EP0073690A1 - Verfahren zur katalytischen Wasserstoffumwandlung von schweren Kohlenwasserstoffen mit einem dispergierten Katalysator und kohleartige Teilchen - Google Patents
Verfahren zur katalytischen Wasserstoffumwandlung von schweren Kohlenwasserstoffen mit einem dispergierten Katalysator und kohleartige Teilchen Download PDFInfo
- Publication number
- EP0073690A1 EP0073690A1 EP82401336A EP82401336A EP0073690A1 EP 0073690 A1 EP0073690 A1 EP 0073690A1 EP 82401336 A EP82401336 A EP 82401336A EP 82401336 A EP82401336 A EP 82401336A EP 0073690 A1 EP0073690 A1 EP 0073690A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- cenospheres
- weight
- nickel
- vanadium
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 229930195733 hydrocarbon Natural products 0.000 title claims abstract description 28
- 150000002430 hydrocarbons Chemical class 0.000 title claims abstract description 28
- 238000000034 method Methods 0.000 title claims abstract description 24
- 230000003197 catalytic effect Effects 0.000 title claims abstract description 18
- 230000008569 process Effects 0.000 title claims abstract description 17
- 239000003054 catalyst Substances 0.000 title claims abstract description 14
- 239000002245 particle Substances 0.000 title abstract description 11
- 239000002184 metal Substances 0.000 claims abstract description 27
- 229910052751 metal Inorganic materials 0.000 claims abstract description 26
- 239000004215 Carbon black (E152) Substances 0.000 claims abstract description 22
- 150000002739 metals Chemical class 0.000 claims abstract description 16
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims abstract description 8
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 8
- 239000011593 sulfur Substances 0.000 claims abstract description 8
- 239000012535 impurity Substances 0.000 claims abstract description 7
- 239000004071 soot Substances 0.000 claims abstract description 6
- 238000002485 combustion reaction Methods 0.000 claims abstract description 5
- 229910052739 hydrogen Inorganic materials 0.000 claims description 22
- 239000001257 hydrogen Substances 0.000 claims description 22
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 21
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 19
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 17
- 229910052750 molybdenum Inorganic materials 0.000 claims description 17
- 239000011733 molybdenum Substances 0.000 claims description 17
- 150000002736 metal compounds Chemical class 0.000 claims description 12
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical compound S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 claims description 11
- 229910052759 nickel Inorganic materials 0.000 claims description 11
- 229910052720 vanadium Inorganic materials 0.000 claims description 11
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 claims description 9
- 150000001875 compounds Chemical class 0.000 claims description 9
- 229910052742 iron Inorganic materials 0.000 claims description 9
- 239000000203 mixture Substances 0.000 claims description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 8
- 238000006243 chemical reaction Methods 0.000 claims description 8
- 229910000037 hydrogen sulfide Inorganic materials 0.000 claims description 8
- 239000000243 solution Substances 0.000 claims description 8
- 239000002904 solvent Substances 0.000 claims description 8
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 6
- 239000007864 aqueous solution Substances 0.000 claims description 6
- 239000007788 liquid Substances 0.000 claims description 5
- 238000009835 boiling Methods 0.000 claims description 4
- 229910052757 nitrogen Inorganic materials 0.000 claims description 4
- 239000011148 porous material Substances 0.000 claims description 4
- 229910052799 carbon Inorganic materials 0.000 claims description 3
- 239000000839 emulsion Substances 0.000 claims description 3
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 claims description 2
- 229910052804 chromium Inorganic materials 0.000 claims description 2
- 239000011651 chromium Substances 0.000 claims description 2
- 229910017052 cobalt Inorganic materials 0.000 claims description 2
- 239000010941 cobalt Substances 0.000 claims description 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 claims description 2
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 claims description 2
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims description 2
- 229910052721 tungsten Inorganic materials 0.000 claims description 2
- 239000010937 tungsten Substances 0.000 claims description 2
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 claims 4
- 238000010348 incorporation Methods 0.000 claims 1
- 239000000446 fuel Substances 0.000 abstract description 5
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 abstract 1
- 238000012360 testing method Methods 0.000 description 22
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 15
- 239000000047 product Substances 0.000 description 15
- 229910000765 intermetallic Inorganic materials 0.000 description 12
- 238000001914 filtration Methods 0.000 description 9
- 239000007787 solid Substances 0.000 description 9
- 239000000945 filler Substances 0.000 description 8
- 238000000926 separation method Methods 0.000 description 8
- 150000003839 salts Chemical class 0.000 description 7
- 239000007789 gas Substances 0.000 description 6
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 6
- 239000003849 aromatic solvent Substances 0.000 description 5
- 239000010426 asphalt Substances 0.000 description 5
- 239000003921 oil Substances 0.000 description 4
- 150000004763 sulfides Chemical class 0.000 description 4
- 239000002253 acid Substances 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- -1 bitumens Substances 0.000 description 3
- 239000000571 coke Substances 0.000 description 3
- 239000000470 constituent Substances 0.000 description 3
- 238000004821 distillation Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000005194 fractionation Methods 0.000 description 3
- 238000002309 gasification Methods 0.000 description 3
- 150000002431 hydrogen Chemical class 0.000 description 3
- 239000012263 liquid product Substances 0.000 description 3
- 238000004064 recycling Methods 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 2
- LSDPWZHWYPCBBB-UHFFFAOYSA-N Methanethiol Chemical compound SC LSDPWZHWYPCBBB-UHFFFAOYSA-N 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 238000011437 continuous method Methods 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 238000005336 cracking Methods 0.000 description 2
- 239000010779 crude oil Substances 0.000 description 2
- 238000007324 demetalation reaction Methods 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 239000010881 fly ash Substances 0.000 description 2
- 239000000295 fuel oil Substances 0.000 description 2
- 238000011065 in-situ storage Methods 0.000 description 2
- 229910052976 metal sulfide Inorganic materials 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000005078 molybdenum compound Substances 0.000 description 2
- 150000002752 molybdenum compounds Chemical class 0.000 description 2
- 238000010926 purge Methods 0.000 description 2
- 230000035484 reaction time Effects 0.000 description 2
- 239000011269 tar Substances 0.000 description 2
- 238000011282 treatment Methods 0.000 description 2
- 238000005292 vacuum distillation Methods 0.000 description 2
- HNNQYHFROJDYHQ-UHFFFAOYSA-N 3-(4-ethylcyclohexyl)propanoic acid 3-(3-ethylcyclopentyl)propanoic acid Chemical compound CCC1CCC(CCC(O)=O)C1.CCC1CCC(CCC(O)=O)CC1 HNNQYHFROJDYHQ-UHFFFAOYSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- 241000923606 Schistes Species 0.000 description 1
- 235000015076 Shorea robusta Nutrition 0.000 description 1
- 244000166071 Shorea robusta Species 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 1
- 241001080024 Telles Species 0.000 description 1
- 241001125843 Trichiuridae Species 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- QGAVSDVURUSLQK-UHFFFAOYSA-N ammonium heptamolybdate Chemical compound N.N.N.N.N.N.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.[Mo].[Mo].[Mo].[Mo].[Mo].[Mo].[Mo] QGAVSDVURUSLQK-UHFFFAOYSA-N 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 239000011329 calcined coke Substances 0.000 description 1
- 238000004517 catalytic hydrocracking Methods 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 239000003245 coal Substances 0.000 description 1
- UFMZWBIQTDUYBN-UHFFFAOYSA-N cobalt dinitrate Chemical compound [Co+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O UFMZWBIQTDUYBN-UHFFFAOYSA-N 0.000 description 1
- 229910001981 cobalt nitrate Inorganic materials 0.000 description 1
- 238000010908 decantation Methods 0.000 description 1
- 239000002283 diesel fuel Substances 0.000 description 1
- 229960001484 edetic acid Drugs 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000011790 ferrous sulphate Substances 0.000 description 1
- 235000003891 ferrous sulphate Nutrition 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 230000009969 flowable effect Effects 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 239000002010 green coke Substances 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 239000010763 heavy fuel oil Substances 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 239000002198 insoluble material Substances 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 1
- 229910000359 iron(II) sulfate Inorganic materials 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- GEMHFKXPOCTAIP-UHFFFAOYSA-N n,n-dimethyl-n'-phenylcarbamimidoyl chloride Chemical compound CN(C)C(Cl)=NC1=CC=CC=C1 GEMHFKXPOCTAIP-UHFFFAOYSA-N 0.000 description 1
- KBJMLQFLOWQJNF-UHFFFAOYSA-N nickel(ii) nitrate Chemical compound [Ni+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O KBJMLQFLOWQJNF-UHFFFAOYSA-N 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000008262 pumice Substances 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 239000012429 reaction media Substances 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 239000000779 smoke Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- XMVONEAAOPAGAO-UHFFFAOYSA-N sodium tungstate Chemical compound [Na+].[Na+].[O-][W]([O-])(=O)=O XMVONEAAOPAGAO-UHFFFAOYSA-N 0.000 description 1
- 239000011949 solid catalyst Substances 0.000 description 1
- 239000012265 solid product Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- BUUPQKDIAURBJP-UHFFFAOYSA-N sulfinic acid Chemical compound OS=O BUUPQKDIAURBJP-UHFFFAOYSA-N 0.000 description 1
- 150000003464 sulfur compounds Chemical class 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G49/00—Treatment of hydrocarbon oils, in the presence of hydrogen or hydrogen-generating compounds, not provided for in a single one of groups C10G45/02, C10G45/32, C10G45/44, C10G45/58 or C10G47/00
- C10G49/02—Treatment of hydrocarbon oils, in the presence of hydrogen or hydrogen-generating compounds, not provided for in a single one of groups C10G45/02, C10G45/32, C10G45/44, C10G45/58 or C10G47/00 characterised by the catalyst used
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G49/00—Treatment of hydrocarbon oils, in the presence of hydrogen or hydrogen-generating compounds, not provided for in a single one of groups C10G45/02, C10G45/32, C10G45/44, C10G45/58 or C10G47/00
- C10G49/10—Treatment of hydrocarbon oils, in the presence of hydrogen or hydrogen-generating compounds, not provided for in a single one of groups C10G45/02, C10G45/32, C10G45/44, C10G45/58 or C10G47/00 with moving solid particles
- C10G49/12—Treatment of hydrocarbon oils, in the presence of hydrogen or hydrogen-generating compounds, not provided for in a single one of groups C10G45/02, C10G45/32, C10G45/44, C10G45/58 or C10G47/00 with moving solid particles suspended in the oil, e.g. slurries
Definitions
- the present invention relates to a process for the catalytic hydroconversion of heavy hydrocarbon feedstocks containing asphaltenes and metallic impurities, sulfur and nitrogen.
- the catalytic system of the invention leads, under the conditions of hydroconversion, to the transformation of a portion of the heavy products of the feedstock into products with a lower boiling point and significantly lowers the content of impurities by hydrodemetallization, hydrodesulfurization and hydrodenitrification as well as the value of the Conradson carbon residue.
- cenospheres Another important advantage of the presence of the cenospheres is to allow, at the end of the reaction, easy filtration of the catalyst residues (a) present in the liquid product of the reaction.
- the fines entrained by the gas, during gasification have an average size of less than 10 microns. They contain the metals from the oil, usually vanadium, iron and nickel, and, in addition, the metallic component of the oil-soluble catalytic metal compound which had been added.
- U.S. Patent 4,204,943 describes a catalytic hydroconversion process in which the catalyst consists of carbonaceous or fine particles derived therefrom, the diameter of which is less than 10 microns. These particles and fines come from the gasification of coke.
- US Pat. No. 4,227,995 describes a catalytic hydrodemetallization process in which the catalyst consists of particles of calcined coke or "green coke" having a porosity of less than 0.3 cm3 / g and a specific surface area less than 5 m2 / g, 50 to 80% of the pores having diameters greater than 10 000 hngstrkms (1 pm).
- US Patent 4,299,685 describes a hydrocracking process for heavy oil, the catalyst of which consists of fly ash; fly ash are particles with a high mineral content and low carbon content; under the electron microscope, they have a smooth appearance. Their porosity is low, of the order of 0.3 to 0.4 cm3 / g.
- cenospheres make it a very efficient and inexpensive material for transporting insoluble materials and the metals formed during hydroconversion.
- Their high content of metals (Fe, Ni, V) (approximately 1 to 10% by weight, in total, of these 3 metals) also gives them a catalytic activity of cracking, hydrogenation and demetallation.
- their roughly spherical shape and their relatively large size ensure their easy removal by filtration without clogging the filters.
- cenospheres contain, by weight, 0.1 to 2% vanadium (preferably 0.4 to 2%), 0.1 to 5% iron (preferably 0.4 to 2%) and 0 , 2 to 1% of nickel (preferably 0.5 to 1%), these values not being limiting.
- They also contain carbon, for example 60 to 90% by weight, and sulfur, for example 2 to 10% by weight, as well as common elements such as Na and Ca.
- the specific surface of the cenospheres can be very variable, most often between 2 and 130 m2 / g, preferably 2 to 20 m2 / g.
- the average diameter of the cenospheres is usually greater than 10 ⁇ m, for example between 10 and 200 ⁇ m or between 20 and 200 ⁇ m, more particularly between 20 and 60 ⁇ m.
- Their grain density is usually from 0.3 to 0.8 g / cm3, preferably from 0.4 to 0.6 g / cm3, and their structural density usually from 1.2 to 2.5 g / cm3, from preferably 1.3 to 2.1 g / cm3.
- Their total pore volume is usually 0.8 to 2.5 cm3 / g, preferably 1.2 to 1.7 cm3 / g.
- cenospheres Certain initially spherical cenospheres may have been broken and the invention also covers the use of debris from cenospheres.
- Hydroconversion is a process in which a part of the heavy constituents of the charge is transformed under hydrogen pressure, at high temperature, into products with a lower boiling point.
- This process is particularly well suited for the heaviest hydrocarbon feedstocks having a Conradson carbon residue of up to 50% by weight.
- These fillers also have very high contents by weight of asphaltenes (for example up to 40%), sulfur (for example up to 8%) and metals (for example up to 3000 ppm).
- the catalytic metal compound used in the invention is a finely divided metallic compound preferably originating from a metallic compound soluble in the filler or from an aqueous solution of a metallic salt which is dispersed in the filler or, intermediately , in a hydrocarbon solvent.
- the metallic constituent of these soluble compounds convertible into a solid dispersed catalyst belongs to groups VB, VI B, VII B and or VIII according to the table published by EH Sargent in 1962.
- the preferred metals are molybdenum, vanadium, chromium , tungsten, manganese, iron, nickel, cobalt.
- the preferred compounds are molybdenum naphthenate and molybdenum blue.
- the amount of the soluble metal compound added to the charge is for example between 10 and 1000 ppm, preferably between 50 and 500 ppm counted by weight of metal relative to the charge.
- the metal compound can be added either alone or mixed with one or more compounds of different metals.
- the metallic compound, dissolved in an aqueous solution optionally pre-emulsified with a hydrocarbon can be for example: ammonium or alkali metal heptamolybdate, cobalt nitrate, nickel nitrate, ferrous sulfate or sodium tungstate.
- the preferred compound is ammonium heptamolybdate either alone or as a mixture with another metallic water-soluble compound.
- the amount of metallic compound dissolved in the emulsified aqueous solution is between 10 and 1000 ppm, preferably between 50 and 500 ppm counted by weight of metal.
- the cenospheres most often originate from smoke dedusting installations in large thermal power plants burning heavy industrial fuels, in particular heavy no. 2 fuels.
- cenospheres are mixed with the filler in the proportion of 0.1 to 5% by weight relative to the latter.
- the filler containing the cenospheres, the soluble metallic compound or the metallic salt provided by an aqueous solution or emulsion may or may not be subjected to a pretreatment.
- the purpose of this pretreatment is to transform the metal compound or the metal salt into a finely dispersed solid catalyst comprising from 10 to 1000 ppm, preferably from 50 to 300 ppm by weight of active material counted as elemental metal, based on the weight of filler .
- the pretreatment is carried out in the presence of hydrogen sulphide alone or in admixture with hydrogen at a temperature between 200 and 450 ° C and under a pressure between 25 and 250 bars. During this pretreatment, some or all of the metals contained in the cenospheres are also transformed into metallic sulphides.
- the charge mixed with the constituents of the catalytic system is sent to the hydroconversion reactor where the metallic compound or the metallic salt and the metals contained in the cenospheres are transformed into metallic sulfides under the action feed sulfur and / or sulfur compounds formed during the reaction, especially H 2 S.
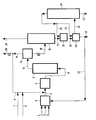
- FIG. 3 describes an embodiment of the method given by way of example.
- the fresh charge, the soluble metallic compound or the emulsion of an aqueous solution of a metallic salt in a hydrocarbon are introduced respectively by lines 1, 2 and 3 into a mixing tank 4.
- This mixture is pumped, line 5, into a pretreatment reactor 6, where it is brought into contact with hydrogen containing from 2 to 10% of hydrogen sulfide.
- Hydrogen is a mixture of fresh hydrogen (line 7) and recycled hydrogen (line 8).
- the hydrogen sulphide is supplied either by recycling (line 8), or by a fresh supply (line 9).
- the temperature is between 200 and 450 ° C, preferably 350/450 ° C, the pressure between 25 and 250 bars, preferably 100/200 bars, the reaction time between 5 minutes and 4 hours, from preferably 10 minutes to 2 hours.
- the pretreated product is introduced (line 10) into the hydroconversion reactor 11.
- the temperature of this reactor is between 380 and 480 ° C, preferably 420 to 460 ° C, the partial pressure of hydrogen between 25 and 250 bars , preferably between 100 and 200 bars, the hydrogen flow rate between 1000 and 5000 liters TPN / liter of charge, preferably between 1000 and 2000 1/1 and the space speed (VVH) defined by the charge volume per hour and per reactor volume between 0.1 and 10, preferably between 0.25 and 5.
- the effluent which leaves the hydroconversion reactor via line 12 comprises gases and a liquid having solids in suspension. It is introduced into a high pressure separator 13. From this separator leaves a gas (line 14) which contains hydrogen, hydrogen sulfide and light hydrocarbons. Part of this gas is recycled, after treatment to remove the hydrogen sulfide, to the pretreatment reactor or the hydroconversion reactor if there is no pretreatment. The other part is eliminated (28) to maintain the partial pressures of hydrogen and hydrogen sulfide at the fixed level.
- This fractionation unit can be a simple vacuum evaporator or a vacuum distillation column.
- the adjustment of the separation between distillate and residue is done to obtain a flowable and pumpable residue under industrial conditions.
- the residue drawn off through line 17 is mixed in tank 18 with an aromatic solvent with a boiling point of between 100 and 220 ° C., introduced through line 25.
- This solvent reduces the viscosity and allows a phase to be obtained.
- which is treated in a separation unit 20, joined at 18 by line 19. In this separation unit, the solids are separated by filtration or centrifugation or decantation.
- the filtered or centrifuged solids are washed with the same aromatic solvent (line 26), in the separation unit 20, to remove the oily products which coat the sulphides of the catalytic metals, the sulphides of the metals contained in the feed, the cenospheres more or less charged with metals and metal sulfides and insoluble in the aromatic solvent.
- a fraction of these solids is eliminated via line 21. They can be burned, carbonated or treated in order to recover the metals.
- the other fraction is recycled to the hydroconversion reactor (line 22), this via the mixing tank 4, the residual aromatic solvent can either be kept or eliminated.
- the aromatic solvent is withdrawn, which is reinjected into the mixer 18 via line 25, and into the separation unit 20, through line 26, for washing the filtered or centrifuged solids.
- the hydrotreated residue (line 27), largely free of metals, sulfur, nitrogen and asphaltenes. This residue is either burned, or gasified, or diluted to make a heavy fuel oil No. 2.
- a 250 ml stainless steel autoclave is used.
- the gas-liquid contact is ensured by shaking.
- a test is made with 30 g of filler.
- the autoclave after loading the soluble molybdenum compound, the cenospheres and the charge, is closed and weighed at atmospheric pressure, scanned with hydrogen and subjected to a hydrogen pressure of 100 bars for one hour to check sealing.
- the autoclave is first filled with hydrogen sulphide at 10 bars, then it is completed up to 100 bars with hydrogen.
- the mixture is heated to 380 ° C., left for 1 hour, cooled to room temperature, relaxed, scanned with hydrogen and then the test is repeated as indicated above.
- the reaction medium is diluted with toluene and filtered.
- the solids are washed with hot toluene.
- the two toluene solutions, for filtration and washing, are evaporated at 100 ° C. under 0.025 bar.
- the hydrocarbons entrained with toluene are analyzed.
- the evaporation residue constitutes the hydro-converted product.
- the weight balance must be greater than 95% for a test to be considered valid.
- the charge containing the soluble metallic compound and the cenospheres is mixed in line with hydrogen containing from 3 to 7% of hydrogen sulfide, then is brought to the reaction temperature through an oven, consisting of five heating elements. It then enters the bottom of a reactor, consisting of a vertical tube.
- the reactor effluent is cooled to 150 ° C and passed through a high pressure separator. The gas from this separator is recycled after being washed with water. A purge allows the partial pressures of hydrogen and hydrogen sulfide to be adjusted.
- the hydroconverted product is drawn off at the base of the high pressure separator.
- the operation is carried out batchwise with 30 g of Safanya asphalt diluted with 35% by volume of diesel oil at 420 ° C. for two hours; initial hydrogen pressure: 100 bars; no pretreatment.
- Various tests are carried out: test without catalyst, test with cenospheres alone, test with molybdenum naphthenate alone, test with molybdenum naphthenate plus cenospheres.
- the cenospheres allow the fixation of vanadium, nickel and molybdenum.
- Molybdenum is not found in the liquid hydroconverted product.
- Example 2 The tests indicated in this example are carried out under the same conditions as in Example 1. A molybdenum blue in 5.8% solution in a C 7 - Cg alcohol is used as the molybdenum compound.
- Table III collates the results of these tests. These tests confirm the results obtained with molybdenum naphthenate, namely the presence of cenospheres increases the hydrometallization activity and reduces the weight of insoluble matter.
- Example 2 The procedure is as in Example 1, but adding to the hydrocarbon feedstock, in addition to cobalt naphthenate and the cenospheres, 0.5% by weight, relative to the feedstock, of cenospheres recovered at the end of the Example 1 and washed with hot toluene.
- the addition of these recovered cenospheres makes it possible, as shown in Table IV, to reduce the supply of fresh molybdenum naphthenate to 100 ppm, without significant modification of the results.
- the charge is mixed with molybdenum naphthenate (500 ppm by weight of molybdenum) and 1% by weight of cenospheres, identical to those used in Example 1. It is introduced into the preheating oven at the rate of 1 liter / h , where it is brought to 430 ° C., temperature at which it enters the reaction chamber.
- molybdenum naphthenate 500 ppm by weight of molybdenum
- cenospheres identical to those used in Example 1. It is introduced into the preheating oven at the rate of 1 liter / h , where it is brought to 430 ° C., temperature at which it enters the reaction chamber.
- the total pressure is 150 bars.
- the recycled hydrogen is introduced online just before the preheater, with an H 2 / Hydrocarbon ratio equal to 1000 liters per liter, the hydrogen being considered at normal temperature and pressure.
- Hydrogen contains 2 to 3% hydrogen sulfide.
- the space velocity, charge volume per hour and per reactor volume, is equal to 1.2, which corresponds to a residence time in the reactor of 54 minutes.
- Table V shows the results obtained after 100 hours of operation under the preceding conditions.
- Table VI provides the filtration rates for these various products as well as their viscosity at 50 ° C.
Landscapes
- Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Production Of Liquid Hydrocarbon Mixture For Refining Petroleum (AREA)
- Catalysts (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR8115665A FR2511389A1 (fr) | 1981-08-11 | 1981-08-11 | Procede d'hydroconversion catalytique d'hydrocarbures lourds en phase liquide et en presence d'un catalyseur disperse et de particules charbonneuses |
| FR8115665 | 1981-08-11 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0073690A1 true EP0073690A1 (de) | 1983-03-09 |
| EP0073690B1 EP0073690B1 (de) | 1985-06-19 |
Family
ID=9261443
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP82401336A Expired EP0073690B1 (de) | 1981-08-11 | 1982-07-16 | Verfahren zur katalytischen Wasserstoffumwandlung von schweren Kohlenwasserstoffen mit einem dispergierten Katalysator und kohleartige Teilchen |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US4431520A (de) |
| EP (1) | EP0073690B1 (de) |
| JP (1) | JPS58108294A (de) |
| CA (1) | CA1191804A (de) |
| DE (1) | DE3264271D1 (de) |
| FR (1) | FR2511389A1 (de) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0096382A2 (de) * | 1982-06-05 | 1983-12-21 | VEBA OEL Entwicklungs-Gesellschaft mbH | Verfahren zur Hydrierung von Schweröl, Bitumen und dergl. |
| EP0106221A2 (de) * | 1982-10-19 | 1984-04-25 | Rheinische Braunkohlenwerke AG. | Verfahren zum Hydrieren von Schwer- und Rückstandsölen und dafür verwendete Katalysatoren |
| FR2548206A1 (fr) * | 1983-06-29 | 1985-01-04 | Inst Francais Du Petrole | Procede de formation de melanges de sels metalliques solubles, principalement du vanadium et du nickel, et utilisation des melanges formes comme catalyseurs d'hydrotraitement d'hydrocarbures lourds, en phase liquide |
| FR2556000A1 (fr) * | 1983-12-02 | 1985-06-07 | Asahi Chemical Ind | Procede de conversion d'un hydrocarbure lourd en un produit de plus de valeur ou plus leger |
| GB2159168A (en) * | 1984-05-25 | 1985-11-27 | Gulf Research Development Co | Process for cracking high metals content feedstocks using a cracking catalyst mixture containing antimony and/or tin |
| EP0216306A2 (de) * | 1985-09-27 | 1987-04-01 | Rheinische Braunkohlenwerke AG. | Verbesserte Katalysatoren zur Hydrierung von Schwer- und Rückstandsölen sowie festen kohlenstoffhaltigen Substanzen, ihre Herstellung und Verfahren unter Verwendung derselben |
| FR2594137A1 (fr) * | 1986-02-10 | 1987-08-14 | Inst Francais Du Petrole | Procede d'hydrotraitement d'hydrocarbures lourds en phase liquide en presence d'un catalyseur disperse |
| FR2603598A1 (fr) * | 1986-09-10 | 1988-03-11 | Inst Francais Du Petrole | Procede d'hydroconversion d'une charge lourde d'hydrocarbures |
| EP0263522A2 (de) * | 1986-10-08 | 1988-04-13 | VEBA OEL Technologie und Automatisierung GmbH | Verfahren zur hydrierenden Konversion von Schwer- und Rückstandsölen |
| EP0271337A2 (de) * | 1986-12-12 | 1988-06-15 | Asahi Kasei Kogyo Kabushiki Kaisha | Additiv für die Hydroumwandlung eines Schweröls |
| US4770764A (en) * | 1983-03-19 | 1988-09-13 | Asahi Kasei Kogyo Kabushiki Kaisha | Process for converting heavy hydrocarbon into more valuable product |
Families Citing this family (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL8203780A (nl) * | 1981-10-16 | 1983-05-16 | Chevron Res | Werkwijze voor de hydroverwerking van zware koolwaterstofhoudende olien. |
| US4592827A (en) * | 1983-01-28 | 1986-06-03 | Intevep, S.A. | Hydroconversion of heavy crudes with high metal and asphaltene content in the presence of soluble metallic compounds and water |
| US4518488A (en) * | 1983-02-28 | 1985-05-21 | Standard Oil Company (Indiana) | Metal-containing active carbon and methods for making and using same |
| US4863892A (en) * | 1983-08-16 | 1989-09-05 | Phillips Petroleum Company | Antifoulants comprising tin, antimony and aluminum for thermal cracking processes |
| US4732664A (en) * | 1984-11-26 | 1988-03-22 | Intevep, S.A. | Process for solid separation from hydroprocessing liquid product |
| US4637870A (en) * | 1985-04-29 | 1987-01-20 | Exxon Research And Engineering Company | Hydrocracking with phosphomolybdic acid and phosphoric acid |
| US4708784A (en) * | 1986-10-10 | 1987-11-24 | Phillips Petroleum Company | Hydrovisbreaking of oils |
| US4853110A (en) * | 1986-10-31 | 1989-08-01 | Exxon Research And Engineering Company | Method for separating arsenic and/or selenium from shale oil |
| DE3912807A1 (de) * | 1989-04-19 | 1990-11-08 | Gfk Kohleverfluessigung Gmbh | Katalysator bzw. additiv zur hydrierenden behandlung von schweren erdoelen bzw. schweren erdoelfraktionen sowie verfahren zur gewinnung des katalysators |
| US5000836A (en) * | 1989-09-26 | 1991-03-19 | Betz Laboratories, Inc. | Method and composition for retarding coke formation during pyrolytic hydrocarbon processing |
| US5319119A (en) * | 1991-03-15 | 1994-06-07 | Asahi Kasei Kogyo Kabushiki Kaisha | Oleophilic molybdenum compound for use in hydroconversion of a hydrocarbon and a method for producing the same |
| US5951849A (en) * | 1996-12-05 | 1999-09-14 | Bp Amoco Corporation | Resid hydroprocessing method utilizing a metal-impregnated, carbonaceous particle catalyst |
| US5954945A (en) * | 1997-03-27 | 1999-09-21 | Bp Amoco Corporation | Fluid hydrocracking catalyst precursor and method |
| US5807478A (en) * | 1997-05-16 | 1998-09-15 | Exxon Research And Engineering Company | Bitumen modification using fly ash derived from bitumen coke |
| JP3404522B2 (ja) * | 1999-10-29 | 2003-05-12 | 独立行政法人産業技術総合研究所 | 重質油の水素化処理方法 |
| US20030234010A1 (en) * | 2002-06-25 | 2003-12-25 | Redmond Scott D. | Methods and apparatus for converting internal combustion engine (ICE) vehicles to hydrogen fuel |
| US20040016769A1 (en) * | 2002-03-15 | 2004-01-29 | Redmond Scott D. | Hydrogen storage, distribution, and recovery system |
| US7169489B2 (en) * | 2002-03-15 | 2007-01-30 | Fuelsell Technologies, Inc. | Hydrogen storage, distribution, and recovery system |
| US7011768B2 (en) * | 2002-07-10 | 2006-03-14 | Fuelsell Technologies, Inc. | Methods for hydrogen storage using doped alanate compositions |
| US20040065171A1 (en) * | 2002-10-02 | 2004-04-08 | Hearley Andrew K. | Soild-state hydrogen storage systems |
| US7737072B2 (en) * | 2004-09-10 | 2010-06-15 | Chevron Usa Inc. | Hydroprocessing bulk catalyst and uses thereof |
| EP1754770B1 (de) * | 2005-08-16 | 2017-01-18 | Research Institute of Petroleum | Verfahren zur Hydrierung von schweren Kohlenwasserstoffe |
| JP5566896B2 (ja) * | 2007-10-31 | 2014-08-06 | シェブロン ユー.エス.エー. インコーポレイテッド | 水素化処理のバルク触媒及びその使用 |
| US8722556B2 (en) * | 2007-12-20 | 2014-05-13 | Chevron U.S.A. Inc. | Recovery of slurry unsupported catalyst |
| US7818969B1 (en) | 2009-12-18 | 2010-10-26 | Energyield, Llc | Enhanced efficiency turbine |
| US10041004B2 (en) * | 2014-11-06 | 2018-08-07 | Uop Llc | Processes for producing deashed pitch |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2156777A1 (de) * | 1971-10-18 | 1973-06-01 | Universal Oil Prod Co | |
| US3923635A (en) * | 1974-06-17 | 1975-12-02 | Exxon Research Engineering Co | Catalytic upgrading of heavy hydrocarbons |
| US4178227A (en) * | 1978-03-24 | 1979-12-11 | Exxon Research & Engineering Co. | Combination hydroconversion, fluid coking and gasification |
| US4204943A (en) * | 1978-03-24 | 1980-05-27 | Exxon Research & Engineering Co. | Combination hydroconversion, coking and gasification |
| FR2443492A1 (fr) * | 1978-12-06 | 1980-07-04 | Lummus Co | Procede de demetallisation d'une charge d'hydrocarbures contenant des metaux |
| US4226742A (en) * | 1978-07-14 | 1980-10-07 | Exxon Research & Engineering Co. | Catalyst for the hydroconversion of heavy hydrocarbons |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4125455A (en) * | 1973-09-26 | 1978-11-14 | Texaco Inc. | Hydrotreating heavy residual oils |
| NL7412337A (nl) * | 1974-09-18 | 1976-03-22 | Shell Int Research | Werkwijze voor de bereiding van nieuwe kata- lysatoren. |
| US3978000A (en) * | 1975-03-19 | 1976-08-31 | American Cyanamid Company | Catalysts based on carbon supports |
| US4192735A (en) * | 1976-07-02 | 1980-03-11 | Exxon Research & Engineering Co. | Hydrocracking of hydrocarbons |
| CA1124194A (en) * | 1979-03-05 | 1982-05-25 | Ramaswami Ranganathan | Hydrocracking of heavy oils/fly ash slurries |
-
1981
- 1981-08-11 FR FR8115665A patent/FR2511389A1/fr active Granted
-
1982
- 1982-07-16 DE DE8282401336T patent/DE3264271D1/de not_active Expired
- 1982-07-16 EP EP82401336A patent/EP0073690B1/de not_active Expired
- 1982-08-11 US US06/407,217 patent/US4431520A/en not_active Expired - Fee Related
- 1982-08-11 CA CA000409246A patent/CA1191804A/fr not_active Expired
- 1982-08-11 JP JP57140478A patent/JPS58108294A/ja active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2156777A1 (de) * | 1971-10-18 | 1973-06-01 | Universal Oil Prod Co | |
| US3923635A (en) * | 1974-06-17 | 1975-12-02 | Exxon Research Engineering Co | Catalytic upgrading of heavy hydrocarbons |
| US4178227A (en) * | 1978-03-24 | 1979-12-11 | Exxon Research & Engineering Co. | Combination hydroconversion, fluid coking and gasification |
| US4204943A (en) * | 1978-03-24 | 1980-05-27 | Exxon Research & Engineering Co. | Combination hydroconversion, coking and gasification |
| US4226742A (en) * | 1978-07-14 | 1980-10-07 | Exxon Research & Engineering Co. | Catalyst for the hydroconversion of heavy hydrocarbons |
| FR2443492A1 (fr) * | 1978-12-06 | 1980-07-04 | Lummus Co | Procede de demetallisation d'une charge d'hydrocarbures contenant des metaux |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0096382A3 (en) * | 1982-06-05 | 1985-05-08 | Veba Oel Entwicklungs-Gesellschaft Mbh | Process for the hydrogenation of heavy oils, bitumen and the like |
| EP0096382A2 (de) * | 1982-06-05 | 1983-12-21 | VEBA OEL Entwicklungs-Gesellschaft mbH | Verfahren zur Hydrierung von Schweröl, Bitumen und dergl. |
| EP0106221A2 (de) * | 1982-10-19 | 1984-04-25 | Rheinische Braunkohlenwerke AG. | Verfahren zum Hydrieren von Schwer- und Rückstandsölen und dafür verwendete Katalysatoren |
| EP0106221A3 (de) * | 1982-10-19 | 1985-05-15 | Rheinische Braunkohlenwerke AG. | Verfahren zum Hydrieren von Schwer- und Rückstandsölen und dafür verwendete Katalysatoren |
| US4770764A (en) * | 1983-03-19 | 1988-09-13 | Asahi Kasei Kogyo Kabushiki Kaisha | Process for converting heavy hydrocarbon into more valuable product |
| FR2548206A1 (fr) * | 1983-06-29 | 1985-01-04 | Inst Francais Du Petrole | Procede de formation de melanges de sels metalliques solubles, principalement du vanadium et du nickel, et utilisation des melanges formes comme catalyseurs d'hydrotraitement d'hydrocarbures lourds, en phase liquide |
| FR2556000A1 (fr) * | 1983-12-02 | 1985-06-07 | Asahi Chemical Ind | Procede de conversion d'un hydrocarbure lourd en un produit de plus de valeur ou plus leger |
| GB2159168A (en) * | 1984-05-25 | 1985-11-27 | Gulf Research Development Co | Process for cracking high metals content feedstocks using a cracking catalyst mixture containing antimony and/or tin |
| GB2159168B (en) * | 1984-05-25 | 1989-05-10 | Gulf Research Development Co | Process for cracking high metals content feedstocks using a cracking catalyst mixture containing antimony and/or tin |
| EP0216306A2 (de) * | 1985-09-27 | 1987-04-01 | Rheinische Braunkohlenwerke AG. | Verbesserte Katalysatoren zur Hydrierung von Schwer- und Rückstandsölen sowie festen kohlenstoffhaltigen Substanzen, ihre Herstellung und Verfahren unter Verwendung derselben |
| EP0216306A3 (de) * | 1985-09-27 | 1987-08-05 | Rheinische Braunkohlenwerke AG. | Verbesserte Katalysatoren zur Hydrierung von Schwer- und Rückstandsölen sowie festen kohlenstoffhaltigen Substanzen, ihre Herstellung und Verfahren unter Verwendung derselben |
| EP0235003A1 (de) * | 1986-02-10 | 1987-09-02 | Institut Français du Pétrole | Verfahren zur Hydrobehandlung von schweren Kohlenwasserstoffen in flüssiger Phase in Gegenwart eines dispergierten Katalysators |
| FR2594137A1 (fr) * | 1986-02-10 | 1987-08-14 | Inst Francais Du Petrole | Procede d'hydrotraitement d'hydrocarbures lourds en phase liquide en presence d'un catalyseur disperse |
| FR2603598A1 (fr) * | 1986-09-10 | 1988-03-11 | Inst Francais Du Petrole | Procede d'hydroconversion d'une charge lourde d'hydrocarbures |
| EP0263522A2 (de) * | 1986-10-08 | 1988-04-13 | VEBA OEL Technologie und Automatisierung GmbH | Verfahren zur hydrierenden Konversion von Schwer- und Rückstandsölen |
| EP0263522A3 (en) * | 1986-10-08 | 1989-04-12 | Intevep S.A. | Process for the hydroconversion of heavy and residual oils |
| EP0516187A2 (de) * | 1986-10-08 | 1992-12-02 | VEBA OEL Technologie und Automatisierung GmbH | Verfahren zur hydrierenden Konversion von Schwer- und Rückstandsölen |
| EP0516187A3 (en) * | 1986-10-08 | 1992-12-09 | Veba Oel Technologie Gmbh | Process for the hydroconversion of heavy and residual oils |
| EP0271337A2 (de) * | 1986-12-12 | 1988-06-15 | Asahi Kasei Kogyo Kabushiki Kaisha | Additiv für die Hydroumwandlung eines Schweröls |
| EP0271337A3 (en) * | 1986-12-12 | 1989-04-12 | Asahi Kasei Kogyo Kabushiki Kaisha | An additive for the hydroconversion of a heavy hydrocarbon oil |
Also Published As
| Publication number | Publication date |
|---|---|
| JPS58108294A (ja) | 1983-06-28 |
| US4431520A (en) | 1984-02-14 |
| FR2511389B1 (de) | 1983-11-18 |
| DE3264271D1 (en) | 1985-07-25 |
| EP0073690B1 (de) | 1985-06-19 |
| FR2511389A1 (fr) | 1983-02-18 |
| CA1191804A (fr) | 1985-08-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0073690B1 (de) | Verfahren zur katalytischen Wasserstoffumwandlung von schweren Kohlenwasserstoffen mit einem dispergierten Katalysator und kohleartige Teilchen | |
| US7585406B2 (en) | Process for hydroconverting of a heavy hydrocarbonaceous feedstock | |
| US4226742A (en) | Catalyst for the hydroconversion of heavy hydrocarbons | |
| RU2352616C2 (ru) | Способ переработки тяжелого сырья, такого как тяжелая сырая нефть и кубовые остатки | |
| RU2360944C2 (ru) | Комплексный способ конверсии содержащего уголь сырья в жидкие продукты | |
| US8679322B2 (en) | Hydroconversion process for heavy and extra heavy oils and residuals | |
| US5496464A (en) | Hydrotreating of heavy hydrocarbon oils in supercritical fluids | |
| EP0343045A1 (de) | Katalytische Zusammensetzung, die ein Metallsulfid in Form einer Suspension mit einer Asphalt enthaltenden Flüssigkeit enthält und Verfahren zur Hydroviskoreduktion von Kohlenwasserstoffen | |
| US4831003A (en) | Catalyst composition and process of making | |
| FR2499584A1 (fr) | Procede de demetallisation et d'hydrocraquage simultanes d'huiles hydrocarbonees lourdes | |
| FR2792947A1 (fr) | Procede d'hydrocraquage d'huiles lourdes | |
| JP2008512557A (ja) | 重油のアップグレードにおける活性スラリー触媒組成物のリサイクル法 | |
| FR2753984A1 (fr) | Procede de conversion d'une fraction lourde d'hydrocarbures impliquant une hydrodemetallisation en lit bouillonnant de catalyseur | |
| WO2011128517A2 (fr) | Procede d'hydroconversion de charges petrolieres via une technologie en slurry permettant la recuperation des metaux du catalyseur et de la charge mettant en oeuvre une etape d'extraction | |
| EP0235003B1 (de) | Verfahren zur Hydrobehandlung von schweren Kohlenwasserstoffen in flüssiger Phase in Gegenwart eines dispergierten Katalysators | |
| FR2753982A1 (fr) | Procede catalytique en plusieurs etapes de conversion d'une fraction lourde d'hydrocarbures | |
| FR2636069A1 (fr) | Systeme catalytique pour l'hydroconversion des huiles lourdes | |
| FR2504144A1 (fr) | Nouveau procede d'hydrotraitement d'hydrocarbures lourds en presence de metaux reduits | |
| CN115916928A (zh) | 利用氢和水的重油提质工艺 | |
| FR2512457A1 (fr) | Procede de liquefaction du charbon | |
| FR2540883A1 (fr) | Procede d'hydrocraquage d'huiles lourdes en presence d'additif melange sec forme de charbon ou d'escarbilles et d'un compose metallique | |
| FR2479255A1 (fr) | Procede d'hydrocraquage d'hydrocarbures lourds utilisant du gaz de synthese | |
| BE1026853A1 (fr) | Procédé de traitement simultané | |
| FR2551452A1 (fr) | Procede pour la valorisation de petroles bruts lourds | |
| US4597855A (en) | Upgrading of residual oils using a selenium catalyst wherein sulfur and metallic impurities are reduced |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): BE DE GB IT NL |
|
| 17P | Request for examination filed |
Effective date: 19830811 |
|
| ITF | It: translation for a ep patent filed | ||
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): BE DE GB IT NL |
|
| REF | Corresponds to: |
Ref document number: 3264271 Country of ref document: DE Date of ref document: 19850725 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19910624 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19910723 Year of fee payment: 10 |
|
| ITTA | It: last paid annual fee | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19910731 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19910814 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19920716 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Effective date: 19920731 |
|
| BERE | Be: lapsed |
Owner name: INSTITUT FRANCAIS DU PETROLE Effective date: 19920731 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19930201 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee | ||
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19920716 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19930401 |