EP2018418B1 - Substrat für das wachstum kultivierter zellen in drei dimensionen - Google Patents
Substrat für das wachstum kultivierter zellen in drei dimensionen Download PDFInfo
- Publication number
- EP2018418B1 EP2018418B1 EP07732504.1A EP07732504A EP2018418B1 EP 2018418 B1 EP2018418 B1 EP 2018418B1 EP 07732504 A EP07732504 A EP 07732504A EP 2018418 B1 EP2018418 B1 EP 2018418B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- cells
- cell
- cell culture
- agent
- substrate according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Not-in-force
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/0068—General culture methods using substrates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12M—APPARATUS FOR ENZYMOLOGY OR MICROBIOLOGY; APPARATUS FOR CULTURING MICROORGANISMS FOR PRODUCING BIOMASS, FOR GROWING CELLS OR FOR OBTAINING FERMENTATION OR METABOLIC PRODUCTS, i.e. BIOREACTORS OR FERMENTERS
- C12M25/00—Means for supporting, enclosing or fixing the microorganisms, e.g. immunocoatings
- C12M25/14—Scaffolds; Matrices
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/067—Hepatocytes
- C12N5/0671—Three-dimensional culture, tissue culture or organ culture; Encapsulated cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2533/00—Supports or coatings for cell culture, characterised by material
- C12N2533/30—Synthetic polymers
Definitions
- the invention relates to a cell culture substrate comprising a polymerised high internal phase emulsion polymer (polyHIPE) adapted for installation and use in existing cell culture plastic-ware for the growth of cells, typically mammalian cells and the use of the substrate in a cell culture system for analysis of proliferation, differentiation and function of cells.
- polyHIPE polymerised high internal phase emulsion polymer
- eukaryotic cells for example mammalian cells
- cell culture of mammalian cells requires a sterile vessel, usually manufactured from plastics (typically polystyrene), defined growth medium and, in some examples, feeder cells and serum, typically calf serum.
- the feeder cells function to provide signals which stimulate cell proliferation and/or maintain cells in an undifferentiated state and can influence cell function.
- the culturing of prokaryotic cells, for example bacterial cells is also an established technique and has been used for many years for the production of valuable molecules.
- the culturing of mammalian cells has many applications and there are numerous in vitro assays and models where cell culture is used for experimentation and research; for example the use of cells in tissue engineering; the use of mammalian expression systems for the production of recombinant protein and the use of mammalian cells in the initial screening of drugs.
- tissue engineering is a science which has implications with respect to many areas of clinical and cosmetic surgery. More particularly, tissue engineering relates to the replacement and/or restoration and/or repair of damaged and/or diseased tissues to return the tissue and/or organ to a functional state. For example, tissue engineering is useful in the provision of skin grafts to repair wounds occurring as a consequence of: contusions, or burns, or failure of tissue to heal due to venous or diabetic ulcers. Tissue engineering requires in vitro culturing of replacement tissue followed by surgical application of the tissue to a wound to be repaired.
- Eukaryotic systems include the use of mammalian cells, e.g. Chinese Hamster Ovary cells; insect cells e.g. Spodoptera spp; or yeast e.g. Saccharomyces spp, Pichia spp.
- the large scale production of recombinant proteins requires a high standard of quality control since many of these proteins are used as pharmaceuticals, for example: growth hormone; leptin; erythropoietin; prolactin; TNF, interleukins; granulocyte colony stimulating factor (G-CSF); granulocyte macrophage colony stimulating factor (GM-CSF); ciliary neurotrophic factor (CNTF); cardiotrophin-1 (CT-1); leukemia inhibitory factor (LIF); oncostatin M (OSM); interferon, IFN ⁇ , IFN ⁇ .
- growth hormone for example: growth hormone; leptin; erythropoietin; prolactin; TNF, interleukins; granulocyte colony stimulating factor (G-CSF); granulocyte macrophage colony stimulating factor (GM-CSF); ciliary neurotrophic factor (CNTF); cardiotrophin-1 (CT-1); leukemia inhibitory factor (LIF); oncostatin M (OSM); interfer
- vaccines particularly subunit vaccines, (vaccines based on a defined antigen, for example gp120 of HIV)
- a defined antigen for example gp120 of HIV
- Post-translation processing includes the proteolytic processing of precursor proteins and the addition or removal of chemical groups (e.g. phosphorylation, prenylation, glucosylation, farnesylation).
- mammalian cells are used in initial drug screening to determine whether a lead therapeutic (e.g. a small molecule agonist or antagonist, a monoclonal antibody, peptide therapeutic, nucleic acid aptamer, small inhibitory RNA (siRNA)) has efficacy before animal trials are undertaken.
- a lead therapeutic e.g. a small molecule agonist or antagonist, a monoclonal antibody, peptide therapeutic, nucleic acid aptamer, small inhibitory RNA (siRNA)
- siRNA small inhibitory RNA
- Cell culture systems are known in the art and have been available to the skilled person for many years. Cell culture typically involves the growth of cells in monolayer culture under sterile conditions in closed cell culture vessels. More recently cell culture systems have been developed that provide means by which cells can be cultured in 3 dimensions to more closely resemble the situation found in vivo.
- WO2003/014334 discloses an in vitro cell culture method which provides a culture regime that allows prostate epithelial cells to form prostate-like-acini which closely resemble prostate acini found in vivo. These have utility in testing the efficacy of anti cancer agents with respect to controlling proliferation or metastasis of prostate cancer cells since transformed prostate epithelial cells also form acini in the cell culture system.
- cell culture substrates are described in WO00/34454 , which comprises microcellular polymeric materials which are described as polyHIPE polymers. These polymers form reticulate structures of pores that interconnect with one another to provide a substrate to which cells can attach and proliferate.
- the process for the formation of polyHIPEs allows pore volume to be accurately controlled with pore volume varying from 75% to 97%. Pore sizes can vary between 0.1 to 1000 micron and the diameter of the interconnecting members from a few microns to 100 microns.
- the polyHIPEs can be combined with additional components that facilitate cell proliferation and/or differentiation. PolyHIPEs are therefore versatile substrates on which cells can attach and proliferate in a cell culture system. Processes for the preparation of polyHIPEs are well known in the art and also disclosed in WO2004/005355 and WO2004/004880 .
- PolyHIPEs are commercially available and comprise for example oil phase monomers styrene, divinyl benzene and a surfactant, for example Span 80 sorbitan monooleate.
- the rigidity of the polymer formed during processing of the polyHIPE may be affected by the inclusion of a monomer such as 2-ethylhexyl acrylate.
- the process for the formation of polyHIPE from an emulsion is initiated by the addition of a catalyst such as ammonium persulphate.
- styrene concentration can vary from 15 %(w/w) to 78 % (w/w); surfactant concentration varies between 14 %(w/w) and 15 %(w/w) and the addition of the monomer 2-ethylhexyl acrylate varies between 60 %(w/w) and 62 %(w/w).
- the disclosures in these applications relate to the production of unitary cell supports to which cells attach and grow.
- the resultant polyHIPEs formed by these processes have pore volumes that vary from 75% to 97%.
- polyHIPE that has superior properties specifically designed for the routine culture of cells, typically mammalian cells, when compared to polyHIPEs formed by prior art processes.
- the polyHIPEs thus formed have a porosity of around 90% and are further processed into thin membranes or layers (for example, by microtome sectioning) to produce a cell culture substrate comprising a plurality of thin polyHIPE adapted to fit existing cell culture vessels.
- the polyHIPE is also modified by the inclusion of organic monomers and polymers to provide a cell culture substrate tailored to specific cell-types.
- the cell culture system herein disclosed can be applied to both eukaryotic cells and prokaryotic cells to provide the means to produce cell cultures that mirror more closely in vivo conditions to provide a more reliable cell culture system that has applications, for example in tissue engineering, recombinant protein production and drug screening.
- cell culture substrate comprising a plurality of sectioned microcellular polymeric material wherein said sections are 50-1000 microns and wherein the pore volume of the microcellular polymeric material is between 88% and 92%.
- Pore volume is defined as the fraction of the total volume of the material that is comprised of pores, and is determined by the droplet fraction of the parent emulsion.
- said pore volume is about 90%.
- membranes of microcellular polymeric material with a pore volume of about 90% are a surprisingly effective substrate for cell growth.
- cell adherence, proliferation and function are significantly affected by the structure of the polymeric material.
- the cells adhere better to 90% porosity materials and proliferate well and show enhanced function over cells grown on polymeric materials with different porosities (for example, 95% pore volume).
- the proliferation and function of cells grown on 90% polymeric materials is significantly improved compared to the growth of cells on conventional 2-dimensional tissue culture plastic.
- said substrate comprises a hydrophobic elastomer at a concentration of between 20 %(w/w) and 40% (w/w) of the total monomer content.
- said hydrophobic elastomer is provided at a concentration of between 25 %(w/w) and 35%(w/w).
- concentration is selected from the group consisting of 26%(w/w); 27%(w/w); 28%(w/w); 29%(w/w); 30%(w/w); 31%(w/w); 32%(w/w); 33%(w/w); or 34 %(w/w).
- said hydrophobic elastomer is provided at a concentration of 30% (w/w).
- said elastomer is selected from the group consisting of: 2-ethylhexyl acrylate; n-butyl acrylate and n-hexyl acrylate.
- said elastomer is 2-ethylhexyl acrylate.
- said 2-ethylhexyl acrylate is provided at between 28 %(w/w) and 32% (w/w); preferably 2-ethylhexyl acrylate is provided at about 30% (w/w).
- said cell culture substrate comprises polyvinyl.
- said polyvinyl is polystyrene; preferably a polystyrene comprising a styrene monomer and divinylbenzene.
- said cell culture substrate comprises a surfactant.
- said surfactant is provided at a concentration of 20-30% (w/w) of the monomer phase of the emulsion; preferably 24-26 %(w/w) and most preferably around 25%(w/w).
- said cell culture substrate comprises a plurality of sectioned microcellular polymeric material wherein said sections are 50-1000 microns thick; preferably said sections are approximately 500-750 microns thick. More preferably still said sections are 100-200 microns thick.
- said cell culture substrate comprises a plurality of sectioned microcellular polymeric material wherein said sections are 50-250 microns thick; preferably said sections are approximately 150 microns thick.
- said cell culture substrate comprises a plurality of sectioned microcellular polymeric material wherein said sections are 50-150 microns thick; preferably said sections are approximately 120 microns thick.
- said sectioned microcellular material is approximately 300 microns thick.
- said cell culture substrate comprises a further organic monomer.
- said organic monomer is selected from the group consisting of: N-butyl methacrylate, n-hexyl methacrylate, cyclohexyl acrylate, cyclohexyl methacrylate, phenyl acrylate, phenyl methacrylate, 3-vinylbenzyl chloride, 4-vinylbenzyl chloride, para-acetoxystyrene.
- said cell culture substrate comprises a further organic polymer.
- said organic polymer is selected from the group consisting of: Poly(n-butyl methacrylate), poly(n-hexyl methacrylate), poly(cyclohexyl acrylate), poly(cyclohexyl methacrylate), poly(phenyl acrylate), poly(phenyl methacrylate), poly(3-vinylbenzyl chloride), poly(4-vinylbenzyl chloride), poly(para-acetoxystyrene).
- said cell culture substrate comprises a surface that has been modified by the provision of a coating that facilitates the attachment, proliferation and/or differentiation of cells attached to the surface.
- said modification is the provision of a proteinaceous coating.
- said proteinaceous coating comprises at least one molecule selected from the group consisting of: laminin, collagen, for example cell supports like Matrigel, fibronectin, non-collagen based peptide matrices.
- said proteinaceous coating comprises a poly-amino acid coating.
- Poly-amino acids have properties that mimic proteins and in particular proteins to which cells can attach and grow.
- Poly-amino acids can be homopolymers or heteropolymers.
- Examples of poly amino acids useful in cell culture include poly L ornthine and poly L lysine. Proteinaceous coatings are well known in the art. For example see Culture of Animal Cells, Ian Freshney, Wiley-Liss 1994 .
- the surface of said cell culture substrate is physically modified.
- said substrate comprises a surface that is modified by gas plasma treatment.
- Gas plasma treatment of cell culture substrates is known in the art.
- the plasma treatment can be used to alter the physical properties of a cell culture surface.
- ammonia and oxygen have been used as gas plasmas to improve cell attachment and proliferation on cell culture products.
- the process involves the excitation of gaseous products at low pressures and ambient temperatures by radio-frequency energy.
- the plasmas contain free electrons and other metastable particles which upon collision with polymeric surfaces can modify the surface by breaking chemical bonds. This creates free radicals which also modify the polymer surface.
- a cell culture vessel comprising a cell culture substrate according to the invention.
- Cell culture vessel is defined as any means suitable to contain the above described cell culture substrate.
- an example of such a vessel is a petri dish; cell culture bottle or flask or multiwell culture dishes or well insert.
- Multiwell culture dishes are multiwell microtitre plates with formats such as 6, 12, 48, 96 and 384 wells which are typically used for compatibility with automated loading and robotic handling systems.
- high throughput screens use homogeneous mixtures of agents with an indicator compound that is either converted or modified resulting in the production of a signal. The signal is measured by suitable means (for example detection of fluorescence emission, optical density, or radioactivity) followed by integration of the signals from each well containing the cells, substrate/agent and indicator compound.
- said cell culture vessel comprising said cell culture substrate further comprises a cell and cell culture media.
- said cell is a eukaryotic cell; preferably said eukaryotic cell is selected from the group consisting of: a mammalian cell; a plant cell; a fungal cell; a slime mold.
- said mammalian cell is a primate cell; preferably said primate cell is a human cell.
- said mammalian cell is selected from the group consisting of: an epidermal keratinocyte; a fibroblast (e.g. dermal, corneal; intestinal mucosa, oral mucosa, bladder, urethral, prostate, liver) an epithelial cell (e.g. corneal, dermal, corneal; intestinal mucosa, oral mucosa, bladder, urethral, prostate, liver); a neuronal glial cell or neural cell; a hepatocyte or hepatocyte stellate cell; a mesenchymal cell; a muscle cell (cardiomyocyte, or myotube cell); a kidney cell; a blood cell (e.g. CD4+ lymphocyte, CD8+ lymphocyte; a pancreatic P cell; or an endothelial cell);
- a fibroblast e.g. dermal, corneal; intestinal mucosa, oral mucosa, bladder, urethral, prostate, liver
- an epithelial cell
- said cell is a cell line derived from tumour tissue.
- said mammalian cell is a stem cell except human embryonic stem cell.
- said stem cell is selected from the group consisting of: haemopoietic stem cell; neural stem cell; bone stem cell; muscle stem cell; mesenchymal stem cell; epithelial stem cell (derived from organs such as the skin, gastrointestinal mucosa, kidney, bladder, mammary glands, uterus, prostate and endocrine glands such as the pituitary); endodermal stem cell (derived from organs such as the liver, pancreas, lung and blood vessels); embryonic stem cell except human embryonic stem cell; embryonic germ cell except human embryonic ferm cell; embryonal carcinoma stem cell except human embryonal carcinoma stem cell.
- said embryonic stem cell/embryonic germ cell is a pluripotent cell and not a totipotent cell.
- said cell is a prokaryotic cell; preferably a bacterial cell.
- said cell or cell line is genetically modified.
- said cell culture vessel is a bioreactor; preferably said bioreactor is designed to scale-up the proliferation, differentiation and function of the said cell type.
- said cells are mammalian cells; preferably human cells.
- said cells are hepatocytes.
- said cells are prokaryotic cells; preferably bacterial cells.
- microorganisms are used in the cell culture method according to the invention, they are grown or cultured in the manner with which the skilled worker is familiar, depending on the host organism.
- a liquid medium comprising a carbon source, usually in the form of sugars, a nitrogen source, usually in the form of organic nitrogen sources such as yeast extract or salts such as ammonium sulfate, trace elements such as salts of iron, manganese and magnesium and, if appropriate, vitamins, at temperatures of between 0°C and 100°C, preferably between 10°C and 60°C, while gassing in oxygen.
- the pH of the liquid medium can either be kept constant, that is to say regulated during the culturing period, or not.
- the cultures can be grown batchwise, semi-batchwise or continuously.
- Nutrients can be provided at the beginning of the fermentation or fed in semi-continuously or continuously.
- the products produced can be isolated from the organisms as described above by processes known to the skilled worker, for example by extraction, distillation, crystallization, if appropriate precipitation with salt, and/or chromatography. To this end, the organisms can advantageously be disrupted beforehand.
- the pH value is advantageously kept between pH 4 and 12, preferably between pH 6 and 9, especially preferably between pH 7 and 8.
- these media which can be employed in accordance with the invention usually comprise one or more carbon sources, nitrogen sources, inorganic salts, vitamins and/or trace elements.
- Preferred carbon sources are sugars, such as mono-, di- or polysaccharides.
- Examples of carbon sources are glucose, fructose, mannose, galactose, ribose, sorbose, ribulose, lactose, maltose, sucrose, raffinose, starch or cellulose.
- Sugars can also be added to the media via complex compounds such as molasses or other by-products from sugar refining. The addition of mixtures of a variety of carbon sources may also be advantageous.
- oils and fats such as, for example, soya oil, sunflower oil, peanut oil and/or coconut fat, fatty acids such as, for example, palmitic acid, stearic acid and/or linoleic acid, alcohols and/or polyalcohols such as, for example, glycerol, methanol and/or ethanol, and/or organic acids such as, for example, acetic acid and/or lactic acid.
- Nitrogen sources are usually organic or inorganic nitrogen compounds or materials comprising these compounds.
- nitrogen sources comprise ammonia in liquid or gaseous form or ammonium salts such as ammonium sulfate, ammonium chloride, ammonium phosphate, ammonium carbonate or ammonium nitrate, nitrates, urea, amino acids or complex nitrogen sources such as comsteep liquor, soya meal, soya protein, yeast extract, meat extract and others.
- the nitrogen sources can be used individually or as a mixture.
- Inorganic salt compounds which may be present in the media comprise the chloride, phosphorus and sulfate salts of calcium, magnesium, sodium, cobalt, molybdenum, potassium, manganese, zinc, copper and iron.
- Inorganic sulfur-containing compounds such as, for example, sulfates, sulfites, dithionites, tetrathionates, thiosulfates, sulfides, or else organic sulfur compounds such as mercaptans and thiols may be used as sources of sulfur for the production of sulfur-containing fine chemicals, in particular of methionine.
- Phosphoric acid, potassium dihydrogenphosphate or dipotassium hydrogenphosphate or the corresponding sodium-containing salts may be used as sources of phosphorus.
- Chelating agents may be added to the medium in order to keep the metal ions in solution.
- Particularly suitable chelating agents comprise dihydroxyphenols such as catechol or protocatechuate and organic acids such as citric acid.
- a method to screen for an agent wherein said agent affects the proliferation, differentiation or function of a cell comprising the steps of:
- said cell is a hepatocyte.
- said screening method includes the steps of: collating the activity data in (iii) above; converting the collated data into a data analysable form; and optionally providing an output for the analysed data.
- Assay devices include standard multiwell microtitre plates with formats such as 6, 12, 48, 96 and 384 wells which are typically used for compatibility with automated loading and robotic handling systems.
- high throughput screens use homogeneous mixtures of agents with an indicator compound which is either converted or modified resulting in the production of a signal.
- the signal is measured by suitable means (for example detection of fluorescence emission, optical density, or radioactivity) followed by integration of the signals from each well containing the cells, agent and indicator compound.
- agent includes any small molecule, antibody, polypeptide, peptide, aptamer, double stranded or small inhibitory RNA. These can be an agonist or an antagonist.
- Small molecule antagonists include chemotherapeutic agents useful in the treatment of diseases such as cancer.
- Antibodies or immunoglobulins are a class of structurally related proteins consisting of two pairs of polypeptide chains, one pair of light (L) (low molecular weight) chain ( ⁇ or ⁇ ), and one pair of heavy (H) chains ( ⁇ , ⁇ , ⁇ , ⁇ and ⁇ ), all four linked together by disulphide bonds. Both H and L chains have regions that contribute to the binding of antigen and that are highly variable from one Ig molecule to another. In addition, H and L chains contain regions that are non-variable or constant. The L chains consist of two domains. The carboxy-terminal domain is essentially identical among L chains of a given type and is referred to as the "constant" (C) region.
- C constant
- variable region contains complementarity determining regions or CDR's which form an antigen binding pocket.
- the binding pockets comprise H and L variable regions which contribute to antigen recognition. It is possible to create single variable regions, so called single chain antibody variable region fragments (scFv's). If a hybridoma exists for a specific monoclonal antibody it is well within the knowledge of the skilled person to isolate scFv's from mRNA extracted from said hybridoma via RT PCR. Alternatively, phage display screening can be undertaken to identify clones expressing scFv's.
- domain antibodies are the smallest binding part of an antibody (approximately 13kDa). Examples of this technology is disclosed in US6, 248, 516 , US6,291, 158 , US6,127, 197 and EP0368684 .
- Aptamers are small, usually stabilised, nucleic acid molecules which comprise a binding domain for a target molecule.
- a screening method to identify aptamers is described in US 5,270,163 .
- Aptamers are typically oligonucleotides which may be single stranded oligodeoxynucleotides, oligoribonucleotides, or modified oligodeoxynucleotide or oligoribonucleotides.
- siRNA small inhibitory or interfering RNA
- the siRNA molecule comprises two complementary strands of RNA (a sense strand and an antisense strand) annealed to each other to form a double stranded RNA molecule.
- the siRNA molecule is typically derived from exons of the gene which is to be ablated. The mechanism of RNA interference is being elucidated. Many organisms respond to the presence of double stranded RNA by activating a cascade that leads to the formation of siRNA.
- siRNAs small fragments
- the siRNA acts as a guide for the RNase complex to cleave mRNA complementary to the antisense strand of the siRNA thereby resulting in destruction of the mRNA.
- An agent based on a siRNA would have value in determining the function of a specific gene in cell proliferation and/or differentiation.
- said cell is a hepatocyte.
- said method includes the additional steps of:
- tumour cells produce a number of tumour cell specific antigens, some of which are presented at the tumour cell surface. These are generally referred to as tumour rejection antigens and are derived from larger polypeptides referred to as tumour rejection antigen precursors.
- tumour rejection antigens are presented via HLA's to the immune system. The immune system recognises these molecules as foreign and naturally selects and destroys cells expressing these antigens.
- tumour rejection antigens have been developed based on dominant tumour rejection antigens to provide individuals with a preformed defence to the establishment of a tumour.
- the method according to the invention provides a means to identify tumour rejection antigens and precursors which will have utility with respect to the vaccine development to provoke the patients own immune system to deter the establishment of tumours.
- an in vitro method to analyse the development of cancerous cells from normal cells comprising
- said cells are hepatocytes.
- agents capable of transforming a normal cell into a transformed cell with many of the features of cancerous cells include, by example only, viruses, DNA intercalating agents, oncogenes and telomerase genes.
- cancer refers to cells having the capacity for autonomous growth, i.e., an abnormal state or condition characterized by rapidly proliferating cell growth.
- the term is meant to include all types of cancerous growths or oncogenic processes, metastatic tissues or malignantly transformed cells, tissues, or organs, irrespective of histopathologic type or stage of invasiveness.
- cancer includes malignancies of the various organ systems, such as those affecting, for example, lung, breast, thyroid, lymphoid, gastrointestinal, and genito-urinary tract, as well as adenocarcinomas which include malignancies such as most colon cancers, renal-cell carcinoma, prostate cancer and/or testicular tumours, non-small cell carcinoma of the lung, cancer of the small intestine and cancer of the esophagus.
- carcinoma is art recognized and refers to malignancies of epithelial or endocrine tissues including respiratory system carcinomas, gastrointestinal system carcinomas, genitourinary system carcinomas, testicular carcinomas, breast carcinomas, prostatic carcinomas, endocrine system carcinomas, and melanomas. Exemplary carcinomas include those forming from tissue of the cervix, lung, prostate, breast, head and neck, colon and ovary.
- carcinosarcomas e.g., which include malignant tumours composed of carcinomatous and sarcomatous tissues.
- An "adenocarcinoma” refers to a carcinoma derived from glandular tissue or in which the tumor cells form recognizable glandular structures.
- sarcoma is art recognized and refers to malignant tumours of mesenchymal derivation.
- a process for the formation of a high internal phase polymer having a pore volume of between 88% and 92% comprising the steps of:
- said hydrophobic elastomer is provided at a concentration of between 25 %(w/w) and 35%(w/w); preferably said hydrophobic elastomer is provided at a concentration of about 30% (w/w).
- said elastomer is selected from the group consisting of: 2-ethylhexyl acrylate; n-butyl acrylate and n-hexyl acrylate.
- the temperature of the preparation in ii) is heated to a temperature of between 50°C and 80°C.
- said preparation in ii) is heated to 50°C or 60°C or 80°C.
- said preparation in i) comprises a styrene monomer.
- said preparation in i) comprises divinyl benzene.
- said preparation in i) comprises a surfactant that is provided at a concentration of 24-26 %(w/w) and most preferably around 25%(w/w).
- the preparation in i) comprises 60%(w/w) styrene; 30% (w/w) 2-ethylhexyl acrylate; 10% (w/w) divinylbenzene and 25% surfactant.
- said high internal phase emulsion polymer in step iv) is sectioned; preferably said polymer is sectioned into a thin membrane or layer.
- said polymer is engineered into a thin membrane or layer of approximately 50-150 microns thick; preferably said membranes are approximately 120 microns thick.
- a substrate comprising a high internal phase emulsion polymer to determine the liver toxicity of an agent.
- said agent is a chemotherapeutic agent.
- said agent is a viral gene therapy vector.
- a method to test the liver toxicity of an agent comprising the steps of:
- said agent is a chemotherapeutic agent.
- said agent is a viral gene therapy vector.
- a method for the growth and differentiation of a keratinocyte and/or keratinocyte precursor stem cell comprising:
- said fibroblast feeder cells are dermal fibroblasts.
- said fibroblast feeder cells are selected from the group consisting of: corneal fibroblasts, intestinal mucosa fibroblasts, oral mucosa fibroblasts, urethral fibroblasts, or bladder fibroblasts.
- said keratinocytes are epidermal keratinocytes.
- said fibroblasts are human fibroblasts.
- said keratinocytes are human keratinocytes.
- said preparation further comprises collagen.
- collagen is type 1 collagen.
- said collagen is provided as a gel.
- said collagen is provided in a solution.
- At least said keratinocytes are displaced to contact air thereby inducing keratinocyte stratification.
- an apparatus for the culture of cells comprising a cell culture substrate according to any of claims 1-31, a cell culture vessel and an insert adapted to co-operate with said cell culture vessel and contain said cell culture substrate and said cells.
- said cell culture substrate comprises fibroblasts and keratinocytes.
- Divinylbenzene (Aldrich; 80 vol % divinylbenzene, the remainder being m- and p-ethylstyrene), 2-ethylhexyl acrylate (Aldrich; 99 %) and styrene (Aldrich; 99 %) were passed through a column of basic activated alumina (Aldrich; Brockmann 1) to remove any inhibitor (4-tert-butylcatechol for styrene and divinylbenzene and hydroquinone or monomethyl ether hydroquinone for 2-ethyhexyl acrylate).
- PolyHIPE foams were prepared using the polymerisation of a HIPE.
- the polymers were engineered into 120 micron thick membranes. This can be achieved using a microtome or vibrotome should thicker sections (up to 1mm) be required.
- Membranes of polymeric material were then sterilized using absolute ethanol, hydrated through a series of graded ethanol solutions and subsequently washed ( ⁇ 3) with sterile phosphate buffered saline (PBS) prior to use.
- PBS sterile phosphate buffered saline
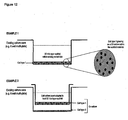
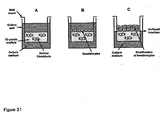
- Membranes can be mounted directly into the bottom of existing cell culture plastic-ware (e.g. 6-welled plate) or adhered to a cell culture well insert (see Figures 12 and 13 ).
- Mercury intrusion porosimetry analysis was performed using a Micromeritics AutoPore III 9420. Intrusion and extrusion mercury contact angles of 130° were used. Penetrometers with a stem volume of 1.836 mL and a bulb volume of 5 mL were used. The intrusion volume always comprised between 45 and 80 % of the stem volume. Intrusion pressures for the PolyHIPEs never exceeded 200 psi.
- the self diffusion coefficient of water was measured using a 500 MHz Varian Unity Inova 500 narrow bore spectrometer equipped with a Performa II gradient pulse amplifier and an actively shielded 5 mm indirect direction probe. Automated z gradient shimming based on deuterium spin echoes was used. The temperature used for all measurements was 25 +/- 0.1 °C. Water diffusion coefficients were measured using a pulse sequence incorporating pulsed-field gradients such as the bipolar pulse pair stimulated echo (BPPSTE) pulse sequence. Diffusion coefficients are obtained from BPPTSE spectra by monitoring signal attenuation as a function of the applied magnetic field gradient amplitude and fitting eq. (2) to the experimental results.
- BPPSTE bipolar pulse pair stimulated echo
- I I 0 exp - D ⁇ ⁇ G 2 ⁇ ⁇ - ⁇ / 2 - ⁇ / 3
- I the resonance intensity measured for a given gradient amplitude
- G the intensity in the absence of the gradient pulse
- ⁇ the gyromagnetic ratio
- ⁇ the duration of the bipolar gradient pulse pair
- ⁇ the diffusion delay time
- ⁇ is a short gradient recovery delay time during which relaxation and spin-spin coupling evolution are not significant.
- HepG2 The human hepatic carcinoma cell line, HepG2, was obtained from the American Type Culture Collection (ATCC). HepG2 cells were cultured at 37°C in 5% CO 2 in growth medium (Dulbecco's modified Eagle medium (D-MEM, Gibco/BRL) supplemented with 10% (v/v) foetal calf serum (FBS, Gibco/BRL), 100 ⁇ g.mL -1 penicillin and 10 ⁇ g.mL -1 streptomycin (Gibco/BRL)). Cells were passaged every 5-7 days. Confluent cultures of cells were washed with PBS, detached using trypsin/EDTA solution and cell number determined using a hemocytometer.
- D-MEM Dulbecco's modified Eagle medium
- FBS foetal calf serum
- Suspensions of HepG2 cells were then seeded at equal densities either directly into wells of a standard 6-welled plate (Nunc) or into modified well-inserts mounted with the polymer and located in wells of a 6-well plate. Cultures were maintained in growth medium which was changed every 3-4 days or as required.
- the number of viable cells was determined using a commercially available colorimetric assay (Promega) based on Mosmann's original method for measuring cell activity involving the conversion of a tetrazolium salt into a blue formazan product detectable by a spectrophotometer (570nm) [32].
- the assay was performed according to the manufacturer's instructions on HepG2 cells cultured on 2-D and 3-D substrates for various periods under alternative growth conditions.
- Cells were seeded on 2-D and 3-D surfaces in triplicate and left to settle and adhere for 24 hours. The medium was then changed and replaced with medium containing different concentrations of MTX (no MTX (vehicle alone, control), 8 ⁇ M, 31 ⁇ M, and 125 ⁇ M). Cells were subsequently incubated for 1, 3, 7 or 10 days, after which cultures were sampled and assayed for cell number/viability and levels of albumin and transglutaminase were determined.
- albumin is often used as an indicator of hepatocyte metabolic activity.
- Levels of albumin were determined using a commercially available kit (Bioassay systems) based on an established method that utilizes bromocresol green which forms a coloured complex specifically with albumin that is detectable at 620nm.
- Known quantities of human albumin were used to establish the standard curve.
- Specific levels of albumin secretion were normalised to total protein levels (as determined by a standard Bradford assay).
- Pelleted cells were subsequently rinsed in 0.1M phosphate buffer and immersed in 1% OsO 4 (aq.) solution for 1 hour, then dehydrated in 50%, 70%, 95% and 100% ethanol for 5min, for times for each respective ethanol change.
- the dehydrated cell pellets were then soaked in resin (Araldite CY212) for 60min at 37°C. When set, ultra thin sections of the resin embedded material were produced and subsequently imaged by TEM (Hitachi H7600).
- Tissue transglutaminase is a cross-linking enzyme which has recently been suggested to play a role in the formation of fibrotic lesions in experimental settings. The leakage of this enzyme is often used as a marker for in vitro toxicity testing and its presence indicates damage to cell membranes.
- tissue transglutaminase Several in vivo and in vitro experimental model systems show a direct relationship between the expression and activity of tissue transglutaminase, suppression of cell growth and programmed cell death [33-35]. The level of transglutaminase was analysed by means of a quantitative enzymatic assay (Sigma, UK) as previously described [36].
- the differential plot of intrusion versus interconnect diameter shows an increase in interconnect size as the aqueous phase temperature is increased.
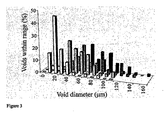
- the plot also shows that, as the aqueous phase temperature is increased, a material with a narrower distribution at higher interconnect diameters and a tail extending in the lower interconnect diameter range exists. This suggests that, for each emulsion, a limiting interconnect diameter exists. This is in contrast to the void size distribution, where a broader distribution is obtained as the temperature is increased.
- the ratio of the average interconnect ( ⁇ d>) and void diameters ( ⁇ D>) provides a measure of the degree of interconnection.
- the values for the materials prepared in this study are shown in Table 1. As the temperature is increased, the degree of interconnection ( ⁇ d>/ ⁇ D>) of the PolyHIPE material decreases. This suggests that, as the aqueous phase temperature is increased, the emulsion stability decreases.
- Emulsion partial destabilisation can be induced by the presence of organic additives in the aqueous phase.
- additives should be partially soluble in both the continuous and the internal phase of the emulsion, which can thus enhance diffusion of water molecules from droplet to droplet and promote Ostwald ripening.
- Lissant reported 3 that addition of cosolvents, such as acetone or methanol, can disrupt the interfacial film due to their solubility in both phases. These additives may dilute the interfacial layer and cause some of the surfactant to migrate into the bulk phase, therefore promoting coalescence of the emulsion droplets.
- THF tetrahydrofuran
- Each component was added to HIPEs in increasing quantities until phase separation occurred. It was found that the emulsion could accept much higher quantities of methanol than either THF or PEG, and this is particularly apparent if one considers the molar quantities of each (0.1 mol methanol, 0.02 mol THF and 0.005 mol PEG). This is due to the greater partitioning of methanol into the aqueous phase, at least in comparison to THF.
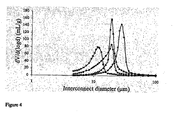
- the interconnect distribution curves are similar in nature for all additives used ( Figure 7 ). As the concentration of additive is increased, there is a tendency towards materials with a higher average interconnect diameter and a narrower size distribution. The exception to this trend is when THF is used as the additive. In this case a broad distribution is still obtained at high THF concentration. When PEG was used as the additive, the average interconnect diameter values increased steadily with PEG concentration in the aqueous phase (Table 1). This effect was not as pronounced for THF or methanol; for these additives, much higher concentrations were needed to produce a significant change in the interconnect diameter. In the case of THF, this was unexpected as it had the most significant effect on the average void diameter.
- the degree of interconnection decreases following initial addition of the organic component, which is brought about by the large increase in void diameter compared to interconnect diameter.
- the degree of interconnection increases steadily with the concentration in the aqueous phase.
- the degree of interconnection continued to decrease with increasing concentration. This is because THF has no significant effect on the interconnect size until the concentration reaches 1.5 %.
- the octanol/water partition coefficient value (log P ow ) for the additives indicates that THF partitions more into the oil phase than methanol, resulting in a less stable emulsion in the presence of THF.
- the surfactant concentration has little effect on the interconnect diameter when THF is used as the additive ( Figure 11a ).
- increasing the surfactant concentration from 20 to 25 % w/w results in a peak shift towards larger interconnect diameters ( Figure 11b ) in the presence of methanol.
- the surfactant concentration is increased from 25 % to 30 % w/w smaller interconnect diameters are obtained, although the ⁇ d>/ ⁇ D> ratios (Table 3) for both materials are similar since this is also accompanied by a decrease in ⁇ D>.
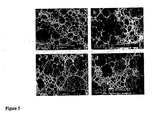
- polyHIPE materials The ability to control the structure of polyHIPE materials is critical to ensure the optimal growth of cultured cells in a 3-dimensional fashion.
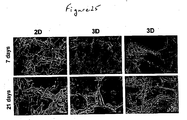
- the approach to using thin layers with large surface areas allows: (1) Good access of the cells into the structure of the material by either static or dynamic seeding; allows good access of oxygen and nutrients (in some cases from both sides of the membrane - see Figure 12 , example 1) and removal of waste materials and carbon dioxide therefore minimising the chance of necrosis occurring as found in polymer scaffolds of larger dimensions.
- hepatocytes possess polarity with two or three basal surfaces facing the sinusoid while adjacent cells form the bile canaliculi.
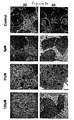
- Micrographs of cells grown in 3-D revealed adjacent hepatocytes often shared microvilli-lined channels lined with tight junctions. This observation suggests that cultured HepG2 cells may be polarized and capable of forming channels that resemble bile canaliculi [9]. These structures are known to be rich in microvilli and components of bile metabolised in the cells are normally secreted into the canaliculi.
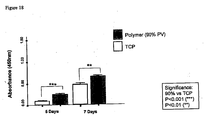
- Figure 27A illustrates the MTT absorbance values for hepatocytes grown on 2-D control surfaces and our 3-D scaffolds. Viable cells were successfully cultured on both substrates for up to 21 days. The assay revealed that cell viability was significantly enhanced when grown in 3-D. It should be noted that cells grown on scaffolds have a greater surface area on which to attach and grow, compared to planar surfaces, where space per cell is restricted. Where possible, this difference has been taken into account and values were normalised for in vitro assays.
- FIG. 27B shows the time courses of albumin secretion on 2-D tissue culture plastic and the 3-D scaffolds. Values have been normalised to account for any differences in cell number. It can be seen clearly that there is a significantly higher albumin concentration in cultures grown on 3-D surfaces compared to cells grown in 2-D for all the time points that were tested. Albumin levels in cultures grown on flat tissue culture plastic peaked at day 14 and then decreased rapidly at 21 days. This did not occur in cultures grown on 3-D scaffolds indicating that the 3-D environment is more conducive to cell function.
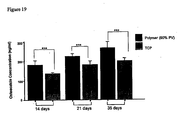
- HepG2 cells grown in 2-D and 3-D formats were treated with various concentrations of MTX to evaluate their tolerance to a well known cytotoxin. Following each treatment period, cultures were then studied for biochemical ( Figure 28 ) and morphological changes ( Figure 29 & 30 ).
- Figure 28A illustrates cell viability after 1 and 7 days treated with varying concentrations of MTX.
- Treatment of HepG2 monolayers with MTX resulted in a gradual increase of absorbance at 15 ⁇ M MTX after 24 hours but absorbance levels started to drop at 7 days (data not shown).
- the viability of cells grown on 2-D surfaces was visibly reduced especially at the higher levels of the cytotoxin.
- sensitivity to MTX was not evident in the lesser concentrations of MTX; only at 62 ⁇ M MTX was there a significant decrease in absorbance levels compared to control values.
- transglutaminase was performed as a test for HepG2 cell toxicity in response to increasing concentrations of MTX.
- levels of transglutaminase were found to be minimal and at similar levels.
- 2-D cultures secreted significantly higher levels of transglutaminase which increased in a dose dependent manner unlike cells grown in 3-D culture.
- HepG2 cells grown in 3-D culture and exposed to MTX were significantly more resistant to the effects of the cytotoxin.
- the ultra-structure of cells treated with lower concentrations of MTX possessed normal organelles in their cytoplasm (RER, ribosomes, mitochondria and lipid droplets).
- the nuclei displayed normal heterochromatin and nucleoli. These features were well preserved throughout most of the concentrations of MTX tested.
- the nuclear membrane had an irregular morphology and other sub-cellular features, such as mitochondria, which appeared to be slightly abnormal. It is likely therefore at higher concentrations of MTX, cells in 3-D cultures are starting to undergo changes similar to those experienced by cells cultured in 2-D as seen significantly lower concentrations of the cytotoxin.
- the growth of cells on styrene-based polymeric scaffolds adapted for use in existing cell culture plastic-ware provides the opportunity for 3-dimensional cell growth in vitro.
- Cell behaviour is influenced by the environment in which cells grow and cell growth in 3-dimensions is more realistic and more closely resembles the growth conditions cells normally experience in the body.
- the apparatus described herein provides an opportunity for researchers to routinely grow cells in 3-dimensions which will be invaluable for more accurate read-outs from cell models and assays.
- the apparatus is also inert, easy to use, can be sterilised, is cheap to manufacture and produce, it is robust and reproducible, has an indefinite shelf-life and is adaptable to many applications.
- polystyrene scaffold In a further application for the use of the polystyrene scaffold, we have developed an organotypic model of mammalian skin consisting of a stratified sheet of epidermal keratinocytes grown at the media/air interface on a layer of dermal fibroblasts in the presence or absence of a collagen gel or solution-coating within the scaffold.
- This system enables long term growth and maintenance of polarised epithelia that closely resemble native skin.
- the technology can be used to investigate the function of skin epithelial cells in a broad range of applications, including basic science, development of pharmaceuticals and assessment of compound toxicity.
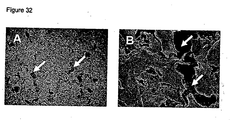
- an organotypic coculture is set up by seeding epidermal keratinocytes (e.g. HaCaT cells) onto the surface of the fibroblasts ( Figure 31b ).
- epidermal keratinocytes e.g. HaCaT cells
- the surface of the polystyrene scaffold is raised to the air-liquid interface. Air exposure induces stratification of the keratinocytes ( Figures 31c and 33 ).
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biomedical Technology (AREA)
- Zoology (AREA)
- Chemical & Material Sciences (AREA)
- Biotechnology (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Immunology (AREA)
- Sustainable Development (AREA)
- Gastroenterology & Hepatology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Apparatus Associated With Microorganisms And Enzymes (AREA)
- Manufacture Of Porous Articles, And Recovery And Treatment Of Waste Products (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Claims (18)
- Ein Zellkultursubstrat, das eine Pluralität eines zerlegten polymerisierten High Internal Phase-Emulsionspolymerisats enthält, wobei besagte Teile 50-1000 Mikrometer groß sind, und worin das Porenvolumen des polymerisierten High Internal Phase-Emulsionspolymerisats zwischen 88% und 92% beträgt.
- Ein Substrat gemäß Anspruch 1, wobei besagtes Substrat ein hydrophobes Elastomer mit einer Konzentration zwischen 20% (w/w) und 40% (w/w) enthält.
- Ein Substrat gemäß Anspruch 2, wobei besagtes Elastomer aus der Gruppe, bestehend aus 2-Ethylhexylacrylat, n-Butylacrylat und n-Hexylacrylat, gewählt wird.
- Ein Substrat gemäß jedwedem der Ansprüche 1-3, wobei besagtes Zellkultursubstrat eine oberflächenaktive Substanz enthält.
- Ein Substrat gemäß Anspruch 4, wobei besagte oberflächenaktive Substanz in einer Konzentration von 20-30% (w/w) oder zwischen 24-26% (w/w) oder ungefähr 25% (w/w) bereitgestellt wird.
- Ein Verfahren zur Bildung eines High Internal Phase-Polymers, welches ein Porenvolumen zwischen 88% und 92% aufweist, und die folgenden Schritte umfasst:i) Bilden eines Präparats, das eine High Internal Phase-Emulsion enthält, welches ein hydrophobes Elastomer in einer Konzentration von zwischen 20% (w/w) und 40% (w/w) sowie eine oberflächenaktive Substanz enthält, die in einer Konzentration von 20-30% (w/w) bereitgestellt wird;ii) Bilden eines Präparats, das einen Katalysator enthält;iii) Vereinen der Präparate von (i) und (ii);iv) Inkubieren des vereinigten Präparats, um die Bildung eines High Internal Phase-Emulsionspolymerisats zu ermöglichen; undv) Zerlegen besagten Polymers in Teile, die 50-1000 Mikrometer dick sind.
- Ein Verfahren für die Züchtung von Zellen, bestehend aus folgenden Schritten:i) Bereitstellen eines Zellkulturgefäßes, welches Folgendes enthält:a) Zellen,b) Ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5;c) Zellkulturmedium, welches ausreicht, um das Wachstum besagter Zellen zu unterstützen; undii) Bereitstellen von Zellkulturbedingungen, die die Proliferation und/oder Differenzierung besagter Zellen fördern.
- Ein Zellkulturgefäß, welches ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5 enthält.
- Ein Verfahren, um auf einen Wirkstoff hin zu untersuchen, worin besagter Wirkstoff die Proliferation, Differenzierung oder Arbeitsweise einer Zelle beeinflusst. Dieses umfasst folgende Schritte:i) Bereitstellen einer Zellkultur, die mindestens eine Zelle und ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5 umfasst;ii) Hinzufügen mindestens eines zu prüfenden Wirkstoffs; undiii) Überwachen der Aktivität des Wirkstoffs im Hinblick auf die Proliferation, Differenzierung oder Arbeitsweise besagter Zellen.
- Ein Verfahren für die Bestimmung von Genen, die mit der Zelldifferenzierung in Zusammenhang stehen, bestehend aus folgenden Schritten:i) Bereitstellen einer Zellkultur, die mindestens eine Zelle und ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5 enthält;ii) Extrahieren von Nucleinsäure aus Zellen, die in besagter Zellkultur enthalten sind;iii) Besagte extrahierte Nucleinsäure mit einem Nucleinsäure-Array in Verbindung bringen; undiv) Detektieren eines Signals, welches das Binden besagter Nucleinsäure an einen Bindungspartner an besagtem Nucleinsäure-Array anzeigt.
- Ein in vitro-Verfahren zur Analyse der Entwicklung karzinöser Zellen aus normalen Zellen, das Folgendes umfasst:i) Bilden eines Präparats, das ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5 einschließlich Zellen enthält;ii) Hinzufügen mindestens eines Wirkstoffs, der in der Lage ist, eine Zelltransformation herbeizuführen; undiii) Überwachen der Auswirkung bzw. Nichtauswirkung besagten Wirkstoffs auf die Transformation besagter Zellen.
- Der Einsatz eines Substrats, das ein High Internal Phase-Emulsionspolymerisat gemäß jedwedem der Ansprüche 1-5 enthält, an Kulturzellen.
- Der Einsatz eines Substrats, das ein High Internal Phase-Emulsionspolymerisat gemäß jedwedem der Ansprüche 1-5 enthält, um die Lebertoxizität eines Wirkstoffs zu bestimmen.
- Ein Verfahren zur Prüfung der Lebertoxizität eines Wirkstoffs, welches folgende Schritte umfasst:i) Bereitstellen einer Zellkultur, die mindestens eine Hepatozytzelle und ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5 enthält;ii) Hinzufügen mindestens eines zu prüfenden Wirkstoffs; undiii) Überwachen der Aktivität des Wirkstoffs im Hinblick auf die Proliferation, Differenzierung oder Arbeitsweise besagter Hepatozytzellen als ein Maßstab für die Toxizität des Wirkstoffs.
- Ein Verfahren für Wachstum und Differenzierung eines Keratinozyts und/oder einer Keratinozyten-Vorläuferstammzelle, welches folgendes umfasst:i) Bilden eines Präparats, das ein Zellkultursubstrat gemäß jedwedem des Ansprüche 1-5, Fibroblast-Fütterzellen und Zellkulturmedium enthält;ii) Züchten besagter Fütterzellen, um ein Zellkultursubstrat zu erbringen, welches mit besagten Fütterzellen reichlich belegt ist;iii) Besagtes belegtes Substrat mit Keratinozyten und/oder einer Keratinozyten-Vorläuferstammzellen in Verbindung bringen; undiv) Züchten des vereinten Zellpräparats unter Bedingungen, die für Wachstum und Differenzierung besagter Keratinozyten und/oder einer Keratinozyten-Vorläuferstammzellen förderlich sind.
- Ein Verfahren zur Prüfung eines Wirkstoffs, welches Folgendes umfasst:i) Bilden eines Präparats, das ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5 enthält, welches einen zu prüfenden Wirkstoff beinhaltet;ii) Überwachen der Wirkung besagten Wirkstoffs auf das Keratinozytenwachstum und/oder Differenzierung, wenn mit einem Kontrollpräparat verglichen wird, welches besagten Wirkstoff nicht enthält.
- Ein Gerät für das Züchten von Zellen, welches Folgendes enthält: ein Zellkultursubstrat gemäß jedwedem der Ansprüche 1-5, ein Zellkulturgefäß und einen Einsatz, der angepasst ist, um mit besagtem Zellkulturgefäß zusammenzuwirken und besagtes Zellkultursubstrat und besagte Zellen zu enthalten.
- Die Verwendung eines Zellkultursubstrats gemäß jedwedem der Ansprüche 1-5 zur Gewinnung von differenzierter Hautverbundstruktur.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0608403A GB0608403D0 (en) | 2006-04-28 | 2006-04-28 | Substrate for the routine growth of cultured cells in three dimensions |
| GB0610120A GB0610120D0 (en) | 2006-05-20 | 2006-05-20 | Substrate for the routine growth of cultured cells in three dimensions |
| GB0700592A GB0700592D0 (en) | 2007-01-12 | 2007-01-12 | Substrate for the routine growth of cultured cells in three D dimensions |
| PCT/GB2007/001464 WO2007125288A1 (en) | 2006-04-28 | 2007-04-24 | Substrate for the growth of cultured cells in three dimensions |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2018418A1 EP2018418A1 (de) | 2009-01-28 |
| EP2018418B1 true EP2018418B1 (de) | 2014-12-10 |
Family
ID=38290111
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP07732504.1A Not-in-force EP2018418B1 (de) | 2006-04-28 | 2007-04-24 | Substrat für das wachstum kultivierter zellen in drei dimensionen |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20100048411A1 (de) |
| EP (1) | EP2018418B1 (de) |
| JP (2) | JP2009535025A (de) |
| AU (1) | AU2007245453B2 (de) |
| BR (1) | BRPI0710777A2 (de) |
| CA (1) | CA2650488A1 (de) |
| IL (1) | IL194894A (de) |
| MX (1) | MX2008013885A (de) |
| WO (1) | WO2007125288A1 (de) |
Families Citing this family (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007125288A1 (en) * | 2006-04-28 | 2007-11-08 | Reinnervate Limited | Substrate for the growth of cultured cells in three dimensions |
| WO2009059115A1 (en) * | 2007-10-31 | 2009-05-07 | Trustees Of The University Of Pennsylvania | Culture based screening assay and methods of use thereof to identify agents which modulate tumor development, invasion and differentiation |
| CN103097517A (zh) | 2010-06-11 | 2013-05-08 | 塞拉帝思股份公司 | 用于提高多能干细胞分化成肝细胞的3维支架 |
| US9534206B2 (en) * | 2010-12-16 | 2017-01-03 | General Electric Company | Cell carrier, associated methods for making cell carrier and culturing cells using the same |
| US9453196B2 (en) | 2010-12-16 | 2016-09-27 | General Electric Company | Cell carrier, methods of making and use |
| US9453197B2 (en) | 2010-12-16 | 2016-09-27 | General Electric Company | Methods of making cell carrier |
| US9926523B2 (en) * | 2010-12-16 | 2018-03-27 | General Electric Company | Cell carriers and methods for culturing cells |
| US9518249B2 (en) * | 2010-12-16 | 2016-12-13 | General Electric Company | Cell carrier, associated methods for making cell carrier and culturing cells using the same |
| US8981025B2 (en) | 2011-02-10 | 2015-03-17 | Corning Incorporated | Polymerizable catonic peptide monomers and polymers |
| CN103502426B (zh) * | 2011-02-28 | 2018-03-13 | 哈佛大学校长及研究员协会 | 细胞培养系统 |
| WO2013050921A1 (en) | 2011-10-03 | 2013-04-11 | Piramal Enterprises Limited | Hollow polymer microspheres as three-dimensional cell culture matrix |
| JP2013215152A (ja) * | 2012-04-11 | 2013-10-24 | Vessel Inc | 3次元細胞培養プレートとその製造方法 |
| US20170363614A1 (en) * | 2014-12-22 | 2017-12-21 | Enumeral Biomedical Holdings, Inc. | Methods For Screening Therapeutic Compounds |
| KR102150108B1 (ko) * | 2016-07-05 | 2020-08-31 | 한국과학기술원 | 세포시트 제작방법 및 응용을 위한 고분자 박막 배양플레이트 제작방법 및 용도 |
| AU2017326179B2 (en) | 2016-09-13 | 2022-12-08 | President And Fellows Of Harvard College | Methods relating to intestinal organ-on-a-chip |
| US20190276852A1 (en) * | 2016-11-14 | 2019-09-12 | Paean Biotechnology Inc | Method for delivering exogenous mitochondria into cells |
| EP3431582A1 (de) * | 2017-07-18 | 2019-01-23 | Koninklijke Philips N.V. | Zellkulturmaterialen |
| WO2022034573A1 (en) * | 2020-08-09 | 2022-02-17 | Technion Research & Development Foundation Limited | Highly porous poly(lactic acid) monoliths |
| DE102021201935A1 (de) | 2021-01-08 | 2022-07-14 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung eingetragener Verein | Hochporöse Nanofaservliese als Trägerstruktur für stromales Gewebe |
| EP4274928B1 (de) | 2021-01-08 | 2025-01-08 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Hochporöse nanofaservliese als trägerstruktur für stromales gewebe |
| WO2024071109A1 (ja) * | 2022-09-30 | 2024-04-04 | インテグリカルチャー株式会社 | 細胞培養装置、細胞培養システム、培養肉、及び培養上清 |
| CN117625394B (zh) * | 2023-12-01 | 2024-12-06 | 东莞合安机电有限公司 | 一种荔枝菌菌种培养液、制备方法及培养方法 |
| WO2025244512A1 (ko) * | 2024-05-23 | 2025-11-27 | 주식회사 엘지화학 | 세포 배양용 마이크로 캐리어, 세포 배양용 마이크로 캐리어의 제조 방법 및 이를 이용한 세포 배양 조성물 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5266476A (en) * | 1985-06-18 | 1993-11-30 | Yeda Research & Development Co., Ltd. | Fibrous matrix for in vitro cell cultivation |
| IL75554A (en) * | 1985-06-18 | 1993-01-14 | Yeda Res & Dev | Matrix for cell cultivation in vitro |
| GB8729889D0 (en) * | 1987-12-22 | 1988-02-03 | Unilever Plc | Bio-catalysts support systems |
| AU8681098A (en) * | 1997-08-01 | 1999-03-08 | Massachusetts Institute Of Technology | Three-dimensional polymer matrices |
| US6281257B1 (en) * | 1998-04-27 | 2001-08-28 | The Regents Of The University Of Michigan | Porous composite materials |
| GB9826701D0 (en) * | 1998-12-05 | 1999-01-27 | Univ Newcastle | Microcellular polymers as cell growth media and novel polymers |
| TW591106B (en) * | 2000-10-06 | 2004-06-11 | Ind Tech Res Inst | Carrier for cell attachment or fixation and its process for preparation |
| WO2007125288A1 (en) * | 2006-04-28 | 2007-11-08 | Reinnervate Limited | Substrate for the growth of cultured cells in three dimensions |
-
2007
- 2007-04-24 WO PCT/GB2007/001464 patent/WO2007125288A1/en not_active Ceased
- 2007-04-24 BR BRPI0710777-3A patent/BRPI0710777A2/pt not_active IP Right Cessation
- 2007-04-24 MX MX2008013885A patent/MX2008013885A/es not_active Application Discontinuation
- 2007-04-24 AU AU2007245453A patent/AU2007245453B2/en not_active Ceased
- 2007-04-24 EP EP07732504.1A patent/EP2018418B1/de not_active Not-in-force
- 2007-04-24 JP JP2009507144A patent/JP2009535025A/ja active Pending
- 2007-04-24 US US12/298,742 patent/US20100048411A1/en not_active Abandoned
- 2007-04-24 CA CA002650488A patent/CA2650488A1/en not_active Abandoned
-
2008
- 2008-10-23 IL IL194894A patent/IL194894A/en not_active IP Right Cessation
-
2014
- 2014-02-04 JP JP2014019401A patent/JP2014097068A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| JP2009535025A (ja) | 2009-10-01 |
| US20100048411A1 (en) | 2010-02-25 |
| BRPI0710777A2 (pt) | 2012-01-10 |
| CA2650488A1 (en) | 2007-11-08 |
| JP2014097068A (ja) | 2014-05-29 |
| WO2007125288A1 (en) | 2007-11-08 |
| EP2018418A1 (de) | 2009-01-28 |
| MX2008013885A (es) | 2009-02-11 |
| AU2007245453B2 (en) | 2013-11-28 |
| IL194894A0 (en) | 2011-08-01 |
| IL194894A (en) | 2013-08-29 |
| AU2007245453A1 (en) | 2007-11-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2018418B1 (de) | Substrat für das wachstum kultivierter zellen in drei dimensionen | |
| Anada et al. | An oxygen-permeable spheroid culture system for the prevention of central hypoxia and necrosis of spheroids | |
| Lee et al. | Engineering liver tissue spheroids with inverted colloidal crystal scaffolds | |
| US11130935B2 (en) | Microfluid device and three-dimensional microculture method for cell | |
| CA2351156A1 (en) | A bioprocess for the generation of pluripotent cell derived cells and tissues | |
| US20200256850A1 (en) | Method for evaluating effect of cytokine on metabolic activity of cytochrome p450, and drug screening method | |
| CN114317439A (zh) | 一种培养肿瘤类器官的方法 | |
| CN105039245A (zh) | 一种利用3d打印技术促进人未成熟卵母细胞体外成熟的方法 | |
| CN101484574A (zh) | 供培养的细胞三维生长的底物 | |
| EP4190891A1 (de) | Verfahren zur herstellung einer zellmasse | |
| Iguchi et al. | Proliferation of normal mouse uterine luminal epithelial cells in serum-free collagen gel culture | |
| EP3000869A1 (de) | Zellkulturverfahren, träger für kulturen in partikelform sowie partikelumfassendes zellaggregat | |
| CN114457007A (zh) | 一种基于微孔板的均一的单类器官模型及其制备方法 | |
| JP2003501081A (ja) | 幹細胞の増殖工程 | |
| CN112481193A (zh) | 三维培养肠及肠癌组织类器官的标准化培养基及培养方法 | |
| KR102116741B1 (ko) | 설치류 유래의 지방 전구세포를 포함하는 3 차원 세포배양 구조체, 이를 이용한 3 차원 세포배양 방법, 및 이를 이용한 약물 스크리닝 방법 | |
| JP2003507035A (ja) | 三次元バイオリアクターにおいて、骨髄から機能的な破骨細胞をエクスビボで産生する方法 | |
| EP4229179A1 (de) | Kugelförmige selbstangeordnete peptidhydrogele mit zellen | |
| CN113667632A (zh) | 细胞培养基添加剂、细胞培养基及细胞体外扩增的方法 | |
| Källén et al. | 3D Culture in Functionalized FN‐Silk Networks Facilitate Proliferation, Differentiation and Phenotypic Stability of Mature Human Primary Cells and Stem Cells | |
| EP4397750A1 (de) | Verfahren zur herstellung von gefässrandzellen der innenohrstria, verfahren zur beurteilung eines chemischen wirkstoffs und zellkultur zur beurteilung eines chemischen wirkstoffs | |
| WO2018067476A1 (en) | Methods and kits for production of tissue equivalents from cryopreserved cells | |
| Habra et al. | Robust Formation of Optimal Single Spheroids towards Cost-Effective In-Vitro 3-Dimensional Tumor Models | |
| Wang et al. | Dictating the fate of female germline stem cells using stiffness-programmed DNA hydrogels | |
| JP2025509608A (ja) | 分離ウェルマイクロプレートを使用する細胞培養適用法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20081121 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA HR MK RS |
|
| 17Q | First examination report despatched |
Effective date: 20090710 |
|
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1129418 Country of ref document: HK |
|
| DAX | Request for extension of the european patent (deleted) | ||
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20130925 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: PRZYBORSKI, STEFAN ALEXANDER Inventor name: CAMERON, NEIL |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20140722 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE SI SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Ref country code: CH Ref legal event code: NV Representative=s name: MICHELI AND CIE SA, CH |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 700674 Country of ref document: AT Kind code of ref document: T Effective date: 20150115 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602007039615 Country of ref document: DE Effective date: 20150122 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 602007039615 Country of ref document: DE Representative=s name: PATENTANWALTSKANZLEI MEYER, DE |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PCOW Free format text: NEW ADDRESS: NETPARK INCUBATOR THOMAS WRIGHT WAY SEDGEFIELD, CO. DURHAM TS21 3FD (GB) |
|
| RAP2 | Party data changed (patent owner data changed or rights of a patent transferred) |
Owner name: REINNERVATE LIMITED |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R081 Ref document number: 602007039615 Country of ref document: DE Owner name: REINNERVATE LTD., SEDGEFIELD, GB Free format text: FORMER OWNER: REINNERVATE LTD., OLD ELVET, DURHAM, GB Effective date: 20150211 Ref country code: DE Ref legal event code: R082 Ref document number: 602007039615 Country of ref document: DE Representative=s name: PATENTANWALTSKANZLEI MEYER, DE Effective date: 20150211 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 700674 Country of ref document: AT Kind code of ref document: T Effective date: 20141210 Ref country code: NL Ref legal event code: VDEP Effective date: 20141210 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20141210 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150311 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20150306 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150410 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20150422 Year of fee payment: 9 Ref country code: CH Payment date: 20150505 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150410 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20150423 Year of fee payment: 9 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602007039615 Country of ref document: DE |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| 26N | No opposition filed |
Effective date: 20150911 |
|
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: WD Ref document number: 1129418 Country of ref document: HK |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: LU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150424 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150424 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602007039615 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20160424 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20161230 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160430 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160424 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20161101 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160502 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160430 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20070424 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20141210 |