WO2021090888A1 - タンパク質の製造方法 - Google Patents
タンパク質の製造方法 Download PDFInfo
- Publication number
- WO2021090888A1 WO2021090888A1 PCT/JP2020/041377 JP2020041377W WO2021090888A1 WO 2021090888 A1 WO2021090888 A1 WO 2021090888A1 JP 2020041377 W JP2020041377 W JP 2020041377W WO 2021090888 A1 WO2021090888 A1 WO 2021090888A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- medium
- concentration
- culture
- less
- phosphoric acid
- Prior art date
Links
- 108090000623 proteins and genes Proteins 0.000 title claims abstract description 197
- 102000004169 proteins and genes Human genes 0.000 title claims abstract description 119
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 24
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims abstract description 265
- 210000004102 animal cell Anatomy 0.000 claims abstract description 141
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims abstract description 133
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 claims abstract description 87
- 229910052700 potassium Inorganic materials 0.000 claims abstract description 87
- 239000011591 potassium Substances 0.000 claims abstract description 87
- 238000012258 culturing Methods 0.000 claims abstract description 81
- 238000000034 method Methods 0.000 claims description 119
- MTCFGRXMJLQNBG-UHFFFAOYSA-N serine Chemical compound OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 claims description 84
- 150000001412 amines Chemical class 0.000 claims description 79
- 229960001231 choline Drugs 0.000 claims description 71
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 claims description 71
- 239000013076 target substance Substances 0.000 claims description 61
- 230000001965 increasing effect Effects 0.000 claims description 34
- KIDHWZJUCRJVML-UHFFFAOYSA-N putrescine Chemical group NCCCCN KIDHWZJUCRJVML-UHFFFAOYSA-N 0.000 claims description 30
- 230000010412 perfusion Effects 0.000 claims description 20
- 241001522306 Serinus serinus Species 0.000 claims description 11
- 230000014509 gene expression Effects 0.000 abstract description 19
- 239000002609 medium Substances 0.000 description 312
- 210000004027 cell Anatomy 0.000 description 136
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 62
- 229960001153 serine Drugs 0.000 description 61
- 229910019142 PO4 Inorganic materials 0.000 description 42
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 42
- 239000010452 phosphate Substances 0.000 description 42
- 108010085895 Laminin Proteins 0.000 description 36
- 102000007547 Laminin Human genes 0.000 description 36
- 239000000306 component Substances 0.000 description 32
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 30
- 239000007640 basal medium Substances 0.000 description 25
- 150000001413 amino acids Chemical group 0.000 description 23
- 230000016784 immunoglobulin production Effects 0.000 description 21
- 239000013598 vector Substances 0.000 description 20
- 150000003839 salts Chemical class 0.000 description 17
- 230000000694 effects Effects 0.000 description 14
- 239000012526 feed medium Substances 0.000 description 13
- 229940024606 amino acid Drugs 0.000 description 12
- 230000004663 cell proliferation Effects 0.000 description 12
- 108020004414 DNA Proteins 0.000 description 11
- 238000002360 preparation method Methods 0.000 description 11
- 230000035755 proliferation Effects 0.000 description 11
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 10
- 229930182816 L-glutamine Natural products 0.000 description 8
- 230000035611 feeding Effects 0.000 description 8
- 239000001963 growth medium Substances 0.000 description 8
- 229910000403 monosodium phosphate Inorganic materials 0.000 description 8
- 235000019799 monosodium phosphate Nutrition 0.000 description 8
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 8
- 241000894007 species Species 0.000 description 8
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 7
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 7
- QWJSAWXRUVVRLH-UHFFFAOYSA-M choline bitartrate Chemical compound C[N+](C)(C)CCO.OC(=O)C(O)C(O)C([O-])=O QWJSAWXRUVVRLH-UHFFFAOYSA-M 0.000 description 7
- 229960004874 choline bitartrate Drugs 0.000 description 7
- 239000012634 fragment Substances 0.000 description 7
- 239000008103 glucose Substances 0.000 description 7
- 150000007523 nucleic acids Chemical class 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- 238000011218 seed culture Methods 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 238000003756 stirring Methods 0.000 description 7
- 229960002964 adalimumab Drugs 0.000 description 6
- 239000006285 cell suspension Substances 0.000 description 6
- 230000002708 enhancing effect Effects 0.000 description 6
- 238000011156 evaluation Methods 0.000 description 6
- 238000002474 experimental method Methods 0.000 description 6
- 230000006872 improvement Effects 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- -1 agmatin Chemical compound 0.000 description 5
- 239000002585 base Substances 0.000 description 5
- 239000006143 cell culture medium Substances 0.000 description 5
- 210000000349 chromosome Anatomy 0.000 description 5
- 239000003102 growth factor Substances 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- 239000013028 medium composition Substances 0.000 description 5
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 4
- 241000699802 Cricetulus griseus Species 0.000 description 4
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 4
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 4
- 102000003982 Parathyroid hormone Human genes 0.000 description 4
- 108090000445 Parathyroid hormone Proteins 0.000 description 4
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 4
- 102000036639 antigens Human genes 0.000 description 4
- 108091007433 antigens Proteins 0.000 description 4
- 230000000975 bioactive effect Effects 0.000 description 4
- 229910052739 hydrogen Inorganic materials 0.000 description 4
- 239000001257 hydrogen Substances 0.000 description 4
- 150000002500 ions Chemical class 0.000 description 4
- 239000000199 parathyroid hormone Substances 0.000 description 4
- 229960001319 parathyroid hormone Drugs 0.000 description 4
- 102000005962 receptors Human genes 0.000 description 4
- 108020003175 receptors Proteins 0.000 description 4
- 230000000717 retained effect Effects 0.000 description 4
- 229910052708 sodium Inorganic materials 0.000 description 4
- 239000011734 sodium Substances 0.000 description 4
- ATHGHQPFGPMSJY-UHFFFAOYSA-N spermidine Chemical compound NCCCCNCCCN ATHGHQPFGPMSJY-UHFFFAOYSA-N 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- 238000013518 transcription Methods 0.000 description 4
- 230000035897 transcription Effects 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 108010059616 Activins Proteins 0.000 description 3
- 102000005606 Activins Human genes 0.000 description 3
- 241000282552 Chlorocebus aethiops Species 0.000 description 3
- 108020004705 Codon Proteins 0.000 description 3
- 102000053602 DNA Human genes 0.000 description 3
- 108090000386 Fibroblast Growth Factor 1 Proteins 0.000 description 3
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 239000000488 activin Substances 0.000 description 3
- 238000004113 cell culture Methods 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 210000004748 cultured cell Anatomy 0.000 description 3
- 229960000633 dextran sulfate Drugs 0.000 description 3
- 239000013604 expression vector Substances 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- MYWUZJCMWCOHBA-VIFPVBQESA-N methamphetamine Chemical compound CN[C@@H](C)CC1=CC=CC=C1 MYWUZJCMWCOHBA-VIFPVBQESA-N 0.000 description 3
- 230000035772 mutation Effects 0.000 description 3
- 108020004707 nucleic acids Proteins 0.000 description 3
- 102000039446 nucleic acids Human genes 0.000 description 3
- 210000001672 ovary Anatomy 0.000 description 3
- 108091033319 polynucleotide Proteins 0.000 description 3
- 102000040430 polynucleotide Human genes 0.000 description 3
- 239000002157 polynucleotide Substances 0.000 description 3
- 238000000746 purification Methods 0.000 description 3
- 230000010076 replication Effects 0.000 description 3
- PFNFFQXMRSDOHW-UHFFFAOYSA-N spermine Chemical compound NCCCNCCCCNCCCN PFNFFQXMRSDOHW-UHFFFAOYSA-N 0.000 description 3
- 238000011282 treatment Methods 0.000 description 3
- 239000013603 viral vector Substances 0.000 description 3
- 102000009027 Albumins Human genes 0.000 description 2
- 108010088751 Albumins Proteins 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- 241000271566 Aves Species 0.000 description 2
- 102100023995 Beta-nerve growth factor Human genes 0.000 description 2
- 102000004219 Brain-derived neurotrophic factor Human genes 0.000 description 2
- 108090000715 Brain-derived neurotrophic factor Proteins 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- 101100283604 Caenorhabditis elegans pigk-1 gene Proteins 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 241000282693 Cercopithecidae Species 0.000 description 2
- 101100007328 Cocos nucifera COS-1 gene Proteins 0.000 description 2
- 241000699800 Cricetinae Species 0.000 description 2
- 102000004127 Cytokines Human genes 0.000 description 2
- 108090000695 Cytokines Proteins 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000003951 Erythropoietin Human genes 0.000 description 2
- 108090000394 Erythropoietin Proteins 0.000 description 2
- 108050001049 Extracellular proteins Proteins 0.000 description 2
- 102100026745 Fatty acid-binding protein, liver Human genes 0.000 description 2
- 101710188974 Fatty acid-binding protein, liver Proteins 0.000 description 2
- 101710189565 Fatty acid-binding protein, liver-type Proteins 0.000 description 2
- 102000003971 Fibroblast Growth Factor 1 Human genes 0.000 description 2
- 102000003974 Fibroblast growth factor 2 Human genes 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 102000004269 Granulocyte Colony-Stimulating Factor Human genes 0.000 description 2
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 description 2
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 2
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 2
- 108010043121 Green Fluorescent Proteins Proteins 0.000 description 2
- 102000004144 Green Fluorescent Proteins Human genes 0.000 description 2
- 101000599951 Homo sapiens Insulin-like growth factor I Proteins 0.000 description 2
- 102000002265 Human Growth Hormone Human genes 0.000 description 2
- 108010000521 Human Growth Hormone Proteins 0.000 description 2
- 239000000854 Human Growth Hormone Substances 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- 102100037852 Insulin-like growth factor I Human genes 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 2
- 108010025020 Nerve Growth Factor Proteins 0.000 description 2
- 108010038512 Platelet-Derived Growth Factor Proteins 0.000 description 2
- 102000010780 Platelet-Derived Growth Factor Human genes 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- 241000288906 Primates Species 0.000 description 2
- 102000001708 Protein Isoforms Human genes 0.000 description 2
- 108010029485 Protein Isoforms Proteins 0.000 description 2
- 241000508269 Psidium Species 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 2
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 2
- 241000283984 Rodentia Species 0.000 description 2
- 102000040739 Secretory proteins Human genes 0.000 description 2
- 108091058545 Secretory proteins Proteins 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 102000036693 Thrombopoietin Human genes 0.000 description 2
- 108010041111 Thrombopoietin Proteins 0.000 description 2
- 108010009583 Transforming Growth Factors Proteins 0.000 description 2
- 102000009618 Transforming Growth Factors Human genes 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 description 2
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 2
- 102100039037 Vascular endothelial growth factor A Human genes 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- 241000269370 Xenopus <genus> Species 0.000 description 2
- 238000009825 accumulation Methods 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 229940077737 brain-derived neurotrophic factor Drugs 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 239000012228 culture supernatant Substances 0.000 description 2
- 230000002354 daily effect Effects 0.000 description 2
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 2
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 229940088598 enzyme Drugs 0.000 description 2
- 229940105423 erythropoietin Drugs 0.000 description 2
- 230000003203 everyday effect Effects 0.000 description 2
- 239000000284 extract Substances 0.000 description 2
- 108091006047 fluorescent proteins Proteins 0.000 description 2
- 102000034287 fluorescent proteins Human genes 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- 239000005090 green fluorescent protein Substances 0.000 description 2
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid Chemical compound CCCCCCC(O)=O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 2
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 description 2
- 229940088597 hormone Drugs 0.000 description 2
- 239000005556 hormone Substances 0.000 description 2
- 230000028993 immune response Effects 0.000 description 2
- 102000028557 immunoglobulin binding proteins Human genes 0.000 description 2
- 108091009323 immunoglobulin binding proteins Proteins 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 229940053128 nerve growth factor Drugs 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 229960004838 phosphoric acid Drugs 0.000 description 2
- 230000001766 physiological effect Effects 0.000 description 2
- 239000013600 plasmid vector Substances 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- OXCMYAYHXIHQOA-UHFFFAOYSA-N potassium;[2-butyl-5-chloro-3-[[4-[2-(1,2,4-triaza-3-azanidacyclopenta-1,4-dien-5-yl)phenyl]phenyl]methyl]imidazol-4-yl]methanol Chemical compound [K+].CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=2C(=CC=CC=2)C2=N[N-]N=N2)C=C1 OXCMYAYHXIHQOA-UHFFFAOYSA-N 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 230000003248 secreting effect Effects 0.000 description 2
- FQENQNTWSFEDLI-UHFFFAOYSA-J sodium diphosphate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]P([O-])(=O)OP([O-])([O-])=O FQENQNTWSFEDLI-UHFFFAOYSA-J 0.000 description 2
- 229940063673 spermidine Drugs 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- 102000003390 tumor necrosis factor Human genes 0.000 description 2
- 230000035899 viability Effects 0.000 description 2
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- GJFNRSDCSTVPCJ-UHFFFAOYSA-N 1,8-bis(dimethylamino)naphthalene Chemical compound C1=CC(N(C)C)=C2C(N(C)C)=CC=CC2=C1 GJFNRSDCSTVPCJ-UHFFFAOYSA-N 0.000 description 1
- TUSDEZXZIZRFGC-UHFFFAOYSA-N 1-O-galloyl-3,6-(R)-HHDP-beta-D-glucose Natural products OC1C(O2)COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC1C(O)C2OC(=O)C1=CC(O)=C(O)C(O)=C1 TUSDEZXZIZRFGC-UHFFFAOYSA-N 0.000 description 1
- GYSCBCSGKXNZRH-UHFFFAOYSA-N 1-benzothiophene-2-carboxamide Chemical compound C1=CC=C2SC(C(=O)N)=CC2=C1 GYSCBCSGKXNZRH-UHFFFAOYSA-N 0.000 description 1
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 1
- 102000004400 Aminopeptidases Human genes 0.000 description 1
- 108090000915 Aminopeptidases Proteins 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- 102000055006 Calcitonin Human genes 0.000 description 1
- 108060001064 Calcitonin Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 102000005367 Carboxypeptidases Human genes 0.000 description 1
- 108010006303 Carboxypeptidases Proteins 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 108010059892 Cellulase Proteins 0.000 description 1
- 102000012286 Chitinases Human genes 0.000 description 1
- 108010022172 Chitinases Proteins 0.000 description 1
- 239000004381 Choline salt Substances 0.000 description 1
- 108700010070 Codon Usage Proteins 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 102000029816 Collagenase Human genes 0.000 description 1
- 108060005980 Collagenase Proteins 0.000 description 1
- GHVNFZFCNZKVNT-UHFFFAOYSA-N Decanoic acid Natural products CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 101100118093 Drosophila melanogaster eEF1alpha2 gene Proteins 0.000 description 1
- 206010059866 Drug resistance Diseases 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 108010059378 Endopeptidases Proteins 0.000 description 1
- 102000005593 Endopeptidases Human genes 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- HTQBXNHDCUEHJF-XWLPCZSASA-N Exenatide Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)NCC(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 HTQBXNHDCUEHJF-XWLPCZSASA-N 0.000 description 1
- 108010011459 Exenatide Proteins 0.000 description 1
- 102000018389 Exopeptidases Human genes 0.000 description 1
- 108010091443 Exopeptidases Proteins 0.000 description 1
- 239000001263 FEMA 3042 Substances 0.000 description 1
- 102100031706 Fibroblast growth factor 1 Human genes 0.000 description 1
- 102100028412 Fibroblast growth factor 10 Human genes 0.000 description 1
- 102100024785 Fibroblast growth factor 2 Human genes 0.000 description 1
- 102000003972 Fibroblast growth factor 7 Human genes 0.000 description 1
- 108090000385 Fibroblast growth factor 7 Proteins 0.000 description 1
- 102100028071 Fibroblast growth factor 7 Human genes 0.000 description 1
- 102100037362 Fibronectin Human genes 0.000 description 1
- 108010067306 Fibronectins Proteins 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 102400000321 Glucagon Human genes 0.000 description 1
- 108060003199 Glucagon Proteins 0.000 description 1
- 108010073324 Glutaminase Proteins 0.000 description 1
- 102000009127 Glutaminase Human genes 0.000 description 1
- 101000917237 Homo sapiens Fibroblast growth factor 10 Proteins 0.000 description 1
- 101001060261 Homo sapiens Fibroblast growth factor 7 Proteins 0.000 description 1
- 101000629635 Homo sapiens Signal recognition particle receptor subunit alpha Proteins 0.000 description 1
- 102000008100 Human Serum Albumin Human genes 0.000 description 1
- 108091006905 Human Serum Albumin Proteins 0.000 description 1
- 108010093096 Immobilized Enzymes Proteins 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- 101710128836 Large T antigen Proteins 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 1
- 108010014251 Muramidase Proteins 0.000 description 1
- 102000016943 Muramidase Human genes 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 description 1
- 108010062010 N-Acetylmuramoyl-L-alanine Amidase Proteins 0.000 description 1
- 125000001429 N-terminal alpha-amino-acid group Chemical group 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 239000012124 Opti-MEM Substances 0.000 description 1
- 102000016978 Orphan receptors Human genes 0.000 description 1
- 108070000031 Orphan receptors Proteins 0.000 description 1
- 102000004264 Osteopontin Human genes 0.000 description 1
- 108010081689 Osteopontin Proteins 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- 241000282579 Pan Species 0.000 description 1
- LRBQNJMCXXYXIU-PPKXGCFTSA-N Penta-digallate-beta-D-glucose Natural products OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@@H]2[C@H]([C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-PPKXGCFTSA-N 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 108700008625 Reporter Genes Proteins 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 108010071390 Serum Albumin Proteins 0.000 description 1
- 102000007562 Serum Albumin Human genes 0.000 description 1
- 102100026900 Signal recognition particle receptor subunit alpha Human genes 0.000 description 1
- 108020004682 Single-Stranded DNA Proteins 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- 102000005157 Somatostatin Human genes 0.000 description 1
- 108010056088 Somatostatin Proteins 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 108010049264 Teriparatide Proteins 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- 101710120037 Toxin CcdB Proteins 0.000 description 1
- 108060008539 Transglutaminase Proteins 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 102100035140 Vitronectin Human genes 0.000 description 1
- 108010031318 Vitronectin Proteins 0.000 description 1
- 241000269368 Xenopus laevis Species 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 108010023082 activin A Proteins 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 210000004100 adrenal gland Anatomy 0.000 description 1
- 238000001042 affinity chromatography Methods 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 125000000539 amino acid group Chemical group 0.000 description 1
- 238000003277 amino acid sequence analysis Methods 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000003927 aminopyridines Chemical class 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 150000004982 aromatic amines Chemical class 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- BBBFJLBPOGFECG-VJVYQDLKSA-N calcitonin Chemical compound N([C@H](C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(N)=O)C(C)C)C(=O)[C@@H]1CSSC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1 BBBFJLBPOGFECG-VJVYQDLKSA-N 0.000 description 1
- 229960004015 calcitonin Drugs 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 239000000679 carrageenan Substances 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 229940113118 carrageenan Drugs 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 150000003943 catecholamines Chemical class 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 229940106157 cellulase Drugs 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 235000019417 choline salt Nutrition 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 229960002424 collagenase Drugs 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 229960002086 dextran Drugs 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- ZPWVASYFFYYZEW-UHFFFAOYSA-L dipotassium hydrogen phosphate Chemical compound [K+].[K+].OP([O-])([O-])=O ZPWVASYFFYYZEW-UHFFFAOYSA-L 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 229960003638 dopamine Drugs 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 238000004520 electroporation Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000003028 enzyme activity measurement method Methods 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 229940031098 ethanolamine Drugs 0.000 description 1
- 229960001519 exenatide Drugs 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 230000001605 fetal effect Effects 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000005194 fractionation Methods 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- LRBQNJMCXXYXIU-QWKBTXIPSA-N gallotannic acid Chemical compound OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@H]2[C@@H]([C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-QWKBTXIPSA-N 0.000 description 1
- 238000001641 gel filtration chromatography Methods 0.000 description 1
- MASNOZXLGMXCHN-ZLPAWPGGSA-N glucagon Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)C(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 MASNOZXLGMXCHN-ZLPAWPGGSA-N 0.000 description 1
- 229960004666 glucagon Drugs 0.000 description 1
- 108010000320 glucan 1,6-alpha-isomaltosidase Proteins 0.000 description 1
- 229940022353 herceptin Drugs 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 210000001822 immobilized cell Anatomy 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 229910017053 inorganic salt Inorganic materials 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 108010028309 kalinin Proteins 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 210000003292 kidney cell Anatomy 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 229960000448 lactic acid Drugs 0.000 description 1
- 108010038862 laminin 10 Proteins 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 238000001638 lipofection Methods 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 230000002934 lysing effect Effects 0.000 description 1
- 239000004325 lysozyme Substances 0.000 description 1
- 229960000274 lysozyme Drugs 0.000 description 1
- 235000010335 lysozyme Nutrition 0.000 description 1
- 108010026228 mRNA guanylyltransferase Proteins 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229940099690 malic acid Drugs 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 239000012533 medium component Substances 0.000 description 1
- 210000002752 melanocyte Anatomy 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 102000035118 modified proteins Human genes 0.000 description 1
- 108091005573 modified proteins Proteins 0.000 description 1
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 1
- 235000019796 monopotassium phosphate Nutrition 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229960003301 nivolumab Drugs 0.000 description 1
- 230000001254 nonsecretory effect Effects 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 210000001706 olfactory mucosa Anatomy 0.000 description 1
- 210000000287 oocyte Anatomy 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 229940116315 oxalic acid Drugs 0.000 description 1
- 239000006174 pH buffer Substances 0.000 description 1
- WLJNZVDCPSBLRP-UHFFFAOYSA-N pamoic acid Chemical compound C1=CC=C2C(CC=3C4=CC=CC=C4C=C(C=3O)C(=O)O)=C(O)C(C(O)=O)=CC2=C1 WLJNZVDCPSBLRP-UHFFFAOYSA-N 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- NIXKBAZVOQAHGC-UHFFFAOYSA-N phenylmethanesulfonic acid Chemical compound OS(=O)(=O)CC1=CC=CC=C1 NIXKBAZVOQAHGC-UHFFFAOYSA-N 0.000 description 1
- PJNZPQUBCPKICU-UHFFFAOYSA-N phosphoric acid;potassium Chemical compound [K].OP(O)(O)=O PJNZPQUBCPKICU-UHFFFAOYSA-N 0.000 description 1
- NFIYTPYOYDDLGO-UHFFFAOYSA-N phosphoric acid;sodium Chemical compound [Na].OP(O)(O)=O NFIYTPYOYDDLGO-UHFFFAOYSA-N 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 210000004560 pineal gland Anatomy 0.000 description 1
- 229920001184 polypeptide Polymers 0.000 description 1
- 239000011736 potassium bicarbonate Substances 0.000 description 1
- 235000015497 potassium bicarbonate Nutrition 0.000 description 1
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 235000011181 potassium carbonates Nutrition 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 238000002331 protein detection Methods 0.000 description 1
- 238000001742 protein purification Methods 0.000 description 1
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical compound C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 150000003248 quinolines Chemical class 0.000 description 1
- SBYHFKPVCBCYGV-UHFFFAOYSA-N quinuclidine Chemical compound C1CC2CCN1CC2 SBYHFKPVCBCYGV-UHFFFAOYSA-N 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- UQDJGEHQDNVPGU-UHFFFAOYSA-N serine phosphoethanolamine Chemical compound [NH3+]CCOP([O-])(=O)OCC([NH3+])C([O-])=O UQDJGEHQDNVPGU-UHFFFAOYSA-N 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 230000007928 solubilization Effects 0.000 description 1
- 238000005063 solubilization Methods 0.000 description 1
- NHXLMOGPVYXJNR-ATOGVRKGSA-N somatostatin Chemical compound C([C@H]1C(=O)N[C@H](C(N[C@@H](CO)C(=O)N[C@@H](CSSC[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(=O)N1)[C@@H](C)O)NC(=O)CNC(=O)[C@H](C)N)C(O)=O)=O)[C@H](O)C)C1=CC=CC=C1 NHXLMOGPVYXJNR-ATOGVRKGSA-N 0.000 description 1
- 229960000553 somatostatin Drugs 0.000 description 1
- 229940063675 spermine Drugs 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 229920002258 tannic acid Polymers 0.000 description 1
- 229940033123 tannic acid Drugs 0.000 description 1
- 235000015523 tannic acid Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 210000002105 tongue Anatomy 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 102000003601 transglutaminase Human genes 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 229960000575 trastuzumab Drugs 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 1
- 238000004704 ultra performance liquid chromatography Methods 0.000 description 1
- 241000701161 unidentified adenovirus Species 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- 229960005486 vaccine Drugs 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P21/00—Preparation of peptides or proteins
- C12P21/02—Preparation of peptides or proteins having a known sequence of two or more amino acids, e.g. glutathione
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/0018—Culture media for cell or tissue culture
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
- C07K16/241—Tumor Necrosis Factors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/10—Immunoglobulins specific features characterized by their source of isolation or production
- C07K2317/14—Specific host cells or culture conditions, e.g. components, pH or temperature
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
- C12N2500/10—Metals; Metal chelators
- C12N2500/12—Light metals, i.e. alkali, alkaline earth, Be, Al, Mg
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/32—Amino acids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/42—Organic phosphate, e.g. beta glycerophosphate
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/46—Amines, e.g. putrescine
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/105—Insulin-like growth factors [IGF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2510/00—Genetically modified cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2510/00—Genetically modified cells
- C12N2510/02—Cells for production
Definitions
- the present invention relates to a method for culturing animal cells.
- One aspect of the present invention relates to a method for producing a target substance such as a protein by an animal cell.
- Animal cells are used as an expression host for recombinant proteins.
- Patent Document 1 As a method for producing a recombinant protein as an expression host of a Chinese hamster ovary-derived cell line (CHO), a fed-batch medium containing phosphoric acid is flowed so that the phosphoric acid concentration in the medium is 1.5 to 3.5 mM. A method of adding has been reported (Patent Document 1).
- Patent Document 2 a method of culturing cells using a medium containing a high concentration of choline
- Patent Document 3 a method of culturing cells using a medium containing a high concentration of serine
- Patent Document 4 A method of culturing cells using a medium containing an amine (Patent Document 4) is known.
- An object of the present invention is to provide a novel technique for improving the culture performance of animal cells (for example, proliferation of animal cells and production of target substance by animal cells).
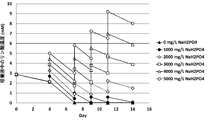
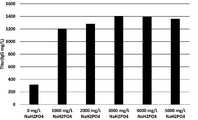
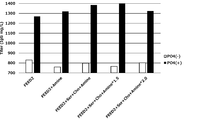
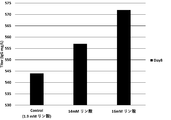
- the present inventors increase the concentration of specific components such as phosphate and potassium in the medium when culturing animal cells to increase the culture results of animal cells (for example, proliferation of animal cells and target substances by animal cells).
- the present invention has been completed by finding that the production) is improved.
- the present invention can be exemplified as follows.
- the method the culture is carried out under the conditions (A) and / or (B) below.
- the method, wherein the target substance is a protein.
- a method of culturing animal cells Including culturing animal cells in medium The method: the culture is carried out under the conditions (A) and / or (B) below.
- the method of the present invention is a method for culturing animal cells, which comprises culturing the animal cells in a medium, and the culture enhances the phosphoric acid concentration and / or the potassium concentration in the medium. It is a method that is carried out under the above conditions.
- the target substance may be produced by culturing animal cells. That is, when an animal cell has a target substance-producing ability, the target substance can be produced by culturing the cell. That is, one aspect of the method of the present invention is a method for producing a target substance, which comprises culturing an animal cell capable of producing the target substance in a medium and recovering the target substance, and the culture is the medium. The method may be carried out under conditions in which the concentration of phosphoric acid and / or the concentration of potassium in the medium is increased.
- the target substance is not particularly limited as long as it can be produced by animal cells.
- examples of the target substance include proteins.
- a protein produced as a target substance is also referred to as a "target protein”.
- Animal cells are not particularly limited. The animal cell can be appropriately selected depending on various conditions such as the use of the animal cell. For example, when an animal cell is used for producing a target protein, the animal cell is not particularly limited as long as it can express the target protein.
- Animal cells are also referred to as "hosts," "expression hosts,” or "host cells.” Animals include mammals, birds and amphibians. Animals include mammals in particular. Mammals include rodents and primates. Rodents include hamsters, mice, rats and guinea pigs. Examples of hamsters include Chinese hamsters. Primates include humans, monkeys and chimpanzees. Examples of monkeys include African green monkeys. Examples of birds include chickens. Amphibians include Xenopus laevis.
- tissue or cell from which the animal cell is derived is not particularly limited.
- Tissues or cells from which animal cells are derived include ovaries, kidneys, adrenal glands, tongue epithelium, olfactory epithelium, pineal gland, thyroid gland, and melanocytes.
- Examples of Chinese hamster cells include Chinese hamster ovary-derived cell lines (CHO). Specific examples of CHO include CHO-DG44 and CHO-K1. Examples of human cells include human fetal kidney cell-derived cell lines (HEK). Specific examples of HEK include HEK293 and HEK293T. Examples of African green monkey cells include African green monkey renal cell-derived cell lines (COS). Specific examples of COS include COS-1.
- Specific examples of Xenopus cells include Xenopus oocytes.
- Animal cell capable of producing a target substance means an animal cell capable of producing a target substance.
- animal cells capable of producing a target substance means an ability to produce a target substance (for example, express a target protein) and accumulate it in a culture to a extent that it can be recovered when cultured in a medium. It may mean an animal cell having.
- "Accumulation in culture” may specifically mean accumulation in medium, cell surface, intracellular, or a combination thereof. The case where the target substance accumulates extracellularly (for example, in the medium or on the cell surface) is also referred to as "secretion" or "secretory production” of the target substance.
- the animal cell may have a secretory production ability (the ability to secrete and produce the target substance) of the target substance.
- the accumulated amount of the target substance may be, for example, 10 ⁇ g / L or more, 1 mg / L or more, 100 mg / L or more, or 1 g / L or more as the accumulated amount in the culture.
- the animal cell may have the ability to produce one kind of target substance, or may have the ability to produce two or more kinds of target substances.
- the animal cell may be one that originally has the ability to produce the target substance, or may be one that has been modified to have the ability to produce the target substance.
- the animal cells may be modified so as to enhance the inherent ability to produce the target substance.
- the animal cell having the target substance-producing ability can be obtained, for example, by imparting the target substance-producing ability to the animal cell as described above, or by enhancing the target substance-producing ability of the animal cell as described above. ..
- the target protein-producing ability can be imparted or enhanced by introducing a gene encoding the target protein.
- the gene encoding the target protein is also referred to as the "target protein gene".
- the target protein is not particularly limited as long as it can be expressed using animal cells as a host.
- the protein may be a host-derived protein or a heterologous protein.
- the "heterologous protein” refers to a protein that is exogenous to a host that produces the protein (ie, an animal cell capable of producing the protein of interest).
- the target protein may be, for example, a naturally occurring protein, a modified protein thereof, or an artificially designed amino acid sequence protein.
- the target protein may be, for example, a protein derived from a microorganism, a protein derived from a plant, a protein derived from an animal, or a protein derived from a virus.
- the protein of interest may be, in particular, a protein of human origin.
- the target protein may be a monomeric protein or a multimeric protein.
- the target protein may be a secretory protein or a non-secretory protein.
- the "protein” also includes what is called a peptide, such as an oligopeptide or a polypeptide.
- target protein examples include enzymes, bioactive proteins, receptor proteins, antigen proteins, and other proteins.
- Enzymes include cellulase, transglutaminase, protein glutaminase, isomaltodextranase, protease, endopeptidase, exopeptidase, aminopeptidase, carboxypeptidase, collagenase, and chitinase.
- bioactive proteins include growth factors (growth factors), hormones, cytokines, and antibody-related molecules.
- Growth factors include epidermal growth factor (EGF), insulin-like growth factor-1 (IGF-1), and transforming growth factor (TGF). ), Nerve growth factor (NGF), Brain-derived neurotrophic factor (BDNF), Vascular endothelial growth factor (VEGF), Granulocyte- colony stimulating factor (G-CSF), granulocyte-macrophage-colony stimulating factor (GM-CSF), platelet-derived growth factor (PDGF), erythropoietin (EPO), Thrombopoietin (TPO), acidic fibroblast growth factor (aFGF or FGF1), basic fibroblast growth factor (bFGF or FGF2), keratinocyte growth factor ; KGF-1 or FGF7, KGF-2 or FGF10), hepatocellular growth factor (HGF), stem cell factor (SCF), activin (Activin). Examples of activin include activin A, C, and E.
- hormones include insulin, glucagon, somatostatin, human growth hormone (hGH), parathyroid hormone (PTH), calcitonin, and exenatide.
- Cytokines include interleukin, interferon, and tumor necrosis factor (TNF).
- the bioactive protein may be the whole protein or a part thereof.
- a portion having a physiological activity can be mentioned.
- Specific examples of the portion having physiological activity include the physiologically active peptide Teriparatide consisting of 34 amino acid residues at the N-terminal of a mature parathyroid hormone (PTH).
- Antibody-related molecule may mean a protein comprising a molecular species consisting of a single domain selected from the domains constituting a complete antibody or a combination of two or more domains. Domains that make up a complete antibody include the heavy chain domains VH, CH1, CH2, and CH3, and the light chain domains VL and CL.
- the antibody-related molecule may be a monomeric protein or a multimeric protein as long as it contains the above-mentioned molecular species. When the antibody-related molecule is a multimeric protein, it may be a homomultimer composed of a single type of subunit, or a heteromultimer composed of two or more types of subunits. May be good.
- antibody-related molecules include complete antibodies, Fab, F (ab'), F (ab') 2 , Fc, dimers consisting of heavy chain (H chain) and light chain (L chain), and Fc. Fusion protein, heavy chain (H chain), light chain (L chain), single chain Fv (scFv), sc (Fv) 2 , disulfide bond Fv (sdFv), diabody, VHH fragment (nanobody®) Be done. More specific examples of antibody-related molecules include Trastuzumab, Adalimumab, and Nivolumab.
- the receptor protein examples include a physiologically active protein and a receptor protein for other physiologically active substances.
- Other bioactive substances include neurotransmitters such as dopamine.
- the receptor protein may be an orphan receptor whose corresponding ligand is unknown.
- the antigen protein is not particularly limited as long as it can elicit an immune response.
- the antigen protein can be appropriately selected, for example, according to the target of the assumed immune response.
- the antigen protein can be used, for example, as a vaccine.
- LFABP Liver-type fatty acid-binding protein
- fluorescent protein include Green Fluorescent Protein (GFP).
- immunoglobulin binding protein include Protein A, Protein G, and Protein L.
- albumin include human serum albumin.
- fibroin-like protein include those disclosed in WO2017 / 090665 and WO2017 / 171001.
- Laminin is a protein having a heterotrimeric structure consisting of ⁇ chain, ⁇ chain, and ⁇ chain.
- laminin include mammalian laminin.
- the laminin subunit chains ie, ⁇ chain, ⁇ chain, and ⁇ chain
- the laminin subunit chains include 5 types of ⁇ chain ( ⁇ 1 to ⁇ 5), 3 types of ⁇ chain ( ⁇ 1 to ⁇ 3), and 3 types of ⁇ chain ( ⁇ 1). ⁇ ⁇ 3) can be mentioned.
- Laminin constitutes various isoforms by combining these subunit chains.
- laminin specifically, for example, laminin 111, laminin 121, laminin 211, laminin 213, laminin 221, laminin 311, laminin 321, laminin 332, laminin 411, laminin 421, laminin 423, laminin 511, laminin 521, laminin. 523 can be mentioned.
- a partial sequence of laminin includes laminin E8, which is an E8 fragment of laminin.
- laminin E8 has a heterotrimeric structure consisting of an ⁇ chain E8 fragment ( ⁇ chain E8), a ⁇ chain E8 fragment ( ⁇ chain E8), and a ⁇ chain E8 fragment ( ⁇ chain E8). It is a protein that has.
- the subunit chains of laminin E8 are collectively referred to as “E8 subunit chains”.
- Examples of the E8 subunit chain include the E8 fragment of the laminin subunit chain exemplified above.
- Laminin E8 constitutes various isoforms by combining these E8 subunit chains.
- Specific examples of the laminin E8 include laminin 111E8, laminin 121E8, laminin 211E8, laminin 221E8, laminin 332E8, laminin 421E8, laminin 411E8, laminin 511E8, and laminin 521E8.
- the target protein may be, for example, a protein having a known or natural amino acid sequence of the above-mentioned protein. Further, the target protein may be, for example, a variant of a protein having a known or natural amino acid sequence of the above-mentioned protein. Variants include proteins having an amino acid sequence in which one or several amino acids are substituted, deleted, inserted or added at one or several positions in a known or natural amino acid sequence. Specifically, "1 or several" means, for example, 1 to 50, 1 to 40, 1 to 30, preferably 1 to 20, more preferably 1 to 10, and even more preferably 1. It may mean up to 5, particularly preferably 1 to 3.
- Variants include, for example, 50% or more, 65% or more, 80% or more, preferably 90% or more, more preferably 95% or more, still more preferably 97% or more, particularly with respect to the entire known or natural amino acid sequence.
- a protein having an amino acid sequence having 99% or more identity is also mentioned.
- the protein specified by the species of origin is not limited to the protein itself found in the species, but includes proteins having the amino acid sequence of the protein found in the species and variants thereof.
- Variants may or may not be found in the species. That is, for example, "human-derived protein” is not limited to the protein itself found in humans, but includes proteins having an amino acid sequence of proteins found in humans and variants thereof.
- the target protein gene is not particularly limited as long as it encodes the target protein as described above.
- the target protein gene may be, for example, a gene having a known or natural base sequence of a gene encoding a protein as described above. Further, the target protein gene may be, for example, a variant of a gene having a known or natural base sequence of a gene encoding a protein as described above.
- the protein gene of interest may be modified, for example, to encode a protein having a variant sequence as exemplified above.
- the target protein gene may be one in which any codon is replaced with an equivalent codon.
- the target protein gene may be modified to have an optimum codon, for example, depending on the codon usage frequency of the host cell.
- the term "gene” is not limited to DNA as long as it encodes the corresponding expression product, and may include any polynucleotide. That is, the "target protein gene” may mean any polynucleotide encoding the target protein.
- the target protein gene may be DNA, RNA, or a combination thereof.
- the target protein gene may be single-stranded or double-stranded.
- the target protein gene may be single-stranded DNA or single-stranded RNA.
- the target protein gene may be double-stranded DNA, double-stranded RNA, or a hybrid strand consisting of a DNA strand and an RNA strand.
- the protein gene of interest may contain both DNA and RNA residues in a single polynucleotide strand.
- the protein gene of interest may or may not contain an intron.
- the mode of the target protein gene can be appropriately selected according to various conditions such as the means for expressing the target protein.
- the expression “having a (amino acid or base) sequence” means “including the (amino acid or base) sequence” and includes the case of “consisting of the (amino acid or base) sequence”.
- the target protein is expressed from the target protein gene. That is, an animal cell capable of producing a target protein has a target protein gene. Specifically, an animal cell having a target protein-producing ability has a target protein gene capable of expressing it. It is sufficient that the animal cell having the target protein-producing ability has the target protein gene until the target protein is expressed to a desired degree. That is, the animal cell capable of producing the target protein may or may not have the target protein gene after the expression of the target protein.
- "expression of target protein gene” and "expression of target protein” may be used synonymously.
- the target protein gene can be obtained by cloning from an organism having the target protein gene. Nucleic acids such as genomic DNA and cDNA containing the same gene can be used for cloning. The target protein gene can also be obtained by chemical synthesis (Gene, 60 (1), 115-127 (1987)).
- the acquired target protein gene can be used as it is or after being appropriately modified. That is, the variant can be obtained by modifying the target protein gene.
- the gene can be modified by a known method.
- a site-specific mutation method can be used to introduce a desired mutation into a target site of DNA.
- a site-specific mutation method a method using PCR (Higuchi, R., 61, in PCR technology, Erlich, H. A. Eds., Stockton press (1989); Carter, P., Meth. In Enzymol., 154, 382 (1987)) and methods using phage (Kramer, W. and Frits, H. J., Meth. In Enzymol., 154, 350 (1987); Kunkel, T. A. et al., Meth . In Enzymol., 154, 367 (1987)).
- the variant of the target protein gene may be directly obtained by chemical synthesis.
- the form of introducing the target protein gene into the host cell is not particularly limited.
- the target protein gene may be retained in the host cell so that it can be expressed. Specifically, for example, when a target protein gene is introduced in a form that requires transcription of DNA or the like, the target protein gene is retained in a host cell so as to be expressible under the control of a promoter functioning in the host cell. Just do it.
- the protein gene of interest may be extrachromosomally present or introduced onto the chromosome. When introducing two or more genes, each gene need only be retained in the host cell so that it can be expressed.
- the promoter for expressing the target protein gene is not particularly limited as long as it functions in the host cell.
- "Promoter functioning in a host cell” means a promoter having promoter activity in a host cell.
- the promoter may be a host cell-derived promoter or a heterologous promoter.
- the promoter may be a promoter unique to the target protein gene or a promoter of another gene.
- the promoter may be a stronger promoter than the unique promoter of the target protein gene.
- Promoters that function in animal cells include the SV40 promoter, EF1a promoter, RSV promoter, CMV promoter, and SRalpha promoter. Further, as the promoter, a highly active form of a conventional promoter may be obtained and used by using various reporter genes. Methods for assessing promoter strength and examples of potent promoters are described in Goldstein et al.'S paper (Prokaryotic promoters in biotechnology. Biotechnol. Annu. Rev., 1, 105-128
- the target protein gene can be introduced into a host cell using, for example, a vector containing the same gene.
- a vector containing a target protein gene is also referred to as an "expression vector of the target protein gene".
- the expression vector of the target protein gene can be constructed, for example, by ligating a DNA fragment containing the target protein gene with the vector.
- the gene By introducing the expression vector of the target protein gene into the host cell, the gene can be introduced into the host cell.
- the vector may comprise a marker such as a drug resistance gene.
- the vector may include an expression regulatory sequence such as a promoter for expressing the inserted gene.
- the vector can be appropriately selected according to various conditions such as the type of host cell and the form of introduction of the target protein gene.
- Examples of vectors that can be used for gene transfer into animal cells include plasmid vectors and viral vectors.
- the viral vector include a retrovirus vector and an adenovirus vector.
- Examples of the plasmid vector include a pcDNA series vector (pcDNA3.1 and the like; Thermo Fisher Scientific), a pBApo-CMV series vector (Takara Bio), and pCI-neo (Promega).
- the vector can be integrated into the chromosome of the host cell, can autonomously replicate outside the chromosome of the host cell, or can be temporarily retained outside the chromosome of the host cell.
- a vector having a viral origin of replication can replicate autonomously outside the chromosomes of animal cells.
- the pcDNA series vector has an SV40 origin of replication and can autonomously replicate extrachromosomally in host cells expressing the SV40 large T antigen (COS-1, HEK293T, etc.).
- the target protein gene can also be introduced into a host cell by, for example, introducing a nucleic acid fragment containing the gene into the host cell.
- nucleic acid fragments include linear DNA and linear RNA.
- linear RNA include mRNA and cRNA.
- the method for introducing nucleic acids such as vectors and nucleic acid fragments into host cells can be appropriately selected according to various conditions such as the type of host cells.
- Examples of the method for introducing a nucleic acid such as a vector or a nucleic acid fragment into an animal cell include a DEAE dextran method, a calcium phosphate method, a lipofection method, an electroporation method, and a microinjection method.
- the vector is a viral vector
- the vector can be introduced into the host cell by infecting the host cell with the vector (virus).
- a cell that originally has the target protein gene may be modified and used so that the expression of the target protein gene is increased.
- Increased gene expression means that the per-cell expression level of the gene is increased as compared to unmodified cells.
- unmodified cell as used herein means a control cell that has not been modified to increase the expression of the target gene. Examples of unmodified cells include wild-type cells and cells from which they are modified.
- Methods for increasing the expression of the target protein gene include increasing the number of copies of the target protein gene and improving the transcription efficiency and translation efficiency of the target protein gene. An increase in the number of copies of the target protein gene can be achieved by introducing the target protein gene into a host cell. The introduction of the target protein gene can be carried out as described above.
- the target protein gene to be introduced may be derived from a host cell or a heterologous gene. Improvements in transcription efficiency and translation efficiency of the target protein gene can be achieved by modifying the expression regulatory sequences of genes such as promoters. For example, improvement in transcription efficiency of a target protein gene can be achieved by replacing the promoter of the target protein gene with a stronger promoter.
- Animal cell culture is carried out under conditions in which the phosphate concentration and / or potassium concentration in the medium is increased. Culturing of animal cells may be carried out, for example, under conditions in which at least the concentration of phosphoric acid in the medium is increased. "Conditions in which the phosphoric acid concentration and / or potassium concentration in the medium is enhanced” are also referred to as “phosphoric acid / potassium enhancing conditions”. The “condition for enhancing the phosphoric acid concentration in the medium” is also particularly referred to as the “phosphoric acid enhancing condition”. "Conditions in which the potassium concentration in the medium is enhanced” are also particularly referred to as “potassium-enhanced conditions".
- Control conditions include general conditions used for culturing animal cells. Specific examples of the control condition include conditions that do not satisfy the phosphoric acid / potassium enhancement conditions exemplified below.
- the culture results of animal cells can be improved. Specifically, by culturing the animal cells under the phosphate / potassium enhancing condition, the culture performance of the animal cells can be improved as compared with the case where the animal cells are cultured under the control condition. Further, for example, by culturing the animal cells under the condition that both the phosphate concentration and the potassium concentration in the medium are enhanced, the animal cells are cultured under the condition that only one of the phosphate concentration and the potassium concentration in the medium is enhanced. The culture performance of animal cells may be improved as compared with the case. Improvements in the culture results of animal cells include improvement of animal cell proliferation, improvement of animal cell viability, improvement of target substance production by animal cells, and extension of animal cell culture period.
- culturing animal cells under phosphate / potassium-enhanced conditions may, for example, improve the proliferation and / or viability of the animal cells.
- culturing animal cells under phosphate / potassium-enhanced conditions may improve the proliferation and / or viability of the animal cells as compared to culturing the animal cells under control conditions.
- the viable cell density of the animal cells is increased during culturing, for example, 1.5 times or more, 2 times or more, 2.5 times or more the viable cell density at the start of culturing.
- the density may reach times or more, 80 times or more, 90 times or more, 100 times or more, 200 times or more, 500 times or more, or 1000 times or more.
- the viable cell density of the animal cells can be increased, for example, 1.5 ⁇ 10 7 cells / mL or more and 1.6 ⁇ 10 7 cells / mL or more during the culture.
- the density may reach 5 ⁇ 10 7 cells / mL or more, 7 ⁇ 10 7 cells / mL or more, 1 ⁇ 10 8 cells / mL or more, or 1 ⁇ 10 9 cells / mL or more.
- the viable cell count of animal cells can be measured, for example, using a live-and-dead cell autoanalyzer Vi-CELL TM XR (Beckman Coulter) or a flow cytometer guava easy Cyte (Luminex).
- the animal cell has a target substance-producing ability such as a target protein-producing ability
- culturing the animal cell under the phosphate / potassium-enhanced condition improves, for example, the production of the target substance such as the target protein by the animal cell.
- the animal cell has a target substance-producing ability such as a target protein-producing ability
- the animal cell is cultured under the phosphate / potassium-enhanced condition as compared with the case where the animal cell is cultured under the control condition.
- the production of target substances such as target proteins by animal cells may be improved.
- the culture period of animal cells may be extended by culturing animal cells under phosphate / potassium enhancing conditions. Specifically, by culturing the animal cells under the phosphate / potassium-enhanced condition, the culture period of the animal cells may be extended as compared with the case where the animal cells are cultured under the control condition.
- the extension of the culture period of the animal cells includes the extension of the period during which the growth of the animal cells continues, the extension of the period during which the animal cells are maintained (that is, the survival of the animal cells), and the extension of the target substance such as the target protein by the animal cells. There is an extension of the period during which production continues.
- one aspect of the method of the present invention may be a method of improving the proliferation of animal cells.
- one aspect of the method of the present invention may be a method for improving the survival rate of animal cells.
- one aspect of the method of the present invention may be a method for improving the production of a target substance by an animal cell.
- one aspect of the method of the present invention may be a method of extending the culture period of animal cells.
- “Culture of animal cells” is not limited to those intended for the proliferation of animal cells, but also includes those not intended for the proliferation of animal cells, such as maintenance of animal cells and production of target substances by animal cells. Good.
- the medium composition and culture conditions are not particularly limited as long as the purpose of culturing animal cells can be achieved, except that the phosphoric acid concentration and / or potassium concentration in the medium is enhanced.
- the medium composition and culture conditions can be configured so that animal cells proliferate.
- the purpose is to maintain animal cells
- the medium composition and culture conditions can be configured so that the animal cells are maintained (that is, the animal cells survive).
- the purpose is to produce a target substance such as a target protein
- the medium composition and culture conditions can be configured so that the target substance is produced (for example, the target protein is expressed). If not intended for the growth of animal cells, the animal cells may or may not grow during culturing.
- the animal cells may proliferate during culturing, even if they are not intended for the growth of the animal cells.
- the medium composition and culture conditions can be appropriately set according to various conditions such as the type of animal cells.
- the culture is carried out using, for example, the normal medium and normal conditions used for culturing animal cells as they are or with appropriate modifications, except that the phosphoric acid concentration and / or potassium concentration in the medium is enhanced. can do.
- Culturing can be carried out using, for example, a liquid medium.
- Specific media that can be used for culturing animal cells include D-MEM (Dulbecco's Modified Eagle Medium), CELLiST Basal Media BASAL4P (Ajinomoto Co., Ltd.), Opti-MEM (Thermo Fisher Scientific), and RPMI 1640 (Thermo Fisher Scientific).
- the medium contains various medium components such as carbon source, amino acid, vitamin, inorganic salt, phosphoric acid, choline, amine, pH buffer, growth factor, serum, serum albumin, selective drug, gene expression inducer and the like. You can. Glucose is mentioned as a carbon source.
- amino acids examples include 20 types of amino acids constituting proteins and their derivatives.
- the amino acid may be, for example, L-form.
- Specific examples of amino acids include glutamine and serine.
- glutamine examples include L-glutamine.
- Examples of serine include L-serine.
- Amines include 1,4-butanediamine (also called "putresin"), agmatin, methylamine, dimethylamine, trimethylamine, ethylamine, diethylamine, triethylamine, ethylenediamine, ethanolamine, diethanolamine, triethanolamine, N, N-diisopropyl.
- Examples thereof include ethylamine, tetramethylethylenediamine, hexamethylenediamine, spermidine, spermin and amantazine.
- aromatic amines include aniline, phenethylamine, toluidine, catecholamines, and 1,8-bis (dimethylamino) naphthalene.
- Heterocyclic amines include pyrrolidine, piperidine, piperidine, morpholin, quinuclidine, 1,4-diazabicyclo [2.2.2] octane, pyrrole, pyrazole, imidazole, pyridine, pyridazine, pyrimidine, pyrazine, oxazole, thiazole, 4-dimethyl.
- Aminopyridine can be mentioned.
- Amines include, among others, 1,4-butanediamine, ethanolamine, spermidine, spermine. Amines include, more particularly, 1,4-butanediamine.
- any component that can form a salt may be used as a free form, as a salt, or as a combination thereof. That is, for example, the term “amino acid” may mean a free amino acid, a salt thereof, or a combination thereof, unless otherwise specified. Further, for example, the term “phosphoric acid” may mean free phosphoric acid, or a salt thereof, or a combination thereof, unless otherwise specified. Also, for example, the term “choline” may mean free choline, or a salt thereof, or a combination thereof, unless otherwise specified. Further, for example, the term “amine” may mean a free amine, a salt thereof, or a combination thereof, unless otherwise specified.
- the salt is not particularly limited as long as it can be used for culturing animal cells.
- a salt for an acidic group such as a phosphoric acid group or a carboxyl group
- a salt with an alkali metal such as ammonium salt, sodium and potassium
- a salt with an alkaline earth metal such as calcium and magnesium
- an amine as exemplified above.
- salts with basic amino acids such as arginine and lysine.
- a salt for a basic group such as an amino group
- a salt with an inorganic acid such as hydrochloric acid, sulfuric acid, phosphoric acid, nitric acid, hydrobromic acid, acetic acid, citric acid, benzoic acid, maleic acid and fumaric acid.
- Salts with organic carboxylic acids such as tartaric acid, succinic acid, tannic acid, butyric acid, hibenzic acid, pamoic acid, enanthic acid, decanoic acid, theocric acid, salicylic acid, lactic acid, oxalic acid, mandelic acid, malic acid, methanesulfonic acid , Benzylsulfonic acid, p-toluenesulfonic acid and other salts with organic sulfonic acids.
- examples of the salt of phosphoric acid include sodium dihydrogen phosphate, disodium hydrogen phosphate, potassium dihydrogen phosphate, and dipotassium hydrogen phosphate.
- choline salt includes choline bitartrate.
- the seeding amount of animal cells at the start of culture is, for example, 1 ⁇ 10 3 cells / mL or more, 1 ⁇ 10 4 cells / mL or more, 1 ⁇ 10 5 cells / mL or more, 1 ⁇ 10 6 cells / mL or more, or It may be 1 ⁇ 10 7 cells / mL or more, 1 ⁇ 10 10 cells / mL or less, 1 ⁇ 10 9 cells / mL or less, 1 ⁇ 10 8 cells / mL or less, 1 ⁇ 10 7 cells / mL or less, It may be 1 ⁇ 10 6 cells / mL or less, 1 ⁇ 10 5 cells / mL or less, or 1 ⁇ 10 4 cells / mL or less.
- the culture may be divided into a seed culture and a main culture.
- the culture conditions of the seed culture and the main culture may or may not be the same.
- the target substance may be produced at least during the main culture period.
- animal cells may be sufficiently proliferated by seed culture, and then a target substance such as a target protein may be produced by main culture.
- Culturing can be carried out by batch culture, fed-batch culture, continuous culture, or a combination thereof.
- fed-batch culture or continuous culture include perfusion culture.
- the medium at the start of culturing is also referred to as "initial medium” or “basal medium”.
- the medium supplied to the culture system (for example, the initial medium) in the fed-batch culture or the continuous culture is also referred to as "fed-batch medium”.
- supplying a fed-batch medium to a culture system in fed-batch culture or continuous culture is also referred to as "fed-batch".
- Fed-batch may be carried out throughout the period of the culture or only for a part of the period of the culture. Fed-batch may be carried out continuously or intermittently.
- withdrawal of the culture broth may be carried out.
- the withdrawal of the culture solution may be, for example, withdrawal of the culture solution containing animal cells or withdrawal of the culture supernatant.
- animal cells may be collected from the extracted culture medium and returned to the culture system.
- Withdrawal of the culture medium may be carried out throughout the whole period of the culture, or may be carried out only during a part of the culture.
- the withdrawal of the culture solution may be carried out continuously or intermittently.
- the withdrawal and feeding of the culture medium may or may not be performed at the same time.
- the amount of the culture solution to be withdrawn is preferably the same as the amount of the fed-batch medium to be fed.
- the “equivalent amount” may mean, for example, an amount of 93 to 107% (v / v) with respect to the amount of fed-batch medium to be fed.
- the culture forms of the seed culture and the main culture may or may not be the same.
- Various components such as phosphoric acid may be contained in the initial medium, the fed-batch medium, or both. That is, in the process of culturing, various components such as phosphoric acid may be supplied to the medium alone or in any combination. All of these components may be supplied once or multiple times, or may be supplied continuously.
- the composition of the initial medium and the fed-batch medium eg, the type and / or concentration of the components contained
- the composition of the initial medium and the fed-batch medium may or may not be the same. That is, the type of the component contained in the initial medium may or may not be the same as the type of the component contained in the fed-batch medium. Further, the concentration of each component contained in the initial medium may or may not be the same as the concentration of each component contained in the fed-batch medium.
- the composition of the initial medium and the fed-batch medium may be the same.
- two or more fed-batch media having different compositions may be used.
- the composition of each fed-batch medium may or may not be the same.
- various components such as phosphoric acid may be supplied to the medium in a form such as powder that is not contained in the fed-batch medium.
- the culture may be carried out in a CO 2 containing atmosphere such as 5% CO 2.
- the pH of the medium may be, for example, near neutral. "Near neutral” may mean, for example, pH 6-8, pH 6.5-7.5, or pH 6.8-7.2.
- the pH of the medium can be adjusted as needed.
- the pH of the medium is various alkaline or acidic substances such as ammonia gas, aqueous ammonia, sodium carbonate, sodium bicarbonate, potassium carbonate, potassium bicarbonate, magnesium carbonate, sodium hydroxide, potassium hydroxide, calcium hydroxide, magnesium hydroxide, etc. Can be adjusted using.
- the culture temperature may be, for example, 36-38 ° C.
- the culture period is, for example, 0.5 days or more, 1 day or more, 2 days or more, 3 days or more, 4 days or more, 5 days or more, 6 days or more, 7 days or more, 8 days or more, 9 days or more, 10 days. More than 12 days, more than 15 days, or more than 20 days, 50 days or less, 40 days or less, 30 days or less, 25 days or less, 20 days or less, 15 days or less, 12 days or less, 10 days Hereinafter, it may be 9 days or less, 8 days or less, or 7 days or less, and may be a consistent combination thereof. Specifically, the culture period may be, for example, 1 to 30 days, 3 to 25 days, or 5 to 20 days. At the time of culturing, gene expression such as expression of the target protein may be induced as appropriate.
- Various components such as phosphoric acid may be present in the medium in any form.
- a component is a component that can be ionized, the component may or may not be ionized in the medium.
- Concentration of a certain component may mean the total concentration of the dissolved component when the component can be present in a liquid in a plurality of forms. That is, for example, "concentration of a certain component” may mean the sum of the concentration of the molecule and the concentration of ions of the component when the component can be present in the liquid in the form of molecules and ions.
- concentration of a component in a medium may mean the total concentration of the component dissolved in the medium when the component can be present in the medium in multiple forms. That is, for example, "concentration of a certain component in the medium” means the sum of the concentration of the molecule of the component and the concentration of ions in the medium when the component can be present in the medium in the form of molecules and ions. May mean.
- phosphoric acid concentration may mean the total concentration of dissolved phosphoric acid molecular species. That is, the “phosphoric acid concentration", specifically, the concentration of molecular phosphoric acid (H 3 PO 4) and phosphoric acid ions (PO 4 3-, HPO 4 2- , and H 2 PO 4 -) concentration May mean the sum of.
- phosphoric acid concentration in the medium may mean the total concentration of phosphoric acid molecular species dissolved in the medium. That is, for example, the "phosphoric acid concentration in the medium”, specifically, phosphoric acid ions in the concentration and medium phosphate molecules in the medium (H 3 PO 4) (PO 4 3-, HPO 4 2 -, and H 2 PO 4 -) may refer to the total concentration of.
- the degree of enhancement of the phosphoric acid concentration and / or potassium concentration in the medium is not particularly limited as long as the culture performance of the animal cells (for example, the proliferation of the animal cells or the production of the target substance by the animal cells) is improved.
- the degree of enhancement of the phosphoric acid concentration and / or potassium concentration in the medium can be appropriately set according to various conditions such as the type of animal cells, the length of the culture period, and the desired production amount of the target substance.
- “Culturing is carried out under conditions of enhanced phosphate and / or potassium concentration in the medium” means, for example, that the phosphate and / or potassium concentration in the medium is within a predetermined range. Good.
- the phosphoric acid concentration in the medium is, for example, 0.2 mM or more, 0.5 mM or more, 1 mM or more, 1.5 mM or more, 2 mM or more, 2.5 mM or more, 3 mM or more, 3.5 mM or more, 4 mM or more, 4.5 mM.
- the phosphoric acid concentration in the medium may be 4 mM or more, in particular.
- the phosphoric acid concentration in the medium may be 11 mM or more, more particularly.
- the phosphoric acid concentration in the medium may be, for example, 0.2 to 100 mM, 4 to 70 mM, 11 to 50 mM, 11 to 40 mM, 11 to 30 mM, 11 to 27 mM, or 10 to 25 mM. ..
- the potassium concentration in the medium is, for example, 0.2 mM or more, 0.5 mM or more, 1 mM or more, 2 mM or more, 3 mM or more, 4 mM or more, 5 mM or more, 6 mM or more, 7 mM or more, 8 mM or more, 9 mM or more, or 10 mM or more. It may be 50 mM or less, 40 mM or less, 30 mM or less, 20 mM or less, 15 mM or less, 12 mM or less, 10 mM or less, 9 mM or less, 8 mM or less, 7 mM or less, 6 mM or less, or 5 mM or less, and their contradictions.
- the potassium concentration in the medium may be, in particular, 1 mM or higher. Specifically, the potassium concentration in the medium may be, for example, 0.2 to 50 mM, 0.5 to 30 mM, or 1 to 10 mM.
- Phosphoric acid concentration and / or potassium concentration in the medium may be enhanced during the entire period of the culture, or may be enhanced only during a part of the culture. That is, "the culture is carried out under the condition that the phosphoric acid concentration and / or the potassium concentration in the medium is enhanced” means that the phosphoric acid concentration and / or the potassium concentration in the medium is enhanced during at least a part of the culture. It is sufficient, and it is not necessary that the phosphate concentration and / or potassium concentration in the medium is enhanced during the entire period of the culture.
- the phosphoric acid concentration and / or potassium concentration in the medium may be increased to the above-exemplified concentration during the entire period of the culture, or may be increased to the above-exemplified concentration only during a part of the culture.
- Phosphoric acid and / or potassium may be contained in the medium at the above-exemplified concentration at the start of culturing, or may be supplied to the medium at the above-exemplified concentration after the start of culturing. Further, phosphoric acid and / or potassium is contained in the medium at the above-exemplified concentration at the start of culturing, and is further supplied to the medium at the above-exemplified concentration after the start of culturing (for example, after consumption). You may. When the culture is divided into seed culture and main culture, the phosphate concentration and / or potassium concentration in the medium should be at least during the main culture, that is, during the entire main culture or a part of the main culture.
- the phosphate concentration and / or potassium concentration in the medium may or may not be enhanced during the seed culture period.
- the description about the culture for example, “culture period (culture period)” or “culture start" can be read as that for the main culture.
- the “partial period” is not particularly limited as long as the culture performance of the animal cells (for example, the proliferation of the animal cells and the production of the target substance by the animal cells) is improved.
- the “partial period” can be appropriately set according to various conditions such as the type of animal cells, the length of the culture period, and the desired production amount of the target substance.
- “Partial period” is, for example, 50% or more, 60% or more, 70% or more, 80% or more, 90% or more, 95% or more, 97% or more, or 99% or more of the entire culture period. It may be there.
- the "partial period” is, for example, 0.5 days or more, 1 day or more, 2 days or more, 3 days or more, 4 days or more, 5 days or more, 6 days or more, 7 days or more, 8 days or more, The period may be 9 days or more, 10 days or more, 12 days or more, or 15 days or more.
- the phosphoric acid concentration and / or potassium concentration in the medium may be enhanced to the above-exemplified concentration, for example, as an average value over the entire culture period. That is, "culture is carried out under the condition that the phosphate concentration and / or potassium concentration in the medium is increased to a certain concentration” or "culture is carried out in a medium having a certain phosphoric acid concentration and / or potassium concentration”. By may mean that the average value of phosphoric acid concentration and / or potassium concentration in the medium throughout the whole period of the culture is within the range of the concentration.
- the "average value of phosphoric acid concentration and / or potassium concentration in the medium throughout the entire culture period” is not particularly limited as long as the fluctuation of phosphoric acid concentration and / or potassium concentration during the entire culture period can be grasped. May mean, for example, the average value of phosphate and / or potassium concentration in the medium measured every 60 minutes, every 30 minutes, every 20 minutes, or every 10 minutes throughout the culture.
- the culture is carried out under the condition that the concentration of phosphoric acid and / or the concentration of potassium in the medium is increased” means that, for example, the amount of phosphoric acid and / or potassium supplied to the medium is within a predetermined range. It may mean.
- the amount of phosphoric acid supplied to the medium is, for example, 0.05 mM or more, 0.1 mM or more, 0.15 mM or more, 0.2 mM or more, 0.25 mM or more, 0.3 mM or more per day throughout the culture.
- 0.35 mM or more 0.35 mM or more, 0.4 mM or more, 0.45 mM or more, 0.5 mM or more, 0.55 mM or more, 0.6 mM or more, 0.65 mM or more, 0.7 mM or more, 0.75 mM or more, 0.8 mM or more , 0.85 mM or more, 0.9 mM or more, 0.95 mM or more, or 1 mM or more, 5 mM or less, 4 mM or less, 3 mM or less, 2 mM or less, or 1 mM or less, and there is no contradiction between them. It may be a combination.
- the amount of phosphoric acid supplied to the medium may be 0.05 mM or more per day, especially throughout the culture.
- the amount of phosphoric acid supplied to the medium may be 0.5 mM or more per day, more particularly throughout the entire culture.
- the amount of phosphoric acid supplied to the medium is, for example, 0.05 to 5 mM, 0.1 to 4 mM, 0.4 to 3 mM, or 0.7 to 2 mM per day throughout the culture. It may be.
- the amount of potassium supplied to the medium is, for example, 0.05 mM or more, 0.1 mM or more, 0.15 mM or more, 0.2 mM or more, 0.25 mM or more, 0.3 mM or more per day throughout the culture.
- the amount of potassium supplied to the medium may be 0.2 mM or more per day, especially throughout the culture. Specifically, the amount of potassium supplied to the medium may be, for example, 0.05 to 5 mM, 0.1 to 3 mM, or 0.2 to 1 mM per day throughout the culture.
- Phosphoric acid and / or potassium may be continuously supplied to the medium or intermittently supplied to the medium, for example. Phosphoric acid and / or potassium may be supplied to the medium daily or every few days, for example.
- Supply of phosphoric acid and / or potassium to the medium for example, viable cell density of the animal cells 1.5 ⁇ 10 6 cells / mL or more, 1.6 ⁇ 10 6 cells / mL or more, 1.7 ⁇ 10 6 cells / mL or more, 1.8 ⁇ 10 6 cells / mL or more, 1.9 ⁇ 10 6 cells / mL or more, 2 ⁇ 10 6 cells / mL or more, 3 ⁇ 10 6 cells / mL or more, 5 ⁇ 10 6 cells It may be carried out at a density of / mL or higher, 7 ⁇ 10 6 cells / mL or higher, 1 ⁇ 10 7 cells / mL or higher, or 1 ⁇ 10 8 cells / mL or higher.
- Culturing may be carried out in the presence of amines, choline, and / or serins. Culturing may be carried out, for example, in the presence of amines, choline, and / or serins, at least when the phosphate concentration in the medium is enhanced. Culturing may be carried out, for example, in the presence of at least amine. Culturing may be carried out, for example, in the presence of at least choline and / or serin.
- “Culture is performed in the presence of amines, choline, and / or serin” may mean, for example, that the medium contains amines, choline, and / or serine.
- the amine concentration in the medium is, for example, 0.001 mM or more, 0.002 mM or more, 0.005 mM or more, 0.01 mM or more, 0.02 mM or more, 0.03 mM or more, 0.04 mM or more, 0.05 mM or more, 0.
- the amine concentration in the medium may be, in particular, 0.002 mM or more.
- the amine concentration in the medium may be, more particularly, 0.007 mM or more.
- the amine concentration in the medium may be, for example, 0.002-0.5 mM, 0.005-0.2 mM, or 0.007-0.1 mM.
- the choline concentration in the medium is, for example, 0.1 mM or more, 0.2 mM or more, 0.5 mM or more, 1 mM or more, 2 mM or more, 3 mM or more, 4 mM or more, 5 mM or more, 6 mM or more, 7 mM or more, 8 mM or more, 9 mM or more.
- the choline concentration in the medium may be 0.2 mM or more, in particular.
- the choline concentration in the medium may be, more particularly, 1 mM or more.
- the choline concentration in the medium may be, for example, 0.2 to 50 mM, 0.5 to 20 mM, or 1 to 10 mM.
- the serine concentration in the medium is, for example, 0.5 mM or more, 1 mM or more, 2 mM or more, 3 mM or more, 4 mM or more, 5 mM or more, 6 mM or more, 7 mM or more, 8 mM or more, 9 mM or more, 10 mM or more, 12 mM or more, 15 mM or more, It may be 20 mM or more, 25 mM or more, 30 mM or more, 35 mM or more, 40 mM or more, 50 mM or more, 60 mM or more, 70 mM or more, 80 mM or more, 90 mM or more, or 100 mM or more, 500 mM or less, 400 mM or less, 300 mM or less, 200 mM.
- the serine concentration in the medium may be particularly 2 mM or more.
- the serine concentration in the medium may be more than 10 mM, more particularly.
- the serine concentration in the medium may be, for example, 1 to 200 mM, 2 to 100 mM, 4 to 50 mM, 2 to 500 mM, 5 to 200 mM, or 10 to 100 mM.
- Amine, choline, and / or serine may be contained in the medium for the entire period of the culture, or may be contained in the medium only for a part of the period of the culture. That is, "culture is performed in the presence of amines, choline, and / or serine" or "culture is performed in a medium containing amine, choline, and / or serine” means that amine, choline, and It is sufficient that / or serine is contained in the medium for at least a part of the culture, and it is not necessary that amine, choline, and / or serine be contained in the medium for the entire duration of the culture.
- Amine, choline, and / or serine may be contained in the medium at the above-exemplified concentrations, for example, during the entire period of the culture, and may be contained in the medium at the above-exemplified concentrations only during a part of the culture. May be good. That is, "culture is carried out in the presence of certain amine, choline and / or serine" or "culture is carried out in medium of certain amine, choline and / or serine concentration".
- the amine concentration, choline concentration, and / or serine concentration in the medium is within the range of the concentration during at least a part of the culture, and the amine concentration, choline concentration, and / or serine concentration in the medium is It does not have to be within the concentration range for the entire duration of the culture.
- the amine concentration, choline concentration, and / or serine concentration in the medium may be set to the above-exemplified concentration, for example, as an average value over the entire culture period. That is, "culture is carried out in the presence of certain amine, choline, and / or serine” or "culture is carried out in medium of certain amine, choline, and / or serine concentrations.” May mean that the average value of amine, choline, and / or serine concentrations in the medium throughout the culture is within that concentration range.
- the "average value of amine concentration, choline concentration, and / or serine concentration in the medium throughout the entire culture period” means that the fluctuation of amine concentration, choline concentration, and / or serine concentration during the entire culture period can be grasped. This is not particularly limited, but for example, the amine concentration, choline concentration, and / or serine concentration in the medium measured every 60 minutes, 30 minutes, 20 minutes, or 10 minutes throughout the culture period. It may mean an average value.
- culture is carried out in the presence of amines, choline, and / or serin may mean, for example, that the medium is supplied with amine, choline, and / or serine.
- the amount of amine supplied to the medium is, for example, 0.0002 mM or more, 0.0005 mM or more, 0.001 mM or more, 0.0015 mM or more, 0.002 mM or more, 0.0025 mM or more, per day throughout the culture. It may be 0.003 mM or more, 0.0035 mM or more, 0.004 mM or more, 0.0045 mM or more, or 0.005 mM or more, 0.025 mM or less, 0.02 mM or less, 0.015 mM or less, 0.01 mM or less.
- the supply of amine to the medium may be 0.0005 mM or more per day, especially throughout the culture.
- the amount of amine supplied to the medium is, for example, 0.0005 to 0.025 mM mM, 0.001 to 0.015 mM, 0.002 to 0.007 mM, or 0.003 to 0 per day throughout the culture. It may be .004 mM.
- the amount of choline supplied to the medium is, for example, 0.02 mM or more, 0.05 mM or more, 0.1 mM or more, 0.15 mM or more, 0.2 mM or more, 0.25 mM or more per day throughout the culture. It may be 0.3 mM or more, 0.35 mM or more, 0.4 mM or more, 0.45 mM or more, or 0.5 mM or more, 2.5 mM or less, 2 mM or less, 1.5 mM or less, 1 mM or less, 0.7 mM. Hereinafter, it may be 0.5 mM or less, 0.2 mM or less, or 0.1 mM or less, and may be a consistent combination thereof.
- the supply of choline to the medium may be 0.05 mM or more per day, especially throughout the culture.
- the supply of choline to the medium is, for example, 0.05-2.5 mM, 0.1-1.5 mM, 0.2-0.7 mM, or 0.3-0 per day throughout the culture. It may be .5 mM.
- the amount of serin supplied to the medium is, for example, 0.1 mM or more, 0.2 mM or more, 0.3 mM or more, 0.4 mM or more, 0.5 mM or more, 0.6 mM or more, per day throughout the culture.
- the supply of serin to the medium may be 0.5 mM or more per day, especially throughout the culture.
- the amount of serin supplied to the medium is, for example, 0.2 to 10 mM, 0.4 to 5 mM, 0.8 to 3 mM, 0.5 to 25 mM, 1 to 15 mM, 2 to per day throughout the culture. It may be 7 mM, or 3.5-5 mM.
- Amine, choline, and / or serine may be supplied to the medium continuously or intermittently, for example. Amines, choline, and / or serine may be supplied to the medium daily or every few days, for example.
- the phosphoric acid concentration in the fed-batch medium is not particularly limited as long as the desired amount of phosphoric acid can be supplied.
- the phosphoric acid concentration in the fed-batch medium may be, for example, the phosphoric acid concentration in the above-exemplified medium.
- the phosphoric acid concentration in the fed-batch medium may be the phosphoric acid concentration in the above-exemplified medium.
- the phosphoric acid concentration in the fed-batch medium is, for example, more than 1 times, 1.1 times or more, 1.3 times or more, 1.5 times or more, 2 times or more the phosphoric acid concentration in the above-exemplified medium.
- the concentration may be 3 times or more, 5 times or more, 7 times or more, 10 times or more, 15 times or more, or 20 times or more, and 100 times or less, 70 times or less, 50 times or less, 30 times or less, 20 times or more.
- the concentration may be 10 times or less, or 5 times or less, and may be a consistent combination thereof.
- the phosphoric acid concentration in the fed-batch medium may be, for example, 10 mM or more, 20 mM or more, 30 mM or more, 50 mM or more, 70 mM or more, 100 mM or more, 150 mM or more, or 200 mM or more, 1000 mM or less, 700 mM or less, It may be 500 mM or less, 300 mM or less, 200 mM or less, 100 mM or less, 70 mM or less, or 50 mM or less, and may be a consistent combination thereof. Specifically, the phosphoric acid concentration in the fed-batch medium may be, for example, 10 to 1000 mM, 30 to 500 mM, or 50 to 200 mM.
- the amine concentration in the fed-batch medium is not particularly limited as long as the desired amount of amine can be supplied.
- the amine concentration in the fed-batch medium may be, for example, the amine concentration in the above-exemplified medium.
- the amine concentration in the fed-batch medium may be the amine concentration in the above-exemplified medium.
- the amine concentration in the fed-batch medium is, for example, more than 1 times, 1.1 times or more, 1.3 times or more, 1.5 times or more, 2 times or more, or 3 times the amine concentration in the above-exemplified medium.
- the concentration may be 5 times or more, 7 times or more, 10 times or more, 15 times or more, or 20 times or more, 100 times or less, 70 times or less, 50 times or less, 30 times or less, 20 times or less,
- the concentration may be 10 times or less, or 5 times or less, and may be a consistent combination thereof.
- the amine concentration in the fed-batch medium is, for example, 0.05 mM or more, 0.1 mM or more, 0.2 mM or more, 0.3 mM or more, 0.5 mM or more, 0.7 mM or more, 1 mM or more, or 1 mM or more.
- the amine concentration in the fed-batch medium may be, for example, 0.05 to 5 mM, 0.1 to 3 mM, or 0.2 to 1 mM.
- the choline concentration in the fed-batch medium is not particularly limited as long as the desired amount of choline can be supplied.
- the choline concentration in the fed-batch medium may be, for example, the choline concentration in the above-exemplified medium.
- the choline concentration in the fed-batch medium may be the choline concentration in the above-exemplified medium.
- the choline concentration in the fed-batch medium is, for example, more than 1 times, 1.1 times or more, 1.3 times or more, 1.5 times or more, 2 times or more, and 3 times the choline concentration in the above-exemplified medium.
- the concentration may be 5 times or more, 7 times or more, 10 times or more, 15 times or more, or 20 times or more, 100 times or less, 70 times or less, 50 times or less, 30 times or less, 20 times or less,
- the concentration may be 10 times or less, or 5 times or less, and may be a consistent combination thereof.
- the choline concentration in the fed-batch medium may be, for example, 5 mM or more, 10 mM or more, 20 mM or more, 30 mM or more, 50 mM or more, 70 mM or more, or 100 mM or more, and 500 mM or less, 300 mM or less, 200 mM or less, 100 mM.
- the choline concentration in the fed-batch medium may be, for example, 5 to 500 mM, 10 to 300 mM, or 20 to 100 mM.
- the serine concentration in the fed-batch medium is not particularly limited as long as the desired amount of serine to be supplied can be obtained.
- the serine concentration in the fed-batch medium may be, for example, the serine concentration in the above-exemplified medium.
- the serine concentration in the fed-batch medium may be the serine concentration in the above-exemplified medium.
- the serine concentration in the fed-batch medium is, for example, more than 1 times, 1.1 times or more, 1.3 times or more, 1.5 times or more, 2 times or more and 3 times the serine concentration in the above-exemplified medium.
- the concentration may be 5 times or more, 7 times or more, 10 times or more, 15 times or more, or 20 times or more, 100 times or less, 70 times or less, 50 times or less, 30 times or less, 20 times or less, The concentration may be 10 times or less, or 5 times or less, and may be a consistent combination thereof.
- the serine concentration in the fed-batch medium may be, for example, 20 mM or more, 30 mM or more, 50 mM or more, 70 mM or more, 100 mM or more, 200 mM or more, 300 mM or more, 500 mM or more, 700 mM or more, or 1000 mM or more, and 5000 mM.
- the serine concentration in the fed-batch medium may be, for example, 30 to 2000 mM, 50 to 1000 mM, 70 to 500 mM, 50 to 5000 mM, 100 to 3000 mM, or 200 to 1000 mM.
- the concentration of various components can be measured, for example, by a known method used for detecting or identifying a compound. Such methods include, for example, HPLC, UPLC, LC / MS, GC / MS, NMR. These methods can also be used to confirm that the target substance has been produced. One of these methods may be used alone, or two or more thereof may be used in combination as appropriate.
- Animal cells can be cultured as described above.
- the target substance is produced (for example, the target protein is expressed) by culturing the animal cell as described above, and thus the target substance is contained.
- the culture to be used is obtained.
- the target substance such as the target protein may be accumulated in the medium, the cell surface, the cell, or a combination thereof.
- the production of the target protein can be confirmed by a known method used for protein detection or identification.
- a known method used for protein detection or identification examples include SDS-PAGE, Western blotting, mass spectrometry, N-terminal amino acid sequence analysis, and enzyme activity measurement.
- SDS-PAGE SDS-PAGE
- Western blotting Western blotting
- mass spectrometry mass spectrometry
- N-terminal amino acid sequence analysis N-terminal amino acid sequence analysis
- enzyme activity measurement One of these methods may be used alone, or two or more thereof may be used in combination as appropriate.
- the target protein can be recovered as appropriate. Specifically, the target protein can be recovered as an appropriate fraction containing the target protein. Examples of such a fraction include cultures, culture supernatants, cultured cells, and processed products of cultured cells (crushed products, lysates, extracts (cell-free extracts). Cultured cells include, for example, It may be obtained in the form of immobilized cells immobilized with a carrier such as acrylamide or carrageenan.
- the target protein may be further purified to the desired degree.
- the target protein When the target protein accumulates in the medium, the target protein can be purified from the supernatant after removing the solid content of cells or the like from the culture by centrifugation or the like.
- the target protein When the target protein accumulates in the cell, the target protein can be purified from the processed product after being subjected to a treatment such as crushing, lysing, or extracting the cell, for example.
- the cells can be recovered from the culture by centrifugation or the like.
- Treatments such as cell disruption, lysis, and extraction can be performed by known methods. Examples of such a method include an ultrasonic crushing method, a dynomill method, a bead crushing method, a French press crushing method, and a lysozyme treatment. One of these methods may be used alone, or two or more thereof may be used in combination as appropriate.
- the target protein When the target protein accumulates on the cell surface, the target protein can be purified from the solubilized product after being solubilized, for example.
- Solubilization can be performed by a known method. Such methods include, for example, increasing the salt concentration and using a surfactant. One of these methods may be used alone, or two or more thereof may be used in combination as appropriate.
- Purification of the target protein can be performed by a known method used for protein purification.
- a known method used for protein purification include sulfur fractionation, ion exchange chromatography, hydrophobic chromatography, affinity chromatography, gel filtration chromatography, and isoelectric point precipitation.
- sulfur fractionation ion exchange chromatography
- hydrophobic chromatography affinity chromatography
- gel filtration chromatography gel filtration chromatography
- isoelectric point precipitation One of these methods may be used alone, or two or more thereof may be used in combination as appropriate.
- the target protein may be obtained in a free state or in a state of an immobilized enzyme immobilized on a solid phase such as a resin.
- the recovered target protein may be appropriately formulated.
- the dosage form is not particularly limited and can be appropriately set according to various conditions such as the intended use of the target protein. Dosage forms include, for example, liquids, suspensions, powders, tablets, pills and capsules. In formulation, pharmacologically acceptable additives such as excipients, binders, disintegrants, lubricants, stabilizers, flavoring agents, odorants, fragrances, diluents, surfactants, etc. Can be used.
- the medium of the present invention is a medium for culturing animal cells in which the phosphoric acid concentration and / or the potassium concentration is enhanced.
- the medium of the present invention can be used, for example, in the method of the present invention.
- the description of the medium used in the method of the present invention can be applied mutatis mutandis.
- the medium of the present invention contains phosphoric acid and / or potassium.
- the medium of the present invention may further contain amines, choline, and / or serine.
- the medium of the present invention may contain amines, choline, and / or serine, for example, if it contains at least phosphoric acid.
- the medium of the present invention may contain, for example, at least an amine.
- the medium of the present invention may contain, for example, at least choline and / or serine.
- the medium of the present invention may be, for example, a basal medium (initial medium) or a fed-batch medium.
- the fed-batch medium may be used for, for example, fed-batch culture or continuous culture.
- the fed-batch medium may be, for example, one used for perfusion culture.
- the description of the phosphoric acid concentration in the medium (including the description of the phosphoric acid concentration in the feed medium) in the method of the present invention exemplified above can be applied mutatis mutandis.
- the description of the potassium concentration in the medium (including the description of the potassium concentration in the fed-batch medium) in the method of the present invention exemplified above can be applied mutatis mutandis.
- the description of the amine concentration in the medium (including the description of the amine concentration in the fed-batch medium) in the method of the present invention exemplified above can be applied mutatis mutandis.
- the description of the choline concentration in the medium (including the description of the choline concentration in the fed-batch medium) in the method of the present invention exemplified above can be applied mutatis mutandis.
- the description of the serine concentration in the medium (including the description of the serine concentration in the feed medium) in the method of the present invention exemplified above can be applied mutatis mutandis.
- Example 1 Evaluation of the effect of improving CHO cell proliferation and antibody production by adding phosphoric acid to the feeding medium
- the effect of improving CHO cell proliferation and antibody production by adding phosphoric acid to the feeding medium was evaluated. evaluated.
- CHO cells cells of the CHO DG-44 lineage modified to produce the antibody Adalimumab were used.
- Basal medium CELLiST registered trademark
- Basal media (Ajinomoto Co., Ltd .: BASAL4P; containing phosphoric acid with a final concentration of 1.9 mM), LR3-IGF-1 (final concentration 10 ⁇ g / mL), sodium dextran sulfate (Final concentration 400 mg / L) and L-Glutamine (final concentration 6 mM) were added to prepare a basal medium.
- Feed media (Ajinomoto Co., Ltd .: FEED2)
- glucose final concentration 75 g / L
- sodium dihydrogen phosphate final concentration 0, 125, 250, 500, 1000
- 4000 or 7000 mg / L (ie, 0.0, 1.0, 2.1, 4.2, 8.3, 33.3, or 58.3 mM) was added to prepare seven fed-batch media with different phosphate concentrations.
- Cell culture medium was sampled on days 4, 7, 9, 11, and 14 of the start of culture, and the number of viable cells was measured using a living-and-dead cell autoanalyzer Vi-CELL TM XR (Beckman Coulter), and the amount of antibody produced. was measured using Octet QK (FORTEBIO).
- Example 2 Evaluation of the effect of improving CHO cell proliferation and antibody production by adding phosphoric acid to the feed medium
- the effect of improving CHO cell proliferation and antibody production by adding phosphoric acid to the feed medium was evaluated. evaluated.
- CHO cells cells of the CHO DG-44 lineage modified to produce the antibody Adalimumab were used.
- Basal medium CELLiST registered trademark
- Basal media (Ajinomoto Co., Ltd .: BASAL4P; containing phosphoric acid with a final concentration of 1.9 mM), LR3-IGF-1 (final concentration 10 ⁇ g / mL), diphosphate Sodium hydrogen hydrogen (115 mg / L (ie, final concentration 2.9 mM)), sodium dextran sulfate (final concentration 400 mg / L) and L-Glutamine (final concentration 6 mM) were added to prepare a basal medium.
- Basal media (Ajinomoto Co., Ltd .: BASAL4P; containing phosphoric acid with a final concentration of 1.9 mM
- LR3-IGF-1 final concentration 10 ⁇ g / mL
- diphosphate Sodium hydrogen hydrogen 115 mg / L (ie, final concentration 2.9 mM)
- sodium dextran sulfate final concentration 400 mg / L
- L-Glutamine final concentration
- Feed media (Ajinomoto Co., Ltd .: FEED2)
- glucose final concentration 75 g / L
- sodium dihydrogen phosphate final concentration 0, 1000, 2000, 3000, 4000
- 5000 mg / L ie 0.0, 8.3, 16.7, 25, 33.3, or 41.7 mM
- Cell culture medium was sampled on days 4, 7, 9, 11, and 14 of the start of culture, and the number of viable cells was measured using a living-and-dead cell autoanalyzer Vi-CELL TM XR (Beckman Coulter), and the amount of antibody produced. was measured using CEDEX Bio HT (Roche Diagnostics).
- Example 3 Evaluation of the effect of adding phosphoric acid and serine, choline, or amine to the feed medium to improve CHO cell proliferation and antibody production
- phosphoric acid and serine, choline, etc. to the feed medium
- the effect of adding amine on improving CHO cell proliferation and antibody production was evaluated.
- CHO cells cells of the CHO S lineage modified to produce the antibody Adalimumab were used.
- Basal medium CELLiST registered trademark
- Basal media (Ajinomoto Co., Ltd .: BASAL4P; containing phosphoric acid with a final concentration of 1.9 mM), LR3-IGF-1 (final concentration 10 ⁇ g / mL), diphosphate Sodium hydrogen hydrogen (115 mg / L (ie, total final concentration 2.9 mM with phosphate contained in Basal media)), sodium dextran sulfate (final concentration 400 mg / L), and L-Glutamine (final concentration 6 mM) ) was added to prepare a basal medium.
- Basal media Ajinomoto Co., Ltd .: BASAL4P
- LR3-IGF-1 final concentration 10 ⁇ g / mL
- diphosphate Sodium hydrogen hydrogen 115 mg / L (ie, total final concentration 2.9 mM with phosphate contained in Basal media)
- sodium dextran sulfate final concentration 400 mg / L
- Feed media (Ajinomoto Co., Ltd .: FEED2), glucose (final concentration 75 g / L), sodium dihydrogen phosphate (final concentration 0 or 2500 mg / L (ie, that is) 0.0 or 20.8 mM)), and if necessary, the following components (A), (B), (C), or (D) were added to prepare 10 types of fed-batch media: (A) 1,4-Butanediamine (final concentration 12.8 mg / L (ie 0.145 mM)); (B) Serine (final concentration 20,000 mg / L (ie 190 mM)), choline bitartrate (final concentration 4,400 mg / L (ie 17.4 mM)), and 1,4-butanediamine (final concentration 12.8 mg /).
- Cell culture medium was sampled on days 4, 7, 9, 11 and 14 of the start of culture, the number of viable cells was measured using a flow cytometer guava easyCyte (Luminex), and the amount of antibody produced was measured by CEDEX Bio HT (Roche. It was measured using Diagnostics).
- Luminex flow cytometer guava easyCyte
- CEDEX Bio HT Roche. It was measured using Diagnostics.
- Example 4 Evaluation of CHO cell proliferation and antibody production improving effect by addition of phosphoric acid or potassium to the feed medium
- CHO cell proliferation and CHO cell proliferation by addition of phosphoric acid or potassium to the feed medium The effect of improving antibody production was evaluated.
- CHO cells cells of the CHO S lineage modified to produce the antibody Herceptin were used.
- Cell culture medium was sampled on days 4, 7, 9, 11, and 14 of the start of culture, and the number of viable cells was measured using a living-and-dead cell autoanalyzer Vi-CELL TM XR (Beckman Coulter), and the amount of antibody produced. was measured using CEDEX Bio HT (Roche Diagnostics).
- cell culture medium was sampled on the 4th, 7th, 9th, and 11th days after the start of culture, and the glucose concentration was measured using CEDEX Bio HT (Roche Diagnostics), and the final glucose concentration was 11 g / A 500 g / L glucose aqueous solution was added so as to have L.
- Example 5 Evaluation of the effect of improving antibody production by phosphate-enriched medium in Perfusion culture
- the effect of improving CHO cell proliferation and antibody production by adding phosphate to the basal medium and flow medium was applied to Perfusion culture. Evaluated.
- CHO cells cells of the CHO S lineage modified to produce the antibody Adalimumab were used.
- the medium exchange was carried out by centrifuging the culture medium, removing it so that 3.68 mL of the centrifugal supernatant remained, adding 6.32 mL of the same fresh fed-batch medium as the basal medium, and stirring. After changing the medium, the culture was restarted. The number of living cells was measured using a living-and-dead cell autoanalyzer Vi-CELL TM XR (Beckman Coulter), and the amount of antibody produced was measured using CEDEX Bio HT (Roche Diagnostics).
- Example 6 Evaluation of the effect of improving antibody production by the phosphate-enriched medium in Perfusion culture
- the effect of improving CHO cell proliferation and antibody production by adding phosphate to the flow medium was evaluated in Perfusion culture. ..
- CHO cells cells of the CHO S lineage modified to produce the antibody Adalimumab were used.
- the medium exchange was carried out by centrifuging the culture medium, removing it so that 3.68 mL of the centrifugal supernatant remained, and adding 6.32 mL of fresh fed-batch medium. After changing the medium, the culture was restarted.
- the number of living cells was measured using a living-and-dead cell autoanalyzer Vi-CELL TM XR (Beckman Coulter), and the amount of antibody produced was measured using CEDEX Bio HT (Roche Diagnostics).
- animal cells for example, proliferation of animal cells and production of target substance by animal cells.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Biomedical Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Cell Biology (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Biophysics (AREA)
- Immunology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Abstract
Description
[1]
目的物質の製造方法であって、
目的物質生産能を有する動物細胞を培地で培養すること;および
目的物質を回収すること
を含み、
前記培養が、下記(A)および/または(B)の条件で実施される、方法:
(A)前記培地中のリン酸濃度を4mM以上に増強した条件;
(B)前記培地中のカリウム濃度を1mM以上に増強した条件。
[2]
前記目的物質が、タンパク質である、前記方法。
[3]
動物細胞の培養方法であって、
動物細胞を培地で培養すること
を含み、
前記培養が、下記(A)および/または(B)の条件で実施される、方法:
(A)前記培地中のリン酸濃度を4mM以上に増強した条件;
(B)前記培地中のカリウム濃度を1mM以上に増強した条件。
[4]
前記培養が、少なくとも、前記培地中のリン酸濃度を増強した条件で実施される、前記方法。
[5]
前記培養の際の前記培地中のリン酸濃度が、11mM以上である、前記方法。
[6]
リン酸および/またはカリウムが、培養開始時に前記濃度で前記培地に含有される、前記方法。
[7]
リン酸および/またはカリウムが、培養開始後に前記濃度で前記培地に含有されるように該培地に供給される、前記方法。
[8]
リン酸および/またはカリウムが、培養の全期間を通じての平均値として、前記濃度で前記培地に含有される、前記方法。
[9]
リン酸が、培養の全期間を通じて、一日当たり、0.05mM以上の供給量で前記培地に供給される、前記方法。
[10]
リン酸が、培養の全期間を通じて、一日当たり、0.5mM以上の供給量で前記培地に供給される、前記方法。
[11]
カリウムが、培養の全期間を通じて、一日当たり、0.2mM以上の供給量で前記培地に供給される、前記方法。
[12]
前記培養が、リン酸濃度が4mM以上の流加培地を用いた灌流培養により実施される、前記方法。
[13]
前記培養が、リン酸濃度が11mM以上の流加培地を用いた灌流培養により実施される、前記方法。
[14]
前記培養が、アミンの存在下で実施される、前記方法。
[15]
前記アミンが、1,4-ブタンジアミンである、前記方法。
[16]
前記培養が、コリンおよび/またはセリンの存在下で実施される、前記方法。
[17]
前記培養の際の前記培地中のアミン濃度が、0.002mM以上である、前記方法。
[18]
前記培養の際の前記培地中のコリン濃度が、0.2mM以上である、前記方法。
[19]
前記培養の際の前記培地中のセリン濃度が、2mM以上である、前記方法。
[20]
前記アミン、コリン、および/またはセリンが、培養開始時に前記濃度で前記培地に含有される、前記方法。
[21]
前記アミン、コリン、および/またはセリンが、培養開始後に前記濃度で前記培地に含有されるように該培地に供給される、前記方法。
[22]
前記アミン、コリン、および/またはセリンが、培養の全期間を通じての平均値として、前記濃度で前記培地に含有される、前記方法。
[23]
前記アミンが、培養の全期間を通じて、一日当たり、0.0005mM以上の供給量で前記培地に供給される、前記方法。
[24]
コリンが、培養の全期間を通じて、一日当たり、0.05mM以上の供給量で前記培地に供給される、前記方法。
[25]
セリンが、培養の全期間を通じて、一日当たり、0.5mM以上の供給量で前記培地に供給される、前記方法。
[26]
リン酸濃度および/またはカリウム濃度が増強された、動物細胞培養用の培地。
[27]
少なくとも、リン酸濃度が増強された、前記培地。
[28]
さらに、アミンを含有する、前記培地。
[29]
前記アミンが、1,4-ブタンジアミンである、前記培地。
[30]
さらに、コリンおよび/またはセリンを含有する、前記培地。
[31]
初発培地である、前記培地。
[32]
流加培地である、前記培地。
本発明の方法は、動物細胞の培養方法であって、動物細胞を培地で培養することを含み、前記培養が前記培地中のリン酸濃度および/またはカリウム濃度を増強した条件で実施される方法である。
本発明の培地は、リン酸濃度および/またはカリウム濃度が増強された、動物細胞培養用の培地である。本発明の培地は、例えば、本発明の方法に用いることができる。
本実施例では、流加培地へのリン酸添加によるCHO細胞の増殖及び抗体生産の向上効果を評価した。CHO細胞としては、抗体Adalimumabを生産するよう改変されたCHO DG-44系統の細胞を用いた。
CELLiST(登録商標) Basal media(味の素株式会社:BASAL4P;終濃度1.9 mMのリン酸を含有する)にLR3-IGF-1(終濃度10 μg/mL)、デキストラン硫酸ナトリウム(終濃度400 mg/L)、およびL-Glutamine(終濃度 6 mM)を添加し、基礎培地を調製した。
CELLiST(登録商標) Feed media(味の素株式会社:FEED2)にグルコース(終濃度75 g/L)とリン酸二水素ナトリウム(終濃度0、125、250、500、1000、4000、または7000 mg/L(すなわち、0.0、1.0、2.1、4.2、8.3、33.3、または58.3 mM))を添加し、リン酸濃度の異なる7種類の流加培地を調製した。
調製した基礎培地を用いて3×105 cells/mLに調整したCHO細胞懸濁液30 mLを125 mLフラスコ(CORNING:431143)へ播種し、14日間培養を行った。培養は、全ての実験群についてn=2で実施した。培養温度は37℃、撹拌速度は110 rpmとした。調製した流加培地を、培養開始4、7、9、11日目にそれぞれ2.1 mL添加した。培養開始4、7、9、11、および14日目に細胞培養液をサンプリングし、生細胞数を生死細胞オートアナライザーVi-CELLTM XR(ベックマン・コールター社)を用いて測定し、抗体産生量をOctet QK(FORTEBIO社)を用いて測定した。
生細胞数の推移および培養14日目の抗体産生量を図1および2に示す。流加培地中のリン酸濃度の増大に伴い、生細胞数と抗体産生量が向上することが明らかとなった。
本実施例では、流加培地へのリン酸添加によるCHO細胞の増殖及び抗体生産の向上効果を評価した。CHO細胞としては、抗体Adalimumabを生産するよう改変されたCHO DG-44系統の細胞を用いた。
CELLiST(登録商標) Basal media(味の素株式会社:BASAL4P;終濃度1.9 mMのリン酸を含有する)にLR3-IGF-1(終濃度10 μg/mL)、リン酸二水素ナトリウム(115 mg/L(すなわち、終濃度2.9 mM))、デキストラン硫酸ナトリウム(終濃度400 mg/L)とL-Glutamine(終濃度 6 mM)を添加し、基礎培地を調製した。
CELLiST(登録商標) Feed media(味の素株式会社:FEED2)にグルコース(終濃度75 g/L)とリン酸二水素ナトリウム(終濃度0、1000、2000、3000、4000、または5000 mg/L(すなわち、0.0、8.3、16.7、25、33.3、または41.7 mM))を添加し、リン酸濃度の異なる6種類の流加培地を調製した。
調製した基礎培地を用いて3×105 cells/mLに調整したCHO細胞懸濁液30 mLを125 mLフラスコ(CORNING:431143)へ播種し、14日間培養を行った。培養は、全ての実験群についてn=2で実施した。培養温度は37℃、撹拌速度は110 rpmとした。調製した流加培地を、培養開始4、7、9、11日目にそれぞれ2.1 mL添加した。培養開始4、7、9、11、および14日目に細胞培養液をサンプリングし、生細胞数を生死細胞オートアナライザーVi-CELLTM XR(ベックマン・コールター社)を用いて測定し、抗体産生量をCEDEX Bio HT(ロシュ・ダイアグノスティックス社)を用いて測定した。
培養液中のリン酸濃度の推移および培養14日目の抗体産生量を図3および4に示す。終濃度2000 mg/Lリン酸二水素ナトリウムを含有する流加培地を添加することにより、先行特許(US6,924,124)に記載のリン酸濃度(培養液中のリン酸濃度1.5~3.5 mM)を再現した。先行特許の条件より高濃度のリン酸を含有する流加培地を添加することで、先行特許の条件と比較して抗体の生産性が向上することが明らかとなった。
本実施例では、流加培地へのリン酸ならびにセリン、コリン、またはアミンの添加によるCHO細胞の増殖及び抗体生産の向上効果を評価した。CHO細胞としては、抗体Adalimumabを生産するよう改変されたCHO S系統の細胞を用いた。
CELLiST(登録商標) Basal media(味の素株式会社:BASAL4P;終濃度1.9 mMのリン酸を含有する)にLR3-IGF-1(終濃度10 μg/mL)、リン酸二水素ナトリウム(115 mg/L(すなわち、Basal mediaに含有されるリン酸との合計で終濃度2.9 mM))、デキストラン硫酸ナトリウム(終濃度400 mg/L)、およびL-Glutamine(終濃度 6 mM)を添加し、基礎培地を調製した。
CELLiST(登録商標) Feed media(味の素株式会社:FEED2)にグルコース(終濃度75 g/L)、リン酸二水素ナトリウム(終濃度0または2500 mg/L(すなわち、0.0または20.8 mM))、および必要により以下の成分(A)、(B)、(C)、または(D)を添加し、10種類の流加培地を調製した:
(A)1,4-ブタンジアミン(終濃度12.8 mg/L(すなわち、0.145 mM));
(B)セリン(終濃度20,000 mg/L(すなわち、190 mM))、重酒石酸コリン(終濃度4,400 mg/L(すなわち、17.4 mM))、および1,4-ブタンジアミン(終濃度12.8 mg/L(すなわち、0.145 mM));
(C)1.5倍濃度の成分(B)(すなわち、セリン:30,000 mg/L , 重酒石酸コリン:6,600 mg/L, および1,4-ブタンジアミン:19.2 mg/L);
(D)2倍濃度の成分(B)(すなわち、セリン:40,000 mg/L , 重酒石酸コリン:8,800 mg/L, および1,4-ブタンジアミン:25.6 mg/L)。
調製した基礎培地を用いて3×105 cells/mLに調整したCHO細胞懸濁液を4 mLを6wellフラスコ(CORNING:3471)へ播種し、14日間培養を行った。培養は、全ての実験群についてn=2で実施した。培養温度は37℃、撹拌速度は110 rpmとした。調製した流加培地を、培養開始4、7、9、11日目にそれぞれ280 μL添加した。培養開始4、7、9、11、14日目に細胞培養液をサンプリングし、生細胞数をフローサイトメーターguava easyCyte(Luminex社)を用いて測定し、抗体産生量をCEDEX Bio HT(ロシュ・ダイアグノスティックス社)を用いて測定した。
培養14日目の抗体産生量を図5に示す。流加培地中へリン酸二水素ナトリウムを添加しない条件では、セリン、重酒石酸コリン、または1,4-ブタンジアミンの添加効果は認められなかった。一方、流加培地へリン酸二水素ナトリウムを添加した条件では、1,4-ブタンジアミンを添加することで抗体の生産性が向上し、セリン、重酒石酸コリン、および1,4-ブタンジアミンを添加することで抗体の生産性がさらに向上することが明らかとなった。特に、セリン、重酒石酸コリン、および1,4-ブタンジアミンを1.5倍濃度で添加した場合に最大の抗体濃度を示した。
本実施例では、流加培地へのリン酸またはカリウムの添加によるCHO細胞の増殖及び抗体生産の向上効果を評価した。CHO細胞としては、抗体Herceptinを生産するよう改変されたCHO S系統の細胞を用いた。
CELLiST(登録商標) Basal media(味の素株式会社:BASAL3;終濃度1.9 mMのリン酸を含有する)にLR3-IGF-1(終濃度10 μg/mL)とL-Glutamine(終濃度 6 mM)を添加し、基礎培地を調製した。
CELLiST(登録商標) Feed media(味の素:FEED2)にリン酸水素二ナトリウム(終濃度0または4,510 mg/L(すなわち、0.0または20.8 mM))と塩化カリウム(終濃度0または2,850 mg/L(すなわち、0.0または38.2 mM))を添加し、4種類の流加培地を調製した。
調製した基礎培地を用いて3×105 cells/mLに調整したCHO細胞懸濁液30 mLを125 mLフラスコ(CORNING:431143)へ播種し、14日間培養を行った。培養は、全ての実験群についてn=2で実施した。培養温度は37℃、撹拌速度は110 rpmとした。調製した流加培地を、培養開始4、6、8、10、12日目にそれぞれ1.2 mL添加した。培養開始4、7、9、11、および14日目に細胞培養液をサンプリングし、生細胞数を生死細胞オートアナライザーVi-CELLTM XR(ベックマン・コールター社)を用いて測定し、抗体産生量をCEDEX Bio HT(ロシュ・ダイアグノスティックス社)を用いて測定した。また、培養開始4、7、9、11日目に細胞培養液をサンプリングし、グルコース濃度をCEDEX Bio HT(ロシュ・ダイアグノスティックス社)を用いて測定し、グルコースの終濃度が11 g/Lとなるように500 g/Lグルコース水溶液を添加した。
培養14日目の抗体産生量を図6に示す。流加培地中へリン酸および/またはカリウムを添加した群では、無添加群と比較して抗体生産量は高値を示した。また、リン酸およびカリウムを添加した群では、リン酸単独添加群およびカリウム単独添加群と比較して抗体産生量は高値を示した。
本実施例では、基礎培地および流加培地へのリン酸添加によるCHO細胞の増殖及び抗体生産の向上効果をPerfusion培養にて評価した。CHO細胞としては、抗体Adalimumabを生産するよう改変されたCHO S系統の細胞を用いた。
CELLiST(登録商標) Basal media(味の素株式会社:BASAL4P;終濃度1.9 mMのリン酸を含有する)にLR3-IGF-1(終濃度10 μg/mL)、リン酸二水素ナトリウム(添加量0, 1450, または1690 mg/L(すなわち、Basal mediaに含有されるリン酸との合計で終濃度1.9, 14, または16 mM))、およびL-Glutamine(終濃度 6 mM)を添加し、基礎培地および流加培地を調製した。
調製した基礎培地を用いて25×105 cells/mLに調整した10 mLのCHO細胞懸濁液をマイクロバイオリアクターambr15(sartorius:001-0881)を用いて8日間培養を行った。培養中は、pH 7.2±0.1、DOを飽和濃度に対して50%、培養温度37℃、攪拌速度=1000 rpmとなるように管理した。また、細胞数が200×105cells/mLに到達した後、毎日細胞数を測定し、培地交換後の培養液中の細胞濃度が200×105 cells/mLとなるように過剰な細胞培養液を除いた後、培地交換を実施した。培地交換は、培養液を遠心分離し、遠心上清が3.68 mL残るように取り除き、基礎培地と同一の新鮮な流加培地を6.32 mL添加し、撹拌することで実施した。培地交換後に、培養を再開した。生細胞数を生死細胞オートアナライザーVi-CELLTM XR(ベックマン・コールター社)を用いて測定し、抗体産生量をCEDEX Bio HT(ロシュ・ダイアグノスティックス社)を用いて測定した。
培養8日目の抗体産生量を図7に示す。基礎培地および流加培地のリン酸濃度を14 mM以上に増強した場合には、基礎培地および流加培地のリン酸濃度を増強しない場合と比較して、抗体濃度が増加することが明らかとなった。
本実施例では、流加培地へのリン酸添加によるCHO細胞の増殖及び抗体生産の向上効果をPerfusion培養にて評価した。CHO細胞としては、抗体Adalimumabを生産するよう改変されたCHO S系統の細胞を用いた。
CELLiST(登録商標) Basal media(味の素株式会社:BASAL4P;終濃度1.9 mMのリン酸を含有する)にLR3-IGF-1(終濃度10 μg/mL)とL-Glutamine(終濃度 6 mM)を添加し、基礎培地を調製した。
(2)流加培地の調製
CELLiST(登録商標) Basal media(味の素株式会社:BASAL4P;終濃度1.9 mMのリン酸を含有する)にLR3-IGF-1(終濃度10 μg/mL)、リン酸二水素ナトリウム(添加量0, 490, または730 mg/L(すなわち、Basal mediaに含有されるリン酸との合計で終濃度1.9, 6, または8 mM))、およびL-Glutamine(終濃度6 mM)を添加し、流加培地を調製した。
調製した基礎培地を用いて25×105 cells/mLに調整した10 mLのCHO細胞懸濁液をマイクロバイオリアクターambr15(sartorius:001-0881)を用いて12日間培養を行った。培養中は、pH 7.2±0.1、DOを飽和濃度に対して50%、培養温度37℃、攪拌速度=1000 rpmとなるように管理した。また、細胞数が150×105 cells/mLに到達した後、毎日細胞数を測定し、培地交換後の培養液中の細胞濃度が150×105 cells/mLとなるように、過剰な細胞培養液を除いた後、培地交換を実施した。培地交換は、培養液を遠心分離し、遠心上清が3.68 mL残るように取り除き、新鮮な流加培地を6.32 mL添加することで実施した。培地交換後に、培養を再開した。生細胞数を生死細胞オートアナライザーVi-CELLTM XR(ベックマン・コールター社)を用いて測定し、抗体産生量をCEDEX Bio HT(ロシュ・ダイアグノスティックス社)を用いて測定した。
培養8日目の抗体産生量を図8に示す。流加培地のリン酸濃度を6 mM以上に増強した場合には、流加培地のリン酸濃度を増強しない場合と比較して、抗体濃度が増加することが明らかとなった。
Claims (32)
- 目的物質の製造方法であって、
目的物質生産能を有する動物細胞を培地で培養すること;および
目的物質を回収すること
を含み、
前記培養が、下記(A)および/または(B)の条件で実施される、方法:
(A)前記培地中のリン酸濃度を4mM以上に増強した条件;
(B)前記培地中のカリウム濃度を1mM以上に増強した条件。 - 前記目的物質が、タンパク質である、請求項1に記載の方法。
- 動物細胞の培養方法であって、
動物細胞を培地で培養すること
を含み、
前記培養が、下記(A)および/または(B)の条件で実施される、方法:
(A)前記培地中のリン酸濃度を4mM以上に増強した条件;
(B)前記培地中のカリウム濃度を1mM以上に増強した条件。 - 前記培養が、少なくとも、前記培地中のリン酸濃度を増強した条件で実施される、請求項1~3のいずれか1項に記載の方法。
- 前記培養の際の前記培地中のリン酸濃度が、11mM以上である、請求項1~4のいずれか1項に記載の方法。
- リン酸および/またはカリウムが、培養開始時に前記濃度で前記培地に含有される、請求項1~5のいずれか1項に記載の方法。
- リン酸および/またはカリウムが、培養開始後に前記濃度で前記培地に含有されるように該培地に供給される、請求項1~6のいずれか1項に記載の方法。
- リン酸および/またはカリウムが、培養の全期間を通じての平均値として、前記濃度で前記培地に含有される、請求項1~7のいずれか1項に記載の方法。
- リン酸が、培養の全期間を通じて、一日当たり、0.05mM以上の供給量で前記培地に供給される、請求項1~8のいずれか1項に記載の方法。
- リン酸が、培養の全期間を通じて、一日当たり、0.5mM以上の供給量で前記培地に供給される、請求項1~9のいずれか1項に記載の方法。
- カリウムが、培養の全期間を通じて、一日当たり、0.2mM以上の供給量で前記培地に供給される、請求項1~10のいずれか1項に記載の方法。
- 前記培養が、リン酸濃度が4mM以上の流加培地を用いた灌流培養により実施される、請求項1~11のいずれか1項に記載の方法。
- 前記培養が、リン酸濃度が11mM以上の流加培地を用いた灌流培養により実施される、請求項1~12のいずれか1項に記載の方法。
- 前記培養が、アミンの存在下で実施される、請求項1~13のいずれか1項に記載の方法。
- 前記アミンが、1,4-ブタンジアミンである、請求項14に記載の方法。
- 前記培養が、コリンおよび/またはセリンの存在下で実施される、請求項1~15のいずれか1項に記載の方法。
- 前記培養の際の前記培地中のアミン濃度が、0.002mM以上である、請求項14~16のいずれか1項に記載の方法。
- 前記培養の際の前記培地中のコリン濃度が、0.2mM以上である、請求項16または17に記載の方法。
- 前記培養の際の前記培地中のセリン濃度が、2mM以上である、請求項16~18のいずれか1項に記載の方法。
- 前記アミン、コリン、および/またはセリンが、培養開始時に前記濃度で前記培地に含有される、請求項14~19に記載の方法。
- 前記アミン、コリン、および/またはセリンが、培養開始後に前記濃度で前記培地に含有されるように該培地に供給される、請求項14~20のいずれか1項に記載の方法。
- 前記アミン、コリン、および/またはセリンが、培養の全期間を通じての平均値として、前記濃度で前記培地に含有される、請求項14~21のいずれか1項に記載の方法。
- 前記アミンが、培養の全期間を通じて、一日当たり、0.0005mM以上の供給量で前記培地に供給される、請求項14~22のいずれか1項に記載の方法。
- コリンが、培養の全期間を通じて、一日当たり、0.05mM以上の供給量で前記培地に供給される、請求項16~23のいずれか1項に記載の方法。
- セリンが、培養の全期間を通じて、一日当たり、0.5mM以上の供給量で前記培地に供給される、請求項16~24のいずれか1項に記載の方法。
- リン酸濃度および/またはカリウム濃度が増強された、動物細胞培養用の培地。
- 少なくとも、リン酸濃度が増強された、請求項26に記載の培地。
- さらに、アミンを含有する、請求項26または27に記載の培地。
- 前記アミンが、1,4-ブタンジアミンである、請求項28に記載の培地。
- さらに、コリンおよび/またはセリンを含有する、請求項26~29のいずれか1項に記載の培地。
- 初発培地である、請求項26~30のいずれか1項に記載の培地。
- 流加培地である、請求項26~30のいずれか1項に記載の培地。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020227017195A KR20220097421A (ko) | 2019-11-05 | 2020-11-05 | 단백질의 제조 방법 |
| CN202080076873.4A CN114667351A (zh) | 2019-11-05 | 2020-11-05 | 蛋白质的制造方法 |
| EP20885757.3A EP4056678A4 (en) | 2019-11-05 | 2020-11-05 | METHOD FOR PRODUCING A PROTEIN |
| JP2021555106A JPWO2021090888A1 (ja) | 2019-11-05 | 2020-11-05 | |
| US17/730,750 US20220251622A1 (en) | 2019-11-05 | 2022-04-27 | Method for Producing Protein |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019200753 | 2019-11-05 | ||
| JP2019-200753 | 2019-11-05 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/730,750 Continuation US20220251622A1 (en) | 2019-11-05 | 2022-04-27 | Method for Producing Protein |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021090888A1 true WO2021090888A1 (ja) | 2021-05-14 |
Family
ID=75848562
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2020/041377 WO2021090888A1 (ja) | 2019-11-05 | 2020-11-05 | タンパク質の製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20220251622A1 (ja) |
| EP (1) | EP4056678A4 (ja) |
| JP (1) | JPWO2021090888A1 (ja) |
| KR (1) | KR20220097421A (ja) |
| CN (1) | CN114667351A (ja) |
| WO (1) | WO2021090888A1 (ja) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01222793A (ja) * | 1988-02-29 | 1989-09-06 | Morinaga & Co Ltd | モノクローナル抗体生産増強法 |
| JPH01247097A (ja) * | 1988-03-30 | 1989-10-02 | Morinaga & Co Ltd | モノクローナル抗体生産増強法 |
| US6924124B1 (en) | 2002-08-23 | 2005-08-02 | Immunex Corporation | Feeding strategies for cell culture |
| WO2007077217A2 (en) | 2006-01-04 | 2007-07-12 | Baxter International Inc. | Oligopeptide-free cell culture media |
| WO2008136398A1 (ja) | 2007-04-26 | 2008-11-13 | Chugai Seiyaku Kabushiki Kaisha | 高濃度アミノ酸含有培地を用いた細胞の培養方法 |
| WO2011134920A1 (en) * | 2010-04-26 | 2011-11-03 | Novartis Ag | Improved cell culture medium |
| WO2011134921A1 (en) | 2010-04-26 | 2011-11-03 | Novartis Ag | Improved cell culture medium |
| WO2017090665A1 (ja) | 2015-11-25 | 2017-06-01 | 味の素株式会社 | フィブロイン様タンパク質の製造法 |
| WO2017171001A1 (ja) | 2016-03-31 | 2017-10-05 | 味の素株式会社 | フィブロイン様タンパク質改変体および細胞培養方法 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SG10201510384UA (en) * | 2006-09-13 | 2016-01-28 | Abbvie Inc | Cell culture improvements |
| CN102994441A (zh) * | 2012-09-19 | 2013-03-27 | 上海瀚康生物医药科技有限公司 | 一种细胞培养基及其制备方法和用途 |
-
2020
- 2020-11-05 WO PCT/JP2020/041377 patent/WO2021090888A1/ja unknown
- 2020-11-05 KR KR1020227017195A patent/KR20220097421A/ko unknown
- 2020-11-05 CN CN202080076873.4A patent/CN114667351A/zh active Pending
- 2020-11-05 JP JP2021555106A patent/JPWO2021090888A1/ja active Pending
- 2020-11-05 EP EP20885757.3A patent/EP4056678A4/en active Pending
-
2022
- 2022-04-27 US US17/730,750 patent/US20220251622A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01222793A (ja) * | 1988-02-29 | 1989-09-06 | Morinaga & Co Ltd | モノクローナル抗体生産増強法 |
| JPH01247097A (ja) * | 1988-03-30 | 1989-10-02 | Morinaga & Co Ltd | モノクローナル抗体生産増強法 |
| US6924124B1 (en) | 2002-08-23 | 2005-08-02 | Immunex Corporation | Feeding strategies for cell culture |
| WO2007077217A2 (en) | 2006-01-04 | 2007-07-12 | Baxter International Inc. | Oligopeptide-free cell culture media |
| WO2008136398A1 (ja) | 2007-04-26 | 2008-11-13 | Chugai Seiyaku Kabushiki Kaisha | 高濃度アミノ酸含有培地を用いた細胞の培養方法 |
| WO2011134920A1 (en) * | 2010-04-26 | 2011-11-03 | Novartis Ag | Improved cell culture medium |
| WO2011134921A1 (en) | 2010-04-26 | 2011-11-03 | Novartis Ag | Improved cell culture medium |
| WO2017090665A1 (ja) | 2015-11-25 | 2017-06-01 | 味の素株式会社 | フィブロイン様タンパク質の製造法 |
| WO2017171001A1 (ja) | 2016-03-31 | 2017-10-05 | 味の素株式会社 | フィブロイン様タンパク質改変体および細胞培養方法 |
Non-Patent Citations (7)
| Title |
|---|
| GENE, vol. 60, no. 1, 1987, pages 115 - 127 |
| GOLDSTEIN ET AL.: "Prokaryotic Promoters in Biotechnology", BIOTECHNOL. ANNU. REV., vol. 1, 1995, pages 105 - 128, XP008038474 |
| HIGUCHI, R.: "PCR Technology", 1989, STOCKTON PRESS |
| KRAMER, W.FRITS, H.J., METH. IN ENZYMOL., vol. 154, 1987, pages 367 |
| S. SATO, H. MURAKAMI, T. SUGAHARA, T. IKEGAMI, K. YAMADA, H. OMURA, S. HASHIZUME : "Stimulation of monoclonal antibody production by human-human hybridoma cells with an elevated concentration of potassium or sodium phosphate in serum-free medium", CYTOTECHNOLOGY, vol. 2, no. 1, 1 February 1989 (1989-02-01), Dordrecht, pages 63 - 67, XP009536658, ISSN: 0920-9069, DOI: 10.1007/BF00365416 * |
| See also references of EP4056678A4 |
| SHUICHI HASHIZUME, KAZUHIKO KURODA, MASANORI KAMEI: "Generation and production of human monoclonal antibody", NIPPON NOGEIKAGAKU KAISHI, vol. 62, no. 10, 15 October 1988 (1988-10-15), pages 1513 - 1516, XP055929703, ISSN: 0002-1407, DOI: 10.1271/nogeikagaku1924.62.1513 * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4056678A1 (en) | 2022-09-14 |
| US20220251622A1 (en) | 2022-08-11 |
| EP4056678A4 (en) | 2023-12-06 |
| JPWO2021090888A1 (ja) | 2021-05-14 |
| KR20220097421A (ko) | 2022-07-07 |
| CN114667351A (zh) | 2022-06-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101540124B1 (ko) | 항노화 화합물을 사용하는 단백질의 생산 방법 | |
| US11661579B2 (en) | Cell culture method using amino acid-enriched medium | |
| CN107922920B (zh) | 补充牛磺酸的细胞培养基和使用方法 | |
| KR102229380B1 (ko) | 무혈청 세포 배양 배지 | |
| JP2022546230A (ja) | 細胞培養方法 | |
| JP2013535981A (ja) | 成長因子不含適合細胞の細胞培養 | |
| WO2021090888A1 (ja) | タンパク質の製造方法 | |
| JP6108170B2 (ja) | 細胞培養用培地 | |
| CA2852021C (en) | Addition of iron to improve cell culture | |
| WO2023120637A1 (ja) | 動物細胞培養用の組成物 | |
| WO2021187411A1 (ja) | 動物細胞の培養方法 | |
| WO2023125389A1 (en) | Novel markers for recombinant production system | |
| Lobanova et al. | Biologicals Technology Optimization of Culturing of CHO Cells Expressing Recombinant Interferon-β |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20885757 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2021555106 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20227017195 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2020885757 Country of ref document: EP Effective date: 20220607 |