WO2020004649A1 - 被膜形成組成物 - Google Patents
被膜形成組成物 Download PDFInfo
- Publication number
- WO2020004649A1 WO2020004649A1 PCT/JP2019/025927 JP2019025927W WO2020004649A1 WO 2020004649 A1 WO2020004649 A1 WO 2020004649A1 JP 2019025927 W JP2019025927 W JP 2019025927W WO 2020004649 A1 WO2020004649 A1 WO 2020004649A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- acid
- extract
- group
- hair
- comparative example
- Prior art date
Links
- 0 C*C(C(*)=O)NC(C(*)*C(*)=O)=C Chemical compound C*C(C(*)=O)NC(C(*)*C(*)=O)=C 0.000 description 2
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/362—Polycarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/042—Gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/361—Carboxylic acids having more than seven carbon atoms in an unbroken chain; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/365—Hydroxycarboxylic acids; Ketocarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/02—Preparations for cleaning the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/12—Preparations containing hair conditioners
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/04—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing only normal peptide links
- C07K5/06—Dipeptides
- C07K5/06008—Dipeptides with the first amino acid being neutral
- C07K5/06017—Dipeptides with the first amino acid being neutral and aliphatic
- C07K5/06026—Dipeptides with the first amino acid being neutral and aliphatic the side chain containing 0 or 1 carbon atom, i.e. Gly or Ala
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D177/00—Coating compositions based on polyamides obtained by reactions forming a carboxylic amide link in the main chain; Coating compositions based on derivatives of such polymers
- C09D177/04—Polyamides derived from alpha-amino carboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q1/00—Make-up preparations; Body powders; Preparations for removing make-up
- A61Q1/02—Preparations containing skin colorants, e.g. pigments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/06—Preparations for styling the hair, e.g. by temporary shaping or colouring
Definitions
- the present invention relates to a film-forming composition, and more particularly, to a film-forming composition containing at least one lipid peptide type compound.
- a film formed on the skin or on the surface of the hair provides an effective barrier on the skin or on the surface of the hair, and thus suppresses evaporation of moisture in the skin or the hair, It plays various important roles in personal care products, such as enhancing the retention of active ingredients.
- ⁇ ⁇ ⁇ Recent health awareness, especially dry skin awareness, has led to a demand for a cosmetic composition having a high moisturizing effect.
- skin care preparations that exhibit a high moisturizing effect include coating cosmetics having a self-organized structure, such as those using a layered ⁇ -gel (Patent Document 1).
- the interlamellar lipids in the stratum corneum which is the outermost layer of the skin, also have a self-assembled lamellar structure, which suppresses invasion of substances from the outside and evaporation of moisture from the inside into the skin. It has the function of keeping moisturizing properties and skin flexibility.
- Patent Documents 2 and 3 hair preparations using polypeptides have been proposed.
- JP 2016-6030 A JP-A-10-77210 JP-A-2002-308756
- the layered ⁇ -gel In order to form the layered ⁇ -gel, it is necessary to mix a plurality of components at a specific ratio. Therefore, when the layered ⁇ -gel is used in combination with another component, the plurality of components and the other component are mixed with each other. Adjustment of the mixing ratio may be complicated. In addition, when a polymer such as a polypeptide is used as a film-forming agent, there is a sense of sharpness peculiar to the polymer.
- An object of the present invention is to provide a composition that can more easily form a film having excellent moisture retention. Further, an object of the present invention is to provide a composition capable of forming a coating film which has almost no feeling of crispness and has improved usability.
- the present inventors have found that a film is formed on the skin or the hair surface only by using at least one kind of lipid peptide type compound. 1. Excellent moisture retention. 2. suppression of moisture adsorption; 3. Promote penetration into the hair; 4. Adsorption of negatively charged components; The inventors have found that coalescence of oil components is suppressed, and have completed the present invention. In addition, the present inventors have found that the above-mentioned coating film has almost no crispness and the like, and has a good feeling of use, and thus completed the present invention.
- the present invention as a first aspect, relates to a film-forming composition
- a film-forming composition comprising at least one lipid peptide-type compound comprising a low-molecular lipid peptide or a pharmaceutically usable salt thereof.
- the lipid peptide-type compound is a compound in which a peptide portion having a repeating amino acid bonding structure is bonded to a lipid portion comprising an aliphatic group having 9 to 23 carbon atoms.
- the lipid peptide-type compound comprises at least one of the compounds represented by the following formulas (1) to (3) or a pharmaceutically usable salt thereof, A film-forming composition according to the second aspect.
- R 1 represents an aliphatic group having 9 to 23 carbon atoms
- R 2 represents a hydrogen atom or an alkyl group having 1 to 4 carbon atoms which may have a branched chain having 1 or 2 carbon atoms
- R 3 represents a — (CH 2 ) n —X group, n represents a number of 1 to 4, and X represents 1 to 3 amino groups, guanidino groups, —CONH 2 groups, or 1 to 3 nitrogen atoms.
- R 4 represents an aliphatic group having 9 to 23 carbon atoms
- R 5 to R 7 each independently represent a hydrogen atom or a carbon atom which may have a branched chain having 1 or 2 carbon atoms.
- R 8 represents an aliphatic group having 9 to 23 carbon atoms
- R 9 to R 12 each independently represent a hydrogen atom or a carbon atom which may have a branched chain having 1 or 2 carbon atoms.
- the present invention relates to a method for producing a film, comprising using the film-forming composition according to any one of the first to third aspects.
- the present invention relates to a film formed using the film-forming composition according to any one of the first to third aspects.
- a film excellent in moisture retention can be formed by containing at least one lipid peptide type compound. That is, since it is not necessary to adjust the ratio of each component in the composition excessively, it is possible to easily form a film having excellent moisture retention. Further, according to the present invention, when blended in cosmetics or the like, excellent moisture retention can be imparted to cosmetics. Furthermore, according to the present invention, it is possible to form a coating film having a good feeling of use with almost no feeling of snapping.
- the effect of suppressing the reflection of light, the effect of having water repellency, the effect of having fingerability and combability, the effect of preventing makeup twist, and the luster are imparted.
- the film having at least one or more of the following effects can be formed.
- the composition of the present invention has high biosafety and is particularly useful for pharmaceuticals and cosmetics. Useful.
- FIG. 1 is a scanning electron microscope (SEM) image in Example 1. [(a) SEM image immediately after setting a sample on a scanning electron microscope (numbers in the figure are representative mineral oil droplets) Position)). (B) SEM image of the dried sample (numbers in the figure represent the positions of the mineral oil droplets shown in (a)). ]
- FIG. 2 is a photograph of the appearance of Example 3 and Comparative Example 3 [(a) is a photograph of the appearance of Example 3. (B) is an appearance photograph of Comparative Example 3. ].
- FIG. 3 is a scanning electron microscope (SEM) image in Example 3 and Comparative Example 3. [(a) SEM image of Example 3. (B) SEM image of Comparative Example 3. ].
- FIG. 1 is a scanning electron microscope (SEM) image in Example 1. [(a) SEM image immediately after setting a sample on a scanning electron microscope (numbers in the figure are representative mineral oil droplets) Position)). (B) SEM image of the dried sample (numbers in the figure
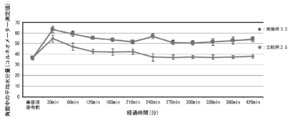
- FIG. 4 is a diagram showing the results of shampoo moisture retention tests (hair moisture retention) in Examples 4 to 8 and Comparative Example 4.
- FIG. 5 is a diagram showing the results of the contact angle measurement in Example 9 and Comparative Example 5 [(a) is a diagram showing the results of the contact angle measurement in Example 9; (B) It is a figure which shows the result of the contact angle measurement of the comparative example 5. ].
- FIG. 6 is a diagram showing the results (moisture retention of hair) of the conditioner moisture retention test in Example 10 and Comparative Example 6.
- FIG. 7 is a scanning electron microscope (SEM) image of the hair after the conditioner moisture retention test in Example 10 and Comparative Example 6. [(a) SEM image of Example 10. (B) SEM image of Comparative Example 6. ] FIG.
- SEM scanning electron microscope
- FIG. 8 is a diagram showing measurement results of frictional resistance in Example 11 and Comparative Example 7.
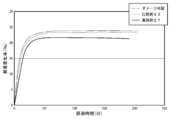
- FIG. 9 is a diagram showing the results of the reflected light intensity measurement in Example 13 and Comparative Example 9.
- FIG. 10 is a photograph of the appearance of Example 14 and Comparative Example 10. [(a) A photograph of the appearance of Comparative Example 10. (B) A photograph of the appearance of Example 14. ].
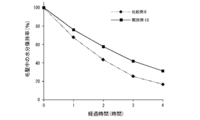
- FIG. 11 is a diagram showing the results of quantification of malic acid in Example 18 and Comparative Example 14.
- FIG. 12 is a scanning electron microscope (SEM) image of Example 19, Comparative Example 15, and the hair after the conditioner treatment on the damaged hair [(a) SEM image of damaged hair. (B) SEM image of Example 19. (C) An SEM image of Comparative Example 15. ]
- SEM scanning electron microscope
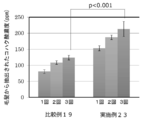
- FIG. 13 is a diagram showing the results of the dynamic friction coefficient measurement in Example 21 and Comparative Example 17.
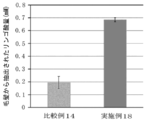
- FIG. 14 is a diagram showing the results of quantifying the amount of succinic acid in the conditioner-treated hair in Example 23 and Comparative Example 19.
- FIG. 15 is a graph showing the results of evaluation of the amounts of succinic acid and lipid peptides in human hair in Example 24 and Comparative Example 20.
- FIG. 16 is a diagram showing the evaluation results of the hardness of the conditioner-treated hair surface of Example 26, Comparative Example 22, and black and damaged hair.
- FIG. 17 is a diagram showing the results of evaluating water absorption and desorption after conditioner treatment on Example 27, Comparative Example 23, and damaged hair.
- FIG. 18 is a diagram showing the results of observing the time-dependent change of shine due to sebum floating in Example 28 and Comparative Example 24.
- FIG. 19 is a diagram illustrating the moisturizing effect of the serum in Example 30 and Comparative Example 26.
- FIG. 20 is a diagram showing the results of quantifying the amount of keratin in the hair treated with the conditioner in Example 33, Example 34, and Comparative Example 28.
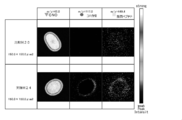
- FIG. 21 is a scanning electron microscope (SEM) image of the hair of Example 36, Comparative Example 30, and damaged hair after the conditioner treatment [(a) SEM image of Example 36. (B) SEM image of Comparative Example 30. (C) SEM image of damaged hair. ]
- the present invention relates to a film-forming composition
- a film-forming composition comprising at least one lipid peptide-type compound comprising a low-molecular lipid peptide or a pharmaceutically usable salt thereof.
- the film-forming composition according to the present invention means all those which bring industrial utility value by utilizing film-forming properties, and in particular, for forming a film on the surface of skin or hair which is a film-forming object. It means a composition, but does not limit the application to paints, coating materials, sealers, primers, correction fluids and the like.
- the above-mentioned lipid peptide-type compound is preferably a compound in which a peptide portion having a repeating bonding structure of amino acids is bonded to a lipid portion composed of an aliphatic group having 9 to 23 carbon atoms.
- lipid peptide-type compound examples include compounds (lipid peptides) represented by the following formulas (1) to (3) or a pharmaceutically usable salt thereof (a lipid portion which is a hydrophobic portion and a hydrophilic portion). Or a low molecular weight compound having a peptide moiety as a site).
- R 1 represents an aliphatic group having 9 to 23 carbon atoms
- R 1 is a linear aliphatic group having 11 to 23 carbon atoms which can have 0 to 2 unsaturated bonds.
- Specific examples of the lipid moiety (acyl group) composed of R 1 and an adjacent carbonyl group include lauroyl, dodecylcarbonyl, myristoyl, tetradecylcarbonyl, palmitoyl, margaroyl, oleoyl, and elideyl.
- R 2 contained in the peptide moiety represents a hydrogen atom or an alkyl group having 1 to 4 carbon atoms and having a branched chain having 1 or 2 carbon atoms.
- the alkyl group having 1 to 4 carbon atoms which may have a branched chain having 1 or 2 carbon atoms means that the main chain has 1 to 4 carbon atoms and the branched chain having 1 or 2 carbon atoms.
- alkyl group which may have, and specific examples thereof include a methyl group, an ethyl group, an n-propyl group, an i-propyl group, an n-butyl group, an i-butyl group, a sec-butyl group and a tert-butyl group. And the like.
- R 2 is preferably a hydrogen atom or an alkyl group having 1 to 3 carbon atoms which may have a branched chain having 1 carbon atom, and more preferably a hydrogen atom.
- An alkyl group having 1 to 3 carbon atoms which may have a branched chain having 1 carbon atom means an alkyl group having 1 to 3 carbon atoms in the main chain and having a branched chain having 1 carbon atom.
- methyl group an ethyl group, an n-propyl group, an i-propyl group, an i-butyl group and a sec-butyl group, and preferably a methyl group, an i-propyl group, It is an i-butyl group or a sec-butyl group.
- R 3 represents a — (CH 2 ) nX group.
- n represents a number of 1 to 4
- X represents an amino group, a guanidino group, a —CONH 2 group, or a 5-membered ring having 1 to 3 nitrogen atoms or Represents a 6-membered ring or a fused heterocyclic ring composed of a 5-membered ring and a 6-membered ring.
- X is preferably an amino group, a guanidino group, a carbamoyl group (—CONH 2 group), a pyrrole group, an imidazole group, a pyrazole group, or an indole group. Preferably it is an imidazole group.
- n is preferably 1 or 2, and more preferably 1.
- the — (CH 2 ) n- group is preferably an aminomethyl group, a 2-aminoethyl group, a 3-aminopropyl group, a 4-aminobutyl group, a carbamoylmethyl group, a 2-carbamoylethyl group, or a 3-carbamoyl group.
- a lipid peptide particularly suitable as a lipid peptide type compound is a compound formed from the following lipid part and peptide part (amino acid assembly part).
- abbreviations of amino acids include alanine (Ala), asparagine (Asn), glutamine (Gln), glycine (Gly), histidine (His), isorosine (Ile), leucine (Leu), and lysine (Lysine). Lys), tryptophan (Trp), and valine (Val).
- Lauroyl-Gly-His Lauroyl-Gly-Gln, Lauroyl-Gly-Asn, Lauroyl-Gly-Trp, Lauroyl-Gly-Lys, Lauroyl-Ala-His, Lauroyl-Ala-Gln, Lauroyl-Ala-Asn, Lauroyl -Ala-Trp, Lauroyl-Ala-Lys; Myristoyl-Gly-His, Myristoyl-Gly-Gln, Myristoyl-Gly-Asn, Myristoyl-Gly-Trp, Myristoyl-Gly-Lys, Myristoyl-Ala-His, Myristoyl-Ala -Gln, myristoyl-Ala-Asn, myristoyl-Ala-Trp, myristoyl-Ala-Lys; palmitoyl-Gly-His, palmitoyl-Gly-Gln, palmi
- lauroyl-Gly-His lauroyl-Ala-His, myristoyl-Gly-His, myristoyl-Ala-His; palmitoyl-Gly-His, palmitoyl-Ala-His; stearoyl-Gly-His, stearoyl-Ala. -His.
- R 4 represents an aliphatic group having 9 to 23 carbon atoms, and specific preferred examples thereof include the same groups as those defined for R 1 above.
- R 5 to R 7 are each independently a hydrogen atom, an alkyl group having 1 to 4 carbon atoms, which may have a branched chain having 1 or 2 carbon atoms, or-( CH 2 ) nX, and at least one of R 5 to R 7 represents a — (CH 2 ) nX group.
- n a number from 1 to 4
- X represents an amino group, a guanidino group, a —CONH 2 group, or a 5- or 6-membered ring having 1 to 3 nitrogen atoms, or a 5- and 6-membered ring Represents a fused heterocyclic ring composed of
- preferred specific examples of R 5 to R 7 include the same groups as defined for R 2 and R 3 above.
- a preferred lipid peptide is a compound formed from the following lipid part and peptide part (amino acid assembly part). Lauroyl-Gly-Gly-His, Myristoyl-Gly-Gly-His, Myristoyl-Gly-Gly-Gln, Myristoyl-Gly-Gly-Asn, Myristoyl-Gly-Gly-Trp, Myristoyl-Gly-Gly-Lys, Myristoyl Gly-Ala-His, Myristoyl-Gly-Ala-Gln, Myristoyl-Gly-Ala-Asn, Myristoyl-Gly-Ala-Trp, Myristoyl-Gly-Ala-Lys, Myristoyl-Aly-Gly-His, Myristoyl-Ala- Gly-Gln, myristoyl-Ala-Gly-Asn, myristoyl-Ala-Asn, myristoyl-A
- lauroyl-Gly-Gly-His myristoyl-Gly-Gly-His, palmitoyl-Gly-Gly-His, palmitoyl-Gly-His-Gly, palmitoyl-His-Gly-Gly, stearoyl. -Gly-Gly-His.
- R 8 represents an aliphatic group having 9 to 23 carbon atoms, and preferred specific examples include the same groups as defined for R 1 above.
- R 9 to R 12 each independently represent a hydrogen atom, an alkyl group having 1 to 4 carbon atoms, which may have a branched chain having 1 or 2 carbon atoms, or-( CH 2 ) nX, and at least one of R 9 to R 12 represents a — (CH 2 ) nX group.
- n a number from 1 to 4
- X represents an amino group, a guanidino group, a —CONH 2 group, or a 5- or 6-membered ring having 1 to 3 nitrogen atoms, or a 5- and 6-membered ring Represents a fused heterocyclic ring composed of
- preferred specific examples of R 9 to R 12 include the same groups as defined for R 2 and R 3 above.
- preferred lipid peptide-type compounds include lauroyl-Gly-Gly-Gly-His, myristoyl-Gly-Gly-Gly-His, and palmitoyl-Gly-Gly-Gly-Gly-His. , Palmitoyl-Gly-Gly-His-Gly, palmitoyl-Gly-His-Gly-Gly, palmitoyl-His-Gly-Gly-Gly, stearoyl-Gly-Gly-Gly-Gly-Gly-His and the like.
- the content of the lipid peptide type compound is, for example, 0.01 to 30% by mass, preferably 0.02 to 10% by mass, more preferably 0.03% by mass, based on the total mass of the film-forming composition. To 5% by mass.
- the lipid peptide type compound at least one of the compounds (lipid peptides) represented by the above formulas (1) to (3) or pharmaceutically usable salts thereof may be used. More preferably, these compounds can be used alone or in combination of two or more.
- composition of the present invention may contain water, alcohol, polyhydric alcohol, or a mixed solution thereof in addition to at least one kind of the above-mentioned lipid peptide type compound.
- Examples of the water include purified water, purified water, hard water, soft water, natural water, deep sea water, electrolytic alkaline ionized water, electrolytic acidic ionized water, ionized water, and cluster water.
- the alcohol is a monohydric alcohol, for example, an alcohol having 1 to 6 carbon atoms which is dissolved in water at an arbitrary ratio, specifically, methanol, ethanol, 2-propanol, i-butanol and the like, and higher alcohols. Alcohol, specifically, oleyl alcohol, phenoxy alcohol and the like can be mentioned.
- the polyhydric alcohol is a dihydric or higher alcohol, such as propylene glycol, 1,3-butanediol, 2-ethyl-1,3-hexanediol, glycerin, isopentyldiol, ethylhexanediol, and erythrulose.
- a polyhydric alcohol when a polyhydric alcohol is contained, its content can be, for example, 1% by mass to 60% by mass, and preferably 1% by mass to 30% by mass, based on the total mass of the composition. In the present invention, when a polyhydric alcohol is contained, the polyhydric alcohol can be used alone or in combination of two or more.
- composition of the present invention can contain, if necessary, a cosmetic additive, a quasi-drug additive, an additive usable as a pharmaceutical additive, and the like.
- additional components such as physiologically active substances and functional substances to be added to skin external preparations such as cosmetics, quasi-drugs or pharmaceuticals include, for example, pigments, oily bases, humectants, feel improvers, surfactants other than the above.
- Agents polymers, thickening / gelling agents, solvents, antioxidants, reducing agents, oxidizing agents, preservatives, antibacterial agents, bactericides, chelating agents, pH adjusters, acids, alkalis, powders, inorganic salts, UV absorbers, whitening agents, vitamins and their derivatives, hair growth agents, blood circulation promoters, stimulants, hormones, anti-wrinkles, anti-aging agents, tightening agents, cooling agents, warming agents, promoting wound healing Agent, stimulant, analgesic, cell activator, plant / animal / microbial extract, antipruritic, exfoliating / dissolving agent, antiperspirant, freshener, astringent, enzyme, nucleic acid, fragrance, pigment, coloring agent, Dye, anti-inflammatory, anti-inflammatory, anti-asthma, anti-chronic obstructive pulmonary disease, anti-allergy, immune Modifiers, anti-infective agents and antifungal agents.
- the pigment examples include inorganic white pigments such as titanium dioxide and zinc oxide; inorganic red pigments such as red iron oxide (iron oxide) and iron titanate; inorganic brown pigments such as ⁇ -iron oxide; Inorganic yellow pigments; inorganic black pigments such as black iron oxide and low titanium oxide; inorganic purple pigments such as mango violet and cobalt violet; inorganic green pigments such as chromium oxide, chromium hydroxide, and cobalt titanate; Pearl pigments such as titanium oxide coated mica, titanium oxide coated bismuth oxychloride, titanium oxide coated talc, colored titanium oxide coated mica, bismuth oxychloride, fish scale foil; talc, sericite, mica, Extender pigments such as kaolin, calcium carbonate, magnesium carbonate, silicic anhydride, barium sulfate, aluminum hydroxide; Aluminum powder, copper powder, metal powder pigments such as gold; surface treated inorganic and metallic powder pigments; zirconium, organic pigments such as barium
- oil base examples include higher (polyhydric) alcohols such as oleyl alcohol, jojoba alcohol, chimyl alcohol, seraky alcohol, butyl alcohol, hexyldecanol, isostearyl alcohol, 2-octyldodecanol, and dimer diol; benzyl alcohol and the like.
- higher (polyhydric) alcohols such as oleyl alcohol, jojoba alcohol, chimyl alcohol, seraky alcohol, butyl alcohol, hexyldecanol, isostearyl alcohol, 2-octyldodecanol, and dimer diol
- benzyl alcohol and the like.
- Aralkyl alcohols and derivatives thereof isostearic acid, behenic acid, undecylenic acid, 12-hydroxystearic acid, palmitooleic acid, oleic acid, linoleic acid, linoleic acid, erucic acid, docosahexaenoic acid, eicosapentaenoic acid, isohexadecanoic acid, Anteisohenicosanoic acid, long-chain branched fatty acid, dimer acid, hydrogenated dimer acid, etc .; liquid paraffin (mineral oil), heavy liquid isoparaffin, light liquid isoparaffin, ⁇ -olefin Rigomer, polyisobutene, hydrogenated polyisobutene, polybutene, squalane, olive-derived squalane, squalene, petrolatum, hydrocarbons such as solid paraffin; candelilla wax, carnauba wax, rice wax, wood wax, beeswax,
- humectant / feel improver examples include polyols such as glycerin, trimethylolpropane, pentaerythritol, hexylene glycol, diglycerin, polyglycerin, diethylene glycol, dipropylene glycol, polypropylene glycol, and ethylene glycol / propylene glycol copolymer.
- polyols such as glycerin, trimethylolpropane, pentaerythritol, hexylene glycol, diglycerin, polyglycerin, diethylene glycol, dipropylene glycol, polypropylene glycol, and ethylene glycol / propylene glycol copolymer.
- glycol alkyl ethers such as diethylene glycol monoethyl ether (ethoxydiglycol), ethylene glycol monoethyl ether, ethylene glycol monobutyl ether, and diethylene glycol dibutyl ether; (eicosane diacid / tetradecane diacid) polyglyceryl-10, tetradecane di -Soluble esters such as polyglyceryl-10 acid; sorbitol, xylitol, erythritol, man Sugar alcohols such as toll and maltitol; glucose, fructose, galactose, mannose, threose, xylose, arabinose, fucose, ribose, deoxyribose, maltose, trehalose, lactose, raffinose, gluconic acid, glucuronic acid, cyclodextrins ( ⁇ -, ⁇
- Preferred examples of the surfactant include an anionic surfactant, a nonionic surfactant, a cationic surfactant, an amphoteric surfactant, and a polymer surfactant.
- Preferred examples of the surfactant include, for example, fatty acid salts such as potassium laurate and potassium myristate; alkyl sulfate salts such as sodium lauryl sulfate, triethanolamine lauryl sulfate and ammonium lauryl sulfate; Polyoxyethylene alkyl sulfates such as sodium laureth sulfate and triethanolamine laureth sulfate; cocoyl methyl taurine sodium, cocoyl methyl taurine potassium, lauroyl methyl taurine sodium, myristoyl methyl taurine sodium, lauroyl methyl alanine sodium, lauroyl sarcosine sodium, lauroyl sarcosine tri Acyl N-methyl amino acid salts such as ethanolamine
- Polyoxyethylene alkyl ethers having various polyoxyethylene addition numbers such as poly (ethylene behenyl ether) s, isosteares (polyoxyethylene isostearyl ether), octyldodeces (polyoxyethylene octyldodecyl ether); polyoxyethylene alkylphenyl Ether: polyoxyethylene hydrogenated castor oil, polyoxyethylene castor oil, polyoxyethylene hydrogenated castor oil monoisostearate, polyoxyethylene hydrogenated castor oil triisostearate, polyoxyethylene hydrogenated castor oil monopyroglutamic acid monoisostearate diester Oil and hydrogenated castor oil derivatives, such as polyoxyethylene hydrogenated castor oil and maleic acid; polyoxyethylene phytosterol; polyoxyethylene cholesterol Polyoxyethylene cholestanol; polyoxyethylene lanolin; polyoxyethylene reduced lanolin; polyoxyethylene / polyoxypropylene cetyl ether, polyoxyethylene / polyoxypropylene 2-decyltetradecy
- Alkyl glyceryl ethers polyhydric alcohol alkyl ethers; polyoxyethylene alkylamines; condensates of tetrapolyoxyethylene / tetrapolyoxypropylene-ethylenediamine; natural surfactants such as saponins and sophorolipids; polyoxyethylene fatty acid amides;
- coconut oil fatty acid monoethanolamide cocamide MEA
- coconut oil fatty acid diethanolamide cocamide DEA
- lauric acid monoethanolamide Lauramide MEA
- lauric acid diethanolamide lauric acid diethanolamide
- lauramide MIPA lauric acid monoisopropanolamide
- palmitic acid monoethanolamide partamide MEA
- palmitic acid diethanolamide partamide DEA
- coconut oil fatty acid methylethanolamide Fatty acid alkanolamides such as cocamidomethyl MEA
- alkyl dimethylamine oxides such as lauramine oxide, cocamine oxide, stearamine oxide, and behen
- Silicone-based nonionic surfactants include alkyltrimethylammonium chlorides such as behentrimonium chloride, stearyltrimonium chloride, cetrimonium chloride, lauryltrimonium chloride; stearyltrimonium bromide Alkyltrimethylammonium bromide; dialkyldimethylammonium chlorides such as distearyldimonium chloride and dicocodimonium chloride; fatty acid amidoamines such as stearamidopropyldimethylamine and stearamidoethyldiethylamine and salts thereof; alkyl ethers such as stearoxypropyldimethylamine Amines and salts or quaternary salts thereof; ethyl sulfate long-chain branched fatty acid (12-31) aminopropylethyldimethylammonium, ethyl Fatty acid amide type quaternary ammonium salts such as lanolin
- -Dimethylamino acid betaine imidazoline betaines such as sodium cocoamphoacetate and sodium lauroamphoacetate; alkylsulfobetaines such as alkyldimethyltaurine; sulfuric acid betaines such as alkyldimethylaminoethanol sulfate; phosphoric acids such as alkyldimethylaminoethanol phosphate; Betaine; sphingolipids such as phosphatidylcholine, phosphatidylethanolamine, phosphatidylserine, sphingomyelin, lysolecithin, hydrogenated soybean phospholipids, Phospholipids such as hydrogenated soybean phospholipids, hydrogenated egg yolk phospholipids, partially hydrogenated egg yolk phospholipids, hydroxylated lecithin; silicone amphoteric surfactants; polymer surfactants such as polyvinyl alcohol and sodium alginate , Starch derivatives, tragacanth gum, acrylic acid / alky
- the solvent examples include lower alcohols such as ethanol, 2-propanol (isopropyl alcohol), butanol and isobutyl alcohol; glycols such as propylene glycol, diethylene glycol, dipropylene glycol and isopentyl diol; diethylene glycol monoethyl ether (ethoxydiglycol).
- lower alcohols such as ethanol, 2-propanol (isopropyl alcohol), butanol and isobutyl alcohol
- glycols such as propylene glycol, diethylene glycol, dipropylene glycol and isopentyl diol
- diethylene glycol monoethyl ether ethoxydiglycol
- Glycol ethers such as ethylene glycol monoethyl ether, ethylene glycol monobutyl ether, triethylene glycol monoethyl ether, diethylene glycol diethyl ether, diethylene glycol dibutyl ether, propylene glycol monoethyl ether and dipropylene glycol monoethyl ether; ethylene glycol monoethyl ether Acetate, diethylene glycol Glycol ether esters such as ethyl ether acetate and propylene glycol monoethyl ether acetate; glycol esters such as diethoxyethyl succinate and ethylene glycol disuccinate; benzyl alcohol, benzyloxyethanol, propylene carbonate, dialkyl carbonate, acetone, and acetic acid Preferred examples include ethyl, N-methylpyrrolidone and toluene.
- tocopherol derivatives such as tocopherol (vitamin E) and tocopherol acetate; BHT, BHA; gallic acid derivatives such as propyl gallate; vitamin C (ascorbic acid) and / or derivatives thereof; erythorbic acid and derivatives thereof; Sulfites such as sodium sulfite; bisulfites such as sodium bisulfite; thiosulfates such as sodium thiosulfate; metabisulfite; thiotaurine, hypotaurine; thioglycerol, thiourea, thioglycolic acid, and cysteine hydrochloride are preferred.
- thioglycolic acid As the reducing agent, thioglycolic acid, cysteine, cysteamine and the like are preferable.
- oxidizing agent preferred are aqueous hydrogen peroxide, ammonium persulfate, sodium bromate, percarbonate and the like.
- Preservatives, antibacterial agents, and bactericides include hydroxybenzoic acid such as methylparaben, ethylparaben, propylparaben, and butylparaben and salts or esters thereof; salicylic acid; sodium benzoate; phenoxyethanol; methylchloroisothiazolinone; Isothiazolinone derivatives such as linone; imidazolinium urea; dehydroacetic acid and salts thereof; phenols; halogenated bisphenols such as triclosan, acid amides, quaternary ammonium salts; trichlorocarbanide, zinc pyrithione, benzalkonium chloride, chloride Benzethonium, sorbic acid, chlorhexidine, chlorhexidine gluconate, halocarban, hexachlorophen, hinokitiol; phenol, isopropylphenol, cresol, Mall, para chlorophenol, pheny
- the chelating agent examples include edetates (ethylenediaminetetraacetate) such as EDTA, EDTA2Na, EDTA3Na, and EDTA4Na; hydroxyethylethylenediaminetriacetate such as HEDTA3Na; pentetate (diethylenetriaminepentaacetate); phytic acid; etidronic acid and the like Phosphonic acid and salts thereof such as sodium salt thereof; polyaspartic acid, polyamino acids such as polyglutamic acid; sodium polyphosphate, sodium metaphosphate, phosphoric acid; sodium citrate, citric acid, alanine, dihydroxyethylglycine, gluconic acid, Ascorbic acid, succinic acid and tartaric acid are preferred.
- edetates ethylenediaminetetraacetate
- HEDTA3Na EDTA3Na
- EDTA4Na hydroxyethylethylenediaminetriacetate
- pH adjusters, acids and alkalis include ascorbic acid, citric acid, sodium citrate, lactic acid, sodium lactate, potassium lactate, glycolic acid, succinic acid, acetic acid, sodium acetate, malic acid, tartaric acid, fumaric acid, phosphoric acid, Hydrochloric acid, sulfuric acid, monoethanolamine, diethanolamine, triethanolamine, isopropanolamine, triisopropanolamine, 2-amino-2-methyl-1,3-propanediol, 2-amino-2-hydroxymethyl-1,3-propane Preferred examples include diol, arginine, sodium hydroxide, potassium hydroxide, aqueous ammonia, guanidine carbonate, and ammonium carbonate.
- Powders include mica, talc, kaolin, sericite, montmorillonite, kaolinite, mica, muscovite, phlogopite, synthetic mica, biotite, biotite, permiculite, magnesium carbonate, calcium carbonate, aluminum silicate, silicate Barium, calcium silicate, magnesium silicate, strontium silicate, metal tungstate, magnesium, zeolite, barium sulfate, calcined calcium sulfate, calcium phosphate such as tricalcium phosphate, fluorine apatite, hydroxyapatite, ceramic powder, bentonite, smectite , Clay, mud, metal soap (eg, zinc myristate, calcium palmitate, aluminum stearate), calcium carbonate, red iron oxide, yellow iron oxide, black iron oxide, ultramarine, navy blue, carbon black, acid Titanium, fine and ultrafine titanium oxide, zinc oxide, fine and ultrafine zinc oxide, alumina, silica, fumed
- Copolymer resin powder polyester powder, benzoguanamine resin powder
- Organic powders and surface-treated powders of various sizes and shapes such as polyethylene terephthalate / polymethyl methacrylate laminate powder, polyethylene terephthalate / aluminum / epoxy laminate powder, urethane powder, silicone powder, Teflon (registered trademark) powder, etc.
- Organic-inorganic composite powders are preferred.
- sodium chloride-containing salts such as common salt, normal salt, rock salt, sea salt, natural salt
- sodium phosphates such as 1Na / 2Na / 3Na phosphate, phosphoric acid Potassiums, calcium phosphates, and magnesium phosphates are preferred.
- the ultraviolet absorber examples include para-aminobenzoic acid, para-aminobenzoic acid monoglycerin ester, N, N-dipropoxypara-aminobenzoic acid ethyl ester, N, N-diethoxypara-aminobenzoic acid ethyl ester, N, N-dimethylpara-aminobenzoic acid ethyl ester Benzoic acid-based ultraviolet absorbers such as esters, N, N-dimethylparaaminobenzoic acid butyl ester, N, N-dimethylparaaminobenzoic acid methyl ester; anthranilic acid-based ultraviolet absorbers such as homomenthyl-N-acetylanthranilate; salicylic acid And salicylic acids such as sodium salts thereof, amyl salicylate, menthyl salicylate, homomenthyl salicylate, octyl salicylate, phenyl salicylate, benzyl sal
- whitening agents include hydroquinone glycosides such as arbutin and ⁇ -arbutin and esters thereof; ascorbic acid phosphates such as ascorbic acid, sodium ascorbic acid phosphate and magnesium ascorbic acid phosphate; ascorbic acid Fatty acid esters of ascorbic acid such as tetraisopalmitic acid ester, alkyl ethers of ascorbic acid such as ethyl ether of ascorbic acid, glucosides of ascorbic acid such as ascorbic acid-2-glucoside and fatty acid esters thereof, ascorbic acid sulfate ester, tocopheryl ascorbyl phosphate And other ascorbic acid derivatives; kojic acid, ellagic acid, tranexamic acid and its derivatives, ferulic acid and its derivatives, placenta extract, glutathione, oryzanol, butylreso Shinoru, oil-soluble Kamomiraekisu, oil-soluble phosphat
- vitamin A such as retinol, retinol acetate, retinol palmitate; thiamine hydrochloride, thiamine sulfate, riboflavin, riboflavin acetate, pyridoxine hydrochloride, pyridoxine dioctanoate, pyridoxine dipalmitate, Flavin adenine dinucleotide, cyanocobalamin, folic acids, nicotinic acids such as nicotinamide / benzyl nicotinate, vitamin Bs such as choline; vitamin Cs such as ascorbic acid and salts thereof such as sodium; vitamin D; vitamins E such as ⁇ , ⁇ , ⁇ -tocopherol; other vitamins such as pantothenic acid and biotin; phosphate salts of ascorbic acid such as sodium salt of ascorbic acid phosphate and magnesium salt of ascorbic acid phosphate; Fatty acid esters of ascorbic
- hair-growth agents examples include plant extracts and tinctures such as assembly extract, pepper tincture, ginger tincture, ginger extract, and cantharis tincture; capsaicin, nonylate valenylamide, zingerone, ictamol, tannic acid , Borneol, cyclanderate, cinnarizine, tolazoline, acetylcholine, verapamil, cepharanthin, ⁇ -oryzanol, vitamin E and derivatives such as tocopherol nicotinate / tocopherol acetate, ⁇ -oryzanol, nicotinic acid and nicotinamide / nicotinic acid benzyl ester Derivatives such as inositol hexanicotinate, nicotine alcohol, etc., allantoin, photosensitizer 301, photosensitizer 401, capronium chloride, pentadecan
- estradiol estrone
- ethinyl estradiol cortisone
- hydrocortisone hydrocortisone
- prednisone prednisone and the like are preferred.
- Other therapeutic agents such as anti-wrinkle agents, anti-aging agents, tightening agents, cooling agents, warming agents, wound healing promoters, stimulants, analgesics, cell activators, etc. include retinols, retinoic acids, retinoin Acid tocopheryl; lactic acid, glycolic acid, gluconic acid, fruit acid, salicylic acid and derivatives such as glycosides / esterified products thereof, hydroxycapric acid, long-chain ⁇ -hydroxy fatty acid, ⁇ - such as long-chain ⁇ -hydroxy fatty acid cholesteryl and the like.
- ⁇ -hydroxy acids and derivatives thereof ⁇ -aminobutyric acid, ⁇ -amino- ⁇ -hydroxybutyric acid; carnitine; carnosine; creatine; ceramides, sphingosines; caffeine, xanthine, and derivatives thereof; coenzyme Q10, carotene, lycopene , Astaxanthin, lutein, ⁇ -lipoic acid, nanocolloidal platinum, fullerenes, etc.
- Catechins flavones such as quercetin; isoflavones; gallic acid and ester sugar derivatives; tannins, sesamin, protoanthocyanidins, chlorogenic acid, polyphenols such as apple polyphenols; rutin and glycosides Derivatives such as hesperidin and glycosides; lignan glycosides; licorice extract-related substances such as glabridine, glabrene, liquiritin, isoliquiritin; lactoferrin; shogaol, gingerol; fragrance substances such as menthol, cedrol and derivatives thereof; Preferred are capsaicin, vanillin and the like and derivatives; insect repellents such as diethyltoluamide; complexes of physiologically active substances with cyclodextrins.
- Plant / animal / microbial extracts include iris extract, ashitaba extract, asnaro extract, asparagus extract, avocado extract, amateur extract, almond extract,retea extract, arnica extract, aloe extract, apricot extract, apricot nucleus extract, ginkgo extract , Inchiko extract, Fennel extract, Turmeric extract, Oolong tea extract, Owaurushi extract, Eijitsu extract, Echinashi leaf extract, Emisoso extract, Japanese gourd extract, Oubaku extract, Ouren extract, Barley extract, Panax ginseng extract, Hypericum perforatum extract, Odorikosou extract, Ononisu extract , Dutch mustard extract, orange extract, seawater dried product, seaweed extract, oyster leaf extract, oyster extract, hydrolyzed elastin, hydrolyzed wheat powder, water content Silk, cuckoo extract, chamomile extract, oil-soluble chamomile extract, carrot extract, sagebrush extract, oats wheat extract, calcade extract,
- antipruritic agents examples include diphenhydramine hydrochloride, chlorpheniramine maleate, camphor, substance-P inhibitor and the like.
- exfoliating / dissolving agent examples include salicylic acid, sulfur, resorcin, selenium sulfide, and pyridoxine.
- antiperspirants examples include chlorohydroxyaluminum, aluminum chloride, zinc oxide, zinc paraphenolsulfonate, and the like.
- cooling agent examples include menthol, methyl salicylate, and the like.
- Astringents include citric acid, tartaric acid, lactic acid, aluminum / potassium sulfate, tannic acid and the like.
- Enzymes include superoxide dismutase, catalase, lysozyme chloride, lipase, papain, pancreatin, protease and the like.
- nucleic acids As the nucleic acids, ribonucleic acids and salts thereof, deoxyribonucleic acids and salts thereof, and disodium adenosine triphosphate are preferred.
- acetyl cedrene, amyl cinnamaldehyde, allyl amyl glycolate, ⁇ -ionone, iso-e super isobutyl quinoline, iris oil, iron, indole, ylang-ylang oil, undecanaal, undecenal, ⁇ -undecalactone, Estragole, Eugenol, Oak Moss, Opoponax Resinoid, Orange Oil, Eugenol, Orlanthol, Garrack Solid, Carvacrol, L-Carbon, Camphor, Cannon, Carrot Seed Oil, Clove Oil, Methyl Cinnarate, Geraniol, Geranilnitrile , Isobornyl acetate, geranyl acetate, dimethylbenzylcarbinyl acetate, styryl acetate, ceryl acetate, terpinel acetate, pt-butylcyclohexyl acetate, vetivery
- Pigments, colorants and dyes include Brown No. 201, Black No. 401, Purple No. 201, Purple No. 401, Blue No. 1, Blue No. 2, Blue No. 201, Blue No. 202, Blue No. 203, Blue No. 204, Blue No.
- Yellow No. 202-2 Yellow No. 203, Yellow No. 204, Yellow No. 205, Yellow No. 4, Yellow No. 401, Yellow No. 402, Yellow No. 403-1, Yellow No. 404, Yellow No. 405, Yellow No. 406, Yellow No. 407, yellow No.
- anti-inflammatory and anti-inflammatory agents include glycyrrhizic acid and derivatives thereof, glycyrrhetinic acid derivatives, salicylic acid derivatives, hinokitiol, guaiazulene, allantoin, indomethacin, ketoprofen, ibuprofen, diclofenac, loxoprofen, celecoxib, infliximab, etanercorcept, zinc oxide, hydrochloride, and hydrochloride.
- Prednisone, diphedramine hydrochloride, chlorpheniramine maleate; and plant extracts such as a peach leaf extract and a pine leaf extract are preferred.
- Anti-asthma, anti-chronic obstructive pulmonary disease, anti-allergy, immunomodulators include aminophylline, theophylline, steroids (fluticasone, beclomethasone, etc.), leukotriene antagonists, thromboxane inhibitors, intal, ⁇ 2 agonist (Formoterol, salmeterol, albuterol, tulobuterol, clenbuterol, epinephrine, etc.), tiotropium, ipratropium, dextromethorphan, dimethorphan, bromhexine, tranilast, ketotifen, azelastine, cetirizine, chlorpheniramine, mexetalimscrotaline, mextrezine , Cytokine regulators, interferon, omalizumab, and protein / antibody preparations are preferred.
- Oseltamivir, zanamivir and itraconazole are preferred as anti-infectives and antifungals.
- cosmetic raw material standards cosmetic varieties compounding component standards, Japanese Cosmetic Industry Federation component label name list, INCI dictionary (The International Cosmetic Ingredient Dictionary and Handbook), quasi-drug raw material standards, Japanese Pharmacopoeia, pharmaceutical additive standards Ingredients listed in the Official Compendium of Food Additives, etc., and listed in Japanese and foreign patent publications and patent publications (including published publications and republication) whose international patent classification IPC belongs to the classification of A61K7 and A61K8 It is possible to incorporate known cosmetic components, pharmaceutical components, food components, and the like, such as those described above, in a known combination and in a blending ratio and blending amount.
- the film-forming composition of the present invention can be produced, for example, by mixing and stirring at least one kind of lipid peptide type compound, water and, if desired, other components while heating, and then allowing the mixture to stand and cool.
- the present invention also relates to a method for producing a film, characterized by using the above-mentioned film-forming composition.
- the method for producing the film is not particularly limited as long as the film can be produced, and examples include a method in which the above-mentioned film-forming composition is applied to an object to be applied and then dried by natural drying or heat drying.
- the solid obtained here was dissolved in a mixed solution of 600 g of water and 750 g of methanol, and 30.5 mL (183.2 mmol) of 6N hydrochloric acid was added to neutralize the solid to precipitate a solid, which was filtered.
- the obtained solid was dissolved in a mixed solution of 120 g of tetrahydrofuran and 30 g of water at 60 ° C, 150 g of ethyl acetate was added, and the mixture was cooled from 60 ° C to 30 ° C. Thereafter, the precipitated solid was filtered.
- the obtained solid was dissolved in a solvent of 120 g of tetrahydrofuran and 60 g of acetonitrile, heated to 60 ° C., stirred for 1 hour, cooled, and filtered.
- the obtained solid was washed with 120 g of water, filtered, and dried under reduced pressure to obtain 26.9 g of white crystals of N-palmitoyl-Gly-His free body (hereinafter, also simply referred to as Pal-GH) (yield: 65). %).
- Preparation Example 1 Preparation of premix
- Pal-GH, stearic acid, 1,2-hexanediol, polyoxyethylene lauryl ether (manufactured by Nikko Chemicals) and water as additives were added to a 300 mL beaker at the ratio shown in Table 1 below, and the liquid temperature was 80 ° C.
- the mixture was heated and stirred at 200 rpm to obtain a uniform solution.
- the mixture was cooled with stirring, and when the liquid temperature reached 60 ° C., the mixture was allowed to stand and cooled to prepare an ES-01 premix.
- Example 1 Gelation test and observation of gel morphology According to Table 2, propylene glycol alginate, POE60-hardened castor oil, mineral oil and pure water were placed in a 300 mL tall beaker, and heated and stirred at 80 ° C. The stirring was performed at 200 rpm using a LABORATORY HIGH MIXER manufactured by AS ONE Corporation. Next, the ES-01 premix heated to 80 ° C. was added, and the mixture was further heated and stirred for 5 minutes. After the completion of the heating and stirring, the mixture was stirred and cooled until the liquid temperature reached about 50 ° C., and gel formation was confirmed. Gel formation was confirmed by the test tube inversion method, and the state where the fluidity of the dispersion liquid was lost and the liquid did not flow down even when the tall beaker was inverted was determined to be gelation.
- FIG. 1 shows observation images of a sample immediately after being placed in the scanning electron microscope and a sample that has been placed in the scanning electron microscope for a predetermined time and dried.
- Example 2 According to Table 3, propylene glycol alginate, POE60 hydrogenated castor oil, and pure water were placed in a 300 mL tall beaker, and heated and stirred at 80 ° C. The stirring was performed at 200 rpm using a LABORATORY HIGH MIXER manufactured by AS ONE Corporation. Next, the ES-01 premix heated to 80 ° C. was added, and the mixture was further heated and stirred for 5 minutes. After completion of the heating and stirring, the mixture was stirred and cooled until the liquid temperature reached about 50 ° C., and gel formation was confirmed. Gel formation was confirmed by the test tube inversion method, and the state where the fluidity of the dispersion liquid was lost and the liquid did not flow down even when the tall beaker was inverted was determined to be gelation.
- Example 3 and Comparative Example 3 Reflection light intensity measurement According to Table 4, the gel obtained in Example 2 or the liquid of Comparative Example 2 was weighed, and squalane was added thereto. After the addition, a uniform thickener or liquid was obtained using a spatula. Then, the thickened material of Example 3 or the liquid of Comparative Example 3 shown in Table 4 was applied to Bioskin (manufactured by Bureaux Co., Ltd.) at a concentration of 10 mg / cm 2, and was placed in a thermostat at 35 ° C. And dried.
- Bioskin manufactured by Bureaux Co., Ltd.
- the coating obtained by the composition of the present invention has about half the reflected light intensity as compared with the coating obtained by the composition not containing the lipid peptide type compound (Comparative Example 3). (Example 3).
- the film-forming composition of the present invention was able to form a film capable of suppressing light reflection.
- Example 4 and Comparative Example 4 Moisture retention test of shampoo According to Table 5, A phase was weighed in a 300 mL tall beaker. On the other hand, ES-01 premix similarly heated and dissolved in purified water heated to 70 ° C. was weighed to prepare phase B. Phase B was added to Phase A heated to 70 ° C. Thereafter, the mixture was stirred and cooled until the liquid temperature reached about 40 ° C.
- a human hair bundle (black hair, 1 g, 10 cm) (manufactured by Kadoya Co., Ltd.) was reciprocated 10 times with a cyclon sponge, and was similarly reciprocated 10 times with a non-cyclon sponge contact surface.
- 10 g of the composition of Example 4 or Comparative Example 4 was weighed into a 50-mL sample tube, and the hair bundle subjected to the above-described treatment was added thereto, followed by stirring with a mix rotor for 3 minutes. About 300 mL of pure water was put into a 300 mL tall beaker, and a hair bundle immersed in shampoo was put therein, and washed for 30 seconds.
- Examples 5 to 8 Moisture retention test of shampoo According to Table 6, each component was weighed in a 300 mL tall beaker. The mixture was heated to 70 ° C., and when a uniform solution was obtained, the mixture was stirred and cooled until the liquid temperature reached about 40 ° C. Then, the moisture retention was determined in the same procedure as in Example 4. The result is shown in FIG. Here, Pal-H represents palmitoyl histidine.
- the shampoo to which the lipid peptide type compound was added had an improved hair moisturizing rate as compared to the shampoo without the lipid peptide type compound (Comparative Example 4) (Examples 4 to 8). ).
- the moisturizing rate of hair was particularly improved and the moisturizing effect was excellent.
- Example 9 and Comparative Example 5 According to Table 7, the phases B and C were dissolved by heating. After heating the phase D and performing a homomixer (5000 rpm, 3 minutes) treatment, the above-mentioned heated phases B and C were added, and a homomixer (5000 rpm, 3 minutes) treatment was further performed. After the homomixer treatment, the phase A was slowly added, and the mixture was further treated with a homomixer (5000 rpm, 3 minutes). Thereafter, the mixture was stirred and cooled until the liquid temperature reached about 40 ° C.
- a homomixer 5000 rpm, 3 minutes
- Example 9 100 ⁇ L of the composition of Example 9 or Comparative Example 5 was uniformly applied to artificial leather cut to 2 cm ⁇ 2 cm, dried at room temperature for 30 minutes, and a contact angle meter Drop @ Master @ DMC-MCS (manufactured by Kyowa Kogyo Co., Ltd.) was used. To evaluate the contact angle. The result is shown in FIG.
- the contact angle of the coating obtained by the composition of the present invention was larger than that of the coating obtained by the composition not containing the lipid peptide type compound (Comparative Example 5).
- Example 9 The film forming composition of the present invention was able to form a film having good water repellency.
- Example 10 and Comparative Example 6 Moisture retention test of conditioner Phase A was weighed according to Table 8, and heated to 75 ° C to obtain a uniform solution. On the other hand, components other than the ES-01 premix of the phase B were heated to 75 ° C., and the ES-01 premix heated to 75 ° C. was added thereto. Next, the phase B was added to the phase A heated to 75 ° C., and the mixture was stirred and cooled to around room temperature.
- a human hair bundle (black hair, 1 g, 10 cm) (manufactured by Kadoya Co., Ltd.) was reciprocated 10 times with a cyclon sponge, and was similarly reciprocated 10 times with a non-cyclon sponge contact surface.
- 4.0 g of the composition (conditioner) of Example 10 or Comparative Example 6 was placed on the hair bundle that had been subjected to the above-mentioned treatment, and after being hand-adapted, about 300 mL of pure water was placed in a 300 mL tall beaker. Washing was performed for 30 seconds by immersion and shaking in the object. Further, rewashing was performed in a separately prepared 300 mL tall beaker containing about 300 mL of pure water.
- FIG. 6 shows the result.
- SEM scanning electron microscope
- the conditioner to which the lipid peptide-type compound was added was improved in the moisturizing rate of the hair and superior in the moisturizing effect as compared to the conditioner without the lipid peptide-type compound (Comparative Example 6) ( Example 10). Further, the SEM image shown in FIG. 7 confirmed that the conditioner to which the lipid peptide type compound was added formed a film on the hair. On the other hand, the conditioner to which no lipid peptide type compound was added did not form a film on the hair.
- Formulation Example 2 Liquid Crystal Cream According to the composition shown in Table 10, A, B, C, D and E were prepared and mixed to prepare a liquid crystal cream.
- UV milk (O / W emulsion) According to the composition shown in Table 11, A, B, C and D were prepared and mixed to prepare a UV milk (O / W emulsion).
- Formulation Example 6 Shampoo According to the composition shown in Table 14, A, B, C and D were prepared and mixed to prepare a shampoo.
- Example 11 and Comparative Example 7 Evaluation of Hair After Conditioner Treatment as Fingers As Example 11, a composition having the same composition as the composition of Example 10 was prepared according to Table 8 above. Further, as Comparative Example 7, a composition having the same composition as the composition of Comparative Example 6 was prepared. 10 g of the composition (conditioner) of Example 11 or Comparative Example 7 was uniformly applied to the entire human hair bundle (black hair, 1 g, 10 cm) (manufactured by Kadoya), and after 5 minutes, the applied conditioner was washed. . After standing at room temperature overnight, the frictional resistance was measured using a tactile TYPE33 (Shinto Kagaku Co., Ltd.) and used as an index for the finger. FIG. 8 shows the frictional resistance of Example 11 and Comparative Example 7. As Reference Example 1, the frictional resistance of the untreated human hair bundle was also measured under the same conditions.
- the hair treated with the conditioner had lower friction resistance than the hair not treated with the conditioner (Example 11 and Comparative Example 7).
- the hair treated with the conditioner to which the lipid peptide-type compound was added had remarkably small frictional resistance, and had good fingering properties to the hair (Example 11).
- the film-forming composition of the present invention was able to form a film having low frictional resistance, that is, a film having good finger piercing properties and comb piercing properties.
- Example 12 Preparation of O / W Liquid Foundation According to Table 15 below, an O / W liquid foundation in which 17% by mass of the pigment was uniformly dispersed was prepared. In addition, the amount of each component in Table 15 represents mass% (wt%) based on the total mass of the O / W liquid foundation.
- phase A In a 200 mL beaker (manufactured by HARIO Corporation), 4.0 g of NIKKOL Nikolurs 41, 14.0 g of NIKKOL NATURAL OILS, and 1.0 g of NIKKOL Nicogard 88 were added, and the mixture was heated and stirred at 75 ° C. for 10 minutes (phase A). The phase A is added to the phase B while stirring slowly, and the resulting mixture is emulsified at 5,000 rpm for 3 minutes at 75 ° C. using a homomixer (QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.). Was. Further, 1.0 g (phase D) of ES-01 premix heated to 75 ° C.
- phase B 10 minutes
- a 200 mL beaker manufactured by HARIO Corporation
- 4.0 g of NIKKOL Nikolurs 41, 14.0 g of NIKKOL NATURAL OILS, and 1.0 g of NIKKOL Nicogard 88 were added, and the mixture was heated and stirred at 75 ° C. for 10 minutes (phase A).
- the phase A is added to the phase B while stirring slowly, and the resulting mixture is emulsified at 5,000 rpm for 3 minutes at 75 ° C. using a homomixer (QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.).
- QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.
- Example 13 and Comparative Example 9 Reflection Light Intensity Measurement Reflection was performed using the O / W liquid foundation of Example 12 as Example 13 and the O / W liquid foundation of Comparative Example 8 as Comparative Example 9. The light intensity was measured.
- the ⁇ / W liquid foundation of Example 13 or the ⁇ / W liquid foundation of Comparative Example 9 was applied to Bioskin (manufactured by Beulux Co., Ltd.) to a concentration of 10 mg / cm 2 and placed in a thermostat at 35 ° C. And dried.
- a variable-angle photometer GP-5 manufactured by Murakami Color Research Laboratory Co., Ltd.
- FIG. 9 shows the measurement results.
- Example 14 and Comparative Example 10 Evaluation of cosmetic twist
- the O / W liquid foundation of Example 12 was used as the composition of Example 14, and the O / W liquid foundation of Comparative Example 8 was used as the composition of Comparative Example 10.
- On human skin 0.5 g of the ⁇ / W liquid foundation of Example 14 or ⁇ / W liquid foundation of Comparative Example 10 was applied, and after evenly spreading, 0.5 g of artificial sebum was dropped and allowed to stand at room temperature for 30 minutes. Placed.
- FIG. 10 shows external appearance photographs of Example 14 and Comparative Example 10 after standing at room temperature for 30 minutes.
- Example 15 and Comparative Example 11 Preparation of Conditioner Containing Malic Acid According to Table 16 below, a foundation containing 1.5% by mass (wt%) of malic acid was prepared. Phase A was heated and stirred to 75 ° C. in a 200 mL beaker (manufactured by HARIO Corporation). Phase B and phase C were mixed in a 200 mL beaker, and heated and stirred to 75 ° C. The A phase was added to the mixed phase of the B phase and the C phase, and emulsification was performed at 75 rpm at 5,000 rpm for 5 minutes using a homomixer (QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.).
- a homomixer QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.
- phase D heated to 75 ° C. was added, and the mixture was heated and stirred at 75 ° C. for 5 minutes, and then stirred and cooled until the temperature reached 40 ° C. In the above steps, all stirring was performed at 200 rpm.
- Example 16 and Comparative Example 12 Moisturizing test of human hair (1) Preparation procedure of damaged hair sample (damaged hair) 50 bundles of human hair (BS-BA, manufactured by Belux Inc.) (per bundle) , About 10 cm, about 1 g), apply one set of Gatsby EX High Bleach (manufactured by Mandom Co., Ltd.) (mixture of 18 g of powder, 70 mL of water, 35 g of cream) and leave it at room temperature for 1 hour to decolorize the hair Processing was performed. Thereafter, the hair was washed with distilled water, and dried for 30 minutes using a constant temperature dryer (OF-300B, manufactured by AS ONE Corporation) set at 65 ° C. The above operation was repeated three times in total.
- BS-BA manufactured by Belux Inc.
- the hair was immersed in 1 L of an aqueous solution in which 1% by mass of sodium dodecyl sulfate (manufactured by Fujifilm Wako Pure Chemical Industries, Ltd.) was dissolved in distilled water, and washed with distilled water. Damaged hair samples (bleach hair) were prepared by removing excess water with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.) and then drying overnight at room temperature.
- sodium dodecyl sulfate manufactured by Fujifilm Wako Pure Chemical Industries, Ltd.
- Damaged hair samples were prepared by removing excess water with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.) and then drying overnight at room temperature.
- the black hair treated with the conditioner had a lower water loss rate than the black hair not treated with the conditioner (Reference Examples 2 and 3) (Example 16 and Comparative Example 12).
- the black hair treated with the conditioner to which the lipid peptide type compound was added had a lower water loss rate and improved the moisturizing effect of human hair.
- Nystack registered trademark
- NW-10 manufactured by Nichiban Co., Ltd.
- the black hair treated with the conditioner had higher reflected light intensity than the black hair not treated with the conditioner, and gloss was imparted to the hair (the gloss recovered) (Example 17 and Comparative Example) 13).
- the black hair treated with the conditioner to which the lipid peptide type compound was added had particularly high reflected light intensity, and the gloss of the hair was further improved (the gloss was recovered more).
- Example 18 and Comparative Example 14 Quantification of Malic Acid in Human Hair After Conditioner Treatment As Example 18, human hair treated in (2) of Example 16 was used, and as Comparative Example 14, Comparative Example 12 was used. Using the human hair treated in (2), the amount of malic acid in the human hair was evaluated. The quantification of malic acid was evaluated using the EnzyChrom TM Malate Assay Kit (EMAL-100) manufactured by BioAssay Systems. 200 milligrams of the human hair of Example 18 or Comparative Example 14 was weighed into a 50 mL sample tube, and 4 mL of purified water was added thereto, followed by ultrasonication for 60 minutes to extract malic acid.
- EEL-100 EnzyChrom TM Malate Assay Kit
- the film-forming composition of the present invention was able to form a film having a high retention of the active ingredient (a coating that could contain a large amount of the active ingredient).
- Example 19 Comparative Example 15; SEM measurement of conditioner-treated hair
- 1 g of the conditioner prepared in Example 10 and Comparative Example 6 was applied to a bundle of damaged hair (10 cm, 50 pieces) prepared in the same procedure as (1) of Example 16, It was left still for 5 minutes. Thereafter, the resultant was washed by shaking in a 300 mL tall beaker containing 300 mL of purified water, and excess purified water was removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature.
- K-dry manufactured by Nippon Paper Crecia Co., Ltd.
- Example 20 Comparative Example 16; Evaluation of moisture absorption and desorption of hair treated with conditioner
- Example 20 a bundle of hair (approximately 10 cm, approximately 1 g) produced by the same procedure as in Example 16 (1) was subjected to the same treatment as in Example 16 (2). After shearing to 1 cm, 16 mg of hair was weighed, and the amount of moisture absorption / desorption was evaluated using a moisture absorption / desorption measuring device IGAsorp (manufactured by Heiden Isokeyama). After measuring the wet weight of each sample under the condition of a temperature of 25 ° C. and a humidity of 40%, the sample is dried at a temperature of 25 ° C.
- IGAsorp manufactured by Heiden Isokeyama
- the relative humidity was increased stepwise from 0% to 90% in increments of 10%, and the moisture content in the hair was calculated from the equilibrium weight of the sample at each relative humidity point. Further, even when the relative humidity was gradually decreased from 90% to 0% in steps of 10%, the moisture content in the hair was calculated from the equilibrium weight of the sample at each relative humidity point.
- the maximum equilibration time at each humidity was 360 minutes, the above procedure was performed by automatic measurement by a program, and the equilibrium weight of the sample was determined by the asymptote method based on the LDF (linear propulsion approximation) model.
- the equilibrium weight of the sample in the case where the LDF model was not applied was calculated by moving-averaging the measured value after 180 minutes of measurement.
- the results of the obtained moisture percentage are shown in Table 19 (when the humidity rises) and Table 20 (when the humidity falls).
- Comparative Example 16 the results of human hair treated in (2) of Comparative Example 12 and the results of damaged hair are also shown in Table 19. In comparison with the damaged hair not subjected to the conditioner treatment, Comparative Example 16 suppressed the adsorption of moisture, and Example 19 tended to further suppress the adsorption of moisture.
- Example 21, Comparative Example 17; Dynamic friction coefficient of hair treated with conditioner As Example 21, one bundle (about 10 cm, about 1 g) of damaged hair prepared by the same procedure as in Example 16 (1) was subjected to the same treatment as in Example 16 (2). Was measured and used as an index for the finger. The kinetic friction coefficient was measured using a KES-SE friction tester (manufactured by Kato Tech Co., Ltd.), and a 10 mm square silicon sensor was used as the sensor. The hair bundle treated in the same manner as (2) of Comparative Example 12 was also measured, and Comparative Example 17 was obtained. FIG. 13 shows the results of both. The hair provided in Example 21 tended to have a lower coefficient of kinetic friction on the surface of the hair as compared with Comparative Example 17.
- Example 22, Comparative Example 18; Preparation of Conditioner Containing Succinic Acid According to the following Table 21, a conditioner containing 1.5% by mass of succinic acid was prepared. Phase A was heated and stirred to 75 ° C. in a 200 mL beaker (manufactured by HARIO Corporation). The phase B and the phase C were mixed in a 200 mL beaker, and heated and stirred to 75 ° C. The A phase was added to the B + C phase, and emulsified at 5,000 rpm for 5 minutes at 75 ° C. using a homomixer (QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.). Thereafter, phase D heated to 75 ° C. was added, and the mixture was heated and stirred at 75 ° C. for 5 minutes, and then stirred and cooled until the temperature reached 40 ° C. In the above steps, all stirring was performed at 200 rpm.
- Example 23 Comparative Example 19; Evaluation of succinic acid content of conditioner-treated hair
- Example 23 and Comparative Example 19 one gram of the conditioner prepared in Example 22 and Comparative Example 18 was taken by hand, and one bundle (about 10 cm) of damaged hair made in the same procedure as (1) of Example 16 was taken. , About 1 g) and allowed to stand for 5 minutes. Thereafter, the resultant was washed by rocking in a 300 mL tall beaker containing 300 mL of water, excess water was removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature. The above process was performed once, twice, and three times to produce three types of samples.
- Each of the prepared damaged hair samples was cut into a length of 1 cm.
- the quantification of succinic acid was evaluated using a Succinate colorimetric Assay Kit (manufactured by Megazyme). 200 mg of each of the human hairs treated in Example 23 and Comparative Example 19 was weighed in a 50 mL sample tube, and 4 mL of purified water was added thereto, followed by sonication for 60 minutes to extract succinic acid.
- Example 24, Comparative Example 20; Evaluation of penetration amount of succinic acid and lipid peptide in human hair As Example 24 and Comparative Example 20, 1 g of the conditioner prepared in Example 22 and Comparative Example 18 was taken by hand, and a bundle of damaged hair (about 10 cm, About 1 g) and allowed to stand for 5 minutes. Thereafter, the resultant was washed by rocking in a 300 mL tall beaker containing 300 mL of water, excess water was removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature. The above processing was repeated for three days, and the time-of-flight secondary ion mass spectrometer TOF.
- Example 25 Comparative Example 21; Measurement of zeta potential of hair surface treated with conditioner
- Example 25 and Comparative Example 21 one gram of the conditioner prepared in Example 22 and Comparative Example 18 was taken by hand, and a bundle of damaged hair (approximately 10 cm) prepared in the same procedure as (1) of Example 16 was taken. , About 1 g) and allowed to stand for 5 minutes. Thereafter, the resultant was washed by rocking in a 300 mL tall beaker containing 300 mL of water, excess water was removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature.
- K-dry manufactured by Nippon Paper Crecia Co., Ltd.
- the surface zeta potential of the above hair sample was evaluated by a solid surface zeta potential device SurPASS (manufactured by Anton-Paar). 1 mM KCl was prepared for the electrolyte, and the measurement was performed at a temperature of 23 ° C. The results are shown in Table 22. In Comparative Example 21, it was confirmed that the surface potential was more cationic than that in the bleached hair subjected to the bleaching treatment, and in Example 25, the value became more cationic.
- Example 26 Comparative Example 22; Hardness evaluation of conditioner-treated hair surface
- Example 26 and Comparative Example 22 one gram of the conditioner prepared in Example 22 and Comparative Example 18 was taken by hand, and a bundle of damaged hair (approximately 10 cm) made in the same procedure as (1) of Example 16 was taken. , About 1 g) and allowed to stand for 5 minutes. Thereafter, the resultant was washed by rocking in a 300 mL tall beaker containing 300 mL of water, excess water was removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature.
- K-dry manufactured by Nippon Paper Crecia Co., Ltd.
- the hardness of the surface of each of the hair samples was measured with a Hyditron TI980 tribo indenter (manufactured by Bruker).
- the indentation depth was set to 1 ⁇ m, the indenter was a Berkovich type, and the average value measured ten times was calculated.
- FIG. 16 shows the results. It was shown that the bleached damaged hair had a harder hair surface than untreated black hair. Comparative Example 22 showed that the hair surface became softer than that of the damaged hair, and Example 26 showed that the surface became softer.
- Example 27 Comparative Example 23; Evaluation of moisture absorption and desorption of hair treated with conditioner
- Example 27 and Comparative Example 23 one gram of the conditioner prepared in Example 22 and Comparative Example 18 was taken by hand, and a bundle (about 10 cm) of damaged hair made in the same procedure as (1) of Example 16 was taken. , About 1 g) and allowed to stand for 5 minutes. Thereafter, the resultant was washed by rocking in a 300 mL tall beaker containing 300 mL of water, excess water was removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature.
- K-dry manufactured by Nippon Paper Crecia Co., Ltd.
- the prepared treated sample was sheared to 1 cm, and the amount of water adsorbed and desorbed on 3 mg of human hair was evaluated using a water adsorption and desorption measuring device IGAsorp (manufactured by Heiden Isokeyama).
- the hair was dried for 2 days in a relative humidity of 0%, and the amount of mass change when the humidity was increased to 90% relative humidity at a stretch was measured.
- the above procedure was performed by automatic measurement by a program. The results are shown in FIG. As compared with the damaged hair that was not subjected to the conditioner treatment, Comparative Example 23 suppressed the adsorption of moisture, and Example 27 tended to further suppress the adsorption.
- Example 28 Comparative Example 24; Temporal change of shine due to sebum floating
- O / W liquid foundation of Example 12 As Example 28 and the O / W liquid foundation of Comparative Example 8 as Comparative Example 24, the change of shine over time due to sebum floating was evaluated.
- the O / W liquid foundations of Example 12 and Comparative Example 8 were applied to the face of the subject, living as usual, and the surface of the face was applied from 1 hour after application of each foundation to 6 hours after every other hour.
- the gloss was measured using Glossymeter GL200 (manufactured by Courage + khazaka electronic).
- FIG. 18 shows the results. In Example 28, as compared with Comparative Example 24, even after 6 hours, shine caused by sebum floating over time was suppressed.
- Example 29, Comparative Example 25; Preparation of serum According to Table 23 below, a serum containing 0.005% of hyaluronic acid was prepared. Phase A was heated and stirred to 75 ° C. in a 200 mL beaker (manufactured by HARIO Corporation). The B, C, and D phases were mixed in a 200 mL beaker, and heated and stirred to 75 ° C. Phase A was added to phase B + C + D and stirred at 75 ° C. for 5 minutes. Thereafter, the mixture was stirred and cooled to 45 ° C., phase E was added, and the mixture was stirred and cooled to 35 ° C. In the above steps, all stirring was performed at 200 rpm.
- Example 30, Comparative Example 26; Moisturizing effect of serum The moisturizing effect was evaluated using the serum of Example 29 as Example 30 and the serum of Comparative Example 25 as Comparative Example 26.
- the serum of Example 29 and Comparative Example 25 was applied to the inside of the forearm of the subject, and in an environment of 50% humidity and a temperature of 24 ° C., the serum was applied from 1 hour to 7 hours every 1 hour.
- the horny water content of the forearm was measured using Corneometer CM825 (Courage + Khazaka electronic). Ten places were randomly extracted from the applied forearm and measured, and the average was taken as the measurement result. The result is shown in FIG. In Example 30, as compared with Comparative Example 26, a high water content was retained in the keratin even after 7 hours, and it was confirmed that there was a moisturizing effect.
- Example 31, Example 32, Comparative Example 27; Preparation of keratin-containing conditioner According to Table 24 below, a conditioner containing 1.5% by mass of keratin was prepared. Phase A was heated and stirred to 75 ° C. in a 200 mL beaker (manufactured by HARIO Corporation). The phase B and the phase C were mixed in a 200 mL beaker, and heated and stirred to 75 ° C. The A phase was added to the B + C phase, and the mixture was emulsified at 5,000 rpm for 5 minutes at 75 ° C. using a homomixer (QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.).
- a homomixer QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.
- phase D heated to 75 ° C. was added, and the mixture was heated and stirred at 75 ° C. for 5 minutes, and then stirred and cooled until the temperature reached 40 ° C. In the above steps, all stirring was performed at 200 rpm.
- Example 33, Example 34, Comparative Example 28; Evaluation of keratin content in hair treated with conditioner As Example 33, Example 34, and Comparative Example 28, the conditioners adjusted in Example 31, Example 32, and Comparative Example 27 were prepared by hand in the same procedure as (1) of Example 16 by taking each 1 g of the conditioner. It was applied to one bundle (about 10 cm, about 1 g) of damaged hair and allowed to stand for 5 minutes. After that, it is washed by shaking in a 300 mL tall beaker containing 300 mL of methanol (manufactured by Junsei Chemical Co., Ltd.), excess methanol is removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature. Was.
- Each of the prepared damaged hair samples was cut into a length of 1 cm.
- the quantification of keratin was evaluated using Keratin Orange ⁇ -Keratin Assay Kit (manufactured by biocolor).
- 5 mg of each of the human hairs treated in Examples and Comparative Examples was weighed into 1.5 mL microtubes (manufactured by Eppendorf), 1 mL of Digestion reagent was added, and vortexed for 1 hour.
- Reference standard ⁇ -Keratin 5.0 mg / mL
- Digestion reagent was diluted with Digestion reagent to prepare a sample for a calibration curve (0, 1, 2, 3, 4, 5 mg / mL).
- Neutralizing solution (1 M HCl) was added to 200 ⁇ L of the hair sample and 200 ⁇ L of the calibration curve sample to neutralize the pH of each solution.
- 50 ⁇ L of Dye reagent was added to the sample, mixed by vortex, and allowed to stand for 30 minutes to react.
- 450 ⁇ L of Saturated solution ((NH 4 ) 2 SO 4 ) was added to the sample.
- Saturated solution ((NH 4 ) 2 SO 4 ) was added to the sample.
- the sample was centrifuged at 12,000 rpm for 10 minutes, the supernatant was discarded, and 50 ⁇ L of Digestion reagent was added, and the mixture was vortexed sufficiently to solubilize keratin.
- FIG. 20 shows the results of the test. Compared with Comparative Example 28, a higher amount of keratin was extracted from the hairs of Example 33 and Example 34.
- Example 35 Comparative Example 29; Preparation of 18-MEA-containing conditioner
- a conditioner containing 1.5% by mass of 18-methyleicoic acid (18-MEA) was prepared.
- Phase A was heated and stirred to 75 ° C. in a 200 mL beaker (manufactured by HARIO Corporation).
- the phase B and the phase C were mixed in a 200 mL beaker, and heated and stirred to 75 ° C.
- the A phase was added to the B + C phase, and emulsification was performed at 75 rpm at 5,000 rpm for 5 minutes using a homomixer (QUICK HOMO MIXER LR-1A manufactured by Mizuho Industry Co., Ltd.).
- phase D heated to 75 ° C. was added, and the mixture was heated and stirred at 75 ° C. for 5 minutes, and then stirred and cooled until the temperature reached 40 ° C. In the above steps, all stirring was performed at 200 rpm.
- Example 36 Comparative Example 30; SEM measurement of hair treated with conditioner
- Example 36 and Comparative Example 30 one gram of the conditioner prepared in Example 35 and Comparative Example 29 was taken by hand, and one bundle (about 10 cm) of damaged hair made in the same procedure as (1) of Example 16 was taken. , About 1 g) and allowed to stand for 5 minutes. Thereafter, the resultant was washed by shaking in a 300 mL tall beaker containing 300 mL of purified water, and excess purified water was removed with K-dry (manufactured by Nippon Paper Crecia Co., Ltd.), and dried overnight at room temperature.
- K-dry manufactured by Nippon Paper Crecia Co., Ltd.
- FIG. 21 shows the measurement results of the damaged hair.
- the structure of the cuticle surface can be observed, whereas in Example 36, the cuticle surface was covered with the coating.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Dermatology (AREA)
- Organic Chemistry (AREA)
- Emergency Medicine (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dispersion Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Medicinal Chemistry (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Cosmetics (AREA)
Abstract
Description
また、ポリペプチドのような高分子を皮膜形成剤に使用した場合、高分子特有のツッパリ感があるので、その使用感の改善が求められていた。
また、本発明の目的は、ツッパリ感などがほとんどなく、使用感が改善された被膜を形成することができる組成物を提供することである。
また、本発明者は、上記被膜はツッパリ感等がほとんどなく、使用感が良好であることも見出し、本発明を完成にした。
第2観点として、前記脂質ペプチド型化合物が、炭素原子数9乃至23の脂肪族基からなる脂質部に、アミノ酸の繰り返し結合構造を有するペプチド部が結合された化合物である、第1観点に記載の被膜形成組成物に関する。
第3観点として、前記脂質ペプチド型化合物が、下記式(1)乃至式(3)で表される化合物又はその薬学的に使用可能な塩のうちの少なくとも1種からなることを特徴とする、第2観点に記載の被膜形成組成物に関する。
第5観点として、第1観点乃至第3観点のいずれか1つに記載の被膜形成組成物を用いて作成された被膜に関する。
また、本発明によれば、化粧料などに配合すると、優れた保湿性を化粧品に付与することができる。
さらに、本発明によれば、ツッパリ感などがほとんどなく、使用感が良好な被膜を形成することができる。
R1及び隣接するカルボニル基で構成される脂質部(アシル基)の具体例としては、ラウロイル基、ドデシルカルボニル基、ミリストイル基、テトラデシルカルボニル基、パルミトイル基、マルガロイル基、オレオイル基、エライドイル基、リノレオイル基、ステアロイル基、バクセノイル基、オクタデシルカルボニル基、アラキドイル基、エイコシルカルボニル基、ベヘノイル基、エルカノイル基、ドコシルカルボニル基、リグノセイル基、ネルボノイル基等を挙げることができ、特に好ましいものとして、ラウロイル基、ミリストイル基、パルミトイル基、マルガロイル基、ステアロイル基、オレオイル基、エライドイル基及びベヘノイル基が挙げられる。
上記炭素原子数1若しくは2の分岐鎖を有し得る炭素原子数1乃至4のアルキル基とは、主鎖の炭素原子数が1乃至4であり、かつ炭素原子数1若しくは2の分岐鎖を有し得るアルキル基を意味し、その具体例としては、メチル基、エチル基、n-プロピル基、i-プロピル基、n-ブチル基、i-ブチル基、sec-ブチル基又はtert-ブチル基などが挙げられる。
炭素原子数1の分岐鎖を有し得る炭素原子数1乃至3のアルキル基とは、主鎖の炭素原子数が1乃至3であり、かつ炭素原子数1の分岐鎖を有し得るアルキル基を意味し、その具体例としては、メチル基、エチル基、n-プロピル基、i-プロピル基、i-ブチル基又はsec-ブチル基などが挙げられ、好ましくはメチル基、i-プロピル基、i-ブチル基又はsec-ブチル基である。
上記R3を表す-(CH2)n-X基において、Xは好ましくはアミノ基、グアニジノ基、カルバモイル基(-CONH2基)、ピロール基、イミダゾール基、ピラゾール基又はインドール基であり、より好ましくはイミダゾール基である。また、上記-(CH2)n-X基において、nは好ましくは1又は2であり、より好ましくは1である。
従って、上記-(CH2)n-基は、好ましくはアミノメチル基、2-アミノエチル基、3-アミノプロピル基、4-アミノブチル基、カルバモイルメチル基、2-カルバモイルエチル基、3-カルバモイルブチル基、2-グアニジノエチル基、3-グアニジノブチル基、ピロールメチル基、4-イミダゾールメチル基、ピラゾールメチル基、又は3-インドールメチル基を表し、より好ましくは4-アミノブチル基、カルバモイルメチル基、2-カルバモイルエチル基、3-グアニジノブチル基、4-イミダゾールメチル基又は3-インドールメチル基を表し、さらに好ましくは4-イミダゾールメチル基である。
上記式(2)において、R5乃至R7は、それぞれ独立して、水素原子、又は炭素原子数1若しくは2の分枝鎖を有し得る炭素原子数1乃至4のアルキル基、又は-(CH2)n-X基を表し、且つR5乃至R7のうち少なくとも一つ以上が-(CH2)n-X基を表す。nは1乃至4の数を表し、Xはアミノ基、グアニジノ基、-CONH2基、又は窒素原子を1乃至3個有し得る5員環若しくは6員環、又は5員環と6員環から構成される縮合複素環を表す。ここでR5乃至R7の好ましい具体例としては、前出のR2及びR3で定義したものと同じ基が挙げられる。
上記式(3)において、R9乃至R12は、それぞれ独立して、水素原子、又は炭素原子数1若しくは2の分枝鎖を有し得る炭素原子数1乃至4のアルキル基、又は-(CH2)n-X基を表し、且つR9乃至R12のうち少なくとも一つ以上が-(CH2)n-X基を表す。nは1乃至4の数を表し、Xはアミノ基、グアニジノ基、-CONH2基、又は窒素原子を1乃至3個有し得る5員環若しくは6員環、又は5員環と6員環から構成される縮合複素環を表す。ここでR9乃至R12の好ましい具体例としては、前出のR2及びR3で定義したものと同じ基が挙げられる。
なお、本発明において、脂質ペプチド型化合物は、上記式(1)乃至式(3)で表される化合物(脂質ペプチド)又はその薬学的な使用可能な塩のうちの少なくとも1種を用いることがより好ましく、これら化合物は単独で、又は2種以上を組み合わせて用いることができる。
なお、本発明において、多価アルコールを含む場合、多価アルコールは単独で、又は二種以上を組み合わせて用いられ得る。
本発明の組成物は、必要に応じて一般に化粧品用添加剤や、医薬部外品用添加剤及び医薬用添加剤として使用可能な添加剤などを含むことができる。
化粧品、医薬部外品又は医薬等の皮膚外用剤に配合される生理活性物質及び機能性物質等の添加成分としては、例えば顔料、油性基剤、保湿剤、感触向上剤、上記以外の界面活性剤、高分子、増粘・ゲル化剤、溶剤、酸化防止剤、還元剤、酸化剤、防腐剤、抗菌剤、殺菌剤、キレート剤、pH調整剤、酸、アルカリ、粉体、無機塩、紫外線吸収剤、美白剤、ビタミン類及びその誘導体類、育毛用薬剤、血行促進剤、刺激剤、ホルモン類、抗しわ剤、抗老化剤、ひきしめ剤、冷感剤、温感剤、創傷治癒促進剤、刺激緩和剤、鎮痛剤、細胞賦活剤、植物・動物・微生物エキス、鎮痒剤、角質剥離・溶解剤、制汗剤、清涼剤、収れん剤、酵素、核酸、香料、色素、着色剤、染料、消炎剤、抗炎症剤、抗喘息、抗慢性閉塞性肺疾患、抗アレルギー、免疫調整剤、抗感染症剤及び抗真菌剤等が挙げられる。
これらその他添加剤の含有量は、その種類によって種々変化し得るが、組成物の総質量に対して、例えば0.001質量%乃至20質量%、あるいは0.01質量%乃至10質量%程度とすることができる。
ン等のリン脂質類;シリコーン系両性界面活性剤等;高分子界面活性剤としては、ポリビニルアルコール、アルギン酸ナトリウム、デンプン誘導体、トラガントガム、アクリル酸・メタアクリル酸アルキル共重合体;シリコーン系各種界面活性剤が好ましいものとして挙げられる。
本発明の被膜形成組成物は、例えば、少なくとも1種の脂質ペプチド型化合物、水及び所望によりその他の成分を加熱しながら混合、撹拌した後、静置放冷することによって製造され得る。
本発明はまた上記の被膜形成組成物を使用することを特徴とする被膜の製造方法に関する。
被膜を製造する方法としては、被膜を製造することができれば特に制限されず、上記の被膜形成組成物を被塗布物に塗布した後、自然乾燥や加熱乾燥などにより乾燥させる方法などが挙げられる。
<試薬>
メタノール:関東化学株式会社(特級)
i-プロパノール:関東化学株式会社(1級)
トルエン:関東化学株式会社(1級)
酢酸:関東化学株式会社(1級)
グリシンメチルエステル塩酸塩:東京化成工業株式会社
L-ヒスチジン:東京化成工業株式会社、協和発酵バイオ株式会社
ナトリウムメトキサイド 28%メタノール溶液:和光純薬工業株式会社(28% ナトリウムメトキシドメタノール溶液)
炭酸ナトリウム:純正化学株式会社(1級)
塩酸:関東化学株式会社(1級)
アセトニトリル:関東化学株式会社(特級)
ISTA:2-(4,4-ジメチルペンタン-2-イル)-5,7,7-トリメチルオクタン酸[日産化学工業(株)製ファインオキソコール(登録商標)イソステアリン酸]
2-ヘプチルウンデカン酸:[日産化学工業(株)製]
塩化チオニル:東京化成工業株式会社
アセトン:関東化学株式会社(1級)
テトラヒドロフラン:関東化学株式会社(1級)
酢酸エチル:関東化学株式会社(1級)
N,N-ジメチルホルムアミド(DMF):関東化学株式会社(1級)
<分析機器>
NMR:JNM-ECP300(日本電子株式会社製)
LC-MS:Waters ACQUITY
pHメーター:メトラートレド製
500mLの4つ口フラスコに、L-ヒスチジン14.2g(91.6mmol)、N-パルミトイル-Gly-メチル30.0g(91.6mmol)、トルエン300gを投入し、塩基であるナトリウムメトキサイド 28%メタノール溶液35.3g(183.2mmol)を加え、油浴で60℃に加熱し1時間撹拌を続けた。その後、油浴を外し、25℃まで放冷し、この溶液をアセトン600gで再沈殿させ、ろ取した。ここで得られた固体を、水600gとメタノール750gの混合溶液に溶解し、ここに6規定塩酸30.5mL(183.2mmol)を加えて中和し固体を析出させ、ろ過した。次に、得られた固体をテトラヒドロフラン120gと水30gの混合溶液に60℃で溶解させ、酢酸エチル150gを加え、60℃から30℃まで冷却した。その後、析出した固体をろ過した。さらに得られた固体を、テトラヒドロフラン120gとアセトニトリル60g溶剤中に溶解し、60℃に加熱し、1時間撹拌した後に冷却し、ろ過した。ここで得られた固体を水120gで洗浄し、ろ過後に減圧乾燥を行いN-パルミトイル-Gly-Hisフリー体(以下、単にPal-GHとも称する)の白色の結晶、26.9g(収率65%)を得た。
100mL四ツ口フラスコに、ISTA20.0g(70.3mmol)、DMF154mg、トルエン20.0gを仕込み、0℃に温調した。塩化チオニル8.78g(73.8mmol)を15分かけて滴下した後に、徐々に30℃に昇温した。30℃で2時間撹拌した後、トルエン20.0gを仕込んだ。50℃に昇温し20gのトルエンを減圧留去することで塩化チオニルを除去する操作を2回繰り返した(酸クロライド溶液)。
上記反応容器とは別に、500mL四ツ口フラスコにイオン交換水40.0gとグリシンメチルエステル塩酸塩11.5g(91.4mmol)、トルエン80.0gを投入し、塩基である炭酸ナトリウム8.6g(80.9mmol)、イオン交換水60.0gを投入し、攪拌した。その後、この中に上記酸クロライド溶液を反応温度25±5℃で1時間かけて滴下し、25℃で3時間攪拌した後、10%食塩水200.0gを追加して60℃で分液操作を行った。得られた有機層にトルエン160.0gを追加し、共沸脱水を行い、2-(4,4-ジメチルペンタン-2-イル)-5,7,7-トリメチルオクタノイルグリシンメチルエステル24.5g(収率98%)を得た。
500mL四ツ口フラスコに、L-ヒスチジン10.7g(68.9mmol)とトルエン49.0gを加え、塩基であるナトリウムメトキサイド 28%メタノール溶液12.6g(65.5mmol)を滴下したところに、合成例2(1)で得られた2-(4,4-ジメチルペンタン-2-イル)-5,7,7-トリメチルオクタノイルグリシンメチルエステル24.5g(68.9mmol)を、メタノール9.8gとともに投入し70℃に昇温した。その後、塩基であるナトリウムメトキサイド 28%メタノール溶液10.0g(51.7mmol)の滴下を開始し、約70℃で3時間攪拌を続けた。反応終了後pH調整のため酢酸を5.0g(82.6mmol)加えてpH7に調整し、イオン交換水122.5gとi-プロパノール49.0gを加えて60℃で分液を行った。得られた水層に6N塩酸13.8g(82.7mmol)を加え、メタノール24.5gを添加してからアセトニトリル489.8gに再沈殿を行った。得られたスラリーをろ過し、湿品をメタノールで溶解させた後に活性炭(特製白鷺)4.9gを投入して1時間撹拌した後に濾別し、再度アセトニトリル489.8gに再沈殿を行った。得られた湿品を60℃で減圧乾燥し、2-(4,4-ジメチルペンタン-2-イル)-5,7,7-トリメチルオクタノイルグリシルヒスチジン(以下、単にFO-GHとも称する)26.6g(収率86%)を得た。
・1H-NMR(300MHz,DMSO-d6,δppm):8.02(2H,m),7.55(1H,s),6.80(1H,s),4.38(1H,m),3.68(2H,m),2.88(2H,m),1.94(1H,m),1.51(4H,m),1.12(3H,m),0.87(27H,m)
・MS(ESI)m/z:479.29(M+H+)
原料に2-ヘプチルウンデカン酸を用いた以外は合成例2(1)と同様の操作で、2-ヘプチルウンデカン酸グリシンメチルエステル24.1g(収率96%)を得た。
2-(4,4-ジメチルペンタン-2-イル)-5,7,7-トリメチルオクタノイルグリシンメチルエステルの替りに2-ヘプチルウンデカン酸グリシンメチルエステルを用いた以外は合成例2(2)と同様の操作で、2-ヘプチルウンデカノイルグリシルヒスチジン(以下、単にIS-Ste-GHとも称する)23.1g(収率86%)を得た。
・1H-NMR(300MHz,DMSO-d6,δppm):8.07(1H,t,J=6.0Hz),8.04(1H,d,J=7.5Hz),7.57(1H,s),6.82(1H,s),4.39(1H,q,J=7.5Hz),3.68(2H,m),2.28(2H,m),2.16(1H,m),1.42(2H,m),1.21(26H,m),0.85(3H,m)
・MS(ESI)m/z:479.18(M+H+)
Pal-GH、添加剤としてステアリン酸、1,2-ヘキサンジオール、ポリオキシエチレンラウリルエーテル(日光ケミカルズ社製)及び水を、下記の表1に示す割合にて300mLビーカーに加え、液温80℃、200rpmで加熱撹拌を行い、均一溶液とした。撹拌しながら、冷却を行い、液温が60℃になったところで静置放冷を行い、ES-01プレミックスを調製した。
表2に従い、300mLトールビーカーに、アルギン酸プロピレングリコール、POE60硬化ヒマシ油、ミネラルオイル及び純水を入れ、80℃で加熱撹拌を行った。なお撹拌はアズワン(株)製のLABORATORY HIGH MIXERを用いて、200rpmで行った。次いで、80℃に加熱したES-01プレミックスを加え、さらに加熱撹拌を5分間行った。加熱撹拌終了後、液温が約50℃程度になるまで撹拌冷却し、ゲル形成を確認した。ゲル形成の確認は試験管倒置法により行い、分散液の流動性が失われて、トールビーカーを倒置しても液が流れ落ちない状態をゲル化と判定した。
表2に従い、300mLトールビーカーに、アルギン酸プロピレングリコール、POE60硬化ヒマシ油、ミネラルオイル及び純水を入れ、80℃で加熱撹拌を行った。なお撹拌はアズワン(株)製のLABORATORY HIGH MIXERを用いて、200rpmで行った。加熱撹拌終了後、液温が約50℃程度になるまで撹拌冷却した。
表3に従い、300mLトールビーカーに、アルギン酸プロピレングリコール、POE60硬化ヒマシ油、及び純水を入れ、80℃で加熱撹拌を行った。なお撹拌はアズワン(株)製のLABORATORY HIGH MIXERを用いて、200rpmで行った。次いで、80℃に加熱したES-01プレミックスを加え、さらに加熱撹拌を5分間行った。加熱撹拌終了後、液温が約50℃程度になるまで撹拌冷却し、ゲル形成を確認した。ゲル形成の確認は試験管倒置法により行い、分散液の流動性が失われて、トールビーカーを倒置しても液が流れ落ちない状態をゲル化と判定した。
表3に従い、300mLトールビーカーに、アルギン酸プロピレングリコール、POE60硬化ヒマシ油、及び純水を入れ、80℃で加熱撹拌を行った。なお撹拌はアズワン(株)製のLABORATORY HIGH MIXERを用いて、200rpmで行った。加熱撹拌終了後、液温が約50℃程度になるまで撹拌冷却した。
表4に従って、実施例2で得られたゲル又は比較例2の液体を秤量し、そこへスクワランを添加した。添加後スパチュラを用いて均一の増粘物又は液体を得た。
そして、バイオスキン((株)ビューラックス製)上に表4に示した実施例3の増粘物又は比較例3の液体を10mg/cm2となるように塗布し、35℃の恒温槽内にて乾燥した。変角光度計GP-5((株)村上色彩技術研究所製)を用いて、入射光-30°とした場合の30°の反射光強度を測定した。
また、実施例3及び比較例3の外観写真およびSEM画像をそれぞれ図2及び図3に示す。
本発明の被膜形成組成物は、光の反射を抑制し得る被膜を形成することができた。
表5に従い、300mLトールビーカーにA相を秤量した。一方で、70℃に加温した精製水に対して、同様に加熱溶解させたES-01プレミックスを秤量し、B相を調製した。70℃に加熱したA相に対して、B相を添加した。その後、液温が40℃程度になるまで撹拌冷却した。
表6に従い、300mLトールビーカーに各成分を秤量した。70℃に加熱し、均一溶液となったところで、液温が40℃程度になるまで撹拌冷却した。そして、実施例4と同様の手順で、保湿率を求めた。その結果を図4に示す。ここで、Pal-Hは、パルミトイルヒスチジンを表す。
表7に従い、B相及びC相を加熱溶解した。D相を加熱し、ホモミキサー(5000rpm、3分間)処理を実施した後に、前述の加熱したB相およびC相を添加し、さらにホモミキサー(5000rpm、3分間)処理を実施した。ホモミキサー処理後に、A相をゆっくりと加え、さらにホモミキサー(5000rpm、3分間)処理を行った。その後、液温が40℃程度になるまで撹拌冷却した。
本発明の被膜形成組成物は、良好な撥水性を有する被膜を形成することができた。
表8に従いA相を秤量し、75℃に加熱することで均一な溶液とした。他方、B相のES-01プレミックスを除く成分を75℃に加温し、そこへ75℃に加温したES-01プレミックスを添加した。次いで、75℃に加温しているA相へB相を添加し、室温付近まで撹拌冷却を行った。
また、試験終了後の毛髪を走査型電子顕微鏡(SEM)観察した。その結果を図7に示す。
また、図7に示したSEM画像により、脂質ペプチド型化合物を添加したコンディショナーは毛髪上に被膜を形成していることが確認できた。一方、脂質ペプチド型化合物未添加のコンディショナーは毛髪上に被膜を形成しなかった。
実施例11として、上記表8に従い、実施例10の組成物と同じ組成の組成物を調製した。また、比較例7として、比較例6の組成物と同じ組成の組成物を調製した。
実施例11又は比較例7の組成物(コンディショナー)10gを、人毛毛束(黒髪、1g、10cm)(毛束屋社製)全体に均一に塗布し、5分後、塗布したコンディショナーを洗浄した。1晩室温で静置後、触感系TYPE33(新東科学(株)社)を用いて摩擦抵抗力を測定し、指通りの指標とした。
実施例11及び比較例7の摩擦抵抗力を図8に示す。参考例1として、コンディショナー未処理の人毛毛束の摩擦抵抗力も同条件にて測定を行った。
本発明の被膜形成組成物は摩擦抵抗力が小さい被膜、すなわち、良好な指通し性や櫛通し性を有する被膜を形成することができた。
下記表15に従い、顔料17質量%を均一に分散したO/Wリキッドファンデーションを調製した。なお、表15中の各成分の量は、O/Wリキッドファンデーションの総質量に基づく質量%(wt%)を表す。
200mLビーカー(HARIO株式会社製)中でフレッシュカラーベースAQUA 34.0g、精製水140.6g、ステアロイルメチルタウリンNa(NIKKOL SMT)0.2g、LPA(レシチン)0.2g、エデト酸二ナトリウム(EDTA-2Na)0.2g、ヒドロキシプロピルセルロース0.4g、キサンタンガム(KELTROL CG-SFT)0.4gを混合し、75℃で10分間加熱撹拌した(B相)。200mLビーカー(HARIO株式会社製)中でNIKKOL ニコムルス41 4.0g、NIKKOL NATURAL OILS 14.0g、NIKKOL ニコガード88 1.0gを加えて75℃で10分間加熱撹拌した(A相)。B相に対してA相をゆっくりと撹拌しながら添加し、出来上がった混合物を75℃中でホモミキサー(みづほ工業株式会社製QUICK HOMO MIXER LR-1A)を用いて5000rpmで3分間乳化処理を行った。さらに、75℃に加熱したES-01プレミックス1.0g(D相)を添加して、75℃中で2分間加熱撹拌した後に、40℃になるまで室温で撹拌冷却を行った。40℃になったことを確認し、NIKKOL NET-813-1 4.0g(C相)を添加して、35℃になるまで撹拌冷却を行った。以上の工程において、撹拌は全て200rpmで行った。
下記表15に従い、顔料17質量%を均一に配合したO/Wリキッドファンデーションを調製した。
200mLビーカー(HARIO株式会社製)中でフレッシュカラーベースAQUA 34.0g、精製水141.6g、ステアロイルメチルタウリンNa(NIKKOL SMT)0.2g、LPA(レシチン)0.2g、エデト酸二ナトリウム(EDTA-2Na)0.2g、ヒドロキシプロピルセルロース0.4g、キサンタンガム(KELTROL CG-SFT)0.4gを混合し、75℃で10分間加熱撹拌した(B相)。200mLビーカー(HARIO株式会社製)中でNIKKOL ニコムルス41 4.0g、NIKKOL NATURAL OILS 14.0g、NIKKOL ニコガード88 1.0gを加えて75℃で10分間加熱撹拌した(A相)。B相に対してA相をゆっくりと撹拌しながら添加し、出来上がった混合物を75℃中でホモミキサー(みづほ工業株式会社製QUICK HOMO MIXER LR-1A)を用いて5000rpmで3分間乳化処理を行った。その後、40℃になるまで室温で撹拌冷却を行い、NIKKOL NET-813-1 4.0g(C相)を添加し、35℃になるまで撹拌冷却を行った。以上の工程において、撹拌は全て200rpmで行った。
実施例13として、実施例12のO/Wリキッドファンデーションを、また比較例9として、比較例8のO/Wリキッドファンデーションをそれぞれ用いて、反射光強度を測定した。
バイオスキン((株)ビューラックス製)上に実施例13のО/Wリキッドファンデーション又は比較例9のО/Wリキッドファンデーションを10mg/cm2となるように塗布し、35℃の恒温槽内にて乾燥した。変角光度計GP-5((株)村上色彩技術研究所製)を用いて、入射光-45°とした場合の45°の反射光強度を測定した。測定結果を図9に示す。
本発明の被膜形成組成物は、光の反射を抑制し得る被膜を形成することができた。
実施例14の組成物として、実施例12のO/Wリキッドファンデーションを、また比較例10の組成物として、比較例8のO/Wリキッドファンデーションをそれぞれ用いた。
ヒト皮膚上に、実施例14のО/Wリキッドファンデーション又は比較例10のО/Wリキッドファンデーションを0.5g塗布し、均一に伸ばした後に、人工皮脂を0.5g滴下して30分室温静置した。30分室温静置後の、実施例14及び比較例10の外観写真を図10に示す。
本発明の被膜形成組成物は、皮脂との会合を抑制し、化粧ヨレが発生し難い被膜を形成することができた。
下記表16に従い、リンゴ酸1.5質量%(wt%)を配合したファンデーションを調製した。
200mLビーカー(HARIO株式会社製)中でA相を75℃まで加熱撹拌した。B相とC相とを200mLビーカー中で混合し、75℃まで加熱撹拌した。B相とC相との混合相に対してA相を添加し、75℃中でホモミキサー(みづほ工業株式会社製QUICK HOMO MIXER LR-1A)を用いて5000rpmで5分間乳化処理を行った。その後、75℃に加熱したD相を添加し、75℃で5分間加熱撹拌を行い、40℃になるまで撹拌冷却を行った。以上の工程において、撹拌は全て200rpmで行った。
(1)ダメージヘアサンプル(ダメージ毛髪)の作製手順
人毛黒髪(BS-B-A、(株)ビューラックス製)50束(1束当たり、約10cm、約1g)に対し、ギャツビーEXハイブリーチ((株)マンダム製)を1セット(パウダー18g、水70mL、クリーム35gの混合物)塗布し、1時間室温で放置することで髪の脱色処理を行った。その後、髪を蒸留水で洗浄し、65℃に設定した定温乾燥機(OF-300B、アズワン(株)製)で30分間乾燥させた。上記操作を計三回繰り返した。その後毛髪を、ドデシル硫酸ナトリウム(富士フイルム和光純薬(株)製)1質量%を蒸留水に溶解させた水溶液1Lに浸漬させ、蒸留水で洗浄した。ケイドライ(日本製紙クレシア(株)製)で余分な水を取った後、室温で一晩乾燥させることで、ダメージヘアサンプル(ブリーチ髪)を作製した。
調製した上記ダメージヘアサンプルを蒸留水で濡らした後、ケイドライ(日本製紙クレシア(株)製)で余分な水を取った。実施例15又は比較例11で調製したサンプルを1gずつ手に取って髪に塗布し、5分間静置した。その後、300mLの水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な水を取り、室温で一晩乾燥させた。
(3)毛髪の保湿試験
上記ヘアサンプルを1cmの長さに切り揃え、薬包皿に0.7g量り取り、65℃に設定した定温乾燥機(OF-300B、アズワン(株)製)で40分間乾燥させた。その後、再び質量を計測し、毛髪の質量減少量を水分減少量として、髪からの水分損失率を下記式から算出した(表17)。
水分損失率(%)=(1-(乾燥後の毛髪質量)/(乾燥前の毛髪質量))*100
マイクロスライドガラスS1111(松浪硝子工業(株)製)上にナイスタック(登録商標)NW-10(ニチバン(株)製)を用いて、黒色の画用紙PI-N86D((株)マルアイ製)を貼り付けた。その上に、上記保湿試験後の毛髪サンプルを平行に隙間なく並べ、ナイスタック(登録商標)NW-10(ニチバン(株)製)で張り付けた。反射光分布の測定は、変角光度計GP-5((株)村上色彩技術研究所製)にて実施した。測定入射光は-45°とし、45°における反射光強度を測定した。
実施例18として、実施例16の(2)で処理された人毛を、また比較例14として、比較例12の(2)で処理された人毛を用いて、人毛中のリンゴ酸の定量を評価した。
リンゴ酸の定量は、BioAssaySystems社製のEnzyChromTM Malate Assay Kit(EMAL-100)により評価した。
実施例18又は比較例14の人毛を50mLサンプル管に200ミリグラム秤量し、精製水4mLを加えて60分間超音波処理することでリンゴ酸を抽出した。96wellプレートに各抽出液20μL、アッセイバッファー74μL、EnzymeA 1μL、EnzymeB 1μLとNAD/MTT試薬8μLを添加した。15分後、上記手順で調製したwell中の吸光度(565nm)をプレートリーダー(TECAN社製)にて測定し、サンプル自身の吸光度を引くことで正味の吸光度を算出した。試験の結果を図11に示す。
実施例19および比較例15として、実施例10および比較例6で調製したコンディショナー1gを実施例16の(1)と同様の手順で作成したダメージ毛髪の束(10cm、50本)に塗布し、5分間静置した。その後、300mLの精製水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な精製水を取り、室温で一晩乾燥させた。走査型電子顕微鏡(SEM)[Miniscope(登録商標)TM3000((株)日立ハイテクノロジーズ製)]を用いて、上記各毛髪表面観察を実施した。結果を図12に示す。ブリーチ処理をしたダメージ毛髪や比較例15と比較して実施例19は毛髪表面が滑らかになっている様子が観測された。
実施例20として、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に、実施例16の(2)と同様の処理を施した毛束を1cmにせん断した後に、16mgの毛髪を秤量し、水分吸脱着量を水分吸脱着測定装置IGAsorp(ハイデンアイソケヤマ社製)を用いて評価した。温度25℃、湿度40%の条件下において、各試料の湿潤重量を測定した後、温度25℃、湿度0%条件下において乾燥を行い、試料重量の変化率が0.0001mg/minを下回るまで乾燥を継続した。相対湿度を0%から90%まで10%刻みで段階的に上昇させ、各相対湿度点における試料の平衡重量から毛髪中の水分率を算出した。更に、相対湿度を90%から0%まで10%刻みで段階的に降下させた場合にも、各相対湿度点における試料の平衡重量から毛髪中の水分率を算出した。各湿度での最大平衡化時間を360分間とし、上記手順はプログラムによる自動測定で行い、試料の平衡重量の決定は、LDF(線形推進力近似)モデルに基づく漸近線法を採用し、緩和状態99%下における重量値を算出することを目的とし、LDFモデルが適用されなかった場合の試料の平衡重量は、180分間測定後の測定値を移動平均化して算出したものを採用した。得られた水分率の結果を表19(湿度上昇時)と表20(湿度降下時)に示す。比較例16として、比較例12の(2)で処理された人毛結果と、ダメージ毛髪の結果も、表19に示した。コンディショナー処理していないダメージ毛髪と比較して、比較例16は水分の吸着を抑制し、実施例19は更に抑制する傾向であった。
実施例21として、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に、実施例16の(2)と同様の処理を施した毛束表面の動摩擦係数を測定し、指通りの指標とした。動摩擦係数はKES-SE摩擦感テスター(カトーテック(株)社製)を用いて測定し、センサーは10mm角のシリコンセンサーを用いた。比較例12の(2)と同様の処理をした毛束についても、測定を行い、比較例17とした。両者の結果を図13に示す。実施例21に施した毛髪は、比較例17と比較して毛髪表面の動摩擦係数が低い傾向であった。
下記表21に従い、コハク酸1.5質量%を配合したコンディショナーを調製した。200mLビーカー(HARIO株式会社製)中でA相を75℃まで加熱撹拌した。B相とC相を200mLビーカー中で混合し、75℃まで加熱撹拌した。B+C相に対してA相を添加し、75℃中でホモミキサー(QUICK HOMO MIXER LR-1Aみづほ工業株式会社製)を用いて5,000rpmで5分間乳化処理を行った。その後、75℃に加熱したD相を添加し、75℃で5分間加熱撹拌を行い、40℃になるまで撹拌冷却を行った。以上の工程において、撹拌は全て200rpmで行った。
実施例23および比較例19として、実施例22および比較例18で調製したコンディショナーを1gずつ手に取って、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に塗布し、5分間静置した。その後、300mLの水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な水を取り、室温で一晩乾燥させた。上記処理を1回実施、2回実施、3回実施した3種類のサンプルを作製した。作製した各ダメージヘアサンプルを1cmの長さに切り揃えた。コハク酸の定量は、Succinate colorimetric Assay Kit(Megazyme社製)を用いて評価した。実施例23及び比較例19で処理された人毛をそれぞれ50mLサンプル管に200mg秤量し、精製水4mLを加えて60分間超音波処理することでコハク酸を抽出した。96wellプレートに各抽出液10μL、精製水210μL、Buffer 20μL、NADH 20μL、ATP/PEP/CoA 20μL、PK/L-LDH 2μLを添加し、3分間静置後にSCSを2μL加えた。上記手順で調製したwell中の吸光度(340nm)をプレートリーダー(TECAN社製)にて測定し、サンプル自身の吸光度を引くことで正味の吸光度を算出した。試験の結果を図14に示す。処理日数依存的に、毛髪からのコハク酸抽出量は向上し、比較例19と比較して実施例23の人毛からは、約2倍のコハク酸が抽出された。
実施例24および比較例20として、実施例22および比較例18で調製したコンディショナーを1gずつ手に取って実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に塗布し、5分間静置した。その後、300mLの水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な水を取り、室温で一晩乾燥させた。上記の処理を3日間繰り返して実施し、飛行時間型二次イオン質量分析計TOF.SIMS5((株)日立ハイテクサイエンス社製)を用いてコハク酸およびES-01プレミックス中に含まれる脂質ペプチドの分析を行った。各毛髪サンプルの毛髪中央付近をサンプリングし、断面を作成した。一次イオン源はBiを選択した。結果の毛髪断面図を図15に示す。比較例20と比較して、実施例24の毛髪断面からは高いコハク酸強度が確認できた。また、実施例24は、ES-01プレミックス中の脂質ペプチドが毛髪表面に局在していることが確認できた。
実施例25および比較例21として、実施例22および比較例18で調整したコンディショナーを1gずつ手に取って、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に塗布し、5分間静置した。その後、300mLの水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な水を取り、室温で一晩乾燥させた。上記毛髪サンプルの表面ゼータ電位を固体表面ゼータ電位装置SurPASS(Anton-Paar社製)により評価した。電解質には1mM KClを調製し、温度23℃の条件で測定した。結果を表22に示す。比較例21は、ブリーチ処理を行ったダメージ毛髪と比較して表面電位がカチオン性に寄り、実施例25はさらにその値がカチオン性になることが確認できた。
実施例26および比較例22として、実施例22および比較例18で調整したコンディショナーを1gずつ手に取って、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に塗布し、5分間静置した。その後、300mLの水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な水を取り、室温で一晩乾燥させた。上記各毛髪サンプル表面の硬さをハイジトロンTI980トライボインデンター(Bruker社製)により測定した。押し込み深さを1μmに設定、圧子はバーコビッチ型を用い、10回測定した平均値を算出した。結果を図16に示す。ブリーチ処理をしたダメージ毛髪は、未処理である黒髪と比較して毛髪の表面が硬くなることが示された。ダメージ毛髪と比較して比較例22は毛髪表面が柔らかくなり、実施例26はさらに表面が柔らかくなることが示された。
実施例27および比較例23として、実施例22および比較例18で調製したコンディショナーを1gずつ手に取って、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に塗布し、5分間静置した。その後、300mLの水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な水を取り、室温で一晩乾燥させた。作製した処理サンプルを1cmにせん断し、3mg秤量した人毛の水分吸脱着量を水分吸脱着測定装置IGAsorp(ハイデンアイソケヤマ社製)を用いて評価した。相対湿度0%中で2日間毛髪を乾燥させ、なりゆきで相対湿度90%まで一気に湿度を上げた際の質量変化量を測定した。上記手順はプログラムによる自動測定で行った。結果を図17に示す。コンディショナー処理していないダメージ毛髪と比較して、比較例23は水分の吸着を抑制し、実施例27は更に抑制する傾向であった。
実施例28として、実施例12のO/Wリキッドファンデーションを、また比較例24として、比較例8のO/Wリキッドファンデーションをそれぞれ用いて、皮脂浮きによる経時的なテカリの変化を評価した。実施例12及び比較例8のO/Wリキッドファンデーションを被験者の顔面に塗布し、普段通りの生活を行い、各ファンデーションを塗布して1時間後から1時間おきに6時間後まで、顔面の表面光沢をGlossymeter GL200(Courage+khazaka electronic社製)を用いて測定した。結果を図18に示す。実施例28は比較例24と比較して、6時間後も経時的な皮脂浮きによるテカリを抑制した。
下記表23に従い、ヒアルロン酸を0.005%配合した美容液を調製した。200mLビーカー(HARIO株式会社製)中でA相を75℃まで加熱撹拌した。B相、C相、D相を200mLビーカー中で混合し、75℃まで加熱撹拌した。B+C+D相に対してA相を添加し、75℃中で5分間撹拌した。その後、45℃になるまで撹拌冷却し、E相を添加して35℃になるまで撹拌冷却を行った。以上の工程において、撹拌は全て200rpmで行った。
実施例30として、実施例29の美容液を、また比較例26として、比較例25の美容液を用いて、保湿効果を評価した。実施例29及び比較例25の美容液を被験者の前腕の内側に塗布し、湿度50%、温度24℃の環境下にて、美容液を塗布して1時間後から1時間おきに7時間後まで前腕の角質水分量をCorneometer CM825(Courage+khazaka electronic社製)を用いて測定した。塗布した前腕部分から無作為に10か所を抽出して測定し、その平均値を測定結果とした。結果を図19に示す。実施例30は比較例26と比較して、7時間後も角質中に高い水分量を保持しており、保湿効果があることを確認した。
下記表24に従い、ケラチン1.5質量%を配合したコンディショナーを調製した。200mLビーカー(HARIO株式会社製)中でA相を75℃まで加熱撹拌した。B相とC相を200mLビーカー中で混合し、75℃まで加熱撹拌した。B+C相に対してA相を添加し、75℃中でホモミキサー(QUICK HOMO MIXER LR-1Aみづほ工業株式会社製)を用いて5000rpmで5分間乳化処理を行った。その後、75℃に加熱したD相を添加し、75℃で5分間加熱撹拌を行い、40℃になるまで撹拌冷却を行った。以上の工程において、撹拌は全て200rpmで行った。
実施例33、実施例34および比較例28として、実施例31、実施例32および比較例27で調整したコンディショナーを1gずつ手に取って、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に塗布し、5分間静置した。その後、300mLのメタノール(純正化学(株)製)を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分なメタノールを取り、室温で一晩乾燥させた。作製した各ダメージヘアサンプルを1cmの長さに切り揃えた。ケラチンの定量は、Keratin Orange α-Keratin Assay Kit(biocolor社製)を用いて評価した。実施例及び比較例で処理された人毛をそれぞれ1.5mLマイクロチューブ(eppendorf社製)に5mgずつ秤量し、1mLずつDigestion reagentを加えて1時間ボルテックスで混和した。Reference standard(α―Keratin 5.0mg/mL)をDigestion reagentで希釈し、検量線用試料を調製した(0、1、2、3、4、5mg/mL)。毛髪試料および検量線試料200μLにNeutralising solution(1M HCl)を200μLずつ加え、各溶液のpHを中和した。試料にDye reagentを50μLずつ加え、ボルテックスで混和した後に、30分間静置して反応させた。各毛髪試料から抽出されたケラチンを塩析させるため、試料にSaturated solution((NH4)2SO4)を450μLずつ加えた。結合しなかったOrange Gを除去するため、試料を12,000rpmで10分間遠心した後に、上清を捨て、Digestion reagentを50μLずつ加え、ボルテックスで十分に混和させてケラチンを可溶化した。調製した各試料を200μLずつ96wellプレートに移し、吸光度(480nm)にてプレートリーダー(TECAN社製)により測定し、サンプル自身の吸光度を引くことで正味の吸光度を算出した。試験の結果を図20に示す。比較例28と比較して実施例33および実施例34の毛髪からは、高いケラチン量が抽出された。
下記表25に従い、18-メチルエイコ酸(18-MEA)1.5質量%を配合したコンディショナーを調製した。200mLビーカー(HARIO株式会社製)中でA相を75℃まで加熱撹拌した。B相とC相を200mLビーカー中で混合し、75℃まで加熱撹拌した。B+C相に対してA相を添加し、75℃中でホモミキサー(QUICK HOMO MIXER LR-1Aみづほ工業株式会社製)を用いて5000rpmで5分間乳化処理を行った。その後、75℃に加熱したD相を添加し、75℃で5分間加熱撹拌を行い、40℃になるまで撹拌冷却を行った。以上の工程において、撹拌は全て200rpmで行った。
実施例36および比較例30として、実施例35および比較例29で調製したコンディショナーを1gずつ手に取って、実施例16の(1)と同様の手順で作成したダメージ毛髪の1束(約10cm、約1g)に塗布し、5分間静置した。その後、300mLの精製水を入れた300mLトールビーカー内で揺らすように洗浄し、ケイドライ(日本製紙クレシア(株)製)で余分な精製水を取り、室温で一晩乾燥させた。この操作を10回繰り返した後に、ショットキー電界放出形走査電子顕微鏡JSM-7800F(日本電子(株)製)を用いて作成した各毛髪表面観察を実施した。サンプルの固定はカーボンテープを用い、加速電圧は0.7kVの条件で測定した。ダメージ毛髪の測定結果と併せて図21に示す。ダメージ毛髪や比較例30はキューティクル表面の構造が観察できるのに対し、実施例36はキューティクル表面が被膜で覆われている様子が確認できた。
Claims (5)
- 低分子脂質ペプチド又はその薬学的に使用可能な塩からなる少なくとも1種の脂質ペプチド型化合物を含有することを特徴とする被膜形成組成物。
- 前記脂質ペプチド型化合物が、炭素原子数9乃至23の脂肪族基からなる脂質部に、アミノ酸の繰り返し結合構造を有するペプチド部が結合された化合物である、請求項1に記載の被膜形成組成物。
- 前記脂質ペプチド型化合物が、下記式(1)乃至式(3)で表される化合物又はその薬学的に使用可能な塩のうちの少なくとも1種からなることを特徴とする、請求項2に記載の被膜形成組成物。
- 請求項1乃至請求項3のいずれか1項に記載の被膜形成組成物を使用することを特徴とする被膜の製造方法。
- 請求項1乃至請求項3のいずれか1項に記載の被膜形成組成物を用いて作成された被膜。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020217002238A KR20210025070A (ko) | 2018-06-29 | 2019-06-28 | 피막형성 조성물 |
| CN201980056353.4A CN112638359A (zh) | 2018-06-29 | 2019-06-28 | 被膜形成用组合物 |
| EP22155607.9A EP4014958A1 (en) | 2018-06-29 | 2019-06-28 | Coating film-forming composition |
| EP19827474.8A EP3808329A4 (en) | 2018-06-29 | 2019-06-28 | COATING FILM FORMING COMPOSITION |
| US17/256,729 US20210259941A1 (en) | 2018-06-29 | 2019-06-28 | Coating film-forming composition |
| JP2020527698A JP7460962B2 (ja) | 2018-06-29 | 2019-06-28 | 被膜形成組成物 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018125136 | 2018-06-29 | ||
| JP2018-125136 | 2018-06-29 | ||
| JP2018233469 | 2018-12-13 | ||
| JP2018-233469 | 2018-12-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020004649A1 true WO2020004649A1 (ja) | 2020-01-02 |
Family
ID=68984647
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/025927 WO2020004649A1 (ja) | 2018-06-29 | 2019-06-28 | 被膜形成組成物 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20210259941A1 (ja) |
| EP (2) | EP3808329A4 (ja) |
| JP (1) | JP7460962B2 (ja) |
| KR (1) | KR20210025070A (ja) |
| CN (1) | CN112638359A (ja) |
| TW (1) | TW202017558A (ja) |
| WO (1) | WO2020004649A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022065433A1 (ja) * | 2020-09-28 | 2022-03-31 | 日産化学株式会社 | 毛髪用化粧料 |
| WO2022092284A1 (ja) * | 2020-10-30 | 2022-05-05 | 日産化学株式会社 | 脂質ペプチドとショ糖エステルを含む組成物 |
| KR20220106873A (ko) * | 2021-01-22 | 2022-08-01 | 주식회사 코스메카코리아 | 미세먼지 흡착방지용 화장료 조성물의 제조방법 및 그에 의해 제조된 미세먼지 흡착방지용 화장료 조성물 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115612321B (zh) * | 2022-09-27 | 2024-01-19 | 常州君合科技股份有限公司 | 一种环保型水性锌铝防腐涂液及其制备方法 |
| CN115975483B (zh) * | 2022-12-19 | 2023-08-22 | 浦诺菲新材料有限公司 | 自修复涂层涂布液的制备方法,以及汽车漆面保护膜 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1077210A (ja) | 1996-09-06 | 1998-03-24 | Nakanihon Seni Kogyo Kyodo Kumiai | 化粧料用組成物及びその製造方法並びにこれを配合した化粧品 |
| JP2002308756A (ja) | 2001-03-05 | 2002-10-23 | L'oreal Sa | ポリアミノ酸の保湿剤としての用途とそれを含有する化粧品用又は製薬用組成物 |
| WO2010013555A1 (ja) * | 2008-08-01 | 2010-02-04 | 日産化学工業株式会社 | 新規脂質ジペプチド並びにゲル |
| JP2010047534A (ja) * | 2008-08-22 | 2010-03-04 | Kyushu Univ | 酵素応答性ゲル |
| WO2010106981A1 (ja) * | 2009-03-16 | 2010-09-23 | 日産化学工業株式会社 | 低分子ゲル化剤からなるスプレー用基材 |
| JP2016006030A (ja) | 2014-05-30 | 2016-01-14 | 花王株式会社 | 皮膚化粧料 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2760634B1 (fr) * | 1997-03-14 | 2002-10-11 | Oreal | Composition aqueuse topique solide ayant l'aspect d'un gel permettant la formation d'un film lors de son application |
| DE602005013422D1 (de) * | 2004-11-22 | 2009-04-30 | Symrise Gmbh & Co Kg | Formulierungen mit ceramiden und/oder pseudoceramiden und (alpha-)bisabolol zur behandlung von hautschäden |
| FR2911779B1 (fr) * | 2007-01-30 | 2009-04-24 | Lvmh Rech | Composition contenant un extrait d'ambre |
| JP2009073764A (ja) * | 2007-09-20 | 2009-04-09 | Fujifilm Corp | 生体用粘着ゲルシートおよびそれを用いたシート状化粧料 |
| JP2014503551A (ja) * | 2011-01-10 | 2014-02-13 | ダウ コーニング コーポレーション | パーソナルケア及びヘルスケア組成物 |
| BR112016014595B1 (pt) * | 2013-12-25 | 2021-05-18 | Nissan Chemical Industries, Ltd | material à base de sólido para uso tópico sobre a pele,composição aquosa e método de produção do mesmo. |
| JP2016027035A (ja) * | 2014-06-30 | 2016-02-18 | ロート製薬株式会社 | 皮膚外用組成物、化粧料及び皮膚外用組成物の変色抑制方法 |
| JP6975516B2 (ja) * | 2014-06-30 | 2021-12-01 | ロート製薬株式会社 | 外用組成物、化粧料、経皮吸収促進用組成物、外用組成物における有効成分の経皮吸収性を高める方法、経皮投与型医薬及び点眼用組成物 |
| EP3212688B1 (en) * | 2014-10-31 | 2022-04-13 | Lubrizol Advanced Materials, Inc. | Thermoplastic polyurethane film for delivery of active agents to skin surfaces |
| DE102014225582A1 (de) * | 2014-12-11 | 2015-10-01 | Henkel Ag & Co. Kgaa | Stylinggel mit Spinnen-Seidenprotein und Polyacrylsäure |
| CN106109358A (zh) * | 2016-08-25 | 2016-11-16 | 安迪美生物科技(北京)有限公司 | 一种多肽蚕丝补水修护面膜及其制备方法 |
-
2019
- 2019-06-28 CN CN201980056353.4A patent/CN112638359A/zh active Pending
- 2019-06-28 WO PCT/JP2019/025927 patent/WO2020004649A1/ja unknown
- 2019-06-28 EP EP19827474.8A patent/EP3808329A4/en active Pending
- 2019-06-28 EP EP22155607.9A patent/EP4014958A1/en not_active Withdrawn
- 2019-06-28 JP JP2020527698A patent/JP7460962B2/ja active Active
- 2019-06-28 US US17/256,729 patent/US20210259941A1/en active Pending
- 2019-06-28 TW TW108122947A patent/TW202017558A/zh unknown
- 2019-06-28 KR KR1020217002238A patent/KR20210025070A/ko unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1077210A (ja) | 1996-09-06 | 1998-03-24 | Nakanihon Seni Kogyo Kyodo Kumiai | 化粧料用組成物及びその製造方法並びにこれを配合した化粧品 |
| JP2002308756A (ja) | 2001-03-05 | 2002-10-23 | L'oreal Sa | ポリアミノ酸の保湿剤としての用途とそれを含有する化粧品用又は製薬用組成物 |
| WO2010013555A1 (ja) * | 2008-08-01 | 2010-02-04 | 日産化学工業株式会社 | 新規脂質ジペプチド並びにゲル |
| JP2010047534A (ja) * | 2008-08-22 | 2010-03-04 | Kyushu Univ | 酵素応答性ゲル |
| WO2010106981A1 (ja) * | 2009-03-16 | 2010-09-23 | 日産化学工業株式会社 | 低分子ゲル化剤からなるスプレー用基材 |
| JP2016006030A (ja) | 2014-05-30 | 2016-01-14 | 花王株式会社 | 皮膚化粧料 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022065433A1 (ja) * | 2020-09-28 | 2022-03-31 | 日産化学株式会社 | 毛髪用化粧料 |
| WO2022092284A1 (ja) * | 2020-10-30 | 2022-05-05 | 日産化学株式会社 | 脂質ペプチドとショ糖エステルを含む組成物 |
| EP4230265A1 (en) * | 2020-10-30 | 2023-08-23 | Nissan Chemical Corporation | Composition containing lipid peptide and sucrose ester |
| EP4230265A4 (en) * | 2020-10-30 | 2024-05-08 | Nissan Chemical Corporation | COMPOSITION CONTAINING A LIPID PEPTIDE AND A SUCCAROSE ESTER |
| KR20220106873A (ko) * | 2021-01-22 | 2022-08-01 | 주식회사 코스메카코리아 | 미세먼지 흡착방지용 화장료 조성물의 제조방법 및 그에 의해 제조된 미세먼지 흡착방지용 화장료 조성물 |
| KR102552912B1 (ko) | 2021-01-22 | 2023-07-10 | 주식회사 코스메카코리아 | 미세먼지 흡착방지용 화장료 조성물의 제조방법 및 그에 의해 제조된 미세먼지 흡착방지용 화장료 조성물 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202017558A (zh) | 2020-05-16 |
| CN112638359A (zh) | 2021-04-09 |
| JPWO2020004649A1 (ja) | 2021-07-08 |
| JP7460962B2 (ja) | 2024-04-03 |
| EP3808329A1 (en) | 2021-04-21 |
| KR20210025070A (ko) | 2021-03-08 |
| EP3808329A4 (en) | 2021-08-18 |
| US20210259941A1 (en) | 2021-08-26 |
| EP4014958A1 (en) | 2022-06-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5971487B2 (ja) | 化粧料の製造方法、化粧料用ゲルの調製方法及び化粧料原料に配合される高分子増粘剤の使用量を軽減する方法 | |
| JP6624380B2 (ja) | 脂質ペプチド型化合物を含有するスティック状基材 | |
| EP3747911A1 (en) | Functional polysaccharide particle | |
| WO2011052613A1 (ja) | 化粧料及び皮膚外用剤、並びに医療用機器 | |
| JP6734568B2 (ja) | 脂質ペプチド型化合物を含有するスティック状基材 | |
| JP7460962B2 (ja) | 被膜形成組成物 | |
| JP6802711B2 (ja) | 脂質ペプチド型化合物を含有する保湿基材 | |
| JP4931568B2 (ja) | 表面処理粉体を含有する化粧料 | |
| JPWO2015005219A1 (ja) | 分散液及びヒドロゲル形成方法 | |
| EP3251696A1 (en) | Transdermally absorbable base material comprising lipid peptide-type compound | |
| US20220251393A1 (en) | Pigments containing cellulose | |
| JP5521025B2 (ja) | 浸透促進剤 | |
| WO2021132653A1 (ja) | 汚染防止材料 | |
| JP6694166B2 (ja) | 脂質ペプチド型化合物を含有するスティック状基材の硬度調整方法 | |
| KR20200108447A (ko) | 분체를 포함하는 조성물 | |
| JPWO2018207947A1 (ja) | 多糖含有スティック状基材 | |
| WO2022092284A1 (ja) | 脂質ペプチドとショ糖エステルを含む組成物 | |
| JPWO2018190383A1 (ja) | 顔料又は粉体用分散剤を含有する分散液 | |
| WO2021132668A1 (ja) | 浸透促進材料 | |
| JP2023048158A (ja) | 脂質ペプチドとノニオン性界面活性剤を含む機能性液状組成物 | |
| JP2023091264A (ja) | 付着抑制方法 | |
| JP2022189546A (ja) | 脂質の流動化を高める組成物 | |
| JPWO2018207948A1 (ja) | 高含水スティック状基材 | |
| JP2024037077A (ja) | シクロペンタシロキサン代替組成物及びそれを含む化粧料 | |
| WO2015194054A1 (ja) | フェニルエチルアミン誘導体又はシネフリン含有デスモプラキン発現誘導剤又はデスモプラキン細胞膜局在誘導剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19827474 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020527698 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20217002238 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019827474 Country of ref document: EP Effective date: 20210113 |