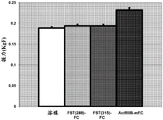
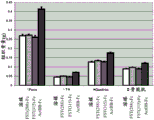
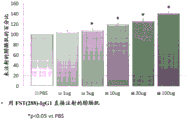
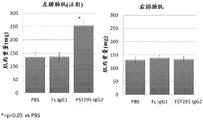
CN106795224B - 用促滤泡素抑制素多肽治疗病症的方法和组合物 - Google Patents
用促滤泡素抑制素多肽治疗病症的方法和组合物 Download PDFInfo
- Publication number
- CN106795224B CN106795224B CN201580041740.2A CN201580041740A CN106795224B CN 106795224 B CN106795224 B CN 106795224B CN 201580041740 A CN201580041740 A CN 201580041740A CN 106795224 B CN106795224 B CN 106795224B
- Authority
- CN
- China
- Prior art keywords
- follistatin
- fst
- muscle
- polypeptides
- seq
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4702—Regulators; Modulating activity
- C07K14/4703—Inhibitors; Suppressors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/04—Drugs for disorders of the muscular or neuromuscular system for myasthenia gravis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/06—Anabolic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4707—Muscular dystrophy
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K19/00—Hybrid peptides, i.e. peptides covalently bound to nucleic acids, or non-covalently bound protein-protein complexes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/1703—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- A61K38/1709—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/475—Growth factors; Growth regulators
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biochemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Gastroenterology & Hepatology (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Zoology (AREA)
- Physical Education & Sports Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Neurology (AREA)
- General Chemical & Material Sciences (AREA)
- Toxicology (AREA)
- Immunology (AREA)
- Epidemiology (AREA)
- Endocrinology (AREA)
- Marine Sciences & Fisheries (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
- Medicinal Preparation (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110533308.2A CN113583104A (zh) | 2014-06-04 | 2015-06-04 | 促滤泡素抑制素多肽、其组合物及其使用方法 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462007908P | 2014-06-04 | 2014-06-04 | |
| US62/007908 | 2014-06-04 | ||
| PCT/US2015/034245 WO2015187977A1 (en) | 2014-06-04 | 2015-06-04 | Methods and compositions for treatment of disorders with follistatin polypeptides |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110533308.2A Division CN113583104A (zh) | 2014-06-04 | 2015-06-04 | 促滤泡素抑制素多肽、其组合物及其使用方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN106795224A CN106795224A (zh) | 2017-05-31 |
| CN106795224B true CN106795224B (zh) | 2021-05-18 |
Family
ID=54767384
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201580041740.2A Expired - Fee Related CN106795224B (zh) | 2014-06-04 | 2015-06-04 | 用促滤泡素抑制素多肽治疗病症的方法和组合物 |
| CN202110533308.2A Pending CN113583104A (zh) | 2014-06-04 | 2015-06-04 | 促滤泡素抑制素多肽、其组合物及其使用方法 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110533308.2A Pending CN113583104A (zh) | 2014-06-04 | 2015-06-04 | 促滤泡素抑制素多肽、其组合物及其使用方法 |
Country Status (12)
| Country | Link |
|---|---|
| US (2) | US10023621B2 (cg-RX-API-DMAC7.html) |
| EP (2) | EP3152237B1 (cg-RX-API-DMAC7.html) |
| JP (3) | JP6568110B2 (cg-RX-API-DMAC7.html) |
| KR (4) | KR102305109B1 (cg-RX-API-DMAC7.html) |
| CN (2) | CN106795224B (cg-RX-API-DMAC7.html) |
| AU (2) | AU2015269333B2 (cg-RX-API-DMAC7.html) |
| BR (1) | BR112016028520A2 (cg-RX-API-DMAC7.html) |
| CA (1) | CA2950754C (cg-RX-API-DMAC7.html) |
| EA (2) | EA202090632A1 (cg-RX-API-DMAC7.html) |
| MA (2) | MA51075A (cg-RX-API-DMAC7.html) |
| MX (2) | MX382908B (cg-RX-API-DMAC7.html) |
| WO (1) | WO2015187977A1 (cg-RX-API-DMAC7.html) |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7612041B2 (en) | 2005-11-23 | 2009-11-03 | Acceleron Pharma Inc. | Isolated activin-binding ActRIIa polypeptide comprising the SEQ ID NO: 7 and uses for promoting bone growth |
| US20100015144A1 (en) | 2008-06-26 | 2010-01-21 | Acceleron Pharma Inc. | Methods for dosing an activin-actriia antagonist and monitoring of treated patients |
| EA201590719A1 (ru) | 2013-01-25 | 2015-12-30 | Шир Хьюман Дженетик Терапис, Инк. | Фоллистатин в лечении мышечной дистрофии дюшенна |
| JP6568110B2 (ja) | 2014-06-04 | 2019-08-28 | アクセルロン ファーマ, インコーポレイテッド | フォリスタチンポリペプチドによる障害の処置のための方法および組成物 |
| US10010498B2 (en) | 2014-06-04 | 2018-07-03 | Acceleron Pharma Inc. | Methods for treatment of amyotrophic lateral sclerosis with follistatin fusion proteins |
| BR122023023170A2 (pt) | 2014-06-13 | 2024-02-20 | Acceleron Pharma Inc. | Uso de um antagonista de actrii no tratamento ou prevenção de úlcera cutânea associada com beta-talassemia |
| JP2018510637A (ja) * | 2015-03-26 | 2018-04-19 | アクセルロン ファーマ, インコーポレイテッド | フォリスタチン関連融合タンパク質およびその使用 |
| KR20180137487A (ko) * | 2016-03-04 | 2018-12-27 | 샤이어 휴먼 지네틱 테라피즈 인크. | 재조합 폴리스타틴-fc 융합 단백질 및 듀시엔형 근이영양증 치료에서의 용도 |
| US10905766B2 (en) | 2017-03-23 | 2021-02-02 | Oregon State University | Therapeutic compositions and methods for treatment of muscle wasting diseases |
| KR101822633B1 (ko) * | 2017-08-22 | 2018-01-26 | (주)진셀팜 | 폴리스타틴 유래 생리활성 펩타이드, 및 이의 용도 |
| WO2019046903A1 (en) * | 2017-09-08 | 2019-03-14 | Baker Heart and Diabetes Institute | THERAPEUTIC METHOD FOR INCREASING MUSCLE MASS IN A SUBJECT |
| CN108218985B (zh) * | 2017-12-12 | 2021-04-30 | 山西农业大学 | 一种利用特征多肽制备fst蛋白特异性抗体的方法及其应用 |
| AU2019233929B2 (en) * | 2018-03-16 | 2025-05-29 | Immusoft Corporation | B cells genetically engineered to secrete follistatin and methods of using the same to treat follistatin-related diseases, conditions, disorders and to enhance muscle growth and strength |
| WO2019178759A1 (zh) * | 2018-03-21 | 2019-09-26 | 维多利亚生物医学控股股份有限公司 | 促进局部肌肉增长、减缓或防止局部肌肉萎缩的组合物及其用途 |
| WO2019191204A1 (en) * | 2018-03-28 | 2019-10-03 | Acceleron Pharma Inc. | Follistatin polypeptides for the treatment of muscle contracture |
| CA3100734A1 (en) * | 2018-05-17 | 2019-11-21 | Housey Pharmaceutical Research Laboratories, L.L.C. | Inhibition of follistatin |
| WO2020046466A1 (en) * | 2018-08-29 | 2020-03-05 | Myos Rens Technology, Inc. | Methods for alleviating, inhibiting or reversing muscle disuse atrophy in mammals |
| WO2022098812A1 (en) | 2020-11-04 | 2022-05-12 | Keros Therapeutics, Inc. | Methods of treating iron overload |
| EP4540274A1 (en) | 2022-06-15 | 2025-04-23 | UCB Biopharma SRL | Follistatin-fc fusion proteins |
| CA3259309A1 (en) | 2022-06-15 | 2023-12-21 | UCB Biopharma SRL | Fusion protein for the prevention, treatment or improvement of kidney disease |
| WO2024121351A1 (en) * | 2022-12-07 | 2024-06-13 | Lodberg Andreas | Extracellular matrix binding compounds for the localized loading of therapeutic or diagnostic agents |
| WO2025157262A1 (en) * | 2024-01-25 | 2025-07-31 | Sichuan Real & Best Biotech Co., Ltd. | Nucleic acids encoding follistatin and uses thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1993048A (zh) * | 2002-02-21 | 2007-07-04 | 惠氏公司 | 包含促滤泡素抑制素结构域的蛋白质 |
| WO2009158035A3 (en) * | 2008-06-26 | 2010-06-03 | Acceleron Pharma Inc. | Methods for dosing an activin-actriia antagonist and monitoring of treated patients |
Family Cites Families (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0272253A4 (en) | 1986-03-07 | 1990-02-05 | Massachusetts Inst Technology | METHOD FOR IMPROVING GLYCOPROTE INSTABILITY. |
| US5182375A (en) | 1987-08-28 | 1993-01-26 | The Salk Institute For Biological Studies | DNA encoding follistatin |
| US5041538A (en) | 1987-08-28 | 1991-08-20 | The Salk Institute For Biological Studies | Mammalian follistatin |
| ATE288923T1 (de) * | 1991-02-08 | 2005-02-15 | Progenics Pharm Inc | Cd4-gamma2- und cd4-igg2-chimären |
| WO1994006456A1 (en) | 1992-09-16 | 1994-03-31 | Genentech, Inc. | Protection against liver damage by hgf |
| US6686198B1 (en) | 1993-10-14 | 2004-02-03 | President And Fellows Of Harvard College | Method of inducing and maintaining neuronal cells |
| JPH09503673A (ja) | 1993-10-14 | 1997-04-15 | プレジデント・アンド・フエローズ・オブ・ハーバード・カレツジ | ニューロン細胞の誘導および維持法 |
| US5541087A (en) | 1994-09-14 | 1996-07-30 | Fuji Immunopharmaceuticals Corporation | Expression and export technology of proteins as immunofusins |
| US5545616A (en) | 1994-09-22 | 1996-08-13 | Genentech, Inc. | Method for predicting and/or preventing preterm labor |
| GB2306481A (en) | 1995-10-21 | 1997-05-07 | Univ Manchester | Pharmaceutical comprising a stimulator of activin and/or inhibin |
| US6656475B1 (en) | 1997-08-01 | 2003-12-02 | The Johns Hopkins University School Of Medicine | Growth differentiation factor receptors, agonists and antagonists thereof, and methods of using same |
| WO1999006559A1 (en) | 1997-08-01 | 1999-02-11 | The Johns Hopkins University School Of Medicine | Methods to identify growth differentiation factor (gdf) receptors |
| US6891082B2 (en) | 1997-08-01 | 2005-05-10 | The Johns Hopkins University School Of Medicine | Transgenic non-human animals expressing a truncated activintype II receptor |
| US6004937A (en) | 1998-03-09 | 1999-12-21 | Genetics Institute, Inc. | Use of follistatin to modulate growth and differentiation factor 8 [GDF-8] and bone morphogenic protein 11 [BMP-11] |
| DE60027135T2 (de) | 1999-01-21 | 2007-01-11 | Metamorphix, Inc. | Inhibitoren für wachstumdifferenzierungsfaktor und ihre anwendungen |
| EP1174149A1 (en) | 1999-04-19 | 2002-01-23 | Kyowa Hakko Kogyo Co., Ltd. | Proliferation inhibitor for androgen-independent tumor |
| WO2001009368A1 (en) | 1999-07-30 | 2001-02-08 | The General Hospital Corporation | Follistatin antagonists |
| JP4487376B2 (ja) | 2000-03-31 | 2010-06-23 | 味の素株式会社 | 腎疾患治療剤 |
| AUPR638101A0 (en) | 2001-07-13 | 2001-08-09 | Bioa Pty Limited | Composition and method for treatment of disease |
| JP4429729B2 (ja) | 2002-02-21 | 2010-03-10 | ワイス エルエルシー | Gasp1;フォリスタチンドメイン含有タンパク質 |
| JP4429728B2 (ja) | 2002-02-21 | 2010-03-10 | ワイス エルエルシー | フォリスタチン(follistatin)ドメイン含有タンパク質 |
| ATE474593T1 (de) | 2003-03-21 | 2010-08-15 | Celldex Therapeutics Ltd | Behandlung von allergischen erkrankungen unter verwendung eines modulators des notch signaling pathway |
| JP5110877B2 (ja) | 2003-06-02 | 2012-12-26 | ワイス・エルエルシー | 神経筋障害を処置するための、コルチコステロイドと組み合わせたミオスタチン(gdf8)インヒビターの使用 |
| US20070135336A1 (en) | 2003-09-15 | 2007-06-14 | De Kretser David | Follistatin isoforms and uses thereof |
| US7355018B2 (en) * | 2003-09-30 | 2008-04-08 | Regeneron Pharmaceuticals, Inc. | Modified IGF1 polypeptides with increased stability and potency |
| WO2005032578A1 (en) | 2003-10-06 | 2005-04-14 | Monash University | Therapeutic method |
| JP4688483B2 (ja) | 2004-04-15 | 2011-05-25 | 株式会社テクノネットワーク四国 | フォリスタチン変異体ポリペプチド |
| EP1771470B1 (en) | 2004-07-23 | 2013-06-26 | Acceleron Pharma Inc. | Actrii receptor polypeptides, methods and compositions |
| EP1778275A2 (en) | 2004-08-12 | 2007-05-02 | Wyeth | Combination therapy for diabetes, obesity, and cardiovascular diseases using gdf-8 inhibitors |
| NZ538097A (en) | 2005-02-07 | 2006-07-28 | Ovita Ltd | Method and compositions for improving wound healing |
| AU2006283725B2 (en) | 2005-08-19 | 2012-02-16 | The Trustees Of The University Of Pennsylvania | Antagonist antibodies against GDF-8 and uses in treatment of ALS and other GDF-8-associated disorders |
| MX2008007324A (es) | 2005-12-06 | 2009-03-04 | Amgen Inc | Usos de antagonistas de miostatina. |
| WO2008030367A2 (en) | 2006-09-01 | 2008-03-13 | The General Hospital Corporation | Selective myostatin inhibitors |
| EP2097095B1 (en) | 2006-11-29 | 2016-01-27 | Nationwide Children's Hospital | Myostatin inhibition for enhancing muscle and/or improving muscle function |
| US20100028332A1 (en) | 2006-12-18 | 2010-02-04 | Acceleron Pharma Inc. | Antagonists of actriib and uses for increasing red blood cell levels |
| EP2468290B1 (en) | 2006-12-18 | 2015-04-01 | Acceleron Pharma, Inc. | Activin-ActRII Antagonists for use in treating anemia |
| US8895016B2 (en) | 2006-12-18 | 2014-11-25 | Acceleron Pharma, Inc. | Antagonists of activin-actriia and uses for increasing red blood cell levels |
| DK2235064T3 (en) | 2008-01-07 | 2016-01-11 | Amgen Inc | A process for the preparation of heterodimeric Fc molecules using electrostatic control effects |
| JP5773868B2 (ja) * | 2008-06-26 | 2015-09-02 | アクセルロン ファーマ, インコーポレイテッド | アクチビン−ActRIIaのアンタゴニストおよび赤血球レベルを高めるためのその使用 |
| MX340451B (es) * | 2009-09-09 | 2016-07-08 | Acceleron Pharma Inc | Antagonistas de actriib y dosificacion y usos de los mismos. |
| JP5813114B2 (ja) * | 2010-08-25 | 2015-11-17 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | Il−18r1に対する抗体およびその使用 |
| MX352929B (es) | 2010-11-05 | 2017-12-13 | Zymeworks Inc | DISEÑO DE ANTICUERPOS HETERODIMÉRICOS ESTABLES CON MUTACIONES EN EL DOMINIO Fc. |
| EP2834260A4 (en) | 2012-04-02 | 2016-08-10 | Moderna Therapeutics Inc | MODIFIED POLYNUCLEOTIDES FOR THE PREPARATION OF MEMBRANE PROTEINS |
| ES2663993T3 (es) | 2012-05-17 | 2018-04-17 | Paranta Biosciences Limited | Uso de folistatina o de un inhibidor de activina para prevenir o tratar la disfunción del injerto de tejido |
| ES3049487T3 (en) * | 2012-06-27 | 2025-12-16 | Meiragtx Uk Ii Ltd | Combination for treating an inflammatory disorder |
| EA201590719A1 (ru) * | 2013-01-25 | 2015-12-30 | Шир Хьюман Дженетик Терапис, Инк. | Фоллистатин в лечении мышечной дистрофии дюшенна |
| WO2014187807A1 (en) * | 2013-05-21 | 2014-11-27 | Arcarios B.V. | Follistatin derivatives |
| US10010498B2 (en) | 2014-06-04 | 2018-07-03 | Acceleron Pharma Inc. | Methods for treatment of amyotrophic lateral sclerosis with follistatin fusion proteins |
| JP6568110B2 (ja) | 2014-06-04 | 2019-08-28 | アクセルロン ファーマ, インコーポレイテッド | フォリスタチンポリペプチドによる障害の処置のための方法および組成物 |
| JP2018510637A (ja) | 2015-03-26 | 2018-04-19 | アクセルロン ファーマ, インコーポレイテッド | フォリスタチン関連融合タンパク質およびその使用 |
| JP2020519291A (ja) | 2017-05-12 | 2020-07-02 | シャイアー ヒューマン ジェネティック セラピーズ インコーポレイテッド | 組み換えフォリスタチン−fc融合タンパク質およびデュシェンヌ型筋ジストロフィーの治療における使用 |
-
2015
- 2015-06-04 JP JP2016570806A patent/JP6568110B2/ja not_active Expired - Fee Related
- 2015-06-04 CA CA2950754A patent/CA2950754C/en active Active
- 2015-06-04 WO PCT/US2015/034245 patent/WO2015187977A1/en not_active Ceased
- 2015-06-04 EA EA202090632A patent/EA202090632A1/ru unknown
- 2015-06-04 KR KR1020207018965A patent/KR102305109B1/ko not_active Expired - Fee Related
- 2015-06-04 MA MA051075A patent/MA51075A/fr unknown
- 2015-06-04 EA EA201692529A patent/EA035455B1/ru not_active IP Right Cessation
- 2015-06-04 AU AU2015269333A patent/AU2015269333B2/en not_active Ceased
- 2015-06-04 CN CN201580041740.2A patent/CN106795224B/zh not_active Expired - Fee Related
- 2015-06-04 BR BR112016028520A patent/BR112016028520A2/pt not_active Application Discontinuation
- 2015-06-04 MX MX2016015868A patent/MX382908B/es unknown
- 2015-06-04 MA MA051074A patent/MA51074A/fr unknown
- 2015-06-04 KR KR1020177000297A patent/KR102077286B1/ko not_active Expired - Fee Related
- 2015-06-04 EP EP15802909.0A patent/EP3152237B1/en active Active
- 2015-06-04 EP EP20160287.7A patent/EP3721892B1/en active Active
- 2015-06-04 US US14/731,009 patent/US10023621B2/en active Active
- 2015-06-04 KR KR1020217029755A patent/KR20210119546A/ko not_active Ceased
- 2015-06-04 CN CN202110533308.2A patent/CN113583104A/zh active Pending
- 2015-06-04 KR KR1020207003791A patent/KR102132144B1/ko not_active Expired - Fee Related
-
2016
- 2016-12-01 MX MX2021006017A patent/MX2021006017A/es unknown
-
2018
- 2018-06-12 US US16/006,025 patent/US10954279B2/en active Active
-
2019
- 2019-08-01 JP JP2019142333A patent/JP6856716B2/ja not_active Expired - Fee Related
-
2020
- 2020-08-03 AU AU2020210315A patent/AU2020210315A1/en not_active Abandoned
-
2021
- 2021-03-18 JP JP2021044545A patent/JP2021095416A/ja active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1993048A (zh) * | 2002-02-21 | 2007-07-04 | 惠氏公司 | 包含促滤泡素抑制素结构域的蛋白质 |
| WO2009158035A3 (en) * | 2008-06-26 | 2010-06-03 | Acceleron Pharma Inc. | Methods for dosing an activin-actriia antagonist and monitoring of treated patients |
Non-Patent Citations (3)
| Title |
|---|
| Amita Datta-Mannan et al.An Engineered Human Follistatin Variant: Insights into the Pharmacokinetic and Pharmocodynamic Relationships of a Novel Molecule with Broad Therapeutic Potential.《Journal of Pharmacology and Experimental Therapeutics》.2013,第344卷(第3期),第616-623页. * |
| An Engineered Human Follistatin Variant: Insights into the Pharmacokinetic and Pharmocodynamic Relationships of a Novel Molecule with Broad Therapeutic Potential;Amita Datta-Mannan et al;《Journal of Pharmacology and Experimental Therapeutics》;20130301;第344卷(第3期);第616-623页 * |
| Follistatin: A Novel Therapeutic for the Improvement of Muscle Regenerations;Benjamin C.Yaden et al;《Journal of Pharmacology and Experimental Therapeutics》;20140313;第349卷(第2期);第355-371页 * |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN106795224B (zh) | 用促滤泡素抑制素多肽治疗病症的方法和组合物 | |
| US11497792B2 (en) | Methods for treatment of Duchenne muscular dystrophy with follistatin polypeptides | |
| DK2332977T3 (en) | ActRII receptor polypeptides | |
| EP3805259A1 (en) | Truncated actriib-fc fusion proteins | |
| TW202021980A (zh) | 衍生自ActRIIB的變體與其用途 | |
| CN107636154A (zh) | 促滤泡素抑制素相关的融合蛋白及其用途 | |
| WO2019191204A1 (en) | Follistatin polypeptides for the treatment of muscle contracture | |
| HK1238658A1 (en) | Methods and compositions for treatment of disorders with follistatin polypeptides | |
| HK1236544A1 (en) | Methods and compositions for treatment of disorders with follistatin polypeptides | |
| HK1236544B (en) | Methods and compositions for treatment of disorders with follistatin polypeptides | |
| HK1238658B (zh) | 用促滤泡素抑制素多肽治疗病症的方法和组合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1238658 Country of ref document: HK |
|
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20210518 |