WO2013112040A1 - Use of certain trioxygenated benzene derivatives in body fat management - Google Patents
Use of certain trioxygenated benzene derivatives in body fat management Download PDFInfo
- Publication number
- WO2013112040A1 WO2013112040A1 PCT/MY2012/000008 MY2012000008W WO2013112040A1 WO 2013112040 A1 WO2013112040 A1 WO 2013112040A1 MY 2012000008 W MY2012000008 W MY 2012000008W WO 2013112040 A1 WO2013112040 A1 WO 2013112040A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compounds
- plant
- range
- weight
- Prior art date
Links
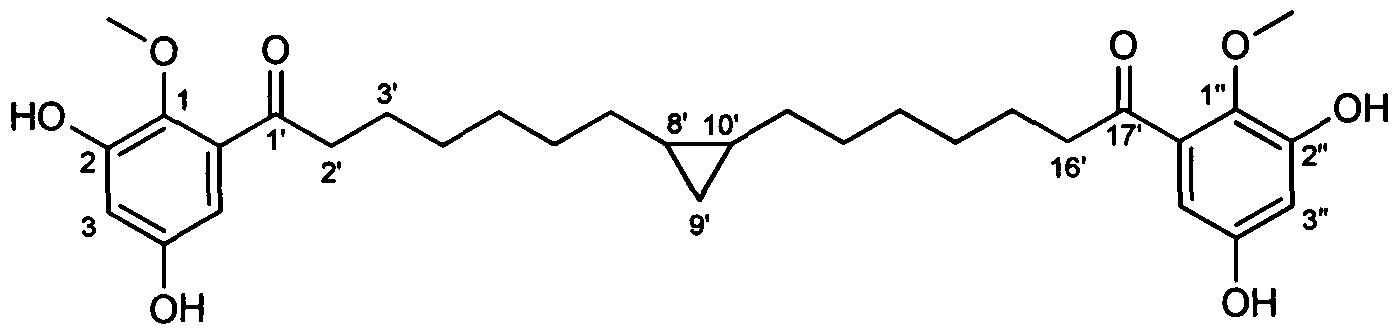
- RTLZZNDZZMKWBD-UHFFFAOYSA-N COc(c(O)cc(O)c1)c1C(CCCCCCC1C(CCCCCCC(c2cc(O)cc(O)c2OC)=O)C1)=O Chemical compound COc(c(O)cc(O)c1)c1C(CCCCCCC1C(CCCCCCC(c2cc(O)cc(O)c2OC)=O)C1)=O RTLZZNDZZMKWBD-UHFFFAOYSA-N 0.000 description 2
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/06—Preparations for care of the skin for countering cellulitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/347—Phenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/37—Esters of carboxylic acids
- A61K8/375—Esters of carboxylic acids the alcohol moiety containing more than one hydroxy group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9789—Magnoliopsida [dicotyledons]
Definitions
- the invention primarily relates to the use of certain trioxygenated benzene derivatives of formula (I) as defined herein or a corresponding plant extract as body fat modulating agents, preferably as anti-cellulite actives and/or for the reduction of the body fat content, in particular for reduction of body fat and/or for the management of obesity.
- the present invention also relates to corresponding methods and to certain novel compounds of formula (I).
- the invention further relates to specific plant extract formulations obtainable from a plant of the family Myrsinaceae, preferably from the genus Labisia, and to compositions, in particular cosmetic or pharmaceutical formulations, comprising an effective amount of one or more compounds of formula (I) or of a corresponding plant extract formulation.
- Weight control and body fat management today are a concern, and excessive fat deposits play an important role in various fields, such as obesity which may result in several health problems, or cellulite which is a cosmetic-aesthetic problem.
- Adipose tissue is a connective tissue that functions as a major storage site for body fat in the form of triglycerides. Adipose tissue can increase in size by increase in adipocyte size (hypertrophic growth) and/or by the increase in adipocyte number (hyperplastic growth).
- Fat metabolism in the fat tissue of humans in order to reduce the stored lipid quantity, can in principle be regulated by three Routes:
- Route (i) inhibition of the differentiation of preadipocytes.
- the differentiation of the precursor cells of the fat cells called preadipocytes to the real fat cells, called adipocytes, which may store triglycerides, can be inhibited.
- adipocytes which may store triglycerides
- Route (i) prevents the build up of cellulite in that the number of fat cells does not increase. This process of differentiation from preadipocytes to adipocytes is called adipogenesis.
- Route (ii) inhibition of the lipogenesis in adipocytes.
- the storage of triglycerides in the adipocytes also called lipogenesis
- an inhibition of Route (ii) prevents the storage of further triglycerides (fats) in the cell and existing fat cells do not store any new fat. Owing to the natural fat metabolism, when Route (ii) is inhibited, the fat content in the cell decreases.
- Route (iii) stimulation of lipoylsis in adipocytes.
- An augmented/increased hydrolysis of lipids already stored in the adipocytes - also called lipolysis - is possible by targeted stimulation.
- stimulation of Route (iii) increases the breakdown of the fats already present in the cell while an inhibiting, i.e. antagonistic effect with respect to Route (iii) on the other hand inhibits or prevents the breakdown of fat.
- the differentiation of cells is the changing of the control of the gene activity of a cell so that various protein stores are provided in the cells by means of transcription and protein biosynthesis and the cells differ according to appearance and function.
- adipocytes only express enzymes, which are necessary for the storing of fats, after differentiation.
- the undifferentiated preadipocytes these enzymes are not expressed or only to a very small extent.
- an important approach for diverence prevention may be to control the regulatory system of adipocytes proliferation and accumulation of triglyceride.
- adiposity body fat
- atherosclerosis where more than two of the preceding disorders are present, the condition is often called "metabolic syndrome”
- hyperlipidemia coronary heart disease, stroke, breast and colon cancer
- sleep apnoea gallbladder disease
- gastroesophageal reflux disease fatty liver, gout or thromboembolism.
- Obesity is one of the main factors in the development of cardiovascular diseases.
- the levels of cholesterol, blood pressure, blood sugar and uric acid in obese people are usually higher than those of persons of normal weight.
- the morbidity from coronary heart disease among the overweight people is increased as well.
- mortality will rise about 1% when body weight increases by 0.5 kg and the death rate will increase 74% when body weight exceeds 25% of the standard.
- the prevalence of obesity in the United States has more than doubled since the turn of the last century (whole population) and more than tripled within the last 30 years among children aged from 6 to 11.
- being overweight is considered by the majority of the Western population as unattractive. Determinants of obesity include social factors, psychological factors, genetic factors, developmental factors and decreased physical activity.
- WO 2004/082700 describes compositions comprising as essential constituent the extracts from four plants and an animal, and other plant extracts in the treatment of obesity. Only a combination of 14 extracts was examined in the example and shown to reduce the weight of mice by 6.4 to 9.2 %. Due to the large number of extracts administered, it is not possible to understand which of the extracts is responsible for the effects observed, in particular whether omission of one of more of these extracts would be possible without impairing the desired effects.
- WO 2004/096252 describes compositions comprising mushroom powder, extract, or a derivative thereof in combination with a liver protecting agent, wherein the mushrooms were selected on the basis of a hunger suppressing activity and fat burning capability.
- Adipostatin A (5-n-pentadecylresorcinol) is known as inhibitor of glycerol-3-phosphate dehydrogenase and for markedly inhibiting triglyceride accumulation in murine 3T3-L1 cells (see Journal of Antibiotics 1992, 886-891).
- 3T3-L1 cells a clone of Swiss/3T3 fibroblast, are transformed to adipose cells under certain culture conditions being accompanied by an increase of activities of many lipogenic enzymes. Therefore, adipocyte differentiation testing in murine 3T3-L1 cells can be regarded as a good model to study these effects.
- WO 2009/021622 A1 discloses the use of certain compounds or extracts comprising such compounds obtainable from plants from the genus from the group Schisandra, lllicium, Kadsura, Steganotaenia and Magnolia as active ingredient in the regulation of body weight and/or fat loss.
- Cellulite is also known under the synonyms protrusio cutis and colloquially as orange peel skin. It is a cosmetic-aesthetic problem which is accompanied by the formation of dimples and indentations of the skin and nodule formation of the subcutaneous fat tissue. Cellulite can occur on any part of the human body, but the outer side and the back of the thighs as well as the buttocks are most frequently affected. Breasts, lower stomach, upper arms or neck are also sometimes affected by cellulite. Cellulite may be regularly found on parts of the human body with excessive fat deposits, but overweight is not a prerequisite for its occurrence. Slim women increasingly also have pronounced cellulite symptoms. However, there is probably a correlation between the severity of the cellulite and the percentage of fat in the tissue.
- the gender-specific anatomic structure of the skin of human beings has a great influence on the development of cellulite.
- cellulite can only seldom be observed in men, while, on the other hand, about 80% - 90% of all women are affected, in particular Caucasian women.
- the structure of the dermis in particular, has an effect on the skin relief.
- the fat chambers in men when the skin is pressed together, are held back by intersecting connective tissue septa and the clamp-like enclosure of the fat cells connected therewith.
- women the fat chambers separated from one another in a tubular manner bulge up when being pressed together.
- the visible pattern of the cellulite is based on an increase in fat cushions in the subcutis and a reduction in the circulation conditions in the blood and lymph vessels.
- the cause is therefore partly a predisposed weakening of the connective tissue with simultaneous occurrence of enlarged fat cell chambers with stress, sports activity, smoking, pregnancies and female hormones (estrogen and progesterone) playing a part, in addition to genetic factors.
- Cellulitis is to be clearly separated and distinguished from the cosmetic phenomenon of cellulite.
- Cellulitis is a bacterial infection of the subcutaneous tissue, which in many cases may be a serious illness, and in contrast to cellulite, has to be treated therapeutically.
- the conventional treatment methods for cellulite attempt to encourage the blood circulation of the relevant skin parts and to positively influence the connective tissue structure, for example by massage, lymph drainage, diet, sport, magnetic fields or else liposuction (removing fat by suction).
- Cosmetic preparations which have the prophylaxis and treatment of cellulite as a goal have already been proposed in the literature. They mostly influence adipose tissue or adipocytes by a specific activity.
- EP 1 234 572 describes a cosmetic preparation of at least one isoflavone aglycone, in particular genistein and/or daidzein, for treating cellulite.
- the isoflavone aglycone is in this case combined with an algae extract.
- Genistein is described there as an active ingredient, which inhibits the multiplication of precursor fat cells and in addition the enzyme phosphodiesterase.
- isoflavones Preparations containing certain isoflavones are also described in DE 100 09 423, the isoflavones being described as materials with an anti-estrogen effect and used because of this effect. Daidzein, genistein, glycitein, formononetin and others are preferred isoflavones there.
- WO 2006/063714 teaches compositions for topical administration, containing a PDE3 inhibitor as active ingredient, for use in the treatment of cellulite and proposes pharmaceutical compositions comprising drugs like anagrelide, cilostazol, pimobendan, milrinone, amrinone, olprinone, enoximone, cilostamide, vesnarinone and trequinsin.
- the primary object of the present invention was to identify alternative active substances and compositions that can reduce the body fat content, and/or reduce the lipid quantity contained in fat tissue in mammals, preferably in human beings.
- Said substances and compositions should show a, preferably improved, activity regarding differentiation of preadipocytes and/or lipogenesis in adipocytes (cf. Route (i) and/or Route (ii) as described above). More particularly, said substances and compositions should be able to promote the loss of body fat and thus be useful in the treatment of obesity and to show activity with respect to the prophylaxis and treatment of cellulite. Additionally, said substances should preferably be naturally occurring compounds.
- the present invention relates to the use of a compound of formula (I) or a physiologically, preferably cosmetically, acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the physiologically acceptable salts thereof
- R 1 denotes hydrogen or an alkyl moiety having 1 to 4 carbon atoms
- R 2 denotes methyl or an acyl moiety having 1 to 4 carbon atoms
- each R 3 denotes hydrogen or both R 3 together denote an oxygen atom
- Y denotes an organic radical having 6 to 24 carbon atoms.
- the compounds of formula (I) and/or their salts as defined herein and certain extracts (as defined hereinafter) of plants or parts of plants of the family Myrsinaceae comprising one or more of said compounds of formula (I) and/or salts thereof can reduce the body fat content, and/or reduce the lipid quantity contained in fat tissue, and/or inhibit the differentiation of preadipocytes, and/or inhibit of the lipogenesis in adipocytes, and/or modulate the adipocyte differentiation.
- a cosmetic use or a cosmetic method is free of any therapeutic (side) effects.
- Cosmetic especially refers to a non-therapeutic use, that is, a use not for treatment of a disease or condition for its cure or symptom reduction but merely to improve the aesthetic appearance of an individual.
- a therapeutic or pharmaceutical use or method is considered as medical treatment, optionally with cosmetic (side) effects.
- cellulite is prevented, treated or reduced by a cosmetic composition containing one or more compounds of formula (I) by influencing the above described Routes (i) and/or (ii), most preferably by influencing the above described Routes (i) and (ii).
- the compounds of formula (I) and/or their salts as defined herein and certain extracts comprising one or more of said compounds of formula (I) or physiologically acceptable salts thereof are actives for the reduction of body fat, i.e. to achieve fat loss, and thereby thus are capable of allowing weight control in humans and thus the treatment of obesity. Further, they are cosmetic actives for the cosmetic applications mentioned herein, in particular for prevention, treatment and/or reduction of cellulite.
- the compounds of formula (I) may be used in the context of the present invention in all their isomeric forms, i.e. - depending on their structure - as enantiomers, diastereomers, syn-/anti-isomers, cis-/trans-isomers, epimers as well as (E)-/(Z)-isomers.
- the compounds of formula (I) can be used in the context of the present invention in the form of the pure stereoisomeric form or in the form of any mixture of stereoisomers.
- the compounds of formula (I) can also be used in the context of the present invention in the form of the pure enantiomers or in the form of any mixture of enantiomers, in the latter case racemates being preferred.
- extract either a direct extract (in liquid or preferably dried form), e.g. obtained as described below, or preferably a further enriched extract (obtainable e.g. by one or more further purification steps after extraction, e.g. chromatography, for example as described below) containing one or more, preferably two or more compounds of formula (I) is meant.
- administered or “administering” herein is meant administration of a prophylactically and/or therapeutically effective dose of a compound of formula (I) or a mixture of compounds of formula (I), or an extract comprising one or more the compounds of formula (I), to a human being in need of such treatment.
- effective amount or “effective dose” herein is meant an amount or a dose that produces the (therapeutic or cosmetic) effects for which it is administered, especially a reduction of weight, in particular (due to) the reduction of body fat.
- a "patient” or “subject” for the purposes of the present invention relates to mammals, especially human beings.
- the compounds of formula (I) or a mixture of compounds of formula (I), or a plant extract comprising one or more compounds of formula (I) are applicable to both humans and mammals.
- the patient is a human.
- the patients will be treated either in prophylactic or therapeutic intention, the latter e.g. to avoid regain in weight after a weight (especially body fat) reduction (e.g. to avoid the yo-yo effect), or to avoid weight gain (especially due to an increase in body fat) ab initio.
- dry refers to matter (such as an extract, a composition etc.) without water and without organic solvents, in particular being free of water and free of substances having a boiling point of less than 300°C at 1013 mbar.
- liquid and solid refer to the state of matter, e.g. a compound, carrier or composition, at 25°C and 1013 mbar.
- the terms “reduction of body fat” and/or “management of obesity” especially refer to the effects achieved by administration of one or more compounds of formula (I) or preferably an (especially further enriched) extract comprising one or more, preferably two or more, compounds of formula (I) either a lower weight gain can be observed or preferably a weight loss can be observed, in comparison to a control without such administration. More preferably, the weight loss is due to a reduction of the body fat.
- treatment of obesity is meant to either prophylactically avoid a weight gain in a mammal, preferably a human being, or preferably to reduce the body weight, especially to reduce the body fat in a mammal, preferably a human being.
- Salts of the compounds of formula (I) may be formed, for example, by reacting a compound of formula (I) with an amount of acid or base, such as an equivalent amount, in a medium such as one in which the salt precipitates or in an aqueous medium followed by lyophilization. Also ion exchangers can be used to form salts from free forms or free forms from salts of a compound of formula (I).
- a compound of formula (I) a mixture of compounds of formula (I) or one or more compounds of formula (I) are mentioned, this intends to include the free (enriched or substantially pure) form and/or one or more (especially pharmaceutically or nutraceutically salts (alone or together referred to as pharmaceutically acceptable salts hereinafter)) where salt-forming groups are present, tautomer(s) (where tautomerism, e.g. of the oxo/enol type, is possible), or mixtures of two or more of these specific forms.
- Preferred physiologically, preferably cosmetically, pharmaceutically or nutraceutically acceptable salts of compounds of formula (I) are those in which the one, several or preferably all counterions (counteracting cation) are selected from the group consisting of Na ⁇ K + , NH 4 + , trialkylammonium NHR' 3 ⁇ Ca 2+ , Mg 2+ , Zn 2+ and Al 3+ .
- each R' independently of the other radicals R' denotes an alkyl group having 1 to 30 C-atoms, preferably having 4 to 22 C-atoms.
- Particular preferred counterions in physiologically, preferably cosmetically, pharmaceutically or nutraceutically, acceptable salts of compounds of formula (I) are selected from the group consisting of Na + , K + , Ca 2+ and Mg 2+ and mixtures thereof.
- trioxygenated benzene derivatives corresponding to formula (I) are known per se, some of these compounds are described as occurring naturally.
- R z denotes methyl or n-propyl.
- the compounds of formula (I) are natural compounds, i.e. compounds that are present in and can be isolated or extracted from natural sources (especially from plant sources mentioned in detail hereinafter) without chemical synthesis steps (though they may also be prepared by chemical synthesis).
- Preferred compounds used in accordance to the present invention are compounds of formula (I) and/or the physiologically acceptable salts thereof, wherein
- R 1 denotes hydrogen, methyl or ethyl, and/or
- R 2 denotes methyl, formyl or acetyl
- Y denotes an organic radical having 8 to 22 carbon atoms.
- the compounds used in accordance to the present invention are compounds of formula (I) and/or the physiologically acceptable salts thereof, wherein
- R 1 denotes hydrogen or methyl
- R 2 denotes methyl or acetyl
- Y denotes an organic radical having 8 to 22 carbon atoms and consists of carbon, hydrogen and oxygen, wherein Y contains 0, 1 , 2, 3, 4 or 5 oxygen atoms.
- the compounds used in accordance to the present invention are compounds of formula (I) and/or the physiologically acceptable salts thereof, wherein
- Y denotes a n-alkyl radical having 8 to 12 carbon atoms
- Y denotes an n-alkenyl radical having 8 to 12 carbon atoms and 1 , 2 or 3 carbon-carbon double bonds, or
- Y denotes an organic radical having 17 to 21 carbon atoms consisting of carbon, hydrogen and 3 or 4 oxygen atoms. Due to their higher activity regarding the effects to be achieved in the context of the present invention, particularly preferred compounds of formula (I) and/or the physiologically acceptable salts thereof are selected from the group consisting of:
- Kacip Fatimah (Labisia pumila - which is synonymous to Labisia pothonia or Ardisia pumila) has been used by many generations of the Malay women to induce and facilitate childbirth as well as a post-partum medicine.
- the decoction of the leaves and roots is valued to help delay conception and regain strength after childbirth.
- the decoction may also be given 1-2 months before childbirth.
- Modern perceptions of its health benefits include being a useful herb to contract the uterus after childbirth; firm and tone abdominal muscles; tighten the vaginal wall and tissue; allay painful or difficult menstruation, cramping, and irregular periods; and to generally alleviate fatigue and promote emotional well-being.
- Kacip Fatimah used in traditional Malay medicine may also be referred to as Selusoh fatimah, Rumpur siti fatimah, Akar fatimah, Kacit fatimah, Kachip Fatima, Kachip patima, Kunchi fatimah, Pokok pinggang, Rumput palis, Tadah matahari, Mata pelanduk rimba (Malay), Bunga belangkas hutan or Sangkoh (Iban).
- Antioxidant components found in Labisia pumila include ascorbic acid (vitamin C), beta carotene, anthocyanins, and certain flavonoids (J. Med. Plants Res. 2009; 3(4):217-223).
- the genus Labisia belongs to the family Myrsinaceae and contains several species.
- the species Labisia pumila is further subdivided into several varieties (var. pumila, var. alata, var. discoplacenta, var. lanceolata, var. malintangensis, var. neriifolia, var. sessilifolia).
- L. pumila var. alata seems to be the most commonly used in traditional medicine.
- Labisia pumila is distributed throughout South-East Asia and is found in throughout Malaysia, Sumatra, Java, Borneo, Thailand, Cambodia, Indonesia, etc. The plants can be found in the undergrowth of tropical forests. It can be cultivated in shaded places on moist sandy loam soil, with liberal application of organic matter and fertilizers.
- Labisia pumila is a small herbaceous shrub.
- the root is tough and woody with long primary roots and few secondary roots. There are few leaves pointing upwards.
- the whole leaf is about 5-35 cm long and 2-8 cm wide and finely toothed with numerous veins. It is of a dark green color on the adaxial and lighter green on the abaxial side.
- Labisia pumila is preferred, in particular Labisia pumila var. alata, Labisia pumila var. lanceolate, and Labisia pumila var. pumila.
- J. Pharm. Pharmacol. 1999, 51 (Supplement): 236 discloses that the ethanolic extracts showed different profiles in thin layer chromatography between leaves and roots and between the two varieties Labisia pumila var. alata and Labisia pumila var. pumila. Additionally, two benzoquinoid compounds from Labisia pumila var. alata leaves and roots were identified.
- WO 2009/064155 and WO 2009/064156 describe a water-soluble extract obtained by extraction of extraction of Labisia pumila plant material with water, in particular extracts obtained from dried leaves of Labisia pumila and distilled water in a ratio of 1 : 8, as having the capability to develop a pharmaceutical composition for hepatoprotective or immunostimulating activity.
- US 2007/0082069 A1 and US 2010/0311180 A1 relate to processes for preparing a water- soluble extract by extraction of one part of dried Labisia pumila plant material with 6 parts of water, said extract having a reproducible chemical profile by reverse-phase HPLC, using 3,4,5-trihydroxybenzoic acid as marker substance.
- WO 2010/147449 A1 describes a process for extracting the leaves of Labisia pumila with water into a free flowing powder. Further, WO 2010/147449 A1 describes that an aqueous leaf extract of Labisia pumila resulted in an increase of the body weight of mice after 14 days.
- KR 2009/0113723 A relates to compositions containing an extract or a fermentation product of Labisia pumila providing activity of UV protection, whitening, anti-aging and anti-oxidation activity and improving skin function.
- the Labisia pumila extract according to KR 2009/0113723 A is obtained using as organic solvent ethanol, hexane, or acetone.
- the cosmetic composition may be in the form of soap, lotion, cream, pack and cosmetic liquid.
- WO 2009/116848 A1 discloses a standardized aqueous extract of Labisia pumila that can reduce the risk or progression of cardiovascular diseases, said extract is described as being capable to influence pathophysiological changes, including regulation of various related genes, associated with insulin resistance, obesity, metabolic syndrome, diabetes and aging.
- Zulfiqar et al. isolated certain alkyl phenols and saponins from a methanolic extract from the powdered roots of Labisia pumila (Phytochemistry 2011 , doi:10.1016/j.phytochem.2011.06.014, article in press). Some of the isolated compounds showed moderate in vitro antibacterial activity.
- the total phenolic content and the total flavanoid content in extracts obtained by extraction of Labisia pumila leaves with 100% methanol, 100% water, 60% methanol and 100% acetonitrile was determined by Chua et al., Food Chemistry 127 (2011) 1186-1192.
- flavonoids and phenolic acids were detected by LC/MS methodology (e.g. flavonols like quercetin, flavanols like catechin and epigallocatechin, benzoic acids like gallic acid, vanillic acid or salicylic acid, and cinnamic acids, e.g. coumaric acid and caffeic acid.
- PCOS polycystic ovary syndrome
- a plant extract preferably an enriched plant extract, according to the present invention comprises an effective amount of one or more compounds of formula (I) as defined herein and/or a physiologically acceptable salt thereof, preferably in a total amount in the range from 0.1 to 40 wt.%, more preferably from 0.2 to 20 wt.%, particularly preferably from 0.5 to 15 wt.%, most preferably from 1 to 8 wt.%, most preferably from 1.5 to 6 wt.%, based on the total weight of the plant extract.
- the present invention relates to a corresponding use or method, wherein one or more compounds of formula (I) and/or physiologically acceptable salts thereof as defined above, preferably as defined in any of the above mentioned preferred embodiments, wherein these are used as part or in the form of a plant extract, preferably a plant extract from a plant of the family Myrsinaceae, more preferably from the genus Labisia, more preferably from Labisia pumila.
- the total weight share of one or more compounds of formula (I) in an extract used according to the invention is in the range from 0.1 to 25 % by weight, more preferably from 0.25 to 15 %, particularly preferably from 1 to 10 %, most preferably from 1.25 to 5 %, based on the total dry weight of the extract.
- the extracts or compounds according to the invention may be used as such, in the form or pharmaceutical or nutraceutical formulations (the latter term including food additives) or in the form of functional food.
- the compounds or mixture of compounds of formula (I), especially extracts comprising one or more compounds of formula (I), are used as supplement, this means that the compound(s), extract or a pharmaceutical or nutraceutical formulation comprising it or them can be added to any other nutrient or pharmaceutical or nutraceutical, preferably other than (exclude especially mixtures known). Thus they can especially serve as food supplement. However, the compound(s), extract or formulations may also be administered as such.
- “Nutraceuticals”, “Functional Food”, or “Functional Food products” (sometimes also called “Foodsceuticals”, “Medicinal Food” or “Designer Food”) for use according to the present invention are defined as food products (including beverages) suitable for human consumption - the expression comprises any fresh or processed food having a health- promoting and/or disease-preventing property beyond the basic nutritional function of supplying nutrients, including food made from functional food ingredients or fortified with health-promoting additives, especially with effects in the prophylaxis or treatment of obesity, especially allowing for body weight reduction and/or body weight maintenance, appetite suppression, the provision of satiety or similar changes in metabolism, and in which an extract, a compound or compound mixture of compounds of formula (I), respectively, according to the invention is used as an ingredient (especially additive) as health benefit agent, especially in an effective amount.
- the functional food products or pharmaceutical products may be manufactured according to any suitable process, preferably comprising extraction of one or more compounds of formula (I) and admixing to a functional food product or at least one physiologically, preferably cosmetically, nutraceutically or pharmaceutically, acceptable carrier.
- a functional food or a pharmaceutical or nutraceutical formulation comprising a compound, more preferably a compound mixture, for use according to the present invention, can be obtained by
- drying e.g. freeze-drying, spray-drying and evaporation
- granulation e.g. to syrups, formed via concentration and/or with the aid of thickeners
- concentrating e.g. to syrups, formed via concentration and/or with the aid of thickeners
- pasteurizing sterilizing, freezing, dissolving, dispersing, filtering, centrifuging, confectioning, and the like.
- the functional food products according to the invention may be of any food type. They may comprise one or more common food ingredients in addition to the food product, such as flavours, fragrances, sugars, fruit, minerals, vitamins, stabilizers, thickeners, dietary fibers, protein, amino acids or the like in appropriate amounts, or mixtures of two or more thereof, in accordance with the desired type of food product.
- Examples of basic food products and thus of functional food products according to the invention are fruit or juice products, such as orange and grapefruit, tropical fruits, banana, apple, peach, blackberry, cranberry, plum, prune, apricot, cherry, peer, strawberry, marionberry, black currant, red currant, tomato, vegetable, e.g. carrot, or blueberry juice, soy-based beverages, or concentrates thereof, respectively; lemonades; extracts, e.g.
- dairy type products such as milk, dairy spreads, quark, cheese, cream cheese, custards, puddings, mousses, milk type drinks and yoghurt
- frozen confectionary products such as ice-cream, frozen yoghurt, sorbet, ice milk, frozen custard, water-ices, granitas and frozen fruit purees
- baked goods such as bread, cakes, biscuits, cookies or crackers
- spreads e.g. margarine, butter, peanut butter honey
- snacks e.g.
- meat fish or fish or meat products such as sausages, burgers, meat loafs, meatballs, meat extracts, canned or tinned fish or meat, meat vol-au-vent, meat or fish soup, meat or fish skewers, fish fingers; or the like.
- One or more other customary additives may be present, such as flavour, fragrances or other additives, such as one or more selected from stabilizers, e.g. thickeners; colouring agents, such as edible pigments or food dyes; bulking agents, such as fruit pulp, e.g.
- polyols such as xylitol, mannitol, maltitol or the like
- preservatives such as sodium or potassium benzoate, sodium or calcium carbonate or other food grade preservatives
- antioxidants such as ascorbic acid, carotinoids, tocopherols or polyphenols
- mono-, oligo- or polysaccharides such as glucose, fructose, sucrose, soy- oligosaccharides, xylo-oligosaccharides, galacto-oligosacharides
- other artificial or natural non- or low-caloric sweeteners such as aspartame or acesulfame
- bitterness blockers acidifiers in the form of edible acids, such as citric acids, acetic acid, lactic acid, adipic acid; flavours, e.g.
- artificial or natural e.g. botanical flavours
- emulsifiers e.g. thiols, e.g. allylic thiols
- diluents e.g. maltodextrose
- wetting agents e.g. glycerol
- stabilizers coatings
- isotonic agents absorption promoting or delaying agents; and/or the like.
- the one or more compounds of formula (I) or compound mixtures thereof according to the invention can also be comprised in confectioned formulations to be added to foods including beverages, e.g. in the form of powders or granules, e.g. freeze-dried or spray- dried, concentrates, solutions, dispersions or other instant form, or the like.
- the present invention relates to a composition, preferably a topically or orally administrable cosmetic or pharmaceutical composition, comprising
- an effective amount of (the preferred) compounds of formula (I) relates to a total amount of one, two or more compounds, preferably of the preferred compounds, of formula (I) sufficient to exhibit an activity in the above described Routes (i) and (ii) i.e. to influence one or more of said Routes in the desired way in the sense of the present invention.
- a preferred composition according to the present invention comprises one or more compounds selected from the following groups (b) and/or (c):
- the group of stimulators of beta-oxidation preferably L-carnitine.
- the lipolysis stimulant is present in a quantity sufficient to stimulate lipolysis.
- the one or more stimulators of the transport or oxidation of free fatty acids are present in a quantity sufficient to stimulate the transport or oxidation of free fatty acids.
- An advantageous preparation according to the invention additionally contains anti-cellulite active ingredients from group (b-i) of inhibitors of phosphodiesterase selected from the group of xanthines, preferably those described in US 2010/0285153 A1.
- Preferred xanthines are methyl xanthines, preferably selected from the group consisting of caffeine, theobromine and theophylline; the most preferred methyl xanthine in the sense of the present invention is caffeine.
- An alternatively preferred active ingredient from group (b-i) is aminophylline, a theophylline derivative.
- the xanthines or methyl xanthines may preferably be used as pure materials or in the form of plant extracts.
- preferred compositions contain combinations of the compound of formula (I) with an agonist of beta-adrenergic receptors of adipocytes.
- Preferred agonists of beta-adrenergic receptors are [beta]-phenylethylamines can preferably be used as pure substances, in the form of their respective hydrochlorides or in the form of plant extracts.
- Preferred agonists of beta-adrenergic receptors are adrenaline, noradrenaline, metanephrine, macromerine, normacromerine, hordenine, N-methyltyramine, dopamine, octopamine, tyramine, 2-phenylethylamine, phenylethanolamine, epinine (N- methyldopamine), synephrine, ephedrine, pseudoephedrine, norephedrine and isoprenaline.
- synephrine-containing extracts such as, for example, orange blossom extract.
- the invention in one aspect of the present invention thus relates to (improved), preferably cosmetic, compositions comprising (b) one or more lipolysis stimulants, wherein one, a plurality or all of the lipolysis stimulants of component (b) are preferably selected from the group consisting of
- (b-i) xanthines preferably selected from the group consisting of caffeine, theobromine and theophylline, and
- (b-ii) [beta]-phenylethylamines preferably selected from the group consisting of adrenaline, noradrenaline, metanephrine, macromerine, normacromerine, hordenine, N- methyltyramine, dopamine, octopamine, tyramine, 2-phenylethylamine, phenylethanolamine, epinine (N-methyldopamine), synephrine, ephedrine, pseudoephedrine, norephedrine and isoprenaline.
- the total quantity of compounds of formula (I) is in the range of from 0.01 - 10 % by weight, preferably in the range of from 0.025 - 5 % by weight and particularly preferably in the range of from 0.05 - 3 % by weight and most preferably in the range of from 0.1 - 2 % by weight
- the total quantity of phosphodiesterase inhibitors (b-i) is in the range of from 0.005 - 10 % by weight, preferably in the range of from 0.05 - 5 % by weight and more preferably in the range of from 0.5 - 2.5 % by weight
- the total quantity of agonists of beta-adrenergic receptors (b-ii) is in the range of from 0.0001 - 0.10 % by weight, preferably in the range of from 0.001 - 0.05 % by weight and more preferably in the range of from 0.002 - 0.02 % by weight, in
- compositions can be prepared in various forms, such as granules, tablets, pills, pellets, syrups, solutions, dispersions, suppositories, capsules, suspensions, salves, lotions and the like.
- Pharmaceutical grade or food grade organic or inorganic carriers and/or diluents suitable for oral and topical use can be used to formulate compositions containing the therapeutically-active compounds.
- Diluents known in the art include aqueous media, vegetable and animal oils and fats.
- compositions may also include one or more of the following: carrier proteins such as serum albumin; buffers; fillers such as microcrystalline cellulose, lactose, corn and other starches; binding agents; sweeteners and other flavouring or fragrancing agents; colouring agents; and polyethylene glycol.
- carrier proteins such as serum albumin
- buffers such as microcrystalline cellulose, lactose, corn and other starches
- binding agents such as microcrystalline cellulose, lactose
- the compounds of formula (I) or a mixture of compounds of formula (I) or extracts comprising one or more compounds of formula (I) may be administered alone or in combination with other active agents, preferably on or more other anti-obesity agents.
- anti-obesity agents that may be combined with one or more compounds of formula (I) or the salts thereof, antilipidemics, e.g.
- atorvastatin cerivastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, simvastatin, anti-obesity drugs, such as suppressants of the appetite, stimulators of the body's metabolism, or drugs or compositions interfering with the body's ability to absorb specific nutrients, such as sibutramine, diethylpropion, phendimetrazine, phentermine, fenfluramine, lipase inhibitors, such as orlistat; anorectics, such as dexedrine; cannabinoid receptor antagonists, such as rimonabant; acarbose; or the like, can be mentioned.
- specific nutrients such as sibutramine, diethylpropion, phendimetrazine, phentermine, fenfluramine, lipase inhibitors, such as orlistat
- anorectics such as dexedrine
- cannabinoid receptor antagonists
- Other helpful drugs or active agents may be administered, e.g. psychoactive agents, agents that help in the treatment of addictive behaviour, e.g. nicotine addiction, or the like, especially in so far as they help to support the prophylaxis or treatment according to the invention intended.
- psychoactive agents agents that help in the treatment of addictive behaviour, e.g. nicotine addiction, or the like, especially in so far as they help to support the prophylaxis or treatment according to the invention intended.
- Weight loss diets such as food combining, Hay diet, Atkins diet (low-carbohydrate diet), cabbage soup diet, diabetic diet, fat resistance diet, slimming world diet, low-fat diet, Pritkin diet, low-carbohydrate diet, low protein diet, negative calorie diet, raw food diet, weight watchers diet are possible examples of appropriate diets.
- the compounds of formula (I) or a mixture of compounds of formula (I) or an extract comprising one or more compounds of formula (I) of the present invention are particularly useful for controlling the body weight, preferably the treatment of obesity, adiposity, and/or further immune or inflammatory disorders.
- Natural compounds of formula (I) or extracts comprising one or more of natural compounds of formula (I), for use according to the present invention are isolated from the plant or from parts of plants, preferably the roots, of Labisia pumila.
- the extracts used according to the invention may be obtained by using solvents of a certain polarity capable to extract one or more compounds of formula (I) from plant material.
- the extraction leading to the extract can, for example, start from whole plants or plant parts (collectively referred to also a plant material) as defined above, e.g. from leaves or roots.
- the plant material may be used without prior treatment or after e.g. drying, chopping, milling or grinding or combinations of two or more thereof.
- the extract may be prepared by any extraction method known in the art, however, the condition being that it is capable to extract one or more naturally occurring compounds of formula (I).
- supercritical fluids such as nitrous oxide, sulphur dioxide, fluorocarbons or especially carbon dioxide, with or without one or more organic modifiers, e.g. co- solvents such as methanol or ethanol, halogenated hydrocarbons such as chloroform, which allow adjusting the solvating power
- the extraction can be followed by a further step for enrichment, e.g. solvent partition (e.g. of an extract filled up with water and partitioned between a hydrophilic phase, e.g.
- a hydrophobic phase e.g. an essentially apolar solvent forming a separate phase in the presence of water, e.g. an alkane, such as pentanes or hexanes, or an only weekly polar solvent, such as an ester, e.g. ethyl acetate, an ether, e.g. ethyl ether, or a halogenated hydrocarbons, such as methylene chloride, and/or by chromatography, e.g. preparative high performance chromatography.
- water e.g. an alkane, such as pentanes or hexanes
- an only weekly polar solvent such as an ester, e.g. ethyl acetate, an ether, e.g. ethyl ether, or a halogenated hydrocarbons, such as methylene chloride, and/or by chromatography, e.g. preparative high performance chromatography.
- the extract is further extracted after optional concentration e.g. by evaporation extracted with a solvent not miscible with water to remove impurities into the organic phase.
- reprocessing is possible, e.g. by filtering (e.g. through paper, sintered glass, charcoal (also allowing for decoloration) or silica).
- the extraction can be made at lowered or elevated or ambient temperature, e.g. in the range from -20 °C to the boiling point of the solvent or solvent mixture employed, e.g. from 20 °C to said boiling point.
- the extraction may be improved by moving the solvent and/or the plant material, e.g. by stirring, or by ultrasound, or by milling and/or chopping during extraction, or the like.
- Plant extracts for use in accordance with the present invention can be prepared using plants or especially plant parts, in particular roots, of Labisia pumila.
- Plant parts are, e.g. leaves, bark, flowers, buds, fruits, stems, shoots, roots or other parts of plants, and they or the plants can be complete, hackled, crushed, chopped, broken up, homogenized, dried, fermented or treated otherwise.
- the compounds of formula (I) or a mixture of compounds of formula (I) or an extract comprising one or more compounds of formula (I) of the present invention can be prepared by extracting and preferably enriching up to isolating them from the plants or parts of the plants.
- Auxiliary means such as (especially ultrasonic) sonication, temperature/heating (preferably at temperatures in the range from 20°C to 120 °C), stirring, re-extraction, evaporation or the like, may be used to allow for appropriate extraction, enrichment and purification.
- Extraction preferably takes place with a polar solvent or solvent mixture, e.g. water and/or an alcohol, such as ethanol, and/or with a liquid or superfluid gas, especially superfluid C0 2 .
- a polar solvent or solvent mixture e.g. water and/or an alcohol, such as ethanol, and/or with a liquid or superfluid gas, especially superfluid C0 2 .
- a further embodiment of the invention relates to the use of a plant extract comprising one ore more compounds of formula (I) and/or physiologically acceptable salts thereof (as defined above), wherein the solvent for extraction is an aqueous solvent; yet another specific embodiment of this embodiment is where the solvent used for extraction essentially consists or consist of a mixture of water and a Ci-C 8 -alkanol with one or more hydroxy groups, preferably a C C 8 -alkanol, a (VCe-alkandiol or a Ci-C 8 -alkantriol, such as ethanol/water, isopropanol/water, monopropylene glycol/water or glycerol/water, wherein the total vol/vol ratio (v/v) of said alkanol or alkanols with one or more hydroxy groups to water is in the range from 3 : 97 to 98 : 2, preferably from 25 : 75 to 96 : 4.
- the extraction of the plant or a part of a plant preferably is carried out with a mixture of water and an alcohol having 1 to 3 carbon atoms, preferably with a mixture of ethanol and water, wherein the volume ratio (v/v) of said alcohol : water in the range of 1 : 2 to 30 : 1 , more preferably in the range of 1 : 1 to 25 : 1 , most preferably in the range of 3 : 2 to 20 : 1 , preferably at a temperature below 75 °C, more preferably in the range from 15 to 60 °C, even preferably in the range from 20 to 50 °C.
- the weight ratio of (preferably dried) plant material, preferably roots, to the total amount of aqueous solvent used in the extraction preferably is in the range from 2 : 1 to 1 : 4, more preferably in the range from 1 : 1 to 1 : 3, even more preferably in the range from 1 : 1 to 1 : 2.
- the following method for producing a preferred plant extract for use in accordance with the present invention comprises the following steps: providing one or more roots of a plant or a part of a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, and optionally drying the plant or a part of a plant of step (i), extraction of said plant or a part of a plant provided in step (i) with a mixture of water and an alcohol having 1 to 3 carbon atoms, preferably with a mixture of ethanol and water, wherein the volume ratio (v/v) of said alcohol : water in the range of 1 : 2 to 30 : 1 , more preferably in the range of 1 : 1 to 25 : 1 , most preferably in the range of 3 : 2 to 20 : 1 , preferably at a temperature below 75 °C, more preferably in the range from 15 to 60 °C, even preferably in the range from 20 to 50 °C, optionally partially or fully
- the extracts can subsequently be further enriched by one or more additional purification steps, such as distribution, precipitation (e.g. crystallization) or especially chromatography, by which it is possible to obtain further enriched extracts or isolated compounds of formula (I).
- additional purification steps such as distribution, precipitation (e.g. crystallization) or especially chromatography, by which it is possible to obtain further enriched extracts or isolated compounds of formula (I).
- the present invention relates to a plant extract formulation, preferably in solid form, obtainable from a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, particularly preferably from the roots of Labisia pumila, comprising
- solid carrier substances preferably one or more solid carrier substances selected from the group consisting of maltodextrins, silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stearate salts, preferably magnesium stearate, alginates, tragacanth, gelatins, calcium silicates, cellulose and cellulose derivatives, preferably microcrystalline cellulose or methyl cellulose, polyvinylpyrrolidones, and propylhydroxybenzoates.
- maltodextrins silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stearate salts, preferably magnesium stearate, alginates, tragacanth, gelatins, calcium silicates, cellulose and cellulose derivatives, preferably micro
- the present invention relates to a method for producing a plant extract formulation according to the present invention, comprising the following steps: (i) providing a plant or a part of a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, particularly preferably from the roots of Labisia pumila,
- step (i-a) optionally drying the plant or a part of a plant of step (i), (ii) extraction of said plant or a part of a plant provided in step (i) or (i-a) with a mixture of water and an alcohol having 1 to 3 carbon atoms, preferably with a mixture of ethanol and water, wherein the volume ratio (v/v) of said alcohol : water in the range of 1 : 2 to 30 : 1 , more preferably in the range of 1 : 1 to 25 : 1 , most preferably in the range of 3 : 2 to 20 : 1 ,
- step (iii) mixing the extract obtained in step (ii) or (ii-a) with one or more solid carrier substances, preferably one or more solid carrier substances selected from the group consisting of maltodextrins, silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stearate salts, preferably magnesium stearate, alginates, tragacanth, gelatins, calcium silicates, cellulose and cellulose derivatives, preferably microcrystalline cellulose or methyl cellulose, polyvinylpyrrolidones, and propylhydroxybenzoates,
- one or more solid carrier substances selected from the group consisting of maltodextrins, silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stea
- step (iv) optionally drying the mixture obtained in step (iii), preferably by spray-drying or freeze-drying.
- a mixture of compounds of formula (I), plant extract, plant extract formulation or (cosmetic or pharmaceutical) composition comprises the three substances of formula (I) (as defined above)
- a pharmaceutical or nutraceutical composition comprising a compound of formula (I), or a mixture of compounds of formula (I), or especially a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient together with a pharmaceutically acceptable diluent or carrier, especially for use in the therapeutic and/or prophylactic treatment mentioned under (1).
- a pharmaceutical or nutraceutical composition for the treatment as mentioned under (1) comprising a compound of formula (I), or a mixture of compounds of formula (I), or especially a (preferably further enriched) extract comprising one or more compounds of formula (I), and a pharmaceutically acceptable diluent or carrier, as active ingredient supplement to a food.
- a functional food comprising a compound of formula (I), or a mixture of compounds of formula (I), or especially a (preferably further enriched) extract, as active ingredient for the treatment as mentioned under (1 ).
- a method for the treatment as mentioned under (1), especially any one or more of obesity, and/or excess body fat, in a subject in need of such treatment comprising administering a pharmaceutically or nutraceutically effective amount of a compound of formula (I), a mixture of compounds of formula (I), or a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient, especially to an individual in need thereof.
- a method or use as defined under (4) comprising co-administration, e.g. concomitantly or in sequence, of a therapeutically effective amount of compound of formula (I), or a mixture of compounds of formula (I), or a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient and a different pharmaceutically active compound and/or a pharmaceutically acceptable salt thereof, said different pharmaceutically active compound and/or salt thereof being especially for use in the treatment as mentioned under (1).
- a combination product comprising a therapeutically effective amount of a compound of formula (I), or a mixture of compounds of formula (I), or a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient, and a different pharmaceutically active compound and/or a pharmaceutically acceptable salt thereof, said second pharmaceutically active compound being especially for use or of use in the treatment mentioned under (1).
- the use or method is such that the compound(s) of formula (I) and/or salts thereof (as defined herein) or mixtures thereof are the active ingredient, i.e.
- the prophylactic and/or therapeutic treatment or regulation of body weight and/or fat loss and/or management of obesity, especially decrease (reduction) of body weight, more especially decrease (reduction) of body fat, are especially preferred embodiments according to the invention.
- the pharmaceutical or nutraceutical preparations may be sterilized and/or may contain carrier materials or adjuvants such as preservatives, stabilizers, binders, disintegrants, wetting agents, skin or mucuous membrane penetration enhancers, emulsifiers, salts for varying the osmotic pressure and/or buffers, or other ingredients, excipients or carrier materials known in the art.
- carrier materials or adjuvants such as preservatives, stabilizers, binders, disintegrants, wetting agents, skin or mucuous membrane penetration enhancers, emulsifiers, salts for varying the osmotic pressure and/or buffers, or other ingredients, excipients or carrier materials known in the art.
- the compounds can be administered by a variety of routes including oral, rectal, topical, transdermal, subcutaneous, intravenous, intramuscular, and intranasal, preferably they are administered orally or topically.
- Another embodiment of the present invention is a pharmaceutical formulation comprising an effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable carrier, diluent or excipient therefor.
- physiologically, preferably cosmetically, pharmaceutically and/or nutraceutically, acceptable it is meant that the carrier, diluent or excipient is compatible with the other ingredients of the formulation and not be deleterious to the recipient thereof.
- the active ingredient will usually be admixed with a carrier, or diluted by a carrier, or enclosed within a carrier which may be in the form of a capsule, sachet, paper or other container.
- a carrier which may be in the form of a capsule, sachet, paper or other container.
- the carrier serves as a diluent, it may be a solid, semi-solid or liquid material which acts as a vehicle, excipient or medium for the active ingredient.
- compositions according to the present invention can be in the form of tablets, pills, powders, lozenges, sachets, cachets, elixirs, suspensions, emulsions, solutions, syrups, aerosols, (as a solid or in a liquid medium), ointments containing, for example, up to 10% by weight of the active compound, soft and hard gelatin capsules, suppositories, sterile injectable solutions, sterile packaged powders, and the like.
- Suitable carriers, excipients, and diluents are lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, alginates, tragacanth, gelatin, calcium silicate, microcrystalline cellulose, polyvinylpyrrolidone, cellulose, water syrup, methyl cellulose, methylhydroxybenzoates, propylhydroxybenzoates, talc, magnesium stearate and mineral oil.
- the formulations may additionally include lubricating agents, wetting agents, sweetening agents, flavoring agents, and the like.
- the compositions of the invention may be formulated so as to provide quick, sustained or delayed release of the active ingredient after administration to the patient by employing procedures well known in the art.
- compositions are preferably formulated in a unit dosage from, each dosage generally containing form 0.1 to 500 mg, and preferably from 0.25 to 300 mg, more preferably from 0.5 to 200 mg of the compound of formula (I).
- unit dosage form refers to physically discrete units suitable as unitary dosages for human subjects and other mammals, each unit containing a predetermined quantity of active material calculated to produce the desired therapeutic effect, in association with a suitable pharmaceutical carrier.
- the effects of the compounds of formula (I) and/or their salts may be explained by a modulation of adipocyte triglyceride accumulation and in particular have an inhibitory effect on adipocyte differentiation. Therefore the extracts of the invention are useful in the modulation of triglyceride incorporation in adipocytes and thus can be used for modulation of body fat content in mammals (including humans) including the skin of said mammals.
- the invention relates to the use of an extract from Labisia pumila for reducing the body fat composition of a mammal (preferably a human), comprising modulation of adipocyte differentiation by applying said composition to individuals orally and/or topically.
- the invention relates to the use of one or more compounds of formula (I) (as defined above) in the reduction of body fat content, thereby resulting in body weight management, in particular body weight reduction.
- the invention relates to the use of one or more compounds of formula (I) and/or the physiologically acceptable salts thereof (as defined above) - based on the modulation of adipocyte differentiation - for skin slimming, in particular for dermal slimming, and/or for improving the appearance of skin.
- a further aspect of the invention relates to the use of one or more compounds of formula (I) and/or the physiologically acceptable salts thereof (as defined above), where the improving of the cosmetic appearance is a more juvenile appearance of the skin.
- the compounds of formula (I) can easily be incorporated in these concentrations in common cosmetic or dermatological formulations such as pump sprays, aerosol sprays, creams, ointments, tinctures, lotions and the like.
- the dermatologic composition is an oil, an ointments, a tincture, a cream, a gel, a solution, a lotion, a spray, a powder, a shampoo, a soap, a cosmetic pencil or a perfume.
- a more specific embodiment here relates to the case where the dermatologic composition is an ointment, a cream, a gel or a solution.
- the "body fat content” can preferably be determined with a body fat analysis (BFA) using dual-energy X-ray absorptiometry (DEXA) using a four-compartment model that divides the body into water, bone mineral mass, fat tissue mass and residual fat-free soft (lean) mass).
- BFA body fat analysis
- DEXA dual-energy X-ray absorptiometry
- a four-compartment model that divides the body into water, bone mineral mass, fat tissue mass and residual fat-free soft (lean) mass.
- X-rays of two different energies are used to scan the body, one of which is absorbed more strongly by fat than the other.
- a computer can subtract one image from the other, and the difference indicates the amount of fat relative to other tissues at each point. A sum over the entire image enables calculation of the overall body composition.
- the GE Lunar Piximus2 DEXA scanner gives accurate information on differences in body composition.
- One scanning procedure takes up to five minutes per individual and provides data on fat mass, lean mass, bone mineral content and bone mineral density.
- the amount of subcutaneous fat in a subject can be measured using any method available for the measurement of subcutaneous fat. Methods of measuring subcutaneous fat are known in the art, for example, those described in US 6,530,886, incorporated herein by reference.
- the compounds of formula (I) also show a pronounced effect in the treatment of cellulite, measurable by means of echographic determination of the subcutis layer thickness, in particular to prevent the increased formation of fat stores in the skin and/or cellulite, in that the lipid content in the human subcutaneous fat tissue is reduced.
- the invention therefore relates to cosmetic compositions (preparations), containing a corresponding effective quantity of one or more compounds of formula (I), in particular for the topical treatment and prevention of increased formation of fat stores in the skin and/or cellulite.
- compositions according to the invention are for external application on the skin of humans.
- compositions according to the present invention preferably are provided as ointments, tinctures, creams, gels, solutions, lotions, sprays, aerosols, dry powders, suspensions or the like, preferably in the form of an ointments, gels, creams, lotions, microemulsions, nanoemulsions, o/w-emulsions, w/o-emulsions or multiple emulsions.
- a composition according to the present invention preferably for topical administration to the skin, can be prepared by mixing one or more compounds of formula (I) and/or the salts thereif (as defined above), preferably in the form of a plant extract or a plant extract formulation (as defined above) with one or more non-toxic solid or liquid carriers customarily used in such compositions.
- These compositions may comprise from 5 to 99 wt.%, preferably from 20 to 98 wt.%, more preferably from 30 to 96 wt.% of one or more carriers, based on the total weight of the composition.
- Cream-base cosmetic compositions containing the active agent used in accordance with this invention, can be composed of aqueous emulsions containing a fatty acid alcohol, semi-solid petroleum hydrocarbon, ethylene glycol and an emulsifying agent.
- Ointment formulations comprising the extract in accordance with this invention, for example, can comprise admixtures of a semi-solid petroleum hydrocarbon or fats with a solvent dispersion of the active material.
- Cream compositions comprising the extract for use in this invention can, for example, comprise emulsions formed from a water phase, e.g. using a humectant, a viscosity stabilizer and water, an oil phase of a fatty acid alcohol, a semi-solid petroleum hydrocarbon and an emulsifying agent and a phase containing the active agent dispersed in a aqueous stabilizer-buffer solution.
- Stabilizers may be added to the topical composition. Any conventional stabilizer can be utilized in accordance with this invention.
- fatty acid alcohol components function as a stabilizer. These fatty acid alcohol components are derived from the reduction of a long- chain saturated fatty acid containing at least 14 carbon atoms.
- gels can be used utilising standard gel carriers.
- compositions used according to the invention e.g. preservatives, bactericides, substances impeding foaming, colorants, e.g. dyes or pigments, thickeners (e.g. silica, aluminium silicates, polysaccharides or their derivatives, e.g.
- moisturizers including urea, lactic acid, pyrrolidone carbonic acid, hyluronic acid, polysaccharides rich in fucose, and/or glycerol
- fats oils, waxes or other customary additives, such as polyols, alcohols, polymers, foam stabilizers, electrolytes, other organic solvents, silicones (e.g. cyclomethicone, hexamethylcyclotrisiloxane, polydimethylsiloxane, poly(methylphenyl- siloxane)), perfumes, UV protectors, such as metal oxides which may also be coated, e.g. Ti0 2 , or organic UV absorbers, such as methylene bis-benztriazolyl tetramethylbutylphenol or 4-(tert.-butyl)-4'-methoxydibenzoylmethane) and the like.
- antioxidants conventional antioxidants or mixtures of conventional antioxidants can be incorporated into the topical preparations containing the aforementioned active agent.
- conventional antioxidants which can be utilized in these preparations are included N-methyl-a-tocopherolamine, tocopherols, butylated hydroxyanisole, butylated hydroxytoluene, ethoxyquin, vitamin C, ascorbyl palmitate, vitamin A and the like.
- preferred cosmetic preparations according to the invention containing one or more compounds of formula (I) preferably also contain active ingredients which prevent a breakdown of the connective tissue. Such preparations show improved efficacy in the prophylaxis and cosmetic treatment of cellulite.
- MMPs matrix-metallo-proteinases
- Such preparations are particularly effective in the prophylaxis and cosmetic treatment of cellulite.
- These enzymes are in a position to break down macromolecules of the extra-cellular matrix (ECM) / of the connective tissue, also including the collagens, proteolytically.
- ECM extra-cellular matrix
- MMP-1 matrix-metallo-proteinase-1
- MMP-2 matrix- metalloproteinase-2
- MMP-9 matrix-metallo-proteinase-9
- An inhibition of MMPs is possible, for example, by the addition of ursolic acid, retinyl palmitate, propyl gallate, precocenes, 6- hydroxy-7-methoxy-2,2-dimethyl-1 (2H)-benzopyran, 3,4-dihydro-6-hydroxy-7-methoxy- 2,2-dimethyl-1 (2H)-benzopyran.
- An addition of peptides, which inhibit MMPs, to preparations according to the invention is also advantageous to inhibit MMPs. Proteins or glycoproteins from soya and hydrolysed proteins from rice, pea or lupine also inhibit MMPs and are therefore a suitable addition.
- a combination with a plant extract, which inhibits MMPs is also advantageous. To be mentioned here by way of example is an extract from shitake mushrooms. The combination with extracts from the leaves of the Rosaceae family, sub-family Rosoideae, is also advantageous.
- MMP inhibitors to be preferably used in combination in the scope of the present invention are retinyl palmitate, propyl gallate, precocenes, 6-hydroxy-7-methoxy-2,2-dimethy-1 (2H)- benzopyran, 3,4-dihydro-6-hydroxy-7-methoxy-2,2-dimethyl-1 (2H)-benzopyran, benzamidine Oenothera biennis root extract, ellagic acid and ellagitannins (for example from pomegranate), soya seed extract, ursolic acid, soya isoflavones, sage extracts, lentinus edodes extracts, and extracts from green and/or black tea.
- the combination of active ingredients, which encourage the formation of collagen in the tissue is furthermore advantageous in preferred cosmetic preparations according to the invention containing one or more compounds of formula (I).
- Such preparations are particularly effective in the prophylaxis and cosmetic treatment of cellulite.
- Individual substances frequently used to increase collagen synthesis are, for example, ingredients such as ascorbic acid and their derivatives, retinol and derivatives of retinol or plant extracts such as, for example, extracts of aloe and centella species.
- peptidic materials and their derivatives such as, for example, carnitine, carnosine, creatine, matrikine peptides (e.g.
- lysyl-threonyl-threonyl-lysyl-serine and further peptidic structures such as palmitoylated pentapeptides are also included in the frequently used active ingredients increasing collagen synthesis.
- compounds such as asiatic acid, madecassic acid, madecassoside, asiaticoside, extracts of Centella asiatica, niacinamide, astaxanthine, glucans, for example from yeast and oats, soya extracts and soya isoflavones such as genistein and daidzein, rutin, chrysin, morin, betel nut alkaloids, forskolin, betulinic acid, extracts of Plantago species, TGF-beta, extracts from Ginkgo biloba, glutamine and glycolic acid are also used as collagen synthesis stimulators.
- compositions according to the invention containing one or more compounds of formula (I) further additionally comprise one or more matrix-metalloproteinase inhibitors, and/or one or more collagen synthesis stimulators.
- Such preparations are particularly effective in the prophylaxis and cosmetic treatment of cellulite.
- a method comprises the step of topical application onto the skin, in particular on the thighs (in particular the outer side and the back of the thighs) and/or the buttocks, of a human, preferably a woman.
- the present invention further relates to a pharmaceutical composition comprising a pharmaceutically active amount of one or more compounds of formula (I) as defined herein, preferably for preventing, treating or reducing cellulite. Further, the present invention also relates to a method of treatment of cellulite, comprising the following step: application, preferably topical application, of an effective amount of a compound of formula (I) or a cosmetically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the salts thereof as defined herein or of a cosmtic composition as defined herein. In particular, the present invention relates to a method for the non-therapeutic, preferably cosmetic,
- (iii) modulation of adipocyte differentiation comprising the following step: application of an effective amount of a compound of formula (I) or a physiologically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the salts thereof as defined hereinbefore, a plant extract formulation as defined above or a cosmetic composition as defined above.
- the present invention relates to a compound of formula (I) or a pharmaceutically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the pharmaceutically acceptable salts thereof as defined hereinbefore, a plant extract formulation as defined above, or a composition as defined above, for use in a prophylactic or therapeutic method for treating adiposity (obesity).
- the present invention relates to a pharmaceutical composition
- a pharmaceutical composition comprising an effective amount of one or more compounds of formula (I) as defined above, or a plant extract formulation as defined above, for use in a therapeutic method for preventing and/or treating obesity.
- the present invention also relates to a method for the therapeutic reduction of the body fat content or the therapeutic treatment of obesity, comprising administering, preferably orally, an effective amount of one or more compounds of formula (I) as defined hereinbefore, a plant extract formulation as defined above or a composition as defined above, said effective amount being sufficient to
- compositions according to the invention containing one or more compounds of formula (I) may additionally contain are selected from the following group: preservatives, in particular those described in US 2006/0089413, antimicrobial agents, such as e.g.
- antibacterial agents or agents to treat yeast and mold in particular those described in WO 2005/123101 , antiacne and sebum reducing agents, in particular those described in WO 2008/046791 , compounds against ageing of the skin, in particular those described in WO 2005/123101 , further anti-cellulite agents, in particular those described in WO 2007/077541 , antidandruff agents, in particular those described in WO 2008/046795, antiirritants (antiinflammatory agents, irritation-preventing agents, irritation- inhibiting agents), in particular those described in WO 2007/042472 and US 2006/0089413, antioxidants, in particular those described in WO 2005/123101 , carrier materials, in particular those described in WO 2005/123101 , chelating agents, in particular those described in WO 2005/123101 , deodorizing agents and antiperspirants, in particular those described in WO 2005/123101 , moisture regulators (moisture-donating agents, moisturizing substance, moisture-retaining substances), in particular those described in WO 2005/123
- Preferred liquid carrier substances which may be a component of a preparation according to the invention are selected from the group consisting of glycerol, 1 ,2-propylene glycol, 1 ,2-butylene glycol, 1 ,3-butylene glycol, 1 ,2-pentanediol, 1 ,5-pentanediol, 1 ,2-hexanediol, 1 ,6-hexanediol, 1 ,2-octanediol, 1 ,2-decanediol, ethanol, water and mixtures of two or more of said liquid carrier materials with water.
- these preparations according to the invention may be produced using preservatives, solubilizers or antioxidants.
- Skin care agents preferably skin moisture retention regulators or skin repair agents, preferably are selected from the group consisting of sodium lactate, urea and derivatives, glycerol, 1 ,2-pentanediol, collagen, elastin or hyaluronic acid, diacyl adipates, petrolatum, urocanic acid, lecithin, allantoin, panthenol, phytantriol, lycopene, (pseudo-)ceramides [preferably Ceramide 2, hydroxypropyl bispalmitamide MEA, cetyloxypropyl glyceryl methoxypropyl myristamide, N-(1-hexadecanoyl)-4-hydroxy-L-proline (1-hexadecyl) ester, hydroxyethyl palmityl oxy hydroxy propyl palmitamide], glycosphingolipids, cholesterol, phytosterols, chitosan, chondroitin sulfate,
- compositions according to the invention are cosmetic, in particular dermatological preparations, which are composed as conventional (apart from the compound(s) of formula (I)) and are used for cosmetic, in particular dermatological light protection, for treatment, care and cleaning of the skin and/or hair or as a make-up product in decorative cosmetics.
- preparations of this type can be used, for example, as day protection cream, day or night cream, eye cream, sun protection or after-sun lotion, nourishing cream, a care mask, gel pads, facial tonic, moist care and cleaning tissues, cleaning milk, cleaning soap, foam or shower bath, deodorant, antiperspirant, hair shampoo, hair care agent, hair conditioner, hair colorant, hair styling agent and in this case preferably be present as an emulsion, lotion, milk, fluid, cream, hydro dispersion gel, balm, spray, alcoholic or aqueous/alcoholic solution, foam, powder, liquid soap, piece of soap, shampoo, roll-on, stick or make-up.
- hair treatment agents the use is preferably directed at the base of the hair or the scalp.
- Example 1 Extraction and Extracts Leaves and roots from Labisia pumila var. alata (obtained from plants harvested in Malaysia) were used in the following experiments.
- extract E-6 contained the highest amount of compounds of formula (I). It should be mentioned that the total amount of compounds of formula (I) (as defined above) identified in the respective extracts E-3 to E-6, based on the total amount of extract obtained, was comparatively small (see also Table 2 of example 2.2).
- Example 2 Extracts and pure compound isolated from roots of Labisia oumila
- the n-heptane extract (BTP0056-5-1) was dried (Na 2 S0 4 ) and the solvent evaporated under reduced pressure.
- the remaining water phase was extracted three times with ethyl acetate.
- the combined three ethyl acetate extracts (BTP0056-5-2) were dried (Na 2 S0 4 ) and the solvent evaporated under reduced pressure.
- the remaining water phase (BTP0056-5-3) was also evaporated under reduced pressure and the amounts for the three crude extracts were determined:
- Extract BTP0056-5-2 was selected as starting material for isolating pure compounds.
- the initial separation steps were performed as MPLC separations on reverse phase material in multigram scale allowing separating the most part of the matrix from the analytically detectable compounds.
- a HPLC-setup was used comprising reverse phase separation columns with capacity for up to 200 mg material per separation.
- the gradients for elution were chosen according to the separation problem. Generally the systems were based on water / acetonitrile mixtures.
- Case by case the final separation step for the isolation of pure compounds was performed on a PS1 column (Molecular filtration Chromatography, column provided by Merck, Darmstadt, Germany) using acetonitrile as solvent under isocratic conditions. Every fraction was dried by using a vacuum concentrator and the yield was determined.
- NMR spectra were recorded in DMSO-cfe on a Bruker DRX500 spectrometer at 300 K, operating at 500.13 MHz proton frequency. Structure elucidation was done by thorough interpretation of 1D and 2D NMR spectra, combined with HPLC-MS/UV data including extracted UV as well as positive and negative mode ESI spectra.
- Retention times were obtained by LC- MS/UV analyses performed using an Agilent HP1100 (Agilent, Waldbronn, Germany) liquid chromatograph coupled with a LCQTM (Trademark by Finnigan) Deca XPplus mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) in the positive and negative electrospray ionization (ESI) mode.
- a Waters symmetry column Waters Symmetry® (Trademark by Waters) C 8, 3.5 ⁇ , 2.1 mm x 150 mm, Waters GmbH, Eschborn, Germany) was used as stationary phase with a flow rate of 0.4 ml/min at 40°C.
- Mobile phase A 0.1 % formic acid in water
- mobile phase B 0.1 % formic acid in acetonitrile
- the UV/Vis (ultraviolet/visible light) spectra were recorded between 200-500 nm
- the LC-MS Liquid Chromatography-Mass Spectrometry coupling
- Example 3 Biological activities - Adipocyte testing
- Example 3.1 Adipocyte testing of pure compounds
- 3T3-L1 mouse embryonic fibroblast-like adipocyte cells were cultured and allowed to adhere in 3T3-L1 Pre-adipocyte medium (PM-1-L1), for 1 day . Cells were further incubated for 4 days in PM-1-L1 medium until they were confluent.
- Phase 2 Adipogenesis After additional 48 hrs medium was changed to 3T3-L2 Differentiation Medium (DM-2-L1) containing the test compounds or controls.
- DM-2-L1 Differentiation Medium
- PPAR peroxisome proliferator-activated receptor
- TNF-alpha known inhibitor of PPAR-gamma expression in adipocytes
- PM-1-L1 (3T3-L1 Preadipocyte Medium): Dulbecco's modified Eagle's medium (DMEM), 4.5 g/l glucose, HEPES buffer pH 7.4, 10 % Bovine Calf Serum (BCS), Antibiotics (Penicillin , Streptomycin, Amphotericin B, each 100 pg/ml)
- DMEM Dulbecco's modified Eagle's medium
- BCS Bovine Calf Serum
- Antibiotics Penicillin , Streptomycin, Amphotericin B, each 100 pg/ml
- DM-2-L1 (3T3-L1 Differentiation Medium): DMEM / Ham's F-12 medium (1 :1 , v/v), HEPES pH 7.4, 10% Fetal Bovine Serum (FBS), Biotin, Pantothenate, Human insulin (167 nmol/l), Dexamethasone (1 u-mol/l), Antibiotics (Penicillin, Streptomycin, Amphotericin B, each 100 Mg/ml), Isobutylmethylxanthine , PPAR-gamma agonist (Isoproterenol)
- AM-1-L1 (3T3-L1 Adipocyte Medium): DMEM / Ham's F-12 medium (1 :1 , v/v), HEPES pH 7.4, Fetal Bovine Serum (FBS), Biotin, Pantothenate, Human insulin (167 nmol/l) , Dexamethasone ( ⁇ ⁇ / ⁇ ), Antibiotics (Penicillin, Streptomycin, Amphotericin B, each 100 Mg/ml)
- Table 3.1 Test of individual compounds of obtained from Labisia pumila extracts on the adipogenesis and lipogenesis of murine 3T3-L1 cells (Phases 2 and 3, above)
- Adipostatin A (CAS 3158-56-3)
- TNF-alpha (positive control) 10 ng/ml 15.9
- Example 3.2 Adipocyte testing of extracts Four extracts (E-1 to E-4) according to Example 2 were tested in the assay according to example 3.1 at a concentration of 150 Mg/ml. Said extracts were tested in adipogenesis (Phase 2), lipogenesis phase (Phase 3) and adipogenesis and lipogensis (Phases 2 and 3), respectively, as described in example 3.1 above.
- Table 3.2 Test of extracts from Labisia pumila on adipogenesis murine 3T3-L1 cells (Phase 2, above)
- Example 4 In vivo feeding study with male rats An animal study was performed to prove efficacy (e.g. blood lipid lowering effects) of the compounds of formula (I) (as defined above) in form of extract E-4 (as described above) of the invention in a feeding model of weight gain and body composition.
- efficacy e.g. blood lipid lowering effects
- mice 20 male Sprague Dawley rats (approx. 8 weeks old) were fed for a two week assimilation phase with standard diet (SD, see below). Thereafter the rats were separated into two groups (each 10 rats), one treatment and one control group, that were further fed for eight weeks with SD.
- the treatment group received extract E-4 formulated in a vehicle (see below) via gavage catheter under light anesthesia five times per week (Monday - Friday) once a day in a dosage of 100 mg extract E-4 / kg body weight.
- the control group received the vehicle only.
- the room temperature was maintained at 22 ⁇ 3°C and the relative humidity was kept at 60% + 15%.
- the light/dark period was 12/12 hours.
- Drinking tap water was continuously available ad libitum via drinking bottles.
- Standard diet SD: casein 21.0 wt.%, cornstarch 50.04 wt.%, maltodextrin 3.0 wt.%, sucrose 5.0 wt.%, soybean oil 3.0 wt.%, cellulose 10.0 wt.%, vitamin mix 1.0 wt.%, mineral mix 6.5 wt.%, choline 0.20 wt.%, L-cystine 0.25 wt.%, butylated hydroxytoluol 0.01 wt.%.
- Vehicle The vehicle is composed from 10% (v/v) DMSO (Fisher Scientific D/4121/PB15), 10% (v/v) Cremophor EL (Sigma C5135), 70% (v/v) Saline (0.9% NaCI) and 10% (v/v) HPMC solution (SIGMA H7509).
- DMSO DMSO
- Cremophor EL Sigma C5135
- 70% (v/v) Saline 0.9% NaCI
- HPMC solution SIGMA H7509
- the body weight was measured throughout the study once per weak.
- Body fat analysis (BFA) was performed using dual-energy X-ray absorptiometry (DEXA) at the beginning of the treatment phase (day 0), at the middle and the end of the study.
- DEXA dual-energy X-ray absorptiometry
- the body weight gain and the body fat content in the treatment group receiving extract E- 4 obtained from the Labisia pumila was lower compared to the control group.
- the findings show that a noticeably reduction in fat deposition can be obtained by including one or more compounds of formula (I) (as defined above) in form of an extract in a mammal diet. The observed fat reduction thus confirms the fat-reducing activity of the compounds of formula (I).
- Example 5 In vivo feeding study with male rats
- mice Male Sprague-Dawley weanling rats of three weeks old were used in this study. They were allotted to two treatment groups of five rats each when they weighed an average of about 65 g. The animals were fed carbohydrate enriched diets ad libitum for a period of 31 days. The diet contained the following ingredients: casein 17.42 wt.%, methionine 0.11 wt.%, cornstarch 49.82 wt.%, sucrose 16.60 wt.%, corn oil 6.00 wt.%, cellulose 4.00 wt.%, vitamin mix 0.85 wt.%, mineral mix 5.00 wt.%, choline 0.20 wt.% (referred to as "diet-1").
- the control group received the above “diet-1”.
- the other group received "diet-2", corresponding to "diet-1" to which a total amount of 0.25 wt.% compounds of formula (I) (BTP-00056-8-6 and BTP-00056- 15-03 in a ratio of 1 : 3) was added, based on the weight of "diet-1".
- the body weight gain and the body fat content in animals receiving "diet-2" comprising compounds of formula (I) obtained from the Labisia pumila was lower compared to animals that were fed "diet-1".
- Body fat analysis (BFA) was performed using dual-energy X-ray absorptiometry (DEXA).
- the findings show that a significant reduction in fat deposition can be obtained by including one or more compounds of formula (I) (as defined above) in a mammal diet.
- the observed fat reduction thus confirms the fat-reducing activity of the compounds of formula (I)-
- Example 6 Skin Testing - Slimming and anti-cellulite
- a body lotion comprising a mixture consisting of BTP-00056-24-04 (1 wt.%), BTP-00056- 8-6 (55 wt.%), BTP-00056-15-03 (23 wt.%), BTP-00056-1 -03 (15 wt.%), BTP-00056-16- 03 (5 wt.%) and BTP-00056- 12-03 (1 wt.%) (as described above) in a final total concentration of 0.5 wt.% was tested by a panel of 30 healthy women (Caucasian type). Each of the test subjects treated one leg for two months with a composition according to the invention and the other leg with a control preparation free of compounds according to formula (I).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Emergency Medicine (AREA)
- Dermatology (AREA)
- Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Botany (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Medicines Containing Plant Substances (AREA)
- Cosmetics (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention primarily relates to the use of certain trioxygenated benzene derivatives of formula (I) or a corresponding plant extract as body fat modulating agents, preferably as anti-cellulite actives and/or for the reduction of the body fat content, in particular for reduction of body weight and/or for the management of obesity. The present invention also relates to corresponding methods and to certain novel compounds of formula (I). The invention further relates to specific plant extract formulations obtainable from a plant of the family Myrsinaceae, preferably from the genus Labisia, and to compositions, in particular cosmetic or pharmaceutical formulations, comprising an effective amount of one or more compounds of formula (I) or of a corresponding plant extract formulation.
Description
USE OF CERTAIN TRIOXYGENATED BENZENE DERIVATIVES IN BODY FAT MANAGEMENT
The invention primarily relates to the use of certain trioxygenated benzene derivatives of formula (I) as defined herein or a corresponding plant extract as body fat modulating agents, preferably as anti-cellulite actives and/or for the reduction of the body fat content, in particular for reduction of body fat and/or for the management of obesity. The present invention also relates to corresponding methods and to certain novel compounds of formula (I). The invention further relates to specific plant extract formulations obtainable from a plant of the family Myrsinaceae, preferably from the genus Labisia, and to compositions, in particular cosmetic or pharmaceutical formulations, comprising an effective amount of one or more compounds of formula (I) or of a corresponding plant extract formulation. Weight control and body fat management today are a concern, and excessive fat deposits play an important role in various fields, such as obesity which may result in several health problems, or cellulite which is a cosmetic-aesthetic problem.
Adipose tissue is a connective tissue that functions as a major storage site for body fat in the form of triglycerides. Adipose tissue can increase in size by increase in adipocyte size (hypertrophic growth) and/or by the increase in adipocyte number (hyperplastic growth).
Fat metabolism in the fat tissue of humans, in order to reduce the stored lipid quantity, can in principle be regulated by three Routes:
Route (i): inhibition of the differentiation of preadipocytes. The differentiation of the precursor cells of the fat cells called preadipocytes to the real fat cells, called adipocytes, which may store triglycerides, can be inhibited. Expressed more simply, an inhibition of Route (i) prevents the build up of cellulite in that the number of fat cells does not increase. This process of differentiation from preadipocytes to adipocytes is called adipogenesis.
Route (ii): inhibition of the lipogenesis in adipocytes. The storage of triglycerides in the adipocytes (also called lipogenesis) can be prevented or inhibited. Expressed more simply, an inhibition of Route (ii) prevents the storage of further triglycerides (fats) in the
cell and existing fat cells do not store any new fat. Owing to the natural fat metabolism, when Route (ii) is inhibited, the fat content in the cell decreases.
Route (iii): stimulation of lipoylsis in adipocytes. An augmented/increased hydrolysis of lipids already stored in the adipocytes - also called lipolysis - is possible by targeted stimulation. Expressed more simply, stimulation of Route (iii) increases the breakdown of the fats already present in the cell while an inhibiting, i.e. antagonistic effect with respect to Route (iii) on the other hand inhibits or prevents the breakdown of fat.
The differentiation of cells is the changing of the control of the gene activity of a cell so that various protein stores are provided in the cells by means of transcription and protein biosynthesis and the cells differ according to appearance and function. Thus, adipocytes only express enzymes, which are necessary for the storing of fats, after differentiation. In their precursor cells, the undifferentiated preadipocytes, these enzymes are not expressed or only to a very small extent.
Increases of the number of adipocytes in early life stage and the content of triglyceride in adults are thought to be the cause of corpulence and obesity. Therefore, an important approach for corpulence prevention may be to control the regulatory system of adipocytes proliferation and accumulation of triglyceride.
Excessive accumulation of body fat, i.e. obesity (= adiposity), especially with excessive fat in the ventral region and surrounding the viscera)) can be dangerous and has been linked to health problems such as type II diabetes, hypertension, heart disease, atherosclerosis (where more than two of the preceding disorders are present, the condition is often called "metabolic syndrome"), hyperlipidemia, coronary heart disease, stroke, breast and colon cancer, sleep apnoea, gallbladder disease, gastroesophageal reflux disease, fatty liver, gout or thromboembolism. Obesity is one of the main factors in the development of cardiovascular diseases. As a side effect the levels of cholesterol, blood pressure, blood sugar and uric acid in obese people are usually higher than those of persons of normal weight. The morbidity from coronary heart disease among the overweight people is increased as well. Among the people aged 40-50, mortality will rise about 1% when body weight increases by 0.5 kg and the death rate will increase 74% when body weight exceeds 25% of the standard. The prevalence of obesity in the United States has more
than doubled since the turn of the last century (whole population) and more than tripled within the last 30 years among children aged from 6 to 11. Furthermore, being overweight is considered by the majority of the Western population as unattractive. Determinants of obesity include social factors, psychological factors, genetic factors, developmental factors and decreased physical activity.
Great interest by consumers in the maintenance or reduction of their body fat can be found. This leads to a demand for products useful for these purposes. Preferred are such food products which can conveniently be consumed as part of the daily diet, for example meal replacer products, such as meal replacer bars and beverages. These are usually designed for use as a single-serving food product to replace one or two meals a day.
WO 2004/082700 describes compositions comprising as essential constituent the extracts from four plants and an animal, and other plant extracts in the treatment of obesity. Only a combination of 14 extracts was examined in the example and shown to reduce the weight of mice by 6.4 to 9.2 %. Due to the large number of extracts administered, it is not possible to understand which of the extracts is responsible for the effects observed, in particular whether omission of one of more of these extracts would be possible without impairing the desired effects.
WO 2004/096252 describes compositions comprising mushroom powder, extract, or a derivative thereof in combination with a liver protecting agent, wherein the mushrooms were selected on the basis of a hunger suppressing activity and fat burning capability.
Adipostatin A (5-n-pentadecylresorcinol) is known as inhibitor of glycerol-3-phosphate dehydrogenase and for markedly inhibiting triglyceride accumulation in murine 3T3-L1 cells (see Journal of Antibiotics 1992, 886-891).
3T3-L1 cells, a clone of Swiss/3T3 fibroblast, are transformed to adipose cells under certain culture conditions being accompanied by an increase of activities of many lipogenic enzymes. Therefore, adipocyte differentiation testing in murine 3T3-L1 cells can be regarded as a good model to study these effects.
WO 2009/021622 A1 discloses the use of certain compounds or extracts comprising such compounds obtainable from plants from the genus from the group Schisandra, lllicium, Kadsura, Steganotaenia and Magnolia as active ingredient in the regulation of body weight and/or fat loss.
Cellulite is also known under the synonyms protrusio cutis and colloquially as orange peel skin. It is a cosmetic-aesthetic problem which is accompanied by the formation of dimples and indentations of the skin and nodule formation of the subcutaneous fat tissue. Cellulite can occur on any part of the human body, but the outer side and the back of the thighs as well as the buttocks are most frequently affected. Breasts, lower stomach, upper arms or neck are also sometimes affected by cellulite. Cellulite may be regularly found on parts of the human body with excessive fat deposits, but overweight is not a prerequisite for its occurrence. Slim women increasingly also have pronounced cellulite symptoms. However, there is probably a correlation between the severity of the cellulite and the percentage of fat in the tissue.
The gender-specific anatomic structure of the skin of human beings (humans) has a great influence on the development of cellulite. Thus, for example, cellulite can only seldom be observed in men, while, on the other hand, about 80% - 90% of all women are affected, in particular Caucasian women. The structure of the dermis, in particular, has an effect on the skin relief. Thus, the fat chambers in men, when the skin is pressed together, are held back by intersecting connective tissue septa and the clamp-like enclosure of the fat cells connected therewith. On the other hand, in women, the fat chambers separated from one another in a tubular manner bulge up when being pressed together.
In addition, the visible pattern of the cellulite is based on an increase in fat cushions in the subcutis and a reduction in the circulation conditions in the blood and lymph vessels. The cause is therefore partly a predisposed weakening of the connective tissue with simultaneous occurrence of enlarged fat cell chambers with stress, sports activity, smoking, pregnancies and female hormones (estrogen and progesterone) playing a part, in addition to genetic factors.
Cellulitis is to be clearly separated and distinguished from the cosmetic phenomenon of cellulite. Cellulitis is a bacterial infection of the subcutaneous tissue, which in many cases may be a serious illness, and in contrast to cellulite, has to be treated therapeutically.
As mentioned above, even healthy women are affected by cellulite. It should be stressed that cellulite itself is not an illness and thus its treatment is not to be regarded as therapy. Light or moderate cellulite, which is considered as healthy skin, is not a condition or blemish that requires medication and is not regarded as a pathological state. In contrast thereto, heavy cellulite may be accompanied by side or after effects like pain or other medical symptoms. Medical specialists can clearly distinguish between light or moderate cellulite and heavy cellulite and medical specialists also decide whether a treatment of cellulite in the medical sense is advisable or needed.
The conventional treatment methods for cellulite attempt to encourage the blood circulation of the relevant skin parts and to positively influence the connective tissue structure, for example by massage, lymph drainage, diet, sport, magnetic fields or else liposuction (removing fat by suction).
In the literature, the use of several cosmetic products is described for the prophylaxis and treatment of cellulite. But their effectiveness is often very limited because of the very complex mechanism of fat cell metabolism.
Cosmetic preparations which have the prophylaxis and treatment of cellulite as a goal have already been proposed in the literature. They mostly influence adipose tissue or adipocytes by a specific activity.
EP 1 234 572 describes a cosmetic preparation of at least one isoflavone aglycone, in particular genistein and/or daidzein, for treating cellulite. The isoflavone aglycone is in this case combined with an algae extract. Genistein is described there as an active ingredient, which inhibits the multiplication of precursor fat cells and in addition the enzyme phosphodiesterase.
Preparations containing certain isoflavones are also described in DE 100 09 423, the isoflavones being described as materials with an anti-estrogen effect and used because of
this effect. Daidzein, genistein, glycitein, formononetin and others are preferred isoflavones there.
WO 2006/063714 teaches compositions for topical administration, containing a PDE3 inhibitor as active ingredient, for use in the treatment of cellulite and proposes pharmaceutical compositions comprising drugs like anagrelide, cilostazol, pimobendan, milrinone, amrinone, olprinone, enoximone, cilostamide, vesnarinone and trequinsin.
US 2007/043109 A1 suggests certain diterpenes as slimming agents. As explained above, adipocyte metabolism is a complex mechanism which needs the alteration of different pathways within the fat metabolism to be effective. Influencing only one of the different pathways is generally not effective in humans because in parallel other pathways are influenced which in their turn lead to an increase of stored lipid quantity, leading to an adverse effect. The effectiveness of the substances proposed in the prior art so far is often very limited.
The primary object of the present invention was to identify alternative active substances and compositions that can reduce the body fat content, and/or reduce the lipid quantity contained in fat tissue in mammals, preferably in human beings. Said substances and compositions should show a, preferably improved, activity regarding differentiation of preadipocytes and/or lipogenesis in adipocytes (cf. Route (i) and/or Route (ii) as described above). More particularly, said substances and compositions should be able to promote the loss of body fat and thus be useful in the treatment of obesity and to show activity with respect to the prophylaxis and treatment of cellulite. Additionally, said substances should preferably be naturally occurring compounds.
It has now been found that this primary object can be achieved by using one or more compounds of formula (I) or a physiologically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the salts thereof. In a first aspect, the present invention relates to the use of a compound of formula (I) or a physiologically, preferably cosmetically, acceptable salt of a compound of formula (I) or a
mixture containing two or more of these compounds or the physiologically acceptable salts thereof
(i) for the cosmetic prevention, treatment or reduction of cellulite, and/or (ii) for the non-therapeutic, preferably cosmetic, reduction of the lipid quantity contained in subcutaneous fat tissue, and/or inhibition of the differentiation of preadipocytes,
and/or
inhibition of the lipogenesis in adipocytes,
and/or
(iii) as cosmetic anti-cellulite active,
R1 denotes hydrogen or an alkyl moiety having 1 to 4 carbon atoms, R2 denotes methyl or an acyl moiety having 1 to 4 carbon atoms, each R3 denotes hydrogen or both R3 together denote an oxygen atom, and Y denotes an organic radical having 6 to 24 carbon atoms.
Surprisingly, it now has been found that the compounds of formula (I) and/or their salts as defined herein and certain extracts (as defined hereinafter) of plants or parts of plants of the family Myrsinaceae comprising one or more of said compounds of formula (I) and/or salts thereof can reduce the body fat content, and/or reduce the lipid quantity contained in
fat tissue, and/or inhibit the differentiation of preadipocytes, and/or inhibit of the lipogenesis in adipocytes, and/or modulate the adipocyte differentiation.
In particular, in was found that the compounds of formula (I) and/or their salts (as defined herein) show a superior activity compared to adipostatin A (see example 3.1 below). Without wishing to be bound by theory it is assumed that the trioxygenated benzene moiety and the substitution pattern on the benzene ring of the compounds of formula (I) is responsible for the improved activity. The compounds of formula (I) and/or their salts as defined herein and certain extracts obtainable from plants of the family Myrsinaceae, preferably of the genus Labisia, in particular of Labisia pumila, were shown in own experiments to be potent inhibitors of adipocyte trigyceride accumulation and thus can be used for body fat reduction, and in particular are effective in achieving weight loss and/or reducing weight gain (i.e. less weight gain) in comparison to an untreated control group.
There is no indication hitherto that the compounds used in accordance with the present invention are suitable for the cosmetic treatment of cellulite. Compounds of formula (I) and compositions (preparations) according to the invention, comprising one or more compounds of formula (I) influence cellulite with regard to the stored lipid quantity, in that the lipid content in the body is reduced.
In the context of the present invention, a cosmetic use or a cosmetic method is free of any therapeutic (side) effects. "Cosmetic" especially refers to a non-therapeutic use, that is, a use not for treatment of a disease or condition for its cure or symptom reduction but merely to improve the aesthetic appearance of an individual.
In the context of the present invention, a therapeutic or pharmaceutical use or method is considered as medical treatment, optionally with cosmetic (side) effects. In accordance with the present invention, cellulite is prevented, treated or reduced by a cosmetic composition containing one or more compounds of formula (I) by influencing the
above described Routes (i) and/or (ii), most preferably by influencing the above described Routes (i) and (ii).
To determine whether a compound exhibits an activity in the sense of the present invention corresponding to Route (i) [inhibition of adipogenesis (differentiation of preadipocytes)] and/or Route (ii) [inhibition of the lipogenesis in adipocytes], preferably tests are performed in accordance with the examples given below.
By inhibiting proliferation of preadipocytes the number of precursor cells is reduced and the process of adipogenesis (Route (i)) indirectly results in a lower number of adipocytes. Surprisingly it was found that the compounds of formula (I) and/or the salts thereof (as defined herein) inhibit the proliferation of preadipocytes.
In summary, the compounds of formula (I) and/or their salts as defined herein and certain extracts comprising one or more of said compounds of formula (I) or physiologically acceptable salts thereof are actives for the reduction of body fat, i.e. to achieve fat loss, and thereby thus are capable of allowing weight control in humans and thus the treatment of obesity. Further, they are cosmetic actives for the cosmetic applications mentioned herein, in particular for prevention, treatment and/or reduction of cellulite.
The compounds of formula (I) may be used in the context of the present invention in all their isomeric forms, i.e. - depending on their structure - as enantiomers, diastereomers, syn-/anti-isomers, cis-/trans-isomers, epimers as well as (E)-/(Z)-isomers. The compounds of formula (I) can be used in the context of the present invention in the form of the pure stereoisomeric form or in the form of any mixture of stereoisomers. The compounds of formula (I) can also be used in the context of the present invention in the form of the pure enantiomers or in the form of any mixture of enantiomers, in the latter case racemates being preferred. To the extent that compounds of formula (I) and physiologically acceptable salts thereof may exist in their tautomeric form, all such tautomeric forms are contemplated herein as part of the present invention.
In the compounds of formula (I) at least one phenolic hydroxyl group is present. Thus, the compounds of formula (I) may be in the free form or in the form of salts. The term "salt(s)", as employed herein, denotes salts formed with inorganic and/or organic bases. "Obtainable" means that a product (e.g. extract or compound) may be obtained by a certain method, and preferably is obtained by said method.
Where ratios or percentages are given, these refer to the weight (e.g. percent by weight, wt.%), unless indicated otherwise.
"Comprising" or "including" wherever used herein is meant not to be limiting to any elements stated subsequently to such term but rather to encompass one or more further elements not specifically mentioned with or without functional importance, that is, the listed steps, elements or options need not be exhaustive. In contrast, "containing" would be used where the elements are limited to those specifically after "containing".
By the term "extract", either a direct extract (in liquid or preferably dried form), e.g. obtained as described below, or preferably a further enriched extract (obtainable e.g. by one or more further purification steps after extraction, e.g. chromatography, for example as described below) containing one or more, preferably two or more compounds of formula (I) is meant.
By "administered" or "administering" herein is meant administration of a prophylactically and/or therapeutically effective dose of a compound of formula (I) or a mixture of compounds of formula (I), or an extract comprising one or more the compounds of formula (I), to a human being in need of such treatment. By "effective amount" or "effective dose" herein is meant an amount or a dose that produces the (therapeutic or cosmetic) effects for which it is administered, especially a reduction of weight, in particular (due to) the reduction of body fat.
A "patient" or "subject" for the purposes of the present invention relates to mammals, especially human beings. Thus, the compounds of formula (I) or a mixture of compounds of formula (I), or a plant extract comprising one or more compounds of formula (I), are
applicable to both humans and mammals. In the preferred embodiment the patient is a human. The patients will be treated either in prophylactic or therapeutic intention, the latter e.g. to avoid regain in weight after a weight (especially body fat) reduction (e.g. to avoid the yo-yo effect), or to avoid weight gain (especially due to an increase in body fat) ab initio.
The terms "dry", "dried form", "dry weight" and the like refer to matter (such as an extract, a composition etc.) without water and without organic solvents, in particular being free of water and free of substances having a boiling point of less than 300°C at 1013 mbar.
The terms "liquid" and "solid" refer to the state of matter, e.g. a compound, carrier or composition, at 25°C and 1013 mbar.
The terms "reduction of body fat" and/or "management of obesity" (especially leading to body weight and/or fat reduction or for treating, including preventing, obesity), especially refer to the effects achieved by administration of one or more compounds of formula (I) or preferably an (especially further enriched) extract comprising one or more, preferably two or more, compounds of formula (I) either a lower weight gain can be observed or preferably a weight loss can be observed, in comparison to a control without such administration. More preferably, the weight loss is due to a reduction of the body fat. Thus, most preferably, with "treatment of obesity" is meant to either prophylactically avoid a weight gain in a mammal, preferably a human being, or preferably to reduce the body weight, especially to reduce the body fat in a mammal, preferably a human being.
"Slimming", in particular "dermal slimming", according to the present invention describes an improved appearance of skin, mainly based on adipocyte differentiation modulation (inhibition of triglyceride uptake). Pharmaceutically (or nutraceutically) acceptable (i.e. non-toxic, physiologically acceptable) salts are preferred, although other salts are also useful, e.g. in isolation or purification steps which may be employed during preparation. Salts of the compounds of formula (I) may be formed, for example, by reacting a compound of formula (I) with an
amount of acid or base, such as an equivalent amount, in a medium such as one in which the salt precipitates or in an aqueous medium followed by lyophilization. Also ion exchangers can be used to form salts from free forms or free forms from salts of a compound of formula (I).
Wherever in the present disclosure a compound of formula (I), a mixture of compounds of formula (I) or one or more compounds of formula (I) are mentioned, this intends to include the free (enriched or substantially pure) form and/or one or more (especially pharmaceutically or nutraceutically salts (alone or together referred to as pharmaceutically acceptable salts hereinafter)) where salt-forming groups are present, tautomer(s) (where tautomerism, e.g. of the oxo/enol type, is possible), or mixtures of two or more of these specific forms.
Preferred physiologically, preferably cosmetically, pharmaceutically or nutraceutically acceptable salts of compounds of formula (I) are those in which the one, several or preferably all counterions (counteracting cation) are selected from the group consisting of Na\ K+, NH4 +, trialkylammonium NHR'3\ Ca2+, Mg2+, Zn2+ and Al3+.
In trialkylammonium NHR'3 +, preferably each R' independently of the other radicals R' denotes an alkyl group having 1 to 30 C-atoms, preferably having 4 to 22 C-atoms. Particular preferred counterions in physiologically, preferably cosmetically, pharmaceutically or nutraceutically, acceptable salts of compounds of formula (I) are selected from the group consisting of Na+, K+, Ca2+ and Mg2+ and mixtures thereof. Several trioxygenated benzene derivatives corresponding to formula (I) are known per se, some of these compounds are described as occurring naturally.
4-hydroxy-2-methoxy-6-pentadecylphenyl acetate (Ry = n-propyl) and 4-hydroxy-2- methoxy-6-tridecylphenyl acetate of formula (Ry = methyl)
were identified from the roots and stems of Ardisia virens Kurz (syn. Ardisia polysticta, genus: Ardisia; family: Myrsinaceae) (see Phytochemistry 2009, 70, 2064-71). -pentadec-8-en-1 -yl]phenyl acetate of formula
was identified - together with 4-hydroxy-2-methoxy-6-pentadecylphenyl acetate (vide supra) and several known alkyl resorcinols - from the roots of Ardisia brevicaulis Diels (see Chem. Biodiv. 2010, 7, 2901-7).
The following compounds of formula (I) are reported to be naturally occuring:
from the fruit of Ardisia colorata, and from Iris spp. the following were reported:
wherein Rz denotes methyl or n-propyl.
Preferably, the compounds of formula (I) are natural compounds, i.e. compounds that are present in and can be isolated or extracted from natural sources (especially from plant sources mentioned in detail hereinafter) without chemical synthesis steps (though they may also be prepared by chemical synthesis).
In own investigations we were able to identify and to subsequently isolate the following compounds of formula (I) from certain extracts obtained from of Labisia pumila:
It should be noted that these compounds were essentially found in the roots of Labisia pumila. They were not found in water extracts from Labisia pumila leafs, and only in trace amounts in water extracts from Labisia pumila roots (see also example 1 below). The best
way found to obtain these three compounds is to extract the roots from Labisia pumila with an aqueous alcoholic mixture, in particular as described in more detail below.
Preferred compounds used in accordance to the present invention are compounds of formula (I) and/or the physiologically acceptable salts thereof, wherein
R1 denotes hydrogen, methyl or ethyl, and/or
R2 denotes methyl, formyl or acetyl, and/or Y denotes an organic radical having 8 to 22 carbon atoms.
More preferably, the compounds used in accordance to the present invention are compounds of formula (I) and/or the physiologically acceptable salts thereof, wherein
R1 denotes hydrogen or methyl,
R2 denotes methyl or acetyl, and Y denotes an organic radical having 8 to 22 carbon atoms and consists of carbon, hydrogen and oxygen, wherein Y contains 0, 1 , 2, 3, 4 or 5 oxygen atoms.
More preferably, the compounds used in accordance to the present invention are compounds of formula (I) and/or the physiologically acceptable salts thereof, wherein
Y denotes a n-alkyl radical having 8 to 12 carbon atoms, Y denotes an n-alkenyl radical having 8 to 12 carbon atoms and 1 , 2 or 3 carbon-carbon double bonds, or
Y denotes an organic radical having 17 to 21 carbon atoms consisting of carbon, hydrogen and 3 or 4 oxygen atoms.
Due to their higher activity regarding the effects to be achieved in the context of the present invention, particularly preferred compounds of formula (I) and/or the physiologically acceptable salts thereof are selected from the group consisting of:
It is evident to those skilled in the art that the compounds formula (I), also the naturally occurring compounds formula (I), can be synthesized according to known methods, for example those described in US 5,352,690 (disclosing certain 1 ,2,4-trioxygenated benzene derivatives and their use as leukotriene antagonists), March's Advanced Organic Chemistry: Reaction, Mechanisms and Structure, 5th ed. by Michael B. Smith, Jerry March, Wiley-lnterscience; 2001 ; Classics in Total Synthesis: Targets, Strategies, Methods by Nicolaou, Sorensen, John Wiley & Son Ltd, 1996 and Angew. Chem. Int. Ed. Engl. 2000, 39 (1): 4 -122.
S. Foster extensively compiled (research) information available to date on Malysian traditional medicinal plants, including Labisia pumila (S. Foster, Balancing nature and wellness: Malaysian traditions of "Ramuan" - The history, culture, biodiversity and scientific assimilation of medicinal plants in Malaysia, HerbalGram. 2009; 84: 30-43; American Botanical Council).
"Kacip Fatimah" (Labisia pumila - which is synonymous to Labisia pothonia or Ardisia pumila) has been used by many generations of the Malay women to induce and facilitate childbirth as well as a post-partum medicine. The decoction of the leaves and roots is valued to help delay conception and regain strength after childbirth. The decoction may also be given 1-2 months before childbirth. Modern perceptions of its health benefits include being a useful herb to contract the uterus after childbirth; firm and tone abdominal muscles; tighten the vaginal wall and tissue; allay painful or difficult menstruation, cramping, and irregular periods; and to generally alleviate fatigue and promote emotional well-being. Other reported traditional uses of the plants include treating dysentery, flatulence, rheumatism and gonorrhoea.
"Kacip Fatimah" used in traditional Malay medicine may also be referred to as Selusoh fatimah, Rumpur siti fatimah, Akar fatimah, Kacit fatimah, Kachip Fatima, Kachip patima, Kunchi fatimah, Pokok pinggang, Rumput palis, Tadah matahari, Mata pelanduk rimba (Malay), Bunga belangkas hutan or Sangkoh (Iban).
In Malaysia, "Kacip Fatimah" has not only been used for centuries, but it is taken on a regular basis by Malay women today. As traditional and conventional products solely water decoctions are used. The fresh or dried plant is boiled in a cooking pot, either alone or in decoction with other herbs. The plants are usually boiled and water soluble extract taken as a drink. The plant or parts or parts of the plant as well as formulations with other herbs, are readily available in powders or extracts in pills, capsules, mixtures with tea or coffee, and as a canned beverage. Used regularly it is believed to increase vitality, libido, and energy. As chemical constituents from the leaves and the roots of Labisia pumlia var. alata benzoquinoid compounds, certain alkenyl resorcinols, and triterpenoid compounds are reported (Phytochemistry 2011 , doi:10.1016/j.phytochem.2011.06.014, article in press).
Antioxidant components found in Labisia pumila include ascorbic acid (vitamin C), beta carotene, anthocyanins, and certain flavonoids (J. Med. Plants Res. 2009; 3(4):217-223).
Several commercial products, such as capsules or tablets, reputed to contain this plant or parts thereof, or extracts of said plant are available in Malaysia. However, no general or official specification exists at present. Also, extraction processes still lack reproducibility and reliable profiling methods. In particular, only few information and data for individual substances contained in said plant or extracts thereof are available, in particular relating to specific cosmetic or therapeutic effects of said compounds.
The genus Labisia belongs to the family Myrsinaceae and contains several species. The species Labisia pumila is further subdivided into several varieties (var. pumila, var. alata, var. discoplacenta, var. lanceolata, var. malintangensis, var. neriifolia, var. sessilifolia). Of these varieties, L. pumila var. alata seems to be the most commonly used in traditional medicine.
Labisia pumila is distributed throughout South-East Asia and is found in throughout Malaysia, Sumatra, Java, Borneo, Thailand, Cambodia, Indonesia, etc. The plants can be found in the undergrowth of tropical forests. It can be cultivated in shaded places on moist sandy loam soil, with liberal application of organic matter and fertilizers.
Labisia pumila is a small herbaceous shrub. The root is tough and woody with long primary roots and few secondary roots. There are few leaves pointing upwards. The whole leaf is about 5-35 cm long and 2-8 cm wide and finely toothed with numerous veins. It is of a dark green color on the adaxial and lighter green on the abaxial side.
In the context of the present invention, the species Labisia pumila is preferred, in particular Labisia pumila var. alata, Labisia pumila var. lanceolate, and Labisia pumila var. pumila.
Jamal et al. in article "Kacip Fatimah: A Malay Traditional Herb for Pregnant Women" (in "Phytochemicals and Biopharmaceutics from the Malaysian Rain Forest", edited by A. Ali, K. Shaari and Z. Zakaria, Forest Research Institute Malaysia (FRIM) in Kepong, 1999, 166- 176) investigated water and ethanol extracts of powdered roots and leaves of Labisia pumila var. alata and Labisia pumila var. pumila. A hot ethanol extract of Labisia pumila var. alata root was found to be weakly estrogenic in vitro at concentrations between 10 and 75 pg/ml and could contribute to its reputed use for enhancing and speeding parturition, whereas other extracts did not show any significant estrogenic activity.
According to J. Pharm. Pharmacol. 1998, 50 (Supplement): 79 ethanol and water extracts of both Labisia pumila var. alata and Labisia pumila var. pumila roots and leaves did not show any significant estrogenic activity.
J. Pharm. Pharmacol. 1999, 51 (Supplement): 236 discloses that the ethanolic extracts showed different profiles in thin layer chromatography between leaves and roots and between the two varieties Labisia pumila var. alata and Labisia pumila var. pumila. Additionally, two benzoquinoid compounds from Labisia pumila var. alata leaves and roots were identified.
WO 2009/064155 and WO 2009/064156 describe a water-soluble extract obtained by extraction of extraction of Labisia pumila plant material with water, in particular extracts obtained from dried leaves of Labisia pumila and distilled water in a ratio of 1 : 8, as having the capability to develop a pharmaceutical composition for hepatoprotective or immunostimulating activity.
US 2007/0082069 A1 and US 2010/0311180 A1 relate to processes for preparing a water- soluble extract by extraction of one part of dried Labisia pumila plant material with 6 parts of water, said extract having a reproducible chemical profile by reverse-phase HPLC, using 3,4,5-trihydroxybenzoic acid as marker substance. WO 2010/147449 A1 describes a process for extracting the leaves of Labisia pumila with water into a free flowing powder. Further, WO 2010/147449 A1 describes that an aqueous leaf extract of Labisia pumila resulted in an increase of the body weight of mice after 14 days.
KR 2009/0113723 A relates to compositions containing an extract or a fermentation product of Labisia pumila providing activity of UV protection, whitening, anti-aging and anti-oxidation activity and improving skin function. The Labisia pumila extract according to KR 2009/0113723 A is obtained using as organic solvent ethanol, hexane, or acetone. The cosmetic composition may be in the form of soap, lotion, cream, pack and cosmetic liquid. WO 2009/116848 A1 discloses a standardized aqueous extract of Labisia pumila that can reduce the risk or progression of cardiovascular diseases, said extract is described as being capable to influence pathophysiological changes, including regulation of various related genes, associated with insulin resistance, obesity, metabolic syndrome, diabetes and aging. In WO 2009/116848 A1 a standardized aqueous extract of Labisia pumila leaves prepared by the method as disclosed in Malaysian patent application PI 20054784 (corresponding to US 2007/0082069 A1 , vide supra) was used.
Zulfiqar et al. isolated certain alkyl phenols and saponins from a methanolic extract from the powdered roots of Labisia pumila (Phytochemistry 2011 ,
doi:10.1016/j.phytochem.2011.06.014, article in press). Some of the isolated compounds showed moderate in vitro antibacterial activity.
The total phenolic content and the total flavanoid content in extracts obtained by extraction of Labisia pumila leaves with 100% methanol, 100% water, 60% methanol and 100% acetonitrile was determined by Chua et al., Food Chemistry 127 (2011) 1186-1192. Several flavonoids and phenolic acids were detected by LC/MS methodology (e.g. flavonols like quercetin, flavanols like catechin and epigallocatechin, benzoic acids like gallic acid, vanillic acid or salicylic acid, and cinnamic acids, e.g. coumaric acid and caffeic acid.
Several publications describe activities of Labisia pumila extracts regarding body weight and adipocyte cells. The following table gives a brief summary thereof.
Journal / Source
Malays. J. Med. Sci. 2005; In vivo toxicologival study (teratogenicity) in female rats 12(2): 13-21
Result: An aaueous extract of Labisia oumila sliahtly increased maternal body weight.
Indian Journal of Evaluation Of The Female Reproductive Toxicity Of The Pharmacology 2006; 38, Aqueous Extract Of Labisia pumila var. alata in rats
355-356
Result: There were no changes in maternal body weiaht.
Journal of Medicine 2007; In vivo study on ovariectomized rats; search for estrogenic 1(1) activity of Labisia pumila extracts
[J. Med. Biol. Sci. 2007; Result: The results implied a possible role of a water 1 (1)] extract of Labisia pumila var. alata in modulating postmenopausal adiposity in a manner similar to that reported for estrogen through initiation of the lipolysis process in adipose tissues.
Maturitas 2009; 62(1 ):91- In vivo study on ovariectomized rats; search for estrogenic
97 activity of Labisia pumila extracts
Result: The effects of and compared to estroaen replacement (ERT), on body weight gain, uterus weight, adipose tissue mRNA and protein levels of adipokines in ovariectomized female Sprague-Dawley rats were investigated. The study provides evidence that a standardized water extract of Labisia pumila var. alata contains biologically active phytoestrogens with uterotrophic effect that could regulate body weight gain at least partly by modulating secretion of leptin and resistin, and expression of the adipokines in adipose tissues.
J. Ethnopharmacology The effect of an extract from Labisia pumila var. alata on 2010; 127(2): 346-51 body composition and metabolic features in a rat model of polycystic ovary syndrome (PCOS) was investigated.
Result: The mechanism of action was associated with a uterotrophic effect and regulating body weight gain by changing the expression and secretion of adipokines, leptin, and resistin in adipose tissue. Body weight development was not affected, but uterine weight was increased, indicating potential estrogenic effects. No effects on body composition or adipocyte size were observed. http://www.kacipfatima.net/ In vivo toxicology study (90 day sub- chronic, male and female rats)
Result: There was no decrease in weekly bodv weights of the animals tn treated groups as compared to control group in both male groups.
Possible estrogenic activity thus has been proposed as an explanation for traditional use by pregnant women, and two studies have shown a tendency towards possible estrogenic activity. In summary, the findings reported in the publications cited above are quite diverting and even contradictory in results and interpretations. There is hence no guidance provided in the prior art as to the compounds and substances that could be responsible or crucial for achieving the effects reported. Said investigations were carried out with water extracts from Labisia pumila which at most contain trace amounts of compounds of formula (I) and/or physiologically acceptable salts thereof as defined in the context of the present invention. Water extracts from Labisia pumila do not contain an effective amount of compounds of formula (I) and/or physiologically acceptable salts thereof as defined in the context of the present invention. In contrast to the results reported in the prior art in our investigations the compounds of formula (I) and/or the physiologically acceptable salts thereof did not exhibit lipolysis activity (Route (iii), vide supra).
A plant extract, preferably an enriched plant extract, according to the present invention comprises an effective amount of one or more compounds of formula (I) as defined herein and/or a physiologically acceptable salt thereof, preferably in a total amount in the range from 0.1 to 40 wt.%, more preferably from 0.2 to 20 wt.%, particularly preferably from 0.5 to 15 wt.%, most preferably from 1 to 8 wt.%, most preferably from 1.5 to 6 wt.%, based on the total weight of the plant extract.
In a preferred embodiment the present invention relates to a corresponding use or method, wherein one or more compounds of formula (I) and/or physiologically acceptable salts thereof as defined above, preferably as defined in any of the above mentioned preferred embodiments, wherein these are used as part or in the form of a plant extract, preferably a plant extract from a plant of the family Myrsinaceae, more preferably from the genus Labisia, more preferably from Labisia pumila.
Preferably, the total weight share of one or more compounds of formula (I) in an extract used according to the invention is in the range from 0.1 to 25 % by weight, more preferably from 0.25 to 15 %, particularly preferably from 1 to 10 %, most preferably from 1.25 to 5 %, based on the total dry weight of the extract.
For testing purposes, it is possible to conduct clinical trials, e.g. clinical trials analogous to those described in WO 2004/096252 or WO 2004/082700 (which are incorporated here by reference, especially with regard to the description of the tests), but only using one or more compounds of formula (I) or an extract comprising such compound(s) of formula (I) as described for the present invention.
The extracts or compounds according to the invention may be used as such, in the form or pharmaceutical or nutraceutical formulations (the latter term including food additives) or in the form of functional food.
Where the compounds or mixture of compounds of formula (I), especially extracts comprising one or more compounds of formula (I), are used as supplement, this means that the compound(s), extract or a pharmaceutical or nutraceutical formulation comprising it or them can be added to any other nutrient or pharmaceutical or nutraceutical, preferably other than (exclude especially mixtures known). Thus they can especially serve as food supplement. However, the compound(s), extract or formulations may also be administered as such.
"Nutraceuticals", "Functional Food", or "Functional Food products" (sometimes also called "Foodsceuticals", "Medicinal Food" or "Designer Food") for use according to the present invention are defined as food products (including beverages) suitable for human consumption - the expression comprises any fresh or processed food having a health- promoting and/or disease-preventing property beyond the basic nutritional function of supplying nutrients, including food made from functional food ingredients or fortified with health-promoting additives, especially with effects in the prophylaxis or treatment of obesity, especially allowing for body weight reduction and/or body weight maintenance, appetite suppression, the provision of satiety or similar changes in metabolism, and in which an extract, a compound or compound mixture of compounds of formula (I),
respectively, according to the invention is used as an ingredient (especially additive) as health benefit agent, especially in an effective amount.
The functional food products or pharmaceutical products may be manufactured according to any suitable process, preferably comprising extraction of one or more compounds of formula (I) and admixing to a functional food product or at least one physiologically, preferably cosmetically, nutraceutically or pharmaceutically, acceptable carrier.
Preferably, a functional food or a pharmaceutical or nutraceutical formulation comprising a compound, more preferably a compound mixture, for use according to the present invention, can be obtained by
(a) extraction of one or more compounds and/or mixture of compounds of formula (I) from the plant or parts of the plant Labisia pumila, and
(b) mixing the resulting one or more compounds and/or mixtures of compounds as active ingredient in the preparation of the functional food product with the other constituents thereof or in order to obtain a pharmaceutical or nutraceutical formulation with one or more carrier materials or with a solvent, e.g. water or an aqueous solvent (e.g. to give a juice or dispersion or solution).
Further processing steps may precede and/or follow, such as drying (e.g. freeze-drying, spray-drying and evaporation), granulation, agglomeration, concentrating (e.g. to syrups, formed via concentration and/or with the aid of thickeners), pasteurizing, sterilizing, freezing, dissolving, dispersing, filtering, centrifuging, confectioning, and the like.
When one or more compounds and/or a compound mixture according to the invention are added to a food product or pharmaceutical or nutraceutical, this also results in a functional food product or pharmaceutical or nutraceutical formulation according to the invention.
Further additives may be included, such as vitamins, minerals, e.g. in the form of mineral salts, unsaturated fatty acids or oils or fats comprising them, other extracts, or the like.
The functional food products according to the invention may be of any food type. They may comprise one or more common food ingredients in addition to the food product, such as flavours, fragrances, sugars, fruit, minerals, vitamins, stabilizers, thickeners, dietary fibers, protein, amino acids or the like in appropriate amounts, or mixtures of two or more thereof, in accordance with the desired type of food product.
Examples of basic food products and thus of functional food products according to the invention are fruit or juice products, such as orange and grapefruit, tropical fruits, banana, apple, peach, blackberry, cranberry, plum, prune, apricot, cherry, peer, strawberry, marionberry, black currant, red currant, tomato, vegetable, e.g. carrot, or blueberry juice, soy-based beverages, or concentrates thereof, respectively; lemonades; extracts, e.g. coffee, tea, green tea; dairy type products, such as milk, dairy spreads, quark, cheese, cream cheese, custards, puddings, mousses, milk type drinks and yoghurt; frozen confectionary products, such as ice-cream, frozen yoghurt, sorbet, ice milk, frozen custard, water-ices, granitas and frozen fruit purees; baked goods, such as bread, cakes, biscuits, cookies or crackers; spreads, e.g. margarine, butter, peanut butter honey; snacks, e.g. chocolate bars, muesli bars; pasta products or other cereal products, such as muesli; ready-to-serve-dishes; frozen food; tinned food; syrups; oils, such as salad oil; sauces, such as salad dressings, mayonnaise; fillings; dips; chewing gums; sherbet; spices; cooking salt; instant drink powders, such as instant coffee, instant tee or instant cocoa powder; instant powders e.g. for pudding or other desserts; meat fish or fish or meat products, such as sausages, burgers, meat loafs, meatballs, meat extracts, canned or tinned fish or meat, meat vol-au-vent, meat or fish soup, meat or fish skewers, fish fingers; or the like.
One or more other customary additives may be present, such as flavour, fragrances or other additives, such as one or more selected from stabilizers, e.g. thickeners; colouring agents, such as edible pigments or food dyes; bulking agents, such as fruit pulp, e.g. in dried form; polyols, such as xylitol, mannitol, maltitol or the like; preservatives, such as sodium or potassium benzoate, sodium or calcium carbonate or other food grade preservatives; antioxidants, such as ascorbic acid, carotinoids, tocopherols or polyphenols; mono-, oligo- or polysaccharides, such as glucose, fructose, sucrose, soy- oligosaccharides, xylo-oligosaccharides, galacto-oligosacharides; other artificial or natural
non- or low-caloric sweeteners, such as aspartame or acesulfame; bitterness blockers; acidifiers in the form of edible acids, such as citric acids, acetic acid, lactic acid, adipic acid; flavours, e.g. artificial or natural (e.g. botanical flavours); emulsifiers; thiols, e.g. allylic thiols; diluents, e.g. maltodextrose; wetting agents, e.g. glycerol; stabilizers; coatings; isotonic agents; absorption promoting or delaying agents; and/or the like.
The one or more compounds of formula (I) or compound mixtures thereof according to the invention can also be comprised in confectioned formulations to be added to foods including beverages, e.g. in the form of powders or granules, e.g. freeze-dried or spray- dried, concentrates, solutions, dispersions or other instant form, or the like.
In another aspect, the present invention relates to a composition, preferably a topically or orally administrable cosmetic or pharmaceutical composition, comprising
(a) an effective amount of one, two or more compounds of formula (I) and/or a physiologically acceptable salt thereof as defined above, or of a plant extract formulation as defined above, said effective amount being sufficient to reduce the lipid quantity contained in fat tissue, preferably subcutaneous fat tissue, and/or to inhibit the differentiation of preadipocytes, and/or to inhibit the lipogenesis in adipocytes, and preferably additionally one or more liquid or solid carrier substances.
The (particularly) preferred compounds of formula (I) of the present invention are preferably used in the preferred compositions indicated hereinbefore or hereinafter. In the context of the present invention an effective amount of (the preferred) compounds of formula (I) relates to a total amount of one, two or more compounds, preferably of the preferred compounds, of formula (I) sufficient to exhibit an activity in the above described Routes (i) and (ii) i.e. to influence one or more of said Routes in the desired way in the sense of the present invention.
Since the compounds of formula (I) do not stimulate lipolysis in adipocytes (Route (iii) as described above), it is many cases advantageous and preferred to combine the one or more compounds of formula (I) and/or the salts thereof (as defined above) with active ingredients to stimulate lipolysis (Route (iii)). Therefore, a preferred composition according to the present invention comprises one or more compounds selected from the following groups (b) and/or (c):
(b) one or more lipolysis stimulants, preferably selected from
(b-i) the group of phosphodiesterase inhibitors, and
(b-ii) the group of agonists of beta-adrenergic receptors, (c) one or more stimulators of the transport or oxidation of free fatty acids, preferably selected from
(c-i) the group of promoters of the transport of free fatty acids in the mitochondria, preferably coenzyme A, and
(c-ii) the group of stimulators of beta-oxidation, preferably L-carnitine. Preferably, the lipolysis stimulant is present in a quantity sufficient to stimulate lipolysis.
Preferably, the one or more stimulators of the transport or oxidation of free fatty acids are present in a quantity sufficient to stimulate the transport or oxidation of free fatty acids.
An advantageous preparation according to the invention additionally contains anti-cellulite active ingredients from group (b-i) of inhibitors of phosphodiesterase selected from the group of xanthines, preferably those described in US 2010/0285153 A1. Preferred xanthines are methyl xanthines, preferably selected from the group consisting of caffeine, theobromine and theophylline; the most preferred methyl xanthine in the sense of the present invention is caffeine. An alternatively preferred active ingredient from group (b-i) is aminophylline, a theophylline derivative. The xanthines or methyl xanthines may preferably be used as pure materials or in the form of plant extracts.
Also preferred compositions contain combinations of the compound of formula (I) with an agonist of beta-adrenergic receptors of adipocytes.
Preferred agonists of beta-adrenergic receptors are [beta]-phenylethylamines can preferably be used as pure substances, in the form of their respective hydrochlorides or in the form of plant extracts.
Preferred agonists of beta-adrenergic receptors are adrenaline, noradrenaline, metanephrine, macromerine, normacromerine, hordenine, N-methyltyramine, dopamine, octopamine, tyramine, 2-phenylethylamine, phenylethanolamine, epinine (N- methyldopamine), synephrine, ephedrine, pseudoephedrine, norephedrine and isoprenaline.
If synephrine is used, it is preferred to use synephrine-containing extracts, such as, for example, orange blossom extract.
The invention in one aspect of the present invention thus relates to (improved), preferably cosmetic, compositions comprising (b) one or more lipolysis stimulants, wherein one, a plurality or all of the lipolysis stimulants of component (b) are preferably selected from the group consisting of
(b-i) xanthines, preferably selected from the group consisting of caffeine, theobromine and theophylline, and
(b-ii) [beta]-phenylethylamines, preferably selected from the group consisting of adrenaline, noradrenaline, metanephrine, macromerine, normacromerine, hordenine, N- methyltyramine, dopamine, octopamine, tyramine, 2-phenylethylamine, phenylethanolamine, epinine (N-methyldopamine), synephrine, ephedrine, pseudoephedrine, norephedrine and isoprenaline.
In a preferred, preferably cosmetic and/or topical, composition according to the present invention
the total quantity of compounds of formula (I) is in the range of from 0.01 - 10 % by weight, preferably in the range of from 0.025 - 5 % by weight and particularly preferably in the range of from 0.05 - 3 % by weight and most preferably in the range of from 0.1 - 2 % by weight, and/or the total quantity of phosphodiesterase inhibitors (b-i) is in the range of from 0.005 - 10 % by weight, preferably in the range of from 0.05 - 5 % by weight and more preferably in the range of from 0.5 - 2.5 % by weight, and/or the total quantity of agonists of beta-adrenergic receptors (b-ii) is in the range of from 0.0001 - 0.10 % by weight, preferably in the range of from 0.001 - 0.05 % by weight and more preferably in the range of from 0.002 - 0.02 % by weight, in each case based on the total weight of the composition.
The pharmaceutical or nutraceutical formulation (= compositions) according to the present invention can be prepared in various forms, such as granules, tablets, pills, pellets, syrups, solutions, dispersions, suppositories, capsules, suspensions, salves, lotions and the like. Pharmaceutical grade or food grade organic or inorganic carriers and/or diluents suitable for oral and topical use can be used to formulate compositions containing the therapeutically-active compounds. Diluents known in the art include aqueous media, vegetable and animal oils and fats. Stabilizing agents, wetting and emulsifying agents, salts for varying the osmotic pressure or buffers for securing an adequate pH value, and skin penetration enhancers can be used as auxiliary agents. The compositions may also include one or more of the following: carrier proteins such as serum albumin; buffers; fillers such as microcrystalline cellulose, lactose, corn and other starches; binding agents; sweeteners and other flavouring or fragrancing agents; colouring agents; and polyethylene glycol. Those additives are well known in the art, and are used in a variety of formulations.
The compounds of formula (I) or a mixture of compounds of formula (I) or extracts comprising one or more compounds of formula (I) may be administered alone or in combination with other active agents, preferably on or more other anti-obesity agents. Among other anti-obesity agents that may be combined with one or more compounds of formula (I) or the salts thereof, antilipidemics, e.g. atorvastatin, cerivastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, simvastatin, anti-obesity drugs, such as suppressants of the appetite, stimulators of the body's metabolism, or drugs or compositions interfering with the body's ability to absorb specific nutrients, such as sibutramine, diethylpropion, phendimetrazine, phentermine, fenfluramine, lipase inhibitors, such as orlistat; anorectics, such as dexedrine; cannabinoid receptor antagonists, such as rimonabant; acarbose; or the like, can be mentioned. Other helpful drugs or active agents may be administered, e.g. psychoactive agents, agents that help in the treatment of addictive behaviour, e.g. nicotine addiction, or the like, especially in so far as they help to support the prophylaxis or treatment according to the invention intended.
Weight loss diets, such as food combining, Hay diet, Atkins diet (low-carbohydrate diet), cabbage soup diet, diabetic diet, fat resistance diet, slimming world diet, low-fat diet, Pritkin diet, low-carbohydrate diet, low protein diet, negative calorie diet, raw food diet, weight watchers diet are possible examples of appropriate diets.
The compounds of formula (I) or a mixture of compounds of formula (I) or an extract comprising one or more compounds of formula (I) of the present invention are particularly useful for controlling the body weight, preferably the treatment of obesity, adiposity, and/or further immune or inflammatory disorders.
Natural compounds of formula (I) or extracts comprising one or more of natural compounds of formula (I), for use according to the present invention are isolated from the plant or from parts of plants, preferably the roots, of Labisia pumila. The extracts used according to the invention may be obtained by using solvents of a certain polarity capable to extract one or more compounds of formula (I) from plant material.
The extraction leading to the extract can, for example, start from whole plants or plant parts (collectively referred to also a plant material) as defined above, e.g. from leaves or roots. The plant material may be used without prior treatment or after e.g. drying, chopping, milling or grinding or combinations of two or more thereof.
The extract may be prepared by any extraction method known in the art, however, the condition being that it is capable to extract one or more naturally occurring compounds of formula (I). Alternatively, also supercritical fluids, such as nitrous oxide, sulphur dioxide, fluorocarbons or especially carbon dioxide, with or without one or more organic modifiers, e.g. co- solvents such as methanol or ethanol, halogenated hydrocarbons such as chloroform, which allow adjusting the solvating power, can be used for extraction. In a further embodiment of the invention, the extraction can be followed by a further step for enrichment, e.g. solvent partition (e.g. of an extract filled up with water and partitioned between a hydrophilic phase, e.g. with water and/or one or more polar organic solvents, and a hydrophobic phase, e.g. an essentially apolar solvent forming a separate phase in the presence of water, e.g. an alkane, such as pentanes or hexanes, or an only weekly polar solvent, such as an ester, e.g. ethyl acetate, an ether, e.g. ethyl ether, or a halogenated hydrocarbons, such as methylene chloride, and/or by chromatography, e.g. preparative high performance chromatography.
Preferably, if organic solvents are used, the extract is further extracted after optional concentration e.g. by evaporation extracted with a solvent not miscible with water to remove impurities into the organic phase.
Other reprocessing is possible, e.g. by filtering (e.g. through paper, sintered glass, charcoal (also allowing for decoloration) or silica). The extraction can be made at lowered or elevated or ambient temperature, e.g. in the range from -20 °C to the boiling point of the solvent or solvent mixture employed, e.g. from 20 °C to said boiling point. The extraction may be improved by moving the solvent and/or the plant material, e.g. by stirring, or by ultrasound, or by milling and/or chopping
during extraction, or the like.
Plant extracts for use in accordance with the present invention can be prepared using plants or especially plant parts, in particular roots, of Labisia pumila.
Plant parts are, e.g. leaves, bark, flowers, buds, fruits, stems, shoots, roots or other parts of plants, and they or the plants can be complete, hackled, crushed, chopped, broken up, homogenized, dried, fermented or treated otherwise. The compounds of formula (I) or a mixture of compounds of formula (I) or an extract comprising one or more compounds of formula (I) of the present invention can be prepared by extracting and preferably enriching up to isolating them from the plants or parts of the plants. Auxiliary means such as (especially ultrasonic) sonication, temperature/heating (preferably at temperatures in the range from 20°C to 120 °C), stirring, re-extraction, evaporation or the like, may be used to allow for appropriate extraction, enrichment and purification.
Extraction preferably takes place with a polar solvent or solvent mixture, e.g. water and/or an alcohol, such as ethanol, and/or with a liquid or superfluid gas, especially superfluid C02.
A further embodiment of the invention relates to the use of a plant extract comprising one ore more compounds of formula (I) and/or physiologically acceptable salts thereof (as defined above), wherein the solvent for extraction is an aqueous solvent; yet another specific embodiment of this embodiment is where the solvent used for extraction essentially consists or consist of a mixture of water and a Ci-C8-alkanol with one or more hydroxy groups, preferably a C C8-alkanol, a (VCe-alkandiol or a Ci-C8-alkantriol, such as ethanol/water, isopropanol/water, monopropylene glycol/water or glycerol/water, wherein the total vol/vol ratio (v/v) of said alkanol or alkanols with one or more hydroxy groups to water is in the range from 3 : 97 to 98 : 2, preferably from 25 : 75 to 96 : 4.
The extraction of the plant or a part of a plant preferably is carried out with a mixture of water and an alcohol having 1 to 3 carbon atoms, preferably with a mixture of ethanol and
water, wherein the volume ratio (v/v) of said alcohol : water in the range of 1 : 2 to 30 : 1 , more preferably in the range of 1 : 1 to 25 : 1 , most preferably in the range of 3 : 2 to 20 : 1 , preferably at a temperature below 75 °C, more preferably in the range from 15 to 60 °C, even preferably in the range from 20 to 50 °C. The weight ratio of (preferably dried) plant material, preferably roots, to the total amount of aqueous solvent used in the extraction preferably is in the range from 2 : 1 to 1 : 4, more preferably in the range from 1 : 1 to 1 : 3, even more preferably in the range from 1 : 1 to 1 : 2.
The following method for producing a preferred plant extract for use in accordance with the present invention comprises the following steps: providing one or more roots of a plant or a part of a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, and optionally drying the plant or a part of a plant of step (i), extraction of said plant or a part of a plant provided in step (i) with a mixture of water and an alcohol having 1 to 3 carbon atoms, preferably with a mixture of ethanol and water, wherein the volume ratio (v/v) of said alcohol : water in the range of 1 : 2 to 30 : 1 , more preferably in the range of 1 : 1 to 25 : 1 , most preferably in the range of 3 : 2 to 20 : 1 , preferably at a temperature below 75 °C, more preferably in the range from 15 to 60 °C, even preferably in the range from 20 to 50 °C, optionally partially or fully removing the alcohol having 1 to 3 carbon atoms of step (ii), preferably at a temperature below 70 °C, more preferably in the range from 15 to 55 °C, optionally drying the mixture obtained in step (ii) or (ii-a), preferably by spray- drying or freeze-drying.
Preferably, the extracts can subsequently be further enriched by one or more additional purification steps, such as distribution, precipitation (e.g. crystallization) or especially
chromatography, by which it is possible to obtain further enriched extracts or isolated compounds of formula (I).
In another aspect, the present invention relates to a plant extract formulation, preferably in solid form, obtainable from a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, particularly preferably from the roots of Labisia pumila, comprising
(a) one or more of the particularly preferred compounds of formula (I) as defined above or a physiologically acceptable salt thereof in a total amount effective, preferably the total amount being in the range from 0.1 to 15 wt.%, more preferably from 0.2 to 10 wt.%, particularly preferably from 0.5 to 6 wt.%, most preferably from 1 to 5 wt.%, most preferably from 1.25 to 4 wt.%, based on the total weight of the plant extract formulation,
(b) one or more further substances naturally occurring in said plant from, wherein said one or more further substances are not particularly preferred compounds of formula (I) as defined for constituent (a) or a physiologically acceptable salt thereof, and
(c) one or more solid carrier substances, preferably one or more solid carrier substances selected from the group consisting of maltodextrins, silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stearate salts, preferably magnesium stearate, alginates, tragacanth, gelatins, calcium silicates, cellulose and cellulose derivatives, preferably microcrystalline cellulose or methyl cellulose, polyvinylpyrrolidones, and propylhydroxybenzoates.
In a further aspect, the present invention relates to a method for producing a plant extract formulation according to the present invention, comprising the following steps: (i) providing a plant or a part of a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, particularly preferably from the roots of Labisia pumila,
(i-a) optionally drying the plant or a part of a plant of step (i),
(ii) extraction of said plant or a part of a plant provided in step (i) or (i-a) with a mixture of water and an alcohol having 1 to 3 carbon atoms, preferably with a mixture of ethanol and water, wherein the volume ratio (v/v) of said alcohol : water in the range of 1 : 2 to 30 : 1 , more preferably in the range of 1 : 1 to 25 : 1 , most preferably in the range of 3 : 2 to 20 : 1 ,
(ii-a) optionally partially or fully removing the alcohol having 1 to 3 carbon atoms of step
(ii) , preferably at a temperature below 70 °C, more preferably in the range from 15 to 55
°C,
(iii) mixing the extract obtained in step (ii) or (ii-a) with one or more solid carrier substances, preferably one or more solid carrier substances selected from the group consisting of maltodextrins, silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stearate salts, preferably magnesium stearate, alginates, tragacanth, gelatins, calcium silicates, cellulose and cellulose derivatives, preferably microcrystalline cellulose or methyl cellulose, polyvinylpyrrolidones, and propylhydroxybenzoates,
(iv) optionally drying the mixture obtained in step (iii), preferably by spray-drying or freeze-drying.
Several compounds of formula (I) can be obtained from natural sources. The method for isolation and detection can comprise high pressure liquid chromatography (HPLC) on reversed phase silica gel (C18) with water/acetonitrile-gradient as an elution solvent with UV as well as MS detection which are used for the product analysis and production optimization. It is evident to those having ordinary skill in this art that compounds of formula (I) can be synthesized according to standard methods leading to compounds identical with the naturally occurring compounds according to appropriate methods (as mentioned above).
In a preferred embodiment according to the present invention, a mixture of compounds of formula (I), plant extract, plant extract formulation or (cosmetic or pharmaceutical) composition comprises the three substances of formula (I) (as defined above)
and preferably additionally one, two or all of the following compounds:
Where "use" is mentioned, this especially refers to one or more of the following embodiments of the invention which can be inserted wherever use is mentioned:
(1) A compound of formula (I), or a mixture of compounds of formula (I), or especially a (preferably further enriched) extract comprising one or more compounds of formula (I), for use in therapeutic (including prophylactic) treatment of a mammal, especially a human, for the reduction of body weight and/or fat loss and/or for the management of obesity, especially for decreasing the body weight, more especially for decreasing the body fat; or simply for maintenance of a healthy body, e.g. a low body mass index.
(2) A pharmaceutical or nutraceutical composition comprising a compound of formula (I), or a mixture of compounds of formula (I), or especially a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient together with a
pharmaceutically acceptable diluent or carrier, especially for use in the therapeutic and/or prophylactic treatment mentioned under (1).
(2') A pharmaceutical or nutraceutical composition for the treatment as mentioned under (1) comprising a compound of formula (I), or a mixture of compounds of formula (I), or especially a (preferably further enriched) extract comprising one or more compounds of formula (I), and a pharmaceutically acceptable diluent or carrier, as active ingredient supplement to a food.
(3) A functional food comprising a compound of formula (I), or a mixture of compounds of formula (I), or especially a (preferably further enriched) extract, as active ingredient for the treatment as mentioned under (1 ).
(4) A method for the treatment as mentioned under (1), especially any one or more of obesity, and/or excess body fat, in a subject in need of such treatment, comprising administering a pharmaceutically or nutraceutically effective amount of a compound of formula (I), a mixture of compounds of formula (I), or a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient, especially to an individual in need thereof.
(5) The use of a compound of formula (I), or a mixture of compounds of formula (I), or a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient for the manufacture of a medicament or nutraceutical or food supplement for the treatment mentioned under (1 ).
(6) A method or use as defined under (4), comprising co-administration, e.g. concomitantly or in sequence, of a therapeutically effective amount of compound of formula (I), or a mixture of compounds of formula (I), or a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient and a different pharmaceutically active compound and/or a pharmaceutically acceptable salt thereof, said different pharmaceutically active compound and/or salt thereof being especially for use in the treatment as mentioned under (1).
(7) A combination product comprising a therapeutically effective amount of a compound of formula (I), or a mixture of compounds of formula (I), or a (preferably further enriched) extract comprising one or more compounds of formula (I), as active ingredient, and a different pharmaceutically active compound and/or a pharmaceutically acceptable salt thereof, said second pharmaceutically active compound being especially for use or of use in the treatment mentioned under (1).
For any of the uses and methods described in the present invention, the use or method is such that the compound(s) of formula (I) and/or salts thereof (as defined herein) or mixtures thereof are the active ingredient, i.e. they are (alone or in admixture with other known active ingredients) capable of achieving the intended effects and are thus themselves the important active principle for the treatment(s) mentioned. Throughout the present specification, the prophylactic and/or therapeutic treatment or regulation of body weight and/or fat loss and/or management of obesity, especially decrease (reduction) of body weight, more especially decrease (reduction) of body fat, are especially preferred embodiments according to the invention.
The pharmaceutical or nutraceutical preparations may be sterilized and/or may contain carrier materials or adjuvants such as preservatives, stabilizers, binders, disintegrants, wetting agents, skin or mucuous membrane penetration enhancers, emulsifiers, salts for varying the osmotic pressure and/or buffers, or other ingredients, excipients or carrier materials known in the art.
The compounds can be administered by a variety of routes including oral, rectal, topical, transdermal, subcutaneous, intravenous, intramuscular, and intranasal, preferably they are administered orally or topically.
The compounds of the present invention are preferably formulated prior to administration. Therefore, another embodiment of the present invention is a pharmaceutical formulation comprising an effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable carrier, diluent or excipient therefor.
By physiologically, preferably cosmetically, pharmaceutically and/or nutraceutically, acceptable it is meant that the carrier, diluent or excipient is compatible with the other ingredients of the formulation and not be deleterious to the recipient thereof.
The present pharmaceutical formulations are prepared by known procedures using well known and readily available ingredients. In making the compositions of the present
invention, the active ingredient will usually be admixed with a carrier, or diluted by a carrier, or enclosed within a carrier which may be in the form of a capsule, sachet, paper or other container. When the carrier serves as a diluent, it may be a solid, semi-solid or liquid material which acts as a vehicle, excipient or medium for the active ingredient. The compositions according to the present invention can be in the form of tablets, pills, powders, lozenges, sachets, cachets, elixirs, suspensions, emulsions, solutions, syrups, aerosols, (as a solid or in a liquid medium), ointments containing, for example, up to 10% by weight of the active compound, soft and hard gelatin capsules, suppositories, sterile injectable solutions, sterile packaged powders, and the like.
Examples of suitable carriers, excipients, and diluents are lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, alginates, tragacanth, gelatin, calcium silicate, microcrystalline cellulose, polyvinylpyrrolidone, cellulose, water syrup, methyl cellulose, methylhydroxybenzoates, propylhydroxybenzoates, talc, magnesium stearate and mineral oil. The formulations may additionally include lubricating agents, wetting agents, sweetening agents, flavoring agents, and the like. The compositions of the invention may be formulated so as to provide quick, sustained or delayed release of the active ingredient after administration to the patient by employing procedures well known in the art.
In a preferred embodiment, the compositions are preferably formulated in a unit dosage from, each dosage generally containing form 0.1 to 500 mg, and preferably from 0.25 to 300 mg, more preferably from 0.5 to 200 mg of the compound of formula (I). The term "unit dosage form" refers to physically discrete units suitable as unitary dosages for human subjects and other mammals, each unit containing a predetermined quantity of active material calculated to produce the desired therapeutic effect, in association with a suitable pharmaceutical carrier.
The effects of the compounds of formula (I) and/or their salts (as defined above) may be explained by a modulation of adipocyte triglyceride accumulation and in particular have an inhibitory effect on adipocyte differentiation. Therefore the extracts of the invention are useful in the modulation of triglyceride incorporation in adipocytes and thus can be used for modulation of body fat content in mammals (including humans) including the skin of
said mammals.
In a preferred embodiment, the invention relates to the use of an extract from Labisia pumila for reducing the body fat composition of a mammal (preferably a human), comprising modulation of adipocyte differentiation by applying said composition to individuals orally and/or topically.
In another aspect, the invention relates to the use of one or more compounds of formula (I) (as defined above) in the reduction of body fat content, thereby resulting in body weight management, in particular body weight reduction.
In yet another aspect, the invention relates to the use of one or more compounds of formula (I) and/or the physiologically acceptable salts thereof (as defined above) - based on the modulation of adipocyte differentiation - for skin slimming, in particular for dermal slimming, and/or for improving the appearance of skin.
In yet a further aspect, the invention relates to the use of one or more compounds of formula (I) and/or the physiologically acceptable salts thereof (as defined above), wherein the improving of the cosmetic appearance is (= manifests itself in) a more smooth, supple and/or soft appearance of skin in case of touch.
A further aspect of the invention relates to the use of one or more compounds of formula (I) and/or the physiologically acceptable salts thereof (as defined above), where the improving of the cosmetic appearance is a more juvenile appearance of the skin. The compounds of formula (I) can easily be incorporated in these concentrations in common cosmetic or dermatological formulations such as pump sprays, aerosol sprays, creams, ointments, tinctures, lotions and the like.
Another aspect of the invention refers to a corresponding use wherein the dermatologic composition is an oil, an ointments, a tincture, a cream, a gel, a solution, a lotion, a spray, a powder, a shampoo, a soap, a cosmetic pencil or a perfume. A more specific embodiment here relates to the case where the dermatologic composition is an ointment, a cream, a gel or a solution.
The following definitions of more general terms, as well as definitions given already above, can replace the more general terms in any of the embodiments of the invention, in any combination, thus leading to more specific embodiments all of which are also comprised by the present invention.
The "body fat content" can preferably be determined with a body fat analysis (BFA) using dual-energy X-ray absorptiometry (DEXA) using a four-compartment model that divides the body into water, bone mineral mass, fat tissue mass and residual fat-free soft (lean) mass). In this method X-rays of two different energies are used to scan the body, one of which is absorbed more strongly by fat than the other. A computer can subtract one image from the other, and the difference indicates the amount of fat relative to other tissues at each point. A sum over the entire image enables calculation of the overall body composition.
For example, the GE Lunar Piximus2 DEXA scanner gives accurate information on differences in body composition. One scanning procedure takes up to five minutes per individual and provides data on fat mass, lean mass, bone mineral content and bone mineral density.
The amount of subcutaneous fat in a subject can be measured using any method available for the measurement of subcutaneous fat. Methods of measuring subcutaneous fat are known in the art, for example, those described in US 6,530,886, incorporated herein by reference.
The compounds of formula (I) also show a pronounced effect in the treatment of cellulite, measurable by means of echographic determination of the subcutis layer thickness, in particular to prevent the increased formation of fat stores in the skin and/or cellulite, in that the lipid content in the human subcutaneous fat tissue is reduced. The invention therefore relates to cosmetic compositions (preparations), containing a corresponding effective quantity of one or more compounds of formula (I), in particular for the topical treatment and prevention of increased formation of fat stores in the skin and/or cellulite.
The topic dermatologic (cosmetic) compositions according to the invention are for external
application on the skin of humans.
For topical administration to the skin, the compositions according to the present invention preferably are provided as ointments, tinctures, creams, gels, solutions, lotions, sprays, aerosols, dry powders, suspensions or the like, preferably in the form of an ointments, gels, creams, lotions, microemulsions, nanoemulsions, o/w-emulsions, w/o-emulsions or multiple emulsions.
A composition according to the present invention, preferably for topical administration to the skin, can be prepared by mixing one or more compounds of formula (I) and/or the salts thereif (as defined above), preferably in the form of a plant extract or a plant extract formulation (as defined above) with one or more non-toxic solid or liquid carriers customarily used in such compositions. These compositions may comprise from 5 to 99 wt.%, preferably from 20 to 98 wt.%, more preferably from 30 to 96 wt.% of one or more carriers, based on the total weight of the composition.
In preparing the topical compositions described above, customary additives such as preservatives, thickeners, perfumes and the like used in the art of cosmetic compounding of topical preparation can be used. Cream-base cosmetic compositions containing the active agent, used in accordance with this invention, can be composed of aqueous emulsions containing a fatty acid alcohol, semi-solid petroleum hydrocarbon, ethylene glycol and an emulsifying agent.
Ointment formulations comprising the extract in accordance with this invention, for example, can comprise admixtures of a semi-solid petroleum hydrocarbon or fats with a solvent dispersion of the active material. Cream compositions comprising the extract for use in this invention can, for example, comprise emulsions formed from a water phase, e.g. using a humectant, a viscosity stabilizer and water, an oil phase of a fatty acid alcohol, a semi-solid petroleum hydrocarbon and an emulsifying agent and a phase containing the active agent dispersed in a aqueous stabilizer-buffer solution. Stabilizers may be added to the topical composition. Any conventional stabilizer can be utilized in accordance with this invention. For example, fatty acid alcohol components function as a stabilizer. These fatty acid alcohol components are derived from the reduction of a long-
chain saturated fatty acid containing at least 14 carbon atoms. Alternatively gels can be used utilising standard gel carriers.
One or more further additives may be present in the compositions used according to the invention, e.g. preservatives, bactericides, substances impeding foaming, colorants, e.g. dyes or pigments, thickeners (e.g. silica, aluminium silicates, polysaccharides or their derivatives, e.g. xanthane or guar gum or hydroxypropylmethylcellulose), moisturizers (including urea, lactic acid, pyrrolidone carbonic acid, hyluronic acid, polysaccharides rich in fucose, and/or glycerol), fats, oils, waxes or other customary additives, such as polyols, alcohols, polymers, foam stabilizers, electrolytes, other organic solvents, silicones (e.g. cyclomethicone, hexamethylcyclotrisiloxane, polydimethylsiloxane, poly(methylphenyl- siloxane)), perfumes, UV protectors, such as metal oxides which may also be coated, e.g. Ti02, or organic UV absorbers, such as methylene bis-benztriazolyl tetramethylbutylphenol or 4-(tert.-butyl)-4'-methoxydibenzoylmethane) and the like.
Among the antioxidants, conventional antioxidants or mixtures of conventional antioxidants can be incorporated into the topical preparations containing the aforementioned active agent. Among the conventional antioxidants which can be utilized in these preparations are included N-methyl-a-tocopherolamine, tocopherols, butylated hydroxyanisole, butylated hydroxytoluene, ethoxyquin, vitamin C, ascorbyl palmitate, vitamin A and the like.
As the occurrence of cellulite, in addition to an increased storage of fat in the fat tissue, is generally also accompanied by a breakdown of the connective tissue, preferred cosmetic preparations according to the invention containing one or more compounds of formula (I) preferably also contain active ingredients which prevent a breakdown of the connective tissue. Such preparations show improved efficacy in the prophylaxis and cosmetic treatment of cellulite.
Active ingredients are advantageous here which inhibit matrix-metallo-proteinases (MMPs). Such preparations are particularly effective in the prophylaxis and cosmetic treatment of cellulite. These enzymes are in a position to break down macromolecules of the extra-cellular matrix (ECM) / of the connective tissue, also including the collagens, proteolytically. In particular the matrix-metallo-proteinase-1 (MMP-1), matrix-
metalloproteinase-2 (MMP-2) and matrix-metallo-proteinase-9 (MMP-9) are responsible for the breakdown of the connective tissue of the skin. An inhibition of MMPs is possible, for example, by the addition of ursolic acid, retinyl palmitate, propyl gallate, precocenes, 6- hydroxy-7-methoxy-2,2-dimethyl-1 (2H)-benzopyran, 3,4-dihydro-6-hydroxy-7-methoxy- 2,2-dimethyl-1 (2H)-benzopyran. An addition of peptides, which inhibit MMPs, to preparations according to the invention, is also advantageous to inhibit MMPs. Proteins or glycoproteins from soya and hydrolysed proteins from rice, pea or lupine also inhibit MMPs and are therefore a suitable addition. A combination with a plant extract, which inhibits MMPs is also advantageous. To be mentioned here by way of example is an extract from shitake mushrooms. The combination with extracts from the leaves of the Rosaceae family, sub-family Rosoideae, is also advantageous.
MMP inhibitors to be preferably used in combination in the scope of the present invention are retinyl palmitate, propyl gallate, precocenes, 6-hydroxy-7-methoxy-2,2-dimethy-1 (2H)- benzopyran, 3,4-dihydro-6-hydroxy-7-methoxy-2,2-dimethyl-1 (2H)-benzopyran, benzamidine Oenothera biennis root extract, ellagic acid and ellagitannins (for example from pomegranate), soya seed extract, ursolic acid, soya isoflavones, sage extracts, lentinus edodes extracts, and extracts from green and/or black tea.
In order to counteract the breakdown of the connective tissue, the combination of active ingredients, which encourage the formation of collagen in the tissue (collagen synthesis stimulators, collagen stimulants), is furthermore advantageous in preferred cosmetic preparations according to the invention containing one or more compounds of formula (I). Such preparations are particularly effective in the prophylaxis and cosmetic treatment of cellulite. Individual substances frequently used to increase collagen synthesis are, for example, ingredients such as ascorbic acid and their derivatives, retinol and derivatives of retinol or plant extracts such as, for example, extracts of aloe and centella species. Moreover peptidic materials and their derivatives, such as, for example, carnitine, carnosine, creatine, matrikine peptides (e.g. lysyl-threonyl-threonyl-lysyl-serine) and further peptidic structures such as palmitoylated pentapeptides are also included in the frequently used active ingredients increasing collagen synthesis. Furthermore, compounds such as asiatic acid, madecassic acid, madecassoside, asiaticoside, extracts of Centella asiatica, niacinamide, astaxanthine, glucans, for example from yeast and oats, soya extracts and soya isoflavones such as genistein and daidzein, rutin, chrysin, morin,
betel nut alkaloids, forskolin, betulinic acid, extracts of Plantago species, TGF-beta, extracts from Ginkgo biloba, glutamine and glycolic acid are also used as collagen synthesis stimulators. Particularly preferred here is the addition of a combination of aloe vera extract, raspberry extract and magnesium ascorbyl phosphate. Thus, further preferred cosmetic or pharmaceutical, preferably topical, preparations according to the invention containing one or more compounds of formula (I) further additionally comprise one or more matrix-metalloproteinase inhibitors, and/or one or more collagen synthesis stimulators.
Such preparations are particularly effective in the prophylaxis and cosmetic treatment of cellulite.
The (particularly) preferred aspects and embodiments mentioned hereinbefore or hereinafter relating to compounds of formula (I) or compositions (preparations) comprising one or more compounds of formula (I) according to the present invention also apply to (particularly) preferred aspects and embodiments, uses and methods in accordance with the present invention.
Preferably, a method comprises the step of topical application onto the skin, in particular on the thighs (in particular the outer side and the back of the thighs) and/or the buttocks, of a human, preferably a woman.
The present invention further relates to a pharmaceutical composition comprising a pharmaceutically active amount of one or more compounds of formula (I) as defined herein, preferably for preventing, treating or reducing cellulite. Further, the present invention also relates to a method of treatment of cellulite, comprising the following step: application, preferably topical application, of an effective amount of a compound of formula (I) or a cosmetically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the salts thereof as defined herein or of a cosmtic composition as defined herein.
In particular, the present invention relates to a method for the non-therapeutic, preferably cosmetic,
(i) prevention, treatment or reduction of cellulite, and/or
(ii) reduction of the lipid quantity contained in fat tissue, preferably in subcutaneous fat tissue and/or inhibition of the differentiation of preadipocytes, and/or inhibition of the lipogenesis in adipocytes, and/or
(iii) modulation of adipocyte differentiation, comprising the following step: application of an effective amount of a compound of formula (I) or a physiologically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the salts thereof as defined hereinbefore, a plant extract formulation as defined above or a cosmetic composition as defined above.
In particular, the present invention relates to a compound of formula (I) or a pharmaceutically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the pharmaceutically acceptable salts thereof as defined hereinbefore, a plant extract formulation as defined above, or a composition as defined above, for use in a prophylactic or therapeutic method for treating adiposity (obesity).
In particular, the present invention relates to a pharmaceutical composition comprising an effective amount of one or more compounds of formula (I) as defined above, or a plant extract formulation as defined above, for use in a therapeutic method for preventing and/or treating obesity.
The present invention also relates to a method for the therapeutic reduction of the body fat content or the therapeutic treatment of obesity, comprising administering, preferably orally, an effective amount of one or more compounds of formula (I) as defined
hereinbefore, a plant extract formulation as defined above or a composition as defined above, said effective amount being sufficient to
(i) reduce the body fat content, and/or
(ii) reduce the lipid quantity contained in fat tissue, and/or (iii) inhibit the differentiation of preadipocytes, and/or inhibit of the lipogenesis in adipocytes, and/or (iv) modulate the adipocyte differentiation.
Substances and auxiliaries which a composition according to the invention containing one or more compounds of formula (I) may additionally contain are selected from the following group: preservatives, in particular those described in US 2006/0089413, antimicrobial agents, such as e.g. antibacterial agents or agents to treat yeast and mold, in particular those described in WO 2005/123101 , antiacne and sebum reducing agents, in particular those described in WO 2008/046791 , compounds against ageing of the skin, in particular those described in WO 2005/123101 , further anti-cellulite agents, in particular those described in WO 2007/077541 , antidandruff agents, in particular those described in WO 2008/046795, antiirritants (antiinflammatory agents, irritation-preventing agents, irritation- inhibiting agents), in particular those described in WO 2007/042472 and US 2006/0089413, antioxidants, in particular those described in WO 2005/123101 , carrier materials, in particular those described in WO 2005/123101 , chelating agents, in particular those described in WO 2005/123101 , deodorizing agents and antiperspirants, in particular those described in WO 2005/123101 , moisture regulators (moisture-donating agents, moisturizing substance, moisture-retaining substances), in particular those described in WO 2005/123101 , osmolytes, in particular those described in WO 2005/123101 , compatible solutes, in particular those described in WO 01/76572 and WO 02/15868, proteins and protein hydrolysates, in particular those described in WO 2005/123101 and WO 2008/46676, skin-lightening agents, in particular those described in WO 2007/110415, skin-tanning agents, in particular those described in WO 2006/045760, cooling agents, in particular those described in WO 2005/123101 , skin-cooling agents, in particular those described in WO 2005/123101 , skin warming agents, in particular those
described in WO 2005/123101 , UV-absorbing agents, in particular those described in WO 2005/123101 , UV filters, in particular those described in WO 2005/123101 , insect repellents, in particular those described in WO 2005/123101 , plant parts, plant extracts, in particular those described in WO 2005/123101 , vitamins, in particular those described in WO 2005/123101 , emulsifiers, in particular those described in WO 2005/123101 , gelling agents, in particular those described in WO 2005/123101 , oils in particular those described in WO 2005/123101 , waxes in particular those described in WO 2005/123101 , fats in particular those described in WO 2005/123101 , phospholipids, in particular those described in WO 2005/123101 , saturated fatty acids and mono- or polyunsaturated fatty acids and [alpha]-hydroxy acids and polyhydroxy-fatty acids and esters of saturated and/or unsaturated branched and/or unbranched alkane carboxylic acids, in particular those described in WO 2005/123101 , surface-active substances (surfactants) in particular those described in WO 2005/123101 , skin repair agents comprising cholesterol and/or fatty acids and/or ceramides and/or pseudoceramides, in particular those described in WO 2006/053912, dyestuffs and colorants and pigments, in particular those described in WO 2005/123101 , aroma chemicals and flavors and fragrances, in particular those described in S. Arctander, Perfume and Flavor Chemicals, private publishing house, Montclair, N.J., 1969 and Surburg, Panten, Common Fragrance and Flavor Materials, 5th Edition, Wiley- VCH, Weinheim 2006, preferably those explicitly mentioned in US 2008/0070825, alcohols and polyols, in particular those described in WO 2005/123101 , organic solvents, in particular those described in WO 2005/123101 , silicones and silicone oils and silicone derivatives in particular those described in WO 2008/046676, virucides, abrasives, astringents, antiseptic agents, antistatics, binders, buffers, cell stimulants, cleansing agents, care agents, depilatory agents, softeners, enzymes, essential oils, in particular those described in US 2008/0070825, fibres, film-forming agents, fixatives, foam-forming agents, foam stabilizers, substances for preventing foaming, foam boosters, gel-forming agents, hair growth activators, hair growth inhibitors, hair care agents, hair-setting agents, hair-straightening agents, hair-smoothening, bleaching agents, strengthening agents, stain-removing agents, optieaHy brightening agents, impregnating agents, dirt-repeHent agents, friction-reducing agents, lubricants, opacifying agents, plasticizing agents, covering agents, polish, gloss agents, polymers in particular those described in WO 2008/046676, powders, peptides, mono-, di- and oligosaccharides, re-oiling agents, abrading agents, skin-soothing agents, skin-cleansing agents, skin care agents, skin-
healing agents, skin-protecting agents, skin-softening agents, skin-smoothing agents, nourishing agents, skin-warming agents, stabilizers, detergents, fabric conditioning agents, suspending agents, thickeners, yeast extracts, algae or microalgae extracts, animal extracts, liquefiers, color-protecting agents, and electrolytes.
Preferred liquid carrier substances, which may be a component of a preparation according to the invention are selected from the group consisting of glycerol, 1 ,2-propylene glycol, 1 ,2-butylene glycol, 1 ,3-butylene glycol, 1 ,2-pentanediol, 1 ,5-pentanediol, 1 ,2-hexanediol, 1 ,6-hexanediol, 1 ,2-octanediol, 1 ,2-decanediol, ethanol, water and mixtures of two or more of said liquid carrier materials with water. Optionally, these preparations according to the invention may be produced using preservatives, solubilizers or antioxidants.
Skin care agents, preferably skin moisture retention regulators or skin repair agents, preferably are selected from the group consisting of sodium lactate, urea and derivatives, glycerol, 1 ,2-pentanediol, collagen, elastin or hyaluronic acid, diacyl adipates, petrolatum, urocanic acid, lecithin, allantoin, panthenol, phytantriol, lycopene, (pseudo-)ceramides [preferably Ceramide 2, hydroxypropyl bispalmitamide MEA, cetyloxypropyl glyceryl methoxypropyl myristamide, N-(1-hexadecanoyl)-4-hydroxy-L-proline (1-hexadecyl) ester, hydroxyethyl palmityl oxy hydroxy propyl palmitamide], glycosphingolipids, cholesterol, phytosterols, chitosan, chondroitin sulfate, lanolin, lanolin esters, amino acids, vitamin E and derivatives (preferably tocopherol, tocopheryl acetate), alpha-hydroxy acids (preferably citric acid, lactic acid, malic acid) and derivatives thereof, mono-, di- and oligosaccharides, preferably glucose, galactose, fructose, mannose, laevulose and lactose, polysugars, such as [beta]-glucans, in particular 1 ,3-1 ,4-[beta]-glucan from oats, alpha-hydroxy-fatty acids, triterpenic acids, such as betulic acid or ursolic acid, and algae extracts, preferably selected from the group consisting of glycerol, 1 ,2-pentanediol, urea, hyaluronic acid, allantoin, panthenol, lanolin, alpha-hydroxy acids (preferably citric acid, lactic acid), vitamin E and derivatives (preferably tocopherol, tocopheryl acetate).
Important areas of application for the preparations according to the invention are cosmetic, in particular dermatological preparations, which are composed as conventional (apart from the compound(s) of formula (I)) and are used for cosmetic, in particular dermatological light protection, for treatment, care and cleaning of the skin and/or hair or
as a make-up product in decorative cosmetics. Accordingly, preparations of this type, depending on their structure, can be used, for example, as day protection cream, day or night cream, eye cream, sun protection or after-sun lotion, nourishing cream, a care mask, gel pads, facial tonic, moist care and cleaning tissues, cleaning milk, cleaning soap, foam or shower bath, deodorant, antiperspirant, hair shampoo, hair care agent, hair conditioner, hair colorant, hair styling agent and in this case preferably be present as an emulsion, lotion, milk, fluid, cream, hydro dispersion gel, balm, spray, alcoholic or aqueous/alcoholic solution, foam, powder, liquid soap, piece of soap, shampoo, roll-on, stick or make-up. In hair treatment agents, the use is preferably directed at the base of the hair or the scalp. The present invention is further explained by the following examples. The specific examples which follow illustrate the methods in which the compositions of the present invention may be prepared, components therein and their use, as well as other embodiments of the invention, but are not to be construed as limiting the invention in scope.
Examples
Example 1 : Extraction and Extracts Leaves and roots from Labisia pumila var. alata (obtained from plants harvested in Malaysia) were used in the following experiments.
Method A: Hot water extraction
20 g of either dry leaves or dry roots were ground into a powder using a lab mill and afterwards extracted in a stirred suspension with 300 ml water for 30 min. at 90 °C. Evaporating water was condensed and refluxed. The suspension was filtered and the filtrate was evaporated to dryness under reduced pressure at a maximum temperature of 40° C. Method B: Extraction using organic solvents
20 g of either dried leaves or dried roots were ground into a powder using a lab mill and subsequently extracted with 300 ml of either 70 vol.% ethanol in water or 95 vol.% ethanol in water for 30 min. at 40°C by using ultrasonication.
The suspension was filtered and the filtrate was evaporated to dryness under reduced pressure at a maximum temperature of 40°C. Table 1 : Extracts prepared according methods A and B, conditions of preparation and yields
Of these extracts E-1 to E-6, extract E-6 contained the highest amount of compounds of formula (I). It should be mentioned that the total amount of compounds of formula (I) (as defined above) identified in the respective extracts E-3 to E-6, based on the total amount of extract obtained, was comparatively small (see also Table 2 of example 2.2).
Example 2: Extracts and pure compound isolated from roots of Labisia oumila
Example 2.1 : Preparation of the crude extract and liquid -liquid enrichment
1200 g of Labisia pumila roots (BTP0056) were ground into a powder using a lab mill and subsequently extracted at 25°C with 2000 ml aqueous ethanol (ethanol : water = 95 : 5 (v/v)) twice by using ultrasonication. The resulting solution was separated from the remaining plant material and the ethanol removed under reduced pressure. To the remaining water phase additional water was added to a final volume of 400 ml and subsequently extracted with n-heptane and ethyl acetate by liquid / liquid separation. The n-heptane extract (BTP0056-5-1) was dried (Na2S04) and the solvent evaporated under reduced pressure. The remaining water phase was extracted three times with ethyl acetate. The combined three ethyl acetate extracts (BTP0056-5-2) were dried (Na2S04) and the solvent evaporated under reduced pressure. The remaining water phase (BTP0056-5-3) was also evaporated under reduced pressure and the amounts for the three crude extracts were determined:
Example 2.2 Isolation of pure compounds
Extract BTP0056-5-2 was selected as starting material for isolating pure compounds. The initial separation steps were performed as MPLC separations on reverse phase material in multigram scale allowing separating the most part of the matrix from the analytically detectable compounds. For the separation of the single compounds in preparative scale a HPLC-setup was used comprising reverse phase separation columns with capacity for up to 200 mg material per separation. The gradients for elution were chosen according to the separation problem. Generally the systems were based on water / acetonitrile mixtures. Case by case the final separation step for the isolation of pure compounds was performed on a PS1 column (Molecular filtration Chromatography, column provided by Merck,
Darmstadt, Germany) using acetonitrile as solvent under isocratic conditions. Every fraction was dried by using a vacuum concentrator and the yield was determined.
Structure elucidation:
(2)-4-hydroxy-2-methoxy-6-(pentadec-10-en-1 -y l)pheny I acetate
Atom 13C mult. (J in [Hz])
(DMSO-cf6, (DMSO-d6, 500 MHz)
125 MHz)
1 130.1
2 151.4 —
3 98.0 6.31 , d (1.5)
4 155.4 —
5 106.9 6.18, d (1.5)
6 135.4 — r 29.5 2.30, t (7.5)
2' 29.4 1.41 , m
3' - 7' 28.6-28.9 1.23, m
8' 29.1 1.28, m
9' 26.6 1.98, m
io' - ir 129.6 5.32, m
12' 26.3 1.99, m
13' 31.4 1.28, m
14' 21.7 1.28, m
15' 13.8 0.85, t (6.8)
1 " 168.8
2" 20.2 2.20, s
OCH3 55.5 3.66, s
4-OH 9.32, br s +: m/z = 391.2843)
7,7'-(cyclopropane-1 ,2-diyl)bis(1-(3,5-dihydroxy-2-methoxyphenyl)heptan-1-one)
Atom 13C Ή, mult. (J in [Hz])
(DMSO-d6, (DMSO-d6, 500 MHz)
125 MHz)
1 , 1" 138.9
2, 2" 151.2
3, 3" 106.9 6.46, d (1.4) , 4" 153.3
5, 5" 104.3 6.25, d (1.4)
6, 6" 134.1 —
1 ', 17' 202.7 —
2', 16' 42.4 2.83, t (6.8)
3', 15' 23.8 1.53, m
4', 14' 28.7 1.27, m
5', 13' 29.6 1.29, m
6', 12' 28.1 1.29, m
7', 11 ' 28.1 1.29, m
8', 10' 15.3 1.13, m; 1.34, m
9' 10.7 -0.33, m; 0.53, m
1-OCH3, 60.8 6.65, s
1"-OCH3,
2-ΟΗ, 9.54, s
2"-ΟΗ
4-ΟΗ, 9.26, s 4"-ΟΗ
HRMS, positive ESI mode: m/z = 543.2952 (calcd for [M+H]+: m/z = 543.2952)
NMR spectra were recorded in DMSO-cfe on a Bruker DRX500 spectrometer at 300 K, operating at 500.13 MHz proton frequency. Structure elucidation was done by thorough interpretation of 1D and 2D NMR spectra, combined with HPLC-MS/UV data including extracted UV as well as positive and negative mode ESI spectra. The minimum NMR
012 000008
58 dataset used consisted of 1H NMR, 1H,1H-COSY, 1H,13C-HSQC, 1H, 3C-HMBC spectra. In case of previously unknown compounds, 13C NMR data were recorded. The molecular formulae were confirmed by HR-ESIMS. Molecular formulae and elemental compositions were confirmed by high resolution ESI mass spectrometry using a Bruker MicroTOF (Bruker Daltonik GmbH, Leipzig, Germany) instrument coupled with an Agilent 1100 HPLC system as described before. Sodium formate was used for internal calibration. Structures and relevant data of the isolated pure compounds are summarised in Table 2.
Retention times, given for the isolated compounds in Table 2, were obtained by LC- MS/UV analyses performed using an Agilent HP1100 (Agilent, Waldbronn, Germany) liquid chromatograph coupled with a LCQTM (Trademark by Finnigan) Deca XPplus mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) in the positive and negative electrospray ionization (ESI) mode. A Waters symmetry column (Waters Symmetry® (Trademark by Waters) C 8, 3.5 μω, 2.1 mm x 150 mm, Waters GmbH, Eschborn, Germany) was used as stationary phase with a flow rate of 0.4 ml/min at 40°C. Mobile phase A: 0.1 % formic acid in water, mobile phase B: 0.1 % formic acid in acetonitrile; gradient: 0-1 min. 98 % A, from 1-21 min. to 100 % B, from 21-27 min 100 % B. The UV/Vis (ultraviolet/visible light) spectra were recorded between 200-500 nm, the LC-MS (Liquid Chromatography-Mass Spectrometry coupling) spectra were recorded in the range of molecular weights between 160 and 1.600 g/mol. Table 2: Compounds isolated in pure form from Labisia pumila roots
12000008
59
BTP0056-16-3 25.67 334.54 3-methoxy-5- pentadecylphenol
39 mg
BTP0056-23-3 29.00 622.98 7.0 mg
Example 3: Biological activities - Adipocyte testing Example 3.1 : Adipocyte testing of pure compounds
The purified compounds (obtained and isolated from Labisia pumila roots according to Example 2.2) were tested at 20 and 2 μΜ for their influence on the differentiation of murine 3T3-L1 pre-adipocytes to adult adipocytes. Tests have been conducted at Zen-Bio Inc. (3200 East Hwy 54 Suite 104, Research Triangle Park, NC 27709, USA) according to technologies well known in the art, based on the glycerol content of cultivated cells. For details see http.V/www.zen-bio.com/companv/index.php or http:/ www. biocat.com/bc/pdf/ZenBio 3T3 L1 User Manual.pdf. Phase 1 : Initial culturing of the pre-adipocytes
In brief, 3T3-L1 mouse embryonic fibroblast-like adipocyte cells were cultured and allowed to adhere in 3T3-L1 Pre-adipocyte medium (PM-1-L1), for 1 day . Cells were further incubated for 4 days in PM-1-L1 medium until they were confluent. Phase 2: Adipogenesis
After additional 48 hrs medium was changed to 3T3-L2 Differentiation Medium (DM-2-L1) containing the test compounds or controls.
Phase 3: Lipogenesis
After further three days incubation the media was exchanged to 3T3-L1 Adipocyte Medium (AM-1-L1) containing the compounds. The cultures were fed on day 13, 15 and 17 with AM-1-L1 containing the compounds. Cells were maintained at 37°C in 95% humidified air with 5% C02 Assay read out
At day 20, cells were washed with phosphate buffered saline (PBS) and lysed with a lysis buffer. Triglyceride content was assessed by the measurement of glycerol. The amount of glycerol liberated from triglyceride was determined via absorption at 540 nm using of a colorimetric assay kit. Glycerol concentrations were expressed as micromoles per milliliter.
As reference and positive control, the known PPAR-gamma (PPAR = peroxisome proliferator-activated receptor) agonist isoproterenol was used. TNF-alpha was used as positive control (known inhibitor of PPAR-gamma expression in adipocytes).
Tissue culture media:
PM-1-L1 (3T3-L1 Preadipocyte Medium): Dulbecco's modified Eagle's medium (DMEM), 4.5 g/l glucose, HEPES buffer pH 7.4, 10 % Bovine Calf Serum (BCS), Antibiotics (Penicillin , Streptomycin, Amphotericin B, each 100 pg/ml)
DM-2-L1 (3T3-L1 Differentiation Medium): DMEM / Ham's F-12 medium (1 :1 , v/v), HEPES pH 7.4, 10% Fetal Bovine Serum (FBS), Biotin, Pantothenate, Human insulin (167 nmol/l), Dexamethasone (1 u-mol/l), Antibiotics (Penicillin, Streptomycin, Amphotericin B, each 100 Mg/ml), Isobutylmethylxanthine , PPAR-gamma agonist (Isoproterenol)
AM-1-L1 (3T3-L1 Adipocyte Medium): DMEM / Ham's F-12 medium (1 :1 , v/v), HEPES pH 7.4, Fetal Bovine Serum (FBS), Biotin, Pantothenate, Human insulin (167 nmol/l) ,
Dexamethasone (Ι μηιοΙ/Ι), Antibiotics (Penicillin, Streptomycin, Amphotericin B, each 100 Mg/ml)
Table 3.1 : Test of individual compounds of obtained from Labisia pumila extracts on the adipogenesis and lipogenesis of murine 3T3-L1 cells (Phases 2 and 3, above)
BTP-00056-23-03
20 μΜ 16.5
2 μΜ 281.4
BTP-00056-14-03
(CAS 52483-20-2)
20 μΜ 33.4
BTP-00056-16-03
2 μΜ 265.5
(CAS 144078-14-8)
20 μΜ 23.1
BTP-00056-12-03
2 μΜ 240.4
Adipostatin A (CAS 3158-56-3)
DMSO (Vehicle) 0.1% 234.6
TNF-alpha (positive control) 10 ng/ml 15.9
PPAR agonist (reference) 10 μΜ 242.0
Example 3.2: Adipocyte testing of extracts Four extracts (E-1 to E-4) according to Example 2 were tested in the assay according to example 3.1 at a concentration of 150 Mg/ml. Said extracts were tested in adipogenesis (Phase 2), lipogenesis phase (Phase 3) and adipogenesis and lipogensis (Phases 2 and 3), respectively, as described in example 3.1 above. Table 3.2: Test of extracts from Labisia pumila on adipogenesis murine 3T3-L1 cells (Phase 2, above)
Table 3.3: Test of extracts from Labisia pumila on lipogenesis of murine 3T3-L1 cells (Phase 3, above)
Sample Concentration Average amount of
Glycerol [μΜ]
E-1 150 Mg/ml 116.38
E-2 150 Mg/ml 109.20
E-3 150 Mg/ml 24.15
E-4 150 Mg/ml 19.03
Positive control (TNF-alpha) 10 ng / ml 66.68
PPAR-gamma agonist 10 MM 175.81
(reference)
Vehicle (DMSO) 0.1 % 172.23
Table 3.4: Test of extracts from Labisia pumila on the adipogenesis and lipogenesis of murine 3T3-L1 cells (Phases 2 and 3, above)
Average amount of
Sample Concentration
Glycerol [MM]
E-1 150 pg/ml 89.73
E-2 150 Mg/ml 31.32
E-3 150 Mg/ml 4.68
E-4 150 g/ml 4.17
Positive control (TNF-alpha) 10 ng / ml 10.83
PPAR-gamma agonist 10 MM 92.30
(reference)
Vehicle (DMSO) 0.1 % 105.11
Example 4: In vivo feeding study with male rats An animal study was performed to prove efficacy (e.g. blood lipid lowering effects) of the compounds of formula (I) (as defined above) in form of extract E-4 (as described above) of the invention in a feeding model of weight gain and body composition.
For this purpose 20 male Sprague Dawley rats (approx. 8 weeks old) were fed for a two week assimilation phase with standard diet (SD, see below). Thereafter the rats were separated into two groups (each 10 rats), one treatment and one control group, that were further fed for eight weeks with SD. The treatment group received extract E-4 formulated in a vehicle (see below) via gavage catheter under light anesthesia five times per week (Monday - Friday) once a day in a dosage of 100 mg extract E-4 / kg body weight. The control group received the vehicle only.
The room temperature was maintained at 22 ± 3°C and the relative humidity was kept at 60% + 15%. The light/dark period was 12/12 hours. Drinking tap water was continuously available ad libitum via drinking bottles.
Standard diet = SD: casein 21.0 wt.%, cornstarch 50.04 wt.%, maltodextrin 3.0 wt.%, sucrose 5.0 wt.%, soybean oil 3.0 wt.%, cellulose 10.0 wt.%, vitamin mix 1.0 wt.%, mineral mix 6.5 wt.%, choline 0.20 wt.%, L-cystine 0.25 wt.%, butylated hydroxytoluol 0.01 wt.%.
Vehicle: The vehicle is composed from 10% (v/v) DMSO (Fisher Scientific D/4121/PB15), 10% (v/v) Cremophor EL (Sigma C5135), 70% (v/v) Saline (0.9% NaCI) and 10% (v/v) HPMC solution (SIGMA H7509). For test purposes the DMSO solution contained the extract E-4 at a dose of 100 / kg body weight.
The body weight was measured throughout the study once per weak. Body fat analysis (BFA) was performed using dual-energy X-ray absorptiometry (DEXA) at the beginning of the treatment phase (day 0), at the middle and the end of the study.
The body weight gain and the body fat content in the treatment group receiving extract E- 4 obtained from the Labisia pumila was lower compared to the control group. The findings show that a noticeably reduction in fat deposition can be obtained by including one or more compounds of formula (I) (as defined above) in form of an extract in a mammal diet. The observed fat reduction thus confirms the fat-reducing activity of the compounds of formula (I). Example 5: In vivo feeding study with male rats
Male Sprague-Dawley weanling rats of three weeks old were used in this study. They were allotted to two treatment groups of five rats each when they weighed an average of about 65 g. The animals were fed carbohydrate enriched diets ad libitum for a period of 31 days. The diet contained the following ingredients: casein 17.42 wt.%, methionine 0.11 wt.%, cornstarch 49.82 wt.%, sucrose 16.60 wt.%, corn oil 6.00 wt.%, cellulose 4.00 wt.%, vitamin mix 0.85 wt.%, mineral mix 5.00 wt.%, choline 0.20 wt.% (referred to as "diet-1").
The control group received the above "diet-1". The other group received "diet-2", corresponding to "diet-1" to which a total amount of 0.25 wt.% compounds of formula (I) (BTP-00056-8-6 and BTP-00056- 15-03 in a ratio of 1 : 3) was added, based on the weight of "diet-1".
The body weight gain and the body fat content in animals receiving "diet-2" comprising compounds of formula (I) obtained from the Labisia pumila was lower compared to animals that were fed "diet-1". Body fat analysis (BFA) was performed using dual-energy X-ray absorptiometry (DEXA).
The findings show that a significant reduction in fat deposition can be obtained by including one or more compounds of formula (I) (as defined above) in a mammal diet. The observed fat reduction thus confirms the fat-reducing activity of the compounds of formula (I)-
Example 6: Skin Testing - Slimming and anti-cellulite
A body lotion comprising a mixture consisting of BTP-00056-24-04 (1 wt.%), BTP-00056- 8-6 (55 wt.%), BTP-00056-15-03 (23 wt.%), BTP-00056-1 -03 (15 wt.%), BTP-00056-16- 03 (5 wt.%) and BTP-00056- 12-03 (1 wt.%) (as described above) in a final total concentration of 0.5 wt.% was tested by a panel of 30 healthy women (Caucasian type). Each of the test subjects treated one leg for two months with a composition according to the invention and the other leg with a control preparation free of compounds according to formula (I). After two months a test panel of trained examiners assessed the improvement in the cellulite appearance using a scale of 1 (just perceivable improvement) to 5 (complete elimination of the cellulite pattern). It was observed that a cosmetic composition comprising an effective amount of compounds of formula (I) (as defined above) when applied topically to the skin can achieve a slimming effect. In addition, skin appearance was improved.
Claims
1. Use of a compound of formula (I) or a physiologically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the physiologically acceptable salts thereof
(i) for the cosmetic prevention, treatment or reduction of cellulite, and/or
(ii) for the non-therapeutic, preferably cosmetic, reduction of the lipid quantity contained in subcutaneous fat tissue, and/or
inhibition of the differentiation of preadipocytes, and/or
inhibition of the lipogenesis in adipocytes,
and/or
(iii) as cosmetic anti-cellulite active,
R1 denotes hydrogen or an alkyl moiety having 1 to 4 carbon atoms, R2 denotes methyl or an acyl moiety having 1 to 4 carbon atoms, each R3 denotes hydrogen or both R3 together denote an oxygen atom, and Y denotes an organic radical having 6 to 24 carbon atoms.
Use according to claim 1 , wherein in formula (I) R1 denotes hydrogen, methyl or ethyl, and/or
R2 denotes methyl, formyl or acetyl, and/or
Y denotes an organic radical having 8 to 22 carbon atoms.
Use according to claim 1 or 2, wherein in formula (I) R1 denotes hydrogen or methyl, R2 denotes methyl or acetyl, and
Y denotes an organic radical having 8 to 22 carbon atoms and consists of carbon, hydrogen and oxygen, wherein Y contains 0, 1 , 2, 3, 4 or 5 oxygen atoms.
Use according to any one of claims 1 to 3, wherein
Y denotes a n-alkyl radical having 8 to 12 carbon atoms,
Y denotes an n-alkenyl radical having 8 to 12 carbon atoms and 1 , 2 or 3 carbon- carbon double bonds, or
Y denotes an organic radical having 17 to 21 carbon atoms consisting of carbon, hydrogen and 3 or 4 oxygen atoms.
Use according to any one of claims 1 to 5, wherein one or more compounds of formula (I) and/or a physiologically acceptable salt thereof as defined in any one of claims 1 to 5, preferably as defined in claim 5, are used in the form of a plant extract, preferably a plant extract from a plant of the family Myrsinaceae, more preferably from the genus Labisia, more preferably from Labisia pumila.
Plant extract formulation, preferably in solid form, obtainable from a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, particularly preferably from the roots of Labisia pumila, comprising
(a) one or more compounds of formula (I) as defined in claim 5 or a physiologically acceptable salt thereof in a total amount effective, preferably the total amount being in the range from 0.1 to 15 wt.%, more preferably from 0.2 to 10 wt.%, particularly preferably from 0.5 to 6 wt.%, most preferably from 1 to 5 wt.%, most preferably from 1.25 to 4 wt.%, based on the total weight of the plant extract formulation,
(b) one or more further substances naturally occurring in said plant from, wherein said one or more further substances are not compounds of formula (I) as defined in claim 5 or a physiologically acceptable salt thereof, and
(c) one or more solid carrier substances, preferably one or more solid carrier substances selected from the group consisting of maltodextrins, silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stearate salts, preferably magnesium stearate, alginates, tragacanth, gelatins, calcium silicates, cellulose and cellulose derivatives, preferably microcrystalline cellulose or methyl cellulose, polyvinylpyrrolidones, and propylhydroxybenzoates.
8. Method for producing a plant extract formulation according to claim 7, comprising the following steps:
(i) providing a plant or a part of a plant of the family Myrsinaceae, preferably from the genus Labisia, more preferably from Labisia pumila, particularly preferably from the roots of Labisia pumila,
(i-a) optionally drying the plant or a part of a plant of step (i),
(ii) extraction of said plant or a part of a plant provided in step (i) or (i-a) with a mixture of water and an alcohol having 1 to 3 carbon atoms, preferably with
a mixture of ethanol and water, wherein the volume ratio (v/v) of said alcohol : water in the range of 1 : 2 to 30 : 1 , more preferably in the range of 1 : 1 to 25 : 1 , most preferably in the range of 3 : 2 to 20 : 1 ,
(ii-a) optionally partially or fully removing the alcohol having 1 to 3 carbon atoms of step (ii), preferably at a temperature below 70 °C, more preferably in the range from 15 to 55 °C,
(iii) mixing the extract obtained in step (ii) or (ii-a) with one or more solid carrier substances, preferably one or more solid carrier substances selected from the group consisting of maltodextrins, silica, talc, lactose, dextrose, sucrose, sorbitol, mannitol, starches, gum acacia, calcium phosphate, orally acceptable stearate salts, preferably magnesium stearate, alginates, tragacanth, gelatins, calcium silicates, cellulose and cellulose derivatives, preferably microcrystalline cellulose or methyl cellulose, polyvinylpyrrolidones, and propylhydroxybenzoates,
(iv) optionally drying the mixture obtained in step (iii), preferably by spray- drying or freeze-drying.
9. Composition, preferably topically or orally administrable cosmetic or pharmaceutical composition, comprising
(a) an effective amount of one, two or more compounds of formula (I) and/or a physiologically acceptable salt thereof as defined in one of claims 1 to 5, or of a plant extract formulation as defined in claim 7, said effective amount being sufficient to reduce the lipid quantity contained in fat tissue, preferably subcutaneous fat tissue, and/or to inhibit the differentiation of preadipocytes, and/or
to inhibit the lipogenesis in adipocytes, and preferably additionally one or more liquid or solid carrier substances.
10. Composition according to claim 9, comprising one or more compounds selected from the following groups (b) and/or (c):
(b) one or more lipolysis stimulants, preferably selected from
(b-i) the group of phosphodiesterase inhibitors, and (b-ii) the group of agonists of beta-adrenergic receptors,
(c) one or more stimulators of the transport or oxidation of free fatty acids, preferably selected from
(c-i) the group of promoters of the transport of free fatty acids in the mitochondria, preferably coenzyme A, and
(c-ii) the group of stimulators of beta-oxidation, preferably L-carnitine.
11. Composition according to claim 9 or 10, comprising (b) one or more lipolysis stimulants, wherein one, a plurality or all of the lipolysis stimulants of component (b) are preferably selected from the group consisting of
(b-i) xanthines, preferably selected from the group consisting of caffeine, theobromine and theophylline, and
(b-ii) [beta]-phenylethylamines, preferably selected from the group consisting of adrenaline, noradrenaline, metanephrine, macromerine, normacromerine,
hordenine, N-methyltyramine, dopamine, octopamine, tyramine, 2- phenylethylamine, phenylethanolamine, epinine (N-methyldopamine), synephrine, ephedrine, pseudoephedrine, norephedrine and isoprenaline.
Composition according to one of claims 9 to 11 , wherein, in each case based on the total weight of the composition, the total quantity of compounds of formula (I) is in the range of from 0.01 - 10 % by weight, preferably in the range of from 0.025 - 5 % by weight and particularly preferably in the range of from 0.05 - 3 % by weight and most preferably in the range of from 0.1 - 2 % by weight, and/or the total quantity of phosphodiesterase inhibitors (b-i) is in the range of from 0.005 - 10 % by weight, preferably in the range of from 0.05 - 5 % by weight and more preferably in the range of from 0.5 - 2.5 % by weight, and/or the total quantity of agonists of beta-adrenergic receptors (b-ii) is in the range of from 0.0001 - 0.10 % by weight, preferably in the range of from 0.001 - 0.05 % by weight and more preferably in the range of from 0.002 - 0.02 % by weight.
Compound of formula (I) or a physiologically acceptable salt thereof, selected from the group consisting of: o o o o'
Method for the non-therapeutic, preferably cosmetic,
(i) prevention, treatment or reduction of cellulite, and/or
(ii) reduction of the lipid quantity contained in fat tissue, preferably in subcutaneous fat tissue and/or inhibition of the differentiation of preadipocytes, and/or inhibition of the lipogenesis in adipocytes, and/or
(iii) modulation of adipocyte differentiation, comprising the following step: application of an effective amount of a compound of formula (I) or a physiologically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the salts thereof as defined in any one of claims 1 to 5 or 13, a plant extract formulation as defined in claim 7 or a cosmetic composition as defined in any one of claims 9 to 12.
15. Compound of formula (I) or a pharmaceutically acceptable salt of a compound of formula (I) or a mixture containing two or more of these compounds or the pharmaceutically acceptable salts thereof as defined in any one of the claims 1 to
5 or 13, a plant extract formulation as defined in claim 7, or a composition as defined in any one of claims 9 to 12, for use in a prophylactic or therapeutic method for treating adiposity.
Pharmaceutical composition comprising an effective amount of one or more compounds of formula (I) as defined in claim 4, 5 or 13, or a plant extract formulation as defined in claim 7, for use in a therapeutic method for preventing and/or treating obesity.
Method for the therapeutic reduction of the body fat content or the therapeutic treatment of obesity, comprising administering, preferably orally, an effective amount of one or more compounds of formula (I) as defined in any one of claims 1 to 5 or 13, a plant extract formulation as defined in claim 7 or a composition as defined in any one of claims 9 to 12, said effective amount being sufficient to
(i) reduce the body fat content, and/or
(ii) reduce the lipid quantity contained in fat tissue, and/or
(iii) inhibit the differentiation of preadipocytes, and/or inhibit of the lipogenesis in adipocytes, and/or
(iv) modulate the adipocyte differentiation.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MYPI2014001278A MY170724A (en) | 2012-01-27 | 2012-01-27 | Use of certain trioxygenated benzene derivatives in body fat management |
| PCT/MY2012/000008 WO2013112040A1 (en) | 2012-01-27 | 2012-01-27 | Use of certain trioxygenated benzene derivatives in body fat management |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/MY2012/000008 WO2013112040A1 (en) | 2012-01-27 | 2012-01-27 | Use of certain trioxygenated benzene derivatives in body fat management |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013112040A1 true WO2013112040A1 (en) | 2013-08-01 |
Family
ID=45688211
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/MY2012/000008 WO2013112040A1 (en) | 2012-01-27 | 2012-01-27 | Use of certain trioxygenated benzene derivatives in body fat management |
Country Status (2)
| Country | Link |
|---|---|
| MY (1) | MY170724A (en) |
| WO (1) | WO2013112040A1 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016093692A1 (en) * | 2014-12-08 | 2016-06-16 | Universiti Sains Malaysia | Standardised extract of labisia pumila for weight reduction and nanoformulation thereof |
| CN106562939A (en) * | 2015-10-08 | 2017-04-19 | 中国科学院微生物研究所 | Application of Compound AJ-4 in the Preparation of Medicines for Treating or Preventing Influenza Viruses |
| WO2018026263A3 (en) * | 2016-08-01 | 2018-04-26 | Universiti Kebangsaan Malaysia | A process for extracting irisresorcinol from labisia pumila |
| WO2023080772A1 (en) | 2021-11-02 | 2023-05-11 | Medika Natura Sdn. Bhd. | Labisia pumila extract composition and its pharmaceutical formulation |
Citations (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5352690A (en) | 1992-07-01 | 1994-10-04 | Eli Lilly And Company | 1,2,4-trioxygenated benzene derivatives useful as leukotriene antagonists |
| WO1997028115A1 (en) * | 1996-02-02 | 1997-08-07 | Merck & Co., Inc. | Antidiabetic agents |
| DE10009423A1 (en) | 2000-02-28 | 2001-09-06 | Henkel Kgaa | Cosmetic or pharmaceutical preparation especially for treatment of cellulite comprises nerve fibre stimulator or depolariser, phosphodiesterase inhibitor and antiestrogen |
| WO2001076572A2 (en) | 2000-04-12 | 2001-10-18 | bitop Aktiengesellschaft für biotechnische Optimierung | Use of compatible solutes as substances having free radical scavenging properties |
| WO2002015868A2 (en) | 2000-08-18 | 2002-02-28 | Bitop Gesellschaft Für Biotechnische Optimierung Mbh | Cosmetic formulation containing at least one di-sugar alcohol phosphate of c3 to c6 sugar alcohols |
| EP1234572A1 (en) | 2001-02-26 | 2002-08-28 | Mibelle AG Cosmetics | Skin treatment compositions containing isoflavone aglycones |
| US6530886B1 (en) | 1999-10-08 | 2003-03-11 | Tanita Corporation | Method and apparatus for measuring subcutaneous fat using ultrasonic wave |
| WO2004082700A1 (en) | 2003-03-19 | 2004-09-30 | Korea Institute Of Oriental Medicine | Composition for treatment and prevention of obesity and adult disease |
| WO2004096252A1 (en) | 2003-04-25 | 2004-11-11 | Primavera Biosciences, Inc. | Compositions and methods for weight loss |
| WO2005123101A1 (en) | 2004-06-18 | 2005-12-29 | Symrise Gmbh & Co. Kg | Blackberry extract |
| US20060089413A1 (en) | 2002-11-25 | 2006-04-27 | Symrise Gmbh & Co. Kg | Anthranilic acid amides and derivatives thereof as cosmetic and pharmaceutical agents |
| WO2006045760A1 (en) | 2004-10-25 | 2006-05-04 | Symrise Gmbh & Co. Kg | Use of glycosylated flavanones for the browning of skin or hair |
| WO2006053912A1 (en) | 2004-11-22 | 2006-05-26 | Symrise Gmbh & Co. Kg | Formulations comprising ceramides and/or pseudoceramides and (alpha-)bisabolol for combating skin damage |
| WO2006063714A1 (en) | 2004-12-14 | 2006-06-22 | Menarini Ricerche S.P.A. | Pharmaceutical compositions for the treatment of cellulite |
| US20070043109A1 (en) | 2003-05-20 | 2007-02-22 | Sederma Sas | Slimming cosmetic composition |
| US20070082069A1 (en) | 2005-10-12 | 2007-04-12 | Gov't Of Malaysia, As Rep. By The Ministry Of Science, Technology And Innovation | Process for preparation of labisia pumila extract |
| WO2007042472A1 (en) | 2005-10-14 | 2007-04-19 | Symrise Gmbh & Co. Kg | Synergistic mixtures of bisabolol and ginger extract |
| WO2007077541A2 (en) | 2006-01-04 | 2007-07-12 | Sederma | Cosmetic composition comprising glaucine and its use |
| WO2007110415A2 (en) | 2006-03-27 | 2007-10-04 | Symrise Gmbh & Co. Kg | Use of diacetyl trimers and cosmetic or therapeutic formulations containing these compounds |
| US20080070825A1 (en) | 2006-09-16 | 2008-03-20 | Symrise Gmbh & Co. Kg | 2-Methyl-2-alkenyl-substituted 1,3-dioxanes as odoriferous substances |
| WO2008046791A1 (en) | 2006-10-20 | 2008-04-24 | Symrise Gmbh & Co. Kg | Use of 1,2-decanediol for sebum reduction and cosmetic and/or dermatological formulations comprising 1,2-decanediol |
| WO2008046676A1 (en) | 2006-10-20 | 2008-04-24 | Henkel Ag & Co. Kgaa | Cosmetic composition comprising purine and/or a purine derivative and taurine |
| WO2008046795A1 (en) | 2006-10-20 | 2008-04-24 | Symrise Gmbh & Co. Kg | Use of c10-c14-alkanediols in the preparation of a composition for the prophylaxis and/or treatment of malassezia-induced dandruff formation and compositions comprising c10-c14-alkanediols |
| WO2009021622A1 (en) | 2007-08-16 | 2009-02-19 | Intermed Discovery Gmbh | Extracts with liver-x-receptor modulators, compounds and their use especially in weight control |
| WO2009064156A1 (en) | 2007-11-15 | 2009-05-22 | Holista Biotechnology Sdn Bhd | Novel composition obtained from labisia pumila extract for hepatoprotective activity |
| WO2009064155A1 (en) | 2007-11-15 | 2009-05-22 | Holista Biotechnology Sdn Bhd | Immunopotentiating composition from labisia pumila extract |
| WO2009116848A1 (en) | 2008-03-19 | 2009-09-24 | Government Of Malaysia | Labisia pumila extracts for reducing the risk of cardiovascular diseases |
| KR20090113723A (en) | 2008-04-28 | 2009-11-02 | 동국대학교 산학협력단 | Cosmetic composition and pharmaceutical composition comprising extract or fermentation product of Ravizi pumila |
| US20100285153A1 (en) | 2006-04-07 | 2010-11-11 | Kao Corporation | Lipolysis Stimulator |
| WO2010147449A1 (en) | 2009-06-16 | 2010-12-23 | Holista Biotech Sdn. Bhd. | Adaptogenic activity of labisia pumila extract |
-
2012
- 2012-01-27 WO PCT/MY2012/000008 patent/WO2013112040A1/en active Application Filing
- 2012-01-27 MY MYPI2014001278A patent/MY170724A/en unknown
Patent Citations (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5352690A (en) | 1992-07-01 | 1994-10-04 | Eli Lilly And Company | 1,2,4-trioxygenated benzene derivatives useful as leukotriene antagonists |
| WO1997028115A1 (en) * | 1996-02-02 | 1997-08-07 | Merck & Co., Inc. | Antidiabetic agents |
| US6530886B1 (en) | 1999-10-08 | 2003-03-11 | Tanita Corporation | Method and apparatus for measuring subcutaneous fat using ultrasonic wave |
| DE10009423A1 (en) | 2000-02-28 | 2001-09-06 | Henkel Kgaa | Cosmetic or pharmaceutical preparation especially for treatment of cellulite comprises nerve fibre stimulator or depolariser, phosphodiesterase inhibitor and antiestrogen |
| WO2001076572A2 (en) | 2000-04-12 | 2001-10-18 | bitop Aktiengesellschaft für biotechnische Optimierung | Use of compatible solutes as substances having free radical scavenging properties |
| WO2002015868A2 (en) | 2000-08-18 | 2002-02-28 | Bitop Gesellschaft Für Biotechnische Optimierung Mbh | Cosmetic formulation containing at least one di-sugar alcohol phosphate of c3 to c6 sugar alcohols |
| EP1234572A1 (en) | 2001-02-26 | 2002-08-28 | Mibelle AG Cosmetics | Skin treatment compositions containing isoflavone aglycones |
| US20060089413A1 (en) | 2002-11-25 | 2006-04-27 | Symrise Gmbh & Co. Kg | Anthranilic acid amides and derivatives thereof as cosmetic and pharmaceutical agents |
| WO2004082700A1 (en) | 2003-03-19 | 2004-09-30 | Korea Institute Of Oriental Medicine | Composition for treatment and prevention of obesity and adult disease |
| WO2004096252A1 (en) | 2003-04-25 | 2004-11-11 | Primavera Biosciences, Inc. | Compositions and methods for weight loss |
| US20070043109A1 (en) | 2003-05-20 | 2007-02-22 | Sederma Sas | Slimming cosmetic composition |
| WO2005123101A1 (en) | 2004-06-18 | 2005-12-29 | Symrise Gmbh & Co. Kg | Blackberry extract |
| WO2006045760A1 (en) | 2004-10-25 | 2006-05-04 | Symrise Gmbh & Co. Kg | Use of glycosylated flavanones for the browning of skin or hair |
| WO2006053912A1 (en) | 2004-11-22 | 2006-05-26 | Symrise Gmbh & Co. Kg | Formulations comprising ceramides and/or pseudoceramides and (alpha-)bisabolol for combating skin damage |
| WO2006063714A1 (en) | 2004-12-14 | 2006-06-22 | Menarini Ricerche S.P.A. | Pharmaceutical compositions for the treatment of cellulite |
| US20070082069A1 (en) | 2005-10-12 | 2007-04-12 | Gov't Of Malaysia, As Rep. By The Ministry Of Science, Technology And Innovation | Process for preparation of labisia pumila extract |
| US20100311180A1 (en) | 2005-10-12 | 2010-12-09 | Gov't Of Malaysia, As Rep. By The Ministry Of Science, Technology And Innovation | Process for preparation of labisia pumila extract |
| WO2007042472A1 (en) | 2005-10-14 | 2007-04-19 | Symrise Gmbh & Co. Kg | Synergistic mixtures of bisabolol and ginger extract |
| WO2007077541A2 (en) | 2006-01-04 | 2007-07-12 | Sederma | Cosmetic composition comprising glaucine and its use |
| WO2007110415A2 (en) | 2006-03-27 | 2007-10-04 | Symrise Gmbh & Co. Kg | Use of diacetyl trimers and cosmetic or therapeutic formulations containing these compounds |
| US20100285153A1 (en) | 2006-04-07 | 2010-11-11 | Kao Corporation | Lipolysis Stimulator |
| US20080070825A1 (en) | 2006-09-16 | 2008-03-20 | Symrise Gmbh & Co. Kg | 2-Methyl-2-alkenyl-substituted 1,3-dioxanes as odoriferous substances |
| WO2008046676A1 (en) | 2006-10-20 | 2008-04-24 | Henkel Ag & Co. Kgaa | Cosmetic composition comprising purine and/or a purine derivative and taurine |
| WO2008046795A1 (en) | 2006-10-20 | 2008-04-24 | Symrise Gmbh & Co. Kg | Use of c10-c14-alkanediols in the preparation of a composition for the prophylaxis and/or treatment of malassezia-induced dandruff formation and compositions comprising c10-c14-alkanediols |
| WO2008046791A1 (en) | 2006-10-20 | 2008-04-24 | Symrise Gmbh & Co. Kg | Use of 1,2-decanediol for sebum reduction and cosmetic and/or dermatological formulations comprising 1,2-decanediol |
| WO2009021622A1 (en) | 2007-08-16 | 2009-02-19 | Intermed Discovery Gmbh | Extracts with liver-x-receptor modulators, compounds and their use especially in weight control |
| WO2009064156A1 (en) | 2007-11-15 | 2009-05-22 | Holista Biotechnology Sdn Bhd | Novel composition obtained from labisia pumila extract for hepatoprotective activity |
| WO2009064155A1 (en) | 2007-11-15 | 2009-05-22 | Holista Biotechnology Sdn Bhd | Immunopotentiating composition from labisia pumila extract |
| WO2009116848A1 (en) | 2008-03-19 | 2009-09-24 | Government Of Malaysia | Labisia pumila extracts for reducing the risk of cardiovascular diseases |
| KR20090113723A (en) | 2008-04-28 | 2009-11-02 | 동국대학교 산학협력단 | Cosmetic composition and pharmaceutical composition comprising extract or fermentation product of Ravizi pumila |
| WO2010147449A1 (en) | 2009-06-16 | 2010-12-23 | Holista Biotech Sdn. Bhd. | Adaptogenic activity of labisia pumila extract |
Non-Patent Citations (23)
| Title |
|---|
| ANGEW. CHEM. INT. ED. ENGL., vol. 39, no. 1, 2000, pages 44 - 122 |
| ANONYMOUS: "Wholesale kacip fatimah", 18 March 2011 (2011-03-18), XP002686546, Retrieved from the Internet <URL:http://www.tongkatali-france.com/wholesalekacipfatimah.htm> [retrieved on 20121105] * |
| CHEM. BIODIV., vol. 7, 2010, pages 2901 - 7 |
| CHEN L-P ET AL: "Antitumor effect of resorcinol derivatives from the roots of Ardisia brevicaulis by inducing apoptosis", JOURNAL OF ASIAN NATURAL PRODUCTS RESEARCH, GORDON AND BREACH, CHUR, CH, vol. 13, no. 8, 1 January 2011 (2011-01-01), pages 734 - 743, XP009163972, ISSN: 1028-6020, DOI: 10.1080/10286020.2011.587412 * |
| CHUA ET AL., FOOD CHEMISTRY, vol. 127, 2011, pages 1186 - 1192 |
| FRANZ-JOSEF MARNER ET AL: "Phenols and Quinones from Seeds of Different Iris Species", HELVETICA CHIMICA ACTA, 1 January 1992 (1992-01-01), pages 1557 - 1562, XP055042322, Retrieved from the Internet <URL:http://onlinelibrary.wiley.com/store/10.1002/hlca.19920750509/asset/19920750509_ftp.pdf?v=1&t=h8qy9zqr&s=fe42b9dc39dc906210c99500cb6868da74b3e2fc> [retrieved on 20121026] * |
| J. MED. PLANTS RES., vol. 3, no. 4, 2009, pages 217 - 223 |
| J. PHARM. PHARMACOL., vol. 50, 1998, pages 79 |
| J. PHARM. PHARMACOL., vol. 51, 1999, pages 236 |
| JAMAL ET AL.: "Phytochemicals and Biopharmaceutics from the Malaysian Rain Forest", 1999, FOREST RESEARCH INSTITUTE MALAYSIA, article "Kacip Fatimah: A Malay Traditional Herb for Pregnant Women", pages: 166 - 176 |
| JOURNAL OF ANTIBIOTICS, 1992, pages 886 - 891 |
| KATSURA SEKI ET AL: "Phenols and a dioxotetrahydrodibenzofuran from seeds of Iris pallasii", PHYTOCHEMISTRY, vol. 38, no. 4, 1 March 1995 (1995-03-01), pages 965 - 973, XP055042320, ISSN: 0031-9422, DOI: 10.1016/0031-9422(94)00731-8 * |
| LI BAO ET AL: "Two New Resorcinol Derivatives with Strong Cytotoxicity from the Roots of Ardisia brevicaulisDiels", CHEMISTRY & BIODIVERSITY, vol. 7, no. 12, 1 December 2010 (2010-12-01), pages 2901 - 2907, XP055042028, ISSN: 1612-1872, DOI: 10.1002/cbdv.200900349 * |
| MANNERAS L ET AL: "Beneficial metabolic effects of the Malaysian herb Labisia pumila var. alata in a rat model of polycystic ovary syndrome", JOURNAL OF ETHNOPHARMACOLOGY, ELSEVIER SCIENTIFIC PUBLISHERS LTD, IE, vol. 127, no. 2, 3 February 2010 (2010-02-03), pages 346 - 351, XP026853055, ISSN: 0378-8741, [retrieved on 20091031] * |
| MEGUMI SUMINO: "Ardisiphenols and other antioxidant principles from the fruits of Ardisia colorata.", CHEMICAL & PHARMACEUTICAL BULLETIN, vol. 50, no. 11, 1 November 2002 (2002-11-01), pages 1484 - 1487, XP055041904, ISSN: 0009-2363 * |
| MICHAEL B. SMITH; JERRY MARCH: "March's Advanced Organic Chemistry: Reaction, Mechanisms and Structure, 5th ed.", 2001, WILEY-INTERSCIENCE |
| NICOLAOU, SORENSEN: "Classics in Total Synthesis: Targets, Strategies, Methods", 1996, JOHN WILEY & SON LTD |
| PHYTOCHEMISTRY, 2011 |
| PHYTOCHEMISTRY, vol. 70, 2009, pages 2064 - 71 |
| S. ARCTANDER: "Perfume and Flavor Chemicals", 1969, PRIVATE PUBLISHING HOUSE |
| S. FOSTER: "Balancing nature and wellness: Malaysian traditions of ''Ramuan'' - The history, culture, biodiversity and scientific assimilation of medicinal plants in Malaysia", HERBALGRAM, vol. 84, 2009, pages 30 - 43 |
| SURBURG, PANTEN: "Common Fragrance and Flavor Materials, 5th ed.", 2006, WILEY-VCH |
| ZULFIQAR ALI ET AL: "Alkyl phenols and saponins from the roots of(Kacip Fatimah)", PHYTOCHEMISTRY, PERGAMON PRESS, GB, vol. 72, no. 16, 27 June 2011 (2011-06-27), pages 2075 - 2080, XP028294904, ISSN: 0031-9422, [retrieved on 20110705], DOI: 10.1016/J.PHYTOCHEM.2011.06.014 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016093692A1 (en) * | 2014-12-08 | 2016-06-16 | Universiti Sains Malaysia | Standardised extract of labisia pumila for weight reduction and nanoformulation thereof |
| CN106562939A (en) * | 2015-10-08 | 2017-04-19 | 中国科学院微生物研究所 | Application of Compound AJ-4 in the Preparation of Medicines for Treating or Preventing Influenza Viruses |
| CN106562939B (en) * | 2015-10-08 | 2019-01-15 | 中国科学院微生物研究所 | Application of compound AJ-4 in preparing medicine for treating or preventing influenza virus |
| WO2018026263A3 (en) * | 2016-08-01 | 2018-04-26 | Universiti Kebangsaan Malaysia | A process for extracting irisresorcinol from labisia pumila |
| WO2023080772A1 (en) | 2021-11-02 | 2023-05-11 | Medika Natura Sdn. Bhd. | Labisia pumila extract composition and its pharmaceutical formulation |
Also Published As
| Publication number | Publication date |
|---|---|
| MY170724A (en) | 2019-08-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10688142B2 (en) | Methods of treatment using a lipid extract of Passiflora seeds concentrated in its unsaponifiable fraction | |
| KR101800498B1 (en) | Composition for improving skin wrinkle comprising extract or compounds derived from unripe apple | |
| KR101436199B1 (en) | Composition for Improving Skin Conditions Comprising Hordenine | |
| CN107530258B (en) | Composition for promoting hair growth or hair restoration and for anti-inflammatory | |
| JP6055667B2 (en) | Collagen production promoter | |
| US9445987B2 (en) | Ceramide production enhancer and moisturizer | |
| WO2014034802A1 (en) | Transglutaminase activator | |
| KR20140012456A (en) | Composition for improving skin conditions comprising akebia saponin d | |
| WO2013112040A1 (en) | Use of certain trioxygenated benzene derivatives in body fat management | |
| JP6709783B2 (en) | Composition for preventing hair loss or promoting hair growth containing alpine wormwood extract | |
| KR20250009941A (en) | Composition for cortisone reductase inhibition | |
| KR100975819B1 (en) | A composition for preventing hair loss and promoting hair growth containing hydroxya extract or 4-methyl-honokiol separated therefrom | |
| KR20210131598A (en) | Composition for preventing hair loss or promoting hair growth, comprising Camellia japonica pericarp extract as an active ingredient | |
| KR101050484B1 (en) | Skin whitening composition containing lactone compound as an active ingredient | |
| US9730872B2 (en) | Method for improving skin conditions with veratric acid or acceptable salt thereof as an active ingredient | |
| KR102292114B1 (en) | Composition for Improving Skin Conditions Having Moisturizing, Anti-Inflammatory Caused by Fine Dust and Pore Shrinkage Property Comprising Plant Complex Extracts as Active Ingredient | |
| KR102264006B1 (en) | Composition for inhibiting production of melanin and promoting decomposition of melanin | |
| KR101069906B1 (en) | A composition for improving skin conditions comprising extract of Aster subulatus Michx. | |
| JP5419258B2 (en) | Cosmetics | |
| JP7028803B2 (en) | Whitening agent | |
| KR101526435B1 (en) | Compositions for skin-whitening comprising extract of Vitis amurensis ruprecht | |
| KR102810314B1 (en) | A cosmetic composition for skin whitening comprising a mixed extract of Geranium kunthii and Zephyrnathes candida as an active ingredient, and a pharmaceutical composition for preventing or treating diseases of melanin hyperpigmentation | |
| KR102360927B1 (en) | Composition for preventing hair loss and promoting hair growth comprising runcinatside | |
| KR20250069743A (en) | Composition for antioxidant or skin whitening comprising Sebok extracts | |
| KR20060031007A (en) | Cosmetic composition containing cheonma extract having a skin whitening effect and a compound separated therefrom |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12704556 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 32PN | Ep: public notification in the ep bulletin as address of the adressee cannot be established |
Free format text: NOTING OF LOSS OF RIGHTS PURSUANT TO RULE 112(1) EPC (EPO FORM 1205A DATED 09/02/2015) |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12704556 Country of ref document: EP Kind code of ref document: A1 |