WO2020116628A1 - 糖および甘味料の呈する味質が改善した茶飲料 - Google Patents
糖および甘味料の呈する味質が改善した茶飲料 Download PDFInfo
- Publication number
- WO2020116628A1 WO2020116628A1 PCT/JP2019/047868 JP2019047868W WO2020116628A1 WO 2020116628 A1 WO2020116628 A1 WO 2020116628A1 JP 2019047868 W JP2019047868 W JP 2019047868W WO 2020116628 A1 WO2020116628 A1 WO 2020116628A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- calcium
- potassium
- sodium
- tea beverage
- rebaudioside
- Prior art date
Links
- 235000013616 tea Nutrition 0.000 title claims abstract description 310
- 235000000346 sugar Nutrition 0.000 title claims abstract description 124
- 235000003599 food sweetener Nutrition 0.000 title claims abstract description 55
- 239000003765 sweetening agent Substances 0.000 title claims abstract description 55
- 235000019640 taste Nutrition 0.000 title description 84
- 150000008163 sugars Chemical class 0.000 title description 14
- 239000011575 calcium Substances 0.000 claims abstract description 150
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 claims abstract description 148
- 239000011591 potassium Substances 0.000 claims abstract description 147
- 229910052700 potassium Inorganic materials 0.000 claims abstract description 147
- 239000011734 sodium Substances 0.000 claims abstract description 147
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 claims abstract description 146
- 229910052791 calcium Inorganic materials 0.000 claims abstract description 145
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 claims abstract description 142
- 229910052708 sodium Inorganic materials 0.000 claims abstract description 142
- RPYRMTHVSUWHSV-CUZJHZIBSA-N rebaudioside D Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O RPYRMTHVSUWHSV-CUZJHZIBSA-N 0.000 claims abstract description 120
- GSGVXNMGMKBGQU-PHESRWQRSA-N rebaudioside M Chemical compound C[C@@]12CCC[C@](C)([C@H]1CC[C@@]13CC(=C)[C@@](C1)(CC[C@@H]23)O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)[C@H]1O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O)C(=O)O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)[C@H]1O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O GSGVXNMGMKBGQU-PHESRWQRSA-N 0.000 claims abstract description 53
- 239000000284 extract Substances 0.000 claims abstract description 52
- GHBNZZJYBXQAHG-KUVSNLSMSA-N (2r,3r,4s,5s,6r)-2-[[(2r,3s,4s,5r,6r)-6-[[(3s,8s,9r,10r,11r,13r,14s,17r)-17-[(2r,5r)-5-[(2s,3r,4s,5s,6r)-4,5-dihydroxy-3-[(2r,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-6-[[(2r,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy Chemical compound C([C@H]1O[C@H]([C@@H]([C@@H](O)[C@@H]1O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@H](CC[C@@H](C)[C@@H]1[C@]2(C[C@@H](O)[C@@]3(C)[C@H]4C(C([C@@H](O[C@H]5[C@@H]([C@@H](O)[C@H](O)[C@@H](CO[C@H]6[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O6)O)O5)O)CC4)(C)C)=CC[C@H]3[C@]2(C)CC1)C)C(C)(C)O)O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O GHBNZZJYBXQAHG-KUVSNLSMSA-N 0.000 claims abstract description 42
- TVJXHJAWHUMLLG-UHFFFAOYSA-N mogroside V Natural products CC(CCC(OC1OC(COC2OC(CO)C(O)C(O)C2OC3OC(CO)C(O)C(O)C3O)C(O)C(O)C1O)C(C)(C)O)C4CCC5(C)C6CC=C7C(CCC(OC8OC(COC9OC(CO)C(O)C(O)C9O)C(O)C(O)C8O)C7(C)C)C6(C)C(O)CC45C TVJXHJAWHUMLLG-UHFFFAOYSA-N 0.000 claims abstract description 42
- 229930188195 rebaudioside Natural products 0.000 claims abstract description 25
- RLLCWNUIHGPAJY-RYBZXKSASA-N Rebaudioside E Natural products O=C(O[C@H]1[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O2)[C@@H](O)[C@@H](O)[C@H](CO)O1)[C@]1(C)[C@@H]2[C@@](C)([C@@H]3[C@@]4(CC(=C)[C@@](O[C@@H]5[C@@H](O[C@@H]6[C@@H](O)[C@H](O)[C@@H](O)[C@H](CO)O6)[C@H](O)[C@@H](O)[C@H](CO)O5)(C4)CC3)CC2)CCC1 RLLCWNUIHGPAJY-RYBZXKSASA-N 0.000 claims abstract description 13
- RLLCWNUIHGPAJY-SFUUMPFESA-N rebaudioside E Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O RLLCWNUIHGPAJY-SFUUMPFESA-N 0.000 claims abstract description 13
- 239000000892 thaumatin Substances 0.000 claims abstract description 11
- 235000010436 thaumatin Nutrition 0.000 claims abstract description 11
- 229960003975 potassium Drugs 0.000 claims description 147
- 229960005069 calcium Drugs 0.000 claims description 144
- 238000000034 method Methods 0.000 claims description 90
- 244000269722 Thea sinensis Species 0.000 claims description 89
- 239000008123 high-intensity sweetener Substances 0.000 claims description 71
- 235000013615 non-nutritive sweetener Nutrition 0.000 claims description 71
- 235000013361 beverage Nutrition 0.000 claims description 60
- 229930006000 Sucrose Natural products 0.000 claims description 51
- 239000005720 sucrose Substances 0.000 claims description 51
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 claims description 50
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 claims description 47
- 235000013399 edible fruits Nutrition 0.000 claims description 42
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 claims description 32
- 239000008103 glucose Substances 0.000 claims description 32
- 239000002994 raw material Substances 0.000 claims description 25
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 claims description 24
- 239000001110 calcium chloride Substances 0.000 claims description 24
- 229910001628 calcium chloride Inorganic materials 0.000 claims description 24
- 229960002713 calcium chloride Drugs 0.000 claims description 24
- 235000011148 calcium chloride Nutrition 0.000 claims description 24
- 239000001103 potassium chloride Substances 0.000 claims description 24
- 235000011164 potassium chloride Nutrition 0.000 claims description 24
- 239000000203 mixture Substances 0.000 claims description 23
- 229960002816 potassium chloride Drugs 0.000 claims description 23
- 239000012141 concentrate Substances 0.000 claims description 21
- 229920001542 oligosaccharide Polymers 0.000 claims description 20
- 150000002482 oligosaccharides Chemical class 0.000 claims description 20
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 claims description 19
- 229930091371 Fructose Natural products 0.000 claims description 18
- 239000005715 Fructose Substances 0.000 claims description 18
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 18
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 18
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 claims description 18
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 claims description 17
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 claims description 17
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 claims description 17
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 16
- 239000008101 lactose Substances 0.000 claims description 16
- 238000004519 manufacturing process Methods 0.000 claims description 14
- 150000008442 polyphenolic compounds Chemical class 0.000 claims description 14
- 235000013824 polyphenols Nutrition 0.000 claims description 14
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 claims description 12
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 claims description 12
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical group [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 12
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 claims description 12
- OQZCJRJRGMMSGK-UHFFFAOYSA-M potassium metaphosphate Chemical compound [K+].[O-]P(=O)=O OQZCJRJRGMMSGK-UHFFFAOYSA-M 0.000 claims description 12
- FGIUAXJPYTZDNR-UHFFFAOYSA-N potassium nitrate Chemical compound [K+].[O-][N+]([O-])=O FGIUAXJPYTZDNR-UHFFFAOYSA-N 0.000 claims description 12
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 claims description 11
- 235000010413 sodium alginate Nutrition 0.000 claims description 11
- 239000000661 sodium alginate Substances 0.000 claims description 11
- 229940005550 sodium alginate Drugs 0.000 claims description 11
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 claims description 10
- PFTAWBLQPZVEMU-DZGCQCFKSA-N (+)-catechin Chemical compound C1([C@H]2OC3=CC(O)=CC(O)=C3C[C@@H]2O)=CC=C(O)C(O)=C1 PFTAWBLQPZVEMU-DZGCQCFKSA-N 0.000 claims description 9
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 claims description 9
- 235000005487 catechin Nutrition 0.000 claims description 9
- ADRVNXBAWSRFAJ-UHFFFAOYSA-N catechin Natural products OC1Cc2cc(O)cc(O)c2OC1c3ccc(O)c(O)c3 ADRVNXBAWSRFAJ-UHFFFAOYSA-N 0.000 claims description 9
- 229950001002 cianidanol Drugs 0.000 claims description 9
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical group OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 claims description 8
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 8
- 230000002708 enhancing effect Effects 0.000 claims description 8
- SHWNNYZBHZIQQV-UHFFFAOYSA-J EDTA monocalcium diisodium salt Chemical compound [Na+].[Na+].[Ca+2].[O-]C(=O)CN(CC([O-])=O)CCN(CC([O-])=O)CC([O-])=O SHWNNYZBHZIQQV-UHFFFAOYSA-J 0.000 claims description 7
- 239000011780 sodium chloride Substances 0.000 claims description 7
- CHHHXKFHOYLYRE-UHFFFAOYSA-M 2,4-Hexadienoic acid, potassium salt (1:1), (2E,4E)- Chemical compound [K+].CC=CC=CC([O-])=O CHHHXKFHOYLYRE-UHFFFAOYSA-M 0.000 claims description 6
- KQMCGGGTJKNIMC-UHFFFAOYSA-N 2-hydroxy-3-propyl-2h-furan-5-one Chemical compound CCCC1=CC(=O)OC1O KQMCGGGTJKNIMC-UHFFFAOYSA-N 0.000 claims description 6
- CYDQOEWLBCCFJZ-UHFFFAOYSA-N 4-(4-fluorophenyl)oxane-4-carboxylic acid Chemical compound C=1C=C(F)C=CC=1C1(C(=O)O)CCOCC1 CYDQOEWLBCCFJZ-UHFFFAOYSA-N 0.000 claims description 6
- XWNSFEAWWGGSKJ-UHFFFAOYSA-N 4-acetyl-4-methylheptanedinitrile Chemical compound N#CCCC(C)(C(=O)C)CCC#N XWNSFEAWWGGSKJ-UHFFFAOYSA-N 0.000 claims description 6
- 239000001736 Calcium glycerylphosphate Substances 0.000 claims description 6
- 102000011632 Caseins Human genes 0.000 claims description 6
- 108010076119 Caseins Proteins 0.000 claims description 6
- LKDRXBCSQODPBY-JDJSBBGDSA-N D-allulose Chemical compound OCC1(O)OC[C@@H](O)[C@@H](O)[C@H]1O LKDRXBCSQODPBY-JDJSBBGDSA-N 0.000 claims description 6
- 239000004153 Potassium bromate Substances 0.000 claims description 6
- HLCFGWHYROZGBI-JJKGCWMISA-M Potassium gluconate Chemical compound [K+].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O HLCFGWHYROZGBI-JJKGCWMISA-M 0.000 claims description 6
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 claims description 6
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 claims description 6
- FAPWYRCQGJNNSJ-UBKPKTQASA-L calcium D-pantothenic acid Chemical compound [Ca+2].OCC(C)(C)[C@@H](O)C(=O)NCCC([O-])=O.OCC(C)(C)[C@@H](O)C(=O)NCCC([O-])=O FAPWYRCQGJNNSJ-UBKPKTQASA-L 0.000 claims description 6
- VSGNNIFQASZAOI-UHFFFAOYSA-L calcium acetate Chemical compound [Ca+2].CC([O-])=O.CC([O-])=O VSGNNIFQASZAOI-UHFFFAOYSA-L 0.000 claims description 6
- 235000011092 calcium acetate Nutrition 0.000 claims description 6
- 239000001639 calcium acetate Substances 0.000 claims description 6
- 229960005147 calcium acetate Drugs 0.000 claims description 6
- 235000010410 calcium alginate Nutrition 0.000 claims description 6
- 239000000648 calcium alginate Substances 0.000 claims description 6
- 229960002681 calcium alginate Drugs 0.000 claims description 6
- YYRMJZQKEFZXMX-UHFFFAOYSA-L calcium bis(dihydrogenphosphate) Chemical compound [Ca+2].OP(O)([O-])=O.OP(O)([O-])=O YYRMJZQKEFZXMX-UHFFFAOYSA-L 0.000 claims description 6
- 229910000019 calcium carbonate Inorganic materials 0.000 claims description 6
- 229960003563 calcium carbonate Drugs 0.000 claims description 6
- 235000010216 calcium carbonate Nutrition 0.000 claims description 6
- FNAQSUUGMSOBHW-UHFFFAOYSA-H calcium citrate Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O.[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O FNAQSUUGMSOBHW-UHFFFAOYSA-H 0.000 claims description 6
- 239000001354 calcium citrate Substances 0.000 claims description 6
- 229960004256 calcium citrate Drugs 0.000 claims description 6
- 229940062672 calcium dihydrogen phosphate Drugs 0.000 claims description 6
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 claims description 6
- 239000001201 calcium disodium ethylene diamine tetra-acetate Substances 0.000 claims description 6
- 235000011188 calcium disodium ethylene diamine tetraacetate Nutrition 0.000 claims description 6
- 239000000279 calcium ferrocyanide Substances 0.000 claims description 6
- 235000012251 calcium ferrocyanide Nutrition 0.000 claims description 6
- 239000004227 calcium gluconate Substances 0.000 claims description 6
- 229960004494 calcium gluconate Drugs 0.000 claims description 6
- 235000013927 calcium gluconate Nutrition 0.000 claims description 6
- 229940095618 calcium glycerophosphate Drugs 0.000 claims description 6
- UHHRFSOMMCWGSO-UHFFFAOYSA-L calcium glycerophosphate Chemical compound [Ca+2].OCC(CO)OP([O-])([O-])=O UHHRFSOMMCWGSO-UHFFFAOYSA-L 0.000 claims description 6
- 235000019299 calcium glycerylphosphate Nutrition 0.000 claims description 6
- FUFJGUQYACFECW-UHFFFAOYSA-L calcium hydrogenphosphate Chemical compound [Ca+2].OP([O-])([O-])=O FUFJGUQYACFECW-UHFFFAOYSA-L 0.000 claims description 6
- 239000000920 calcium hydroxide Substances 0.000 claims description 6
- 229910001861 calcium hydroxide Inorganic materials 0.000 claims description 6
- 229940095643 calcium hydroxide Drugs 0.000 claims description 6
- 235000011116 calcium hydroxide Nutrition 0.000 claims description 6
- BRPQOXSCLDDYGP-UHFFFAOYSA-N calcium oxide Chemical compound [O-2].[Ca+2] BRPQOXSCLDDYGP-UHFFFAOYSA-N 0.000 claims description 6
- 239000000292 calcium oxide Substances 0.000 claims description 6
- ODINCKMPIJJUCX-UHFFFAOYSA-N calcium oxide Inorganic materials [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 claims description 6
- 229940087373 calcium oxide Drugs 0.000 claims description 6
- 235000012255 calcium oxide Nutrition 0.000 claims description 6
- 229960002079 calcium pantothenate Drugs 0.000 claims description 6
- 239000001506 calcium phosphate Substances 0.000 claims description 6
- 229910000389 calcium phosphate Inorganic materials 0.000 claims description 6
- 239000000378 calcium silicate Substances 0.000 claims description 6
- 229910052918 calcium silicate Inorganic materials 0.000 claims description 6
- 229960003340 calcium silicate Drugs 0.000 claims description 6
- MCFVRESNTICQSJ-RJNTXXOISA-L calcium sorbate Chemical compound [Ca+2].C\C=C\C=C\C([O-])=O.C\C=C\C=C\C([O-])=O MCFVRESNTICQSJ-RJNTXXOISA-L 0.000 claims description 6
- 235000010244 calcium sorbate Nutrition 0.000 claims description 6
- 239000004303 calcium sorbate Substances 0.000 claims description 6
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 claims description 6
- 235000013539 calcium stearate Nutrition 0.000 claims description 6
- 239000008116 calcium stearate Substances 0.000 claims description 6
- 229940078456 calcium stearate Drugs 0.000 claims description 6
- OEUVSBXAMBLPES-UHFFFAOYSA-L calcium stearoyl-2-lactylate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC(=O)OC(C)C(=O)OC(C)C([O-])=O.CCCCCCCCCCCCCCCCCC(=O)OC(C)C(=O)OC(C)C([O-])=O OEUVSBXAMBLPES-UHFFFAOYSA-L 0.000 claims description 6
- 229940095672 calcium sulfate Drugs 0.000 claims description 6
- 235000011132 calcium sulphate Nutrition 0.000 claims description 6
- OKHHGHGGPDJQHR-YMOPUZKJSA-L calcium;(2s,3s,4s,5s,6r)-6-[(2r,3s,4r,5s,6r)-2-carboxy-6-[(2r,3s,4r,5s,6r)-2-carboxylato-4,5,6-trihydroxyoxan-3-yl]oxy-4,5-dihydroxyoxan-3-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylate Chemical compound [Ca+2].O[C@@H]1[C@H](O)[C@H](O)O[C@@H](C([O-])=O)[C@H]1O[C@H]1[C@@H](O)[C@@H](O)[C@H](O[C@H]2[C@H]([C@@H](O)[C@H](O)[C@H](O2)C([O-])=O)O)[C@H](C(O)=O)O1 OKHHGHGGPDJQHR-YMOPUZKJSA-L 0.000 claims description 6
- NEEHYRZPVYRGPP-UHFFFAOYSA-L calcium;2,3,4,5,6-pentahydroxyhexanoate Chemical compound [Ca+2].OCC(O)C(O)C(O)C(O)C([O-])=O.OCC(O)C(O)C(O)C(O)C([O-])=O NEEHYRZPVYRGPP-UHFFFAOYSA-L 0.000 claims description 6
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 claims description 6
- VEJCUEBBRSCJRP-UHFFFAOYSA-L calcium;hydron;phosphonato phosphate Chemical compound [Ca+2].OP(O)(=O)OP([O-])([O-])=O VEJCUEBBRSCJRP-UHFFFAOYSA-L 0.000 claims description 6
- 235000019700 dicalcium phosphate Nutrition 0.000 claims description 6
- ZPWVASYFFYYZEW-UHFFFAOYSA-L dipotassium hydrogen phosphate Chemical compound [K+].[K+].OP([O-])([O-])=O ZPWVASYFFYYZEW-UHFFFAOYSA-L 0.000 claims description 6
- WDRWZVWLVBXVOI-QTNFYWBSSA-L dipotassium;(2s)-2-aminopentanedioate Chemical compound [K+].[K+].[O-]C(=O)[C@@H](N)CCC([O-])=O WDRWZVWLVBXVOI-QTNFYWBSSA-L 0.000 claims description 6
- WPUMTJGUQUYPIV-JIZZDEOASA-L disodium (S)-malate Chemical compound [Na+].[Na+].[O-]C(=O)[C@@H](O)CC([O-])=O WPUMTJGUQUYPIV-JIZZDEOASA-L 0.000 claims description 6
- PXEDJBXQKAGXNJ-QTNFYWBSSA-L disodium L-glutamate Chemical compound [Na+].[Na+].[O-]C(=O)[C@@H](N)CCC([O-])=O PXEDJBXQKAGXNJ-QTNFYWBSSA-L 0.000 claims description 6
- BJHIKXHVCXFQLS-PQLUHFTBSA-N keto-D-tagatose Chemical compound OC[C@@H](O)[C@H](O)[C@H](O)C(=O)CO BJHIKXHVCXFQLS-PQLUHFTBSA-N 0.000 claims description 6
- 235000019691 monocalcium phosphate Nutrition 0.000 claims description 6
- 235000013923 monosodium glutamate Nutrition 0.000 claims description 6
- 235000010408 potassium alginate Nutrition 0.000 claims description 6
- 239000000737 potassium alginate Substances 0.000 claims description 6
- MZYRDLHIWXQJCQ-YZOKENDUSA-L potassium alginate Chemical group [K+].[K+].O1[C@@H](C([O-])=O)[C@@H](OC)[C@H](O)[C@H](O)[C@@H]1O[C@@H]1[C@@H](C([O-])=O)O[C@@H](O)[C@@H](O)[C@H]1O MZYRDLHIWXQJCQ-YZOKENDUSA-L 0.000 claims description 6
- GRLPQNLYRHEGIJ-UHFFFAOYSA-J potassium aluminium sulfate Chemical compound [Al+3].[K+].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O GRLPQNLYRHEGIJ-UHFFFAOYSA-J 0.000 claims description 6
- 229940094037 potassium bromate Drugs 0.000 claims description 6
- 235000019396 potassium bromate Nutrition 0.000 claims description 6
- 239000001508 potassium citrate Substances 0.000 claims description 6
- 229960002635 potassium citrate Drugs 0.000 claims description 6
- QEEAPRPFLLJWCF-UHFFFAOYSA-K potassium citrate (anhydrous) Chemical compound [K+].[K+].[K+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O QEEAPRPFLLJWCF-UHFFFAOYSA-K 0.000 claims description 6
- 235000011082 potassium citrates Nutrition 0.000 claims description 6
- 239000000276 potassium ferrocyanide Substances 0.000 claims description 6
- 239000004224 potassium gluconate Substances 0.000 claims description 6
- 235000013926 potassium gluconate Nutrition 0.000 claims description 6
- 229960003189 potassium gluconate Drugs 0.000 claims description 6
- 229940086065 potassium hydrogentartrate Drugs 0.000 claims description 6
- 229940093932 potassium hydroxide Drugs 0.000 claims description 6
- 235000011118 potassium hydroxide Nutrition 0.000 claims description 6
- PHZLMBHDXVLRIX-UHFFFAOYSA-M potassium lactate Chemical compound [K+].CC(O)C([O-])=O PHZLMBHDXVLRIX-UHFFFAOYSA-M 0.000 claims description 6
- 235000011085 potassium lactate Nutrition 0.000 claims description 6
- 239000001521 potassium lactate Substances 0.000 claims description 6
- 229960001304 potassium lactate Drugs 0.000 claims description 6
- RWPGFSMJFRPDDP-UHFFFAOYSA-L potassium metabisulfite Chemical compound [K+].[K+].[O-]S(=O)S([O-])(=O)=O RWPGFSMJFRPDDP-UHFFFAOYSA-L 0.000 claims description 6
- 235000010263 potassium metabisulphite Nutrition 0.000 claims description 6
- 229940099402 potassium metaphosphate Drugs 0.000 claims description 6
- 235000010333 potassium nitrate Nutrition 0.000 claims description 6
- 239000004323 potassium nitrate Substances 0.000 claims description 6
- 235000019828 potassium polyphosphate Nutrition 0.000 claims description 6
- 235000010241 potassium sorbate Nutrition 0.000 claims description 6
- 239000004302 potassium sorbate Substances 0.000 claims description 6
- 229940069338 potassium sorbate Drugs 0.000 claims description 6
- OTYBMLCTZGSZBG-UHFFFAOYSA-L potassium sulfate Chemical compound [K+].[K+].[O-]S([O-])(=O)=O OTYBMLCTZGSZBG-UHFFFAOYSA-L 0.000 claims description 6
- 229910052939 potassium sulfate Inorganic materials 0.000 claims description 6
- 235000011151 potassium sulphates Nutrition 0.000 claims description 6
- 239000002336 ribonucleotide Substances 0.000 claims description 6
- 235000019265 sodium DL-malate Nutrition 0.000 claims description 6
- HELHAJAZNSDZJO-OLXYHTOASA-L sodium L-tartrate Chemical compound [Na+].[Na+].[O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O HELHAJAZNSDZJO-OLXYHTOASA-L 0.000 claims description 6
- 235000010378 sodium ascorbate Nutrition 0.000 claims description 6
- PPASLZSBLFJQEF-RKJRWTFHSA-M sodium ascorbate Substances [Na+].OC[C@@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RKJRWTFHSA-M 0.000 claims description 6
- 229960005055 sodium ascorbate Drugs 0.000 claims description 6
- 229910000029 sodium carbonate Inorganic materials 0.000 claims description 6
- 229940080237 sodium caseinate Drugs 0.000 claims description 6
- 239000001509 sodium citrate Substances 0.000 claims description 6
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 claims description 6
- SRRKNRDXURUMPP-UHFFFAOYSA-N sodium disulfide Chemical compound [Na+].[Na+].[S-][S-] SRRKNRDXURUMPP-UHFFFAOYSA-N 0.000 claims description 6
- 229940073490 sodium glutamate Drugs 0.000 claims description 6
- 239000001540 sodium lactate Substances 0.000 claims description 6
- 235000011088 sodium lactate Nutrition 0.000 claims description 6
- 229940005581 sodium lactate Drugs 0.000 claims description 6
- 239000001394 sodium malate Substances 0.000 claims description 6
- 239000001488 sodium phosphate Substances 0.000 claims description 6
- 229910000162 sodium phosphate Inorganic materials 0.000 claims description 6
- 229910052938 sodium sulfate Inorganic materials 0.000 claims description 6
- 235000011152 sodium sulphate Nutrition 0.000 claims description 6
- 239000001433 sodium tartrate Substances 0.000 claims description 6
- 229960002167 sodium tartrate Drugs 0.000 claims description 6
- 235000011004 sodium tartrates Nutrition 0.000 claims description 6
- PPASLZSBLFJQEF-RXSVEWSESA-M sodium-L-ascorbate Chemical compound [Na+].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RXSVEWSESA-M 0.000 claims description 6
- WTWSHHITWMVLBX-DKWTVANSSA-M sodium;(2s)-2-aminobutanedioate;hydron Chemical compound [Na+].[O-]C(=O)[C@@H](N)CC(O)=O WTWSHHITWMVLBX-DKWTVANSSA-M 0.000 claims description 6
- FMYOMWCQJXWGEN-UHFFFAOYSA-M sodium;2,3,4,5,6,7-hexahydroxyheptanoate Chemical compound [Na+].OCC(O)C(O)C(O)C(O)C(O)C([O-])=O FMYOMWCQJXWGEN-UHFFFAOYSA-M 0.000 claims description 6
- XOGGUFAVLNCTRS-UHFFFAOYSA-N tetrapotassium;iron(2+);hexacyanide Chemical compound [K+].[K+].[K+].[K+].[Fe+2].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-] XOGGUFAVLNCTRS-UHFFFAOYSA-N 0.000 claims description 6
- RYCLIXPGLDDLTM-UHFFFAOYSA-J tetrapotassium;phosphonato phosphate Chemical compound [K+].[K+].[K+].[K+].[O-]P([O-])(=O)OP([O-])([O-])=O RYCLIXPGLDDLTM-UHFFFAOYSA-J 0.000 claims description 6
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 claims description 6
- 235000013337 tricalcium citrate Nutrition 0.000 claims description 6
- 229940078499 tricalcium phosphate Drugs 0.000 claims description 6
- 229910000391 tricalcium phosphate Inorganic materials 0.000 claims description 6
- 235000019731 tricalcium phosphate Nutrition 0.000 claims description 6
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 claims description 6
- BCZXFFBUYPCTSJ-UHFFFAOYSA-L Calcium propionate Chemical compound [Ca+2].CCC([O-])=O.CCC([O-])=O BCZXFFBUYPCTSJ-UHFFFAOYSA-L 0.000 claims description 5
- WQZGKKKJIJFFOK-IVMDWMLBSA-N D-allopyranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@H](O)[C@@H]1O WQZGKKKJIJFFOK-IVMDWMLBSA-N 0.000 claims description 5
- MKJXYGKVIBWPFZ-UHFFFAOYSA-L calcium lactate Chemical compound [Ca+2].CC(O)C([O-])=O.CC(O)C([O-])=O MKJXYGKVIBWPFZ-UHFFFAOYSA-L 0.000 claims description 5
- 239000001527 calcium lactate Substances 0.000 claims description 5
- 229960002401 calcium lactate Drugs 0.000 claims description 5
- 235000011086 calcium lactate Nutrition 0.000 claims description 5
- 235000010331 calcium propionate Nutrition 0.000 claims description 5
- 239000004330 calcium propionate Substances 0.000 claims description 5
- NIDRASOKXCQPKX-DFWYDOINSA-L calcium;(2s)-2-aminopentanedioate Chemical compound [Ca+2].[O-]C(=O)[C@@H](N)CCC([O-])=O NIDRASOKXCQPKX-DFWYDOINSA-L 0.000 claims description 5
- MHPZZOJHWAYMJY-ZKAUOVOBSA-L dipotassium;(2e,4e,6e,8e,10e,12e,14e,16e,18e)-4,8,13,17-tetramethylicosa-2,4,6,8,10,12,14,16,18-nonaenedioate Chemical compound [K+].[K+].[O-]C(=O)/C=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(\C)/C=C/C=C(\C)/C=C/C([O-])=O MHPZZOJHWAYMJY-ZKAUOVOBSA-L 0.000 claims description 5
- AVTYONGGKAJVTE-OLXYHTOASA-L potassium L-tartrate Chemical compound [K+].[K+].[O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O AVTYONGGKAJVTE-OLXYHTOASA-L 0.000 claims description 5
- 235000012249 potassium ferrocyanide Nutrition 0.000 claims description 5
- 229940093928 potassium nitrate Drugs 0.000 claims description 5
- 229910000404 tripotassium phosphate Inorganic materials 0.000 claims description 5
- 235000019798 tripotassium phosphate Nutrition 0.000 claims description 5
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 4
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 4
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 claims description 4
- 229940084030 carboxymethylcellulose calcium Drugs 0.000 claims description 4
- 159000000000 sodium salts Chemical class 0.000 claims 2
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric Acid Chemical compound [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 claims 1
- 235000011171 Thladiantha grosvenorii Nutrition 0.000 abstract description 2
- 241001409321 Siraitia grosvenorii Species 0.000 abstract 1
- HELXLJCILKEWJH-NCGAPWICSA-N rebaudioside A Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HELXLJCILKEWJH-NCGAPWICSA-N 0.000 description 46
- 239000001512 FEMA 4601 Substances 0.000 description 41
- HELXLJCILKEWJH-SEAGSNCFSA-N Rebaudioside A Natural products O=C(O[C@H]1[C@@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1)[C@@]1(C)[C@@H]2[C@](C)([C@H]3[C@@]4(CC(=C)[C@@](O[C@H]5[C@H](O[C@H]6[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O6)[C@@H](O[C@H]6[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O6)[C@H](O)[C@@H](CO)O5)(C4)CC3)CC2)CCC1 HELXLJCILKEWJH-SEAGSNCFSA-N 0.000 description 41
- HELXLJCILKEWJH-UHFFFAOYSA-N entered according to Sigma 01432 Natural products C1CC2C3(C)CCCC(C)(C(=O)OC4C(C(O)C(O)C(CO)O4)O)C3CCC2(C2)CC(=C)C21OC(C1OC2C(C(O)C(O)C(CO)O2)O)OC(CO)C(O)C1OC1OC(CO)C(O)C(O)C1O HELXLJCILKEWJH-UHFFFAOYSA-N 0.000 description 41
- 235000019203 rebaudioside A Nutrition 0.000 description 41
- 238000011156 evaluation Methods 0.000 description 36
- 230000001953 sensory effect Effects 0.000 description 31
- 235000013336 milk Nutrition 0.000 description 24
- 239000008267 milk Substances 0.000 description 24
- 210000004080 milk Anatomy 0.000 description 24
- AEQDJSLRWYMAQI-UHFFFAOYSA-N 2,3,9,10-tetramethoxy-6,8,13,13a-tetrahydro-5H-isoquinolino[2,1-b]isoquinoline Chemical compound C1CN2CC(C(=C(OC)C=C3)OC)=C3CC2C2=C1C=C(OC)C(OC)=C2 AEQDJSLRWYMAQI-UHFFFAOYSA-N 0.000 description 20
- 238000006243 chemical reaction Methods 0.000 description 20
- 235000008504 concentrate Nutrition 0.000 description 20
- 239000000176 sodium gluconate Substances 0.000 description 20
- 235000012207 sodium gluconate Nutrition 0.000 description 20
- 229940005574 sodium gluconate Drugs 0.000 description 20
- 230000000694 effects Effects 0.000 description 17
- 229940083542 sodium Drugs 0.000 description 16
- 239000000796 flavoring agent Substances 0.000 description 15
- 235000019634 flavors Nutrition 0.000 description 15
- 235000006468 Thea sinensis Nutrition 0.000 description 14
- 238000002156 mixing Methods 0.000 description 11
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- 235000019658 bitter taste Nutrition 0.000 description 10
- 150000001875 compounds Chemical class 0.000 description 10
- 230000001965 increasing effect Effects 0.000 description 9
- 239000007788 liquid Substances 0.000 description 9
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 9
- 235000013305 food Nutrition 0.000 description 8
- 235000020333 oolong tea Nutrition 0.000 description 8
- 235000019606 astringent taste Nutrition 0.000 description 7
- 230000001747 exhibiting effect Effects 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 235000009024 Ceanothus sanguineus Nutrition 0.000 description 6
- 240000003553 Leptospermum scoparium Species 0.000 description 6
- 235000015459 Lycium barbarum Nutrition 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 239000004376 Sucralose Substances 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 238000002474 experimental method Methods 0.000 description 6
- 238000000605 extraction Methods 0.000 description 6
- 150000003839 salts Chemical class 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 235000019408 sucralose Nutrition 0.000 description 6
- BAQAVOSOZGMPRM-QBMZZYIRSA-N sucralose Chemical compound O[C@@H]1[C@@H](O)[C@@H](Cl)[C@@H](CO)O[C@@H]1O[C@@]1(CCl)[C@@H](O)[C@H](O)[C@@H](CCl)O1 BAQAVOSOZGMPRM-QBMZZYIRSA-N 0.000 description 6
- 244000228451 Stevia rebaudiana Species 0.000 description 5
- -1 acidulant Substances 0.000 description 5
- 235000009508 confectionery Nutrition 0.000 description 5
- 229930182470 glycoside Natural products 0.000 description 5
- 235000009569 green tea Nutrition 0.000 description 5
- WBZFUFAFFUEMEI-UHFFFAOYSA-M Acesulfame k Chemical compound [K+].CC1=CC(=O)[N-]S(=O)(=O)O1 WBZFUFAFFUEMEI-UHFFFAOYSA-M 0.000 description 4
- 241000196324 Embryophyta Species 0.000 description 4
- 239000001329 FEMA 3811 Substances 0.000 description 4
- 244000303040 Glycyrrhiza glabra Species 0.000 description 4
- 235000006200 Glycyrrhiza glabra Nutrition 0.000 description 4
- 239000000619 acesulfame-K Substances 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 235000020279 black tea Nutrition 0.000 description 4
- 150000002338 glycosides Chemical class 0.000 description 4
- LPLVUJXQOOQHMX-QWBHMCJMSA-N glycyrrhizinic acid Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@H](O[C@@H]1O[C@@H]1C([C@H]2[C@]([C@@H]3[C@@]([C@@]4(CC[C@@]5(C)CC[C@@](C)(C[C@H]5C4=CC3=O)C(O)=O)C)(C)CC2)(C)CC1)(C)C)C(O)=O)[C@@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O LPLVUJXQOOQHMX-QWBHMCJMSA-N 0.000 description 4
- 229940094952 green tea extract Drugs 0.000 description 4
- 229940069445 licorice extract Drugs 0.000 description 4
- ITVGXXMINPYUHD-CUVHLRMHSA-N neohesperidin dihydrochalcone Chemical compound C1=C(O)C(OC)=CC=C1CCC(=O)C(C(=C1)O)=C(O)C=C1O[C@H]1[C@H](O[C@H]2[C@@H]([C@H](O)[C@@H](O)[C@H](C)O2)O)[C@@H](O)[C@H](O)[C@@H](CO)O1 ITVGXXMINPYUHD-CUVHLRMHSA-N 0.000 description 4
- 229940089953 neohesperidin dihydrochalcone Drugs 0.000 description 4
- 235000010434 neohesperidine DC Nutrition 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 102000004169 proteins and genes Human genes 0.000 description 4
- 108090000623 proteins and genes Proteins 0.000 description 4
- 230000001954 sterilising effect Effects 0.000 description 4
- 235000001453 Glycyrrhiza echinata Nutrition 0.000 description 3
- 235000017382 Glycyrrhiza lepidota Nutrition 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 235000001465 calcium Nutrition 0.000 description 3
- 235000013339 cereals Nutrition 0.000 description 3
- 235000014113 dietary fatty acids Nutrition 0.000 description 3
- 239000000194 fatty acid Substances 0.000 description 3
- 229930195729 fatty acid Natural products 0.000 description 3
- LPLVUJXQOOQHMX-UHFFFAOYSA-N glycyrrhetinic acid glycoside Natural products C1CC(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2=O)C(O)=O)C)(C)CC2)(C)C2C(C)(C)C1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O LPLVUJXQOOQHMX-UHFFFAOYSA-N 0.000 description 3
- 229960004949 glycyrrhizic acid Drugs 0.000 description 3
- UYRUBYNTXSDKQT-UHFFFAOYSA-N glycyrrhizic acid Natural products CC1(C)C(CCC2(C)C1CCC3(C)C2C(=O)C=C4C5CC(C)(CCC5(C)CCC34C)C(=O)O)OC6OC(C(O)C(O)C6OC7OC(O)C(O)C(O)C7C(=O)O)C(=O)O UYRUBYNTXSDKQT-UHFFFAOYSA-N 0.000 description 3
- 235000019410 glycyrrhizin Nutrition 0.000 description 3
- 235000020688 green tea extract Nutrition 0.000 description 3
- 235000008216 herbs Nutrition 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 230000003993 interaction Effects 0.000 description 3
- 229940010454 licorice Drugs 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 238000004659 sterilization and disinfection Methods 0.000 description 3
- 210000001779 taste bud Anatomy 0.000 description 3
- WRPAFPPCKSYACJ-ZBYJYCAASA-N (2r,3r,4s,5s,6r)-2-[[(2r,3s,4s,5r,6r)-6-[[(3s,8r,9r,10s,11r,13r,14s,17r)-17-[(5r)-5-[(2s,3r,4s,5s,6r)-4,5-dihydroxy-6-(hydroxymethyl)-3-[(2r,3s,4r,5r,6s)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxan-2-yl]oxy-6-hydroxy-6-methylheptan-2-yl]-11-hydrox Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@H](CCC(C)[C@@H]1[C@]2(C[C@@H](O)[C@@]3(C)[C@@H]4C(C([C@@H](O[C@H]5[C@@H]([C@@H](O)[C@H](O)[C@@H](CO[C@H]6[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O6)O)O5)O)CC4)(C)C)=CC[C@@H]3[C@]2(C)CC1)C)C(C)(C)O)[C@H]1O[C@@H](CO)[C@H](O)[C@@H](O)[C@@H]1O WRPAFPPCKSYACJ-ZBYJYCAASA-N 0.000 description 2
- YTKBWWKAVMSYHE-OALUTQOASA-N (3s)-3-[3-(3-hydroxy-4-methoxyphenyl)propylamino]-4-[[(2s)-1-methoxy-1-oxo-3-phenylpropan-2-yl]amino]-4-oxobutanoic acid Chemical compound C([C@@H](C(=O)OC)NC(=O)[C@H](CC(O)=O)NCCCC=1C=C(O)C(OC)=CC=1)C1=CC=CC=C1 YTKBWWKAVMSYHE-OALUTQOASA-N 0.000 description 2
- 239000004394 Advantame Substances 0.000 description 2
- 241000512259 Ascophyllum nodosum Species 0.000 description 2
- 235000017166 Bambusa arundinacea Nutrition 0.000 description 2
- 235000017491 Bambusa tulda Nutrition 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- 239000004378 Glycyrrhizin Substances 0.000 description 2
- 101000659765 Homo sapiens Taste receptor type 1 member 2 Proteins 0.000 description 2
- 101000659774 Homo sapiens Taste receptor type 1 member 3 Proteins 0.000 description 2
- 240000005979 Hordeum vulgare Species 0.000 description 2
- 235000007340 Hordeum vulgare Nutrition 0.000 description 2
- 108010093901 N-(N-(3-(3-hydroxy-4-methoxyphenyl) propyl)-alpha-aspartyl)-L-phenylalanine 1-methyl ester Proteins 0.000 description 2
- 244000082204 Phyllostachys viridis Species 0.000 description 2
- 235000015334 Phyllostachys viridis Nutrition 0.000 description 2
- 102100035948 Taste receptor type 1 member 2 Human genes 0.000 description 2
- 102100035942 Taste receptor type 1 member 3 Human genes 0.000 description 2
- 235000019453 advantame Nutrition 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 239000011425 bamboo Substances 0.000 description 2
- 235000012241 calcium silicate Nutrition 0.000 description 2
- BLORRZQTHNGFTI-ZZMNMWMASA-L calcium-L-ascorbate Chemical compound [Ca+2].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-] BLORRZQTHNGFTI-ZZMNMWMASA-L 0.000 description 2
- 239000002967 calcium-L-ascorbate Substances 0.000 description 2
- 235000005937 calcium-L-ascorbate Nutrition 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 229940105329 carboxymethylcellulose Drugs 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 238000012364 cultivation method Methods 0.000 description 2
- VFLDPWHFBUODDF-FCXRPNKRSA-N curcumin Chemical compound C1=C(O)C(OC)=CC(\C=C\C(=O)CC(=O)\C=C\C=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-FCXRPNKRSA-N 0.000 description 2
- 235000013365 dairy product Nutrition 0.000 description 2
- 238000007865 diluting Methods 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 238000004993 emission spectroscopy Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 235000019225 fermented tea Nutrition 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 235000013373 food additive Nutrition 0.000 description 2
- 239000002778 food additive Substances 0.000 description 2
- 108091005708 gustatory receptors Proteins 0.000 description 2
- 239000008233 hard water Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 229930191869 mogroside IV Natural products 0.000 description 2
- OKGRRPCHOJYNKX-UHFFFAOYSA-N mogroside IV A Natural products C1CC2(C)C3CC=C(C(C(OC4C(C(O)C(O)C(COC5C(C(O)C(O)C(CO)O5)O)O4)O)CC4)(C)C)C4C3(C)C(O)CC2(C)C1C(C)CCC(C(C)(C)O)OC(C(C(O)C1O)O)OC1COC1OC(CO)C(O)C(O)C1O OKGRRPCHOJYNKX-UHFFFAOYSA-N 0.000 description 2
- WRPAFPPCKSYACJ-UHFFFAOYSA-N mogroside IV E Natural products C1CC2(C)C3CC=C(C(C(OC4C(C(O)C(O)C(COC5C(C(O)C(O)C(CO)O5)O)O4)O)CC4)(C)C)C4C3(C)C(O)CC2(C)C1C(C)CCC(C(C)(C)O)OC1OC(CO)C(O)C(O)C1OC1OC(CO)C(O)C(O)C1O WRPAFPPCKSYACJ-UHFFFAOYSA-N 0.000 description 2
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 2
- 235000019796 monopotassium phosphate Nutrition 0.000 description 2
- PJNZPQUBCPKICU-UHFFFAOYSA-N phosphoric acid;potassium Chemical compound [K].OP(O)(O)=O PJNZPQUBCPKICU-UHFFFAOYSA-N 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 229940093956 potassium carbonate Drugs 0.000 description 2
- 235000011181 potassium carbonates Nutrition 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 230000002335 preservative effect Effects 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- QSRAJVGDWKFOGU-WBXIDTKBSA-N rebaudioside c Chemical compound O[C@@H]1[C@H](O)[C@@H](O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](O[C@H]2[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O2)O)[C@H](O)[C@@H](CO)O[C@H]1O[C@]1(CC[C@H]2[C@@]3(C)[C@@H]([C@](CCC3)(C)C(=O)O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O3)O)CC3)C(=C)C[C@]23C1 QSRAJVGDWKFOGU-WBXIDTKBSA-N 0.000 description 2
- 235000019600 saltiness Nutrition 0.000 description 2
- 210000000697 sensory organ Anatomy 0.000 description 2
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 235000019202 steviosides Nutrition 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 150000003445 sucroses Chemical class 0.000 description 2
- 229940092665 tea leaf extract Drugs 0.000 description 2
- RMLYXMMBIZLGAQ-UHFFFAOYSA-N (-)-monatin Natural products C1=CC=C2C(CC(O)(CC(N)C(O)=O)C(O)=O)=CNC2=C1 RMLYXMMBIZLGAQ-UHFFFAOYSA-N 0.000 description 1
- XFZJEEAOWLFHDH-UHFFFAOYSA-N (2R,2'R,3R,3'R,4R)-3,3',4',5,7-Pentahydroxyflavan(48)-3,3',4',5,7-pentahydroxyflavan Natural products C=12OC(C=3C=C(O)C(O)=CC=3)C(O)CC2=C(O)C=C(O)C=1C(C1=C(O)C=C(O)C=C1O1)C(O)C1C1=CC=C(O)C(O)=C1 XFZJEEAOWLFHDH-UHFFFAOYSA-N 0.000 description 1
- 239000001100 (2S)-5,7-dihydroxy-2-(3-hydroxy-4-methoxyphenyl)chroman-4-one Substances 0.000 description 1
- XJIPREFALCDWRQ-UYQGGQRHSA-N (2r,3r,4s,5s,6r)-2-[[(2r,3s,4s,5r,6s)-3,4-dihydroxy-6-[(3r,6r)-2-hydroxy-6-[(3s,8s,9r,10r,11r,13r,14s,17r)-11-hydroxy-4,4,9,13,14-pentamethyl-3-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-2,3,7,8,10,11,12,15,16,17-decahydro-1h-cyclop Chemical compound C([C@H]1O[C@H]([C@@H]([C@@H](O)[C@@H]1O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@H](CC[C@@H](C)[C@@H]1[C@]2(C[C@@H](O)[C@@]3(C)[C@H]4C(C([C@@H](O[C@H]5[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O5)O)CC4)(C)C)=CC[C@H]3[C@]2(C)CC1)C)C(C)(C)O)O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O XJIPREFALCDWRQ-UYQGGQRHSA-N 0.000 description 1
- RMLYXMMBIZLGAQ-HZMBPMFUSA-N (2s,4s)-4-amino-2-hydroxy-2-(1h-indol-3-ylmethyl)pentanedioic acid Chemical compound C1=CC=C2C(C[C@](O)(C[C@H](N)C(O)=O)C(O)=O)=CNC2=C1 RMLYXMMBIZLGAQ-HZMBPMFUSA-N 0.000 description 1
- CGGWHBLPUUKEJC-HRTKKJOOSA-N (3S,8R,9R,10R,13R,14S,17R)-17-[(2R,5R)-5-[(2S,3R,4S,5S,6R)-4,5-dihydroxy-3-[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-6-[[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]oxan-2-yl]oxy-6-hydroxy-6-methylheptan-2-yl]-4,4,9,13,14-pentamethyl-3-[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]oxan-2-yl]oxy-1,2,3,7,8,10,12,15,16,17-decahydrocyclopenta[a]phenanthren-11-one Chemical compound C[C@H](CC[C@@H](O[C@@H]1O[C@H](CO[C@@H]2O[C@H](CO)[C@@H](O)[C@H](O)[C@H]2O)[C@@H](O)[C@H](O)[C@H]1O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O)C(C)(C)O)[C@H]1CC[C@@]2(C)[C@H]3CC=C4[C@@H](CC[C@H](O[C@@H]5O[C@H](CO[C@@H]6O[C@H](CO)[C@@H](O)[C@H](O)[C@H]6O)[C@@H](O)[C@H](O)[C@H]5O)C4(C)C)[C@]3(C)C(=O)C[C@]12C CGGWHBLPUUKEJC-HRTKKJOOSA-N 0.000 description 1
- CGGWHBLPUUKEJC-UHFFFAOYSA-N 11-oxomogroside V Natural products C1CC2(C)C3CC=C(C(C(OC4C(C(O)C(O)C(COC5C(C(O)C(O)C(CO)O5)O)O4)O)CC4)(C)C)C4C3(C)C(=O)CC2(C)C1C(C)CCC(C(C)(C)O)OC(C(C(O)C1O)OC2C(C(O)C(O)C(CO)O2)O)OC1COC1OC(CO)C(O)C(O)C1O CGGWHBLPUUKEJC-UHFFFAOYSA-N 0.000 description 1
- GOLORTLGFDVFDW-UHFFFAOYSA-N 3-(1h-benzimidazol-2-yl)-7-(diethylamino)chromen-2-one Chemical compound C1=CC=C2NC(C3=CC4=CC=C(C=C4OC3=O)N(CC)CC)=NC2=C1 GOLORTLGFDVFDW-UHFFFAOYSA-N 0.000 description 1
- CWVRJTMFETXNAD-FWCWNIRPSA-N 3-O-Caffeoylquinic acid Natural products O[C@H]1[C@@H](O)C[C@@](O)(C(O)=O)C[C@H]1OC(=O)\C=C\C1=CC=C(O)C(O)=C1 CWVRJTMFETXNAD-FWCWNIRPSA-N 0.000 description 1
- GMVPRGQOIOIIMI-DODZYUBVSA-N 7-[(1R,2R,3R)-3-hydroxy-2-[(3S)-3-hydroxyoct-1-enyl]-5-oxocyclopentyl]heptanoic acid Chemical compound CCCCC[C@H](O)C=C[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O GMVPRGQOIOIIMI-DODZYUBVSA-N 0.000 description 1
- 240000006274 Actinidia polygama Species 0.000 description 1
- 235000016413 Actinidia polygama Nutrition 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 240000006914 Aspalathus linearis Species 0.000 description 1
- 235000012984 Aspalathus linearis Nutrition 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 235000021537 Beetroot Nutrition 0.000 description 1
- JMGZEFIQIZZSBH-UHFFFAOYSA-N Bioquercetin Natural products CC1OC(OCC(O)C2OC(OC3=C(Oc4cc(O)cc(O)c4C3=O)c5ccc(O)c(O)c5)C(O)C2O)C(O)C(O)C1O JMGZEFIQIZZSBH-UHFFFAOYSA-N 0.000 description 1
- PZIRUHCJZBGLDY-UHFFFAOYSA-N Caffeoylquinic acid Natural products CC(CCC(=O)C(C)C1C(=O)CC2C3CC(O)C4CC(O)CCC4(C)C3CCC12C)C(=O)O PZIRUHCJZBGLDY-UHFFFAOYSA-N 0.000 description 1
- 235000019224 Camellia sinensis var Qingmao Nutrition 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 241000951471 Citrus junos Species 0.000 description 1
- AFSDNFLWKVMVRB-UHFFFAOYSA-N Ellagic acid Chemical compound OC1=C(O)C(OC2=O)=C3C4=C2C=C(O)C(O)=C4OC(=O)C3=C1 AFSDNFLWKVMVRB-UHFFFAOYSA-N 0.000 description 1
- ATJXMQHAMYVHRX-CPCISQLKSA-N Ellagic acid Natural products OC1=C(O)[C@H]2OC(=O)c3cc(O)c(O)c4OC(=O)C(=C1)[C@H]2c34 ATJXMQHAMYVHRX-CPCISQLKSA-N 0.000 description 1
- 229920002079 Ellagic acid Polymers 0.000 description 1
- 240000006890 Erythroxylum coca Species 0.000 description 1
- 239000001776 FEMA 4720 Substances 0.000 description 1
- 240000008620 Fagopyrum esculentum Species 0.000 description 1
- 235000009419 Fagopyrum esculentum Nutrition 0.000 description 1
- QUQPHWDTPGMPEX-UHFFFAOYSA-N Hesperidine Natural products C1=C(O)C(OC)=CC=C1C1OC2=CC(OC3C(C(O)C(O)C(COC4C(C(O)C(O)C(C)O4)O)O3)O)=CC(O)=C2C(=O)C1 QUQPHWDTPGMPEX-UHFFFAOYSA-N 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 240000000691 Houttuynia cordata Species 0.000 description 1
- 235000013719 Houttuynia cordata Nutrition 0.000 description 1
- 235000003368 Ilex paraguariensis Nutrition 0.000 description 1
- 241000824268 Kuma Species 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- 235000017858 Laurus nobilis Nutrition 0.000 description 1
- 240000000599 Lentinula edodes Species 0.000 description 1
- 235000001715 Lentinula edodes Nutrition 0.000 description 1
- VTAJIXDZFCRWBR-UHFFFAOYSA-N Licoricesaponin B2 Natural products C1C(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2)C(O)=O)C)(C)CC2)(C)C2C(C)(C)CC1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O VTAJIXDZFCRWBR-UHFFFAOYSA-N 0.000 description 1
- CWVRJTMFETXNAD-KLZCAUPSSA-N Neochlorogenin-saeure Natural products O[C@H]1C[C@@](O)(C[C@@H](OC(=O)C=Cc2ccc(O)c(O)c2)[C@@H]1O)C(=O)O CWVRJTMFETXNAD-KLZCAUPSSA-N 0.000 description 1
- 239000004384 Neotame Substances 0.000 description 1
- ZVKOASAVGLETCT-UOGKPENDSA-N Norbixin Chemical compound OC(=O)/C=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(\C)/C=C/C=C(\C)/C=C/C(O)=O ZVKOASAVGLETCT-UOGKPENDSA-N 0.000 description 1
- JERYLJRGLVHIEW-UENHKZIGSA-N Norbixin Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C(=O)O)C=CC=CC=CC(=O)O JERYLJRGLVHIEW-UENHKZIGSA-N 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- 244000124853 Perilla frutescens Species 0.000 description 1
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 1
- 244000046052 Phaseolus vulgaris Species 0.000 description 1
- CWEZAWNPTYBADX-UHFFFAOYSA-N Procyanidin Natural products OC1C(OC2C(O)C(Oc3c2c(O)cc(O)c3C4C(O)C(Oc5cc(O)cc(O)c45)c6ccc(O)c(O)c6)c7ccc(O)c(O)c7)c8c(O)cc(O)cc8OC1c9ccc(O)c(O)c9 CWEZAWNPTYBADX-UHFFFAOYSA-N 0.000 description 1
- MOJZMWJRUKIQGL-FWCKPOPSSA-N Procyanidin C2 Natural products O[C@@H]1[C@@H](c2cc(O)c(O)cc2)Oc2c([C@H]3[C@H](O)[C@@H](c4cc(O)c(O)cc4)Oc4c3c(O)cc(O)c4)c(O)cc(O)c2[C@@H]1c1c(O)cc(O)c2c1O[C@@H]([C@H](O)C2)c1cc(O)c(O)cc1 MOJZMWJRUKIQGL-FWCKPOPSSA-N 0.000 description 1
- GIPHUOWOTCAJSR-UHFFFAOYSA-N Rebaudioside A. Natural products C1CC2C3(C)CCCC(C)(C(=O)OC4C(C(O)C(O)C(CO)O4)O)C3CCC2(C2)CC(=C)C21OC1OC(CO)C(O)C(O)C1OC(C1O)OC(CO)C(O)C1OC1OC(CO)C(O)C(O)C1O GIPHUOWOTCAJSR-UHFFFAOYSA-N 0.000 description 1
- QNVSXXGDAPORNA-UHFFFAOYSA-N Resveratrol Natural products OC1=CC=CC(C=CC=2C=C(O)C(O)=CC=2)=C1 QNVSXXGDAPORNA-UHFFFAOYSA-N 0.000 description 1
- 239000004288 Sodium dehydroacetate Substances 0.000 description 1
- QFVOYBUQQBFCRH-UHFFFAOYSA-N Steviol Natural products C1CC2(C3)CC(=C)C3(O)CCC2C2(C)C1C(C)(C(O)=O)CCC2 QFVOYBUQQBFCRH-UHFFFAOYSA-N 0.000 description 1
- 239000004383 Steviol glycoside Substances 0.000 description 1
- UEDUENGHJMELGK-HYDKPPNVSA-N Stevioside Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O UEDUENGHJMELGK-HYDKPPNVSA-N 0.000 description 1
- 208000025371 Taste disease Diseases 0.000 description 1
- 244000125380 Terminalia tomentosa Species 0.000 description 1
- 235000005212 Terminalia tomentosa Nutrition 0.000 description 1
- 244000185386 Thladiantha grosvenorii Species 0.000 description 1
- LUKBXSAWLPMMSZ-OWOJBTEDSA-N Trans-resveratrol Chemical compound C1=CC(O)=CC=C1\C=C\C1=CC(O)=CC(O)=C1 LUKBXSAWLPMMSZ-OWOJBTEDSA-N 0.000 description 1
- YGCFIWIQZPHFLU-UHFFFAOYSA-N acesulfame Chemical compound CC1=CC(=O)NS(=O)(=O)O1 YGCFIWIQZPHFLU-UHFFFAOYSA-N 0.000 description 1
- 229960005164 acesulfame Drugs 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- ZVKOASAVGLETCT-UOAMSCJGSA-N all-trans norbixin Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C(=O)O)C=CC=C(/C)C=CC(=O)O ZVKOASAVGLETCT-UOAMSCJGSA-N 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000001670 anatto Substances 0.000 description 1
- 235000012665 annatto Nutrition 0.000 description 1
- 235000010208 anthocyanin Nutrition 0.000 description 1
- 239000004410 anthocyanin Substances 0.000 description 1
- 229930002877 anthocyanin Natural products 0.000 description 1
- 150000004636 anthocyanins Chemical class 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 235000009697 arginine Nutrition 0.000 description 1
- 235000019568 aromas Nutrition 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- QUQPHWDTPGMPEX-UTWYECKDSA-N aurantiamarin Natural products COc1ccc(cc1O)[C@H]1CC(=O)c2c(O)cc(O[C@@H]3O[C@H](CO[C@@H]4O[C@@H](C)[C@H](O)[C@@H](O)[C@H]4O)[C@@H](O)[C@H](O)[C@H]3O)cc2O1 QUQPHWDTPGMPEX-UTWYECKDSA-N 0.000 description 1
- 210000000270 basal cell Anatomy 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 235000013921 calcium diglutamate Nutrition 0.000 description 1
- 235000010957 calcium stearoyl-2-lactylate Nutrition 0.000 description 1
- UMVAYAXXQSFULN-QHTZZOMLSA-L calcium;(2s)-2-aminopentanedioate;hydron Chemical compound [Ca+2].[O-]C(=O)[C@@H](N)CCC(O)=O.[O-]C(=O)[C@@H](N)CCC(O)=O UMVAYAXXQSFULN-QHTZZOMLSA-L 0.000 description 1
- SMHNUIFHMAGAFL-UHFFFAOYSA-N calcium;2-hydroxypropanoic acid Chemical compound [Ca].CC(O)C(O)=O SMHNUIFHMAGAFL-UHFFFAOYSA-N 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 229940074393 chlorogenic acid Drugs 0.000 description 1
- 235000001368 chlorogenic acid Nutrition 0.000 description 1
- CWVRJTMFETXNAD-JUHZACGLSA-N chlorogenic acid Chemical compound O[C@@H]1[C@H](O)C[C@@](O)(C(O)=O)C[C@H]1OC(=O)\C=C\C1=CC=C(O)C(O)=C1 CWVRJTMFETXNAD-JUHZACGLSA-N 0.000 description 1
- FFQSDFBBSXGVKF-KHSQJDLVSA-N chlorogenic acid Natural products O[C@@H]1C[C@](O)(C[C@@H](CC(=O)C=Cc2ccc(O)c(O)c2)[C@@H]1O)C(=O)O FFQSDFBBSXGVKF-KHSQJDLVSA-N 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- BMRSEYFENKXDIS-KLZCAUPSSA-N cis-3-O-p-coumaroylquinic acid Natural products O[C@H]1C[C@@](O)(C[C@@H](OC(=O)C=Cc2ccc(O)cc2)[C@@H]1O)C(=O)O BMRSEYFENKXDIS-KLZCAUPSSA-N 0.000 description 1
- APSNPMVGBGZYAJ-GLOOOPAXSA-N clematine Natural products COc1cc(ccc1O)[C@@H]2CC(=O)c3c(O)cc(O[C@@H]4O[C@H](CO[C@H]5O[C@@H](C)[C@H](O)[C@@H](O)[C@H]5O)[C@@H](O)[C@H](O)[C@H]4O)cc3O2 APSNPMVGBGZYAJ-GLOOOPAXSA-N 0.000 description 1
- 235000008957 cocaer Nutrition 0.000 description 1
- ZPUCINDJVBIVPJ-LJISPDSOSA-N cocaine Chemical compound O([C@H]1C[C@@H]2CC[C@@H](N2C)[C@H]1C(=O)OC)C(=O)C1=CC=CC=C1 ZPUCINDJVBIVPJ-LJISPDSOSA-N 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 229920002770 condensed tannin Polymers 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 108010010165 curculin Proteins 0.000 description 1
- 235000012754 curcumin Nutrition 0.000 description 1
- 239000004148 curcumin Substances 0.000 description 1
- 229940109262 curcumin Drugs 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 229940109275 cyclamate Drugs 0.000 description 1
- HCAJEUSONLESMK-UHFFFAOYSA-N cyclohexylsulfamic acid Chemical compound OS(=O)(=O)NC1CCCCC1 HCAJEUSONLESMK-UHFFFAOYSA-N 0.000 description 1
- 230000002354 daily effect Effects 0.000 description 1
- 238000005238 degreasing Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- VFLDPWHFBUODDF-UHFFFAOYSA-N diferuloylmethane Natural products C1=C(O)C(OC)=CC(C=CC(=O)CC(=O)C=CC=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 229930004069 diterpene Natural products 0.000 description 1
- 150000004141 diterpene derivatives Chemical class 0.000 description 1
- 229960002852 ellagic acid Drugs 0.000 description 1
- 235000004132 ellagic acid Nutrition 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- IVTMALDHFAHOGL-UHFFFAOYSA-N eriodictyol 7-O-rutinoside Natural products OC1C(O)C(O)C(C)OC1OCC1C(O)C(O)C(O)C(OC=2C=C3C(C(C(O)=C(O3)C=3C=C(O)C(O)=CC=3)=O)=C(O)C=2)O1 IVTMALDHFAHOGL-UHFFFAOYSA-N 0.000 description 1
- 230000003203 everyday effect Effects 0.000 description 1
- 238000010304 firing Methods 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 235000021255 galacto-oligosaccharides Nutrition 0.000 description 1
- 150000003271 galactooligosaccharides Chemical class 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 239000001685 glycyrrhizic acid Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 210000004209 hair Anatomy 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229940025878 hesperidin Drugs 0.000 description 1
- VUYDGVRIQRPHFX-UHFFFAOYSA-N hesperidin Natural products COc1cc(ccc1O)C2CC(=O)c3c(O)cc(OC4OC(COC5OC(O)C(O)C(O)C5O)C(O)C(O)C4O)cc3O2 VUYDGVRIQRPHFX-UHFFFAOYSA-N 0.000 description 1
- QUQPHWDTPGMPEX-QJBIFVCTSA-N hesperidin Chemical compound C1=C(O)C(OC)=CC=C1[C@H]1OC2=CC(O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@@H](CO[C@H]4[C@@H]([C@H](O)[C@@H](O)[C@H](C)O4)O)O3)O)=CC(O)=C2C(=O)C1 QUQPHWDTPGMPEX-QJBIFVCTSA-N 0.000 description 1
- 239000000833 heterodimer Substances 0.000 description 1
- 235000020340 honeybush tea Nutrition 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- GOMNOOKGLZYEJT-UHFFFAOYSA-N isoflavone Chemical compound C=1OC2=CC=CC=C2C(=O)C=1C1=CC=CC=C1 GOMNOOKGLZYEJT-UHFFFAOYSA-N 0.000 description 1
- CJWQYWQDLBZGPD-UHFFFAOYSA-N isoflavone Natural products C1=C(OC)C(OC)=CC(OC)=C1C1=COC2=C(C=CC(C)(C)O3)C3=C(OC)C=C2C1=O CJWQYWQDLBZGPD-UHFFFAOYSA-N 0.000 description 1
- 235000008696 isoflavones Nutrition 0.000 description 1
- 229930013686 lignan Natural products 0.000 description 1
- 235000009408 lignans Nutrition 0.000 description 1
- 150000005692 lignans Chemical class 0.000 description 1
- 238000004811 liquid chromatography Methods 0.000 description 1
- 235000011477 liquorice Nutrition 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 235000020331 mate tea Nutrition 0.000 description 1
- 235000019656 metallic taste Nutrition 0.000 description 1
- FAARLWTXUUQFSN-UHFFFAOYSA-N methylellagic acid Natural products O1C(=O)C2=CC(O)=C(O)C3=C2C2=C1C(OC)=C(O)C=C2C(=O)O3 FAARLWTXUUQFSN-UHFFFAOYSA-N 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- JLYBBRAAICDTIS-UHFFFAOYSA-N mogrol Natural products CC12C(O)CC3(C)C(C(CCC(O)C(C)(C)O)C)CCC3(C)C1CC=C1C2CCC(O)C1(C)C JLYBBRAAICDTIS-UHFFFAOYSA-N 0.000 description 1
- 229930189775 mogroside Natural products 0.000 description 1
- 239000004570 mortar (masonry) Substances 0.000 description 1
- ARGKVCXINMKCAZ-UHFFFAOYSA-N neohesperidine Natural products C1=C(O)C(OC)=CC=C1C1OC2=CC(OC3C(C(O)C(O)C(CO)O3)OC3C(C(O)C(O)C(C)O3)O)=CC(O)=C2C(=O)C1 ARGKVCXINMKCAZ-UHFFFAOYSA-N 0.000 description 1
- 235000019412 neotame Nutrition 0.000 description 1
- HLIAVLHNDJUHFG-HOTGVXAUSA-N neotame Chemical compound CC(C)(C)CCN[C@@H](CC(O)=O)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 HLIAVLHNDJUHFG-HOTGVXAUSA-N 0.000 description 1
- 108010070257 neotame Proteins 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 210000004126 nerve fiber Anatomy 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- XIMSTDAFGWROBF-UHFFFAOYSA-N oxathiazine 2-oxide Chemical compound O=S1OC=CC=N1 XIMSTDAFGWROBF-UHFFFAOYSA-N 0.000 description 1
- 239000003002 pH adjusting agent Substances 0.000 description 1
- FDPIMTJIUBPUKL-UHFFFAOYSA-N pentan-3-one Chemical compound CCC(=O)CC FDPIMTJIUBPUKL-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 229920002414 procyanidin Polymers 0.000 description 1
- HGVVOUNEGQIPMS-UHFFFAOYSA-N procyanidin Chemical compound O1C2=CC(O)=CC(O)=C2C(O)C(O)C1(C=1C=C(O)C(O)=CC=1)OC1CC2=C(O)C=C(O)C=C2OC1C1=CC=C(O)C(O)=C1 HGVVOUNEGQIPMS-UHFFFAOYSA-N 0.000 description 1
- 235000020339 pu-erh tea Nutrition 0.000 description 1
- FDRQPMVGJOQVTL-UHFFFAOYSA-N quercetin rutinoside Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC=2C(C3=C(O)C=C(O)C=C3OC=2C=2C=C(O)C(O)=CC=2)=O)O1 FDRQPMVGJOQVTL-UHFFFAOYSA-N 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 235000021283 resveratrol Nutrition 0.000 description 1
- 229940016667 resveratrol Drugs 0.000 description 1
- 235000020330 rooibos tea Nutrition 0.000 description 1
- 235000005493 rutin Nutrition 0.000 description 1
- IKGXIBQEEMLURG-BKUODXTLSA-N rutin Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](C)O[C@@H]1OC[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](OC=2C(C3=C(O)C=C(O)C=C3OC=2C=2C=C(O)C(O)=CC=2)=O)O1 IKGXIBQEEMLURG-BKUODXTLSA-N 0.000 description 1
- ALABRVAAKCSLSC-UHFFFAOYSA-N rutin Natural products CC1OC(OCC2OC(O)C(O)C(O)C2O)C(O)C(O)C1OC3=C(Oc4cc(O)cc(O)c4C3=O)c5ccc(O)c(O)c5 ALABRVAAKCSLSC-UHFFFAOYSA-N 0.000 description 1
- 229960004555 rutoside Drugs 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 235000019643 salty taste Nutrition 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- XJIPREFALCDWRQ-UHFFFAOYSA-N siamenoside I Natural products C1CC2(C)C3CC=C(C(C(OC4C(C(O)C(O)C(CO)O4)O)CC4)(C)C)C4C3(C)C(O)CC2(C)C1C(C)CCC(C(C)(C)O)OC(C(C(O)C1O)OC2C(C(O)C(O)C(CO)O2)O)OC1COC1OC(CO)C(O)C(O)C1O XJIPREFALCDWRQ-UHFFFAOYSA-N 0.000 description 1
- 125000004436 sodium atom Chemical group 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 235000019259 sodium dehydroacetate Nutrition 0.000 description 1
- 229940079839 sodium dehydroacetate Drugs 0.000 description 1
- HRZFUMHJMZEROT-UHFFFAOYSA-L sodium disulfite Chemical compound [Na+].[Na+].[O-]S(=O)S([O-])(=O)=O HRZFUMHJMZEROT-UHFFFAOYSA-L 0.000 description 1
- 239000004320 sodium erythorbate Substances 0.000 description 1
- 235000010352 sodium erythorbate Nutrition 0.000 description 1
- 235000010262 sodium metabisulphite Nutrition 0.000 description 1
- JXKPEJDQGNYQSM-UHFFFAOYSA-M sodium propionate Chemical compound [Na+].CCC([O-])=O JXKPEJDQGNYQSM-UHFFFAOYSA-M 0.000 description 1
- 235000010334 sodium propionate Nutrition 0.000 description 1
- 239000004324 sodium propionate Substances 0.000 description 1
- 229960003212 sodium propionate Drugs 0.000 description 1
- 235000010265 sodium sulphite Nutrition 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 235000019345 sodium thiosulphate Nutrition 0.000 description 1
- DSOWAKKSGYUMTF-GZOLSCHFSA-M sodium;(1e)-1-(6-methyl-2,4-dioxopyran-3-ylidene)ethanolate Chemical compound [Na+].C\C([O-])=C1/C(=O)OC(C)=CC1=O DSOWAKKSGYUMTF-GZOLSCHFSA-M 0.000 description 1
- RBWSWDPRDBEWCR-RKJRWTFHSA-N sodium;(2r)-2-[(2r)-3,4-dihydroxy-5-oxo-2h-furan-2-yl]-2-hydroxyethanolate Chemical compound [Na+].[O-]C[C@@H](O)[C@H]1OC(=O)C(O)=C1O RBWSWDPRDBEWCR-RKJRWTFHSA-N 0.000 description 1
- 235000014347 soups Nutrition 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- QFVOYBUQQBFCRH-VQSWZGCSSA-N steviol Chemical compound C([C@@]1(O)C(=C)C[C@@]2(C1)CC1)C[C@H]2[C@@]2(C)[C@H]1[C@](C)(C(O)=O)CCC2 QFVOYBUQQBFCRH-VQSWZGCSSA-N 0.000 description 1
- 229940032084 steviol Drugs 0.000 description 1
- 229930182488 steviol glycoside Natural products 0.000 description 1
- 235000019411 steviol glycoside Nutrition 0.000 description 1
- 150000008144 steviol glycosides Chemical class 0.000 description 1
- OHHNJQXIOPOJSC-UHFFFAOYSA-N stevioside Natural products CC1(CCCC2(C)C3(C)CCC4(CC3(CCC12C)CC4=C)OC5OC(CO)C(O)C(O)C5OC6OC(CO)C(O)C(O)C6O)C(=O)OC7OC(CO)C(O)C(O)C7O OHHNJQXIOPOJSC-UHFFFAOYSA-N 0.000 description 1
- 229940013618 stevioside Drugs 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 235000019605 sweet taste sensations Nutrition 0.000 description 1
- 210000000225 synapse Anatomy 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 229920001864 tannin Polymers 0.000 description 1
- 235000018553 tannin Nutrition 0.000 description 1
- 239000001648 tannin Substances 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 210000005182 tip of the tongue Anatomy 0.000 description 1
- 238000012549 training Methods 0.000 description 1
- 238000011282 treatment Methods 0.000 description 1
- 239000001226 triphosphate Substances 0.000 description 1
- 235000011178 triphosphate Nutrition 0.000 description 1
- UNXRWKVEANCORM-UHFFFAOYSA-N triphosphoric acid Chemical compound OP(O)(=O)OP(O)(=O)OP(O)(O)=O UNXRWKVEANCORM-UHFFFAOYSA-N 0.000 description 1
- DRSKVOAJKLUMCL-MMUIXFKXSA-N u2n4xkx7hp Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(O)=O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O DRSKVOAJKLUMCL-MMUIXFKXSA-N 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L27/00—Spices; Flavouring agents or condiments; Artificial sweetening agents; Table salts; Dietetic salt substitutes; Preparation or treatment thereof
- A23L27/30—Artificial sweetening agents
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23F—COFFEE; TEA; THEIR SUBSTITUTES; MANUFACTURE, PREPARATION, OR INFUSION THEREOF
- A23F3/00—Tea; Tea substitutes; Preparations thereof
- A23F3/16—Tea extraction; Tea extracts; Treating tea extract; Making instant tea
- A23F3/163—Liquid or semi-liquid tea extract preparations, e.g. gels, liquid extracts in solid capsules
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23F—COFFEE; TEA; THEIR SUBSTITUTES; MANUFACTURE, PREPARATION, OR INFUSION THEREOF
- A23F3/00—Tea; Tea substitutes; Preparations thereof
- A23F3/40—Tea flavour; Tea oil; Flavouring of tea or tea extract
- A23F3/405—Flavouring with flavours other than natural tea flavour or tea oil
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
- A23L2/56—Flavouring or bittering agents
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
- A23L2/60—Sweeteners
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L27/00—Spices; Flavouring agents or condiments; Artificial sweetening agents; Table salts; Dietetic salt substitutes; Preparation or treatment thereof
- A23L27/20—Synthetic spices, flavouring agents or condiments
- A23L27/202—Aliphatic compounds
- A23L27/2024—Aliphatic compounds having oxygen as the only hetero atom
- A23L27/2028—Carboxy compounds
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L27/00—Spices; Flavouring agents or condiments; Artificial sweetening agents; Table salts; Dietetic salt substitutes; Preparation or treatment thereof
- A23L27/30—Artificial sweetening agents
- A23L27/33—Artificial sweetening agents containing sugars or derivatives
- A23L27/36—Terpene glycosides
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L27/00—Spices; Flavouring agents or condiments; Artificial sweetening agents; Table salts; Dietetic salt substitutes; Preparation or treatment thereof
- A23L27/40—Table salts; Dietetic salt substitutes
- A23L27/45—Salt substitutes completely devoid of sodium chloride
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23F—COFFEE; TEA; THEIR SUBSTITUTES; MANUFACTURE, PREPARATION, OR INFUSION THEREOF
- A23F3/00—Tea; Tea substitutes; Preparations thereof
- A23F3/16—Tea extraction; Tea extracts; Treating tea extract; Making instant tea
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
Definitions
- the present invention relates to a tea beverage in which the taste quality exhibited by sugar and a sweetener is improved, and a method for producing the tea beverage.
- the taste receptors that receive tastes are called taste buds, and are present in a wide variety of fungiform papillae centered around the tip of the tongue, circumvallate papillae and foliate papillae that exist in a limited area behind the tongue.
- the taste bud is a cell aggregate composed of cells called elongated taste cells and basal cells. Taste cells emit fine hairs toward the lingual surface, and at the bottom of the cell form taste nerve fibers and synapses that invade taste buds, and the taste we normally feel is sent to the brain via the taste nerve and recognized as taste information.
- T1R2 and T1R3 are known as sweet taste receptors. It has been reported that T1R2 and T1R3 form a heterodimer (Non-Patent Documents 1 to 3).
- Non-Patent Document 4 As an example of the contrasting effect, which is one of the interaction of tastes, it has long been known that sweetness increases when salt is added to soup powder. There is an example that reports the interaction between saltiness and sweetness focusing on this phenomenon, but the interaction between sweetness and saltiness has a somewhat strong sweetness (15% solution) and a relatively high salt concentration (0.1-0.2%). It has been concluded that it is necessary (Non-Patent Document 4).
- the present inventors have added not only natural sugar but also a specific high-intensity sweetener, and by adding low-concentration sodium and low-concentration potassium and/or calcium, the natural sugar and specific high-intensity sweetener For the first time, it succeeded in increasing the sweetness and improving the taste quality based on the combination of ingredients. It was unexpected that the effect of improving the taste was synergistically improved by adding a specific amount of sodium and/or a specific amount of potassium and/or calcium.
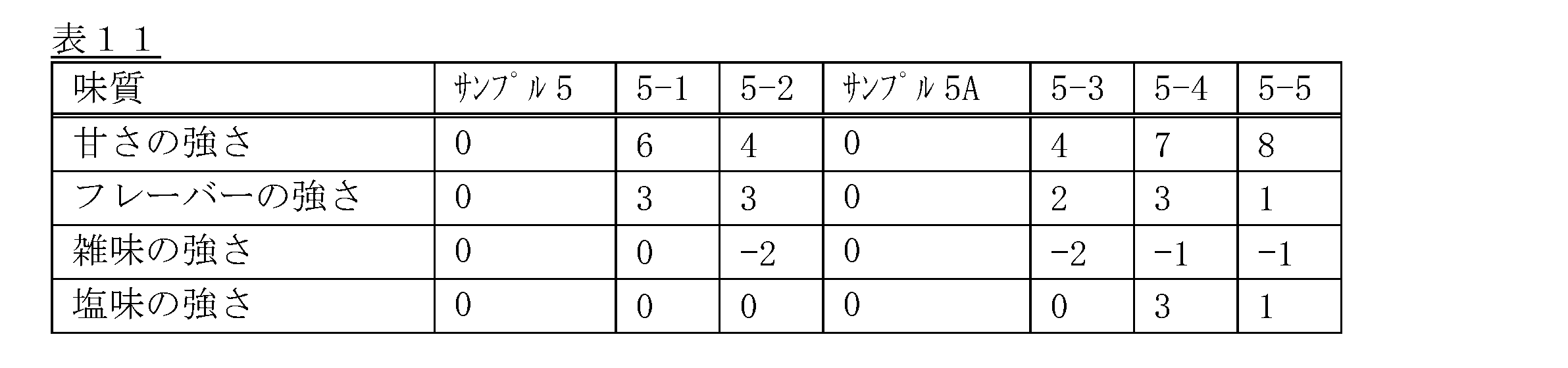
- the present invention is as follows. [1] (A) An amount of natural sugar equivalent to sweetness intensity X1, (B) a sweetener having a high sweetness of an amount equivalent to sweetness intensity X2, (C) less than 50 mg/100 ml sodium, and (d) 0.1-52 mg/100 ml potassium and/or 0.1-52 mg/100 ml calcium,
- the high-potency sweetener comprises at least one high-potency sweetener b1 selected from the group consisting of rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, rebaudioside E, Rakan fruit extract, mogroside V and thaumatin, A tea beverage with 0.1 ⁇ (X1+X2) ⁇ 20.
- the tea beverage according to [1] which has a sodium content of 7 mg/100 ml or more and less than 40 mg/100 ml.
- the tea beverage according to any one of [1] to [5] which contains 200 to 600 ppm of polyphenol.
- the natural sugar is at least one selected from the group consisting of glucose, sucrose, fructose, maltose, oligosaccharides, isomerized sugar, lactose, psicose, allose, tagatose and combinations thereof, [1] to [7]
- the high-intensity sweetener b1 is at least one selected from the group consisting of rebaudioside M, rebaudioside D, Rakan fruit extract, mogroside V, and combinations thereof. [1] to [8] The described tea beverage.
- the sodium is sodium chloride, sodium hydroxide, sodium malate, sodium sulfate, sodium citrate, sodium phosphate, sodium carbonate, sodium disulfide, sodium bicarbonate, sodium alginate, sodium alginate, sodium glucoheptanate, glucone.
- [1] to [9] which is at least one selected from the group consisting of sodium acidate, sodium glutamate, sodium tartrate, sodium aspartate, sodium lactate, sodium caseinate, sodium ascorbate and mixtures thereof.
- the potassium is potassium alginate, potassium chloride, potassium citrate, potassium gluconate, potassium L-glutamate, potassium bromate, DL-potassium hydrogen tartrate, potassium L-tartrate, potassium nitrate, potassium hydroxide, potassium sorbate, carbonic acid.
- the calcium is L-ascorbate, calcium alginate, calcium disodium ethylenediaminetetraacetate, calcium chloride, carboxymethyl cellulose calcium, calcium citrate, calcium glycerophosphate, calcium gluconate, calcium L-glutamate, calcium silicate, calcium acetate, Calcium oxide, calcium hydroxide, calcium stearate, calcium stearoyl lactate, calcium sorbate, calcium carbonate, calcium lactate, calcium pantothenate, calcium dihydrogen pyrophosphate, calcium ferrocyanide, calcium propionate, 5'-ribonucleotide calcium, [1] to [11], which is at least one selected from the group consisting of calcium sulfate, tricalcium phosphate, calcium monohydrogen phosphate, calcium dihydrogen phosphate and mixtures thereof.
- Tea drink [13] Contains 17 mg/100 ml or more and less than 40 mg/100 ml of sodium, contains 10 to 21 mg/100 ml of potassium and/or 10 to 22 mg/100 ml of calcium, has an energy of 50 Kcal/100 ml or less, and has X1+X2 of 6 or more, The tea beverage according to any one of [1] to [12]. [14] The tea beverage according to any one of [1] to [13], which is packaged in a container.
- the high-potency sweetener comprises at least one high-potency sweetener b1 selected from the group consisting of rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, rebaudioside E, Rakan fruit extract, mogroside V and thaumatin, A method for producing a tea beverage, wherein 0.1 ⁇ (X1+X2) ⁇ 20.
- the natural sugar is at least one selected from the group consisting of glucose, sucrose, fructose, maltose, oligosaccharides, isomerized sugar, lactose, psicose, allose, tagatose and combinations thereof, [15]-[21] The method according to any one of 1.
- the high-intensity sweetener b1 is at least one selected from the group consisting of rebaudioside M, rebaudioside D, Rakan fruit extract, mogroside V, and combinations thereof. [15] to [22] The method described.
- the sodium is sodium chloride, sodium hydroxide, sodium malate, sodium sulfate, sodium citrate, sodium phosphate, sodium carbonate, sodium disulfide, sodium bicarbonate, sodium alginate, sodium alginate, sodium glucoheptanate, glucone.
- [15] to [23] which is at least one selected from the group consisting of sodium acidate, sodium glutamate, sodium tartrate, sodium aspartate, sodium lactate, sodium caseinate, sodium ascorbate and mixtures thereof. The method described in.
- the potassium is potassium alginate, potassium chloride, potassium citrate, potassium gluconate, potassium L-glutamate, potassium bromate, DL-potassium hydrogen tartrate, potassium L-tartrate, potassium nitrate, potassium hydroxide, potassium sorbate, carbonic acid.
- Potassium, potassium lactate, potassium norbixin, potassium pyrosulfite, tetrapotassium pyrophosphate, potassium ferrocyanide, potassium polyphosphate, potassium metaphosphate, potassium aluminum sulfate, potassium sulfate, tripotassium phosphate, dipotassium hydrogenphosphate, phosphoric acid The method according to any one of [15] to [24], which is at least one selected from the group consisting of potassium dihydrogen and mixtures thereof.
- the calcium is L-ascorbate, calcium alginate, calcium disodium ethylenediaminetetraacetate, calcium chloride, carboxymethyl cellulose calcium, calcium citrate, calcium glycerophosphate, calcium gluconate, calcium L-glutamate, calcium silicate, calcium acetate, Calcium oxide, calcium hydroxide, calcium stearate, calcium stearoyl lactate, calcium sorbate, calcium carbonate, calcium lactate, calcium pantothenate, calcium dihydrogen pyrophosphate, calcium ferrocyanide, calcium propionate, 5'-ribonucleotide calcium, [15] to [25], which is at least one selected from the group consisting of calcium sulfate, tricalcium phosphate, calcium monohydrogen phosphate, calcium dihydrogen phosphate, and mixtures thereof.
- the tea beverage contains 17 mg/100 ml or more and less than 40 mg/100 ml of sodium, 10 to 21 mg/100 ml of potassium and/or 10 to 22 mg/100 ml of calcium, energy of 50 Kcal/100 ml or less, and X1+X2 is The method according to any one of [15] to [26], which is 6 or more. [28] The method according to any one of [15] to [27], which uses Camellia sinensis tea leaves.
- a sweetener having a high sweetness of an amount equivalent to sweetness intensity X2 A method for enhancing sweetness of a tea beverage, which comprises (c) less than 50 mg/100 ml of sodium, and (d) 0.1 to 52 mg/100 ml of potassium and/or 0.1 to 52 mg/100 ml of calcium.
- the method of the present invention provides a method of increasing the sweetness of a tea beverage and exhibiting a good taste quality, rather than the simple sweetness obtained by increasing the amounts of natural sugar and high-potency sweetener used.
- the method of the present invention provides a tea beverage exhibiting a good taste in which the sweetness is increased by means other than controlling the amounts of sugar and sweetener used.
- the present invention will be described in detail.
- the following embodiments are examples for explaining the present invention and are not intended to limit the present invention only to these embodiments.
- the present invention can be implemented in various forms without departing from the gist thereof.
- All the documents cited in this specification, as well as the publications, patent publications and other patent documents, are incorporated herein by reference.
- the description “the content of the component A is X mg/100 ml” means “the content of the component A is X mg per 100 ml of the beverage”.
- the description “the content of the component B is Yppm” means “the content of the component B is Yppm with respect to the total amount (100% by mass) of the beverage”.
- tea beverage of the present invention provides the following tea beverage (hereinafter referred to as "tea beverage of the present invention") as a first aspect.
- A An amount of natural sugar equivalent to sweetness intensity X1
- B a sweetener having a high sweetness of an amount equivalent to sweetness intensity X2,
- C less than 50 mg/100 ml of sodium
- D contains 0.1 to 52 mg/100 ml of potassium and/or 0.1 to 52 mg/100 ml of calcium
- the high-intensity sweetener is at least one high-intensity sweetener selected from the group consisting of rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, rebaudioside E, Rakan fruit extract, mogroside V and thaumatin. including b1, A tea beverage with 0.1 ⁇ (X1+X2) ⁇ 20.
- the tea beverage of the present invention exhibits sweetness of sweetness intensity X
- the components exhibiting sweetness are (a) a specific amount of a predetermined natural sugar and (b) a specific amount of a predetermined high-potency sweetener.
- the sweetness exhibited by the tea beverage of the present invention should be, in calculation, the sum of the sweetness intensity when the component (a) is added to the tea beverage and the sweetness intensity when the component (b) is added to the tea beverage. is there.
- the sweetness exhibited by (a) the natural sugar and (b) the high intensity sweetener is increased.
- the tea beverage of the present invention exhibits sweetness exceeding the total sweetness intensity.
- the present invention means that, in addition to these components (a) to (d), additional components such as milk, acidulant, flavor and extract can be contained.
- additional components such as milk, acidulant, flavor and extract
- the tea beverage of the present invention does not contain a sweetening component other than the components (a) and (b) as a sweetener.
- the tea beverage according to the preferred embodiment of the present invention exhibits the effect of improving the taste quality in addition to the increase in sweetness.
- the effect of eliciting the flavor (flavor) can be confirmed not only by sensory evaluation but also by analysis of the amount of volatilized aroma components by gas chromatography.
- by increasing the addition amount of K and/or Ca instead of Na the flavor of the tea beverage can be improved while suppressing an increase in salty taste.
- the tea beverage includes a processed product produced by using raw leaves plucked from a tea tree (scientific name: Camellia sinensis) as a raw material.
- tea beverages include barley tea, sweet tea, goya tea, coca tea, rooibos tea, matatabi tea, vine tea, pearl barley tea, yuzu tea, honeybush tea, jinseki tea, dokudami tea, kuma bamboo tea, bamboo tea, herbs.
- the tea beverage is a processed product produced from raw leaves plucked from a tea tree (scientific name: Camellia sinensis).
- the raw leaves that can be used in the present invention are not limited in their variety, production area, cultivation method, tea season, etc. as long as they are tea trees.
- tea tree varieties include Yabukita, Yutaka Midori, Oku Midori, Kaori Sayama, Kanaya Midori, Sae Midori, and Asatsuyu.
- production areas include Shizuoka, Kagoshima, Mie, Kumamoto, Fukuoka, Kyoto, Miyazaki, Saitama, and the like.
- cultivation methods include open field, cover and gyokuro.
- tea season include the first tea, the second tea, the third tea, the fourth tea, the winter-spring-autumn tea, and the cutting number.
- Tea beverages produced using tea tree leaves are generally finished by performing operations such as heating freshly picked leaves with steam and drying to obtain rough tea, firing rough tea, and selecting.
- the above steps are merely examples, and the present invention is not limited to this.
- the order of steps may be changed, another step may be added, or some steps may be omitted.
- Examples of the steps that can be added include a step of grinding finished tea with a mortar or the like when the tea beverage is a matcha beverage.
- oolong tea beverages by using semi-fermented tea leaves obtained by semi-fermenting raw leaves black tea beverages by using fermented tea leaves, using tea leaves fermented green tea is a non-fermented tea by microorganisms such as mold.
- a black tea beverage such as Pu'er tea can be produced.
- varieties used for manufacturing these general ones can be used.
- Tea leaves derived from tea tree leaves and tea leaves for outside tea may be mixed and used.
- the produced green tea, oolong tea, black tea, black tea, etc. can be used alone as a tea beverage, but can also be mixed tea beverages by appropriately mixing each in a suitable ratio. Furthermore, a tea beverage produced by using these tea leaf extract liquids may be added with an extract liquid such as grains and herbs to prepare a tea beverage.
- the tea beverage of the present invention preferably contains polyphenol.
- Polyphenols include polyphenols derived from raw materials such as tea leaves, tea leaves, cereals, and herbs, or polyphenols added as food additives as needed. Examples thereof include anthocyanin, resveratrol, isoflavone, lignan, hesperidin, curcumin, catechin, tannin, proanthocyanin, rutin, chlorogenic acid, ellagic acid, coumarin, procyanidin and the like.
- the polyphenol content is preferably 200 to 600 ppm, particularly preferably 300 to 500 ppm, based on the total amount (100% by mass) of the tea beverage.
- the polyphenol content can be measured by any generally known method.
- the content of catechin in polyphenol is preferably 200 to 600 ppm, particularly preferably 300 to 500 ppm, based on the total amount (100% by mass) of the tea beverage.
- the "natural sugar” is represented by the general formula of C m (H 2 O) n (where m and n represent independent natural numbers), and includes a human-digestible D-form carbohydrate, Examples thereof include glucose, sucrose, fructose, maltose, oligosaccharides (eg fructooligosaccharides, maltooligosaccharides, isomaltooligosaccharides, galactooligosaccharides, etc.), isomerized sugars, lactose, psicose, allose, tagatose and combinations thereof.
- oligosaccharides eg fructooligosaccharides, maltooligosaccharides, isomaltooligosaccharides, galactooligosaccharides, etc.
- isomerized sugars lactose, psicose, allose, tagatose and combinations thereof.
- Examples of combinations of glucose, sucrose, fructose, maltose, oligosaccharides, isomerized sugars, and lactose include the following. Specifically, sucrose and isomerized sugar, sucrose and glucose, sucrose and fructose, sucrose and maltose, sucrose and oligosaccharide, sucrose and lactose, isomerized sugar and oligosaccharide, sucrose and psicose, sucrose and isomerized sugar and glucose.
- Sucrose and isomerized sugar and fructose sucrose and isomerized sugar and maltose, sucrose and isomerized sugar and oligosaccharide, sucrose and isomerized sugar and glucose and oligosaccharide, sucrose and glucose and fructose and oligosaccharide, glucose and sucrose and Examples include a combination of fructose, maltose, oligosaccharide, isomerized sugar, lactose, tagatose, and the like.
- sugar derived from milk is also included in natural sugars.
- the present invention keeps the concentration of natural sugar low, has low energy (that is, low calorie), but has a strong sweetness when ingested due to the combination of natural sugar, a high-potency sweetener, sodium and potassium and/or calcium. felt. Therefore, the amount of sweetness intensity X1 of natural sugar is preferably an amount that provides energy of 50 Kcal/100 ml or less.
- the energy of the tea beverage of the present invention is 0 to 50 Kcal/100 ml, 0 to 45 Kcal/100 ml, 0 to 40 Kcal/100 ml, 0 to 35 Kcal/100 ml, 0 to 30 Kcal/100 ml, 0 to 25 Kcal/100 ml, 0 to 22Kcal/100ml, 0-20Kcal/100ml, 0-15Kcal/100ml, 0-10Kcal/100ml, 0-5Kcal/100ml, 5-50Kcal/100ml, 5-45Kcal/100ml, 5-40Kcal/100ml, 5-35Kcal/ 100 ml, 5-30 Kcal/100 ml, 5-25 Kcal/100 ml, 5-20 Kcal/100 ml, 5-15 Kcal/100 ml, 5-10 Kcal/100 ml, 10-50 Kcal/100 ml, 10-45 Kcal/100 ml, 10-40 Kcal/100 ml, 10-35K
- the energy of the tea beverage of the present invention is also 0-32Kcal/100ml, 0-24Kcal/100ml, 0-8Kcal/100ml, 0-4Kcal/100ml, 4-32Kcal/100ml, 4-24Kcal/100ml, depending on the embodiment. It may be 4-8 Kcal/100 ml, 8-32 Kcal/100 ml, 8-24 Kcal/100 ml, 24-32 Kcal/100 ml. When a component with high calories such as milk is contained, the total calorie of natural sugar and components such as milk is preferably 50 Kcal/100 ml or less.
- X1 of "sweetness intensity X1" is 0 to 0.5, 0 to 1.0, 0 to 1.5, 0 to 2.0, 0 to 2.5, 0 to 3.0, 0 to 3.5, 0 to 4.0, 0 to 4.5, 0 to 5.0, 0-5.5, 0-6.0, 0-6.5, 0-7.0, 0-7.5, 0-8.0, 0-8.25, 0-8.5, 0-8.75, 0-9.0, 0-9.25, 0-9.5, 0- 9.75, 0 to 10.0, 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 to 5.5, 0.05 to 6.0, 0.05 to 6.5, 0.05 to 7.0, 0.05 to 7.5, 0.05 to 8.0, 0.05 to 8.25, 0.05 to 8.5, 0.05 to 8.75, 0.05 to 9.0,
- X1 is also 0 to 10.5, 0 to 11.0, 0 to 11.5, 0 to 12.0, 0 to 12.5, 0 to 13.0, 0 to 13.5, 0 to 14.0, 0 to 14.5, 0 to 15.0, 0.05 to 10.5, 0.05 to 11.0, 0.05 ⁇ 11.5, 0.05 ⁇ 12.0, 0.05 ⁇ 12.5, 0.05 ⁇ 13.0, 0.05 ⁇ 13.5, 0.05 ⁇ 14.0, 0.05 ⁇ 14.5, 0.05 ⁇ 15.0, 0.1 ⁇ 10.5, 0.1 ⁇ 11.0, 0.1 ⁇ 11.5, 0.1 ⁇ 12.0, 0.1 ⁇ 12.5, 0.1 ⁇ 13.0, 0.1 ⁇ 13.5, 0.1 ⁇ 14.0, 0.1 ⁇ 14.5, 0.1 ⁇ 15.0, 0.5 ⁇ 10.5, 0.5 ⁇ 11.0, 0.5 ⁇ 11.5, 0.5 ⁇ 12.0, 0.5 ⁇ 12.5, 0.5 ⁇ 13.0, 0.5 ⁇ 13.5, 0.5 ⁇ 14.0, 0.5 ⁇ 14.5, 0.5 ⁇
- the amount corresponding to the sweetness intensity X1 of the natural sugar refers to the amount (concentration) at which the sweetness of the sweetness intensity X1 is exhibited under the condition that the natural sugar is dissolved in 20° C. water having the same volume as the tea beverage of the present invention.
- the sweetness intensity means the sweetness of a substance.
- the sweetness degree of glucose is 0.6 to 0.7 (center value 0.65).
- the sugar having a wide range of sweetness uses the central value thereof unless otherwise specified.
- the “high-potency sweetener” means a compound having a stronger sweetness than sucrose, and may be a naturally derived compound, a synthetic compound, or a combination of naturally derived compounds and synthetic compounds. High-intensity sweetener in the same amount as sucrose, 5 times or more, 10 times or more, 50 times or more, 100 times or more, 500 times or more, 1000 times or more, 5000 times or more, 10000 times or more, 50000 times more than sucrose. As a result, it has a sweetness of 100,000 times or more.
- the presence of the natural sugar and the high-intensity sweetener in the tea beverage of the present invention in terms of weight ratio indicates “natural sugar: high-intensity sweetener”. Is 5:1 to 10:1, 50:1 to 100:1, 50:1 to 200:1, 500:1 to 1000:1, 5000:1 to 10000:1, 50000:1 to 100000:1 ..
- the high intensity sweetener at least one high intensity sweetener b1 selected from the group consisting of rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, rebaudioside E, Rakan fruit extract, mogroside V and thaumatin.
- the content of the high intensity sweetener b1 with respect to the total amount (100% by weight) of the high intensity sweetener which is the component (b) is preferably 50% by mass or more, more preferably 60% by mass or more, and further preferably 70% by mass.
- the content is preferably at least mass%, more preferably at least 80 mass%, particularly preferably at least 90 mass%.
- the high-intensity sweetener which is the component (b), may consist essentially of the high-intensity sweetener b1.
- “consisting essentially of the high-intensity sweetener b1” means that it is unavoidable in the process of preparing the high-intensity sweetener b1 (such as purification and biosynthesis of stevia extract or lo-kan fruit extract). It means that impurities such as other steviol glycosides and mogrosides contained therein may be contained.
- Rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, rebaudioside E may be directly extracted from stevia, or may be obtained by adding glucose to a compound having another structure contained in the stevia extract.
- the Rakan fruit extract as a sweetener is an extract of Rakan fruit containing a sweet substance derived from Rakan fruit, and is licensed and marketed as a food additive in each country including Japan.
- sweet substances derived from Lokhan fruit include mogroside V, mogroside IV, 11-oxo-mogroside V, and siamenoside I.
- Mogroside V is one of the main mogrol glycosides contained in Rakan fruit, and it has been reported that it has high-quality sweetness characteristics similar to those of sucrose, as compared with rebaudioside A. In addition, the sweetness of mogroside V is approximately 300 times that of sucrose (MurataY et al., Nippon Shokuhin Kagaku Kogaku Kaishi Vol. 53, No.10, 527-533 (2006) ).
- Mogroside V can be obtained by purifying from an extract of Rakan fruit (for example, an alcohol extract of Rakan fruit) by chromatography or the like. Alternatively, mogroside V may be obtained by adding glucose to a compound having another structure contained in the Rakan fruit extract.
- the Rakan fruit extract preferably contains mogroside V, and the ratio thereof is not limited, and is 10% by weight or more, 15% by weight or more, 20% by weight or more, 25% by weight or more, based on the dry weight of the entire Rakan fruit extract. % By weight, 35% by weight, 40% by weight, 45% by weight, 50% by weight or more, 55% by weight or more, 60% by weight or more, 65% by weight or more, 70% by weight or more, 75% by weight or more You can The content of mogroside V can be determined by a known method such as liquid chromatography.
- the Rakan fruit extract is obtained by mixing the fruits of Rakan fruit (Siraitia grosvenorii) with an appropriate solvent (for example, an aqueous solvent such as water, an alcohol solvent such as ethanol or methanol, a water-containing ethanol or a mixed solvent of an alcohol solvent such as water-containing methanol). Etc.), and then optionally subjected to treatments such as degreasing, purification, concentration and drying.
- an appropriate solvent for example, an aqueous solvent such as water, an alcohol solvent such as ethanol or methanol, a water-containing ethanol or a mixed solvent of an alcohol solvent such as water-containing methanol. Etc.
- Mogroside V may be highly pure, for example, having a purity of 80% or more, 85% or more, 90% or more, 91% or more, 92% or more, 93% or more, 94% or more, 95% or more, 96. % Or more, 97% or more, or 98% or more, or the like.
- the higher the purity of the mogroside V obtained by purifying the Rakan fruit extract the less the content of the Rakan fruit extract components other than the Mogroside V.
- Thaumatin is a protein-based sweetener extracted from plants.
- other high-intensity sweetener b2 may be used in combination with the high-intensity sweetener b1 described above.
- specific examples of the other high-intensity sweetener b2 include peptide-based sweeteners such as aspartame, neotame, and aritame; sucrose derivatives such as sucralose; synthetic sweeteners such as acesulfame K, saccharin, advantame, cyclamate, and zultin; Examples include protein-based sweeteners extracted from plants other than thaumatin such as monerin, curculin, and brazein; high-intensity sweeteners other than protein-based sweeteners extracted from plants; taumarin; neohesperidin dihydrochalcone.
- the sucrose derivative is obtained by substituting the OH group or H group of sucrose with another substituent, and examples thereof include a halogen derivative (sucralose) of sucrose, an oxathiazinone dioxide derivative, and a sugar. Examples thereof include alcohol, aldonic acid, and uronic acid.
- high-intensity sweeteners other than protein-based sweeteners extracted from plants include sweeteners present in stevia extracts other than rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, and rebaudioside E; other than mogroside V. And sweeteners present in the extract of licorice; glycosides thereof; and the like. Moreover, monatin, glycyrrhizin, etc. are also mentioned.
- Sweeteners and glycosides present in stevia extracts other than rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, and rebaudioside E include steviol, stevioside, rebaudioside A, rebaudioside B, rebaudioside C and the like.
- Examples of sweeteners and glycosides thereof present in the Rakan fruit extract other than mogroside V include mogroside IV.
- a licorice extract is a glycyrrhizic acid-based ingredient obtained from the roots or rhizomes of laurel liquorice, butterfly licorice or licorice.
- Examples of licorice extract include licorice extract, glycyrrhizin and licorice extract.
- the high-intensity sweetener b1 at least one selected from the group consisting of rebaudioside M, rebaudioside D, Rakan fruit extract, mogroside V and combinations thereof is suitable. Particularly, as the high-intensity sweetener b1, rebaudioside D, rebaudioside M, or a mixture of rebaudioside D and rebaudioside M is suitable.
- Rebaudioside D or Rebaudioside M has few negative aromas such as astringency and metallic taste as seen in Rebaudioside A, has characteristics such as high-quality sweetness, and is expected to be used in food and beverage fields (Japan. Chemical Society of Japan (5), (1981) 726-735, "Sweet Steroid Leaf Sweet Diterpene Glycosides-Synthesis of Rebaudioside-A, -D, -E and Related Glycosides and Correlation between Sweetness and Chemical Structure-” , Kasai, Kaneda, Tanaka, Yamazaki, Sakamoto, Morimoto, Okada, Kitahata, Furukawa).
- rebaudioside D or rebaudioside M when used alone, it is excellent in that it has less unpleasant taste than rebaudioside A and has a sweetness similar to sucrose.
- Rebaudioside D or rebaudioside M has about 200 times the sweetness of sucrose.
- Examples of the combination of the high intensity sweetener b1 and the combination of the high intensity sweetener b1 and another high intensity sweetener b2 include the following. Specifically, rebaudioside D and rebaudioside M, rebaudioside D and rebaudioside A, rebaudioside M and rebaudioside A, rebaudioside M and mogroside V, rebaudioside D and mogroside V, rebaudioside D and advantame, rebaudioside D and acesulfameside D, rebaudioside D and acesulfame D, Sucralose, rebaudioside M and rebaudioside D and rebaudioside A, rebaudioside M and rebaudioside D and mogroside V, rebaudioside M and rebaudioside D and adventame, rebaudioside M and rebaudioside D and acesulfame K, rebaudioside M and rebaudioside
- rebaudioside D and mogroside V rebaudioside D and rahan fruit extract, rebaudioside M and rahan fruit extract, rebaudioside M and rebaudioside D and rahan fruit extract, rebaudioside A and rebaudioside M and rebaudioside D and rahan fruit extract, rebaudioside A and rebaudioside M and rebaudioside D and rakaneo fruit A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudioside A and rebaudio
- X2 of "sweetness intensity X2" is 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 ⁇ 5.5, 0.5 ⁇ 1.0, 0.5 ⁇ 1.5, 0.5 ⁇ 2.0, 0.5 ⁇ 2.5, 0.5 ⁇ 3.0, 0.5 ⁇ 3.5, 0.5 ⁇ 4.0, 0.5 ⁇ 4.5, 0.5 ⁇ 5.0, 0.5 ⁇ 5.5, 1.0 ⁇ 1.5, 1.0 ⁇ 2.0, 1.0-2.5, 1.0-3.0, 1.0-3.5, 1.0-4.0, 1.0-4.5, 1.0-5.0, 1.0-5.5, 1.5-2.0, 1.5-2.5, 1.5-3.0, 1.5-3.5, 1.5-4.0, 1.5-4.5, 1.5-5.0, 1.5-5.5, 2.0-2.5, 2.0-3.0, 2.0-3.5, 2.0-4.0, 2.0-4.5, 2.0-5.0, 1.0-5.5
- X2 is also 0.05 to 6.0, 0.05 to 6.5, 0.05 to 7.0, 0.05 to 7.5, 0.05 to 8.0, 0.05 to 8.5, 0.05 to 9.0, 0.05 to 9.5, 0.05 to 10.0, 0.05 to 10.5, 0.05 to 11.0, 0.05 to 11.5, 0.05 ⁇ 12.0, 0.05 ⁇ 13.0, 0.05 ⁇ 14.0, 0.05 ⁇ 15.0, 0.05 ⁇ 16.0, 0.05 ⁇ 17.0, 0.05 ⁇ 18.0, 0.5 ⁇ 6.0, 0.5 ⁇ 6.5, 0.5 ⁇ 7.0, 0.5 ⁇ 7.5, 0.5 ⁇ 8.0, 0.5 ⁇ 8.5, 0.5 ⁇ 9.0, 0.5 ⁇ 9.5, 0.5 ⁇ 10.0, 0.5 ⁇ 10.5, 0.5 ⁇ 11.0, 0.5 ⁇ 11.5, 0.5 ⁇ 12.0, 0.5 ⁇ 13.0, 0.5 ⁇ 14.0, 0.5 ⁇ 15.0, 0.5 ⁇ 16.0, 0.5 ⁇ 17.0, 0.5-18.0,
- the amount equivalent to the sweetness intensity X2 of the high-intensity sweetener refers to the amount at which the sweetness intensity X2 is sweetened under the condition that the high-intensity sweetener is dissolved in 20°C of water of the same volume as the tea beverage of the present invention.
- the sweetness intensity here also means the sweetness of the substance.
- sweetness intensity of sucrose per unit concentration Brix is defined as sweetness 1
- sweetness of rebaudioside D is 200 to 250 (center value 225)
- sweetness of rebaudioside M is 200 to 260 (center value 230).
- Sweetness of rebaudioside N is 230-240 (center value 235), sweetness of rebaudioside O is 200-250 (center value 225), sweetness of rebaudioside N is 200-250 (center value 225), sweetness of rebaudioside E
- the degree of sweetness is 70-80 (center value 75), the sweetness of Rakan fruit extract is 110-150 (center value 130), the sweetness of mogroside V is 240-300 (center value 270), the sweetness of thaumatin is 2,000 ..
- the value obtained by multiplying these sweetnesses by the concentration of the high-potency sweetener in the tea beverage (w/v% (can be equated with w/w% in the case of beverages)) is the sweetness of the high-sweetness sweetener. It becomes strength.
- the central value of the high-sweetness sweetener having a wide range of sweetness is used unless otherwise specified.
- the content of the high intensity sweetener may be P2ppm, where P2ppm is the amount corresponding to the sweetness intensity X2.
- P2 is 20-550, 25-550, 30-550, 35-550, 40-550, 45-550, 50-550, 55-550, 20-540, 25-540, 30-540, 35 ⁇ 540, 40-540, 45-540, 50-540, 55-540, 20-530, 25-530, 30-530, 35-530, 40-530, 45-530, 50-530, 55-530 , 20-520, 25-520, 30-520, 35-520, 40-520, 45-520, 50-520, 55-520, 20-510, 25-510, 30-510, 35-510, 40 ⁇ 510, 45-510, 50-510, 55-510, 20-505, 25-505, 30-505, 35-505, 40-505, 45-505, 50-505, 55-505, 20-500 , 25-500, 30-500, 35-500, 40-500, 45-500, 50-500, 55-500, 20-
- P2 is also 1-1500, 1-1200, 5-1200, 1-1000, 5-1000, 10-1000, 1-900, 5-900, 10-900, 15-900, 20-900, 25- 900, 30-900, 35-900, 40-900, 45-900, 50-900, 55-900, 1-800, 5-800, 10-800, 15-800, 20-800, 25-800, 30-800, 35-800, 40-800, 45-800, 50-800, 55-800, 1-700, 5-700, 10-700, 15-700, 20-700, 25-700, 30- 700, 35-700, 40-700, 45-700, 50-700, 55-700, 1-600, 5-600, 10-600, 15-600, 20-600, 25-600, 30-600, 35-600, 40-600, 45-600, 50-600, 55-600, 1-550, 1-540, 1-530, 1-520, 1-510, 1-505, 1-500, 1- 495, 1-490, 5-550, 5-540, 5-530, 5-520, 5-510, 5-505, 5-500, 5-4
- the present invention comprises (c) less than 50 mg/100 ml of sodium, which means that the content of sodium atoms is less than 50 mg/100 ml.
- the content of sodium is 0.1 mg/100 ml or more and less than 50 mg/100 ml, 0.1 to 45 mg/100 ml, 0.1 to 40 mg/100 ml, 0.1 to 35 mg/100 ml, 0.1 to 30 mg/100 ml, 0.1 to 25 mg/ 100 ml, 0.1-20 mg/100 ml, 0.1-19 mg/100 ml, 0.1-18 mg/100 ml, 0.1-17 mg/100 ml, 0.1-16 mg/100 ml, 0.1-15 mg/100 ml, 0.1-14 mg/100 ml, 0.1-13 mg/100 ml, 0.1-12 mg/100 ml, 0.1-11 mg/100 ml, 0.1-10 mg/100 ml, 1 mg/100 ml or more but less than 50
- the sodium content may be 0.1 to 22 mg/100 ml, 0.1 to 21 mg/100 ml, 1 to 22 mg/100 ml, 1 to 21 mg/100 ml, 4 to 40 mg/100 ml, 4 to 35 mg/100 ml, depending on the embodiment.
- the amount of sodium added to the tea beverage is 0.1 mg/100 ml or more and less than 50 mg/100 ml, 0.1 to 45 mg/100 ml, 0.1 to 40 mg/100 ml, 0.1 to 35 mg/100 ml, 0.1 to 30 mg/100 ml, 0.1 ⁇ 25mg/100ml, 0.1-20mg/100ml, 0.1-19mg/100ml, 0.1-18mg/100ml, 0.1-17mg/100ml, 0.1-16mg/100ml, 0.1-15mg/100ml, 0.1-14mg/100ml, 0.1-13mg /100 ml, 0.1-12 mg/100 ml, 0.1-11 mg/100 ml, 0.1-10 mg/100 ml, 1 mg/100 ml or more and less than 50 mg/100 ml, 1-45 mg/100 ml, 1-40 mg/100 ml, 1-35 mg/100 ml, 1- 30mg/100ml
- Sodium is not particularly limited in its form as long as it is contained in the tea beverage of the present invention in an ingestible state, for example, sodium chloride, sodium hydroxide, sodium malate, sodium sulfate, sodium citrate, Sodium phosphate, sodium carbonate, sodium disulfide, sodium bicarbonate, sodium alginate, sodium alginate, sodium glucoheptanate, sodium gluconate, sodium glutamate, sodium tartrate, sodium aspartate, sodium lactate, sodium caseinate, sodium ascorbate And at least one form selected from the group consisting of a mixture thereof.
- sodium may be contained in tea leaves, and in that case, sodium obtained when tea is extracted from tea leaves is also included in the sodium contained in the tea beverage of the present invention.
- sodium derived from a sodium component used as a preservative for example, sodium benzoate, sodium sulfite, sodium hyposulfite, sodium dehydroacetate, sodium pyrosulfite, sodium propionate, etc.
- a sodium component used as a preservative for example, sodium benzoate, sodium sulfite, sodium hyposulfite, sodium dehydroacetate, sodium pyrosulfite, sodium propionate, etc.
- the content of sodium in a beverage can be measured by an atomic absorption method.
- the value calculated from the blending amount may be used.
- the present invention may include (c) 0.1 to 52 mg/100 ml of potassium, which means that the content of potassium atoms is 0.1 to 52 mg/100 ml.
- the content of potassium ranges from 0.1 to 50 mg/100 ml, 0.1 to 45 mg/100 ml, 0.1 to 40 mg/100 ml, 0.1 to 35 mg/100 ml, 0.1 to 32 mg/100 ml, 0.1 to 30 mg/100 ml, 0.1 to 25 mg/100 ml, 0.1-21 mg/100 ml, 0.1-20 mg/100 ml, 0.1-15 mg/100 ml, 0.1-10 mg/100 ml, 0.1-5 mg/100 ml, 0.5-52 mg/100 ml, 0.5-50 mg/100 ml, 0.5-45 mg/ 100 ml, 0.5-40 mg/100 ml, 0.5-35 mg/100 ml, 0.5-30 mg/100 ml, 0.5-25 mg
- the amount of potassium added to tea beverages is 0.1-52mg/100ml, 0.1-50mg/100ml, 0.1-45mg/100ml, 0.1-40mg/100ml, 0.1-35mg/100ml, 0.1-30mg/100ml , 0.1-25mg/100ml, 0.1-20mg/100ml, 0.1-19mg/100ml, 0.1-18mg/100ml, 0.1-17mg/100ml, 0.1-16mg/100ml, 0.1-15mg/100ml, 0.1-14mg/100ml, 0.1 ⁇ 13mg/100ml, 0.1-12mg/100ml, 0.1-11mg/100ml, 0.1-10mg/100ml, 1-50mg/100ml, 1-45mg/100ml, 1-40mg/100ml, 1-35mg/100ml, 1-30mg /100ml, 1-25mg/100ml, 1-20mg
- the form of potassium is not particularly limited as long as it is contained in the tea beverage of the present invention in an ingestible state.
- It may be in at least one form selected from the group consisting of potassium polyphosphate, potassium metaphosphate, potassium aluminum sulfate, potassium sulfate, tripotassium phosphate, dipotassium hydrogen phosphate, potassium dihydrogen phosphate and mixtures thereof.
- potassium may be contained in tea leaves, and in that case, potassium obtained when extracting components from tea leaves is also included in potassium contained in the tea beverage of the present invention.
- the tea beverage contains milk, milk, dairy products, and other milk components, potassium contained in the milk components is also included.
- the content of potassium in a beverage can be measured by an atomic absorption method.
- a value calculated from the blending amount may be used.
- the present invention may comprise (c) 0.1-52 mg/100 ml of calcium, which means that the content of calcium atoms is 0.1-52 mg/100 ml.
- the content of calcium is 0.1-50 mg/100 ml, 0.1-45 mg/100 ml, 0.1-40 mg/100 ml, 0.1-35 mg/100 ml, 0.1-32 mg/100 ml, 0.1-30 mg/100 ml, 0.1- 25 mg/100 ml, 0.1-22 mg/100 ml, 0.1-20 mg/100 ml, 0.1-19 mg/100 ml, 0.1-18 mg/100 ml, 0.1-17 mg/100 ml, 0.1-16 mg/100 ml, 0.1-15 mg/100 ml, 0.1-14 mg/ 100 ml, 0.1 to 13 mg/100 ml, 0.1 to 12 mg/100 ml, 0.1 to 11 mg/100 ml, 0.1 to 10 mg/100 ml
- the amount of calcium added to tea beverages is 0.1-52mg/100ml, 0.1-50mg/100ml, 0.1-45mg/100ml, 0.1-40mg/100ml, 0.1-35mg/100ml, 0.1-30mg/100ml , 0.1-25mg/100ml, 0.1-20mg/100ml, 0.1-19mg/100ml, 0.1-18mg/100ml, 0.1-17mg/100ml, 0.1-16mg/100ml, 0.1-15mg/100ml, 0.1-14mg/100ml, 0.1 ⁇ 13mg/100ml, 0.1-12mg/100ml, 0.1-11mg/100ml, 0.1-10mg/100ml, 1-52mg/100ml, 1-50mg/100ml, 1-45mg/100ml, 1-40mg/100ml, 1-35mg /100ml, 1-30mg/100ml, 1-25mg
- the form of calcium is not particularly limited as long as it is contained in the tea beverage of the present invention in an ingestible state, and examples thereof include calcium L-ascorbate, calcium alginate, calcium disodium ethylenediaminetetraacetate, and calcium chloride.
- It may be at least one form selected from the group consisting of: Further, although calcium may be contained in tea leaves and water (particularly hard water), in that case, calcium obtained when extracting components from tea leaves is also included in calcium contained in the tea beverage of the present invention. .. In addition, when the tea beverage contains milk, milk, milk products, and other milk components, calcium contained in the milk components is also included.
- the content of calcium in the beverage can be measured by ICP emission spectrometry. If the blending amount of the calcium-containing compound blended in the beverage is known, a value calculated from the blending amount may be used. In addition, for example, when an additive containing two or more atoms selected from sodium, potassium, and calcium, such as ethylenediaminetetraacetate calcium disodium, is mixed, the compounding amount is such that the amounts of sodium, calcium, and sodium are as described above. It is prepared so that it may become the range of.
- the tea beverage of the present invention exhibits enhanced sweetness. Whether or not the sweetness of the tea beverage of the present invention is enhanced can be evaluated by panelists trained in sensory. Furthermore, the sweetness intensity X3 of the tea beverage of the present invention is a standard tea beverage that serves as a reference for sweetness, the sweetness intensity 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, , 14, 15 prepared by shaking the sucrose concentration, panelists compare the sweetness of the tea beverage of the present invention and the sweetness of the reference tea beverage, to measure the sweetness of the tea beverage of the present invention You can Note that the standard tea beverages with sweetness intensity of 1, 2, ... 15 should be sucrose content of 1g/100g, 2g/100g, ... 15g/100g compared to tea beverages without added sucrose.
- a reference tea beverage having the closest sweetness to the tea beverage of the present invention is selected, and sucrose is added to the selected reference tea beverage. It is adjusted to exhibit the same sweetness as the tea beverage of the present invention by adding, at that time, from the sucrose content contained in the adjusted reference tea beverage, the sweetness intensity X3 of the tea beverage of the present invention is measured. You can also
- Another method for measuring the sweetness of the tea beverage of the present invention is, for example, the sweetness intensity rating (VAS method) using the Visual Analogue Scale.
- VAS method the sweetness intensity rating
- the literature of Jaw Function Magazine (2014) 20 pp.115-129 (“Construction of a screening test method for taste function in 4 basic tastes” Toyota et al.) can be referred to.
- the evaluator determined that the lower end of the sweetness intensity was “not sweet at all” and the upper end was “I can't think of anything sweeter than this”. Evaluation is performed by expressing the sweetness intensity felt at that time by a position on a straight line using a paper on which a vertical line indicating the intensity is drawn.
- X3 is 4.0-20, 4.0-15, 4.0-12.5, 4.0-10, 4.5-20, 4.5-15, 4.5-12.5, 4.5-10, 5.0-20, 5.0-15, 5.0-12.5, 5.0- 10, 5.5-20, 5.5-15, 5.5-12.5, 5.5-10, 6.0-20, 6.0-15, 6.0-12.5, 6.0-10, 6.5-20, 6.5-15, 6.5-12.5, 6.5-10, 7.0-20, 7.0-15, 7.0-12.5, 7.0-10, 7.5-20, 7.5-15, 7.5-12.5, 7.5-10, 7.5-9, 7.5-8, 8.0-20, 8.0-20, 8.0- 15, 8.0 to 12.5, 8.0 to 10, 8.5 to 20, 8.5 to 15, 8.5 to 12.5, 8.5 to 10, 9.0 to 20, 9.0 to 15, 9.0 to 12.5, 9.0 to 10, 9.5 to 20, 9.5 to 15, It may be 9.5 to 12.5, 9.5 to 10, 10.0 to 20, 10.0 to 15, 10.0 to 12.5, 10.5 to 20, 10.5 to 15, and 10.5 to 12.5.
- X3 is also 4.0-18, 4.0-16, 4.0-15.5, 4.0-14, 4.5-18, 4.5-16, 4.5-15.5, 4.5-14, 5.0-18, 5.0-16, 5.0-15.5, 5.0- 14, 5.5-18, 5.5-16, 5.5-15.5, 5.5-14, 6.0-18, 6.0-16, 6.0-15.5, 6.0-14, 6.5-18, 6.5-16, 6.5-15.5, 6.5-14, 7.0-18, 7.0-16, 7.0-15.5, 7.0-14, 7.5-18, 7.5-16, 7.5-15.5, 7.5-14, 7.5-9, 7.5-8, 8.0-18, 8.0-16, 8.0- 15.5, 8.0-14, 8.5-18, 8.5-16, 8.5-15.5, 8.5-14, 9.0-18, 9.0-16, 9.0-15.5, 9.0-14, 9.5-18, 9.5-16, 9.5-15.5, It may be 9.5 to 14, 10.0 to 18, 10.0 to 16, 10.0 to 15.5, 10.5 to 18, 10.5 to 16, 10.5 to 15.5.
- the tea beverage of the present invention comprises (A) An amount of natural sugar whose sweetness intensity X1 is equivalent to 0.1 to 5, (B) a high intensity sweetener with an amount of sweetness intensity X2 equivalent to 0.1 to 5, (C) 7 mg/100 ml or more and 40 mg/100 ml or less of sodium, and (d) 0.1 to 52 mg/100 ml of potassium and/or 0.1 to 52 mg/100 ml of calcium, A tea beverage having a sweetness intensity X3 of 5.5 to 12.5 due to the components (a) to (d).
- the tea beverage of the present invention comprises (A) An amount of natural sugar having a sweetness intensity X1 of 3 to 5, (B) A high-intensity sweetener whose sweetness intensity X2 is equivalent to 1 to 5, (C) 17 mg/100 ml or more and less than 40 mg/100 ml of sodium, and (d) 0.1 to 32 mg/100 ml of potassium and/or 0.1 to 32 mg/100 ml of calcium, A tea beverage having a sweetness intensity X3 of 5.5 to 12.5 due to the components (a) to (d).
- the tea beverage of the present invention further has energy of 50 Kcal/100 ml or less and X1+X2 of 6.0 or more.
- the sweetness intensity X1 of natural sugar, the sweetness intensity X2 of the high-intensity sweetener, the sodium content, the potassium and/or calcium content, the sweetness intensity X3 of the tea beverage and the energy of the tea beverage are 0.1. Any value may be used as long as ⁇ (X1+X2) ⁇ 20 is satisfied.
- "Sweetness intensity X1" 0.1-0.5, 0.1-1.0, 0.1-1.5, 0.1-2.0, 0.1-2.5, 0.1-3.0, 0.1-3.5, 0.1-4.0, 0.1-4.5, 0.1-5.0, 0.5-1.0, 0.5 ⁇ 1.5, 0.5 ⁇ 2.0, 0.5 ⁇ 2.5, 0.5 ⁇ 3.0, 0.5 ⁇ 3.5, 0.5 ⁇ 4.0, 0.5 ⁇ 4.5, 0.5 ⁇ 5.0, 1.0 ⁇ 1.5, 1.0 ⁇ 2.0, 1.0 ⁇ 2.5, 1.0 ⁇ 3.0, 1.0 ⁇ 3.5, 1.0-4.0, 1.0-4.5, 1.0-5.0, 1.5-2.0, 1.5-2.5, 1.5-3.0, 1.5-3.5, 1.5-4.0, 1.5-4.5, 1.5-5.0, 2.0-2.5, 2.0-3.0, 2.0-3.5, 2.0-4.0, 2.0-4.5, 2.0-5.0, 2.0-5.5, 2.5-3.0, 2.5-3.5, 2.5-4.0, 2.5-4.5, 2.5-5.0, 3.0-3.5, 3.0-4.0, 3.
- "Sweetness intensity X2" 0.1-0.5, 0.1-1.0, 0.1-1.5, 0.1-2.0, 0.1-2.5, 0.1-3.0, 0.1-3.5, 0.1-4.0, 0.1-4.5, 0.1-5.0, 0.5-1.0, 0.5 ⁇ 1.5, 0.5 ⁇ 2.0, 0.5 ⁇ 2.5, 0.5 ⁇ 3.0, 0.5 ⁇ 3.5, 0.5 ⁇ 4.0, 0.5 ⁇ 4.5, 0.5 ⁇ 5.0, 1.0 ⁇ 1.5, 1.0 ⁇ 2.0, 1.0 ⁇ 2.5, 1.0 ⁇ 3.0, 1.0 ⁇ 3.5, 1.0-4.0, 1.0-4.5, 1.0-5.0, 1.5-2.0, 1.5-2.5, 1.5-3.0, 1.5-3.5, 1.5-4.0, 1.5-4.5, 1.5-5.0, 2.0-2.5, 2.0-3.0, 2.0-3.5, 2.0-4.0, 2.0-4.5, 2.0-5.0, 2.5-3.0, 2.5-3.5, 2.5-4.0, 2.5-4.5, 2.5-5.0, 3.0-3.5, 3.0-4.0, 3.0-4.5, 3.
- Sodium content 7-40mg/100ml, 7mg/100ml or more and less than 40mg/100ml, 7-35mg/100ml, 7-30mg/100ml, 7-25mg/100ml, 7-20mg/100ml, 7-19mg/ 100 ml, 7-18 mg/100 ml, 7-17 mg/100 ml, 7-16 mg/100 ml, 7-15 mg/100 ml, 7-14 mg/100 ml, 7-13 mg/100 ml, 7-12 mg/100 ml, 7-11 mg/100 ml, 7-10mg/100ml, 10-40mg/100ml, 10-35mg/100ml, 10-30mg/100ml, 10-25mg/100ml, 10-20mg/100ml, 10-19mg/100ml, 10-18mg/100ml, 10- 17mg/100ml, 10-16mg/100ml, 10-15mg/100ml, 15-40mg
- “Potassium content” 0.1-52mg/100ml, 0.1-50mg/100ml, 0.1-45mg/100ml, 0.1-40mg/100ml, 0.1-35mg/100ml, 0.1-32mg/100ml, 0.1-30mg/100ml, 0.1 ⁇ 25mg/100ml, 0.1-21mg/100ml, 0.1-20mg/100ml, 0.1-15mg/100ml, 0.1-10mg/100ml, 0.1-5mg/100ml, 0.5-52mg/100ml, 0.5-50mg/100ml, 0.5-45mg /100ml, 0.5-40mg/100ml, 0.5-35mg/100ml, 0.5-30mg/100ml, 0.5-25mg/100ml, 0.5-21mg/100ml, 0.5-20mg/100ml, 0.5-15mg/100ml, 0.5-10mg/
- “Calcium content” 0.1 to 52 mg/100 ml, 0.1 to 50 mg/100 ml, 0.1 to 45 mg/100 ml, 0.1 to 40 mg/100 ml, 0.1 to 35 mg/100 ml, 0.1 to 32 mg/100 ml, 0.1 to 30 mg/100 ml, 0.1 ⁇ 25mg/100ml, 0.1-22mg/100ml, 0.1-20mg/100ml, 0.1-19mg/100ml, 0.1-18mg/100ml, 0.1-17mg/100ml, 0.1-16mg/100ml, 0.1-15mg/100ml, 0.1-14mg /100ml, 0.1-13mg/100ml, 0.1-12mg/100ml, 0.1-11mg/100ml, 0.1-10mg/100ml, 1-50mg/100ml, 1-45mg/100ml, 1-40mg/100ml, 1-35mg/100ml , 1
- the sweetness intensity X1 of natural sugar is not limited to the combination of the numerical values exemplified above, but within the range of 0.1 ⁇ (X1+X2) ⁇ 20, the sweetness intensity X1, sweetness intensity X2, sodium content, potassium and/or potassium described in the present specification. It is possible to combine any of the values of calcium content, sweetness intensity X3 and energy.
- the tea beverage of the present invention includes antioxidants (sodium erythorbate, etc.), emulsifiers (sucrose fatty acid ester, sorbitan fatty acid ester, polyglycerin fatty acid ester, etc.), flavors (tea flavor, etc.) as long as the effects of the present invention are not impaired. ) And the like can be appropriately mixed.
- the present invention provides the following tea beverages (hereinafter referred to as "tea beverage A of the present invention").
- tea beverage A An amount of natural sugar equivalent to sweetness intensity X1
- B a high-intensity sweetener selected from the group consisting of rebaudioside M, rebaudioside D, and combinations thereof in an amount equivalent to sweetness intensity X2,
- C 17 mg/100 ml or more and less than 40 mg/100 ml sodium, and
- a tea beverage exhibiting sweetness of sweetness intensity X3 due to the components (a) to (d), and 0.1 ⁇ (X1+X2) ⁇ 20.
- sweetness intensity X1 sweetness intensity X2, sodium content, potassium and/or calcium content, sweetness intensity X3, high-potency sweetener concentration (content) P2, and energy are preferred.
- the range is as follows, for example. These values can be arbitrarily combined within the range of 0.1 ⁇ (X1+X2) ⁇ 20.
- “Sweetness intensity X1" 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 to 5.5, 0.05 to 6.0, 0.05 to 6.5, 0.05 to 7.0, 0.05 to 7.5, 0.05 to 8.0, 0.05 to 8.25, 0.05 to 8.5, 0.05 to 8.75, 0.05 to 9.0, 0.05 to 9.25, 0.05 to 9.5, 0.05 to 9.75, 0.05 to 10.0, 0.1 ⁇ 0.5, 0.1 ⁇ 1.0, 0.1 ⁇ 1.5, 0.1 ⁇ 2.0, 0.1 ⁇ 2.5, 0.1 ⁇ 3.0, 0.1 ⁇ 3.5, 0.1 ⁇ 4.0, 0.1 ⁇ 4.5, 0.1 ⁇ 5.0, 0.1 ⁇ 5.5, 0.1 ⁇ 5.9, 0.1 ⁇ 6.0, 0.1 ⁇ 6.5, 0.1 7.0,
- “Sweetness intensity X2" 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 to 5.5, 0.5 ⁇ 1.0, 0.5 ⁇ 1.5, 0.5 ⁇ 2.0, 0.5 ⁇ 2.5, 0.5 ⁇ 3.0, 0.5 ⁇ 3.5, 0.5 ⁇ 4.0, 0.5 ⁇ 4.5, 0.5 ⁇ 5.0, 0.5 ⁇ 5.5, 1.0 ⁇ 1.5, 1.0 ⁇ 2.0, 1.0 ⁇ 2.5, 1.0-3.0, 1.0-3.5, 1.0-4.0, 1.0-4.5, 1.0-5.0, 1.0-5.5, 1.5-2.0, 1.5-2.5, 1.5-3.0, 1.5-3.5, 1.5-4.0, 1.5-4.5, 1.5-5.0, 1.5-5.5, 2.0-2.5, 2.0-3.0, 2.0-3.5, 2.0-4.0, 2.0-4.5, 2.0-5.0, 2.0-5.5, 1.5-
- Sodium content 17-40 mg/100 ml, 17 mg/100 ml or more and less than 40 mg/100 ml, 17-35 mg/100 ml, 17-30 mg/100 ml, 17-25 mg/100 ml, 17-20 mg/100 ml, 22-40 mg/ 100 ml, 22-35 mg/100 ml, 22-30 mg/100 ml, 22-25 mg/100 ml, 27-40 mg/100 ml, 27-35 mg/100 ml, 27-30 mg/100 ml.
- Constent of potassium 10-21mg/100ml, 10-20mg/100ml, 10-15mg/100ml, 12-21mg/100ml, 12-20mg/120ml, 12-15mg/120ml, 15-21mg/100ml, 15 ⁇ 20mg/100ml.
- Calcium content 10-22mg/100ml, 10-20mg/100ml, 10-19mg/100ml, 10-18mg/100ml, 10-17mg/100ml, 10-16mg/100ml, 10-15mg/100ml, 15 ⁇ 22mg/100ml, 15-20mg/100ml, 20-25mg/100ml, 20-22mg/100ml.
- the forms of natural sugar including an example of a combination of glucose, sucrose, fructose, maltose, oligosaccharide, isomerized sugar and lactose
- sodium, potassium and calcium are the same as those of the tea beverage of the present invention. It is the same as the definition described in the item.
- the present invention provides the following tea beverage (hereinafter, referred to as “tea beverage B of the present invention”).
- tea beverage B of the present invention An amount of natural sugar equivalent to sweetness intensity X1
- B An amount of Rakan fruit extract equivalent to sweetness intensity X2
- C 17 mg/100 ml or more and less than 40 mg/100 ml of sodium
- a tea beverage exhibiting sweetness of sweetness intensity X3 due to the components (a) to (d), and 0.1 ⁇ (X1+X2) ⁇ 20.
- the sweetness intensity X1, the sweetness intensity X2, the content of sodium, the content of potassium and/or calcium, the sweetness intensity X3, the content P2 of the Rakan fruit extract, and the preferable range of energy are, for example, It is as follows. These values can be arbitrarily combined within the range of 0.1 ⁇ (X1+X2) ⁇ 20.
- “Sweetness intensity X1" 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 to 5.5, 0.05 to 6.0, 0.05 to 6.5, 0.05 to 7.0, 0.05 to 7.5, 0.05 to 8.0, 0.05 to 8.25, 0.05 to 8.5, 0.05 to 8.75, 0.05 to 9.0, 0.05 to 9.25, 0.05 to 9.5, 0.05 to 9.75, 0.05 to 10.0, 0.1 ⁇ 0.5, 0.1 ⁇ 1.0, 0.1 ⁇ 1.5, 0.1 ⁇ 2.0, 0.1 ⁇ 2.5, 0.1 ⁇ 3.0, 0.1 ⁇ 3.5, 0.1 ⁇ 4.0, 0.1 ⁇ 4.5, 0.1 ⁇ 5.0, 0.1 ⁇ 5.5, 0.1 ⁇ 5.9, 0.1 ⁇ 6.0, 0.1 ⁇ 6.5, 0.1 7.0,
- “Sweetness intensity X2" 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 to 5.5, 0.5 ⁇ 1.0, 0.5 ⁇ 1.5, 0.5 ⁇ 2.0, 0.5 ⁇ 2.5, 0.5 ⁇ 3.0, 0.5 ⁇ 3.5, 0.5 ⁇ 4.0, 0.5 ⁇ 4.5, 0.5 ⁇ 5.0, 0.5 ⁇ 5.5, 1.0 ⁇ 1.5, 1.0 ⁇ 2.0, 1.0 ⁇ 2.5, 1.0-3.0, 1.0-3.5, 1.0-4.0, 1.0-4.5, 1.0-5.0, 1.0-5.5, 1.5-2.0, 1.5-2.5, 1.5-3.0, 1.5-3.5, 1.5-4.0, 1.5-4.5, 1.5-5.0, 1.5-5.5, 2.0-2.5, 2.0-3.0, 2.0-3.5, 2.0-4.0, 2.0-4.5, 2.0-5.0, 2.0-5.5, 1.5-
- Sodium content 17-40 mg/100 ml, 17 mg/100 ml or more and less than 40 mg/100 ml, 17-35 mg/100 ml, 17-30 mg/100 ml, 17-25 mg/100 ml, 17-20 mg/100 ml, 22-40 mg/ 100 ml, 22-35 mg/100 ml, 22-30 mg/100 ml, 22-25 mg/100 ml, 27-40 mg/100 ml, 27-35 mg/100 ml, 27-30 mg/100 ml.
- Constent of potassium 10-21mg/100ml, 10-20mg/100ml, 10-15mg/100ml, 12-21mg/100ml, 12-20mg/120ml, 12-15mg/120ml, 15-21mg/100ml, 15 ⁇ 20mg/100ml.
- Calcium content 10-22mg/100ml, 10-20mg/100ml, 10-19mg/100ml, 10-18mg/100ml, 10-17mg/100ml, 10-16mg/100ml, 10-15mg/100ml, 15 ⁇ 22mg/100ml, 15-20mg/100ml, 20-25mg/100ml, 20-22mg/100ml.
- the forms of natural sugar including an example of a combination of glucose, sucrose, fructose, maltose, oligosaccharide, isomerized sugar and lactose
- sodium, potassium and calcium are the tea of the present invention. It is the same as the definition described in the item of beverage.
- the present invention provides the following tea beverage (hereinafter, referred to as “tea beverage C of the present invention”).
- tea beverage C of the present invention An amount of natural sugar equivalent to sweetness intensity X1,
- B an amount of mogroside V equivalent to sweetness intensity X2,
- C 17 mg/100 ml or more and less than 40 mg/100 ml sodium, and
- D 10-21 mg/100 ml potassium and/or 10-22 mg/100 ml calcium
- a tea beverage exhibiting sweetness of sweetness intensity X3 due to the components (a) to (d), and 0.1 ⁇ (X1+X2) ⁇ 20.
- sweetness intensity X1, sweetness intensity X2, sodium content, potassium and/or calcium content, sweetness intensity X3, mogroside V content P2 (ppm), and preferable range of energy Is for example, as follows. These values can be arbitrarily combined within the range of 0.1 ⁇ (X1+X2) ⁇ 20.
- “Sweetness intensity X1" 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 to 5.5, 0.05 to 6.0, 0.05 to 6.5, 0.05 to 7.0, 0.05 to 7.5, 0.05 to 8.0, 0.05 to 8.25, 0.05 to 8.5, 0.05 to 8.75, 0.05 to 9.0, 0.05 to 9.25, 0.05 to 9.5, 0.05 to 9.75, 0.05 to 10.0, 0.1 ⁇ 0.5, 0.1 ⁇ 1.0, 0.1 ⁇ 1.5, 0.1 ⁇ 2.0, 0.1 ⁇ 2.5, 0.1 ⁇ 3.0, 0.1 ⁇ 3.5, 0.1 ⁇ 4.0, 0.1 ⁇ 4.5, 0.1 ⁇ 5.0, 0.1 ⁇ 5.5, 0.1 ⁇ 5.9, 0.1 ⁇ 6.0, 0.1 ⁇ 6.5, 0.1 7.0,
- “Sweetness intensity X2" 0.05 to 0.5, 0.05 to 1.0, 0.05 to 1.5, 0.05 to 2.0, 0.05 to 2.5, 0.05 to 3.0, 0.05 to 3.5, 0.05 to 4.0, 0.05 to 4.5, 0.05 to 5.0, 0.05 to 5.5, 0.5 ⁇ 1.0, 0.5 ⁇ 1.5, 0.5 ⁇ 2.0, 0.5 ⁇ 2.5, 0.5 ⁇ 3.0, 0.5 ⁇ 3.5, 0.5 ⁇ 4.0, 0.5 ⁇ 4.5, 0.5 ⁇ 5.0, 0.5 ⁇ 5.5, 1.0 ⁇ 1.5, 1.0 ⁇ 2.0, 1.0 ⁇ 2.5, 1.0-3.0, 1.0-3.5, 1.0-4.0, 1.0-4.5, 1.0-5.0, 1.0-5.5, 1.5-2.0, 1.5-2.5, 1.5-3.0, 1.5-3.5, 1.5-4.0, 1.5-4.5, 1.5-5.0, 1.5-5.5, 2.0-2.5, 2.0-3.0, 2.0-3.5, 2.0-4.0, 2.0-4.5, 2.0-5.0, 2.0-5.5, 1.5-
- Sodium content 17-40 mg/100 ml, 17 mg/100 ml or more and less than 40 mg/100 ml, 17-35 mg/100 ml, 17-30 mg/100 ml, 17-25 mg/100 ml, 17-20 mg/100 ml, 22-40 mg/ 100 ml, 22-35 mg/100 ml, 22-30 mg/100 ml, 22-25 mg/100 ml, 27-40 mg/100 ml, 27-35 mg/100 ml, 27-30 mg/100 ml.
- Constent of potassium 10-21mg/100ml, 10-20mg/100ml, 10-15mg/100ml, 12-21mg/100ml, 12-20mg/120ml, 12-15mg/120ml, 15-21mg/100ml, 15 ⁇ 20mg/100ml.
- Calcium content 10-22mg/100ml, 10-20mg/100ml, 10-19mg/100ml, 10-18mg/100ml, 10-17mg/100ml, 10-16mg/100ml, 10-15mg/100ml, 15 ⁇ 22mg/100ml, 15-20mg/100ml, 20-25mg/100ml, 20-22mg/100ml.
- the forms of natural sugars including examples of combinations of glucose, sucrose, fructose, maltose, oligosaccharides, isomerized sugars and lactose
- sodium, potassium and calcium are teas of the present invention. It is the same as the definition described in the item of beverage.
- the present invention provides, as yet another aspect, the following method for producing a tea beverage with increased sweetness (hereinafter, referred to as “method of the present invention”).
- the high-potency sweetener comprises at least one high-potency sweetener b1 selected from the group consisting of rebaudioside M, rebaudioside D, rebaudioside N, rebaudioside O, rebaudioside E, Rakan fruit extract, mogroside V and thaumatin, A method for producing a tea beverage, wherein the components (a) to (d) exhibit a sweetness of sweetness intensity X3 and 0.1 ⁇
- the tea beverage produced by the method of the present invention is the tea beverage of the present invention described in the above item "1. Tea beverage with improved taste quality exhibited by natural sugar and a high-intensity sweetener".
- the "raw material” in the method of the present invention may be each material necessary for producing a tea beverage, such as tea leaves or an aqueous medium, or a mixture thereof (that is, an extract from tea leaves), and a preservative. , Additional ingredients such as flavors, carriers, milk and the like may be included.
- the “raw material” may be composed of a plurality of materials. However, regardless of the types of raw materials, the finally produced tea beverage of the present invention does not contain, as a sweetener, a component exhibiting sweetness other than (a) and (b).
- any of the following steps (i) to (iii) may be performed first.
- (I) (a) A step of adding an amount of natural sugar equivalent to sweetness intensity X1 and (b) an amount of high intensity sweetener equivalent to sweetness intensity X2 (ii) (c) Sodium concentration in the beverage is 50 mg/100 ml Step (iii) (d) adding potassium so that the potassium content in the beverage is 0.1 to 52 mg/100 ml and/or the calcium content in the beverage is 0.1 to 52 mg The process of adding calcium to 100ml
- step (i) (a) sweetness intensity X1 equivalent amount of natural sugar and (b) sweetness intensity X2 amount equivalent to high intensity sweetener will be added to the raw material, but (a) sweetness intensity X1 A considerable amount of natural sugar and (b) a sweetness intensity X2 equivalent amount of high intensity sweetener may be added separately. Further, even when (a) the amount of natural sugar corresponding to the sweetness intensity X1 is added, it is not necessary to add the amount of natural sugar equivalent to the sweetness intensity X1 at once, and the natural sugar may be added several times. Similarly, (b) it is not necessary to add an amount of natural sugar equivalent to the sweetness intensity X2 at once, even when adding a high-intensity sweetener in an amount equivalent to the sweetness intensity X2.
- Sodium added to the raw material in the step (ii) is, for example, sodium chloride, sodium hydroxide, sodium malate, sodium sulfate, sodium citrate, sodium phosphate, sodium carbonate, sodium disulfide, sodium bicarbonate, sodium alginate, arginine.
- Sodium acid, sodium glucoheptanate, sodium gluconate, sodium glutamate, sodium tartrate, sodium aspartate, sodium lactate, sodium caseinate may be in at least one form selected from the group consisting of sodium ascorbate and mixtures thereof. ..
- the tea leaf extract may also contain sodium.
- the total content of tea leaf-derived sodium and additional sodium is adjusted to be less than 50 mg/100 ml.
- Potassium that can be added to the raw materials in the step (iii) is, for example, potassium alginate, potassium chloride, potassium citrate, potassium gluconate, potassium L-glutamate, potassium bromate, DL-potassium hydrogen tartrate, potassium L-tartrate, potassium nitrate.
- Calcium which can be added to the raw material in the step (iii) is, for example, calcium L-ascorbate, calcium alginate, calcium disodium ethylenediamine tetraacetate, calcium chloride, calcium carboxymethyl cellulose, calcium citrate, calcium glycerophosphate, calcium gluconate, L-.
- It may be in at least one form selected from the group consisting of calcium acidate, 5'-ribonucleotide calcium, calcium sulfate, tricalcium phosphate, calcium monohydrogen phosphate, calcium dihydrogen phosphate and mixtures thereof.
- tea leaves extract and water may contain calcium, in which case the total content of calcium from tea leaves and additional calcium should be 0.1-52 mg/100 ml. adjust.
- the calcium content in the milk components is also taken into consideration.
- addition means not only the operation of actually adding any of the components (a) to (d) to the raw materials, but also the tea finally produced through the production process of the tea beverage of the present invention. It also means an operation of adjusting the contents of the components (a) to (d) in the beverage to sweetness intensity X1 equivalent amount, sweetness intensity X2 equivalent amount, less than 50 mg/100 ml and 0.1 to 52 mg/100 ml, respectively.
- the first raw material contains milk, cereals, beans and its extract in addition to the components extracted from tea leaves into an aqueous medium such as water, and therefore the raw materials are preliminarily composed of components (a) to (d).
- the second raw material for mixing with the first raw material also includes components (a) to (d), and the mixture of the first and second raw materials
- the tea beverage of the invention When the tea beverage of the invention can be produced, there is no operation of individually adding the components (a) to (d) to the raw materials, but in the method of the present invention, the tea beverage of the present invention finally produced.
- Is (a) an amount of natural sugar equivalent to sweetness intensity X1, (b) an amount of high intensity sweetener equivalent to sweetness intensity X2, (c) sodium less than 50 mg/100 ml and (d) 0.1 to 52 mg/100 ml potassium. And/or steps (i)-(iii) are considered to have been performed as long as they contain 0.1-52 mg/100 ml calcium.
- the method for producing a beverage of the present invention includes the step of filling the container with the tea beverage.
- a predetermined amount of the tea beverage can be filled in the can and retort sterilized at 120 to 125° C. for about 5 to 20 minutes for heat sterilization.
- PET bottles paper packs and bottled beverages, for example, UHT sterilization at 130 to 145°C for about 2 to 120 seconds is performed, and a predetermined amount is hot packed or aseptically filled at low temperature.
- a packaged beverage can be obtained.
- “tea beverage”, “natural sugar”, “sweetness intensity X1”, “high intensity sweetener”, “sweetness intensity X2”, sodium content, potassium and/or calcium content The form of sodium in the tea beverage, the “sweetness intensity X3” and the energy are the same as the definitions described in the item of the tea beverage, and the numerical values described in the item of the tea beverage are applied as they are. Further, examples of “combination of glucose, sucrose, fructose, maltose, oligosaccharide, isomerized sugar and lactose” and “combination of high intensity sweetener” are also the same as the examples described in the above-mentioned tea beverage item.
- the tea beverage produced by the method of the present invention (A) An amount of natural sugar whose sweetness intensity value X1 is equivalent to 0.1 to 5, (B) A high-intensity sweetener whose sweetness intensity value X2 is equivalent to 0.1 to 3; (C) less than 50 mg/100 ml sodium, and (d) 0.1-52 mg/100 ml potassium and/or 0.1-52 mg/100 ml calcium,
- the tea beverage has a sweetness intensity X3 of 5.5 to 12.5 due to the components (a) to (d) and 0.2 ⁇ (X1+X2) ⁇ 12.5.
- the tea beverage produced by the method of the present invention (A) An amount of natural sugar having a sweetness intensity X1 of 3 to 5, (B) High intensity sweetener whose sweetness intensity X2 is equivalent to 1 to 3, (C) less than 50 mg/100 ml sodium, and (d) 0.1-32 mg/100 ml potassium and/or 0.1-32 mg/100 ml calcium, A tea beverage having a sweetness intensity X3 of 5.5 to 12.5 due to the components (a) to (d) and 4 ⁇ (X1+X2) ⁇ 12.5.
- the tea beverage produced by the method of the present invention (A) An amount of natural sugar whose sweetness intensity value X1 is equivalent to 0.1 to 5, (B) A high-intensity sweetener whose sweetness intensity value X2 is equivalent to 0.1 to 3; (C) 7 mg/100 ml or more and less than 40 mg/100 ml of sodium, and (d) 0.1-52 mg/100 ml of potassium and/or 0.1-52 mg/100 ml of calcium,
- the tea beverage has a sweetness intensity of X3 of 2.0 to 12.0 and 0.2 ⁇ (X1+X2) ⁇ 12.5 due to the components (a) to (d).
- the tea beverage produced by the method of the present invention (A) An amount of natural sugar having a sweetness intensity X1 of 3 to 5, (B) High intensity sweetener whose sweetness intensity X2 is equivalent to 1 to 3, (C) 7 mg/100 ml or more and less than 40 mg/100 ml of sodium, and (d) 0.1-32 mg/100 ml of potassium and/or 0.1-32 mg/100 ml of calcium, A tea beverage having a sweetness intensity X3 of 5.5 to 12.0 due to the components (a) to (d) and 4 ⁇ (X1+X2) ⁇ 12.5.
- the present invention provides the following method (hereinafter referred to as “method A of the present invention”).
- a high-intensity sweetener selected from the group consisting of rebaudioside M, rebaudioside D, and combinations thereof in an amount of (i) (a) sweetness intensity X1 of natural sugar and (b) amount of sweetness intensity X2 to raw materials
- the process of (Ii) (c) a step of adding sodium such that the sodium content in the beverage is 17 mg/100 ml or more and less than 40 mg/100 ml, (Iii) (d)
- a step of adding potassium so that the potassium content in the beverage becomes 0.1 to 52 mg/100 ml and/or calcium is added so that the calcium content in the beverage becomes 0.1 to 52 mg/100 ml
- Process including, A method for producing a tea beverage, wherein the components (a) to (d) exhibit a sweetness of sweetness intensity X3 and 0.1 ⁇ (X1+X2) ⁇ 20.
- the “sweetener”, the “sweetness intensity X2”, the content of sodium, the content of potassium and/or calcium, the form of sodium, potassium and/or calcium in the tea beverage, the “sweetness intensity X3” and the energy are the above-mentioned tea beverage A.
- the definition and the like are the same as those in the item, and the numerical values and the like described in the item of the tea beverage A are directly applied.
- the tea beverage in the method A of the present invention corresponds to the tea beverage A of the present invention, and the matters described above regarding the tea beverage A of the present invention apply as they are. Furthermore, the order of “raw materials”, “addition”, steps (i), (ii) and (iii) and the addition mode of each component in the method A of the present invention are the same as the definitions described above for the method of the present invention. ..
- the present invention provides the following method (hereinafter, referred to as “method B of the present invention”).
- a step of adding (i) (a) an amount of sweetness X1 of natural sugar and (b) an amount of sweetness intensity X2 of Rakan fruit extract to the raw materials; (Ii) (c) a step of adding sodium such that the sodium content in the beverage is 17 mg/100 ml or more and less than 40 mg/100 ml, (Iii) (d)
- a step of adding potassium so that the potassium content in the beverage becomes 0.1 to 52 mg/100 ml and/or calcium is added so that the calcium content in the beverage becomes 0.1 to 52 mg/100 ml
- Process including, A method for producing a tea beverage, wherein the components (a) to (d) exhibit a sweetness of sweetness intensity X3 and 0.1 ⁇ (X1+X2) ⁇ 20.
- the definition and the like are the same, and the numerical values and the like are the same as those described in the item of the tea beverage B.
- the tea beverage in the method B of the present invention corresponds to the tea beverage B of the present invention, and the matters described above regarding the tea beverage B of the present invention apply as they are.
- the “raw materials”, the “addition”, the order of steps (i), (ii) and (iii) and the addition mode of each component in the method B of the present invention are the same as the definitions described above for the method of the present invention. ..
- the present invention provides the following method (hereinafter, referred to as “method C of the present invention”).
- a step of adding (i) (a) a natural sugar amount of sweetness X1 and (b) a mogroside V amount of sweetness X2 to the raw materials; (Ii) (c) a step of adding sodium such that the sodium content in the beverage is 17 mg/100 ml or more and less than 40 mg/100 ml, (Iii) (d)
- a step of adding potassium so that the potassium content in the beverage becomes 0.1 to 52 mg/100 ml and/or calcium is added so that the calcium content in the beverage becomes 0.1 to 52 mg/100 ml
- Process including, A method for producing a tea beverage, wherein the components (a) to (d) exhibit a sweetness of sweetness intensity X3 and 0.1 ⁇ (X1+X2) ⁇ 20.
- “tea beverage” "natural sugar” (including an example of a combination of glucose, sucrose, fructose, maltose, oligosaccharide, isomerized sugar, and lactose), "sweetness intensity X1", “mogroside V”.
- the “sweetness intensity X2”, the content of sodium, the content of potassium and/or calcium, the form of sodium, potassium and/or calcium in the tea beverage, the “sweetness intensity X3” and the energy are as described in the above tea beverage C.
- the definition is the same as that described above, and the numerical values and the like are the same as those described in the item of the tea beverage C.
- the tea beverage in the method C of the present invention corresponds to the tea beverage C of the present invention, and the matters described above regarding the tea beverage C of the present invention apply as they are. Furthermore, the order of “raw materials”, “addition”, steps (i), (ii) and (iii) and the addition mode of each component in the method C of the present invention are the same as the definitions described above for the method of the present invention. .
- the sweetness intensity X1 of natural sugar, the sweetness intensity X2 of the high intensity sweetener, the content of sodium, potassium and/or calcium and the sweetness intensity X3 exhibited by the tea beverage are 0.1. Any value may be used as long as ⁇ (X1+X2) ⁇ 20 is satisfied.
- sweetness intensity X1, sweetness intensity X2, sodium content, potassium and/or calcium It is possible to combine any of the content, the sweetness intensity X3 and the energy value.
- the present invention provides, as another embodiment, a concentrate for providing the tea beverage of the present invention (hereinafter, referred to as “concentrate of the present invention”).
- the concentrate of the invention comprises (A) An amount of natural sugar equivalent to sweetness intensity X4, (B) Sweetness intensity X5 equivalent amount of high intensity sweetener, (C) less than 500 mg/100 ml sodium, and (d) 1-520 mg/100 ml potassium and/or 1-520 mg/100 ml calcium, (X4+X5) ⁇ 200, preferably 1 ⁇ (X4+X5) ⁇ 200, more preferably 50 ⁇ (X4+X5) ⁇ 200.
- the concentrate of the present invention is used to provide a tea beverage by diluting it at an arbitrary ratio.
- the "tea beverage” is the same as that described in "1. Tea beverage with improved taste quality exhibited by natural sugar and high-intensity sweetener”.
- the concentrate of the invention can be used in beverages as a syrup or stock solution. In that case, it can be used after diluting 2-fold, 3-fold, 4-fold, 5-fold, 6-fold, 7-fold, 8-fold, 9-fold or 10-fold.
- the concentrate of the present invention is preferable in terms of storage stability and transportability because it is concentrated.
- the concentrate of the present invention may be solid or liquid.
- the concentrate of the present invention is a 2 to 10-fold concentrate of the tea beverage of the present invention, preferably a 3 to 9-fold concentrate, more preferably a 4 to 8-fold concentrate, and further preferably 5 to It is a 7-fold concentrate.
- a concentrate according to one aspect of the invention is a 5 times concentrate of the tea beverage of the invention, (A) An amount of natural sugar equivalent to sweetness intensity X6, (B) High intensity sweetener with an amount equivalent to sweetness intensity X7, (C) less than 250 mg/100 ml sodium, and (d) 1-260 mg/100 ml potassium and/or 1-260 mg/100 ml calcium, (X6+X7) ⁇ 100, preferably 0.5 ⁇ (X6+X7) ⁇ 100, more preferably 20 ⁇ (X6+X7) ⁇ 100.
- a concentrate according to one aspect of the invention is a 6-fold concentrate of the tea beverage of the invention, (A) An amount of natural sugar equivalent to sweetness intensity X8, (B) High intensity sweetener with an amount equivalent to sweetness intensity X9, (C) less than 300 mg/100 ml sodium, and (d) 0.6-312 mg/100 ml potassium and/or 0.6-312 mg/100 ml calcium, (X8+X9) ⁇ 120, preferably 0.6 ⁇ (X8+X9) ⁇ 120, more preferably 30 ⁇ (X8+X9) ⁇ 120.
- a concentrate according to another aspect of the invention is an 8-fold concentrate of the tea beverage of the invention, (A) An amount of natural sugar equivalent to sweetness intensity X10, (B) A sweetener having a sweetness level of X11 and a high-intensity sweetener, (C) less than 400 mg/100 ml sodium, and (d) 0.8-416 mg/100 ml potassium and/or 0.8-416 mg/100 ml calcium, (X10+X11) ⁇ 160, preferably 0.8 ⁇ (X10+X11) ⁇ 160, more preferably 40 ⁇ (X10+X11) ⁇ 160.
- the present invention provides, as another embodiment, a method for enhancing sweetness of a tea beverage (hereinafter, referred to as “sweetness enhancing method of the present invention”).
- the method for enhancing sweetness of the present invention provides a tea beverage comprising (a) an amount of natural sugar equivalent to sweetness intensity X1, (B) a sweetener having a high sweetness of an amount equivalent to sweetness intensity X2, It is characterized by containing (c) less than 50 mg/100 ml of sodium, and (d) 0.1 to 52 mg/100 ml of potassium and/or 0.1 to 52 mg/100 ml of calcium.
- the sweetness of the tea beverage is enhanced, and the sum of the sweetness intensity when the component (a) is simply added to the tea beverage and the component (b) is added to the tea beverage. It is possible to provide a tea beverage having a sweetness exceeding the above.
- “tea beverage”, “natural sugar”, “sweetness intensity X1”, “high intensity sweetener”, “sweetness intensity X2”, sodium content, potassium and/or calcium content The amount, the form of sodium, potassium and/or calcium in the tea beverage, the “sweetness intensity X3” and the energy are the same as the definitions described in the item of the tea beverage, and the numerical values are the values described in the item of the tea beverage. Remains true.
- examples of “combination of glucose, sucrose, fructose, maltose, oligosaccharide, isomerized sugar and lactose” and “combination of high intensity sweetener” are also the same as the examples described in the above-mentioned tea beverage item.
- the phrase “at least” means that the number of specific items may be equal to or more than the listed numbers. Further, in the present application, the word “about” exists within a range of ⁇ 25%, ⁇ 10%, ⁇ 5%, ⁇ 3%, ⁇ 2% or ⁇ 1% of the numerical value that the subject follows “about”. Means to do. For example, "about 10" means in the range of 7.5 to 12.5.
- the sodium and potassium contents contained in the base beverage before the addition of various additives were measured by the atomic absorption method using SpectrAA240FS (manufactured by Agilent Technologies), and the calcium content was measured. The amount was measured by ICP emission spectrometry using 5100VDV (manufactured by Agilent Technologies) and ICPE-9000 (manufactured by Shimadzu Corporation). Further, the sodium content of the beverage sample after blending the various additives, to the sodium content in the base beverage measured by the above method, added the sodium content contained in the various additives calculated from the blended amount It is a thing. The same applies to potassium and calcium.
- Example 1 Evaluation of taste improvement effect of oolong tea beverage by sodium, potassium and calcium Experimental method Natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ , rebaudioside D (RebD) (purity 95% or more), sodium gluconate, and chloride in the ratios shown in Table 2 below.
- Oolong tea extract with potassium and calcium chloride (sodium content: 10 (mg/100 ml), potassium content: 0.2 mg/100 ml, calcium content: 11.3 mg/100 ml, polyphenol content: 400 (ppm), energy : 0 (kcal/100 ml)) to prepare a beverage sample. Further, a solution containing neither sodium gluconate, potassium chloride nor calcium chloride was used as Sample 1. Brix in Table 2 was calculated from the concentration of natural sugar, sodium content was calculated from the sodium gluconate addition amount and sodium content derived from tea leaves, and potassium content was added potassium chloride and potassium content derived from tea leaves.
- the calcium content is calculated from the calcium chloride addition amount and the tea leaf-derived calcium content, and the energy (kcal/100 ml) is the energy of the components derived from RebD, sodium, potassium and calcium being 0 (kcal/100 ml).
- the energy is the energy of the components derived from RebD, sodium, potassium and calcium being 0 (kcal/100 ml).
- sample 1-1 has 20 mg/100 ml of Na added
- sample 1-2 has 20 mg/100 ml of total added Na+K
- sample 1-3 has total added amount of Na+Ca. Is 20 mg/100 ml.
- the taste improving effect of sodium, potassium and calcium was verified by sensory comparing the tastes of these beverage samples.
- the taste qualities that are sensory evaluation items are "sweetness”, “flavor (fragrance) strength”, “miscellaneous taste (bitterness, astringency, etc.) strength", and “salty strength”. Trained persons (4 persons) became panelists and verified.
- the “sensory evaluation score” for each taste quality was calculated for each beverage sample based on the common taste quality judgment standard that each panelist had through daily training.
- the “sensory evaluation score” is a numerical value of the difference in taste from Sample 1 in the range of ⁇ 3.0 to +3.0, with the degree of taste of Sample 1 being “0” (reference).
- the criteria for each item of the "sensory evaluation score” are as follows.
- the "sensory evaluation score” calculated by each panel was defined as a “conversion score” according to the following criteria, and the total value of the conversion scores of the four panelists for each taste was calculated.
- Conversion score -Conversion score "3": Sensory evaluation score is +1.5 or more.
- -Conversion score "0” Sensory evaluation score is more than -0.5 and less than +0.5.
- -Conversion score "-1” Sensory evaluation score is higher than -1.0 and lower than -0.5.
- -Conversion score "-2" Sensory evaluation score is higher than -1.5 and lower than -1.0.
- -Conversion score "-3” Sensory evaluation score is -1.5 or less.
- sweetness and “flavor intensity” the larger the converted score total value is, the more preferable.
- poor taste intensity and “salty strength” are converted score total values. Is smaller, the more preferable.
- Results Table 3 shows the calculated results of the converted score total values of the respective taste qualities.
- Example 2 Evaluation of taste improving effect of green tea beverage by sodium, potassium and calcium Experimental method
- natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ , rebaudioside D (RebD) (purity of 95% or more) in the ratios shown in Table 4 below.
- RebD rebaudioside D
- sodium gluconate, potassium chloride and calcium chloride in green tea extract sodium content: 6 (mg/100 ml), potassium content: 10.7 mg/100 ml, calcium content: 0.2 mg/100 ml, catechin content: 300) (ppm), energy: 0 (kcal/100 ml)
- a solution containing neither sodium gluconate, potassium chloride nor calcium chloride was used as Sample 2.
- Brix in Table 4 was calculated from the concentration of natural sugar, sodium content was calculated from the sodium gluconate concentration and sodium content derived from tea leaves, and potassium content was calculated from the potassium chloride concentration and potassium content derived from tea leaves.
- the calcium content was calculated from the calcium chloride concentration and the calcium content derived from tea leaves, and the energy (kcal/100 ml) was calculated with the energy of the components derived from RebD, sodium, potassium and calcium being 0 (kcal/100 ml). .. As shown in Table 4, sample 2-1 had a Na addition amount of 20 mg/100 ml, sample 2-2 had a total addition amount of Na+K of 20 mg/100 ml, and sample 2-3 had a total addition amount of Na+Ca. Is 20 mg/100 ml.
- Results Table 5 shows the calculated results of the converted score total values of the respective taste qualities.
- Example 3 Evaluation of taste improvement effect of oolong tea by difference in potassium and/or calcium concentration Experimental method As in Example 1, natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ , rebaudioside D (RebD) (purity of 95% or more) in the ratios shown in Table 6 below.
- natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ , rebaudioside D (RebD) (purity of 95% or more) in the ratios shown in Table 6 below.
- Brix in Table 6 is calculated from the concentration of natural sugar, sodium content is calculated from sodium gluconate addition amount and sodium content from tea leaves, potassium content is potassium chloride addition amount and potassium content from tea leaves
- the calcium content was calculated from the added amount of calcium chloride and the calcium content derived from tea leaves, and the energy (kcal/100 ml) was calculated from 0 (kcal/100 ml) derived from RebD, sodium, potassium and calcium.
- Example 4 Evaluation of taste improvement effect of green tea due to difference in potassium and/or calcium concentration Experimental method As in Example 1, natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ , rebaudioside D (RebD) (purity of 95% or more) in the ratios shown in Table 8 below.
- natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ , rebaudioside D (RebD) (purity of 95% or more) in the ratios shown in Table 8 below.
- sodium gluconate, potassium chloride and calcium chloride green tea extract (catechin content: 300 (ppm), sodium content: 6 mg/100 ml, potassium content: 10.7 mg/100 ml, calcium content: 0.2 mg/100 ml) , Energy: 0 (kcal/100 ml)) to prepare a beverage sample.
- a solution containing neither sodium gluconate, potassium chloride nor calcium chloride was designated as Sample 4.
- Brix in Table 8 is calculated from the concentration of natural sugar, sodium content is calculated from the sodium gluconate addition amount and sodium content derived from tea leaves, and potassium content is potassium chloride addition amount and potassium content derived from tea leaves.
- the calcium content was calculated from the added amount of calcium chloride and the calcium content derived from tea leaves, and the energy (kcal/100 ml) was calculated from 0 (kcal/100 ml) derived from RebD, sodium, potassium and calcium.
- Example 5 Evaluation of taste improving effect of oolong tea by various high-intensity sweeteners and sodium and potassium and/or calcium Experimental Method
- natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ and high-potency sweeteners ⁇ Mogroside V (MogV) at the ratios shown in Table 10 below.
- Brix in Table 10 is calculated from the concentration of natural sugar, sodium content is calculated from sodium gluconate addition amount and sodium content derived from tea leaves, potassium content is potassium chloride addition amount and potassium content derived from tea leaves
- the calcium content was calculated from the amount of calcium chloride added and the calcium content derived from tea leaves, and the energy (kcal/100 ml) was calculated as 0 (kcal/100 ml) derived from MogV, RebM, sodium, potassium and calcium. ..
- sample 5-3 had an added amount of Na of 20 mg/100 ml
- samples 5-1 and 5-4 had a total added amount of Na+K of 20 mg/100 ml.
- the total amount of Na+Ca added was 20 mg/100 ml.
- the taste improving effect of sodium, potassium and calcium was verified by sensory comparing the tastes of these beverage samples.
- the tastes that are sensory evaluation items are “sweetness intensity”, “flavor intensity”, “miscellaneous bitterness (bitterness, astringency, etc.) intensity” and “salt intensity”.
- Those who were trained in sensuality (4 people) were panelists and verified. Specifically, the same evaluation as in Example 1 was performed, and the “sensory evaluation score” calculated by each panel was used as a “conversion score” to calculate the total conversion score of four panelists for each taste.
- For Samples 5 to 5-2 the score of Sample 5 was used as the standard (0 point), and for Samples 5A to 5-5, the score of Sample 5A was used as the standard (0 point).
- Results Table 11 shows the calculated results of the converted score total values of the respective taste qualities.
- Example 6 Evaluation of taste improving effect of green tea by various high-intensity sweeteners and sodium and potassium and/or calcium Experimental Method
- natural sugars ⁇ sucrose (manufactured by Taiheiyo Sugar Co., Ltd.), glucose (manufactured by Showa Sangyo Co., Ltd.) ⁇ and high-potency sweeteners ⁇ mogroside V (MogV) at the ratios shown in Table 12 below.
- Brix in Table 12 is calculated from the concentration of natural sugar, sodium content is calculated from sodium gluconate addition amount and sodium content derived from tea leaves, potassium content is potassium chloride addition amount and potassium content derived from tea leaves
- the calcium content was calculated from the amount of calcium chloride added and the calcium content derived from tea leaves, and the energy (kcal/100 ml) was calculated as 0 (kcal/100 ml) derived from MogV, RebM, sodium, potassium and calcium. ..
- the sample 6-3 has a Na addition amount of 20 mg/100 ml
- the samples 6-1 and 6-4 have a total addition amount of Na+K of 20 mg/100 ml.
- the total amount of Na+Ca added was 20 mg/100 ml.
- the taste improving effect of sodium, potassium and calcium was verified by sensory comparing the tastes of these beverage samples.
- the tastes that are sensory evaluation items are “sweetness intensity”, “flavor intensity”, “miscellaneous bitterness (bitterness, astringency, etc.) intensity” and “salt intensity”.
- Those who were trained in sensuality (4 people) were panelists and verified. Specifically, the same evaluation as in Example 1 was performed, and the “sensory evaluation score” calculated by each panel was used as a “conversion score” to calculate the total conversion score of four panelists for each taste.
- For Samples 6 to 6-2 the score of Sample 6 was used as the standard (0 point), and for Samples 6A to 6-5, the score of Sample 6A was used as the standard (0 point).
- Results Table 13 shows the calculated results of the converted score total values of the respective taste qualities.
- the method of the present invention provides a method of increasing the sweetness of a tea beverage or a sweetening composition and providing a good taste, not a simple sweetness obtained by increasing the amounts of natural sugar and a high-potency sweetener used. To be done.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Nutrition Science (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Dispersion Chemistry (AREA)
- Tea And Coffee (AREA)
- Seasonings (AREA)
Abstract
Description
しかしながら、低カロリー飲食品にするためには天然糖使用量を低く抑える必要があり、この点が低カロリーかつ良好な味質を呈する飲食品を提供する際の障害となっている。
[1]
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、および
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含み、
前記高甘味度甘味料が、レバウジオシドM、レバウジオシドD、レバウジオシドN、レバウジオシドO、レバウジオシドE、羅漢果抽出物、モグロシドVおよびソーマチンからなる群より選択される少なくとも1つの高甘味度甘味料b1を含み、
0.1<(X1+X2)≦20である、茶飲料。
[2]
ナトリウム含有量が7mg/100ml以上40mg/100ml未満である、[1]に記載の茶飲料。
[3]
0.1~32mg/100mlのカリウムおよび/または0.1~32mg/100mlのカルシウムを含む、[1]または[2]に記載の茶飲料。
[4]
エネルギーが50Kcal/100ml以下である、[1]~[3]のいずれか一項に記載の茶飲料。
[5]
X1が0.1~5.9である、[1]~[4]のいずれか一項に記載の茶飲料。
[6]
ポリフェノールを200~600ppm含む、[1]~[5]のいずれか一項に記載の茶飲料。
[7]
カテキンを200~600ppm含む、[6]に記載の茶飲料。
[8]
前記天然糖は、グルコース、スクロース、フルクトース、マルトース、オリゴ糖、異性化糖、乳糖、プシコース、アロース、タガトースおよびその組み合わせからなる群より選択される少なくとも1つである、[1]~[7]のいずれか一項に記載の茶飲料。
[9]
前記高甘味度甘味料b1は、レバウジオシドM、レバウジオシドD、羅漢果抽出物、モグロシドVおよびその組み合わせからなる群より選択される少なくとも1つである、[1]~[8]のいずれか一項に記載の茶飲料。
[10]
前記ナトリウムは、塩化ナトリウム、水酸化ナトリウム、リンゴ酸ナトリウム、硫酸ナトリウム、クエン酸ナトリウム、リン酸ナトリウム、炭酸ナトリウム、二硫化ナトリウム、重炭酸ナトリウム、アルギン酸ナトリウム、アルギニン酸ナトリウム、グルコヘプタン酸ナトリウム、グルコン酸ナトリウム、グルタミン酸ナトリウム、酒石酸ナトリウム、アスパラギン酸ナトリウム、乳酸ナトリウム、カゼインナトリウム、アスコルビン酸ナトリウムおよびその混合物からなる群より選択される少なくとも1つである、[1]~[9]のいずれか一項に記載の茶飲料。
[11]
前記カリウムは、アルギン酸カリウム、塩化カリウム、クエン酸カリウム、グルコン酸カリウム、L-グルタミン酸カリウム、臭素酸カリウム、DL-酒石酸水素カリウム、L-酒石酸水素カリウム、硝酸カリウム、水酸化カリウム、ソルビン酸カリウム、炭酸カリウム、乳酸カリウム、ノルビキシンカリウム、ピロ亜硫酸カリウム、ピロリン酸四カリウム、フェロシアン化カリウム、ポリリン酸カリウム、メタリン酸カリウム、硫酸アルミニウムカリウム、硫酸カリウム、リン酸三カリウム、リン酸水素二カリウム、リン酸二水素カリウムおよびその混合物からなる群より選択される少なくとも1つである、[1]~[10]のいずれか一項に記載の茶飲料。
[12]
前記カルシウムは、L-アスコルビン酸カルシウム、アルギン酸カルシウム、エチレンジアミン四酢酸カルシウムニナトリウム、塩化カルシウム、カルボキシメチルセルロースカルシウム、クエン酸カルシウム、グリセロリン酸カルシウム、グルコン酸カルシウム、L-グルタミン酸カルシウム、ケイ酸カルシウム、酢酸カルシウム、酸化カルシウム、水酸化カルシウム、ステアリン酸カルシウム、ステアロイル乳酸カルシウム、ソルビン酸カルシウム、炭酸カルシウム、乳酸カルシウム、パントテン酸カルシウム、ピロリン酸二水素カルシウム、フェロシアン化カルシウム、プロピオン酸カルシウム、5'-リボヌクレオチドカルシウム、硫酸カルシウム、リン酸三カルシウム、リン酸一水素カルシウム、リン酸二水素カルシウムおよびその混合物からなる群より選択される少なくとも1つである、[1]~[11]のいずれか一項に記載の茶飲料。
[13]
17mg/100ml以上40mg/100ml未満のナトリウムを含み、10~21mg/100mlのカリウムおよび/または10~22mg/100mlのカルシウムを含み、エネルギーが50Kcal/100ml以下、かつX1+X2が6以上である、[1]~[12]のいずれか一項に記載の茶飲料。
[14]
容器詰めされている、[1]~[13]のいずれか一項に記載の茶飲料。
[15]
原材料に対し
(i)(a)甘味強度X1の量の天然糖および(b)甘味強度X2の量の高甘味度甘味料を添加する工程と、
(ii)(c)茶飲料に含まれるナトリウム含有量が50mg/100ml未満となるように、ナトリウムを添加する工程と、
(iii)(d)飲料中のカリウム含有量が0.1~52mg/100mlとなるようにカリウムを添加する工程および/または飲料中のカルシウム含有量が0.1~52mg/100mlとなるようにカルシウムを添加する工程と、
を含み、
前記高甘味度甘味料が、レバウジオシドM、レバウジオシドD、レバウジオシドN、レバウジオシドO、レバウジオシドE、羅漢果抽出物、モグロシドVおよびソーマチンからなる群より選択される少なくとも1つの高甘味度甘味料b1を含み、
0.1<(X1+X2)≦20である、茶飲料
を製造する方法。
[16]
前記茶飲料においてナトリウム含有量が7mg/100ml以上40mg/100ml未満である、[15]に記載の方法。
[17]
前記茶飲料が、0.1~32mg/100mlのカリウムおよび/または0.1~32mg/100mlのカルシウムを含む、[15]または[16]に記載の方法。
[18]
前記茶飲料のエネルギーが50Kcal/100ml以下である、[15]~[17]のいずれか一項に記載の方法。
[19]
X1が0.1~5.9である、[15]~[18]のいずれか一項に記載の方法。
[20]
前記茶飲料がポリフェノールを200~600ppm含む、[15]~[19]のいずれか一項に記載の方法。
[21]
前記茶飲料がカテキンを200~600ppm含む、[20]に記載の方法。
[22]
前記天然糖は、グルコース、スクロース、フルクトース、マルトース、オリゴ糖、異性化糖、乳糖、プシコース、アロース、タガトースおよびその組み合わせからなる群より選択される少なくとも1つである、[15]~[21]のいずれか一項に記載の方法。
[23]
前記高甘味度甘味料b1は、レバウジオシドM、レバウジオシドD、羅漢果抽出物、モグロシドVおよびその組み合わせからなる群より選択される少なくとも1つである、[15]~[22]のいずれか一項に記載の方法。
[24]
前記ナトリウムは、塩化ナトリウム、水酸化ナトリウム、リンゴ酸ナトリウム、硫酸ナトリウム、クエン酸ナトリウム、リン酸ナトリウム、炭酸ナトリウム、二硫化ナトリウム、重炭酸ナトリウム、アルギン酸ナトリウム、アルギニン酸ナトリウム、グルコヘプタン酸ナトリウム、グルコン酸ナトリウム、グルタミン酸ナトリウム、酒石酸ナトリウム、アスパラギン酸ナトリウム、乳酸ナトリウム、カゼインナトリウム、アスコルビン酸ナトリウムおよびその混合物からなる群より選択される少なくとも1つである、[15]~[23]のいずれか一項に記載の方法。
[25]
前記カリウムは、アルギン酸カリウム、塩化カリウム、クエン酸カリウム、グルコン酸カリウム、L-グルタミン酸カリウム、臭素酸カリウム、DL-酒石酸水素カリウム、L-酒石酸水素カリウム、硝酸カリウム、水酸化カリウム、ソルビン酸カリウム、炭酸カリウム、乳酸カリウム、ノルビキシンカリウム、ピロ亜硫酸カリウム、ピロリン酸四カリウム、フェロシアン化カリウム、ポリリン酸カリウム、メタリン酸カリウム、硫酸アルミニウムカリウム、硫酸カリウム、リン酸三カリウム、リン酸水素二カリウム、リン酸二水素カリウムおよびその混合物からなる群より選択される少なくとも1つである、[15]~[24]のいずれか一項に記載の方法。
[26]
前記カルシウムは、L-アスコルビン酸カルシウム、アルギン酸カルシウム、エチレンジアミン四酢酸カルシウムニナトリウム、塩化カルシウム、カルボキシメチルセルロースカルシウム、クエン酸カルシウム、グリセロリン酸カルシウム、グルコン酸カルシウム、L-グルタミン酸カルシウム、ケイ酸カルシウム、酢酸カルシウム、酸化カルシウム、水酸化カルシウム、ステアリン酸カルシウム、ステアロイル乳酸カルシウム、ソルビン酸カルシウム、炭酸カルシウム、乳酸カルシウム、パントテン酸カルシウム、ピロリン酸二水素カルシウム、フェロシアン化カルシウム、プロピオン酸カルシウム、5'-リボヌクレオチドカルシウム、硫酸カルシウム、リン酸三カルシウム、リン酸一水素カルシウム、リン酸二水素カルシウムおよびその混合物からなる群より選択される少なくとも1つである、[15]~[25]のいずれか一項に記載の方法。
[27]
前記茶飲料が、17mg/100ml以上40mg/100ml未満のナトリウムを含み、10~21mg/100mlのカリウムおよび/または10~22mg/100mlのカルシウムを含み、エネルギーが50Kcal/100ml以下、かつX1+X2が6以上である、[15]~[26]のいずれか一項に記載の方法。
[28]
Camellia sinensisの茶葉を使用する、[15]~[27]のいずれか一項に記載の方法。
[29]
(a)甘味強度X4相当の量の天然糖、
(b)甘味強度X5相当の量の高甘味度甘味料、
(c)500mg/100ml未満のナトリウム、ならびに
(d)1~520mg/100mlのカリウムおよび/または1~520mg/100mlのカルシウム
を含み、
(X4+X5)≦200である、[1]に記載の茶飲料を提供するための濃縮物。
[30]
茶飲料に
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、ならびに
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含有させることを特徴とする、茶飲料の甘味増強方法。
なお、本明細書において引用した全ての文献、および公開公報、特許公報その他の特許文献は、参照として本明細書に組み込むものとする。
本明細書において、例えば、「A成分の含有量がXmg/100mlである」との記載は、「飲料100mlに対して、A成分がXmg含まれている」ことを意味する。また、例えば、「B成分の含有量がYppmである」との記載は、「飲料の全量(100質量%)に対して、B成分がYppm含まれている」ことを意味する。
本発明は、第1の態様として、以下の茶飲料(以下、「本発明の茶飲料」という)を提供する。
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、ならびに、
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含み、
前記高甘味度甘味料が、レバウジオシドM、レバウジオシドD、レバウジオシドN、レバウジオシドO、レバウジオシドE、羅漢果抽出物、モグロシドV(Mogroside V)およびソーマチンからなる群より選択される少なくとも1つの高甘味度甘味料b1を含み、
0.1<(X1+X2)≦20である、茶飲料。
本発明の茶飲料は、前記成分(a)~(d)により甘味強度X3の甘味を呈する。
本願明細書において、茶飲料には、茶樹(学名:Camellia sinensis)から摘採した生葉を原料として製造される加工品が含まれる。あるいは、茶飲料には、麦茶、甘茶、ゴーヤ茶、コカ茶、ルイボス茶、マタタビ茶、蔓茶、ハトムギ茶、ゆず茶、ハニーブッシュ茶、陣皮茶、ドクダミ茶、熊笹茶、竹茶、ハーブ茶、昆布茶、梅昆布茶、マテ茶、そば茶、ハブ茶、甜茶、紫蘇茶、羅漢茶、椎茸茶等の茶外茶と呼ばれる、茶樹以外から採取した原料を用いて製造される加工品も含まれる。
本発明において「天然糖」とは、Cm(H2O)nの一般式(ここでmとnは独立の自然数を表す)で表され、ヒトが消化可能なD体の炭水化物を含み、その例としてはグルコース、スクロース、フルクトース、マルトース、オリゴ糖(例えば、フラクトオリゴ糖、マルトオリゴ糖、イソマルトオリゴ糖、ガラクトオリゴ糖等)、異性化糖、乳糖、プシコース、アロース、タガトースおよびその組み合わせが挙げられる。
グルコース、スクロース、フルクトース、マルトース、オリゴ糖、異性化糖、乳糖の組み合わせの例としては、以下が挙げられる。具体的には、スクロースと異性化糖、スクロースとグルコース、スクロースとフルクトース、スクロースとマルトース、スクロースとオリゴ糖、スクロースと乳糖、異性化糖とオリゴ糖、スクロースとプシコース、スクロースと異性化糖とグルコース、スクロースと異性化糖とフルクトース、スクロースと異性化糖とマルトース、スクロースと異性化糖とオリゴ糖、スクロースと異性化糖とグルコースとオリゴ糖、スクロースとグルコースとフルクトースとオリゴ糖、グルコースとスクロースとフルクトースとマルトースとオリゴ糖と異性化糖と乳糖とタガトース、などの組み合わせが挙げられる。また、乳分由来の糖も、天然糖に含まれる。
天然糖の甘味強度X1相当の量とは、本発明の茶飲料と同等体積の20℃の水に天然糖を溶かした条件で甘味強度X1の甘味を呈する量(濃度)をいう。
本明細書において「高甘味度甘味料」とは、ショ糖に比べて強い甘味を有する化合物を意味し、天然由来化合物、合成化合物または天然由来化合物および合成化合物の組み合わせであってもよい。高甘味度甘味料はショ糖と同量において、ショ糖より5倍以上、10倍以上、50倍以上、100倍以上、500倍以上、1000倍以上、5000倍以上、10000倍以上、50000倍以上、100000倍以上の甘味を呈する。高甘味度甘味料はこのように非常に強い甘味を呈するため、本発明の茶飲料における天然糖と高甘味度甘味料の存在を重量比で表すと、「天然糖:高甘味度甘味料」は5:1~10:1、50:1~100:1、50:1~200:1、500:1~1000:1、5000:1~10000:1、50000:1~100000:1となる。
本発明は(c)50mg/100ml未満のナトリウムを含むが、これはナトリウム原子の含有量が50mg/100ml未満であることを意味する。ナトリウムの含有量は、実施態様に応じて、0.1mg/100ml以上50mg/100ml未満、0.1~45mg/100ml、0.1~40mg/100ml、0.1~35mg/100ml、0.1~30mg/100ml、0.1~25mg/100ml、0.1~20mg/100ml、0.1~19mg/100ml、0.1~18mg/100ml、0.1~17mg/100ml、0.1~16mg/100ml、0.1~15mg/100ml、0.1~14mg/100ml、0.1~13mg/100ml、0.1~12mg/100ml、0.1~11mg/100ml、0.1~10mg/100ml、1mg/100ml以上50mg/100ml未満、1~45mg/100ml、1~40mg/100ml、1~35mg/100ml、1~30mg/100ml、1~25mg/100ml、1~20mg/100ml、1~19mg/100ml、1~18mg/100ml、1~17mg/100ml、1~16mg/100ml、1~15mg/100ml、1~14mg/100ml、1~13mg/100ml、1~12mg/100ml、1~11mg/100ml、1~10mg/100ml、5mg/100ml以上50mg/100ml未満、5~45mg/100ml、5~40mg/100ml、5~35mg/100ml、5~30mg/100ml、5~25mg/100ml、5~20mg/100ml、5~19mg/100ml、5~18mg/100ml、5~17mg/100ml、5~16mg/100ml、5~15mg/100ml、5~14mg/100ml、5~13mg/100ml、5~12mg/100ml、5~11mg/100ml、5~10mg/100ml、7mg/100ml以上50mg/100ml未満、7~45mg/100ml、7~40mg/100ml、7mg/100ml以上40mg/100ml未満、7~35mg/100ml、7~30mg/100ml、7~25mg/100ml、7~20mg/100ml、7~19mg/100ml、7~18mg/100ml、7~17mg/100ml、7~16mg/100ml、7~15mg/100ml、7~14mg/100ml、7~13mg/100ml、7~12mg/100ml、7~11mg/100ml、7~10mg/100ml、10mg/100ml以上50mg/100ml未満、10~45mg/100ml、10~40mg/100ml、10~35mg/100ml、10~30mg/100ml、10~25mg/100ml、10~20mg/100ml、10~19mg/100ml、10~18mg/100ml、10~17mg/100ml、10~16mg/100ml、10~15mg/100ml、15mg/100ml以上50mg/100ml未満、15~45mg/100ml、15~40mg/100ml、15~35mg/100ml、15~30mg/100ml、15~25mg/100ml、15~20mg/100ml、17mg/100ml以上50mg/100ml未満、17~45mg/100ml、17mg/100ml以上40mg/100ml未満、17~40mg/100ml、17~35mg/100ml、17~30mg/100ml、17~25mg/100ml、17~20mg/100ml、17~19mg/100ml、17~18mg/100ml、20mg/100ml以上50mg/100ml未満、20~45mg/100ml、20~40mg/100ml、20~35mg/100ml、20~30mg/100ml、20~25mg/100ml、25mg/100ml以上50mg/100ml未満、25~45mg/100ml、25~40mg/100ml、25~35mg/100ml、25~30mg/100mlであってもよい。
本発明は(c)0.1~52mg/100mlのカリウムを含みうるが、これはカリウム原子の含有量が0.1~52mg/100mlであることを意味する。カリウムの含有量は、実施態様に応じて、0.1~50mg/100ml、0.1~45mg/100ml、0.1~40mg/100ml、0.1~35mg/100ml、0.1~32mg/100ml、0.1~30mg/100ml、0.1~25mg/100ml、0.1~21mg/100ml、0.1~20mg/100ml、0.1~15mg/100ml、0.1~10mg/100ml、0.1~5mg/100ml、0.5~52mg/100ml、0.5~50mg/100ml、0.5~45mg/100ml、0.5~40mg/100ml、0.5~35mg/100ml、0.5~30mg/100ml、0.5~25mg/100ml、0.5~21mg/100ml、0.5~20mg/100ml、0.5~15mg/100ml、0.5~10mg/100ml、0.5~5mg/100ml、1~52mg/100ml、1~50mg/100ml、1~45mg/100ml、1~40mg/100ml、1~35mg/100ml、1~32mg/100ml、1~30mg/100ml、1~25mg/100ml、1~21mg/100ml、1~20mg/100ml、1~15mg/100ml、1~10mg/100ml、1~5mg/100ml、5~52mg/100ml、5~50mg/100ml、5~45mg/100ml、5~40mg/100ml、5~35mg/100ml、5~32mg/100ml、5~30mg/100ml、5~25mg/100ml、5~21mg/100ml、5~20mg/100ml、5~15mg/100ml、5~10mg/100ml、5~5mg/100ml、10~52mg/100ml、10~50mg/100ml、10~45mg/100ml、10~40mg/100ml、10~35mg/100ml、10~32mg/100ml、10~30mg/100ml、10~25mg/100ml、10~21mg/100ml、10~20mg/100ml、10~15mg/100ml、12~52mg/120ml、12~50mg/120ml、12~45mg/120ml、12~40mg/120ml、12~35mg/120ml、12~32mg/100ml、12~30mg/120ml、12~25mg/120ml、12~21mg/100ml、12~20mg/120ml、12~15mg/120ml、15~52mg/100ml、15~50mg/100ml、15~45mg/100ml、15~40mg/100ml、15~35mg/100ml、15~32mg/100ml、15~30mg/100ml、15~25mg/100ml、15~21mg/100ml、15~20mg/100ml、20~52mg/100ml、20~50mg/100ml、20~45mg/100ml、20~40mg/100ml、20~35mg/100ml、20~32mg/100ml、20~30mg/100ml、20~25mg/100mlであってもよい。
本発明は(c)0.1~52mg/100mlのカルシウムを含みうるが、これはカルシウム原子の含有量が0.1~52mg/100mlであることを意味する。カルシウムの含有量は、実施態様に応じて、0.1~50mg/100ml、0.1~45mg/100ml、0.1~40mg/100ml、0.1~35mg/100ml、0.1~32mg/100ml、0.1~30mg/100ml、0.1~25mg/100ml、0.1~22mg/100ml、0.1~20mg/100ml、0.1~19mg/100ml、0.1~18mg/100ml、0.1~17mg/100ml、0.1~16mg/100ml、0.1~15mg/100ml、0.1~14mg/100ml、0.1~13mg/100ml、0.1~12mg/100ml、0.1~11mg/100ml、0.1~10mg/100ml、1~50mg/100ml、1~45mg/100ml、1~40mg/100ml、1~35mg/100ml、1~32mg/100ml、1~30mg/100ml、1~25mg/100ml、1~22mg/100ml、1~20mg/100ml、1~19mg/100ml、1~18mg/100ml、1~17mg/100ml、1~16mg/100ml、1~15mg/100ml、1~14mg/100ml、1~13mg/100ml、1~12mg/100ml、1~11mg/100ml、1~10mg/100ml、5~50mg/100ml、5~45mg/100ml、5~40mg/100ml、5~35mg/100ml、5~32mg/100ml、5~30mg/100ml、5~25mg/100ml、5~22mg/100ml、5~20mg/100ml、5~19mg/100ml、5~18mg/100ml、5~17mg/100ml、5~16mg/100ml、5~15mg/100ml、5~14mg/100ml、5~13mg/100ml、5~12mg/100ml、5~11mg/100ml、5~10mg/100ml、10~50mg/100ml、10~45mg/100ml、10~40mg/100ml、10~35mg/100ml、10~32mg/100ml、10~30mg/100ml、10~25mg/100ml、10~22mg/100ml、10~20mg/100ml、10~19mg/100ml、10~18mg/100ml、10~17mg/100ml、10~16mg/100ml、10~15mg/100ml、12~52mg/120ml、12~50mg/120ml、12~45mg/120ml、12~40mg/120ml、12~35mg/120ml、12~32mg/100ml、12~30mg/120ml、12~25mg/120ml、12~22mg/100ml、12~20mg/120ml、12~15mg/120ml、15~50mg/100ml、15~45mg/100ml、15~40mg/100ml、15~35mg/100ml、15~32mg/100ml、15~30mg/100ml、15~25mg/100ml、15~22mg/100ml、15~20mg/100ml、20~50mg/100ml、20~45mg/100ml、20~40mg/100ml、20~35mg/100ml、20~32mg/100ml、20~30mg/100ml、20~25mg/100ml、25~50mg/100ml、25~45mg/100ml、25~40mg/100ml、25~35mg/100ml、25~32mg/100ml、25~30mg/100mlであってもよい。
なお、飲料に配合されたカルシウム含有化合物の配合量が判明している場合には、その配合量から算出された値を用いてもよい。また、例えば、エチレンジアミン四酢酸カルシウムニナトリウムのような、ナトリウム、カリウム、及びカルシウムから選ばれる2以上の原子を含む添加剤を配合する場合、その配合量は、ナトリウム、カルシウムおよびナトリウムの量が上述の範囲となるように調製される。
さらには、上記測定において本発明の茶飲料よりも甘味が少ない基準茶飲料のうち、本発明の茶飲料と最も甘味の近い基準茶飲料を選択し、当該選択された基準茶飲料にショ糖を追加することによって本発明の茶飲料と同じ甘味を呈するように調整し、その際に、調整後の基準茶飲料に含まれるショ糖含有量から、本発明の茶飲料について甘味強度X3を測定することもできる。
(a)甘味強度X1が0.1~5相当の量の天然糖、
(b)甘味強度X2が0.1~5相当の量の高甘味度甘味料、
(c)7mg/100ml以上40mg/100ml以下のナトリウム、並びに
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3が5.5~12.5の甘味を呈する茶飲料である。
(a)甘味強度X1が3~5相当の量の天然糖、
(b)甘味強度X2が1~5相当の量の高甘味度甘味料、
(c)17mg/100ml以上40mg/100ml未満のナトリウム、並びに
(d)0.1~32mg/100mlのカリウムおよび/または0.1~32mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3が5.5~12.5の甘味を呈する茶飲料である。好ましくは、本発明の茶飲料は、さらに、エネルギーが50Kcal/100ml以下、かつ、X1+X2が6.0以上である。
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の、レバウジオシドM、レバウジオシドDおよびその組み合わせからなる群より選択される高甘味度甘味料、
(c)17mg/100ml以上40mg/100ml未満のナトリウム、および
(d)10~21mg/100mlのカリウムおよび/または10~22mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3の甘味を呈し、0.1<(X1+X2)≦20である、茶飲料。
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の羅漢果抽出物、
(c)17mg/100ml以上40mg/100ml未満のナトリウム、並びに
(d)10~21mg/100mlのカリウムおよび/または10~22mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3の甘味を呈し、0.1<(X1+X2)≦20である、茶飲料。
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量のモグロシドV、
(c)17mg/100ml以上40mg/100ml未満のナトリウム、および
(d)10~21mg/100mlのカリウムおよび/または10~22mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3の甘味を呈し、0.1<(X1+X2)≦20である、茶飲料。
(a)0.1~0.5 w/v%、0.1~1.0 w/v%、0.1~1.5 w/v%、0.1~2.0 w/v%、0.1~2.5 w/v%、0.1~3.0 w/v%、0.1~3.5 w/v%、0.1~4.0 w/v%、0.1~4.5 w/v%、0.1~5.0 w/v%、0.1~5.5 w/v%、0.1~5.9 w/v%、0.5~1.0 w/v%、0.5~1.5 w/v%、0.5~2.0 w/v%、0.5~2.5 w/v%、0.5~3.0 w/v%、0.5~3.5 w/v%、0.5~4.0 w/v%、0.5~4.5 w/v%、0.5~5.0 w/v%、0.5~5.5 w/v%、0.5~5.9 w/v%、1.0~1.5 w/v%、1.0~2.0 w/v%、1.0~2.5 w/v%、1.0~3.0 w/v%、1.0~3.5 w/v%、1.0~4.0 w/v%、1.0~4.5 w/v%、1.0~5.0 w/v%、1.0~5.5 w/v%、1.0~5.9 w/v%、1.5~2.0 w/v%、1.5~2.5 w/v%、1.5~3.0 w/v%、1.5~3.5 w/v%、1.5~4.0 w/v%、1.5~4.5 w/v%、1.5~5.0 w/v%、1.5~5.5 w/v%、1.5~5.9 w/v%、2.0~2.5 w/v%、2.0~3.0 w/v%、2.0~3.5 w/v%、2.0~4.0 w/v%、2.0~4.5 w/v%、2.0~5.0 w/v%、2.0~5.5 w/v%または2.0~5.9 w/v%の天然糖と、
(b)20~550ppm、25~550 ppm、30~550 ppm、35~550 ppm、40~550 ppm、45~550 ppm、50~550 ppm、55~550 ppm、20~540 ppm、25~540 ppm、30~540 ppm、35~540 ppm、40~540 ppm、45~540 ppm、50~540 ppm、55~540 ppm、20~530 ppm、25~530 ppm、30~530 ppm、35~530 ppm、40~530 ppm、45~530 ppm、50~530 ppm、55~530 ppm、20~520 ppm、25~520 ppm、30~520 ppm、35~520 ppm、40~520 ppm、45~520 ppm、50~520 ppm、55~520 ppm、20~510 ppm、25~510 ppm、30~510 ppm、35~510 ppm、40~510 ppm、45~510 ppm、50~510 ppm、55~510ppm、20~505ppm、25~505ppm、30~505ppm、35~505ppm、40~505ppm、45~505ppm、50~505ppm、55~505ppm、20~500ppm、25~500ppm、30~500ppm、35~500ppm、40~500ppm、45~500ppm、50~500ppm、55~500ppm、20~495ppm、25~495ppm、30~495ppm、35~495ppm、40~495ppm、45~495ppm、50~495ppm、55~495ppm、20~490ppm、25~490ppm、30~490ppm、35~490ppm、40~490ppm、45~490ppm、50~490ppmまたは55~490ppmのレバウジオシドM、レバウジオシドD、羅漢果抽出物、モグロシドVおよびその組み合わせからなる群より選択される少なくとも1つの高甘味度甘味料と、
(c)0.1mg/100ml以上50mg/100ml未満、0.1~45mg/100ml、0.1~40mg/100ml、0.1~35mg/100ml、0.1~30mg/100ml、0.1~25mg/100ml、0.1~20mg/100ml、0.1~19mg/100ml、0.1~18mg/100ml、0.1~17mg/100ml、0.1~16mg/100ml、0.1~15mg/100ml、0.1~14mg/100ml、0.1~13mg/100ml、0.1~12mg/100ml、0.1~11mg/100ml、0.1~10mg/100ml、1mg/100ml以上50mg/100ml未満、1~45mg/100ml、1~40mg/100ml、1~35mg/100ml、1~30mg/100ml、1~25mg/100ml、1~20mg/100ml、1~19mg/100ml、1~18mg/100ml、1~17mg/100ml、1~16mg/100ml、1~15mg/100ml、1~14mg/100ml、1~13mg/100ml、1~12mg/100ml、1~11mg/100ml、1~10mg/100ml、5mg/100ml以上50mg/100ml未満、5~45mg/100ml、5~40mg/100ml、5~35mg/100ml、5~30mg/100ml、5~25mg/100ml、5~20mg/100ml、5~19mg/100ml、5~18mg/100ml、5~17mg/100ml、5~16mg/100ml、5~15mg/100ml、5~14mg/100ml、5~13mg/100ml、5~12mg/100ml、5~11mg/100ml、5~10mg/100ml、7mg/100ml以上50mg/100ml未満、7~45mg/100ml、7~40mg/100ml、7mg/100ml以上40mg/100ml未満、7~35mg/100ml、7~30mg/100ml、7~25mg/100ml、7~20mg/100ml、7~19mg/100ml、7~18mg/100ml、7~17mg/100ml、7~16mg/100ml、7~15mg/100ml、7~14mg/100ml、7~13mg/100ml、7~12mg/100ml、7~11mg/100ml、7~10mg/100ml、10mg/100ml以上50mg/100ml未満、10~45mg/100ml、10~40mg/100ml、10~35mg/100ml、10~30mg/100ml、10~25mg/100ml、10~20mg/100ml、10~19mg/100ml、10~18mg/100ml、10~17mg/100ml、10~16mg/100ml、10~15mg/100ml、15mg/100ml以上50mg/100ml未満、15~45mg/100ml、15~40mg/100ml、15~35mg/100ml、15~30mg/100ml、15~25mg/100ml、15~20mg/100ml、17mg/100ml以上50mg/100ml未満、17~45mg/100ml、17~40mg/100ml、17mg/100ml以上40mg/100ml未満、17~35mg/100ml、17~30mg/100ml、17~25mg/100ml、17~20mg/100ml、17~19mg/100ml、17~18mg/100ml、20mg/100ml以上50mg/100ml未満、20~45mg/100ml、20~40mg/100ml、20~35mg/100ml、20~30mg/100ml、20~25mg/100ml、25mg/100ml以上50mg/100ml未満、25~45mg/100ml、25~40mg/100ml、25~35mg/100ml、25~30mg/100ml、0.1~22mg/100ml、0.1~21mg/100ml、1~22mg/100ml、1~21mg/100ml、4~40mg/100ml、4~35mg/100ml、4~34mg/100ml、4~33mg/100ml、4~32mg/100ml、4~31mg/100ml、4~30mg/100ml、4~29mg/100ml、4~26mg/100ml、4~25mg/100ml、4~22mg/100ml、4~21mg/100ml、4~20mg/100ml、4~19mg/100ml、4~18mg/100ml、4~17mg/100ml、4~16mg/100ml、4~15mg/100ml、4~14mg/100ml、4~13mg/100ml、4~12mg/100ml、4~11mg/100ml、4~10mg/100ml、5~34mg/100ml、5~33mg/100ml、5~32mg/100ml、5~31mg/100ml、5~29mg/100ml、5~22mg/100ml、5~21mg/100ml、10~34mg/100ml、10~33mg/100ml、10~32mg/100ml、10~31mg/100ml、10~29mg/100ml、10~22mg/100ml、10~21mg/100ml、11.5~34mg/100ml、11.5~33mg/100ml、11.5~32mg/100ml、11.5~31mg/100ml、11.5~30mg/100ml、11.5~29mg/100ml、11.5~22mg/100ml、11.5~21mg/100ml、11.5~20mg/100ml、11.5~19mg/100ml、11.5~18mg/100ml、11.5~17mg/100ml、11.5~16mg/100ml、11.5~15mg/100ml、11.5~14mg/100ml、11.5~13mg/100ml、11.5~12mg/100ml、5.75~34.5mg/100ml、5.75~28.75mg/100ml、5.75~23mg/100ml、5.75~17.25mg/100ml、5.75~11.5mg/100ml、11.5~34.5mg/100ml、11.5~28.75mg/100ml、11.5~23mg/100ml、11.5~17.25mg/100ml、17.25~34.5mg/100ml、17.25~28.75mg/100ml、17.25~23mg/100ml、23~34.5mg/100ml、23~28.75mg/100mlまたは28.75~34.5mg/100mlのナトリウムと
(d)0.1~52mg/100ml、0.1~50mg/100ml、0.1~45mg/100ml、0.1~40mg/100ml、0.1~35mg/100ml、0.1~32mg/100ml、0.1~30mg/100ml、0.1~25mg/100ml、0.1~21mg/100ml、0.1~20mg/100ml、0.1~15mg/100ml、0.1~10mg/100ml、0.1~5mg/100ml、0.5~52mg/100ml、0.5~50mg/100ml、0.5~45mg/100ml、0.5~40mg/100ml、0.5~35mg/100ml、0.5~30mg/100ml、0.5~25mg/100ml、0.5~21mg/100ml、0.5~20mg/100ml、0.5~15mg/100ml、0.5~10mg/100ml、0.5~5mg/100ml、1~52mg/100ml、1~50mg/100ml、1~45mg/100ml、1~40mg/100ml、1~35mg/100ml、1~32mg/100ml、1~30mg/100ml、1~25mg/100ml、1~21mg/100ml、1~20mg/100ml、1~15mg/100ml、1~10mg/100ml、1~5mg/100ml、5~52mg/100ml、5~50mg/100ml、5~45mg/100ml、5~40mg/100ml、5~35mg/100ml、5~32mg/100ml、5~30mg/100ml、5~25mg/100ml、5~21mg/100ml、5~20mg/100ml、5~15mg/100ml、5~10mg/100ml、5~5mg/100ml、10~52mg/100ml、10~50mg/100ml、10~45mg/100ml、10~40mg/100ml、10~35mg/100ml、10~32mg/100ml、10~30mg/100ml、10~25mg/100ml、10~21mg/100ml、10~20mg/100ml、10~15mg/100ml、12~52mg/120ml、12~50mg/120ml、12~45mg/120ml、12~40mg/120ml、12~35mg/120ml、12~32mg/100ml、12~30mg/120ml、12~25mg/120ml、12~21mg/100ml、12~20mg/120ml、12~15mg/120ml、15~52mg/100ml、15~50mg/100ml、15~45mg/100ml、15~40mg/100ml、15~35mg/100ml、15~32mg/100ml、15~30mg/100ml、15~25mg/100ml、15~21mg/100ml、15~20mg/100ml、20~52mg/100ml、20~50mg/100ml、20~45mg/100ml、20~40mg/100ml、20~35mg/100ml、20~32mg/100ml、20~30mg/100ml、20~25mg/100mlのカリウム、および/または0.1~52mg/100ml、0.1~50mg/100ml、0.1~45mg/100ml、0.1~40mg/100ml、0.1~35mg/100ml、0.1~32mg/100ml、0.1~30mg/100ml、0.1~25mg/100ml、0.1~22mg/100ml、0.1~20mg/100ml、0.1~19mg/100ml、0.1~18mg/100ml、0.1~17mg/100ml、0.1~16mg/100ml、0.1~15mg/100ml、0.1~14mg/100ml、0.1~13mg/100ml、0.1~12mg/100ml、0.1~11mg/100ml、0.1~10mg/100ml、1~50mg/100ml、1~45mg/100ml、1~40mg/100ml、1~35mg/100ml、1~32mg/100ml、1~30mg/100ml、1~25mg/100ml、1~22mg/100ml、1~20mg/100ml、1~19mg/100ml、1~18mg/100ml、1~17mg/100ml、1~16mg/100ml、1~15mg/100ml、1~14mg/100ml、1~13mg/100ml、1~12mg/100ml、1~11mg/100ml、1~10mg/100ml、5~50mg/100ml、5~45mg/100ml、5~40mg/100ml、5~35mg/100ml、5~32mg/100ml、5~30mg/100ml、5~25mg/100ml、5~22mg/100ml、5~20mg/100ml、5~19mg/100ml、5~18mg/100ml、5~17mg/100ml、5~16mg/100ml、5~15mg/100ml、5~14mg/100ml、5~13mg/100ml、5~12mg/100ml、5~11mg/100ml、5~10mg/100ml、10~50mg/100ml、10~45mg/100ml、10~40mg/100ml、10~35mg/100ml、10~32mg/100ml、10~30mg/100ml、10~25mg/100ml、10~22mg/100ml、10~20mg/100ml、10~19mg/100ml、10~18mg/100ml、10~17mg/100ml、10~16mg/100ml、10~15mg/100ml、12~52mg/120ml、12~50mg/120ml、12~45mg/120ml、12~40mg/120ml、12~35mg/120ml、12~32mg/100ml、12~30mg/120ml、12~25mg/120ml、12~22mg/100ml、12~20mg/120ml、12~15mg/120ml、15~50mg/100ml、15~45mg/100ml、15~40mg/100ml、15~35mg/100ml、15~32mg/100ml、15~30mg/100ml、15~25mg/100ml、15~22mg/100ml、15~20mg/100ml、20~50mg/100ml、20~45mg/100ml、20~40mg/100ml、20~35mg/100ml、20~32mg/100ml、20~30mg/100ml、20~25mg/100ml、25~50mg/100ml、25~45mg/100ml、25~40mg/100ml、25~35mg/100ml、25~32mg/100ml、25~30mg/100mlのカルシウムと
を含む茶飲料が提供される。
天然糖と高甘味度甘味料の配合量を低めに設定することによりエネルギー(Kcal/100ml)を低レベルに抑えた茶飲料であっても、ヒトが感知しない程度の低濃度のナトリウムに加えて低濃度のカリウムおよび/またはカルシウムを添加させることにより天然糖と高甘味度甘味料に基づく甘味を増大させることが可能である。
したがって、本発明は、さらなる別の態様として、以下の甘味が増大された茶飲料の製造方法(以下、「本発明の方法」という)を提供する。
原材料に対し
(i)(a)甘味強度X1相当の量の天然糖および(b)甘味強度X2相当の量の高甘味度甘味料を添加する工程と、
(ii)(c)飲料中のナトリウム濃度(含有量)が50mg/100ml未満となるようにナトリウムを添加する工程と、
(iii)(d)飲料中のカリウム含有量が0.1~52mg/100mlとなるようにカリウムを添加する工程および/または飲料中のカルシウム含有量が0.1~52mg/100mlとなるようにカルシウムを添加する工程と、
を含み、
前記高甘味度甘味料が、レバウジオシドM、レバウジオシドD、レバウジオシドN、レバウジオシドO、レバウジオシドE、羅漢果抽出物、モグロシドVおよびソーマチンからなる群より選択される少なくとも1つの高甘味度甘味料b1を含み、
前記成分(a)~(d)により甘味強度X3の甘味を呈し、0.1<(X1+X2)≦20である、茶飲料
を製造する方法。
(i)(a)甘味強度X1相当の量の天然糖および(b)甘味強度X2相当の量の高甘味度甘味料を添加する工程
(ii)(c)飲料中のナトリウム濃度が50mg/100ml未満となるようにナトリウムを添加する工程
(iii)(d)飲料中のカリウム含有量が0.1~52mg/100mlとなるようにカリウムを添加する工程および/または飲料中のカルシウム含有量が0.1~52mg/100mlとなるようにカルシウムを添加する工程
さらに(a)甘味強度X1相当の量の天然糖を添加する場合であっても、一度に甘味強度X1相当の量の天然糖を添加する必要はなく、数回に分けて添加してよい。同様に(b)甘味強度X2相当の量の高甘味度甘味料を添加する場合であっても、一度に甘味強度X2相当の量の天然糖を添加する必要はなく、数回に分けて添加してよい。
また、別の態様として、天然糖と高甘味度甘味料の混合物を数回に分けて添加して、最終的に製造される茶飲料に含まれる天然糖と高甘味度甘味料の量がそれぞれ甘味強度X1相当量と甘味強度X2相当量になるように調整することも可能である。
工程(ii)において原材料に加えるナトリウムは、例えば、塩化ナトリウム、水酸化ナトリウム、リンゴ酸ナトリウム、硫酸ナトリウム、クエン酸ナトリウム、リン酸ナトリウム、炭酸ナトリウム、二硫化ナトリウム、重炭酸ナトリウム、アルギン酸ナトリウム、アルギニン酸ナトリウム、グルコヘプタン酸ナトリウム、グルコン酸ナトリウム、グルタミン酸ナトリウム、酒石酸ナトリウム、アスパラギン酸ナトリウム、乳酸ナトリウム、カゼインナトリウム、アスコルビン酸ナトリウムおよびその混合物からなる群より選択される少なくとも1つの形態にあってもよい。また、茶葉の抽出物にもナトリウムが含まれている場合があるが、その際は、茶葉由来のナトリウムと追加のナトリウムの合計含有量が50mg/100ml未満となるように調整する。
工程(iii)において原材料に加えうるカリウムは、例えば、アルギン酸カリウム、塩化カリウム、クエン酸カリウム、グルコン酸カリウム、L-グルタミン酸カリウム、臭素酸カリウム、DL-酒石酸水素カリウム、L-酒石酸水素カリウム、硝酸カリウム、水酸化カリウム、ソルビン酸カリウム、炭酸カリウム、乳酸カリウム、ノルビキシンカリウム、ピロ亜硫酸カリウム、ピロリン酸四カリウム、フェロシアン化カリウム、ポリリン酸カリウム、メタリン酸カリウム、硫酸アルミニウムカリウム、硫酸カリウム、リン酸三カリウム、リン酸水素二カリウム、リン酸二水素カリウムおよびその混合物からなる群より選択される少なくとも1つの形態にあってもよい。また、茶葉の抽出物にもカリウムが含まれているため、この茶葉由来のカリウムと追加のカリウムの合計含有量が0.1~52mg/100mlとなるように調整する。また、茶飲料に乳、牛乳及び乳製品等の乳分が含まれる場合は、その乳分に含まれるカリウム含有量も考慮する。
工程(iii)において原材料に加えうるカルシウムは、例えば、L-アスコルビン酸カルシウム、アルギン酸カルシウム、エチレンジアミン四酢酸カルシウムニナトリウム、塩化カルシウム、カルボキシメチルセルロースカルシウム、クエン酸カルシウム、グリセロリン酸カルシウム、グルコン酸カルシウム、L-グルタミン酸カルシウム、ケイ酸カルシウム、酢酸カルシウム、酸化カルシウム、水酸化カルシウム、ステアリン酸カルシウム、ステアロイル乳酸カルシウム、ソルビン酸カルシウム、炭酸カルシウム、乳酸カルシウム、パントテン酸カルシウム、ピロリン酸二水素カルシウム、フェロシアン化カルシウム、プロピオン酸カルシウム、5'-リボヌクレオチドカルシウム、硫酸カルシウム、リン酸三カルシウム、リン酸一水素カルシウム、リン酸二水素カルシウムおよびその混合物からなる群より選択される少なくとも1つの形態にあってもよい。また、茶葉の抽出物や水(特に硬水)にもカルシウムが含まれる場合があるが、その場合は、この茶葉由来のカルシウムと追加のカルシウムの合計含有量が0.1~52mg/100mlとなるように調整する。また、茶飲料に乳、牛乳及び乳製品等の乳分が含まれる場合は、その乳分に含まれるカルシウム含有量も考慮する。
例えば、第1の原材料に、茶葉から水等の水性媒体中に抽出された成分以外に乳分、穀類、豆類やその抽出物が含まれ、そのために原材料に予め成分(a)~(d)のいずれか1つ以上が含まれ、第1の原材料と混合するための第2の原材料においても成分(a)~(d)が含まれ、第1および第2の原材料を混合することにより本発明の茶飲料が製造できた場合、成分(a)~(d)を個別に原材料に追加する操作は存在していないが、本発明の方法では、最終的に製造される本発明の茶飲料が(a)甘味強度X1相当の量の天然糖、(b)甘味強度X2相当の量の高甘味度甘味料、(c)50mg/100ml未満のナトリウムおよび(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウムを含む限り、工程(i)~(iii)は行われたものとみなす。
(a)甘味強度値X1が0.1~5相当の量の天然糖、
(b)甘味強度値X2が0.1~3相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、および
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3が5.5~12.5の甘味を呈し、0.2<(X1+X2)≦12.5である、茶飲料である。
(a)甘味強度X1が3~5相当の量の天然糖、
(b)甘味強度X2が1~3相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、および
(d)0.1~32mg/100mlのカリウムおよび/または0.1~32mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3が5.5~12.5の甘味を呈し、4<(X1+X2)≦12.5である、茶飲料である。
(a)甘味強度値X1が0.1~5相当の量の天然糖、
(b)甘味強度値X2が0.1~3相当の量の高甘味度甘味料、
(c)7mg/100ml以上40mg/100ml未満のナトリウム、および
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3が2.0~12.0の甘味を呈し、0.2<(X1+X2)≦12.5である、茶飲料である。
(a)甘味強度X1が3~5相当の量の天然糖、
(b)甘味強度X2が1~3相当の量の高甘味度甘味料、
(c)7 mg/100ml以上40mg/100ml未満のナトリウム、および
(d)0.1~32mg/100mlのカリウムおよび/または0.1~32mg/100mlのカルシウム
を含み、
前記成分(a)~(d)により甘味強度X3が5.5~12.0の甘味を呈し、4<(X1+X2)≦12.5である、茶飲料である。
原材料に対し
(i)(a)甘味強度X1の量の天然糖および(b)甘味強度X2の量の、レバウジオシドM、レバウジオシドDおよびその組み合わせからなる群より選択される高甘味度甘味料を添加する工程と、
(ii)(c)飲料中のナトリウム含有量が17mg/100ml以上40mg/100ml未満となるようにナトリウムを添加する工程と、
(iii)(d)飲料中のカリウム含有量が0.1~52mg/100mlとなるようにカリウムを添加する工程および/または飲料中のカルシウム含有量が0.1~52mg/100mlとなるようにカルシウムを添加する工程と、
を含む、
前記成分(a)~(d)により甘味強度X3の甘味を呈し、0.1<(X1+X2)≦20である、茶飲料
を製造する方法。
原材料に対し
(i)(a)甘味強度X1の量の天然糖および(b)甘味強度X2の量の羅漢果抽出物を添加する工程と、
(ii)(c)飲料中のナトリウム含有量が17mg/100ml以上40mg/100ml未満となるようにナトリウムを添加する工程と、
(iii)(d)飲料中のカリウム含有量が0.1~52mg/100mlとなるようにカリウムを添加する工程および/または飲料中のカルシウム含有量が0.1~52mg/100mlとなるようにカルシウムを添加する工程と、
を含む、
前記成分(a)~(d)により甘味強度X3の甘味を呈し、0.1<(X1+X2)≦20である、茶飲料
を製造する方法。
原材料に対し
(i)(a)甘味強度X1の量の天然糖および(b)甘味強度X2の量のモグロシドVを添加する工程と、
(ii)(c)飲料中のナトリウム含有量が17mg/100ml以上40mg/100ml未満となるようにナトリウムを添加する工程と、
(iii)(d)飲料中のカリウム含有量が0.1~52mg/100mlとなるようにカリウムを添加する工程および/または飲料中のカルシウム含有量が0.1~52mg/100mlとなるようにカルシウムを添加する工程と、
を含む、
前記成分(a)~(d)により甘味強度X3の甘味を呈し、0.1<(X1+X2)≦20である、茶飲料
を製造する方法。
本発明は、別の実施形態として、上記本発明の茶飲料を提供するための濃縮物(以下、「本発明の濃縮物」という)を提供する。本発明の一態様によれば、本発明の濃縮物は、
(a)甘味強度X4相当の量の天然糖、
(b)甘味強度X5相当の量の高甘味度甘味料、
(c)500mg/100ml未満のナトリウム、ならびに
(d)1~520mg/100mlのカリウムおよび/または1~520mg/100mlのカルシウム
を含み、
(X4+X5)≦200、好ましくは1<(X4+X5)≦200、より好ましくは50<(X4+X5)≦200である。
(a)甘味強度X6相当の量の天然糖、
(b)甘味強度X7相当の量の高甘味度甘味料、
(c)250mg/100ml未満のナトリウム、ならびに
(d)1~260mg/100mlのカリウムおよび/または1~260mg/100mlのカルシウム
を含み、
(X6+X7)≦100、好ましくは0.5<(X6+X7)≦100、より好ましくは20<(X6+X7)≦100である。
(a)甘味強度X8相当の量の天然糖、
(b)甘味強度X9相当の量の高甘味度甘味料、
(c)300mg/100ml未満のナトリウム、ならびに
(d)0.6~312mg/100mlのカリウムおよび/または0.6~312mg/100mlのカルシウム
を含み、
(X8+X9)≦120、好ましくは0.6<(X8+X9)≦120、より好ましくは30<(X8+X9)≦120である。
(a)甘味強度X10相当の量の天然糖、
(b)甘味強度X11相当の量の高甘味度甘味料、
(c)400mg/100ml未満のナトリウム、ならびに
(d)0.8~416mg/100mlのカリウムおよび/または0.8~416mg/100mlのカルシウム
を含み、
(X10+X11)≦160、好ましくは0.8<(X10+X11)≦160、より好ましくは40<(X10+X11)≦160である。
本発明は、別の実施形態として、茶飲料の甘味増強方法(以下、「本発明の甘味増強方法」という)を提供する。本発明の一態様によれば、本発明の甘味増強方法は、茶飲料に
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、ならびに
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含有させることを特徴とする。
実験方法
下記の表2に示した比率で天然糖{スクロース(太平洋製糖株式会社製)、グルコース(昭和産業株式会社製)}、レバウジオシドD(RebD)(純度95%以上)、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムを烏龍茶抽出液(ナトリウム含有量:10(mg/100ml)、カリウム含有量:0.2mg/100ml、カルシウム含有量:11.3mg/100ml、ポリフェノール含有量:400(ppm)、エネルギー:0(kcal/100ml))に溶解して、飲料サンプルを調製した。また、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムのいずれも添加していない溶液をサンプル1とした。
なお、表2におけるBrixは天然糖の濃度から算出し、ナトリウム含有量はグルコン酸ナトリウム添加量および茶葉由来のナトリウム含有量から算出し、カリウム含有量は塩化カリウム添加量および茶葉由来のカリウム含有量から算出し、カルシウム含有量は塩化カルシウム添加量および茶葉由来のカルシウム含有量から算出し、エネルギー(kcal/100ml)は、RebD、ナトリウム、カリウムおよびカルシウム由来の成分のエネルギーを0(kcal/100ml)として算出した。表2に示すように、サンプル1-1はNaの添加量が20mg/100mlであり、サンプル1-2はNa+Kの合計添加量が20mg/100mlであり、サンプル1-3はNa+Caの合計添加量が20mg/100mlである。
まず、各パネラーが日頃の訓練により備えている共通の味質判断基準に基づき、各飲料サンプルについて、味質ごとの「官能評価スコア」を算出した。
「官能評価スコア」は、サンプル1の味質の程度を「0」(基準)として、-3.0~+3.0の範囲でサンプル1との味質の違いを数値化したものである。「官能評価スコア」の各項目の基準としては以下のとおりである。
(官能評価スコア)
・「+3.0」:サンプル1と比べて、対象となる味質が非常に強いと感じる。
・「+2.0」:サンプル1と比べて、対象となる味質が強いと感じる。
・「+1.0」:サンプル1と比べて、対象となる味質がやや強いと感じる。
・「0」 :対象となる味質が、サンプル1と同じである。
・「-1.0」:サンプル1と比べて、対象となる味質がやや弱いと感じる。
・「-2.0」:サンプル1と比べて、対象となる味質が弱いと感じる。
・「-3.0」:サンプル1と比べて、対象となる味質が非常に弱いと感じる。
なお、上記基準に照らし、例えば、「+1.0」と「+2.0」の間の味質と判断した場合には「+1.5」と、0.5刻みでスコアをつけた。
(換算スコア)
・換算スコア「3」:官能評価スコアが+1.5以上。
・換算スコア「2」:官能評価スコアが+1.0以上+1.5未満。
・換算スコア「1」:官能評価スコアが+0.5以上+1.0未満。
・換算スコア「0」:官能評価スコアが-0.5超+0.5未満。
・換算スコア「-1」:官能評価スコアが-1.0超-0.5以下。
・換算スコア「-2」:官能評価スコアが-1.5超-1.0以下。
・換算スコア「-3」:官能評価スコアが-1.5以下。
なお、「甘さの強さ」および「フレーバーの強さ」については、換算スコア合計値が大きいほど好ましく、また、「雑味の強さ」および「塩味の強さ」については換算スコア合計値が小さいほど好ましい。
実験方法
実施例1と同様に、下記の表4に示した比率で天然糖{スクロース(太平洋製糖株式会社製)、グルコース(昭和産業株式会社製)}、レバウジオシドD(RebD)(純度95%以上)、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムを緑茶抽出液(ナトリウム含有量:6(mg/100ml)、カリウム含有量:10.7mg/100ml、カルシウム含有量:0.2mg/100ml、カテキン含有量:300(ppm)、エネルギー:0(kcal/100ml))に溶解して、飲料サンプルを調製した。また、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムのいずれも添加していない溶液をサンプル2とした。なお、表4におけるBrixは天然糖の濃度から算出し、ナトリウム含有量はグルコン酸ナトリウム濃度および茶葉由来のナトリウム含有量から算出し、カリウム含有量は塩化カリウム濃度および茶葉由来のカリウム含有量から算出し、カルシウム含有量は塩化カルシウム濃度および茶葉由来のカルシウム含有量から算出し、エネルギー(kcal/100ml)は、RebD、ナトリウム、カリウムおよびカルシウム由来の成分のエネルギーを0(kcal/100ml)として算出した。表4に示すように、サンプル2-1はNaの添加量が20mg/100mlであり、サンプル2-2はNa+Kの合計添加量が20mg/100mlであり、サンプル2-3はNa+Caの合計添加量が20mg/100mlである。
実験方法
実施例1と同様に、下記の表6に示した比率で天然糖{スクロース(太平洋製糖株式会社製)、グルコース(昭和産業株式会社製)}、レバウジオシドD(RebD)(純度95%以上)、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムを烏龍茶抽出液(ポリフェノール含有量:400(ppm)、ナトリウム含有量:10mg/100ml、カリウム含有量:0.2mg/100ml、カルシウム含有量:11.3mg/100ml、エネルギー:0(kcal/100ml))に溶解して、飲料サンプルを調製した。また、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムのいずれも添加していない溶液をサンプル3とした。なお、表6におけるBrixは天然糖の濃度から算出し、ナトリウム含有量はグルコン酸ナトリウム添加量および茶葉由来のナトリウム含有量から算出し、カリウム含有量は塩化カリウム添加量および茶葉由来のカリウム含有量から算出し、カルシウム含有量は塩化カルシウム添加量および茶葉由来のカルシウム含有量から算出し、エネルギー(kcal/100ml)はRebD、ナトリウム、カリウムおよびカルシウム由来を0(kcal/100ml)として算出した。
サンプル3-7、3-8では沈殿が生じていた。
実験方法
実施例1と同様に、下記の表8に示した比率で天然糖{スクロース(太平洋製糖株式会社製)、グルコース(昭和産業株式会社製)}、レバウジオシドD(RebD)(純度95%以上)、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムを緑茶抽出液(カテキン含有量:300(ppm)、ナトリウム含有量:6mg/100ml、カリウム含有量:10.7mg/100ml、カルシウム含有量:0.2mg/100ml、エネルギー:0(kcal/100ml))に溶解して、飲料サンプルを調製した。また、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムのいずれも添加していない溶液をサンプル4とした。なお、表8におけるBrixは天然糖の濃度から算出し、ナトリウム含有量はグルコン酸ナトリウム添加量および茶葉由来のナトリウム含有量から算出し、カリウム含有量は塩化カリウム添加量および茶葉由来のカリウム含有量から算出し、カルシウム含有量は塩化カルシウム添加量および茶葉由来のカルシウム含有量から算出し、エネルギー(kcal/100ml)はRebD、ナトリウム、カリウムおよびカルシウム由来を0(kcal/100ml)として算出した。
サンプル4-7,4-8では、沈殿が生じていた。
実験方法
実施例1と同様に、下記の表10に示した比率で天然糖{スクロース(太平洋製糖株式会社製)、グルコース(昭和産業株式会社製)}、高甘味度甘味料{モグロシドV(MogV)(純度95%以上)またはレバウジオシドM(RebM)(純度99%以上)}、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムを烏龍茶抽出液(ナトリウム含有量:10mg/100ml、カリウム含有量:0.2mg/100ml、カルシウム含有量:11.3mg/100ml、ポリフェノール含有量:400(ppm)、エネルギー:0(kcal/100ml))に溶解して、飲料サンプルを調製した。また、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムのいずれも添加していない溶液をそれぞれサンプル5(MogV含有)とサンプル5A(RebM含有)とした。なお、表10におけるBrixは天然糖の濃度から算出し、ナトリウム含有量はグルコン酸ナトリウム添加量および茶葉由来のナトリウム含有量から算出し、カリウム含有量は塩化カリウム添加量および茶葉由来のカリウム含有量から算出し、カルシウム含有量は塩化カルシウム添加量および茶葉由来のカルシウム含有量から算出し、エネルギー(kcal/100ml)はMogV、RebM、ナトリウム、カリウムおよびカルシウム由来を0(kcal/100ml)として算出した。表10に示すように、サンプル5-3はNaの添加量が20mg/100mlであり、サンプル5-1、5-4はNa+Kの合計添加量が20mg/100mlであり、サンプル5-2、5-5はNa+Caの合計添加量が20mg/100mlであった。
実験方法
実施例1と同様に、下記の表12に示した比率で天然糖{スクロース(太平洋製糖株式会社製)、グルコース(昭和産業株式会社製)}、高甘味度甘味料{モグロシドV(MogV)(純度95%以上)またはレバウジオシドM(RebM)(純度99%以上)}、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムを緑茶抽出液(ナトリウム含有量:6mg/100ml、カリウム含有量:10.7mg/100ml、カルシウム含有量:0.2mg/100ml、カテキン含有量:300(ppm)、エネルギー:0(kcal/100ml))に溶解して、飲料サンプルを調製した。また、グルコン酸ナトリウム、塩化カリウムおよび塩化カルシウムのいずれも添加していない溶液をそれぞれサンプル6(MogV含有)とサンプル6A(RebM含有)とした。なお、表12におけるBrixは天然糖の濃度から算出し、ナトリウム含有量はグルコン酸ナトリウム添加量および茶葉由来のナトリウム含有量から算出し、カリウム含有量は塩化カリウム添加量および茶葉由来のカリウム含有量から算出し、カルシウム含有量は塩化カルシウム添加量および茶葉由来のカルシウム含有量から算出し、エネルギー(kcal/100ml)はMogV、RebM、ナトリウム、カリウムおよびカルシウム由来を0(kcal/100ml)として算出した。表12に示すように、サンプル6-3はNaの添加量が20mg/100mlであり、サンプル6-1、6-4はNa+Kの合計添加量が20mg/100mlであり、サンプル6-2、6-5はNa+Caの合計添加量が20mg/100mlであった。
Claims (30)
- (a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、および
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含み、
前記高甘味度甘味料が、レバウジオシドM、レバウジオシドD、レバウジオシドN、レバウジオシドO、レバウジオシドE、羅漢果抽出物、モグロシドVおよびソーマチンからなる群より選択される少なくとも1つの高甘味度甘味料b1を含み、
0.1<(X1+X2)≦20である、茶飲料。 - ナトリウム含有量が7mg/100ml以上40mg/100ml未満である、請求項1に記載の茶飲料。
- 0.1~32mg/100mlのカリウムおよび/または0.1~32mg/100mlのカルシウムを含む、請求項1または2に記載の茶飲料。
- エネルギーが50Kcal/100ml以下である、請求項1~3のいずれか一項に記載の茶飲料。
- X1が0.1~5.9である、請求項1~4のいずれか一項に記載の茶飲料。
- ポリフェノールを200~600ppm含む、請求項1~5のいずれか一項に記載の茶飲料。
- カテキンを200~600ppm含む、請求項6に記載の茶飲料。
- 前記天然糖は、グルコース、スクロース、フルクトース、マルトース、オリゴ糖、異性化糖、乳糖、プシコース、アロース、タガトースおよびその組み合わせからなる群より選択される少なくとも1つである、請求項1~7のいずれか一項に記載の茶飲料。
- 前記高甘味度甘味料b1は、レバウジオシドM、レバウジオシドD、羅漢果抽出物、モグロシドVおよびその組み合わせからなる群より選択される少なくとも1つである、請求項1~8のいずれか一項に記載の茶飲料。
- 前記ナトリウムは、塩化ナトリウム、水酸化ナトリウム、リンゴ酸ナトリウム、硫酸ナトリウム、クエン酸ナトリウム、リン酸ナトリウム、炭酸ナトリウム、二硫化ナトリウム、重炭酸ナトリウム、アルギン酸ナトリウム、アルギニン酸ナトリウム、グルコヘプタン酸ナトリウム、グルコン酸ナトリウム、グルタミン酸ナトリウム、酒石酸ナトリウム、アスパラギン酸ナトリウム、乳酸ナトリウム、カゼインナトリウム、アスコルビン酸ナトリウムおよびその混合物からなる群より選択される少なくとも1つである、請求項1~9のいずれか一項に記載の茶飲料。
- 前記カリウムは、アルギン酸カリウム、塩化カリウム、クエン酸カリウム、グルコン酸カリウム、L-グルタミン酸カリウム、臭素酸カリウム、DL-酒石酸水素カリウム、L-酒石酸水素カリウム、硝酸カリウム、水酸化カリウム、ソルビン酸カリウム、炭酸カリウム、乳酸カリウム、ノルビキシンカリウム、ピロ亜硫酸カリウム、ピロリン酸四カリウム、フェロシアン化カリウム、ポリリン酸カリウム、メタリン酸カリウム、硫酸アルミニウムカリウム、硫酸カリウム、リン酸三カリウム、リン酸水素二カリウム、リン酸二水素カリウムおよびその混合物からなる群より選択される少なくとも1つである、請求項1~10のいずれか一項に記載の茶飲料。
- 前記カルシウムは、L-アスコルビン酸カルシウム、アルギン酸カルシウム、エチレンジアミン四酢酸カルシウムニナトリウム、塩化カルシウム、カルボキシメチルセルロースカルシウム、クエン酸カルシウム、グリセロリン酸カルシウム、グルコン酸カルシウム、L-グルタミン酸カルシウム、ケイ酸カルシウム、酢酸カルシウム、酸化カルシウム、水酸化カルシウム、ステアリン酸カルシウム、ステアロイル乳酸カルシウム、ソルビン酸カルシウム、炭酸カルシウム、乳酸カルシウム、パントテン酸カルシウム、ピロリン酸二水素カルシウム、フェロシアン化カルシウム、プロピオン酸カルシウム、5'-リボヌクレオチドカルシウム、硫酸カルシウム、リン酸三カルシウム、リン酸一水素カルシウム、リン酸二水素カルシウムおよびその混合物からなる群より選択される少なくとも1つである、請求項1~11のいずれか一項に記載の茶飲料。
- 17mg/100ml以上40mg/100ml未満のナトリウムを含み、10~21mg/100mlのカリウムおよび/または10~22mg/100mlのカルシウムを含み、エネルギーが50Kcal/100ml以下、かつX1+X2が6以上である、請求項1~12のいずれか一項に記載の茶飲料。
- 容器詰めされている、請求項1~13のいずれか一項に記載の茶飲料。
- 原材料に対し
(i)(a)甘味強度X1の量の天然糖および(b)甘味強度X2の量の高甘味度甘味料を添加する工程と、
(ii)(c)茶飲料に含まれるナトリウム含有量が50mg/100ml未満となるように、ナトリウムを添加する工程と、
(iii)(d)飲料中のカリウム含有量が0.1~52mg/100mlとなるようにカリウムを添加する工程および/または飲料中のカルシウム含有量が0.1~52mg/100mlとなるようにカルシウムを添加する工程と、
を含み、
前記高甘味度甘味料が、レバウジオシドM、レバウジオシドD、レバウジオシドN、レバウジオシドO、レバウジオシドE、羅漢果抽出物、モグロシドVおよびソーマチンからなる群より選択される少なくとも1つの高甘味度甘味料b1を含み、
0.1<(X1+X2)≦20である、茶飲料
を製造する方法。 - 前記茶飲料においてナトリウム含有量が7mg/100ml以上40mg/100ml未満である、請求項15に記載の方法。
- 前記茶飲料が、0.1~32mg/100mlのカリウムおよび/または0.1~32mg/100mlのカルシウムを含む、請求項15または16に記載の方法。
- 前記茶飲料のエネルギーが50Kcal/100ml以下である、請求項15~17のいずれか一項に記載の方法。
- X1が0.1~5.9である、請求項15~18のいずれか一項に記載の方法。
- 前記茶飲料がポリフェノールを200~600ppm含む、請求項15~19のいずれか一項に記載の方法。
- 前記茶飲料がカテキンを200~600ppm含む、請求項20に記載の方法。
- 前記天然糖は、グルコース、スクロース、フルクトース、マルトース、オリゴ糖、異性化糖、乳糖、プシコース、アロース、タガトースおよびその組み合わせからなる群より選択される少なくとも1つである、請求項15~21のいずれか一項に記載の方法。
- 前記高甘味度甘味料b1は、レバウジオシドM、レバウジオシドD、羅漢果抽出物、モグロシドVおよびその組み合わせからなる群より選択される少なくとも1つである、請求項15~22のいずれか一項に記載の方法。
- 前記ナトリウムは、塩化ナトリウム、水酸化ナトリウム、リンゴ酸ナトリウム、硫酸ナトリウム、クエン酸ナトリウム、リン酸ナトリウム、炭酸ナトリウム、二硫化ナトリウム、重炭酸ナトリウム、アルギン酸ナトリウム、アルギニン酸ナトリウム、グルコヘプタン酸ナトリウム、グルコン酸ナトリウム、グルタミン酸ナトリウム、酒石酸ナトリウム、アスパラギン酸ナトリウム、乳酸ナトリウム、カゼインナトリウム、アスコルビン酸ナトリウムおよびその混合物からなる群より選択される少なくとも1つである、請求項15~23のいずれか一項に記載の方法。
- 前記カリウムは、アルギン酸カリウム、塩化カリウム、クエン酸カリウム、グルコン酸カリウム、L-グルタミン酸カリウム、臭素酸カリウム、DL-酒石酸水素カリウム、L-酒石酸水素カリウム、硝酸カリウム、水酸化カリウム、ソルビン酸カリウム、炭酸カリウム、乳酸カリウム、ノルビキシンカリウム、ピロ亜硫酸カリウム、ピロリン酸四カリウム、フェロシアン化カリウム、ポリリン酸カリウム、メタリン酸カリウム、硫酸アルミニウムカリウム、硫酸カリウム、リン酸三カリウム、リン酸水素二カリウム、リン酸二水素カリウムおよびその混合物からなる群より選択される少なくとも1つである、請求項15~24のいずれか一項に記載の方法。
- 前記カルシウムは、L-アスコルビン酸カルシウム、アルギン酸カルシウム、エチレンジアミン四酢酸カルシウムニナトリウム、塩化カルシウム、カルボキシメチルセルロースカルシウム、クエン酸カルシウム、グリセロリン酸カルシウム、グルコン酸カルシウム、L-グルタミン酸カルシウム、ケイ酸カルシウム、酢酸カルシウム、酸化カルシウム、水酸化カルシウム、ステアリン酸カルシウム、ステアロイル乳酸カルシウム、ソルビン酸カルシウム、炭酸カルシウム、乳酸カルシウム、パントテン酸カルシウム、ピロリン酸二水素カルシウム、フェロシアン化カルシウム、プロピオン酸カルシウム、5'-リボヌクレオチドカルシウム、硫酸カルシウム、リン酸三カルシウム、リン酸一水素カルシウム、リン酸二水素カルシウムおよびその混合物からなる群より選択される少なくとも1つである、請求項15~25のいずれか一項に記載の方法。
- 前記茶飲料が、17mg/100ml以上40mg/100ml未満のナトリウムを含み、10~21mg/100mlのカリウムおよび/または10~22mg/100mlのカルシウムを含み、エネルギーが50Kcal/100ml以下、かつX1+X2が6以上である、請求項15~26のいずれか一項に記載の方法。
- Camellia sinensisの茶葉を使用する、請求項15~27のいずれか一項に記載の方法。
- (a)甘味強度X4相当の量の天然糖、
(b)甘味強度X5相当の量の高甘味度甘味料、
(c)500mg/100ml未満のナトリウム、および
(d)1~520mg/100mlのカリウムおよび/または1~520mg/100mlのカルシウム
を含み、
(X4+X5)≦200である、請求項1に記載の茶飲料を提供するための濃縮物。 - 茶飲料に
(a)甘味強度X1相当の量の天然糖、
(b)甘味強度X2相当の量の高甘味度甘味料、
(c)50mg/100ml未満のナトリウム、および
(d)0.1~52mg/100mlのカリウムおよび/または0.1~52mg/100mlのカルシウム
を含有させることを特徴とする、茶飲料の甘味増強方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2019393558A AU2019393558A1 (en) | 2018-12-07 | 2019-12-06 | Tea beverage in which quality of taste resulting from sugars and sweeteners is improved |
| CN201980080662.5A CN113163784A (zh) | 2018-12-07 | 2019-12-06 | 糖及甜味剂所呈现的味质得到改善的茶饮料 |
| US17/299,965 US20220015384A1 (en) | 2018-12-07 | 2019-12-06 | Tea beverage in which quality of taste resulting from sugars and sweeteners is improved |
| EP19892252.8A EP3892102A4 (en) | 2018-12-07 | 2019-12-06 | TEA DRINK IN WHICH THE TASTE QUALITY OF A SUGAR AND A SWEETENER IS IMPROVED |
| SG11202105908SA SG11202105908SA (en) | 2018-12-07 | 2019-12-06 | Tea beverage in which quality of taste resulting from sugars and sweeteners is improved |
| JP2020560056A JP7385599B2 (ja) | 2018-12-07 | 2019-12-06 | 糖および甘味料の呈する味質が改善した茶飲料 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018-230340 | 2018-12-07 | ||
| JP2018230340 | 2018-12-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020116628A1 true WO2020116628A1 (ja) | 2020-06-11 |
Family
ID=70973483
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/047868 WO2020116628A1 (ja) | 2018-12-07 | 2019-12-06 | 糖および甘味料の呈する味質が改善した茶飲料 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20220015384A1 (ja) |
| EP (1) | EP3892102A4 (ja) |
| JP (1) | JP7385599B2 (ja) |
| CN (1) | CN113163784A (ja) |
| AU (1) | AU2019393558A1 (ja) |
| SG (1) | SG11202105908SA (ja) |
| WO (1) | WO2020116628A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022145482A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
| WO2022145483A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
| WO2022145481A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
| WO2022145484A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023215812A1 (en) * | 2022-05-04 | 2023-11-09 | The Coca-Cola Company | Tea beverages with improved taste |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008017834A (ja) * | 2006-06-16 | 2008-01-31 | Kao Corp | 容器詰飲料 |
| JP2010521164A (ja) * | 2007-03-14 | 2010-06-24 | ザ コンセントレイト マニュファクチャリング カンパニー オブ アイルランド | 甘味付けされた飲料 |
| JP2014082960A (ja) * | 2012-10-22 | 2014-05-12 | Inabata Koryo Kk | ポリフェノール含有飲料の苦渋味マスキング剤及び該飲料の苦渋味のマスキング方法 |
| WO2018044588A1 (en) * | 2016-08-29 | 2018-03-08 | Pepsico, Inc. | Compositions comprising rebaudioside j |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8029846B2 (en) * | 2007-03-14 | 2011-10-04 | The Concentrate Manufacturing Company Of Ireland | Beverage products |
| US20080226773A1 (en) * | 2007-03-14 | 2008-09-18 | Concentrate Manufacturing Company Of Ireland | Beverage Sweetened with Rebaudioside A |
| CN103564379A (zh) * | 2012-07-18 | 2014-02-12 | 成都华高瑞甜科技有限公司 | 复合甜味剂和制法、及增强甜菊苷甜度的方法 |
-
2019
- 2019-12-06 SG SG11202105908SA patent/SG11202105908SA/en unknown
- 2019-12-06 JP JP2020560056A patent/JP7385599B2/ja active Active
- 2019-12-06 EP EP19892252.8A patent/EP3892102A4/en active Pending
- 2019-12-06 WO PCT/JP2019/047868 patent/WO2020116628A1/ja unknown
- 2019-12-06 US US17/299,965 patent/US20220015384A1/en active Pending
- 2019-12-06 AU AU2019393558A patent/AU2019393558A1/en active Pending
- 2019-12-06 CN CN201980080662.5A patent/CN113163784A/zh active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008017834A (ja) * | 2006-06-16 | 2008-01-31 | Kao Corp | 容器詰飲料 |
| JP2010521164A (ja) * | 2007-03-14 | 2010-06-24 | ザ コンセントレイト マニュファクチャリング カンパニー オブ アイルランド | 甘味付けされた飲料 |
| JP2014082960A (ja) * | 2012-10-22 | 2014-05-12 | Inabata Koryo Kk | ポリフェノール含有飲料の苦渋味マスキング剤及び該飲料の苦渋味のマスキング方法 |
| WO2018044588A1 (en) * | 2016-08-29 | 2018-03-08 | Pepsico, Inc. | Compositions comprising rebaudioside j |
Non-Patent Citations (9)
| Title |
|---|
| "Food composition table 2016; 7th ed.", 1 April 2016, WOMEN'S NUTRITION UNIVERSITY PRESS, JP, ISBN: 978-4-7895-1016-5, article KAGAWA, YOSHIKO: "Passage; Coffee and cocoa", pages: 210 - 213, XP009528697 * |
| AYUMI UCHIDANAO TAKAGIRIEKO HORIKIRIMIHO MATSUEYUMIKO UCHIYAMAMASASHI OMORI, RESEARCH BULLETIN OF OTSUMA WOMEN'S UNIVERSITY FOR HOME ECONOMICS -NO.49, 2013 |
| FERNSTROM J. D.MUNGER S. D.SCLAFANI A.DE ARAUJO I. E.ROBERTS A.MOLINARY S., J. NUTR., vol. 142, 2012, pages 1134S - 1141S |
| KASAIKANEDATANAKAYAMASAKISAKAMOTOMORIMOTOOKADAKITAHATAFURUKAWA: "Sweet Diterpene-Glycoside of Leaves of Stevia rebaudiana Bertoni Synthesis and Structure-Sweetness Relationship of Rebaudiosides-A, -D, -E and Their Related glycosides", NIPPON KAGAKU KAISHI, vol. 5, 1981, pages 726 - 735 |
| LI XSTASZEWSKI LXU HDURICK KZOLLER MADLER E., PROC NATL ACAD SCI U S A., vol. 99, no. 7, 2002, pages 4692 - 4696 |
| MURATA Y. ET AL., NIPPON SHOKUHIN KAGAKU KOGAKU KAISHI, vol. 53, no. 10, 2006, pages 527 - 533 |
| See also references of EP3892102A4 |
| TOYOTA ET AL.: "Construction of a Screening Test for Gustatory Function in Four Basic Tastes", THE JOURNAL OF JAPANESE SOCIETY OF STOMATOGNATHIC FUNCTION, vol. 20, 2014, pages 115 - 129 |
| ZHAO G. Q.ZHANG Y.HOON M. A.CHANDRASHEKAR J.ERLENBACH I.RYBA N. J. P.ZUKERL C. S., CELL, vol. 115, 2003, pages 255 - 266 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022145482A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
| WO2022145483A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
| WO2022145481A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
| WO2022145484A1 (ja) | 2020-12-28 | 2022-07-07 | サントリーホールディングス株式会社 | 甘味の増大した経口組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP7385599B2 (ja) | 2023-11-22 |
| SG11202105908SA (en) | 2021-07-29 |
| JPWO2020116628A1 (ja) | 2021-10-21 |
| CN113163784A (zh) | 2021-07-23 |
| AU2019393558A1 (en) | 2021-07-01 |
| US20220015384A1 (en) | 2022-01-20 |
| EP3892102A1 (en) | 2021-10-13 |
| EP3892102A4 (en) | 2022-08-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7385599B2 (ja) | 糖および甘味料の呈する味質が改善した茶飲料 | |
| JP7385598B2 (ja) | 糖および甘味料の呈する味質が改善した茶飲料 | |
| JP7486433B2 (ja) | 糖および甘味料の呈する味質が改善したコーヒー飲料 | |
| TWI412326B (zh) | Containers | |
| JP7486432B2 (ja) | 糖および甘味料の呈する味質が改善したコーヒー飲料 | |
| WO2020116634A1 (ja) | 糖および甘味料の呈する味質が改善したフレーバーウォーター | |
| MX2012014810A (es) | Producto que comprende estevia. | |
| JPWO2020116641A1 (ja) | 糖および甘味料の呈する味質が改善した発泡性飲料 | |
| WO2020116627A1 (ja) | 糖および甘味料の呈する味質が改善したフレーバーウォーター | |
| JPWO2020116639A1 (ja) | 糖および甘味料の呈する味質が改善した発泡性飲料 | |
| WO2020116633A1 (ja) | 糖および甘味料の呈する味質が改善した果汁飲料 | |
| WO2020116626A1 (ja) | 糖および甘味料の呈する味質が改善した果汁飲料 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19892252 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020560056 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2019393558 Country of ref document: AU Date of ref document: 20191206 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019892252 Country of ref document: EP Effective date: 20210707 |