JP5643092B2 - コンタクトレンズのパッケージング溶液 - Google Patents
コンタクトレンズのパッケージング溶液 Download PDFInfo
- Publication number
- JP5643092B2 JP5643092B2 JP2010522932A JP2010522932A JP5643092B2 JP 5643092 B2 JP5643092 B2 JP 5643092B2 JP 2010522932 A JP2010522932 A JP 2010522932A JP 2010522932 A JP2010522932 A JP 2010522932A JP 5643092 B2 JP5643092 B2 JP 5643092B2
- Authority
- JP
- Japan
- Prior art keywords
- packaging solution
- contact lens
- solution
- lens
- packaging
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65B—MACHINES, APPARATUS OR DEVICES FOR, OR METHODS OF, PACKAGING ARTICLES OR MATERIALS; UNPACKING
- B65B25/00—Packaging other articles presenting special problems
- B65B25/008—Packaging other articles presenting special problems packaging of contact lenses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L12/00—Methods or apparatus for disinfecting or sterilising contact lenses; Accessories therefor
- A61L12/08—Methods or apparatus for disinfecting or sterilising contact lenses; Accessories therefor using chemical substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L12/00—Methods or apparatus for disinfecting or sterilising contact lenses; Accessories therefor
- A61L12/02—Methods or apparatus for disinfecting or sterilising contact lenses; Accessories therefor using physical phenomena, e.g. electricity, ultrasonics or ultrafiltration
- A61L12/04—Heat
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B1/00—Optical elements characterised by the material of which they are made; Optical coatings for optical elements
- G02B1/04—Optical elements characterised by the material of which they are made; Optical coatings for optical elements made of organic materials, e.g. plastics
- G02B1/041—Lenses
- G02B1/043—Contact lenses
-
- G—PHYSICS
- G02—OPTICS
- G02C—SPECTACLES; SUNGLASSES OR GOGGLES INSOFAR AS THEY HAVE THE SAME FEATURES AS SPECTACLES; CONTACT LENSES
- G02C7/00—Optical parts
- G02C7/02—Lenses; Lens systems ; Methods of designing lenses
- G02C7/04—Contact lenses for the eyes
Description
断りない限り、本明細書で使用されるすべての専門用語は、本発明が属する技術分野の当業者によって一般に理解されるものと同じ意味を有する。一般に、本明細書で使用される術語及び実験手順は当該技術で周知であり、一般に使用されている。これらの手順には、当該技術及び種々の一般的参考文献で提供されているような従来法が使用される。ある語が単数形で記載されている場合、本発明者らはその語の複数形をも考慮している。本明細書で使用される術語及び以下に記載する実験手順は、当該技術で周知であり、一般に使用されているものである。本開示を通して使用される以下の用語は、断りない限り、以下の意味を有するものと理解されなければならない。
a.本明細書で使用されるポリマー材料(モノマー又はマクロマー材料を含む)の「分子量」とは、断りない限り、又は試験条件がそうではないことを指示しない限り、数平均分子量をいう。
b.レンズに関していう「視認用色付け」とは、装用者がレンズ貯蔵、消毒又は洗浄パッケージ内の透明な溶液中のレンズを容易に見つけることを可能にするためのレンズの染色(又は着色)をいう。レンズの視認用色付けに染料及び/又は顔料を使用することができるということは当該技術で周知である。
レンズパッケージング溶液
コポリマー845をISPから、PEG400(Sentry Carbowax 400)をDowから入手した。表1に示すように1Lの水中で様々な成分を溶解させることによって、溶液を調整した。ここで、HECは成分の一つとして用いられ、溶液はHECを溶解させるために約80℃で調製した。
Advancedマイクロオスモメーターモデル3300を各パッケージング溶液のオスモル濃度を決定するために用いた(各溶液に対し2つの独立した実験結果を平均した)。パッケージング溶液の粘度は、Brookfield Viscometerを用いて、異なる回転速度(3rpm、6rpm及び12rpm)での3回の測定の平均によって得た。各パッケージング溶液のpH値は、Fisher Acumet 25 pH計(各溶液に対し2つの独立した実験結果を平均した)を用いた。結果を表2にまとめた。
細胞毒性評価
実施例1で調製した各パッケージング溶液についての、24時間暴露時での細胞毒性の試験をL−929(L929)マウス繊維芽細胞を用いて行った。アラマーブルー試験(AB)並びにニュートラルレッド取り込み及び放出試験(NRUR)を溶液の細胞毒性評価に用いた。96Wellプレート型中にて試験サンプルの溶液を1:1、1:3及び1:7に希釈し(最終的な溶液濃度は50、25、12.5%)細胞単層を24時間曝露した。この試験では、、対照と比較しての、初期の曝露及び試験溶液を用いてのインキュベーション後の制御された環境での成長した細胞を、生存及び成長力についてモニターした。10ppmにおけるBAKを陽性対照として用いた。
溶液調製
種々の溶液を、表3に示すようにリン酸緩衝生理食塩水中で種々の成分を溶解させることで調製した。各溶液はHPMCを溶解させるために約80℃で調製した。濃度、pH及びオスモル濃度を、実施例1に記載の方法によって測定し、結果を表3にまとめた。
O2OPTIX(商標)レンズを、パッケージング溶液(Y1,Y2、Y3、及びY4の一つ)を含むブリスタパッケージにパッケージングし、封止し、オートクレーブ処理を行った。
緩衝生理食塩水(対照)、Y1、Y2、Y3又はY4のパッケージング溶液中に包装されたレンズの、イオン透過性、酸素透過性、反射指数、光透過率、水分含有量、弾性率に有意な変化はみられない。試験の行われた溶液に包装された全てのレンズは細胞毒性試験に合格した。本発明におけるパッケージング溶液中に包装されたレンズの、水の接触角(3つのレンズに対する計測の平均)、脂質取り込み、及びタンパク質取り込みを表にまとめた。
市販されているNight&Dayレンズの他にAcuvue Advance及びO2Optixをパッケージング食塩水から取り出し、パッケージング溶液(下記)で3度すすいだ後、パッケージング溶液に一晩浸した。翌日にレンズを10μg/mLのホスホチジルエタノールアミンを加えた新しい24wellプレートに置き、再度一晩浸した。24時間後レンズを34.5℃のインキュベーション及びアルミニウムホイルから動かし、緩衝液を3度交換し、1mLのリン酸緩衝生理食塩水(PBS)を加えた新しい24wellプレートに置き、Victor II Wallacにて測定した。
1%アミノコート
PBS中0.25%HEC
0.5%コポリマー845
PBS中2%PVP
PBS中0.1%HEC及び1%PVP
PBS中1%PEG400及び2%PVP
PBS中1%PEG400及び2%PVA
PBS中0.5%PEG400及び1%K90PVP
PBS中0.5%PEG400及び2%K90PVP
PBS中0.25%PEG400及び1%K90PVP
PBS中0.25%PEG400及び2%K90PVP
PBS中0.5%コポリマー845及び0.5%アミノコート
PBS中0.5%コポリマー845及び0.5%PEG400
PBS中0.1%ヒドロキシプロピルグァーガム
対照標準PBS
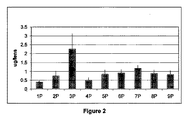
パッケージング溶液(下記)を、吸収生産工程(imbibed production process)(プラズマ処理後の乾燥レンズのための水和溶液としてパッケージング溶液を使用することによって、レンズにパッケージング溶液を「吸収」させた工程)及びレンズの湿潤性試験にて用いた。吸収生産工程において、パッケージング溶液はまた、一杯の生理食塩水(すなわち、レンズパッケージ内に)のために用いられた。
1P.生理食塩水パッケージングに2%コリドンK90を添加
2P.生理食塩水パッケージングに1%コポリマー845を添加
3P.標準生産ラインの生理食塩水中に包装されたNight&Day対照レンズ
4P.プラズマ処理を施した後、2%コリドンK90に浸して包装された乾燥レンズ
5P.プラズマ処理を施した後、1%コポリマー845に浸して包装された乾燥レンズ
6P.生理食塩水パッケージに0.25%PEG4000/1%コリドンK90を添加
7P.生理食塩水パッケージに0.5%コポリマー845/0.5%PEG400を添加
8P.プラズマ処理を施した後、0.25%PEG4000/1%コリドンK90に浸して包装された乾燥レンズ
9P.プラズマ処理を施した後、0.5%コポリマー845/0.5%PEG400に浸して包装された乾燥レンズ
Claims (8)
- パッケージング溶液及びパッケージング溶液に浸漬されたシリコーンヒドロゲルコンタクトレンズを含む、封止され、滅菌されたパッケージを含む、眼科用製品であって、
ここで、パッケージング溶液は、
ビニルピロリドンとジメチルアミノエチルメタクリレートとのコポリマー;
600以下の平均分子量を有するポリエチレングリコール;
α−オキソ多酸又はその塩;及び
0.001重量%〜2重量%の一つ以上の緩衝剤を含み、
パッケージング溶液は、200〜450mOsm/kgのオスモル濃度を有し、パッケージング溶液は、25℃で1.2〜2.5センチポアズの粘度を有し、6.0〜8.0のpHを有し、そして、パッケージング溶液中のギ酸の量が、α−オキソ多酸又はその塩なしの溶液中で検出される量の80%以下である、眼科用製品。 - α−オキソ多酸が、クエン酸、2−ケトグルタル酸及びリンゴ酸からなる群より選択される、請求項1記載の眼科用製品。
- α−オキソ多酸が、クエン酸である、請求項1又は2記載の眼科用製品。
- ヒドロゲルコンタクトレンズが、その中に一つ以上の浸出可能な潤滑剤を含む、請求項1〜3いずれか1項記載の眼科用製品。
- 装用者の初期不快感を緩和しうる、ソフトコンタクトレンズを製造する方法であって、
a)パッケージング溶液を含む容器に、シリコーンヒドロゲルコンタクトレンズをパッケージングすることであって、ここで
パッケージング溶液は、ビニルピロリドンとジメチルアミノエチルメタクリレートとのコポリマーを含み、
パッケージング溶液は、さらに、600以下の平均分子量を有するポリエチレングリコール、α−オキソ多酸又はその塩、及び、0.001重量%〜2重量%の一つ以上の緩衝剤を含み、
パッケージング溶液は、200〜450mOsm/kgのオスモル濃度を有し、パッケージング溶液は、25℃で1.2〜2.5センチポアズの粘度を有し、パッケージング溶液は、6.0〜8.0のpHを有し、そして、パッケージング溶液中のギ酸の量が、α−オキソ多酸又はその塩なしの溶液中で検出される量の80%以下であること:ならびに
b)パッケージング中のシリコーンヒドロゲルコンタクトレンズを滅菌してソフトコンタクトレンズを得ること
により、シリコーンヒドロゲルコンタクトレンズに、湿潤性の増加、摩擦の減少、レンズのクッションによる初期のコンディショニング、及び/又はレンズ上への沈着物の付着性の減少を提供する工程を含む方法。 - α−オキソ多酸が、クエン酸、2−ケトグルタル酸及びリンゴ酸からなる群より選択される、請求項5記載の方法。
- α−オキソ多酸が、クエン酸である、請求項6記載の方法。
- シリコーンヒドロゲルコンタクトレンズが、その中に一つ以上の浸出可能な潤滑剤を含む、請求項5〜7いずれか1項記載の方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US96933707P | 2007-08-31 | 2007-08-31 | |
| US60/969,337 | 2007-08-31 | ||
| PCT/US2008/010146 WO2009032122A1 (en) | 2007-08-31 | 2008-08-27 | Contact lens packaging solutions |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2014151165A Division JP5821140B2 (ja) | 2007-08-31 | 2014-07-24 | コンタクトレンズのパッケージング溶液 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2010538321A JP2010538321A (ja) | 2010-12-09 |
| JP2010538321A5 JP2010538321A5 (ja) | 2011-07-21 |
| JP5643092B2 true JP5643092B2 (ja) | 2014-12-17 |
Family
ID=40260610
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2010522932A Expired - Fee Related JP5643092B2 (ja) | 2007-08-31 | 2008-08-27 | コンタクトレンズのパッケージング溶液 |
| JP2014151165A Expired - Fee Related JP5821140B2 (ja) | 2007-08-31 | 2014-07-24 | コンタクトレンズのパッケージング溶液 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2014151165A Expired - Fee Related JP5821140B2 (ja) | 2007-08-31 | 2014-07-24 | コンタクトレンズのパッケージング溶液 |
Country Status (7)
| Country | Link |
|---|---|
| US (2) | US8689971B2 (ja) |
| EP (1) | EP2188655B1 (ja) |
| JP (2) | JP5643092B2 (ja) |
| KR (1) | KR101535692B1 (ja) |
| AT (1) | ATE540327T1 (ja) |
| CA (1) | CA2692831C (ja) |
| WO (1) | WO2009032122A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014222362A (ja) * | 2007-08-31 | 2014-11-27 | ノバルティス アーゲー | コンタクトレンズのパッケージング溶液 |
Families Citing this family (60)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090173045A1 (en) * | 2008-01-09 | 2009-07-09 | Yu-Chin Lai | Packaging Solutions |
| US20110114517A1 (en) * | 2009-11-17 | 2011-05-19 | Kasey Jon Minick | Hydrogen peroxide solution and kit for disinfecting contact lenses |
| KR102411923B1 (ko) | 2010-07-30 | 2022-06-22 | 알콘 인코포레이티드 | 수분이 풍부한 표면을 갖는 실리콘 히드로겔 렌즈 |
| US8798332B2 (en) | 2012-05-15 | 2014-08-05 | Google Inc. | Contact lenses |
| US9158133B1 (en) | 2012-07-26 | 2015-10-13 | Google Inc. | Contact lens employing optical signals for power and/or communication |
| US8857981B2 (en) | 2012-07-26 | 2014-10-14 | Google Inc. | Facilitation of contact lenses with capacitive sensors |
| US9523865B2 (en) | 2012-07-26 | 2016-12-20 | Verily Life Sciences Llc | Contact lenses with hybrid power sources |
| US9298020B1 (en) | 2012-07-26 | 2016-03-29 | Verily Life Sciences Llc | Input system |
| US8919953B1 (en) | 2012-08-02 | 2014-12-30 | Google Inc. | Actuatable contact lenses |
| US9696564B1 (en) | 2012-08-21 | 2017-07-04 | Verily Life Sciences Llc | Contact lens with metal portion and polymer layer having indentations |
| US8971978B2 (en) | 2012-08-21 | 2015-03-03 | Google Inc. | Contact lens with integrated pulse oximeter |
| US9111473B1 (en) | 2012-08-24 | 2015-08-18 | Google Inc. | Input system |
| US8820934B1 (en) | 2012-09-05 | 2014-09-02 | Google Inc. | Passive surface acoustic wave communication |
| US20140192315A1 (en) | 2012-09-07 | 2014-07-10 | Google Inc. | In-situ tear sample collection and testing using a contact lens |
| US9398868B1 (en) | 2012-09-11 | 2016-07-26 | Verily Life Sciences Llc | Cancellation of a baseline current signal via current subtraction within a linear relaxation oscillator-based current-to-frequency converter circuit |
| US10010270B2 (en) | 2012-09-17 | 2018-07-03 | Verily Life Sciences Llc | Sensing system |
| US9326710B1 (en) | 2012-09-20 | 2016-05-03 | Verily Life Sciences Llc | Contact lenses having sensors with adjustable sensitivity |
| US8870370B1 (en) | 2012-09-24 | 2014-10-28 | Google Inc. | Contact lens that facilitates antenna communication via sensor impedance modulation |
| US8960898B1 (en) | 2012-09-24 | 2015-02-24 | Google Inc. | Contact lens that restricts incoming light to the eye |
| US8979271B2 (en) | 2012-09-25 | 2015-03-17 | Google Inc. | Facilitation of temperature compensation for contact lens sensors and temperature sensing |
| US8989834B2 (en) | 2012-09-25 | 2015-03-24 | Google Inc. | Wearable device |
| US20140088372A1 (en) | 2012-09-25 | 2014-03-27 | Google Inc. | Information processing method |
| US8985763B1 (en) | 2012-09-26 | 2015-03-24 | Google Inc. | Contact lens having an uneven embedded substrate and method of manufacture |
| US9884180B1 (en) | 2012-09-26 | 2018-02-06 | Verily Life Sciences Llc | Power transducer for a retinal implant using a contact lens |
| US8821811B2 (en) | 2012-09-26 | 2014-09-02 | Google Inc. | In-vitro contact lens testing |
| US8960899B2 (en) | 2012-09-26 | 2015-02-24 | Google Inc. | Assembling thin silicon chips on a contact lens |
| US9063351B1 (en) | 2012-09-28 | 2015-06-23 | Google Inc. | Input detection system |
| US8965478B2 (en) | 2012-10-12 | 2015-02-24 | Google Inc. | Microelectrodes in an ophthalmic electrochemical sensor |
| US9176332B1 (en) | 2012-10-24 | 2015-11-03 | Google Inc. | Contact lens and method of manufacture to improve sensor sensitivity |
| US9757056B1 (en) | 2012-10-26 | 2017-09-12 | Verily Life Sciences Llc | Over-molding of sensor apparatus in eye-mountable device |
| MY172901A (en) | 2012-12-17 | 2019-12-13 | Alcon Inc | Method for making improved uv-absorbing ophthalmic lenses |
| US8874182B2 (en) | 2013-01-15 | 2014-10-28 | Google Inc. | Encapsulated electronics |
| US9289954B2 (en) | 2013-01-17 | 2016-03-22 | Verily Life Sciences Llc | Method of ring-shaped structure placement in an eye-mountable device |
| US9636016B1 (en) | 2013-01-25 | 2017-05-02 | Verily Life Sciences Llc | Eye-mountable devices and methods for accurately placing a flexible ring containing electronics in eye-mountable devices |
| US20140209481A1 (en) | 2013-01-25 | 2014-07-31 | Google Inc. | Standby Biasing Of Electrochemical Sensor To Reduce Sensor Stabilization Time During Measurement |
| US9161712B2 (en) | 2013-03-26 | 2015-10-20 | Google Inc. | Systems and methods for encapsulating electronics in a mountable device |
| US9113829B2 (en) | 2013-03-27 | 2015-08-25 | Google Inc. | Systems and methods for encapsulating electronics in a mountable device |
| WO2014167611A1 (ja) * | 2013-04-12 | 2014-10-16 | ロート製薬株式会社 | コンタクトレンズ用組成物およびそれを用いたコンタクトレンズパッケージ |
| US20140371560A1 (en) | 2013-06-14 | 2014-12-18 | Google Inc. | Body-Mountable Devices and Methods for Embedding a Structure in a Body-Mountable Device |
| US9084561B2 (en) | 2013-06-17 | 2015-07-21 | Google Inc. | Symmetrically arranged sensor electrodes in an ophthalmic electrochemical sensor |
| US9948895B1 (en) | 2013-06-18 | 2018-04-17 | Verily Life Sciences Llc | Fully integrated pinhole camera for eye-mountable imaging system |
| US9685689B1 (en) | 2013-06-27 | 2017-06-20 | Verily Life Sciences Llc | Fabrication methods for bio-compatible devices |
| US9028772B2 (en) | 2013-06-28 | 2015-05-12 | Google Inc. | Methods for forming a channel through a polymer layer using one or more photoresist layers |
| US9307901B1 (en) | 2013-06-28 | 2016-04-12 | Verily Life Sciences Llc | Methods for leaving a channel in a polymer layer using a cross-linked polymer plug |
| US9492118B1 (en) | 2013-06-28 | 2016-11-15 | Life Sciences Llc | Pre-treatment process for electrochemical amperometric sensor |
| US9814387B2 (en) | 2013-06-28 | 2017-11-14 | Verily Life Sciences, LLC | Device identification |
| MY180543A (en) | 2013-12-17 | 2020-12-01 | Novartis Ag | A silicone hydrogel lens with a crosslinked hydrophilic coating |
| US9572522B2 (en) | 2013-12-20 | 2017-02-21 | Verily Life Sciences Llc | Tear fluid conductivity sensor |
| US9654674B1 (en) | 2013-12-20 | 2017-05-16 | Verily Life Sciences Llc | Image sensor with a plurality of light channels |
| US9366570B1 (en) | 2014-03-10 | 2016-06-14 | Verily Life Sciences Llc | Photodiode operable in photoconductive mode and photovoltaic mode |
| US9184698B1 (en) | 2014-03-11 | 2015-11-10 | Google Inc. | Reference frequency from ambient light signal |
| US9789655B1 (en) | 2014-03-14 | 2017-10-17 | Verily Life Sciences Llc | Methods for mold release of body-mountable devices including microelectronics |
| US20150366311A1 (en) * | 2014-06-19 | 2015-12-24 | Coopervision International Holding Company, Lp | Protection of Contact Lenses from Microbial Contamination Caused by Handling |
| SG10201901379TA (en) | 2014-08-26 | 2019-03-28 | Novartis Ag | Method for applying stable coating on silicone hydrogel contact lenses |
| TWI742016B (zh) * | 2015-11-30 | 2021-10-11 | 日商樂敦製藥股份有限公司 | 眼科組成物 |
| MY186090A (en) * | 2015-12-03 | 2021-06-21 | Alcon Inc | Contact lens packaging solutions |
| EP3391101B1 (en) | 2015-12-15 | 2020-07-08 | Alcon Inc. | Method for applying stable coating on silicone hydrogel contact lenses |
| US11746308B2 (en) | 2017-12-04 | 2023-09-05 | Nof Corporation | Soft contact lens treatment solution |
| WO2019116139A1 (en) | 2017-12-13 | 2019-06-20 | Novartis Ag | Weekly and monthly disposable water gradient contact lenses |
| TWI798452B (zh) * | 2019-06-17 | 2023-04-11 | 晶碩光學股份有限公司 | 隱形眼鏡產品 |
Family Cites Families (119)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3549747A (en) * | 1968-02-20 | 1970-12-22 | Flow Pharma Inc | Contact lens wetting solution and method of using same |
| USB724600I5 (ja) * | 1968-04-26 | |||
| US3639576A (en) * | 1968-06-19 | 1972-02-01 | Barnes Hind Pharm Inc | Resterilizing contact lens solution |
| GB1340517A (en) | 1969-12-01 | 1973-12-12 | Burton Parsons Chemicals Inc | Ophthalmic solution |
| US4013576A (en) | 1973-11-21 | 1977-03-22 | Wesley-Jessen Inc. | Contact lens treating composition |
| JPS5111853U (ja) * | 1974-07-12 | 1976-01-28 | ||
| US4141665A (en) * | 1977-09-14 | 1979-02-27 | Snapp Jr Edward A | Lockable joint for articulated members |
| US4287175A (en) * | 1978-06-22 | 1981-09-01 | Merck & Co., Inc. | Contact lens wetting agents |
| US4409205A (en) * | 1979-03-05 | 1983-10-11 | Cooper Laboratories, Inc. | Ophthalmic solution |
| US4323467A (en) * | 1980-11-24 | 1982-04-06 | Syntex (U.S.A.) Inc. | Contact lens cleaning, storing and wetting solutions |
| JPS57186733A (en) * | 1981-05-13 | 1982-11-17 | Toyo Contact Lens Co Ltd | Agent for use in contact lenses |
| JPS5870208A (ja) | 1981-10-22 | 1983-04-26 | Toyo Contact Lens Co Ltd | コンタクトレンズ用洗浄剤 |
| US4551461A (en) * | 1982-06-01 | 1985-11-05 | Sherman Laboratories, Inc. | Soft contact lens ambient temperature disinfectant and rinsing solution and method |
| US4626292A (en) * | 1982-06-01 | 1986-12-02 | Sherman Laboratories, Inc. | Soft contact lens wetting and preservation method |
| US4529535A (en) * | 1982-06-01 | 1985-07-16 | Sherman Laboratories, Inc. | Soft contact lens wetting solution containing preservative system and method |
| GB8422950D0 (en) * | 1984-09-11 | 1984-10-17 | Warne K J | Hydrogel |
| US4808239A (en) | 1984-12-28 | 1989-02-28 | Alcon Laboratories, Inc. | Method of cleaning contact lens using compositions containing polyether carboxylic acid surfactant |
| US4748189A (en) * | 1985-04-19 | 1988-05-31 | Ciba-Geigy Corporation | Ophthalmic solutions and methods for improving the comfort and safety of contact lenses |
| GB8601967D0 (en) * | 1986-01-28 | 1986-03-05 | Coopervision Optics | Manufacturing contact lenses |
| US4786436A (en) * | 1986-01-31 | 1988-11-22 | Bausch & Lomb Incorporated | Wetting solutions for contact lenses |
| US4783488A (en) * | 1987-01-31 | 1988-11-08 | Bausch & Lomb Incorporated | Contact lens wetting solution |
| US5141665A (en) | 1987-03-31 | 1992-08-25 | Sherman Laboratories, Inc. | Cleaning, conditioning, storing and wetting system and method for rigid gas permeable contact lenses and other contact lenses |
| US5322667A (en) * | 1987-03-31 | 1994-06-21 | Sherman Pharmaceuticals, Inc. | Preservative system for ophthalmic and contact lens solutions and method for cleaning disinfecting and storing contact lenses |
| EP0315836A3 (en) | 1987-10-30 | 1990-10-17 | HÜLS AMERICA INC. (a Delaware corporation) | Polyorganosiloxane based interpenetrating network polymers and methods of making |
| US5089053A (en) * | 1989-11-09 | 1992-02-18 | Polymer Technology Corporation | Contact lens cleaning material and method |
| US5157093A (en) * | 1990-05-10 | 1992-10-20 | Ciba-Geigy Corporation | Hydroxyethyl cellulose derivatives containing pendant (meth)acryloyl units bound through urethane groups and hydrogel contact lenses made therefrom |
| AU647880B2 (en) * | 1991-02-28 | 1994-03-31 | Ciba-Geigy Ag | Contact lenses made from thermoformable material |
| NZ242358A (en) * | 1991-05-10 | 1994-03-25 | Allergan Inc | Use of thiol compounds to inhibit deposits on a contact lens |
| US5260001A (en) | 1992-08-03 | 1993-11-09 | Bausch & Lomb Incorporated | Spincasting process for producing a series of contact lenses having desired shapes |
| WO1994009794A1 (en) | 1992-10-28 | 1994-05-11 | Cytrx Corporation | Ophthalmic solution and method of use |
| US5364601A (en) * | 1992-12-30 | 1994-11-15 | Bausch & Lomb Incorporated | Treating of contact lenses with compositions comprising PVP-H202 |
| EP0681485A1 (en) * | 1993-01-26 | 1995-11-15 | Allergan, Inc. | Compositions and methods to disinfect contact lenses |
| US5401327A (en) * | 1993-06-18 | 1995-03-28 | Wilmington Partners L.P. | Method of treating contact lenses |
| ES2144525T3 (es) * | 1993-06-18 | 2000-06-16 | Polymer Technology Corp | Composicion para limpiar y humedecer lentes de contacto. |
| AU7061594A (en) | 1993-06-18 | 1995-01-17 | Polymer Technology Corporation | Contact lens solution containing peo and cationic cellulose |
| US5405878A (en) | 1993-06-18 | 1995-04-11 | Wilmington Partners L.P. | Contact lens solution containing cationic glycoside |
| EP0703965B1 (en) * | 1993-06-18 | 1999-01-20 | Polymer Technology Corporation | Composition for cleaning and wetting contact lenses |
| US5382599A (en) * | 1993-10-13 | 1995-01-17 | Allergan, Inc. | Method of inhibiting protozoan growth in eye care products using a polyvalent cation chelating agent |
| US5712356A (en) * | 1993-11-26 | 1998-01-27 | Ciba Vision Corporation | Cross-linkable copolymers and hydrogels |
| AU1373195A (en) * | 1993-12-21 | 1995-07-10 | Bausch & Lomb Incorporated | Method for increasing hydrophilicity of contact lenses |
| WO1995034327A1 (en) | 1994-06-15 | 1995-12-21 | Alcon Laboratories, Inc. | Improved method for storing contact lenses |
| BR9508909A (pt) * | 1994-09-09 | 1998-01-13 | Novo Nordisk As | Processo produto e tablete ou cápsula de limpeza desinfecção e preservação de lentes de contato e uso de um inibidor de enzima |
| US5700559A (en) * | 1994-12-16 | 1997-12-23 | Advanced Surface Technology | Durable hydrophilic surface coatings |
| EP0733918B1 (en) | 1995-03-24 | 2003-07-30 | Ocular Research of Boston, Inc. | Hydrogel lens pre-coated with lipid layer |
| US5731087A (en) * | 1995-06-07 | 1998-03-24 | Union Carbide Chemicals & Plastics Technology Corporation | Lubricious coatings containing polymers with vinyl and carboxylic acid moieties |
| US5605661A (en) * | 1995-08-18 | 1997-02-25 | Alcon Laboratories, Inc. | Methods of using liquid enzyme compositions containing mixed polyols |
| GB9524452D0 (en) | 1995-11-30 | 1996-01-31 | Award Plc | Contact lens packaging |
| US5800412A (en) * | 1996-10-10 | 1998-09-01 | Sts Biopolymers, Inc. | Hydrophilic coatings with hydrating agents |
| US5882687A (en) | 1997-01-10 | 1999-03-16 | Allergan | Compositions and methods for storing contact lenses |
| US5800807A (en) * | 1997-01-29 | 1998-09-01 | Bausch & Lomb Incorporated | Ophthalmic compositions including glycerin and propylene glycol |
| DE69728276T2 (de) | 1997-04-03 | 2005-04-21 | Ophtecs Corp Osaka | Einkomponenten-zusammensetzung zur desinfektion, neutralisation und reinigung von kontaktlinsen und verfahren zur desinfektion, neutralisation und reinigung |
| FR2762849B1 (fr) * | 1997-05-05 | 1999-06-18 | Essilor Int | Solution aqueuse d'entretien des lentilles de contact |
| GB9711818D0 (en) * | 1997-06-06 | 1997-08-06 | Bausch & Lomb | Contact lens packing solutions and methods for improving the comfort of disposable contact lenses |
| US7654728B2 (en) * | 1997-10-24 | 2010-02-02 | Revalesio Corporation | System and method for therapeutic application of dissolved oxygen |
| US20030129083A1 (en) * | 1997-11-26 | 2003-07-10 | Advanced Medical Optics, Inc. | Multi purpose contact lens care compositions including propylene glycol or glycerin |
| US7052131B2 (en) | 2001-09-10 | 2006-05-30 | J&J Vision Care, Inc. | Biomedical devices containing internal wetting agents |
| US5998498A (en) * | 1998-03-02 | 1999-12-07 | Johnson & Johnson Vision Products, Inc. | Soft contact lenses |
| US6367929B1 (en) | 1998-03-02 | 2002-04-09 | Johnson & Johnson Vision Care, Inc. | Hydrogel with internal wetting agent |
| US6822016B2 (en) * | 2001-09-10 | 2004-11-23 | Johnson & Johnson Vision Care, Inc. | Biomedical devices containing internal wetting agents |
| US6348507B1 (en) * | 1998-05-05 | 2002-02-19 | Bausch & Lomb Incorporated | Surface treatment of silicone hydrogel contact lenses |
| WO1999057177A1 (en) * | 1998-05-05 | 1999-11-11 | Bausch & Lomb Incorporated | Plasma surface treatment of silicone hydrogel contact lenses |
| JP3883739B2 (ja) * | 1998-05-22 | 2007-02-21 | 株式会社メニコン | コンタクトレンズ用殺菌液 |
| AU4980199A (en) | 1998-07-08 | 2000-02-01 | Sunsoft Corporation | Interpenetrating polymer network hydrophilic hydrogels for contact lens |
| AU1180300A (en) * | 1998-11-16 | 2000-06-05 | Rohto Pharmaceutical Co., Ltd. | Liquid ophthalmic preparations |
| US6274133B1 (en) * | 1998-12-22 | 2001-08-14 | Bausch & Lomb Incorporated | Method for treating extended-wear contact lenses in the eyes |
| US6037328A (en) * | 1998-12-22 | 2000-03-14 | Bausch & Lomb Incorporated | Method and composition for rewetting and preventing deposits on contact lens |
| US6630243B2 (en) | 1999-05-20 | 2003-10-07 | Bausch & Lomb Incorporated | Surface treatment of silicone hydrogel contact lenses comprising hydrophilic polymer chains attached to an intermediate carbon coating |
| US6482799B1 (en) * | 1999-05-25 | 2002-11-19 | The Regents Of The University Of California | Self-preserving multipurpose ophthalmic solutions incorporating a polypeptide antimicrobial |
| EP1214883B1 (en) | 1999-09-20 | 2003-11-26 | Menicon Co., Ltd. | Liquid preparation for contact lenses |
| BR0014552B1 (pt) | 1999-10-07 | 2011-07-12 | método de redução do módulo de young e bronzeamento (delta) de um hidrogel de silicone, hidrogéis de silicone, lentes de contato, método de produção de um polìmero e macrÈmero útil para preparar hidrogéis de silicone. | |
| AU1583601A (en) | 1999-11-05 | 2001-06-06 | Bausch & Lomb Incorporated | Surface treatment of non-plasma treated silicone hydrogel contact lenses |
| CA2389013A1 (en) * | 1999-12-08 | 2001-06-14 | The Procter & Gamble Company | Compositions including ether-capped poly(oxyalkylated) alcohol wetting agents |
| US20020018732A1 (en) * | 2000-04-21 | 2002-02-14 | Hung William M. | Preserving compositions containing chitosan and processes for making water soluble O-acetylated chitosan and chitosan |
| US6428839B1 (en) * | 2000-06-02 | 2002-08-06 | Bausch & Lomb Incorporated | Surface treatment of medical device |
| US20020182315A1 (en) * | 2000-11-01 | 2002-12-05 | Heiler David J. | Surface treatment of non-plasma treated silicone hydrogel contact lenses |
| DE60140007D1 (de) | 2000-11-08 | 2009-11-05 | Fxs Ventures Llc | Verbesserte ophthalmische und kontaktlinsenlösungen mit einfachen sacchariden als konservierungsverbesserer |
| US6634748B1 (en) * | 2000-11-15 | 2003-10-21 | Johnson & Johnson Vision Care, Inc. | Methods of stabilizing silicone hydrogels against hydrolytic degradation |
| US6867172B2 (en) * | 2000-12-07 | 2005-03-15 | Johnson & Johnson Vision Care, Inc. | Methods of inhibiting the adherence of lenses to their packaging |
| US6531432B2 (en) | 2000-12-07 | 2003-03-11 | Johnson & Johnson Vision Care, Inc. | Contact lens packaging solutions |
| US20020115578A1 (en) * | 2000-12-14 | 2002-08-22 | Groemminger Suzanne F. | Composition for cleaning and wetting contact lenses |
| US6805836B2 (en) | 2000-12-15 | 2004-10-19 | Bausch & Lomb Incorporated | Prevention of preservative uptake into biomaterials |
| US6702983B2 (en) * | 2001-05-15 | 2004-03-09 | Bausch & Lomb Incorporated | Low ionic strength method and composition for reducing bacterial attachment to biomaterials |
| US6815074B2 (en) * | 2001-05-30 | 2004-11-09 | Novartis Ag | Polymeric materials for making contact lenses |
| US7879267B2 (en) * | 2001-08-02 | 2011-02-01 | J&J Vision Care, Inc. | Method for coating articles by mold transfer |
| US20030095230A1 (en) * | 2001-08-02 | 2003-05-22 | Neely Frank L. | Antimicrobial lenses and methods of their use related patent applications |
| JP2003057610A (ja) * | 2001-08-09 | 2003-02-26 | Ophtecs Corp | コンタクトレンズ用液剤組成物 |
| US6528464B1 (en) * | 2001-08-17 | 2003-03-04 | Bausch & Lomb Incorporated | Composition and method for inhibiting uptake of biguanide antimicrobials by hydrogels |
| US6617291B1 (en) * | 2001-11-08 | 2003-09-09 | Francis X. Smith | Ophthalmic and contact lens solutions |
| US20040028645A1 (en) * | 2001-12-13 | 2004-02-12 | Masood Chowhan | Artificial tear composition adapted to be used with contact lenses |
| IL165910A0 (en) * | 2002-07-03 | 2006-01-15 | Pericor Science Inc | Compositions of hyaluronic acid and methods of use |
| US20080299179A1 (en) * | 2002-09-06 | 2008-12-04 | Osman Rathore | Solutions for ophthalmic lenses containing at least one silicone containing component |
| US6926965B2 (en) * | 2002-09-11 | 2005-08-09 | Novartis Ag | LbL-coated medical device and method for making the same |
| CN1684722A (zh) * | 2002-09-30 | 2005-10-19 | 博士伦公司 | 减少细菌在生物材料和生物医学装置上的粘附 |
| US7550418B2 (en) * | 2002-12-13 | 2009-06-23 | Novartis Ag | Lens care composition and method |
| US7037469B2 (en) * | 2003-03-19 | 2006-05-02 | Bausch & Lomb, Inc. | Method and composition for reducing contact lens swelling |
| US20060073185A1 (en) * | 2002-12-13 | 2006-04-06 | Bausch & Lomb Incorporated | Method and composition for contact lenses |
| US20040115270A1 (en) * | 2002-12-13 | 2004-06-17 | Dharmendra Jani | Absorption and controlled release of polyethers from hydrogel biomaterials |
| US20040120982A1 (en) * | 2002-12-19 | 2004-06-24 | Zanini Diana | Biomedical devices with coatings attached via latent reactive components |
| US20040119176A1 (en) * | 2002-12-23 | 2004-06-24 | Bausch & Lomb Incorporated | Method for manufacturing lenses |
| EP2283876A1 (en) * | 2002-12-23 | 2011-02-16 | Johnson and Johnson Vision Care, Inc. | Contact lens packages containing additives |
| US20040120916A1 (en) * | 2002-12-23 | 2004-06-24 | Advanced Medical Optics, Inc. | Contact lens care compositions, methods of use and preparation which protect ocular tissue membrane integrity |
| US20040137079A1 (en) | 2003-01-08 | 2004-07-15 | Cook James N. | Contact lens and eye drop rewetter compositions and methods |
| WO2004091438A2 (en) | 2003-04-15 | 2004-10-28 | Fxs Ventures, Llc | Improved ophthalmic and contact lens solutions containing peptides as representative enhancers |
| WO2005011966A1 (en) | 2003-07-24 | 2005-02-10 | Provis Limited | Methods and apparatus for use in contact lens manufacture and packaging |
| US7416737B2 (en) * | 2003-11-18 | 2008-08-26 | Johnson & Johnson Vision Care, Inc. | Antimicrobial lenses, processes to prepare them and methods of their use |
| US20050119141A1 (en) | 2003-12-01 | 2005-06-02 | Irene Quenville | Stability enhancement of solutions containing antimicrobial agents |
| JP2005208628A (ja) * | 2003-12-25 | 2005-08-04 | Lion Corp | コンタクトレンズ用プレケア剤及びコンタクトレンズのプレケア方法 |
| US7786185B2 (en) | 2004-03-05 | 2010-08-31 | Johnson & Johnson Vision Care, Inc. | Wettable hydrogels comprising acyclic polyamides |
| JPWO2006009101A1 (ja) | 2004-07-21 | 2008-05-01 | 千寿製薬株式会社 | コンタクトレンズ用装着液 |
| WO2006033728A1 (en) * | 2004-08-20 | 2006-03-30 | Noveon, Inc. | Associative thickeners for aqueous systems |
| SG155241A1 (en) * | 2004-08-27 | 2009-09-30 | Asahikasei Aime Co Ltd | Silicone hydrogel contact lenses |
| US7247692B2 (en) * | 2004-09-30 | 2007-07-24 | Johnson & Johnson Vision Care, Inc. | Biomedical devices containing amphiphilic block copolymers |
| ATE452659T1 (de) | 2004-10-01 | 2010-01-15 | Menicon Co Ltd | Verfahren zur sterilisation verpackter kontaktlinsen mit verpackungslösung |
| JP4642449B2 (ja) | 2004-12-06 | 2011-03-02 | 株式会社メニコン | コンタクトレンズ用液剤組成物 |
| CN103933614B (zh) * | 2005-02-14 | 2016-03-02 | 庄臣及庄臣视力保护公司 | 舒适的眼用器件及其制造方法 |
| JP5116211B2 (ja) * | 2005-03-03 | 2013-01-09 | ロート製薬株式会社 | 粘膜適用組成物 |
| US9804295B2 (en) * | 2005-05-05 | 2017-10-31 | Novartis Ag | Ophthalmic devices for sustained delivery of active compounds |
| US20070195261A1 (en) * | 2006-02-17 | 2007-08-23 | Jurgen Vogt | Method for sterilization of hydrogel contact lenses |
| JP5643092B2 (ja) * | 2007-08-31 | 2014-12-17 | ノバルティス アーゲー | コンタクトレンズのパッケージング溶液 |
-
2008
- 2008-08-27 JP JP2010522932A patent/JP5643092B2/ja not_active Expired - Fee Related
- 2008-08-27 KR KR1020107006936A patent/KR101535692B1/ko active IP Right Grant
- 2008-08-27 EP EP08795627A patent/EP2188655B1/en not_active Not-in-force
- 2008-08-27 AT AT08795627T patent/ATE540327T1/de active
- 2008-08-27 WO PCT/US2008/010146 patent/WO2009032122A1/en active Application Filing
- 2008-08-27 CA CA2692831A patent/CA2692831C/en not_active Expired - Fee Related
- 2008-08-27 US US12/229,771 patent/US8689971B2/en active Active
-
2014
- 2014-02-17 US US14/181,890 patent/US9162784B2/en active Active
- 2014-07-24 JP JP2014151165A patent/JP5821140B2/ja not_active Expired - Fee Related
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014222362A (ja) * | 2007-08-31 | 2014-11-27 | ノバルティス アーゲー | コンタクトレンズのパッケージング溶液 |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2692831C (en) | 2016-05-17 |
| US20140237945A1 (en) | 2014-08-28 |
| JP2010538321A (ja) | 2010-12-09 |
| KR101535692B1 (ko) | 2015-07-09 |
| WO2009032122A1 (en) | 2009-03-12 |
| EP2188655B1 (en) | 2012-01-04 |
| US8689971B2 (en) | 2014-04-08 |
| KR20100049125A (ko) | 2010-05-11 |
| ATE540327T1 (de) | 2012-01-15 |
| US20090057164A1 (en) | 2009-03-05 |
| EP2188655A1 (en) | 2010-05-26 |
| JP5821140B2 (ja) | 2015-11-24 |
| US9162784B2 (en) | 2015-10-20 |
| JP2014222362A (ja) | 2014-11-27 |
| CA2692831A1 (en) | 2009-03-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5821140B2 (ja) | コンタクトレンズのパッケージング溶液 | |
| JP5880614B2 (ja) | コンタクトレンズ製品 | |
| JP6595120B2 (ja) | コンタクトレンズパッケージング溶液 | |
| AU2013224663B2 (en) | Contact Lens Products |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A524 | Written submission of copy of amendment under article 19 pct |
Free format text: JAPANESE INTERMEDIATE CODE: A524 Effective date: 20110606 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110606 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20130125 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130416 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130712 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130722 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130815 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130822 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130906 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20140325 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140724 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20140909 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20141021 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20141030 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5643092 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |