WO2012173003A2 - 医療用組織マーカー及びその製造方法 - Google Patents
医療用組織マーカー及びその製造方法 Download PDFInfo
- Publication number
- WO2012173003A2 WO2012173003A2 PCT/JP2012/064235 JP2012064235W WO2012173003A2 WO 2012173003 A2 WO2012173003 A2 WO 2012173003A2 JP 2012064235 W JP2012064235 W JP 2012064235W WO 2012173003 A2 WO2012173003 A2 WO 2012173003A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- phospholipid
- hydrophilic solvent
- cluster
- emulsion
- medical tissue
- Prior art date
Links
- 239000003550 marker Substances 0.000 title claims abstract description 56
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 23
- 239000002904 solvent Substances 0.000 claims abstract description 90
- 239000000839 emulsion Substances 0.000 claims abstract description 52
- 150000003904 phospholipids Chemical class 0.000 claims abstract description 48
- 239000002872 contrast media Substances 0.000 claims abstract description 35
- 239000003995 emulsifying agent Substances 0.000 claims abstract description 29
- 239000002775 capsule Substances 0.000 claims abstract description 26
- 239000007850 fluorescent dye Substances 0.000 claims abstract description 25
- 230000002209 hydrophobic effect Effects 0.000 claims description 28
- 239000000725 suspension Substances 0.000 claims description 19
- 235000008753 Papaver somniferum Nutrition 0.000 claims description 14
- 240000001090 Papaver somniferum Species 0.000 claims description 14
- 239000002562 thickening agent Substances 0.000 claims description 14
- 125000004494 ethyl ester group Chemical group 0.000 claims description 13
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 claims description 10
- 239000000787 lecithin Substances 0.000 claims description 10
- 229940067606 lecithin Drugs 0.000 claims description 10
- 235000010445 lecithin Nutrition 0.000 claims description 10
- 238000003756 stirring Methods 0.000 claims description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 5
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 claims description 3
- 210000000056 organ Anatomy 0.000 abstract description 29
- 230000015572 biosynthetic process Effects 0.000 abstract description 3
- 238000003786 synthesis reaction Methods 0.000 abstract 2
- 230000000699 topical effect Effects 0.000 abstract 1
- 230000000694 effects Effects 0.000 description 50
- 210000001519 tissue Anatomy 0.000 description 50
- 238000000034 method Methods 0.000 description 22
- 239000000243 solution Substances 0.000 description 20
- 210000002784 stomach Anatomy 0.000 description 19
- 239000007924 injection Substances 0.000 description 18
- 238000002347 injection Methods 0.000 description 18
- 239000000203 mixture Substances 0.000 description 15
- 239000003921 oil Substances 0.000 description 15
- 238000002073 fluorescence micrograph Methods 0.000 description 14
- 239000012071 phase Substances 0.000 description 12
- 241000282898 Sus scrofa Species 0.000 description 10
- 239000007983 Tris buffer Substances 0.000 description 9
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N dodecahydrosqualene Natural products CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 description 9
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 9
- 238000013170 computed tomography imaging Methods 0.000 description 8
- YYGNTYWPHWGJRM-UHFFFAOYSA-N (6E,10E,14E,18E)-2,6,10,15,19,23-hexamethyltetracosa-2,6,10,14,18,22-hexaene Chemical compound CC(C)=CCCC(C)=CCCC(C)=CCCC=C(C)CCC=C(C)CCC=C(C)C YYGNTYWPHWGJRM-UHFFFAOYSA-N 0.000 description 7
- BHEOSNUKNHRBNM-UHFFFAOYSA-N Tetramethylsqualene Natural products CC(=C)C(C)CCC(=C)C(C)CCC(C)=CCCC=C(C)CCC(C)C(=C)CCC(C)C(C)=C BHEOSNUKNHRBNM-UHFFFAOYSA-N 0.000 description 7
- 239000006185 dispersion Substances 0.000 description 7
- 229960004657 indocyanine green Drugs 0.000 description 7
- MOFVSTNWEDAEEK-UHFFFAOYSA-M indocyanine green Chemical compound [Na+].[O-]S(=O)(=O)CCCCN1C2=CC=C3C=CC=CC3=C2C(C)(C)C1=CC=CC=CC=CC1=[N+](CCCCS([O-])(=O)=O)C2=CC=C(C=CC=C3)C3=C2C1(C)C MOFVSTNWEDAEEK-UHFFFAOYSA-M 0.000 description 7
- 229940031439 squalene Drugs 0.000 description 7
- TUHBEKDERLKLEC-UHFFFAOYSA-N squalene Natural products CC(=CCCC(=CCCC(=CCCC=C(/C)CCC=C(/C)CC=C(C)C)C)C)C TUHBEKDERLKLEC-UHFFFAOYSA-N 0.000 description 7
- JQWAHKMIYCERGA-UHFFFAOYSA-N (2-nonanoyloxy-3-octadeca-9,12-dienoyloxypropoxy)-[2-(trimethylazaniumyl)ethyl]phosphinate Chemical compound CCCCCCCCC(=O)OC(COP([O-])(=O)CC[N+](C)(C)C)COC(=O)CCCCCCCC=CCC=CCCCCC JQWAHKMIYCERGA-UHFFFAOYSA-N 0.000 description 6
- 238000010521 absorption reaction Methods 0.000 description 6
- 239000007864 aqueous solution Substances 0.000 description 6
- 230000008859 change Effects 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 5
- 108010010803 Gelatin Proteins 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 238000010586 diagram Methods 0.000 description 5
- 229920000159 gelatin Polymers 0.000 description 5
- 239000008273 gelatin Substances 0.000 description 5
- 235000019322 gelatine Nutrition 0.000 description 5
- 235000011852 gelatine desserts Nutrition 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 4
- 229920001817 Agar Polymers 0.000 description 4
- 108010049003 Fibrinogen Proteins 0.000 description 4
- 102000008946 Fibrinogen Human genes 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- 239000007995 HEPES buffer Substances 0.000 description 4
- 238000003745 diagnosis Methods 0.000 description 4
- 210000002969 egg yolk Anatomy 0.000 description 4
- 229940012952 fibrinogen Drugs 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 238000001000 micrograph Methods 0.000 description 4
- 239000008363 phosphate buffer Substances 0.000 description 4
- 238000001356 surgical procedure Methods 0.000 description 4
- 229930006000 Sucrose Natural products 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 239000008272 agar Substances 0.000 description 3
- -1 although not limited Proteins 0.000 description 3
- 239000008346 aqueous phase Substances 0.000 description 3
- 229960001506 brilliant green Drugs 0.000 description 3
- HXCILVUBKWANLN-UHFFFAOYSA-N brilliant green cation Chemical compound C1=CC(N(CC)CC)=CC=C1C(C=1C=CC=CC=1)=C1C=CC(=[N+](CC)CC)C=C1 HXCILVUBKWANLN-UHFFFAOYSA-N 0.000 description 3
- 150000001720 carbohydrates Chemical class 0.000 description 3
- 239000000499 gel Substances 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 229960003988 indigo carmine Drugs 0.000 description 3
- KHLVKKOJDHCJMG-QDBORUFSSA-L indigo carmine Chemical compound [Na+].[Na+].N/1C2=CC=C(S([O-])(=O)=O)C=C2C(=O)C\1=C1/NC2=CC=C(S(=O)(=O)[O-])C=C2C1=O KHLVKKOJDHCJMG-QDBORUFSSA-L 0.000 description 3
- 235000012738 indigotine Nutrition 0.000 description 3
- 239000004179 indigotine Substances 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 239000002504 physiological saline solution Substances 0.000 description 3
- 230000035945 sensitivity Effects 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- 210000004876 tela submucosa Anatomy 0.000 description 3
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerol Natural products OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- YCOXTKKNXUZSKD-UHFFFAOYSA-N as-o-xylenol Natural products CC1=CC=C(O)C=C1C YCOXTKKNXUZSKD-UHFFFAOYSA-N 0.000 description 2
- 159000000009 barium salts Chemical class 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- DIOQZVSQGTUSAI-UHFFFAOYSA-N decane Chemical compound CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- SNRUBQQJIBEYMU-UHFFFAOYSA-N dodecane Chemical compound CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 2
- 238000001839 endoscopy Methods 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 238000003384 imaging method Methods 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 230000009878 intermolecular interaction Effects 0.000 description 2
- SNHMUERNLJLMHN-UHFFFAOYSA-N iodobenzene Chemical compound IC1=CC=CC=C1 SNHMUERNLJLMHN-UHFFFAOYSA-N 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 238000005191 phase separation Methods 0.000 description 2
- 238000001338 self-assembly Methods 0.000 description 2
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- 229920000936 Agarose Polymers 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- 102000012422 Collagen Type I Human genes 0.000 description 1
- 108010022452 Collagen Type I Proteins 0.000 description 1
- 102000000503 Collagen Type II Human genes 0.000 description 1
- 108010041390 Collagen Type II Proteins 0.000 description 1
- 102000001187 Collagen Type III Human genes 0.000 description 1
- 108010069502 Collagen Type III Proteins 0.000 description 1
- 102000012432 Collagen Type V Human genes 0.000 description 1
- 108010022514 Collagen Type V Proteins 0.000 description 1
- GUBGYTABKSRVRQ-CUHNMECISA-N D-Cellobiose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-CUHNMECISA-N 0.000 description 1
- ZAQJHHRNXZUBTE-NQXXGFSBSA-N D-ribulose Chemical compound OC[C@@H](O)[C@@H](O)C(=O)CO ZAQJHHRNXZUBTE-NQXXGFSBSA-N 0.000 description 1
- ZAQJHHRNXZUBTE-UHFFFAOYSA-N D-threo-2-Pentulose Natural products OCC(O)C(O)C(=O)CO ZAQJHHRNXZUBTE-UHFFFAOYSA-N 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 108010094028 Prothrombin Proteins 0.000 description 1
- 102100027378 Prothrombin Human genes 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 230000032900 absorption of visible light Effects 0.000 description 1
- 230000004931 aggregating effect Effects 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- PYMYPHUHKUWMLA-WDCZJNDASA-N arabinose Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)C=O PYMYPHUHKUWMLA-WDCZJNDASA-N 0.000 description 1
- PYMYPHUHKUWMLA-UHFFFAOYSA-N arabinose Natural products OCC(O)C(O)C(O)C=O PYMYPHUHKUWMLA-UHFFFAOYSA-N 0.000 description 1
- SRBFZHDQGSBBOR-UHFFFAOYSA-N beta-D-Pyranose-Lyxose Natural products OC1COC(O)C(O)C1O SRBFZHDQGSBBOR-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- 230000010109 chemoembolization Effects 0.000 description 1
- 229940096422 collagen type i Drugs 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 230000001079 digestive effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 210000001156 gastric mucosa Anatomy 0.000 description 1
- 238000002695 general anesthesia Methods 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 229920001600 hydrophobic polymer Polymers 0.000 description 1
- 239000003350 kerosene Substances 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- JXTPJDDICSTXJX-UHFFFAOYSA-N n-Triacontane Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCC JXTPJDDICSTXJX-UHFFFAOYSA-N 0.000 description 1
- 239000002120 nanofilm Substances 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 229940039716 prothrombin Drugs 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 238000004062 sedimentation Methods 0.000 description 1
- 238000004088 simulation Methods 0.000 description 1
- 229940083466 soybean lecithin Drugs 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 229940032094 squalane Drugs 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000009210 therapy by ultrasound Methods 0.000 description 1
- 238000002834 transmittance Methods 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/0013—Luminescence
- A61K49/0017—Fluorescence in vivo
- A61K49/0019—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules
- A61K49/0021—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules the fluorescent group being a small organic molecule
- A61K49/0032—Methine dyes, e.g. cyanine dyes
- A61K49/0034—Indocyanine green, i.e. ICG, cardiogreen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/0002—General or multifunctional contrast agents, e.g. chelated agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/0013—Luminescence
- A61K49/0017—Fluorescence in vivo
- A61K49/0019—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules
- A61K49/0021—Fluorescence in vivo characterised by the fluorescent group, e.g. oligomeric, polymeric or dendritic molecules the fluorescent group being a small organic molecule
- A61K49/0023—Di-or triarylmethane dye
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/006—Biological staining of tissues in vivo, e.g. methylene blue or toluidine blue O administered in the buccal area to detect epithelial cancer cells, dyes used for delineating tissues during surgery
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/001—Preparation for luminescence or biological staining
- A61K49/0063—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres
- A61K49/0069—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres the agent being in a particular physical galenical form
- A61K49/0076—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres the agent being in a particular physical galenical form dispersion, suspension, e.g. particles in a liquid, colloid, emulsion
- A61K49/0084—Preparation for luminescence or biological staining characterised by a special physical or galenical form, e.g. emulsions, microspheres the agent being in a particular physical galenical form dispersion, suspension, e.g. particles in a liquid, colloid, emulsion liposome, i.e. bilayered vesicular structure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/04—X-ray contrast preparations
- A61K49/0433—X-ray contrast preparations containing an organic halogenated X-ray contrast-enhancing agent
- A61K49/0438—Organic X-ray contrast-enhancing agent comprising an iodinated group or an iodine atom, e.g. iopamidol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/04—X-ray contrast preparations
- A61K49/0433—X-ray contrast preparations containing an organic halogenated X-ray contrast-enhancing agent
- A61K49/0447—Physical forms of mixtures of two different X-ray contrast-enhancing agents, containing at least one X-ray contrast-enhancing agent which is a halogenated organic compound
- A61K49/0461—Dispersions, colloids, emulsions or suspensions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/04—X-ray contrast preparations
- A61K49/0433—X-ray contrast preparations containing an organic halogenated X-ray contrast-enhancing agent
- A61K49/0447—Physical forms of mixtures of two different X-ray contrast-enhancing agents, containing at least one X-ray contrast-enhancing agent which is a halogenated organic compound
- A61K49/0461—Dispersions, colloids, emulsions or suspensions
- A61K49/0466—Liposomes, lipoprotein vesicles, e.g. HDL or LDL lipoproteins, phospholipidic or polymeric micelles
Abstract
Description
(医療用組織マーカー)
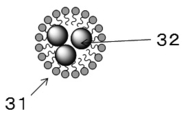
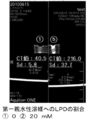
本実施形態に係る医療用組織マーカーは、リン脂質と近赤外蛍光色素が複合して形成されたベシクルと、リン脂質とX線造影剤とが複合して形成されたエマルションを親水性溶媒に内包させ、乳化剤により複数のカプセルを形成、凝集させたクラスター(以下「クラスター」という。)を有する。図1は、本実施形態に係る医療用組織マーカーにおけるクラスター1のイメージ図であり、図2は、クラスター1に含まれるベシクル2のイメージ図であり、図3は、クラスター1に含まれるエマルション3のイメージ図である。
ここで、上記医療用組織マーカーの製造方法(以下「本製造方法」という。)の一例について、詳細に説明する。図4は、本製造方法の概略を示す図である。
(医療用組織マーカー)
本実施形態に係る医療用組織マーカーは、上記実施形態1とほぼ同じであるが、疎水性溶媒に、第一の親水性溶媒、乳化剤を加えて懸濁液を形成する際、疎水性溶媒にX線造影剤を加えている点が実施形態1と異なる。異なる点について以下説明する。
ここで本実施形態に係る医療用組織マーカーの製造方法について説明する。本実施形態におけるクラスターの製造方法も、ほぼ実施形態1と同様であるが、上記実施形態の(2)疎水性溶媒に、上記第一の親水性溶媒と乳化剤を加えて懸濁液を形成する工程が少し異なる。具体的には、疎水性溶媒に第一の親水性溶媒、乳化剤を加えて懸濁液を形成する際、X線造影剤も加えておく点が実施形態1と異なる。
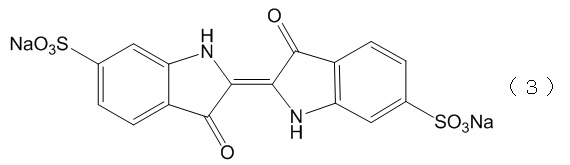
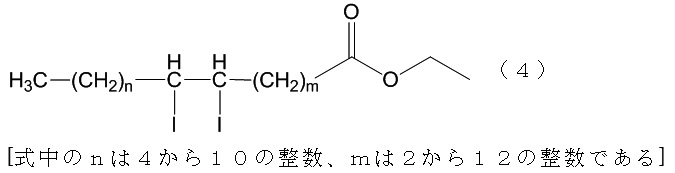
本実施例では、第一の親水性溶媒としてTRIS塩酸緩衝液を、近赤外蛍光色素としてインドシアニングリーン(以下「ICG」という。)を、X線造影剤として、ヨード化ケシ油エチルエステル(以下「LPG」という。)を、リン脂質として卵黄レシチンを、採用した。なお、この場合における増粘剤として、スクロースを採用した。
本実施例では、第一の親水性溶媒にだけLPGをいれた以外は上記実施例1と同様の材料、方法を用いて医療用組織マーカーを作製した。以下異なる点を中心に説明する。
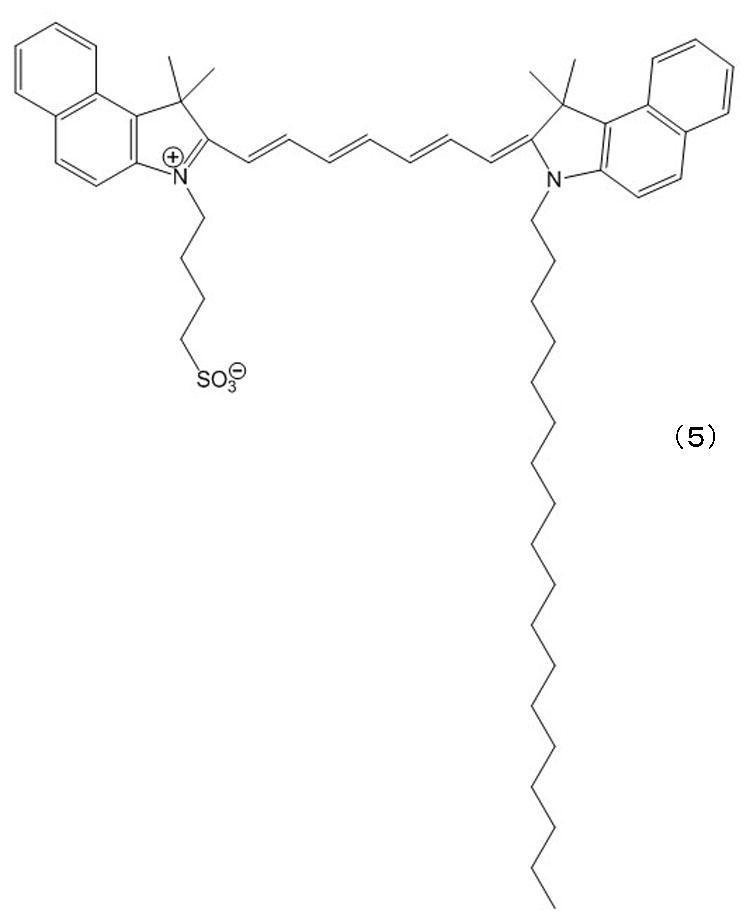
本実施例では、ICGの誘導体である下記式(5)で示されるICG-8を用いた医療用組織マーカーを以下の手順で作製した。まず、室温下において、ガラス管に、50mM、pH7.8となるようTRIS緩衝液を1mL準備し、そこにICG-8 3.2×10-2mM、LPD40mg/mL、卵黄レシチン30mMとなるよう加え、攪拌し、ベシクル及びエマルションを形成した。
Claims (7)
- リン脂質及び近赤外蛍光色素が複合して形成されたベシクルと、前記リン脂質及びX線造影剤が複合して形成されたエマルションを含み、前記ベシクルと前記エマルションを親水性溶媒に内包させ、乳化剤により複数のカプセルを形成、凝集させたクラスターを有する医療用組織マーカー。
- 前記X線造影剤は、ヨード化ケシ油エチルエステルを含む請求項1記載の医療用組織マーカー。
- 前記リン脂質は、レシチン及びフォスファチジルコリンの少なくともいずれかである請求項1記載の医療用組織マーカー。
- 前記親水性溶媒は、水と食用増粘剤を含む、請求項1記載の医療用組織マーカー。
- 第一の親水性溶媒に近赤外蛍光色素、X線造影剤及びリン脂質を加えて攪拌し、
疎水性溶媒に、前記第一の親水性溶媒、乳化剤を加えて懸濁液を形成し、
前記懸濁液と第二の親水性溶媒とを用いて遠心分離する、医療用組織マーカーの製造方法。 - 前記疎水性溶媒に、前記第一の親水性溶媒、乳化剤を加えて懸濁液を形成する際、X線造影剤も加える請求項5記載の医療用組織マーカーの製造方法。
- 前記X線造影剤は、ヨード化ケシ油エチルエステルを含む請求項5記載の医療用組織マーカーの製造方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP12801298.6A EP2719398B1 (en) | 2011-06-13 | 2012-05-31 | Medical tissue-marker and manufacturing method for same |
| US14/126,034 US10022459B2 (en) | 2011-06-13 | 2012-05-31 | Medical tissue-marker and manufacturing method for the same |
| CA2839200A CA2839200C (en) | 2011-06-13 | 2012-05-31 | Medical tissue-marker and manufacturing method for same |
| JP2013520507A JP5959118B2 (ja) | 2011-06-13 | 2012-05-31 | 医療用組織マーカー及びその製造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011130901 | 2011-06-13 | ||
| JP2011-130901 | 2011-06-13 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2012173003A2 true WO2012173003A2 (ja) | 2012-12-20 |
| WO2012173003A3 WO2012173003A3 (ja) | 2013-02-14 |
Family
ID=47357558
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/064235 WO2012173003A2 (ja) | 2011-06-13 | 2012-05-31 | 医療用組織マーカー及びその製造方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US10022459B2 (ja) |
| EP (1) | EP2719398B1 (ja) |
| JP (1) | JP5959118B2 (ja) |
| CA (1) | CA2839200C (ja) |
| WO (1) | WO2012173003A2 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015118113A1 (fr) * | 2014-02-07 | 2015-08-13 | Guerbet | Composition destinée à vectoriser un agent anticancéreux |
| US10716861B2 (en) | 2015-08-04 | 2020-07-21 | Guerbet | Composition intended to vectorise an anti-cancer agent |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7048096B2 (ja) | 2018-11-14 | 2022-04-05 | 株式会社日本医療機器開発機構 | 開窓用部分を有するステントグラフト |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007262062A (ja) | 2006-03-03 | 2007-10-11 | Koyo Chemical Kk | 組織マーカー |
| JP2008069107A (ja) | 2006-09-14 | 2008-03-27 | Pentax Corp | 内視鏡用組織蛍光染色剤 |
| JP2010266295A (ja) | 2009-05-13 | 2010-11-25 | Chiba Univ | 蛍光組織マーカー及びその製造方法 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DK175531B1 (da) * | 1986-12-15 | 2004-11-22 | Nexstar Pharmaceuticals Inc | Leveringsvehikel med amphiphil-associeret aktiv bestanddel |
| ATE89171T1 (de) * | 1987-06-11 | 1993-05-15 | Kabi Pharmacia Ab | Jod enthaltende emulsion. |
| JP2005220045A (ja) * | 2004-02-04 | 2005-08-18 | Konica Minolta Medical & Graphic Inc | 蛍光造影剤 |
| JP2005263647A (ja) * | 2004-03-16 | 2005-09-29 | Konica Minolta Medical & Graphic Inc | エマルション粒子含有造影剤 |
-
2012
- 2012-05-31 EP EP12801298.6A patent/EP2719398B1/en not_active Not-in-force
- 2012-05-31 CA CA2839200A patent/CA2839200C/en not_active Expired - Fee Related
- 2012-05-31 US US14/126,034 patent/US10022459B2/en active Active
- 2012-05-31 WO PCT/JP2012/064235 patent/WO2012173003A2/ja active Application Filing
- 2012-05-31 JP JP2013520507A patent/JP5959118B2/ja active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007262062A (ja) | 2006-03-03 | 2007-10-11 | Koyo Chemical Kk | 組織マーカー |
| JP2008069107A (ja) | 2006-09-14 | 2008-03-27 | Pentax Corp | 内視鏡用組織蛍光染色剤 |
| JP2010266295A (ja) | 2009-05-13 | 2010-11-25 | Chiba Univ | 蛍光組織マーカー及びその製造方法 |
Non-Patent Citations (8)
| Title |
|---|
| "Intermedia", 2008, article "All about ICG fluorescent Navigation Surgery" |
| AHKOH SEIHIRO: "basic research for hepatic artery chemoembolotherapy using lipiodol emulsion mixed with lecithin", TOKYO MEDICAL WOMEN'S COLLEGE MAGAZINE, vol. 60, 1990, pages 999 - 1010 |
| J. V. FRANGIONI, CURRENT OPINION IN CHEMICAL BIOLOGY, vol. 7, 2003, pages 626 - 634 |
| R. ASHIDA ET AL., ENDOSCOPY, vol. 38, 2006, pages 190 - 192 |
| S. ITO ET AL., ENDOSCOPY, vol. 33, 2001, pages 849 - 853 |
| S. TAOKA ET AL., DIGESTIVE ENDOSCOPY, vol. 11, 1999, pages 321 - 326 |
| S. YONEYA ET AL., INVESTIGATIVE OPHTHALMOLOGY AND VISUAL SCIENCE, vol. 39, 1998, pages 1286 - 1290 |
| See also references of EP2719398A4 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015118113A1 (fr) * | 2014-02-07 | 2015-08-13 | Guerbet | Composition destinée à vectoriser un agent anticancéreux |
| FR3017295A1 (fr) * | 2014-02-07 | 2015-08-14 | Guerbet Sa | Composition destinee a vectoriser un agent anticancereux |
| KR20160130992A (ko) * | 2014-02-07 | 2016-11-15 | 게르브 | 항암제를 벡터화하기 위한 조성물 |
| KR102396686B1 (ko) | 2014-02-07 | 2022-05-11 | 게르브 | 항암제를 벡터화하기 위한 조성물 |
| US10716861B2 (en) | 2015-08-04 | 2020-07-21 | Guerbet | Composition intended to vectorise an anti-cancer agent |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2012173003A1 (ja) | 2015-02-23 |
| US10022459B2 (en) | 2018-07-17 |
| WO2012173003A3 (ja) | 2013-02-14 |
| EP2719398A2 (en) | 2014-04-16 |
| CA2839200C (en) | 2016-06-14 |
| US20140219924A1 (en) | 2014-08-07 |
| EP2719398B1 (en) | 2016-01-20 |
| EP2719398A4 (en) | 2014-12-31 |
| JP5959118B2 (ja) | 2016-08-02 |
| CA2839200A1 (en) | 2012-12-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11344632B2 (en) | Indocyanine compound, synthesis method and purification method thereof, diagnostic composition using the indocyanine compound, and device for measuring biokinetics and device for visualizing circulation using the diagnostic composition | |
| JP7312822B2 (ja) | 注入型ヒドロゲルシステムおよび病変の標識方法 | |
| CA2733959C (en) | Nanocrystal nano-emulsion | |
| CA2733942C (en) | Fluorescent emulsion of indocyanine green | |
| JP5959118B2 (ja) | 医療用組織マーカー及びその製造方法 | |
| JP6721907B2 (ja) | 生体分解性腫瘍封止剤 | |
| JP2010266295A (ja) | 蛍光組織マーカー及びその製造方法 | |
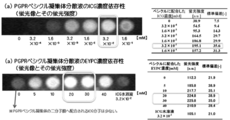
| WO2013109963A1 (en) | Fluorescent compositions with enhanced fluorescence and methods based thereon | |
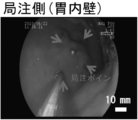
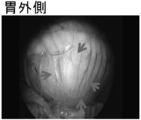
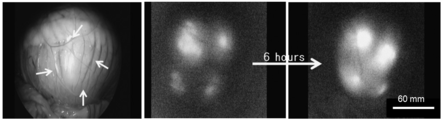
| Hatayama et al. | Application of a novel near infrared-fluorescence giant vesicle-and polymerasome-based tissue marker for endoscopic and laparoscopic navigation | |
| KR101571149B1 (ko) | 근적외선 형광다이를 함유하는 요오드화 오일 유제 및 이의 이용 | |
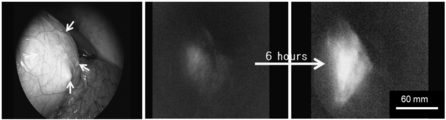
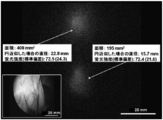
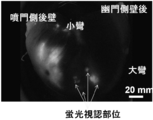
| Hayashi et al. | Development of a non-blurring, dual-imaging tissue marker for gastrointestinal tumor localization | |
| Ma et al. | High-precision detection and navigation surgery of colorectal cancer micrometastases | |
| CN104125825B (zh) | 用于诊断和治疗激素依赖性癌症及合成甾族激素的器官的癌症的制剂 | |
| JP5765695B2 (ja) | 蛍光組織マーカー及びその製造方法 | |
| KR102449537B1 (ko) | 근적외선을 이용하여 진단 생검 또는 종양 제거 수술 중에 종양 식별과 절제면 구분을 위한 종양 표적 진단 조영제 | |
| JP2005263647A (ja) | エマルション粒子含有造影剤 | |
| US20140065067A1 (en) | Radiopaque iodinated products intended for use medical imaging and their methods | |
| Mendez et al. | Short-Wave Infrared Emitting Nanocomposites for Fluorescence-Guided Surgery | |
| JP4990137B2 (ja) | 中性子捕捉製剤およびその用途 | |
| Kaliyaperumal et al. | Construct Fluorescent Solid Lipid Nanoparticles from Bacterial Outer Membrane Vesicles to Study their Properties and Potential Applications as In Vivo and Invitro Imaging Agents | |
| CN115252825A (zh) | 一种胶质瘤细胞膜包覆的聚合物点探针及其制备方法与应用 | |
| Hayashi et al. | Development of a Non-blurring, Dual-Imaging (X-Ray | |
| Class et al. | Patent application title: FLUORESCENT COMPOSITIONS WITH ENHANCED FLUORESCENCE AND METHODS BASED THEREON Inventors: Jay E. Reeder (Syracuse, NY, US) Jed-Sian Cheng (Syracuse, NY, US) Yachao Zhang (Syracuse, NY, US) Ronald W. Wood (Rochester, NY, US) Assignees: The Research Foundation of the State of University of New York | |
| KR20160150080A (ko) | 광음향 조영효과 및 항암효과를 나타내는 알부민 나노입자 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12801298 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2013520507 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2839200 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012801298 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14126034 Country of ref document: US |