EP0017188B1 - Anode für die Alkalichlorid-Elektrolyse und Verfahren zur Herstellung von Chlor - Google Patents
Anode für die Alkalichlorid-Elektrolyse und Verfahren zur Herstellung von Chlor Download PDFInfo
- Publication number
- EP0017188B1 EP0017188B1 EP80101671A EP80101671A EP0017188B1 EP 0017188 B1 EP0017188 B1 EP 0017188B1 EP 80101671 A EP80101671 A EP 80101671A EP 80101671 A EP80101671 A EP 80101671A EP 0017188 B1 EP0017188 B1 EP 0017188B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- anode
- bridge
- copper bolt
- conical
- titanium
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired
Links
- 238000005868 electrolysis reaction Methods 0.000 title claims description 7
- 238000000034 method Methods 0.000 title claims description 6
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 title claims description 3
- 229910052801 chlorine Inorganic materials 0.000 title claims description 3
- 239000000460 chlorine Substances 0.000 title claims description 3
- 238000004519 manufacturing process Methods 0.000 title claims description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 title description 2
- 239000003513 alkali Substances 0.000 title 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims description 21
- 229910052802 copper Inorganic materials 0.000 claims description 21
- 239000010949 copper Substances 0.000 claims description 21
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 19
- 239000010936 titanium Substances 0.000 claims description 12
- 229910052719 titanium Inorganic materials 0.000 claims description 12
- 229910001514 alkali metal chloride Inorganic materials 0.000 claims description 5
- 229910000497 Amalgam Inorganic materials 0.000 claims description 3
- 239000004020 conductor Substances 0.000 claims description 3
- 238000005260 corrosion Methods 0.000 claims description 2
- 230000007797 corrosion Effects 0.000 claims description 2
- 229910052751 metal Inorganic materials 0.000 claims 1
- 239000002184 metal Substances 0.000 claims 1
- 238000009826 distribution Methods 0.000 description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 2
- 229910052753 mercury Inorganic materials 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 229910000743 fusible alloy Inorganic materials 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 230000005923 long-lasting effect Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 229910001092 metal group alloy Inorganic materials 0.000 description 1
- 238000001465 metallisation Methods 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25B—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES FOR THE PRODUCTION OF COMPOUNDS OR NON-METALS; APPARATUS THEREFOR
- C25B11/00—Electrodes; Manufacture thereof not otherwise provided for
- C25B11/02—Electrodes; Manufacture thereof not otherwise provided for characterised by shape or form
- C25B11/03—Electrodes; Manufacture thereof not otherwise provided for characterised by shape or form perforated or foraminous
Definitions
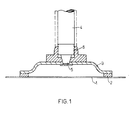
- Titanium anodes consist of a lattice-shaped or reticular titanium anode surface, which is arranged opposite and parallel to the mercury cathode and immersed in the electrolyte, which overlays the mercury cathode. Titanium anodes also consist of cross conductors of larger cross-section welded firmly to the titanium anode surface, which in turn are welded with a bridge of even larger cross-section made of titanium.
- the current is usually supplied via copper bolts which are arranged perpendicular to the titanium structure and conductively connected to the bridge.
- a problem is the attachment of the copper bolts to the bridge of the titanium structure.
- the connection between the titanium structure and the copper bolt is intended to be a good mechanical connection on the one hand, since the titanium structure is held by copper bolts and an adjustment that has been carried out over longer periods of time is to be guaranteed and on the other hand a good one to represent electrical contact.
- the titanium structure should be detachable from the copper bolt without great effort and without damaging the contact surfaces, so that the titanium structure can be easily handled when the active layer of the titanium anode surface is replaced at regular intervals.
- DE-A-2 031 525 e.g. B. proposed to produce the electrical contact between the copper bolt and the bridge of the titanium structure via a low-melting metal alloy that is liquid under operating conditions of the electrolytic cell.
- the mechanical connection is also ensured by screwing.
- the difficulties encountered with this proposal lie in the fact that such low-melting alloys generally do not wet titanium at the expense of good electrical contact and, moreover, the removal of the copper bolt is made more difficult.
- the object of the present invention is therefore to provide an anode for alkali metal chloride electrolysis which avoids the disadvantages mentioned.
- the present invention relates to an anode for the alkali metal chloride electrolysis according to the amalgam process, consisting of a lattice-shaped or reticular planar titanium anode surface, optionally current distribution rails, a bridge serving as a primary conductor rail and at least one copper bolt as a power supply, which is characterized in that the copper bolt has a thread in its lower region and is conical below the thread, the bridge has a threaded nut and a conical bore for receiving the conical region of the copper bolt, and the copper bolt and bridge are screwed together in a positive and non-positive manner.
- the conical surfaces on the copper bolt and / or the bridge can be protected against corrosion by surface metallization.
- surface metallization nickel or platinum layers are suitable.
- the cone ratio of the cone is expediently between 1: 5 and 1:15, a cone ratio of about 1:10 is preferred.
- the present invention furthermore relates to a process for the production of chlorine by electrolysis of alkali metal chloride using the anode according to the invention.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Electrodes For Compound Or Non-Metal Manufacture (AREA)
- Electrolytic Production Of Non-Metals, Compounds, Apparatuses Therefor (AREA)
- Electrolytic Production Of Metals (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19792914414 DE2914414A1 (de) | 1979-04-10 | 1979-04-10 | Anode fuer die alkalichlorid-elektrolyse und verfahren zur herstellung von chlor |
| DE2914414 | 1979-04-10 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0017188A1 EP0017188A1 (de) | 1980-10-15 |
| EP0017188B1 true EP0017188B1 (de) | 1983-01-12 |
Family
ID=6067933
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP80101671A Expired EP0017188B1 (de) | 1979-04-10 | 1980-03-28 | Anode für die Alkalichlorid-Elektrolyse und Verfahren zur Herstellung von Chlor |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US4323438A (show.php) |
| EP (1) | EP0017188B1 (show.php) |
| CA (1) | CA1157810A (show.php) |
| DE (2) | DE2914414A1 (show.php) |
| IN (1) | IN153771B (show.php) |
| NO (1) | NO153307C (show.php) |
| PL (1) | PL123695B1 (show.php) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6103096A (en) * | 1997-11-12 | 2000-08-15 | International Business Machines Corporation | Apparatus and method for the electrochemical etching of a wafer |
| US6106687A (en) * | 1998-04-28 | 2000-08-22 | International Business Machines Corporation | Process and diffusion baffle to modulate the cross sectional distribution of flow rate and deposition rate |
| US6251251B1 (en) | 1998-11-16 | 2001-06-26 | International Business Machines Corporation | Anode design for semiconductor deposition |
| US6113759A (en) * | 1998-12-18 | 2000-09-05 | International Business Machines Corporation | Anode design for semiconductor deposition having novel electrical contact assembly |
| US6261426B1 (en) | 1999-01-22 | 2001-07-17 | International Business Machines Corporation | Method and apparatus for enhancing the uniformity of electrodeposition or electroetching |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE542233C (de) * | 1931-02-03 | 1932-01-21 | Conradty Fa C | In Zellen fuer waesserige Elektrolyse horizontal einzubauende Anodenplatte |
| SE307568B (show.php) * | 1965-02-04 | 1969-01-13 | Uddeholms Ab | |
| DE1592044B2 (de) * | 1965-11-08 | 1972-12-21 | Friedrich Uhde Gmbh, 4600 Dortmund | Elektrolysezelle |
| US3536460A (en) * | 1966-12-28 | 1970-10-27 | Great Lakes Carbon Corp | Connections between electrical conductors and carbon bodies and method of making same |
| US3616445A (en) | 1967-12-14 | 1971-10-26 | Electronor Corp | Titanium or tantalum base electrodes with applied titanium or tantalum oxide face activated with noble metals or noble metal oxides |
| DK119671B (da) | 1968-12-23 | 1971-02-08 | Nordisk Ventilator | Skovlhjul til en centrifugalblæser. |
| GB1267993A (en) * | 1969-04-28 | 1972-03-22 | Marston Excelsior Ltd | Improvements in or relating to electrodes |
| GB1304518A (show.php) * | 1969-06-27 | 1973-01-24 | ||
| BE756437A (fr) * | 1969-09-22 | 1971-03-01 | Progil | Bac d'electrolyse avec nouvel ensemble anodique |
| US3912616A (en) * | 1973-05-31 | 1975-10-14 | Olin Corp | Metal anode assembly |
| BE806280A (fr) * | 1973-10-19 | 1974-02-15 | Solvay | Assemblage d'anodes pour une cellule d'electrolyse a electrodes verticales |
| DE2426674A1 (de) * | 1974-06-01 | 1975-12-04 | Bayer Ag | Anoden fuer elektrolysezellen |
| DE2717931C3 (de) * | 1977-04-22 | 1980-08-07 | Heraeus-Elektroden Gmbh, 6450 Hanau | Elektroden mit austauschbaren aktiven Flächen für Elektrolysezellen, vorzugsweise Chloralkalielektrolysezellen |
| US4121994A (en) * | 1977-11-17 | 1978-10-24 | Hooker Chemicals & Plastics Corp. | Anode support means for an electrolytic cell |
-
1979
- 1979-04-10 DE DE19792914414 patent/DE2914414A1/de not_active Withdrawn
-
1980
- 1980-03-05 IN IN156/DEL/80A patent/IN153771B/en unknown
- 1980-03-21 US US06/132,564 patent/US4323438A/en not_active Expired - Lifetime
- 1980-03-27 NO NO800890A patent/NO153307C/no unknown
- 1980-03-28 EP EP80101671A patent/EP0017188B1/de not_active Expired
- 1980-03-28 DE DE8080101671T patent/DE3061573D1/de not_active Expired
- 1980-04-08 PL PL1980223327A patent/PL123695B1/pl unknown
- 1980-04-08 CA CA000349318A patent/CA1157810A/en not_active Expired
Also Published As
| Publication number | Publication date |
|---|---|
| NO153307C (no) | 1986-02-19 |
| EP0017188A1 (de) | 1980-10-15 |
| PL223327A1 (show.php) | 1981-01-16 |
| US4323438A (en) | 1982-04-06 |
| DE3061573D1 (en) | 1983-02-17 |
| NO153307B (no) | 1985-11-11 |
| NO800890L (no) | 1980-10-13 |
| CA1157810A (en) | 1983-11-29 |
| PL123695B1 (en) | 1982-11-30 |
| IN153771B (show.php) | 1984-08-18 |
| DE2914414A1 (de) | 1980-10-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE2113795C3 (de) | Elektrode für elektrolytische Verfahren als Sauerstoffanode | |
| DE2832664C2 (de) | Elektrolysezelle für die Elektrolyse von Meerwasser | |
| EP0089475B1 (de) | Beschichtete Ventilmetallanode zur elektrolytischen Gewinnung von Metallen oder Metalloxiden | |
| EP0017188B1 (de) | Anode für die Alkalichlorid-Elektrolyse und Verfahren zur Herstellung von Chlor | |
| DE1191136B (de) | Hygrometerzelle | |
| EP0036677A1 (de) | Elektrolysezelle | |
| DE2046479B2 (de) | Anodenanordnung in einer elektrolysezelle | |
| EP1388595B1 (de) | Verfahren und Vorrichtung zur Bildung von Erdalkalicarbonat | |
| DE3003927C2 (de) | Kathode für die elektrolytische Raffination von Kupfer | |
| DE2406085A1 (de) | Verfahren zur herstellung von verbundkoerpern und elektrolytische zellen mit derartigen verbundkoerpern | |
| DE3406797C2 (de) | Beschichtete Ventilmetallanode zur elektrolytischen Gewinnung von Metallen oder Metalloxiden | |
| DE2949495C2 (de) | Elektrode für Elektrolysezellen | |
| EP0753604B1 (de) | Anode zur elektrolytischen Gewinnung von Metallen | |
| DE2023751A1 (de) | Vorrichtung zum Elektrolysleren von Salzwasser | |
| DE2041250A1 (de) | Dimensionsstabile Anode | |
| DE1467075B2 (de) | Anode zur elektrolytischen Herstellung von Chlor | |
| DE2833499A1 (de) | Anode fuer elektrolytische zellen | |
| DE2232903C3 (de) | Verfahren zur elektrolytischen Raffination von Kupfer unter Verwendung von Titanelektroden | |
| DE2844558A1 (de) | Elektrode fuer die verwendung in einem elektrolytischen verfahren | |
| DE3443338C2 (de) | Kathode zur Herstellung von Elektrolyt-Mangandioxid | |
| DE3005795A1 (de) | Beschichtete metallanode zur elektrolytischen gewinnung von metallen | |
| DE4318533A1 (de) | Bipolare Elektrolysezelle | |
| DE1210426C2 (de) | Verfahren zur elektrolytischen Herstellung von Phosphin | |
| DE2311877A1 (de) | Elektroden fuer elektrochemische verfahren | |
| DE1240827B (de) | Grossflaechige, Schlitze bzw. OEffnungen aufweisende Elektrode fuer elektrochemischeProzesse, insbesondere fuer die Chloralkali-elektrolyse |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed | ||
| AK | Designated contracting states |
Designated state(s): BE DE FR GB IT NL SE |
|
| ITF | It: translation for a ep patent filed | ||
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): BE DE FR GB IT NL SE |
|
| ET | Fr: translation filed | ||
| REF | Corresponds to: |
Ref document number: 3061573 Country of ref document: DE Date of ref document: 19830217 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19890222 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19890224 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19890228 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 19890330 Year of fee payment: 10 |
|
| ITTA | It: last paid annual fee | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19890331 Year of fee payment: 10 Ref country code: GB Payment date: 19890331 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Effective date: 19900328 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Effective date: 19900329 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Effective date: 19900331 |
|
| BERE | Be: lapsed |
Owner name: BAYER A.G. Effective date: 19900331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19901001 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee | ||
| GBPC | Gb: european patent ceased through non-payment of renewal fee | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Effective date: 19901130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19901201 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| EUG | Se: european patent has lapsed |
Ref document number: 80101671.8 Effective date: 19910109 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |