CN113413359A - 慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法 - Google Patents
慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法 Download PDFInfo
- Publication number
- CN113413359A CN113413359A CN202110707748.5A CN202110707748A CN113413359A CN 113413359 A CN113413359 A CN 113413359A CN 202110707748 A CN202110707748 A CN 202110707748A CN 113413359 A CN113413359 A CN 113413359A
- Authority
- CN
- China
- Prior art keywords
- parts
- chinese medicine
- traditional chinese
- inner shell
- sinus
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000003814 drug Substances 0.000 title claims abstract description 70
- 239000000843 powder Substances 0.000 title claims abstract description 59
- 208000004396 mastitis Diseases 0.000 title claims abstract description 35
- 238000002360 preparation method Methods 0.000 title claims abstract description 12
- 241000045403 Astragalus propinquus Species 0.000 claims abstract description 16
- 235000006533 astragalus Nutrition 0.000 claims abstract description 16
- 239000009526 Fructus Akebiae Substances 0.000 claims abstract description 13
- 239000004863 Frankincense Substances 0.000 claims abstract description 12
- 244000048199 Hibiscus mutabilis Species 0.000 claims abstract description 12
- 235000003973 Hibiscus mutabilis Nutrition 0.000 claims abstract description 12
- 235000017309 Hypericum perforatum Nutrition 0.000 claims abstract description 11
- 244000141009 Hypericum perforatum Species 0.000 claims abstract description 11
- 235000005035 Panax pseudoginseng ssp. pseudoginseng Nutrition 0.000 claims abstract description 11
- 235000003140 Panax quinquefolius Nutrition 0.000 claims abstract description 11
- 235000008434 ginseng Nutrition 0.000 claims abstract description 11
- 240000000691 Houttuynia cordata Species 0.000 claims abstract description 10
- 235000013719 Houttuynia cordata Nutrition 0.000 claims abstract description 10
- 239000002994 raw material Substances 0.000 claims abstract description 10
- 235000003717 Boswellia sacra Nutrition 0.000 claims abstract description 8
- 240000007551 Boswellia serrata Species 0.000 claims abstract description 8
- 235000012035 Boswellia serrata Nutrition 0.000 claims abstract description 8
- 235000006965 Commiphora myrrha Nutrition 0.000 claims abstract description 8
- 240000007232 Illicium verum Species 0.000 claims abstract description 8
- 235000008227 Illicium verum Nutrition 0.000 claims abstract description 8
- 235000007265 Myrrhis odorata Nutrition 0.000 claims abstract description 8
- 210000000582 semen Anatomy 0.000 claims abstract description 8
- 241000031023 Amana edulis Species 0.000 claims abstract description 6
- 241000208340 Araliaceae Species 0.000 claims description 10
- 238000000034 method Methods 0.000 claims description 8
- 240000009023 Myrrhis odorata Species 0.000 claims description 7
- 239000000203 mixture Substances 0.000 claims description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 6
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 claims description 6
- 230000001954 sterilising effect Effects 0.000 claims description 6
- 238000004659 sterilization and disinfection Methods 0.000 claims description 6
- 241000766380 Iphigenia Species 0.000 claims description 5
- 239000002274 desiccant Substances 0.000 claims description 5
- 239000000341 volatile oil Substances 0.000 claims description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 5
- 241000717739 Boswellia sacra Species 0.000 claims description 4
- 241001057584 Myrrha Species 0.000 claims description 4
- 238000007789 sealing Methods 0.000 claims description 4
- 238000001256 steam distillation Methods 0.000 claims description 4
- 235000006463 Brassica alba Nutrition 0.000 claims description 3
- 241000202726 Bupleurum Species 0.000 claims description 3
- 235000003990 Monochoria hastata Nutrition 0.000 claims description 3
- 240000000178 Monochoria vaginalis Species 0.000 claims description 3
- 241001671204 Stemona Species 0.000 claims description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 3
- 239000012153 distilled water Substances 0.000 claims description 3
- 238000001035 drying Methods 0.000 claims description 3
- 238000001914 filtration Methods 0.000 claims description 3
- 238000002156 mixing Methods 0.000 claims description 3
- 229910052760 oxygen Inorganic materials 0.000 claims description 3
- 239000001301 oxygen Substances 0.000 claims description 3
- 244000128879 sarson Species 0.000 claims description 3
- 238000007873 sieving Methods 0.000 claims description 3
- UAJTZZNRJCKXJN-UHFFFAOYSA-M sodium;2-dodecoxy-2-oxoethanesulfonate Chemical compound [Na+].CCCCCCCCCCCCOC(=O)CS([O-])(=O)=O UAJTZZNRJCKXJN-UHFFFAOYSA-M 0.000 claims description 3
- 241001454294 Tetranychus Species 0.000 claims description 2
- 240000007311 Commiphora myrrha Species 0.000 abstract 1
- 244000131316 Panax pseudoginseng Species 0.000 abstract 1
- 230000000694 effects Effects 0.000 description 18
- 208000033809 Suppuration Diseases 0.000 description 16
- 230000001737 promoting effect Effects 0.000 description 13
- 230000008961 swelling Effects 0.000 description 11
- 206010061218 Inflammation Diseases 0.000 description 7
- 230000004054 inflammatory process Effects 0.000 description 7
- 230000000844 anti-bacterial effect Effects 0.000 description 6
- 230000002401 inhibitory effect Effects 0.000 description 6
- 230000003179 granulation Effects 0.000 description 5
- 238000005469 granulation Methods 0.000 description 5
- 230000035876 healing Effects 0.000 description 5
- 230000006651 lactation Effects 0.000 description 5
- 241000894006 Bacteria Species 0.000 description 4
- 230000003110 anti-inflammatory effect Effects 0.000 description 4
- 230000017531 blood circulation Effects 0.000 description 4
- 239000000284 extract Substances 0.000 description 4
- 230000001105 regulatory effect Effects 0.000 description 4
- 208000025865 Ulcer Diseases 0.000 description 3
- 230000000202 analgesic effect Effects 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 230000036039 immunity Effects 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 238000011084 recovery Methods 0.000 description 3
- 231100000331 toxic Toxicity 0.000 description 3
- 230000002588 toxic effect Effects 0.000 description 3
- 230000036269 ulceration Effects 0.000 description 3
- 208000002628 Granulomatous mastitis Diseases 0.000 description 2
- 206010066170 Plasma cell mastitis Diseases 0.000 description 2
- 241000191967 Staphylococcus aureus Species 0.000 description 2
- 206010000269 abscess Diseases 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 210000000481 breast Anatomy 0.000 description 2
- 210000000265 leukocyte Anatomy 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000017423 tissue regeneration Effects 0.000 description 2
- MBDOYVRWFFCFHM-SNAWJCMRSA-N (2E)-hexenal Chemical compound CCC\C=C\C=O MBDOYVRWFFCFHM-SNAWJCMRSA-N 0.000 description 1
- 241000358234 Balanophora japonica Species 0.000 description 1
- 241000282461 Canis lupus Species 0.000 description 1
- 206010007247 Carbuncle Diseases 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 206010062717 Increased upper airway secretion Diseases 0.000 description 1
- 241001479578 Packera contermina Species 0.000 description 1
- 241001250596 Pleione Species 0.000 description 1
- 208000004880 Polyuria Diseases 0.000 description 1
- 241000589517 Pseudomonas aeruginosa Species 0.000 description 1
- 241000194017 Streptococcus Species 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 230000005784 autoimmunity Effects 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 238000000546 chi-square test Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000003467 diminishing effect Effects 0.000 description 1
- 230000035619 diuresis Effects 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000002481 ethanol extraction Methods 0.000 description 1
- 230000003203 everyday effect Effects 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 229930003935 flavonoid Natural products 0.000 description 1
- 150000002215 flavonoids Chemical class 0.000 description 1
- 235000017173 flavonoids Nutrition 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000002949 hemolytic effect Effects 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 210000005075 mammary gland Anatomy 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 208000026435 phlegm Diseases 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 229940126680 traditional chinese medicines Drugs 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0087—Galenical forms not covered by A61K9/02 - A61K9/7023
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/23—Apiaceae or Umbelliferae (Carrot family), e.g. dill, chervil, coriander or cumin
- A61K36/233—Bupleurum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/28—Asteraceae or Compositae (Aster or Sunflower family), e.g. chamomile, feverfew, yarrow or echinacea
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/29—Berberidaceae (Barberry family), e.g. barberry, cohosh or mayapple
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/31—Brassicaceae or Cruciferae (Mustard family), e.g. broccoli, cabbage or kohlrabi
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/32—Burseraceae (Frankincense family)
- A61K36/324—Boswellia, e.g. frankincense
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/32—Burseraceae (Frankincense family)
- A61K36/328—Commiphora, e.g. mecca myrrh or balm of Gilead
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/34—Campanulaceae (Bellflower family)
- A61K36/344—Codonopsis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/36—Caryophyllaceae (Pink family), e.g. babysbreath or soapwort
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/38—Clusiaceae, Hypericaceae or Guttiferae (Hypericum or Mangosteen family), e.g. common St. Johnswort
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/48—Fabaceae or Leguminosae (Pea or Legume family); Caesalpiniaceae; Mimosaceae; Papilionaceae
- A61K36/481—Astragalus (milkvetch)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/78—Saururaceae (Lizard's-tail family)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/88—Liliopsida (monocotyledons)
- A61K36/898—Orchidaceae (Orchid family)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M31/00—Devices for introducing or retaining media, e.g. remedies, in cavities of the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/14—Drugs for genital or sexual disorders; Contraceptives for lactation disorders, e.g. galactorrhoea
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2236/00—Isolation or extraction methods of medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicine
- A61K2236/30—Extraction of the material
- A61K2236/33—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones
- A61K2236/331—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones using water, e.g. cold water, infusion, tea, steam distillation or decoction
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2236/00—Isolation or extraction methods of medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicine
- A61K2236/30—Extraction of the material
- A61K2236/33—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones
- A61K2236/333—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones using mixed solvents, e.g. 70% EtOH
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2236/00—Isolation or extraction methods of medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicine
- A61K2236/30—Extraction of the material
- A61K2236/39—Complex extraction schemes, e.g. fractionation or repeated extraction steps
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2236/00—Isolation or extraction methods of medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicine
- A61K2236/50—Methods involving additional extraction steps
- A61K2236/51—Concentration or drying of the extract, e.g. Lyophilisation, freeze-drying or spray-drying
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Health & Medical Sciences (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Botany (AREA)
- Medical Informatics (AREA)
- Alternative & Traditional Medicine (AREA)
- Mycology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Rheumatology (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Pain & Pain Management (AREA)
- Hematology (AREA)
- Gynecology & Obstetrics (AREA)
- Pregnancy & Childbirth (AREA)
- Endocrinology (AREA)
- Reproductive Health (AREA)
- Medicines Containing Plant Substances (AREA)
- Medicinal Preparation (AREA)
Abstract
本发明公开了一种慢性乳腺炎窦道中药引流条,包括引流壳体及填充在中药粉剂;所述引流壳体包括内壳及外壳,所述外壳可相对内壳旋转,所述内壳设置为齿轮柱状空腔结构,所述内壳均匀布置有若干凸起部,相邻两凸起部之间设置有一凹槽部,所述中药粉剂设置在内壳的凹槽部位置处。本发明还公开了一种用于根据权利要求1‑5任一项所述慢性乳腺炎窦道中药引流条的中药粉剂及其制备方法,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡5~30份、乳香5~30份、没药5~30份、绵黄芪5~30份、狼头花5~15份、霸天伞5~15份、鱼腥草5~15份、八角乌5~15份、木芙蓉5~15份、四叶参5~15份、贯叶金丝桃5~15份、预知子5~15份、白芥子5~15份、山慈菇10~30份。
Description
技术领域
本发明属于中药技术领域,具体涉及一种慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法。
背景技术
慢性乳腺炎又称非哺乳期乳腺炎,是一种良性的发生在非哺乳期的乳腺炎症,常见的两种亚型为浆细胞性乳腺炎和肉芽肿性乳腺炎,由于临床症状相似,发病阶段相似,病理也有时难以区分,所以临床治疗也相似。慢性乳腺炎发病机制较为复杂,目前研究认为与自身免疫和理化刺激等相关,临床具有病程长、易反复、对乳房外形影响较大等特点。相较于西医学激素、抗生素、手术的治疗方案,中医学治疗有一定特色及优势,众多临床研究也证实了中医药治疗的有效性。慢性乳腺炎一般分为四期:溢液期、肿块期、脓肿期和破溃期,破溃期往往见窦道形成,通往内部脓腔,该期对于乳房病灶最终愈合及复发有重要意义,若排脓不畅、新肉不生,会影响整个疾病病程及预后,目前临床常用的为纸捻引流、药线引流等方法,有一定的疗效,但是效果不佳,因此,本发明从中医学角度为解决,破溃期排脓不畅、新肉不生的问题。
发明内容
发明目的:针对现有技术中存在问题或不足,本发明提供一种慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法,来解决慢性乳腺炎尤其是破溃期所形成窦道排脓及恢复问题。
为实现上述发明目的,本发明的实施例提供一种慢性乳腺炎窦道中药引流条,其特征在于,包括引流壳体及填充在中药粉剂;所述引流壳体包括内壳及外壳,所述外壳可相对内壳旋转,所述内壳设置为齿轮柱状空腔结构,所述内壳均匀布置有若干凸起部,相邻两凸起部之间设置有一凹槽部,所述中药粉剂设置在内壳的凹槽部位置处。
进一步的,所述内壳在每一凸起部沿轴向方向均匀设置有多个通孔;所述内壳内部设置有海绵。
优选的,所述海绵中间位置设置有柱状干燥剂。
进一步的,所述外壳的前端位置处具有导向头部,所述导向头部设置为圆锥状结构。
进一步的,所述外壳和内壳的尾端均设置为圆柱状空腔结构的端部,所述内壳的端部延伸出外壳的端部2cm;所述外壳的外部外侧设置有固定抓持部,所述内壳的端部还设置有旋转把手且通过旋转把手可旋转内壳,所述内壳的端部还具有一可开闭的密封塞。
本发明的实施例还提供一种用于慢性乳腺炎窦道中药引流条的中药粉剂,其特征在于,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡5~30份、乳香5~30份、没药5~30份、绵黄芪5~30份、狼头花5~15份、霸天伞5~15份、鱼腥草5~15份、八角乌5~15份、木芙蓉5~15份、四叶参5~15份、贯叶金丝桃5~15份、预知子5~15份、白芥子5~15份、山慈菇10~30份。
进一步的,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡7~15份、乳香10~20份、没药8~18份、绵黄芪10~22份、狼头花7~10份、霸天伞8~12份、鱼腥草7~10份、八角乌7~12份、木芙蓉7~10份、四叶参7~12份、贯叶金丝桃9~12份、预知子9~12份、白芥子7~10份、山慈菇15~20份。
优选的,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡12份、乳香18份、没药13份、绵黄芪15份、狼头花9份、霸天伞9份、鱼腥草8份、八角乌9份、木芙蓉9份、四叶参8份、贯叶金丝桃10份、预知子10份、白芥子9份、山慈菇18份。
本发明的实施例另外还提供一种慢性乳腺炎窦道中药引流条用中药粉剂的制备方法,其特征在于,包括以下制备过程:
(1)将配方量的绵黄芪粉碎,制成绵黄芪粉,加入65%~90%乙醇提取两次,每次2~3小时,过滤,收集第一提取液;
(2)将配方量的鱼腥草、八角乌、乳香、没药切片,用水蒸汽蒸馏法提取其挥发油,收集为第二提取液;
(3)将配方量的柴胡、狼头花、霸天伞、木芙蓉、四叶参、贯叶金丝桃、预知子、白芥子、山慈菇混合后加入足量的蒸馏水,煎煮30~60分钟,过80目筛,得第三提取液;
(4)将步骤(1)所得第一提取液和步骤(2)所得第二提取液加入步骤(3)制得的第三提取液内,采取柜式减压干燥器减压干燥法,制得干燥药粉;
(5)利用中药超微粉碎机将步骤(4)所得药粉进一步粉碎成100目以上的细粉;
(6)在臭氧浓度50g/m3、气流速度40m/s、气源氧气纯度>85%、灭菌温度小于40℃条件下对步骤(5)所得细粉进行臭氧灭菌,最大条件保存药物成分,制备成中药粉剂。
优选的,所述步骤(5)中制得的细粉要求:100%过100目筛,95%过120目筛。
本发明的上述技术方案的有益效果如下:
(1)本发明的慢性乳腺炎窦道中药引流条,在未使用时,外壳与内壳之间呈封闭状态,中药粉剂置于外壳与内壳的凹槽部位置处;使用时,将该中药引流条置于窦道内,旋转内壳,将中药粉剂与外部窦道接触,中药粉剂利于恢复。
(2)本发明的中药粉剂中,本发明采用的上述中药原料具有以下特点:柴胡,有活血止痛、消肿生肌的作用,其提取物能够镇痛、消炎、升高白细胞,促进肉芽生长;绵黄芪,具有益气升阳,托毒排脓,敛疮生肌的功效,其有效成分具有抑菌、抗炎、镇痛的作用,还能够促进组织新生;乳香、没药具有活血行气止痛、消肿生肌的功效,其有效成分具有镇痛、消炎、升高白细胞的作用,能够加速炎症渗出排泄,促进伤口愈合。预知子,有疏肝理气,活血止痛,散结的功效,现代药理研究表明预知子具有抑菌、免疫调节等作用。贯叶金丝桃:有疏肝解郁,清热利湿,消肿止痛的功效,其有效成分具有抗菌镇痛的作用。狼头花:功用清热解毒,排脓消肿,通乳。其水提取物有效成分具有抗炎、镇痛、调节免疫的作用。霸天伞具有解毒托疮,活血消肿,排脓生肌的功效。其有效成分多种黄酮类成分具有抑菌、镇痛、抗炎消肿及促进组织修复的作用,其提取液对G+及G-菌均有一定抑菌作用。鱼腥草,具有清热解毒,消痈排脓的功效,其有效成分具有抗炎镇痛作用,挥发油对乙型溶血性链球菌、金黄色葡萄球菌、铜绿假单胞菌、大肠埃希菌均有一定的抑制作用,其中对金黄色葡萄球菌抑制作用最大。八角乌具有清热解毒,凉血止血,散结消肿的功效,其叶挥发油中主要成分为已烯醛,根含千里光酸,具有抗炎抑菌镇痛作用。木芙蓉具有清热解毒,消肿排脓的作用,其有效成分具有抗炎抑菌的作用。四叶参具有解毒消肿,排脓,通乳的作用,其水提物具有抑菌,调节免疫的作用。白芥子具有散结通络止痛的作用,其有效成分具有抑菌作用。山慈菇具有清热解毒,化痰散结的作用,其有效成分具有抗炎、抗菌的作用。
(3)本发明的中药粉剂制备方法过程中,通过将材料分成多组,采用不同的提取方式(乙醇提取、水蒸汽蒸馏法、煎煮方式)分别进行有效成分的提取,能够有效保护所提取药物的有效成分,临床疗效更优。
附图说明
图1为本发明的慢性乳腺炎窦道中药引流条的结构示意图;
图2为图1的A-A剖视图。
附图标记说明:1、内壳;2、外壳;3、凸起部;4、海绵;5、柱状干燥剂;6、导向头部;7、固定抓持部;8、旋转把手;9、中药粉剂。
具体实施方式
为使本发明要解决的技术问题、技术方案和优点更加清楚,下面将结合具体实施例进行详细描述。
一种慢性乳腺炎窦道中药引流条,包括引流壳体及填充在中药粉剂9;所述引流壳体包括内壳1及外壳2,所述外壳2可相对内壳1旋转,所述内壳1设置为齿轮柱状空腔结构,所述内壳1均匀布置有若干凸起部3,相邻两凸起部3之间设置有一凹槽部,所述中药粉剂9设置在内壳1的凹槽部位置处。该中药引流条在使用时,通过导向头部6将引流壳体置入窦道内,通过旋转内壳1,实现中药粉剂9与窦道内相接触,利于窦道内部恢复。
在本实施例中,所述内壳1在每一凸起部3沿轴向方向均匀设置有多个通孔;所述内壳1内部设置有海绵4。通过在内壳1的凸起部上设置有若干通孔,实现海绵4对外侧窦道内的脓液吸附,利于窦道内部恢复。
优选的,所述海绵4中间位置设置有柱状干燥剂5,柱状干燥剂5对海绵4进行吸附,延长海绵4使用时间。
在本实施例中,所述外壳2的前端位置处具有导向头部6,所述导向头部6设置为圆锥状结构,导向头部6呈圆锥状,便于整个中药引流条进入窦道内。
在本实施例中,所述外壳2和内壳1的尾端均设置为圆柱状空腔结构的端部,所述内壳2的端部延伸出外壳2的端部2cm;所述外壳2的外部外侧设置有固定抓持部7,所述内壳1的端部还设置有旋转把手8且通过旋转把手8可旋转内壳1,所述内壳1的端部还具有一可开闭的密封塞。本发明使用时,一手握持外壳端部的固定抓持部,另外一只手旋转把手来旋转内壳,优选的,可以通过刻度线的方式来确定所需旋转的角度,可作为本发明的补充,将内壳中的中药粉剂与窦道接触,从而起到治疗的作用;通过打开密封塞,可以更换海绵。
本发明还提供一种用于慢性乳腺炎窦道中药引流条的中药粉剂,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡12份、乳香18份、没药13份、绵黄芪15份、狼头花9份、霸天伞9份、鱼腥草8份、八角乌9份、木芙蓉9份、四叶参8份、贯叶金丝桃10份、预知子10份、白芥子9份、山慈菇18份。
按照上述配方,取配方量的各中药原料,按照下述制备方法制备慢性乳腺炎窦道中药引流条用中药粉剂,包括以下制备过程:
(1)将配方量的绵黄芪粉碎,制成绵黄芪粉,加入65%~90%乙醇提取两次,每次2~3小时,过滤,收集第一提取液;
(2)将配方量的鱼腥草、八角乌、乳香、没药切片,用水蒸汽蒸馏法提取其挥发油,收集为第二提取液;
(3)将配方量的柴胡、狼头花、霸天伞、木芙蓉、四叶参、贯叶金丝桃、预知子、白芥子、山慈菇混合后加入足量的蒸馏水,煎煮30~60分钟,过80目筛,得第三提取液;
(4)将步骤(1)所得第一提取液和步骤(2)所得第二提取液加入步骤(3)制得的第三提取液内,采取柜式减压干燥器减压干燥法,制得干燥药粉;
(5)利用中药超微粉碎机将步骤(4)所得药粉进一步粉碎成100目以上的细粉;所制得的细粉要求:100%过100目筛,95%过120目筛。
(6)在臭氧浓度50g/m3、气流速度40m/s、气源氧气纯度>85%、灭菌温度小于40℃条件下对步骤(5)所得细粉进行臭氧灭菌,最大条件保存药物成分,制备成中药粉剂。
利用上述配方和方法制备的慢性乳腺炎窦道中药引流条经过大鼠实验证明无毒副作用。
下面结合实验数据以证明本发明制备的中药粉剂可用于慢性乳腺炎窦道形成。
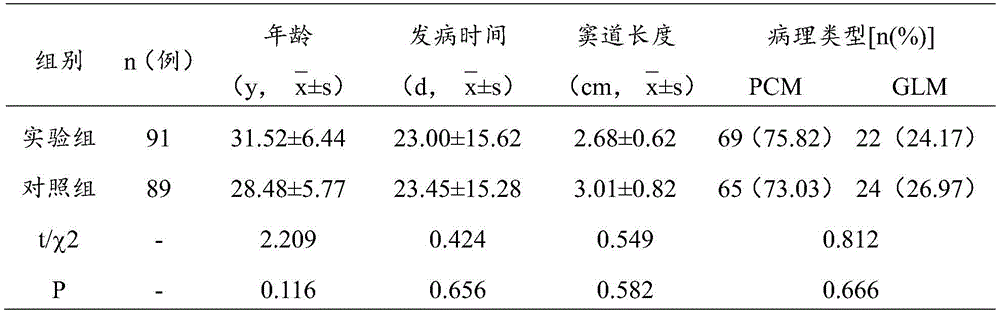
临床实验纳入南通市中医院2018~2020年门诊及住院慢性乳腺炎窦道形成患者共计180人,按照随机数字表法均分为实验组(91人)和对照组(89人),实验组和对照组一般资料(年龄、发病时间、病理类型、窦道长度)无统计学差异,实验组和对照组的统计数据如下表1所示。
表中,PCM就是浆细胞性乳腺炎,GLM是肉芽肿性乳腺炎,是非哺乳期乳腺炎的两种典型类型,t是T检验的统计量,χ2是卡方检验的统计量,P为对结果的判定,P>0.05,这两组数据差异没有统计学意义。
治疗方法:实验组将本发明制备的引流条置入窦道,旋转内壳的使药粉与脓腔接触,每日更换海绵内芯,三日更换引流条,直至无脓液,后窦道内以棉签塞入本发明中药粉剂,直至痊愈;对照组采用传统八二丹药线引流法。
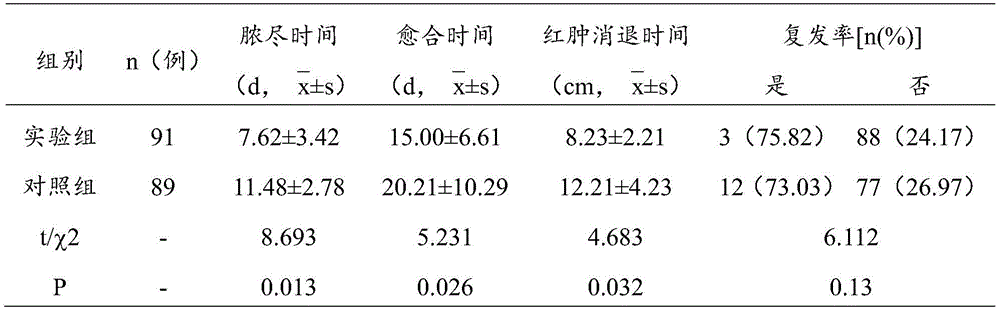
观察指标:①脓尽时间:以无脓液流出为准,计算治疗时长;②愈合时间:窦道完全愈合的时长;③红肿消退时间:窦道周围红肿消退的时间;④复发率:半年后复查,统计复发率。实验结果如表2所示:
表2实验组和对照组在脓尽、愈合、红肿消退时间及复发率对比表
如表2的实验结果显示,实验组在运用本发明后,相对于传统八二丹药线引流法治疗,能够加速脓液生成与排出,促进窦道愈合,红肿消退更为迅速,复发率更低,差异具有统计学意义(P<0.05)。
以上所述是本发明的优选实施方式,应当指出,对于本技术领域的普通技术人员来说,在不脱离本发明所述原理的前提下,还可以作出若干改进和润饰,这些改进和润饰也应视为本发明的保护范围。
Claims (10)
1.一种慢性乳腺炎窦道中药引流条,其特征在于,包括引流壳体及填充在中药粉剂;所述引流壳体包括内壳及外壳,所述外壳可相对内壳旋转,所述内壳设置为齿轮柱状空腔结构,所述内壳均匀布置有若干凸起部,相邻两凸起部之间设置有一凹槽部,所述中药粉剂设置在内壳的凹槽部位置处。
2.根据权利要求1所述的一种慢性乳腺炎窦道中药引流条,其特征在于,所述内壳在每一凸起部沿轴向方向均匀设置有多个通孔;所述内壳内部设置有海绵。
3.根据权利要求2所述的一种慢性乳腺炎窦道中药引流条,其特征在于,所述海绵中间位置设置有柱状干燥剂。
4.根据权利要求1所述的一种慢性乳腺炎窦道中药引流条,其特征在于,所述外壳的前端位置处具有导向头部,所述导向头部设置为圆锥状结构。
5.根据权利要求1所述的一种慢性乳腺炎窦道中药引流条,其特征在于,所述外壳和内壳的尾端均设置为圆柱状空腔结构的端部,所述内壳的端部延伸出外壳的端部2cm;所述外壳的外部外侧设置有固定抓持部,所述内壳的端部还设置有旋转把手且通过旋转把手可旋转内壳,所述内壳的端部还具有一可开闭的密封塞。
6.一种用于根据权利要求1-5任一项所述慢性乳腺炎窦道中药引流条的中药粉剂,其特征在于,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡5~30份、乳香5~30份、没药5~30份、绵黄芪5~30份、狼头花5~15份、霸天伞5~15份、鱼腥草5~15份、八角乌5~15份、木芙蓉5~15份、四叶参5~15份、贯叶金丝桃5~15份、预知子5~15份、白芥子5~15份、山慈菇10~30份。
7.根据权利要求1所述的一种慢性乳腺炎窦道中药引流条用中药粉剂,其特征在于,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡7~15份、乳香10~20份、没药8~18份、绵黄芪10~22份、狼头花7~10份、霸天伞8~12份、鱼腥草7~10份、八角乌7~12份、木芙蓉7~10份、四叶参7~12份、贯叶金丝桃9~12份、预知子9~12份、白芥子7~10份、山慈菇15~20份。
8.根据权利要求1所述一种慢性乳腺炎窦道中药引流条用中药粉剂,其特征在于,所述中药粉剂取下述重量配比的主要原料制备而成:柴胡12份、乳香18份、没药13份、绵黄芪15份、狼头花9份、霸天伞9份、鱼腥草8份、八角乌9份、木芙蓉9份、四叶参8份、贯叶金丝桃10份、预知子10份、白芥子9份、山慈菇18份。
9.一种根据权利要求6-8任一项所述的慢性乳腺炎窦道中药引流条用中药粉剂的制备方法,其特征在于,包括以下制备过程:
(1)将配方量的绵黄芪粉碎,制成绵黄芪粉,加入65%~90%乙醇提取两次,每次2~3小时,过滤,收集第一提取液;
(2)将配方量的鱼腥草、八角乌、乳香、没药切片,用水蒸汽蒸馏法提取其挥发油,收集为第二提取液;
(3)将配方量的柴胡、狼头花、霸天伞、木芙蓉、四叶参、贯叶金丝桃、预知子、白芥子、山慈菇混合后加入足量的蒸馏水,煎煮30~60分钟,过80目筛,得第三提取液;
(4)将步骤(1)所得第一提取液和步骤(2)所得第二提取液加入步骤(3)制得的第三提取液内,采取柜式减压干燥器减压干燥法,制得干燥药粉;
(5)利用中药超微粉碎机将步骤(4)所得药粉进一步粉碎成100目以上的细粉;
(6)在臭氧浓度50g/m3、气流速度40m/s、气源氧气纯度>85%、灭菌温度小于40℃条件下对步骤(5)所得细粉进行臭氧灭菌,最大条件保存药物成分,制备成中药粉剂。
10.根据权利要求9所述的一种慢性乳腺炎窦道中药引流条用中药粉剂的制备方法,其特征在于,所述步骤(5)中制得的细粉要求:100%过100目筛,95%过120目筛。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110707748.5A CN113413359B (zh) | 2021-06-24 | 2021-06-24 | 慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110707748.5A CN113413359B (zh) | 2021-06-24 | 2021-06-24 | 慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113413359A true CN113413359A (zh) | 2021-09-21 |
| CN113413359B CN113413359B (zh) | 2023-08-29 |
Family
ID=77716579
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110707748.5A Active CN113413359B (zh) | 2021-06-24 | 2021-06-24 | 慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113413359B (zh) |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4250882A (en) * | 1979-01-26 | 1981-02-17 | Medical Dynamics, Inc. | Wound drainage device |
| US4642105A (en) * | 1984-02-21 | 1987-02-10 | Toter Kimberly A M | Gastric drainage system |
| TW393323B (en) * | 1997-03-31 | 2000-06-11 | Fischell Robert | Stent delivery catheter system |
| CN101361894A (zh) * | 2008-08-29 | 2009-02-11 | 蒋保珍 | 治疗乳腺炎症的药物 |
| CN102151325A (zh) * | 2011-04-13 | 2011-08-17 | 苑学 | 一种治疗乳腺疾病的中药 |
| CN102552432A (zh) * | 2012-03-17 | 2012-07-11 | 张磊磊 | 一种治疗乳腺炎的外用中草药 |
| CN102973897A (zh) * | 2012-12-06 | 2013-03-20 | 赵辉 | 一种治疗乳腺增生及乳腺炎的中药组合物 |
| DE202014006977U1 (de) * | 2014-08-26 | 2014-10-28 | Norbert Neubauer | Drainagemittel zur Durchführung von Vakuumwundbehandlungen |
| CN204411114U (zh) * | 2014-12-18 | 2015-06-24 | 青岛明月生物医用材料有限公司 | 一种医用引流条 |
| CN104826036A (zh) * | 2015-06-10 | 2015-08-12 | 沈洪彦 | 一种治疗乳腺增生的复合中药组合物及制备方法 |
| CN206081313U (zh) * | 2016-07-28 | 2017-04-12 | 高丽彩 | 一种阴道给药装置 |
| CN111973720A (zh) * | 2020-09-29 | 2020-11-24 | 禹州济德医院 | 一种治疗乳腺炎、乳腺增生的中药组合物及其制备方法 |
| DE202021000349U1 (de) * | 2021-01-30 | 2021-02-19 | Norbert Neubauer | Drainagestrip - Matte |
| DE202021000555U1 (de) * | 2021-02-13 | 2021-03-11 | Norbert Neubauer | Drainagestrip-Strangdrainage |
| CN112472978A (zh) * | 2020-11-30 | 2021-03-12 | 重庆市人民医院 | 一种窦道换药器 |
-
2021
- 2021-06-24 CN CN202110707748.5A patent/CN113413359B/zh active Active
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4250882A (en) * | 1979-01-26 | 1981-02-17 | Medical Dynamics, Inc. | Wound drainage device |
| US4642105A (en) * | 1984-02-21 | 1987-02-10 | Toter Kimberly A M | Gastric drainage system |
| TW393323B (en) * | 1997-03-31 | 2000-06-11 | Fischell Robert | Stent delivery catheter system |
| CN101361894A (zh) * | 2008-08-29 | 2009-02-11 | 蒋保珍 | 治疗乳腺炎症的药物 |
| CN102151325A (zh) * | 2011-04-13 | 2011-08-17 | 苑学 | 一种治疗乳腺疾病的中药 |
| CN102552432A (zh) * | 2012-03-17 | 2012-07-11 | 张磊磊 | 一种治疗乳腺炎的外用中草药 |
| CN102973897A (zh) * | 2012-12-06 | 2013-03-20 | 赵辉 | 一种治疗乳腺增生及乳腺炎的中药组合物 |
| DE202014006977U1 (de) * | 2014-08-26 | 2014-10-28 | Norbert Neubauer | Drainagemittel zur Durchführung von Vakuumwundbehandlungen |
| CN204411114U (zh) * | 2014-12-18 | 2015-06-24 | 青岛明月生物医用材料有限公司 | 一种医用引流条 |
| CN104826036A (zh) * | 2015-06-10 | 2015-08-12 | 沈洪彦 | 一种治疗乳腺增生的复合中药组合物及制备方法 |
| CN206081313U (zh) * | 2016-07-28 | 2017-04-12 | 高丽彩 | 一种阴道给药装置 |
| CN111973720A (zh) * | 2020-09-29 | 2020-11-24 | 禹州济德医院 | 一种治疗乳腺炎、乳腺增生的中药组合物及其制备方法 |
| CN112472978A (zh) * | 2020-11-30 | 2021-03-12 | 重庆市人民医院 | 一种窦道换药器 |
| DE202021000349U1 (de) * | 2021-01-30 | 2021-02-19 | Norbert Neubauer | Drainagestrip - Matte |
| DE202021000555U1 (de) * | 2021-02-13 | 2021-03-11 | Norbert Neubauer | Drainagestrip-Strangdrainage |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113413359B (zh) | 2023-08-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN105663243A (zh) | 一种治疗鼻炎和/或鼻窦炎的中药组合物及其药物 | |
| CN109223637B (zh) | 山豆根复方中药牙膏及制备方法 | |
| CN100471509C (zh) | 一种用于治疗中耳炎、外耳道炎的药物及其制备方法 | |
| CN101632754B (zh) | 一种治疗咽炎的中药组合物及其制备方法 | |
| CN114246878A (zh) | 一种中药提取物组合物及其制备方法和应用 | |
| CN1813917A (zh) | 一种治疗急性分泌性中耳炎的药物及其制备方法 | |
| CN113413359A (zh) | 慢性乳腺炎窦道中药引流条、其所用中药粉剂及制备方法 | |
| CN105232603A (zh) | 一种艾叶提取物的制备方法 | |
| CN106581256B (zh) | 一种用于治疗鼻窦炎的中药组合物、药物制剂及其制备方法 | |
| CN109528998A (zh) | 一种妇科外用洗消药品及其制备方法 | |
| CN113730474A (zh) | 一种治疗咳嗽变异性哮喘的中药配方 | |
| CN111514230A (zh) | 一种麻杏宣肺中药胶囊及其制备方法 | |
| CN1207050C (zh) | 治疗慢性鼻炎的中药及其生产方法 | |
| CN106214804B (zh) | 一种用于祛痤疮的中药组合物及其应用 | |
| CN109045219B (zh) | 一种用于治疗母兔乳房炎的红茂草复方外用膏剂 | |
| CN112972578A (zh) | 一种治疗牛皮癣的膏剂及其制备方法 | |
| CN101053593B (zh) | 胜红清热颗粒及其制备方法 | |
| CN108771702A (zh) | 一种用于人体肛周皮肤的外用熏洗抑菌剂 | |
| CN103520315A (zh) | 一种治疗妇科阴道炎的药物组合物及其制备方法 | |
| CN116392518B (zh) | 东紫苏及其提取物在制备慢性压力脑区病变药物中的应用 | |
| CN110664934B (zh) | 抗菌止痒的中药组合物及其制备方法和应用 | |
| CN106138745B (zh) | 一种用于治疗鼻炎的中药制剂及其制备方法 | |
| CN107875269B (zh) | 一种治疗妇科炎症的中药组合物及其制备方法 | |
| CN106039149A (zh) | 一种治疗前列腺疾病的药物及其制备方法 | |
| CN106177117A (zh) | 治疗慢性鼻炎的中药滴剂及其制备方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |