WO2013183363A1 - 炭化水素資源回収ダウンホールツール用部材 - Google Patents
炭化水素資源回収ダウンホールツール用部材 Download PDFInfo
- Publication number
- WO2013183363A1 WO2013183363A1 PCT/JP2013/061075 JP2013061075W WO2013183363A1 WO 2013183363 A1 WO2013183363 A1 WO 2013183363A1 JP 2013061075 W JP2013061075 W JP 2013061075W WO 2013183363 A1 WO2013183363 A1 WO 2013183363A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- thickness
- decomposition
- downhole tool
- water
- polyglycolic acid
- Prior art date
Links
- 239000004215 Carbon black (E152) Substances 0.000 title claims abstract description 8
- 229930195733 hydrocarbon Natural products 0.000 title claims abstract description 8
- 150000002430 hydrocarbons Chemical class 0.000 title claims abstract description 8
- 229920000954 Polyglycolide Polymers 0.000 claims abstract description 62
- 239000004633 polyglycolic acid Substances 0.000 claims abstract description 62
- 229920005989 resin Polymers 0.000 claims abstract description 41
- 239000011347 resin Substances 0.000 claims abstract description 41
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 35
- 230000009467 reduction Effects 0.000 claims abstract description 27
- 238000000354 decomposition reaction Methods 0.000 claims description 52
- 239000012736 aqueous medium Substances 0.000 claims description 4
- 238000011084 recovery Methods 0.000 claims description 3
- 238000000465 moulding Methods 0.000 abstract description 8
- 238000013461 design Methods 0.000 abstract description 5
- 230000015556 catabolic process Effects 0.000 abstract description 3
- 238000006731 degradation reaction Methods 0.000 abstract description 2
- 239000003208 petroleum Substances 0.000 abstract description 2
- 230000000052 comparative effect Effects 0.000 description 14
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 13
- 238000012360 testing method Methods 0.000 description 13
- 230000035515 penetration Effects 0.000 description 9
- 229920001432 poly(L-lactide) Polymers 0.000 description 9
- JVTAAEKCZFNVCJ-REOHCLBHSA-N L-lactic acid Chemical compound C[C@H](O)C(O)=O JVTAAEKCZFNVCJ-REOHCLBHSA-N 0.000 description 8
- 229920006237 degradable polymer Polymers 0.000 description 8
- 230000004888 barrier function Effects 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- 239000004626 polylactic acid Substances 0.000 description 7
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 6
- 230000008859 change Effects 0.000 description 6
- 229920000747 poly(lactic acid) Polymers 0.000 description 6
- 238000005227 gel permeation chromatography Methods 0.000 description 5
- 230000007246 mechanism Effects 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 239000002994 raw material Substances 0.000 description 5
- 229920001577 copolymer Polymers 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 229920001519 homopolymer Polymers 0.000 description 4
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 238000003825 pressing Methods 0.000 description 4
- 239000012488 sample solution Substances 0.000 description 4
- BYEAHWXPCBROCE-UHFFFAOYSA-N 1,1,1,3,3,3-hexafluoropropan-2-ol Chemical compound FC(F)(F)C(O)C(F)(F)F BYEAHWXPCBROCE-UHFFFAOYSA-N 0.000 description 3
- 238000007654 immersion Methods 0.000 description 3
- 230000001771 impaired effect Effects 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 230000008439 repair process Effects 0.000 description 3
- 229920003232 aliphatic polyester Polymers 0.000 description 2
- 239000004568 cement Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 238000002425 crystallisation Methods 0.000 description 2
- 230000008025 crystallization Effects 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 239000000539 dimer Substances 0.000 description 2
- 238000001125 extrusion Methods 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 235000014655 lactic acid Nutrition 0.000 description 2
- 239000004310 lactic acid Substances 0.000 description 2
- JJTUDXZGHPGLLC-UHFFFAOYSA-N lactide Chemical compound CC1OC(=O)C(C)OC1=O JJTUDXZGHPGLLC-UHFFFAOYSA-N 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- 239000004576 sand Substances 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- UYCAUPASBSROMS-AWQJXPNKSA-M sodium;2,2,2-trifluoroacetate Chemical compound [Na+].[O-][13C](=O)[13C](F)(F)F UYCAUPASBSROMS-AWQJXPNKSA-M 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- RKDVKSZUMVYZHH-UHFFFAOYSA-N 1,4-dioxane-2,5-dione Chemical compound O=C1COC(=O)CO1 RKDVKSZUMVYZHH-UHFFFAOYSA-N 0.000 description 1
- 229920002101 Chitin Polymers 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 235000021120 animal protein Nutrition 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 238000009412 basement excavation Methods 0.000 description 1
- GSCLMSFRWBPUSK-UHFFFAOYSA-N beta-Butyrolactone Chemical compound CC1CC(=O)O1 GSCLMSFRWBPUSK-UHFFFAOYSA-N 0.000 description 1
- VEZXCJBBBCKRPI-UHFFFAOYSA-N beta-propiolactone Chemical compound O=C1CCO1 VEZXCJBBBCKRPI-UHFFFAOYSA-N 0.000 description 1
- 229920001222 biopolymer Polymers 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 238000012662 bulk polymerization Methods 0.000 description 1
- 238000011088 calibration curve Methods 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical group OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000011951 cationic catalyst Substances 0.000 description 1
- 238000004581 coalescence Methods 0.000 description 1
- 238000000748 compression moulding Methods 0.000 description 1
- 239000012792 core layer Substances 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 239000012760 heat stabilizer Substances 0.000 description 1
- 229920006158 high molecular weight polymer Polymers 0.000 description 1
- 239000004790 ingeo Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000001746 injection moulding Methods 0.000 description 1
- 239000011256 inorganic filler Substances 0.000 description 1
- 229910003475 inorganic filler Inorganic materials 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920001308 poly(aminoacid) Polymers 0.000 description 1
- 229920000111 poly(butyric acid) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 229960000380 propiolactone Drugs 0.000 description 1
- 230000002940 repellent Effects 0.000 description 1
- 239000005871 repellent Substances 0.000 description 1
- 238000007151 ring opening polymerisation reaction Methods 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 238000007711 solidification Methods 0.000 description 1
- 230000008023 solidification Effects 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000003856 thermoforming Methods 0.000 description 1
- 229920005992 thermoplastic resin Polymers 0.000 description 1
- -1 tin halide Chemical class 0.000 description 1
- 238000004078 waterproofing Methods 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- PAPBSGBWRJIAAV-UHFFFAOYSA-N ε-Caprolactone Chemical compound O=C1CCCCCO1 PAPBSGBWRJIAAV-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH DRILLING; MINING
- E21B—EARTH DRILLING, e.g. DEEP DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B33/00—Sealing or packing boreholes or wells
- E21B33/10—Sealing or packing boreholes or wells in the borehole
- E21B33/12—Packers; Plugs
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH DRILLING; MINING
- E21B—EARTH DRILLING, e.g. DEEP DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B17/00—Drilling rods or pipes; Flexible drill strings; Kellies; Drill collars; Sucker rods; Cables; Casings; Tubings
- E21B17/02—Couplings; joints
- E21B17/04—Couplings; joints between rod or the like and bit or between rod and rod or the like
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH DRILLING; MINING
- E21B—EARTH DRILLING, e.g. DEEP DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B23/00—Apparatus for displacing, setting, locking, releasing, or removing tools, packers or the like in the boreholes or wells
- E21B23/001—Self-propelling systems or apparatus, e.g. for moving tools within the horizontal portion of a borehole
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH DRILLING; MINING
- E21B—EARTH DRILLING, e.g. DEEP DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B23/00—Apparatus for displacing, setting, locking, releasing, or removing tools, packers or the like in the boreholes or wells
- E21B23/14—Apparatus for displacing, setting, locking, releasing, or removing tools, packers or the like in the boreholes or wells for displacing a cable or cable-operated tool, e.g. for logging or perforating operations in deviated wells
Definitions
- the present invention relates to a member for forming a downhole for repairing hydrocarbon resources such as oil and gas or a tool for repair itself or a part thereof.
- Downholes underground excavations
- hydrocarbon resources including oil and gas (hereinafter sometimes referred to as “petroleum”) from the ground.
- tools such as flack plugs (decomposable plugs), bridge plugs, cement retainers, perforation guns, ball sealers, seal plugs, packers, etc. (hereinafter collectively referred to as “downhole tools”) for repair are as follows: In many cases, it is disposed of in the downhole as it is without being collected after use, or disposed of by dropping it down (examples of such downhole tools or their modes of use are disclosed in, for example, Patent Documents 1 to 5). Be looked at).
- the whole or a member (downhole tool member) constituting a joint for promoting disintegration be formed of a degradable polymer.
- degradable polymers include polysaccharides such as starch or dextrin; animal protein polymers such as chitin and chitosan; polylactic acid (PLA, typically poly L-lactic acid (PLLA)), polyglycolic acid (PGA), aliphatic polyesters such as polybutyric acid and polyvaleric acid; and polyamino acids, polyethylene oxide and the like (Patent Documents 1 and 2).
- PLA poly L-lactic acid
- PGA polyglycolic acid
- aliphatic polyesters such as polybutyric acid and polyvaleric acid
- Patent Documents 1 and 2 a technique for designing the strength and time until the downhole tool member collapses using these degradable polymers has not always been satisfactory. This is because it was difficult to accurately determine the degradation behavior of the degradable polymer.
- the main object of the present invention is to provide a member for a downhole tool capable of more accurate design of strength and time until collapse through appropriate selection and molding of a degradable polymer. Objective.
- the hydrocarbon resource recovery downhole tool member of the present invention was developed to achieve the above-mentioned object, and the effective thickness of the polyglycolic acid resin having a weight average molecular weight of 70,000 or more is that of surface decomposition. It consists of a molded body having a thickness that is 1/2 or more of the critical thickness, and is characterized in that the rate of thickness reduction in water is constant with time.
- the polyglycolic acid resin not only has excellent initial strength, but according to its appropriately designed molded body, it differs from other degradable polymers specifically in water. It has been found that the thickness reduction rate of the film exhibits a constant characteristic with respect to time (in other words, a linear thickness reduction rate). Therefore, it is possible to design the strength and holding time by setting the effective thickness that contributes to the strength maintenance of the downhole tool member and the required characteristics such as plug or seal according to the maintenance time until the member collapses become.
- the above-mentioned linear thickness reduction rate characteristic of the polyglycolic acid resin molded article is that the hydrolysis proceeds as surface decomposition through the excellent water (vapor) barrier property of the polyglycolic acid resin molded article (in other words, For example, the rate of progress of water molecules from the surface to the interface between the low molecular weight polymer layer that has been hydrolyzed and does not exhibit barrier properties in the molded body and the core layer composed of undecomposed high molecular weight polymer This is because penetration proceeds at a rate that is almost the same as the rate of penetration), and in the form of polyglycolic acid resin fine particles that do not have a clear interface or other degradable polymers with poor barrier properties. It cannot be obtained by decomposition.
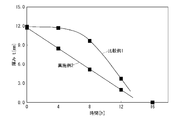
- the rate of decrease in the effective thickness of the molded article is moderate at first, but increases rapidly from the middle (see Comparative Examples below).
- the effective thickness of the polyglycolic acid resin molded body (that is, the thickness of the portion governing the characteristics of the molded body formed as a tool member) is greater than the critical thickness as the thickness of the boundary that changes from block decomposition to surface decomposition.
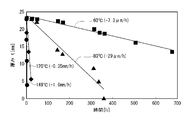
- the schematic diagram of the principal part of the flap plug as an example of a downhaul tool The graph which shows the time change data of the thickness in various temperature of a PGA molded object.
- the graph which shows the temperature dependence of the thickness decreasing speed of a PGA molded object (Arrhenius plot).
- the polyglycolic acid resin (PGA resin) used in the present invention is added to a glycolic acid homopolymer (that is, polyglycolic acid (PGA)) consisting only of a glycolic acid unit (—OCH 2 —CO—) as a repeating unit.
- Glycolic acid co-polymer containing other monomer (comonomer) units preferably hydroxyl carboxylic acid units such as lactic acid, in a proportion of 50% by weight or less, preferably 30% by weight or less, more preferably 10% by weight or less. Includes coalescence.
- the downhole tool member of the present invention can exhibit surface decomposition characteristics due to the excellent barrier property of the polyglycolic acid resin, and if included excessively, the excellent barrier property is impaired, resulting in In particular, the linearity of the rate of thickness reduction is impaired, which is not preferable.
- the polyglycolic acid resin those having a weight average molecular weight of 70,000 or more, preferably 100,000 to 500,000 are used.
- the weight average molecular weight is less than 70,000, the initial strength characteristics required for the tool member are impaired.
- the weight average molecular weight exceeds 500,000, molding processability is deteriorated, which is not preferable.
- glycolide which is a dimer of glycolic acid
- catalyst for example, tin organic carboxylate, tin halide, halogenated halide. It is preferable to employ a method of ring-opening polymerization by heating to a temperature of about 120 to 250 ° C. in the presence of a cationic catalyst such as antimony) and substantially in the absence of a solvent (ie, bulk polymerization conditions).
- lactide typified by lactide which is a dimer of lactic acid
- lactones for example, caprolactone, ⁇ -propiolactone, ⁇ -butyrolactone 1 It is preferable to use more than one species.
- the melting point (Tm) of polyglycolic acid resin is generally 200 ° C. or higher.
- polyglycolic acid (PGA) has a melting point of about 220 ° C., a glass transition temperature of about 38 ° C., and a crystallization temperature of about 90 ° C.
- the melting points of these polyglycolic acid resins vary depending on the molecular weight of the polyglycolic acid resin, the type of comonomer used, and the like.
- the downhole tool member is usually formed of a polyglycolic acid resin alone, but for the purpose of controlling its degradability and the like, other aliphatic polyesters (for example, the above-mentioned glycolic acid copolymer) are used.
- Other thermoplastic resins such as aromatic polyesters, elastomers, and the like.
- the addition amount is an amount that does not prevent the polyglycolic acid resin from existing as a matrix resin, more specifically Specifically, it should be suppressed to less than 30% by weight, preferably less than 20% by weight, more preferably less than 10% by weight.
- the polyglycolic acid resin has a heat stabilizer, a light stabilizer, an inorganic filler, a plasticizer, a moisture-proofing agent, a waterproofing agent, a water repellent, a lubricant, a decomposition as long as it does not contradict the purpose of the present invention.
- Various additives such as an accelerator and a decomposition retarder can be added.
- thermoforming method such as injection molding, melt extrusion molding, solidification extrusion molding, compression molding, centrifugal molding, or the like, or necessary for the polyglycolic acid resin (and optionally other optional components) obtained as described above
- Various downhaul tools such as flack plugs, bridge plugs, cement retainers, perforation guns, ball sealers, seal plugs, packers, etc., as exemplified in Patent Documents 1 to 5, etc. Is formed into the shape of a member constituting the whole or a part thereof. At that time, in order to improve the controllability of the tool collapse time due to the linearity of thickness reduction, for example, as shown in FIG.
- the joint portion between the cylindrical, prismatic, or hollow rod-shaped members 11-11 made of a porous resin or metal is composed of the member 12 made of polyglycolic acid resin, and the overall shape constitutes, for example, a generally rod-shaped tool 10. It can also be done.
- the thickness t from the surface 12a exposed to the water of the member 12 (more practically, in the aqueous working environment medium where the downhole tool exists) to the side surface of the protruding portion 11a of the member 11 becomes an effective thickness. It will dominate the time until the collapse or decomposition of.
- the effective thickness in such a case is 1/2 of the critical thickness.
- the diameter may be taken as the effective thickness.
- the obtained polyglycolic acid resin molded product is subjected to a heat treatment at a crystallization temperature Tc1 at the time of temperature rise (about 90 ° C. for glycolic acid homopolymer) and below the melting point for about 1 minute to 10 hours. It is also preferable to improve the water vapor barrier property and the linearity of the thickness reduction rate by improving the weight crystallinity to about 20% or more, particularly 30 to 60%.
- the effective thickness of the polyglycolic acid resin molded body constituting the downhole tool member is set to 1/2 or more of the critical thickness for surface decomposition.
- the critical thickness Lc of this surface decomposition was determined as follows.
- the thickness reduction rate in water was investigated using a PGA molded piece (thickness: 23 mm) (Example 1 described later). As a result, it was found that the thickness (one side) decreased at a constant rate with respect to time (FIG. 2). Further, it was found that the molecular weight of the undecomposed portion was not changed from the molecular weight before decomposition, and the molded piece was decomposed by the surface decomposition mechanism. At this time, since the penetration rate of water is the controlling factor of the decomposition rate, it can be said that the thickness reduction rate (decomposition rate) is equivalent to the penetration rate of water.
- V water penetration rate
- K absolute temperature
- the critical thickness Lc of the material whose PGA decomposition mechanism changes from bulk decomposition to surface decomposition is estimated from the following equation (3) based on the results of the above equations (1) and (2) at each temperature (K). Can do.
- Critical thickness Lc 2 ⁇ ⁇ ⁇ V. . . (3)
- the critical thickness of PGA was 770 ⁇ m in 40 ° C. water, 812 ⁇ m in 60 ° C. water, and 852 ⁇ m in 80 ° C. water.
- the effective thickness of the polyglycolic acid resin molded body constituting the downhole tool member is set to 1 / (1) of the critical thickness ( ⁇ ) of surface decomposition under the environmental conditions of the downhole tool mainly determined by temperature.
- the collapse time of the downhole tool can be designed based on the linearity of the thickness reduction rate of the downhole tool member.
- the effective thickness of the PGA resin molded body constituting the downhole tool member is a required characteristic of the tool member (for example, a bonding strength characteristic in the case of a coupling member, or a plug or sealer in the case of being used as a plug or sealer itself). It is defined as the reduced thickness allowed before the seal function) disappears.
- the effective thickness of the tool member molded body is at least 1 times the critical thickness when both main surfaces are exposed to the working environment aqueous medium, and at least 1/2 the critical thickness when only one surface is exposed, In each case, considering the strength retention characteristics, it is generally preferable to set the value to 1.2 times or more, more preferably 1.5 times or more of the above value.
- the member for downhaul tool of the present invention has a value above the above and for a predetermined time in a working environment aqueous medium at a predetermined temperature of 20 to 180 ° C., for example, for operations such as formation, repair or expansion of the downhole tool. It is formed with an effective thickness designed to spontaneously collapse after it has been used. However, in order to promote the collapse after the end of the work, if necessary, the ambient environment temperature is increased by injection of, for example, heating steam, and the collapse. Can also be promoted.
- HFIP hexafluoroisopropanol
- the molding piece for the measurement test of the thickness reduction rate by immersion in water was formed as follows. First, a 5 mm thick resin sheet was produced by press molding using a stainless steel mold 5 cm long and 5 mm deep. The pressing conditions were 260 ° C., preheating for 4 minutes, and pressurization was 5 MPa for 2 minutes. After pressing, the plate was quenched with a water cooling plate. Subsequently, several sheets produced were overlapped, and a molded piece having a predetermined thickness (12 mm or 23 mm) was produced by press molding.
- the pressing conditions were 260 ° C., preheating for 7 minutes, pressurization was 5 Mpa for 3 minutes, and after pressing, the plate was quenched with a water cooling plate.
- the produced molded piece was crystallized by performing heat treatment in an oven at 120 ° C. for 1 hour and then used for the experiment.
- Example 1 A predetermined amount of a molded piece having a thickness of 23 mm obtained as described above using a glycolic acid homopolymer (PGA, manufactured by Kureha Co., Ltd.) having an initial molecular weight Mw of 200,000 is prepared.
- PGA glycolic acid homopolymer
- Example 2 Using the same PGA as used in Example 1, four 12 mm-thick molded pieces were prepared by the above method and subjected to an underwater decomposition test at a temperature of 149 ° C., respectively. Was measured.
- Example 1 A test piece having a thickness of 12 mm was prepared in the same manner as in Example 2 except that crystalline polylactic acid having a weight average molecular weight of 260,000 (PLLA, “Ingeo Biopolymer 4032D” manufactured by Nature Works) was used. The time change of thickness reduction was measured.
- PLLA crystalline polylactic acid having a weight average molecular weight of 260,000
- Example 2 exhibits a good thickness reduction rate linearity
- the PLA molded piece of Comparative Example 1 initially shows a gradual reduction rate, and the thickness reduction rate increases abruptly from the middle. Thus, no linearity is observed in the thickness reduction rate.
- Example 3 An underwater decomposition test was conducted in the same manner as in Example 2 except that the temperature was 120 ° C.
- Example 4 An underwater decomposition test was conducted in the same manner as in Example 2 except that an 800 ml glass bottle was used instead of the autoclave and the container was stored in an oven set at 80 ° C.
- Example 5 An underwater decomposition test was conducted in the same manner as in Example 2 except that an 800 ml glass bottle was used instead of the autoclave and the container was stored in an oven set at 60 ° C.
- Example 8 A molded piece was obtained in the same manner as in Example 2 except that a composition in which 90 parts by weight of PGA used in Example 1 was mixed with 10 parts by weight of crystalline polylactic acid (PLLA) used in Comparative Example 1 was used as a raw material for the molded piece. Obtained and subjected to an underwater decomposition test.
- a composition in which 90 parts by weight of PGA used in Example 1 was mixed with 10 parts by weight of crystalline polylactic acid (PLLA) used in Comparative Example 1 was used as a raw material for the molded piece. Obtained and subjected to an underwater decomposition test.
- PLLA crystalline polylactic acid
- a molded piece was obtained in the same manner as in Example 2 except that a composition in which 30 parts by weight of PLLA used in Comparative Example 1 was mixed with 70 parts by weight of PGA used in Example 1 as a raw material for the molded piece was subjected to an underwater decomposition test. I did it.
- Example 3 The same procedure as in Example 1 was performed except that a composition obtained by mixing 50 parts by weight of PLLA used in Comparative Example 1 with 50 parts by weight of PGA used in Example 1 was used as a raw material for the molded pieces.
- a tool for forming or repairing a downhole for recovering hydrocarbon resources including oil and gas, or a member for forming a part thereof (member for a downhole tool)
- a molded product of polyglycolic acid resin having a weight average molecular weight of 70,000 or more and an effective thickness of 1/2 or more of the critical thickness of surface decomposition, it gives linearity of the rate of thickness reduction in water. This provides a member for a downhaul tool that allows a more accurate design of strength and time to collapse.
Abstract
Description
本発明で使用するポリグリコール酸樹脂(PGA樹脂)は、繰り返し単位としてグリコール酸単位(-OCH2-CO-)のみからなるグリコール酸単独重合体(すなわちポリグリコ-ル酸(PGA))に加えて、他の単量体(コモノマー)単位、好ましくは乳酸等のヒドロキシルカルボン酸単位、を50重量%以下、好ましくは30重量%以下、更に好ましくは10重量%以下、の割合で含むグリコール酸共重合体を含む。他の単量体単位を含む共重合体とすることにより、ポリグリコ-ル酸樹脂の加水分解速度、結晶性等をある程度調整することができる。但し、本発明のダウンホールツール用部材が表面分解特性を示し得るのは、ポリグリコール酸樹脂のもつ優れたバリア性によるものであり、過剰に含ませると、優れたバリア性が損なわれ、結果的に厚み減少速度の直線性が損なわれるので、好ましくない。
本発明においては、ダウンホールツール部材を構成するポリグリコール酸樹脂成形体の有効厚みを、表面分解の臨界厚みの1/2以上に設定する。本発明者らの研究によれば、この表面分解の臨界厚みLcは以下のようにして決定された。
τ=exp(8240/K - 20.7) ...(1)
V=exp(21.332-8519.7/K) ...(2)
臨界厚みLc = 2×τ×V ...(3)
その結果、PGAの臨界厚みは40℃水中で770μm、60℃水中で812μm、80℃水中で852μmであった。
ダウンホールツール部材を構成するPGA樹脂成形体の有効厚みは、当該ツール部材の要求特性(例えば、結合部材である場合は結合強度特性、それ自体がプラグあるいはシーラーとして用いられる場合は、そのプラグあるいはシール機能)を消失するまでに許容される減少厚み、として定義される。ツール部材成形体の有効厚みは、その主たる二表面がともに作業環境水性媒体に露出される場合は臨界厚みの1倍以上、一面のみが露出される場合は臨界厚みの1/2以上、とし、それぞれの場合について、強度保持特性を考慮して、一般に、上記値の1.2倍以上、更には1.5倍以上、に設定することが好ましい。
ポリグリコ-ル酸(PGA)およびポリ乳酸(PLA)の重量平均分子量(Mw)は、各10mgの試料を、トリフルオロ酢酸ナトリウムを5mMの濃度で溶解させたヘキサフルオロイソプロパノール(HFIP)に、溶解させて10mLとした後、メンブレンフィルタ―で濾過して試料溶液を得た。この試料溶液の10μlをゲルパーミエーションクロマトグラフィー(GPC)装置に注入して、下記条件で分子量を測定した。なお、試料溶液は、溶解後、30分以内にGPC装置に注入した。
<GPC測定条件>
装置:Shimazu LC-9A,
カラム:昭和電工(株)製 HFIP-806M 2本(直列接続)+プレカラム:HFIP-LG 1本
カラム温度:40℃、
溶離液:トリフルオロ酢酸ナトリウムを5mMの濃度で溶解させたHFIP溶液、
流速:1mL/分、
検出器:示差屈折率計
分子量校正:分子量の異なる標準分子量のポリメタクリル酸メチル5種(POLYMER LABORATORIES Ltd.製)を用いて作成した分子量の検量線データを使用。
後記実施例および比較例の樹脂(組成物)について、水中浸漬による厚み減少速度の測定試験用成型片を、以下のようにして形成した。
まず縦横5cm且つ深さ5mmのステンレスの型枠を用いて、プレス成型により厚み5mmの樹脂シートを作製した。プレス条件は、260℃、予熱4分間、加圧は5MPaで2分間とし、プレス後には水冷却板にて急冷した。次いで作製したシートを数枚重ね合わせ、プレス成型により所定の厚み(12mmまたは23mm)の成型片を作製した。プレス条件は260℃、予熱7分間、加圧は5Mpaで3分間とし、プレス後には水冷却板にて急冷した。作製した成型片をオーブン120℃で1時間熱処理を行うことで結晶化させたのち実験に使用した。
1L-オートクレーブ中に、上記のようにして得た樹脂成型片の1個を入れ、脱イオン水を満たして、所定の温度および時間の浸漬試験を行なった。ついで浸漬後の成型片を取り出し、その断面を切り出して、ドライルーム内に一晩放置し乾燥させた。その芯部(未分解の硬い部分)の厚みを測定して、当初厚みとの差により、減少厚み(両面からの減少厚みの合計量の1/2=Δt)を測定した。
当初分子量Mw=20万であるグリコール酸単独重合体(PGA,(株)クレハ製)を用いて上記のようにして得た厚み23mmの成型片の所定量を用意し、温度60℃、80℃、120℃、149℃で、それぞれ上記の方法で水中分解試験を行い、減少厚み(片側)(=Δt)の時間変化を測定した。結果を図2にプロットして示す。図2のプロットを見れば、いずれの温度においても良好な厚み減少速度の直線性が認められる。図2のデータをもとに、片側の厚み変化速度の対数値ln(Δt/h)を縦軸に、絶対温度の逆数(1/K)を横軸にとったアレニウスプロットを図3に示す。これから前述した厚み減少速度(片側)(=V)の温度依存性を示す式(2)(以下に再掲する)が得られた。
V=Δt(mm)/h=exp(21.332-8519.7/K) ...(2)
実施例1で用いたものと同じPGAを用いて厚み12mmの成型片を4枚、上記の方法により調製し、温度149℃で、それぞれ上記の方法で水中分解試験を行い、厚み減少の時間変化を測定した。
重量平均分子量が26万である結晶性ポリ乳酸(PLLA,Nature Works製「Ingeo Biopolymer 4032D」)を用いる以外は実施例2と同様にして厚み12mmの試験片を調製し、水中分解試験を行い、厚み減少の時間変化を測定した。
温度を120℃とした以外は実施例2と同様に、水中分解試験を行なった。
容器としてオートクレーブにかわり800mlガラス瓶を使用し、80℃に設定したオーブンに保管した以外は実施例2と同様に、水中分解試験を行なった。
容器としてオートクレーブにかわり800mlガラス瓶を使用し、60℃に設定したオーブンに保管した以外は実施例2と同様に、水中分解試験を行なった。
成型片の原料として実施例1で用いたPGA50重量部にタルク(日本タルク製、「ミクロエースL-1」、体積基準50%平均粒子径=5μm)を50重量部混合した組成物を使用した以外は実施例2と同様に成型片を得、水中分解試験を行なった。
成型片の原料として実施例1で用いたPGA50重量部にケイ砂(JFEミネラル株式会社製、珪砂8号、粒子径範囲=150-212μm)を50重量部混合した組成物を使用した以外は実施例2と同様に成型片を得、水中分解試験を行なった。
成型片の原料として実施例1で用いたPGA90重量部に比較例1で用いた結晶性ポリ乳酸(PLLA)を10重量部混合した組成物を使用した以外は実施例2と同様に成型片を得、水中分解試験を行なった。
成型片の原料として実施例1で用いたPGA50重量部に比較例1で用いたPLLAを50重量部混合した組成物を使用した以外は実施例1と同様に行なった。
Claims (7)
- 重量平均分子量が7万以上であるポリグリコール酸樹脂の、有効厚みが表面分解の臨界厚みの1/2以上である成形体からなり、水中での厚み減少速度が時間に対して一定であることを特徴とする、炭化水素資源回収ダウンホールツール用部材。
- ポリグリコール酸樹脂成形体が結晶化処理されている請求項1に記載のダウンホールツール用部材。
- その主たる二表面のうち一面のみが作業環境水性媒体に露出されるダウンホールツール用部材であり、有効厚みが表面分解の臨界厚みの1/2以上に設定されている請求項1または2に記載のダウンホールツール用部材。
- 有効厚みが表面分解の臨界厚みの3/4以上に設定されている請求項3に記載のダウンホールツール用部材。
- その主たる二表面がともに作業環境水性媒体に露出されるダウンホールツール用部材であり、有効厚みが表面分解の臨界厚み以上に設定されている請求項1または2に記載のダウンホールツール用部材。
- 有効厚みが表面分解の臨界厚みの1.5倍以上に設定されている請求項5に記載のダウンホールツール用部材。
- 複数の非水中分解性の部材間の結合部を形成する部材であり、全体形状が概ね棒状のツールを構成する請求項1~6のいずれかに記載のダウンホールツール用部材。
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201380013534.1A CN104204404B (zh) | 2012-06-07 | 2013-04-12 | 烃资源回收钻井工具用构件 |
| EP13801222.4A EP2860344A4 (en) | 2012-06-07 | 2013-04-12 | ELEMENT FOR DOWNHOLE TOOL FOR HYDROCARBON RESOURCE COLLECTION WELL |
| EP19184094.1A EP3569815A1 (en) | 2012-06-07 | 2013-04-12 | Member for hydrocarbon resource collection downhole tool |
| JP2014519870A JP6084609B2 (ja) | 2012-06-07 | 2013-04-12 | 炭化水素資源回収ダウンホールツール用部材 |
| CA2868975A CA2868975C (en) | 2012-06-07 | 2013-04-12 | Member for hydrocarbon resource collection downhole tool |
| MX2014012613A MX2014012613A (es) | 2012-06-07 | 2013-04-12 | Miembro para herramienta de fondo de pozo para recuperacion de recursos de hidrocarburos. |
| AU2013272915A AU2013272915B2 (en) | 2012-06-07 | 2013-04-12 | Member for hydrocarbon resource collection downhole tool |
| US14/395,654 US9267351B2 (en) | 2012-06-07 | 2013-04-12 | Member for hydrocarbon resource collection downhole tool |
| US14/984,667 US10030464B2 (en) | 2012-06-07 | 2015-12-30 | Member for hydrocarbon resource collection downhole tool |
| US16/010,733 US10626694B2 (en) | 2012-06-07 | 2018-06-18 | Downhole tool member for hydrocarbon resource recovery |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012130055 | 2012-06-07 | ||
| JP2012-130055 | 2012-06-07 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/395,654 A-371-Of-International US9267351B2 (en) | 2012-06-07 | 2013-04-12 | Member for hydrocarbon resource collection downhole tool |
| US14/984,667 Continuation US10030464B2 (en) | 2012-06-07 | 2015-12-30 | Member for hydrocarbon resource collection downhole tool |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013183363A1 true WO2013183363A1 (ja) | 2013-12-12 |
Family
ID=49711764
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/061075 WO2013183363A1 (ja) | 2012-06-07 | 2013-04-12 | 炭化水素資源回収ダウンホールツール用部材 |
Country Status (8)
| Country | Link |
|---|---|
| US (3) | US9267351B2 (ja) |
| EP (2) | EP3569815A1 (ja) |
| JP (1) | JP6084609B2 (ja) |
| CN (2) | CN104204404B (ja) |
| AU (1) | AU2013272915B2 (ja) |
| CA (1) | CA2868975C (ja) |
| MX (1) | MX2014012613A (ja) |
| WO (1) | WO2013183363A1 (ja) |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014010267A1 (ja) | 2012-07-10 | 2014-01-16 | 株式会社クレハ | 炭化水素資源回収ダウンホールツール用部材 |
| WO2014024827A1 (ja) | 2012-08-08 | 2014-02-13 | 株式会社クレハ | 炭化水素資源回収用ボールシーラーならびにその製造方法及びそれを用いる坑井の処理方法 |
| WO2015098913A1 (ja) * | 2013-12-27 | 2015-07-02 | 株式会社クレハ | ダウンホールツール用の拡径可能な環状の分解性シール部材、及び坑井掘削用プラグ、並びに坑井掘削方法 |
| WO2015098801A1 (ja) * | 2013-12-26 | 2015-07-02 | 株式会社クレハ | ダウンホールツールまたはダウンホールツール部材及び分解性樹脂組成物、並びに炭化水素資源の回収方法 |
| WO2015098597A1 (ja) * | 2013-12-26 | 2015-07-02 | 株式会社クレハ | 炭化水素資源回収用ボールシーラーならびにその製造方法及びそれを用いる坑井の処理方法 |
| WO2015098849A1 (ja) * | 2013-12-27 | 2015-07-02 | 株式会社クレハ | 分解性を有するゴム材料から形成される拡径可能な環状のゴム部材を備える坑井掘削用プラグ |
| WO2015133545A1 (ja) * | 2014-03-07 | 2015-09-11 | 株式会社クレハ | ダウンホールツール用分解性ゴム部材、分解性シール部材、分解性保護部材、ダウンホールツール、及び坑井掘削方法 |
| WO2015133544A1 (ja) * | 2014-03-07 | 2015-09-11 | 株式会社クレハ | 崩壊性のダウンホールツール用シール部材、ダウンホールツール、及び坑井掘削方法 |
| WO2016047502A1 (ja) * | 2014-09-22 | 2016-03-31 | 株式会社クレハ | 反応性金属を含有するダウンホールツール部材及び分解性樹脂組成物を含有するダウンホールツール部材を備えるダウンホールツール、並びに坑井掘削方法 |
| JP2016060900A (ja) * | 2014-09-22 | 2016-04-25 | 株式会社クレハ | 反応性金属及び分解性樹脂組成物を含有する坑井掘削用組成物、坑井掘削用成形品、及び坑井掘削方法 |
| US20160289374A1 (en) * | 2015-03-30 | 2016-10-06 | Kureha Corporation | Polyglycolic acid molded article, component for downhole tool, and method of producing polyglycolic acid molded article |
| US9926764B2 (en) | 2014-03-11 | 2018-03-27 | Kureha Corporation | Molded product having effective thickness of 1 mm or more and containing aliphatic polyester resin, and downhole tool member for hydrocarbon resource recovery |
| WO2020013216A1 (ja) * | 2018-07-10 | 2020-01-16 | 株式会社クレハ | ダウンホールツールおよび坑井掘削方法 |
| WO2022075484A1 (ja) | 2020-10-09 | 2022-04-14 | 株式会社クレハ | プラグ、ダウンホールツールおよび坑井処理方法 |
| WO2023053955A1 (ja) | 2021-09-29 | 2023-04-06 | 株式会社クレハ | 樹脂組成物、ダウンホールツールまたはその部材、プラグおよび坑井処理方法 |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9500061B2 (en) | 2008-12-23 | 2016-11-22 | Frazier Technologies, L.L.C. | Downhole tools having non-toxic degradable elements and methods of using the same |
| US10337279B2 (en) | 2014-04-02 | 2019-07-02 | Magnum Oil Tools International, Ltd. | Dissolvable downhole tools comprising both degradable polymer acid and degradable metal alloy elements |
| CA2868975C (en) * | 2012-06-07 | 2017-02-14 | Kureha Corporation | Member for hydrocarbon resource collection downhole tool |
| JP6327946B2 (ja) * | 2013-05-31 | 2018-05-23 | 株式会社クレハ | 分解性材料から形成されるマンドレルを備える坑井掘削用プラグ |
| JP6327933B2 (ja) | 2013-06-28 | 2018-05-23 | 株式会社クレハ | ダウンホールツール用ゴム部材、及びダウンホールツール、並びに炭化水素資源の回収方法 |
| CA2886988C (en) | 2014-04-02 | 2017-08-29 | Magnum Oil Tools International, Ltd. | Dissolvable aluminum downhole plug |
| CN114829740A (zh) | 2019-12-18 | 2022-07-29 | 索尔维特殊聚合物美国有限责任公司 | 包含支化聚(羟基酸)的井下工具构件 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050205266A1 (en) | 2004-03-18 | 2005-09-22 | Todd Bradley I | Biodegradable downhole tools |
| US20050205265A1 (en) | 2004-03-18 | 2005-09-22 | Todd Bradley L | One-time use composite tool formed of fibers and a biodegradable resin |
| JP2005534746A (ja) * | 2002-07-31 | 2005-11-17 | ダウ・コーニング・コーポレイション | 掘削のフルイドロス制御用シリコーン樹脂 |
| JP2008088364A (ja) * | 2006-10-04 | 2008-04-17 | Mitsui Chemicals Inc | 脂肪族ポリエステル系樹脂組成物およびその成形体 |
| US20090101334A1 (en) | 2007-10-18 | 2009-04-23 | Belgin Baser | Multilayered ball sealer and method of use thereof |
| US7621336B2 (en) | 2004-08-30 | 2009-11-24 | Halliburton Energy Services, Inc. | Casing shoes and methods of reverse-circulation cementing of casing |
| US7762342B2 (en) | 2003-10-22 | 2010-07-27 | Baker Hughes Incorporated | Apparatus for providing a temporary degradable barrier in a flow pathway |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20030125508A1 (en) | 2001-10-31 | 2003-07-03 | Kazuyuki Yamane | Crystalline polyglycolic acid, polyglycolic acid composition and production process thereof |
| US20050113263A1 (en) | 2002-10-28 | 2005-05-26 | Brown J. E. | Differential etching in acid fracturing |
| US7265079B2 (en) | 2002-10-28 | 2007-09-04 | Schlumberger Technology Corporation | Self-destructing filter cake |
| US20060058197A1 (en) | 2004-09-15 | 2006-03-16 | Brown J E | Selective fracture face dissolution |
| US7775278B2 (en) | 2004-09-01 | 2010-08-17 | Schlumberger Technology Corporation | Degradable material assisted diversion or isolation |
| US7350572B2 (en) | 2004-09-01 | 2008-04-01 | Schlumberger Technology Corporation | Methods for controlling fluid loss |
| US7275596B2 (en) | 2005-06-20 | 2007-10-02 | Schlumberger Technology Corporation | Method of using degradable fiber systems for stimulation |
| US7380600B2 (en) * | 2004-09-01 | 2008-06-03 | Schlumberger Technology Corporation | Degradable material assisted diversion or isolation |
| US20080202764A1 (en) * | 2007-02-22 | 2008-08-28 | Halliburton Energy Services, Inc. | Consumable downhole tools |
| US7757773B2 (en) * | 2007-07-25 | 2010-07-20 | Schlumberger Technology Corporation | Latch assembly for wellbore operations |
| US7775286B2 (en) * | 2008-08-06 | 2010-08-17 | Baker Hughes Incorporated | Convertible downhole devices and method of performing downhole operations using convertible downhole devices |
| US9127527B2 (en) * | 2009-04-21 | 2015-09-08 | W. Lynn Frazier | Decomposable impediments for downhole tools and methods for using same |
| CA2868975C (en) * | 2012-06-07 | 2017-02-14 | Kureha Corporation | Member for hydrocarbon resource collection downhole tool |
| CA2872240C (en) * | 2012-07-10 | 2016-12-20 | Kureha Corporation | Downhole tool member for hydrocarbon resource recovery |
-
2013
- 2013-04-12 CA CA2868975A patent/CA2868975C/en active Active
- 2013-04-12 US US14/395,654 patent/US9267351B2/en active Active
- 2013-04-12 EP EP19184094.1A patent/EP3569815A1/en not_active Withdrawn
- 2013-04-12 MX MX2014012613A patent/MX2014012613A/es unknown
- 2013-04-12 JP JP2014519870A patent/JP6084609B2/ja active Active
- 2013-04-12 CN CN201380013534.1A patent/CN104204404B/zh active Active
- 2013-04-12 WO PCT/JP2013/061075 patent/WO2013183363A1/ja active Application Filing
- 2013-04-12 AU AU2013272915A patent/AU2013272915B2/en active Active
- 2013-04-12 CN CN201611043322.XA patent/CN106761546B/zh active Active
- 2013-04-12 EP EP13801222.4A patent/EP2860344A4/en not_active Ceased
-
2015
- 2015-12-30 US US14/984,667 patent/US10030464B2/en active Active
-
2018
- 2018-06-18 US US16/010,733 patent/US10626694B2/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005534746A (ja) * | 2002-07-31 | 2005-11-17 | ダウ・コーニング・コーポレイション | 掘削のフルイドロス制御用シリコーン樹脂 |
| US7762342B2 (en) | 2003-10-22 | 2010-07-27 | Baker Hughes Incorporated | Apparatus for providing a temporary degradable barrier in a flow pathway |
| US20050205266A1 (en) | 2004-03-18 | 2005-09-22 | Todd Bradley I | Biodegradable downhole tools |
| US20050205265A1 (en) | 2004-03-18 | 2005-09-22 | Todd Bradley L | One-time use composite tool formed of fibers and a biodegradable resin |
| US7621336B2 (en) | 2004-08-30 | 2009-11-24 | Halliburton Energy Services, Inc. | Casing shoes and methods of reverse-circulation cementing of casing |
| JP2008088364A (ja) * | 2006-10-04 | 2008-04-17 | Mitsui Chemicals Inc | 脂肪族ポリエステル系樹脂組成物およびその成形体 |
| US20090101334A1 (en) | 2007-10-18 | 2009-04-23 | Belgin Baser | Multilayered ball sealer and method of use thereof |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2860344A4 |
Cited By (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014010267A1 (ja) | 2012-07-10 | 2014-01-16 | 株式会社クレハ | 炭化水素資源回収ダウンホールツール用部材 |
| WO2014024827A1 (ja) | 2012-08-08 | 2014-02-13 | 株式会社クレハ | 炭化水素資源回収用ボールシーラーならびにその製造方法及びそれを用いる坑井の処理方法 |
| US9914871B2 (en) | 2013-12-26 | 2018-03-13 | Kureha Corporation | Ball sealer for hydrocarbon resource recovery, method for manufacturing same, and method for treating borehole using same |
| JPWO2015098801A1 (ja) * | 2013-12-26 | 2017-03-23 | 株式会社クレハ | ダウンホールツールまたはダウンホールツール部材及び分解性樹脂組成物、並びに炭化水素資源の回収方法 |
| WO2015098597A1 (ja) * | 2013-12-26 | 2015-07-02 | 株式会社クレハ | 炭化水素資源回収用ボールシーラーならびにその製造方法及びそれを用いる坑井の処理方法 |
| JP5955474B2 (ja) * | 2013-12-26 | 2016-07-20 | 株式会社クレハ | 炭化水素資源回収用ボールシーラーならびにその製造方法及びそれを用いる坑井の処理方法 |
| WO2015098801A1 (ja) * | 2013-12-26 | 2015-07-02 | 株式会社クレハ | ダウンホールツールまたはダウンホールツール部材及び分解性樹脂組成物、並びに炭化水素資源の回収方法 |
| JP2015143458A (ja) * | 2013-12-27 | 2015-08-06 | 株式会社クレハ | ダウンホールツール用の拡径可能な環状の分解性シール部材、及び坑井掘削用プラグ、並びに坑井掘削方法 |
| US10619084B2 (en) | 2013-12-27 | 2020-04-14 | Kureha Corporation | Plug for well drilling provided with diametrically expandable annular rubber member formed from degradable rubber material |
| WO2015098913A1 (ja) * | 2013-12-27 | 2015-07-02 | 株式会社クレハ | ダウンホールツール用の拡径可能な環状の分解性シール部材、及び坑井掘削用プラグ、並びに坑井掘削方法 |
| CN105637174B (zh) * | 2013-12-27 | 2019-05-03 | 株式会社吴羽 | 钻井工具用可扩径的环状分解性密封构件、钻井用堵塞器、以及钻井方法 |
| US10208559B2 (en) | 2013-12-27 | 2019-02-19 | Kureha Corporation | Diameter-expandable annular degradable seal member for downhole tool, plug for well drilling, and method for well drilling |
| JP2015143459A (ja) * | 2013-12-27 | 2015-08-06 | 株式会社クレハ | 分解性を有するゴム材料から形成される拡径可能な環状のゴム部材を備える坑井掘削用プラグ |
| CN105593464A (zh) * | 2013-12-27 | 2016-05-18 | 株式会社吴羽 | 具备由具有降解性的橡胶材料形成的可扩径的环状橡胶构件的钻井用堵塞器 |
| CN105637174A (zh) * | 2013-12-27 | 2016-06-01 | 株式会社吴羽 | 钻井工具用可扩径的环状分解性密封构件、钻井用堵塞器、以及钻井方法 |
| WO2015098849A1 (ja) * | 2013-12-27 | 2015-07-02 | 株式会社クレハ | 分解性を有するゴム材料から形成される拡径可能な環状のゴム部材を備える坑井掘削用プラグ |
| US10280699B2 (en) | 2014-03-07 | 2019-05-07 | Kureha Corporation | Degradable rubber member for downhole tools, degradable seal member, degradable protecting member, downhole tool, and method for well drilling |
| WO2015133544A1 (ja) * | 2014-03-07 | 2015-09-11 | 株式会社クレハ | 崩壊性のダウンホールツール用シール部材、ダウンホールツール、及び坑井掘削方法 |
| WO2015133545A1 (ja) * | 2014-03-07 | 2015-09-11 | 株式会社クレハ | ダウンホールツール用分解性ゴム部材、分解性シール部材、分解性保護部材、ダウンホールツール、及び坑井掘削方法 |
| CN110242244A (zh) * | 2014-03-07 | 2019-09-17 | 株式会社吴羽 | 钻井用堵塞器 |
| US9926764B2 (en) | 2014-03-11 | 2018-03-27 | Kureha Corporation | Molded product having effective thickness of 1 mm or more and containing aliphatic polyester resin, and downhole tool member for hydrocarbon resource recovery |
| JP2016060900A (ja) * | 2014-09-22 | 2016-04-25 | 株式会社クレハ | 反応性金属及び分解性樹脂組成物を含有する坑井掘削用組成物、坑井掘削用成形品、及び坑井掘削方法 |
| WO2016047502A1 (ja) * | 2014-09-22 | 2016-03-31 | 株式会社クレハ | 反応性金属を含有するダウンホールツール部材及び分解性樹脂組成物を含有するダウンホールツール部材を備えるダウンホールツール、並びに坑井掘削方法 |
| JP2016061127A (ja) * | 2014-09-22 | 2016-04-25 | 株式会社クレハ | 反応性金属を含有するダウンホールツール部材及び分解性樹脂組成物を含有するダウンホールツール部材を備えるダウンホールツール、並びに坑井掘削方法 |
| JP2016190901A (ja) * | 2015-03-30 | 2016-11-10 | 株式会社クレハ | ポリグリコール酸成形物、ダウンホールツール用部材、及び、ポリグリコール酸成形物の製造方法 |
| US9868231B2 (en) * | 2015-03-30 | 2018-01-16 | Kureha Corporation | Polyglycolic acid molded article, component for downhole tool, and method of producing polyglycolic acid molded article |
| US20160289374A1 (en) * | 2015-03-30 | 2016-10-06 | Kureha Corporation | Polyglycolic acid molded article, component for downhole tool, and method of producing polyglycolic acid molded article |
| WO2020013216A1 (ja) * | 2018-07-10 | 2020-01-16 | 株式会社クレハ | ダウンホールツールおよび坑井掘削方法 |
| GB2590023A (en) * | 2018-07-10 | 2021-06-16 | Kureha Corp | Downhole tool and well-drilling method |
| GB2590023B (en) * | 2018-07-10 | 2022-04-27 | Kureha Corp | Downhole tool and well-drilling method |
| US11428064B2 (en) | 2018-07-10 | 2022-08-30 | Kureha Corporation | Downhole tool and well-drilling method |
| WO2022075484A1 (ja) | 2020-10-09 | 2022-04-14 | 株式会社クレハ | プラグ、ダウンホールツールおよび坑井処理方法 |
| WO2023053955A1 (ja) | 2021-09-29 | 2023-04-06 | 株式会社クレハ | 樹脂組成物、ダウンホールツールまたはその部材、プラグおよび坑井処理方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20150096741A1 (en) | 2015-04-09 |
| US10030464B2 (en) | 2018-07-24 |
| CN106761546A (zh) | 2017-05-31 |
| EP2860344A4 (en) | 2016-01-06 |
| CN106761546B (zh) | 2020-05-08 |
| JPWO2013183363A1 (ja) | 2016-01-28 |
| US20160108696A1 (en) | 2016-04-21 |
| EP2860344A1 (en) | 2015-04-15 |
| CN104204404B (zh) | 2017-01-18 |
| AU2013272915A1 (en) | 2014-10-09 |
| US9267351B2 (en) | 2016-02-23 |
| CA2868975C (en) | 2017-02-14 |
| CN104204404A (zh) | 2014-12-10 |
| MX2014012613A (es) | 2015-01-19 |
| CA2868975A1 (en) | 2013-12-12 |
| EP3569815A1 (en) | 2019-11-20 |
| US20180298714A1 (en) | 2018-10-18 |
| US10626694B2 (en) | 2020-04-21 |
| AU2013272915B2 (en) | 2015-12-10 |
| JP6084609B2 (ja) | 2017-02-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6084609B2 (ja) | 炭化水素資源回収ダウンホールツール用部材 | |
| JP6117784B2 (ja) | 炭化水素資源回収ダウンホールツール用部材 | |
| JP6151255B2 (ja) | 炭化水素資源回収用ボールシーラーならびにその製造方法及びそれを用いる坑井の処理方法 | |
| JP6207529B2 (ja) | ポリ−l−乳酸固化押出成形物の応用及びポリ−l−乳酸固化押出成形物の製造方法 | |
| JP6363362B2 (ja) | 炭化水素資源回収用ダウンホールツール部材 | |
| JPWO2012050187A1 (ja) | 石油掘削補助用分散液 | |
| RU2588530C1 (ru) | Элемент для скважинного инструмента для извлечения углеводородных ресурсов | |
| CA3104631C (en) | Downhole tool and well-drilling method | |
| WO2022230962A1 (ja) | ダイバーティングエージェント及びこれを用いた坑井の亀裂の閉塞方法 | |
| CA3213356A1 (en) | Molded product and processed article | |
| JP2016083919A (ja) | ポリグリコール酸から形成される素形材または成形品の保管方法 | |
| JP2017155094A (ja) | 脂肪族ポリエステル樹脂成形物およびその製造方法 | |
| Labropoulos et al. | Modeling the Dynamic Rheological Bebavior of Agar Based Aqueous Binders | |
| JP2016186055A (ja) | ポリグリコール酸組成物および一時目止め材 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13801222 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2868975 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2013272915 Country of ref document: AU Date of ref document: 20130412 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2014/012613 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14395654 Country of ref document: US |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112014023400 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2014519870 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2014137001 Country of ref document: RU Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 112014023400 Country of ref document: BR Kind code of ref document: A2 Effective date: 20140922 |