CN103379957A - 来自火焰喷射热解的催化剂载体和用于自热丙烷脱氢的催化剂 - Google Patents
来自火焰喷射热解的催化剂载体和用于自热丙烷脱氢的催化剂 Download PDFInfo
- Publication number
- CN103379957A CN103379957A CN2012800086618A CN201280008661A CN103379957A CN 103379957 A CN103379957 A CN 103379957A CN 2012800086618 A CN2012800086618 A CN 2012800086618A CN 201280008661 A CN201280008661 A CN 201280008661A CN 103379957 A CN103379957 A CN 103379957A
- Authority
- CN
- China
- Prior art keywords
- catalyst
- particle
- carrier
- weight
- pyrolysis
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000003054 catalyst Substances 0.000 title claims abstract description 65
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 title claims description 22
- 238000006356 dehydrogenation reaction Methods 0.000 title claims description 19
- 239000001294 propane Substances 0.000 title claims description 11
- 238000005118 spray pyrolysis Methods 0.000 title description 8
- 239000002245 particle Substances 0.000 claims abstract description 44
- 239000002243 precursor Substances 0.000 claims abstract description 42
- 238000000197 pyrolysis Methods 0.000 claims abstract description 37
- 150000001875 compounds Chemical class 0.000 claims abstract description 34
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 claims abstract description 26
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims abstract description 20
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 claims abstract description 11
- 239000000443 aerosol Substances 0.000 claims abstract description 10
- 239000000377 silicon dioxide Substances 0.000 claims abstract description 9
- 238000000034 method Methods 0.000 claims description 31
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 24
- 238000002360 preparation method Methods 0.000 claims description 20
- 239000000203 mixture Substances 0.000 claims description 19
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 17
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 15
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 14
- 229910052746 lanthanum Inorganic materials 0.000 claims description 11
- 229910052718 tin Inorganic materials 0.000 claims description 11
- 229910052792 caesium Inorganic materials 0.000 claims description 10
- 238000007598 dipping method Methods 0.000 claims description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 10
- 238000001354 calcination Methods 0.000 claims description 9
- 238000010438 heat treatment Methods 0.000 claims description 9
- 238000005470 impregnation Methods 0.000 claims description 9
- 229910052697 platinum Inorganic materials 0.000 claims description 8
- 229910052726 zirconium Inorganic materials 0.000 claims description 8
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 claims description 7
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 claims description 7
- TVFDJXOCXUVLDH-UHFFFAOYSA-N caesium atom Chemical compound [Cs] TVFDJXOCXUVLDH-UHFFFAOYSA-N 0.000 claims description 7
- UQEAIHBTYFGYIE-UHFFFAOYSA-N hexamethyldisiloxane Chemical compound C[Si](C)(C)O[Si](C)(C)C UQEAIHBTYFGYIE-UHFFFAOYSA-N 0.000 claims description 7
- FZLIPJUXYLNCLC-UHFFFAOYSA-N lanthanum atom Chemical group [La] FZLIPJUXYLNCLC-UHFFFAOYSA-N 0.000 claims description 7
- 229910052622 kaolinite Inorganic materials 0.000 claims description 6
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 claims description 6
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 claims description 6
- 239000002253 acid Substances 0.000 claims description 5
- 238000001035 drying Methods 0.000 claims description 5
- OFBQJSOFQDEBGM-UHFFFAOYSA-N n-pentane Natural products CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 claims description 5
- 239000001273 butane Substances 0.000 claims description 3
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 claims description 3
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 claims 2
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 abstract 1
- 235000012239 silicon dioxide Nutrition 0.000 abstract 1
- 229910052814 silicon oxide Inorganic materials 0.000 abstract 1
- 229910052845 zircon Inorganic materials 0.000 abstract 1
- GFQYVLUOOAAOGM-UHFFFAOYSA-N zirconium(iv) silicate Chemical compound [Zr+4].[O-][Si]([O-])([O-])[O-] GFQYVLUOOAAOGM-UHFFFAOYSA-N 0.000 abstract 1
- 239000000243 solution Substances 0.000 description 23
- 239000007789 gas Substances 0.000 description 17
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 10
- 238000001694 spray drying Methods 0.000 description 8
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 229910052751 metal Inorganic materials 0.000 description 6
- 239000002184 metal Substances 0.000 description 6
- 229910004298 SiO 2 Inorganic materials 0.000 description 5
- 239000012159 carrier gas Substances 0.000 description 5
- 238000005516 engineering process Methods 0.000 description 5
- 239000002737 fuel gas Substances 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- 238000001556 precipitation Methods 0.000 description 5
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 5
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 4
- CYPPCCJJKNISFK-UHFFFAOYSA-J kaolinite Chemical compound [OH-].[OH-].[OH-].[OH-].[Al+3].[Al+3].[O-][Si](=O)O[Si]([O-])=O CYPPCCJJKNISFK-UHFFFAOYSA-J 0.000 description 4
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- YOBOXHGSEJBUPB-MTOQALJVSA-N (z)-4-hydroxypent-3-en-2-one;zirconium Chemical compound [Zr].C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O.C\C(O)=C\C(C)=O YOBOXHGSEJBUPB-MTOQALJVSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 238000002485 combustion reaction Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000001257 hydrogen Substances 0.000 description 3
- 229910052739 hydrogen Inorganic materials 0.000 description 3
- UADUAXMDVVGCGW-UHFFFAOYSA-N propanoic acid;zirconium Chemical compound [Zr].CCC(O)=O UADUAXMDVVGCGW-UHFFFAOYSA-N 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- HBHXRPHNNKAWQL-UHFFFAOYSA-N 2-ethylhexanoic acid;zirconium Chemical compound [Zr].CCCCC(CC)C(O)=O HBHXRPHNNKAWQL-UHFFFAOYSA-N 0.000 description 2
- DUFCMRCMPHIFTR-UHFFFAOYSA-N 5-(dimethylsulfamoyl)-2-methylfuran-3-carboxylic acid Chemical compound CN(C)S(=O)(=O)C1=CC(C(O)=O)=C(C)O1 DUFCMRCMPHIFTR-UHFFFAOYSA-N 0.000 description 2
- YPIFGDQKSSMYHQ-UHFFFAOYSA-N 7,7-dimethyloctanoic acid Chemical compound CC(C)(C)CCCCCC(O)=O YPIFGDQKSSMYHQ-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 150000007942 carboxylates Chemical group 0.000 description 2
- 238000006555 catalytic reaction Methods 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 150000002430 hydrocarbons Chemical class 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical group [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 2
- -1 isopropyl zirconia Chemical compound 0.000 description 2
- MRELNEQAGSRDBK-UHFFFAOYSA-N lanthanum(3+);oxygen(2-) Chemical compound [O-2].[O-2].[O-2].[La+3].[La+3] MRELNEQAGSRDBK-UHFFFAOYSA-N 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- 229910000510 noble metal Inorganic materials 0.000 description 2
- 239000012454 non-polar solvent Substances 0.000 description 2
- BPYXFMVJXTUYRV-UHFFFAOYSA-J octanoate;zirconium(4+) Chemical compound [Zr+4].CCCCCCCC([O-])=O.CCCCCCCC([O-])=O.CCCCCCCC([O-])=O.CCCCCCCC([O-])=O BPYXFMVJXTUYRV-UHFFFAOYSA-J 0.000 description 2
- 150000003891 oxalate salts Chemical class 0.000 description 2
- IJZKJRUCRGJGKO-UHFFFAOYSA-N oxalic acid;zirconium Chemical compound [Zr].OC(=O)C(O)=O IJZKJRUCRGJGKO-UHFFFAOYSA-N 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000002798 polar solvent Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 238000010791 quenching Methods 0.000 description 2
- 239000011877 solvent mixture Substances 0.000 description 2
- 239000007921 spray Substances 0.000 description 2
- GBNDTYKAOXLLID-UHFFFAOYSA-N zirconium(4+) ion Chemical compound [Zr+4] GBNDTYKAOXLLID-UHFFFAOYSA-N 0.000 description 2
- OBETXYAYXDNJHR-SSDOTTSWSA-M (2r)-2-ethylhexanoate Chemical compound CCCC[C@@H](CC)C([O-])=O OBETXYAYXDNJHR-SSDOTTSWSA-M 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- 229910052684 Cerium Inorganic materials 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 1
- 239000004258 Ethoxyquin Substances 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- NHTMVDHEPJAVLT-UHFFFAOYSA-N Isooctane Chemical compound CC(C)CC(C)(C)C NHTMVDHEPJAVLT-UHFFFAOYSA-N 0.000 description 1
- 229910002422 La(NO3)3·6H2O Inorganic materials 0.000 description 1
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 1
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 229910003902 SiCl 4 Inorganic materials 0.000 description 1
- 229910002808 Si–O–Si Inorganic materials 0.000 description 1
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 1
- 229910021627 Tin(IV) chloride Inorganic materials 0.000 description 1
- 229910007926 ZrCl Inorganic materials 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- CUJRVFIICFDLGR-UHFFFAOYSA-N acetylacetonate Chemical compound CC(=O)[CH-]C(C)=O CUJRVFIICFDLGR-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000004703 alkoxides Chemical group 0.000 description 1
- OBETXYAYXDNJHR-UHFFFAOYSA-N alpha-ethylcaproic acid Natural products CCCCC(CC)C(O)=O OBETXYAYXDNJHR-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- MMCOUVMKNAHQOY-UHFFFAOYSA-N carbonoperoxoic acid Chemical compound OOC(O)=O MMCOUVMKNAHQOY-UHFFFAOYSA-N 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- GWXLDORMOJMVQZ-UHFFFAOYSA-N cerium Chemical compound [Ce] GWXLDORMOJMVQZ-UHFFFAOYSA-N 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- 238000000975 co-precipitation Methods 0.000 description 1
- 239000007859 condensation product Substances 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000002242 deionisation method Methods 0.000 description 1
- JVSWJIKNEAIKJW-UHFFFAOYSA-N dimethyl-hexane Natural products CCCCCC(C)C JVSWJIKNEAIKJW-UHFFFAOYSA-N 0.000 description 1
- DECIPOUIJURFOJ-UHFFFAOYSA-N ethoxyquin Chemical compound N1C(C)(C)C=C(C)C2=CC(OCC)=CC=C21 DECIPOUIJURFOJ-UHFFFAOYSA-N 0.000 description 1
- 229940093500 ethoxyquin Drugs 0.000 description 1
- 235000019285 ethoxyquin Nutrition 0.000 description 1
- 238000010304 firing Methods 0.000 description 1
- 150000004675 formic acid derivatives Chemical class 0.000 description 1
- YKIAZARODMJWBV-UHFFFAOYSA-N formic acid;lanthanum Chemical compound [La].OC=O YKIAZARODMJWBV-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- OXHNIMPTBAKYRS-UHFFFAOYSA-H lanthanum(3+);oxalate Chemical compound [La+3].[La+3].[O-]C(=O)C([O-])=O.[O-]C(=O)C([O-])=O.[O-]C(=O)C([O-])=O OXHNIMPTBAKYRS-UHFFFAOYSA-H 0.000 description 1
- JLRJWBUSTKIQQH-UHFFFAOYSA-K lanthanum(3+);triacetate Chemical compound [La+3].CC([O-])=O.CC([O-])=O.CC([O-])=O JLRJWBUSTKIQQH-UHFFFAOYSA-K 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000000320 mechanical mixture Substances 0.000 description 1
- 239000003863 metallic catalyst Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- SYSQUGFVNFXIIT-UHFFFAOYSA-N n-[4-(1,3-benzoxazol-2-yl)phenyl]-4-nitrobenzenesulfonamide Chemical class C1=CC([N+](=O)[O-])=CC=C1S(=O)(=O)NC1=CC=C(C=2OC3=CC=CC=C3N=2)C=C1 SYSQUGFVNFXIIT-UHFFFAOYSA-N 0.000 description 1
- VRQWWCJWSIOWHG-UHFFFAOYSA-J octadecanoate;zirconium(4+) Chemical compound [Zr+4].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O VRQWWCJWSIOWHG-UHFFFAOYSA-J 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 150000001283 organosilanols Chemical class 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- CMXPERZAMAQXSF-UHFFFAOYSA-M sodium;1,4-bis(2-ethylhexoxy)-1,4-dioxobutane-2-sulfonate;1,8-dihydroxyanthracene-9,10-dione Chemical compound [Na+].O=C1C2=CC=CC(O)=C2C(=O)C2=C1C=CC=C2O.CCCCC(CC)COC(=O)CC(S([O-])(=O)=O)C(=O)OCC(CC)CCCC CMXPERZAMAQXSF-UHFFFAOYSA-M 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 150000003606 tin compounds Chemical class 0.000 description 1
- HPGGPRDJHPYFRM-UHFFFAOYSA-J tin(iv) chloride Chemical compound Cl[Sn](Cl)(Cl)Cl HPGGPRDJHPYFRM-UHFFFAOYSA-J 0.000 description 1
- YJGJRYWNNHUESM-UHFFFAOYSA-J triacetyloxystannyl acetate Chemical compound [Sn+4].CC([O-])=O.CC([O-])=O.CC([O-])=O.CC([O-])=O YJGJRYWNNHUESM-UHFFFAOYSA-J 0.000 description 1
- 239000002912 waste gas Substances 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/08—Heat treatment
- B01J37/082—Decomposition and pyrolysis
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G25/00—Compounds of zirconium
- C01G25/02—Oxides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J21/00—Catalysts comprising the elements, oxides, or hydroxides of magnesium, boron, aluminium, carbon, silicon, titanium, zirconium, or hafnium
- B01J21/06—Silicon, titanium, zirconium or hafnium; Oxides or hydroxides thereof
- B01J21/066—Zirconium or hafnium; Oxides or hydroxides thereof
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J21/00—Catalysts comprising the elements, oxides, or hydroxides of magnesium, boron, aluminium, carbon, silicon, titanium, zirconium, or hafnium
- B01J21/06—Silicon, titanium, zirconium or hafnium; Oxides or hydroxides thereof
- B01J21/08—Silica
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/38—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals
- B01J23/54—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals combined with metals, oxides or hydroxides provided for in groups B01J23/02 - B01J23/36
- B01J23/56—Platinum group metals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/38—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals
- B01J23/54—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals combined with metals, oxides or hydroxides provided for in groups B01J23/02 - B01J23/36
- B01J23/56—Platinum group metals
- B01J23/62—Platinum group metals with gallium, indium, thallium, germanium, tin or lead
- B01J23/622—Platinum group metals with gallium, indium, thallium, germanium, tin or lead with germanium, tin or lead
- B01J23/626—Platinum group metals with gallium, indium, thallium, germanium, tin or lead with germanium, tin or lead with tin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/38—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals
- B01J23/54—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals combined with metals, oxides or hydroxides provided for in groups B01J23/02 - B01J23/36
- B01J23/56—Platinum group metals
- B01J23/63—Platinum group metals with rare earths or actinides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/90—Regeneration or reactivation
- B01J23/96—Regeneration or reactivation of catalysts comprising metals, oxides or hydroxides of the noble metals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/60—Catalysts, in general, characterised by their form or physical properties characterised by their surface properties or porosity
- B01J35/61—Surface area
- B01J35/613—10-100 m2/g
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/02—Impregnation, coating or precipitation
- B01J37/03—Precipitation; Co-precipitation
- B01J37/036—Precipitation; Co-precipitation to form a gel or a cogel
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/34—Irradiation by, or application of, electric, magnetic or wave energy, e.g. ultrasonic waves ; Ionic sputtering; Flame or plasma spraying; Particle radiation
- B01J37/349—Irradiation by, or application of, electric, magnetic or wave energy, e.g. ultrasonic waves ; Ionic sputtering; Flame or plasma spraying; Particle radiation making use of flames, plasmas or lasers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J38/00—Regeneration or reactivation of catalysts, in general
- B01J38/04—Gas or vapour treating; Treating by using liquids vaporisable upon contacting spent catalyst
- B01J38/10—Gas or vapour treating; Treating by using liquids vaporisable upon contacting spent catalyst using elemental hydrogen
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J38/00—Regeneration or reactivation of catalysts, in general
- B01J38/04—Gas or vapour treating; Treating by using liquids vaporisable upon contacting spent catalyst
- B01J38/12—Treating with free oxygen-containing gas
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B33/00—Silicon; Compounds thereof
- C01B33/113—Silicon oxides; Hydrates thereof
- C01B33/12—Silica; Hydrates thereof, e.g. lepidoic silicic acid
- C01B33/18—Preparation of finely divided silica neither in sol nor in gel form; After-treatment thereof
- C01B33/187—Preparation of finely divided silica neither in sol nor in gel form; After-treatment thereof by acidic treatment of silicates
- C01B33/193—Preparation of finely divided silica neither in sol nor in gel form; After-treatment thereof by acidic treatment of silicates of aqueous solutions of silicates
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C5/00—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms
- C07C5/32—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms by dehydrogenation with formation of free hydrogen
- C07C5/327—Formation of non-aromatic carbon-to-carbon double bonds only
- C07C5/333—Catalytic processes
- C07C5/3335—Catalytic processes with metals
- C07C5/3337—Catalytic processes with metals of the platinum group
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/12—Surface area
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2521/00—Catalysts comprising the elements, oxides or hydroxides of magnesium, boron, aluminium, carbon, silicon, titanium, zirconium or hafnium
- C07C2521/06—Silicon, titanium, zirconium or hafnium; Oxides or hydroxides thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2521/00—Catalysts comprising the elements, oxides or hydroxides of magnesium, boron, aluminium, carbon, silicon, titanium, zirconium or hafnium
- C07C2521/06—Silicon, titanium, zirconium or hafnium; Oxides or hydroxides thereof
- C07C2521/08—Silica
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2523/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00
- C07C2523/02—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00 of the alkali- or alkaline earth metals or beryllium
- C07C2523/04—Alkali metals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2523/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00
- C07C2523/10—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00 of rare earths
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2523/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00
- C07C2523/14—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00 of germanium, tin or lead
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2523/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00
- C07C2523/38—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00 of noble metals
- C07C2523/40—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00 of noble metals of the platinum group metals
- C07C2523/42—Platinum
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2523/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00
- C07C2523/38—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00 of noble metals
- C07C2523/54—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group C07C2521/00 of noble metals combined with metals, oxides or hydroxides provided for in groups C07C2523/02 - C07C2523/36
- C07C2523/56—Platinum group metals
- C07C2523/62—Platinum group metals with gallium, indium, thallium, germanium, tin or lead
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/52—Improvements relating to the production of bulk chemicals using catalysts, e.g. selective catalysts
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/584—Recycling of catalysts
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Inorganic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Optics & Photonics (AREA)
- Plasma & Fusion (AREA)
- Toxicology (AREA)
- Thermal Sciences (AREA)
- Dispersion Chemistry (AREA)
- Catalysts (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
Abstract
本发明提供一种制备催化剂载体粒子的方法,所述催化剂载体粒子含有二氧化锆和任选地氧化硅,所述方法包括以下步骤:(i)制备含有二氧化锆前体化合物和任选地二氧化硅前体化合物的溶液,(ii)将所述溶液转化成气溶胶,(iii)将气溶胶加入直接或间接加热的热解区中,(iv)进行热解,和(v)从热解气体分离出所形成的催化剂粒子。
Description
描述
本发明涉及氧化物催化剂载体和由其制备的催化剂粒子,其制备方法以及所述催化剂粒子用作脱氢催化剂的用途。
通过浸渍工艺或喷雾干燥制备脱氢催化剂的方法是已知的。在这些方法中,通过浸渍工艺将催化活性金属施用到氧化物载体或硅酸盐载体上,或者通过将共沉淀的氧化物前体进行喷雾干燥来制备催化剂。
DE-A 196 54 391描述了制备脱氢催化剂的方法,其中用Pt(NO3)2和Sn(OAc)2的溶液浸渍基本上单斜的ZrO2,或者用Pt(NO3)2的第一溶液浸渍ZrO2,然后用La(NO3)3的第二溶液浸渍。浸渍的载体进行干燥,然后煅烧。如此获得的催化剂用作脱氢催化剂,例如用于丙烷向丙烯的脱氢反应。
催化剂载体是按照常规方式通过溶胶-凝胶工艺、盐的沉淀、相应酸的脱水、干混、淤浆化或喷雾干燥制备的。例如,对于ZrO2·Al2O3·SiO2混合氧化物的制备,先通过合适的含锆前体的沉淀制备具有高水含量的通式ZrO2·xH2O的氧化锆。合适的锆前体例如是Zr(NO3)4、ZrOCl2或ZrCl4。实际的沉淀通过加入碱例如NaOH、KOH、Na2CO3和NH3进行,例如参见EP-A 0 849 224所述。
为了制备ZrO2·SiO2混合氧化物,含锆的前体可以与含硅的前体混合。非常合适的用于SiO2的前体例如是SiO2的含水溶胶,例如LudoxTM。这两种组分可以例如通过简单的机械混合或通过在喷雾塔中喷雾干燥来混合。
一种已知的通过火焰喷射热解制备金属催化剂的方法描述在Pisduangnawakij等,Applied Catalysis A:General3701-6,2009中。其中,含有铂和锡的前体化合物和载体氧化铝在二甲苯中的溶液被转化成气溶胶,将其在惰性载气中在热解反应器中在比前体化合物的分解温度更高的温度下处理,然后将所形成的细碎金属从载气分离出来。
本发明要解决的技术问题是提供一种便宜且省时的制备用于脱氢催化剂的氧化物载体的方法,其中所得的脱氢催化剂应当在活性和选择性方面与在现有技术中完全通过浸渍工艺或喷雾干燥制得的催化剂相当。
此问题通过一种制备催化剂载体粒子的方法实现,所述催化剂载体粒子含有二氧化锆和任选地氧化硅,所述方法包括以下步骤:
(i)制备含有二氧化锆和任选氧化硅的前体化合物的溶液,
(ii)将所述溶液转化成气溶胶,
(iii)将气溶胶加入直接或间接加热的热解区中,
(iv)进行热解,和
(v)从热解气体分离出所形成的催化剂粒子。
能形成氧化物的前体化合物是作为气溶胶加入热解区的。优选的是,加入热解区的气溶胶是通过仅仅一种溶液的雾化获得的,所述溶液含有所有的能形成氧化物的前体化合物。以此方式,总是确保所制得的粒子的组成是均匀和恒定的。在制备要被转化成气溶胶的溶液期间,各个组分因此优选选择使得在溶液中所含的能形成氧化物的前体能彼此均匀地溶解,直到溶液进行雾化。或者,也可以使用几种不同的溶液,其一起含有能形成氧化物的前体。所述一种或多种溶液可以同时含有极性和非极性的溶剂或溶剂混合物。
在热解区中,所述氧化物前体进行分解和/或氧化,同时形成氧化物。热解一般导致具有不同比表面的球形粒子。
在热解区中的温度是处于足够形成氧化物的温度,通常是500-2000℃。热解优选在900-1500℃的温度下进行。
热解反应器可以从外部间接地加热,例如通过电炉加热。由于在间接加热中需要从外部向内部的温度梯度,电炉必须比热解所需的温度显著更热。间接加热需要热稳定性炉材料和昂贵的反应器构造,但是所需的气体总量小于在火焰反应器情况下的气体总量。
在一个优选实施方案中,热解区通过火焰加热(火焰喷射热解)。热解区则包含点燃装置。对于直接加热,使用常规的可燃气体,但是优选使用氢气、甲烷或乙烯。在热解区中的温度可以通过可燃气体的量与气体总量之间的比率而按照需要调节。为了保持气体总量低、但能达到尽可能高的温度,也可以向热解区供应纯氧气来代替空气作为用于可燃气体的燃烧的氧气源。气体的总量也包含用于气溶胶的载气和气溶胶的已蒸发的溶剂。向热解区供应的一种或多种气溶胶优选直接加入火焰中。虽然空气一般优选作为用于气溶胶的载气,但是也可以使用氮气、CO2、O2或可燃气体,例如氢气、甲烷、乙烯、丙烷或丁烷。
在本发明方法的另一个实施方案中,热解区通过电子等离子体或感应等离子体加热。
火焰喷射热解设备通常包含用于要雾化的液体的储存容器,用于载气、可燃气体和含氧气体的进料管线,中心气溶胶喷嘴以及布置在其周围的环形燃烧器,用于气体-固体分离的装置,其包含过滤元件和用于固体的排料装置,以及用于废气的出口。这些粒子用骤冷气体冷却,例如用氮气或空气。
为了产生平衡的温度分布,燃烧空间是隔热的,所述燃烧空间优选是管形的。
由于热解,获得热解气体,其含有具有不同比表面的球形粒子。所得粒子的尺寸分布基本上直接来自被加入热解区的气溶胶的液滴谱和所用的一种或多种溶液的浓度。
优选,在从热解气体分离所形成的粒子之前,将热解气体冷却,从而排除粒子的烧结。因此,热解区优选含有冷却区,其与热解反应器的燃烧空间邻接。通常需要将热解气体和在其中所含的催化剂粒子冷却到约100-500℃的温度,这取决于所用的过滤元件。优选冷却到约100-150℃。在离开热解区之后,含有催化剂粒子且被部分冷却的热解气体进入用于从热解气体分离粒子的装置,此装置包含过滤元件。为了冷却,加入骤冷气体,例如氮气、空气或被水润湿的空气。
合适的能形成二氧化锆的前体化合物是醇盐,例如乙氧化锆(IV)、正丙氧化锆(IV)、异丙氧化锆(IV)、正丁氧化锆(IV)和叔丁氧化锆(IV)。在本发明方法的一个优选实施方案中,丙氧化锆(IV),优选作为在正丙醇中的溶液形式,用作ZrO2前体化合物。
其它合适的能形成二氧化锆的前体化合物是羧酸盐,例如乙酸锆、丙酸锆、草酸锆、辛酸锆、2-乙基己酸锆、新癸酸锆、乙酸锆、丙酸锆、草酸锆、辛酸锆、2-乙基己酸锆、新癸酸锆和/或硬脂酸锆、丙酸锆。在本发明方法的另一个优选实施方案中,乙酰丙酮酸锆(IV)用作前体化合物。
在一个实施方案中,前体化合物另外含有二氧化硅前体化合物。用于二氧化硅的可能前体是有机硅烷,以及SiCl4与低级醇或低级羧酸的反应产物。也可以使用上述有机硅烷和/或有机硅烷醇的具有Si-O-Si单元的缩合物。优选使用硅氧烷。也可以使用SiO2。在本发明方法的一个优选实施方案中,前体化合物含有六甲基二硅氧烷作为能形成二氧化硅的前体化合物。
极性和非极性的溶剂或溶剂混合物可以用于制备用于形成气溶胶的一种或多种溶液。
优选的极性溶剂是水、甲醇、乙醇、正丙醇、异丙醇、正丁醇、叔丁醇、正丙醇、正丁醇、乙醚、叔丁基甲基醚、四氢呋喃、C1-C8羧酸、乙酸乙酯以及它们的混合物。
在本发明方法的一个优选实施方案中,一种或多种前体化合物、优选全部的前体化合物溶解在乙酸、乙醇和水的混合物中。优选,此混合物含有30-75重量%的乙酸、30-75重量%的乙醇和0-20重量%的水。尤其是,乙酰丙酮酸锆(IV)和六甲基二硅氧烷溶解在乙酸、乙醇和水的混合物中。
优选的非极性溶剂是甲苯、二甲苯、正庚烷、正戊烷、辛烷、异辛烷、环己烷、乙酸甲酯、乙酸乙酯或乙酸丁酯,或它们的混合物。具有5-15个碳原子的烃或烃混合物也是合适的。二甲苯是尤其优选的。
尤其是,乙基己酸锆(IV)和六甲基二硅氧烷溶解在二甲苯中。
通过喷射热解获得的催化剂载体粒子优选具有36-70m2/g的比表面。
所得的催化剂载体粒子然后用含有铂、锡和至少一种选自镧和铯的其它元素的化合物的一种或多种溶液浸渍。浸渍后的催化剂载体粒子进行干燥和煅烧。
所以本发明还涉及一种制备催化剂粒子的方法,所述催化剂粒子含有在含二氧化锆的载体上的铂和锡和至少一种选自镧和铯的其它元素,其中所述方法包括步骤(i)至(v)以及另外包括以下步骤:
(vi)所得的催化剂载体粒子用含有铂、锡和至少一种选自镧和铯的其它元素的化合物的一种或多种溶液浸渍,
(vii)浸渍后的催化剂载体粒子进行干燥和煅烧。
通常,所用的前体化合物是能通过煅烧转化成相应氧化物的化合物。例如,合适的是相应金属的氢氧化物、碳酸盐、草酸盐、乙酸盐、氯化物或混合羟基碳酸盐。
通常,脱氢活性组分是通过浸渍施用的。但是,代替浸渍,脱氢活性组分也可以通过其它方法施用,例如喷洒金属盐前体。铂优选作为H2PtCl6或Pt(NO3)2使用。水和有机溶剂都适合用作溶剂。水和低级醇是特别合适的,例如甲醇和乙醇。
当使用贵金属作为脱氢活性组分时,合适的前体也是相应的贵金属溶胶,其可以通过公知方法之一制备,例如通过在稳定剂例如PVP和还原剂的存在下还原金属盐。关于制备技术的细节,可以参见德国专利申请DE195 00 366。
在催化剂中作为脱氢活性组分的铂的含量是0.01-5重量%,优选0.05-1重量%,尤其优选0.05-0.5重量%。
另外,催化剂含有至少0.01-10重量%、优选0.05-2重量%的锡。合适的锡化合物是羧酸盐,例如乙酸锡(II)、2-乙基己酸锡或氯化锡(II)。
在一个优选实施方案中,Pt的载荷是0.05-1重量%,Sn的载荷是0.05-2重量%。
此外,活性物质可以含有以下额外组分,其中含有至少铯或镧:
-铯和任选地钾,含量为0.1-10重量%。能通过煅烧转化成氧化物的化合物,例如氢氧化物、碳酸盐、草酸盐、乙酸盐或甲酸盐,用作铯或钾的氧化物前体。
-镧和任选地铈,含量为0.1-10重量%。如果使用镧,例如氧化镧碳酸盐、La(OH)3、La2(CO3)3、La(NO3)3,、甲酸镧、乙酸镧和草酸镧适合用作前体盐。
在催化剂载体上施用活性组分之后,煅烧在400-1000℃的温度下进行,优选500-700℃,尤其优选550-650℃。
本发明还涉及可通过本发明方法获得的载体和催化剂粒子。它们优选具有20-70m2/g的比表面。
在一个优选实施方案中,催化剂载体具有以下百分比组成:30-99.5重量%的ZrO2,0.5-25重量%的SiO2。催化剂粒子另外含有0.1-1重量%的Pt,0.1-10重量%的Sn、La和/或Cs,基于载体的质量计,其中含有至少Sn和至少La或Cs。
本发明还涉及催化剂粒子作为氢化催化剂或脱氢催化剂的用途。链烷、例如丁烷和丙烷、以及乙基苯优选进行脱氢。
本发明催化剂用于将丙烷脱氢成丙烯的用途是尤其优选的。
通过以下实施例更详细地说明本发明。
实施例
所用的化学品:
乙酰丙酮酸锆Zr(acac)2(98%)
丙氧化锆(IV)Zr(OPr)4(70%在1-丙醇中)
六甲基二硅氧烷(HMDSO)(98%)
CsNO3
KNO3
SnCl2·2H2O
La(NO3)3·6H2O
乙酸(100%)、乙醇(96%)和水(去离子)的混合物
二甲苯(异构体的混合物)
制备前体化合物的溶液
溶剂是HoAc:EtOH:H2O,其重量比为4.6:4.6:1。乙酸-乙醇混合物是新鲜制备的。用于Sn和Zr的前体化合物溶解在所述溶剂中。
实施例中的前体化合物的极性溶液的组成列在表1中。
表1:用于非极性混合物(二甲苯)的前体化合物溶液的组成
| [g] | 物质 | 纯度[重量%] |
| 374.40 | 乙基己酸锆(IV) | 97 |
| 10.11 | 六甲基二硅氧烷 | 99 |
通过火焰喷射热解制备催化剂载体粒子
含有前体化合物的溶液是通过活塞泵经由双组分喷嘴供应的,并用相应量的空气雾化。为了达到相应的温度,有时使用来自乙烯-空气混合物的支持火焰,其是经由位于喷嘴周围的环形燃烧器提供的。压降保持恒定为1.1巴。
火焰合成条件总结在表2中。
表2:通过火焰喷射热解制备的载体的试验参数
袋式过滤器用于分离这些粒子。这些过滤器可以通过向滤袋施加5巴压力的氮气吹扫来清洁。
火焰合成的载体的浸渍
浸渍是根据EP 1 074 301中的实施例4所述进行的。将SnCl2和H2PtCl6在乙醇中的溶液倒在筛分级分为1-2mm的火焰合成的SiO2/ZrO2载体上。过量的溶液在旋转蒸发器中除去,并且固体物质进行干燥和煅烧。为此,加入CsNO3和La(NO3)3的水溶液,并除去上清液。在干燥和煅烧之后,获得BET表面积为23m2的催化剂。
参比催化剂
根据EP 1 074 301的参比催化剂含有95重量%ZrO2、5重量%SiO2(载体)、0.5重量%Pt、1重量%Sn、3%La、0.5重量%Cs和0.2重量%K(活性和促进剂金属,基于载体质量计),其根据实施例4所述通过湿化学方式制备。载体是通过将根据溶胶/凝胶工艺沉淀所得的氧化物混合物进行喷雾干燥制备的。
催化检测
丙烷脱氢反应是在约600℃下进行:21Nl/h总气体(20Nl/h丙烷,1Nl/h氮气作为内标),5g/h水。再生在400℃下进行:2小时21Nl/h N2+4Nl/h空气;2小时25Nl/h空气;1小时25Nl/h氢气。
在催化试验中研究了转化率、长期稳定性和对形成丙烯的选择性。从火焰合成以及随后浸渍获得的催化剂显示在最佳操作条件下,在丙烷向丙烯的自热脱氢中达到48%转化率和95%选择性。
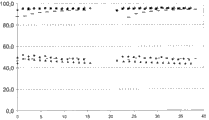
为了对比,图1显示了参比催化剂(-)和本发明催化剂(■)的活性和选择性,其中参比催化剂具有通过沉淀和喷雾干燥制备的载体,本发明催化剂具有从火焰合成衍生的载体,其中额外组分在每种情况下通过浸渍施用。也显示了具有相同组成的仅仅通过火焰合成的催化剂的结果(▲)。对于丙烷向丙烯的自热脱氢反应,在横轴上绘制单位为小时的时间,在纵轴上绘制转化率(40-50%)和选择性(>80%)。
可见,三种催化剂具有相当的性能。参比催化剂具有较低的初始选择性。但是,在数星期的试验周期中,其等同于本发明的催化剂。因此,火焰合成的催化剂以及在额外元素的湿化学施用之后的火焰合成的载体(根据本发明)的性能就象老化的催化剂那样,后者的载体是通过喷雾干燥制备的。
Claims (16)
1.一种制备催化剂载体粒子的方法,所述催化剂载体粒子含有二氧化锆和任选地氧化硅,所述方法包括以下步骤:
(i)制备含有二氧化锆前体化合物和任选地二氧化硅前体化合物的溶液,
(ii)将所述溶液转化成气溶胶,
(iii)将气溶胶加入直接或间接加热的热解区中,
(iv)进行热解,
(v)从热解气体分离出所形成的催化剂粒子。
2.权利要求1的方法,其中热解区用火焰加热。
3.权利要求1或2的方法,其中二氧化锆前体化合物含有乙基己酸锆(IV)。
4.权利要求1-3中任一项的方法,其中二氧化硅前体化合物含有六甲基二硅氧烷。
5.权利要求1-4中任一项的方法,其中二氧化锆前体化合物含有丙氧化锆(IV)。
6.权利要求1-5中任一项的方法,其中一种或多种前体化合物溶解在乙酸、乙醇和水的混合物中。
7.权利要求1-6中任一项的方法,其中一种或多种前体化合物溶解在二甲苯中。
8.权利要求1-7中任一项的方法,其中热解在900-1500℃的温度下进行。
9.催化剂载体粒子,其可以通过根据权利要求1-8中任一项所述的方法获得。
10.一种制备催化剂粒子的方法,所述催化剂粒子含有在含二氧化锆的载体上的铂和锡和至少一种选自镧和铯的其它元素,所述方法包括如权利要求1-8中任一项所述的步骤(i)至(v)以及另外包括以下步骤:
(vi)所得的催化剂载体粒子用含有铂、锡和至少一种选自镧和铯的其它元素的化合物的一种或多种溶液浸渍,
(vii)浸渍后的催化剂载体粒子进行干燥和煅烧。
11.可通过权利要求10的方法获得的催化剂粒子。
12.权利要求11的催化剂粒子,其中所述催化剂粒子含有0.05-1重量%的Pt和0.05-2重量%的Sn。
13.权利要求11或12的催化剂粒子,其具有20-70m2/g的比表面。
14.权利要求11-13中任一项的催化剂粒子,其含有作为载体的30-99.5重量%的ZrO2和0.5-25重量%的SiO2,0.1-1重量%的Pt,0.1-10重量%的Sn、La和/或Cs,基于载体的质量计,其中含有至少Sn和至少La或Cs。
15.权利要求11-14中任一项的催化剂粒子作为脱氢催化剂的用途。
16.权利要求15的用途,所述催化剂粒子用于丙烷向丙烯的脱氢反应或丁烷向丁烯的脱氢反应。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP11151958 | 2011-01-25 | ||
| EP111519583 | 2011-01-25 | ||
| PCT/IB2012/050303 WO2012101567A2 (de) | 2011-01-25 | 2012-01-23 | Katalysatorträger aus der flammen-spraypyrolyse und katalysator für die autotherme propandehydrierung |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN103379957A true CN103379957A (zh) | 2013-10-30 |
Family
ID=46581216
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2012800086618A Pending CN103379957A (zh) | 2011-01-25 | 2012-01-23 | 来自火焰喷射热解的催化剂载体和用于自热丙烷脱氢的催化剂 |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP2667968A2 (zh) |
| JP (1) | JP2014511259A (zh) |
| KR (1) | KR20140006909A (zh) |
| CN (1) | CN103379957A (zh) |
| BR (1) | BR112013019047A2 (zh) |
| WO (1) | WO2012101567A2 (zh) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106753391A (zh) * | 2016-11-28 | 2017-05-31 | 新沂市中诺新材料科技有限公司 | 一种用于钝化修复土壤中重金属的纳米材料及制备方法 |
| CN111821996A (zh) * | 2019-04-18 | 2020-10-27 | 清华大学 | 高效抗水热和抗硫性能的脱汞催化剂及其制备方法与应用 |
| CN115069246A (zh) * | 2021-03-15 | 2022-09-20 | 中国石油化工股份有限公司 | 一种负载型银催化剂及其制备方法和应用 |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8933262B2 (en) | 2011-05-24 | 2015-01-13 | Basf Se | Process for preparing polyisocyanates from biomass |
| CN103566933B (zh) * | 2012-08-08 | 2016-09-07 | 中国石油化工股份有限公司 | 一种醋酸酯加氢制乙醇用催化剂及其制备方法 |
| CN104248953A (zh) * | 2013-06-28 | 2014-12-31 | 中国石油化工股份有限公司 | 一种醋酸酯加氢制乙醇用催化剂及其制备方法 |
| KR101814451B1 (ko) | 2015-11-10 | 2018-01-04 | 희성촉매 주식회사 | 안정화 활성금속 착체를 이용한 직쇄형 탄화수소류 탈수소화 촉매 제조방법 |
| KR101716170B1 (ko) | 2015-11-10 | 2017-03-14 | 희성촉매 주식회사 | 안정화 활성금속 복합체를 이용한 직쇄형 경질탄화수소류 탈수소화 촉매 제조방법 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6057030A (en) * | 1997-07-21 | 2000-05-02 | Kanebo Ltd. | Porous ceramic body and kiln furniture made from a porous ceramic body |
| CN1283519A (zh) * | 1999-08-06 | 2001-02-14 | Basf公司 | 具有双峰孔半径分布的催化剂 |
| CN101433863A (zh) * | 2007-11-15 | 2009-05-20 | 中国石油化工股份有限公司 | 一种复合氧化物载体及其制备方法 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3701805A (en) * | 1971-09-27 | 1972-10-31 | Pfizer | Preparation of citraconic and itaconic acids |
| CN1513600A (zh) * | 2003-08-19 | 2004-07-21 | 丁章云 | 一种催化剂活性载体及其制备 |
| CN101204654A (zh) * | 2006-12-20 | 2008-06-25 | 中国科学院生态环境研究中心 | 一种低温催化氧化苯系物的负载型贵金属催化剂及其制备方法 |
-
2012
- 2012-01-23 WO PCT/IB2012/050303 patent/WO2012101567A2/de active Application Filing
- 2012-01-23 BR BR112013019047A patent/BR112013019047A2/pt not_active IP Right Cessation
- 2012-01-23 KR KR1020137022258A patent/KR20140006909A/ko not_active Application Discontinuation
- 2012-01-23 EP EP12739435.1A patent/EP2667968A2/de not_active Withdrawn
- 2012-01-23 CN CN2012800086618A patent/CN103379957A/zh active Pending
- 2012-01-23 JP JP2013549930A patent/JP2014511259A/ja not_active Withdrawn
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6057030A (en) * | 1997-07-21 | 2000-05-02 | Kanebo Ltd. | Porous ceramic body and kiln furniture made from a porous ceramic body |
| CN1283519A (zh) * | 1999-08-06 | 2001-02-14 | Basf公司 | 具有双峰孔半径分布的催化剂 |
| CN101433863A (zh) * | 2007-11-15 | 2009-05-20 | 中国石油化工股份有限公司 | 一种复合氧化物载体及其制备方法 |
Non-Patent Citations (1)
| Title |
|---|
| SUKANYA PISDUANGDAW ET AL: ""Characteristics and catalytic properties of Pt–Sn/Al2O3 nanoparticles synthesized by one-step flame spray pyrolysis in the dehydrogenation of propane"", 《APPLIED CATALYSIS A:GENERAL》, vol. 370, 13 August 2009 (2009-08-13), pages 1 - 6 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106753391A (zh) * | 2016-11-28 | 2017-05-31 | 新沂市中诺新材料科技有限公司 | 一种用于钝化修复土壤中重金属的纳米材料及制备方法 |
| CN111821996A (zh) * | 2019-04-18 | 2020-10-27 | 清华大学 | 高效抗水热和抗硫性能的脱汞催化剂及其制备方法与应用 |
| CN111821996B (zh) * | 2019-04-18 | 2022-11-29 | 清华大学 | 高效抗水热和抗硫性能的脱汞催化剂及其制备方法与应用 |
| CN115069246A (zh) * | 2021-03-15 | 2022-09-20 | 中国石油化工股份有限公司 | 一种负载型银催化剂及其制备方法和应用 |
| CN115069246B (zh) * | 2021-03-15 | 2023-08-15 | 中国石油化工股份有限公司 | 一种负载型银催化剂及其制备方法和应用 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2667968A2 (de) | 2013-12-04 |
| WO2012101567A3 (de) | 2013-01-03 |
| BR112013019047A2 (pt) | 2017-03-28 |
| WO2012101567A2 (de) | 2012-08-02 |
| JP2014511259A (ja) | 2014-05-15 |
| KR20140006909A (ko) | 2014-01-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103379957A (zh) | 来自火焰喷射热解的催化剂载体和用于自热丙烷脱氢的催化剂 | |
| CN1220659C (zh) | 烃类的脱氢方法 | |
| CN101218025B (zh) | 层状组合物及其制备和应用方法 | |
| CN108236955A (zh) | 一种草酸二甲酯加氢合成乙醇用催化剂的制备方法以及由此得到的催化剂和其应用 | |
| Wang et al. | Effect of promoters on the selective hydrogenolysis of glycerol over Pt/W-containing catalysts | |
| JP2012527339A (ja) | モノリス触媒とその使用方法 | |
| CN108144610B (zh) | 火焰喷射裂解法制备的铜基加氢催化剂及其制备和应用 | |
| JP2015525668A (ja) | イリジウム含有活性物質の存在下における炭化水素の二酸化炭素改質のための高圧方法 | |
| AU2012343061A1 (en) | Fischer-Tropsch synthesis cobalt nano-catalyst based on porous material confinement, and preparation method therefor | |
| WO2015183059A1 (ko) | 활성 및 수명 특성이 향상된 피셔-트롭쉬 촉매의 제조방법 | |
| CN101190413B (zh) | 一种石脑油重整催化剂及其制备方法 | |
| KR20070122571A (ko) | 세리아-함유 지지체 상의 금촉매 | |
| EP1283746B1 (en) | A catalyst support and a supported metal catalyst, a process for their preparation, and the use of the catalyst | |
| Pandya et al. | Cascade dehydrative amination of glycerol to oxazoline | |
| CN103379958A (zh) | 通过火焰喷射热解制备的用于自热丙烷脱氢的催化剂 | |
| US20070190347A1 (en) | Gold-and cerium-oxide-based composition, method for the preparation and the use thereof in the form of a catalyst, in particular for carbon monoxide oxidation | |
| CN101288851B (zh) | 一种经孔壁碳修饰的sba-15负载的高分散镍催化剂及其制备方法 | |
| US8680005B2 (en) | Catalyst from flame-spray pyrolysis and catalyst for autothermal propane dehydrogenation | |
| US20120190537A1 (en) | Catalyst support from flame-spray pyrolysis and catalyst for autothermal propane dehydrogenation | |
| CA2018991C (en) | Process for the preparation of vinyl acetate | |
| Wu et al. | Direct oxidation of methane to methanol using CuMoO 4 | |
| EP3651898B1 (en) | Catalysts based on pd/ceo2 and preparation method thereof | |
| US9598644B1 (en) | Method of CO and/or CO2 hydrogenation to higher hydrocarbons using doped mixed-metal oxides | |
| CN111569872B (zh) | 一种活性炭-钯镓锡液态合金复合催化剂及其制备方法和应用 | |
| WO2021250567A1 (en) | A process for reconstructing a spent dehydrogenation catalyst |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20131030 |