EP0564404B1 - Procédé de teinture du cuir avec des mélanges de colorants - Google Patents
Procédé de teinture du cuir avec des mélanges de colorants Download PDFInfo
- Publication number
- EP0564404B1 EP0564404B1 EP93810208A EP93810208A EP0564404B1 EP 0564404 B1 EP0564404 B1 EP 0564404B1 EP 93810208 A EP93810208 A EP 93810208A EP 93810208 A EP93810208 A EP 93810208A EP 0564404 B1 EP0564404 B1 EP 0564404B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- formula
- dye
- process according
- hydrogen
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 Cc(c(CNC(*)=O)c1C)cc(C)c1Nc(cc(*)c(N)c1C(c2ccccc22)=O)c1C2=O Chemical compound Cc(c(CNC(*)=O)c1C)cc(C)c1Nc(cc(*)c(N)c1C(c2ccccc22)=O)c1C2=O 0.000 description 6
- OXNVYWGBVGZKAH-RQZHXJHFSA-N Nc(ccc1c2c(O)cc(S(O)(=O)=O)c1)c2/N=N\c(cc(cc1)Cl)c1Oc1ccccc1Cl Chemical compound Nc(ccc1c2c(O)cc(S(O)(=O)=O)c1)c2/N=N\c(cc(cc1)Cl)c1Oc1ccccc1Cl OXNVYWGBVGZKAH-RQZHXJHFSA-N 0.000 description 1
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P3/00—Special processes of dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form, classified according to the material treated
- D06P3/02—Material containing basic nitrogen
- D06P3/04—Material containing basic nitrogen containing amide groups
- D06P3/32—Material containing basic nitrogen containing amide groups leather skins
- D06P3/3206—Material containing basic nitrogen containing amide groups leather skins using acid dyes
- D06P3/3213—Material containing basic nitrogen containing amide groups leather skins using acid dyes monoazo
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0046—Mixtures of two or more azo dyes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P3/00—Special processes of dyeing or printing textiles, or dyeing leather, furs, or solid macromolecular substances in any form, classified according to the material treated
- D06P3/02—Material containing basic nitrogen
- D06P3/04—Material containing basic nitrogen containing amide groups
- D06P3/32—Material containing basic nitrogen containing amide groups leather skins
- D06P3/3206—Material containing basic nitrogen containing amide groups leather skins using acid dyes
- D06P3/3226—Material containing basic nitrogen containing amide groups leather skins using acid dyes dis-polyazo
Definitions
- the present invention relates to a method for dyeing leather with dye mixtures, in particular a method for trichromatic dyeing of leather, and to the leather dyed by the method.
- the present invention had for its object to provide a method for dyeing leather with dye mixtures, according to which dyeing in brilliant shades is possible.
- Particularly suitable dye mixtures should also enable the trichromatic dyeing of leather in brilliant shades.
- the invention thus relates to a process for dyeing leather with dye mixtures, which is characterized in that at least two metal-free dyes each having a single sulfo or carboxy group and a molecular weight between 450 and 700 are used.
- the dyes of formulas (1), (2) and (3) each have a single sulfo group.
- Halogen means fluorine, bromine, iodine or especially chlorine.
- a dye mixture is preferably used which contains a yellow dye of the formula (1), a red dye of the formula (2) and a blue dye of the formula (3).
- R 1 is preferably methyl.
- R 3 preferably denotes chlorine and R 4 preferably denotes sulfo or hydrogen.
- R 5 are sulfo or a group or the phenyl radicals in the latter two groups being unsubstituted or substituted by chlorine.
- R 6 are hydrogen, chlorine or a group wherein R 7 is hydrogen, methyl or ethyl, R 1 is methyl and n is 0, 1 or 2.
- a particularly preferred embodiment of the process according to the invention consists in that the yellow dye of the formula used.
- a further preferred embodiment of the process according to the invention is characterized in that a red dye of the formula (2) is used in which R 8 sulfo or means, wherein R 7 is methyl or ethyl.
- a very particularly preferred embodiment of the process according to the invention consists in that the red dye of the formula used.
- a further preferred embodiment of the process according to the invention is characterized in that a blue dye of the formula (3) is used in which R 14 is methyl and R 12 and R 13 are hydrogen or methyl.
- dyes of the formula (3) in which R 15 is hydrogen, methyl or -CH 2 -NH-CO-CH 2 -Cl preference is given to using dyes of the formula (3) in which R 15 is hydrogen, methyl or -CH 2 -NH-CO-CH 2 -Cl, and also dyes of the formula (3) in which R 16 is -SO 2 - NH-C 2 H 4 -OH, -CH 2 -NH-CO-CH 2 -Cl or -CH 2 -NH-CO-C 6 H 5 means.
- a very particularly preferred embodiment of the process according to the invention consists in that the blue dye of the formula used.
- the parachor of which is between 900 and 1200 and the log P of which is between 1 and 4.
- the Parachor is used according to the article by OR Quayle [Chem. Rev. 53 439 (1953)] and log P means the relative lipophilicity, the calculation of which by C. Hansch et al. [J. Med. Chem. 16 , 1207 (1973)].
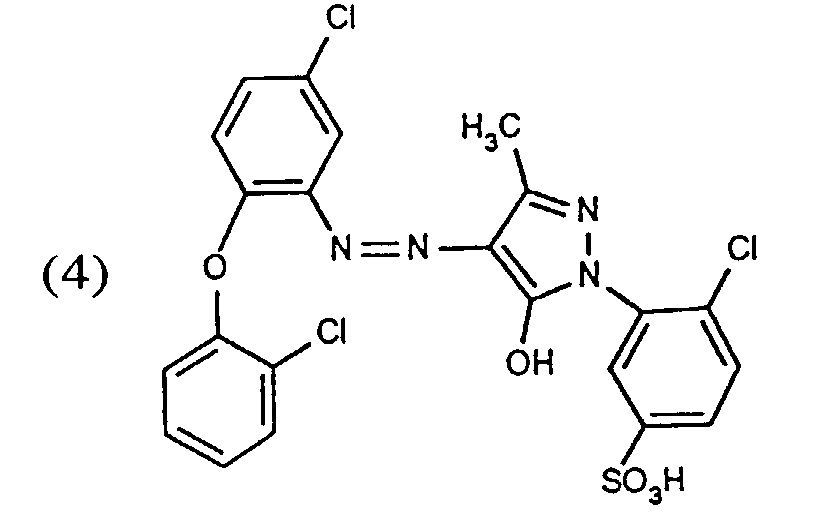
- a very particularly important embodiment of the process according to the invention consists in using a dye mixture containing the yellow dye of the formula (4), the red dye of the formula (5) and the blue dye of the formula (6) for trichromatic dyeing.
- dyes of the formulas (2), (3) or (4) can be used in the dyeing process according to the invention. This can e.g. be useful if the shade should be cloudy or darker or in a slightly changed color than is achievable with the dyes mentioned.
- Preferably used as further dyes are those which likewise have a sulfo or carboxy group and a molecular weight between 450 and 700.
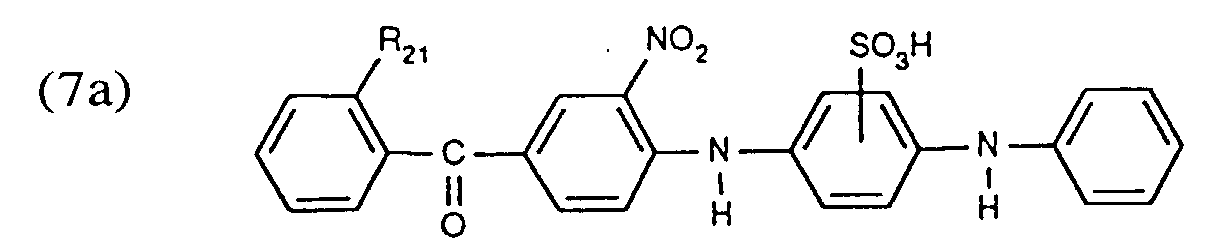
- a black dye of the formula is used above all as an additional dye where R 19 is hydrogen, C 1 -C 4 alkyl, C 1 -C 4 alkoxy or C 1 -C 4 alkanoylamino and R 20 is chlorine or nitro, or a brown dye of the formula wherein R 21 is -COOH, -CONH 2 , -SO 3 H or -SO 3 NH 2 .
- the black dye of the formula is preferably used as an additional black dye and the brown dye of the formula is preferably used as the additional brown dye
- the dyes according to the invention are anionic in nature and are advantageously used in the form of their salts, in particular alkali metal salts or ammonium salts.
- the alkali salts are in particular the lithium, potassium or sodium salts and ammonium salts are also understood to mean salts of organic amines, for example those which are substituted by C 1 -C 6 -alkyl or hydroxy-C 1 -C 6 -alkyl groups are.
- the dyes used in the process according to the invention are known or can be are produced in a manner known per se.
- the amounts in which the dyes used according to the invention are used in the dye baths can vary within wide limits depending on the desired depth of color. In general, amounts of dye of 0.1 to 10, preferably 0.5 to 4,% by weight, based on the weight of the leather to be dyed, are advantageous.
- the dye liquor can also contain other additives, e.g. Acids, bases or salts to adjust the desired pH value, as well as auxiliaries such as Wetting agents, greasing agents, color-deepening aids, leveling agents, penetrants and / or anti-foaming agents.
- additives e.g. Acids, bases or salts to adjust the desired pH value, as well as auxiliaries such as Wetting agents, greasing agents, color-deepening aids, leveling agents, penetrants and / or anti-foaming agents.
- the method according to the invention has the great advantage that it is not only suitable for a specific type of leather, but can also be used for the various types of leather, for example chrome leather, retanned leather or suede from goat, cow or pig.
- the dyeing is preferably carried out by the exhaust process, e.g. at a liquor ratio of 1: 1.5 to 1:20, preferably 1: 2 to 1:10, and at temperatures of 20 to 100 ° C, preferably 30 to 60 ° C.
- the leather can be pretreated be subjected, for example, to a neutralization or roll.
- the dyeing time varies depending on the type of leather and the desired depth of color, but is generally between 30 and 180 minutes. After the dyeing, the leather is rinsed and finished as usual.
- the process according to the invention gives leather dyeings in brilliant shades and with good fastness properties, such as Light and wet fastness properties. Since the individual dyes penetrate approximately the same depth into the leather, the nuance of the colors on the surface and in the deeper layers of the leather is the same.
- the individual dyes are mixed with one another for use in dyeing.
- the dyes can be prepared individually as stock solutions for the dyeing process or formulated together in a single stock solution.
- the dye trichromatic mixtures containing at least one yellow dye of the formula (1), at least one red dye of the formula (2) and at least one blue dye of the formula (3) are known from EP-A-0 181 292.
- Preferred dye trichromatic mixtures contain 1 to 98% by weight of a yellow dye of the formula (1), 1 to 98% by weight of a red dye of the formula (2) and 1 to 98% by weight of a blue dye of the formula (3) , in particular 10 to 70% by weight of a yellow dye of the formula (1), 10 to 50% by weight of a red dye of the formula (2) and 10 to 50% by weight of a blue dye of the formula (3).
Claims (24)
- Procédé pour la teinture du cuir avec des mélanges de colorants, caractérisé en ce que l'on utilisé au moins deux colorants exempts de métaux comportant chacun un seul groupe sulfo et ayant une masse moléculaire comprise entre 450 et 700, choisis dans le groupe formé para) un colorant jaune de formule
- Procédé conforme à la revendication 1, caractérisé en ce que l'on utilise pour la teinture en trichromie un mélange de colorant contenant un colorant jaune de formule (1), un colorant rouge de formule (2) et un colorant bleu de formule (3).
- Procédé conforme aux revendications 1 et 2, caractérisé en ce que l'on utilise un colorant jaune de formule (1) dans lequel R1 est un résidu méthyle.
- Procédé conforme à une des revendications 1 à 3, caractérisé en ce que l'on utilise un colorant j aune de formule (1) dans lequel R3 représente un atome de chlore.
- Procédé conforme à une des revendications 1 à 4, caractérisé en ce que l'on utilise un colorant jaune de formule (1) dans lequel R4 est un groupe sulfo ou un atome d'hydrogène.
- Procédé conforme à une des revendications 1 à 5, caractérisé en ce que l'on utilise un colorant j aune de formule (1) dans lequel R5 est un groupe sulfo ou un groupe O-phényle ou -SO2-O-phényle, les résidus phényle de ces deux derniers groupes pouvant être non substitués ou chlorés.
- Procédé conforme à une des revendications 1 à 6, caractérisé en ce que l'on utilise un colorant jaune de formule (1) dans lequel R6 est un atome d'hydrogène ou de chlore ou un groupe
- Procédé conforme à une des revendications 1 à 10, caractérisé en ce que l'on utilise un colorant rouge de formule (2) dans lequel R10 est un atome d'hydrogène ou un groupe chlorométhylcarbonylamino.
- Procédé conforme à une des revendications 1 à 11, caractérisé en ce que l'on utilise un colorant rouge de formule (2) dans lequel R11 est un atome d'hydrogène ou de chlore.
- Procédé conforme à une des revendications 1 à 13, caractérisé en ce que l'on utilise un colorant bleu de formule (3) dans lequel R14 représente un groupe méthyle et R12 et R13 représentent un atome d'hydrogène ou un groupe méthyle.
- Procédé conforme à une des revendications 1 à 14, caractérisé en ce que l'on utilise un colorant bleu de formule (3) dans lequel R15 représente un atome d'hydrogène, un groupe méthyle ou -CH2-NH-CO-CH2-Cl.
- Procédé conforme à une des revendications 1 à 15, caractérisé en ce que l'on utilise un colorant bleu de formule (3) dans lequel R16 représente un groupe -SO2-NH-C2H4-OH, -CH2-NH-CO-CH2-Cl ou -CH2-NHCO-C6H5.
- Procédé conforme à une des revendications 1 à 17, caractérisé en ce que l'on utilise au moins deux colorants ayant un parachor compris entre 900 et 1200 et dont la valeur de log P est comprise entre 1 et 4.
- Procédé conforme à une des revendications 1 à 18, caractérisé en ce que l'on utilise en plus un colorant noir de formule
- Procédé conforme aux revendications 1, 2, 8, 13 et 17, caractérisé en ce que l'on utilise pour la teinture en trichromie un mélange de colorants contenant le colorant jaune de formule (4), le colorant rouge de formule (5) et le colorant bleu de formule (6).
- Procédé conforme à une des revendications 1 à 21, caractérisé en ce que l'on utilise un bain de teinture contenant en plus un agent de renforcement de la couleur.
- Procédé conforme à la revendication 22, caractérisé en ce que l'on utilise un bain de teinture contenant, comme agent de renforcement de la couleur, une alkylamine éthoxylée.
- Cuir teint selon le procédé conforme aux revendications 1 à 23.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH105392 | 1992-04-01 | ||
| CH1053/92 | 1992-04-01 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0564404A1 EP0564404A1 (fr) | 1993-10-06 |
| EP0564404B1 true EP0564404B1 (fr) | 1997-06-25 |
Family
ID=4201357
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP93810208A Expired - Lifetime EP0564404B1 (fr) | 1992-04-01 | 1993-03-23 | Procédé de teinture du cuir avec des mélanges de colorants |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US5354341A (fr) |
| EP (1) | EP0564404B1 (fr) |
| JP (1) | JPH0649778A (fr) |
| DE (1) | DE59306811D1 (fr) |
| ES (1) | ES2105192T3 (fr) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19851389A1 (de) * | 1998-11-07 | 2000-05-11 | Dystar Textilfarben Gmbh & Co | Gelbe Farbstoffmischungen von wasserlöslichen faserreaktiven Azofarbstoffen und ihre Verwendung |
| US7001435B2 (en) * | 2002-05-30 | 2006-02-21 | Automotive Technology, Inc. | Leather treatment |
| AU2011248537B2 (en) | 2010-04-28 | 2014-04-17 | University Of Georgia Research Foundation, Inc. | Photochemical cross-linkable polymers, methods of marking photochemical cross-linkable polymers, methods of using photochemical cross-linkable polymers, and methods of making articles containing photochemical cross-linkable polymers |
| US9315628B2 (en) | 2011-07-15 | 2016-04-19 | University Of Georgia Research Foundation, Inc. | Permanent attachment of agents to surfaces containing C-H functionality |
| US9839213B2 (en) | 2011-10-14 | 2017-12-12 | The University Of Georgia Research Foundation, Inc. | Photochemical cross-linkable polymers, methods of making photochemical cross-linkable polymers, methods of using photochemical cross-linkable polymers, and methods of making articles containing photochemical cross-linkable polymers |
| CN103275516B (zh) * | 2013-06-06 | 2016-05-11 | 恒升化工(启东)有限公司 | 一种含活性基团的酸性黄色染料的生产方法 |
| CN114575171A (zh) * | 2021-12-22 | 2022-06-03 | 浙江中辉裘革科技有限公司 | 一种羊皮低温染色用染料及其染色工艺 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2226336A (en) * | 1988-12-22 | 1990-06-27 | Sandoz Ltd | Trichromatic reactive dye mixture |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH191165A (de) * | 1936-03-05 | 1937-05-31 | Geigy Ag J R | Verfahren zur Darstellung eines neuen Monoazofarbstoffes. |
| US3819690A (en) * | 1971-04-19 | 1974-06-25 | Ciba Geigy Ag | Acid nitro dyestuffs,their manufacture and use |
| JPS4996010A (fr) * | 1972-10-03 | 1974-09-11 | ||
| LU78597A1 (de) * | 1977-11-28 | 1979-06-13 | Ciba Geigy Ag | Neue anthrachinonverbindungen,deren herstellung und verwendung |
| EP0061670B2 (fr) * | 1981-03-23 | 1994-06-01 | Ciba-Geigy Ag | Procédé de teinture de cuir et de peaux avec des mélanges de colorants |
| EP0092512B1 (fr) * | 1982-04-08 | 1986-04-30 | Ciba-Geigy Ag | Procédé de teinture ou d'impression en trois couleurs |
| DE3326954A1 (de) * | 1982-08-06 | 1984-02-09 | Sandoz-Patent-GmbH, 7850 Lörrach | Anionische monoazoverbindungen, verfahren zur herstellung und ihre verwendung bei faerbe- und druckverfahren |
| CH654845A5 (de) * | 1982-08-06 | 1986-03-14 | Sandoz Ag | Anionische monoazoverbindungen, verfahren zur herstellung und ihre verwendung bei faerbe- und druckverfahren. |
| EP0127579B1 (fr) * | 1983-05-25 | 1987-10-07 | Ciba-Geigy Ag | Procédé de teinture ou d'impression en trois couleurs |
| DE3578870D1 (de) * | 1984-11-08 | 1990-08-30 | Ciba Geigy Ag | Verfahren zum kontinuierlichen trichromie-faerben von synthetischen polyamidmaterialien. |
| US4652269A (en) * | 1984-11-08 | 1987-03-24 | Ciba-Geigy Corporation | Process for the continuous trichromatic dyeing of synthetic polyamide materials: including a blue anthraquinone reactive dye |
| US4799933A (en) * | 1986-01-21 | 1989-01-24 | Ciba-Geigy Corporation | Process for the trichromatic dyeing of leather |
| GB2236542B (en) * | 1989-10-06 | 1992-04-15 | Sandoz Ltd | Dye mixtures and their use in trichromatic dyeing processes |
| EP0425435A1 (fr) * | 1989-10-23 | 1991-05-02 | Ciba-Geigy Ag | Procédé de teinture ou d'impression trichromique |
-
1993
- 1993-03-23 EP EP93810208A patent/EP0564404B1/fr not_active Expired - Lifetime
- 1993-03-23 DE DE59306811T patent/DE59306811D1/de not_active Expired - Fee Related
- 1993-03-23 ES ES93810208T patent/ES2105192T3/es not_active Expired - Lifetime
- 1993-03-26 US US08/037,194 patent/US5354341A/en not_active Expired - Fee Related
- 1993-04-01 JP JP5074906A patent/JPH0649778A/ja not_active Withdrawn
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2226336A (en) * | 1988-12-22 | 1990-06-27 | Sandoz Ltd | Trichromatic reactive dye mixture |
Also Published As
| Publication number | Publication date |
|---|---|
| DE59306811D1 (de) | 1997-07-31 |
| EP0564404A1 (fr) | 1993-10-06 |
| ES2105192T3 (es) | 1997-10-16 |
| US5354341A (en) | 1994-10-11 |
| JPH0649778A (ja) | 1994-02-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0501197A1 (fr) | Procédé de teinture pour cuir aves des colorants au soufre insolubles dans l'eau | |
| EP0061670B1 (fr) | Procédé de teinture de cuir et de peaux avec des mélanges de colorants | |
| EP0564404B1 (fr) | Procédé de teinture du cuir avec des mélanges de colorants | |
| DE2818653C2 (fr) | ||
| EP0577556B1 (fr) | Procédé pour teindre du cuir par trichromie avec des mélanges de colorants | |
| EP0558450B1 (fr) | Procédé pour teindre du cuir avec des mélanges de colorants | |
| DE2835035B2 (de) | Verfahren zum Färben von Cellulosefasern mit Reaktivfarbstoffen nach der Ausziehmethode | |
| DE1444281A1 (de) | Verfahren zum Faerben von geformten Gebilden aus Polyurethan mit wasserloeslichen Farbstoffen | |
| EP0147783A2 (fr) | Mélanges de colorants azoiques dispersés bleus pour la teinture de fibres synthétiques | |
| EP0531968A1 (fr) | Mélange de colorants réactifs solubles dans l'eau et son utilisation | |
| EP0905201B1 (fr) | Mélanges de colorants réactifs solubles dans l'eau et leur utilisation en teinture | |
| EP0548014A1 (fr) | Mélanges de colorants et leur utilisation | |
| EP0024014A1 (fr) | Procédé de retannage de cuirs tannés au minéral avec des acides sulfoniques aromatiques | |
| EP0648816B1 (fr) | Teinture du cuir avec des mélanges de colorants | |
| EP0222269B1 (fr) | Procédé de teinture de tissu de laine en pièce | |
| EP0059876A1 (fr) | Procédé pour colorer des matériaux en mélanges, en fibres de polyester et en fibres kératiniques | |
| EP0341206A1 (fr) | Colorants formazane réactifs avec les fibres | |
| EP0651028A1 (fr) | Procédé de teinture ou d'impression en trichromie | |
| EP0497238B1 (fr) | Procédé de teinture du cuir | |
| DE2638236A1 (de) | Verfahren zum faerben von leder durch gleichzeitige anwendung saurer und basischer farbstoffe | |
| DE874759C (de) | Verfahren zum Faerben von Fasermaterial mit Kuepenfarbstoffen | |
| EP0264346A1 (fr) | Procédé de teinture de matières fibreuses en polyamides naturels ou synthétiques avec des colorants à complexes métallifères 1:1 | |
| DE3427806C2 (fr) | ||
| EP0099336B1 (fr) | Complexes de cobalt 1:2 de colorants azoiques | |
| DE1283801B (de) | Verfahren zur Fertigstellung von Faerbungen und Drucken auf Polyamidfasern |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): BE CH DE ES FR GB IT LI NL |
|
| 17P | Request for examination filed |
Effective date: 19940316 |
|
| 17Q | First examination report despatched |
Effective date: 19941219 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: CIBA SC HOLDING AG |
|
| RAP3 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: CIBA SPECIALTY CHEMICALS HOLDING INC. |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE CH DE ES FR GB IT LI NL |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| ET | Fr: translation filed | ||
| REF | Corresponds to: |
Ref document number: 59306811 Country of ref document: DE Date of ref document: 19970731 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19970909 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2105192 Country of ref document: ES Kind code of ref document: T3 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19980129 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19980212 Year of fee payment: 6 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19980518 Year of fee payment: 6 |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 19990201 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19990217 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 19990316 Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990331 |
|
| BERE | Be: lapsed |
Owner name: CIBA SPECIALTY CHEMICALS HOLDING INC. Effective date: 19990331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19991001 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19991130 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 19991001 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000323 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000324 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000331 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000331 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20000323 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20010910 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050323 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20070331 Year of fee payment: 15 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20081001 |