WO2011127051A1 - Modulators of the gpr119 receptor and the treatment of disorders related thereto - Google Patents
Modulators of the gpr119 receptor and the treatment of disorders related thereto Download PDFInfo
- Publication number
- WO2011127051A1 WO2011127051A1 PCT/US2011/031243 US2011031243W WO2011127051A1 WO 2011127051 A1 WO2011127051 A1 WO 2011127051A1 US 2011031243 W US2011031243 W US 2011031243W WO 2011127051 A1 WO2011127051 A1 WO 2011127051A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- cyclohexyloxy
- alkyl
- methylsulfonyl
- yloxy
- Prior art date
Links
- 0 *CC*1(C(*)**2=CC1)C(*)C2O[C@](CC1)CC[C@]1OC1CCN(*)CC1 Chemical compound *CC*1(C(*)**2=CC1)C(*)C2O[C@](CC1)CC[C@]1OC1CCN(*)CC1 0.000 description 6
- WIGIZIANZCJQQY-RUCARUNLSA-N CCC1=C(C)CN(C(NCCc(cc2)ccc2S(NC(N[C@H]2CC[C@H](C)CC2)=O)(=O)=O)=O)C1=O Chemical compound CCC1=C(C)CN(C(NCCc(cc2)ccc2S(NC(N[C@H]2CC[C@H](C)CC2)=O)(=O)=O)=O)C1=O WIGIZIANZCJQQY-RUCARUNLSA-N 0.000 description 1
- IWYJYHUNXVAVAA-OAHLLOKOSA-N CN(C(C=C(N1Cc2cc(F)ccc2C#N)N(CCC2)C[C@@H]2N)=O)C1=O Chemical compound CN(C(C=C(N1Cc2cc(F)ccc2C#N)N(CCC2)C[C@@H]2N)=O)C1=O IWYJYHUNXVAVAA-OAHLLOKOSA-N 0.000 description 1
- NVSWJKWHLUTHLP-CPJSRVTESA-N C[C@@H](CC(c1nnn[nH]1)(c(c(CC1)c2)ccc2C(N(C)C)=O)c(cc2)c1cc2C(N(C)C)=O)NCC(N(CCC1)[C@@H]1C#N)=O Chemical compound C[C@@H](CC(c1nnn[nH]1)(c(c(CC1)c2)ccc2C(N(C)C)=O)c(cc2)c1cc2C(N(C)C)=O)NCC(N(CCC1)[C@@H]1C#N)=O NVSWJKWHLUTHLP-CPJSRVTESA-N 0.000 description 1
- OUDSBRTVNLOZBN-UHFFFAOYSA-N Cc(cc1)ccc1S(NC(NN1CCCCCC1)=O)(=O)=O Chemical compound Cc(cc1)ccc1S(NC(NN1CCCCCC1)=O)(=O)=O OUDSBRTVNLOZBN-UHFFFAOYSA-N 0.000 description 1
- UOCHFMVPMIYRHJ-QWRGUYRKSA-N N[C@@H](CN(C1)c2ncnc(N(CC3)CC3(F)F)n2)[C@H]1N(CC(CC1)(F)F)C1=O Chemical compound N[C@@H](CN(C1)c2ncnc(N(CC3)CC3(F)F)n2)[C@H]1N(CC(CC1)(F)F)C1=O UOCHFMVPMIYRHJ-QWRGUYRKSA-N 0.000 description 1
- QWEWGXUTRTXFRF-KBPBESRZSA-N O=C([C@H](C1)NC[C@H]1N(CC1)CCN1c1ncccn1)N(CC1)CC1(F)F Chemical compound O=C([C@H](C1)NC[C@H]1N(CC1)CCN1c1ncccn1)N(CC1)CC1(F)F QWEWGXUTRTXFRF-KBPBESRZSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/36—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D211/40—Oxygen atoms
- C07D211/44—Oxygen atoms attached in position 4
- C07D211/46—Oxygen atoms attached in position 4 having a hydrogen atom as the second substituent in position 4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
Definitions
- the present invention relates to compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, that are useful as a single agent or in combination with one or more additional pharmacetical agents, such as, an inhibitor of DPP-IV, a biguanide, an SGLT2 inhibitor, or an alpha-glucosidase inhibitor, in the treatment of, for example, a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level; a metabolic-related disorder; type 2 diabetes; obesity; and complications related thereto.
- a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level; a metabolic-related disorder; type 2 diabetes; obesity; and complications related thereto.
- Diabetes mellitus is a serious disease afflicting over 100 million people worldwide.
- Diabetes mellitus is a serious disease afflicting over 100 million people worldwide.
- Diabetes mellitus is a diagnostic term for a group of disorders characterized by abnormal glucose homeostasis resulting in elevated blood sugar.
- IDDM insulin-dependent diabetes mellitus
- NIDDM non-insulin-dependent diabetes mellitus
- the etiology of the different types of diabetes is not the same; however, everyone with diabetes has two things in common: overproduction of glucose by the liver and little or no ability to move glucose out of the blood into the cells where it becomes the body's primary fuel.
- Diabetes is a syndrome with interrelated metabolic, vascular, and neuropathic components.
- the metabolic syndrome generally characterized by hyperglycemia, comprises alterations in carbohydrate, fat and protein metabolism caused by absent or markedly reduced insulin secretion and/or ineffective insulin action.
- the vascular syndrome consists of abnormalities in the blood vessels leading to cardiovascular, retinal and renal complications. Abnormalities in the peripheral and autonomic nervous systems are also part of the diabetic syndrome.
- IDDM is characterized by low or undetectable levels of endogenous insulin production caused by destruction of the insulin-producing ⁇ cells of the pancreas, the characteristic that most readily distinguishes IDDM from NIDDM. IDDM, once termed juvenile-onset diabetes, strikes young and older adults alike.
- NIDDM type 2
- NIDDM Approximately 90 to 95% of people with diabetes have type 2 (or NIDDM). NIDDM subjects produce insulin, but the cells in their bodies are insulin resistant: the cells don't respond properly to the hormone, so glucose accumulates in their blood. NIDDM is characterized by a relative disparity between endogenous insulin production and insulin requirements, leading to elevated blood glucose levels. In contrast to IDDM, there is always some endogenous insulin production in NIDDM; many NIDDM patients have normal or even elevated blood insulin levels, while other NIDDM patients have inadequate insulin production (Rotwein, R. et al. N. Engl. J. Med. 308, 65-71 (1983)). Most people diagnosed with NIDDM are age 30 or older, and half of all new cases are age 55 and older. Compared with whites and Asians, NIDDM is more common among Native Americans, African- Americans, Latinos, and Hispanics. In addition, the onset can be insidious or even clinically inapparent, making diagnosis difficult.
- NIDDM neurodegenerative disease
- Kidney disease also called nephropathy
- Diabetes occurs when the kidney's "filter mechanism” is damaged and protein leaks into urine in excessive amounts and eventually the kidney fails. Diabetes is also a leading cause of damage to the retina at the back of the eye and increases risk of cataracts and glaucoma.
- diabetes is associated with nerve damage, especially in the legs and feet, which interferes with the ability to sense pain and contributes to serious infections. Taken together, diabetes complications are one of the nation's leading causes of death.
- Obesity and diabetes are among the most common human health problems in industrialized societies. In industrialized countries a third of the population is at least 20% overweight. In the United States, the percentage of obese people has increased from 25% at the end of the 1970's, to 33% at the beginning the 1990's. Obesity is one of the most important risk factors for NIDDM. Definitions of obesity differ, but in general, a subject weighing at least 20% more than the recommended weight for his/her height and build is considered obese. The risk of developing NIDDM is tripled in subjects 30% overweight, and three-quarters with NIDDM are overweight.
- Obesity which is the result of an imbalance between caloric intake and energy expenditure, is highly correlated with insulin resistance and diabetes in experimental animals and human.
- Whether someone is classified as overweight or obese can be determined by a number of different methods, such as, on the basis of their body mass index (BMI) which is calculated by dividing body weight (kg) by height squared (m 2 ).
- BMI body mass index
- m 2 height squared
- Overweight is defined as a BMI in the range 25-30 kg/m 2
- obesity as a BMI greater than 30 kg/m 2 (see TABLE below).
- body fat content greater than 25% and 30% in males and females, respectively.
- Coronary insufficiency, atheromatous disease, and cardiac insufficiency are at the forefront of the cardiovascular complication induced by obesity. It is estimated that if the entire population had an ideal weight, the risk of coronary insufficiency would decrease by 25% and the risk of cardiac insufficiency and of cerebral vascular accidents by 35%. The incidence of coronary diseases is doubled in subjects less than 50 years of age who are 30% overweight.
- Atherosclerosis is a complex disease characterized by inflammation, lipid accumulation, cell death and fibrosis. Atherosclerosis is characterized by cholesterol deposition and monocyte infiltration into the subendothelial space, resulting in foam cell formation. Thrombosis subsequent to atherosclerosis leads to myocardial infarction and stroke. Atherosclerosis is the leading cause of mortality in many countries, including the United States. (See, e.g., Ruggeri, Nat Med (2002) 8: 1227-1234; Arehart et al, Circ Res, Circ. Res. (2008) 102:986-993.)
- Osteoporosis is a disabling disease characterized by the loss of bone mass and microarchitectural deterioration of skeletal structure leading to compromised bone strength, which predisposes a patient to increased risk of fragility fractures. Osteoporosis affects more than 75 million people in Europe, Japan and the United States, and causes more than 2.3 million fractures in Europe and the United States alone. In the United States, osteoporosis affects at least 25% of all post-menopausal white women, and the proportion rises to 70% in women older than 80 years. One in three women older than 50 years will have an osteoporotic fracture that causes a considerable social and financial burden on society. The disease is not limited to women; older men also can be affected.
- IBD Inflammatory bowel disease
- Crohn's disease ulcerative colitis
- ulcerative proctitis U.S. medical costs of inflammatory bowel disease for 1990 have been estimated to be $1.4 to $1.8 billion. Lost productivity has been estimated to have added an additional $0.4 to $0.8 billion, making the estimated cost of inflammatory bowel disease $1.8 to $2.6 billion.
- Enteritis refers to inflammation of the intestine, especially the small intestine, a general condition that can have any of numerous different causes. Enterocolitis refers to inflammation of the small intestine and colon.
- CD Crohn's disease
- Ileitis is CD of the ileum which is the third part of the small intestine.
- Crohn's colitis is CD affecting part or all of the colon.
- Ulcerative colitis is an inflammatory disease of the large intestine, commonly called the colon. UC causes inflammation and ulceration of the inner lining of the colon and rectum. The inflammation of UC is usually most severe in the rectal area with severity diminishing (at a rate that varies from patient to patient) toward the cecum, where the large and small intestine join. Inflammation of the rectum is called proctitis. Inflammation of the sigmoid colon (located just above the rectum) is called sigmoiditis. Inflammation involving the entire colon is termed pancolitis. The inflammation causes the colon to empty frequently resulting in diarrhea. As the lining of the colon is destroyed ulcers form releasing mucus, pus and blood. Ulcerative proctitis is a form of UC that affects only the rectum.
- GPR119 is a G protein-coupled receptor (GPR119; e.g. , human GPR119, GenBank ®
- GPR119 activation leads to elevation of a level of intracellular cAMP, consistent with GPR119 being coupled to Gs.
- Agonists to GPR119 stimulate glucose-dependent insulin secretion in vitro and lower an elevated blood glucose level in vivo; see, e.g. , International Applications WO
- GPR119 has also been referred to as RUP3 (see, International Application WO 00/31258) and as Glucose-Dependent Insulinotropic Receptor GDIR (see, Jones, et. al. Expert Opin. Ther. Patents (2009), 19(10): 1339-1359).
- GPR119 agonists also stimulate the release of Glucose-dependent Insulinotropic Poloypeptide (GIP), Glucagon-Like Peptide-1 (GLP-1), and at least one other L-cell peptide, Peptide YY (PYY) (Jones, et. al. Expert Opin. Ther. Patents (2009), 19(10): 1339-1359); for specific references related to GPR119 agonists and the release of:
- GIP Insulinotropic Poloypeptide
- GLP-1 Glucagon-Like Peptide-1
- PYY Peptide YY
- GLP-1 see Shah, Current Opinion in Drug Discovery & Development, (2009) 12:519-532; Jones, et al., Ann. Rep. Med. Chem. , (2009) 44: 149-170; Schwartz et. al., Cell Metabolism, 2010, 11 :445-447; and WO 2006/076231 ; and
- GPR119 agonists enhance incretin release and therefore can be used in treatment of disorders related to the incretins, such as, GIP, GLP-1, and PYY.
- GIP and GLP-1 are substrates for the enzyme DPP-IV. Jones and co-workers (Jones, et al., Ann. Rep. Med. Chem.
- G Glucose-dependent Insulinotropic Poloypeptide
- GIP Glucose-dependent insulinotropic polypeptide
- gastric inhibitory polypeptide is a peptide incretin hormone of 42 amino acids that is released from duodenal endocrine K cells after meal ingestion. The amount of GIP released is largely dependent on the amount of glucose consumed. GIP has been shown to stimulate glucose-dependent insulin secretion in pancreatic beta cells. GIP mediates its actions through a specific G protein-coupled receptor, namely GIPR.
- GIP contains an alanine at position 2, it is an excellent substrate for dipeptidyl peptidase-4 (DPP-IV), an enzyme regulating the degradation of GIP.
- DPP-IV dipeptidyl peptidase-4
- Full-length GIP(l-42) is rapidly converted to bioinactive GIP(3-42) within minutes of secretion from the gut K cell. Inhibition of DPP-IV has been shown to augment GIP bioactivity.
- DPP-IV dipeptidyl peptidase-4
- Analysis of full length bioactive GIP, for example in blood, can be carried out using N-terminal-specific assays (see, e.g., Deacon et al, Clin Endocrinol Metab (2000) 85:3575-3581).
- GIP has been shown to promote bone formation.
- GIP has been shown to activate osteoblastic receptors, resulting in increases in collagen type I synthesis and alkaline phosphatase activity, both associated with bone formation.
- GIP has been shown to inhibit osteoclast activity and differentiation in vitro.
- GIP administration has been shown to prevent the bone loss due to ovariectomy.
- GIP receptor (GIPR) knockout mice evidence a decreased bone size, lower bone mass, altered bone microarchitecture and biochemical properties, and altered parameters for bone turnover, especially in bone formation.
- Patent No. 6,410,508 for the treatment of reduced bone mineralization by administration of GIP peptide.
- current GIP peptide agonists suffer from a lack of oral bioavailability, negatively impacting patient compliance.
- An attractive alternative approach is to develop an orally active composition for increasing an endogenous level of GIP activity.
- GLP-1 Glucagon-Like Peptide-1
- Glucagon-like peptide-1 (GLP-1) is an incretin hormone derived from the
- GLP-1 mediates its actions through a specific G protein-coupled receptor (GPCR), namely GLP-1R.
- GPCR G protein-coupled receptor
- GLP-1R G protein-coupled receptor
- GLP-1 is best characterized as a hormone that regulates glucose homeostasis.
- GLP-1 has been shown to stimulate glucose-dependent insulin secretion and to increase pancreatic beta cell mass.
- GLP-1 has also been shown to reduce the rate of gastric emptying and to promote satiety.
- GLP-1 peptide agonists in controlling blood glucose in Type 2 diabetics has been demonstrated in several clinical studies [see, e.g., Nauck et al., Drug News Perspect (2003) 16:413-422], as has its efficacy in reducing body mass [Zander et al., Lancet (2002) 359:824- 830].
- GLP-1 receptor agonists are additionally useful in protecting against myocardial infarction and against cognitive and neurodegenerative disorders.
- GLP-1 has been shown to be cardioprotective in a rat model of myocardial infarction [Bose et al., Diabetes (2005) 54: 146- 151], and GLP-1 R has been shown in rodent models to be involved in learning and
- GLP-1 peptide agonists suffer from a lack of oral bioavailability, negatively impacting patient compliance. Efforts to develop orally bioavailable non-peptidergic, small- molecule agonists of GLP-1 R have so far been unsuccessful [Mentlein, Expert Opin Investig Drugs (2005) 14:57-64]. An attractive alternative approach is to develop an orally active composition for increasing an endogenous level of GLP-1 in the blood.
- Peptide YY is a 36 amino acid peptide originally isolated in 1980 from porcine intestine (Tatemoto et al, Nature (1980) 285:417-418). PYY is secreted from enteroendocrine L- cells within both the large and small intestine.
- PYY expression in rat was reported to also extend to alpha cells of the islets of Langerhans and to cells in the medulla oblongata (Ekblad et al, Peptides (2002) 23:251-261 ; PYY is released into the circulation as PYYi_ 36 and PYY 3 . 36 (Eberlein et al, Peptides (1989) 10:797-803).
- PYY 3 - 36 is generated from PYYi_ 36 by cleavage of the N-terminal Tyr and Pro residues by dipeptidyl peptidase IV.
- PYY 3 . 36 is the predominant form of PYY in human postprandial plasma (Grandt et al, Regul. Pept.
- PYYi- 36 and PYY 3 . 36 have been reported to have comparable agonist activity at ⁇ Y2 receptor (Y2R), a G protein- coupled receptor (Parker et al, Br. J. Pharmacol. (2008) 153:420-431); however, PYY 3 . 36 has been reported to be a high-affinity Y2R selective agonist (Keire et al, Am. J. Physiol.
- mice peripheral administration (Morley et al, Life Sciences (1987) 41 :2157-2165).
- Peripheral administration of PYY 3 . 36 has been reported to markedly reduce food intake and weight gain in rats, to decrease appetite and food intake in humans, and to decrease food intake in mice, but not in Y2R-null mice, which was said to suggest that the food intake effect requires the Y2R.
- infusion of PYY 3 . 36 was found to significantly decrease appetite and reduce food intake by 33% over 24 hours.
- Peripheral administration of PYY 3 . 36 has been reported to reduce food intake, body weight gain and glycemic indices in diverse rodent models of metabolic diseases of both sexes (Pittner et al, Int. J. Obes. Relat. Metab. Disord. (2004) 28:963-971). It has been reported that blockade of Y2R with the specific antagonist BIIE-246 attenuates the effect of peripherally administered endogenous and exogenous PYY 3 _ 36 for reducing food intake (Abbott et al, Brain Res (2005) 1043: 139-144).
- peripheral administration of a novel long- acting selective Y2R polyethylene gly col-conjugated peptide agonist reduces food intake and improves glucose metabolism (glucose disposal, plasma insulin and plasma glucose) in rodents (Ortiz et al, JPET (2007) 323:692-700; Lamb et al, . Med. Chem. (2007) 50:2264-2268). It has been reported that PYY ablation in mice leads to the development of hyperinsulinemia and obesity (Boey et al, Diabetologia (2006) 49: 1360-1370). It has been reported that peripheral administration of a long-acting, potent and highly selective Y2R agonist inhibits food intake and promotes fat metabolism in mice (Balasubramaniam et al, Peptides (2007) 28:235-240).
- Y2R agonists such as PYY 1 36 and PYY 3 . 36 can confer protection against epileptic seizures, such as against kainate seizures (El Bahh et al, Eur. J.
- Y2R agonists such as PYY 1 36 and PYY 3 . 36 act as proabsorbtive (or anti-secretory) hormones, increasing upon intravenous administration the absorption of both water and sodium in various parts of the bowel (Bilchik et al, Gastroenterol. (1993) 105: 1441- 1448; Liu et al, . Surg. Res. (1995) 58:6-11 ; Nightingale et al, Gut (1996) 39:267-272; Liu et al, Am Surg (1996) 62:232-236; Balasubramaniam et al, . Med. Chem. (2000) 43:3420-3427). It has been reported that Y2R agonists such as PYY analogues inhibit secretion and promote absorption and growth in the intestinal epithelium (Balasubramaniam et al, . Med. Chem.
- Y2R agonists such as PYY 1 36 and PYY 3 . 36 can confer protection against inflammatory bowel disease such as ulcerative colitis and Crohn's disease (WO 03/105763). It has been reported that PYY-deficient mice exhibit an osteopenic phenotype, i.e. that PYY can increase bone mass and/or can confer protection against loss of bone mass (e.g., decreases loss of bone mass) (Wortley et al, Gastroenterol. (2007) 133: 1534-1543). It has been reported that PYY 3 . 36 can confer protection in rodent models of pancreatitis (Vona-Davis et al, Peptides (2007) 28:334-338).
- PYY and Y2R agonists such as PYY 3 . 36 can suppress tumor growth in the cases of, e.g., pancreatic cancer such as pancreatic ductal adenocarcinoma, breast cancer such as breast infiltrative ductal adenocarcinoma, colon cancer such as colon
- Adiponectin is an adipokine with potent anti-inflammatory properties (Ouchi et al, Clin Chim Acta (2007) 380:24-30; Tilg et al, Nat. Rev. Immunol. (2006) 6:772-783).
- Adiponectin exerts anti-atherogenic effects by targeting vascular endothelial cells and macrophages and insulin-sensitizing effects, predominantly in muscle and liver (Kubota et al, . Biol. Chem.
- adiponectin has been implicated in high density lipoprotein (HDL) assembly (Oku et al, FEBS Letters (2007) 581 :5029-5033).
- HDL high density lipoprotein
- Adiponectin has been found to ameliorate the abnormalities of metabolic syndrome, including insulin resistance, hyperglycemia, and dyslipidemia, in a mouse model of obesity-linked metabolic syndrome associated with decreased adiponectin levels (Hara et al, Diabetes Care (2006) 29: 1357-1362). Adiponectin has been reported to stimulate angiogenesis in response to tissue ischemia (Shibata et al, . Biol. Chem. (2004) 279:28670-28674).
- Adiponectin has been reported to prevent cerebral ischemic injury through endothelial nitric oxide synthase-dependent mechanisms (Nishimura et al, Circulation (2008) 117:216-223). Adiponectin has been reported to confer protection against myocardial ischemia-reperfusion injury (Shibata et al, Nat Med (2005) 11 : 1096-1103; Tao et al, Circulation (2007) 115: 1408- 1416). Adiponectin has been reported to confer protection against myocardial ischaemia- reperfusion injury via AMP-activated protein kinase, Akt, and nitric oxide (Gonon et al, Cardiovasc Res. (2008) 78: 116-122).
- Adiponectin has been reported to confer protection against the development of systolic dysfunction following myocardial infarction, through its abilities to suppress cardiac hypertrophy and interstitial fibrosis, and proctect against myocyte and capillary loss (Shibata et al, . Mol. Cell Cardiol. (2007) 42: 1065-1074). Adiponectin has been reported to confer protection against inflammatory lung disease; adiponectin-deficient mice exhibit an emphysema-like phenotype (Summer et al, Am J. Physiol. Lung Cell Mol. Physiol (March 7, 2008)).
- Adiponectin has been reported to confer protection against allergic airway inflammation and airway hyperresponsiveness such as may be associated with asthma (Shore et al, . Allergy Clin. Immunol (2006) 118:389-395). Adiponectin has been suggested to confer protection against pulmonary arterial hypertension by virtue of its insulin-sensitizing effects (Hansmann et al, Circulation (2007) 115: 1275-1284). Adiponectin has been reported to ameliorate obesity- related hypertension, with said amelioration of hypertension being associated in part with upregulated prostacyclin expression (Ohashi et al, Hypertension (2006) 47: 1108-1116).
- Adiponectin has been reported to decrease tumor necrosis factor (TNF)-a-induced expression of the adhesion molecules VCAM-1, E-selectin and ICAM-1 in human aortic endothelial cells (HAECs) (Ouchi et al, Circulation (1999) 100:2473-2476) and to inhibit production of TNF-a in macrophages (Yokota et al, Blood (2000) 96: 1723-1732). Adiponectin has been reported to confer protection against restenosis after vascular intervention (Matsuda et al, Biol Chem
- TNF-a-mediated inflammatory conditions encompass rheumatoid arthritis, inflammatory bowel disease such as Crohn's disease, ankylosing spondylitis, psoriasis, ischemic brain injury, cardiac allograft rejection, asthma, and the like (Bradley, Pathol (2008) 214: 149-160). See, e.g.,
- One aspect of the present invention is directed to compounds, as described herein, and pharmaceutically acceptable salts, solvates, and hydrates thereof, which bind to and modulate the activity of a GPCR, referred to herein as GPR119, and uses thereof.
- One aspect of the present invention encompasses, inter alia, certain cyclohexane derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N or CR 4 ;
- Z is N or CR 5 ;
- X is N, N(O), or CR 6 ;
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , C(0)OR 9 , and C(0)SR 9 ; or R 1 is selected from the group consisting of heteroaryl and phenyl, each optionally substituted with one or more substituents selected independently from the group consisting of C 2 -C6 alkenyl, C 1 -C6 alkoxy, C 1 -C6 alkyl, halogen, C 1 -C6 haloalkoxy, and C 1 -C6 haloalkyl;
- R 2 is selected from the group consisting of H, C 1 -C6 alkyl, cyano, C3-C6 cycloalkyl, halogen, C 1 -C6 haloalkyl, heteroaryl, heterocyclyl, S(0) n R n , and C(0)NR 12 R 13 ; wherein said C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-Cs alkylsulfonyl, cyano, and C(0)NR 12 R 13 ; said C 3 -C 6 cycloalkyl is optionally substituted with C(0)NR 12 R 13 ; said heteroaryl is optionally substituted with C 1 -C6 alkyl; and said heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-C 6 alkylsulfonyl, hydroxyl, and halogen;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, Q- C 6 alkyl, Ci-C 6 alkylsulfonyl, and halogen;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein said C 3 -C 6 cycloalkyl is optionally substituted with one or more Ci-C 6 alkyl;
- R 8 is selected from the group consisting of C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, heteroaryl, phenyl, and C(0)OR 9 ; wherein said C 3 -C 6 cycloalkyl and said heteroaryl are each optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 haloalkyl and C 1 -C6 alkyl;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C6 cycloalkyl, C 1 -C6 haloalkyl, heterocyclyl, and phenyl; said C 1 -C6 alkyl and said C 3 -C6 cycloalkyl are each optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl, halogen, hydroxyl, C 1 -C6 alkoxy, and R 10 , wherein said C 1 -C6 alkoxy is optionally substituted with phenyl;
- R 10 is heterocyclyl optionally substituted with C 1 -C6 alkyl
- R 11 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C 6 cycloalkyl, C 1 -C6 haloalkyl, and heterocyclyl; wherein said C 1 -C6 alkyl and heterocyclyl are each optionally substituted with one or more substituents selected independently from the group consisting of halogen, hydroxyl, and NR 12 R 13 ; and said heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-C 6 alkyl, and hydroxyl;
- R 12 and R 13 are each independently selected from the group consisting of H, Ci-C 6 alkyl, and C 3 -C 6 cycloalkyl; wherein said Ci-C 6 alkyl is optionally substituted with hydroxyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of cyano, halogen, hydroxyl, and C 1 -C6 alkoxy; and
- n 0, 1, or 2.
- compositions comprising a compound of the present invention.
- compositions comprising a compound of the present invention and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to methods for preparing a composition comprising the step of admixing a compound of the present invention and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to pharmaceutical products selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention.
- compositions comprising a compound of the present invention and a second pharmaceutical agent.
- One aspect of the present invention pertains to methods for preparing a composition comprising the step of admixing a compound of the present invention and a second
- compositions comprising a compound of the present invention, a second pharmaceutical agent, and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to methods for preparing a composition
- methods for preparing a composition comprising the step of admixing a compound of the present invention, a second pharmaceutical agent, and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to compositions obtained by the methods of the present invention as described herein.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention and a second pharmaceutical agent.
- One aspect of the present invention pertains to methods for modulating the activity of a GPRl 19 receptor, comprising administering to an individual in need thereof, a therapeutically effective amount of: a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention.
- One aspect of the present invention pertains to the use of a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention; in the manufacture of a medicament for modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention; for use in a method of treating the human or animal by therapy.
- One aspect of the present invention pertains to a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention; for use in a method of modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention; for use in a method of treating the human or animal by therapy.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention; for modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, and pharmaceutical products, as described herein, for agonizing the GPRl 19 receptor.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, and pharmaceutical products, as described herein, increasing the secretion of an incretin.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, and pharmaceutical products, as described herein, increasing a blood incretin level.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, and pharmaceutical products, as described herein, treating a disorder, wherein the disorder is selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level; a condition characterized by low bone mass; a neurological disorder; a metabolic-related disorder; and obesity.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, and pharmaceutical products, as described herein, in combination with a second pharmaceutical agent.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, and pharmaceutical products, as described herein, in combination with a second pharmaceutical agent, wherein the second pharmaceutical agent is selected from: an inhibitor of DPP-IV, a biguanide, an alpha-glucosidase inhibitor, an insulin analogue, a sulfonylurea, a SGLT2 inhibitor, a meglitinide, a thiazolidinedione, and an anti-diabetic peptide analogue.
- Figure 1 shows the in vivo effects of Compound 28 on glucose homeostasis in male diabetic ZDF rats (oral glucose tolerance test (oGTT)).
- Figure 2 shows the in vivo effects of Compound 28 on percent glycemic inhibition in male diabetic ZDF rats.
- Figure 3 shows the in vivo effects of Compound 62 on glucose homeostasis in male diabetic ZDF rats (oral glucose tolerance test (oGTT)).
- Figure 4 shows the in vivo effects of Compound 62 on percent glycemic inhibition in male diabetic ZDF rats.
- Figure 5 shows the in vivo effects of Compound 28 on glucose homeostasis in male 129SVE mice (oral glucose tolerance test (oGTT)).
- Figure 6 shows the in vivo effects of Compound 62 on percent glycemic inhibition in male 129SVE mice.
- Figure 7 shows the in vivo effects of representative compounds of the present invention on incretin hormone GIP release.
- Figure 8 shows a general synthetic scheme for the preparation of compounds of Formula (la) utilizing cyclohexane-l,4-diol as the starting material. It is understood that Is, 4s (i.e., cis), ⁇ r,4r (i.e., trans), or a mixture of ⁇ s, s and lr,4r (i.e., cis and trans) cyclohexane-1,4- diol can be used in the preparation of compounds of Formula (la).
- Figure 9 shows a general synthetic method for the preparation of the useful intermediate teri-butyl 4-(4-hydroxycyclohexyl oxy)piperidine-l-carboxylate as substantially pure ⁇ s, s (i.e., cis), and substantially stereochemically pure lr,4r (i.e., trans).
- Figure 10 shows a general synthetic method for the preparation of useful intermediates ( 1 s,4s)A-( 1 -methylpiperidin-4-yloxy)cyclohexanol (i.e. , cis)and ( 1 r,4r)-4-( 1 -methylpiperidin-4- yloxy)cyclohexanol (i.e., trans), see Example 1.114.
- Figure 11 shows a general synthetic scheme for the preparation of intermediates that are useful in the synthesis of compounds of Formula (la).
- Figure 12 shows a general synthetic scheme for the preparation of certain compounds of Formula (la), wherein R 1 is an optionally substituted oxadiazolyl group.
- Figure 13 shows a general synthetic scheme for the preparation of certain compounds of Formula (la), wherein R 1 is -S(0) 2 R 7 , -C(0)R 7 , or -CH 2 R 8 .
- Figure 14 shows a general synthetic scheme for the preparation of certain compounds of Formula (la), wherein R 1 is C(0)OR 9 , an optionally substituted heteroaryl, or an optionally substituted phenyl.
- Figure 15 shows a general synthetic scheme for the preparation of lr,4r (i.e., trans) compounds of Formula (la), ls,4s (i-e., cis) compounds of Formula (la) can be prepared in an analogous manner with the exception that Method A would be used with (1 ⁇ ,4 ⁇ )-4-(1- methylpiperidin-4-yloxy)cyclohexanol to retain the cis stereochemistry while Method B would be used with (lr,4r)-4-(l-methylpiperidin-4-yloxy)cyclohexanol to invent the stereocenter thus providing the cis stereochemistry.
- Method A would be used with (1 ⁇ ,4 ⁇ )-4-(1- methylpiperidin-4-yloxy)cyclohexanol to retain the cis stereochemistry
- Method B would be used with (lr,4r)-4-(l-methylpiperidin-4-yloxy)cyclohexanol to invent the stereocenter thus providing the cis
- Figure 16 shows the in vivo effects of Compound 83 on glucose homeostasis in male diabetic ZDF rats (oral glucose tolerance test (oGTT)).
- Figure 17 shows the in vivo effects of Compound 83 on percent glycemic inhibition in male diabetic ZDF rats.
- Figure 18 shows a powder X-ray diffraction (PXRD) pattern for Compound 28.
- Figure 19 shows two powder X-ray diffraction (PXRD) patterns for Compound 83, one sample prepared from a slurry in ethanol and a second sample that was ground prior to PXRD analysis.
- PXRD powder X-ray diffraction
- Figure 20 shows a powder X-ray diffraction (PXRD) pattern for Compound 85.
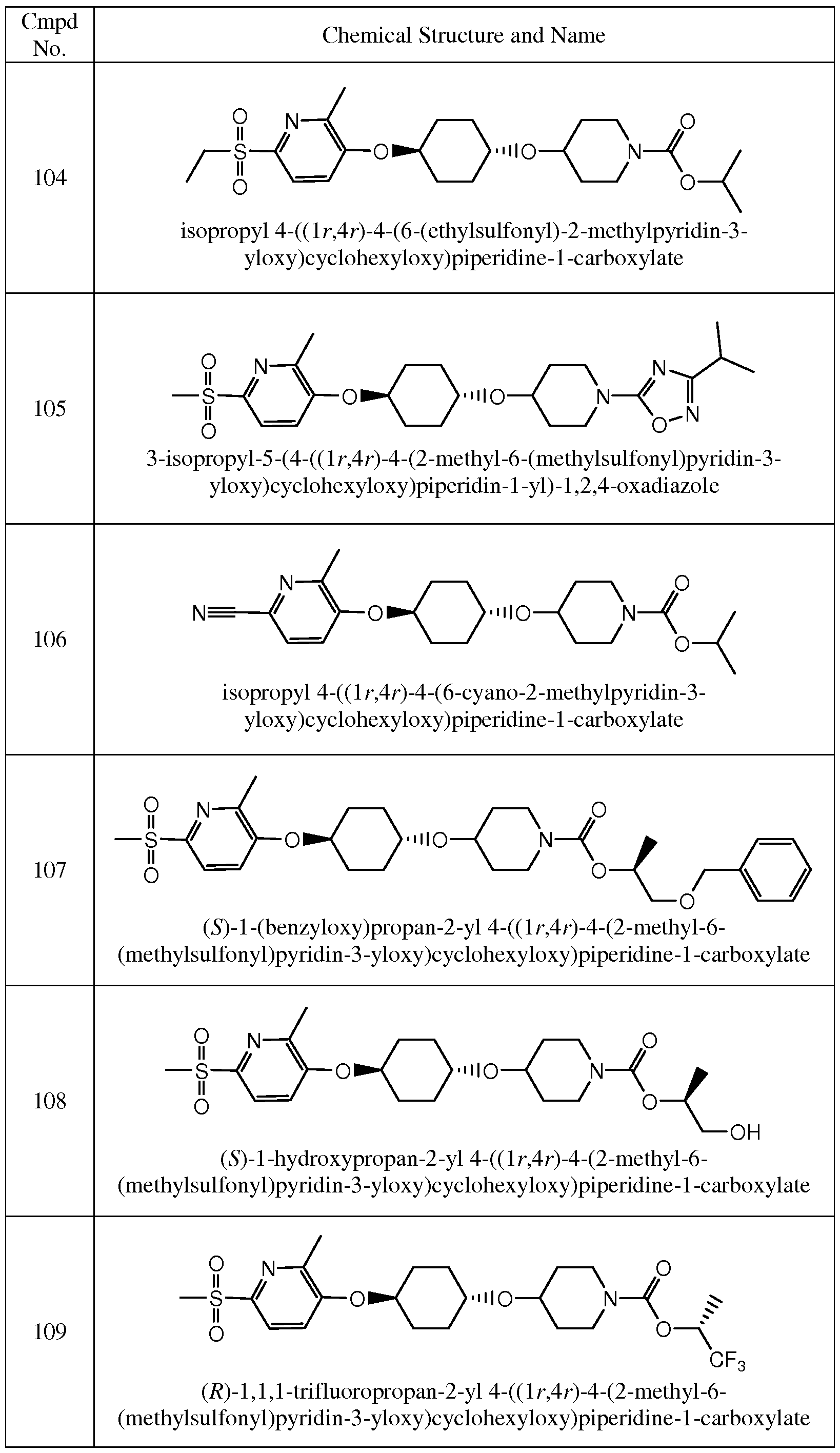
- Figure 21 shows a powder X-ray diffraction (PXRD) pattern for Compound 109.
- Figure 22 shows a powder X-ray diffraction (PXRD) pattern for Compound 122.
- FIG. 23 shows a thermogravimetric analysis (TGA) thermogram and a differential scanning calorimetry (DSC) thermogram for Compound 28.
- TGA thermogravimetric analysis
- DSC differential scanning calorimetry
- Figure 24 shows a TGA thermogram and a DSC thermogram for Compound 83.
- Figure 25 shows a TGA thermogram and a DSC thermogram for Compound 85.
- Figure 26 shows a TGA thermogram and a DSC thermogram for Compound 109.
- Figure 27 shows a TGA thermogram and a DSC thermogram for Compound 122.
- agonist refers to a moiety that interacts with and activates a G-protein-coupled receptor, for instance a GPR119-receptor, and can thereby initiate a physiological or pharmacological response characteristic of that receptor.
- a G-protein-coupled receptor for instance a GPR119-receptor
- an agonist may activate an intracellular response upon binding to a receptor, or enhance GTP binding to a membrane.
- antagonist refers to a moiety that competitively binds to the receptor at the same site as an agonist (for example, the endogenous ligand), but which does not activate the intracellular response initiated by the active form of the receptor and can thereby inhibit the intracellular responses by an agonist or partial agonist.
- An antagonist does not diminish the baseline intracellular response in the absence of an agonist or partial agonist.
- hydrate refers to a compound of the invention or a salt thereof, that further includes a stoichiometric or non-stoichiometric amount of water bound by non-covalent intermolecular forces.
- solvate refers to a compound of the invention or a salt, thereof, that further includes a stoichiometric or non-stoichiometric amount of a solvent bound by non-covalent intermolecular forces.
- Preferred solvents are volatile, non-toxic, and/or acceptable for administration to humans in trace amounts.
- the term "in need of treatment” and the term “in need thereof when referring to treatment are used interchangeably and refer to a judgment made by a caregiver (e.g. physician, nurse, nurse practitioner, etc. in the case of humans; veterinarian in the case of animals, including non-human mammals) that an individual or animal requires or will benefit from treatment. This judgment is made based on a variety of factors that are in the realm of a caregiver's expertise, but that includes the knowledge that the individual or animal is ill, or will become ill, as the result of a disease, condition or disorder that is treatable by the compounds of the invention. Accordingly, the compounds of the invention can be used in a protective or preventive manner; or compounds of the invention can be used to alleviate, inhibit or ameliorate the disease, condition or disorder.
- a caregiver e.g. physician, nurse, nurse practitioner, etc. in the case of humans; veterinarian in the case of animals, including non-human mammals
- mice refers to any animal, including mammals, preferably mice, rats, other rodents, rabbits, dogs, cats, swine, cattle, sheep, horses, or primates, and most preferably humans.
- inverse agonist refers to a moiety that binds to the endogenous form of the receptor or to the constitutively activated form of the receptor and which inhibits the baseline intracellular response initiated by the active form of the receptor below the normal base level of activity which is observed in the absence of an agonist or partial agonist, or decreases GTP binding to a membrane.
- the baseline intracellular response is inhibited in the presence of the inverse agonist by at least 30%, more preferably by at least 50% and most preferably by at least 75%, as compared with the baseline response in the absence of the inverse agonist.
- modulate or modulating refers to an increase or decrease in the amount, quality, response or effect of a particular activity, function or molecule.
- pharmaceutical composition refers to a composition comprising at least one active ingredient; including but not limited to, salts, solvates, and hydrates of compounds of the present invention, whereby the composition is amenable to investigation for a specified, efficacious outcome in a mammal (for example, without limitation, a human).
- a mammal for example, without limitation, a human.
- terapéuticaally effective amount refers to the amount of active compound or pharmaceutical agent that elicits the biological or medicinal response in a tissue, system, animal, individual or human that is being sought by a researcher, veterinarian, medical doctor or other clinician or caregiver or by an individual, which includes one or more of the following:
- Preventing the disease for example, preventing a disease, condition or disorder in an individual that may be predisposed to the disease, condition or disorder but does not yet experience or display the pathology or symptomatology of the disease;
- Inhibiting the disease for example, inhibiting a disease, condition or disorder in an individual that is experiencing or displaying the pathology or symptomatology of the disease, condition or disorder (i.e., arresting further development of the pathology and/or
- Ameliorating the disease for example, ameliorating a disease, condition or disorder in an individual that is experiencing or displaying the pathology or symptomatology of the disease, condition or disorder (i.e., reversing the pathology and/or symptomatology).
- amino refers to the group -NH 2 .
- C 1 -C6 alkoxy refers to a radical comprising a C 1 -C6 alkyl group attached directly to an oxygen atom, wherein C 1 -C6 alkyl has the same definition as found herein. Some embodiments contain 1 to 5 carbons. Some embodiments contain 1 to 4 carbons. Some embodiments contain 1 to 3 carbons. Some embodiments contain one or two carbons. Examples of an alkoxy group include, but are not limited to methoxy, ethoxy, «-propoxy, isopropoxy, n- butoxy, i-butoxy, isobutoxy, s-butoxy, and the like.
- C 1 -C6 alkyl refers to a straight or branched carbon radical containing 1 to 6 carbons. Some embodiments contain 1 to 5 carbons. Some embodiments contain 1 to 4 carbons. Some embodiments contain 1 to 3 carbons. Some embodiments contain one or two carbons. Examples of an alkyl group include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, n- butyl, s- butyl, isobutyl, f-butyl, pentyl, isopentyl, f-pentyl, neopentyl, 1-methylbutyl [i.e.
- Ci-C 6 alkylsulfonyl refers to a radical comprising a Ci-C 6 alkyl group attached to the sulfur of a sulfonyl group, wherein the Ci-C 6 alkyl radical has the same definition as described herein.
- Examples include, but are not limited to, methylsulfonyl, ethylsulfonyl, «-propylsulfonyl, isopropylsulfonyl, «-butylsulfonyl, s-butylsulfonyl, isobutylsulfonyl, i-butylsulfonyl, and the like.
- C 3 -C6 cycloalkyl refers to a saturated ring radical containing 3 to 6 carbons. Some embodiments contain 3 to 4 carbons. Some embodiments contain 3 to 5 carbons. Some embodiments contain 4 to 6 carbons. Some embodiments contain 5 to 6 carbons. Examples include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and the like.
- cyano refers to the group -CN.
- C 1 -C6 haloalkoxy refers to a radical comprising a C 1 -C6 haloalkyl group directly attached to an oxygen atom, wherein C 1 -C6 haloalkyl has the same definition as found herein.
- Examples include, but are not limited to, difluoromethoxy, trifluoromethoxy, 2,2,2- trifluoroethoxy, pentafluoroethoxy, 2-fluoropropan-2-yloxy, 1 , 1-difluoropropyloxy, 1 ,3- difluoropropan-2-yloxy, (5)-l -fluoropropan-2-yloxy, (R)-l-fluoropropan-2-yloxy, 1 ,1 , 1- trifluoropropan-2-yloxy, 1 , 1 ,1 , 3,3, 3-hexafluoropropan-2-yloxy, and the like.
- Ci-C 6 haloalkyl refers to a radical comprising a Ci-C 6 alkyl group substituted with one or more halogens, wherein Ci-C 6 alkyl has the same definition as found herein.
- the Ci-C 6 haloalkyl may be fully substituted in which case it can be represented by the formula C q L 2q+ i, wherein L is a halogen and "q" is 1 , 2, 3, 4, 5 or 6. When more than one halogen is present then they may be the same or different and selected from: fluorine, chlorine, bromine, and iodine. In some embodiments, haloalkyl contains 1 to 5 carbons.
- haloalkyl contains 1 to 4 carbons. In some embodiments, haloalkyl contains 1 to 3 carbons. In some embodiments, haloalkyl contains one or two carbons.
- haloalkyl groups include, but are not limited to, fluoromethyl, difluoromethyl, trifluoromethyl, 2,2,2- trifluoroethyl, pentafluoroethyl, 2-fluoropropan-2-yl, 1 , 1-difluoropropyl, l ,3-difluoropropan-2- yl, (5)-l-fluoropropan-2-yl, (R)-l -fluoropropan-2-yl, l ,l , l-trifluoropropan-2-yl, 1 , 1, 1 ,3,3,3- hexafluoropropan-2-yl, and the like.
- halogen refers to a fluoro, chloro, bromo or iodo group.
- heteroaryl refers to a ring system containing 5 to 10 ring atoms, that may contain a single ring or two fused rings, and wherein at least one ring is aromatic and at least one ring atom of the aromatic ring is a heteroatom selected from, for example: O, S and N, wherein N is optionally substituted with H, Q-C4 acyl, Q-C4 alkyl, or O (i.e., forming an N- oxide) and S is optionally substituted with one or two oxygens.
- the aromatic ring contains one heteroatom.
- the aromatic ring contains two heteroatoms.
- the aromatic ring contains three heteroatoms.
- Examples include furanyl, thienyl, pyrrolyl, imidazolyl, oxazolyl, thiazolyl, isoxazolyl, pyrazolyl, isothiazolyl, oxadiazolyl, triazolyl, tetrazolyl, thiadiazolyl, pyridinyl, pyrazinyl, pyrimidinyl, pyridazinyl, triazinyl, quinolinyl, isoquinolinyl, cinnolinyl, phthalazinyl, quinazolinyl, quinoxalinyl, indolyl, isoindolyl, indazolyl, indolizinyl, purinyl, naphthyridinyl, pteridinyl, carbazolyl, acridinyl.
- Some embodiments are directed to 5-membered heteroaryl rings.
- Examples of a 5-membered heteroaryl ring include furanyl, thienyl, pyrrolyl, imidazolyl, oxazolyl, thiazolyl, isoxazolyl, pyrazolyl, isothiazolyl, oxadiazolyl, triazolyl, tetrazolyl, thiadiazolyl, and the like.
- Some embodiments are directed to 6-membered heteroaryl rings. Examples of a 6-membered heteroaryl ring include pyridinyl, pyrazinyl, pyrimidinyl, pyridazinyl, triazinyl, and the like.
- Some embodiments are directed to 8 to 10-membered heteroaryl rings.
- Examples of a 8 to 10-membered heteroaryl ring include quinolinyl, isoquinolinyl, cinnolinyl, phthalazinyl, quinazolinyl, quinoxalinyl, indolyl, isoindolyl, indazolyl, indolizinyl, purinyl, naphthyridinyl, pteridinyl, carbazolyl, acridinyl. phenazinyl,
- heterocyclyl refers to a non-aromatic ring radical containing 3 to 7 ring atoms, wherein one, two or three ring atoms are heteroatoms is selected independently from, for example: O, S, and N, wherein N is optionally substituted with ⁇ , Ci-C 4 acyl or Ci-C 4 alkyl; and S is optionally substituted with one or two oxygens.
- heterocyclyl group examples include, but are not limited to, aziridinyl, azetidinyl, piperidinyl, morpholinyl, piperazinyl, pyrrolidinyl, [l,3]-dioxolanyl, thiomo holinyl, [l,4]oxazepanyl, l,l-dioxothiomo holinyl, azepanyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydrothiopyranyl, l-oxo-hexahydro- ⁇ 4 -thiopyranyl, l,l-dioxo-hexahydro- ⁇ 6 -thiopyranyl, and the like.
- hydroxyl refers to the group - ⁇ .
- phenyl refers to the group -CeH 5 .
- One aspect of the present invention provides, inter alia, compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- R 1 , R 2 , R 3 , Q, Z, X, and variables related thereto i.e., R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , and R 13 ), have the same definitions as described herein, supra and infra.
- One aspect of the present invention encompasses, inter alia, certain cyclohexane derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N or CR 4 ;
- Z is N or CR 5 ;
- X is N or CR 6 ;
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of heteroaryl and phenyl, each optionally substituted with one or more substituents selected independently from the group consisting of C 2 -C6 alkenyl, C 1 -C6 alkoxy, C 1 -C6 alkyl, halogen, C 1 -C6 haloalkoxy, and C 1 -C6 haloalkyl;
- R 2 is selected from the group consisting of H, C 1 -C6 alkyl, cyano, C3-C6 cycloalkyl, halogen, C C 6 haloalkyl, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ; wherein said C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-Ce alkylsulfonyl, cyano, and C(0)NR 12 R 13 ; said C 3 -C 6 cycloalkyl is optionally substituted with C(0)NR 12 R 13 ; said heteroaryl is optionally substituted with Ci-C 6 alkyl; and said heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-C 6 alkylsulfonyl, hydroxyl, and halogen;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, Q- C 6 alkyl, Ci-C 6 alkylsulfonyl, and halogen;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein said C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of heteroaryl, phenyl, and C(0)OR 9 ; wherein said heteroaryl is optionally substituted with C 1 -C6 alkyl;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C6 cycloalkyl, C 1 -C6 haloalkyl, heterocyclyl, and phenyl; wherein said C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, C 1 -C6 alkoxy, and R 10 , wherein said C 1 -C6 alkoxy is optionally substituted with phenyl; and said C 3 -C6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl and halogen; R 10 is heterocyclyl optionally substituted with Ci-C 6 alkyl;
- R 11 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, and heterocyclyl; wherein said Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and NR 12 R 13 ; and said heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, C 1 -C6 alkyl, and hydroxyl; and
- R 12 and R 13 are each independently selected from the group consisting of H, C 1 -C6 alkyl, and C3-C6 cycloalkyl; wherein said C 1 -C6 alkyl is optionally substituted with hydroxyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of cyano, halogen, hydroxyl, and C 1 -C6 alkoxy.
- One aspect of the present invention encompasses, inter alia, certain cyclohexane derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N or CR 4 ;
- Z is N or CR 5 ;
- X is N or CR 6 ;
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is heteroaryl or phenyl, each optionally substituted with one or more substituents selected independently from the group consisting of C 2 -C 6 alkenyl, Ci-C 6 alkoxy, Ci-C 6 alkyl, halogen, Ci-C 6 haloalkoxy, and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of H, Ci-C 6 alkyl, cyano, C 3 -C 6 cycloalkyl, halogen, Ci-C 6 haloalkyl, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ; wherein said C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-Cs alkylsulfonyl, cyano, and C(0)NR 12 R 13 ; said C 3 -C 6 cycloalkyl is optionally substituted with C(0)NR 12 R 13 ; said heteroaryl is optionally substituted with C 1 -C6 alkyl; and said heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, C 1 -C6 alkylsulfonyl, hydroxyl, and halogen;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, Ci_6 alkyl, Ci_6 alkylsulfonyl, and halogen;
- R 7 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, and C 1 -C6 haloalkyl; wherein said C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of heteroaryl, phenyl, and C(0)OR 9 ; wherein said heteroaryl is optionally substituted with Ci-C 6 alkyl;
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, and heterocyclyl; wherein said Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and R 10 ; and said C 3 - C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 10 is heterocyclyl optionally substituted with Ci-C 6 alkyl
- R 11 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, and heterocyclyl; wherein said C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and NR 12 R 13 , and said heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, C 1 -C6 alkyl, and hydroxyl; and
- R 12 and R 13 are each independently selected from the group consisting of H, C 1 -C6 alkyl, and C3-C6 cycloalkyl; wherein said C 1 -C6 alkyl is optionally substituted with hydroxyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of cyano, halogen, and hydroxyl.
- some embodiments of the present invention include every combination of one or more embodiments pertaining to the chemical groups represented by the variables and generic chemical formulae as described herein or every combination of one or more compounds of Formula (la) together/in combination with every combination of one or more pharmaceutical agents, such as an inhibitor of DPP-IV, a biguanide, an alpha-glucosidase inhibitor, and the like, either specifically disclosed herein or specifically disclosed in any reference recited herein just as if each and every combination was individually and explicitly recited.
- pharmaceutical agents such as an inhibitor of DPP-IV, a biguanide, an alpha-glucosidase inhibitor, and the like, either specifically disclosed herein or specifically disclosed in any reference recited herein just as if each and every combination was individually and explicitly recited.
- substituted indicates that at least one hydrogen atom of the chemical group is replaced by a non-hydrogen substituent or group, the non-hydrogen substituent or group can be monovalent or divalent. When the substituent or group is divalent, then it is understood that this group is further substituted with another substituent or group.
- a chemical group herein when a chemical group herein is "substituted" it may have up to the full valance of substitution; for example, a methyl group can be substituted by 1, 2, or 3 substituents, a methylene group can be substituted by one or two substituents, a phenyl group can be substituted by 1, 2, 3, 4, or 5 substituents, a naphthyl group can be substituted by 1, 2, 3, 4, 5, 6, or 7 substituents, and the like.
- substituted with one or more substituents refers to the substitution of a group with one substituent up to the total number of substituents physically allowed by the group.
- a group when a group is substituted with more than one group they can be identical or they can be different.
- Tautomeric forms can be in equilibrium or sterically locked into one form by appropriate substitution. It is understood that the various tautomeric forms are within the scope of the compounds of the present invention.
- meso isomers Such meso isomers may be referred to as cis and trans.
- the cis meso isomers of compounds of Formula (la) are named herein using the designation (Is, 4s) and the trans meso isomers of compounds of Formula (la) are named herein using the designation (lr,4r) as shown below:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Ic) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Ie) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N or CR 4 .
- Q is N.
- Q is CR 4 .
- Z is N or CR 5
- Z is N.
- Z is CR 5 .
- X is N, N(O), or CR 6 .
- X is N or CR 6 .
- X is N.
- X is N(O). It is understood that the group N(O) refers to an N- oxide group.
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (If) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- X is CR 6 .
- Q is N or CR 4 ; Z is N or CR 5 ; and X is N or CR 6 .
- Q is CR 4
- Z is CR 5
- X is CR 6 .
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Ig) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- each variable in Formula (Ig) has the same meaning as described herein, supra and infra.
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Ii) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
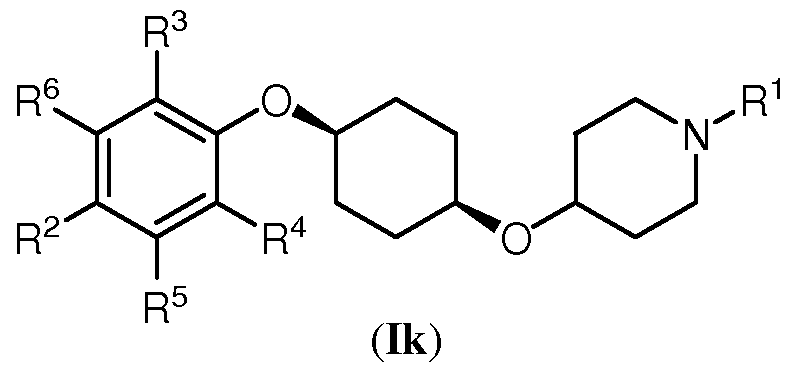
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Ik) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N
- Z is CR 5
- X is CR 6 .
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Im) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Io) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Iq) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N
- Z is CR 5
- X is N
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Is) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N
- Z is N
- X is CR 6 .
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Iu) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is CR 4
- Z is CR 5
- X is N.
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Iw) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Ix) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Iy) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N
- Z is CR 5
- X is N or CR 6 .
- Q is N or CR 4
- Z is CR 5
- X is N.
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (Ila) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (lie) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives selected from compounds of Formula (He) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexane derivatives wherein at least one Q, Z, and X is other than N.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of heteroaryl and phenyl, each optionally substituted with one or more substituents selected independently from the group consisting of C 2 -C6 alkenyl, C 1 -C6 alkoxy, C 1 -C6 alkyl, halogen, C 1 -C6 haloalkoxy, and C 1 -C6 haloalkyl;
- R 7 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, and C 1 -C6 haloalkyl; wherein the C3-C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 8 is selected from the group consisting of heteroaryl, phenyl, and C(0)OR 9 ; wherein the heteroaryl is optionally substituted with C 1 -C6 alkyl;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, heterocyclyl, and phenyl; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, Ci-C 6 alkoxy, and R 10 , wherein the Ci-C 6 alkoxy is optionally substituted with phenyl; and the C 3 -C 6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of Ci-C 6 alkyl and halogen; and
- R 10 is heterocyclyl optionally substituted with Ci-C 6 alkyl.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 ,
- R 1 is selected from the group consisting of 1,2,4- oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of prop-l-en-2-yl, ethoxy, methoxy, terf -butyl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2-fluoropropan-2-yl, and trifluoromethyl;
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl;
- R 8 is selected from the group consisting of 1 ,2,4-oxadiazolyl, cyclopropyl, 1,1,2,2- tetrafluoroethyl, cyclobutyl, trifluoromethyl, and C(0)OR 9 ; wherein said 1,2,4-oxadiazole, cyclopropyl, and cyclobutyl are each optionally substituted with one group selected from the group consisting of isopropyl and trifluoromethyl; and
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert-butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1-methylcyclopropyl, 2-methylcyclopropyl, 1,3- difluoropropan-2-yl, l-fluoropropan-2-yl, l,l,l,3,3,3-hexafluoropropan-2-yl, 1,1,1- trifluoropropan-2-yl, tetrahydrofuran-3-yl, 1 -hydro xypropan-2-yl, phenyl, 2,2,3,3- tetrafluorocyclobutyl, l-(benzyloxy)propan-2-yl, l,l,l-trifluoro-2-methylpropan-2-yl, and cyclopropyl.
- R 1 is selected from the group consisting of H, S, S
- R 1 is selected from the group consisting of heteroaryl and phenyl, each optionally substituted with one or more substituents selected independently from the group consisting of C 2 -C 6 alkenyl, Ci-C 6 alkoxy, Ci-C 6 alkyl, halogen, Ci-C 6 haloalkoxy, and Ci-C 6 haloalkyl;
- R 7 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, and C 1 -C6 haloalkyl; wherein the C3-C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 8 is selected from the group consisting of heteroaryl, phenyl, and C(0)OR 9 ; wherein the heteroaryl is optionally substituted with C 1 -C6 alkyl;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, and heterocyclyl; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and R 10 ; and the C 3 - (, cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent; and
- R 10 is heterocyclyl optionally substituted with Ci-C 6 alkyl.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of a five-membered heteroaryl, phenyl, and a six-membered heteroaryl, each optionally substituted with one or two substituents selected independently from the group consisting of C 2 -C 6 alkenyl, Q-C4 alkoxy, Ci-C 6 alkyl, halogen, Q-C4 haloalkoxy, and Ci-C 6 haloalkyl;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, heterocyclyl, and phenyl; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, C 1 -C6 alkoxy, and R 10 , wherein the C 1 -C6 alkoxy is optionally substituted with phenyl; and the C3-C6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl and halogen; and
- R 10 is heterocyclyl optionally substituted with one C 1 -C6 alkyl substituent.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of a five-membered heteroaryl, phenyl, and a six-membered heteroaryl, each optionally substituted with one or two substituents selected independently from the group consisting of C 2 -C 6 alkenyl, Q-C4 alkoxy, Ci-C 6 alkyl, halogen, Q-C4 haloalkoxy, and Ci-C 6 haloalkyl; R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, and heterocyclyl; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and R 10 ; and the C 3 - (, cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent; and
- R 10 is heterocyclyl optionally substituted with one C 1 -C6 alkyl substituent.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of C 2 -C6 alkenyl, C 1 -C4 alkoxy, Ci-C 6 alkyl, halogen, Q-C4 haloalkoxy, and Ci-C 6 haloalkyl;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, heterocyclyl, and phenyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, Ci-C 6 alkoxy, and R 10 , wherein the C 1 -C6 alkoxy is optionally substituted with phenyl; and the C 3 -C6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl and halogen; and
- R 10 is heterocyclyl optionally substituted with one C 1 -C6 alkyl substituent.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of C 2 -C6 alkenyl, C 1 -C 4 alkoxy, C 1 -C6 alkyl, halogen, C 1 -C 4 haloalkoxy, and C 1 -C6 haloalkyl;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, and heterocyclyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and R 10 ; and the C 3 - C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent; and
- R 10 is heterocyclyl optionally substituted with one Ci-C 6 alkyl substituent.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of prop-l-en-2-yl, ethoxy, methoxy, tert-butyl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2-fluoropropan- 2-yl, and trifluoromethyl;
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl;
- R 8 is selected from the group consisting of 1 ,2,4-oxadiazolyl and C(0)OR 9 ; wherein the 1 ,2,4-oxadiazole is optionally substituted with one isopropyl group; and
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert-butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1-methylcyclopropyl, 2-methylcyclopropyl, 1,3- difluoropropan-2-yl, l-fluoropropan-2-yl, l,l,l,3,3,3-hexafluoropropan-2-yl, 1,1,1- trifluoropropan-2-yl, tetrahydrofuran-3-yl, 1 -hydro xypropan-2-yl, phenyl, 2,2,3,3- tetrafluorocyclobutyl, and l-(benzyloxy)propan-2-yl.
- R 1 is selected from the group consisting of H, S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of prop-l-en-2-yl, ethoxy, methoxy, tert-butyl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2-fluoropropan- 2-yl, and trifluoromethyl;
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl;
- R 8 is selected from the group consisting of 1 ,2,4-oxadiazolyl and C(0)OR 9 ; wherein the 1 ,2,4-oxadiazole is optionally substituted with one isopropyl group; and
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert-butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1-methylcyclopropyl, 2-methylcyclopropyl, 1,3- difluoropropan-2-yl, l-fluoropropan-2-yl, l,l,l,3,3,3-hexafluoropropan-2-yl, 1,1,1- trifluoropropan-2-yl, tetrahydrofuran-3-yl, and l-hydroxypropan-2-yl.
- R 1 is selected from the group consisting of H
- cyclopropylsulfonyl isopropylsulfonyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2- methylcyclopropanecarbonyl, 2,2-difluorobutanoyl, (3-isopropyl-l ,2,4-oxadiazol-5-yl)methyl, 2- teri-butoxy-2-oxoethyl, teri-butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl, cyclopentyloxycarbonyl, (1,1,1 ,3,3,3-hexalluoropropan-2-yloxy)carbonyl, ((3-methyloxetan-3- yl)methoxy)carbonyl, (l-methylcyclopropoxy)carbonyl, sec-butoxycarbonyl, (tetrahydrofuran-
- R 1 is selected from the group consisting of cyclopropylsulfonyl, isopropylsulfonyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2-methylcyclopropanecarbonyl, 2,2- difluorobutanoyl, (3-isopropyl-l,2,4-oxadiazol-5-yl)methyl, 2-teri-butoxy-2-oxoethyl, tert- butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl, cyclopentyloxycarbonyl, (1,1,1,3,3,3- hexafluoropropan-2-yloxy)carbonyl, ((3-methyloxetan-3-yl)methoxy)carbonyl, (1- methylcyclopropoxy)carbonyl, iec-butoxycarbonyl, (tetrahydrofuran-3-yloxy)carbonyl, (1,1,1
- R 1 is selected from the group consisting of H,
- cyclopropylsulfonyl isopropylsulfonyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2- methylcyclopropanecarbonyl, 2,2-difluorobutanoyl, (3-isopropyl-l ,2,4-oxadiazol-5-yl)methyl,
- R 1 is selected from the group consisting of H
- cyclopropylsulfonyl isopropylsulfonyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2- methylcyclopropanecarbonyl, 2,2-difluorobutanoyl, (3-isopropyl-l ,2,4-oxadiazol-5-yl)methyl, 2-teri-butoxy-2-oxoethyl, teri-butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl, cyclopentyloxycarbonyl, (1,1,1 ,3,3,3-hexafluoropropan-2-yloxy)carbonyl, ((3-methyloxetan-3- yl)methoxy)carbonyl, (l-methylcyclopropoxy)carbonyl, sec-butoxycarbonyl, (tetrahydrofuran- 3-yloxy)carbonyl, (1,1,1 -trifluoropropan-2-yloxy
- R 1 is selected from the group consisting of S(0) 2 R 7 and C(0)R 7 ;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent.
- R 1 is selected from the group consisting of S(0) 2 R 7 and C(0)R 7 ; and
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl.
- R 1 is selected from the group consisting of cyclopropylsulfonyl, isopropylsulionyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2-methylcyclopropanecarbonyl, and 2,2- difluorobutanoyl.
- R 1 is CH 2 R 8 ;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one C 1 -C6 alkyl substituent; and
- R 9 is Ci-C 6 alkyl.
- R 1 is CH 2 R 8 ;
- R 8 is selected from the group consisting of 1 ,2,4-oxadiazolyl and C(0)OR 9 ; wherein the 1 ,2,4-oxadiazole is optionally substituted with one isopropyl group; and
- R 9 is tert-butyl
- R 1 is selected from the group consisting of (3-isopropyl- 1,2,4- oxadiazol-5-yl)methyl and 2-teri-butoxy-2-oxoethyl.
- R 1 is C(0)OR 9 ;
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, heterocyclyl, and phenyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, Ci-C 6 alkoxy, and R 10 , wherein Ci-C 6 alkoxy is optionally substituted with phenyl; and the C 3 -C 6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl and halogen; and
- R 10 is heterocyclyl optionally substituted with one C 1 -C6 alkyl substituent.
- R 1 is C(0)OR 9 ;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, and heterocyclyl; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and R 10 ; and the C 3 - (, cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent; and
- R 10 is heterocyclyl optionally substituted with one C 1 -C6 alkyl substituent.
- R 1 is C(0)OR 9 ;
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert-butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1-methylcyclopropyl, l,3-difluoropropan-2-yl, 1- fluoropropan-2-yl, 1,1,1 ,3,3,3-hexafluoropropan-2-yl, 1,1,1 -trifluoropropan-2-yl, tetrahydrofuran-3-yl, l-hydroxypropan-2-yl, phenyl, 2,2,3,3-tetrafluorocyclobutyl, and 1-
- R 1 is C(0)OR 9 ;
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert-butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1-methylcyclopropyl, l,3-difluoropropan-2-yl, 1- fluoropropan-2-yl, 1,1,1 ,3,3,3-hexafluoropropan-2-yl, 1,1,1 -trifluoropropan-2-yl,
- R 1 is selected from the group consisting of teri-butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl, cyclopentyloxycarbonyl, (1,1,1,3,3,3- hexafluoropropan-2-yloxy)carbonyl, ((3-methyloxetan-3-yl)methoxy)carbonyl, (1- methylcyclopropoxy)carbonyl, sec-butoxycarbonyl, (tetrahydrofuran-3-yloxy)carbonyl, (1,1,1- trifluoropropan-2-yloxy)carbonyl, (l,3-difluoropropan-2-yloxy)carbonyl, (l-fluoropropan-2- yloxy)carbonyl, (l-hydroxypropan-2-yloxy)carbonyl, phenoxycarbonyl, (2,2,3,3- tetrafluorocyclobutoxy)carbonyl, and ( 1 -(benzyl,
- R 1 is selected from the group consisting of teri-butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl, cyclopentyloxycarbonyl, (1,1,1,3,3,3- hexafluoropropan-2-yloxy)carbonyl, ((3-methyloxetan-3-yl)methoxy)carbonyl, (1- methylcyclopropoxy)carbonyl, sec-butoxycarbonyl, (tetrahydrofuran-3-yloxy)carbonyl, (1,1,1- trifluoropropan-2-yloxy)carbonyl, (l,3-difluoropropan-2-yloxy)carbonyl, (l-fluoropropan-2- yloxy)carbonyl, and (l-hydroxypropan-2-yloxy)carbonyl.
- R 1 is selected from the group consiting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of C 2 -C 6 alkenyl, C 1 -C4 alkoxy, C 1 -C6 alkyl, halogen, Ci-C 4 haloalkoxy, and C 1 -C6 haloalkyl.
- R 1 is selected from the group consiting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of prop-l-en-2-yl, ethoxy, methoxy, tert-bv yl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2-fluoropropan-
- R 1 is selected from the group consisting of 3-isopropyl- 1,2,4- oxadiazol-5-yl, 5-isopropyl-l,2,4-oxadiazol-3-yl, 3-(2-fluoropropan-2-yl)-l,2,4-oxadiazol-5-yl,
- R 1 is selected from the group consisting of 3-isopropyl- 1,2,4- oxadiazol-5-yl, 5-isopropyl-l,2,4-oxadiazol-3-yl, 3-(2-fluoropropan-2-yl)-l,2,4-oxadiazol-5-yl, 3-teri-butyl-l,2,4-oxadiazol-5-yl, 3-(prop-l-en-2-yl)-l,2,4-oxadiazol-5-yl, p-tolyl, 4- (trifluoromethyl)phenyl, 4-(trifluoromethoxy)phenyl, 4-methoxyphenyl, 3- (trifluoromethyl)phenyl, 4-fluorophenyl, 4-chloro-2-fluorophenyl, 5-ethyl-pyrimidin-2-yl, 5- chloro-pyrimidin-2-yl, 5-methyl-pyrimidin-2-yl, 5-(trifluoromethyl)pyridin
- R 1 is selected from the group consisting of 3-isopropyl- 1,2,4- oxadiazol-5-yl, 5-isopropyl-l,2,4-oxadiazol-3-yl, 3-(2-fluoropropan-2-yl)-l,2,4-oxadiazol-5-yl, 3-teri-butyl-l,2,4-oxadiazol-5-yl, 3-(prop-l-en-2-yl)-l,2,4-oxadiazol-5-yl, and 5-(2- fluoropropan-2-yl)-l,2,4-oxadiazol-3-yl.
- R 1 is selected from the group consisting of 3-isopropyl- 1,2,4- oxadiazol-5-yl, 5-isopropyl-l,2,4-oxadiazol-3-yl, 3-(2-fluoropropan-2-yl)-l,2,4-oxadiazol-5-yl, 3-teri-butyl-l,2,4-oxadiazol-5-yl, and 3-(prop-l-en-2-yl)-l,2,4-oxadiazol-5-yl.
- R 1 is selected from the group consisting of p-tolyl, 4- (trifluoromethyl)phenyl, 4-(trifluoromethoxy)phenyl, 4-methoxyphenyl, 3- (trifluoromethyl)phenyl, 4-fluorophenyl, and 4-chloro-2-fluorophenyl.
- R 1 is selected from the group consisting of 5-ethyl-pyrimidin-2- yl, 5-chloro-pyrimidin-2-yl, 5-methyl-pyrimidin-2-yl, 5-(trifluoromethyl)pyridin-2-yl, 3-methyl- pyridazin-6-yl, 2-methyl-pyrazin-5-yl, 5-chloro-pyridin-2-yl, 3-ethoxy-pyridazin-6-yl, 5-fluoro- pyridin-2-yl, and 5-methoxy-pyrimidin-2-yl.
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- R 1 is In some embodiments, R s ((3-methyloxetan-3-yl)methoxy)carbonyl.
- R s ( 1 -methylcyclopropoxy)carbonyl.
- R sec-butoxycarbonyl
- R s tetrahydrofuran-3-yloxycarbonyl
- R s (1,1,1 -triiluoropropan-2-yloxy)carbonyl.
- R s (l,3-difluoropropan-2-yloxy)carbonyl.
- R s ( 1 -fluoropropan-2-yloxy)carbonyl.
- R 3-isopropyl-l,2,4-oxadiazol-5-yl.
- R s 5-isopropyl-l,2,4-oxadiazol-3-yl.
- R 3-(2-fluoropropan-2-yl)-l ,2,4-oxadiazol-5-yl.
- R s 3-teri-butyl-l,2,4-oxadiazol-5-yl.
- R 3-(prop-l-en-2-yl)-l,2,4-oxadiazol-5-yl.
- R s p-tolyl In some embodiments, R s p-tolyl.
- R 4-(trifluoromethyl)phenyl.
- R 4-(trifluoromethoxy)phenyl.
- R 4-methoxyphenyl
- R s 3-(trifluoromethyl)phenyl.
- R 4-fluorophenyl
- R 4-chloro-2 -fluorophenyl.
- R s 5-ethyl-pyrimidin-2-yl.
- R s 5-chloro-pyrimidin-2-yl.
- R s 5-methyl-pyrimidin-2-yl.
- R s 5-(trifluoromethyl)pyridin-2-yl.
- R s 3-methyl-pyridazin-6-yl.
- R s 2-methyl-pyrazin-5-yl.
- R s 5-chloro-pyridin-2-yl.
- R s 3-ethoxy-pyridazin-6-yl.
- R s 5-iluoro-pyridin-2-yl.
- R s 5-methoxy-pyrimidin-2-yl.
- R s ( 1 -hydroxypropan-2-yloxy)carbonyl.
- R s phenoxycarbonyl
- R s 5-(2-fluoropropan-2-yl)-l ,2,4-oxadiazol-3-yl.
- R s (2,2,3, 3-tetrafluorocyclobutoxy)carbonyl.
- R s ( 1 -(benzyloxy)propan-2-yloxy)carbonyl.
- R s isopropylthiocarbonyl.
- R s 5-methylpyridin-2-yl.
- R s 5-ethylpyridin-2-yl.
- R 1 is (l,l,l-trifluoro-2-methylpropan-2-yloxy)carbonyl.
- R 1 is cyclopropylthiocarbonyl.
- R 1 is (l-(trifluoromethyl)cyclopropyl)methyl.
- R 1 is 2,2,3, 3-tetrafluoropropyl.
- R 1 is (l-(trifluoromethyl)cyclobutyl)methyl.
- R 1 is 2,2,2-trifluoroethyl.
- R 2 is selected from the group consisting of H, C 1 -C6 alkyl, cyano, C3-C6 cycloalkyl, halogen, C 1 -C6 haloalkyl, heteroaryl, heterocyclyl, S(0) 2 R n , and
- C(0)NR 12 R 13 wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, C 1 -C6 alkylsulfonyl, cyano, and C(0)NR 12 R 13 ; the C 3 -C 6 cycloalkyl is optionally substituted with C(0)NR 12 R 13 ; the heteroaryl is optionally substituted with C 1 -C6 alkyl; and the heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-C 6 alkylsulfonyl, hydroxyl, and halogen;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, Q- C 6 alkyl, Ci-C 6 alkylsulfonyl, and halogen;
- R 11 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, and heterocyclyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and NR 12 R 13 ; and the heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-C 6 alkyl, and hydroxyl; and
- R 12 and R 13 are each independently selected from the group consisting of H, C 1 -C6 alkyl, and C3-C6 cycloalkyl; wherein the C 1 -C6 alkyl is optionally substituted with hydroxyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of cyano, halogen, hydroxyl, and C 1 -C6 alkoxy.
- R 2 is selected from the group consisting of H, C 1 -C6 alkyl, cyano, C3-C6 cycloalkyl, halogen, C 1 -C6 haloalkyl, heteroaryl, heterocyclyl, S(0) 2 R n , and
- C(0)NR 12 R 13 wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, C 1 -C6 alkylsulfonyl, cyano, and C(0)NR 12 R 13 ; the C 3 -C 6 cycloalkyl is optionally substituted with C(0)NR 12 R 13 ; the heteroaryl is optionally substituted with Ci-C 6 alkyl; and the heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, Ci-C 6 alkylsulfonyl, hydroxyl, and halogen; R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, Q-
- R 11 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, and heterocyclyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and NR 12 R 13 ; and the heterocyclyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, C 1 -C6 alkyl, and hydroxyl; and
- R 12 and R 13 are each independently selected from the group consisting of H, C 1 -C6 alkyl, and C3-C6 cycloalkyl; wherein the C 1 -C6 alkyl is optionally substituted with hydroxyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of cyano, halogen, and hydroxyl.
- R 2 is selected from the group consisting of H, C 1 -C6 alkyl, cyano, halogen, heteroaryl, heterocyclyl, S(0) n R n , and C(0)NR 12 R 13 ; wherein said C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, cyano, and C(0)NR 12 R 13 ;
- R 11 is selected from the group consiting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and heterocyclyl; wherein said heterocyclyl is optionally substituted with one or two halogens; and
- R 12 and R 1 1 3 J are each independently 12 13
- Ci-C 6 alkyl or R and R together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of halogen and Ci-C 6 alkoxy.
- R 2 is selected from the group consisting of H, Ci-C 6 alkyl, cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, cyano, and C(0)NR 12 R 13 ;
- R 11 is selected from the group consiting of C 1 -C6 alkyl and C 3 -C6 cycloalkyl;
- R 12 and R 1 1 3 J are each independently 12 13
- R 2 is selected from the group consisting of H, C 1 -C6 alkyl, cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ; wherein the Ci-C 6 alkyl is optionally substituted with cyano;
- R 11 is Ci-Ce alkyl
- R 12 and R 13 are each independently Ci-C 6 alkyl.
- R 2 is selected from the group consisting of H, methyl, isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, fluoro, pyridazin-4-yl, lH-l ,2,4-triazol-l-yl, l ,l -dioxo-thiomorpholin-4-yl, morpholin-4-yl, 2-cyanoethyl, cyclopropylsulfonyl, 2-amino-3-(3,3-difluoroazetidin-l-yl)-3-oxopropyl, ethylsulfonyl, pyrimidin-5-yl, 3-methoxyazetidine-l -carbonyl, and 3,3-difluoroazetidin-l -ylsulfon
- R 2 is selected from the group consisting of ⁇ , methyl, isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, fluoro, pyridazin-4-yl, lH-l ,2,4-triazol-l-yl, l ,l -dioxo-thiomorpholin-4-yl, morpholin-4-yl, 2-cyanoethyl, cyclopropylsulfonyl, 2-amino-3-(3,3-difluoroazetidin-l-yl)-3-oxopropyl, ethylsulfonyl, pyrimidin-5-yl, and 3-methoxyazetidine-l -carbonyl.
- R 2 is selected from the group consisting of ⁇ , methyl, isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, fluoro, pyridazin-4-yl, lH-l ,2,4-triazol-l-yl, l ,l -dioxo-thiomorpholin-4-yl, morpholin-4-yl, and 2-cyanoethyl.
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of ⁇ , methyl, methylsulfonyl, and fluoro.
- R 2 is selected from the group consisting of ⁇ , Ci-C 6 alkyl, cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ; wherein the C C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, cyano, and C(0)NR 12 R 13 ;
- R 3 is selected from the group consisting of ⁇ and C 1 -C4 alkylsulfonyl
- R 4 is selected from the group consisting of ⁇ and halogen
- R 5 is selected from the group consisting of ⁇ , halogen and Ci-C 6 alkyl
- R 6 is selected from the group consisting of ⁇ and halogen
- R 11 is selected from the group consisting of C 1 -C6 alkyl and C3-C6 cycloalkyl;
- R and R 1J are each independently C 1 -C6 alkyl; or R and R together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of halogen and C 1 -C6 alkoxy.
- R 2 is selected from the group consisting of ⁇ , C 1 -C6 alkyl, cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ; wherein the C C 6 alkyl is optionally substituted with cyano;
- R 3 is selected from the group consisting of ⁇ and C 1 -C4 alkylsulfonyl
- R 4 is selected from the group consisting of ⁇ and halogen
- R 5 is selected from the group consisting of ⁇ , halogen, and Ci-C 6 alkyl
- R 6 is selected from the group consisting of ⁇ and halogen;
- R 11 is Ci-Ce alkyl;
- R 12 and R 13 are each independently Ci-C 6 alkyl.
- R 2 is selected from the group consisting of H, methyl, isopropylsulfonyl,
- R 3 is selected from the group consisting of ⁇ and methylsulionyl
- R 4 is selected from the group consisting of ⁇ and fluoro
- R 5 is selected from the group consisting of ⁇ , fluoro, and methyl
- R 6 is selected from the group consisting of ⁇ and fluoro.
- R 2 is selected from the group consisting of ⁇ , methyl, isopropylsulfonyl,
- R 3 is selected from the group consisting of ⁇ and methylsulionyl
- R 4 is selected from the group consisting of ⁇ and fluoro
- R 5 is selected from the group consisting of ⁇ , fluoro, and methyl
- R 6 is selected from the group consisting of ⁇ and fluoro.
- R 2 is ⁇ .
- R 2 is C 1 -C4 alkylsulfonyl.
- R 2 is cyano
- R 2 is C 2 -C dialkylcarboxamide.
- R 2 is halogen
- R 2 is heteroaryl
- R 2 is a five-membered heteroaryl.
- R 2 is a six-membered heteroaryl.
- R 2 is a heterocyclyl
- R 2 is C 1 -C6 alkyl optionally substituted with one cyano group.
- R 2 is isopropylsulfonyl.
- R 2 is methylsulionyl.
- R 2 is cyano
- R 2 is dimethylcarbamoyl.
- R 2 is bromo
- R 2 is 2-cyanoethyl
- R 2 is lH-l ,2,4-triazol-l -yl. In some embodiments, R 2 is pyridazin-4-yl.
- R 2 is l,l-dioxo-thiomo holin-4-yl.
- R 2 is morpholin-4-yl.
- R 2 is cyclopropylsulfonyl.
- R 2 is 2-amino-3-(3,3-difluoroazetidin-l
- R 2 is ethylsulfonyl, pyrimidin-5-yl.
- R 2 is 3-methoxyazetidine-l-carbonyl.
- R 2 is 3,3-difluoroazetidin-l -ylsulfonyl.
- R 3 is H.
- R 3 is C 1 -C4 alkylsulfonyl.
- R 3 is methylsulfonyl
- R 4 is H.
- R 4 is halogen
- R 4 is fluoro
- R 5 is H.
- R 5 is halogen
- R 5 is Ci-Ce alkyl.
- R 5 is fluoro
- R 5 is methyl
- R 6 is H.
- R 6 is halogen
- R 6 is fluoro
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C6 cycloalkyl, C 1 -C6 haloalkyl, heterocyclyl, and phenyl; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, C 1 -C6 alkoxy, and R 10 , wherein the C 1 -C6 alkoxy is optionally substituted with phenyl; and the C 3 -C6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl and halogen; and
- R 12 and R 13 are each independently selected from the group consisting of H, C 1 -C6 alkyl, and C 3 -C 6 cycloalkyl; wherein the Ci-C 6 alkyl is optionally substituted with hydroxyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of cyano, halogen, hydroxyl, and Ci-C 6 alkoxy.
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, and heterocyclyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and R 10 ; and the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent; and R 12 and R 13 are each independently selected from the group consisting of H, Ci-C 6 alkyl, and C3-C6 cycloalkyl; wherein the C 1 -C6 alkyl is optionally substituted with hydroxyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or more substituents selected independently from the group consisting of cyano, halogen, and hydroxyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ig) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of Q-C 4 alkoxy, Ci-C 6 alkyl, halogen, Q-C 4 haloalkoxy, and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of H, Ci-C 6 alkyl, cyano, heteroaryl, and S(0) 2 R n ; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, cyano, and C(0)NR 12 R 13 ;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, C 1 -C6 alkylsulfonyl, and halogen;
- R 7 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C6 cycloalkyl, and C 1 -C6 haloalkyl; wherein the C 3 -C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one Q- C 6 alkyl substituent;
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and
- Ci-C 6 haloalkyl wherein the Ci-C 6 alkyl is optionally substituted with one R 10 substituent; and the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 10 is heterocyclyl optionally substituted with one Ci-C 6 alkyl substituent
- R 11 is Ci-Cs alkyl
- R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or two halogens.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ig) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of C 1 -C4 alkoxy,
- R 2 is selected from the group consisting of H, Ci-C 6 alkyl, cyano, heteroaryl, and S(0) 2 R n ; wherein the Ci-C 6 alkyl is optionally substituted with cyano;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, Ci-C 6 alkylsulfonyl, and halogen;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, or C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one d- (, alkyl substituent;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C6 cycloalkyl, and C 1 -C6 haloalkyl; wherein the C 1 -C6 alkyl is optionally substituted with one R 10 substituent; and the C 3 -C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 10 is heterocyclyl optionally substituted with one C 1 -C6 alkyl substituent; and R 11 is Ci-C 6 alkyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ig) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of ethoxy, methoxy, tert-b tyl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2- fluoropropan-2-yl, and trifluoromethyl;
- R 2 is selected from the group consisting of H, methylsulfonyl, cyano, 1H-1,2,4- triazol-l-yl, 2-cyanoethyl, and 2-amino-3-(3,3-difluoroazetidin-l-yl)-3-oxopropyl;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of ⁇ , methylsulfonyl, and fluoro;
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl;
- R 8 is selected from the group consisting of 1 ,2,4-oxadiazolyl and C(0)OR 9 ; wherein the 1 ,2,4-oxadiazole is optionally substituted with one isopropyl group; and
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert- butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1 -methylcyclopropyl, 1,1,1,3,3,3- hexafluoropropan-2-yl, and l,l,l-trifluoropropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ig) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of ethoxy, methoxy, tert-butyl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2- fluoropropan-2-yl, and trifluoromethyl;
- R 2 is selected from the group consisting of H, methylsulfonyl, cyano, 1H-1,2,4- triazol-l-yl, and 2-cyanoethyl;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of ⁇ , methylsulfonyl, and fluoro;
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl;
- R 8 is selected from the group consisting of 1,2,4-oxadiazolyl and C(0)OR 9 ; wherein the 1 ,2,4-oxadiazole is optionally substituted with one isopropyl group; and
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert- butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1 -methylcyclopropyl, 1,1,1,3,3,3- hexafluoropropan-2-yl, and l,l,l-trifluoropropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ig) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of cyclopropylsulfonyl, isopropylsulfonyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2-methylcyclopropanecarbonyl,
- 2,2-difluorobutanoyl (3-isopropyl-l,2,4-oxadiazol-5-yl)methyl, 2-tert-b toxy-2- oxoethyl, teri-butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl,
- R 2 is selected from the group consisting of H, methylsulfonyl, cyano, 1H-1,2,4- triazol-l-yl, 2-cyanoethyl, and 2-amino-3-(3,3-difluoroazetidin-l-yl)-3-oxopropyl;
- R 3 is selected from the group consisting of ⁇ and methylsulfonyl
- R 4 is selected from the group consisting of ⁇ and fluoro
- R 5 is selected from the group consisting of ⁇ , and fluoro
- R 6 is selected from the group consisting of ⁇ and fluoro.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ig) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of cyclopropylsulfonyl, isopropylsulfonyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2-methylcyclopropanecarbonyl, 2,2-difluorobutanoyl, (3-isopropyl-l,2,4-oxadiazol-5-yl)methyl, 2-tert-b toxy-2- oxoethyl, teri-butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl,
- R 2 is selected from the group consisting of H, methylsulfonyl, cyano, 1H-1 ,2,4- triazol-l-yl, and 2-cyanoethyl;
- R 3 is selected from the group consisting of ⁇ and methylsulfonyl
- R 4 is selected from the group consisting of ⁇ and fluoro
- R 5 is selected from the group consisting of ⁇ , and fluoro; and R 6 is selected from the group consisting of ⁇ and fluoro.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ii) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of C 1 -C4 alkoxy, Ci-C 6 alkyl, halogen, Ci-C haloalkoxy, and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of H, Ci-C 6 alkyl, cyano, heteroaryl, and S(0) 2 R n ; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of amino, cyano, and C(0)NR 12 R 13 ;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, C 1 -C6 alkylsulfonyl, and halogen;
- R 7 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, and C 1 -C6 haloalkyl; wherein the C3-C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one d- C 6 alkyl substituent;
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the Ci-C 6 alkyl is optionally substituted with one R 10 substituent; and the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 10 is heterocyclyl optionally substituted with one Ci-C 6 alkyl substituent;
- R 11 is Ci-C 6 alkyl;
- R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with one or two halogens.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ii) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and
- R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of Ci-C 4 alkoxy, C 1 -C6 alkyl, halogen, Ci-C 4 haloalkoxy, and C 1 -C6 haloalkyl;
- R 2 is selected from the group consisting of H, Ci-C 6 alkyl, cyano, heteroaryl, and S(0) 2 R n ; wherein the Ci-C 6 alkyl is optionally substituted with cyano;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H, Ci-C 6 alkylsulfonyl, and halogen;
- R 7 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, and Ci-C 6 haloalkyl; wherein the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent;
- R 8 is selected from the group consisting of a five-membered heteroaryl and C(0)OR 9 ; wherein the five-membered heteroaryl is optionally substituted with one Q- (, alkyl substituent;
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, and
- C 1 -C6 haloalkyl wherein the C 1 -C6 alkyl is optionally substituted with one R 10 substituent; and the C 3 -C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 10 is heterocyclyl optionally substituted with one C 1 -C6 alkyl substituent; and R 11 is Ci-C 6 alkyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ii) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of ethoxy, methoxy, tert-b tyl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2- fluoropropan-2-yl, and trifluoromethyl;
- R 2 is selected from the group consisting of H, methylsulfonyl, cyano, 1H-1,2,4- triazol-l-yl, 2-cyanoethyl, and 2-amino-3-(3,3-difluoroazetidin-l-yl)-3-oxopropyl;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl;
- R 8 is selected from the group consisting of 1 ,2,4-oxadiazolyl and C(0)OR 9 ; wherein the 1 ,2,4-oxadiazole is optionally substituted with one isopropyl group; and
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert- butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1-methylcyclopropyl, 1,1,1,3,3,3- hexafluoropropan-2-yl, and l,l,l-trifluoropropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ii) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of S(0) 2 R 7 , C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1,2,4-oxadiazolyl, phenyl, pyrimidinyl, pyridinyl, pyridazinyl, and pyrazinyl, each optionally substituted with one or two substituents selected independently from the group consisting of ethoxy, methoxy, tert-butyl, ethyl, isopropyl, methyl, chloro, fluoro, trifluoromethoxy, 2- fluoropropan-2-yl, and trifluoromethyl;
- R 2 is selected from the group consisting of H, methylsulfonyl, cyano, 1H-1,2,4- triazol-l-yl, and 2-cyanoethyl;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of
- R 7 is selected from the group consisting of 2,2-dimethylpropyl, isopropyl, 2- methylcyclopropyl, 1,1-difluoropropyl, and cyclopropyl;
- R 8 is selected from the group consisting of 1,2,4-oxadiazolyl and C(0)OR 9 ; wherein the 1 ,2,4-oxadiazole is optionally substituted with one isopropyl group; and
- R 9 is selected from the group consisting of isobutyl, isopropyl, sec -butyl, tert- butyl, cyclopentyl, (3-methyloxetan-3-yl)methyl, 1-methylcyclopropyl, 1,1,1,3,3,3- hexafluoropropan-2-yl, and l,l,l-trifluoropropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ii) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein: R 1 is selected from the group consisting of cyclopropylsulfonyl,
- R 2 is selected from the group consisting of H, methylsulfonyl, cyano, 1H-1,2,4- triazol-l-yl, 2-cyanoethyl, and 2-amino-3-(3,3-difluoroazetidin-l-yl)-3-oxopropyl;
- R 3 is selected from the group consisting of ⁇ and methylsulfonyl
- R 4 is selected from the group consisting of ⁇ and fluoro
- R 5 is selected from the group consisting of ⁇ and fluoro
- R 6 is selected from the group consisting of ⁇ and fluoro.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ii) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of cyclopropylsulfonyl, isopropylsulfonyl, 3-isobutyryl, 3,3-dimethylbutanoyl, 2-methylcyclopropanecarbonyl,
- 2,2-difluorobutanoyl (3-isopropyl- l,2,4-oxadiazol-5-yl)methyl, 2-tert-b toxy-2- oxoethyl, teri-butoxycarbonyl, isopropoxycarbonyl, isobutoxycarbonyl,
- R 3 is selected from the group consisting of ⁇ and methylsulfonyl
- R 4 is selected from the group consisting of ⁇ and fluoro
- R 5 is selected from the group consisting of ⁇ and fluoro
- R 6 is selected from the group consisting of ⁇ and fluoro.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ik) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- R 1 is C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with one Ci-C 6 alkyl substituent;
- R 2 is selected from the group consisting of ⁇ and S(0) 2 R n ;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of ⁇ and halogen;
- R 9 is Ci-Ce alkyl
- R 11 is Ci-Ce alkyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ik) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with one or more substituents selected independently from the group consisting of ethyl and isopropyl;
- R 2 is methylsulfonyl
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of ⁇ , and fluoro;
- R 9 is tert-butyl
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ik) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of teri-butoxycarbonyl, 3-isopropyl- l ,2,4-oxadiazol-5-yl, and 5-ethyl-pyrimidin-2-yl;
- R 2 is methylsulfonyl;
- R 3 is H
- R 4 is selected from the group consisting of H and fluoro
- R 5 is H
- R 6 is H.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ; or
- Q is CR 4 ; Z is CR 5 ; and X is N;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of C 2 -C 6 alkenyl, Ci-C 6 alkyl, and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of H, cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H and C C 6 alkyl;
- R 7 is Ci-Ce haloalkyl
- R 8 is a five-membered heteroaryl optionally substituted with one C 1 -C6 alkyl substituent
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C6 cycloalkyl, C 1 -C6 haloalkyl, heterocyclyl, and phenyl; wherein the C 1 -C6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, C 1 -C6 alkoxy, and R 10 , wherein C 1 -C6 alkoxy is optionally substituted with phenyl; and the C 3 -C6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl and halogen;
- R 11 is selected from the group consisting of C 1 -C6 alkyl and C 3 -C6 cycloalkyl;
- R 12 and R 13 are each independently selected from the group consisting of Ci-C 6 alkyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with Ci-C 6 alkoxy.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ; or
- Q is CR 4 ; Z is CR 5 ; and X is N;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of prop-1- en-2-yl, ethyl, 2-fluoropropan-2-yl, and isopropyl;
- R 2 is selected from the group consisting of H, isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l,2,4-triazol-l- yl, l,l-dioxo-thiomorpholin-4-yl, morpholin-4-yl, cyclopropylsulfonyl, ethylsulfonyl, pyrimidin-5-yl, and 3-methoxyazetidine-l-carbonyl;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of ⁇ and methyl;
- R 7 is 1,1-difluoropropyl
- R 8 is 1 ,2,4-oxadiazolyl optionally substituted with one isopropyl group; and R 9 is selected from the group consisting of isopropyl, tert-butyl, 1- methylcyclopropyl, l,3-difluoropropan-2-yl, 1 -fluoropropan-2-yl, tetrahydrofuran-3-yl, l-hydroxypropan-2-yl, phenyl, 2,2,3,3-tetrafluorocyclobutyl, 1,1,1,3,3,3- hexafluoropropan-2-yl, l,l,l-trifluoropropan-2-yl, 1 -(benzyloxy)propan-2-yl, and 1- hydroxypropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ; or
- Q is CR 4 ; Z is CR 5 ; and X is N;
- R 1 is selected from the group consisting of 2,2-difluorobutanoyl, (3-isopropyl- l,2,4-oxadiazol-5-yl)methyl, teri-butoxycarbonyl, isopropoxycarbonyl, (1- methylcyclopropoxy)carbonyl, (tetrahydrofuran-3-yloxy)carbonyl, (1 ,3-difluoropropan- 2-yloxy)carbonyl, (l-fluoropropan-2-yloxy)carbonyl, 3-(2-fluoropropan-2-yl)- 1,2,4- oxadiazol-5-yl, 3-(prop-l-en-2-yl)-l,2,4-oxadiazol-5-yl, 5-ethyl-pyrimidin-2-yl, (1- hydroxypropan-2-yloxy)carbonyl, phenoxycarbonyl, 5-isopropyl-l ,2,4-oxadiazol-3-yl, 3-iso
- R 2 is selected from the group consisting of H, isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l,2,4-triazol-l- yl, l,l-dioxo-thiomorpholin-4-yl, mo holin-4-yl, cyclopropylsulionyl, ethylsulfonyl, pyrimidin-5-yl, and 3-methoxyazetidine-l-carbonyl;
- R 3 is selected from the group consisting of ⁇ , and methyl
- R 4 is ⁇
- R 5 is selected from the group consisting of ⁇ and methyl; and R 6 is ⁇ .
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of C 2 -C 6 alkenyl, Ci-C 6 alkyl, and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ;
- R 3 , R 5 , and R 6 are each independently selected from the group consisting of H and C 1 -C6 alkyl;
- R 7 is Ci-Cs haloalkyl
- R 8 is a five-membered heteroaryl optionally substituted with one C 1 -C6 alkyl substituent
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C 3 -C6 cycloalkyl, C 1 -C6 haloalkyl, and heterocyclyl; wherein the C 1 -C6 alkyl is optionally substituted with hydroxyl; and the C 3 -C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 11 is Ci-C 6 alkyl
- R 12 and R 13 are each independently Ci-C 6 alkyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of prop-1- en-2-yl, ethyl, and 2-fluoropropan-2-yl;
- R 2 is selected from the group consisting of isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l,2,4-triazol-l-yl, 1,1-dioxo- thiomorpholin-4-yl, and morpholin-4-yl;
- R 3 , R 5 , and R 6 are each independently selected from the group consisting of ⁇ and methyl;
- R 7 is 1,1-difluoropropyl
- R 8 is 1 ,2,4-oxadiazolyl optionally substituted with one isopropyl group; and R 9 is selected from the group consisting of isopropyl, tert-butyl, 1- methylcyclopropyl, l,3-difluoropropan-2-yl, 1 -fluoropropan-2-yl, tetrahydrofuran-3-yl, and l-hydroxypropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (la) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ;
- R 1 is selected from the group consisting of 2,2-difluorobutanoyl, (3-isopropyl- l,2,4-oxadiazol-5-yl)methyl, teri-butoxycarbonyl, isopropoxycarbonyl, (1- methylcyclopropoxy)carbonyl, (tetrahydrofuran-3-yloxy)carbonyl, (1 ,3-difluoropropan- 2-yloxy)carbonyl, (l-fluoropropan-2-yloxy)carbonyl, 3-(2-fluoropropan-2-yl)- 1,2,4- oxadiazol-5-yl, 3-(prop-l-en-2-yl)-l,2,4-oxadiazol-5-yl, 5-ethyl-pyrimidin-2-yl, and (1- hydroxypropan-2-yloxy)carbonyl;
- R 2 is isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l,2,4-triazol-l-yl, l,l-dioxo-thiomorpholin-4-yl, and morpholin-4- yl;
- R 3 is ⁇
- R 5 is selected from the group consisting of ⁇ , and methyl; and R 6 is H.
- one Q, Z, and X is N, and compounds are selected from compounds of Formula (Im) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- one Q, Z, and X is N, the stereochemistry is (lr,4r) (i.e., trans).
- compounds are selected from compounds of Formula (Io) and pharmaceutically accept
- one Q, Z, and X is N, the stereochemistry is (ls,4s) (i.e., cis).
- compounds are selected from compounds of Formula (Iq) and pharmaceutically accept
- two Q, Z, and X are N, and the stereochemistry is (lr,4r) (i.e., trans).
- compounds are selected from compounds of Formula (Is) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- two Q, Z, and X are N, the stereochemistry is (lr,4r) (i.e., trans).
- compounds are selected from compounds of Formula (Iu) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ic) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ; or
- Q is CR 4 ; Z is CR 5 ; and X is N;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of C 2 -C 6 alkenyl, Ci-C 6 alkyl, and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of H, cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of H and C C 6 alkyl;
- R 7 is Ci-Ce haloalkyl;
- R 8 is a five-membered heteroaryl optionally substituted with one Ci-C 6 alkyl substituent
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, heterocyclyl, and phenyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl, C 1 -C6 alkoxy, and R 10 , wherein C 1 -C6 alkoxy is optionally substituted with phenyl; and the C3-C6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of C 1 -C6 alkyl and halogen;
- R 11 is selected from the group consisting of C 1 -C6 alkyl and C3-C6 cycloalkyl;
- R 12 and R 1 1 3 J are each independently 12 13
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ic) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ; or
- Q is CR 4 ; Z is CR 5 ; and X is N;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of prop-1- en-2-yl, ethyl, 2-fluoropropan-2-yl, and isopropyl;
- R 2 is selected from the group consisting of H, isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l,2,4-triazol-l- yl, l,l-dioxo-thiomorpholin-4-yl, morpholin-4-yl, cyclopropylsulfonyl, ethylsulfonyl, pyrimidin-5-yl, and 3-methoxyazetidine-l-carbonyl;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of
- R 7 is 1,1-difluoropropyl
- R 8 is 1 ,2,4-oxadiazolyl optionally substituted with one isopropyl group; and R 9 is selected from the group consisting of isopropyl, tert-butyl, 1- methylcyclopropyl, l,3-difluoropropan-2-yl, 1 -fluoropropan-2-yl, tetrahydrofuran-3-yl, l-hydroxypropan-2-yl, phenyl, 2,2,3,3-tetrafluorocyclobutyl, 1,1,1,3,3,3- hexafluoropropan-2-yl, l,l,l-trifluoropropan-2-yl, and l-hydroxypropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ic) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ; or
- Q is CR 4 ; Z is CR 5 ; and X is N;
- R 1 is selected from the group consisting of 2,2-difluorobutanoyl, (3-isopropyl- l ,2,4-oxadiazol-5-yl)methyl, teri-butoxycarbonyl, isopropoxycarbonyl, (1- methylcyclopropoxy)carbonyl, (tetrahydrofuran-3-yloxy)carbonyl, (1 ,3-difluoropropan-
- R 2 is selected from the group consisting of H, isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l ,2,4-triazol-l - yl, l , l-dioxo-thiomorpholin-4-yl, mo holin-4-yl, cyclopropylsulfonyl, ethylsulfonyl, pyrimidin-5-yl, and 3-methoxyazetidine-l -carbonyl;
- R 3 is selected from the group consisting of ⁇ and methyl
- R 4 is ⁇
- R 5 is selected from the group consisting of ⁇ and methyl; and R 6 is ⁇ .
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ic) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of C 2 -C 6 alkenyl, Ci-C 6 alkyl, and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of cyano, halogen, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of
- R 7 is Ci-C 6 haloalkyl
- R 8 is a five-membered heteroaryl optionally substituted with one Ci-C 6 alkyl substituent
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, and heterocyclyl; wherein the C 1 -C6 alkyl is optionally substituted with hydroxyl; and the C3-C6 cycloalkyl is optionally substituted with one C 1 -C6 alkyl substituent;
- R 11 is Ci-C 6 alkyl
- R 12 and R 13 are each independently C 1 -C6 alkyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ic) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- Q is N; Z is N; and X is CR 6 ;
- R 1 is selected from the group consisting of C(0)R 7 , CH 2 R 8 , and C(0)OR 9 ; or R 1 is selected from the group consisting of 1 ,2,4-oxadiazolyl and pyrimidinyl, each optionally substituted with 1 substituent selected from the group consisting of prop-1- en-2-yl, ethyl, and 2-fluoropropan-2-yl;
- R 2 is selected from the group consisting of isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l,2,4-triazol-l-yl, 1,1-dioxo- thiomorpholin-4-yl, and morpholin-4-yl;
- R 3 , R 4 , R 5 , and R 6 are each independently selected from the group consisting of
- R 7 is 1,1-difluoropropyl
- R 8 is 1 ,2,4-oxadiazolyl optionally substituted with one isopropyl group; and R 9 is selected from the group consisting of isopropyl, tert-butyl, 1- methylcyclopropyl, l,3-difluoropropan-2-yl, 1 -fluoropropan-2-yl, tetrahydrofuran-3-yl, and l-hydroxypropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Ic) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N; Z is CR 5 ; and X is CR 6 ; or
- Q is N; Z is CR 5 ; and X is N; or
- R 1 is selected from the group consisting of 2,2-difluorobutanoyl, (3-isopropyl- l ,2,4-oxadiazol-5-yl)methyl, teri-butoxycarbonyl, isopropoxycarbonyl, (1- methylcyclopropoxy)carbonyl, (tetrahydrofuran-3-yloxy)carbonyl, (1 ,3-difluoropropan- 2-yloxy)carbonyl, ( 1 -fluoropropan-2-yloxy)carbonyl, 3-(2-fluoropropan-2-yl)- 1 ,2,4- oxadiazol-5-yl, 3-(prop-l -en-2-yl)-l ,2,4-oxadiazol-5-yl, 5-ethyl-pyrimidin-2-yl, and (1- hydroxypropan-2-yloxy)carbonyl;
- R 2 is selected from the group consisting of isopropylsulfonyl, methylsulfonyl, cyano, dimethylcarbamoyl, bromo, pyridazin-4-yl, lH-l ,2,4-triazol-l -yl, 1 , 1-dioxo- thiomo holin-4-yl, and morpholin-4-yl;
- R 3 is ⁇
- R 5 is selected from the group consisting of ⁇ and methyl
- R 6 is ⁇ .
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (He) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- Q is N or CR 4 ;
- R 1 is selected from the group consisting of C(0)R 7 and C(0)OR 9 ; or R 1 is 1 ,2,4- oxadiazolyl optionally substituted with 1 substituent selected from the group consisting of C 2 -C alkenyl, C 1 -C6 alkyl, and C 1 -C6 haloalkyl;
- R 2 is selected from the group consisting of H, cyano, heteroaryl, heterocyclyl, S(0) 2 R n , and C(0)NR 12 R 13 ;
- R 3 , R 4 , and R 5 are each independently selected from the group consisting of H and C 1 -C6 alkyl;
- R 7 is Ci-Cs haloalkyl
- R 9 is selected from the group consisting of C 1 -C6 alkyl, C3-C6 cycloalkyl, C 1 -C6 haloalkyl, heterocyclyl, and phenyl; wherein the Ci-C 6 alkyl is optionally substituted with one or more substituents selected independently from the group consisting of hydroxyl and Ci-C 6 alkoxy, wherein Ci-C 6 alkoxy is optionally substituted with phenyl; and the C 3 -C 6 cycloalkyl is optionally substituted with one or more substituents selected independently from the group consisting of Ci-C 6 alkyl and halogen; R 11 is selected from the group consisting of Ci-C 6 alkyl and C 3 -C 6 cycloalkyl; and
- R 12 and R 1 1 3 J are each independently Ci-C 6 alkyl; or R 12 and R 13 together with the nitrogen to which they are both bonded form a heterocyclyl optionally substituted with Ci-C 6 alkoxy.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (He) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N or CR 4 ;
- R 1 is selected from the group consisting of C(0)R 7 and C(0)OR 9 ; or R 1 is 1,2,4- oxadiazolyl optionally substituted with 1 substituent selected from the group consisting of prop-l-en-2-yl, 2-fluoropropan-2-yl, and isopropyl;
- R 2 is selected from the group consisting of H, isopropylsulfonyl, methylsulfonyl, cyano, pyridazin-4-yl, lH-l,2,4-triazol-l-yl, l,l-dioxo-thiomo holin- 4-yl, morpholin-4-yl, cyclopropylsulfonyl, bromo, dimethylcarbamoyl, ethylsulfonyl, pyrimidin-5-yl, and 3-methoxyazetidine-l-carbonyl;
- R 3 , R 4 , and R 5 are each independently selected from the group consisting of ⁇ and methyl;
- R 7 is 1,1-difluoropropyl
- R 9 is selected from the group consisting of isopropyl, tert-butyl, 1- methylcyclopropyl, l,3-difluoropropan-2-yl, 1 -fluoropropan-2-yl, tetrahydrofuran-3-yl, l-hydroxypropan-2-yl, phenyl, 2,2,3,3-tetrafluorocyclobutyl, 1,1,1,3,3,3- hexafluoropropan-2-yl, l,l,l-trifluoropropan-2-yl, l-(benzyloxy)propan-2-yl, and 1- hydroxypropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (He) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- Q is N or CR 4 ;
- R 1 is selected from the group consisting of 2,2-difluorobutanoyl, tert- butoxycarbonyl, isopropoxycarbonyl, (l-methylcyclopropoxy)carbonyl,
- R 3 , R 4 , and R 5 are each independently selected from the group consisting of ⁇ and methyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Is) and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- R 1 is selected from the group consisting of C(0)R 7 and C(0)OR 9 ; or R 1 is 1,2,4- oxadiazolyl optionally substituted with 1 substituent selected from the group consisting of C 2 -C 6 alkenyl and Ci-C 6 haloalkyl;
- R 2 is selected from the group consisting of cyano, heteroaryl, heterocyclyl, and
- R 3 and R 5 are each ⁇ ;
- R 7 is Ci-C 6 haloalkyl
- R 9 is selected from the group consisting of Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 haloalkyl, and heterocyclyl; wherein the Ci-C 6 alkyl is optionally substituted with hydroxyl; and the C 3 -C 6 cycloalkyl is optionally substituted with one Ci-C 6 alkyl substituent; and
- R 11 is Ci-C 6 alkyl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Is) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of C(0)R 7 and C(0)OR 9 ; or R 1 is 1,2,4- oxadiazolyl optionally substituted with 1 substituent selected from the group consisting of prop-l-en-2-yl and 2-fluoropropan-2-yl;
- R 2 is selected from the group consisting of isopropylsulfonyl, methylsulfonyl, cyano, pyridazin-4-yl, lH-l,2,4-triazol-l-yl, l,l-dioxo-thiomo holin-4-yl, and n ⁇ holin-4-yl;
- R 3 and R 5 are each ⁇ ; R 7 is 1 ,1-difluoropropyl; and
- R 9 is selected from the group consisting of isopropyl, tert-butyl, 1- methylcyclopropyl, l ,3-difluoropropan-2-yl, 1 -fluoropropan-2-yl, tetrahydrofuran-3-yl, and l-hydroxypropan-2-yl.
- One aspect of the present invention encompasses certain cyclohexyl derivatives selected from compounds of Formula (Is) and pharmaceutically acceptable salts, solvates, and hydrates thereof, wherein:
- R 1 is selected from the group consisting of 2,2-difluorobutanoyl, tert- butoxycarbonyl, isopropoxycarbonyl, ( 1 -methylcyclopropoxy)carbonyl,
- R 2 is selected from the group consisting of isopropylsulfonyl, methylsulfonyl, cyano, pyridazin-4-yl, lH-l ,2,4-triazol-l -yl, l , l-dioxo-thiomo holin-4-yl, and morpholin-4-yl; and
- R 3 and R 5 are each ⁇ .
- Some embodiments of the present invention include every combination of one or more compounds selected from the following group shown in Table A.
- individual compounds and chemical genera of the present invention for example those compounds found in Table A including, isomers, diastereoisomers and enantiomers thereof, encompass all pharmaceutically acceptable salts, solvates, and hydrates, thereof.
- mesoisomers of individual compounds and chemical genera of the present invention for example those compounds found in Table A, encompass all pharmaceutically acceptable salts, solvates and particularly hydrates, thereof.
- the compounds of the Formula (la) of the present invention may be prepared according to relevant published literature procedures that are used by one skilled in the art. Exemplary reagents and procedures for these reactions appear hereinafter in the working Examples.
- Protection and deprotection may be carried out by procedures generally known in the art (see, for example, Greene, T. W. and Wuts, P. G. M., Protecting Groups in Organic Synthesis, 3 rd Edition, 1999 [Wiley]).
- the present invention embraces, each isomer, each diastereoisomer, each enantiomer and mixtures thereof of each compound and generic formulae disclosed herein just as if they were each individually disclosed with the specific stereochemical designation for each chiral carbon. Separation of the individual isomers and enatiomers (such as, by chiral HPLC, recrystallization of diastereoisomeric mixtures and the like) or selective synthesis (such as, by enantiomeric selective syntheses and the like) of the individual isomers can be accomplished by application of various methods which are well known to practitioners in the art.
- compositions Compositions, Methods, Indications, Pharmaceutical Products, Combinations, and Uses of Compoounds of the Present Invention.
- compositions comprising a compound of the present invention.
- compositions comprising a compound of the present invention and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to methods for preparing a composition comprising the step of admixing a compound of the present invention and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to pharmaceutical products selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention.
- compositions comprising a compound of the present invention and a second pharmaceutical agent.
- a pharmaceutical agent and “a second pharmaceutical agent”
- these terms in some aspects be further limited to a pharmaceutical agent that is not a compound of Formula (la).
- a pharmaceutical agent and “a second pharmaceutical agent” may refer to a pharmaceutical agent that is not detectable or has an EC 50 that is greater than a value selected from: 50 ⁇ , 10 ⁇ , 1 ⁇ , and 0.1 ⁇ in a GPR119 receptor activity assay as described in Example 4.
- One aspect of the present invention pertains to methods for preparing a composition comprising the step of admixing a compound of the present invention and a second
- compositions comprising a compound of the present invention, a second pharmaceutical agent, and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to methods for preparing a composition
- methods for preparing a composition comprising the step of admixing a compound of the present invention, a second pharmaceutical agent, and a pharmaceutically acceptable carrier.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention and a second pharmaceutical agent.
- One aspect of the present invention pertains to compositions obtained by the methods of the present invention as described herein.
- One aspect of the present invention pertains to methods for modulating the activity of a
- GPR119 receptor comprising administering to an individual in need thereof, a therapeutically effective amount of: a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention.
- One aspect of the present invention pertains to the use of a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention; in the manufacture of a medicament for modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention; for use in a method of treating the human or animal by therapy.
- One aspect of the present invention pertains to a compound of the present invention; a composition of the present invention; or a pharmaceutical product of the present invention; for use in a method of modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention; for use in a method of treating the human or animal by therapy.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention; for modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to methods for modulating the activity of a GPRl 19 receptor, comprising administering to an individual in need thereof, a therapeutically effective amount of a compound of the present invention in combination with a therapeutically effective amount of a second pharmaceutical agent.
- One aspect of the present invention pertains to methods for agonizing a GPRl 19 receptor, comprising administering to an individual in need thereof, a therapeutically effective amount of a compound of the present invention in combination with a therapeutically effective amount of a second pharmaceutical agent.
- One aspect of the present invention pertains to methods for the treatment of a disorder selected from: a GPRl 19-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic -related disorder; and obesity; in an individual; comprising administering to said individual in need thereof, a therapeutically effective amount of a compound of the present invention in combination with a therapeutically effective amount of a second pharmaceutical agent.
- One aspect of the present invention pertains to the use of a compound of the present invention in combination with a second pharmaceutical agent in the manufacture of a medicament for modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to the use of a compound of the present invention in combination with a second pharmaceutical agent in the manufacture of a medicament for agonizing a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to the use of a compound of the present invention in combination with a second pharmaceutical agent, in the manufacture of a medicament for the treatment of a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic -related disorder; and obesity.
- a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic -related disorder; and obesity.
- One aspect of the present invention pertains to a compound of the present invention for use in combination with a second pharmaceutical agent for use in a method of treating the human or animal by therapy.
- One aspect of the present invention pertains to a compound of the present invention for use in combination with a second pharmaceutical agent for modulating the activity of a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to a compound of the present invention for use in combination with a second pharmaceutical agent for agonizing a GPRl 19 receptor in an individual.
- One aspect of the present invention pertains to a compound of the present invention for use in combination with a second pharmaceutical agent for the treatment of a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic - related disorder; and obesity; in an individual.
- a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic - related disorder; and obesity; in an individual.
- the second pharmaceutical agent is selected from: an inhibitor of DPP-IV, a biguanide, an alpha-glucosidase inhibitor, a sulfonylurea, a SGLT2 inhibitor, and a meglitinide. In some embodiments, the second pharmaceutical agent is selected from:
- sitagliptin sitagliptin, vildagliptin, saxagliptin, alogliptin, linagliptin, phenformin, metformin, buformin, acarbose, miglitol, voglibose, tolbutamide, acetohexamide, tolazamide, chlorpropamide, glipizide, glibenclamide, glimepiride, gliclazide, dapagliflozin, remigliflozin, and sergliflozin.
- the disorder is type 2 diabetes. In some embodiments, the disorder is hyperglycemia. In some embodiments, the disorder is hyperlipidemia. In some embodiments, the disorder is hypertriglyceridemia. In some embodiments, the disorder is type 1 diabetes. In some embodiments, the disorder is dyslipidemia. In some embodiments, the disorder is syndrome X. In some embodiments, the disorder is obesity.
- One aspect of the present invention pertains to the use of a pharmaceutical agent in combination with a compound of the present invention, in the manufacture of a medicament for modulating the activity of a GPR119 receptor in an individual.
- One aspect of the present invention pertains to the use of a pharmaceutical agent in combination with a compound of the present invention, in the manufacture of a medicament for increasing the secretion of an incretin level in an individual.
- One aspect of the present invention pertains to the use of a pharmaceutical agent in combination with a compound of the present invention, in the manufacture of a medicament for increasing a blood incretin level in an individual.
- One aspect of the present invention pertains to the use of a pharmaceutical agent in combination with a compound of the present invention, in the manufacture of a medicament for the treatment of a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic -related disorder; and obesity.
- a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic -related disorder; and obesity.
- One aspect of the present invention pertains to a pharmaceutical agent for use in combination with a compound of the present invention, for use in a method of treating the human or animal by therapy.
- One aspect of the present invention pertains to a pharmaceutical agent for use in combination with a compound of the present invention, for use in combination with a pharmaceutical agent for modulating the activity of a GPR119 receptor in an individual.
- One aspect of the present invention pertains to a pharmaceutical agent for use in combination with a compound of the present invention, for increasing the secretion of an incretin level in an individual.
- One aspect of the present invention pertains to a pharmaceutical agent for use in combination with a compound of the present invention, for use in a method for increasing a blood incretin level in an individual.
- One aspect of the present invention pertains to a pharmaceutical agent for use in combination with a compound of the present invention, for the treatment of a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic- related disorder; and obesity; in an individual.
- a disorder selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level, a condition characterized by low bone mass; a neurological disorder; a metabolic- related disorder; and obesity; in an individual.
- the pharmaceutical agent is selected from: an inhibitor of DPP- IV, a biguanide, an alpha-glucosidase inhibitor, a sulfonylurea, a SGLT2 inhibitor, and a meglitinide.
- the pharmaceutical agent is selected from: sitagliptin, vildagliptin, saxagliptin, alogliptin, linagliptin, phenformin, metformin, buformin, acarbose, miglitol, voglibose, tolbutamide, acetohexamide, tolazamide, chlorpropamide, glipizide, glibenclamide, glimepiride, gliclazide, dapagliflozin, remigliflozin, and sergliflozin.
- the disorder is type 2 diabetes. In some embodiments, the disorder is hyperglycemia.
- the disorder is hyperlipidemia. In some embodiments, the disorder is hypertriglyceridemia. In some embodiments, the disorder is type 1 diabetes. In some embodiments, the disorder is dyslipidemia. In some embodiments, the disorder is syndrome X. In some embodiments, the disorder is obesity.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention and a second pharmaceutical agent; for use in a method of treating the human or animal by therapy.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention and a second pharmaceutical agent; for modulating the activity of a GPR119 receptor in an individual.
- One aspect of the present invention pertains to methods for modulating the activity of a GPR119 receptor, comprising administering to an individual in need thereof, a therapeutically effective amount of a compound of the present invention and an inhibitor of DPP-IV.
- One aspect of the present invention pertains to compounds of the present invention for use in combination with an inhibitor of DPP-IV for modulating the activity of a GPR119 receptor in an individual.
- One aspect of the present invention pertains to inhibitors of DPP-IV in combination with a compound of the present invention, for use in modulating the activity of a GPR119 receptor.
- One aspect of the present invention pertains to a pharmaceutical product selected from: a pharmaceutical composition, a formulation, a dosage form, a combined preparation, a twin pack, and a kit; comprising a compound of the present invention and an inhibitor of DPP-IV; for modulating the activity of a GPR119 receptor in an individual.
- One aspect of the present invention pertains to the use of a compound of the present invention and an inhibitor of DPP-IV in the manufacture of a medicament for modulating the activity of a GPR119 receptor in an individual.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, pharmaceutical agents, pharmaceutical products, and inhibitors of DPP-IV, as described herein, wherein modulating the activity of a GPR119 receptor is agonizing the
- GPR119 receptor in an individual is GPR119 receptor in an individual.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, pharmaceutical agents, pharmaceutical products, and inhibitors of DPP-IV, as described herein, wherein modulating the activity of a GPR119 receptor is increasing the secretion of an incretin in an individual.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, pharmaceutical agents, pharmaceutical products, and inhibitors of DPP-IV, as described herein, wherein modulating the activity of a GPRl 19 receptor is increasing a blood incretin level in an individual.
- One aspect of the present invention pertains to compounds, methods, compositions, uses of compounds, pharmaceutical agents, pharmaceutical products, and inhibitors of DPP-IV, as described herein, wherein modulating the activity of a GPRl 19 receptor treating a disorder, wherein the disorder is selected from: a GPR119-receptor-related disorder; a condition ameliorated by increasing a blood incretin level; a condition characterized by low bone mass; a neurological disorder; a metabolic -related disorder; and obesity.
- the pharmaceutical product comprises a pharmaceutical composition. In some embodiments, the pharmaceutical product comprises a formulation. In some embodiments, the pharmaceutical product comprises a dosage form. In some
- the pharmaceutical product comprises a combined preparation. In some embodiments, the pharmaceutical product comprises a twin pack. In some embodiments, the pharmaceutical product comprises a kit.
- the compound and the pharmaceutical agent or second pharmaceutical agent are administered simultaneously. In some embodiments, the compound and the pharmaceutical agent or second pharmaceutical agent are administered separately. In some embodiments, the compound and the pharmaceutical agent or second pharmaceutical agent are administered sequentially.
- the incretin is GLP-1. In some embodiments, the incretin is GIP. In some embodiments, the incretin is PYY.
- the compound and the pharmaceutical agent or second pharmaceutical agent are provided in amounts which give a synergistic effect in treating the disorder.
- the amount of the compound alone is substantially
- the amount of the pharmaceutical agent alone is substantially therapeutically ineffective at treating the disorder.
- One aspect of the present invention pertains to methods for preparing a pharmaceutical product, as described herein, comprising: mixing the compound of the present invention with a first pharmaceutically acceptable carrier to prepare a compound dosage form, mixing the second pharmaceutical agent with a second pharmaceutically acceptable carrier to prepare a second pharmaceutical agent dosage form, and providing the compound dosage form and the second pharmaceutical agent dosage form in a combined dosage form for simultaneous, separate, or sequential use.
- the first pharmaceutically acceptable carrier and the second pharmaceutically acceptable carrier are different.
- the different pharmaceutically acceptable carriers are suitable for administration by the same route or different routes.
- the first pharmaceutically acceptable carrier and the second pharmaceutically acceptable carrier are substantially the same.
- the substantially the same pharmaceutically acceptable carriers are suitable for administration by the same route.
- the substantially the same pharmaceutically acceptable carriers are suitable for oral administration.
- the pharmaceutical agent or the second pharmaceutical agent is selected from: an inhibitor of DPP-IV, a biguanide, an alpha-glucosidase inhibitor, an insulin analogue, a sulfonylurea, a SGLT2 inhibitor, a meglitinide, a thiazolidinedione, and an antidiabetic peptide analogue.
- the pharmaceutical agent or the second pharmaceutical agent is selected from: an inhibitor of DPP-IV, a biguanide, an alpha- glucosidase inhibitor, a sulfonylurea, a SGLT2 inhibitor, and a meglitinide.
- the pharmaceutical agent or the second pharmaceutical agent is selected from: an inhibitor of DPP-IV, a biguanide, and an alpha-glucosidase inhibitor.
- the pharmaceutical agent or the second pharmaceutical agent is an inhibitor of DPP-IV.
- the pharmaceutical agent or the second pharmaceutical agent is a biguanide.
- the pharmaceutical agent or the second pharmaceutical agent is an alpha- glucosidase inhibitor.
- the pharmaceutical agent or the second pharmaceutical agent is a sulfonylurea.
- the pharmaceutical agent or the second pharmaceutical agent is a SGLT2 inhibitor.
- the pharmaceutical agent or the second pharmaceutical agent is a meglitinide.
- the pharmaceutical agent or the second pharmaceutical agent is a biguanide selected from the following compounds and pharmaceutically acceptable salts, solvates, and hydrates thereof: metformin, phenformin, buformin, and proguanil.
- the pharmaceutical agent or the second pharmaceutical agent is an alpha-glucosidase inhibitor selected from the following compounds and pharmaceutically acceptable salts, solvates, and hydrates thereof: acarbose, miglitol, and voglibose.
- the pharmaceutical agent or the second pharmaceutical agent is a sulfonylurea selected from the following compounds and pharmaceutically acceptable salts, solvates, and hydrates thereof: here herein
- the pharmaceutical agent or the second pharmaceutical agent is a SGLT2 inhibitor selected from the following compounds and pharmaceutically acceptable salts, solvates, and hydrates thereof: In some embodiments, the pharmaceutical agent or the second pharmaceutical agent is a meglitinide selected from the following compounds and pharmaceutically acceptable salts, solvates, and hydrates thereof:
- One aspect of the present invention pertains to methods for weight management, comprising administering to an individual in need thereof, a therapeutically effective amount of a compound of the present invention in combination with a therapeutically effective amount of a pharmaceutical agent, such as any agent described herein; wherein the compound and the pharmaceutical agent.
- the weight management comprises weight loss. In some embodiments, the weight management comprises maintenance of weight loss. In some embodiments, the weight management further comprises a reduced-calorie diet. In some embodiments, the weight management further comprises a program of regular exercise. In some embodiments, the weight management further comprises both a reduced-calorie diet and a program of regular exercise.
- the individual in need of weight management is a patient with an initial body mass of index > 40 kg/m 2 ; > 39 kg/m 2 ; > 38 kg/m 2 ; > 37 kg/m 2 ; > 36 kg/m 2 ; > 35 kg/m 2 ; > 34 kg/m 2 ; > 33 kg/m 2 ; > 32 kg/m 2 ; > 31 kg/m 2 ; > 30 kg/m 2 ; > 29 kg/m 2 ; > 28 kg/m 2 ; > 27 kg/m 2 ; > 26 kg/m 2 ; > 25 kg/m 2 ; > 24 kg/m 2 ; > 23 kg/m 2 ; > 22 kg/m 2 ; > 21 kg/m 2 ; or > 20 kg/m 2 ; and the patient optionally has at least one or at least two weight related comorbid condition(s).
- the comorbid condition(s) when present are selected from: hypertension, dyslipidemia, cardiovascular disease, glucose intolerance, and sleep apnea.
- a compound as described herein or a pharmaceutical composition thereof can be utilized for modulating the activity of GPR119- receptor-related diseases, conditions and/or disorders as described herein.
- modulating the activity includes the treatment of a GPR119- receptor-related disorder.
- a GPR119-receptor-related disorder is a condition ameliorated by increasing a blood incretin level.
- a GPR119- receptor-related disorder is a condition characterized by low bone mass.
- a GPR119-receptor-related disorder is a neurological disorder.
- a GPR119-receptor-related disorder is a metabolic-related disorder.
- a GPR119-receptor-related disorder is obesity
- Some embodiments of the present invention include every combination of one or more conditions characterized by low bone mass selected from: osteopenia, osteoporosis, rheumatoid arthritis, osteoarthritis, periodontal disease, alveolar bone loss, osteotomy bone loss, childhood idiopathic bone loss, Paget' s disease, bone loss due to metastatic cancer, osteolytic lesions, curvature of the spine, and loss of height.
- the neurological disorder selected from: stroke and
- Some embodiments of the present invention include every combination of one or more metabolic -related disorders selected from: type 1 diabetes, type 2 diabetes mellitus, and conditions associated therewith, such as, but not limited to, coronary heart disease, ischemic stroke, restenosis after angioplasty, peripheral vascular disease, intermittent claudication, myocardial infarction (e.g.
- necrosis and apoptosis dyslipidemia, post-prandial lipemia, conditions of impaired glucose tolerance (IGT), conditions of impaired fasting plasma glucose, metabolic acidosis, ketosis, arthritis, osteoporosis, hypertension, congestive heart failure, left ventricular hypertrophy, peripheral arterial disease, diabetic retinopathy, macular degeneration, cataract, diabetic nephropathy, glomerulosclerosis, chronic renal failure, diabetic neuropathy, metabolic syndrome, syndrome X, premenstrual syndrome, coronary heart disease, angina pectoris, thrombosis, atherosclerosis, myocardial infarction, transient ischemic attacks, stroke, vascular restenosis, hyperglycemia, hyperinsulinemia, hyperlipidemia, hypertriglyceridemia, insulin resistance, impaired glucose metabolism, erectile dysfunction, skin and connective tissue disorders, foot ulcerations and ulcerative colitis, endothelial dysfunction and impaired vascular compliance.
- ITT impaired glucose tolerance
- Some embodiments of the present invention include every combination of one or more metabolic -related disorders selected from: diabetes, type 1 diabetes, type 2 diabetes, inadequate glucose tolerance, impaired glucose tolerance, insulin resistance, hyperglycemia,
- hyperlipidemia hypertriglyceridemia, hypercholesterolemia, dyslipidemia, atherosclerosis, stroke, syndrome X, hypertension, pancreatic beta-cell insufficiency, enteroendocrine cell insufficiency, glucosuria, metabolic acidosis, cataracts, diabetic nephropathy, diabetic neuropathy, peripheral neuropathy, diabetic coronary artery disease, diabetic cerebrovascular disease, diabetic peripheral vascular disease, diabetic retinopathy, metabolic syndrome, a condition related to diabetes, myocardial infarction, learning impairment, memory impairment, a neurodegenerative disorder, a condition ameliorated by increasing a blood GLP-1 level in an individual with a neurodegenerative disorder, excitotoxic brain damage caused by severe epileptic seizures, Alzheimer's disease, Parkinson's disease, Huntington's disease, prion- associated disease, stroke, motor-neuron disease, traumatic brain injury, spinal cord injury, and obesity.
- the disorder is type 2 diabetes. In some embodiments, the disorder is hyperglycemia. In some embodiments, the disorder is hyperlipidemia. In some embodiments, the disorder is hypertriglyceridemia. In some embodiments, the disorder is type 1 diabetes. In some embodiments, the disorder is dyslipidemia. In some embodiments, the disorder is syndrome X. In some embodiments, the disorder is obesity.
- Formulations may be prepared by any suitable method, typically by uniformly mixing the active compound(s) with liquids or finely divided solid carriers, or both, in the required proportions and then, if necessary, forming the resulting mixture into a desired shape.
- excipients such as binding agents, fillers, acceptable wetting agents, tabletting lubricants and disintegrants may be used in tablets and capsules for oral
- Liquid preparations for oral administration may be in the form of solutions, emulsions, aqueous or oily suspensions and syrups.
- the oral preparations may be in the form of dry powder that can be reconstituted with water or another suitable liquid vehicle before use. Additional additives such as suspending or emulsifying agents, non-aqueous vehicles (including edible oils), preservatives and flavorings and colorants may be added to the liquid preparations.
- Parenteral dosage forms may be prepared by dissolving the compound of the invention in a suitable liquid vehicle and filter sterilizing the solution before filling and sealing an appropriate vial or ampule. These are just a few examples of the many appropriate methods well known in the art for preparing dosage forms.
- a compound of the present invention can be formulated into pharmaceutical compositions using techniques well known to those in the art. Suitable pharmaceutically- acceptable carriers, outside those mentioned herein, are known in the art; for example, see Remington, The Science and Practice of Pharmacy, 20 th Edition, 2000, Lippincott Williams & Wilkins, (Editors: Gennaro et al.)
- a compound of the invention may, in an alternative use, be administered as a raw or pure chemical, it is preferable however to present the compound or active ingredient as a pharmaceutical formulation or composition further comprising a pharmaceutically acceptable carrier.
- Transdermal patches dispense a drug at a controlled rate by presenting the drug for absorption in an efficient manner with minimal degradation of the drug.
- transdermal patches comprise an impermeable backing layer, a single pressure sensitive adhesive and a removable protective layer with a release liner.
- the compounds of the invention may thus be placed into the form of pharmaceutical formulations and unit dosages thereof and in such form may be employed as solids, such as tablets or filled capsules, or liquids such as solutions, suspensions, emulsions, elixirs, gels or capsules filled with the same, all for oral use, in the form of suppositories for rectal administration; or in the form of sterile injectable solutions for parenteral (including subcutaneous) use.
- Such pharmaceutical compositions and unit dosage forms thereof may comprise conventional ingredients in conventional proportions, with or without additional active compounds or principles and such unit dosage forms may contain any suitable effective amount of the active ingredient commensurate with the intended daily dosage range to be employed.
- the pharmaceutical composition may be in the form of, for example, a tablet, capsule, suspension or liquid.
- the pharmaceutical composition is preferably made in the form of a dosage unit containing a particular amount of the active ingredient.
- dosage units are capsules, tablets, powders, granules or a suspension, with conventional additives such as lactose, mannitol, corn starch or potato starch; with binders such as crystalline cellulose, cellulose derivatives, acacia, corn starch or gelatins; with disintegrators such as corn starch, potato starch or sodium carboxymethyl-cellulose; and with lubricants such as talc or magnesium stearate.
- the active ingredient may also be administered by injection as a composition wherein, for example, saline, dextrose or water may be used as a suitable pharmaceutically acceptable carrier.
- active ingredient defined in the context of a “pharmaceutical composition” refers to a component of a pharmaceutical composition that provides the primary pharmacological effect, as opposed to an "inactive ingredient” which would generally be recognized as providing no pharmaceutical benefit.
- the dose when using the compounds of the present invention can vary within wide limits and as is customary and is known to the physician, it is to be tailored to the individual conditions in each individual case. It depends, for example, on the nature and severity of the illness to be treated, on the condition of the patient, on the compound employed or on whether an acute or chronic disease state is treated or prophylaxis conducted or on whether further active compounds are administered in addition to the compounds of the present invention.
- Representative doses of the present invention include, but not limited to, about 0.001 mg to about 5000 mg, about 0.001 mg to about 2500 mg, about 0.001 mg to about 1000 mg, 0.001 mg to about 500 mg, 0.001 mg to about 250 mg, about 0.001 mg to 100 mg, about 0.001 mg to about 50 mg and about 0.001 mg to about 25 mg.
- Multiple doses may be administered during the day, especially when relatively large amounts are deemed to be needed, for example 2, 3 or 4 doses. Depending on the individual and as deemed appropriate from the patient's physician or caregiver it may be necessary to deviate upward or downward from the doses described herein.
- the amount of active ingredient, or an active salt or derivative thereof, required for use in treatment will vary not only with the particular salt selected but also with the route of administration, the nature of the condition being treated and the age and condition of the patient and will ultimately be at the discretion of the attendant physician or clinician.
- a model system typically an animal model
- these extrapolations may merely be based on the weight of the animal model in comparison to another, such as a mammal, preferably a human, however, more often, these extrapolations are not simply based on weights, but rather incorporate a variety of factors.
- compositions of this invention are selected in accordance with a variety factors as cited above.
- the actual dosage regimen employed may vary widely and therefore may deviate from a preferred dosage regimen and one skilled in the art will recognize that dosage and dosage regimen outside these typical ranges can be tested and, where appropriate, may be used in the methods of this invention.
- the desired dose may conveniently be presented in a single dose or as divided doses administered at appropriate intervals, for example, as two, three, four or more sub-doses per day.
- the sub-dose itself may be further divided, e.g., into a number of discrete loosely spaced administrations.
- the daily dose can be divided, especially when relatively large amounts are administered as deemed appropriate, into several, for example 2, 3 or 4 part administrations. If appropriate, depending on individual behavior, it may be necessary to deviate upward or downward from the daily dose indicated.
- the compounds of the present invention can be administrated in a wide variety of oral and parenteral dosage forms. It will be obvious to those skilled in the art that the following dosage forms may comprise, as the active component, either a compound of the invention or a pharmaceutically acceptable salt, solvate, or hydrate of a compound of the invention.
- a suitable pharmaceutically acceptable carrier can be either solid, liquid or a mixture of both.
- Solid form preparations include powders, tablets, pills, capsules, cachets, suppositories and dispersible granules.
- a solid carrier can be one or more substances which may also act as diluents, flavoring agents, solubilizers, lubricants, suspending agents, binders, preservatives, tablet disintegrating agents, or an encapsulating material.
- the carrier is a finely divided solid which is in a mixture with the finely divided active component.
- the active component is mixed with the carrier having the necessary binding capacity in suitable proportions and compacted to the desire shape and size.
- the powders and tablets may contain varying percentage amounts of the active compound.
- a representative amount in a powder or tablet may contain from 0.5 to about 90 percent of the active compound; however, an artisan would know when amounts outside of this range are necessary.
- Suitable carriers for powders and tablets are magnesium carbonate, magnesium stearate, talc, sugar, lactose, pectin, dextrin, starch, gelatin, tragacanth, methylcellulose, sodium carboxymethylcellulose, a low melting wax, cocoa butter and the like.
- the term "preparation” refers to the formulation of the active compound with encapsulating material as carrier providing a capsule in which the active component, with or without carriers, is surrounded by a carrier, which is thus in association with it. Similarly, cachets and lozenges are included.
- Tablets, powders, capsules, pills, cachets and lozenges can be used as solid forms suitable for oral administration.
- a low melting wax such as an admixture of fatty acid glycerides or cocoa butter
- the active component is dispersed homogeneously therein, as by stirring.
- the molten homogenous mixture is then poured into convenient sized molds, allowed to cool and thereby to solidify.
- Formulations suitable for vaginal administration may be presented as pessaries, tampons, creams, gels, pastes, foams or sprays containing in addition to the active ingredient such carriers as are known in the art to be appropriate.
- Liquid form preparations include solutions, suspensions and emulsions, for example, water or water-propylene glycol solutions.
- parenteral injection liquid preparations can be formulated as solutions in aqueous polyethylene glycol solution.
- injectable preparations for example, sterile injectable aqueous or oleaginous suspensions may be formulated according to the known art using suitable dispersing or wetting agents and suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a nontoxic parenterally acceptable diluent or solvent, for example, as a solution in 1,3-butanediol.
- the acceptable vehicles and solvents that may be employed are water, Ringer's solution and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- fatty acids such as oleic acid find use in the preparation of injectables.
- the compounds according to the present invention may thus be formulated for parenteral administration (e.g. by injection, for example bolus injection or continuous infusion) and may be presented in unit dose form in ampoules, pre-filled syringes, small volume infusion or in multi-dose containers with an added preservative.
- the pharmaceutical compositions may take such forms as suspensions, solutions, or emulsions in oily or aqueous vehicles and may contain formulatory agents such as suspending, stabilizing and/or dispersing agents.
- the active ingredient may be in powder form, obtained by aseptic isolation of sterile solid or by lyophilization from solution, for constitution with a suitable vehicle, e.g. sterile, pyrogen-free water, before use.
- a suitable vehicle e.g. sterile, pyrogen-free water
- Aqueous formulations suitable for oral use can be prepared by dissolving or suspending the active component in water and adding suitable colorants, flavors, stabilizing and thickening agents, as desired.
- Aqueous suspensions suitable for oral use can be made by dispersing the finely divided active component in water with viscous material, such as natural or synthetic gums, resins, methylcellulose, sodium carboxymethylcellulose, or other well-known suspending agents.
- viscous material such as natural or synthetic gums, resins, methylcellulose, sodium carboxymethylcellulose, or other well-known suspending agents.
- solid form preparations which are intended to be converted, shortly before use, to liquid form preparations for oral administration.
- liquid forms include solutions, suspensions and emulsions.
- These preparations may contain, in addition to the active component, colorants, flavors, stabilizers, buffers, artificial and natural sweeteners, dispersants, thickeners, solubilizing agents and the like.
- the compounds according to the invention may be formulated as ointments, creams or lotions, or as a transdermal patch.
- Ointments and creams may, for example, be formulated with an aqueous or oily base with the addition of suitable thickening and/or gelling agents.
- Lotions may be formulated with an aqueous or oily base and will in general also contain one or more emulsifying agents, stabilizing agents, dispersing agents, suspending agents, thickening agents, or coloring agents.
- Formulations suitable for topical administration in the mouth include lozenges comprising active agent in a flavored base, usually sucrose and acacia or tragacanth; pastilles comprising the active ingredient in an inert base such as gelatin and glycerin or sucrose and acacia; and mouthwashes comprising the active ingredient in a suitable liquid carrier.
- Solutions or suspensions are applied directly to the nasal cavity by conventional means, for example with a dropper, pipette or spray.
- the formulations may be provided in single or multi-dose form. In the latter case of a dropper or pipette, this may be achieved by the patient administering an appropriate, predetermined volume of the solution or suspension. In the case of a spray, this may be achieved for example by means of a metering atomizing spray pump.
- Administration to the respiratory tract may also be achieved by means of an aerosol formulation in which the active ingredient is provided in a pressurized pack with a suitable propellant.
- aerosol formulation in which the active ingredient is provided in a pressurized pack with a suitable propellant.
- the compounds of the present invention or pharmaceutical compositions comprising them are administered as aerosols, for example as nasal aerosols or by inhalation, this can be carried out, for example, using a spray, a nebulizer, a pump nebulizer, an inhalation apparatus, a metered inhaler or a dry powder inhaler.
- Pharmaceutical forms for administration of the compounds of the present invention as an aerosol can be prepared by processes well known to the person skilled in the art.
- solutions or dispersions of the compounds of the present invention in water, water/alcohol mixtures or suitable saline solutions can be employed using customary additives, for example benzyl alcohol or other suitable preservatives, absorption enhancers for increasing the bioavailability, solubilizers, dispersants and others and, if appropriate, customary propellants, for example include carbon dioxide,
- CFCs such as, dichlorodifluoromethane, trichlorofluorome thane, or dichlorotetrafluoroe thane; and the like.
- the aerosol may conveniently also contain a surfactant such as lecithin.
- the dose of drug may be controlled by provision of a metered valve.
- the compound In formulations intended for administration to the respiratory tract, including intranasal formulations, the compound will generally have a small particle size for example of the order of 10 microns or less. Such a particle size may be obtained by means known in the art, for example by micronization. When desired, formulations adapted to give sustained release of the active ingredient may be employed.
- the active ingredients may be provided in the form of a dry powder, for example, a powder mix of the compound in a suitable powder base such as lactose, starch, starch derivatives such as hydroxypropylmethyl cellulose and polyvinylpyrrolidone (PVP).
- a powder mix of the compound in a suitable powder base such as lactose, starch, starch derivatives such as hydroxypropylmethyl cellulose and polyvinylpyrrolidone (PVP).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Physical Education & Sports Medicine (AREA)
- Diabetes (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Rheumatology (AREA)
- Endocrinology (AREA)
- Neurosurgery (AREA)
- Child & Adolescent Psychology (AREA)
- Neurology (AREA)
- Emergency Medicine (AREA)
- Biomedical Technology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Hydrogenated Pyridines (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013503841A JP2013523819A (en) | 2010-04-06 | 2011-04-05 | GPR119 receptor modulators and treatment of disorders related thereto |
| EP11715123A EP2556056A1 (en) | 2010-04-06 | 2011-04-05 | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| CN2011800275776A CN102918027A (en) | 2010-04-06 | 2011-04-05 | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| CA2795513A CA2795513A1 (en) | 2010-04-06 | 2011-04-05 | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| AU2011237775A AU2011237775A1 (en) | 2010-04-06 | 2011-04-05 | Modulators of the GPR119 receptor and the treatment of disorders related thereto |
| BR112012025592A BR112012025592A2 (en) | 2010-04-06 | 2011-04-05 | gpr119 receptor modulators and the treatment of disorders related thereto |
| MX2012011631A MX2012011631A (en) | 2010-04-06 | 2011-04-05 | Modulators of the gpr119 receptor and the treatment of disorders related thereto. |
| US13/639,652 US20130023494A1 (en) | 2010-04-06 | 2011-04-05 | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US34186610P | 2010-04-06 | 2010-04-06 | |
| US61/341,866 | 2010-04-06 | ||
| US40186310P | 2010-08-20 | 2010-08-20 | |
| US61/401,863 | 2010-08-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011127051A1 true WO2011127051A1 (en) | 2011-10-13 |
Family
ID=44151966
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2011/031243 WO2011127051A1 (en) | 2010-04-06 | 2011-04-05 | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20130023494A1 (en) |
| EP (1) | EP2556056A1 (en) |
| JP (1) | JP2013523819A (en) |
| CN (1) | CN102918027A (en) |
| AU (1) | AU2011237775A1 (en) |
| BR (1) | BR112012025592A2 (en) |
| CA (1) | CA2795513A1 (en) |
| MX (1) | MX2012011631A (en) |
| WO (1) | WO2011127051A1 (en) |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012170702A1 (en) * | 2011-06-08 | 2012-12-13 | Arena Pharmaceuticals, Inc. | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| WO2012170867A1 (en) | 2011-06-09 | 2012-12-13 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of gpr-119 |
| EP2576507A1 (en) * | 2010-06-04 | 2013-04-10 | Sanofi | Hexafluoroisopropyl carbamate derivatives, their preparation and their therapeutic application |
| WO2013055910A1 (en) * | 2011-10-12 | 2013-04-18 | Arena Pharmaceuticals, Inc. | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| WO2014011926A1 (en) | 2012-07-11 | 2014-01-16 | Elcelyx Therapeutics, Inc. | Compositions comprising statins, biguanides and further agents for reducing cardiometabolic risk |
| WO2014074700A1 (en) * | 2012-11-08 | 2014-05-15 | Arena Pharmaceuticals, Inc. | Antagonists of gpr119 receptor and the treatment of disorders related thereto |
| WO2014074668A1 (en) | 2012-11-08 | 2014-05-15 | Arena Pharmaceuticals, Inc. | Modulators of gpr119 and the treatment of disorders related thereto |
| US8754226B2 (en) | 2010-05-17 | 2014-06-17 | Array Biopharma Inc. | Piperidinyl-substituted lactams as GPR119 modulators |
| EP2771000A1 (en) * | 2011-10-24 | 2014-09-03 | Merck Sharp & Dohme Corp. | Substituted piperidinyl compounds useful as gpr119 agonists |
| US8933083B2 (en) | 2003-01-14 | 2015-01-13 | Arena Pharmaceuticals, Inc. | 1,2,3-trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto such as diabetes and hyperglycemia |
| JP2015522559A (en) * | 2012-06-12 | 2015-08-06 | チョン クン ダン ファーマシューティカル コーポレーション | Piperidine derivatives as GPR119 agonists |
| US9422266B2 (en) | 2011-09-30 | 2016-08-23 | Merck Sharp & Dohme Corp. | Substituted cyclopropyl compounds, compositions containing such compounds and methods of treatment |
| JP2016538296A (en) * | 2013-11-26 | 2016-12-08 | チョン クン ダン ファーマシューティカル コーポレーション | Amide derivatives as GPR119 agonists |
| US10894787B2 (en) | 2010-09-22 | 2021-01-19 | Arena Pharmaceuticals, Inc. | Modulators of the GPR119 receptor and the treatment of disorders related thereto |
| US11007175B2 (en) | 2015-01-06 | 2021-05-18 | Arena Pharmaceuticals, Inc. | Methods of treating conditions related to the S1P1 receptor |
| US11534424B2 (en) | 2017-02-16 | 2022-12-27 | Arena Pharmaceuticals, Inc. | Compounds and methods for treatment of primary biliary cholangitis |
| US11884626B2 (en) | 2015-06-22 | 2024-01-30 | Arena Pharmaceuticals, Inc. | Crystalline L-arginine salt of (R)-2-(7-(4-cyclopentyl-3-(trifluoromethyl)benzyloxy)-1,2,3,4-tetrahydrocyclo-penta [b]indol-3-yl)acetic acid(Compound1) for use in S1P1 receptor-associated disorders |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101526592B1 (en) * | 2009-09-17 | 2015-06-05 | 주식회사 엘지생활건강 | Anti-wrinkle composition |
| EP2528894A1 (en) | 2010-01-27 | 2012-12-05 | Arena Pharmaceuticals, Inc. | Processes for the preparation of (r)-2-(7-(4-cyclopentyl-3-(trifluoromethyl)benzyloxy)-1,2,3,4-tetrahydrocyclopenta[b]indol-3-yl)acetic acid and salts thereof |
| CN102731492B (en) * | 2011-03-30 | 2016-06-29 | 江苏恒瑞医药股份有限公司 | Cyclohexanes derivant, its preparation method and in application pharmaceutically |
| AR111199A1 (en) * | 2017-03-31 | 2019-06-12 | Takeda Pharmaceuticals Co | GPR40 AGONIST AROMATIC COMPOUND |
| WO2023034530A1 (en) | 2021-09-02 | 2023-03-09 | Teon Therapeutics, Inc. | Methods of improving growth and function of immune cells |
Citations (241)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1991016339A1 (en) | 1990-04-14 | 1991-10-31 | New England Medical Center Hospitals, Inc. | Inhibitors of dipeptidyl-aminopeptidase type iv |
| DD296075A5 (en) | 1989-08-07 | 1991-11-21 | Martin-Luther-Universitaet Halle-Wittenberg,De | PROCESS FOR THE PREPARATION OF NEW INHIBITORS OF DIPEPTIDYL PEPTIDASE IV |
| WO1993008259A2 (en) | 1991-10-22 | 1993-04-29 | New England Medical Center Hospitals, Inc. | Inhibitors of dipeptidyl-aminopeptidase type iv |
| CA2123128A1 (en) | 1991-11-22 | 1993-05-27 | Roger Snow | Method for making a prolineboronate ester |
| WO1995015309A1 (en) | 1993-12-03 | 1995-06-08 | Ferring B.V. | Dp-iv-serine protease inhibitors |
| WO1995029691A1 (en) | 1994-04-28 | 1995-11-09 | Georgia Tech Research Corporation | Proline phosphonate derivatives |
| DE19616486A1 (en) | 1996-04-25 | 1997-10-30 | Knoell Hans Forschung Ev | Method of lowering blood glucose levels in mammals |
| JPH1081666A (en) | 1996-06-12 | 1998-03-31 | Ishihara Sangyo Kaisha Ltd | Phthalimide derivative or salt thereof, its production and medicinal composition containing the same |
| WO1998018763A1 (en) | 1996-10-25 | 1998-05-07 | Tanabe Seiyaku Co., Ltd. | Tetrahydroisoquinoline derivatives |
| WO1998019998A2 (en) | 1996-11-07 | 1998-05-14 | Novartis Ag | N-substituted 2-cyanopyrrolidines |
| JPH10182613A (en) | 1996-10-25 | 1998-07-07 | Tanabe Seiyaku Co Ltd | Tetraphydroisoquinoline derivative |
| CA2289125A1 (en) | 1997-05-07 | 1998-11-12 | Trustees Of Tufts College | Potentiation of the immune response through delivery of compounds binding a cytoplasmic dipeptidase |
| WO1998050046A1 (en) | 1997-05-07 | 1998-11-12 | Trustees Of Tufts College | Use of cd26 inhibitor for the manufacture of a medicament for the treatment of hiv |
| WO1999016864A1 (en) | 1997-09-29 | 1999-04-08 | Point Therapeutics, Inc. | Stimulation of hematopoietic cells in vitro |
| WO1999025719A1 (en) | 1997-11-18 | 1999-05-27 | Zaidan Hojin Biseibutsu Kagaku Kenkyu Kai | Novel physiologically active substance sulphostin, process for producing the same, and use thereof |
| WO1999056753A1 (en) | 1998-05-04 | 1999-11-11 | Point Therapeutics, Inc. | Hematopoietic stimulation |
| DE19823831A1 (en) | 1998-05-28 | 1999-12-02 | Probiodrug Ges Fuer Arzneim | New pharmaceutical use of isoleucyl thiazolidide and its salts |
| WO1999062914A1 (en) | 1998-06-05 | 1999-12-09 | Point Therapeutics, Inc. | Cyclic boroproline compounds |
| WO1999067278A1 (en) | 1998-06-24 | 1999-12-29 | Probiodrug Gesellschaft für Arzneimittelforschung mbH | Prodrugs of dipeptidyl peptidase iv inhibitors |
| DE19834591A1 (en) | 1998-07-31 | 2000-02-03 | Probiodrug Ges Fuer Arzneim | Use of substances that decrease the activity of dipeptidyl peptidase IV to increase blood sugar levels, e.g. for treating hypoglycemia |
| CA2339537A1 (en) | 1998-08-21 | 2000-03-02 | Barbara Wallner | Regulation of substrate activity |
| WO2000023421A1 (en) | 1998-10-22 | 2000-04-27 | Idun Pharmaceuticals, Inc. | (SUBSTITUTED)ACYL DIPEPTIDYL INHIBITORS OF THE ICE/ced-3 FAMILY OF CYSTEINE PROTEASES |
| WO2000031258A2 (en) | 1998-11-20 | 2000-06-02 | Arena Pharmaceuticals, Inc. | Human orphan g protein-coupled receptors |
| WO2000034241A1 (en) | 1998-12-10 | 2000-06-15 | Novartis Ag | N-substituted 2-cyanopyrrolidines |
| JP2000191616A (en) | 1998-12-24 | 2000-07-11 | Senju Pharmaceut Co Ltd | New peptidylaldehyde derivative and medicine containing the same |
| WO2000056297A2 (en) | 1999-03-23 | 2000-09-28 | Ferring B.V. | Pharmaceutical compositions comprising dipeptidy peptidase iv inhibitors for the promotion of growth |
| WO2000056296A2 (en) | 1999-03-23 | 2000-09-28 | Ferring Bv | Compositions for improving fertility |
| WO2000069868A1 (en) | 1999-05-17 | 2000-11-23 | Zaidan Hojin Biseibutsu Kagaku Kenkyu Kai | Sulphostin analogues and processes for the preparation of sulphostin and analogues thereof |
| WO2000071135A1 (en) | 1999-05-25 | 2000-11-30 | Point Therapeutics, Inc. | Anti-tumor comprising boroproline compounds |
| WO2001034594A1 (en) | 1999-11-12 | 2001-05-17 | Guilford Pharmaceuticals, Inc. | Dipeptidyl peptidase iv inhibitors and methods of making and using dipeptidyl peptidase iv inhibitors |
| WO2001052825A2 (en) | 2000-01-21 | 2001-07-26 | Novartis Ag | Combinations comprising dipeptidylpeptidase-iv inhibitors and antidiabetic agents |
| WO2001055105A1 (en) | 2000-01-24 | 2001-08-02 | Novo Nordisk A/S | N-substituted 2-cyanopyroles and -pyrrolines which are inhibitors of the enzyme dpp-iv |
| WO2001068603A2 (en) | 2000-03-10 | 2001-09-20 | Bristol-Myers Squibb Co. | Cyclopropyl-fused pyrrolidine-based inhibitors of dipeptidyl iv, processes for their preparation, and their use |
| WO2001081337A1 (en) | 2000-04-26 | 2001-11-01 | Ferring B.V. | Inhibitors of dipeptidyl peptidase iv |
| WO2001081304A1 (en) | 2000-04-26 | 2001-11-01 | Ferring Bv | Inhibitors of dipeptidyl peptidase iv |
| WO2001096295A2 (en) | 2000-06-13 | 2001-12-20 | Novartis Ag | 2-cyanopyrrolidine derivatives and their use as medicaments |
| WO2001097808A1 (en) | 2000-06-19 | 2001-12-27 | Smithkline Beecham Plc | Combinations of depeptidyl peptidase iv inhibitors and other antidiabetic agents for the treatment of diabete mellitus |
| WO2002001427A1 (en) | 2000-06-29 | 2002-01-03 | Worldsmart Technology Pty Ltd | Protecting against impulse expenditure |
| WO2002002560A2 (en) | 2000-07-04 | 2002-01-10 | Novo Nordisk A/S | Purine-2,6-diones which are inhibitors of the enzyme dipeptidyl peptidase iv (dpp-iv) |
| US20020006899A1 (en) | 1998-10-06 | 2002-01-17 | Pospisilik Andrew J. | Use of dipeptidyl peptidase IV effectors for lowering blood pressure in mammals |
| WO2002014271A1 (en) | 2000-08-10 | 2002-02-21 | Mitsubishi Pharma Corporation | Proline derivatives and use thereof as drugs |
| WO2002030890A1 (en) | 2000-10-06 | 2002-04-18 | Tanabe Seiyaku Co., Ltd. | Nitrogenous five-membered ring compounds |
| US6380398B2 (en) | 2000-01-04 | 2002-04-30 | Novo Nordisk A/S | Therapeutically active and selective heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| WO2002034900A1 (en) | 2000-10-27 | 2002-05-02 | The University Of Sydney | Dipeptidyl peptidases |
| WO2002038541A1 (en) | 2000-11-10 | 2002-05-16 | Taisho Pharmaceutical Co., Ltd. | Cyanopyrrolidine derivatives |
| US6410508B1 (en) | 1998-10-07 | 2002-06-25 | Med College Georgia Res Inst | Glucose-dependent insulinotropic peptide for use as an osteotropic hormone |
| CA2433090A1 (en) | 2000-12-27 | 2002-07-04 | Kyowa Hakko Kogyo Co., Ltd. | Dipeptidyl peptidase iv inhibitor |
| WO2002055088A1 (en) | 2001-01-16 | 2002-07-18 | Nippon Kayaku Kabushiki Kaisha | Remedies for bone marrow suppression and infectious diseases and white blood cell count elevating agents |
| US6432969B1 (en) | 2000-06-13 | 2002-08-13 | Novartis Ag | N-(substituted glycyl)-2 cyanopyrrolidines, pharmaceutical compositions containing them and their use in inhibiting dipeptidyl peptidase-IV |
| WO2002062764A1 (en) | 2001-02-02 | 2002-08-15 | Takeda Chemical Industries, Ltd. | Fused heterocyclic compounds |
| WO2002068420A1 (en) | 2001-02-24 | 2002-09-06 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Xanthine derivative, production and use thereof as a medicament |
| JP2002265439A (en) | 2001-03-08 | 2002-09-18 | Mitsubishi Pharma Corp | Cyanopyrrolidine derivative and its use for medicine |
| EP1245568A1 (en) | 2001-03-28 | 2002-10-02 | Les Laboratoires Servier | Sulfonyl derivatives of amino acids and their use as inhibitors of dipeptidyl-peptidase IV (DPP IV) |
| WO2002076450A1 (en) | 2001-03-27 | 2002-10-03 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2002083109A1 (en) | 2001-04-11 | 2002-10-24 | Ferring Bv | Treatment of type 2 diabetes with inhibitors of dipeptidyl peptidase iv |
| WO2002083128A1 (en) | 2001-04-12 | 2002-10-24 | Bristol-Myers Squibb Company | 2,1-oxazoline and 1,2-pyrazoline-based inhibitors of dipeptidyl peptidase iv and method |
| EP1258476A1 (en) | 2001-05-15 | 2002-11-20 | Les Laboratoires Servier | Alpha-amino acid derivatives, method for their preparation and their use as dipeptidyl-peptidase IV inhibitors (DPP IV) |
| JP2002356471A (en) | 2000-10-06 | 2002-12-13 | Tanabe Seiyaku Co Ltd | Aliphatic nitrogen-containing five-membered ring compound |
| JP2002356472A (en) | 2000-10-06 | 2002-12-13 | Tanabe Seiyaku Co Ltd | Nitrogen-containing five-membered ring compound |
| WO2003000180A2 (en) | 2001-06-20 | 2003-01-03 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| WO2003000250A1 (en) | 2001-06-25 | 2003-01-03 | Ferring Bv | 3-fluoro-pyrrolidines as antidiabetic agents |
| WO2003000181A2 (en) | 2001-06-20 | 2003-01-03 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| WO2003002593A2 (en) | 2001-06-27 | 2003-01-09 | Probiodrug Ag | Peptide structures useful for competitive modulation of dipeptidyl peptidase iv catalysis |
| WO2003002596A2 (en) | 2001-06-27 | 2003-01-09 | Probiodrug Ag | Use of dipeptidyl peptidase iv inhibitors as therapeutics for neurological disorders |
| WO2003002531A2 (en) | 2001-06-27 | 2003-01-09 | Smithkline Beecham Corporation | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| WO2003002530A2 (en) | 2001-06-27 | 2003-01-09 | Smithkline Beecham Corporation | Pyrrolidines as dipeptidyl peptidase inhibitors |
| WO2003002553A2 (en) | 2001-06-27 | 2003-01-09 | Smithkline Beecham Corporation | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| WO2003004496A1 (en) | 2001-07-03 | 2003-01-16 | Novo Nordisk A/S | Dpp-iv-inhibiting purine derivatives for the treatment of diabetes |
| WO2003004498A1 (en) | 2001-07-06 | 2003-01-16 | Merck & Co., Inc. | Beta-amino tetrahydroimidazo (1, 2-a) pyrazines and tetrahydrotrioazolo (4, 3-a) pyrazines as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2003022871A2 (en) | 2001-09-06 | 2003-03-20 | Probiodrug Ag | Peptides having a c- terminal hydroxylamino group as inhibitors of dipeptidyl peptidase i |
| WO2003024965A2 (en) | 2001-09-19 | 2003-03-27 | Novo Nordisk A/S | Heterocyclic compounds that are inhibitors of the enzyme dpp-iv |
| WO2003024942A1 (en) | 2001-09-14 | 2003-03-27 | Mitsubishi Pharma Corporation | Thiazolidine derivative and medicinal use thereof |
| WO2003035057A1 (en) | 2001-10-23 | 2003-05-01 | Ferring B.V. | Inhibitors of dipeptidyl peptidase iv |
| WO2003035067A1 (en) | 2001-10-23 | 2003-05-01 | Ferring B.V. | Novel dipeptidyl peptidase iv (dp-iv) inhibitors as anti-diabetic agents |
| WO2003037327A1 (en) | 2001-10-26 | 2003-05-08 | F. Hoffmann-La-Roche Ag | N-substituted pyrrolidin derivatives as dipeptidyl peptidase iv inhibitors |
| WO2003038123A2 (en) | 2001-10-31 | 2003-05-08 | Novartis Ag | Methods to treat diabetes and related conditions based on polymorphisms in the tcf1 gene |
| WO2003040174A2 (en) | 2001-11-09 | 2003-05-15 | Probiodrug Ag | Substituted amino ketone compounds |
| US20030105077A1 (en) | 2001-07-03 | 2003-06-05 | Kanstrup Anders Bendtz | Heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| CA2466870A1 (en) | 2001-11-26 | 2003-06-05 | Trustees Of Tufts College | Methods for treating autoimmune disorders, and reagents related thereto |
| US20030119738A1 (en) | 2001-09-06 | 2003-06-26 | Andre Niestroj | Novel inhibitors of dipeptidyl peptidase I |
| US20030119750A1 (en) | 2001-06-27 | 2003-06-26 | Hans-Ulrich Demuth | Use of dipeptidyl peptidase IV inhibitors |
| US20030125304A1 (en) | 2001-11-09 | 2003-07-03 | Hans-Ulrich Demuth | Substituted amino ketone compounds |
| US20030130199A1 (en) | 2001-06-27 | 2003-07-10 | Von Hoersten Stephan | Dipeptidyl peptidase IV inhibitors and their uses as anti-cancer agents |
| WO2003055881A1 (en) | 2001-12-27 | 2003-07-10 | F. Hoffmann-La Roche Ag | Pyrido(2,1-a)isoquinoline derivatives as dpp-iv inhibitors |
| WO2003057144A2 (en) | 2001-12-26 | 2003-07-17 | Guilford Pharmaceuticals | Change inhibitors of dipeptidyl peptidase iv |
| WO2003068748A1 (en) | 2002-02-13 | 2003-08-21 | F. Hoffmann-La Roche Ag | Novel pyridine- and quinoline-derivatives |
| WO2003068757A1 (en) | 2002-02-13 | 2003-08-21 | F. Hoffmann-La Roche Ag | Novel pyridin- and pyrimidin-derivatives |
| EP1338592A1 (en) | 2002-02-22 | 2003-08-27 | Nippon Zoki Pharmaceutical Co., Ltd. | Novel 2-phenylpiperazine derivatives |
| JP2003238566A (en) | 2001-02-02 | 2003-08-27 | Takeda Chem Ind Ltd | Condensed heterocyclic compound |
| EP1338651A1 (en) | 2000-12-01 | 2003-08-27 | Yamanouchi Pharmaceutical Co. Ltd. | Method of screening remedy for diabetes |
| US20030162820A1 (en) | 2002-02-28 | 2003-08-28 | Hans-Ulrich Demuth | Glutaminyl based DPIV inhibitors |
| WO2003072528A2 (en) | 2002-02-08 | 2003-09-04 | Idun Pharmaceuticals, Inc. | (substituted)acyl dipeptidyl inhibitors of the ice/ced-3 family of cysteine proteases |
| US6617340B1 (en) | 1999-07-29 | 2003-09-09 | Novartis Ag | N-(substituted glycyl)-pyrrolidines, pharmaceutical compositions containing them and their use in inhibiting dipeptidyl peptidase-IV |
| WO2003074500A2 (en) | 2002-03-06 | 2003-09-12 | Sanofi-Aventis | N-aminoacetyl-pyrrolidine-2-carbonitriles and their use as ddp-iv inhibitors |
| WO2003080633A1 (en) | 2002-03-25 | 2003-10-02 | Nippon Kayaku Kabushiki Kaisha | Novel $g(a)-amino-n-(diaminophosphinyl)lactam derivative |
| WO2003082817A2 (en) | 2002-03-25 | 2003-10-09 | Merck & Co., Inc. | Beta-amino heterocyclic dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2003084940A1 (en) | 2002-04-08 | 2003-10-16 | Alangudi Sankaranarayanan | Thiazolidine-4-carbonitriles and analogues and their use as dipeptidyl-peptidas inhibitors |
| JP2003300977A (en) | 2002-04-10 | 2003-10-21 | Sumitomo Pharmaceut Co Ltd | Xanthine derivative |
| JP2003327532A (en) | 2002-05-10 | 2003-11-19 | Takeda Chem Ind Ltd | Peptidase inhibitor |
| WO2003095425A1 (en) | 2002-05-09 | 2003-11-20 | Taisho Pharmaceutical Co.,Ltd. | Cyanopyrrolidine derivatives |
| WO2003099279A1 (en) | 2002-05-29 | 2003-12-04 | Novartis Ag | Combination of a dpp iv inhibitor and a cardiovascular compound |
| WO2003101448A1 (en) | 2002-06-03 | 2003-12-11 | Novartis Ag | The use of substituted cyanopyrrolidines and combination preparations containing them for treating hyperlipidemia and associated diseases |
| WO2003101958A2 (en) | 2002-06-04 | 2003-12-11 | Pfizer Products Inc. | Flourinated cyclic amides as dipeptidyl peptidase iv inhibitors |
| WO2003104229A1 (en) | 2002-06-06 | 2003-12-18 | エーザイ株式会社 | Novel fused imidazole derivative |
| WO2003106456A2 (en) | 2002-06-14 | 2003-12-24 | Sanofi-Synthelabo | New compounds |
| WO2003105763A2 (en) | 2002-06-14 | 2003-12-24 | Amylin Pharmaceuticals, Inc. | Prevention and/or treatment of inflammatory bowel disease using pyy or agonists thereof |
| WO2004000327A1 (en) | 2002-06-24 | 2003-12-31 | Zaidan Hojin Biseibutsu Kagaku Kenkyu Kai | Therapeutic agent for type 2 diabetes |
| JP2004002367A (en) | 2002-04-04 | 2004-01-08 | Tanabe Seiyaku Co Ltd | Pharmaceutical composition |
| JP2004002368A (en) | 2002-04-04 | 2004-01-08 | Tanabe Seiyaku Co Ltd | Pharmaceutical composition |
| WO2004004661A2 (en) | 2002-07-09 | 2004-01-15 | Point Therapeutics, Inc. | Boroproline compound combination therapy |
| WO2004007468A1 (en) | 2002-07-15 | 2004-01-22 | Merck & Co., Inc. | Piperidino pyrimidine dipeptidyl peptidase inhibitors for the treatment of diabetes |
| WO2004007446A1 (en) | 2002-07-10 | 2004-01-22 | Yamanouchi Pharmaceutical Co., Ltd. | Novel azetidine derivative or salt thereof |
| WO2004007517A1 (en) | 2002-07-11 | 2004-01-22 | Aventis Pharma Deutschland Gmbh | Novel thiophenylglycoside derivatives, methods for production thereof, medicaments comprising said compounds and use thereof |
| JP2004026820A (en) | 2002-05-09 | 2004-01-29 | Taisho Pharmaceut Co Ltd | Dipeptidyl peptidase iv inhibitor |
| WO2004009544A1 (en) | 2002-07-23 | 2004-01-29 | Yamanouchi Pharmaceutical Co., Ltd. | 2-cyano-4-fluoropyrrolidine derivative or its salt |
| JP2004035574A (en) | 2000-10-06 | 2004-02-05 | Tanabe Seiyaku Co Ltd | Aliphatic nitrogenous five-membered ring compound |
| JP2004043429A (en) | 2002-02-25 | 2004-02-12 | Eisai Co Ltd | New xanthine derivative and dppiv inhibitor |
| WO2004014860A2 (en) | 2002-08-08 | 2004-02-19 | Takeda Pharmaceutical Company Limited | Fused heterocyclic compounds as peptidase inhibitors |
| DE10238477A1 (en) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New purine derivatives, their production and their use as medicines |
| WO2004018468A2 (en) | 2002-08-21 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the production thereof and the use of the same as medicaments |
| DE10238243A1 (en) | 2002-08-21 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New 8-(3-amino-piperidin-1-yl)-xanthine derivatives are dipeptidylpeptidase-IV inhibitors useful for, e.g. treating diabetes mellitus, arthritis or obesity |
| DE10238470A1 (en) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New xanthine derivatives, their production and their use as medicines |
| WO2004020407A1 (en) | 2002-08-29 | 2004-03-11 | Taisho Pharmaceutical Co.,Ltd. | Benzenesulfonate of 4-fluoro-2-cyanopyrrolidine derivative |
| US6710040B1 (en) | 2002-06-04 | 2004-03-23 | Pfizer Inc. | Fluorinated cyclic amides as dipeptidyl peptidase IV inhibitors |
| WO2004026822A2 (en) | 2002-09-19 | 2004-04-01 | Abbott Laboratories | Pharmaceutical compositions as inhibitors of dipeptidyl peptidase-iv (dpp-iv) |
| WO2004033455A2 (en) | 2002-10-08 | 2004-04-22 | Novo Nordisk A/S | Hemisuccinate salts of heterocyclic dpp-iv inhibitors |
| WO2004032836A2 (en) | 2002-10-07 | 2004-04-22 | Merck & Co., Inc. | Antidiabetic beta-amino heterocylcic dipeptidyl peptidase inhibitors |
| US20040082570A1 (en) | 2002-02-25 | 2004-04-29 | Eisai Co., Ltd. | Xanthine derivative and DPPIV inhibitor |
| WO2004037169A2 (en) | 2002-10-18 | 2004-05-06 | Merck & Co., Inc. | Beta-amino heterocyclic dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004037181A2 (en) | 2002-10-23 | 2004-05-06 | Bristol-Myers Squibb Company | Glycinenitrile-based inhibitors of dipeptidyl peptidase iv and methods |
| DE10251927A1 (en) | 2002-11-08 | 2004-05-19 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New 1,7,8-trisubstituted xanthine derivatives, are dipeptidylpeptidase-IV inhibitors useful e.g. for treating diabetes mellitus type I or II, arthritis or obesity |
| US20040097510A1 (en) | 2002-08-21 | 2004-05-20 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| WO2004041795A1 (en) | 2002-10-30 | 2004-05-21 | Guilford Pharmaceuticals Inc. | Novel inhibitors of dipeptidyl peptidase iv |
| WO2004043940A1 (en) | 2002-11-07 | 2004-05-27 | Merck & Co., Inc. | Phenylalanine derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004046106A1 (en) | 2002-11-18 | 2004-06-03 | Pfizer Products Inc. | Dipeptidyl peptidase iv inhibiting fluorinated cyclic amides |
| WO2004048379A1 (en) | 2002-11-01 | 2004-06-10 | Sumitomo Pharmaceuticals Co., Ltd. | Xanthine compound |
| WO2004050658A1 (en) | 2002-12-03 | 2004-06-17 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel substituted imidazo-pyridinones and imidazo-pyridazeiones, the production and use thereof as medicaments |
| WO2004050022A2 (en) | 2002-12-04 | 2004-06-17 | Merck & Co., Inc. | Phenylalanine derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004052362A1 (en) | 2002-12-10 | 2004-06-24 | Novartis Ag | Combination of an dpp-iv inhibitor and a ppar-alpha compound |
| DE10256264A1 (en) | 2002-12-03 | 2004-06-24 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New trisubstituted dihydro-imidazo-pyridazinone or imidazo-pyridinone derivatives, useful as dipeptidylpeptidase-IV inhibitors for e.g. treating diabetes mellitus type I or II, arthritis or obesity |
| WO2004052850A2 (en) | 2002-12-09 | 2004-06-24 | Bristol-Myers Squibb Company | Methods and compounds producing dipeptidyl peptidase iv inhibitors and intermediates thereof |
| WO2004058266A1 (en) | 2002-12-20 | 2004-07-15 | Merck & Co., Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| US20040138214A1 (en) | 2002-11-08 | 2004-07-15 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Xanthine derivatives, the preparation thereof and their use as pharmaceutical compositions |
| WO2004065380A1 (en) | 2003-01-14 | 2004-08-05 | Arena Pharmaceuticals Inc. | 1,2,3-trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prpphylaxis and treatment of disorders related thereto such as diabetes and hyperglycemia |
| WO2004064778A2 (en) | 2003-01-17 | 2004-08-05 | Merck & Co. Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004067509A1 (en) | 2003-01-31 | 2004-08-12 | Sanwa Kagaku Kenkyusho Co., Ltd. | Compound inhibiting dipeptidyl peptidase iv |
| WO2004069162A2 (en) | 2003-01-31 | 2004-08-19 | Merck & Co., Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004071454A2 (en) | 2003-02-13 | 2004-08-26 | Guilford Pharmaceuticals Inc. | Substituted azetidine compounds as inhibitors of dipeptidyl peptidase iv |
| JP2004244412A (en) | 2003-01-20 | 2004-09-02 | Kotobuki Seiyaku Kk | 2-cyanopyrrolidine derivative having substituent at 4-position, method for producing the same, and medicament containing the same |
| WO2004076434A1 (en) | 2003-02-28 | 2004-09-10 | Aic | Dipeptidyl peptidase inhibitors |
| WO2004076413A2 (en) | 2003-02-24 | 2004-09-10 | Arena Pharmaceuticals, Inc. | Phenyl- and pyridylpiperidine-derivatives as modulators of glucose metabolism |
| WO2004076433A1 (en) | 2003-02-28 | 2004-09-10 | Aic | Dipeptidyl peptidase inhibitors |
| WO2004080990A1 (en) | 2003-03-14 | 2004-09-23 | Astellas Pharma Inc. | C-glycoside derivatives and salts thereof |
| WO2004085378A1 (en) | 2003-03-19 | 2004-10-07 | Merck & Co. Inc. | Process for the preparation of chiral beta amino acid derivatives by asymmetric hydrogenation |
| WO2004085661A2 (en) | 2003-03-24 | 2004-10-07 | Merck & Co., Inc | Process to chiral beta-amino acid derivatives |
| US6803357B1 (en) | 1998-02-02 | 2004-10-12 | New England Medical Center Hospitals, Inc. | Method of regulating glucose metabolism, and reagents related thereto |
| WO2004087053A2 (en) | 2003-03-25 | 2004-10-14 | Syrrx, Inc. | Dipeptidyl peptidase inhibitors |
| WO2004087650A2 (en) | 2003-03-27 | 2004-10-14 | Merck & Co. Inc. | Process and intermediates for the preparation of beta-amino acid amide dipeptidyl peptidase-iv inhibitors |
| EP1469873A2 (en) | 2001-11-26 | 2004-10-27 | Trustees of Tufts College | Peptidomimetic inhibitors of post-proline cleaving enzymes |
| WO2004092128A1 (en) | 2003-04-10 | 2004-10-28 | Smithkline Beecham Corporation | Anhydrous crystalline forms of (2s, 4s)-1-{(2r)-2-amino-3-‘4-methoxybenzyl)sulfonyl!-3-methylbutanoyl}-4-fluoropyrrolindine-2-carbonitrile |
| US6812350B2 (en) | 2002-06-04 | 2004-11-02 | Pfizer Inc. | Synthesis of 3,3,4,4-tetrafluoropyrrolidine and novel dipeptidyl peptidase-IV inhibitor compounds |
| WO2004096806A1 (en) | 2003-04-30 | 2004-11-11 | Sumitomo Pharmaceuticals Co. Ltd. | Fused imidazole derivative |
| JP2004315496A (en) | 2002-08-08 | 2004-11-11 | Takeda Chem Ind Ltd | Fused heterocyclic ring compound |
| WO2004099134A2 (en) | 2003-05-05 | 2004-11-18 | Prosidion Ltd. | Glutaminyl based dp iv-inhibitors |
| WO2004104215A2 (en) | 2003-05-21 | 2004-12-02 | Bayer Healthcare Ag | Diagnostics and therapeutics for diseases associated with dipeptidylpeptidase 7 (dpp7) |
| WO2004103993A1 (en) | 2003-05-14 | 2004-12-02 | Syrrx, Inc. | Dipeptidyl peptidase inhibitors |
| WO2004104216A2 (en) | 2003-05-21 | 2004-12-02 | Bayer Healthcare Ag | Diagnostics and therapeutics for diseases associated with dipeptidylpeptidase iv (dpp4) |
| WO2004103276A2 (en) | 2003-05-14 | 2004-12-02 | Merck & Co., Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004108730A1 (en) | 2003-06-05 | 2004-12-16 | Fujisawa Pharmaceutical Co., Ltd. | 1h-imidazo`4,5-d!pyridazines as dpp-iv inhibitors for the treatment of niddm |
| US20040259903A1 (en) | 2003-06-20 | 2004-12-23 | Markus Boehringer | Pyrido [2,1-a] isoquinoline derivatives |
| WO2004110375A2 (en) | 2003-06-06 | 2004-12-23 | Merck & Co., Inc. | Combination therapy for the treatment of diabetes |
| US20040259902A1 (en) | 2003-06-20 | 2004-12-23 | Markus Boehringer | Pyrido [2,1-a] isoquinoline derivatives |
| WO2004111041A1 (en) | 2003-06-12 | 2004-12-23 | Fujisawa Pharmaceutical Co., Ltd. | Pyrrolidine, thiazolidine and oxazolidine compounds which inhibit dipeptidyl peptidase-iv (dpp) |
| WO2004111051A1 (en) | 2003-06-18 | 2004-12-23 | Boehringer Ingelheim International Gmbh | Imidazo-pyridazinone derivatives and imidazo-pyridone derivatives, production thereof, and use thereof as medicaments |
| WO2004110436A1 (en) | 2003-06-06 | 2004-12-23 | Merck & Co., Inc. | Fused indoles as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004112701A2 (en) | 2003-06-17 | 2004-12-29 | Merck & Co., Inc. | Cyclohexylglycine derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2005003135A1 (en) | 2003-06-24 | 2005-01-13 | Merck & Co., Inc. | Phosphoric acid salt of a dipeptidyl peptidase-iv inhibitor |
| JP2005023038A (en) | 2003-07-04 | 2005-01-27 | Taisho Pharmaceut Co Ltd | Therapeutic agent for chronic renal disease |
| WO2005007647A1 (en) * | 2003-07-11 | 2005-01-27 | Arena Pharmaceuticals, Inc. | Trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto |
| WO2005009956A1 (en) | 2003-07-21 | 2005-02-03 | Smithkline Beecham Corporation | (2s,4s)-4-fluoro-1-[4-fluoro-beta-(4-fluorophenyl)-l-phenylalanyl]-2-pyrrolidinecarbonitrile p-toluenesulfonic acid salt and anhydrous crystalline forms thereof |
| WO2005011581A2 (en) | 2003-07-31 | 2005-02-10 | Merck & Co., Inc. | Hexahydrodiazepinones as dipeptidyl peptidase-iv inhibitors for the treatment or prevention of diabetes |
| WO2005012312A1 (en) | 2003-07-25 | 2005-02-10 | Sanofi-Aventis Deutschland Gmbh | Novel cyano thiazolides, methods for the production thereof, and use thereof as a medicament |
| WO2005012249A2 (en) | 2003-08-01 | 2005-02-10 | Bristol-Myers Squibb Company | Adamantyglycine- based inhibitors of dipeptidyl peptidase iv for the treatment of diabetes |
| WO2005012308A1 (en) | 2003-07-25 | 2005-02-10 | Sanofi-Aventis Deutschland Gmbh | Novel cyanopyrrolidides, methods for the production thereof, and use of the same as medicaments |
| WO2005012326A1 (en) | 2003-08-01 | 2005-02-10 | Tanabe Seiyaku Co., Ltd. | Novel compounds having inhibitory activity against sodium-dependant transporter |
| US20050043292A1 (en) | 2003-08-20 | 2005-02-24 | Pfizer Inc | Fluorinated lysine derivatives as dipeptidyl peptidase IV inhibitors |
| WO2005020920A2 (en) | 2003-09-02 | 2005-03-10 | Merck & Co., Inc. | Novel crystalline forms of a phosphoric acid salt of a dipeptidyl peptidase-iv inhibitor |
| US20050059716A1 (en) | 2003-07-25 | 2005-03-17 | Aventis Pharma Deutschland Gmbh | Novel bicyclic cyanoheterocycles, process for their preparation and their use as medicaments |
| US20050059724A1 (en) | 2003-07-25 | 2005-03-17 | Aventis Pharma Deutschland Gmbh | Novel cyanopyrrolidides, process for their preparation and their use as medicaments |
| WO2005023762A1 (en) | 2003-09-04 | 2005-03-17 | Abbott Laboratories | Pyrrolidine-2-carbonitrile derivatives and their use as inhibitors of dipeptidyl peptidase-iv (dpp-iv) |
| WO2005026148A1 (en) | 2003-09-08 | 2005-03-24 | Takeda San Diego, Inc. | Dipeptidyl peptidase inhibitors |
| WO2005025554A2 (en) | 2003-09-09 | 2005-03-24 | Japan Tobacco Inc. | Dipeptidyl peptidase iv inhibitor |
| WO2005030127A2 (en) | 2003-09-23 | 2005-04-07 | Merck & Co., Inc. | Novel crystalline form of a phosphoric acid salt of a dipeptidyl peptidase-iv inhibitor |
| WO2005030751A2 (en) | 2003-09-08 | 2005-04-07 | Takeda Pharmaceutical Company Limited | Dipeptidyl peptidase inhibitors |
| WO2005033099A2 (en) | 2003-10-03 | 2005-04-14 | Glenmark Pharmaceuticals Ltd. | Novel dipeptidyl peptidase iv inhibitors; processes for their preparation and compositions thereof |
| WO2005032590A1 (en) | 2003-10-03 | 2005-04-14 | Takeda Pharmaceutical Company Limited | Remedy for diabetes |
| WO2005034940A2 (en) | 2003-10-15 | 2005-04-21 | Imtm Gmbh | Dual alanyl aminopeptidase and dipeptidyl peptidase iv inhibitors for functionally influencing different cells and for treating immunological, inflammatory, neuronal and other diseases |
| WO2005037828A1 (en) | 2003-10-20 | 2005-04-28 | Lg Life Sciences Ltd. | Novel inhibitors of dpp-iv, methods of preparing the same, and pharmaceutical compositions containing the same as an active agent |
| WO2005037779A2 (en) | 2003-10-15 | 2005-04-28 | Imtm Gmbh | Novel dipeptidyl peptidase iv inhibitors used for functionally influencing different cells and treating immunological, inflammatory, neuronal, and other diseases |
| WO2005040095A1 (en) | 2003-10-16 | 2005-05-06 | Astrazeneca Ab | Inhibitors of dipeptidyl peptidase iv |
| WO2005042488A1 (en) | 2003-10-31 | 2005-05-12 | Takeda Pharmaceutical Company Limited | Pyridine compounds as inhibitors of dipeptidyl peptidase iv |
| WO2005044195A2 (en) | 2003-11-04 | 2005-05-19 | Merck & Co., Inc. | Fused phenylalanine derivatives as dipeptidyl peptidase-iv inhibitors for the treatment or prevention of diabetes |
| WO2005047297A1 (en) | 2003-11-12 | 2005-05-26 | Phenomix Corporation | Heterocyclic boronic acid compounds |
| WO2005049022A2 (en) | 2003-11-17 | 2005-06-02 | Novartis Ag | Use of dipeptidyl peptidase iv inhibitors |
| WO2005058849A1 (en) | 2003-12-15 | 2005-06-30 | Glenmark Pharmaceuticals Ltd. | New dipeptidyl peptidase in inhibitors; process for their preparation and compositions containing them |
| WO2005063750A1 (en) | 2003-12-23 | 2005-07-14 | Boehringer Ingelheim International Gmbh | Bicyclic imidazole compounds, the production thereof and their use as medicaments |
| WO2005072530A1 (en) | 2004-01-16 | 2005-08-11 | Merck & Co., Inc. | Novel crystalline salts of a dipeptidyl peptidase-iv inhibitor |
| WO2005075426A1 (en) | 2004-02-03 | 2005-08-18 | Glenmark Pharmaceuticals Ltd. | Novel dipeptidyl peptidase iv inhibitors; processes for their preparation and compositions thereof |
| WO2005079795A2 (en) | 2004-02-20 | 2005-09-01 | Novartis Ag | Dpp-iv inhibitors for treating neurodegeneration and cognitive disorders |
| WO2005082348A2 (en) | 2004-02-23 | 2005-09-09 | Trustees Of Tufts College | Inhibitors of dipeptidylpeptidase iv for regulating glucose metabolism |
| WO2005082849A1 (en) | 2004-02-23 | 2005-09-09 | Trustees Of Tufts College | Lactams as conformationally constrained peptidomimetic inhibitors |
| WO2005087235A1 (en) | 2004-03-09 | 2005-09-22 | National Health Research Institutes | Pyrrolidine compounds |
| WO2005092877A1 (en) | 2004-03-16 | 2005-10-06 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzol derivatives, drugs containing said compounds, the use thereof and method for the production thereof |
| WO2005095381A1 (en) | 2004-03-15 | 2005-10-13 | Takeda Pharmaceutical Company Limited | Dipeptidyl peptidase inhibitors |
| WO2005116014A1 (en) | 2004-05-12 | 2005-12-08 | Pfizer Products Inc. | Proline derivatives and their use as dipeptidyl peptidase iv inhibitors |
| WO2006040625A1 (en) | 2004-10-12 | 2006-04-20 | Glenmark Pharmaceuticals S.A. | Novel dipeptidyl peptidase iv inhibitors, pharmaceutical compositions containing them, and process for their preparation |
| WO2006068163A1 (en) | 2004-12-24 | 2006-06-29 | Dainippon Sumitomo Pharma Co., Ltd. | Bicyclic pyrrole derivatives |
| WO2006076231A2 (en) | 2005-01-10 | 2006-07-20 | Arena Pharmaceuticals, Inc. | Combination therapy for the treatment of diabetes and conditions related thereto and for the treatment of conditions ameliorated by increasing a blood glp-1 level |
| WO2006080421A1 (en) | 2005-01-28 | 2006-08-03 | Chugai Seiyaku Kabushiki Kaisha | Spiroketal derivative and use thereof as diabetic medicine |
| WO2006088129A1 (en) | 2005-02-18 | 2006-08-24 | Mitsubishi Pharma Corporation | Salt of proline derivative, solvate thereof, and production method thereof |
| WO2006100181A2 (en) | 2005-03-22 | 2006-09-28 | F. Hoffmann-La Roche Ag | New salt and polymorphs of a dpp-iv inhibitor |
| WO2006116157A2 (en) | 2005-04-22 | 2006-11-02 | Alantos Pharmaceuticals Holding, Inc. | Dipeptidyl peptidase-iv inhibitors |
| WO2007014815A1 (en) | 2005-07-29 | 2007-02-08 | Trasmetal S.P.A. | Paint booth for plants for painting products |
| WO2007019255A2 (en) | 2005-08-04 | 2007-02-15 | Novartis Ag | Salts of vildagliptin |
| WO2007027651A2 (en) | 2005-08-30 | 2007-03-08 | Abbott Laboratories | Pharmaceutical compositions as inhibitors of dipeptidyl peptidase-iv (dpp-iv) |
| WO2007035372A2 (en) | 2005-09-16 | 2007-03-29 | Takeda Pharmaceutical Company Limited | Polymorphs of benzoate salt of 2-[[6-[(3r)-3-amino-1-piperidinyl]-3,4-dihydro-3-methyl-2,4-dioxo-1(2h)-pyrimidinyl]methyl]-benzonitrile and methods of use therefor |
| WO2007071576A1 (en) | 2005-12-21 | 2007-06-28 | F. Hoffmann-La Roche Ag | New salt and polymorph of dpp-iv inhibitor |
| US20070167468A1 (en) | 2004-08-06 | 2007-07-19 | Sanofi-Aventis Deutschland Gmbh | Substituted bicyclic 8-pyrr0lidinoxanthines, methods for their production, pharmaceutical formulations and their use |
| WO2007120689A2 (en) | 2006-04-11 | 2007-10-25 | Arena Pharmaceuticals, Inc. | Methods of using gpr119 receptor to identify compounds useful for increasing bone mass in an individual |
| WO2007120702A2 (en) | 2006-04-11 | 2007-10-25 | Arena Pharmaceuticals, Inc. | Use of gpr119 receptor agonists for increasing bone mass and for treating osteoporosis, and combination therapy relating thereto |
| WO2007128721A1 (en) | 2006-05-04 | 2007-11-15 | Boehringer Ingelheim Internationalgmbh | Polymorphs |
| WO2007148185A2 (en) | 2006-06-21 | 2007-12-27 | Pfizer Products Inc. | Substituted 3 -amino- pyrrolidino-4 -lactams as dpp inhibitors |
| WO2008027273A2 (en) | 2006-08-30 | 2008-03-06 | Phenomix Corporation | Solid citrate and tartrate salts of dpp-iv inhibitors |
| EP1902730A1 (en) | 2005-06-09 | 2008-03-26 | Banyu Pharmaceutical Co., Ltd. | Npy y2 agonist for use as therapeutic agent for disease accompanied by diarrhea |
| WO2008067465A1 (en) | 2006-11-29 | 2008-06-05 | Takeda Pharmaceutical Company Limited | Polymorphs of succinate salt of 2-[6-(3-amino-piperidin-1-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2h-pyrimidin-1-ylmethy]-4-fluor-benzonitrile and methods of use therefor |
| WO2008070692A2 (en) * | 2006-12-06 | 2008-06-12 | Smithkline Beecham Corporation | Bicyclic compounds and use as antidiabetics |
| US20080146818A1 (en) | 2004-02-05 | 2008-06-19 | Yasumichi Fukuda | Bicycloester Derivative |
| WO2008109591A1 (en) | 2007-03-08 | 2008-09-12 | Lexicon Pharmaceuticals, Inc. | Phlorizin analogs as inhibitors of sodium glucose co-transporter 2 |
| WO2008114857A1 (en) | 2007-03-22 | 2008-09-25 | Kyorin Pharmaceutical Co., Ltd. | Method for producing aminoacetylpyrrolidinecarbonitrile derivative |
| WO2009084497A1 (en) | 2007-12-28 | 2009-07-09 | Dainippon Sumitomo Pharma Co., Ltd. | Methyl-substituted piperidine derivative |
| WO2009126245A1 (en) | 2008-04-07 | 2009-10-15 | Arena Pharmaceuticals, Inc. | Methods of using a g protein-coupled receptor to identify peptide yy (pyy) secretagogues and compounds useful in the treatment of conditions modulated by pyy |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW438587B (en) * | 1995-06-20 | 2001-06-07 | Takeda Chemical Industries Ltd | A pharmaceutical composition for prophylaxis and treatment of diabetes |
| JP2004067575A (en) * | 2002-08-06 | 2004-03-04 | Yaizu Suisankagaku Industry Co Ltd | Promoter for effect of therapeutic agent for diabetes |
| UA86943C2 (en) * | 2003-07-11 | 2009-06-10 | Арена Фармасьютикалз, Инк. | Trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto |
| WO2008128832A1 (en) * | 2007-04-18 | 2008-10-30 | Nicox S.A. | Nitroderivatives of non-peptidic renin inhibitors for the treatment of cardiovascular, renal and chronic liver disease, inflammations and metabolic syndrome |
| WO2010074271A1 (en) * | 2008-12-26 | 2010-07-01 | 武田薬品工業株式会社 | Therapeutic agent for diabetes |
-
2011
- 2011-04-05 EP EP11715123A patent/EP2556056A1/en not_active Withdrawn
- 2011-04-05 BR BR112012025592A patent/BR112012025592A2/en not_active IP Right Cessation
- 2011-04-05 MX MX2012011631A patent/MX2012011631A/en not_active Application Discontinuation
- 2011-04-05 CA CA2795513A patent/CA2795513A1/en not_active Abandoned
- 2011-04-05 JP JP2013503841A patent/JP2013523819A/en not_active Withdrawn
- 2011-04-05 US US13/639,652 patent/US20130023494A1/en not_active Abandoned
- 2011-04-05 WO PCT/US2011/031243 patent/WO2011127051A1/en active Application Filing
- 2011-04-05 AU AU2011237775A patent/AU2011237775A1/en not_active Abandoned
- 2011-04-05 CN CN2011800275776A patent/CN102918027A/en active Pending
Patent Citations (410)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DD296075A5 (en) | 1989-08-07 | 1991-11-21 | Martin-Luther-Universitaet Halle-Wittenberg,De | PROCESS FOR THE PREPARATION OF NEW INHIBITORS OF DIPEPTIDYL PEPTIDASE IV |
| WO1991016339A1 (en) | 1990-04-14 | 1991-10-31 | New England Medical Center Hospitals, Inc. | Inhibitors of dipeptidyl-aminopeptidase type iv |
| EP0528858A1 (en) | 1990-04-14 | 1993-03-03 | New England Medical Center Inc | Inhibitors of dipeptidyl-aminopeptidase type iv. |
| JPH05508624A (en) | 1990-04-14 | 1993-12-02 | ニュー イングランド メデカル センター ホスピタルズ インク | Dipeptidyl-aminopeptidase type 4 inhibitor |
| WO1993008259A2 (en) | 1991-10-22 | 1993-04-29 | New England Medical Center Hospitals, Inc. | Inhibitors of dipeptidyl-aminopeptidase type iv |
| EP0610317A1 (en) | 1991-10-22 | 1994-08-17 | New England Medical Center | Inhibitors of dipeptidyl-aminopeptidase type iv |
| EP1050540A2 (en) | 1991-10-22 | 2000-11-08 | New England Medical Center Hospitals, Inc. | Inhibitors of dipeptidyl-aminopeptidase type IV |
| CA2123128A1 (en) | 1991-11-22 | 1993-05-27 | Roger Snow | Method for making a prolineboronate ester |
| WO1993010127A1 (en) | 1991-11-22 | 1993-05-27 | Boehringer Ingelheim Pharmaceuticals, Inc. | Method for making a prolineboronate ester |
| JPH07501078A (en) | 1991-11-22 | 1995-02-02 | ベーリンガー インゲルハイム ファーマシューティカルズ インコーポレイテッド | Production method of proline boronate ester |
| EP0641347A1 (en) | 1991-11-22 | 1995-03-08 | Boehringer Ingelheim Pharmaceuticals Inc. | Method for making a prolineboronate ester |
| EP0731789A1 (en) | 1993-12-03 | 1996-09-18 | Ferring B.V. | Dp-iv-serine protease inhibitors |
| JPH09509921A (en) | 1993-12-03 | 1997-10-07 | フェーリング ベスローテン フェンノートシャップ | Enzyme inhibitors |
| WO1995015309A1 (en) | 1993-12-03 | 1995-06-08 | Ferring B.V. | Dp-iv-serine protease inhibitors |
| WO1995029691A1 (en) | 1994-04-28 | 1995-11-09 | Georgia Tech Research Corporation | Proline phosphonate derivatives |
| DE19616486A1 (en) | 1996-04-25 | 1997-10-30 | Knoell Hans Forschung Ev | Method of lowering blood glucose levels in mammals |
| WO1997040832A1 (en) | 1996-04-25 | 1997-11-06 | Probiodrug Gesellschaft für Arzneimittelforschung mbH | Use of dipeptidyl peptidase iv effectors for lowering the blood glucose level in mammals |
| JP2001510442A (en) | 1996-04-25 | 2001-07-31 | プロビオドラッグ ゲゼルシャフト フュル アルツナイミッテルフォルシュング エムベーハー | Use of dipeptidyl peptidase (IV) effector for lowering blood glucose in mammals |
| US6303661B1 (en) | 1996-04-25 | 2001-10-16 | Probiodrug | Use of dipeptidyl peptidase IV effectors for lowering the blood glucose level in mammals |
| JPH1081666A (en) | 1996-06-12 | 1998-03-31 | Ishihara Sangyo Kaisha Ltd | Phthalimide derivative or salt thereof, its production and medicinal composition containing the same |
| WO1998018763A1 (en) | 1996-10-25 | 1998-05-07 | Tanabe Seiyaku Co., Ltd. | Tetrahydroisoquinoline derivatives |
| JPH10182613A (en) | 1996-10-25 | 1998-07-07 | Tanabe Seiyaku Co Ltd | Tetraphydroisoquinoline derivative |
| WO1998019998A2 (en) | 1996-11-07 | 1998-05-14 | Novartis Ag | N-substituted 2-cyanopyrrolidines |
| JP2000511559A (en) | 1996-11-07 | 2000-09-05 | ノバルティス アクチエンゲゼルシャフト | N-substituted 2-cyanopyrrolidine |
| WO1998050066A1 (en) | 1997-05-07 | 1998-11-12 | Trustees Of Tufts College | Potentiation of the immune response through delivery of compounds binding a cytoplasmic dipeptidase |
| EP0980249A1 (en) | 1997-05-07 | 2000-02-23 | Trustees Of Tufts College | Use of cd26 inhibitor for the manufacture of a medicament for the treatment of hiv |
| CA2289125A1 (en) | 1997-05-07 | 1998-11-12 | Trustees Of Tufts College | Potentiation of the immune response through delivery of compounds binding a cytoplasmic dipeptidase |
| WO1998050046A1 (en) | 1997-05-07 | 1998-11-12 | Trustees Of Tufts College | Use of cd26 inhibitor for the manufacture of a medicament for the treatment of hiv |
| US6100234A (en) | 1997-05-07 | 2000-08-08 | Tufts University | Treatment of HIV |
| CA2289124A1 (en) | 1997-05-07 | 1998-11-12 | Trustees Of Tufts College | Use of cd26 inhibitor for the manufacture of a medicament for the treatment of hiv |
| US6040145A (en) | 1997-05-07 | 2000-03-21 | Tufts University | Potentiation of the immune response |
| EP0975359A1 (en) | 1997-05-07 | 2000-02-02 | Trustees Of Tufts College | Potentiation of the immune response through delivery of compounds binding a cytoplasmic dipeptidase |
| WO1999016864A1 (en) | 1997-09-29 | 1999-04-08 | Point Therapeutics, Inc. | Stimulation of hematopoietic cells in vitro |
| EP1043328A1 (en) | 1997-11-18 | 2000-10-11 | Zaidan Hojin Biseibutsu Kagaku Kenkyu Kai | Novel physiologically active substance sulphostin, process for producing the same, and use thereof |
| WO1999025719A1 (en) | 1997-11-18 | 1999-05-27 | Zaidan Hojin Biseibutsu Kagaku Kenkyu Kai | Novel physiologically active substance sulphostin, process for producing the same, and use thereof |
| US6803357B1 (en) | 1998-02-02 | 2004-10-12 | New England Medical Center Hospitals, Inc. | Method of regulating glucose metabolism, and reagents related thereto |
| WO1999056753A1 (en) | 1998-05-04 | 1999-11-11 | Point Therapeutics, Inc. | Hematopoietic stimulation |
| EP1215207A2 (en) | 1998-05-28 | 2002-06-19 | Probiodrug AG | Salts of isoleucyl-thiazolidine and -pyrrolidine and their use as dipeptidylpeptidase inhibitors |
| US20030134802A1 (en) | 1998-05-28 | 2003-07-17 | Hans-Ulrich Demuth | Novel effectors of dipepetidyl peptidase IV |
| EP1304327A2 (en) | 1998-05-28 | 2003-04-23 | Probiodrug AG | Glutamine-thiazolidide and -pyrrolidide and their use as dipeptidylpeptidase IV inhibitors |
| JP2002516318A (en) | 1998-05-28 | 2002-06-04 | プロバイオドラッグ ゲゼルシャフト フュア アルツナイミッテルフォルシュンク エムベーハー | Novel effector of dipeptidyl peptidase IV |
| EP1082314A1 (en) | 1998-05-28 | 2001-03-14 | Probiodrug Gesellschaft für Arzneimittelforschung mbH | New dipeptidyl peptidase iv effectors |
| WO1999061431A1 (en) | 1998-05-28 | 1999-12-02 | Probiodrug Gesellschaft für Arzneimittelforschung mbH | New dipeptidyl peptidase iv effectors |
| DE19823831A1 (en) | 1998-05-28 | 1999-12-02 | Probiodrug Ges Fuer Arzneim | New pharmaceutical use of isoleucyl thiazolidide and its salts |
| JP2002517401A (en) | 1998-06-05 | 2002-06-18 | ポイント セラピューティクス, インコーポレイテッド | Cyclic boroproline compound |
| WO1999062914A1 (en) | 1998-06-05 | 1999-12-09 | Point Therapeutics, Inc. | Cyclic boroproline compounds |
| JP2003524591A (en) | 1998-06-24 | 2003-08-19 | プロバイオドラッグ ゲゼルシャフト フュア アルツナイミッテルフォルシュンク エムベーハー | Prodrugs of DPIV inhibitors |
| WO1999067278A1 (en) | 1998-06-24 | 1999-12-29 | Probiodrug Gesellschaft für Arzneimittelforschung mbH | Prodrugs of dipeptidyl peptidase iv inhibitors |
| US20020049164A1 (en) | 1998-06-24 | 2002-04-25 | Hans-Ulrich Demuth | Prodrugs of DP IV-inhibitors |
| DE19828113A1 (en) | 1998-06-24 | 2000-01-05 | Probiodrug Ges Fuer Arzneim | Prodrugs of Dipeptidyl Peptidase IV Inhibitors |
| DE19834591A1 (en) | 1998-07-31 | 2000-02-03 | Probiodrug Ges Fuer Arzneim | Use of substances that decrease the activity of dipeptidyl peptidase IV to increase blood sugar levels, e.g. for treating hypoglycemia |
| EP0995440A1 (en) | 1998-07-31 | 2000-04-26 | Probiodrug Gesellschaft für Arzneimittelforschung mbH | Method to increase the blood, glucose level in mammals |
| WO2000010549A1 (en) | 1998-08-21 | 2000-03-02 | Point Therapeutics, Inc. | Regulation of substrate activity |
| CA2339537A1 (en) | 1998-08-21 | 2000-03-02 | Barbara Wallner | Regulation of substrate activity |
| EP1104293A1 (en) | 1998-08-21 | 2001-06-06 | Point Therapeutics, Inc. | Regulation of substrate activity |
| US20020006899A1 (en) | 1998-10-06 | 2002-01-17 | Pospisilik Andrew J. | Use of dipeptidyl peptidase IV effectors for lowering blood pressure in mammals |
| US6410508B1 (en) | 1998-10-07 | 2002-06-25 | Med College Georgia Res Inst | Glucose-dependent insulinotropic peptide for use as an osteotropic hormone |
| US6242422B1 (en) | 1998-10-22 | 2001-06-05 | Idun Pharmacueticals, Inc. | (Substituted)Acyl dipeptidyl inhibitors of the ice/ced-3 family of cysteine proteases |
| EP1123272A1 (en) | 1998-10-22 | 2001-08-16 | Idun Pharmaceuticals, Inc. | (substituted)acyl dipeptidyl inhibitors of the ice/ced-3 family of cysteine proteases |
| WO2000023421A1 (en) | 1998-10-22 | 2000-04-27 | Idun Pharmaceuticals, Inc. | (SUBSTITUTED)ACYL DIPEPTIDYL INHIBITORS OF THE ICE/ced-3 FAMILY OF CYSTEINE PROTEASES |
| JP2002527504A (en) | 1998-10-22 | 2002-08-27 | アイドゥン ファーマシューティカルズ, インコーポレイテッド | ICE / ced-3 substituted acyldipeptidyl inhibitors of cysteine proteases |
| WO2000031258A2 (en) | 1998-11-20 | 2000-06-02 | Arena Pharmaceuticals, Inc. | Human orphan g protein-coupled receptors |
| JP2002531547A (en) | 1998-12-10 | 2002-09-24 | ノバルティス アクチエンゲゼルシャフト | N-substituted 2-cyanopyrrolidine |
| WO2000034241A1 (en) | 1998-12-10 | 2000-06-15 | Novartis Ag | N-substituted 2-cyanopyrrolidines |
| EP1137635A1 (en) | 1998-12-10 | 2001-10-04 | Novartis AG | N-substituted 2-cyanopyrrolidines |
| US6166063A (en) | 1998-12-10 | 2000-12-26 | Novartis Ag | N-(substituted glycyl)-2-cyanopyrrolidines, pharmaceutical compositions containing them and their use in inhibiting dipeptidyl peptidase-IV |
| JP2000191616A (en) | 1998-12-24 | 2000-07-11 | Senju Pharmaceut Co Ltd | New peptidylaldehyde derivative and medicine containing the same |
| WO2000056296A2 (en) | 1999-03-23 | 2000-09-28 | Ferring Bv | Compositions for improving fertility |
| WO2000056297A2 (en) | 1999-03-23 | 2000-09-28 | Ferring B.V. | Pharmaceutical compositions comprising dipeptidy peptidase iv inhibitors for the promotion of growth |
| JP2000327689A (en) | 1999-05-17 | 2000-11-28 | Microbial Chem Res Found | Sulfostin analogue, and production of sulfostin and its analogue |
| WO2000069868A1 (en) | 1999-05-17 | 2000-11-23 | Zaidan Hojin Biseibutsu Kagaku Kenkyu Kai | Sulphostin analogues and processes for the preparation of sulphostin and analogues thereof |
| WO2000071135A1 (en) | 1999-05-25 | 2000-11-30 | Point Therapeutics, Inc. | Anti-tumor comprising boroproline compounds |
| US6617340B1 (en) | 1999-07-29 | 2003-09-09 | Novartis Ag | N-(substituted glycyl)-pyrrolidines, pharmaceutical compositions containing them and their use in inhibiting dipeptidyl peptidase-IV |
| WO2001034594A1 (en) | 1999-11-12 | 2001-05-17 | Guilford Pharmaceuticals, Inc. | Dipeptidyl peptidase iv inhibitors and methods of making and using dipeptidyl peptidase iv inhibitors |
| EP1228061A1 (en) | 1999-11-12 | 2002-08-07 | Guilford Pharmaceuticals Inc. | Dipeptidyl peptidase iv inhibitors and methods of making and using dipeptidyl peptidase iv inhibitors |
| JP2003535034A (en) | 1999-11-12 | 2003-11-25 | ギルフォード ファーマシューティカルズ インコーポレイテッド | Dipeptidyl peptidase IV inhibitors and methods for producing and using dipeptidyl peptidase IV inhibitors |
| US6380398B2 (en) | 2000-01-04 | 2002-04-30 | Novo Nordisk A/S | Therapeutically active and selective heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| WO2001052825A2 (en) | 2000-01-21 | 2001-07-26 | Novartis Ag | Combinations comprising dipeptidylpeptidase-iv inhibitors and antidiabetic agents |
| EP1248604A2 (en) | 2000-01-21 | 2002-10-16 | Novartis AG | Combinations comprising dipeptidylpeptidase-iv inhibitors and antidiabetic agents |
| US20020103384A1 (en) | 2000-01-24 | 2002-08-01 | Anders Kanstrup | Therapeutically active and selective heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| EP1254113A1 (en) | 2000-01-24 | 2002-11-06 | Novo Nordisk A/S | N-substituted 2-cyanopyroles and -pyrrolines which are inhibitors of the enzyme dpp-iv |
| US6645995B2 (en) | 2000-01-24 | 2003-11-11 | Novo Nordisk A/S | Therapeutically active and selective heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| JP2003520849A (en) | 2000-01-24 | 2003-07-08 | ノボ ノルディスク アクティーゼルスカブ | N-substituted 2-cyanopyrroles and -pyrrolines which are inhibitors of the enzyme DPP-IV |
| WO2001055105A1 (en) | 2000-01-24 | 2001-08-02 | Novo Nordisk A/S | N-substituted 2-cyanopyroles and -pyrrolines which are inhibitors of the enzyme dpp-iv |
| US6395767B2 (en) | 2000-03-10 | 2002-05-28 | Bristol-Myers Squibb Company | Cyclopropyl-fused pyrrolidine-based inhibitors of dipeptidyl peptidase IV and method |
| JP2003531118A (en) | 2000-03-10 | 2003-10-21 | ブリストル−マイヤーズ スクイブ カンパニー | Inhibitors and methods for dipeptidyl peptidase IV having a cyclopropyl-fused pyrrolidine skeleton |
| WO2001068603A2 (en) | 2000-03-10 | 2001-09-20 | Bristol-Myers Squibb Co. | Cyclopropyl-fused pyrrolidine-based inhibitors of dipeptidyl iv, processes for their preparation, and their use |
| EP1261586A2 (en) | 2000-03-10 | 2002-12-04 | Bristol-Myers Squibb Company | Cyclopropyl-fused pyrrolidine-based inhibitors of dipeptidyl peptidase iv, processes for their preparation, and their use |
| US20030216450A1 (en) | 2000-04-26 | 2003-11-20 | Evans David Michael | Inhibitors of dipeptidyl peptidase IV |
| WO2001081337A1 (en) | 2000-04-26 | 2001-11-01 | Ferring B.V. | Inhibitors of dipeptidyl peptidase iv |
| JP2003531204A (en) | 2000-04-26 | 2003-10-21 | フェリング ベスローテン フェンノートシャップ | Inhibitors of dipeptidyl peptidase IV |
| EP1282600A1 (en) | 2000-04-26 | 2003-02-12 | Ferring BV | Inhibitors of dipeptidyl peptidase iv |
| WO2001081304A1 (en) | 2000-04-26 | 2001-11-01 | Ferring Bv | Inhibitors of dipeptidyl peptidase iv |
| EP1280797A1 (en) | 2000-04-26 | 2003-02-05 | Ferring B.V. | Inhibitors of dipeptidyl peptidase iv |
| JP2003531191A (en) | 2000-04-26 | 2003-10-21 | フェリング ベスローテン フェンノートシャップ | Inhibitors of dipeptidyl peptidase IV |
| US6432969B1 (en) | 2000-06-13 | 2002-08-13 | Novartis Ag | N-(substituted glycyl)-2 cyanopyrrolidines, pharmaceutical compositions containing them and their use in inhibiting dipeptidyl peptidase-IV |
| JP2004503531A (en) | 2000-06-13 | 2004-02-05 | ノバルティス アクチエンゲゼルシャフト | 2-Cyanopyrrolidine derivatives and their use as drugs |
| EP1296974A2 (en) | 2000-06-13 | 2003-04-02 | Novartis AG | 2-cyanopyrrolidine derivatives and their use as medicaments |
| WO2001096295A2 (en) | 2000-06-13 | 2001-12-20 | Novartis Ag | 2-cyanopyrrolidine derivatives and their use as medicaments |
| WO2001097808A1 (en) | 2000-06-19 | 2001-12-27 | Smithkline Beecham Plc | Combinations of depeptidyl peptidase iv inhibitors and other antidiabetic agents for the treatment of diabete mellitus |
| JP2003535898A (en) | 2000-06-19 | 2003-12-02 | スミスクライン ビーチャム パブリック リミテッド カンパニー | Combination of dipeptidyl peptidase IV inhibitors and other antidiabetic agents for the treatment of diabetes mellitus |
| WO2002001427A1 (en) | 2000-06-29 | 2002-01-03 | Worldsmart Technology Pty Ltd | Protecting against impulse expenditure |
| EP1301187A2 (en) | 2000-07-04 | 2003-04-16 | Novo Nordisk A/S | Heterocyclic compounds, which are inhibitors of the enzyme dpp-iv |
| US20040034014A1 (en) | 2000-07-04 | 2004-02-19 | Kanstrup Anders Bendtz | Heterocyclic compounds, which are inhibitors of the enzyme DPP-IV |
| WO2002002560A2 (en) | 2000-07-04 | 2002-01-10 | Novo Nordisk A/S | Purine-2,6-diones which are inhibitors of the enzyme dipeptidyl peptidase iv (dpp-iv) |
| WO2002014271A1 (en) | 2000-08-10 | 2002-02-21 | Mitsubishi Pharma Corporation | Proline derivatives and use thereof as drugs |
| US20040063935A1 (en) | 2000-10-06 | 2004-04-01 | Kosuke Yasuda | Aliphatic nitrogenous five-membered ring compounds |
| JP2004035574A (en) | 2000-10-06 | 2004-02-05 | Tanabe Seiyaku Co Ltd | Aliphatic nitrogenous five-membered ring compound |
| JP2002356471A (en) | 2000-10-06 | 2002-12-13 | Tanabe Seiyaku Co Ltd | Aliphatic nitrogen-containing five-membered ring compound |
| JP2002356472A (en) | 2000-10-06 | 2002-12-13 | Tanabe Seiyaku Co Ltd | Nitrogen-containing five-membered ring compound |
| US6849622B2 (en) | 2000-10-06 | 2005-02-01 | Tanabe Seiyaku Co., Ltd. | Aliphatic nitrogenous five-membered ring compounds |
| US20040229926A1 (en) | 2000-10-06 | 2004-11-18 | Tanabe Seiyaku Co., Ltd. | Aliphatic nitrogen - containing 5 - membered ring compound |
| WO2002030890A1 (en) | 2000-10-06 | 2002-04-18 | Tanabe Seiyaku Co., Ltd. | Nitrogenous five-membered ring compounds |
| WO2002030891A1 (en) | 2000-10-06 | 2002-04-18 | Tanabe Seiyaku Co., Ltd. | Aliphatic nitrogenous five-membered ring compounds |
| WO2002034900A1 (en) | 2000-10-27 | 2002-05-02 | The University Of Sydney | Dipeptidyl peptidases |
| US20040072892A1 (en) | 2000-11-10 | 2004-04-15 | Hiroshi Fukushima | Cyanopyrrolidine derivatives |
| US20070112059A1 (en) | 2000-11-10 | 2007-05-17 | Taisho Pharmaceutical Co., Ltd. | Cyanopyrrolidine derivatives |
| WO2002038541A1 (en) | 2000-11-10 | 2002-05-16 | Taisho Pharmaceutical Co., Ltd. | Cyanopyrrolidine derivatives |
| EP1333025A1 (en) | 2000-11-10 | 2003-08-06 | Taisho Pharmaceutical Co., Ltd | Cyanopyrrolidine derivatives |
| EP1338651A1 (en) | 2000-12-01 | 2003-08-27 | Yamanouchi Pharmaceutical Co. Ltd. | Method of screening remedy for diabetes |
| US20040180925A1 (en) | 2000-12-27 | 2004-09-16 | Kenji Matsuno | Dipeptidylpeptidase-IV inhibitor |
| EP1354882A1 (en) | 2000-12-27 | 2003-10-22 | Kyowa Hakko Kogyo Co., Ltd. | Dipeptidyl peptidase iv inhibitor |
| CA2433090A1 (en) | 2000-12-27 | 2002-07-04 | Kyowa Hakko Kogyo Co., Ltd. | Dipeptidyl peptidase iv inhibitor |
| WO2002051836A1 (en) | 2000-12-27 | 2002-07-04 | Kyowa Hakko Kogyo Co., Ltd. | Dipeptidyl peptidase iv inhibitor |
| WO2002055088A1 (en) | 2001-01-16 | 2002-07-18 | Nippon Kayaku Kabushiki Kaisha | Remedies for bone marrow suppression and infectious diseases and white blood cell count elevating agents |
| WO2002062764A1 (en) | 2001-02-02 | 2002-08-15 | Takeda Chemical Industries, Ltd. | Fused heterocyclic compounds |
| EP1355886A1 (en) | 2001-02-02 | 2003-10-29 | Takeda Chemical Industries, Ltd. | Fused heterocyclic compounds |
| JP2003238566A (en) | 2001-02-02 | 2003-08-27 | Takeda Chem Ind Ltd | Condensed heterocyclic compound |
| US20040087587A1 (en) | 2001-02-24 | 2004-05-06 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Xanthine derivatives, the preparation thereof and their use as pharmaceutical compositions |
| WO2002068420A1 (en) | 2001-02-24 | 2002-09-06 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Xanthine derivative, production and use thereof as a medicament |
| US20020198205A1 (en) | 2001-02-24 | 2002-12-26 | Frank Himmelsbach | Xanthine derivatives, the preparation thereof and their use as pharmaceutical compositions |
| US20040077645A1 (en) | 2001-02-24 | 2004-04-22 | Frank Himmelsbach | Xanthine derivatives,production and use thereof as medicament |
| JP2004522786A (en) | 2001-02-24 | 2004-07-29 | ベーリンガー インゲルハイム ファルマ ゲゼルシャフト ミット ベシュレンクテル ハフツング ウント コンパニー コマンディトゲゼルシャフト | Xanthine derivatives, their preparation and their use as pharmaceutical compositions |
| JP2002265439A (en) | 2001-03-08 | 2002-09-18 | Mitsubishi Pharma Corp | Cyanopyrrolidine derivative and its use for medicine |
| EP1385508A1 (en) | 2001-03-27 | 2004-02-04 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| US20040106656A1 (en) | 2001-03-27 | 2004-06-03 | Ashton Wallace T | Dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| JP2004525929A (en) | 2001-03-27 | 2004-08-26 | メルク エンド カムパニー インコーポレーテッド | Dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2002076450A1 (en) | 2001-03-27 | 2002-10-03 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| EP1245568A1 (en) | 2001-03-28 | 2002-10-02 | Les Laboratoires Servier | Sulfonyl derivatives of amino acids and their use as inhibitors of dipeptidyl-peptidase IV (DPP IV) |
| US6716843B2 (en) | 2001-03-28 | 2004-04-06 | Les Laboratoires Servier | Alpha-amino acid sulphonyl compounds |
| US20030087950A1 (en) | 2001-03-28 | 2003-05-08 | Denanteuil Guillaume | New alpha-amino acid sulphonyl compounds |
| FR2822826A1 (en) | 2001-03-28 | 2002-10-04 | Servier Lab | NOVEL ALPHA-AMINO ACID SULPHONYL DERIVATIVES, PROCESS FOR PREPARING THEM AND PHARMACEUTICAL COMPOSITIONS CONTAINING SAME |
| WO2002083109A1 (en) | 2001-04-11 | 2002-10-24 | Ferring Bv | Treatment of type 2 diabetes with inhibitors of dipeptidyl peptidase iv |
| JP2004525179A (en) | 2001-04-11 | 2004-08-19 | フェリング ベスローテン フェンノートシャップ | Treatment of type II diabetes with dipeptidyl peptidase IV inhibitors |
| US6573287B2 (en) | 2001-04-12 | 2003-06-03 | Bristo-Myers Squibb Company | 2,1-oxazoline and 1,2-pyrazoline-based inhibitors of dipeptidyl peptidase IV and method |
| WO2002083128A1 (en) | 2001-04-12 | 2002-10-24 | Bristol-Myers Squibb Company | 2,1-oxazoline and 1,2-pyrazoline-based inhibitors of dipeptidyl peptidase iv and method |
| US20020183367A1 (en) | 2001-04-12 | 2002-12-05 | Sulsky Richard B. | 2,1-Oxazoline and 1,2-pyrazoline-based inhibitors of dipeptidyl peptidase IV and method |
| JP2004532220A (en) | 2001-04-12 | 2004-10-21 | ブリストル−マイヤーズ スクイブ カンパニー | Dipeptidyl peptidase IV 2,1-oxazoline and 1,2-pyrazolin based inhibitors and methods |
| EP1377288A1 (en) | 2001-04-12 | 2004-01-07 | Bristol-Myers Squibb Company | 2,1-oxazoline and 1,2-pyrazoline-based inhibitors of dipeptidyl peptidase iv and method |
| EP1258476A1 (en) | 2001-05-15 | 2002-11-20 | Les Laboratoires Servier | Alpha-amino acid derivatives, method for their preparation and their use as dipeptidyl-peptidase IV inhibitors (DPP IV) |
| JP2002363157A (en) | 2001-05-15 | 2002-12-18 | Lab Servier | NEW alpha-AMINO ACID COMPOUND, METHOD FOR PREPARING THE SAME AND PHARMACEUTICAL COMPOSITION CONTAINING THE SAME |
| US6706742B2 (en) | 2001-05-15 | 2004-03-16 | Les Laboratories Servier | Alpha-amino-acid compounds |
| US20030078247A1 (en) | 2001-05-15 | 2003-04-24 | De Nanteuil Guillaume | Alpha-amino-acid compounds |
| FR2824825A1 (en) | 2001-05-15 | 2002-11-22 | Servier Lab | NOVEL ALPHA-AMINO-ACID DERIVATIVES, THEIR PREPARATION PROCESS AND THE PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| WO2003000181A2 (en) | 2001-06-20 | 2003-01-03 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| EP1406872A2 (en) | 2001-06-20 | 2004-04-14 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| EP1406622A2 (en) | 2001-06-20 | 2004-04-14 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| JP2005500308A (en) | 2001-06-20 | 2005-01-06 | メルク エンド カムパニー インコーポレーテッド | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| JP2004535433A (en) | 2001-06-20 | 2004-11-25 | メルク エンド カムパニー インコーポレーテッド | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| WO2003000180A2 (en) | 2001-06-20 | 2003-01-03 | Merck & Co., Inc. | Dipeptidyl peptidase inhibitors for the treatment of diabetes |
| EP1399154A1 (en) | 2001-06-25 | 2004-03-24 | Ferring BV | 3-fluoro-pyrrolidines as antidiabetic agents |
| WO2003000250A1 (en) | 2001-06-25 | 2003-01-03 | Ferring Bv | 3-fluoro-pyrrolidines as antidiabetic agents |
| JP2004534815A (en) | 2001-06-25 | 2004-11-18 | フェリング ベスローテン フェンノートシャップ | 3-Fluoro-pyrrolidine as antidiabetic drug |
| WO2003002530A2 (en) | 2001-06-27 | 2003-01-09 | Smithkline Beecham Corporation | Pyrrolidines as dipeptidyl peptidase inhibitors |
| EP1399470A2 (en) | 2001-06-27 | 2004-03-24 | Probiodrug AG | Dipeptidyl peptidase iv inhibitors and their uses as anti-cancer agents |
| JP2004530729A (en) | 2001-06-27 | 2004-10-07 | プロバイオドラッグ アーゲー | Peptide structures useful for antagonizing dipeptidyl peptidase IV catalysis |
| WO2003002553A2 (en) | 2001-06-27 | 2003-01-09 | Smithkline Beecham Corporation | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| JP2004521149A (en) | 2001-06-27 | 2004-07-15 | プロバイオドラッグ アーゲー | Novel dipeptidyl peptidase IV inhibitors and their use as anticancer agents |
| US20030119750A1 (en) | 2001-06-27 | 2003-06-26 | Hans-Ulrich Demuth | Use of dipeptidyl peptidase IV inhibitors |
| WO2003002593A2 (en) | 2001-06-27 | 2003-01-09 | Probiodrug Ag | Peptide structures useful for competitive modulation of dipeptidyl peptidase iv catalysis |
| JP2004535445A (en) | 2001-06-27 | 2004-11-25 | スミスクライン ビーチャム コーポレーション | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| WO2003072556A1 (en) | 2001-06-27 | 2003-09-04 | Prosidion Ltd. | Glutaminyl based dpiv inhibitors |
| EP1399469A2 (en) | 2001-06-27 | 2004-03-24 | Probiodrug AG | Peptide structures useful for competitive modulation of dipeptidyl peptidase iv catalysis |
| WO2003002531A2 (en) | 2001-06-27 | 2003-01-09 | Smithkline Beecham Corporation | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| EP1399471A2 (en) | 2001-06-27 | 2004-03-24 | Probiodrug AG | Use of dipeptidyl peptidase iv inhibitors as therapeutics for neurological disorders |
| EP1399433A2 (en) | 2001-06-27 | 2004-03-24 | Smithkline Beecham Corporation | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| JP2004534836A (en) | 2001-06-27 | 2004-11-18 | プロバイオドラッグ アーゲー | Novel use of dipeptidyl peptidase IV inhibitors |
| EP1399420A2 (en) | 2001-06-27 | 2004-03-24 | SmithKline Beecham Corporation | Pyrrolidines as dipeptidyl peptidase inhibitors |
| WO2003002595A2 (en) | 2001-06-27 | 2003-01-09 | Probiodrug Ag | Dipeptidyl peptidase iv inhibitors and their uses as anti-cancer agents |
| JP2005500321A (en) | 2001-06-27 | 2005-01-06 | スミスクライン ビーチャム コーポレーション | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| WO2003002596A2 (en) | 2001-06-27 | 2003-01-09 | Probiodrug Ag | Use of dipeptidyl peptidase iv inhibitors as therapeutics for neurological disorders |
| US20030130199A1 (en) | 2001-06-27 | 2003-07-10 | Von Hoersten Stephan | Dipeptidyl peptidase IV inhibitors and their uses as anti-cancer agents |
| EP1406873A2 (en) | 2001-06-27 | 2004-04-14 | Smithkline Beecham Corporation | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| EP1404675A1 (en) | 2001-07-03 | 2004-04-07 | Novo Nordisk A/S | Dpp-iv-inhibiting purine derivatives for the treatment of diabetes |
| JP2005502624A (en) | 2001-07-03 | 2005-01-27 | ノボ ノルディスク アクティーゼルスカブ | Purine derivatives inhibiting DPP-IV for the treatment of diabetes |
| US20030105077A1 (en) | 2001-07-03 | 2003-06-05 | Kanstrup Anders Bendtz | Heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| WO2003004496A1 (en) | 2001-07-03 | 2003-01-16 | Novo Nordisk A/S | Dpp-iv-inhibiting purine derivatives for the treatment of diabetes |
| US6869947B2 (en) | 2001-07-03 | 2005-03-22 | Novo Nordisk A/S | Heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| EP1412357A1 (en) | 2001-07-06 | 2004-04-28 | Merck & Co., Inc. | Beta-amino tetrahydroimidazo(1,2-a)pyrazines and tetrahydrotriazolo(4,3-a)pyrazines as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| US20030100563A1 (en) | 2001-07-06 | 2003-05-29 | Edmondson Scott D. | Beta-amino heterocyclic dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2003004498A1 (en) | 2001-07-06 | 2003-01-16 | Merck & Co., Inc. | Beta-amino tetrahydroimidazo (1, 2-a) pyrazines and tetrahydrotrioazolo (4, 3-a) pyrazines as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| US6699871B2 (en) | 2001-07-06 | 2004-03-02 | Merck & Co., Inc. | Beta-amino heterocyclic dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| JP2004536115A (en) | 2001-07-06 | 2004-12-02 | メルク エンド カムパニー インコーポレーテッド | Β-aminotetrahydroimidazo (1,2-A) pyrazines and tetrahydrotriazolo (4,3-A) pyrazines as dipeptidyl peptidase inhibitors for treating or preventing diabetes |
| JP2005505531A (en) | 2001-08-17 | 2005-02-24 | プロビオドルグ アーゲー | Novel dipeptidyl peptidase IV inhibitors and their use to reduce blood pressure levels |
| WO2003015775A1 (en) | 2001-08-17 | 2003-02-27 | Probiodrug Ag | New dipeptidyl peptidase iv inhibitors and their uses for lowering blood pressure levels |
| US20030119738A1 (en) | 2001-09-06 | 2003-06-26 | Andre Niestroj | Novel inhibitors of dipeptidyl peptidase I |
| DE10143840A1 (en) | 2001-09-06 | 2003-03-27 | Probiodrug Ag | New acylated hydroxamates useful for the treatment of e.g. wound healing |
| WO2003022871A2 (en) | 2001-09-06 | 2003-03-20 | Probiodrug Ag | Peptides having a c- terminal hydroxylamino group as inhibitors of dipeptidyl peptidase i |
| WO2003024942A1 (en) | 2001-09-14 | 2003-03-27 | Mitsubishi Pharma Corporation | Thiazolidine derivative and medicinal use thereof |
| EP1426366A1 (en) | 2001-09-14 | 2004-06-09 | Mitsubishi Pharma Corporation | Thiazolidine derivative and medicinal use thereof |
| US20040259883A1 (en) | 2001-09-14 | 2004-12-23 | Hiroshi Sakashita | Thiazolidine derivative and medicinal use thereof |
| WO2003024965A2 (en) | 2001-09-19 | 2003-03-27 | Novo Nordisk A/S | Heterocyclic compounds that are inhibitors of the enzyme dpp-iv |
| US20030199528A1 (en) | 2001-09-19 | 2003-10-23 | Kanstrup Anders B. | Hetrocyclic compounds that are inhibitors of the enzyme DPP-IV |
| EP1463727A2 (en) | 2001-09-19 | 2004-10-06 | Novo Nordisk A/S | Heterocyclic compounds that are inhibitors of the enzyme dpp-iv |
| EP1450794A1 (en) | 2001-10-23 | 2004-09-01 | Ferring B.V. | Novel dipeptidyl peptidase iv (dp-iv) inhibitors as anti-diabetic agents |
| US20050004205A1 (en) | 2001-10-23 | 2005-01-06 | Evans David M | Novel dipeptidyl peptidase iv (dp-iv) inhibitors as anti-diabetic agents |
| EP1446116A1 (en) | 2001-10-23 | 2004-08-18 | Ferring B.V. | Inhibitors of dipeptidyl peptidase iv |
| WO2003035067A1 (en) | 2001-10-23 | 2003-05-01 | Ferring B.V. | Novel dipeptidyl peptidase iv (dp-iv) inhibitors as anti-diabetic agents |
| WO2003035057A1 (en) | 2001-10-23 | 2003-05-01 | Ferring B.V. | Inhibitors of dipeptidyl peptidase iv |
| WO2003037327A1 (en) | 2001-10-26 | 2003-05-08 | F. Hoffmann-La-Roche Ag | N-substituted pyrrolidin derivatives as dipeptidyl peptidase iv inhibitors |
| US6861440B2 (en) | 2001-10-26 | 2005-03-01 | Hoffmann-La Roche Inc. | DPP IV inhibitors |
| US20030130281A1 (en) | 2001-10-26 | 2003-07-10 | Markus Boehringer | DPP IV inhibitors |
| EP1441719A1 (en) | 2001-10-26 | 2004-08-04 | F. Hoffmann-La Roche Ag | N-substituted pyrrolidin derivatives as dipeptidyl peptidase iv inhibitors |
| JP2005507261A (en) | 2001-10-31 | 2005-03-17 | ノバルティス アクチエンゲゼルシャフト | Methods for treating diabetes and related conditions based on polymorphisms in the TCF1 gene |
| WO2003038123A2 (en) | 2001-10-31 | 2003-05-08 | Novartis Ag | Methods to treat diabetes and related conditions based on polymorphisms in the tcf1 gene |
| US20030125304A1 (en) | 2001-11-09 | 2003-07-03 | Hans-Ulrich Demuth | Substituted amino ketone compounds |
| WO2003040174A2 (en) | 2001-11-09 | 2003-05-15 | Probiodrug Ag | Substituted amino ketone compounds |
| EP1442049A2 (en) | 2001-11-09 | 2004-08-04 | Probiodrug AG | Substituted amino ketone compounds |
| EP1469873A2 (en) | 2001-11-26 | 2004-10-27 | Trustees of Tufts College | Peptidomimetic inhibitors of post-proline cleaving enzymes |
| WO2003045228A2 (en) | 2001-11-26 | 2003-06-05 | Trustees Of Tufts College | Methods for treating autoimmune disorders, and reagents related thereto |
| CA2466870A1 (en) | 2001-11-26 | 2003-06-05 | Trustees Of Tufts College | Methods for treating autoimmune disorders, and reagents related thereto |
| WO2003057144A2 (en) | 2001-12-26 | 2003-07-17 | Guilford Pharmaceuticals | Change inhibitors of dipeptidyl peptidase iv |
| EP1465891A2 (en) | 2001-12-26 | 2004-10-13 | Guilford Pharmaceuticals Inc. | Inhibitors of dipeptidyl peptidase iv |
| WO2003057666A2 (en) | 2001-12-26 | 2003-07-17 | Guilford Pharmaceuticals | Inhibitors of dipeptidyl peptidase iv |
| US20040176406A1 (en) | 2001-12-27 | 2004-09-09 | Gobbi Luca Claudio | Pyrido [2,1-a] isoquinoline derivatives |
| US20030149071A1 (en) | 2001-12-27 | 2003-08-07 | Gobbi Luca Claudio | Pyrido [2,1-a] isoquinoline derivatives |
| WO2003055881A1 (en) | 2001-12-27 | 2003-07-10 | F. Hoffmann-La Roche Ag | Pyrido(2,1-a)isoquinoline derivatives as dpp-iv inhibitors |
| EP1461337A1 (en) | 2001-12-27 | 2004-09-29 | F. Hoffmann-La Roche Ag | Pyrido[2,1-a]isoquinoline derivatives as dpp-iv inhibitors |
| US6727261B2 (en) | 2001-12-27 | 2004-04-27 | Hoffman-La Roche Inc. | Pyrido[2,1-A]Isoquinoline derivatives |
| WO2003072528A2 (en) | 2002-02-08 | 2003-09-04 | Idun Pharmaceuticals, Inc. | (substituted)acyl dipeptidyl inhibitors of the ice/ced-3 family of cysteine proteases |
| US20030232788A1 (en) | 2002-02-08 | 2003-12-18 | Idun Pharmaceuticals, Inc. | (Substituted)acyl dipeptidyl inhibitors of the ICE/ced-3 family of cysteine proteases |
| EP1476435A1 (en) | 2002-02-13 | 2004-11-17 | F. Hoffmann-La Roche Ag | Novel pyridin- and pyrimidin-derivatives |
| US20030216382A1 (en) | 2002-02-13 | 2003-11-20 | Markus Boehringer | Pyridine and pyrimidine derivatives |
| US6867205B2 (en) | 2002-02-13 | 2005-03-15 | Hoffman-La Roche Inc. | Pyridine and pyrimidine derivatives |
| US6800650B2 (en) | 2002-02-13 | 2004-10-05 | Hoffmann-La Roche Inc. | Pyridine and quinoline derivatives |
| WO2003068748A1 (en) | 2002-02-13 | 2003-08-21 | F. Hoffmann-La Roche Ag | Novel pyridine- and quinoline-derivatives |
| WO2003068757A1 (en) | 2002-02-13 | 2003-08-21 | F. Hoffmann-La Roche Ag | Novel pyridin- and pyrimidin-derivatives |
| US20030195188A1 (en) | 2002-02-13 | 2003-10-16 | Markus Boehringer | Pyridine and quinoline derivatives |
| EP1476429A1 (en) | 2002-02-13 | 2004-11-17 | F. Hoffmann-La Roche Ag | Novel pyridine- and quinoline-derivatives |
| EP1338592A1 (en) | 2002-02-22 | 2003-08-27 | Nippon Zoki Pharmaceutical Co., Ltd. | Novel 2-phenylpiperazine derivatives |
| US20040082570A1 (en) | 2002-02-25 | 2004-04-29 | Eisai Co., Ltd. | Xanthine derivative and DPPIV inhibitor |
| JP2004043429A (en) | 2002-02-25 | 2004-02-12 | Eisai Co Ltd | New xanthine derivative and dppiv inhibitor |
| EP1480961A1 (en) | 2002-02-28 | 2004-12-01 | Prosidion Limited | Glutaminyl based dpiv inhibitors |
| US20030162820A1 (en) | 2002-02-28 | 2003-08-28 | Hans-Ulrich Demuth | Glutaminyl based DPIV inhibitors |
| WO2003074500A2 (en) | 2002-03-06 | 2003-09-12 | Sanofi-Aventis | N-aminoacetyl-pyrrolidine-2-carbonitriles and their use as ddp-iv inhibitors |
| WO2003080633A1 (en) | 2002-03-25 | 2003-10-02 | Nippon Kayaku Kabushiki Kaisha | Novel $g(a)-amino-n-(diaminophosphinyl)lactam derivative |
| WO2003082817A2 (en) | 2002-03-25 | 2003-10-09 | Merck & Co., Inc. | Beta-amino heterocyclic dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| EP1489088A1 (en) | 2002-03-25 | 2004-12-22 | Nippon Kayaku Kabushiki Kaisha | Novel alpha-amino-n-(diaminophosphinyl)lactam derivative |
| EP1490335A2 (en) | 2002-03-25 | 2004-12-29 | Merck & Co., Inc. | Beta-amino heterocyclic dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| JP2004002368A (en) | 2002-04-04 | 2004-01-08 | Tanabe Seiyaku Co Ltd | Pharmaceutical composition |
| JP2004002367A (en) | 2002-04-04 | 2004-01-08 | Tanabe Seiyaku Co Ltd | Pharmaceutical composition |
| EP1492777A1 (en) | 2002-04-08 | 2005-01-05 | Torrent Pharmaceuticals Ltd | Thiazolidine-4-carbonitriles and analogues and their use as dipeptidyl-peptidase inhibitors |
| WO2003084940A1 (en) | 2002-04-08 | 2003-10-16 | Alangudi Sankaranarayanan | Thiazolidine-4-carbonitriles and analogues and their use as dipeptidyl-peptidas inhibitors |
| US20030225102A1 (en) | 2002-04-08 | 2003-12-04 | Torrent Pharmaceuticals Ltd. | Novel compounds and therapeutic uses thereof |
| JP2003300977A (en) | 2002-04-10 | 2003-10-21 | Sumitomo Pharmaceut Co Ltd | Xanthine derivative |
| JP2004026820A (en) | 2002-05-09 | 2004-01-29 | Taisho Pharmaceut Co Ltd | Dipeptidyl peptidase iv inhibitor |
| WO2003095425A1 (en) | 2002-05-09 | 2003-11-20 | Taisho Pharmaceutical Co.,Ltd. | Cyanopyrrolidine derivatives |
| JP2003327532A (en) | 2002-05-10 | 2003-11-19 | Takeda Chem Ind Ltd | Peptidase inhibitor |
| WO2003099279A1 (en) | 2002-05-29 | 2003-12-04 | Novartis Ag | Combination of a dpp iv inhibitor and a cardiovascular compound |
| WO2003101448A1 (en) | 2002-06-03 | 2003-12-11 | Novartis Ag | The use of substituted cyanopyrrolidines and combination preparations containing them for treating hyperlipidemia and associated diseases |
| US6812350B2 (en) | 2002-06-04 | 2004-11-02 | Pfizer Inc. | Synthesis of 3,3,4,4-tetrafluoropyrrolidine and novel dipeptidyl peptidase-IV inhibitor compounds |
| WO2003101958A2 (en) | 2002-06-04 | 2003-12-11 | Pfizer Products Inc. | Flourinated cyclic amides as dipeptidyl peptidase iv inhibitors |
| US6710040B1 (en) | 2002-06-04 | 2004-03-23 | Pfizer Inc. | Fluorinated cyclic amides as dipeptidyl peptidase IV inhibitors |
| US20040242898A1 (en) | 2002-06-04 | 2004-12-02 | Pfizer Inc | Synthesis of 3,3,4,4-tetrafluoropyrrolidine and novel dipeptidyl peptidase-IV inhibitor compounds |
| EP1513808A2 (en) | 2002-06-04 | 2005-03-16 | Pfizer Products Inc. | Fluorinated cyclic amides as dipeptidyl peptidase iv inhibitors |
| WO2003104229A1 (en) | 2002-06-06 | 2003-12-18 | エーザイ株式会社 | Novel fused imidazole derivative |
| US20040116328A1 (en) | 2002-06-06 | 2004-06-17 | Eisai Co., Ltd. | Condensed imidazole derivatives |
| EP1517907A2 (en) | 2002-06-14 | 2005-03-30 | Sanofi-Aventis | Azabicyclo-octane and nonane derivatives with ddp-iv inhibiting activity |
| WO2003106456A2 (en) | 2002-06-14 | 2003-12-24 | Sanofi-Synthelabo | New compounds |
| WO2003105763A2 (en) | 2002-06-14 | 2003-12-24 | Amylin Pharmaceuticals, Inc. | Prevention and/or treatment of inflammatory bowel disease using pyy or agonists thereof |
| WO2004000327A1 (en) | 2002-06-24 | 2003-12-31 | Zaidan Hojin Biseibutsu Kagaku Kenkyu Kai | Therapeutic agent for type 2 diabetes |
| JP2004026678A (en) | 2002-06-24 | 2004-01-29 | Microbial Chem Res Found | Therapeutic agent for type 2 diabetes |
| WO2004004661A2 (en) | 2002-07-09 | 2004-01-15 | Point Therapeutics, Inc. | Boroproline compound combination therapy |
| WO2004007446A1 (en) | 2002-07-10 | 2004-01-22 | Yamanouchi Pharmaceutical Co., Ltd. | Novel azetidine derivative or salt thereof |
| WO2004007517A1 (en) | 2002-07-11 | 2004-01-22 | Aventis Pharma Deutschland Gmbh | Novel thiophenylglycoside derivatives, methods for production thereof, medicaments comprising said compounds and use thereof |
| WO2004007468A1 (en) | 2002-07-15 | 2004-01-22 | Merck & Co., Inc. | Piperidino pyrimidine dipeptidyl peptidase inhibitors for the treatment of diabetes |
| WO2004009544A1 (en) | 2002-07-23 | 2004-01-29 | Yamanouchi Pharmaceutical Co., Ltd. | 2-cyano-4-fluoropyrrolidine derivative or its salt |
| JP2004315496A (en) | 2002-08-08 | 2004-11-11 | Takeda Chem Ind Ltd | Fused heterocyclic ring compound |
| WO2004014860A2 (en) | 2002-08-08 | 2004-02-19 | Takeda Pharmaceutical Company Limited | Fused heterocyclic compounds as peptidase inhibitors |
| WO2004018468A2 (en) | 2002-08-21 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the production thereof and the use of the same as medicaments |
| DE10238243A1 (en) | 2002-08-21 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New 8-(3-amino-piperidin-1-yl)-xanthine derivatives are dipeptidylpeptidase-IV inhibitors useful for, e.g. treating diabetes mellitus, arthritis or obesity |
| US20040097510A1 (en) | 2002-08-21 | 2004-05-20 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| WO2004018469A1 (en) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel purine derivatives, production and use thereof as medicaments |
| WO2004018467A2 (en) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Phenacyl xanthine derivatives as dpp-iv inhibitor |
| DE10238470A1 (en) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New xanthine derivatives, their production and their use as medicines |
| DE10238477A1 (en) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New purine derivatives, their production and their use as medicines |
| WO2004020407A1 (en) | 2002-08-29 | 2004-03-11 | Taisho Pharmaceutical Co.,Ltd. | Benzenesulfonate of 4-fluoro-2-cyanopyrrolidine derivative |
| WO2004026822A2 (en) | 2002-09-19 | 2004-04-01 | Abbott Laboratories | Pharmaceutical compositions as inhibitors of dipeptidyl peptidase-iv (dpp-iv) |
| WO2004032836A2 (en) | 2002-10-07 | 2004-04-22 | Merck & Co., Inc. | Antidiabetic beta-amino heterocylcic dipeptidyl peptidase inhibitors |
| WO2004033455A2 (en) | 2002-10-08 | 2004-04-22 | Novo Nordisk A/S | Hemisuccinate salts of heterocyclic dpp-iv inhibitors |
| WO2004037169A2 (en) | 2002-10-18 | 2004-05-06 | Merck & Co., Inc. | Beta-amino heterocyclic dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004037181A2 (en) | 2002-10-23 | 2004-05-06 | Bristol-Myers Squibb Company | Glycinenitrile-based inhibitors of dipeptidyl peptidase iv and methods |
| WO2004041795A1 (en) | 2002-10-30 | 2004-05-21 | Guilford Pharmaceuticals Inc. | Novel inhibitors of dipeptidyl peptidase iv |
| WO2004048379A1 (en) | 2002-11-01 | 2004-06-10 | Sumitomo Pharmaceuticals Co., Ltd. | Xanthine compound |
| WO2004043940A1 (en) | 2002-11-07 | 2004-05-27 | Merck & Co., Inc. | Phenylalanine derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| DE10251927A1 (en) | 2002-11-08 | 2004-05-19 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New 1,7,8-trisubstituted xanthine derivatives, are dipeptidylpeptidase-IV inhibitors useful e.g. for treating diabetes mellitus type I or II, arthritis or obesity |
| WO2004041820A1 (en) | 2002-11-08 | 2004-05-21 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel xanthine derivatives, the production and the use thereof in the form of drugs |
| US20040138214A1 (en) | 2002-11-08 | 2004-07-15 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Xanthine derivatives, the preparation thereof and their use as pharmaceutical compositions |
| WO2004046106A1 (en) | 2002-11-18 | 2004-06-03 | Pfizer Products Inc. | Dipeptidyl peptidase iv inhibiting fluorinated cyclic amides |
| US20040110817A1 (en) | 2002-11-18 | 2004-06-10 | Pfizer Inc | Dipeptidyl peptidase IV inhibiting fluorinated cyclic amides |
| WO2004050658A1 (en) | 2002-12-03 | 2004-06-17 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel substituted imidazo-pyridinones and imidazo-pyridazeiones, the production and use thereof as medicaments |
| DE10256264A1 (en) | 2002-12-03 | 2004-06-24 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | New trisubstituted dihydro-imidazo-pyridazinone or imidazo-pyridinone derivatives, useful as dipeptidylpeptidase-IV inhibitors for e.g. treating diabetes mellitus type I or II, arthritis or obesity |
| WO2004050022A2 (en) | 2002-12-04 | 2004-06-17 | Merck & Co., Inc. | Phenylalanine derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004052850A2 (en) | 2002-12-09 | 2004-06-24 | Bristol-Myers Squibb Company | Methods and compounds producing dipeptidyl peptidase iv inhibitors and intermediates thereof |
| WO2004052362A1 (en) | 2002-12-10 | 2004-06-24 | Novartis Ag | Combination of an dpp-iv inhibitor and a ppar-alpha compound |
| WO2004058266A1 (en) | 2002-12-20 | 2004-07-15 | Merck & Co., Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004065380A1 (en) | 2003-01-14 | 2004-08-05 | Arena Pharmaceuticals Inc. | 1,2,3-trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prpphylaxis and treatment of disorders related thereto such as diabetes and hyperglycemia |
| WO2004064778A2 (en) | 2003-01-17 | 2004-08-05 | Merck & Co. Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| JP2004244412A (en) | 2003-01-20 | 2004-09-02 | Kotobuki Seiyaku Kk | 2-cyanopyrrolidine derivative having substituent at 4-position, method for producing the same, and medicament containing the same |
| WO2004069162A2 (en) | 2003-01-31 | 2004-08-19 | Merck & Co., Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004067509A1 (en) | 2003-01-31 | 2004-08-12 | Sanwa Kagaku Kenkyusho Co., Ltd. | Compound inhibiting dipeptidyl peptidase iv |
| WO2004071454A2 (en) | 2003-02-13 | 2004-08-26 | Guilford Pharmaceuticals Inc. | Substituted azetidine compounds as inhibitors of dipeptidyl peptidase iv |
| WO2004076413A2 (en) | 2003-02-24 | 2004-09-10 | Arena Pharmaceuticals, Inc. | Phenyl- and pyridylpiperidine-derivatives as modulators of glucose metabolism |
| WO2004076433A1 (en) | 2003-02-28 | 2004-09-10 | Aic | Dipeptidyl peptidase inhibitors |
| WO2004076434A1 (en) | 2003-02-28 | 2004-09-10 | Aic | Dipeptidyl peptidase inhibitors |
| WO2004080990A1 (en) | 2003-03-14 | 2004-09-23 | Astellas Pharma Inc. | C-glycoside derivatives and salts thereof |
| WO2004085378A1 (en) | 2003-03-19 | 2004-10-07 | Merck & Co. Inc. | Process for the preparation of chiral beta amino acid derivatives by asymmetric hydrogenation |
| WO2004085661A2 (en) | 2003-03-24 | 2004-10-07 | Merck & Co., Inc | Process to chiral beta-amino acid derivatives |
| WO2004087053A2 (en) | 2003-03-25 | 2004-10-14 | Syrrx, Inc. | Dipeptidyl peptidase inhibitors |
| WO2004087650A2 (en) | 2003-03-27 | 2004-10-14 | Merck & Co. Inc. | Process and intermediates for the preparation of beta-amino acid amide dipeptidyl peptidase-iv inhibitors |
| WO2004092128A1 (en) | 2003-04-10 | 2004-10-28 | Smithkline Beecham Corporation | Anhydrous crystalline forms of (2s, 4s)-1-{(2r)-2-amino-3-‘4-methoxybenzyl)sulfonyl!-3-methylbutanoyl}-4-fluoropyrrolindine-2-carbonitrile |
| WO2004096806A1 (en) | 2003-04-30 | 2004-11-11 | Sumitomo Pharmaceuticals Co. Ltd. | Fused imidazole derivative |
| WO2004099134A2 (en) | 2003-05-05 | 2004-11-18 | Prosidion Ltd. | Glutaminyl based dp iv-inhibitors |
| WO2004103276A2 (en) | 2003-05-14 | 2004-12-02 | Merck & Co., Inc. | 3-amino-4-phenylbutanoic acid derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| US20040254226A1 (en) | 2003-05-14 | 2004-12-16 | Syrrx, Inc. | Dipeptidyl peptidase inhibitors |
| WO2004103993A1 (en) | 2003-05-14 | 2004-12-02 | Syrrx, Inc. | Dipeptidyl peptidase inhibitors |
| WO2004104215A2 (en) | 2003-05-21 | 2004-12-02 | Bayer Healthcare Ag | Diagnostics and therapeutics for diseases associated with dipeptidylpeptidase 7 (dpp7) |
| WO2004104216A2 (en) | 2003-05-21 | 2004-12-02 | Bayer Healthcare Ag | Diagnostics and therapeutics for diseases associated with dipeptidylpeptidase iv (dpp4) |
| WO2004108730A1 (en) | 2003-06-05 | 2004-12-16 | Fujisawa Pharmaceutical Co., Ltd. | 1h-imidazo`4,5-d!pyridazines as dpp-iv inhibitors for the treatment of niddm |
| WO2004110375A2 (en) | 2003-06-06 | 2004-12-23 | Merck & Co., Inc. | Combination therapy for the treatment of diabetes |
| WO2004110436A1 (en) | 2003-06-06 | 2004-12-23 | Merck & Co., Inc. | Fused indoles as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004111041A1 (en) | 2003-06-12 | 2004-12-23 | Fujisawa Pharmaceutical Co., Ltd. | Pyrrolidine, thiazolidine and oxazolidine compounds which inhibit dipeptidyl peptidase-iv (dpp) |
| WO2004112701A2 (en) | 2003-06-17 | 2004-12-29 | Merck & Co., Inc. | Cyclohexylglycine derivatives as dipeptidyl peptidase inhibitors for the treatment or prevention of diabetes |
| WO2004111051A1 (en) | 2003-06-18 | 2004-12-23 | Boehringer Ingelheim International Gmbh | Imidazo-pyridazinone derivatives and imidazo-pyridone derivatives, production thereof, and use thereof as medicaments |
| DE10327439A1 (en) | 2003-06-18 | 2005-01-05 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel imidazopyridazinone and imidazopyridone derivatives, their production and their use as pharmaceuticals |
| US20040259902A1 (en) | 2003-06-20 | 2004-12-23 | Markus Boehringer | Pyrido [2,1-a] isoquinoline derivatives |
| WO2005000846A1 (en) | 2003-06-20 | 2005-01-06 | F.Hoffmann-La Roche Ag | Hexahydropyridoisoqinolines as dpp-iv inhibitors |
| US20040259903A1 (en) | 2003-06-20 | 2004-12-23 | Markus Boehringer | Pyrido [2,1-a] isoquinoline derivatives |
| WO2005000848A1 (en) | 2003-06-20 | 2005-01-06 | F. Hoffmann-La Roche Ag | Pyrido` 2, 1-a - isoquinoline derivatives as dpp-iv inhibitors |
| US20050032804A1 (en) | 2003-06-24 | 2005-02-10 | Cypes Stephen Howard | Phosphoric acid salt of a dipeptidyl peptidase-IV inhibitor |
| WO2005003135A1 (en) | 2003-06-24 | 2005-01-13 | Merck & Co., Inc. | Phosphoric acid salt of a dipeptidyl peptidase-iv inhibitor |
| JP2005023038A (en) | 2003-07-04 | 2005-01-27 | Taisho Pharmaceut Co Ltd | Therapeutic agent for chronic renal disease |
| WO2005007647A1 (en) * | 2003-07-11 | 2005-01-27 | Arena Pharmaceuticals, Inc. | Trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto |
| WO2005009956A1 (en) | 2003-07-21 | 2005-02-03 | Smithkline Beecham Corporation | (2s,4s)-4-fluoro-1-[4-fluoro-beta-(4-fluorophenyl)-l-phenylalanyl]-2-pyrrolidinecarbonitrile p-toluenesulfonic acid salt and anhydrous crystalline forms thereof |
| DE10333935A1 (en) | 2003-07-25 | 2005-02-24 | Aventis Pharma Deutschland Gmbh | New bicyclic cyano-heterocycles, process for their preparation and their use as pharmaceuticals |
| US20050059716A1 (en) | 2003-07-25 | 2005-03-17 | Aventis Pharma Deutschland Gmbh | Novel bicyclic cyanoheterocycles, process for their preparation and their use as medicaments |
| WO2005012312A1 (en) | 2003-07-25 | 2005-02-10 | Sanofi-Aventis Deutschland Gmbh | Novel cyano thiazolides, methods for the production thereof, and use thereof as a medicament |
| US20050059724A1 (en) | 2003-07-25 | 2005-03-17 | Aventis Pharma Deutschland Gmbh | Novel cyanopyrrolidides, process for their preparation and their use as medicaments |
| WO2005012308A1 (en) | 2003-07-25 | 2005-02-10 | Sanofi-Aventis Deutschland Gmbh | Novel cyanopyrrolidides, methods for the production thereof, and use of the same as medicaments |
| WO2005011581A2 (en) | 2003-07-31 | 2005-02-10 | Merck & Co., Inc. | Hexahydrodiazepinones as dipeptidyl peptidase-iv inhibitors for the treatment or prevention of diabetes |
| WO2005012326A1 (en) | 2003-08-01 | 2005-02-10 | Tanabe Seiyaku Co., Ltd. | Novel compounds having inhibitory activity against sodium-dependant transporter |
| US20050038020A1 (en) | 2003-08-01 | 2005-02-17 | Hamann Lawrence G. | Adamantylglycine-based inhibitors of dipeptidyl peptidase IV and methods |
| WO2005012249A2 (en) | 2003-08-01 | 2005-02-10 | Bristol-Myers Squibb Company | Adamantyglycine- based inhibitors of dipeptidyl peptidase iv for the treatment of diabetes |
| WO2005019168A2 (en) | 2003-08-20 | 2005-03-03 | Pfizer Products Inc. | Fluorinated lysine derivatives as dipeptidyl peptidase iv inhibitors |
| US20050043292A1 (en) | 2003-08-20 | 2005-02-24 | Pfizer Inc | Fluorinated lysine derivatives as dipeptidyl peptidase IV inhibitors |
| WO2005020920A2 (en) | 2003-09-02 | 2005-03-10 | Merck & Co., Inc. | Novel crystalline forms of a phosphoric acid salt of a dipeptidyl peptidase-iv inhibitor |
| WO2005023762A1 (en) | 2003-09-04 | 2005-03-17 | Abbott Laboratories | Pyrrolidine-2-carbonitrile derivatives and their use as inhibitors of dipeptidyl peptidase-iv (dpp-iv) |
| WO2005026148A1 (en) | 2003-09-08 | 2005-03-24 | Takeda San Diego, Inc. | Dipeptidyl peptidase inhibitors |
| WO2005030751A2 (en) | 2003-09-08 | 2005-04-07 | Takeda Pharmaceutical Company Limited | Dipeptidyl peptidase inhibitors |
| WO2005025554A2 (en) | 2003-09-09 | 2005-03-24 | Japan Tobacco Inc. | Dipeptidyl peptidase iv inhibitor |
| WO2005030127A2 (en) | 2003-09-23 | 2005-04-07 | Merck & Co., Inc. | Novel crystalline form of a phosphoric acid salt of a dipeptidyl peptidase-iv inhibitor |
| WO2005032590A1 (en) | 2003-10-03 | 2005-04-14 | Takeda Pharmaceutical Company Limited | Remedy for diabetes |
| WO2005033099A2 (en) | 2003-10-03 | 2005-04-14 | Glenmark Pharmaceuticals Ltd. | Novel dipeptidyl peptidase iv inhibitors; processes for their preparation and compositions thereof |
| WO2005037779A2 (en) | 2003-10-15 | 2005-04-28 | Imtm Gmbh | Novel dipeptidyl peptidase iv inhibitors used for functionally influencing different cells and treating immunological, inflammatory, neuronal, and other diseases |
| WO2005034940A2 (en) | 2003-10-15 | 2005-04-21 | Imtm Gmbh | Dual alanyl aminopeptidase and dipeptidyl peptidase iv inhibitors for functionally influencing different cells and for treating immunological, inflammatory, neuronal and other diseases |
| WO2005040095A1 (en) | 2003-10-16 | 2005-05-06 | Astrazeneca Ab | Inhibitors of dipeptidyl peptidase iv |
| WO2005037828A1 (en) | 2003-10-20 | 2005-04-28 | Lg Life Sciences Ltd. | Novel inhibitors of dpp-iv, methods of preparing the same, and pharmaceutical compositions containing the same as an active agent |
| WO2005042488A1 (en) | 2003-10-31 | 2005-05-12 | Takeda Pharmaceutical Company Limited | Pyridine compounds as inhibitors of dipeptidyl peptidase iv |
| WO2005044195A2 (en) | 2003-11-04 | 2005-05-19 | Merck & Co., Inc. | Fused phenylalanine derivatives as dipeptidyl peptidase-iv inhibitors for the treatment or prevention of diabetes |
| WO2005047297A1 (en) | 2003-11-12 | 2005-05-26 | Phenomix Corporation | Heterocyclic boronic acid compounds |
| WO2005049022A2 (en) | 2003-11-17 | 2005-06-02 | Novartis Ag | Use of dipeptidyl peptidase iv inhibitors |
| WO2005058849A1 (en) | 2003-12-15 | 2005-06-30 | Glenmark Pharmaceuticals Ltd. | New dipeptidyl peptidase in inhibitors; process for their preparation and compositions containing them |
| WO2005063750A1 (en) | 2003-12-23 | 2005-07-14 | Boehringer Ingelheim International Gmbh | Bicyclic imidazole compounds, the production thereof and their use as medicaments |
| WO2005072530A1 (en) | 2004-01-16 | 2005-08-11 | Merck & Co., Inc. | Novel crystalline salts of a dipeptidyl peptidase-iv inhibitor |
| WO2005075426A1 (en) | 2004-02-03 | 2005-08-18 | Glenmark Pharmaceuticals Ltd. | Novel dipeptidyl peptidase iv inhibitors; processes for their preparation and compositions thereof |
| US20080146818A1 (en) | 2004-02-05 | 2008-06-19 | Yasumichi Fukuda | Bicycloester Derivative |
| WO2005079795A2 (en) | 2004-02-20 | 2005-09-01 | Novartis Ag | Dpp-iv inhibitors for treating neurodegeneration and cognitive disorders |
| WO2005082348A2 (en) | 2004-02-23 | 2005-09-09 | Trustees Of Tufts College | Inhibitors of dipeptidylpeptidase iv for regulating glucose metabolism |
| WO2005082849A1 (en) | 2004-02-23 | 2005-09-09 | Trustees Of Tufts College | Lactams as conformationally constrained peptidomimetic inhibitors |
| WO2005087235A1 (en) | 2004-03-09 | 2005-09-22 | National Health Research Institutes | Pyrrolidine compounds |
| WO2005095381A1 (en) | 2004-03-15 | 2005-10-13 | Takeda Pharmaceutical Company Limited | Dipeptidyl peptidase inhibitors |
| WO2005092877A1 (en) | 2004-03-16 | 2005-10-06 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzol derivatives, drugs containing said compounds, the use thereof and method for the production thereof |
| WO2005116014A1 (en) | 2004-05-12 | 2005-12-08 | Pfizer Products Inc. | Proline derivatives and their use as dipeptidyl peptidase iv inhibitors |
| US20070167468A1 (en) | 2004-08-06 | 2007-07-19 | Sanofi-Aventis Deutschland Gmbh | Substituted bicyclic 8-pyrr0lidinoxanthines, methods for their production, pharmaceutical formulations and their use |
| WO2006040625A1 (en) | 2004-10-12 | 2006-04-20 | Glenmark Pharmaceuticals S.A. | Novel dipeptidyl peptidase iv inhibitors, pharmaceutical compositions containing them, and process for their preparation |
| US20090192129A1 (en) | 2004-12-12 | 2009-07-30 | Dainippon Sumitomo Pharma Co., Ltd. | Bicyclic pyrrole derivatives |
| WO2006068163A1 (en) | 2004-12-24 | 2006-06-29 | Dainippon Sumitomo Pharma Co., Ltd. | Bicyclic pyrrole derivatives |
| WO2006076231A2 (en) | 2005-01-10 | 2006-07-20 | Arena Pharmaceuticals, Inc. | Combination therapy for the treatment of diabetes and conditions related thereto and for the treatment of conditions ameliorated by increasing a blood glp-1 level |
| WO2006080421A1 (en) | 2005-01-28 | 2006-08-03 | Chugai Seiyaku Kabushiki Kaisha | Spiroketal derivative and use thereof as diabetic medicine |
| WO2006088129A1 (en) | 2005-02-18 | 2006-08-24 | Mitsubishi Pharma Corporation | Salt of proline derivative, solvate thereof, and production method thereof |
| US20090216016A1 (en) | 2005-02-18 | 2009-08-27 | Mitsubishi Pharma Corporation | Salt of proline derivative, solvate thereof, and production method thereof |
| WO2006100181A2 (en) | 2005-03-22 | 2006-09-28 | F. Hoffmann-La Roche Ag | New salt and polymorphs of a dpp-iv inhibitor |
| WO2006116157A2 (en) | 2005-04-22 | 2006-11-02 | Alantos Pharmaceuticals Holding, Inc. | Dipeptidyl peptidase-iv inhibitors |
| EP1902730A1 (en) | 2005-06-09 | 2008-03-26 | Banyu Pharmaceutical Co., Ltd. | Npy y2 agonist for use as therapeutic agent for disease accompanied by diarrhea |
| WO2007014815A1 (en) | 2005-07-29 | 2007-02-08 | Trasmetal S.P.A. | Paint booth for plants for painting products |
| WO2007019255A2 (en) | 2005-08-04 | 2007-02-15 | Novartis Ag | Salts of vildagliptin |
| WO2007027651A2 (en) | 2005-08-30 | 2007-03-08 | Abbott Laboratories | Pharmaceutical compositions as inhibitors of dipeptidyl peptidase-iv (dpp-iv) |
| WO2007035372A2 (en) | 2005-09-16 | 2007-03-29 | Takeda Pharmaceutical Company Limited | Polymorphs of benzoate salt of 2-[[6-[(3r)-3-amino-1-piperidinyl]-3,4-dihydro-3-methyl-2,4-dioxo-1(2h)-pyrimidinyl]methyl]-benzonitrile and methods of use therefor |
| WO2007071576A1 (en) | 2005-12-21 | 2007-06-28 | F. Hoffmann-La Roche Ag | New salt and polymorph of dpp-iv inhibitor |
| WO2007120689A2 (en) | 2006-04-11 | 2007-10-25 | Arena Pharmaceuticals, Inc. | Methods of using gpr119 receptor to identify compounds useful for increasing bone mass in an individual |
| WO2007120702A2 (en) | 2006-04-11 | 2007-10-25 | Arena Pharmaceuticals, Inc. | Use of gpr119 receptor agonists for increasing bone mass and for treating osteoporosis, and combination therapy relating thereto |
| WO2007128721A1 (en) | 2006-05-04 | 2007-11-15 | Boehringer Ingelheim Internationalgmbh | Polymorphs |
| WO2007148185A2 (en) | 2006-06-21 | 2007-12-27 | Pfizer Products Inc. | Substituted 3 -amino- pyrrolidino-4 -lactams as dpp inhibitors |
| WO2008027273A2 (en) | 2006-08-30 | 2008-03-06 | Phenomix Corporation | Solid citrate and tartrate salts of dpp-iv inhibitors |
| WO2008067465A1 (en) | 2006-11-29 | 2008-06-05 | Takeda Pharmaceutical Company Limited | Polymorphs of succinate salt of 2-[6-(3-amino-piperidin-1-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2h-pyrimidin-1-ylmethy]-4-fluor-benzonitrile and methods of use therefor |
| WO2008070692A2 (en) * | 2006-12-06 | 2008-06-12 | Smithkline Beecham Corporation | Bicyclic compounds and use as antidiabetics |
| WO2008109591A1 (en) | 2007-03-08 | 2008-09-12 | Lexicon Pharmaceuticals, Inc. | Phlorizin analogs as inhibitors of sodium glucose co-transporter 2 |
| WO2008114857A1 (en) | 2007-03-22 | 2008-09-25 | Kyorin Pharmaceutical Co., Ltd. | Method for producing aminoacetylpyrrolidinecarbonitrile derivative |
| WO2009084497A1 (en) | 2007-12-28 | 2009-07-09 | Dainippon Sumitomo Pharma Co., Ltd. | Methyl-substituted piperidine derivative |
| WO2009126245A1 (en) | 2008-04-07 | 2009-10-15 | Arena Pharmaceuticals, Inc. | Methods of using a g protein-coupled receptor to identify peptide yy (pyy) secretagogues and compounds useful in the treatment of conditions modulated by pyy |
Non-Patent Citations (124)
| Title |
|---|
| "Prevention and Management of Osteopoosis", 2003, WORLD HEALTH ORGANIZATION TECHNICAL REPORT SERIES 921 |
| '1'SUKIYAMA ET AL., MOL ENDOCRINOL, vol. 20, 2006, pages 1644 - 1651 |
| ABBOTT ET AL., BRAIN RES, vol. 1043, 2005, pages 139 - 144 |
| ABE ET AL., J. NA.T PROD., vol. 67, 2004, pages 999 - 1004 |
| ADRIAN ET AL., GASTROENTEROL., vol. 89, 1985, pages 1070 - 1077 |
| AHREN ET AL., DIABETES CARE, vol. 25, 2002, pages 869 - 875 |
| AHREN ET AL., ENDOCRINOLOGY, vol. 146, 2005, pages 2055 - 2059 |
| ALREN ET AL., J. CLIN. ENDOCRINOL. METAB., vol. 89, 2004, pages 2078 - 2084 |
| AREHART ET AL., CIRC RES, CIRC. RES., vol. 102, 2008, pages 986 - 993 |
| ATIK ET AL., CLIN ORTHOP RELAT RES, vol. 443, 2006, pages 19 - 24 |
| BALASUBRAMANIAM ET AL., J. MED. CHEM., vol. 43, 2000, pages 3420 - 3427 |
| BALASUBRAMANIAM ET AL., PEPTIDES, vol. 28, 2007, pages 235 - 240 |
| BALASUBRAMANIAM, J. MED. CHEM., vol. 43, 2000, pages 3420 - 3427 |
| BATTERHAM ET AL., NATURE, vol. 418, 2002, pages 650 - 654 |
| BEHRE, SCAND J CLIN LAB INVEST, vol. 67, 2007, pages 449 - 458 |
| BERGE ET AL., JOURNAL OF PHARMACEUTICAL SCIENCES, vol. 66, 1977, pages 1 - 19 |
| BILCHIK ET AL., GASTROENTEROL., vol. 105, 1993, pages 1441 - 1448 |
| BOEY ET AL., DIABETOLOGIA, vol. 49, 2006, pages 1360 - 1370 |
| BOEY ET AL., NEUROPEPTIDES, vol. 42, 2008, pages 19 - 30 |
| BOLLAG ET AL., ENDOCRINOLOGY, vol. 141, 2000, pages 1228 - 1235 |
| BOLLAG ET AL., MOL CELL ENDOCRINOL, vol. 177, 2001, pages 35 - 41 |
| BOSE ET AL., DIABETES, vol. 54, 2005, pages 146 - 151 |
| BRADLEY, J PATHOL, vol. 214, 2008, pages 149 - 160 |
| BRANCATI, F. L. ET AL., ARC.H. LNTEM. MED., vol. 159, 1999, pages 957 - 963 |
| CALDWELL ET AL., BIOORG. MED.CHEM. LETT., vol. 14, 2004, pages 1265 - 1268 |
| CHAUDHRI ET AL., ANNU REV PHYSIOL, vol. 70, 2008, pages 239 - 255 |
| COLLIER, T. L., J. LABELLED COMPD. RADIOPHARM., vol. 42, 1999, pages S264 - S266 |
| COX, PEPTIDES, vol. 28, 2007, pages 345 - 351 |
| CRUZE ET AL., PEPTIDES, vol. 28, 2007, pages 269 - 280 |
| DEACON ET AL., J CLIN ENDOCRINOL METAB, vol. 85, 2000, pages 3575 - 3581 |
| DEACON, REGUL PEPT, vol. 128, 2005, pages 117 - 124 |
| DRUCKER, CELL METAB, vol. 3, 2006, pages 153 - 165 |
| DURING ET AL., NAT. MED., vol. 9, 2003, pages 1173 - 1179 |
| EBERLEIN ET AL., PEPTIDES, vol. 10, 1989, pages 797 - 803 |
| EDMONDSON ET AL., BIOORG. MED. CHEM. LETT., vol. 14, 2004, pages 5151 - 5155 |
| EDWARD B. ROCHE: "Biureversible Carriers in Drug Design", 1987, AMERICAN PHARMACEUTICAL ASSOCIATION AND PERGAMON PRESS |
| EKBLAD ET AL., PEPTIDES, vol. 23, 2002, pages 251 - 261 |
| EKSTRAND ET AL., PNAS USA, vol. 100, 2003, pages 6033 - 6038 |
| EL BAHH ET AL., EUR. J. NEUROSCI., vol. 22, 2005, pages 1417 - 1430 |
| GOMEZ ET AL., AM. J. PHYSIOL., vol. 268, 1995, pages G71 - G81 |
| GONON ET AL., CARDIOVASC RES., vol. 78, 2008, pages 116 - 122 |
| GRANDT ET AL., REGUL. PEPT., vol. 51, 1994, pages 151 - 159 |
| GREENE, T. W., WUTS, P. G. M.: "Protecting Groups in Organic Synthesis, 3 rd Edition", 1999, WILEY |
| GREIG ET AL., ANN N YACAD SCI, vol. 1035, 2004, pages 290 - 315 |
| GRISE ET AL., J. SURG. RES., vol. 82, 1999, pages 151 - 155 |
| GUERRE-MILLO, DIABETES & METABOLISM, vol. 34, 2009, pages 12 - 18 |
| H.-U. DEMUTH: "Type 2 diabetes-therapy with DPP-IV inhibitors", BIOCHIM. BIOPHYS. ACTA, vol. 1751, 2005, pages 33 - 44 |
| HANSMANN, CIRCULATION, vol. 115, 2007, pages 1275 - 1284 |
| HARA ET AL., DIABETES CARE, vol. 29, 2006, pages 1357 - 1362 |
| HARRY G. BRITAIN: "Polymorphism in Pharmaceutical Solids", vol. 95, 1999, MARCEL DEKKER, INC., article K.J. GUILLORY: "Generation of Polymorphs, IIydrates, Solvates, and Amorphous Solids", pages: 202 - 209 |
| HAY ET AL., J CLIN GASTROENTEROL, vol. 14, 1992, pages 309 - 317 |
| HILL, J. 0., SCIENCE, vol. 280, 1998, pages 1371 - 1374 |
| JONES ET AL., ANN. REP. MED. CHEM, vol. 44, 2009, pages 149 - 170 |
| JONES ET AL., ANN. REP. MED. CHEM., vol. 44, 2009, pages 149 - 170 |
| JONES, EXPERT OPIN. THER. PATENTS, vol. 19, no. 10, 2009, pages 1339 - 1359 |
| K. AUGUSTYNS ET AL.: "Inhibitors of proline-specific dipeptidyl peptidases: DPP-IV inhibitors as a novel approach for the treatment of type 2 diabetes", EXPERT OPIN. THER. PATENTS, vol. 15, 2005, pages 1387 - 1407, XP002453370, DOI: doi:10.1517/13543776.15.10.1387 |
| KEIGHLEY ET AL., AILMENT PHARMACOL THER, vol. 18, 2003, pages 66 - 70 |
| KEIRE ET AL., ?M. J. PHYSIOL. GASTROINTEST. LIVER PHYSIOL., vol. 279, 2000, pages G126 - G131 |
| KUBOTA ET AL., J. BIOL. CHEM., vol. 277, 2002, pages 25863 - 25866 |
| LAMB ET AL., J. MED. CHEM., vol. 50, 2007, pages 2264 - 2268 |
| LE BAS, M.-D., J. LABELLED COMPD. RADIVPHARM., vol. 44, 2001, pages S280 - S282 |
| LE STUNFF ET AL., DIABETES, vol. 43, 1989, pages 696 - 702 |
| LEE ET AL., J. CLIN. INVEST., vol. 111, 2003, pages 1853 - 1862 |
| LEE ET AL., PEPTIDES, vol. 24, 2003, pages 99 - 106 |
| LIU ET AL., AM SURG, vol. 62, 1996, pages 232 - 236 |
| LIU ET AL., J. SURG. RE,S., vol. 58, 1995, pages 707 - 712 |
| LIU ET AL., J. SURG. RES., vol. 58, 1995, pages 6 - 11 |
| LIU ET AL., SURGERY, vol. 118, 1995, pages 229 - 236 |
| LUNDBERG ET AL., PNAS USA, vol. 79, 1982, pages 4471 - 4475 |
| MAEDA ET AL., NAT. MED., vol. 8, 2002, pages 731 - 737 |
| MARSO ET AL., DIABETES CARE, 5 February 2008 (2008-02-05) |
| MATSUDA ET AL., J BIOL CHEM, vol. 277, 2002, pages 37487 - 37491 |
| MCFADDEN ET AL., AM. J. SURG., vol. 188, 2004, pages 516 - 519 |
| MCINTOSH ET AL., REGUL PEPT, vol. 128, 2005, pages 159 - 165 |
| MENTLEIN, EXPERT OPIN INVESTIG DRUGS, vol. 14, 2005, pages 57 - 64 |
| MORLEY ET AL., LIFE SCIENCE,S, vol. 41, 1987, pages 2157 - 2165 |
| NAUCK ET AL., DIABETES, vol. 53, no. 3, 2004, pages 5190 - 196 |
| NAUCK, DRUG NEWS PERSPECT, vol. 16, 2003, pages 413 - 422 |
| NIGHTINGALE ET AL., GUT, vol. 39, 1996, pages 267 - 272 |
| NISHIMURA ET AL., CIRCULATION, vol. 117, 2008, pages 216 - 223 |
| O'BRIEN, M., DAILY, B., SCHURRIA, M.: "Assay for DPPIV activity using a homogeneous, luminescent method", CELL NOTES, vol. 11, 2005 |
| OHASHI ET AL., HYPERTENSION, vol. 47, 2006, pages 1108 - 1116 |
| OKADA ET AL., ENDOCRINOLOGY, 1993, pages 180 |
| OKU ET AL., H'B'BS LETTERS, vol. 581, 2007, pages 5029 - 5033 |
| ORTIZ ET AL., JPET, vol. 323, 2007, pages 692 - 700 |
| OUCHI ET AL., CIRCULATION, vol. 100, 1999, pages 2473 - 2476 |
| OUCHI ET AL., CLIN CHIM ACTA, vol. 380, 2007, pages 24 - 30 |
| PARKER ET AL., BR. J. PHARMACOL., vol. 153, 2008, pages 420 - 431 |
| PEARSON, NURSING TIMES, vol. 100, 2004, pages 86 - 90 |
| PEDERSON, P., DIAB. METAB. REV., vol. 5, 1989, pages 505 - 509 |
| PERRY, I. J. ET AL., BMJ, vol. 310, 1995, pages 560 - 564 |
| PETERS ET AL., BIOORG. MED. CHEM. LETT., vol. 14, 2004, pages 1491 - 1493 |
| PITTNER ET AL., INT. J. OBES. RELAT. METAB. DISORD., vol. 28, 2004, pages 963 - 971 |
| RAISZ, J CLIN INVEST, vol. 115, 2005, pages 3318 - 3325 |
| REMINGTON: "The Science and Practice of Pharmacy, 20th Edition", 2000, LIPPINCOTT WILLIAMS & WILKINS |
| RENSHAW ET AL., CURRENT DRUG TARGETS, vol. 6, 2005, pages 171 - 179 |
| RIMOIN, D. L.: "Emery and Rimoin's Principles and Practice of Medical Genetics 3rd Ed.", vol. 1, 1996, pages: 1401 - 1402 |
| ROTWEIN, R. ET AL., N. ENGL. J MED., vol. 308, 1983, pages 65 - 71 |
| RUGGERI, NAT MED, vol. 8, 2002, pages 1227 - 1234 |
| SCHWARTZ, CELL METABOLISM, vol. 11, 2010, pages 445 - 447 |
| SCHWARTZ, CELL METAHNLI,SM, vol. 11, 2010, pages 445 - 447 |
| SHAH, CURRENT OPINION IN DRUG DISCOVERY & DEVELOPMENT, vol. 12, 2009, pages 519 - 532 |
| SHIBATA ET AL., J. BIOL. CHEM., vol. 279, 2004, pages 28670 - 28674 |
| SHIBATA ET AL., J. MOL. CELL CARDIOL., vol. 42, 2007, pages 1065 - 1074 |
| SHIBATA ET AL., NAT MED, vol. 11, 2005, pages 1096 - 1103 |
| SHORE ET AL., J. ALLERGY CLIN. IMMUNOL, vol. 118, 2006, pages 389 - 395 |
| SUMMER ET AL., AM J. PHYSIOL. LUNG CELL MOL. PHYSIOL, 7 March 2008 (2008-03-07) |
| T. HIGUCHI, V. STELLA: "Pro-drugs as Novel Delivery Systems", A.C.S. SYMPOSIUM SERIES, vol. 14 |
| TAO ET AL., CIRCULATION, vol. 115, 2007, pages 1408 - 1416 |
| TATEMOTO ET AL., NATURE, vol. 285, 1980, pages 417 - 418 |
| TILG ET AL., NAT. REV. IMMUNOL., vol. 6, 2006, pages 772 - 783 |
| TSENG ET AL., PEPTIDES, vol. 23, 2002, pages 389 - 395 |
| UENO ET AL., REGUL PEPT, vol. 145, 2008, pages 12 - 16 |
| VILLHAUER ET AL., J. MED. CHEM., vol. 45, 2002, pages 2362 - 2365 |
| VILLHAUER ET AL., J. MED. CHEM., vol. 46, 2003, pages 2774 - 2789 |
| VONA-DAVIS ET AL., PEPTIDES, vol. 28, 2007, pages 334 - 338 |
| WOLDBYE ET AL., NEUROBIOLOGY OF DISEASE, vol. 20, 2005, pages 760 - 772 |
| WORTLEY ET AL., GASTROENTEROL., vol. 133, 2007, pages 1534 - 1543 |
| XIE ET AL., BONE, vol. 37, 2005, pages 759 - 769 |
| YAMAMOTO ET AL., CLINICAL SCIENCE, vol. 103, 2002, pages 137 - 142 |
| YOKOTA ET AL., BLOOD, vol. 96, 2000, pages 1723 - 1732 |
| ZANDER ET AL., LANCET, vol. 359, 2002, pages 824 - 830 |
| ZHONG ET AL., AM J PHYSIOL ENDOCRINOL METAB, vol. 292, 2007, pages E543 - E548 |
| ZHU, G-D., J. ORG. CHEM., vol. 67, 2002, pages 943 - 948 |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8933083B2 (en) | 2003-01-14 | 2015-01-13 | Arena Pharmaceuticals, Inc. | 1,2,3-trisubstituted aryl and heteroaryl derivatives as modulators of metabolism and the prophylaxis and treatment of disorders related thereto such as diabetes and hyperglycemia |
| US8754226B2 (en) | 2010-05-17 | 2014-06-17 | Array Biopharma Inc. | Piperidinyl-substituted lactams as GPR119 modulators |
| EP2576507A1 (en) * | 2010-06-04 | 2013-04-10 | Sanofi | Hexafluoroisopropyl carbamate derivatives, their preparation and their therapeutic application |
| EP2576507B1 (en) * | 2010-06-04 | 2017-04-05 | Sanofi | Hexafluoroisopropyl carbamate derivatives, their preparation and their therapeutic application |
| US10894787B2 (en) | 2010-09-22 | 2021-01-19 | Arena Pharmaceuticals, Inc. | Modulators of the GPR119 receptor and the treatment of disorders related thereto |
| WO2012170702A1 (en) * | 2011-06-08 | 2012-12-13 | Arena Pharmaceuticals, Inc. | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| WO2012170867A1 (en) | 2011-06-09 | 2012-12-13 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of gpr-119 |
| US9422266B2 (en) | 2011-09-30 | 2016-08-23 | Merck Sharp & Dohme Corp. | Substituted cyclopropyl compounds, compositions containing such compounds and methods of treatment |
| WO2013055910A1 (en) * | 2011-10-12 | 2013-04-18 | Arena Pharmaceuticals, Inc. | Modulators of the gpr119 receptor and the treatment of disorders related thereto |
| EP2771000A1 (en) * | 2011-10-24 | 2014-09-03 | Merck Sharp & Dohme Corp. | Substituted piperidinyl compounds useful as gpr119 agonists |
| EP2771000A4 (en) * | 2011-10-24 | 2015-04-08 | Merck Sharp & Dohme | Substituted piperidinyl compounds useful as gpr119 agonists |
| US9018200B2 (en) | 2011-10-24 | 2015-04-28 | Merck Sharp & Dohme Corp. | Substituted piperidinyl compounds useful as GPR119 agonists |
| US9944600B2 (en) | 2012-06-12 | 2018-04-17 | Chong Kun Dang Pharmaceutical Corp. | Piperidine derivatives for GPR119 agonist |
| JP2015522559A (en) * | 2012-06-12 | 2015-08-06 | チョン クン ダン ファーマシューティカル コーポレーション | Piperidine derivatives as GPR119 agonists |
| JP2017105785A (en) * | 2012-06-12 | 2017-06-15 | チョン クン ダン ファーマシューティカル コーポレーション | Piperidine derivative as GPR119 agonist |
| JP2019104741A (en) * | 2012-06-12 | 2019-06-27 | チョン クン ダン ファーマシューティカル コーポレーション | Piperidine derivatives as gpr119 agonist |
| WO2014011926A1 (en) | 2012-07-11 | 2014-01-16 | Elcelyx Therapeutics, Inc. | Compositions comprising statins, biguanides and further agents for reducing cardiometabolic risk |
| WO2014074668A1 (en) | 2012-11-08 | 2014-05-15 | Arena Pharmaceuticals, Inc. | Modulators of gpr119 and the treatment of disorders related thereto |
| WO2014074700A1 (en) * | 2012-11-08 | 2014-05-15 | Arena Pharmaceuticals, Inc. | Antagonists of gpr119 receptor and the treatment of disorders related thereto |
| JP2016538296A (en) * | 2013-11-26 | 2016-12-08 | チョン クン ダン ファーマシューティカル コーポレーション | Amide derivatives as GPR119 agonists |
| US9776987B2 (en) | 2013-11-26 | 2017-10-03 | Chong Kun Dang Pharmaceutical Corp | Amide derivatives for GPR119 agonist |
| US11007175B2 (en) | 2015-01-06 | 2021-05-18 | Arena Pharmaceuticals, Inc. | Methods of treating conditions related to the S1P1 receptor |
| US11884626B2 (en) | 2015-06-22 | 2024-01-30 | Arena Pharmaceuticals, Inc. | Crystalline L-arginine salt of (R)-2-(7-(4-cyclopentyl-3-(trifluoromethyl)benzyloxy)-1,2,3,4-tetrahydrocyclo-penta [b]indol-3-yl)acetic acid(Compound1) for use in S1P1 receptor-associated disorders |
| US11534424B2 (en) | 2017-02-16 | 2022-12-27 | Arena Pharmaceuticals, Inc. | Compounds and methods for treatment of primary biliary cholangitis |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102918027A (en) | 2013-02-06 |
| EP2556056A1 (en) | 2013-02-13 |
| JP2013523819A (en) | 2013-06-17 |
| BR112012025592A2 (en) | 2019-09-24 |
| US20130023494A1 (en) | 2013-01-24 |
| AU2011237775A1 (en) | 2012-11-22 |
| CA2795513A1 (en) | 2011-10-13 |
| MX2012011631A (en) | 2013-01-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20130023494A1 (en) | Modulators of the gpr119 receptor and the treatment of disorders related thereto | |
| US20210340133A1 (en) | Modulators of the gpr119 receptor and the treatment of disorders related thereto | |
| WO2012170702A1 (en) | Modulators of the gpr119 receptor and the treatment of disorders related thereto | |
| US20140066369A1 (en) | Modulators Of The GPR119 Receptor And The Treatment Of Disorders Related Thereto | |
| WO2013055910A1 (en) | Modulators of the gpr119 receptor and the treatment of disorders related thereto | |
| US20140038889A1 (en) | Modulators Of The GPR119 Receptor And The Treatment Of Disorders Related Thereto | |
| US20140018371A1 (en) | Modulators Of The GPR119 Receptor And The Treatment Of Disorders Related Thereto | |
| US20140051714A1 (en) | Modulators Of The GPR119 Receptor And The Treatment Of Disorders Related Thereto | |
| WO2011005929A1 (en) | Piperidine derivative and its use for the treatment of diabets and obesity | |
| EA018703B1 (en) | Compounds and compositions as modulators of gpr119 activity | |
| JP5935154B2 (en) | N-cyclopropyl-N-piperidinylbenzamide as a GPR119 modulator | |
| JP2013507366A (en) | Compounds and compositions as GPR119 activity modulators | |
| JP7106623B2 (en) | Compounds active against nuclear receptors | |
| TW202304891A (en) | Benzo[d][1,3]dioxole derivatives | |
| WO2014074668A1 (en) | Modulators of gpr119 and the treatment of disorders related thereto | |
| EA040124B1 (en) | GPR119 RECEPTOR MODULATORS AND THE TREATMENT OF RELATED DISORDERS | |
| JP2024514826A (en) | 2-((4-((S)-2-(4-chloro-2-fluorophenyl)-2-methylbenzo[D][1,3]dioxol-4-yl)piperidin-1-yl)methyl)-1-(((S)-oxetan-2-yl)methyl)-1H-imidazole derivatives as activators of the GLP1 receptor for the treatment of obesity |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180027577.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11715123 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2795513 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013503841 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13639652 Country of ref document: US Ref document number: MX/A/2012/011631 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 8933/DELNP/2012 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011715123 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2011237775 Country of ref document: AU Date of ref document: 20110405 Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112012025592 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112012025592 Country of ref document: BR Kind code of ref document: A2 Effective date: 20121005 |