EP0334840B1 - Colorant bleu pour procede d'enregistrement electrophotographique a action regulatrice positive - Google Patents
Colorant bleu pour procede d'enregistrement electrophotographique a action regulatrice positive Download PDFInfo
- Publication number
- EP0334840B1 EP0334840B1 EP87900805A EP87900805A EP0334840B1 EP 0334840 B1 EP0334840 B1 EP 0334840B1 EP 87900805 A EP87900805 A EP 87900805A EP 87900805 A EP87900805 A EP 87900805A EP 0334840 B1 EP0334840 B1 EP 0334840B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- toner
- parts
- colorant
- blue
- colouring agent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
- G03G9/08—Developers with toner particles
- G03G9/09—Colouring agents for toner particles
- G03G9/0906—Organic dyes
- G03G9/0912—Indigoid; Diaryl and Triaryl methane; Oxyketone dyes
Definitions
- the present invention relates to the use of a blue colorant based on a triaminotriphenylmethane dye for coloring toners and developers for electrophotographic recording processes, which is specifically substituted as a highly crystalline sulfate salt and thus has particularly favorable charge control properties. Because of its color, the colorant is also suitable as a color-providing component for blue and green toners or developers or as a fining agent for black and brown toners or developers. In electrophotographic recording methods, for example, a "latent charge image" is generated on a photoconductor.
- the photoconductor used is then cleaned and is available for a new recording process.
- control agents also called charge control agents
- the extent of the control effect is important because a higher effectiveness allows a lower amount to be used.
- toner binders alone generally have a large change in the chargeability as a function of the activation time, it is the task of a charge control agent to set the sign and amount of the toner chargeability on the one hand and the driftability drift of the toner binder on the other counteract and ensure constant toner chargeability.
- Charge control agents which cannot prevent the toner or developer from showing a high charge drift (aging) over a long period of use, which can even cause the toner or developer to undergo charge reversal, are therefore unsuitable in practice.
- the aim of the present invention was therefore to find a charge control agent with a positive control effect, which can be adjusted as a function of concentration and whose charge control properties help the toner or developer to show as little aging as possible, and also as a colorant for blue and green or as a fining additive for black and yellow , red and brown toner or developer is suitable.
- nigrosines for example, nigrosines, quaternary ammonium compounds (US Pat. No. 4,560,635) or metal complexes (EP 0 141 377) are frequently used as charge control agents.
- Triphenylmethane dyes as positive charge control agents are also described in numerous patents, but the compounds claimed to date have hardly been used because they have always had certain deficiencies.
- Japanese patent applications 58-97056 and 56-46248 describe the use of special rosaniline sulfonic acid derivatives as positive charge control agents.
- DE-OS 3 527 306 describes the chlorides of certain triphenylmethane dyes as positive charge control agents.
- Japanese patent application 60-107654 the effect of halogenated triphenylmethane dyes
- Japanese patent applications 59-77447, 54-84732, 61-6661 and 52-113739 the effect of some Salts (triphenylborates, chlorides, hydrogen sulfates) of special triphenylmethane dye derivatives are described.
- color base sulfate is the product of the reaction of a phenylimino-2,5-cyclohexadien-1-ylidene-methylene) -bis-diphenylamine, the "color base” ", with sulfuric acid
- color base sulfate is the product of the reaction of a phenylimino-2,5-cyclohexadien-1-ylidene-methylene) -bis-diphenylamine, the "color base” ", with sulfuric acid
- this highly crystalline color base sulfate gives toners or developers surprisingly favorable properties with regard to their aging in triboelectric charging and is therefore particularly suitable for practical use.
- this colorant is suitable as a coloring component for blue and green or as a finishing component for black, yellow, red and brown toners or developers.
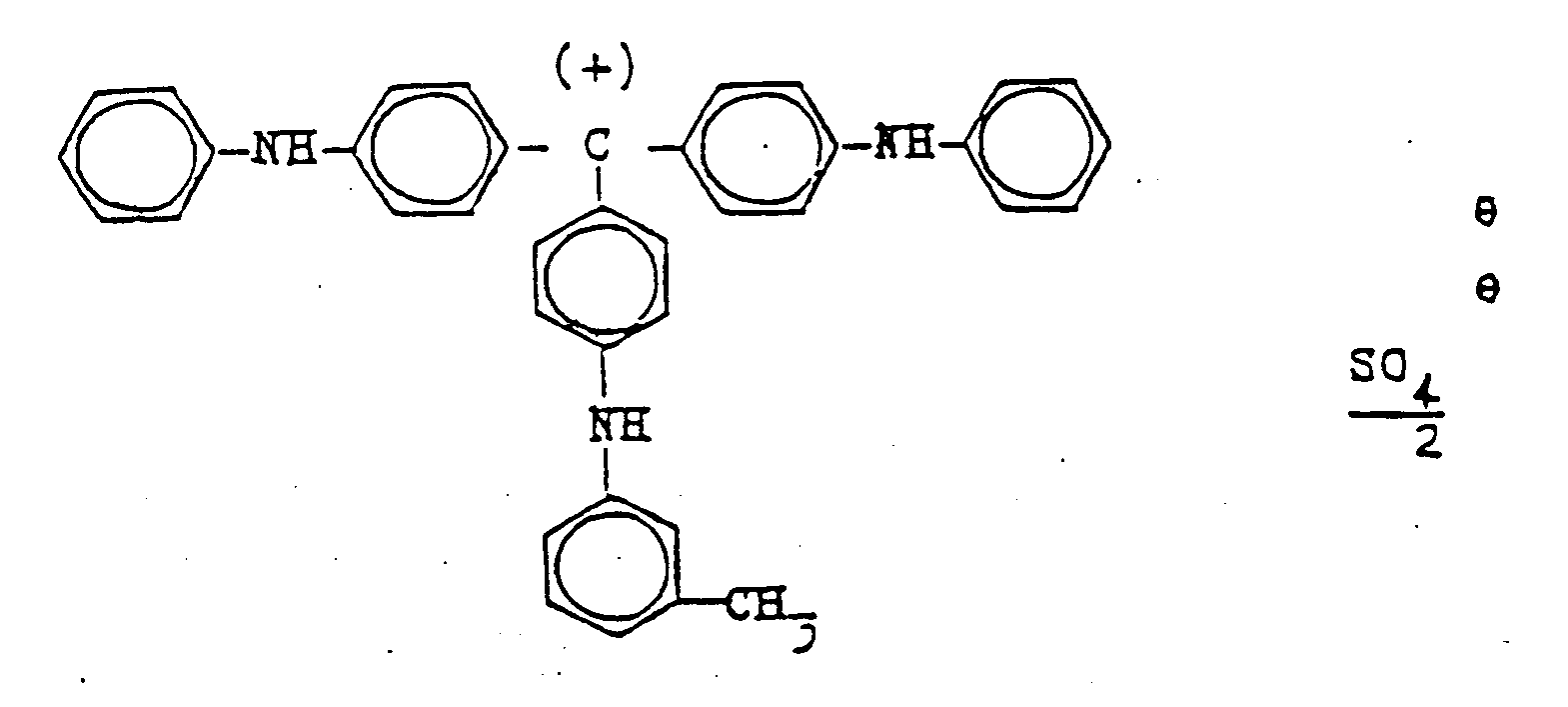
- the present invention thus relates to the use of the blue colorant, consisting essentially of the compound of the formula which in the X-ray diffraction diagram by a strong band at 2 ⁇ ° (CuK ⁇ ) 18.47, three medium-strong bands 2 ⁇ ° (CuK ⁇ ) 5.97; 12.01; 13.90 and weak broad bands at 2 ⁇ ° (CuK ⁇ ) 20.0; 21.7; 22.5; 24.8; 28.2; 30.7; 32.2 is identified as a positive charge control agent for electrophotographic toners and developers, which are used for electrophotographic copying or duplication of originals and for printing electronically, optically or magnetically stored information or in color proofing, and its use as a coloring component for blue and green toners or developers or as fining agents for black, red, yellow and brown toners or developers.
- a toner with 5% by weight of the colorant according to the invention has a chargeability of +49.1 ⁇ C / g
- a toner with 1 or 0.5% by weight of the colorant according to the invention has a chargeability of +21.5 ⁇ C / g or +15, 8 ⁇ C / g (see Examples 1 to 3 below).
- the monosulfonic acid (Reflex Blue R, CI Pigment Blue 61) of the claimed compound is used instead of the color base sulfate according to the invention, this shows a much lower charge control property in the corresponding toner.
- control agent In contrast to the control agent according to the invention, an addition of 0.5 percent by weight of this control agent (monosulfonic acid) is therefore no longer sufficient to counteract the triboelectric, negative self-chargeability of the resin (toner binder). With regard to the constancy of its charge control effect (aging), poorer properties can be seen up to the reversal of the polarity of the chargeability (comparative example 6).
- this control means is no longer able to counteract the triboelectric negative self-chargeability of the resin sufficiently.
- the highly crystalline color base sulfate according to the invention has, compared to, for example, the chlorides of the corresponding color base, significantly more favorable charge control properties (comparative example 5), just as toners or developers with the highly crystalline modification of the color base sulfate according to the invention have a significantly better long-term durability (lower signs of aging) than those with an X-ray amorphous Modification of the color base sulfate (comparative example 4).
- triphenylmethane color base (CI Solvent Blue 125) used in Comparative Examples 9, 10 and 11 is not suitable in practice, on the one hand because of its only moderate charge control ability, and on the other hand because of the poor consistency of its charge control effect (aging), which leads to the reversal of polarity of the toner leads (Comparative Example 9).
- the colorant according to the invention differs in its significantly stronger, positive charge control behavior and in the significantly better constancy of its charge control effect.
- the color and control agent according to the invention allows a toner or developer to be colored at the same time or to be beautiful and, depending on the concentration, to be controlled in a targeted triboelectric manner, which among other things. has the advantage that, in addition to the colorant, additional control agent substance does not have to be incorporated into the already complex toner formulations. This eliminates problems of compatibility, miscibility or tax funds migration.
- either dried and ground colorant or an aqueous dispersion or a press cake can be used to incorporate the colorant according to the invention into the toner binder.
- the level of electrostatic charging of the toner using the claimed colorant was measured on standard systems under the same conditions (such as the same dispersion times, same particle size distribution, same particle shape) at 23 ° C. and 50% relative atmospheric humidity.
- the toner is activated in a two-component developer by swirling the toner with a carrier (3 parts toner to 97 parts carrier) on a roller bench (150 revolutions per minute).
- the particle size is of great influence when determining the Q / M value. Care was therefore taken to ensure that the toner samples obtained from the sightings and listed in the examples below were uniform in particle size distribution.
- the desired particle fraction was activated with a carrier made of magnetite particles of size 50 to 200 ⁇ m of the 90 ⁇ m type Xerographic Carrier from Plasma Materials Inc., coated with a styrene-methacrylate copolymer 90:10.
- the measurement is carried out on a customary Q / M measuring stand (cf. J.H. Dessauer, H.E. Clark, "Xerography and related Processes", Focal Press, N.Y. 1965, page 289); by using a sieve with a mesh size of 25 ⁇ m (508 mesh per inch), from Gebrüder Kufferath, Düren, it was ensured that no carrier could be carried away during the toner blow-out.
- the Q / M value was determined to be + 49.1 ⁇ C / g.
- the Q / M value was determined to be + 34.7 ⁇ C / g.
- the Q / M value was determined to be + 28.4 ⁇ C / g.
- the Q / M value was determined to be + 21.5 ⁇ C / g.
- the Q / M value was determined to be + 15.8 ⁇ C / g.
- Example 2 The procedure was as described in Example 1, with the difference that instead of 5 parts of the colorant according to the invention, 5 parts of an X-ray amorphous form of the colorant according to the invention were used.
- the Q / M value was determined to be + 51.8 ⁇ C / g.
- the Q / M value was determined to be + 39.1 ⁇ C / g.
- the Q / M value was determined to be + 22.3 ⁇ C / g.
- the Q / M value was determined to be + 1.0 ⁇ C / g.
- the Q / M value was determined to be - 0.4 ⁇ C / g.
- the Q / M value was determined to be + 3.2 ⁇ C / g.
- the Q / M value was determined to be + 18.6 ⁇ C / g.
- the Q / M value was determined to be + 1.8 ⁇ C / g.
- the Q / M value was determined to be 5.2 ⁇ C / g.
- the Q / M value was determined to be + 4.8 ⁇ C / g.
- the Q / M value was determined to be 5.4 ⁇ C / g.
- Example 2 The procedure was as described in Example 1, with the difference that instead of 5 parts of the colorant claimed, 5 parts of the free base of a triaminotriphenylmethane colorant were incorporated into the toner.
- the colorant used for comparison is the C.I. Solvent Blue 66 (brilliant blue base SM).
- the Q / M value was determined to be + 29.2 ⁇ C / g.
- the Q / M value was determined to be + 12.7 ⁇ C / g.
- the Q / M value was determined to be - 4.8 ⁇ C / g.
- the Q / M value was determined to be + 7.2 ⁇ C / g.
- the Q / M value was determined to be + 4.0 ⁇ C / g.
- the Q / M value was determined to be - 6.5 ⁇ C / g.
Landscapes
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Developing Agents For Electrophotography (AREA)
- Color Printing (AREA)
Abstract
Claims (1)
- Utilisation de la matière colorante bleue qui est essentiellement constituée du composé de la formule :
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE3641525 | 1986-12-05 | ||
| DE19863641525 DE3641525A1 (de) | 1986-12-05 | 1986-12-05 | Blaues farbmittel fuer elektrophotographische aufzeichnungsverfahren mit positiver steuerwirkung |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0334840A1 EP0334840A1 (fr) | 1989-10-04 |
| EP0334840B1 true EP0334840B1 (fr) | 1993-03-10 |
Family
ID=6315505
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP87900805A Expired - Lifetime EP0334840B1 (fr) | 1986-12-05 | 1987-01-07 | Colorant bleu pour procede d'enregistrement electrophotographique a action regulatrice positive |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US5061585A (fr) |
| EP (1) | EP0334840B1 (fr) |
| JP (1) | JPH0774918B2 (fr) |
| KR (1) | KR950003308B1 (fr) |
| AU (1) | AU605475B2 (fr) |
| DE (2) | DE3641525A1 (fr) |
| WO (1) | WO1988004442A2 (fr) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0623941B1 (fr) * | 1993-03-09 | 1997-08-06 | Hoechst Celanese Corporation | Electrets en polymère ayant une stabilité de charge ameliorée |
| DE4447593C2 (de) | 1994-10-05 | 2000-12-07 | Clariant Gmbh | Toner für elektrophotographische Entwickler, enthaltend ein Azogelbpigment |
| US6159649A (en) * | 1996-06-13 | 2000-12-12 | Clariant Gmbh | Electrophotographic, resin-containing, electret, or inkjet compositions containing magenta azo pigment and use thereof |
| DE10251394A1 (de) * | 2002-11-05 | 2004-05-13 | Clariant Gmbh | Blaues Farbmittel mit besonders hoher Reinheit und positiver triboelektrischer Steuerwirkung |
| US7442481B2 (en) | 2004-04-27 | 2008-10-28 | Xsys Print Solutions Us Llc | Charge control agent |
| US7305836B2 (en) * | 2004-05-19 | 2007-12-11 | Eden Innovations Ltd. | Cryogenic container and superconductivity magnetic energy storage (SMES) system |
| EP3885480B1 (fr) | 2010-10-25 | 2024-09-11 | SWM Luxembourg Sarl | Matériau de filtration utilisant des mélanges de fibres qui contiennent des fibres ayant une forme stratégique et/ou des agents permettant de supprimer les charges |
| JP6264903B2 (ja) * | 2014-01-29 | 2018-01-24 | 東洋インキScホールディングス株式会社 | 有機el表示装置用青色着色組成物、カラーフィルタ、および有機el表示装置 |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2010820A1 (en) * | 1968-06-13 | 1970-02-20 | Eastman Kodak Co | Electrostatic image development |
| JPS5111455A (fr) * | 1974-07-19 | 1976-01-29 | Ricoh Kk | |
| JPS52113739A (en) * | 1976-03-19 | 1977-09-24 | Canon Inc | Electrostatically developing toner |
| JPS6040020B2 (ja) * | 1977-12-19 | 1985-09-09 | 株式会社リコー | 電子写真用正荷電シアントナー |
| JPS5579456A (en) * | 1978-12-13 | 1980-06-14 | Ricoh Co Ltd | Dry type electrophotographic toner |
| JPS5646248A (en) * | 1979-09-20 | 1981-04-27 | Mitsubishi Chem Ind Ltd | Dry type developer |
| JPS5897056A (ja) * | 1981-12-07 | 1983-06-09 | Mitsubishi Chem Ind Ltd | 静電荷現像用トナ− |
| JPS5977447A (ja) * | 1982-10-27 | 1984-05-02 | Ricoh Co Ltd | 電気的潜像現像用トナ− |
| EP0141377B1 (fr) * | 1983-11-04 | 1990-05-02 | Hodogaya Chemical Co., Ltd. | Complexes métalliques |
| JPS60107654A (ja) * | 1983-11-17 | 1985-06-13 | Hodogaya Chem Co Ltd | 電子写真用トナ− |
| JPS616661A (ja) * | 1984-06-20 | 1986-01-13 | Konishiroku Photo Ind Co Ltd | 磁性体分散型マイクロキヤリア |
| JPS6136758A (ja) * | 1984-07-30 | 1986-02-21 | Ricoh Co Ltd | 乾式電子写真用正荷電性トナ− |
| US4560635A (en) * | 1984-08-30 | 1985-12-24 | Xerox Corporation | Toner compositions with ammonium sulfate charge enhancing additives |
-
1986
- 1986-12-05 DE DE19863641525 patent/DE3641525A1/de not_active Withdrawn
-
1987
- 1987-01-07 KR KR1019880700919A patent/KR950003308B1/ko not_active Expired - Lifetime
- 1987-01-07 WO PCT/EP1987/000003 patent/WO1988004442A2/fr not_active Ceased
- 1987-01-07 JP JP62500817A patent/JPH0774918B2/ja not_active Expired - Lifetime
- 1987-01-07 US US07/664,471 patent/US5061585A/en not_active Expired - Lifetime
- 1987-01-07 DE DE8787900805T patent/DE3784736D1/de not_active Expired - Lifetime
- 1987-01-07 EP EP87900805A patent/EP0334840B1/fr not_active Expired - Lifetime
- 1987-01-07 AU AU68957/87A patent/AU605475B2/en not_active Expired
Also Published As
| Publication number | Publication date |
|---|---|
| DE3784736D1 (de) | 1993-04-15 |
| WO1988004442A2 (fr) | 1988-06-16 |
| AU6895787A (en) | 1988-06-30 |
| WO1988004442A3 (fr) | 1988-07-28 |
| EP0334840A1 (fr) | 1989-10-04 |
| KR950003308B1 (ko) | 1995-04-10 |
| US5061585A (en) | 1991-10-29 |
| DE3641525A1 (de) | 1988-06-16 |
| KR890700238A (ko) | 1989-03-10 |
| JPH0774918B2 (ja) | 1995-08-09 |
| AU605475B2 (en) | 1991-01-17 |
| JPH02501506A (ja) | 1990-05-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0367162B1 (fr) | Utilisation de composés d'ammonium et d'immonium incolores, à haut degré de fluoruration comme agents de contrôle de charges pour des procédés d'enregistrement électrophotographique | |
| DE4447593C2 (de) | Toner für elektrophotographische Entwickler, enthaltend ein Azogelbpigment | |
| DE3144017C2 (de) | Elektrostatographischer Toner | |
| EP0001785B1 (fr) | Encre électrostatographique classée granulométriquements mélange de développement et procédé de développement utilisant un tel mélange | |
| EP0247576B1 (fr) | Colorant magenta pour l'enregistrement électrophotographique | |
| DE3788024T2 (de) | Toner für elektrophotographische Verfahren, die eine Phenolverbindung enthalten. | |
| EP0551336B1 (fr) | Composes sulfoniques, oxydes sulfoniques ou sulfures d'aryle et d'aralkyle utilises comme regulateurs de charge | |
| EP0359123B1 (fr) | Procédé pour influencer distinctement l'effet triboélectrique de pigments azoiques | |
| EP0393503B1 (fr) | Utilisation de composés incolores de phosphonium ayant un degré de substitution au fluor élevé, dans des procédés d'enregistrement électrophotographique | |
| EP0334840B1 (fr) | Colorant bleu pour procede d'enregistrement electrophotographique a action regulatrice positive | |
| DE1522583A1 (de) | Elektrophotographisches Verfahren | |
| EP0362703B1 (fr) | Quinaoridones ayant des effets triboélectriques sélectivement déterminés | |
| DE69008509T2 (de) | Farbelektrofotografie für hoch qualitative Halbtonbilder. | |
| DE68915431T2 (de) | Teilchenförmiges Tonermaterial. | |
| DE3784116T2 (de) | Toner fuer elektrophotographische verfahren, die salze von chromkomplexen enthalten. | |
| DE69023336T2 (de) | Partikeltonermaterial. | |
| DE60212050T2 (de) | Harzteilchen für Ladungssteuerung und Toner für die Entwicklung elektrostatischer Bilder | |
| DE69316095T2 (de) | Ladungskontrollmittel und positiv aufladbarer Toner zur Entwicklung elektrostatischer Bilder | |
| DE69026842T2 (de) | Entwickler für elektrostatische latente Bilder | |
| EP0230950B1 (fr) | Agent colorant pour des procédés d'enregistrement électrophotographique | |
| DE69125509T2 (de) | Di- und trikationische negative Ladungssteuerungsmittel für Tonerzusammensetzung | |
| DE68924059T2 (de) | Toner für elektrophotographie. | |
| DE69410978T2 (de) | Mittels Reibung aufladendes Element, für positiv aufladbaren Toner | |
| DE3628894A1 (de) | Kondensationsprodukte 5-gliedriger heterocyclen mit triboelektrischer steuerwirkung fuer elektrophotographische aufzeichnungsverfahren | |
| DE69013529T2 (de) | Partikeltonermaterial. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19890519 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): CH DE FR GB IT LI |
|
| 17Q | First examination report despatched |
Effective date: 19911007 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): CH DE FR GB IT LI |
|
| REF | Corresponds to: |
Ref document number: 3784736 Country of ref document: DE Date of ref document: 19930415 |
|
| ITF | It: translation for a ep patent filed | ||
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 19930517 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: HOECHST AKTIENGESELLSCHAFT TRANSFER- CLARIANT GMBH |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050107 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20051209 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20051212 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20051215 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20051216 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PFA Owner name: CLARIANT PRODUKTE (DEUTSCHLAND) GMBH Free format text: CLARIANT GMBH#BRUENINGSTRASSE 50#65929 FRANKFURT AM MAIN (DE) -TRANSFER TO- CLARIANT PRODUKTE (DEUTSCHLAND) GMBH#BRUENINGSTRASSE 50#65929 FRANKFURT AM MAIN (DE) |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: CD |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20070106 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 |