WO2022085593A1 - ポリエステルエラストマー樹脂組成物及び発泡成形体 - Google Patents
ポリエステルエラストマー樹脂組成物及び発泡成形体 Download PDFInfo
- Publication number
- WO2022085593A1 WO2022085593A1 PCT/JP2021/038277 JP2021038277W WO2022085593A1 WO 2022085593 A1 WO2022085593 A1 WO 2022085593A1 JP 2021038277 W JP2021038277 W JP 2021038277W WO 2022085593 A1 WO2022085593 A1 WO 2022085593A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- foamed
- polyester elastomer
- molded product
- thermoplastic polyester
- resin composition
- Prior art date
Links
- 239000006260 foam Substances 0.000 title claims abstract description 95
- 239000011342 resin composition Substances 0.000 title claims abstract description 71
- 229920000728 polyester Polymers 0.000 title claims abstract description 34
- 229920001971 elastomer Polymers 0.000 title description 18
- 239000000806 elastomer Substances 0.000 title description 17
- 229920006346 thermoplastic polyester elastomer Polymers 0.000 claims abstract description 99
- 239000002562 thickening agent Substances 0.000 claims abstract description 33
- 125000001931 aliphatic group Chemical group 0.000 claims abstract description 26
- 238000010097 foam moulding Methods 0.000 claims abstract description 20
- 239000004417 polycarbonate Substances 0.000 claims abstract description 13
- 229920000515 polycarbonate Polymers 0.000 claims abstract description 13
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 claims abstract description 10
- 125000003118 aryl group Chemical group 0.000 claims abstract description 9
- 229920000570 polyether Polymers 0.000 claims abstract description 6
- 229920003232 aliphatic polyester Polymers 0.000 claims abstract description 5
- 239000010410 layer Substances 0.000 claims description 65
- 150000001875 compounds Chemical class 0.000 claims description 43
- 239000002344 surface layer Substances 0.000 claims description 42
- 125000003700 epoxy group Chemical group 0.000 claims description 20
- 238000005259 measurement Methods 0.000 claims description 9
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 claims description 4
- 239000004721 Polyphenylene oxide Substances 0.000 claims description 4
- 150000002009 diols Chemical class 0.000 abstract description 9
- 239000000470 constituent Substances 0.000 abstract 1
- 239000000047 product Substances 0.000 description 114
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 50
- 238000005187 foaming Methods 0.000 description 42
- 238000000034 method Methods 0.000 description 32
- -1 aromatic dicarboxylic acids Chemical class 0.000 description 29
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 27
- 239000004088 foaming agent Substances 0.000 description 27
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 24
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 21
- 239000007789 gas Substances 0.000 description 20
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 18
- 239000004593 Epoxy Substances 0.000 description 17
- VPKDCDLSJZCGKE-UHFFFAOYSA-N carbodiimide group Chemical group N=C=N VPKDCDLSJZCGKE-UHFFFAOYSA-N 0.000 description 14
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 14
- 239000000463 material Substances 0.000 description 14
- 239000000126 substance Substances 0.000 description 14
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 13
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 13
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 13
- 229920001577 copolymer Polymers 0.000 description 13
- 239000000178 monomer Substances 0.000 description 13
- 229910052757 nitrogen Inorganic materials 0.000 description 13
- 238000002347 injection Methods 0.000 description 12
- 239000007924 injection Substances 0.000 description 12
- 238000000465 moulding Methods 0.000 description 12
- 239000000203 mixture Substances 0.000 description 11
- 239000011162 core material Substances 0.000 description 10
- WOZVHXUHUFLZGK-UHFFFAOYSA-N dimethyl terephthalate Chemical compound COC(=O)C1=CC=C(C(=O)OC)C=C1 WOZVHXUHUFLZGK-UHFFFAOYSA-N 0.000 description 10
- 238000001746 injection moulding Methods 0.000 description 10
- 239000001569 carbon dioxide Substances 0.000 description 9
- 229910002092 carbon dioxide Inorganic materials 0.000 description 9
- 238000011049 filling Methods 0.000 description 9
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 9
- 239000002994 raw material Substances 0.000 description 9
- 229920005989 resin Polymers 0.000 description 9
- 239000011347 resin Substances 0.000 description 9
- KKEYFWRCBNTPAC-UHFFFAOYSA-N Terephthalic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-N 0.000 description 8
- 230000000052 comparative effect Effects 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 238000002844 melting Methods 0.000 description 8
- 230000008018 melting Effects 0.000 description 8
- 229920000642 polymer Polymers 0.000 description 8
- 239000002253 acid Substances 0.000 description 7
- 239000000654 additive Substances 0.000 description 7
- 125000000524 functional group Chemical group 0.000 description 7
- 239000012071 phase Substances 0.000 description 7
- 238000006243 chemical reaction Methods 0.000 description 6
- 238000002156 mixing Methods 0.000 description 6
- 229920005992 thermoplastic resin Polymers 0.000 description 6
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 6
- 229920002554 vinyl polymer Polymers 0.000 description 6
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 5
- 238000005520 cutting process Methods 0.000 description 5
- YPFDHNVEDLHUCE-UHFFFAOYSA-N propane-1,3-diol Chemical compound OCCCO YPFDHNVEDLHUCE-UHFFFAOYSA-N 0.000 description 5
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- 239000005058 Isophorone diisocyanate Substances 0.000 description 4
- KXBFLNPZHXDQLV-UHFFFAOYSA-N [cyclohexyl(diisocyanato)methyl]cyclohexane Chemical compound C1CCCCC1C(N=C=O)(N=C=O)C1CCCCC1 KXBFLNPZHXDQLV-UHFFFAOYSA-N 0.000 description 4
- 150000001412 amines Chemical class 0.000 description 4
- 125000004432 carbon atom Chemical group C* 0.000 description 4
- 238000007872 degassing Methods 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 239000000945 filler Substances 0.000 description 4
- 230000009477 glass transition Effects 0.000 description 4
- 230000001771 impaired effect Effects 0.000 description 4
- 239000012948 isocyanate Substances 0.000 description 4
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 230000008719 thickening Effects 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 239000004594 Masterbatch (MB) Substances 0.000 description 3
- 125000002723 alicyclic group Chemical group 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 229910001873 dinitrogen Inorganic materials 0.000 description 3
- 239000012530 fluid Substances 0.000 description 3
- XXMIOPMDWAUFGU-UHFFFAOYSA-N hexane-1,6-diol Chemical compound OCCCCCCO XXMIOPMDWAUFGU-UHFFFAOYSA-N 0.000 description 3
- 239000011261 inert gas Substances 0.000 description 3
- 150000002513 isocyanates Chemical group 0.000 description 3
- 229920001610 polycaprolactone Polymers 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- 229920001451 polypropylene glycol Polymers 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- CDMDQYCEEKCBGR-UHFFFAOYSA-N 1,4-diisocyanatocyclohexane Chemical compound O=C=NC1CCC(N=C=O)CC1 CDMDQYCEEKCBGR-UHFFFAOYSA-N 0.000 description 2
- ALVZNPYWJMLXKV-UHFFFAOYSA-N 1,9-Nonanediol Chemical compound OCCCCCCCCCO ALVZNPYWJMLXKV-UHFFFAOYSA-N 0.000 description 2
- SXFJDZNJHVPHPH-UHFFFAOYSA-N 3-methylpentane-1,5-diol Chemical compound OCCC(C)CCO SXFJDZNJHVPHPH-UHFFFAOYSA-N 0.000 description 2
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- 239000004793 Polystyrene Substances 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- 125000004018 acid anhydride group Chemical group 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 238000004378 air conditioning Methods 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- 230000001588 bifunctional effect Effects 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 239000007795 chemical reaction product Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 description 2
- 125000005442 diisocyanate group Chemical group 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 239000003063 flame retardant Substances 0.000 description 2
- 239000011810 insulating material Substances 0.000 description 2
- QQVIHTHCMHWDBS-UHFFFAOYSA-N isophthalic acid Chemical compound OC(=O)C1=CC=CC(C(O)=O)=C1 QQVIHTHCMHWDBS-UHFFFAOYSA-N 0.000 description 2
- 238000004898 kneading Methods 0.000 description 2
- 239000007791 liquid phase Substances 0.000 description 2
- 238000000691 measurement method Methods 0.000 description 2
- RXOHFPCZGPKIRD-UHFFFAOYSA-N naphthalene-2,6-dicarboxylic acid Chemical compound C1=C(C(O)=O)C=CC2=CC(C(=O)O)=CC=C21 RXOHFPCZGPKIRD-UHFFFAOYSA-N 0.000 description 2
- BDJRBEYXGGNYIS-UHFFFAOYSA-N nonanedioic acid Chemical compound OC(=O)CCCCCCCC(O)=O BDJRBEYXGGNYIS-UHFFFAOYSA-N 0.000 description 2
- OEIJHBUUFURJLI-UHFFFAOYSA-N octane-1,8-diol Chemical compound OCCCCCCCCO OEIJHBUUFURJLI-UHFFFAOYSA-N 0.000 description 2
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 2
- 230000000704 physical effect Effects 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 238000012805 post-processing Methods 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- CXMXRPHRNRROMY-UHFFFAOYSA-N sebacic acid Chemical compound OC(=O)CCCCCCCCC(O)=O CXMXRPHRNRROMY-UHFFFAOYSA-N 0.000 description 2
- 239000002689 soil Substances 0.000 description 2
- 229920001169 thermoplastic Polymers 0.000 description 2
- 239000004416 thermosoftening plastic Substances 0.000 description 2
- 238000004448 titration Methods 0.000 description 2
- 239000013585 weight reducing agent Substances 0.000 description 2
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical group C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 1
- NNOZGCICXAYKLW-UHFFFAOYSA-N 1,2-bis(2-isocyanatopropan-2-yl)benzene Chemical compound O=C=NC(C)(C)C1=CC=CC=C1C(C)(C)N=C=O NNOZGCICXAYKLW-UHFFFAOYSA-N 0.000 description 1
- FKTHNVSLHLHISI-UHFFFAOYSA-N 1,2-bis(isocyanatomethyl)benzene Chemical compound O=C=NCC1=CC=CC=C1CN=C=O FKTHNVSLHLHISI-UHFFFAOYSA-N 0.000 description 1
- SBJCUZQNHOLYMD-UHFFFAOYSA-N 1,5-Naphthalene diisocyanate Chemical compound C1=CC=C2C(N=C=O)=CC=CC2=C1N=C=O SBJCUZQNHOLYMD-UHFFFAOYSA-N 0.000 description 1
- RTBFRGCFXZNCOE-UHFFFAOYSA-N 1-methylsulfonylpiperidin-4-one Chemical compound CS(=O)(=O)N1CCC(=O)CC1 RTBFRGCFXZNCOE-UHFFFAOYSA-N 0.000 description 1
- QFGCFKJIPBRJGM-UHFFFAOYSA-N 12-[(2-methylpropan-2-yl)oxy]-12-oxododecanoic acid Chemical class CC(C)(C)OC(=O)CCCCCCCCCCC(O)=O QFGCFKJIPBRJGM-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- QPBYBLZYMNWGMO-UHFFFAOYSA-N 2,2,3-trimethyloxirane Chemical compound CC1OC1(C)C QPBYBLZYMNWGMO-UHFFFAOYSA-N 0.000 description 1
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 description 1
- SDQROPCSKIYYAV-UHFFFAOYSA-N 2-methyloctane-1,8-diol Chemical compound OCC(C)CCCCCCO SDQROPCSKIYYAV-UHFFFAOYSA-N 0.000 description 1
- WSQZNZLOZXSBHA-UHFFFAOYSA-N 3,8-dioxabicyclo[8.2.2]tetradeca-1(12),10,13-triene-2,9-dione Chemical group O=C1OCCCCOC(=O)C2=CC=C1C=C2 WSQZNZLOZXSBHA-UHFFFAOYSA-N 0.000 description 1
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 description 1
- MEJAPGGFIJZHEJ-UHFFFAOYSA-N 5-acetamido-1,3,4-thiadiazole-2-sulfonyl chloride Chemical class CC(=O)NC1=NN=C(S(Cl)(=O)=O)S1 MEJAPGGFIJZHEJ-UHFFFAOYSA-N 0.000 description 1
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 1
- ATRRKUHOCOJYRX-UHFFFAOYSA-N Ammonium bicarbonate Chemical compound [NH4+].OC([O-])=O ATRRKUHOCOJYRX-UHFFFAOYSA-N 0.000 description 1
- XFXPMWWXUTWYJX-UHFFFAOYSA-N Cyanide Chemical compound N#[C-] XFXPMWWXUTWYJX-UHFFFAOYSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 239000005057 Hexamethylene diisocyanate Substances 0.000 description 1
- HDONYZHVZVCMLR-UHFFFAOYSA-N N=C=O.N=C=O.CC1CCCCC1 Chemical compound N=C=O.N=C=O.CC1CCCCC1 HDONYZHVZVCMLR-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- WNTSGVPAFRQERO-UHFFFAOYSA-N OC12C(C=CC3=CC(=CC=C13)O)C1C(COCC3C2O3)O1 Chemical compound OC12C(C=CC3=CC(=CC=C13)O)C1C(COCC3C2O3)O1 WNTSGVPAFRQERO-UHFFFAOYSA-N 0.000 description 1
- ALQSHHUCVQOPAS-UHFFFAOYSA-N Pentane-1,5-diol Chemical compound OCCCCCO ALQSHHUCVQOPAS-UHFFFAOYSA-N 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 1
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical class C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 description 1
- YIMQCDZDWXUDCA-UHFFFAOYSA-N [4-(hydroxymethyl)cyclohexyl]methanol Chemical compound OCC1CCC(CO)CC1 YIMQCDZDWXUDCA-UHFFFAOYSA-N 0.000 description 1
- 239000011358 absorbing material Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 239000012773 agricultural material Substances 0.000 description 1
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 1
- 239000001099 ammonium carbonate Substances 0.000 description 1
- 235000012501 ammonium carbonate Nutrition 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 150000008064 anhydrides Chemical class 0.000 description 1
- JFCQEDHGNNZCLN-UHFFFAOYSA-N anhydrous glutaric acid Natural products OC(=O)CCCC(O)=O JFCQEDHGNNZCLN-UHFFFAOYSA-N 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 239000002216 antistatic agent Substances 0.000 description 1
- 150000001540 azides Chemical class 0.000 description 1
- 125000000751 azo group Chemical group [*]N=N[*] 0.000 description 1
- VJRITMATACIYAF-UHFFFAOYSA-N benzenesulfonohydrazide Chemical compound NNS(=O)(=O)C1=CC=CC=C1 VJRITMATACIYAF-UHFFFAOYSA-N 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- QRUDEWIWKLJBPS-UHFFFAOYSA-N benzotriazole Chemical compound C1=CC=C2N[N][N]C2=C1 QRUDEWIWKLJBPS-UHFFFAOYSA-N 0.000 description 1
- 239000012964 benzotriazole Substances 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 239000011489 building insulation material Substances 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000001718 carbodiimides Chemical class 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 238000010411 cooking Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- 239000003484 crystal nucleating agent Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- QYQADNCHXSEGJT-UHFFFAOYSA-N cyclohexane-1,1-dicarboxylate;hydron Chemical compound OC(=O)C1(C(O)=O)CCCCC1 QYQADNCHXSEGJT-UHFFFAOYSA-N 0.000 description 1
- ZWAJLVLEBYIOTI-UHFFFAOYSA-N cyclohexene oxide Chemical group C1CCCC2OC21 ZWAJLVLEBYIOTI-UHFFFAOYSA-N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 238000013016 damping Methods 0.000 description 1
- 238000005262 decarbonization Methods 0.000 description 1
- 230000006837 decompression Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 150000001991 dicarboxylic acids Chemical class 0.000 description 1
- GPLRAVKSCUXZTP-UHFFFAOYSA-N diglycerol Chemical compound OCC(O)COCC(O)CO GPLRAVKSCUXZTP-UHFFFAOYSA-N 0.000 description 1
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical compound C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000003912 environmental pollution Methods 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 239000006081 fluorescent whitening agent Substances 0.000 description 1
- XJRPXNWUCVZVJP-UHFFFAOYSA-N formyl azide Chemical compound [N-]=[N+]=NC=O XJRPXNWUCVZVJP-UHFFFAOYSA-N 0.000 description 1
- ANSXAPJVJOKRDJ-UHFFFAOYSA-N furo[3,4-f][2]benzofuran-1,3,5,7-tetrone Chemical compound C1=C2C(=O)OC(=O)C2=CC2=C1C(=O)OC2=O ANSXAPJVJOKRDJ-UHFFFAOYSA-N 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- VOZRXNHHFUQHIL-UHFFFAOYSA-N glycidyl methacrylate Chemical compound CC(=C)C(=O)OCC1CO1 VOZRXNHHFUQHIL-UHFFFAOYSA-N 0.000 description 1
- RRAMGCGOFNQTLD-UHFFFAOYSA-N hexamethylene diisocyanate Chemical compound O=C=NCCCCCCN=C=O RRAMGCGOFNQTLD-UHFFFAOYSA-N 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000001023 inorganic pigment Substances 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 239000006078 metal deactivator Substances 0.000 description 1
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000003607 modifier Substances 0.000 description 1
- 239000006082 mold release agent Substances 0.000 description 1
- ALIFPGGMJDWMJH-UHFFFAOYSA-N n-phenyldiazenylaniline Chemical compound C=1C=CC=CC=1NN=NC1=CC=CC=C1 ALIFPGGMJDWMJH-UHFFFAOYSA-N 0.000 description 1
- KYTZHLUVELPASH-UHFFFAOYSA-N naphthalene-1,2-dicarboxylic acid Chemical compound C1=CC=CC2=C(C(O)=O)C(C(=O)O)=CC=C21 KYTZHLUVELPASH-UHFFFAOYSA-N 0.000 description 1
- DFQICHCWIIJABH-UHFFFAOYSA-N naphthalene-2,7-diol Chemical compound C1=CC(O)=CC2=CC(O)=CC=C21 DFQICHCWIIJABH-UHFFFAOYSA-N 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 150000002832 nitroso derivatives Chemical class 0.000 description 1
- 239000002667 nucleating agent Substances 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000012860 organic pigment Substances 0.000 description 1
- UHHKSVZZTYJVEG-UHFFFAOYSA-N oxepane Chemical compound C1CCCOCC1 UHHKSVZZTYJVEG-UHFFFAOYSA-N 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 150000003018 phosphorus compounds Chemical class 0.000 description 1
- LGRFSURHDFAFJT-UHFFFAOYSA-N phthalic anhydride Chemical compound C1=CC=C2C(=O)OC(=O)C2=C1 LGRFSURHDFAFJT-UHFFFAOYSA-N 0.000 description 1
- 229920001515 polyalkylene glycol Polymers 0.000 description 1
- 239000003505 polymerization initiator Substances 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920000166 polytrimethylene carbonate Polymers 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000001294 propane Substances 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 230000001012 protector Effects 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- JZWFDVDETGFGFC-UHFFFAOYSA-N salacetamide Chemical group CC(=O)NC(=O)C1=CC=CC=C1O JZWFDVDETGFGFC-UHFFFAOYSA-N 0.000 description 1
- 239000003566 sealing material Substances 0.000 description 1
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N sodium azide Substances [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000007711 solidification Methods 0.000 description 1
- 230000008023 solidification Effects 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 150000003440 styrenes Chemical class 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- DVKJHBMWWAPEIU-UHFFFAOYSA-N toluene 2,4-diisocyanate Chemical compound CC1=CC=C(N=C=O)C=C1N=C=O DVKJHBMWWAPEIU-UHFFFAOYSA-N 0.000 description 1
- RUELTTOHQODFPA-UHFFFAOYSA-N toluene 2,6-diisocyanate Chemical compound CC1=C(N=C=O)C=CC=C1N=C=O RUELTTOHQODFPA-UHFFFAOYSA-N 0.000 description 1
- 238000005809 transesterification reaction Methods 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- SRPWOOOHEPICQU-UHFFFAOYSA-N trimellitic anhydride Chemical compound OC(=O)C1=CC=C2C(=O)OC(=O)C2=C1 SRPWOOOHEPICQU-UHFFFAOYSA-N 0.000 description 1
- 229940124543 ultraviolet light absorber Drugs 0.000 description 1
- 229920001567 vinyl ester resin Polymers 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
- PAPBSGBWRJIAAV-UHFFFAOYSA-N ε-Caprolactone Chemical group O=C1CCCCCO1 PAPBSGBWRJIAAV-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L67/00—Compositions of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Compositions of derivatives of such polymers
- C08L67/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
- C08L67/025—Polyesters derived from dicarboxylic acids and dihydroxy compounds containing polyether sequences
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C44/00—Shaping by internal pressure generated in the material, e.g. swelling or foaming ; Producing porous or cellular expanded plastics articles
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F212/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an aromatic carbocyclic ring
- C08F212/02—Monomers containing only one unsaturated aliphatic radical
- C08F212/04—Monomers containing only one unsaturated aliphatic radical containing one ring
- C08F212/06—Hydrocarbons
- C08F212/08—Styrene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/32—Esters containing oxygen in addition to the carboxy oxygen containing epoxy radicals
- C08F220/325—Esters containing oxygen in addition to the carboxy oxygen containing epoxy radicals containing glycidyl radical, e.g. glycidyl (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/02—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds
- C08G63/12—Polyesters derived from hydroxycarboxylic acids or from polycarboxylic acids and polyhydroxy compounds derived from polycarboxylic acids and polyhydroxy compounds
- C08G63/16—Dicarboxylic acids and dihydroxy compounds
- C08G63/18—Dicarboxylic acids and dihydroxy compounds the acids or hydroxy compounds containing carbocyclic rings
- C08G63/181—Acids containing aromatic rings
- C08G63/183—Terephthalic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G63/00—Macromolecular compounds obtained by reactions forming a carboxylic ester link in the main chain of the macromolecule
- C08G63/66—Polyesters containing oxygen in the form of ether groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/0014—Use of organic additives
- C08J9/0028—Use of organic additives containing nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/0061—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof characterized by the use of several polymeric components
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/34—Chemical features in the manufacture of articles consisting of a foamed macromolecular core and a macromolecular surface layer having a higher density than the core
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29C—SHAPING OR JOINING OF PLASTICS; SHAPING OF MATERIAL IN A PLASTIC STATE, NOT OTHERWISE PROVIDED FOR; AFTER-TREATMENT OF THE SHAPED PRODUCTS, e.g. REPAIRING
- B29C44/00—Shaping by internal pressure generated in the material, e.g. swelling or foaming ; Producing porous or cellular expanded plastics articles
- B29C44/34—Auxiliary operations
- B29C44/36—Feeding the material to be shaped
- B29C44/38—Feeding the material to be shaped into a closed space, i.e. to make articles of definite length
- B29C44/42—Feeding the material to be shaped into a closed space, i.e. to make articles of definite length using pressure difference, e.g. by injection or by vacuum
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29K—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES B29B, B29C OR B29D, RELATING TO MOULDING MATERIALS OR TO MATERIALS FOR MOULDS, REINFORCEMENTS, FILLERS OR PREFORMED PARTS, e.g. INSERTS
- B29K2067/00—Use of polyesters or derivatives thereof, as moulding material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29K—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES B29B, B29C OR B29D, RELATING TO MOULDING MATERIALS OR TO MATERIALS FOR MOULDS, REINFORCEMENTS, FILLERS OR PREFORMED PARTS, e.g. INSERTS
- B29K2067/00—Use of polyesters or derivatives thereof, as moulding material
- B29K2067/006—PBT, i.e. polybutylene terephthalate
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29K—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES B29B, B29C OR B29D, RELATING TO MOULDING MATERIALS OR TO MATERIALS FOR MOULDS, REINFORCEMENTS, FILLERS OR PREFORMED PARTS, e.g. INSERTS
- B29K2105/00—Condition, form or state of moulded material or of the material to be shaped
- B29K2105/04—Condition, form or state of moulded material or of the material to be shaped cellular or porous
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2101/00—Manufacture of cellular products
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2110/00—Foam properties
- C08G2110/0041—Foam properties having specified density
- C08G2110/005—< 50kg/m3
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2203/00—Foams characterized by the expanding agent
- C08J2203/06—CO2, N2 or noble gases
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2203/00—Foams characterized by the expanding agent
- C08J2203/08—Supercritical fluid
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2205/00—Foams characterised by their properties
- C08J2205/04—Foams characterised by their properties characterised by the foam pores
- C08J2205/044—Micropores, i.e. average diameter being between 0,1 micrometer and 0,1 millimeter
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2367/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2367/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2425/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an aromatic carbocyclic ring; Derivatives of such polymers
- C08J2425/02—Homopolymers or copolymers of hydrocarbons
- C08J2425/04—Homopolymers or copolymers of styrene
- C08J2425/08—Copolymers of styrene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J9/00—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof
- C08J9/04—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof using blowing gases generated by a previously added blowing agent
- C08J9/12—Working-up of macromolecular substances to porous or cellular articles or materials; After-treatment thereof using blowing gases generated by a previously added blowing agent by a physical blowing agent
- C08J9/122—Hydrogen, oxygen, CO2, nitrogen or noble gases
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2201/00—Properties
- C08L2201/08—Stabilised against heat, light or radiation or oxydation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2203/00—Applications
- C08L2203/14—Applications used for foams
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2207/00—Properties characterising the ingredient of the composition
- C08L2207/04—Thermoplastic elastomer
Definitions
- the present invention relates to a thermoplastic polyester elastomer resin composition suitable for foam molding using counter pressure, and also relates to a foam molded body in which the thermoplastic polyester elastomer resin composition forms a continuous phase. More specifically, the foamed molded product of the present invention is a foamed molded product having excellent impact resilience and surface smoothness, and it is possible to provide a foamed molded product that has been foamed twice or more.
- Thermoplastic polyester elastomer is excellent in injection moldability and extrusion moldability, has high mechanical strength, has rubber properties such as elastic recovery, impact resistance, and flexibility, and is a material with excellent cold resistance. It is used in applications such as electronic parts, textiles, films, and sports parts.
- Thermoplastic polyester elastomer is used for automobile parts, especially parts used in high temperature environment and automobile interior parts, because it has excellent heat aging resistance, light resistance, and wear resistance. Further, in recent years, the weight of resin parts has been reduced, and the application of foam molded products can be mentioned as one of the means for achieving the purpose.
- Patent Document 1 As one of the high-fold foaming methods for weight reduction, there is a core back injection foam molding method that moves the mold in the mold opening direction at the time of foaming. Further, since the bubbles in the foam layer become fine, the impact resilience becomes high (Patent Document 1).
- the foam molded product produced by the core back injection foam molding method has a non-foam skin layer on the surface layer and the above-mentioned foam layer on the inner layer, and has a sandwich structure of the non-foam skin layer and the foam layer in the thickness direction, and is non-foam.
- the presence of the skin layer of the foam layer alleviates the resilience of the foam layer, lowers the rebound resilience, and causes unevenness on the surface of the molded product due to defects such as swirl marks and avatars, resulting in surface smoothness. Inferior to.
- Patent Document 2 if the short shot injection foam molding method is used, it is possible to produce a foam molded product having a thin skin layer, but there is a problem that the foaming ratio is low and the weight is inferior (Patent Document 2).
- urethane foam having a high resilience of 60% or more is preferably adopted, and Patent Document 3 proposes a manufacturing method thereof.
- urethane foam has a problem of environmental pollution because cyanide gas or the like is generated at the time of combustion.
- the present invention has been made in view of the current state of the prior art, and an object of the present invention is to provide a thermoplastic polyester elastomer resin composition suitable for a foam molded product having excellent lightness, impact resilience and surface smoothness. It is in.
- the present inventor has diligently studied the composition of the thermoplastic polyester elastomer and the thickener to be blended in order to achieve the above object.
- the hard segment and the soft segment of the thermoplastic polyester elastomer have a specific ratio, and a thickener is added to obtain a thermoplastic polyester elastomer resin composition adjusted to a specific melt viscosity, and this thermoplastic polyester is obtained.
- a foamed molded product that exhibits good foamable formability that combines fine foaming and surface smoothness, enables weight reduction by high-magnification foaming, and has an extremely high impact elastic modulus. I found that I could get it.
- the foam injection molding method by the counter pressure method in which gas is injected into the cavity of the mold and the thermoplastic resin melted under pressure is injected, the above-mentioned high-quality polyester elastomer foam molded product can be obtained.
- the characteristics of the thermoplastic polyester elastomer resin composition (melt tension, crystallization temperature, gas retention, etc.) are suitable for the foam molding method by the counter pressure method, and it is suitable for the process of pressurizing and releasing the counter pressure.
- a foamed molded article that can withstand and has the desired high-fold foaming and surface smoothness can be obtained, and the present invention has been completed.
- the present invention constitutes the following (1) to (8).
- a hard segment made of a polyester containing an aromatic dicarboxylic acid and an aliphatic and / or an aliphatic diol, and at least one selected from an aliphatic polyether, an aliphatic polyester, and an aliphatic polycarbonate. It is characterized by containing 0.05 to 4.5 parts by mass of the thickener with respect to 100 parts by mass of the thermoplastic polyester elastomer to which the soft segments are bonded and the content of the soft segments is 25 to 90% by mass.
- thermoplastic polyester elastomer resin composition according to (1) wherein the thickener is a reactive compound having an epoxy group.
- the foam molded product made of the thermoplastic polyester elastomer resin composition of the present invention not only has excellent lightness, but also exhibits extremely high impact resilience and has excellent surface smoothness. Furthermore, it is possible to provide a foamed molded product that can be applied to parts that require high reliability because it has a uniform foaming state despite a high foaming ratio, high heat resistance, water resistance, and molding stability. can. Then, by using the foam injection molding method by the counter pressure method, it has the above-mentioned excellent characteristics having an arbitrary desired shape by preparing a corresponding mold without performing post-processing such as cutting. A foam molded product can be obtained.
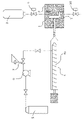
- FIG. 1 It is a schematic block diagram for demonstrating an example of the manufacturing method of the foam molded article of this invention.
- the thermoplastic polyester elastomer used in the present invention is formed by bonding a hard segment and a soft segment.
- the hard segment consists of polyester.
- the aromatic dicarboxylic acid constituting the hard segment polyester an ordinary aromatic dicarboxylic acid is widely used and is not particularly limited, but the main aromatic dicarboxylic acid is terephthalic acid or naphthalenedicarboxylic acid (among isomers). 2,6-naphthalenedicarboxylic acid is preferable).
- the content of these aromatic dicarboxylic acids is preferably 70 mol% or more, more preferably 80 mol% or more, based on the total dicarboxylic acids constituting the polyester of the hard segment.
- dicarboxylic acid components include aromatic dicarboxylic acids such as diphenyldicarboxylic acid, isophthalic acid, and 5-sodium sulfoisophthalic acid, alicyclic dicarboxylic acids such as cyclohexanedicarboxylic acid and tetrahydrohydride phthalic acid, succinic acid, and glutaric acid.
- aromatic dicarboxylic acids such as diphenyldicarboxylic acid, isophthalic acid, and 5-sodium sulfoisophthalic acid
- alicyclic dicarboxylic acids such as cyclohexanedicarboxylic acid and tetrahydrohydride phthalic acid
- succinic acid succinic acid
- glutaric acid examples thereof include aliphatic dicarboxylic acids such as adipic acid, azelaic acid, sebacic acid, dodecanedioic acid, dimer acid and hydrogenated dimer acid.
- thermoplastic polyester elastomer used in the present invention as the aliphatic or alicyclic diol constituting the polyester of the hard segment, a general aliphatic or alicyclic diol is widely used, and is not particularly limited, but mainly. It is desirable that it is an alkylene glycol having 2 to 8 carbon atoms. Specific examples thereof include ethylene glycol, 1,3-propylene glycol, 1,4-butanediol, 1,6-hexanediol, and 1,4-cyclohexanedimethanol. Among these, ethylene glycol or 1,4-butanediol is preferable.
- the components constituting the above-mentioned hard segment polyester include butylene terephthalate unit (unit consisting of terephthalic acid and 1,4-butanediol) or butylene naphthalate unit (2,6-naphthalenedicarboxylic acid and 1,4-butanediol).
- a unit consisting of (a unit consisting of) is preferable from the viewpoint of physical properties, formability, and cost performance.
- the aromatic polyester is a normal polyester. It can be easily obtained according to the manufacturing method. Further, it is desirable that the polyester has a number average molecular weight of 10,000 to 40,000.
- the soft segment of the thermoplastic polyester elastomer used in the present invention is at least one selected from aliphatic polyethers, aliphatic polyesters, and aliphatic polycarbonates.
- Examples of the aliphatic polyether include poly (ethylene oxide) glycol, poly (propylene oxide) glycol, poly (tetramethylene oxide) glycol, poly (hexamethylene oxide) glycol, poly (trimethylethylene oxide) glycol, and both ethylene oxide and propylene oxide.
- Examples thereof include a polymer, an ethylene oxide adduct of poly (propylene oxide) glycol, and a copolymer of ethylene oxide and tetrahydrofuran.
- ethylene oxide adducts of poly (tetramethylene oxide) glycol and poly (propylene oxide) glycol are preferable from the viewpoint of elastic properties.
- Examples of the aliphatic polyester include poly ( ⁇ -caprolactone), polyenant lactone, polycaprilolactone, and polybutylene adipate.
- poly ( ⁇ -caprolactone) and polybutylene adipate are preferable from the viewpoint of elastic properties.
- the aliphatic polycarbonate is preferably composed mainly of aliphatic diol residues having 2 to 12 carbon atoms.
- these aliphatic diols include ethylene glycol, 1,3-propylene glycol, 1,4-butanediol, 1,5-pentanediol, 1,6-hexanediol, 1,8-octanediol, and 2, 2-Diol-1,3-propanediol, 3-methyl-1,5-pentanediol, 2,4-diol-1,5-pentanediol, 1,9-nonanediol, 2-methyl-1,8- Examples include octanediol.
- thermoplastic polyester elastomer an aliphatic diol having 5 to 12 carbon atoms is preferable from the viewpoint of the flexibility and low temperature characteristics of the obtained thermoplastic polyester elastomer.
- these components may be used alone or in combination of two or more, if necessary, based on the cases described below.
- an aliphatic polycarbonate diol constituting the soft segment of the thermoplastic polyester elastomer used in the present invention and having good low temperature characteristics those having a low melting point (for example, 70 ° C. or lower) and a low glass transition temperature are preferable.
- an aliphatic polycarbonate diol composed of 1,6-hexanediol used for forming a soft segment of a thermoplastic polyester elastomer has a low glass transition temperature of about -60 ° C and a melting point of about 50 ° C. Good low temperature characteristics.
- the aliphatic polycarbonate diol obtained by copolymerizing the above aliphatic polycarbonate diol with an appropriate amount of, for example, 3-methyl-1,5-pentanediol has a glass transition point with respect to the original aliphatic polycarbonate diol.
- the melting point is lowered or becomes amorphous, it corresponds to an aliphatic polycarbonate diol having good low temperature characteristics.
- the aliphatic polycarbonate diol composed of 1,9-nonane diol and 2-methyl-1,8-octane diol has a melting point of about 30 ° C. and a glass transition temperature of about ⁇ 70 ° C., which are sufficiently low.
- an aliphatic polyether is preferable from the viewpoint of solving the problem of the present invention.
- thermoplastic polyester elastomer used in the present invention is preferably a copolymer containing terephthalic acid, 1,4-butanediol, and poly (tetramethylene oxide) glycol as main components.
- terephthalic acid is preferably 40 mol% or more, more preferably 70 mol% or more, further preferably 80 mol% or more, and 90 mol%. The above is particularly preferable.
- the total of 1,4-butanediol and poly (tetramethylene oxide) glycol is preferably 40 mol% or more, more preferably 70 mol% or more, and 80. It is more preferably mol% or more, and particularly preferably 90 mol% or more.
- the number average molecular weight of the poly (tetramethylene oxide) glycol is preferably 500 to 4000. If the number average molecular weight is less than 500, it may be difficult to develop elastomeric properties. On the other hand, when the number average molecular weight exceeds 4000, the compatibility with the hard segment component is lowered, and it may be difficult to copolymerize in a block shape.
- the number average molecular weight of the poly (tetramethylene oxide) glycol is more preferably 800 or more and 3000 or less, and further preferably 1000 or more and 2500 or less.
- the content of the soft segment is 25 to 90% by mass, preferably 40 to 90% by mass, more preferably 55 to 90% by mass, and further preferably 65 to 90% by mass. %. If the content of the soft segment is lower than 25% by mass, the crystallinity is high and therefore the impact resilience is inferior, and if it exceeds 90% by mass, the crystallinity is too low and the foam formability tends to be inferior.
- thermoplastic polyester elastomer used in the present invention can be produced by a known method. For example, a method of transesterifying a lower alcohol diester of a dicarboxylic acid, an excess amount of low molecular weight glycol, and a soft segment component in the presence of a catalyst to polycondensate the resulting reaction product, a dicarboxylic acid and an excess amount of glycol and soft. A method in which the segment components are esterified in the presence of a catalyst and the obtained reaction product is polycondensed. A hard segment polyester is prepared in advance, and the soft segment component is added to the polyester to be randomized by a transesterification reaction.
- Any method may be used, such as a method of connecting the hard segment and the soft segment with a chain binder, and a method of adding ⁇ -caprolactone monomer to the hard segment when poly ( ⁇ -caprolactone) is used for the soft segment. ..
- the thickener in the present invention is a reactive compound having a hydroxyl group or a functional group capable of reacting with a carboxyl group of the thermoplastic polyester elastomer (hereinafter, may be simply referred to as a reactive compound), and the functional group capable of reacting.
- the group is preferably at least one selected from an epoxy group (glycidyl group), an acid anhydride group, a carbodiimide group and an isocyanate group, and contains two or more functional groups per molecule.
- the functional group is more preferably an epoxy group (glycidyl group).
- the thickener in the present invention is preferably a reactive compound having an epoxy group (glycidyl group), a reactive compound having a carbodiimide group, and more preferably a reactive compound having an epoxy group (glycidyl group). ..
- reactive compound having an epoxy group may be referred to as “compound having an epoxy group”. Since the thickener reacts with and binds to the polymer chain of the thermoplastic polyester elastomer, it also acts as a cross-linking agent.
- the thickener is a compound having an epoxy group
- a polyfunctional epoxy compound having two or more epoxy groups specifically, 1,6-dihydroxynaphthalenediglycidyl ether having two epoxy groups or 1 , 3-Bis (oxylanylmethoxy) benzene 1,3,5-tris (2,3-epoxidepropyl) -1,3,5-triazine-2,4,6 (1H,) with three epoxy groups 3H, 5H) -trione and diglycerol triglycidyl ether, 1-chloro-2,3-epoxide propane, formaldehyde, 2,7-naphthalenediol polycondensate with four epoxy groups and pentaerythritol polyglycidyl ether can be mentioned.
- a polyfunctional epoxy compound having heat resistance in the skeleton is preferable.
- a bifunctional or tetrafunctional epoxy compound having a naphthalene structure as a skeleton, or a trifunctional epoxy compound having a triazine structure as a skeleton is preferable.
- bifunctional or 3 Functional epoxy compounds are preferred.
- vinyl aromatic monomer (X) containing 2 or more glycidyl groups per molecule, having a weight average molecular weight of 4000 to 25000, and 20 to 99% by mass of vinyl aromatic monomer (X), and 1 to 80% by mass of glycidyl (meth).
- examples thereof include a copolymer composed of an acrylate (Y) and a vinyl group-containing monomer (Z) other than (X) which does not contain 0 to 79% by mass of an epoxy group.
- the thickener used in the present invention contains two or more glycidyl groups per molecule, has a weight average molecular weight of 4000 to 25000, and has a vinyl aromatic monomer (X) of 20 to 99% by mass, 1 to 80.
- a styrene-based polymer composed of a vinyl group-containing monomer (Z) other than (X) containing 0 to 79% by mass of glycidyl (meth) acrylate (Y) and 0 to 79% by weight of an epoxy group is a thermoplastic polyester. It is preferable because it has good compatibility with the elastomer and has a wider molecular weight distribution.
- the copolymer is composed of 20 to 99% by mass of (X), 1 to 80% by mass of (Y), and 0 to 40% by mass of (Z), and more preferably 25 to 90% by mass of (X). It is a copolymer composed of% by mass, (Y) 10 to 75% by mass, and (Z) 0 to 35% by mass. Since these compositions affect the concentration of functional groups that contribute to the reaction with the thermoplastic polyester elastomer, it is preferable to appropriately control the above range.
- Examples of the vinyl aromatic monomer (X) include styrene and ⁇ -methylstyrene.
- Examples of the glycidyl (meth) acrylate (Y) include glycidyl (meth) acrylic acid, (meth) acrylic acid ester having a cyclohexene oxide structure, (meth) acrylic glycidyl ether, and the like, and among these, reactive. Glysidyl (meth) acrylate is preferred because of its high value.
- Examples of the vinyl group-containing monomer (Z) other than the epoxy group-free (X) include methyl (meth) acrylate, ethyl (meth) acrylate, propyl (meth) acrylate, and butyl (meth) acrylate. , 2-Ethylhexyl (meth) acrylate, Cyclohexyl (meth) acrylate, Stearyl (meth) acrylate, methoxyethyl (meth) acrylate and other alkyl groups having 1 to 22 carbon atoms (alkyl groups are linear and branched.
- esters include esters, (meth) acrylic acid benzyl esters, (meth) acrylic acid phenoxyalkyl esters, (meth) acrylic acid isobornyl esters, and (meth) acrylic acid alkoxysilylalkyl esters.
- vinyl esters such as (meth) acrylamide, (meth) acrylic dialkylamide and vinyl acetate, vinyl ethers, aromatic vinyl-based monomers such as (meth) allyl ethers, and ⁇ -olefin monomers such as ethylene and propylene. Etc. can also be used as the vinyl group-containing monomer (Z) other than the epoxy group-free (X).
- the weight average molecular weight of the copolymer is preferably 4000 to 25000.
- the weight average molecular weight is more preferably 5000 to 15000. If the weight average molecular weight is less than 4000, the unreacted copolymer may volatilize in the molding process or bleed out to the surface of the molded product, causing surface contamination.
- the weight average molecular weight exceeds 25,000, the reaction with the thermoplastic polyester elastomer is delayed and the effect of increasing the molecular weight is insufficient, and the compatibility between the copolymer and the thermoplastic polyester elastomer is deteriorated. , There is a high possibility that the inherent properties of thermoplastic polyester elastomers, such as heat resistance, will deteriorate.
- the epoxy value of the copolymer is preferably 400 to 2500 equivalents / 1 ⁇ 10 6 g, more preferably 500 to 1500 equivalents / 1 ⁇ 10 6 g, still more preferably 600 to 1000 equivalents / 1 ⁇ 10. It is 6 g. If the epoxy value is less than 400 equivalents / 1 ⁇ 10 6 g, the thickening effect may not be exhibited, while if it exceeds 2500 equivalents / 1 ⁇ 10 6 g, the thickening effect becomes excessive and the moldability is improved. May have an adverse effect.
- a polycarbodiimide compound When the thickener is a compound having a carbodiimide group, a polycarbodiimide compound can be used.
- Polycarbodiimide compounds are advantageous in that they efficiently reduce the acid value.
- the polycarbodiimide compound can be obtained, for example, by a decarbonization reaction of the diisocyanate compound.
- the diisocyanate compound that can be used here include 4,4'-diphenylmethane diisocyanate, 4,4'-diphenyldimethylmethane diisocyanate, 1,3-phenylenediocyanate, 1,4-phenylenediocyanate, and 2,4-tolylene diisocyanate.
- 2,6-Toluene diisocyanate 1,5-naphthylene diisocyanate, hexamethylene diisocyanate, dicyclohexylmethane diisocyanate, cyclohexane-1,4-diisocyanate, xylylene diisocyanate, isophorone diisocyanate, methylcyclohexanediisocyanate, tetramethylxylylene diisocyanate, Examples thereof include 1,3,5-triisopropylphenylene-2,4-diisocyanate. Only one of these may be used, or two or more thereof may be copolymerized and used.
- a branched structure may be introduced, or a functional group other than a carbodiimide group or an isocyanate group may be introduced by copolymerization.
- the terminal isocyanate can be used as it is, the degree of polymerization may be controlled by reacting the terminal isocyanate, or a part of the terminal isocyanate may be blocked.
- polycarbodiimide compound alicyclic polycarbodiimide derived from dicyclohexylmethane diisocyanate, cyclohexane-1,4-diisocyanate, isophorone diisocyanate and the like is particularly preferable, and polycarbodiimide derived from dicyclohexylmethane diisocyanate and isophorone diisocyanate is particularly preferable.
- the polycarbodiimide compound preferably contains 2 to 50 carbodiimide groups per molecule in terms of stability and handleability. More preferably, each molecule contains 5 to 30 carbodiimide groups.
- the number of carbodiimides in the polycarbodiimide molecule corresponds to the degree of polymerization if the polycarbodiimide is obtained from a diisocyanate compound. For example, the degree of polymerization of polycarbodiimide obtained by connecting 21 diisocyanate compounds in a chain is 20 and the number of carbodiimide groups in the molecular chain is 20.
- a polycarbodiimide compound is a mixture of molecules of various lengths, and the number of carbodiimide groups is expressed as an average value. If it has a carbodiimide group in the above range and is solid at around room temperature, it can be powdered, so that it has excellent workability and compatibility when mixed with a thermoplastic polyester elastomer, and also in terms of uniform reactivity and bleed-out resistance. preferable.
- the number of carbodiimide groups can be measured, for example, by using a conventional method (a method of dissolving with an amine and performing back titration with hydrochloric acid).
- the polycarbodiimide compound has an isocyanate group at the terminal, and the isocyanate group content is preferably 0.5 to 4% by mass, which is preferable from the viewpoint of stability and handleability. More preferably, the isocyanate group content is 1 to 3% by mass. In particular, it is preferable that the polycarbodiimide derived from dicyclohexylmethane diisocyanate or isophorone diisocyanate has an isocyanate group content in the above range.
- the isocyanate group content can be measured by using a conventional method (a method of dissolving with amine and performing back titration with hydrochloric acid).
- the thickener is a compound having an isocyanate group
- examples thereof include the above-mentioned polycarbodiimide compound containing an isocyanate group and the isocyanate compound which is a raw material of the above-mentioned polycarbodiimide compound.
- the thickener is a compound having an acid anhydride group
- a compound containing 2 to 4 anhydrides per molecule is preferable in terms of stability and handleability.
- examples of such a compound include phthalic acid anhydride, trimellitic acid anhydride, pyromellitic acid anhydride and the like.
- the content of the thickener is 0.05 to 4.5 parts by mass, preferably 0.1 to 4 parts by mass, and more preferably 0.1 to 4 parts by mass with respect to 100 parts by mass of the thermoplastic polyester elastomer. 3 parts by mass.
- a thickener is contained, if it is less than 0.05 parts by mass, the targeted molecular chain extension effect is insufficient, and if it exceeds 4.5 parts by mass, the thickening effect becomes excessive and the moldability is adversely affected. And tends to affect the mechanical properties of the molded product.
- the thickener is an epoxy compound, if it exceeds 4.5 parts by mass, the surface of the molded product may be uneven due to the cohesive curing of the epoxy compound.
- the thickener is a carbodiimide compound, if it exceeds 4.5 parts by mass, the basicity of the polycarbodiimide compound causes hydrolysis of the thermoplastic polyester elastomer, which tends to affect the mechanical properties.
- a vinyl aromatic monomer (X) containing an epoxy group-containing compound particularly, two or more glycidyl groups per molecule, having a weight average molecular weight of 4000 to 25000, and 20 to 99% by mass.
- a compound having a highly reactive functional group such as a carbodiimide group is used in combination with an epoxy compound, the molecular weight distribution of the resin composition after thickening tends to be narrowed.
- the injection pressure during injection molding may increase, the foaming nuclei may disappear, and the foaming ratio may decrease. Therefore, in the resin composition of the present invention, it is preferable not to use the epoxy compound and the carbodiimide compound together as the thickener.
- thermoplastic Polyester Elastomer Resin Composition Further, in the thermoplastic polyester elastomer used in the present invention, various additives, fillers, and other kinds of polymers can be blended depending on the purpose, in addition to the thickener.
- the type of additive is not particularly limited, and various additives usually used for foam molding can be used.
- Light stabilizers such as systems, ultraviolet light absorbers, lubricants, crystal nucleating agents, fillers, flame retardants, flame retardant aids, mold release agents, antistatic agents, molecular modifiers such as peroxides, metal deactivators , Organic and inorganic nucleating agents, neutralizing agents, acid inhibitors, antibacterial agents, fluorescent whitening agents, organic and inorganic pigments and dyes, as well as for the purpose of imparting flame retardancy and thermal stability.
- thermoplastic polyester elastomer resin composition of the present invention preferably occupies 80% by mass or more, more preferably 90% by mass or more, and 95% by mass or more in total of the thermoplastic polyester elastomer and the thickener. It is more preferable to occupy.
- thermoplastic polyester elastomer resin composition of the present invention can be produced by mixing the above-mentioned components, various additives and fillers, and other kinds of polymers as necessary, and melt-kneading them.
- melt-kneading method any method known to those skilled in the art may be used, and a single-screw extruder, a twin-screw extruder, a pressure kneader, a Banbury mixer and the like can be used. Above all, it is preferable to use a twin-screw extruder.
- thermoplastic polyester elastomer used in the present invention, it is also possible to calculate from the proton integral ratio of 1 H-NMR measured by dissolving the sample in a solvent such as deuterated chloroform. ..
- the MFR (melt flow rate) of the thermoplastic polyester elastomer resin composition of the present invention is 5 to 30 g when measured at a load of 2,160 g and a measurement temperature of 230 ° C. according to the measurement method described in ASTM D1238. It is preferably / 10 min. Since the MFR is 5 to 30 g / 10 min, when the thermoplastic polyester elastomer resin composition is foam-molded, the bubble wall is less likely to be broken, and a foam-molded article having a high foaming ratio in which fine bubbles are formed is produced. Obtainable. It is particularly effective in foam molding using counter pressure, which will be described later.
- the MFR is lower than 5 g / 10 min, the fluidity tends to decrease and the foaming followability tends to decrease, and if it is higher than 30 g / 10 min, coarse bubbles tend to be generated and the foaming characteristics tend to deteriorate.
- thermoplastic polyester elastomer resin composition of the present invention contains a reactant obtained by reacting the thermoplastic polyester elastomer with the thickener, but the thermoplastic polyester elastomer not reacting with the thickener and the increase in release. It may contain a thickener. This is because it is difficult to completely react the thermoplastic polyester elastomer with the thickener.
- the relationship between the content of the thermoplastic polyester elastomer and the thickener described above is considered in terms of the mass derived from the thickener with respect to the mass derived from the thermoplastic polyester elastomer in the thermoplastic polyester elastomer resin composition. The relationship of this content is the same in the foam molded product described later.
- the foam molded product of the present invention is obtained by using the above-mentioned thermoplastic polyester elastomer resin composition, and is not subjected to any post-processing such as cutting. That is, it is a molded body that does not have a cutting surface.
- the cutting surface in this case means a surface obtained by cutting a non-foamed skin layer or the like from a foam molded body, and does not refer to a surface generated when unnecessary parts derived from the mold such as a gate portion are removed. not.
- the foamed molded product of the present invention is a surface layer consisting only of a foamed region having a bubble density of 10% or less and no non-foamed portion as a surface layer from the surface of the foamed molded product to a depth of 1000 ⁇ m (hereinafter, surface layer (hereinafter, surface layer)). It is a foamed molded product (A) composed of (may be abbreviated as A)), or a surface layer (A) in which a foamed region in which the non-foamed portion is present and a foamed region in which the non-foamed portion is not present coexist.
- the surface layer refers to a surface layer composed of a single surface, and for example, in the case of a rectangular parallelepiped foam molded product, it has six surface layers and refers to one of them.
- all six surfaces of the foam molded product (A) are composed of a surface layer (A)
- the foam molded product (B) is composed of at least one surface layer (B). ..
- the foamed molded product having a non-foamed skin layer produced by the method does not fall under the above-mentioned foamed molded product (B).
- the area of the foamed region in which the non-foamed portion is present is preferably 60% or less, more preferably 50% or less, still more preferably 30% or less, among the area of the single surface.
- the foamed molded product (A) is obtained. Further, in the foam molded product (B), the area of the foamed region where the non-foamed portion is present is preferably 50% or less, more preferably 40% or less, still more preferably 25% or less, based on the total surface area of the foamed molded product. In the foamed molded product (A), the impact resilience is improved because the non-foamed portion in which no bubbles do not exist is not present in the surface layer of the foamed molded product.
- the foamed region in which the non-foamed portion is present has a low rebound resilience because there are few bubbles, and the foamed region in which the non-foamed portion is not present has a high repulsive elasticity. It is possible to have different impact resilience depending on the location.
- the surface of the foam molded product of the present invention does not necessarily have to be a flat surface, and may be a curved surface or a surface having protrusions or the like. It is one of the points of the present invention that a foam molded product having an arbitrary desired shape can be obtained by preparing a corresponding mold.
- the above-mentioned non-foaming portion refers to a phase formed of a thermoplastic polyester elastomer resin composition with almost no bubbles, and is a portion having a bubble density of 10% or less.
- the bubble density is calculated by image processing a cross-sectional photograph of a sample for cross-section observation in the surface layer of the foamed molded product taken by a scanning electron microscope. Specifically, it is as described in the section of Examples.
- a portion that does not correspond to a non-foamed portion is referred to as a foamed portion or a foamed layer.
- the surface of the foamed portion is made of an extremely thin resin layer (so-called thin skin) formed by contacting the molten resin composition before foaming or the foamed molten resin composition with the surface of the mold, and the thickness thereof is 50 ⁇ m or less. be.
- the non-foaming portion having a bubble density of 10% or less is measured in a region of 200 ⁇ m ⁇ 200 ⁇ m, the thin resin layer on the surface of the foamed portion is “a non-foaming portion having a bubble density of 10% or less”. Does not apply to.
- the foamed molded product of the present invention does not have a sandwich structure in which a non-foamed layer is provided on both sides of the foamed layer (in other words, a structure in which the foamed layer is sandwiched between the non-foamed layers from both sides).
- the foam-molded article of the present invention is the foam-molded article (A) or the foam-molded article (B) described above.
- the foam molded body (A) is a foam molded body having no non-foaming layer and is composed of a single foam layer, and the foam molded body (B) is one of the surfaces of the foam molded body when the expression is changed.
- foam molded product in which a non-foaming portion (non-foaming skin layer) is present only in the portion.
- the size of the foamed molded product of the present invention is not particularly limited, and if a mold can be manufactured, a foamed molded product of a desired size can be obtained.
- a bubble having an aspect ratio of 2.0 or less is considered as a circular bubble, and a bubble having an aspect ratio of more than 2.0 is considered as a flat bubble.
- a flat cell layer having a large average aspect ratio of bubbles exists in the foamed region where the non-foamed portion of the surface layer does not exist. Since the surface of the foamed molded product becomes smooth and the appearance is excellent as the aspect ratio of the flat cell of the surface layer becomes large, the average aspect ratio of the flat cell layer is preferably 4.0 to 15.0. If it is less than 4.0, unevenness is generated on the surface of the molded body, and the surface smoothness is impaired.
- the surface smoothness tends to be impaired.
- the average aspect ratio of the flat cell layer is more preferably 4.0 to 10.0.
- a circular bubble layer having a small average aspect ratio of bubbles exists in the inner layer deeper than 1000 ⁇ m from the surface. Since the circular cell of the inner layer has a smaller aspect ratio and the impact resilience is improved, the average aspect ratio of the circular cell layer is preferably 1.0 to 2.0, and when it exceeds 2.0, the impact resilience is increased. It tends to decrease.
- the bubbles in the surface layer become flat bubbles because the resin composition flows along the surface of the mold during foam molding.
- the foamed molded product of the present invention preferably has a flat bubble layer having an average aspect ratio of bubbles of 4.0 to 15.0 in the foamed region where the non-foamed portion of the surface layer does not exist, and further, from the surface. It is a preferred embodiment to have a circular bubble layer having an average aspect ratio of bubbles of 1.0 to 2.0 in the inner layer deeper than 1000 ⁇ m.
- the foam layer is composed of a resin continuous phase and independent bubbles.
- the resin continuous phase means a portion formed of the cured thermoplastic polyester elastomer resin composition and having no cavity.
- the diameter (cell diameter) of the bubbles in the circular bubble layer of the inner layer deeper than 1000 ⁇ m from the surface described above is uniform and does not vary, the characteristics differ depending on the size.
- the cell diameter is small, and specifically, the average cell diameter is preferably 10 to 350 ⁇ m.
- the average cell diameter is less than 10 ⁇ m, the internal pressure of the molded product is low, the pressure at the time of forming the non-foamed skin layer is insufficient, and the appearance such as sink marks tends to be deteriorated.
- the average cell diameter is more preferably 50 to 350 ⁇ m, and further. It is preferably 100 to 300 ⁇ m, and particularly preferably 130 to 300 ⁇ m. Further, most of the circular bubble layer is composed of the above-mentioned fine bubbles having an average cell diameter of 10 to 350 ⁇ m, but also includes coarse bubbles having a slightly larger cell diameter.
- Coarse bubbles are generated when the molten resin is unstable, such as when the molten resin composition and the foaming agent are melt-mixed in a molding machine during the foam molding process, the melt viscosity of the molten resin composition is reduced in continuous molding. It is difficult to uniformly melt and mix the composition and the foaming agent, and it is presumed that the composition and the foaming agent are formed by the non-uniformly dispersed foaming agent.
- the maximum cell diameter of coarse bubbles is preferably 100 to 1000 ⁇ m. When the maximum cell diameter exceeds 1000 ⁇ m, the load bearing capacity is low and the elastic modulus tends to be low. The maximum cell diameter is more preferably 200 to 1000 ⁇ m, still more preferably 400 to 950 ⁇ m, in order to achieve the density for exhibiting high impact resilience, which will be described later.
- the density of the foam molded product of the present invention is preferably 0.01 to 0.70 g / cm 3 . Since the density of a general polyester elastomer is about 1.0 to 1.4 g / cm 3 , it can be said that the foam molded product of the present invention is sufficiently light in weight. It is more preferably 0.1 to 0.60 g / cm 3 , still more preferably 0.1 to 0.45 g / cm 3 , and particularly preferably 0.1 to 0.35 g / cm 3 . .. If the density is less than 0.01 g / cm 3 , sufficient strength cannot be obtained and the mechanical properties tend to be inferior, and if it exceeds 0.70 g / cm 3 , the impact resilience tends to be inferior.
- the average cell diameter is within a specific range and the density is within a specific range, and as a result, a high elastic modulus can be realized. Further, by appropriately selecting the above-mentioned configuration, a higher elastic modulus of impact of 50 to 90% can be realized. In order to exhibit high resilience characteristics, the elastic modulus of the foam molded product is preferably 60 to 90%.
- the foam molded product of the present invention is not only excellent in lightness, but also exhibits an extremely high elastic modulus and is also excellent in surface smoothness. Furthermore, to provide a polyester-based foamed molded product that can be applied to parts that require high reliability because it has a uniform foaming state despite a high foaming ratio, and has high heat resistance, water resistance, and molding stability. Can be done. Therefore, for example, it can be used for the following purposes. However, the use of the foam molded product of the present invention is not limited to the following uses. Examples of applications include automobile materials, civil engineering supplies, construction supplies, home appliances, OA equipment, sports supplies, stationery, toys, medical supplies, food containers, agricultural materials, and the like.
- Specific examples include automobile mechanical members, engine components, automobile exterior materials, automobile interior members, cushioning materials, sealing materials, car seats, deadening, door trims, sun visors, automobile vibration damping materials, sound absorbing materials, and heat insulating materials (heat insulating materials).
- anti-vibration material cushioning material, civil joint, anti-collapse panel, protective material, lightweight soil, filling, artificial soil, tatami core material, building insulation material, building joint material, face door material, building curing material, reflection Materials, industrial trays, tubes, pipe covers, air-conditioning insulation pipes, gasket cores, concrete molds, TVs, refrigerator-freezers, cooking equipment, washing machines, air-conditioning equipment, lighting equipment, computers, optomagnetic discs, copy machines, facsimiles , Printers, shoes, protectors, gloves, exercise equipment, etc.
- a foaming method of impregnating the thermoplastic polyester elastomer resin composition with a high-pressure gas and then reducing the pressure (releasing the pressure) is preferable.
- gas is injected into the cavity of the mold when the foaming agent and the thermoplastic polyester elastomer resin composition are melt-mixed and injection-molded.
- a foam injection molding method by a counter pressure method in which a thermoplastic polyester elastomer resin composition melted under a pressurized state is injected is preferable. Specifically, as shown in FIG.
- thermoplastic polyester elastomer resin composition a nitrogen gas for pressurization is injected into a cavity 3 formed of a plurality of molded molds 1 and 2 by using a counter pressure device 8, and a predetermined value is obtained.
- a pressure is applied to bring the thermoplastic polyester elastomer resin composition into a pressurized state, and the thermoplastic polyester elastomer resin composition in a molten state is used as a chemical foaming agent and / or an inert gas in a supercritical state (hereinafter, may be collectively referred to as “foaming agent”).
- the gas applied to the cavity is rapidly discharged from the electromagnetic valve 10 by counter pressure, whereby the thermoplastic polyester elastomer resin is started.
- the composition foams The predetermined pressure is preferably 0.01 MPa to 29.0 MPa.
- the pressure referred to here is a gauge pressure.
- the predetermined time is preferably 1 to 60 seconds.
- immediately before degassing the gas applied to the cavity by the rapid counter pressure described above at the same time as degassing, or immediately after degassing, or after degassing a predetermined time, one mold is used. By moving 2 in the mold opening direction to increase the volume of the cavity 3, it is also possible to combine it with a core back injection foam molding method for obtaining a foam molded product.
- thermoplastic polyester elastomer resin composition and the foaming agent can be mixed in the plasticization region 4a of the injection molding machine 4 before being filled in the cavity 3.
- foam molding is performed as described above, by appropriately adjusting the gas pressure of the counter pressure and the filling amount of the resin composition according to the material, a foam molded product having a desired foaming ratio and impact resilience can be obtained. ..
- the gas pressure of the counter pressure affects the fineness of bubbles and the foaming ratio, and by applying the gas pressure to the cavity even at a low pressure, the decompression rate is improved and high-magnification foaming becomes possible.
- the pressure of the counter pressure is preferably 0.01 MPa to 29.0 MPa, more preferably 0.05 MPa to 15.0 MPa, and further preferably 0.5 MPa to 10.0 MPa.
- the pressure referred to here is a gauge pressure.
- the filling amount of the resin composition is preferably 10% to 55%, more preferably 10% to 50%, still more preferably 10% to 40%, and particularly preferably 10% to 30% of the cavity volume. %.
- the reason why the foamed molded product of the present invention becomes the foamed molded product (A) or the foamed molded product (B) described above will be described.
- the schematic structure of the foam molded product (B) is shown in FIG.
- the foam molded product (A) has a structure in which there is no foamed region 25 in which the non-foamed portion 21 exists in the foamed molded product (B), and only the foamed region 24 in which the non-foamed portion 21 does not exist.
- the cavity of the mold pressurized by counter pressure is filled with the melted thermoplastic polyester elastomer resin composition together with the foaming agent within the range of 10% to 55% of the cavity volume.
- a non-foamed portion is formed by touching a part of the molten resin composition with the mold and cooling it. After that, by releasing the pressure in the cavity, foaming of the thermoplastic polyester elastomer resin composition occurs and a foamed molded product is obtained, but the non-foamed portion remains in a part of the surface layer of the foamed molded product and foams. A molded product (B) is obtained.
- the molten resin composition does not touch the mold at the time of filling, the area is very small even if it is touched, or the pressure in the cavity is released before it is sufficiently cooled even if it is touched.
- a foamed molded product (A) in which the foamed portion is not formed on the surface layer can be obtained.
- the thermoplastic polyester elastomer resin composition foams, it flows along the surface of the mold, so that the bubbles become flat cells.
- the chemical foaming agent that can be used to obtain the foamed molded product of the present invention is added to the resin composition melted in the resin melting zone of the molding machine as a gas component serving as a foaming nucleus or a source thereof. ..
- a gas component serving as a foaming nucleus or a source thereof.
- inorganic compounds such as ammonium carbonate, sodium bicarbonate and azide compounds, and organic compounds such as azo compounds, sulfohydrazide compounds and nitroso compounds can be used.
- the azide compound include terephthalazide and P-third butylbenz azide.
- examples of the azo compound include diazocarboxylic amide (ADCA), 2,2-azoisobutyronitrile, azohexahydrobenzonitrile, and diazoaminobenzene, and among them, ADCA is preferably used.
- examples of the sulfohydrazide compound include benzenesulfohydrazide, benzene1,3-disulfohydrazide, diphenylsulfone-3,3-disulfone hydrazide, diphenyloxide-4,4-disulfone hydrazide-, and the like.
- N, N-dinitrosopentaethylenetetramine (DNPT) and the like can be exemplified.
- the chemical foaming agent is based on a thermoplastic resin having a melting point lower than the decomposition temperature of the chemical foaming agent in order to be uniformly dispersed in the thermoplastic polyester elastomer resin composition. It can also be used as a foaming agent master batch.
- the base thermoplastic resin is not particularly limited as long as it has a melting point lower than the decomposition temperature of the chemical foaming agent, and examples thereof include polystyrene (PS), polyethylene (PE), polypropylene (PP), and the like.
- the blending ratio of the chemical foaming agent and the thermoplastic resin is preferably 10 to 100 parts by mass with respect to 100 parts by mass of the thermoplastic resin.
- the amount of the chemical foaming agent is less than 10 parts by mass, the amount of the masterbatch for the thermoplastic polyester elastomer resin composition may be too large to cause deterioration of the physical properties. If it exceeds 100 parts by mass, it becomes difficult to make a masterbatch due to the problem of dispersibility of the chemical foaming agent.
- carbon dioxide and / or nitrogen can be used as the inert gas.
- the amount thereof is preferably 0.05 to 30 parts by mass, preferably 0.1 to 20 parts by mass with respect to 100 parts by mass of the thermoplastic polyester elastomer resin composition. It is more preferably by mass. If the amount of carbon dioxide and / or nitrogen in the supercritical state is less than 0.05 parts by mass, it becomes difficult to obtain uniform and fine bubbles, and if it exceeds 30 parts by mass, the appearance of the surface of the molded body tends to be impaired.
- carbon dioxide or nitrogen in a supercritical state used as a foaming agent can be used alone, carbon dioxide and nitrogen may be mixed and used.
- nitrogen tends to be suitable for forming finer cells, and carbon dioxide can inject more gas and obtain higher foaming ratios. Since it is suitable, it may be arbitrarily mixed with respect to the state of the adjusted foam structure, and the mixing ratio in the case of mixing is preferably in the range of 1: 9 to 9: 1 in terms of molar ratio.
- foaming agent used in the present invention nitrogen in a supercritical state is more preferable from the viewpoint of uniform fine foaming.
- the molten thermoplastic polyester elastomer resin composition and the foaming agent are mixed in the plasticized region 4a of the injection molding machine 4. do it.
- carbon dioxide and / or nitrogen in a supercritical state is used as a foaming agent, for example, as shown in FIG. 1, carbon dioxide and / or nitrogen in a gaseous state is directly or pressurized by a booster pump 6 from a gas cylinder 5.
- a method of injecting into the injection molding machine 4 or the like can be adopted.
- These carbon dioxide and / or nitrogen need to be in a supercritical state inside the molding machine from the viewpoint of solubility, permeability, and diffusibility in the thermoplastic polyester elastomer resin composition in a molten state. ..
- the supercritical state can eliminate the distinction between the gas phase and the liquid phase in a certain temperature range and pressure range when raising the temperature and pressure of the substance producing the gas phase and the liquid phase.
- the state is called the critical temperature and the critical pressure at this time. That is, since a substance has both gas and liquid characteristics in a supercritical state, the fluid generated in this state is called a critical fluid. Since such a critical fluid has a higher density than a gas and a lower viscosity than a liquid, it has a characteristic that it is extremely easy to diffuse in a substance.
- Thermoplastic polyester elastomer (Polyester elastomer A) According to the method described in JP-A-9-59491, dimethyl terephthalate, 1,4-butanediol, and poly (tetramethylene oxide) glycol having a number average molecular weight of 2000 are used as raw materials, and the soft segment content is 76% by mass.
- the thermoplastic polyester elastomer of No. 1 was produced and used as Polyester Elastomer A.
- polyester elastomer B According to the method described in JP-A-9-59491, dimethyl terephthalate, 1,4-butanediol, and poly (tetramethylene oxide) glycol having a number average molecular weight of 2000 are used as raw materials, and the soft segment content is 83% by mass.
- the thermoplastic polyester elastomer of No. 1 was produced and used as Polyester Elastomer B.
- polyester elastomer C According to the method described in JP-A-9-59491, dimethyl terephthalate, 1,4-butanediol, and poly (tetramethylene oxide) glycol having a number average molecular weight of 1000 are used as raw materials, and the soft segment content is 67% by mass.
- the thermoplastic polyester elastomer of No. 1 was produced and used as Polyester Elastomer C.
- polyester elastomer D According to the method described in JP-A-9-59491, dimethyl terephthalate, 1,4-butanediol, and poly (tetramethylene oxide) glycol having a number average molecular weight of 1000 are used as raw materials, and the soft segment content is 55% by mass.
- the thermoplastic polyester elastomer of No. 1 was produced and used as Polyester Elastomer D.
- polyester elastomer E According to the method described in JP-A-9-59491, dimethyl terephthalate, 1,4-butanediol, and poly (tetramethylene oxide) glycol having a number average molecular weight of 1000 are used as raw materials, and the soft segment content is 28% by mass.
- the thermoplastic polyester elastomer of No. 1 was produced and used as Polyester Elastomer E.
- this styrene-based copolymer had a mass average molecular weight of 8500 and a number average molecular weight of 3300.
- the epoxy valence is 670 equivalents / 1 ⁇ 106 g, the epoxy valence (average number of epoxy groups per molecule) is 2.2, and one molecule has two or more glycidyl groups.
- TEPIC-S Commercially available epoxy compounds (Nissan Chemical Industries, Ltd.
- TPIC-S 1,3,5-Tris (2,3-epoxypropyl) -1,3,5-triazine-2,4,6 (1H, 3H, 5H) ) -Trion
- LA-1 A commercially available polycarbodiimide (“Carbodilite LA-1” manufactured by Nisshinbo Inc.) was prepared.
- thermoplastic Polyester Elastomer Resin Composition According to the compounding composition shown in Table 1, the thermoplastic polyester elastomer and the thickener were melt-kneaded using a twin-screw extruder and then pelletized to obtain pellets of the thermoplastic polyester elastomer resin composition. The physical characteristics of each resin composition were measured by the method described later and were as shown in Table 1.
- MFR Melt flow rate
- the MFR (melt flow rate) of the thermoplastic polyester elastomer resin composition was measured at a load of 2,160 g and a measurement temperature of 230 ° C. according to the measurement method described in ASTM D1238.
- a thermoplastic polyester elastomer resin composition dried (100 ° C., 3 to 5 hours) so as to have a water content of 0.1% by mass or less was used.
- thermoplastic polyester elastomer resin composition obtained above, a foam molded product was produced by the above-mentioned counter pressure method.
- a mold having a mold clamping force of 10000 kN and forming a cavity having a width of 360 mm, a length of 190 mm, and a thickness of 15.0 mm was used. Nitrogen gas at the pressure shown in Table 1 (counter pressure pressure) was injected into the cavity of this mold using counter pressure.
- thermoplastic polyester elastomer resin composition In the plasticized region of an electric injection molding machine having a screw with a screw diameter of 60 mm and a screw stroke of 300 mm, nitrogen in a supercritical state was injected into a molten thermoplastic polyester elastomer resin composition, and the temperature was adjusted to 50 ° C.
- the cavity of the mold was injection-filled with a short shot from the gate (the central portion of the surface having a width of 360 mm and a length of 190 mm) and the amount of the resin composition shown in Table 1 (the amount of the resin composition filled with respect to the cavity volume).
- the thermoplastic polyester elastomer resin composition was foamed by rapidly removing the nitrogen gas pressurized by counter pressure to obtain a foamed molded product.
- Comparative Example 3 A foam molded product was produced by a mold expansion method (core back injection foam molding method).
- a mold As a mold, a cavity with a width of 360 mm, a length of 190 mm, and a thickness of 3.0 mm can be formed when the mold is fastened, and when the core is backed in the mold opening direction, the width is the same, the length is the same, and the thickness is 3.0 mm + core back.
- a mold for producing a flat plate consisting of a fixing mold and an operating mold capable of forming a cavity having an amount (mm) was used.
- the foamed molded body is cut at the center surface (ww'plane of FIG. 3) of the foamed molded body, and the sample for cross-section observation is used. bottom. Further, the foam molded product was cut on a plane perpendicular to the cut plane (vv'plane in FIG. 3) to prepare a sample for cross-section observation.
- a cross-sectional photograph of a sample for cross-section observation in the surface layer of the foamed molded product was taken with a scanning electron microscope SU1510 manufactured by Hitachi High-Technologies Corporation.
- the cross-sectional photograph was image-processed, and the bubble density was calculated by the following formula in the region of 200 ⁇ m ⁇ 200 ⁇ m in the surface layer from the surface of the foamed molded body to the depth of 1000 ⁇ m, and the portion where the bubble density was 10% or less was not excluded. It was a foamed part.
- Bubble density (%) [total area of bubbles ( ⁇ m 2 ) / 40,000 ( ⁇ m 2 )] ⁇ 100
- the observation region of 200 ⁇ m ⁇ 200 ⁇ m is near the surface, around 500 ⁇ m, and around 1000 ⁇ m in the surface layer from the surface to a depth of 1000 ⁇ m (the surface of FIG. 2).
- the foamed molded body estimated to have no non-foamed portion at 5 points in each of the depths of the white squares in the layer 22) in the surface layer from the surface to a depth of 1000 ⁇ m, near the surface, at a depth of around 500 ⁇ m, and in depth.
- a foam molded body (B) having a surface layer in which non-foamed regions are mixed It is classified into three categories: a foam molded body (B) having a surface layer in which non-foamed regions are mixed, and a foam molded body (C) having a surface layer consisting only of a foamed region in which a non-foamed portion (non-foamed skin layer) is present. bottom.
- the foamed region in which the non-foamed portion is present is specified from the visual observation and the above cross-sectional photograph in the area of the surface of the foamed molded product, and the ratio of the area thereof. was calculated by the following equation.
- the foam molded product (B) has a plurality of surfaces corresponding to the surface layer (B)
- the one having a larger ratio is adopted.
- Percentage of the area of the foamed region where the non-foamed portion is present [Area of the foamed region where the non-foamed portion is present (mm 2 ) / Area of the surface of the foamed molded product (mm 2 )] ⁇ 100
- Ratio of the area of the foamed region where the non-foamed portion is present to the total surface area (%) [Area of the foamed region where the non-foamed portion is present (mm 2 ) / Surface area of the foamed molded product (mm 2 )] ⁇ 100
- a cross-sectional photograph of a sample for observing a cross-section of a foamed molded product was taken with a scanning electron microscope SU1510 manufactured by Hitachi High-Technologies Corporation. It was confirmed that the surface layer up to a depth of 1000 ⁇ m from the surface had a layer composed of flat bubbles, and the inner layer deeper than 1000 ⁇ m from the surface had a layer composed of circular bubbles.
- Cross-sectional photographs were image-processed and the major and minor axes of at least 100 adjacent flat and circular bubbles were measured with a caliper.
- the average aspect ratio (length of major axis / length of minor axis) of these 100 pieces was obtained, and this was performed at any three points of each layer, and the average value of the three average values obtained at the three points was averaged.
- the rebound resilience of the foamed region in which the non-foamed portion does not exist is defined as the repulsive elastic modulus (A)
- the repulsive elastic modulus of the foamed region in which the non-foamed portion exists is defined as the repulsive elastic modulus (B).
- the foamed molded product (A) composed of only the foamed region in which the non-foamed portion does not exist, or the foamed molded product (A).
- the foamed molded product (B) has a surface layer in which a foamed region having a non-foamed portion and a foamed region not having a non-foamed portion coexist. Since the average cell diameter and the maximum cell diameter of the bubbles have a predetermined size, they are lightweight and exhibit high impact resilience.
- the foam molded products (B) of Examples 2 to 6 and 8 to 11 and Comparative Examples 1 and 2 all have a non-foamed portion having a maximum area on the surface opposite to the surface having the gate and have a gate.
- the surface also had a non-foamed portion having almost the same area. Further, from Examples 2 to 6 and 8 to 11, it can be seen that the foamed region in which the non-foamed portion does not exist has a higher rebound resilience than the foamed region in which the non-foamed portion exists.
- Comparative Example 1 is a foam-molded polyester elastomer containing no thickener, and although it shows a high elastic modulus, the maximum cell diameter of the bubbles in the circular bubble layer becomes large. Compared with Examples 1 to 6 using the same polyester elastomer A, the impact modulus is lowered. In Comparative Example 2, the value is lower than the predetermined MFR, the fluidity is lowered, and the foam moldability is inferior, so that the mold followability is deteriorated.
- Comparative Example 3 is obtained by foam molding by the core back injection foam molding method, and although the weight can be sufficiently reduced, the entire foam molded body is composed of a sandwich structure of a non-foam layer and a foam layer. In addition, this causes irregularities on the entire surface of the molded product, resulting in poor surface smoothness.
- the foam molded product of the present invention is not only excellent in lightness, but also exhibits an extremely high elastic modulus and is also excellent in surface smoothness. Furthermore, to provide a polyester foam molded product that can be applied to parts that require high reliability because it has a uniform foaming state despite a high foaming ratio, and has high heat resistance, water resistance, and molding stability. Can be done.
- Mold (for fixing) 2 Mold (for operation) 3 Cavity 4 Injection molding machine 4a Plasticization area 5 Gas cylinder 6 Boost pump 7 Pressure control valve 8 Counter pressure device 9 Solenoid valve for intake 10 Solenoid valve for exhaust 21 Non-foaming part 22 Surface layer 23 Inner layer 24 Foaming without non-foaming part Region 25 Foamed region with non-foamed portion
Abstract
Description
(1) 芳香族ジカルボン酸と脂肪族及び/又は脂環族ジオールとを構成成分とするポリエステルからなるハードセグメントと、脂肪族ポリエーテル、脂肪族ポリエステル、及び脂肪族ポリカーボネートから選ばれる少なくとも1種のソフトセグメントが結合され、ソフトセグメントの含有量が25~90質量%である熱可塑性ポリエステルエラストマー100質量部に対して、増粘剤0.05~4.5質量部の割合で含有することを特徴とするカウンタープレッシャーを用いた発泡成形用熱可塑性ポリエステルエラストマー樹脂組成物。
(2) 増粘剤がエポキシ基を有する反応性化合物であることを特徴とする、(1)に記載の熱可塑性ポリエステルエラストマー樹脂組成物。
(3) 荷重2,160g、測定温度230℃におけるMFRが、5~30g/10minであることを特徴とする、(1)または(2)に記載の熱可塑性ポリエステルエラストマー樹脂組成物。
(4) (1)~(3)のいずれかに記載の熱可塑性ポリエステルエラストマー樹脂組成物が連続相をなす発泡成形体であり、該発泡成形体の表面から1000μmの深さまでの表面層として、気泡密度が10%以下である非発泡部分が存在しない発泡領域のみからなる表面層で構成される発泡成形体であるか、もしくは、前記非発泡部分が存在する発泡領域と前記非発泡部分が存在しない発泡領域が混在する表面層を有する発泡成形体であり、密度が0.01~0.70g/cm3である発泡成形体。
(5) 前記表面層の非発泡部分が存在しない発泡領域に、気泡の平均アスペクト比が4.0~15.0である扁平気泡層を有することを特徴とする(4)に記載の発泡成形体。
(6) さらに、表面から1000μmより深い内層に気泡の平均アスペクト比が1.0~2.0である円形気泡層を有することを特徴とする(4)または(5)に記載の発泡成形体。
(7) 反発弾性率が50~90%であることを特徴とする、(4)~(6)のいずれかに記載の発泡成形体。
(8) 内層の円形気泡層の気泡における、平均セル径が10~350μmであり、最大セル径が100~1000μmである、(6)または(7)に記載の発泡成形体。
本発明で使用する熱可塑性ポリエステルエラストマーは、ハードセグメントとソフトセグメントが結合してなる。ハードセグメントは、ポリエステルからなる。ハードセグメントのポリエステルを構成する芳香族ジカルボン酸としては、通常の芳香族ジカルボン酸が広く用いられ、特に限定されないが、主たる芳香族ジカルボン酸としては、テレフタル酸又はナフタレンジカルボン酸(異性体の中では2,6-ナフタレンジカルボン酸が好ましい)であることが望ましい。これらの芳香族ジカルボン酸の含有量は、ハードセグメントのポリエステルを構成する全ジカルボン酸中、70モル%以上であることが好ましく、80モル%以上であることがより好ましい。その他のジカルボン酸成分としては、ジフェニルジカルボン酸、イソフタル酸、5-ナトリウムスルホイソフタル酸などの芳香族ジカルボン酸、シクロヘキサンジカルボン酸、テトラヒドロ無水フタル酸などの脂環族ジカルボン酸、コハク酸、グルタル酸、アジピン酸、アゼライン酸、セバシン酸、ドデカン二酸、ダイマー酸、水添ダイマー酸などの脂肪族ジカルボン酸などが挙げられる。これらは、樹脂の融点を大きく低下させない範囲で用いられることができ、その量は全酸成分の30モル%以下が好ましく、より好ましくは20モル%以下である。
本発明における増粘剤は、熱可塑性ポリエステルエラストマーの持つヒドロキシル基あるいはカルボキシル基と反応し得る官能基を有する反応性化合物(以下、単に反応性化合物と称することがある)であり、反応し得る官能基としては、エポキシ基(グリシジル基)、酸無水物基、カルボジイミド基およびイソシアネート基から選ばれる少なくとも一種であることが好ましく、該官能基は1分子あたり2個以上含有する。該官能基は、エポキシ基(グリシジル基)がより好ましい。本発明における増粘剤は、エポキシ基(グリシジル基)を有する反応性化合物、カルボジイミド基を有する反応性化合物であることが好ましく、エポキシ基(グリシジル基)を有する反応性化合物であることがより好ましい。以下、例えば、「エポキシ基(グリシジル基)を有する反応性化合物」は、「エポキシ基を持つ化合物」のように称することもある。増粘剤は、熱可塑性ポリエステルエラストマーのポリマー鎖と反応して結合することから、架橋剤の働きもする。
増粘剤を含有する場合、0.05質量部未満であると、目標とした分子鎖延長効果が不十分であり、4.5質量部を超えると、増粘効果が過剰となり成形性に悪影響を与えたり、成形品の機械的特性に影響を与える傾向がある。増粘剤がエポキシ化合物の場合、4.5質量部を超えると、エポキシ化合物の凝集硬化によって成形品表面に凸凹が生じることがある。増粘剤がカルボジイミド化合物の場合、4.5質量部を超えると、ポリカルボジイミド化合物の塩基性により熱可塑性ポリエステルエラストマーの加水分解が生じ機械的特性に影響を与える傾向がある。
さらに、本発明に用いる熱可塑性ポリエステルエラストマーには、上記増粘剤以外にも、目的に応じて種々の添加剤やフィラー、他種ポリマーを配合することができる。添加剤の種類は特に限定されず、発泡成形に通常使用される各種添加剤を用いることができる。具体的には、添加剤として、公知のヒンダードフェノール系、硫黄系、燐系、アミン系の酸化防止剤、ヒンダードアミン系のほかベンゾトリアゾール系、ベンゾフェノン系、ベンゾエート系、トリアゾール系、ニッケル系、サリチル系等の光安定剤、紫外光吸収剤、滑剤、結晶核剤、充填剤、難燃剤、難燃助剤、離型剤、帯電防止剤、過酸化物等の分子調整剤、金属不活性剤、有機および無機系の核剤、中和剤、制酸剤、防菌剤、蛍光増白剤、有機および無機系の顔料や染料のほか、難燃性付与や熱安定性付与の目的で使用される有機および無機系の燐化合物などが挙げられる。添加剤やフィラー、他種ポリマーの配合量(含有量)は、気泡の形成等を損なわない範囲で適宜選択することができ、通常の熱可塑性樹脂の成形に用いられる配合量(含有量)を採用することができる。本発明の熱可塑性ポリエステルエラストマー樹脂組成物としては、熱可塑性ポリエステルエラストマーと増粘剤の合計で、80質量%以上を占めることが好ましく、90質量%以上を占めることがより好ましく、95質量%以上を占めることがさらに好ましい。
本発明の発泡成形体は、前述した熱可塑性ポリエステルエラストマー樹脂組成物を用いて得られたものであり、切削などの後加工は一切していないものになる。すわなち、切削面を有さない成形体である。この場合の切削面とは、発泡成形体から非発泡スキン層等を切削した面を意味するものであり、ゲート部等の金型由来の不要個所を除いた際に発生する面を指すものではない。
本発明の発泡成形体は、表面層の非発泡部分が存在しない発泡領域に、気泡の平均アスペクト比が大きい扁平気泡層が存在していることが好ましい。表面層の扁平気泡は、アスペクト比が大きくなることで発泡成形体表面が平滑となり外観に優れるため、扁平気泡層の平均アスペクト比は4.0~15.0であることが好ましい。4.0を下回ると、成形体表面に凹凸が発生し、表面平滑性が損なわれ、15.0を上回ると、気泡が引き延ばされることで破泡を起こすことで成形体表面に凹凸が発生し、表面平滑性が損なわれる傾向にある。優れた表面平滑性を有するためには、扁平気泡層の平均アスペクト比は、4.0~10.0であることがより好ましい。さらに、表面から1000μmより深い内層に気泡の平均アスペクト比が小さい円形気泡層が存在していることが好ましい。内層の円形気泡は、アスペクト比が小さくなることで反発弾性が向上するため、円形気泡層の平均アスペクト比は1.0~2.0であることが好ましく、2.0を上回ると反発弾性が低下する傾向にある。表面層の気泡が、扁平気泡となるのは、発泡成形時に樹脂組成物が金型の面に沿って流動するからである。本発明の発泡成形体としては、表面層の非発泡部分が存在しない発泡領域に、気泡の平均アスペクト比が4.0~15.0である扁平気泡層を有することが好ましく、さらに、表面から1000μmより深い内層に気泡の平均アスペクト比が1.0~2.0である円形気泡層を有することが好ましい態様である。
用途としては、自動車用材料、土木用品、建築用品、家電、OA機器、スポーツ用品、文具、玩具、医療用品、食品容器、農業用資材などが挙げられる。具体例としては、自動車用機構部材、エンジン構成材、自動車外装材、自動車内装部材、クッション材、シール材カーシート、デッドニング、ドアトリム、サンバイザー、自動車用制振材・吸音材・断熱材(保温材)、防振材、緩衝材、土木目地、つらら防止パネル、保護材、軽量土、盛土、人工土壌、畳芯材、建築用断熱材、建築目地材、面戸材、建築養生材、反射材、工業用トレー、チューブ、パイプカバー、エアコン断熱配管、ガスケット芯材、コンクリート型枠、テレビ、冷凍冷蔵庫、調理機器、洗濯機、空調機器、照明器具、コンピュータ、光磁気ディスク、コピー機、ファクシミリ、プリンター、シューズ、プロテクター、グローブ、運動用具類などが挙げられる。
本発明の発泡成形体の発泡方法については、熱可塑性ポリエステルエラストマー樹脂組成物に高圧のガスを含浸させた後、減圧する(圧力を解放する)発泡方法が好ましい。なかでも、成形サイクル性やコスト、均質発泡を得られる成形方法として、発泡剤と熱可塑性ポリエステルエラストマー樹脂組成物を溶融混合して射出成形する際に、金型のキャビティ内にガスを注入し、加圧状態下に溶融した熱可塑性ポリエステルエラストマー樹脂組成物を射出するカウンタープレッシャー法による発泡射出成形方法が好ましい。具体的には、図1に示すように、型締めされた複数の金型1、2で形成されるキャビティ3内に、カウンタープレッシャー装置8を用いて加圧用の窒素ガスを注入し、所定の圧力を掛けて加圧状態にし、そこへ溶融状態の熱可塑性ポリエステルエラストマー樹脂組成物を化学発泡剤および/または超臨界状態の不活性ガス(以下、まとめて「発泡剤」と称することもある)とともに射出を開始し、ついで樹脂組成物の充填が完了した直後、ないし所定の時間をおいてから、急速にカウンタープレッシャーでキャビティに掛けたガスを電磁バルブ10から抜くことで、熱可塑性ポリエステルエラストマー樹脂組成物が発泡するものである。前記の所定の圧力としては、0.01MPa~29.0MPaが好ましい。ここで言う圧力は、ゲージ圧である。前記の所定の時間としては、1~60秒が好ましい。ここで、前述した急速にカウンタープレッシャーでキャビティに掛けたガスを抜く直前、もしくはガスを抜くと同時、もしくはガスを抜いた直後、もしくはガスを抜いて所定の時間をおいてから、一つの金型2を型開き方向へ移動してキャビティ3の容積を拡大させることにより、発泡成形体を得るコアバック射出発泡成形法と組み合わせることも可能である。
具体的には、化学発泡剤としては、炭酸アンモニウム及び重炭素酸ソーダ、アジド化合物等の無機化合物、並びにアゾ化合物、スルホヒドラジド化合物、ニトロソ化合物等の有機化合物等が使用できる。上記アジド化合物としては、テレフタルアジド及びP-第三ブチルベンズアジド等が例示できる。さらに上記アゾ化合物としては、ジアゾカルボンアミド(ADCA)、2,2-アゾイソブチロニトリル、アゾヘキサヒドロベンゾニトリル、及びジアゾアミノベンゼン等が例示でき、中でもADCAが好まれて活用されている。上記スルホヒドラジド化合物としては、ベンゼンスルホヒドラジド、ベンゼン1,3-ジスルホヒドラジド、ジフェニルスルホン-3,3-ジスルホンヒドラジド及びジフェニルオキシド-4,4-ジスルホンヒドラジド-等が例示でき、上記ニトロソ化合物としては、N,N-ジニトロソペンタエチレンテトラミン(DNPT)等が例示できる。
熱可塑性ポリエステルエラストマー;
(ポリエステルエラストマーA)
特開平9-59491号公報に記載の方法に準じて、ジメチルテレフタレート、1,4-ブタンジオール、及び数平均分子量2000のポリ(テトラメチレンオキシド)グリコールを原料として、ソフトセグメント含有量が76質量%の熱可塑性ポリエステルエラストマーを製造して、これをポリエステルエラストマーAとした。
(ポリエステルエラストマーB)
特開平9-59491号公報に記載の方法に準じて、ジメチルテレフタレート、1,4-ブタンジオール、及び数平均分子量2000のポリ(テトラメチレンオキシド)グリコールを原料として、ソフトセグメント含有量が83質量%の熱可塑性ポリエステルエラストマーを製造して、これをポリエステルエラストマーBとした。
(ポリエステルエラストマーC)
特開平9-59491号公報に記載の方法に準じて、ジメチルテレフタレート、1,4-ブタンジオール、及び数平均分子量1000のポリ(テトラメチレンオキシド)グリコールを原料として、ソフトセグメント含有量が67質量%の熱可塑性ポリエステルエラストマーを製造して、これをポリエステルエラストマーCとした。
(ポリエステルエラストマーD)
特開平9-59491号公報に記載の方法に準じて、ジメチルテレフタレート、1,4-ブタンジオール、及び数平均分子量1000のポリ(テトラメチレンオキシド)グリコールを原料として、ソフトセグメント含有量が55質量%の熱可塑性ポリエステルエラストマーを製造して、これをポリエステルエラストマーDとした。
(ポリエステルエラストマーE)
特開平9-59491号公報に記載の方法に準じて、ジメチルテレフタレート、1,4-ブタンジオール、及び数平均分子量1000のポリ(テトラメチレンオキシド)グリコールを原料として、ソフトセグメント含有量が28質量%の熱可塑性ポリエステルエラストマーを製造して、これをポリエステルエラストマーEとした。
(エポキシ基を持つ化合物:スチレン系共重合体)
オイルジャケットを備えた容量1リットルの加圧式攪拌槽型反応器のオイルジャケット温度を、200℃に保った。一方、スチレン(St)89質量部、グリシジルメタクリレート(GMA)11質量部、キシレン(Xy)15質量部及び重合開始剤としてジターシャリーブチルパーオキサイド(DTBP)0.5質量部からなる単量体混合液を原料タンクに仕込んだ。これを一定の供給速度(48g/分、滞留時間:12分)で原料タンクから反応器に連続供給し、反応器の内容液質量が約580gで一定になるように反応液を反応器の出口から連続的に抜き出した。その時の反応器内温は、約210℃に保った。反応器内部の温度が安定してから36分経過後から、抜き出した反応液を減圧度30kPa、温度250℃に保った薄膜蒸発機に導き、連続的に揮発成分を除去して、スチレン系共重合体を得た。このスチレン系共重合体は、GPC分析(ポリスチレン換算値)によると質量平均分子量8500、数平均分子量3300であった。また、エポキシ価は670当量/1×106g、エポキシ価数(1分子当りの平均エポキシ基の数)は2.2であり、グリシジル基を1分子中に2個以上有するものである。
(エポキシ基を持つ化合物:TEPIC-S)
市販のエポキシ化合物(日産化学社製「TEPIC-S」、1,3,5-トリス(2,3-エポキシプロピル)-1,3,5-トリアジン-2,4,6(1H,3H,5H)-トリオン)を用意した。
(カルボジイミド基を持つ化合物:LA-1)
市販のポリカルボジイミド(日清紡社製「カルボジライト LA-1」)を用意した。
表1に記載の配合組成に従って、熱可塑性ポリエステルエラストマーと増粘剤を、二軸スクリュー式押出機を用いて溶融混練した後、ペレット化して熱可塑性ポリエステルエラストマー樹脂組成物のペレットを得た。各樹脂組成物の物性について、後述する方法で測定したところ表1に示す通りであった。
熱可塑性ポリエステルエラストマー樹脂組成物のMFR(メルトフローレート)は、ASTM D1238に記載されている測定法に準じて、荷重2,160g、測定温度230℃にて測定を実施した。測定は、水分率が0.1質量%以下になるように乾燥(100℃、3~5時間)した熱可塑性ポリエステルエラストマー樹脂組成物を用いた。
次に、上記で得られた熱可塑性ポリエステルエラストマー樹脂組成物を用いて上述したカウンタープレッシャー法にて発泡成形体を作製した。金型としては、型締め力が10000kNであり、型締めすると幅360mm、長さ190mm、厚み15.0mmのキャビティを形成するものを用いた。この金型のキャビティ内に、カウンタープレッシャーを用いて表1に記載の圧力(カウンタープレッシャー圧力)の窒素ガスを注入した。スクリュー径60mm、スクリューストローク300mmのスクリューを持つ電動射出成形機の可塑化領域で、溶融状態の熱可塑性ポリエステルエラストマー樹脂組成物に超臨界状態とした窒素を注入し、表面温度50℃に温調した前記の金型のキャビティに、ゲート(幅360mm、長さ190mmの面の中央部)から、表1に記載の樹脂組成物量(キャビティ体積に対する樹脂組成物充填量)をショートショットで射出充填した。充填した直後、急速にカウンタープレッシャーで加圧した窒素ガスを抜くことで、熱可塑性ポリエステルエラストマー樹脂組成物を発泡させて発泡成形体を得た。
金型拡張法(コアバック射出発泡成形法)にて、発泡成形体を作製した。金型としては、型締めすると幅360mm、長さ190mm、厚み3.0mmのキャビティを形成することができ、型開き方向へコアバックさせると同幅、同長さで厚みが3.0mm+コアバック量(mm)であるキャビティを形成することができる固定用金型および稼働用金型からなる平板作製用の金型を用いた。具体的には、金型の型締め力が10000kN、スクリュー径60mm、スクリューストローク300mmのスクリューを持つ電動射出成形機の可塑化領域で、超臨界状態とした窒素を注入し、表面温度50℃に温調された金型にフルパックで射出充填後、射出外圧と内部からの発泡圧力によって700μm程度の非発泡スキン層が形成された段階で、稼働用金型を型開き方向へ、12.0mm移動させて、キャビティの容積を拡大させて、発泡成形体を得た。
発泡成形体の表面を目視で観察し、成形収縮による凸凹部(ヒケ)の有無やアバタの発生から非発泡部分の存在があるかどうかを推定した。凸凹部及び/又はアバタがあり非発泡部分が存在すると推定した発泡成形体の場合、非発泡部分が中心にくる面(図2のw-w’面)で発泡成形体を切って、断面観察用サンプルとした。さらにその切断面と垂直な面(図2のv-v’面)で発泡成形体を切って、断面観察用サンプルとした。凸凹部及びアバタがなく非発泡部分が存在しないと推定した発泡成形体の場合、発泡成形体の中心面(図3のw-w’面)で発泡成形体を切って、断面観察用サンプルとした。さらにその切断面と垂直な面(図3のv-v’面)で発泡成形体を切って、断面観察用サンプルとした。
日立ハイテクノロジーズ製の走査電子顕微鏡SU1510により、発泡成形体の表面層における断面観察用サンプルの断面写真を撮影した。断面写真を画像処理し、発泡成形体の表面から1000μmの深さまでの表面層において、200μm×200μmの領域にて、次式により気泡密度を算出し、気泡密度が10%以下となる部分を非発泡部分とした。
気泡密度(%)=[気泡の合計面積(μm2)/40,000(μm2)]×100
200μm×200μmの観察領域は、非発泡部分が存在すると推定した発泡成形体の場合、表面から1000μmの深さまでの表面層において、表面付近、深さ500μm付近、深さ1000μm付近(図2の表面層22にある白い四角の深さ)でそれぞれ5ヶ所、非発泡部分が存在しないと推定した発泡成形体の場合、表面から1000μmの深さまでの表面層において、表面付近、深さ500μm付近、深さ1000μm付近(図3の表面層22にある白い四角の深さ)でそれぞれ5ヶ所を測定した。この測定で表面から深さ方向の3ヶ所全てが、非発泡部分が存在しないと判断できた場合、その領域は非発泡部分が存在しない発泡領域のみからなる領域であるとした。
これにより、非発泡部分の有無を判断し、非発泡部分が存在しない発泡領域のみからなる表面層で構成される発泡成形体(A)、非発泡部分が存在する発泡領域と非発泡部分が存在しない発泡領域が混在する表面層を有する発泡成形体(B)、及び非発泡部分(非発泡スキン層)が存在する発泡領域のみからなる表面層を有する発泡成形体(C)の3つに分類した。
発泡成形体(B)の表面層(B)において、発泡成形体の表面の面積の内、目視による観察や上記の断面写真から、非発泡部分が存在する発泡領域を特定し、その面積の割合を、次式により算出した。発泡成形体(B)に表面層(B)に相当する面が複数存在する場合は、この割合が大きい方を採用する。
非発泡部分が存在する発泡領域の面積の割合(%)=[非発泡部分が存在する発泡領域の面積(mm2)/発泡成形体の表面の面積(mm2)]×100
上記「非発泡部分が存在する発泡領域の面積の割合」の方法と同様に、「非発泡部分が存在する発泡領域の面積」を特定した。発泡成形体(B)に表面層(B)に相当する面が複数存在する場合は、全ての面の「非発泡部分が存在する発泡領域の面積」を合計した。次式により、発泡成形体(B)の全表面積に対する非発泡部分が存在する発泡領域の面積の割合を算出した。
全表面積に対する非発泡部分が存在する発泡領域の面積の割合(%)=[非発泡部分が存在する発泡領域の面積(mm2)/発泡成形体の表面積(mm2)]×100
日立ハイテクノロジーズ製の走査電子顕微鏡SU1510により、発泡成形体の断面観察用サンプルの断面写真を撮影した。表面から1000μmの深さまでの表面層に扁平気泡からなる層があること、表面から1000μmより深い内層に円形気泡からなる層があることを確認した。断面写真を画像処理し、少なくとも100個の隣接する扁平気泡及び円形気泡の長軸と短軸をノギスで測定した。それら100個の平均アスペクト比(長軸の長さ/短軸の長さ)を求め、これをそれぞれの層の任意の三箇所において行い、三箇所で得られた3つの平均値を平均した値を、それぞれ扁平気泡層及び円形気泡層の平均アスペクト比とした。
発泡成形体の寸法をノギスで測定し、その質量を電子天秤にて測定し、次式により算出した。
密度(g/cm3)=発泡成形体の質量/発泡成形体の体積
上記円形気泡層の気泡の平均アスペクト比の測定の際に、円形気泡の面積円相当径をセル径とし、それら100個の平均値を求めた。これを円形気泡層の任意の三箇所において行い、三箇所で得られた3つの平均値を平均した値を平均セル径とした。
なお、上記の観察した気泡のセル径の中で最大のセル径を最大セル径とした。
JIS K 6400に記載されている方法にて測定を実施した。手動計測試験機を用い、規定の落下高さ(H)から発泡成形体に鋼球を落下させ、跳ね返った最大の高さ(h)を読み取り、以下の計算式にて反発弾性率を算出した。
反発弾性率(%)=(跳ね返った最大高さ(h)/落下高さ(H))×100
一分間以内に3回の測定を行い、その中央値を求め、反発弾性率を算出した。非発泡部分が存在しない発泡領域の反発弾性率を反発弾性率(A)、非発泡部分が存在する発泡領域の反発弾性率を反発弾性率(B)とした。
発泡成形体の表面平滑性を目視にて、以下のように3段階の評価を行った。
〇:成形体表面に凹凸が認められない
△:成形体表面の一部に凹凸が認められる
×:成形体表面の全体に凹凸が認められる
金型キャビティ形状である幅360mm、長さ190mm、厚み15.0mmの寸法において、発泡後の成形体厚みが、14.0mm以上であれば金型に追従できているとして「〇」、14.0mm未満であれば金型に追従できていないとして「×」とした。
2 金型(稼働用)
3 キャビティ
4 射出成形機
4a 可塑化領域
5 ガスボンベ
6 昇圧ポンプ
7 圧力制御バルブ
8 カウンタープレッシャー装置
9 吸気用電磁バルブ
10 排気用電磁バルブ
21 非発泡部分
22 表面層
23 内層
24 非発泡部分が存在しない発泡領域
25 非発泡部分が存在する発泡領域
Claims (8)
- 芳香族ジカルボン酸と脂肪族及び/又は脂環族ジオールとを構成成分とするポリエステルからなるハードセグメントと、脂肪族ポリエーテル、脂肪族ポリエステル、及び脂肪族ポリカーボネートから選ばれる少なくとも1種のソフトセグメントが結合され、ソフトセグメントの含有量が25~90質量%である熱可塑性ポリエステルエラストマー100質量部に対して、増粘剤0.05~4.5質量部の割合で含有することを特徴とするカウンタープレッシャーを用いた発泡成形用熱可塑性ポリエステルエラストマー樹脂組成物。
- 増粘剤がエポキシ基を有する反応性化合物であることを特徴とする、請求項1に記載の熱可塑性ポリエステルエラストマー樹脂組成物。
- 荷重2,160g、測定温度230℃におけるMFRが、5~30g/10minであることを特徴とする、請求項1または2に記載の熱可塑性ポリエステルエラストマー樹脂組成物。
- 請求項1~3のいずれかに記載の熱可塑性ポリエステルエラストマー樹脂組成物が連続相をなす発泡成形体であり、該発泡成形体の表面から1000μmの深さまでの表面層として、気泡密度が10%以下である非発泡部分が存在しない発泡領域のみからなる表面層で構成される発泡成形体であるか、もしくは、前記非発泡部分が存在する発泡領域と前記非発泡部分が存在しない発泡領域が混在する表面層を有する発泡成形体であり、密度が0.01~0.70g/cm3である発泡成形体。
- 前記表面層の非発泡部分が存在しない発泡領域に、気泡の平均アスペクト比が4.0~15.0である扁平気泡層を有することを特徴とする請求項4に記載の発泡成形体。
- さらに、表面から1000μmより深い内層に気泡の平均アスペクト比が1.0~2.0である円形気泡層を有することを特徴とする請求項4または5に記載の発泡成形体。
- 反発弾性率が50~90%であることを特徴とする、請求項4~6のいずれかに記載の発泡成形体。
- 内層の円形気泡層の気泡における、平均セル径が10~350μmであり、最大セル径が100~1000μmである、請求項6または7に記載の発泡成形体。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202180071645.2A CN116583395A (zh) | 2020-10-20 | 2021-10-15 | 聚酯弹性体树脂组合物以及发泡成形体 |
| EP21882734.3A EP4234202A1 (en) | 2020-10-20 | 2021-10-15 | Polyester elastomer resin composition and foam molded body |
| US18/032,416 US20230383118A1 (en) | 2020-10-20 | 2021-10-15 | Polyester elastomer resin composition and foam molded product |
| JP2022515586A JPWO2022085593A1 (ja) | 2020-10-20 | 2021-10-15 |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020176133 | 2020-10-20 | ||
| JP2020-176133 | 2020-10-20 | ||
| JP2021-074976 | 2021-04-27 | ||
| JP2021074976A JP7088362B1 (ja) | 2020-10-20 | 2021-04-27 | 発泡成形体およびその製造方法 |
| JP2021-115867 | 2021-07-13 | ||
| JP2021115867 | 2021-07-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022085593A1 true WO2022085593A1 (ja) | 2022-04-28 |
Family
ID=81290460
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2021/038277 WO2022085593A1 (ja) | 2020-10-20 | 2021-10-15 | ポリエステルエラストマー樹脂組成物及び発泡成形体 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20230383118A1 (ja) |
| EP (1) | EP4234202A1 (ja) |
| JP (1) | JPWO2022085593A1 (ja) |
| TW (1) | TW202222938A (ja) |
| WO (1) | WO2022085593A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023037957A1 (ja) * | 2021-09-08 | 2023-03-16 | 東洋紡株式会社 | 熱可塑性ポリエステルエラストマー樹脂組成物及び発泡成形体 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH059491A (ja) | 1991-06-28 | 1993-01-19 | Cosmo Sogo Kenkyusho:Kk | 塑性加工油組成物 |
| JPH06506724A (ja) * | 1991-04-05 | 1994-07-28 | マサチユーセツツ・インステイテユート・オブ・テクノロジー | 超微孔質発泡材料 |
| JP2003138054A (ja) * | 2001-11-05 | 2003-05-14 | Toray Ind Inc | 液晶ポリエステル樹脂発泡成形品 |
| JP2003342343A (ja) | 2002-05-29 | 2003-12-03 | Inoac Corp | 高反発弾性フォーム |
| JP2011068819A (ja) | 2009-09-28 | 2011-04-07 | Japan Polypropylene Corp | ポリプロピレン系射出発泡成形体及びその製造方法 |
| JP2013032492A (ja) * | 2011-07-05 | 2013-02-14 | Nitto Denko Corp | ポリエステル系エラストマー発泡体及び発泡部材 |
| JP6358369B1 (ja) | 2017-06-27 | 2018-07-18 | 東洋紡株式会社 | 熱可塑性ポリエステルエラストマー樹脂発泡成形体およびその製造方法 |
| JP2019006926A (ja) * | 2017-06-27 | 2019-01-17 | 東洋紡株式会社 | 熱可塑性ポリエステルエラストマー樹脂組成物およびその発泡成形体 |
-
2021
- 2021-10-15 EP EP21882734.3A patent/EP4234202A1/en active Pending
- 2021-10-15 JP JP2022515586A patent/JPWO2022085593A1/ja active Pending
- 2021-10-15 WO PCT/JP2021/038277 patent/WO2022085593A1/ja active Application Filing
- 2021-10-15 US US18/032,416 patent/US20230383118A1/en active Pending
- 2021-10-18 TW TW110138462A patent/TW202222938A/zh unknown
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06506724A (ja) * | 1991-04-05 | 1994-07-28 | マサチユーセツツ・インステイテユート・オブ・テクノロジー | 超微孔質発泡材料 |
| JPH059491A (ja) | 1991-06-28 | 1993-01-19 | Cosmo Sogo Kenkyusho:Kk | 塑性加工油組成物 |
| JP2003138054A (ja) * | 2001-11-05 | 2003-05-14 | Toray Ind Inc | 液晶ポリエステル樹脂発泡成形品 |
| JP2003342343A (ja) | 2002-05-29 | 2003-12-03 | Inoac Corp | 高反発弾性フォーム |
| JP2011068819A (ja) | 2009-09-28 | 2011-04-07 | Japan Polypropylene Corp | ポリプロピレン系射出発泡成形体及びその製造方法 |
| JP2013032492A (ja) * | 2011-07-05 | 2013-02-14 | Nitto Denko Corp | ポリエステル系エラストマー発泡体及び発泡部材 |
| JP6358369B1 (ja) | 2017-06-27 | 2018-07-18 | 東洋紡株式会社 | 熱可塑性ポリエステルエラストマー樹脂発泡成形体およびその製造方法 |
| JP2019006926A (ja) * | 2017-06-27 | 2019-01-17 | 東洋紡株式会社 | 熱可塑性ポリエステルエラストマー樹脂組成物およびその発泡成形体 |
| JP2019006927A (ja) * | 2017-06-27 | 2019-01-17 | 東洋紡株式会社 | 熱可塑性ポリエステルエラストマー樹脂発泡成形体およびその製造方法 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023037957A1 (ja) * | 2021-09-08 | 2023-03-16 | 東洋紡株式会社 | 熱可塑性ポリエステルエラストマー樹脂組成物及び発泡成形体 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4234202A1 (en) | 2023-08-30 |
| US20230383118A1 (en) | 2023-11-30 |
| JPWO2022085593A1 (ja) | 2022-04-28 |
| TW202222938A (zh) | 2022-06-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6358369B1 (ja) | 熱可塑性ポリエステルエラストマー樹脂発泡成形体およびその製造方法 | |
| JP7103003B2 (ja) | 熱可塑性ポリエステルエラストマー樹脂組成物およびその発泡成形体 | |
| JP7256981B2 (ja) | 熱可塑性ポリエステルエラストマー樹脂の発泡体からなる吸音材 | |
| JP6358368B1 (ja) | 熱可塑性ポリエステルエラストマー樹脂組成物およびその発泡成形体 | |
| JP7054448B6 (ja) | 熱可塑性ポリエステルエラストマー発泡成形体およびその製造方法 | |
| WO2020017450A1 (ja) | 熱可塑性ポリエステルエラストマー樹脂発泡成形体およびその製造方法 | |
| WO2022085593A1 (ja) | ポリエステルエラストマー樹脂組成物及び発泡成形体 | |
| JP6380638B1 (ja) | 熱可塑性ポリエステルエラストマー樹脂組成物およびその発泡成形体 | |
| JP7088398B2 (ja) | 発泡成形体 | |
| JPWO2020050213A1 (ja) | 発泡成形用熱可塑性ポリエステルエラストマー樹脂組成物およびその発泡成形体 | |
| JP7088362B1 (ja) | 発泡成形体およびその製造方法 | |
| WO2023037957A1 (ja) | 熱可塑性ポリエステルエラストマー樹脂組成物及び発泡成形体 | |
| JP2022143482A (ja) | 熱可塑性ポリエステルエラストマー樹脂組成物、及びそれからなる発泡成形体 | |
| JP7147285B2 (ja) | 熱可塑性ポリエステルエラストマー組成物、熱可塑性ポリエステルエラストマー発泡成形体およびその製造方法 | |
| JP2021063176A (ja) | 発泡成形用熱可塑性ポリエステルエラストマー樹脂組成物、発泡成形体、および発泡成形体の製造方法 | |
| JP2021063177A (ja) | 発泡成形用熱可塑性ポリエステルエラストマー樹脂組成物、発泡成形体、及び発泡成形体の製造方法 | |
| CN116583395A (zh) | 聚酯弹性体树脂组合物以及发泡成形体 | |
| WO2021054398A1 (ja) | 熱可塑性ポリエステルエラストマー樹脂組成物からなる発泡成形体、及び吸音材 | |
| JP2023114927A (ja) | ポリエステルエラストマー樹脂組成物及び発泡成形体 | |
| CN116507471A (zh) | 发泡成形体及其制造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2022515586 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21882734 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18032416 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202180071645.2 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2021882734 Country of ref document: EP Effective date: 20230522 |