WO2022154045A1 - 可溶型clec2を用いた癌患者における血栓症リスクの予測方法 - Google Patents
可溶型clec2を用いた癌患者における血栓症リスクの予測方法 Download PDFInfo
- Publication number
- WO2022154045A1 WO2022154045A1 PCT/JP2022/000891 JP2022000891W WO2022154045A1 WO 2022154045 A1 WO2022154045 A1 WO 2022154045A1 JP 2022000891 W JP2022000891 W JP 2022000891W WO 2022154045 A1 WO2022154045 A1 WO 2022154045A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cancer
- concentration
- risk
- thrombosis
- soluble
- Prior art date
Links
- 201000011510 cancer Diseases 0.000 title claims abstract description 105
- 206010028980 Neoplasm Diseases 0.000 title claims abstract description 103
- 208000007536 Thrombosis Diseases 0.000 title claims abstract description 70
- 238000000034 method Methods 0.000 title claims abstract description 61
- 102100032529 C-type lectin domain family 1 member B Human genes 0.000 claims abstract description 41
- 210000004369 blood Anatomy 0.000 claims abstract description 29
- 239000008280 blood Substances 0.000 claims abstract description 29
- 101000942284 Homo sapiens C-type lectin domain family 1 member B Proteins 0.000 claims abstract 8
- 101710160442 C-type lectin domain family 1 member B Proteins 0.000 claims description 33
- 238000012502 risk assessment Methods 0.000 claims description 19
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 17
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 17
- 201000002528 pancreatic cancer Diseases 0.000 claims description 17
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 17
- 208000003174 Brain Neoplasms Diseases 0.000 claims description 14
- 238000012544 monitoring process Methods 0.000 claims description 13
- 230000015271 coagulation Effects 0.000 claims description 10
- 238000005345 coagulation Methods 0.000 claims description 10
- 238000001356 surgical procedure Methods 0.000 claims description 10
- 208000035134 Trousseau syndrome Diseases 0.000 claims description 7
- 229940127218 antiplatelet drug Drugs 0.000 claims description 7
- 239000003550 marker Substances 0.000 claims description 6
- 239000003814 drug Substances 0.000 claims description 5
- 230000000694 effects Effects 0.000 claims description 5
- 229940079593 drug Drugs 0.000 claims description 4
- 230000023597 hemostasis Effects 0.000 claims description 4
- 206010041823 squamous cell carcinoma Diseases 0.000 claims description 4
- 206010027406 Mesothelioma Diseases 0.000 claims description 3
- 210000003679 cervix uteri Anatomy 0.000 claims description 3
- 210000003238 esophagus Anatomy 0.000 claims description 3
- 210000004072 lung Anatomy 0.000 claims description 3
- 208000014767 Myeloproliferative disease Diseases 0.000 claims description 2
- 239000000090 biomarker Substances 0.000 abstract description 7
- 238000011156 evaluation Methods 0.000 abstract description 6
- 238000001727 in vivo Methods 0.000 abstract description 4
- 230000035602 clotting Effects 0.000 abstract 1
- 210000001772 blood platelet Anatomy 0.000 description 58
- 206010047249 Venous thrombosis Diseases 0.000 description 25
- 206010051055 Deep vein thrombosis Diseases 0.000 description 22
- 239000003154 D dimer Substances 0.000 description 17
- 239000000523 sample Substances 0.000 description 17
- 108010052295 fibrin fragment D Proteins 0.000 description 16
- 238000011282 treatment Methods 0.000 description 16
- 206010014522 Embolism venous Diseases 0.000 description 15
- 208000004043 venous thromboembolism Diseases 0.000 description 15
- 102000004169 proteins and genes Human genes 0.000 description 14
- 108090000623 proteins and genes Proteins 0.000 description 14
- 239000003146 anticoagulant agent Substances 0.000 description 13
- 229940127219 anticoagulant drug Drugs 0.000 description 13
- 238000005259 measurement Methods 0.000 description 12
- 210000004027 cell Anatomy 0.000 description 11
- 238000002560 therapeutic procedure Methods 0.000 description 10
- 201000010099 disease Diseases 0.000 description 9
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 9
- 208000032843 Hemorrhage Diseases 0.000 description 8
- 208000034158 bleeding Diseases 0.000 description 8
- 230000000740 bleeding effect Effects 0.000 description 8
- 206010020608 Hypercoagulation Diseases 0.000 description 7
- 230000004913 activation Effects 0.000 description 7
- 230000015572 biosynthetic process Effects 0.000 description 7
- 230000010118 platelet activation Effects 0.000 description 7
- 206010003178 Arterial thrombosis Diseases 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 230000002980 postoperative effect Effects 0.000 description 6
- 206010062608 Endocarditis noninfective Diseases 0.000 description 5
- 208000024369 Libman-Sacks endocarditis Diseases 0.000 description 5
- 102100037265 Podoplanin Human genes 0.000 description 5
- 101710118150 Podoplanin Proteins 0.000 description 5
- 230000005856 abnormality Effects 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 238000003745 diagnosis Methods 0.000 description 5
- 238000003018 immunoassay Methods 0.000 description 5
- 201000007261 marantic endocarditis Diseases 0.000 description 5
- 208000016135 nonbacterial thrombotic endocarditis Diseases 0.000 description 5
- 108010073385 Fibrin Proteins 0.000 description 4
- 102000009123 Fibrin Human genes 0.000 description 4
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 4
- 208000024659 Hemostatic disease Diseases 0.000 description 4
- 241000282412 Homo Species 0.000 description 4
- 206010027476 Metastases Diseases 0.000 description 4
- 230000008859 change Effects 0.000 description 4
- 229950003499 fibrin Drugs 0.000 description 4
- 230000003027 hypercoagulation Effects 0.000 description 4
- 230000009401 metastasis Effects 0.000 description 4
- 230000036470 plasma concentration Effects 0.000 description 4
- 238000004393 prognosis Methods 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 3
- 208000005189 Embolism Diseases 0.000 description 3
- 102000002262 Thromboplastin Human genes 0.000 description 3
- 108010000499 Thromboplastin Proteins 0.000 description 3
- 229960001138 acetylsalicylic acid Drugs 0.000 description 3
- 210000000601 blood cell Anatomy 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 3
- 239000011859 microparticle Substances 0.000 description 3
- 239000008363 phosphate buffer Substances 0.000 description 3
- 239000002953 phosphate buffered saline Substances 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000012475 sodium chloride buffer Substances 0.000 description 3
- BYKRNSHANADUFY-UHFFFAOYSA-M sodium octanoate Chemical compound [Na+].CCCCCCCC([O-])=O BYKRNSHANADUFY-UHFFFAOYSA-M 0.000 description 3
- 208000010110 spontaneous platelet aggregation Diseases 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- 201000005665 thrombophilia Diseases 0.000 description 3
- 230000001732 thrombotic effect Effects 0.000 description 3
- 210000003462 vein Anatomy 0.000 description 3
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- 108010039209 Blood Coagulation Factors Proteins 0.000 description 2
- 102000015081 Blood Coagulation Factors Human genes 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 2
- 206010033128 Ovarian cancer Diseases 0.000 description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 description 2
- 208000010378 Pulmonary Embolism Diseases 0.000 description 2
- 208000005718 Stomach Neoplasms Diseases 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 230000002159 abnormal effect Effects 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 229960002685 biotin Drugs 0.000 description 2
- 239000011616 biotin Substances 0.000 description 2
- 239000003114 blood coagulation factor Substances 0.000 description 2
- 238000010241 blood sampling Methods 0.000 description 2
- 229940098773 bovine serum albumin Drugs 0.000 description 2
- 206010008118 cerebral infarction Diseases 0.000 description 2
- 208000026106 cerebrovascular disease Diseases 0.000 description 2
- 238000002512 chemotherapy Methods 0.000 description 2
- 230000007812 deficiency Effects 0.000 description 2
- 206010017758 gastric cancer Diseases 0.000 description 2
- 208000005017 glioblastoma Diseases 0.000 description 2
- 230000001900 immune effect Effects 0.000 description 2
- 238000003317 immunochromatography Methods 0.000 description 2
- 208000032839 leukemia Diseases 0.000 description 2
- 210000000265 leukocyte Anatomy 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 210000003141 lower extremity Anatomy 0.000 description 2
- 201000005202 lung cancer Diseases 0.000 description 2
- 208000020816 lung neoplasm Diseases 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 210000003593 megakaryocyte Anatomy 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 210000000056 organ Anatomy 0.000 description 2
- 230000001575 pathological effect Effects 0.000 description 2
- 239000000106 platelet aggregation inhibitor Substances 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 201000011549 stomach cancer Diseases 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 208000011580 syndromic disease Diseases 0.000 description 2
- 230000009885 systemic effect Effects 0.000 description 2
- 201000005060 thrombophlebitis Diseases 0.000 description 2
- 230000036962 time dependent Effects 0.000 description 2
- 208000019553 vascular disease Diseases 0.000 description 2
- UAIUNKRWKOVEES-UHFFFAOYSA-N 3,3',5,5'-tetramethylbenzidine Chemical compound CC1=C(N)C(C)=CC(C=2C=C(C)C(N)=C(C)C=2)=C1 UAIUNKRWKOVEES-UHFFFAOYSA-N 0.000 description 1
- 208000030090 Acute Disease Diseases 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 208000003343 Antiphospholipid Syndrome Diseases 0.000 description 1
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 206010003658 Atrial Fibrillation Diseases 0.000 description 1
- 206010004146 Basal cell carcinoma Diseases 0.000 description 1
- 206010004593 Bile duct cancer Diseases 0.000 description 1
- 206010052384 Biliary cyst Diseases 0.000 description 1
- 206010005003 Bladder cancer Diseases 0.000 description 1
- 208000018084 Bone neoplasm Diseases 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 102000003930 C-Type Lectins Human genes 0.000 description 1
- 108090000342 C-Type Lectins Proteins 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 206010007559 Cardiac failure congestive Diseases 0.000 description 1
- 208000031229 Cardiomyopathies Diseases 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 206010008088 Cerebral artery embolism Diseases 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- 241000700114 Chinchillidae Species 0.000 description 1
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 208000000461 Esophageal Neoplasms Diseases 0.000 description 1
- 108010058861 Fibrin Fibrinogen Degradation Products Proteins 0.000 description 1
- 241000597000 Freesia Species 0.000 description 1
- 206010018338 Glioma Diseases 0.000 description 1
- 206010019280 Heart failures Diseases 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 1
- 206010023825 Laryngeal cancer Diseases 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- 108010052285 Membrane Proteins Proteins 0.000 description 1
- 102000018697 Membrane Proteins Human genes 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 208000021908 Myocardial disease Diseases 0.000 description 1
- 206010029164 Nephrotic syndrome Diseases 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- 206010030155 Oesophageal carcinoma Diseases 0.000 description 1
- 206010033799 Paralysis Diseases 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 206010050902 Postoperative thrombosis Diseases 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 101800004937 Protein C Proteins 0.000 description 1
- 102000017975 Protein C Human genes 0.000 description 1
- 102000029301 Protein S Human genes 0.000 description 1
- 108010066124 Protein S Proteins 0.000 description 1
- 229940096437 Protein S Drugs 0.000 description 1
- 206010037340 Pulmonary artery thrombosis Diseases 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 208000006265 Renal cell carcinoma Diseases 0.000 description 1
- 208000004756 Respiratory Insufficiency Diseases 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 101800001700 Saposin-D Proteins 0.000 description 1
- 206010040047 Sepsis Diseases 0.000 description 1
- 208000000453 Skin Neoplasms Diseases 0.000 description 1
- 206010068771 Soft tissue neoplasm Diseases 0.000 description 1
- 108010057517 Strep-avidin conjugated horseradish peroxidase Proteins 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 208000024770 Thyroid neoplasm Diseases 0.000 description 1
- 206010062129 Tongue neoplasm Diseases 0.000 description 1
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 208000002495 Uterine Neoplasms Diseases 0.000 description 1
- 206010046996 Varicose vein Diseases 0.000 description 1
- 208000024248 Vascular System injury Diseases 0.000 description 1
- 208000012339 Vascular injury Diseases 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 230000004520 agglutination Effects 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 230000000259 anti-tumor effect Effects 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 229940041181 antineoplastic drug Drugs 0.000 description 1
- 239000004019 antithrombin Substances 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 230000003143 atherosclerotic effect Effects 0.000 description 1
- 208000026900 bile duct neoplasm Diseases 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 230000023555 blood coagulation Effects 0.000 description 1
- 238000004820 blood count Methods 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 230000001269 cardiogenic effect Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 210000000038 chest Anatomy 0.000 description 1
- 208000011654 childhood malignant neoplasm Diseases 0.000 description 1
- 208000006990 cholangiocarcinoma Diseases 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000002405 diagnostic procedure Methods 0.000 description 1
- 230000010102 embolization Effects 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 201000004101 esophageal cancer Diseases 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- 239000000208 fibrin degradation product Substances 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 208000035474 group of disease Diseases 0.000 description 1
- 231100000226 haematotoxicity Toxicity 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 208000013210 hematogenous Diseases 0.000 description 1
- 108010036302 hemoglobin AS Proteins 0.000 description 1
- 230000002439 hemostatic effect Effects 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 206010073071 hepatocellular carcinoma Diseases 0.000 description 1
- 231100000844 hepatocellular carcinoma Toxicity 0.000 description 1
- 208000029824 high grade glioma Diseases 0.000 description 1
- 208000013403 hyperactivity Diseases 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 238000011221 initial treatment Methods 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 201000010849 intracranial embolism Diseases 0.000 description 1
- 206010023841 laryngeal neoplasm Diseases 0.000 description 1
- 239000004816 latex Substances 0.000 description 1
- 229920000126 latex Polymers 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 201000011614 malignant glioma Diseases 0.000 description 1
- 208000020984 malignant renal pelvis neoplasm Diseases 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000001617 migratory effect Effects 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 230000001400 myeloablative effect Effects 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 208000009928 nephrosis Diseases 0.000 description 1
- 231100001027 nephrosis Toxicity 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 229940127066 new oral anticoagluant drug Drugs 0.000 description 1
- 230000001453 nonthrombogenic effect Effects 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- 230000000414 obstructive effect Effects 0.000 description 1
- 201000008968 osteosarcoma Diseases 0.000 description 1
- 230000001991 pathophysiological effect Effects 0.000 description 1
- 230000007310 pathophysiology Effects 0.000 description 1
- 239000013610 patient sample Substances 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 210000003240 portal vein Anatomy 0.000 description 1
- 230000035935 pregnancy Effects 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 229960000856 protein c Drugs 0.000 description 1
- 210000001147 pulmonary artery Anatomy 0.000 description 1
- 230000002685 pulmonary effect Effects 0.000 description 1
- 238000003127 radioimmunoassay Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000029865 regulation of blood pressure Effects 0.000 description 1
- 208000015347 renal cell adenocarcinoma Diseases 0.000 description 1
- 201000007444 renal pelvis carcinoma Diseases 0.000 description 1
- 201000004193 respiratory failure Diseases 0.000 description 1
- 210000005245 right atrium Anatomy 0.000 description 1
- 210000005241 right ventricle Anatomy 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 238000003118 sandwich ELISA Methods 0.000 description 1
- 208000013223 septicemia Diseases 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 201000000849 skin cancer Diseases 0.000 description 1
- 239000003998 snake venom Substances 0.000 description 1
- 239000012064 sodium phosphate buffer Substances 0.000 description 1
- 238000011272 standard treatment Methods 0.000 description 1
- 208000014754 thrombocytosis disease Diseases 0.000 description 1
- 230000002885 thrombogenetic effect Effects 0.000 description 1
- 201000002510 thyroid cancer Diseases 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 201000006134 tongue cancer Diseases 0.000 description 1
- 238000013175 transesophageal echocardiography Methods 0.000 description 1
- 238000010967 transthoracic echocardiography Methods 0.000 description 1
- 201000005112 urinary bladder cancer Diseases 0.000 description 1
- 206010046766 uterine cancer Diseases 0.000 description 1
- 208000027185 varicose disease Diseases 0.000 description 1
- PJVWKTKQMONHTI-UHFFFAOYSA-N warfarin Chemical compound OC=1C2=CC=CC=C2OC(=O)C=1C(CC(=O)C)C1=CC=CC=C1 PJVWKTKQMONHTI-UHFFFAOYSA-N 0.000 description 1
- 229960005080 warfarin Drugs 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/574—Immunoassay; Biospecific binding assay; Materials therefor for cancer
- G01N33/57484—Immunoassay; Biospecific binding assay; Materials therefor for cancer involving compounds serving as markers for tumor, cancer, neoplasia, e.g. cellular determinants, receptors, heat shock/stress proteins, A-protein, oligosaccharides, metabolites
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6893—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to diseases not provided for elsewhere
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/705—Assays involving receptors, cell surface antigens or cell surface determinants
- G01N2333/7056—Selectin superfamily, e.g. LAM-1, GlyCAM, ELAM-1, PADGEM
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/22—Haematology
- G01N2800/226—Thrombotic disorders, i.e. thrombo-embolism irrespective of location/organ involved, e.g. renal vein thrombosis, venous thrombosis
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/86—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving blood coagulating time or factors, or their receptors
Definitions
- the present invention relates to a method for predicting cancer-related thrombosis using soluble CLEC2.
- Thrombus formed in blood vessels is considered to be a serious life-threatening factor among a wide range of human diseases, and it is known that the risk of thrombus formation increases in various diseases.
- the risk of thrombosis is high in acute diseases such as chronic obstructive pulmonary disease (COPD), infectious diseases, and septicemia, advanced cancer, pregnancy, nephrosis syndrome, inflammatory bowel disease, and myeloablative disease.
- COPD chronic obstructive pulmonary disease
- pancreatic cancer and brain tumors have a high risk of thrombosis.
- brain tumors were 48/1000 / year and pancreatic cancer was 58.9 / 1000 / year, compared with 13.6 / 1000 / year for cancer as a whole (non-).
- Patent Document 2 It is considered that the cause is that the coagulation system is activated by the cancer cells expressing tissue factor (TF) and constantly releasing TF-positive Microparticles (MPs) into the blood.
- TF tissue factor
- MPs TF-positive Microparticles
- VTE venous thromboembolism
- CAT cancer-associated thrombosis
- VTE Venous thromboembolism
- PE pulmonary arterial thrombosis
- VTE Venous thromboembolism
- DVT deep vein thrombosis
- PE pulmonary artery thrombosis
- DVT cancer-related CAT
- CAT varies depending on the type of cancer, and it has been reported that it is high in intra-abdominal and chest cancers, brain cancers, and cancers of unknown cause. In addition, the incidence of CAT is also high in cancer types with a high mortality rate, and it is considered that there is a correlation between the malignancy of cancer and the incidence of CAT.
- the period when the incidence of CAT is high is the first 3 months after being diagnosed with cancer, and special attention should be paid to the start time of cancer treatment. It has also been reported that CAT recurrence and bleeding are likely to occur in cancer patients after the onset of CAT during anticoagulant therapy, and careful management during treatment is required. Therefore, in the so-called perioperative period, risk management involving many medical personnel is required.
- Non-Patent Document 1 Venous and arterial thrombosis is often associated with cancer patients (Non-Patent Document 1), and clinically, the advanced stage, tumor volume, length of hospital stay, etc. increase the risk of thrombosis. It has been known.
- Trousseau syndrome is known as an example of hypercoagulation associated with cancer. Trousseau syndrome refers to "hypercoagulation associated with malignant tumors and associated migratory thrombophlebitis", but malignant tumors are often discovered for the first time after the onset of cerebral infarction. Therefore, in Japan, it is being understood as “systemic (particularly brain) embolism caused by DIC-related thrombosis associated with malignant tumors and nonbacterial thrombotic endocarditis (NBTE)".
- NBTE nonbacterial thrombotic endocarditis
- CAT shall include Trousseau syndrome.
- CAT malignant tumors that cause CAT are overwhelmingly adenocarcinomas such as lung cancer, pancreatic cancer, gastric cancer, and ovarian cancer (mutin-producing tumors).
- Head MRI often presents with multiple embolism, and about half have nonbacterial thromboendocarditis (NBTE), but the detection rate by transthoracic echocardiography is low, and transesophageal echocardiography It is said to be useful for diagnosis.
- NBTE nonbacterial thromboendocarditis
- Non-Patent Document 3 There is a description about the management of CAT in domestic and foreign guidelines, and anticoagulant therapy is to be performed as the initial treatment. There is no option. In the treatment of CAT, it is necessary to consider that thrombus is likely to be formed and bleeding is likely to occur at the same time, and it is necessary to consider that the influence of thrombophilia is present not only in veins but also in arterial thrombi.
- Podopranin is drawing attention as a thrombophilia predisposition for cancer patients.
- Podopranin is a membrane protein highly expressed on the surface of many cancer cells such as squamous cell carcinoma (lung, esophagus, cervix, etc.), mesothelioma, brain tumor, etc., and is involved in cancer infiltration.
- Kunida et al. Found that podoplanin promotes cancer metastasis through platelet aggregation (Non-Patent Document 4). It was also reported that podoplanin is highly expressed in brain tumors and osteosarcomas and has a high platelet aggregation ability.
- a novel anti-podopranin antibody induces a high antitumor effect through cytotoxic activity and suppresses metastasis, demonstrating that podoplanin is useful as a therapeutic target for cancer metastasis.
- C-type lectin-like receptor 2 (CLEC2), which is a receptor for podoplanin, was identified on platelets as a receptor for platelet-activated snake venom rhodocytin.
- CLEC2 C-type lectin-like receptor 2
- podoplanin The binding between CLEC2 and podoplanin has various pathophysiological roles, and its action of promoting hematogenous metastasis of tumors has been confirmed. Since CLEC2 is expressed almost specifically in platelets and megakaryocytes in humans, it is considered that thrombus is stabilized by homophyllically binding to platelet activation dependence in bloodstream (Non-Patent Document 5). ).
- the prognosis of a wide range of thrombosis is extremely poor, and an early and accurate diagnostic method is indispensable for implementing appropriate treatment that greatly improves the mortality rate. Therefore, even in cancer patients, in order to evaluate the risk of thrombosis, it is necessary to adequately manage the perioperative blood hypercoagulation state, and to work on prevention and early treatment.
- Patent Document 1 A method for diagnosing venous thromboembolism and stratifying risk, not only coagulation and hemostasis-related markers but also a method in which a plurality of markers such as blood pressure regulation, inflammation, myocardial disorder, and lung disorder are combined (Patent Document 1).
- Patent Document 2 A method combining measurement of D-dimer and measurement of coagulation factor activity using fibrin formation as an index (Patent Document 2) has been known, but none of them can be easily carried out, and thrombosis formation is accurate. Since it is difficult to say that the risk of the disease can be evaluated, there was no marker that could be used for risk evaluation and monitoring that could meet the needs in clinical practice.
- an object of the present invention is to develop a biomarker that better reflects thrombus formation in vivo and to provide a method that enables perioperative risk assessment of CAT in cancer patients.
- sCLEC2 concentration of soluble CLEC2
- CAT concentration of soluble CLEC2
- the present invention provides: [1] A method for assessing the risk of cancer-related thrombosis in the perioperative period of a cancer patient, which comprises a step of measuring the concentration of soluble CLEC2 present in blood collected from a cancer patient. [2] A method for assessing the risk of cancer-related thrombosis during the perioperative period of cancer patients. (1) A step of providing a sample derived from a patient who may have cancer-related thrombosis or a patient who has been diagnosed with cancer-related thrombosis. (2) A step of determining the soluble CLEC2 concentration in the sample, (3) A step of correlating the soluble CLEC2 concentration with the presence or absence of cancer-related thrombosis or the likelihood of outcome in the patient.
- the method of [1] including.
- [3] A method for assessing the risk of cancer-related thrombosis during the perioperative period of a cancer patient, which includes the soluble CLEC2 concentration and the presence or absence of cancer-related thrombosis in the patient, or the possibility of outcome.
- the method of [1] or [2], wherein the step of correlating is assessing whether the patient is at risk for cancer-related thrombosis based on changes in the soluble CLEC2 concentration.
- [4] A method for risk assessment of cancer-related thrombosis in the perioperative period of a cancer patient, which comprises using at least one coagulation and hemostasis-related marker in addition to the soluble CLEC2 concentration [1]. ] To [3].
- [5] A method for assessing the risk of cancer-related thrombosis in the perioperative period of a cancer patient, in which a value obtained by dividing the soluble CLEC-2 concentration by the platelet count is used instead of the soluble CLEC2 concentration.
- the method according to any one of [1] to [4].
- [6] A method for assessing the risk of cancer-related thrombosis in the perioperative period of a cancer patient, in which a sample derived from the cancer patient is provided over time from preoperative to 30 days after the operation, and the risk A method for predicting the effect of an antiplatelet agent by continuously monitoring the evaluation.
- the cancer is selected from the group consisting of pancreatic cancer, squamous cell carcinoma (lung, esophagus, cervix, etc.), mesothelioma, brain tumor, advanced cancer, and myeloproliferative disease, [1] to [ 6] Any method. [8] The method according to any one of [1] to [7], wherein the cancer-related thrombosis is Trousseau syndrome.
- the method of the present invention By measuring the sCLEC2 concentration present in the blood of a cancer patient, which is the method of the present invention, it is possible to perform an early, simple and accurate risk assessment of CAT. In addition, in the perioperative CAT onset prediction monitoring including the period after treatment such as surgery and chemotherapy in cancer patients, the prediction accuracy is improved compared to the conventional blood marker test and platelet aggregation test. Be expected.
- a method for risk assessment of CAT which comprises measuring (or determining) the soluble CLEC2 concentration (or the value obtained by dividing the soluble CLEC-2 concentration by the platelet count) in a sample.
- a method for assisting in CAT risk assessment which comprises measuring (or determining) the soluble CLEC2 concentration (or the soluble CLEC-2 concentration divided by the platelet count) in a sample.
- An in vitro risk assessment method for CAT which comprises measuring (or determining) the soluble CLEC2 concentration (or the value obtained by dividing the soluble CLEC-2 concentration by the platelet count) in a sample.
- Use of an antibody capable of detecting soluble CLEC2 concentration in the manufacture of a CAT risk assessment kit Included is a method of measuring (or determining) soluble CLEC2 concentration (or soluble CLEC-2 concentration divided by platelet count) in a sample to provide the information needed for CAT risk assessment. ..
- cancer refers to a group of diseases in which cells are infiltrated and spread from the site of origin, that is, the site of origin to other parts of the body, due to uncontrolled proliferation of cells, and cancer that develops from epithelial cells. It is not limited to cancers limited to. It is assumed to be suitable for cancers in which it is known that podopranin or its receptor CLEC2, which will be described later, is involved. In general, cancer is often classified according to the organ, tissue, shape, etc.
- basal cell carcinoma which is a cancer that develops from cells constituting the capsule, and myeloid proliferative disease.
- organ for example, brain tumor, tongue cancer, laryngeal cancer, thyroid cancer, esophageal cancer, gastric cancer, colon cancer, hepatocellular carcinoma, biliary cyst cancer, bile duct cancer, pancreatic cancer, lung cancer, middle splenoma, breast cancer , Ovarian cancer, cervical cancer, uterine body cancer (uterine cancer), renal cell cancer, renal pelvis cancer, prostate cancer, bladder cancer, skin cancer, bone and soft tissue tumor, leukemia, malignant lymphoma, childhood cancer, etc. , Not limited to this. It also includes those advanced cancers.
- organ for example, brain tumor, tongue cancer, laryngeal cancer, thyroid cancer, esophageal cancer, gastric cancer, colon cancer, hepatocellular carcinoma, biliary cyst cancer, bile duct cancer, pancreatic cancer, lung cancer, middle splenoma, breast cancer , Ovarian cancer, cervical cancer, uterine body cancer (uterine cancer), renal cell cancer, renal pelvis cancer, prostate
- cancer-related thrombosis is understood as a general term for cancer-related thrombosis.
- Thrombosis that can occur in cancer patients includes venous thromboembolism associated with the cancer itself, vascular disorders associated with chemotherapy, and the coagulation system, such as blood flow stagnation due to cancer, dehydration, lying down, and increased coagulation system due to cancer cells.
- Venous thromboembolism associated with cancer treatment history of venous thromboembolism, obesity, such as hyperactivity, vascular injury associated with catheters, stagnation, portal stagnation associated with hypertonic hypertension, and vascular disorders of the portal vein.
- Trousseau syndrome which is positioned as one aspect of this CAT, is not limited to the pathological condition in which a stroke symptom is exhibited due to hypercoagulation in the CAT, and thrombosis associated with DIC associated with a malignant tumor. And systemic embolism due to nonbacterial thrombotic endocarditis (NBTE).
- NBTE nonbacterial thrombotic endocarditis
- the "perioperative period” means a series of periods including before and after the surgery from the outpatient department where the surgery is decided to hospitalization, anesthesia / surgery, postoperative recovery, discharge / rehabilitation.
- CLEC2 is a platelet activation receptor belonging to the C-type lectin family, which is usually present in the platelet membrane, but is released into the blood with the activation of platelets. ..
- soluble CLEC2 sCLEC2
- sCLEC2 refers to a molecule derived from CLEC2 or CLEC2 that is released from such platelets and detected in blood (in buffer, if incubated in buffer). Means.
- SCLEC2 includes a protein having a molecular weight of about 40 kDa, a protein having a molecular weight of about 32 kDa, and a protein having a molecular weight of about 25 kDa in SDS polyacrylamide gel electrophoresis (SDS-PAGE) under reducing conditions. It is presumed that the protein having a molecular weight of about 40 kDa and the protein having a molecular weight of about 32 kDa are present on the surface of the platelet membrane and are released in a state of being contained in the microparticles produced by the activation of platelets. It is considered that sugar chains are added to these.
- a protein having a molecular weight of about 25 kDa is cleaved by a protease with activation of platelets and released from platelets.
- the amount of sCLEC2 as described above is measured.
- sCLEC2 may collectively detect a protein having a molecular weight of about 40 kDa, a protein having a molecular weight of about 32 kDa, and a protein having a molecular weight of about 25 kDa, or may detect only a protein having a molecular weight of about 25 kDa.
- the sCLEC2 concentration used in the present invention only the sCLEC2 concentration may be used, or the value obtained by dividing the sCLEC2 concentration by the platelet count may be used.
- the sCLEC2 concentration is interpreted to include both the case where the sCLEC2 concentration is used and the case where the sCLEC2 concentration is divided by the platelet count.
- the sample used for the measurement is preferably derived from humans, but a sample derived from an animal other than humans may be used in order to grasp the pathological condition of the experimental animal.
- the experimental animal is not particularly limited, and examples thereof include guinea pigs, rats, mice, and chinchillas.
- thrombotic hemostatic diseases As used herein, “hemostatic” means that platelets and coagulation factors are conjugated to effectively and appropriately stop bleeding or bleeding.
- thrombogenic hemostatic disease includes, but is not limited to, conditions and diseases including, but not limited to, excessive bleeding and abnormal blood coagulation. In particular, it can be suitably used in risk prediction of venous thromboembolism (VTE) and cancer-related thrombosis (CAT), which is a VTE associated with cancer.
- VTE venous thromboembolism
- CAT cancer-related thrombosis
- the sCLEC2 concentration value is higher than that of a healthy person or a non-thrombogenic hemostatic disease group, it can be said that there is a high possibility or risk of suffering from CAT thrombotic hemostatic disease.
- the sCLEC2 concentration can be compared preoperatively and postoperatively and used as a risk prediction for thrombosis.
- sCLEC2 concentration is measured in patients with pancreatic cancer and brain tumor, and if the ratio becomes high, it can be judged that platelet activation in vivo has occurred, and antiplatelet drugs such as aspirin are used as primary drugs. It is considered possible to use it as a preventive measure. Furthermore, sCLEC2 concentration measurement will be performed in patients taking antiplatelet drugs such as aspirin and clovidogrel, and if the value is high, the dose of antiplatelet drug should be increased, or change to a different type of antiplatelet drug or additional administration should be considered. It is also possible.
- the method for detecting the presence of sCLEC2 is not particularly limited, but an immunological method using an antibody that recognizes sCLEC2 (hereinafter, this may be referred to as "anti-sCLEC2 antibody”) is preferable.
- immunological methods for detecting proteins include immunoassay using labeled antibodies such as enzyme immunoassay (ELISA method), chemoluminescence immunoassay, fluorescent antibody method, radioimmunoassay, and immunochromatography. Any commonly known method such as a measuring method, a Western blotting method, a latex agglutination method, or an immunoturbidimetric method can be used, but among these, from the viewpoint of simplicity of operation and measurement accuracy.
- An immunoassay method using a labeled antibody is preferably used. For intraoperative diagnosis, it is desired that a rapid result can be obtained, and therefore chemiluminescent immunoassay, immunochromatography, or the like is particularly preferably used.
- a sample is collected from a target subject (particularly a patient) using a blood collection tube for plasma blood collection.
- a blood collection tube for plasma blood collection Considering the measurement of platelet count, EDTA-containing blood collection tube is preferable, but heparin and citric acid-containing blood collection tubes can also be used, and those skilled in the art can appropriately select and use an appropriate blood collection tube from these. can do.
- Samples for measuring plasma sCLEC2 concentration and measuring platelet count may be obtained from one blood collection tube, but if blood is collected at the same time, different blood collection tubes may be used.
- the plasma sCLEC2 concentration is measured using, for example, plasma centrifuged at 2000 g for about 20 minutes, but the centrifugation conditions are not limited to this, and a measurement system using whole blood may be used.
- measurement of plasma sCLEC2 concentration will be described as an example, but the present invention is not limited thereto.
- whole blood containing an anticoagulant such as EDTA is used.
- the sCLEC2 concentration in the cancer patient-derived sample and the sCLEC2 concentration in the healthy person-derived sample may be appropriately set and used from the comparison of sCLEC2 concentration, or the sCLEC2 concentration can be compared from the time-dependent records of sCLEC2 concentration, such as comparison between preoperative and postoperative and comparison between the first day and several days after the operation in the same patient.
- a CAT risk assessment may be performed when significant fluctuations in CAT are detected.
- the platelet count is usually measured using an automatic blood cell counter (blood cell counter), but a blood cell counter and a microscope. It is also possible to count using.
- the sCLEC2 concentration in plasma is represented by, for example, pg / mL
- the blood platelet count is represented by, for example, 1000 cells / mm3
- the sCLEC2 concentration / platelet count is calculated.
- Any unit such as ng / mL or ng / L may be used for the concentration of sCLEC2 used here, and any unit such as 10000 platelets / mm 3 can be used for the platelet count, but a unified unit is used for comparison. Should be used.
- the sCLEC2 concentration / platelet count can take different values, but they are essentially the same concept.
- the ratio is calculated using the measured value obtained from the clinical laboratory device that measures the sCLEC2 concentration and the measured value that comes out from the blood calculator that measures the platelet count.
- This calculation is suitable for daily medical care to be calculated automatically on a hospital inspection system, a hospital system, or a system such as an electronic medical record connected to both measuring instruments, but the data of the two measuring instruments is used.
- a connecting system may be constructed, or a machine capable of simultaneously measuring the sCLEC2 concentration and the platelet count can be constructed. Alternatively, both data may be used for manual calculation.
- the sCLEC2 concentration in plasma or the sCLEC2 concentration / platelet count and the correlation between the degree of platelet activation and various diseases are, for example, a determination threshold or original data for calculating a determination threshold. It may be used as statistical processing data or the like.
- SCLEC2 is released into the blood with the activation of platelets.
- Existing platelet activation markers such as PF4 and ⁇ TG, have been problematic in that granules are stimulated by physical pressure from blood sampling to cause non-specific release, whereas sCLEC2 induces platelet activation. It is a signal transduction-dependent release mechanism that can be a marker that more accurately reflects the activation of platelets in vivo.
- CLEC2 is a specific marker with few false positives because its expression is almost limited in the platelet / megakaryocyte system in humans. Therefore, by measuring sCLEC2, it is possible to diagnose the activated state of platelets at an early stage, and it can be used as a method for predicting the risk of thrombosis.
- Plasma sCLEC2 concentration tends to be high in individuals with high platelets, and plasma sCLEC2 concentration tends to be low in individuals with low platelets.
- the value obtained by dividing the plasma sCLEC2 concentration by the blood platelet count may be used, taking advantage of the fact that the sCLEC2 concentration has a positive correlation with the blood platelet count.
- the plasma sCLEC2 concentration is represented by pg / mL (A)
- the blood platelet count is represented by 1000 cells / mm 3 (B)
- the number obtained by dividing A by B is used as an index of platelet activation. can do.
- plasma sCLEC2 concentration is measured and used for risk prediction by observing the change in the concentration. It is possible. For example, by appropriately setting the sampling period according to the background information for each patient, it is possible to obtain a more detailed profile, observe the condition, and evaluate the risk. It is used not only to predict the risk of CAT for patients who are observed to increase significantly from preoperatively, but also to monitor the post-drug status of patients diagnosed with CAT. can do.
- CAT has a high recurrence rate, and it is thought that there are many cases in which long-term anticoagulant therapy is required.
- anticoagulant therapy for CAT may increase the risk of bleeding, and anticancer drugs may cause hematological toxicity that reduces leukocytes (especially neutrophils), red blood cells, and platelets.
- leukocytes especially neutrophils
- red blood cells especially platelets.
- anticoagulant therapy policy it is difficult to determine the duration of anticoagulant therapy for CAT because it must be determined in consideration of bleeding risk and prognosis, no clear criteria have been given for anticoagulant therapy policy. Therefore, by using the present invention to evaluate the risk of CAT in a patient, it may be an aid for appropriately determining a treatment policy.
- the frequency of concentration measurement used for monitoring may be appropriately set according to the background information including the treatment history of the individual patient and the treatment policy. In that case, risk assessment is possible by collecting samples at regular intervals from preoperative to 30 days after surgery, but it is assumed that the situation will differ depending on the patient's background. Therefore, for example, the monitoring period and timing are appropriately set, for example, by observing the occurrence of CAT regularly during the 7th to 10th days after surgery, which requires special attention. Can be carried out.
- the present invention for the purpose of assisting the risk assessment based on the sCLEC2 concentration, it may be used in combination with a biomarker currently reported to be useful as a risk assessment of CAT.
- CAT risk assessment can be performed more accurately, which is preferable.
- D-dimer or the like may be used for postoperative monitoring as in the present invention.
- the sCLEC2 concentration fluctuates in the case diagnosed as CAT, the D-dimer does not fluctuate at all and the risk of CAT cannot be evaluated. It was found by the examination of the present inventors that there is a case.
- thrombus This may depend on the mechanism by which the thrombus is formed. That is, by monitoring the fibrin-based thrombus formation with D-dimer and the platelet-based thrombus formation with sCLEC2 over time, there is a possibility that the risk assessment of CAT can be performed more accurately. That is, platelet-dominated thrombi can be predicted.
- the present invention may be used in combination with other findings such as image evaluation.
- diagnosis is performed by performing image diagnosis on a patient suspected of having CAT, and it is expected that the accuracy will be improved by using it in combination with this information, which is preferable. ..
- treatment of thrombosis includes aspirin in the case of thrombosis in an artery where coagulation progresses due to activation and aggregation of platelets such as myocardial infarction, atherosclerotic cerebral infarction, and obstructive arteriosclerosis.
- platelets such as myocardial infarction, atherosclerotic cerebral infarction, and obstructive arteriosclerosis.
- thrombosis such as deep vein thrombosis, atrial fibrillation, cardiogenic cerebral embolism, and pulmonary embolism, in which the coagulation system is activated in the veins and atria and coagulation progresses, warfarin, DOAC, etc.
- a treatment method that administers an anticoagulant is selected.
- CAT which is a cancer-related thrombosis
- an anticoagulant is currently used as a standard treatment method.
- the need for postoperative care and treatment means can be evaluated and estimated in a reliable manner. For example, it is possible to quickly determine the therapeutic effect and predict the risk with reliable accuracy, and it is possible to select a drug having a lower risk of bleeding, so that a great benefit can be obtained from the method of the present invention. ..
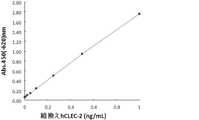
- Example 1 Measurement of sCLEC2 in human plasma >> The sCLEC2 concentration in plasma was measured according to Example 6 of Japanese Patent No. 4961595.
- a sandwich ELISA system was constructed using mouse anti-human sCLEC2 antibody. That is, the 1-11D5 antibody (F (ab) ' 2 ) purified with 0.05 mol / L carbonate buffer (pH 9.5) was diluted to 10 ⁇ g / mL, and 100 ⁇ L / well was added to the immunoplate (Maxisorp; NUNC). did.
- the prepared biotin-labeled 3-11E6 antibody (F (ab) ' 2 -biotin) was added to 1.0 ⁇ g / mL in 10% SuperBlock, 0.1% sodium octanate, 0.14 mol / L sodium chloride / PB. And 100 ⁇ L / well was added to each well. After reacting at 37 ° C. for 1 hour, the cells were washed 3 times in the same manner.
- AMDEX streptavidin-conjugated horseradish peroxidase (GE Healthcare) was then diluted with 10% SuperBlock, 0.1% sodium octanate, 0.14 mol / L sodium chloride / PB and into each well. 100 ⁇ L / well was added. After reacting at 37 ° C. for 1 hour, the mixture was washed 5 times in the same manner, and 100 ⁇ L / well of each well was added with a 3,3', 5,5'-tetramethylbenzidine (TMB) solution. After reacting at room temperature for about 20 minutes, the reaction was stopped with a 1 mol / L sulfuric acid solution.
- TMB 3,3', 5,5'-tetramethylbenzidine
- FIG. 1 shows a standard curve prepared by using the hsCLEC2 protein as a standard product.
- Example 2 Measurement of sCLEC2 in plasma sample of pancreatic cancer patient >> 14 cases of pancreatic cancer patient plasma and 10 cases of healthy subjects purchased from BiolVT (Westbury, NY) were diluted with buffer and measured by the method of Example 1. The results of the obtained pancreatic cancer patients are shown in Table 1. In addition, the average value ⁇ SE of sCLEC2 concentration in healthy subjects and pancreatic cancer patients was calculated and shown in FIG. The sCLEC2 concentration was significantly (p ⁇ 0.05) higher than that of healthy subjects. From this, it was clarified that platelets are activated by the influence of cancer in patients with pancreatic cancer. Next, when pancreatic cancer patients were classified according to the presence or absence of blood abnormalities, as shown in FIG.
- sCLEC2 was higher in patients with blood abnormalities than in patients without blood abnormalities.
- 1382.2 pg / mL which is about 8 times higher than the average value of healthy subjects, was found in patients diagnosed with an abnormal coagulation profile, and it was revealed that the value was high in patients at high risk of thrombosis.
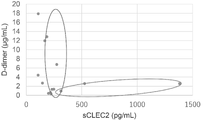
- VTE monitoring generally uses D-dimer in addition to image evaluation. Therefore, D-dimer of 14 cases of pancreatic cancer patient plasma purchased from BiolVT was measured with LPIA Genesis D-dimer reagent (LSI Rulece) and compared with the sCLEC2 concentration measured in Example 2 (FIG. 4). As a result, two groups were observed: a group of patients in which D-dimer was elevated relative to sCLEC2 and a group of patients in which sCLEC2 was elevated relative to D-dimer. Thrombosis is roughly classified into venous thrombosis and arterial thrombosis.
- Venous thrombosis is considered to be a fibrin-based thrombosis
- arterial thrombosis is considered to be mainly composed of platelets.
- D-dimer a fibrin degradation product
- fibrin-based venous thrombosis is commonly used as a biomarker for fibrin-based venous thrombosis, but there is no commonly used biomarker for arterial thrombosis such as platelet-based Truseau syndrome.
- the fact that a group in which D-dimer did not rise and sCLEC2 increased was observed in this example means that sCLEC2 captures the pathophysiology of platelet-based arterial thrombosis, which is unknown by the conventional D-dimer test. It has been shown that sCLEC2 is useful for the examination of Trousseau syndrome.
- Example 3 Measurement of sCLEC2 in plasma sample of brain tumor patient
- Plasma was collected from a sample obtained by general blood sampling 7 to 10 days after surgery for a brain tumor centered on malignant glioma admitted to neurosurgery and a healthy person with consent, and sCLEC2 was collected by the method of Example 1. The concentration was measured. The results of the obtained samples are shown in Tables 2 and 3.
- the sCLEC2 concentration 7 to 10 days after the operation tended to be higher in DVT / PE cases than in cases without DVT / PE (p 0.239).
- Table 4 shows the results of the mean value, standard deviation, and median value in the cases with DVT / PE and the cases without DVT / PE.
- DVT was screened based on the D-dimer value and clinical symptoms, and was identified 10 days after surgery on average. This indicates that platelets may be activated in patients who developed DVT / PE during the perioperative period of brain tumors.
- DVT monitoring generally uses a D-dimer in addition to image evaluation. Therefore, the D-dimer concentration, sCLEC2 concentration, and C2PAC value of patients (glioblastoma, 71 years old, female) who were able to collect blood over time were evaluated (FIG. 6). This case was identified as DVT 7 days after surgery and anticoagulant therapy was started. It was considered that the sCLEC2 and C2PAC levels were maximum on the 7th day when they were identified as DVT, and then gradually decreased due to the effect of anticoagulant therapy. On the other hand, D-dimer showed a relatively high value 14 days after the start of anticoagulant therapy in addition to the identification of DVT, but no significant change was observed. The sCLEC2 and C2PAC levels responded more sensitively to treatment, and in this case, the sCLEC2 and C2PAC levels were estimated to be the most useful for monitoring the therapeutic effect for monitoring anticoagulant therapy.
- the method for predicting thrombosis risk is applied by measuring blood sCLEC2 of the present invention, the risk of thrombosis in a patient can be predicted before thrombosis occurs, and appropriate treatment can be started promptly. It becomes. Therefore, the thrombosis risk prediction method of the present invention can be applied to a wide range of fields such as medicine and biology, and is particularly useful in the field of clinical examination.
Abstract
Description
更に、癌患者のsCLEC2濃度を経時的に測定してその変動を観察することによって、早期かつ簡便で正確なCATのリスク評価を行うことができる方法を確立することができた。数十名の患者サンプルを用いて本法を検証し、膵臓癌及び脳腫瘍患者CATリスク予測及びモニタリングに有用であることを示した。
[1]癌患者から採取した血液中に存在する、可溶型CLEC2濃度を測定する工程を含む、癌患者周術期における癌関連血栓症のリスク評価を行う方法。
[2]癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、
(1)癌関連血栓症を有する可能性のある患者、または癌関連血栓症と診断された患者に由来する試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記可溶型CLEC2濃度と、前記患者における癌関連血栓症の存在もしくは非存在、または転帰の可能性とを相関させる工程、
を含む、[1]の方法。
[3]癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記可溶型CLEC2濃度と、前記患者における癌関連血栓症の存在もしくは非存在、または転帰の可能性とを相関させる工程が、前記可溶型CLEC2濃度の変化に基づいて、癌関連血栓症のリスクを前記患者が有するかどうかを評価することを含む、[1]又は[2]の方法。
[4]癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記可溶型CLEC2濃度に加えて、少なくとも1つの凝固および止血関連マーカーを使用することを含む、[1]~[3]のいずれかの方法。
[5]癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記可溶型CLEC2濃度に代えて、可溶型CLEC-2濃度を血小板数で除した値を使用することを特徴とする、[1]~[4]のいずれかの方法。
[6]癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記癌患者に由来する試料の提供を、術前から術後30日までの間経時的に行い、リスク評価を継続してモニタリングすることにより、抗血小板剤の効果判定の予測方法。
[7]前記癌が、膵臓癌、扁平上皮癌(肺、食道、子宮頸部等)、中皮腫、脳腫瘍、進行癌、骨髄増殖性疾患からなる群から選択される、[1]~[6]のいずれかの方法。
[8]前記癌関連血栓症が、トルーソー症候群である、[1]~[7]のいずれかの方法。
例えば、本発明には、
試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値)を測定(又は決定)することを特徴とする、CATのリスク評価を行う方法、
試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値)を測定(又は決定)することを特徴とする、CATのリスク評価を補助する方法、
CATのリスク評価のために、試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値)を測定(又は決定)する方法、
試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値)を測定(又は決定)することを特徴とする、CATのin vitroリスク評価方法、
可溶型CLEC2濃度を検出できる抗体の、CATのリスク評価用キットの製造における使用、
CATのリスク評価に必要な情報を提供するために、試料中の可溶型CLEC2濃度(又は可溶型CLEC-2濃度を血小板数で除した値)を測定(又は決定)する方法
が含まれる。
特許第4961595号明細書の実施例6に従って、血漿中のsCLEC2濃度を測定した。
マウス抗ヒトsCLEC2抗体を用いてサンドイッチELISA系を構築した。すなわち、0.05mol/L炭酸緩衝液(pH9.5)で精製した1-11D5抗体(F(ab)’2)を10μg/mLに希釈し、イムノプレート(Maxisorp; NUNC)に100μL/ウェル添加した。4℃で一晩反応後、0.05%Tween-20を含むリン酸緩衝生理食塩水(PBS)で3回洗浄し、1%ウシ血清アルブミン(BSA)を含むPBSを各ウェルに200μL添加しブロッキングした。次にスタンダードとして用いるヒトsCLEC2(hsCLEC2)蛋白質を10% SuperBlock(Thermo Fisher Scientific)、0.1%オクタン酸ナトリウム、0.14mol/L塩化ナトリウム/リン酸緩衝液(PB)を用いて標準品希釈系列を調製した。ヒト血漿は同バッファーで5倍以上に希釈した。それぞれ100μL/ウェルで添加し、37℃で1時間半反応させた後、同様に3回洗浄した。次に調製したビオチン標識した3-11E6抗体(F(ab)’2-ビオチン)を10%SuperBlock、0.1%オクタン酸ナトリウム、0.14mol/L塩化ナトリウム/PBにて1.0μg/mLに希釈し、各ウェルに100μL/ウェル添加した。37℃で1時間、反応させた後、同様の方法で3回洗浄した。次いで、AMDEXストレプトアビジン標識ホースラディッシュペルオキシダーゼ(AMDEX streptavidin-conjugated horseradish peroxidase; GE Healthcare)を10%SuperBlock、0.1%オクタン酸ナトリウム、0.14mol/L塩化ナトリウム/PBにて希釈し、各ウェルに100μL/ウェル添加した。37℃で1時間反応させた後、同様の方法で5回洗浄し、3,3’,5,5’-テトラメチルベンジジン(TMB)溶液を各ウェル100μL/ウェル添加した。室温で約20分間反応後、1mol/L硫酸溶液で反応を停止した。プレート分光光度計(BIO-TEK INSTRUMENTS社/EL312e)で450nm(-620nm)の吸光度を測定した。図1にhsCLEC2蛋白質を標準品として用いて作成した標準曲線を示した。
BiolVT社(Westbury, NY)より購入した膵臓癌患者血漿14例及び健常人10例をバッファーで希釈し、実施例1の方法により測定した。得られた膵臓癌患者の結果を表1に示した。また、健常人と膵臓癌患者のsCLEC2濃度の平均値±SEを算出し図2に示した。sCLEC2濃度は健常人に比較して有意(p<0.05)に上昇していた。このことから、膵臓癌患者では癌の影響により血小板が活性化していることが明らかになった。次に膵臓癌患者を血液異常の有無により分類したところ、図3に示すように血液異常がある患者で、血液異常がない患者に比較して、sCLEC2が高値を示した。その中でも異常凝固プロファイルと診断された患者では1382.2pg/mLと健常人平均値の約8倍高値であり、血栓症リスクの高い患者で高値を示すことが明らかになった。
脳神経外科に入院した悪性神経膠腫を中心とする脳腫瘍の術後7~10日の一般採血で得られた検体及び同意の得られた健常人より血漿を採血し、実施例1の方法によりsCLEC2濃度を測定した。得られた検体の結果を表2、表3に示した。図5にDVT/PE合併症例(n=10)、DVT/PE非合併症例(n=48)、及び健常人(n=15)のsCLEC2濃度を示した。術後7~10日後でのsCLEC2濃度はDVT/PE症例でDVT/PE非合併症例と比較して高値傾向を示した(p=0.239)。また、DVT/PE合併症例とDVT/PE非合併症例における平均値、標準偏差、中央値の結果を表4に示した。なお、DVTはDダイマー値と臨床症状でスクリーニングを行い、術後平均10日で同定されていた。このことから、脳腫瘍の周術期にDVT/PEを発症した患者では血小板が活性化している可能性が示された。また、CLEC2は血小板に発現し、血小板数の影響を受ける可能性があることから、sCLEC2濃度を血小板数で割った値(sCLEC2濃度pg/mLを血小板数1000個/μLで除したもの。以下、C2PACと表記する場合がある)により両群を比較したところ、DVT/PE非合併症例と比べて、DVT/PE合併症例で有意な上昇が示された(p=0.032、表2、表3、図6)。
Claims (8)
- 癌患者から採取した血液中に存在する、可溶型CLEC2濃度を測定する工程を含む、癌患者周術期における癌関連血栓症のリスク評価を行う方法。
- 癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、
(1)癌関連血栓症を有する可能性のある患者、または癌関連血栓症と診断された患者に由来する試料を提供する工程、
(2)前記試料中の可溶型CLEC2濃度を決定する工程、
(3)前記可溶型CLEC2濃度と、前記患者における癌関連血栓症の存在もしくは非存在、または転帰の可能性とを相関させる工程、
を含む、請求項1に記載の方法。 - 癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記可溶型CLEC2濃度と、前記患者における癌関連血栓症の存在もしくは非存在、または転帰の可能性とを相関させる工程が、前記可溶型CLEC2濃度の変化に基づいて、癌関連血栓症のリスクを前記患者が有するかどうかを評価することを含む、請求項1又は2に記載の方法。
- 癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記可溶型CLEC2濃度に加えて、少なくとも1つの凝固および止血関連マーカーを使用することを含む、請求項1~3のいずれか一項に記載の方法。
- 癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記可溶型CLEC2濃度に代えて、可溶型CLEC-2濃度を血小板数で除した値を使用することを特徴とする、請求項1~4のいずれか一項に記載の方法。
- 癌患者周術期における癌関連血栓症のリスク評価を行う方法であって、前記癌患者に由来する試料の提供を、術前から術後30日までの間経時的に行い、リスク評価を継続してモニタリングすることにより、抗血小板剤の効果判定の予測方法。
- 前記癌が、膵臓癌、扁平上皮癌(肺、食道、子宮頸部等)、中皮腫、脳腫瘍、進行癌、骨髄増殖性疾患からなる群から選択される、請求項1~6のいずれか一項に記載の方法。
- 前記癌関連血栓症が、トルーソー症候群である、請求項1~7のいずれか一項に記載の方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202280009906.2A CN116761895A (zh) | 2021-01-13 | 2022-01-13 | 使用了可溶型clec2的癌症患者的血栓症风险的预测方法 |
| JP2022575626A JPWO2022154045A1 (ja) | 2021-01-13 | 2022-01-13 | |
| KR1020237025829A KR20230131222A (ko) | 2021-01-13 | 2022-01-13 | 가용형 clec2를 이용한 암 환자에 있어서의 혈전증리스크의 예측 방법 |
| EP22739450.9A EP4279606A1 (en) | 2021-01-13 | 2022-01-13 | Method for predicting risk for thrombosis in cancer patient using soluble clec2 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021003671 | 2021-01-13 | ||
| JP2021-003671 | 2021-01-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022154045A1 true WO2022154045A1 (ja) | 2022-07-21 |
Family
ID=82448474
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/000891 WO2022154045A1 (ja) | 2021-01-13 | 2022-01-13 | 可溶型clec2を用いた癌患者における血栓症リスクの予測方法 |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP4279606A1 (ja) |
| JP (1) | JPWO2022154045A1 (ja) |
| KR (1) | KR20230131222A (ja) |
| CN (1) | CN116761895A (ja) |
| WO (1) | WO2022154045A1 (ja) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008539694A (ja) * | 2005-04-25 | 2008-11-20 | 国立大学法人山梨大学 | Clec−2シグナル伝達により止血疾患を治療するための組成物および方法 |
| JP2008544224A (ja) | 2005-06-09 | 2008-12-04 | バイオサイト インコーポレイテッド | 静脈血栓塞栓症の診断のための方法および組成物 |
| WO2012128082A1 (ja) * | 2011-03-22 | 2012-09-27 | 公益財団法人がん研究会 | マウス抗Aggrusモノクローナル抗体 |
| JP2014070942A (ja) * | 2012-09-28 | 2014-04-21 | Mitsubishi Chemical Medience Corp | 可溶型clec−2に基づく血小板活性化測定方法 |
| JP2019507345A (ja) | 2016-02-18 | 2019-03-14 | ダイアグノスティカ スターゴ | 静脈血栓塞栓症に対して特異的なdダイマーをアッセイするための方法、ならびに肺塞栓症および深部静脈血栓症を診断するためのその使用 |
| WO2021172493A1 (ja) * | 2020-02-28 | 2021-09-02 | 株式会社Lsiメディエンス | 可溶型clec-2と血小板数に基づく血小板活性化測定方法 |
-
2022
- 2022-01-13 KR KR1020237025829A patent/KR20230131222A/ko unknown
- 2022-01-13 CN CN202280009906.2A patent/CN116761895A/zh active Pending
- 2022-01-13 WO PCT/JP2022/000891 patent/WO2022154045A1/ja active Application Filing
- 2022-01-13 JP JP2022575626A patent/JPWO2022154045A1/ja active Pending
- 2022-01-13 EP EP22739450.9A patent/EP4279606A1/en active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008539694A (ja) * | 2005-04-25 | 2008-11-20 | 国立大学法人山梨大学 | Clec−2シグナル伝達により止血疾患を治療するための組成物および方法 |
| JP4961595B2 (ja) | 2005-04-25 | 2012-06-27 | 国立大学法人山梨大学 | Clec−2シグナル伝達により止血疾患を治療するための組成物および方法 |
| JP2008544224A (ja) | 2005-06-09 | 2008-12-04 | バイオサイト インコーポレイテッド | 静脈血栓塞栓症の診断のための方法および組成物 |
| WO2012128082A1 (ja) * | 2011-03-22 | 2012-09-27 | 公益財団法人がん研究会 | マウス抗Aggrusモノクローナル抗体 |
| JP2014070942A (ja) * | 2012-09-28 | 2014-04-21 | Mitsubishi Chemical Medience Corp | 可溶型clec−2に基づく血小板活性化測定方法 |
| JP2019507345A (ja) | 2016-02-18 | 2019-03-14 | ダイアグノスティカ スターゴ | 静脈血栓塞栓症に対して特異的なdダイマーをアッセイするための方法、ならびに肺塞栓症および深部静脈血栓症を診断するためのその使用 |
| WO2021172493A1 (ja) * | 2020-02-28 | 2021-09-02 | 株式会社Lsiメディエンス | 可溶型clec-2と血小板数に基づく血小板活性化測定方法 |
Non-Patent Citations (11)
| Title |
|---|
| FREESIA HORSTED ET AL., PLOS MED, vol. 9, no. 7, July 2012 (2012-07-01), pages e1001275 |
| KATSUE SUZUKI-INOUE: "Roles of the CLEC-2-podoplanin interaction in tumor progression", PLATELETS, vol. 29, no. 8, 1 January 2018 (2018-01-01), GB , pages 786 - 792, XP009538258, ISSN: 0953-7104, DOI: 10.1080/09537104.2018.1478401 * |
| KUNITA A ET AL., AM J PATHOL., vol. 170, 2007, pages 1337 - 1347 |
| LYMAN GH ET AL., J CLIN ONCOL, vol. 31, no. 17, 2013, pages 2189 - 2204 |
| MAKOTO SANO, RYOTA TAKAHASHI, HIDEAKI IJICHI, HIROSHI MIYABAYASHI, KAZUYOSHI ISHIGAKI, SUGURU MIZUNO, YOSUKE NAKAI, KEISUKE TATEIS: "PCA1: Elevated plasma sVCAM-1 and ANP are risk predictors for pancreatic cancer-related thrombosis/thromboembolism", PANCREAS, vol. 35, no. 3, 17 July 2020 (2020-07-17), JP , pages A266, XP009538271, ISSN: 0913-0071, DOI: 10.2958/suizo.35.A1 * |
| MIR SEYED NAZARI PEGAH, RIEDL JULIA, PABINGER INGRID, AY CIHAN: "The role of podoplanin in cancer-associated thrombosis", THROMBOSIS RESEARCH, ELSEVIER, AMSTERDAM, NL, vol. 164, 1 April 2018 (2018-04-01), AMSTERDAM, NL , pages S34 - S39, XP055951090, ISSN: 0049-3848, DOI: 10.1016/j.thromres.2018.01.020 * |
| OSADA MAKOTO: "Examination for clinical applications and practical use of blood sCLEC-2 measurement method", GRANT-IN-AID FOR SCIENTIFIC RESEARCH, FINAL RESEARCH REPORT., 1 January 2016 (2016-01-01), XP055951088, Retrieved from the Internet <URL:https://kaken.nii.ac.jp/ja/file/KAKENHI-PROJECT-25460677/25460677seika.pdf> * |
| SHIRAI, T.; INOUE, O; HIRAYAMA, K.; ENDO, H.; SATO-UTIDA, H.; FUJII, H.; SUZUKI-INOUE, K.; OZAKI, Y.: "CLEC-2 facilitates hematogeneous tumor metastasis and in vitro tumor growth, but not in vivo tumor growth", JOURNAL OF THROMBOSIS AND HAEMOSTASIS, BLACKWELL PUBLISHING, OXFORD, vol. 13, no. Suppl. 2, 31 May 2015 (2015-05-31), GB , pages 226, XP009538357, ISSN: 1538-7933 * |
| SHIRAI, TOSHIAKI: "Platelet CLEC-2 promotes hematogenous metastasis and thromboinflammation in tumor-bearing mice", JAPANESE JOURNAL OF THROMBOSIS AND HEMOSTASIS, NIHON KESSEN SHIKETSU GAKKAI , TOKYO, vol. 31, no. 4, 1 January 2020 (2020-01-01), JP , pages 452 - 459, XP009538356, ISSN: 0915-7441, DOI: 10.2491/jjsth.31.452 * |
| SUZUKI-INOUE KATSUE: "Platelets and cancer-associated thrombosis: focusing on the platelet activation receptor CLEC-2 and podoplanin", BLOOD, vol. 134, no. 22, 28 November 2019 (2019-11-28), pages 1912 - 1918, XP055951087, DOI: 10.1182/blood.2019001388 * |
| TROUSSEAU A: "Phlegmasia alba dolens", CLINIQUE MEDICALE DE 1'HOTEL DIEU DE PARIS, vol. 3, pages 654 - 712 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4279606A1 (en) | 2023-11-22 |
| KR20230131222A (ko) | 2023-09-12 |
| JPWO2022154045A1 (ja) | 2022-07-21 |
| CN116761895A (zh) | 2023-09-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Blann et al. | Vascular endothelial growth factor and its receptor, Flt-1, in the plasma of patients with coronary or peripheral atherosclerosis, or Type II diabetes | |
| Shorr et al. | D-dimer assay predicts mortality in critically ill patients without disseminated intravascular coagulation or venous thromboembolic disease | |
| Tian et al. | The diagnostic value of serum C-reactive protein, procalcitonin, interleukin-6 and lactate dehydrogenase in patients with severe acute pancreatitis | |
| Takahashi et al. | Diagnostic accuracy of procalcitonin and presepsin for infectious disease in patients with acute kidney injury | |
| EP4113524A1 (en) | Method for measuring platelet activation based on soluble clec-2 and platelet count | |
| US10656163B2 (en) | Method for determining risk of pre-eclampsia | |
| JP6639392B2 (ja) | 胎盤成長因子2の選択的測定法 | |
| JP2009539080A (ja) | 急性冠状動脈症候群のリスクを有する個体の識別のためのmrp8/14レベルの使用 | |
| EP2496946B1 (en) | NT-pro ANP AND sFlt-1 FOR THE DIFFERENTIATION BETWEEN CIRCULATORY AND ISCHEMIC EVENTS | |
| Akdur et al. | Diagnostic utility of hematological indices in predicting adverse outcomes and severity of acute pancreatitis based on BISAP and modified Glasgow score | |
| Samouilidou et al. | Prognostic and diagnostic value of endocan in kidney diseases | |
| WO2022154045A1 (ja) | 可溶型clec2を用いた癌患者における血栓症リスクの予測方法 | |
| JP2004503254A (ja) | リポタンパク質と急性期タンパク質との複合体を検出し、系の不全または致死のリスクの増加を予測する方法 | |
| Ding et al. | Increased serum concentrations of signal peptide-Cub-Egf domain-containing protein-1 in patients with aneurysmal subarachnoid hemorrhage | |
| Jotheeswaran et al. | Role of inflammatory and nutritional markers in predicting complications after pancreaticoduodenectomy | |
| El Lehleh et al. | Diagnostic value of platelet indices, carbohydrate antigen 19-9 and carcinoembryonic antigen in differentiating malignant from benign gastric ulcers | |
| Gombert et al. | Increase of urinary TIMP-2 and IGFBP7 as potential predictor of acute kidney injury requiring renal replacement therapy and patients’ outcome following complex endovascular and open thoracic abdominal aortic aneurysm surgery–a prospective observational study | |
| WO2022255349A1 (ja) | 可溶型clec2を用いた出血性脳卒中のリスク評価法 | |
| Ye et al. | Correlation and diagnostic value of serum cys-C, RBP4, and NGAL with the condition of patients with traumatic acute kidney injury | |
| WO2022255348A1 (ja) | 可溶型clec2を用いた急性脳血管障害のリスク評価方法 | |
| JPWO2014034168A1 (ja) | 早期腎障害の評価マーカーとその測定方法 | |
| JP7419341B2 (ja) | 心房細動の評価における循環DKK3(Dickkopf関連タンパク質3) | |
| JP6004322B2 (ja) | 心血管イベントの発症リスクの検査方法 | |
| Abdulaziz et al. | Fecal Calgranulin C a Novel Noninvasive Marker in Predicting Activity and Severity of Ulcerative Colitis Compared with Colonoscopy. | |
| Mariusz Wójcik et al. | Evaluation of platelet indexes as potential biomarkers of suspected pulmonary embolism |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22739450 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2022575626 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280009906.2 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 20237025829 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022739450 Country of ref document: EP Effective date: 20230814 |