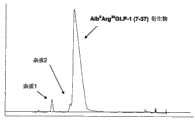
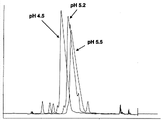
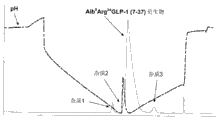
CN102770440A - 纯化肽的制备性rp-hplc方法 - Google Patents
纯化肽的制备性rp-hplc方法 Download PDFInfo
- Publication number
- CN102770440A CN102770440A CN2011800118239A CN201180011823A CN102770440A CN 102770440 A CN102770440 A CN 102770440A CN 2011800118239 A CN2011800118239 A CN 2011800118239A CN 201180011823 A CN201180011823 A CN 201180011823A CN 102770440 A CN102770440 A CN 102770440A
- Authority
- CN
- China
- Prior art keywords
- peptide
- value
- polypeptide
- glp
- gradient
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
- C07K1/14—Extraction; Separation; Purification
- C07K1/16—Extraction; Separation; Purification by chromatography
- C07K1/20—Partition-, reverse-phase or hydrophobic interaction chromatography
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Analytical Chemistry (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP10155023.4 | 2010-03-01 | ||
| EP10155023 | 2010-03-01 | ||
| US30964210P | 2010-03-02 | 2010-03-02 | |
| US61/309642 | 2010-03-02 | ||
| PCT/EP2011/052964 WO2011107447A1 (en) | 2010-03-01 | 2011-03-01 | Preparative rp-hplc method for purifying peptides |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102770440A true CN102770440A (zh) | 2012-11-07 |
Family
ID=42727392
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2011800118239A Pending CN102770440A (zh) | 2010-03-01 | 2011-03-01 | 纯化肽的制备性rp-hplc方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US9422330B2 (enExample) |
| EP (1) | EP2542565A1 (enExample) |
| JP (1) | JP5992836B2 (enExample) |
| CN (1) | CN102770440A (enExample) |
| WO (1) | WO2011107447A1 (enExample) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104994924A (zh) * | 2013-01-29 | 2015-10-21 | 诺伊兰健康科学私人有限公司 | 使用在反相柱上的替代固定相纯化有机化合物 |
| CN105223296A (zh) * | 2015-10-16 | 2016-01-06 | 江苏开元医药化工有限公司 | 一类多肽的纯化方法 |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2012313045A1 (en) * | 2011-09-22 | 2014-04-10 | Hochschule Ostwestfalen-Lippe | Method for producing peptide fractions and use thereof |
| US8921113B2 (en) | 2012-12-21 | 2014-12-30 | Dionex Corporation | Buffer kit and method of generating a linear pH gradient |
| EP2976331B1 (en) | 2013-03-21 | 2017-03-01 | Sanofi-Aventis Deutschland GmbH | Synthesis of hydantoin containing peptide products |
| MX365465B (es) | 2013-03-21 | 2019-06-04 | Sanofi Aventis Deutschland | Sintesis de productos peptidicos que contienen imida ciclica. |
| CA2924821A1 (en) * | 2013-09-20 | 2015-03-26 | Davuluri, Ramamohan Rao | Purification of organic compounds by surfactant mediated preparative hplc |
| EP3433268B1 (en) | 2016-03-23 | 2021-07-28 | Bachem Holding AG | Purification of glucagon-like peptide 1 analogs |
| US12269844B2 (en) | 2019-03-25 | 2025-04-08 | Scinopharm Taiwan, Ltd. | Process for purifying semaglutide and liraglutide |
| TWI738260B (zh) | 2019-03-25 | 2021-09-01 | 台灣神隆股份有限公司 | 純化利拉魯肽之方法 |
| US11022585B2 (en) | 2019-06-09 | 2021-06-01 | Dionex Corporation | Methods and systems for optimizing buffer conditions with liquid chromatography |
| KR102691149B1 (ko) | 2021-01-20 | 2024-08-05 | 바이킹 테라퓨틱스 인코포레이티드 | 대사 및 간 질환 치료를 위한 조성물 및 방법 |
| CN115703825B (zh) * | 2021-08-17 | 2026-01-02 | 派格生物医药(杭州)股份有限公司 | 艾塞那肽变体及其聚乙二醇缀合物的制备方法 |
| US12421282B2 (en) | 2021-09-15 | 2025-09-23 | Viking Therapeutics, Inc. | Compositions and methods for the treatment of metabolic and liver disorders |
| CN114656550A (zh) * | 2021-12-28 | 2022-06-24 | 深圳翰宇药业股份有限公司 | 一种利拉鲁肽/索马鲁肽的纯化方法及应用 |
| WO2024147150A1 (en) * | 2023-01-02 | 2024-07-11 | Zelle Biotechnology Pvt. Ltd. | Chromatographic detection, estimation and separation of amino acids in hydrolysates of monoclonal antibodies |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1232470A (zh) * | 1996-08-30 | 1999-10-20 | 诺沃挪第克公司 | Glp-1衍生物 |
| CN1839155A (zh) * | 2003-08-21 | 2006-09-27 | 诺沃挪第克公司 | 胰高血糖素样肽的纯化 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE4028120C2 (de) * | 1990-09-05 | 1996-09-19 | Hoechst Ag | Verfahren zur Reinigung von Insulin und/oder Insulinderivaten |
| US5424286A (en) | 1993-05-24 | 1995-06-13 | Eng; John | Exendin-3 and exendin-4 polypeptides, and pharmaceutical compositions comprising same |
| US6767887B1 (en) | 1996-06-05 | 2004-07-27 | Roche Diagnostics Gmbh | Exendin analogues, processes for their preparation and medicaments containing them |
| EP1076066A1 (en) | 1999-07-12 | 2001-02-14 | Zealand Pharmaceuticals A/S | Peptides for lowering blood glucose levels |
| JP2007526235A (ja) | 2003-08-21 | 2007-09-13 | ノボ ノルディスク アクティーゼルスカブ | グルカゴン様ペプチドの精製 |
| WO2007071768A1 (en) | 2005-12-23 | 2007-06-28 | Novo Nordisk A/S | Purification of proteins using preparative reverse phase chromatography (rpc) |
| WO2007071767A1 (en) | 2005-12-23 | 2007-06-28 | Novo Nordisk Health Care Ag | Purification of vitamin k-dependent polypeptides using preparative reverse phase chromatography (rpc) |
-
2011
- 2011-03-01 EP EP11705589A patent/EP2542565A1/en not_active Withdrawn
- 2011-03-01 US US13/580,204 patent/US9422330B2/en not_active Expired - Fee Related
- 2011-03-01 WO PCT/EP2011/052964 patent/WO2011107447A1/en not_active Ceased
- 2011-03-01 JP JP2012555389A patent/JP5992836B2/ja not_active Expired - Fee Related
- 2011-03-01 CN CN2011800118239A patent/CN102770440A/zh active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1232470A (zh) * | 1996-08-30 | 1999-10-20 | 诺沃挪第克公司 | Glp-1衍生物 |
| CN1839155A (zh) * | 2003-08-21 | 2006-09-27 | 诺沃挪第克公司 | 胰高血糖素样肽的纯化 |
Non-Patent Citations (3)
| Title |
|---|
| KALISZAN R ET AL: "pH gradient reversed-phase HPLC", 《ANALYTICAL CHEMISTRY》 * |
| TOMASZ BACZEK ET AL: "pH gradient reversed-phase liquid chromatography as a fractionation tool for the separation of peptides", 《TALANTA》 * |
| WICZLING P ET AL: "Determination of PKa by pH gradient reversed-phase HPLC", 《ANALYTICAL CHEMISTRY》 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104994924A (zh) * | 2013-01-29 | 2015-10-21 | 诺伊兰健康科学私人有限公司 | 使用在反相柱上的替代固定相纯化有机化合物 |
| CN105223296A (zh) * | 2015-10-16 | 2016-01-06 | 江苏开元医药化工有限公司 | 一类多肽的纯化方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5992836B2 (ja) | 2016-09-14 |
| WO2011107447A1 (en) | 2011-09-09 |
| EP2542565A1 (en) | 2013-01-09 |
| US9422330B2 (en) | 2016-08-23 |
| JP2013521485A (ja) | 2013-06-10 |
| US20120322976A1 (en) | 2012-12-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102770440A (zh) | 纯化肽的制备性rp-hplc方法 | |
| JP7011643B2 (ja) | グルカゴン様ペプチド1類似体の精製 | |
| JP5755398B2 (ja) | 伸長されたglp−1化合物 | |
| JP4585037B2 (ja) | アシル化glp−1化合物 | |
| JP6179864B2 (ja) | グルカゴン/glp−1レセプタコ−アゴニスト | |
| JP5756407B2 (ja) | ポリペプチドの向流精製 | |
| KR102002783B1 (ko) | 포도당 의존성 인슐리노트로핀 폴리펩타이드 유사물질, 이의 약학적 조성물 및 응용 | |
| CN101868476B (zh) | 胰高血糖素样肽-1衍生物及其制药用途 | |
| EP2464655B1 (en) | Chromatographic processes | |
| US20110313131A1 (en) | Reversed phase hplc purification of a glp-1 analogue | |
| JP6110312B2 (ja) | インスリンの精製 | |
| MXPA06006746A (es) | Analogos de glp-1 novedosos ligados a agentes similares a albumina. | |
| EP4095149A1 (en) | Triple agonist for glucagon-like peptide-1 receptor, glucagon receptor, and gastric inhibitory polypeptide receptor | |
| EP2905289A1 (en) | Method for purifying teriparatide (PTH1-34) | |
| CA3018627C (en) | Purification of glucagon-like peptide 1 analogs | |
| CA2908295A1 (en) | Method for purifying teriparatide |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20121107 |
|
| RJ01 | Rejection of invention patent application after publication |