EP0366834B1 - Verfahren zur Herstellung von Eisenoxidpulver - Google Patents
Verfahren zur Herstellung von Eisenoxidpulver Download PDFInfo
- Publication number
- EP0366834B1 EP0366834B1 EP88121026A EP88121026A EP0366834B1 EP 0366834 B1 EP0366834 B1 EP 0366834B1 EP 88121026 A EP88121026 A EP 88121026A EP 88121026 A EP88121026 A EP 88121026A EP 0366834 B1 EP0366834 B1 EP 0366834B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- solution
- iron oxide
- value
- powdered iron
- chloride solution
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 title claims abstract description 116
- 239000012256 powdered iron Substances 0.000 title claims abstract description 59
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 11
- FBAFATDZDUQKNH-UHFFFAOYSA-M iron chloride Chemical compound [Cl-].[Fe] FBAFATDZDUQKNH-UHFFFAOYSA-M 0.000 claims abstract description 47
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims abstract description 24
- 230000001590 oxidative effect Effects 0.000 claims abstract description 15
- 150000001875 compounds Chemical class 0.000 claims abstract description 12
- 239000000706 filtrate Substances 0.000 claims abstract description 6
- 238000000034 method Methods 0.000 claims description 30
- 229910001035 Soft ferrite Inorganic materials 0.000 claims description 17
- 229910000831 Steel Inorganic materials 0.000 claims description 15
- 230000005587 bubbling Effects 0.000 claims description 15
- 238000001914 filtration Methods 0.000 claims description 15
- 239000010959 steel Substances 0.000 claims description 15
- 229910000859 α-Fe Inorganic materials 0.000 claims description 9
- -1 aqueous ammonia Chemical class 0.000 claims description 4
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 claims description 3
- 239000003513 alkali Substances 0.000 claims description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 claims description 3
- 238000011282 treatment Methods 0.000 abstract description 10
- 239000000243 solution Substances 0.000 description 114
- 235000013980 iron oxide Nutrition 0.000 description 61
- VBMVTYDPPZVILR-UHFFFAOYSA-N iron(2+);oxygen(2-) Chemical class [O-2].[Fe+2] VBMVTYDPPZVILR-UHFFFAOYSA-N 0.000 description 18
- 239000012535 impurity Substances 0.000 description 15
- 238000005554 pickling Methods 0.000 description 13
- 238000010907 mechanical stirring Methods 0.000 description 12
- 239000013078 crystal Substances 0.000 description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 10
- 230000008569 process Effects 0.000 description 10
- 229910052698 phosphorus Inorganic materials 0.000 description 8
- 230000009467 reduction Effects 0.000 description 8
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 7
- 229910052742 iron Inorganic materials 0.000 description 7
- 239000011574 phosphorus Substances 0.000 description 7
- 239000002699 waste material Substances 0.000 description 7
- 239000004744 fabric Substances 0.000 description 6
- 238000007670 refining Methods 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 230000007423 decrease Effects 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 3
- 239000000428 dust Substances 0.000 description 3
- 239000011812 mixed powder Substances 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- 230000035699 permeability Effects 0.000 description 3
- 235000011007 phosphoric acid Nutrition 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 230000008901 benefit Effects 0.000 description 2
- 229910052681 coesite Inorganic materials 0.000 description 2
- 229910052906 cristobalite Inorganic materials 0.000 description 2
- 238000010494 dissociation reaction Methods 0.000 description 2
- 230000005593 dissociations Effects 0.000 description 2
- AMWRITDGCCNYAT-UHFFFAOYSA-L hydroxy(oxo)manganese;manganese Chemical compound [Mn].O[Mn]=O.O[Mn]=O AMWRITDGCCNYAT-UHFFFAOYSA-L 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 239000003595 mist Substances 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 239000007921 spray Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 229910052682 stishovite Inorganic materials 0.000 description 2
- 229910052905 tridymite Inorganic materials 0.000 description 2
- 238000000108 ultra-filtration Methods 0.000 description 2
- 239000011787 zinc oxide Substances 0.000 description 2
- 229910000975 Carbon steel Inorganic materials 0.000 description 1
- 206010013457 Dissociation Diseases 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000010962 carbon steel Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 208000018459 dissociative disease Diseases 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 239000008235 industrial water Substances 0.000 description 1
- 229910000358 iron sulfate Inorganic materials 0.000 description 1
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 150000001455 metallic ions Chemical class 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G49/00—Compounds of iron
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B3/00—Extraction of metal compounds from ores or concentrates by wet processes
- C22B3/04—Extraction of metal compounds from ores or concentrates by wet processes by leaching
- C22B3/06—Extraction of metal compounds from ores or concentrates by wet processes by leaching in inorganic acid solutions, e.g. with acids generated in situ; in inorganic salt solutions other than ammonium salt solutions
- C22B3/10—Hydrochloric acid, other halogenated acids or salts thereof
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G49/00—Compounds of iron
- C01G49/0018—Mixed oxides or hydroxides
- C01G49/0072—Mixed oxides or hydroxides containing manganese
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G49/00—Compounds of iron
- C01G49/02—Oxides; Hydroxides
- C01G49/06—Ferric oxide [Fe2O3]
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/01—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics
- C04B35/26—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics based on ferrites
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B3/00—Extraction of metal compounds from ores or concentrates by wet processes
- C22B3/20—Treatment or purification of solutions, e.g. obtained by leaching
- C22B3/44—Treatment or purification of solutions, e.g. obtained by leaching by chemical processes
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B47/00—Obtaining manganese
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/42—Magnetic properties
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/80—Compositional purity
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P10/00—Technologies related to metal processing
- Y02P10/20—Recycling
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S423/00—Chemistry of inorganic compounds
- Y10S423/01—Waste acid containing iron
Definitions
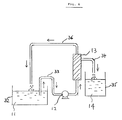
- FIG. 1 shows a diagrammatic view of an apparatus for carrying out such a process, wherein the flow is explained below.
- a reservoir 1 is filled with a waste hydrochloric pickling solution 6, and the reservoir 1 is connected to the first inlet 26 of a contact tower 2 by means of a conduit 31.
- a second inlet 25 of the contact tower 2 is supplied with hot gas 3 from a roasting furnace 5.
- the roasting furnace 5 has a first inlet 20 through which hot air 10 is supplied, while an outlet 22 of the roasting furnace 5 is connected through a conduit 23 to a dust collector 7 which in turn is connected by a conduit 24 to the second inlet 25 of the contact tower 2.
- the invention has been made after having extensively studied the influences of the impurities contained in powdered iron oxide which had been produced by the oxidizing roasting of a crude iron chloride solution of the ferrite properties of a soft ferrite. These studies revealed that the ferrite properties are remarkably improved by controlling the P content of powdered iron oxides so that the P content is not more than 0,005 % by weight. Also, it was found by the inventors that the P content contained in a crude iron chloride solution can be decreased to a very low content by an advantageous new treatment.
- the present invention provides methods for producing powdered iron oxide by oxidizing roasting an iron chloride solution when P has been removed intensively from this solution, wherein the described methods for removing phosphorus P from the solution present the striking advantage that they can be carried out very easily, with the result that powdered iron oxide can be produced in a simple and cheap manner as compared with the conventional crystal refining method.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Inorganic Chemistry (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Geology (AREA)
- Ceramic Engineering (AREA)
- Environmental & Geological Engineering (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Geochemistry & Mineralogy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Structural Engineering (AREA)
- General Chemical & Material Sciences (AREA)
- Compounds Of Iron (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
- Magnetic Ceramics (AREA)
- Soft Magnetic Materials (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
Claims (4)
- Verfahren zum Herstellen von pulverförmigem Eisenoxid durch oxidierendes Rösten einer Eisenchloridlösung,
gekennzeichnet durch folgende Verfahrensschritte:- der pH-Wert einer rohen Eisenchloridlösung wird auf einen Wert in dem Bereich von 2,5 bis 4,5 eingestellt,- die Lösung wird einem Hindurchleiten von Luftblasen ausgesetzt, das solange fortgesetzt wird, bis eine Lösung erhalten wird, deren pH-Wert nicht kleiner als 1,5 ist und die unlösliche Fe³⁺ Verbindungen in einer Menge von 0,01 bis 0,1 Gew.-%, gerechnet als Fe, enthält,- die unlöslichen Bestandteile werden von der Lösung durch Filtrierung getrennt und- das resultierende Filtrat oder die gereinigte Lösung werden dann geröstet und oxidiert, um pulverförmiges Eisenoxid zu erzeugen, das einen P-Gehalt von nicht mehr als 0,005 Gew.-% besitzt. - Verfahren nach Anspruch 1,
dadurch gekennzeichnet,
daß die Einstellung des pH-Wertes durchgeführt wird durch Verwendung einer Alkaliverbindung, wie z.B. von wässrigem Ammoniak, und/oder durch Lösen von Stahlschrott oder Walzzunder in eine Eisenchloridlösung. - Verfahren nach Anspruch 1 oder 2,
dadurch gekennzeichnet,
daß das Hindurchleiten von Luftblasen mindestens 20 Minuten lang fortgesetzt wird. - Verwendung des Eisenoxides, erhalten mit einem Verfahren nach einem der Ansprüche 1 bis 3, zur Herstellung von Ferrit, insbesondere von weichem Ferrit.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP63259448A JP2672985B2 (ja) | 1988-10-17 | 1988-10-17 | 鉄とマンガンを含有する精製溶液およびその製造方法 |
| JP259448/88 | 1988-10-17 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0366834A2 EP0366834A2 (de) | 1990-05-09 |
| EP0366834A3 EP0366834A3 (en) | 1990-06-13 |
| EP0366834B1 true EP0366834B1 (de) | 1995-03-01 |
Family
ID=17334219
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP88121026A Expired - Lifetime EP0366834B1 (de) | 1988-10-17 | 1988-12-15 | Verfahren zur Herstellung von Eisenoxidpulver |
| EP89119089A Expired - Lifetime EP0364904B1 (de) | 1988-10-17 | 1989-10-13 | Gereinigte Lösung, die Eisen und Mangan enthält und Verfahren zu deren Herstellung |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP89119089A Expired - Lifetime EP0364904B1 (de) | 1988-10-17 | 1989-10-13 | Gereinigte Lösung, die Eisen und Mangan enthält und Verfahren zu deren Herstellung |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US5032367A (de) |
| EP (2) | EP0366834B1 (de) |
| JP (1) | JP2672985B2 (de) |
| KR (2) | KR910004860B1 (de) |
| AT (1) | ATE119137T1 (de) |
| DE (2) | DE3853211T2 (de) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR920016348A (ko) * | 1991-02-05 | 1992-09-24 | 도오사끼 시노부 | 고순도 산화철 및 그 제조방법 |
| US8287738B2 (en) * | 2009-04-21 | 2012-10-16 | E I Du Pont De Nemours And Company | Process for removing impurities from iron chloride solutions |
| US8182779B2 (en) * | 2009-04-21 | 2012-05-22 | E. I. Du Pont De Nemours And Company | Process for removing phosphorus-containing colloids and their precursors from iron chloride solutions |
| WO2012094559A1 (en) * | 2011-01-06 | 2012-07-12 | Jan Pavlicek | Process for the purification of iron salt solutions |
| CN105540950A (zh) * | 2015-12-24 | 2016-05-04 | 浙江奇彩环境科技股份有限公司 | 一种铁黄反应液资源化处理的方法 |
| CN106702141B (zh) * | 2016-12-30 | 2018-03-13 | 山东盛荣新材料有限公司 | 一种利用赤泥制备赤铁锰铝复合材料的方法 |
| KR20180085173A (ko) | 2017-01-18 | 2018-07-26 | 박영길 | 슈퍼 엔지니어링 플라스틱 재생 시스템 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US1773235A (en) * | 1927-04-06 | 1930-08-19 | Lafayette M Hughes | Two-stage method of chlorinating ores |
| FR976966A (fr) * | 1942-05-22 | 1951-03-27 | Colin & Cie | Procédés de récupération du fer et autres métaux à partir de bains de décapage usés et autres déchets ou de solutions de leurs sels et appareillage pour la miseen oeuvre de ces procédés |
| US2423385A (en) * | 1944-07-27 | 1947-07-01 | Chemical Foundation Inc | Process of pickling and of recovering pickling agent |
| IE34837B1 (en) * | 1970-05-15 | 1975-09-03 | Electrolyt Zinc Australasia | Method of removing dissolved ferric iron from iron-bearing solutions |
| US3903236A (en) * | 1972-11-27 | 1975-09-02 | Deepsea Ventures Inc | Method for obtaining metal values by the halidation of a basic manganiferous ore with ferric chloride pre-treatment |
| JPS5211604B2 (de) * | 1972-07-27 | 1977-04-01 | ||
| GB1463099A (en) * | 1974-09-05 | 1977-02-02 | Nippon Electric Co | Process for treating acidic waste containing ferrous ions |
| CA1061568A (en) * | 1975-01-17 | 1979-09-04 | Antoine Van Peteghem | Process for extracting metal values from manganiferous ocean floor nodule ore |
| US4120156A (en) * | 1977-06-08 | 1978-10-17 | The Garrett Corporation | Turbocharger control |
| JPS5973439A (ja) * | 1982-10-19 | 1984-04-25 | Nippon Steel Corp | 塩酸酸洗廃液から酸化鉄を製造する方法 |
| JPS6259530A (ja) * | 1985-09-06 | 1987-03-16 | Japan Metals & Chem Co Ltd | 高純度マンガン化合物の製造方法 |
| JPS62235221A (ja) * | 1986-04-02 | 1987-10-15 | Japan Metals & Chem Co Ltd | 高純度酸化鉄の製造法 |
| JPS63117915A (ja) * | 1986-11-07 | 1988-05-21 | Sumitomo Metal Mining Co Ltd | マンガンの少ない酸化鉄用原料の製法 |
| JP2862875B2 (ja) * | 1988-05-23 | 1999-03-03 | ケミライト工業株式会社 | フェライト用の酸化鉄 |
-
1988
- 1988-10-17 JP JP63259448A patent/JP2672985B2/ja not_active Expired - Fee Related
- 1988-12-15 EP EP88121026A patent/EP0366834B1/de not_active Expired - Lifetime
- 1988-12-15 AT AT88121026T patent/ATE119137T1/de not_active IP Right Cessation
- 1988-12-15 DE DE3853211T patent/DE3853211T2/de not_active Expired - Lifetime
- 1988-12-16 US US07/285,451 patent/US5032367A/en not_active Expired - Lifetime
- 1988-12-17 KR KR1019880016917A patent/KR910004860B1/ko not_active IP Right Cessation
-
1989
- 1989-10-12 KR KR1019890014642A patent/KR910006422B1/ko not_active IP Right Cessation
- 1989-10-13 DE DE68912060T patent/DE68912060T2/de not_active Expired - Fee Related
- 1989-10-13 EP EP89119089A patent/EP0364904B1/de not_active Expired - Lifetime
- 1989-10-17 US US07/422,798 patent/US5200159A/en not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| KR900006239A (ko) | 1990-05-07 |
| US5200159A (en) | 1993-04-06 |
| EP0364904B1 (de) | 1994-01-05 |
| JP2672985B2 (ja) | 1997-11-05 |
| KR910004860B1 (ko) | 1991-07-15 |
| US5032367A (en) | 1991-07-16 |
| EP0366834A3 (en) | 1990-06-13 |
| EP0366834A2 (de) | 1990-05-09 |
| DE3853211D1 (de) | 1995-04-06 |
| DE68912060T2 (de) | 1994-08-11 |
| ATE119137T1 (de) | 1995-03-15 |
| DE68912060D1 (de) | 1994-02-17 |
| DE3853211T2 (de) | 1995-10-26 |
| EP0364904A3 (en) | 1990-06-20 |
| JPH02107529A (ja) | 1990-04-19 |
| KR910006422B1 (ko) | 1991-08-24 |
| EP0364904A2 (de) | 1990-04-25 |
| KR900006240A (ko) | 1990-05-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5129945A (en) | Scrap treatment method for rare earth transition metal alloys | |
| EP0427341B1 (de) | Verfahren zur selektiven Abtrennung eines Nichteisenmetalles | |
| EP0366834B1 (de) | Verfahren zur Herstellung von Eisenoxidpulver | |
| CN1972870A (zh) | 完全利用橄榄石组分的方法 | |
| CA1214382A (en) | Method for regenerating pickling acids | |
| CN116002647A (zh) | 一种以高铁拜耳法赤泥为铁源制备磷酸铁的方法 | |
| US4812302A (en) | Process for preparing high purity Mn3 O4 | |
| JPH0952716A (ja) | めっき廃液からのソフトフェライト用複合酸化物粉末の製造方法 | |
| JPH0582330B2 (de) | ||
| EP0343283B1 (de) | Eisenoxid für Ferrit | |
| JP2776423B2 (ja) | 酸化鉄紛の製造方法 | |
| KR100328064B1 (ko) | 산세폐액의 정제방법 | |
| WO2005068358A1 (en) | Production of 'useful material(s)' from waste acid issued from the production of titanium dioxyde | |
| JP2668287B2 (ja) | ニッケルアノード廃棄物を再生利用することにより高純度ニッケル塩化物を製造する方法 | |
| US3220795A (en) | Treatment of ferrophosphorus | |
| CN1099728A (zh) | 从亚铁盐水溶液中去除杂质的方法 | |
| KR100220866B1 (ko) | 결정화법을 이용한 폐산화망간의 정제방법 | |
| US5853692A (en) | Process for manufacturing high purity nickel chloride by recycling waste nickel anode | |
| JP3245926B2 (ja) | Mn−Znフェライトの製造方法 | |
| KR100220865B1 (ko) | 중화법을 이용한 폐산화망간의 정제방법 | |
| RU2148555C1 (ru) | Способ получения пятиокиси ванадия | |
| KR100627489B1 (ko) | 고순도 ZnCl2 용액의 제조방법 및 이를 이용한 고순도ZnO의 제조방법 | |
| KR100226895B1 (ko) | 산화철중의 염소이온 제거방법 | |
| KR0119001B1 (ko) | 소프트 페라이트 산화철 제조용 염화철 용액의 제조방법 | |
| RU1790997C (ru) | Способ получени ферромагнитного сорбирующего реагента |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE DE FR GB IT NL SE |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE DE FR GB IT NL SE |
|
| 17P | Request for examination filed |
Effective date: 19900615 |
|
| 17Q | First examination report despatched |
Effective date: 19920406 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE DE FR GB IT NL SE |
|
| REF | Corresponds to: |
Ref document number: 119137 Country of ref document: AT Date of ref document: 19950315 Kind code of ref document: T |
|
| ET | Fr: translation filed | ||
| REF | Corresponds to: |
Ref document number: 3853211 Country of ref document: DE Date of ref document: 19950406 |
|
| ITF | It: translation for a ep patent filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20041205 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20041206 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20041208 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20041215 Year of fee payment: 17 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20051215 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20051215 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20051216 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060701 |
|
| EUG | Se: european patent has lapsed | ||
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20051215 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060831 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 20060701 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20060831 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20070222 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20071212 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20071213 Year of fee payment: 20 |
|
| BERE | Be: lapsed |
Owner name: *CHEMIRITE LTD Effective date: 20071231 Owner name: *TDK CORP. Effective date: 20071231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071231 |