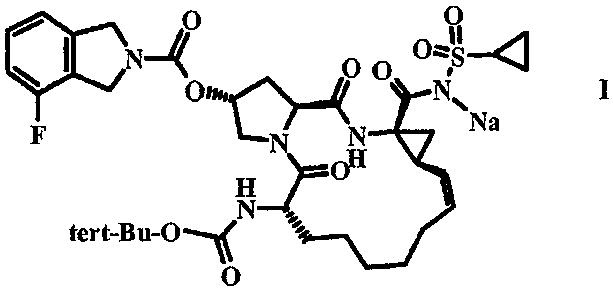
JP5523552B2 - 薬物動態を改善する方法 - Google Patents
薬物動態を改善する方法 Download PDFInfo
- Publication number
- JP5523552B2 JP5523552B2 JP2012506491A JP2012506491A JP5523552B2 JP 5523552 B2 JP5523552 B2 JP 5523552B2 JP 2012506491 A JP2012506491 A JP 2012506491A JP 2012506491 A JP2012506491 A JP 2012506491A JP 5523552 B2 JP5523552 B2 JP 5523552B2
- Authority
- JP
- Japan
- Prior art keywords
- ritonavir
- cytochrome
- inhibitor
- formula
- inhibitors
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/426—1,3-Thiazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/095—Sulfur, selenium, or tellurium compounds, e.g. thiols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/407—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with other heterocyclic ring systems, e.g. ketorolac, physostigmine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Oncology (AREA)
- Virology (AREA)
- Communicable Diseases (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Engineering & Computer Science (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Description
何かの存在に付けられる「a」または「an」は、成句として、本明細書で用いられるように、その存在が1またはそれより多くであることを意味し、例えば、化合物(a compound)は、1種またはそれより多くの化合物、または、少なくとも1種の化合物を意味する。そのようなものとして、用語「1つの(a)」(または「an」)、「1またはそれより多くの」および「少なくとも1つの」は、本明細書において同義的に用いることができる。
本発明のさらにその他の実施態様において、HCVを治療するために、または、HCVを治療する医薬品を製造するために、α−、β−またはγ−インターフェロン、および/または、サイモシン、および/または、リバビリン、および/または、R7128を、R7227およびリトナビルと共に使用することが提供される。
本発明の実施態様において、式Iで示される化合物、リトナビル、および、任意に阻害剤使用に関する指示を含む説明書き(informational insert)を含む薬剤パックが提供される。
投与前の21日以内に、被検者をこの研究に参加できるかどうかに関してスクリーニングした。この研究に、14人の健康な志願者を登録した(n=14/グループ)。以下に投与スケジュールを説明する:
1日目、R7227投与の前にPKサンプル(5mL)を回収し(投与前)、さらに、R7227投与から0.5、1、1.5、2、2.5、3、3.5、4、6、8、12および24時間後に回収した。
Yij=μ+τi+sj+εij
式中、Yijは、分析されるPKパラメーターを意味し、μは、変換した可変値の全体の平均を意味し、τiは、治療の母数効果を意味し;sjは、被検者の変量効果を意味し;εijは(エラー)を意味する。ランダム偏差εijは、独立しており、ゼロ平均で正規分布しおり、共通の分散σ2を有すると考えられる。比較のためのτR7227+リトナビルのグループとτR7227のグループについて、ANOVAモデルから、比較グループに関して残差分散σ2および90%の信頼限界が推測された。対数変換した可変値(AUC0→∞およびCmax)に関して、真のグループ平均の比率、および、それに対応する未変換の可変値の平均の比率に関する最小二乗平均の差を累乗することと変換した値に関する信頼限界それぞれによって信頼限界を計算した。
Claims (13)
- 式Iで示される化合物およびリトナビルが、別々の投薬形態である、請求項1に記載の医薬。
- 前記別々の投薬形態が、ほぼ同時に投与される、請求項2に記載の医薬。
- 式Iで示される化合物およびリトナビルが、単一の投与形態で投与される、請求項1に記載の医薬。
- 請求項1で定義された式Iで示される化合物、その遊離塩基または医薬的に許容される塩と、チトクロームP450モノオキシゲナーゼ阻害剤またはその医薬的に許容される塩を含むC型肝炎ウイルス感染の治療薬であって、ここで該チトクロームP450モノオキシゲナーゼ阻害剤は、リトナビルまたはその医薬的に許容される塩である、上記治療薬。
- さらに、免疫調節剤、および/または、抗ウイルス剤、および/または、HCVのNS3/4Aプロテアーゼのその他の阻害剤、および/または、NS5Bポリメラーゼの阻害剤、および/または、広域ウイルス阻害剤、および/または、その他のチトクロームP−450阻害剤から選択される少なくとも1種の追加の物質を含む、請求項5に記載の治療薬。
- C型肝炎ウイルスNS3/4Aプロテアーゼ阻害剤R7227の生物学的利用率を増加させる医薬品を製造するための、R7227とチトクロームP450モノオキシゲナーゼ阻害剤との使用であって、ここで該チトクロームP450モノオキシゲナーゼ阻害剤の量は、チトクロームP450モノオキシゲナーゼ阻害剤の非存在下のR7227の血中濃度と比較して、R7227の血中濃度を高めるのに十分であり、ここで該チトクロームP450モノオキシゲナーゼ阻害剤は、リトナビルまたはその医薬的に許容される塩である、上記使用。
- 前記R7227およびリトナビルが、別々の投薬形態である、請求項7に記載の使用。
- 前記R7227およびリトナビルが、同時に投与される、請求項7に記載の使用。
- R7227およびリトナビル、ならびに少なくとも1種の追加の物質の使用であって、ここで該少なくとも1種の追加の物質は、免疫調節剤;抗ウイルス剤;HCVのNS3/4Aプロテアーゼのその他の阻害剤;NS5Bポリメラーゼの阻害剤;広域ウイルス阻害剤、および、その他のチトクロームP−450阻害剤から選択される、請求項7に記載の使用。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17272209P | 2009-04-25 | 2009-04-25 | |
| US61/172,722 | 2009-04-25 | ||
| PCT/EP2010/055317 WO2010122087A1 (en) | 2009-04-25 | 2010-04-22 | Methods for improving pharmacokinetics |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2012524753A JP2012524753A (ja) | 2012-10-18 |
| JP5523552B2 true JP5523552B2 (ja) | 2014-06-18 |
Family
ID=42236685
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012506491A Active JP5523552B2 (ja) | 2009-04-25 | 2010-04-22 | 薬物動態を改善する方法 |
Country Status (36)
| Country | Link |
|---|---|
| US (4) | US20100272682A1 (ja) |
| EP (1) | EP2421527B1 (ja) |
| JP (1) | JP5523552B2 (ja) |
| KR (1) | KR101471238B1 (ja) |
| CN (2) | CN105233249A (ja) |
| AR (1) | AR076388A1 (ja) |
| AU (1) | AU2010240893B2 (ja) |
| BR (1) | BRPI1015147B1 (ja) |
| CA (1) | CA2758644C (ja) |
| CL (1) | CL2011002643A1 (ja) |
| CO (1) | CO6440593A2 (ja) |
| CR (1) | CR20110551A (ja) |
| DK (1) | DK2421527T3 (ja) |
| EC (1) | ECSP11011420A (ja) |
| ES (1) | ES2683736T3 (ja) |
| HK (1) | HK1215858A1 (ja) |
| HR (1) | HRP20181306T1 (ja) |
| HU (1) | HUE040182T2 (ja) |
| IL (1) | IL215620A (ja) |
| LT (1) | LT2421527T (ja) |
| MA (1) | MA33212B1 (ja) |
| MX (2) | MX351185B (ja) |
| MY (1) | MY169734A (ja) |
| NZ (1) | NZ595917A (ja) |
| PE (1) | PE20120638A1 (ja) |
| PL (1) | PL2421527T3 (ja) |
| PT (1) | PT2421527T (ja) |
| RS (1) | RS57501B1 (ja) |
| RU (1) | RU2591830C2 (ja) |
| SG (1) | SG175328A1 (ja) |
| SI (1) | SI2421527T1 (ja) |
| TR (1) | TR201809600T4 (ja) |
| TW (1) | TWI468160B (ja) |
| UA (1) | UA103801C2 (ja) |
| WO (1) | WO2010122087A1 (ja) |
| ZA (1) | ZA201107421B (ja) |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2358736A1 (en) * | 2008-10-15 | 2011-08-24 | Intermune, Inc. | Therapeutic antiviral peptides |
| AR075584A1 (es) | 2009-02-27 | 2011-04-20 | Intermune Inc | COMPOSICIONES TERAPEUTICAS QUE COMPRENDEN beta-D-2'-DESOXI-2'-FLUORO-2'-C-METILCITIDINA Y UN DERIVADO DE ACIDO ISOINDOL CARBOXILICO Y SUS USOS. COMPUESTO. |
| EP2455068A1 (en) * | 2010-11-09 | 2012-05-23 | F. Hoffmann-La Roche AG | Pharmaceutical composition for treating HCV infections |
| EP2583677A3 (en) | 2011-10-21 | 2013-07-03 | Abbvie Inc. | Methods for treating HCV comprising at least two direct acting antiviral agent, ribavirin but not interferon. |
| DE202012012955U1 (de) | 2011-10-21 | 2014-07-14 | Abbvie Inc. | Eine Kombination aus mindestens zwei direkt wirkenden antiviralen Wirkstoffen (DAAs) für die Verwendung zur Behandlung von HCV |
| US8466159B2 (en) | 2011-10-21 | 2013-06-18 | Abbvie Inc. | Methods for treating HCV |
| US8492386B2 (en) | 2011-10-21 | 2013-07-23 | Abbvie Inc. | Methods for treating HCV |
| WO2013106631A1 (en) | 2012-01-11 | 2013-07-18 | Abbvie Inc. | Processes for making hcv protease inhibitors |
| EP2866807A2 (en) * | 2012-06-27 | 2015-05-06 | Abbvie Inc. | Methods for treating hcv |
| US20140100364A1 (en) | 2012-10-08 | 2014-04-10 | Abbvie Inc. | Compounds Useful For Making HCV Protease Inhibitors |
| US11311519B2 (en) | 2014-05-01 | 2022-04-26 | Eiger Biopharmaceuticals, Inc. | Treatment of hepatitis delta virus infection |
| PL3137078T3 (pl) * | 2014-05-01 | 2019-08-30 | Eiger Biopharmaceuticals, Inc. | Leczenie zakażenia wirusem zapalenia wątroby typu delta |
| US10076512B2 (en) | 2014-05-01 | 2018-09-18 | Eiger Biopharmaceuticals, Inc. | Treatment of hepatitis delta virus infection |
| WO2016172342A1 (en) | 2015-04-21 | 2016-10-27 | Eiger Biopharmaceuticals, Inc. | Pharmaceutical compositions comprising lonafarnib and ritonavir |
| EA201892448A1 (ru) | 2016-04-28 | 2019-06-28 | Эмори Юниверсити | Алкинсодержащие нуклеотидные и нуклеозидные терапевтические композиции и связанные с ними способы применения |
| CN117105928B (zh) * | 2023-08-22 | 2024-03-26 | 上海蓝木化工有限公司 | 一种蛋白酶抑制剂及其制备方法 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2170020C (en) | 1992-12-29 | 2005-08-02 | Dale J. Kempf | Retroviral protease inhibiting compounds |
| IL110752A (en) | 1993-09-13 | 2000-07-26 | Abbott Lab | Liquid semi-solid or solid pharmaceutical composition for an HIV protease inhibitor |
| US5559158A (en) | 1993-10-01 | 1996-09-24 | Abbott Laboratories | Pharmaceutical composition |
| IL111991A (en) | 1994-01-28 | 2000-07-26 | Abbott Lab | Liquid pharmaceutical composition of HIV protease inhibitors in organic solvent |
| US6037157A (en) * | 1995-06-29 | 2000-03-14 | Abbott Laboratories | Method for improving pharmacokinetics |
| IL141438A0 (en) | 2000-02-23 | 2002-03-10 | Pfizer Prod Inc | Method of increasing the bioavailability and tissue penetration of azithromycin |
| KR20030081297A (ko) * | 2000-07-21 | 2003-10-17 | 쉐링 코포레이션 | C형 간염 바이러스의 ns3-세린 프로테아제억제제로서의 신규한 펩티드 |
| WO2004054586A1 (en) | 2002-12-16 | 2004-07-01 | Boehringer Ingelheim International Gmbh | Use of a combination containing a non-nucleoside reverse transcriptase inhibitor (nnrti) with an inhibitor of cytochrome p450, such as protease inhibitors |
| JP2007509950A (ja) * | 2003-10-27 | 2007-04-19 | バーテックス ファーマシューティカルズ インコーポレイテッド | Hcv処置の組合せ剤 |
| WO2008033466A2 (en) * | 2006-09-14 | 2008-03-20 | Combinatorx (Singapore) Pre. Ltd. | Compositions and methods for treatment of viral diseases |
| US20120220520A1 (en) * | 2006-10-17 | 2012-08-30 | Van T Klooster Gerben Albert Eleutherius | Bioavailable combinations for hcv treatment |
-
2010
- 2010-04-22 PL PL10714883T patent/PL2421527T3/pl unknown
- 2010-04-22 KR KR1020117027997A patent/KR101471238B1/ko active Active
- 2010-04-22 HR HRP20181306TT patent/HRP20181306T1/hr unknown
- 2010-04-22 SI SI201031725T patent/SI2421527T1/sl unknown
- 2010-04-22 CN CN201510706536.XA patent/CN105233249A/zh active Pending
- 2010-04-22 DK DK10714883.5T patent/DK2421527T3/en active
- 2010-04-22 JP JP2012506491A patent/JP5523552B2/ja active Active
- 2010-04-22 BR BRPI1015147-8A patent/BRPI1015147B1/pt active IP Right Grant
- 2010-04-22 MX MX2013013597A patent/MX351185B/es unknown
- 2010-04-22 MY MYPI2011005073A patent/MY169734A/en unknown
- 2010-04-22 EP EP10714883.5A patent/EP2421527B1/en active Active
- 2010-04-22 AU AU2010240893A patent/AU2010240893B2/en active Active
- 2010-04-22 MA MA34273A patent/MA33212B1/fr unknown
- 2010-04-22 LT LTEP10714883.5T patent/LT2421527T/lt unknown
- 2010-04-22 RU RU2011142651/15A patent/RU2591830C2/ru active
- 2010-04-22 RS RS20180898A patent/RS57501B1/sr unknown
- 2010-04-22 CA CA2758644A patent/CA2758644C/en active Active
- 2010-04-22 TR TR2018/09600T patent/TR201809600T4/tr unknown
- 2010-04-22 TW TW99112703A patent/TWI468160B/zh active
- 2010-04-22 MX MX2011011105A patent/MX2011011105A/es active IP Right Grant
- 2010-04-22 ES ES10714883.5T patent/ES2683736T3/es active Active
- 2010-04-22 NZ NZ595917A patent/NZ595917A/en unknown
- 2010-04-22 PT PT107148835T patent/PT2421527T/pt unknown
- 2010-04-22 HU HUE10714883A patent/HUE040182T2/hu unknown
- 2010-04-22 SG SG2011077732A patent/SG175328A1/en unknown
- 2010-04-22 CN CN2010800181204A patent/CN102413827A/zh active Pending
- 2010-04-22 UA UAA201113756A patent/UA103801C2/ru unknown
- 2010-04-22 WO PCT/EP2010/055317 patent/WO2010122087A1/en not_active Ceased
- 2010-04-22 PE PE2011001850A patent/PE20120638A1/es active IP Right Grant
- 2010-04-23 US US12/766,051 patent/US20100272682A1/en not_active Abandoned
- 2010-04-23 AR ARP100101352A patent/AR076388A1/es not_active Application Discontinuation
-
2011
- 2011-10-06 IL IL215620A patent/IL215620A/en active IP Right Grant
- 2011-10-10 ZA ZA2011/07421A patent/ZA201107421B/en unknown
- 2011-10-12 CO CO11135691A patent/CO6440593A2/es active IP Right Grant
- 2011-10-19 CR CR20110551A patent/CR20110551A/es unknown
- 2011-10-24 CL CL2011002643A patent/CL2011002643A1/es unknown
- 2011-10-25 EC EC2011011420A patent/ECSP11011420A/es unknown
-
2016
- 2016-04-05 HK HK16103828.4A patent/HK1215858A1/zh unknown
-
2017
- 2017-01-12 US US15/404,398 patent/US20170119739A1/en not_active Abandoned
-
2018
- 2018-07-11 US US16/033,177 patent/US20180318267A1/en not_active Abandoned
-
2020
- 2020-04-08 US US16/842,855 patent/US10918626B2/en active Active
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5523552B2 (ja) | 薬物動態を改善する方法 | |
| TW201424733A (zh) | 劑量型式 | |
| CN101743008A (zh) | 用于治疗或预防登革热感染的抗病毒药 | |
| TW201023858A (en) | Synergistic combinations of a macrocyclic inhibitor of HCV and a nucleoside | |
| EP2922596B1 (en) | Heterocyclyl carboxamides for treating viral diseases | |
| HK1168552A (en) | Methods for improving pharmacokinetics | |
| AU2013203132A1 (en) | Methods for improving pharmacokinetics | |
| Abd Elguaad | Study on HCV direct acting drugs in treatment of chronic hepatitis C | |
| Hofmann et al. | 754 IMPACT OF RIBAVIRIN ON SUBGENOMIC HEPATITIS C VIRUS RNA DECAY DURING TREATMENT WITH INTERFERON ALFA AND THE PROTEASE INHIBITOR TELAPREVIR | |
| JP2007153837A (ja) | C型肝炎ウイルス粒子形成・細胞外放出を抑制する方法 | |
| WO2017062840A1 (en) | Combination therapy for the treatment of hepatitis c virus | |
| HK1178451A (en) | Dose forms comprising vx-950 and their dosage regimen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130618 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130815 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130822 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20131115 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20131212 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140213 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20140310 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20140408 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5523552 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |