JP2010042272A - 一部が蛋白質に結合した物質を取除く装置 - Google Patents
一部が蛋白質に結合した物質を取除く装置 Download PDFInfo
- Publication number
- JP2010042272A JP2010042272A JP2009231605A JP2009231605A JP2010042272A JP 2010042272 A JP2010042272 A JP 2010042272A JP 2009231605 A JP2009231605 A JP 2009231605A JP 2009231605 A JP2009231605 A JP 2009231605A JP 2010042272 A JP2010042272 A JP 2010042272A
- Authority
- JP
- Japan
- Prior art keywords
- blood
- fluid
- flow rate
- carrier
- dialysis fluid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000000126 substance Substances 0.000 title claims abstract description 50
- 102000004169 proteins and genes Human genes 0.000 title description 12
- 108090000623 proteins and genes Proteins 0.000 title description 12
- 210000004369 blood Anatomy 0.000 claims abstract description 134
- 239000008280 blood Substances 0.000 claims abstract description 134
- 239000012530 fluid Substances 0.000 claims abstract description 62
- 108010088751 Albumins Proteins 0.000 claims abstract description 36
- 102000009027 Albumins Human genes 0.000 claims abstract description 36
- 230000017531 blood circulation Effects 0.000 claims abstract description 32
- 238000012546 transfer Methods 0.000 claims abstract description 25
- 239000012528 membrane Substances 0.000 claims description 58
- 238000000746 purification Methods 0.000 claims description 33
- 239000000463 material Substances 0.000 claims description 23
- 210000002966 serum Anatomy 0.000 claims description 4
- 238000000502 dialysis Methods 0.000 abstract description 31
- 239000000969 carrier Substances 0.000 abstract description 4
- 238000004140 cleaning Methods 0.000 abstract description 4
- 239000000385 dialysis solution Substances 0.000 description 111
- 238000000108 ultra-filtration Methods 0.000 description 27
- 238000000034 method Methods 0.000 description 16
- 230000000694 effects Effects 0.000 description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 14
- 210000004027 cell Anatomy 0.000 description 12
- 239000012141 concentrate Substances 0.000 description 10
- 210000002381 plasma Anatomy 0.000 description 10
- 238000001631 haemodialysis Methods 0.000 description 9
- 230000000322 hemodialysis Effects 0.000 description 9
- 238000002615 hemofiltration Methods 0.000 description 9
- 238000002156 mixing Methods 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- 238000011282 treatment Methods 0.000 description 8
- 239000003792 electrolyte Substances 0.000 description 7
- 230000009471 action Effects 0.000 description 6
- 238000010586 diagram Methods 0.000 description 6
- 210000001124 body fluid Anatomy 0.000 description 5
- 239000010839 body fluid Substances 0.000 description 5
- 239000000243 solution Substances 0.000 description 5
- 238000001179 sorption measurement Methods 0.000 description 5
- 230000008859 change Effects 0.000 description 4
- 230000008878 coupling Effects 0.000 description 4
- 238000010168 coupling process Methods 0.000 description 4
- 238000005859 coupling reaction Methods 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 239000003053 toxin Substances 0.000 description 4
- 231100000765 toxin Toxicity 0.000 description 4
- 108700012359 toxins Proteins 0.000 description 4
- 239000002699 waste material Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 238000013459 approach Methods 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 102000001554 Hemoglobins Human genes 0.000 description 2
- 108010054147 Hemoglobins Proteins 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- 239000004952 Polyamide Substances 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 230000036772 blood pressure Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 230000001276 controlling effect Effects 0.000 description 2
- DDRJAANPRJIHGJ-UHFFFAOYSA-N creatinine Chemical compound CN1CC(=O)NC1=N DDRJAANPRJIHGJ-UHFFFAOYSA-N 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 230000009977 dual effect Effects 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 238000011221 initial treatment Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229920002492 poly(sulfone) Polymers 0.000 description 2
- 229920002647 polyamide Polymers 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 2
- SVUOLADPCWQTTE-UHFFFAOYSA-N 1h-1,2-benzodiazepine Chemical compound N1N=CC=CC2=CC=CC=C12 SVUOLADPCWQTTE-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- XFXPMWWXUTWYJX-UHFFFAOYSA-N Cyanide Chemical compound N#[C-] XFXPMWWXUTWYJX-UHFFFAOYSA-N 0.000 description 1
- XUIIKFGFIJCVMT-GFCCVEGCSA-N D-thyroxine Chemical compound IC1=CC(C[C@@H](N)C(O)=O)=CC(I)=C1OC1=CC(I)=C(O)C(I)=C1 XUIIKFGFIJCVMT-GFCCVEGCSA-N 0.000 description 1
- 206010012335 Dependence Diseases 0.000 description 1
- WDJUZGPOPHTGOT-OAXVISGBSA-N Digitoxin Natural products O([C@H]1[C@@H](C)O[C@@H](O[C@@H]2C[C@@H]3[C@@](C)([C@@H]4[C@H]([C@]5(O)[C@@](C)([C@H](C6=CC(=O)OC6)CC5)CC4)CC3)CC2)C[C@H]1O)[C@H]1O[C@@H](C)[C@H](O[C@H]2O[C@@H](C)[C@@H](O)[C@@H](O)C2)[C@@H](O)C1 WDJUZGPOPHTGOT-OAXVISGBSA-N 0.000 description 1
- LTMHDMANZUZIPE-AMTYYWEZSA-N Digoxin Natural products O([C@H]1[C@H](C)O[C@H](O[C@@H]2C[C@@H]3[C@@](C)([C@@H]4[C@H]([C@]5(O)[C@](C)([C@H](O)C4)[C@H](C4=CC(=O)OC4)CC5)CC3)CC2)C[C@@H]1O)[C@H]1O[C@H](C)[C@@H](O[C@H]2O[C@@H](C)[C@H](O)[C@@H](O)C2)[C@@H](O)C1 LTMHDMANZUZIPE-AMTYYWEZSA-N 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 206010036790 Productive cough Diseases 0.000 description 1
- 206010065604 Suicidal behaviour Diseases 0.000 description 1
- 229940123445 Tricyclic antidepressant Drugs 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 238000005349 anion exchange Methods 0.000 description 1
- -1 aromatic amino acids Chemical class 0.000 description 1
- 229940049706 benzodiazepine Drugs 0.000 description 1
- BPYKTIZUTYGOLE-UHFFFAOYSA-N billirubin-IXalpha Natural products N1C(=O)C(C)=C(C=C)C1=CC1=C(C)C(CCC(O)=O)=C(CC2=C(C(C)=C(C=C3C(=C(C=C)C(=O)N3)C)N2)CCC(O)=O)N1 BPYKTIZUTYGOLE-UHFFFAOYSA-N 0.000 description 1
- 239000013060 biological fluid Substances 0.000 description 1
- 230000036760 body temperature Effects 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 229910002091 carbon monoxide Inorganic materials 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 229940109239 creatinine Drugs 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000007872 degassing Methods 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000009795 derivation Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 229960000648 digitoxin Drugs 0.000 description 1
- WDJUZGPOPHTGOT-XUDUSOBPSA-N digitoxin Chemical compound C1[C@H](O)[C@H](O)[C@@H](C)O[C@H]1O[C@@H]1[C@@H](C)O[C@@H](O[C@@H]2[C@H](O[C@@H](O[C@@H]3C[C@@H]4[C@]([C@@H]5[C@H]([C@]6(CC[C@@H]([C@@]6(C)CC5)C=5COC(=O)C=5)O)CC4)(C)CC3)C[C@@H]2O)C)C[C@@H]1O WDJUZGPOPHTGOT-XUDUSOBPSA-N 0.000 description 1
- 229960005156 digoxin Drugs 0.000 description 1
- LTMHDMANZUZIPE-PUGKRICDSA-N digoxin Chemical compound C1[C@H](O)[C@H](O)[C@@H](C)O[C@H]1O[C@@H]1[C@@H](C)O[C@@H](O[C@@H]2[C@H](O[C@@H](O[C@@H]3C[C@@H]4[C@]([C@@H]5[C@H]([C@]6(CC[C@@H]([C@@]6(C)[C@H](O)C5)C=5COC(=O)C=5)O)CC4)(C)CC3)C[C@@H]2O)C)C[C@@H]1O LTMHDMANZUZIPE-PUGKRICDSA-N 0.000 description 1
- LTMHDMANZUZIPE-UHFFFAOYSA-N digoxine Natural products C1C(O)C(O)C(C)OC1OC1C(C)OC(OC2C(OC(OC3CC4C(C5C(C6(CCC(C6(C)C(O)C5)C=5COC(=O)C=5)O)CC4)(C)CC3)CC2O)C)CC1O LTMHDMANZUZIPE-UHFFFAOYSA-N 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 206010013663 drug dependence Diseases 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 230000009931 harmful effect Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 230000003907 kidney function Effects 0.000 description 1
- 230000003908 liver function Effects 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000002636 mycotoxin Substances 0.000 description 1
- 238000001139 pH measurement Methods 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 231100000572 poisoning Toxicity 0.000 description 1
- 230000000607 poisoning effect Effects 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920002239 polyacrylonitrile Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000011076 safety test Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 210000003802 sputum Anatomy 0.000 description 1
- 208000024794 sputum Diseases 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229940034208 thyroxine Drugs 0.000 description 1
- XUIIKFGFIJCVMT-UHFFFAOYSA-N thyroxine-binding globulin Natural products IC1=CC(CC([NH3+])C([O-])=O)=CC(I)=C1OC1=CC(I)=C(O)C(I)=C1 XUIIKFGFIJCVMT-UHFFFAOYSA-N 0.000 description 1
- 239000003029 tricyclic antidepressant agent Substances 0.000 description 1
- 229960004799 tryptophan Drugs 0.000 description 1
- 229940005605 valeric acid Drugs 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1601—Control or regulation
- A61M1/1603—Regulation parameters
- A61M1/1605—Physical characteristics of the dialysate fluid
- A61M1/1607—Physical characteristics of the dialysate fluid before use, i.e. upstream of dialyser
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1621—Constructional aspects thereof
- A61M1/1633—Constructional aspects thereof with more than one dialyser unit
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1621—Constructional aspects thereof
- A61M1/1647—Constructional aspects thereof with flow rate measurement of the dialysis fluid, upstream and downstream of the dialyser
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1621—Constructional aspects thereof
- A61M1/165—Constructional aspects thereof with a dialyser bypass on the dialysis fluid line
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1692—Detection of blood traces in dialysate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/14—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis
- A61M1/16—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes
- A61M1/1694—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes with recirculating dialysing liquid
- A61M1/1696—Dialysis systems; Artificial kidneys; Blood oxygenators ; Reciprocating systems for treatment of body fluids, e.g. single needle systems for hemofiltration or pheresis with membranes with recirculating dialysing liquid with dialysate regeneration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/34—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration
- A61M1/3403—Regulation parameters
- A61M1/341—Regulation parameters by measuring the filtrate rate or volume
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/34—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration
- A61M1/342—Adding solutions to the blood, e.g. substitution solutions
- A61M1/3424—Substitution fluid path
- A61M1/3431—Substitution fluid path upstream of the filter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/34—Filtering material out of the blood by passing it through a membrane, i.e. hemofiltration or diafiltration
- A61M1/342—Adding solutions to the blood, e.g. substitution solutions
- A61M1/3455—Substitution fluids
- A61M1/3465—Substitution fluids using dialysate as substitution fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M1/00—Suction or pumping devices for medical purposes; Devices for carrying-off, for treatment of, or for carrying-over, body-liquids; Drainage systems
- A61M1/36—Other treatment of blood in a by-pass of the natural circulatory system, e.g. temperature adaptation, irradiation ; Extra-corporeal blood circuits
- A61M1/3621—Extra-corporeal blood circuits
- A61M1/3623—Means for actively controlling temperature of blood
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D61/00—Processes of separation using semi-permeable membranes, e.g. dialysis, osmosis or ultrafiltration; Apparatus, accessories or auxiliary operations specially adapted therefor
- B01D61/24—Dialysis ; Membrane extraction
- B01D61/243—Dialysis
- B01D61/244—Dialysis comprising multiple dialysis steps
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D61/00—Processes of separation using semi-permeable membranes, e.g. dialysis, osmosis or ultrafiltration; Apparatus, accessories or auxiliary operations specially adapted therefor
- B01D61/24—Dialysis ; Membrane extraction
- B01D61/28—Apparatus therefor
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3317—Electromagnetic, inductive or dielectric measuring means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3324—PH measuring means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3331—Pressure; Flow
- A61M2205/3334—Measuring or controlling the flow rate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3368—Temperature
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3379—Masses, volumes, levels of fluids in reservoirs, flow rates
- A61M2205/3393—Masses, volumes, levels of fluids in reservoirs, flow rates by weighing the reservoir
Landscapes
- Health & Medical Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Urology & Nephrology (AREA)
- Engineering & Computer Science (AREA)
- Vascular Medicine (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Hematology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Emergency Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Water Supply & Treatment (AREA)
- Physics & Mathematics (AREA)
- Fluid Mechanics (AREA)
- Cardiology (AREA)
- External Artificial Organs (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
Abstract
【解決手段】浄化流体流量と濾過器の物質移動係数koAとを、従来の透析器に比較して大幅に増大させ、浄化流体流量が少なくとも2000ml/分であり、物質移動係数koAが少なくとも2000ml/分である。また、濾過器の物質移動係数koAと血液流量との間の比率は少なくとも5であり、浄化流体流量と血液流量との間の比率が少なくとも5である構成とする。
【選択図】図1
Description
ここで、eは指数関数を示し、指数fは数2で計算される。
物質の大部分が担体に結合し、濃度勾配が小さな場合には、移送を向上させるように膜自体を改善すれば良好な結果が達成される。これは米国特許第5744042号に示唆されており、それによれば膜はその内外両面をアルブミンで被覆するようにアルブミンを下塗りされており、アルブミンは表面に付着されている。したがって、膜を横断して移送させるための仲介部分として作用することのできる場所が膜内部に形成される。
本発明のさらに他の実施例では、約4000ml/分の大流量の透析流体が約4000ml/分のkoAを有する大きな膜と、約5g/lのアルブミン血清のような担体の濃度と組合わされる。この実施例は高価なアルブミンの濃度が制限され、透析処置の効果が許容レベルに保たれるので有利である。
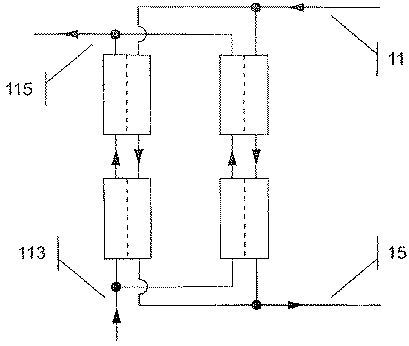
1. 血液回路と、流体回路と、流体室を血液室から隔離する半透膜を有する濾過器とを含み、血液および浄化流体を混合し、前記混合液を血液室に通して導くための手段と、流体室への限外濾過が生じるように膜を隔てて圧力勾配を与える手段とを備え、流体室は浄化流体流量と患者の望ましい重量減との合計に寸法が等しく構成され、血液から一部が担体に結合した物質を除去するための装置であって、
濾過器の水透過係数LpAが少なくとも10ml/分/mmHgであり、
浄化流体流量が少なくとも1000ml/分であり、
浄化流体流量と血液流量との間の比率が少なくとも5であることを特徴とする装置。
2. 濾過器が直列または並列、またはその組合せで配置された幾つかの濾過器によって置き換えられた前記1項又は特許請求の範囲の請求項1から11までのいずれか一項に記載された装置。
3. 血液が患者に戻される前に血液を加熱するためにヒーターが配置された前記1項又は2項、又は特許請求の範囲の請求項1から11までのいずれか一項に記載された装置。
4. 血液が患者に戻される前の血液の通路に沿う最終の透析器がヒーターである前記3項に記載された装置。
Claims (11)
- 血液回路と、流体回路と、流体室を血液室から隔離する半透膜を有する濾過器とを含み、血液は血液室を通して導かれ、浄化流体は流体室を通して導かれるように構成され、血液から一部が担体に結合した物質を除去するための装置であって、
前記血液から前記各担体に結合した物質を除去するための濾過器の物質移動係数koAが少なくとも2000ml/分であり、
濾過器の物質移動係数koAと血液流量との間の比率が少なくとも5であり、
浄化流体流量が少なくとも2000ml/分であり、
浄化流体流量と血液流量との間の比率が少なくとも5であることを特徴とする装置。 - 濾過器の物質移動係数koAと血液流量との間の比率が少なくとも10であり、
浄化流体流量と血液流量との間の比率が少なくとも10である請求項1に記載された装置。 - 濾過器の物質移動係数koAが少なくとも5000ml/分であり、
浄化流体流量が少なくとも5000ml/分である請求項1または請求項2に記載された装置。 - 血液の流量Qbと、血漿中に溶解した分屑に関する除去すべき物質の全量を表す係数αとの積に関してパラメータが選ばれ、
濾過器の物質移動係数koAがこの積の値の少なくとも10%であり、
浄化流体流量がこの積の値の少なくとも10%である請求項1、請求項2または請求項3に記載された装置。 - 濾過器の物質移動係数koAがこの積の値の少なくとも100%であり、および(または)
浄化流体流量がこの積の値の少なくとも100%である請求項4に記載された装置。 - 血液回路と、流体回路と、流体室を血液室から隔離する半透膜を有する濾過器とを含み、血液が血液室を通して導かれ、浄化流体が流体室を通して導かれるように構成され、血液から一部が担体に結合した物質を除去するための装置であって、
前記血液から前記各担体に結合した物質を除去するための濾過器の物質移動係数koAが少なくとも2000ml/分であり、
濾過器の物質移動係数koAと血液流量との間の比率が少なくとも5であり、
一部が担体に結合している血液中の物質に結合することのできる担体を浄化流体が含んでいることを特徴とする装置。 - 血液回路と、流体回路と、流体室を血液室から隔離する半透膜を有する濾過器とを含み、血液が血液室を通して導かれ、浄化流体が流体室を通して導かれるように構成され、血液から一部が担体に結合した物質を除去するための装置であって、
一部が担体に結合している血液中の物質に結合することのできる担体を含む流体によって膜が事前処理されており、
浄化流体流量が少なくとも2000ml/分であり、
浄化流体流量と血液流量との間の比率が少なくとも10であることを特徴とする装置。 - 一部が担体に結合している血液中の物質に結合することのできる担体を含む流体で膜が事前処理されている請求項6に記載された装置。
- 一部が担体に結合している血液中の物質に結合することのできる担体を浄化流体が含んでいる請求項7に記載された装置。
- 担体がアルブミン血清である請求項6、請求項7、請求項8または請求項9に記載された装置。
- アルブミン血清の濃度が10g/lを超える請求項10に記載された装置。
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SE0200956A SE0200956D0 (sv) | 2002-03-27 | 2002-03-27 | Method and device for the removal of protein bound substances |
| SE0200956-1 | 2002-03-27 | ||
| SE0201317-5 | 2002-05-02 | ||
| SE0201317A SE522692C2 (sv) | 2002-03-27 | 2002-05-02 | Förfarande och anordning för avlägsnande av delvis proteinbundna substanser |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003577978A Division JP4456367B2 (ja) | 2002-03-27 | 2003-03-25 | 一部が蛋白質に結合した物質を取除く方法および装置 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013196882A Division JP5711330B2 (ja) | 2002-03-27 | 2013-09-24 | 一部が蛋白質に結合した物質を取除く装置 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010042272A true JP2010042272A (ja) | 2010-02-25 |
| JP5438456B2 JP5438456B2 (ja) | 2014-03-12 |
Family
ID=26655692
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003577978A Expired - Lifetime JP4456367B2 (ja) | 2002-03-27 | 2003-03-25 | 一部が蛋白質に結合した物質を取除く方法および装置 |
| JP2009231605A Expired - Lifetime JP5438456B2 (ja) | 2002-03-27 | 2009-10-05 | 一部が蛋白質に結合した物質を取除く装置 |
| JP2013196882A Expired - Lifetime JP5711330B2 (ja) | 2002-03-27 | 2013-09-24 | 一部が蛋白質に結合した物質を取除く装置 |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003577978A Expired - Lifetime JP4456367B2 (ja) | 2002-03-27 | 2003-03-25 | 一部が蛋白質に結合した物質を取除く方法および装置 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013196882A Expired - Lifetime JP5711330B2 (ja) | 2002-03-27 | 2013-09-24 | 一部が蛋白質に結合した物質を取除く装置 |
Country Status (11)
| Country | Link |
|---|---|
| US (3) | US7615158B2 (ja) |
| EP (1) | EP1490129B1 (ja) |
| JP (3) | JP4456367B2 (ja) |
| KR (1) | KR101044980B1 (ja) |
| CN (1) | CN100502959C (ja) |
| AT (1) | ATE441444T1 (ja) |
| AU (1) | AU2003214752A1 (ja) |
| DE (1) | DE60329088D1 (ja) |
| ES (1) | ES2333002T3 (ja) |
| SE (1) | SE522692C2 (ja) |
| WO (1) | WO2003080154A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012019957A (ja) * | 2010-07-14 | 2012-02-02 | Asahi Kasei Kuraray Medical Co Ltd | 血液透析システム及び透析液流速度の算出方法 |
Families Citing this family (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7160719B2 (en) * | 2002-06-07 | 2007-01-09 | Mayo Foundation For Medical Education And Research | Bioartificial liver system |
| ES2447296T3 (es) | 2006-10-13 | 2014-03-11 | Bluesky Medical Group Incorporated | Control de presión de una bomba de vacío médica |
| GB0621452D0 (en) * | 2006-10-27 | 2006-12-06 | Ucl Business Plc | Therapy for liver disease |
| GB0715211D0 (en) * | 2007-08-06 | 2007-09-12 | Smith & Nephew | Apparatus |
| US9408954B2 (en) | 2007-07-02 | 2016-08-09 | Smith & Nephew Plc | Systems and methods for controlling operation of negative pressure wound therapy apparatus |
| GB0715259D0 (en) | 2007-08-06 | 2007-09-12 | Smith & Nephew | Canister status determination |
| US12121648B2 (en) | 2007-08-06 | 2024-10-22 | Smith & Nephew Plc | Canister status determination |
| US8672869B2 (en) * | 2007-10-30 | 2014-03-18 | Bellco S.R.L. | Kit, system and method of treating myeloma patients |
| US8206591B2 (en) * | 2008-10-16 | 2012-06-26 | Fresenius Medical Care Holdings, Inc. | Method of removing protein-bound deleterious substances during extracorporeal renal replacement treatment |
| CN102421467B (zh) * | 2009-03-13 | 2015-04-22 | 梅约医学教育与研究基金会 | 生物人工肝 |
| EP2380610B1 (en) * | 2010-04-20 | 2014-05-07 | Gambro Lundia AB | High cut-off hemodialysis membrane for use in liver dialysis |
| JP6001660B2 (ja) | 2011-08-02 | 2016-10-05 | メドトロニック,インコーポレイテッド | 制御された追従的な容積を有する流路を有する血液透析システム |
| EP2744537B1 (en) | 2011-08-16 | 2018-01-24 | Medtronic, Inc. | Modular hemodialysis system |
| EP2730303B1 (en) * | 2012-11-08 | 2015-08-12 | Gambro Lundia AB | Albumin pump control in a MARS treatment apparatus |
| CN104394903B (zh) | 2012-12-14 | 2017-08-15 | 甘布罗伦迪亚股份公司 | 生物流体的净化 |
| US10543052B2 (en) | 2013-02-01 | 2020-01-28 | Medtronic, Inc. | Portable dialysis cabinet |
| US9623164B2 (en) | 2013-02-01 | 2017-04-18 | Medtronic, Inc. | Systems and methods for multifunctional volumetric fluid control |
| US10850016B2 (en) | 2013-02-01 | 2020-12-01 | Medtronic, Inc. | Modular fluid therapy system having jumpered flow paths and systems and methods for cleaning and disinfection |
| US10010663B2 (en) | 2013-02-01 | 2018-07-03 | Medtronic, Inc. | Fluid circuit for delivery of renal replacement therapies |
| DE102013112038A1 (de) * | 2013-10-31 | 2015-04-30 | Fresenius Medical Care AG & Co. KGaA | Blutbehandlungsvorrichtung mit erhöhter Patientensicherheit und Verfahren |
| US11033673B2 (en) | 2014-02-17 | 2021-06-15 | Humanity Life Extension Llc | Systems and methods for treating blood |
| US10098993B2 (en) * | 2014-12-10 | 2018-10-16 | Medtronic, Inc. | Sensing and storage system for fluid balance |
| US9895479B2 (en) | 2014-12-10 | 2018-02-20 | Medtronic, Inc. | Water management system for use in dialysis |
| US9713665B2 (en) | 2014-12-10 | 2017-07-25 | Medtronic, Inc. | Degassing system for dialysis |
| US10874787B2 (en) | 2014-12-10 | 2020-12-29 | Medtronic, Inc. | Degassing system for dialysis |
| US9950103B2 (en) * | 2016-03-03 | 2018-04-24 | Micromedics Inc. | Combination kidney and liver dialysis system and method |
| CN105688301A (zh) * | 2016-03-08 | 2016-06-22 | 刘瑞清 | 血液净化用漏血监测系统 |
| US10456515B2 (en) * | 2016-06-06 | 2019-10-29 | The Trustees Of Columbia University In The City Of New York | Wearable ultrafiltration devices methods and systems |
| DE102016007828A1 (de) * | 2016-06-29 | 2018-01-04 | Fresenius Medical Care Deutschland Gmbh | Steuereinheit zur Bluterkennung in einer Dialysierflüssigkeitsabführleitung einer Blutbehandlungsvorrichtung und Blutbehandlungsvorrichtung |
| US11278654B2 (en) | 2017-12-07 | 2022-03-22 | Medtronic, Inc. | Pneumatic manifold for a dialysis system |
| US11033667B2 (en) | 2018-02-02 | 2021-06-15 | Medtronic, Inc. | Sorbent manifold for a dialysis system |
| US11110215B2 (en) | 2018-02-23 | 2021-09-07 | Medtronic, Inc. | Degasser and vent manifolds for dialysis |
| US11147907B2 (en) | 2018-12-10 | 2021-10-19 | Humanity Life Extension Llc | Systems and methods for treating blood |
| US12128165B2 (en) | 2020-04-27 | 2024-10-29 | Mozarc Medical Us Llc | Dual stage degasser |
| EP3991770B1 (en) | 2020-10-30 | 2024-10-30 | Bellco S.r.l. | Dialysis cassette with pump features |
| US12318528B2 (en) | 2020-10-30 | 2025-06-03 | Mozarc Medical Us Llc | Variable orifice fistula graft |
| EP4008376A1 (en) | 2020-12-03 | 2022-06-08 | Medtronic, Inc. | Flexible tube routing accessory for peritoneal dialysis system |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4000072A (en) * | 1973-09-19 | 1976-12-28 | Takeda Chemical Industries, Ltd. | Artificial kidney apparatus |
| JPS53105894A (en) * | 1977-02-26 | 1978-09-14 | Musashi Eng Kk | Artificial kidney with recycle circuit |
| JPH02258035A (ja) * | 1981-11-14 | 1990-10-18 | Toyobo Co Ltd | 透析用中空繊維膜及びその製造方法 |
| JPH07506765A (ja) * | 1993-03-19 | 1995-07-27 | テラクリン アクチエンゲゼルシャフト | 蛋白質含有液から蛋白質−結合物質を透析により分離する膜と方法 |
Family Cites Families (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4191182A (en) * | 1977-09-23 | 1980-03-04 | Hemotherapy Inc. | Method and apparatus for continuous plasmaphersis |
| DE2838414C2 (de) * | 1978-09-02 | 1984-10-31 | Fresenius AG, 6380 Bad Homburg | Vorrichtung zur Hämodialyse und zum Entziehen von Ultrafiltrat |
| US4367040A (en) * | 1979-05-29 | 1983-01-04 | Tokyo Shibaura Denki Kabushiki Kaisha | Multi-channel optical sensing system |
| JPS5838563A (ja) * | 1981-08-31 | 1983-03-07 | 中川 成之輔 | 中空糸型血液透析装置 |
| US4435289A (en) * | 1981-12-23 | 1984-03-06 | Romicon, Inc. | Series ultrafiltration with pressurized permeate |
| DE3313421C2 (de) * | 1983-04-13 | 1985-08-08 | Fresenius AG, 6380 Bad Homburg | Einrichtung zum Regeln der Ultrafiltrationsrate bei Vorrichtungen zum extrakorporalen Reinigen von Blut |
| US4906375A (en) * | 1984-07-14 | 1990-03-06 | Fresenius, Ag | Asymmetrical microporous hollow fiber for hemodialysis |
| DE3444671A1 (de) * | 1984-12-07 | 1986-06-12 | Fresenius AG, 6380 Bad Homburg | Haemodiafiltrationsgeraet |
| SE453361B (sv) * | 1985-06-04 | 1988-02-01 | Gambro Ab | System for styrning av en medicinsk behandling, t ex dialys |
| US4784495A (en) * | 1987-02-06 | 1988-11-15 | Gambro Ab | System for preparing a fluid intended for a medical procedure by mixing at least one concentrate in powder form with water |
| IT1215765B (it) * | 1988-01-22 | 1990-02-22 | Grace W R & Co | Dispositivo di emodiafiltrazione erelativo procedimento di emodiafiltrazione. |
| US5762798A (en) * | 1991-04-12 | 1998-06-09 | Minntech Corporation | Hollow fiber membranes and method of manufacture |
| US5536412A (en) * | 1992-02-06 | 1996-07-16 | Hemocleanse, Inc. | Hemofiltration and plasmafiltration devices and methods |
| FR2723002B1 (fr) * | 1994-07-26 | 1996-09-06 | Hospal Ind | Dispositif et procede pour preparer un liquide de traitement par filtration |
| US5682584A (en) * | 1994-07-28 | 1997-10-28 | Minolta Co., Ltd. | Developer mixing and transporting device |
| US5626759A (en) * | 1994-08-01 | 1997-05-06 | Regents Of The University Of Colorado | Blood treatment device with moving membrane |
| US6117100A (en) * | 1997-06-06 | 2000-09-12 | Powers; Kathleen M. | Hemodialysis-double dialyzers in parallel |
| JP4115626B2 (ja) | 1998-05-25 | 2008-07-09 | 旭化成クラレメディカル株式会社 | 中空糸膜型血液透析器の製造方法 |
| JP3934340B2 (ja) * | 1998-11-09 | 2007-06-20 | 旭化成メディカル株式会社 | 血液浄化器 |
| JP4211168B2 (ja) * | 1999-12-21 | 2009-01-21 | 東レ株式会社 | 透析器の製造方法および滅菌法 |
| US6716356B2 (en) * | 2000-01-11 | 2004-04-06 | Nephros, Inc. | Thermally enhanced dialysis/diafiltration system |
| US6572641B2 (en) * | 2001-04-09 | 2003-06-03 | Nxstage Medical, Inc. | Devices for warming fluid and methods of use |
| US8029454B2 (en) * | 2003-11-05 | 2011-10-04 | Baxter International Inc. | High convection home hemodialysis/hemofiltration and sorbent system |
-
2002
- 2002-05-02 SE SE0201317A patent/SE522692C2/sv not_active IP Right Cessation
-
2003
- 2003-03-25 KR KR1020047013920A patent/KR101044980B1/ko not_active Expired - Lifetime
- 2003-03-25 AT AT03710580T patent/ATE441444T1/de not_active IP Right Cessation
- 2003-03-25 US US10/500,478 patent/US7615158B2/en not_active Expired - Lifetime
- 2003-03-25 CN CNB038037572A patent/CN100502959C/zh not_active Expired - Lifetime
- 2003-03-25 DE DE60329088T patent/DE60329088D1/de not_active Expired - Lifetime
- 2003-03-25 AU AU2003214752A patent/AU2003214752A1/en not_active Abandoned
- 2003-03-25 ES ES03710580T patent/ES2333002T3/es not_active Expired - Lifetime
- 2003-03-25 JP JP2003577978A patent/JP4456367B2/ja not_active Expired - Lifetime
- 2003-03-25 EP EP03710580A patent/EP1490129B1/en not_active Expired - Lifetime
- 2003-03-25 WO PCT/SE2003/000497 patent/WO2003080154A1/en not_active Ceased
-
2009
- 2009-09-28 US US12/567,994 patent/US7879242B2/en not_active Expired - Lifetime
- 2009-10-05 JP JP2009231605A patent/JP5438456B2/ja not_active Expired - Lifetime
-
2010
- 2010-12-22 US US12/976,653 patent/US8048310B2/en not_active Expired - Fee Related
-
2013
- 2013-09-24 JP JP2013196882A patent/JP5711330B2/ja not_active Expired - Lifetime
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4000072A (en) * | 1973-09-19 | 1976-12-28 | Takeda Chemical Industries, Ltd. | Artificial kidney apparatus |
| JPS53105894A (en) * | 1977-02-26 | 1978-09-14 | Musashi Eng Kk | Artificial kidney with recycle circuit |
| JPH02258035A (ja) * | 1981-11-14 | 1990-10-18 | Toyobo Co Ltd | 透析用中空繊維膜及びその製造方法 |
| JPH07506765A (ja) * | 1993-03-19 | 1995-07-27 | テラクリン アクチエンゲゼルシャフト | 蛋白質含有液から蛋白質−結合物質を透析により分離する膜と方法 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012019957A (ja) * | 2010-07-14 | 2012-02-02 | Asahi Kasei Kuraray Medical Co Ltd | 血液透析システム及び透析液流速度の算出方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4456367B2 (ja) | 2010-04-28 |
| US20050115898A1 (en) | 2005-06-02 |
| JP5438456B2 (ja) | 2014-03-12 |
| JP5711330B2 (ja) | 2015-04-30 |
| US7615158B2 (en) | 2009-11-10 |
| DE60329088D1 (de) | 2009-10-15 |
| KR101044980B1 (ko) | 2011-06-29 |
| SE0201317D0 (sv) | 2002-05-02 |
| ES2333002T3 (es) | 2010-02-16 |
| US20110089112A1 (en) | 2011-04-21 |
| ATE441444T1 (de) | 2009-09-15 |
| EP1490129A1 (en) | 2004-12-29 |
| CN100502959C (zh) | 2009-06-24 |
| WO2003080154A1 (en) | 2003-10-02 |
| JP2014028290A (ja) | 2014-02-13 |
| JP2005520643A (ja) | 2005-07-14 |
| US20100044313A1 (en) | 2010-02-25 |
| EP1490129B1 (en) | 2009-09-02 |
| SE0201317L (sv) | 2003-09-28 |
| AU2003214752A1 (en) | 2003-10-08 |
| KR20040093102A (ko) | 2004-11-04 |
| CN1630540A (zh) | 2005-06-22 |
| US8048310B2 (en) | 2011-11-01 |
| SE522692C2 (sv) | 2004-03-02 |
| US7879242B2 (en) | 2011-02-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5711330B2 (ja) | 一部が蛋白質に結合した物質を取除く装置 | |
| JP4729225B2 (ja) | 熱促進型透析/濾過透析システム | |
| EP1809410B1 (en) | Ionic enhanced dialysis/disfiltration system | |
| JP4436569B2 (ja) | 浸透圧差による透過型濾過システム | |
| EP2452705B1 (en) | Device for the treatment of blood with selective extraction of solutes | |
| JP2002521162A (ja) | 効率的な血液透析濾過法 | |
| EA029566B1 (ru) | Устройство для гемодиафильтрации | |
| NL8402870A (nl) | Werkwijze en inrichting voor het behandelen van bloedplasma. | |
| EA018353B1 (ru) | Устройство для диализа | |
| JP3353466B2 (ja) | 血液透析装置の透析液管理装置 | |
| JPS63139561A (ja) | 血液の浄化方法および装置 | |
| JPS5846962A (ja) | 血液処理用装置 | |
| JP3413985B2 (ja) | 選択分離膜およびそれを用いた血液処理装置 | |
| JP6337175B2 (ja) | 急性又は慢性腎不全患者の血液からタンパク質結合毒素を除去するための透析法に使用される医薬組成物。 | |
| JPH07313591A (ja) | 血液透析装置の透析液管理装置 | |
| UA46256A (uk) | Спосіб збільшення елімінації токсичних речовин через мембрану діалізатора |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20111021 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20120123 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20120126 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120215 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120803 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20121105 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20121108 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20121203 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20121206 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130104 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130109 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130129 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20130524 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130924 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20131125 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20131210 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20131213 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5438456 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| EXPY | Cancellation because of completion of term |