EP1548142A1 - Tôle d'acier à résistance élevée laminée à froid, ayant une excellente adhesivité d'une couche de revêtement - Google Patents
Tôle d'acier à résistance élevée laminée à froid, ayant une excellente adhesivité d'une couche de revêtement Download PDFInfo
- Publication number
- EP1548142A1 EP1548142A1 EP04028368A EP04028368A EP1548142A1 EP 1548142 A1 EP1548142 A1 EP 1548142A1 EP 04028368 A EP04028368 A EP 04028368A EP 04028368 A EP04028368 A EP 04028368A EP 1548142 A1 EP1548142 A1 EP 1548142A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- steel sheet
- mass
- oxides
- coating film
- film adhesion
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C30/00—Coating with metallic material characterised only by the composition of the metallic material, i.e. not characterised by the coating process
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/005—Ferrite
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/008—Martensite
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0221—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the working steps
- C21D8/0226—Hot rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0221—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the working steps
- C21D8/0236—Cold rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0278—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips involving a particular surface treatment
Definitions
- the present invention relates to a high-strength cold-rolled steel sheet excellent in coating film adhesion, and more particularly, to a cold-rolled steel sheet which has a tensile strength no lower than 550 MPa and is suitable for use as a steel sheet for automobile parts on account of its excellent coating film adhesion.
- Si is less influential in reducing ductility and is effective in increasing strength while retaining ductility.
- Si content is poor in chemical treatability and hence in coating film adhesion. Consequently, it was necessary to reduce Si content in the case where chemical treatability is important.
- excess Si forms an Si-containing intergranular oxide on the surface of steel sheet, thereby causing cracks to occur and aggravating coating film adhesion.
- IF steel Interstitial Free steel.
- IF steel is limited in carbon content (no more than 0.005%) and has its texture controlled by a specific recrystallization temperature, so that it is improved in deep drawability.
- IF steel with a very low carbon content will not achieve the high strength intended by the present invention.
- the retained austenite-containing steel sheet mentioned above contains such alloying elements as C, Si, Mn, and A1 in large amounts so as to secure retained austenite. Therefore, it is poor in weldability.
- An example of steel having an Si/Mn ratio lower than 1 is mild steel nearly free of Si, which is known to have good chemical treatability.
- a certain amount of Si is necessary for steel to have both high strength and good ductility, and hence there is a limit of reducing the Si content to keep the Si/Mn ratio below 1.
- a steel sheet does not always exhibit good chemical treatability even though it has a Si/Mn ratio lower than 1, for a certain Si content and an adequately controlled Mn content.
- the present invention was completed in view of the foregoing. It is an object of the present invention to provide a cold-rolled steel sheet characterized by a tensile strength no lower than 500 MPa and excellent coating film adhesion and weldability.
- the present invention is directed to a high-strength cold-rolled steel sheet excellent in coating film adhesion, which is a DP (Dual Phase) steel sheet of ferrite-tempered martensite type containing no more than 1 mass% of C (excluding 0 mass%), 0.05 to 2 mass% of Si, and 1 to 5 mass% of Mn, having a tensile strength no lower than 550 MPa, satisfying the equation (1) below, and being characterized by its surface in which there exist Si-Mn complex oxides no larger than 5 ⁇ m in diameter of the equivalent circle as many as 10 or more per 100 ⁇ m 2 and the coverage of oxides composed mainly of Si on the surface of steel sheet is no more than 10% of surface area (requirement (I)).
- DP Direct Phase
- the equivalent circle means the circle of the same area as the Si-Mn complex oxide.
- This steel sheet will be referred to as "Steel sheet 1 of the present invention” hereinafter.
- [Si]/[Mn] ⁇ 0.4 where [Si] denotes an Si content (in mass%) and [Mn] denotes an Mn content (in mass%).
- oxides composed mainly of Si means those oxides in which Si (as one of the constituents excluding oxygen) accounts for no less than 70% in atomic ratio. Such oxides are considered to be amorphous according to the result of analysis.
- the ratio of the surface area of steel sheet which is covered by the oxides composed mainly of Si was obtained by observation under a TEM (Transmission Electron Microscope), quantitative analysis and mapping of Si, O, Mn, and Fe by EDX (Energy Dispersive X-ray), and image analysis of these data.
- Observation under a TEM was accomplished by using an extraction replica, which is explained in Examples given later.
- Observation under a TEM for an extraction replica may be replaced by surface mapping for Si, O, Mn, and Fe by AES (Auger Electron Spectroscopy) at a magnification of 2000 to 5000, and the resulting data may be used for image analysis.
- the present invention is directed also to a high-strength cold-rolled steel sheet excellent in coating film adhesion, which is a DP (Dual Phase) steel sheet of ferrite-tempered martensite type containing no more than 1 mass% of C (excluding 0 mass%), no more than 2 mass% of Si (excluding 0 mass%), and 1 to 5 mass% of Mn, having a tensile strength no lower than 550 MPa, and being characterized by its surface whose cross section does not show cracks with a width no larger than 3 ⁇ m and a depth no smaller than 5 ⁇ m in arbitrary ten fields of observation under an SEM (Scanning Electron Microscope) with a magnification of 2000 (requirement (II)).
- This steel sheet will be referred to as "Steel sheet 2 of the present invention” hereinafter.
- Fig. 1 which is a schematic sectional view of the steel sheet. They are found by observing the vicinity of the surface of the steel sheet under an SEM with a magnification of 2000 (Model S-4500 of Hitachi Ltd.).
- the present invention is directed also to a high-strength cold-rolled steel sheet excellent in coating film adhesion, which is a DP (Dual Phase) steel sheet of ferrite-tempered martensite type containing no more than 1 mass% of C (excluding 0 mass%), 0.05 to 2 mass% of Si, and 1 to 5 mass% of Mn, having a tensile strength no lower than 550 MPa, satisfying the equation (1) above, and meeting the above-mentioned requirements (I) and (II). (This steel sheet will be referred to as "Steel sheet 3 of the present invention" hereinafter.)
- the steel sheets of the present invention should preferably have a composition specified by the equations (2) and (3) below as an additional requirement, so that they exhibit good weldability.
- [C], [Si], [Mn], [P], and [S] denote the content (in mass%) of these elements.
- the steel sheet according to the present invention has a high strength in excess of 550 MPa, exhibits good chemical treatability, and/or good coating film adhesion owing to controlled fine cracks, and provides good weldability. It is suitable for automotive parts. It can be produced without cladding or expensive elements.
- Fig. 1 is a schematic diagram showing cracks in the cross section of the steel sheet.
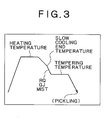
- Fig. 2 is a diagram illustrating (part of) one manufacturing process in Examples.
- Fig. 3 is a diagram illustrating (part of) another manufacturing process in Examples.
- Fig. 4 is an electron micrograph (TEM) of sample in experiment No. 1 in Examples. (Extraction replica, ⁇ 15000)
- Fig. 5 is an electron micrograph (TEM) of sample in experiment No. 29 in Examples. (Extraction replica, ⁇ 15000)
- Fig. 6 is an electron micrograph (TEM) of sample in experiment No. 34 in Examples. (Extraction replica, ⁇ 15000)
- Fig. 7 is an electron micrograph (SEM) showing the cross section near the surface of the steel sheet in experiment No. 1 in Examples.
- Fig. 8 is an electron micrograph (SEM) showing the cross section near the surface of the steel sheet in experiment No. 29 in Examples.
- Fig. 9 is an electron micrograph (SEM) showing the cross section near the surface of the steel sheet in experiment No. 34 in Examples.
- Fig. 10 is an electron micrograph (SEM) showing the surface of the steel sheet (after chemical treatment) in experiment No. 1 in Examples.
- Fig. 11 is an electron micrograph (SEM) showing the surface of the steel sheet (after chemical treatment) in experiment No. 29 in Examples.
- Fig. 12 is an electron micrograph (SEM) showing the surface of the steel sheet (after chemical treatment) in experiment No. 34 in Examples.
- the present inventors have carried out a series of researches to obtain a high-strength steel sheet excellent in coating film adhesion and proposed a technique for improving the chemical treatability of a steel sheet containing Si in a comparatively large amount.
- This technique is intended to improve chemical treatability by finely dispersing amorphous Si oxides detrimental to chemical treatability while controlling the annealing atmosphere.
- major oxides that occur when the Si content is relatively low Si content: 0.05 to 2% as defined in the present invention
- Si-Mn complex oxides rather than amorphous Si oxides. It is considered that these complex oxides are also detrimental to coating film adhesion.
- the present inventors searched for the possibility of positively using Si-Mn complex oxides for improvement of chemical treatability.
- the electrochemical inhomogeneous field mentioned above should preferably be of the same size.
- the present invention specifies that there should exist Si-Mn complex oxides no larger than 5 ⁇ m in diameter of the equivalent circle as many as 10 or more per 100 ⁇ m 2 (or 1 per 10 ⁇ m 2 or more on average), with the average distance between particles of complex oxides being several micrometers. This condition is necessary for easy formation of the electrochemical inhomogeneous field of the above-specified size.
- the number of particles of Si-Mn complex oxides should preferably be 50 or more per 100 ⁇ m 2 , more preferably 100 per or more 100 ⁇ m 2 , and most desirably 150 or more per 100 ⁇ m 2 , because the electrochemical inhomogeneous field does not necessarily occur in every particle of the Si-Mn complex oxides which are present.
- An example of the Si-Mn complex oxides is Mn 2 SiO 4 . It is considered that about 50 nm is the maximum observable size of Si-Mn complex oxides.
- the Si-Mn complex oxides functioning as nucleating sites for zinc phosphate crystals will not contribute to good chemical treatability if there exist other substances detrimental to chemical treatment. Hence, the resulting steel sheet will be poor in coating film adhesion.
- oxides composed mainly of Si are present on the surface of steel sheet, zinc phosphate crystals do not form on them, which leads to considerably poor chemical treatability. Consequently, the present invention requires that the coverage of oxides composed mainly of Si on the surface of steel sheet should be no more than 10% of surface area.
- the present inventors had previously proposed a technique of improving chemical treatability by finely dispersing oxides composed mainly of Si, as mentioned above.
- the presence of oxides should be minimized in the present invention which is intended to utilize the action of Si-Mn complex oxides as mentioned above. Therefore, the coverage of oxides composed mainly of Si on the surface of steel sheet should preferably be no more than 5% of surface area, most desirably 0% of surface area.
- Sharp cracks present on the surface of the steel sheet prevent zinc phosphate crystals from sticking to them at the time of chemical treatment. As the result, corrosion readily proceeds there, aggravating coating film adhesion. For this plausible reason, it is important to minimize the occurrence of sharp cracks in order to improve coating film adhesion.
- the present inventors had previously proposed a technique of improving coating film adhesion by restricting to 10 ⁇ m or less the depth of linear oxides (narrower than 30 nm) composed of Si and oxygen. This technique is based on the assumption that continuous annealing will not be followed by pickling. However, in common practice, continuous annealing is followed by pickling, and pickling removes linear oxides, thereby causing cracks to occur.
- the present inventors conceived that it would be possible to improve coating film adhesion more by controlling cracks than by regulating the depth of linear oxides (as in the technology they had previously proposed) and they investigated the shape of cracks to be controlled. As the result, it was found that zinc phosphate crystals hardly stick to cracks having a width approximately equal to or smaller than their particle diameter. This holds true particularly for cracks deeper than 5 ⁇ m. Thus, according to the present invention, cracks to be controlled are limited to those which are narrower than 3 ⁇ m and deeper than 5 ⁇ m.
- the steel sheet according to the present invention is required to have the following chemical composition so that it has controlled cracks for efficient deposition of the above-mentioned oxides and it exhibits the characteristic properties of high-strength steel sheet.
- oxides composed mainly of Si adversely affect chemical treatability, it is more desirable to suppress them as much as possible rather than finely dispersing them.
- the object of suppressing such oxides can be achieved if the [Si]/[Mn] ratio in the chemical composition is no larger than 0.4, preferably no larger than 0.3.
- Carbon is essential for strength.
- the minimum carbon content is 0.05 mass%. An excess carbon content aggravates weldability. Therefore, the carbon content should be no larger than 1 mass%, preferably no larger than 0.23 mass%, and more preferably no larger than 0.15 mass%.
- Si 0.05 to 2 mass% (for steel sheets 1 and 3)
- Si increases strength without decreasing ductility, it may be contained in the steel sheet.
- a certain amount of Si is necessary for the Si-Mn complex oxides with a diameter of the equivalent circle no larger than 5 ⁇ m to form as much as specified by the requirement (I) mentioned above.
- a minimum amount of Si for this purpose is 0.05 mass%.
- An adequate amount should be no less than 0.15 mass%, preferably no less than 0.3 mass%, and more preferably no less than 0.5 mass%. Si in an excess amount brings about solid solution hardening more than necessary, which leads to an increased rolling load. Therefore, the content of Si should be no larger than 2 mass%, preferably no larger than 1.5 mass%.
- Mn is also essential for strength; however, excess Mn is detrimental to ductility.
- An adequate content of Mn should be no less than 1 mass%, preferably no less than 2 mass%, and no more than 5 mass%, preferably no more than 3.5 mass%.
- the steel sheet according to the present invention should contain the above-mentioned elements, with the remainder being substantially iron. It may contain inevitable impurities, such as A1 no more than 1 mass%, N no more than 0.01 mass%, and O no more than 0.01 mass%, originating from raw materials or incorporated depending on production conditions. It may be positively incorporated with additional elements, such as Cr, Mo, Ni, Ti, Nb, V, P, and B, in an amount not harmful to the effect of the present invention.
- the amount of these additional elements to strengthen the steel sheet is specified as follows. Cr : 0.1 to 1 mass% Mo : 0.1 to 1 mass% Ni : 0.1 to 1 mass% Ti : 0.005 to 0.1 mass% Nb : 0.005 to 0.1 mass% V : 0.0005 to 0.01 mass% P : 0.005 t0 0.1 mass% B : 0.0003 to 0.01 mass%
- Cr 0.1 to 1 mass%
- Mo 0.1 to 1 mass%
- Ni 0.1 to 1 mass%
- Ti 0.005 to 0.1 mass%
- Nb 0.005 to 0.1 mass%
- V 0.0005 to 0.01 mass%
- P 0.005 t0 0.1 mass%
- B 0.0003 to 0.01 mass%
- the present invention covers a steel sheet having a strength no lower than 550 MPa (preferably no lower than 750 MPa, more preferably no lower than 900 MPa).
- the steel sheet should contain C, Si, and Mn (and optionally P) in an adequate amount as specified below according to strength and weldability desired.
- the present invention covers a DP (Dual Phase) steel sheet of ferrite-tempered martensite type.
- the steel may be composed solely of ferrite and tempered martensite, or it may additionally contain pearlite, bainite, and retained austenite in an amount not harmful to the effect of the present invention. They inevitably remain in the manufacturing process, but they should be as little as possible.
- the shape of the oxides that separate out on the surface thereof should be controlled according to the requirement (I) mentioned above.
- This object is achieved not only by the controlled steel composition as mentioned above but also by pickling (that follows hot rolling) with hydrochloric acid (1 to 18 mass%) at 70 to 90°C for about 40 seconds or more (preferably 60 seconds or more) and continuous annealing in an atmosphere with a dew point no higher than -40°C, preferably no higher than -45°C.
- the total dipping time should be 40 seconds at the minimum.
- the hot rolled steel sheet need undergo pickling.
- the winding temperature for hot rolling should be no higher than 500°C, preferably no higher than 480°C;
- the hot rolled steel sheet should be dipped in hydrochloric acid (1 to 18 mass%) at 70 to 90°C for 40 seconds or more, preferably 60 seconds or more;
- Continuous annealing should be performed in an atmosphere with a dew point no higher than -40°C, preferably no higher than -45°C;
- the hardening start temperature at the time of annealing (which may be referred to as "slow cooling end temperature”) should be no higher than 550°C, preferably from 400 to 450°C.
- the production conditions should be such that generation of grain boundary oxides which can become start points for the cracks is restrained.
- the present invention does not specify other manufacturing conditions than mentioned above.
- the steel sheet may be produced in the usual way by melting, casting, and hot rolling.
- the manufacturing process in Examples that follow involves pickling that follows continuous annealing, pickling is not mandatory in the present invention.
- a steel with the chemical composition shown in Table 1 was prepared by melting, and the resulting steel was cast into a slab, which underwent hot rolling, followed by pickling. Winding and pickling were performed under the conditions shown in Tables 2 and 3. Pickling involved an aqueous solution of hydrochloric acid (1 to 18 mass%) at 70 to 90°C. Pickling was followed by cold rolling, which gave a 1.4 mm thick steel sheet.
- the steel sheet underwent continuous annealing by either of the processes shown in Figs. 2 and 3.
- the process shown in Fig. 2 involves cooling with water quenching (WQ) that follows soaking and slow cooling.
- the process shown in Fig. 3 involves cooling with mist, gas blowing (GJ), or water-cooled roll quenching (RQ).
- the heating temperature, slow cooling end temperature, and tempering temperature shown in Tables 2 and 3 correspond to those shown in Figs. 2 and 3.
- the dew point is that of the atmosphere in the continuous annealing furnace. After cooling, the steel sheet underwent tempering. In the process shown Fig. 2, pickling was carried out before and/or after tempering.
- the thus obtained steel sheet was examined for mechanical properties and coating film adhesion. All of the steel sheet samples were found to be composed mainly of ferrite and tempered martensite.
- an extraction replica is prepared from the surface of the steel sheet. Then, it is observed under a TEM (Model H-800 of Hitachi, Ltd.) with a magnification of 15000. An average number of particles (per 100 ⁇ m 2 ) is counted in arbitrary 20 fields of observation.
- the ratio of the surface area of steel sheet which is covered by the oxides composed mainly of Si was obtained by observation of a sample under a TEM and ensuing image analysis.
- the sample was prepared by the extraction replica method consisting of four steps (a) to (d) as explained in the following.
- the treated sample was photographed in ten fields of observation (each measuring 13 by 11 cm) through a TEM with a magnification of 15000.
- the resulting electron micrograph was examined to measure the area covered by oxides composed mainly of Si (or oxides in which Si as the constituents excluding oxygen accounts for no less than 70% in atomic ratio). In this way there was obtained the coverage of oxides composed mainly of Si.
- Samples in Nos. 32, 38, and 40 meet the requirement for the steel sheet 1 of the present invention and hence they are excellent in chemical treatability and coating film adhesion.
- the results suggest that it is necessary to control the winding temperature and slow cooling end temperature for the steel sheet to have good coating film adhesion, with cracks properly controlled.
- Samples in Nos. 34 to 36 meet the requirement for the steel sheet 2 of the present invention and hence they are free of cracks and excellent in coating film adhesion.
- the results suggest that it is necessary to control the composition and the shape of the oxides that separate out on the surface of the steel sheet for the steel sheet to have good chemical treatability and coating film adhesion.
- samples in Nos. 29, 30, 31, 33, 39, and 41 do not meet the requirement for the steel sheets (1 to 3) of the present invention and hence they are poor in coating film adhesion.
- samples in Nos. 29 to 31 do not meet the requirement for [Si]/[Mn] ratio and hence they do not give oxides having the shape specified in the present invention.
- they have many cracks (because they are not produced under the desired conditions) and they are poor in coating film adhesion.
- Samples in Nos. 33, 39, and 41 are not produced under the desirable conditions and hence they do not have oxides with the shape specified in the present invention. They have cracks and are poor in coating film adhesion.
- Sample in No. 37 meets the requirements and hence is excellent in coating film adhesion; but it cannot be formed satisfactorily on account of its poor ductility.
- Samples in Nos. 1 to 27 meet the requirement for the steel sheet 3 of the present invention (or the requirements for the steel sheets (1 and 2) of the present invention) and they also satisfy the equations (2) and (3) and hence they are excellent in chemical treatability, coating film adhesion (free of cracks), and weldability.
- Sample in No. 28 meets the requirements for the steel sheet 3 of the present invention; however, the results suggest that the composition should satisfy the equations (2) and (3) for the steel sheet to exhibit good weldability.
- Figs. 4 indicates that sample in No. 1 has fine Si-Mn complex oxides but does not have oxides composed mainly of Si.
- Fig. 5 indicates that sample in No. 29 is covered with oxides composed mainly of Si.
- Fig. 6 indicates that sample in No. 34 does not have fine Si-Mn complex oxides although it has particulate matter (which is rust).
- Figs. 7 to 9 indicate that sample in No. 1 is free from cracks.
- Fig. 8 indicates that sample in No. 29 has cracks, 5 ⁇ m deep.
- Fig. 9 indicates that sample in No. 34 is free of cracks and hence excellent in coating film adhesion.
- Figs. 10 indicates that sample in No. 1 has fine zinc phosphate crystals free of interstice.
- Fig. 11 indicates that sample in No. 29 has small zinc phosphate crystals with large interstices.
- Fig. 12 indicates that sample in No. 34 has large zinc phosphate crystals with large interstices.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Heat Treatment Of Sheet Steel (AREA)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE602004015922T DE602004015922T3 (de) | 2003-12-25 | 2004-11-30 | Hochfestes, Kaltgewalztes Stahlblech mit Ausgezeichneter Adhesion von Beschichtung |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003429151 | 2003-12-25 | ||
| JP2003429151A JP3934604B2 (ja) | 2003-12-25 | 2003-12-25 | 塗膜密着性に優れた高強度冷延鋼板 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1548142A1 true EP1548142A1 (fr) | 2005-06-29 |
| EP1548142B1 EP1548142B1 (fr) | 2008-08-20 |
| EP1548142B2 EP1548142B2 (fr) | 2013-07-03 |
Family
ID=34545001
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP04028368.1A Expired - Fee Related EP1548142B2 (fr) | 2003-12-25 | 2004-11-30 | Tôle d'acier à résistance élevée laminée à froid, ayant une excellente adhesivité d'une couche de revêtement |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20050139293A1 (fr) |
| EP (1) | EP1548142B2 (fr) |
| JP (1) | JP3934604B2 (fr) |
| AT (1) | ATE405685T1 (fr) |
| DE (1) | DE602004015922T3 (fr) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2433746A (en) * | 2005-12-27 | 2007-07-04 | Kobe Steel Ltd | Steel sheet with excellent weldability |
| EP1865085A1 (fr) * | 2005-03-31 | 2007-12-12 | Kabushiki Kaisha Kobe Seiko Sho | Tole d acier laminee a froid de resistance elevee, excellente en termes d adherence des revetements, d aptitude au faconnage et de resistance a la fragilisation par l'hydrogene, et composant en acier pour automobiles |
| WO2008102009A1 (fr) * | 2007-02-23 | 2008-08-28 | Corus Staal Bv | Feuillard d'acier à résistance élevée, laminé à froid et trempé en continu, et procédé de fabrication dudit feuillard |
| WO2009021897A1 (fr) * | 2007-08-15 | 2009-02-19 | Thyssenkrupp Steel Ag | Acier biphasé, produit plat constitué d'un tel acier biphasé et procédé de fabrication d'un produit plat |
| EP2123786A1 (fr) * | 2008-05-21 | 2009-11-25 | ArcelorMittal France | Procédé de fabrication de tôles d'aciers dual phase laminées à froid à trés haute résistance et tôles ainsi produites |
| CN102317490A (zh) * | 2009-03-30 | 2012-01-11 | 新日本制铁株式会社 | 渗碳钢部件 |
| WO2015014333A3 (fr) * | 2013-07-30 | 2015-11-05 | Salzgitter Flachstahl Gmbh | Acier multiphase à haute résistance, micro-allié et contenant du silicium, présentant une résistance minimale à la traction de 750 mpa et des propriétés améliorées et procédé de fabrication d'une bande à partir de cet acier |
| EP2785889A4 (fr) * | 2011-11-28 | 2016-03-02 | Arcelormittal Investigación Y Desarrollo Sl | Aciers double-phase comportant une teneur élevée en silicium dotés d'une ductilité améliorée |
| EP2623630A4 (fr) * | 2010-09-30 | 2016-11-23 | Jfe Steel Corp | Tôle d'acier à haute résistance et procédé de production associé |
| US9598743B2 (en) | 2010-09-29 | 2017-03-21 | Jfe Steel Corporation | High strength steel sheet and method for manufacturing the same |
| EP3418419A4 (fr) * | 2016-03-31 | 2019-01-23 | JFE Steel Corporation | Tôle d'acier mince, tôle d'acier plaquée, procédé de production de tôle d'acier laminée à chaud, procédé de production de tôle d'acier laminée à froid entièrement dure, procédé de production de tôle d'acier mince et procédé de production de tôle d'acier plaquée |
| EP2829626B1 (fr) * | 2012-03-19 | 2019-06-26 | JFE Steel Corporation | Procédé de fabrication d'une feuille d'acier galvanisé par immersion à chaud à résistance élevée et feuille d'acier galvanisé par immersion à chaud à résistance élevée |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2450066B (en) * | 2006-03-31 | 2011-03-30 | Kobe Steel Ltd | High-strength cold rolled steel sheet excellent in chemical conversion treatment property |
| KR100851189B1 (ko) * | 2006-11-02 | 2008-08-08 | 주식회사 포스코 | 저온인성이 우수한 초고강도 라인파이프용 강판 및 그제조방법 |
| ES2367713T3 (es) * | 2007-08-15 | 2011-11-07 | Thyssenkrupp Steel Europe Ag | Acero de fase dual, producto plano de un acero de fase dual tal y procedimiento para la fabricación de un producto plano. |
| CN102066597B (zh) * | 2008-06-13 | 2013-03-06 | 株式会社神户制钢所 | 异种材料接合用钢材、异种材料接合体及异种材料接合方法 |
| JP5399681B2 (ja) | 2008-10-08 | 2014-01-29 | Jfeスチール株式会社 | 化成処理性に優れた高加工性高強度鋼管およびその製造方法 |
| JP5810499B2 (ja) * | 2010-09-29 | 2015-11-11 | Jfeスチール株式会社 | 高強度鋼板の製造方法 |
| JP5834388B2 (ja) * | 2010-09-29 | 2015-12-24 | Jfeスチール株式会社 | 高強度鋼板の製造方法 |
| JP5637530B2 (ja) * | 2010-10-26 | 2014-12-10 | Jfeスチール株式会社 | 高延性で、化成処理性に優れる780MPa以上の引張強度を有する超高強度冷延鋼板 |
| JP5853683B2 (ja) | 2011-01-25 | 2016-02-09 | Jfeスチール株式会社 | 化成処理性および塗装後耐食性に優れた冷延鋼板の製造方法 |
| JP5637230B2 (ja) | 2013-02-28 | 2014-12-10 | Jfeスチール株式会社 | 高強度冷延鋼板の製造方法 |
| CN105121687A (zh) * | 2013-04-18 | 2015-12-02 | 新日铁住金株式会社 | 表面硬化用钢材和表面硬化钢部件 |
| WO2016147549A1 (fr) | 2015-03-18 | 2016-09-22 | Jfeスチール株式会社 | Tôle d'acier laminée à froid à haute résistance et son procédé de fabrication |
| BR112017014368A2 (pt) | 2015-03-23 | 2018-01-02 | Nippon Steel & Sumitomo Metal Corporation | lâmina de aço laminada a quente e método de fabricação da mesma, e método de fabricação de lâmina de aço laminada a frio |
| KR102519960B1 (ko) * | 2018-11-09 | 2023-04-10 | 제이에프이 스틸 가부시키가이샤 | 지르코늄계 화성 처리용 냉연 강판 및 그의 제조 방법 그리고 지르코늄계 화성 처리 강판 및 그의 제조 방법 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04276060A (ja) * | 1991-02-28 | 1992-10-01 | Sumitomo Metal Ind Ltd | 冷延鋼板及びその製造方法 |
| JPH09310163A (ja) * | 1996-05-20 | 1997-12-02 | Kawasaki Steel Corp | プレス加工性及びメッキ密着性に優れる高強度溶融亜鉛メッキ鋼板 |
| JPH10158784A (ja) * | 1996-11-27 | 1998-06-16 | Kawasaki Steel Corp | 高強度熱延鋼板 |
| JP2001342543A (ja) * | 2000-03-30 | 2001-12-14 | Nippon Steel Corp | 穴拡げ性と延性に優れた高強度熱延鋼板及びその製造方法 |
| JP2003147486A (ja) * | 2002-11-05 | 2003-05-21 | Kawasaki Steel Corp | 高強度冷延鋼板 |
| JP2004323969A (ja) * | 2003-04-10 | 2004-11-18 | Kobe Steel Ltd | 化成処理性に優れる高強度冷延鋼板 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0768583B2 (ja) * | 1984-03-07 | 1995-07-26 | 住友金属工業株式会社 | 高張力冷延鋼板の製造法 |
| JP2562049B2 (ja) * | 1989-01-11 | 1996-12-11 | 株式会社神戸製鋼所 | 局部変形能にすぐれる高強度冷延鋼板の製造方法 |
| JP2793284B2 (ja) * | 1989-08-29 | 1998-09-03 | 株式会社神戸製鋼所 | 焼付硬化性の優れた超高強度冷延鋼板の製造方法 |
| JP3254107B2 (ja) * | 1995-05-19 | 2002-02-04 | 株式会社神戸製鋼所 | 耐遅れ破壊特性にすぐれる超高強度鋼板及びその製造方法 |
| JPH11193418A (ja) * | 1997-12-29 | 1999-07-21 | Kobe Steel Ltd | 平坦性に優れた高強度冷延鋼板の製造方法 |
| CA2310335C (fr) * | 1998-09-29 | 2009-05-19 | Kawasaki Steel Corporation | Feuille mine d'acier haute resistance, feuille d'acier allie haute resistance revetue de zinc et galvanisee a chaud et procede de production correspondant |
| CN1170954C (zh) * | 1999-02-25 | 2004-10-13 | 川崎制铁株式会社 | 钢板、热镀钢板和合金化热镀钢板的制备方法 |
| EP1193322B1 (fr) † | 2000-02-29 | 2006-07-05 | JFE Steel Corporation | Tole d'acier laminee a froid a haute resistance presentant d'excellentes proprietes de durcissement par vieillissement par l'ecrouissage |
| JP2002173714A (ja) * | 2000-09-29 | 2002-06-21 | Kawasaki Steel Corp | 高張力溶融めっき鋼板およびその製造方法 |
| JP4085583B2 (ja) † | 2001-02-27 | 2008-05-14 | Jfeスチール株式会社 | 高強度冷延溶融亜鉛メッキ鋼板およびその製造方法 |
| FR2830260B1 (fr) * | 2001-10-03 | 2007-02-23 | Kobe Steel Ltd | Tole d'acier a double phase a excellente formabilite de bords par etirage et procede de fabrication de celle-ci |
-
2003
- 2003-12-25 JP JP2003429151A patent/JP3934604B2/ja not_active Expired - Lifetime
-
2004
- 2004-11-30 DE DE602004015922T patent/DE602004015922T3/de active Active
- 2004-11-30 US US10/998,950 patent/US20050139293A1/en not_active Abandoned
- 2004-11-30 EP EP04028368.1A patent/EP1548142B2/fr not_active Expired - Fee Related
- 2004-11-30 AT AT04028368T patent/ATE405685T1/de active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04276060A (ja) * | 1991-02-28 | 1992-10-01 | Sumitomo Metal Ind Ltd | 冷延鋼板及びその製造方法 |
| JPH09310163A (ja) * | 1996-05-20 | 1997-12-02 | Kawasaki Steel Corp | プレス加工性及びメッキ密着性に優れる高強度溶融亜鉛メッキ鋼板 |
| JPH10158784A (ja) * | 1996-11-27 | 1998-06-16 | Kawasaki Steel Corp | 高強度熱延鋼板 |
| JP2001342543A (ja) * | 2000-03-30 | 2001-12-14 | Nippon Steel Corp | 穴拡げ性と延性に優れた高強度熱延鋼板及びその製造方法 |
| JP2003147486A (ja) * | 2002-11-05 | 2003-05-21 | Kawasaki Steel Corp | 高強度冷延鋼板 |
| JP2004323969A (ja) * | 2003-04-10 | 2004-11-18 | Kobe Steel Ltd | 化成処理性に優れる高強度冷延鋼板 |
Non-Patent Citations (6)
| Title |
|---|
| DATABASE WPI Section Ch Week 200477, Derwent World Patents Index; Class M24, AN 2004-780743, XP002314973 * |
| PATENT ABSTRACTS OF JAPAN vol. 017, no. 073 (C - 1026) 15 February 1993 (1993-02-15) * |
| PATENT ABSTRACTS OF JAPAN vol. 1998, no. 04 31 March 1998 (1998-03-31) * |
| PATENT ABSTRACTS OF JAPAN vol. 1998, no. 11 30 September 1998 (1998-09-30) * |
| PATENT ABSTRACTS OF JAPAN vol. 2002, no. 04 4 August 2002 (2002-08-04) * |
| PATENT ABSTRACTS OF JAPAN vol. 2003, no. 09 3 September 2003 (2003-09-03) * |
Cited By (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2671961A1 (fr) * | 2005-03-31 | 2013-12-11 | Kabushiki Kaisha Kobe Seiko Sho | Feuille d'acier laminée à froid à haute résistance mécanique et pièces d'automobiles en acier ayant d'excellentes propriétés d'adhésion de film de revêtement, maniabilité et résistivité face à la fragilisation par l'hydrogène |
| EP2671960A1 (fr) * | 2005-03-31 | 2013-12-11 | Kabushiki Kaisha Kobe Seiko Sho | Feuille d'acier laminée à froid à haute résistance mécanique et pièces d'automobiles en acier ayant d'excellentes propriétés d'adhésion de film de revêtement, maniabilité et résistivité face à la fragilisation par l'hydrogène |
| EP1865085A4 (fr) * | 2005-03-31 | 2010-07-28 | Kobe Steel Ltd | Tole d acier laminee a froid de resistance elevee, excellente en termes d adherence des revetements, d aptitude au faconnage et de resistance a la fragilisation par l'hydrogene, et composant en acier pour automobiles |
| US8986468B2 (en) | 2005-03-31 | 2015-03-24 | Kobe Steel, Ltd. | High-strength cold-rolled steel sheet excellent in coating adhesion, workability and hydrogen embrittlement resistance, and steel component for automobile |
| EP2679699A3 (fr) * | 2005-03-31 | 2014-08-20 | Kabushiki Kaisha Kobe Seiko Sho | Feuille d'acier laminée à froid à haute résistance mécanique et pièces d'automobiles en acier ayant d'excellentes propriétés d'adhésion de film de revêtement, maniabilité et résistivité face à la fragilisation par l'hydrogène |
| EP1865085A1 (fr) * | 2005-03-31 | 2007-12-12 | Kabushiki Kaisha Kobe Seiko Sho | Tole d acier laminee a froid de resistance elevee, excellente en termes d adherence des revetements, d aptitude au faconnage et de resistance a la fragilisation par l'hydrogene, et composant en acier pour automobiles |
| GB2433746B (en) * | 2005-12-27 | 2009-07-01 | Kobe Steel Ltd | Steel sheet having excellent weldability |
| US9085816B2 (en) | 2005-12-27 | 2015-07-21 | Kobe Steel, Ltd. | Steel sheet having excellent weldability |
| GB2433746A (en) * | 2005-12-27 | 2007-07-04 | Kobe Steel Ltd | Steel sheet with excellent weldability |
| WO2008102009A1 (fr) * | 2007-02-23 | 2008-08-28 | Corus Staal Bv | Feuillard d'acier à résistance élevée, laminé à froid et trempé en continu, et procédé de fabrication dudit feuillard |
| EP3421634A1 (fr) * | 2007-02-23 | 2019-01-02 | Tata Steel IJmuiden B.V. | Bande d'acier haute résistance laminée à froid recuite en continu et procédé pour fabriquer ledit acier |
| EP2028282A1 (fr) | 2007-08-15 | 2009-02-25 | ThyssenKrupp Steel AG | Acier en phase double, produit plat à partir d'un tel acier en phase double et son procédé de fabrication |
| WO2009021897A1 (fr) * | 2007-08-15 | 2009-02-19 | Thyssenkrupp Steel Ag | Acier biphasé, produit plat constitué d'un tel acier biphasé et procédé de fabrication d'un produit plat |
| RU2470087C2 (ru) * | 2008-05-21 | 2012-12-20 | Арселормитталь Инвестигасьон И Десарролло Сл | Способ производства холоднокатаных листов из двухфазной стали, обладающей очень высокой прочностью, и полученные таким способом листы |
| WO2009150319A1 (fr) * | 2008-05-21 | 2009-12-17 | Arcelormittal Investigacion Y Desarrollo Sl | Procede de fabrication de toles d'aciers dual phase laminees a froid a tres haute resistance et toles ainsi produites |
| EP2123786A1 (fr) * | 2008-05-21 | 2009-11-25 | ArcelorMittal France | Procédé de fabrication de tôles d'aciers dual phase laminées à froid à trés haute résistance et tôles ainsi produites |
| US10190187B2 (en) | 2008-05-21 | 2019-01-29 | Arcelormittal | Manufacturing method for very high-strength, cold-rolled, dual-phase steel sheets |
| US8801873B2 (en) | 2009-03-30 | 2014-08-12 | Nippon Steel & Sumitomo Metal Corporation | Carburized steel part |
| CN102317490A (zh) * | 2009-03-30 | 2012-01-11 | 新日本制铁株式会社 | 渗碳钢部件 |
| CN102317490B (zh) * | 2009-03-30 | 2013-09-11 | 新日铁住金株式会社 | 渗碳钢部件 |
| US9598743B2 (en) | 2010-09-29 | 2017-03-21 | Jfe Steel Corporation | High strength steel sheet and method for manufacturing the same |
| EP2623630A4 (fr) * | 2010-09-30 | 2016-11-23 | Jfe Steel Corp | Tôle d'acier à haute résistance et procédé de production associé |
| US9534270B2 (en) | 2010-09-30 | 2017-01-03 | Jfe Steel Corporation | High strength steel sheet and method for manufacturing the same |
| RU2601037C2 (ru) * | 2011-11-28 | 2016-10-27 | Арселормитталь Инвестигасьон И Десарролло С.Л. | Высококремнистые двухфазные стали с улучшенной пластичностью |
| EP2785889A4 (fr) * | 2011-11-28 | 2016-03-02 | Arcelormittal Investigación Y Desarrollo Sl | Aciers double-phase comportant une teneur élevée en silicium dotés d'une ductilité améliorée |
| EP2829626B1 (fr) * | 2012-03-19 | 2019-06-26 | JFE Steel Corporation | Procédé de fabrication d'une feuille d'acier galvanisé par immersion à chaud à résistance élevée et feuille d'acier galvanisé par immersion à chaud à résistance élevée |
| US10449751B2 (en) | 2012-03-19 | 2019-10-22 | Jfe Steel Corporation | Method for manufacturing high strength galvanized steel sheet |
| WO2015014333A3 (fr) * | 2013-07-30 | 2015-11-05 | Salzgitter Flachstahl Gmbh | Acier multiphase à haute résistance, micro-allié et contenant du silicium, présentant une résistance minimale à la traction de 750 mpa et des propriétés améliorées et procédé de fabrication d'une bande à partir de cet acier |
| US10612113B2 (en) | 2013-07-30 | 2020-04-07 | Salzgitter Flachstahl Gmbh | Micro-alloyed high-strength multi-phase steel containing silicon and having a minimum tensile strength of 750 MPA and improved properties and method for producing a strip from said steel |
| EP3418419A4 (fr) * | 2016-03-31 | 2019-01-23 | JFE Steel Corporation | Tôle d'acier mince, tôle d'acier plaquée, procédé de production de tôle d'acier laminée à chaud, procédé de production de tôle d'acier laminée à froid entièrement dure, procédé de production de tôle d'acier mince et procédé de production de tôle d'acier plaquée |
| US11453926B2 (en) | 2016-03-31 | 2022-09-27 | Jfe Steel Corporation | Steel sheet, plated steel sheet, method for producing hot-rolled steel sheet, method for producing cold-rolled full hard steel sheet, method for producing steel sheet, and method for producing plated steel sheet |
Also Published As
| Publication number | Publication date |
|---|---|
| DE602004015922D1 (de) | 2008-10-02 |
| EP1548142B1 (fr) | 2008-08-20 |
| JP3934604B2 (ja) | 2007-06-20 |
| EP1548142B2 (fr) | 2013-07-03 |
| US20050139293A1 (en) | 2005-06-30 |
| JP2005187863A (ja) | 2005-07-14 |
| ATE405685T1 (de) | 2008-09-15 |
| DE602004015922T3 (de) | 2013-09-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1548142B1 (fr) | Tôle d'acier à résistance élevée laminée à froid, ayant une excellente adhesivité d'une couche de revêtement | |
| CN105143488B (zh) | 热轧钢板及其制造方法 | |
| KR100948998B1 (ko) | 도막 밀착성, 가공성 및 내수소취화 특성이 우수한 고강도 냉연 강판 및 자동차용 강 부품 | |
| JP3889768B2 (ja) | 塗膜密着性と延性に優れた高強度冷延鋼板および自動車用鋼部品 | |
| CA2714117C (fr) | Tole d'acier revetue de zinc en bain fondu a haute resistance presentant une excellente aptitude au faconnage et son procede de fabrication | |
| US20120180909A1 (en) | High strength cold rolled steel sheet and method for manufacturing the same | |
| CN112805395B (zh) | 热轧钢板及其制造方法 | |
| EP2527482B1 (fr) | Procédé de production d'une tôle en acier galvanisé au trempé à haute résistance présentant une excellente stabilité matérielle et une excellente aptitude au traitement | |
| CN112840045B (zh) | 热轧钢板及其制造方法 | |
| TW201317366A (zh) | 高強度熔融鍍鋅鋼板及其製造方法 | |
| KR20230038239A (ko) | 열간 프레스 부재 및 그 제조 방법 | |
| US20240052466A1 (en) | Steel sheet, member, and methods for manufacturing the same | |
| JP5659604B2 (ja) | 高強度鋼板およびその製造方法 | |
| JP7136335B2 (ja) | 高強度鋼板及びその製造方法 | |
| CN113366126B (zh) | 高强度钢板及其制造方法 | |
| JP2007297700A (ja) | 燃料タンク用高強度溶融アルミニウムめっき鋼板及びその製造方法 | |
| EP4043593B1 (fr) | Tôle en acier hautement résistante ainsi que procédé de fabrication de celle-ci, et élément d'absorption de chocs | |
| EP4043594B1 (fr) | Tôle en acier hautement résistante ainsi que procédé de fabrication de celle-ci, et élément d'absorption de chocs | |
| JP7444097B2 (ja) | 熱延鋼板およびその製造方法 | |
| WO2023095870A1 (fr) | Tôle d'acier galvanisée | |
| WO2021125283A1 (fr) | Tôle d'acier et son procédé de fabrication | |
| EP4350015A1 (fr) | Tôle d'acier, élément, et procédés de fabrication de ceux-ci | |
| EP4350016A1 (fr) | Tôle d'acier, élément, et procédés de fabrication de ceux-ci | |
| US20240068065A1 (en) | Steel sheet, member, and methods for manufacturing the same | |
| WO2023132344A1 (fr) | Tôle en acier, et procédé de fabrication de celle-ci |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LU MC NL PL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL HR LT LV MK YU |
|
| 17P | Request for examination filed |
Effective date: 20050714 |
|
| AKX | Designation fees paid |
Designated state(s): AT DE FR GB |
|
| 17Q | First examination report despatched |
Effective date: 20071026 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT DE FR GB |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 602004015922 Country of ref document: DE Date of ref document: 20081002 Kind code of ref document: P |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| 26 | Opposition filed |
Opponent name: CORUS STAAL BV Effective date: 20090520 |
|
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PLAB | Opposition data, opponent's data or that of the opponent's representative modified |
Free format text: ORIGINAL CODE: 0009299OPPO |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PLAB | Opposition data, opponent's data or that of the opponent's representative modified |
Free format text: ORIGINAL CODE: 0009299OPPO |
|
| R26 | Opposition filed (corrected) |
Opponent name: TATA STEEL IJMUIDEN BV Effective date: 20090520 |
|
| PUAH | Patent maintained in amended form |
Free format text: ORIGINAL CODE: 0009272 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT MAINTAINED AS AMENDED |
|
| 27A | Patent maintained in amended form |
Effective date: 20130703 |
|
| AK | Designated contracting states |
Kind code of ref document: B2 Designated state(s): AT DE FR GB |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R102 Ref document number: 602004015922 Country of ref document: DE Effective date: 20130703 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 12 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 13 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20191119 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20191014 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20191025 Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20191129 Year of fee payment: 16 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602004015922 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 405685 Country of ref document: AT Kind code of ref document: T Effective date: 20201130 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20201130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20201130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20201130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210601 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20201130 |