JP5339552B2 - 体液成分の分析器具 - Google Patents
体液成分の分析器具 Download PDFInfo
- Publication number
- JP5339552B2 JP5339552B2 JP2011509102A JP2011509102A JP5339552B2 JP 5339552 B2 JP5339552 B2 JP 5339552B2 JP 2011509102 A JP2011509102 A JP 2011509102A JP 2011509102 A JP2011509102 A JP 2011509102A JP 5339552 B2 JP5339552 B2 JP 5339552B2
- Authority
- JP
- Japan
- Prior art keywords
- sample
- plate
- flow path
- reaction chamber
- hole
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 210000001124 body fluid Anatomy 0.000 title claims description 38
- 239000010839 body fluid Substances 0.000 title claims description 32
- 239000007788 liquid Substances 0.000 claims description 32
- 239000002699 waste material Substances 0.000 claims description 32
- 238000004458 analytical method Methods 0.000 claims description 30
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 19
- 239000011148 porous material Substances 0.000 claims description 18
- 239000012491 analyte Substances 0.000 claims description 12
- 238000011144 upstream manufacturing Methods 0.000 claims description 9
- 230000008859 change Effects 0.000 claims description 7
- 230000003287 optical effect Effects 0.000 claims description 7
- 230000002940 repellent Effects 0.000 claims description 7
- 239000005871 repellent Substances 0.000 claims description 7
- 230000035699 permeability Effects 0.000 claims description 5
- 238000012546 transfer Methods 0.000 claims description 5
- 238000010030 laminating Methods 0.000 claims description 2
- 238000007789 sealing Methods 0.000 claims description 2
- 238000009423 ventilation Methods 0.000 claims description 2
- 102000017011 Glycated Hemoglobin A Human genes 0.000 description 17
- 108091005995 glycated hemoglobin Proteins 0.000 description 17
- 239000000126 substance Substances 0.000 description 13
- 102000001554 Hemoglobins Human genes 0.000 description 11
- 108010054147 Hemoglobins Proteins 0.000 description 11
- 239000000463 material Substances 0.000 description 8
- 230000000903 blocking effect Effects 0.000 description 7
- 238000005259 measurement Methods 0.000 description 7
- 238000000034 method Methods 0.000 description 6
- 239000013076 target substance Substances 0.000 description 6
- 239000002245 particle Substances 0.000 description 5
- 238000010586 diagram Methods 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 239000007850 fluorescent dye Substances 0.000 description 4
- 238000002372 labelling Methods 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 238000003860 storage Methods 0.000 description 4
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 210000003743 erythrocyte Anatomy 0.000 description 3
- 239000000439 tumor marker Substances 0.000 description 3
- XPDXVDYUQZHFPV-UHFFFAOYSA-N Dansyl Chloride Chemical compound C1=CC=C2C(N(C)C)=CC=CC2=C1S(Cl)(=O)=O XPDXVDYUQZHFPV-UHFFFAOYSA-N 0.000 description 2
- 206010018910 Haemolysis Diseases 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002301 cellulose acetate Polymers 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- 230000008588 hemolysis Effects 0.000 description 2
- 230000002949 hemolytic effect Effects 0.000 description 2
- 230000008105 immune reaction Effects 0.000 description 2
- 239000004816 latex Substances 0.000 description 2
- 229920000126 latex Polymers 0.000 description 2
- 230000000149 penetrating effect Effects 0.000 description 2
- -1 polyethylene terephthalate Polymers 0.000 description 2
- 229920000139 polyethylene terephthalate Polymers 0.000 description 2
- 239000005020 polyethylene terephthalate Substances 0.000 description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- 238000002834 transmittance Methods 0.000 description 2
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- AUYYCJSJGJYCDS-LBPRGKRZSA-N Thyrolar Chemical class IC1=CC(C[C@H](N)C(O)=O)=CC(I)=C1OC1=CC=C(O)C(I)=C1 AUYYCJSJGJYCDS-LBPRGKRZSA-N 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 239000012790 adhesive layer Substances 0.000 description 1
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 1
- 238000009739 binding Methods 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 230000009918 complex formation Effects 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000004907 flux Effects 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 238000003317 immunochromatography Methods 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 238000005375 photometry Methods 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000005495 thyroid hormone Substances 0.000 description 1
- 229940036555 thyroid hormone Drugs 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/543—Immunoassay; Biospecific binding assay; Materials therefor with an insoluble carrier for immobilising immunochemicals
- G01N33/54366—Apparatus specially adapted for solid-phase testing
- G01N33/54386—Analytical elements
- G01N33/54387—Immunochromatographic test strips
- G01N33/54388—Immunochromatographic test strips based on lateral flow
- G01N33/54389—Immunochromatographic test strips based on lateral flow with bidirectional or multidirectional lateral flow, e.g. wherein the sample flows from a single, common sample application point into multiple strips, lanes or zones
Landscapes
- Health & Medical Sciences (AREA)
- Immunology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Food Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Automatic Analysis And Handling Materials Therefor (AREA)
Description
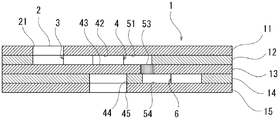
図1〜図4に、本発明に係る体液成分の分析器具の第1の実施の形態を示す。図1に平面図、図2に断面図を示し、図3に分解斜視図を示す。本実施の形態は、赤血球を溶血した試料をもとにヘモグロビンA1c(以下糖化ヘモグロビン)を測定するための形態となっている。
図9に、本発明の第2の実施形態に係る体液成分の分析器具100について、分解斜視図を示す。以下においては、第1の実施形態との相違点を中心に説明し、対応する箇所には同一の符号を付して説明する。
11 第1プレート
12 第2プレート
13 第3プレート
14 第4プレート
15 第5プレート
100 体液成分の分析器具
2 試料供給口
21 貫通孔
22 貫通孔
23 濾過手段
24 血漿貯留室
25 供給口保持体
3 第1の流路
31 溝
4 反応室
41 貫通孔
42 標識抗体保持部
421 標識抗体
422 可溶性保持体
43 固定化抗体保持部
431 固定化抗体
44 貫通孔
45 貫通孔
5 第2の流路
51 上流部
511 溝
511a 端部
52 保持孔
53 閉塞部材
54 下流部
541 溝
541a 端部
6 廃液室
61 貫通孔
81 糖化ヘモグロビン
82 未糖化ヘモグロビン
Claims (5)
- 試料が内部に供給される試料供給口と、
前記試料供給口から延出する第1の流路と、
前記第1の流路に連通する反応室と、
前記反応室から延出する第2の流路と、
前記第2の流路に連通する廃液室と、
前記第2の流路を閉塞するよう設けられた閉塞部材とを備え、
前記閉塞部材は、通気性を有するとともに、所定の圧力より低い圧では通水せず、前記所定の圧力以上の圧力で通水し、
前記反応室を取り囲み対面する2つの壁面が光透過性を有し、
前記2つの壁面のいずれか一方に前記試料に含まれる分析対象物質と特異的に結合する抗体が固定化されており、
さらに前記反応室に前記分析対象物質と特異的に結合する標識された抗体が遊離可能に保持されており、
前記廃液室を取り囲む壁面の少なくとも一部が通気性を有する
ことを特徴とする体液成分の分析器具。 - 前記閉塞部材が撥水処理された多孔質材料からなる
ことを特徴とする請求項1に記載の体液成分の分析器具。 - 不通気性でありかつ不通水性の保持プレートを備え、
前記保持プレートに、前記第2の流路の一部を構成する貫通孔が、板厚方向に貫通して設けられており、
前記貫通孔に前記閉塞部材が収容されており、
前記第2の流路のうち前記反応室と前記貫通孔とを連通する上流部分が、前記保持プレートの一の面側に配置されており、
前記第2の流路のうち前記貫通孔と前記廃液室とを連通する下流部分が、前記保持プレートの他の面側に配置されている
ことを特徴とする請求項1または2に記載の体液成分の分析器具。 - 前記廃液室が、不通水性で通気性のある通気プレートと、不通気性でありかつ不通水性の封止プレートとを積層して形成されている
ことを特徴とする請求項1〜3のいずれかに記載の体液成分の分析器具。 - 請求項1〜4にいずれかに記載の体液成分の分析器具を用い、
試料を前記試料供給口に供給する工程と、
前記所定の圧力より低い第1の圧力を前記使用供給口に与えて前記試料を前記反応室に移送する工程と、
所定の時間が経過した後、前記所定の圧力以上の第2の圧力を前記試料供給口に与えて前記試料を前記廃液室に移送する工程と、
前記反応室の光学的変化量を測定する工程とを備える
ことを特徴とする体液成分の分析方法。
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2009/057466 WO2010119501A1 (ja) | 2009-04-13 | 2009-04-13 | 体液成分の分析器具 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2010119501A1 JPWO2010119501A1 (ja) | 2012-10-22 |
| JP5339552B2 true JP5339552B2 (ja) | 2013-11-13 |
Family
ID=42982185
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011509102A Expired - Fee Related JP5339552B2 (ja) | 2009-04-13 | 2009-04-13 | 体液成分の分析器具 |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JP5339552B2 (ja) |
| WO (1) | WO2010119501A1 (ja) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003254896A (ja) * | 2002-02-28 | 2003-09-10 | Kokusai Gijutsu Kaihatsu Co Ltd | 試料カートリッジ、試料分析装置及び方法 |
| JP2008539419A (ja) * | 2005-04-30 | 2008-11-13 | シェーン ユウ、ジャエ | バイオディスクおよびバイオドライバ装置、これを用いた分析方法 |
| WO2009034649A1 (ja) * | 2007-09-14 | 2009-03-19 | Ttm Co., Ltd. | 液状流体の検査器具および検査方法 |
-
2009
- 2009-04-13 JP JP2011509102A patent/JP5339552B2/ja not_active Expired - Fee Related
- 2009-04-13 WO PCT/JP2009/057466 patent/WO2010119501A1/ja not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003254896A (ja) * | 2002-02-28 | 2003-09-10 | Kokusai Gijutsu Kaihatsu Co Ltd | 試料カートリッジ、試料分析装置及び方法 |
| JP2008539419A (ja) * | 2005-04-30 | 2008-11-13 | シェーン ユウ、ジャエ | バイオディスクおよびバイオドライバ装置、これを用いた分析方法 |
| WO2009034649A1 (ja) * | 2007-09-14 | 2009-03-19 | Ttm Co., Ltd. | 液状流体の検査器具および検査方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2010119501A1 (ja) | 2010-10-21 |
| JPWO2010119501A1 (ja) | 2012-10-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20080213133A1 (en) | Flow analysis apparatus and method | |
| JP6037184B2 (ja) | 多孔質媒体を利用したアッセイ装置 | |
| EP1776574B1 (en) | Mechanical cartridge with test strip fluid control features for use in a fluid analyte meter | |
| JP3553045B2 (ja) | バイオセンサ | |
| CN202886374U (zh) | 一个进行多个测试的设备 | |
| EP2285491A1 (en) | Flow control in microfluidic systems | |
| TWI498166B (zh) | 以表面電漿共振定量分析之自動操作檢測程序的多孔性薄膜微流體裝置 | |
| JPWO2001090754A1 (ja) | バイオセンサ | |
| WO2009068583A2 (en) | Separation and detection device with means for optimization of the capillary drag force | |
| CN211905402U (zh) | 检测芯片及检测系统 | |
| US20210055284A1 (en) | Microchip immunoassay device having precise incubation time control and signal scaling and related methods | |
| JP6779433B2 (ja) | 検出装置及び検出方法 | |
| CN105445454A (zh) | 一种可定量的免疫层析装置 | |
| EP4006553A1 (en) | Blood group antigen testing component | |
| CN211014324U (zh) | 检测芯片及检测系统 | |
| JP5339552B2 (ja) | 体液成分の分析器具 | |
| WO2006080438A1 (ja) | イムノクロマト用試験具およびこれを用いた半定量方法 | |
| EP4071463B1 (en) | Reaction test paper, detection chip and detection system | |
| JP5224276B2 (ja) | 体液成分の分析器具 | |
| CN212989175U (zh) | 一种复合检测芯片 | |
| CN112088308A (zh) | 利用免疫化学诊断方法的靶抗原检测用纸基三维结构的微芯片及利用其的靶抗原检测方法 | |
| US20240157355A1 (en) | Interface to lateral flow | |
| EP1717585A1 (en) | Microchip and analysis method using the same | |
| JP6043990B2 (ja) | 体液試料移送機構および体液試料移送方法、ならびに体液成分分析装置および体液成分分析方法 | |
| JP7337371B2 (ja) | イムノクロマトグラフィー測定装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20130716 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20130802 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5339552 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313117 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |