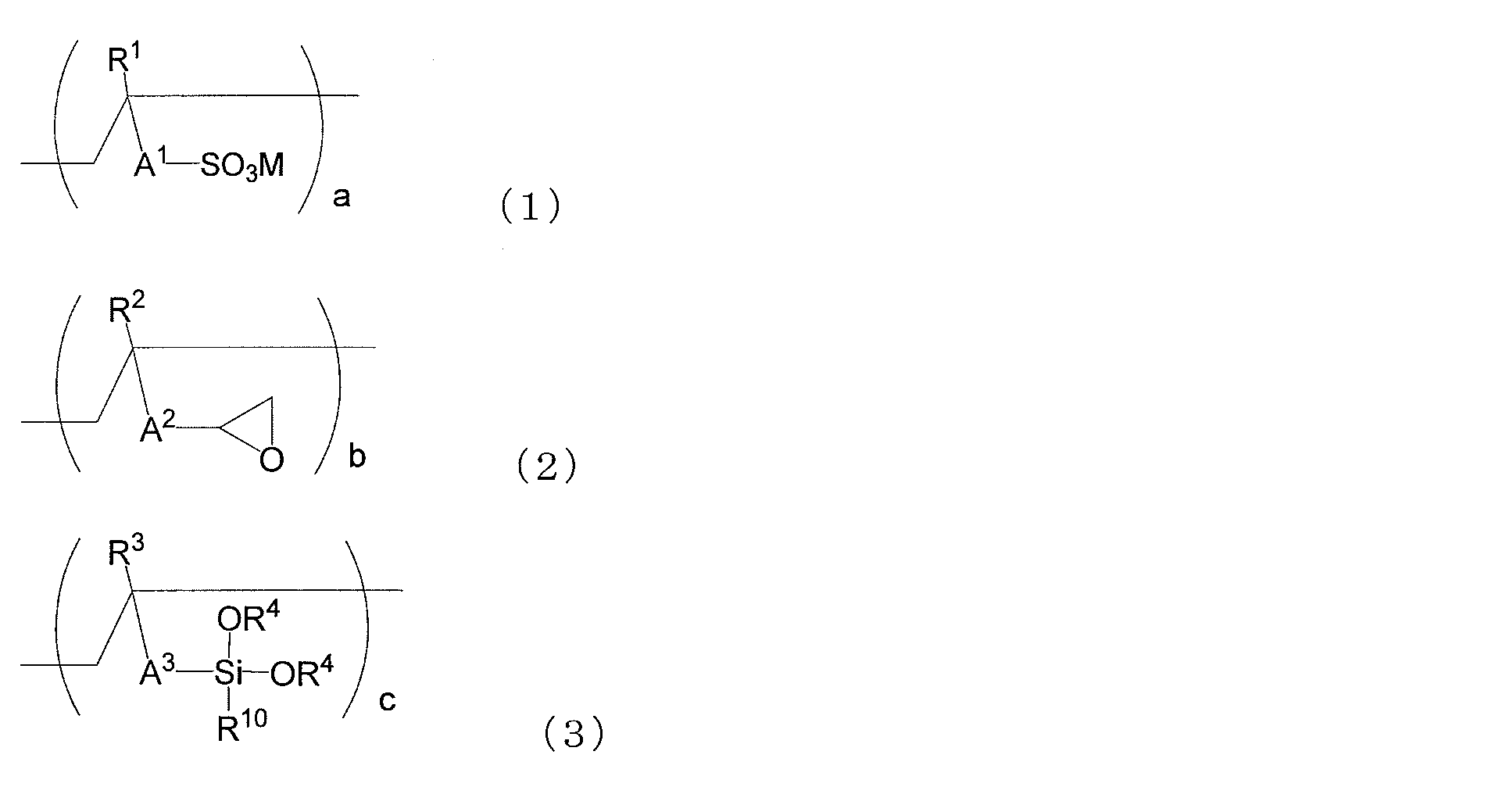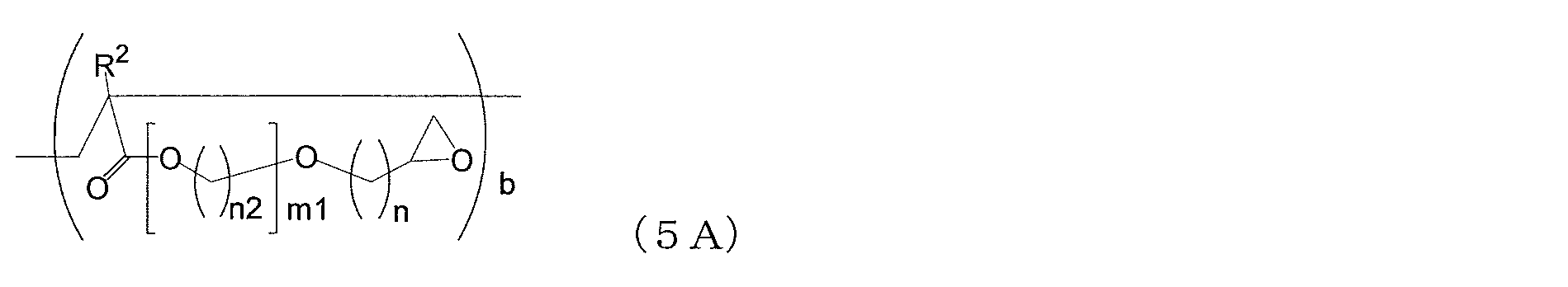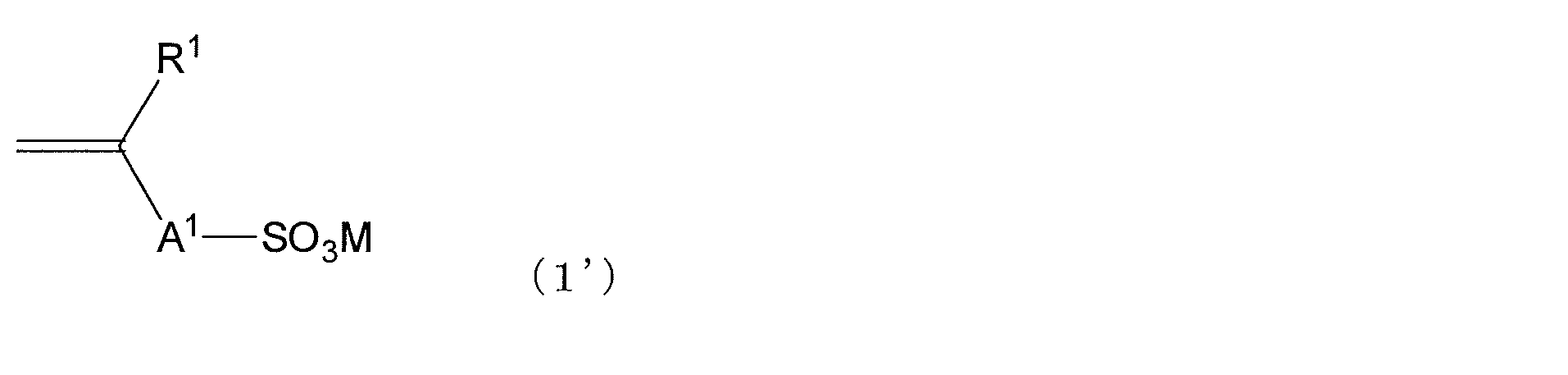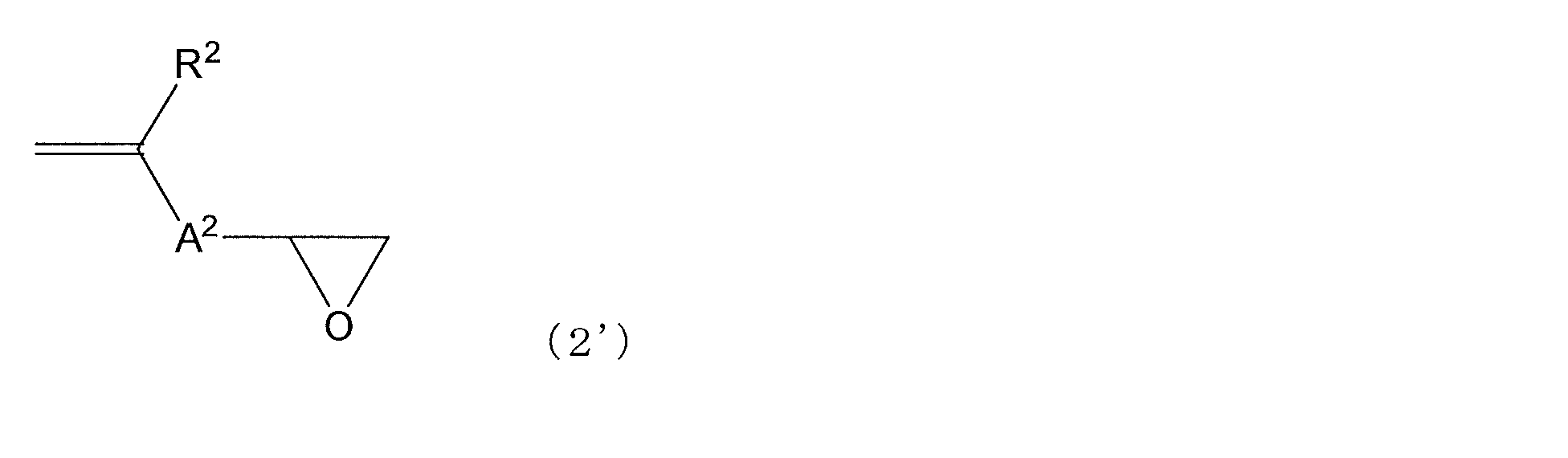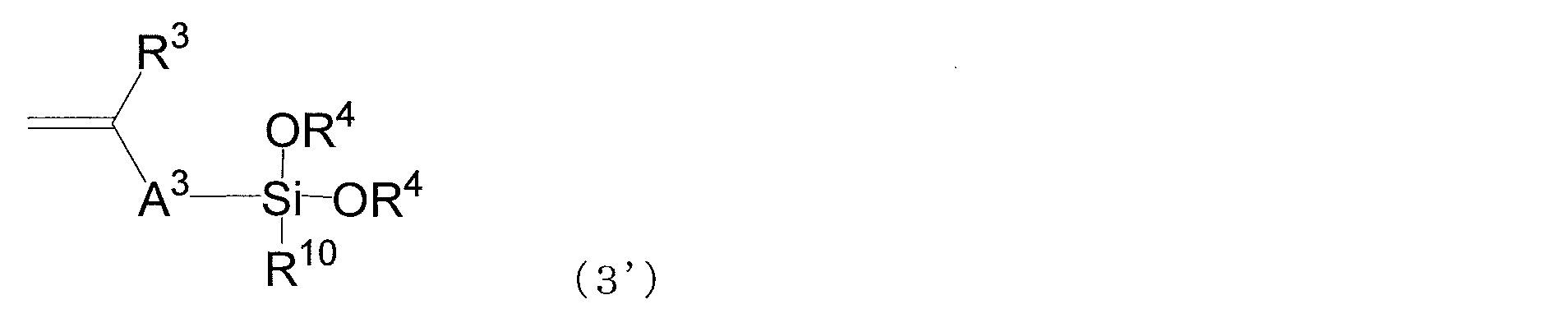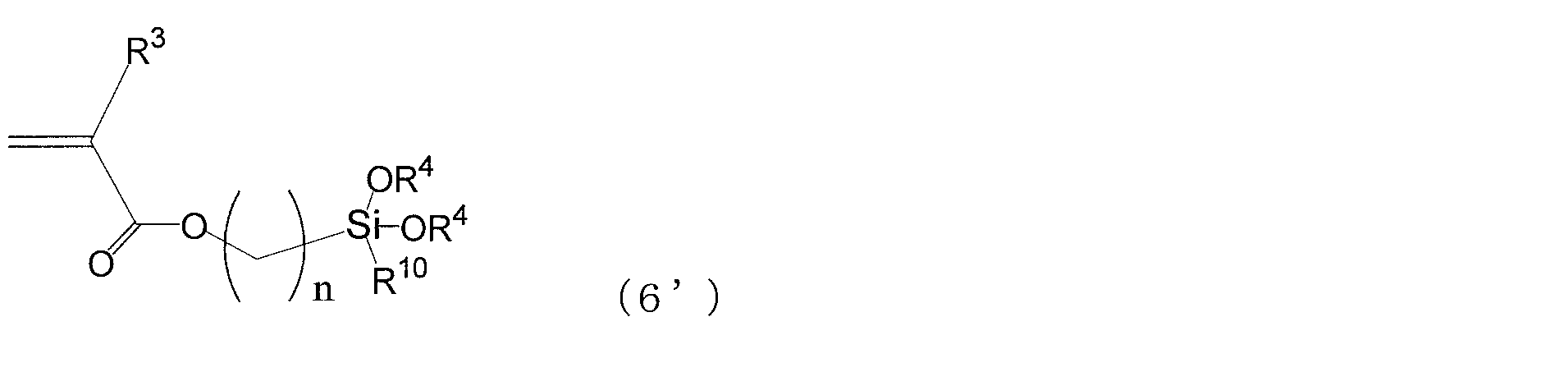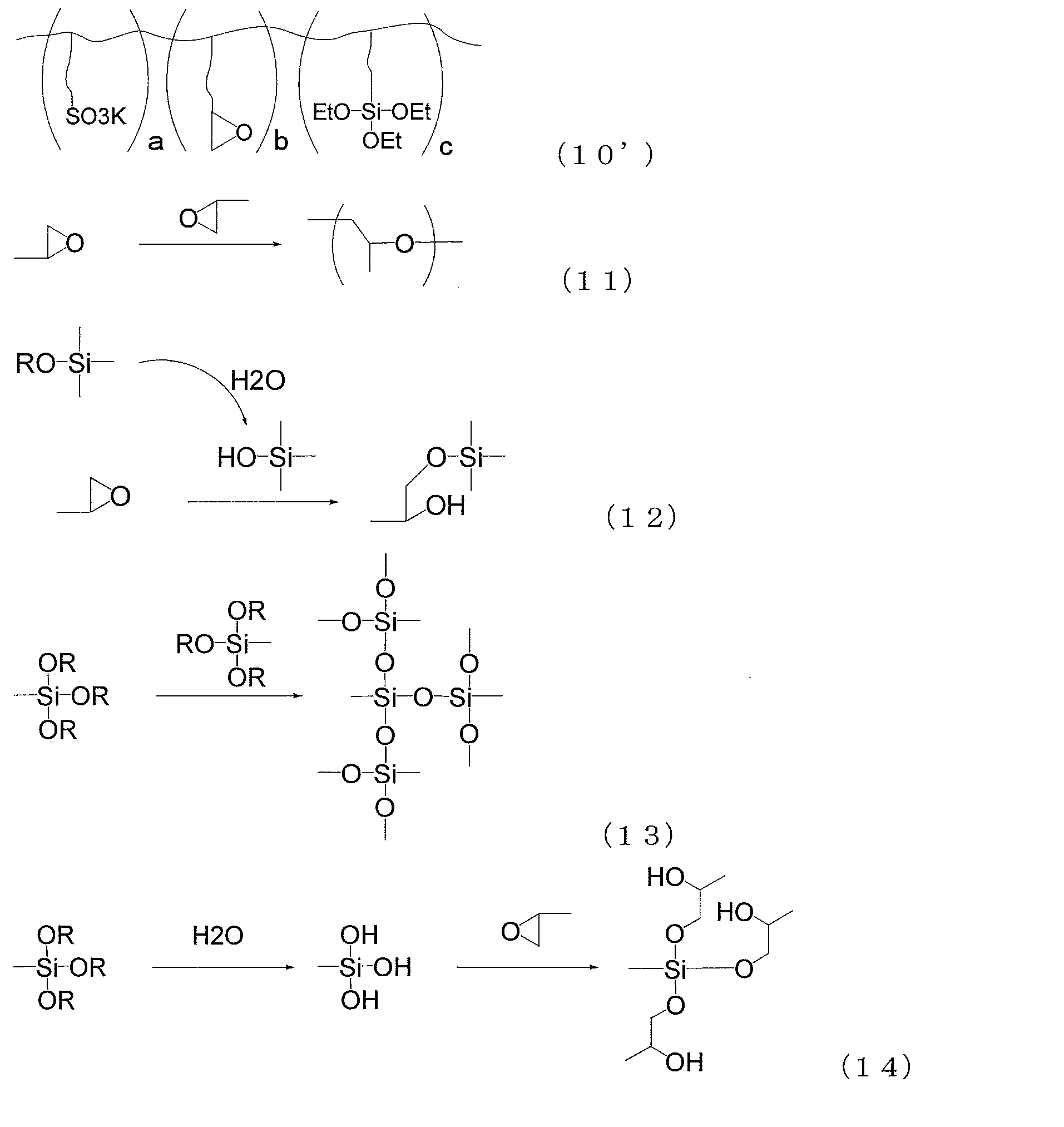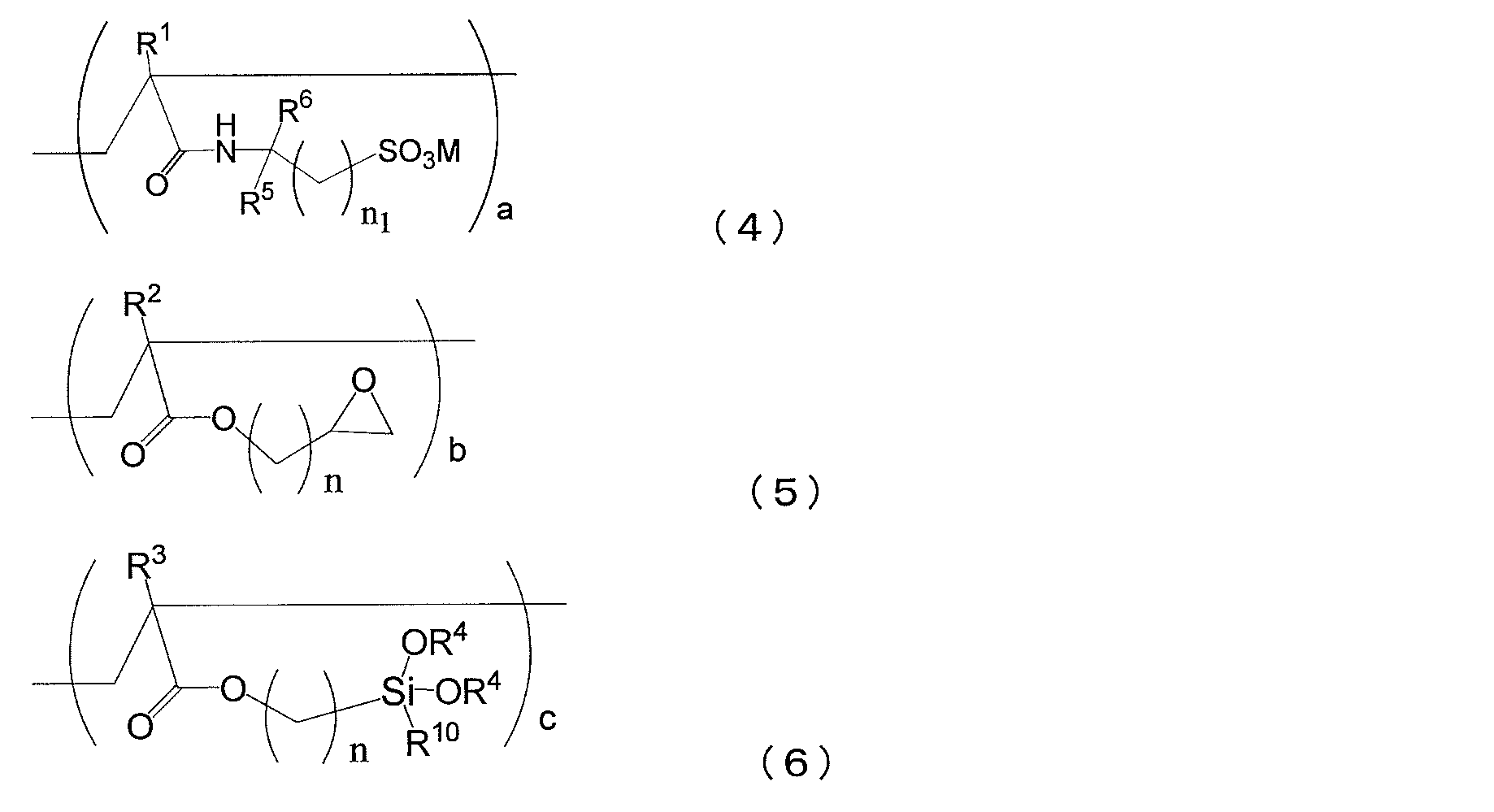WO2016017619A1 - スルホン酸系共重合体とアミノ樹脂からなる親水性材料 - Google Patents
スルホン酸系共重合体とアミノ樹脂からなる親水性材料 Download PDFInfo
- Publication number
- WO2016017619A1 WO2016017619A1 PCT/JP2015/071331 JP2015071331W WO2016017619A1 WO 2016017619 A1 WO2016017619 A1 WO 2016017619A1 JP 2015071331 W JP2015071331 W JP 2015071331W WO 2016017619 A1 WO2016017619 A1 WO 2016017619A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- copolymer
- represented
- reaction
- film
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/02—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by structural features of a fibrous or filamentary layer
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L33/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- C08L33/04—Homopolymers or copolymers of esters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B13/00—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material
- B32B13/04—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material comprising such water setting substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B13/06—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material comprising such water setting substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material of metal
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B13/00—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material
- B32B13/04—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material comprising such water setting substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B13/12—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material comprising such water setting substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B13/00—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material
- B32B13/14—Layered products comprising a a layer of water-setting substance, e.g. concrete, plaster, asbestos cement, or like builders' material next to a fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/043—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of metal
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/082—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising vinyl resins; comprising acrylic resins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/085—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising polyolefins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/09—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising polyesters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/092—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising epoxy resins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/095—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising polyurethanes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/098—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising condensation resins of aldehydes, e.g. with phenols, ureas or melamines
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/18—Layered products comprising a layer of metal comprising iron or steel
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/20—Layered products comprising a layer of metal comprising aluminium or copper
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B23/00—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose
- B32B23/04—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising such cellulosic plastic substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B23/00—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose
- B32B23/04—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising such cellulosic plastic substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B23/042—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising such cellulosic plastic substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material of metal
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B23/00—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose
- B32B23/04—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising such cellulosic plastic substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B23/08—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising such cellulosic plastic substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B23/00—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose
- B32B23/10—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose next to a fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B23/00—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose
- B32B23/20—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising esters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/06—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/06—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B27/08—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/12—Layered products comprising a layer of synthetic resin next to a fibrous or filamentary layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/16—Layered products comprising a layer of synthetic resin specially treated, e.g. irradiated
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
- B32B27/302—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers comprising aromatic vinyl (co)polymers, e.g. styrenic (co)polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
- B32B27/304—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers comprising vinyl halide (co)polymers, e.g. PVC, PVDC, PVF, PVDF
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
- B32B27/306—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers comprising vinyl acetate or vinyl alcohol (co)polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
- B32B27/308—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers comprising acrylic (co)polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/32—Layered products comprising a layer of synthetic resin comprising polyolefins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/34—Layered products comprising a layer of synthetic resin comprising polyamides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
- B32B27/365—Layered products comprising a layer of synthetic resin comprising polyesters comprising polycarbonates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/38—Layered products comprising a layer of synthetic resin comprising epoxy resins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/40—Layered products comprising a layer of synthetic resin comprising polyurethanes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/42—Layered products comprising a layer of synthetic resin comprising condensation resins of aldehydes, e.g. with phenols, ureas or melamines
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/22—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by the presence of two or more layers which are next to each other and are fibrous, filamentary, formed of particles or foamed
- B32B5/24—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by the presence of two or more layers which are next to each other and are fibrous, filamentary, formed of particles or foamed one layer being a fibrous or filamentary layer
- B32B5/26—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by the presence of two or more layers which are next to each other and are fibrous, filamentary, formed of particles or foamed one layer being a fibrous or filamentary layer another layer next to it also being fibrous or filamentary
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B7/00—Layered products characterised by the relation between layers; Layered products characterised by the relative orientation of features between layers, or by the relative values of a measurable parameter between layers, i.e. products comprising layers having different physical, chemical or physicochemical properties; Layered products characterised by the interconnection of layers
- B32B7/04—Interconnection of layers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B9/00—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00
- B32B9/002—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising natural stone or artificial stone
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B9/00—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00
- B32B9/005—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising one layer of ceramic material, e.g. porcelain, ceramic tile
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B9/00—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00
- B32B9/04—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising such particular substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B9/00—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00
- B32B9/04—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising such particular substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B9/041—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising such particular substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material of metal
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B9/00—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00
- B32B9/04—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising such particular substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B9/045—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising such particular substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B9/00—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00
- B32B9/04—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising such particular substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B9/047—Layered products comprising a layer of a particular substance not covered by groups B32B11/00 - B32B29/00 comprising such particular substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material made of fibres or filaments
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/32—Esters containing oxygen in addition to the carboxy oxygen containing epoxy radicals
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
- C08F220/56—Acrylamide; Methacrylamide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F230/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal
- C08F230/04—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal
- C08F230/08—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal containing silicon
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/20—Manufacture of shaped structures of ion-exchange resins
- C08J5/22—Films, membranes or diaphragms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/01—Use of inorganic substances as compounding ingredients characterized by their specific function
- C08K3/013—Fillers, pigments or reinforcing additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L33/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- C08L33/24—Homopolymers or copolymers of amides or imides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L43/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing boron, silicon, phosphorus, selenium, tellurium or a metal; Compositions of derivatives of such polymers
- C08L43/04—Homopolymers or copolymers of monomers containing silicon
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L57/00—Compositions of unspecified polymers obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C08L57/06—Homopolymers or copolymers containing elements other than carbon and hydrogen
- C08L57/10—Homopolymers or copolymers containing elements other than carbon and hydrogen containing oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/02—Polyamines
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/24—Homopolymers or copolymers of amides or imides
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D143/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing boron, silicon, phosphorus, selenium, tellurium, or a metal; Coating compositions based on derivatives of such polymers
- C09D143/04—Homopolymers or copolymers of monomers containing silicon
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D161/00—Coating compositions based on condensation polymers of aldehydes or ketones; Coating compositions based on derivatives of such polymers
- C09D161/20—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
- C09D161/26—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with heterocyclic compounds
- C09D161/28—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with heterocyclic compounds with melamine
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/16—Antifouling paints; Underwater paints
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/16—Antifouling paints; Underwater paints
- C09D5/1606—Antifouling paints; Underwater paints characterised by the anti-fouling agent
- C09D5/1637—Macromolecular compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/24—Electrically-conducting paints
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/20—All layers being fibrous or filamentary
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/24—All layers being polymeric
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/06—Coating on the layer surface on metal layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/10—Coating on the layer surface on synthetic resin layer or on natural or synthetic rubber layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/20—Inorganic coating
- B32B2255/205—Metallic coating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/26—Polymeric coating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/28—Multiple coating on one surface
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/10—Inorganic fibres
- B32B2262/101—Glass fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/20—Properties of the layers or laminate having particular electrical or magnetic properties, e.g. piezoelectric
- B32B2307/21—Anti-static
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/40—Properties of the layers or laminate having particular optical properties
- B32B2307/412—Transparent
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/50—Properties of the layers or laminate having particular mechanical properties
- B32B2307/536—Hardness
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/50—Properties of the layers or laminate having particular mechanical properties
- B32B2307/554—Wear resistance
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/50—Properties of the layers or laminate having particular mechanical properties
- B32B2307/584—Scratch resistance
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/712—Weather resistant
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/728—Hydrophilic
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2419/00—Buildings or parts thereof
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2437/00—Clothing
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2439/00—Containers; Receptacles
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2457/00—Electrical equipment
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2457/00—Electrical equipment
- B32B2457/20—Displays, e.g. liquid crystal displays, plasma displays
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2457/00—Electrical equipment
- B32B2457/20—Displays, e.g. liquid crystal displays, plasma displays
- B32B2457/208—Touch screens
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2479/00—Furniture
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2535/00—Medical equipment, e.g. bandage, prostheses or catheter
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2551/00—Optical elements
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2551/00—Optical elements
- B32B2551/08—Mirrors
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2590/00—Signboards, advertising panels, road signs
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2605/00—Vehicles
- B32B2605/08—Cars
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2605/00—Vehicles
- B32B2605/12—Ships
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2605/00—Vehicles
- B32B2605/18—Aircraft
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/32—Esters containing oxygen in addition to the carboxy oxygen containing epoxy radicals
- C08F220/325—Esters containing oxygen in addition to the carboxy oxygen containing epoxy radicals containing glycidyl radical, e.g. glycidyl (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
- C08F220/58—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing oxygen in addition to the carbonamido oxygen, e.g. N-methylolacrylamide, N-(meth)acryloylmorpholine
- C08F220/585—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing oxygen in addition to the carbonamido oxygen, e.g. N-methylolacrylamide, N-(meth)acryloylmorpholine and containing other heteroatoms, e.g. 2-acrylamido-2-methylpropane sulfonic acid [AMPS]
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F230/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal
- C08F230/04—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal
- C08F230/08—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal containing silicon
- C08F230/085—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal containing silicon the monomer being a polymerisable silane, e.g. (meth)acryloyloxy trialkoxy silanes or vinyl trialkoxysilanes
Definitions
- the present invention provides a hydrophilic cured product (for example, a film made of the cured product) having antifogging properties, antifouling properties, and antistatic properties, and excellent wear resistance and weather resistance, and uses of the cured product About.
- Non-patent Document 1 As a method of solving the problem of fogging, an antifogging paint in which a reactive surfactant is added to an acrylic oligomer has been proposed, and a cured product obtained from the antifogging paint (for example, a film made of the cured product) It is said that hydrophilicity and water absorption are improved (Non-patent Document 1). Further, as a method for solving the problem of dirt, for example, the surface is improved in hydrophilicity, and the dirt (hydrophobic substance etc.) adhering to the outer wall etc. is lifted up by rain or watering etc. and efficiently removed. An antifouling material having a cleaning property (antifouling property) has attracted attention (Non-Patent Documents 2 and 3).
- the present inventors have proposed a cured product (for example, a monolayer film) in which an anionic hydrophilic group is inclined (concentrated) on the surface as a proposal for solving the problems of “cloudiness” and “dirt” ( Patent Document 1).
- the cured product (for example, a film) obtained by the present invention is transparent and highly hydrophilic, and has an antifogging property, an antifouling property, an antistatic property, a fast drying property (fast removal of attached water), and chemical resistance. It is excellent and hard and has excellent scratch resistance.
- Non-patent Document 4 a method of coating an inorganic compound on the substrate surface.
- a representative example is a method in which alkoxysilane is applied to a hard coat of a spectacle lens by a sol-gel reaction (Non-patent Document 4).
- Alkoxysilane hard coat is very hard due to its dense structure, and its wear is comparable to glass, but on the other hand, the hard coat is easy to break, difficult to dye, cloudy, easy to get dirty. There are problems such as being easily fixed.
- Patent Document 2 a method of blending hydroxysilane with a melamine resin and a silicon compound having an epoxy group
- Patent Document 3 a method of blending hydroxysilane with an epoxy compound and an aluminum complex
- Patent Document 4 a method of blending an acrylic polymer having a hydroxyl group with hydroxysilane
- Patent Document 5 As a method for imparting antifogging properties, a method of blending a styrenesulfonic acid polymer with alkoxysilane (Patent Document 5) has been proposed.
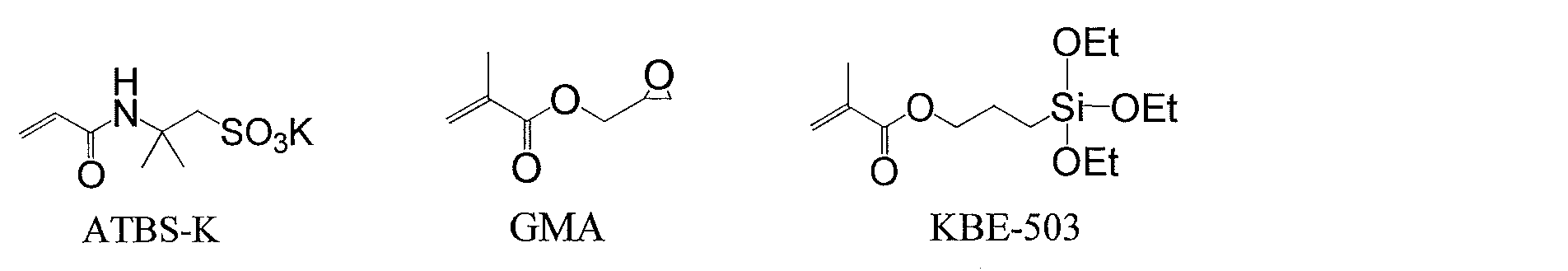
- a polymerizable unsaturated monomer having an epoxy group a polymerizable unsaturated monomer having an acid group such as a sulfonic acid group, a polymerizable unsaturated monomer having a hydroxyl group
- a copolymer compound (A) obtained by emulsion polymerization of a polymerizable unsaturated monomer having a hydrolyzable silyl group with a use amount of 0.1 to 10 wt% based on the total amount of monomers is added to a zirconium compound (B ) And a silane coupling agent (C) are known (Patent Document 6).
- a polymerizable unsaturated monomer containing no epoxy group, acid group or hydroxyl group, a polymerizable unsaturated monomer having an epoxy group, and an acid group such as a sulfonic acid group A polymerizable unsaturated monomer, a polymerizable unsaturated monomer having a hydroxyl group, a polymerizable unsaturated monomer having a hydrolyzable silyl group, and a polymerizable unsaturated monomer having a cyclic ureido group having a specific structure are added to the total amount of monomers.
- a treatment agent comprising a core-shell type resin (A) emulsion-polymerized within a range of 0.1 to 5 wt%, respectively, and a zirconium compound (B) and a silane coupling agent (C) (Patent Document 7) )It has been known.
- Patent Document 8 a method of obtaining a film made of a highly hydrophilic cured product by reacting a copolymer having a sulfonic acid group and an alkoxysilyl group with an alkoxysilane (Patent Document 8) is also known.
- Patent Document 9 a method for obtaining a film made of a highly hydrophilic cured product by reacting a copolymer having a sulfonic acid group and an epoxy group with an alkoxysilane.
- Patent Documents 8 and 9 are also preferable proposals because the hydrophilicity tends to be high, but when stored or heated at room temperature for a long period of time, pollutants in the atmosphere are likely to adhere, and the attached contamination It has been found by the present inventors that the substance has a problem that it is difficult to desorb.
- the object of the present invention is excellent in the balance between hydrophilicity and wear resistance, maintains high hydrophilicity even after washing with water, and is resistant to contaminants (or easily desorbed) even when stored or heated for a long time. It is in providing the hardened
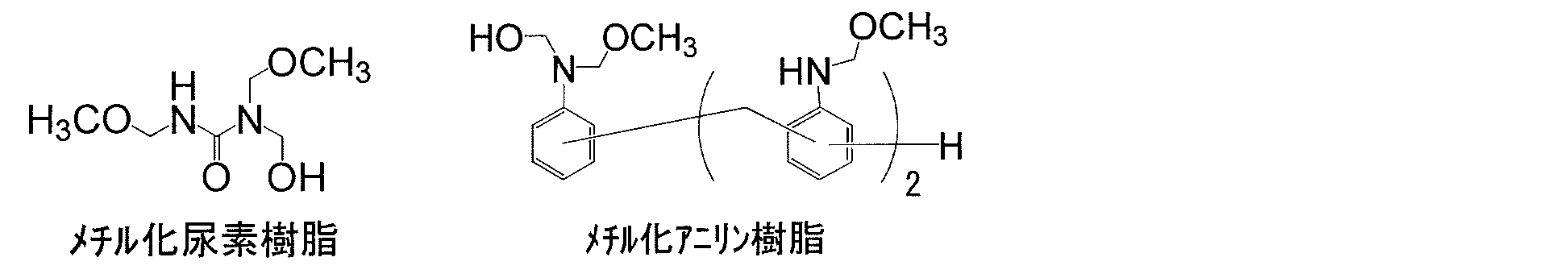
- alkoxysilane (hydroxyl-containing silane) used as a copolymerization component is the main cause of pollutants in the air that are easily attached and difficult to desorb, and overcome this problem.
- a cured product obtained by curing the composition for example, a film made of the cured product
- the present invention relates to the following [1] to [9].
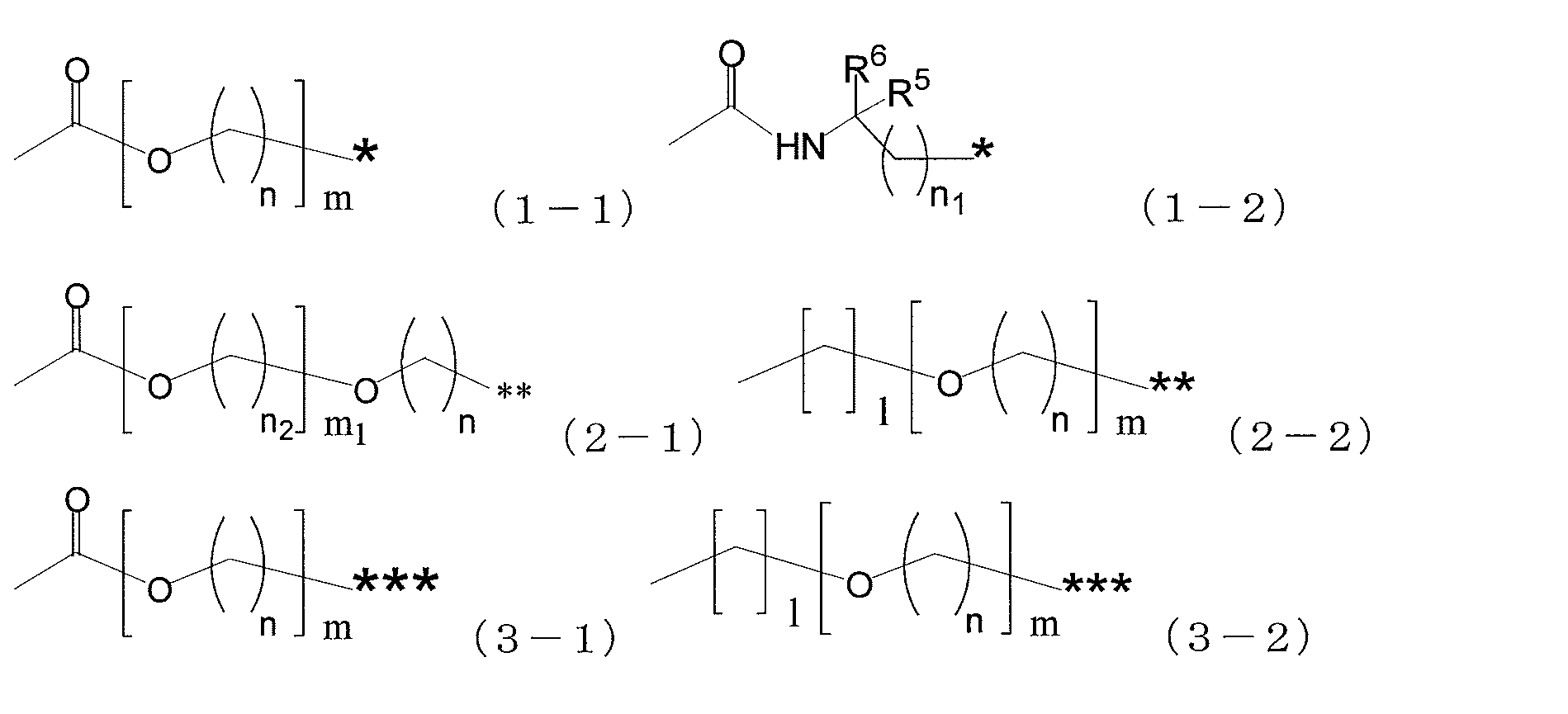
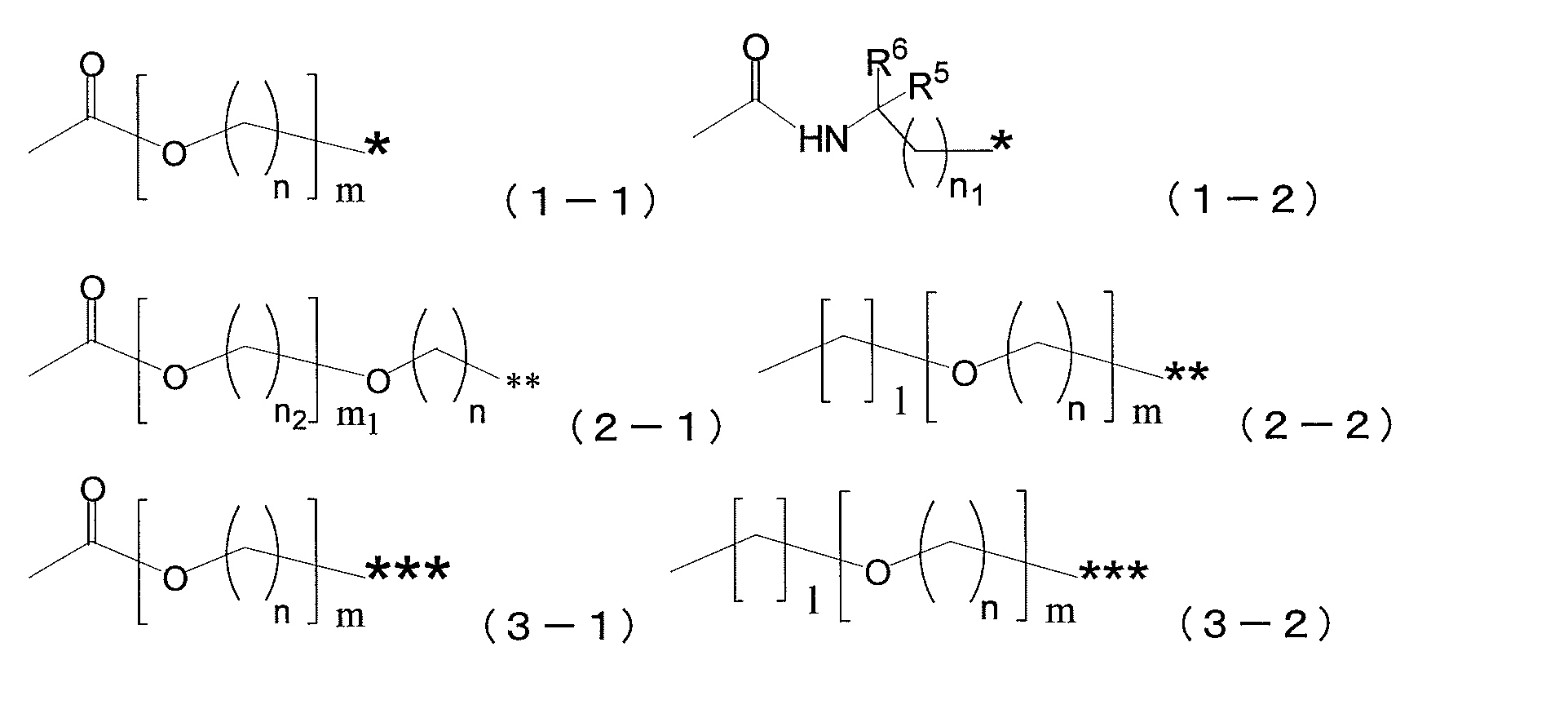
- a 1 represents a single bond, a divalent hydrocarbon group having 1 to 10 carbon atoms, a group represented by the following formula (1-1), or a group represented by the following formula (1-2).
- a 2 represents a single bond, a divalent hydrocarbon group having 1 to 10 carbon atoms, a group represented by the following formula (2-1), or a group represented by the following formula (2-2).
- a 3 represents a single bond, a divalent hydrocarbon group having 1 to 10 carbon atoms, a group represented by the following formula (3-1), or a group represented by the following formula (3-2).
- R 1 , R 2 , and R 3 each independently represent a hydrogen atom or a methyl group
- R 4 represents a hydrogen atom, a methyl group, an ethyl group, a propyl group, or a butyl group, and R 4 may be the same as or different from each other
- R 10 represents a hydrogen atom, a methyl group, an ethyl group, a propyl group, a butyl group, a methoxy group, an ethoxy group, a propoxy group, or a butoxy group
- M represents a hydrogen atom, an alkali metal ion, a half-valent alkaline earth metal ion, an ammonium ion, or an amine ion; In the following formulas (1-1), (1-2), (2-1
- the copolymer (i) is obtained from the above composition which is a copolymer (i3-1) containing structural units represented by the following general formulas (4), (5) and (6) [ 1].
- n is an integer of 1 to 10
- n1 is an integer of 0 to 10
- R 1 , R 2 , R 3 , R 5 , and R 6 each independently represent a hydrogen atom or a methyl group
- R 4 represents a hydrogen atom, a methyl group, an ethyl group, a propyl group, or a butyl group, and R 4 may be the same as or different from each other
- R 10 represents a hydrogen atom, a methyl group, an ethyl group, a propyl group, a butyl group, a methoxy group, an ethoxy group, a propoxy group, or a butoxy group
- M represents a hydrogen atom, an alkali metal ion, a half-valent alkaline earth metal ion, an ammonium ion, or an amine ion.
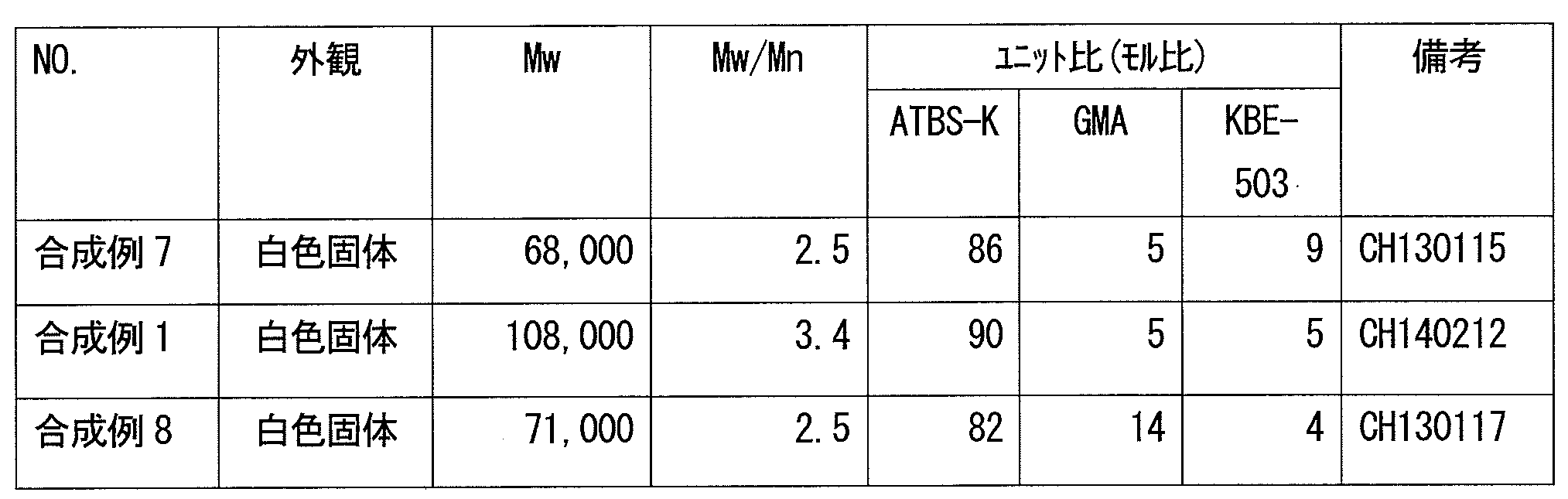
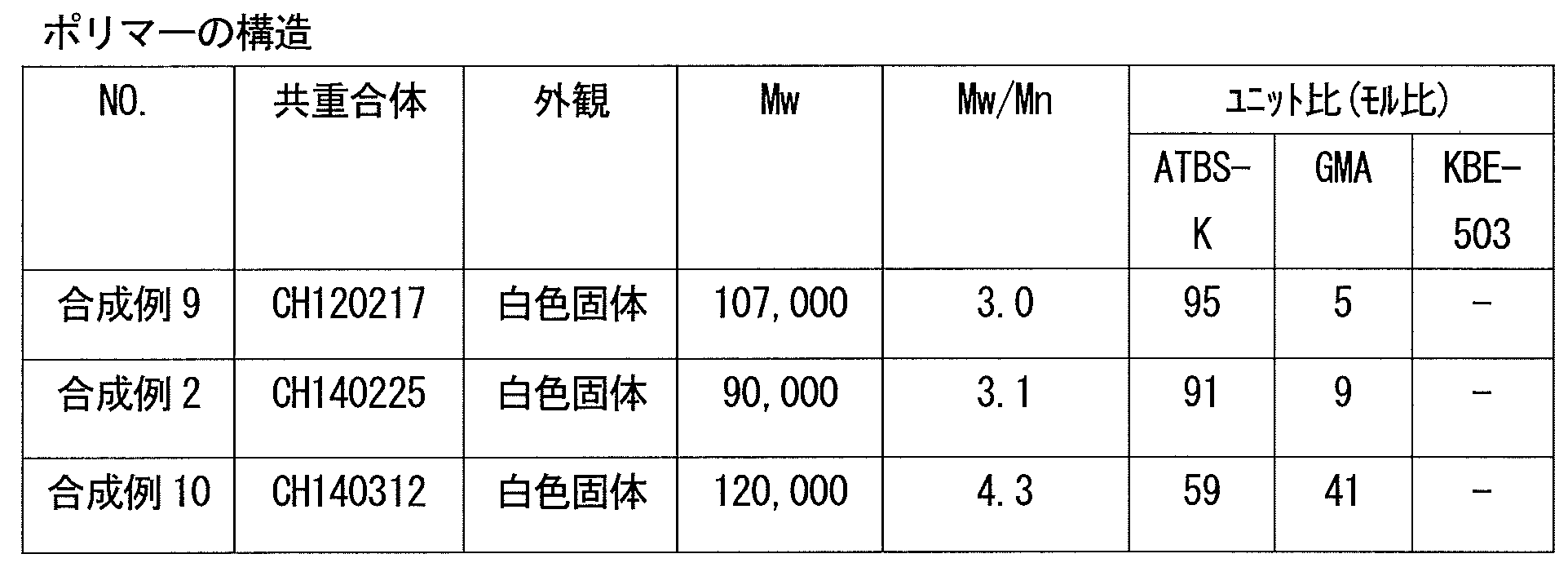
- the copolymer (i) is obtained from the above composition having a weight average molecular weight in terms of standard polymethyl methacrylate measured by gel permeation chromatography (GPC) of 500 to 3,000,000.
- the cured product according to [1] or [2] obtained from a composition having a weight average molecular weight of 500 to 3,000,000 as measured by GPC of the copolymer (i).
- R 30 represents a hydrogen atom, an alkyl group having 1 to 10 carbon atoms, a hydroxymethyl group, or an alkoxymethyl group having 1 to 10 carbon atoms
- R 40 represents a hydroxyl group, a hydrogen atom
- q190 is an integer of 1 to 90
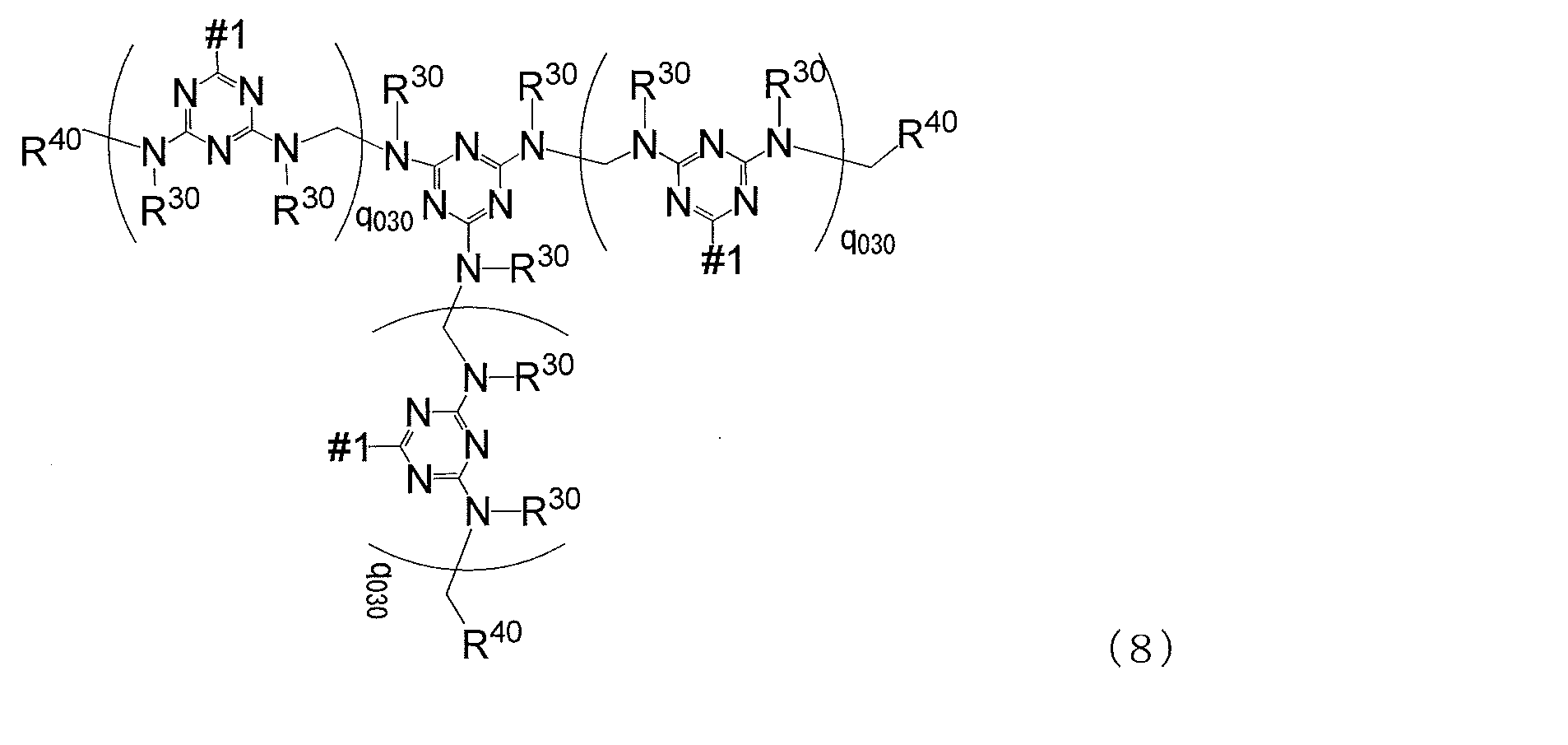
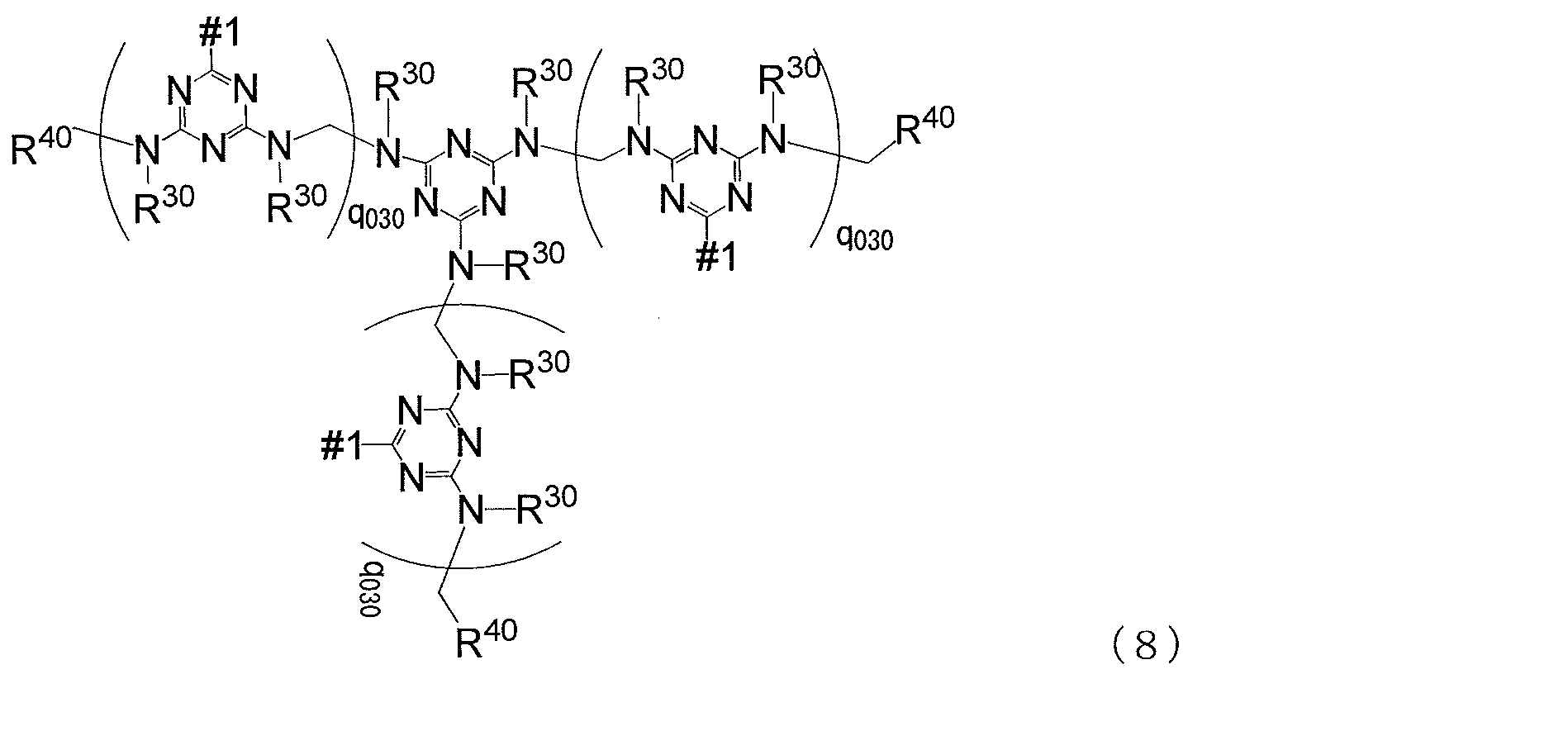
- MC is represented by any one of the following general formulas (8) to (10) Represents a mother nucleus
- # 2 is a bond that bonds to # 1 in the following general formulas (8) to (10), and the numbers of # 1 and # 2 are the same
- q 030 is an integer of 0 to 30, q 030 may be the same as or different from each other, R 30 and R 40 are the same as defined in formula (7)
- q 050 is an integer of 0 to 50
- X represents an oxygen
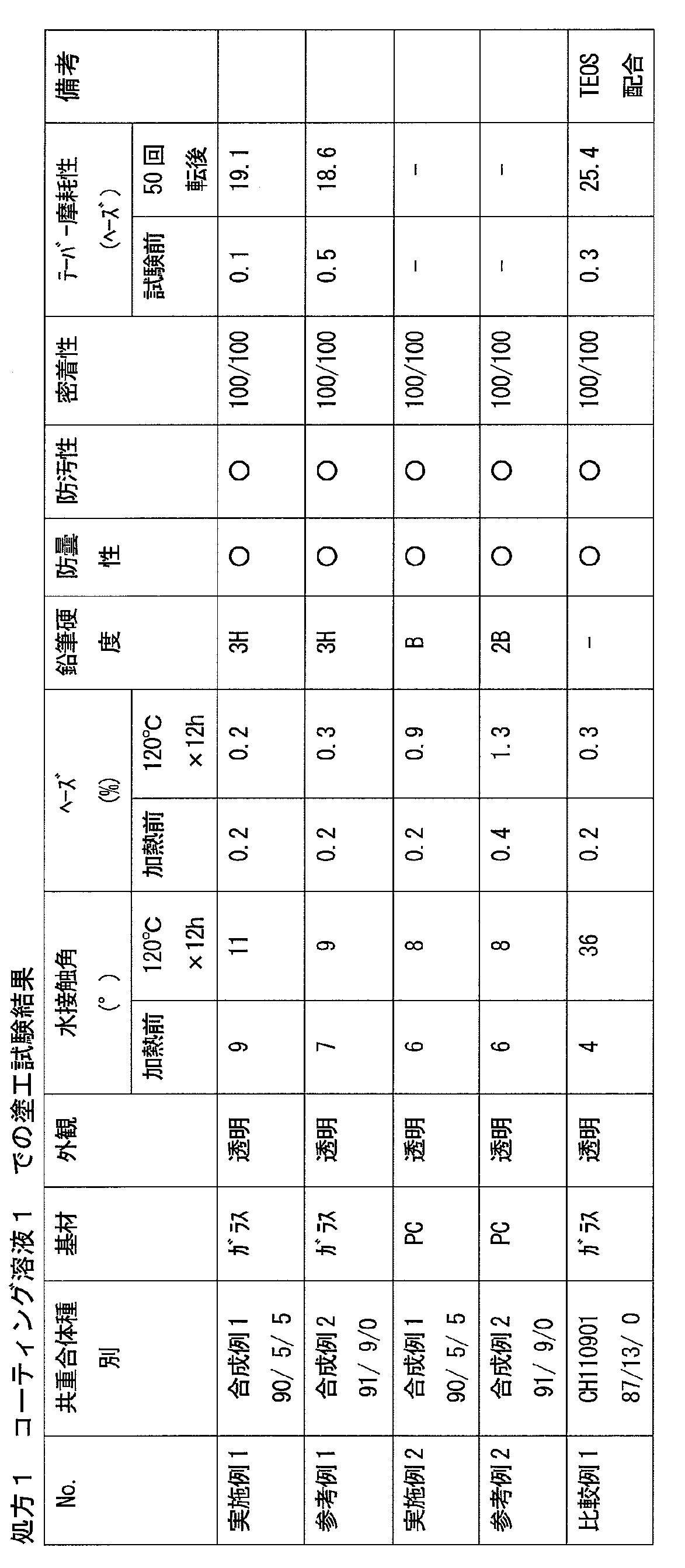
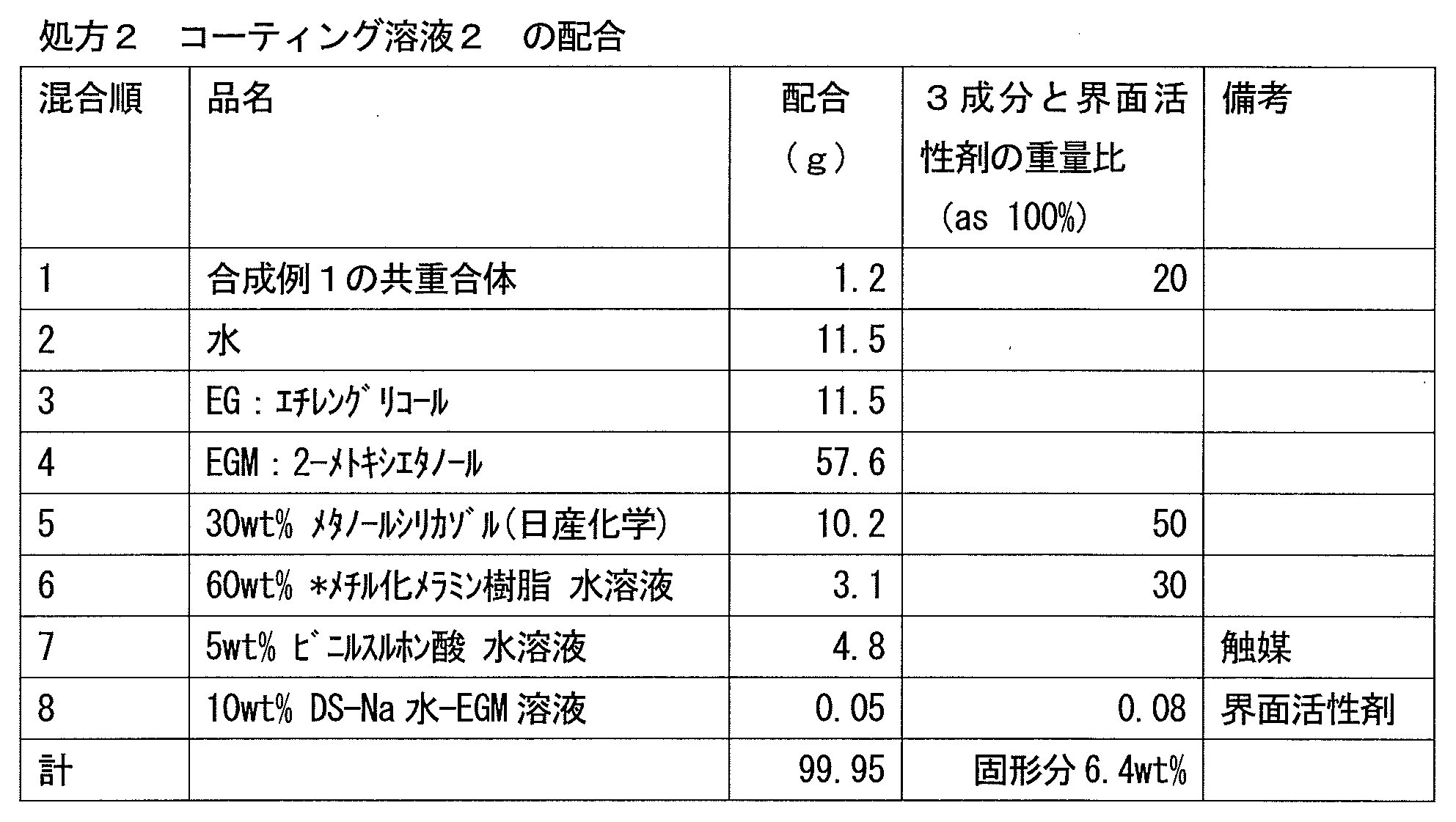
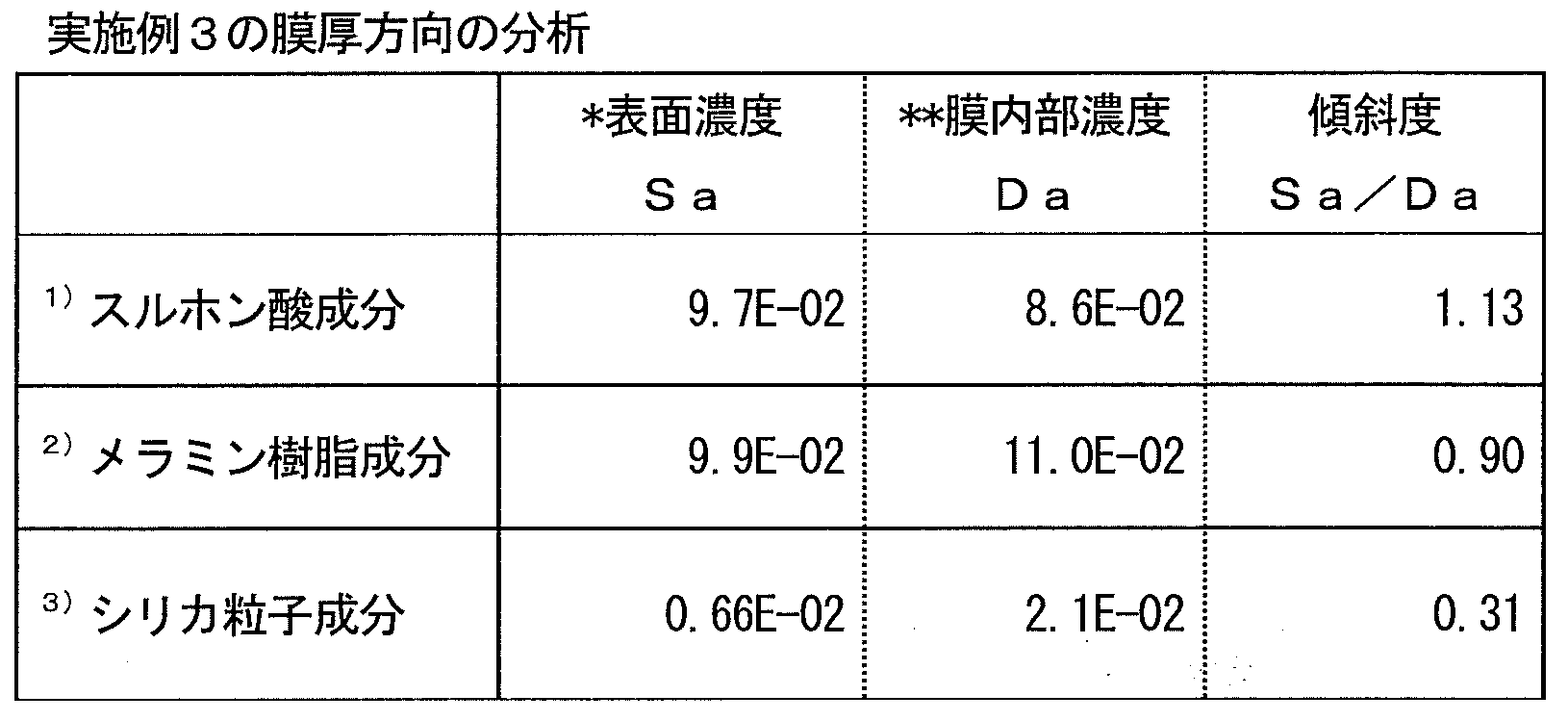
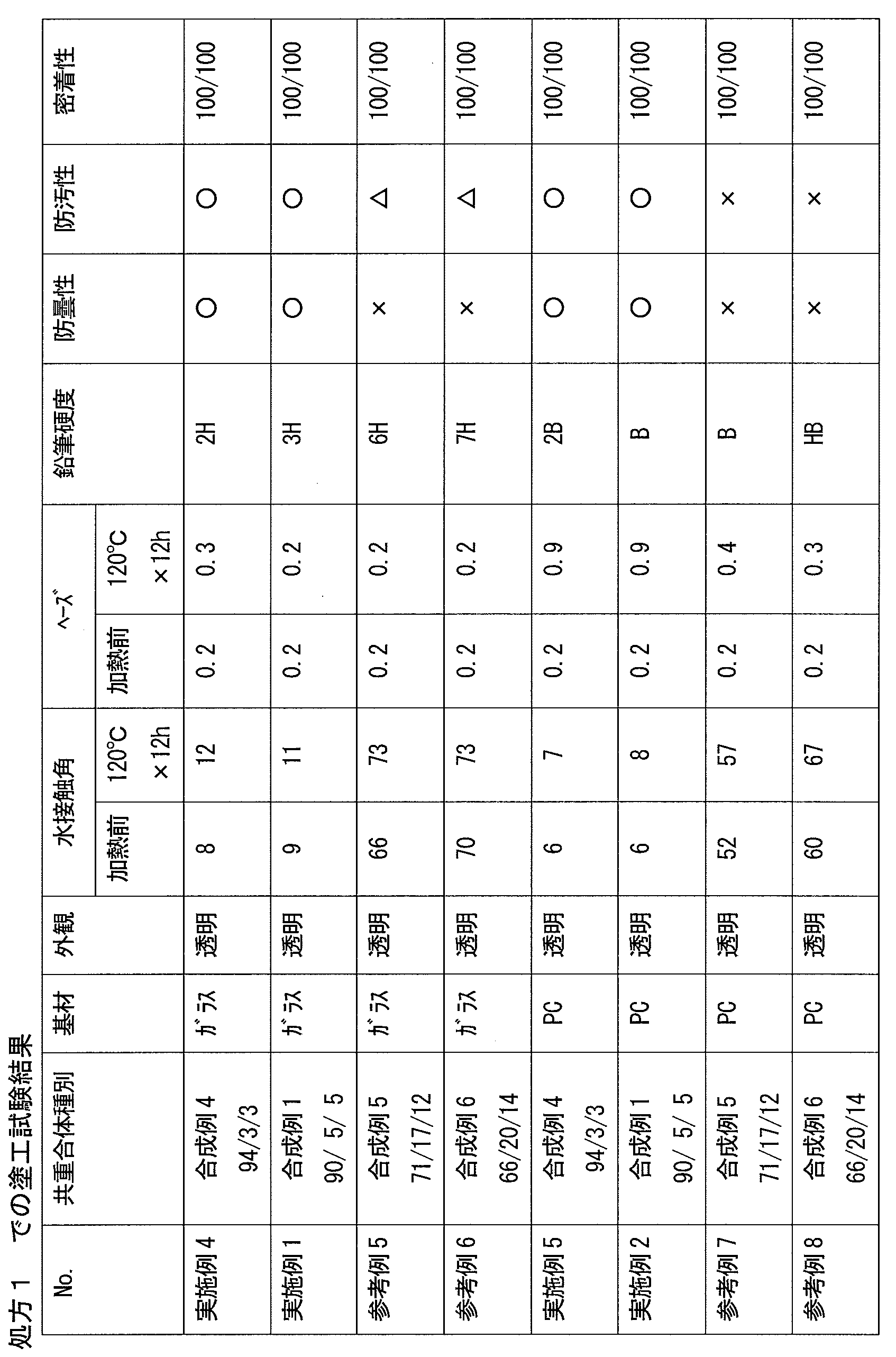
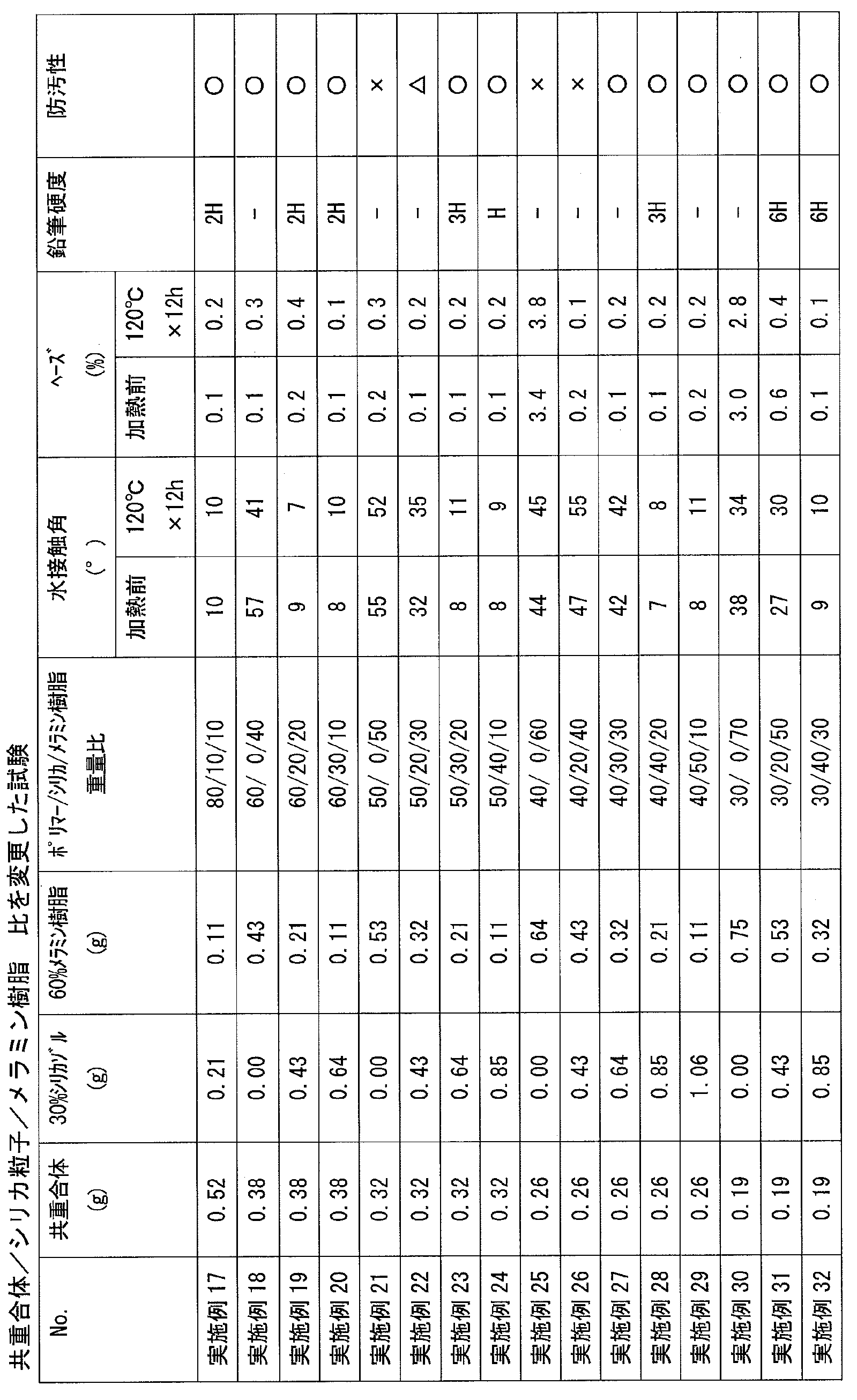
- the cured product obtained by the present invention and the film made of the cured product have an excellent balance of hydrophilicity, hardness, and abrasion resistance, and can suppress a decrease in hydrophilicity due to water, contaminants, and the like.
- membrane obtained by this invention can be used also as a laminated body by laminating
- the composition used for forming the cured product of the present invention includes a copolymer (i).
- the copolymer (i) is characterized by including structural units represented by (1), (2) and (3).
- R 1 , R 2 and R 3 each independently represent a hydrogen atom or a methyl group
- R 4 represents a hydrogen atom, a methyl group or an ethyl group.
- a propyl group, or a butyl group, R 4 may be the same or different from each other
- R 10 represents a hydrogen atom, a methyl group, an ethyl group, a propyl group, a butyl group, a methoxy group, an ethoxy group, a propoxy group, Or represents a butoxy group.
- M represents a hydrogen atom, an alkali metal ion, a half-valent alkaline earth metal ion, an ammonium ion or an amine ion.
- a 1 is a single bond, a divalent hydrocarbon group having 1 to 10 carbon atoms, a group represented by the following formula (1-1), or A group represented by the following formula (1-2), wherein A 2 is a single bond, a divalent hydrocarbon group having 1 to 10 carbon atoms, a group represented by the following formula (2-1), or Represents a group represented by the following formula (2-2), and A 3 represents a single bond, a divalent hydrocarbon group having 1 to 10 carbon atoms, a group represented by the following formula (3-1), or A group represented by the following formula (3-2) is shown.
- n is an integer of 1 to 10
- m is an integer of 1 to 6. It is.
- n1 is an integer of 0 to 10.
- n2 is an integer of 1 to 10
- m1 is an integer of 0 to 6.
- l is an integer of 0-4.
- R 5 and R 6 each independently represent a hydrogen atom or a methyl group.
- the copolymer (i) exhibits hydrophilicity and cross-linking reactivity by including the above-mentioned structural unit, and the composition containing the copolymer (i) has an excellent balance between hydrophilicity and wear resistance, and is hydrophilic by water. Therefore, it is possible to produce a cured product having a small decrease in property and excellent weather resistance, for example, a film made of the cured product.
- a 1 in the above formula (1) is preferably a single bond, methylene, phenylene, a group represented by the above formula (1-1), or a group represented by the above formula (1-2).
- the group represented by 1-2) is more preferable.
- R 1 and M in the above formula (4) are the same as those in the above formula (1), and the definitions of R 5 , R 6 and n1 are those in the above formula (1-2). Is the same.
- M in the above formulas (1) and (4) is a hydrogen atom, an alkali metal ion, a half-valent alkaline earth metal ion, an ammonium ion, or an amine ion, but in the resulting copolymer (i)
- SO 3 M is not in the form of a free acid, and among these M, alkali metal ions, half-valent alkaline earth metal ions, ammonium ions, and amine ions are preferable.
- alkali metal ion sodium ion, potassium ion, and rubidium ion are preferable.
- alkaline earth metal ions calcium ions and magnesium ions are preferable.
- ammonium ion tetrahydroammonium ion (NH 4 + ) is preferable.
- the amine ion include trihydro-methylamine ion, trihydro-ethylamine ion, trihydro-propylamine ion, trihydro-isopropylamine ion, trihydro-butylamine ion, trihydro-cyclohexylamine ion, trihydro-benzylamine ion, dihydro-dimethylamine.
- Ions, hydro-triethylamine ions, trihydro-ethanolamine ions, dihydro-diethanolamine ions, hydro-triethanolamine ions are preferred.
- a 3 in the above formula (3) is preferably a single bond, methylene, phenylene, or a group represented by the above formula (3-1), and preferably a group represented by the above formula (3-1). More preferred.
- the ratio a of the structural unit represented by the formula (1) having a sulfonic acid-containing group may be increased.
- the ratio of the structural unit represented by the formula (2) having a group contributing to the crosslinking reaction and the structural unit of (3) is relatively Toughness, wear resistance, chemical resistance, etc., because the crosslinking density of a cured product (for example, a film made of the cured product) formed from the composition containing the copolymer (i) is lowered. Etc. tend to decrease and may not be desirable.
- the copolymer (i) in the case of a copolymer containing a large amount of the structural unit represented by (3), the hardness, wear resistance, etc. tend to improve, but the hydrophilicity tends to decrease. It is in. Therefore, in applications where high hydrophilicity is required, the copolymer (i3) tends to be preferable as the copolymer (i).
- a monomer mixture containing a polymerizable functional group having a carbon-carbon double bond corresponding to and a compound having an alkoxysilyl group may be polymerized.
- the ratios a, b, and c of the structural units (1), (2), and (3) that can be included in the copolymer (i) are monomers for polymerizing the copolymer (i).
- the monomer corresponding to the structural unit represented by Formula (1), the monomer corresponding to the structural unit represented by Formula (2), and the structure represented by Formula (3) contained in the mixture It can be controlled by the charging ratio of the monomer corresponding to the unit.
- Examples of the compound having a polymerizable functional group having a carbon-carbon double bond corresponding to the structural unit represented by the formula (1) and an SO 3 M group include compounds represented by the following general formula (1 ′). It is done.
- a sulfonic acid compound having a vinyl group a sulfonic acid compound having an allyl group, a sulfonic acid compound having an isopropenyl group, and a sulfonic acid compound having a styryl group
- Acryloyloxy group or methacryloyloxy group (hereinafter, acryloyloxy and methacryloyloxy may be collectively referred to as (meth) acryloyloxy, and acryl and methacryl may be collectively referred to as (meth) acrylic)
- a sulfonic acid compound having an acrylamide group or a methacrylamide group hereinafter, acrylamide and methacrylamide may be collectively referred to as (meth) acrylamide); preferable.
- sulfonic acid compound having a vinyl group vinyl sulfonic acid and alkali metal salts or ammonium salts thereof such as lithium vinyl sulfonate are preferable.
- sulfonic acid compound having an allyl group allyl sulfonic acid, sodium allyl sulfonate, potassium allyl sulfonate and the like are preferable.
- sulfonic acid compounds having an isopropenyl group include isopropenyl sulfonic acid, sodium 4-isopropenylbenzene-1-sulfonate, sodium 3-isopropenylbenzene-1-sulfonate, and 2-isopropenylbenzene-1-sulfone.
- Sodium acid, potassium 4-isopropenylbenzene-1-sulfonate and the like are preferable.
- sulfonic acid compound having a styryl group 4-styrene sulfonic acid, 2-styrene sulfonic acid and alkali metal salts, alkaline earth metal salts, ammonium salts thereof such as lithium 4-styrene sulfonate are preferable.
- an alkali metal salt of sulfomethyl (meth) acrylate an alkaline earth metal salt of sulfomethyl (meth) acrylate, such as a sulfomethyl (meth) acrylate sodium salt, is preferable.
- sulfonic acid compound having a (meth) acrylamide group a compound represented by the following formula (4 ') is preferable.
- R 1 , R 5 , R 6 , M, and n1 in the above formula (4 ′) are the same as those in the above formula (4).
- Examples of the compound represented by the above formula (4 ′) include 1- (meth) acrylamide-methanesulfonic acid, 1- (meth) acrylamide-potassium methanesulfonate, 2- (meth) acrylamide-ethanesulfonic acid, 2- (Meth) acrylamide-sodium ethanesulfonate, 2- (meth) acrylamide-propanesulfonic acid, 2- (meth) acrylamide-potassium propanesulfonate, 2- (meth) acrylamide-2-methyl-propanesulfonic acid ((meta ) Acrylamide-t-butylsulfonic acid), 2- (meth) acrylamide-2-methyl-propanesulfonic acid sodium salt, 2- (meth) acrylamide-2-methyl-propanesulfonic acid potassium salt, 2- (meth) acrylamide -2-Rubidium propane sulfonate 2- (meth) acrylamide-2-methyl-propanesulfonic acid calcium
- a sulfonic acid compound having a (meth) acrylamide group is preferable, a compound represented by the above formula (4 ′) is more preferable, and 2- (meth) acrylamide-2-methyl-propyl is preferable.
- M is preferably an alkali metal ion other than a hydrogen atom, a half-valent alkaline earth metal ion, an ammonium ion, or an amine ion in the compound represented by the general formula (1 ') will be described.
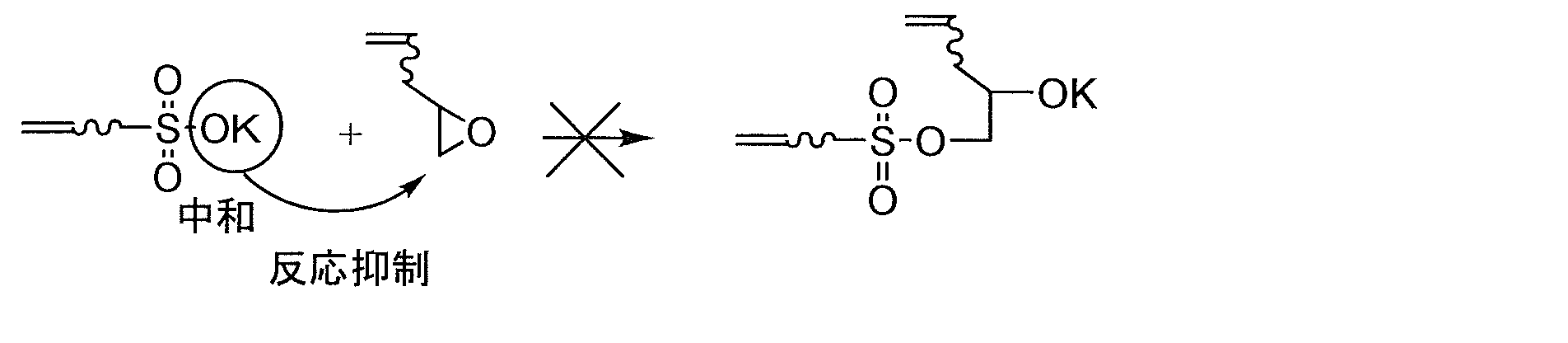
- the sulfonic acid group is represented by a compound having an epoxy group described below (typically represented by the following general formula (2 ′)). May react with an epoxy group contained in the compound) to form a gel.
- a typical reaction formula for the reaction between the epoxy group and the sulfonic acid group is described below.
- alkali metal ions alkaline earth metal ions, ammonium ions, and amine ions, which are counter cations
- alkali metal ions that tend to have high reaction inhibiting power and stability tend to be preferable.
- the alkali metals sodium or potassium is preferable, and potassium is more preferable. The reason is not clear, but when the counter cation is potassium, the thermal stability may be higher than that of sodium.
- DSC chart thermal stability comparison data of representative compounds among the compounds having a polymerizable functional group having a carbon-carbon double bond and a sulfonic acid-containing group is shown in FIG.
- Examples of the compound having a polymerizable functional group having a carbon-carbon double bond corresponding to the structural unit represented by the formula (2) and an epoxy group include a compound represented by the following general formula (2 ′).
- an epoxy compound having a vinyl group an epoxy compound having a vinyl ether group, an epoxy compound having an allyl ether group, an epoxy compound having an isopropenyl ether group, and an epoxy having a styryl group
- an epoxy compound having a (meth) acryloyloxy group is relatively preferred.
- epoxy compound having a vinyl group vinyl-cyclohexene monooxide, butadiene-monooxide, pentadiene-monooxide, hexadiene-monooxide and the like are preferable.
- epoxy compounds having a vinyl ether group examples include vinyl glycidyl ether, butanediol-divinyl ether monooxide, cyclohexanedimethanol-divinyl ether monooxide, 4-glycidyloxymethyl-1-vinyloxymethyl-cyclohexane, diethylene glycol-divinyl ether monooxide. Tripropylene glycol-divinyl ether monoxide, 4-vinyloxy-1-glycidyloxy-butane and the like are preferable.
- Examples of the epoxy compound having an allyl ether group include allyl-glycidyl ether, allyl-epoxy ether, butanediol-diallyl ether monooxide, cyclohexanedimethanol-diallyl ether monooxide, 4-glycidyloxymethyl-1-allyloxymethyl-cyclohexane.
- Diethylene glycol-diallyl ether monooxide, tripropylene glycol-diallyl ether monooxide, 4-allyloxy-1-glycidyloxy-butane and the like are preferable.
- Examples of the epoxy compound having an isopropenyl ether group include isopropenyl glycidyl ether, isopropenyl epoxy ether, butanediol-diisopropenyl ether monooxide, cyclohexanedimethanol-diisopropenyl ether monooxide, 4-glycidyloxymethyl-1- Isopropenyloxymethyl-cyclohexane, diethylene glycol-diisopropenyl ether monooxide, tripropylene glycol-diisopropenyl ether monooxide, 4-isopropenyloxy-1-glycidyloxy-butane, 4-isopropenyl-1-glycidyloxy- Benzene and the like are preferred.
- epoxy compounds having a styryl group examples include divinylbenzene-monooxide, 4-glycidyloxy-styrene, 3-glycidyloxy-styrene, 2-glycidyloxy-styrene, 4-epoxyoxy-styrene, styrylcarboxylic acid epoxy ester, styryl. Carboxylic acid glycidyl ester and the like are preferable.
- the epoxy compound having a (meth) acryloyloxy group a compound represented by the following formula (5 ′) is preferable.
- Examples of the compound represented by the above formula (5 ′) include glycidyl- (meth) acrylate, epoxy- (meth) acrylate, 2-glycidyloxy-ethyl- (meth) acrylate, 3-glycidyloxy-propyl- ( (Meth) acrylate, 4-glycidyloxy-butyl- (meth) acrylate, 6-glycidyloxy-hexyl- (meth) acrylate, 5-glycidyloxy-3-oxapentyl- (meth) acrylate, 3-glycidyloxy-2- Hydroxy-propyl- (meth) acrylate, 2,3-bis (glycidyloxy) -propyl- (meth) acrylate, trimethylolpropane-diglycidyl ether- (meth) acrylate, ⁇ 4-glycidyloxyphenyl ⁇ - ⁇ (4 -(Meth) acryloyloxy- -
- an epoxy compound having a (meth) acryloyloxy group, an epoxy compound having an allyl ether group, and an epoxy compound having a styryl group are preferable, and glycidyl (meth) acrylate, 4- Glycidyloxy-butyl- (meth) acrylate, allyl glycidyl ether, and 4-glycidyloxystyrene are more preferable.
- Examples of the compound having a polymerizable functional group having a carbon-carbon double bond corresponding to the structural unit represented by the formula (3) and an alkoxysilyl group include compounds represented by the following general formula (3 ′). .
- An alkoxysilyl compound having an isopropenyl ether group, an alkoxysilyl compound having a styryl group, and an alkoxysilyl compound having a (meth) acryloyloxy group are relatively preferable.
- alkoxysilyl compounds having a vinyl group examples include vinyl-trimethoxysilane, vinyl-triethoxysilane, vinyl-tripropoxysilane, vinyl-triisopropoxysilane, vinyl-tributoxysilane, vinyl-methyldimethoxysilane, and vinyl-phenyl. Dimethoxysilane, vinyl-ethyldiethoxysilane, vinyl-diethylmonoethoxysilane, vinyl-dimethylmonobutoxysilane and the like are preferable.
- alkoxysilyl compound having a vinyl ether group vinyloxy-ethyltrimethoxysilane, vinyloxy-propyltrimethoxysilane and the like are preferable.
- alkoxysilyl compound having an allyl group examples include allyltrimethoxysilane, allyltriethoxysilane, allyltripropoxysilane, allyltriisopropoxysilane, allyltributoxysilane, isopropenyltriethoxysilane, allylmethyldimethoxysilane, allylphenyldimethoxy.
- Silane, allylethyldiethoxysilane, allyldiethylmonoethoxysilane, allyldimethylmonobutoxysilane and the like are preferable.
- alkoxysilyl compound having an allyl ether group allyloxy-ethyltrimethoxysilane, allyloxy-propyltrimethoxysilane, allyloxy-propyltriethoxysilane and the like are preferable.
- alkoxysilyl compound having an isopropenyl group 4-isopropenyl-1-trimethoxysilyl-benzene, 4-isopropenyl-1-triethoxysilyl-benzene and the like are preferable.
- alkoxysilyl compound having an isopropenyl ether group isopropenyloxy-propyltrimethoxysilane, isopropenyloxy-propyltriethoxysilane and the like are preferable.
- alkoxysilyl compound having a styryl group styryl-trimethoxysilane, styryl-triethoxysilane, styryl-tributoxysilane, styryl-methyldimethoxysilane and the like are preferable.
- alkoxysilyl compound having a (meth) acryloyloxy group a compound represented by the following formula (6 ') is preferable.
- R 3 , R 4 , R 10 and n in the above formula (6 ′) are the same as those in the above formula (6).
- Examples of the compound represented by the above formula (6 ′) include (meth) acryloyloxy-ethyltrimethoxysilane, (meth) acryloyloxy-propyl-trimethoxysilane, and (meth) acryloyloxy-butyl-trimethoxysilane.
- an alkoxysilyl compound having a vinyl group an alkoxysilyl compound having a styryl group, and an alkoxysilyl compound having a (meth) acryloyloxy group are preferable.
- Vinyl-trimethoxysilane More preferred are vinyl-triethoxysilane, styryl-trimethoxysilane, styryl-triethoxysilane, (meth) acryloyloxy-propyl-trimethoxysilane, and (meth) acryloyloxy-propyl-triethoxysilane.
- the copolymer (i) may contain other structural units other than the structural units represented by the general formulas (1) to (3).
- the other structural unit can be obtained, for example, by further adding a compound corresponding to the other structural unit to the monomer mixture containing the compounds represented by the above (1 ′) to (3 ′) and polymerizing. Can do.
- Examples of compounds corresponding to other structural units include acrylic acid, methacrylic acid, methyl (meth) acrylate, butyl (meth) acrylate, isobornyl (meth) acrylate, tetrahydrofurfuryl (meth) acrylate, and phenyl (meth) acrylate.
- Tribromophenyl (meth) acrylate Tribromophenyl (meth) acrylate, hydroxyethyl (meth) acrylate, ethyl phosphate (meth) acrylate, tetramethylpiperidyl (meth) acrylate, perfluorooctylethyl (meth) acrylate, thioglycidyl (meth) acrylate, styrene
- Examples include acrylonitrile, divinylbenzene, and allyl (meth) acrylate.
- divinylbenzene and allyl (meth) acrylate it is desirable to use it in a small quantity so that the copolymer (i) may not be gelled.
- the ratio (molar ratio) (a + b + c) / d of the total number of structural units (a + b + c) of the structural units represented by formulas (1), (2), and (3) to the number of other structural units (d) is The range is usually 100/0 to 30/70, more preferably 100/0 to 50/50, and still more preferably 100/0 to 60/40.
- the molar ratio (a + b + c) / d is usually in the range of 99.9 / 0.1 to 30/70, more preferably 99/1 to 50. / 50, more preferably 95/5 to 60/40.
- (a + b + c) / d is 70/30 or more, preferably 80/20 or more.
- the ratio (mass ratio) (Wa + Wb + Wc) / Wd of the total weight (Wa + Wb + Wc) of the structural units represented by the formulas (1), (2), and (3) and the weight (Wd) of the other structural units Is preferably in the range of 100/0 to 30/70, more preferably in the range of 100/0 to 50/50, and still more preferably in the range of 100/0 to 60/40.
- the copolymer (i) used in the present invention is typically a compound represented by the formula (1 ′), a compound represented by the formula (2 ′), a compound represented by the formula (3 ′), In addition, a monomer mixture containing a compound corresponding to another constituent unit contained as necessary is obtained by solution polymerization in the presence of a polymerization initiator.
- a polymerization initiator there is no restriction
- the number of repeating structural units and the molecular weight of the copolymer (i) used in the present invention are mainly controlled by the type of solvent, the concentration of the compound (monomer), the amount of polymerization initiator, the reaction temperature, and the like.
- the number of repeating structural units of the copolymer (i) is usually in the range of 1 to 10,000, preferably in the range of 3 to 3,000, and more preferably in the range of 30 to 1,500.
- the weight average molecular weight (Mw) in terms of standard polymethyl methacrylate measured by GPC of the copolymer (i) is usually in the range of 500 to 3,000,000, but is preferably from the viewpoint of durability and solubility. Is from 1,000 to 1,000,000, more preferably from 10,000 to 500,000.
- the ratio of the weight average molecular weight (Mw) to the number average molecular weight (Mn) of the copolymer (i) used in the present invention the molecular weight distribution Mw / Mn is usually 1 to 10, preferably 1 to 6, more preferably 1. ⁇ 4.
- Mw / Mn is within this range, the copolymer (i) or the composition containing the copolymer (i) has excellent solubility or dispersibility in a solvent, and is obtained by curing the composition. It tends to be excellent in transparency or smoothness of a film made of a cured product, for example, the cured product.
- a radical polymerization initiator is preferable.
- radical polymerization initiators examples include azo compounds (azo radical polymerization initiators) and organic peroxides.
- examples of the azo compound include azobisisobutyronitrile (AIBN), and examples of the organic peroxide include ketone peroxides, diacyl peroxides (such as benzoyl peroxide), hydroperoxides, dialkyl peroxides, Examples include peroxyketals, alkyl peresters, and percarbonates.
- radical polymerization initiators such as organic peroxides, particularly alkyl peresters such as t-butylperoxy-2-ethylhexanoate are preferred.
- the addition amount of these polymerization initiators is included in the compound represented by the formula (1 ′), the compound represented by the formula (2 ′), the compound represented by the formula (3 ′), and others as necessary. Is approximately in the range of 0.01 to 10 wt%, preferably in the range of 0.1 to 5 wt%, more preferably in the range of 0.2 to 3 wt%, with respect to the total weight of the compounds corresponding to .
- the polymerization solvent is not particularly limited as long as it does not cause problems such as inhibiting the polymerization reaction, but the compound represented by the formula (1 ′), the compound represented by the formula (2 ′), the formula (3) A highly polar solvent having a high solubility of the compound represented by ') and a compound corresponding to another structural unit contained as necessary tends to be good.
- Examples of such a polymerization solvent include methanol, ethanol, isopropanol (IPA), 1-propanol, 1-butanol, 1-pentanol, isopentanol, 1-hexanol, 1-octanol, cyclohexanol, benzyl alcohol, Alcohols such as ethylene glycol, propylene glycol, ethylene glycol monomethyl ether (2-methoxyethanol), propylene glycol monomethyl ether (1-methoxy-2-propanol), acetonitrile, sulfolane, dimethyl sulfoxide, N, N-dimethylformamide (DMF) ), N, N-dimethylacetamide (DMAc), aprotic polar solvents such as N, N-dimethylhumidazolidinone (DMI), water, and mixtures thereof. It is.
- IPA isopropanol
- 1-butanol 1-pentanol
- isopentanol
- the polymerization temperature is mainly set by the 10-hour half-life temperature of the radical polymerization initiator, but is generally in the range of room temperature to 200 ° C, preferably in the range of 30 to 120 ° C, more preferably in the range of 40 to 100 ° C. It is.
- the copolymer (i) may be used as a cured product, film or laminate for applications requiring high transparency, an amorphous polymer (with low crystallinity) having high transparency. , Tm (melting point) is not measured, or heat of fusion is small, which corresponds to an amorphous polymer or a latent crystalline polymer).
- Such a highly transparent copolymer (i) can be produced, for example, by setting each constituent unit ratio of formulas (1) to (3) within a desired range.
- these core / shell structures generally tend to be micron-sized particles. It tends to become secondary particles and eventually become large micron-sized particle aggregates.
- these micron-sized core / shell structures have a particle size that exceeds a quarter wavelength (about 100 nm) of light, so that light is scattered and transparency is lowered. Can not be used.
- the copolymer (i) used in the present invention does not form a higher order structure such as a core shell.
- the core-shell structure formed of two kinds of polymers or polymer raw materials or the like tends to have two Tg (glass transition points) observed.
- Such a copolymer (i) that does not form a higher-order structure can be prepared, for example, by dissolving a compound (monomer) serving as each structural unit in a solvent and performing polymerization (solution polymerization). it can.
- the copolymer (i) thus produced is usually a high molecular weight body having a large number of sulfonic acid-containing groups and often has a property of being soluble only in water. Therefore, in this case, unless a large amount of water is used as the polymerization solvent, the copolymer is precipitated from the polymerization solution as the polymerization reaction proceeds.
- the desired copolymer can be obtained simply by filtering and drying after completion of the polymerization reaction.
- a target copolymer is obtained by a method of adding a solvent, mixing and stirring, and drying a filter cake obtained by filtration.
- the copolymer (i) of the present invention is cured mainly by a crosslinking reaction with an amino resin (ii) described later to be a cured product of the present invention.
- the copolymer (i) is composed of an epoxy group and an alkoxysilyl group. Since at least one group selected from the groups is contained, the copolymers (i) may cause a cross-linking reaction to become a partial skeleton of the cured product of the present invention.
- the reaction of the epoxy group and alkoxysilyl group is usually accelerated by heating.
- a method for promoting a reaction other than heating for example, a method of irradiating microwaves, which is a kind of radiation, can be cited.
- the reaction between epoxy groups is represented by the general formula (11), and it is preferable to react by heating.
- the heating temperature is approximately in the range of 30 to 250 ° C, preferably in the range of 30 to 200 ° C, and more preferably in the range of 30 to 150 ° C.
- This reaction between epoxy groups tends to be accelerated by the presence of a catalyst typified by a cation such as an acid and an anion such as a base.
- an epoxy group and an alkoxysilyl group hardly react directly, and usually a reaction occurs between a silanol group obtained by hydrolyzing an alkoxysilyl group and an epoxy group.
- the reaction between the epoxy group and the alkoxysilyl group is also preferably performed by heating.
- the heating temperature is approximately in the range of 30 to 300 ° C, preferably in the range of 50 to 250 ° C, and more preferably in the range of 100 to 200 ° C.
- the hydrolysis reaction of the alkoxysilyl group and the reaction between the epoxy group and the silanol group tend to be accelerated by the presence of a catalyst typified by a cation such as an acid and an anion such as a base. Even when such a catalyst is used, it is preferable to react by heating.
- the heating temperature is approximately in the range of 30 to 250 ° C, preferably in the range of 30 to 200 ° C, and more preferably in the range of 30 to 180 ° C.
- reaction formula between alkoxysilyl groups is represented by the general formula (13), and it is preferable to react by heating.
- the heating temperature is approximately in the range of 30 to 250 ° C, preferably in the range of 30 to 200 ° C, and more preferably in the range of 30 to 180 ° C.
- the alkoxysilyl group is hydrolyzed relatively easily by moisture and converted into a silanol group.
- This silanol group is highly reactive, and the reaction between silanol groups occurs more easily than the reaction between alkoxysilyl groups. Therefore, the reaction between alkoxysilyl groups is usually carried out as a reaction between silanol groups hydrolyzed by moisture, or a reaction between a silanol group and an alkoxysilyl group.
- the reaction between the silanol groups and the reaction between the silanol group and the alkoxysilyl group are preferably performed by heating.
- the heating temperature is approximately in the range of 30 to 200 ° C, preferably in the range of 30 to 180 ° C, and more preferably in the range of 30 to 150 ° C.
- Reactions between alkoxysilyl groups, hydrolysis reactions of alkoxysilyl groups, reactions between alkoxysilyl groups and silanol groups, and reactions between silanol groups include cations such as acids, anions such as bases, alkoxy titanium and tin oxide, etc.
- the reaction tends to be accelerated by the presence of a catalyst typified by a metal compound.
- the cured product (for example, film) of the present invention can be obtained by curing a composition of the copolymer (i) and the amino resin (ii).
- a copolymer having a sulfonic acid group, an epoxy group, and an alkoxysilyl group in the molecule (the above general formula (10 ′)) and a melamine resin that is a typical amino resin (the following general formula (15))
- a reaction for example, crosslinking by a polycondensation reaction of a hydroxymethyl group or an alkoxymethyl group represented by the following general formulas (16) to (19)
- the polycondensation reaction with these hydroxymethyl groups or alkoxymethyl groups usually proceeds by heating.
- a curing method other than heating for example, a method of curing by irradiating a microwave which is a kind of radiation can be cited.
- reaction between the hydroxymethyl group and the epoxy group is represented by the general formula (16). This reaction proceeds slowly, and this reaction tends to be promoted by adding Arrhenius acid or Lewis acidic compound as a catalyst.
- the reaction temperature is approximately in the range of 30 to 300 ° C, preferably in the range of 50 to 250 ° C, more preferably in the range of 80 to 180 ° C.
- reaction between the alkoxymethyl group and the epoxy group is represented by the general formula (17), since the reaction tends to be less advanced than the reaction (16) between the hydroxymethyl group and the epoxy group, the epoxy group is usually A condensation reaction occurs with a secondary hydroxy group produced by a ring-opening reaction with an active hydrogen group such as silanol.
- the reaction between the alkoxymethyl group and the secondary hydroxy group is slow as in the reaction of the general formula (16), and when Arrhenius acid or Lewis acidic compound is added as a catalyst, this reaction tends to be promoted.
- the reaction temperature is approximately in the range of 30 to 300 ° C, preferably in the range of 50 to 250 ° C, more preferably in the range of 80 to 180 ° C.
- reaction temperature is approximately in the range of 20 to 300 ° C, preferably in the range of 40 to 250 ° C, more preferably in the range of 80 to 180 ° C.
- reaction between the alkoxymethyl group and the silanol group produced by hydrolysis of the alkoxysilyl group derived from the copolymer (i) is represented by the general formula (19).
- This reaction also tends to be faster than the reaction with the secondary alcohol represented by the general formula (17), similarly to the reaction represented by the general formula (18).
- Arrhenius acid or Lewis acidic compound may be added as a catalyst.
- the reaction temperature is approximately in the range of 20 to 300 ° C, preferably in the range of 40 to 250 ° C, more preferably in the range of 80 to 180 ° C.
- the amino resin (ii) used in the present invention is a resin produced by polycondensation of a compound containing an amino group and formaldehyde.
- melamine resin that is polycondensation with melamine
- urea that is polycondensation with urea (Urea) resin
- aniline resin which is polycondensation with aniline.
- these amino resins also include alkylated amino resins in which some or all of the hydroxy groups produced by the reaction of formaldehyde are substituted with alkoxy groups.
- the amino resin (ii) is preferably an amino resin (ii1) represented by the following general formula (7).
- R 30 represents a hydrogen atom, an alkyl group having 1 to 10 carbon atoms, a hydroxymethyl group, or an alkoxymethyl group having 1 to 10 carbon atoms
- R 40 represents a hydroxyl group, a hydrogen atom, carbon Represents an alkyl group having 1 to 10 carbon atoms or an alkoxy group having 1 to 10 carbon atoms
- q190 is an integer of 1 to 90
- MC is a host represented by any one of the following general formulas (8) to (10): Represents a nucleus
- # 2 is a bond that bonds to # 1 in the following general formulas (8) to (10), and the numbers of # 1 and # 2 are the same.
- q 030 is an integer of 0 to 30, q 030 may be the same as or different from each other, and R 30 and R 40 are the same as defined in formula (7).
- q 050 is an integer of 0 to 50
- X represents an oxygen atom or a sulfur atom
- R 30 and R 40 are the same as defined in formula (7).
- q 050 is an integer of 0 to 50.
- R 30 is preferably an alkoxymethyl group or a methylol group
- R 40 is preferably an alkoxy group having 1 to 10 carbon atoms
- q 030 is preferably an integer of 0 to 10.
- R 30 is preferably an alkoxymethyl group or a methylol group
- R 40 is preferably an alkoxy group having 1 to 10 carbon atoms
- q 050 is preferably an integer of 1 to 10.
- q 050 is preferably an integer of 1 to 10.
- the melamine resin examples include hydrogenated melamine resin, methylated melamine resin, ethylated melamine resin, n-propylated melamine resin, isopropylated melamine resin, n-butylated melamine resin, isobutylated melamine resin, and n-hexyl.
- urea (urea) resins include, for example, hydrogenated urea (urea) resin, methylated urea (urea) resin, ethylated urea (urea) resin, n-propylated urea (urea) resin, and isopropylated urea.
- Rea n-butylated urea (urea) resin, isobutylated urea (urea) resin, n-hexylated urea (urea) resin, n-octylated urea (urea) resin, n-decylated urea (urea) ) Resin, n-dodecylated urea (urea) resin and the like.
- aniline resin examples include hydrogenated aniline resin, methylated aniline resin, ethylated aniline resin, n-propylated aniline resin, isopropylated aniline resin, n-butylated aniline resin, isobutylated aniline resin, and n-hexyl. And aniline resin, n-octylated aniline resin, n-decylated aniline resin, and n-dodecylated aniline resin.
- the weight ratio (i) / (ii) between the copolymer (i) and the amino resin (ii) contained in the composition used in the present invention is approximately in the range of 99/1 to 1/99, preferably 95/5. It is in the range of ⁇ 5 / 95, more preferably in the range of 90/10 to 10/90.
- composition to be a cured product of the present invention for example, a film made of the cured product
- other components are used as long as the effects of the present invention are not impaired. Can be included.
- Inorganic particles (iii) can be included.
- inorganic particles (iii) include silver particles, copper particles, copper oxide particles, silica particles, hollow silica particles, alumina particles, iron oxide particles, cobalt oxide particles, zirconia particles, titanium dioxide particles, and oxidation.
- Antimony particles and the like can be mentioned. Among them, silica particles, hollow silica particles, zirconia particles, and titanium dioxide particles are preferable, and silica particles, zirconia particles, and titanium dioxide particles are more preferable.
- endless particles also include endless particles whose surface is modified with an organic group having an alkyl group or a (meth) acryloyl group for the purpose of improving dispersibility. Furthermore, as these inorganic particles (iii), from the viewpoint of ensuring transparency, inorganic particles having a diameter of nanometer size tend to be preferable. Particles (silica nanoparticles, zirconium dioxide nanoparticles, titanium dioxide nanoparticles, etc.) tend to be more preferable.
- the composition includes the copolymer (i), the amino resin (ii), and the inorganic Copolymer (i) 5 to 98 parts by weight, amino resin (ii) 1 to 70 parts by weight, and inorganic particles (iii) 1 to 90 parts by weight with respect to 100 parts by weight of the total weight of the particles (iii) More preferably, the copolymer (i) is contained in an amount of 10 to 70 parts by weight, the amino resin (ii) in an amount of 5 to 40 parts by weight, and the inorganic particles (iii) in an amount of 25 to 75 parts by weight. 20) to 60 parts by weight, amino resin (ii) 10 to 30 parts by weight, and inorganic particles (iii) 30 to 70 parts by weight are more preferable.
- the composition includes compounds having an epoxy group other than the copolymer (i), compounds having a hydroxyl group, compounds having a mercapto group, compounds having a carboxyl group, compounds having an amino group, acid anhydrides, and the like. Reactive compounds can be included.
- a polyvalent epoxy compound having two or more epoxy groups in the molecule is preferable.
- the polyvalent epoxy compound include bisphenol A bis (glycidyl ether), bisphenol F bis (glycidyl ether), hydrogenated bisphenol A bis (glycidyl ether), N, N ′, N ′′ -trisglycidyl-isocyanurate, and isocyanate.
- Nurate polyglycidyl ether (Nissan Chemical TEPIC-PAS B22, TEPIC-PAS B26), phenol novolac type polyglycidyl ether (DIC Corporation N-730, Mitsubishi Chemical 152), 1.1.2.2.-tetrakis (4- Glycidyloxy-phenyl) ethane, N, N, N ′, N′-tetraglycidyl-diaminodiphenylmethane, trimethylolpropane-triglycidyl ether, neopentyl glycol diglycidyl ether, butanediol jig Sidyl ether, polyethylene glycol diglycidyl ether (Mitsubishi Chemical YDE205), cyclohexanedicarboxylic acid diglycidyl ester, phthalic acid diglycidyl ester, 3,4-epoxycyclohexylmethyl-3 ′, 4′-epoxycyclohe
- the reaction in the reaction route (B) is mainly performed in the reaction of the above formula (20) (reaction with the epoxy group of the copolymer (i)).
- the reaction in the reaction route (G) mainly occurs and curing occurs.
- the compound having a hydroxyl group is preferably a polyvalent hydroxyl compound having two or more hydroxyl groups.
- polyhydric hydroxyl compounds include ethylene glycol, diethylene glycol, 1,2-propylene glycol, glycerin, trimethylolpropane, pentaerythritol, dipentaerythritol, xylylenediol, resorcin, bisphenol A, and phenol formaldehyde resin (Mitsui Chemicals).
- a condensation reaction product of melamine and formaldehyde a condensation reaction product of melamine, formaldehyde and lower alcohol, a condensation reaction product of urea and formaldehyde, a condensation reaction product of urea, formaldehyde and lower alcohol.
- Other examples of the compound having a hydroxyl group include a condensation reaction product of melamine and lower alcohol, a condensation reaction product of urea and lower alcohol, and the like. Since these are easily hydrolyzed with water to generate hydroxyl groups, they can be used as the compounds having a hydroxyl group of the present invention.
- the reaction in the reaction route (C) is performed in the reaction of the above formula (20) (reaction with the epoxy group of the copolymer (i)).
- the reaction in the reaction route (H) mainly occurs and curing occurs.
- the compound having a mercapto group is preferably a polyvalent mercapto compound having two or more mercapto groups.
- examples of the polyvalent mercapto compound include glycerin dithioglycolate.
- a polyvalent mercapto compound exemplified in paragraph [0120] of the pamphlet of International Publication No. 2014/168122 can also be used as the polyvalent mercapto compound.
- reaction route (D) When a compound having a carboxyl group is used as the reactive compound, in the reaction of the above formula (20) (reaction with the epoxy group of the copolymer (i)), the reaction in the reaction route (D) is performed. In the reaction of the above formula (21) (reaction with the alkoxysilyl group of the copolymer (i)), the reaction in the reaction route (I) mainly occurs and curing occurs.
- the compound having a carboxyl group is preferably a polyvalent carboxyl compound having two or more carboxyl groups.
- the polyvalent carboxyl compound include maleic acid.
- the polyvalent carboxyl compound illustrated by paragraph [0122] of international publication 2014/168122 pamphlet can also be used as said polyvalent carboxyl compound.
- reaction route (E) When a compound having an amino group is used as the reactive compound, in the reaction of the above formula (20) (reaction with the epoxy group of the copolymer (i)), the reaction in the reaction route (E) is performed. In the reaction of the above formula (21) (reaction with the alkoxysilyl group of the copolymer (j)), the reaction in the reaction route (I) mainly occurs and curing occurs.
- the compound having an amino group is preferably a polyvalent amino compound having two or more amino groups.
- the polyvalent amino compound include phenylenediamine.
- the polyvalent amino compound illustrated by paragraph [0124] of international publication 2014/168122 pamphlet can also be used as the said polyvalent amino compound.
- the reaction in the reaction route (G) mainly occurs in the reaction of the above formula (20) (reaction with the epoxy group of the copolymer (i)).
- the reaction in the reaction route (L) mainly occurs and curing occurs.
- acid anhydride examples include maleic anhydride and succinic anhydride.
- acid anhydride illustrated in paragraph [0126] of international publication 2014/168122 pamphlet can also be used as the said acid anhydride.
- an acid catalyst for the purpose of improving the curing rate of the composition of the present invention, an acid catalyst, a base catalyst, or the like may be used.
- the acid catalyst examples include hydrochloric acid and sulfuric acid. Further, the acid catalyst exemplified in paragraph [0139] of International Publication No. 2014/168122 can also be used as the acid catalyst, and the base catalyst exemplified in the same paragraph can be used as the base catalyst.
- the addition amount of the acid catalyst or the base catalyst is preferably in the range of 0.1 to 20% by weight, and in the range of 0.2 to 10% by weight with respect to the total of the copolymer (i) and the amino resin (ii). Is more preferable, and the range of 0.3 to 5% by weight is more preferable.
- the above composition is used.
- An additive or a modifier may be added to the product.
- additives or modifiers include UV absorbers, HALS (hindered amine light stabilizers), antioxidants, radical scavengers, fillers other than inorganic particles, pigments, color correction agents, and high refractive index agents. , Fragrances, surfactants, antifoaming agents, leveling agents, sagging prevention materials, and other modifiers.
- the UV absorber and HALS are added mainly for further improving weather resistance.
- the antioxidant and radical scavenger are added mainly for improving heat resistance or preventing deterioration.
- the filler is added mainly for improving wear or imparting toughness.
- a filler exemplified in paragraph [0180] of International Publication No. 2014/168122 Pamphlet can be used.
- pigments and dyes, color complementary agents, high refractive index agents, fragrances and the like can be used, and those exemplified as these additives can be used.
- a cured product having a gradient of sulfonic acid concentration that is preferably used in the present invention typically a film made of the cured product, a degree of gradient Sa / Da ⁇ 1.1
- the addition of the surfactant described above is performed. It is as effective as selective use of polar solvents.
- surfactant for example, a surfactant exemplified in paragraph [0181] of International Publication No. 2014/168122 Pamphlet can be used.
- sodium lauryl sulfate, sodium disterylsulfosuccinate, sodium dialkylsulfosuccinate (Kao Perex TR, Perex OT-P), sorbitan stearate, and lauryl betaine tend to be preferable.
- the antifoaming agent, leveling agent, and sagging prevention material are added mainly for the purpose of improving coating properties, imparting ideological smoothness, and improving the appearance of a cured product (for example, a film made of the cured product). .
- an antifoaming agent for example, an antifoaming agent exemplified in paragraph [0182] of International Publication No. 2014/168122 Pamphlet can be used.
- leveling agent for example, a leveling agent exemplified in paragraph [0183] of International Publication No. 2014/168122 Pamphlet can be used.
- the sagging prevention material for example, the sagging prevention material exemplified in paragraph [0184] of International Publication No. 2014/168122 Pamphlet can be used.
- Examples of the other modifiers include polyacrylate and polymethacrylate.
- the composition to be the cured product of the present invention may contain a hydrolyzable silicon compound other than the copolymer (i) and a hydrolysis product thereof, for example, alkoxysilane, halogenated silane, and hydroxylane. Good.
- a hydrolyzable silicon compound and its hydrolysis product are included in the composition, the resulting cured product (for example, a film made of the cured product) adsorbs contaminants in the atmosphere. It may be easy and may be difficult to desorb and is easily contaminated. Therefore, when maintaining hydrophilicity for a long period of time is a problem, it may be desirable not to actively add these hydrolyzable silicon compounds and their hydrolysis products.
- the concentration of the sulfonic acid-containing group derived from the copolymer (i) is from the inside of the cured product to the cured product. In some cases, it is concentrated (inclined) in such a manner that it gradually becomes higher toward the outer surface direction. And it is estimated that the hydrophilicity of the hardened
- the main principle of forming this inclined structure is that “when the polar solvent added in advance is evaporated, the hydrophilic copolymer (i) having a sulfonic acid-containing group is accompanied by evaporation of the polar solvent, And “concentrate and cure on the surface by adding a surfactant and accompanying the surfactant to the surface”.
- a cured product layer for example, a film
- Sa is the sulfonic acid concentration on the outer surface in the opposite direction to the base material, and the intermediate point between the interface contacting the base material and the outer surface
- the gradient of the copolymer (i) of the present invention having a sulfonic acid-containing group is represented by a sulfonic acid concentration ratio (Sa / Da). That is, a large concentration ratio (Sa / Da) of sulfonic acid indicates that a large amount of sulfonic acid is concentrated on the outer surface of the cured product layer.
- the “intermediate point between the interface in contact with the substrate and the outer surface” is usually a point where the depth from the outer surface is 1 ⁇ 2 of the film thickness toward the interface in contact with the substrate ( In this specification, this point is also referred to as “a point at which the film thickness is 1 ⁇ 2”.)
- the phrase “acid” and “acid concentration”, respectively, - means "concentration of the -SO 3 M group”"SO 3 M group” and.
- the gradient ⁇ concentration ratio of sulfonic acid (Sa / Da) ⁇ of a cured product (typically a film made of the cured product) obtained by curing the composition of the present invention is usually 1.01 to 1000.
- the range is preferably 1.1 to 100, and more preferably 1.2 to 60.
- the lower limit value of the inclination is more preferably 1.1 or more.
- the film thickness of the film formed from the cured product formed by curing the composition of the present invention on the substrate is not particularly limited, but is approximately in the range of 0.0001 to 3000 ⁇ m, preferably 0.00.
- the range is from 01 to 300 ⁇ m, more preferably from 0.1 to 30 ⁇ m.
- the cured product (typically the membrane) having a gradient of sulfonic acid concentration exhibits higher hydrophilicity.
- more hydrophilic co-polymers are used. Coalescence (i) is required.
- a cured product having a sulfonic acid concentration gradient typically a film made of the cured product
- it has a high crosslink density due to oxymethylamino bonds while having high hydrophilicity.
- Property, wear resistance, durability (hydrophilicity maintenance), etc. can be improved.
- a cured product (typically a film comprising the cured product) obtained by curing the composition of the present invention has a gradient in the concentration of —SO 3 M groups, the hydrophilicity of the obtained cured product It is thought that the balance between the property (such as water contact angle) and the hardness is better.
- any solvent may be used as long as it can uniformly dissolve or disperse the above-described components. Such solvents may be used alone or in combination of two or more.
- the hydrophilic copolymer (i) is inclined in the thickness direction (sulfonic acid is cured of the cured product).
- a highly polar solvent having a high SP value solubility parameter ⁇
- one or more solvents having an SP value (solubility parameter ⁇ ) of at least 9.3 or more should be used. Is preferred.
- this solvent is used in combination with a solvent having an SP value of 9.3 or more. It is preferable that it has a lower boiling point (faster evaporation rate) than a solvent having an SP value of 9.3 or more.
- the SP value (solubility parameter ⁇ ) (cal / cm 3 ) 1/2 of the solvent is a value calculated by the following formulas (1) to (5).
- (1) latent heat of vaporization per mol Hb 21 ⁇ (273 + Tb) [unit: cal / mol], Tb: boiling point of solvent (° C.)
- the surface of the applied mixture in contact with the outside air is accompanied by a solvent and has a hydrophilic sulfonic acid-containing group.
- the copolymer (i) moves and the hydrophilic sulfonic acid-containing groups are concentrated on the surface thereof, and hydrophilic groups are formed on the outer surface of the cured product (typically a film) obtained in the present invention.
- a gradient structure in which sulfonic acid-containing groups are concentrated is formed.
- the solubility parameter ⁇ (cal / cm 3 ) 1/2 is less than 9.3, the above-described interaction is weakened, and thus the above-described inclined structure is not sufficiently formed.
- the solubility parameter ⁇ (cal / cm 3 ) 1/2 is preferably 9.3 or more, more preferably 10 or more, and 11 or more. Further preferred.
- the gradient structure is formed and fixed (cured) by evaporating the solvent according to the heating conditions (temperature, time, catalyst, curing material, air volume, etc.). It is normal. Therefore, in the point of proceeding curing while forming the above-described inclined structure, the solvent tends to be selected based on the boiling point (evaporation rate) in accordance with the curing temperature. Specifically, a solvent having a boiling point of 30 to 300 ° C., a solvent having a boiling point of 40 to 250 ° C. is more preferable, and a solvent having a boiling point of 50 to 210 ° C. is more preferable. In the case of a mixed solvent containing two or more solvents, the boiling point of the solvent having the highest boiling point contained in these mixed solvents may be in the above range.
- Examples of the solvent having a solubility parameter ⁇ (cal / cm 3 ) 1/2 of 9.3 or more and a boiling point of 50 to 210 ° C. that can be used as the solvent include alcohols, ketones, carboxylic acids, carboxylic acid esters, ethers, Solvents classified into amide, nitrile, water and the like can be mentioned. Specific examples of the solvent include those described in paragraph [0167] of International Publication No. 2014/168122 Pamphlet.
- ⁇ 21.4 (cal / cm 3 ) 1/2 ⁇ water having the highest solubility parameter ⁇ and alcohol are preferable.
- alcohols methanol, ethanol, 1-propanol, 2-methoxyethanol (EGM), 2-ethoxyethanol, 2-methoxypropanol (PGM), 1-butanol, 1-pentanol, 2-methyl-1-butanol Primary alcohols such as 1-pentanol tend to be preferable. These alcohols can be used alone, but are also preferably used by mixing with water.
- the above solvent having a solubility parameter ⁇ (cal / cm 3 ) 1/2 of 9.3 or more contained in the solvent may be used alone or in combination of two or more.
- the solvent is a mixed solvent containing two or more solvents
- at least one of the solvents only needs to satisfy the conditions of the solubility parameter.
- the solubility parameter of the one solvent contained in the mixed solvent satisfies the above conditions
- the hydrophilic sulfonic acid-containing group derived from the copolymer (i) and the one solvent have a constant mutual relationship.
- a hydrophilic sulfonic acid-containing group is brought together with the one solvent on the surface of the applied mixture in contact with the outside air. This is because there is no change in the movement of the copolymer (i) having, and as a result, hydrophilic sulfonic acid-containing groups are concentrated on the surface.
- the solubility parameter ⁇ (cal / cm 3 ) 1/2 of the solvent having the highest boiling point contained in the mixed solvent is preferably 9.3 or more.
- the solubility parameter ⁇ (cal / cm 3 ) 1/2 of the solvent on the high boiling side is higher than that of the solvent on the low boiling point, so that a cured product having a sulfonic acid concentration inclined on the surface is easily formed. ,preferable.
- the weight ratio of the solvent having the highest solubility parameter / the other solvent is preferably 99.9 / 0.1 to 1/99, more preferably 99/1 to The range is 10/90, more preferably 98/2 to 30/70.
- the solvent other than water is a low polarity solvent that separates from water
- the amount of water added is large, or the evaporation rate is higher than necessary.
- a fast (low boiling point) solvent is mixed, in the solvent removal step, the applied composition of the present invention tends to form water droplets, and a cured product having a transparent and smooth surface due to a decrease in leveling property (typically A film made of the cured product may not be obtained. Therefore, when selecting a mixed solvent with water, it is important to first make the composition of the present invention a uniform solution or a uniform dispersion by using a highly polar solvent that is easily compatible with water.
- the weight ratio of water / solvent other than water is 80/20 to 1/99. Is relatively preferable, more preferably 70/30 to 5/95, and still more preferably 60/40 to 10/90.
- Examples of the method for selecting the type of solvent other than water to be mixed with water include a method for selecting a highly polar solvent having a solubility parameter ⁇ (cal / cm 3 ) 1/2 of 9.3 or more.
- the evaporation rate ratio R is calculated by the following simple calculation formulas (A) to (B).
- (A) Evaporation rate saturated vapor pressure at solvent removal temperature (mmHg) ⁇ ⁇ (molecular weight)
- (B) Evaporation rate ratio with respect to water R evaporation rate of solvent other than water / water evaporation rate
- the evaporation rate of water at 50 ° C. is calculated to be 92.6.
- the solvent evaporation rate ratio R is calculated as follows.
- a —SO 3 M group (M represents a hydrogen atom, an alkali metal, an alkaline earth metal, or an ammonium ion) formed on a substrate and N—CH 2 —
- a cured product having an O structure (typically a film made of the cured product), comprising an SO 3 M group concentration (Sa) on the outer surface of the cured product, an interface contacting the substrate, and the outer surface A cured product (typically a film made of the cured product) having a SO 3 M group concentration (Da) ratio (Sa / Da) of 2 to 1000 at an intermediate point
- Sa SO 3 M group concentration
- the gradient ⁇ concentration ratio of sulfonic acid (Sa / Da) ⁇ of the cured product of the present invention is usually in the range of 1.01 to 1000, preferably 1.1 to 1000. It is in the range of 100, more preferably in the range of 1.2 to 60. In any case, the lower limit value is more preferably 1.1 or more.
- the cured product of the present invention has hydrophilicity and N—CH 2 —O structure, so that it is excellent in hardness, wear resistance, weather resistance and the like.
- the cured product of the present invention obtained by curing the composition containing the copolymer (i) and amino resin (ii) thus obtained, for example, a film made of the cured product can be formed.
- curing may mean that, for example, when the solvent is removed from the composition, the solubility in the solvent is typically reduced or lost.
- the group typically contained in the composition typically an epoxy group
- a network (crosslinked structure) mainly composed of oxymethylamino bonds is formed in the cured product.
- Such curing is preferably performed by heating, for example.
- the heating temperature is approximately in the range of 30 to 300 ° C, preferably in the range of 40 to 200 ° C, more preferably in the range of 50 to 180 ° C.
- the heating time is usually in the range of 0.02 hours to 200 hours, preferably 0.1 hours to 8.0 hours, more preferably 0.3 hours to 4.0 hours.
- curing can be done by methods other than heating.
- a known polyfunctional (meth) acrylate, a known polyfunctional epoxy compound, or a known polyfunctional vinyl compound is added to the above composition, and a UV radical polymerization initiator or UV cation is added as necessary. It can be cured by adding a polymerization initiator or the like and irradiating with ultraviolet rays (UV) which is a kind of radiation.
- UV ultraviolet rays
- energy rays having a wavelength range of 0.0001 to 800 nm can be used as radiation.
- the radiation is classified into ⁇ rays, ⁇ rays, ⁇ rays, X rays, electron rays, ultraviolet rays, visible light, microwaves, etc., depending on the composition of the copolymer (i) and amino resin (ii).
- ultraviolet rays are preferable, and the output peak of ultraviolet rays is preferably in the range of 200 to 450 nm, more preferably in the range of 210 to 445 nm, still more preferably in the range of 220 to 430 nm, and particularly preferably in the range of 230 to 400 nm.
- an ultraviolet lamp As the type of the ultraviolet lamp, an electrodeless UV (ultraviolet) lamp with less infrared and high illuminance is preferable to a normal electroded UV (ultraviolet) lamp. Further, when an ultraviolet absorber or HALS is added to the composition containing the copolymer (i) and the amino resin (ii), an ultraviolet lamp having a peak intensity at 240 to 270 nm was used in terms of output characteristics. Tends to be preferable.
- the atmosphere for curing the composition containing the copolymer (i) and the amino resin (ii) may be an inert gas atmosphere such as nitrogen, but is preferably an air atmosphere.
- the humidity of the atmosphere is preferably as low as possible because the surface of the cured product tends to be non-uniform under high humidity, but is preferably in the range of 20 to 70% RH, and in the range of 30 to 60% RH. Is more preferable, and the range of 40 to 60% RH is more preferable.
- the film which is an example of the cured product is a film (Z1) having a thickness exceeding 100 nm (0.1 ⁇ m). Films having such thicknesses are excellent in wear resistance and durability, so that they are used for interior and exterior coatings of buildings and transportation equipment (vehicles, ships and aircraft), interior and exterior coatings of household appliances and electrical appliances, and used in them. It is preferably used as a coating for fixtures and parts. In particular, it is useful for outdoor coating applications that require weather resistance.
- a surface with high hardness can be formed without being affected by the base material (generally a soft substrate).
- the film coated on the material is easily damaged by the influence of the base material).
- This high-hardness surface coating film makes it difficult to be damaged, and even if the surface is somewhat shaved due to stress or the like beyond assumption, the film remains in most cases, so that the film performance is easily maintained (that is, durable) Tend to improve).
- the film (Z1) can be produced, for example, by applying the composition containing the copolymer (i) and the amino resin (ii) to a substrate described later and curing the composition.
- Examples of methods for applying the composition to a substrate include brush coating, spray coating, wire bar, bar coater, blade, roll coating, spin coating, dipping, and other known coating methods. Is mentioned.
- the coating amount may be set so that the thickness of the film (Z1) is in a desired range.
- the thickness of the film (Z1) exceeds 0.001 ⁇ m (1 nm) to 0.1 ⁇ m (100 nm) or less when hardness is not required, and exceeds about 0.1 ⁇ m (100 nm) when hardness is required. It is 500 ⁇ m or less, preferably more than 0.1 ⁇ m and 100 ⁇ m or less, more preferably 1 to 50 ⁇ m.
- the thickness of the film (Z1) tends to be relatively thick, approximately in the range of more than 0.1 ⁇ m (100 nm) to 500 ⁇ m or less, preferably in the range of 1 ⁇ m to 200 ⁇ m, Preferably it is the range of 3 micrometers or more and 100 micrometers or less.
- the layer (Z1) which consists of a film
- a laminate having at least one layer and a substrate can be produced, and the resulting laminate can be used as it is.
- the material used as the base material examples include PMMA, polycarbonate (PC), PET, ABS, triacetyl cellulose (TAC), polyvinyl chloride (vinyl chloride), polyethylene (PE), polypropylene (PP), and polylactic acid (PLA). ), Poly (thio) urethane resin, poly (thio) urea resin, and (thio) epoxy resin and other organic materials, glass, iron, stainless steel, aluminum, nickel, zinc, gold, silver, copper, metal oxides, Composites such as SMC (Sheet Molding Compound) that combines inorganic materials such as ceramics, cement, slate, marble, granite and mortar, inorganic materials such as glass fiber and calcium carbonate, and organic materials such as unsaturated polyester resin Materials and the like.
- PC polycarbonate
- PET ABS
- PV chloride polyvinyl chloride
- PE polyethylene
- PP polypropylene
- PVA polylactic acid
- Such an organic base material, inorganic base material, and composite base material made of such organic material, inorganic material, and composite material can be used as they are, but they may be used after various surface treatments are performed.
- the surface treatment for example, the adhesion between the substrate and the layer made of the film (Z) can be enhanced.
- the substrate subjected to such surface treatment include a substrate obtained by metal plating the substrate surface, a substrate obtained by chemically treating the substrate surface with a chemical such as a zinc phosphate aqueous solution, and a corona treatment.
- Examples of the coating agent used for the primer treatment, undercoat treatment, or anchor coat treatment include polyester resins, polyamide resins, polyurethane resins, epoxy resins, phenol resins, (meth) acrylic resins, polyvinyl acetate.
- a coating agent containing a resin such as a polyethylene resin, a polyolefin resin such as polyethylene and polypropylene, or a copolymer or modified resin thereof, a cellulose resin, or the like as a main component of the vehicle can be used.
- the coating agent may be either a solvent type coating agent or an aqueous type coating agent.
- silane coupling agent coating agents include silane coupling agent coating agents, mixed coating agents of silane coupling agents and fillers, modified polyolefin coating agents, ethyl vinyl alcohol coating agents, polyethyleneimine coating agents, polybutadiene coating agents.
- Agent polyurethane coating agent; polyester polyurethane emulsion coating agent, polyvinyl chloride emulsion coating agent, urethane acrylic emulsion coating agent, silicon acrylic emulsion coating agent, vinyl acetate acrylic emulsion coating agent, acrylic emulsion coating agent; Styrene-butadiene copolymer latex coating agent, acrylonitrile-butadiene copolymer latex coating agent, methyl methacrylate-butadiene copolymer latex coating agent, chloroprene latex coating agent, polybutadiene rubber rubber latex coating agent, poly An acrylic ester latex coating agent, a polyvinylidene chloride latex coating agent, a polybutadiene latex coating agent
- These coating agents can be applied, for example, by the gravure coating method, reverse roll coating method, knife coating method, kiss coating method, etc. 0.005 g / m 2 to 5 g / m 2 .
- the silane coupling agent-based coating agent the mixed coating agent of the silane coupling agent and the filler, and the trade names “Takelac TM ” and “Takenate TM ” (both made by Mitsui Chemicals)
- a typical polyurethane-based coating agent is more preferable.
- silane coupling agent-based coating agent and the mixed coating agent of the silane coupling agent and the filler are characterized in that the coating agent contains a silane coupling agent.
- Typical silane coupling agents contained in the coating agent include, for example, vinyltrichlorosilane, vinyltrimethoxysilane, vinyltriethoxysilane, 2- (3,4-epoxycyclohexyl) ethyltrimethoxysilane, and 3-glycidyloxy.
- silane coupling agents a silane coupling agent having an epoxy group, a mercapto group, or an amino group is relatively preferred, a silane coupling agent having an amino group is more preferred, and bis (trimethoxysilylpropyl) is further preferred.
- Amine, bis (triethoxysilylpropyl) amine, N, N′-bis (trimethoxysilylpropyl) ethylenediamine, and N, N′-bis (triethoxysilylpropyl) ethylenediamine are most preferred.
- the polyurethane-based coating agent has a urethane bond in the main chain or side chain of the resin contained in the coating agent.
- a polyurethane-type coating agent is a coating agent containing the polyurethane obtained by making polyol and isocyanate compounds, such as polyester polyol, polyether polyol, or acrylic polyol, react, for example.
- polyester polyols such as condensation polyester polyols and lactone polyester polyols are mixed with isocyanate compounds such as tolylene diisocyanate, hexamethylene diisocyanate, isophorone diisocyanate, norbornane diisocyanate methyl, and xylene diisocyanate.
- isocyanate compounds such as tolylene diisocyanate, hexamethylene diisocyanate, isophorone diisocyanate, norbornane diisocyanate methyl, and xylene diisocyanate.
- the method of mixing the polyol compound and the isocyanate compound is not particularly limited.
- the mixing ratio is not particularly limited, but if the isocyanate compound is too small, it may cause curing failure, so that the OH group of the polyol compound and the NCO group of the isocyanate compound are in the range of 2/1 to 1/40 in terms of equivalents. Is preferred.
- the film thickness of the coating agent used for the primer treatment, undercoat treatment, or anchor coat treatment can be arbitrarily determined according to the application.
- it in the case of coating an optical system substrate that often requires antireflection properties, it is in the range of approximately 0.0001 ⁇ m (0.1 nm) to 0.1 ⁇ m (100 nm), and 0.001 ⁇ m (1 nm). It is more preferable if it is in the range of -0.05 ⁇ m (50 nm).
- it is approximately in the range of 0.1 to 400 ⁇ m, more preferably in the range of 0.5 to 200 ⁇ m, and still more preferably in the range of 1 to 100 ⁇ m.
- the surface treatment can be performed for the purpose other than the improvement of adhesion, for example, for the purpose of imparting antireflection properties.
- a glazing group having fine irregularities formed on the surface is used as the substrate subjected to such a surface treatment.
- coated the coating material to the surface of these base materials and formed the coating film can also be used.
- the above substrate can be used alone as a substrate, but can also be used as a laminate substrate in which a plurality of substrates selected from an organic substrate, an inorganic substrate, and a composite substrate are laminated.
- optical system substrate used for optical articles and optical devices represented by lenses, glasses, cameras, display devices (displays), projection devices, etc.
- transparent A transparent substrate made of a material having a property can also be used.
- the laminate having the base material and the layer (Z1) may be provided with various functional layers.
- Examples of such a functional layer include a hard coat layer and an antireflection (AR) layer.
- AR antireflection
- Examples of the hard coat layer include a layer formed from an acrylic material and a layer formed from a silica material.
- antireflection layer examples include a layer formed of a low refractive index material, a multilayer antireflection layer in which layers of a low refractive index material and layers of a high refractive index material are alternately stacked.
- the functional layer may be provided outside the base material and the layer (Z1), but may be provided between the base material and the layer (Z1), for example, on the base material.
- the hard coat layer and the antireflection layer are preferably provided between the base material layer and the layer (Z1), for example, provided on the base material.
- the laminate of the present invention may have a plurality of the above functional layers.
- each layer of the laminate is made as thin as possible.
- the functional layer can be provided in the laminate by a known method.
- the layer in contact with the layer comprising the film (Z1) of the present invention (Z1) is at it is one preferred embodiment of stacking such that a layer composed mainly of SiO 2 is there.
- the structural unit a and formula (2) represented by the formula (1) of the copolymer (i) As for the ratio of the structural unit b represented by the formula (3) and the structural unit c represented by the formula (3), the larger the structural unit a represented by the formula (1), the higher the hydrophilicity.
- the structural unit a represented by the formula (1) is in the range of 99.0 to 60.0
- the structural unit b represented by the formula (2) is in the range of 0.5 to 20.0
- the formula ( The structural unit c represented by 3) is preferably in the range of 0.5 to 20.0
- a is in the range of 98.0 to 70.0
- b is in the range of 1.0 to 15.0
- the ratio (i) / (ii) of the weight of the copolymer (i) and the converted weight of the amino resin (ii) should be higher for the same reason as described above. It tends to be favorable.
- the weight ratio is preferably in the range of 99/1 to 40/60, more preferably in the range of 95/5 to 60/40.
- the film (Z1) is an antifogging film, an antifouling film, or an antistatic film
- the substrate is coated with the antifogging film, the antifouling film, or the antistatic film, for example.
- a laminated body is obtained.
- the substrate is a film
- a pressure-sensitive adhesive layer described later can be provided on the surface on which the film of the present invention is not formed, and a release film can be provided on the surface of the pressure-sensitive adhesive layer.
- the adhesive layer is laminated on the other side of the base film
- the laminated film having the film of the present invention is used as an antifogging film and an antifouling film, and glass, a mirror such as a bathroom, a display material surface of a display, a television, etc. It can be easily attached to signboards, advertisements, information boards such as information boards, signs for railways, roads, etc., outer walls of buildings, window glass and the like.
- a well-known adhesive can be used.
- the adhesive include acrylic adhesives, rubber adhesives, vinyl ether polymer adhesives, and silicone adhesives.
- the thickness of the adhesive layer is usually in the range of 2 to 50 ⁇ m, preferably in the range of 5 to 30 ⁇ m.
- the surface of the film in contact with the outside air may be covered with a coating material.
- the film covered with the covering material and the laminate having the film can prevent the film from being damaged or soiled during transportation, storage, display, or the like.
- the coating material when the coating material is applied with a composition containing the copolymer (i) and the amino resin (ii) and is heated or irradiated with infrared rays as described above, the surface of the coating material is removed from dust and contaminants in the atmosphere.
- the coating material can be cured by heating or irradiating with an infrared ray while keeping the coating material in close contact with the coated product, and the product of the present invention can be produced with the film and coating material of the present invention laminated on a substrate or the like. .
- the laminated body which prevented the damage
- Examples of the material preferably used as the covering material include, for example, polyvinyl alcohol (PVA), vinyl alcohol polymers such as ethylene / vinyl alcohol copolymer, polyacrylamide, polyisopropylacrylamide, and polyacrylonitrile. , Polycarbonate (PC), polymethyl methacrylate (PMMA), polyethylene terephthalate (PET), polystyrene (PS), biaxially oriented polypropylene (OPP), and the like.
- PVA polyvinyl alcohol
- PVD vinyl alcohol polymers
- PMMA polymethyl methacrylate
- PET polyethylene terephthalate
- PS polystyrene
- OPP biaxially oriented polypropylene
- the laminate of the present invention can be made into laminates of various forms by devising the shape of the substrate.
- membrane (Z1) and laminated body obtained by this invention can be used with forms, such as a film, a sheet
- the film (Z1) can also be used as a primer layer.
- composition containing the copolymer (i) and the amino resin (ii) can be obtained as a cured product having various shapes, such as a film or a molded body, by curing in a mold having various shapes. it can.
- the film (Z1) obtained by the present invention is excellent in hydrophilicity, durability, abrasion resistance, and weather resistance, and has high antifogging properties, antifouling properties, antistatic properties, and quick drying properties (water evaporation).
- the water contact angle of the membrane (Z1) obtained by the present invention is usually 30 ° or less, preferably 20 ° or less, more preferably 10 ° or less.
- a film having a water contact angle of not more than the above upper limit has high hydrophilicity and is easy to blend (wet) with water and is excellent as a hydrophilic material.
- the cured product of the present invention obtained by curing the composition containing the copolymer (i) and the amino resin (ii), for example, the film (Z1) comprising the cured product is preferably immersed in water at 25 ° C.
- the change in the water contact angle before and after the ultrasonic treatment for 10 minutes is usually within 20 °, preferably within 10 °, and more preferably within 5 °.
- the composition containing the copolymer (i) and amino resin (ii) used in the present invention which is water-soluble or very familiar with water, has sufficiently advanced the reaction of the groups contained in the composition.
- a cured product having such characteristics can be produced by curing the composition by heating or the like as described above.
- the film (Z1) obtained according to the present invention is, for example, an antifogging material, an antifogging coating (hereinafter also referred to as an antifogging coat), an antifouling material, an antifouling coating or a self-cleaning coat, an antistatic material, and a quick-drying material. It is also useful for quick-drying coatings and antistatic coatings or dust adhesion prevention coatings.
- the film of the present invention when used as an antifogging coat, water droplets spread on the film surface and a water film can be formed, so that the antifogging effect is excellent. It is excellent in antifouling effect because it can be removed by floating the dirt. Further, the film of the present invention is excellent in antistatic properties, and is useful for antistatic materials, antistatic coatings, and dust adhesion preventing coatings.
- the laminate obtained by the present invention is also excellent in hydrophilicity and durability, and is useful as an antifogging material, an antifouling material, an antistatic material and the like.
- the laminate obtained by laminating the film of the present invention on a substrate made of a transparent material such as transparent resin and glass has transparency, hydrophilicity, antifogging property, antifouling property, and antistatic property. It can be used as a laminate having excellent properties, quick drying properties, anti-condensation properties, weather resistance, and abrasion resistance.
- Articles for equipment Building materials such as exterior wall products, interior wall products, floors, furniture, bathroom items, toilets, kitchen items such as sinks, ventilators and range peripherals, toilet items, piping items, etc.
- the structure of the copolymer (i) was evaluated as follows.
- the unit ratio (1) / (2) / (3) of the unit (1) having a sulfonic acid-containing group, the unit (2) having an epoxy group, and the unit (3) having a trialkoxysilyl group is 13 C -Analyzed by NMR. The measurement conditions are described below.
- Mw Weight average molecular weight
- Mw weight average molecular weight
- Mn number average molecular weight
- the physical properties of the coating film were evaluated as follows.
- ⁇ 120 ° C heated dirt adhesion test> The sample was kept in a 120 ° C. hot air dryer for 12 hours, cooled to room temperature, scrubbed with Bencot (Asahi Kasei) under running water, and dried with an air gun to obtain a sample for evaluation. Evaluation was performed by measuring the water contact angle before and after the test, and it was judged that the larger the increase (gap) of the water contact angle, the more contaminated.
- ⁇ Adhesion test> The test was conducted according to JIS K5600-5-6 (adhesion-cross-cut method). In addition, evaluation was represented by the number of the squares which did not peel (converted) by converting 25 squares into 100 squares.
- the sample formed by forming the coat layer 20 on the base material 10 is cut obliquely, and the outer surface is measured using a time-of-flight secondary ion mass spectrometer (TOF-SIMS).
- TOF-SIMS time-of-flight secondary ion mass spectrometer
- Da sulfonic acid concentration
- the slope (Sa / Da) represented by the sulfone concentration ratio at the midpoint between the surface and the outer surface was determined.
- the coat layer 20 is the film of the present invention.
- the sulfonic acid concentration was measured using a time-of-flight secondary ion mass spectrometer (TOF-SIMS) at a half point, the inner surface of the coat layer in contact with the substrate 10. Evaluation Evaluation was performed using the following formula. The relative intensity (relative to the total detected ions) was used as the ion concentration at each measurement point.
- TOF-SIMS time-of-flight secondary ion mass spectrometer
- Inclination degree Sa / Da sulfonic acid concentration at coat layer surface 40 / sulfonic acid concentration at point 1/2 of coating layer 20 ⁇ Measurement of film thickness> (Measurement equipment and conditions)
- Apparatus Field emission transmission electron microscope (FE-TEM): JEM-2200FS (manufactured by JEOL) Accelerating voltage: 200 kV FIB (Focused Ion Beam System, Focused Ion Beam System) processing equipment: SMI2050 (manufactured by Seiko Instruments Inc.) Sample preparation, etc.
- FE-TEM Field emission transmission electron microscope
- JEM-2200FS manufactured by JEOL
- Accelerating voltage 200 kV FIB (Focused Ion Beam System, Focused Ion Beam System) processing equipment: SMI2050 (manufactured by Seiko Instruments Inc.) Sample preparation, etc.
- GMA glycidyl methacrylate
- KBE-503 methacryloyloxypropyltriethoxysilane
- a mixed liquid of 8 g and a mixed liquid of 1.15 g of t-butylperoxy-2-ethylhexanoate (hereinafter abbreviated as perbutyl-O) as a polymerization initiator and 11.5 g of methanol were prepared. Each of these was charged into a reaction flask in which the resulting neutralized mixture was heated to reflux (internal temperature: 66 ° C.) in one-third portions over 2 hours. Polymerization was carried out for 8 hours under the conditions.
- Example 1 ⁇ Preparation of formulation 1 coating solution 1> Each component was mixed according to the mixing order shown in Table 2 below, and finally a coating solution 1 was prepared through a filter having an average pore size of 0.5 ⁇ m.
- the obtained coated substrate was pre-dried in an oven at 50 ° C. for 1 minute and then heated in an oven at 120 ° C. for 1 hour to obtain a primer-treated substrate formed from a silane coupling agent having a thickness of 5 nm on the substrate. It was.
- the coating liquid 1 On the primer layer of the primer-treated substrate, the coating liquid 1 is applied with a bar coater # 30, pre-dried in an oven at 50 ° C. for 1 minute, heated at 120 ° C. for 2 hours, and on the primer layer, A coating film having a thickness of 3 ⁇ m was formed.
- a laminate in which a primer layer and a coating film (total thickness of 3.005 ⁇ m) were formed on a base material (glass plate) was obtained.
- the laminate is cooled to room temperature, and the coating film is scrubbed with Bencot M-3 II (Asahi Kasei Fibers Co., Ltd.) under running water, and the film surface is dried with an air gun.
- Bencot M-3 II Asahi Kasei Fibers Co., Ltd.
- the physical properties were evaluated (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property, adhesion, Taber abrasion property). The results are listed in Table 3.
- the coating solution 1 was prepared, the primer composition was prepared, the primer layer was formed, and the coating film was formed in the same manner as in Example 1 except that the copolymer of Synthesis Example 1 was changed to the copolymer of Synthesis Example 2.
- a laminate having a primer layer and a coating film formed on a substrate (glass plate) was obtained.
- the physical properties of the obtained coating film were evaluated in the same manner as in Example 1 according to the method described above. The results are listed in Table 3.
- Example 2 Preparation of coating solution 1, preparation of primer composition, formation of primer layer, formation of coating film, as in Example 1, except that the substrate for forming the primer layer is a polycarbonate plate (hereinafter abbreviated as PC plate).
- PC plate polycarbonate plate
- this neutralized mixture was heated to reflux (internal temperature 63 ° C.), and a mixed solution of 5.14 g (0.036 mol) of GMA and 0.13 g of perbutyl-O as a polymerization initiator was charged. The mixture was stirred for 4.5 hours for polymerization.
- a solution was prepared by mixing 5.8 g of the produced copolymer (CH110901 polymer) with 40 g of water, and under stirring conditions, this solution was mixed with 35 g of 2-methoxyethanol (hereinafter abbreviated as EGM), tetraethoxysilane ( Hereinafter abbreviated as TEOS.) 20.1 g and 6 g of 5 wt% sulfuric acid water were mixed.
- EGM 2-methoxyethanol
- TEOS. tetraethoxysilane
- 20.1 g and 6 g of 5 wt% sulfuric acid water were mixed.
- the obtained mixed solution was passed through a filter having an average pore size of 0.5 ⁇ m, and 106.9 g of a colorless transparent coating composition 1 having a solid content (total amount of copolymer and SiO 2 converted TEOS) of 11 wt% was obtained. Obtained.
- the polymer / TEOS (SiO 2 equivalent) weight ratio in this composition
- Example 3 (Coating test) In the same manner as in Example 1, except that the coating solution 1 was applied to the substrate by the bar coater # 30, and the coating composition 1 was applied to the substrate by the bar coater # 18. Preparation, formation of a primer layer, and formation of a coating film were performed to obtain a laminate in which the primer layer and the coating film were formed on a substrate (glass plate). The physical properties of the obtained coating film were evaluated in the same manner as in Example 1 according to the method described above. The results are listed in Table 3.
- Example 3 Each component was mixed according to the mixing order of the following Table 4 (formulation 2), and finally the coating solution 2 was prepared through a filter having an average pore size of 0.5 ⁇ m.
- Physical property evaluation (appearance, water contact angle, haze, antifouling property, adhesion, pencil hardness, Taber abrasion property) of the obtained coating film was performed according to the method described above. The results are listed in Table 5. Further, the inclination of the coating film was measured by TOF-SIMS. The results are listed in Table 6.
- the coating solution 2 was prepared, the primer composition was prepared, the primer layer was formed, and the coating film was formed in the same manner as in Example 3 except that the copolymer of Synthesis Example 1 was changed to the copolymer of Synthesis Example 2.
- a laminate having a primer layer and a coating film formed on a substrate (glass plate) was obtained.
- the physical properties of the obtained coating film were evaluated in the same manner as in Example 3 according to the method described above. The results are listed in Table 5.
- Example 5 (Coating test) In the same manner as in Example 5, except that the coating solution 2 was applied to the substrate by the bar coater # 30, and the coating composition 2 was applied to the substrate by the bar coater # 18. Preparation, formation of a primer layer, and formation of a coating film were performed to obtain a laminate in which the primer layer and the coating film were formed on a substrate (glass plate). The physical properties of the obtained coating film were evaluated in the same manner as in Example 5 according to the method described above. The results are listed in Table 5.
- ATBS acrylamide-t-butylsulfonic acid
- the crystallized copolymer is filtered, and the obtained filter cake is washed with 500 ml of methanol, and the taken-up filter cake is reduced under reduced pressure (less than 100 mmHg) until no change in weight is observed at 50 ° C.
- the product was sufficiently dried to obtain 53.3 g (yield 50%) of a white copolymer.
- the crystallized copolymer is filtered, and the obtained filter cake is washed with 500 ml of methanol, and the taken-up filter cake is reduced under reduced pressure (less than 100 mmHg) until no change in weight is observed at 50 ° C. After sufficiently drying, 53.2 g (yield 41%) of a white copolymer was obtained.
- Example 4 Preparation of coating solution 1, preparation of primer composition, primer layer as in Example 1, except that the copolymer of Synthesis Example 1 is changed to the copolymer described in the section of copolymer type in Table 8 below. And a coating film were formed to obtain a laminate in which a primer layer and a coating film were formed on a substrate (glass plate). The physical properties of the resulting coating film were evaluated (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property, adhesion) according to the above-described methods. The results are listed in Table 8. Table 8 shows the results of Example 1 again.
- Example 5 Preparation of coating solution 1, preparation of primer composition, primer layer, as in Example 2, except that the copolymer of Synthesis Example 1 is changed to the copolymer described in the section of copolymer type in Table 8 below. And a coating film were formed to obtain a laminate in which a primer layer and a coating film were formed on a substrate (PC plate). The physical properties of the resulting coating film were evaluated (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property, adhesion) according to the above-described methods. The results are listed in Table 8. Table 8 shows the results of Example 2 again.
- the crystallized copolymer is filtered, and the obtained filter cake is washed with 700 ml of methanol, and then the taken-up filter cake is reduced under reduced pressure (less than 100 mmHg) until no change in weight is observed at 50 ° C. After sufficiently drying, 69.2 g (yield 52%) of a white copolymer was obtained.
- Example 6 and 7 Preparation of coating solution 1, preparation of primer composition, primer layer, as in Example 1, except that the copolymer of Synthesis Example 1 is changed to the copolymer described in the section of copolymer type in Table 10 below. And a coating film were formed to obtain a laminate in which a primer layer and a coating film were formed on a substrate (glass plate). The physical properties of the resulting coating film (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property) were evaluated according to the above-described methods. The results are listed in Table 10. Table 10 shows the results of Example 1 again.
- Example 8 and 9 Preparation of coating solution 2, preparation of primer composition, primer layer, as in Example 3, except that the copolymer of Synthesis Example 1 is changed to the copolymer described in the section of copolymer type in Table 10 below. And a coating film were formed to obtain a laminate in which a primer layer and a coating film were formed on a substrate (glass plate). The physical properties of the resulting coating film (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property) were evaluated according to the above-described methods. The results are listed in Table 10. Table 10 shows the results of Example 3 again.
- Example 10 to 16 The coating solution 1 was prepared in the same manner as in Example 1, except that the amount of DS-Na (surfactant) contained in the coating solution 1 was changed to the amount shown in Table 13, the preparation of the primer composition, the primer layer Formation of the coating film was performed to obtain a laminate in which the primer layer and the coating film were formed on the substrate (glass plate). The physical properties of the resulting coating film were evaluated (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property). The results are listed in Table 13.
- Example 1 except that the amount of the copolymer of Synthesis Example 1 contained in the coating solution 1, the amount of 30 wt% methanol silica sol, and the amount of 60 wt% methylated melamine resin aqueous solution were changed to the amounts shown in Tables 14 and 15.
- Preparation of coating solution 1 preparation of primer composition, formation of primer layer and formation of coating film were carried out in the same manner as above to obtain a laminate in which the primer layer and the coating film were formed on the substrate (glass plate).
- the physical properties of the resulting coating film were evaluated (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifouling property). The results are listed in Tables 14 and 15.
- reaction solution was cooled to room temperature, and then 189.0 g (1.05 mol) of 30 wt% sodium methoxide (methanol solution) was added dropwise at an internal temperature of 30 ° C. or lower in a water bath. Neutralized.
- the precipitated copolymer (methylated aniline resin) is filtered, and the filter cake is washed with 1000 ml of methanol and then sufficiently dried under reduced pressure (less than 100 mmHg) at 25-30 ° C. until no weight change is observed (2 days ) To obtain 110 g of an orange copolymer.
- Example 43 The coating solution was the same as in Example 1 except that the methylated melamine resin contained in the coating solution 1 was changed to the amino resin described in Table 16 (Example 43: methylated urea resin, Example 44: methylated aniline resin).
- Preparation of No. 1 preparation of a primer composition, formation of a primer layer, and formation of a coating film were performed to obtain a laminate in which the primer layer and the coating film were formed on a substrate (glass plate).
- the physical properties of the resulting coating film were evaluated (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property, adhesion). The results are listed in Table 16. In Table 16 below, the results of Example 1 are listed again.
- the obtained polymerization solution was concentrated under reduced pressure using a rotary evaporator, and 400 g of isopropanol and 400 g of cyclohexane were added to the resulting residue and mixed vigorously.
- the precipitated polymer was filtered off, and the obtained filter cake was washed with ethanol, and the taken-out filter cake was sufficiently dried under reduced pressure (less than 100 mmHg) at 50 ° C. until no weight change was observed. 52.5 g (yield 91%) of copolymer was obtained.
- Example 45 The coating solution 1 was prepared, the primer composition was prepared, the primer layer was formed, and the coating film was formed in the same manner as in Example 1 except that the copolymer of Synthesis Example 1 was changed to the copolymer of Synthesis Example 12.
- a laminate having a primer layer and a coating film formed on a substrate (glass plate) was obtained.
- the physical properties of the resulting coating film were evaluated (appearance, 120 ° C. heat stain adhesion test, pencil hardness, antifogging property, antifouling property, adhesion). The results are listed in Table 17.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Ceramic Engineering (AREA)
- Materials Engineering (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Structural Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Paints Or Removers (AREA)
Abstract
Description
A1は、単結合、炭素数1~10である2価の炭化水素基、下記式(1-1)で表される基、または下記式(1-2)で表される基を示し、
A2は、単結合、炭素数1~10である2価の炭化水素基、下記式(2-1)で表される基、または下記式(2-2)で表される基を示し、
A3は、単結合、炭素数1~10である2価の炭化水素基、下記式(3-1)で表される基、または下記式(3-2)で表される基を示し、
R1、R2、およびR3は、それぞれ独立して、水素原子またはメチル基を示し、
R4は、水素原子、メチル基、エチル基、プロピル基、またはブチル基を示し、R4は互いに同一でも異なっていてもよく、
R10は、水素原子、メチル基、エチル基、プロピル基、ブチル基、メトキシ基、エトキシ基、プロポキシ基、またはブトキシ基を示し、
Mは、水素原子、アルカリ金属イオン、1/2価のアルカリ土類金属イオン、アンモニウムイオン、またはアミンイオンを示し;
下記式(1-1)、(1-2)、(2-1)、(2-2)、(3-1)、および(3-2)において、nおよびn2は、それぞれ独立して、1~10の整数であり、n1は0~10の整数であり、mは1~6の整数であり、m1は0~6の整数であり、lは0~4の整数であり、R5およびR6は、それぞれ独立して、水素原子またはメチル基を示し、*はSO3Mと結合する側の端部、**はエポキシ基と結合する側の端部、***はSi原子と結合する側の端部を示す。)
nは1~10の整数であり、n1は0~10の整数であり、
R1、R2、R3、R5、およびR6は、それぞれ独立して、水素原子またはメチル基を示し、
R4は、水素原子、メチル基、エチル基、プロピル基、またはブチル基を示し、R4は互いに同一でも異なっていてもよく、
R10は、水素原子、メチル基、エチル基、プロピル基、ブチル基、メトキシ基、エトキシ基、プロポキシ基、またはブトキシ基を示し、
Mは、水素原子、アルカリ金属イオン、1/2価のアルカリ土類金属イオン、アンモニウムイオン、またはアミンイオンを示す。)
〔3〕共重合体(i)のゲルパーミエーションクロマトグラフィー(GPC)により測定した標準ポリメチルメタクリレート換算の重量平均分子量が500~3,000,000である上記組成物から得られる〔1〕~〔2〕のいずれかに記載の硬化物。
上記共重合体(i)のGPCにより測定した重量平均分子量が500~3,000,000である組成物から得られる〔1〕または〔2〕に記載の硬化物。
下記式(8)において、q030は0~30の整数であり、q030は互いに同一でも異なっていてもよく、R30およびR40は式(7)中の定義と同一であり、
下記式(9)において、q050は0~50の整数であり、Xは酸素原子または硫黄原子を示し、R30およびR40は式(7)中の定義と同一であり、
下記式(10)において、q050は0~50の整数である。)
(メタ)アクリロイルオキシ基を有するエポキシ化合物としては、下記式(5’)で表される化合物が好ましい。
エポキシ基同士の反応は一般式(11)で表され、加熱して反応させることが好ましい。加熱温度は、おおよそ30~250℃の範囲であり、好ましくは30~200℃の範囲、さらに好ましくは30~150℃の範囲である。このエポキシ基同士の反応は、酸などのカチオンおよび塩基などのアニオンに代表される触媒の存在によって、反応が加速される傾向にある。
エポキシ基とアルコキシシリル基の反応は、一般式(12)および一般式(14)で表される。
アルコキシシリル基同士の反応式は、一般式(13)で表され、加熱して反応させることが好ましい。加熱温度は、おおよそ30~250℃の範囲であり、好ましくは30~200℃の範囲、さらに好ましくは30~180℃の範囲である。
ヒドロキシメチル基とエポキシ基との反応は一般式(16)で表される。本反応は進行が遅く、アレニウス酸またはルイス酸性の化合物等を触媒として添加するとこの反応が促進される傾向にある。反応温度は、おおよそ30~300℃の範囲であり、好ましくは50~250℃の範囲、さらに好ましくは80~180℃の範囲である。
アルコキシメチル基とエポキシ基との反応は一般式(17)で表されるが、上記のヒドロキシメチル基とエポキシ基との反応(16)よりも進行しにくい傾向にあるため、通常はエポキシ基がシラノール等の活性水素基と開環反応して生成した2級ヒドロキシ基と、縮合反応が起こる。このアルコキシメチル基と2級ヒドロキシ基との反応も一般式(16)の反応と同様に遅く、アレニウス酸またはルイス酸性の化合物等を触媒として添加するとこの反応が促進される傾向にある。反応温度は、おおよそ30~300℃の範囲であり、好ましくは50~250℃の範囲、さらに好ましくは80~180℃の範囲である。
ヒドロキシメチル基と共重合体(i)由来のアルコキシシリル基が加水分解されて生成したシラノール基との反応が一般式(17)で表される。本反応は一般式(17)で表される2級アルコールとの反応よりも早い傾向にある。反応速度の向上を目的として、アレニウス酸またはルイス酸性の化合物等を触媒として添加する場合もある。反応温度は、おおよそ20~300℃の範囲であり、好ましくは40~250℃の範囲、さらに好ましくは80~180℃の範囲である。
アルコキシメチル基と共重合体(i)由来のアルコキシシリル基が加水分解されて生成したシラノール基との反応は一般式(19)で表される。本反応も一般式(18)で表される反応と同様に、一般式(17)で表される2級アルコールとの反応よりも早い傾向にある。反応速度の向上を目的として、アレニウス酸またはルイス酸性の化合物等を触媒として添加する場合もある。反応温度は、おおよそ20~300℃の範囲であり、好ましくは40~250℃の範囲、さらに好ましくは80~180℃の範囲である。
(1)1mol当たりの蒸発潜熱 Hb=21×(273+Tb)〔単位:cal/mol〕,Tb:溶媒の沸点(℃)
(2)25℃での1mol当たりの蒸発潜熱 H25=Hb×{1+0.175×(Tb-25)/100}〔単位:cal/mol〕,Tb:溶媒の沸点(℃)
(3)分子間結合エネルギー E=H25-596〔単位:cal/mol〕
(4)溶媒1ml(cm3)当たりの分子間結合エネルギー E1=E×D/Mw〔単位:cal/cm3〕,D:密度(g/cm3),Mw:溶媒の分子量
(5)SP値: 溶解パラメーター σ=(E1)1/2 〔単位:(cal/cm3)1/2〕
このようなSP値(溶解パラメーターσ)(cal/cm3)1/2が9.3以上の溶媒を用いることにより、共重合体(i)に由来する親水性のスルホン酸含有基と一定の相互作用を有するため、この混合物を基材に塗布して、その混合物から溶媒を除去する際に、塗布された混合物の外気に接する表面に溶媒と同伴して親水性のスルホン酸含有基を有する共重合体(i)が移動して、その表面に親水性のスルホン酸含有基が濃縮されることになり、本発明で得られる硬化物(典型的には膜)の外表面に親水基のスルホン酸含有基が集中した傾斜構造が形成される。
(A) 蒸発速度=溶媒除去温度での飽和蒸気圧(mmHg)×√(分子量)
(B) 水に対する蒸発速度比R=水以外の溶媒の蒸発速度/水の蒸発速度
例えば、50℃における水の蒸発速度は92.6と計算され、50℃で溶媒除去を行う場合の代表的な溶媒の蒸発速度比Rは、およそ以下の通り計算される。
スチレン-ブタジエン共重合体ラテックスコート剤、アクリルニトリル-ブタジエン共重合体ラテックスコート剤、メチルメタアクリレート-ブタジエン共重合体ラテックスコート剤、クロロプレンラテックスコート剤、ポリブタジェンラテックスのゴム系ラテックスコート剤、ポリアクリル酸エステルラテックスコート剤、ポリ塩化ビニリデンラテックスコート剤、ポリブタジエンラテックスコート剤、あるいはこれらラテックスコート剤に含まれる樹脂のカルボン酸変性物ラテックスもしくはディスパージョンからなるコート剤が好ましい。
スルホン酸含有基を有するユニット(1)、エポキシ基を有するユニット(2)のユニット、およびトリアルコキシシリル基を有するユニット(3)のユニット比(1)/(2)/(3)は13C-NMRにより分析した。測定条件を以下に記載する。
* 装置: ブルカー・バイオスピン製 AVANCEIII cryo-500型核磁気共鳴装置
* 測定核: 13C(125MHz)
* 測定モード: シングルパルスプロトンブロードバンドデカップリング
* パルス幅: 45°(5.0μ秒)
* ポイント数: 64K
* 測定範囲: -25~225ppm
* 積算回数: 1000回
* 測定溶媒: D2O
* 測定温度: 室温
* 試料濃度: 40mg/0.6ml-D2O
(ユニット比(1)/(2)/(3)の解析)
下記式(200)のf炭素のピーク(57~59ppm付近)、下記式(300)のk炭素のピーク(51~52ppm付近)、および下記式(400)のt炭素のピーク(4~6ppm付近)の積分強度比で算出した。
即ち、ユニット比(1)/(2)/(3)=f炭素ピークの積分強度/k炭素ピークの積分強度/t炭素ピークの積分強度とした。
Mw(重量平均分子量)、および重量平均分子量(Mw)と数平均分子量(Mn)の比 分子量分布 Mw/MnはGPCにより分析した。測定条件を以下に記載する。
* 装置: 日本分光(株) GPC-900
* カラム: 昭和電工(株) Shodex Asahipac「GF-7M HQ」,Φ7.5mm×300mm
* 測定温度: 40℃
* 溶離液: 水/メタノール/NaHPO4/NaHPO4・2H2O=850.0/150.0/2.7/7.3(重量比)
* 流速: 0.5ml/min.
*分子量校正: 分子量既知のポリメチルメタクリレートにより行った。
得られたコーティング膜の透明性を目視により確認した。
協和界面科学社製の水接触角測定装置CA-V型を用いて、1サンプルについて3箇所測定し、これら値の平均値を水接触角の値とした。
日本電色工業社製のヘーズメーターNDH2000を用いて、1サンプルについて4箇所測定し、これら値の平均値をヘーズの値とした。
120℃温風乾燥機中に12時間保持し、室温まで冷却後、流水下においてベンコット(旭化成)で表面を擦り洗い、エアガンで乾燥して評価用サンプルとした。評価は試験前後の水接触角を測定することで行い、水接触角の上昇幅(ギャップ)が大きいほど汚染された、と判断した。
JIS K5600-5-4: 引っかき硬度(鉛筆法)に準じて試験を行った。
呼気により曇らなかった場合を〇、僅かに曇った場合を△、曇った場合を×とした。
ゼブラ(株)製の油性マーカー「マッキー極細」(黒,品番MO-120-MC-BK)でマークし、その上に水滴を垂らして30秒間放置し、ティッシュペーパーでふき取った。マークがふき取れた場合を〇、僅かに残った場合を△、ふき取れずに残った場合を×とした。
JIS K5600-5-6(付着性-クロスカット法)に準じて試験を行った。なお、評価は25マスを100マスに換算して剥離しなかった(密着していた)マスの数で表した。
試験前のサンプルについて、上記方法によりヘーズの測定を行った。その後、JIS K 7204に準拠して、以下の条件によりテーバー摩耗試験を行い、テーバー摩耗試験後のサンプルについてもヘーズの測定を行った。ヘーズの上昇幅が大きいほど耐摩耗性が低い(摩耗しやすい)と判断した。
(テーバー摩耗試験条件)
測定機器: ロータリーアブレージョンテスター ,(株)東洋精機製作所
磨耗輪: C180 OXF
荷重: 500g(250g+250g)×2
<傾斜度の測定>
図2に示す試料調製の通り、基材10の上にコート層20を形成してなるサンプルを斜めに切断し、飛行時間型2次イオン質量分析装置(TOF-SIMS)を用いて、外表面におけるスルホン酸濃度(Sa)と、基材10に接する界面と前記外表面との中間地点におけるスルホン酸濃度(Da)とを測定し、その値から外気に接する膜の外表面と膜の内表面と外表面との中間地点のスルホン濃度比で表される傾斜度(Sa/Da)を求めた。ここで、本発明の積層体において、コート層20が本発明の膜となる。
(分析装置と測定条件)
TOF-SIMS; ION・TOF社製 TOF-SIMS 5
1次イオン; Bi3 2+ (加速電圧25kV)
測定面積; 300~340μm2 測定には帯電補正用電子銃を使用
試料調製等
図2に示す通りに、基材10の表面にコート層20が設けられたサンプルを切削方向30に向かって、精密斜め切削を行った後、10mm×10mm程度の大きさに切り出し、測定面にメッシュを当て、サンプルホルダーに固定し、外気と接するコート層表面40および膜の内部であるコート層内部50(膜厚1/2の地点、基材10に接するコート層の内表面)で飛行時間型2次イオン質量分析装置(TOF-SIMS)を用いてスルホン酸濃度を測定した。
評価
評価は以下の計算式で行った。なお、各測定点のイオン濃度は、相対強度(トータル検出イオンに対する)を用いた。
傾斜度Sa/Da(スルホン酸濃度比,傾斜度)=コート層表面40でのスルホン酸濃度/コート層20の膜厚1/2の地点でのスルホン酸濃度
<膜厚の測定>
(測定装置および条件)
装置 :電界放出型透過電子顕微鏡(FE-TEM): JEM-2200FS(日本電子製)
加速電圧 : 200 kV
FIB(Focused Ion Beam System,集束イオンビームシステム)加工装置 : SMI2050(セイコーインスツルメンツ社製)
試料調製等
試料の凸面中央部を切り出した後、試料最表面にPtコートおよびカーボン蒸着を行った。これをFIB加工により薄膜化し、測定検体とした。測定検体を電界放出型透過電子顕微鏡(FE-TEM)で観察し、膜厚を測定した。
減圧下で脱ガスされたメタノール483.3gを反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク28.0g(0.424モル)を徐々に加えて完溶させた。次にアクリルアミド-t-ブチルスルホン酸(以下ATBSと略す。)89.9g(0.424モル)を分割装入して中和(pH=7.2)を行い、アクリルアミド-t-ブチルスルホン酸カリウム塩(以下ATBS-Kと略す。)を含む中和混合物を作製した。
GMA 7.54g(0.0530モル)およびKBE-503 15.40g(0.0530モル)を、GMA 5.48g(0.0386モル)のみに変更する以外は、合成例1と同様に共重合体の作製を行い、その結果、白色の共重合体 108.8g(収率97%)が得られた。得られた共重合体のGPC分析を行った結果、重量平均分子量 Mw=90,000、分子量分布 Mw/Mn=3.1であった。また、13C-NMR分析を行った結果、共重合体の構成単位比率 ATBS-Kユニット/GMAユニット/KBE-503ユニット=91/9/0であった。なお、エポキシ基が開環したユニットは検出されなかった。結果を表1に掲載する。
GMA 7.54g(0.0530モル)およびKBE-503 15.40g(0.0530モル)を、KBE-503 30.8g(0.1061モル)に変更する以外は、合成例1と同様に共重合体の作製を行い、その結果、白色の共重合体 84.7g(収率62%)が得られた。得られた共重合体のGPC分析を行った結果、重量平均分子量 Mw=76,000、分子量分布 Mw/Mn=2.7であった。また、13C-NMR分析を行った結果、共重合体の構成単位比率 ATBS-Kユニット/GMAユニット/KBE-503ユニット=90/0/10であった。結果を表1に掲載する。
<処方1 コーティング溶液1 の調製>
下記表2の混合順に従い各成分を混合し、最後に平均孔径0.5μmのフィルターを通してコーティング溶液1を調製した。
撹拌条件下、シランカップリング剤であるビス(トリメトキシシリルプロピル)アミン(以下KBM-666Pと略す。)0.5gに、2-メトキシエタノール(以下EGMと略す。)94.5g、および5水5.0gを混合して、固形分0.5wt%のプライマー用組成物を調製した。
基材であるよく洗浄されたガラス板(表面の水接触角 8°未満)を、スピンコーター(MIKASA SPINCOATER 1H-DX2)にセットし、500rpmの回転速度で回転させながら、調製したプライマー用組成物(固形分0.5wt%)を滴下し、滴下5秒後4000rpmに回転速度を上昇し、さらにその回転数で10秒間基材を回転させて、プライマー用組成物を基材表面に均一に塗布した。得られた塗布基材を50℃のオーブンで1分間予備乾燥した後、120℃のオーブンで1時間加熱し、基材上に、厚み5nmのシランカップリング剤から形成されたプライマー処理基板を得た。
上記プライマー処理基材のプライマー層の上に、上記コーティング液1をバーコーター#30で塗布し、50℃のオーブンで1分間予備乾燥した後、120℃×2時間加熱し、プライマー層上に、厚み3μmのコーティング膜を形成した。
合成例1の共重合体を、合成例2の共重合体に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価を上述の方法に従って実施例1と同様に行った。結果を表3に掲載する。
プライマー層を形成する基材をポリカーボネート板(以下PC板と略す。)とする以外は、実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(PC板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価を上述の方法に従って実施例1と同様に行った。結果を表3に掲載する。
プライマー層を形成する基材をPC板とする以外は、実施例2と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(PC板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価を上述の方法に従って実施例1と同様に行った。結果を表3に掲載する。
(CH110901ポリマーの製造)
減圧下で脱ガスされたメタノール535.5gを反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク23.6g(0.357モル)を徐々に加えて完溶させた。次にATBS 75.7g(0.357モル)を分割装入して中和(pH=7.5)を行い、ATBS-Kを含む中和混合物を作製した。
製造された共重合体(CH110901ポリマー) 5.8gに水40gを混合して溶液を作製した後、撹拌条件下、この溶液に2-メトキシエタノール(以下EGMと略す。)35g、テトラエトキシシラン(以下TEOSと略す。)20.1g、および5wt%硫酸水6gを混合した。得られた混合液を平均孔径0.5μmのフィルターに通して、固形分(共重合体およびSiO2換算のTEOSの合計量)固形分11wt%の無色透明なコーティング用組成物1 106.9gを得た。この組成物中の重合体/TEOS(SiO2換算)重量比は50/50である。
コーティング溶液1のバーコーター#30による基材への塗布を、上記コーティング用組成物1のバーコーター#18による基材への塗布に変更する以外は、実施例1と同様に、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価を上述の方法に従って実施例1と同様に行った。結果を表3に掲載する。
下記表4(処方2)の混合順に従い各成分を混合し、最後に平均孔径0.5μmのフィルターを通してコーティング溶液2を調製した。
合成例1の共重合体を、合成例2の共重合体に変更する以外は実施例3と同様にコーティング溶液2の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価を上述の方法に従って実施例3と同様に行った。結果を表5に掲載する。
合成例1の共重合体を、合成例3の共重合体に変更し、10wt% DS-Na水-EGM溶液(界面活性剤)の添加量を0.05gから0.5gに変更(10倍に変更)する以外は実施例3と同様にコーティング溶液2の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価を上述の方法に従って実施例3と同様に行った。結果を表5に掲載する。
(コーティング用組成物2の調製)
比較例1で製造された共重合体(CH110901ポリマー) 2.3gに水30gを混合して溶液を作製した後、撹拌条件下、この溶液にEGM 35g、TEOS 32.1g、および5wt%硫酸水6gを混合した。得られた混合液を平均孔径0.5μmのフィルターに通して、固形分(共重合体およびSiO2換算のTEOSの合計量)固形分11wt%の無色透明なコーティング用組成物2 105.4gを得た。この組成物中の重合体/TEOS(SiO2換算)重量比は20/80である。
コーティング溶液2のバーコーター#30による基材への塗布を、上記コーティング用組成物2のバーコーター#18による基材への塗布に変更する以外は、実施例5と同様に、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価を上述の方法に従って実施例5と同様に行った。結果を表5に掲載する。
**TOF-SIMS分析による、親水性膜の膜厚1/2の地点内部のイオン強度
1)TOS-SIMS スルホン酸(SO3 -)強度
2)TOS-SIMS アミノ基(C2N3 -)強度
3)TOS-SIMS シリカ粒子(Si+)強度
[合成例4]<共重合体94/3/3 AFM0110の製造>
減圧下で脱ガスされたメタノール756.6gを反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク50.46g(0.7645モル)を徐々に加えて完溶させた。次にアクリルアミド-t-ブチルスルホン酸(以下ATBSと略す。)162.0g(0.7645モル)を分割装入して中和(pH=7.6)を行い、ATBS-Kを含む中和混合物を作製した。
減圧下で脱ガスされたメタノール322.5gと同様に脱ガスされたエタノール118.6を反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク15.00g(0.2273モル)を徐々に加えて完溶させた。次にアクリルアミド-t-ブチルスルホン酸(以下ATBSと略す。)48.16g(0.2273モル)を分割装入して中和(pH=7.4)を行い、ATBS-Kを含む中和混合物を作製した。
減圧下で脱ガスされたメタノール347.9gと同様に脱ガスされたエタノール173.9を反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク15.00g(0.2273モル)を徐々に加えて完溶させた。次にアクリルアミド-t-ブチルスルホン酸(以下ATBSと略す。)48.40g(0.2284モル)を分割装入して中和(pH=7.4)を行い、ATBS-Kを含む中和混合物を作製した。
合成例1の共重合体を、下記表8の共重合体種別の項に記載の共重合体に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性、密着性)を上述の方法に従って行った。結果を表8に掲載する。なお表8には実施例1の結果を再度掲載する。
合成例1の共重合体を、下記表8の共重合体種別の項に記載の共重合体に変更する以外は実施例2と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(PC板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性、密着性)を上述の方法に従って行った。結果を表8に掲載する。なお表8には実施例2の結果を再度掲載する。
減圧下で脱ガスされたメタノール1150.0gを反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク28.0g(0.424モル)を徐々に加えて完溶させた。次にATBS 89.9g(0.424モル)を分割装入して中和(pH=7.3)を行い、ATBS-Kを含む中和混合物を作製した。
GMA 11.31g(0.0795モル)、KBE-503 7.7g(0.0265モル)に変更する以外は、合成例7と同様に共重合体の作製を行い、その結果、白色の共重合体 92.2g(収率73%)が得られた。得られた共重合体のGPC分析を行った結果、重量平均分子量 Mw=71,000、分子量分布 Mw/Mn=2.5であった。また、13C-NMR分析を行った結果、共重合体の構成単位比率 ATBS-Kユニット/GMAユニット/KBE-503ユニット=82/14/4であった。なお、エポキシ基が開環したユニットは検出されなかった。結果を表9に掲載する。
合成例1の共重合体を、下記表10の共重合体種別の項に記載の共重合体に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性)を上述の方法に従って行った。結果を表10に掲載する。なお表10には実施例1の結果を再度掲載する。
合成例1の共重合体を、下記表10の共重合体種別の項に記載の共重合体に変更する以外は実施例3と同様にコーティング溶液2の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性)を上述の方法に従って行った。結果を表10に掲載する。なお表10には実施例3の結果を再度掲載する。
減圧下で脱ガスされたメタノール780.0gを反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク46.26g(0.7009モル)を徐々に加えて完溶させた。ATBS 150.0g(0.7078モル)を分割装入して中和(pH=7.7)を行い、ATBS-Kを含む中和混合物を作製した。
減圧下で脱ガスされたメタノール215.0gと脱ガスされたエタノール215.0gとを反応フラスコに装入し、攪拌しながら純度85wt%のKOHフレーク20.0g(0.303モル)を徐々に加えて完溶させた。ATBS 64.2g(0.303モル)を分割装入して中和(pH=7.4)を行い、ATBS-Kを含む中和混合物を作製した。
合成例1の共重合体を、下記表12の共重合体種別の項に記載の共重合体に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性)を上述の方法に従って行った。結果を表12に掲載する。なお表12には参考例1の結果を再度掲載する。
合成例1の共重合体を、下記表12の共重合体種別の項に記載の共重合体に変更する以外は実施例3と同様にコーティング溶液2の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性)を上述の方法に従って行った。結果を表12に掲載する。なお表12には参考例3の結果を再度掲載する。
コーティング溶液1に含まれるDS-Na(界面活性剤)の添加量を表13に記載の量に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性)を行った。結果を表13に掲載する。
コーティング溶液1に含まれる合成例1の共重合体の量、30wt%メタノールシリカゾルの量、および60wt%メチル化メラミン樹脂水溶液の量を表14および15に記載の量に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防汚性)を行った。結果を表14および15に掲載する。
反応フラスコにアニリン 94.0g(1.0モル)とメタノール 160g を仕込み、水浴下で、内容物を撹拌しながら35wt%塩酸 109.6g(1.0モル)を滴下し、次いで37wt%ホルムアルデヒド水溶液 178.4g(2.2モル)を装入した後、Reflux(内温67℃)で5時間反応させた。
コーティング溶液1に含まれるメチル化メラミン樹脂を表16に記載のアミノ樹脂(実施例43:メチル化尿素樹脂、実施例44:メチル化アニリン樹脂)に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性、密着性)を行った。結果を表16に掲載する。なお、下記表16には実施例1の結果を再度掲載する。
反応フラスコに、3-スルホプロピルアクリレート・ナトリウム塩(以下SPA-Naと略す。)52.43g(0.2425モル)、4-グリシジルオキシ-ブチルアクリレート(以下GOBAと略す。) 2.43g(0.0121モル)、3-アクリロイルオキシ-プロピルトリメトキシシラン(以下KBM-5103と略す。) 2.84g(0.0121モル)、および減圧下で脱ガスされたメタノール488.9gを装入して混合液を作製した。
合成例1の共重合体を、合成例12の共重合体に変更する以外は実施例1と同様にコーティング溶液1の調製、プライマー組成物の調製、プライマー層の形成、コーティング膜の形成を行い、基材(ガラス板)上にプライマー層およびコーティング膜が形成された積層体を得た。得られたコーティング膜の物性評価(外観、120℃加熱汚れ付着試験、鉛筆硬度、防曇性、防汚性、密着性)を行った。結果を表17に掲載する。
Claims (9)
- 下記一般式(1)、(2)および(3)で表される構成単位を含む共重合体(i)と、
アミノ樹脂(ii)と、
を含む組成物から得られる硬化物。
A1は、単結合、炭素数1~10である2価の炭化水素基、下記式(1-1)で表される基、または下記式(1-2)で表される基を示し、
A2は、単結合、炭素数1~10である2価の炭化水素基、下記式(2-1)で表される基、または下記式(2-2)で表される基を示し、
A3は、単結合、炭素数1~10である2価の炭化水素基、下記式(3-1)で表される基、または下記式(3-2)で表される基を示し、
R1、R2、およびR3は、それぞれ独立して、水素原子またはメチル基を示し、
R4は、水素原子、メチル基、エチル基、プロピル基、またはブチル基を示し、R4は互いに同一でも異なっていてもよく、
R10は、水素原子、メチル基、エチル基、プロピル基、ブチル基、メトキシ基、エトキシ基、プロポキシ基、またはブトキシ基を示し、
Mは、水素原子、アルカリ金属イオン、1/2価のアルカリ土類金属イオン、アンモニウムイオン、またはアミンイオンを示し;
下記式(1-1)、(1-2)、(2-1)、(2-2)、(3-1)、および(3-2)において、nおよびn2は、それぞれ独立して、1~10の整数であり、n1は0~10の整数であり、mは1~6の整数であり、m1は0~6の整数であり、lは0~4の整数であり、R5およびR6は、それぞれ独立して、水素原子またはメチル基を示し、*はSO3Mと結合する側の端部、**はエポキシ基と結合する側の端部、***はSi原子と結合する側の端部を示す。)
- 共重合体(i)が、下記一般式(4)、(5)および(6)で表される構成単位を含む共重合体(i3-1)である上記組成物から得られる請求項1に記載の硬化物。
nは1~10の整数であり、n1は0~10の整数であり、
R1、R2、R3、R5、およびR6は、それぞれ独立して、水素原子またはメチル基を示し、
R4は、水素原子、メチル基、エチル基、プロピル基、またはブチル基を示し、R4は互いに同一でも異なっていてもよく、
R10は、水素原子、メチル基、エチル基、プロピル基、ブチル基、メトキシ基、エトキシ基、プロポキシ基、またはブトキシ基を示し、
Mは、水素原子、アルカリ金属イオン、1/2価のアルカリ土類金属イオン、アンモニウムイオン、またはアミンイオンを示す。) - 共重合体(i)のゲルパーミエーションクロマトグラフィー(GPC)により測定した標準ポリメチルメタクリレート換算の重量平均分子量が500~3,000,000である上記組成物から得られる請求項1または2に記載の硬化物。
- アミノ樹脂(ii)が、下記一般式(7)で表されるアミノ樹脂(ii1)である上記組成物から得られる請求項1~3のいずれか1項に記載の硬化物。
下記式(8)において、q030は0~30の整数であり、q030は互いに同一でも異なっていてもよく、R30およびR40は式(7)中の定義と同一であり、
下記式(9)において、q050は0~50の整数であり、Xは酸素原子または硫黄原子を示し、R30およびR40は式(7)中の定義と同一であり、
下記式(10)において、q050は0~50の整数である。)
- 共重合体(i)と上記アミノ樹脂(ii)との重量比(i)/(ii)が99/1~1/99の範囲にある上記組成物から得られる請求項1~4のいずれか1項に記載の硬化物。
- さらに無機粒子(iii)を含む上記組成物から得られる請求項1~5のいずれか1項に記載の硬化物。
- 共重合体(i)5~98重量部、アミノ樹脂(ii)1~70重量部、および無機粒子(iii)1~90重量部(ただし、共重合体(i)、アミノ樹脂(ii)および無機粒子(iii)の合計重量が100重量部とする。)を含む上記組成物から得られる請求項6に記載の硬化物。
- 請求項1~7のいずれか1項に記載の硬化物からなる厚みが0.01~300μmである膜(Z1)。
- 請求項8に記載の膜(Z1)を少なくとも1層有する積層体。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020177002112A KR101910753B1 (ko) | 2014-07-31 | 2015-07-28 | 술폰산계 공중합체와 아미노 수지로 이루어지는 친수성 재료 |
| US15/329,131 US9976050B2 (en) | 2014-07-31 | 2015-07-28 | Hydrophilic materials including sulfonate copolymer and amino resin |
| EP15827650.1A EP3176216B1 (en) | 2014-07-31 | 2015-07-28 | Hydrophilic material comprising sulfonate copolymer and amino resin |
| CN201580040385.7A CN106574099B (zh) | 2014-07-31 | 2015-07-28 | 包含磺酸系共聚物和氨基树脂的亲水性材料 |
| JP2016538356A JP6434519B2 (ja) | 2014-07-31 | 2015-07-28 | スルホン酸系共重合体とアミノ樹脂からなる親水性材料 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014156146 | 2014-07-31 | ||
| JP2014-156146 | 2014-07-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016017619A1 true WO2016017619A1 (ja) | 2016-02-04 |
Family
ID=55217519
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/071331 Ceased WO2016017619A1 (ja) | 2014-07-31 | 2015-07-28 | スルホン酸系共重合体とアミノ樹脂からなる親水性材料 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9976050B2 (ja) |
| EP (1) | EP3176216B1 (ja) |
| JP (1) | JP6434519B2 (ja) |
| KR (1) | KR101910753B1 (ja) |
| CN (1) | CN106574099B (ja) |
| TW (1) | TW201609926A (ja) |
| WO (1) | WO2016017619A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20190038867A (ko) | 2016-08-26 | 2019-04-09 | 미쯔이가가꾸가부시끼가이샤 | 반사 방지성 적층체 |
| KR20190038868A (ko) | 2016-08-26 | 2019-04-09 | 미쯔이가가꾸가부시끼가이샤 | 방담성 적층체 |
| JP2019522062A (ja) * | 2016-04-14 | 2019-08-08 | ビーエーエスエフ ソシエタス・ヨーロピアBasf Se | 水膨潤性ポリマーコアとゾルゲルコーティングとを含む被覆されたポリマー粒子 |
| JPWO2021045136A1 (ja) * | 2019-09-06 | 2021-03-11 |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10025468B2 (en) * | 2013-11-15 | 2018-07-17 | Google Llc | Personalization of map content via an application programming interface |
| BR112017024689B1 (pt) | 2015-06-17 | 2022-04-12 | Clariant International Ltd | Polímeros hidrossolúveis ou incháveis em água, seu processo de produção, seu uso, processo de cimentação de perfurações profundas utilizando uma lama de cimento e mistura polimérica |
| WO2018108611A1 (en) | 2016-12-12 | 2018-06-21 | Clariant International Ltd | Use of bio-based polymer in a cosmetic, dermatological or pharmaceutical composition |
| JP7032402B2 (ja) | 2016-12-12 | 2022-03-08 | クラリアント・インターナシヨナル・リミテツド | ある特定のレベルのバイオベース炭素を含むポリマー |
| WO2018108665A1 (en) | 2016-12-15 | 2018-06-21 | Clariant International Ltd | Water-soluble and/or water-swellable hybrid polymer |
| EP3554646B1 (en) | 2016-12-15 | 2025-03-19 | Clariant International Ltd | Water-soluble and/or water-swellable hybrid polymer |
| ES3017507T3 (en) | 2016-12-15 | 2025-05-13 | Clariant Int Ltd | Water-soluble and/or water-swellable hybrid polymer |
| US11306170B2 (en) | 2016-12-15 | 2022-04-19 | Clariant International Ltd. | Water-soluble and/or water-swellable hybrid polymer |
| AU2018235066A1 (en) | 2017-03-15 | 2019-10-17 | Samsonite Ip Holdings S.Á R.L. | Biaxially oriented thermoplastic polymer laminate films for luggage articles and methods of making the same |
| JP6568294B2 (ja) * | 2018-02-21 | 2019-08-28 | デクセリアルズ株式会社 | 活性エネルギー線硬化性樹脂組成物、防曇防汚積層体、及びその製造方法、物品、並びに防曇方法 |
| CN115028830B (zh) * | 2022-05-10 | 2023-12-22 | 武汉中科先进材料科技有限公司 | 一种用于防雾涂层的亲水树脂及其制备方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0948945A (ja) * | 1995-05-31 | 1997-02-18 | Nippon Carbide Ind Co Inc | 水性被覆剤組成物 |
| WO2008143143A1 (ja) * | 2007-05-15 | 2008-11-27 | Fujifilm Corporation | 親水性コーティング組成物及びこれを用いた親水性部材 |
| WO2014168123A1 (ja) * | 2013-04-12 | 2014-10-16 | 三井化学株式会社 | 共重合体または組成物からなる膜 |
| WO2014168122A1 (ja) * | 2013-04-12 | 2014-10-16 | 三井化学株式会社 | 共重合体及びそれからなる親水性材料 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5622365A (en) | 1979-08-02 | 1981-03-02 | Nippon Sheet Glass Co Ltd | Coating composition |
| JPS61166824A (ja) | 1986-01-24 | 1986-07-28 | Toray Ind Inc | 硬化性樹脂組成物 |
| JPH05287232A (ja) * | 1992-04-08 | 1993-11-02 | Kansai Paint Co Ltd | 塗料組成物 |
| JPH06166847A (ja) | 1992-11-30 | 1994-06-14 | Mitsubishi Kasei Corp | ハードコート |
| JPH1121512A (ja) | 1997-07-01 | 1999-01-26 | Asahi Denka Kogyo Kk | 親水性コーティング用樹脂組成物 |
| JPH11106439A (ja) * | 1997-10-03 | 1999-04-20 | Nippon Nsc Ltd | アクリル系水系樹脂 |
| JP2006089589A (ja) | 2004-09-24 | 2006-04-06 | Daicel Chem Ind Ltd | 金属表面用水分散性樹脂処理剤及び表面処理金属板 |
| JP2006342221A (ja) | 2005-06-08 | 2006-12-21 | Daicel Chem Ind Ltd | 水分散性樹脂組成物 |
| CN101309760B (zh) | 2005-12-02 | 2012-07-25 | 三井化学株式会社 | 单层膜及由其构成的亲水性材料 |
| JP2009062463A (ja) | 2007-09-06 | 2009-03-26 | Fujifilm Corp | 光硬化型親水性組成物 |
| JP2013020103A (ja) * | 2011-07-12 | 2013-01-31 | Mitsubishi Paper Mills Ltd | ネガ型感光性平版印刷版 |
| KR101588988B1 (ko) * | 2011-10-14 | 2016-01-26 | 미쓰이 가가쿠 가부시키가이샤 | 조성물 및 그것으로 이루어지는 막 |
-
2015
- 2015-07-28 EP EP15827650.1A patent/EP3176216B1/en not_active Not-in-force
- 2015-07-28 CN CN201580040385.7A patent/CN106574099B/zh not_active Expired - Fee Related
- 2015-07-28 JP JP2016538356A patent/JP6434519B2/ja not_active Expired - Fee Related
- 2015-07-28 US US15/329,131 patent/US9976050B2/en active Active
- 2015-07-28 KR KR1020177002112A patent/KR101910753B1/ko not_active Expired - Fee Related
- 2015-07-28 WO PCT/JP2015/071331 patent/WO2016017619A1/ja not_active Ceased
- 2015-07-29 TW TW104124531A patent/TW201609926A/zh unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0948945A (ja) * | 1995-05-31 | 1997-02-18 | Nippon Carbide Ind Co Inc | 水性被覆剤組成物 |
| WO2008143143A1 (ja) * | 2007-05-15 | 2008-11-27 | Fujifilm Corporation | 親水性コーティング組成物及びこれを用いた親水性部材 |
| WO2014168123A1 (ja) * | 2013-04-12 | 2014-10-16 | 三井化学株式会社 | 共重合体または組成物からなる膜 |
| WO2014168122A1 (ja) * | 2013-04-12 | 2014-10-16 | 三井化学株式会社 | 共重合体及びそれからなる親水性材料 |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019522062A (ja) * | 2016-04-14 | 2019-08-08 | ビーエーエスエフ ソシエタス・ヨーロピアBasf Se | 水膨潤性ポリマーコアとゾルゲルコーティングとを含む被覆されたポリマー粒子 |
| JP7004666B2 (ja) | 2016-04-14 | 2022-01-21 | ビーエーエスエフ ソシエタス・ヨーロピア | 水膨潤性ポリマーコアとゾルゲルコーティングとを含む被覆されたポリマー粒子 |
| KR20190038867A (ko) | 2016-08-26 | 2019-04-09 | 미쯔이가가꾸가부시끼가이샤 | 반사 방지성 적층체 |
| KR20190038868A (ko) | 2016-08-26 | 2019-04-09 | 미쯔이가가꾸가부시끼가이샤 | 방담성 적층체 |
| US11338562B2 (en) | 2016-08-26 | 2022-05-24 | Mitsui Chemicals, Inc. | Antifogging laminate |
| JPWO2021045136A1 (ja) * | 2019-09-06 | 2021-03-11 | ||
| JP7510945B2 (ja) | 2019-09-06 | 2024-07-04 | 株式会社カネカ | 水溶性(メタ)アクリル系樹脂およびその利用 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2016017619A1 (ja) | 2017-05-18 |
| TW201609926A (zh) | 2016-03-16 |
| US20170210937A1 (en) | 2017-07-27 |
| US9976050B2 (en) | 2018-05-22 |
| EP3176216A4 (en) | 2018-01-24 |
| EP3176216B1 (en) | 2019-04-24 |
| KR101910753B1 (ko) | 2018-10-22 |
| CN106574099A (zh) | 2017-04-19 |
| JP6434519B2 (ja) | 2018-12-05 |
| EP3176216A1 (en) | 2017-06-07 |
| CN106574099B (zh) | 2019-06-21 |
| KR20170023140A (ko) | 2017-03-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6434519B2 (ja) | スルホン酸系共重合体とアミノ樹脂からなる親水性材料 | |
| JP6283718B2 (ja) | 共重合体及びそれからなる親水性材料 | |
| JP6239086B2 (ja) | 共重合体または組成物からなる膜 | |
| JP5788014B2 (ja) | 組成物及びそれからなる膜 | |
| JP2019131763A (ja) | イオン性基を有するポリマーの製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15827650 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016538356 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20177002112 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15329131 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015827650 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015827650 Country of ref document: EP |