JP6272476B2 - 赤血球溶解のための試薬の使用ならびに該試薬に関連する方法およびキット - Google Patents
赤血球溶解のための試薬の使用ならびに該試薬に関連する方法およびキット Download PDFInfo
- Publication number
- JP6272476B2 JP6272476B2 JP2016527179A JP2016527179A JP6272476B2 JP 6272476 B2 JP6272476 B2 JP 6272476B2 JP 2016527179 A JP2016527179 A JP 2016527179A JP 2016527179 A JP2016527179 A JP 2016527179A JP 6272476 B2 JP6272476 B2 JP 6272476B2
- Authority
- JP
- Japan
- Prior art keywords
- cells
- mmol
- reagent
- red blood
- lysis
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 210000003743 erythrocyte Anatomy 0.000 title claims description 120
- 239000003153 chemical reaction reagent Substances 0.000 title claims description 82
- 230000009089 cytolysis Effects 0.000 title claims description 62
- 238000000034 method Methods 0.000 title claims description 46
- 210000004027 cell Anatomy 0.000 claims description 63
- 210000000265 leukocyte Anatomy 0.000 claims description 60
- 210000004369 blood Anatomy 0.000 claims description 53
- 239000008280 blood Substances 0.000 claims description 53
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical group OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 claims description 41
- 239000007995 HEPES buffer Substances 0.000 claims description 36
- 239000002738 chelating agent Substances 0.000 claims description 29
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical group OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 claims description 26
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 claims description 13
- 239000002953 phosphate buffered saline Substances 0.000 claims description 13
- 208000005443 Circulating Neoplastic Cells Diseases 0.000 claims description 10
- 238000001514 detection method Methods 0.000 claims description 10
- 238000004090 dissolution Methods 0.000 claims description 10
- 238000002955 isolation Methods 0.000 claims description 9
- 230000002934 lysing effect Effects 0.000 claims description 8
- 239000001397 quillaja saponaria molina bark Substances 0.000 claims description 5
- 229930182490 saponin Natural products 0.000 claims description 5
- 150000007949 saponins Chemical class 0.000 claims description 5
- 238000011534 incubation Methods 0.000 claims description 4
- 210000002358 circulating endothelial cell Anatomy 0.000 claims description 3
- 239000000306 component Substances 0.000 description 24
- 239000000872 buffer Substances 0.000 description 22
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 18
- 238000011084 recovery Methods 0.000 description 14
- 239000012139 lysis buffer Substances 0.000 description 12
- 239000000203 mixture Substances 0.000 description 12
- 238000012360 testing method Methods 0.000 description 11
- 235000019270 ammonium chloride Nutrition 0.000 description 9
- 239000000243 solution Substances 0.000 description 9
- 206010028980 Neoplasm Diseases 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 150000002500 ions Chemical class 0.000 description 7
- -1 water Chemical compound 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical group N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 239000007864 aqueous solution Substances 0.000 description 6
- 230000003204 osmotic effect Effects 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 5
- 239000010836 blood and blood product Substances 0.000 description 5
- 229940125691 blood product Drugs 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 108090000623 proteins and genes Proteins 0.000 description 5
- 210000000130 stem cell Anatomy 0.000 description 5
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 4
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 4
- 241000124008 Mammalia Species 0.000 description 4
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 4
- 230000002378 acidificating effect Effects 0.000 description 4
- 210000000601 blood cell Anatomy 0.000 description 4
- 239000007853 buffer solution Substances 0.000 description 4
- 238000010790 dilution Methods 0.000 description 4
- 239000012895 dilution Substances 0.000 description 4
- 102000004169 proteins and genes Human genes 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- GLNADSQYFUSGOU-GPTZEZBUSA-J Trypan blue Chemical compound [Na+].[Na+].[Na+].[Na+].C1=C(S([O-])(=O)=O)C=C2C=C(S([O-])(=O)=O)C(/N=N/C3=CC=C(C=C3C)C=3C=C(C(=CC=3)\N=N\C=3C(=CC4=CC(=CC(N)=C4C=3O)S([O-])(=O)=O)S([O-])(=O)=O)C)=C(O)C2=C1N GLNADSQYFUSGOU-GPTZEZBUSA-J 0.000 description 3
- 229910021529 ammonia Inorganic materials 0.000 description 3
- 239000003146 anticoagulant agent Substances 0.000 description 3
- 229940127219 anticoagulant drug Drugs 0.000 description 3
- 238000013459 approach Methods 0.000 description 3
- 201000011510 cancer Diseases 0.000 description 3
- 230000003833 cell viability Effects 0.000 description 3
- 238000005119 centrifugation Methods 0.000 description 3
- 239000012141 concentrate Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- 239000001103 potassium chloride Substances 0.000 description 3
- 235000011164 potassium chloride Nutrition 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 230000035899 viability Effects 0.000 description 3
- FTEDXVNDVHYDQW-UHFFFAOYSA-N BAPTA Chemical compound OC(=O)CN(CC(O)=O)C1=CC=CC=C1OCCOC1=CC=CC=C1N(CC(O)=O)CC(O)=O FTEDXVNDVHYDQW-UHFFFAOYSA-N 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 2
- 102000003846 Carbonic anhydrases Human genes 0.000 description 2
- 108090000209 Carbonic anhydrases Proteins 0.000 description 2
- 102100025064 Cellular tumor antigen p53 Human genes 0.000 description 2
- 108050006400 Cyclin Proteins 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 101000721661 Homo sapiens Cellular tumor antigen p53 Proteins 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- QPCDCPDFJACHGM-UHFFFAOYSA-N N,N-bis{2-[bis(carboxymethyl)amino]ethyl}glycine Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(=O)O)CCN(CC(O)=O)CC(O)=O QPCDCPDFJACHGM-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 2
- 102000040856 WT1 Human genes 0.000 description 2
- 108700020467 WT1 Proteins 0.000 description 2
- 101150084041 WT1 gene Proteins 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000001569 carbon dioxide Substances 0.000 description 2
- 229910002092 carbon dioxide Inorganic materials 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 2
- 230000006037 cell lysis Effects 0.000 description 2
- 239000013522 chelant Substances 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 238000007865 diluting Methods 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 238000010494 dissociation reaction Methods 0.000 description 2
- 230000005593 dissociations Effects 0.000 description 2
- 230000002526 effect on cardiovascular system Effects 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- DEFVIWRASFVYLL-UHFFFAOYSA-N ethylene glycol bis(2-aminoethyl)tetraacetic acid Chemical compound OC(=O)CN(CC(O)=O)CCOCCOCCN(CC(O)=O)CC(O)=O DEFVIWRASFVYLL-UHFFFAOYSA-N 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 239000005556 hormone Substances 0.000 description 2
- 229940088597 hormone Drugs 0.000 description 2
- 230000004054 inflammatory process Effects 0.000 description 2
- 208000032839 leukemia Diseases 0.000 description 2
- 230000002101 lytic effect Effects 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 108020004707 nucleic acids Proteins 0.000 description 2
- 102000039446 nucleic acids Human genes 0.000 description 2
- 150000007523 nucleic acids Chemical class 0.000 description 2
- 230000001991 pathophysiological effect Effects 0.000 description 2
- 229960003330 pentetic acid Drugs 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 239000002562 thickening agent Substances 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- 210000005167 vascular cell Anatomy 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- SMRPGWBDLOQHOS-UHFFFAOYSA-N 5-[4,5-dihydroxy-6-(hydroxymethyl)-3-(3,4,5-trihydroxy-6-methyloxan-2-yl)oxyoxan-2-yl]oxy-3,4-dihydroxy-6-[[9-hydroxy-4-(hydroxymethyl)-4,6a,6b,8a,11,11,14b-heptamethyl-14-oxo-2,3,4a,5,6,7,8,9,10,12,12a,14a-dodecahydro-1H-picen-3-yl]oxy]oxane-2-carboxylic acid Chemical compound OC1C(O)C(O)C(C)OC1OC1C(OC2C(OC(C(O)C2O)C(O)=O)OC2C(C3C(C4C(C5(CCC6(C)C(O)CC(C)(C)CC6C5=CC4=O)C)(C)CC3)(C)CC2)(C)CO)OC(CO)C(O)C1O SMRPGWBDLOQHOS-UHFFFAOYSA-N 0.000 description 1
- LVRVABPNVHYXRT-BQWXUCBYSA-N 52906-92-0 Chemical compound C([C@H](N)C(=O)N[C@H](C(=O)N1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)NCC(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCC(N)=O)C(O)=O)C(C)C)C1=CC=CC=C1 LVRVABPNVHYXRT-BQWXUCBYSA-N 0.000 description 1
- ZKRFOXLVOKTUTA-KQYNXXCUSA-N 9-(5-phosphoribofuranosyl)-6-mercaptopurine Chemical compound O[C@@H]1[C@H](O)[C@@H](COP(O)(O)=O)O[C@H]1N1C(NC=NC2=S)=C2N=C1 ZKRFOXLVOKTUTA-KQYNXXCUSA-N 0.000 description 1
- 102100027308 Apoptosis regulator BAX Human genes 0.000 description 1
- 108050006685 Apoptosis regulator BAX Proteins 0.000 description 1
- 102100021569 Apoptosis regulator Bcl-2 Human genes 0.000 description 1
- 102000036365 BRCA1 Human genes 0.000 description 1
- 108700020463 BRCA1 Proteins 0.000 description 1
- 101150072950 BRCA1 gene Proteins 0.000 description 1
- 102000052609 BRCA2 Human genes 0.000 description 1
- 108700020462 BRCA2 Proteins 0.000 description 1
- 208000012639 Balance disease Diseases 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 101150008921 Brca2 gene Proteins 0.000 description 1
- 102000000905 Cadherin Human genes 0.000 description 1
- 108050007957 Cadherin Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000282832 Camelidae Species 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 102000003952 Caspase 3 Human genes 0.000 description 1
- 108090000397 Caspase 3 Proteins 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 102000016736 Cyclin Human genes 0.000 description 1
- 102000002427 Cyclin B Human genes 0.000 description 1
- 108010068150 Cyclin B Proteins 0.000 description 1
- 102000003910 Cyclin D Human genes 0.000 description 1
- 108090000259 Cyclin D Proteins 0.000 description 1
- 102000003909 Cyclin E Human genes 0.000 description 1
- 108090000257 Cyclin E Proteins 0.000 description 1
- 102100034157 DNA mismatch repair protein Msh2 Human genes 0.000 description 1
- 102000012199 E3 ubiquitin-protein ligase Mdm2 Human genes 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 108010014173 Factor X Proteins 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 102100024785 Fibroblast growth factor 2 Human genes 0.000 description 1
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 1
- 101710088083 Glomulin Proteins 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 102000001398 Granzyme Human genes 0.000 description 1
- 108060005986 Granzyme Proteins 0.000 description 1
- 206010018910 Haemolysis Diseases 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 101000971171 Homo sapiens Apoptosis regulator Bcl-2 Proteins 0.000 description 1
- 101001134036 Homo sapiens DNA mismatch repair protein Msh2 Proteins 0.000 description 1
- 101001034314 Homo sapiens Lactadherin Proteins 0.000 description 1
- 101001005719 Homo sapiens Melanoma-associated antigen 3 Proteins 0.000 description 1
- 101000669513 Homo sapiens Metalloproteinase inhibitor 1 Proteins 0.000 description 1
- 101000835893 Homo sapiens Mothers against decapentaplegic homolog 4 Proteins 0.000 description 1
- 101001133056 Homo sapiens Mucin-1 Proteins 0.000 description 1
- 101000642478 Homo sapiens Serpin B3 Proteins 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 102100039648 Lactadherin Human genes 0.000 description 1
- 229910015837 MSH2 Inorganic materials 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 102000002274 Matrix Metalloproteinases Human genes 0.000 description 1
- 108010000684 Matrix Metalloproteinases Proteins 0.000 description 1
- 101150094768 Mcam gene Proteins 0.000 description 1
- 102100025082 Melanoma-associated antigen 3 Human genes 0.000 description 1
- 102100039364 Metalloproteinase inhibitor 1 Human genes 0.000 description 1
- 208000032818 Microsatellite Instability Diseases 0.000 description 1
- 102100025725 Mothers against decapentaplegic homolog 4 Human genes 0.000 description 1
- 102000002419 Motilin Human genes 0.000 description 1
- 101800002372 Motilin Proteins 0.000 description 1
- 102100034256 Mucin-1 Human genes 0.000 description 1
- 108010026664 MutL Protein Homolog 1 Proteins 0.000 description 1
- 102000013609 MutL Protein Homolog 1 Human genes 0.000 description 1
- 101710135898 Myc proto-oncogene protein Proteins 0.000 description 1
- 102100038895 Myc proto-oncogene protein Human genes 0.000 description 1
- 108700020796 Oncogene Proteins 0.000 description 1
- 102000043276 Oncogene Human genes 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 102000004503 Perforin Human genes 0.000 description 1
- 108010056995 Perforin Proteins 0.000 description 1
- KHGNFPUMBJSZSM-UHFFFAOYSA-N Perforine Natural products COC1=C2CCC(O)C(CCC(C)(C)O)(OC)C2=NC2=C1C=CO2 KHGNFPUMBJSZSM-UHFFFAOYSA-N 0.000 description 1
- 102100021768 Phosphoserine aminotransferase Human genes 0.000 description 1
- 102100036691 Proliferating cell nuclear antigen Human genes 0.000 description 1
- 102100023832 Prolyl endopeptidase FAP Human genes 0.000 description 1
- 108010072866 Prostate-Specific Antigen Proteins 0.000 description 1
- 108010029485 Protein Isoforms Proteins 0.000 description 1
- 102000001708 Protein Isoforms Human genes 0.000 description 1
- 102000007568 Proto-Oncogene Proteins c-fos Human genes 0.000 description 1
- 108010071563 Proto-Oncogene Proteins c-fos Proteins 0.000 description 1
- FCLZCOCSZQNREK-UHFFFAOYSA-N Pyrrolidine, hydrochloride Chemical compound Cl.C1CCNC1 FCLZCOCSZQNREK-UHFFFAOYSA-N 0.000 description 1
- 208000007660 Residual Neoplasm Diseases 0.000 description 1
- 108010005173 SERPIN-B5 Proteins 0.000 description 1
- 102100031770 SH2B adapter protein 1 Human genes 0.000 description 1
- 108050003189 SH2B adapter protein 1 Proteins 0.000 description 1
- 102000003800 Selectins Human genes 0.000 description 1
- 108090000184 Selectins Proteins 0.000 description 1
- 102100036383 Serpin B3 Human genes 0.000 description 1
- 102100030333 Serpin B5 Human genes 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- 101710150448 Transcriptional regulator Myc Proteins 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 239000013504 Triton X-100 Substances 0.000 description 1
- 229920004890 Triton X-100 Polymers 0.000 description 1
- 102000044209 Tumor Suppressor Genes Human genes 0.000 description 1
- 108700025716 Tumor Suppressor Genes Proteins 0.000 description 1
- 102100039094 Tyrosinase Human genes 0.000 description 1
- 108060008724 Tyrosinase Proteins 0.000 description 1
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 1
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000001464 adherent effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 230000001640 apoptogenic effect Effects 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 238000001574 biopsy Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 239000012503 blood component Substances 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 230000003139 buffering effect Effects 0.000 description 1
- 125000005587 carbonate group Chemical group 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 125000002843 carboxylic acid group Chemical group 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 230000022131 cell cycle Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 210000005266 circulating tumour cell Anatomy 0.000 description 1
- 238000005352 clarification Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000004925 denaturation Methods 0.000 description 1
- 230000036425 denaturation Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 238000002405 diagnostic procedure Methods 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 238000006911 enzymatic reaction Methods 0.000 description 1
- 210000003979 eosinophil Anatomy 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 210000003608 fece Anatomy 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000000684 flow cytometry Methods 0.000 description 1
- 210000000245 forearm Anatomy 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 238000003205 genotyping method Methods 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 210000003714 granulocyte Anatomy 0.000 description 1
- 230000008588 hemolysis Effects 0.000 description 1
- 230000023597 hemostasis Effects 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 102000006495 integrins Human genes 0.000 description 1
- 108010044426 integrins Proteins 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 210000004324 lymphatic system Anatomy 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 101150024228 mdm2 gene Proteins 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 230000001394 metastastic effect Effects 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 230000000921 morphogenic effect Effects 0.000 description 1
- 230000004899 motility Effects 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- MGFYIUFZLHCRTH-UHFFFAOYSA-N nitrilotriacetic acid Chemical compound OC(=O)CN(CC(O)=O)CC(O)=O MGFYIUFZLHCRTH-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 210000004882 non-tumor cell Anatomy 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 229910052755 nonmetal Inorganic materials 0.000 description 1
- 229930192851 perforin Natural products 0.000 description 1
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 235000015497 potassium bicarbonate Nutrition 0.000 description 1
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 1
- 239000011736 potassium bicarbonate Substances 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- AAEVYOVXGOFMJO-UHFFFAOYSA-N prometryn Chemical compound CSC1=NC(NC(C)C)=NC(NC(C)C)=N1 AAEVYOVXGOFMJO-UHFFFAOYSA-N 0.000 description 1
- 208000015445 pyruvate dehydrogenase deficiency Diseases 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- PPRSVUXPYPBULA-UHFFFAOYSA-N saponin A Natural products CC1(C)CCC2(CCC3(C)C(=CCC4C5(C)CCC(OC6OC(CO)C(O)C(O)C6=O)C(C)(C)C5CCC34C)C2C1)C(=O)O PPRSVUXPYPBULA-UHFFFAOYSA-N 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 238000004062 sedimentation Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 238000005563 spheronization Methods 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 150000003512 tertiary amines Chemical group 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 239000003634 thrombocyte concentrate Substances 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5094—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for blood cell populations
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/574—Immunoassay; Biospecific binding assay; Materials therefor for cancer
Description
塩化アンモニウムは、全血中の赤血球の溶解を可能にする、浸透性塩として記載された。古典的な塩化アンモニウム溶解緩衝液は、150mM NH4Cl、1mM KHCO3および0.1mM EDTAを含有する。赤血球は、この緩衝液を用いて、定量的に枯渇させることも可能であるが、約30%またはそれより多くの白血球が失われる(例えば、Meryman H. Red Cell Structure and Function 1969; 352−367、Sass M. Am J Physiol. 1979;236(5):C238−43、Claus R.ら Folia Hematol. 1985; 5: 683−688、Terstappenら J Immun Methods. 1989: 103−112を参照されたい)。
− 2.5mmol/l〜12mmol/l HEPES、
− 60mmol/l〜120mmol/l NH4 +/NH3、
− 0.04mmol/l〜0.8mmol/lキレート剤、および
− 存在する場合、0.15mmol/l〜0.8mmol/l CO3 2-/CO3 -
の範囲である、前記試薬によって解決された。
− 2.5mmol/l〜12mmol/l HEPES、
− 60mmol/l〜120mmol/l NH4 +/NH3、
− 0.04mmol/l〜0.8mmol/lキレート剤、および
− 存在する場合、0.15mmol/l〜0.8mmol/l CO3 2-/CO3 -
の範囲である、前記試薬の使用に関する。
− 3mmol/l〜11mmol/l HEPES、好ましくは、3mmol/l〜10mmol/l HEPES、より好ましくは、3.5mmol/l〜4.5mmol/l HEPES、
− 70mmol/l〜100mmol/l NH4 +/NH3、好ましくは75mmol/l〜85mmol/l NH4 +/NH3、
− 0.05mmol/l〜0.5mmol/lキレート剤、好ましくは0.06mmol/l〜0.2mmol/lキレート剤、より好ましくは0.07mmol/l〜0.1mmol/lキレート剤、および/または
− 存在する場合、0.3mmol/l〜0.6mmol/l CO3 2-/CO3 -、好ましくは0.3mmol/l〜0.5mmol/l CO3 2-/CO3 -、より好ましくは0.35mmol/l〜0.45mmol/l CO3 2-/CO3 -
の範囲の最終濃度中で存在する。
特定の態様において、赤血球または赤血球を含有する試料は、哺乳動物から、特に哺乳動物家畜動物、例えばネコ、イヌ、ウサギ、またはモルモット、あるいは農場動物、例えばウシ、ウマ、ヤギ、ヒツジ、ブタまたはラクダから得られる。非常に特定の態様において、赤血球または赤血球を含有する試料は、ヒトから得られる。
a)赤血球を含む試料を提供し;
b)請求項1〜5のいずれかに定義するような試薬と試料をインキュベーションし、それによって赤血球を溶解し;そして
c)赤血球破片を除去してもよい
工程を含む、前記方法を提供する。
d)上に記載するように、赤血球を含む試料から、特に血液試料から、赤血球以外の細胞を検出するかまたは単離する
工程をさらに含む。
好ましくは、赤血球以外の細胞は、白血球または循環腫瘍細胞、特に循環腫瘍細胞である(上記の詳細もまた参照されたい)。
第三の側面において、本開示は
− 本明細書に開示するような使用および方法の文脈において上に定義するような赤血球溶解のための試薬;および
− 赤血球破片を除去するための試薬;および
− 本明細書に開示するような任意の方法を実行するためのあってもよい使用説明書
を含む、赤血球を含む試料から白血球を単離するためのキットに関する。
別に示さない限り、全血の1部分を、溶解緩衝液の部分(以下に定義する通り)と混合し、室温で10分間インキュベーションし、そして300xgで15分間遠心分離する。上清を廃棄し、そして細胞ペレットを4部分の溶解緩衝液に再懸濁し、そして300xgで15分間遠心分離する。上清を廃棄し、そして細胞ペレットを、0.3mM EDTAを含有するPBS 4部分に再懸濁し、そして300xgで15分間遠心分離する。上清を廃棄し、そして細胞ペレットを、0.3mM EDTAを含有するPBSの別個の量に再懸濁する。
最初の試験において、上記プロトコルにしたがって、慣用的製品の適切さを試験した。以下の慣用的製品を用いた。EasySep(登録商標)赤血球溶解緩衝液(StemCell Technologies、カタログ番号20110)、HetaSep(登録商標)(StemCell Technologies、カタログ番号07806)、Stromatolyser NR溶解(Sysmex、カタログ番号SNR−200、SNR−210A)。
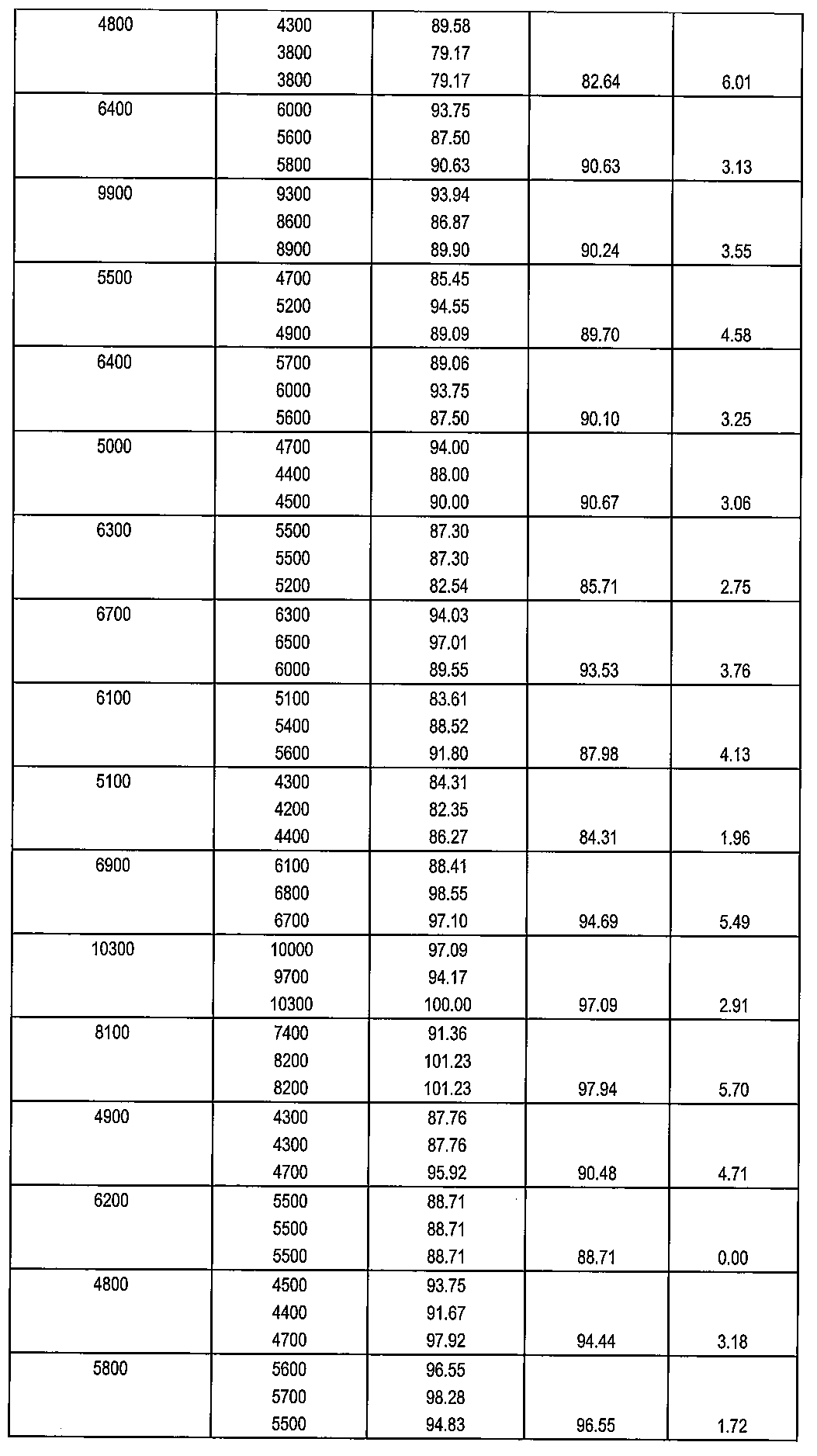
表1:白血球(WBC)の回収率に対する赤血球溶解のための慣用的製品の影響
実施例2:NH4Clを含まない試薬中での赤血球溶解
第二の試験において、上記プロトコルにしたがって、NH4Clを含まない試薬の適切さを試験した。結果を以下の表2に示す:
表2:白血球(WBC)の回収に対するNH4Clを含まない試薬の影響
実施例3:NH4Clを含む、異なる試薬中での赤血球溶解
第三の試験において、上記プロトコルにしたがって、NH4Clを含む、異なる試薬の適切さを試験した。結果を以下の表3に示す:
表3:白血球(WBC)の回収に対する、NH4Clを含む異なる試薬の影響
第四の試験において、上記プロトコルにしたがって、NH4ClおよびHEPESを含む、異なる試薬の適切さを試験した。結果を以下の表4に示す:
表4:白血球(WBC)の回収に対する、NH4ClおよびHEPESを含む、異なる試薬の影響
第五の試験において、上記プロトコルにしたがって、80mM NH4Cl+10mM Hepes+0.1mM EDTAを含む試薬の適切さを試験した。WBCおよびRBCの回収に加えて、回収されたWBCの生存性をトリパンブルー排除試験によって測定した。したがって、WBCを、トリパンブルー(Sigma、カタログ番号T8154−20ML)(希釈WBC懸濁物:トリパンブルー=1:2)で染色し、そしてC−Chip計数チャンバー(Biochrom、カタログ番号P DHC−N01)を用いて、染色された細胞(死んだ細胞)を計数した。結果を以下の表5に示す:
表5:白血球(WBC)および赤血球(RBC)の回収および生存度に対する試薬の影響
実施例6:NH4ClおよびHEPESを含む、異なる試薬中での赤血球溶解
第六の試験において、上記プロトコルにしたがって、NH4ClおよびHEPESを含む、異なる試薬の適切さを試験した。結果を以下の表6に示す:
表6:白血球(WBC)の回収および溶解後のpH値に対する試薬組成の影響
第七の試験において、現存する赤血球溶解プロトコルの有効性を、示すように試験した。結果を以下の表7に示す:
表7:白血球の回収に対する、慣用的赤血球溶解プロトコルの影響
Claims (15)
- 赤血球溶解のための試薬の使用であって、試薬がHEPES(4−(2−ヒドロキシエチル)−1−ピペラジンエタンスルホン酸)、NH4 +/NH3、キレート剤およびあってもよいCO3 2−/CO3 −を含むか、またはこれらからなり、赤血球溶解中の最終濃度が
− 2.5mmol/l〜12mmol/l HEPES、
− 60mmol/l〜120mmol/l NH4 +/NH3、
− 0.04mmol/l〜0.8mmol/l キレート剤、および
− 存在する場合、0.15mmol/l〜0.8mmol/l CO3 2−/CO3 −
の範囲であって、
当該試薬はサポニンを含まない、前記試薬の使用。 - 溶解中の最終濃度が
− 3mmol/l〜11mmol/l HEPES、
− 70mmol/l〜100mmol/l NH4 +/NH3、
− 0.05mmol/l〜0.5mmol/l キレート剤、および/または
− 存在する場合、0.3mmol/l〜0.6mmol/l CO3 2−/CO3 −
の範囲である、請求項1の使用。 - キレート剤がエチレンジアミン四酢酸(EDTA)である、請求項1または2の使用。
- 試薬のpHが、6.4〜7.7の範囲である、請求項1〜3のいずれか一項の使用。
- 赤血球溶解中、pHが、6.4〜7.7の範囲に維持される、請求項4の使用。
- 赤血球を含む試料から、赤血球以外の細胞の検出、濃縮、または単離において前記試薬を用いる、請求項1〜5のいずれか一項の使用。
- 赤血球以外の細胞が、白血球、循環内皮細胞、または循環腫瘍細胞である、請求項6の使用。
- 赤血球を溶解する方法であって
a)赤血球を含む試料を提供し;
b)請求項1〜5のいずれか一項に定義される試薬と該試料をインキュベーションし、それによって赤血球を溶解し;そして
c)赤血球破片を除去してもよい
工程を含む、前記方法。 - 前記試料が、血液試料または赤血球および他の細胞を含む試料である、請求項8の方法。
- d)赤血球を含む試料から、赤血球以外の細胞を検出するかまたは単離する
工程をさらに含む、請求項8または9の方法。 - 赤血球以外の細胞が、白血球または循環腫瘍細胞である、請求項10の方法。
- 工程b)のインキュベーションが、最大30分間である、請求項8〜11のいずれか一項の方法。
- − 請求項1〜5のいずれか一項に定義される赤血球溶解のための試薬;および
− 赤血球破片を除去するための試薬
を含む、赤血球を含む試料から白血球を単離するためのキット。 - − 請求項8〜12のいずれか一項の方法を実行するための使用説明書
をさらに含む、請求項13のキット。 - 赤血球破片を除去するための試薬が、キレート剤を含むリン酸緩衝生理食塩水(PBS)である、請求項13または14のキット。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP13005219 | 2013-11-05 | ||
| EP13005219.4 | 2013-11-05 | ||
| PCT/EP2014/073519 WO2015067546A1 (en) | 2013-11-05 | 2014-11-03 | Use of a reagent for the lysis of erythrocytes as well as methods and kits relating thereto |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2016536586A JP2016536586A (ja) | 2016-11-24 |
| JP6272476B2 true JP6272476B2 (ja) | 2018-01-31 |
Family
ID=49546227
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016527179A Active JP6272476B2 (ja) | 2013-11-05 | 2014-11-03 | 赤血球溶解のための試薬の使用ならびに該試薬に関連する方法およびキット |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US20160252498A1 (ja) |
| EP (1) | EP3066467B1 (ja) |
| JP (1) | JP6272476B2 (ja) |
| CN (1) | CN105683753B (ja) |
| CA (1) | CA2926304C (ja) |
| ES (1) | ES2913337T3 (ja) |
| HK (1) | HK1225798B (ja) |
| WO (1) | WO2015067546A1 (ja) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110770587B (zh) * | 2017-05-08 | 2023-10-20 | 拜克门寇尔特公司 | 用于裂解红细胞的组合物和方法 |
| CN108279145A (zh) * | 2017-12-19 | 2018-07-13 | 中国医学科学院医学生物学研究所 | 树鼩细胞免疫表型分析中外周血红细胞裂解方法 |
| CN109632734A (zh) * | 2018-11-30 | 2019-04-16 | 张丽英 | 一种利用atp生物发光反应检测食品中黄曲霉菌的方法 |
| CN109655607B (zh) * | 2019-01-25 | 2021-02-23 | 广东菲鹏生物有限公司 | 红细胞裂解试剂及其应用 |
| US11623034B2 (en) | 2020-01-03 | 2023-04-11 | Fenwal, Inc. | System and method to lyse and remove red blood cells from a cell product |
| CN111781042B (zh) * | 2020-07-08 | 2023-07-07 | 青海省畜牧兽医科学院 | 一种附红细胞体检测试剂盒及样品处理方法 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5702884A (en) * | 1996-03-12 | 1997-12-30 | Johnson & Johnson Clinical Diagnostics, Inc. | Whole blood sample preparation for polymerase chain reaction using ammonium chloride and a carboxylic acid or metal carboxylate for selective red blood cell lysis |
| FR2813891B1 (fr) * | 2000-09-14 | 2005-01-14 | Immunotech Sa | Reactif multifonctionnel pour erythrocytes mettant en jeu des carbamates et applications |
| JP2002181809A (ja) * | 2000-12-15 | 2002-06-26 | Asahi Medical Co Ltd | 血液処理剤、それによる血液処理方法及び白血球分析方法 |
| DE10242016A1 (de) * | 2002-09-11 | 2004-03-25 | Esplora GmbH c/o TU Darmstadt Institut für Biochemie | Verfahren zur Identifizierung BHS-spezifischer Proteine und Fragmente davon |
| CN1419120A (zh) * | 2002-12-18 | 2003-05-21 | 北京市桑浩博科技发展有限公司 | 血红蛋白目测比色装置 |
| CN101327324A (zh) * | 2007-06-21 | 2008-12-24 | 中国科学院上海生命科学研究院 | 治疗自身免疫疾病的方法和组合物 |
-
2014
- 2014-11-03 ES ES14792815T patent/ES2913337T3/es active Active
- 2014-11-03 EP EP14792815.4A patent/EP3066467B1/en active Active
- 2014-11-03 WO PCT/EP2014/073519 patent/WO2015067546A1/en active Application Filing
- 2014-11-03 JP JP2016527179A patent/JP6272476B2/ja active Active
- 2014-11-03 CA CA2926304A patent/CA2926304C/en active Active
- 2014-11-03 CN CN201480060814.2A patent/CN105683753B/zh active Active
-
2016
- 2016-05-04 US US15/146,273 patent/US20160252498A1/en not_active Abandoned
- 2016-12-09 HK HK16114040A patent/HK1225798B/zh unknown
-
2019
- 2019-12-20 US US16/722,193 patent/US20200141929A1/en active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| HK1225798B (zh) | 2017-09-15 |
| EP3066467B1 (en) | 2022-03-16 |
| ES2913337T3 (es) | 2022-06-01 |
| CA2926304A1 (en) | 2015-05-14 |
| CN105683753B (zh) | 2018-04-06 |
| CN105683753A (zh) | 2016-06-15 |
| WO2015067546A1 (en) | 2015-05-14 |
| US20160252498A1 (en) | 2016-09-01 |
| EP3066467A1 (en) | 2016-09-14 |
| CA2926304C (en) | 2020-04-07 |
| US20200141929A1 (en) | 2020-05-07 |
| JP2016536586A (ja) | 2016-11-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6272476B2 (ja) | 赤血球溶解のための試薬の使用ならびに該試薬に関連する方法およびキット | |
| JP7278987B2 (ja) | 全血サンプルの安定化 | |
| US9671321B2 (en) | Methods and compositions for exosome isolation | |
| JP6179508B2 (ja) | 希少細胞の検出方法 | |
| JP2019013250A (ja) | 生物学的試料の安定化 | |
| KR20040053115A (ko) | 분석용 세포 및 생물학적 시험편의 안정화 | |
| US20150132738A1 (en) | Method For Identification Of Non-Hematogeneous Karocytes Enriched From Body Fluid Of Humans Or Animals | |
| JP6545198B2 (ja) | 周囲温度での血液サンプル中の代謝的に活性な細胞の安定化 | |
| US20230131940A1 (en) | Methods of enumerating particles present in a cell composition | |
| TW201439515A (zh) | 用於高嚴苛度細胞處理及抗原修復之組成物 | |
| ES2846224T3 (es) | Enriquecimiento de células tumorales circulantes agotando glóbulos blancos | |
| KR20210144848A (ko) | 세포 외 소포의 보존 안정화제 및 보존 안정화 방법 | |
| JP6617495B2 (ja) | 腫瘍細胞の検出方法 | |
| JP2020030180A (ja) | 血液試料の溶血剤 | |
| CA3067946A1 (en) | Process and composition for the stabilization of cell-free nucleic acids and cells | |
| Hinson | Advances in Transfusion Medicine for Veterinary Patients | |
| WO2018220601A1 (en) | Sample storage system and methods of use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170222 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20170222 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170517 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170816 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170822 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20171204 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20171228 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6272476 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |