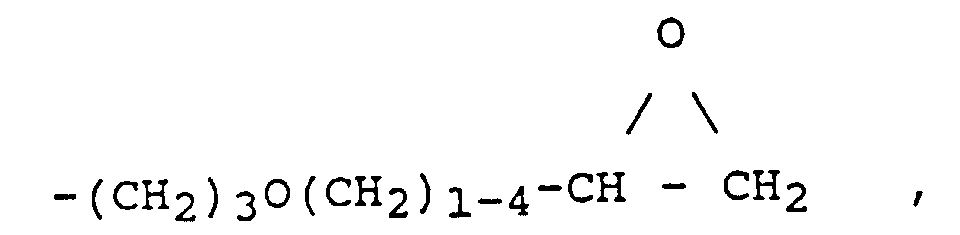EP0881325B1 - Siliconweichmacher für Jeans - Google Patents
Siliconweichmacher für Jeans Download PDFInfo
- Publication number
- EP0881325B1 EP0881325B1 EP98109267A EP98109267A EP0881325B1 EP 0881325 B1 EP0881325 B1 EP 0881325B1 EP 98109267 A EP98109267 A EP 98109267A EP 98109267 A EP98109267 A EP 98109267A EP 0881325 B1 EP0881325 B1 EP 0881325B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- radicals
- plasticizer
- radical
- organopolysiloxane
- silicone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 229920001296 polysiloxane Polymers 0.000 title claims description 65
- 239000004902 Softening Agent Substances 0.000 title description 2
- 239000000839 emulsion Substances 0.000 claims description 36
- 239000004744 fabric Substances 0.000 claims description 30
- 125000000217 alkyl group Chemical group 0.000 claims description 22
- COHYTHOBJLSHDF-UHFFFAOYSA-N indigo powder Natural products N1C2=CC=CC=C2C(=O)C1=C1C(=O)C2=CC=CC=C2N1 COHYTHOBJLSHDF-UHFFFAOYSA-N 0.000 claims description 13
- 235000000177 Indigofera tinctoria Nutrition 0.000 claims description 12
- 229940097275 indigo Drugs 0.000 claims description 12
- 238000000034 method Methods 0.000 claims description 12
- -1 polyethylene Polymers 0.000 description 73
- 239000004014 plasticizer Substances 0.000 description 48
- 150000003254 radicals Chemical class 0.000 description 35
- 239000003921 oil Substances 0.000 description 30
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 22
- 150000001412 amines Chemical class 0.000 description 22
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 16
- 239000004215 Carbon black (E152) Substances 0.000 description 11
- 150000002191 fatty alcohols Chemical class 0.000 description 11
- 229930195733 hydrocarbon Natural products 0.000 description 11
- 239000004530 micro-emulsion Substances 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- 229920013822 aminosilicone Polymers 0.000 description 10
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 10
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 9
- 239000003995 emulsifying agent Substances 0.000 description 8
- 229920000742 Cotton Polymers 0.000 description 7
- 239000004698 Polyethylene Substances 0.000 description 7
- 238000004383 yellowing Methods 0.000 description 7
- 239000004593 Epoxy Substances 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 239000004064 cosurfactant Substances 0.000 description 6
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 238000002156 mixing Methods 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- 229920002545 silicone oil Polymers 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 125000004432 carbon atom Chemical group C* 0.000 description 5
- 238000005562 fading Methods 0.000 description 5
- 239000007789 gas Substances 0.000 description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 238000005406 washing Methods 0.000 description 5
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 4
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 4
- 239000004907 Macro-emulsion Substances 0.000 description 4
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 4
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 4
- 150000001735 carboxylic acids Chemical class 0.000 description 4
- 235000014113 dietary fatty acids Nutrition 0.000 description 4
- 239000000975 dye Substances 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 239000000194 fatty acid Substances 0.000 description 4
- 229930195729 fatty acid Natural products 0.000 description 4
- ZSIAUFGUXNUGDI-UHFFFAOYSA-N hexan-1-ol Chemical compound CCCCCCO ZSIAUFGUXNUGDI-UHFFFAOYSA-N 0.000 description 4
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 4
- 229910052500 inorganic mineral Inorganic materials 0.000 description 4
- 239000011707 mineral Substances 0.000 description 4
- 239000002245 particle Substances 0.000 description 4
- XNLICIUVMPYHGG-UHFFFAOYSA-N pentan-2-one Chemical compound CCCC(C)=O XNLICIUVMPYHGG-UHFFFAOYSA-N 0.000 description 4
- 229920000573 polyethylene Polymers 0.000 description 4
- 229910052710 silicon Inorganic materials 0.000 description 4
- 239000004753 textile Substances 0.000 description 4
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 3
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-O ammonium group Chemical group [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 3
- PGMYKACGEOXYJE-UHFFFAOYSA-N anhydrous amyl acetate Natural products CCCCCOC(C)=O PGMYKACGEOXYJE-UHFFFAOYSA-N 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- BTANRVKWQNVYAZ-UHFFFAOYSA-N butan-2-ol Chemical compound CCC(C)O BTANRVKWQNVYAZ-UHFFFAOYSA-N 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 238000004040 coloring Methods 0.000 description 3
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical group COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 3
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 3
- 238000004043 dyeing Methods 0.000 description 3
- 150000004665 fatty acids Chemical class 0.000 description 3
- QQZOPKMRPOGIEB-UHFFFAOYSA-N n-butyl methyl ketone Natural products CCCCC(C)=O QQZOPKMRPOGIEB-UHFFFAOYSA-N 0.000 description 3
- MWUXSHHQAYIFBG-UHFFFAOYSA-N nitrogen oxide Inorganic materials O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 3
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 229920006395 saturated elastomer Polymers 0.000 description 3
- 239000010703 silicon Substances 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- BBMCTIGTTCKYKF-UHFFFAOYSA-N 1-heptanol Chemical compound CCCCCCCO BBMCTIGTTCKYKF-UHFFFAOYSA-N 0.000 description 2
- CETWDUZRCINIHU-UHFFFAOYSA-N 2-heptanol Chemical compound CCCCCC(C)O CETWDUZRCINIHU-UHFFFAOYSA-N 0.000 description 2
- ZPVFWPFBNIEHGJ-UHFFFAOYSA-N 2-octanone Chemical compound CCCCCCC(C)=O ZPVFWPFBNIEHGJ-UHFFFAOYSA-N 0.000 description 2
- RHLVCLIPMVJYKS-UHFFFAOYSA-N 3-octanone Chemical compound CCCCCC(=O)CC RHLVCLIPMVJYKS-UHFFFAOYSA-N 0.000 description 2
- YVBCULSIZWMTFY-UHFFFAOYSA-N 4-Heptanol Natural products CCCC(O)CCC YVBCULSIZWMTFY-UHFFFAOYSA-N 0.000 description 2
- HCFAJYNVAYBARA-UHFFFAOYSA-N 4-heptanone Chemical compound CCCC(=O)CCC HCFAJYNVAYBARA-UHFFFAOYSA-N 0.000 description 2
- MWRSABPHNREIIX-UHFFFAOYSA-N 9,9-dimethyldecan-1-ol Chemical compound CC(C)(C)CCCCCCCCO MWRSABPHNREIIX-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- 102000013142 Amylases Human genes 0.000 description 2
- 108010065511 Amylases Proteins 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- RZKSECIXORKHQS-UHFFFAOYSA-N Heptan-3-ol Chemical compound CCCCC(O)CC RZKSECIXORKHQS-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- 239000012190 activator Substances 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 238000004061 bleaching Methods 0.000 description 2
- XUPYJHCZDLZNFP-UHFFFAOYSA-N butyl butanoate Chemical compound CCCCOC(=O)CCC XUPYJHCZDLZNFP-UHFFFAOYSA-N 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- HUCVOHYBFXVBRW-UHFFFAOYSA-M caesium hydroxide Chemical compound [OH-].[Cs+] HUCVOHYBFXVBRW-UHFFFAOYSA-M 0.000 description 2
- 125000002091 cationic group Chemical group 0.000 description 2
- WOWHHFRSBJGXCM-UHFFFAOYSA-M cetyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+](C)(C)C WOWHHFRSBJGXCM-UHFFFAOYSA-M 0.000 description 2
- 239000007859 condensation product Substances 0.000 description 2
- MLIREBYILWEBDM-UHFFFAOYSA-N cyanoacetic acid Chemical compound OC(=O)CC#N MLIREBYILWEBDM-UHFFFAOYSA-N 0.000 description 2
- 229940028356 diethylene glycol monobutyl ether Drugs 0.000 description 2
- 125000003700 epoxy group Chemical group 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 150000002170 ethers Chemical class 0.000 description 2
- 238000007046 ethoxylation reaction Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- CATSNJVOTSVZJV-UHFFFAOYSA-N heptan-2-one Chemical compound CCCCCC(C)=O CATSNJVOTSVZJV-UHFFFAOYSA-N 0.000 description 2
- NGAZZOYFWWSOGK-UHFFFAOYSA-N heptan-3-one Chemical compound CCCCC(=O)CC NGAZZOYFWWSOGK-UHFFFAOYSA-N 0.000 description 2
- QNVRIHYSUZMSGM-UHFFFAOYSA-N hexan-2-ol Chemical compound CCCCC(C)O QNVRIHYSUZMSGM-UHFFFAOYSA-N 0.000 description 2
- ZOCHHNOQQHDWHG-UHFFFAOYSA-N hexan-3-ol Chemical compound CCCC(O)CC ZOCHHNOQQHDWHG-UHFFFAOYSA-N 0.000 description 2
- AOGQPLXWSUTHQB-UHFFFAOYSA-N hexyl acetate Chemical compound CCCCCCOC(C)=O AOGQPLXWSUTHQB-UHFFFAOYSA-N 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- YKYONYBAUNKHLG-UHFFFAOYSA-N n-Propyl acetate Natural products CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 description 2
- SJWFXCIHNDVPSH-UHFFFAOYSA-N octan-2-ol Chemical compound CCCCCCC(C)O SJWFXCIHNDVPSH-UHFFFAOYSA-N 0.000 description 2
- NMRPBPVERJPACX-UHFFFAOYSA-N octan-3-ol Chemical compound CCCCCC(O)CC NMRPBPVERJPACX-UHFFFAOYSA-N 0.000 description 2
- JCGNDDUYTRNOFT-UHFFFAOYSA-N oxolane-2,4-dione Chemical compound O=C1COC(=O)C1 JCGNDDUYTRNOFT-UHFFFAOYSA-N 0.000 description 2
- JYVLIDXNZAXMDK-UHFFFAOYSA-N pentan-2-ol Chemical compound CCCC(C)O JYVLIDXNZAXMDK-UHFFFAOYSA-N 0.000 description 2
- AQIXEPGDORPWBJ-UHFFFAOYSA-N pentan-3-ol Chemical compound CCC(O)CC AQIXEPGDORPWBJ-UHFFFAOYSA-N 0.000 description 2
- FDPIMTJIUBPUKL-UHFFFAOYSA-N pentan-3-one Chemical compound CCC(=O)CC FDPIMTJIUBPUKL-UHFFFAOYSA-N 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 229920000151 polyglycol Polymers 0.000 description 2
- 239000010695 polyglycol Substances 0.000 description 2
- FGIUAXJPYTZDNR-UHFFFAOYSA-N potassium nitrate Chemical compound [K+].[O-][N+]([O-])=O FGIUAXJPYTZDNR-UHFFFAOYSA-N 0.000 description 2
- CPRMKOQKXYSDML-UHFFFAOYSA-M rubidium hydroxide Chemical compound [OH-].[Rb+] CPRMKOQKXYSDML-UHFFFAOYSA-M 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- 229920002554 vinyl polymer Polymers 0.000 description 2
- ULPMRIXXHGUZFA-UHFFFAOYSA-N (R)-4-Methyl-3-hexanone Natural products CCC(C)C(=O)CC ULPMRIXXHGUZFA-UHFFFAOYSA-N 0.000 description 1
- FKTXDTWDCPTPHK-UHFFFAOYSA-N 1,1,1,2,3,3,3-heptafluoropropane Chemical compound FC(F)(F)[C](F)C(F)(F)F FKTXDTWDCPTPHK-UHFFFAOYSA-N 0.000 description 1
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 description 1
- RRQYJINTUHWNHW-UHFFFAOYSA-N 1-ethoxy-2-(2-ethoxyethoxy)ethane Chemical compound CCOCCOCCOCC RRQYJINTUHWNHW-UHFFFAOYSA-N 0.000 description 1
- BMVXCPBXGZKUPN-UHFFFAOYSA-N 1-hexanamine Chemical compound CCCCCCN BMVXCPBXGZKUPN-UHFFFAOYSA-N 0.000 description 1
- OAYXUHPQHDHDDZ-UHFFFAOYSA-N 2-(2-butoxyethoxy)ethanol Chemical compound CCCCOCCOCCO OAYXUHPQHDHDDZ-UHFFFAOYSA-N 0.000 description 1
- SBASXUCJHJRPEV-UHFFFAOYSA-N 2-(2-methoxyethoxy)ethanol Chemical group COCCOCCO SBASXUCJHJRPEV-UHFFFAOYSA-N 0.000 description 1
- IEORSVTYLWZQJQ-UHFFFAOYSA-N 2-(2-nonylphenoxy)ethanol Chemical compound CCCCCCCCCC1=CC=CC=C1OCCO IEORSVTYLWZQJQ-UHFFFAOYSA-N 0.000 description 1
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 1
- QNVRIHYSUZMSGM-LURJTMIESA-N 2-Hexanol Natural products CCCC[C@H](C)O QNVRIHYSUZMSGM-LURJTMIESA-N 0.000 description 1
- PLFJWWUZKJKIPZ-UHFFFAOYSA-N 2-[2-[2-(2,6,8-trimethylnonan-4-yloxy)ethoxy]ethoxy]ethanol Chemical compound CC(C)CC(C)CC(CC(C)C)OCCOCCOCCO PLFJWWUZKJKIPZ-UHFFFAOYSA-N 0.000 description 1
- IHEDBVUTTQXGSJ-UHFFFAOYSA-M 2-[bis(2-oxidoethyl)amino]ethanolate;titanium(4+);hydroxide Chemical compound [OH-].[Ti+4].[O-]CCN(CC[O-])CC[O-] IHEDBVUTTQXGSJ-UHFFFAOYSA-M 0.000 description 1
- WLJVXDMOQOGPHL-PPJXEINESA-N 2-phenylacetic acid Chemical compound O[14C](=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-PPJXEINESA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- SLRMQYXOBQWXCR-UHFFFAOYSA-N 2154-56-5 Chemical compound [CH2]C1=CC=CC=C1 SLRMQYXOBQWXCR-UHFFFAOYSA-N 0.000 description 1
- NMRPBPVERJPACX-QMMMGPOBSA-N 3-Octanol Natural products CCCCC[C@@H](O)CC NMRPBPVERJPACX-QMMMGPOBSA-N 0.000 description 1
- 125000004179 3-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(Cl)=C1[H] 0.000 description 1
- PFCHFHIRKBAQGU-UHFFFAOYSA-N 3-hexanone Chemical compound CCCC(=O)CC PFCHFHIRKBAQGU-UHFFFAOYSA-N 0.000 description 1
- AFPHTEQTJZKQAQ-UHFFFAOYSA-N 3-nitrobenzoic acid Chemical compound OC(=O)C1=CC=CC([N+]([O-])=O)=C1 AFPHTEQTJZKQAQ-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- OTLNPYWUJOZPPA-UHFFFAOYSA-N 4-nitrobenzoic acid Chemical compound OC(=O)C1=CC=C([N+]([O-])=O)C=C1 OTLNPYWUJOZPPA-UHFFFAOYSA-N 0.000 description 1
- PXRKCOCTEMYUEG-UHFFFAOYSA-N 5-aminoisoindole-1,3-dione Chemical compound NC1=CC=C2C(=O)NC(=O)C2=C1 PXRKCOCTEMYUEG-UHFFFAOYSA-N 0.000 description 1
- PSBKJPTZCVYXSD-UHFFFAOYSA-N 5-methylheptan-3-one Chemical compound CCC(C)CC(=O)CC PSBKJPTZCVYXSD-UHFFFAOYSA-N 0.000 description 1
- 239000004382 Amylase Substances 0.000 description 1
- 206010063659 Aversion Diseases 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 108010059892 Cellulase Proteins 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical group [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- KHMVXSQLPUNRCF-UHFFFAOYSA-N DL-Adalin Natural products C1CCC2CC(=O)CC1(CCCCC)N2 KHMVXSQLPUNRCF-UHFFFAOYSA-N 0.000 description 1
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Natural products CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical group FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- WJYIASZWHGOTOU-UHFFFAOYSA-N Heptylamine Chemical compound CCCCCCCN WJYIASZWHGOTOU-UHFFFAOYSA-N 0.000 description 1
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 1
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 1
- 229920000881 Modified starch Polymers 0.000 description 1
- 229930182559 Natural dye Natural products 0.000 description 1
- IGFHQQFPSIBGKE-UHFFFAOYSA-N Nonylphenol Natural products CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 1
- CYTYCFOTNPOANT-UHFFFAOYSA-N Perchloroethylene Chemical compound ClC(Cl)=C(Cl)Cl CYTYCFOTNPOANT-UHFFFAOYSA-N 0.000 description 1
- 229920000297 Rayon Polymers 0.000 description 1
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 1
- 229920004482 WACKER® Polymers 0.000 description 1
- YUDRVAHLXDBKSR-UHFFFAOYSA-N [CH]1CCCCC1 Chemical compound [CH]1CCCCC1 YUDRVAHLXDBKSR-UHFFFAOYSA-N 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- MLFHJEHSLIIPHL-UHFFFAOYSA-N acetic acid isopentyl ester Natural products CC(C)CCOC(C)=O MLFHJEHSLIIPHL-UHFFFAOYSA-N 0.000 description 1
- ZOIORXHNWRGPMV-UHFFFAOYSA-N acetic acid;zinc Chemical compound [Zn].CC(O)=O.CC(O)=O ZOIORXHNWRGPMV-UHFFFAOYSA-N 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 239000003619 algicide Substances 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 229910001854 alkali hydroxide Inorganic materials 0.000 description 1
- 229910001860 alkaline earth metal hydroxide Inorganic materials 0.000 description 1
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000002877 alkyl aryl group Chemical group 0.000 description 1
- 125000005211 alkyl trimethyl ammonium group Chemical group 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- OBFQBDOLCADBTP-UHFFFAOYSA-N aminosilicon Chemical compound [Si]N OBFQBDOLCADBTP-UHFFFAOYSA-N 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 235000019418 amylase Nutrition 0.000 description 1
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 150000005840 aryl radicals Chemical class 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 239000003899 bactericide agent Substances 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 238000005282 brightening Methods 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Chemical group BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Chemical group 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- OPNPQXLQERQBBV-UHFFFAOYSA-N carbromal Chemical compound CCC(Br)(CC)C(=O)NC(N)=O OPNPQXLQERQBBV-UHFFFAOYSA-N 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 238000005660 chlorination reaction Methods 0.000 description 1
- 239000000460 chlorine Chemical group 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- FOCAUTSVDIKZOP-UHFFFAOYSA-N chloroacetic acid Chemical compound OC(=O)CCl FOCAUTSVDIKZOP-UHFFFAOYSA-N 0.000 description 1
- 229940106681 chloroacetic acid Drugs 0.000 description 1
- 239000007957 coemulsifier Substances 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000004956 cyclohexylene group Chemical group 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000009990 desizing Methods 0.000 description 1
- 125000003963 dichloro group Chemical group Cl* 0.000 description 1
- 229960005215 dichloroacetic acid Drugs 0.000 description 1
- 229940019778 diethylene glycol diethyl ether Drugs 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 238000004945 emulsification Methods 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- QUPDWYMUPZLYJZ-UHFFFAOYSA-N ethyl Chemical compound C[CH2] QUPDWYMUPZLYJZ-UHFFFAOYSA-N 0.000 description 1
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000002979 fabric softener Substances 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 239000013505 freshwater Substances 0.000 description 1
- 238000003958 fumigation Methods 0.000 description 1
- 230000000855 fungicidal effect Effects 0.000 description 1
- 239000000417 fungicide Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 238000005858 glycosidation reaction Methods 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 150000002402 hexoses Chemical class 0.000 description 1
- 239000008240 homogeneous mixture Substances 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- COHYTHOBJLSHDF-BUHFOSPRSA-N indigo dye Chemical compound N\1C2=CC=CC=C2C(=O)C/1=C1/C(=O)C2=CC=CC=C2N1 COHYTHOBJLSHDF-BUHFOSPRSA-N 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- GJRQTCIYDGXPES-UHFFFAOYSA-N iso-butyl acetate Natural products CC(C)COC(C)=O GJRQTCIYDGXPES-UHFFFAOYSA-N 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- JMMWKPVZQRWMSS-UHFFFAOYSA-N isopropanol acetate Natural products CC(C)OC(C)=O JMMWKPVZQRWMSS-UHFFFAOYSA-N 0.000 description 1
- OQAGVSWESNCJJT-UHFFFAOYSA-N isovaleric acid methyl ester Natural products COC(=O)CC(C)C OQAGVSWESNCJJT-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 230000003641 microbiacidal effect Effects 0.000 description 1
- 229940124561 microbicide Drugs 0.000 description 1
- 239000002855 microbicide agent Substances 0.000 description 1
- 235000019426 modified starch Nutrition 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 239000000978 natural dye Substances 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- GQPLMRYTRLFLPF-UHFFFAOYSA-N nitrous oxide Inorganic materials [O-][N+]#N GQPLMRYTRLFLPF-UHFFFAOYSA-N 0.000 description 1
- 239000012454 non-polar solvent Substances 0.000 description 1
- 239000012875 nonionic emulsifier Substances 0.000 description 1
- 229920000847 nonoxynol Polymers 0.000 description 1
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 1
- IOQPZZOEVPZRBK-UHFFFAOYSA-N octan-1-amine Chemical compound CCCCCCCCN IOQPZZOEVPZRBK-UHFFFAOYSA-N 0.000 description 1
- WOFPPJOZXUTRAU-UHFFFAOYSA-N octan-4-ol Chemical compound CCCCC(O)CCC WOFPPJOZXUTRAU-UHFFFAOYSA-N 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 238000006385 ozonation reaction Methods 0.000 description 1
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 description 1
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- DPBLXKKOBLCELK-UHFFFAOYSA-N pentan-1-amine Chemical compound CCCCCN DPBLXKKOBLCELK-UHFFFAOYSA-N 0.000 description 1
- IGEIPFLJVCPEKU-UHFFFAOYSA-N pentan-2-amine Chemical compound CCCC(C)N IGEIPFLJVCPEKU-UHFFFAOYSA-N 0.000 description 1
- 150000002972 pentoses Chemical group 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- ONJQDTZCDSESIW-UHFFFAOYSA-N polidocanol Chemical compound CCCCCCCCCCCCOCCOCCOCCOCCOCCOCCOCCOCCOCCO ONJQDTZCDSESIW-UHFFFAOYSA-N 0.000 description 1
- 229920000137 polyphosphoric acid Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 239000004323 potassium nitrate Substances 0.000 description 1
- 235000010333 potassium nitrate Nutrition 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 238000009895 reductive bleaching Methods 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- BHRZNVHARXXAHW-UHFFFAOYSA-N sec-butylamine Chemical compound CCC(C)N BHRZNVHARXXAHW-UHFFFAOYSA-N 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 125000000542 sulfonic acid group Chemical group 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 239000012209 synthetic fiber Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- JAELLLITIZHOGQ-UHFFFAOYSA-N tert-butyl propanoate Chemical compound CCC(=O)OC(C)(C)C JAELLLITIZHOGQ-UHFFFAOYSA-N 0.000 description 1
- YBRBMKDOPFTVDT-UHFFFAOYSA-N tert-butylamine Chemical compound CC(C)(C)N YBRBMKDOPFTVDT-UHFFFAOYSA-N 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000009988 textile finishing Methods 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical group OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 1
- 238000009423 ventilation Methods 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 238000009941 weaving Methods 0.000 description 1
- 125000005023 xylyl group Chemical group 0.000 description 1
- 239000004246 zinc acetate Substances 0.000 description 1
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/44—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders
- D06P1/52—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders using compositions containing synthetic macromolecular substances
- D06P1/5264—Macromolecular compounds obtained otherwise than by reactions involving only unsaturated carbon-to-carbon bonds
- D06P1/5292—Macromolecular compounds obtained otherwise than by reactions involving only unsaturated carbon-to-carbon bonds containing Si-atoms
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/44—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders
- D06P1/46—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using insoluble pigments or auxiliary substances, e.g. binders using compositions containing natural macromolecular substances or derivatives thereof
- D06P1/48—Derivatives of carbohydrates
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P5/00—Other features in dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form
- D06P5/02—After-treatment
- D06P5/04—After-treatment with organic compounds
- D06P5/08—After-treatment with organic compounds macromolecular
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/22—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using vat dyestuffs including indigo
- D06P1/228—Indigo
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T442/00—Fabric [woven, knitted, or nonwoven textile or cloth, etc.]
- Y10T442/20—Coated or impregnated woven, knit, or nonwoven fabric which is not [a] associated with another preformed layer or fiber layer or, [b] with respect to woven and knit, characterized, respectively, by a particular or differential weave or knit, wherein the coating or impregnation is neither a foamed material nor a free metal or alloy layer
- Y10T442/2311—Coating or impregnation is a lubricant or a surface friction reducing agent other than specified as improving the "hand" of the fabric or increasing the softness thereof
- Y10T442/2328—Organosilicon containing
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T442/00—Fabric [woven, knitted, or nonwoven textile or cloth, etc.]
- Y10T442/20—Coated or impregnated woven, knit, or nonwoven fabric which is not [a] associated with another preformed layer or fiber layer or, [b] with respect to woven and knit, characterized, respectively, by a particular or differential weave or knit, wherein the coating or impregnation is neither a foamed material nor a free metal or alloy layer
- Y10T442/2352—Coating or impregnation functions to soften the feel of or improve the "hand" of the fabric
Definitions
- the invention relates to tissue dyed with indigo. method for the treatment of a fabric stained with indigo.
- Tissue dyed with indigo is mostly the so-called denim fabric, which is a coarse Cotton fabrics are mainly used to manufacture the so-called blue jeans, i.e. around jeans and other items of clothing based on denim fabrics, e.g. Shirts, Jackets, skirts etc., is used. These clothes are made according to special and characteristic of this type of clothing Process manufactured.
- T-shirts can also be the undyed, ready-made garment subsequently colored according to need.
- Classic jeans are predominantly made of cotton and dyed blue. There are denim articles in a wide variety of colors, but the classic jeans are blue. "Blue Jeans” is a linguistically firmly anchored generic term. Traditionally, the coloring of the jeans must be done with indigo. Indigo is a natural dye, but today it is mainly manufactured synthetically.
- the invention relates to indigo-dyed fabric, which thereby is characterized in that it is at least one organopolysiloxane and contains at least one alkyl polyglycoside.
- Indigo-dyed fabric is basically any form of fabric, but preferably cotton fabric dyed with indigo, Linen fabrics, viscose fabrics, fabrics made of synthetic fibers, whereby Cotton fabrics preferred and denim fabrics particularly preferred is.
- the organopolysiloxanes are preferably Organopolysiloxanes (a), which polar groups on Si-C-bonded Have hydrocarbon radicals, such as preferably amino, Ammonium, epoxy, hydroxy, amido, mercapto, carboxy and / or sulfonic acid groups, their salts or esters.
- hydrocarbon radicals such as preferably amino, Ammonium, epoxy, hydroxy, amido, mercapto, carboxy and / or sulfonic acid groups, their salts or esters.
- optionally substituted hydrocarbyloxy R are preferably a directly bonded to a silicon atom of oxygen atom-bonded substituted or unsubstituted hydrocarbon radicals R according to the examples above-mentioned, in particular alkoxy groups having 1 to 18 carbon atoms and phenoxy radicals, especially the methoxy, ethoxy, n-propoxy , iso-propoxy and phenoxy.
- Preferably at most 5% of the radicals R are optionally substituted hydrocarbonoxy radicals.
- Examples of the divalent C 1 to C 18 hydrocarbon radicals R 1 are preferably saturated straight-chain or branched-chain or cyclic alkylene radicals such as the methylene and ethylene radical and propylene, butylene, pentylene, hexylene, 2-methylpropylene, cyclohexylene and and octadecylene radicals or unsaturated alkylene or arylene radicals, such as the hexenylene radical and phenylene radicals, the n-propylene radical and the 2-methylpropylene radical being particularly preferred.
- saturated straight-chain or branched-chain or cyclic alkylene radicals such as the methylene and ethylene radical and propylene, butylene, pentylene, hexylene, 2-methylpropylene, cyclohexylene and and octadecylene radicals or unsaturated alkylene or arylene radicals, such as the hexenylene radical and phenylene radicals
- hydrocarbon radicals R 2 are preferably the examples given for R.
- halogen-substituted hydrocarbon radicals R 2 are haloalkyl radicals, such as the 3,3,3-trifluoro-n-propyl radical, the 2,2,2,2 ', 2', 2'-hexafluoroisopropyl radical, the heptafluoroisopropyl radical, and haloaryl radicals, such as the above -, m- and p-chlorophenyl.
- Examples of mineral acids that deal with the above amino functional hydrocarbon residues to the corresponding have ammonium-functional residues implemented preferably salt, perchlor, sulfur, sulfurous, saltpetre, nitrous, hydrofluoric, phosphoric, diphosphoric and polyphosphoric acids.
- suitable carboxylic acids are preferred Formic, acetic, propionic, butanoic, citric, Trichloro, dichloro and chloroacetic acid, trifluoroacetic acid, Cyanoacetic acid, phenylacetic acid, benzoic acid, m- and p-nitrobenzoic acid, Oxalic acid, malonic acid and lactic acid.
- Especially the ammonium functional groups obtainable with acetic acid are preferred Hydrocarbon residues.
- amido-functional radicals are preferably the ⁇ -acetamidopropyl radical, partially or fully acetylated ⁇ -aminoethyl- ⁇ -aminopropyl residues.
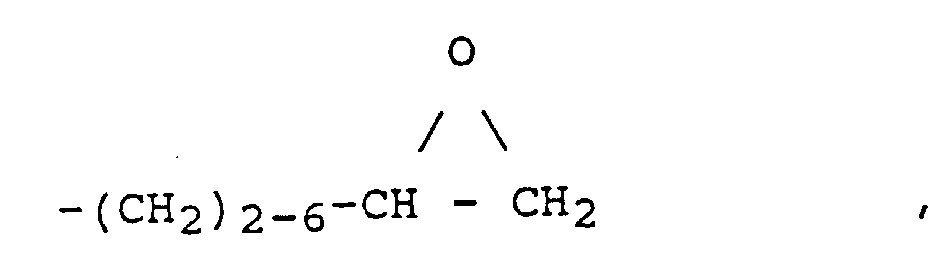
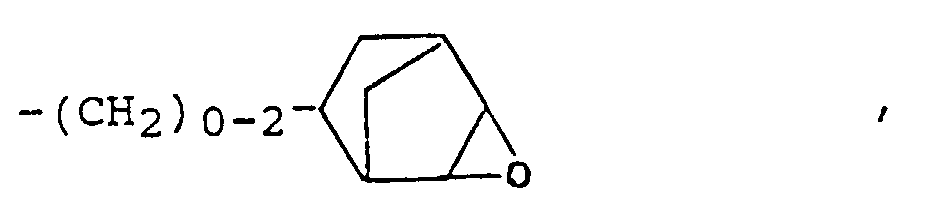
- epoxy-functional radicals R ' are radicals of the general formulas (III) and (IV) where A is an alkyl, alkoxyalkyl, aryl or alkaryl radical.
- the preferred epoxy numbers of the epoxy functional organopolysiloxanes (a) are 0.5 - 0.001 (equiv./100 g), in particular at 0.2 - 0.01 (equiv./100 g).
- the epoxy number of one epoxy functional organopolysiloxane gives the number of equivalents of epoxy, namely the number of moles of epoxy groups, which is contained in 100 grams of organopolysiloxane (a).
- radicals R ' are particularly preferred as the carboxy-functional radicals - (CH 2 ) 10 -COOH, -CH 2 CH (CH 3 ) -COOH and - (CH 2 ) 2 -S-CH 2 -COOH.
- the preferred acid numbers of the carboxy-functional organopolysiloxanes (a) are 1-100 (mg KOH / g), preferably 5-50 and especially 10-30.
- the acid number of a carboxy functional Organopolysiloxane (a) indicates the number Milligrams of potassium hydroxide, which is necessary to get the free Neutralize acids in one gram of carboxy functional Organopolysiloxane (a) are included.
- the radicals R are preferably methyl, ethyl, phenyl, methoxy and / or vinyl radicals. Because of the easier accessibility, preferably 50% of the R radicals, in particular at least 80% of the R radicals, are methyl radicals.
- the organopolysiloxane (mixture) used in emulsions is preferably liquid.
- Process used organopolysiloxanes preferably Viscosities from 100 mPa * s to 1,000,000 mPa * s, respectively measured at 25 ° C.
- an amino-functional organopolysiloxane is used to prepare the ammonium-functional organopolysiloxane (a) which is preferably used in the emulsions according to the invention, it is preferred that it has an amine number from 0.1 to 3.0, in particular from 0.2 to 0.9.
- the amine number of an amino-functional substance is determined as the consumption in cm 3 of 1N hydrochloric acid when titrating 1 g of the amino-functional substance.
- Alkyl polyglycosides with a saturated content are particularly preferred Alkyl radical with an average of 8 to 14 carbon atoms and one average degree of glycosidation n between 1.1 and 3.
- the emulsions used according to the invention have a higher one Stability to foreign electrolytes, such as magnesium and sodium salts, on as corresponding emulsions in which alkyl polyglycol ethers can be used as emulsifiers.
- the emulsions used according to the invention contain relatively small amounts of emulsifiers, in particular 5 to 100 parts by weight of alkyl polyglycosides (b) per 100 parts by weight organopolysiloxanes containing polar groups (a).
- the emulsions used according to the invention have a discontinuous Oil phase which the contains organopolysiloxanes (a) containing polar groups, and a continuous water phase.
- the proportions of the organopolysiloxane (a) and the continuous Water phase can be varied in a wide range, depending on which fixed content in the used according to the invention Emulsions and microemulsions is sought.
- the proportion of organopolysiloxane (a) is between 20 and 70 percent by weight, but especially between 40 and 60 percent by weight of the total weight of the emulsion.
- the emulsions used according to the invention preferably have an average particle size of at most 1 ⁇ m, in particular of at most 300 nm.
- the used according to the invention Microemulsions preferably have an average particle size of at most 150 nm, in particular of at most 20 nm, on.
- the term "emulsions” also includes microemulsions throughout the text.
- the term "microemulsions” only refers on emulsions with an average particle size of at most 150 nm, which are transparent to optically clear. Microemulsions of organopolysiloxanes with alkyl polyglycosides as emulsifiers are not prescribed.
- Examples of particularly suitable cosurfactants are preferred 1-butanol, 2-butanol, 2-methyl-2-propanol, 1-pentanol, 2-pentanol, 3-pentanol, 1-hexanol, 2-hexanol, 3-hexanol, 1-heptanol, 2-heptanol, 3-heptanol, 4-heptanol, 1-octanol, 2-octanol, 3-octanol and 4-octanol; Diethylene glycol monomethyl, ethyl and butyl ether; Diethylene glycol dimethyl and ethyl ether; 1-aminobutane, 2-aminobutane, 2-amino-2-methyl-propane, 1-aminopentane, 2-aminopentane, 1-aminohexane, 1-aminoheptane and 1-aminooctane; Ethyl, propyl, Isopropyl
- 1-Pentanol and 1-hexanol are particularly preferred as cosurfactants and 1-octanol, diethylene glycol monobutyl ether, diethylene glycol dimethyl ether and butyl acetate.
- Emulsions still contain additives. They are special Bactericide, Fungicide, Algicide, Microbicide, fragrances, Corrosion inhibitors, dyes, pigments, thickeners and fillers.
- the used according to the invention Emulsions preferably contain additives in amounts of 0 up to 1 percent by weight, in particular from 0 to 0.2 percent by weight, each based on the total weight of the finished Emulsion.
- Emulsion can be used in any order of emulsifying devices or by stirring together without being charged high shear forces occur. However, it is preferred that first a homogeneous mixture of organopolysiloxane (a), Alkyl polyglycosides (b) and water produced and in this Mix the cosurfactants and additives, if used, be stirred in without applying high shear forces.
- the one exercised on the respective components or mixtures Pressure is preferably that which may be due to the action of Mixing elements increased (atmospheric) pressure; the corresponding the prevailing temperature is preferably the one where appropriate increased by the action of the mixing elements (Room) temperature.
- ammonium-functional organopolysiloxane (a) can be made by adding mineral acids or carboxylic acids to corresponding amino-functional organopolysiloxanes. This addition of acid to the organopolysiloxane (a) can be done before the organopolysiloxane (a) is used becomes.
- the emulsions used according to the invention are particularly suitable preferred amino and / or ammonium functional organopolysiloxanes (a) the ammonium functional residues however in situ when mixing organopolysiloxane (a), alkyl polyglycosides (b) and water by adding those described above Mineral and / or carboxylic acids, especially acetic acid, generated.
- the emulsions used according to the invention can in principle be made in any turbulent mixer, too has previously been used to prepare emulsions.
- mixers that can be used are stirrers such as blades, bars, Anchor, grid, screw, propeller, disc, impeller, Turbine, planetary stirrers, single and twin screw mixers, Mixing turbines, colloid mills, ultrasonic mixers, in-line mixers, Pumps, homogenizers, such as high pressure, turbine and circulation homogenizers.
- the fabric samples were padded with aqueous emulsions or solutions of the test substances, in the case of silicone oils with Solutions in white spirit, so applied that approx. 1.0% Active substance was applied to tissue weight. Finally was dried at 150 ° C for 5 minutes.
- Samples 7 x 5 cm were sewn together and gassed in a glass tube.
- the concentration of ozone / NO x was approx. 200 mg / h, the flow rate 2.0 l / h.
- the oxidizing agent was generated by electrical discharge in air from an ozone generator so that 0 3 was present in addition to NO x .
- the exposure time was 60 minutes at room temperature. The samples were then rinsed and dried.
- the samples are sewn together into a strip with a Weight (approx. 30 g) weighed down.
- the loss caused by plasticizers according to the invention % of color strength is therefore considerably less than when used conventional silicone emulsions (Example 1) and in same order of magnitude as when using e.g. B. polyethylene emulsions.
- the soft grip caused by aminosilicones is the one obtainable with polyethylene, like any textile specialist common, far superior.
- the activator is a long-known promotional product the pull-out capacity of plasticizer emulsions.
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Molecular Biology (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Detergent Compositions (AREA)
Description
Zwar gibt es Denimartikel in den unterschiedlichsten Farbtönen, die klassische Jeans aber ist blau. "Blue Jeans" ist ein sprachlich fest verankerter Gattungsbegriff. Ebenso traditionell muß die Färbung der Jeans mit Indigo erfolgen. Indigo ist ein Naturfarbstoff, der allerdings heute überwiegend synthetisch hergestellt wird.
Dann wird, nach Einstellung eines optimalen pH, vorzugsweise enzymatisch entschlichtet, d. h. die Stärkeschlichte wird durch die Amylaseenzyme zu löslichen Bruchstücken abgebaut und diese ausgespült. Dadurch verliert das Gewebe wenigstens teilweise seine Starre.
Der heutigen Mode entsprechend wird dann ein beträchtlicher Prozentsatz der aufgebrachten Farbe durch Bleichen entfernt, bis das Kleidungsstück nicht mehr dunkelblau, sondern in definierten mittel- bis hellblauen Tönen erscheint. Dieses Bleichen geschieht vorwiegend durch Chlorierung. Alternative Verfahren, wie z. B. die reduktive Bleiche (Sugar Wash) sind ebenfalls bekannt.
Falls modisch erforderlich, wird die Wäsche mit Lavasteinen durchgeführt (sogenannte "Stone-Wash"-Prozess), was den durch Abrasion geschädigten Hosen den sogenannten "Worn Out Look" verleiht.
Hochwertige Jeans aber werden zusätzlich noch im Spülbad mit Textilweichmacher versetzt, um ihnen einen besonders guten und weichen Griff zu verleihen.
Die Verwendung aminofuktioneller Silicone als Weichmacher wäre an sich erwünscht, da diese Substanzen besonders guten Weichgriff ergeben und dank ihrer kationischen Ladung auch Substantivität zur negativ geladenen Baumwolle aufweisen.
Leider besteht - zu Recht - eine Abneigung gegen Siliconweichmacher bei Jeans:
Im Ladengeschäft liegen Jeanswaren oft längere Zeit vor dem Verkauf. Bei Lagerung im Stapel sind dabei nur die Kanten unmittelbar der Wirkung der Atmosphäre ausgesetzt.
Luftvereinigungen, wie Ozon oder Stickoxide (NOx) aber können den blauen Indigofarbstoff schädigen, d. h. zu gelbem Isathin oxydieren.
Jeans können also, speziell an Lagerkanten, ihre Farbe verlieren. Dieses "Ozonfading" macht, speziell, da es nur örtlich auftritt, diese Waren unverkäuflich.
In der Fachwelt besteht Einigkeit, daß Siliconweichmacher dieses Ozonfading zwar nicht verursachen, wohl aber fördern und verstärken. Daher werden die eigentlich besonders effektiven Siliconweichmacher für diesen Einsatzzweck abgelehnt. Siehe auch Melliand Textilberichte, "Vergilbung von indigohaltiger Jeanswear" 11/96, S. 786f, 1996, wo auf Seite 787 unter 6. Einfluß von Weichgriffmittel und Lagerdauer ausgeführt wird:
"Von den untersuchten Weichgriffmitteln (verschiedene Fettsäurekondensationsprodukte, ein aminofunfuntionelles Polysiloxan und ein Griffmittel auf der Basis von Polyurethan) verursacht keines unter Einwirkung von Schadgas Eigenvergilbung. Siliconhaltige Produkte jedoch beschleunigen den Indigoabbau durch Schadgas. Einzelne Fettsäurekondensationsprodukte vermindern die Vergilbung von Jeanswear deutlich.
Je länger Jeanswear der Einwirkung von Schadgas ausgesetzt ist, desto größer ist die Wahrscheinlichkeit der Entstehung irreparabler Aufhellung und Vergilbung. Beobachtungen aus der Praxis zufolge entsteht ein Vergilbungsmaximum, das unter andauernder Aufhellung stationär ist."
Ethoxylierte Emulgatoren hingegen fördern das Ozonfading.
- R1
- einen zweiwertigen C2- bis C6-Kohlenwasserstoffrest,
- R2
- ein Wasserstoffatom, einen Methyl- oder Cyclohexylrest,
- a
- die Werte 2 oder 3 und
- b
- die Werte 0 oder 1.
wobei
- Ozonkonzentration:
- ca. 200 mg/h
- Durchflußrate:
- 2,0 l/h
- Behandlungsdauer:
- 60 min, bei Raumtemperatur
| Denim behandelt mit | Farbstärkeverlust % 0 3 /NOx | Differenz zum Blindwert |
| Wasser (Blindwert) | 5,3 | 0 |
| Si-Weichmacher 1 | 46,2 | 40,9 |
| Si-Weichmacher 2 | 41,9 | 36,6 |
| Si-Weichmacher 3 | 38,2 | 32,9 |
| Si-Weichmacher 4 | 38,3 | 33 |
| Si-Weichmacher 5 | 54,8 | 49,5 |
| Si-Weichmacher 6 | 48,4 | 43,1 |
| Si-Weichmacher 7 | 39,2 | 33,9 |
| PE-Emulsion (Velustrol P 40, Hoechst AG) | 16,4 | 11,1 |
| PE-Emulsion ( Adalin NI, Henkel) | 16,5 | 11,2 |
| kation. Fettweichmacher (quaternisiertes Talkfettsäureamin) (Leomin AFK, Hoechst) | 30,5 | 25,2 |
| PE = Polyethylen |
Microemulsion eines niedrigviskosen, gestopperten Siliconöles mit Aminoethyl-aminopropylgruppen; Ölviskosiät 200 mm2/s, Aminzahl 0,25; emulgiert mit Trimethylnonanol x 6 EO
Macroemulsion eines hochviskosen, niedrigaminierten, reaktiven Öls; Ölviskosität 8000 mm2/s; Aminzahl 0,15; emulgiert mit iso-C13 Fettalkohol x 10 EO
Macroemulsion des gleichen Öles wie Weichmacher 2, emulgiert mit Alkyltrimethylammoniumchlorid und Nonylphenol x 7 EO
Macroemulsion eines reaktiven aminreichen Siliconöles; ölviskosität 1000 mm2/s, Aminzahl 0,6; emulgiert mit einem Gemisch aus C13-Fettalkohol x 6 EO und C13-Fettalkohol x 8 EO
Microemulsion eines gestopperten, aminreichen öles; Ölsivkosität 1000 mm2/s, Aminzahl 0,6; emulgiert mit C13-Fettalkohol x 6 EO
Microemulsion eines reaktiven Öles mittleren Amingehalts; ölviskosität 1000 mm2/s; Aminzahl 0,3; emulgiert mit C13-Fettalkohol x 6 EO
Microemulsion eines cyclohexylaminofunktionellen (thermovergilbungsarmen) Siliconöls; Ölviskosität 1000 mm2/ s; Aminzahl 0,3; emulgiert mit C13-Fettalkohol x 6 EO mit Butyldiglykol als Co-Emulgator
| Denim behandelt mit | Farbstärkeverlust % 0 3 /NOx | Differenz zum Blindwert |
| Wasser (Blindwert) | 5,3 | 0.00 |
| Testbenzin (Blindwert) | 4,5 | 0.00 |
| Aminosiliconöl 1 | 10 | 5,5 |
| Aminosiliconöl 2 | 8,7 | 4,2 |
| Aminosiliconöl 3 | 19,8 | 15,3 |
| Aminosiliconöl 4 | 5,4 | 0,9 |
| C 13 -Fettalkohol x 10 EO (Arlypon IT 10/Grünau) | 51 | 45,7 |
| C 13 -Fettalkohol x 6 EO (Genapol X060, Hoechst) | 34,8 | 29,5 |
| C 13 -Fettalkohol x 8 EO (Genapol X080, Hoechst) | 44,8 | 39,5 |
| Nonylphenolethoxylat (Marlophen NP7, Hüls) | 54,3 | 49 |
| Trimethylnonanol x 6 EO (Tergitol TMN6, ICI) | 38,2 | 32,9 |
| Hexadecyltrimethylammoni-umchlorid ( Genamin CTAC 50, Hoe) | 37,4 | 32,1 |
| Mischemulgator von Silicon-Weichmacher 3 | 37,6 | 32,3 |
17 Teile eines gestopperten hochaminierten Aminosiliconöles (Aminzahl = 0,6; Viscosität = 1000 mm2/s) wurden mit 8,6 Teilen Alkylplyglycosid (70%ig), 74 Teilen vollentsalztem Wasser und 0,3 Teilen Essigsäure 100 %ig zu einer Emulsion verrührt und mit 3,5 g/L Formalinlösung konserviert.
Das Öl war identisch mit dem im Weichmacher 5 verwendeten Produkt.
Als Emulgator diente Glucopon 225 DK (Henkel), ein C8 - C10 Alkylplyglycosid mit 1,7 Zuckereinheiten und 70 % Aktivgehalt.
Analog wurde aus dem auch in Weichmacher 4 eingesetzten reaktiven Öl nach Rezeptur von Weichmacher 8 eine Emulsion bereitet.
Das bereits in den Weichmachern 2 + 3 eingesetzte aminarme, hochviskose Siliconöl wurde zu 17 Teilen mit 9,5 Teilen Glucopon 215 CSUP (Henkel) und 0,08 Teilen Essigsäure emulgiert und mit 3,5 g/L Formalinlösung konserviert.
Glucopon 215 CSUP ist ein C8 - C10 Alkylpolyglykosid mit 1,5 Zuckereinheiten.
17 Teile des bereits in Weichmacher 6 benutzten Aminöles der Aminzahl 0,3 wurden mit 8,6 Teilen Glucopon 225 DK wie bei Weichmacher 8 beschrieben emulgiert, jedoch mit einer auf 0,15 Teile reduzierte Menge an Essigsäure.
| Denim behandelt mit | Farbstärkeverlust % 0 3 /NOx | Differenz zum Blindwert |
| Wasser (Blindwert) | 7 | 0.00 |
| Weichmacher 8 | 20,2 | 13,2 |
| Weichmacher 9 | 26 | 19 |
| Weichmacher 10 | 21 | 14 |
| Weichmacher 11 | 18,8 | 11,8 |
In der industriellen Praxis aber wird der Weichmacher in der Waschmaschine im Ausziehverfahren appliziert.
Claims (5)
- Indigo gefärbtes Gewebe, dadurch gekennzeichnet, daß es zumindest ein Organopolysiloxan und zumindest ein Alkylpolyglykosid enthält.
- Indigo gefärbtes Gewebe nach Anspruch 1, dadurch gekennzeichnet, daß das Organopolysiloxan ein aminofunktionelles Organopolysiloxan ist.
- Indigo gefärbtes Gewebe nach Anspruch 1 oder 2, dadurch gekennzeichnet, daß das Gewichtsverhältnis Organopolysiloxan zu dem Alkylpolyglykosid 10 : 1 bis 0,5 : 1 beträgt.
- Verfahren zur Behandlung eines mit Indigo gefärbten Gewebes nach einem oder mehreren der Ansprüch 1 bis 3, dadurch gekennzeichnet, daß zumindest ein Organopolysiloxan und zumindest ein Alkylpolyglykosid aufgebracht werden.
- Verfahren zur Behandlung eines mit Indigo gefärbten Gewebes nach Anspruch 4, dadurch gekennzeichnet, daß zumindest ein Organopolysiloxan und zumindest ein Alkylpolyglykosid in Form einer Emulsion aufgebracht werden.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19722680 | 1997-05-30 | ||
| DE19722680A DE19722680A1 (de) | 1997-05-30 | 1997-05-30 | Siliconweichmacher für Jeans |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0881325A1 EP0881325A1 (de) | 1998-12-02 |
| EP0881325B1 true EP0881325B1 (de) | 1999-09-29 |
Family
ID=7830941
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP98109267A Expired - Lifetime EP0881325B1 (de) | 1997-05-30 | 1998-05-22 | Siliconweichmacher für Jeans |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US6042615A (de) |
| EP (1) | EP0881325B1 (de) |
| JP (1) | JP2868763B2 (de) |
| BR (1) | BR9801560A (de) |
| DE (2) | DE19722680A1 (de) |
| ES (1) | ES2139467T3 (de) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19915439A1 (de) * | 1999-04-07 | 2000-10-12 | Cognis Deutschland Gmbh | Verfahren zur Stabilisierung von gefärbtem Textilgut gegen Vergilben |
| DE10005855A1 (de) * | 2000-02-10 | 2001-08-23 | Wacker Chemie Gmbh | Flächengebilde aus Polyester |
| DE10034831A1 (de) * | 2000-07-18 | 2002-01-31 | Ciba Sc Pfersee Gmbh | Gemische von Polysiloxanemulsionen |
| DE10040631A1 (de) * | 2000-08-16 | 2002-03-07 | Wolfram Koch | Vergilbungshemmer für Blue Denim Textilien |
| US6632385B2 (en) | 2001-03-23 | 2003-10-14 | First Quality Nonwovens, Inc. | Condrapable hydrophobic nonwoven web and method of making same |
| JP5346586B2 (ja) * | 2005-12-16 | 2013-11-20 | サザンミルズ インコーポレイテッド | 耐熱性を提供する防護服 |
| WO2016032996A1 (en) * | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Method of preparing a detergent composition |
| CN110698691A (zh) * | 2019-11-06 | 2020-01-17 | 常州市宁河新材料科技有限公司 | 一种疏水型氨基硅油乳液的制作方法 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5114426A (en) * | 1988-12-28 | 1992-05-19 | Atochem North America, Inc. | Chemical stonewash methods for treating fabrics |
| DE3925846A1 (de) * | 1989-08-04 | 1991-02-14 | Huels Chemische Werke Ag | Emulgatoren zur herstellung von lagerstabilen, waessrigen polysiloxan- bzw. polysiloxan-paraffinoel-emulsionen |
| DE4131551A1 (de) * | 1991-09-21 | 1993-03-25 | Pfersee Chem Fab | Waessrige dispersionen von stickstoffhaltigen polysiloxanen |
| US5254269A (en) * | 1991-11-26 | 1993-10-19 | Lever Brothers Company, Division Of Conopco, Inc. | Fabric conditioning composition containing an emulsified silicone mixture |
| DE4306796A1 (de) * | 1993-03-04 | 1994-09-08 | Wacker Chemie Gmbh | Emulsionen polare Gruppen enthaltender Organopolysiloxane mit Alkylpolyglykosiden als Emulgatoren |
| US5480567A (en) * | 1994-01-14 | 1996-01-02 | Lever Brothers Company, Division Of Conopco, Inc. | Surfactant mixtures for fabric conditioning compositions |
| DE19603401A1 (de) * | 1996-01-31 | 1997-08-07 | Basf Ag | Verfahren zur Stabilisierung von konfektioniertem Textilgut, das mit verküpbaren Farbstoffen gefärbt ist, gegen Vergilbung |
-
1997
- 1997-05-30 DE DE19722680A patent/DE19722680A1/de not_active Withdrawn
-
1998
- 1998-05-22 EP EP98109267A patent/EP0881325B1/de not_active Expired - Lifetime
- 1998-05-22 DE DE59800032T patent/DE59800032D1/de not_active Expired - Fee Related
- 1998-05-22 ES ES98109267T patent/ES2139467T3/es not_active Expired - Lifetime
- 1998-05-27 US US09/085,744 patent/US6042615A/en not_active Expired - Fee Related
- 1998-05-28 JP JP10147251A patent/JP2868763B2/ja not_active Expired - Fee Related
- 1998-05-29 BR BR9801560-5A patent/BR9801560A/pt not_active Application Discontinuation
Also Published As
| Publication number | Publication date |
|---|---|
| JPH10331074A (ja) | 1998-12-15 |
| JP2868763B2 (ja) | 1999-03-10 |
| DE19722680A1 (de) | 1998-12-03 |
| US6042615A (en) | 2000-03-28 |
| EP0881325A1 (de) | 1998-12-02 |
| BR9801560A (pt) | 1999-12-14 |
| DE59800032D1 (de) | 1999-11-04 |
| ES2139467T3 (es) | 2000-02-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE667744C (de) | Verfahren zur Herstellung von in Wasser loeslichen oder leicht verteilbaren Kondensationsprodukten | |
| DE69220936T2 (de) | Entfernung überschüssigen farbstoffes von neuen textilien | |
| EP0622397B1 (de) | Emulsionen polare Gruppen enthaltender Organopolysiloxane mit Alkylpolyglykosiden als Emulgatoren | |
| DE3519601C2 (de) | ||
| EP0881325B1 (de) | Siliconweichmacher für Jeans | |
| EP0628655B1 (de) | Bleichhilfsmittel | |
| DE2641263A1 (de) | Detachiermittel und verfahren zum reinigen und gegebenenfalls faerben von textilmaterialien | |
| DE102016218163A1 (de) | Verbesserte Färbung von Textilien mit synthetischen Materialien in der Waschmaschine durch modifizierte Silikone | |
| DE3932276C2 (de) | Textilbehandlungsmittel und dessen Verwendung | |
| CH689150B5 (de) | Textilveredlungsmittel. | |
| DE4407801A1 (de) | Behandlung von Textilien | |
| CN107858843B (zh) | 一种含壳聚糖季铵盐的纺织品固色剂及其制备方法 | |
| DE10259291B4 (de) | Hochkonzentrierte, selbstemulgierende Zubereitungen, enthaltend Organopolysiloxane und Alkylammoniumverbindungen und deren Verwendung in wässrigen Systemen | |
| KR20000077318A (ko) | 실록산 유액 | |
| DE19652524C2 (de) | Organopolysiloxane enthaltende Emulsionen, deren Herstellung und Verwendung in wäßrigen Systemen | |
| GB2492641A (en) | Process for inkjet printing of textiles | |
| EP1498541A2 (de) | Verfahren zur Herstellung von Baumwollkettgarnen mit "Invers Denim" Effekt | |
| EP0978586A2 (de) | Wässrige Mikroemulsionen, enthaltend Organopolysiloxane | |
| DE3137044A1 (de) | Imidazolindervate | |
| DE60127117T2 (de) | Farbwiedergewinnungsmittel | |
| EP1127975A1 (de) | Flächengebilde aus Polyester | |
| DE349655C (de) | Verfahren, um Textilfasern aller Art von ihren staerkeartigen, gummiartigen, gelatineartigen und fetten, von der Appretur oder Versteifung u. dgl. herstammenden Stoffen mit Hilfe von Bakterien zu befreien | |
| MXPA98004314A (en) | Silicon softener for je | |
| DE19614628A1 (de) | Vorbehandlung von Textilien | |
| JP2004218180A (ja) | 綿繊維の連続酵素精練方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| 17P | Request for examination filed |
Effective date: 19980522 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): BE CH DE ES FR GB IT LI NL |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| 17Q | First examination report despatched |
Effective date: 19981204 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| AKX | Designation fees paid |
Free format text: BE CH DE ES FR GB IT LI NL |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE CH DE ES FR GB IT LI NL |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REF | Corresponds to: |
Ref document number: 59800032 Country of ref document: DE Date of ref document: 19991104 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19991108 |
|
| ITF | It: translation for a ep patent filed | ||
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2139467 Country of ref document: ES Kind code of ref document: T3 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20050429 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20050517 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20050518 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20050520 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20050609 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20050624 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20050630 Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060522 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060523 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060531 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060531 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060531 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20060531 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20061201 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20061201 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20060522 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 20061201 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20070131 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20060523 |
|
| BERE | Be: lapsed |
Owner name: *WACKER-CHEMIE G.M.B.H. Effective date: 20060531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20070522 |