WO2014021282A1 - 抗白癬菌症外用液剤 - Google Patents
抗白癬菌症外用液剤 Download PDFInfo
- Publication number
- WO2014021282A1 WO2014021282A1 PCT/JP2013/070525 JP2013070525W WO2014021282A1 WO 2014021282 A1 WO2014021282 A1 WO 2014021282A1 JP 2013070525 W JP2013070525 W JP 2013070525W WO 2014021282 A1 WO2014021282 A1 WO 2014021282A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- hydrogen atom
- pyrazol
- dimethyl
- production example
- Prior art date
Links
- 150000001875 compounds Chemical class 0.000 claims abstract description 178
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims abstract description 103
- 150000003839 salts Chemical class 0.000 claims abstract description 39
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 34
- 150000002367 halogens Chemical group 0.000 claims abstract description 34
- 208000002474 Tinea Diseases 0.000 claims abstract description 31
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 25
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims abstract description 22
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims abstract description 19
- 125000002252 acyl group Chemical group 0.000 claims abstract description 17
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims abstract description 15
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims abstract description 8
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims abstract description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 124
- 125000000217 alkyl group Chemical group 0.000 claims description 99
- -1 —NR a R b Chemical group 0.000 claims description 64
- 238000002360 preparation method Methods 0.000 claims description 47
- 239000007788 liquid Substances 0.000 claims description 26
- 241000893966 Trichophyton verrucosum Species 0.000 claims description 21
- 229940079593 drug Drugs 0.000 claims description 14
- 239000003814 drug Substances 0.000 claims description 14
- 229920000642 polymer Polymers 0.000 claims description 12
- 230000002265 prevention Effects 0.000 claims description 11
- 206010017533 Fungal infection Diseases 0.000 claims description 9
- 208000024386 fungal infectious disease Diseases 0.000 claims description 9
- 208000010195 Onychomycosis Diseases 0.000 claims description 8
- 201000005882 tinea unguium Diseases 0.000 claims description 8
- 229920003144 amino alkyl methacrylate copolymer Polymers 0.000 claims description 7
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 claims description 5
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 claims description 5
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 claims description 5
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 claims description 5
- 229920001296 polysiloxane Polymers 0.000 claims description 5
- VQXHPRBERPTLQW-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-methylphenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(C)C=C1O VQXHPRBERPTLQW-UHFFFAOYSA-N 0.000 claims description 4
- 229920000178 Acrylic resin Polymers 0.000 claims description 4
- 239000004925 Acrylic resin Substances 0.000 claims description 4
- 239000002562 thickening agent Substances 0.000 claims description 4
- RVGRUAULSDPKGF-UHFFFAOYSA-N Poloxamer Chemical compound C1CO1.CC1CO1 RVGRUAULSDPKGF-UHFFFAOYSA-N 0.000 claims description 3
- 210000000078 claw Anatomy 0.000 claims description 3
- 229960000502 poloxamer Drugs 0.000 claims description 3
- 229920001983 poloxamer Polymers 0.000 claims description 3
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims description 3
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims description 3
- 229940069328 povidone Drugs 0.000 claims description 3
- UUEVFMOUBSLVJW-UHFFFAOYSA-N oxo-[[1-[2-[2-[2-[4-(oxoazaniumylmethylidene)pyridin-1-yl]ethoxy]ethoxy]ethyl]pyridin-4-ylidene]methyl]azanium;dibromide Chemical compound [Br-].[Br-].C1=CC(=C[NH+]=O)C=CN1CCOCCOCCN1C=CC(=C[NH+]=O)C=C1 UUEVFMOUBSLVJW-UHFFFAOYSA-N 0.000 claims description 2
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 claims 1
- 230000000699 topical effect Effects 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 21
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 abstract description 15
- 230000035699 permeability Effects 0.000 abstract description 11
- 229910003827 NRaRb Inorganic materials 0.000 abstract 1
- 206010067409 Trichophytosis Diseases 0.000 abstract 1
- 230000015572 biosynthetic process Effects 0.000 abstract 1
- 239000011248 coating agent Substances 0.000 abstract 1
- 238000000576 coating method Methods 0.000 abstract 1
- 238000004519 manufacturing process Methods 0.000 description 222
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 173
- 238000005160 1H NMR spectroscopy Methods 0.000 description 120
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 72
- 239000002904 solvent Substances 0.000 description 67
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 60
- 238000006243 chemical reaction Methods 0.000 description 51
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 50
- 239000000243 solution Substances 0.000 description 50
- 239000000203 mixture Substances 0.000 description 49
- 210000000282 nail Anatomy 0.000 description 49
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 49
- 238000004992 fast atom bombardment mass spectroscopy Methods 0.000 description 43
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 42
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 39
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 34
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 29
- 239000000758 substrate Substances 0.000 description 28
- 239000012044 organic layer Substances 0.000 description 26
- 238000010898 silica gel chromatography Methods 0.000 description 25
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 24
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 23
- 239000011541 reaction mixture Substances 0.000 description 23
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 22
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 20
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 20
- 239000002253 acid Substances 0.000 description 19
- 210000003491 skin Anatomy 0.000 description 19
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 18
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 18
- BTANRVKWQNVYAZ-UHFFFAOYSA-N butan-2-ol Chemical compound CCC(C)O BTANRVKWQNVYAZ-UHFFFAOYSA-N 0.000 description 18
- 238000000034 method Methods 0.000 description 18
- 238000012360 testing method Methods 0.000 description 18
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 17
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 16
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 15
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 15
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 14
- 238000006722 reduction reaction Methods 0.000 description 14
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical class CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 13
- YRKCREAYFQTBPV-UHFFFAOYSA-N acetylacetone Chemical compound CC(=O)CC(C)=O YRKCREAYFQTBPV-UHFFFAOYSA-N 0.000 description 13
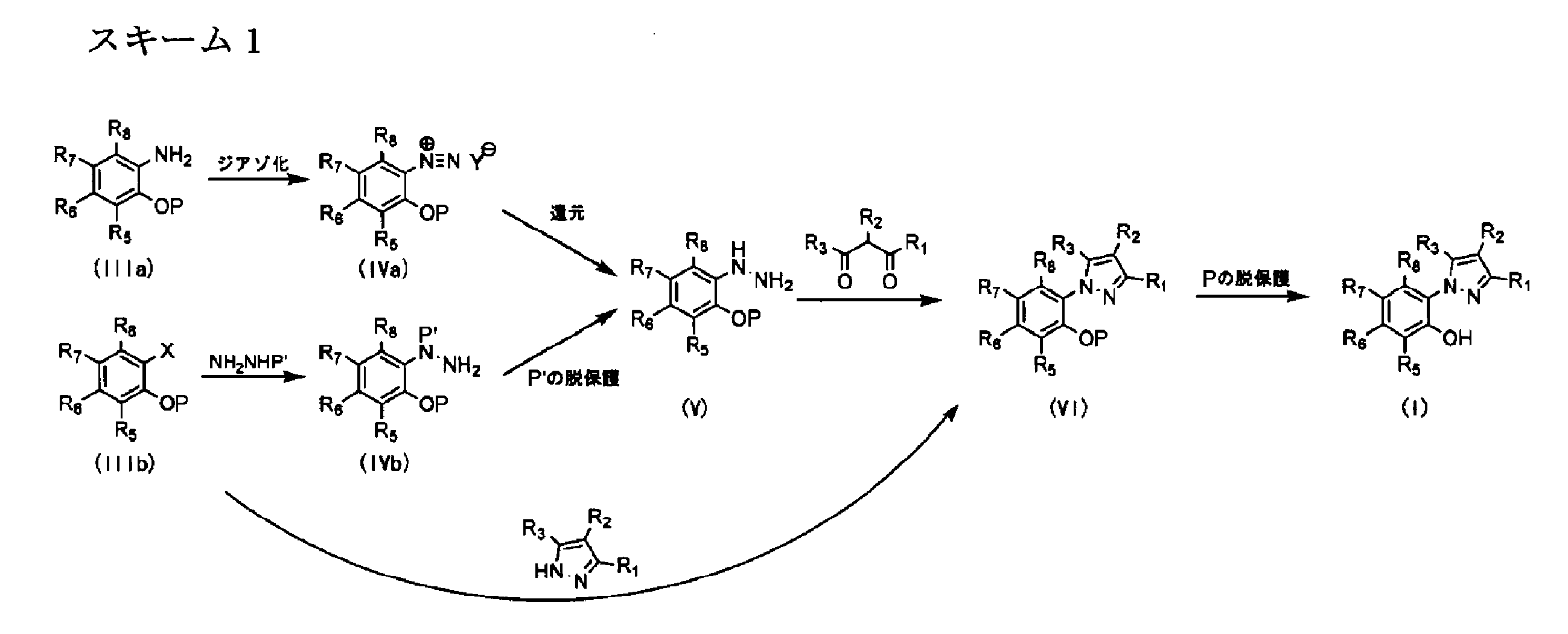
- 238000006193 diazotization reaction Methods 0.000 description 13
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 12
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 12
- 150000001989 diazonium salts Chemical class 0.000 description 11
- 229960003742 phenol Drugs 0.000 description 11
- 238000010992 reflux Methods 0.000 description 11
- BMTGLHWYLKPGMY-UHFFFAOYSA-N 2-pyrazol-1-ylphenol Chemical class OC1=CC=CC=C1N1N=CC=C1 BMTGLHWYLKPGMY-UHFFFAOYSA-N 0.000 description 10
- 241000283690 Bos taurus Species 0.000 description 10
- 241000233866 Fungi Species 0.000 description 10
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 10
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 10
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 10
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 10
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 9
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 9
- 238000001035 drying Methods 0.000 description 9
- 125000006239 protecting group Chemical group 0.000 description 9
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 9
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 8
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 8
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 8
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 8
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 8
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 8
- 239000001257 hydrogen Substances 0.000 description 8
- 229910052739 hydrogen Inorganic materials 0.000 description 8
- AZUYLZMQTIKGSC-UHFFFAOYSA-N 1-[6-[4-(5-chloro-6-methyl-1H-indazol-4-yl)-5-methyl-3-(1-methylindazol-5-yl)pyrazol-1-yl]-2-azaspiro[3.3]heptan-2-yl]prop-2-en-1-one Chemical compound ClC=1C(=C2C=NNC2=CC=1C)C=1C(=NN(C=1C)C1CC2(CN(C2)C(C=C)=O)C1)C=1C=C2C=NN(C2=CC=1)C AZUYLZMQTIKGSC-UHFFFAOYSA-N 0.000 description 7
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 7
- GSOHKPVFCOWKPU-UHFFFAOYSA-N 3-methylpentane-2,4-dione Chemical compound CC(=O)C(C)C(C)=O GSOHKPVFCOWKPU-UHFFFAOYSA-N 0.000 description 7
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 7
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 7
- 150000002429 hydrazines Chemical class 0.000 description 7
- HECSHXUNOVTAIJ-UHFFFAOYSA-N hydron;(2-methoxyphenyl)hydrazine;chloride Chemical compound Cl.COC1=CC=CC=C1NN HECSHXUNOVTAIJ-UHFFFAOYSA-N 0.000 description 7
- 239000002198 insoluble material Substances 0.000 description 7
- 239000010410 layer Substances 0.000 description 7
- 229910052751 metal Inorganic materials 0.000 description 7
- 239000002184 metal Substances 0.000 description 7
- 230000009467 reduction Effects 0.000 description 7
- 229920006395 saturated elastomer Polymers 0.000 description 7
- TXUICONDJPYNPY-UHFFFAOYSA-N (1,10,13-trimethyl-3-oxo-4,5,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-17-yl) heptanoate Chemical compound C1CC2CC(=O)C=C(C)C2(C)C2C1C1CCC(OC(=O)CCCCCC)C1(C)CC2 TXUICONDJPYNPY-UHFFFAOYSA-N 0.000 description 6
- ZNQVEEAIQZEUHB-UHFFFAOYSA-N 2-ethoxyethanol Chemical compound CCOCCO ZNQVEEAIQZEUHB-UHFFFAOYSA-N 0.000 description 6
- YYROPELSRYBVMQ-UHFFFAOYSA-N 4-toluenesulfonyl chloride Chemical compound CC1=CC=C(S(Cl)(=O)=O)C=C1 YYROPELSRYBVMQ-UHFFFAOYSA-N 0.000 description 6
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 6
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 6
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 6
- 229910021626 Tin(II) chloride Inorganic materials 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 239000012298 atmosphere Substances 0.000 description 6
- 239000003054 catalyst Substances 0.000 description 6
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 6
- 239000004210 ether based solvent Substances 0.000 description 6
- 238000011156 evaluation Methods 0.000 description 6
- 235000011187 glycerol Nutrition 0.000 description 6
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 6
- 235000011150 stannous chloride Nutrition 0.000 description 6
- 239000001119 stannous chloride Substances 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 229920003169 water-soluble polymer Polymers 0.000 description 6
- VLRGXXKFHVJQOL-UHFFFAOYSA-N 3-chloropentane-2,4-dione Chemical compound CC(=O)C(Cl)C(C)=O VLRGXXKFHVJQOL-UHFFFAOYSA-N 0.000 description 5
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 5
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 5
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 5
- 241001045770 Trichophyton mentagrophytes Species 0.000 description 5
- 244000052616 bacterial pathogen Species 0.000 description 5
- 238000009903 catalytic hydrogenation reaction Methods 0.000 description 5
- 239000003638 chemical reducing agent Substances 0.000 description 5
- 238000010511 deprotection reaction Methods 0.000 description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 5
- 125000003226 pyrazolyl group Chemical group 0.000 description 5
- 235000010288 sodium nitrite Nutrition 0.000 description 5
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 5
- 239000012453 solvate Substances 0.000 description 5
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 5
- 229920003176 water-insoluble polymer Polymers 0.000 description 5
- DNIAPMSPPWPWGF-VKHMYHEASA-N (+)-propylene glycol Chemical compound C[C@H](O)CO DNIAPMSPPWPWGF-VKHMYHEASA-N 0.000 description 4
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 4
- QPFMBZIOSGYJDE-UHFFFAOYSA-N 1,1,2,2-tetrachloroethane Chemical compound ClC(Cl)C(Cl)Cl QPFMBZIOSGYJDE-UHFFFAOYSA-N 0.000 description 4
- DHKHKXVYLBGOIT-UHFFFAOYSA-N 1,1-Diethoxyethane Chemical compound CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 4
- LZDKZFUFMNSQCJ-UHFFFAOYSA-N 1,2-diethoxyethane Chemical compound CCOCCOCC LZDKZFUFMNSQCJ-UHFFFAOYSA-N 0.000 description 4
- YPFDHNVEDLHUCE-UHFFFAOYSA-N 1,3-propanediol Substances OCCCO YPFDHNVEDLHUCE-UHFFFAOYSA-N 0.000 description 4
- TZZOTGGQFFDSCI-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)benzene-1,4-diol Chemical compound N1=C(C)C=C(C)N1C1=CC(O)=CC=C1O TZZOTGGQFFDSCI-UHFFFAOYSA-N 0.000 description 4
- UFLNGSAFJHIAQR-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC=CC=C1O UFLNGSAFJHIAQR-UHFFFAOYSA-N 0.000 description 4
- QEHQADXVFUDOBF-UHFFFAOYSA-N 2-(4-chloro-3,5-dimethylpyrazol-1-yl)benzene-1,4-diol Chemical compound CC1=C(Cl)C(C)=NN1C1=CC(O)=CC=C1O QEHQADXVFUDOBF-UHFFFAOYSA-N 0.000 description 4
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 4
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 4
- FFDGPVCHZBVARC-UHFFFAOYSA-N N,N-dimethylglycine Chemical compound CN(C)CC(O)=O FFDGPVCHZBVARC-UHFFFAOYSA-N 0.000 description 4
- IOVCWXUNBOPUCH-UHFFFAOYSA-M Nitrite anion Chemical compound [O-]N=O IOVCWXUNBOPUCH-UHFFFAOYSA-M 0.000 description 4
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 4
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 4
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 4
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 4
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 4
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 4
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 230000000996 additive effect Effects 0.000 description 4
- 150000001298 alcohols Chemical class 0.000 description 4
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 4
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 4
- 229940121375 antifungal agent Drugs 0.000 description 4
- 239000000010 aprotic solvent Substances 0.000 description 4
- 239000002585 base Substances 0.000 description 4
- ILAHWRKJUDSMFH-UHFFFAOYSA-N boron tribromide Chemical compound BrB(Br)Br ILAHWRKJUDSMFH-UHFFFAOYSA-N 0.000 description 4
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 4
- 230000003197 catalytic effect Effects 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 235000014113 dietary fatty acids Nutrition 0.000 description 4
- 239000000194 fatty acid Substances 0.000 description 4
- 229930195729 fatty acid Natural products 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 4
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- YKYONYBAUNKHLG-UHFFFAOYSA-N n-Propyl acetate Natural products CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 description 4
- 150000008048 phenylpyrazoles Chemical class 0.000 description 4
- 229910052698 phosphorus Inorganic materials 0.000 description 4
- 229920000166 polytrimethylene carbonate Polymers 0.000 description 4
- 229910052700 potassium Inorganic materials 0.000 description 4
- 239000011591 potassium Substances 0.000 description 4
- 238000001556 precipitation Methods 0.000 description 4
- 239000003755 preservative agent Substances 0.000 description 4
- 229940090181 propyl acetate Drugs 0.000 description 4
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 4
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 4
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 4
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 4
- UJUXBIXHNNSHGH-UHFFFAOYSA-N (4-amino-3-hydroxyphenyl) acetate Chemical compound CC(=O)OC1=CC=C(N)C(O)=C1 UJUXBIXHNNSHGH-UHFFFAOYSA-N 0.000 description 3
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 3
- NRSKRRKWPPDKNI-UHFFFAOYSA-N 2-(1h-pyrazol-5-yl)aniline Chemical class NC1=CC=CC=C1C1=CC=NN1 NRSKRRKWPPDKNI-UHFFFAOYSA-N 0.000 description 3
- IHVOUZNUAKSPLX-UHFFFAOYSA-N 2-(3,4,5-trimethylpyrazol-1-yl)phenol Chemical compound CC1=C(C)C(C)=NN1C1=CC=CC=C1O IHVOUZNUAKSPLX-UHFFFAOYSA-N 0.000 description 3
- AKYRTWWMTOMHLH-UHFFFAOYSA-N 2-(3,5-diethylpyrazol-1-yl)phenol Chemical compound N1=C(CC)C=C(CC)N1C1=CC=CC=C1O AKYRTWWMTOMHLH-UHFFFAOYSA-N 0.000 description 3
- LFCDNGDVVIIYSM-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-3-nitrophenol Chemical compound N1=C(C)C=C(C)N1C1=C(O)C=CC=C1[N+]([O-])=O LFCDNGDVVIIYSM-UHFFFAOYSA-N 0.000 description 3
- OSXPDSAVYIIRIX-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-4-fluorophenol Chemical compound N1=C(C)C=C(C)N1C1=CC(F)=CC=C1O OSXPDSAVYIIRIX-UHFFFAOYSA-N 0.000 description 3
- PRNCRWAGANVDSL-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-4-methoxy-5-methylphenol Chemical compound C1=C(C)C(OC)=CC(N2C(=CC(C)=N2)C)=C1O PRNCRWAGANVDSL-UHFFFAOYSA-N 0.000 description 3
- PRQXJWKTVXILBM-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-(methylamino)phenol Chemical compound OC1=CC(NC)=CC=C1N1C(C)=CC(C)=N1 PRQXJWKTVXILBM-UHFFFAOYSA-N 0.000 description 3
- IBPRZPFTMMGKPM-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-ethylphenol Chemical compound OC1=CC(CC)=CC=C1N1C(C)=CC(C)=N1 IBPRZPFTMMGKPM-UHFFFAOYSA-N 0.000 description 3
- VOPPOYGUVVCYNZ-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-fluorophenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(F)C=C1O VOPPOYGUVVCYNZ-UHFFFAOYSA-N 0.000 description 3
- YSAACJNGJBVKKY-UHFFFAOYSA-N 2-(3-methylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=CN1C1=CC=CC=C1O YSAACJNGJBVKKY-UHFFFAOYSA-N 0.000 description 3
- PGPSHIPZKDVTHH-UHFFFAOYSA-N 2-(4-butyl-3,5-dimethylpyrazol-1-yl)phenol Chemical compound CC1=C(CCCC)C(C)=NN1C1=CC=CC=C1O PGPSHIPZKDVTHH-UHFFFAOYSA-N 0.000 description 3
- JLLVCUBPORACNB-UHFFFAOYSA-N 2-(4-ethyl-3,5-dimethylpyrazol-1-yl)phenol Chemical compound CC1=C(CC)C(C)=NN1C1=CC=CC=C1O JLLVCUBPORACNB-UHFFFAOYSA-N 0.000 description 3
- MOSZMQYLEJOZQJ-UHFFFAOYSA-N 2-(5-amino-3-tert-butylpyrazol-1-yl)phenol Chemical compound N1=C(C(C)(C)C)C=C(N)N1C1=CC=CC=C1O MOSZMQYLEJOZQJ-UHFFFAOYSA-N 0.000 description 3
- NOABAQYHMDCRLP-UHFFFAOYSA-N 2-(5-methylpyrazol-1-yl)phenol Chemical compound CC1=CC=NN1C1=CC=CC=C1O NOABAQYHMDCRLP-UHFFFAOYSA-N 0.000 description 3
- IIDUNAVOCYMUFB-UHFFFAOYSA-N 2-amino-5-fluorophenol Chemical compound NC1=CC=C(F)C=C1O IIDUNAVOCYMUFB-UHFFFAOYSA-N 0.000 description 3
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical class NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 description 3
- SEJWMEIEGBEWOH-UHFFFAOYSA-N 2-chloro-6-(3,5-dimethylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC=CC(Cl)=C1O SEJWMEIEGBEWOH-UHFFFAOYSA-N 0.000 description 3
- DPJCXCZTLWNFOH-UHFFFAOYSA-N 2-nitroaniline Chemical class NC1=CC=CC=C1[N+]([O-])=O DPJCXCZTLWNFOH-UHFFFAOYSA-N 0.000 description 3
- HQKBEOULYYQQAV-UHFFFAOYSA-N 3-hydroxy-4-(3,4,5-trimethylpyrazol-1-yl)benzoic acid Chemical compound CC1=C(C)C(C)=NN1C1=CC=C(C(O)=O)C=C1O HQKBEOULYYQQAV-UHFFFAOYSA-N 0.000 description 3
- DDPGRMMKKXJUAQ-UHFFFAOYSA-N 4-(4-chloro-3,5-dimethylpyrazol-1-yl)-3-hydroxybenzamide Chemical compound CC1=C(Cl)C(C)=NN1C1=CC=C(C(N)=O)C=C1O DDPGRMMKKXJUAQ-UHFFFAOYSA-N 0.000 description 3
- BAYGNUDBTAVCED-UHFFFAOYSA-N 4-chloro-2-(3,5-dimethylpyrazol-1-yl)-5-methylphenol Chemical compound N1=C(C)C=C(C)N1C1=CC(Cl)=C(C)C=C1O BAYGNUDBTAVCED-UHFFFAOYSA-N 0.000 description 3
- LNRBQCSTNUDDGL-UHFFFAOYSA-N 5-(2-nitrophenyl)-1h-pyrazole Chemical class [O-][N+](=O)C1=CC=CC=C1C1=CC=NN1 LNRBQCSTNUDDGL-UHFFFAOYSA-N 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- 229920000936 Agarose Polymers 0.000 description 3
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 3
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 3
- 239000012448 Lithium borohydride Substances 0.000 description 3
- PWXUUAMFGUGJRM-UHFFFAOYSA-N N1=C(C)C=C(O)N1C1=CC=CC=C1O Chemical compound N1=C(C)C=C(O)N1C1=CC=CC=C1O PWXUUAMFGUGJRM-UHFFFAOYSA-N 0.000 description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 3
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 3
- 239000002202 Polyethylene glycol Substances 0.000 description 3
- 241000223238 Trichophyton Species 0.000 description 3
- LGBAXHLMOSOUMB-UHFFFAOYSA-N [4-(3,5-dimethylpyrazol-1-yl)-3-hydroxyphenyl] acetate Chemical compound OC1=CC(OC(=O)C)=CC=C1N1C(C)=CC(C)=N1 LGBAXHLMOSOUMB-UHFFFAOYSA-N 0.000 description 3
- 230000002378 acidificating effect Effects 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 3
- 230000000843 anti-fungal effect Effects 0.000 description 3
- 239000003429 antifungal agent Substances 0.000 description 3
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 3
- 125000005620 boronic acid group Chemical group 0.000 description 3
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 210000004027 cell Anatomy 0.000 description 3
- 239000010949 copper Substances 0.000 description 3
- 229910052802 copper Inorganic materials 0.000 description 3
- 125000004122 cyclic group Chemical group 0.000 description 3
- 239000012954 diazonium Substances 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 150000002148 esters Chemical class 0.000 description 3
- 244000053095 fungal pathogen Species 0.000 description 3
- 229940093915 gynecological organic acid Drugs 0.000 description 3
- 210000000003 hoof Anatomy 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 239000002609 medium Substances 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- OCZXDVNSNDITBS-UHFFFAOYSA-N methyl 4-amino-3-hydroxybenzoate Chemical compound COC(=O)C1=CC=C(N)C(O)=C1 OCZXDVNSNDITBS-UHFFFAOYSA-N 0.000 description 3
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 3
- 150000005181 nitrobenzenes Chemical class 0.000 description 3
- 239000012299 nitrogen atmosphere Substances 0.000 description 3
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 3
- 150000007524 organic acids Chemical class 0.000 description 3
- 235000005985 organic acids Nutrition 0.000 description 3
- 229920003023 plastic Polymers 0.000 description 3
- 239000004033 plastic Substances 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 229920002451 polyvinyl alcohol Polymers 0.000 description 3
- 229910000027 potassium carbonate Inorganic materials 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 description 3
- 230000001953 sensory effect Effects 0.000 description 3
- 229910000029 sodium carbonate Inorganic materials 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 238000003860 storage Methods 0.000 description 3
- JOXIMZWYDAKGHI-UHFFFAOYSA-M toluene-4-sulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-M 0.000 description 3
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 3
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical class OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- 239000011701 zinc Substances 0.000 description 3
- SQTUYFKNCCBFRR-UHFFFAOYSA-N (2,4-dimethoxyphenyl)boronic acid Chemical compound COC1=CC=C(B(O)O)C(OC)=C1 SQTUYFKNCCBFRR-UHFFFAOYSA-N 0.000 description 2
- ZWFOEEHTYYXTGJ-UHFFFAOYSA-N (2-amino-3-methylphenyl) 4-methylbenzenesulfonate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)OC1=CC=CC(C)=C1N ZWFOEEHTYYXTGJ-UHFFFAOYSA-N 0.000 description 2
- GGFCDOUVWPCWSZ-UHFFFAOYSA-N (2-amino-4-methylphenyl) 4-methylbenzenesulfonate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)OC1=CC=C(C)C=C1N GGFCDOUVWPCWSZ-UHFFFAOYSA-N 0.000 description 2
- MQHLMHIZUIDKOO-OKZBNKHCSA-N (2R,6S)-2,6-dimethyl-4-[(2S)-2-methyl-3-[4-(2-methylbutan-2-yl)phenyl]propyl]morpholine Chemical compound C1=CC(C(C)(C)CC)=CC=C1C[C@H](C)CN1C[C@@H](C)O[C@@H](C)C1 MQHLMHIZUIDKOO-OKZBNKHCSA-N 0.000 description 2
- UOGNFTJSCYHMKN-UHFFFAOYSA-N (3-acetyloxy-4-nitrophenyl) acetate Chemical compound CC(=O)OC1=CC=C([N+]([O-])=O)C(OC(C)=O)=C1 UOGNFTJSCYHMKN-UHFFFAOYSA-N 0.000 description 2
- CIHJXKYKTHZEER-UHFFFAOYSA-N (3-hydroxy-4-nitrophenyl) acetate Chemical compound CC(=O)OC1=CC=C([N+]([O-])=O)C(O)=C1 CIHJXKYKTHZEER-UHFFFAOYSA-N 0.000 description 2
- XRDYNHBJKCFHDN-UHFFFAOYSA-N (4-hydrazinyl-3-hydroxyphenyl) acetate;4-methylbenzenesulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1.CC(=O)OC1=CC=C(NN)C(O)=C1 XRDYNHBJKCFHDN-UHFFFAOYSA-N 0.000 description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 2
- UOCLXMDMGBRAIB-UHFFFAOYSA-N 1,1,1-trichloroethane Chemical compound CC(Cl)(Cl)Cl UOCLXMDMGBRAIB-UHFFFAOYSA-N 0.000 description 2
- 229940058015 1,3-butylene glycol Drugs 0.000 description 2
- UCCSDFOEIGJWAF-UHFFFAOYSA-N 1,4-dimethoxy-2-methyl-5-nitrobenzene Chemical compound COC1=CC([N+]([O-])=O)=C(OC)C=C1C UCCSDFOEIGJWAF-UHFFFAOYSA-N 0.000 description 2
- NPOZNJGLBIUCIO-UHFFFAOYSA-N 1-(2,5-dimethoxyphenyl)-3,5-dimethylpyrazole Chemical compound COC1=CC=C(OC)C(N2C(=CC(C)=N2)C)=C1 NPOZNJGLBIUCIO-UHFFFAOYSA-N 0.000 description 2
- JWTURNNUQFVWMT-UHFFFAOYSA-N 1-(2-methoxy-4-nitrophenyl)-3,5-dimethylpyrazole Chemical compound COC1=CC([N+]([O-])=O)=CC=C1N1C(C)=CC(C)=N1 JWTURNNUQFVWMT-UHFFFAOYSA-N 0.000 description 2
- QQPGEFNKQAVDPB-UHFFFAOYSA-N 1-(2-methoxyphenyl)pyrazole Chemical compound COC1=CC=CC=C1N1N=CC=C1 QQPGEFNKQAVDPB-UHFFFAOYSA-N 0.000 description 2
- JFGVNIYHEGSRQK-UHFFFAOYSA-N 1-(3-chloro-2-methoxyphenyl)-3,5-dimethylpyrazole Chemical compound COC1=C(Cl)C=CC=C1N1C(C)=CC(C)=N1 JFGVNIYHEGSRQK-UHFFFAOYSA-N 0.000 description 2
- YQTCQNIPQMJNTI-UHFFFAOYSA-N 2,2-dimethylpropan-1-one Chemical group CC(C)(C)[C]=O YQTCQNIPQMJNTI-UHFFFAOYSA-N 0.000 description 2
- RXATUQSSVSAJAI-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-3-methylphenol Chemical compound N1=C(C)C=C(C)N1C1=C(C)C=CC=C1O RXATUQSSVSAJAI-UHFFFAOYSA-N 0.000 description 2
- MLWFMZSFLZXQMC-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-4,5-dimethylphenol Chemical compound N1=C(C)C=C(C)N1C1=CC(C)=C(C)C=C1O MLWFMZSFLZXQMC-UHFFFAOYSA-N 0.000 description 2
- LSEANPSUWNEYQW-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-4-methylphenol Chemical compound N1=C(C)C=C(C)N1C1=CC(C)=CC=C1O LSEANPSUWNEYQW-UHFFFAOYSA-N 0.000 description 2
- DBZXNHWVWFRXRY-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-(trifluoromethyl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(C(F)(F)F)C=C1O DBZXNHWVWFRXRY-UHFFFAOYSA-N 0.000 description 2
- NRONTMCRANJURU-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-methoxyphenol Chemical compound OC1=CC(OC)=CC=C1N1C(C)=CC(C)=N1 NRONTMCRANJURU-UHFFFAOYSA-N 0.000 description 2
- FHWREENZKRCEBD-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-nitrophenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C([N+]([O-])=O)C=C1O FHWREENZKRCEBD-UHFFFAOYSA-N 0.000 description 2
- PKUDTZBFZDFCHN-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-6-methylphenol Chemical compound N1=C(C)C=C(C)N1C1=CC=CC(C)=C1O PKUDTZBFZDFCHN-UHFFFAOYSA-N 0.000 description 2
- RTVPERWIBHAYIR-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)benzene-1,3-diol Chemical compound N1=C(C)C=C(C)N1C1=C(O)C=CC=C1O RTVPERWIBHAYIR-UHFFFAOYSA-N 0.000 description 2
- QVWXUXQPKDFHAT-UHFFFAOYSA-N 2-(4-chloro-3,5-dimethylpyrazol-1-yl)phenol Chemical compound CC1=C(Cl)C(C)=NN1C1=CC=CC=C1O QVWXUXQPKDFHAT-UHFFFAOYSA-N 0.000 description 2
- YLMNNMAFFFUBJB-UHFFFAOYSA-N 2-(4-fluoro-3,5-dimethylpyrazol-1-yl)phenol Chemical compound CC1=C(F)C(C)=NN1C1=CC=CC=C1O YLMNNMAFFFUBJB-UHFFFAOYSA-N 0.000 description 2
- DOPJTDJKZNWLRB-UHFFFAOYSA-N 2-Amino-5-nitrophenol Chemical compound NC1=CC=C([N+]([O-])=O)C=C1O DOPJTDJKZNWLRB-UHFFFAOYSA-N 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- CIGLUVZPXQKUQW-UHFFFAOYSA-N 2-[3,5-bis(trifluoromethyl)pyrazol-1-yl]phenol Chemical compound OC1=CC=CC=C1N1C(C(F)(F)F)=CC(C(F)(F)F)=N1 CIGLUVZPXQKUQW-UHFFFAOYSA-N 0.000 description 2
- QDGJHZNOQSIFAT-UHFFFAOYSA-N 2-amino-4-chloro-5-methylphenol Chemical compound CC1=CC(O)=C(N)C=C1Cl QDGJHZNOQSIFAT-UHFFFAOYSA-N 0.000 description 2
- ULDFRPKVIZMKJG-UHFFFAOYSA-N 2-amino-4-fluorophenol Chemical compound NC1=CC(F)=CC=C1O ULDFRPKVIZMKJG-UHFFFAOYSA-N 0.000 description 2
- FZCQMIRJCGWWCL-UHFFFAOYSA-N 2-amino-5-chlorophenol Chemical compound NC1=CC=C(Cl)C=C1O FZCQMIRJCGWWCL-UHFFFAOYSA-N 0.000 description 2
- 125000004204 2-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C(OC([H])([H])[H])C([H])=C1[H] 0.000 description 2
- BSKHPKMHTQYZBB-UHFFFAOYSA-N 2-methylpyridine Chemical class CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 2
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 2
- JKWMDHTWYVYZBV-UHFFFAOYSA-N 3-(3,5-dimethylpyrazol-1-yl)benzene-1,2-diol Chemical compound N1=C(C)C=C(C)N1C1=CC=CC(O)=C1O JKWMDHTWYVYZBV-UHFFFAOYSA-N 0.000 description 2
- DVLFYONBTKHTER-UHFFFAOYSA-N 3-(N-morpholino)propanesulfonic acid Chemical compound OS(=O)(=O)CCCN1CCOCC1 DVLFYONBTKHTER-UHFFFAOYSA-N 0.000 description 2
- GNCDAHQSFQRXFZ-UHFFFAOYSA-N 3-[1-(2-hydroxyphenyl)-3,5-dimethylpyrazol-4-yl]propanoic acid Chemical compound CC1=C(CCC(O)=O)C(C)=NN1C1=CC=CC=C1O GNCDAHQSFQRXFZ-UHFFFAOYSA-N 0.000 description 2
- AUCCYVILTRXXKC-UHFFFAOYSA-N 3-amino-2-(3,5-dimethylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=C(C)N1C1=C(N)C=CC=C1O AUCCYVILTRXXKC-UHFFFAOYSA-N 0.000 description 2
- IJFXRHURBJZNAO-UHFFFAOYSA-N 3-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=CC(O)=C1 IJFXRHURBJZNAO-UHFFFAOYSA-N 0.000 description 2
- PJCCSZUMZMCWSX-UHFFFAOYSA-N 4,4-Dimethoxy-2-butanone Chemical compound COC(OC)CC(C)=O PJCCSZUMZMCWSX-UHFFFAOYSA-N 0.000 description 2
- FIGZNYYQCDEDJQ-UHFFFAOYSA-N 4-(3,5-dimethylpyrazol-1-yl)-3-hydroxy-n,n-dimethylbenzamide Chemical compound OC1=CC(C(=O)N(C)C)=CC=C1N1C(C)=CC(C)=N1 FIGZNYYQCDEDJQ-UHFFFAOYSA-N 0.000 description 2
- JQNDLHCBFCODNT-UHFFFAOYSA-N 4-(4-chloro-3,5-dimethylpyrazol-1-yl)benzene-1,3-diol Chemical compound CC1=C(Cl)C(C)=NN1C1=CC=C(O)C=C1O JQNDLHCBFCODNT-UHFFFAOYSA-N 0.000 description 2
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 2
- DMVSQFJOIKGKGJ-UHFFFAOYSA-N 4-chloro-2-(3,5-dimethylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC(Cl)=CC=C1O DMVSQFJOIKGKGJ-UHFFFAOYSA-N 0.000 description 2
- HIQIXEFWDLTDED-UHFFFAOYSA-N 4-hydroxy-1-piperidin-4-ylpyrrolidin-2-one Chemical compound O=C1CC(O)CN1C1CCNCC1 HIQIXEFWDLTDED-UHFFFAOYSA-N 0.000 description 2
- YUBURECKQXVYLX-UHFFFAOYSA-N 4-methoxy-5-methyl-2-nitrophenol Chemical compound COC1=CC([N+]([O-])=O)=C(O)C=C1C YUBURECKQXVYLX-UHFFFAOYSA-N 0.000 description 2
- QRGWBPZHRLREJH-UHFFFAOYSA-N 5-bromo-2-(3,5-dimethylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(Br)C=C1O QRGWBPZHRLREJH-UHFFFAOYSA-N 0.000 description 2
- YAQJFKVJXGQPHX-UHFFFAOYSA-N 5-chloro-2-(3,4,5-trimethylpyrazol-1-yl)phenol Chemical compound CC1=C(C)C(C)=NN1C1=CC=C(Cl)C=C1O YAQJFKVJXGQPHX-UHFFFAOYSA-N 0.000 description 2
- RRTSNQSOAYQWBV-UHFFFAOYSA-N 5-chloro-2-(3,5-dimethylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(Cl)C=C1O RRTSNQSOAYQWBV-UHFFFAOYSA-N 0.000 description 2
- LRBGQZZXBXYJMQ-UHFFFAOYSA-N 5-nitro-2-(3,4,5-trimethylpyrazol-1-yl)phenol Chemical compound CC1=C(C)C(C)=NN1C1=CC=C([N+]([O-])=O)C=C1O LRBGQZZXBXYJMQ-UHFFFAOYSA-N 0.000 description 2
- KPZNCKLMOFGREF-UHFFFAOYSA-N 5-tert-butyl-2-(2-methoxyphenyl)pyrazol-3-amine Chemical compound COC1=CC=CC=C1N1C(N)=CC(C(C)(C)C)=N1 KPZNCKLMOFGREF-UHFFFAOYSA-N 0.000 description 2
- 239000005725 8-Hydroxyquinoline Substances 0.000 description 2
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 2
- 229920001817 Agar Polymers 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- 241001480043 Arthrodermataceae Species 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- SZCYTLPATIGFOZ-UHFFFAOYSA-N COC1=CC=CC=C1N1C(O)=CC(C)=N1 Chemical compound COC1=CC=CC=C1N1C(O)=CC(C)=N1 SZCYTLPATIGFOZ-UHFFFAOYSA-N 0.000 description 2
- 241000222120 Candida <Saccharomycetales> Species 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 102000011782 Keratins Human genes 0.000 description 2
- 108010076876 Keratins Proteins 0.000 description 2
- HSHXDCVZWHOWCS-UHFFFAOYSA-N N'-hexadecylthiophene-2-carbohydrazide Chemical compound CCCCCCCCCCCCCCCCNNC(=O)c1cccs1 HSHXDCVZWHOWCS-UHFFFAOYSA-N 0.000 description 2
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 2
- 239000005642 Oleic acid Substances 0.000 description 2
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N Propionic acid Chemical compound CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- ZTHYODDOHIVTJV-UHFFFAOYSA-N Propyl gallate Chemical compound CCCOC(=O)C1=CC(O)=C(O)C(O)=C1 ZTHYODDOHIVTJV-UHFFFAOYSA-N 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- 241000223229 Trichophyton rubrum Species 0.000 description 2
- 229910021536 Zeolite Inorganic materials 0.000 description 2
- GDEVITLTVZUCFT-UHFFFAOYSA-N [2-(3,5-dimethylpyrazol-1-yl)-5-ethylphenyl] methanesulfonate Chemical compound CS(=O)(=O)OC1=CC(CC)=CC=C1N1C(C)=CC(C)=N1 GDEVITLTVZUCFT-UHFFFAOYSA-N 0.000 description 2
- YLWUPLHXOSZKEE-UHFFFAOYSA-N [3-hydroxy-4-(3,4,5-trimethylpyrazol-1-yl)phenyl] acetate Chemical compound OC1=CC(OC(=O)C)=CC=C1N1C(C)=C(C)C(C)=N1 YLWUPLHXOSZKEE-UHFFFAOYSA-N 0.000 description 2
- GKZKBOUVACTIJJ-UHFFFAOYSA-N [4-(4-chloro-3,5-dimethylpyrazol-1-yl)-3-hydroxyphenyl] acetate Chemical compound OC1=CC(OC(=O)C)=CC=C1N1C(C)=C(Cl)C(C)=N1 GKZKBOUVACTIJJ-UHFFFAOYSA-N 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 150000001242 acetic acid derivatives Chemical class 0.000 description 2
- GRFKUEDWLAGOJX-UHFFFAOYSA-N acetic acid;butyl acetate Chemical class CC(O)=O.CCCCOC(C)=O GRFKUEDWLAGOJX-UHFFFAOYSA-N 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- 239000008272 agar Substances 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 229940024606 amino acid Drugs 0.000 description 2
- 229960003204 amorolfine Drugs 0.000 description 2
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 2
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 229940077388 benzenesulfonate Drugs 0.000 description 2
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 2
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 235000019437 butane-1,3-diol Nutrition 0.000 description 2
- CZBZUDVBLSSABA-UHFFFAOYSA-N butylated hydroxyanisole Chemical compound COC1=CC=C(O)C(C(C)(C)C)=C1.COC1=CC=C(O)C=C1C(C)(C)C CZBZUDVBLSSABA-UHFFFAOYSA-N 0.000 description 2
- 235000010354 butylated hydroxytoluene Nutrition 0.000 description 2
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 2
- 229910000024 caesium carbonate Inorganic materials 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- SCKYRAXSEDYPSA-UHFFFAOYSA-N ciclopirox Chemical compound ON1C(=O)C=C(C)C=C1C1CCCCC1 SCKYRAXSEDYPSA-UHFFFAOYSA-N 0.000 description 2
- 235000015165 citric acid Nutrition 0.000 description 2
- 238000013329 compounding Methods 0.000 description 2
- 238000012790 confirmation Methods 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- OPQARKPSCNTWTJ-UHFFFAOYSA-L copper(ii) acetate Chemical compound [Cu+2].CC([O-])=O.CC([O-])=O OPQARKPSCNTWTJ-UHFFFAOYSA-L 0.000 description 2
- SSJXIUAHEKJCMH-UHFFFAOYSA-N cyclohexane-1,2-diamine Chemical compound NC1CCCCC1N SSJXIUAHEKJCMH-UHFFFAOYSA-N 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 230000037304 dermatophytes Effects 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- DOIRQSBPFJWKBE-UHFFFAOYSA-N dibutyl phthalate Chemical compound CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 description 2
- FLKPEMZONWLCSK-UHFFFAOYSA-N diethyl phthalate Chemical compound CCOC(=O)C1=CC=CC=C1C(=O)OCC FLKPEMZONWLCSK-UHFFFAOYSA-N 0.000 description 2
- XXJWXESWEXIICW-UHFFFAOYSA-N diethylene glycol monoethyl ether Chemical compound CCOCCOCCO XXJWXESWEXIICW-UHFFFAOYSA-N 0.000 description 2
- 108700003601 dimethylglycine Proteins 0.000 description 2
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 2
- 231100000676 disease causative agent Toxicity 0.000 description 2
- GRWZHXKQBITJKP-UHFFFAOYSA-L dithionite(2-) Chemical compound [O-]S(=O)S([O-])=O GRWZHXKQBITJKP-UHFFFAOYSA-L 0.000 description 2
- 239000003623 enhancer Substances 0.000 description 2
- RRAFCDWBNXTKKO-UHFFFAOYSA-N eugenol Chemical compound COC1=CC(CC=C)=CC=C1O RRAFCDWBNXTKKO-UHFFFAOYSA-N 0.000 description 2
- 150000004665 fatty acids Chemical class 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 2
- 230000009036 growth inhibition Effects 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- 238000005984 hydrogenation reaction Methods 0.000 description 2
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 2
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 2
- 229960003943 hypromellose Drugs 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- OWFXIOWLTKNBAP-UHFFFAOYSA-N isoamyl nitrite Chemical compound CC(C)CCON=O OWFXIOWLTKNBAP-UHFFFAOYSA-N 0.000 description 2
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 235000014655 lactic acid Nutrition 0.000 description 2
- 239000004310 lactic acid Substances 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 239000011976 maleic acid Substances 0.000 description 2
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 2
- 229910052987 metal hydride Inorganic materials 0.000 description 2
- 150000004681 metal hydrides Chemical class 0.000 description 2
- 229940098779 methanesulfonic acid Drugs 0.000 description 2
- FWRRTOSKCLOXCW-UHFFFAOYSA-N methyl 3-[1-(2-hydroxyphenyl)-3,5-dimethylpyrazol-4-yl]propanoate Chemical compound CC1=C(CCC(=O)OC)C(C)=NN1C1=CC=CC=C1O FWRRTOSKCLOXCW-UHFFFAOYSA-N 0.000 description 2
- DUJJYTLFGXXUOT-UHFFFAOYSA-N methyl 4-(4-chloro-3,5-dimethylpyrazol-1-yl)-3-hydroxybenzoate Chemical compound OC1=CC(C(=O)OC)=CC=C1N1C(C)=C(Cl)C(C)=N1 DUJJYTLFGXXUOT-UHFFFAOYSA-N 0.000 description 2
- CZXGXYBOQYQXQD-UHFFFAOYSA-N methyl benzenesulfonate Chemical compound COS(=O)(=O)C1=CC=CC=C1 CZXGXYBOQYQXQD-UHFFFAOYSA-N 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 229940078490 n,n-dimethylglycine Drugs 0.000 description 2
- 229910017604 nitric acid Inorganic materials 0.000 description 2
- 150000002826 nitrites Chemical class 0.000 description 2
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- KJIFKLIQANRMOU-UHFFFAOYSA-N oxidanium;4-methylbenzenesulfonate Chemical compound O.CC1=CC=C(S(O)(=O)=O)C=C1 KJIFKLIQANRMOU-UHFFFAOYSA-N 0.000 description 2
- 229960003540 oxyquinoline Drugs 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 2
- 244000052769 pathogen Species 0.000 description 2
- 230000035515 penetration Effects 0.000 description 2
- 150000002989 phenols Chemical class 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 229940068984 polyvinyl alcohol Drugs 0.000 description 2
- 229910000160 potassium phosphate Inorganic materials 0.000 description 2
- 235000011009 potassium phosphates Nutrition 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- MCJGNVYPOGVAJF-UHFFFAOYSA-N quinolin-8-ol Chemical compound C1=CN=C2C(O)=CC=CC2=C1 MCJGNVYPOGVAJF-UHFFFAOYSA-N 0.000 description 2
- 238000007363 ring formation reaction Methods 0.000 description 2
- ZMQAAUBTXCXRIC-UHFFFAOYSA-N safrole Chemical compound C=CCC1=CC=C2OCOC2=C1 ZMQAAUBTXCXRIC-UHFFFAOYSA-N 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- FSYKKLYZXJSNPZ-UHFFFAOYSA-N sarcosine Chemical compound C[NH2+]CC([O-])=O FSYKKLYZXJSNPZ-UHFFFAOYSA-N 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 2
- 235000017557 sodium bicarbonate Nutrition 0.000 description 2
- 239000012279 sodium borohydride Substances 0.000 description 2
- 229910000033 sodium borohydride Inorganic materials 0.000 description 2
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- IOGXOCVLYRDXLW-UHFFFAOYSA-N tert-butyl nitrite Chemical compound CC(C)(C)ON=O IOGXOCVLYRDXLW-UHFFFAOYSA-N 0.000 description 2
- 239000012414 tert-butyl nitrite Substances 0.000 description 2
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- MGSRCZKZVOBKFT-UHFFFAOYSA-N thymol Chemical compound CC(C)C1=CC=C(C)C=C1O MGSRCZKZVOBKFT-UHFFFAOYSA-N 0.000 description 2
- FWPIDFUJEMBDLS-UHFFFAOYSA-L tin(II) chloride dihydrate Chemical compound O.O.Cl[Sn]Cl FWPIDFUJEMBDLS-UHFFFAOYSA-L 0.000 description 2
- 229920002554 vinyl polymer Polymers 0.000 description 2
- 239000010457 zeolite Substances 0.000 description 2
- NUXPNWCDWYQEOW-UHFFFAOYSA-N (2-amino-5-ethylphenyl) methanesulfonate Chemical compound CCC1=CC=C(N)C(OS(C)(=O)=O)=C1 NUXPNWCDWYQEOW-UHFFFAOYSA-N 0.000 description 1
- ARASIKCXYJHRPJ-UHFFFAOYSA-N (2-amino-5-methoxyphenyl) 4-methylbenzenesulfonate Chemical compound COC1=CC=C(N)C(OS(=O)(=O)C=2C=CC(C)=CC=2)=C1 ARASIKCXYJHRPJ-UHFFFAOYSA-N 0.000 description 1
- LFGPRAKKKFAMMK-UHFFFAOYSA-N (2-amino-5-methylphenyl) 4-methylbenzenesulfonate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)OC1=CC(C)=CC=C1N LFGPRAKKKFAMMK-UHFFFAOYSA-N 0.000 description 1
- VNGFIALEVLELLC-UHFFFAOYSA-N (2-amino-5-methylphenyl) 4-methylbenzenesulfonate [2-(3,5-dimethylpyrazol-1-yl)-5-methylphenyl] 4-methylbenzenesulfonate Chemical compound Cc1ccc(cc1)S(=O)(=O)Oc1cc(C)ccc1N.Cc1cc(C)n(n1)-c1ccc(C)cc1OS(=O)(=O)c1ccc(C)cc1 VNGFIALEVLELLC-UHFFFAOYSA-N 0.000 description 1
- KIILXXHXARRJTE-UHFFFAOYSA-N (2-amino-6-methylphenyl) 4-methylbenzenesulfonate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)OC1=C(C)C=CC=C1N KIILXXHXARRJTE-UHFFFAOYSA-N 0.000 description 1
- KZRIXZCUNKWRDF-UHFFFAOYSA-N (2-phenylmethoxyphenyl)hydrazine;hydrochloride Chemical compound Cl.NNC1=CC=CC=C1OCC1=CC=CC=C1 KZRIXZCUNKWRDF-UHFFFAOYSA-N 0.000 description 1
- CUNWUEBNSZSNRX-RKGWDQTMSA-N (2r,3r,4r,5s)-hexane-1,2,3,4,5,6-hexol;(z)-octadec-9-enoic acid Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO.OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO.CCCCCCCC\C=C/CCCCCCCC(O)=O.CCCCCCCC\C=C/CCCCCCCC(O)=O.CCCCCCCC\C=C/CCCCCCCC(O)=O CUNWUEBNSZSNRX-RKGWDQTMSA-N 0.000 description 1
- VIKTUZZYGHKELS-UHFFFAOYSA-N (3-methylphenyl) 4-methylbenzenesulfonate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)OC1=CC=CC(C)=C1 VIKTUZZYGHKELS-UHFFFAOYSA-N 0.000 description 1
- VCJOGQVFTRGPKU-UHFFFAOYSA-N (4-chloro-5-ethyl-2-nitrophenyl) methanesulfonate Chemical compound CCC1=CC(OS(C)(=O)=O)=C([N+]([O-])=O)C=C1Cl VCJOGQVFTRGPKU-UHFFFAOYSA-N 0.000 description 1
- DMGRLHIAFWMKOY-UHFFFAOYSA-N (4-methoxy-5-methyl-2-nitrophenyl) 4-methylbenzenesulfonate Chemical compound C1=C(C)C(OC)=CC([N+]([O-])=O)=C1OS(=O)(=O)C1=CC=C(C)C=C1 DMGRLHIAFWMKOY-UHFFFAOYSA-N 0.000 description 1
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 1
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- 150000005045 1,10-phenanthrolines Chemical class 0.000 description 1
- SSUJUUNLZQVZMO-UHFFFAOYSA-N 1,2,3,4,8,9,10,10a-octahydropyrimido[1,2-a]azepine Chemical compound C1CCC=CN2CCCNC21 SSUJUUNLZQVZMO-UHFFFAOYSA-N 0.000 description 1
- WERYXYBDKMZEQL-UHFFFAOYSA-N 1,4-butanediol Substances OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 1
- IQISOVKPFBLQIQ-UHFFFAOYSA-N 1,4-dimethoxy-2-methylbenzene Chemical compound COC1=CC=C(OC)C(C)=C1 IQISOVKPFBLQIQ-UHFFFAOYSA-N 0.000 description 1
- RDNRQAZPLGCWFD-UHFFFAOYSA-N 1-(2,4-dimethoxyphenyl)-3,5-dimethylpyrazole Chemical compound COC1=CC(OC)=CC=C1N1C(C)=CC(C)=N1 RDNRQAZPLGCWFD-UHFFFAOYSA-N 0.000 description 1
- WYBTUEZSNBFYFN-UHFFFAOYSA-N 1-(2-methoxyphenyl)-3,4,5-trimethylpyrazole Chemical compound COC1=CC=CC=C1N1C(C)=C(C)C(C)=N1 WYBTUEZSNBFYFN-UHFFFAOYSA-N 0.000 description 1
- VQUKSDWAQISZLV-UHFFFAOYSA-N 1-(2-methoxyphenyl)-3,5-bis(trifluoromethyl)pyrazole Chemical compound COC1=CC=CC=C1N1C(C(F)(F)F)=CC(C(F)(F)F)=N1 VQUKSDWAQISZLV-UHFFFAOYSA-N 0.000 description 1
- CDXJFVKAOUJEPO-UHFFFAOYSA-N 1-(2-methoxyphenyl)-3-methylpyrazole Chemical compound COC1=CC=CC=C1N1N=C(C)C=C1 CDXJFVKAOUJEPO-UHFFFAOYSA-N 0.000 description 1
- UJCAVMHKIOQENR-UHFFFAOYSA-N 1-(2-methoxyphenyl)-5-methyl-3-(trifluoromethyl)pyrazole Chemical compound COC1=CC=CC=C1N1C(C)=CC(C(F)(F)F)=N1 UJCAVMHKIOQENR-UHFFFAOYSA-N 0.000 description 1
- JUJWEFGZUFSRPH-UHFFFAOYSA-N 1-(2-methoxyphenyl)-5-methyl-3-(trifluoromethyl)pyrazole 2-[5-methyl-3-(trifluoromethyl)pyrazol-1-yl]phenol Chemical compound COC1=C(C=CC=C1)N1N=C(C=C1C)C(F)(F)F.CC1=CC(=NN1C1=C(C=CC=C1)O)C(F)(F)F JUJWEFGZUFSRPH-UHFFFAOYSA-N 0.000 description 1
- XABBCVPCXVSWNG-UHFFFAOYSA-N 1-(2-methoxyphenyl)-5-methylpyrazole Chemical compound COC1=CC=CC=C1N1C(C)=CC=N1 XABBCVPCXVSWNG-UHFFFAOYSA-N 0.000 description 1
- ASOKPJOREAFHNY-UHFFFAOYSA-N 1-Hydroxybenzotriazole Chemical compound C1=CC=C2N(O)N=NC2=C1 ASOKPJOREAFHNY-UHFFFAOYSA-N 0.000 description 1
- NIUZVSQOXJIHBL-UHFFFAOYSA-N 1-bromo-2,4-dimethoxybenzene Chemical compound COC1=CC=C(Br)C(OC)=C1 NIUZVSQOXJIHBL-UHFFFAOYSA-N 0.000 description 1
- ARIWANIATODDMH-AWEZNQCLSA-N 1-lauroyl-sn-glycerol Chemical compound CCCCCCCCCCCC(=O)OC[C@@H](O)CO ARIWANIATODDMH-AWEZNQCLSA-N 0.000 description 1
- NZJXADCEESMBPW-UHFFFAOYSA-N 1-methylsulfinyldecane Chemical compound CCCCCCCCCCS(C)=O NZJXADCEESMBPW-UHFFFAOYSA-N 0.000 description 1
- SPSPIUSUWPLVKD-UHFFFAOYSA-N 2,3-dibutyl-6-methylphenol Chemical compound CCCCC1=CC=C(C)C(O)=C1CCCC SPSPIUSUWPLVKD-UHFFFAOYSA-N 0.000 description 1
- CYEZXDVLBGFROE-UHFFFAOYSA-N 2,4-Dihydroxy-nitrophenol Chemical compound OC1=CC=C([N+]([O-])=O)C(O)=C1 CYEZXDVLBGFROE-UHFFFAOYSA-N 0.000 description 1
- NAZDVUBIEPVUKE-UHFFFAOYSA-N 2,5-dimethoxyaniline Chemical compound COC1=CC=C(OC)C(N)=C1 NAZDVUBIEPVUKE-UHFFFAOYSA-N 0.000 description 1
- MGZISDFFGSWQIN-UHFFFAOYSA-N 2-(3,5-dimethylpyrazol-1-yl)-5-(hydroxymethyl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(CO)C=C1O MGZISDFFGSWQIN-UHFFFAOYSA-N 0.000 description 1
- MOAXLQJATRVKPD-UHFFFAOYSA-N 2-(4-chloro-3,5-dimethylpyrazol-1-yl)-5-fluorophenol Chemical compound CC1=C(Cl)C(C)=NN1C1=CC=C(F)C=C1O MOAXLQJATRVKPD-UHFFFAOYSA-N 0.000 description 1
- JOSJQBSKIIBMKJ-UHFFFAOYSA-N 2-(5-amino-3-tert-butylpyrazol-1-yl)-5-methylphenol Chemical compound OC1=CC(C)=CC=C1N1C(N)=CC(C(C)(C)C)=N1 JOSJQBSKIIBMKJ-UHFFFAOYSA-N 0.000 description 1
- VHVPQPYKVGDNFY-DFMJLFEVSA-N 2-[(2r)-butan-2-yl]-4-[4-[4-[4-[[(2r,4s)-2-(2,4-dichlorophenyl)-2-(1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-1,2,4-triazol-3-one Chemical compound O=C1N([C@H](C)CC)N=CN1C1=CC=C(N2CCN(CC2)C=2C=CC(OC[C@@H]3O[C@](CN4N=CN=C4)(OC3)C=3C(=CC(Cl)=CC=3)Cl)=CC=2)C=C1 VHVPQPYKVGDNFY-DFMJLFEVSA-N 0.000 description 1
- FOHZVKOJJQALSO-UHFFFAOYSA-N 2-[5-methyl-3-(trifluoromethyl)pyrazol-1-yl]phenol Chemical compound CC1=CC(C(F)(F)F)=NN1C1=CC=CC=C1O FOHZVKOJJQALSO-UHFFFAOYSA-N 0.000 description 1
- FEDLEBCVFZMHBP-UHFFFAOYSA-N 2-amino-3-methylphenol Chemical compound CC1=CC=CC(O)=C1N FEDLEBCVFZMHBP-UHFFFAOYSA-N 0.000 description 1
- KUCWUAFNGCMZDB-UHFFFAOYSA-N 2-amino-3-nitrophenol Chemical compound NC1=C(O)C=CC=C1[N+]([O-])=O KUCWUAFNGCMZDB-UHFFFAOYSA-N 0.000 description 1
- JEASLLCHQHBBGM-UHFFFAOYSA-N 2-amino-4,5-dimethylphenol Chemical compound CC1=CC(N)=C(O)C=C1C JEASLLCHQHBBGM-UHFFFAOYSA-N 0.000 description 1
- MZMUKGFYWNOPAU-UHFFFAOYSA-N 2-amino-4-methylphenol;hydron;chloride Chemical compound Cl.CC1=CC=C(O)C(N)=C1 MZMUKGFYWNOPAU-UHFFFAOYSA-N 0.000 description 1
- DRQWUAAWZFIVTF-UHFFFAOYSA-N 2-amino-5-bromophenol Chemical compound NC1=CC=C(Br)C=C1O DRQWUAAWZFIVTF-UHFFFAOYSA-N 0.000 description 1
- HCPJEHJGFKWRFM-UHFFFAOYSA-N 2-amino-5-methylphenol Chemical compound CC1=CC=C(N)C(O)=C1 HCPJEHJGFKWRFM-UHFFFAOYSA-N 0.000 description 1
- TYBHCHHFJWXIHS-UHFFFAOYSA-N 2-amino-6-methylphenol;hydrochloride Chemical compound Cl.CC1=CC=CC(N)=C1O TYBHCHHFJWXIHS-UHFFFAOYSA-N 0.000 description 1
- VZOZMRMQWMPBIX-UHFFFAOYSA-N 2-aminobenzene-1,3-diol 2-nitrobenzene-1,3-diol Chemical compound NC1=C(O)C=CC=C1O.[N+](=O)([O-])C1=C(O)C=CC=C1O VZOZMRMQWMPBIX-UHFFFAOYSA-N 0.000 description 1
- 125000000143 2-carboxyethyl group Chemical group [H]OC(=O)C([H])([H])C([H])([H])* 0.000 description 1
- CHPCQZSIOGSYBI-UHFFFAOYSA-N 2-methoxy-3,5-dimethyl-4-[2-(trifluoromethyl)phenyl]-1,3-dihydropyrazole Chemical compound CC1N(OC)NC(C)=C1C1=CC=CC=C1C(F)(F)F CHPCQZSIOGSYBI-UHFFFAOYSA-N 0.000 description 1
- LWKZYZZCYQWRTJ-UHFFFAOYSA-N 2-methoxy-4-(trifluoromethyl)aniline Chemical compound COC1=CC(C(F)(F)F)=CC=C1N LWKZYZZCYQWRTJ-UHFFFAOYSA-N 0.000 description 1
- GVBHRNIWBGTNQA-UHFFFAOYSA-N 2-methoxy-4-nitroaniline Chemical compound COC1=CC([N+]([O-])=O)=CC=C1N GVBHRNIWBGTNQA-UHFFFAOYSA-N 0.000 description 1
- 229940080296 2-naphthalenesulfonate Drugs 0.000 description 1
- LEACJMVNYZDSKR-UHFFFAOYSA-N 2-octyldodecan-1-ol Chemical compound CCCCCCCCCCC(CO)CCCCCCCC LEACJMVNYZDSKR-UHFFFAOYSA-N 0.000 description 1
- BGRXBNZMPMGLQI-UHFFFAOYSA-N 2-octyldodecyl tetradecanoate Chemical compound CCCCCCCCCCCCCC(=O)OCC(CCCCCCCC)CCCCCCCCCC BGRXBNZMPMGLQI-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- LUENEMPMZFQNMJ-UHFFFAOYSA-N 3,5-diethyl-1-(2-methoxyphenyl)pyrazole Chemical compound N1=C(CC)C=C(CC)N1C1=CC=CC=C1OC LUENEMPMZFQNMJ-UHFFFAOYSA-N 0.000 description 1
- SDXAWLJRERMRKF-UHFFFAOYSA-N 3,5-dimethyl-1h-pyrazole Chemical compound CC=1C=C(C)NN=1 SDXAWLJRERMRKF-UHFFFAOYSA-N 0.000 description 1
- FPQQSJJWHUJYPU-UHFFFAOYSA-N 3-(dimethylamino)propyliminomethylidene-ethylazanium;chloride Chemical compound Cl.CCN=C=NCCCN(C)C FPQQSJJWHUJYPU-UHFFFAOYSA-N 0.000 description 1
- MBXOOYPCIDHXGH-UHFFFAOYSA-N 3-butylpentane-2,4-dione Chemical compound CCCCC(C(C)=O)C(C)=O MBXOOYPCIDHXGH-UHFFFAOYSA-N 0.000 description 1
- VPZJHTWLWKFPQW-UHFFFAOYSA-N 3-chloro-2-methoxyaniline Chemical compound COC1=C(N)C=CC=C1Cl VPZJHTWLWKFPQW-UHFFFAOYSA-N 0.000 description 1
- GUARKOVVHJSMRW-UHFFFAOYSA-N 3-ethylpentane-2,4-dione Chemical compound CCC(C(C)=O)C(C)=O GUARKOVVHJSMRW-UHFFFAOYSA-N 0.000 description 1
- LELFVORHOKZNGY-UHFFFAOYSA-N 3-fluoropentane-2,4-dione Chemical compound CC(=O)C(F)C(C)=O LELFVORHOKZNGY-UHFFFAOYSA-N 0.000 description 1
- AQGSZYZZVTYOMQ-UHFFFAOYSA-N 3-propylpentane-2,4-dione Chemical compound CCCC(C(C)=O)C(C)=O AQGSZYZZVTYOMQ-UHFFFAOYSA-N 0.000 description 1
- MXZMACXOMZKYHJ-UHFFFAOYSA-N 4,4-dimethyl-3-oxopentanenitrile Chemical compound CC(C)(C)C(=O)CC#N MXZMACXOMZKYHJ-UHFFFAOYSA-N 0.000 description 1
- NSGVABONWZPMKU-UHFFFAOYSA-N 4-(3,5-dimethylpyrazol-1-yl)-3-hydroxybenzamide Chemical compound N1=C(C)C=C(C)N1C1=CC=C(C(N)=O)C=C1O NSGVABONWZPMKU-UHFFFAOYSA-N 0.000 description 1
- VLXZJPCJXSVHFR-UHFFFAOYSA-N 4-(3,5-dimethylpyrazol-1-yl)-3-hydroxybenzoic acid Chemical compound N1=C(C)C=C(C)N1C1=CC=C(C(O)=O)C=C1O VLXZJPCJXSVHFR-UHFFFAOYSA-N 0.000 description 1
- WBRALSDLFWUYFP-UHFFFAOYSA-N 4-(3,5-dimethylpyrazol-1-yl)-3-methoxyaniline Chemical compound COC1=CC(N)=CC=C1N1C(C)=CC(C)=N1 WBRALSDLFWUYFP-UHFFFAOYSA-N 0.000 description 1
- GHCJLLQDZSNIEN-UHFFFAOYSA-N 4-(3,5-dimethylpyrazol-1-yl)benzene-1,3-diol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(O)C=C1O GHCJLLQDZSNIEN-UHFFFAOYSA-N 0.000 description 1
- JVJVGXSDQAHUJV-UHFFFAOYSA-N 4-butyl-1-(2-methoxyphenyl)-3,5-dimethylpyrazole Chemical compound CC1=C(CCCC)C(C)=NN1C1=CC=CC=C1OC JVJVGXSDQAHUJV-UHFFFAOYSA-N 0.000 description 1
- CEZGCEFWOKTFAW-UHFFFAOYSA-N 4-chloro-1-(2-methoxyphenyl)-3,5-dimethylpyrazole Chemical compound COC1=CC=CC=C1N1C(C)=C(Cl)C(C)=N1 CEZGCEFWOKTFAW-UHFFFAOYSA-N 0.000 description 1
- RIZCFPOVCXLBME-UHFFFAOYSA-N 4-chloro-5-ethyl-2-nitrophenol Chemical compound CCC1=CC(O)=C([N+]([O-])=O)C=C1Cl RIZCFPOVCXLBME-UHFFFAOYSA-N 0.000 description 1
- JBMGJOKJUYGIJH-UHFFFAOYSA-N 4-chloro-5-methyl-2-nitrophenol Chemical compound CC1=CC(O)=C([N+]([O-])=O)C=C1Cl JBMGJOKJUYGIJH-UHFFFAOYSA-N 0.000 description 1
- HBTAOSGHCXUEKI-UHFFFAOYSA-N 4-chloro-n,n-dimethyl-3-nitrobenzenesulfonamide Chemical compound CN(C)S(=O)(=O)C1=CC=C(Cl)C([N+]([O-])=O)=C1 HBTAOSGHCXUEKI-UHFFFAOYSA-N 0.000 description 1
- ITIPQDUSULAMDA-UHFFFAOYSA-N 4-ethyl-1-(2-methoxyphenyl)-3,5-dimethylpyrazole Chemical compound CC1=C(CC)C(C)=NN1C1=CC=CC=C1OC ITIPQDUSULAMDA-UHFFFAOYSA-N 0.000 description 1
- YUKLVHZAMSJKAN-UHFFFAOYSA-N 4-fluoro-3,5-dimethyl-1-(2-phenylmethoxyphenyl)pyrazole Chemical compound CC1=C(F)C(C)=NN1C1=CC=CC=C1OCC1=CC=CC=C1 YUKLVHZAMSJKAN-UHFFFAOYSA-N 0.000 description 1
- ACGMPKMQHCHONI-UHFFFAOYSA-N 5-amino-2-(3,4,5-trimethylpyrazol-1-yl)phenol Chemical compound CC1=C(C)C(C)=NN1C1=CC=C(N)C=C1O ACGMPKMQHCHONI-UHFFFAOYSA-N 0.000 description 1
- NKSYEYBOCRUPRR-UHFFFAOYSA-N 5-amino-2-(3,5-dimethylpyrazol-1-yl)phenol Chemical compound N1=C(C)C=C(C)N1C1=CC=C(N)C=C1O NKSYEYBOCRUPRR-UHFFFAOYSA-N 0.000 description 1
- CHUHKTUWWOFHHJ-UHFFFAOYSA-N 5-bromo-2-(3,4,5-trimethylpyrazol-1-yl)phenol Chemical compound CC1=C(C)C(C)=NN1C1=CC=C(Br)C=C1O CHUHKTUWWOFHHJ-UHFFFAOYSA-N 0.000 description 1
- OTQRUQJXEUPVMS-UHFFFAOYSA-N 5-bromo-2-(4-chloro-3,5-dimethylpyrazol-1-yl)phenol Chemical compound CC1=C(Cl)C(C)=NN1C1=CC=C(Br)C=C1O OTQRUQJXEUPVMS-UHFFFAOYSA-N 0.000 description 1
- UFPHCCHSICYZDL-UHFFFAOYSA-N 5-bromo-2-hydrazinylphenol Chemical compound NNC1=CC=C(Br)C=C1O UFPHCCHSICYZDL-UHFFFAOYSA-N 0.000 description 1
- QQURWFRNETXFTN-UHFFFAOYSA-N 5-fluoro-2-nitrophenol Chemical compound OC1=CC(F)=CC=C1[N+]([O-])=O QQURWFRNETXFTN-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 201000002909 Aspergillosis Diseases 0.000 description 1
- 241000228212 Aspergillus Species 0.000 description 1
- 208000036641 Aspergillus infections Diseases 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical compound N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- YFGBIHMBXHFGCN-UHFFFAOYSA-N BrC=1C=CC(=C(C1)O)NN.BrC=1C=CC(=C(C1)O)N1N=C(C(=C1C)C)C Chemical compound BrC=1C=CC(=C(C1)O)NN.BrC=1C=CC(=C(C1)O)N1N=C(C(=C1C)C)C YFGBIHMBXHFGCN-UHFFFAOYSA-N 0.000 description 1
- OUFAREBVAXPNFB-UHFFFAOYSA-N BrC=1C=CC(=C(C1)O)NN.BrC=1C=CC(=C(C1)O)N1N=C(C(=C1C)Cl)C Chemical compound BrC=1C=CC(=C(C1)O)NN.BrC=1C=CC(=C(C1)O)N1N=C(C(=C1C)Cl)C OUFAREBVAXPNFB-UHFFFAOYSA-N 0.000 description 1
- CIUHQAIBISXSCG-UHFFFAOYSA-N BrC=1C=CC(=C(C1)O)NN.BrC=1C=CC(=C(C1)O)N1N=C(C=C1C)C Chemical compound BrC=1C=CC(=C(C1)O)NN.BrC=1C=CC(=C(C1)O)N1N=C(C=C1C)C CIUHQAIBISXSCG-UHFFFAOYSA-N 0.000 description 1
- JQJPBYFTQAANLE-UHFFFAOYSA-N Butyl nitrite Chemical compound CCCCON=O JQJPBYFTQAANLE-UHFFFAOYSA-N 0.000 description 1
- ZRSIFOUEEWNZHM-UHFFFAOYSA-N C(C1=CC=CC=C1)OC1=C(C=CC=C1)N1N=C(C(=C1C)F)C.FC=1C(=NN(C1C)C1=C(C=CC=C1)O)C Chemical compound C(C1=CC=CC=C1)OC1=C(C=CC=C1)N1N=C(C(=C1C)F)C.FC=1C(=NN(C1C)C1=C(C=CC=C1)O)C ZRSIFOUEEWNZHM-UHFFFAOYSA-N 0.000 description 1
- OFRIEHJEBRVQKU-UHFFFAOYSA-N CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)N.CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)N1N=C(C=C1C)C Chemical compound CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)N.CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)N1N=C(C=C1C)C OFRIEHJEBRVQKU-UHFFFAOYSA-N 0.000 description 1
- YJOUALGGLDXCLR-UHFFFAOYSA-N CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)[N+](=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)N Chemical compound CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)[N+](=O)[O-].CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=C(C(=C1)C)OC)N YJOUALGGLDXCLR-UHFFFAOYSA-N 0.000 description 1
- LDASUJZTCBKXGP-UHFFFAOYSA-N CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=CC=C1C)N.CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=CC=C1C)N1N=C(C=C1C)C Chemical compound CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=CC=C1C)N.CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C=CC=C1C)N1N=C(C=C1C)C LDASUJZTCBKXGP-UHFFFAOYSA-N 0.000 description 1
- TVWPTIQPCNSTHF-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C(=CC=C1)C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C(=CC=C1)C)O Chemical compound CC1=NN(C(=C1)C)C1=C(C(=CC=C1)C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C(=CC=C1)C)O TVWPTIQPCNSTHF-UHFFFAOYSA-N 0.000 description 1
- SCQYHLJCPFQKMF-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O.CC1=NN(C(=C1)C)C1=C(C=C(C(=O)N)C=C1)O Chemical compound CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O.CC1=NN(C(=C1)C)C1=C(C=C(C(=O)N)C=C1)O SCQYHLJCPFQKMF-UHFFFAOYSA-N 0.000 description 1
- CFEKXOOYALEGGG-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O Chemical compound CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O CFEKXOOYALEGGG-UHFFFAOYSA-N 0.000 description 1
- MBCIKTBCUOKTNP-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)CO)O Chemical compound CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(=O)O)O.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)CO)O MBCIKTBCUOKTNP-UHFFFAOYSA-N 0.000 description 1
- FFSANWRTGGDXQD-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C)O Chemical compound CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C)O FFSANWRTGGDXQD-UHFFFAOYSA-N 0.000 description 1
- YBCYIQRIIKUKHW-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=C(C=C1)OC)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)OC)O Chemical compound CC1=NN(C(=C1)C)C1=C(C=C(C=C1)OC)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)OC)O YBCYIQRIIKUKHW-UHFFFAOYSA-N 0.000 description 1
- UQAOSZUVGBYDAD-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=C(C=C1)[N+](=O)[O-])O.NC=1C=CC(=C(C1)O)N1N=C(C=C1C)C Chemical compound CC1=NN(C(=C1)C)C1=C(C=C(C=C1)[N+](=O)[O-])O.NC=1C=CC(=C(C1)O)N1N=C(C=C1C)C UQAOSZUVGBYDAD-UHFFFAOYSA-N 0.000 description 1
- SIDGKVYQJFCESH-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=C(N)C=C1)OC.CC1=NN(C(=C1)C)C1=C(C=C(NC)C=C1)OC Chemical compound CC1=NN(C(=C1)C)C1=C(C=C(N)C=C1)OC.CC1=NN(C(=C1)C)C1=C(C=C(NC)C=C1)OC SIDGKVYQJFCESH-UHFFFAOYSA-N 0.000 description 1
- IYFWNIMPPXCONS-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=CC(=C1)C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=CC(=C1)C)O Chemical compound CC1=NN(C(=C1)C)C1=C(C=CC(=C1)C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=CC(=C1)C)O IYFWNIMPPXCONS-UHFFFAOYSA-N 0.000 description 1
- XHVXXRFCQNVJDJ-UHFFFAOYSA-N CC1=NN(C(=C1)C)C1=C(C=CC=C1C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=CC=C1C)O Chemical compound CC1=NN(C(=C1)C)C1=C(C=CC=C1C)C1=CC=C(C=C1)C.CC1=NN(C(=C1)C)C1=C(C=CC=C1C)O XHVXXRFCQNVJDJ-UHFFFAOYSA-N 0.000 description 1
- RCMLSSOURYQLNC-UHFFFAOYSA-N COC1=C(C=CC(=C1)C(F)(F)F)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(F)(F)F)O Chemical compound COC1=C(C=CC(=C1)C(F)(F)F)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)C(F)(F)F)O RCMLSSOURYQLNC-UHFFFAOYSA-N 0.000 description 1
- YQRAHNUASUQFQU-UHFFFAOYSA-N COC1=C(C=CC(=C1)OC)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)O)O Chemical compound COC1=C(C=CC(=C1)OC)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C=C(C=C1)O)O YQRAHNUASUQFQU-UHFFFAOYSA-N 0.000 description 1
- RFSWZVFDUGOHRR-UHFFFAOYSA-N COC1=C(C=CC=C1)N1N=C(C(=C1C)C(=O)OCC)C.OC1=C(C=CC=C1)N1N=C(C(=C1C)C(=O)OCC)C Chemical compound COC1=C(C=CC=C1)N1N=C(C(=C1C)C(=O)OCC)C.OC1=C(C=CC=C1)N1N=C(C(=C1C)C(=O)OCC)C RFSWZVFDUGOHRR-UHFFFAOYSA-N 0.000 description 1
- BEAKTLVOJBJHHD-UHFFFAOYSA-N COC1=C(C=CC=C1)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C=CC=C1)O Chemical compound COC1=C(C=CC=C1)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C=CC=C1)O BEAKTLVOJBJHHD-UHFFFAOYSA-N 0.000 description 1
- GCECEHRVUVFBBV-UHFFFAOYSA-N COC1=C(C=CC=C1OC)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C(=CC=C1)O)O Chemical compound COC1=C(C=CC=C1OC)N1N=C(C=C1C)C.CC1=NN(C(=C1)C)C1=C(C(=CC=C1)O)O GCECEHRVUVFBBV-UHFFFAOYSA-N 0.000 description 1
- CAVHSYCWCFEYAI-UHFFFAOYSA-N COC1=C(N)C=C(C=C1)OC.ClC=1C(=NN(C1C)C1=C(C=CC(=C1)OC)OC)C Chemical compound COC1=C(N)C=C(C=C1)OC.ClC=1C(=NN(C1C)C1=C(C=CC(=C1)OC)OC)C CAVHSYCWCFEYAI-UHFFFAOYSA-N 0.000 description 1
- DRFGYZBUQWKKMY-UHFFFAOYSA-N COC1=C(N)C=CC=C1OC.COC1=C(C=CC=C1OC)N1N=C(C=C1C)C Chemical compound COC1=C(N)C=CC=C1OC.COC1=C(C=CC=C1OC)N1N=C(C=C1C)C DRFGYZBUQWKKMY-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- NPBVQXIMTZKSBA-UHFFFAOYSA-N Chavibetol Natural products COC1=CC=C(CC=C)C=C1O NPBVQXIMTZKSBA-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- MKUNZHYXSNZHAB-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.C(C)C1=NN(C(=C1)CC)C1=C(C=CC=C1)OC Chemical compound Cl.COC1=C(C=CC=C1)NN.C(C)C1=NN(C(=C1)CC)C1=C(C=CC=C1)OC MKUNZHYXSNZHAB-UHFFFAOYSA-N 0.000 description 1
- FUWJLPIEYFVIBJ-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.C(C)C=1C(=NN(C1C)C1=C(C=CC=C1)OC)C Chemical compound Cl.COC1=C(C=CC=C1)NN.C(C)C=1C(=NN(C1C)C1=C(C=CC=C1)OC)C FUWJLPIEYFVIBJ-UHFFFAOYSA-N 0.000 description 1
- FGVKWTWXQHXWRP-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.C(CCC)C=1C(=NN(C1C)C1=C(C=CC=C1)OC)C Chemical compound Cl.COC1=C(C=CC=C1)NN.C(CCC)C=1C(=NN(C1C)C1=C(C=CC=C1)OC)C FGVKWTWXQHXWRP-UHFFFAOYSA-N 0.000 description 1
- BBBKXIQFOMVCKS-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=C(C(=C1C)C)C Chemical compound Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=C(C(=C1C)C)C BBBKXIQFOMVCKS-UHFFFAOYSA-N 0.000 description 1
- IAQMFLQENLRLAX-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=C(C=C1)C Chemical compound Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=C(C=C1)C IAQMFLQENLRLAX-UHFFFAOYSA-N 0.000 description 1
- QKEXYOBLERDHBS-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=C(C=C1C)C Chemical compound Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=C(C=C1C)C QKEXYOBLERDHBS-UHFFFAOYSA-N 0.000 description 1
- OZNJXUZCMUCPJD-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=CC=C1C Chemical compound Cl.COC1=C(C=CC=C1)NN.COC1=C(C=CC=C1)N1N=CC=C1C OZNJXUZCMUCPJD-UHFFFAOYSA-N 0.000 description 1
- UYSSAVBJXFTDHD-UHFFFAOYSA-N Cl.COC1=C(C=CC=C1)NN.ClC=1C(=NN(C1C)C1=C(C=CC=C1)OC)C Chemical compound Cl.COC1=C(C=CC=C1)NN.ClC=1C(=NN(C1C)C1=C(C=CC=C1)OC)C UYSSAVBJXFTDHD-UHFFFAOYSA-N 0.000 description 1
- QCGWNSRNMNNXFX-UHFFFAOYSA-N Cl.ClC=1C=CC(=C(N)C1)OC.ClC=1C=CC(=C(C1)N1N=C(C=C1C)C)OC Chemical compound Cl.ClC=1C=CC(=C(N)C1)OC.ClC=1C=CC(=C(C1)N1N=C(C=C1C)C)OC QCGWNSRNMNNXFX-UHFFFAOYSA-N 0.000 description 1
- HEUAMBHMBLYYKP-UHFFFAOYSA-N ClC=1C=CC(=C(C1)N1N=C(C=C1C)C)OC.ClC1=CC(=C(C=C1)O)N1N=C(C=C1C)C Chemical compound ClC=1C=CC(=C(C1)N1N=C(C=C1C)C)OC.ClC1=CC(=C(C=C1)O)N1N=C(C=C1C)C HEUAMBHMBLYYKP-UHFFFAOYSA-N 0.000 description 1
- 201000007336 Cryptococcosis Diseases 0.000 description 1
- 241001337994 Cryptococcus <scale insect> Species 0.000 description 1
- 241000221204 Cryptococcus neoformans Species 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- ZAKOWWREFLAJOT-CEFNRUSXSA-N D-alpha-tocopherylacetate Chemical compound CC(=O)OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C ZAKOWWREFLAJOT-CEFNRUSXSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 208000007163 Dermatomycoses Diseases 0.000 description 1
- 206010012504 Dermatophytosis Diseases 0.000 description 1
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical class C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 1
- VIZORQUEIQEFRT-UHFFFAOYSA-N Diethyl adipate Chemical compound CCOC(=O)CCCCC(=O)OCC VIZORQUEIQEFRT-UHFFFAOYSA-N 0.000 description 1
- 206010013710 Drug interaction Diseases 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- IMROMDMJAWUWLK-UHFFFAOYSA-N Ethenol Chemical compound OC=C IMROMDMJAWUWLK-UHFFFAOYSA-N 0.000 description 1
- QQZWEECEMNQSTG-UHFFFAOYSA-N Ethyl nitrite Chemical compound CCON=O QQZWEECEMNQSTG-UHFFFAOYSA-N 0.000 description 1
- 229920003148 Eudragit® E polymer Polymers 0.000 description 1
- 229920003151 Eudragit® RL polymer Polymers 0.000 description 1
- 229920003152 Eudragit® RS polymer Polymers 0.000 description 1
- 239000005770 Eugenol Substances 0.000 description 1
- RJHWDLVZRBAHBT-UHFFFAOYSA-N FC1=CC(=C(C=C1)O)[N+](=O)[O-].NC1=C(C=CC(=C1)F)O Chemical compound FC1=CC(=C(C=C1)O)[N+](=O)[O-].NC1=C(C=CC(=C1)F)O RJHWDLVZRBAHBT-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- 229920003114 HPC-L Polymers 0.000 description 1
- 229920003115 HPC-SL Polymers 0.000 description 1
- 229920003116 HPC-SSL Polymers 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- BJIOGJUNALELMI-ONEGZZNKSA-N Isoeugenol Natural products COC1=CC(\C=C\C)=CC=C1O BJIOGJUNALELMI-ONEGZZNKSA-N 0.000 description 1
- VHVOLFRBFDOUSH-NSCUHMNNSA-N Isosafrole Chemical compound C\C=C\C1=CC=C2OCOC2=C1 VHVOLFRBFDOUSH-NSCUHMNNSA-N 0.000 description 1
- VHVOLFRBFDOUSH-UHFFFAOYSA-N Isosafrole Natural products CC=CC1=CC=C2OCOC2=C1 VHVOLFRBFDOUSH-UHFFFAOYSA-N 0.000 description 1
- PWKSKIMOESPYIA-BYPYZUCNSA-N L-N-acetyl-Cysteine Chemical compound CC(=O)N[C@@H](CS)C(O)=O PWKSKIMOESPYIA-BYPYZUCNSA-N 0.000 description 1
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical class NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 1
- QAQJMLQRFWZOBN-LAUBAEHRSA-N L-ascorbyl-6-palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](O)[C@H]1OC(=O)C(O)=C1O QAQJMLQRFWZOBN-LAUBAEHRSA-N 0.000 description 1
- 235000000072 L-ascorbyl-6-palmitate Nutrition 0.000 description 1
- 239000011786 L-ascorbyl-6-palmitate Substances 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical class NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- ARIWANIATODDMH-UHFFFAOYSA-N Lauric acid monoglyceride Natural products CCCCCCCCCCCC(=O)OCC(O)CO ARIWANIATODDMH-UHFFFAOYSA-N 0.000 description 1
- 206010067125 Liver injury Diseases 0.000 description 1
- WSMYVTOQOOLQHP-UHFFFAOYSA-N Malondialdehyde Chemical compound O=CCC=O WSMYVTOQOOLQHP-UHFFFAOYSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical class CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 1
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 1
- WRQNANDWMGAFTP-UHFFFAOYSA-N Methylacetoacetic acid Chemical compound COC(=O)CC(C)=O WRQNANDWMGAFTP-UHFFFAOYSA-N 0.000 description 1
- 241001460074 Microsporum distortum Species 0.000 description 1
- 241000187479 Mycobacterium tuberculosis Species 0.000 description 1
- 208000031888 Mycoses Diseases 0.000 description 1
- IVNCBCSRFZIRLK-UHFFFAOYSA-N NC1=C(C=C(C=C1)F)O.ClC=1C(=NN(C1C)C1=C(C=C(C=C1)F)O)C Chemical compound NC1=C(C=C(C=C1)F)O.ClC=1C(=NN(C1C)C1=C(C=C(C=C1)F)O)C IVNCBCSRFZIRLK-UHFFFAOYSA-N 0.000 description 1
- KYYLMUCKXQPKQV-UHFFFAOYSA-N NC1=C(C=CC=C1C1=C(C=CC(=C1)C)S(=O)(=O)O)C1=C(C=CC(=C1)C)S(=O)(=O)O.CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C(=CC=C1)O)N1N=C(C=C1C)C Chemical compound NC1=C(C=CC=C1C1=C(C=CC(=C1)C)S(=O)(=O)O)C1=C(C=CC(=C1)C)S(=O)(=O)O.CC1=CC=C(C=C1)S(=O)(=O)OC1=C(C(=CC=C1)O)N1N=C(C=C1C)C KYYLMUCKXQPKQV-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 1
- 229910021586 Nickel(II) chloride Inorganic materials 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- IOVCWXUNBOPUCH-UHFFFAOYSA-N Nitrous acid Chemical compound ON=O IOVCWXUNBOPUCH-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 229920002675 Polyoxyl Polymers 0.000 description 1
- UVMRYBDEERADNV-UHFFFAOYSA-N Pseudoeugenol Natural products COC1=CC(C(C)=C)=CC=C1O UVMRYBDEERADNV-UHFFFAOYSA-N 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 239000007868 Raney catalyst Substances 0.000 description 1
- 229910000564 Raney nickel Inorganic materials 0.000 description 1
- 108010077895 Sarcosine Proteins 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 239000005844 Thymol Substances 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical class OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- DTQVDTLACAAQTR-UHFFFAOYSA-M Trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-M 0.000 description 1
- 206010061418 Zygomycosis Diseases 0.000 description 1
- AZFNGPAYDKGCRB-XCPIVNJJSA-M [(1s,2s)-2-amino-1,2-diphenylethyl]-(4-methylphenyl)sulfonylazanide;chlororuthenium(1+);1-methyl-4-propan-2-ylbenzene Chemical compound [Ru+]Cl.CC(C)C1=CC=C(C)C=C1.C1=CC(C)=CC=C1S(=O)(=O)[N-][C@@H](C=1C=CC=CC=1)[C@@H](N)C1=CC=CC=C1 AZFNGPAYDKGCRB-XCPIVNJJSA-M 0.000 description 1
- SVIOXJUULGVHBD-UHFFFAOYSA-N [2-(3,5-dimethylpyrazol-1-yl)-3-methylphenyl] 4-methylbenzenesulfonate Chemical compound N1=C(C)C=C(C)N1C1=C(C)C=CC=C1OS(=O)(=O)C1=CC=C(C)C=C1 SVIOXJUULGVHBD-UHFFFAOYSA-N 0.000 description 1
- HHHSBDLLHBBPQI-UHFFFAOYSA-N [2-(3,5-dimethylpyrazol-1-yl)-4-methylphenyl] 4-methylbenzenesulfonate Chemical compound N1=C(C)C=C(C)N1C1=CC(C)=CC=C1OS(=O)(=O)C1=CC=C(C)C=C1 HHHSBDLLHBBPQI-UHFFFAOYSA-N 0.000 description 1
- IMOVWUXHLQRODN-UHFFFAOYSA-N [2-(3,5-dimethylpyrazol-1-yl)-5-methoxyphenyl] 4-methylbenzenesulfonate Chemical compound C=1C(OC)=CC=C(N2C(=CC(C)=N2)C)C=1OS(=O)(=O)C1=CC=C(C)C=C1 IMOVWUXHLQRODN-UHFFFAOYSA-N 0.000 description 1
- 125000004036 acetal group Chemical group 0.000 description 1
- AOZUYISQWWJMJC-UHFFFAOYSA-N acetic acid;methanol;hydrate Chemical compound O.OC.CC(O)=O AOZUYISQWWJMJC-UHFFFAOYSA-N 0.000 description 1
- 229960004308 acetylcysteine Drugs 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 1
- 229940061720 alpha hydroxy acid Drugs 0.000 description 1
- 150000001280 alpha hydroxy acids Chemical class 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 150000001448 anilines Chemical class 0.000 description 1
- BTFJIXJJCSYFAL-UHFFFAOYSA-N arachidyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCO BTFJIXJJCSYFAL-UHFFFAOYSA-N 0.000 description 1
- 125000000637 arginyl group Chemical class N[C@@H](CCCNC(N)=N)C(=O)* 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- GJTDJAPHKDIQIQ-UHFFFAOYSA-L barium(2+);dinitrite Chemical compound [Ba+2].[O-]N=O.[O-]N=O GJTDJAPHKDIQIQ-UHFFFAOYSA-L 0.000 description 1
- 229940050390 benzoate Drugs 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 229960004365 benzoic acid Drugs 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- SIPUZPBQZHNSDW-UHFFFAOYSA-N bis(2-methylpropyl)aluminum Chemical compound CC(C)C[Al]CC(C)C SIPUZPBQZHNSDW-UHFFFAOYSA-N 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- NEDGUIRITORSKL-UHFFFAOYSA-N butyl 2-methylprop-2-enoate;2-(dimethylamino)ethyl 2-methylprop-2-enoate;methyl 2-methylprop-2-enoate Chemical compound COC(=O)C(C)=C.CCCCOC(=O)C(C)=C.CN(C)CCOC(=O)C(C)=C NEDGUIRITORSKL-UHFFFAOYSA-N 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- MIOPJNTWMNEORI-UHFFFAOYSA-N camphorsulfonic acid Chemical compound C1CC2(CS(O)(=O)=O)C(=O)CC1C2(C)C MIOPJNTWMNEORI-UHFFFAOYSA-N 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 238000010609 cell counting kit-8 assay Methods 0.000 description 1
- 229940082500 cetostearyl alcohol Drugs 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 229960003749 ciclopirox Drugs 0.000 description 1
- BJIOGJUNALELMI-ARJAWSKDSA-N cis-isoeugenol Chemical compound COC1=CC(\C=C/C)=CC=C1O BJIOGJUNALELMI-ARJAWSKDSA-N 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- GVJHHUAWPYXKBD-UHFFFAOYSA-N d-alpha-tocopherol Natural products OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 201000003929 dermatomycosis Diseases 0.000 description 1
- 230000000249 desinfective effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- USLKCMBGQFYUFI-UHFFFAOYSA-N dichloromethane;tribromoborane Chemical compound ClCCl.BrB(Br)Br USLKCMBGQFYUFI-UHFFFAOYSA-N 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical class OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 150000005332 diethylamines Chemical class 0.000 description 1
- 238000000113 differential scanning calorimetry Methods 0.000 description 1
- 229940031578 diisopropyl adipate Drugs 0.000 description 1
- 229940031569 diisopropyl sebacate Drugs 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 1
- 150000004656 dimethylamines Chemical class 0.000 description 1
- XFKBBSZEQRFVSL-UHFFFAOYSA-N dipropan-2-yl decanedioate Chemical compound CC(C)OC(=O)CCCCCCCCC(=O)OC(C)C XFKBBSZEQRFVSL-UHFFFAOYSA-N 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- PSLIMVZEAPALCD-UHFFFAOYSA-N ethanol;ethoxyethane Chemical compound CCO.CCOCC PSLIMVZEAPALCD-UHFFFAOYSA-N 0.000 description 1
- 150000002169 ethanolamines Chemical class 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- QURVBNHKXPVYQH-UHFFFAOYSA-N ethyl 1-(2-hydroxyphenyl)-3,5-dimethylpyrazole-4-carboxylate Chemical compound CC1=C(C(=O)OCC)C(C)=NN1C1=CC=CC=C1O QURVBNHKXPVYQH-UHFFFAOYSA-N 0.000 description 1
- NSBORFLATSJICK-UHFFFAOYSA-N ethyl 1-(2-methoxyphenyl)-3,5-dimethylpyrazole-4-carboxylate Chemical compound CC1=C(C(=O)OCC)C(C)=NN1C1=CC=CC=C1OC NSBORFLATSJICK-UHFFFAOYSA-N 0.000 description 1
- KACZQOKEFKFNDB-UHFFFAOYSA-N ethyl 1h-pyrazole-4-carboxylate Chemical compound CCOC(=O)C=1C=NNC=1 KACZQOKEFKFNDB-UHFFFAOYSA-N 0.000 description 1
- YMCDYRGMTRCAPZ-UHFFFAOYSA-N ethyl 2-acetyl-3-oxobutanoate Chemical compound CCOC(=O)C(C(C)=O)C(C)=O YMCDYRGMTRCAPZ-UHFFFAOYSA-N 0.000 description 1
- TZMFJUDUGYTVRY-UHFFFAOYSA-N ethyl methyl diketone Natural products CCC(=O)C(C)=O TZMFJUDUGYTVRY-UHFFFAOYSA-N 0.000 description 1
- FSXVSUSRJXIJHB-UHFFFAOYSA-M ethyl prop-2-enoate;methyl 2-methylprop-2-enoate;trimethyl-[2-(2-methylprop-2-enoyloxy)ethyl]azanium;chloride Chemical compound [Cl-].CCOC(=O)C=C.COC(=O)C(C)=C.CC(=C)C(=O)OCC[N+](C)(C)C FSXVSUSRJXIJHB-UHFFFAOYSA-M 0.000 description 1
- 150000003947 ethylamines Chemical class 0.000 description 1
- 229960002217 eugenol Drugs 0.000 description 1
- 150000002191 fatty alcohols Chemical class 0.000 description 1
- 210000004904 fingernail bed Anatomy 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 229940050411 fumarate Drugs 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- JFCQEDHGNNZCLN-UHFFFAOYSA-N glutaric acid Chemical compound OC(=O)CCCC(O)=O JFCQEDHGNNZCLN-UHFFFAOYSA-N 0.000 description 1
- 125000005456 glyceride group Chemical group 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 210000004209 hair Anatomy 0.000 description 1
- 231100000234 hepatic damage Toxicity 0.000 description 1
- DGCTVLNZTFDPDJ-UHFFFAOYSA-N heptane-3,5-dione Chemical compound CCC(=O)CC(=O)CC DGCTVLNZTFDPDJ-UHFFFAOYSA-N 0.000 description 1
- QAMFBRUWYYMMGJ-UHFFFAOYSA-N hexafluoroacetylacetone Chemical compound FC(F)(F)C(=O)CC(=O)C(F)(F)F QAMFBRUWYYMMGJ-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 229940071676 hydroxypropylcellulose Drugs 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- APNSGVMLAYLYCT-UHFFFAOYSA-N isobutyl nitrite Chemical compound CC(C)CON=O APNSGVMLAYLYCT-UHFFFAOYSA-N 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- SKRDXYBATCVEMS-UHFFFAOYSA-N isopropyl nitrite Chemical compound CC(C)ON=O SKRDXYBATCVEMS-UHFFFAOYSA-N 0.000 description 1
- 229960004130 itraconazole Drugs 0.000 description 1
- 125000000468 ketone group Chemical group 0.000 description 1
- 239000004922 lacquer Substances 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- 239000006193 liquid solution Substances 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- SDRRRXPWHKVEMP-UHFFFAOYSA-N lithium;triethyl borate Chemical compound [Li].CCOB(OCC)OCC SDRRRXPWHKVEMP-UHFFFAOYSA-N 0.000 description 1
- 230000008818 liver damage Effects 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- 229940049920 malate Drugs 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-M mandelate Chemical compound [O-]C(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-M 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical compound C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 description 1
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 1
- GBMDVOWEEQVZKZ-UHFFFAOYSA-N methanol;hydrate Chemical compound O.OC GBMDVOWEEQVZKZ-UHFFFAOYSA-N 0.000 description 1
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- DYBQVWGWQBASNK-UHFFFAOYSA-N methyl 3-hydroxy-4-(3,4,5-trimethylpyrazol-1-yl)benzoate Chemical compound OC1=CC(C(=O)OC)=CC=C1N1C(C)=C(C)C(C)=N1 DYBQVWGWQBASNK-UHFFFAOYSA-N 0.000 description 1
- QPJVMBTYPHYUOC-UHFFFAOYSA-N methyl benzoate Chemical compound COC(=O)C1=CC=CC=C1 QPJVMBTYPHYUOC-UHFFFAOYSA-N 0.000 description 1
- UCFFGYASXIPWPD-UHFFFAOYSA-N methyl hypochlorite Chemical group COCl UCFFGYASXIPWPD-UHFFFAOYSA-N 0.000 description 1
- 150000003956 methylamines Chemical class 0.000 description 1
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 description 1
- 125000004092 methylthiomethyl group Chemical group [H]C([H])([H])SC([H])([H])* 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 150000004682 monohydrates Chemical class 0.000 description 1
- 201000007524 mucormycosis Diseases 0.000 description 1
- ACTNHJDHMQSOGL-UHFFFAOYSA-N n',n'-dibenzylethane-1,2-diamine Chemical class C=1C=CC=CC=1CN(CCN)CC1=CC=CC=C1 ACTNHJDHMQSOGL-UHFFFAOYSA-N 0.000 description 1
- DNKKLDKIFMDAPT-UHFFFAOYSA-N n,n-dimethylmethanamine;2-methylprop-2-enoic acid Chemical compound CN(C)C.CC(=C)C(O)=O.CC(=C)C(O)=O DNKKLDKIFMDAPT-UHFFFAOYSA-N 0.000 description 1
- PSHKMPUSSFXUIA-UHFFFAOYSA-N n,n-dimethylpyridin-2-amine Chemical compound CN(C)C1=CC=CC=N1 PSHKMPUSSFXUIA-UHFFFAOYSA-N 0.000 description 1
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 1
- 208000026721 nail disease Diseases 0.000 description 1
- KVBGVZZKJNLNJU-UHFFFAOYSA-M naphthalene-2-sulfonate Chemical compound C1=CC=CC2=CC(S(=O)(=O)[O-])=CC=C21 KVBGVZZKJNLNJU-UHFFFAOYSA-M 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- QMMRZOWCJAIUJA-UHFFFAOYSA-L nickel dichloride Chemical compound Cl[Ni]Cl QMMRZOWCJAIUJA-UHFFFAOYSA-L 0.000 description 1
- 235000001968 nicotinic acid Nutrition 0.000 description 1
- 239000011664 nicotinic acid Substances 0.000 description 1
- VQTGUFBGYOIUFS-UHFFFAOYSA-N nitrosylsulfuric acid Chemical compound OS(=O)(=O)ON=O VQTGUFBGYOIUFS-UHFFFAOYSA-N 0.000 description 1
- GQPLMRYTRLFLPF-UHFFFAOYSA-N nitrous oxide Inorganic materials [O-][N+]#N GQPLMRYTRLFLPF-UHFFFAOYSA-N 0.000 description 1
- YCIMNLLNPGFGHC-UHFFFAOYSA-N o-dihydroxy-benzene Natural products OC1=CC=CC=C1O YCIMNLLNPGFGHC-UHFFFAOYSA-N 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- KSCKTBJJRVPGKM-UHFFFAOYSA-N octan-1-olate;titanium(4+) Chemical compound [Ti+4].CCCCCCCC[O-].CCCCCCCC[O-].CCCCCCCC[O-].CCCCCCCC[O-] KSCKTBJJRVPGKM-UHFFFAOYSA-N 0.000 description 1
- 229940073665 octyldodecyl myristate Drugs 0.000 description 1
- BARWIPMJPCRCTP-UHFFFAOYSA-N oleic acid oleyl ester Natural products CCCCCCCCC=CCCCCCCCCOC(=O)CCCCCCCC=CCCCCCCCC BARWIPMJPCRCTP-UHFFFAOYSA-N 0.000 description 1
- 229940055577 oleyl alcohol Drugs 0.000 description 1
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 description 1
- BARWIPMJPCRCTP-CLFAGFIQSA-N oleyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCCOC(=O)CCCCCCC\C=C/CCCCCCCC BARWIPMJPCRCTP-CLFAGFIQSA-N 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 150000002940 palladium Chemical class 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 230000008823 permeabilization Effects 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- RAFRTSDUWORDLA-UHFFFAOYSA-N phenyl 3-chloropropanoate Chemical compound ClCCC(=O)OC1=CC=CC=C1 RAFRTSDUWORDLA-UHFFFAOYSA-N 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- XNGIFLGASWRNHJ-UHFFFAOYSA-L phthalate(2-) Chemical compound [O-]C(=O)C1=CC=CC=C1C([O-])=O XNGIFLGASWRNHJ-UHFFFAOYSA-L 0.000 description 1
- 150000004885 piperazines Chemical class 0.000 description 1
- 150000003053 piperidines Chemical class 0.000 description 1
- 229940068196 placebo Drugs 0.000 description 1
- 239000000902 placebo Substances 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 235000010289 potassium nitrite Nutrition 0.000 description 1
- 239000004304 potassium nitrite Substances 0.000 description 1
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 235000010388 propyl gallate Nutrition 0.000 description 1
- 239000000473 propyl gallate Substances 0.000 description 1
- 229940075579 propyl gallate Drugs 0.000 description 1
- KAOQVXHBVNKNHA-UHFFFAOYSA-N propyl nitrite Chemical compound CCCON=O KAOQVXHBVNKNHA-UHFFFAOYSA-N 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 150000003222 pyridines Chemical class 0.000 description 1
- 150000004040 pyrrolidinones Chemical class 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- GHMLBKRAJCXXBS-UHFFFAOYSA-N resorcinol Chemical compound OC1=CC=CC(O)=C1 GHMLBKRAJCXXBS-UHFFFAOYSA-N 0.000 description 1
- 229960001755 resorcinol Drugs 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 239000010948 rhodium Substances 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 150000003873 salicylate salts Chemical class 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000035807 sensation Effects 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- KKKDGYXNGYJJRX-UHFFFAOYSA-M silver nitrite Chemical compound [Ag+].[O-]N=O KKKDGYXNGYJJRX-UHFFFAOYSA-M 0.000 description 1
- DVQHRBFGRZHMSR-UHFFFAOYSA-N sodium methyl 2,2-dimethyl-4,6-dioxo-5-(N-prop-2-enoxy-C-propylcarbonimidoyl)cyclohexane-1-carboxylate Chemical compound [Na+].C=CCON=C(CCC)[C-]1C(=O)CC(C)(C)C(C(=O)OC)C1=O DVQHRBFGRZHMSR-UHFFFAOYSA-N 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 1
- VSIVTUIKYVGDCX-UHFFFAOYSA-M sodium;4-[2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)tetrazol-2-ium-5-yl]benzene-1,3-disulfonate Chemical compound [Na+].COC1=CC([N+]([O-])=O)=CC=C1[N+]1=NC(C=2C(=CC(=CC=2)S([O-])(=O)=O)S([O-])(=O)=O)=NN1C1=CC=C([N+]([O-])=O)C=C1 VSIVTUIKYVGDCX-UHFFFAOYSA-M 0.000 description 1
- 229960005078 sorbitan sesquioleate Drugs 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229940012831 stearyl alcohol Drugs 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 210000000434 stratum corneum Anatomy 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- HXJUTPCZVOIRIF-UHFFFAOYSA-N sulfolane Chemical compound O=S1(=O)CCCC1 HXJUTPCZVOIRIF-UHFFFAOYSA-N 0.000 description 1
- 150000003871 sulfonates Chemical class 0.000 description 1
- 201000009862 superficial mycosis Diseases 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- DOMXUEMWDBAQBQ-WEVVVXLNSA-N terbinafine Chemical compound C1=CC=C2C(CN(C\C=C\C#CC(C)(C)C)C)=CC=CC2=C1 DOMXUEMWDBAQBQ-WEVVVXLNSA-N 0.000 description 1
- 229960002722 terbinafine Drugs 0.000 description 1
- CBXCPBUEXACCNR-UHFFFAOYSA-N tetraethylammonium Chemical class CC[N+](CC)(CC)CC CBXCPBUEXACCNR-UHFFFAOYSA-N 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical class C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- HNKJADCVZUBCPG-UHFFFAOYSA-N thioanisole Chemical compound CSC1=CC=CC=C1 HNKJADCVZUBCPG-UHFFFAOYSA-N 0.000 description 1
- 229960000790 thymol Drugs 0.000 description 1
- 229910052718 tin Inorganic materials 0.000 description 1
- 239000011135 tin Substances 0.000 description 1
- 235000010384 tocopherol Nutrition 0.000 description 1
- 229960001295 tocopherol Drugs 0.000 description 1
- 229930003799 tocopherol Natural products 0.000 description 1
- 239000011732 tocopherol Substances 0.000 description 1
- 229940042585 tocopherol acetate Drugs 0.000 description 1
- 229940100613 topical solution Drugs 0.000 description 1
- BJIOGJUNALELMI-UHFFFAOYSA-N trans-isoeugenol Natural products COC1=CC(C=CC)=CC=C1O BJIOGJUNALELMI-UHFFFAOYSA-N 0.000 description 1
- TUQOTMZNTHZOKS-UHFFFAOYSA-N tributylphosphine Chemical compound CCCCP(CCCC)CCCC TUQOTMZNTHZOKS-UHFFFAOYSA-N 0.000 description 1
- FAQYAMRNWDIXMY-UHFFFAOYSA-N trichloroborane Chemical compound ClB(Cl)Cl FAQYAMRNWDIXMY-UHFFFAOYSA-N 0.000 description 1
- IMNIMPAHZVJRPE-UHFFFAOYSA-N triethylenediamine Chemical compound C1CN2CCN1CC2 IMNIMPAHZVJRPE-UHFFFAOYSA-N 0.000 description 1
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical class CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 1
- NHDIQVFFNDKAQU-UHFFFAOYSA-N tripropan-2-yl borate Chemical compound CC(C)OB(OC(C)C)OC(C)C NHDIQVFFNDKAQU-UHFFFAOYSA-N 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
- A61K9/7015—Drug-containing film-forming compositions, e.g. spray-on
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
Definitions
- the present invention is for the prevention or treatment of ringworm mycosis comprising a novel 2- (1H-pyrazol-1-yl) phenol derivative or a pharmaceutically acceptable salt thereof suitable as an anti- ringworm fungus agent as an active ingredient. It relates to a liquid for external use.
- Mycosis is a disease caused by fungi infecting humans and animals.
- Typical human mycosis is Candida caused by Candida, Cryptococcosis caused by Cryptococcus, Aspergillosis caused by Aspergillus, zygomycosis caused by zygomycetes, and dermatophytosis caused by Trichophyton (pathogen) Fungi and mycosis (Nanzan-do, second revised edition), p42-45; Non-patent document 1).
- Ringworm that is one of the pathogens of mycosis is a dermatophyte that can be a causative agent of ringworm, and has the property of keratin resolution. This ability invades the skin, nails, and hairs to cause ringworm (pathogenic fungi and mycosis (Nanyamado, 2nd revised edition), p184-187; Non-Patent Document 2).
- Onychomycosis is a nail disease caused by dermatophytes, which is said to be more than 90% of the causative agents of onychomycosis in Japan, and is a disease with symptoms such as cloudiness, thickening, destruction and deformation of the nail plate.
- dermatophytes which is said to be more than 90% of the causative agents of onychomycosis in Japan, and is a disease with symptoms such as cloudiness, thickening, destruction and deformation of the nail plate.
- the drug in order to exert its efficacy, the drug itself has a strong anti-tinea fungus activity, and in addition to the epidermis stratum corneum and nail that is the site of infection, It is necessary to reach the concentration at a high level.
- the external antifungal agent used for normal dermatomycosis cannot reach the nail keratin, the inside, and the nail bed at a sufficient concentration, and a sufficient effect is not obtained.
- nail lacquers for external use such as amorolfine and ciclopirox are approved and used.
- the penetration into the nail is very low, and further penetration cannot be expected to reach the nail mother cell.
- Non-Patent Document 4 As a compound having 2- (1H-pyrazol-1-yl) phenol skeleton, 2- (3,5-dimethyl-1H-pyrazol-1-yl having methyl groups at the 3-position and 5-position of the pyrazole ring ) Phenol is known (Takeda Institute Annual Report (1963) 22, p27; Non-Patent Document 4). These are intended to prevent the growth of Mycobacterium tuberculosis. However, Non-Patent Document 4 does not disclose 2- (1H-pyrazol-1-yl) phenol compounds other than 2- (3,5-dimethyl-1H-pyrazol-1-yl) phenol.
- 2- (1H-pyrazol-1-yl) is a compound having a 2- (1H-pyrazol-1-yl) phenol skeleton.
- Phenol, 2- (3,5-Dimethyl-1H-pyrazol-1-yl) -1,4-benzenediol, 2- (4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl) -1 , 4-Benzenediol and the like are known. These are used for various chemical reactions, electroluminescent element compound raw materials (Patent No. 4284169: Patent Document 1), light stabilizers (Spanish Publication No. 20158648: Patent Document 2), and the like.
- Patent Document 3 discloses 2- (5-amino-3-tert-butyl-1H-pyrazol-1-yl) -5-methylphenol.
- an object of the present invention is to provide an external liquid for nail and / or skin having anti-tinea fungi activity and having high nail permeability.
- the present inventors apply a liquid for external use containing a compound having a 2- (1H-pyrazol-1-yl) phenol skeleton represented by the following formula (I) or a salt thereof and a polymer to the nail and / or skin. And has a high nail permeability, high storage stability, high anti-tinea fungus activity, and a drug precipitation inhibitory effect, and based on these findings, the present invention was completed.
- the present invention comprises a film forming agent, a general formula (Where R 1 represents a hydrogen atom, C 1-6 alkyl, or trifluoromethyl, R 2 represents a hydrogen atom, C 1-6 alkyl, halogen, —COO (C 1-6 alkyl), or (CH 2 ) 1-3 COOR (R represents a hydrogen atom or C 1-6 alkyl).
- R 3 represents a hydrogen atom, C 1-6 alkyl, amino, trifluoromethyl, or OR (R represents a hydrogen atom or C 1-6 alkyl); R 4 represents a hydroxyl group, R 5 represents a hydrogen atom, C 1-6 alkyl, hydroxyl group, or halogen, R 6 represents a hydrogen atom, C 1-6 alkyl, trifluoromethyl, halogen, amino, —NR a R b , nitro, hydroxy C 1-6 alkyl, —CONR a R b , —COO (C 1-6 alkyl) ), —COOH, — (CH 2 ) 1-3 COOR, or OR a (R represents a hydrogen atom or C 1-6 alkyl, R a and R b may be the same or different, and a hydrogen atom Represents C 1-6 alkyl, or C 1-6 acyl), R 7 represents a hydrogen atom, C 1-6 alkyl, —OR (R represents a hydrogen
- the external preparation of the present invention not only exhibits high antifungal activity against ringworm, but also has high permeability to the nail, so that the nail and / or for the prevention or treatment of ringworm mycosis, particularly nail ringworm It is useful as an external skin solution.
- C 1-6 alkyl denotes an alkyl group having from 1 to 6 carbon atoms.
- the alkyl group may be chain, branched or cyclic.
- C 1-6 alkyl groups such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, hexyl, etc.
- C 3-6 cycloalkyl such as cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl Group and the like.
- C 1-6 acyl part of the "C 1-6 acyl” group, an acyl group having from 1 to 6 carbon atoms.
- the acyl group may be chain, branched or cyclic. Examples include formyl, acetyl, propionyl, butyryl, isobutyryl and the like. Preferably, formyl, acetyl, propionyl, butyryl and the like are mentioned.
- halogen includes fluorine, chlorine, bromine, iodine and the like.
- R represents a hydrogen atom or C 1-6 alkyl
- C 1-6 alkyl has the same meaning as the above “C 1-6 alkyl”.
- R a represents a hydrogen atom, C 1-6 alkyl, or C 1-6 acyl
- R a represents a hydrogen atom, C 1-6 alkyl, or C 1-6 acyl
- —OR a Represents a hydroxyl group, C 1-6 alkyloxy, or C 1-6 acyloxy.
- C 1-6 alkyl has the same meaning as the above “C 1-6 alkyl”.
- the present invention comprises a film-forming agent, a general formula And a liquid for external use for nails and / or skin for preventing or treating ringworm bacilli.
- R 1 represents a hydrogen atom, C 1-6 alkyl, or trifluoromethyl, preferably a hydrogen atom or C 1-6 alkyl, more preferably a C 1-6 alkyl. is there. Specific examples of R 1 include a hydrogen atom, methyl, ethyl and the like.
- R 2 represents a hydrogen atom, C 1-6 alkyl, halogen, —COO (C 1-6 alkyl), or — (CH 2 ) 1-3 COOR (R represents a hydrogen atom or C Represents a 1-6 alkyl), preferably a hydrogen atom, C 1-6 alkyl, or halogen.
- R 2 include a hydrogen atom, methyl, ethyl, propyl, butyl, chlorine atom, or 2-carboxyethyl.
- R 3 represents a hydrogen atom, C 1-6 alkyl, amino, trifluoromethyl, —OR (R represents a hydrogen atom or C 1-6 alkyl), preferably a hydrogen atom Or C 1-6 alkyl, more preferably C 1-6 alkyl. Specific examples of R 3 include methyl or ethyl.
- R 4 represents a hydroxyl group.
- R 5 represents a hydrogen atom, C 1-6 alkyl, hydroxyl group, or halogen, preferably a hydrogen atom.
- R 6 represents a hydrogen atom, C 1-6 alkyl, trifluoromethyl, halogen, amino, —NR a R b , nitro, hydroxy C 1-6 alkyl, —CONR a R b , — COO (C 1-6 alkyl), - COOH, - (CH 2) 1-3 COOR or oR a
- R represents a hydrogen atom or C 1-6 alkyl, R a and R b, be the same or different
- R represents a hydrogen atom, C 1-6 alkyl, or C 1-6 acyl
- R a and R b may be the same or different, a hydrogen atom, C 1-6 alkyl, Other represents represents)
- R 6 examples include a hydrogen atom, methyl, ethyl, methoxy, chlorine atom, bromine atom, fluorine atom, hydroxyl group, acetoxy, amino, nitro, carboxyl, hydroxymethyl, trifluoromethyl, methylamino, carbamoyl, Another example is N, N-dimethylcarbamoyl.
- R 7 represents a hydrogen atom, C 1-6 alkyl, —OR (R represents a hydrogen atom or C 1-6 alkyl), or halogen, and preferably represents a hydrogen atom.
- R 8 represents a hydrogen atom, C 1-6 alkyl, hydroxyl group, amino, or nitro, preferably a hydrogen atom, C 1-6 alkyl, amino, or nitro, and more preferably a hydrogen atom. Specific examples of R 8 include a hydrogen atom, methyl, nitro, or amino.
- R 1 is a hydrogen atom or C 1-6 alkyl
- R 2 is a hydrogen atom, C 1-6 alkyl, halogen, or — (CH 2 ) 1-3 COOR (R represents a hydrogen atom or C 1-6 alkyl);
- R 3 is a hydrogen atom or C 1-6 alkyl,
- R 4 is a hydroxyl group,
- R 5 is a hydrogen atom
- R 6 is a hydrogen atom, C 1-6 alkyl, trifluoromethyl, halogen, amino, —NR a R b , nitro, hydroxy C 1-6 alkyl, —CONR a R b , —COOH, or OR a (R a and R b may be the same or different and each represents a hydrogen atom, C 1-6 alkyl, or C 1-6 acyl),
- R 7 is a hydrogen atom,
- A is a hydrogen atom, A
- a more preferred compound of general formula (I) is: R 1 is C 1-6 alkyl, R 2 is a hydrogen atom, C 1-6 alkyl, or halogen; R 3 is C 1-6 alkyl; R 4 is a hydroxyl group, R 5 is a hydrogen atom, R 6 represents a hydrogen atom, C 1-6 alkyl, halogen, or OR a (R a represents a hydrogen atom, C 1-6 alkyl, or C 1-6 acyl); R 7 is a hydrogen atom, R 8 is a compound which is a hydrogen atom (corresponding to claim 3).
- a more preferred compound of the general formula (I) is that R 1 is C 1-6 alkyl, and R 2 is A hydrogen atom, R 3 is C 1-6 alkyl, R 4 is a hydroxyl group, R 5 is a hydrogen atom, R 6 is C 1-6 alkyl, and R 7 is It is a compound that is a hydrogen atom and R 8 is a hydrogen atom (corresponding to claim 4).
- Examples of the compound of the general formula (I) contained in the nail and / or external skin solution for prevention or treatment of ringworm bacilli of the present invention include the following compounds.
- Production Example 1 2- (3,5-Dimethyl-1H-pyrazol-1-yl) phenol
- Production Example 2 2- (3,5-Dimethyl-1H-pyrazol-1-yl) -4-fluorophenol Production
- Production Example 3 2- (1H-pyrazol-1-yl) phenol production example 4 2- (5-hydroxy-3-methyl-1H-pyrazol-1-yl) phenol production example 5 2- (5-methyl-3-trifluoro) Methyl-1H-pyrazol-1-yl) phenol production example 6 2- (3,5-bistrifluoromethyl-1H-pyrazol-1-yl) phenol production example 7 2- (3-methyl-1H-pyrazol-1- Yl) phenol production example 8 2- (5-methyl-1H-pyrazol-1-yl) phenol production example 9 2- (3,4,5-trimethyl-1H
- the compound of the general formula (I) contained in the external preparation for nail and / or skin for prevention or treatment of trichomycosis of the present invention can also be used as a salt.
- any pharmaceutically acceptable salt can be used, and specific examples thereof include pharmaceutically acceptable salts derived from inorganic acids, organic acids or bases.
- “Pharmaceutically acceptable salts” are known in the art. For example, S. M. Berge et al. Describe pharmaceutically acceptable salts in detail in Journal of Pharmaceutical Sciences, 66 p.1 et seq. (1977).
- Typical acid addition salts include inorganic acid salts such as hydrochloride, sulfate, nitrate, hydrobromide, hydroiodide, phosphate; acetate, trifluoroacetate, lactate, citric acid Organic carboxylic acids such as acid, oxalate, succinate, glutarate, malate, tartrate, fumarate, mandelate, maleate, benzoate, nicotinate, phthalate Organic sulfonates such as methanesulfonate, ethanesulfonate, 2-hydroxyethanesulfonate, benzenesulfonate, p-toluenesulfonate, 2-naphthalenesulfonate, camphorsulfonate; Examples include, but are not limited to, acidic amino acid salts such as aspartate and glutamate.
- acid addition salts with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid and phosphoric acid; and organic acids such as oxalic acid, maleic acid, methanesulfonic acid, p-toluenesulfonic acid and citric acid Salt. More preferred are salts with hydrochloric acid, hydrobromic acid, sulfuric acid and methanesulfonic acid.
- the base addition salt can be produced in situ by reacting a carboxylic acid or phenolic hydroxyl group-containing moiety with an appropriate base during the final isolation and purification process of the compound of the present invention.
- Pharmaceutically acceptable salts include alkali metal salts such as lithium salt, sodium salt and potassium salt; alkaline earth metal salts such as calcium salt and magnesium salt; aluminum salt, ammonium salt, and methylamine salt; Dimethylamine salt, ethylamine salt, diethylamine salt, trimethylamine salt, triethylamine salt, tetramethylammonium salt, tetraethylammonium salt, pyridine salt, picoline salt, ethanolamine salt, diethanolamine salt, triethanolamine salt, trishydroxymethylaminomethane salt, Organic base salts such as piperidine salts, piperazine salts, dicyclohexylamine salts, N, N-dibenzylethylenediamine salts; basic amino acid salts such as arginine salts
- Preferred base addition salts include addition salts with sodium, potassium, calcium, ethanolamine and trishydroxymethylaminomethane. More preferred are addition salts with sodium, potassium and trishydroxymethylaminomethane.
- solvate means various stoichiometric complexes formed by a solute (in the present invention, a compound of formula (I) or a salt thereof) and a solvent.
- a solvent for the purposes of the present invention is preferably a pharmaceutically acceptable solvent that does not interfere with the biological activity of the solute.
- suitable solvents for solvates include, but are not limited to, water, methanol, ethanol, ethylene glycol, propylene glycol, ethyl acetate and butyl acetate.
- Solvents for the solvate preferably include water, ethanol and ethyl acetate.
- the 2- (1H-pyrazol-1-yl) phenol derivative represented by the general formula (I) contained in the external preparation of the present invention can be produced by any method. Although it can synthesize
- each symbol of the compound is as described above, P and P ′ represent hydrogen or a suitable protecting group, X represents a halogen or a suitable boronic acid group, and Y was used for the reaction.
- “suitable boronic acid” represents a boronic acid or a boronic acid ester.
- a method for obtaining a hydrazine derivative (V) or a salt thereof from a 2-hydroxyaniline derivative (IIIa) or a salt thereof via a diazonium compound (IVa) is described in Organic Synthesis Collective Volume 1, p. 442-445, J. Org. Chem., 21, p.394-399, 1956, WO2007 / 083320, US6852890.
- nitrites such as potassium nitrite, calcium nitrite, silver nitrite, sodium nitrite, barium nitrite, nitrosyl sulfate or ethyl nitrite, isoamyl nitrite, isobutyl nitrite, isopropyl nitrite, tert-nitrite
- Nitrous esters such as butyl, n-butyl nitrite, and n-propyl nitrite can be used.
- nitrite such as sodium nitrite or isoamyl nitrite and tert-butyl nitrite is used.
- P and P ′ include a methyl group, an isopropyl group, an allyl group, a tert-butyl group, a methoxymethyl group, and a methylthiomethyl group.
- Alkyl groups such as benzyl group and 9-anthrylmethyl group, acyl groups such as pivaloyl group and benzoyl group, or sulfonyl groups such as p-toluenesulfonyl group and methanesulfonyl group. It is not a thing.
- Preferred protecting groups include methyl, p-toluenesulfonyl and methanesulfonyl groups.
- the amount of the reagent used in the diazotization reaction is preferably 1 to 10 equivalents, more preferably 1 to 3 equivalents, relative to the 2-hydroxyaniline derivative (IIIa).
- the above diazotization reaction is carried out with water, an organic solvent mixed with water at an arbitrary ratio, for example, methanol, ethanol, 2-propanol, acetic acid, trifluoroacetic acid, tetrahydrofuran, 1,4-dioxane, dimethyl Formamide and dimethyl sulfoxide can be used. Further, a plurality of these solvents can be mixed and used. Preferably, water, a water-methanol mixture, and a water-methanol-acetic acid mixture are used. Further, the diazotization reaction is carried out under acidic conditions in order to ensure the solubility of the substrate aniline derivative and to generate nitrous acid in the reaction system.
- an organic solvent mixed with water at an arbitrary ratio for example, methanol, ethanol, 2-propanol, acetic acid, trifluoroacetic acid, tetrahydrofuran, 1,4-dioxane, dimethyl Formamide and dimethyl sulfoxide can be used. Further,
- Examples of the acid used include hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, acetic acid, trifluoroacetic acid, and phosphoric acid. Preferred are hydrochloric acid, acetic acid and trifluoroacetic acid. Furthermore, these acids can also be used as a solvent.
- the above diazotization reaction may be carried out by using alcohols such as methanol, ethanol, methoxyethanol, ethoxyethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol and 2-methyl-2-propanol when nitrites are used.
- alcohols such as methanol, ethanol, methoxyethanol, ethoxyethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol and 2-methyl-2-propanol when nitrites are used.
- Ether solvents such as diethyl ether, diisopropyl ether, tetrahydrofuran, methyl-tert-butyl ether, diphenyl ether and 1,4-dioxane, acetates such as methyl acetate, ethyl acetate, propyl acetate and butyl acetate, N, Aprotic solvents such as N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone and dimethylsulfoxide, and halogen solvents such as dichloromethane, chloroform and 1,2-dichloroethane can be used. Further, a plurality of these solvents can be mixed and used. Preferred is a mixed solution of methanol, ethanol and ethanol-diethyl ether.
- the temperature of the diazotization reaction is preferably -50 ° C to 50 ° C, more preferably -30 ° C to 10 ° C, regardless of whether nitrite is used or nitrite is used. More preferably, it is between -10 ° C and 5 ° C.
- the above-mentioned diazotization reaction varies depending on the substrate and reaction conditions, but is often completed within 5 minutes to 12 hours, usually within 3 hours.
- the concentration of the substrate in the diazotization reaction solution is not particularly limited, but can be performed in the range of 0.1 mM to 10 M. Preferably, it is in the range of 1 mM to 1M.
- stannous chloride or a hydrate thereof For the reduction from the diazonium compound (IVa) to the hydrazine derivative (V), stannous chloride or a hydrate thereof, sulfite, bisulfite, dithionite, triphenylphosphine, or the like can be used ( Organic Synthesis Collective Volume 1, p.442-445, J.Org.Chem., 21, 394-399, 1956, WO2007 / 083320, US6852890, US2007 / 0105866, J.Am.Chem.Soc., 92, p.853-859, 1970). A method using stannous chloride, dithionite, or sulfite is preferable.
- the above reduction reaction can be carried out following the diazotization reaction.
- a hydrazine derivative (V) or a salt thereof is synthesized by adding a reducing reagent to the reaction solution or adding a diazotization reaction solution to the reducing reagent solution without isolating generally unstable diazonium compounds. Is possible.
- the amount of the reducing agent is preferably 1 to 30 equivalents, more preferably 1 to 10 equivalents, relative to the corresponding diazonium compound.
- the solvent used in the reduction reaction may be the same as the solvent used in the diazotization reaction, and may be added as appropriate, but is preferably the same as the diazotization reaction.
- the temperature of the above reduction reaction varies depending on the type of reducing agent, but is preferably -50 ° C to 120 ° C, more preferably -10 ° C to 70 ° C. More preferably, it is between -10 ° C and 30 ° C.
- the hydrazine derivative (V) or a salt thereof can be synthesized from the compound (IIIb) without passing through the diazonium compound (IVa). That is, the hydrazine derivative (V) or a salt thereof can also be obtained by reacting compound (IIIb) with hydrazine or P′-protected hydrazine in the presence or absence of a suitable catalyst.
- the phenylpyrazole derivative (VI) can also be synthesized by reacting compound (IIIb) with an appropriate pyrazole in the presence or absence of an appropriate catalyst.
- any substance that is inactive in steps other than deprotection may be used.
- P ′ include methoxycarbonyl group, ethoxycarbonyl group, tert-butoxycarbonyl group, benzyloxycarbonyl Examples include, but are not limited to, alkyloxycarbonyl groups such as groups, acyl groups such as pivaloyl groups and benzoyl groups, or sulfonyl groups such as p-toluenesulfonyl groups and methanesulfonyl groups.
- a preferred protecting group includes a t-butoxycarbonyl group.
- the amount of hydrazine or P′-protected hydrazine or pyrazole used in the above reaction is preferably 1 to 30 equivalents, more preferably 1 to 5 equivalents, relative to compound (IIIb).
- the solvent suitable for the above reaction varies depending on the substrate and reaction conditions, but N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, dimethyl sulfoxide, sulfolane, acetonitrile and propionitrile
- Aprotic solvents such as 1,4-dioxane, ether solvents such as 1,2-dimethoxyethane and 1,2-diethoxyethane, chloroform, 1,2-dichloroethane, 1,1,2,2-
- Nonlimiting examples include halogenated solvents such as tetrachloroethane and 1,1,1-trichloroethane. Further, a plurality of these solvents can be mixed and used.
- the reaction can be performed without using a solvent.
- a solvent Preferably, N, N-dimethylformamide, N-methylpyrrolidone, propionitrile, dimethyl sulfoxide is used, or the reaction is carried out without solvent.
- copper or palladium coordinated with an appropriate ligand in addition to a copper or palladium salt, copper or palladium coordinated with an appropriate ligand can be used in a catalytic amount or a stoichiometric amount or more.
- 1,4-diazabicyclo [2,2,2] octane, 1,8-diazabicyclo [5,4,0] undecene, pyridine, organic bases such as N, N-dimethylaminopyridine, tert-butoxypotassium , Tert-butoxy sodium, or an inorganic base such as potassium carbonate, cesium carbonate, or potassium phosphate is preferably used. More preferred are pyridine, potassium carbonate, cesium carbonate, potassium phosphate and the like.
- Suitable ligands include tributylphosphine, triphenylphosphine, N-methylglycine, N, N-dimethylglycine, 1,2-diaminocyclohexane, 1,10-phenanthroline derivative, 8-hydroxyquinoline, picoline Examples thereof include, but are not limited to, acid and 2,2′-bipyridine. N, N-dimethylglycine, 1,2-diaminocyclohexane and 8-hydroxyquinoline are preferable.
- the temperature of the above reaction is preferably 10 ° C. to 200 ° C., more preferably 20 ° C. to 150 ° C., although it varies depending on the type of substrate, the presence / absence of the catalyst, and the type. At this time, the reaction may be accelerated by irradiation with microwaves.
- the above reaction varies depending on the type of substrate, the presence or absence of the catalyst, and the type, but is often completed within 15 minutes to 96 hours, usually within 48 hours.
- the substrate concentration of the above reaction is not particularly limited, but it is usually 1 mM to neat (no solvent).
- the preferred range is 10 mM to 10M.
- the phenylhydrazole derivative (VI) in which a pyrazole ring is formed can be synthesized by reacting the obtained hydrazine derivative (V) or a salt thereof with 1,3-diketone or a chemical equivalent thereof.
- the chemical equivalent means a compound in which a carbonyl group is protected with an acetal group and can be easily converted into a ketone group with an acid or the like present in the system during a pyrazole ring formation reaction.
- the amount of 1,3-diketone or chemical equivalent thereof used in the above reaction is preferably 1 to 20 equivalents, more preferably 1 to 5 equivalents, relative to compound (V).
- the solvent suitable for the above reaction varies depending on the substrate and reaction conditions, but methanol, ethanol, methoxyethanol, ethoxyethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol, 2-methyl-2- Alcohols such as propanol, glycerol and 1,3-propanediol, diethyl ether, diisopropyl ether, tetrahydrofuran, methyl-tert-butyl ether, diphenyl ether, 1,4-dioxane, diethylene glycol dimethyl ether, 1,2-dimethoxyethane and 1, Ether solvents such as 2-diethoxyethane, acetates such as methyl acetate, ethyl acetate, propyl acetate and butyl acetate, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone , Dimethyl sulfoxide, ace
- an appropriate acid can be added in a catalytic amount or 1 equivalent or more.
- Suitable acids include hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, acetic acid, trifluoroacetic acid and phosphoric acid. Hydrochloric acid and trifluoroacetic acid are preferred.
- the temperature of the above reaction varies depending on the type of substrate, but is preferably 10 ° C to 200 ° C, more preferably 40 ° C to 120 ° C.
- the above reaction varies depending on the type of substrate, but is often completed within 15 minutes to 24 hours, usually within 12 hours.
- the concentration of the substrate for the above reaction is not particularly limited, but is usually 0.1 mM to 1 M.
- the preferred range is 10 mM to 1 M.
- the obtained phenylpyrazole derivative (VI) can be led to the compound (I) of the present invention by deprotection of a protecting group, if necessary.
- a protecting group for the deprotection reaction, an appropriate method may be used according to the used P with reference to Green, Protective Groups Organic Synthesis (5th), 1999, John Wieley & Sons.
- P is a methyl group, boron tribromide, aluminum chloride, etc.
- P is a benzyl group, catalytic hydrogenation reduction, etc.
- P is a p-toluenesulfonyl group, sodium hydroxide or hydroxide It is preferable to use potassium or the like.
- the compound (I) of the present invention can be further subjected to further chemical modification of one or both of the benzene ring side chain and the pyrazole ring side chain by an organic chemical reaction as usually used.
- a compound having a carboxyl group is esterified, amidated, reduced to an alcohol, etc.
- a compound having an amino group is alkylated, acylated, carbamatized, etc. It is also possible to apply.
- the compound (I) of the present invention can also be synthesized using a 2-nitroaniline derivative (IIIc) or a nitrobenzene derivative (IIId) having a halogen or boronic acid group at the 2-position as shown in Scheme 2.
- P ′ represents hydrogen or a suitable protecting group
- X represents a halogen or a suitable boronic acid group
- Y represents a dissociated ion of the acid used in the reaction.
- the diazotization of the 2-nitroaniline derivative (IIIc), the reduction of the resulting diazonium compound, and the cyclization reaction of the pyrazole ring are in accordance with the above-mentioned production methods (IIIa) to (V) and (V) to (VI). Can be synthesized.
- the reaction from the nitrobenzene derivative (IIId) to the 2-nitrophenylpyrazole derivative (VIII) can be synthesized according to the production method of (IIIb) to (VI) described above. Alternatively, it can be synthesized according to the method described in J. Org. Chem., Vol.76, P654-660, 2011.
- the hydrogenation pressure is 1 to 80 atm, preferably 1 to 5 atm.
- Suitable solvents for the above reaction vary depending on the substrate and reaction conditions, but methanol, ethanol, methoxyethanol, ethoxyethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol and 2-methyl-2- Alcohols such as propanol, diethyl ether, diisopropyl ether, tetrahydrofuran, methyl-t-butyl ether, diphenyl ether, ether solvents such as diethylene glycol dimethyl ether and 1,4-dioxane, methyl acetate, ethyl acetate, propyl acetate and butyl acetate Acetates such as N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, dimethyl sulfoxide, glycerol, 1,3-propanediol, 1,2-dimethoxyethane, 1,2- Diethoxyethane, water and Although acid but not
- metals such as palladium, platinum, rhodium and nickel and complexes thereof, and these compounds adsorbed on activated carbon or salts thereof are used.
- Palladium carbon and Raney nickel are preferred.
- the temperature of the catalytic hydroreduction reaction varies depending on the type of substrate and catalyst, but is preferably 0 ° C to 100 ° C, more preferably 10 ° C to 50 ° C.
- the catalytic hydrogenation reduction reaction varies depending on the type of substrate, but is often completed within 15 minutes to 24 hours, usually within 12 hours.
- the substrate concentration for the catalytic hydrogenation reduction reaction is not particularly limited, but is usually 0.1 mM to 1 M.
- the preferred range is 1 mM to 100 mM.
- the metal used is iron, tin, zinc, etc., and it is necessary to add an acid to both.
- the acid to be used hydrochloric acid, sulfuric acid, acetic acid, trifluoroacetic acid and phosphoric acid can be used, and hydrochloric acid is preferred. Furthermore, these acids can also be used as a solvent.
- the amount of metal used in the above reaction is preferably 1 to 100 equivalents, more preferably 3 to 15 equivalents, relative to compound (VIII).
- Suitable solvents for the above reaction vary depending on the substrate and reaction conditions, but water, acetic acid, methanol, ethanol, methoxyethanol, ethoxyethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol and 2- Alcohols such as methyl-2-propanol, ethereal solvents such as diethyl ether, diisopropyl ether, tetrahydrofuran, diphenyl ether, diethylene glycol dimethyl ether and 1,4-dioxane, such as methyl acetate, ethyl acetate, propyl acetate and butyl acetate Acetates, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone, dimethyl sulfoxide, glycerol, 1,3-propanediol, 1,2-dimethoxyethane and 1,2-diethoxy Ethane can be mentioned but this is limited
- the temperature of the above reaction varies depending on the substrate type and conditions, but is preferably 0 ° C. to 100 ° C., more preferably 20 ° C. to 50 ° C.
- the above reaction varies depending on the type and conditions of the substrate, but it is often completed within 1 to 24 hours, usually within 12 hours.
- the concentration of the substrate for the above reaction is not particularly limited, but is usually 0.1 mM to 1 M.
- the preferred range is 1 mM to 100 mM.
- lithium borohydride, sodium borohydride, potassium borohydride, zinc borohydride, lithium triethoxyborane, diisobutylaluminum hydride, etc. are used as reagents, but preferably hydrogenation Lithium borohydride or sodium borohydride.
- stannous chloride, nickel (II) chloride, etc. may give good results.
- the amount of the reducing agent used in the above reaction is preferably 1 to 50 equivalents, more preferably 1 to 5 equivalents, relative to compound (VIII).
- Suitable solvents for the above reaction vary depending on the type of substrate and reducing agent, but diethyl ether, diisopropyl ether, tetrahydrofuran, diphenyl ether, 1,4-dioxane, diethylene glycol dimethyl ether, 1,2-dimethoxyethane, and 1,2- And ether solvents such as diethoxyethane, alcohols such as methanol, ethanol, methoxyethanol, ethoxyethanol, 1-propanol, 2-propanol, 1-butanol, 2-butanol and 2-methyl-2-propanol However, it is not limited to this. Further, a plurality of these solvents can be mixed and used. Preferred are methanol and a mixture of methanol and diethylene glycol dimethyl ether.
- the temperature of the above reaction varies depending on the type of substrate and conditions, but is preferably -80 ° C to 100 ° C, more preferably -20 ° C to 80 ° C.
- the above reaction varies depending on the type and conditions of the substrate, but is often completed within 15 minutes to 24 hours, usually within 12 hours.
- the concentration of the substrate for the above reaction is not particularly limited, but is usually 0.1 mM to 1 M.
- the preferred range is 1 mM to 100 mM.
- the obtained 2-aminophenylpyrazole derivative (IX) can be diazotized according to the above-mentioned production method from (IIIa) to (IVa) and JP-A-8-53401.
- reaction from the diazonium compound to (I) can be carried out according to the methods described in JP-A-8-188545 and JP-A-11-60528.
- the hydrolysis of the diazonium salt can be carried out by heating in water or a solvent containing water under acidic conditions.
- the solvent suitable for the above reaction varies depending on the substrate and reaction conditions, but water, acetic acid, trifluoroacetic acid, methanol, ethanol, methoxyethanol, ethoxyethanol, glycerol, 1,3-propanediol, 1-propanol, 2 Of alcohols such as -propanol, 1-butanol, 2-butanol and 2-methyl-2-propanol, tetrahydrofuran, diethylene glycol dimethyl ether, 1,2-dimethoxyethane, 1,2-diethoxyethane and 1,4-dioxane
- aprotic solvents such as ether solvents, N, N-dimethylformamide, N, N-dimethylacetamide, N-methylpyrrolidone and dimethylsulfoxide are not limited thereto. Further, a plurality of these solvents can be mixed and used. It is preferable to use water, sulfuric acid, hydrochloric acid, trifluoroacetic acid,
- the temperature of the above reaction varies depending on the type and conditions of the substrate, but is preferably 20 ° C to 200 ° C, more preferably 50 ° C to 150 ° C.
- the above reaction varies depending on the type and conditions of the substrate, but is often completed within 10 minutes to 24 hours, usually within 12 hours.
- the concentration of the substrate for the above reaction is not particularly limited, but is usually 0.1 mM to 1 M.
- the preferred range is 1 mM to 100 mM.
- the nail and / or skin external liquid for prevention or treatment of ringworm bacilli of the present invention contains the compound represented by the above general formula (I) or a salt thereof together with the film forming agent.
- a film-forming agent any film-forming agent generally used in nail or external skin liquid preparations can be used.
- Such a film-forming agent contains a solvent and an additive.
- the additive include a polymer
- the solvent include an organic solvent.
- the polymer include water-soluble polymers and water-insoluble polymers.
- water-soluble polymer examples include copolyvidone, povidone, hydroxypropylcellulose, hypromellose, polyvinyl alcohol, and the like
- water-insoluble polymer examples include acrylic resin alkanolamine, or aminoalkyl methacrylate copolymer RS, aminoalkyl methacrylate copolymer E
- examples include polyvinyl acetal diethylaminoacetate
- the solvent examples include lower alcohol, ethyl acetate, butyl acetate and the like
- examples of the lower alcohol include methanol, ethanol, isopropanol and the like.
- ethanol is used.
- Water-soluble polymer-containing film-forming agent examples include, for example, copolyvidone (Plaston S-630, manufactured by Ashland) , Plusdon K-17, Plusdon K-25, Plusdon K-29 / 32, Plusdon K-90, Plusdon K-90D), Hydroxypropylcellulose (Nippon Soda HPC-SSL, HPC-SL, HPC-L), Hypromellose (TC-5E, TC-5R, TC-5M manufactured by Shin-Etsu Chemical Co., Ltd.) and polyvinyl alcohol (GOHSENOL manufactured by Nippon Synthetic Chemical).
- Water-insoluble polymer-containing film-forming agent examples include acrylic resin alkanolamines (plus size L-53 series, plus size L-9000 series manufactured by Kyoyo Chemical Industry), aminoalkyl methacrylate copolymers RS (Evonik's Eudragit RS, Eudragit RL, etc.), aminoalkyl methacrylate copolymer E (Evonik's Eudragit E), polyvinyl acetal diethylaminoacetate (Sankyo AEA Sankyo), and the like.
- Compounding ratio of compound of general formula (I) or salt thereof in the total amount of the liquid agent is determined appropriately depending on the type of compound. can do.
- the compound of general formula (I) or a salt thereof is blended with a water-soluble polymer-containing film-forming agent or a water-insoluble film-forming agent, the compound of general formula (I) or a salt thereof is added to the total amount of the solution.
- the total amount is preferably 1 to 50% by mass, more preferably 1 to 30% by mass, still more preferably 1 to 20% by mass, and particularly preferably 5 to 15% by mass.
- Additives In the nail and / or skin external liquid of the present invention, in addition to the above-mentioned film forming agent, preservatives, solubilizers, permeation accelerators, antioxidants, and viscous agents that are usually used for external preparations and the like. Etc. can be contained.
- preservatives examples include organic preservatives (cationic activators, phenols, sorbates, salicylates, dehydroacetates, benzoates, etc.), inorganic preservatives (silver, copper, zinc, etc.) Antibacterial zeolite obtained by ion exchange of antibacterial metal ions with zeolite, etc.) can be blended.
- solubilizer examples include polyhydric alcohols (glycerin, sorbitol, ethylene glycol, propylene glycol, dipropylene glycol, 1,3-butylene glycol, 1,3-tetramethylene glycol, polyethylene glycol, etc.), phenols (thymol) , Safrole, isosafrole, eugenol, isoeugenol, etc., higher alcohols (benzyl alcohol, oleyl alcohol, cetyl alcohol, stearyl alcohol, cetostearyl alcohol, octyldodecanol, etc.), ester surfactants (sorbitan sesquioleate, Polyoxyethylene hydrogenated castor oil, polyoxyl stearate), fatty acid esters (isopropyl myristate, octyldodecyl myristate, oleyl oleate, List solubilizers such as diethyl phthalate, dibutyl phthalate
- permeation enhancers include fatty acids, fatty acid esters (isopropyl myristate, palmitic isopropyl, diisopropyl sebacate, diethyl sebacate, diisopropyl adipate, diethyl adipate, etc.), fatty acid amides, fatty alcohols, 2- (2-ethoxy Ethoxy) -ethanol, ester of glycerol, glycerol monolaurate, propylene glycol, polyethylene glycol, unsaturated polyglycolized glyceride, saturated polyglyceride, ⁇ -hydroxy acid, dimethyl sulfoxide, decylmethyl sulfoxide, pyrrolidones, salicylic acid, lactic acid, Dimethylformamide, dimethylacetamide, sodium dodecyl sulfate, phospholipid, oleic acid, oleic acid / 2- (2-ethoxyethoxy) ethanol, protein It can be
- the nail and / or skin topical solution of the present invention comprises a thickening agent, preferably a silicone derivative, hydrophobized hydroxypropylmethylcellulose, or poloxamer.
- Production Example 14 2- (3,5-Diethyl-1H-pyrazol-1-yl) phenol a) From 3,5-diethyl-1- (2-methoxyphenyl) -1H-pyrazole 2-methoxyphenylhydrazine hydrochloride 174.6 mg and 3,5-heptanedione 135.5 ⁇ l, the same method as in Production Example 1a) As a result, 209.8 mg of the title compound was obtained.
- Production Example 58 4- (4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl) -3-hydroxyphenyl acetate Same as Production Example 54a) using 1.0 g of 4-amino-3-hydroxyphenyl acetate 30 mg of the title compound was obtained from 0.9 g of 4-hydrazinyl-3-hydroxyphenyl acetate 4-methylbenzenesulfonate and 1.2 g of 3-chloropentane-2,4-dione prepared in the same manner as in Production Example 54b). .
- Production Example 59 3-hydroxy-4- (3,4,5-trimethyl-1H-pyrazol-1-yl) phenyl acetate 1.0 g of 4-amino-3-hydroxyphenyl acetate was used in the same manner as in Production Example 54a). 230 mg of the title compound was obtained in the same manner as in Production Example 54b) from 0.6 g of the prepared 4-hydrazinyl-3-hydroxyphenyl acetate 4-methylbenzenesulfonate and 1.0 g of 3-methylpentane-2,4-dione.
- Production Example 63 4- (4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl) -benzene-1,3-diol
- Production Example 44 means “Compound of Production Example 44”.
- Production Example 44 10 g Copolyvidone 10 g Ethanol 100mL Production Example 44 and copolyvidone were sequentially dissolved in a part of ethanol to make a total of 100 mL with ethanol to obtain a preparation. Further, referring to the above method, an external liquid preparation containing the following polymer was produced instead of copolyvidone.
- the enthalpy was calculated for the melting peak of Production Example 44 near 105 ° C., and compared with the melting enthalpy measured by Production Example 44 powder alone. As shown in Table 2, for any of the polymers, there was a tendency for the amount of drug present in a crystalline state in the film to decrease as the amount of polymer added relative to the amount of drug increased.
- bovine hoof slice Cut 0.1 mm thick bovine claw into an appropriate size and immerse in disinfecting ethanol. After drying, place a cylindrical plastic (about 2.5mm in height, about 5mm in width, about 1mm in thickness) on the bovine hoof, fix it with an adhesive, and let it stand overnight for complete adhesion.
- Anti- ringworm fungus activity test Anti- ringworm fungus activity measurement of the compound of the general formula (I) contained in the external preparation of the present invention was carried out by the following method. The evaluation compound was used by dissolving in dimethyl sulfoxide (DMSO). As a test medium, RMI1640 (RPMI1640) medium containing 0.165M 3-morpholinopropanesulfonic acid (MOPS) was used. As a test bacterium, Trichophyton mentagrophytes ATCC18748 or Trichophyton rubrum ATCC10218 was used.
- DMSO dimethyl sulfoxide
- MOPS 3-morpholinopropanesulfonic acid
- WST8 Cell Counting Kit 8
- the permeabilization activity against the human nail of the external solution of the present invention was measured as follows using a tertube model of Medpharma. First, a test preparation was prepared. The test preparation was a preparation for external use of Production Example 44, and the concentration of Production Example 44 was adjusted to 0% (placebo), 2%, 5%, 10% and 15%, and prepared by adding copolyvidone (10%) and ethanol. . After 2 ⁇ l of the test preparation was applied onto the human nail at the top of the tertube cell, the tertube cell was cultured at 20 to 25 ° C. for 7 days.
- the inhibition circle length against the growth of Trichophyton rubrum inoculated on the agar in the lower receiver of the Turtube cell was measured, and the anti-tinea activity of the test preparation that permeated the human nail was confirmed.
- the external liquid preparation of Production Example 44 formed a blocking circle having a size depending on the concentration of the active substance (2%, 5%, 10% and 15%).
- a clinical product of cyclopirox and a clinical product of amorolfine were applied, no inhibition circle was formed. From the above, it was found that the liquid preparation for external use of the present application permeates the human nail and exhibits anti-tinea activity in a concentration-dependent manner.
- the permeable anti- ringworm activity of bovine nails of the externally applied solution of the present invention was measured as follows. After applying 2 ⁇ L of the test preparation prepared in 5 (2) to the 0.1mm thick bovine nail on the bovine nail in the cylindrical plastic and covering with parafilm, Trichophyton Mentagrophytes (T mentagrophytes) Trichophyton mentagrophytes were cultured for 4 days at an incubation temperature of 28 ° C. on an agar medium inoculated with ATCC18748. Thereafter, the inhibition circle length against the growth was measured, and the anti-tinea fungus activity of the test preparation that permeated the bovine nail was confirmed.
- the external preparation containing the general formula (I) and polymer as active ingredients easily forms a film when applied to the skin, etc., and has strong anti-tinea fungus activity. And it was confirmed that it has high stability, nail permeability, and the effect of suppressing the precipitation of active ingredients. It also has quick drying properties, and it was shown that both water-soluble polymers and water-insoluble polymers are preferable as the additive polymer.
- copolyvidone, povidone, acrylic resin alkanolamine, and aminoalkyl methacrylate copolymer are preferred. Further, it is also preferable to add a thickener, and a silicone derivative is particularly preferable.
- a nail and / or skin external solution having anti-tinea fungus activity and higher nail permeability is provided.
Landscapes
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Dermatology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
R1は、水素原子、C1-6アルキル、またはトリフルオロメチルを表し、
R2は、水素原子、C1-6アルキル、ハロゲン、-COO(C1-6アルキル)、または(CH2)1-3COOR(Rは、水素原子またはC1-6アルキルを表す)を表し、
R3は、水素原子、C1-6アルキル、アミノ、トリフルオロメチル、またはOR(Rは、水素原子またはC1-6アルキルを表す)を表し、
R4は、水酸基を表し、
R5は、水素原子、C1-6アルキル、水酸基、またはハロゲンを表し、
R6は、水素原子、C1-6アルキル、トリフルオロメチル、ハロゲン、アミノ、-NRaRb、ニトロ、ヒドロキシC1-6アルキル、-CONRaRb、-COO(C1-6アルキル)、-COOH、-(CH2)1-3COOR、またはORa(Rは、水素原子またはC1-6アルキルを表し、RaおよびRbは、同一でも異なっていてもよく、水素原子、C1-6アルキル、またはC1-6アシルを表す)を表し、
R7は、水素原子、C1-6アルキル、-OR(Rは、水素原子またはC1-6アルキルを表す)、またはハロゲンを表し、
R8は、水素原子、C1-6アルキル、水酸基、アミノ、またはニトロを表す。)で示される化合物またはその塩と、を含有する、白癬菌症の予防または治療用の爪および/または皮膚用外用液剤に関する。
上述のとおり、本発明は、皮膜形成剤と、一般式
R1が、水素原子またはC1-6アルキルであり、
R2が、水素原子、C1-6アルキル、ハロゲン、または-(CH2)1-3COOR(Rは、水素原子またはC1-6アルキルを表す)であり、
R3が、水素原子、またはC1-6アルキルであり、
R4が、水酸基であり、
R5が、水素原子であり、
R6が、水素原子、C1-6アルキル、トリフルオロメチル、ハロゲン、アミノ、-NRaRb、ニトロ、ヒドロキシC1-6アルキル、-CONRaRb、-COOH、またはORa(RaおよびRbは同一でも異なっていてもよく、水素原子、C1-6アルキル、またはC1-6アシルを表す)を表し、
R7が、水素原子であり、
R8が、水素原子、C1-6アルキル、アミノ、またはニトロである化合物である(請求項2に対応)。
R1が、C1-6アルキルであり、
R2が、水素原子、C1-6アルキル、またはハロゲンであり、
R3が、C1-6アルキルであり、
R4が、水酸基であり、
R5が、水素原子であり、
R6が、水素原子、C1-6アルキル、ハロゲン、またはORa(Raは、水素原子、C1-6アルキル、またはC1-6アシルを表す)を表し、
R7が、水素原子であり、
R8が、水素原子である化合物である(請求項3に対応)。
製造例1 2-(3,5-ジメチル-1H-ピラゾ-ル-1-イル)フェノール
製造例2 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-4-フルオロフェノール
製造例3 2-(1H-ピラゾール-1-イル)フェノール
製造例4 2-(5-ヒドロキシ-3-メチル-1H-ピラゾール-1-イル)フェノール
製造例5 2-(5-メチル-3-トリフルオロメチル-1H-ピラゾール-1-イル)フェノール
製造例6 2-(3,5-ビストリフルオロメチル-1H-ピラゾール-1-イル)フェノール
製造例7 2-(3-メチル-1H-ピラゾール-1-イル)フェノール
製造例8 2-(5-メチル-1H-ピラゾール-1-イル)フェノール
製造例9 2-(3,4,5-トリメチル-1H-ピラゾール-1-イル)フェノール
製造例10 2-(5-アミノ-3-tert-ブチル-1H-ピラゾール-1-イル)フェノール
製造例11 4-クロロ-2-(3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例12 2-クロロ-6-(3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例13 2-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例14 2-(3,5-ジエチル-1H-ピラゾール-1-イル)フェノール
製造例15 3-(3,5-ジメチル-1H-ピラゾール-1-イル)ベンゼン-1,2-ジオール
製造例16 2-(3,5-ジメチル-1H-ピラゾール-1-イル)ベンゼン-1,4-ジオール
製造例17 2-(4-エチル-3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例18 5-フルオロ-2-(3,4,5,-トリメチル-1H-ピラゾール-1-イル)フェノール
製造例19 2-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)ベンゼン-1,4-ジオール
製造例20 4-フルオロ-2-(3,4,5,-トリメチル-1H-ピラゾール-1-イル)フェノール
製造例21 2-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)-5-フルオロフェノール
製造例22 1-(2-ヒドロキシフェニル)-3,5-ジメチル-1H-ピラゾール-4-カルボン酸エチル
製造例23 3-(1-(2-ヒドロキシフェニル)-3,5-ジメチル-1H-ピラゾール-4-イル)プロパン酸メチル
製造例24 2-(4-ブチル-3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例25 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-フルオロフェノール
製造例26 5-クロロ-2-(3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例27 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ニトロフェノール
製造例28 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-ニトロフェノール
製造例29 3-(1-(2-ヒドロキフェニル)-3,5-ジメチル-1H-ピラゾール-4-イル)プロピオン酸
製造例30 5-クロロ-2-(3,4,5-トリメチル-1H-ピラゾール-1-イル)フェノール
製造例31 5-アミノ-2-(3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例32 5-ニトロ-2-(3,4,5-トリメチル-1H-ピラゾール-1-イル)フェノール
製造例33 4-(3,5-ジメチル-1H-ピラゾール-1-イル)ベンゼン-1,3-ジオール
製造例34 5-アミノ-2-(3,4,5-トリメチル-1H-ピラゾール-1-イル)フェノール
製造例35 4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンゼンカルボン酸メチル
製造例36 3-アミノ-2-(3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例37 4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンゼンカルボン酸
製造例38 4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシ-N,N-ジメチルベンズアミド
製造例39 4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンズアミド
製造例40 3-ヒドロキシ-4-(3,4,5-トリメチル-1H-ピラゾール-1-イル)ベンゼンカルボン酸
製造例41 3-ヒドロキシ-4-(3,4,5-トリメチル-1H-ピラゾール-1-イル)ベンズアミド
製造例42 4-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンズアミド
製造例43 2-(3,5-ジメチル-1H-ピラゾール-1-イル)ベンゼン-1,3-ジオール
製造例44 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-メチルフェノール
製造例45 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-メトキシフェノール
製造例46 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-メチルフェノール
製造例47 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-ヒドロキシメチルフェノール
製造例48 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-メチルアミノフェノール
製造例49 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-4-メチルフェノール
製造例50 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-トリフルオロメチルフェノール
製造例51 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-6-メチルフェノール
製造例52 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-エチルフェノール
製造例53 2-(4-フルオロ-3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例54 5-ブロモ-2-(3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例55 5-ブロモ-2-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)フェノール
製造例56 5-ブロモ-2-(3,4,5-トリメチル-1H-ピラゾール-1-イル)フェノール
製造例57 4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシフェニル アセテート
製造例58 4-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシフェニル アセテート
製造例59 3-ヒドロキシ-4-(3,4,5-トリメチル-1H-ピラゾール-1-イル)フェニル アセテート
製造例60 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-4-メトキシ-5-メチルフェノール
製造例61 4-クロロ-2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-メチルフェノール
製造例62 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-4,5-ジメチルフェノール
製造例63 4-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)-ベンゼン-1,3-ジオール
本発明の外用液剤に含有される一般式(I)で表される2-(1H-ピラゾール-1-イル)フェノール誘導体は、任意の方法で合成することができるが、例えばスキーム1に示す方法またはこれに準じた方法によって合成することができる。なお、スキーム1中、化合物の各記号は、前述のとおりであり、PおよびP'は水素または適当な保護基を表し、Xはハロゲンまたは適当なボロン酸基を表し、Yは反応に用いた酸の解離イオンを表す。本明細書中、「適当なボロン酸」とは、ボロン酸またはボロン酸エステルを表す。
本発明の白癬菌症の予防または治療用の爪および/または皮膚用外用液剤は、上述の一般式(I)で示される化合物またはその塩を皮膜形成剤と共に含有する。このような皮膜形成剤としては、爪または皮膚用外用液剤において一般的に使用される任意の皮膜形成剤を用いることができる。このような皮膜形成剤としては、溶媒および添加剤を含有する。添加剤としては、例えばポリマーが挙げられ、溶媒としては有機溶媒などを挙げることができる。
ポリマーとしては、水溶性ポリマーおよび水不溶性ポリマーなどが挙げられる。
水溶性ポリマーとしては、コポリビドン、ポビドン、ヒドロキシプロピルセルロース、または、ヒプロメロース、ポリビニルアルコールなどが挙げられ、水不溶性ポリマーとしては、アクリル樹脂アルカノールアミン、または、アミノアルキルメタクリレートコポリマーRS、アミノアルキルメタクリレートコポリマーE、ポリビニルアセタールジエチルアミノアセテートなどが挙げられる。
溶媒としては、低級アルコール、酢酸エチル、酢酸ブチルなどが挙げられ、低級アルコールとしてはメタノール、エタノール、イソプロパノールなどが挙げられる。好ましくはエタノールが挙げられる。
水溶性ポリマー含有皮膜形成剤としては、例えば、コポリビドン(Ashland社製 プラスドンS-630、または、BASF社製 コリドンVA64)、ポビドン(Ashland社製 プラスドンK-12、プラスドンK-17、 プラスドンK-25、プラスドンK-29/32、プラスドンK-90、プラスドンK-90D)、ヒドロキシプロピルセルロース(日本曹達製 HPC-SSL、HPC-SL、HPC-L)、ヒプロメロース(信越化学工業製 TC-5E、TC-5R、TC-5M)、ポリビニルアルコール(日本合成化学製 ゴーセノール)が挙げられる。
水不溶性ポリマー含有皮膜形成剤としては、例えば、アクリル樹脂アルカノールアミン(互応化学工業製 プラスサイズ L-53シリーズ、プラスサイズ L-9000シリーズ)、アミノアルキルメタクリレートコポリマーRS(エボニック社製 オイドラギットRS、オイドラギットRL等)、アミノアルキルメタクリレートコポリマーE(エボニック社製 オイドラギットE)、ポリビニルアセタールジエチルアミノアセテート(三共製 AEA三共)などが挙げられる。
本発明の爪および/または皮膚用外用液剤における一般式(I)の化合物またはその塩の配合割合は、化合物の種類によって適宜決定することができる。一般式(I)の化合物またはその塩を水溶性ポリマー含有皮膜形成剤、または、水不溶性皮膜形成剤と共に配合する場合には、一般式(I)の化合物またはその塩を、該液剤量全体に対して、全質量基準で好ましくは1~50質量%、より好ましくは1~30質量%、更により好ましくは1~20質量%、特に好ましくは5~15質量%配合する。
その他の添加剤
本発明の爪および/または皮膚用外用液剤には、上記皮膜形成剤の他に、通常外用剤等に用いられる防腐剤、溶解剤、透過促進剤、酸化防止剤、粘稠剤などを含有させることができる。
溶解剤としては、例えば、多価アルコール類(グリセリン、ソルビトール、エチレングリコール、プロピレングリコール、ジプロピレングリコール、1,3-ブチレングリコール、1,3-テトラメチレングリコール、ポリエチレングリコール等)、フェノール類(チモール、サフロール、イソサフロール、オイゲノール、イソオイゲノール等)、高級アルコール類(ベンジルアルコール、オレイルアルコール、セチルアルコール、ステアリルアルコール、セトステアリルアルコール、オクチルドデカノール等)、エステル系界面活性剤(セスキオレイン酸ソルビタン、ポリオキシエチレン硬化ヒマシ油、ステアリン酸ポリオキシル等)、脂肪酸エステル類(ミリスチン酸イソプロピル、ミリスチン酸オクチルドデシル、オレイン酸オレイル、フタル酸ジエチル、フタル酸ジブチル等)、(メタ)アクリル酸エステル類(アミノアルキルメタクリレートコポリマー等)、および有機酸類(乳酸、クエン酸、酒石酸、マレイン酸、リンゴ酸等)などの溶解剤を挙げることができる。
触感を向上させる粘稠剤としては、例えば、シリコーン誘導体(環状シリコーン、ジメチルポリシロキサン等)、疎水化ヒドロキシプロピルメチルセルロース、ポロクサマーなどが挙げられる。したがって、1つの実施形態において、本発明の爪および/または皮膚用外用液剤は、粘稠剤、好ましくはシリコーン誘導体、疎水化ヒドロキシプロピルメチルセルロース、またはポロクサマーを含む。
本発明の外用液剤を製造するには、溶媒の一部に有効成分である一般式(I)の化合物またはその塩、および、皮膜形成剤等を順に溶解させ、溶媒で量を調整し製剤を得た。また、本発明の外用液剤を製造するには、通常の外用液剤を製造する方法を適宜用いることができる。以下で、本発明を実施例によって説明するが、本発明はこれらの実施例に限定されるものではない。なお、実施例中、「%」は、「質量%」を意味する。また、実施例中の「室温」は通常約1℃から約40℃を示す。
製造例1 2-(3,5-ジメチル-1H-ピラゾ-ル-1-イル)フェノール
a)1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩 3.50gをエタノール60mlに溶解し、アセチルアセトン2.06mlを加えて1時間加熱還流した。反応混合物に水150mlを加え、飽和炭酸ナトリウム水溶液で中和し、酢酸エチル150mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=2:1)で精製し、表題化合物3.88gを得た。
1H-NMR(CDCl3);δ(ppm) 2.08(3H, s), 2.29(3H, s), 3.78(3H, s), 5.95(1H, s), 6.98-7.03(2H, m), 7.29-7.32(1H, m), 7.34-7.39(1H, m).
MS(ESI); m/z 203(M+H)+
1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール3.88gを塩化メチレン40mlに溶解し、三臭化ホウ素1M塩化メチレン溶液32mlを加えて室温にて1.5時間攪拌した。反応混合物を水150mlに加え、1N-水酸化ナトリウムで中和し、酢酸エチル150mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=3:1)で精製し、表題化合物2.83gを得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H,s), 2.38(3H, s), 6.02(1H, s), 6.87-6.6.91(1H, m), 7.06-7.09(1H, m), 7.16-7.20(1H, m).
MS(ESI); m/z 189(M+H)+
a)2-アミノ-4-フルオロフェノール
4-フルオロ-2-ニトロフェノール300mgをエタノール3mlに溶解し、10%パラジウム/炭素120mgを加え、水素雰囲気下、室温にて1時間攪拌した。不溶物を濾過した後、濾液を減圧下留去して、表題化合物を211mg得た。
1H-NMR(DMSO-d6);δ(ppm) 4.80(2H, s), 6.09-6.14(1H, m), 6.34-6.37(1H, m), 6.53-6.57(1H, m), 8.93(1H, s).
MS(FAB); m/z 128(M+H)+
2-アミノ-4-フルオロフェノール100mgに5N-塩酸0.8mlを加え、亜硝酸ナトリウム 65mgを水 0.2mlに溶解した溶液を0℃にて滴下し、30分攪拌した。次いで、塩化第一スズ 249mgを5N-塩酸0.46mlに溶解した溶液を0℃にて滴下し、0℃にて30分、次いで室温にて2時間攪拌した。溶媒を減圧下にて留去し、エタノール2.5mlおよびアセチルアセトン81μlを加えて4時間加熱還流した。反応混合物に水50mlを加え、飽和炭酸水素ナトリウム溶液で中和し、酢酸エチル50mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=2:1)で精製し、表題化合物を20.6mg得た。
1H-NMR(CDCl3);δ(ppm) 2.24(3H, s), 2.33(3H, s), 5.99(1H, s), 6.82-6.90(2H, m), 6.95-6.98(1H, m).
MS(FAB); m/z 207(M+H)+
a)1-(2-メトキシフェニル)-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩 200mgをエタノール5mlに溶解し、マロンアルデヒド ビスジメチルアセタール)189μlを加えて2時間加熱還流した。反応混合物に水50mlを加え、飽和炭酸ナトリウム水溶液で中和し、酢酸エチル60mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去して、表題化合物179.4mgを得た。
1H-NMR(CDCl3);δ(ppm) 3.87(3H,s), 6.42(1H, d, J=2.4Hz), 7.02-7.07(2H, m), 7.27-7.32(1H, m), 7.68-7.72(2H, m), 8.01(1H, d, J=2.4Hz).
MS(FAB); m/z 175(M+H)+
1-(2-メトキシフェニル)-1H-ピラゾール178mgより製造例1b)と同様の方法にて表題化合物を121mg得た。
1H-NMR(CDCl3);δ(ppm) 6.49(1H, d, J=2.4Hz), 6.88-6.92(1H, m), 7.08-7.10(1H, m), 7.14-7.18(1H, m), 7.35-7.37(1H, m), 7.72(1H, s), 7.99(1H, d, J=2.4Hz).
MS(ESI); m/z 161(M+H)+
a)5-ヒドロキシ-1-(2-メトキシフェニル)-3-メチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩150mgおよびアセト酢酸メチル93μlより製造例1a)と同様の方法にて表題化合物を55.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.11(3H, s), 3.76(3H, s), 6.88-6.94(3H, m), 7.19-7.34(2H, m).
MS(ESI); m/z 204(M+H)+
5-ヒドロキシ-1-(2-メトキシフェニル)-3-メチル-1H-ピラゾール52mgより製造例1b)と同様の方法にて表題化合物を32.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.17(3H, s), 5.30(1H, s), 6.88-6.95(2H, m), 7.15(1H, t, J=7.6Hz), 7.37(1H, d, J=7.6Hz).
MS(FAB); m/z 191(M+H)+
a)1-(2-メトキシフェニル)-5-メチル-3-トリフルオロメチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩300mgを2-メトキシエタノール1.3mlに溶解し、酢酸2.5mlおよび1,1,1-トリフルオロ-2,4-ペンタンジオン208μlを加えて1時間40分加熱還流した。溶媒を減圧下留去し、酢酸エチル50mlを加え、飽和炭酸水素ナトリウム溶液50mlおよび、飽和食塩水50mlで洗浄た有機層を無水硫酸マグネシウムで乾燥させ、減圧下溶媒を留去して、表題化合物485.6mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.14(3H, s), 3.78(3H, s), 6.40(1H, s), 7.00-7.07(2H, m), 7.31-7.33(1H, m), 7.41-7.45(1H, m).
MS(ESI); m/z 257(M+H)+
1-(2-メトキシフェニル)-5-メチル-3-(トリフルオロメチル)-1H-ピラゾールより製造例1b)と同様の方法にて表題化合物を320.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.41(3H, s), 6.52(1H, s), 6.95-6.99(1H, m), 7.11-7.13(1H, m), 7.20-7.24(1H, m), 7.28-7.32(1H, m).
MS(FAB); m/z 243(M+H)+
a)1-(2-メトキシフェニル)-3,5-ビストリフルオロメチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩300mgおよびヘキサフルオロアセチルアセトン243μlより製造例5a)と同様の方法にて表題化合物を得た。
1H-NMR(CDCl3);δ(ppm) 3.77(3H, s), 7.01-7.06(3H, m), 7.33(1H, d, J=7.6Hz), 7.49(1H, d, J=7.6Hz).
MS(ESI); m/z 311(M+H)+
上記a)で得られた1-(2-メトキシフェニル)-3,5-ビストリフルオロメチル)-1H-ピラゾールより製造例1b)と同様の方法にて表題化合物を455.6mg得た。
1H-NMR(CDCl3);δ(ppm) 6.99-7.04(1H, m), 7.07-7.08(1H, m), 7.10(1H, s), 7.32-7.41(2H, m).
MS(FAB); m/z 297(M+H)+
a)1-(2-メトキシフェニル)-3-メチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩200mgおよび4,4-ジメトキシブタン-2-オン151μlより製造例1a)と同様の方法にて表題化合物を115.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.31(3H, s), 3.80(3H, s), 6.14(1H, d, J=2.4Hz), 6.95-6.99(2H, m), 7.18-7.22(1H, m), 7.61-7.63(1H, m), 7.84(1H, d, J=2.4Hz).
MS(FAB); m/z 189(M+H)+
1-(2-メトキシフェニル)-3-メチル-1H-ピラゾール115mgより製造例1b)と同様の方法にて表題化合物を76mg得た。
1H-NMR(CDCl3);δ(ppm) 2.36(3H, s), 6.24(1H, d, J=2.4Hz), 6.85-6.89(1H, m), 7.05-7.14(2H, m), 7.29-7.31(1H, m), 7.86(1H, d, J=2.4Hz).
MS(FAB); m/z 175(M+H)+
a)1-(2-メトキシフェニル)-5-メチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩200mgおよび4,4-ジメトキシブタン-2-オン151μlより製造例1a)と同様の方法にて表題化合物を70mg得た。
1H-NMR(CDCl3);δ(ppm) 2.07(3H, s), 3.71(3H, s), 6.09(1H, s), 6.94-6.99(2H, m), 7.24-7.26(1H, m), 7.31-7.35(1H, m), 7.52(1H, s).
MS(FAB); m/z 189(M+H)+
1-(2-メトキシフェニル)-5-メチル-1H-ピラゾール69mgより製造例1b)と同様の方法にて表題化合物を45.5mg得た。
1H-NMR(CDCl3);δ(ppm) 2.41(3H, s), 6.27(1H, d, J=1.6Hz), 6.90-6.94(1H, m), 7.10-7.12(1H, m), 7.19-7.24(2H, m), 7.66(1H, d, J=1.6Hz).
MS(FAB); m/z 175(M+H)+
a)1-(2-メトキシフェニル)-3,4,5-トリメチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩 200mgをエタノール4mlに溶解し、3-メチル-2,4-ペンタンジオン134μlを加えて3時間加熱還流した。反応混合物に水50mlを加え、飽和炭酸ナトリウム水溶液で中和し、酢酸エチル60mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去して、表題化合物を209.8mg得た。
1H-NMR(CDCl3);δ(ppm) 1.98(3H, s), 2.02(3H, s), 2.24(3H, s), 3.80(3H, s), 6.99-7.04(2H, m), 7.29-7.31(1H, m), 7.34-7.39(1H, m).
MS(FAB); m/z 217(M+H)+
1-(2-メトキシフェニル)-3,4,5-トリメチル-1H-ピラゾール209mgより製造例1b)と同様の方法にて表題化合物を104mg得た。
1H-NMR(CDCl3);δ(ppm) 1.99(3H, s), 2.25(3H, s), 2.31(3H, s), 6.90(1H, t, J=8.0Hz), 7.08(1H, d, J=8.0Hz), 7.06-7.20(2H, m), 9.89(1H, s).
MS(ESI); m/z 203(M+H)+
a)3-tert-ブチル-1-(2-メトキシフェニル)-1H-ピラゾール-5-アミン
2-メトキシフェニルヒドラジン塩酸塩300mgおよび4,4-ジメチル-3-オキソペンタンニトリル215mgおよび酢酸40μlより製造例1a)と同様の方法にて表題化合物を310.3mg得た。
1H-NMR(CDCl3);δ(ppm) 1.32(9H, s), 3.80(2H, s), 3.86(3H, s), 5.51(1H, s), 7.01-7.08(2H, m), 7.30-7.35(1H, m), 7.45-7.47(1H, m).
MS(FAB); m/z 246(M+H)+
3-tert-ブチル-1-(2-メトキシフェニル)-1H-ピラゾール-5-アミン100mgより製造例1b)と同様の方法にて表題化合物を66.2mg得た。
1H-NMR(CDCl3);δ(ppm) 1.30(9H, s), 3.93(2H, s), 5.55(1H, s), 6.90(1H, dt, J=1.6, 8.0Hz), 7.08(1H, dd, J=1.6, 8.0Hz), 7.17(1H, dt, J=1.6, 8.0Hz), 7.47(1H, dd, J=1.6, 8.0Hz), 10.39(1H, brs).
MS(FAB); m/z 232(M+H)+
a)1-(5-クロロ-2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール
5-クロロ-2-メトキシアニリン塩酸塩 388mgより製造例2b)と同様の方法にて表題化合物を104.8mg得た。
1H-NMR(CDCl3);δ(ppm) 2.11(3H, s), 2.29(3H, s), 3.79(3H, s), 5.96(1H, s), 6.93(1H, dd, J=2.4, 7.2Hz), 7.33-7.35(2H, m).
MS(FAB); m/z 237(M+H)+
1-(5-クロロ-2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール104.8mgより製造例1b)と同様の方法にて表題化合物を67.3mg得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.43(3H, s), 6.05(1H, s), 7.02(1H, d, J=8.8Hz), 7.14-7.20(2H, m), 10.08(1H, s).
MS(FAB); m/z 223(M+H)+
a)1-(3-クロロ-2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール
3-クロロ-o-アニシジン158mgより製造例2b)と同様の方法にて表題化合物を30.6mg得た。
1H-NMR(CDCl3);δ(ppm) 2.14(3H, s), 2.29(3H, s), 3.49(3H, s), 5.99(1H,s), 7.12(1H, t, J=8.0Hz), 7.27-7.31(1H, m), 7.43-7.46(1H, m).
MS(ESI); m/z 236(M+H)+
1-(3-クロロ-2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール63.4 mgより製造例1b)と同様の方法にて表題化合物を27.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.40(3H, s), 6.05(1H, s), 6.86(1H, t, J=8.0Hz),7.13-7.15(1H, m), 7.26-7.31(1H, m), 10.66(1H, s).
MS(FAB); m/z 223(M+H)+
a)4-クロロ-1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩174.6mg、および3-クロロペンタン-2, 4-ジオン114μlより、製造例1a)と同様の方法にて表題化合物を193.9mg得た。
1H-NMR(CDCl3);δ(ppm) 2.09(3H,s), 2.29(3H, s), 3.81(3H, s), 7.00-7.06(2H, m), 7.30(1H, dd, J=1.6, 7.6Hz), 7.38-7.43(1H, m).
MS(FAB); m/z 237(M+H)+
4-クロロ-1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール193mgより製造例1b)と同様の方法にて表題化合物を151.9mg得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.37(3H, s), 6.91-6.95(1H, m), 7.09(1H, dd, J=1.6, 8.0Hz), 7.17(1H, dd, J=1.6, 8.0Hz), 7.21-7.25(1H, m), 9.23(1H, s).
MS(FAB); m/z 223(M+H)+
a)3,5-ジエチル-1-(2-メトキシフェニル)- 1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩174.6mg、および3,5-ヘプタンジオン135.5μlより、製造例1a)と同様の方法にて表題化合物を209.8mg得た。
1H-NMR(CDCl3);δ(ppm) 1.14-1.18(3H, m), 1.25-1.31(3H, m), 2.42(2H, q, J=7.2Hz), 2.67-2.73(2H, m), 3.78(3H, s), 6.03(1H, s), 6.99-7.04(2H, m), 7.31-7.40(2H, m).
MS(FAB); m/z 231(M+H)+
3,5-ジエチル-1-(2-メトキシフェニル)-1H-ピラゾール207mgより製造例1b)と同様の方法にて表題化合物を159.2mg得た。
1H-NMR(CDCl3);δ(ppm) 1.24-1.31(6H, m), 2.69(2H, q, J=7.6Hz), 2.76(2H, q, J=7.6Hz), 6.11(1H, s), 6.90(1H, q, J=7.6Hz), 7.09(1H, d, J=7.6Hz), 7.19-7.22(2H, m), 9.69(1H, s).
MS(FAB); m/z 217(M+H)+
a)1-(2,3-ジメトキシフェニル)-3,5-ジメチル-1H-ピラゾール
2,3-ジメトキシアニリン306mgより製造例2b)と同様の方法にて表題化合物を189mg得た。
1H-NMR(CDCl3);δ(ppm) 2.14(3H, s), 2.29(3H, s), 3.53(3H, s), 3.91(3H, s), 5.96(1H, s), 6.96-6.70(2H, m), 7.12(1H, t, J=8.0Hz).
MS(FAB); m/z 233(M+H)+
1-(2,3-ジメトキシフェニル)-3,5-ジメチル-1H-ピラゾール186mgより製造例1b)と同様の方法にて表題化合物を28.9mg得た。
1H-NMR(CDCl3);δ(ppm) 2.26(3H, s), 2.38(3H, s), 5.99(1H, s), 6.75-6.85(3H, m).
MS(FAB); m/z 205(M+H)+
a)1-(2,5-ジメトキシフェニル)-3,5-ジメチル-1H-ピラゾール
2,5-ジメトキシアニリン306mgより製造例2b)と同様の方法にて表題化合物を260.3mg得た。
1H-NMR(CDCl3);δ(ppm) 2.11(3H, s), 2.30(3H, s), 3.73(3H, s), 3.78(3H, s), 5.96(1H, s), 6.91-6.93(3H, m).
1-(2,5-ジメトキシフェニル)-3,5-ジメチル-1H-ピラゾール260.3mgより製造例1b)と同様の方法にて表題化合物を162.6mg得た。
1H-NMR(DMSO-d6);δ(ppm) 2.07(3H, s), 2.14(3H, s), 5.93(1H, s), 6.56(1H, d, J=2.8Hz), 6.66-6.69(1H, m), 6.80(1H, d, J=8.8Hz), 9.02(1H, s), 9.17(1H, s).
MS(FAB); m/z 205(M+H)+
a)4-エチル-1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩200mg、および3-エチル-2,4-ペンタンジオン155μlより、製造例1a)と同様の方法にて表題化合物を236.6mg得た。
1H-NMR(CDCl3);δ(ppm) 1.13(3H, t, J=7.6Hz), 2.03(3H, s), 2.27(3H, s), 2.42(2H, q, J=7.6Hz), 3.79(3H, s), 6.99-7.04(2H, m), 7.30-7.39(2H, m).
MS(FAB); m/z 231(M+H)+
4-エチル-1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール232mgより製造例1b)と同様の方法にて表題化合物を196.2mg得た。
1H-NMR(CDCl3);δ(ppm) 1.12(3H, t, J=7.6Hz), 2.27(3H, s), 2.33(3H, s), 2.43(2H, q, J=7.6Hz), 6.88-6.92(1H, m), 7.08-7.10(1H, m), 7.06-7.19(2H, m), 9.90(1H, s).
MS(FAB); m/z 217(M+H)+
2-アミノ-5-フルオロフェノール201.2mgおよび3-メチル-2,4-ペンタンジオン184μlより製造例1b)と同様の方法にて表題化合物を114.7mg得た。
1H-NMR(CDCl3);δ(ppm) 1.99(3H, s), 2.23(3H, s), 2.28(3H, s), 6.59-6.64(1H, m), 6.78-6.81(1H, m), 7.10-7.14(1H, m).
MS(ESI); m/z 221(M+H)+
a)4-クロロ-1-(2,5-ジメトキシフェニル)-3,5-ジメチル-1H-ピラゾール
2,5-ジメトキシアニリン306mgおよび3-クロロペンタン-2,4-ジオン228μlより製造例2b)と同様の方法にて表題化合物を333.7mg得た。
1H-NMR(CDCl3);δ(ppm) 2.10(3H, s), 2.29(3H, s), 3.74(3H, s), 3.78(3H, s), 6.94-6.97(2H, m), 7.26(1H, s).
MS(FAB); m/z 267(M+H)+
4-クロロ-1-(2,5-ジメトキシフェニル)-3,5-ジメチル-1H-ピラゾール329mgより製造例1b)と同様の方法にて表題化合物を184.8mg得た。
1H-NMR(DMSO-d6);δ(ppm) 2.05(3H, s), 2.15(3H, s), 6.58(1H, d, J=2.8Hz), 6.72(1H, dd, J=2.8, 8.8Hz), 6.82(1H, d, J=8.8Hz), 9.11(1H, s), 9.33(1H, s).
MS(FAB); m/z 239(M+H)+
2-アミノ-4-フルオロフェノール111mgおよび3-メチル-2,4-ペンタンジオン102μlより製造例2b)と同様の方法にて表題化合物を84.8mg得た。
1H-NMR(CDCl3);δ(ppm) 1.99(3H, s), 2.23(3H, s), 2.24(3H, s), 2.34(3H, s), 6.87-6.95(2H, m), 7.00-7.03(1H, m), 9.90(1H, s).
MS(FAB); m/z 221(M+H)+
2-アミノ-5-フルオロフェノール100mgおよび3-クロロペンタン-2,4-ジオン90μlより製造例2b)と同様の方法にて表題化合物を62mg得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.35(3H, s), 6.62-6.67(1H, m), 6.78-6.81(1H, m), 7.10-7.14(1H, m), 9.44(1H, s).
MS(FAB); m/z 241(M+H)+
a)1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール-4-カルボン酸エチル
2-メトキシフェニルヒドラジン塩酸塩175mg、および2-アセチル-3-オキソブタン酸エチル156μlより、製造例1a)と同様の方法にて表題化合物を82.6mg得た。
1H-NMR(CDCl3);δ(ppm) 1.38(3H, t, J=7.6Hz), 2.33(3H, s), 2.50(3H, s), .80(3H, s), 4.32(2H, q, J=7.6Hz), 7.02-7.08(2H, m), 7.30-7.32(1H, m), 7.41-7.45(1H, m).
MS(ESI); m/z 275(M+H)+
1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール-4-カルボン酸エチル82mgより製造例1b)と同様の方法にて表題化合物を21mg得た。
1H-NMR(CDCl3);δ(ppm) 1.39(3H, t, J=7.2Hz), 2.50(3H, s), 2.61(3H, s), 4.34(2H, q, J=7.2Hz), 6.96(1H, t, J=8.4Hz), 7.10(1H, d, J=8.4Hz), 7.17(1H, d, J=8.4Hz), 7.26-7.30(1H, m), 8.76(1H, s).
MS(FAB); m/z 261(M+H)+
a)3-(1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール-4-イル)プロパン酸メチル
2-メトキシフェニルヒドラジン塩酸塩174.6mg、および4-アセチル-5-オキソヘキサン酸メチル175μlより、製造例1a)と同様の方法にて表題化合物を96mg得た。
1H-NMR(CDCl3);δ(ppm) 2.04(3H, s), 2.27(3H, s), 2.49-2.54(2H, m), 2.74-2.78(2H, m), 3.68(3H, s), 3.79(3H, s), 6.99-7.02(2H, m), 7.28-7.31(1H, m), 7.35-7.39(1H, m).
MS(FAB); m/z 289(M+H)+
3-(1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール-4-イル)プロパン酸メチル96mg より製造例1b)と同様の方法にて表題化合物を36.8mg得た。
1H-NMR(CDCl3);δ(ppm) 2.28(3H, s), 2.34(3H, s), 2.51(2H, t, J=8.0Hz), 2.77(2H, t, J=8.0Hz), 3.69(3H, s), 6.91(1H, t, J=6.8Hz), 7.08-7.10(1H, m), 7.17-7.26(2H, m), 9.73(1H, s).
MS(ESI); m/z 275(M+H)+
a)4-ブチル-1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール
2-メトキシフェニルヒドラジン塩酸塩175mg、および3-n-ブチル2,4-ペンタンジオン168μlより、製造例1a)と同様の方法にて表題化合物を273mg得た。
1H-NMR(CDCl3);δ(ppm) 0.94(3H, t, J=7.2Hz), 1.32-1.39(2H, m), 1.45-1.50(2H, m), 2.01(3H, m), 2.26(3H, s), 2.39(2H, q, J=7.2Hz), 3.79(3H, s), 6.99-7.04(2H, m), 7.30-7.38(2H, m).
MS(FAB); m/z 259(M+H)+
4-ブチル-1-(2-メトキシフェニル)-3,5-ジメチル-1H-ピラゾール273mgより製造例1b)と同様の方法にて表題化合物を208.8mg得た。
1H-NMR(CDCl3);δ(ppm) 0.95(3H, t, J=7.6Hz), 1.33-1.39(2H, m), 1.43-1.48(2H, m), 2.26(3H, s), 2.32(3H, s), 2.40(2H, t, J=7.6Hz), 6.88-6.92(1H, m), 7.08(1H, d, J=8.4Hz), 7.16-7.20(2H, m), 9.93(1H, s).
MS(FAB); m/z 245(M+H)+
a)2-アミノ-5-フルオロフェノール
5-フルオロ-2-ニトロフェノール314mgより、製造例2a)と同様の方法にて表題化合物を252.5mg得た。
1H-NMR(DMSO-d6);δ(ppm) 6.33-6.38(1H, m), 6.45-6.48(1H, m), 6.51-6.55(1H, m).
MS(FAB); m/z 128(M+H)+
2-アミノ-5-フルオロフェノール150mgより製造例2b)と同様の方法にて表題化合物を89.6mg得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.37(3H, s), 6.03(1H, s), 6.60-6.65(1H, m), 6.78-6.81(1H, m), 7.13-7.16(1H, m).
MS(ESI); m/z 207(M+H)+
2-アミノ-5-クロロフェノール287mgより製造例2b)と同様の方法にて表題化合物を168.8mg得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.40(3H, s), 6.05(1H, s), 6.87-6.90(1H, m), 7.10-7.14(2H, m), 10.23(1H, s).
MS(FAB); m/z 223(M+H)+
2-アミノ-3-ニトロフェノール308mgより製造例2b)と同様の方法にて、表題化合物を75.4mg得た。
1H-NMR(CD3OD);δ(ppm) 2.14(3H, s), 2.19(3H, s), 6.04(1H, s), 7.27(1H, dd, J=1.6, 7.6Hz), 7.44(1H, dd, J=1.6, 7.6Hz), 7.50(1H, t, J=7.6Hz).
MS(FAB); m/z 234(M+H)+
2-アミノ-5-ニトロフェノール308mgより製造例2b)と同様の方法にて表題化合物を101mg得た。
1H-NMR(CDCl3);δ(ppm) 2.33(3H, s), 2.50(3H, s), 6.13(1H, s), 7.38(1H, d, J=7.6Hz), 7.79-7.81(1H, m), 7.95(1H, s).
M S(ESI); m/z 234(M+H)+
3-(1-(2-ヒドロキシフェニル)-3,5-ジメチル-1Hピラゾール-4-イル)プロパン酸メチル29.9mgをメタノール0.6mlに溶解し、1N-水酸化ナトリウム0.29mlを加えて室温にて3.5時間攪拌した。反応混合物に水20mlを加え、1N-塩酸で中和し、酢酸エチル20mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去して、表題化合物を7.5mg得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.34(3H, s), 2.56(2H, t, J=7.6Hz), 2.78(2H, t, J=7.6Hz), 6.89-6.93(1H, m), 7.08-7.10(1H, m), 7.16-7.22(2H, m).
MS(FAB); m/z 261(M+H)+
2-アミノ- 5-クロロフェノール287mgおよび3-メチル-2,4-ペンタンジオン233μlより製造例2b)と同様の方法にて表題化合物を152mg得た。
1H-NMR(CDCl3);δ(ppm) 1.99(3H, s), 2.24(3H, s), 2.31(3H, s), 6.86-6.89(1H, m), 7.09-7.11(2H, m).
MS(FAB); m/z 227(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-ニトロフェノール 86mgをエタノール1.7mlに溶解し、10%パラジウム/炭素43mgを加え、水素雰囲気下、室温にて45分攪拌した。不溶物を濾過した後、溶媒を減圧下留去して、表題化合物を30.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.28(3H, s), 2.33(3H, s), 3.72(2H, s), 5.99(1H, s), 6.22(1H, dd, J=2.4, 8.4Hz), 6.40(1H, d, J=2.4Hz), 6.97(1H, d, J=8.4Hz), 9.27(1H, s).
MS(FAB); m/z 204(M+H)+
2-アミノ-5-ニトロフェノール308mgおよび3-メチル-2,4-ペンタンジオン233μlより製造例2b)と同様の方法にて表題化合物を289.5mg得た。
1H-NMR(CDCl3);δ(ppm) 2.02(3H, s), 2.24(3H, s), 2.40(3H, s), 7.33(1H, d, J=8.8Hz), 7.79(1H, dd, J=2.4, 8.8Hz), 7.94(1H, d, J=2.4Hz).
MS(FAB); m/z 248(M+H)+
a)2,4-ジメトキシフェニルボロン酸
1-ブロモ-2,4-ジメトキシベンゼン576μlをテトラヒドロフラン5.8mlに溶解し、アルゴン雰囲気下-78℃にてn-ブチルリチウム1.6mol/l ヘキサン溶液3mlを滴下した。次いで、ホウ酸トリイソプロピル1.1mlを加え、-78℃にて40分攪拌した後、室温で2時間攪拌した。反応混合物に水40mlおよび5N-塩酸1mlを加え、酢酸エチル50mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=2:1)を用いて精製して表題化合物を610.2mg得た。
1H-NMR(CDCl3);δ(ppm) 3.85(3H, s), 3.89(3H, s), 5.81(2H, s), 6.46(1H, s), 6.56(1H, dd, J=2.0, 8.4Hz), 7.77(1H, d, J=8.4Hz).
2,4-ジメトキシフェニルボロン酸610.2mgを塩化メチレン6mlに溶解し、3,5-ジメチルピラゾール387mg、酢酸銅(II) 730mgおよびピリジン948μlを加えて、室温にて終夜攪拌した。反応混合物に水60mlを加え、酢酸エチル60mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=3:2)を用いて精製して表題化合物を81.7mg得た。
1H-NMR(CDCl3);δ(ppm) 2.07(3H, s), 2.29(3H, s), 3.77(3H, s), 3.85(3H, s), 5.94(1H, s), 6.52-6.54(2H, m), 7.22-7.24(1H, m).
MS(FAB); m/z 233(M+H)+
1-(2,4-ジメトキシフェニル)-3,5-ジメチル-1H-ピラゾール 118.4mgを塩化メチレン2.3mlに溶解し、三臭化ホウ素1M塩化メチレン溶液1.7mlを加えて室温にて1時間攪拌した。反応混合物を水30mlに加え、1N-水酸化ナトリウムで中和し、酢酸エチル50mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、分取用薄層シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=1:2)を用いて精製して表題化合物を71.7mg得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.33(3H,s), 6.01(1H, s), 6.37(1H, dd, J=2.8, 8.8Hz), 6.53(1H, d, J=2.8Hz), 7.03(1H, d, J=8.8Hz).
MS(ESI); m/z 205(M+H)+
5-ニトロ-2-(3,4,5-トリメチル-1H-ピラゾール-1-イル)フェノール200mgより製造例31と同様の方法にて、表題化合物を118.4mg得た。
1H-NMR(CDCl3);δ(ppm) 1.97(3H, s), 2.23(3H, s), 2.24(3H, s), 3.70(2H, s), 6.22(1H, dd, J=2.4, 8.4Hz), 6.40(1H, d, J=2.4Hz), 6.95(1H, d, J=8.4Hz), 9.34(1H, s).
MS(FAB); m/z 218(M+H)+
4-アミノ-3-ヒドロキシベンゼンカルボン酸メチル334mgより製造例2b)と同様の方法にて表題化合物を239.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.31(3H, s), 2.45(3H, s), 3.92(3H, s), 6.08(1H, s), 7.26-7.29(1H, m), 7.60(1H, dd, J=1.6, 8.0Hz), 7.76(1H, d, J=1.6Hz).
MS(ESI); m/z 247(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ニトロフェノール56.6mgより製造例31と同様の方法にて、表題化合物を38.9mg得た。
1H-NMR(CDCl3);δ(ppm) 2.14(3H, s), 2.25(3H, s), 3.57(2H, s), 6.02(1H, s), 6.32-6.37(2H, m), 7.02(1H, t, J=8.0Hz).
MS(FAB); m/z 204(M+H)+
4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンゼンカルボン酸メチル100mgをメタノール1mlに溶解し、1N-水酸化ナトリウム1.6mlを加えて室温にて3.5時間攪拌した。反応混合物に水20mlを加え、1N-塩酸で中和し、酢酸エチル20mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去して、表題化合物を40.8mg得た。
1H-NMR(CD3OD);δ(ppm) 2.16(3H, s), 2.24(3H, s), 6.04(1H, s), 7.30(1H, d, J=8.4Hz), 7.59(1H, dd, J=2.0, 8.4Hz), 7.64(1H, d, J=2.0Hz).
MS(ESI); m/z 233(M+H)+
4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンゼンカルボン酸36mgをジメチルホルムアミド0.4mlに溶解し、1-(3-ジメチルアミノプロピル)-3-エチルカルボジイミド塩酸塩36mg、1-ヒドロキシベンゾトリアゾール25mg、ジメチルアミン(2.0M THF溶液)93μlを加えて室温で4時間攪拌した。反応混合物に水10mlを加え、酢酸エチル15mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、分取用薄層シリカゲルカラムクロマトグラフィー(酢酸エチル)を用いて精製して表題化合物を11.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.31(3H, s), 2.41(3H, s), 3.02(3H, s), 3.11(3H, s), 6.06(1H, s), 6.97(1H, d, J=8.0Hz), 7.23(1H, d, J=8.0Hz), 7.26(1H, s).
MS(ESI); m/z 259(M+H)+
4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンゼンカルボン酸73.1mgおよびアンモニア水38μlより製造例38と同様の方法にて、表題化合物を34.3mg得た。
1H-NMR(CD3OD);δ(ppm) 2.16(3H, s), 2.24(3H, s), 6.04(1H, s), 7.29(1H, d, J=8.4Hz), 7.41 (1H, dd, J=2.0, 8.4Hz), 7.50(1H, d, J=2.0Hz).
MS(ESI); m/z 232(M+H)+
a)3-ヒドロキシ-4-(3,4,5-トリメチル-1H-ピラゾール-1-イル)-ベンゼンカルボン酸メチル
4-アミノ-3-ヒドロキシベンゼンカルボン酸メチル167mgおよび3-メチル-2,4-ペンタンジオン116μlより製造例2b)と同様の方法にて表題化合物を110.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.00(3H, s), 2.26(3H, s), 2.36(3H, s), 3.92(3H, s), 7.23-7.26(1H, m), 7.58-7.60(1H, m), 7.49(1H, s), 10.53(1H, s).
MS(ESI); m/z 261(M+H)+
3-ヒドロキシ-4-(3,4,5-トリメチル-1H-ピラゾール-1-イル)-ベンゼンカルボン酸メチル110mgより製造例37と同様の方法にて、表題化合物を68.7mg得た。
1H-NMR(CD3OD);δ(ppm) 1.99(3H, s), 2.09(3H, s), 2.20(3H, s), 7.28(1H, d, J=8.0Hz),7.59(1H, dd, J=2.0, 8.0Hz), 7.63(1H, d, J=2.0Hz).
MS(ESI); m/z 247(M+H)+
3-ヒドロキシ-4-(3,4,5-トリメチル-1H-ピラゾール-1-イル)ベンゼンカルボン酸65mgおよびアンモニア水32μlより製造例38と同様の方法にて、表題化合物を33.3mg得た。
1H-NMR(CD3OD);δ(ppm) 1.99(3H, s), 2.09(3H, s), 2.19(3H, s), 7.27(1H, d, J=8.0Hz),7.40(1H, dd, J=2.0, 8.0Hz), 7.57(1H, d, J=2.0Hz).
MS(ESI); m/z 246(M+H)+
a)4-(4-クロロ3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシ安息香酸メチル
4-アミノ-3-ヒドロキシ安息香酸メチル167mgおよび3-クロロペンタン-2,4-ジオン114μlより製造例2b)と同様の方法にて表題化合物を69.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.32(3H, s), 2.42(3H, s), 3.93(3H, s), 7.24-7.26(1H, m), 7.60-7.66(1H, m), 7.77(1H, s).
メチル 4-(4-クロロ3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシ安息香酸69.1mgより製造例37と同様の方法にて、表題化合物を39.2mg得た。
1H-NMR(CD3OD);δ(ppm) 2.15(3H, s), 2.24(3H, s), 7.32-7.35(1H, m), 7.59-7.66(2H, m).
MS(ESI); m/z 267(M+H)+
4-(4-クロロ-3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシ安息香酸37.9mgおよびアンモニア水1 7μlより製造例38と同様の方法にて、表題化合物を12.3mg得た。
1H-NMR(CD3OD);δ(ppm) 2.14(3H, s), 2.24(3H, s), 7.29-7.32(1H, m), 7.41-7.44(1H, m), 7.50(1H, s).
MS(ESI); m/z 266(M+H)+
a)2-アミノベンゼン-1,3-ジオール
2-ニトロレゾルシノール465mgをエタノール9.3mlに溶解し、10%パラジウム/炭素230mgを加え、水素雰囲気下、室温にて1時間攪拌した。不溶物を濾過した後、溶媒を減圧下留去して、表題化合物を338.5mg得た。
1H-NMR(DMSO-d6);δ(ppm) 6.20-6.28(3H, m).
MS(ESI); m/z 126(M+H)+
2-アミノベンゼン-1,3-ジオール100mgをジクロロメタン2mlに溶解し、トリエチルアミン 234μlおよびp-トルエンスルホニルクロライド 320mgを加え、室温にて1.5時間攪拌した。反応混合物に水20mlを加え、酢酸エチル20mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=4:1)を用いて精製して表題化合物を278.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.46(6H, s), 3.83(2H, s), 6.48(1H, t, J=8.1Hz), 6.79(2H, d, J=8.3Hz), 7.32(4H, d, J=8.1Hz), 7.72(4H, d, J=8.3Hz).
MS(ESI); m/z 434(M+H)+
2-アミノ-1,3-フェニレン ビス(4-メチルベンゼンスルホネート)278mgに5N-塩酸0.64mlを加え、亜硝酸ナトリウム 58mgを水 0.4mlに溶解した溶液を0℃にて滴下し、30分攪拌した。次いで、塩化第一スズ 289mgを5N-塩酸0.32mlに溶解した溶液を0℃にて滴下し、1時間攪拌した。溶媒を減圧下にて留去し、エタノール1.3mlおよびアセチルアセトン66μlを加えて2時間加熱還流した。反応混合物に水50mlを加え、飽和炭酸水素ナトリウム溶液で中和し、酢酸エチル50mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=4:1~1:1)を用いて精製して表題化合物を62.9mg得た。
1H-NMR(CDCl3);δ(ppm) 2.15(3H, s), 2.17(3H, s), 2.41(3H, s), 5.91(1H, s), 6.93-6.-96(2H, m), 7.14(2H, d, J=8.0Hz), 7.18-7.26(1H, m), 7.35(2H, d, J=8.0Hz).
MS(ESI); m/z 359(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシフェニル 4-メチルベンゼンスルホネート62mgに水酸化カリウム97mgをエタノール1.5mlおよび水1.5mlに溶解した溶液を加え、3.5時間加熱還流した。反応混合物に水20mlを加え、酢酸エチル20mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、分取用薄層シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=1:1)を用いて精製し、表題化合物を20.8mg得た。
1H-NMR(CDCl3);δ(ppm) 2.15(3H, s), 2.16(3H, s), 5.98(1H, s), 6.45(2H, d, J=8.4Hz), 7.00(1H, t, J=8.4Hz).
MS(ESI); m/z 205(M+H)+
a)2-アミノ-5-メチルフェニル 4-メチルベンゼンスルホネート
2-アミノ-5-メチルフェノール 400mgをジクロロメタン 6.5mlに溶解し、トリエチルアミン 476μlおよびp-トルエンスルホニルクロライド 619mgを加え、室温にて1時間攪拌した。反応混合物に水60mlを加え、酢酸エチル60mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=3:1)を用いて精製して表題化合物を730.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.15(3H, s), 2.46(3H, s), 3.63(2H, brs), 6.61-6.63(1H, m), 6.67(1H, s), 6.82-6.85(1H, m), 6.33(2H, d, J=8.4Hz), 7.78(2H, d, J=8.4Hz).MS(ESI); m/z 278(M+H)+
2-アミノ-5-メチルフェニル 4-メチルベンゼンスルホネート 453mgに5N-塩酸1.6mlを加え、亜硝酸ナトリウム 146mgを水 1mlに溶解した溶液を0℃にて滴下し、30分攪拌した。次いで、塩化第一スズ 736mgを5N-塩酸0.8mlに溶解した溶液を0℃にて滴下し、1時間攪拌した。溶媒を減圧下にて留去し、エタノール3.2mlおよびアセチルアセトン167μlを加えて2時間加熱還流した。反応混合物に水50mlを加え、飽和炭酸水素ナトリウム溶液で中和し、酢酸エチル80mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=2:1)を用いて精製して表題化合物を199.6mg得た。
1H-NMR(CDCl3);δ(ppm) 2.09(3H, s), 2.11(3H, s), 2.41(3H,s), 2.43(3H, s), 5.82(1H, s), 7.15-7.21(4H, m), 7.36-7.38(3H, m).
MS(ESI); m/z 357(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-メチルフェニル 4-メチルベンゼンスルホネート199.6mgに水酸化カリウム314 mgをエタノール4mlおよび水4mlに溶解した溶液を加え、1時間加熱還流した。反応混合物に水20mlを加え、酢酸エチル20mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、分取用薄層シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=4:1)を用いて精製し、所望の画分を1,4-ジオキサンにて溶解後、凍結乾燥することにより表題化合物を66.2mg得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.33(3H,s), 2.37(3H,s), 6.01(1H, s), 6.70(1H, d, J=8.0Hz), 6.90(1H, s), 7.07(1H, d, J=8.0Hz), 9.64(1H, brs).
MS(ESI); m/z 203(M+H)+
a)2-アミノ-5-メトキシフェニル 4-メチルベンゼンスルホネート
2-ヒドロキシ-4-メトキシアニリン 塩酸塩176mgより、製造例44 a)と同様の方法にて表題化合物を220.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.46(3H, s), 3.62(3H, s), 6.41(1H, s), 6.65-6.66(2H, m), 7.34(2H, d, J=8.8Hz), 7.79(2H, d, J=8.8Hz).
2-アミノ-5-メトキシフェニル 4-メチルベンゼンスルホネート220mgより、製造例44b)と同様の方法にて表題化合物を93.7mg得た。
1H-NMR(CDCl3);δ(ppm) 2.08(3H, s), 2.11(3H, s), 2.41(3H, s), 3.85(3H, s), 5.82(1H, s), 6.87(1H, dd, J=2.4, 8.4Hz), 7.06(1H, d, J=2.4Hz), 7.17(2H, d, J=8.4Hz), 7.20-7.24(1H, m), 7.39(2H, d, J=8.4Hz).
MS(ESI); m/z 373(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-メトキシフェニル 4-メチルベンゼンスルホネート93mgより製造例44c)と同様の方法にて表題化合物を30.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.27(3H, s), 2.33(3H, s), 3.80(3H, s), 6.00(1H, s), 6.45(1H, dd, J=2.8, 8.4Hz), 6.61(1H, d, J=2.8Hz), 7.08(1H, d, J=8.4Hz), 9.67(1H, brs).
MS(ESI); m/z 219(M+H)+
a)2-アミノ-3-メチルフェニル 4-メチルベンゼンスルホネート
2-アミノ-3-メチルフェノール 200mgより、製造例43b)と同様の方法にて、表題化合物を374.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.14(3H, s), 2.46(3H, s), 3.79(2H,s), 6.51(1H, t, J=8.0Hz), 6.63(1H, d, J=8.0Hz), 6.91(1H, d, J=8.0Hz), 7.32(2H, d, J=8.4Hz), 7.78(2H, d, J=8.4Hz).
MS(ESI); m/z 278(M+H)+
2-アミノ-3-メチルフェニル 4-メチルベンゼンスルホネート374mgより製造例43c)と同様の方法にて、表題化合物を269.9mg得た。
1H-NMR(CDCl3);δ(ppm) 2.01(3H, s), 2.02(3H, s), 2.16(3H, s), 2.43(3H, s), 5.88(1H, s), 7.19-7.24(3H, m), 7.32(2H, d, J=5.2Hz), 7.51(2H, d, J=8.4Hz).
MS(ESI); m/z 357(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-メチルフェニル 4-メチルベンゼンスルホネート269mgより製造例43d)と同様の方法にて、表題化合物を79.2mg得た。
1H-NMR(CD3OD);δ(ppm) 1.92(3H, s), 2.01(3H, s), 2.24(3H, s), 6.02(1H, s), 6.79(2H, d, J=7.6Hz), 7.18(1H, t, J=7.6Hz).
MS(ESI); m/z 203(M+H)+
4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-ヒドロキシベンゼンカルボン酸メチル 132mgをテトラヒドロフラン 2.6mlに溶解し、リチウムボロハイドライド 58mgを加えて50℃にて3.5時間攪拌した。反応混合物に水20mlを加え、酢酸エチル20mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、分取用薄層シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=1:2)を用いて精製して表題化合物を12.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.28(3H,s), 2.44(3H, s), 4.64(2H, s), 6.02(1H, s), 6.90(1H, d, J=8.0Hz), 7.02(1H, s), 7.17(1H, d, J=8.0Hz).
MS(ESI); m/z 219(M+H)+
a)1-(2-メトキシ-4-ニトロフェニル)-3,5-ジメチル-1H-ピラゾール
2-メトキシ-4-ニトロアニリン700mgより、製造例43c)と同様の方法にて表題化合物を498mg得た。
1H-NMR(CDCl3);δ(ppm) 2.13(3H, s), 2.30(3H, s), 3.93(3H, s), 6.02(1H, s), 7.53(1H, d, J=8.4Hz), 7.89(1H, d, J=2.0Hz), 7.95(1H, dd, J=2.0, 8.4Hz).
MS(ESI); m/z 248(M+H)+
1-(2-メトキシ-4-ニトロフェニル)-3,5-ジメチル-1H-ピラゾール495mgより、製造例31と同様の方法にて表題化合物を417.6mg得た。
1H-NMR(CDCl3);δ(ppm) 2.06(3H, s), 2.28(3H, s), 3.72(3H, s), 3.81(2H, brs), 5.92(1H, s), 6.28-6.31(2H, m), 7.06(1H, d, J=8.4Hz).
MS(ESI); m/z 218(M+H)+
4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-メトキシアニリン200mgをジメチルホルムアミド6mlに溶解し、ヨウ化メチル160μlおよび炭酸カリウム636mgを加えて室温にて2.5時間攪拌した。反応混合物に水50mlを加えて酢酸エチル50mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、分取用薄層シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=1:3)を用いて精製して表題化合物を48.7mg得た。
1H-NMR(CDCl3);δ(ppm) 2.07(3H, s), 2.28(3H, s), 2.86(3H, s), 3.74(3H, s), 3.95(1H, brs), 5.92(1H, s), 6.17(1H, d, J=2.4Hz), 6.21(1H, dd, J=2.4, 8.4Hz), 7.09(1H, d, J=8.4Hz).
MS(ESI); m/z 232(M+H)+
4-(3,5-ジメチル-1H-ピラゾール-1-イル)-3-メトキシ-N-メチルアニリン58.7mgより、製造例33c)と同様の方法にて表題化合物を25mg得た。
1H-NMR(CDCl3);δ(ppm) 2.28(3H, s), 2.32(3H, s), 2.83(3H, d, J=1.2Hz), 5.98(1H, s), 6.13-6.16(1H, m), 6.31-6.32(1H, m), 6.99(1H, dd, J=1.2, 8.4Hz).
MS(ESI); m/z 218(M+H)+
a)2-アミノ-4-メチルフェニル 4-メチルベンゼンスルホネート
2-アミノ-4-メチルフェノール塩酸塩 479mgより、製造例43b)と同様の方法にて、表題化合物を770.1mg得た。
1H-NMR(CDCl3);δ(ppm) 2.21(3H, s), 2.46(3H, s), 3.74(2H, brs), 6.39(1H, d, J=8.0Hz), 6.53(1H, s), 6.62(1H, d, J=8.0Hz), 7.32(2H, d, J=8.0Hz), 7.77(2H, d, J=8.0Hz).
MS(ESI); m/z 278(M+H)+
2-アミノ-4-メチルフェニル 4-メチルベンゼンスルホネート400mgより製造例43c)と同様の方法にて、表題化合物を137.3mg得た。
1H-NMR(CDCl3);δ(ppm) 2.10(3H, s), 2.12(3H, s), 2.35(3H, s), 2.41(3H, s), 5.82(1H, s), 7.13-7.15(3H, m), 7.20-7.22(1H, m), 7.33-7.36(2H, m), 7.42(2H, d, J=8.4Hz).
MS(ESI); m/z 357(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-4-メチルフェニル 4-メチルベンゼンスルホネート167mgより製造例43d)と同様の方法にて、表題化合物を60.4mg得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.31(3H, s), 2.39(3H, s), 6.02(1H, s), 6.97-7.02(3H, m), 9.43(1H, s).
MS(ESI); m/z 203(M+H)+
a)1-(2-メトキシ-4-(トリフルオロメチルフェニル)-3,5-ジメチル-1H-ピラゾール
2-メトキシ-4-トリフルオロメチルアニリン191mgより、製造例43c)と同様の方法にて表題化合物を95.5mg得た。
1H-NMR(CDCl3);δ(ppm) 2.05(3H,s), 2.30(3H, s), 3.86(3H, s), 5.99(1H,s), 7.22(1H,s), 7.31(1H, d, J=8.0Hz), 7.46(1H, d, J=8.0Hz).
MS(ESI); m/z 271(M+H)+
1-(2-メトキシ-4-トリフルオロメチルフェニル)-3,5-ジメチル-1H-ピラゾール95.5mgより製造例33c)と同様の方法にて表題化合物を25.7mg得た。
1H-NMR(CDCl3);δ(ppm) 2.31(3H, s), 2.45(3H, s), 6.08(1H,s), 7.15-7.18(1H, m), 7.30-7.35(2H, m), 10.64(1H,s).
MS(ESI); m/z 257(M+H)+
a)2-アミノ-6-メチルフェニル 4-メチルベンゼンスルホネート
6-アミノ-o-クレゾール塩酸塩 200mgより、製造例43b)と同様の方法にて、表題化合物を159.9mg得た。
1H-NMR(CDCl3);δ(ppm) 2.06(3H, s), 2.48(3H, s), 3.96(2H, s), 6.53-6.55(1H, m), 6.59-6.61(1H, m), 6.93(1H, t, J=7.6Hz), 7.37(2H, d, J=8.0Hz), 7.90(2H, d, J=8.0Hz).
MS(ESI); m/z 278(M+H)+
2-アミノ-6-メチルフェニル 4-メチルベンゼンスルホネート159.5mgより製造例43c)と同様の方法にて、表題化合物を48.8mg得た。
1H-NMR(CDCl3);δ(ppm) 2.09(3H, s), 2.10(3H, s), 2.43(3H, s), 2.49(3H, s), 5.67(1H, s), 7.13-7.32(5H, m), 7.48(2H, d, J=8.0Hz).
MS(ESI); m/z 357(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-6-メチルフェニル 4-メチルベンゼンスルホネート48.5mgより製造例43d)と同様の方法にて、表題化合物を12mg得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.31(3H, s), 2.38(3H, s), 6.03(1H, s), 6.81(1H, t, J=8.0Hz), 7.03-7.09(2H, m), 9.79(1H, s).
MS(ESI); m/z 203(M+H)+
a)4-クロロ-5-エチル-2-ニトロフェニル メタンスルホネート
4-クロロ-5-エチル-2-ニトロフェノール150mgをジクロロメタン1.5mlに溶解し、トリエチルアミン156μlおよびメタンスルホニルクロライド69μlを加えて室温で30分攪拌した。反応混合物に水50mlを加え、酢酸エチル60mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去して、表題化合物を200.7mg得た。
1H-NMR(CDCl3);δ(ppm) 1.28(3H, t, J=7.6Hz), 2.83(2H, q, J=7.6Hz), 3.37(3H, s), 7.42(1H, s), 8.09(1H, s).
4-クロロ-5-エチル-2-ニトロフェニル メタンスルホネート200mgをエタノール4mlに溶解し、10%パラジウム/炭素200mgを加えて、水素雰囲気下、室温にて4時間攪拌した。不溶物を濾過した後、溶媒を減圧下留去して、表題化合物を74.9mg得た。
1H-NMR(CD3OD);δ(ppm) 1.26(3H, t, J=7.6Hz), 2.73(2H,q, J=7.6Hz), 3.47(3H, s), 7.28-7.46(3H, m).
MS(ESI); m/z 216(M+H)+
2-アミノ-5-エチルフェニル メタンスルホネート74.5mgより、製造例43c)と同様の方法にて表題化合物を28.1mg得た。
1H-NMR(CDCl3);δ(ppm) 1.28(3H, t, J=7.6Hz), 2.17(3H, s), 2.26(3H, s), 2.65(3H, s), 2.73(2H, q, J=7.6Hz), 6.00(1H, s), 7.24-7.26(1H, m), 7.33-7.37(2H, m).
MS(ESI); m/z 295(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-エチルフェニル メタンスルホネート28mgをメタノール0.1mlに溶解し、5N-塩酸0.07mlを加えて30分加熱還流した。反応混合物に水15mlを加え、酢酸エチル15mlで抽出した。有機層を無水硫酸マグネシウムで乾燥した後、溶媒を減圧下にて留去し、分取用薄層シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=4:1)を用いて精製して表題化合物を7.4mg得た。
1H-NMR(CDCl3);δ(ppm) 1.24(3H, t, J=7.6Hz), 2.29(3H, s), 2.38(3H, s), 2.63(2H, q, J=7.6Hz), 6.02(1H, s), 6.73-6.75(1H, m), 6.94(1H, s), 7.10(1H, d, J=8.0Hz), 9.67(1H, s).
MS(ESI); m/z 217(M+H)+
a)1-(2-ベンジルオキシフェニル)-4-フルオロ-3,5-ジメチル-1H-ピラゾール
2-ベンジルオキシフェニルヒドラジン塩酸塩500mgと3-フルオロペンタン-2,4-ジオン 251mgをエタノール12mlに加え、1.5時間加熱還流した。反応液をそのまま減圧濃縮し、シリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=6:1~5:1)を用いて精製して表題化合物417mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.07(3H, s), 2.30(3H, s), 5.04(2H, s), 7.03-7.07(2H, m), 7.24-7.37(7H, m).
MS(ESI); m/z 297(M+H)+
1-(2-ベンジルオキシフェニル)-4-フルオロ-3,5-ジメチル-1H-ピラゾール 416mgをメタノール16mlに溶解し、10%パラジウム/炭素42.6mgを加え、水素雰囲気下、室温にて1昼夜攪拌した。不溶物を濾過した後、濾液を減圧下留去して得られた残滓をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=5:1)を用いて精製して表題化合物290mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.28(3H, s), 2.33(3H, s), 6.90(1H, t, J=8.0Hz), 7.06(1H, d, J=8.0Hz), 7.15(1H, d, J=8.0Hz), 7.19(1H, t, J=8.0Hz).
MS(ESI); m/z 207(M+H)+
a)5-ブロモ-2-ヒドラジニルフェノール 4-メチルベンゼンスルホネート
2-アミノ-5-ブロモフェノール1.0gをエタノール7mlに懸濁し、-10℃で濃塩酸1.5mlを滴下した。さらに同温度で亜硝酸tert-ブチル636mgを滴下し、同温度で1時間攪拌し、ジアゾニウム塩溶液を得た。別のフラスコに塩化第一スズ2水和物2.49g、p-トルエンスルホン酸1水和物1.08g、エタノール15mlを加え、-10℃で15分攪拌した。この溶液中に、先に調製したジアゾニウム塩溶液を-10℃で滴下し、同温度で1時間攪拌した。tert-ブチルメチルエーテル30mlを加え、15分攪拌後、得られた沈殿を濾取し、0.9gの表題化合物を得た。
5-ブロモ-2-ヒドラジニルフェノール 4-メチルベンゼンスルホネート0.9g、アセチルアセトン0.8gをエタノール25ml中に加え、1時間加熱還流した。室温まで冷却し、そのまま減圧濃縮して得られた残滓を酢酸エチル50mlに溶解し、飽和重曹水20mlで2回洗浄した。有機層を無水硫酸ナトリウムで脱水後、減圧濃縮して得られた残滓をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=4:1)で精製して表題化合物650mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.40(3H, s), 6.05(1H, s), 7.01-7.08(2H, m), 7.26(1H, s).
MS(ESI);m/z 267(M+H)+
製造例54a)と同様にして調製した5-ブロモ-2-ヒドラジニルフェノール 4-メチルベンゼンスルホネート0.9gと3-クロロペンタン-2,4-ジオン1.07gから製造例54b)と同様の方法にて表題化合物558mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.38(3H, s), 7.02-7.08(2H, m), 7.27(1H, d, J=2.0Hz), 9.62(1H, s).
MS(ESI);m/z 303(M+H)+
製造例54a)と同様にして調製した5-ブロモ-2-ヒドラジニルフェノール 4-メチルベンゼンスルホネート0.9gと3-メチルペンタン-2,4-ジオン0.9gから製造例54b)と同様の方法にて表題化合物320mgを得た。
1H-NMR(CDCl3);δ(ppm) 1.98(3H, s), 2.24(3H, s), 2.31(3H, s), 7.00-7.06(2H, m), 7.25(1H, d, J=1.6 Hz)), 10.34(1H, s).
MS(ESI);m/z 281(M+H)+
a)4-ニトロ-1,3-フェニレン ジアセテート
窒素雰囲気下、4-ニトロベンゼン-1,3-ジオール5.0gを塩化メチレン50mlに溶解し、氷冷下ピリジン5.35g、4-ジメチルアミノピリジン0.39g、無水酢酸8.12gを順次加えた。室温まで昇温し、1時間攪拌した。反応液を水50ml、1N-塩酸100ml、飽和重曹水100ml、飽和食塩水100mlで順次洗い、無水硫酸ナトリウムで脱水後、減圧濃縮し、表題化合物7.4gを得た。
1H-NMR(CDCl3);δ(ppm) 2.33(3H, s), 2.37(3H, s), 7.09(1H, d, J=2.4 Hz), 7.18(1H, dd, J=2.4, 9.2 Hz), 8.16(1H, d, J=9.2 Hz).
窒素雰囲気下、4-ニトロ-1,3-フェニレン ジアセテート1.0gをクロロホルム25mlに溶解し、氷冷下、塩化アルミニウム2.23gを加えた。室温まで昇温し、3時間攪拌した。水100mlを加え、塩化メチレン30mlで3回抽出した。有機層を合わせ、1N-塩酸25ml、飽和食塩水25mlで順次洗い、無水硫酸ナトリウムで脱水後、減圧濃縮した。得られた残滓をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=20:1)で精製して表題化合物610mgを得た。
1H-NMR(CDCl3):δ(ppm) 2.32(3H, s), 6.77(1H, dd, J=2.4 Hz, J=9.2 Hz), 6.95(1H, d, J=2.4 Hz), 8.14(1H, d, J=9.2 Hz), 10.70(1H, s).
MS(ESI);m/z 196(M-H)-
3-ヒドロキシ-4-ニトロフェニル アセテート3.0gを酢酸エチル50mlに溶解し、10%パラジウム/炭素300mgを加え、水素雰囲気下、室温にて10時間攪拌した。不溶物を濾過した後、濾液を減圧下留去して得られた残滓をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=1:1)で精製して表題化合物2.48gを得た。
1H-NMR(DMSO-d6);δ(ppm) 2.17(3H, s), 4.45(2H, brs), 6.28(1H, dd, J=2.4, 8.4Hz), 6.40(1H, d, J=2.4Hz), 6.53(1H, d, J=8.4Hz), 9.26(1H, brs).
MS(ESI);m/z 167(M+)
4-アミノ-3-ヒドロキシフェニル アセテート1.0gを用い、製造例54a)と同様にして調製した4-ヒドラジニル-3-ヒドロキシフェニル アセテート 4-メチルベンゼンスルホネート0.9gとアセチルアセトン0.9gから製造例54b)と同様の方法にて表題化合物330mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.30(3H, s), 2.41(3H, s), 6.04(1H, s), 6.68(1H, dd, J=2.4, 8.8Hz), 6.85(1H, d, J=2.4Hz), 7.19(1H, d, J=8.8 Hz), 10.14(1H, s).
MS(ESI);m/z 247(M+H)+
4-アミノ-3-ヒドロキシフェニル アセテート1.0gを用い、製造例54a)と同様にして調製した4-ヒドラジニル-3-ヒドロキシフェニル アセテート 4-メチルベンゼンスルホネート0.9gと3-クロロペンタン-2,4-ジオン1.2gから製造例54b)と同様の方法にて表題化合物30mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.30(3H, s), 2.31(3H, s), 2.39(3H, s), 6.70(1H, dd, J=2.8, 8.8Hz), 6.86(1H, d, J=2.8Hz), 7.17(1H, d, J=8.8 Hz), 9.50(1H, s).
MS(ESI);m/z 281(M+H)+
4-アミノ-3-ヒドロキシフェニル アセテート1.0gを用い、製造例54a)と同様にして調製した4-ヒドラジニル-3-ヒドロキシフェニル アセテート 4-メチルベンゼンスルホネート0.6gと3-メチルペンタン-2,4-ジオン1.0gから製造例54b)と同様の方法にて表題化合物230mgを得た。
1H-NMR(CDCl3);δ(ppm) 1.99(3H, s), 2.24(3H, s), 2.30(3H, s), 2.32(3H, s), 6.66(1H, dd, J=2.8, 8.4Hz), 6.83(1H, d, J=2.8Hz), 7.17(1H, d, J=8.4 Hz), 10.22(1H, s).
MS(ESI);m/z 261(M+H)+
a)1,4-ジメトキシ-2-メチル-5-ニトロベンゼン
2,5-ジメトキシトルエン6.0gを酢酸20mlに溶解し、40℃で発煙硝酸(d=1.50)4.32gの酢酸10ml溶液を5分間かけて滴下した。同温度で30分攪拌し、室温まで冷却後、さらに30分攪拌した。反応液を冷水300mlで希釈し、生じた沈殿を濾取し、冷水100mlで洗浄した。減圧下乾燥し、表題化合物7.5gを得た。
1H-NMR(CDCl3);δ(ppm) 2.28(3H, s), 3.84(3H, s), 3.92(3H, s), 6.90(1H, s), 7.40(1H, s).
MS(ESI);m/z 198(M+H)+
1,4-ジメトキシ-2-メチル-5-ニトロベンゼン6.0gの塩化メチレン30ml溶液を-20℃に冷却し、三塩化ホウ素1M塩化メチレン溶液30mlを同温度で滴下した。室温まで昇温後、16時間攪拌し、飽和重曹水50mlに反応液を加え、酢酸エチル100mlで3回抽出した。有機層を合わせ、水100ml、飽和食塩水50mlで洗浄後、無水硫酸ナトリウムで脱水し、減圧濃縮した。得られた残滓をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=9:1)で精製して表題化合物4.25gを得た。
1H-NMR(CDCl3);δ(ppm) 2.26(3H, s), 3.84(3H, s), 6.94(1H, s), 7.39(1H, s), 10.46(1H, s).
MS(ESI);m/z 182(M-H)-
窒素雰囲気下、4-メトキシ-5-メチル-2-ニトロフェノール8.0gの塩化メチレン80ml溶液にp-トルエンスルホニルクロライド9.15gを室温で加え、0℃まで冷却した。ここにトリエチルアミン4.86gを加え、2時間攪拌した。反応液を水150mlにあけ、塩化メチレン150mlで抽出した。有機層を水100ml、飽和食塩水50mlで洗浄し、無水硫酸ナトリウムで脱水後、減圧濃縮した。得られた残滓をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=9:1)で精製して表題化合物12.0gを得た。
1H-NMR(CDCl3);δ(ppm) 2.26(3H, s), 2.46(3H, s), 3.87(3H, s), 7.19(1H, s), 7.33(1H, s), 7.34(2H, d, J=8.4Hz), 7.77(2H, d, J=8.4Hz).
MS(ESI);m/z 336(M-H)-
4-メトキシ-5-メチル-2-ニトロフェニル 4-メチルベンゼンスルホネート4.0gを用い、製造例57c)と同様の方法にて表題化合物3.2gを得た。
1H-NMR(DMSO-d6);δ(ppm) 1.91(3H, s), 2.41(3H, s), 3.64(3H, s), 4.73(2H, brs), 6.25(1H, s), 6.64(1H, s), 7.43(2H, d, J=8.4Hz), 7.79(2H, d, J=8.4Hz).
MS(ESI);m/z 308(M+H)+
2-アミノ-4-メトキシ-5-メチルフェニル 4-メチルベンゼンスルホネート1.0gを用い、製造例54a)、54b)と同様の方法にて表題化合物400mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.10(6H, s), 2.25(3H, s), 2.41(3H, s), 3.79(3H, s), 5.80(1H, s), 6.71(1H, s), 7.15(2H, d, J=8.4Hz), 7.27(1H, s), 7.35(2H, d, J=8.4Hz).
MS(ESI);m/z 387(M+H)+
2-(3,5-ジメチル-1H-ピラゾール-1-イル)-4-メトキシ-5-メチルフェニル 4-メチルベンゼンスルホネート200mgを用い、製造例29と同様の方法にて表題化合物70mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.21(3H, s), 2.29(3H, s), 2.39(3H, s), 3.78(3H, s), 6.02(1H, s), 6.67(1H, s), 6.89(1H, s), 8.87(1H, s).
MS(ESI);m/z 233(M+H)+
a)2-アミノ-4-クロロ-5-メチルフェノール
4-クロロ-5-メチル-2-ニトロフェノール5.0gのメタノール100ml溶液中に塩酸で活性化した亜鉛末8.71gおよび塩化アンモニウム7.1gの水20ml溶液を0℃で添加した。この懸濁液を室温で4時間攪拌後、不溶物をセライトを用いて濾過し、酢酸エチル100mlで洗浄した。濾液と洗液を合わせて減圧濃縮した。得られた残滓をシリカゲルカラムクロマトグラフィー(ヘキサン:酢酸エチル=3:2)で精製して表題化合物1.4gを得た。
1H-NMR(DMSO-d6);δ(ppm) 2.10(3H, s), 4.57(2H, brs), 6.54(1H, s), 6.58(1H, s), 9.11(1H, s).
MS(ESI);m/z 157(M)+
2-アミノ-4-クロロ-5-メチルフェノール1.0gを用い、製造例54a)、54b)と同様の方法にて表題化合物120mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.29(3H, s), 2.34(3H, s), 2.41(3H, s), 6.03(1H, s), 6.96(1H, s), 7.19(1H, s), 9.88(1H, s).
MS(ESI);m/z 237(M+H)+
2-アミノ-4,5-ジメチルフェノール1.0gを用い、製造例54a)、54b)と同様の方法にて表題化合物630mgを得た。
1H-NMR(CDCl3);δ(ppm) 2.21(3H, s), 2.24(3H, s), 2.29(3H, s), 2.38(3H, s), 6.00(1H, s), 6.88(1H, s), 6.94(1H, s), 9.28(1H, s).
MS(ESI);m/z 217(M+H)+
製造例58においてシリカゲルカラムクロマトグラフィー精製中に副産物として表題化合物200mgを得た。
1H-NMR(CD3OD);δ(ppm) 2.07(3H, s), 2.20(3H, s), 6.36(1H, dd, J=2.4, 8.4Hz), 6.42(1H, d, J=2.4Hz), 6.98(1H, d, J=8.4Hz).
MS(ESI);m/z 239(M+H)+
実施例1
以下で、本発明を実施例によって説明するが、本発明はこれらの実施例に限定されるものではない。なお、以下において「製造例44」とは、「製造例44の化合物」を意味する。
製造例44 10 g
コポリビドン 10 g
エタノール 100mL
エタノールの一部に製造例44及びコポリビドンを順に溶解させ、エタノールで合計100mLにして製剤を得た。
また上記方法を参照に、コポリビドンの代わりに下記ポリマーを含有する外用液剤を製造した。
製造例44(1w/v%)と各種添加剤(5w/v%)をエタノール(またはエタノール/酢酸エチル混液(7:3))に溶解させ、ガラスバイアルに入れ、施栓したものの保存試験を行った。40℃/75%RHで3箇月間保存した後、表1の通り、製造例44の残存率はいずれの場合も90%以上であり、製造例44残存率を低下させる添加剤は認められなかった。
本発明の外用液剤を爪に塗布した際に、爪上で薬物が析出することを防止するため、薬物と高分子の相溶性を示差走査熱量測定(DSC)により評価した。高分子と薬物を各比率でエタノールに溶解し、DSC用アルミパン中に溶液50 μLを滴下し、60℃で1時間乾燥した。乾燥物(皮膜)を示差走査熱量計により測定した(DSC測定条件:クリンプパン、測定範囲40℃~120℃、昇温速度は製造例44粉末の場合は3℃/min、その他の試料は5℃/min、n=1)。各試料において、105℃付近の製造例44融解ピークにつきエンタルピーを算出し、製造例44粉末単独で測定した際の融解エンタルピーと比較した。
表2に示した通り、いずれの高分子についても、薬物量に対する高分子添加量の増加に伴い、皮膜中において結晶状態で存在する薬物量が減少する傾向が認められた。
ウシ爪を用いて、本発明の外用液剤の爪透過性について調べた。
厚み0.1mmのウシ爪を適当な大きさに切り、消毒用エタノールに浸す。乾燥させた後、ウシ蹄上に筒状のプラスチック(高さ約2.5mm、幅約5mm、厚み約1mm)を載せ、接着剤で固定し一晩放置して、完全に接着させる。
1%低融点アガロースをオートクレーブした後、シャーレに80mLずつ分注して固める。そこから金属製のコルクホールボーラーでアガロース片を円柱形にくりだし(高さ約1.5cm、直径約2cm、容量約5mL)、その上にプラスチックを固定したウシ蹄をのせる。調製した試験製剤1μLを筒状プラスチック内のウシ爪に塗布し、パラフィルムで上部をカバーしたのち、28℃7日間放置し、ウシ蹄を取り除く。低融点アガロースに蒸留水35mLを添加し、70℃にてゲルを溶解させ、溶液をLC-MSにて製造例44の爪透過量を定量する。
表4に示したように、いずれの試験製剤においても、製造例44の高い爪透過性が認められた。
本発明の外用液剤に含有される一般式(I)の化合物の抗白癬菌活性測定は以下の方法で実施した。評価化合物はジメチルスルホキシド(DMSO)に溶解して使用した。試験用培地としては、0.165Mの3-モルホリノプロパンスルホン酸(MOPS)を含有するアールピーエムアイ1640(RPMI1640)培地を用いた。試験菌としてトリコフィトン・メンタグロファイテス(T. mentagrophytes)ATCC18748もしくはトリコフィトン・ルブラム(T. rubrum)ATCC10218を用いた。試験菌種を1×104 conidia/mlの濃度で100μl分注し、DMSO濃度が1%となるように96ウェルハーフエリアプレート上で評価化合物と混合して、培養温度28℃にて3日間(トリコフィトン・メンタグロファイテス)もしくは4日間(トリコフィトン・ルブラム)培養した。その後、Cell Counting Kit8(WST8)を5μl添加して、450nmと595nmでの吸光度を測定し、バックグラウンド値とした。その後、28℃にて5時間(トリコフィトン・メンタグロファイテス)もしくは一晩(トリコフィトン・ルブラム)培養して発色させ、再度450nmと595nmでの吸光度を測定し、バックグラウンド値との差で生育阻害率を計算、80%生育阻害濃度をMIC値(μg/ml)とした。
本発明外用液剤のヒト爪に対する透過性抗白癬菌活性はメドファーマ社のターチューブモデルを用いて以下のように測定した。
まず始めに被験製剤を調製した。被験製剤は製造例44の外用液剤であり、製造例44の濃度を0%(プラセボ)、2%、5%、10%および15%とし、コポリビドン(10%)およびエタノールを添加して調製した。
被験製剤をターチューブセル上部のヒト爪上に2μl適用したのち、ターチューブセルを20℃から25℃で7日間培養した。その後、ターチューブセル下部レシーバー内の寒天上に接種した白癬菌(トリコフィトン ルブラム)の発育に対する阻止円長を測定し、ヒト爪を透過した被験製剤の抗白癬菌活性を確認した。
その結果、製造例44の外用液剤は活性物質の濃度(2%、5%、10%および15%)に依存した大きさの阻止円を形成した。一方、シクロピロックスの臨床製剤、アモロルフィンの臨床製剤を適用した際にはまったく阻止円を形成しなかった。
以上により、本願外用液剤は、ヒト爪を透過し、抗白癬菌活性を濃度依存的に示すことがわかった。
本発明外用液剤のウシ爪に対する透過性抗白癬菌活性は以下のように測定した。5(2)で調製した厚み0.1mmのウシ爪に調製した試験製剤2μLを筒状プラスチック内のウシ爪に塗布し、パラフィルムでカバーした後、試験菌としてトリコフィトン・メンタグロファイテス(T. mentagrophytes)ATCC18748を接種した寒天培地上に静置して、培養温度28℃にて4日間トリコフィトン・メンタグロファイテスを培養した。その後、その発育に対する阻止円長を測定し、ウシ爪を透過した被験製剤の抗白癬菌活性を確認した。
上記2から10の評価結果より、有効成分である一般式(I)およびポリマーを含有する外用液剤は、皮膚等に塗布することによって容易に皮膜を形成し、強い抗白癬菌活性を有し、かつ、高い安定性、爪透過性、有効成分の析出抑制効果を有することが確認された。また速乾性も有しており、添加剤であるポリマーとしては、水溶性ポリマーおよび、水不溶性ポリマーともに好ましいことが示された。とりわけ、コポリビドン、ポビドン、アクリル樹脂アルカノールアミン、および、アミノアルキルメタクリレートコポリマーが好ましい。また、さらに粘稠剤を添加することも好ましく、とりわけ、シリコーン誘導体が好ましい。
Claims (14)
- 皮膜形成剤と、一般式
R1は、水素原子、C1-6アルキル、またはトリフルオロメチルを表し、
R2は、水素原子、C1-6アルキル、ハロゲン、-COO(C1-6アルキル)、または(CH2)1-3COOR(Rは、水素原子またはC1-6アルキルを表す)を表し、
R3は、水素原子、C1-6アルキル、アミノ、トリフルオロメチル、またはOR(Rは、水素原子またはC1-6アルキルを表す)を表し、
R4は、水酸基を表し、
R5は、水素原子、C1-6アルキル、水酸基、またはハロゲンを表し、
R6は、水素原子、C1-6アルキル、トリフルオロメチル、ハロゲン、アミノ、-NRaRb、ニトロ、ヒドロキシC1-6アルキル、-CONRaRb、-COO(C1-6アルキル)、-COOH、-(CH2)1-3COOR、またはORa(Rは、水素原子またはC1-6アルキルを表し、RaおよびRbは、同一でも異なっていてもよく、水素原子、C1-6アルキル、またはC1-6アシルを表す)を表し、
R7は、水素原子、C1-6アルキル、-OR(Rは、水素原子またはC1-6アルキルを表す)、またはハロゲンを表し、
R8は、水素原子、C1-6アルキル、水酸基、アミノ、またはニトロを表す。)
で示される化合物またはその塩と、を含有する、白癬菌症の予防または治療用の爪および/または皮膚用外用液剤。 - R1が、水素原子またはC1-6アルキルであり、
R2が、水素原子、C1-6アルキル、ハロゲン、または-(CH2)1-3COOR(Rは、水素原子またはC1-6アルキルを表す)であり、
R3が、水素原子、またはC1-6アルキルであり、
R4が、水酸基であり、
R5が、水素原子であり、
R6が、水素原子、C1-6アルキル、トリフルオロメチル、ハロゲン、アミノ、-NRaRb、ニトロ、ヒドロキシC1-6アルキル、-CONRaRb、-COOH、またはORa(RaおよびRbは同一でも異なっていてもよく、水素原子、C1-6アルキル、またはC1-6アシルを表す)を表し、
R7が、水素原子であり、
R8が、水素原子、C1-6アルキル、アミノ、またはニトロである、
一般式(I)で示される化合物またはその塩を含有する、請求項1記載の爪および/または皮膚用外用液剤。 - R1が、C1-6アルキルであり、
R2が、水素原子、C1-6アルキル、またはハロゲンであり、
R3が、C1-6アルキルであり、
R4が、水酸基であり、
R5が、水素原子であり、
R6が、水素原子、C1-6アルキル、ハロゲン、またはORa(Raは、水素原子、C1-6アルキル、またはC1-6アシルを表す)を表し、
R7が、水素原子であり、
R8が、水素原子である、
一般式(I)で示される化合物またはその塩を含有する、請求項1記載の爪および/または皮膚用外用液剤。 - R1が、C1-6アルキルであり、R2が、水素原子であり、R3が、C1-6アルキルであり、R4が、水酸基であり、R5が、水素原子であり、R6が、C1-6アルキルであり、R7が、水素原子であり、R8が、水素原子である一般式(I)で示される化合物またはその塩を含有する、請求項1記載の爪および/または皮膚用外用液剤。
- 2-(3,5-ジメチル-1H-ピラゾール-1-イル)-5-メチルフェノールを含有する、請求項1記載の爪および/または皮膚用外用液剤。
- 皮膜形成剤がポリマーを含んでなる、請求項1~5のいずれか1項記載の爪および/または皮膚用外用液剤。
- ポリマーが、コポリビドン、ポビドン、アクリル樹脂アルカノールアミン、または、アミノアルキルメタクリレートコポリマーである、請求項6記載の爪および/または皮膚用外用液剤。
- 低級アルコールを含んでなる、請求項1~7のいずれか1項記載の爪および/または皮膚用外用液剤。
- 粘稠剤を含んでなる、請求項1~8のいずれか1項記載の爪および/または皮膚用外用液剤。
- 粘稠剤がシリコーン誘導体、疎水化ヒドロキシプロピルメチルセルロース、またはポロクサマーである請求項9記載の爪および/または皮膚用外用液剤。
- 一般式(I)で示される化合物またはその塩を、液剤量全体に対して1~30質量%の割合で含有する、請求項1~10のいずれか1項記載の爪および/または皮膚用外用液剤。
- 一般式(I)で示される化合物またはその塩を、液剤量全体に対して1~20質量%の割合で含有する、請求項1~10のいずれか1項記載の爪および/または皮膚用外用液剤。
- 一般式(I)で示される化合物またはその塩を、液剤量全体に対して5~15質量%の割合で含有する、請求項1~10のいずれか1項記載の爪および/または皮膚用外用液剤。
- 爪白癬の予防または治療用である、請求項1~13のいずれか1項記載の爪および/または皮膚用外用液剤。
Priority Applications (14)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2879846A CA2879846A1 (en) | 2012-07-30 | 2013-07-30 | Topical liquid agent for the treatment of dermatophytosis |
| KR20157004795A KR20150038314A (ko) | 2012-07-30 | 2013-07-30 | 항백선균증 외용액제 |
| AU2013297602A AU2013297602A1 (en) | 2012-07-30 | 2013-07-30 | Antitrichophytosis solution for external use |
| EP13825029.5A EP2889031A4 (en) | 2012-07-30 | 2013-07-30 | ANTI-TRICHOPHYTIC SOLUTION FOR EXTERNAL USE |
| RU2015106906A RU2015106906A (ru) | 2012-07-30 | 2013-07-30 | Местнодействующий жидкий агент для лечения дерматофитоза |
| JP2014528154A JP6132842B2 (ja) | 2012-07-30 | 2013-07-30 | 抗白癬菌症外用液剤 |
| SG11201500637QA SG11201500637QA (en) | 2012-07-30 | 2013-07-30 | Antitrichophytosis solution for external use |
| MX2015001416A MX2015001416A (es) | 2012-07-30 | 2013-07-30 | Agente liquido topico para el tratamiento de la dermatofitosis. |
| CN201380040338.3A CN104661661A (zh) | 2012-07-30 | 2013-07-30 | 抗白癣菌症外用液剂 |
| BR112015001235A BR112015001235A2 (pt) | 2012-07-30 | 2013-07-30 | agente líquido tópico para o tratamento de dermatofitose |
| IL236993A IL236993A0 (en) | 2012-07-30 | 2015-01-29 | A liquid substance to administer for the treatment of dermatophytosis |
| PH12015500224A PH12015500224A1 (en) | 2012-07-30 | 2015-01-30 | Topical liquid agent for the treatment of dermatophytosis |
| ZA2015/00809A ZA201500809B (en) | 2012-07-30 | 2015-02-04 | Antitrichophytosis solution for external use |
| HK15111656.5A HK1210718A1 (en) | 2012-07-30 | 2015-11-26 | Antitrichophytosis solution for external use |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012069306 | 2012-07-30 | ||
| JPPCT/JP2012/069306 | 2012-07-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014021282A1 true WO2014021282A1 (ja) | 2014-02-06 |
Family
ID=49995097
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/070525 WO2014021282A1 (ja) | 2012-07-30 | 2013-07-30 | 抗白癬菌症外用液剤 |
Country Status (20)
| Country | Link |
|---|---|
| US (1) | US20140030209A1 (ja) |
| EP (1) | EP2889031A4 (ja) |
| JP (1) | JP6132842B2 (ja) |
| KR (1) | KR20150038314A (ja) |
| CN (1) | CN104661661A (ja) |
| AR (1) | AR092358A1 (ja) |
| AU (1) | AU2013297602A1 (ja) |
| BR (1) | BR112015001235A2 (ja) |
| CA (1) | CA2879846A1 (ja) |
| CL (1) | CL2015000218A1 (ja) |
| CO (1) | CO7170168A2 (ja) |
| HK (1) | HK1210718A1 (ja) |
| IL (1) | IL236993A0 (ja) |
| MX (1) | MX2015001416A (ja) |
| PH (1) | PH12015500224A1 (ja) |
| RU (1) | RU2015106906A (ja) |
| SG (1) | SG11201500637QA (ja) |
| TW (1) | TW201410239A (ja) |
| WO (1) | WO2014021282A1 (ja) |
| ZA (1) | ZA201500809B (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019151282A1 (ja) * | 2018-01-31 | 2019-08-08 | Meiji Seikaファルマ株式会社 | 爪白癬の予防又は治療用外用液剤 |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW201410273A (zh) | 2012-07-30 | 2014-03-16 | Meiji Seika Pharma Co Ltd | 抗白癬菌症貼附劑 |
| CA2957880A1 (en) | 2014-08-13 | 2016-02-18 | Meiji Seika Pharma Co., Ltd. | Crystalline anti-trichophyton agents and preparation process thereof |
| ES2766424T3 (es) * | 2015-03-19 | 2020-06-12 | Daiichi Sankyo Co Ltd | Preparación sólida que contienen agente oxidante |
| ES2821733T3 (es) | 2015-03-19 | 2021-04-27 | Daiichi Sankyo Co Ltd | Preparación sólida que contiene colorante |
| CN107311838A (zh) * | 2017-07-13 | 2017-11-03 | 柳州丰康泰科技有限公司 | 一种新的合成环丙基溴的方法 |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6368569A (ja) * | 1986-08-26 | 1988-03-28 | バイエル・アクチエンゲゼルシヤフト | 置換1‐アリール‐3‐tert.‐ブチル‐ピラゾール |
| ES2015648A6 (es) | 1989-05-05 | 1990-09-01 | Consejo Superior Investigacion | Procedeimiento de sintesis de mono y bis (3-r3-4-r4-5-r5-pirazol-1-il)1,4-dihidroxibencenos. |
| JPH0853401A (ja) | 1994-04-28 | 1996-02-27 | Hoechst Ag | 芳香族ジアゾニウム塩およびそれらの放射線感応性混合物における使用 |
| JPH08188545A (ja) | 1995-01-05 | 1996-07-23 | Kanto Denka Kogyo Co Ltd | フルオロフェノール類の製造方法 |
| JPH1160528A (ja) | 1997-08-12 | 1999-03-02 | Ube Ind Ltd | 4−クロロ−3−トリフルオロメチルフェノールの製法 |
| WO2003005999A2 (en) | 2001-07-11 | 2003-01-23 | Boehringer Ingelheim Pharmaceuticals, Inc. | Methods of treating cytokine mediated diseases |
| WO2004033432A1 (ja) * | 2002-10-09 | 2004-04-22 | Ssp Co., Ltd. | 抗真菌活性を有する新規ピラゾール化合物 |
| US6852890B1 (en) | 2000-03-03 | 2005-02-08 | Sumitomo Chemical Company, Limited | Process for the preparation of phenylhydrazines |
| US20070105866A1 (en) | 2005-11-04 | 2007-05-10 | Amira Pharmaceuticals, Inc. | 5-lipoxygenase-activating protein (flap) inhibitors |
| WO2007083320A2 (en) | 2006-01-19 | 2007-07-26 | Matrix Laboratories Ltd | Conversion of aromatic diazonium salt to aryl hydrazine |
| JP4284169B2 (ja) | 2002-12-28 | 2009-06-24 | 三星モバイルディスプレイ株式會社 | 赤色発光化合物およびそれを採用した有機電界発光素子 |
| WO2012102404A1 (ja) * | 2011-01-30 | 2012-08-02 | Meiji Seikaファルマ株式会社 | 外用抗真菌剤 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5391367A (en) * | 1993-07-28 | 1995-02-21 | Pfizer Inc. | Antifungal nail solution |
| EP1144383A1 (en) * | 1999-01-14 | 2001-10-17 | Merck Frosst Canada & Co. | Synthesis of 4-[(5-substituted or unsubstituted phenyl)-3-substituted-1h-pyrazol-1-yl]benzenesulfonamides |
| KR101093990B1 (ko) * | 2003-03-21 | 2011-12-16 | 넥스메드 홀딩스 인코포레이티드 | 항진균성 네일 코트 및 사용 방법 |
| WO2008024978A2 (en) * | 2006-08-24 | 2008-02-28 | Serenex, Inc. | Tetrahydroindolone and tetrahydroindazolone derivatives |
| CN101011560A (zh) * | 2007-02-13 | 2007-08-08 | 贵州神奇集团控股有限公司 | 治疗脚手癣、灰指甲的中西复方制剂及其制备方法和质控方法 |
-
2013
- 2013-07-30 US US13/954,804 patent/US20140030209A1/en not_active Abandoned
- 2013-07-30 BR BR112015001235A patent/BR112015001235A2/pt not_active IP Right Cessation
- 2013-07-30 WO PCT/JP2013/070525 patent/WO2014021282A1/ja active Application Filing
- 2013-07-30 AU AU2013297602A patent/AU2013297602A1/en not_active Abandoned
- 2013-07-30 CA CA2879846A patent/CA2879846A1/en not_active Abandoned
- 2013-07-30 JP JP2014528154A patent/JP6132842B2/ja not_active Expired - Fee Related
- 2013-07-30 KR KR20157004795A patent/KR20150038314A/ko not_active Application Discontinuation
- 2013-07-30 AR ARP130102697A patent/AR092358A1/es unknown
- 2013-07-30 TW TW102127227A patent/TW201410239A/zh unknown
- 2013-07-30 MX MX2015001416A patent/MX2015001416A/es unknown
- 2013-07-30 RU RU2015106906A patent/RU2015106906A/ru not_active Application Discontinuation
- 2013-07-30 EP EP13825029.5A patent/EP2889031A4/en not_active Withdrawn
- 2013-07-30 SG SG11201500637QA patent/SG11201500637QA/en unknown
- 2013-07-30 CN CN201380040338.3A patent/CN104661661A/zh active Pending
-
2014
- 2014-12-23 CO CO14281705A patent/CO7170168A2/es unknown
-
2015
- 2015-01-28 CL CL2015000218A patent/CL2015000218A1/es unknown
- 2015-01-29 IL IL236993A patent/IL236993A0/en unknown
- 2015-01-30 PH PH12015500224A patent/PH12015500224A1/en unknown
- 2015-02-04 ZA ZA2015/00809A patent/ZA201500809B/en unknown
- 2015-11-26 HK HK15111656.5A patent/HK1210718A1/xx unknown
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6368569A (ja) * | 1986-08-26 | 1988-03-28 | バイエル・アクチエンゲゼルシヤフト | 置換1‐アリール‐3‐tert.‐ブチル‐ピラゾール |
| ES2015648A6 (es) | 1989-05-05 | 1990-09-01 | Consejo Superior Investigacion | Procedeimiento de sintesis de mono y bis (3-r3-4-r4-5-r5-pirazol-1-il)1,4-dihidroxibencenos. |
| JPH0853401A (ja) | 1994-04-28 | 1996-02-27 | Hoechst Ag | 芳香族ジアゾニウム塩およびそれらの放射線感応性混合物における使用 |
| JPH08188545A (ja) | 1995-01-05 | 1996-07-23 | Kanto Denka Kogyo Co Ltd | フルオロフェノール類の製造方法 |
| JPH1160528A (ja) | 1997-08-12 | 1999-03-02 | Ube Ind Ltd | 4−クロロ−3−トリフルオロメチルフェノールの製法 |
| US6852890B1 (en) | 2000-03-03 | 2005-02-08 | Sumitomo Chemical Company, Limited | Process for the preparation of phenylhydrazines |
| WO2003005999A2 (en) | 2001-07-11 | 2003-01-23 | Boehringer Ingelheim Pharmaceuticals, Inc. | Methods of treating cytokine mediated diseases |
| WO2004033432A1 (ja) * | 2002-10-09 | 2004-04-22 | Ssp Co., Ltd. | 抗真菌活性を有する新規ピラゾール化合物 |
| JP4284169B2 (ja) | 2002-12-28 | 2009-06-24 | 三星モバイルディスプレイ株式會社 | 赤色発光化合物およびそれを採用した有機電界発光素子 |
| US20070105866A1 (en) | 2005-11-04 | 2007-05-10 | Amira Pharmaceuticals, Inc. | 5-lipoxygenase-activating protein (flap) inhibitors |
| WO2007083320A2 (en) | 2006-01-19 | 2007-07-26 | Matrix Laboratories Ltd | Conversion of aromatic diazonium salt to aryl hydrazine |
| WO2012102404A1 (ja) * | 2011-01-30 | 2012-08-02 | Meiji Seikaファルマ株式会社 | 外用抗真菌剤 |
Non-Patent Citations (18)
| Title |
|---|
| "Experimental Chemistry, 4th ed.", pages: 159 - 266 |
| "Pathogenic fungi and mycosis, Revised 2nd Edition", NANZANDO CO., LTD., pages: 184 - 187 |
| "pathogenic fungi and mycosis, Revised 2nd Edition", NANZANDO CO., LTD., pages: 42 - 45 |
| ANANDARAJAGOPAL, K. ET AL.: "Antiepileptic and antimicrobial activities of novel 1- (unsubstituted/substituted)-3,5-dimethyl-lH- pyrazole derivatives", INTERNATIONAL JOURNAL OF CHEMTECH RESEARCH, vol. 2, no. 1, 2010, pages 45 - 49 * |
| ANNUAL REPORT OF TAKEDA RESEARCH CENTER, vol. 22, 1963, pages 27 |
| BIOORG. MED. CHEM. LETT., vol. 18, 2008, pages 4438 - 4441 |
| BR. J. OF DERMATOL., vol. 139, no. 4, 1998, pages 665 |
| GREEN: "Protective Groups in Organic Synthesis, 5th ed.", 1999, JOHN WIELEY & SONS |
| J. ORG. CHEM., vol. 21, 1956, pages 394 - 399 |
| J. ORG. CHEM., vol. 76, 2011, pages 654 - 660 |
| J.AM.CHEM.SOC., vol. 92, 1970, pages 853 - 859 |
| J.ORG.CHEM., vol. 21, 1956, pages 394 - 399 |
| J.ORG.CHEM., vol. 70, 2005, pages 5164 - 5173 |
| J.ORG.CHEM., vol. 72, 2007, pages 6190 - 6199 |
| ORGANIC LETTERS, vol. 3, 2001, pages 3803 - 3805 |
| ORGANIC SYNTHESIS COLLECTIVE, vol. 1, pages 442 - 445 |
| S. M. BERGE ET AL.: "describe pharmaceutically acceptable salts", JOURNAL OF PHARMACEUTICAL SCIENCES, 1977, pages 66 |
| See also references of EP2889031A4 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019151282A1 (ja) * | 2018-01-31 | 2019-08-08 | Meiji Seikaファルマ株式会社 | 爪白癬の予防又は治療用外用液剤 |
| WO2019150485A1 (ja) * | 2018-01-31 | 2019-08-08 | Meiji Seikaファルマ株式会社 | 爪白癬の予防又は治療用外用液剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201410239A (zh) | 2014-03-16 |
| KR20150038314A (ko) | 2015-04-08 |
| BR112015001235A2 (pt) | 2017-07-04 |
| CL2015000218A1 (es) | 2015-05-22 |
| JPWO2014021282A1 (ja) | 2016-07-21 |
| AU2013297602A1 (en) | 2015-02-19 |
| RU2015106906A (ru) | 2016-09-20 |
| HK1210718A1 (en) | 2016-05-06 |
| EP2889031A1 (en) | 2015-07-01 |
| SG11201500637QA (en) | 2015-03-30 |
| MX2015001416A (es) | 2015-05-08 |
| ZA201500809B (en) | 2016-04-28 |
| CO7170168A2 (es) | 2015-01-28 |
| US20140030209A1 (en) | 2014-01-30 |
| JP6132842B2 (ja) | 2017-05-24 |
| AR092358A1 (es) | 2015-04-15 |
| EP2889031A4 (en) | 2016-01-06 |
| CA2879846A1 (en) | 2014-02-06 |
| PH12015500224A1 (en) | 2015-04-06 |
| CN104661661A (zh) | 2015-05-27 |
| IL236993A0 (en) | 2015-03-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6132842B2 (ja) | 抗白癬菌症外用液剤 | |
| JP5362128B2 (ja) | 外用抗真菌剤 | |
| Giornal et al. | Synthesis of diversely fluorinated pyrazoles as novel active agrochemical ingredients | |
| US10123978B2 (en) | Patch for treating dermatophytosis | |
| CA2568993A1 (fr) | Sel de sodium monohydrate du s-tenatoprazole et application en tant qu'inhibiteur de la pompe a protons | |
| AU2015210334A1 (en) | Topical antifungal agent | |
| Verma et al. | Synthesis and characterisation of 3-acetylindole derivatives and evaluation of their anti-inflammatory and anti-microbial Activity | |
| CN112079744A (zh) | 芳香酰腙衍生物及其作为na抑制剂的应用 | |
| CA2872832A1 (fr) | Composition pharmaceutique d'un inhibiteur du virus du papillome | |
| NZ613856B2 (en) | Topical antifungal agent | |
| WO2018190324A1 (ja) | ピリドン化合物を有効成分として含有する抗真菌剤およびその使用方法 | |
| Jadhav et al. | Synthesis and Biological Evaluation of New Pyridine Derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13825029 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2014528154 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14281705 Country of ref document: CO |
|
| ENP | Entry into the national phase |
Ref document number: 2879846 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IDP00201500415 Country of ref document: ID |
|
| WWE | Wipo information: entry into national phase |
Ref document number: P115/2015 Country of ref document: AE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015000218 Country of ref document: CL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 236993 Country of ref document: IL Ref document number: MX/A/2015/001416 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12015500224 Country of ref document: PH |
|
| ENP | Entry into the national phase |
Ref document number: 2013297602 Country of ref document: AU Date of ref document: 20130730 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20157004795 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2013825029 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: A201501771 Country of ref document: UA Ref document number: 2013825029 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2015106906 Country of ref document: RU Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112015001235 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112015001235 Country of ref document: BR Kind code of ref document: A2 Effective date: 20150119 |