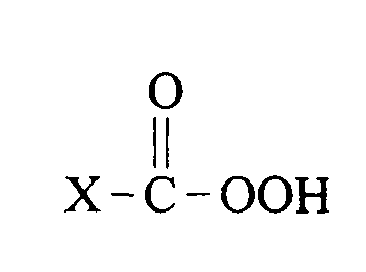EP1212398B1 - Bleaching detergent compositions - Google Patents
Bleaching detergent compositions Download PDFInfo
- Publication number
- EP1212398B1 EP1212398B1 EP00959694A EP00959694A EP1212398B1 EP 1212398 B1 EP1212398 B1 EP 1212398B1 EP 00959694 A EP00959694 A EP 00959694A EP 00959694 A EP00959694 A EP 00959694A EP 1212398 B1 EP1212398 B1 EP 1212398B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- weight
- acid
- composition
- preferred
- sulfate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/48—Medical, disinfecting agents, disinfecting, antibacterial, germicidal or antimicrobial compositions
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/02—Inorganic compounds ; Elemental compounds
- C11D3/04—Water-soluble compounds
- C11D3/10—Carbonates ; Bicarbonates
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/20—Organic compounds containing oxygen
- C11D3/2075—Carboxylic acids-salts thereof
- C11D3/2082—Polycarboxylic acids-salts thereof
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/20—Organic compounds containing oxygen
- C11D3/2075—Carboxylic acids-salts thereof
- C11D3/2086—Hydroxy carboxylic acids-salts thereof
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3902—Organic or inorganic per-compounds combined with specific additives
- C11D3/3905—Bleach activators or bleach catalysts
- C11D3/3907—Organic compounds
- C11D3/3915—Sulfur-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3902—Organic or inorganic per-compounds combined with specific additives
- C11D3/3905—Bleach activators or bleach catalysts
- C11D3/3907—Organic compounds
- C11D3/3917—Nitrogen-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3942—Inorganic per-compounds
Definitions
- This invention provides specific sanitising compositions and methods for improved sanitisation of fabrics.
- Hydrogen peroxide bleach containing dishwashing compositions are described in DE 4232170 and DE19547430.
- weakly alkaline machine dish-washing compositions comprising sodium citrate, alkali metal hydrogen carbonate, bleaching agents, bleach activators and enzymes.
- part of the sodium citrate is replaced with a water soluble natural substance selected from mono and/or disaccharides, sugar acids and protein hydrolysates.
- bleach is not always compatible with other detergent ingredients, such as enzymes, or moreover, the levels of bleach required to obtain disinfecting are too high and may damage the fabrics.
- peroxyacid bleach precursors can be the preferred bleach species to deliver sanitisation of fabrics during the wash. They found however that a problem with these bleach precursors is that they form the peroxyacid bleach at a high, alkaline pH, but that at such a high, alkaline pH the peroxyacid, which is found to be the active species which gives the sanitisation benefits, dissociates to the inactive form, resulting thus in reduced sanitisation.
- a preferred bleaching ingredient in detergents is a percarbonate salt.
- the percarbonate makes the solution very alkaline, which is as set out above not desirable when aiming to keep high levels of the active species, i.e. the peroxyacid itself, and thus the antimicrobial performance thereof. Also many builders provide an alkaline pH in the wash, which is not desirable when seeking an efficient sanitisation.
- the inventors have now found a way to provide efficient antimicrobial performance and/or sanitisation whilst a good cleaning of both bleachable and non-bleachable stains is achieved. They found that when the pH of the detergent in the wash water is controlled, namely from 8.8 to 9.9, preferably from 9.0 or even 9.2 to 9.85 or even to 9.8, in particular achieved by using specific levels of acid and alkaline ingredients and optionally buffers, excellent sanitisation and cleaning is obtained, whilst only limited amounts of precursor are required, making the compositions more storage stable and safer to use. They found that these detergent compositions can then even contain percarbonate bleach.
- compositions of the invention are preferably used to effectively provide sanitisation of fabrics or, effectively inactivate micro-organisms, in particular bacteria which are difficult to inactivate, such as in particular E. Hirae, but also S. Aureus, E Coli and Ps Aeruginosa.
- the invention relates to a laundry detergent composition
- a laundry detergent composition comprising, at least 15% by weight of a carbonate source and a bleach system which contains at least 2.5% by weight of a peroxyacid bleach precursor and a hydrogen peroxide source, which may include a salt of a percarbonate and may thus be part of the carbonate source, and at least 7% by weight of an acid, preferably an organic acid, more preferably including a organic carboxylic acid, characterised in that a 1% by weight mixture of the composition in demineralised water at a temperature of 20°C obtained after 10 minutes stirring at a speed of 200rpm, provides a pH from 8.8 to 9.9 and which further comprises an anionic surfactant.
- the invention also relates to a method of providing sanitisation during the wash by use of a composition of the invention and a method for inactivate E. Hirae, S. Aureus, E. Coli and Ps Aeruginosa.
- the inventors have also found that, when the laundry compositions of the invention are for use in automatic laundry methods, improved antimicrobial performance or sanitisation of the fabrics is achieved when the composition is delivered directly into the drum of the washing machine, for example by use of a dispensing device.
- One embodiment of the invention thus relates to a washing method for washing fabrics whereby the composition of the invention is introduced into the drum of a washing machine by placing a dispensing device comprising the composition in the drum prior to the introduction of wash water.
- the composition of the invention is preferably such that a 1% mixture of the composition in demineralised water, at a temperature of 20°C. obtained after 10 minutes stirring at a speed of200rpm, provides a pH from 9.0 to 9.85 or even from 9.2 to 9.85 or even to 9.8, or even from 9.4 to 9.8.
- the pH is measured after stirring the mixture, for 10 minutes at a constant temperature, by any conventional pH measurement method.
- '1% mixture is meant any mixture, dispersion or solution of 1 weight unit of the composition in 99 weight units of demineralised water, and it is thus to be understood that if the composition is not dissolved fully after 10 minutes, the pH is measured for such a mixture or dispersion.
- 'reduction of the activity of the micro-organisms' includes a reduction of the activity as defined in the CEN method prEN1276, 1993, (ComInstitut Euroworth de Normalisation) and CEN method prEN 1650.
- the reduction therein is preferably represented by a reduction of the specific micro-organism or bacteria by at least 10 5 , more preferably at least 2x 10 5 , or even 3x 10 5 .
- the reduction of the activity of the micro-organisms when used herein includes the reduction of any of the processes conducted by the micro-organism, including secretion of products but preferably the growth of the micro-organisms.
- the invention also provides a method for reduction of the activity of micro-organisms, or preferably specific bacteria, comprising the steps of contacting the micro-organism with an aqueous liquor comprising the composition of the invention such that the peroxyacid provided by the precursor is present at a level of at least 100 ppm in the liquor, per 10 6 micro-organisms.
- the composition is such that at least the activity of E. Hirae, but more preferably also of S. Aureus, E Coli and Ps Aeruginosa, is reduced by at least 10 5 , preferably at least 2 x 10 5 or even 3 x 10 5 , as measured by the above CEN method.
- the activity reduction is done during a laundering process.
- the composition of the invention can be contacted with the specific micro-organisms in solid form, but preferably the bleaching agent is present in a liquor in contact with the micro-organisms.
- the specific micro-organisms preferably the specific bacteria
- the bacteria can also be present in the liquor used for the cleaning or sanitisation process or in the equipment used in the process, such as the washing machine or dispensing device.
- composition of the invention is particularly useful in inactivating micro-organisms having cell walls comprising relatively high levels of peptidoglycan, in particular gram positive bacteria, which can be present in soils or stains on the fabrics or surfaces, in particular in body soils.
- the amount of composition required to obtain effective reduction of the activity of the specific micro-organisms depends on various factors, such as the amount of micro-organisms present, the conditions of the sanitisation or cleaning process, including the other compounds present and the temperature of the cleaning process.
- the liquor used preferably comprises at least 100 ppm of the peroxyacid per 10 6 micro-organisms, more preferably at least 200 ppm or even 250 ppm.
- the reduction of the activity of the specific micro-organisms, having a peptidoglycan-containing cell walls can be determined by the Petrocci and Clarke method, as described in JOAC 1981, but is preferably determined for the purpose of this invention by the CEN method prEN1276, 1993 for bacteria and CEN method prEN1650 for yeast.
- Such a CEN method involves, for example, the preparation of gram positive bacterial inocula conform the CEN method, pages 7 and further, preparation of a solution comprising the hydrophobic bleaching agent at a level of about 250ppm, conducting the test following the CEN method, incubation TSA plates for 24 hours at 36°C; and subsequently counting of the bacteria colonies on the plates.
- the composition comprises at least 7% by weight of one or more acids, preferably an organic acid, preferably including at lest an organic carboxylic acid.
- acids preferably an organic acid, preferably including at lest an organic carboxylic acid.
- the exact levels will depend on the other ingredients of the detergent composition and the alkalinity thereof, so that the level of the acid is adjusted to provide the required pH.
- the acid is present at a level of at least 8% by weight of the composition or even at least 9% or even at least 10% by weight of the composition, with as upper limit a level of preferably 30% or even 20% by weight of the composition.
- Suitable acids to be used herein include materials which not only help to provide the required pH of the formulation, but which also have a secondary function in the composition, such as acting as a chelating agent, builder and/or effervescence source.
- Useful inorganic acids include boric acid, bisulphite salts preferably sodium salts thereof.
- the acid comprises at least an organic carboxylic acid.
- Such acids include mono- or polycarboxylic acids preferably citric acid, adipic acid, glutaric acid, 3 chetoglutaric acid, citramalic acid, tartaric acid, maleic acid, fumaric acid, malic acid, succinic acid, malonic acid but also polymeric or oligomeric polycarboxylic acids, such as acrylic acid polymers or maleic acid polymers, or copolymers of maleic acid and acrylic acid.
- organic carboxylic acid herein comprises at least 7% by weight of the composition of maleic acid, malic acid or citric acid, or mixtures thereof.
- mixtures of organic carboxylic acids and inorganic acids are used, for example in ratio's of from 1:1 to 10:1 1 or even from 2:1 to 5:1.
- the acid may be present as a separate component or in solid compositions of the invention, the acid may be incorporated in a granule together with other detergent ingredients.
- the acid is a particulate material whereof at least 75%, preferably at least 85% or even at least 90% or even at least 95% or even at least 99% by volume, has a particle size from 1 to 500 microns and more preferably from 1 to 350 microns and it may even be preferred that at least 65% or even at least 75% or even at least 85% has a particle size from 1.0 to 250 microns or even from 1.0 to 150 microns.
- particle sizes can be determined by any method known in the art, in particular by laser light scattering or difraction technique, such as with Malvem 2600 or Sympatec Helos laser light scattering equipment (or difractometer).
- the particle size distribution and mean is preferably similar to particle size distribution and mean of the other components of the composition, as described hereinafter.
- Salts of inorganic acids and/or salts or esters of organic carboxylic acids may also be present as additional components, in particular because it may be beneficial to buffer the composition at the required pH.
- the composition comprises a salt of citric acid, maleic acid, malic acid, glutaric acid or tartaric acid or borate salt, or mixtures thereof.
- composition of the present invention comprises at least 15% by weight of a carbonate source.
- Preferred carbonate sources include carbonate, bicarbonate and percarbonate salts and preferably the carbonate source comprises a mixture of three salts.
- the percarbonate salt when present, forms part of both the carbonate source and the hydrogen peroxide source as defined herein.
- the composition comprises a percarbonate salt to provide hydrogen peroxide and carbonate.
- the composition when the composition comprises at least 10% by weight of a percarbonate salt, as hydrogen peroxide source and carbonate source, the composition also contains at least 5% by weight of another carbonate source.
- the composition comprises a mixture of carbonate sources, preferably including a carbonate salt, bicarbonate salt and percarbonate salt. Then, the composition comprises preferably at least 5% by weight of a carbonate salt, at least 2% by weight of a bicarbonate salt and at least 8% by weight of a percarbonate salt, more preferably at least 7% by weight of a carbonate salt , at least 4% by weight of a bicarbonate salt and at least 15% by weight of a percarbonate salt.
- Suitable carbonates sources to be used herein include carbonate and hydrogen carbonate or bicarbonate of earth alkali or alkali metals and sodium and potassium salts.
- the inventors found that it may be particularly useful to include a bicarbonate salt into the compositions, because it has been found that the bicarbonate salt provides a buffered pH at around the pH value needed to form the peroxyacids from the precursors, for example of TAED as described hereinafter.
- the composition comprises at least 1%, preferably at least 2% or even at least 4% by weight of the composition of an alkali or earth alkali salt of bicarbonate, preferably sodium bicarbonate.
- This salt may be present up to any level, preferably up to 20% by weight of the composition, more preferably up to 15% or even up to 10% by weight of the composition.
- the carbonate, bicarbonate and percarbonate salts herein my be present as separate particulate components or may be incorporated in detergent granules together with other detergent ingredients.
- the carbonate and bicarbonate material when present in other detergent granules, may preferably have a volume median particle size from 1 to 500 microns, whereby preferably at least 60%, preferably at least 70% or even at least 80% or even at least 90% by volume, has a particle size of from 0.5 to 1180 microns.
- the carbonate or bicarbonate salts has a volume median particle size of 10 to 375, whereby preferably at least 60 %, or even at least 70% or even at least 80% or even at least 90% by volume, has a particle size of from 1 to 850 microns; or even preferably a volume median particle size from 10 to 250 microns, whereby preferably at least 60 %, preferably at least 70% or even at least 80% or even at least 90% by volume, has a particle size of from 5 to 425 microns.
- the required particle size of the carbonate and/or bicarbonate salt is obtained by grinding a larger particle size material, optionally followed by selecting the material with the required particle size by any suitable method.
- the carbonate source is incorporated in the composition in such a manner that it is capable to react with the acid to provide effervescing upon contact with water.
- compositions of the invention comprise a hydrogen peroxide source.
- this is a persalt such a salts of percarbonate, perborate, perphosphate, peroxymonopersulfate and persilicate salts.
- the inorganic perhydrate salts are normally the alkali metal salts.
- the alkali metal salt of percarbonate, perborate, or mixtures thereof, are the preferred inorganic perhydrate salts for use herein.
- Preferred perborate is sodium perborate in the form of the monohydrate or tetrahydrate, respectively of nominal formula NaBO 2 H 2 O 2 and NaBO 2 H 2 O 2 .3H 2 O.
- Other suitable oxygen bleaches include persulphates, particularly potassium persulphate K 2 S 2 O 8 and sodium persulphate Na 2 S 2 O 8 .
- an alkali metal salt of percarbonate preferably sodium percarbonate, which then forms also part of the carbonate source, described above.
- compositions in the present invention comprise from 5% to 50% by weight of the total composition of a hydrogen peroxide source, preferably from 10% to 45% and more preferably from 15% to 35% by weight or even form 17% or even 19% to 30% or even to 25% by weight of the composition.
- the persalts may be coated by any coating material, preferably comprising a sulphate salt, such as magnesium sulphate or preferably sodium sulphate, carbonate or bicarbonate salt or mixtures thereof.
- a sulphate salt such as magnesium sulphate or preferably sodium sulphate, carbonate or bicarbonate salt or mixtures thereof.
- the granular compositions herein comprise a peroxyacid bleach precursor.
- the peroxyacid bleach precursor is present at a level of from 3% to 10% or even 3.5% to 8% or even 4% to 6% by weight of the composition.
- Peroxyacid bleach precursors are compounds which react with hydrogen peroxide in a perhydrolysis reaction to produce a peroxyacid.
- peroxyacid bleach precursors may be represented as where L is a leaving group and X is essentially any functionality, such that on perhydroloysis the structure of the peroxyacid produced is
- Suitable peroxyacid bleach precursor compounds typically contain one or more N- or O-acyl groups, which precursors can be selected from a wide range of classes.
- Suitable classes include anhydrides, esters, imides, lactams and acylated derivatives of imidazoles and oximes. Examples of useful materials within these classes are disclosed in GB-A-1586789.
- Suitable esters are disclosed in GB-A-836988, 864798, 1147871, 2143231 and EP-A-0170386.
- L group The leaving group, hereinafter L group, must be sufficiently reactive for the perhydrolysis reaction to occur within the optimum time frame (e.g., a wash cycle). However, if L is too reactive, this activator will be difficult to stabilize for use in a bleaching composition.
- Preferred L groups are selected from the group consisting of: and mixtures thereof, wherein R 1 is an alkyl, aryl, or alkaryl group containing from 1 to 14 carbon atoms, R 3 is an alkyl chain containing from 1 to 8 carbon atoms, R 4 is H or R 3 , and Y is H or a solubilizing group. Any of R 1 , R 3 and R 4 may be substituted by essentially any functional group including, for example alkyl, hydroxy, alkoxy, halogen, amine, nitrosyl, amide and ammonium or alkyl ammmonium groups.
- the preferred solubilizing groups are -SO 3 - M + , -CO 2 - M + , -SO 4 - M + , -N + (R 3 ) 4 X - and O ⁇ --N(R 3 ) 3 and most preferably -SO 3 - M + and -CO 2 - M + wherein R 3 is an alkyl chain containing from 1 to 4 carbon atoms, M is a cation which provides solubility to the bleach activator and X is an anion which provides solubility to the bleach activator.
- M is an alkali metal, ammonium or substituted ammonium cation, with sodium and potassium being most preferred, and X is a halide, hydroxide, methylsulfate or acetate anion.
- Preferred examples of such compounds are tetracetyl ethylene diamine, (TAED), sodium 3, 5, 5-trimethyl hexanoyloxybenzene sulphonate, nonylamide of peroxyadipic acid and n-nonanoyloxybenzenesulphonate (NOBS), -decanoyloyloxybenzenesulphonate, dodecanoyloxybenzenesulphonate and acetyl triethyl citrate (ATC), Phenolsulphonate Ester of N-nonanoyl-6-aminocaproic acid, aliphatic diacyl peroxide (DAP) having the general formula R-C(O)-O-O-(O)C-R1, wherein R and R1 can be the same or different and are linear or branched aliphatic groups having from 6 to 20 carbon atoms.
- DAP aliphatic diacyl peroxide
- N-acyl caprolactam selected from the group consisting of substituted or unsubstituted benzoyl caprolactam, octanyl caprolactam, nonanoyl caprolactam, hexanoyl caprolactam, decanoyl caprolactam, undecenoyl caprolactam, formyl caprolactam, acetyl caprolactam, propanoyl caprolactam, butanoyl caprolactam pentanoyl caprolactam.
- Amide substituted alkyl peroxyacid precursor compounds are suitable herein, including those of the following general formulae: wherein R 1 is an alkyl group with from 1 to 14 carbon atoms, R 2 is an alkylene group containing from 1 to 14 carbon atoms, and R 5 is H or an alkyl group containing 1 to 10 carbon atoms and L can be essentially any leaving group, preferably a oxybenzene sulfonate.
- Amide substituted bleach activator compounds of this type are described in EP-A-0170386.
- Nonanamido caproyl oxybenzene sulfonate preferably in the form of the sodium salt, NACA-OBS, is a preferred precursor of this type.
- a highly preferred bleach precursor herein is TAED, NACA-OBS, DOBS and/ or NOBS.
- the compositions herein may comprise mixtures of said bleach activators, because it has been found that this may result in improved sanitisation.
- Highly preferred is a mixture of TAED and a precursor selected from NOBS, DOBS or NACA-OBS. It has been found that further improved sanitisation is achieved when such mixed precursor systems are used, in particular when the ratio of TAED to NOBS, DOBS or NACA-OBS is from 1:2 to 2:1.
- the precursor may be in the form of a separate detergent granule, which may be coated, or in the form of a detergent granule comprising also other detergent actives. It is preferably present in the form of a particulate component having a similar particle size distribution and mean particle size as the other detergent particles.
- the inventors also found that it can be beneficial when the precursor and hydrogen peroxide source are present intimately mixed with one another, for example in a granule.
- This mixture or granule is preferably being substantially free of organic acids.
- the peroxy acid formed of the precursor is not part of the acid component of the composition of the invention.
- Machine laundry methods herein typically comprise treating soiled laundry with an aqueous wash solution in a washing machine having dissolved or dispensed therein an effective amount of a detergent in accord with the invention.
- laundry washing methods are envisaged herein.
- an effective amount of the laundry detergent composition for laundry washing is normally meant from 10g to 300g of product dissolved or dispersed in a wash solution of volume from 5 to 65 litres, as are typical product dosages and wash solution volumes commonly employed in conventional machine laundry methods. Dosage is dependent upon the particular conditions such as water hardness and degree of soiling of the soiled laundry.
- the reduction of the activity of the micro-organisms present in the wash and on the soiled fabrics preferably takes place during a washing method, preferably an automatic or hand laundry, at low temperatures up to 60°C or even up to 45°C or even around 30°C.
- the detergent composition may be dispensed for example, from the drawer dispenser of a washing machine or may be sprinkled over the soiled laundry placed in the machine.
- a dispensing device is employed in the washing method, to introduce the composition of the invention directly in the drum of the wash. It has been found that improved sanitisation is then achieved.
- Any dispensing device can be used, including preferred dispensing devices for use with the composition of the invention have been described in the following patents; GB-B-2, 157, 717, GB-B-2, 157, 718, EP-A-0201376, EP-A-0288345 and EP-A-0288346, WO94/11562.
- compositions of the present invention can be prepared with different bulk densities, preferably being from 300 to 1200 g/l, preferably from 500 to 1100 g/l or even 550g/l to 900g/l.
- These compositions are preferably solid compositions, preferably solid laundry detergents, which can be made by a variety of methods well known in the art, including dry-mixing, spray drying, extrusion, roller compaction, agglomeration and combinations thereof.
- the solid compositions may have any form, preferably granular or in the form of a tablet or bar.
- the composition is made by mixing all or part of the granules, including those made by agglomeration or spray-drying, and subsequently adding a binder and mixing or agglomerating the granules and binder to form the, preferably agglomerated detergent granules. These may be of the required particle size or they may be sieved to obtain particles of the required size.
- the acid, or part thereof, and the carbonate source or part thereof are present in an intimate mixture with one another, preferably in a granule, which means for the purpose of the invention that the acid and carbonate source are preferably homogeneously mixed.
- the weight ratio of acid to carbonate source in the intimate mixture or the effervescent granule is preferably from 0.1 to 10, preferably from 0.5 to 2.5 and more preferably from 1 to 2.
- the acid is preferably present in such a granule at a level of from 5% to 85% by weight of the total granule, preferably from 10% to 75%, more preferably from 15% to 60% and most preferably from 10% to 50%.
- the carbonate source is preferably present in such a granule at a level of from 5% to 90% by weight of the total, preferably from 10% to 80%, more preferably from 20% to 75% and most preferably from 30% to 65%.
- the granule is preferably substantially free of water, i.e. no water has been added or present other than the moisture of the raw materials themselves.
- the level of water is below 5% by weight of the total intimate mixture or granule, preferably below 3% and more preferably below 1.5%.
- a desiccant is present in the intimate mixture or the granule, such as overdried inorganic and organic salts, anhydrous salts, in particular overdried silicates and aluminosilicates, anhydrous silicates and/or sulphate salts.
- the granules are preferably obtainable by a process comprising a granulation step, preferably comprising the step of dry-powder compaction or pressure agglomeration. While all binding mechanisms can occur in pressure agglomeration, adhesion forces between the solid particles, i.e., between the acid, carbonate source and optionally the binder if present, play an especially important role.
- pressure agglomeration is an essentially dry process that forms new entities (i.e., dry effervescent granules) from solid particles (i.e., the acid, bicarbonate, carbonate source and optionally the binder) by applying external forces to densify a more or less defined bulk mass or volume and create binding mechanisms between the solid particles providing strength to the new entity, i.e. the high external force applied brings the solid particles closely together.
- new entities i.e., dry effervescent granules
- solid particles i.e., the acid, bicarbonate, carbonate source and optionally the binder
- the effervescent granules may optionally comprise a binder or a mixture thereof.
- the granules comprise up to 50 % by weight of the total granule of a binder or a mixture thereof, preferably up to 35% and more preferably up to 20%.
- Suitable binders to use herein are those known to those skilled in the art and include anionic surfactants like C6-C20 alkyl or alkylaryl sulphonates or sulphates, preferably C8-C20 alkylbenzene sulphonates, cellulose derivatives such as carboxymethylcellulose and homo- or copolymeric polycarboxylic acid or their salts, nonionic surfactants, preferably C10-C20 alcohol ethoxylates containing from 5-100 moles of ethylene oxide per mole of alcohol and more preferably the C15-C20 primary alcohol ethoxylates containing from 20-100 moles of ethylene oxide per mole of alcohol.
- anionic surfactants like C6-C20 alkyl or alkylaryl sulphonates or sulphates, preferably C8-C20 alkylbenzene sulphonates, cellulose derivatives such as carboxymethylcellulose and homo- or copolymeric polycarboxylic acid or their salts
- binders include the polymeric materials like polyvinylpyrrolidones with an average molecular weight of from 12 000 to 700 000 and polyethylene glycols with an average weight of from 600 to 10 000. Copolymers of maleic anhydride with ethylene, methylvinyl ether, methacrylic acid or acrylic acid are other examples of polymeric binders. Others binders further include C 10-C20 mono and diglycerol ethers as well as C10-C20 fatty acids.
- the effervescent granules may have any particle size, the preferred particle size depending on the application and the component of the granule.
- the effervescing granules described above preferably comprise no percarbonate salt as carbonate source, but preferably comprise carbonate salts and/ or bicarbonate salts.
- the composition preferably comprises granules whereof at least 60%, more preferably at least 80% by weight have an average particle size, by weight, of from 400 microns to 1400 microns, preferably from 500 microns to 1100 microns or even 750 to 1000 microns.
- the composition comprises less than 20% or even less than 10% or even less than 5% by weight of particulate components of a particle size of less than 300 microns, or even less than 425 microns or even less than 600 microns; it may also be preferred that the composition comprises less than 20% or even less than 10% or even less than 5% by weight of the composition, of particulate components of a particle size of more than 1700 microns, or even more than 1400 microns or even more than 1180 microns.
- the compositions may comprise different granular components, which each have the above particle size requirements, and thus have a similar particle size distribution and mean particle size.
- Such preferred perfume granules of encapsulated perfume oils are described in co-pending European patent application EP-A-965326. Preferred levels of perfume granules will depend on the level of perfume oils therein, but generally the level is from about 0.05 to 8% or even 0.5% to 5% or even 0.8% to 3% by weight of the composition. Moreover, preferably the composition also comprises perfume components which are not in the form of encapsulated perfume oil granules, but preferably in the form of perfume components sprayed onto the detergent granules of the composition.
- compositions herein preferably contain one or more additional detergent components selected from surfactants, bleach catalysts, additional builders, additional organic polymeric compounds, enzymes, suds suppressors, lime soap, dispersants, soil suspension and anti-redeposition agents soil releasing agents, brightners, photobleaching agents and additional corrosion inhibitors.
- additional detergent components selected from surfactants, bleach catalysts, additional builders, additional organic polymeric compounds, enzymes, suds suppressors, lime soap, dispersants, soil suspension and anti-redeposition agents soil releasing agents, brightners, photobleaching agents and additional corrosion inhibitors.
- the levels of detergent ingredients are to be chosen such that the pH of 1% by weight of composition in demineralised water is as defined above.
- the level thereof is up to about 12% by weight or even up to 10% by weight.
- the detergent compositions herein preferably contain one or more surfactants.
- the surfactant may comprise any surfactant known in the art selected from anionic, nonionic, cationic, ampholytic, amphoteric and zwitterionic surfactants and mixtures thereof.
- the detergent composition may comprise surfactant which is not present in the intimate mixture with the crystalline layered silicate, but present in the other detergent components.
- compositions in accord with the present invention comprise an anionic surfactant.
- anionic surfactants useful for detersive purposes can be comprised in the detergent composition. These can include salts (including, for example, sodium, potassium, ammonium, and substituted ammonium salts such as mono-, di- and triethanolamine salts) of the anionic sulfate, sulfonate, carboxylate and sarcosinate surfactants. Anionic sulfate and sulfonate surfactants are preferred.
- surfactants systems comprising a sulfonate and a sulfate surfactant, preferably a linear or branched alkyl benzene sulfonate and alkyl ethoxylsulfates, as described herein, preferably combined with a cationic surfactants as described herein.
- anionic sulfonate surfactants particularly preferred herein include the salts of C 5 -C 20 linear or branched alkylbenzene sulfonates, but also alkyl ester sulfonates, C 6 -C 22 primary or secondary alkane sulfonates, C 6 -C 24 olefin sulfonates, sulfonated polycarboxylic acids, alkyl glycerol sulfonates, fatty acyl glycerol sulfonates, fatty oleyl glycerol sulfonates, and any mixtures thereof. Most preferred are C 9 -C 14 linear alkyl benzene sulfonates.
- Anionic sulfate surfactants suitable for use herein include the linear and branched primary and secondary alkyl sulfates, alkyl ethoxysulfates, fatty oleoyl glycerol sulfates, alkyl phenol ethylene oxide ether sulfates, the C 5 -C 17 acyl-N-(C 1 -C 4 alkyl) and -N-(C 1 -C 2 hydroxyalkyl) glucamine sulfates, and sulfates of alkylpolysaccharides such as the sulfates of alkylpolyglucoside (the nonionic nonsulfated compounds being described herein).
- Alkyl sulfate surfactants are preferably selected from the linear and branched primary C 10 -C 18 alkyl sulfates, more preferably the C 11 -C 15 branched chain alkyl sulfates and the C 12 -C 14 linear chain alkyl sulfates.
- Alkyl ethoxysulfate surfactants are preferably selected from the group consisting of the C 10 -C 18 alkyl sulfates which have been ethoxylated with from 0.5 to 20 moles of ethylene oxide per molecule. More preferably, the alkyl ethoxysulfate surfactant is a C 11- C 18 , most preferably C 11 -C 15 alkyl sulfate which has been ethoxylated with from 0.5 to 7, preferably from 1 to 5, moles of ethylene oxide per molecule.
- a particularly preferred aspect of the invention employs mixtures of the preferred alkyl sulfate and/ or sulfonate and alkyl ethoxysulfate surfactants. Such mixtures have been disclosed in PCT Patent Application No. WO 93/18124.
- R, R 1 , and R 2 are each independently selected from hydrogen and C 1 -C 3 alkyl (preferably methyl), provided R, R 1 , and R 2 are not all hydrogen and, when z is 0, at least R or R 1 is not hydrogen;
- w is an integer from 0 to 13;
- x is an integer from 0 to 13;
- y is an integer from 0 to 13;
- z is an integer from 0 to 13; and
- w + x + y + z is from 7 to 13.
- mid-chain branched surfactants compounds for use in the detergent compositions herein are mid-chain branched primary alkyl sulfonate and, even more preferably, sulfate surfactants. It should be understood that for the purpose of the invention, it may be preferred that the surfactant system comprises a mixture of two or more mid-chain branched primary alkyl sulfate or sulphonate surfactants.
- Preferred mid-chain branched primary alkyl_sulfate surfactants are of the formula
- These surfactants have a linear primary alkyl sulfate chain backbone (i.e., the longest linear carbon chain which includes the sulfated carbon atom) which preferably comprises from 12 to 19 carbon atoms and their branched primary alkyl moieties comprise preferably a total of at least 14 and preferably no more than 20, carbon atoms.
- the average total number of carbon atoms for the branched primary alkyl moieties is preferably within the range of from greater than 14.5 to about 17.5.
- the surfactant system preferably comprises at least one branched primary alkyl sulfate surfactant compound having a longest linear carbon chain of not less than 12 carbon atoms or not more than 19 carbon atoms, and the total number of carbon atoms including branching must be at least 14, and further the average total number of carbon atoms for the branched primary alkyl moiety is within the range of greater than 14.5 to about 17.5.
- R, R 1 , and R 2 are each independently selected from hydrogen and C 1 -C 3 alkyl group (preferably hydrogen or C 1 -C 2 alkyl, more preferably hydrogen or methyl, and most preferably methyl), provided R, R 1 , and R 2 are not all hydrogen. Further, when z is 1, at least R or R 1 is not hydrogen.
- Preferred mono-methyl branched primary alkyl sulfates are selected from the group consisting of: 3-methyl pentadecanol sulfate, 4-methyl pentadecanol sulfate, 5-methyl pentadecanol sulfate, 6-methyl pentadecanol sulfate, 7-methyl pentadecanol sulfate, 8-methyl pentadecanol sulfate, 9-methyl pentadecanol sulfate, 10-methyl pentadecanol sulfate, 11-methyl pentadecanol sulfate, 12-methyl pentadecanol sulfate, 13-methyl pentadecanol sulfate, 3-methyl hexadecanol sulfate, 4-methyl hexadecanol sulfate, 5-methyl hexadecanol sulfate, 6-methyl hexadecanol
- Preferred di-methyl branched primary alkyl sulfates are selected from the group consisting of: 2,3-methyl tetradecanol sulfate, 2,4-methyl tetradecanol sulfate, 2,5-methyl tetradecanol sulfate, 2,6-methyl tetradecanol sulfate, 2,7-methyl tetradecanol sulfate, 2,8-methyl tetradecanol sulfate, 2,9-methyl tetradecanol sulfate, 2,10-methyl tetradecanol sulfate, 2,11-methyl tetradecanol sulfate, 2,12-methyl tetradecanol sulfate, 2,3-methyl pentadecanol sulfate, 2,4-methyl pentadecanol sulfate, 2,5-methyl pentadecanol sulfate, 2,6-methyl penta
- any alkoxylated nonionic surfactants are suitable herein.
- the ethoxylated and propoxylated nonionic surfactants are preferred.
- Preferred alkoxylated surfactants can be selected from the classes of the nonionic condensates of alkyl phenols, nonionic ethoxylated alcohols, nonionic ethoxylated/propoxylated fatty alcohols, nonionic ethoxylate/propoxylate condensates with propylene glycol, and the nonionic ethoxylate condensation products with propylene oxide/ethylene diamine adducts.
- Nonionic surfactant can be present in the detergent compositions. It may be preferred that the level of ethoxylated nonionic surfactants in the intimate mixture are below 10% by weight of the mixture, preferably even 5% by weight.
- the condensation products of aliphatic alcohols with from I to 25 moles of alkylene oxide, particularly ethylene oxide and/or propylene oxide, are suitable for use herein.
- the alkyl chain of the aliphatic alcohol can either be straight or branched, primary or secondary, and generally contains from 6 to 22 carbon atoms.
- Particularly preferred are the condensation products of alcohols having an alkyl group containing from 8 to 20 carbon atoms with from 2 to 10 moles of ethylene oxide per mole of alcohol.
- Suitable cationic surfactants to be used herein include the quaternary ammonium surfactants.
- the quaternary ammonium surfactant is a mono C 6 -C 16 , preferably C 8 -C 14 N-alkyl or alkenyl ammonium surfactants wherein the remaining N positions are substituted by methyl, hydroxyethyl or hydroxypropyl groups.
- Preferred are also the mono-alkoxylated and bis-alkoxylated amine surfactants.
- cationic ester surfactants Another suitable group of cationic surfactants which can be used in the detergent compositions or components thereof herein are cationic ester surfactants.
- the cationic ester surfactant is a, preferably water dispersible, compound having surfactant properties comprising at least one ester (i.e. -COO-) linkage and at least one cationically charged group.
- Suitable cationic ester surfactants including choline ester surfactants, have for example been disclosed in US Patents No.s 4228042, 4239660 and 4260529.
- composition herein can contain a transition metal containing bleach catalyst.
- bleach catalysts include the manganese-based complexes disclosed in U.S. Pat. 5,246,621 and U.S. Pat. 5,244,594. Preferred examples of these catalysts include Mn IV 2 (u-O) 3 (1,4,7-trimethyl-1,4,7-triazacyclononane) 2 -(PF 6 ) 2 , Mn III 2 (u-O) 1 (u-OAc) 2 (1,4,7-trimethyl-1,4,7-triazacyclononane) 2 -(ClO 4 ) 2 , Mn IV 4 (u-O) 6 (1,4,7-triazacyclononane) 4 -(ClO 4 ) 2 , Mn III Mn IV 4 (u-O) 1 (u-OAc) 2 -(1,4,7-trimethyl-1,4,7-triazacyclononane) 2 -(ClO 4 ) 3 , and mixtures thereof.
- ligands suitable for use herein include 1,5,9-trimethyl-1,5,9-triazacyclododecane, 2-methyl-1,4,7-triazacyclononane, 2-methyl-1,4,7-triazacyclononane, 1,2,4,7-tetramethyl-1,4,7-triazacyclononane, and mixtures thereof.
- the bleach catalysts useful herein may also be selected as appropriate for the present invention.
- suitable bleach catalysts see U.S. Pat. 4,246,612 and U.S. Pat. 5,227,084. See also U.S. Pat. 5,194,416 which teaches mononuclear manganese (IV) complexes such as Mn(1,4,7-trimethyl-1,4,7-triazacyclononane)(OCH 3 ) 3 -(PF 6 ).
- Still another type of bleach catalyst is a water-soluble complex of manganese (III), and/or (IV) with a ligand which is a non-carboxylate polyhydroxy compound having at least three consecutive C-OH groups.
- Preferred ligands include sorbitol, iditol, dulsitol, mannitol, xylithol, arabitol, adonitol, meso- erythritol, meso-inositol, lactose, and mixtures thereof.
- U.S. Pat. 5,114,611 teaches a bleach catalyst comprising a complex of transition metals, including Mn, Co, Fe, or Cu, with an non-(macro)-cyclic ligand.
- B is a bridging group selected from O, S.
- R 5 , R 6 , and R 7 can each be H, alkyl, or aryl groups, including substituted or unsubstituted groups.
- Preferred ligands include pyridine, pyridazine, pyrimidine, pyrazine, imidazole, pyrazole, and triazole rings.
- said rings may be substituted with substituents such as alkyl, aryl, alkoxy, halide, and nitro.
- substituents such as alkyl, aryl, alkoxy, halide, and nitro.
- Particularly preferred is the ligand 2,2'-bispyridylamine.
- Preferred bleach catalysts include Co, Cu, Mn, Fe,-bispyridylmethane and -bispyridylamine complexes.
- Highly preferred catalysts include Co(2,2'-bispyridylamine)Cl 2 , Di(isothiocyanato)bispyridylamine-cobalt (II), trisdipyridylamine-cobalt(II) perchlorate, Co(2,2-bispyridylamine) 2 O 2 ClO 4 , Bis-(2,2'-bispyridylamine) copper(II) perchlorate, tris(di-2-pyridylamine) iron(II) perchlorate, and mixtures thereof.
- binuclear Mn complexed with tetra-N-dentate and bi-N-dentate ligands including N 4 Mn III (u-O) 2 Mn IV N 4 ) + and [Bipy 2 Mn III (u-O) 2 Mn IV bipy 2 ]-(ClO 4 ) 3 .
- bleach catalysts are described, for example, in European patent application, publication no. 408,131 (cobalt complex catalysts), European patent applications, publication nos. 384,503, and 306,089 (metallo-porphyrin catalysts), U.S. 4,728,455 (manganese/multidentate ligand catalyst), U.S. 4,711,748 and European patent application, publication no. 224,952, (absorbed manganese on aluminosilicate catalyst), U.S. 4,601,845 (aluminosilicate support with manganese and zinc or magnesium salt), U.S. 4,626,373 (manganese/ligand catalyst), U.S. 4,119,557 (ferric complex catalyst), German Pat.
- the detergent compositions of the invention preferably comprise additional water-soluble and/ or water insoluble builder material.
- Preferred are silicates, aluminosilicates, crystalline layered silicates and phosphate salts.
- compositions may for example comprise phosphate-containing builder material, preferably comprises tetrasodium pyrophosphate or even more preferably anhydrous sodium tripolyphosphate, present at a level of from 0.5% to 60%, more preferably from 5% to 50%, more preferably from 8% to 40.
- phosphate-containing builder material preferably comprises tetrasodium pyrophosphate or even more preferably anhydrous sodium tripolyphosphate, present at a level of from 0.5% to 60%, more preferably from 5% to 50%, more preferably from 8% to 40.
- Suitable water-soluble builder compounds include the water soluble (poly)carboxylate salts and borates.
- the carboxylate or polycarboxylate builder can be momomeric or oligomeric in type although monomeric polycarboxylates are generally preferred for reasons of cost and performance.
- the(poly)carboxylates and/ or polymeric or oligomeric (poly)carboxylates are present at levels of less than 5%, preferably less than 3% or even less than 2% or even 0% by weight of the compositions.
- Examples of largely water insoluble builders include the sodium aluminosilicates.
- Suitable aluminosilicate zeolites have the unit cell formula Na z [(AlO 2 ) z (SiO 2 ) y ], xH 2 O wherein z and y are at least 6; the molar ratio of z to y is from 1.0 to 0.5 and x is at least 5, preferably from 7.5 to 276, more preferably from 10 to 264.
- the aluminosilicate material are in hydrated form and are preferably crystalline, containing from 10% to 28%, more preferably from 18% to 22% water in bound form.
- the aluminosilicate zeolites can be naturally occurring materials, but are preferably synthetically derived. Synthetic crystalline aluminosilicate ion exchange materials are available under the designations Zeolite A, Zeolite B, Zeolite P, Zeolite X, Zeolite HS and mixtures thereof. Zeolite A has the formula: Na 12 [AlO 2 ) 12 (SiO 2 ) 12 ]. xH 2 O wherein x is from 20 to 30, especially 27. Zeolite X has the formula Na 86 [(AlO 2 ) 86 (SiO 2 ) 106 ]. 276 H 2 O.
- Zeolite MAP is zeolite MAP builder.
- Zeolite MAP is described in EP 384070A (Unilever). It is defined as an alkali metal aluminosilicate of the zeolite P type having a silicon to aluminium ratio not greater than 1.33, preferably within the range from 0.9 to 1.33 and more preferably within the range of from 0.9 to 1.2.
- compositions herein may also comprise additional silicate material, including amorphous silicate material, meta-silicates, preferably at least crystalline layered silicate material such as sold under the trade name SKS-6.
- the silicate material is preferably present at a level of less than 20% by weight of the compositions, preferably less than 15% by weight or even less than 10% by weight.
- Heavy metal ion sequestrant are also useful additional ingredients herein.
- heavy metal ion sequestrant it is meant herein components which act to sequester (chelate) heavy metal ions. These components may also have calcium and magnesium chelation capacity, but preferentially they show selectivity to binding heavy metal ions such as iron, manganese and copper.
- Heavy metal ion sequestrants are generally present at a level of from 0.005% to 10%, preferably from 0.1% to 5%, more preferably from 0.25% to 7.5% and most preferably from 0.3% to 2% by weight of the compositions.
- Suitable heavy metal ion sequestrants for use herein include organic phosphonates, such as the amino alkylene poly (alkylene phosphonates), alkali metal ethane 1-hydroxy disphosphonates and nitrilo trimethylene phosphonates.
- Preferred among the above species are diethylene triamine penta (methylene phosphonate), ethylene diamine tri (methylene phosphonate) hexamethylene diamine tetra (methylene phosphonate) and hydroxy-ethylene 1,1 diphosphonate, 1,1 hydroxyethane diphosphonic acid and 1,1 hydroxyethane dimethylene phosphonic acid.
- Suitable heavy metal ion sequestrant for use herein include nitrilotriacetic acid and polyaminocarboxylic acids such as ethylenediaminotetracetic acid, ethylenediamine disuccinic acid, ethylenediamine diglutaric acid, 2-hydroxypropylenediamine disuccinic acid or any salts thereof.
- Suitable heavy metal ion sequestrants for use herein are iminodiacetic acid derivatives such as 2-hydroxyethyl diacetic acid or glyceryl imino diacetic acid, described in EP-A-317,542 and EP-A-399,133.
- iminodiacetic acid-N-2-hydroxypropyl sulfonic acid and aspartic acid N-carboxymethyl N-2-hydroxypropyl-3-sulfonic acid sequestrants described in EP-A-516,102 are also suitable herein.
- EP-A-476,257 describes suitable amino based sequestrants.
- EP-A-510,331 describes suitable sequestrants derived from collagen, keratin or casein.
- EP-A-528,859 describes a suitable alkyl iminodiacetic acid sequestrant. Dipicolinic acid and 2-phosphonobutane-1,2,4-tricarboxylic acid are alos suitable.
- Glycinamide-N,N'-disuccinic acid (GADS), ethylenediamine-N-N'-diglutaric acid (EDDG) and 2-hydroxypropylenediamine-N-N'-disuccinic acid (HPDDS) are also suitable.
- diethylenetriamine pentacetic acid ethylenediamine-N,N'-disuccinic acid (EDDS) and 1,1 hydroxyethane diphosphonic acid or the alkali metal, alkaline earth metal, ammonium, or substituted ammonium salts thereof, or mixtures thereof.
- EDDS ethylenediamine-N,N'-disuccinic acid
- 1,1 hydroxyethane diphosphonic acid or the alkali metal, alkaline earth metal, ammonium, or substituted ammonium salts thereof, or mixtures thereof.
- Another preferred ingredient useful herein is one or more additional enzymes.
- Preferred additional enzymatic materials include the commercially available lipases, cutinases, amylases, neutral and alkaline proteases, cellulases, endolases, esterases, pectinases, lactases and peroxidases conventionally incorporated into detergent compositions. Suitable enzymes are discussed in US Patents 3,519,570 and 3,533,139.
- protease enzymes include those sold under the tradenames Alcalase, Savinase, Primase, Durazym, and Esperase by Novo Industries A/S (Denmark), those sold under the tradename Maxatase, Maxacal and Maxapem by Gist-Brocades, those sold by Genencor International, and those sold under the tradename Opticlean and Optimase by Solvay Enzymes.
- Protease enzyme may be incorporated into the compositions in accordance with the invention at a level of from 0.0001 % to 4% active enzyme by weight of the composition.
- Additional organic polymeric compounds are preferred additional components of the compositions herein or the agglomerates herein, where they may act such as to bind the agglomerate components together.
- organic polymeric compound it is meant herein essentially any polymeric organic compound commonly used as binder, dispersants, and anti-redeposition and soil suspension agents in detergent compositions, including any of the high molecular weight organic polymeric compounds described as clay flocculating agents herein, including quaternised ethoxylated (poly) amine clay-soil removal/ anti-redeposition agent in accord with the invention.
- Organic polymeric compound is typically incorporated in the detergent compositions of the invention at a level of from 0.01% to 30%, preferably from 0.1% to 15%, most preferably from 0.5% to 10% by weight of the compositions.
- Organic polymeric compounds suitable for incorporation in the detergent compositions herein include cellulose derivatives such as methylcellulose, sodium carboxymethylcellulose, hydroxypropylmethylcellulose and hydroxyethylcellulose. Highly preferred is also - or amide-modified carboxymethyl cellulose and - or amide-modified celluloses, or derivatives thereof
- organic polymeric compounds are the polyethylene glycols, particularly those of molecular weight 1000-10000, more particularly 2000 to 8000 and most preferably about 4000.
- the detergent compositions of the invention when formulated for use in machine washing compositions, may comprise a suds suppressing system present at a level of from 0.01% to 15%, preferably from 0.02% to 10%, most preferably from 0.05% to 3% by weight of the composition.
- Suitable suds suppressing systems for use herein may comprise essentially any known antifoam compound, including, for example silicone antifoam compounds and 2-alkyl alcanol antifoam compounds.
- composition herein may be a detergent which provides softening through the wash.
- Preferred may be that the composition comprises a clay softening agent, and preferably also a flocculating agent.
- Suitable smectite clays include those selected from the classes of the montmorillonites, hectorites, volchonskoites, nontronites, saponites and sauconites, particularly those having an alkali or alkaline earth metal ion within the crystal lattice structure.
- Sodium or calcium montmorillonite are particularly preferred.
- Preferred are bentonite clays.
- Preferred as clay flocculating agents herein are organic polymeric materials having an average weight of from 100,000 to 10,000,000, preferably from 150,000 to 5,000,000, more preferably from 200,000 to 2,000,000.
- Suitable organic polymeric materials comprise homopolymers or copolymers containing monomeric units selected from alkylene oxide, particularly ethylene oxide, acrylamide, acrylic acid, vinyl alcohol, vinyl pyrrolidone, and ethylene imine. Homopolymers of, on particular, ethylene oxide, but also acrylamide and acrylic acid are preferred.
- EP-A-299,575 and EP-A-313,146 in the name of the Procter and Gamble Company describe preferred organic polymeric clay flocculating agents for use herein.
- compositions of the invention include perfumes, such as the encapsultyed perfumes described above, brightners, speckles, including colours or dyes, filler salts, with sodium sulfate being a preferred filler salt.
- neutralizing agents e.g., less than about 20% by weight
- additional buffering agents e.g., phase regulants, hydrotropes, enzyme stabilizing agents, polyacids, suds regulants, opacifiers, anti-oxidants, bactericides and dyes, such as those described in US Patent 4,285,841 to Barrat et al., issued August 25, 1981 can be present.
- LAS Sodium linear C11-13 alkyl benzene sulfonate
- TAS Sodium tallow alkyl sulfate
- CxyAS Sodium C1x - C1y alkyl sulfate
- C46SAS Sodium C14-C16 secondary (2,3) alkyl sulfate
- CxyEzS Sodium C1x-C1y alkyl sulfate condensed with z moles of ethylene oxide
- CxyEz C 1 x-C 1 y predominantly linear primary alcohol condensed with an average of z moles of ethylene oxide
- Soap Sodium linear alkyl carboxylate derived from an 80/20 mixture of tallow and coconut
- Amylase Amylolytic enzyme, having 1.6% by weight of active enzyme, sold by NOVO Industries A/S under the tradename Termamyl 120T
- Lipase Lipolytic enzyme, having 2.0% by weight of active enzyme, sold by NOVO Industries A/S under the tradename Lipolase or lipolytic enzyme, having 2.0% by weight of active enzyme, sold by NOVO Industries A/S under the tradename
- Lipolase Ultra PB4 Particles containing sodium perborate tetrahydrate of nominal formula NaBO2.3H2 O, the particles having a weight average particle size of 950 microns, 85% particles having a particle size of from 850 microns to 950 microns PB1 : Particle containing anhydrous sodium perborate bleach of nominal formula NaBO2.H 202, the particles having a weight average particle size of 800 microns, 85% particles having a particle size of from 750 microns to 950 microns
- Percarbonate Particle containing sodium percarbonate of nominal formula 2Na2
- HEDP 1,1-hydroxyethane diphosphonic acid
- PEGx Polyethylene glycol, with a molecular weight of x (typically 4,000)
- PEO Polyethylene oxide, with an average molecular weight of 50,000
- TEPAE Tetraethylenepentaamine ethoxylate
- PVI Polyvinyl imidosole, with an average molecular weight of 20,000
- PVP Polyvinylpyrolidone polymer, with an average molecular weight of 60,000
- PVNO Polyvinylpyridine N-oxide polymer, with an average molecular weight of 50,000
- PVPVI Copolymer of polyvinylpyrolidone and vinylimidazole, with an average molecular weight of 20,000
- compositions are in accordance with the invention and have a pH in the wash, as measured by the method defined herein, of around 9.4 -9.85:
- a B C D E F G Spray-dried Granules - LAS 8.0 10.0 15.0 5.0 - TAS - 1.0 - 0.5 - MBAS 4.0 - 5.0 - C 45 AS - - 1.0 - C 45 AE 3 S - - 1.0 1.0 - QAS 1.0 1.0 - DTPA, HEDP and/or EDDS 0.3 0.3 0.5 0.3 - MgSO 4 0.5 0.5 0.1 0.1 - Sodium carbonate 3.0 7.0 5.0 4.0 - Sodium sulphate - - - - - Sodium silicate 1.6R - - - 0.3 - STPP - - - - - Zeolite A 16.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Emergency Medicine (AREA)
- Detergent Compositions (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Description
- This invention provides specific sanitising compositions and methods for improved sanitisation of fabrics.
- Hydrogen peroxide bleach containing dishwashing compositions are described in DE 4232170 and DE19547430. In the first of these references, weakly alkaline machine dish-washing compositions are described comprising sodium citrate, alkali metal hydrogen carbonate, bleaching agents, bleach activators and enzymes. In the second of these, in addition, part of the sodium citrate is replaced with a water soluble natural substance selected from mono and/or disaccharides, sugar acids and protein hydrolysates.
- Research has shown that hydrogen peroxide bleach in laundry detergents also has a disinfecting action and, more recently, detergent manufacturers have become interested in the disinfecting properties of these detergents. Peroxyacid bleach precursors are also known to provide antimicrobial action.
- However, not all bleach systems which have sufficient disinfecting properties are suitable for detergents: bleach is not always compatible with other detergent ingredients, such as enzymes, or moreover, the levels of bleach required to obtain disinfecting are too high and may damage the fabrics.
- The inventors found that peroxyacid bleach precursors can be the preferred bleach species to deliver sanitisation of fabrics during the wash. They found however that a problem with these bleach precursors is that they form the peroxyacid bleach at a high, alkaline pH, but that at such a high, alkaline pH the peroxyacid, which is found to be the active species which gives the sanitisation benefits, dissociates to the inactive form, resulting thus in reduced sanitisation.
- Also, a preferred bleaching ingredient in detergents is a percarbonate salt. The percarbonate makes the solution very alkaline, which is as set out above not desirable when aiming to keep high levels of the active species, i.e. the peroxyacid itself, and thus the antimicrobial performance thereof. Also many builders provide an alkaline pH in the wash, which is not desirable when seeking an efficient sanitisation.
- Moreover, many detergent ingredients also require high, alkaline pH conditions in the wash, which are not compatible with the preferred pH of the wash required to have a high level of peroxyacid.
- Thus it is difficult to formulate detergents in such a way that preferably small amounts peroxyacid bleach precursors provide efficient sanitisation whilst at the same time excellent cleaning is achieved.
- The inventors have now found a way to provide efficient antimicrobial performance and/or sanitisation whilst a good cleaning of both bleachable and non-bleachable stains is achieved. They found that when the pH of the detergent in the wash water is controlled, namely from 8.8 to 9.9, preferably from 9.0 or even 9.2 to 9.85 or even to 9.8, in particular achieved by using specific levels of acid and alkaline ingredients and optionally buffers, excellent sanitisation and cleaning is obtained, whilst only limited amounts of precursor are required, making the compositions more storage stable and safer to use. They found that these detergent compositions can then even contain percarbonate bleach.
- The specific compositions of the invention are preferably used to effectively provide sanitisation of fabrics or, effectively inactivate micro-organisms, in particular bacteria which are difficult to inactivate, such as in particular E. Hirae, but also S. Aureus, E Coli and Ps Aeruginosa.
- The invention relates to a laundry detergent composition comprising, at least 15% by weight of a carbonate source and a bleach system which contains at least 2.5% by weight of a peroxyacid bleach precursor and a hydrogen peroxide source, which may include a salt of a percarbonate and may thus be part of the carbonate source, and at least 7% by weight of an acid, preferably an organic acid, more preferably including a organic carboxylic acid, characterised in that a 1% by weight mixture of the composition in demineralised water at a temperature of 20°C obtained after 10 minutes stirring at a speed of 200rpm, provides a pH from 8.8 to 9.9 and which further comprises an anionic surfactant.
- The invention also relates to a method of providing sanitisation during the wash by use of a composition of the invention and a method for inactivate E. Hirae, S. Aureus, E. Coli and Ps Aeruginosa.
- The inventors have also found that, when the laundry compositions of the invention are for use in automatic laundry methods, improved antimicrobial performance or sanitisation of the fabrics is achieved when the composition is delivered directly into the drum of the washing machine, for example by use of a dispensing device. One embodiment of the invention thus relates to a washing method for washing fabrics whereby the composition of the invention is introduced into the drum of a washing machine by placing a dispensing device comprising the composition in the drum prior to the introduction of wash water.
- The composition of the invention is preferably such that a 1% mixture of the composition in demineralised water, at a temperature of 20°C. obtained after 10 minutes stirring at a speed of200rpm, provides a pH from 9.0 to 9.85 or even from 9.2 to 9.85 or even to 9.8, or even from 9.4 to 9.8. The pH is measured after stirring the mixture, for 10 minutes at a constant temperature, by any conventional pH measurement method.
- With the term '1% mixture' is meant any mixture, dispersion or solution of 1 weight unit of the composition in 99 weight units of demineralised water, and it is thus to be understood that if the composition is not dissolved fully after 10 minutes, the pH is measured for such a mixture or dispersion.
- When used herein, 'reduction of the activity of the micro-organisms' includes a reduction of the activity as defined in the CEN method prEN1276, 1993, (Comité Européen de Normalisation) and CEN method prEN 1650. The reduction therein is preferably represented by a reduction of the specific micro-organism or bacteria by at least 105, more preferably at least 2x 105, or even 3x 105.
- The reduction of the activity of the micro-organisms when used herein includes the reduction of any of the processes conducted by the micro-organism, including secretion of products but preferably the growth of the micro-organisms.
- The invention also provides a method for reduction of the activity of micro-organisms, or preferably specific bacteria, comprising the steps of contacting the micro-organism with an aqueous liquor comprising the composition of the invention such that the peroxyacid provided by the precursor is present at a level of at least 100 ppm in the liquor, per 106 micro-organisms.
- Preferably, the composition is such that at least the activity of E. Hirae, but more preferably also of S. Aureus, E Coli and Ps Aeruginosa, is reduced by at least 105, preferably at least 2 x 105 or even 3 x 105, as measured by the above CEN method.
- In particular, the activity reduction is done during a laundering process. Hereby, the composition of the invention can be contacted with the specific micro-organisms in solid form, but preferably the bleaching agent is present in a liquor in contact with the micro-organisms.
- The specific micro-organisms, preferably the specific bacteria, can be present on the surfaces or fabrics to be cleaned and sanitised, for example on the fabrics to be cleaned in a laundry process. The bacteria can also be present in the liquor used for the cleaning or sanitisation process or in the equipment used in the process, such as the washing machine or dispensing device.
- The composition of the invention is particularly useful in inactivating micro-organisms having cell walls comprising relatively high levels of peptidoglycan, in particular gram positive bacteria, which can be present in soils or stains on the fabrics or surfaces, in particular in body soils.
- The amount of composition required to obtain effective reduction of the activity of the specific micro-organisms depends on various factors, such as the amount of micro-organisms present, the conditions of the sanitisation or cleaning process, including the other compounds present and the temperature of the cleaning process.
- In the method of the invention, the liquor used preferably comprises at least 100 ppm of the peroxyacid per 106 micro-organisms, more preferably at least 200 ppm or even 250 ppm.
- The reduction of the activity of the specific micro-organisms, having a peptidoglycan-containing cell walls can be determined by the Petrocci and Clarke method, as described in JOAC 1981, but is preferably determined for the purpose of this invention by the CEN method prEN1276, 1993 for bacteria and CEN method prEN1650 for yeast.
- Such a CEN method involves, for example, the preparation of gram positive bacterial inocula conform the CEN method, pages 7 and further, preparation of a solution comprising the hydrophobic bleaching agent at a level of about 250ppm, conducting the test following the CEN method, incubation TSA plates for 24 hours at 36°C; and subsequently counting of the bacteria colonies on the plates.
- This is compared with the results of the reference and the reduction of bacteria growth is calculated, for the defined contacting time.
- The composition comprises at least 7% by weight of one or more acids, preferably an organic acid, preferably including at lest an organic carboxylic acid. The exact levels will depend on the other ingredients of the detergent composition and the alkalinity thereof, so that the level of the acid is adjusted to provide the required pH.
- Preferably the acid is present at a level of at least 8% by weight of the composition or even at least 9% or even at least 10% by weight of the composition, with as upper limit a level of preferably 30% or even 20% by weight of the composition.
- Suitable acids to be used herein include materials which not only help to provide the required pH of the formulation, but which also have a secondary function in the composition, such as acting as a chelating agent, builder and/or effervescence source. Useful inorganic acids include boric acid, bisulphite salts preferably sodium salts thereof.
- Preferably, the acid comprises at least an organic carboxylic acid. Such acids include mono- or polycarboxylic acids preferably citric acid, adipic acid, glutaric acid, 3 chetoglutaric acid, citramalic acid, tartaric acid, maleic acid, fumaric acid, malic acid, succinic acid, malonic acid but also polymeric or oligomeric polycarboxylic acids, such as acrylic acid polymers or maleic acid polymers, or copolymers of maleic acid and acrylic acid.
- Most preferably the organic carboxylic acid herein comprises at least 7% by weight of the composition of maleic acid, malic acid or citric acid, or mixtures thereof.
- It may be preferred that mixtures of organic carboxylic acids and inorganic acids are used, for example in ratio's of from 1:1 to 10:1 1 or even from 2:1 to 5:1.
- The acid may be present as a separate component or in solid compositions of the invention, the acid may be incorporated in a granule together with other detergent ingredients. In the latter case, it may be preferred that the acid is a particulate material whereof at least 75%, preferably at least 85% or even at least 90% or even at least 95% or even at least 99% by volume, has a particle size from 1 to 500 microns and more preferably from 1 to 350 microns and it may even be preferred that at least 65% or even at least 75% or even at least 85% has a particle size from 1.0 to 250 microns or even from 1.0 to 150 microns.
- These particle sizes can be determined by any method known in the art, in particular by laser light scattering or difraction technique, such as with Malvem 2600 or Sympatec Helos laser light scattering equipment (or difractometer).
- When the acid is present as a separate particle in the solid composition of the invention, the particle size distribution and mean is preferably similar to particle size distribution and mean of the other components of the composition, as described hereinafter.
- Salts of inorganic acids and/or salts or esters of organic carboxylic acids may also be present as additional components, in particular because it may be beneficial to buffer the composition at the required pH. For example, it may be preferred that the composition comprises a salt of citric acid, maleic acid, malic acid, glutaric acid or tartaric acid or borate salt, or mixtures thereof.
- The composition of the present invention comprises at least 15% by weight of a carbonate source. Preferred carbonate sources include carbonate, bicarbonate and percarbonate salts and preferably the carbonate source comprises a mixture of three salts.
- It is to be understood that the percarbonate salt when present, forms part of both the carbonate source and the hydrogen peroxide source as defined herein. Thus, in a preferred embodiment of the invention, the composition comprises a percarbonate salt to provide hydrogen peroxide and carbonate. For example, when the composition comprises at least 10% by weight of a percarbonate salt, as hydrogen peroxide source and carbonate source, the composition also contains at least 5% by weight of another carbonate source.
- The level of carbonate sources will depend on the nature of the detergent. Also, if percarbonate salt is used as hydrogen peroxide source, reduced levels of other carbonate sources may only be needed. Preferably, the composition comprises a mixture of carbonate sources, preferably including a carbonate salt, bicarbonate salt and percarbonate salt. Then, the composition comprises preferably at least 5% by weight of a carbonate salt, at least 2% by weight of a bicarbonate salt and at least 8% by weight of a percarbonate salt, more preferably at least 7% by weight of a carbonate salt , at least 4% by weight of a bicarbonate salt and at least 15% by weight of a percarbonate salt.
- Suitable carbonates sources to be used herein include carbonate and hydrogen carbonate or bicarbonate of earth alkali or alkali metals and sodium and potassium salts. The inventors found that it may be particularly useful to include a bicarbonate salt into the compositions, because it has been found that the bicarbonate salt provides a buffered pH at around the pH value needed to form the peroxyacids from the precursors, for example of TAED as described hereinafter.
- In a preferred embodiment of the invention, the composition comprises at least 1%, preferably at least 2% or even at least 4% by weight of the composition of an alkali or earth alkali salt of bicarbonate, preferably sodium bicarbonate. This salt may be present up to any level, preferably up to 20% by weight of the composition, more preferably up to 15% or even up to 10% by weight of the composition.
- The carbonate, bicarbonate and percarbonate salts herein my be present as separate particulate components or may be incorporated in detergent granules together with other detergent ingredients.
- The carbonate and bicarbonate material, when present in other detergent granules, may preferably have a volume median particle size from 1 to 500 microns, whereby preferably at least 60%, preferably at least 70% or even at least 80% or even at least 90% by volume, has a particle size of from 0.5 to 1180 microns. More preferably, the carbonate or bicarbonate salts has a volume median particle size of 10 to 375, whereby preferably at least 60 %, or even at least 70% or even at least 80% or even at least 90% by volume, has a particle size of from 1 to 850 microns; or even preferably a volume median particle size from 10 to 250 microns, whereby preferably at least 60 %, preferably at least 70% or even at least 80% or even at least 90% by volume, has a particle size of from 5 to 425 microns.
- It may be preferred that the required particle size of the carbonate and/or bicarbonate salt is obtained by grinding a larger particle size material, optionally followed by selecting the material with the required particle size by any suitable method.
- Preferably, the carbonate source is incorporated in the composition in such a manner that it is capable to react with the acid to provide effervescing upon contact with water.
- The compositions of the invention comprise a hydrogen peroxide source. Preferably, this is a persalt such a salts of percarbonate, perborate, perphosphate, peroxymonopersulfate and persilicate salts. The inorganic perhydrate salts are normally the alkali metal salts. The alkali metal salt of percarbonate, perborate, or mixtures thereof, are the preferred inorganic perhydrate salts for use herein. Preferred perborate is sodium perborate in the form of the monohydrate or tetrahydrate, respectively of nominal formula NaBO2H2O2 and NaBO2H2O2.3H2O. Other suitable oxygen bleaches include persulphates, particularly potassium persulphate K2S2O8 and sodium persulphate Na2S2O8.
- Highly preferred is an alkali metal salt of percarbonate, preferably sodium percarbonate, which then forms also part of the carbonate source, described above.
- Typically, the compositions in the present invention comprise from 5% to 50% by weight of the total composition of a hydrogen peroxide source, preferably from 10% to 45% and more preferably from 15% to 35% by weight or even form 17% or even 19% to 30% or even to 25% by weight of the composition.
- The persalts may be coated by any coating material, preferably comprising a sulphate salt, such as magnesium sulphate or preferably sodium sulphate, carbonate or bicarbonate salt or mixtures thereof.
- Preferred persalts and methods for making them are described in for example
- The granular compositions herein comprise a peroxyacid bleach precursor. Preferably, the peroxyacid bleach precursor is present at a level of from 3% to 10% or even 3.5% to 8% or even 4% to 6% by weight of the composition.
- Peroxyacid bleach precursors are compounds which react with hydrogen peroxide in a perhydrolysis reaction to produce a peroxyacid. Generally peroxyacid bleach precursors may be represented as
- Suitable peroxyacid bleach precursor compounds typically contain one or more N- or O-acyl groups, which precursors can be selected from a wide range of classes. Suitable classes include anhydrides, esters, imides, lactams and acylated derivatives of imidazoles and oximes. Examples of useful materials within these classes are disclosed in GB-A-1586789. Suitable esters are disclosed in GB-A-836988, 864798, 1147871, 2143231 and EP-A-0170386.
- The leaving group, hereinafter L group, must be sufficiently reactive for the perhydrolysis reaction to occur within the optimum time frame (e.g., a wash cycle). However, if L is too reactive, this activator will be difficult to stabilize for use in a bleaching composition.
- Preferred L groups are selected from the group consisting of:
- The preferred solubilizing groups are -SO3 -M+, -CO2 -M+, -SO4 -M+, -N+(R3)4X- and O<--N(R3)3 and most preferably -SO3 -M+ and -CO2 -M+ wherein R3 is an alkyl chain containing from 1 to 4 carbon atoms, M is a cation which provides solubility to the bleach activator and X is an anion which provides solubility to the bleach activator. Preferably, M is an alkali metal, ammonium or substituted ammonium cation, with sodium and potassium being most preferred, and X is a halide, hydroxide, methylsulfate or acetate anion.
- Preferred examples of such compounds are tetracetyl ethylene diamine, (TAED), sodium 3, 5, 5-trimethyl hexanoyloxybenzene sulphonate, nonylamide of peroxyadipic acid and n-nonanoyloxybenzenesulphonate (NOBS), -decanoyloyloxybenzenesulphonate, dodecanoyloxybenzenesulphonate and acetyl triethyl citrate (ATC), Phenolsulphonate Ester of N-nonanoyl-6-aminocaproic acid, aliphatic diacyl peroxide (DAP) having the general formula R-C(O)-O-O-(O)C-R1, wherein R and R1 can be the same or different and are linear or branched aliphatic groups having from 6 to 20 carbon atoms.
- Also particularly preferred are N-acyl caprolactam selected from the group consisting of substituted or unsubstituted benzoyl caprolactam, octanyl caprolactam, nonanoyl caprolactam, hexanoyl caprolactam, decanoyl caprolactam, undecenoyl caprolactam, formyl caprolactam, acetyl caprolactam, propanoyl caprolactam, butanoyl caprolactam pentanoyl caprolactam.
- Examples of other uitable compounds are disclosed in British Patent GB 1 586 769 , GB 2 143 231, US 4 818 425 and US 4 259 201.
- Amide substituted alkyl peroxyacid precursor compounds are suitable herein, including those of the following general formulae:
- A highly preferred bleach precursor herein is TAED, NACA-OBS, DOBS and/ or NOBS. The compositions herein may comprise mixtures of said bleach activators, because it has been found that this may result in improved sanitisation. Highly preferred is a mixture of TAED and a precursor selected from NOBS, DOBS or NACA-OBS. It has been found that further improved sanitisation is achieved when such mixed precursor systems are used, in particular when the ratio of TAED to NOBS, DOBS or NACA-OBS is from 1:2 to 2:1.
- The precursor may be in the form of a separate detergent granule, which may be coated, or in the form of a detergent granule comprising also other detergent actives. It is preferably present in the form of a particulate component having a similar particle size distribution and mean particle size as the other detergent particles.
- The inventors also found that it can be beneficial when the precursor and hydrogen peroxide source are present intimately mixed with one another, for example in a granule. This mixture or granule is preferably being substantially free of organic acids.
- It should be understood that for the purpose of the invention, the peroxy acid formed of the precursor is not part of the acid component of the composition of the invention.
- Machine laundry methods herein typically comprise treating soiled laundry with an aqueous wash solution in a washing machine having dissolved or dispensed therein an effective amount of a detergent in accord with the invention. In particular laundry washing methods are envisaged herein. For example, an effective amount of the laundry detergent composition for laundry washing is normally meant from 10g to 300g of product dissolved or dispersed in a wash solution of volume from 5 to 65 litres, as are typical product dosages and wash solution volumes commonly employed in conventional machine laundry methods. Dosage is dependent upon the particular conditions such as water hardness and degree of soiling of the soiled laundry.
- The reduction of the activity of the micro-organisms present in the wash and on the soiled fabrics preferably takes place during a washing method, preferably an automatic or hand laundry, at low temperatures up to 60°C or even up to 45°C or even around 30°C.
- The detergent composition may be dispensed for example, from the drawer dispenser of a washing machine or may be sprinkled over the soiled laundry placed in the machine.
- In one highly preferred use aspect a dispensing device is employed in the washing method, to introduce the composition of the invention directly in the drum of the wash. It has been found that improved sanitisation is then achieved.
- Any dispensing device can be used, including preferred dispensing devices for use with the composition of the invention have been described in the following patents; GB-B-2, 157, 717, GB-B-2, 157, 718, EP-A-0201376, EP-A-0288345 and EP-A-0288346, WO94/11562.
- The compositions of the present invention can be prepared with different bulk densities, preferably being from 300 to 1200 g/l, preferably from 500 to 1100 g/l or even 550g/l to 900g/l. These compositions are preferably solid compositions, preferably solid laundry detergents, which can be made by a variety of methods well known in the art, including dry-mixing, spray drying, extrusion, roller compaction, agglomeration and combinations thereof. The solid compositions may have any form, preferably granular or in the form of a tablet or bar.
- It may be preferred that the composition is made by mixing all or part of the granules, including those made by agglomeration or spray-drying, and subsequently adding a binder and mixing or agglomerating the granules and binder to form the, preferably agglomerated detergent granules. These may be of the required particle size or they may be sieved to obtain particles of the required size.
- In a highly preferred embodiment, the acid, or part thereof, and the carbonate source or part thereof, are present in an intimate mixture with one another, preferably in a granule, which means for the purpose of the invention that the acid and carbonate source are preferably homogeneously mixed. For optimum effervescence in aqueous medium the weight ratio of acid to carbonate source in the intimate mixture or the effervescent granule is preferably from 0.1 to 10, preferably from 0.5 to 2.5 and more preferably from 1 to 2.
- The acid is preferably present in such a granule at a level of from 5% to 85% by weight of the total granule, preferably from 10% to 75%, more preferably from 15% to 60% and most preferably from 10% to 50%.
- The carbonate source is preferably present in such a granule at a level of from 5% to 90% by weight of the total, preferably from 10% to 80%, more preferably from 20% to 75% and most preferably from 30% to 65%.
- The granule is preferably substantially free of water, i.e. no water has been added or present other than the moisture of the raw materials themselves. Typically, the level of water is below 5% by weight of the total intimate mixture or granule, preferably below 3% and more preferably below 1.5%.
- It may be preferred that a desiccant is present in the intimate mixture or the granule, such as overdried inorganic and organic salts, anhydrous salts, in particular overdried silicates and aluminosilicates, anhydrous silicates and/or sulphate salts.
- The granules are preferably obtainable by a process comprising a granulation step, preferably comprising the step of dry-powder compaction or pressure agglomeration. While all binding mechanisms can occur in pressure agglomeration, adhesion forces between the solid particles, i.e., between the acid, carbonate source and optionally the binder if present, play an especially important role. This is because pressure agglomeration, especially high pressure agglomeration, is an essentially dry process that forms new entities (i.e., dry effervescent granules) from solid particles (i.e., the acid, bicarbonate, carbonate source and optionally the binder) by applying external forces to densify a more or less defined bulk mass or volume and create binding mechanisms between the solid particles providing strength to the new entity, i.e. the high external force applied brings the solid particles closely together.
- The effervescent granules may optionally comprise a binder or a mixture thereof. Typically, the granules comprise up to 50 % by weight of the total granule of a binder or a mixture thereof, preferably up to 35% and more preferably up to 20%. Suitable binders to use herein are those known to those skilled in the art and include anionic surfactants like C6-C20 alkyl or alkylaryl sulphonates or sulphates, preferably C8-C20 alkylbenzene sulphonates, cellulose derivatives such as carboxymethylcellulose and homo- or copolymeric polycarboxylic acid or their salts, nonionic surfactants, preferably C10-C20 alcohol ethoxylates containing from 5-100 moles of ethylene oxide per mole of alcohol and more preferably the C15-C20 primary alcohol ethoxylates containing from 20-100 moles of ethylene oxide per mole of alcohol. Of these tallow alcohol ethoxylated with 25 moles of ethylene oxide per mole of alcohol (TAE25) or 50 moles of ethylene oxide per mole of alcohol (TAE50) are preferred. Other preferred binders include the polymeric materials like polyvinylpyrrolidones with an average molecular weight of from 12 000 to 700 000 and polyethylene glycols with an average weight of from 600 to 10 000. Copolymers of maleic anhydride with ethylene, methylvinyl ether, methacrylic acid or acrylic acid are other examples of polymeric binders. Others binders further include C 10-C20 mono and diglycerol ethers as well as C10-C20 fatty acids.
- The effervescent granules may have any particle size, the preferred particle size depending on the application and the component of the granule.
- The inventors have found that it can be beneficial for the sanitisation and cleaning performance that the acid herein is separated from the precursor and preferably any other bleach components present including any hydrogen peroxide source, including percarbonate salt, if present. Thus, the effervescing granules described above, preferably comprise no percarbonate salt as carbonate source, but preferably comprise carbonate salts and/ or bicarbonate salts.
- In a preferred embodiment, the composition preferably comprises granules whereof at least 60%, more preferably at least 80% by weight have an average particle size, by weight, of from 400 microns to 1400 microns, preferably from 500 microns to 1100 microns or even 750 to 1000 microns. It may be preferred that the composition comprises less than 20% or even less than 10% or even less than 5% by weight of particulate components of a particle size of less than 300 microns, or even less than 425 microns or even less than 600 microns; it may also be preferred that the composition comprises less than 20% or even less than 10% or even less than 5% by weight of the composition, of particulate components of a particle size of more than 1700 microns, or even more than 1400 microns or even more than 1180 microns.
Thus, the compositions may comprise different granular components, which each have the above particle size requirements, and thus have a similar particle size distribution and mean particle size. - The inventors found that improved sanitisation is achieved when the composition contains perfumes or perfume components with a high fabric substantivity. It is believed that this is due to the perfume oils which remain on the surface of the washed fabric and which can thus provide sanitisation during the subsequent use of the fabric.
- Such preferred perfume granules of encapsulated perfume oils are described in co-pending European patent application EP-A-965326. Preferred levels of perfume granules will depend on the level of perfume oils therein, but generally the level is from about 0.05 to 8% or even 0.5% to 5% or even 0.8% to 3% by weight of the composition. Moreover, preferably the composition also comprises perfume components which are not in the form of encapsulated perfume oil granules, but preferably in the form of perfume components sprayed onto the detergent granules of the composition.
- The compositions herein preferably contain one or more additional detergent components selected from surfactants, bleach catalysts, additional builders, additional organic polymeric compounds, enzymes, suds suppressors, lime soap, dispersants, soil suspension and anti-redeposition agents soil releasing agents, brightners, photobleaching agents and additional corrosion inhibitors.
- It is to be understood that the levels of detergent ingredients are to be chosen such that the pH of 1% by weight of composition in demineralised water is as defined above. For example, it may be preferred that when crystalline layered silicates or amorphous silicates are used, the level thereof is up to about 12% by weight or even up to 10% by weight.
- The detergent compositions herein preferably contain one or more surfactants. The surfactant may comprise any surfactant known in the art selected from anionic, nonionic, cationic, ampholytic, amphoteric and zwitterionic surfactants and mixtures thereof.
- It should be understood that for the purpose of the invention the detergent composition may comprise surfactant which is not present in the intimate mixture with the crystalline layered silicate, but present in the other detergent components.
- The compositions in accord with the present invention comprise an anionic surfactant. Essentially any anionic surfactants useful for detersive purposes can be comprised in the detergent composition. These can include salts (including, for example, sodium, potassium, ammonium, and substituted ammonium salts such as mono-, di- and triethanolamine salts) of the anionic sulfate, sulfonate, carboxylate and sarcosinate surfactants. Anionic sulfate and sulfonate surfactants are preferred.
- Highly preferred are surfactants systems comprising a sulfonate and a sulfate surfactant, preferably a linear or branched alkyl benzene sulfonate and alkyl ethoxylsulfates, as described herein, preferably combined with a cationic surfactants as described herein.
- Highly preferred herein are anionic sulfonate surfactants. Particularly suitable for use herein include the salts of C5-C20 linear or branched alkylbenzene sulfonates, but also alkyl ester sulfonates, C6-C22 primary or secondary alkane sulfonates, C6-C24 olefin sulfonates, sulfonated polycarboxylic acids, alkyl glycerol sulfonates, fatty acyl glycerol sulfonates, fatty oleyl glycerol sulfonates, and any mixtures thereof. Most preferred are C9-C14 linear alkyl benzene sulfonates.
- Anionic sulfate surfactants suitable for use herein include the linear and branched primary and secondary alkyl sulfates, alkyl ethoxysulfates, fatty oleoyl glycerol sulfates, alkyl phenol ethylene oxide ether sulfates, the C5-C17 acyl-N-(C1-C4 alkyl) and -N-(C1-C2 hydroxyalkyl) glucamine sulfates, and sulfates of alkylpolysaccharides such as the sulfates of alkylpolyglucoside (the nonionic nonsulfated compounds being described herein).
- Alkyl sulfate surfactants are preferably selected from the linear and branched primary C10-C18 alkyl sulfates, more preferably the C11-C15 branched chain alkyl sulfates and the C12-C14 linear chain alkyl sulfates.
- Alkyl ethoxysulfate surfactants are preferably selected from the group consisting of the C10-C18 alkyl sulfates which have been ethoxylated with from 0.5 to 20 moles of ethylene oxide per molecule. More preferably, the alkyl ethoxysulfate surfactant is a C11-C18, most preferably C11-C15 alkyl sulfate which has been ethoxylated with from 0.5 to 7, preferably from 1 to 5, moles of ethylene oxide per molecule.
- A particularly preferred aspect of the invention employs mixtures of the preferred alkyl sulfate and/ or sulfonate and alkyl ethoxysulfate surfactants. Such mixtures have been disclosed in PCT Patent Application No. WO 93/18124.
- Highly preferred herein, in particular for providing an improved surfactant performance, are alkyl chain, mid-chain branched surfactant compounds of the formula
- The most preferred mid-chain branched surfactants compounds for use in the detergent compositions herein are mid-chain branched primary alkyl sulfonate and, even more preferably, sulfate surfactants. It should be understood that for the purpose of the invention, it may be preferred that the surfactant system comprises a mixture of two or more mid-chain branched primary alkyl sulfate or sulphonate surfactants.
-
- These surfactants have a linear primary alkyl sulfate chain backbone (i.e., the longest linear carbon chain which includes the sulfated carbon atom) which preferably comprises from 12 to 19 carbon atoms and their branched primary alkyl moieties comprise preferably a total of at least 14 and preferably no more than 20, carbon atoms. In the surfactant system comprising more than one of these sulfate surfactants, the average total number of carbon atoms for the branched primary alkyl moieties is preferably within the range of from greater than 14.5 to about 17.5. Thus, the surfactant system preferably comprises at least one branched primary alkyl sulfate surfactant compound having a longest linear carbon chain of not less than 12 carbon atoms or not more than 19 carbon atoms, and the total number of carbon atoms including branching must be at least 14, and further the average total number of carbon atoms for the branched primary alkyl moiety is within the range of greater than 14.5 to about 17.5.
- R, R1, and R2 are each independently selected from hydrogen and C1-C3 alkyl group (preferably hydrogen or C1-C2 alkyl, more preferably hydrogen or methyl, and most preferably methyl), provided R, R1, and R2 are not all hydrogen. Further, when z is 1, at least R or R1 is not hydrogen.
- Preferred mono-methyl branched primary alkyl sulfates are selected from the group consisting of: 3-methyl pentadecanol sulfate, 4-methyl pentadecanol sulfate, 5-methyl pentadecanol sulfate, 6-methyl pentadecanol sulfate, 7-methyl pentadecanol sulfate, 8-methyl pentadecanol sulfate, 9-methyl pentadecanol sulfate, 10-methyl pentadecanol sulfate, 11-methyl pentadecanol sulfate, 12-methyl pentadecanol sulfate, 13-methyl pentadecanol sulfate, 3-methyl hexadecanol sulfate, 4-methyl hexadecanol sulfate, 5-methyl hexadecanol sulfate, 6-methyl hexadecanol sulfate, 7-methyl hexadecanol sulfate, 8-methyl hexadecanol sulfate, 9-methyl hexadecanol sulfate, 10-methyl hexadecanol sulfate, 11-methyl hexadecanol sulfate, 12-methyl hexadecanol sulfate, 13-methyl hexadecanol sulfate, 14-methyl hexadecanol sulfate, and mixtures thereof.
- Preferred di-methyl branched primary alkyl sulfates are selected from the group consisting of: 2,3-methyl tetradecanol sulfate, 2,4-methyl tetradecanol sulfate, 2,5-methyl tetradecanol sulfate, 2,6-methyl tetradecanol sulfate, 2,7-methyl tetradecanol sulfate, 2,8-methyl tetradecanol sulfate, 2,9-methyl tetradecanol sulfate, 2,10-methyl tetradecanol sulfate, 2,11-methyl tetradecanol sulfate, 2,12-methyl tetradecanol sulfate, 2,3-methyl pentadecanol sulfate, 2,4-methyl pentadecanol sulfate, 2,5-methyl pentadecanol sulfate, 2,6-methyl pentadecanol sulfate, 2,7-methyl pentadecanol sulfate, 2,8-methyl pentadecanol sulfate, 2,9-methyl pentadecanol sulfate, 2,10-methyl pentadecanol sulfate, 2,11-methyl pentadecanol sulfate, 2,12-methyl pentadecanol sulfate, 2,13-methyl pentadecanol sulfate, and mixtures thereof.
- Essentially any alkoxylated nonionic surfactants are suitable herein. The ethoxylated and propoxylated nonionic surfactants are preferred.
- Preferred alkoxylated surfactants can be selected from the classes of the nonionic condensates of alkyl phenols, nonionic ethoxylated alcohols, nonionic ethoxylated/propoxylated fatty alcohols, nonionic ethoxylate/propoxylate condensates with propylene glycol, and the nonionic ethoxylate condensation products with propylene oxide/ethylene diamine adducts.
- Nonionic surfactant can be present in the detergent compositions. It may be preferred that the level of ethoxylated nonionic surfactants in the intimate mixture are below 10% by weight of the mixture, preferably even 5% by weight.
- The condensation products of aliphatic alcohols with from I to 25 moles of alkylene oxide, particularly ethylene oxide and/or propylene oxide, are suitable for use herein. The alkyl chain of the aliphatic alcohol can either be straight or branched, primary or secondary, and generally contains from 6 to 22 carbon atoms. Particularly preferred are the condensation products of alcohols having an alkyl group containing from 8 to 20 carbon atoms with from 2 to 10 moles of ethylene oxide per mole of alcohol.
- Suitable cationic surfactants to be used herein include the quaternary ammonium surfactants. Preferably the quaternary ammonium surfactant is a mono C6-C16, preferably C8-C14 N-alkyl or alkenyl ammonium surfactants wherein the remaining N positions are substituted by methyl, hydroxyethyl or hydroxypropyl groups. Preferred are also the mono-alkoxylated and bis-alkoxylated amine surfactants.
- Another suitable group of cationic surfactants which can be used in the detergent compositions or components thereof herein are cationic ester surfactants. The cationic ester surfactant is a, preferably water dispersible, compound having surfactant properties comprising at least one ester (i.e. -COO-) linkage and at least one cationically charged group.
- Suitable cationic ester surfactants, including choline ester surfactants, have for example been disclosed in US Patents No.s 4228042, 4239660 and 4260529.
- The composition herein can contain a transition metal containing bleach catalyst.
- One suitable type of bleach catalyst is disclosed in U.S. Pat. 4,430,243.
- Other types of bleach catalysts include the manganese-based complexes disclosed in U.S. Pat. 5,246,621 and U.S. Pat. 5,244,594. Preferred examples of these catalysts include MnIV 2(u-O)3(1,4,7-trimethyl-1,4,7-triazacyclononane)2-(PF6)2, MnIII 2(u-O)1(u-OAc)2(1,4,7-trimethyl-1,4,7-triazacyclononane)2-(ClO4)2, MnIV 4(u-O)6(1,4,7-triazacyclononane)4-(ClO4)2, MnIIIMnIV 4(u-O)1 (u-OAc)2-(1,4,7-trimethyl-1,4,7-triazacyclononane)2-(ClO4)3, and mixtures thereof. Others are described in European patent application publication no. 549,272. Other ligands suitable for use herein include 1,5,9-trimethyl-1,5,9-triazacyclododecane, 2-methyl-1,4,7-triazacyclononane, 2-methyl-1,4,7-triazacyclononane, 1,2,4,7-tetramethyl-1,4,7-triazacyclononane, and mixtures thereof.
- The bleach catalysts useful herein may also be selected as appropriate for the present invention. For examples of suitable bleach catalysts see U.S. Pat. 4,246,612 and U.S. Pat. 5,227,084. See also U.S. Pat. 5,194,416 which teaches mononuclear manganese (IV) complexes such as Mn(1,4,7-trimethyl-1,4,7-triazacyclononane)(OCH3)3-(PF6).
- Still another type of bleach catalyst, as disclosed in U.S. Pat. 5,114,606, is a water-soluble complex of manganese (III), and/or (IV) with a ligand which is a non-carboxylate polyhydroxy compound having at least three consecutive C-OH groups. Preferred ligands include sorbitol, iditol, dulsitol, mannitol, xylithol, arabitol, adonitol, meso- erythritol, meso-inositol, lactose, and mixtures thereof.
- U.S. Pat. 5,114,611 teaches a bleach catalyst comprising a complex of transition metals, including Mn, Co, Fe, or Cu, with an non-(macro)-cyclic ligand. Said ligands are of the formula:
- Other examples include binuclear Mn complexed with tetra-N-dentate and bi-N-dentate ligands, including N4MnIII(u-O)2MnIVN4)+and [Bipy2MnIII(u-O)2MnIVbipy2]-(ClO4)3.
- Other bleach catalysts are described, for example, in European patent application, publication no. 408,131 (cobalt complex catalysts), European patent applications, publication nos. 384,503, and 306,089 (metallo-porphyrin catalysts), U.S. 4,728,455 (manganese/multidentate ligand catalyst), U.S. 4,711,748 and European patent application, publication no. 224,952, (absorbed manganese on aluminosilicate catalyst), U.S. 4,601,845 (aluminosilicate support with manganese and zinc or magnesium salt), U.S. 4,626,373 (manganese/ligand catalyst), U.S. 4,119,557 (ferric complex catalyst), German Pat. specification 2,054,019 (cobalt chelant catalyst) Canadian 866,191 (transition metal-containing salts), U.S. 4,430,243 (chelants with manganese cations and non-catalytic metal cations), and U.S. 4,728,455 (manganese gluconate catalysts).
- In addition to the organic carboxylic acids, which may act as builders, and the carbonate salts, which may act as builders, the detergent compositions of the invention preferably comprise additional water-soluble and/ or water insoluble builder material. Preferred are silicates, aluminosilicates, crystalline layered silicates and phosphate salts.
- The compositions may for example comprise phosphate-containing builder material, preferably comprises tetrasodium pyrophosphate or even more preferably anhydrous sodium tripolyphosphate, present at a level of from 0.5% to 60%, more preferably from 5% to 50%, more preferably from 8% to 40.
- Suitable water-soluble builder compounds include the water soluble (poly)carboxylate salts and borates. The carboxylate or polycarboxylate builder can be momomeric or oligomeric in type although monomeric polycarboxylates are generally preferred for reasons of cost and performance.
- It may be preferred that the(poly)carboxylates and/ or polymeric or oligomeric (poly)carboxylates are present at levels of less than 5%, preferably less than 3% or even less than 2% or even 0% by weight of the compositions.
- Examples of largely water insoluble builders include the sodium aluminosilicates.
- Suitable aluminosilicate zeolites have the unit cell formula Naz[(AlO2)z(SiO2)y], xH2O wherein z and y are at least 6; the molar ratio of z to y is from 1.0 to 0.5 and x is at least 5, preferably from 7.5 to 276, more preferably from 10 to 264. The aluminosilicate material are in hydrated form and are preferably crystalline, containing from 10% to 28%, more preferably from 18% to 22% water in bound form.
- The aluminosilicate zeolites can be naturally occurring materials, but are preferably synthetically derived. Synthetic crystalline aluminosilicate ion exchange materials are available under the designations Zeolite A, Zeolite B, Zeolite P, Zeolite X, Zeolite HS and mixtures thereof. Zeolite A has the formula:
Na12[AlO2)12 (SiO2)12]. xH2O
wherein x is from 20 to 30, especially 27. Zeolite X has the formula Na86 [(AlO2)86(SiO2)106]. 276 H2O. - Another preferred aluminosilicate zeolite is zeolite MAP builder. Zeolite MAP is described in EP 384070A (Unilever). It is defined as an alkali metal aluminosilicate of the zeolite P type having a silicon to aluminium ratio not greater than 1.33, preferably within the range from 0.9 to 1.33 and more preferably within the range of from 0.9 to 1.2.
- The compositions herein may also comprise additional silicate material, including amorphous silicate material, meta-silicates, preferably at least crystalline layered silicate material such as sold under the trade name SKS-6. The silicate material is preferably present at a level of less than 20% by weight of the compositions, preferably less than 15% by weight or even less than 10% by weight.
- Heavy metal ion sequestrant are also useful additional ingredients herein. By heavy metal ion sequestrant it is meant herein components which act to sequester (chelate) heavy metal ions. These components may also have calcium and magnesium chelation capacity, but preferentially they show selectivity to binding heavy metal ions such as iron, manganese and copper.
- Heavy metal ion sequestrants are generally present at a level of from 0.005% to 10%, preferably from 0.1% to 5%, more preferably from 0.25% to 7.5% and most preferably from 0.3% to 2% by weight of the compositions.
- Suitable heavy metal ion sequestrants for use herein include organic phosphonates, such as the amino alkylene poly (alkylene phosphonates), alkali metal ethane 1-hydroxy disphosphonates and nitrilo trimethylene phosphonates.
- Preferred among the above species are diethylene triamine penta (methylene phosphonate), ethylene diamine tri (methylene phosphonate) hexamethylene diamine tetra (methylene phosphonate) and hydroxy-ethylene 1,1 diphosphonate, 1,1 hydroxyethane diphosphonic acid and 1,1 hydroxyethane dimethylene phosphonic acid.
- Other suitable heavy metal ion sequestrant for use herein include nitrilotriacetic acid and polyaminocarboxylic acids such as ethylenediaminotetracetic acid, ethylenediamine disuccinic acid, ethylenediamine diglutaric acid, 2-hydroxypropylenediamine disuccinic acid or any salts thereof.
- Other suitable heavy metal ion sequestrants for use herein are iminodiacetic acid derivatives such as 2-hydroxyethyl diacetic acid or glyceryl imino diacetic acid, described in EP-A-317,542 and EP-A-399,133. The iminodiacetic acid-N-2-hydroxypropyl sulfonic acid and aspartic acid N-carboxymethyl N-2-hydroxypropyl-3-sulfonic acid sequestrants described in EP-A-516,102 are also suitable herein. The β-alanine-N,N'-diacetic acid, aspartic acid-N,N'-diacetic acid, aspartic acid-N-monoacetic acid and iminodisuccinic acid sequestrants described in EP-A-509,382 are also suitable.
- EP-A-476,257 describes suitable amino based sequestrants. EP-A-510,331 describes suitable sequestrants derived from collagen, keratin or casein. EP-A-528,859 describes a suitable alkyl iminodiacetic acid sequestrant. Dipicolinic acid and 2-phosphonobutane-1,2,4-tricarboxylic acid are alos suitable. Glycinamide-N,N'-disuccinic acid (GADS), ethylenediamine-N-N'-diglutaric acid (EDDG) and 2-hydroxypropylenediamine-N-N'-disuccinic acid (HPDDS) are also suitable.
- Especially preferred are diethylenetriamine pentacetic acid, ethylenediamine-N,N'-disuccinic acid (EDDS) and 1,1 hydroxyethane diphosphonic acid or the alkali metal, alkaline earth metal, ammonium, or substituted ammonium salts thereof, or mixtures thereof.
- Another preferred ingredient useful herein is one or more additional enzymes.
- Preferred additional enzymatic materials include the commercially available lipases, cutinases, amylases, neutral and alkaline proteases, cellulases, endolases, esterases, pectinases, lactases and peroxidases conventionally incorporated into detergent compositions. Suitable enzymes are discussed in US Patents 3,519,570 and 3,533,139.
- Preferred commercially available protease enzymes include those sold under the tradenames Alcalase, Savinase, Primase, Durazym, and Esperase by Novo Industries A/S (Denmark), those sold under the tradename Maxatase, Maxacal and Maxapem by Gist-Brocades, those sold by Genencor International, and those sold under the tradename Opticlean and Optimase by Solvay Enzymes. Protease enzyme may be incorporated into the compositions in accordance with the invention at a level of from 0.0001 % to 4% active enzyme by weight of the composition.
- Additional organic polymeric compounds, not being the carboxylic acids herein, are preferred additional components of the compositions herein or the agglomerates herein, where they may act such as to bind the agglomerate components together.
- By organic polymeric compound it is meant herein essentially any polymeric organic compound commonly used as binder, dispersants, and anti-redeposition and soil suspension agents in detergent compositions, including any of the high molecular weight organic polymeric compounds described as clay flocculating agents herein, including quaternised ethoxylated (poly) amine clay-soil removal/ anti-redeposition agent in accord with the invention.
- Organic polymeric compound is typically incorporated in the detergent compositions of the invention at a level of from 0.01% to 30%, preferably from 0.1% to 15%, most preferably from 0.5% to 10% by weight of the compositions.
- Organic polymeric compounds suitable for incorporation in the detergent compositions herein include cellulose derivatives such as methylcellulose, sodium carboxymethylcellulose, hydroxypropylmethylcellulose and hydroxyethylcellulose. Highly preferred is also - or amide-modified carboxymethyl cellulose and - or amide-modified celluloses, or derivatives thereof
- Further useful organic polymeric compounds are the polyethylene glycols, particularly those of molecular weight 1000-10000, more particularly 2000 to 8000 and most preferably about 4000.
- The detergent compositions of the invention, when formulated for use in machine washing compositions, may comprise a suds suppressing system present at a level of from 0.01% to 15%, preferably from 0.02% to 10%, most preferably from 0.05% to 3% by weight of the composition.
- Suitable suds suppressing systems for use herein may comprise essentially any known antifoam compound, including, for example silicone antifoam compounds and 2-alkyl alcanol antifoam compounds.
- The composition herein may be a detergent which provides softening through the wash. Preferred may be that the composition comprises a clay softening agent, and preferably also a flocculating agent.
- Specific examples of suitable smectite clays include those selected from the classes of the montmorillonites, hectorites, volchonskoites, nontronites, saponites and sauconites, particularly those having an alkali or alkaline earth metal ion within the crystal lattice structure. Sodium or calcium montmorillonite are particularly preferred. Preferred are bentonite clays.
- Preferred as clay flocculating agents herein are organic polymeric materials having an average weight of from 100,000 to 10,000,000, preferably from 150,000 to 5,000,000, more preferably from 200,000 to 2,000,000.
- Suitable organic polymeric materials comprise homopolymers or copolymers containing monomeric units selected from alkylene oxide, particularly ethylene oxide, acrylamide, acrylic acid, vinyl alcohol, vinyl pyrrolidone, and ethylene imine. Homopolymers of, on particular, ethylene oxide, but also acrylamide and acrylic acid are preferred.
- European Patents No.s EP-A-299,575 and EP-A-313,146 in the name of the Procter and Gamble Company describe preferred organic polymeric clay flocculating agents for use herein.
- Other optional ingredients suitable for inclusion in the compositions of the invention include perfumes, such as the encapsultyed perfumes described above, brightners, speckles, including colours or dyes, filler salts, with sodium sulfate being a preferred filler salt.
- Also, minor amounts (e.g., less than about 20% by weight) of neutralizing agents, additional buffering agents, phase regulants, hydrotropes, enzyme stabilizing agents, polyacids, suds regulants, opacifiers, anti-oxidants, bactericides and dyes, such as those described in US Patent 4,285,841 to Barrat et al., issued August 25, 1981 can be present.
- Abbreviations used in the examples
LAS : Sodium linear C11-13 alkyl benzene sulfonate TAS : Sodium tallow alkyl sulfate CxyAS : Sodium C1x - C1y alkyl sulfate C46SAS : Sodium C14-C16 secondary (2,3) alkyl sulfate CxyEzS : Sodium C1x-C1y alkyl sulfate condensed with z moles of ethylene oxide CxyEz : C 1 x-C 1 y predominantly linear primary alcohol condensed with an average of z moles of ethylene oxide QAS : R2.N+(CH3)2(C2H4OH) with R2 = C12 - C14 QAS : R2.N+(CH3)2(C2H4OH) with R2 = C8 - C 11 Soap : Sodium linear alkyl carboxylate derived from an 80/20 mixture of tallow and coconut fatty acids STS : Sodium toluene sulphonate STPP : Anhydrous sodium tripolyphosphate Zeolite A : Hydrated sodium aluminosilicate of formula Na12(A1O2SiO2)12.27H2O having a primary particle size in the range from 0.1 to 10 micrometers (weight expressed on an anhydrous basis) NaSKS-6 : average particle size Crystalline layered silicate of formula δ- Na2Si2O5 of 25 microns Carbonate : Anydrous sodium carbonate Bicarbonate : Anhydrous sodium bicarbonate Silicate : Amorphous sodium silicate (SiO2:Na2O = 2.0:1) Sulfate : Anhydrous sodium sulfate Mg sulfate : Anhydrous magnesium sulfate Citrate : Tri-sodium citrate dihydrate MA/AA : Copolymer of 1:4 maleic/acrylic acid, average molecular weight about 70,000 AA : Sodium polyacrylate polymer of average molecular weight 4,500 CMC : Sodium carboxymethyl cellulose Cellulose ether : Methyl cellulose ether with a degree of polymerization of 650 available from Shin Etsu Chemicals Protease : Proteolytic enzyme, having 3.3% by weight of active enzyme, sold by NOVO Industries A/S under the tradename Savinase or proteolytic enzyme, having 4% by weight of active enzyme, as described in WO 95/10591, sold by Genencor Int. Inc. Amylase : Amylolytic enzyme, having 1.6% by weight of active enzyme, sold by NOVO Industries A/S under the tradename Termamyl 120T Lipase : Lipolytic enzyme, having 2.0% by weight of active enzyme, sold by NOVO Industries A/S under the tradename Lipolase or lipolytic enzyme, having 2.0% by weight of active enzyme, sold by NOVO Industries A/S under the tradename Lipolase Ultra PB4 : Particles containing sodium perborate tetrahydrate of nominal formula NaBO2.3H2 O, the particles having a weight average particle size of 950 microns, 85% particles having a particle size of from 850 microns to 950 microns PB1 : Particle containing anhydrous sodium perborate bleach of nominal formula NaBO2.H 202, the particles having a weight average particle size of 800 microns, 85% particles having a particle size of from 750 microns to 950 microns Percarbonate : Particle containing sodium percarbonate of nominal formula 2Na2CO3.3H2O2, the particles having a weight average particle size of 550 to 850 microns, 5% or less having a particle size of less than 300 microns and 7% or less having a particle size of more than 1180 microns NOBS : Particle comprising nonanoyloxybenzene sulfonate in the form of the sodium salt, the particles having a weight average particle size of 550 microns to 900 microns NAC-OBS : Particle comprising (6-nonamidocaproyl) oxybenzene sulfonate, the particles having a weight average particle size of from 550 microns to 900 microns TAED I : Particle containing tetraacetylethylenediamine, the particles having a weight average particle size of from 550 microns to 900 microns DTPA : Diethylene triamine pentaacetic acid DTPMP : Diethylene triamine penta (methylene phosphonate), marketed by Monsanto under the Tradename Dequest 2060 Photoactivated : Sulfonated zinc phthlocyanine encapsulated in bleach (1) dextrin soluble polymer Photoactivated : Sulfonated alumino phthlocyanine encapsulated in bleach (2) dextrin soluble polymer Brightener : Disodium 4,4'-bis(2-sulphostyryl)biphenyl or Disodium 4,4'-bis(4-anilino-6-morpholino-1.3.5-triazin-2-yl)amino) stilbene-2:2'-disulfonate EDDS : Ethylenediamine-N,N'-disuccinic acid, (S,S) isomer in the form of its sodium salt. HEDP : 1,1-hydroxyethane diphosphonic acid PEGx : Polyethylene glycol, with a molecular weight of x (typically 4,000) PEO : Polyethylene oxide, with an average molecular weight of 50,000 TEPAE : Tetraethylenepentaamine ethoxylate PVI : Polyvinyl imidosole, with an average molecular weight of 20,000 PVP : Polyvinylpyrolidone polymer, with an average molecular weight of 60,000 PVNO : Polyvinylpyridine N-oxide polymer, with an average molecular weight of 50,000 PVPVI : Copolymer of polyvinylpyrolidone and vinylimidazole, with an average molecular weight of 20,000 QEA : bis((C2H5O)(C2H4O)n)(CH3) -N+-C6H12-N+-(CH3) bis((C2H5O)-(C2H4 O))n, wherein n = from 20 to 30 SRP : Anionically end capped poly esters Silicone antifoam : Polydimethylsiloxane foam controller with siloxane or suds suppresser oxyalkylene copolymer as dispersing agent with a ratio of said foam controller to said dispersing agent of 10:1 to 100:1 - In the following examples all levels are quoted as % by weight of the composition:
TABLE I The following compositions are in accordance with the invention and have a pH in the wash, as measured by the method defined herein, of around 9.4 -9.85: A B C D E F G Spray-dried Granules - LAS 8.0 10.0 15.0 5.0 - TAS - 1.0 - 0.5 - MBAS 4.0 - 5.0 - C45AS - - 1.0 - C45AE3S - - 1.0 1.0 - QAS 1.0 1.0 - DTPA, HEDP and/or EDDS 0.3 0.3 0.5 0.3 - MgSO4 0.5 0.5 0.1 0.1 - Sodium carbonate 3.0 7.0 5.0 4.0 - Sodium sulphate - - - - - Sodium silicate 1.6R - - - 0.3 - STPP - - - - - Zeolite A 16. 0 18.0 20.0 20.0 - SKS-6 8.0 - - - MA/AA or AA 1.0 2.0 - - - PEG 4000 - 2.0 - - QEA 1.0 - - 1.0 - Brightener 0.05 0.05 0.05 - - Silicone oil 0.01 0.01 0.01 - - Agglomerate TAS 0.5 - 0.8 LAS 3.0 - - 2.0 - - MBAS - - - 14.0 8.0 C45AS - - 4.0 - 4.0 C45AE3 - - 1.0 - 1.0 Carbonate 1.0 - - 7.0 - 3.0 Sodium citrate 2.0 - - - Citric acid 2.0 - - - 9.0 - QEA - - 1.0 1.0 - SRP - - 0.2 - - Zeolite A 2.0 - - 15.0 15.0 25.0 Sodium silicate - - 0.5 - - PEG - - - - - MA/AA or A.A - - - - 4.0 1.0 0.8 Builder Agglomerates SKS-6 6.0 - - - 7.0 - - LAS 4.0 - - - 6.0 - - Dry-add particulate components Effervescence granule of carbonate/ bicarbonate/cit ric acid 1:1:1 10.0 15.0 15.0 - - 12.0 Sodium bicarbonate - - 4.0 2.0 8.0 4.0 5.0 Sodium carbonate 3.0 - 2.0 2.0 - 5.0 NOBS 3.0 3.0 4.5 - - - - TAED 2.5 2.0 - 6.0 4.5 5.0 - NACA-OBS - - - - - 4.5 Citric acid 5.0 5.0 3.0 - 7.0 7.0 10.0 Maleic acid - - - 10.0 Citrate - - - - - 10.0 2.0 Percarbonate 15.0 6.0 6.0 20.0 18.0 16.0 PB1 and/ or PB4 - 6.0 - - - Photobleach 0.02 0.02 0.02 0.1 - Enzymes (amylase, protease, lipase) 1.3 - 0.5 0.2 1.0 7.0 - Perfume (encapsulated) - 0.5 0.5 - 0.5 1.0 2.0 Suds suppressor 1.0 0.6 0.3 - 0.3 0.5 0.9 Soap 0.5 0.2 0.3 3.0 0.3 - - SKS-6 - - - 4.0 6.0 - - Bentonite clay - - - - - - - Dyed carbonate (blue, green) 0.5 0.5 1.0 2.0 0.5 1.0 - Spray-on Brightener 0.2 0.2 0.3 0.1 0.6 Dye - - - 0.3 - C25AE5 - - - - 0.7 Perfume 1.0 0.5 1.1 0.8 0.5 0.5 1.0 total balanced to 100%
Claims (6)
- A laundry detergent composition comprising a bleach system which contains a hydrogen peroxide source and at least 2.5% by weight of a peroxyacid bleach precursor, at least 15% by weight of a carbonate source, which may include the hydrogen peroxide source, at least 7% by weight of an acid, preferably an organic acid, whereby a 1% by weight mixture of the composition in demineralised water provides a pH from 8.8 to 9.9, and which further comprises an anionic surfactant.
- A composition according to claim 1, which comprises at least 7% by weight of an acid selected from carboxylic acids, preferably from citric acid, maleic acid, malic acid or mixtures thereof and whereby the carbonate source comprises at least 5% by weight of the composition of an alkali or earth alkali carbonate salt; at least 8% by weight of the composition of an alkali or earth alkali percarbonate salt and at least 2% by weight of the composition of an alkali or earth alkali bicarbonate salt.
- A composition according to claim 1 or 2 whereby the peroxyacid bleach precursor is present at a level of 3% to 10% by weight of the composition, or even 4% to 6% by weight and whereby the composition comprises from 10% to 45% by weight, more preferably from 15% to 35% by weight of the hydrogen peroxide source, preferably a percarbonate salt which when present forms part of the carbonate source.
- A composition according to any preceding claim whereby the pH is from 9.2 to 9.85 or even from 9.4 to 9.8.
- A composition according to any preceding claim comprising at least two peroxyacid bleach precursors, preferably TAED and a precursor selected from NOBS, DOBS or NACA-OBS.
- Washing method for washing textile in a washing machine whereby a composition according to any of claims 1 to 5 is introduced in a dispensing device which is then introduced in the drum of the machine prior to the introduction of wash water.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB9920678 | 1999-09-02 | ||
| GB9920678A GB2353800A (en) | 1999-09-02 | 1999-09-02 | Antibacterial detergent compositions |
| PCT/US2000/023964 WO2001016279A1 (en) | 1999-09-02 | 2000-08-31 | Bleaching detergent compositions |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1212398A1 EP1212398A1 (en) | 2002-06-12 |
| EP1212398B1 true EP1212398B1 (en) | 2006-06-14 |
Family
ID=10860174
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP00959694A Expired - Lifetime EP1212398B1 (en) | 1999-09-02 | 2000-08-31 | Bleaching detergent compositions |
Country Status (12)
| Country | Link |
|---|---|
| EP (1) | EP1212398B1 (en) |
| JP (1) | JP2003508590A (en) |
| CN (1) | CN1292062C (en) |
| AR (1) | AR035559A1 (en) |
| AT (1) | ATE329995T1 (en) |
| AU (1) | AU7097000A (en) |
| BR (1) | BR0013761B1 (en) |
| CA (1) | CA2382722A1 (en) |
| DE (1) | DE60028790D1 (en) |
| GB (1) | GB2353800A (en) |
| MX (1) | MXPA02002396A (en) |
| WO (1) | WO2001016279A1 (en) |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6010729A (en) | 1998-08-20 | 2000-01-04 | Ecolab Inc. | Treatment of animal carcasses |
| US7150884B1 (en) | 2000-07-12 | 2006-12-19 | Ecolab Inc. | Composition for inhibition of microbial growth |
| US6514556B2 (en) | 2000-12-15 | 2003-02-04 | Ecolab Inc. | Method and composition for washing poultry during processing |
| GB0118932D0 (en) * | 2001-08-02 | 2001-09-26 | Unilever Plc | Improvements relating to laundry compositions |
| US7771737B2 (en) | 2004-01-09 | 2010-08-10 | Ecolab Inc. | Medium chain peroxycarboxylic acid compositions |
| BRPI0506713A (en) | 2004-01-09 | 2007-05-02 | Ecolab Inc | medium chain peroxycarboxylic acid compositions |
| US7507429B2 (en) | 2004-01-09 | 2009-03-24 | Ecolab Inc. | Methods for washing carcasses, meat, or meat products with medium chain peroxycarboxylic acid compositions |
| US8999175B2 (en) | 2004-01-09 | 2015-04-07 | Ecolab Usa Inc. | Methods for washing and processing fruits, vegetables, and other produce with medium chain peroxycarboxylic acid compositions |
| US7887641B2 (en) | 2004-01-09 | 2011-02-15 | Ecolab Usa Inc. | Neutral or alkaline medium chain peroxycarboxylic acid compositions and methods employing them |
| US7504123B2 (en) | 2004-01-09 | 2009-03-17 | Ecolab Inc. | Methods for washing poultry during processing with medium chain peroxycarboxylic acid compositions |
| US7754670B2 (en) | 2005-07-06 | 2010-07-13 | Ecolab Inc. | Surfactant peroxycarboxylic acid compositions |
| US7759299B2 (en) * | 2006-07-24 | 2010-07-20 | Ecolab Inc. | Warewashing composition for use in automatic dishwashing machines |
| US7547421B2 (en) | 2006-10-18 | 2009-06-16 | Ecolab Inc. | Apparatus and method for making a peroxycarboxylic acid |
| US8075857B2 (en) | 2006-10-18 | 2011-12-13 | Ecolab Usa Inc. | Apparatus and method for making a peroxycarboxylic acid |
| GB201117231D0 (en) * | 2011-10-06 | 2011-11-16 | Givaudan Sa | Composition |
| CN102657231A (en) * | 2012-05-02 | 2012-09-12 | 裴理 | Environmentally-friendly high-efficiency insecticide |
| JP5700469B2 (en) * | 2012-12-28 | 2015-04-15 | 花王株式会社 | Powder detergent composition for clothing |
| JP6407682B2 (en) * | 2014-11-27 | 2018-10-17 | 花王株式会社 | Method for producing powder detergent composition for clothing |
| US20190136440A1 (en) * | 2016-06-09 | 2019-05-09 | Conopco, Inc., D/B/A Unilever | Laundry products |
| EP3469132A1 (en) | 2016-06-09 | 2019-04-17 | Unilever PLC | Laundry liquid mixing apparatus |
| TR202004710U5 (en) | 2017-10-05 | 2021-01-21 | Unilever Nv | Methods and apparatuses for specialized laundry. |
| CN107875425B (en) * | 2017-11-27 | 2020-06-30 | 中国人民解放军陆军防化学院 | Method for disinfecting the interior of a facility |
| WO2019160948A1 (en) | 2018-02-14 | 2019-08-22 | Ecolab Usa Inc. | Compositions and methods for the reduction of biofilm and spores from membranes |
| GB2571336A (en) | 2018-02-26 | 2019-08-28 | Unilever Plc | Methods and system for monitoring and replenishing one or more laundry components |
| CN114630934A (en) | 2019-11-01 | 2022-06-14 | 联合利华知识产权控股有限公司 | Recyclable automatic dosing container |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB8724900D0 (en) * | 1987-10-23 | 1987-11-25 | Unilever Plc | Detergent bleach compositions |
| DK166548B1 (en) * | 1991-03-15 | 1993-06-07 | Cleantabs As | PHOSPHATE-FREE MACHINE DISHWASH |
| US5423997A (en) * | 1991-05-31 | 1995-06-13 | Colgate Palmolive Co. | Spray dried powdered automatic dishwashing composition containing enzymes |
| EP0550077A1 (en) * | 1992-01-03 | 1993-07-07 | The Procter & Gamble Company | Granular laundry bleaching composition |
| DE69230098T2 (en) * | 1992-06-02 | 2000-05-25 | The Procter & Gamble Co., Cincinnati | Bleaching composition for laundry |
| DE4232170C2 (en) * | 1992-09-25 | 1999-09-16 | Henkel Kgaa | Weakly alkaline dish detergent |
| WO1996005283A1 (en) * | 1994-08-11 | 1996-02-22 | The Procter & Gamble Company | Detergent composition |
| DE69628784T2 (en) * | 1995-04-12 | 2004-02-12 | Cleantabs A/S | Bleach tablets |
| DE19547730A1 (en) * | 1995-12-20 | 1997-06-26 | Henkel Kgaa | Weakly alkaline dish washing |
| BR9710914A (en) * | 1996-07-29 | 1999-08-17 | Procter & Gamble | Activating asymmetric acyclical imide and compositions using the same |
| CN1249210C (en) * | 1996-10-15 | 2006-04-05 | 普罗格特-甘布尔公司 | Asymmetrical imide bleach activators and compositions employing the same |
| EP0932657B1 (en) * | 1996-10-15 | 2002-06-12 | The Procter & Gamble Company | Asymmetrical bleach activators and compositions employing the same |
-
1999
- 1999-09-02 GB GB9920678A patent/GB2353800A/en not_active Withdrawn
-
2000
- 2000-08-31 MX MXPA02002396A patent/MXPA02002396A/en active IP Right Grant
- 2000-08-31 JP JP2001520827A patent/JP2003508590A/en active Pending
- 2000-08-31 AU AU70970/00A patent/AU7097000A/en not_active Abandoned
- 2000-08-31 EP EP00959694A patent/EP1212398B1/en not_active Expired - Lifetime
- 2000-08-31 CN CN00815349.3A patent/CN1292062C/en not_active Expired - Fee Related
- 2000-08-31 DE DE60028790T patent/DE60028790D1/en not_active Expired - Lifetime
- 2000-08-31 CA CA002382722A patent/CA2382722A1/en not_active Abandoned
- 2000-08-31 AT AT00959694T patent/ATE329995T1/en not_active IP Right Cessation
- 2000-08-31 WO PCT/US2000/023964 patent/WO2001016279A1/en active IP Right Grant
- 2000-08-31 BR BRPI0013761-8A patent/BR0013761B1/en not_active IP Right Cessation
- 2000-09-01 AR ARP000104601A patent/AR035559A1/en not_active Application Discontinuation
Also Published As
| Publication number | Publication date |
|---|---|
| GB2353800A (en) | 2001-03-07 |
| AR035559A1 (en) | 2004-06-16 |
| MXPA02002396A (en) | 2002-08-20 |
| WO2001016279A1 (en) | 2001-03-08 |
| EP1212398A1 (en) | 2002-06-12 |
| CA2382722A1 (en) | 2001-03-08 |
| ATE329995T1 (en) | 2006-07-15 |
| AU7097000A (en) | 2001-03-26 |
| BR0013761A (en) | 2002-05-07 |
| CN1387563A (en) | 2002-12-25 |
| CN1292062C (en) | 2006-12-27 |
| JP2003508590A (en) | 2003-03-04 |
| DE60028790D1 (en) | 2006-07-27 |
| GB9920678D0 (en) | 1999-11-03 |
| BR0013761B1 (en) | 2014-04-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6548467B2 (en) | Sanitizing compositions and methods | |
| EP1212398B1 (en) | Bleaching detergent compositions | |
| JP4393582B2 (en) | Detergents containing heavy metal sequestering agents and delayed release peroxyacid bleach systems | |
| EP1165732B1 (en) | Fabric softening component | |
| JP2001524145A (en) | Detergent particles | |
| EP1165733B1 (en) | Detergent compositions | |
| EP1119604B1 (en) | Foaming system and detergent compositions containing the same | |
| ES2239320T3 (en) | DETERGENTS CONTAINING A DETERGENCE REINFORCER AND A DELAYED RELEASE ENZYME. | |
| CZ134797A3 (en) | Process of washing clothes by making use of a solid bleaching detergent dosage method | |
| GB2294705A (en) | Bleaching compositions | |
| US6881717B1 (en) | Fabric softening component | |
| GB2355722A (en) | Detergent compositions and methods for cleaning | |
| GB2348436A (en) | Detergent compositions | |
| JP3773540B2 (en) | Detergent composition | |
| GB2355721A (en) | Detergent compositions | |
| GB2339194A (en) | Layered crystalline silicate as detergent builder component | |
| EP1124929B1 (en) | Bleach-containing detergent composition | |
| MXPA99003687A (en) | Detergent compositions | |
| GB2355723A (en) | Detergent compositions | |
| WO2001012767A1 (en) | Disintegrating component and detergent composition containing it | |
| ES2237126T3 (en) | METHOD TO DISPENSE. | |
| MXPA99009397A (en) | Detergent particle | |
| JP2002524575A (en) | Distribution method | |
| MXPA01003515A (en) | Foaming system and detergent compositions containing the same | |
| MXPA99003686A (en) | Detergent compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20020329 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: BAKER, ASHLEY, SUTHERLAND Inventor name: HENSMAN, TIFFANY ALICE Inventor name: GOVERS, KRISTIEN, GRETA Inventor name: GRAY, PETER, GERARD |
|
| 17Q | First examination report despatched |
Effective date: 20030919 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20060614 Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 Ref country code: LI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 Ref country code: CH Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 60028790 Country of ref document: DE Date of ref document: 20060727 Kind code of ref document: P |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060831 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060914 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060914 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060915 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060925 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20061114 |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| 26 | Opposition filed |
Opponent name: UNILEVER PLC Effective date: 20070308 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| EN | Fr: translation not filed | ||
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060915 Ref country code: FR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070309 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20060614 |
|
| PLCK | Communication despatched that opposition was rejected |
Free format text: ORIGINAL CODE: EPIDOSNREJ1 |
|
| APBM | Appeal reference recorded |
Free format text: ORIGINAL CODE: EPIDOSNREFNO |
|
| APBP | Date of receipt of notice of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA2O |
|
| APAH | Appeal reference modified |
Free format text: ORIGINAL CODE: EPIDOSCREFNO |
|
| APBQ | Date of receipt of statement of grounds of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA3O |
|
| APBU | Appeal procedure closed |
Free format text: ORIGINAL CODE: EPIDOSNNOA9O |
|
| PLBN | Opposition rejected |
Free format text: ORIGINAL CODE: 0009273 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: OPPOSITION REJECTED |
|
| 27O | Opposition rejected |
Effective date: 20111021 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20190830 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 Expiry date: 20200830 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20200830 |