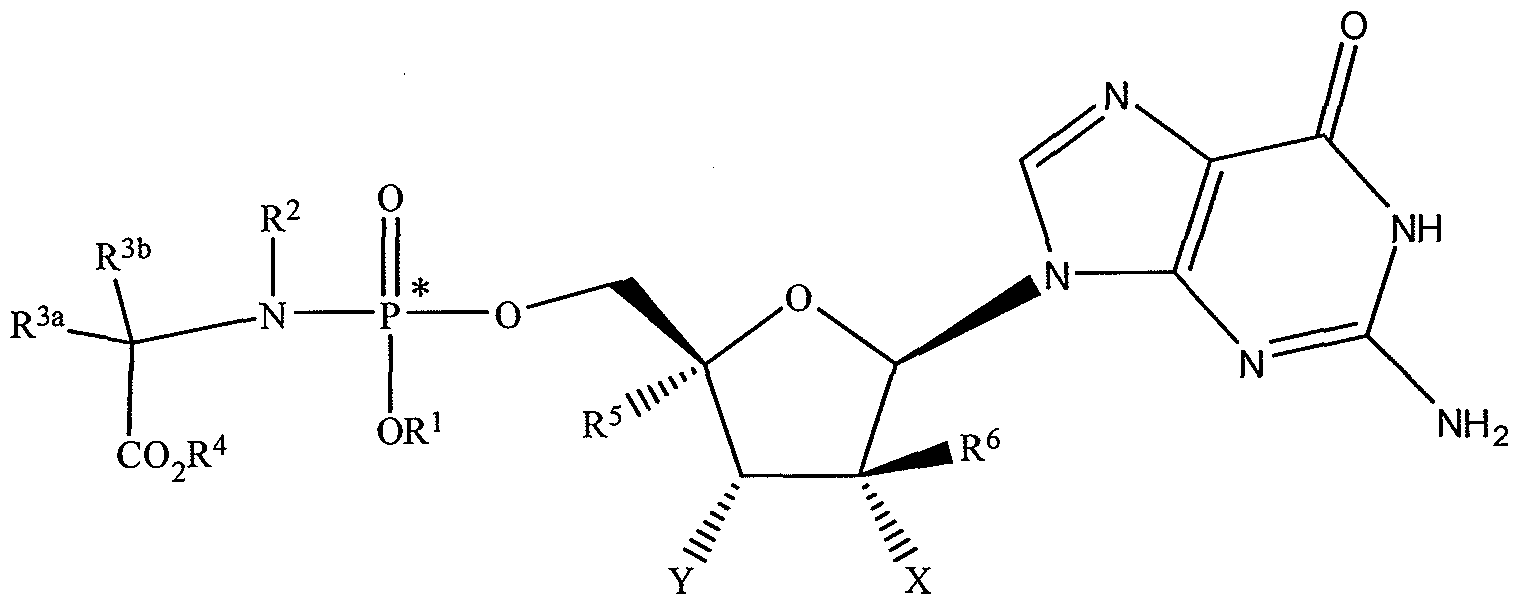
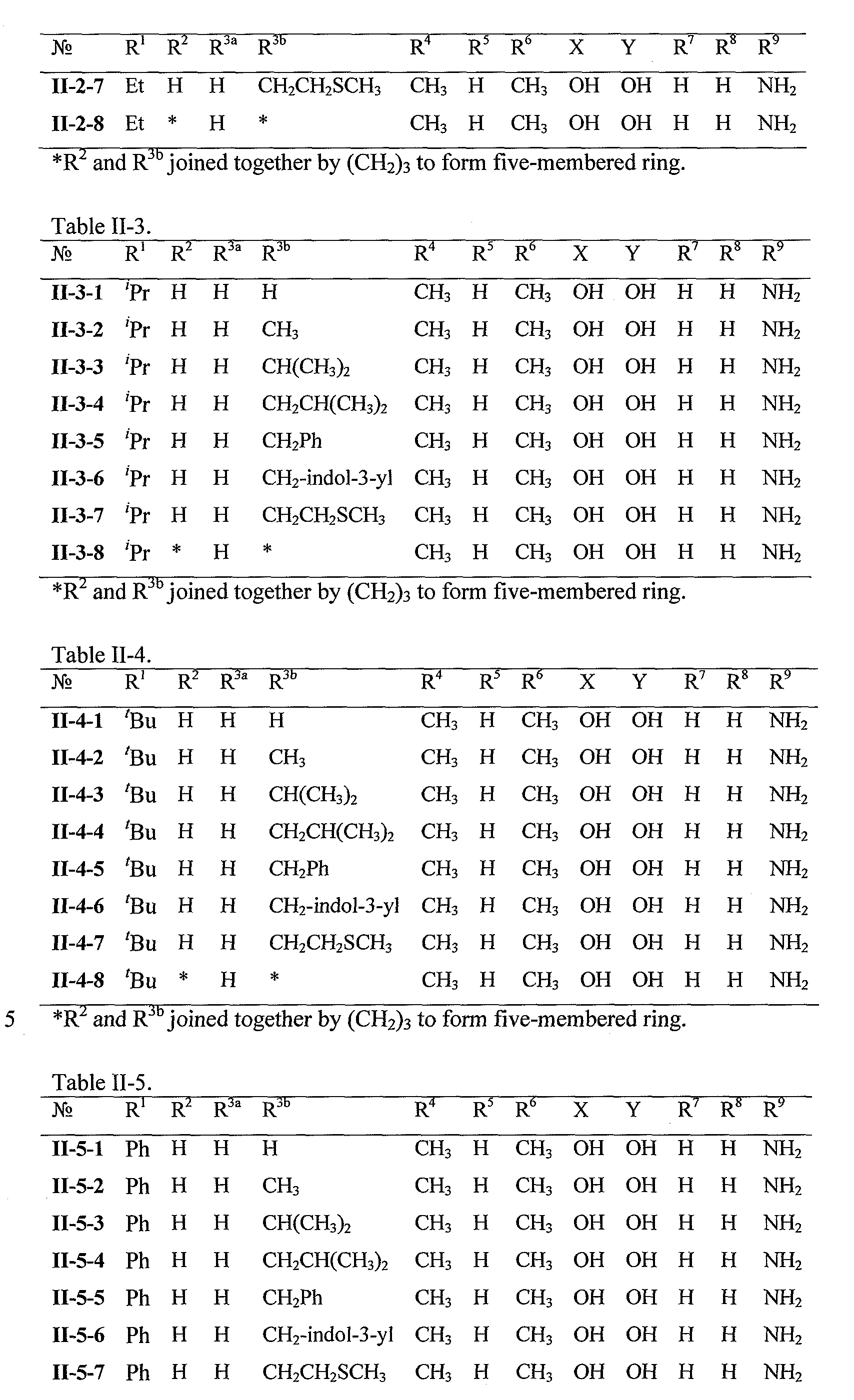
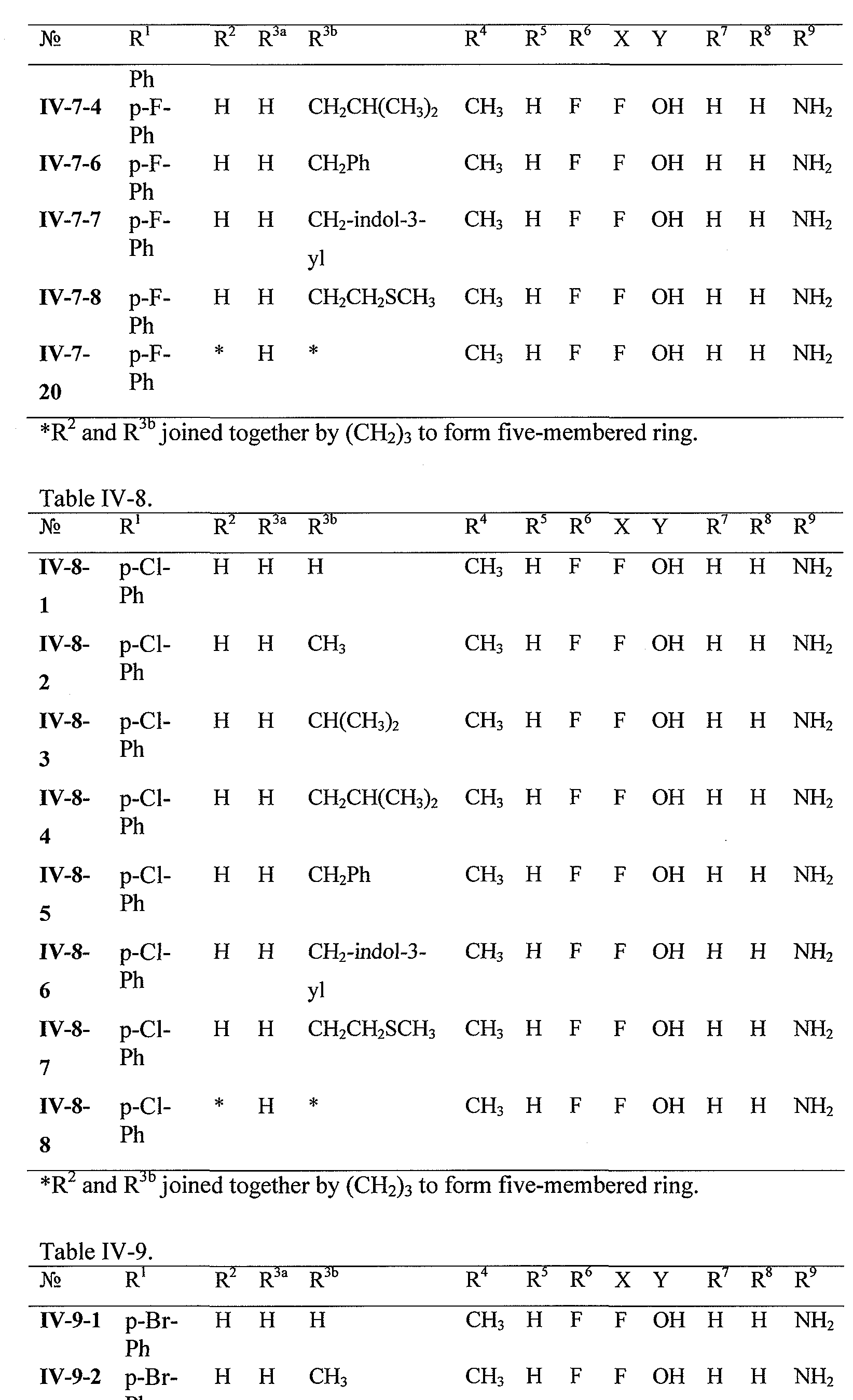
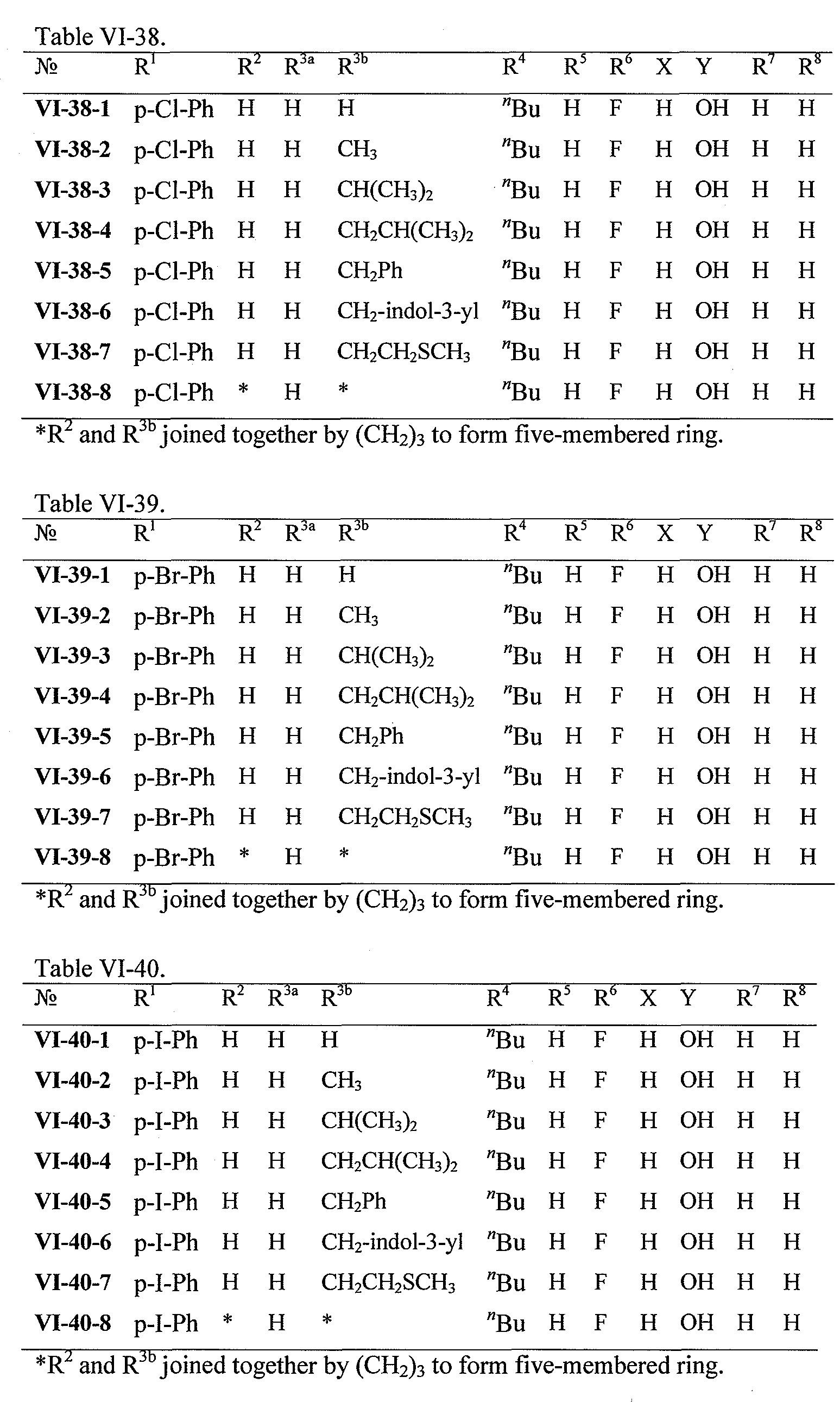
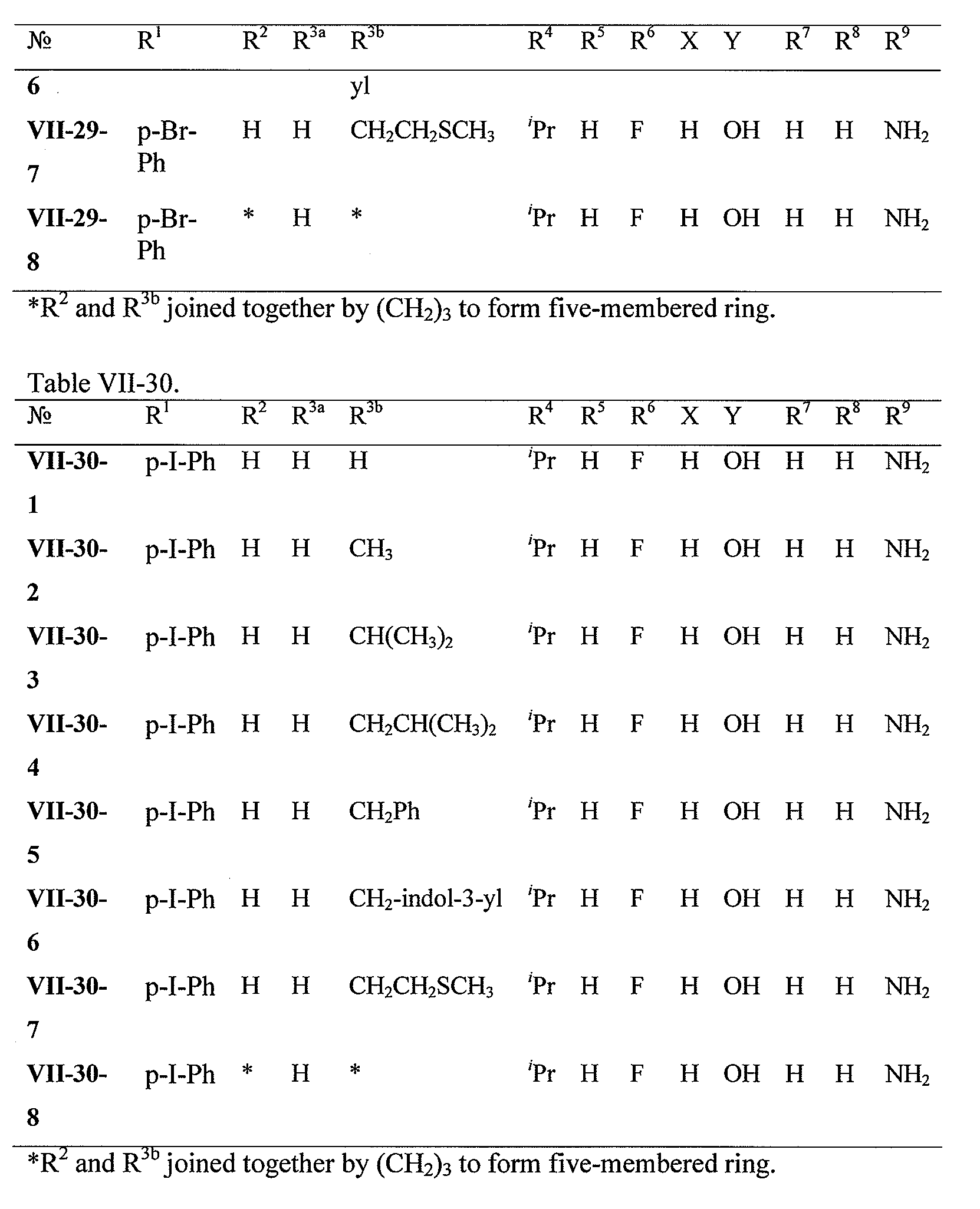
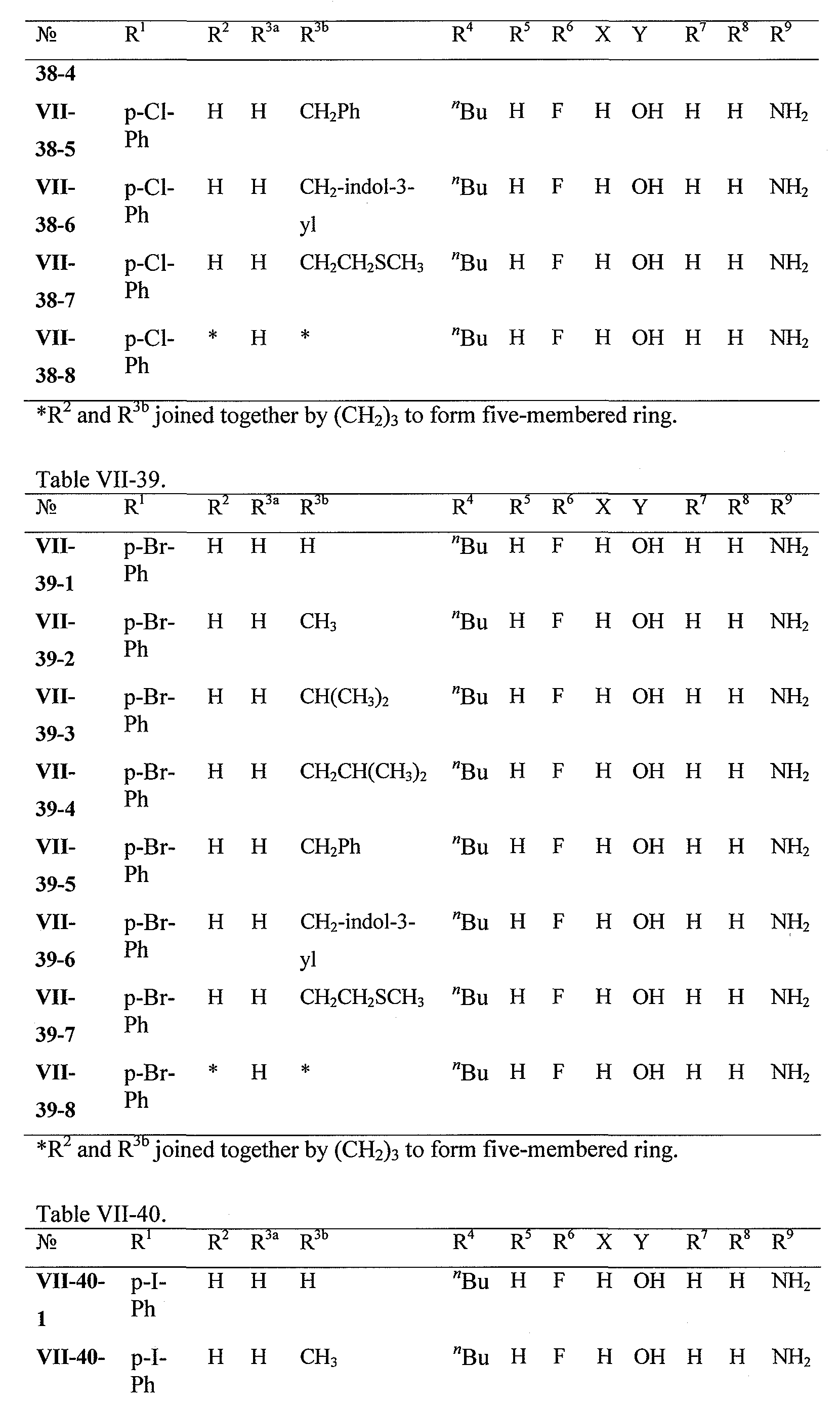
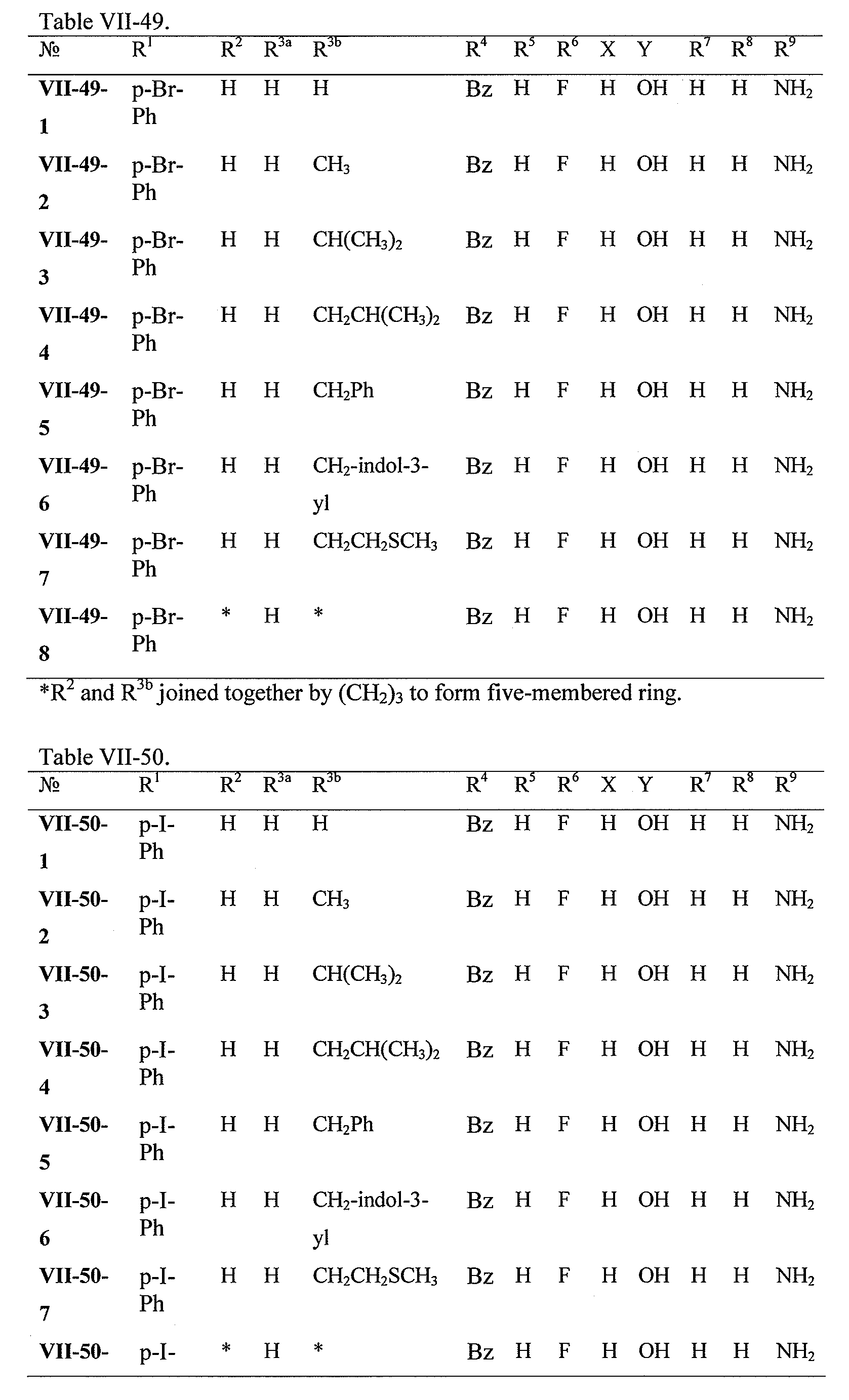
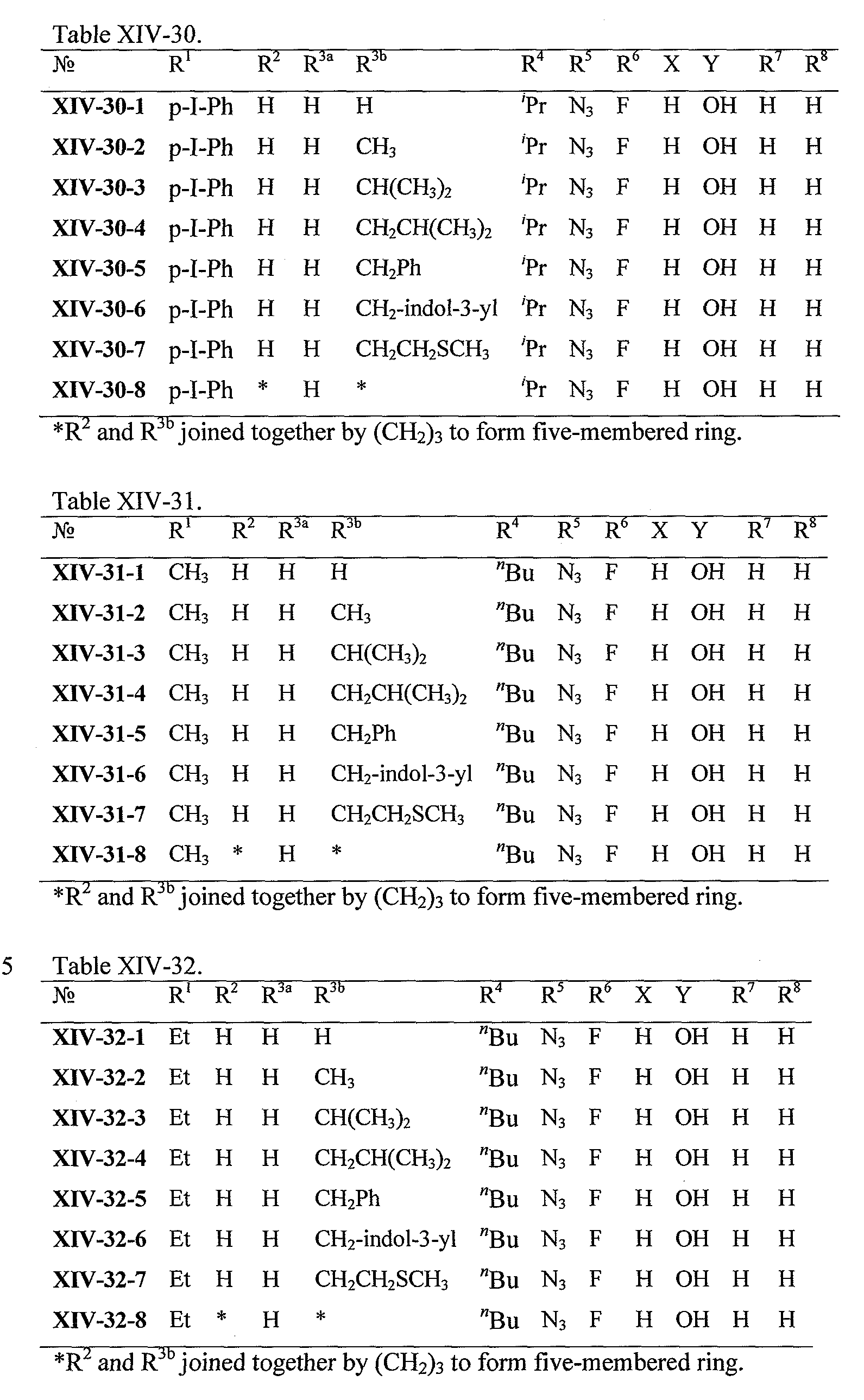
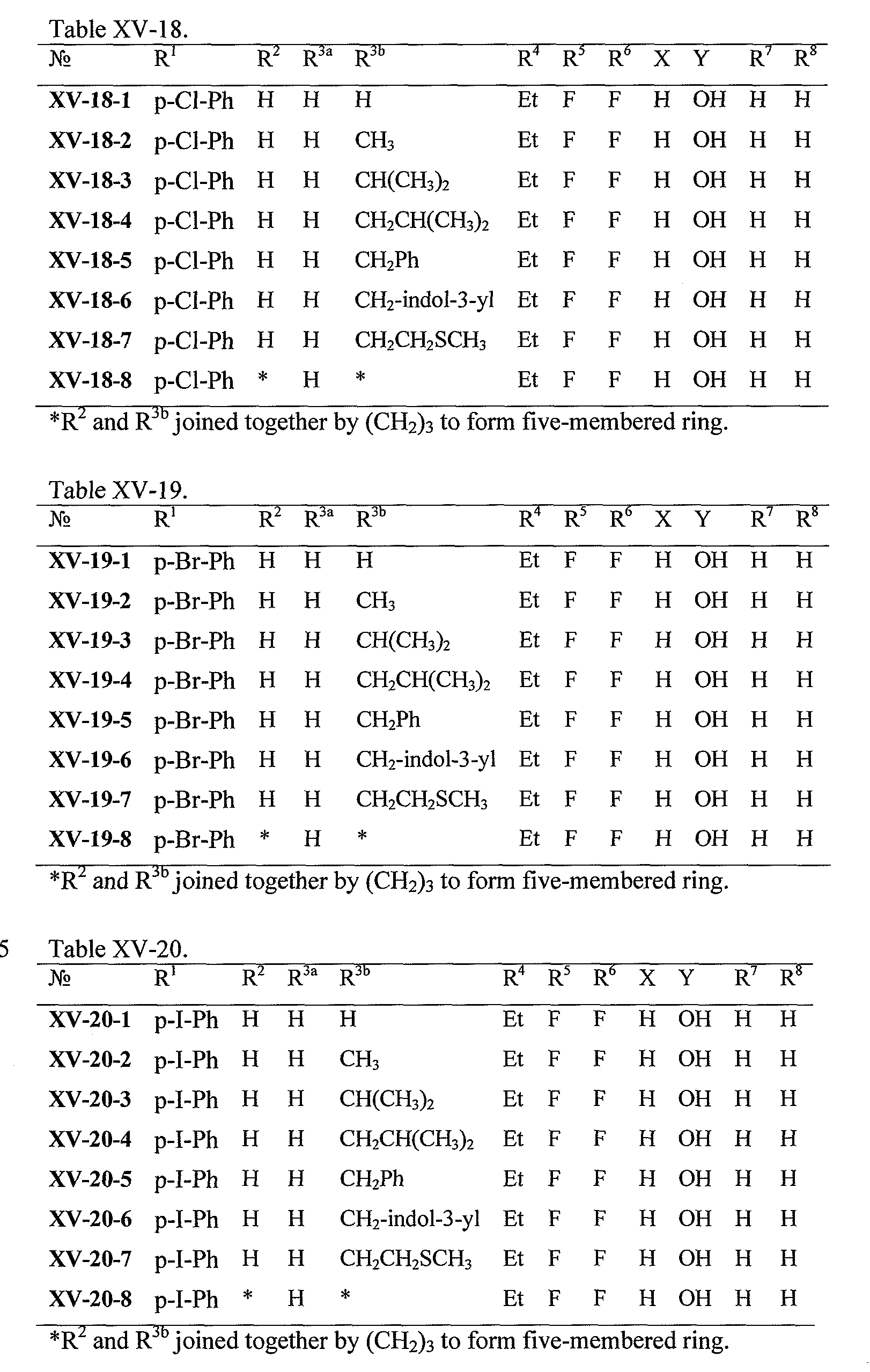
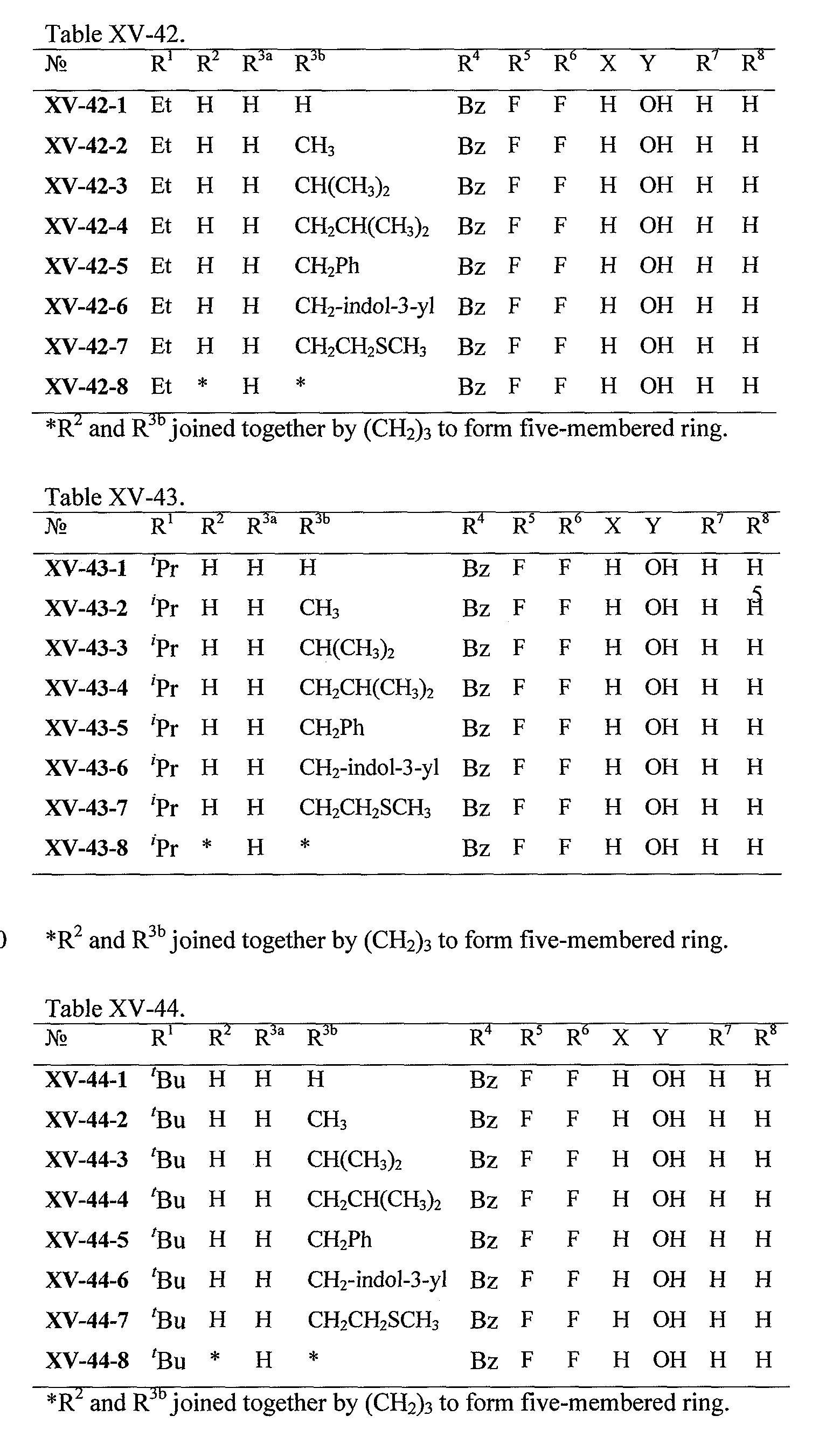
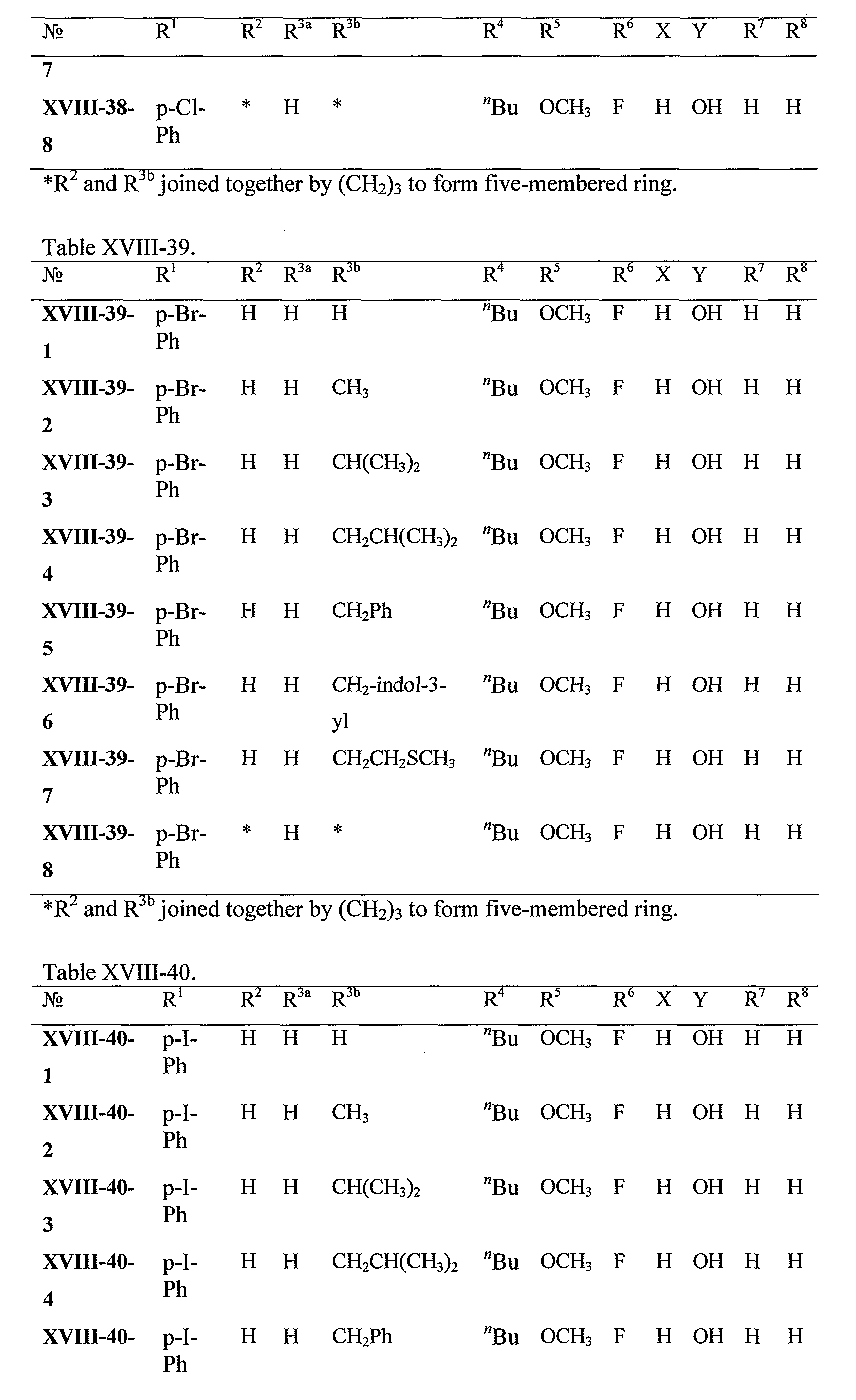
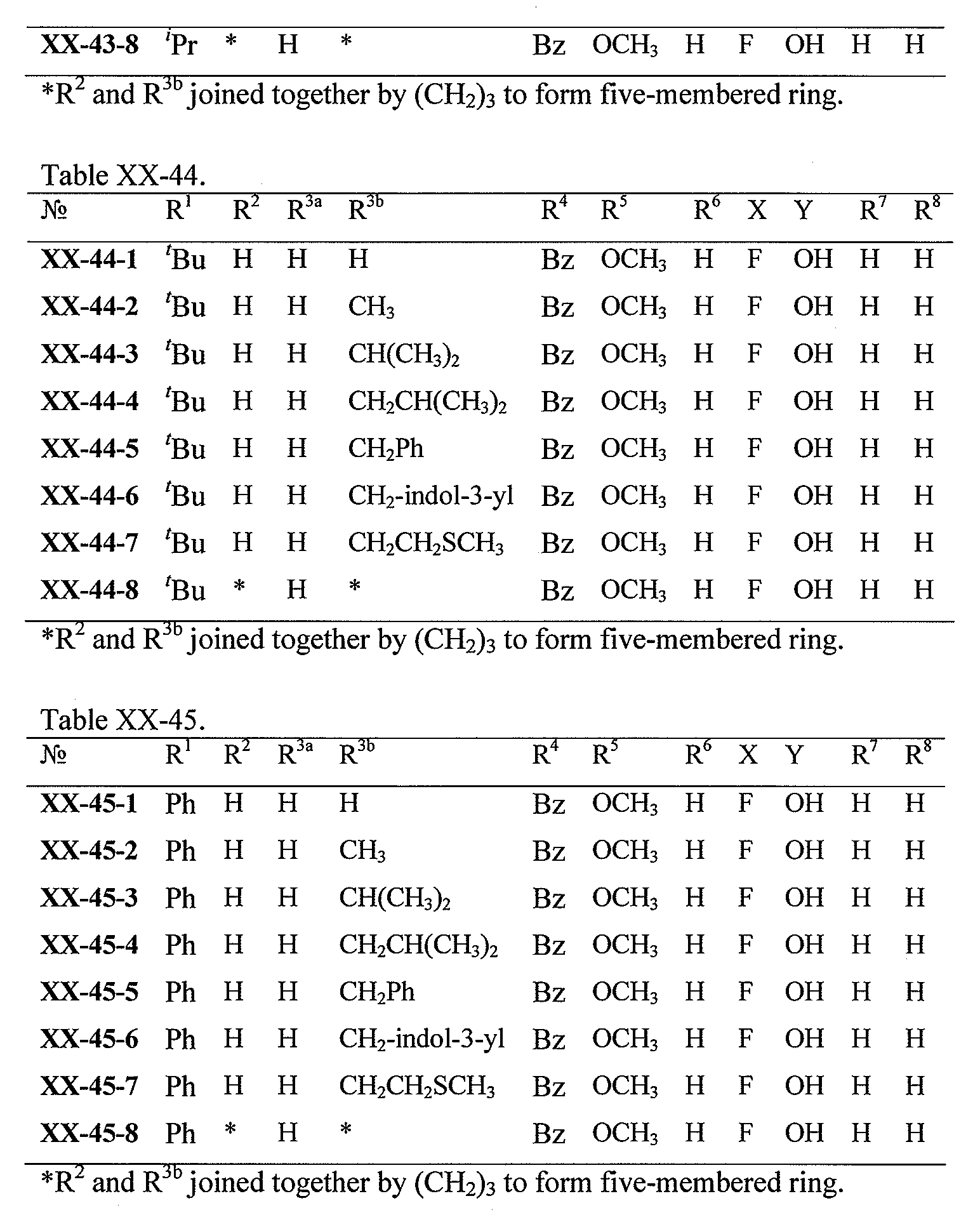
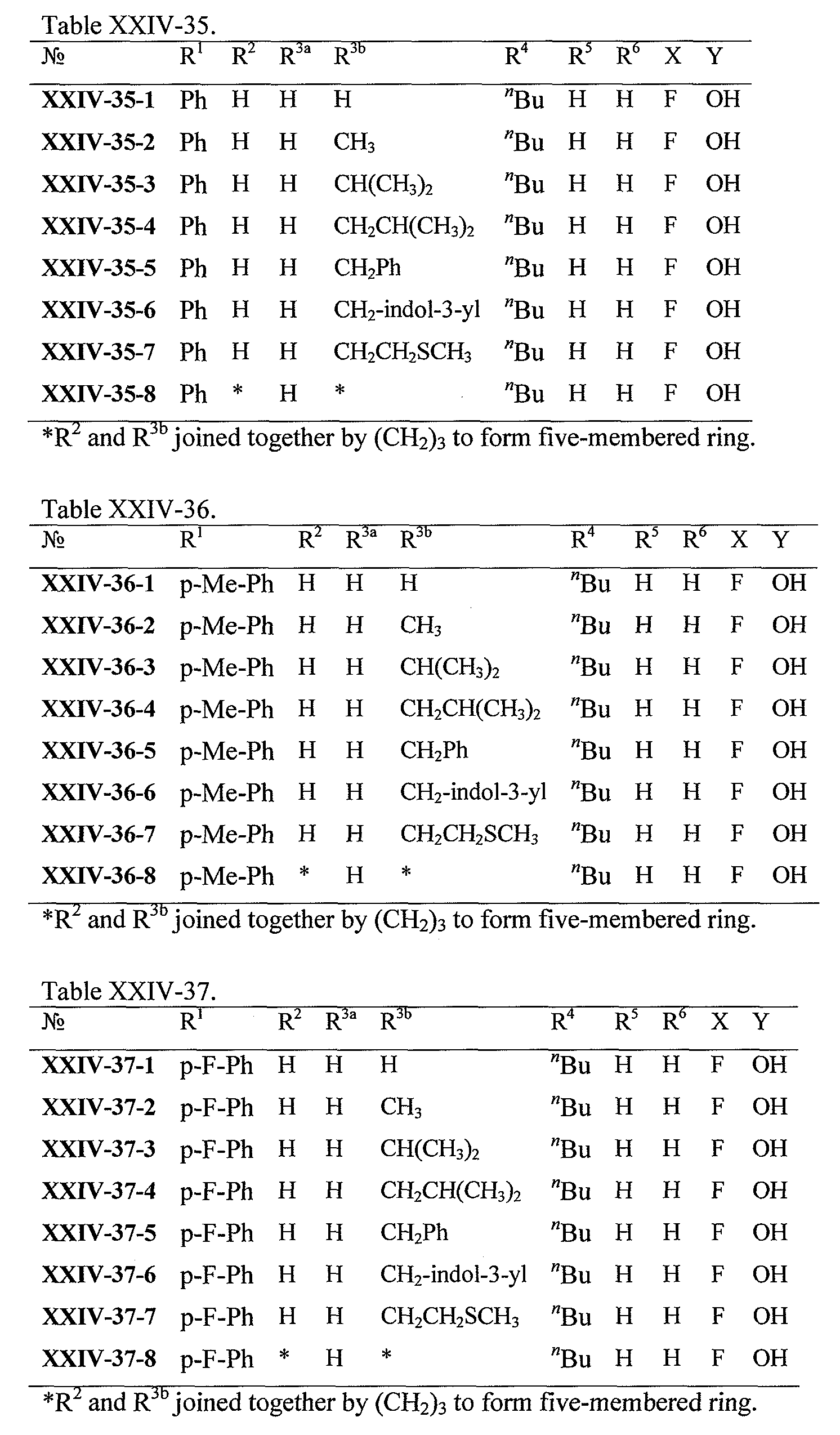
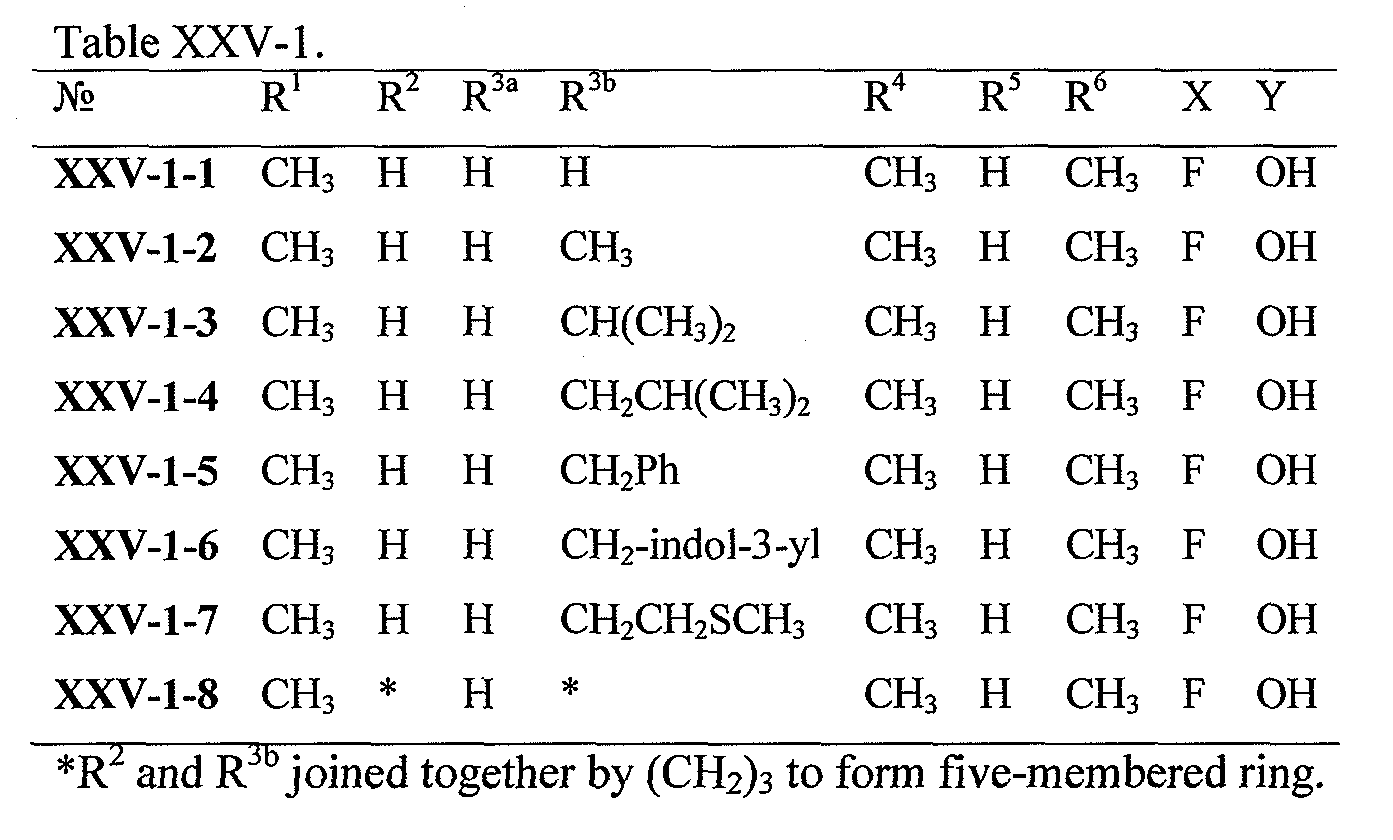
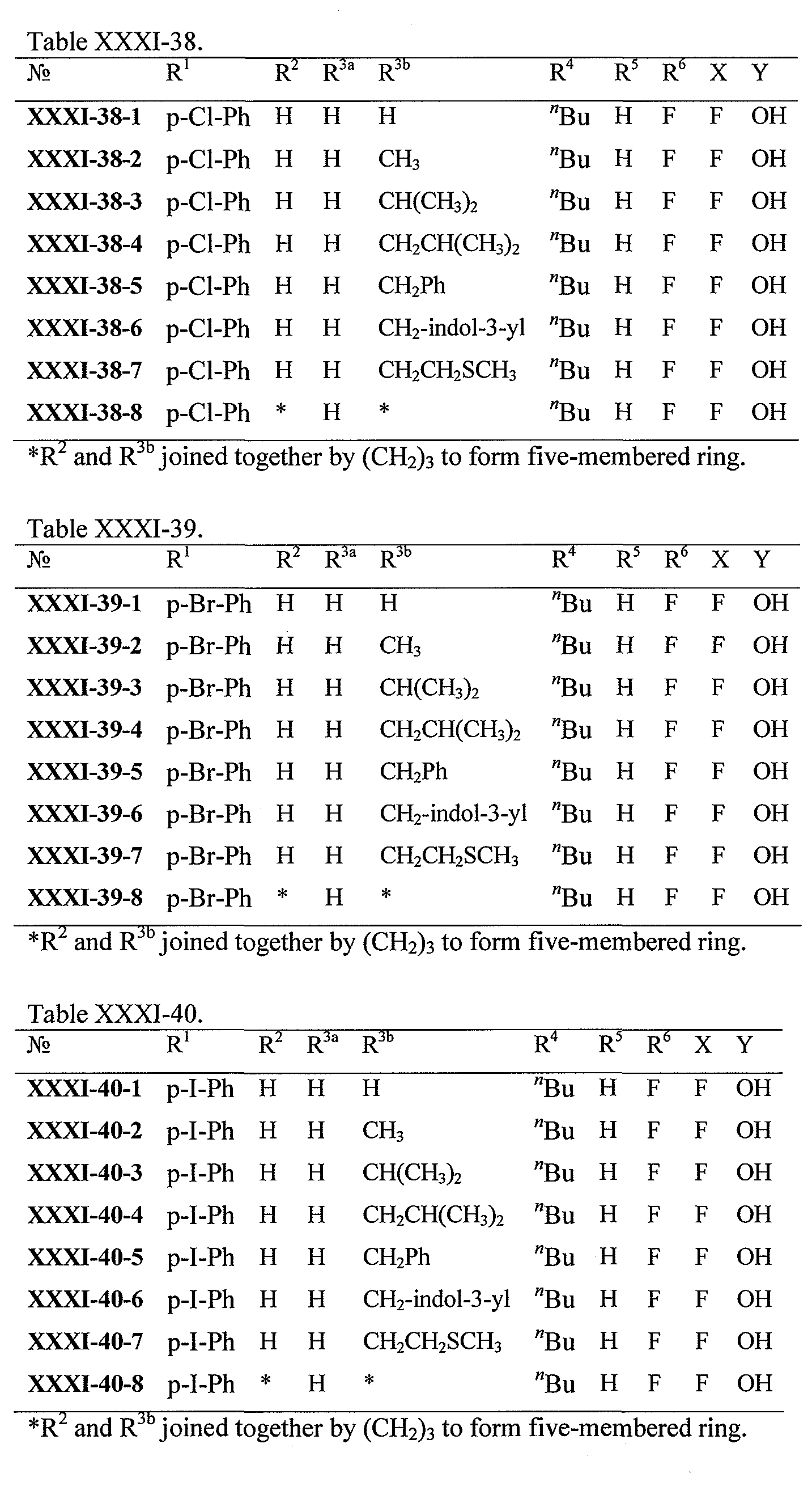
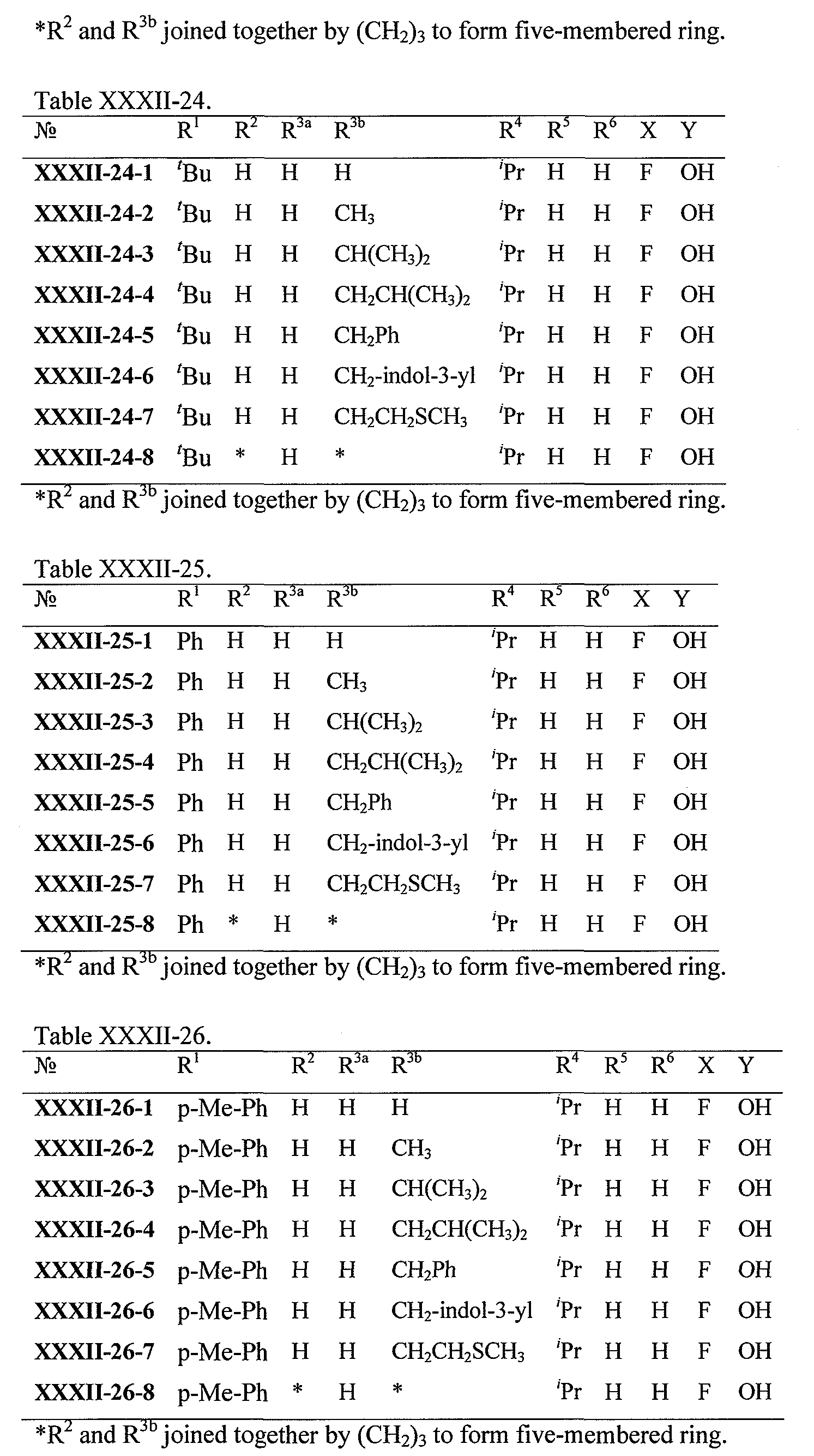
WO2008121634A2 - Nucleoside phosphoramidate prodrugs - Google Patents
Nucleoside phosphoramidate prodrugs Download PDFInfo
- Publication number
- WO2008121634A2 WO2008121634A2 PCT/US2008/058183 US2008058183W WO2008121634A2 WO 2008121634 A2 WO2008121634 A2 WO 2008121634A2 US 2008058183 W US2008058183 W US 2008058183W WO 2008121634 A2 WO2008121634 A2 WO 2008121634A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- fluoro
- hydroxy
- phosphorylamino
- phenoxy
- Prior art date
Links
- -1 Nucleoside phosphoramidate Chemical class 0.000 title claims description 331
- 239000002777 nucleoside Substances 0.000 title claims description 26
- 229940002612 prodrug Drugs 0.000 title abstract description 7
- 239000000651 prodrug Substances 0.000 title abstract description 7
- 150000001875 compounds Chemical class 0.000 claims abstract description 74
- 150000003839 salts Chemical class 0.000 claims abstract description 18
- 150000003833 nucleoside derivatives Chemical class 0.000 claims abstract description 16
- 239000012453 solvate Substances 0.000 claims abstract description 16
- 238000011282 treatment Methods 0.000 claims abstract description 12
- 238000000034 method Methods 0.000 claims abstract 8
- 125000000217 alkyl group Chemical group 0.000 claims description 529
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 369
- 229910052731 fluorine Inorganic materials 0.000 claims description 328
- 229910052794 bromium Inorganic materials 0.000 claims description 311
- 229910052739 hydrogen Inorganic materials 0.000 claims description 311
- 229910052801 chlorine Inorganic materials 0.000 claims description 289
- 229910052740 iodine Inorganic materials 0.000 claims description 249
- 239000001257 hydrogen Substances 0.000 claims description 245
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 228
- 125000006656 (C2-C4) alkenyl group Chemical group 0.000 claims description 223
- 125000006650 (C2-C4) alkynyl group Chemical group 0.000 claims description 165
- 125000002861 (C1-C4) alkanoyl group Chemical group 0.000 claims description 138
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 135
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 claims description 130
- 125000004432 carbon atom Chemical group C* 0.000 claims description 128
- 150000002431 hydrogen Chemical group 0.000 claims description 124
- 229910052736 halogen Inorganic materials 0.000 claims description 121
- 150000002367 halogens Chemical class 0.000 claims description 120
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 116
- VUWZPRWSIVNGKG-UHFFFAOYSA-N fluoromethane Chemical compound F[CH2] VUWZPRWSIVNGKG-UHFFFAOYSA-N 0.000 claims description 113
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 103
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 102
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 98
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 97
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 94
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 91
- 229910004749 OS(O)2 Inorganic materials 0.000 claims description 90
- 125000003342 alkenyl group Chemical group 0.000 claims description 82
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 82
- 229920002554 vinyl polymer Polymers 0.000 claims description 82
- 125000002252 acyl group Chemical group 0.000 claims description 80
- CBOIHMRHGLHBPB-UHFFFAOYSA-N hydroxymethyl Chemical compound O[CH2] CBOIHMRHGLHBPB-UHFFFAOYSA-N 0.000 claims description 80
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 75
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 74
- 125000003118 aryl group Chemical group 0.000 claims description 74
- 125000001188 haloalkyl group Chemical group 0.000 claims description 69
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 claims description 68
- 125000003545 alkoxy group Chemical group 0.000 claims description 67
- 125000000304 alkynyl group Chemical group 0.000 claims description 65
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 claims description 54
- 229910052757 nitrogen Inorganic materials 0.000 claims description 54
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 51
- 125000006302 indol-3-yl methyl group Chemical group [H]N1C([H])=C(C2=C([H])C([H])=C([H])C([H])=C12)C([H])([H])* 0.000 claims description 51
- 125000004055 thiomethyl group Chemical group [H]SC([H])([H])* 0.000 claims description 51
- 125000000266 alpha-aminoacyl group Chemical group 0.000 claims description 48
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 48
- JNCMHMUGTWEVOZ-UHFFFAOYSA-N F[CH]F Chemical compound F[CH]F JNCMHMUGTWEVOZ-UHFFFAOYSA-N 0.000 claims description 47
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 47
- 125000004122 cyclic group Chemical group 0.000 claims description 46
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 45
- MDFFNEOEWAXZRQ-UHFFFAOYSA-N aminyl Chemical compound [NH2] MDFFNEOEWAXZRQ-UHFFFAOYSA-N 0.000 claims description 39
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical group NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 claims description 38
- 125000001072 heteroaryl group Chemical group 0.000 claims description 38
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 36
- 125000000623 heterocyclic group Chemical group 0.000 claims description 34
- 125000001624 naphthyl group Chemical group 0.000 claims description 33
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 32
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 30
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 28
- 241000711549 Hepacivirus C Species 0.000 claims description 27
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical group N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 claims description 25
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 claims description 24
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims description 23
- XXJGBENTLXFVFI-UHFFFAOYSA-N 1-amino-methylene Chemical compound N[CH2] XXJGBENTLXFVFI-UHFFFAOYSA-N 0.000 claims description 22
- AQIAIZBHFAKICS-UHFFFAOYSA-N methylaminomethyl Chemical compound [CH2]NC AQIAIZBHFAKICS-UHFFFAOYSA-N 0.000 claims description 22
- VMWJCFLUSKZZDX-UHFFFAOYSA-N n,n-dimethylmethanamine Chemical compound [CH2]N(C)C VMWJCFLUSKZZDX-UHFFFAOYSA-N 0.000 claims description 22
- 125000001316 cycloalkyl alkyl group Chemical group 0.000 claims description 20
- 229940104302 cytosine Drugs 0.000 claims description 19
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 18
- 125000004076 pyridyl group Chemical group 0.000 claims description 18
- 125000003107 substituted aryl group Chemical group 0.000 claims description 18
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 claims description 16
- 125000004442 acylamino group Chemical group 0.000 claims description 14
- 150000001345 alkine derivatives Chemical class 0.000 claims description 14
- JCXJVPUVTGWSNB-UHFFFAOYSA-N nitrogen dioxide Inorganic materials O=[N]=O JCXJVPUVTGWSNB-UHFFFAOYSA-N 0.000 claims description 14
- 125000003003 spiro group Chemical group 0.000 claims description 14
- 125000006376 (C3-C10) cycloalkyl group Chemical group 0.000 claims description 13
- 125000000520 N-substituted aminocarbonyl group Chemical group [*]NC(=O)* 0.000 claims description 12
- 125000006577 C1-C6 hydroxyalkyl group Chemical group 0.000 claims description 11
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 claims description 11
- 125000000547 substituted alkyl group Chemical group 0.000 claims description 11
- 208000015181 infectious disease Diseases 0.000 claims description 10
- 125000004426 substituted alkynyl group Chemical group 0.000 claims description 10
- 125000005346 substituted cycloalkyl group Chemical group 0.000 claims description 10
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 claims description 9
- 150000003212 purines Chemical class 0.000 claims description 9
- 241000710780 Bovine viral diarrhea virus 1 Species 0.000 claims description 7
- 239000003814 drug Substances 0.000 claims description 7
- 239000003795 chemical substances by application Substances 0.000 claims description 6
- 230000003612 virological effect Effects 0.000 claims description 6
- 241000710842 Japanese encephalitis virus Species 0.000 claims description 3
- 241000710772 Yellow fever virus Species 0.000 claims description 3
- 229940051021 yellow-fever virus Drugs 0.000 claims description 3
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims description 2
- ARKKGZQTGXJVKW-VPCXQMTMSA-N 1-[(2r,3r,4r,5r)-3-fluoro-4-hydroxy-5-(hydroxymethyl)-3-methyloxolan-2-yl]pyrimidine-2,4-dione Chemical compound C[C@@]1(F)[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C=C1 ARKKGZQTGXJVKW-VPCXQMTMSA-N 0.000 claims 26
- NYPIRLYMDJMKGW-VPCXQMTMSA-N 4-amino-1-[(2r,3r,4r,5r)-3-fluoro-4-hydroxy-5-(hydroxymethyl)-3-methyloxolan-2-yl]pyrimidin-2-one Chemical compound C[C@@]1(F)[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)N=C(N)C=C1 NYPIRLYMDJMKGW-VPCXQMTMSA-N 0.000 claims 16
- BDZBACWYNJKMNY-SCOGAWTLSA-N CCCCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=CC=C1Cl Chemical compound CCCCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=CC=C1Cl BDZBACWYNJKMNY-SCOGAWTLSA-N 0.000 claims 9
- DUPAZHRIXWOLCW-YZMWRSGDSA-N C([C@H]1O[C@H]([C@]([C@@H]1O)(C)F)N1C=2N=CN=C(C=2N=C1)NCCC)OC1=CC=CC=C1OP(=O)=N[C@@H](C)C(=O)OC Chemical compound C([C@H]1O[C@H]([C@]([C@@H]1O)(C)F)N1C=2N=CN=C(C=2N=C1)NCCC)OC1=CC=CC=C1OP(=O)=N[C@@H](C)C(=O)OC DUPAZHRIXWOLCW-YZMWRSGDSA-N 0.000 claims 6
- FQESRLYGFUVGPA-WBAFMLPDSA-N CC(C)CCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CC(C)CCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 FQESRLYGFUVGPA-WBAFMLPDSA-N 0.000 claims 6
- RCQSIGGBMAPEGP-HVYVWJKCSA-N CC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=C(C)C(OC[C@@H]2[C@H]([C@@](C)(F)[C@H](N3C(NC(=O)C=C3)=O)O2)O)=C1 Chemical compound CC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=C(C)C(OC[C@@H]2[C@H]([C@@](C)(F)[C@H](N3C(NC(=O)C=C3)=O)O2)O)=C1 RCQSIGGBMAPEGP-HVYVWJKCSA-N 0.000 claims 6
- IFOHCQHTSSRCLU-WSOBARQNSA-N CC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 IFOHCQHTSSRCLU-WSOBARQNSA-N 0.000 claims 6
- XVVLESMRYHJYLT-RFQRIQSBSA-N CC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 XVVLESMRYHJYLT-RFQRIQSBSA-N 0.000 claims 6
- HGPLXBBXBDOENH-HVYVWJKCSA-N CC(C)OC(=O)[C@H](CC)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CC(C)OC(=O)[C@H](CC)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 HGPLXBBXBDOENH-HVYVWJKCSA-N 0.000 claims 6
- ZZEKVAJKGTWNAQ-WBAFMLPDSA-N CCCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 ZZEKVAJKGTWNAQ-WBAFMLPDSA-N 0.000 claims 6
- UBZQTBUWRWYPNN-KOECYIFESA-N CCCCOC(=O)[C@@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCCCOC(=O)[C@@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 UBZQTBUWRWYPNN-KOECYIFESA-N 0.000 claims 6
- SHKZANSNKKBWQA-WBAFMLPDSA-N CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(C)C(OC[C@@H]2[C@H]([C@@](C)(F)[C@H](N3C(NC(=O)C=C3)=O)O2)O)=C1 Chemical compound CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(C)C(OC[C@@H]2[C@H]([C@@](C)(F)[C@H](N3C(NC(=O)C=C3)=O)O2)O)=C1 SHKZANSNKKBWQA-WBAFMLPDSA-N 0.000 claims 6
- YMXCXDMMCZIPTD-LDZBRYNHSA-N CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 YMXCXDMMCZIPTD-LDZBRYNHSA-N 0.000 claims 6
- ZVMLEWDXBRNZHB-LNYBLPJFSA-N CCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1Cl Chemical compound CCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1Cl ZVMLEWDXBRNZHB-LNYBLPJFSA-N 0.000 claims 6
- WARCXNDRHZHSSB-AGXWGAAFSA-N CCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 WARCXNDRHZHSSB-AGXWGAAFSA-N 0.000 claims 6
- VKAWEFORVLYFEN-UXHNPPQISA-N COC(=O)[C@H](C(C)C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Br)C=C1 Chemical compound COC(=O)[C@H](C(C)C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Br)C=C1 VKAWEFORVLYFEN-UXHNPPQISA-N 0.000 claims 6
- JNWADQXDGSDGAO-NKFUEYJGSA-N COC(=O)[C@H](C(C)C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound COC(=O)[C@H](C(C)C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 JNWADQXDGSDGAO-NKFUEYJGSA-N 0.000 claims 6
- QWNDHYSAMZFJNQ-JLIQXZNTSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 QWNDHYSAMZFJNQ-JLIQXZNTSA-N 0.000 claims 6
- FMFBRWNGWVRMFX-GYTLQMSYSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC(N)=NC(=C3N=C2)N2CCC2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC(N)=NC(=C3N=C2)N2CCC2)O1 FMFBRWNGWVRMFX-GYTLQMSYSA-N 0.000 claims 6
- WSCRTCPIDGUHEZ-YZMWRSGDSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(=C3N=C2)N2CCC2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(=C3N=C2)N2CCC2)O1 WSCRTCPIDGUHEZ-YZMWRSGDSA-N 0.000 claims 6
- YLYOIOGVOSFDGY-HSYUBHGISA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(N)=C3N=C2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(N)=C3N=C2)O1 YLYOIOGVOSFDGY-HSYUBHGISA-N 0.000 claims 6
- VOTXAYYTTMFMIV-BIWAAWSLSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(NC4CCC4)=C3N=C2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(NC4CCC4)=C3N=C2)O1 VOTXAYYTTMFMIV-BIWAAWSLSA-N 0.000 claims 6
- DHLZPCBLMQUZHR-TXVRXSMZSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(NC4CCCC4)=C3N=C2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(NC4CCCC4)=C3N=C2)O1 DHLZPCBLMQUZHR-TXVRXSMZSA-N 0.000 claims 6
- VNUWVEUIPKYTOC-LDZBRYNHSA-N COC1=CC=C(OP(=O)=N[C@@H](C)C(=O)OC(C)C)C(OC[C@@H]2[C@H]([C@@](C)(F)[C@H](N3C(NC(=O)C=C3)=O)O2)O)=C1 Chemical compound COC1=CC=C(OP(=O)=N[C@@H](C)C(=O)OC(C)C)C(OC[C@@H]2[C@H]([C@@](C)(F)[C@H](N3C(NC(=O)C=C3)=O)O2)O)=C1 VNUWVEUIPKYTOC-LDZBRYNHSA-N 0.000 claims 6
- VFQNQWCLKGJBRM-BZMMVCHVSA-N FC(F)COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound FC(F)COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 VFQNQWCLKGJBRM-BZMMVCHVSA-N 0.000 claims 6
- JIOYWNZWTJDVJP-XKNCWQHRSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=C(C3=CC=CC=C3C=C2)OP(=O)=N[C@@H](C)C(=O)OC)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=C(C3=CC=CC=C3C=C2)OP(=O)=N[C@@H](C)C(=O)OC)C=CC(=O)NC1=O JIOYWNZWTJDVJP-XKNCWQHRSA-N 0.000 claims 6
- BAWWJIHVLYRFKN-CWPHUKAXSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(F)=CC=C2OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(F)=CC=C2OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O BAWWJIHVLYRFKN-CWPHUKAXSA-N 0.000 claims 6
- XXSSJGOUHCBSTL-GENZXHNBSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(F)=CC=C2OP(=O)=N[C@@H](CC)C(=O)OC2CCCCC2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(F)=CC=C2OP(=O)=N[C@@H](CC)C(=O)OC2CCCCC2)C=CC(=O)NC1=O XXSSJGOUHCBSTL-GENZXHNBSA-N 0.000 claims 6
- SGHWDHYFAMOYBZ-FDUYRLPTSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O SGHWDHYFAMOYBZ-FDUYRLPTSA-N 0.000 claims 6
- WJQRLMGSDQNSRZ-TXCNUOAPSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](CC)C(=O)OC2CCCC2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](CC)C(=O)OC2CCCC2)C=CC(=O)NC1=O WJQRLMGSDQNSRZ-TXCNUOAPSA-N 0.000 claims 6
- SGHWDHYFAMOYBZ-VWVFGUNASA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O SGHWDHYFAMOYBZ-VWVFGUNASA-N 0.000 claims 6
- RPNFCMVSCNDSHV-QGTWPWHYSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC=2C(C)=CC=C(C=2)OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC=2C(C)=CC=C(C=2)OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O RPNFCMVSCNDSHV-QGTWPWHYSA-N 0.000 claims 6
- OVWGLVJEHZKRNC-RUAZOORXSA-N O=C([C@H](C)N=P(=O)OC1=CC=C(C=C1OC[C@@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC)OCC1=CC=CC=C1 Chemical compound O=C([C@H](C)N=P(=O)OC1=CC=C(C=C1OC[C@@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC)OCC1=CC=CC=C1 OVWGLVJEHZKRNC-RUAZOORXSA-N 0.000 claims 6
- XBDQKXXYIPTUBI-UHFFFAOYSA-N Propionic acid Substances CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 claims 6
- CRFIKMCLYGGDBH-WXWBDWACSA-N [2-[[(2R,3R,4R,5R)-5-(2-amino-6-oxo-1H-purin-9-yl)-4-fluoro-3-hydroxy-4-methyloxolan-2-yl]methoxy]phenoxy]-[(2S)-1-methoxy-1-oxopropan-2-yl]imino-oxidophosphanium Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC(N)=NC(O)=C3N=C2)O1 CRFIKMCLYGGDBH-WXWBDWACSA-N 0.000 claims 6
- OPMOATVQKBAMRU-INGMEPTHSA-N CC(C)OC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 Chemical compound CC(C)OC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 OPMOATVQKBAMRU-INGMEPTHSA-N 0.000 claims 5
- JIIZCRNZGKHOPG-BJTVNIROSA-N CCC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCC(C)OC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 JIIZCRNZGKHOPG-BJTVNIROSA-N 0.000 claims 5
- AIYHMZUCOYXOEM-SCOGAWTLSA-N CCCCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 Chemical compound CCCCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 AIYHMZUCOYXOEM-SCOGAWTLSA-N 0.000 claims 5
- QETWHEVWPGAKTI-UENURJEQSA-N CCCCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1Cl Chemical compound CCCCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1Cl QETWHEVWPGAKTI-UENURJEQSA-N 0.000 claims 5
- YAZDOTJHYJRYGL-LNYBLPJFSA-N CCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C(Cl)=C1 Chemical compound CCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C(Cl)=C1 YAZDOTJHYJRYGL-LNYBLPJFSA-N 0.000 claims 5
- IMBOULUSWXCYHH-PTMWTUGHSA-N CCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(F)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 IMBOULUSWXCYHH-PTMWTUGHSA-N 0.000 claims 5
- CZLPPJRZYLHGKC-VVHMCBODSA-N COC(=O)CN=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound COC(=O)CN=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 CZLPPJRZYLHGKC-VVHMCBODSA-N 0.000 claims 5
- YMTFFYKACJYOGK-RXIBCSDBSA-N COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Br)C=C1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Br)C=C1 YMTFFYKACJYOGK-RXIBCSDBSA-N 0.000 claims 5
- FQFOGVIZBOMEON-KPTXFZLCSA-N COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C(Cl)=C1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C(Cl)=C1 FQFOGVIZBOMEON-KPTXFZLCSA-N 0.000 claims 5
- JBXVXZFDVGWIFV-KPTXFZLCSA-N COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1Cl Chemical compound COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1Cl JBXVXZFDVGWIFV-KPTXFZLCSA-N 0.000 claims 5
- NOHLZRRJRHFSTN-CDHYSNCASA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 NOHLZRRJRHFSTN-CDHYSNCASA-N 0.000 claims 5
- GUPUEXHYQAAWAP-CWROUZLESA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC(N)=NC(NC4CCC4)=C3N=C2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC(N)=NC(NC4CCC4)=C3N=C2)O1 GUPUEXHYQAAWAP-CWROUZLESA-N 0.000 claims 5
- WEXPAHYSJXHRKF-XUKIZRNXSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC(N)=NC(NC4CCCC4)=C3N=C2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC(N)=NC(NC4CCCC4)=C3N=C2)O1 WEXPAHYSJXHRKF-XUKIZRNXSA-N 0.000 claims 5
- LOQDAAAHUARCIH-AEFBQFJTSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(=C3N=C2)N2CC2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(=C3N=C2)N2CC2)O1 LOQDAAAHUARCIH-AEFBQFJTSA-N 0.000 claims 5
- FXRDXXWITIOTNG-OAKNYHCFSA-N COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(=C3N=C2)N2CCCCC2)O1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C3=NC=NC(=C3N=C2)N2CCCCC2)O1 FXRDXXWITIOTNG-OAKNYHCFSA-N 0.000 claims 5
- MSRFHJCLOMVKFL-AGXWGAAFSA-N COC(=O)[C@H](CC)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound COC(=O)[C@H](CC)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 MSRFHJCLOMVKFL-AGXWGAAFSA-N 0.000 claims 5
- XBFIIITYIAGZIX-QRBJZHLOSA-N N([C@@H](C)C(=O)OCC=1C=CC=CC=1)=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=CC=C1Cl Chemical compound N([C@@H](C)C(=O)OCC=1C=CC=CC=1)=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=CC=C1Cl XBFIIITYIAGZIX-QRBJZHLOSA-N 0.000 claims 5
- ABZZMUCRTKTLHS-BGJJMRAMSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=C(C3=CC=CC=C3C=C2)OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=C(C3=CC=CC=C3C=C2)OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC=CC=2)C=CC(=O)NC1=O ABZZMUCRTKTLHS-BGJJMRAMSA-N 0.000 claims 5
- JKSMVJROHCQZBP-SZCJYMSISA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(Br)=CC=C2OP(=O)=N[C@@H](C)C(=O)OC2CCCCC2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(Br)=CC=C2OP(=O)=N[C@@H](C)C(=O)OC2CCCCC2)C=CC(=O)NC1=O JKSMVJROHCQZBP-SZCJYMSISA-N 0.000 claims 5
- OMHWHWSBOAYSEZ-SZCJYMSISA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(F)=CC=C2OP(=O)=N[C@@H](C)C(=O)OC2CCCCC2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC(F)=CC=C2OP(=O)=N[C@@H](C)C(=O)OC2CCCCC2)C=CC(=O)NC1=O OMHWHWSBOAYSEZ-SZCJYMSISA-N 0.000 claims 5
- JMELFAZAZNPJDW-OVRMDKARSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](C)C(=O)OC2CCCCC2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](C)C(=O)OC2CCCCC2)C=CC(=O)NC1=O JMELFAZAZNPJDW-OVRMDKARSA-N 0.000 claims 5
- KCRZRDIQSDNESC-GENZXHNBSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](CC)C(=O)OC2CCCCC2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](CC)C(=O)OC2CCCCC2)C=CC(=O)NC1=O KCRZRDIQSDNESC-GENZXHNBSA-N 0.000 claims 5
- PHAFKEPCGPSNPS-RDXHDDKDSA-N [2-[[(2R,3R,4R,5R)-5-(3-amino-2-oxo-4H-pyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyloxolan-2-yl]methoxy]phenoxy]-[(2S)-1-methoxy-3-methyl-1-oxobutan-2-yl]imino-oxidophosphanium Chemical compound COC(=O)[C@H](C(C)C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(N(N)CC=C2)=O)O1 PHAFKEPCGPSNPS-RDXHDDKDSA-N 0.000 claims 5
- WXDHRBJOTTXIPB-UXHNPPQISA-N [[(2R,3R,4R,5R)-5-(3-amino-2-oxo-4H-pyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyloxolan-2-yl]-(4-bromophenoxy)methoxy]-[(2S)-1-methoxy-3-methyl-1-oxobutan-2-yl]imino-oxidophosphanium Chemical compound COC(=O)[C@H](C(C)C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(N(N)CC=C2)=O)O1)O)OC1=CC=C(Br)C=C1 WXDHRBJOTTXIPB-UXHNPPQISA-N 0.000 claims 5
- MBDAKGRZCKTFKV-INGMEPTHSA-N CC(C)OC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=CC=C1Cl Chemical compound CC(C)OC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=CC=C1Cl MBDAKGRZCKTFKV-INGMEPTHSA-N 0.000 claims 4
- RVHKSZIFYYPRSW-DVRJWERCSA-N CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(OC)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=C(OC)C=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 RVHKSZIFYYPRSW-DVRJWERCSA-N 0.000 claims 4
- ZNRYONSQVQXDJP-RVMRSRQISA-N CCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 Chemical compound CCOC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 ZNRYONSQVQXDJP-RVMRSRQISA-N 0.000 claims 4
- LBZRKUREAHTTKY-RXIBCSDBSA-N COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 Chemical compound COC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Cl)C=C1 LBZRKUREAHTTKY-RXIBCSDBSA-N 0.000 claims 4
- PYCJKVWYPDPKTE-CWPHUKAXSA-N N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC(F)=CC=2)C=CC(=O)NC1=O Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COC2=CC=CC=C2OP(=O)=N[C@@H](C)C(=O)OCC=2C=CC(F)=CC=2)C=CC(=O)NC1=O PYCJKVWYPDPKTE-CWPHUKAXSA-N 0.000 claims 4
- 238000004519 manufacturing process Methods 0.000 claims 4
- DHWPNTXNFNHSFE-IJDMJUJQSA-N methyl (2s)-1-[[(2r,3r,4r,5r)-5-(2,4-dioxopyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyloxolan-2-yl]methoxy-phenoxyphosphoryl]pyrrolidine-2-carboxylate Chemical compound COC(=O)[C@@H]1CCCN1P(=O)(OC=1C=CC=CC=1)OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 DHWPNTXNFNHSFE-IJDMJUJQSA-N 0.000 claims 4
- UBZQTBUWRWYPNN-HVYVWJKCSA-N CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 Chemical compound CCCCOC(=O)[C@H](C)N=P(=O)OC1=CC=CC=C1OC[C@@H]1[C@@H](O)[C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1 UBZQTBUWRWYPNN-HVYVWJKCSA-N 0.000 claims 3
- AAWWGAODHYFKSU-PDQOIFANSA-N C([C@H]1O[C@H]([C@]([C@@H]1O)(C)F)N1C=2N=C(N)N=C(C=2N=C1)N(CC)CC)OC1=CC=CC=C1OP(=O)=N[C@@H](C)C(=O)OC Chemical compound C([C@H]1O[C@H]([C@]([C@@H]1O)(C)F)N1C=2N=C(N)N=C(C=2N=C1)N(CC)CC)OC1=CC=CC=C1OP(=O)=N[C@@H](C)C(=O)OC AAWWGAODHYFKSU-PDQOIFANSA-N 0.000 claims 2
- ZGDHFKIGFWNEHY-GYTLQMSYSA-N C([C@H]1O[C@H]([C@]([C@@H]1O)(C)F)N1C=2N=C(N)N=C(C=2N=C1)NCCC)OC1=CC=CC=C1OP(=O)=N[C@@H](C)C(=O)OC Chemical compound C([C@H]1O[C@H]([C@]([C@@H]1O)(C)F)N1C=2N=C(N)N=C(C=2N=C1)NCCC)OC1=CC=CC=C1OP(=O)=N[C@@H](C)C(=O)OC ZGDHFKIGFWNEHY-GYTLQMSYSA-N 0.000 claims 2
- URXASQSEFNHSRL-INGMEPTHSA-N CC(C)OC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Br)C=C1 Chemical compound CC(C)OC(=O)[C@H](C)N=P(=O)OC([C@H]1[C@H]([C@@](C)(F)[C@H](N2C(NC(=O)C=C2)=O)O1)O)OC1=CC=C(Br)C=C1 URXASQSEFNHSRL-INGMEPTHSA-N 0.000 claims 2
- 241000725619 Dengue virus Species 0.000 claims 2
- 241000709661 Enterovirus Species 0.000 claims 2
- 241000991587 Enterovirus C Species 0.000 claims 2
- 241000709721 Hepatovirus A Species 0.000 claims 2
- 241000710886 West Nile virus Species 0.000 claims 2
- 239000003085 diluting agent Substances 0.000 claims 2
- 239000000546 pharmaceutical excipient Substances 0.000 claims 2
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims 2
- 238000011321 prophylaxis Methods 0.000 claims 2
- 241000124008 Mammalia Species 0.000 abstract description 3
- PTMHPRAIXMAOOB-UHFFFAOYSA-L phosphoramidate Chemical compound NP([O-])([O-])=O PTMHPRAIXMAOOB-UHFFFAOYSA-L 0.000 abstract description 3
- 208000036142 Viral infection Diseases 0.000 abstract description 2
- 239000002253 acid Substances 0.000 abstract description 2
- 230000009385 viral infection Effects 0.000 abstract description 2
- 229910002092 carbon dioxide Inorganic materials 0.000 description 102
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical compound O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 72
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 37
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 32
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 32
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 32
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 32
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 30
- 125000004800 4-bromophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Br 0.000 description 25
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 25
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 description 25
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 21
- 229910003204 NH2 Inorganic materials 0.000 description 20
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 19
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 18
- 241000710778 Pestivirus Species 0.000 description 14
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 13
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 12
- 241000711557 Hepacivirus Species 0.000 description 12
- 241000700605 Viruses Species 0.000 description 11
- 108090000623 proteins and genes Proteins 0.000 description 10
- 150000001413 amino acids Chemical class 0.000 description 9
- 229910052760 oxygen Inorganic materials 0.000 description 9
- 235000018102 proteins Nutrition 0.000 description 9
- 102000004169 proteins and genes Human genes 0.000 description 9
- 101800001554 RNA-directed RNA polymerase Proteins 0.000 description 8
- 229940024606 amino acid Drugs 0.000 description 8
- 235000001014 amino acid Nutrition 0.000 description 8
- 229910052799 carbon Inorganic materials 0.000 description 8
- 125000005843 halogen group Chemical group 0.000 description 8
- 239000003112 inhibitor Substances 0.000 description 8
- 108700026244 Open Reading Frames Proteins 0.000 description 7
- 201000010099 disease Diseases 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 7
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 6
- 101710144111 Non-structural protein 3 Proteins 0.000 description 6
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 6
- 230000010076 replication Effects 0.000 description 6
- 108060004795 Methyltransferase Proteins 0.000 description 5
- 108010076039 Polyproteins Proteins 0.000 description 5
- 108700008776 hepatitis C virus NS-5 Proteins 0.000 description 5
- 125000003835 nucleoside group Chemical group 0.000 description 5
- 239000001301 oxygen Substances 0.000 description 5
- 238000002560 therapeutic procedure Methods 0.000 description 5
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 4
- 125000002877 alkyl aryl group Chemical group 0.000 description 4
- 125000003710 aryl alkyl group Chemical group 0.000 description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- 210000004027 cell Anatomy 0.000 description 4
- 230000000670 limiting effect Effects 0.000 description 4
- 230000026731 phosphorylation Effects 0.000 description 4
- 238000006366 phosphorylation reaction Methods 0.000 description 4
- 239000001226 triphosphate Substances 0.000 description 4
- 235000011178 triphosphate Nutrition 0.000 description 4
- 230000029812 viral genome replication Effects 0.000 description 4
- 241000283690 Bos taurus Species 0.000 description 3
- 241000710781 Flaviviridae Species 0.000 description 3
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 3
- 101800001020 Non-structural protein 4A Proteins 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- 102000012479 Serine Proteases Human genes 0.000 description 3
- 108010022999 Serine Proteases Proteins 0.000 description 3
- 108010067390 Viral Proteins Proteins 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 125000005842 heteroatom Chemical group 0.000 description 3
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 239000002773 nucleotide Substances 0.000 description 3
- 125000003729 nucleotide group Chemical group 0.000 description 3
- 230000008520 organization Effects 0.000 description 3
- 239000010452 phosphate Substances 0.000 description 3
- 125000004437 phosphorous atom Chemical group 0.000 description 3
- 125000001424 substituent group Chemical group 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- UNXRWKVEANCORM-UHFFFAOYSA-N triphosphoric acid Chemical compound OP(O)(=O)OP(O)(=O)OP(O)(O)=O UNXRWKVEANCORM-UHFFFAOYSA-N 0.000 description 3
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 2
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 description 2
- 125000006583 (C1-C3) haloalkyl group Chemical group 0.000 description 2
- 125000006274 (C1-C3)alkoxy group Chemical group 0.000 description 2
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- 229930024421 Adenine Natural products 0.000 description 2
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 2
- 239000004475 Arginine Substances 0.000 description 2
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 2
- 241001118702 Border disease virus Species 0.000 description 2
- 241000710777 Classical swine fever virus Species 0.000 description 2
- 101710118188 DNA-binding protein HU-alpha Proteins 0.000 description 2
- 206010012310 Dengue fever Diseases 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 206010016654 Fibrosis Diseases 0.000 description 2
- 241000710831 Flavivirus Species 0.000 description 2
- 241000531123 GB virus C Species 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 102000006992 Interferon-alpha Human genes 0.000 description 2
- 108010047761 Interferon-alpha Proteins 0.000 description 2
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 2
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 2
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 2
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 2
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 2
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 2
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 2
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 2
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 2
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 2
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 2
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 2
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 2
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 2
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 2
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 2
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 2
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 2
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 2
- 239000004472 Lysine Substances 0.000 description 2
- 101710144128 Non-structural protein 2 Proteins 0.000 description 2
- 101800001019 Non-structural protein 4B Proteins 0.000 description 2
- 101800001014 Non-structural protein 5A Proteins 0.000 description 2
- 101710199667 Nuclear export protein Proteins 0.000 description 2
- 241001494479 Pecora Species 0.000 description 2
- 108091005804 Peptidases Proteins 0.000 description 2
- 108091000080 Phosphotransferase Proteins 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 2
- 108030002536 RNA-directed RNA polymerases Proteins 0.000 description 2
- IWUCXVSUMQZMFG-AFCXAGJDSA-N Ribavirin Chemical compound N1=C(C(=O)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 IWUCXVSUMQZMFG-AFCXAGJDSA-N 0.000 description 2
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 2
- 101710172711 Structural protein Proteins 0.000 description 2
- 241000282887 Suidae Species 0.000 description 2
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 2
- 239000004473 Threonine Substances 0.000 description 2
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 2
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical compound O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 2
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 2
- 229960000643 adenine Drugs 0.000 description 2
- 229960003767 alanine Drugs 0.000 description 2
- 235000004279 alanine Nutrition 0.000 description 2
- 125000003282 alkyl amino group Chemical group 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 2
- 229960003121 arginine Drugs 0.000 description 2
- 125000001769 aryl amino group Chemical group 0.000 description 2
- 229960001230 asparagine Drugs 0.000 description 2
- 235000009582 asparagine Nutrition 0.000 description 2
- 229940009098 aspartate Drugs 0.000 description 2
- 125000002837 carbocyclic group Chemical group 0.000 description 2
- 230000001413 cellular effect Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 230000007882 cirrhosis Effects 0.000 description 2
- 208000019425 cirrhosis of liver Diseases 0.000 description 2
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 2
- 229960002433 cysteine Drugs 0.000 description 2
- 235000018417 cysteine Nutrition 0.000 description 2
- 201000002950 dengue hemorrhagic fever Diseases 0.000 description 2
- VILAVOFMIJHSJA-UHFFFAOYSA-N dicarbon monoxide Chemical compound [C]=C=O VILAVOFMIJHSJA-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 238000009509 drug development Methods 0.000 description 2
- 241001493065 dsRNA viruses Species 0.000 description 2
- 230000002068 genetic effect Effects 0.000 description 2
- 229930195712 glutamate Natural products 0.000 description 2
- 229940049906 glutamate Drugs 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- 235000004554 glutamine Nutrition 0.000 description 2
- 229960002743 glutamine Drugs 0.000 description 2
- UYTPUPDQBNUYGX-UHFFFAOYSA-N guanine Chemical compound O=C1NC(N)=NC2=C1N=CN2 UYTPUPDQBNUYGX-UHFFFAOYSA-N 0.000 description 2
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 229960002885 histidine Drugs 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- 150000004677 hydrates Chemical class 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- FDGQSTZJBFJUBT-UHFFFAOYSA-N hypoxanthine Chemical compound O=C1NC=NC2=C1NC=N2 FDGQSTZJBFJUBT-UHFFFAOYSA-N 0.000 description 2
- 238000009169 immunotherapy Methods 0.000 description 2
- 229960000310 isoleucine Drugs 0.000 description 2
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 2
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 2
- 229960003136 leucine Drugs 0.000 description 2
- 244000144972 livestock Species 0.000 description 2
- 229960003646 lysine Drugs 0.000 description 2
- 230000001404 mediated effect Effects 0.000 description 2
- 229930182817 methionine Natural products 0.000 description 2
- 229960004452 methionine Drugs 0.000 description 2
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 2
- 150000004712 monophosphates Chemical class 0.000 description 2
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 239000002547 new drug Substances 0.000 description 2
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 2
- 108020004707 nucleic acids Proteins 0.000 description 2
- 102000039446 nucleic acids Human genes 0.000 description 2
- 150000007523 nucleic acids Chemical class 0.000 description 2
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 2
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 2
- 229960005190 phenylalanine Drugs 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 102000020233 phosphotransferase Human genes 0.000 description 2
- 125000004193 piperazinyl group Chemical group 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 229960002429 proline Drugs 0.000 description 2
- 235000013930 proline Nutrition 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000006239 protecting group Chemical group 0.000 description 2
- 229960000329 ribavirin Drugs 0.000 description 2
- HZCAHMRRMINHDJ-DBRKOABJSA-N ribavirin Natural products O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1N=CN=C1 HZCAHMRRMINHDJ-DBRKOABJSA-N 0.000 description 2
- 229960001153 serine Drugs 0.000 description 2
- 235000004400 serine Nutrition 0.000 description 2
- 230000000405 serological effect Effects 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- 229960002898 threonine Drugs 0.000 description 2
- 235000008521 threonine Nutrition 0.000 description 2
- RWQNBRDOKXIBIV-UHFFFAOYSA-N thymine Chemical compound CC1=CNC(=O)NC1=O RWQNBRDOKXIBIV-UHFFFAOYSA-N 0.000 description 2
- 125000002221 trityl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C([*])(C1=C(C(=C(C(=C1[H])[H])[H])[H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- 229960004799 tryptophan Drugs 0.000 description 2
- 229960004441 tyrosine Drugs 0.000 description 2
- 235000002374 tyrosine Nutrition 0.000 description 2
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 2
- 229960004295 valine Drugs 0.000 description 2
- 239000004474 valine Substances 0.000 description 2
- JIDDDPVQQUHACU-YFKPBYRVSA-N (2s)-pyrrolidine-2-carbaldehyde Chemical group O=C[C@@H]1CCCN1 JIDDDPVQQUHACU-YFKPBYRVSA-N 0.000 description 1
- 125000000027 (C1-C10) alkoxy group Chemical group 0.000 description 1
- 125000006272 (C3-C7) cycloalkyl group Chemical group 0.000 description 1
- FYADHXFMURLYQI-UHFFFAOYSA-N 1,2,4-triazine Chemical compound C1=CN=NC=N1 FYADHXFMURLYQI-UHFFFAOYSA-N 0.000 description 1
- 125000006083 1-bromoethyl group Chemical group 0.000 description 1
- 125000004972 1-butynyl group Chemical group [H]C([H])([H])C([H])([H])C#C* 0.000 description 1
- 125000001478 1-chloroethyl group Chemical group [H]C([H])([H])C([H])(Cl)* 0.000 description 1
- 125000004776 1-fluoroethyl group Chemical group [H]C([H])([H])C([H])(F)* 0.000 description 1
- HNEGJTWNOOWEMH-UHFFFAOYSA-N 1-fluoropropane Chemical group [CH2]CCF HNEGJTWNOOWEMH-UHFFFAOYSA-N 0.000 description 1
- UUFQTNFCRMXOAE-UHFFFAOYSA-N 1-methylmethylene Chemical compound C[CH] UUFQTNFCRMXOAE-UHFFFAOYSA-N 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- 125000000530 1-propynyl group Chemical group [H]C([H])([H])C#C* 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000004778 2,2-difluoroethyl group Chemical group [H]C([H])(*)C([H])(F)F 0.000 description 1
- 125000005999 2-bromoethyl group Chemical group 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 1
- 125000004777 2-fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- SVXNJCYYMRMXNM-UHFFFAOYSA-N 5-amino-2h-1,2,4-triazin-3-one Chemical compound NC=1C=NNC(=O)N=1 SVXNJCYYMRMXNM-UHFFFAOYSA-N 0.000 description 1
- NPYPQKXJJZZSAX-UHFFFAOYSA-N 5-benzylpyrimidine Chemical class C=1N=CN=CC=1CC1=CC=CC=C1 NPYPQKXJJZZSAX-UHFFFAOYSA-N 0.000 description 1
- LZGICTBSBIWVFA-UHFFFAOYSA-N 5-bromo-2-ethenylpyrimidine Chemical compound BrC1=CN=C(C=C)N=C1 LZGICTBSBIWVFA-UHFFFAOYSA-N 0.000 description 1
- HXXVIKZQIFTJOQ-UHFFFAOYSA-N 5-ethenylpyrimidine Chemical compound C=CC1=CN=CN=C1 HXXVIKZQIFTJOQ-UHFFFAOYSA-N 0.000 description 1
- DNWRLMRKDSGSPL-UHFFFAOYSA-N 5-iodopyrimidine Chemical compound IC1=CN=CN=C1 DNWRLMRKDSGSPL-UHFFFAOYSA-N 0.000 description 1
- LRSASMSXMSNRBT-UHFFFAOYSA-N 5-methylcytosine Chemical compound CC1=CNC(=O)N=C1N LRSASMSXMSNRBT-UHFFFAOYSA-N 0.000 description 1
- NOYDQGFVFOQSAJ-UHFFFAOYSA-N 5-nitropyrimidine Chemical compound [O-][N+](=O)C1=CN=CN=C1 NOYDQGFVFOQSAJ-UHFFFAOYSA-N 0.000 description 1
- 108091027075 5S-rRNA precursor Proteins 0.000 description 1
- ZKBQDFAWXLTYKS-UHFFFAOYSA-N 6-Chloro-1H-purine Chemical compound ClC1=NC=NC2=C1NC=N2 ZKBQDFAWXLTYKS-UHFFFAOYSA-N 0.000 description 1
- PVRBGBGMDLPYKG-UHFFFAOYSA-N 6-benzyl-7h-purine Chemical compound N=1C=NC=2N=CNC=2C=1CC1=CC=CC=C1 PVRBGBGMDLPYKG-UHFFFAOYSA-N 0.000 description 1
- DBCMWACNZJYUHS-UHFFFAOYSA-N 6-ethenyl-7h-purine Chemical compound C=CC1=NC=NC2=C1NC=N2 DBCMWACNZJYUHS-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- MSSXOMSJDRHRMC-UHFFFAOYSA-N 9H-purine-2,6-diamine Chemical compound NC1=NC(N)=C2NC=NC2=N1 MSSXOMSJDRHRMC-UHFFFAOYSA-N 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- OMSPNIGCBQFQFQ-UHFFFAOYSA-N BrC1=CC=NC(C=C)=N1 Chemical compound BrC1=CC=NC(C=C)=N1 OMSPNIGCBQFQFQ-UHFFFAOYSA-N 0.000 description 1
- 125000006374 C2-C10 alkenyl group Chemical group 0.000 description 1
- JGLMVXWAHNTPRF-CMDGGOBGSA-N CCN1N=C(C)C=C1C(=O)NC1=NC2=CC(=CC(OC)=C2N1C\C=C\CN1C(NC(=O)C2=CC(C)=NN2CC)=NC2=CC(=CC(OCCCN3CCOCC3)=C12)C(N)=O)C(N)=O Chemical compound CCN1N=C(C)C=C1C(=O)NC1=NC2=CC(=CC(OC)=C2N1C\C=C\CN1C(NC(=O)C2=CC(C)=NN2CC)=NC2=CC(=CC(OCCCN3CCOCC3)=C12)C(N)=O)C(N)=O JGLMVXWAHNTPRF-CMDGGOBGSA-N 0.000 description 1
- 208000001490 Dengue Diseases 0.000 description 1
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 1
- 241001337814 Erysiphe glycines Species 0.000 description 1
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 1
- 241000414862 GBV-A-like agents Species 0.000 description 1
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 1
- 206010061192 Haemorrhagic fever Diseases 0.000 description 1
- 208000005176 Hepatitis C Diseases 0.000 description 1
- UGQMRVRMYYASKQ-UHFFFAOYSA-N Hypoxanthine nucleoside Natural products OC1C(O)C(CO)OC1N1C(NC=NC2=O)=C2N=C1 UGQMRVRMYYASKQ-UHFFFAOYSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 108010011356 Nucleoside phosphotransferase Proteins 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- 241000682990 Pegivirus A Species 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 208000004571 Pestivirus Infections Diseases 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical compound OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- 229940123066 Polymerase inhibitor Drugs 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 206010037660 Pyrexia Diseases 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 1
- 101800001838 Serine protease/helicase NS3 Proteins 0.000 description 1
- 208000009714 Severe Dengue Diseases 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 108020000999 Viral RNA Proteins 0.000 description 1
- 208000003152 Yellow Fever Diseases 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 description 1
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 1
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 230000000840 anti-viral effect Effects 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 239000003443 antiviral agent Substances 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 125000005160 aryl oxy alkyl group Chemical group 0.000 description 1
- 125000004104 aryloxy group Chemical group 0.000 description 1
- 125000000613 asparagine group Chemical group N[C@@H](CC(N)=O)C(=O)* 0.000 description 1
- 125000000254 aspartoyl group Chemical group 0.000 description 1
- HONIICLYMWZJFZ-UHFFFAOYSA-N azetidine Chemical compound C1CNC1 HONIICLYMWZJFZ-UHFFFAOYSA-N 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 125000004267 aziridin-2-yl group Chemical group [H]N1C([H])([H])C1([H])* 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000010836 blood and blood product Substances 0.000 description 1
- 125000005997 bromomethyl group Chemical group 0.000 description 1
- 210000004899 c-terminal region Anatomy 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 238000000423 cell based assay Methods 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- LXWYCLOUQZZDBD-LIYNQYRNSA-N csfv Chemical compound C([C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)[C@@H](C)CC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(O)=O)C1=CC=C(O)C=C1 LXWYCLOUQZZDBD-LIYNQYRNSA-N 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 1
- 208000025729 dengue disease Diseases 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 239000001177 diphosphate Substances 0.000 description 1
- XPPKVPWEQAFLFU-UHFFFAOYSA-J diphosphate(4-) Chemical compound [O-]P([O-])(=O)OP([O-])([O-])=O XPPKVPWEQAFLFU-UHFFFAOYSA-J 0.000 description 1
- 235000011180 diphosphates Nutrition 0.000 description 1
- 229940072240 direct acting antivirals Drugs 0.000 description 1
- 229940125371 direct-acting antiviral drugs Drugs 0.000 description 1
- 238000007876 drug discovery Methods 0.000 description 1
- 239000003596 drug target Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 206010014599 encephalitis Diseases 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- XRECTZIEBJDKEO-UHFFFAOYSA-N flucytosine Chemical compound NC1=NC(=O)NC=C1F XRECTZIEBJDKEO-UHFFFAOYSA-N 0.000 description 1
- 229960004413 flucytosine Drugs 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- 229960002949 fluorouracil Drugs 0.000 description 1
- 125000000404 glutamine group Chemical group N[C@@H](CCC(N)=O)C(=O)* 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000005802 health problem Effects 0.000 description 1
- 208000010710 hepatitis C virus infection Diseases 0.000 description 1
- 206010073071 hepatocellular carcinoma Diseases 0.000 description 1
- 231100000844 hepatocellular carcinoma Toxicity 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000031891 intestinal absorption Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 208000019423 liver disease Diseases 0.000 description 1
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- HRDXJKGNWSUIBT-UHFFFAOYSA-N methoxybenzene Chemical group [CH2]OC1=CC=CC=C1 HRDXJKGNWSUIBT-UHFFFAOYSA-N 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- 125000004372 methylthioethyl group Chemical group [H]C([H])([H])SC([H])([H])C([H])([H])* 0.000 description 1
- 239000006225 natural substrate Substances 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- LBQAJLBSGOBDQF-UHFFFAOYSA-N nitro azanylidynemethanesulfonate Chemical compound [O-][N+](=O)OS(=O)(=O)C#N LBQAJLBSGOBDQF-UHFFFAOYSA-N 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 208000025858 pestivirus infectious disease Diseases 0.000 description 1
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 1
- PTMHPRAIXMAOOB-UHFFFAOYSA-N phosphoramidic acid Chemical group NP(O)(O)=O PTMHPRAIXMAOOB-UHFFFAOYSA-N 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000019833 protease Nutrition 0.000 description 1
- 235000019419 proteases Nutrition 0.000 description 1
- 230000004853 protein function Effects 0.000 description 1
- 230000002797 proteolythic effect Effects 0.000 description 1
- FVLAYJRLBLHIPV-UHFFFAOYSA-N pyrimidin-5-amine Chemical compound NC1=CN=CN=C1 FVLAYJRLBLHIPV-UHFFFAOYSA-N 0.000 description 1
- XVIAPHVAGFEFFN-UHFFFAOYSA-N pyrimidine-5-carbonitrile Chemical compound N#CC1=CN=CN=C1 XVIAPHVAGFEFFN-UHFFFAOYSA-N 0.000 description 1
- 238000009790 rate-determining step (RDS) Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 125000000548 ribosyl group Chemical group C1([C@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 150000003871 sulfonates Chemical class 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 238000003419 tautomerization reaction Methods 0.000 description 1
- ILMRJRBKQSSXGY-UHFFFAOYSA-N tert-butyl(dimethyl)silicon Chemical group C[Si](C)C(C)(C)C ILMRJRBKQSSXGY-UHFFFAOYSA-N 0.000 description 1
- 125000001981 tert-butyldimethylsilyl group Chemical group [H]C([H])([H])[Si]([H])(C([H])([H])[H])[*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000000037 tert-butyldiphenylsilyl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1[Si]([H])([*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 125000000341 threoninyl group Chemical group [H]OC([H])(C([H])([H])[H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 229940113082 thymine Drugs 0.000 description 1
- 125000004665 trialkylsilyl group Chemical group 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000002264 triphosphate group Chemical group [H]OP(=O)(O[H])OP(=O)(O[H])OP(=O)(O[H])O* 0.000 description 1
- 125000005454 tryptophanyl group Chemical group 0.000 description 1
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 229940035893 uracil Drugs 0.000 description 1
- 229960005486 vaccine Drugs 0.000 description 1
- 210000002845 virion Anatomy 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
- A61K31/7068—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines having oxo groups directly attached to the pyrimidine ring, e.g. cytidine, cytidylic acid
- A61K31/7072—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines having oxo groups directly attached to the pyrimidine ring, e.g. cytidine, cytidylic acid having two oxo groups directly attached to the pyrimidine ring, e.g. uridine, uridylic acid, thymidine, zidovudine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/16—Purine radicals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
- A61K31/7076—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines containing purines, e.g. adenosine, adenylic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/16—Antivirals for RNA viruses for influenza or rhinoviruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/20—Antivirals for DNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H17/00—Compounds containing heterocyclic radicals directly attached to hetero atoms of saccharide radicals
- C07H17/02—Heterocyclic radicals containing only nitrogen as ring hetero atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/06—Pyrimidine radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/06—Pyrimidine radicals
- C07H19/10—Pyrimidine radicals with the saccharide radical esterified by phosphoric or polyphosphoric acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/16—Purine radicals
- C07H19/20—Purine radicals with the saccharide radical esterified by phosphoric or polyphosphoric acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
- A61K31/7068—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines having oxo groups directly attached to the pyrimidine ring, e.g. cytidine, cytidylic acid
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/06—Pyrimidine radicals
- C07H19/073—Pyrimidine radicals with 2-deoxyribosyl as the saccharide radical
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- the present invention pertains to nucleoside phosphoramidates and their use as agents for treating viral diseases. These compounds are inhibitors of RNA-dependent RNA viral replication and are useful as inhibitors of HCV NS5B polymerase, as inhibitors of HCV replication and for treatment of hepatitis C infection in mammals.
- the invention provides novel chemical compounds, and the use of these compounds alone or in combination with other antiviral agents for treating HCV infection.
- Hepatitis C virus (HCV) infection is a major health problem that leads to chronic liver disease, such as cirrhosis and hepatocellular carcinoma, in a substantial number of infected individuals, estimated to be 2-15% of the world's population.
- chronic liver disease such as cirrhosis and hepatocellular carcinoma
- According to the World Health Organization there are more than 200 million infected individuals worldwide, with at least 3 to 4 million people being infected each year. Once infected, about 20% of people clear the virus, but the rest can harbor HCV the rest of their lives.
- Ten to twenty percent of chronically infected individuals eventually develop liver-destroying cirrhosis or cancer.
- the viral disease is transmitted parenterally by contaminated blood and blood products, contaminated needles, or sexually and vertically from infected mothers or carrier mothers to their offspring.
- Current treatments for HCV infection which are restricted to immunotherapy with recombinant interferon- ⁇ alone or in combination with the nucleoside analog ribavirin, are of limited clinical benefit.
- the HCV virion is an enveloped positive-strand RNA virus with a single oligoribonucleotide genomic sequence of about 9600 bases which encodes a polyprotein of about 3,010 amino acids.
- the protein products of the HCV gene consist of the structural proteins C, El, and E2, and the non-structural proteins NS2, NS3, NS4A and NS4B, and NS5A and NS5B.
- the nonstructural (NS) proteins are believed to provide the catalytic machinery for viral replication.
- the NS3 protease releases NS5B, the RNA-dependent RNA polymerase from the polyprotein chain.
- HCV NS5B polymerase is required for the synthesis of a double-stranded RNA from a single-stranded viral RNA that serves as a template in the replication cycle of HCV. Therefore, NS5B polymerase is considered to be an essential component in the HCV replication complex (K. Ishi, et al, Heptology, 1999, 29: 1227-1235; V. Lohmann, et al., Virology, 1998, 249: 108- 118). Inhibition of HCV NS5B polymerase prevents formation of the double- stranded HCV RNA and therefore constitutes an attractive approach to the development of HCV-specific antiviral therapies.
- HCV belongs to a much larger family of viruses that share many common features.
- the Flaviviridae family of viruses comprises at least three distinct genera: pestiviruses, which cause disease in cattle and pigs; flavivruses, which are the primary cause of diseases such as dengue fever and yellow fever; and hepaciviruses, whose sole member is HCV.
- the flavivirus genus includes more than 68 members separated into groups on the basis of serological relatedness (Calisher et al., J. Gen. Virol, 1993,70,37-43). Clinical symptoms vary and include fever, encephalitis and hemorrhagic fever (Fields Virology, Editors: Fields, B. N., Knipe, D. M., and Howley, P.
- Flaviviruses of global concern that are associated with human disease include the Dengue Hemorrhagic Fever viruses (DHF), yellow fever virus, shock syndrome and Japanese encephalitis virus (Halstead, S. B., Rev. Infect. Dis., 1984, 6, 251-264; Halstead, S. B., Science, 239:476-481, 1988; Monath, T. P., New Eng. J. Med, 1988, 319, 64 1- 643).
- DHF Dengue Hemorrhagic Fever viruses
- Yellow fever virus yellow fever virus
- shock syndrome and Japanese encephalitis virus
- the pestivirus genus includes bovine viral diarrhea virus (BVDV), classical swine fever virus (CSFV, also called hog cholera virus) and border disease virus (BDV) of sheep (Moennig, V. et al. Adv. Vir. Res. 1992, 41, 53- 98). Pestivirus infections of domesticated livestock (cattle, pigs and sheep) cause significant economic losses worldwide. BVDV causes mucosal disease in cattle and is of significant economic importance to the livestock industry (Meyers, G. and Thiel, H.J., Advances in Virus Research, 1996, 47, 53-118; Moennig V., et al, Adv. Vir. Res. 1992, 41, 53-98). Human pestiviruses have not been as extensively characterized as the animal pestiviruses. However, serological surveys indicate considerable pestivirus exposure in humans.
- BVDV bovine viral diarrhea virus
- CSFV classical swine fever virus
- BDV border disease virus
- Pestiviruses and hepaciviruses are closely related virus groups within the Flaviviridae family.
- Other closely related viruses in this family include the GB virus A, GB virus A-like agents, GB virus-B and GB virus-C (also called hepatitis G virus, HGV).
- the hepacivirus group (hepatitis C virus; HCV) consists of a number of closely related but genotypically distinguishable viruses that infect humans. There are at least 6 HCV genotypes and more than 50 subtypes.
- bovine viral diarrhea virus Due to the similarities between pestiviruses and hepaciviruses, combined with the poor ability of hepaciviruses to grow efficiently in cell culture, bovine viral diarrhea virus (BVDV) is often used as a surrogate to study the HCV virus.
- BVDV bovine viral diarrhea virus
- RNA viruses possess a single large open reading frame (ORF) encoding all the viral proteins necessary for virus replication. These proteins are expressed as a polyprotein that is co- and post-translationally processed by both cellular and virus-encoded proteinases to yield the mature viral proteins.
- the viral proteins responsible for the replication of the viral genome RNA are located within approximately the carboxy-terminal. Two- thirds of the ORF are termed nonstructural (NS) proteins.
- NS nonstructural
- the mature nonstructural (NS) proteins in sequential order from the amino-terminus of the nonstructural protein coding region to the carboxy-terminus of the ORF, consist of p7, NS2, NS3, NS4A, NS4B, NS5A, and NS5B.
- the NS proteins of pestiviruses and hepaciviruses share sequence domains that are characteristic of specific protein functions.
- the NS3 proteins of viruses in both groups possess amino acid sequence motifs characteristic of serine proteinases and of helicases (Gorbalenya et al., Nature, 1988, 333, 22; Bazan and Fletterick Virology , 1989,171,637-639; Gorbalenya et al., Nucleic Acid Res., 1989, 17, 3889-3897).
- the NS5B proteins of pestiviruses and hepaciviruses have the motifs characteristic of RNA-directed RNA polymerases (Koonin, E. V. and DoIj a, V.V., Crir. Rev. Biochem. Molec. Biol. 1993, 28, 375-430).
- NS3 serine proteinase is responsible for all proteolytic processing of polyprotein precursors downstream of its position in the ORF (Wiskerchen and Collett, Virology, 1991, 184, 341-350; Bartenschlager et al., J. Virol. 1993, 67, 3835-3844; Eckart et al. Biochem. Biophys. Res. Comm. 1993,192, 399-406; Grakoui et al., J. Virol. 1993, 67, 2832-2843; Grakoui et al., Proc. Natl.
- NS4A protein acts as a cofactor with the NS3 serine protease (Bartenschlager et al., J. Virol. 1994, 68, 5045-5055; Failla et al., J. Virol. 1994, 68, 3753-3760; Xu et al., J. Virol., 1997, 71:53 12-5322).
- the NS3 protein of both viruses also functions as a helicase (Kim et al., Biochem. Biophys. Res. Comm., 1995, 215, 160-166; Jin and Peterson, Arch. Biochem. Biophys., 1995, 323, 47-53; Warrener and Collett, J. Virol. 1995, 69, 1720- 1726).
- the NS5B proteins of pestiviruses and hepaciviruses have the predicted RNA-directed RNA polymerases activity (Behrens et al., EMBO, 1996, 15, 12-22; Lechmann et al., J. Virol, 1997, 71, 8416-8428; Yuan et al., Biochem. Biophys. Res. Comm. 1997, 232, 231-235; Hagedorn, PCT WO 97/12033; Zhong et al, J. Virol., 1998, 72, 9365-9369).
- RNA-dependent RNA polymerase is absolutely essential for replication of the single-stranded, positive sense, RNA genome and this enzyme has elicited significant interest among medicinal chemists.
- Inhibitors of HCV NS5B as potential therapies for HCV infection have been reviewed: Tan, S.-L., et al., Nature Rev. Drug Discov., 2002, 1 , 867-881 ; Walker, M.P. et al., Exp. Opin. Investigational Drugs, 2003, 12, 1269-1280; Ni, Z-J., et al., Current Opinion in Drug Discovery and Development, 2004, 7, 446- 459; Beaulieu, P.
- Nucleoside inhibitors of NS5B polymerase can act either as a non-natural substrate that results in chain termination or as a competitive inhibitor which competes with nucleotide binding to the polymerase.
- the nucleoside analog must be taken up by the cell and converted in vivo to a triphosphate to compete for the polymerase nucleotide binding site. This conversion to the triphosphate is commonly mediated by cellular kinases which imparts additional structural requirements on a potential nucleoside polymerase inhibitor. Unfortunately, this limits the direct evaluation of nucleosides as inhibitors of HCV replication to cell-based assays capable of in situ phosphorylation.
- nucleoside phosphoramidate prodrugs have been shown to be precursors of the active nucleoside triphosphate and to inhibit viral replication when administered to viral infected whole cells (McGuigan, C, et al., J. Med.
- nucleosides are also limiting the utility of nucleosides as viable therapeutic agents. These poor properties can limit the intestinal absorption of an agent and limit uptake into the target tissue or cell.
- prodrugs of nucleosides have been employed. It has been demonstrated that preparation of nucleoside phosphoramidates improves the systemic absorption of a nucleoside and furthermore, the phosphoramidate moiety of these "pronucleotides" is masked with neutral lipophilic groups to obtain a suitable partition coefficient to optimize uptake and transport into the cell dramatically enhancing the intracellular concentration of the nucleoside monophosphate analog relative to administering the parent nucleoside alone. Enzyme-mediated hydrolysis of the phosphate ester moiety produces a nucleoside monophosphate wherein the rate limiting initial phosphorylation is unnecessary.
- the present invention is directed toward novel phosphoramidate prodrugs of nucleoside derivatives for the treatment of viral infections in mammals, which is a compound, its stereoisomers, salts (acid or basic addition salts), hydrates, solvates, or crystalline forms thereof, represented by the following structure:
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1 - 6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R r ) 2 , COR 1 - , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-20 alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1- is -OR' or -N(R )
- R 2 is hydrogen, C I-10 alkyl, R 3a or R 3b and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms, C(O)CR 3a R 3b NHR', where n is 2 to 4 and R 1 , R 3a , and R 3b ;
- R 3a is CH 3 , -CH 2 CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , -CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 - imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH 2 ((4'-OH)-Ph), CH 2 SH, or lower cycloalkyl and R 3b is H, where R 3 is independently hydrogen or alkyl, which
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy, di(lower alkyl)-amino, or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) p OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN,
- the base is a naturally occurring or modified purine or pyrimidine base represented by the following structures:
- Z is N or CR 12 ;
- R 7 , R 8 ,R 9 , R 10 , and R n are independently H, F, Cl, Br, I, OH, OR', SH,
- R' is an optionally substituted alkyl , which includes, but is not limited to, an optionally substituted C 1-20 alkyl, an optionally substituted C 1-10 alkyl, an optionally substituted lower alkyl; an optionally substituted cycloalkyl; an optionally substituted alkynyl of C 2 -C 6 , an optionally substituted lower alkenyl of C 2 -C 6 , or optionally substituted acyl, which includes but is not limited to C(O) alkyl, C(O)(C 1-20 alkyl), C(O)(C 1-10 alkyl), or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms; and
- R 12 is H, halogen (including F, Cl, Br, I), OH, OR', SH, SR', NH 2 , NHR',
- a or “an” entity refers to one or more of that entity; for example, a compound refers to one or more compounds or at least one compound.
- a compound refers to one or more compounds or at least one compound.
- the terms “a” (or “an”), “one or more”, and “at least one” can be used interchangeably herein.
- both R's can be carbon, both R's can be nitrogen, or one R' can be carbon and the other nitrogen.
- alkenyl refers to an unsubstituted hydrocarbon chain radical having from 2 to 10 carbon atoms having one or two olefinic double bonds, preferably one olefinic double bond.
- C 2-N alkenyl refers to an alkenyl comprising 2 to N carbon atoms, where N is an integer having the following values: 3, 4, 5, 6, 7, 8, 9, or 10.
- C 2-10 alkenyl refers to an alkenyl comprising 2 to 10 carbon atoms.
- C 2-4 alkenyl refers to an alkenyl comprising 2 to 4 carbon atoms.
- halogenated alkenyl refers to an alkenyl comprising at least one of F, Cl, Br, and I.
- alkyl refers to an unbranched or branched chain, saturated, monovalent hydrocarbon residue containing 1 to 30 carbon atoms.
- C 1- M alkyl refers to an alkyl comprising 1 to M carbon atoms, where M is an integer having the following values: 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30.
- C 1-4 alkyl refers to an alkyl containing 1 to 4 carbon atoms.
- lower alkyl denotes a straight or branched chain hydrocarbon residue comprising 1 to 6 carbon atoms.
- C 1-20 alkyl refers to an alkyl comprising 1 to 20 carbon atoms.
- C 1 - K ) alkyl refers to an alkyl comprising 1 to 10 carbons.
- alkyl groups include, but are not limited to, lower alkyl groups include methyl, ethyl, propyl, /-propyl, «-butyl, /-butyl, r-butyl or pentyl, isopentyl, neopentyl, hexyl, heptyl, and octyl.
- the term (ar)alkyl or (heteroaryl)alkyl indicate the alkyl group is optionally substituted by an aryl or a heteroaryl group respectively.
- cycloalkyl refers to an unsubstituted or substituted carbocycle, in which the carbocycle contains 3 to 10 carbon atoms; preferably 3 to 8 carbon atoms; more preferably 3 to 6 carbon atoms (i.e., lower cycloalkyls).
- Examples of cycloalkyl groups include, but are not limited to, cyclopropyl, 2- methyl-cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl.
- cycloalkyl alkyl refers to an additionally unsubstituted or substituted alkyl substituted by a lower cycloalkyl.
- cycloalkyl alkyls include, but are not limited to, any one of methyl, ethyl, propyl, i-propyl, n-butyl, i-butyl, t-butyl or pentyl, isopentyl, neopentyl, hexyl, heptyl, and octyl that is substituted with cyclopropyl, 2-methyl-cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl.
- cycloheteroalkyl refers to an unsubstituted or substituted heterocycle, in which the heterocycle contains 2 to 9 carbon atoms; preferably 2 to 7 carbon atoms; more preferably 2 to 5 carbon atoms.
- Examples of cycloheteroalkyls include, but are not limited to, aziridin-2-yl, N-C 1 - 3 -alkyl- aziridin-2-yl, azetidinyl, N-C 1 .
- N-C 1-3 - alkyl-cycloheteroalkyls include, but are not limited to, N-methyl-aziridin-2-yl, N-methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-2-yl, N-methyl-piperidin-3-yl, and N-methyl-piperidin-4-yl.
- R 4 the point of attachment between the cycloheteroalkyl ring carbon and the oxygen occurs at any one of m'
- heterocycle refers to an unsubstituted or substituted heterocycle containing carbon, hydrogen, and at least one of N, O, and S, where the C and N can be trivalent or tetravalent, i.e., sp 2 - or sp 3 -hybridized.
- heterocycles include, but are not limited to, aziridine, azetidine, pyrrolidine, piperidine, imidazole, oxazole, piperazine, etc.
- piperazine as related to R 10 for NR' 2 , the corresponding opposite nitrogen atom of the piperazinyl is substituted by a lower alkyl represented by the following structure:
- the opposite nitrogen of the piperazinyl is substituted by a methyl group.
- halogenated alkyl refers to an unbranched or branched chain alkyl comprising at least one of F, Cl, Br, and I.
- C 1-M haloalkyl refers to an alkyl comprising 1 to M carbon atoms that comprises at least one of F, Cl, Br, and I, where M is an integer having the following values:
- C r3 haloalkyl refers to a haloalkyl comprising 1 to 3 carbons and at least one of F, Cl, Br, and I.
- halogenated lower alkyl refers to a haloalkyl comprising 1 to 3 carbons and at least one of F, Cl, Br, and I.
- lower haloalkyl refers to a haloalkyl comprising 1 to 6 carbon atoms and at least one of F, Cl, Br, and I. Examples include, but are not limited to, fluoromethyl, chloromethyl, bromomethyl, iodomethyl, difluoromethyl, dichloromethyl, dibromomethyl, diiodomethyl, trifluoromethyl, trichloromethyl, tribromomethyl, triiodomethyl, 1 -fluoroethyl, 1-chloroethyl, 1-bromoethyl, 1- iodoethyl, 2-fluoroethyl, 2-chloroethyl, 2-bromoethyl, 2-iodoethyl, 2,2- difluoroethyl, 2,2-dichloroethyl, 2,2-dibromomethyl, 2-2-diiodomethyl, 3- fluoropropyl, 3-ch
- alkynyl refers to an unbranched or branched hydrocarbon chain radical having from 2 to 10 carbon atoms, preferably 2 to 5 carbon atoms, and having one triple bond.
- C 2-N alkynyl refers to an alkynyl comprising 2 to N carbon atoms, where N is an integer having the following values: 3, 4, 5, 6, 7, 8, 9, or 10.
- C C 2-4 alkynyl refers to an alkynyl comprising 2 to 4 carbon atoms.
- C 2-10 alkynyl refers to an alkynyl comprising 2 to 10 carbons. Examples include, but are limited to, ethynyl, 1- propynyl, 2-propynyl, 1-butynyl, 2-butynyl or 3-butynyl.
- halogenated alkynyl refers to an unbranched or branched hydrocarbon chain radical having from 2 to 10 carbon atoms, preferably 2 to 5 carbon atoms, and having one triple bond and at least one of F, Cl, Br, and I.
- cycloalkyl refers to a saturated carbocyclic ring comprising 3 to 8 carbon atoms, i.e. cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl.
- C 3-7 cycloalkyl refers to a cycloalkyl comprising 3 to 7 carbons in the carbocyclic ring.
- alkoxy refers to an -O-alkyl group or an -O-cycloalkyl group, wherein alkyl and cycloalkyl are as defined above.
- -O-alkyl groups include, but are not limited to, methoxy, ethoxy, n-propyloxy, i- propyloxy, n-butyloxy, i-butyloxy, t-butyloxy.
- “Lower alkoxy” as used herein denotes an alkoxy group with a "lower alkyl” group as previously defined.
- C 1-10 alkoxy refers to an-O-alkyl wherein alkyl is C 1 .io.
- Examples of -O-cycloalkyl groups include, but are not limited to, -O-c-propyl, -O-c-butyl, -O-c-pentyl, and -O-c-hexyl.
- halogenated alkoxy refers to an -O-alkyl group in which the alkyl group comprises at least one of F, Cl, Br, and I.
- halogenated lower alkoxy refers to an -O-(lower alkyl) group in which the lower alkyl group comprises at least one of F, Cl, Br, and I.
- amino acid includes naturally occurring and synthetic ⁇ , ⁇ ⁇ or ⁇ amino acids, and includes but is not limited to, amino acids found in proteins, i.e. glycine, alanine, valine, leucine, isoleucine, methionine, phenylalanine, tryptophan, proline, serine, threonine, cysteine, tyrosine, asparagine, glutamine, aspartate, glutamate, lysine, arginine and histidine.
- the amino acid is in the L-configuration.
- the amino acid can be a derivative of alanyl, valinyl, leucinyl, isoleucinyl, prolinyl, phenylalaninyl, tryptophanyl, methioninyl, glycinyl, serinyl, threoninyl, cysteinyl, tyrosinyl, asparaginyl, glutaminyl, aspartoyl, glutaroyl, lysinyl, argininyl, histidinyl, ⁇ -alanyl, ⁇ -valinyl, ⁇ -leucinyl, ⁇ -isoleucinyl, ⁇ -prolinyl, ⁇ - phenylalaninyl, ⁇ -tryptophanyl, ⁇ -methioninyl, ⁇ -glycinyl, ⁇ -serinyl, ⁇ - threoninyl, ⁇ -cystein
- amino acid When the term amino acid is used, it is considered to be a specific and independent disclosure of each of the esters of ⁇ , ⁇ ⁇ or ⁇ glycine, alanine, valine, leucine, isoleucine, methionine, phenylalanine, tryptophan, proline, serine, threonine, cysteine, tyrosine, asparagine, glutamine, aspartate, glutamate, lysine, arginine and histidine in the D and L-configurations.
- aminoacyl includes N,N-unsubstituted, N,N-monosubstituted, and N,N-disubstituted derivatives of naturally occurring and synthetic ⁇ , ⁇ ⁇ or ⁇ amino acyls, where the amino acyls are derived from amino acids.
- the amino- nitrogen can be substituted or unsubstituted. When the amino-nitrogen is substituted, the nitrogen is either mono- or di-substituted, where the substituent bound to the amino-nitrogen is a lower alkyl or an alkaryl.
- the expression "O(aminoacyl)" is used. It is understood that the C3' carbon of the ribose is bound to the oxygen "O", which is then bound to the carbonyl carbon of the aminoacyl.
- alkylamino or arylamino refer to an amino group that has one or two alkyl or aryl substituents, respectively.
- protected refers to a group that is added to an oxygen, nitrogen, or phosphorus atom to prevent its further reaction or for other purposes. A wide variety of oxygen and nitrogen protecting groups are known to those skilled in the art of organic synthesis.
- Non- limiting examples include: C(O)-alkyl, C(O)Ph, C(O)aryl, CH 3 , CH 2 -alkyl, CH 2 - alkenyl, CH 2 Ph, CH 2 -aryl, CH 2 O-alkyl, CH 2 O-aryl, SO 2 -alkyl, SO 2 -aryl, tert- butyldimethylsilyl, tert-butyldiphenylsilyl, and 1,3-(I 9 1,3,3- tetraisopropyldisiloxanylidene).
- aryl refers to substituted or unsubstituted phenyl (Ph), biphenyl, or naphthyl, preferably the term aryl refers to substituted or unsubstituted phenyl.
- the aryl group can be substituted with one or more moieties selected from among hydroxyl, F, Cl, Br, I, amino, alkylamino, arylamino, alkoxy, aryloxy, nitro, cyano, sulfonic acid, sulfate, phosphonic acid, phosphate, and phosphonate, either unprotected, or protected as necessary, as known to those skilled in the art, for example, as taught in T. W. Greene and P.G. M. Wuts, "Protective Groups in Organic Synthesis," 3rd ed., John Wiley & Sons, 1999.
- alkaryl or “alkylaryl” refer to an alkyl group with an aryl substituent, such as benzyl.
- aryl or “arylalkyl” refer to an aryl group with an alkyl substituent.
- di(lower alkyl)amino-lower alkyl refers to a lower alkyl substituted by an amino group that is itself substituted by two lower alkyl groups. Examples include, but are not limited to, (CH 3 ) 2 NCH 2 , (CH 3 ) 2 NCH 2 CH 2 , (CHa) 2 NCH 2 CH 2 CH 2 , etc.
- the examples above show lower alkyls substituted at the terminus carbon atom with an N,N-dimethyl-amino substituent. These are intended as examples only and are not intended to limit the meaning of the term "di(lower alkyl)amino-lower alkyl" so as to require the same.
- the lower alkyl chain can be substituted with an N,N-di(lower alkyl)-amino at any point along the chain, e.g., CH 3 CH(N-(lower alkyl) 2 )CH 2 CH 2 .
- halo includes chloro, bromo, iodo and fluoro.
- acyl refers to a substituent containing a carbonyl moiety and a non-carbonyl moiety. The carbonyl moiety contains a double-bond between the carbonyl carbon and a heteroatom, where the heteroatom is selected from among O, N and S. When the heteroatom is N, the N is substituted by a lower alkyl.
- the non-carbonyl moiety is selected from straight, branched, and cyclic alkyl, which includes, but is not limited to, a straight, branched, or cyclic C 1-20 alkyl, C 1-10 alkyl, or lower alkyl; alkoxyalkyl, including methoxymethyl; aralkyl, including benzyl; aryloxyalkyl, such as phenoxymethyl; or aryl, including phenyl optionally substituted with halogen (F, Cl, Br, I), hydroxyl, C 1 to C 4 alkyl, or C 1 to C 4 alkoxy, sulfonate esters, such as alkyl or aralkyl sulphonyl, including methanesulfonyl, the mono, di or triphosphate ester, trityl or monomethoxytrityl, substituted benzyl, trialkylsilyl (e.g. dimethyl-t-butylsilyl) or dipheny
- lower acyl refers to an acyl group in which the non-carbonyl moiety is lower alkyl.
- purine or "pyrimidine” base includes, but is not limited to, adenine, N 6 -alkylpurines, N 6 -acylpurines (wherein acyl is C(O)(alkyl, aryl, alkylaryl, or arylalkyl), N 6 -benzylpurine, N 6 -halopurine, N 6 -vinylpurine, N 6 - acetylenic purine, N 6 -acyl purine, N 6 -hydroxyalkyl purine, N 6 - allcylaminopurine, N 6 -thioallcyl purine, N 2 -alkylpurines, N 2 -alkyl-6-thiopurines, thymine, cytosine, 5-fluorocytosine, 5-methylcytosine, 6-azapyrimidine, including 6-azacytosine, 2- and/or 4-mercaptopyrmidine, uracil, 5-halouracil, including 5-fluorouraci
- Purine bases include, but are not limited to, guanine, adenine, hypoxanthine, 2,6- diaminopurine, and 6-chloropurine. Functional oxygen and nitrogen groups on the base can be protected as necessary or desired. Suitable protecting groups are well known to those skilled in the art, and include trimethylsilyl, dimethylhexylsilyl, t-butyldimethylsilyl, and r-butyldiphenylsilyl, trityl, alkyl groups, and acyl groups such as acetyl and propionyl, methanesulfonyl, and p- toluenesulfonyl.
- P* means that the phosphorous atom is chiral and that it has a corresponding Cahn-Ingold-Prelog designation of "R” or "S” which have their accepted plain meanings. It is contemplated that compounds of the formula I are racemic because the chirality at phosphorous. Applicants contemplate use of the racemate and/or the resolved enantiomers. In some instances, an asterisk does not appear next to the phosphoroamidate phosphorous atom. In these instances, it is understood that the phosphorous atom is chiral and that one of ordinary skill understands this to be so unless the substituents bound to the phosphorous exclude the possibility of chirality at phosphorous, such as in P(O)Cl 3 .
- An aspect of the invention is directed to a compound, its salts, hydrates, solvates, crystalline forms, and the like represented by formula I:
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R r ) 2 , COR 1 - , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, Q -20 alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1 " is -OR 1 or -N(R r)
- R 2 is hydrogen, C 1-10 alkyl, R 3a or R 3b and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms,
- R 3a is CH 3 , -CH 2 CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , -CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 - imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy, di(lower alkyl)-amino, or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), O(C M o acyl), O(C 1-4 alkyl), O(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2-4 alkynyl), S(C 2-4 alkenyl), SO(C 1-4 acyl), SO(C 1-4 alkyl), SO(C 2-4 alkynyl), SO(C 2-4 alkenyl),
- the base is a naturally occurring or modified purine or pyrimidine base represented by the following structures:
- Z is N or CR 12 ;
- R 7 , R 8 ,R 9 , R 10 , and R n are independently H, F, Cl, Br, I, OH, OR', SH,
- R' is an optionally substituted alkyl , which includes, but is not limited to, an optionally substituted C 1-20 alkyl, an optionally substituted C 1 .io alkyl, an optionally substituted lower alkyl; an optionally substituted cycloalkyl; an optionally substituted alkynyl of C 2 -C 6 , an optionally substituted lower alkenyl of C 2 -C 6 , or optionally substituted acyl, which includes but is not limited to C(O) alkyl, C(O)(C 1-20 alkyl), C(O)(C 1-10 alkyl), or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms; and
- R 12 is H, halogen (including F, Cl, Br, I), OH, OR', SH, SR', NH 2 , NHR',
- a first embodiment of the invention is directed to a compound represented by formula 1-1 :
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R r ) 2 , COR 1 - , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-2 O alkyl, C 1-10 alkyl, or C !-6 alkyl, R 1" is -OR or -N(R 1-6
- R 2 is hydrogen, C 1-10 alkyl, R 3a or R 3b and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms, C(O)CR 32 R 313 NHR 1 , where n is 2 to 4 and R 1 , R 3a , and R 3b ;
- R 3a and R 3b are (i) independently selected from hydrogen, C 1-10 alkyl, cycloalkyl, -(CH 2 ) C (NR 3' ) 2 , C, -6 hydroxyalkyl, -CH 2 SH, -(CH 2 ) 2 S(O) d Me, -
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), O(C 1-]0 acyl), O(C 1-4 alkyl), O(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2 ⁇ alkynyl), S(C 2-4 alkenyl), SO(C 1-4 acyl), SO(C 1-4 alkyl), SO(C 2-4 alkynyl), SO(C 2-4 alkenyl), SO 2 (C 1-4 acyl), SO 2 (C
- R 7 , R 8 ,R 9 are independently H, F, Cl, Br, I, OH, OR', SH, SR',
- a first aspect of the first embodiment is directed to a compound represented by formula 1-1
- R 1 is hydrogen, n-alkyl or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 1-6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl;
- R 2 is hydrogen or CH 3 ;
- R 3a and R 3b are independently (i) R 3a is hydrogen and R 3b and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms (v) R 3b is hydrogen and R 3a and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms, where c is 1 to 6, d is 0 to 2, e is 0 to 3, f is 3 to 5, n is 2 to 4, and where R 3 is independently hydrogen or C 1-6 alkyl and R 3 " is -OR' or -N(R 3' ) 2 ); (vi) R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, - CH 2 CH 2 SCH 3 , CH 2
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy, di(lower alkyl)-amino, or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, CN, CH 3 , vinyl, OCH 3 , OCH 2 CH 3 , CH 2 OH, CH 2 (halo), such as CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I, with the provisos that when X is OH , base is cytosine and R 6 is H, R 5 cannot be N 3 ;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, vinyl, N 3 , CN, Cl, Br, F, I, O(C 1-6 acyl),
- R 7 , R 8 ,R 9 are independently H, F, Cl, Br, I, OH, OR', SH, SR 1 , NH 2 , NHR', NR' 2 , lower alkyl, halogenated (F, Cl, Br, I) lower alkyl, lower alkenyl of C 2 -C 6 , CO 2 H, CO 2 R', CONH 2 , CONHR', CONR' 2 , wherein R' is a C- 2 o alkyl; a C 1-20 cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl.
- a second aspect of the first embodiment is directed to a compound represented by formula 1-1
- R 1 is hydrogen, n-alkyl or a substituted or unsubstituted phenyl, where the substitutent of the substituted phenyl is at least one of a C 1-3 alkyl, a C 1-3 alkoxy, F, Cl, Br, I, nitro, cyano, and a C 1-3 haloalkyl;
- R 2 is hydrogen or CH 3 ;
- R 3a and R 3b are independently (i) R 3a is hydrogen and R 3b and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms (v) R 3b is hydrogen and R 3a and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms, where c is 1 to 6, d is 0 to 2, e is 0 to 3, f is 3 to 5, n is 2 to 4, and where R 3 is independently hydrogen or C 1-6 alkyl and R 3- is -OR' or -N(R 3' ) 2 ); (vi) R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -
- R 3a is CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3- yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, r Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, di(lower alkyl)amino-lower alkyl, or aminoacyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 (halo), such as CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I, with the provisos that when X is OH , base is cytosine and R 6 is H, R 5 cannot be N 3 ;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 , NH 2 , NHCH 3 , NH(vinyl), NH(acetyl), NH(C(O)CH 3 ), N(CH 3 ) 2 , N(C(O)CH 3 ) 2 , or O(aminoacyl);
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 ; and G) R 9 is selected from among OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , OC(O)(C 1-20 alkyl), which include but are not limited to OC(O)(CH 2 ) s CH 3 , NHC(O)(C 1-20 alkyl), which include but are not limited to NHC(O)(CH 2 ) S CH 3 , and N(C(O)(CH 2 ) S CH 3 ) 2 ,
- a third aspect of the first embodiment is directed to a compound represented by formula 1-1
- R 1 is hydrogen, n-alkyl or a substituted or unsubstituted phenyl, where the substitutent of the substituted phenyl is at least one of a CH 3 , OCH 3 , F, Cl, Br, I, nitro, cyano, and a CH 3-q X q , where X is F, Cl, Br, or I, and q is 1-3;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, lower haloalkyl, di(lower alkyl)amino-lower alkyl, or aminoacyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 (halo), such as CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I, with the provisos that when X is OH , base is cytosine and R 6 is H, R 5 cannot be N 3 ;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ,
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 , wherein R' is a C 1-20 alkyl; a C 1-20 cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl'; and
- R 9 is selected from among OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , OC(O)(C 1-20 alkyl), which include but are not limited to OC(O)(CH 2 ) S CH 3 , NHC(O)(C 1-20 alkyl), which include but are not limited to NHC(O)(CH 2 ) S CH 3 , and N(C(O)(CH 2 ) S CH 3 ) 2 , which include but is not limited to N(C(O)(CH 2 ) S CH 3 ) 2 , where s is an integer selected from 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, and 19.
- a fourth aspect of the first embodiment is directed to a compound represented by formula 1-2
- R 1 is hydrogen, methyl, ethyl, w-propyl, /-propyl, or a substituted or unsubstituted phenyl, where the substitutent of the substituted phenyl is at least one of a CH 3 , OCH 3 , F, Cl, Br, I, nitro, cyano, and a CH3 -q X q , where X is F, Cl, Br, or I, and q is 1-3;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 )2, CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H,
- R 4 is hydrogen, C 1 _io alkyl, C 1- ]O alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C ⁇ io haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I, with the provisos that when X is OH , base is cytosine and R 6 is H, R 5 cannot be N 3 ;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 , NH 2 , NHCH 3 , NH(vinyl), NH(acetyl), NH(C(O)CH 3 ), N(CH 3 ) 2 , N(C(O)CH 3 ) 2 , or O(aminoacy 1) ;
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 , wherein R' is a C 1-20 alkyl; a C 1-20 cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl; and
- R 9 is selected from among OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 ,
- a fifth aspect of the first embodiment is directed to a compound represented by formula 1-2
- R 1 is hydrogen, methyl, ethyl, n-propyl, i-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , - CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 -imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH 2 ((4'- OH)-Ph), CH 2 SH, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, lower haloalkyl, di(lower alkyl)amino-lower alkyl, or aminoacyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F,
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 , or O(aminoacyl);
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 , wherein R 1 is a C 1-2 O alkyl; a C 1-20 cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl; and
- R 9 is selected from among OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 ,
- a sixth aspect of the first embodiment is directed to a compound represented by formula 1-2
- R ! is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, OMe, CN, CH 2 F, F, Cl, Br, or I;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, or N 3 , OCH 3 , OC(O)CH 3 , or O(aminoacyl);
- R 7 and R 8 are independently H, F, Br, SCH 3 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 ; and
- O) R 9 is selected from among OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , OC(O)(C 1-20 alkyl), which include but are not limited to
- NHC(O)(C 1-20 alkyl which include but are not limited to NHC(O)(CH 2 ) s CH 3
- N(C(O)(CH 2 ) S CH 3 ) 2 which include but is not limited to N(C(O)(CH 2 ) S CH 3 ) 2 , where s is an integer selected from 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, and 19.
- a seventh aspect of the first embodiment is directed to a compound represented by formula 1-2
- R ! is hydrogen, methyl, ethyl, «-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, r Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, or N 3 ;
- Y is OH, OCH 3 , OC(O)CH 3 , or O(aminoacyl);
- R 7 and R 8 are independently H, F, Br, SCH 3 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 ; and
- R 9 is selected from among OH, OCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , OC(O)(C 1-20 alkyl), which include but are not limited to OC(O)(CH 2 ) S CH 3 , NHC(O)(C 1-20 alkyl), which include but are not limited to NHC(O)(CH 2 ) S CH 3 , and N(C(O)(CH 2 ) S CH 3 ) 2 , which include but is not limited to
- N(C(O)(CH 2 ) S CH 3 ) 2 where s is an integer selected from among 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, and 19.
- An eighth aspect of the first embodiment is directed to a compound represented by formula 1-2
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, or N 3 ;
- Y is OH, OCH 3 , OC(O)CH 3 , or O(aminoacyl);
- R 7 and R 8 are independently H, F, Br, SCH 3 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 ;
- R 9 is selected from among OH, OCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 ,
- OC(O)(C 1-20 alkyl which include but are not limited to OC(O)(CH 2 ) S CH 3
- NHC(O)(C 1-20 alkyl) which include but are not limited to NHC(O)(CH 2 ) S CH 3
- N(C(O)(CH 2 ) S CH 3 ) 2 which include but is not limited to N(C(O)(CH 2 ) S CH 3 ) 2 , where s is an integer selected from among 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, and 19.
- a second embodiment of the invention is directed to a compound represented by formula I in which the base is a structure represented by formula b above, wherein R 1 , R 2 , R 3a , R 3b , R 4 , R 5 , R 6 , X, Y, R 7 , and R 8 are defined in the Summary of the Invention section above.
- a first aspect of the second embodiment is directed to a compound represented by formula 1-3
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1 -6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C) -6 alkyl, -SO 2 N(R r ) 2 , COR !- , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-20 alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1- is -OR' or -N(R')
- R 2 is hydrogen or CH 3 ;
- R 4 is hydrogen, C 1-1 O alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN,
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OR', SH, SR',
- the second aspect of the second embodiment is directed to a compound represented by formula 1-3
- R 1 is hydrogen, n-alkyl or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 1-6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1 _ 6 haloalkyl;
- R 2 is hydrogen or CH 3 ;
- R 3a and R 3b are independently (i) R 3a is hydrogen and R 3b and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms (v) R 3b is hydrogen and R 3a and R 2 together are (CH 2 ),, so as to form a cyclic ring that includes the adjoining N and C atoms, where c is 1 to 6, d is 0 to 2, e is 0 to 3, f is 3 to 5, n is 2 to 4, and where R 3 is independently hydrogen or C 1-6 alkyl and R 3- is -OR' or -N(R 3' ) 2 ); (vi) R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, - CH 2 CH 2 SCH 3 , CH 2 CO 2
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy or halogen, Q-, 0 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, CN, CH 3 , vinyl, OCH 3 , OCH 2 CH 3 , CH 2 OH, CH 2 (halo), such as CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, vinyl, N 3 , CN, Cl, Br, F, I, O(C 1-6 acyl),
- NH 2 NH(C 1-4 alkyl), NH(C 2-4 alkenyl), NH(C 2-4 alkynyl), NH(C 1-4 acyl), N(C 1-4 alkyl) 2 , N(C 1-18 acyl) 2 , wherein alkyl, alkynyl, alkenyl and vinyl are optionally substituted by N 3 , CN, one to three halogen (Cl, Br, F, I), NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), 0(C 1-4 acyl), 0(C 1-4 alkyl), 0(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 acyl), S(C 1-4
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OR', SH, SR', NH 2 , NHR', NR' 2 , lower alkyl, halogenated (F, Cl, Br, I) lower alkyl, lower alkenyl of C 2 -C 6 , CO 2 H, CO 2 R', CONH 2 , CONHR 1 , C0NR' 2 , wherein R' is a Q- 20 alkyl; a C 1-20 cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl.
- the third aspect of the second embodiment is directed to a compound represented by formula 1-3
- R 1 is hydrogen, n-alkyl or a substituted or unsubstituted phenyl, where the substitutent of the substituted phenyl is at least one of a C 1-3 alkyl, a C 1-3 alkoxy, F, Cl, Br, I, nitro, cyano, and a C 1-3 haloalkyl;
- R 2 is hydrogen, CH 3 , R 3a or R 3b and R 2 together are (CH 2 ) 3 so as to form a cyclic ring that includes the adjoining N and C atoms, C(O)CR 33 R 313 NHR 1 , where n is 2 to 4 and R 1 , R 3a , and R 3b are as defined herein;
- R 3a and R 3b are independently (i) R 3a is hydrogen and R 3b and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms (v) R 3b is hydrogen and R 3a and R 2 together are (CH 2 ) n so as to form a cyclic ring that includes the adjoining N and C atoms, where c is 1 to 6, d is 0 to 2, e is 0 to 3, f is 3 to 5, n is 2 to 4, and where R 3 is independently hydrogen or C 1-6 alkyl and R 3- is -OR 1 or -N(R 3' ) 2 ); (vi) R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, - CH 2 CH 2 SCH 3 , CH 2 CO
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, t Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 (halo), such as CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , halogen, NH 2 , or N 3
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 , NH 2 , NHCH 3 , NH(vinyl), NH(acetyl), NH(C(O)CH 3 ), N(CH 3 ) 2 , N(C(O)CH 3 ) 2 ;
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 .
- the fourth aspect of the second embodiment is directed to a compound represented by formula 1-3
- R 1 is hydrogen, n-alkyl or a substituted or unsubstituted phenyl, where the substitutent of the substituted phenyl is at least one of a CH 3 , OCH 3 ,
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , -
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 (halo), such as CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 , NH 2 , NHCH 3 , NH(vinyl), NH(acetyl), NH(C(O)CH 3 ), N(CH 3 ) 2 , N(C(O)CH 3 ) 2 ;
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OCH 3 , SH,
- SCH 3 NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 , wherein R' is a C 1-20 alkyl; a C 1-20 cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl.
- the fifth aspect of the second embodiment is directed to a compound represented by formula 1-4
- R is hydrogen, methyl, ethyl, n-propyl, /-propyl, or a substituted or unsubstituted phenyl, where the substitutent of the substituted phenyl is at least one of a CH 3 , OCH 3 , F, Cl, Br, I, nitro, cyano, and a CH 3-q X q , where X is F, Cl, Br, or I, and q is 1-3;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , - CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 -imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH 2 ((4'- OH)-Ph), CH 2 SH, or lower cycloalkyl or R 3a is CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, N 3 , CH 2 CN, CH 2 N 3 , CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , halogen, including F, Cl, Br, or I;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 , NH 2 , NHCH 3 , NH(vinyl), NH(acetyl), NH(C(O)CH 3 ), N(CH 3 ) 2 , N(C(O)CH 3 ) 2 ;
- R 7 and R 8 are independently H, F, Cl, Br, 1, OH, OCH 3 , SH, SCH 3 , NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 , wherein R' is a C 1-20 alkyl; a C 1-2 O cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl,
- the sixth aspect of the second embodiment is directed to a compound represented by formula 1-4 wherein
- R 1 is hydrogen, methyl, ethyl, «-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2 , N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ;
- R 7 and R 8 are independently H, F, Cl, Br, I, OH, OCH 3 , SH,
- SCH 3 NH 2 , NHCH 3 , N(CH 3 ) 2 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 H, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 , wherein R' is a C 1-20 alkyl; a C 1-20 cycloalkyl; a C 2 -C 6 alkenyl, a C 2 -C 6 alkynyl.
- the seventh aspect of the second embodiment is directed to a compound represented by formula 1-4
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, OMe, CN, CH 2 F, F, Cl, Br, or I;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH3, or OC(O)CH 3 ;
- R 7 and R 8 are independently H, F, Br, SCH 3 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 ;
- the eighth aspect of the second embodiment is directed to a compound represented by formula 1-4
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2, OCH 3 , or OC(O)CH 3 ;
- R 7 and R 8 are independently H, F, Br, SCH 3 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 .
- the ninth aspect of the second embodiment is directed to a compound represented by formula 1-4
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, or N 3 ;
- Y is OH, OCH 3 , or OC(O)CH 3 ;
- R 7 and R 8 are independently H, F, Br, SCH 3 , CH 3 , CH 3-q X q , where X is F, Cl, Br, or I and q is 1 to 3, vinyl, CO 2 CH 3 , CONH 2 , CONHCH 3 , or CON(CH 3 ) 2 .
- a third embodiment of the invention is directed to a compound represented by formula I in which the base is a structure represented by formula c above, wherein R 1 , R 2 , R 3a , R 3b , R 4 , R 5 , R 6 , X, Y, Z, R 10 , R 11 , and R 12 are defined in the Summary of the Invention section above; with the proviso that R 11 is not H.
- a first aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R r ) 2 , COR 1- , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-20 alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1- is -OR' or -N(R r ) 2
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), O(C 1-!0 acyl), 0(C 1-4 alkyl), 0(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2-4 alkynyl), S(C 2-4 alkenyl), SO(C 1-4 acyl), SO(C 1-4 alkyl), SO(C 2-4 alkynyl), SO(C 2-4 alkenyl),
- R' is an optionally substituted alkyl , which includes, but is not limited to, an optionally substituted C 1-20 alkyl, an optionally substituted C 1-10 alkyl, an optionally substituted lower alkyl; an optionally substituted cycloalkyl; an optionally substituted alkynyl of C 2 -C 6 , an optionally substituted lower alkenyl of C 2 -C 6 , or optionally substituted acyl, which includes but is not limited to C(O) alkyl, C(O)(C 1-20 alkyl), C(O)(C 1-10 alkyl), or C(O)(lower alkyl) or alternatively, in the instance of NR 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms; and
- (j) Z is N or CR 12 ;
- a second aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H.
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- a third aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, ethyl, n-propyl, z-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, ! Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I; with the proviso that X is OH,
- R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH 3 , or OC(O)CH 3 ;
- R 10 and R 1 x are independently H, F, Br, I, OH, OR 1 , NH 2 , NHR',
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- a fourth aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, ethyl, rc-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- a fifth aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- (j) Z is N or CR 12 ;
- a sixth aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, ethyl, w-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CHa) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , - CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 -imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH 2 ((4'- OH)-Ph), CH 2 SH, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H.
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2 , N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ;
- R 10 is NH 2 and R 11 is H, F, Br, I, OH, OR', NH 2 , NHR', NR 2 ,
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- a seventh aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl -piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I; with the proviso that X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- An eighth aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- a ninth aspect of the third embodiment is directed to a compound represented by formula 1-5
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, r Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, or N-methyl-pyrrolidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- a tenth aspect of the third embodiment is directed to a compound represented by formula 1-6
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R r ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R r ) 2 , COR 1- , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-20 alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1 - is -OR or -N(R r r
- R 2 is hydrogen or CH 3 ;
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), 0(C 1-10 acyl), 0(C 1-4 alkyl), 0(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2-4 alkynyl), S(C 2-4 alkenyl), SO(C 1-4 acyl), SO(C 1-4 alkyl), SO(C
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- (j) Z is N or CR 12 ;
- An eleventh aspect of the third embodiment is directed to a compound represented by formula 1-6
- R 1 is hydrogen, methyl, ethyl, w-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , - CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 -imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH 2 ((4'- OH)-Ph), CH 2 SH, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-raethyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2 , N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ;
- R 10 and R 1 ' are independently H, F, Br, I, OH, OR', NH 2 , NHR',
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- a twelvth aspect of the third embodiment is directed to a compound represented by formula 1-6
- R 1 is hydrogen, methyl, ethyl, r ⁇ -propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, r Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms; and Q) Z is N or CR 12 ; and
- a thirteenth aspect of the third embodiment is directed to a compound represented by formula 1-6
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- G) Z is N or CR 12 ;
- a fourteenth aspect of the third embodiment is directed to a compound represented by formula 1-6
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F,
- R 5 cannot be H
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- (j) Z is N or CR 12 ;
- a fifteenth aspect of the third embodiment is directed to a compound represented by formula 1-6
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , Q -6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R 1' ) 2 , COR 1" , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-2 O alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1- is -OR or -N(R r )
- R 2 is hydrogen or CH 3 ;
- R 4 is hydrogen, C 1-10 alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C 1-10 haloalkyl, C 3 _io cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), 0(C 1-10 acyl), 0(C 1-4 alkyl), 0(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2-4 alkynyl), S(C 2-4 alkenyl), SO(C 1-4 acyl), SO(C 1-4 alkyl), SO(C 2-4 alkynyl), SO(C 2-4 alkenyl), SO 2 (C 1-4 acyl), SO 2 (C
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- (j) Z is N or CR 12 ;
- An sixteenth aspect of the third embodiment is directed to a compound represented by formula 1-6
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , - CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 -imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH 2 ((4'- OH)-Ph), CH 2 SH, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F,
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2 , N 3 , CN, Cl, Br, F, I, OC(O)CH 3 ,
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- a seventeenth aspect of the third embodiment is directed to a compound represented by formula 1-6 wherein
- R 1 is hydrogen, methyl, ethyl, rc-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- An eighteenth aspect of the third embodiment is directed to a compound represented by formula 1-6 wherein
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- (j) Z is N or CR 12 ;
- a ninteenth aspect of the third embodiment is directed to a compound represented by formula 1-6 wherein
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- (j) Z is N or CR 12 ;
- a fourth embodiment of the invention is directed to a compound represented by formula I in which the base is a structure represented by formula c above, where R 11 is H and R 2 , R 3a , R 3b , R 4 , R 5 , R 6 , X, and Y are defined in the Summary of the Invention section above.
- a first aspect of the fourth embodiment is directed to a compound represented by formula 1-7
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1 -6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R r ) 2 , COR 1- , and -SO 2 C L6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-20 alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1- is -OR' or -N(R r )
- R 2 is hydrogen or CH 3 ;
- R 3a is CH 3 , -CH 2 CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , -CH 2 CH 2 CH 2 NHC(NH)NH 2 , CH 2 - imidazol-4-yl, CH 2 OH, CH(OH)CH 3 , CH 2 ((4'-OH)-Ph), CH 2 SH, or lower cycloalkyl and R 3b is H, where R 3 is independently hydrogen or alkyl, which
- R 4 is hydrogen, C 1-1 O alkyl, C 1-10 alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C 1-10 haloalkyl, C 3-10 cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH),
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 14 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), O(C 1-]0 acyl), 0(C 1-4 alkyl), 0(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2-4 alkynyl), S(C 2-4 alkenyl), SO(C 14 acyl), SO(C 1-4 alkyl), SO(C 14 acyl
- R' is an optionally substituted alkyl , which includes, but is not limited to, an optionally substituted C 1-20 alkyl, an optionally substituted C 1-10 alkyl, an optionally substituted lower alkyl; an optionally substituted cycloalkyl; an optionally substituted alkynyl of C 2 -C 6 , an optionally substituted lower alkenyl of C 2 -C 6 , or optionally substituted acyl, which includes but is not limited to C(O) alkyl, C(O)(C 1-20 alkyl), C(O)(C 1-10 alkyl), or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms; and
- (j) Z is N or CR 12 ; and R 12 is H, halogen (including F, Cl, Br, I), OH, OR', SH, SR 1 , NH 2 , NHR', NR 2 , NO 2 lower alkyl of C 1 -C 6 , halogenated (F, Cl, Br, I) lower alkyl of C 1 -C 6 , lower alkenyl of C 2 -C 6 , halogenated (F, Cl, Br, I) lower alkenyl of C 2 -C 6 , lower alkynyl of C 2 -C 6 , halogenated (F, Cl, Br, I) lower alkynyl of C 2 -C 6 , lower alkoxy of C 1 -C 6 , halogenated (F, Cl, Br, I) lower alkoxy of C 1 -C 6 , CO 2 H, CO 2 R',
- a second aspect of the fourth embodiment is directed to a compound represented by formula 1-7
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H.
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , halogen, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R 1 comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- a third aspect of the third embodiment is directed to a compound represented by formula 1-7
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I; with the proviso that X is OH,
- R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- a fourth aspect of the fourth embodiment is directed to a compound represented by formula 1-7
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, z-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di ⁇ ower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F,
- R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R 1 comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- Q) Z is N or CR 12 ;
- a fifth aspect of the fourth embodiment is directed to a compound represented by formula 1-7
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- CO R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- (j) Z is N or CR 12 ;
- a sixth aspect of the fourth embodiment is directed to a compound represented by formula 1-8
- R 1 is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1 -6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R !' ) 2 , COR 1 ", and -SO 2 C 1-6 alkyl; (R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-2 O alkyl, Q.io alkyl, or C 1-6 alkyl, R ! - is -OR' or
- R 2 is hydrogen or CH 3 ;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), OC 1-10 haloalkyl, O(aminoacyl), 0(C 1-10 acyl), 0(C 1-4 alkyl), 0(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2-4 alkynyl), S(C 2-4 alkenyl), SO(C 1-4 acyl), SO(C 1-4 alkyl), SO(C 2-4 alkynyl), SO(C 2-4 alkenyl), SO 2 (C 1-4 alkyl), SO 2 (C 1-4 alkyl), SO 2 (C 1-4 alky
- R 10 is H, F, Br, I, OH, OR', NH 2 , NHR', NR' 2 , CO 2 R', CONH 2 ,
- each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- G) Z is N or CR 12 ;
- a seventh aspect of the fourth embodiment is directed to a compound represented by formula 1-8
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , halogen, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2 , N 3 , CN, Cl, Br, F, I, OC(O)CH 3 ,
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- O) Z is N or CR 12 ;
- An eighth aspect of the fourth embodiment is directed to a compound represented by formula 1-8
- R 1 is hydrogen, methyl, ethyl, n-propyl, z-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl , N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H; (f) R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH 3 , or OC(O)CH 3 ;
- R 1 is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms; and
- O) Z is N or CR 12 ;
- a ninth aspect of the fourth embodiment is directed to a compound represented by formula 1-8
- R 1 is hydrogen, methyl, ethyl, n-propyl, z-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- O) Z is N or CR 12 ;
- a tenth aspect of the fourth embodiment is directed to a compound represented by formula 1-8
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- R' is a lower alkyl, a lower cycloalkyl, or C(O)(lower alkyl) or alternatively, in the instance of NR' 2 , each R' comprise at least one C atom that are joined to form a heterocycle comprising at least two carbon atoms;
- Q) Z is N or CR 12 ;
- a fifth embodiment of the invention is directed to a compound represented by formula I in which the base is a structure represented by formula d above, wherein R 1 , R 2 , R 3a , R 3b , R 4 , R 5 , R 6 , X, and Y are defined in the Summary of the Invention section above.
- the first aspect of the fifth embodiment is directed to a compound represented by formula 1-9
- R is hydrogen, n-alkyl; branched alkyl; cycloalkyl; or aryl, which includes, but is not limited to, phenyl or naphthyl, where phenyl or naphthyl are optionally substituted with at least one of C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1 -6 alkoxy, F, Cl, Br, I, nitro, cyano, C 1-6 haloalkyl, -N(R 1 ) 2 , C 1-6 acylamino, - NHSO 2 C 1-6 alkyl, -SO 2 N(R r ) 2 , COR 1- , and -SO 2 C 1-6 alkyl;
- R 1' is independently hydrogen or alkyl, which includes, but is not limited to, C 1-20 alkyl, C 1-10 alkyl, or C 1-6 alkyl, R 1 - is -OR' or -N(R r r
- R 2 is hydrogen or CH 3 ;
- R 4 is hydrogen, C -10 alkyl, C 1-1 O alkyl optionally substituted with a lower alkyl, alkoxy or halogen, C -10 haloalkyl, C 3- io cycloalkyl, cycloalkyl alkyl, cycloheteroalkyl, aminoacyl, di(lower alkyl)amino-lower alkyl, aryl, such as phenyl, heteroaryl, such as, pyridinyl, substituted aryl, or substituted heteroaryl;
- R 5 is H, a lower alkyl, CN, vinyl, O-(lower alkyl), hydroxyl lower alkyl, i.e., -(CH 2 ) P OH, where p is 1 -6, including hydroxyl methyl (CH 2 OH), CH 2 F, N 3 , CH 2 CN, CH 2 NH 2 , CH 2 NHCH 3 , CH 2 N(CH 3 ) 2 , alkyne (optionally substituted), or halogen, including F, Cl, Br, or I, with the provisos that when X is OH, base is cytosine and R 6 is H, R 5 cannot be N 3 and when X is OH, R 6 is CH 3 or CH 2 F and B is a purine base, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OMe, halogen, NH 2 , or N 3 ;
- Y is OH, H, C 1-4 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, vinyl, N 3 , CN, Cl, Br, F, I, NO 2 , OC(O)O(C 1-4 alkyl), OC(O)O(C 1-4 alkyl), OC(O)O(C 2-4 alkynyl), OC(O)O(C 2-4 alkenyl), Od -10 haloalkyl, O(aminoacyl), 0(C 1-10 acyl), 0(C 1-4 alkyl), 0(C 2-4 alkenyl), S(C 1-4 acyl), S(C 1-4 alkyl), S(C 2-4 alkynyl), S(C 2-4 alkenyl), SO(C 1-4 acyl), SO(C 1-4 alkyl), SO(C 2-4 alkynyl), SO(C 2-4 alkenyl), SO 2 (C 1-4 acyl), SO 2 (C
- a second aspect of the fifth embodiment is directed to a compound represented by formula 1-9
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 ,
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H.
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , halogen, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ;
- a third aspect of the fifth embodiment is directed to a compound represented by formula 1-9
- R 1 is hydrogen, methyl, ethyl, «-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, r Pr, n Pr, n Bu, 2-butyl, f Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I; with the proviso that X is OH,
- R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 N 3 , OCH 3 , or OC(O)CH 3 ;
- a fourth aspect of the fifth embodiment is directed to a compound represented by formula 1-9
- R 1 is hydrogen, methyl, ethyl, «-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- a fifth aspect of the fifth embodiment is directed to a compound represented by formula 1-9
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, r Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- a sixth aspect of the fifth embodiment is directed to a compound represented by formula 1-10
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, /-propyl, or a substituted or unsubstituted phenyl, where the substitutent of the substituted phenyl is at least one of a CH 3 , OCH 3 , F, Cl, Br, I, nitro, cyano, and a CH 3-q X q , where X is F, Cl, Br, or I, and q is 1-3;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , -
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, N 3 , CH 2 CN, CH 2 N 3 ,
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 ,
- a seventh aspect of the fifth embodiment is directed to a compound represented by formula 1-10
- R 1 is hydrogen, methyl, ethyl, «-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, CH 2 -indol-3-yl, -CH 2 CH 2 SCH 3 , CH 2 CO 2 H, CH 2 C(O)NH 2 , CH 2 CH 2 COOH, CH 2 CH 2 C(O)NH 2 , CH 2 CH 2 CH 2 CH 2 NH 2 , -
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 3 , OCH 3 , CH 2 OH, CH 2 F, halogen, including F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , F, or CN;
- X is H, OH, F, OCH 3 , Cl, Br, I, NH 2 , or N 3 ;
- Y is OH, H, CH 3 , vinyl, NH 2 , N 3 , CN, Cl, Br, F, I, OC(O)CH 3 , OCH 3 .
- An eighth aspect of the fifth embodiment is directed to a compound represented by formula 1-10
- R 1 is hydrogen, methyl, ethyl, n-propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen or CH 3 ;
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, 1 Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, CN, CH 2 F, F, Cl, Br, or I, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, F, OCH 3 , F, Cl, Br, I, NH 2 or N 3 ;
- Y is H, OH, CH 3 , F, Cl, Br, I, NH 2 or N 3 , OCH 3 , or OC(O)CH 3 ;
- a ninth aspect of the fifth embodiment is directed to a compound represented by formula 1-10
- R 1 is hydrogen, methyl, ethyl, ⁇ -propyl, /-propyl, phenyl, p-tolyl, p-bromo-phenyl, p-chloro-phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, n Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3 -yl, N-methy l-pyrrolidin-3 -y 1, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F, R 5 cannot be H;
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 ;
- a tenth aspect of the fifth embodiment is directed to a compound represented by formula 1-10
- R 1 is hydrogen, methyl, phenyl, p-bromo-phenyl, p-chloro- phenyl, p-fluorophenyl;
- R 2 is hydrogen
- R 3a is H and R 3b is H, CH 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , CH 2 Ph, or lower cycloalkyl;
- R 4 is hydrogen, CH 3 , Et, i Pr, "Pr, n Bu, 2-butyl, 'Bu, benzyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, N-methyl-aziridin-2-yl, N- methyl-azetidin-3-yl, N-methyl-pyrrolidin-3-yl, N-methyl-pyrrolidin-4-yl, N- methyl-piperidin-4-yl, lower haloalkyl, or di(lower alkyl)amino-lower alkyl;
- R 5 is H, with the provisos that when X is OH, R 6 is CH 3 or CH 2 F,
- R 5 cannot be H
- R 6 is H, CH 3 , CH 2 F, CHF 2 , CF 3 , or F;
- X is H, OH, OCH 3 , F, NH 2 or N 3 ;
- Y is OH, NH 2 , OCH 3 , or OC(O)CH 3 .
- the phosphoramidate substituent containing the substituents R 3a and R 3b are depicted without reference to stereochemical structure ⁇ cf. structures 1-1, 1-3, 1-5, 1-7, and 1-9 above). It is contemplated that the compounds recited below embody compounds in which R 3a projects toward the viewer while R 3b projects away from the viewer (cf. structures 1-2, 1-4, 1-6, 1-8, and 1-10). Moreover, it is contemplated that the compounds recited below also embody compounds in which R 3a projects away from the viewer while R 3b projects towards the viewer.
- preferred compounds are those in which R 3a projects towards the viewer and R 3b projects away from the viewer such that the natural L-amino acid (S)-configuration is presented. Additionally, the inventors recognize that the phosphorus atom of the phosphoramidate moiety is another source of chirality.
- the structures below do not specifically depict chirality at phosphorus, the inventors recognize that stereochemical configurations are possible such that in a staggered (or zig- zag) line structure the oxo-substitutent projects towards the viewer while the OR 1 substitutent projects away from the viewer, and vice versa, i.e., where the Cahn-Ingold-Prelog stereochemical designation of phosphorous is either R or S. Therefore, the structures below include all possible stereochemical configurations possible for phosphorus.
- a sixth embodiment of the present invention is directed to a composition for the treatment of any of the viral agents disclosed herein said composition comprising a pharmaceutically acceptable medium selected from among an excipient, carrier, diluent, and equivalent medium and a compound, that is intended to include its salts (acid or basic addition salts), hydrates, solvates, and crystalline forms can be obtained, represented by formula I.
- formulation of the sixth embodiment can contain any of the compounds contemplated in any of the aspects of the first, second, third, fourth, and fifth embodiments or those specifically recited in the tables above or exemplified herein, either alone or in combination with another compound of the present invention.
- the compounds of the present invention may be formulated in a wide variety of oral administration dosage forms and carriers.
- Oral administration can be in the form of tablets, coated tablets, hard and soft gelatin capsules, solutions, emulsions, syrups, or suspensions.
- Compounds of the present invention are efficacious when administered by suppository administration, among other routes of administration.
- the most convenient manner of administration is generally oral using a convenient daily dosing regimen which can be adjusted according to the severity of the disease and the patient's response to the antiviral medication.
- a compound or compounds of the present invention, as well as their pharmaceutically acceptable salts, together with one or more conventional excipients, carriers, or diluents, may be placed into the form of pharmaceutical compositions and unit dosages.
- the pharmaceutical compositions and unit dosage forms may be comprised of conventional ingredients in conventional proportions, with or without additional active compounds and the unit dosage forms may contain any suitable effective amount of the active ingredient commensurate with the intended daily dosage range to be employed.
- the pharmaceutical compositions may be employed as solids, such as tablets or filled capsules, semisolids, powders, sustained release formulations, or liquids such as suspensions, emulsions, or filled capsules for oral use; or in the form of suppositories for rectal or vaginal administration.
- a typical preparation will contain from about 5% to about 95% active compound or compounds (w/w).
- preparation or “dosage form” is intended to include both solid and liquid formulations of the active compound and one skilled in the art will appreciate that an active ingredient can exist in different preparations depending on the desired dose and pharmacokinetic parameters.
- excipient refers to a compound that is used to prepare a pharmaceutical composition, and is generally safe, non-toxic and neither biologically nor otherwise undesirable, and includes excipients that are acceptable for veterinary use as well as human pharmaceutical use.
- the compounds of this invention can be administered alone but will generally be administered in admixture with one or more suitable pharmaceutical excipients, diluents or carriers selected with regard to the intended route of administration and standard pharmaceutical practice.
- a “pharmaceutically acceptable salt” form of an active ingredient may also initially confer a desirable pharmacokinetic property on the active ingredient which were absent in the non-salt form, and may even positively affect the pharmacodynamics of the active ingredient with respect to its therapeutic activity in the body.
- pharmaceutically acceptable salt of a compound as used herein means a salt that is pharmaceutically acceptable and that possesses the desired pharmacological activity of the parent compound.
- Such salts include: (1) acid addition salts, formed with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like; or formed with organic acids such as glycolic acid, pyruvic acid, lactic acid, malonic acid, malic acid, maleic acid, fumaric acid, tartaric acid, citric acid, 3-(4-hydroxybenzoyl)benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, 1,2-ethane-disulfonic acid, 2- hydroxyethanesulfonic acid, benzenesulfonic acid, 4-chlorobenzenesulfonic acid, 2-naphthalenesulfonic acid, 4-toluenesulfonic acid, camphorsulfonic acid, lauryl sulfuric acid, gluconic acid, glutamic acid, salicylic acid, muconic acid, and the like
- Solid form preparations include powders, tablets, pills, capsules, suppositories, and dispersible granules.
- a solid carrier may be one or more substances which may also act as diluents, flavoring agents, solubilizers, lubricants, suspending agents, binders, preservatives, tablet disintegrating agents, or an encapsulating material.
- the carrier In powders, the carrier generally is a finely divided solid which is a mixture with the finely divided active component.
- the active component In tablets, the active component generally is mixed with the carrier having the necessary binding capacity in suitable proportions and compacted in the shape and size desired.
- Suitable carriers include but are not limited to magnesium carbonate, magnesium stearate, talc, sugar, lactose, pectin, dextrin, starch, gelatin, tragacanth, methylcellulose, sodium carboxymethylcellulose, a low melting wax, cocoa butter, and the like.
- Solid form preparations may contain, in addition to the active component, colorants, flavors, stabilizers, buffers, artificial and natural sweeteners, dispersants, thickeners, solubilizing agents, and the like.
- Liquid formulations also are suitable for oral administration include liquid formulation including emulsions, syrups, elixirs and aqueous suspensions. These include solid form preparations which are intended to be converted to liquid form preparations shortly before use. Emulsions may be prepared in solutions, for example, in aqueous propylene glycol solutions or may contain emulsifying agents such as lecithin, sorbitan monooleate, or acacia. Aqueous suspensions can be prepared by dispersing the finely divided active component in water with viscous material, such as natural or synthetic gums, resins, methylcellulose, sodium carboxymethylcellulose, and other well known suspending agents.
- viscous material such as natural or synthetic gums, resins, methylcellulose, sodium carboxymethylcellulose, and other well known suspending agents.
- the compounds of the present invention may be formulated for administration as suppositories.
- a low melting wax such as a mixture of fatty acid glycerides or cocoa butter is first melted and the active component is dispersed homogeneously, for example, by stirring. The molten homogeneous mixture is then poured into convenient sized molds, allowed to cool, and to solidify.
- the compounds of the present invention may be formulated for vaginal administration. Pessaries, tampons, creams, gels, pastes, foams or sprays containing in addition to the active ingredient such carriers as are known in the art to be appropriate.
- Suitable formulations along with pharmaceutical carriers, diluents and expcipients are described in Remington: The Science and Practice of Pharmacy 1995, edited by E. W. Martin, Mack Publishing Company, 19th edition, Easton, Pennsylvania, which is hereby incorporated by reference.
- the compounds of the present invention can also be encapsulated in liposomes, such as those disclosed in U.S. Patent Nos. 6,180,134, 5,192,549, 5,376,380, 6,060,080, 6,132,763, each of which is incorporated by reference.
- a skilled formulation scientist may modify the formulations within the teachings of the specification to provide numerous formulations for a particular route of administration without rendering the compositions of the present invention unstable or compromising their therapeutic activity.
- the modification of the present compounds to render them more soluble in water or other vehicle may be easily accomplished by minor modifications (e.g., salt formulation), which are well within the ordinary skill in the art. It is also well within the ordinary skill of the art to modify the route of administration and dosage regimen of a particular compound in order to manage the pharmacokinetics of the present compounds for maximum beneficial effect in patients.
- a seventh embodiment of the present invention is directed to a use of the compound represented by formula I in the manufacture of a medicament for the treatment of any condition the result of an infection by any one of the following viral agents: hepatitis C virus, West Nile virus, yellow fever virus, dengue virus, rhinovirus, polio virus, hepatitis A virus, bovine viral diarrhea virus and Japanese encephalitis virus.
- the term “medicament” means a substance used in a method of treatment and/or prophylaxis of a subject in need thereof, wherein the substance includes, but is not limited to, a composition, a formulation, a dosage form, and the like, comprising the compound of formula I. It is contemplated that the compound of the use of the compound represented by formula I in the manufacture of a medicament for the treatment of any of the antiviral conditions disclosed herein of the seventh embodiment can be any of the compounds contemplated in any of the aspects of the first, second, third, fourth, fifth embodiments or those specifically recited in the tables above or exemplified herein, either alone or in combination with another compound of the present invention.
- a medicament includes, but is not limited to, any one of the compositions contemplated by the sixth embodiment of the present invention.
- a eighth embodiment of the present invention is directed to a method of treatment and/or prophylaxis in a subject in need thereof said method comprises administering a therapeutically effective amount of the compound represented by formula I to the subject.
- a first aspect of the eighth embodiment is directed to a method of treatment and/or prophylaxis in a subject in need thereof said method comprises administering a therapeutically effective of at least two or more different compounds falling within the scope of the compound represented by formula I to the subject.
- a second aspect of the eighth embodiment is directed to a method of treatment and/or prophylaxis in a subject in need thereof said method comprises alternatively or concurrently administering a therapeutically effective of at least two compounds falling within the scope of the compound represented by formula I to the subject.
- a subject in need thereof is one that has any condition the result of an infection by any of the viral agents disclosed herein, which includes, but is not limited to, hepatitis C virus, West Nile virus, yellow fever virus, dengue virus, rhinovirus, polio virus, hepatitis A virus, bovine viral diarrhea virus or Japanese encephalitis virus, flaviviridae viruses or pestiviruses or hepaciviruses or a viral agent causing symptoms equivalent or comparable to any of the above-listed viruses.
- the viral agents disclosed herein includes, but is not limited to, hepatitis C virus, West Nile virus, yellow fever virus, dengue virus, rhinovirus, polio virus, hepatitis A virus, bovine viral diarrhea virus or Japanese encephalitis virus, flaviviridae viruses or pestiviruses or hepaciviruses or a viral agent causing symptoms equivalent or comparable to any of the above-listed viruses.
- subject means a mammal, which includes, but is not limited to, cattle, pigs, sheep, chicken, turkey, buffalo, llama, ostrich, dogs, cats, and humans, preferably the subject is a human. It is contemplated that in the method of treating a subject thereof of the sixth embodiment can be any of the compounds contemplated in any of the aspects of the first, second, and third embodiments or those specifically recited in the tables above, either alone or in combination with another compound of the present invention.
- terapéuticaally effective amount means an amount required to reduce symptoms of the disease in an individual.
- the dose will be adjusted to the individual requirements in each particular case. That dosage can vary within wide limits depending upon numerous factors such as the severity of the disease to be treated, the age and general health condition of the patient, other medicaments with which the patient is being treated, the route and form of administration and the preferences and experience of the medical practitioner involved.
- a daily dosage of between about 0.1 and about 10 g, including all values in between, such as 0.25, 0.5, 0.75, I 5 1.5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, 7, 7.5, 8, 8.5, 9, and 9.5, per day should be appropriate in monotherapy and/or in combination therapy.
- a preferred daily dosage is between about 0.5 and about 7.5 g per day, more preferred 1.5 and about 6.0 g per day.
- treatment is initiated with a large initial "loading dose" to rapidly reduce or eliminate the virus following by a decreasing the dose to a level sufficient to prevent resurgence of the infection.
- Therapeutic efficacy can be ascertained from tests of liver function including, but not limited to protein levels such as serum proteins (e.g., albumin, clotting factors, alkaline phosphatase, aminotransferases (e.g., alanine transaminase, aspartate transaminase), 5 '-nucleosidase, ⁇ - glutaminyltranspeptidase, etc.), synthesis of bilirubin, synthesis of cholesterol, and synthesis of bile acids; a liver metabolic function, including, but not limited to, carbohydrate metabolism, amino acid and ammonia metabolism.
- serum proteins e.g., albumin, clotting factors, alkaline phosphatase, aminotransferases (e.g., alanine transaminase, aspartate transaminase), 5 '-nucleosidase, ⁇ - glutaminyltranspeptidase, etc.)
- the therapeutic effectiveness may be monitored by measuring HCV-RNA. The results of these tests will allow the dose to be optimized.
- a third aspect of the eighth embodiment is directed to a method of treatment and/or prophylaxis in a subject in need thereof said method comprises administering to the subject a therapeutically effective of a compound represented by formula I and a therapeutically effective amount of another antiviral agent; wherein the administration is concurrent or alternative.
- the time between alternative administration can range between 1-24 hours, which includes any sub-range in between including, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, and 23 hours.
- HCV NS3 protease inhibitors see WO 2008010921, WO 2008010921, EP 1881001, WO 2007015824, WO 2007014925, WO 2007014926, WO 2007014921, WO 2007014920, WO 2007014922, US 2005267018, WO 2005095403, WO 2005037214, WO 2004094452, US 2003187018, WO 200364456, WO 2005028502, and WO 2003006490); HCV NS5B Inhibitors (see US 2007275947, US20072759300, WO2007095269, WO 2007092000, WO 2007076034, WO 200702602, US 2005-98125, WO 2006093801, US 2006166964, WO 2006065590, WO 2006065335, US 2006040927, US 2006040890, WO 2006020082, WO 2006012078, WO 2005123087
- HCV NS4 Inhibitors see WO 2007070556 and WO 2005067900
- HCV NS5a Inhibitors see US 2006276511, WO 2006120252, WO 2006120251, WO 2006100310, WO 2006035061
- Toll-like receptor agonists see WO 2007093901
- other inhibitors see WO 2004035571, WO 2004014852, WO 2004014313, WO 2004009020, WO 2003101993, WO 2000006529.
- a fourth aspect of the eighth embodiment is directed to a method of treatment in a subject in need thereof said method comprises alternatively or concurrently administering a therapeutically effective of a compound represented by formula I and another antiviral agent to the subject. It is understood that the time between alternative administration can range between 1-24 hours, which includes any sub-range in between including, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, and 23 hours.
- a fifth aspect of the eighth embodiment is directed to a method of treatment and/or prophylaxis in a subject in need thereof said method comprises administering to the subject a therapeutically effective of at least one compound represented by formula I and a therapeutically effective amount of another antiviral agent; wherein the administration is concurrent or alternative.
- the time between alternative administration can range between 1-24 hours, which includes any sub-range in between including, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, and 23 hours.
- a sixth aspect of the eighth embodiment is directed to a method of treatment in a subject in need thereof said method comprises alternatively or concurrently administering a therapeutically effective of at least one compound represented by formula I and another antiviral agent to the subject. It is understood that the time between alternative administration can range between 1-24 hours, which includes any sub-range in between including, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, and 23 hours.
- the another antiviral agent includes, but is not limited to interferon - ⁇ , interferon- ⁇ , pegylated interferon- ⁇ , ribavirin, levovirin, viramidine, another nucleoside HCV polymerase inhibitor, a HCV non-nucleoside polymerase inhibitor, a HCV protease inhibitor, a HCV helicase inhibitor or a HCV fusion inhibitor.
- the active compound or its derivative or salt are administered in combination with another antiviral agent the activity may be increased over the parent compound.
- the treatment is combination therapy, such administration may be concurrent or sequential with respect to that of the nucleoside derivatives.
- Constant administration thus includes administration of the agents at the same time or at different times. Administration of two or more agents at the same time can be achieved by a single formulation containing two or more active ingredients or by substantially simultaneous administration of two or more dosage forms with a single active agent.
- references herein to treatment extend to prophylaxis as well as to the treatment of existing conditions.
- treatment also includes treatment or prophylaxis of a disease or a condition associated with or mediated by HCV infection, or the clinical symptoms thereof.
- An ninth embodiment of the present invention is directed to a process for preparing the compound of formula I, which comprises reacting a suitably substituted phosphochloridate compound 4 with a nucleoside analog 5
- substituents R 1 , R 2 , R 3a , R 3b , R 4 , R 5 , X, Y, R 6 , and base have their meanings as disclosed in the Detailed Description of the Invention and X' is a leaving group, such as Cl, Br, I, tosylate, mesylate, trifluoroacetate, trifluorosulfonate, pentafluorophenoxide, p-NO 2 -phenoxide, or other commonly used leaving groups as disclosed in Advanced Organic Chemistry by March, Fourth Edition. Leaving groups and methods that can be used to effect the formation of a phosphoramidate nucleoside conjugate are found in US 20060142238 and WO 2007095269.
- the leaving group is Cl.
- This reaction is performed in an anhydrous aprotic solvent such tetrahydrofuran, dioxane, or both tetrahydrofuran and dioxane, or any functional equivalent thereof, with tetrahydrofuran being the preferred solvent.
- the reaction is typically initiated at a temperature range from -78°C to 4O°C with the preferred reaction temperature being between O°C and room temperature.
- the nucleoside is first stirred with a base (5 to 12 equivalents) such as N- methylimidazole, collidine, pyridine, 2,6-lutidine, 2, 6-'Bu-pyridine, etc.
- a tertiary amine base such as triethylamine, diisopropylethylamine, etc.
- an alkyl Grignard reagent such as tBuMgCl, tBuMgBr, MeMgCl, MeMgBr, etc.
- the phosphorochloridate (3-10 equivalents) is dissolved in the reaction solvent and added to the mixture of the nucleoside and base.
- the reaction is then allowed to stir over a period of time at a temperature between room temperature and 4O°C for a period of 30 min to 24 hr. with the preferred reaction temperature being room temperature and time being 24 hr.
- the solvent is removed from the reaction mixture and the product is purified by chromatography on silica gel.
- a tenth embodiment of the present invention is directed to a product obtained by a process which comprises reacting a suitably substituted phosphochloridate compound 4 with a nucleoside analog 5
- This reaction can be performed in an anhydrous aprotic solvent or other suitable solvent, such as tetrahydrofuran, dioxane, or a mixture of tetrahydrofuran and dioxane, with tetrahydrofuran being the preferred solvent.
- the reaction is typically initiated at a temperature range from -78°C to 4O°C with the preferred reaction temperature being between O°C and room temperature.
- the nucleoside is first stirred with a base (5 to 12 equivalents) such as N- methylimidazole, a tertiary amine base or tButyl Magnesium Chloride.
- a phosphorochloridate (3-10 equivalents (or suitable "phosphoro-(leaving group)- date")) is dissolved in the reaction solvent and added to the mixture of the nucleoside and base.
- the reaction is then allowed to stir over a period of time at a temperature between room temperature and 4O°C for a period of 30 min to 24 hr. with the preferred reaction temperature being room temperature and time being 24 hr.
- the solvent is removed from the reaction mixture and the product is purified by chromatography on silica gel.
- Phosphoramidate compounds of the present invention can be prepared by condensation of a nucleoside analog 5 with a suitably substituted phosphochloridate compound 4 (Scheme 1).
- the nucleoside analog is made by conventional procedures disclosed in any one of U.S. Published Application Nos. 2005/0009737, 2006/0199783, 2006/0122146, and 2007/0197463, each of which is incorporated by reference in its entirety.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Virology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Tropical Medicine & Parasitology (AREA)
- Pulmonology (AREA)
- AIDS & HIV (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Saccharide Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
Claims
Priority Applications (37)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0809654-6A BRPI0809654A2 (en) | 2007-03-30 | 2008-03-26 | Compound, its stereoisomer, salt, hydrate, solvate, or crystalline form thereof and process for preparing same and use thereof, composition for treatment and / or prophylaxis of any viral agents, use of compound, method of treating individual |
| ES08732818.3T ES2492470T3 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| DK08732818.3T DK2203462T3 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| EP14179358.8A EP2826784B1 (en) | 2007-03-30 | 2008-03-26 | Composition comprising a HCV NS3 protease inhibitor and a phosphoramidate prodrug of 2'-deoxy-2'-fluoro-2'-C-methyluridine nucleoside |
| BRPI0823519A BRPI0823519A2 (en) | 2007-03-30 | 2008-03-26 | compound, its stereoisomer, composition and use of said compound or stereoisomer |
| CA2682230A CA2682230C (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| CN200880018024.2A CN101918425B (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| EP14169060.2A EP2801580B1 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| SI200831244T SI2203462T1 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| NZ579880A NZ579880A (en) | 2007-03-30 | 2008-03-26 | (S)-2-{ [(2R,3R,4R,5R)-5-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyl-tetrahydro-furan-2-ylmethoxy]-phenoxy-phosphorylamino} -propionic acid isopropyl ester |
| RU2009139968K RU2651892C2 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidates as antiviral agents |
| MX2009010401A MX2009010401A (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs. |
| JP2010502196A JP5318085B2 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrug |
| RU2009139968A RU2651892C3 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidates as antiviral agents |
| CN201811348434.5A CN109776637B (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| KR1020147036573A KR101527701B1 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| CN202310694712.7A CN116731098A (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| KR1020127004146A KR101525293B1 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| PL14179358T PL2826784T3 (en) | 2007-03-30 | 2008-03-26 | Composition comprising a HCV NS3 protease inhibitor and a phosphoramidate prodrug of 2'-deoxy-2'-fluoro-2'-C-methyluridine nucleoside |
| CN202310695479.4A CN116731099A (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| AU2008232827A AU2008232827C1 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| PL08732818T PL2203462T3 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| KR1020097022652A KR101432860B1 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| EP08732818.3A EP2203462B2 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| EP23197008.8A EP4282482A3 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
| ZA2009/06647A ZA200906647B (en) | 2007-03-30 | 2009-09-23 | Nucleoside phosphoramidate prodrugs |
| IL201239A IL201239A (en) | 2007-03-30 | 2009-09-29 | (s)-2-{[(2r,3r,4r,5r)-5-(2,4-dioxo-3,4-dihydro-2h-pyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyl-tetrahydrofuran-2-ylmethoxy]-phenoxy-phosphorylamino}-propionic acid isopropyl ester or a stereoisomer thereof, (s)-isopropyl 2-(((s)-((2r,3r,4r,5r)-5-(2,4-dioxo-3,4-dihydropyrimidin-yl)methoxy)(phenoxy)phosphoryl)amino)propanoate, a method of synthesizing the nucleotides and uses thereof |
| IL217228A IL217228A (en) | 2007-03-30 | 2011-12-27 | Nucleoside phosphoramidate prodrugs of 2'-deoxy-2'-fluoro-2'-c-methyluridine |
| IL222810A IL222810A0 (en) | 2007-03-30 | 2012-11-01 | Nucleoside phosphorramidate prodrugs |
| HRP20140667AT HRP20140667T1 (en) | 2007-03-30 | 2014-07-14 | Nucleoside phosphoramidate prodrugs |
| CY2014047C CY2014047I1 (en) | 2007-03-30 | 2014-11-18 | NUCLEOSIDE PHOSPHORIMIDE PRODRUGS |
| NL300704C NL300704I2 (en) | 2007-03-30 | 2014-11-19 | |
| LU92600C LU92600I2 (en) | 2007-03-30 | 2014-11-19 | SOVALDI (SOFOSBUVIR) |
| FR14C0082C FR14C0082I2 (en) | 2007-03-30 | 2014-11-19 | NUCLEOSIDE PHOSPHORAMIDATE PRODRUGS |
| LTPA2014040C LTC2203462I2 (en) | 2007-03-30 | 2014-11-21 | Prodrugs of Nucleoside Phosphoramidate |
| PH12014502771A PH12014502771A1 (en) | 2007-03-30 | 2014-12-10 | Nucleoside phosphoramidate prodrugs |
| NO2021031C NO2021031I1 (en) | 2007-03-30 | 2021-07-26 | Sofosbuvir - extended SPC |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US90931507P | 2007-03-30 | 2007-03-30 | |
| US60/909,315 | 2007-03-30 | ||
| US98230907P | 2007-10-24 | 2007-10-24 | |
| US60/982,309 | 2007-10-24 | ||
| US12/053,015 | 2008-03-21 | ||
| US12/053,015 US7964580B2 (en) | 2007-03-30 | 2008-03-21 | Nucleoside phosphoramidate prodrugs |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2008121634A2 true WO2008121634A2 (en) | 2008-10-09 |
| WO2008121634A3 WO2008121634A3 (en) | 2010-05-20 |
Family
ID=39808855
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2008/058183 WO2008121634A2 (en) | 2007-03-30 | 2008-03-26 | Nucleoside phosphoramidate prodrugs |
Country Status (36)
| Country | Link |
|---|---|
| US (11) | US7964580B2 (en) |
| EP (7) | EP2933260A1 (en) |
| JP (8) | JP5318085B2 (en) |
| KR (3) | KR101525293B1 (en) |
| CN (8) | CN109776637B (en) |
| AR (1) | AR066898A1 (en) |
| AU (1) | AU2008232827C1 (en) |
| BR (2) | BRPI0823519A2 (en) |
| CA (1) | CA2682230C (en) |
| CL (1) | CL2008000902A1 (en) |
| CO (1) | CO6260023A2 (en) |
| CY (2) | CY1115488T1 (en) |
| DE (1) | DE202008018643U1 (en) |
| DK (1) | DK2203462T3 (en) |
| ES (3) | ES2492470T3 (en) |
| FR (1) | FR14C0082I2 (en) |
| HK (2) | HK1206033A1 (en) |
| HR (1) | HRP20140667T1 (en) |
| HU (1) | HUS1400059I1 (en) |
| IL (3) | IL201239A (en) |
| LT (1) | LTC2203462I2 (en) |
| LU (1) | LU92600I2 (en) |
| MX (1) | MX2009010401A (en) |
| MY (1) | MY147409A (en) |
| NL (1) | NL300704I2 (en) |
| NO (2) | NO2014029I1 (en) |
| NZ (2) | NZ579880A (en) |
| PH (1) | PH12014502771A1 (en) |
| PL (3) | PL2203462T3 (en) |
| PT (3) | PT2801580T (en) |
| RU (4) | RU2651892C2 (en) |
| SG (2) | SG2014011704A (en) |
| SI (3) | SI2826784T1 (en) |
| TW (1) | TWI357332B (en) |
| WO (1) | WO2008121634A2 (en) |
| ZA (2) | ZA200906647B (en) |
Cited By (121)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010075517A2 (en) | 2008-12-23 | 2010-07-01 | Pharmasset, Inc. | Nucleoside analogs |
| WO2010075554A1 (en) | 2008-12-23 | 2010-07-01 | Pharmasset, Inc. | Synthesis of purine nucleosides |
| WO2010075549A2 (en) | 2008-12-23 | 2010-07-01 | Pharmasset, Inc. | Nucleoside phosphoramidates |
| WO2010135569A1 (en) | 2009-05-20 | 2010-11-25 | Pharmasset, Inc. | N- [ (2 ' r) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -l-alanine 1-methylethyl ester and process for its production |
| WO2011005860A2 (en) | 2009-07-07 | 2011-01-13 | Alnylam Pharmaceuticals, Inc. | 5' phosphate mimics |
| WO2011123672A1 (en) | 2010-03-31 | 2011-10-06 | Pharmasset, Inc. | Purine nucleoside phosphoramidate |
| WO2011123645A2 (en) | 2010-03-31 | 2011-10-06 | Pharmasset, Inc. | Nucleoside phosphoramidates |
| EP2393815A2 (en) * | 2009-02-06 | 2011-12-14 | RFS Pharma, LLC. | Purine nucleoside monophosphate prodrugs for treatment of cancer and viral infections |
| WO2012012465A1 (en) * | 2010-07-19 | 2012-01-26 | Clarke, Michael, O'neil Hanrahan | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| WO2012142523A2 (en) | 2011-04-13 | 2012-10-18 | Gilead Sciences, Inc. | 1'-substituted pyrimidine n-nucleoside analogs for antiviral treatment |
| WO2012158811A3 (en) * | 2011-05-19 | 2013-02-28 | Rfs Pharma, Llc | Purine monophosphate prodrugs for treatment of viral infections |
| WO2013039920A1 (en) * | 2011-09-12 | 2013-03-21 | Idenix Pharmaceuticals, Inc. | Substituted carbonyloxymethylphosphoramidate compounds and pharmaceutical compositions for the treatment of viral infections |
| JP2013514276A (en) * | 2009-12-18 | 2013-04-25 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | HCV combination therapy |
| EP2583680A3 (en) * | 2011-10-21 | 2013-06-12 | Abbvie Inc. | Mono (PSI-7977) or combination treatment of DAAs for use in treating HCV |
| CN103209987A (en) * | 2010-09-22 | 2013-07-17 | 艾丽奥斯生物制药有限公司 | Substituted nucleotide analogs |
| WO2013177219A1 (en) * | 2012-05-22 | 2013-11-28 | Idenix Pharmaceuticals, Inc. | D-amino acid compounds for liver disease |
| US8618076B2 (en) | 2009-05-20 | 2013-12-31 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| WO2014008236A1 (en) | 2012-07-03 | 2014-01-09 | Bristol-Myers Squibb Company | Process for preparing diastereomerically enriched phosphoramidate derivatives of nucleoside compounds for treatment of viral infections |
| WO2014058801A1 (en) * | 2012-10-08 | 2014-04-17 | Idenix Pharmaceuticals, Inc. | 2'-chloro nucleoside analogs for hcv infection |
| WO2014078427A1 (en) * | 2012-11-14 | 2014-05-22 | Idenix Pharmaceuticals, Inc. | D-alanine ester of rp-nucleoside analog |
| WO2014078436A1 (en) * | 2012-11-14 | 2014-05-22 | Idenix Pharmaceuticals, Inc. | D-alanine ester of sp-nucleoside analog |
| WO2014099941A1 (en) * | 2012-12-19 | 2014-06-26 | Idenix Pharmaceuticals, Inc. | 4'-fluoro nucleosides for the treatment of hcv |
| WO2014100505A1 (en) | 2012-12-21 | 2014-06-26 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| PT107665A (en) * | 2011-10-21 | 2014-07-16 | Abbvie Inc | METHODS FOR THE TREATMENT OF HCV COMPREHENDING AT LEAST TWO ANTIVIRAL AGENTS OF DIRECT ACTIVITY, RIBAVIRIN, BUT NOT INTERFERED |
| US8815829B2 (en) | 2008-12-09 | 2014-08-26 | Rfs Pharma, Llc | 3′-azido purine nucleotide prodrugs for treatment of viral infections |
| US8822496B2 (en) | 2009-10-30 | 2014-09-02 | Boehringer Ingelheim International Gmbh | Dosage regimens for HCV combination therapy |
| WO2014137926A1 (en) * | 2013-03-04 | 2014-09-12 | Idenix Pharmaceuticals, Inc. | 3'-deoxy nucleosides for the treatment of hcv |
| WO2014135107A1 (en) | 2013-03-08 | 2014-09-12 | 南京圣和药业有限公司 | Novel nucleoside phosphoramidate compound and use thereof |
| US8841275B2 (en) | 2010-11-30 | 2014-09-23 | Gilead Pharmasset Llc | 2′-spiro-nucleosides and derivatives thereof useful for treating hepatitis C virus and dengue virus infections |
| WO2014148949A1 (en) * | 2013-03-22 | 2014-09-25 | Асави, Ллс | Alkyl 2-{[(2r,3s,5r)-5-(4-amino-2-oxo-2н-pyrimidin-1-yl)-3-hydroxy- tetrahydro-furan-2-yl-methoxy]-phenoxy-phosphoryl-amino}-propionates, nucleoside inhibitors of hcv ns5b rna-polymerase, and methods for producing and use thereof |
| US8853171B2 (en) | 2008-04-23 | 2014-10-07 | Gilead Sciences, Inc. | 1′-substituted carba-nucleoside analogs for antiviral treatment |
| WO2014165542A1 (en) * | 2013-04-01 | 2014-10-09 | Idenix Pharmaceuticals, Inc. | 2',4'-fluoro nucleosides for the treatment of hcv |
| US8877731B2 (en) | 2010-09-22 | 2014-11-04 | Alios Biopharma, Inc. | Azido nucleosides and nucleotide analogs |
| US8889159B2 (en) | 2011-11-29 | 2014-11-18 | Gilead Pharmasset Llc | Compositions and methods for treating hepatitis C virus |
| WO2014186637A1 (en) | 2013-05-16 | 2014-11-20 | Riboscience Llc | 4'-fluor0-2'-methyl substituted nucleoside derivatives |
| WO2014197578A1 (en) * | 2013-06-05 | 2014-12-11 | Idenix Pharmaceuticals, Inc. | 1',4'-thio nucleosides for the treatment of hcv |
| US8933053B2 (en) | 2011-03-01 | 2015-01-13 | Nucana Biomed Limited | Phosphoramidate derivatives of 5-fluoro-2′-deoxyuridine for use in the treatment of cancer |
| JP2015503506A (en) * | 2011-12-22 | 2015-02-02 | アリオス バイオファーマ インク. | Substituted nucleosides, nucleotides and analogs thereof |
| WO2015017713A1 (en) * | 2013-08-01 | 2015-02-05 | Idenix Pharmaceuticals, Inc. | D-amino acid phosphoramidate pronucleotides of halogeno pyrimidine compounds for liver disease |
| WO2015034420A1 (en) | 2013-09-04 | 2015-03-12 | Medivir Ab | Hcv polymerase inhibitors |
| WO2015053662A1 (en) * | 2013-10-11 | 2015-04-16 | Андрей Александрович ИВАЩЕНКО | Substituted (2r, 3r, 5r)-3-hydroxy-(5-pyrimidin-1-yl) tetrahydrofuran-2-ylmethyl aryl phosphoramidates |
| US9012427B2 (en) | 2012-03-22 | 2015-04-21 | Alios Biopharma, Inc. | Pharmaceutical combinations comprising a thionucleotide analog |
| WO2015056213A1 (en) | 2013-10-17 | 2015-04-23 | Medivir Ab | Hcv polymerase inhibitors |
| WO2015061683A1 (en) * | 2013-10-25 | 2015-04-30 | Idenix Pharmaceuticals, Inc. | D-amino acid phosphoramidate and d-alanine thiophosphoramidate pronucleotides of nucleoside compounds useful for the treatment of hcv |
| CN104650171A (en) * | 2013-11-25 | 2015-05-27 | 天津市汉康医药生物技术有限公司 | Sofosbuvir sesquihydrate compound |
| WO2015081297A1 (en) * | 2013-11-27 | 2015-06-04 | Idenix Pharmaceuticals, Inc. | 2'-dichloro and 2'-fluoro-2'-chloro nucleoside analogues for hcv infection |
| RU2553996C1 (en) * | 2013-11-27 | 2015-06-20 | Андрей Александрович Иващенко | Substituted (2r,3r,5r)-3-hydroxy-(5-pyrimidin-1-yl)tetrahydrofuran-2-ylmethyl aryl phosphoramidates |
| WO2015097605A1 (en) | 2013-12-23 | 2015-07-02 | Mylan Laboratories Ltd. | Process for the preparation of sofosbuvir |
| WO2015099989A1 (en) | 2013-12-23 | 2015-07-02 | Gilead Pharmasset Llc | Crystalline forms of antiviral sofosbuvir analogues |
| WO2015101183A1 (en) * | 2014-01-02 | 2015-07-09 | 江苏豪森药业股份有限公司 | Uracil nucleotide analogs, preparation methods therefor and uses thereof |
| US9085573B2 (en) | 2007-03-30 | 2015-07-21 | Gilead Pharmasset Llc | Nucleoside phosphoramidate prodrugs |
| WO2015158317A1 (en) | 2014-04-15 | 2015-10-22 | Zentiva, K.S. | Use of a l,3j5-triazin-2-yl phosphoramidate compound in the synthesis of sofosbuvir |
| WO2015164812A1 (en) * | 2014-04-24 | 2015-10-29 | Cocrystal Pharma, Inc. | 2' -disubstituted nucleoside analogs for treatment of the flaviviridae family of viruses and cancer |
| RU2567854C1 (en) * | 2014-11-11 | 2015-11-10 | Александр Васильевич Иващенко | Nucleoside inhibitors of rna-polymerase hcv ns5b, methods for production and use thereof |
| WO2016008461A1 (en) | 2014-07-17 | 2016-01-21 | Zentiva, K.S. | A new form of sofosbuvir and a method of its preparation |
| AU2014274548B2 (en) * | 2009-05-20 | 2016-02-04 | Gilead Sciences, Inc. | N- [ (2 ' R) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -L-alanine 1-methylethyl ester and process for its production |
| WO2016035006A1 (en) * | 2014-09-01 | 2016-03-10 | Dr. Reddy’S Laboratories Limited | Novel nucleotide analogs, process for the preparation of sofosbuvir and its analogs, novel forms of sofosbuvir and solid dispersion of sofosbuvir |
| WO2016042576A1 (en) | 2014-09-16 | 2016-03-24 | Cadila Healthcare Limited | Co-crystal of sofosbuvir and amino acid and process for preparation thereof |
| US9296777B2 (en) | 2007-11-20 | 2016-03-29 | Gilead Pharmasset Llc | 2′,4′-substituted nucleosides as antiviral agents |
| US9393256B2 (en) | 2011-09-16 | 2016-07-19 | Gilead Pharmasset Llc | Methods for treating HCV |
| WO2016128453A1 (en) | 2015-02-13 | 2016-08-18 | Sandoz Ag | Pharmaceutical compositions comprising ledipasvir and sofosbuvir |
| US9422323B2 (en) | 2012-05-25 | 2016-08-23 | Janssen Sciences Ireland Uc | Uracyl spirooxetane nucleosides |
| ITUB20150109A1 (en) * | 2015-03-05 | 2016-09-05 | Hc Pharma Ag | USEFUL COMPOUNDS FOR THE TREATMENT OF HEPATITIS C |
| US9441007B2 (en) | 2012-03-21 | 2016-09-13 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| WO2016151542A1 (en) * | 2015-03-26 | 2016-09-29 | Quimica Sintetica, S.A. | Nucleoside phosphoramidates useful for the treatment of viral infections and preparation thereof |
| WO2016177300A1 (en) * | 2015-05-07 | 2016-11-10 | 苏州旺山旺水生物医药有限公司 | (2'r)-2'-deoxy-2'-halo-2'-methyl uridine derivative, and preparation method and use thereof |
| WO2016189040A1 (en) | 2015-05-26 | 2016-12-01 | Sandoz Ag | Selective process for synthesis of nucleoside phosphoramidates |
| WO2016196735A2 (en) | 2015-06-03 | 2016-12-08 | Teva Pharmaceuticals International Gmbh | Improved processes for the preparation of sofosbuvir and intermediates thereof |
| WO2016206663A1 (en) | 2015-06-26 | 2016-12-29 | Zentiva, K.S. | A pharmaceutical formulation of sofosbuvir |
| WO2016189443A3 (en) * | 2015-05-23 | 2017-01-12 | Virupaksha Organics Limited | Solid forms of nucleoside phosphoramidate |
| ITUB20152109A1 (en) * | 2015-07-13 | 2017-01-13 | Quim Sintetica S A | PHOSPHORAMIDATE NUCLEOSIDES USEFUL FOR THE TREATMENT OF VIRAL INFECTIONS AND THEIR PREPARATION |
| WO2017077552A1 (en) | 2015-11-03 | 2017-05-11 | Mylan Laboratories Limited | Process for the preparation of sofosbuvir |
| AU2016200676B2 (en) * | 2009-05-20 | 2017-06-01 | Gilead Sciences, Inc. | N-[(2'R)-2 '-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl] -L-alanine 1-methylethyl ester and process for its production |
| US9676797B2 (en) | 2015-09-02 | 2017-06-13 | Abbvie Inc. | Anti-viral compounds |
| KR101765997B1 (en) | 2011-12-20 | 2017-08-07 | 리보사이언스 엘엘씨 | 2',4'-difluoro-2'-methyl substituted nucleoside derivatives as inhibitors of hcv rna replication |
| US9724360B2 (en) | 2014-10-29 | 2017-08-08 | Gilead Sciences, Inc. | Methods for treating Filoviridae virus infections |
| WO2017162169A1 (en) * | 2016-03-25 | 2017-09-28 | 江苏天士力帝益药业有限公司 | Uridine phosphoramide prodrug, preparation method therefor, and medicinal uses thereof |
| WO2017184670A2 (en) | 2016-04-22 | 2017-10-26 | Gilead Sciences, Inc. | Methods for treating zika virus infections |
| WO2017189978A1 (en) | 2016-04-28 | 2017-11-02 | Emory University | Alkyne containing nucleotide and nucleoside therapeutic compositions and uses related thereto |
| WO2017190715A1 (en) | 2016-05-05 | 2017-11-09 | Zentiva, K.S. | An amorphous form of sofosbuvir, a method of its preparation and its stabilization |
| WO2017205078A1 (en) | 2016-05-27 | 2017-11-30 | Gilead Sciences, Inc. | Methods for treating hepatitis b virus infections using ns5a, ns5b or ns3 inhibitors |
| US9862743B2 (en) | 2013-10-11 | 2018-01-09 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| WO2018013937A1 (en) | 2016-07-14 | 2018-01-18 | Atea Pharmaceuticals, Inc. | Beta-d-2'-deoxy-2'-alpha-fluoro-2'-beta-c-substituted-4'-fluoro-n6-substituted-6-amino-2-substituted purine nucleotides for the treatment of hepatitis c virus infection |
| RU2644156C1 (en) * | 2017-02-28 | 2018-02-08 | Александр Васильевич Иващенко | Prodrug of polymerase ns5b hcv inhibitor, method for its preparation and application |
| WO2018033593A1 (en) | 2016-08-19 | 2018-02-22 | Sandoz Ag | Sofosbuvir derivatives for the treatment of hepatitis c |
| RU2659388C1 (en) * | 2017-02-28 | 2018-07-02 | Васильевич Иващенко Александр | Nucleotides including n-[(s)-1-cyclobutoxycarbonyl]phosphoramidate fragment, their analogs and their application |
| WO2018134343A1 (en) | 2017-01-19 | 2018-07-26 | Sandoz Ag | Synthesis of phosphoramidates |
| US10039779B2 (en) | 2013-01-31 | 2018-08-07 | Gilead Pharmasset Llc | Combination formulation of two antiviral compounds |
| US10065958B2 (en) | 2010-07-22 | 2018-09-04 | Gilead Sciences, Inc. | Methods and compounds for treating Paramyxoviridae virus infections |
| WO2019025600A1 (en) | 2017-08-03 | 2019-02-07 | Sandoz Ag | Sofosbuvir hydrate |
| US10202412B2 (en) | 2016-07-08 | 2019-02-12 | Atea Pharmaceuticals, Inc. | β-D-2′-deoxy-2′-substituted-4′-substituted-2-substituted-N6-substituted-6-aminopurinenucleotides for the treatment of paramyxovirus and orthomyxovirus infections |
| US10214553B2 (en) | 2014-06-13 | 2019-02-26 | Teva Pharmaceuticals International Gmbh | Solid state forms of sofosbuvir |
| US10239910B2 (en) | 2016-07-20 | 2019-03-26 | Optimus Drugs (P) Limited | Process for the preparation of sofosbuvir |
| US10251904B2 (en) | 2015-09-16 | 2019-04-09 | Gilead Sciences, Inc. | Methods for treating arenaviridae and coronaviridae virus infections |
| EP3331354A4 (en) * | 2016-07-28 | 2019-08-07 | Asavi, LLC | Phosphoramidate nucleoside prodrug for treating viral diseases and cancer, processes for their preparation and their use |
| EP3377512A4 (en) * | 2015-11-16 | 2019-09-18 | Ichorion Therapeutics, Inc. | Nucleic acid prodrugs |
| US10526363B2 (en) | 2014-08-15 | 2020-01-07 | Merck Sharp & Dohme Corp. | Substituted phosphoramidate compounds and uses thereof |
| US10675296B2 (en) | 2017-07-11 | 2020-06-09 | Gilead Sciences, Inc. | Compositions comprising an RNA polymerase inhibitor and cyclodextrin for treating viral infections |
| US10682368B2 (en) | 2017-03-14 | 2020-06-16 | Gilead Sciences, Inc. | Methods of treating feline coronavirus infections |
| US10682369B2 (en) | 2017-09-21 | 2020-06-16 | Riboscience Llc | 4′-fluoro-2′-methyl substituted nucleoside derivatives as inhibitors of HCV RNA replication |
| US10696708B2 (en) | 2013-06-26 | 2020-06-30 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| USRE48171E1 (en) | 2012-03-21 | 2020-08-25 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US10836787B2 (en) | 2017-05-01 | 2020-11-17 | Gilead Sciences, Inc. | Crystalline forms of (S)-2-ethylbutyl 2-(((S)-(((2R,3S,4R,5R)-5- (4-aminopyrrolo[2,1-f] [1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxytetrahydrofuran-2-yl)methoxy)(phenoxy) phosphoryl)amino)propanoate |
| EP3750544A2 (en) | 2011-11-30 | 2020-12-16 | Emory University | Jak inhibitors for use in the prevention or treatment of viral infection |
| US10912814B2 (en) | 2016-06-02 | 2021-02-09 | Gilead Pharmasset Llc | Combination formulation of three antiviral compounds |
| US10953030B2 (en) | 2013-05-16 | 2021-03-23 | Riboscience Llc | 4′-fluoro-2′-methyl substituted nucleoside derivatives as inhibitors of HCV RNA replication |
| US10988498B2 (en) | 2009-09-21 | 2021-04-27 | Gilead Sciences, Inc. | Processes and intermediates for the preparation of 1′-substituted carba-nucleoside analogs |
| US11116783B2 (en) | 2013-08-27 | 2021-09-14 | Gilead Pharmasset Llc | Combination formulation of two antiviral compounds |
| WO2021202669A2 (en) | 2020-04-01 | 2021-10-07 | Reyoung Corporation | Nucleoside and nucleotide conjugate compounds and uses thereof |
| US11203599B2 (en) | 2014-06-11 | 2021-12-21 | Gilead Pharmasset Llc | Solid forms of an antiviral compound |
| RU2764767C2 (en) * | 2015-03-06 | 2022-01-21 | Атеа Фармасьютикалс, Инк. | β-D-2ʹ-DEOXY-2ʹ-α-FLUORO-2ʹ-β-C-SUBSTITUTED-2-MODIFIED-N6-SUBSTITUTED PURINE NUCLEOTIDES FOR TREATMENT OF DISEASES CAUSED BY HCV |
| US11364257B2 (en) | 2016-06-24 | 2022-06-21 | Emory University | Phosphoramidates for the treatment of hepatitis B virus |
| US11414451B2 (en) | 2017-09-18 | 2022-08-16 | NuCana plc | Floxuridine synthesis |
| US11447518B2 (en) | 2014-12-15 | 2022-09-20 | Emory University | Phosphoramidates for the treatment of hepatitis B virus |
| US11491169B2 (en) | 2020-05-29 | 2022-11-08 | Gilead Sciences, Inc. | Remdesivir treatment methods |
| US11613553B2 (en) | 2020-03-12 | 2023-03-28 | Gilead Sciences, Inc. | Methods of preparing 1′-cyano nucleosides |
| US11660307B2 (en) | 2020-01-27 | 2023-05-30 | Gilead Sciences, Inc. | Methods for treating SARS CoV-2 infections |
| US11701372B2 (en) | 2020-04-06 | 2023-07-18 | Gilead Sciences, Inc. | Inhalation formulations of 1'-cyano substituted carba-nucleoside analogs |
| US11780844B2 (en) | 2022-03-02 | 2023-10-10 | Gilead Sciences, Inc. | Compounds and methods for treatment of viral infections |
| US11814406B2 (en) | 2020-08-27 | 2023-11-14 | Gilead Sciences, Inc. | Compounds and methods for treatment of viral infections |
| US11939347B2 (en) | 2020-06-24 | 2024-03-26 | Gilead Sciences, Inc. | 1′-cyano nucleoside analogs and uses thereof |
Families Citing this family (134)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MY164523A (en) | 2000-05-23 | 2017-12-29 | Univ Degli Studi Cagliari | Methods and compositions for treating hepatitis c virus |
| CA2410579C (en) | 2000-05-26 | 2010-04-20 | Jean-Pierre Sommadossi | Methods and compositions for treating flaviviruses and pestiviruses |
| KR100828453B1 (en) | 2001-01-22 | 2008-05-13 | 머크 앤드 캄파니 인코포레이티드 | Nucleoside derivatives as inhibitors of RNA-dependent RNA viral polymerase |
| MXPA05005192A (en) | 2002-11-15 | 2005-09-08 | Idenix Cayman Ltd | 2aCOE-BRANCHED NUCLEOSIDES AND FLAVIVIRIDAE. |
| BRPI0419345B8 (en) | 2003-05-30 | 2021-05-25 | Gilead Pharmasset Llc | use of (2'r)-2'-deoxy-2'-fluor-2'-c-methyl nucleoside and a pharmaceutical composition comprising it |
| WO2006063149A1 (en) | 2004-12-09 | 2006-06-15 | Regents Of The University Of Minnesota | Nucleosides with antiviral and anticancer activity |
| GB0623493D0 (en) | 2006-11-24 | 2007-01-03 | Univ Cardiff | Chemical compounds |
| US8173621B2 (en) | 2008-06-11 | 2012-05-08 | Gilead Pharmasset Llc | Nucleoside cyclicphosphates |
| EA201190178A1 (en) * | 2009-03-20 | 2012-06-29 | Алиос Биофарма, Инк. | REPLACED NUCLEOSIDE AND NUCLEOTIC ANALOGUES |
| EP3626716A1 (en) | 2009-05-13 | 2020-03-25 | Gilead Pharmasset LLC | Antiviral compounds |
| EP2483288B1 (en) * | 2009-09-29 | 2016-11-16 | Janssen Products, L.P. | Phosphoramidate derivatives of nucleosides |
| US8563530B2 (en) | 2010-03-31 | 2013-10-22 | Gilead Pharmassel LLC | Purine nucleoside phosphoramidate |
| WO2011133212A1 (en) * | 2010-04-20 | 2011-10-27 | New York University | Methods, compounds and pharmaceutical compositions for treating anxiety and mood disorders |
| WO2012094248A1 (en) * | 2011-01-03 | 2012-07-12 | Nanjing Molecular Research, Inc. | O-(substituted benzyl) phosphoramidate compounds and therapeutic use |
| WO2012125900A1 (en) | 2011-03-16 | 2012-09-20 | Enanta Pharmaceuticals, Inc. | 2'-allene-substituted nucleoside derivatives |
| CN103842369A (en) | 2011-03-31 | 2014-06-04 | 埃迪尼克斯医药公司 | Compounds and pharmaceutical compositions for the treatment of viral infections |
| US20130018011A1 (en) * | 2011-06-10 | 2013-01-17 | Hassan Javanbakht | Method of treating dengue fever |
| EP2734535A4 (en) * | 2011-07-19 | 2014-10-22 | Nanjing Molecular Res Inc | 2',3'-DIDEOXY-2'-alpha-FLUORO-2'-beta-C-METHYLNUCLEOSIDES AND PRODRUGS THEREOF |
| CN107353316B (en) | 2011-09-30 | 2020-08-18 | 塔夫斯大学 | Uridine diphosphate derivatives, compositions and methods for treating neurodegenerative diseases |
| EP2768838A1 (en) | 2011-10-14 | 2014-08-27 | IDENIX Pharmaceuticals, Inc. | Substituted 3',5'-cyclic phosphates of purine nucleotide compounds and pharmaceutical compositions for the treatment of viral infections |
| US8492386B2 (en) | 2011-10-21 | 2013-07-23 | Abbvie Inc. | Methods for treating HCV |
| US8466159B2 (en) | 2011-10-21 | 2013-06-18 | Abbvie Inc. | Methods for treating HCV |
| BR112014010295A2 (en) | 2011-10-31 | 2017-04-18 | Gilead Pharmassett Llc | methods and compositions for the treatment of hepatitis c virus |
| MD4430C1 (en) | 2011-11-29 | 2017-03-31 | Gilead Pharmasset Llc | Compositions and methods for treating hepatitis C virus |
| SI2794628T1 (en) * | 2011-12-20 | 2017-07-31 | Riboscience Llc | 4'-azido-3'-fluoro substituted nucleoside derivatives as inhibitors of hcv rna replication |
| US8980865B2 (en) | 2011-12-22 | 2015-03-17 | Alios Biopharma, Inc. | Substituted nucleotide analogs |
| MX2014008008A (en) * | 2011-12-29 | 2014-08-21 | Abbvie Inc | Solid compositions comprising an hcv inhibitor. |
| US9303652B2 (en) | 2012-01-05 | 2016-04-05 | Archipelago Group Llc | Electric blower operable to provide combustion air to a fire |
| EP2814816A2 (en) | 2012-02-14 | 2014-12-24 | University Of Georgia Research Foundation, Inc. | Spiro [2.4]heptanes for treatment of flaviviridae infections |
| US8916538B2 (en) | 2012-03-21 | 2014-12-23 | Vertex Pharmaceuticals Incorporated | Solid forms of a thiophosphoramidate nucleotide prodrug |
| WO2013173759A2 (en) * | 2012-05-17 | 2013-11-21 | Enanta Pharmaceuticals, Inc. | Macrocyclic nucleoside phosphoramidate derivatives |
| US9296778B2 (en) | 2012-05-22 | 2016-03-29 | Idenix Pharmaceuticals, Inc. | 3′,5′-cyclic phosphate prodrugs for HCV infection |
| WO2013177188A1 (en) | 2012-05-22 | 2013-11-28 | Idenix Pharmaceuticals, Inc. | 3',5'-cyclic phosphoramidate prodrugs for hcv infection |
| US9206412B2 (en) * | 2012-05-31 | 2015-12-08 | Colorado State University Research Foundation | Thioxothiazolidine inhibitors |
| WO2013187978A1 (en) * | 2012-06-16 | 2013-12-19 | Nanjing Molecular Research, Inc. | Double-liver-targeting phosphoramidate and phosphonoamidate prodrugs |
| WO2014047117A1 (en) | 2012-09-18 | 2014-03-27 | Bristol-Myers Squibb Company | Process for preparing phosphoramidate derivatives of nucleoside compounds for treatment of viral infections |
| EP2900682A1 (en) | 2012-09-27 | 2015-08-05 | IDENIX Pharmaceuticals, Inc. | Esters and malonates of sate prodrugs |
| JP6523958B2 (en) | 2012-09-28 | 2019-06-05 | タフツ・ユニバーシティ | Uridine diphosphate derivatives, prodrugs, compositions and their use |
| US10723754B2 (en) | 2012-10-22 | 2020-07-28 | Idenix Pharmaceuticals Llc | 2′,4′-bridged nucleosides for HCV infection |
| MX2015006195A (en) * | 2012-11-16 | 2015-12-08 | Univ Cardiff | Process for preparing nucleoside prodrugs. |
| US10034893B2 (en) * | 2013-02-01 | 2018-07-31 | Enanta Pharmaceuticals, Inc. | 5, 6-D2 uridine nucleoside/tide derivatives |
| US20140249101A1 (en) | 2013-03-04 | 2014-09-04 | Gilead Pharmasset Llc | Methods for treating hepatitis c virus infection |
| US9339541B2 (en) | 2013-03-04 | 2016-05-17 | Merck Sharp & Dohme Corp. | Thiophosphate nucleosides for the treatment of HCV |
| EP2970357A1 (en) | 2013-03-13 | 2016-01-20 | IDENIX Pharmaceuticals, Inc. | Amino acid phosphoramidate pronucleotides of 2'-cyano, azido and amino nucleosides for the treatment of hcv |
| JP6591957B2 (en) | 2013-03-13 | 2019-10-16 | タフツ・ユニバーシティ | Uridine nucleoside derivatives, compositions and methods of use |
| US10138265B2 (en) | 2013-03-13 | 2018-11-27 | Tufts University | Uridine nucleoside derivatives, compositions and methods of use |
| EP2984097B1 (en) | 2013-04-12 | 2017-03-29 | Achillion Pharmaceuticals, Inc. | Highly active nucleoside derivative for the treatment of hcv |
| WO2014197400A1 (en) | 2013-06-04 | 2014-12-11 | Gilead Pharmasset Llc | Preventing and treating recurrence of hcv infection after liver transplant |
| CN104231023B (en) * | 2013-06-06 | 2019-02-05 | 南京汇诚制药有限公司 | Tricyclic fused heterocycle nucleoside phosphoramidate compound, preparation method and application |
| DK3043803T3 (en) | 2013-09-11 | 2022-08-01 | Univ Emory | Nucleotide and nucleoside compounds and their applications |
| US9943604B2 (en) | 2013-09-20 | 2018-04-17 | Ionis Pharmaceuticals, Inc. | Targeted therapeutic nucleosides and their use |
| CN104447923B (en) * | 2013-09-23 | 2018-03-30 | 中国药科大学 | Methyl nucleoside derivatives of 2 ' deoxidation, 2 ' fluorine 2 ' and preparation method thereof and the purposes in pharmacy |
| WO2015084741A2 (en) | 2013-12-02 | 2015-06-11 | Gilead Pharmasset Llc | Methods of treating hepatitis c virus infection in subjects with cirrhosis |
| US20170015696A1 (en) | 2014-02-20 | 2017-01-19 | Ratiopharm Gmbh | Solid state forms of sofosbuvir |
| CN103804446A (en) * | 2014-02-27 | 2014-05-21 | 苏州东南药业股份有限公司 | Preparation method of 3,5-dibenzoyl-2-deoxy-2-fluoro-2-methyl-D-ribo-gamma-lactone |
| US20170066779A1 (en) | 2014-03-05 | 2017-03-09 | Idenix Pharmaceuticals Llc | Solid forms of a flaviviridae virus inhibitor compound and salts thereof |
| US20170135990A1 (en) | 2014-03-05 | 2017-05-18 | Idenix Pharmaceuticals Llc | Pharmaceutical compositions comprising a 5,5-fused heteroarylene flaviviridae inhibitor and their use for treating or preventing flaviviridae infection |
| US20170066795A1 (en) * | 2014-03-05 | 2017-03-09 | Idenix Pharmaceuticals Llc | Solid prodrug forms of 2'-chloro-2'-methyl uridine for hcv |
| EP3131914B1 (en) | 2014-04-16 | 2023-05-10 | Idenix Pharmaceuticals LLC | 3'-substituted methyl or alkynyl nucleosides for the treatment of hcv |
| EA201692507A1 (en) | 2014-06-23 | 2017-04-28 | Сановель Илач Санайи Ве Тиджарет А.Ш. | PHARMACEUTICAL COMBINATIONS OF SOFOSBUVIR AND RIBAVIRIN |
| US20170151272A1 (en) | 2014-06-23 | 2017-06-01 | Sanovel Ilac Sanayi Ve Ticaret Anonim Sirketi | A novel pharmaceutical composition of sofosbuvir and ribavirin |
| US20170128482A1 (en) | 2014-06-23 | 2017-05-11 | Sanovel Ilac Sanayi Ve Ticaret A.S. | Modified release pharmaceutical compositions of sofosbuvir and ribavirin |
| CN105315319B (en) * | 2014-07-30 | 2020-11-20 | 南京圣和药业股份有限公司 | Hepatitis C virus inhibitor and application thereof |
| EP3174888A1 (en) | 2014-08-01 | 2017-06-07 | Lupin Limited | A process for the preparation of nucleoside phosphoramidate |
| WO2016033164A1 (en) | 2014-08-26 | 2016-03-03 | Enanta Pharmaceuticals, Inc. | Nucleoside and nucleotide derivatives |
| WO2016038542A2 (en) | 2014-09-10 | 2016-03-17 | Mylan Laboratories Limited | Polymorphic forms of sofosbuvir |
| WO2016064797A1 (en) * | 2014-10-20 | 2016-04-28 | Merck Sharp & Dohme Corp. | Process for making nucleoside phosphoramidate compounds |
| WO2016069975A1 (en) * | 2014-10-31 | 2016-05-06 | Cocrystal Pharma, Inc. | 2',2'-dihalo nucleoside analogs for treatment of the flaviviridae family of viruses and cancer |
| WO2016073756A1 (en) | 2014-11-06 | 2016-05-12 | Enanta Pharmaceuticals, Inc. | Deuterated nucleoside/tide derivatives |
| WO2016074596A1 (en) * | 2014-11-10 | 2016-05-19 | 南京曼杰生物科技有限公司 | Nucleoside phosphoramidate derivative and application thereof |
| WO2016078582A1 (en) * | 2014-11-20 | 2016-05-26 | 南京曼杰生物科技有限公司 | Novel nucleoside phosphoramidate derivatives and applications thereof |
| US9732110B2 (en) | 2014-12-05 | 2017-08-15 | Enanta Pharmaceuticals, Inc. | Nucleoside and nucleotide derivatives |
| CN106146588A (en) * | 2015-03-26 | 2016-11-23 | 常州制药厂有限公司 | A kind of preparation method of Suo Feibuwei |
| CN106188192B (en) * | 2015-04-29 | 2019-09-10 | 刘沛 | Nucleosides phosphoramidic acid/the phosphate derivatives and its medical usage of the amino-acid ester containing D- |
| CN104829672B (en) * | 2015-05-19 | 2018-03-13 | 江苏福瑞生物医药有限公司 | A kind of synthetic method of pharmaceutical intermediate |
| MX2018001073A (en) | 2015-08-06 | 2018-06-12 | Chimerix Inc | Pyrrolopyrimidine nucleosides and analogs thereof useful as antiviral agents. |
| EP3133062A1 (en) | 2015-08-19 | 2017-02-22 | Zaklady Farmaceutyczne Polpharma SA | Process for the preparation of a pharmaceutical agent |
| WO2017029408A1 (en) | 2015-08-20 | 2017-02-23 | Ratiopharm Gmbh | Solid state forms of sofosbuvir |
| CN105061535A (en) * | 2015-09-02 | 2015-11-18 | 江苏科本医药化学有限公司 | Synthetic method of sofosbuvir intermediate |
| CN106674319B (en) * | 2015-11-06 | 2020-04-21 | 博瑞生物医药(苏州)股份有限公司 | Compound for treating hepatitis C |
| CN106674320B (en) * | 2015-11-06 | 2020-04-21 | 博瑞生物医药(苏州)股份有限公司 | HCV (hepatitis C virus) treatment medicine |
| CN106674318B (en) * | 2015-11-06 | 2020-04-21 | 博瑞生物医药(苏州)股份有限公司 | NS5B polymerase inhibitor |
| WO2017093973A1 (en) * | 2015-12-02 | 2017-06-08 | Sun Pharmaceutical Industries Limited | Process for the preparation of pure sofosbuvir |
| ITUB20161079A1 (en) | 2016-02-25 | 2017-08-25 | Hc Pharma Ag | PROCEDURE FOR THE PREPARATION OF SOFOSBUVIR |
| US10738071B2 (en) | 2016-03-17 | 2020-08-11 | Mylan Laboratories Limited | Polymorphic forms of sofosbuvir |
| EP3452447A4 (en) | 2016-05-03 | 2019-12-18 | The Regents of The University of California | Inhibitors of ires-mediated protein synthesis |
| WO2017197046A1 (en) | 2016-05-10 | 2017-11-16 | C4 Therapeutics, Inc. | C3-carbon linked glutarimide degronimers for target protein degradation |
| EP3454862B1 (en) | 2016-05-10 | 2024-09-11 | C4 Therapeutics, Inc. | Spirocyclic degronimers for target protein degradation |
| WO2017197055A1 (en) | 2016-05-10 | 2017-11-16 | C4 Therapeutics, Inc. | Heterocyclic degronimers for target protein degradation |
| CN107501247A (en) * | 2016-06-14 | 2017-12-22 | 安徽贝克联合制药有限公司 | A kind of preparation method of uridine analog derivative |
| WO2017223024A1 (en) * | 2016-06-20 | 2017-12-28 | Merck Sharp & Dohme Corp. | Use of cyclic phosphate substituted nucleoside derivatives for the treatment of viral diseases |
| SI3512863T1 (en) | 2016-09-07 | 2022-04-29 | Atea Pharmaceuticals, Inc. | 2'-substituted-n6-substituted purine nucleotides for rna virus treatment |
| WO2018078536A1 (en) | 2016-10-26 | 2018-05-03 | Lupin Limited | Stable solid dispersion of sofosbuvir and process for preparation thereof |
| WO2018119013A1 (en) | 2016-12-22 | 2018-06-28 | Merck Sharp & Dohme Corp. | Antiviral aliphatic ester prodrugs of tenofovir |
| US11261152B2 (en) | 2017-01-20 | 2022-03-01 | The Regents Of The University Of California | Inhibitors of the N-terminal domain of the androgen receptor |
| SG11201906163TA (en) | 2017-02-01 | 2019-08-27 | Atea Pharmaceuticals Inc | Nucleotide hemi-sulfate salt for the treatment of hepatitis c virus |
| RU2650610C1 (en) | 2017-02-28 | 2018-04-16 | Васильевич Иващенко Александр | Antiviral composition and method of its application |
| RU2656228C9 (en) * | 2017-06-13 | 2019-04-16 | Олег Ростиславович Михайлов | WEAKLY CRYSTALLISED β-MODIFICATION OF (S)-ISOPROPYL 2-((S)-(((2R,3R,4R,5R)-5-(2,4-DIOXO-3,4-DIHYDROPYRIMIDIN-(2H)-YL)-4-FLUORO-3-HYDROXY-4-METHYLTETRAHYDROFURAN-2-YL)METHOXY)-(PHENOXY)PHOSPHORYLAMINO)PROPANOATE, METHOD FOR PRODUCTION THEREOF AND PHARMACEUTICAL COMPOSITION BASED THEREON |
| GB201709471D0 (en) | 2017-06-14 | 2017-07-26 | Nucana Biomed Ltd | Diastereoselective synthesis of hosphate derivatives |
| KR20240033119A (en) | 2017-06-30 | 2024-03-12 | 더 리전트 오브 더 유니버시티 오브 캘리포니아 | Compositions and methods of modulation of hair growth |
| US11111264B2 (en) | 2017-09-21 | 2021-09-07 | Chimerix, Inc. | Morphic forms of 4-amino-7-(3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-2-methyl-7H-pyrrolo[2,3-d]pyrimidine-5-carboxamide and uses thereof |
| JP2021509899A (en) * | 2018-01-05 | 2021-04-08 | セレコー,インコーポレーテッド | Nucleotide prodrug |
| US11708329B2 (en) | 2018-01-30 | 2023-07-25 | The Regents Of The University Of California | Inhibitors of the WNT/beta-catenin pathway |
| EP3773753A4 (en) | 2018-04-10 | 2021-12-22 | ATEA Pharmaceuticals, Inc. | Treatment of hcv infected patients with cirrhosis |
| NL2021049B1 (en) | 2018-06-04 | 2019-12-11 | Academisch Ziekenhuis Leiden | Use of 6'-fluoro-neplanocin a derivatives against chikungunya virus |
| WO2020006489A1 (en) | 2018-06-29 | 2020-01-02 | The Regents Of The University Of California | New molecular tweezers against neurological disorders and viral infections |
| AU2019310595B2 (en) | 2018-07-27 | 2022-11-24 | 1200 Pharma Llc | CDK inhibitors and uses thereof |
| IL280639B (en) | 2018-09-17 | 2022-07-01 | Yungjin Pharmaceutical Co Ltd | Novel thiazole derivatives and pharmaceutically acceptable salts thereof |
| CN114127072B (en) | 2019-03-25 | 2024-10-11 | 加利福尼亚技术学院 | PRMT5 inhibitors and uses thereof |
| CN114096544A (en) | 2019-05-20 | 2022-02-25 | 加州理工学院 | KRAS G12C inhibitors and uses thereof |
| WO2021142221A1 (en) | 2020-01-10 | 2021-07-15 | The Regents Of The University Of California | Compositions and methods for the treatment of neurodegenerative diseases |
| US10874687B1 (en) | 2020-02-27 | 2020-12-29 | Atea Pharmaceuticals, Inc. | Highly active compounds against COVID-19 |
| EP4125810A1 (en) | 2020-03-22 | 2023-02-08 | InspirMed Corp. | Composition of antiviral agent for use in prophylactic or post-exposure treatment of infectious or respiratory diseases |
| CN111467363A (en) * | 2020-04-07 | 2020-07-31 | 中国科学院深圳先进技术研究院 | Application of sofosbuvir in preparation of medicine for preventing and treating coronavirus |
| WO2021263072A1 (en) | 2020-06-25 | 2021-12-30 | Dana-Farber Cancer Institute, Inc. | Methods of treating disease |
| US20230271925A1 (en) | 2020-07-07 | 2023-08-31 | ILAb Co., Ltd. | Novel tnf activity inhibitor compound, and pharmaceutically acceptable salt thereof |
| CA3191166A1 (en) | 2020-08-10 | 2022-02-17 | Dana-Farber Cancer Institute, Inc. | Substituted 3-amino-4-methylbenzenesulfonamides as small molecule inhibitors of ubiquitin-specific protease 28 |
| AU2021325872A1 (en) | 2020-08-10 | 2023-03-16 | Dana-Farber Cancer Institute, Inc. | Substituted 1,2,4-oxadiazoles as small molecule inhibitors of ubiquitin-specific protease 28 |
| AU2021326457A1 (en) | 2020-08-10 | 2023-03-16 | Dana-Farber Cancer Institute, Inc. | Fused tricyclic pyrimidine-thieno-pyridine small molecule inhibitors of ubiquitin-specific protease 28 |
| WO2022076903A1 (en) * | 2020-10-09 | 2022-04-14 | Atea Pharmaceuticals, Inc. | Niran interfering drugs for sars-cov-2 mutant therapy |
| WO2022087433A1 (en) | 2020-10-23 | 2022-04-28 | Dana-Farber Cancer Institute, Inc. | Covalent inhibitors of creatine kinase (ck) and uses thereof for treating and preventing cancer |
| US20240058344A1 (en) | 2020-12-18 | 2024-02-22 | Cornell University | Methods of treating neurodegenerative disorders and stat3-linked cancers using suppressors of electron leak |
| KR20230129455A (en) | 2020-12-21 | 2023-09-08 | 코넬 유니버시티 | Peptide-Linked Drug Delivery Systems |
| WO2022150574A1 (en) | 2021-01-08 | 2022-07-14 | Cornell University | Inhibitors of mycobacterium tuberculosis lipoamide dehydrogenase |
| US20240190809A1 (en) | 2021-05-11 | 2024-06-13 | Awakn Ls Europe Holdings Limited | Therapeutic aminoindane compounds and compositions |
| AU2022279234A1 (en) | 2021-05-21 | 2023-12-07 | Gilead Sciences, Inc. | Pentacyclic derivatives as zika virus inhibitors |
| US20240197652A1 (en) | 2021-05-25 | 2024-06-20 | Awakn Ls Europe Holdings Limited | Ketamine in the treatment of behavioral addictions |
| WO2023028091A1 (en) | 2021-08-23 | 2023-03-02 | Alexander Shulgin Research Institute | Deuterated empathogens |
| US11541071B1 (en) | 2021-12-16 | 2023-01-03 | Ascletis BioScience Co., Ltd | Nucleoside derivatives and methods of use thereof |
| WO2023139163A1 (en) | 2022-01-19 | 2023-07-27 | Awakn Ls Europe Holdings Limited | 1,3-benzodioxole esters and their therapeutic use |
| WO2023156565A1 (en) | 2022-02-16 | 2023-08-24 | Awakn Ls Europe Holdings Limited | Bridged ring compounds and their therapeutic use as cns agents |
| WO2023250157A1 (en) | 2022-06-24 | 2023-12-28 | Cornell University | Inhibitors of mycobacterium tuberculosis lipoamide dehydrogenase |
| WO2024013209A1 (en) | 2022-07-13 | 2024-01-18 | Astrazeneca Ab | Pcsk9 inhibitors and methods of use thereof |
| WO2024134270A1 (en) | 2022-12-19 | 2024-06-27 | Prepaire Labs Limited | Method to identify and alleviate the symptoms of parkinsonism/parkinson's disease |
Citations (77)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2759300A (en) | 1954-08-11 | 1956-08-21 | Pest Control Ltd | Method and means for introducing a predetermined amount of a poisonous material beneath the surface of the soil |
| US5192549A (en) | 1988-09-28 | 1993-03-09 | Yissum Research Development Company Of The Hebrew University Of Jerusalem | Method of amphiphatic drug loading in liposomes by pH gradient |
| US5376380A (en) | 1990-08-21 | 1994-12-27 | Daiichi Pharmaceutical Co., Ltd. | Method of producing liposomal products from freeze or spray-dried preparations of liposomes |
| WO1997012033A1 (en) | 1995-09-27 | 1997-04-03 | Emory University | Recombinant hepatitis c virus rna replicase |
| WO2000006529A1 (en) | 1998-07-27 | 2000-02-10 | Istituto Di Ricerche Di Biologia Molecolare P Angeletti S.P.A. | Diketoacid-derivatives as inhibitors of polymerases |
| US6060080A (en) | 1990-07-16 | 2000-05-09 | Daiichi Pharmaceutical Co., Ltd. | Liposomal products |
| US6132763A (en) | 1988-10-20 | 2000-10-17 | Polymasc Pharmaceuticals Plc | Liposomes |
| US6180134B1 (en) | 1993-03-23 | 2001-01-30 | Sequus Pharmaceuticals, Inc. | Enhanced ciruclation effector composition and method |
| WO2002057287A2 (en) | 2001-01-22 | 2002-07-25 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2003006490A1 (en) | 2001-07-11 | 2003-01-23 | Vertex Pharmaceuticals Incorporated | Bridged bicyclic serine protease inhibitors |
| WO2003010141A2 (en) | 2001-07-25 | 2003-02-06 | Boehringer Ingelheim (Canada) Ltd. | Hepatitis c virus polymerase inhibitors with a heterobicyclic structure |
| WO2003037895A1 (en) | 2001-11-02 | 2003-05-08 | Glaxo Group Limited | 4-(6-membered)-heteroaryl acyl pyrrolidine derivatives as hcv inhibitors |
| WO2003064456A1 (en) | 2002-02-01 | 2003-08-07 | Boehringer Ingelheim International Gmbh | Tripeptides having a hydroxyproline ether of a substituted quinoline for the inhibition of ns3 (hepatitis c) |
| US20030187018A1 (en) | 2002-02-01 | 2003-10-02 | Boehringer Ingelheim International Gmbh | Hepatitis C inhibitor tri-peptides |
| WO2003101993A1 (en) | 2002-06-04 | 2003-12-11 | Neogenesis Pharmaceuticals, Inc. | Pyrazolo` 1,5a! pyrimidine compounds as antiviral agents |
| WO2003105770A2 (en) | 2002-06-17 | 2003-12-24 | Merck & Co., Inc. | Carbocyclic nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2004000858A2 (en) | 2002-06-21 | 2003-12-31 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2004003138A2 (en) | 2002-06-27 | 2004-01-08 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2004002977A1 (en) | 2002-07-01 | 2004-01-08 | Pharmacia & Upjohn Company Llc | Inhibitors of hcv ns5b polymerase |
| WO2004002944A1 (en) | 2002-07-01 | 2004-01-08 | Pharmacia & Upjohn Company | Inhibitors of hcv ns5b polymerase |
| WO2004009020A2 (en) | 2002-07-24 | 2004-01-29 | Merck & Co., Inc. | Pyrrolopyrimidine thionucleoside analogs as antivirals |
| WO2004014852A2 (en) | 2002-08-12 | 2004-02-19 | Bristol-Myers Squibb Company | Iminothiazolidinones as inhibitors of hcv replication |
| WO2004035571A1 (en) | 2002-10-15 | 2004-04-29 | Rigel Pharmaceuticals, Inc. | Substituted indoles and their use as hcv inhibitors |
| WO2004041201A2 (en) | 2002-11-01 | 2004-05-21 | Viropharma Incorporated | Benzofuran compounds, compositions and methods for treatment and prophylaxis of hepatitis c viral infections and associated diseases |
| WO2004065367A1 (en) | 2003-01-22 | 2004-08-05 | Boehringer Ingelheim International Gmbh | Viral polymerase inhibitors |
| US6784166B2 (en) | 2001-06-12 | 2004-08-31 | Syntex (U.S.A.) Llc | 4′-substituted nucleoside derivatives as inhibitors of HCV RNA replication. |
| WO2004094452A2 (en) | 2003-04-16 | 2004-11-04 | Bristol-Myers Squibb Company | Macrocyclic isoquinoline peptide inhibitors of hepatitis c virus |
| WO2004096210A1 (en) | 2003-05-01 | 2004-11-11 | Glaxo Group Limited | Acylated indoline and tetrahydroquinoline derivatives as hcv inhibitors |
| US20040229840A1 (en) | 2002-10-29 | 2004-11-18 | Balkrishen Bhat | Nucleoside derivatives as inhibitors of RNA-dependent RNA viral polymerase |
| US20050009737A1 (en) | 2003-05-30 | 2005-01-13 | Jeremy Clark | Modified fluorinated nucleoside analogues |
| WO2005021568A2 (en) | 2003-08-27 | 2005-03-10 | Biota, Inc. | Novel tricyclic nucleosides or nucleotides as therapeutic agents |
| WO2005028502A1 (en) | 2003-09-18 | 2005-03-31 | Vertex Pharmaceuticals, Incorporated | Inhibitors of serine proteases, particularly hcv ns3-ns4a protease |
| WO2005037214A2 (en) | 2003-10-14 | 2005-04-28 | Intermune, Inc. | Macrocyclic carboxylic acids and acylsulfonamides as inhibitors of hcv replication |
| US20050098125A1 (en) | 2002-07-01 | 2005-05-12 | Thomas Hathaway | Valve lash adjustment apparatus and method |
| US20050154056A1 (en) | 2003-11-07 | 2005-07-14 | Pharmacia & Upjohn Company | Inhibitors of HCV NS5B polymerase |
| WO2005067900A2 (en) | 2004-01-06 | 2005-07-28 | Achillion Pharmaceuticals, Inc. | Azabenzofuran substituted thioureas as inhibitors of viral replication |
| WO2005095403A2 (en) | 2004-03-30 | 2005-10-13 | Intermune, Inc. | Macrocyclic compounds as inhibitors of viral replication |
| WO2005103045A1 (en) | 2004-04-22 | 2005-11-03 | Glaxo Group Limited | Acyl dihydro pyrrole derivatives as hcv inhibitors |
| US20050267018A1 (en) | 2003-10-14 | 2005-12-01 | Blatt Lawrence M | Macrocyclic compounds as inhibitors of viral replication |
| WO2005123087A2 (en) | 2004-06-15 | 2005-12-29 | Merck & Co., Inc. | C-purine nucleoside analogs as inhibitors of rna-dependent rna viral polymerase |
| WO2006012078A2 (en) | 2004-06-24 | 2006-02-02 | Merck & Co., Inc. | Nucleoside aryl phosphoramidates for the treatment of rna-dependent rna viral infection |
| US20060040890A1 (en) | 2004-08-23 | 2006-02-23 | Roche Palo Alto Llc | Anti-viral nucleosides |
| US20060040927A1 (en) | 2004-08-23 | 2006-02-23 | Roche Palo Alto Llc | Heterocyclic antiviral compounds |
| WO2006020082A1 (en) | 2004-08-09 | 2006-02-23 | Bristol-Myers Squibb Company | Inhibitors of hcv replication |
| WO2006035061A1 (en) | 2004-09-30 | 2006-04-06 | Tibotec Pharmaceuticals Ltd. | Hcv inhibiting bi-cyclic pyrimidines |
| US20060122146A1 (en) | 2004-09-14 | 2006-06-08 | Byoung-Kwon Chun | Preparation of 2'-fluoro-2'-alkyl-substituted or other optionally substituted ribofuranosyl pyrimidines and purines and their derivatives |
| WO2006065590A2 (en) | 2004-12-16 | 2006-06-22 | Xtl Biopharmaceuticals Inc. | Pyridine and pyrimidine antiviral compositions |
| WO2006065335A2 (en) | 2004-10-21 | 2006-06-22 | Merck & Co., Inc. | Fluorinated pyrrolo[2,3-d]pyrimidine nucleosides for the treatment of rna-dependent rna viral infection |
| US20060142238A1 (en) | 2003-07-21 | 2006-06-29 | Mcguigan Christopher | Chemical compounds |
| US20060166964A1 (en) | 2004-08-09 | 2006-07-27 | Hudyma Thomas W | Inhibitors of HCV replication |
| US20060194749A1 (en) | 2005-02-28 | 2006-08-31 | Genelabs Technologies, Inc. | Tricyclic-nucleoside prodrugs for treating viral infections |
| US20060199783A1 (en) | 2004-07-21 | 2006-09-07 | Pharmassett, Inc. | Preparation of alkyl-substituted 2-deoxy-2-fluoro-D-ribofuranosyl pyrimidines and purines and their derivatives |
| WO2006093801A1 (en) | 2005-02-25 | 2006-09-08 | Abbott Laboratories | Thiadiazine derivatives useful as anti-infective agents |
| WO2006100310A1 (en) | 2005-03-25 | 2006-09-28 | Tibotec Pharmaceuticals Ltd | Heterobicylic inhibitors of hcv |
| US20060241064A1 (en) | 2005-04-25 | 2006-10-26 | Genelabs Technologies, Inc. | Nucleoside compounds for treating viral infections |
| WO2006120251A1 (en) | 2005-05-12 | 2006-11-16 | Tibotec Pharmaceuticals Ltd. | Pteridines useful as hcv inhibitors and methods for the preparation thereof |
| WO2006120252A2 (en) | 2005-05-12 | 2006-11-16 | Tibotec Pharmaceuticals Ltd. | Pyrido[2,3-d]pyrimidines useful as hcv inhibitors, and methods for the preparation thereof |
| US20060276511A1 (en) | 2005-06-06 | 2006-12-07 | Michael Serrano-Wu | Inhibitors of HCV replication |
| WO2007002602A2 (en) | 2005-06-27 | 2007-01-04 | Depuy Spine, Inc. | Intervertebral disc prosthesis and associated methods |
| WO2007014922A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007014921A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007014920A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007014926A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007014925A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocylic inhibitors of hepatitis c virus |
| WO2007015824A2 (en) | 2005-07-25 | 2007-02-08 | Intermune, Inc. | Novel macrocyclic inhibitors of hepatitis c virus replication |
| WO2007039145A1 (en) | 2005-09-23 | 2007-04-12 | Glaxo Group Limited | C (2) -heteroarylmethyl-c (4) -pyrazinyl-2-yl acyl pyrrolidine compounds and their use for treating viral infections, especially hepatitis c virus |
| WO2007039142A1 (en) | 2005-09-23 | 2007-04-12 | Glaxo Group Limited | C (2) -heteroarylmethyl-c (4) -methoxymethyl acyl pyrrolidine compounds and their use for treating viral infections, especially hepatitis c virus |
| WO2007070556A2 (en) | 2005-12-12 | 2007-06-21 | Genelabs Technologies, Inc. | N-(6-membered aromatic ring)-amido anti-viral compounds |
| WO2007076034A2 (en) | 2005-12-21 | 2007-07-05 | Abbott Laboratories | Anti-viral compounds |
| WO2007088148A1 (en) | 2006-02-01 | 2007-08-09 | Smithkline Beecham Corporation | Derivatives of thiophene carboxilic acid as antiviral agent |
| WO2007092000A1 (en) | 2006-02-06 | 2007-08-16 | Bristol-Myers Squibb Company | Inhibitors of hcv replication |
| WO2007095269A2 (en) | 2006-02-14 | 2007-08-23 | Merck & Co., Inc. | Nucleoside aryl phosphoramidates for the treatment of rna-dependent rna viral infection |
| WO2007093901A1 (en) | 2006-02-17 | 2007-08-23 | Pfizer Limited | 3 -deazapurine derivatives as tlr7 modulators |
| US20070197463A1 (en) | 2005-12-09 | 2007-08-23 | Pharmasset, Inc. | Antiviral nucleosides |
| US20070275947A1 (en) | 2006-05-25 | 2007-11-29 | Bristol-Myers Squibb Company | Cyclopropyl Fused Indolobenzazepine HCV NS5B Inhibitors |
| EP1881001A1 (en) | 2006-07-20 | 2008-01-23 | Tibotec Pharmaceuticals Ltd. | HCV NS-3 serine protease inhibitors |
| WO2008010921A2 (en) | 2006-07-07 | 2008-01-24 | Gilead Sciences, Inc. | Modulators of pharmacokinetic properties of therapeutics |
Family Cites Families (228)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2007275A (en) | 1933-01-09 | 1935-07-09 | Elizabeth Q Kendall | Ingot stripper |
| USRE29835E (en) | 1971-06-01 | 1978-11-14 | Icn Pharmaceuticals | 1,2,4-Triazole nucleosides |
| US3798209A (en) | 1971-06-01 | 1974-03-19 | Icn Pharmaceuticals | 1,2,4-triazole nucleosides |
| US3852267A (en) | 1972-08-04 | 1974-12-03 | Icn Pharmaceuticals | Phosphoramidates of 3{40 ,5{40 -cyclic purine nucleotides |
| JPS5116273A (en) | 1974-07-31 | 1976-02-09 | Hitachi Ltd | DATSUSHOSOCHINIOKERU NH3 CHUNYURYONO SEIGYOHO |
| JPS5238939U (en) | 1975-09-12 | 1977-03-18 | ||
| US4526988A (en) * | 1983-03-10 | 1985-07-02 | Eli Lilly And Company | Difluoro antivirals and intermediate therefor |
| NL8403224A (en) | 1984-10-24 | 1986-05-16 | Oce Andeno Bv | DIOXAPHOSPHORINANS, THEIR PREPARATION AND THE USE FOR SPLITTING OF OPTICALLY ACTIVE COMPOUNDS. |
| US5223263A (en) | 1988-07-07 | 1993-06-29 | Vical, Inc. | Liponucleotide-containing liposomes |
| GB8719367D0 (en) | 1987-08-15 | 1987-09-23 | Wellcome Found | Therapeutic compounds |
| JPH03501253A (en) | 1987-09-22 | 1991-03-22 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | Liposome-based nucleoside analogs for the treatment of AIDS |
| US5705363A (en) | 1989-03-02 | 1998-01-06 | The Women's Research Institute | Recombinant production of human interferon τ polypeptides and nucleic acids |
| US5411947A (en) * | 1989-06-28 | 1995-05-02 | Vestar, Inc. | Method of converting a drug to an orally available form by covalently bonding a lipid to the drug |
| US5194654A (en) * | 1989-11-22 | 1993-03-16 | Vical, Inc. | Lipid derivatives of phosphonoacids for liposomal incorporation and method of use |
| US5463092A (en) | 1989-11-22 | 1995-10-31 | Vestar, Inc. | Lipid derivatives of phosphonacids for liposomal incorporation and method of use |
| US5026687A (en) * | 1990-01-03 | 1991-06-25 | The United States Of America As Represented By The Department Of Health And Human Services | Treatment of human retroviral infections with 2',3'-dideoxyinosine alone and in combination with other antiviral compounds |
| CA2079912C (en) | 1990-04-06 | 2000-07-11 | Gregory Reyes | Hepatitis c virus epitopes |
| WO1991016920A1 (en) | 1990-05-07 | 1991-11-14 | Vical, Inc. | Lipid prodrugs of salicylate and nonsteroidal anti-inflammatory drugs |
| WO1991018914A1 (en) | 1990-05-29 | 1991-12-12 | Vical, Inc. | Synthesis of glycerol di- and triphosphate derivatives |
| DE69115694T2 (en) | 1990-06-13 | 1996-10-17 | Arnold Newton Mass. Glazier | PHOSPHORYLATED PRODRUGS |
| US5372808A (en) | 1990-10-17 | 1994-12-13 | Amgen Inc. | Methods and compositions for the treatment of diseases with consensus interferon while reducing side effect |
| US5543390A (en) * | 1990-11-01 | 1996-08-06 | State Of Oregon, Acting By And Through The Oregon State Board Of Higher Education, Acting For And On Behalf Of The Oregon Health Sciences University | Covalent microparticle-drug conjugates for biological targeting |
| US5543389A (en) | 1990-11-01 | 1996-08-06 | State Of Oregon, Acting By And Through The Oregon State Board Of Higher Education On Behalf Of The Oregon Health Sciences University, A Non Profit Organization | Covalent polar lipid-peptide conjugates for use in salves |
| US5256641A (en) | 1990-11-01 | 1993-10-26 | State Of Oregon | Covalent polar lipid-peptide conjugates for immunological targeting |
| US5149794A (en) * | 1990-11-01 | 1992-09-22 | State Of Oregon | Covalent lipid-drug conjugates for drug targeting |
| US5595732A (en) | 1991-03-25 | 1997-01-21 | Hoffmann-La Roche Inc. | Polyethylene-protein conjugates |
| US5157027A (en) | 1991-05-13 | 1992-10-20 | E. R. Squibb & Sons, Inc. | Bisphosphonate squalene synthetase inhibitors and method |
| CA2112803A1 (en) | 1991-07-12 | 1993-01-21 | Karl Y. Hostetler | Antiviral liponucleosides: treatment of hepatitis b |
| US5554728A (en) | 1991-07-23 | 1996-09-10 | Nexstar Pharmaceuticals, Inc. | Lipid conjugates of therapeutic peptides and protease inhibitors |
| TW224053B (en) | 1991-09-13 | 1994-05-21 | Paul B Chretien | |
| US5676942A (en) | 1992-02-10 | 1997-10-14 | Interferon Sciences, Inc. | Composition containing human alpha interferon species proteins and method for use thereof |
| US5405598A (en) * | 1992-02-24 | 1995-04-11 | Schinazi; Raymond F. | Sensitizing agents for use in boron neutron capture therapy |
| JP3102945B2 (en) | 1992-02-27 | 2000-10-23 | 財団法人野田産業科学研究所 | Hepatitis treatment |
| US5610054A (en) | 1992-05-14 | 1997-03-11 | Ribozyme Pharmaceuticals, Inc. | Enzymatic RNA molecule targeted against Hepatitis C virus |
| US5426183A (en) | 1992-06-22 | 1995-06-20 | Eli Lilly And Company | Catalytic stereoselective glycosylation process for preparing 2'-deoxy-2',2'-difluoronucleosides and 2'-deoxy-2'-fluoronucleosides |
| BR9302427A (en) | 1992-06-22 | 1994-01-11 | Lilly Co Eli | SELECTIVE STEREO PROCESS FOR THE PREPARATION OF A RIBOTURANOSIL DERIVATIVE |
| US5256798A (en) | 1992-06-22 | 1993-10-26 | Eli Lilly And Company | Process for preparing alpha-anomer enriched 2-deoxy-2,2-difluoro-D-ribofuranosyl sulfonates |
| US5301508A (en) | 1992-08-14 | 1994-04-12 | Rubbermaid Incorporated | Thermoelectric portable container |
| CA2105112C (en) * | 1992-09-01 | 2005-08-02 | Thomas C. Britton | A process for anomerizing nucleosides |
| GB9226729D0 (en) | 1992-12-22 | 1993-02-17 | Wellcome Found | Therapeutic combination |
| CN1078478C (en) * | 1993-02-24 | 2002-01-30 | 王瑞駪 | Compositions and methods of application of reactive antiviral polymers |
| EP0746319A4 (en) | 1993-05-12 | 1997-11-05 | Karl Y Hostetler | Acyclovir derivatives for topical use |
| EP0773029A4 (en) | 1993-07-19 | 1997-09-03 | Tokyo Tanabe Co | Hepatitis c virus proliferation inhibitor |
| US7375198B2 (en) * | 1993-10-26 | 2008-05-20 | Affymetrix, Inc. | Modified nucleic acid probes |
| US6156501A (en) | 1993-10-26 | 2000-12-05 | Affymetrix, Inc. | Arrays of modified nucleic acid probes and methods of use |
| US5951974A (en) | 1993-11-10 | 1999-09-14 | Enzon, Inc. | Interferon polymer conjugates |
| ATE214940T1 (en) | 1993-11-10 | 2002-04-15 | Enzon Inc | IMPROVED INTERFERON-POLYMER CONJUGATES |
| WO1995024185A1 (en) | 1994-03-11 | 1995-09-14 | Isis Pharmaceuticals, Inc. | Novel pyrimidine nucleosides |
| DE4447588C2 (en) | 1994-05-03 | 1997-11-20 | Omer Osama Dr Dr Med | Treatment of herpes and hepatitis virus infections and bronchitis |
| AU710074B2 (en) * | 1994-06-22 | 1999-09-16 | Proligo Llc | Novel method of preparation of known and novel 2'-modified nucleosides by intramolecular nucleophilic displacement |
| DE4432623A1 (en) | 1994-09-14 | 1996-03-21 | Huels Chemische Werke Ag | Process for bleaching aqueous surfactant solutions |
| US5738846A (en) | 1994-11-10 | 1998-04-14 | Enzon, Inc. | Interferon polymer conjugates and process for preparing the same |
| US5696277A (en) | 1994-11-15 | 1997-12-09 | Karl Y. Hostetler | Antiviral prodrugs |
| US6391859B1 (en) * | 1995-01-27 | 2002-05-21 | Emory University | [5-Carboxamido or 5-fluoro]-[2′,3′-unsaturated or 3′-modified]-pyrimidine nucleosides |
| US5703058A (en) * | 1995-01-27 | 1997-12-30 | Emory University | Compositions containing 5-fluoro-2',3'-didehydro-2',3'-dideoxycytidine or a mono-, di-, or triphosphate thereof and a second antiviral agent |
| PT806956E (en) | 1995-02-01 | 2003-01-31 | Resprotect Gmbh | USES OF 5-SUBSTITUTED NUCLEOSIDES FOR INHIBITION OF RESISTANCE FORMATION IN CYTOSTATIC TREATMENT |
| GB9505025D0 (en) * | 1995-03-13 | 1995-05-03 | Medical Res Council | Chemical compounds |
| DE19514523A1 (en) | 1995-04-12 | 1996-10-17 | Schering Ag | New cytosine and cytidine derivatives |
| US5908621A (en) | 1995-11-02 | 1999-06-01 | Schering Corporation | Polyethylene glycol modified interferon therapy |
| US5767097A (en) * | 1996-01-23 | 1998-06-16 | Icn Pharmaceuticals, Inc. | Specific modulation of Th1/Th2 cytokine expression by ribavirin in activated T-lymphocytes |
| GB9602028D0 (en) * | 1996-02-01 | 1996-04-03 | Amersham Int Plc | Nucleoside analogues |
| US5980884A (en) | 1996-02-05 | 1999-11-09 | Amgen, Inc. | Methods for retreatment of patients afflicted with Hepatitis C using consensus interferon |
| AU2064297A (en) | 1996-02-29 | 1997-09-16 | Immusol, Inc | Hepatitis c virus ribozymes |
| US5830905A (en) | 1996-03-29 | 1998-11-03 | Viropharma Incorporated | Compounds, compositions and methods for treatment of hepatitis C |
| US5633388A (en) | 1996-03-29 | 1997-05-27 | Viropharma Incorporated | Compounds, compositions and methods for treatment of hepatitis C |
| US5990276A (en) | 1996-05-10 | 1999-11-23 | Schering Corporation | Synthetic inhibitors of hepatitis C virus NS3 protease |
| GB9609932D0 (en) | 1996-05-13 | 1996-07-17 | Hoffmann La Roche | Use of IL-12 and IFN alpha for the treatment of infectious diseases |
| US5891874A (en) | 1996-06-05 | 1999-04-06 | Eli Lilly And Company | Anti-viral compound |
| US5837257A (en) | 1996-07-09 | 1998-11-17 | Sage R&D | Use of plant extracts for treatment of HIV, HCV and HBV infections |
| US5922757A (en) | 1996-09-30 | 1999-07-13 | The Regents Of The University Of California | Treatment and prevention of hepatic disorders |
| EP0961775B1 (en) * | 1996-10-16 | 2004-07-14 | ICN Pharmaceuticals, Inc. | Purine l-nucleosides, analogs and uses thereof |
| US6509320B1 (en) | 1996-10-16 | 2003-01-21 | Icn Pharmaceuticals, Inc. | Purine L-nucleosides, analogs and uses thereof |
| AU738170B2 (en) * | 1996-10-16 | 2001-09-13 | Icn Pharmaceuticals, Inc. | Monocyclic L-nucleosides, analogs and uses thereof |
| US6455690B1 (en) * | 1996-10-16 | 2002-09-24 | Robert Tam | L-8-oxo-7-propyl-7,8-dihydro-(9H)-guanosine |
| ES2169880T3 (en) | 1996-10-18 | 2002-07-16 | Vertex Pharma | INHIBITORS OF SERINE PROTEASES, PARTICULARLY OF THE NS3 PROTEASE OF HEPATITIS VIRUS. |
| GB9623908D0 (en) | 1996-11-18 | 1997-01-08 | Hoffmann La Roche | Amino acid derivatives |
| IL119833A (en) | 1996-12-15 | 2001-01-11 | Lavie David | Hypericum perforatum extracts for the preparation of pharmaceutical compositions for the treatment of hepatitis |
| US6004933A (en) | 1997-04-25 | 1999-12-21 | Cortech Inc. | Cysteine protease inhibitors |
| JP3963488B2 (en) | 1997-06-30 | 2007-08-22 | メルツ ファーマ ゲゼルシャフト ミット ベシュレンクテル ハフツンク ウント コンパニー コマンディゲゼルシャフト アウフ アクチェン | 1-amino-alkylcyclohexane NMDA receptor antagonist |
| IL134233A0 (en) | 1997-08-11 | 2001-04-30 | Boehringer Ingelheim Ca Ltd | Hepatitis c inhibitor peptide analogues |
| ATE206618T1 (en) | 1997-09-21 | 2001-10-15 | Schering Corp | COMBINATION THERAPY FOR REMOVAL OF DETECTED HCV RNA IN PATIENTS WITH CHRONIC HEPATITIS C INFECTION |
| US6703374B1 (en) * | 1997-10-30 | 2004-03-09 | The United States Of America As Represented By The Department Of Health And Human Services | Nucleosides for imaging and treatment applications |
| US5981709A (en) | 1997-12-19 | 1999-11-09 | Enzon, Inc. | α-interferon-polymer-conjugates having enhanced biological activity and methods of preparing the same |
| US7462605B2 (en) | 1998-01-23 | 2008-12-09 | Celmed Oncology (Usa), Inc. | Phosphoramidate compounds and methods of use |
| EP1167972B1 (en) | 1998-01-23 | 2017-03-08 | Kiadis Pharma Intellectual Property B.V. | Enzyme catalyzed therapeutic agents |
| EP2390257A1 (en) * | 1998-02-25 | 2011-11-30 | Emory University | 2'-fluoronucleosides |
| US6787305B1 (en) | 1998-03-13 | 2004-09-07 | Invitrogen Corporation | Compositions and methods for enhanced synthesis of nucleic acid molecules |
| CA2326535A1 (en) | 1998-03-27 | 1999-10-07 | Regents Of The University Of Minnesota | Nucleosides with antiviral and anticancer activity |
| GB9806815D0 (en) | 1998-03-30 | 1998-05-27 | Hoffmann La Roche | Amino acid derivatives |
| DK1066247T3 (en) * | 1998-03-31 | 2007-04-02 | Vertex Pharma | Inhibitors of serine proteases, especially hepatitis C virus NS3 protease |
| TWI277424B (en) | 1998-05-15 | 2007-04-01 | Schering Corp | Combination therapy for eradicating detectable NCV-RNA in antiviral treatment naive patients having chronic hepatitis C infection |
| KR20010052622A (en) | 1998-06-08 | 2001-06-25 | 프리돌린 클라우스너, 롤란드 비. 보레르 | Use of peg-ifn-alpha and ribavirin for the treatment of chronic hepatitis c |
| US6323180B1 (en) | 1998-08-10 | 2001-11-27 | Boehringer Ingelheim (Canada) Ltd | Hepatitis C inhibitor tri-peptides |
| KR100634342B1 (en) | 1998-08-10 | 2006-10-16 | 이데닉스(케이만)리미티드 | ?-L-2'-Deoxy-nucleosides for the treatment of hepatitis B |
| CA2354536A1 (en) | 1998-12-18 | 2000-06-29 | Schering Corporation | Ribavirin-pegylated interferon alfa induction hcv combination therapy |
| AU775601B2 (en) | 1999-07-22 | 2004-08-05 | Celmed Oncology (Usa) Inc. | Enzyme catalyzed therapeutic activation |
| KR20020091829A (en) | 1999-07-30 | 2002-12-06 | 애보트 게엠베하 운트 콤파니 카게 | 2-Pyrazolin-5-ones |
| AU1262001A (en) | 1999-11-04 | 2001-05-14 | Biochem Pharma Inc. | Method for the treatment or prevention of flaviviridae viral infection using nucleoside analogues |
| TR200601784T2 (en) * | 1999-11-12 | 2007-01-22 | Pharmasset Ltd. | Synthesis of 2'-deoxy-L-nucleosides |
| US6495677B1 (en) | 2000-02-15 | 2002-12-17 | Kanda S. Ramasamy | Nucleoside compounds |
| PL364995A1 (en) | 2000-02-18 | 2004-12-27 | Shire Biochem Inc. | Method for the treatment or prevention of flavivirus |
| KR100974917B1 (en) | 2000-04-13 | 2010-08-09 | 파마셋 인코포레이티드 | 3'- or 2'-hydroxymethyl substituted nucleoside derivatives for treatment of hepatitis virus infections |
| US6924270B2 (en) | 2000-04-20 | 2005-08-02 | Schering Corporation | Ribavirin-interferon alfa combination therapy for eradicating detectable HCV-RNA in patients having chronic hepatitis C infection |
| MY164523A (en) * | 2000-05-23 | 2017-12-29 | Univ Degli Studi Cagliari | Methods and compositions for treating hepatitis c virus |
| EA005890B1 (en) | 2000-05-26 | 2005-06-30 | Айденикс (Кайман) Лимитед | Methods for treating hepatitis delta virus infection with beta-l-2'-deoxy-nucleosides |
| US6787526B1 (en) | 2000-05-26 | 2004-09-07 | Idenix Pharmaceuticals, Inc. | Methods of treating hepatitis delta virus infection with β-L-2′-deoxy-nucleosides |
| CA2410579C (en) * | 2000-05-26 | 2010-04-20 | Jean-Pierre Sommadossi | Methods and compositions for treating flaviviruses and pestiviruses |
| FR2810322B1 (en) * | 2000-06-14 | 2006-11-10 | Pasteur Institut | COMBINATORY PRODUCTION OF NUCLEOTIDE AND NUCLEOTIDE ANALOGUES (XiTP) |
| MY141594A (en) | 2000-06-15 | 2010-05-14 | Novirio Pharmaceuticals Ltd | 3'-PRODRUGS OF 2'-DEOXY-ß-L-NUCLEOSIDES |
| US6815542B2 (en) | 2000-06-16 | 2004-11-09 | Ribapharm, Inc. | Nucleoside compounds and uses thereof |
| UA72612C2 (en) * | 2000-07-06 | 2005-03-15 | Pyrido[2.3-d]pyrimidine and pyrimido[4.5-d]pyrimidine nucleoside analogues, prodrugs and method for inhibiting growth of neoplastic cells | |
| AR029851A1 (en) | 2000-07-21 | 2003-07-16 | Dendreon Corp | NEW PEPTIDES AS INHIBITORS OF NS3-SERINA PROTEASA DEL VIRUS DE HEPATITIS C |
| WO2002008251A2 (en) | 2000-07-21 | 2002-01-31 | Corvas International, Inc. | Peptides as ns3-serine protease inhibitors of hepatitis c virus |
| KR100749160B1 (en) | 2000-07-21 | 2007-08-14 | 길리애드 사이언시즈, 인코포레이티드 | Methods for making prodrugs of phosphonate nucleotide analogues |
| AR034127A1 (en) | 2000-07-21 | 2004-02-04 | Schering Corp | IMIDAZOLIDINONES AS INHIBITORS OF NS3-SERINA PROTEASA OF THE HEPATITIS C VIRUS, PHARMACEUTICAL COMPOSITION, A METHOD FOR THEIR PREPARATION, AND THE USE OF THE SAME FOR THE MANUFACTURE OF A MEDICINAL PRODUCT |
| CZ2003195A3 (en) | 2000-07-21 | 2003-04-16 | Schering Corporation | Peptide inhibitors of serine protease NS3 and pharmaceutical preparation containing thereof |
| US7018985B1 (en) | 2000-08-21 | 2006-03-28 | Inspire Pharmaceuticals, Inc. | Composition and method for inhibiting platelet aggregation |
| US6897201B2 (en) * | 2000-08-21 | 2005-05-24 | Inspire Pharmaceuticals, Inc. | Compositions and methods for the treatment of glaucoma or ocular hypertension |
| AR039558A1 (en) * | 2000-08-21 | 2005-02-23 | Inspire Pharmaceuticals Inc | COMPOSITIONS AND METHOD FOR THE TREATMENT OF GLAUCOMA OR OCULAR HYPERTENSION |
| US20030008841A1 (en) | 2000-08-30 | 2003-01-09 | Rene Devos | Anti-HCV nucleoside derivatives |
| AU2002213343A1 (en) | 2000-10-18 | 2002-04-29 | Schering Corporation | Ribavirin-pegylated interferon alfa HCV combination therapy |
| PT1411954E (en) * | 2000-10-18 | 2011-03-16 | Pharmasset Inc | Modified nucleosides for treatment of viral infections and abnormal cellular proliferation |
| US6555677B2 (en) | 2000-10-31 | 2003-04-29 | Merck & Co., Inc. | Phase transfer catalyzed glycosidation of an indolocarbazole |
| AU2002248147B2 (en) | 2000-11-20 | 2006-04-06 | Bristol-Myers Squibb Company | Hepatitis C tripeptide inhibitors |
| ES2324594T3 (en) | 2000-12-12 | 2009-08-11 | Schering Corporation | DIARIL PEPTIDICS USED AS INHIBITORS OF THE SERINE PROTEASE NS3 OF THE HEPATITIS VIRUS C. |
| WO2002048116A2 (en) | 2000-12-13 | 2002-06-20 | Bristol-Myers Squibb Pharma Company | Inhibitors of hepatitis c virus ns3 protease |
| WO2002048157A2 (en) | 2000-12-13 | 2002-06-20 | Bristol-Myers Squibb Pharma Company | Imidazolidinones and their related derivatives as hepatitis c virus ns3 protease inhibitors |
| AU2002232660A1 (en) | 2000-12-15 | 2002-06-24 | Pharmasset Ltd. | Antiviral agents for treatment of flaviviridae infections |
| US7105499B2 (en) * | 2001-01-22 | 2006-09-12 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of RNA-dependent RNA viral polymerase |
| GB0112617D0 (en) | 2001-05-23 | 2001-07-18 | Hoffmann La Roche | Antiviral nucleoside derivatives |
| US20050009775A1 (en) | 2001-06-21 | 2005-01-13 | Howes Peter David | Nucleoside compounds in hcv |
| US6962991B2 (en) * | 2001-09-12 | 2005-11-08 | Epoch Biosciences, Inc. | Process for the synthesis of pyrazolopyrimidines |
| US7227019B2 (en) | 2001-09-13 | 2007-06-05 | Bristol-Myers Squibb Company | Process for the preparation of rebeccamycin and analogs thereof |
| WO2003024461A1 (en) | 2001-09-20 | 2003-03-27 | Schering Corporation | Hcv combination therapy |
| US7138376B2 (en) * | 2001-09-28 | 2006-11-21 | Idenix Pharmaceuticals, Inc. | Methods and compositions for treating hepatitis C virus using 4'-modified nucleosides |
| GB0129945D0 (en) | 2001-12-13 | 2002-02-06 | Mrc Technology Ltd | Chemical compounds |
| EP1569652A4 (en) * | 2001-12-14 | 2008-07-02 | Pharmasset Inc | N sp 4 /sp-acylcytosine nucleosides for treatment of viral iinfections |
| AU2002353165A1 (en) | 2001-12-17 | 2003-06-30 | Ribapharm Inc. | Deazapurine nucleoside libraries and compounds |
| WO2003055896A2 (en) * | 2001-12-21 | 2003-07-10 | Micrologix Biotech Inc. | Anti-viral 7-deaza l-nucleosides |
| WO2003062256A1 (en) | 2002-01-17 | 2003-07-31 | Ribapharm Inc. | 2'-beta-modified-6-substituted adenosine analogs and their use as antiviral agents |
| JP2005527499A (en) * | 2002-02-13 | 2005-09-15 | メルク エンド カムパニー インコーポレーテッド | Methods for inhibiting orthopoxvirus replication using nucleoside compounds |
| AU2003213628A1 (en) | 2002-02-28 | 2003-09-16 | Biota, Inc. | Nucleoside 5'-monophosphate mimics and their prodrugs |
| US7285658B2 (en) * | 2002-02-28 | 2007-10-23 | Biota, Inc. | Nucleotide mimics and their prodrugs |
| US20040014108A1 (en) * | 2002-05-24 | 2004-01-22 | Eldrup Anne B. | Oligonucleotides having modified nucleoside units |
| WO2003106477A1 (en) | 2002-06-01 | 2003-12-24 | Isis Pharmaceuticals, Inc. | Oligomeric compounds that include carbocyclic nucleosides and their use in gene modulation |
| AU2003242672B2 (en) | 2002-06-07 | 2009-12-17 | Universitair Medisch Centrum Utrecht | New compounds for modulating the activity of exhange proteins directly activated by camp (EPACS) |
| PL374792A1 (en) | 2002-06-28 | 2005-10-31 | Idenix (Cayman) Limited | 2' and 3'-nucleoside prodrugs for treating flaviviridae infections |
| US7608600B2 (en) * | 2002-06-28 | 2009-10-27 | Idenix Pharmaceuticals, Inc. | Modified 2′ and 3′-nucleoside prodrugs for treating Flaviviridae infections |
| CN1761677A (en) * | 2002-06-28 | 2006-04-19 | 埃迪尼克斯(开曼)有限公司 | Modified 2' and 3' -nucleoside produgs for treating flaviridae infections |
| CN100348607C (en) | 2002-06-28 | 2007-11-14 | 埃迪尼克斯(开曼)有限公司 | For the treatment of Flaviviridae viral infection in 2 'and 3'-nucleoside prodrugs |
| JP2005533824A (en) | 2002-06-28 | 2005-11-10 | イデニクス(ケイマン)リミテツド | 2'-C-methyl-3'-OL-valine ester ribofuranosyl cytidine for the treatment of Flaviviridae infections |
| CA2490666A1 (en) | 2002-07-16 | 2004-01-22 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| EP1572709A3 (en) | 2002-07-24 | 2005-11-23 | PTC Therapeutics, Inc. | Use of nucleoside compounds for nonsense suppression and the treatment of genetic diseases |
| WO2004011478A2 (en) | 2002-07-25 | 2004-02-05 | Micrologix Biotech Inc. | Anti-viral 7-deaza d-nucleosides and uses thereof |
| JP4173861B2 (en) * | 2002-09-13 | 2008-10-29 | イデニクス(ケイマン)リミテツド | Β-L-2'-deoxynucleoside and combination therapy for the treatment of resistant HBV strains |
| MXPA05005192A (en) | 2002-11-15 | 2005-09-08 | Idenix Cayman Ltd | 2aCOE-BRANCHED NUCLEOSIDES AND FLAVIVIRIDAE. |
| TWI332507B (en) * | 2002-11-19 | 2010-11-01 | Hoffmann La Roche | Antiviral nucleoside derivatives |
| PL377287A1 (en) * | 2002-12-12 | 2006-01-23 | Idenix (Cayman) Limited | Process for the production of 2'-branched nucleosides |
| AU2003225705A1 (en) | 2003-03-07 | 2004-09-30 | Ribapharm Inc. | Cytidine analogs and methods of use |
| WO2004084453A2 (en) | 2003-03-20 | 2004-09-30 | Microbiologica Quimica E Farmaceutica Ltd. | METHODS OF MANUFACTURE OF 2'-DEOXY-β-L-NUCLEOSIDES |
| WO2005002626A2 (en) | 2003-04-25 | 2005-01-13 | Gilead Sciences, Inc. | Therapeutic phosphonate compounds |
| US7407965B2 (en) | 2003-04-25 | 2008-08-05 | Gilead Sciences, Inc. | Phosphonate analogs for treating metabolic diseases |
| WO2004096286A2 (en) | 2003-04-25 | 2004-11-11 | Gilead Sciences, Inc. | Antiviral phosphonate analogs |
| US20050261237A1 (en) | 2003-04-25 | 2005-11-24 | Boojamra Constantine G | Nucleoside phosphonate analogs |
| MXPA05011296A (en) | 2003-04-25 | 2006-01-24 | Gilead Sciences Inc | Kinase inhibitor phosphonate conjugates. |
| US7452901B2 (en) * | 2003-04-25 | 2008-11-18 | Gilead Sciences, Inc. | Anti-cancer phosphonate analogs |
| US7470724B2 (en) | 2003-04-25 | 2008-12-30 | Gilead Sciences, Inc. | Phosphonate compounds having immuno-modulatory activity |
| AU2004233989A1 (en) | 2003-04-25 | 2004-11-11 | Gilead Sciences, Inc. | Anti-cancer phosphonate analogs |
| US20040259934A1 (en) | 2003-05-01 | 2004-12-23 | Olsen David B. | Inhibiting Coronaviridae viral replication and treating Coronaviridae viral infection with nucleoside compounds |
| EP1656093A2 (en) | 2003-05-14 | 2006-05-17 | Idenix (Cayman) Limited | Nucleosides for treatment of infection by corona viruses, toga viruses and picorna viruses |
| US20040229839A1 (en) | 2003-05-14 | 2004-11-18 | Biocryst Pharmaceuticals, Inc. | Substituted nucleosides, preparation thereof and use as inhibitors of RNA viral polymerases |
| WO2004106356A1 (en) | 2003-05-27 | 2004-12-09 | Syddansk Universitet | Functionalized nucleotide derivatives |
| EP1644479A4 (en) | 2003-06-16 | 2008-04-23 | Mark W Grinstaff | Functional synthetic molecules and macromolecules for gene delivery |
| RU2243972C1 (en) | 2003-06-26 | 2005-01-10 | Государственный научный центр вирусологии и биотехнологии "Вектор" | Nucleoside analog phosphoramidates as inhibitors of human immunodeficiency virus reproduction |
| EP1639121A4 (en) | 2003-06-30 | 2008-04-16 | Idenix Cayman Ltd | Synthesis of beta -l-2-deoxy nucleosides |
| WO2005009418A2 (en) | 2003-07-25 | 2005-02-03 | Idenix (Cayman) Limited | Purine nucleoside analogues for treating diseases caused by flaviviridae including hepatitis c |
| US20050148534A1 (en) * | 2003-09-22 | 2005-07-07 | Castellino Angelo J. | Small molecule compositions and methods for increasing drug efficiency using compositions thereof |
| US7348389B2 (en) | 2003-09-22 | 2008-03-25 | E. I. Du Pont De Nemours And Company | Method for achieving recoat adhesion over a fluorinated topcoat |
| JP2007509939A (en) * | 2003-10-27 | 2007-04-19 | ジェネラブズ テクノロジーズ インコーポレーティッド | Nucleoside compounds for treating viral infections |
| JP2007519734A (en) | 2004-01-28 | 2007-07-19 | メルク エンド カムパニー インコーポレーテッド | Aminocyclopentylpyridopyrazinone chemokine receptor activity modulator |
| WO2005082144A1 (en) | 2004-02-25 | 2005-09-09 | Government Of The United States Of America, As Represented By The Secretary, Department Of Health And Human Services Office Of Technology Transfer | Methylation inhibitor compounds |
| WO2005087788A2 (en) | 2004-03-04 | 2005-09-22 | The Regents Of The University Of California | Methods for preparation of nucleoside phosphonate esters |
| BRPI0512360A (en) * | 2004-06-23 | 2008-03-11 | Idenix Cayman Ltd | 5-aza-7-deazapurine derivatives for the treatment of flaviviridae |
| US7217523B2 (en) * | 2004-07-02 | 2007-05-15 | Regents Of The University Of Minnesota | Nucleoside phosphoramidates and nucleoside phosphoramidases |
| NZ552927A (en) | 2004-07-21 | 2010-05-28 | Pharmasset Inc | Preparation of alkyl-substituted 2-deoxy-2-fluoro-D-ribofuranosyl pyrimidines and purines and their derivatives |
| WO2006110157A2 (en) * | 2004-07-27 | 2006-10-19 | Gilead Sciences, Inc. | Nucleoside phosphonate conjugates as anti hiv agents |
| WO2006029081A2 (en) | 2004-09-02 | 2006-03-16 | Neopharm, Inc. | Nucleoside-lipid conjugates, their method of preparation and uses thereof |
| JP2008514639A (en) | 2004-09-24 | 2008-05-08 | イデニクス(ケイマン)リミテツド | Methods and compositions for treating flavivirus, pestivirus and hepacivirus infections |
| EP1814561A4 (en) | 2004-10-29 | 2012-12-19 | Biocryst Pharm Inc | Therapeutic furopyrimidines and thienopyrimidines |
| US20060116557A1 (en) | 2004-11-30 | 2006-06-01 | Alere Medical Incorporated | Methods and systems for evaluating patient data |
| WO2006063149A1 (en) | 2004-12-09 | 2006-06-15 | Regents Of The University Of Minnesota | Nucleosides with antiviral and anticancer activity |
| WO2006063717A2 (en) | 2004-12-16 | 2006-06-22 | Febit Biotech Gmbh | Polymerase-independent analysis of the sequence of polynucleotides |
| EP1674104A1 (en) | 2004-12-24 | 2006-06-28 | Institut National De La Sante Et De La Recherche Medicale (Inserm) | Uridine derivatives as antiviral drugs against a flaviviridae, especially HCV |
| US20060199738A1 (en) | 2005-03-04 | 2006-09-07 | Sumitomo Chemical Company, Limited | Herbicidal composition |
| WO2006121820A1 (en) | 2005-05-05 | 2006-11-16 | Valeant Research & Development | Phosphoramidate prodrugs for treatment of viral infection |
| WO2007027248A2 (en) | 2005-05-16 | 2007-03-08 | Valeant Research & Development | 3', 5' - cyclic nucleoside analogues for treatment of hcv |
| JP2009504704A (en) * | 2005-08-15 | 2009-02-05 | エフ.ホフマン−ラ ロシュ アーゲー | Antiviral 4'-substituted pronucleotide phosphoramidate |
| US8895531B2 (en) | 2006-03-23 | 2014-11-25 | Rfs Pharma Llc | 2′-fluoronucleoside phosphonates as antiviral agents |
| PT2084174E (en) | 2006-10-10 | 2013-10-08 | Hoffmann La Roche | Preparation of nucleosides ribofuranosyl pyrimidines |
| PL216525B1 (en) | 2006-10-17 | 2014-04-30 | Ct Badań Molekularnych I Makromolekularnych Polskiej Akademii Nauk | 5'-0-[(N-acyl) amidophosphate] - and 5'-0- [(N-acyl) amidothiophosphate]- and 5'-0- [N-acyl) amidodithiophosphate] and 5'-0- [N-acyl) amidoselenophosphate] - nucleosides and method for their manufacture |
| GB0623493D0 (en) | 2006-11-24 | 2007-01-03 | Univ Cardiff | Chemical compounds |
| CA2672613A1 (en) | 2006-12-20 | 2008-07-03 | Istituto Di Ricerche Di Biologia Molecolare P. Angeletti S.P.A. | Nucleoside cyclic phosphoramidates for the treatment of rna-dependent rna viral infection |
| US20080261913A1 (en) | 2006-12-28 | 2008-10-23 | Idenix Pharmaceuticals, Inc. | Compounds and pharmaceutical compositions for the treatment of liver disorders |
| EP2124555B1 (en) | 2007-01-05 | 2015-07-08 | Merck Sharp & Dohme Corp. | Nucleoside aryl phosphoramidates for the treatment of rna-dependent rna viral infection |
| EP2144921A2 (en) | 2007-02-27 | 2010-01-20 | K.U. Leuven Research and Development | Phosphate modified nucleosides useful as substrates for polymerases and as antiviral agents |
| US7964580B2 (en) | 2007-03-30 | 2011-06-21 | Pharmasset, Inc. | Nucleoside phosphoramidate prodrugs |
| GB0709791D0 (en) | 2007-05-22 | 2007-06-27 | Angeletti P Ist Richerche Bio | Antiviral agents |
| CN101108870A (en) | 2007-08-03 | 2008-01-23 | 冷一欣 | Process for preparation of nucleoside phosphoric acid ester compound and application thereof |
| JO2778B1 (en) | 2007-10-16 | 2014-03-15 | ايساي انك | Certain Compounds, Compositions and Methods |
| JP5238939B2 (en) | 2007-11-07 | 2013-07-17 | 三菱化学株式会社 | Long fiber reinforced composite resin composition and molded product |
| WO2009086192A1 (en) | 2007-12-21 | 2009-07-09 | Alios Biopharma, Inc. | Biodegradable phosphate protected nucleotide derivatives and their use as cancer, anti viral and anti parasitic agents |
| US8227431B2 (en) | 2008-03-17 | 2012-07-24 | Hetero Drugs Limited | Nucleoside derivatives |
| US20110130440A1 (en) | 2008-03-26 | 2011-06-02 | Alnylam Pharmaceuticals, Inc. | Non-natural ribonucleotides, and methods of use thereof |
| CA2722308C (en) | 2008-04-15 | 2024-02-27 | Rfs Pharma, Llc. | Nucleoside derivatives for treatment of caliciviridae infections, including norovirus infections |
| US8173621B2 (en) | 2008-06-11 | 2012-05-08 | Gilead Pharmasset Llc | Nucleoside cyclicphosphates |
| WO2010009121A2 (en) | 2008-07-15 | 2010-01-21 | University Of Florida Research Foundation, Inc. | Colon stem cells associated with colitis and colorectal cancer and methods of use |
| EP2346329B1 (en) | 2008-10-09 | 2013-08-21 | Anadys Pharmaceuticals, Inc. | A method of inhibiting hepatitis c virus by combination of a 5,6-dihydro-1h-pyridin-2-one and one or more additional antiviral compounds |
| EP2376515A1 (en) | 2008-12-23 | 2011-10-19 | Pharmasset, Inc. | Synthesis of purine nucleosides |
| SG172361A1 (en) | 2008-12-23 | 2011-07-28 | Pharmasset Inc | Nucleoside analogs |
| AR074897A1 (en) | 2008-12-23 | 2011-02-23 | Pharmasset Inc | NUCLEOSID PHOSPHORAMIDATES |
| JP2012514606A (en) | 2009-01-07 | 2012-06-28 | サイネクシス,インコーポレーテッド | Formulation of cyclosporine derivatives and nucleosides for the treatment of HCV |
| TWI576352B (en) | 2009-05-20 | 2017-04-01 | 基利法瑪席特有限責任公司 | Nucleoside phosphoramidates |
| US8618076B2 (en) | 2009-05-20 | 2013-12-31 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| PT3290428T (en) | 2010-03-31 | 2021-12-27 | Gilead Pharmasset Llc | Tablet comprising crystalline (s)-isopropyl 2-(((s)-(((2r,3r,4r,5r)-5-(2,4-dioxo-3,4-dihydropyrimidin-1 (2h)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)(phenoxy)phosphoryl)amino)propanoate |
| EA025311B1 (en) | 2010-07-19 | 2016-12-30 | Гайлид Сайэнсиз, Инк. | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| US20120107278A1 (en) | 2010-10-29 | 2012-05-03 | Pharmasset, Inc. | Abbreviated hcv therapy for hcv infected patients with il28b c/c genotype |
| US20130109647A1 (en) | 2011-10-31 | 2013-05-02 | Gilead Pharmasset Llc | Methods and compositions for treating hepatitis c virus |
| US8889159B2 (en) | 2011-11-29 | 2014-11-18 | Gilead Pharmasset Llc | Compositions and methods for treating hepatitis C virus |
-
2008
- 2008-03-21 US US12/053,015 patent/US7964580B2/en active Active
- 2008-03-26 SI SI200832077T patent/SI2826784T1/en unknown
- 2008-03-26 DE DE202008018643.9U patent/DE202008018643U1/en not_active Expired - Lifetime
- 2008-03-26 CN CN201811348434.5A patent/CN109776637B/en active Active
- 2008-03-26 NZ NZ579880A patent/NZ579880A/en unknown
- 2008-03-26 RU RU2009139968K patent/RU2651892C2/en active Protection Beyond IP Right Term
- 2008-03-26 EP EP15165114.8A patent/EP2933260A1/en not_active Withdrawn
- 2008-03-26 CN CN202310694712.7A patent/CN116731098A/en active Pending
- 2008-03-26 SG SG2014011704A patent/SG2014011704A/en unknown
- 2008-03-26 EP EP20140151876 patent/EP2792680A1/en not_active Withdrawn
- 2008-03-26 CA CA2682230A patent/CA2682230C/en active Active
- 2008-03-26 PT PT141690602T patent/PT2801580T/en unknown
- 2008-03-26 CN CN200880018024.2A patent/CN101918425B/en active Active
- 2008-03-26 JP JP2010502196A patent/JP5318085B2/en active Active
- 2008-03-26 CN CN201811432281.2A patent/CN109970830A/en active Pending
- 2008-03-26 WO PCT/US2008/058183 patent/WO2008121634A2/en active Application Filing
- 2008-03-26 NZ NZ599206A patent/NZ599206A/en unknown
- 2008-03-26 PL PL08732818T patent/PL2203462T3/en unknown
- 2008-03-26 KR KR1020127004146A patent/KR101525293B1/en active IP Right Grant
- 2008-03-26 CN CN201811280509.0A patent/CN109970818A/en active Pending
- 2008-03-26 KR KR1020097022652A patent/KR101432860B1/en active Protection Beyond IP Right Term
- 2008-03-26 SG SG2012015913A patent/SG179445A1/en unknown
- 2008-03-26 SI SI200831244T patent/SI2203462T1/en unknown
- 2008-03-26 PT PT14179358T patent/PT2826784T/en unknown
- 2008-03-26 CN CN202310695479.4A patent/CN116731099A/en active Pending
- 2008-03-26 EP EP08732818.3A patent/EP2203462B2/en active Active
- 2008-03-26 KR KR1020147036573A patent/KR101527701B1/en active IP Right Grant
- 2008-03-26 MY MYPI20094079A patent/MY147409A/en unknown
- 2008-03-26 CN CN201811280064.6A patent/CN109456373A/en active Pending
- 2008-03-26 BR BRPI0823519A patent/BRPI0823519A2/en not_active Application Discontinuation
- 2008-03-26 MX MX2009010401A patent/MX2009010401A/en active IP Right Grant
- 2008-03-26 SI SI200832216T patent/SI2801580T1/en unknown
- 2008-03-26 EP EP14179358.8A patent/EP2826784B1/en active Active
- 2008-03-26 DK DK08732818.3T patent/DK2203462T3/en active
- 2008-03-26 EP EP14179593.0A patent/EP2824109A1/en not_active Withdrawn
- 2008-03-26 PT PT87328183T patent/PT2203462E/en unknown
- 2008-03-26 CN CN201410569402.3A patent/CN104402955A/en active Pending
- 2008-03-26 RU RU2009139968A patent/RU2651892C3/en active IP Right Grant
- 2008-03-26 PL PL14169060.2T patent/PL2801580T3/en unknown
- 2008-03-26 ES ES08732818.3T patent/ES2492470T3/en active Active
- 2008-03-26 AU AU2008232827A patent/AU2008232827C1/en active Active
- 2008-03-26 PL PL14179358T patent/PL2826784T3/en unknown
- 2008-03-26 ES ES14169060T patent/ES2963716T3/en active Active
- 2008-03-26 BR BRPI0809654-6A patent/BRPI0809654A2/en not_active Application Discontinuation
- 2008-03-26 ES ES14179358T patent/ES2745462T3/en active Active
- 2008-03-26 EP EP23197008.8A patent/EP4282482A3/en active Pending
- 2008-03-26 EP EP14169060.2A patent/EP2801580B1/en active Active
- 2008-03-28 AR ARP080101286A patent/AR066898A1/en not_active Application Discontinuation
- 2008-03-28 TW TW097111505A patent/TWI357332B/en active
- 2008-03-28 CL CL2008000902A patent/CL2008000902A1/en unknown
-
2009
- 2009-09-23 ZA ZA2009/06647A patent/ZA200906647B/en unknown
- 2009-09-29 IL IL201239A patent/IL201239A/en active IP Right Grant
- 2009-10-27 CO CO09120744A patent/CO6260023A2/en active IP Right Grant
-
2011
- 2011-05-03 US US13/099,671 patent/US8334270B2/en active Active
- 2011-12-27 IL IL217228A patent/IL217228A/en active IP Right Grant
-
2012
- 2012-01-16 ZA ZA2012/00310A patent/ZA201200310B/en unknown
- 2012-02-17 JP JP2012032352A patent/JP5539419B2/en active Active
- 2012-09-11 US US13/609,614 patent/US8580765B2/en active Active
- 2012-11-01 IL IL222810A patent/IL222810A0/en active IP Right Grant
- 2012-12-07 RU RU2012152811/04A patent/RU2012152811A/en not_active Application Discontinuation
-
2013
- 2013-08-29 US US14/013,237 patent/US9085573B2/en active Active
- 2013-10-18 US US14/057,675 patent/US8735372B2/en active Active
-
2014
- 2014-04-16 US US14/254,737 patent/US8957046B2/en active Active
- 2014-04-17 US US14/255,833 patent/US8906880B2/en active Active
- 2014-04-30 JP JP2014093905A patent/JP5774749B2/en active Active
- 2014-07-14 HR HRP20140667AT patent/HRP20140667T1/en unknown
- 2014-08-14 CY CY20141100659T patent/CY1115488T1/en unknown
- 2014-09-12 JP JP2014186366A patent/JP2015024998A/en active Pending
- 2014-11-18 NO NO2014029C patent/NO2014029I1/en unknown
- 2014-11-18 CY CY2014047C patent/CY2014047I1/en unknown
- 2014-11-18 HU HUS1400059C patent/HUS1400059I1/en unknown
- 2014-11-19 LU LU92600C patent/LU92600I2/en unknown
- 2014-11-19 FR FR14C0082C patent/FR14C0082I2/en active Active
- 2014-11-19 NL NL300704C patent/NL300704I2/nl unknown
- 2014-11-21 LT LTPA2014040C patent/LTC2203462I2/en unknown
- 2014-12-10 PH PH12014502771A patent/PH12014502771A1/en unknown
-
2015
- 2015-03-12 US US14/656,546 patent/US9585906B2/en active Active
- 2015-07-08 HK HK15106528.1A patent/HK1206033A1/en unknown
- 2015-07-08 HK HK15106529.0A patent/HK1206034A1/en unknown
- 2015-07-13 JP JP2015139458A patent/JP2016023187A/en not_active Withdrawn
-
2017
- 2017-01-04 JP JP2017000051A patent/JP6377781B2/en active Active
- 2017-01-20 US US15/411,506 patent/US10183037B2/en active Active
-
2018
- 2018-01-30 RU RU2018103329A patent/RU2018103329A/en unknown
- 2018-07-24 JP JP2018138161A patent/JP2018197245A/en active Pending
-
2019
- 2019-12-10 JP JP2019223231A patent/JP2020100617A/en active Pending
-
2020
- 2020-10-22 US US17/077,267 patent/US11642361B2/en active Active
-
2021
- 2021-07-26 NO NO2021031C patent/NO2021031I1/en unknown
-
2023
- 2023-11-16 US US18/511,973 patent/US12121529B2/en active Active
Patent Citations (82)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2759300A (en) | 1954-08-11 | 1956-08-21 | Pest Control Ltd | Method and means for introducing a predetermined amount of a poisonous material beneath the surface of the soil |
| US5192549A (en) | 1988-09-28 | 1993-03-09 | Yissum Research Development Company Of The Hebrew University Of Jerusalem | Method of amphiphatic drug loading in liposomes by pH gradient |
| US6132763A (en) | 1988-10-20 | 2000-10-17 | Polymasc Pharmaceuticals Plc | Liposomes |
| US6060080A (en) | 1990-07-16 | 2000-05-09 | Daiichi Pharmaceutical Co., Ltd. | Liposomal products |
| US5376380A (en) | 1990-08-21 | 1994-12-27 | Daiichi Pharmaceutical Co., Ltd. | Method of producing liposomal products from freeze or spray-dried preparations of liposomes |
| US6180134B1 (en) | 1993-03-23 | 2001-01-30 | Sequus Pharmaceuticals, Inc. | Enhanced ciruclation effector composition and method |
| WO1997012033A1 (en) | 1995-09-27 | 1997-04-03 | Emory University | Recombinant hepatitis c virus rna replicase |
| WO2000006529A1 (en) | 1998-07-27 | 2000-02-10 | Istituto Di Ricerche Di Biologia Molecolare P Angeletti S.P.A. | Diketoacid-derivatives as inhibitors of polymerases |
| WO2002057287A2 (en) | 2001-01-22 | 2002-07-25 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2002057425A2 (en) | 2001-01-22 | 2002-07-25 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| US6784166B2 (en) | 2001-06-12 | 2004-08-31 | Syntex (U.S.A.) Llc | 4′-substituted nucleoside derivatives as inhibitors of HCV RNA replication. |
| WO2003006490A1 (en) | 2001-07-11 | 2003-01-23 | Vertex Pharmaceuticals Incorporated | Bridged bicyclic serine protease inhibitors |
| WO2003010141A2 (en) | 2001-07-25 | 2003-02-06 | Boehringer Ingelheim (Canada) Ltd. | Hepatitis c virus polymerase inhibitors with a heterobicyclic structure |
| US20060293306A1 (en) | 2001-07-25 | 2006-12-28 | Beaulieu Pierre L | Viral Polymerase Inhibitors |
| WO2003037895A1 (en) | 2001-11-02 | 2003-05-08 | Glaxo Group Limited | 4-(6-membered)-heteroaryl acyl pyrrolidine derivatives as hcv inhibitors |
| WO2003064456A1 (en) | 2002-02-01 | 2003-08-07 | Boehringer Ingelheim International Gmbh | Tripeptides having a hydroxyproline ether of a substituted quinoline for the inhibition of ns3 (hepatitis c) |
| US20030187018A1 (en) | 2002-02-01 | 2003-10-02 | Boehringer Ingelheim International Gmbh | Hepatitis C inhibitor tri-peptides |
| WO2003101993A1 (en) | 2002-06-04 | 2003-12-11 | Neogenesis Pharmaceuticals, Inc. | Pyrazolo` 1,5a! pyrimidine compounds as antiviral agents |
| WO2003105770A2 (en) | 2002-06-17 | 2003-12-24 | Merck & Co., Inc. | Carbocyclic nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2004000858A2 (en) | 2002-06-21 | 2003-12-31 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2004003138A2 (en) | 2002-06-27 | 2004-01-08 | Merck & Co., Inc. | Nucleoside derivatives as inhibitors of rna-dependent rna viral polymerase |
| WO2004002977A1 (en) | 2002-07-01 | 2004-01-08 | Pharmacia & Upjohn Company Llc | Inhibitors of hcv ns5b polymerase |
| WO2004002944A1 (en) | 2002-07-01 | 2004-01-08 | Pharmacia & Upjohn Company | Inhibitors of hcv ns5b polymerase |
| US20050098125A1 (en) | 2002-07-01 | 2005-05-12 | Thomas Hathaway | Valve lash adjustment apparatus and method |
| WO2004002940A1 (en) | 2002-07-01 | 2004-01-08 | Pharmacia & Upjohn Company | Inhibitors of hcv ns5b polymerase |
| WO2004009020A2 (en) | 2002-07-24 | 2004-01-29 | Merck & Co., Inc. | Pyrrolopyrimidine thionucleoside analogs as antivirals |
| WO2004014852A2 (en) | 2002-08-12 | 2004-02-19 | Bristol-Myers Squibb Company | Iminothiazolidinones as inhibitors of hcv replication |
| WO2004014313A2 (en) | 2002-08-12 | 2004-02-19 | Bristol-Myers Squibb Company | Combination pharmaceutical agents as inhibitors of hcv replication |
| WO2004035571A1 (en) | 2002-10-15 | 2004-04-29 | Rigel Pharmaceuticals, Inc. | Substituted indoles and their use as hcv inhibitors |
| US20040229840A1 (en) | 2002-10-29 | 2004-11-18 | Balkrishen Bhat | Nucleoside derivatives as inhibitors of RNA-dependent RNA viral polymerase |
| WO2004041201A2 (en) | 2002-11-01 | 2004-05-21 | Viropharma Incorporated | Benzofuran compounds, compositions and methods for treatment and prophylaxis of hepatitis c viral infections and associated diseases |
| WO2004065367A1 (en) | 2003-01-22 | 2004-08-05 | Boehringer Ingelheim International Gmbh | Viral polymerase inhibitors |
| WO2004094452A2 (en) | 2003-04-16 | 2004-11-04 | Bristol-Myers Squibb Company | Macrocyclic isoquinoline peptide inhibitors of hepatitis c virus |
| WO2004096210A1 (en) | 2003-05-01 | 2004-11-11 | Glaxo Group Limited | Acylated indoline and tetrahydroquinoline derivatives as hcv inhibitors |
| WO2005003147A2 (en) | 2003-05-30 | 2005-01-13 | Pharmasset, Inc. | Modified fluorinated nucleoside analogues |
| US20050009737A1 (en) | 2003-05-30 | 2005-01-13 | Jeremy Clark | Modified fluorinated nucleoside analogues |
| US20060142238A1 (en) | 2003-07-21 | 2006-06-29 | Mcguigan Christopher | Chemical compounds |
| WO2005021568A2 (en) | 2003-08-27 | 2005-03-10 | Biota, Inc. | Novel tricyclic nucleosides or nucleotides as therapeutic agents |
| WO2005028502A1 (en) | 2003-09-18 | 2005-03-31 | Vertex Pharmaceuticals, Incorporated | Inhibitors of serine proteases, particularly hcv ns3-ns4a protease |
| WO2005037214A2 (en) | 2003-10-14 | 2005-04-28 | Intermune, Inc. | Macrocyclic carboxylic acids and acylsulfonamides as inhibitors of hcv replication |
| US20050267018A1 (en) | 2003-10-14 | 2005-12-01 | Blatt Lawrence M | Macrocyclic compounds as inhibitors of viral replication |
| US20050154056A1 (en) | 2003-11-07 | 2005-07-14 | Pharmacia & Upjohn Company | Inhibitors of HCV NS5B polymerase |
| WO2005067900A2 (en) | 2004-01-06 | 2005-07-28 | Achillion Pharmaceuticals, Inc. | Azabenzofuran substituted thioureas as inhibitors of viral replication |
| WO2005095403A2 (en) | 2004-03-30 | 2005-10-13 | Intermune, Inc. | Macrocyclic compounds as inhibitors of viral replication |
| WO2005103045A1 (en) | 2004-04-22 | 2005-11-03 | Glaxo Group Limited | Acyl dihydro pyrrole derivatives as hcv inhibitors |
| WO2005123087A2 (en) | 2004-06-15 | 2005-12-29 | Merck & Co., Inc. | C-purine nucleoside analogs as inhibitors of rna-dependent rna viral polymerase |
| WO2006012078A2 (en) | 2004-06-24 | 2006-02-02 | Merck & Co., Inc. | Nucleoside aryl phosphoramidates for the treatment of rna-dependent rna viral infection |
| US20060199783A1 (en) | 2004-07-21 | 2006-09-07 | Pharmassett, Inc. | Preparation of alkyl-substituted 2-deoxy-2-fluoro-D-ribofuranosyl pyrimidines and purines and their derivatives |
| US20060166964A1 (en) | 2004-08-09 | 2006-07-27 | Hudyma Thomas W | Inhibitors of HCV replication |
| WO2006020082A1 (en) | 2004-08-09 | 2006-02-23 | Bristol-Myers Squibb Company | Inhibitors of hcv replication |
| US20060040890A1 (en) | 2004-08-23 | 2006-02-23 | Roche Palo Alto Llc | Anti-viral nucleosides |
| US20060040927A1 (en) | 2004-08-23 | 2006-02-23 | Roche Palo Alto Llc | Heterocyclic antiviral compounds |
| US20060122146A1 (en) | 2004-09-14 | 2006-06-08 | Byoung-Kwon Chun | Preparation of 2'-fluoro-2'-alkyl-substituted or other optionally substituted ribofuranosyl pyrimidines and purines and their derivatives |
| WO2006035061A1 (en) | 2004-09-30 | 2006-04-06 | Tibotec Pharmaceuticals Ltd. | Hcv inhibiting bi-cyclic pyrimidines |
| WO2006065335A2 (en) | 2004-10-21 | 2006-06-22 | Merck & Co., Inc. | Fluorinated pyrrolo[2,3-d]pyrimidine nucleosides for the treatment of rna-dependent rna viral infection |
| WO2006065590A2 (en) | 2004-12-16 | 2006-06-22 | Xtl Biopharmaceuticals Inc. | Pyridine and pyrimidine antiviral compositions |
| WO2006093801A1 (en) | 2005-02-25 | 2006-09-08 | Abbott Laboratories | Thiadiazine derivatives useful as anti-infective agents |
| US20060194749A1 (en) | 2005-02-28 | 2006-08-31 | Genelabs Technologies, Inc. | Tricyclic-nucleoside prodrugs for treating viral infections |
| WO2006100310A1 (en) | 2005-03-25 | 2006-09-28 | Tibotec Pharmaceuticals Ltd | Heterobicylic inhibitors of hcv |
| US20060241064A1 (en) | 2005-04-25 | 2006-10-26 | Genelabs Technologies, Inc. | Nucleoside compounds for treating viral infections |
| WO2006120251A1 (en) | 2005-05-12 | 2006-11-16 | Tibotec Pharmaceuticals Ltd. | Pteridines useful as hcv inhibitors and methods for the preparation thereof |
| WO2006120252A2 (en) | 2005-05-12 | 2006-11-16 | Tibotec Pharmaceuticals Ltd. | Pyrido[2,3-d]pyrimidines useful as hcv inhibitors, and methods for the preparation thereof |
| US20060276511A1 (en) | 2005-06-06 | 2006-12-07 | Michael Serrano-Wu | Inhibitors of HCV replication |
| WO2007002602A2 (en) | 2005-06-27 | 2007-01-04 | Depuy Spine, Inc. | Intervertebral disc prosthesis and associated methods |
| WO2007015824A2 (en) | 2005-07-25 | 2007-02-08 | Intermune, Inc. | Novel macrocyclic inhibitors of hepatitis c virus replication |
| WO2007014921A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007014920A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007014926A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007014925A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocylic inhibitors of hepatitis c virus |
| WO2007014922A1 (en) | 2005-07-29 | 2007-02-08 | Tibotec Pharmaceuticals Ltd. | Macrocyclic inhibitors of hepatitis c virus |
| WO2007039145A1 (en) | 2005-09-23 | 2007-04-12 | Glaxo Group Limited | C (2) -heteroarylmethyl-c (4) -pyrazinyl-2-yl acyl pyrrolidine compounds and their use for treating viral infections, especially hepatitis c virus |
| WO2007039142A1 (en) | 2005-09-23 | 2007-04-12 | Glaxo Group Limited | C (2) -heteroarylmethyl-c (4) -methoxymethyl acyl pyrrolidine compounds and their use for treating viral infections, especially hepatitis c virus |
| US20070197463A1 (en) | 2005-12-09 | 2007-08-23 | Pharmasset, Inc. | Antiviral nucleosides |
| WO2007070556A2 (en) | 2005-12-12 | 2007-06-21 | Genelabs Technologies, Inc. | N-(6-membered aromatic ring)-amido anti-viral compounds |
| WO2007076034A2 (en) | 2005-12-21 | 2007-07-05 | Abbott Laboratories | Anti-viral compounds |
| WO2007088148A1 (en) | 2006-02-01 | 2007-08-09 | Smithkline Beecham Corporation | Derivatives of thiophene carboxilic acid as antiviral agent |
| WO2007092000A1 (en) | 2006-02-06 | 2007-08-16 | Bristol-Myers Squibb Company | Inhibitors of hcv replication |
| WO2007095269A2 (en) | 2006-02-14 | 2007-08-23 | Merck & Co., Inc. | Nucleoside aryl phosphoramidates for the treatment of rna-dependent rna viral infection |
| WO2007093901A1 (en) | 2006-02-17 | 2007-08-23 | Pfizer Limited | 3 -deazapurine derivatives as tlr7 modulators |
| US20070275947A1 (en) | 2006-05-25 | 2007-11-29 | Bristol-Myers Squibb Company | Cyclopropyl Fused Indolobenzazepine HCV NS5B Inhibitors |
| WO2008010921A2 (en) | 2006-07-07 | 2008-01-24 | Gilead Sciences, Inc. | Modulators of pharmacokinetic properties of therapeutics |
| EP1881001A1 (en) | 2006-07-20 | 2008-01-23 | Tibotec Pharmaceuticals Ltd. | HCV NS-3 serine protease inhibitors |
Non-Patent Citations (52)
| Title |
|---|
| BALZARINI, J. ET AL., PROC. NATIONAL ACAD SCI USA, vol. 93, 1996, pages 7295 - 7299 |
| BARTENSCHLAGER ET AL., J. VIROL., vol. 67, 1993, pages 3835 - 3844 |
| BARTENSCHLAGER ET AL., J. VIROL., vol. 68, 1994, pages 5045 - 5055 |
| BAZAN; FLETTERICK, VIROLOGY, vol. 171, 1989, pages 637 - 639 |
| BEAULIEU, P. L. ET AL., CURRENT OPINION IN INVESTIGATIONAL DRUGS, vol. 5, 2004, pages 838 - 850 |
| BEHRENS ET AL., EMBO, vol. 15, 1996, pages 12 - 22 |
| C. MCGUIGAN ET AL., ANTIVIRAL RES., vol. 17, 1992, pages 311 - 321 |
| CALISHER ET AL., J. GEN. VIROL, vol. 70, 1993, pages 37 - 43 |
| CARROL, S. ET AL., INFECTIOUS DISORDERS-DRUG TARGETS, vol. 6, 2006, pages 17 - 29 |
| CLARK, J. ET AL., J. MED. CHEM., vol. 48, 2005, pages 5504 - 5508 |
| CLARK, J. MED. CHEM., vol. 48, 2005, pages 5504 - 5508 |
| D. CURLEY ET AL., ANTIVIRAL RES., vol. 14, 1990, pages 345 - 356 |
| E. W. MARTIN,: "Remington: The Science and Practice of Pharmacy, 19th edition,", 1995, MACK PUBLISHING COMPANY |
| ECKART ET AL., BIOCHEM. BIOPHYS. RES. COMM., vol. 192, 1993, pages 399 - 406 |
| EISENBERG, E. J. ET AL., NUCLEOSIDES, NUCLEOTIDES AND NUCLEIC ACIDS, vol. 20, 2001, pages 1091 - 1098 |
| FAILLA ET AL., J. VIROL., vol. 68, 1994, pages 3753 - 3760 |
| FIELDS, B. N., KNIPE, D. M., AND HOWLEY, P. M.,: "Fields Virology", 1996, LIPPINCOTT-RAVEN PUBLISHERS, pages: 931 - 959 |
| GORBALENYA ET AL., NATURE, vol. 333, 1988, pages 22 |
| GORBALENYA ET AL., NUCLEIC ACID RES., vol. 17, 1989, pages 3889 - 3897 |
| GRAKOUI ET AL., J. VIROL., vol. 67, 1993, pages 2832 - 2843 |
| GRAKOUI ET AL., PROC. NATL. ACAD SCI. USA, vol. 90, 1993, pages 10583 - 10587 |
| GRIFFITH, R.C. ET AL., ANNUAL REPORTS IN MEDICINAL CHEMISTRY, vol. 39, 2004, pages 223 - 237 |
| HALSTEAD, S. B., REV. INFECT. DIS., vol. 6, 1984, pages 251 - 264 |
| HALSTEAD, S. B., SCIENCE, vol. 239, 1988, pages 476 - 481 |
| HIJIKATA ET AL., J. VIROL., vol. 67, 1993, pages 4665 - 4675 |
| JIN; PETERSON, ARCH. BIOCHEM. BIOPHYS., vol. 323, 1995, pages 47 - 53 |
| K. ISHI ET AL., HEPTOLOGY, vol. 29, 1999, pages 1227 - 1235 |
| KIM ET AL., BIOCHEM. BIOPHYS. RES. COMM., vol. 215, 1995, pages 160 - 166 |
| KOONIN, E.V.; DOLJA, V.V., CRIR. REV. BIOCHEM. MOLEC. BIOL., vol. 28, 1993, pages 375 - 430 |
| LECHMANN ET AL., J. VIROL., vol. 71, 1997, pages 8416 - 8428 |
| LEE, W.A. ET AL., ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, vol. 49, 2005, pages 1898 |
| MARCH: "Advanced Organic Chemistry" |
| MCGUIGAN ET AL., ANTIVIRAL CHEM. CHEMOTHER, vol. 19901, no. 2, pages 107 - 113 |
| MCGUIGAN, C. ET AL., J. MED. CHEM., vol. 39, 1996, pages 1748 - 1753 |
| MEYERS, G.; THIEL, H.J., ADVANCES IN VIRUS RESEARCH, vol. 47, 1996, pages 53 - 118 |
| MOENNIG V. ET AL., ADV. VIR. RES., vol. 41, 1992, pages 53 - 98 |
| MOENNIG, V. ET AL., ADV. VIR. RES., vol. 41, 1992, pages 53 - 98 |
| MONATH, T. P., NEW ENG. J. MED, vol. 319, no. 64, 1988, pages 1 - 643 |
| NI, Z-J. ET AL., CURRENT OPINION IN DRUG DISCOVERY AND DEVELOPMENT, vol. 7, 2004, pages 446 - 459 |
| SIDDIQUI, A. Q. ET AL., J. MED. CHEM., vol. 42, 1999, pages 4122 - 4128 |
| T.W. GREENE; P.G. M. WUTS: "Protective Groups in Organic Synthesis" 3rd ed.,", 1999, JOHN WILEY & SONS |
| TAN, S.-L. ET AL., NATURE REV. DRUG DISCOV., vol. 1, 2002, pages 867 - 881 |
| TOME ET AL., J. VIROL., vol. 67, 1993, pages 4017 - 4026 |
| V. LOHMANN ET AL., VIROLOGY, vol. 249, 1998, pages 108 - 118 |
| VALETTE, G. ET AL., J. MED. CHEM., vol. 39, 1996, pages 1981 - 1990 |
| WALKER, M.P. ET AL., EXP. OPIN. INVESTIGATIONAL DRUGS, vol. 12, 2003, pages 1269 - 1280 |
| WARRENER; COLLETT, J. VIROL., vol. 69, 1995, pages 1720 - 1726 |
| WISKERCHEN; COLLETT, VIROLOGY, vol. 184, 1991, pages 341 - 350 |
| WU, J. ET AL., CURRENT DRUG TARGETS-INFECTIOUS DISORDERS, vol. 3, 2003, pages 207 - 219 |
| XU ET AL., J. VIROL., vol. 71, no. 53, 1997, pages 12 - 5322 |
| YUAN ET AL., BIOCHEM. BIOPHYS. RES. COMM., vol. 232, 1997, pages 231 - 235 |
| ZHONG ET AL., J. VIROL., vol. 72, 1998, pages 9365 - 9369 |
Cited By (314)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9585906B2 (en) | 2007-03-30 | 2017-03-07 | Gilead Pharmasset Llc | Nucleoside phosphoramidate prodrugs |
| US12121529B2 (en) | 2007-03-30 | 2024-10-22 | Gilead Sciences, Inc. | Nucleoside phosphoramidate prodrugs |
| US10183037B2 (en) | 2007-03-30 | 2019-01-22 | Gilead Pharmasset Llc | Nucleoside phosphoramidate prodrugs |
| US11642361B2 (en) | 2007-03-30 | 2023-05-09 | Gilead Sciences, Inc. | Nucleoside phosphoramidate prodrugs |
| US9085573B2 (en) | 2007-03-30 | 2015-07-21 | Gilead Pharmasset Llc | Nucleoside phosphoramidate prodrugs |
| US9676808B2 (en) | 2007-11-20 | 2017-06-13 | Gilead Pharmasset Llc | 2′,4′-substituted nucleosides as antiviral agents |
| US9296777B2 (en) | 2007-11-20 | 2016-03-29 | Gilead Pharmasset Llc | 2′,4′-substituted nucleosides as antiviral agents |
| US8853171B2 (en) | 2008-04-23 | 2014-10-07 | Gilead Sciences, Inc. | 1′-substituted carba-nucleoside analogs for antiviral treatment |
| USRE46762E1 (en) | 2008-04-23 | 2018-03-27 | Gilead Sciences, Inc | 1′-substituted carba-nucleoside analogs for antiviral treatment |
| US8815829B2 (en) | 2008-12-09 | 2014-08-26 | Rfs Pharma, Llc | 3′-azido purine nucleotide prodrugs for treatment of viral infections |
| CN102695513A (en) * | 2008-12-23 | 2012-09-26 | 吉利德制药有限责任公司 | Nucleoside phosphoramidates |
| US8957045B2 (en) | 2008-12-23 | 2015-02-17 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| JP2012513479A (en) * | 2008-12-23 | 2012-06-14 | ギリアド ファーマセット エルエルシー | Synthesis of purine nucleosides |
| WO2010075517A3 (en) * | 2008-12-23 | 2012-06-21 | Pharmasset, Inc. | Nucleoside phosphoramidate prodrugs |
| JP2012515714A (en) * | 2008-12-23 | 2012-07-12 | ギリアド ファーマセット エルエルシー | Nucleoside phosphoramidate |
| JP2012516287A (en) * | 2008-12-23 | 2012-07-19 | ギリアド ファーマセット エルエルシー | Nucleoside analogues |
| WO2010075549A3 (en) * | 2008-12-23 | 2012-08-09 | Pharmasset, Inc. | Nucleoside phosphoramidates |
| WO2010075517A2 (en) | 2008-12-23 | 2010-07-01 | Pharmasset, Inc. | Nucleoside analogs |
| EP3222628A1 (en) | 2008-12-23 | 2017-09-27 | Gilead Pharmasset LLC | Nucleoside phosphoramidates |
| EP2671888A1 (en) | 2008-12-23 | 2013-12-11 | Gilead Pharmasset LLC | 3',5'-cyclic nucleoside phosphate analogues |
| US9045520B2 (en) | 2008-12-23 | 2015-06-02 | Gilead Pharmasset Llc | Synthesis of purine nucleosides |
| WO2010075554A1 (en) | 2008-12-23 | 2010-07-01 | Pharmasset, Inc. | Synthesis of purine nucleosides |
| WO2010075549A2 (en) | 2008-12-23 | 2010-07-01 | Pharmasset, Inc. | Nucleoside phosphoramidates |
| US8551973B2 (en) | 2008-12-23 | 2013-10-08 | Gilead Pharmasset Llc | Nucleoside analogs |
| AU2009329917B2 (en) * | 2008-12-23 | 2016-03-31 | Gilead Pharmasset Llc | Nucleoside analogs |
| JP2015221802A (en) * | 2008-12-23 | 2015-12-10 | ギリアド ファーマセット エルエルシー | Nucleoside analogs |
| JP2015180628A (en) * | 2008-12-23 | 2015-10-15 | ギリアド ファーマセット エルエルシー | synthesis of purine nucleoside |
| US8716262B2 (en) | 2008-12-23 | 2014-05-06 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| JP2013082728A (en) * | 2008-12-23 | 2013-05-09 | Gilead Pharmasset Llc | Synthesis of purine nucleosides |
| EA019341B1 (en) * | 2008-12-23 | 2014-02-28 | Джилид Фармассет, Ллс. | Nucleoside phosphoramidates |
| EA019295B1 (en) * | 2008-12-23 | 2014-02-28 | Джилид Фармассет, Ллс. | Synthesis of purine nucleosides and process for preparing them |
| US9173893B2 (en) | 2009-02-06 | 2015-11-03 | Cocrystal Pharma, Inc. | Purine nucleoside monophosphate prodrugs for treatment of cancer and viral infections |
| EP2393815A2 (en) * | 2009-02-06 | 2011-12-14 | RFS Pharma, LLC. | Purine nucleoside monophosphate prodrugs for treatment of cancer and viral infections |
| EP2393815A4 (en) * | 2009-02-06 | 2012-11-21 | Rfs Pharma Llc | Purine nucleoside monophosphate prodrugs for treatment of cancer and viral infections |
| US8609627B2 (en) | 2009-02-06 | 2013-12-17 | Rfs Pharma, Llc | Purine nucleoside monophosphate prodrugs for treatment of cancer and viral infections |
| KR101774429B1 (en) * | 2009-02-06 | 2017-09-04 | 코크리스탈 파마, 아이엔씨. | Purine nucleoside monophosphate prodrugs for treatment of cancer and viral infections |
| US9206217B2 (en) | 2009-05-20 | 2015-12-08 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| US8735569B2 (en) | 2009-05-20 | 2014-05-27 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| KR101603817B1 (en) * | 2009-05-20 | 2016-03-16 | 길리어드 파마셋 엘엘씨 | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| US9637512B2 (en) | 2009-05-20 | 2017-05-02 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| EP2913337A1 (en) | 2009-05-20 | 2015-09-02 | Gilead Pharmasset LLC | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| CN102459299B (en) * | 2009-05-20 | 2015-09-30 | 吉利德制药有限责任公司 | N-[(2 ' R)-2 '-deoxidation-2 '-fluoro-2 '-methyl-P-phenyl-5 '-uridine acyl group]-ALANINE 1-methylethyl ester and preparation method thereof |
| US8618076B2 (en) | 2009-05-20 | 2013-12-31 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| EP2610264A2 (en) | 2009-05-20 | 2013-07-03 | Gilead Pharmasset LLC | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| US8629263B2 (en) | 2009-05-20 | 2014-01-14 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| US8633309B2 (en) | 2009-05-20 | 2014-01-21 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| EP2610264A3 (en) * | 2009-05-20 | 2014-01-22 | Gilead Pharmasset LLC | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| CN102459299A (en) * | 2009-05-20 | 2012-05-16 | 法莫赛特股份有限公司 | N- [ (2 ' r) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -l-alanine 1-methylethyl ester and process for its production |
| CN105085592A (en) * | 2009-05-20 | 2015-11-25 | 吉利德制药有限责任公司 | N- [ (2 ' R) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -L-alanine 1-methylethyl ester and process for its production |
| EP3321275A1 (en) | 2009-05-20 | 2018-05-16 | Gilead Pharmasset LLC | Crystalline form of sofosbuvir |
| CN105085592B (en) * | 2009-05-20 | 2021-04-27 | 吉利德制药有限责任公司 | N- [ (2' R) -2' -deoxy-2 ' -fluoro-2 ' -methyl-P-phenyl-5 ' -uridylyl ] -L-alanine 1-methylethyl ester and process for preparing same |
| US9284342B2 (en) | 2009-05-20 | 2016-03-15 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| CN105198949A (en) * | 2009-05-20 | 2015-12-30 | 吉利德制药有限责任公司 | Nucleoside Phosphoramidates |
| EP2910562A1 (en) | 2009-05-20 | 2015-08-26 | Gilead Pharmasset LLC | N-[(2'r)-2'-deoxy-2 '-fluoro-2'-methyl-p-phenyl-5 '-uridylyl]-l-alanine 1-methylethyl ester in crystalline form |
| JP2016053045A (en) * | 2009-05-20 | 2016-04-14 | ギリアド ファーマセット エルエルシー | N-[(2 'r)-2 '-deoxy-2 '-fluoro-2 '-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| EA028742B1 (en) * | 2009-05-20 | 2017-12-29 | Джилид Фармассет, Ллс | Intermediate compounds useful for preparing nucleoside phosphoramidates |
| EA026731B1 (en) * | 2009-05-20 | 2017-05-31 | Джилид Фармассет Ллс | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| AU2016200676B2 (en) * | 2009-05-20 | 2017-06-01 | Gilead Sciences, Inc. | N-[(2'R)-2 '-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl] -L-alanine 1-methylethyl ester and process for its production |
| JP2012527477A (en) * | 2009-05-20 | 2012-11-08 | ギリアド ファーマセット エルエルシー | N-[(2'R) -2'-deoxy-2'-fluoro-2'-methyl-P-phenyl-5'-uridylyl] -L-alanine 1-methylethyl ester and process for its preparation |
| WO2010135569A1 (en) | 2009-05-20 | 2010-11-25 | Pharmasset, Inc. | N- [ (2 ' r) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -l-alanine 1-methylethyl ester and process for its production |
| KR20140147144A (en) * | 2009-05-20 | 2014-12-29 | 길리어드 파마셋 엘엘씨 | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| CN104292256A (en) * | 2009-05-20 | 2015-01-21 | 吉利德制药有限责任公司 | N- [ (2 ' R) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -L-alanine 1-methylethyl ester and process for its production |
| EA028709B1 (en) * | 2009-05-20 | 2017-12-29 | Джилид Фармассет, Ллс | N-[(2'R)-2'-DEOXY-2'-FLUORO-2'-METHYL-p-PHENYL-5'-URIDYL]-L-ALANINE 1-METHYLETHYL ESTER AND PROCESS FOR PRODUCTION THEREOF |
| JP2015028060A (en) * | 2009-05-20 | 2015-02-12 | ギリアド ファーマセット エルエルシー | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| AU2014274548B2 (en) * | 2009-05-20 | 2016-02-04 | Gilead Sciences, Inc. | N- [ (2 ' R) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -L-alanine 1-methylethyl ester and process for its production |
| AU2010249481B2 (en) * | 2009-05-20 | 2015-01-22 | Gilead Sciences, Inc. | N- [ (2 ' R) -2 ' -deoxy-2 ' -fluoro-2 ' -methyl-p-phenyl-5 ' -uridylyl] -L-alanine 1-methylethyl ester and process for its production |
| KR101599183B1 (en) * | 2009-05-20 | 2016-03-03 | 길리어드 파마셋 엘엘씨 | N-[(2'r)-2'-deoxy-2'-fluoro-2'-methyl-p-phenyl-5'-uridylyl]-l-alanine 1-methylethyl ester and process for its production |
| WO2011005860A2 (en) | 2009-07-07 | 2011-01-13 | Alnylam Pharmaceuticals, Inc. | 5' phosphate mimics |
| US10988498B2 (en) | 2009-09-21 | 2021-04-27 | Gilead Sciences, Inc. | Processes and intermediates for the preparation of 1′-substituted carba-nucleoside analogs |
| US8822496B2 (en) | 2009-10-30 | 2014-09-02 | Boehringer Ingelheim International Gmbh | Dosage regimens for HCV combination therapy |
| JP2013514276A (en) * | 2009-12-18 | 2013-04-25 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | HCV combination therapy |
| KR101715981B1 (en) | 2010-03-31 | 2017-03-13 | 길리애드 파마셋 엘엘씨 | Nucleoside phosphoramidates |
| CN102858790A (en) * | 2010-03-31 | 2013-01-02 | 吉利德制药有限责任公司 | Nucleoside Phosphoramidates |
| CN102906102A (en) * | 2010-03-31 | 2013-01-30 | 吉利德制药有限责任公司 | Stereoselective synthesis of phosphorus containing actives |
| JP2015205903A (en) * | 2010-03-31 | 2015-11-19 | ギリード・ファーマセット・エルエルシー | nucleoside phosphoramidate |
| US8859756B2 (en) | 2010-03-31 | 2014-10-14 | Gilead Pharmasset Llc | Stereoselective synthesis of phosphorus containing actives |
| JP2013523767A (en) * | 2010-03-31 | 2013-06-17 | ギリード・ファーマセット・エルエルシー | Nucleoside phosphoramidate |
| JP2013525277A (en) * | 2010-03-31 | 2013-06-20 | ギリード・ファーマセット・エルエルシー | Purine nucleoside phosphoramidate |
| WO2011123672A1 (en) | 2010-03-31 | 2011-10-06 | Pharmasset, Inc. | Purine nucleoside phosphoramidate |
| EP2609923A2 (en) | 2010-03-31 | 2013-07-03 | Gilead Pharmasset LLC | Nucleoside Phosphoramidates |
| KR101759369B1 (en) | 2010-03-31 | 2017-07-18 | 길리애드 파마셋 엘엘씨 | Stereoselective synthesis of phosphorus containing actives |
| TWI498117B (en) * | 2010-03-31 | 2015-09-01 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| WO2011123645A2 (en) | 2010-03-31 | 2011-10-06 | Pharmasset, Inc. | Nucleoside phosphoramidates |
| CN104017020A (en) * | 2010-03-31 | 2014-09-03 | 吉利德制药有限责任公司 | Nucleoside phosphoramidates |
| AU2011235112B2 (en) * | 2010-03-31 | 2015-07-09 | Gilead Sciences, Inc. | Nucleoside phosphoramidates |
| WO2011123645A3 (en) * | 2010-03-31 | 2011-12-29 | Pharmasset, Inc. | Nucleoside phosphoramidates |
| EP2609923A3 (en) * | 2010-03-31 | 2014-07-30 | Gilead Pharmasset LLC | Nucleoside Phosphoramidates |
| EA026341B1 (en) * | 2010-03-31 | 2017-03-31 | ГАЙЛИД ФАРМАССЕТ ЭлЭлСи | Crystalline form of nucleoside phosphoramidate |
| WO2011123668A3 (en) * | 2010-03-31 | 2012-05-24 | Pharmasset, Inc. | Stereoselective synthesis of phosphorus containing actives |
| EP3290428A1 (en) | 2010-03-31 | 2018-03-07 | Gilead Pharmasset LLC | Tablet comprising crystalline (s)-isopropyl 2-(((s)-(((2r,3r,4r,5r)-5-(2,4-dioxo-3,4-dihydropyrimidin-1 (2h)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)(phenoxy)phosphoryl)amino)propanoate |
| CN104017020B (en) * | 2010-03-31 | 2017-04-12 | 吉利德制药有限责任公司 | Nucleoside phosphoramidates |
| KR20120138242A (en) * | 2010-03-31 | 2012-12-24 | 길리어드 파마셋 엘엘씨 | Nucleoside phosphoramidates |
| AP3515A (en) * | 2010-03-31 | 2016-01-11 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| KR20130130690A (en) * | 2010-07-19 | 2013-12-02 | 길리애드 사이언시즈, 인코포레이티드 | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| KR20190077606A (en) * | 2010-07-19 | 2019-07-03 | 길리애드 사이언시즈, 인코포레이티드 | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| EP2805960A1 (en) * | 2010-07-19 | 2014-11-26 | Gilead Sciences, Inc. | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| WO2012012465A1 (en) * | 2010-07-19 | 2012-01-26 | Clarke, Michael, O'neil Hanrahan | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| EA025311B1 (en) * | 2010-07-19 | 2016-12-30 | Гайлид Сайэнсиз, Инк. | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| CN103052646A (en) * | 2010-07-19 | 2013-04-17 | 吉里德科学公司 | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| US9487544B2 (en) | 2010-07-19 | 2016-11-08 | Gilead Sciences, Inc. | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| US9090642B2 (en) | 2010-07-19 | 2015-07-28 | Gilead Sciences, Inc. | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| AU2011282241B2 (en) * | 2010-07-19 | 2015-07-30 | Gilead Sciences, Inc. | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| KR102108864B1 (en) | 2010-07-19 | 2020-05-12 | 길리애드 사이언시즈, 인코포레이티드 | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| KR101995598B1 (en) * | 2010-07-19 | 2019-07-02 | 길리애드 사이언시즈, 인코포레이티드 | Methods for the preparation of diasteromerically pure phosphoramidate prodrugs |
| JP2013537527A (en) * | 2010-07-19 | 2013-10-03 | ギリード・サイエンシズ・インコーポレーテッド | Process for the preparation of diastereomeric pure phosphoramidate prodrugs |
| US11492353B2 (en) | 2010-07-22 | 2022-11-08 | Gilead Sciences, Inc. | Methods and compounds for treating Paramyxoviridae virus infections |
| US10696679B2 (en) | 2010-07-22 | 2020-06-30 | Gilead Sciences, Inc. | Methods and compounds for treating paramyxoviridae virus infections |
| US10065958B2 (en) | 2010-07-22 | 2018-09-04 | Gilead Sciences, Inc. | Methods and compounds for treating Paramyxoviridae virus infections |
| CN103209987A (en) * | 2010-09-22 | 2013-07-17 | 艾丽奥斯生物制药有限公司 | Substituted nucleotide analogs |
| JP2016065080A (en) * | 2010-09-22 | 2016-04-28 | アリオス バイオファーマ インク. | Substituted nucleotide analog |
| JP2013537907A (en) * | 2010-09-22 | 2013-10-07 | アリオス バイオファーマ インク. | Substituted nucleotide analogs |
| US9346848B2 (en) | 2010-09-22 | 2016-05-24 | Alios Biopharma, Inc. | Azido nucleosides and nucleotide analogs |
| CN105061534A (en) * | 2010-09-22 | 2015-11-18 | 艾丽奥斯生物制药有限公司 | Substituted nucleotide analogs |
| US8877731B2 (en) | 2010-09-22 | 2014-11-04 | Alios Biopharma, Inc. | Azido nucleosides and nucleotide analogs |
| CN103209987B (en) * | 2010-09-22 | 2017-06-06 | 艾丽奥斯生物制药有限公司 | Substituted nucleotide analog |
| US9278990B2 (en) | 2010-09-22 | 2016-03-08 | Alios Biopharma, Inc. | Substituted nucleotide analogs |
| US8841275B2 (en) | 2010-11-30 | 2014-09-23 | Gilead Pharmasset Llc | 2′-spiro-nucleosides and derivatives thereof useful for treating hepatitis C virus and dengue virus infections |
| US11925658B2 (en) | 2011-03-01 | 2024-03-12 | NuCana plc | Phosphoramidate derivatives of 5-fluoro-2′—deoxyuridine for use in the treatment of cancer |
| US11559542B2 (en) | 2011-03-01 | 2023-01-24 | NuCana plc | Phosphoramidate derivatives of 5-fluoro-2′-deoxyuridine for use in the treatment of cancer |
| US9221866B2 (en) | 2011-03-01 | 2015-12-29 | Nucana Biomed Limited | Phosphoramidate derivatives of 5-fluoro-2′-deoxyuridine for use in the treatment of cancer |
| US8933053B2 (en) | 2011-03-01 | 2015-01-13 | Nucana Biomed Limited | Phosphoramidate derivatives of 5-fluoro-2′-deoxyuridine for use in the treatment of cancer |
| US10022390B2 (en) | 2011-03-01 | 2018-07-17 | NuCana plc | Phosphoramidate derivatives of 5-fluoro-2′-deoxyuridine for use in the treatment of cancer |
| US10993957B2 (en) | 2011-03-01 | 2021-05-04 | NuCana plc | Phosphoramidate derivatives of 5-fluoro-2′-deoxyuridine for use in the treatment of cancer |
| US9655915B2 (en) | 2011-03-01 | 2017-05-23 | Nucana Biomed Limited | Phosphoramidate derivatives of 5-fluoro-2′-deoxyuridine for use in the treatment of cancer |
| WO2012142523A2 (en) | 2011-04-13 | 2012-10-18 | Gilead Sciences, Inc. | 1'-substituted pyrimidine n-nucleoside analogs for antiviral treatment |
| EP2697241B1 (en) * | 2011-04-13 | 2019-06-12 | Gilead Sciences, Inc. | 1'-substituted pyrimidine n-nucleoside analogs for antiviral treatment |
| WO2012158811A3 (en) * | 2011-05-19 | 2013-02-28 | Rfs Pharma, Llc | Purine monophosphate prodrugs for treatment of viral infections |
| WO2013039920A1 (en) * | 2011-09-12 | 2013-03-21 | Idenix Pharmaceuticals, Inc. | Substituted carbonyloxymethylphosphoramidate compounds and pharmaceutical compositions for the treatment of viral infections |
| US10456414B2 (en) | 2011-09-16 | 2019-10-29 | Gilead Pharmasset Llc | Methods for treating HCV |
| US9393256B2 (en) | 2011-09-16 | 2016-07-19 | Gilead Pharmasset Llc | Methods for treating HCV |
| EP2583680A3 (en) * | 2011-10-21 | 2013-06-12 | Abbvie Inc. | Mono (PSI-7977) or combination treatment of DAAs for use in treating HCV |
| PT107665A (en) * | 2011-10-21 | 2014-07-16 | Abbvie Inc | METHODS FOR THE TREATMENT OF HCV COMPREHENDING AT LEAST TWO ANTIVIRAL AGENTS OF DIRECT ACTIVITY, RIBAVIRIN, BUT NOT INTERFERED |
| US9452194B2 (en) | 2011-10-21 | 2016-09-27 | Abbvie Inc. | Methods for treating HCV |
| US8809265B2 (en) | 2011-10-21 | 2014-08-19 | Abbvie Inc. | Methods for treating HCV |
| US8853176B2 (en) | 2011-10-21 | 2014-10-07 | Abbvie Inc. | Methods for treating HCV |
| US9549941B2 (en) | 2011-11-29 | 2017-01-24 | Gilead Pharmasset Llc | Compositions and methods for treating hepatitis C virus |
| US8889159B2 (en) | 2011-11-29 | 2014-11-18 | Gilead Pharmasset Llc | Compositions and methods for treating hepatitis C virus |
| EP3750544A2 (en) | 2011-11-30 | 2020-12-16 | Emory University | Jak inhibitors for use in the prevention or treatment of viral infection |
| KR101765997B1 (en) | 2011-12-20 | 2017-08-07 | 리보사이언스 엘엘씨 | 2',4'-difluoro-2'-methyl substituted nucleoside derivatives as inhibitors of hcv rna replication |
| US9073960B2 (en) | 2011-12-22 | 2015-07-07 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| JP2015503506A (en) * | 2011-12-22 | 2015-02-02 | アリオス バイオファーマ インク. | Substituted nucleosides, nucleotides and analogs thereof |
| US11021509B2 (en) | 2011-12-22 | 2021-06-01 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US10464965B2 (en) | 2011-12-22 | 2019-11-05 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US9441007B2 (en) | 2012-03-21 | 2016-09-13 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US10485815B2 (en) | 2012-03-21 | 2019-11-26 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| USRE48171E1 (en) | 2012-03-21 | 2020-08-25 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US9012427B2 (en) | 2012-03-22 | 2015-04-21 | Alios Biopharma, Inc. | Pharmaceutical combinations comprising a thionucleotide analog |
| WO2013177219A1 (en) * | 2012-05-22 | 2013-11-28 | Idenix Pharmaceuticals, Inc. | D-amino acid compounds for liver disease |
| AU2013266393B2 (en) * | 2012-05-22 | 2017-09-28 | Idenix Pharmaceuticals Llc | D-amino acid compounds for liver disease |
| US10717758B2 (en) * | 2012-05-22 | 2020-07-21 | Idenix Pharmaceuticals Llc | D-amino acid compounds for liver disease |
| US20180258130A1 (en) * | 2012-05-22 | 2018-09-13 | Idenix Pharmaceuticals Llc | D-amino acid compounds for liver disease |
| EA031301B1 (en) * | 2012-05-22 | 2018-12-28 | Иденикс Фармасьютикалз Ллс | D-amino acid chemical compounds for treating liver diseases |
| US10774106B2 (en) | 2012-05-25 | 2020-09-15 | Janssen Sciences Ireland Unlimited Company | Uracyl spirooxetane nucleosides |
| US9845336B2 (en) | 2012-05-25 | 2017-12-19 | Janssen Sciences Ireland Uc | Uracyl spirooxetane nucleosides |
| US9422323B2 (en) | 2012-05-25 | 2016-08-23 | Janssen Sciences Ireland Uc | Uracyl spirooxetane nucleosides |
| US10301347B2 (en) | 2012-05-25 | 2019-05-28 | Janssen Sciences Ireland Unlimited Company | Uracyl spirooxetane nucleosides |
| US10544184B2 (en) | 2012-05-25 | 2020-01-28 | Janssen Sciences Ireland Unlimited Company | Uracyl spirooxetane nucleosides |
| US10040814B2 (en) | 2012-05-25 | 2018-08-07 | Janssen Sciences Ireland Uc | Uracyl spirooxetane nucleosides |
| WO2014008236A1 (en) | 2012-07-03 | 2014-01-09 | Bristol-Myers Squibb Company | Process for preparing diastereomerically enriched phosphoramidate derivatives of nucleoside compounds for treatment of viral infections |
| AU2013329521B2 (en) * | 2012-10-08 | 2018-04-19 | Centre National De La Recherche Scientifique | 2'-chloro nucleoside analogs for HCV infection |
| KR20150084830A (en) * | 2012-10-08 | 2015-07-22 | 아이데닉스 파마슈티칼스, 인코포레이티드 | 2'-chloro nucleoside analogs for hcv infection |
| WO2014058801A1 (en) * | 2012-10-08 | 2014-04-17 | Idenix Pharmaceuticals, Inc. | 2'-chloro nucleoside analogs for hcv infection |
| EA030189B1 (en) * | 2012-10-08 | 2018-07-31 | Иденикс Фармасьютикалз Ллс | 2'-chloro nucleoside analogs for hcv infection |
| KR102001280B1 (en) * | 2012-10-08 | 2019-07-17 | 아이데닉스 파마슈티칼스 엘엘씨 | 2'-chloro nucleoside analogs for hcv infection |
| WO2014078427A1 (en) * | 2012-11-14 | 2014-05-22 | Idenix Pharmaceuticals, Inc. | D-alanine ester of rp-nucleoside analog |
| WO2014078436A1 (en) * | 2012-11-14 | 2014-05-22 | Idenix Pharmaceuticals, Inc. | D-alanine ester of sp-nucleoside analog |
| WO2014099941A1 (en) * | 2012-12-19 | 2014-06-26 | Idenix Pharmaceuticals, Inc. | 4'-fluoro nucleosides for the treatment of hcv |
| US10112966B2 (en) | 2012-12-21 | 2018-10-30 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US11485753B2 (en) | 2012-12-21 | 2022-11-01 | Janssen Pharmaceutica Nv | Substituted nucleosides, nucleotides and analogs thereof |
| US10683320B2 (en) | 2012-12-21 | 2020-06-16 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US10487104B2 (en) | 2012-12-21 | 2019-11-26 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| TWI641615B (en) * | 2012-12-21 | 2018-11-21 | 美商艾洛斯生物製藥公司 | Substituted nucleosides, nucleotides and analogs thereof |
| AU2018203337B2 (en) * | 2012-12-21 | 2019-12-05 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| EP3912984A1 (en) * | 2012-12-21 | 2021-11-24 | Janssen BioPharma, Inc. | 4'-fluoro-nucleosides, 4'-fluoro-nucleotides and analogs thereof for the treatment of hcv |
| US9249174B2 (en) | 2012-12-21 | 2016-02-02 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US10793591B2 (en) | 2012-12-21 | 2020-10-06 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| EP2935303A4 (en) * | 2012-12-21 | 2016-08-10 | Alios Biopharma Inc | Substituted nucleosides, nucleotides and analogs thereof |
| WO2014100505A1 (en) | 2012-12-21 | 2014-06-26 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US10144755B2 (en) | 2012-12-21 | 2018-12-04 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| AU2020200499B2 (en) * | 2012-12-21 | 2021-05-06 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| EP3421482A1 (en) * | 2012-12-21 | 2019-01-02 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US9243022B2 (en) | 2012-12-21 | 2016-01-26 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| US10039779B2 (en) | 2013-01-31 | 2018-08-07 | Gilead Pharmasset Llc | Combination formulation of two antiviral compounds |
| WO2014137926A1 (en) * | 2013-03-04 | 2014-09-12 | Idenix Pharmaceuticals, Inc. | 3'-deoxy nucleosides for the treatment of hcv |
| AU2014225052B2 (en) * | 2013-03-08 | 2016-11-10 | Nanjing Sanhome Pharmaceutical Co., Ltd. | Novel nucleoside phosphoramidate compound and use thereof |
| EP2940031A4 (en) * | 2013-03-08 | 2016-01-27 | Nanjing Sanhome Pharmaceutical Co Ltd | Novel nucleoside phosphoramidate compound and use thereof |
| KR101857337B1 (en) * | 2013-03-08 | 2018-05-11 | 난징 산홈 팔마세우티칼 컴퍼니 리미티드 | Novel nucleoside phosphoramidate compound and use thereof |
| US9963480B2 (en) | 2013-03-08 | 2018-05-08 | Nanjing Sanhome Pharmaceutical Co., Ltd. | Nucleoside phosphoramidate compound and use thereof |
| JP2016510043A (en) * | 2013-03-08 | 2016-04-04 | ナンジン・サンホーム・ファーマシューティカル・カンパニー・リミテッドNanjing Sanhome Pharmaceutical Co., Ltd. | Novel nucleoside phosphoramidate compounds and uses thereof |
| WO2014135107A1 (en) | 2013-03-08 | 2014-09-12 | 南京圣和药业有限公司 | Novel nucleoside phosphoramidate compound and use thereof |
| EA025878B1 (en) * | 2013-03-22 | 2017-02-28 | Александр Васильевич Иващенко | Alkyl 2-{[(2r,3s,5r)-5-(4-amino-2-oxo-2h-pyrimidin-1-yl)-3-hydroxy-tetrahydro-furan-2-yl-methoxy]phenoxy-phosphoryl-amino}propionates, nucleoside inhibitors of hcv ns5b rna-polymerase, methods for production and use thereof |
| WO2014148949A1 (en) * | 2013-03-22 | 2014-09-25 | Асави, Ллс | Alkyl 2-{[(2r,3s,5r)-5-(4-amino-2-oxo-2н-pyrimidin-1-yl)-3-hydroxy- tetrahydro-furan-2-yl-methoxy]-phenoxy-phosphoryl-amino}-propionates, nucleoside inhibitors of hcv ns5b rna-polymerase, and methods for producing and use thereof |
| RU2534613C2 (en) * | 2013-03-22 | 2014-11-27 | Александр Васильевич Иващенко | Alkyl2-{[(2r,3s,5r)-5-(4-amino-2-oxo-2h-pyrimidine-1-yl)- -hydroxy- tetrahydro-furan-2-ylmethoxy]-phenoxy-phosphorylamino}-proptonates, nucleoside inhibitors of rna-polymerase hcv ns5b, methods for producing and using them |
| WO2014165542A1 (en) * | 2013-04-01 | 2014-10-09 | Idenix Pharmaceuticals, Inc. | 2',4'-fluoro nucleosides for the treatment of hcv |
| EP2996695A4 (en) * | 2013-05-16 | 2016-12-14 | Riboscience Llc | 4'-fluor0-2'-methyl substituted nucleoside derivatives |
| US9895442B2 (en) | 2013-05-16 | 2018-02-20 | Riboscience Llc | 4′-fluoro-2′-methyl substituted nucleoside derivatives as inhibitors of HCV RNA replication |
| AU2014265293B2 (en) * | 2013-05-16 | 2019-07-18 | Riboscience Llc | 4'-Fluoro-2'-methyl substituted nucleoside derivatives |
| WO2014186637A1 (en) | 2013-05-16 | 2014-11-20 | Riboscience Llc | 4'-fluor0-2'-methyl substituted nucleoside derivatives |
| US10953030B2 (en) | 2013-05-16 | 2021-03-23 | Riboscience Llc | 4′-fluoro-2′-methyl substituted nucleoside derivatives as inhibitors of HCV RNA replication |
| WO2014197578A1 (en) * | 2013-06-05 | 2014-12-11 | Idenix Pharmaceuticals, Inc. | 1',4'-thio nucleosides for the treatment of hcv |
| US10696708B2 (en) | 2013-06-26 | 2020-06-30 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| WO2015017713A1 (en) * | 2013-08-01 | 2015-02-05 | Idenix Pharmaceuticals, Inc. | D-amino acid phosphoramidate pronucleotides of halogeno pyrimidine compounds for liver disease |
| US11707479B2 (en) | 2013-08-27 | 2023-07-25 | Gilead Sciences, Inc. | Combination formulation of two antiviral compounds |
| US11116783B2 (en) | 2013-08-27 | 2021-09-14 | Gilead Pharmasset Llc | Combination formulation of two antiviral compounds |
| US9481703B2 (en) | 2013-09-04 | 2016-11-01 | Medivir Ab | HCV polymerase inhibitors |
| US9540411B2 (en) | 2013-09-04 | 2017-01-10 | Medivir Ab | HCV polymerase inhibitors |
| US9828408B2 (en) | 2013-09-04 | 2017-11-28 | Medivir Ab | HCV polymerase inhibitors |
| EP3252066A1 (en) | 2013-09-04 | 2017-12-06 | Medivir Ab | Hcv polymerase inhibitors |
| US10106571B2 (en) | 2013-09-04 | 2018-10-23 | Medivir Ab | HCV polymerase inhibitors |
| WO2015034420A1 (en) | 2013-09-04 | 2015-03-12 | Medivir Ab | Hcv polymerase inhibitors |
| US10370401B2 (en) | 2013-10-11 | 2019-08-06 | Janssen Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| EA027990B1 (en) * | 2013-10-11 | 2017-09-29 | Андрей Александрович ИВАЩЕНКО | Substituted (2r,3r,5r)-3-hydroxy-(5-pyrimidin-1-yl)tetrahydrofuran-2-ylmethyl aryl phosphoramidates |
| WO2015053662A1 (en) * | 2013-10-11 | 2015-04-16 | Андрей Александрович ИВАЩЕНКО | Substituted (2r, 3r, 5r)-3-hydroxy-(5-pyrimidin-1-yl) tetrahydrofuran-2-ylmethyl aryl phosphoramidates |
| US9862743B2 (en) | 2013-10-11 | 2018-01-09 | Alios Biopharma, Inc. | Substituted nucleosides, nucleotides and analogs thereof |
| WO2015056213A1 (en) | 2013-10-17 | 2015-04-23 | Medivir Ab | Hcv polymerase inhibitors |
| WO2015061683A1 (en) * | 2013-10-25 | 2015-04-30 | Idenix Pharmaceuticals, Inc. | D-amino acid phosphoramidate and d-alanine thiophosphoramidate pronucleotides of nucleoside compounds useful for the treatment of hcv |
| CN104650171A (en) * | 2013-11-25 | 2015-05-27 | 天津市汉康医药生物技术有限公司 | Sofosbuvir sesquihydrate compound |
| WO2015081297A1 (en) * | 2013-11-27 | 2015-06-04 | Idenix Pharmaceuticals, Inc. | 2'-dichloro and 2'-fluoro-2'-chloro nucleoside analogues for hcv infection |
| RU2553996C1 (en) * | 2013-11-27 | 2015-06-20 | Андрей Александрович Иващенко | Substituted (2r,3r,5r)-3-hydroxy-(5-pyrimidin-1-yl)tetrahydrofuran-2-ylmethyl aryl phosphoramidates |
| WO2015099989A1 (en) | 2013-12-23 | 2015-07-02 | Gilead Pharmasset Llc | Crystalline forms of antiviral sofosbuvir analogues |
| MD20160087A2 (en) * | 2013-12-23 | 2016-12-31 | Gilead Pharmasset Llc. | Crystalline forms of an antiviral compound |
| WO2015097605A1 (en) | 2013-12-23 | 2015-07-02 | Mylan Laboratories Ltd. | Process for the preparation of sofosbuvir |
| US10100075B2 (en) | 2013-12-23 | 2018-10-16 | Mylan Laboratories Limited | Process for the preparation of sofosbuvir |
| WO2015101183A1 (en) * | 2014-01-02 | 2015-07-09 | 江苏豪森药业股份有限公司 | Uracil nucleotide analogs, preparation methods therefor and uses thereof |
| WO2015158317A1 (en) | 2014-04-15 | 2015-10-22 | Zentiva, K.S. | Use of a l,3j5-triazin-2-yl phosphoramidate compound in the synthesis of sofosbuvir |
| WO2015164812A1 (en) * | 2014-04-24 | 2015-10-29 | Cocrystal Pharma, Inc. | 2' -disubstituted nucleoside analogs for treatment of the flaviviridae family of viruses and cancer |
| EP3134423A4 (en) * | 2014-04-24 | 2017-11-29 | Cocrystal Pharma, Inc. | 2' -disubstituted nucleoside analogs for treatment of the flaviviridae family of viruses and cancer |
| US11203599B2 (en) | 2014-06-11 | 2021-12-21 | Gilead Pharmasset Llc | Solid forms of an antiviral compound |
| US10214553B2 (en) | 2014-06-13 | 2019-02-26 | Teva Pharmaceuticals International Gmbh | Solid state forms of sofosbuvir |
| WO2016008461A1 (en) | 2014-07-17 | 2016-01-21 | Zentiva, K.S. | A new form of sofosbuvir and a method of its preparation |
| US10526363B2 (en) | 2014-08-15 | 2020-01-07 | Merck Sharp & Dohme Corp. | Substituted phosphoramidate compounds and uses thereof |
| WO2016035006A1 (en) * | 2014-09-01 | 2016-03-10 | Dr. Reddy’S Laboratories Limited | Novel nucleotide analogs, process for the preparation of sofosbuvir and its analogs, novel forms of sofosbuvir and solid dispersion of sofosbuvir |
| WO2016042576A1 (en) | 2014-09-16 | 2016-03-24 | Cadila Healthcare Limited | Co-crystal of sofosbuvir and amino acid and process for preparation thereof |
| US9949994B2 (en) | 2014-10-29 | 2018-04-24 | Gilead Sciences, Inc. | Methods for treating Filoviridae virus infections |
| US11266666B2 (en) | 2014-10-29 | 2022-03-08 | Gilead Sciences, Inc. | Methods for treating Filoviridae virus infections |
| US10251898B2 (en) | 2014-10-29 | 2019-04-09 | Gilead Sciences, Inc. | Methods for treating Filoviridae virus infections |
| US11344565B2 (en) | 2014-10-29 | 2022-05-31 | Gilead Sciences, Inc. | Methods for the preparation of ribosides |
| US9724360B2 (en) | 2014-10-29 | 2017-08-08 | Gilead Sciences, Inc. | Methods for treating Filoviridae virus infections |
| US10695357B2 (en) | 2014-10-29 | 2020-06-30 | Gilead Sciences, Inc. | Methods for treating filoviridae virus infections |
| RU2567854C1 (en) * | 2014-11-11 | 2015-11-10 | Александр Васильевич Иващенко | Nucleoside inhibitors of rna-polymerase hcv ns5b, methods for production and use thereof |
| US11447518B2 (en) | 2014-12-15 | 2022-09-20 | Emory University | Phosphoramidates for the treatment of hepatitis B virus |
| US11981699B2 (en) | 2014-12-15 | 2024-05-14 | Emory University | Phosphoramidates for the treatment of hepatitis B virus |
| WO2016128453A1 (en) | 2015-02-13 | 2016-08-18 | Sandoz Ag | Pharmaceutical compositions comprising ledipasvir and sofosbuvir |
| ITUB20150109A1 (en) * | 2015-03-05 | 2016-09-05 | Hc Pharma Ag | USEFUL COMPOUNDS FOR THE TREATMENT OF HEPATITIS C |
| RU2764767C2 (en) * | 2015-03-06 | 2022-01-21 | Атеа Фармасьютикалс, Инк. | β-D-2ʹ-DEOXY-2ʹ-α-FLUORO-2ʹ-β-C-SUBSTITUTED-2-MODIFIED-N6-SUBSTITUTED PURINE NUCLEOTIDES FOR TREATMENT OF DISEASES CAUSED BY HCV |
| WO2016151542A1 (en) * | 2015-03-26 | 2016-09-29 | Quimica Sintetica, S.A. | Nucleoside phosphoramidates useful for the treatment of viral infections and preparation thereof |
| US10407456B2 (en) | 2015-03-26 | 2019-09-10 | Quimica Sintetica, S.A. | Nucleoside phosphoramidates useful for the treatment of viral infections and preparation thereof |
| WO2016177300A1 (en) * | 2015-05-07 | 2016-11-10 | 苏州旺山旺水生物医药有限公司 | (2'r)-2'-deoxy-2'-halo-2'-methyl uridine derivative, and preparation method and use thereof |
| WO2016189443A3 (en) * | 2015-05-23 | 2017-01-12 | Virupaksha Organics Limited | Solid forms of nucleoside phosphoramidate |
| WO2016189040A1 (en) | 2015-05-26 | 2016-12-01 | Sandoz Ag | Selective process for synthesis of nucleoside phosphoramidates |
| WO2016196735A2 (en) | 2015-06-03 | 2016-12-08 | Teva Pharmaceuticals International Gmbh | Improved processes for the preparation of sofosbuvir and intermediates thereof |
| US10676498B2 (en) | 2015-06-03 | 2020-06-09 | Teva Pharmaceuticals International Gmbh | Processes for the preparation of sofosbuvir and intermediates thereof |
| WO2016206663A1 (en) | 2015-06-26 | 2016-12-29 | Zentiva, K.S. | A pharmaceutical formulation of sofosbuvir |
| ITUB20152109A1 (en) * | 2015-07-13 | 2017-01-13 | Quim Sintetica S A | PHOSPHORAMIDATE NUCLEOSIDES USEFUL FOR THE TREATMENT OF VIRAL INFECTIONS AND THEIR PREPARATION |
| US10053474B2 (en) | 2015-09-02 | 2018-08-21 | Abbvie Inc. | Anti-viral compounds |
| US9676797B2 (en) | 2015-09-02 | 2017-06-13 | Abbvie Inc. | Anti-viral compounds |
| US10695361B2 (en) | 2015-09-16 | 2020-06-30 | Gilead Sciences, Inc. | Methods for treating arenaviridae and coronaviridae virus infections |
| US11007208B2 (en) | 2015-09-16 | 2021-05-18 | Gilead Sciences, Inc. | Methods for treating arenaviridae and coronaviridae virus infections |
| US11382926B2 (en) | 2015-09-16 | 2022-07-12 | Gilead Sciences, Inc. | Methods for treating Arenaviridae and Coronaviridae virus infections |
| US10251904B2 (en) | 2015-09-16 | 2019-04-09 | Gilead Sciences, Inc. | Methods for treating arenaviridae and coronaviridae virus infections |
| WO2017077552A1 (en) | 2015-11-03 | 2017-05-11 | Mylan Laboratories Limited | Process for the preparation of sofosbuvir |
| EP3377512A4 (en) * | 2015-11-16 | 2019-09-18 | Ichorion Therapeutics, Inc. | Nucleic acid prodrugs |
| US11479576B2 (en) | 2015-11-16 | 2022-10-25 | Avalo Therapeutics, Inc. | Nucleic acid prodrugs |
| US10745435B2 (en) | 2015-11-16 | 2020-08-18 | Cerecor, Inc. | Nucleic acid prodrugs |
| WO2017162169A1 (en) * | 2016-03-25 | 2017-09-28 | 江苏天士力帝益药业有限公司 | Uridine phosphoramide prodrug, preparation method therefor, and medicinal uses thereof |
| CN109071588B (en) * | 2016-03-25 | 2021-07-06 | 江苏天士力帝益药业有限公司 | Uridine phosphamide prodrug, preparation method and application thereof in medicine |
| US10745434B2 (en) | 2016-03-25 | 2020-08-18 | Jiangsu Tasly Diyi Pharmaceutical Co., Ltd. | Uridine phosphoramide prodrug, preparation method therefor, and medicinal uses thereof |
| CN109071588A (en) * | 2016-03-25 | 2018-12-21 | 江苏天士力帝益药业有限公司 | Uridine phosphamide prodrug, preparation method and its application in medicine |
| CN107226831A (en) * | 2016-03-25 | 2017-10-03 | 江苏天士力帝益药业有限公司 | Uridine phosphamide prodrug, its preparation method and its in application pharmaceutically |
| RU2740760C2 (en) * | 2016-03-25 | 2021-01-20 | Цзянсу Тасли Дии Фармасьютикал Ко., Лтд. | Prodrug based on uridine phosphoramide, a method for production thereof and use thereof in medicine |
| WO2017184670A2 (en) | 2016-04-22 | 2017-10-26 | Gilead Sciences, Inc. | Methods for treating zika virus infections |
| US11192914B2 (en) | 2016-04-28 | 2021-12-07 | Emory University | Alkyne containing nucleotide and nucleoside therapeutic compositions and uses related thereto |
| WO2017189978A1 (en) | 2016-04-28 | 2017-11-02 | Emory University | Alkyne containing nucleotide and nucleoside therapeutic compositions and uses related thereto |
| WO2017190715A1 (en) | 2016-05-05 | 2017-11-09 | Zentiva, K.S. | An amorphous form of sofosbuvir, a method of its preparation and its stabilization |
| WO2017205078A1 (en) | 2016-05-27 | 2017-11-30 | Gilead Sciences, Inc. | Methods for treating hepatitis b virus infections using ns5a, ns5b or ns3 inhibitors |
| US10912814B2 (en) | 2016-06-02 | 2021-02-09 | Gilead Pharmasset Llc | Combination formulation of three antiviral compounds |
| US11338007B2 (en) | 2016-06-02 | 2022-05-24 | Gilead Sciences, Inc. | Combination formulation of three antiviral compounds |
| US11364257B2 (en) | 2016-06-24 | 2022-06-21 | Emory University | Phosphoramidates for the treatment of hepatitis B virus |
| US10202412B2 (en) | 2016-07-08 | 2019-02-12 | Atea Pharmaceuticals, Inc. | β-D-2′-deoxy-2′-substituted-4′-substituted-2-substituted-N6-substituted-6-aminopurinenucleotides for the treatment of paramyxovirus and orthomyxovirus infections |
| WO2018013937A1 (en) | 2016-07-14 | 2018-01-18 | Atea Pharmaceuticals, Inc. | Beta-d-2'-deoxy-2'-alpha-fluoro-2'-beta-c-substituted-4'-fluoro-n6-substituted-6-amino-2-substituted purine nucleotides for the treatment of hepatitis c virus infection |
| US10239910B2 (en) | 2016-07-20 | 2019-03-26 | Optimus Drugs (P) Limited | Process for the preparation of sofosbuvir |
| EP3331354A4 (en) * | 2016-07-28 | 2019-08-07 | Asavi, LLC | Phosphoramidate nucleoside prodrug for treating viral diseases and cancer, processes for their preparation and their use |
| WO2018033593A1 (en) | 2016-08-19 | 2018-02-22 | Sandoz Ag | Sofosbuvir derivatives for the treatment of hepatitis c |
| WO2018134343A1 (en) | 2017-01-19 | 2018-07-26 | Sandoz Ag | Synthesis of phosphoramidates |
| RU2644156C1 (en) * | 2017-02-28 | 2018-02-08 | Александр Васильевич Иващенко | Prodrug of polymerase ns5b hcv inhibitor, method for its preparation and application |
| WO2018160089A1 (en) * | 2017-02-28 | 2018-09-07 | Александр Васильевич ИВАЩЕНКО | Prodrug of hcv ns5b polymerase inhibitor and method for producing and using same |
| WO2018160088A1 (en) * | 2017-02-28 | 2018-09-07 | Александр Васильевич ИВАЩЕНКО | Nucleotides containing an n-[(s)-1-cyclobutoxycarbonyl]phosphoramidate fragment, analogs thereof, and use thereof |
| CN110382514A (en) * | 2017-02-28 | 2019-10-25 | 亚历山大·瓦西里耶维奇·伊瓦切恩科 | The prodrug of HCV NS5B polymerase inhibitors and its production and application method |
| RU2659388C1 (en) * | 2017-02-28 | 2018-07-02 | Васильевич Иващенко Александр | Nucleotides including n-[(s)-1-cyclobutoxycarbonyl]phosphoramidate fragment, their analogs and their application |
| US11260070B2 (en) | 2017-03-14 | 2022-03-01 | Gilead Sciences, Inc. | Methods of treating feline coronavirus infections |
| US10682368B2 (en) | 2017-03-14 | 2020-06-16 | Gilead Sciences, Inc. | Methods of treating feline coronavirus infections |
| US12030906B2 (en) | 2017-05-01 | 2024-07-09 | Gilead Sciences, Inc. | Crystalline forms of (s)-2-ethylbutyl 2-(((s)-(((2r,3s,4r,5r)-5-(4-aminopyrrolo[2,1-f] [1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxytetrahydrofuran-2-yl)methoxy) (phenoxy) phosphoryl)amino)propanoate |
| US10836787B2 (en) | 2017-05-01 | 2020-11-17 | Gilead Sciences, Inc. | Crystalline forms of (S)-2-ethylbutyl 2-(((S)-(((2R,3S,4R,5R)-5- (4-aminopyrrolo[2,1-f] [1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxytetrahydrofuran-2-yl)methoxy)(phenoxy) phosphoryl)amino)propanoate |
| US11597742B2 (en) | 2017-05-01 | 2023-03-07 | Gilead Sciences, Inc. | Crystalline forms of (S)-2-ethylbutyl 2-(((S)-(((2R,3S,4R,5R)-5-(4-aminopyrrolo[2,1-f] [1,2,4]triazin-7-yl)-5-cyano-3,4-dihydroxytetrahydrofuran-2-yl)methoxy) (phenoxy) phosphoryl)amino)propanoate |
| US10675296B2 (en) | 2017-07-11 | 2020-06-09 | Gilead Sciences, Inc. | Compositions comprising an RNA polymerase inhibitor and cyclodextrin for treating viral infections |
| US11975017B2 (en) | 2017-07-11 | 2024-05-07 | Gilead Sciences, Inc. | Compositions comprising an RNA polymerase inhibitor and cyclodextrin for treating viral infections |
| US11266681B2 (en) | 2017-07-11 | 2022-03-08 | Gilead Sciences, Inc. | Compositions comprising an RNA polymerase inhibitor and cyclodextrin for treating viral infections |
| WO2019025600A1 (en) | 2017-08-03 | 2019-02-07 | Sandoz Ag | Sofosbuvir hydrate |
| US11414451B2 (en) | 2017-09-18 | 2022-08-16 | NuCana plc | Floxuridine synthesis |
| US10682369B2 (en) | 2017-09-21 | 2020-06-16 | Riboscience Llc | 4′-fluoro-2′-methyl substituted nucleoside derivatives as inhibitors of HCV RNA replication |
| US11351186B2 (en) | 2017-09-21 | 2022-06-07 | Riboscience Llc | 4′-fluoro-2′-methyl substituted nucleoside derivatives as inhibitors of HCV RNA replication |
| US11660307B2 (en) | 2020-01-27 | 2023-05-30 | Gilead Sciences, Inc. | Methods for treating SARS CoV-2 infections |
| US11613553B2 (en) | 2020-03-12 | 2023-03-28 | Gilead Sciences, Inc. | Methods of preparing 1′-cyano nucleosides |
| US12012431B2 (en) | 2020-03-12 | 2024-06-18 | Gilead Sciences, Inc. | Methods of preparing 1′-cyano nucleosides |
| WO2021202669A2 (en) | 2020-04-01 | 2021-10-07 | Reyoung Corporation | Nucleoside and nucleotide conjugate compounds and uses thereof |
| US11701372B2 (en) | 2020-04-06 | 2023-07-18 | Gilead Sciences, Inc. | Inhalation formulations of 1'-cyano substituted carba-nucleoside analogs |
| US11903953B2 (en) | 2020-05-29 | 2024-02-20 | Gilead Sciences, Inc. | Remdesivir treatment methods |
| US11975012B2 (en) | 2020-05-29 | 2024-05-07 | Gilead Sciences, Inc. | Remdesivir treatment methods |
| US11491169B2 (en) | 2020-05-29 | 2022-11-08 | Gilead Sciences, Inc. | Remdesivir treatment methods |
| US11939347B2 (en) | 2020-06-24 | 2024-03-26 | Gilead Sciences, Inc. | 1′-cyano nucleoside analogs and uses thereof |
| US11926645B2 (en) | 2020-08-27 | 2024-03-12 | Gilead Sciences, Inc. | Compounds and methods for treatment of viral infections |
| US11814406B2 (en) | 2020-08-27 | 2023-11-14 | Gilead Sciences, Inc. | Compounds and methods for treatment of viral infections |
| US11845755B2 (en) | 2022-03-02 | 2023-12-19 | Gilead Sciences, Inc. | Compounds and methods for treatment of viral infections |
| US11780844B2 (en) | 2022-03-02 | 2023-10-10 | Gilead Sciences, Inc. | Compounds and methods for treatment of viral infections |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240139227A1 (en) | Nucleoside phosphoramidate prodrugs | |
| US8716263B2 (en) | Synthesis of purine nucleosides | |
| CA2727495C (en) | Nucleoside cyclicphosphates | |
| AU2009329917B2 (en) | Nucleoside analogs | |
| US20110243886A1 (en) | Compounds and pharmaceutical compositions for the treatment of viral infections | |
| JP2005533824A (en) | 2'-C-methyl-3'-OL-valine ester ribofuranosyl cytidine for the treatment of Flaviviridae infections | |
| AU2014233579B2 (en) | Nucleoside phosphoramidate prodrugs |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200880018024.2 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008232827 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 579880 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008732818 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2682230 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2009/010401 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12009501847 Country of ref document: PH |
|
| ENP | Entry into the national phase |
Ref document number: 2010502196 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2008232827 Country of ref document: AU Date of ref document: 20080326 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3658/KOLNP/2009 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 09120744 Country of ref document: CO |
|
| ENP | Entry into the national phase |
Ref document number: 20097022652 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009139968 Country of ref document: RU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 222810 Country of ref document: IL |
|
| ENP | Entry into the national phase |
Ref document number: PI0809654 Country of ref document: BR Kind code of ref document: A2 Effective date: 20090930 |