WO2022255348A1 - 可溶型clec2を用いた急性脳血管障害のリスク評価方法 - Google Patents
可溶型clec2を用いた急性脳血管障害のリスク評価方法 Download PDFInfo
- Publication number
- WO2022255348A1 WO2022255348A1 PCT/JP2022/022076 JP2022022076W WO2022255348A1 WO 2022255348 A1 WO2022255348 A1 WO 2022255348A1 JP 2022022076 W JP2022022076 W JP 2022022076W WO 2022255348 A1 WO2022255348 A1 WO 2022255348A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cerebrovascular accident
- acute cerebrovascular
- concentration
- patient
- soluble
- Prior art date
Links
- 238000000034 method Methods 0.000 title claims abstract description 75
- 230000001154 acute effect Effects 0.000 title abstract description 8
- 230000002490 cerebral effect Effects 0.000 title abstract description 3
- 208000019553 vascular disease Diseases 0.000 title abstract description 3
- 201000010875 transient cerebral ischemia Diseases 0.000 claims abstract description 88
- 208000032109 Transient ischaemic attack Diseases 0.000 claims abstract description 86
- 102100032529 C-type lectin domain family 1 member B Human genes 0.000 claims abstract description 57
- 210000004369 blood Anatomy 0.000 claims abstract description 38
- 239000008280 blood Substances 0.000 claims abstract description 38
- 208000024891 symptom Diseases 0.000 claims abstract description 32
- 101000942284 Homo sapiens C-type lectin domain family 1 member B Proteins 0.000 claims abstract 13
- 208000006011 Stroke Diseases 0.000 claims description 97
- 230000001269 cardiogenic effect Effects 0.000 claims description 95
- 101710160442 C-type lectin domain family 1 member B Proteins 0.000 claims description 44
- 239000003154 D dimer Substances 0.000 claims description 32
- 108010052295 fibrin fragment D Proteins 0.000 claims description 32
- 238000012502 risk assessment Methods 0.000 claims description 22
- 230000015271 coagulation Effects 0.000 claims description 13
- 238000005345 coagulation Methods 0.000 claims description 13
- 238000003018 immunoassay Methods 0.000 claims description 13
- 239000003550 marker Substances 0.000 claims description 13
- 239000003527 fibrinolytic agent Substances 0.000 claims description 8
- 230000003480 fibrinolytic effect Effects 0.000 claims description 8
- 238000011984 electrochemiluminescence immunoassay Methods 0.000 claims description 4
- 230000020764 fibrinolysis Effects 0.000 claims description 4
- 230000008859 change Effects 0.000 claims description 2
- 239000000463 material Substances 0.000 claims description 2
- 230000008569 process Effects 0.000 claims description 2
- 206010008118 cerebral infarction Diseases 0.000 abstract description 104
- 238000003745 diagnosis Methods 0.000 abstract description 27
- 238000003748 differential diagnosis Methods 0.000 abstract description 14
- 238000009534 blood test Methods 0.000 abstract description 10
- 238000011156 evaluation Methods 0.000 abstract description 5
- 230000000747 cardiac effect Effects 0.000 abstract description 3
- 201000008247 brain infarction Diseases 0.000 abstract 1
- 208000026106 cerebrovascular disease Diseases 0.000 description 107
- 210000001772 blood platelet Anatomy 0.000 description 51
- 206010061216 Infarction Diseases 0.000 description 26
- 230000007574 infarction Effects 0.000 description 26
- 208000007536 Thrombosis Diseases 0.000 description 25
- 238000005259 measurement Methods 0.000 description 22
- 238000011282 treatment Methods 0.000 description 17
- 238000003384 imaging method Methods 0.000 description 15
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 14
- 230000010118 platelet activation Effects 0.000 description 14
- 102000004169 proteins and genes Human genes 0.000 description 14
- 108090000623 proteins and genes Proteins 0.000 description 14
- 238000012360 testing method Methods 0.000 description 13
- 210000004556 brain Anatomy 0.000 description 12
- 201000010099 disease Diseases 0.000 description 12
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 11
- 239000003146 anticoagulant agent Substances 0.000 description 11
- 108010073385 Fibrin Proteins 0.000 description 10
- 102000009123 Fibrin Human genes 0.000 description 10
- 229950003499 fibrin Drugs 0.000 description 10
- 239000000243 solution Substances 0.000 description 10
- 229940127219 anticoagulant drug Drugs 0.000 description 9
- 210000004204 blood vessel Anatomy 0.000 description 9
- 239000003153 chemical reaction reagent Substances 0.000 description 9
- 239000000090 biomarker Substances 0.000 description 7
- 230000035945 sensitivity Effects 0.000 description 7
- 229940127218 antiplatelet drug Drugs 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- 239000003814 drug Substances 0.000 description 6
- 230000003902 lesion Effects 0.000 description 5
- 239000000106 platelet aggregation inhibitor Substances 0.000 description 5
- 206010003658 Atrial Fibrillation Diseases 0.000 description 4
- 241000282412 Homo Species 0.000 description 4
- 208000032382 Ischaemic stroke Diseases 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 206010047571 Visual impairment Diseases 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 239000003085 diluting agent Substances 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 239000004816 latex Substances 0.000 description 4
- 229920000126 latex Polymers 0.000 description 4
- 231100000862 numbness Toxicity 0.000 description 4
- 238000011269 treatment regimen Methods 0.000 description 4
- 208000029257 vision disease Diseases 0.000 description 4
- 230000004393 visual impairment Effects 0.000 description 4
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 3
- 108090000790 Enzymes Proteins 0.000 description 3
- 102000004190 Enzymes Human genes 0.000 description 3
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 3
- 206010033799 Paralysis Diseases 0.000 description 3
- 206010040026 Sensory disturbance Diseases 0.000 description 3
- 229960001138 acetylsalicylic acid Drugs 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- KXNPVXPOPUZYGB-XYVMCAHJSA-N argatroban Chemical compound OC(=O)[C@H]1C[C@H](C)CCN1C(=O)[C@H](CCCN=C(N)N)NS(=O)(=O)C1=CC=CC2=C1NC[C@H](C)C2 KXNPVXPOPUZYGB-XYVMCAHJSA-N 0.000 description 3
- 229960003856 argatroban Drugs 0.000 description 3
- 210000001367 artery Anatomy 0.000 description 3
- 230000003143 atherosclerotic effect Effects 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 230000034994 death Effects 0.000 description 3
- 229940088598 enzyme Drugs 0.000 description 3
- 229960002897 heparin Drugs 0.000 description 3
- 229920000669 heparin Polymers 0.000 description 3
- 230000007246 mechanism Effects 0.000 description 3
- 230000009251 neurologic dysfunction Effects 0.000 description 3
- 208000015015 neurological dysfunction Diseases 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 230000003449 preventive effect Effects 0.000 description 3
- 208000027765 speech disease Diseases 0.000 description 3
- PJVWKTKQMONHTI-UHFFFAOYSA-N warfarin Chemical compound OC=1C2=CC=CC=C2OC(=O)C=1C(CC(=O)C)C1=CC=CC=C1 PJVWKTKQMONHTI-UHFFFAOYSA-N 0.000 description 3
- 229960005080 warfarin Drugs 0.000 description 3
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 2
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 2
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 2
- 206010003130 Arrhythmia supraventricular Diseases 0.000 description 2
- 206010008111 Cerebral haemorrhage Diseases 0.000 description 2
- 206010013887 Dysarthria Diseases 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- 239000007995 HEPES buffer Substances 0.000 description 2
- 208000004552 Lacunar Stroke Diseases 0.000 description 2
- 206010051078 Lacunar infarction Diseases 0.000 description 2
- 239000007987 MES buffer Substances 0.000 description 2
- 208000019430 Motor disease Diseases 0.000 description 2
- 208000031481 Pathologic Constriction Diseases 0.000 description 2
- 102000004211 Platelet factor 4 Human genes 0.000 description 2
- 108090000778 Platelet factor 4 Proteins 0.000 description 2
- 208000032851 Subarachnoid Hemorrhage Diseases 0.000 description 2
- 108090000190 Thrombin Proteins 0.000 description 2
- 208000034841 Thrombotic Microangiopathies Diseases 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 230000006793 arrhythmia Effects 0.000 description 2
- 210000000601 blood cell Anatomy 0.000 description 2
- 230000036772 blood pressure Effects 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 210000001715 carotid artery Anatomy 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 230000006735 deficit Effects 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- 230000008034 disappearance Effects 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 238000013399 early diagnosis Methods 0.000 description 2
- 230000002439 hemostatic effect Effects 0.000 description 2
- 208000026762 inability to speak Diseases 0.000 description 2
- 208000028867 ischemia Diseases 0.000 description 2
- 210000003593 megakaryocyte Anatomy 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 229940126619 mouse monoclonal antibody Drugs 0.000 description 2
- HEGSGKPQLMEBJL-RKQHYHRCSA-N octyl beta-D-glucopyranoside Chemical compound CCCCCCCCO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HEGSGKPQLMEBJL-RKQHYHRCSA-N 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 230000002335 preservative effect Effects 0.000 description 2
- 210000001525 retina Anatomy 0.000 description 2
- 230000009863 secondary prevention Effects 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 208000026473 slurred speech Diseases 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 2
- BYKRNSHANADUFY-UHFFFAOYSA-M sodium octanoate Chemical compound [Na+].CCCCCCCC([O-])=O BYKRNSHANADUFY-UHFFFAOYSA-M 0.000 description 2
- 230000036262 stenosis Effects 0.000 description 2
- 208000037804 stenosis Diseases 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- 229960004072 thrombin Drugs 0.000 description 2
- 230000001052 transient effect Effects 0.000 description 2
- 230000000007 visual effect Effects 0.000 description 2
- 102100020970 ATP-binding cassette sub-family D member 2 Human genes 0.000 description 1
- 206010059245 Angiopathy Diseases 0.000 description 1
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 239000005552 B01AC04 - Clopidogrel Substances 0.000 description 1
- 101800003265 Beta-thromboglobulin Proteins 0.000 description 1
- 102000003930 C-Type Lectins Human genes 0.000 description 1
- 108090000342 C-Type Lectins Proteins 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 206010008088 Cerebral artery embolism Diseases 0.000 description 1
- 206010010904 Convulsion Diseases 0.000 description 1
- 206010051055 Deep vein thrombosis Diseases 0.000 description 1
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical compound [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- 208000005189 Embolism Diseases 0.000 description 1
- 206010019280 Heart failures Diseases 0.000 description 1
- 208000016988 Hemorrhagic Stroke Diseases 0.000 description 1
- 101000783774 Homo sapiens ATP-binding cassette sub-family D member 2 Proteins 0.000 description 1
- 208000013016 Hypoglycemia Diseases 0.000 description 1
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 description 1
- 238000000585 Mann–Whitney U test Methods 0.000 description 1
- 208000027530 Meniere disease Diseases 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 208000019695 Migraine disease Diseases 0.000 description 1
- 208000016285 Movement disease Diseases 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 206010028851 Necrosis Diseases 0.000 description 1
- 206010060860 Neurological symptom Diseases 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 102100036154 Platelet basic protein Human genes 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 108010094028 Prothrombin Proteins 0.000 description 1
- 102100027378 Prothrombin Human genes 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 208000005386 Transient Global Amnesia Diseases 0.000 description 1
- 229920004890 Triton X-100 Polymers 0.000 description 1
- 239000013504 Triton X-100 Substances 0.000 description 1
- 206010047249 Venous thrombosis Diseases 0.000 description 1
- 206010047513 Vision blurred Diseases 0.000 description 1
- 208000013449 Visual field disease Diseases 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 230000004520 agglutination Effects 0.000 description 1
- 230000002785 anti-thrombosis Effects 0.000 description 1
- 239000004019 antithrombin Substances 0.000 description 1
- 229940127217 antithrombotic drug Drugs 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000000923 atherogenic effect Effects 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 210000001627 cerebral artery Anatomy 0.000 description 1
- GKTWGGQPFAXNFI-HNNXBMFYSA-N clopidogrel Chemical compound C1([C@H](N2CC=3C=CSC=3CC2)C(=O)OC)=CC=CC=C1Cl GKTWGGQPFAXNFI-HNNXBMFYSA-N 0.000 description 1
- 229960003009 clopidogrel Drugs 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 239000000032 diagnostic agent Substances 0.000 description 1
- 229940039227 diagnostic agent Drugs 0.000 description 1
- 238000012631 diagnostic technique Methods 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- UKWLRLAKGMZXJC-QIECWBMSSA-L disodium;[4-chloro-3-[(3r,5s)-1-chloro-3'-methoxyspiro[adamantane-4,4'-dioxetane]-3'-yl]phenyl] phosphate Chemical compound [Na+].[Na+].O1OC2([C@@H]3CC4C[C@H]2CC(Cl)(C4)C3)C1(OC)C1=CC(OP([O-])([O-])=O)=CC=C1Cl UKWLRLAKGMZXJC-QIECWBMSSA-L 0.000 description 1
- 208000009190 disseminated intravascular coagulation Diseases 0.000 description 1
- 238000002592 echocardiography Methods 0.000 description 1
- QELUYTUMUWHWMC-UHFFFAOYSA-N edaravone Chemical compound O=C1CC(C)=NN1C1=CC=CC=C1 QELUYTUMUWHWMC-UHFFFAOYSA-N 0.000 description 1
- 229950009041 edaravone Drugs 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 230000005713 exacerbation Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 239000000208 fibrin degradation product Substances 0.000 description 1
- 108010073651 fibrinmonomer Proteins 0.000 description 1
- 239000000282 fibrinogen degradation product Substances 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 208000035474 group of disease Diseases 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 208000000122 hyperventilation Diseases 0.000 description 1
- 230000002218 hypoglycaemic effect Effects 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 238000003317 immunochromatography Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 208000020658 intracerebral hemorrhage Diseases 0.000 description 1
- 201000010849 intracranial embolism Diseases 0.000 description 1
- 230000000302 ischemic effect Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 238000000691 measurement method Methods 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 230000006371 metabolic abnormality Effects 0.000 description 1
- 239000011859 microparticle Substances 0.000 description 1
- 206010027599 migraine Diseases 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 208000031225 myocardial ischemia Diseases 0.000 description 1
- 201000003631 narcolepsy Diseases 0.000 description 1
- 230000017074 necrotic cell death Effects 0.000 description 1
- 210000002569 neuron Anatomy 0.000 description 1
- 229940127066 new oral anticoagluant drug Drugs 0.000 description 1
- 230000000474 nursing effect Effects 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 238000011017 operating method Methods 0.000 description 1
- 229940126701 oral medication Drugs 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 208000029308 periodic paralysis Diseases 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000037452 priming Effects 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 229940039716 prothrombin Drugs 0.000 description 1
- 238000003127 radioimmunoassay Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000002000 scavenging effect Effects 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 239000003998 snake venom Substances 0.000 description 1
- 210000000278 spinal cord Anatomy 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 206010042772 syncope Diseases 0.000 description 1
- 229940126585 therapeutic drug Drugs 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 238000013175 transesophageal echocardiography Methods 0.000 description 1
- 238000010967 transthoracic echocardiography Methods 0.000 description 1
- 238000002604 ultrasonography Methods 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/483—Physical analysis of biological material
- G01N33/487—Physical analysis of biological material of liquid biological material
- G01N33/49—Blood
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
Definitions
- the present invention relates to a method for evaluating the risk of acute cerebrovascular accident using soluble CLEC2.
- Stroke is a group of diseases that includes ischemic stroke, which occurs when a blood vessel in the brain is clogged, that is, cerebral infarction, and hemorrhagic stroke, which occurs when a blood vessel in the brain is cut, that is, cerebral hemorrhage and subarachnoid hemorrhage. It is the second most common cause of death, after ischemic heart disease. Stroke is not only a major cause of death, but it often leaves severe aftereffects even after survival, and is a major factor in the need for nursing care. Therefore, it is necessary to have a system that enables prevention, early diagnosis, prompt response after diagnosis, and care and risk management by a wide range of medical professionals.
- cerebral infarction occurs when a blood vessel in the brain narrows or when a blood clot formed in another place blocks a blood vessel in the brain, preventing oxygen and nutrients from reaching the brain. Necrosis of nerve cells in the brain causes various disorders.
- the thrombus that causes cerebral infarction is classified into cardiogenic cerebral embolism, in which a thrombus formed in the heart travels through the blood vessels and clogs an artery in the brain, and noncardiogenic cerebral infarction, which is caused by a thrombus formed in a place other than the heart. broadly classified.
- fibrin is considered to be the main thrombus, and anticoagulants (warfarin and DOAC) that suppress the formation of fibrin are used for treatment and secondary prevention.
- the main thrombus is considered to be platelets, and antiplatelet drugs (aspirin, clopidogrel, etc.) that suppress platelet activation are used for treatment and secondary prevention. That is, since the treatments for cardiogenic cerebral ischemic stroke and non-cardiogenic stroke are clearly different, it is important to correctly diagnose whether it is cardiogenic or non-cardiogenic.
- Cardiogenic cerebral infarction is a relatively large infarct caused by a thrombus formed due to stasis of blood flow in the atria due to atrial fibrillation, which travels through the bloodstream and reaches the brain to cause an infarction.
- the diagnosis is made based on the observation of a large infarction in MRI images and information on past conditions such as atrial fibrillation, arrhythmia, and heart disease.
- non-cardiogenic cerebral infarction is roughly classified into atheromatous and lacunar.
- Atherosclerotic cerebral infarction clogs relatively large arteries and lacunar clogs small blood vessels, so diagnosis is made based on information such as the size of the infarct from MRI images, the state of arteriosclerosis, and metabolic abnormalities from blood tests.
- Atrial fibrillation is detected by an electrocardiogram, but it can be persistent or sudden, and sudden cases are difficult to detect easily. In addition, there are many cases in which images of atherogenic cerebral infarction and cardiogenic cerebral infarction cannot be clearly distinguished.
- TIA transient ischemic attack
- TIA refers to a condition in which blood vessels are constricted or clogged with thrombus, similar to cerebral infarction, but the event is transient, and symptoms similar to cerebral infarction occur transiently.
- TIA was considered to be the disappearance of ischemic symptoms in the brain within 24 hours.
- Today due to advances in imaging technology, there are more and more cases of cerebral infarction being detected by imaging even after symptoms have disappeared. defined as "temporary neurological dysfunction due to focal ischemia of the brain, spinal cord, or retina without acute infarction," and distinguishes between infarction and TIA, regardless of symptom duration.
- a patient with disappearance of symptoms but a cerebral infarction seen on imaging is defined as a cerebral infarction
- a TIA is defined as a patient whose symptoms have disappeared and no cerebral infarction is observed on imaging.
- the WHO World Health Organization
- International Classification of Diseases ICD-11, 2018
- TIA must not have an infarct focus, and the concept of judging by imaging has become a global consensus.
- TIA cerebrovascular diseases
- cerebral infarction cerebral hemorrhage
- subarachnoid hemorrhage subarachnoid hemorrhage
- TIA is clearly distinguished as a separate disease from cerebral infarction. Diagnosis of a TIA is extremely important, even when symptoms have disappeared. That's because TIA patients often have an early onset of stroke. As a result of multiple clinical studies, it has been reported that 10 to 15% of TIA cases develop cerebral infarction within 3 months, and half of them develop within 48 hours, which is conventionally believed. It turns out that stroke often develops much earlier than expected. Based on such data, the Japan Stroke Society recommends that "immediate start of preventive treatment when TIA is suspected" in the "Stroke Treatment Guidelines" issued in 2009. In other words, TIA is said to be a precursor to cerebral infarction, which is a serious disease.
- the presence or absence of a lesion is required for the diagnosis of TIA. (17% within 6 hours, 37% after 24 hours).Also, it is said that many of the cases in which there was no lesion in the first DWI show lesions in the second DWI. That is, according to the definition of cerebral infarction when a lesion is observed on the image and TIA when no lesion is detected, the timing of DWI has a great influence on the diagnosis.
- CLEC2 C-type lectin-like receptor 2
- sCLEC2 soluble CLEC-2
- thrombus formation when a blood vessel is ruptured, platelets are first activated and aggregated to form a primary thrombus. Subsequent thrombi are assumed to form.
- the platelet system and the coagulation system are mutually activated, and a hemostatic thrombus formed of platelets, fibrin, and red blood cells involved in them is observed.
- Cerebral infarction is a serious disease with a high mortality rate, and even if death is avoided, sequelae often remain.
- about 15% of TIA cases are known to cause cerebral infarction, so early diagnosis and preventive treatment are extremely important. This is because if TIA can be diagnosed, appropriate antithrombotic therapy can significantly reduce the probability of subsequent cerebral infarction.
- Patients diagnosed with TIA should be monitored by a doctor for, for example, one week after the onset of cerebral infarction. It is also possible to significantly improve the aftereffects. This is because the earlier the treatment of cerebral infarction, the better, and it is a race against time.
- a diagnosis of TIA is made by interviewing clinical symptoms and assessing the patient's risk. Namely, paralysis of limbs on one side (movement disorder), numbness on one side (sensory disturbance), speech impediment, slurred speech (speech disorder), blurred vision on one side (visual impairment), and numbness on one side
- the main symptom is difficulty in seeing certain things (visual field disorder), but these symptoms disappear within a few minutes to an hour at most.
- Patient risk assessment is also widely performed to compensate for the difficulty of such interviews.
- the ABCD2 score is typical, but it is evaluated by scoring Age (age), Blood Pressure (blood pressure), Clinical Feature (clinical symptoms), Diabetes (diabetes), Duration of Symptoms (duration of symptoms) It is something to do. This is only an evaluation of the risk of developing cerebral infarction afterward, and does not diagnose TIA itself. Thus, there is no objective imaging test or blood test that can augment clinical inquiry in diagnosing TIA.
- an object of the present invention is to provide a simple, rapid and objective risk assessment method by a blood test that can reinforce the clinical symptom inquiry for the diagnosis of TIA, and to provide a risk assessment method for acute cerebrovascular disorders such as cerebral infarction and TIA.
- An object of the present invention is to provide a simple, rapid, and objective blood test-based risk assessment method that can supplement images, electrocardiograms, and conventional blood tests for the differential diagnosis of cardiogenic and non-cardiogenic diseases.
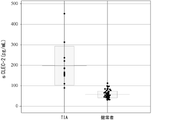
- the present inventors have made intensive studies to solve the above problems. As a result, it was found that the sCLEC2 concentration in the blood of TIA patients was significantly elevated compared to healthy subjects. The present inventors have found that sCLEC2 can be at a high level even in a state where there is no infarction and no thrombus can be confirmed, such as in TIA, but not in a state where there is an infarction and thrombus is present, such as cerebral infarction. This is consistent with the fact that TIA patients have a high probability of having a stroke in the near future.
- an appropriate threshold that is, a cutoff value is required to determine the risk using sCLEC2 concentration.
- the TIA cutoff value for the blood sCLEC2 concentration is preferably 66 pg/mL or more and 148 pg/mL or less.
- infarction images cannot be used to determine cardiogenic or non-cardiogenic cardiogenicity.
- sCLEC2 in the blood of cerebral infarction patients was measured, it was found that sCLEC2 measured values were significantly higher in non-cardiogenic patients than in cardiogenic patients. Based on this knowledge, D-dimer, a biomarker indicating the presence of fibrin thrombi, was simultaneously measured, and the ratio of sCLEC2 to D-dimer (sCLEC2/D-dimer ratio) was calculated. It was found that the sCLEC2/D-dimer ratios in patients with cardiogenic disease were significantly higher than those in cardiogenic patients. That is, sCLEC2 represents platelet activity, and D-dimer represents the presence of fibrin thrombus. By using the ratio of sCLEC2 to D-dimer for determining cardiogenicity, it is possible to provide a method for assisting more accurate diagnosis in addition to conventional methods.
- the present invention provides: [1] Risk of acute cerebrovascular accident, including the step of measuring the concentration of soluble CLEC2 present in blood collected from patients suspected of having acute cerebrovascular accident or diagnosed with acute cerebrovascular accident How to do the evaluation.
- [2] A method for risk assessment of a patient suspected of having acute cerebrovascular accident or a patient diagnosed with acute cerebrovascular accident comprising: (1) providing a blood sample from said patient; (2) determining the concentration of soluble CLEC2 in the sample; (3) correlating the soluble CLEC2 concentration with the presence or absence of acute cerebrovascular accident in the patient, the likelihood of outcome, or whether it is cardiogenic or non-cardiogenic;
- the method of [1] comprising [3] A method for risk assessment of a patient suspected of having acute cerebrovascular accident or a patient diagnosed with acute cerebrovascular accident, wherein the soluble CLEC2 concentration and the presence of acute cerebrovascular accident in the patient or correlating absence, likelihood of outcome, or whether cardiogenic or non-cardiogenic, assessing whether said patient is at risk based on changes
- a method for risk assessment of a patient suspected of having acute cerebrovascular accident or a patient diagnosed with acute cerebrovascular accident comprising: (1) providing a blood sample from said patient; (2) determining the concentration of soluble CLEC2 in the sample; (3) measuring the platelet count in the sample; (4) dividing the soluble CLEC-2 concentration by the platelet count; (5) correlating the value obtained by dividing the soluble CLEC2 concentration by the platelet count with the presence or absence of acute cerebrovascular accident in the patient, the possibility of outcome, or whether it is cardiogenic or non-cardiogenic; process, The method of [1], comprising [6] A method for assessing the risk of acute cerebrovascular accident in a patient suspected of having acute cerebrovascular accident or diagnosed with acute cerebrovascular accident, wherein the acute cerebrovascular accident is transient cerebral ischemia Stroke, and in the step of correlating the value obtained by dividing the soluble CLEC2 concentration by the platelet count with the transient ischemic attack, the cutoff value of the value obtained by dividing the soluble
- a method for assessing the risk of acute cerebrovascular accident in a patient suspected of having acute cerebrovascular accident or diagnosed with acute cerebrovascular accident, wherein said acute cerebrovascular accident is cardiogenic or non-cardiac Any of the methods of [1] to [4], which provides materials for determining whether the substance is priming.
- a method for risk assessment of acute cerebrovascular accident in a patient suspected of having acute cerebrovascular accident or diagnosed with acute cerebrovascular accident comprising: (1) providing a blood sample from said patient; (2) determining the concentration of soluble CLEC2 in the sample; (3) measuring the platelet count in the sample; (4) dividing the soluble CLEC-2 concentration by the coagulation fibrinolysis marker; (5) The value obtained by dividing the soluble CLEC2 concentration by the coagulation fibrinolytic marker, and the presence or absence of acute cerebrovascular accident in the patient, the possibility of outcome, or whether it is cardiogenic or non-cardiogenic correlating; The method of [1], comprising [9] A method for assessing the risk of acute cerebrovascular accident in a patient suspected of having acute cerebrovascular accident or diagnosed with acute cerebrovascular accident, wherein the coagulation and fibrinolysis marker is D-dimer.
- a method for assessing the risk of acute cerebrovascular accident in a patient suspected of having acute cerebrovascular accident or diagnosed with acute cerebrovascular accident, wherein providing a sample derived from the patient is The method of any one of [1] to [9], which is collected from the patient within 48 hours after the onset of symptoms of vascular disorder.
- the step of determining the soluble CLEC2 concentration is a highly sensitive immunoassay, such as a chemiluminescence immunoassay, an electrochemiluminescence immunoassay, or a fluorescence immunoassay; either way.
- Measure (or determine) the soluble CLEC2 concentration in the sample (or the soluble CLEC-2 concentration divided by the platelet count, or the soluble CLEC-2 concentration divided by the coagulation fibrinolytic marker) a method for risk assessment of acute cerebrovascular accident
- Measure (or determine) the soluble CLEC2 concentration in the sample (or the soluble CLEC-2 concentration divided by the platelet count, or the soluble CLEC-2 concentration divided by the coagulation fibrinolytic marker) a method to aid in risk assessment of acute cerebrovascular accident
- the soluble CLEC2 concentration in the sample or the value obtained by dividing the soluble CLEC-2 concentration by the platelet count, or the soluble CLEC-2 concentration with a coagulation fibrinolytic marker method of measuring (or determining)
- FIG. 1 is a graph comparing plasma concentrations of sCLEC2 between TIA patients and healthy subjects.
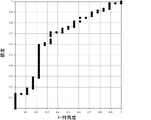
- FIG. 10 is an ROC curve (Receiver Operating Characteristic Curve) when TIA is diagnosed by measuring sCLEC2 concentration in blood.
- FIG. 3 is a graph comparing sCLEC2 concentrations in 3 groups of healthy subjects, cardiogenic cerebral infarction patients, and non-cardiogenic cerebral infarction patients by measuring sCLEC2 concentrations in healthy subjects and cerebral infarction patients. It is a ROC curve when differential diagnosis of cardiogenic cerebral infarction and non-cardiogenic cerebral infarction is made using sCLEC concentration of cerebral infarction patients.
- ROC curve when differential diagnosis of cardiogenic cerebral infarction and non-cardiogenic cerebral infarction is made using the D-dimer concentration of cerebral infarction patients.
- Fig. 10 is a ROC curve when the sCLEC2/D-dimer ratio of cerebral infarction patients is used to make a differential diagnosis between cardiogenic cerebral infarction and non-cardiogenic cerebral infarction.
- CLEC2 is a platelet-activating receptor belonging to the C-type lectin family, normally present in the platelet membrane, but is released into the blood following platelet activation.
- soluble CLEC2 sCLEC2
- sCLEC2 refers to CLEC2 or CLEC2-derived molecules released from such platelets and detected in blood (in buffer when incubated in buffer).
- sCLEC2 is said to include a protein with a molecular weight of about 40 kDa, a protein with a molecular weight of about 32 kDa, a protein with a molecular weight of about 25 kDa, etc. in SDS-polyacrylamide gel electrophoresis (SDS-PAGE) under reducing conditions (Non-Patent Document 1).
- SDS-polyacrylamide gel electrophoresis SDS-polyacrylamide gel electrophoresis
- Non-Patent Document 1 A protein with a molecular weight of about 40 kDa and a protein with a molecular weight of about 32 kDa are present on the platelet membrane surface and are presumed to be released in a state contained in microparticles produced with platelet activation. It is considered that sugar chains are added to these.
- a protein with a molecular weight of about 25 kDa is considered to be cleaved by protease and released from platelets along with platelet activation.
- the amount of sCLEC2 as described above is measured.
- sCLEC2 may detect a protein with a molecular weight of about 40 kDa, a protein with a molecular weight of about 32 kDa, and a protein with a molecular weight of about 25 kDa collectively, or may detect only a protein with a molecular weight of about 25 kDa.
- the sCLEC2 concentration used in the present invention may be used alone or in combination with other biomarkers.
- a value obtained by dividing the sCLEC2 concentration by the platelet count (hereinafter sometimes referred to as C2PAC index) may be used.
- C2PAC index a value obtained by dividing the sCLEC2 concentration by the platelet count
- the sCLEC2 concentration is interpreted to include both the case of using the sCLEC2 concentration and the case of dividing the sCLEC2 concentration by the platelet count.
- the samples used for measurement are preferably derived from humans, but samples derived from animals other than humans may be used to understand the pathology of experimental animals.
- Experimental animals include, but are not limited to, guinea pigs, rats, mice, dogs, and the like.
- the method for detecting the presence of sCLEC2 is not particularly limited, but an immunological method using an antibody that recognizes sCLEC2 (hereinafter sometimes referred to as "anti-sCLEC2 antibody”) is preferred.
- immunologically detecting proteins include enzyme immunoassay (ELISA method), chemiluminescence immunoassay, electrochemiluminescence immunoassay, fluorescence immunoassay, radioimmunoassay, and immunochromatography. Any method can be used as long as it is an immunoassay method using a labeled antibody such as , Western blotting method, latex agglutination method, immunoturbidimetric method, and the like.
- a sample is collected from the target subject (especially a patient), for example, using a blood collection tube for plasma blood collection.
- a collection tube containing citric acid, which leaves little residual platelets, is usually suitable, but a tube containing heparin or EDTA is also possible.
- a blood collection tube containing EDTA is used for blood platelet count measurement, but separate blood collection tubes may be used for simultaneous blood collection.
- Plasma sCLEC2 concentration is measured, for example, by using plasma that has been centrifuged at 2000 g for about 20 minutes, but the conditions for centrifugation are not limited to this, and a measurement system using whole blood may also be used.
- the measurement of plasma sCLEC2 concentration will be described as an example, but the present invention is not limited to this. It is preferable that blood is collected from the patient before 48 hours have passed since the symptoms appeared, but even if 48 hours have passed, it can be used in the present invention. More preferably within 24 hours, still more preferably within 18 hours, and most preferably within 12 hours.
- acute cerebrovascular accident includes, but is not limited to, ischemic stroke such as cerebral infarction and TIA.
- TIA means a case in which cerebral infarction-like symptoms are transiently exhibited, but infarction cannot be confirmed by imaging examination.
- Cerebral infarction-like symptoms include, for example, paralysis of the limbs on one side (motor disorder), numbness on one side (sensory disturbance), inability to speak or speak slurred words (speech disorder), and difficulty seeing in one eye. (visual impairment), difficulty seeing things on one side (visual field impairment), and the like, but are not limited to these.
- a patient with cerebral infarction is a patient with an infarct image confirmed in the brain by MRI, CT, etc., in addition to the above clinical symptoms.
- Cerebral infarction is a state in which a cerebral artery is occluded (infarction), and it is clear that there is a thrombus in this case. It is considered to be In addition, disseminated intravascular coagulation (DIC), thrombotic microangiopathy (TMA), and deep vein thrombosis are pathological conditions in which clots are clearly observed, so it is natural that sCLEC2 is elevated. Conceivable. However, the presence of thrombus cannot be confirmed in a state where infarction cannot be confirmed, such as TIA, and it was thought that thrombosis-related markers (for example, D-dimer is famous) would not rise so much. The concentration of sCLEC2, which is suggested to be used as a biomarker, has increased, and it was surprising that it can be used as a blood biomarker for TIA risk assessment.
- the sCLEC2 concentration value of a patient suspected of having TIA is higher than that of healthy subjects or non-thrombotic disease groups, it can be judged that the possibility of TIA is high. Based on such a comparison, the sCLEC2 concentration before and after prophylactic treatment for cerebral infarction in TIA patients can be compared to predict the risk of developing cerebral infarction.
- the threshold value is appropriately set and used by comparing the sCLEC2 concentration in the patient-derived sample and the sCLEC2 concentration in the healthy subject-derived sample. can do.
- TIA can be evaluated when a significant change in sCLEC2 concentration is detected from the chronological record of sCLEC2 concentration measured before onset in the same patient.
- the blood sCLEC2 concentration is measured in a patient who describes symptoms suspected of TIA, and if the value is higher than that of healthy subjects, the possibility of TIA is high. Furthermore, it is possible to perform a test and administer an antiplatelet drug such as aspirin or an anticoagulant drug such as warfarin according to the risk determination. In addition, sCLEC2 concentration measurement is performed after taking antiplatelet drugs or anticoagulants as treatment after being diagnosed with TIA, and if the level is high, a treatment strategy can be determined, such as changing to a different type of drug or adding an additional dose. It contributes to the study for the purpose.
- an antiplatelet drug such aspirin or an anticoagulant drug such as warfarin
- sCLEC2 concentration measurement is performed after taking antiplatelet drugs or anticoagulants as treatment after being diagnosed with TIA, and if the level is high, a treatment strategy can be determined, such as changing to a different type of drug or adding an additional dose. It contributes to the study for the purpose.
- the original data or statistically processed data for calculating the determination threshold value data showing the correlation between the sCLEC2 concentration in plasma and the degree of platelet activation or various diseases may be used.
- a method for calculating the cut-off value analysis is performed to create an ROC curve (Receiver Operating Characteristic Curve) from the measured sCLEC2 value in plasma, and both the sensitivity and specificity of diagnosis are 80% or more. can be used as a cut-off value.
- a cutoff value can be calculated by performing a similar analysis when using a C2PAC index, which is a value obtained by dividing the sCLEC2 concentration by the platelet count.
- the cutoff value when using the C2PAC index can be 0.7.
- sCLEC2 is released into the blood with platelet activation.
- Existing platelet activation markers such as platelet factor 4 (PF4) and ⁇ -thromboglobulin ( ⁇ TG) have a problem of causing non-specific release due to physical stimulation by blood collection, but sCLEC2 is a signaling-dependent release mechanism that triggers platelet activation, and can be a marker that more accurately reflects platelet activation in vivo.
- PF4 platelet factor 4
- ⁇ TG ⁇ -thromboglobulin
- CLEC2 since CLEC2 is almost exclusively expressed in the platelet/megakaryocyte system in humans, it can be a platelet-specific marker with less false positives. Therefore, by measuring sCLEC2, it becomes possible to diagnose the platelet activation state at an early stage, and it can be used for the diagnosis of TIA.
- the measured sCLEC2 concentration may be divided by platelets and used for risk assessment. If the sCLEC2 concentration decreases with treatment, platelet activation tends to be suppressed. However, if the sCLEC2 concentration continues to be high or increases, the antithrombotic drug should be reviewed, added, or increased. can be done.
- the sCLEC2 concentration divided by the platelet count is referred to as the C2PAC index, and what is described as the sCLEC2 concentration in the diagnosis of TIA, even if not explicitly described as the C2PAC index, All can be performed by replacing the sCLEC2 concentration with the C2PAC index.
- the platelet count is usually measured using an automatic blood cell counter (hematometer), but a blood cell counter and a microscope are used. It is also possible to count using
- the C2PAC index which expresses the sCLEC2 concentration in plasma, for example, in pg/mL, expresses the blood platelet count, for example, in 1,000/mm 3 , and calculates the sCLEC2 concentration/platelet count.
- Any unit such as ng/mL or ng/L may be used for the concentration of sCLEC2 used in , and any unit such as 10,000/ mm3 may be used for the platelet count. Units should be used.
- the sCLEC2 concentration/index can take on different values, but essentially the same concept.
- ratio calculations will often be performed using measurements from a clinical laboratory instrument that measures sCLEC2 concentrations and from a hemocytometer that measures platelet counts.
- This calculation is preferably automatically calculated on a system such as a hospital inspection system, a hospital system, or an electronic medical chart connected to both measuring instruments in daily practice.
- a connecting system may be constructed, or a machine capable of simultaneously measuring sCLEC2 concentration and platelet count may be constructed.
- you may calculate manually using both data.
- those that show the correlation between the sCLEC2 concentration in plasma, the degree of platelet activation, and various diseases are used, for example, as the threshold for judgment, or the original data or statistically processed data for calculating the threshold for judgment.
- the plasma sCLEC2 concentration is divided by the blood platelet count to calculate the amount of sCLEC2 released per platelet, and this is used as an index for diagnosing thrombotic diseases, the platelet activity is not dependent on the blood platelet count. It is preferable because it makes it possible to evaluate the degree of conversion.
- the plasma sCLEC2 concentration is expressed as pg/mL (A)
- the blood platelet count is expressed as 1,000/mm 3 (B)
- the number obtained by dividing A by B is the number of platelet activation. It can be used as an index.
- the present invention does not exclude use in diagnosing cerebral infarction.
- Diagnosis of cerebral infarction includes, for example, unilateral limb paralysis (motor disorder), unilateral numbness (sensory disturbance), inability to speak, slurred speech (speech disorder), and unilateral visual impairment.
- Comprehensive judgment is made based on symptoms such as visual impairment (impaired vision), difficulty seeing objects on one side (visual field impairment), and the possibility of infarction being confirmed by imaging tests such as MRI.
- measuring the sCLEC2 concentration in blood according to the present invention is preferable because it can assist rapid and simple diagnosis of cerebral infarction.
- the present invention can meet this demand.
- blood sCLEC2 concentration is measured in patients diagnosed with acute cerebrovascular accident. At this time, it was confirmed that the sCLEC2 concentration was significantly elevated. If it is higher than the preset threshold, the probability of non-cardiogenic cerebral infarction is high, and if it is lower, it is judged that the probability of cardiogenic cerebral infarction is high. can do.
- the sCLEC2 concentration and other thrombostatic markers can be combined and used for differential diagnosis of cardiogenic cerebral infarction or non-cardiogenic cerebral infarction.
- blood sCLEC2 concentration and D-dimer concentration are measured in patients diagnosed with acute cerebrovascular accident. It is preferable to use samples drawn at the same time for the measurement, but the measurement may be performed using samples drawn separately. A person skilled in the art can appropriately determine the degree of influence even if there is a time difference in collecting blood for each concentration measurement.
- the concentration of sCLEC2 is divided by the concentration of D-dimer and used to calculate the sCLEC2/D-dimer ratio. In doing so, uniform concentration units should be used. It is preferably expressed in ⁇ g/mL, which is a general notation, but is not limited to this.
- Body (TAT) and prothrombin fragment F1+2 can also be used for determination of cardiogenicity/non-cardiogenicity in proportion to sCLEC2.
- thrombologic information such as whether the infarcted thrombus is fibrin-dominant or platelet-dominant, is very useful diagnostically.
- starting treatment is a matter of seconds, but in many cases it is unclear how long it will take to see an outpatient such as an emergency room.
- the differential diagnosis of cardiogenic cerebral infarction or non-cardiogenic cerebral infarction can be performed by a simple method such as the present invention, it will be possible to quickly select an appropriate treatment method. The use of evaluation methods is preferred.
- Anticoagulants and antiplatelet drugs are known as infusion therapeutic agents for the treatment of acute cerebral infarction, but the indications may differ even for the same anticoagulant.
- argatroban and heparin which are anticoagulants, are indicated for non-cardiogenic cerebral infarction, while heparin is indicated for cardiogenic cerebral infarction.
- anticoagulants argatroban
- argatroban if the infarct image is 1.5 cm or more within 2 days of onset, argatroban is indicated, but if it is not large or if it has been more than 3 days, it is not indicated. .
- the practice of the present invention is preferable because it not only enables cerebral infarction to be diagnosed, but also enables rapid differentiation between cardiogenic and non-cardiogenic stroke.
- drugs such as edaravone, which has a completely different mechanism of action from anticoagulants and antiplatelet drugs and are indicated for both cardiogenic and noncardiogenic drugs, are thought to reduce exacerbation of cerebral infarction by scavenging free radicals.
- treatment policies differ from country to country, such as the recent US treatment guidelines not mentioning them.
- the implementation of the present invention not only makes it possible to diagnose cerebral infarction, but also to rapidly distinguish between cardiogenic and non-cardiogenic infarction. preferred because it can be done.
- anticoagulants and antiplatelet drugs are first used in combination, and the next treatment strategy is determined while observing the effects in about 1 to 2 weeks.
- aspirin or warfarin is often selected as an oral drug.
- imaging tests can determine whether it is lacunar cerebral infarction, atherosclerotic cerebral infarction, or cardiogenic cerebral infarction, there is no difference in judgment, but even specialists can make judgments. It is said to be difficult.
- differential diagnosis of whether stroke is cardiogenic or noncardiogenic has been described, the use in the differential diagnosis of whether TIA is cardiogenic or noncardiogenic is described. is also possible.
- differential diagnosis of cardiogenic TIA and non-cardiogenic TIA it can be used in the same manner as in the differential diagnosis of cardiogenic cerebral infarction or non-cardiogenic cerebral infarction.
- TIA since no infarct image is observed in images such as MRI, such differential diagnosis can be made, which is more preferable.
- sCLEC2 concentration in plasma was measured according to the procedure shown below. (Preparation of measurement reagent and preparation of test sample) ⁇ Specimen diluent: 2% sodium octanoate and 0.5% n-octyl- ⁇ -D-glucoside (OG) using 0.1 mol/L HEPES buffer (pH 7.5) containing preservatives They were combined to obtain a specimen diluent.
- the antibody contained in the reagent was prepared as follows using the antibody described in Examples of Japanese Patent No. 6078845.
- ⁇ First antibody solution A mouse monoclonal antibody (11D5) that recognizes sCLEC2 was bound to magnetic latex particles (JSR) and dispersed in a 0.01 mol/L MES buffer solution (pH 6.0) containing a preservative. . -Second antibody solution: Another mouse monoclonal antibody (11E6) that recognizes sCLEC2 is labeled with alkaline phosphatase (ALP) by the maleimide method and dispersed in 0.01 mol/L MES buffer (pH 6.5) containing a preservative.
- ALP alkaline phosphatase
- Luminescent substrate solution 2-chloro-5-(4-methoxyspiro ⁇ 1,2-dioxetane-3,2′-(5′-chloro)-tricyclo[3.3.1.13,7]decane ⁇ - 4-yl)-1-phenylphosphate disodium (CDP-Star®: Applied Biosystems) was used.
- - B/F wash solution A buffer solution containing 0.1 mol/L citric acid (pH 6.5), 0.15 mol/L NaCl, and 0.1% Triton X-100 was used.
- Test sample 1 Recombinant hCLEC-2 protein diluted with buffer (0.025 mol/L HEPES, 0.14 mol/L NaCl, 0.1% sodium octanoate, 0.3% BSA) Test sample 1 was diluted with citrated plasma and used as test sample 2.
- a fully automatic clinical examination system STACIA registered trademark, manufactured by LSI stipulatece
- the sample diluent, the first antibody solution (magnetic latex reagent), and the second antibody solution (enzyme-labeled antibody reagent) were filled in STACIA-dedicated bottles and set in the apparatus. Measurements were carried out according to the operating method of the apparatus described below. Specifically, 40 ⁇ L of the specimen diluent was added to 10 ⁇ L of the sample and heated at 37° C. for several minutes, then 25 ⁇ L of the first antibody solution (magnetic latex reagent) was added and heated at 37° C. for several minutes.
- FIG. 1 shows a standard curve prepared using the hsCLEC2 protein as a standard.
- Example 2 Measurement of sCLEC2 in plasma specimens of TIA patients and healthy subjects>>> Using citrated plasma obtained from patients diagnosed with TIA with no new infarcts seen in interviews about clinical symptoms and MRI, and from healthy subjects, sCLEC2 concentration in plasma, platelet level, The sCLEC2/platelet ratio (C2PAC index) was measured (Table 1, Table 2).
- Example 3 Sensitivity and specificity of diagnosis of TIA by sCLEC2>>
- a ROC curve Receiveiver Operating Characteristic Curve
- Table 4 shows the diagnostic sensitivity and specificity when the cutoff value was varied from 60 pg/mL to 150 pg/mL in this analysis.
- the cut-off values showing both sensitivity and specificity of 80% or more ranged from 66 pg/mL to 148 pg/mL.
- Example 4 sCLEC2, D-dimer, sCLEC2/D-dimer ratio in cerebral infarction and TIA of each disease type>>>
- blood sCLEC2 in cardiogenic cerebral infarction, atherosclerotic cerebral infarction, and lacunar infarction was measured by the method of Example 1 (Fig. 4).
- a significant difference was confirmed by the Mann-Whitney U test between the sCLEC2 concentration in patients with cardiogenic cerebral infarction and the sCLEC2 concentration in patients with non-cardiogenic cerebral infarction, and the p-value was 0.002720 ( ⁇ 0.05).
- sCLEC2 was confirmed to be useful in distinguishing between cardiogenic and non-cardiogenic cerebral infarction. Furthermore, in order to distinguish between cardiogenic cerebral infarction and non-cardiogenic cerebral infarction with higher accuracy, the D-dimer contained in the sample collected from the cerebral infarction patient was subjected to LPIA-Genesis D-Dimer ( (manufactured by LSI Rulece). A fully automatic clinical laboratory analyzer STACIA (manufactured by LSI Rulece) was used for the measurement.
- D-dimer, sCLEC2, and the sCLEC2/D-dimer ratio the ability to differentiate between noncardiogenic cerebral infarction (atheromatous cerebral infarction and lacunar infarction) and cardiogenic cerebral infarction was analyzed with the ROC curve ( 5 to 7).
- the areas under the curve for D-dimer, sCLEC2 and sCLEC2/D-dimer ratio were 0.67, 0.73 and 0.79, respectively, and the sCLEC2/D-dimer ratio showed the best diagnostic ability.
- a representative example of TIA patients suspected of having cardiogenic origin was similarly confirmed using sCLEC2 and the sCLEC2/D-dimer ratio. A trend toward lower values was confirmed.
- the measurement of blood sCLEC2 and sCLEC2/platelet ratio of the present invention can be a clinical test that can be used for TIA diagnosis, and the sCLEC2 measurement reagent can be a TIA clinical test diagnostic agent.
- sCLEC2 in blood and the sCLEC2/D-dimer ratio can be clinical tests that can be used to diagnose cardiogenic/non-cardiogenic cerebral infarction and TIA. It becomes a non-cardiogenic laboratory diagnostic reagent.
Abstract
Description
[1]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者から採取した血液中に存在する、可溶型CLEC2濃度を測定する工程を含む、急性脳血管障害のリスク評価を行う方法。
[2]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者のリスク評価を行う方法であって、
(1)前記患者に由来する血液試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記可溶型CLEC2濃度と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性若しくは非心原性かどうかを相関させる工程、
を含む、[1]の方法。
[3]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者のリスク評価を行う方法であって、前記可溶型CLEC2濃度と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性又は非心原性かどうかを相関させる工程が、前記可溶型CLEC2濃度の変化に基づいて、リスクを前記患者が有するかどうかを評価することを含む、[2]の方法。
[4]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記急性脳血管障害が一過性脳虚血発作であり、可溶型CLEC2濃度と一過性脳虚血発作を相関させる工程で、前記可溶型sCLEC2濃度のカットオフ値が66~148pg/mLである、[2]又は[3]の方法。
[5]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者のリスク評価を行う方法であって、
(1)前記患者に由来する血液試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記試料中の血小板数を測定する工程、
(4)前記可溶型CLEC-2濃度を血小板数で除する工程、
(5)前記可溶型CLEC2濃度を血小板数で除した値と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性若しくは非心原性かどうかを相関させる工程、
を含む、[1]の方法。
[6]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記急性脳血管障害が一過性脳虚血発作であり、可溶型CLEC2濃度を血小板数で除した値と一過性脳虚血発作を相関させる工程で、前記可溶型sCLEC2濃度を血小板数で除した値のカットオフ値が0.55~0.7である、[5]の方法。
[7]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記急性脳血管障害が心原性か非心原性かを判定する材料を提供するものである、[1]~[4]のいずれの方法。
[8]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、
(1)前記患者に由来する血液試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記試料中の血小板数を測定する工程、
(4)前記可溶型CLEC-2濃度を凝固線溶マーカーで除する工程、
(5)前記可溶型CLEC2濃度を凝固線溶マーカーで除した値と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性若しくは非心原性かどうかを相関させる工程、
を含む、[1]の方法。
[9]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記凝固線溶マーカーがD-ダイマーである、[8]の方法。
[10]急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記患者に由来する試料の提供が、急性脳血管障害の症状の発生後48時間以内に、前記患者から採取された、[1]~[9]のいずれかの方法。
[11]可溶型CLEC2濃度を決定する工程が、高感度免疫測定法、例えば、化学発光免疫測定法、電気化学発光免疫測定法、蛍光免疫測定法である、[1]~[10]のいずれかの方法。
試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値、若しくは可溶型CLEC-2濃度を凝固線溶マーカーで除した値)を測定(又は決定)する、急性脳血管障害のリスク評価を行う方法、
試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値、若しくは可溶型CLEC-2濃度を凝固線溶マーカーで除した値)を測定(又は決定)する、急性脳血管障害のリスク評価を補助する方法、
急性脳血管障害のリスク評価のために、試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値、若しくは可溶型CLEC-2濃度を凝固線溶マーカーで除した値)を測定(又は決定)する方法、
試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値、若しくは可溶型CLEC-2濃度を凝固線溶マーカーで除した値)を測定(又は決定)することを特徴とする、急性脳血管障害のin vitroリスク評価方法、
可溶型CLEC2濃度を検出できる抗体の、急性脳血管障害のリスク評価用キットの製造における使用、
急性脳血管障害のリスク評価に必要な情報を提供するために、試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値、若しくは可溶型CLEC-2濃度を凝固線溶マーカーで除した値)を測定(又は決定)する方法
が含まれる。
TIAとは脳梗塞様の症状を一過性に呈するが画像検査で梗塞を確認できないものを意味する。脳梗塞様の症状とは、例えば、片側の手足の麻痺(運動障害)、片側のしびれ(感覚障害)、言葉が出て来ない、ろれつが回らない(言語障害)、片側の目が見えにくくなる(視力障碍)、片側にあるものが見えにくくなる(視野障害)等が挙げられるが、これに限定されるものではない。これらの症状は数分から長くて1時間程度で消失するため、問診により聞き取って診断する以外には方法がないが、症状の程度としては強いものから弱いものまでがあるが、その重篤度は問わない。脳梗塞患者は、上記の臨床症状に加えてMRI、CT等によって脳に梗塞像が確認された患者である。
例えば、カットオフ値を算出するための方法としては、測定した血漿中のsCLEC2の値からROC曲線(Receiver Operating Characteristic Curve)を作成する解析を行って、診断の感度と特異度が共に80%以上を示す濃度をカットオフ値として使用することができる。
本発明の方法において、sCLEC2によるTIAのカットオフ値は66pg/mL~148pg/mL間の任意の値を選ぶことが好適である。また、更に好適な態様として、sCLEC2濃度を血小板数で除した値であるC2PACインデックスを使用した場合にも同様の解析を行って、カットオフ値を算出することができる。C2PACインデックスを使用した場合のカットオフ値は、0.7とすることができる。
また、血漿中のsCLEC2濃度と血小板活性化の程度や各種疾患との相関を示すものを、例えば、判定用閾値、あるいは、判定用閾値を算出するためのオリジナルデータ又は統計処理データなどとして使用してもよい。
血漿中sCLEC2濃度を血中血小板数で除して、血小板当たりのsCLEC2放出量を計算してこれを指標として血栓性疾患の診断を行うと、血中血小板数に依存すること無く、血小板の活性化度合いを評価することが可能となるため、好ましい。具体的には、例えば血漿中sCLEC2濃度をpg/mLで表し(A)、血中血小板数を1,000個/mm3で表し(B)、AをBで除した数字を血小板活性化の指標とすることができる。
その他には、sCLEC2濃度と他の血栓止血マーカーとを組み合わせて心原性脳梗塞、又は非心原性脳梗塞の鑑別診断に使用することもできる。例えば、急性脳血管障害と診断された患者に対して血中のsCLEC2濃度とD-ダイマー濃度を測定する。測定は同時に採血された試料を用いるのが好ましいが、別々に採血をされた試料を使用して測定を実施してもよい。当業者であれば、それぞれの濃度測定をする採血時に時間差があるものであっても、影響の程度を適宜判断して実施することができる。
本実施例では、以下に示す手順に従って、血漿中のsCLEC2濃度を測定した。
(測定用試薬の作製と被検試料の調製)
・検体希釈液:防腐剤を含む0.1mol/LのHEPES緩衝液(pH7.5)を用いて、オクタン酸ナトリウム2%、n-オクチル-β-D-グルコシド(OG)0.5%になるように組み合わせて検体希釈液とした。
試薬に含まれる抗体は、特許第6078845号公報の実施例に記載の抗体を使用し、以下のように調製した。
・第1抗体溶液:磁性ラテックス粒子(JSR社)にsCLEC2を認識するマウスモノクローナル抗体(11D5)を結合させ、防腐剤を含む0.01mol/LのMES緩衝液(pH6.0)に分散させた。
・第2抗体溶液:sCLEC2を認識する別のマウスモノクローナル抗体(11E6)をマレイミド法によりアルカリホスファターゼ(ALP)標識し、防腐剤を含む0.01mol/LのMES緩衝液(pH6.5)に分散させた。
・発光基質溶液:2-クロロ-5-(4-メトキシスピロ{1,2-ジオキセタン-3,2´-(5´-クロロ)-トリシクロ[3.3.1.13,7]デカン}-4-イル)-1-フェニルホスフェート・二ナトリウム(CDP-Star(登録商標):アプライドバイオシステム社)を使用した。
・B/F洗浄液:0.1mol/Lクエン酸(pH6.5)、0.15mol/L NaCl、0.1%TritonX-100、を含む緩衝液を使用した。
・被検試料:組換えhCLEC-2蛋白質を緩衝液(0.025mol/L HEPES、0.14mol/L NaCl、0.1%オクタン酸ナトリウム、0.3%BSA)を用いて希釈したものを被検試料1、クエン酸血漿を用いて希釈したものを被検試料2として使用した。
測定には、全自動臨床検査システムSTACIA(登録商標、LSIメディエンス社製)を使用した。
STACIA専用ボトルに、調製した検体希釈液、第1抗体溶液(磁性ラテックス試薬)、第2抗体溶液(酵素標識抗体試薬)をそれぞれ充填し、装置にセットした。以下前記装置の運転方法に従い測定した。
具体的には、試料10μLに検体希釈液40μLを加え、37℃で数分間加温した後、第1抗体溶液(磁性ラテックス試薬)25μLを加え、37℃で数分間加温した。次いで、B/F分離を行い、50μLの第2抗体溶液(酵素標識抗体試薬)を加え、37℃で数分間加温し、再度B/F分離を行った後、100μLの発光基質溶液を加え、37℃で数分反応後にシグナル強度(カウント)を測定した。
図1にhsCLEC2蛋白質を標準品として用いて作成した標準曲線を示した。
臨床症状の問診及びMRIで新規の梗塞像が見られずTIAと診断された患者、及び健常者から得られたクエン酸血漿を用いて、実施例1の方法で血漿中sCLEC2濃度、血小板値、sCLEC2/血小板比(C2PACインデックス)を測定した(表1、表2)。
また、別途測定したC2PACインデックスの健常者の上限値は0.7であった。
実施例2の測定値を、sCLEC2のカットオフ値を変化させて解析すると、図3のようなROC曲線(Receiver Operating Characteristic Curve)が得られ、その曲線下面積は0.994と非常に良好であった。
この解析でカットオフ値を60pg/mLから150pg/mLに変化させた時の診断の感度と特異度を表4に示す。感度、特異度共に80%以上を示すカットオフ値は66pg/mLから148pg/mLであった。
以上のことより、sCLEC2の測定、並びにsCLEC2/血小板比(C2PACインデックス)は、TIAの診断に対して簡便、且つ正確に客観的なデータを提供する手段となる。
脳梗塞と診断された患者の内、心原性脳梗塞、アテローム性脳梗塞、ラクナ梗塞の血中sCLEC2を実施例1の方法で測定した(図4)。心原性脳梗塞患者のsCLEC2濃度と非心原性脳梗塞患者のsCLEC2濃度とで、マン・ホイットニーU検定により有意差を確認したところ、p値は、0.002720(<0.05)となり、sCLEC2が心原性脳梗塞か非心原性脳梗塞かの鑑別に有用であることが確認された。
更に、より高精度に心原性脳梗塞と非心原性脳梗塞との鑑別を実施するため、上記脳梗塞患者から採取された試料中に含まれるD-ダイマーをLPIA-Genesis D-Dimer(LSIメディエンス社製)にて測定した。測定には全自動臨床検査分析器STACIA(LSIメディエンス社製)を用いた。D-ダイマー、sCLEC2並びにsCLEC2/D-ダイマー比を用いた時の、非心原性脳梗塞(アテローム性脳梗塞とラクナ梗塞)と心原性脳梗塞の鑑別診断能をROC曲線で解析した(図5~図7)。D-ダイマー、sCLEC2、sCLEC2/D-ダイマー比の曲線下面積は各々0.67、0.73、0.79であり、sCLEC2/D-ダイマー比が最も良い診断能を示した。
心原性であることが疑われるTIA患者の代表例についても同様にsCLEC2並びにsCLEC2/D-ダイマー比を用いて確認をしたところ、脳梗塞の場合と同様に、非心原性TIA患者よりも低値となる傾向が確認された。
Claims (11)
- 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者から採取した血液中に存在する、可溶型CLEC2濃度を測定する工程を含む、急性脳血管障害のリスク評価を行う方法。
- 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者のリスク評価を行う方法であって、
(1)前記患者に由来する血液試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記可溶型CLEC2濃度と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性若しくは非心原性かどうかを相関させる工程、
を含む、請求項1に記載の方法。 - 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者のリスク評価を行う方法であって、前記可溶型CLEC2濃度と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性又は非心原性かどうかを相関させる工程が、前記可溶型CLEC2濃度の変化に基づいて、リスクを前記患者が有するかどうかを評価することを含む、請求項2に記載の方法。
- 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記急性脳血管障害が一過性脳虚血発作であり、可溶型CLEC2濃度と一過性脳虚血発作を相関させる工程で、前記可溶型sCLEC2濃度のカットオフ値が66~148pg/mLである、請求項2又は3に記載の方法。
- 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者のリスク評価を行う方法であって、
(1)前記患者に由来する血液試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記試料中の血小板数を測定する工程、
(4)前記可溶型CLEC-2濃度を血小板数で除する工程、
(5)前記可溶型CLEC2濃度を血小板数で除した値と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性若しくは非心原性かどうかを相関させる工程、
を含む、請求項1に記載の方法。 - 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記急性脳血管障害が一過性脳虚血発作であり、可溶型CLEC2濃度を血小板数で除した値と一過性脳虚血発作を相関させる工程で、前記可溶型sCLEC2濃度を血小板数で除した値のカットオフ値が0.55~0.7である、請求項5に記載の方法。
- 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記急性脳血管障害が心原性か非心原性かを判定する材料を提供するものである、請求項1~4のいずれか一項に記載の方法。
- 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、
(1)前記患者に由来する血液試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記試料中の血小板数を測定する工程、
(4)前記可溶型CLEC-2濃度を凝固線溶マーカーで除する工程、
(5)前記可溶型CLEC2濃度を凝固線溶マーカーで除した値と、前記患者における急性脳血管障害の存在若しくは非存在、転帰の可能性、又は心原性若しくは非心原性かどうかを相関させる工程、
を含む、請求項1に記載の方法。 - 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記凝固線溶マーカーがD-ダイマーである、請求項8に記載の方法。
- 急性脳血管障害の罹患が疑われる患者又は急性脳血管障害と診断された患者における急性脳血管障害のリスク評価を行う方法であって、前記患者に由来する試料の提供が、急性脳血管障害の症状の発生後48時間以内に、前記患者から採取された、請求項1~9のいずれか一項に記載の方法。
- 可溶型CLEC2濃度を決定する工程が、高感度免疫測定法、例えば、化学発光免疫測定法、電気化学発光免疫測定法、蛍光免疫測定法である、請求項1~10のいずれか一項に記載の方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023525849A JPWO2022255348A1 (ja) | 2021-05-31 | 2022-05-31 | |
| EP22816094.1A EP4350349A1 (en) | 2021-05-31 | 2022-05-31 | Method for evaluating risk of acute cerebral vascular disease using soluble clec2 |
| CN202280038994.9A CN117813505A (zh) | 2021-05-31 | 2022-05-31 | 使用了可溶型clec2的急性脑血管病的风险评价方法 |
| KR1020237044827A KR20240017000A (ko) | 2021-05-31 | 2022-05-31 | 가용형 clec2를 이용한 급성 뇌혈관 장애의 리스크 평가방법 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-091606 | 2021-05-31 | ||
| JP2021091606 | 2021-05-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022255348A1 true WO2022255348A1 (ja) | 2022-12-08 |
Family
ID=84323407
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/022076 WO2022255348A1 (ja) | 2021-05-31 | 2022-05-31 | 可溶型clec2を用いた急性脳血管障害のリスク評価方法 |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP4350349A1 (ja) |
| JP (1) | JPWO2022255348A1 (ja) |
| KR (1) | KR20240017000A (ja) |
| CN (1) | CN117813505A (ja) |
| WO (1) | WO2022255348A1 (ja) |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050107598A1 (en) * | 2001-12-19 | 2005-05-19 | Baum Peter R. | C-type lectin polypeptide, polynucleotide and methods of making and use thereof |
| JP2007070359A (ja) * | 2005-09-06 | 2007-03-22 | Univ Of Yamanashi | 止血疾患治療用の医薬組成物 |
| EP2269597A1 (en) * | 2009-06-22 | 2011-01-05 | CSL Behring GmbH | CLEC-2 is an essential platelet activating receptor in hemostasis and thrombosis |
| EP2272874A1 (en) * | 2009-07-10 | 2011-01-12 | CSL Behring GmbH | Role of PLD1 in thrombus formation and integrin alpha IIb beta 3 activation |
| JP2014070942A (ja) * | 2012-09-28 | 2014-04-21 | Mitsubishi Chemical Medience Corp | 可溶型clec−2に基づく血小板活性化測定方法 |
| WO2015053381A1 (ja) * | 2013-10-10 | 2015-04-16 | 幸成 加藤 | 抗ポドプラニン抗体 |
| JP2020032797A (ja) | 2018-08-28 | 2020-03-05 | スズキ株式会社 | ニーエアバッグ取付け構造 |
| JP2021003671A (ja) | 2019-06-26 | 2021-01-14 | 稔 甲斐 | 閉塞防止機能付振動篩用網及び振動篩装置 |
| JP2021060870A (ja) | 2019-10-08 | 2021-04-15 | 株式会社デンソー | 電子制御装置 |
| WO2021172493A1 (ja) * | 2020-02-28 | 2021-09-02 | 株式会社Lsiメディエンス | 可溶型clec-2と血小板数に基づく血小板活性化測定方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6078845U (ja) | 1983-11-04 | 1985-06-01 | ライオン株式会社 | 食用廃油固化処理用具 |
-
2022
- 2022-05-31 CN CN202280038994.9A patent/CN117813505A/zh active Pending
- 2022-05-31 KR KR1020237044827A patent/KR20240017000A/ko unknown
- 2022-05-31 JP JP2023525849A patent/JPWO2022255348A1/ja active Pending
- 2022-05-31 EP EP22816094.1A patent/EP4350349A1/en active Pending
- 2022-05-31 WO PCT/JP2022/022076 patent/WO2022255348A1/ja active Application Filing
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050107598A1 (en) * | 2001-12-19 | 2005-05-19 | Baum Peter R. | C-type lectin polypeptide, polynucleotide and methods of making and use thereof |
| JP2007070359A (ja) * | 2005-09-06 | 2007-03-22 | Univ Of Yamanashi | 止血疾患治療用の医薬組成物 |
| EP2269597A1 (en) * | 2009-06-22 | 2011-01-05 | CSL Behring GmbH | CLEC-2 is an essential platelet activating receptor in hemostasis and thrombosis |
| EP2272874A1 (en) * | 2009-07-10 | 2011-01-12 | CSL Behring GmbH | Role of PLD1 in thrombus formation and integrin alpha IIb beta 3 activation |
| JP2014070942A (ja) * | 2012-09-28 | 2014-04-21 | Mitsubishi Chemical Medience Corp | 可溶型clec−2に基づく血小板活性化測定方法 |
| JP6078845B2 (ja) | 2012-09-28 | 2017-02-15 | 株式会社Lsiメディエンス | 可溶型clec−2に基づく血小板活性化測定方法 |
| WO2015053381A1 (ja) * | 2013-10-10 | 2015-04-16 | 幸成 加藤 | 抗ポドプラニン抗体 |
| JP2020032797A (ja) | 2018-08-28 | 2020-03-05 | スズキ株式会社 | ニーエアバッグ取付け構造 |
| JP2021003671A (ja) | 2019-06-26 | 2021-01-14 | 稔 甲斐 | 閉塞防止機能付振動篩用網及び振動篩装置 |
| JP2021060870A (ja) | 2019-10-08 | 2021-04-15 | 株式会社デンソー | 電子制御装置 |
| WO2021172493A1 (ja) * | 2020-02-28 | 2021-09-02 | 株式会社Lsiメディエンス | 可溶型clec-2と血小板数に基づく血小板活性化測定方法 |
Non-Patent Citations (22)
| Title |
|---|
| F. KAZAMA ET AL., PLATELETS, vol. 26, no. 8, 2015, pages 711 - 719 |
| GUO MI, ZHANG HAN, LV QING-WEI, HUANG HANG-BIN, SHEN LIANG-JUN: "Higher plasma C-type lectin-like receptor 2 concentrations for prediction of higher risk of 30-day mortality in isolated severe blunt traumatic brain injury", CLINICA CHIMICA ACTA, vol. 496, 1 September 2019 (2019-09-01), AMSTERDAM, NL , pages 1 - 6, XP093010593, ISSN: 0009-8981, DOI: 10.1016/j.cca.2019.06.014 * |
| HIDEO WADA: "SY3-1 Significance of plasma sCLEC-2 measurement, a platelet activation marker, in diagnosing acute ischemic stroke", JOURNAL OF THE JAPANESE SOCIETY FOR LABORATORY HEMATOLOGY, vol. 23, no. academic conference issue, 4 July 2022 (2022-07-04) - 31 July 2022 (2022-07-31), pages S91, XP009541639, ISSN: 1347-2836 * |
| INOUE KATSUE: "CLEC-2, a platelet-activating receptor responsible for fetal angiolymphatic separation and maintenance of vascular integrity in adults", JOURNAL OF JAPANESE BIOCHEMICAL SOCIETY, vol. 89, no. 3, 25 June 2017 (2017-06-25), pages 377 - 383, XP093010568, DOI: 10.14952/SEIKAGAKU.2017.890377 * |
| INOUE KATSUE: "Clinical research on the measurement -ftof soluble CLEC-2 (diabetes, myocardial infarction,cerebral infarction) (approval number: 955)", UNIVERSITY OF YAMANASHI SCHOOL OF MEDICINE, 8 August 2020 (2020-08-08), pages 1 - 2, XP093010882, Retrieved from the Internet <URL:https://www.med.yamanashi.ac.jp/rinri/files/20200616_2289_%E8%87%A8%E5%BA%8A%E6%A4%9C%E6%9F%BB%E5%8C%BB%E5%AD%A6%E8%AC%9B%E5%BA%A7_%E6%95%97%E8%A1%80%E7%97%87%E6%80%A7DIC%E6%82%A3%E8%80%85%E3%81%AB%E3%81%8A%E3%81%91%E3%82%8BsCLEC2%E6%B8%AC%E5%AE%9A%E3%81%AE%E6%9C%89%E7%94%A8%E6%80%A7%E3%81%AB%E9%96%A2%E3%81%99%E3%82%8B%E8%87%A8%E5%BA%8A%E8%A9%95%E4%BE%A1.pdf> [retrieved on 20230102] * |
| INOUE KATSUE: "Development of antiplatelet drugs, anti-metastatic/neoplastic drugs, and tests targeting the novel receptor CLEC-2 on platelets", RESEARCH-ER.JP, 1 January 2013 (2013-01-01), pages 1 - 5, XP093010900, Retrieved from the Internet <URL:https://research-er.jp/projects/view/106336%3E> [retrieved on 20230102] * |
| KOICHI YAMAGUCHI, SATOMI NAGAYA, MASAHIDE KAWAMURA, SHUHEI MATSUSHITA, KOTARO MORI, SHOICHI SATO, HIROKI TSUJIGUCHI, AKINORI HARA,: "O6-4 Clinical usefulness of soluble C-type lectin-like receptor-2 (sCLEC-2)", JOURNAL OF THE JAPANESE SOCIETY FOR LABORATORY HEMATOLOGY, vol. 22, no. academic conference issue, 18 August 2021 (2021-08-18) - 12 September 2021 (2021-09-12), pages S130, XP009541638, ISSN: 1347-2836 * |
| KOICHI YAMAGUCHI: "Investigation of soluble C-type lectin-type receptor (sCLEC2) in antiphospholipid antibody syndrome", JAPANESE JOURNAL OF THROMBOSIS AND HEMOSTASIS, vol. 31, no. 2, 1 May 2020 (2020-05-01) - 20 June 2020 (2020-06-20), JP , pages 260, XP009541631, ISSN: 0915-7441 * |
| MENG DANYANG, LUO MAN, LIU BEIBEI: "The Role of CLEC-2 and Its Ligands in Thromboinflammation", FRONTIERS IN IMMUNOLOGY, vol. 12, no. 688643, pages 1 - 9, XP093010598, DOI: 10.3389/fimmu.2021.688643 * |
| MENG DANYANG; MA XIAOHUA; LI HUI; WU XUECHUN; CAO YONGJUN; MIAO ZHIGANG; ZHANG XIA: "A Role of the Podoplanin-CLEC-2 Axis in Promoting Inflammatory Response After Ischemic Stroke in Mice", NEUROTOXICITY RESEARCH, vol. 39, no. 2, 1 January 1900 (1900-01-01), CH , pages 477 - 488, XP037407945, ISSN: 1029-8428, DOI: 10.1007/s12640-020-00295-w * |
| MIKA UEDA, SHUHEI MATSUSHITA, TOSHIMITSU SHIRAI, FUMITOMO KAZAMA, NOZOMI ODA, DAISUKE NAGATA, HIROYUKI YOSHIDA, MASAHIDE KAWAMURA,: "O24-4 Performance evaluation of soluble CLEC-2 automated assay reagent based on chemiluminescent enzyme immunoassay", JOURNAL OF THE JAPANESE SOCIETY FOR LABORATORY HEMATOLOGY, vol. 23, no. academic conference issue, 4 July 2022 (2022-07-04) - 31 July 2022 (2022-07-31), pages S176, XP009541629, ISSN: 1347-2836 * |
| NAGATA MAKOTO: "Establishment of a method for measuring plasma sCLEC-2 and its clinical application aiming to prevent arterial thrombosis", GRANTS-IN-AID FOR SCIENTIFIC RESEARCH, 1 January 2013 (2013-01-01), pages 1 - 4, XP093010678 * |
| NIESWANDT B., PLEINES I., BENDER M.: "Platelet adhesion and activation mechanisms in arterial thrombosis and ischaemic stroke : Platelet adhesion and activation mechanisms", JOURNAL OF THROMBOSIS AND HAEMOSTASIS, vol. 9, 1 July 2011 (2011-07-01), GB , pages 92 - 104, XP093010740, ISSN: 1538-7933, DOI: 10.1111/j.1538-7836.2011.04361.x * |
| NISHIGAKI AKISATO, ICHIKAWA YUHUKO, EZAKI MINORU, YAMAMOTO AKITAKA, SUZUKI KENJI, TACHIBANA KEI, KAMON TOSHITAKA, HORIE SHOTARO, M: "Soluble C-Type Lectin-Like Receptor 2 Elevation in Patients with Acute Cerebral Infarction", JOURNAL OF CLINICAL MEDICINE, vol. 10, no. 3408, pages 1 - 8, XP093010596, DOI: 10.3390/jcm10153408 * |
| OSAMU INOUE: "Soluble Platelet Activating Receptor CLEC-2, Production Mechanism and Clinical Application of Soluble CLEC-2", JAPANESE JOURNAL OF THROMBOSIS AND HEMOSTASIS, vol. 31, no. 2, 1 May 2020 (2020-05-01) - 20 June 2020 (2020-06-20), JP , pages 135, XP009541640, ISSN: 0915-7441 * |
| SUZUKI-INOUE KATSUE: "An overview of the novel platelet activation receptor CLEC-2", JOURNAL OF THROMBOSIS AND HEMOSTASIS, vol. 20, no. 4, 1 January 2009 (2009-01-01), pages 401 - 405, XP093010676 * |
| WU X., ZHANG W., LI H., YOU S., SHI J., ZHANG C., SHI R., HUANG Z., CAO Y., ZHANG X.: "Plasma C‐type lectin‐like receptor 2 as a predictor of death and vascular events in patients with acute ischemic stroke", EUROPEAN JOURNAL OF NEUROLOGY, vol. 26, no. 10, 1 October 2019 (2019-10-01), GB , pages 1334 - 1340, XP093010923, ISSN: 1351-5101, DOI: 10.1111/ene.13984 * |
| YAMASHITA ET AL., THROMBOSIS RESEARCH, vol. 178, 2019, pages 54 - 58 |
| YOSHIKI YAMASHITA: "Elevated sCLEC2 plasma levels in patients with thrombotic microangiopathy", JAPANESE JOURNAL OF THROMBOSIS AND HEMOSTASIS, vol. 30, no. 2, 1 May 2019 (2019-05-01) - 22 June 2019 (2019-06-22), JP , pages 427, XP009541641, ISSN: 0915-7441 * |
| YUKO ICHIKAWA, NOZOMU IKEDA, MINORU EZAKI, MOTOKO TANAKA, MITSUO OGAWA, HIKARU MIZUNO, TSUBASA SATO, YUMI WATANABE, KATSUYA SHIRAK: "O19-2 Investigation of plasma sCLEC-2 levels in patients with cerebral infarction", JOURNAL OF THE JAPANESE SOCIETY FOR LABORATORY HEMATOLOGY, vol. 23, no. academic conference issue, 4 July 2022 (2022-07-04) - 31 July 2022 (2022-07-31), pages S161, XP009541628, ISSN: 1347-2836 * |
| ZHANG ET AL., STROKE, vol. 50, 2019, pages 45 - 52 |
| ZHANG XIA, ZHANG WEI, WU XUECHUN, LI HUI, ZHANG CHUNYUAN, HUANG ZHICHAO, SHI RONGFANG, YOU TAO, SHI JIJUN, CAO YONGJUN: "Prognostic Significance of Plasma CLEC-2 (C-Type Lectin-Like Receptor 2) in Patients With Acute Ischemic Stroke", STROKE, vol. 50, no. 1, 1 January 2019 (2019-01-01), US , pages 45 - 52, XP093010739, ISSN: 0039-2499, DOI: 10.1161/STROKEAHA.118.022563 * |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20240017000A (ko) | 2024-02-06 |
| EP4350349A1 (en) | 2024-04-10 |
| JPWO2022255348A1 (ja) | 2022-12-08 |
| CN117813505A (zh) | 2024-04-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Goldhaber et al. | Quantitative plasma D-dimer levels among patients undergoing pulmonary angiography for suspected pulmonary embolism | |
| JP5221333B2 (ja) | 意識障害患者における病態の検出方法及び検出用キット | |
| JPWO2005080982A1 (ja) | A型及びb型急性大動脈解離と急性心筋梗塞の鑑別方法及び鑑別用キット | |
| US20230168260A1 (en) | Method for measuring platelet activation based on soluble clec-2 and platelet count | |
| Poredos et al. | Preclinical carotid atherosclerosis as an indicator of polyvascular disease: a narrative review | |
| JP2009539080A (ja) | 急性冠状動脈症候群のリスクを有する個体の識別のためのmrp8/14レベルの使用 | |
| US20090081703A1 (en) | Method for detecting deep venous thrombosis | |
| Çildag et al. | Pulmonary artery obstruction index and right ventricular dysfunction signs in initial and follow up pulmonary computed tomography angiography in acute pulmonary embolism | |
| WO2022255348A1 (ja) | 可溶型clec2を用いた急性脳血管障害のリスク評価方法 | |
| JPWO2012067151A1 (ja) | CartilageAcidicProtein1蛋白質による脳梗塞の検査方法 | |
| WO2022255349A1 (ja) | 可溶型clec2を用いた出血性脳卒中のリスク評価法 | |
| EP1640719A1 (en) | Method of adjudicating on acute aortic dissection and reagent for adjudication | |
| JP6316200B2 (ja) | サイクロフィリンaによる心血管疾患の検査方法 | |
| JP2008516218A (ja) | 急性冠症候群の疑いを除外するinvitro診断方法 | |
| WO2022154045A1 (ja) | 可溶型clec2を用いた癌患者における血栓症リスクの予測方法 | |
| JP6004322B2 (ja) | 心血管イベントの発症リスクの検査方法 | |
| JP2011237402A (ja) | ガレクチン−3結合蛋白質による脳梗塞の検査方法 | |
| RU2704224C1 (ru) | Способ прогнозирования мультифокального атеросклеротического поражения | |
| JP2012107935A (ja) | Desmoglein−2蛋白質による脳梗塞の検査方法 | |
| Abdullah et al. | Diagnostic Value of D-Dimer’s Serum Level in Patients with Cerebral Venous Thrombosis | |
| Ahmed et al. | D-dimer as Sensitive parameters in COVID-19 (Study of 200 patients) | |
| JP2024027767A (ja) | 可溶型clec2を用いた血小板減少性疾患の診断法 | |
| ARMAGAN et al. | SERUM COPEPTIN LEVEL AS A BIOMARKER FOR DETECTING TRANSIENT ISCHEMIC ATTACK IN THE EMERGENCY ROOM: A PROSPECTIVE CASE-CONTROL STUDY | |
| RU2602914C1 (ru) | Способ оценки степени тяжести воспалительных заболеваний органов малого таза | |
| Reddy | A Study of Carotid Artery Intima Media Thickness and Atherosclerotic Risk Profile in Acute Ischemic Stroke |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22816094 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023525849 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 20237044827 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022816094 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022816094 Country of ref document: EP Effective date: 20240102 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023135647 Country of ref document: RU |