EP0427670A1 - Liquid detergent - Google Patents
Liquid detergent Download PDFInfo
- Publication number
- EP0427670A1 EP0427670A1 EP90810829A EP90810829A EP0427670A1 EP 0427670 A1 EP0427670 A1 EP 0427670A1 EP 90810829 A EP90810829 A EP 90810829A EP 90810829 A EP90810829 A EP 90810829A EP 0427670 A1 EP0427670 A1 EP 0427670A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- liquid detergent
- weight
- detergent
- detergent according
- optical brightener
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 0 *C=II=*c(cc1)cc(C=Cc(cc2)ccc2-c2ccc(C=C[C@]3C(Cl)=CC=C(*)C3)cc2)c1Cl Chemical compound *C=II=*c(cc1)cc(C=Cc(cc2)ccc2-c2ccc(C=C[C@]3C(Cl)=CC=C(*)C3)cc2)c1Cl 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/40—Dyes ; Pigments
- C11D3/42—Brightening agents ; Blueing agents
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3947—Liquid compositions
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/395—Bleaching agents
- C11D3/3956—Liquid compositions
Definitions
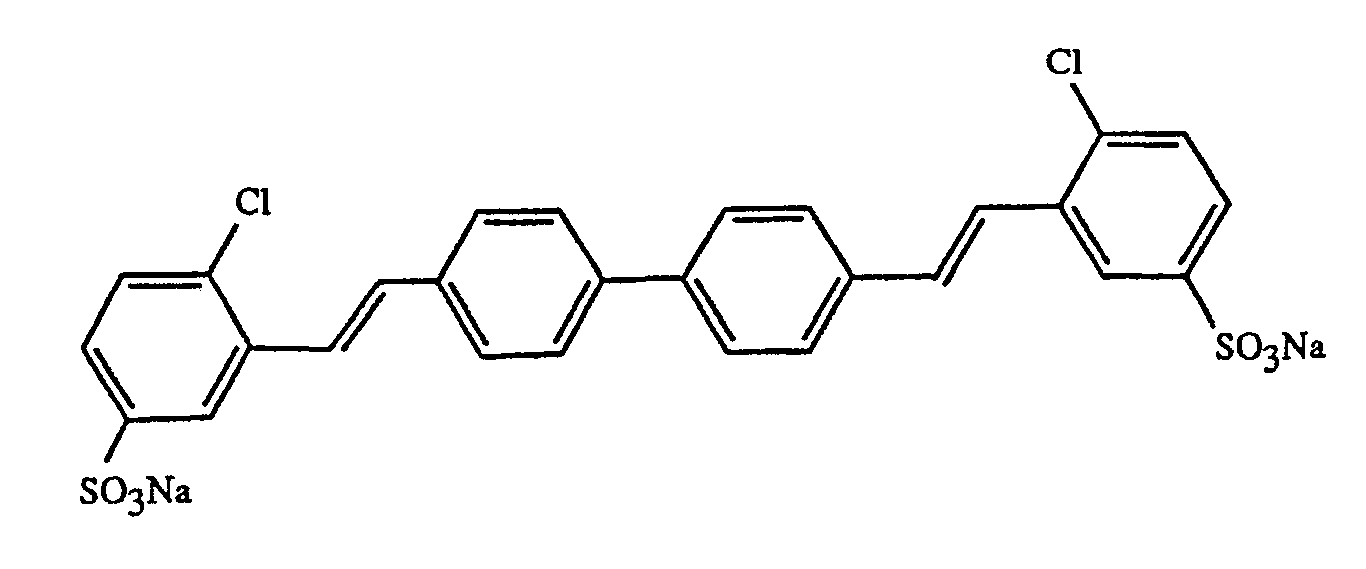
- the present invention relates to liquid detergents which contain 2,2'-dichloro-5,5'-disulfodistyrylbiphenyl as an optical brightener.
- optical brighteners in liquid detergents is generally known. They soak on the items to be washed during treatment and, thanks to their special light absorption / emission properties, lead to an elimination of the yellowish tones.
- EP-A-167 205 proposes to use monosulfonated stilbene triazolyl, triazine or distyrylbiphenyl brighteners. Furthermore, EP-A-0 298 361 describes liquid detergents containing disulfonated distyryl brighteners.
- the brightener does not sediment and the liquid detergent is clear, i.e. without opalescence.
- the invention thus relates to liquid detergents containing optical brighteners, characterized in that the optical brightener present in an amount of 0.01 to 2% by weight, preferably 0.01 to 1% by weight, consists of a disulfonic acid brightener of the formula consists in which M ⁇ is hydrogen or a salt-forming cation.
- liquid detergents which contain the brightener of the formula (I) in an amount of 0.02 to 0.3% by weight.
- M ⁇ in the meaning of a salt-forming cation is preferably alkali metal such as lithium, sodium, potassium and optionally substituted ammonium such as ammonium, mono-, di- or triethanolammonium, mono-, di- or tripropanolammonium or tri- or tetramethylammonium, but especially for Sodium.
- the brighteners of formula (I) are e.g. known from DOS 25 04 276. They are manufactured using known processes.
- Liquid detergents can usually contain 2-60% by weight of anionic, nonionic, cationic or zwitterionic surfactants, 3-50% by weight of builders, 25-95% by weight of solvent and 0-30% by weight of auxiliaries.
- surfactant Generally the surfactant, builder, solvent and adjuvants are mixtures thereof.
- Liquid detergents can contain 1-50% by weight, preferably 10-40% by weight, of anionic surfactants.
- Anionic surfactants are especially those of the sulfate or sulfonate type and soaps.
- Examples include: alkyl or alkylbenzenesulfates and sulfonates (in particular C11-C13 alkylbenzenesulfonate), fatty acid monoglyceryl sulfates and sulfonates, paraffin sulfonates, olefin sulfonates, fatty alcohol ether sulfates, alkylglyceryl ether sulfonates, ethoxylated alkyl and alkyl sulfonates, 1-sulfonyl sulfonates, sulfonates, ethoxylated sulfonic acids, , ⁇ -alkyloxyalkanesulfonates.
- the substances can be in the form of their alkali metal, especially sodium and potassium, ammonium or alkanolamine salts are present.
- Liquid detergents can contain 1-30% by weight, preferably 4-15% by weight, of nonionic surfactants.
- Nonionic surfactants are, for example, by the condensation of ethylene oxide with a hydrocarbon which carries an active hydrogen atom, such as e.g. a hydroxyl, carboxyl or amido group.
- Preferred compounds are C12-C15 fatty alcohols with 4-10 moles of ethylene oxide per mole of alcohol.
- Other nonionic surfactants which can be used are amines and amine oxides, the alkyl radical consisting of about 8-28 carbon atoms.
- Liquid detergents can contain 0-5% by weight, preferably 0.7-2% by weight, of cationic surfactants.
- Cationic surfactants include for example quaternary ammonium compounds, such Cholinesterderivate, C8-C16 alkyl trimethylammonium salts, C8-C16 alkyl di (hydroxyethyl) methyl ammonium salts, C8-C16 Alkylhydroxyethyldimethylammoniumsalze, C8-C16 Alkyloxypropyltrimethylammoniumsalze and C8-C16 Alkyloxypropyldihydroxyethylmethylammoniumsalze; the chlorides, bromides and methyl sulfates are preferred.
- quaternary ammonium compounds such Cholinesterderivate, C8-C16 alkyl trimethylammonium salts, C8-C16 alkyl di (hydroxyethyl) methyl ammonium salts, C8-C16 Alkylhydroxyethyldimethylammoniumsalze, C8-C16 Alkyloxypropyltrimethylammoniums
- Liquid detergents can contain 0-60% by weight, preferably 1-20% by weight, of zwitterionic surfactants.
- Zwitterionic surfactants are, for example, derivatives of secondary or tertiary amines, derivatives of heterocyclic, secondary and tertiary amines or derivatives of quaternary ammonium, quaternary phosphonium or tertiary, sulfonium compounds. All of these compounds have at least one aliphatic optionally branched chain containing about 3-18 carbon atoms and at least one aliphatic substituent containing an anionic water solubilizing group, e.g. a carboxy, sulfonate, sulfate, phosphate or phosphonate group.
- surfactants or surfactant mixtures can be found, for example, in EP-A-0 167 205 and in US Patents 3,929,678, 4,284,532, 4,285,841, 4,321,165 and 4,507,219.
- Liquid detergents may further contain 3-50% by weight, preferably 5-20% by weight, of builders.
- a builder is understood to mean in particular a water-soluble inorganic or organic electrolyte, but also e.g. non-water soluble calcium ion exchange materials.
- builders are: alkali metal carbonates, borates, phosphates, polyphosphates, bicarbonates, silicates, sulfates, chlorides, aminocarboxylates, aminopolyacetates, phytates, polyphosphonates, polycarboxylates (especially citrates), nitrilotriacetic acid, organic amines and amines in particular Triethanolamine), sodium aluminum silicates and optionally ethoxylated (preferably 1-10 mol ethylene oxide per mol fatty acid) C10-C22 fatty acids.
- Liquid detergents can contain 25-95% by weight, preferably 30-60% by weight, of solvent.
- the solvent is to be understood in particular as water and water-miscible organic solvents. Examples of these are lower alcohols (especially ethanol), diethylene glycol ethers and polyethylene glycols.
- the ratio of organic solvent or solvent mixture to water is generally about 1: 8 to 3: 1; however, only water can be used.
- Liquid detergents can furthermore contain 0-30% by weight, preferably 2-15% by weight, of auxiliaries.
- auxiliaries are: enzymes, enzyme stabilizers, foam suppressants, antioxidants, preservatives and disinfectants, fragrances and dyes, complexing agents or sequestering agents, graying inhibitors, dirt removers, opacifiers, hydrotropic compounds and bleach systems.
- Preferred bleaching agent systems are hypochlorites or hypochlorite-releasing substances, and also per compounds, such as perborates, persulfates, perdisulfates, perphosphates, peroxycarboxylic acids or their formation components.
- bleach systems can be added to both the liquid detergent and the wash bath separately.
- auxiliaries can be found in DOS 27 56 583 and EP-A-0 293 040.
- a liquid detergent which contains 0.02 to 0.3% by weight of the compound of the formula is preferred 10 to 30% by weight of alkylbenzenesulfonate, 4 to 15% by weight of polyethoxy fatty alcohol, 5 to 20% by weight of builder, selected from the group containing polycarboxylates, zeolites, polyphosphates and triethanolamine, a lower aliphatic alcohol, auxiliaries and water.
- the brightener 2,2'-dichloro-5,5'-disulfodistyryl-biphenyl can also be used in solid washing powders. This is a major economic advantage in terms of storage and logistics costs. In solid detergents, the brightener not only improves the whitening effect in the wash bath, but also the white aspect of the washing powder.
- the liquid detergents can be obtained by mixing and homogenizing optical brighteners, the surfactants, the builder, the solvent and, if appropriate, the auxiliaries.
- the liquid detergents are used primarily for washing and pretreating textile fabrics, such as fabrics containing cotton. Fabrics containing cotton are preferred.
- the stain test shows only a very slight formation of light stains with a very high degree of lightening.
- the liquid detergent obtained is a clear agent that is very stable in storage.
- the stain test is carried out with the detergent from Example 1, but the brightener of the formula (100) is replaced by that of the formula (200) or (300) or (400).
- Example 2 By spray drying a slurry consisting of 1 part of water and 1 part of detergent A of the following composition 8.4 g linear dodecylbenzenesulfonate 3.1 g tallow alcohol tetradecane ethylene glycol ether (14 AeO) 3.7 g Na soap (mainly from behenic acid and C14-C20 acids) 45.8 g Na tripolyphosphate 7.9 g Na silicate 2.0 g of Mg silicate 1.2 g carboxymethyl cellulose 0.2 g ethylenediamine tetraacetate 22.2 g Na sulfate 0.1 g of the compound of formula (100), (200) or (300) a detergent granulate with a residual moisture content of approx. 5% by weight is produced.
Abstract
Flüssigwaschmittel enthaltend als optischen Aufheller 2,2'-Dichloro-5,5'-disulfodistyrylbiphenyl, Verfahren zur Herstellung und Verwendung des Flüssigwaschmittels zum Waschen und Vorbehandeln von textilen Geweben; es handelt sich um lagerbeständige Flüssigwaschmittel, die nicht sedimentieren, die gegen Bleichsysteme stabil sind und bei der Vorbehandlung keine Flecken bilden.Liquid detergent containing 2,2'-dichloro-5,5'-disulfodistyrylbiphenyl as an optical brightener, process for producing and using the liquid detergent for washing and pretreating textile fabrics; it is storage-stable liquid detergents that do not sediment, that are stable against bleaching systems and that do not stain during pretreatment.
Description
Die vorliegende Erfindung betrifft Flüssigwaschmittel die als optischen Aufheller 2,2′-Dichloro-5,5′-disulfodistyrylbiphenyl enthalten.The present invention relates to liquid detergents which contain 2,2'-dichloro-5,5'-disulfodistyrylbiphenyl as an optical brightener.
Die Verwendung von optischen Aufhellem in flüssigen Waschmitteln ist allgemein bekannt. Sie ziehen während der Behandlung auf das Waschgut auf und führen durch ihre spezielle Lichtabsorption/Emissionseigenschaft zu einer Elimination der gelblichen Töne.The use of optical brighteners in liquid detergents is generally known. They soak on the items to be washed during treatment and, thanks to their special light absorption / emission properties, lead to an elimination of the yellowish tones.
Dieser Effekt ist aber auch verantwortlich für das Auftreten von hellen Flecken wenn Textilgewebe z.B. bei einer Vorbehandlung direkt mit dem flüssigen Waschmittel in Kontakt gerät. In der EP-A-167 205 wird zur Lösung dieses Froblems daher vorgeschlagen, monosulfonierte Stilbentriazolyl-, Triazin- oder Distyrylbiphenyl-Aufheller zu verwenden. Weiterhin werden in der EP-A-0 298 361 flüssige Waschmittel, enthaltend disulfonierte Distyryl-Aufheller, beschrieben.This effect is also responsible for the appearance of light spots when textile fabrics e.g. comes into direct contact with the liquid detergent during pretreatment. To solve this problem, EP-A-167 205 proposes to use monosulfonated stilbene triazolyl, triazine or distyrylbiphenyl brighteners. Furthermore, EP-A-0 298 361 describes liquid detergents containing disulfonated distyryl brighteners.
Es hat sich nun überraschenderweise gezeigt, dass die Bildung von hellen Flecken, bei weiterhin ausgezeichneter Aufhellwirkung verhindert werden kann, wenn 2,2′-Dichloro-5,5′-disulfodistyrylbiphenyl als optischer Aufheller in flüssige Waschmittel eingearbeitet wird.It has now surprisingly been found that the formation of light spots, while still having an excellent brightening effect, can be prevented if 2,2′-dichloro-5,5′-disulfodistyrylbiphenyl is incorporated as an optical brightener in liquid detergent.
Darüberhinaus sedimentiert der Aufheller nicht und das Flüssigwaschmittel ist klar, d.h. ohne Opaleszenz.In addition, the brightener does not sediment and the liquid detergent is clear, i.e. without opalescence.
Gegenstand der Erfindung sind somit Flüssigwaschmittel enthaltend optische Aufheller, dadurch gekennzeichnet, dass der in einer Menge von 0,01 bis 2 Gew.-%, vorzugsweise 0,01 bis 1 Gew.-% vorliegende optische Aufheller aus einem Disulfonsäureaufheller der Formel
Von besonderem praktischen Interesse sind Flüssigwaschmittel, welche den Aufheller der Formel (I) in einer Menge von 0,02 bis 0,3 Gew.-% enthalten.Of particular practical interest are liquid detergents which contain the brightener of the formula (I) in an amount of 0.02 to 0.3% by weight.
M⊕ in der Bedeutung eines salzbildenden Kations steht vorzugsweise für Alkalimetall wie Lithium, Natrium, Kalium sowie gegebenenfalls substituiertes Ammonium wie Ammonium, Mono-, Di- oder Triethanolammonium, Mono-, Di- oder Tripropanolammonium oder Tri- oder Tetramethylammonium, vor allem jedoch für Natrium.M ⊕ in the meaning of a salt-forming cation is preferably alkali metal such as lithium, sodium, potassium and optionally substituted ammonium such as ammonium, mono-, di- or triethanolammonium, mono-, di- or tripropanolammonium or tri- or tetramethylammonium, but especially for Sodium.
Die Aufheller der Formel (I) sind z.B. aus der DOS 25 04 276 bekannt. Ihre Herstellung erfolgt nach bekannten Verfahren.The brighteners of formula (I) are e.g. known from DOS 25 04 276. They are manufactured using known processes.
Ueblicherweise können Flüssigwaschmittel 2-60 Gew.-% anionische, nichtionische, kationische bzw. zwitterionische Tenside, 3-50 Gew.-% Builder, 25-95 Gew.-% Lösungsmittel und 0-30 Gew.-% Hilfsstoffe enthalten.Liquid detergents can usually contain 2-60% by weight of anionic, nonionic, cationic or zwitterionic surfactants, 3-50% by weight of builders, 25-95% by weight of solvent and 0-30% by weight of auxiliaries.
Im allgemeinen handelt es sich bei dem Tensid, bei dem Builder, dem Lösungsmittel und den Hilfsstoffen um Mischungen derselben.Generally the surfactant, builder, solvent and adjuvants are mixtures thereof.
Flüssigwaschmittel können 1-50 Gew.-%, vorzugsweise 10-40 Gew.-% anionische Tenside enthalten.Liquid detergents can contain 1-50% by weight, preferably 10-40% by weight, of anionic surfactants.
Anionische Tenside sind insbesondere solche vom Sulfat- oder Sulfonattyp und Seifen. Beispielsweise seien genannt: Alkyl- oder Alkylbenzolsulfate und -sulfonate (insbesondere C₁₁-C₁₃-Alkylbenzolsulfonat), Fettsäuremonoglycerylsulfate und -sulfonate, Paraffinsulfonate, Olefinsulfonate, Fettalkoholethersulfate, Alkylglycerylethersulfonate, ethoxylierte Alkyl- und Alkylphenolethersulfate, α-Sulfofettsäuren, 2-Acyloxyalkan-1-sulfonsäuren, β-Alkyloxyalkansulfonate. Die Substanzen können in Form ihrer Alkali metall-, insbesondere Natrium- und Kalium-, Ammonium- oder Alkanolaminsalze vorliegen.Anionic surfactants are especially those of the sulfate or sulfonate type and soaps. Examples include: alkyl or alkylbenzenesulfates and sulfonates (in particular C₁₁-C₁₃ alkylbenzenesulfonate), fatty acid monoglyceryl sulfates and sulfonates, paraffin sulfonates, olefin sulfonates, fatty alcohol ether sulfates, alkylglyceryl ether sulfonates, ethoxylated alkyl and alkyl sulfonates, 1-sulfonyl sulfonates, sulfonates, ethoxylated sulfonic acids, , β-alkyloxyalkanesulfonates. The substances can be in the form of their alkali metal, especially sodium and potassium, ammonium or alkanolamine salts are present.
Flüssigwaschmittel können 1-30 Gew.-%, vorzugsweise 4-15 Gew.-% nichtionische Tenside enthalten.Liquid detergents can contain 1-30% by weight, preferably 4-15% by weight, of nonionic surfactants.
Nichtionische Tenside sind beispielsweise durch die Kondensation von Ethylenoxid mit einem Kohlenwasserstoff der ein aktives Wasserstoffatom trägt, wie z.B. eine Hydroxyl-, Carboxyl- oder Amidogruppe, herstellbar. Bevorzugte Verbindungen sind C₁₂-C₁₅-Fettalkohole mit 4-10 mol Ethylenoxid pro mol Alkohol. Weitere nichtionische Tenside welche Verwendung finden können, sind Amine und Aminoxide, wobei der Alkylrest aus etwa 8-28 Kohlenstoffatomen besteht.Nonionic surfactants are, for example, by the condensation of ethylene oxide with a hydrocarbon which carries an active hydrogen atom, such as e.g. a hydroxyl, carboxyl or amido group. Preferred compounds are C₁₂-C₁₅ fatty alcohols with 4-10 moles of ethylene oxide per mole of alcohol. Other nonionic surfactants which can be used are amines and amine oxides, the alkyl radical consisting of about 8-28 carbon atoms.
Flüssigwaschmittel können 0-5 Gew.-%, vorzugsweise 0,7-2 Gew.-% kationische Tenside enthalten.Liquid detergents can contain 0-5% by weight, preferably 0.7-2% by weight, of cationic surfactants.
Kationische Tenside sind beispielsweise quartäre Ammoniumverbindungen, wie Cholinesterderivate, C₈-C₁₆-Alkyltrimethylammoniumsalze, C₈-C₁₆-Alkyldi(hydroxyethyl)methylammoniumsalze, C₈-C₁₆-Alkylhydroxyethyldimethylammoniumsalze, C₈-C₁₆-Alkyloxypropyltrimethylammoniumsalze und C₈-C₁₆-Alkyloxypropyldihydroxyethylmethylammoniumsalze; wobei die Chloride, Bromide und Methylsulfate bevorzugt sind.Cationic surfactants include for example quaternary ammonium compounds, such Cholinesterderivate, C₈-C₁₆ alkyl trimethylammonium salts, C₈-C₁₆ alkyl di (hydroxyethyl) methyl ammonium salts, C₈-C₁₆ Alkylhydroxyethyldimethylammoniumsalze, C₈-C₁₆ Alkyloxypropyltrimethylammoniumsalze and C₈-C₁₆ Alkyloxypropyldihydroxyethylmethylammoniumsalze; the chlorides, bromides and methyl sulfates are preferred.
Flüssigwaschmittel können 0-60 Gew.-%, vorzugsweise 1-20 Gew.-% zwitterionische Tenside enthalten.Liquid detergents can contain 0-60% by weight, preferably 1-20% by weight, of zwitterionic surfactants.
Zwitterionische Tenside sind beispielsweise Derivate von sekundären oder tertiären Aminen, Derivate von heterocyclischen, sekundären und tertiären Aminen oder Derivate von quartären Ammonium-, qartären Phosphonium- oder tertiären, Sulfoniumverbindungen. Alle diese Verbindungen weisen mindestens eine aliphatische gegebenenfalls verzweigte Kette auf, die etwa 3-18 Kohlenstoffatome enthält, und mindestens einen aliphatischen Substituenten, der eine anionische wasserlöslichmachende Gruppe enthält, wie z.B. eine Carboxy-, Sulfonat-, Sulfat-, Phosphat- oder Phosphonatgruppe.Zwitterionic surfactants are, for example, derivatives of secondary or tertiary amines, derivatives of heterocyclic, secondary and tertiary amines or derivatives of quaternary ammonium, quaternary phosphonium or tertiary, sulfonium compounds. All of these compounds have at least one aliphatic optionally branched chain containing about 3-18 carbon atoms and at least one aliphatic substituent containing an anionic water solubilizing group, e.g. a carboxy, sulfonate, sulfate, phosphate or phosphonate group.
Weitere mögliche Tenside oder Tensidgemische finden sich beispielsweise in der EP-A-0 167 205 und in den US-Patenten 3,929,678, 4,284,532, 4,285,841, 4,321,165 und 4,507,219.Further possible surfactants or surfactant mixtures can be found, for example, in EP-A-0 167 205 and in US Patents 3,929,678, 4,284,532, 4,285,841, 4,321,165 and 4,507,219.
Weiterhin können Flüssigwaschmittel 3-50 Gew.-%, vorzugsweise 5-20 Gew.-% Builder enthalten.Liquid detergents may further contain 3-50% by weight, preferably 5-20% by weight, of builders.
Unter einem Builder versteht man insbesondere einen wasserlöslichen anorganischen oder organischen Elektrolyten, aber auch z.B. nicht wasserlösliche Kalzium-Ionenaustausch-Materialien. Beispiele für Builder sind: Alkalimetallcarbonate, -borate, -phosphate, -polyphosphate, -bicarbonate, -silikate, -sulfate, -chloride, Aminocarboxylate, Aminopolyacetate, Phytate, Polyphosphonate, Polycarboxylate (insbesondere Citrate), Nitrilotriessigsäure, organische Amine und Aminsalze (insbesondere Triethanolamin), Natriumaluminiumsilikate und gegebenenfalls ethoxylierte (bevorzugt 1-10 mol Ethylenoxid pro mol Fettsäure) C₁₀-C₂₂-Fettsäuren.A builder is understood to mean in particular a water-soluble inorganic or organic electrolyte, but also e.g. non-water soluble calcium ion exchange materials. Examples of builders are: alkali metal carbonates, borates, phosphates, polyphosphates, bicarbonates, silicates, sulfates, chlorides, aminocarboxylates, aminopolyacetates, phytates, polyphosphonates, polycarboxylates (especially citrates), nitrilotriacetic acid, organic amines and amines in particular Triethanolamine), sodium aluminum silicates and optionally ethoxylated (preferably 1-10 mol ethylene oxide per mol fatty acid) C₁₀-C₂₂ fatty acids.
Weitere mögliche Builder und Gemische von Buildern werden z.B. in der EP-A-0 167 205 und in US 4,321,165 beschrieben.Other possible builders and mixtures of builders are e.g. in EP-A-0 167 205 and in US 4,321,165.
Flüssigwaschmittel können 25-95 Gew.-%, vorzugsweise 30-60 Gew.-% Lösungsmittel enthalten.Liquid detergents can contain 25-95% by weight, preferably 30-60% by weight, of solvent.
Unter dem Lösungsmittel sind insbesondere Wasser und wassermischbare organische Lösungsmittel zu verstehen. Beispiele hierfür sind niedere Alkohole (insbesondere Ethanol), Diethylenglykolether und Polyethylenglykole.The solvent is to be understood in particular as water and water-miscible organic solvents. Examples of these are lower alcohols (especially ethanol), diethylene glycol ethers and polyethylene glycols.
Das Verhältnis organisches Lösungsmittel bzw. Lösungsmittelgemisch zu Wasser beträgt im allgemeinen etwa 1:8 bis 3:1; es kann jedoch auch nur Wasser verwendet werden.The ratio of organic solvent or solvent mixture to water is generally about 1: 8 to 3: 1; however, only water can be used.
Weitere mögliche Lösungsmittelsysteme sind z.B. in der EP-A-0 2,93 040 beschrieben.Other possible solvent systems are e.g. in EP-A-0 2.93 040.
Weiterhin können Flüssigwaschmittel 0-30 Gew.-%, vorzugsweise 2-15 Gew.-% Hilfsstoffe enthalten.Liquid detergents can furthermore contain 0-30% by weight, preferably 2-15% by weight, of auxiliaries.
Beispiele für Hilfsstoffe sind: Enzyme, Enzymstabilisatoren, Schaumdämpfungsmittel, Antioxidantien, Konservierungs- und Desinfektionsmittel, Duft- und Farbstoffe, Komplexbildner bzw. Sequestriermittel, Vergrauungsinhibitoren, Schmutzentferner, Trübungsmittel, hydrotrope Verbindungen und Bleichmittelsysteme.Examples of auxiliaries are: enzymes, enzyme stabilizers, foam suppressants, antioxidants, preservatives and disinfectants, fragrances and dyes, complexing agents or sequestering agents, graying inhibitors, dirt removers, opacifiers, hydrotropic compounds and bleach systems.
Bevorzugte Bleichmittelsysteme sind Hypochlorite oder Hypochlorit freisetzende Substanzen, sowie Perverbindungen, wie Perborate, Persulfate, Perdisulfate, Perphosphate, Peroxycarbonsäuren bzw. deren Bildungskomponenten.Preferred bleaching agent systems are hypochlorites or hypochlorite-releasing substances, and also per compounds, such as perborates, persulfates, perdisulfates, perphosphates, peroxycarboxylic acids or their formation components.
Diese Bleichmittelsysteme können sowohl dem Flüssigwaschmittel als auch dem Waschbad separat zugegeben werden.These bleach systems can be added to both the liquid detergent and the wash bath separately.
Weitere Beispiele für mögliche Hilfsstoffe finden sich in der DOS 27 56 583 und der EP-A-0 293 040.Further examples of possible auxiliaries can be found in DOS 27 56 583 and EP-A-0 293 040.
Bevorzugt ist ein Flüssigwaschmittel, welches 0,02 bis 0,3 Gew.-% der Verbindung der Formel
Der Aufheller 2,2′-Dichloro-5,5′-disulfodistyryl-biphenyl kann auch in festen Waschpulvern eingesetzt werden. Dieses ist im Hinblick auf Lager- und Lgistikkosten ein wesentlicher wirtschaftlicher Vorteil. In festen Waschmitteln verbessert der Aufheller, neben der Aufhellwirkung im Waschbad, auch den Weissaspekt des Waschpulvers.The brightener 2,2'-dichloro-5,5'-disulfodistyryl-biphenyl can also be used in solid washing powders. This is a major economic advantage in terms of storage and logistics costs. In solid detergents, the brightener not only improves the whitening effect in the wash bath, but also the white aspect of the washing powder.
Wegen der hohen Beständigkeit gegenüber Oxidationsmitteln von 2,2′-Dichloro-5,5′-disulfodistyryl-biphenyl können solche festen Waschmittel selbstverständlich auch Persäure-Bleichsysteme enthalten, ohne dass die Zerstörung des Aufhellers bei der Lagerung eintritt.Because of the high resistance to oxidizing agents of 2,2'-dichloro-5,5'-disulfodistyryl-biphenyl, such solid detergents can of course also contain peracid bleaching systems without the brightener being destroyed during storage.
Die Flüssigwaschmittel sind durch Mischen und Homogenisieren von optischem Aufheller, den Tensiden, dem Builder, dem Lösungsmittel und gegebenenfalls den Hilfsstoffen erhältlich.The liquid detergents can be obtained by mixing and homogenizing optical brighteners, the surfactants, the builder, the solvent and, if appropriate, the auxiliaries.
Verwendung finden die flüssigen Waschmittel vor allem zum Waschen und Vorbehandeln von textilen Geweben, wie beispielsweise Baumwolle enthaltende Gewebe. Bevorzugt sind Gewebe, die Baumwolle enthalten.The liquid detergents are used primarily for washing and pretreating textile fabrics, such as fabrics containing cotton. Fabrics containing cotton are preferred.
Die folgenden Beispiele dienen zur Erläuterung der Erfindung; Teile bedeuten Gewichtsteile und Prozente Gewichtsprozente; der Fleckentest wird folgendermassen durchgeführt:
- a) Aufheller/Waschmittel-Formulierung:
0,1 % (100 % Aktivsubstanz) optischer Aufheller werden in einem flüssigen Waschmittel gelöst. 0,6 g dieses Aufheller enthaltenden Waschmittels (A) wird mit 400 ml Wasser (10°- 12° dH) bei einer Temperatur von 30°C verdünnt (Waschflotte B). - b) Ein 20 g Stück gebleichtes Baumwollgewebe wird auf einem Spannrahmen befestigt.
- c) Auf eine vormarkierte, runde Fläche (5 cm Durchmesser) dieses Baumwollgewebes werden mit einer Pipette 0,6 ml der Waschmittellösung (A) gleichmässig aufgetragen, nach 30 Sekunden Einwirkzeit in die vorbereitete Waschflotte (B) gegeben und während 15 Minuten bei 30°C gewaschen. Anschliessend wird mit kaltem Wasser gespült und bei 70°C getrocknet.
- d) Der Unterschied des Weissgrades nach Ganz zwischen der Auftragsfläche und der Umgebung ist ein Mass für das sogenannte Spotting-Verhalten (Bildung von hellen Flecken) und wird bei einfacher Textillage mit einem RFC3-Photometer von Zeiss bestimmt.
15 Teile C₁₁-C₁₃ Alkylbenzolsulfonat
14 Teile C₁₄-C₁₅ Polyethoxyfettalkohol (7 Ethylenoxid)
10 Teile Seife
9 Teile Ethanol
5 Teile Triethanolamin
4 Teile Na-Citrat
43 Teile Wasser
und 0,1 Teile des optischen Aufhellers der Formel
- a) Brightener / detergent formulation:
0.1% (100% active substance) optical brighteners are dissolved in a liquid detergent. 0.6 g of this detergent (A) containing brightener is diluted with 400 ml of water (10 ° -12 ° dH) at a temperature of 30 ° C (wash liquor B). - b) A 20 g piece of bleached cotton fabric is attached to a tenter.
- c) 0.6 ml of the detergent solution (A) are evenly applied to a pre-marked, round surface (5 cm in diameter) of this cotton fabric, added to the prepared wash liquor (B) after 30 seconds of exposure and for 15 minutes at 30 ° C washed. It is then rinsed with cold water and dried at 70 ° C.
- d) The difference in whiteness according to Ganz between the application area and the surroundings is a measure of the so-called spotting behavior (formation of light spots) and is determined with a simple textile layer using an RFC3 photometer from Zeiss.
15 parts of C₁₁-C₁₃ alkylbenzenesulfonate
14 parts of C₁₄-C₁₅ polyethoxy fatty alcohol (7 ethylene oxide)
10 parts of soap
9 parts of ethanol
5 parts of triethanolamine
4 parts Na citrate
43 parts of water
and 0.1 part of the optical brightener of the formula
Der Fleckentest zeigt nur eine sehr geringe Bildung heller Flecken, bei sehr hohem Aufhellgrad.The stain test shows only a very slight formation of light stains with a very high degree of lightening.
Das erhaltene Flüssigwaschmittel stellt ein klares Mittel dar, das sehr lagerbeständig ist.The liquid detergent obtained is a clear agent that is very stable in storage.
Der Fleckentest wird mit dem Waschmittel aus Beispiel 1 durchgeführt, wobei aber der Aufheller der Formel (100) durch denjenigen der Formel (200) bzw. (300) bzw. (400) ersetzt wird.
Die folgende Tabelle zeigt die Weissgradunterschiede zwischen behandelter und unbehandelter Fläche; je kleiner die Differenz ist, umso geringer ist die Spotting-Tendenz.
8,4 g lineares Dodecylbenzolsulfonat
3,1 g Talgalkohol-tetradecan-ethylenglykolether (14 AeO)
3,7 g Na-Seife (vorwiegend aus Behen-Säure und C₁₄-C₂₀-Säuren)
45,8 g Na-Tripolyphosphat
7,9 g Na-Silikat
2,0 g Mg-Silikat
1,2 g Carboxymethylcellulose
0,2 g Ethylendiamin-tetraacetat
22,2 g Na-Sulfat
0,1 g der Verbindung der Formel (100), (200) oder (300)
wird ein Waschmittelgranulat mit einer Restfeuchte von ca. 5 Gew.-% hergestellt.The following table shows the differences in whiteness between treated and untreated surfaces; the smaller the difference, the lower the spotting tendency.
8.4 g linear dodecylbenzenesulfonate
3.1 g tallow alcohol tetradecane ethylene glycol ether (14 AeO)
3.7 g Na soap (mainly from behenic acid and C₁₄-C₂₀ acids)
45.8 g Na tripolyphosphate
7.9 g Na silicate
2.0 g of Mg silicate
1.2 g carboxymethyl cellulose
0.2 g ethylenediamine tetraacetate
22.2 g Na sulfate
0.1 g of the compound of formula (100), (200) or (300)
a detergent granulate with a residual moisture content of approx. 5% by weight is produced.
4 g dieses Waschmittels werden in einem Liter Wasser (12°dH) bei einer Temperatur von 45°C gelöst. Nach 3 Minuten werden 50 g Baumwoll-Gewebe dem Bad zugegeben und während 15 Minuten bei der Temperatur von 45°C gewaschen, dann unter fliessendem Wasser 30 Sekunden gespült, und in einer Schwingmaschine bei einer Tourenzahl von ca. 1000 Umdrehungen/Min. während 30 Sekunden zentrifugiert. Dann wird das Baumwoll4 g of this detergent are dissolved in one liter of water (12 ° dH) at a temperature of 45 ° C. After 3 minutes, 50 g of cotton fabric are added to the bath and washed for 15 minutes at the temperature of 45 ° C, then rinsed under running water for 30 seconds, and in a vibrating machine at a speed of approx. 1000 revolutions / min. centrifuged for 30 seconds. Then the cotton
Gewebe getrocknet und nach der Methode von Ganz mit einem Farbmessgerät (Zeiss RFC3) sein Weissgrad bestimmt.Tissue dried and its whiteness determined using the Ganz method with a color measuring device (Zeiss RFC3).
Dieser Waschvorgang wird unter genau gleichen Bedingungen wiederholt, jedoch unter Zugabe von Hypochlorit, entsprechend 0,5 g Aktivchlor im Liter Wasser, gleichzeitig mit dem Waschmittel. Nach gleicher Behandlung wird der Weissgrad des Versuches ohne Chlor mit demjenigen mit Chlor im Waschbad verglichen: der Weissgrad-Unterschied gibt Auskunft über die Stabilität des Aufhellers gegenüber Hypochlorit, je kleiner die Differenz ist, umso weniger wird der Aufheller durch Chlor zerstört. Folgende Resultate werden erhalten:
Claims (12)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH4003/89 | 1989-11-07 | ||
| CH400389 | 1989-11-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP0427670A1 true EP0427670A1 (en) | 1991-05-15 |
Family
ID=4267993
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP90810829A Withdrawn EP0427670A1 (en) | 1989-11-07 | 1990-10-30 | Liquid detergent |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US5152921A (en) |
| EP (1) | EP0427670A1 (en) |
| JP (1) | JPH03168299A (en) |
| KR (1) | KR910009907A (en) |
| AU (1) | AU6583190A (en) |
| CA (1) | CA2029319A1 (en) |
| NZ (1) | NZ235965A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1993021296A1 (en) * | 1992-04-20 | 1993-10-28 | The Procter & Gamble Company | Aqueous liquid bleach compositions with fluorescent whitening agent and polyvinyl pyrrolidone or polyvinyl alcohol |
| DE19859575B4 (en) * | 1997-12-30 | 2011-12-22 | Kay Chemical Co. | Soaking agent with optical brightener |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH682748A5 (en) * | 1991-11-07 | 1993-11-15 | Ciba Geigy Ag | A storage-stable formulation of optical brightener. |
| GB2318360A (en) * | 1996-10-15 | 1998-04-22 | Ciba Geigy Ag | Fluorescent whitening agent formulation |
| WO1999007636A1 (en) * | 1997-08-11 | 1999-02-18 | The Clorox Company | Crystalline fluorescent whitening agents in aqueous hypochlorite |
| US8883035B2 (en) | 2009-07-27 | 2014-11-11 | Ecolab Usa Inc. | Formulation of a ware washing solid controlling hardness |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0002746A1 (en) * | 1977-12-19 | 1979-07-11 | Henkel Kommanditgesellschaft auf Aktien | Bleaching agent containing per-compounds and optical brighteners and its application in textile treatment |
| DE2808927A1 (en) * | 1978-03-02 | 1979-09-06 | Henkel Kgaa | Liq. detergent for washing textiles - contains di:sulphonated di:phenyl-di:styryl cpd. as optical whitener giving storage stability |
| EP0298361A1 (en) * | 1987-07-03 | 1989-01-11 | Ciba-Geigy Ag | Liquid detergene containing disulfonated optical brighteners |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3849155A (en) * | 1971-06-18 | 1974-11-19 | Ciba Geigy Ag | Brightening of polyacrylonitrile wet tow |
| DE2504276A1 (en) * | 1975-02-01 | 1976-08-05 | Henkel & Cie Gmbh | Detergent compsns. contg. n-alkane-sulphonates - and sulphonated bis-styryl-diphenyl brightener(s) for colour stability on storage |
| US4298490A (en) * | 1978-12-22 | 1981-11-03 | Ciba-Geigy Corporation | Process for the production of washing powders of stabilized or enhanced appearance which contain fluorescent whitening agents |
| GB2076011A (en) * | 1980-05-19 | 1981-11-25 | Procter & Gamble | Coated white diphenyl and stilbene fabric brighteners |
| US4559169A (en) * | 1984-08-17 | 1985-12-17 | The Procter & Gamble Company | Stable liquid detergents containing anionic surfactant and monosulfonated brightener |
-
1990
- 1990-10-30 EP EP90810829A patent/EP0427670A1/en not_active Withdrawn
- 1990-11-02 US US07/608,407 patent/US5152921A/en not_active Expired - Fee Related
- 1990-11-05 CA CA002029319A patent/CA2029319A1/en not_active Abandoned
- 1990-11-05 NZ NZ235965A patent/NZ235965A/en unknown
- 1990-11-05 AU AU65831/90A patent/AU6583190A/en not_active Abandoned
- 1990-11-06 KR KR1019900017868A patent/KR910009907A/en not_active Application Discontinuation
- 1990-11-07 JP JP2300008A patent/JPH03168299A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0002746A1 (en) * | 1977-12-19 | 1979-07-11 | Henkel Kommanditgesellschaft auf Aktien | Bleaching agent containing per-compounds and optical brighteners and its application in textile treatment |
| DE2808927A1 (en) * | 1978-03-02 | 1979-09-06 | Henkel Kgaa | Liq. detergent for washing textiles - contains di:sulphonated di:phenyl-di:styryl cpd. as optical whitener giving storage stability |
| EP0298361A1 (en) * | 1987-07-03 | 1989-01-11 | Ciba-Geigy Ag | Liquid detergene containing disulfonated optical brighteners |
Non-Patent Citations (1)
| Title |
|---|
| RESEARCH DISCLOSURE, no.129, Januar 1975, HAVANT GB, Seite 30 "FLUORESCENT WHITENING AGENTS" * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1993021296A1 (en) * | 1992-04-20 | 1993-10-28 | The Procter & Gamble Company | Aqueous liquid bleach compositions with fluorescent whitening agent and polyvinyl pyrrolidone or polyvinyl alcohol |
| DE19859575B4 (en) * | 1997-12-30 | 2011-12-22 | Kay Chemical Co. | Soaking agent with optical brightener |
Also Published As
| Publication number | Publication date |
|---|---|
| NZ235965A (en) | 1992-05-26 |
| JPH03168299A (en) | 1991-07-22 |
| US5152921A (en) | 1992-10-06 |
| KR910009907A (en) | 1991-06-28 |
| AU6583190A (en) | 1991-05-16 |
| CA2029319A1 (en) | 1991-05-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0158260B1 (en) | Detergent for coloured textiles | |
| DE2948923C2 (en) | ||
| DE2334899C2 (en) | Granular detergent containing builders for washing and softening textiles at the same time | |
| DE2613936C2 (en) | ||
| DE2703020C2 (en) | Detergents containing hydroxyalkylamines | |
| DE2938731C2 (en) | ||
| DE2624483C2 (en) | ||
| CH684485A5 (en) | Liquid detergent. | |
| EP0427670A1 (en) | Liquid detergent | |
| DE1767682A1 (en) | Detergent and cleaning agent mixtures | |
| EP0090310B1 (en) | Agent for the laundering and colour-fast bleaching of textiles | |
| AT394386B (en) | BLEACHING GRINED DETERGENT COMPOSITION | |
| EP0002746A1 (en) | Bleaching agent containing per-compounds and optical brighteners and its application in textile treatment | |
| DE2125249A1 (en) | Builder substances for detergents and cleaning agents | |
| EP0394998B1 (en) | Liquid detergent | |
| DE2401062A1 (en) | PHOSPHATE-FREE DETERGENT MIXTURES | |
| DE2203004A1 (en) | Liquid heavy duty detergent | |
| CH678678B5 (en) | ||
| EP0334372B1 (en) | Liquid, anionic storage-stable detergents containing amphoteric distyryl derivatives | |
| DE2242093C2 (en) | Phosphate-free detergents containing alkyl ether sulfate | |
| AT394575B (en) | Bleach and detergent | |
| DE3812455A1 (en) | Bleach substitute for detergents | |
| DE19859575B4 (en) | Soaking agent with optical brightener | |
| DE2903979A1 (en) | Hydroxy-carboxylic acid amide derivs. - prepd. by acylating hydroxyalkyl alkylene di:amine and used as greying inhibitors in detergent compsns. | |
| DE3121242A1 (en) | Process for bleaching and disinfecting textiles and composition therefor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19901102 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): CH DE ES FR GB IT LI NL SE |
|
| 17Q | First examination report despatched |
Effective date: 19940901 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 19950112 |