WO2004056875A1 - Antibodies against pd-1 and uses therefor - Google Patents
Antibodies against pd-1 and uses therefor Download PDFInfo
- Publication number
- WO2004056875A1 WO2004056875A1 PCT/IB2003/006304 IB0306304W WO2004056875A1 WO 2004056875 A1 WO2004056875 A1 WO 2004056875A1 IB 0306304 W IB0306304 W IB 0306304W WO 2004056875 A1 WO2004056875 A1 WO 2004056875A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- antibody
- antibodies
- binding
- cell
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2818—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against CD28 or CD152
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
Definitions
- the technical field relates to modulation of immune responses regulated by the Programmed Death 1 (PD-1 ) receptor.
- PD-1 Programmed Death 1
- T cells Two major classes of lymphocytes termed T cells and B cells. After encountering an antigen, T cells proliferate and differentiate into antigen-specific effector cells, while B cells proliferate and differentiate into antibody-secreting cells.
- T cell activation is a multi-step process requiring several signaling events between the T cell and an antigen-presenting cell (APC).
- APC antigen-presenting cell
- TcR antigen-specific T cell receptor
- the second, costimulatory, type regulates the magnitude of the response and is delivered through accessory receptors on the T cell.
- a primary costimulatory signal is delivered through the activating CD28 receptor upon engagement of its ligands B7-1 or B7-2.
- engagement of the inhibitory CTLA-4 receptor by the same B7-1 or B7-2 ligands results in attenuation of T cell response.
- CTLA-4 signals antagonize costimulation mediated by CD28.
- CD28 costimulation overrides the CTLA-4 inhibitory effect.
- Temporal regulation of the CD28 and CTLA-4 expression maintains a balance between activating and inhibitory signals and ensures the development of an effective immune response, while safeguarding against the development of autoimmunity.
- ICOS CD28-like costimulatory receptor
- PD-1 Programmed Death 1
- This disclosure relates to modulation of immune responses mediated by the PD-1 receptor.
- PD-1 is a 50-55 kDa type I transmembrane receptor that was originally identified in a T cell line undergoing activation-induced apoptosis. PD-1 is expressed on T cells, B cells, and macrophages.
- the ligands for PD-1 are the B7 family members PD-L1 (B7-H1 ) and PD-L2 (B7-DC).
- PD-1 is a member of the immunoglobulin (Ig) superfamily that contains a single Ig V-like domain in its extracellular region.
- the PD-1 cytoplasmic domain contains two tyrosines, with the most membrane-proximal tyrosine (VAYEEL in mouse PD-1) located within an ITIM (immuno-receptor tyrosine-based inhibitory motif).
- ITIM immunoglobulin
- Human and murine PD-1 proteins share about 60% amino acid identity with conservation of four potential N-glycosylation sites, and residues that define the Ig-V domain.
- the ITIM in the cytoplasmic region and the ITIM-like motif surrounding the carboxy-terminal tyrosine (TEYATI in human and mouse) are also conserved between human and murine orthologues.
- PD-1 is expressed on activated T cells, B cells, and monocytes. Experimental data implicates the interactions of PD-1 with its ligands in down regulation of central and peripheral immune responses. In particular, proliferation in wild-type T cells but not in PD-1 -deficient T cells is inhibited in the presence of PD-L1. Additionally, PD-1 -deficient mice exhibit an autoimmune phenotype. PD-1 deficiency in the C57BL/6 mice results in chronic progressive lupus-like glomerulonephritis and arthritis. In Balb/c mice, PD-1 deficiency leads to severe cardiomyopathy due to the presence of heart- tissue-specific self-reacting antibodies.
- a need exists to provide safe and effective therapeutic methods for immune disorders such as, for example, autoimmune diseases, inflammatory disorders, allergies, transplant rejection, cancer, immune deficiency, and other immune system-related disorders. Modulation of the immune responses involved in these disorders can be accomplished by manipulation of the PD-1 pathway.
- the present disclosure provides antibodies that can act as agonists and/or antagonists of PD-1 , thereby modulating immune responses regulated by PD-1.
- the disclosure further provides anti-PD-1 antibodies that comprise novel antigen-binding fragments.
- Anti-PD-1 antibodies of the invention are capable of (a) specifically binding to PD-1 , including human PD-1 ; (b) blocking PD-1 interactions with its natural ligand(s); or (c) performing both functions.
- the antibodies may possess immunomodulatory properties, i.e., they may be effective in modulating the PD-1 -associated downregulation of immune responses. Depending on the method of use and the desired effect, the antibodies may be used to either enhance or inhibit immune responses.
- Nonlimiting illustrative embodiments of the antibodies are referred to as PD1-17, PD1-28, PD1-33, PD1-35, and PD1-F2.
- Other embodiments comprise a VH and/or V domain of the Fv fragment of PD1-17, PD1-28, PD1-33, PD1-35, or PD1-F2.
- Further embodiments comprise one or more complementarity determining regions (CDRs) of any of these VH and VL domains.
- Other embodiments comprise an H3 fragment of the VH domain of PD1-17, PD1-28, PD1-33, PD1-35, or PD1-F2.
- compositions comprising PD-1 antibodies, and their use in methods of modulating immune response, including methods of treating humans or animals.
- anti-PD-1 antibodies are used to treat or prevent immune disorders by virtue of increasing or reducing the T cell response mediated by TcR/CD28.
- Disorders susceptible to treatment with compositions of the invention include but are not limited to rheumatoid arthritis, multiple sclerosis, inflammatory bowel disease, Crohn's disease, systemic lupus erythematosis, type I diabetes, transplant rejection, graft-versus-host disease, hyperproliferative immune disorders, cancer, and infectious diseases.
- anti-PD-1 antibodies may be used diagnostically to detect PD-1 or its fragments in a biological sample.
- the amount of PD-1 detected may be correlated with the expression level of PD-1 , which, in turn, is correlated with the activation status of immune cells (e.g., activated T cells, B cells, and monocytes) in the subject.
- immune cells e.g., activated T cells, B cells, and monocytes
- the disclosure also provides isolated nucleic acids, which comprise a sequence encoding a VH or VL domain from the Fv fragment of PD1-17, PD1-28, PD1-33, PD1-35, or PD1-F2. Also provided are isolated nucleic acids, which comprise a sequence encoding one or more CDRs from any of the presently disclosed VH and VL domains. The disclosure also provides vectors and host cells comprising such nucleic acids.
- the disclosure further provides a method of producing new VH and V domains and/or functional antibodies comprising all or a portion of such domains derived from the V H or VL domains of PD1-17, PD1-28, PD1-33, PD1-35, or PD1-F2.
- Figures 1 A and 1 B show reactivity of scFv antibodies with human PD-1 as determined by phage ELISA.
- Figures 2A-2C show reactivity of IgG-converted antibodies with human or mouse PD-1 as determined by ELISA.
- Figure 3 shows results of an ELISA demonstrating that selected PD-1 antibodies inhibit binding of PD-L1 to PD-1.
- Figure 4 shows results of an ELISA demonstrating that immunomodulatory PD-1 antibodies bind to distinct sites on PD-1 as determined by cross-blocking ELISA assays.
- FIG. 5 shows results of T-cell proliferation assays demonstrating that co-engagement by TcR and anti-PD-1 antibody PD1-17 or PD-L1.Fc reduces proliferation. Co-engagement by TcR and anti-PD-1 J110 has no effect on proliferation.
- Figure 6 demonstrates enhanced proliferation of primary T cells by PD1-17 in a soluble form.
- antibody refers to an immunoglobulin or a fragment or a derivative thereof, and encompasses any polypeptide comprising an antigen-binding site, regardless whether it is produced in vitro or in vivo.
- the term includes, but is not limited to, polyclonal, monoclonal, monospecific, polyspecific, non-specific, humanized, single-chain, chimeric, synthetic, recombinant, hybrid, mutated, and grafted antibodies.
- antibody also includes antibody fragments such as Fab, F(ab')2, Fv, scFv, Fd, dAb, and other antibody fragments that retain antigen-binding function, i.e., the ability to bind PD-1 specifically. Typically, such fragments would comprise an antigen-binding domain.
- antigen-binding domain refers to a part of an antibody molecule that comprises amino acids responsible for the specific binding between the antibody and the antigen. In instances, where an antigen is large, the antigen-binding domain may only bind to a part of the antigen. A portion of the antigen molecule that is responsible for specific interactions with the antigen-binding domain is referred to as "epitope” or "antigenic determinant.”
- An antigen-binding domain typically comprises an antibody light chain variable region (VL) and an antibody heavy chain variable region (VH), however, it does not necessarily have to comprise both.
- VL antibody light chain variable region
- VH antibody heavy chain variable region
- Fd antibody fragment consists only of a VH domain, but still retains some antigen-binding function of the intact antibody.
- the term "repertoire” refers to a genetically diverse collection of nucleotides derived wholly or partially from sequences that encode expressed immunoglobulins.
- the sequences are generated by in vivo rearrangement of, e.g., V, D, and J segments for H chains and, e.g., V and J segment for L chains.
- the sequences may be generated from a cell line by in vitro stimulation, in response to which the rearrangement occurs.
- part or all of the sequences may be obtained by combining, e.g., unrearranged V segments with D and J segments, by nucleotide synthesis, randomised mutagenesis, and other methods, e.g., as disclosed in U.S. Patent No. 5,565,332.
- binding refers to two molecules forming a complex that is relatively stable under physiologic conditions. Specific binding is characterized by a high affinity and a low to moderate capacity as distinguished from nonspecific binding which usually has a low affinity with a moderate to high capacity. Typically, binding is considered specific when the affinity constant KA is higher than 10 6 M “1 , or more preferably higher than 10 8 M “1 . If necessary, non-specific binding can be reduced without substantially affecting specific binding by varying the binding conditions.
- the appropriate binding conditions such as concentration of antibodies, ionic strength of the solution, temperature, time allowed for binding, concentration of a blocking agent (e.g., serum albumin, milk casein), etc., may be optimized by a skilled artisan using routine techniques. Illustrative conditions are set forth in Examples 1 , 2, 4, 6, and 7.
- substantially as set out means that the relevant CDR, VH, or V L domain of the invention will be either identical to or have only insubstantial differences in the specified regions (e.g., a CDR), the sequence of which is set out. Insubstantial differences include minor amino acid changes, such as substitutions of 1 or 2 out of any 5 amino acids in the sequence of a specified region.
- PD-1 activity refers to one or more immunoregulatory activities associated with PD-1.
- PD-1 is a negative regulator of the TcR/CD28-mediated immune response. Procedures for assessing the PD-1 activity in vivo and in vitro are described in Examples 8, 9, and 10.
- modulate refers to a reduction or an increase in the activity of PD-1 associated with downregulation of T cell responses due to its interaction with an anti-PD-1 antibody, wherein the reduction or increase is relative to the activity of PD-1 in the absence of the same antibody.
- a reduction or an increase in activity is preferably at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the terms “modulatory” and “modulate” are interchangeable with the terms “inhibitory” and “inhibit.”
- modulatory and modulate are interchangeable with the terms “activating” and “activate.”
- the activity of PD-1 can be determined quantitatively using T cell proliferation assays as described in Examples 8 and 9.
- treatment and “therapeutic method” refer to both therapeutic treatment and prophylactic/preventative measures.
- Those in need of treatment may include individuals already having a particular medical disorder as well as those who may ultimately acquire the disorder (i.e., those needing preventative measures).
- the term "effective amount” refers to a dosage or amount that is sufficient to reduce the activity of PD-1 to result in amelioration of symptoms in a patient or to achieve a desired biological outcome, e.g., increased cytolytic activity of T cells, induction of immune tolerance, reduction or increase of the PD-1 activity associated with the negative regulation of T-cell mediated immune response, etc.
- isolated refers to a molecule that is substantially free of its natural environment.
- an isolated protein is substantially free of cellular material or other proteins from the cell or tissue source from which it is derived.
- isolated also refers to preparations where the isolated protein is sufficiently pure to be administered as a pharmaceutical composition, or at least 70-80% (w/w) pure, more preferably, at least 80-90% (w/w) pure, even more preferably, 90-95% pure; and, most preferably, at least 95%, 96%, 97%, 98%, 99%, or 100% (w/w) pure.
- the disclosure provides anti-PD-1 antibodies that comprise novel antigen-binding fragments.
- antibodies can be made, for example, using traditional hybridoma techniques (Kohler and Milstein (1975) Nature, 256: 495-499), recombinant DNA methods (U.S. Patent No. 4,816,567), or phage display performed with antibody libraries (Clackson et al. (1991 ) Nature, 352: 624-628; Marks et al. (1991 ) J. Mol. Biol., 222: 581-597).
- Antibodies A Laboratory Manual, eds. Harlow et al., Cold Spring Harbor Laboratory, 1988.
- the invention is not limited to any particular source, species of origin, method of production.
- Intact antibodies also known as immunoglobulins, are typically tetrameric glycosylated proteins composed of two light (L) chains of approximately 25 kDa each and two heavy (H) chains of approximately 50 kDa each. Two types of light chain, designated as the ⁇ chain and the K chain, are found in antibodies.
- immunoglobulins can be assigned to five major classes: A, D, E, G, and M, and several of these may be further divided into subclasses (isotypes), e.g., IgG-i, lgG 2 , lgG 3 , lgG 4 , IgA-i, and lgA 2 .
- each light chain is composed of an N-terminal variable domain (VL) and a constant domain (CL).
- Each heavy chain is composed of an N-terminal variable domain (VH), three or four constant domains (CH), and a hinge region.
- the CH domain most proximal to VH is designated as CH1.
- the VH and VL domains consist of four regions of relatively conserved sequence called framework regions (FR1 , FR2, FR3, and FR4), which form a scaffold for three regions of hypervariable sequence called complementarity determining regions (CDRs).
- the CDRs contain most of the residues responsible for specific interactions with the antigen.
- the three CDRs are referred to as CDR1 , CDR2, and CDR3.
- CDR constituents on the heavy chain are referred to as H1 , H2, and H3, while CDR constituents on the light chain are referred to as L1 , L2, and L3, accordingly.
- CDR3 and, particularly H3, are the greatest source of molecular diversity within the antigen-binding domain.
- H3, for example, can be as short as two amino acid residues or greater than 26.
- the Fab fragment (Fragment antigen-binding) consists of the VH-CH1 and VL-CL domains covalently linked by a disulfide bond between the constant regions.
- a so-called single chain (sc) Fv fragment (scFv) can be constructed.
- a flexible and adequately long polypeptide links either the C-terminus of the VH to the N-terminus of the VL or the C-terminus of the VL to the N-terminus of the V H .
- Antibody diversity is a result of combinatorial assembly of multiple germline genes encoding variable regions and a variety of somatic events.
- the somatic events include recombination of variable gene segments with diversity (D) and joining (J) gene segments to make a complete VH region and the recombination of variable and joining gene segments to make a complete VL region.
- D diversity
- J joining
- the recombination process itself is imprecise, resulting in the loss or addition of amino acids at the V(D)J junctions.
- the disclosure provides novel CDRs derived from human immunoglobulin gene libraries.
- the structure for carrying a CDR will generally be an antibody heavy or light chain or a portion thereof, in which the CDR is located at a location corresponding to the CDR of naturally occurring VH and VL.
- the structures and locations of immunoglobulin variable domains may be determined, for example, as described in Kabat et al., Sequences of Proteins of Immunological Interest, No. 91-3242, National Institutes of Health Publications, Bethesda, MD, 1991.
- Anti-PD-1 antibodies may optionally comprise antibody constant regions or parts thereof.
- a VL domain may have attached, at its C terminus, antibody light chain constant domains including human CK or C ⁇ chains.
- a specific antigen-binding domain based on a VH domain may have attached all or part of an immunoglobulin heavy chain derived from any antibody isotope, e.g., IgG, IgA, IgE, and IgM and any of the isotope sub-classes, which include but are not limited to, IgG-i and lgG 4 .
- antibodies comprise C-terminal fragments of heavy and light chains of human IgG
- PD1-F2 comprises C-terminal fragments of heavy and light chains of human lgG-
- the DNA and amino acid sequences for the C-terminal fragment of are well known in the art (see, e.g, Kabat et al., Sequences of Proteins of Immunological Interest, No. 91-3242, National Institutes of Health Publications, Bethesda, MD, 1991). Nonlimiting exemplary sequences are set forth in Table 4.
- Certain embodiments comprise a VH and/or VL domain of an Fv fragment from PD1-17, PD1-28, PD1-33, PD1-35, and PD1-F2. Further embodiments comprise at least one CDR of any of these VH and VL domains.
- Antibodies comprising at least one of the CDR sequences set out in SEQ ID NO:2, SEQ ID NO:4, SEQ ID NO:6, SEQ ID NO:8, SEQ ID NO:10, SEQ ID NO:12, SEQ ID NO.14, SEQ ID NOs:16-40, SEQ ID NO:47, or SEQ ID NO:49 are encompassed within the scope of this invention.
- An embodiment for example, comprises an H3 fragment of the VH domain of antibodies chosen from at least one of PD1-17, PD1-28, PD1-33, PD1-35, and PD1-F2.
- the VH and/or VL domains may be germlined, i.e., the framework regions (FRs) of these domains are mutated using conventional molecular biology techniques to match those produced by the germline cells.
- the framework sequences remain diverged from the consensus germline sequences.
- the antibodies specifically bind an epitope within the extracellular domain of human PD-1.
- the predicted extracellular domain consists of a sequence from about amino acid 21 to about amino acid 170 of SEQ ID NO:41 (Swissport Accession No. Q15116).
- the antibodies specifically bind an epitope within the extracellular domain of mouse PD-1 , with an affinity of more than 10 7 M "1 , and preferably more than 10 8 M "1 .
- the amino acid sequence of mouse PD-1 is set out in SEQ ID NO:56 (Accession No. NM_008798) and is as a whole about 60% identical to its human counterpart.
- antibodies of the invention bind to the PD-L-binding domain of PD-1.
- antibodies of the invention may also bind with other proteins, including, for example, recombinant proteins comprising all or a portion of the PD-1 extracellular domain.
- the antibodies of this invention may be used to detect, measure, and inhibit proteins that differ somewhat from PD-1.
- the antibodies are expected to retain the specificity of binding so long as the target protein comprises a sequence which is at least about 60%, 70%, 80%, 90%, 95%, or more identical to any sequence of at least 100, 80, 60, 40, or 20 of contiguous amino acids in the sequence set forth SEQ ID NO:41.
- the percent identity is determined by standard alignment algorithms such as, for example, Basic Local Alignment Tool (BLAST) described in Altshul et al. (1990) J. Mol. Biol., 215: 403-410, the algorithm of Needleman et al. (1970) J. Mol. Biol., 48: 444-453, or the algorithm of Meyers et al. (1988) Comput. Appl. Biosci., 4: 11-17.
- BLAST Basic Local Alignment Tool
- epitope mapping see, e.g., Epitope Mapping Protocols, ed. Morris, Humana Press, 1996) and secondary and tertiary structure analyses can be carried out to identify specific 3D structures assumed by the disclosed antibodies and their complexes with antigens.
- Such methods include, but are not limited to, X-ray crystallography (Engstom (1974) Biochem. Exp. Biol., 11 :7-13) and computer modeling of virtual representations of the presently disclosed antibodies (Fletterick et al. (1986) Computer Graphics and Molecular Modeling, in Current Communications in Molecular Biology, Cold Spring Harbor Laboratory, Cold Spring Harbor, NY). Derivatives
- This disclosure also provides a method for obtaining an antibody specific for PD-1.
- CDRs in such antibodies are not limited to the specific sequences of VH and VL identified in Table 1 and may include variants of these sequences that retain the ability to specifically bind PD-1. Such variants may be derived from the sequences listed in Table 1 by a skilled artisan using techniques well known in the art. For example, amino acid substitutions, deletions, or additions, can be made in the FRs and/or in the CDRs. While changes in the FRs are usually designed to improve stability and immunogenicity of the antibody, changes in the CDRs are typically designed to increase affinity of the antibody for its target. Variants of FRs also include naturally occurring immunoglobulin allotypes.
- Such affinity-increasing changes may be determined empirically by routine techniques that involve altering the CDR and testing the affinity antibody for its target. For example, conservative amino acid substitutions can made within any one of the disclosed CDRs. Various alterations can be made according to the methods described in Antibody Engineering, 2 nd ed., Oxford University Press, ed. Borrebaeck, 1995. These include but are not limited to nucleotide sequences that are altered by the substitution of different codons that encode a functionally equivalent amino acid residue within the sequence, thus producing a "silent" change.
- the nonpolar amino acids include alanine, leucine, isoleucine, valine, proline, phenylalanine, tryptophan, and methionine.
- the polar neutral amino acids include glycine, serine, threonine, cysteine, tyrosine, asparagine, and glutamine.
- the positively charged (basic) amino acids include arginine, lysine, and histidine.
- the negatively charged (acidic) amino acids include aspartic acid and glutamic acid. Substitutes for an amino acid within the sequence may be selected from other members of the class to which the amino acid belongs (see Table 5). Furthermore, any native residue in the polypeptide may also be substituted with alanine (see, e.g., MacLennan et al. (1998) Acta Physiol. Scand. Suppl. 643:55-67; Sasaki et al. (1998) Adv. Biophys. 35:1-24).
- a method for making a VH domain which is an amino acid sequence variant of a VH domain of the invention comprises a step of adding, deleting, substituting, or inserting one or more amino acids in the amino acid sequence of the presently disclosed VH domain, optionally combining the VH domain thus provided with one or more V domains, and testing the VH domain or VH/VL combination or combinations for a specific binding to PD-1 or and, optionally, testing the ability of such antigen-binding domain to modulate PD-1 activity.
- the VL domain may have an amino acid sequence that is identical or is substantially as set out according to Table 1.
- An analogous method can be employed in which one or more sequence variants of a VL domain disclosed herein are combined with one or more VH domains.
- a further aspect of the disclosure provides a method of preparing antigen-binding fragment that specifically binds with PD-1.
- the method comprises:
- VL CDR3 i.e., L3
- the donor nucleic acid may be selected from nucleic acids encoding an amino acid sequence substantially as set out in SEQ ID NO:17-40 or SEQ ID NO:50-55.
- a sequence encoding a CDR of the invention may be introduced into a repertoire of variable domains lacking the respective CDR (e.g., CDR3), using recombinant DNA technology, for example, using methodology described by Marks et al. (Bio/Technology (1992) 10: 779-783).
- consensus primers directed at or adjacent to the 5' end of the variable domain area can be used in conjunction with consensus primers to the third framework region of human VH genes to provide a repertoire of VH variable domains lacking a CDR3.
- the repertoire may be combined with a CDR3 of a particular antibody.
- the CDR3-derived sequences may be shuffled with repertoires of VH or VL domains lacking a CDR3, and the shuffled complete VH or VL domains combined with a cognate VL or VH domain to make the PD-1 -specific antibodies of the invention.
- the repertoire may then be displayed in a suitable host system such as the phage display system such as described in WO92/01047 so that suitable antigen-binding fragments can be selected.
- One such technique, error-prone PCR is described by Gram et al. (Proc. Nat. Acad. Sci. U.S.A. (1992) 89: 3576-3580).
- Another method that may be used is to direct mutagenesis to CDRs of VH or VL genes.
- Such techniques are disclosed by Barbas et al. (Proc. Nat. Acad. Sci. U.S.A. (1994) 91: 3809-3813) and Schier et al. (J. Mol. Biol. (1996) 263: 551-567).
- one or more, or all three CDRs may be grafted into a repertoire of VH or VL domains, which are then screened for an antigen-binding fragment specific for PD-1.
- a portion of an immunoglobulin variable domain will comprise at least one of the CDRs substantially as set out herein and, optionally, intervening framework regions from the scFv fragments as set out herein.
- the portion may include at least about 50% of either or both of FR1 and FR4, the 50% being the C-terminal 50% of FR1 and the N-terminal 50% of FR4. Additional residues at the N-terminal or C-terminal end of the substantial part of the variable domain may be those not normally associated with naturally occurring variable domain regions.
- construction of antibodies by recombinant DNA techniques may result in the introduction of N- or C-terminal residues encoded by linkers introduced to facilitate cloning or other manipulation steps.
- Other manipulation steps include the introduction of linkers to join variable domains to further protein sequences including immunoglobulin heavy chain constant regions, other variable domains (for example, in the production of diabodies), or proteinaceous labels as discussed in further detail below.
- the screening may be accomplished by phage display screening methods using the so-called hierarchical dual combinatorial approach disclosed in WO92/01047, in which an individual colony containing either an H or L chain clone is used to infect a complete library of clones encoding the other chain (L or H) and the resulting two-chain specific binding domain is selected in accordance with phage display techniques as described.
- Anti-PD1 antibodies described herein can be linked to another functional molecule, e.g., another peptide or protein (albumin, another antibody, etc.), toxin, radioisotope, cytotoxic or cytostatic agents.
- the antibodies can be linked by chemical cross-linking or by recombinant methods.
- the antibodies may also be linked to one of a variety of nonproteinaceous polymers, e.g., polyethylene glycol, polypropylene glycol, or polyoxyalkylenes, in the manner set forth in U.S. Patent Nos. 4,640,835; 4,496,689; 4,301 ,144; 4,670,417; 4,791,192; or 4,179,337.
- the antibodies can be chemically modified by covalent conjugation to a polymer, for example, to increase their circulating half-life.
- exemplary polymers and methods to attach them are also shown in U.S. Patent Nos. 4,766,106; 4,179,337; 4,495,285; and 4,609,546.
- the disclosed antibodies may also be altered to have a glycosylation pattern that differs from the native pattern.
- one or more carbohydrate moieties can be deleted and/or one or more glycosylation sites added to the original antibody.
- Addition of glycosylation sites to the presently disclosed antibodies may be accomplished by altering the amino acid sequence to contain glycosylation site consensus sequences known in the art.
- Another means of increasing the number of carbohydrate moieties on the antibodies is by chemical or enzymatic coupling of glycosides to the amino acid residues of the antibody. Such methods are described in WO 87/05330 and in Aplin et al. (1981 ) CRC Crit. Rev. Biochem., 22: 259-306.
- the antibodies may also be tagged with a detectable, or functional, label.
- Detectable labels include radiolabels such as 31 l or 99 Tc, which may also be attached to antibodies using conventional chemistry.
- Detectable labels also include enzyme labels such as horseradish peroxidase or alkaline phosphatase.
- Detectable labels further include chemical moieties such as biotin, which may be detected via binding to a specific cognate detectable moiety, e.g., labeled avidin.
- Antibodies in which CDR sequences differ only insubstantially from those set out in SEQ ID NO:2, SEQ ID NO:4, SEQ ID NO:6, SEQ ID NO:8, SEQ ID NO:10, SEQ ID NO:12, SEQ ID NO:14, SEQ ID NOs:16-40, SEQ ID NO:47, or SEQ ID NO:49 are encompassed within the scope of this invention.
- an amino acid is substituted by a related amino acid having similar charge, hydrophobic, or stereochemical characteristics. Such substitutions would be within the ordinary skills of an artisan.
- more substantial changes can be made in FRs without adversely affecting the binding properties of an antibody.
- Changes to FRs include, but are not limited to, humanizing a non-human derived or engineering certain framework residues that are important for antigen contact or for stabilizing the binding site, e.g., changing the class or subclass of the constant region, changing specific amino acid residues which might alter the effector function such as Fc receptor binding, e.g., as described in U.S. Patent Nos. 5,624,821 and 5,648,260 and Lund et al. (1991) J. Immun. 147: 2657-2662 and Morgan et al. (1995) Immunology 86: 319-324, or changing the species from which the constant region is derived.
- the present disclosure further provides isolated nucleic acids encoding the disclosed antibodies.
- the nucleic acids may comprise DNA or RNA and may be wholly or partially synthetic or recombinant.
- Reference to a nucleotide sequence as set out herein encompasses a DNA molecule with the specified sequence, and encompasses a RNA molecule with the specified sequence in which U is substituted for T, unless context requires otherwise.
- nucleic acids provided herein comprise a coding sequence for a CDR, a VH domain, and/or a VL domain disclosed herein.
- the present disclosure also provides constructs in the form of plasmids, vectors, phagemids, transcription or expression cassettes which comprise at least one nucleic acid encoding a CDR, a VH domain, and/or a VL domain disclosed here.
- the disclosure further provides a host cell which comprises one or more constructs as above.
- nucleic acids encoding any CDR (H1 , H2, H3, L1 , L2, or L3), VH or V domain, as well as methods of making of the encoded products.
- the method comprises expressing the encoded product from the encoding nucleic acid. Expression may be achieved by culturing under appropriate conditions recombinant host cells containing the nucleic acid. Following production by expression a V H or VL domain, or specific binding member may be isolated and/or purified using any suitable technique, then used as appropriate.
- Antigen-binding fragments, VH and/or VL domains, and encoding nucleic acid molecules and vectors may be isolated and/or purified from their natural environment, in substantially pure or homogeneous form, or, in the case of nucleic acid, free or substantially free of nucleic acid or genes of origin other than the sequence encoding a polypeptide with the required function.
- Suitable host cells include bacteria, plant cells, mammalian cells, and yeast and baculovirus systems.
- Mammalian cell lines available in the art for expression of a heterologous polypeptide include Chinese hamster ovary cells, HeLa cells, baby hamster kidney cells, NS0 mouse myeloma cells, and many others.
- a common bacterial host is E. coli. Any protein expression system compatible with the invention may be used to produce the disclosed antibodies. Suitable expression systems include transgenic animals described in Gene Expression Systems, Academic Press, eds. Fernandez et al., 1999.
- Suitable vectors can be chosen or constructed, so that they contain appropriate regulatory sequences, including promoter sequences, terminator sequences, polyadenylation sequences, enhancer sequences, marker genes and other sequences as appropriate.
- Vectors may be plasmids or viral, e.g., phage, or phagemid, as appropriate.
- phage e.g., phagemid
- a further aspect of the disclosure provides a host cell comprising a nucleic acid as disclosed here.
- a still further aspect provides a method comprising introducing such nucleic acid into a host cell.
- the introduction may employ any available technique.
- suitable techniques may include calcium phosphate transfection, DEAE-Dextran, electroporation, liposome-mediated transfection and transduction using retrovirus or other virus, e.g., vaccinia or, for insect cells, baculovirus.
- suitable techniques may include calcium chloride transformation, electroporation and transfection using bacteriophage.
- the introduction of the nucleic acid into the cells may be followed by causing or allowing expression from the nucleic acid, e.g., by culturing host cells under conditions for expression of the gene.
- the disclosed anti-PD-1 antibodies are capable of modulating the PD-1 -associated downregulation of the immune responses.
- the immune response is TcR/CD28-mediated.
- the disclosed antibodies can act as either agonists or antagonists of PD-1 , depending on the method of their use.
- the antibodies can be used to prevent, diagnose, or treat medical disorders in mammals, especially, in humans.
- Antibodies of the invention can also be used for isolating PD-1 or PD-1 -expressing cells.
- the antibodies can be used to treat a subject at risk of or susceptible to a disorder or having a disorder associated with aberrant PD-1 expression or function.
- Antibodies of the invention can be used in methods for induction of tolerance to a specific antigen (e.g., a therapeutic protein).
- a specific antigen e.g., a therapeutic protein
- tolerance is induced against a specific antigen by co- administration of antigen and an anti-PD-1 antibody of the invention.
- Antibodies of the invention can be used in circumstances where a reduction in the level of immune response may be desirable, for example, in certain types of allergy or allergic reactions (e.g., by inhibition of IgE production), autoimmune diseases (e.g., rheumatoid arthritis, type I diabetes mellitus, multiple sclerosis, inflammatory bowel disease, Crohn's disease, and systemic lupus erythematosis), tissue, skin and organ transplant rejection, and graft-versus-host disease (GVHD).
- autoimmune diseases e.g., rheumatoid arthritis, type I diabetes mellitus, multiple sclerosis, inflammatory bowel disease, Crohn's disease, and systemic lupus erythematosis
- tissue skin and organ transplant rejection
- graft-versus-host disease graft-versus-host disease
- the anti-PD-1 antibodies of the invention may be used as agonists to PD-1 in order to enhance the PD-1 -associated attenuation of the immune response.
- co-presentation and physical proximity between positive (i.e., mediated by an antigen receptor, e.g., TcR or BcR) and negative (i.e., PD-1) signals are required.
- the preferred distance is less than or comparable to the size of a naturally occurring antigen-presenting cell, i.e., less than about 100 ⁇ m; more preferably, less than about 50 ⁇ m; and most preferably, less than about 20 ⁇ m.
- the positive (activating) and the negative (inhibiting) signals are provided by a ligand or antibodies immobilized on solid support matrix, or a carrier.
- the solid support matrix may be composed of polymer such as activated agarose, dextran, cellulose, polyvinylidene fluoride (PVDF).
- the solid support matrix may be based on silica or plastic polymers, e.g., as nylon, dacron, polystyrene, polyacrylates, polyvinyls, teflons, etc.
- the matrix can be implanted into the spleen of a patient.
- the matrix may be used for the ex vivo incubation of T cells obtained from a patient, which are then separated and implanted back into the patient.
- the matrix may also be made from a biodegradable material such polyglycolic acid, polyhydroxyalkanoate, collagen, or gelatin so that they can be injected into the patient's peritoneal cavity, and dissolve after some time following the injection.
- the carrier can be shaped to mimic a cell (e.g., bead or microsphere).
- the positive signal is delivered by a T-cell-activating anti-CD3 antibody, which binds TcR.
- Activating anti-CD3 antibodies are known in the art (see, for example, U.S. Patent Nos. 6,405,696 and 5,316,763).
- the ratio between the activating TcR signal and negative PD-1 signal is determined experimentally using conventional procedures known in the art or as described in Examples 8, 9, and 10.
- disorders being treated or prevented by the disclosed methods include but are not limited to infections with microbes (e.g. bacteria), viruses (e.g., systemic viral infections such as influenza, viral skin diseases such as herpes or shingles), or parasites; and cancer (e.g., melanoma and prostate cancers).
- microbes e.g. bacteria
- viruses e.g., systemic viral infections such as influenza, viral skin diseases such as herpes or shingles
- parasites e.g., melanoma and prostate cancers.
- the antibodies act as antagonists of PD-1.
- the antibodies can be used to inhibit or reduce the downregulatory activity associated with PD-1 , i.e., the activity associated with downregulation of TcR/CD28-mediated immune response.
- the antibodies are not coupled to a positive signal such as the TcR-mediated stimulation, e.g., the antibodies are in their soluble, support-unbound, form.
- the antibodies inhibit binding of PD-L to PD-1 with an IC 50 of less than 10 nM, and more preferably less then 5 nM, and most preferably less than 1 nM. Inhibition of PD-L binding can be measured as described in Example 6 or using techniques known in the art.
- the antibodies or antibody compositions of the present invention are administered in therapeutically effective amounts.
- a therapeutically effective amount may vary with the subject's age, condition, and sex, as well as the severity of the medical condition of the subject.
- a therapeutically effective amount of antibody ranges from about 0.001 to about 30 mg/kg body weight, preferably from about 0.01 to about 25 mg/kg body weight, from about 0.1 to about 20 mg/kg body weight, or from about 1 to about 10 mg/kg.
- the dosage may be adjusted, as necessary, to suit observed effects of the treatment. The appropriate dose is chosen based on clinical indications by a treating physician.
- the antibodies may given as a bolus dose, to maximize the circulating levels of antibodies for the greatest length of time after the dose. Continuous infusion may also be used after the bolus dose.
- Immune cells e.g., activated T cells, B cells, or monocytes
- immune responses can be inhibited by removing immune cells from a subject, contacting the immune cells in vitro with an anti-PD-1 antibody of the invention concomitantly with activation of the immune cells (e.g., by antibodies to the TcR and/or BcR antigen receptor).
- the anti-PD-1 antibody should be used in a multivalent form such that PD-1 molecules on the surface of an immune cell become "crosslinked" upon binding to such antibodies.
- the anti-PD-1 antibodies can be bound to solid support, such as beads, or crosslinked via a secondary antibody.
- the immune cells may be then isolated using methods known in the art and reimplanted into the patient.
- the antibodies of the invention can be used as a targeting agent for delivery of another therapeutic or a cytotoxic agent (e.g., a toxin) to a cell expressing PD-1.
- the method includes administering an anti-PD-1 antibody coupled to a therapeutic or a cytotoxic agent or under conditions that allow binding of the antibody to PD-1.
- the antibodies of the invention may also be used to detect the presence of PD-1 in biological samples. The amount of PD-1 detected may be correlated with the expression level of PD-1 , which, in turn, is correlated with the activation status of immune cells (e.g., activated T cells, B cells, and monocytes) in the subject.
- immune cells e.g., activated T cells, B cells, and monocytes
- Detection methods that employ antibodies are well known in the art and include, for example, ELISA, radioimmunoassay, immunoblot, Western blot, immunofluorescence, immunoprecipitation.
- the antibodies may be provided in a diagnostic kit that incorporates one or more of these techniques to detect PD-1.
- a kit may contain other components, packaging, instructions, or other material to aid the detection of the protein.
- the antibodies are intended for diagnostic purposes, it may be desirable to modify them, for example, with a ligand group (such as biotin) or a detectable marker group (such as a fluorescent group, a radioisotope or an enzyme).
- a detectable label such as a fluorescent group, a radioisotope or an enzyme.
- the antibodies of the invention may be labeled using conventional techniques. Suitable detectable labels include, for example, fluorophores, chromophores, radioactive atoms, electron-dense reagents, enzymes, and ligands having specific binding partners. Enzymes are typically detected by their activity. For example, horseradish peroxidase can be detected by its ability to convert tetramethylbenzidine (TMB) to a blue pigment, quantifiable with a spectrophotometer.
- TMB tetramethylbenzidine
- binding partners include, but are not limited to, biotin and avidin or streptavidin, IgG and protein A, and the numerous receptor-ligand couples known in the art. Other permutations and possibilities will be readily apparent to those of ordinary skill in the art, and are considered as equivalents within the scope of the instant invention.
- Antibodies of the invention can be used in screening methods to identify inhibitors of the PD-1 pathway effective as therapeutics.
- a first binding mixture is formed by combining PD-1 and an antibody of the invention; and the amount of binding in the first binding mixture (Mo) is measured.
- a second binding mixture is also formed by combining PD-1 , the antibody, and the compound or agent to be screened, and the amount of binding in the second binding mixture (M-i) is measured.
- a compound to be tested may be another anti-PD-1 antibody, as illustrated in the Examples.
- the amounts of binding in the first and second binding mixtures are then compared, for example, by calculating the M- ⁇ /M 0 ratio.
- the compound or agent is considered to be capable of modulating a PD-1- associated downregulation of immune responses if a decrease in binding in the second binding mixture as compared to the first binding mixture is observed.
- the formulation and optimization of binding mixtures is within the level of skill in the art, such binding mixtures may also contain buffers and salts necessary to enhance or to optimize binding, and additional control assays may be included in the screening assay of the invention.
- Compounds found to reduce the PD-1 -antibody binding by at least about 10% (i.e., M ⁇ /Mo ⁇ 0.9), preferably greater than about 30% may thus be identified and then, if desired, secondarily screened for the capacity to ameliorate a disorder in other assays or animal models as described below.
- the strength of the binding between PD-1 and an antibody can be measured using, for example, an enzyme-linked immunoadsorption assay (ELISA), radio-immunoassay (RIA), surface plasmon resonance-based technology (e.g., Biacore), all of which are techniques well known in the art.
- ELISA enzyme-linked immunoadsorption assay
- RIA radio-immunoassay
- Biacore surface plasmon resonance-based technology
- the compound may then be tested in vitro as described in the Examples or in an animal model (see, generally, Immunologic Defects in Laboratory Animals, eds. Gershwin et al., Plenum Press, 1981), for example, such as the following: the SWR X NZB (SNF1) transgenic mouse model (Uner et al. (1998) J. Autoimmune. 11(3): 233-240), the KRN transgenic mouse (K/BxN) model (Ji et al. (1999) Immunol. Rev. 169: 139); NZB X NZW (B/W) mice, a model for SLE (Riemekasten et al.
- SNF1 SWR X NZB
- K/BxN KRN transgenic mouse
- B/W NZB X NZW mice
- Preliminary doses as, for example, determined according to animal tests, and the scaling of dosages for human administration is performed according to art-accepted practices. Toxicity and therapeutic efficacy can be determined by standard pharmaceutical procedures in cell cultures or experimental animals. The data obtained from the cell culture assays or animal studies can be used in formulating a range of dosage for use in humans. Therapeutically effective dosages achieved in one animal model can be converted for use in another animal, including humans, using conversion factors known in the art (see, e.g., Freireich et al. (1966) Cancer Chemother. Reports, 50(4): 219-244). Pharmaceutical Compositions and Methods of Administration
- compositions comprising anti-PD-1 antibodies. Such compositions may be suitable for pharmaceutical use and administration to patients.
- the compositions typically comprise one or more antibodies of the present invention and a pharmaceutically acceptable excipient.
- pharmaceutically acceptable excipient includes any and all solvents, dispersion media, coatings, antibacterial agents and antifungal agents, isotonic agents, and absorption delaying agents, and the like, that are compatible with pharmaceutical administration. The use of such media and agents for pharmaceutically active substances is well known in the art.
- the compositions may also contain other active compounds providing supplemental, additional, or enhanced therapeutic functions.
- the pharmaceutical compositions may also be included in a container, pack, or dispenser together with instructions for administration.
- a pharmaceutical composition of the invention is formulated to be compatible with its intended route of administration. Methods to accomplish the administration are known to those of ordinary skill in the art.
- the administration may, for example, be intravenous, intraperitoneal, intramuscular, intracavity, subcutaneous or transdermal. It may also be 2004/056875
- compositions which may be topically or orally administered, or which may be capable of transmission across mucous membranes.
- Solutions or suspensions used for intradermal or subcutaneous application typically include one or more of the following components: a sterile diluent such as water for injection, saline solution, fixed oils, polyethylene glycols, glycerin, propylene glycol, or other synthetic solvents; antibacterial agents such as benzyl alcohol or methyl parabens; antioxidants such as ascorbic acid or sodium bisulfite; chelating agents such as ethylenediaminetetraacetic acid; buffers such as acetates, citrates or phosphates; and agents for the adjustment of tonicity such as sodium chloride or dextrose.
- the pH can be adjusted with acids or bases, such as hydrochloric acid or sodium hydroxide.
- Such preparations may be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic.
- compositions suitable for injection include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion.
- suitable carriers include physiological saline, bacteriostatic water, Cremophor EL (BASF, Parsippany, NJ) or phosphate buffered saline (PBS).
- the composition must be sterile and should be fluid to the extent that easy syringability exists. It should be stable under the conditions of manufacture and storage and must be preserved against the contaminating action of microorganisms such as bacteria and fungi.
- the carrier can be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyetheylene glycol, and the like), and suitable mixtures thereof.
- the proper fluidity can be maintained, for 2004/056875
- Prolonged absorption of the injectable compositions can be brought about by including in the composition an agent which delays absorption, for example, aluminum monostearate, and gelatin.
- compositions generally include an inert diluent or an edible carrier. They can be enclosed in gelatin capsules or compressed into tablets. For oral administration, the antibodies can be combined with excipients and used in the form of tablets, troches, or capsules. Pharmaceutically compatible binding agents, and/or adjuvant materials can be included as part of the composition.
- the tablets, pills, capsules, troches, and the like can contain any of the following ingredients, or compounds of a similar nature; a binder such as microcrystalline cellulose, gum tragacanth or gelatin; an excipient such as starch or lactose, a disintegrating agent such as alginic acid, Primogel, or corn starch; a lubricant such as magnesium stearate or Sterotes; a glidant such as colloidal silicon dioxide; a sweetening agent such as sucrose or saccharin; or a flavoring agent such as peppermint, methyl salicylate, or orange flavoring.
- a binder such as microcrystalline cellulose, gum tragacanth or gelatin
- an excipient such as starch or lactose, a disintegrating agent such as alginic acid, Primogel, or corn starch
- a lubricant such as magnesium stearate or Sterotes
- a glidant such as colloidal silicon dioxide
- Systemic administration can also be by transmucosal or transdermal means.
- penetrants appropriate to the barrier to be permeated are used in the formulation.
- penetrants are generally known in the art, and include, for example, detergents, bile salts, and fusidic acid derivatives.
- Transmucosal administration may be accomplished, for example, through the use of lozenges, nasal sprays, inhalers, or suppositories.
- compositions may be capable of transmission across mucous membranes in intestine, mouth, or lungs (e.g., via the FcRn receptor-mediated pathway as described in U.S. Patent No.
- the active compounds may be formulated into ointments, salves, gels, or creams as generally known in the art.
- the antibodies may be delivered in the form of an aerosol spray from pressured container or dispenser, which contains a suitable propellant, e.g., a gas such as carbon dioxide, or a nebulizer.
- the presently disclosed antibodies are prepared with carriers that will protect the compound against rapid elimination from the body, such as a controlled release formulation, including implants and microencapsulated delivery systems.
- a controlled release formulation including implants and microencapsulated delivery systems.
- Biodegradable, biocompatible polymers can be used, such as ethylene vinyl acetate, polyanhydrides, polyglycolic acid, collagen, polyorthoesters, and polylactic acid. Methods for preparation of such formulations will be apparent to those skilled in the art.
- Liposomal suspensions containing the presently disclosed antibodies can also be used as pharmaceutically acceptable carriers. These can be prepared according to methods known to those skilled in the art, for example, as described in U.S. Patent No. 4,522,811.
- dosage unit form refers to physically discrete units suited as unitary dosages for the subject to be treated; each unit containing a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier.
- Toxicity and therapeutic efficacy of the composition of the invention can be determined by standard pharmaceutical procedures in cell cultures or experimental animals, e.g., for determining the LD 50 (the dose lethal to 50% of the population) and the ED 50 (the dose therapeutically effective in 50% of the population).
- the dose ratio between toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio LD 50 /ED 50 .
- Compositions that exhibit large therapeutic indices are preferred.
- the therapeutically effective dose can be estimated initially from cell culture assays.
- suitable bioassays include DNA replication assays, cytokine release assays, transcription-based assays, PD-1/PD-L1 binding assays, creatine kinase assays, assays based on the differentiation of pre-adipocytes, assays based on glucose uptake in adipocytes, immunological assays other assays as, for example, described in the Examples.
- the data obtained from the cell culture assays and animal studies can be used in formulating a range of dosage for use in humans.
- a dose may be formulated in animal models to achieve a circulating plasma concentration range that includes the IC 50 (i.e., the concentration of the antibody which achieves a half-maximal inhibition of symptoms). Circulating levels in plasma may be measured, for example, by high performance liquid chromatography. The effects of any particular dosage can be monitored by a suitable bioassay. The dosage lies preferably within a range of circulating concentrations with little or no toxicity. The dosage may vary depending upon the dosage form employed and the route of administration utilized.
- Blocked phage was added to blocked control fusion protein wells and incubated for 1 hour. The blocked and deselected phage were then transferred to the blocked wells coated with the PD-1 fusion protein and were incubated for an additional hour. Wells were washed 5 times with PBST (PBS containing 0.1% v/v Tween 20), then 5 times with PBS. Bound phage particles were eluted and used to infect 10 ml exponentially growing E. coli TG1. Infected cells were grown in 2TY broth for 1 hour at 37°C, then spread onto 2TYAG plates and incubated overnight at 30°C. Colonies were scraped off the plates into 10 ml 2TY broth and 15% glycerol added for storage at - 70°C.
- PBST PBS containing 0.1% v/v Tween 20
- Glycerol stock cultures from the first round of panning selection were superinfected with helper phage and rescued to give scFv antibody-expressing phage particles for the second round of panning.
- a total of two rounds of panning were carried out in this way for isolation of PD1-17, except in the second round of panning 20 ⁇ g/ml of control protein were used for deselection.
- Clones PD1-28, PD1-33, and PD1-35 were selected following three rounds of selection. Deselection in the second and third rounds was carried out using 10 ⁇ g/ml control fusion protein.
- Antibodies to murine PD-1 were selected by soluble selection using biotinylated murine PD-1 fusion protein at a final concentration of 100 nM.
- Infected cells were incubated for 1 hour at 37°C (30 minutes stationary, 30 minutes shaking at 250 rpm), then spread on 2TYAG plates and incubated overnight at 30°C. Output colonies were scraped off the plates and phage rescued as described above. A second round of soluble selection was carried out as described above.
- a phage ELISA was performed against PD-1 fusion protein and control proteins. Individual E. coli colonies from selection outputs were picked into 96 well plates containing 100 ⁇ l of 2TYAG medium per well. M13K07 helper phage was added to a multiplicity of infection (moi) of 10 to the exponentially growing culture and the plates incubated an additional 1 hour at 37°C. Plates were centrifuged in a benchtop centrifuge at 2000 rpm for 10 minutes. The supernatant was removed and cell pellets were resuspended in 100 ⁇ l 2TYAK and incubated at 30°C overnight with shaking. The next day, plates were centrifuged at 2000 rpm for 10 minutes and phage-containing supernatant from each well was transferred to a fresh 96 well plate. Phage samples were blocked in a final concentration of 3% MPBS prior to ELISA.
- Human or mouse PD-1 fusion protein and control fusion and non-fusion proteins were coated overnight at 4°C onto 96-well mierotiter plates at 0.5-2.5 ⁇ g/ml in PBS. After coating, the solutions were removed from the wells, and the plates blocked for 1 hour in 3% MPBS. Plates were rinsed with PBS and then 50 ⁇ l of pre-blocked phage were added to each well. The plates were incubated for 1 hour and then washed 3 times with PBST followed by 3 washes with PBS.
- PD-1 -binding scFv E. coli clones were streaked out onto 2TYAG plates and incubated overnight at 30°C. Colonies from these plates were sequenced using pCANTAB ⁇ vector sequence oligos to amplify the VH and VL regions from the scFv clone. Unique PD-1 binding clones were assayed for neutralization of PD-L1 binding to PD-1 as described in Example 4. Sequence differences between scFv and IgG formats are due to changes introduced by PCR primers during the conversion from scFv to IgG.
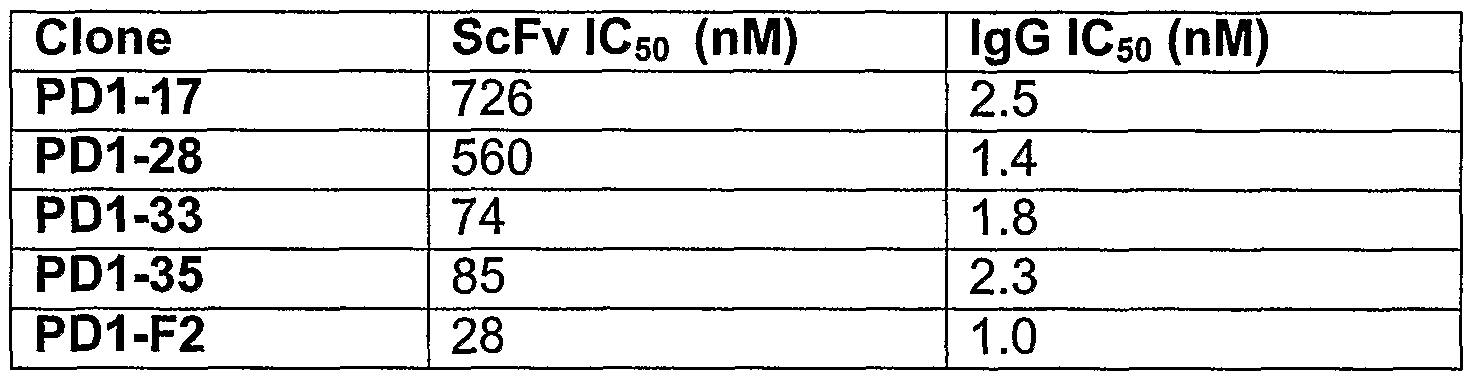
- scFv's (PD1-17, PD1-28, PD1-33, and PD1-35) were tested for the ability to inhibit the binding of biotinylated human PD-L1 fusion protein to human PD-1 fusion protein immobilized on plastic in a 96 well mierotiter plate assay. Binding of biotinylated PD-L1 fusion protein was detected with AMDEX-alkaline phosphatase, and the signal generated was measured by reading the absorbance at 405 nm using a mierotiter plate reader. Data was expressed as a percentage of the total binding and a titration of scFv concentrations was tested to establish clone potency as calculated IC 50 values. Clone potency data for the scFv and IgG antibodies is shown in Table 5.
- PD1-F2 scFv was produced and purified as described above.
- Cells expressing murine PD-1 were added at 10 cells/well in a final volume of 100 ⁇ l to a poly-D-lysine-coated 96 well mierotiter plate. Cells were centrifuged and washed twice in PBS, then blocked with 300 ⁇ l 1% BSA in PBS for 1 hour at room temperature. Blocked cells were washed three times in PBST, prior to addition of 25 ⁇ l/well of assay buffer (0.05% BSA, 0.05% Tween 20 in Dulbecco's PBS) or sample, followed by 25 ⁇ l of biotinylated murine PD-L1 fusion protein at 300 ng/ml. Binding of biotinylated PD-L1 fusion protein was detected with Amdex alkaline phosphatase and signals read as described above. Potencies of PD1-F2 scFv and IgG are shown in Table 6.
- Heavy and light chain V regions from scFv clones were amplified by PCR using clone-specific primers. PCR products were digested with appropriate restriction enzymes and subcloned into vectors containing human IgGi heavy chain constant domain (Takahashi et al. (1982) Cell 29, 671 ) or vectors containing human lambda or kappa light chain constant domains (Hieter et al. (1982) Nature 294, 536). Based on the germlines of the VH and VL segments, it was determined whether kappa or lambda light chain constant domains were used for conversion (Table 7).
- Plasmids were prepared from E. coli cultures by standard techniques and heavy and light chain constructs cotransfected into eukaryotic cells using standard techniques. Secreted IgG was purified using Protein A Sepharose (Pharmacia) and buffer-exchanged into PBS.
- the binding affinity of the anti-mouse PD1 antibody PD1 -F2 was determined with a Surface Plasmon Resonance (SPR) system (BIAcore 3000) (Biacore, Piscataway, NJ) using murine PD-1 fusion immobilized on a CM5 sensor chip.
- SPR Surface Plasmon Resonance
- the concentration of PD1-F2 in the flow cell ranged from 7.81 to 125 nM, while the concentration of the anti-mouse PD1 antibody J43 (eBioscience, San Diego, CA) ranged from 25 nM to 500 nM.
- Example 6 Selected PD-1 Antibodies Inhibit Binding of PD-L1 to PD-1
- Inhibition assays were performed to assess the ability of the antibodies to block binding of PD-L1 to PD-1.
- ELISA was performed as described in Example 2 with modifications. After incubation with a primary, anti-PD-1 antibody for 2 hours at RT, a fixed concentration (1 ⁇ g/ml) of biotin-conjugated PD-L1-lg was added, and the samples were further incubated for 1 hour at RT. After washing, saturating concentrations of avidin-HRP were added, and incubated for 1 hour at RT. Unbound avidin-HRP was washed using PBS/1% BSA. The assay was developed using TMB.
- Results were compared to those obtained with J110 as shown in Figure 3.
- Anti-human PD-1 antibodies J110 and PD1-30 did not inhibit the binding of PD-L1 to PD-1.
- Anti-human antibodies PD1-17, PD1-28, PD1-33, and PD1-35 and anti-mouse antibody PD1-F2 block PD-1/PD-L1 interaction.
- Inhibition assays were performed to map sites recognize by the various human anti-human PD-1 antibodies.
- ELISA was performed as described in Example 6 with minor modifications. After incubation with primary antibody for 2 hours at RT, a fixed concentration (0.25 ⁇ g/ml) of biotin-conjugated anti-PD-1 antibody J110 was added, and the samples were further incubated for 1 hour at RT. After washing, saturating concentrations of avidin-HRP were added, and incubated for 1 hour at RT. Unbound avidin-HRP was washed using PBS/1% BSA. The assay was developed using TMB.
- binding of anti-human PD-1 antibodies defines at least two distinct sites on PD-1.
- Cross-blocking results show that J110 and J116, bind to identical or overlapping sites while PD1-17, 28, 33, and 35 bind to another distinct site.
- Binding of J116 or J110 to PD-1 blocks the binding of J110.
- binding of PD1-17, PD1-28, PD1-33, and PD1-35 do not block binding of J110. This suggests that the tested anti-PD-1 antibodies bind to at least two distinct epitopes: one recognized by J110 and J116, and the other one recognized by PD1-17, PD1-28, PD1-33, and PD1-35.
- CD4+ T cells (5 x 10 4 cells/well) were stimulated with tosyl-beads (Dynal, Great Neck, NY) coated with anti-hCD3 +/- PD-L1-Fc or anti-PD-1 (PD1-17 or J110). Concentration of fusion protein or antibody titer was as indicated in the X-axis of Figure 5. After 72 hours, proliferation was determined by 3 H-thymidine incorporation. Incorporated radioactivity was determined using a LKB 1205 plate reader.
- PD-1 engagement by anti-PD-1 antibody PD1-17 or PD-L1.Fc caused a decrease in T cell proliferation.
- PD1-17 can mimic PD-1 ligands and delivered an inhibitory signal.
- this inhibitory signal results in decreased T cell proliferation and IL-2 production.
- Antibodies PD1- 28, PD1-33, and PD1-35 have the same effect as PD1-17. The effect is dose-dependent, as activation of cells in the presence of increasing concentrations of PD1-17 or PD-L1.Fc results in decreased T cell proliferation.
- control anti-PD-1 antibodies J110 ( Figure 5) or J116 (data not shown), do not inhibit T cell responses and increasing the concentration of J110 has minimal effect on T cell proliferation.
- values are represented as percentage of the anti-CD3 response. "100%” represents CPMs obtained when cells were activated with anti-CD3/murine IgG-coated microspheres.
- CD4+ T cells were pre-activated for 48 hours with anti-CD3/anti-CD28-coated beads, harvested, and restimulated with the indicated concentration of PHA plus 10 ng/ml IL-2 in the presence of PD1-17, J110, or control IgG. Each of the antibodies was added at various concentrations at initiation of the culture. Proliferation was measured at 72 hr.
- Modulation of immune response regulated by PD-1 is useful in instances where an immunosuppressive effect or augmentation of immune response is desired.
- This example describes the use of PD-1 antibodies as PD-1 agonists or antagonists to treat a subject at disease onset or having an established immune disorder or cancer, respectively.
- Subjects at risk for or afflicted with cancer may be in need of immune response augmentation would benefit from treatment with a PD-1 antagonist, such as an anti-PD-1 antibody of the present invention in a soluble form.
- a PD-1 antagonist such as an anti-PD-1 antibody of the present invention in a soluble form.
- antibodies are administered in an outpatient setting by weekly administration at about 0.1-10 mg/kg dose by slow intravenous (IV) infusion.
- the appropriate therapeutically effective dose of an antagonist is selected by a treating clinician and would range approximately from 1 ⁇ g/kg to 20 mg/kg, from 1 ⁇ g/kg to 10 mg/kg, from 1 ⁇ g/kg to 1 mg/kg, from 10 ⁇ g/kg to 1 mg/kg, from 10 ⁇ g/kg to 100 ⁇ g/kg, from 100 ⁇ g to 1 mg/kg, and from 500 ⁇ g/kg to 5 mg/kg.
- the antibodies are also used to prevent and/or to reduce severity and/or symptoms of diseases or conditions that involve an aberrant or undesirable immune response, such as in autoimmune disorders exemplified below.
- MS Multiple sclerosis
- EAE experimental autoimmune encephalitis
- SLE Systemic Lupus Erythematosis
- NZB X NZW mice a mouse model for SLE
- SLE Immunologic Defects in Laboratory Animals, Gershwin et al. eds., Plenum Press, 1981
- PD-1 antibodies of the invention would be administered as PD-1 agonists in ex vivo therapy with a frequency of one per month or less. Treatment duration could range between one month and several years.
- Treatment groups include a placebo group and one to three groups treated with a PD-1 agonist (different doses). Individuals are followed prospectively for one to three years. It is anticipated that individuals receiving treatment would exhibit an improvement.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Diabetes (AREA)
- Rheumatology (AREA)
- Obesity (AREA)
- Communicable Diseases (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Pain & Pain Management (AREA)
- Hematology (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Oncology (AREA)
- Pulmonology (AREA)
- Transplantation (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Physical Education & Sports Medicine (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
Claims
Priority Applications (14)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004561922A JP4511943B2 (en) | 2002-12-23 | 2003-12-22 | Antibody against PD-1 and use thereof |
| CA2508660A CA2508660C (en) | 2002-12-23 | 2003-12-22 | Antibodies against pd-1 and uses therefor |
| CN2003801099298A CN1753912B (en) | 2002-12-23 | 2003-12-22 | Antibodies against PD-1 and uses therefor |
| US10/540,084 US7521051B2 (en) | 2002-12-23 | 2003-12-22 | Methods of upmodulating adaptive immune response using anti-PD-1 antibodies |
| AT03780521T ATE514713T1 (en) | 2002-12-23 | 2003-12-22 | ANTIBODIES TO PD-1 AND THEIR USE |
| BR0316880-8A BR0316880A (en) | 2002-12-23 | 2003-12-22 | Pd-1 Antibodies and Uses |
| AU2003288675A AU2003288675B2 (en) | 2002-12-23 | 2003-12-22 | Antibodies against PD-1 and uses therefor |
| MXPA05006828A MXPA05006828A (en) | 2002-12-23 | 2003-12-22 | Antibodies against pd-1 and uses therefor. |
| EP03780521A EP1576014B1 (en) | 2002-12-23 | 2003-12-22 | Antibodies against pd-1 and uses thereof |
| IL169152A IL169152A (en) | 2002-12-23 | 2005-06-14 | Isolated antibody which binds to an epitope within the extracellular domain of pd - 1 |
| NO20053389A NO336442B1 (en) | 2002-12-23 | 2005-07-12 | Antibodies against PD-1 and their applications. |
| HK06103433.3A HK1083510A1 (en) | 2002-12-23 | 2006-03-17 | Antibodies against pd-1 and uses thereof |
| US12/405,058 US8088905B2 (en) | 2002-12-23 | 2009-03-16 | Nucleic acids encoding antibodies against PD-1 |
| AU2010235966A AU2010235966A1 (en) | 2002-12-23 | 2010-10-22 | Antibodies Against PD-1 and Uses Therefor |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US43535402P | 2002-12-23 | 2002-12-23 | |
| US60/435,354 | 2002-12-23 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/540,084 A-371-Of-International US7521051B2 (en) | 2002-12-23 | 2003-12-22 | Methods of upmodulating adaptive immune response using anti-PD-1 antibodies |
| US12/405,058 Continuation US8088905B2 (en) | 2002-12-23 | 2009-03-16 | Nucleic acids encoding antibodies against PD-1 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004056875A1 true WO2004056875A1 (en) | 2004-07-08 |
Family
ID=32682224
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2003/006304 WO2004056875A1 (en) | 2002-12-23 | 2003-12-22 | Antibodies against pd-1 and uses therefor |
Country Status (14)
| Country | Link |
|---|---|
| US (4) | US7488802B2 (en) |
| EP (1) | EP1576014B1 (en) |
| JP (2) | JP4511943B2 (en) |
| CN (2) | CN1753912B (en) |
| AT (1) | ATE514713T1 (en) |
| AU (2) | AU2003288675B2 (en) |
| BR (1) | BR0316880A (en) |
| CA (1) | CA2508660C (en) |
| ES (1) | ES2367430T3 (en) |
| HK (1) | HK1083510A1 (en) |
| IL (1) | IL169152A (en) |
| MX (1) | MXPA05006828A (en) |
| NO (1) | NO336442B1 (en) |
| WO (1) | WO2004056875A1 (en) |
Cited By (356)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006133396A2 (en) * | 2005-06-08 | 2006-12-14 | Dana-Farber Cancer Institute | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (pd-1) pathway |
| WO2007100098A1 (en) | 2006-03-03 | 2007-09-07 | Kyoto University | Multimer of extracellular domain of cell surface functional molecule |
| US7488802B2 (en) | 2002-12-23 | 2009-02-10 | Wyeth | Antibodies against PD-1 |
| WO2009023566A2 (en) * | 2007-08-09 | 2009-02-19 | Genzyme Corporation | Method of treating autoimmune disease with mesenchymal stem cells |
| JP2009155338A (en) * | 2005-05-09 | 2009-07-16 | Ono Pharmaceut Co Ltd | Cancer therapeutic method singly using human monoclonal antibody to programmed death 1 (pd-1) and anti-pd-1 antibody or concurrently using with other immunotherapy |
| WO2010027423A2 (en) | 2008-08-25 | 2010-03-11 | Amplimmune, Inc. | Compositions of pd-1 antagonists and methods of use |
| WO2010029435A1 (en) * | 2008-09-12 | 2010-03-18 | Isis Innovation Limited | Pd-1 specific antibodies and uses thereof |
| WO2010029434A1 (en) * | 2008-09-12 | 2010-03-18 | Isis Innovation Limited | Pd-1 specific antibodies and uses thereof |
| EP2243493A1 (en) * | 2002-07-03 | 2010-10-27 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| EP2262837A2 (en) * | 2008-03-12 | 2010-12-22 | Merck Sharp & Dohme Corp. | Pd-1 binding proteins |
| WO2011110604A1 (en) | 2010-03-11 | 2011-09-15 | Ucb Pharma, S.A. | Pd-1 antibody |
| WO2011110621A1 (en) | 2010-03-11 | 2011-09-15 | Ucb Pharma, S.A. | Biological products: humanised agonistic anti-pd-1 antibodies |
| WO2011161699A2 (en) | 2010-06-25 | 2011-12-29 | Aurigene Discovery Technologies Limited | Immunosuppression modulating compounds |
| WO2012145493A1 (en) | 2011-04-20 | 2012-10-26 | Amplimmune, Inc. | Antibodies and other molecules that bind b7-h1 and pd-1 |
| WO2012168944A1 (en) | 2011-06-08 | 2012-12-13 | Aurigene Discovery Technologies Limited | Therapeutic compounds for immunomodulation |
| US8354509B2 (en) | 2007-06-18 | 2013-01-15 | Msd Oss B.V. | Antibodies to human programmed death receptor PD-1 |
| WO2013022091A1 (en) | 2011-08-11 | 2013-02-14 | 小野薬品工業株式会社 | Therapeutic agent for autoimmune diseases comprising pd-1 agonist |
| WO2013132317A1 (en) | 2012-03-07 | 2013-09-12 | Aurigene Discovery Technologies Limited | Peptidomimetic compounds as immunomodulators |
| WO2013144704A1 (en) | 2012-03-29 | 2013-10-03 | Aurigene Discovery Technologies Limited | Immunomodulating cyclic compounds from the bc loop of human pd1 |
| WO2013173223A1 (en) * | 2012-05-15 | 2013-11-21 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting pd-1/pd-l1 signaling |
| AU2012204032B2 (en) * | 2005-06-08 | 2014-01-16 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| US8747833B2 (en) | 2004-10-06 | 2014-06-10 | Mayo Foundation For Medical Education And Research | B7-H1 and methods of diagnosis, prognosis, and treatment of cancer |
| WO2014122271A1 (en) | 2013-02-07 | 2014-08-14 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for predicting the survival time of patients suffering from diffuse large b-cell lymphomas |
| WO2014194293A1 (en) | 2013-05-30 | 2014-12-04 | Amplimmune, Inc. | Improved methods for the selection of patients for pd-1 or b7-h4 targeted therapies, and combination therapies thereof |
| WO2014179664A3 (en) * | 2013-05-02 | 2015-02-19 | Anaptysbio, Inc. | Antibodies directed against programmed death-1 (pd-1) |
| WO2014194302A3 (en) * | 2013-05-31 | 2015-02-26 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind pd-1 |
| WO2015033303A1 (en) | 2013-09-06 | 2015-03-12 | Aurigene Discovery Technologies Limited | Cyclic peptidomimetic compounds as immunomodulators |
| WO2015033301A1 (en) | 2013-09-06 | 2015-03-12 | Aurigene Discovery Technologies Limited | 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives as immunomodulators |
| WO2015033299A1 (en) | 2013-09-06 | 2015-03-12 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| WO2015036394A1 (en) * | 2013-09-10 | 2015-03-19 | Medimmune Limited | Antibodies against pd-1 and uses thereof |
| WO2015112900A1 (en) * | 2014-01-24 | 2015-07-30 | Dana-Farber Cancer Institue, Inc. | Antibody molecules to pd-1 and uses thereof |
| WO2015112800A1 (en) * | 2014-01-23 | 2015-07-30 | Regeneron Pharmaceuticals, Inc. | Human antibodies to pd-1 |
| WO2015119944A1 (en) | 2014-02-04 | 2015-08-13 | Incyte Corporation | Combination of a pd-1 antagonist and an ido1 inhibitor for treating cancer |
| WO2015181624A2 (en) | 2014-05-28 | 2015-12-03 | Idenix Pharmaceuticals, Inc | Nucleoside derivatives for the treatment of cancer |
| US9220776B2 (en) | 2011-03-31 | 2015-12-29 | Merck Sharp & Dohme Corp. | Stable formulations of antibodies to human programmed death receptor PD-1 and related treatments |
| US9226958B2 (en) | 2010-10-01 | 2016-01-05 | University Of Georgia Research Foundation, Inc. | Use of Listeria vaccine vectors to reverse vaccine unresponsiveness in parasitically infected individuals |
| WO2016004876A1 (en) | 2014-07-09 | 2016-01-14 | Shanghai Birdie Biotech, Inc. | Anti-pd-l1 combinations for treating tumors |
| US20160075782A1 (en) | 2005-07-01 | 2016-03-17 | E.R. Squibb & Sons, L. L. C. | Human monoclonal antibodies to programmed death ligand 1 (pd-l1) |
| WO2016020856A3 (en) * | 2014-08-05 | 2016-03-31 | MabQuest SA | Immunological reagents binding to pd-1 |
| WO2016057898A1 (en) | 2014-10-10 | 2016-04-14 | Idera Pharmaceuticals, Inc. | Treatment of cancer using tlr9 agonist with checkpoint inhibitors |
| WO2016068801A1 (en) * | 2014-10-27 | 2016-05-06 | Agency For Science, Technology And Research | Anti-pd-1 antibodies |
| WO2016089830A1 (en) | 2014-12-05 | 2016-06-09 | Merck Sharp & Dohme Corp. | Novel tricyclic compounds as inhibitors of mutant idh enzymes |
| WO2016089833A1 (en) | 2014-12-05 | 2016-06-09 | Merck Sharp & Dohme Corp. | Novel tricyclic compounds as inhibitors of mutant idh enzymes |
| WO2016089797A1 (en) | 2014-12-05 | 2016-06-09 | Merck Sharp & Dohme Corp. | Novel tricyclic compounds as inhibitors of mutant idh enzymes |
| WO2016077397A3 (en) * | 2014-11-11 | 2016-09-01 | Sutro Biopharma, Inc. | Anti-pd-1 antibodies, compositions comprising anti-pd-1 antibodies and methods of using anti-pd-1 antibodies |
| WO2016141209A1 (en) | 2015-03-04 | 2016-09-09 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and eribulin for treating cancer |
| WO2016140717A1 (en) | 2015-03-04 | 2016-09-09 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and a vegfr/fgfr/ret tyrosine kinase inhibitor for treating cancer |
| WO2016142833A1 (en) | 2015-03-10 | 2016-09-15 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole and thiadiazole compounds as immunomodulators |
| US9463227B2 (en) | 2011-03-11 | 2016-10-11 | Advaxis, Inc. | Listeria-based adjuvants |
| WO2016189055A1 (en) | 2015-05-27 | 2016-12-01 | Idenix Pharmaceuticals Llc | Nucleotides for the treatment of cancer |
| WO2016196173A1 (en) | 2015-05-29 | 2016-12-08 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and cpg-c type oligonucleotide for treating cancer |
| WO2016196218A1 (en) | 2015-05-31 | 2016-12-08 | Curegenix Corporation | Combination compositions for immunotherapy |
| US9533042B2 (en) | 2007-06-04 | 2017-01-03 | Genentech, Inc. | Anti-notch NRR antibodies and methods using same |
| WO2017007700A1 (en) | 2015-07-06 | 2017-01-12 | Iomet Pharma Ltd. | Pharmaceutical compound |
| WO2017009842A2 (en) | 2015-07-16 | 2017-01-19 | Biokine Therapeutics Ltd. | Compositions and methods for treating cancer |
| WO2017016497A1 (en) * | 2015-07-28 | 2017-02-02 | Harbour Biomed Limited | Anti-pd-1 antibodies and uses thereof |
| EP3130923A1 (en) * | 2008-11-14 | 2017-02-15 | The Brigham and Women's Hospital, Inc. | Therapeutic and diagnostic methods relating to cancer stem cells |
| WO2017027645A1 (en) | 2015-08-13 | 2017-02-16 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| WO2017025016A1 (en) * | 2015-08-10 | 2017-02-16 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibodies |
| US9598491B2 (en) | 2008-11-28 | 2017-03-21 | Emory University | Methods for the treatment of infections and tumors |
| US9603800B2 (en) | 2012-04-12 | 2017-03-28 | Yale University | Methods of treating inflammatory and autoimmune diseases and disorders using nanolipogels |
| WO2017055404A1 (en) | 2015-10-02 | 2017-04-06 | F. Hoffmann-La Roche Ag | Bispecific antibodies specific for pd1 and tim3 |
| WO2017055484A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for determining the metabolic status of lymphomas |
| WO2017071625A1 (en) * | 2015-10-30 | 2017-05-04 | 中山康方生物医药有限公司 | Anti-pd-1 monoclonal antibody, and pharmaceutical composition and use thereof |
| US9644212B2 (en) | 2008-05-19 | 2017-05-09 | Advaxis, Inc. | Dual delivery system for heterologous antigens |
| WO2017079116A2 (en) | 2015-11-03 | 2017-05-11 | Janssen Biotech, Inc. | Antibodies specifically binding pd-1 and tim-3 and their uses |
| US9650639B2 (en) | 2008-05-19 | 2017-05-16 | Advaxis, Inc. | Dual delivery system for heterologous antigens |
| WO2017093933A1 (en) | 2015-12-03 | 2017-06-08 | Glaxosmithkline Intellectual Property Development Limited | Cyclic purine dinucleotides as modulators of sting |
| WO2017098421A1 (en) | 2015-12-08 | 2017-06-15 | Glaxosmithkline Intellectual Property Development Limited | Benzothiadiazine compounds |
| WO2017106061A1 (en) | 2015-12-14 | 2017-06-22 | Macrogenics, Inc. | Bispecific molecules having immunoreactivity with pd-1 and ctla-4, and methods of use thereof |
| WO2017106062A1 (en) | 2015-12-15 | 2017-06-22 | Merck Sharp & Dohme Corp. | Novel compounds as indoleamine 2,3-dioxygenase inhibitors |
| WO2017118634A1 (en) | 2016-01-04 | 2017-07-13 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of pd-1 and tim-3 as a measure for cd8+ cells in predicting and treating renal cell carcinoma |
| EP3192811A1 (en) * | 2009-02-09 | 2017-07-19 | Université d'Aix-Marseille | Pd-1 antibodies and pd-l1 antibodies and uses thereof |
| WO2017155981A1 (en) | 2016-03-07 | 2017-09-14 | Massachusetts Institute Of Technology | Protein-chaperoned t-cell vaccines |
| WO2017153952A1 (en) | 2016-03-10 | 2017-09-14 | Glaxosmithkline Intellectual Property Development Limited | 5-sulfamoyl-2-hydroxybenzamide derivatives |
| WO2017160754A1 (en) | 2016-03-15 | 2017-09-21 | Mersana Therapeutics,Inc. | Napi2b-targeted antibody-drug conjugates and methods of use thereof |
| WO2017160599A1 (en) | 2016-03-14 | 2017-09-21 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Use of cd300b antagonists to treat sepsis and septic shock |
| WO2017160975A1 (en) * | 2016-03-16 | 2017-09-21 | Bristol-Myers Squibb Company | Methods of diagnosing and treating lupus |
| US9783578B2 (en) | 2010-06-25 | 2017-10-10 | Aurigene Discovery Technologies Limited | Immunosuppression modulating compounds |
| WO2017175156A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| WO2017175147A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| WO2017191545A1 (en) | 2016-05-05 | 2017-11-09 | Glaxosmithkline Intellectual Property (No.2) Limited | Enhancer of zeste homolog 2 inhibitors |
| EP3243832A1 (en) | 2016-05-13 | 2017-11-15 | F. Hoffmann-La Roche AG | Antigen binding molecules comprising a tnf family ligand trimer and pd1 binding moiety |
| WO2017212425A1 (en) | 2016-06-08 | 2017-12-14 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds as atf4 pathway inhibitors |
| WO2017212423A1 (en) | 2016-06-08 | 2017-12-14 | Glaxosmithkline Intellectual Property Development Limited | Chemcical compounds |
| WO2017220989A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Anti-pd-l1 and il-2 cytokines |
| US9855342B2 (en) | 2007-04-12 | 2018-01-02 | The Brigham And Women's Hospital, Inc. | Targeting ABCB5 for cancer therapy |
| WO2018015879A1 (en) | 2016-07-20 | 2018-01-25 | Glaxosmithkline Intellectual Property Development Limited | Isoquinoline derivatives as perk inhibitors |
| US9884026B2 (en) | 2013-11-01 | 2018-02-06 | Yale University | Modular particles for immunotherapy |
| WO2018027039A1 (en) | 2016-08-03 | 2018-02-08 | Nextcure, Inc. | Compositions and methods for modulating lair signal transduction |
| WO2018028383A1 (en) | 2016-08-09 | 2018-02-15 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibody formulation |
| US9907849B2 (en) | 2014-07-18 | 2018-03-06 | Advaxis, Inc. | Combination of a PD-1 antagonist and a listeria-based vaccine for treating prostate cancer |
| US9914783B1 (en) | 2016-09-14 | 2018-03-13 | Abbvie Biotherapeutics Inc. | Anti-PD-1 antibodies and their uses |
| US9920123B2 (en) | 2008-12-09 | 2018-03-20 | Genentech, Inc. | Anti-PD-L1 antibodies, compositions and articles of manufacture |
| WO2018053434A1 (en) | 2016-09-16 | 2018-03-22 | The Johns Hopkins University | Protein nanocages with enhanced mucus penetration for targeted tissue and intracellular delivery |
| US9938345B2 (en) | 2014-01-23 | 2018-04-10 | Regeneron Pharmaceuticals, Inc. | Human antibodies to PD-L1 |
| WO2018067423A1 (en) | 2016-10-04 | 2018-04-12 | Merck Sharp & Dohme Corp. | BENZO[b]THIOPHENE COMPOUNDS AS STING AGONISTS |
| WO2018071792A1 (en) | 2016-10-14 | 2018-04-19 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and eribulin for treating urothelial cancer |
| WO2018091542A1 (en) | 2016-11-21 | 2018-05-24 | Idenix Pharmaceuticals Llc | Cyclic phosphate substituted nucleoside derivatives for the treatment of liver diseases |
| WO2018098269A2 (en) | 2016-11-23 | 2018-05-31 | Mersana Therapeutics, Inc. | Peptide-containing linkers for antibody-drug conjugates |
| US9988452B2 (en) | 2014-10-14 | 2018-06-05 | Novartis Ag | Antibody molecules to PD-L1 and uses thereof |
| WO2018100534A1 (en) | 2016-12-01 | 2018-06-07 | Glaxosmithkline Intellectual Property Development Limited | Combination therapy |
| WO2018100535A1 (en) | 2016-12-01 | 2018-06-07 | Glaxosmithkline Intellectual Property Development Limited | Combination therapy |
| US10017738B2 (en) | 2006-05-31 | 2018-07-10 | Children's Medical Center Corporation | ABCB5 positive mesenchymal stem cells as immunomodulators |
| US10016617B2 (en) | 2009-11-11 | 2018-07-10 | The Trustees Of The University Of Pennsylvania | Combination immuno therapy and radiotherapy for the treatment of Her-2-positive cancers |
| US10058599B2 (en) | 2012-03-12 | 2018-08-28 | Advaxis, Inc. | Suppressor cell function inhibition following Listeria vaccine treatment |
| WO2018154520A1 (en) | 2017-02-27 | 2018-08-30 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides as kinase inhibitors |
| WO2018156785A1 (en) * | 2017-02-22 | 2018-08-30 | Sutro Biopharma, Inc. | Pd-1/tim-3 bi-specific antibodies, compositions thereof, and methods of making and using the same |
| CN108473580A (en) * | 2015-12-23 | 2018-08-31 | 杭州尚健生物技术有限公司 | Anti-human 1 antibody of programmed death receptor and its preparation method and application |
| US10064898B2 (en) | 2011-03-11 | 2018-09-04 | Advaxis, Inc. | Listeria-based adjuvants |
| WO2018160538A1 (en) | 2017-02-28 | 2018-09-07 | Mersana Therapeutics, Inc. | Combination therapies of her2-targeted antibody-drug conjugates |
| WO2018162944A1 (en) * | 2017-03-04 | 2018-09-13 | Shenzhen Runshin Bioscience | Recombinant antibodies to programmed death 1 (pd-1) and uses therefor |
| WO2018167778A1 (en) | 2017-03-12 | 2018-09-20 | Yeda Research And Development Co. Ltd. | Methods of diagnosing and prognosing cancer |
| WO2018167780A1 (en) | 2017-03-12 | 2018-09-20 | Yeda Research And Development Co. Ltd. | Methods of prognosing and treating cancer |
| WO2018172508A1 (en) | 2017-03-24 | 2018-09-27 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2018185043A1 (en) | 2017-04-05 | 2018-10-11 | F. Hoffmann-La Roche Ag | Bispecific antibodies specifically binding to pd1 and lag3 |
| WO2018195552A1 (en) | 2017-04-21 | 2018-10-25 | Sillajen, Inc. | Oncolytic vaccinia virus and checkpoint inhibitor combination therapy |
| WO2018226336A1 (en) | 2017-06-09 | 2018-12-13 | Providence Health & Services - Oregon | Utilization of cd39 and cd103 for identification of human tumor reactive cells for treatment of cancer |
| JP2018536632A (en) * | 2015-10-08 | 2018-12-13 | マクロジェニクス,インコーポレーテッド | Combination therapy for the treatment of cancer |
| WO2018225033A1 (en) | 2017-06-09 | 2018-12-13 | Glaxosmithkline Intellectual Property Development Limited | Combination therapy |
| WO2018225093A1 (en) | 2017-06-07 | 2018-12-13 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds as atf4 pathway inhibitors |
| US10160806B2 (en) | 2014-06-26 | 2018-12-25 | Macrogenics, Inc. | Covalently bonded diabodies having immunoreactivity with PD-1 and LAG-3, and methods of use thereof |
| WO2019008506A1 (en) | 2017-07-03 | 2019-01-10 | Glaxosmithkline Intellectual Property Development Limited | N-(3-(2-(4-chlorophenoxy)acetamido)bicyclo[1.1.1]pentan-1-yl)-2-cyclobutane-1-carboxamide derivatives and related compounds as atf4 inhibitors for treating cancer and other diseases |
| WO2019008507A1 (en) | 2017-07-03 | 2019-01-10 | Glaxosmithkline Intellectual Property Development Limited | 2-(4-chlorophenoxy)-n-((1 -(2-(4-chlorophenoxy)ethynazetidin-3-yl)methyl)acetamide derivatives and related compounds as atf4 inhibitors for treating cancer and other diseases |
| GB201820547D0 (en) | 2018-12-17 | 2019-01-30 | Oxford Univ Innovation | Modified antibodies |
| WO2019021208A1 (en) | 2017-07-27 | 2019-01-31 | Glaxosmithkline Intellectual Property Development Limited | Indazole derivatives useful as perk inhibitors |
| EP3444271A1 (en) | 2013-08-08 | 2019-02-20 | Cytune Pharma | Il-15 and il-15raplha sushi domain based modulokines |
| WO2019049061A1 (en) | 2017-09-07 | 2019-03-14 | Glaxosmithkline Intellectual Property Development Limited | 5-(1 h-benzo[d]imidazo-2-yl)-pyridin-2-amine and 5-(3h-imidazo[4,5-b]pyridin-6-yl)-pyridin-2-amine derivatives as c-myc and p300/cbp histone acetyltransferase inhibitors for treating cancer |
| EP3456346A1 (en) | 2015-07-30 | 2019-03-20 | MacroGenics, Inc. | Pd-1 and lag-3 binding molecules and methods of use thereof |
| WO2019053617A1 (en) | 2017-09-12 | 2019-03-21 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| EP3334763A4 (en) * | 2015-08-11 | 2019-04-03 | Wuxi Biologics (Cayman) Inc. | Novel anti-pd-1 antibodies |
| WO2019069269A1 (en) | 2017-10-05 | 2019-04-11 | Glaxosmithkline Intellectual Property Development Limited | Modulators of stimulator of interferon genes (sting) useful in treating hiv |
| WO2019069270A1 (en) | 2017-10-05 | 2019-04-11 | Glaxosmithkline Intellectual Property Development Limited | Modulators of stimulator of interferon genes (sting) |
| WO2019089412A1 (en) | 2017-11-01 | 2019-05-09 | Merck Sharp & Dohme Corp. | Novel substituted tetrahydroquinolin compounds as indoleamine 2,3-dioxygenase (ido) inhibitors |
| WO2019099294A1 (en) | 2017-11-14 | 2019-05-23 | Merck Sharp & Dohme Corp. | Novel substituted biaryl compounds as indoleamine 2,3-dioxygenase (ido) inhibitors |
| WO2019099314A1 (en) | 2017-11-14 | 2019-05-23 | Merck Sharp & Dohme Corp. | Novel substituted biaryl compounds as indoleamine 2,3-dioxygenase (ido) inhibitors |
| WO2019104289A1 (en) | 2017-11-27 | 2019-05-31 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepine antibody conjugates |
| EP3498734A1 (en) | 2014-02-04 | 2019-06-19 | Pfizer Inc | Combination of a pd-1 antagonist and a vegfr inhibitor for treating cancer |
| US10328158B2 (en) | 2014-01-10 | 2019-06-25 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for immunotherapy |
| WO2019126691A1 (en) | 2017-12-21 | 2019-06-27 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepine antibody conjugates |
| WO2019134946A1 (en) | 2018-01-04 | 2019-07-11 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma resistant |
| JP2019519247A (en) * | 2016-04-01 | 2019-07-11 | アケソ・バイオファーマ・インコーポレイテッド | Anti-PD-1 monoclonal antibody |
| WO2019147670A1 (en) | 2018-01-23 | 2019-08-01 | Nextcure, Inc. | B7-h4 antibodies and methods of use thereof |
| US10370448B2 (en) | 2008-09-26 | 2019-08-06 | Emory University | Human anti-PD-1, PD-L1, and PD-L2 antibodies and uses therefor |
| WO2019149716A1 (en) | 2018-01-31 | 2019-08-08 | F. Hoffmann-La Roche Ag | Bispecific antibodies comprising an antigen-binding site binding to lag3 |
| WO2019162325A1 (en) | 2018-02-21 | 2019-08-29 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of sk1 as biomarker for predicting response to immunecheckpoint inhibitors |
| WO2019193540A1 (en) | 2018-04-06 | 2019-10-10 | Glaxosmithkline Intellectual Property Development Limited | Heteroaryl derivatives of formula (i) as atf4 inhibitors |
| WO2019193541A1 (en) | 2018-04-06 | 2019-10-10 | Glaxosmithkline Intellectual Property Development Limited | Bicyclic aromatic ring derivatives of formula (i) as atf4 inhibitors |
| WO2019195124A1 (en) | 2018-04-03 | 2019-10-10 | Merck Sharp & Dohme Corp. | Benzothiophenes and related compounds as sting agonists |
| EP3552615A1 (en) | 2014-07-16 | 2019-10-16 | Transgene SA | Oncolytic virus for expression of immune checkpoint modulators |
| US10457725B2 (en) | 2016-05-13 | 2019-10-29 | Regeneron Pharmaceuticals, Inc. | Methods of treating skin cancer by administering a PD-1 inhibitor |
| US10472419B2 (en) | 2014-01-31 | 2019-11-12 | Novartis Ag | Antibody molecules to TIM-3 and uses thereof |
| WO2019219709A1 (en) | 2018-05-14 | 2019-11-21 | Immunocore Limited | Bifunctional binding polypeptides |
| WO2019219820A1 (en) | 2018-05-16 | 2019-11-21 | Ctxt Pty Limited | Substituted condensed thiophenes as modulators of sting |
| US10493148B2 (en) | 2018-03-02 | 2019-12-03 | Eli Lilly And Company | PD-1 agonist antibodies and uses thereof |
| WO2019231870A1 (en) | 2018-05-31 | 2019-12-05 | Merck Sharp & Dohme Corp. | Novel substituted [1.1.1] bicyclo compounds as indoleamine 2,3-dioxygenase inhibitors |
| WO2019232319A1 (en) | 2018-05-31 | 2019-12-05 | Peloton Therapeutics, Inc. | Compositions and methods for inhibiting cd73 |
| WO2019241730A2 (en) | 2018-06-15 | 2019-12-19 | Flagship Pioneering Innovations V, Inc. | Increasing immune activity through modulation of postcellular signaling factors |
| WO2020012339A1 (en) | 2018-07-09 | 2020-01-16 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| WO2020011972A1 (en) | 2018-07-12 | 2020-01-16 | F-Star Beta Limited | Fc binding fragments comprising cd137 antigne binding side |
| WO2020023268A1 (en) | 2018-07-24 | 2020-01-30 | Amgen Inc. | Combination of lilrb1/2 pathway inhibitors and pd-1 pathway inhibitors |
| US10548988B2 (en) | 2012-07-18 | 2020-02-04 | Birdie Biopharmaceuticals, Inc. | Compounds for targeted immunotherapy |
| WO2020031107A1 (en) | 2018-08-08 | 2020-02-13 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| US10570204B2 (en) | 2013-09-26 | 2020-02-25 | The Medical College Of Wisconsin, Inc. | Methods for treating hematologic cancers |
| WO2020044206A1 (en) | 2018-08-29 | 2020-03-05 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides as kinase inhibitors for use in the treatment cancer |
| WO2020047345A1 (en) | 2018-08-31 | 2020-03-05 | Yale University | Compositions and methods of using cell-penetrating antibodies in combination with immune checkpoint modulators |
| WO2020055702A1 (en) | 2018-09-13 | 2020-03-19 | Merck Sharp & Dohme Corp. | Combination of pd-1 antagonist and lag3 antagonist for treating non-microsatellite instablity-high/proficient mismatch repair colorectal cancer |
| WO2020079692A1 (en) | 2018-10-17 | 2020-04-23 | Biolinerx Ltd. | Treatment of metastatic pancreatic adenocarcinoma |
| WO2020086479A1 (en) | 2018-10-22 | 2020-04-30 | Glaxosmithkline Intellectual Property Development Limited | Dosing |
| US20200131266A1 (en) * | 2017-05-31 | 2020-04-30 | Stcube & Co., Inc. | Methods of treating cancer using antibodies and molecules that immunospecifically bind to btn1a1 |
| WO2020092385A1 (en) | 2018-10-29 | 2020-05-07 | Mersana Therapeutics, Inc. | Cysteine engineered antibody-drug conjugates with peptide-containing linkers |
| WO2020092183A1 (en) | 2018-11-01 | 2020-05-07 | Merck Sharp & Dohme Corp. | Novel substituted pyrazole compounds as indoleamine 2,3-dioxygenase inhibitors |
| WO2020096871A1 (en) | 2018-11-06 | 2020-05-14 | Merck Sharp & Dohme Corp. | Novel substituted tricyclic compounds as indoleamine 2,3-dioxygenase inhibitors |
| WO2020102804A2 (en) | 2018-11-16 | 2020-05-22 | Arqule, Inc. | Pharmaceutical combination for treatment of cancer |
| WO2020104496A1 (en) | 2018-11-20 | 2020-05-28 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Bispecific antibody targeting transferrin receptor 1 and soluble antigen |
| WO2020104479A1 (en) | 2018-11-20 | 2020-05-28 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating cancers and resistant cancers with anti transferrin receptor 1 antibodies |
| WO2020106560A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020106558A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| EP3659622A1 (en) | 2013-08-08 | 2020-06-03 | Cytune Pharma | Combined pharmaceutical composition |
| WO2020112700A1 (en) | 2018-11-30 | 2020-06-04 | Merck Sharp & Dohme Corp. | 9-substituted amino triazolo quinazoline derivatives as adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020112581A1 (en) | 2018-11-28 | 2020-06-04 | Merck Sharp & Dohme Corp. | Novel substituted piperazine amide compounds as indoleamine 2, 3-dioxygenase (ido) inhibitors |
| WO2020110056A1 (en) | 2018-11-30 | 2020-06-04 | Glaxosmithkline Intellectual Property Development Limited | Compounds useful in hiv therapy |
| WO2020115261A1 (en) | 2018-12-07 | 2020-06-11 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| EP3613768A4 (en) * | 2017-04-20 | 2020-06-17 | Suzhou Stainwei Biotech Inc. | Monoclonal antibody antagonizing and inhibiting binding between human pd-1 antigen and ligand thereof, preparation method therefor and application thereof |
| WO2020120592A1 (en) | 2018-12-12 | 2020-06-18 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for predicting and treating melanoma |
| WO2020123453A2 (en) | 2018-12-11 | 2020-06-18 | Theravance Biopharma R&D Ip, Llc | Alk5 inhibitors |
| WO2020127411A1 (en) | 2018-12-19 | 2020-06-25 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating cancers by immuno-modulation using antibodies against cathespin-d |
| WO2020132560A2 (en) | 2018-12-21 | 2020-06-25 | Aim Immunotech Inc. | Compositions and methods for cancer therapy |
| WO2020127885A1 (en) | 2018-12-21 | 2020-06-25 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Compositions for treating cancers and resistant cancers |
| US10695426B2 (en) | 2014-08-25 | 2020-06-30 | Pfizer Inc. | Combination of a PD-1 antagonist and an ALK inhibitor for treating cancer |
| WO2020157131A1 (en) | 2019-01-30 | 2020-08-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for identifying whether a subject suffering from a cancer will achieve a response with an immune-checkpoint inhibitor |
| WO2020161083A1 (en) | 2019-02-04 | 2020-08-13 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for modulating blood-brain barrier |
| WO2020165370A1 (en) | 2019-02-13 | 2020-08-20 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for selecting a cancer treatment in a subject suffering from cancer |
| US10765710B2 (en) | 2014-07-16 | 2020-09-08 | Institut Gustave-Roussy | Combination of oncolytic virus with immune checkpoint modulators |
| WO2020183011A1 (en) | 2019-03-14 | 2020-09-17 | Institut Curie | Htr1d inhibitors and uses thereof in the treatment of cancer |
| WO2020205688A1 (en) | 2019-04-04 | 2020-10-08 | Merck Sharp & Dohme Corp. | Inhibitors of histone deacetylase-3 useful for the treatment of cancer, inflammation, neurodegeneration diseases and diabetes |
| WO2020201095A1 (en) | 2019-03-29 | 2020-10-08 | Institut Curie | Interleukin-2 variants with modified biological activity |
| US10800846B2 (en) | 2015-02-26 | 2020-10-13 | Merck Patent Gmbh | PD-1/PD-L1 inhibitors for the treatment of cancer |
| WO2020221796A1 (en) | 2019-04-30 | 2020-11-05 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2020227159A2 (en) | 2019-05-03 | 2020-11-12 | Flagship Pioneering Innovations V, Inc. | Methods of modulating immune activity |
| WO2020232375A1 (en) | 2019-05-16 | 2020-11-19 | Silicon Swat, Inc. | Oxoacridinyl acetic acid derivatives and methods of use |
| WO2020232378A1 (en) | 2019-05-16 | 2020-11-19 | Silicon Swat, Inc. | Benzo[b][1,8]naphthyridine acetic acid derivatives and methods of use |
| WO2020239558A1 (en) | 2019-05-24 | 2020-12-03 | Pfizer Inc. | Combination therapies using cdk inhibitors |
| US10869924B2 (en) | 2015-06-16 | 2020-12-22 | Merck Patent Gmbh | PD-L1 antagonist combination treatments |
| WO2020260547A1 (en) | 2019-06-27 | 2020-12-30 | Rigontec Gmbh | Design method for optimized rig-i ligands |
| EP3763742A1 (en) | 2014-09-01 | 2021-01-13 | Birdie Biopharmaceuticals Inc. | Anti-pd-l1 conjugates for treating tumors |
| WO2021006199A1 (en) | 2019-07-05 | 2021-01-14 | 小野薬品工業株式会社 | Treatment of hematologic cancer with pd-1/cd3 dual specificity protein |
| WO2020247648A3 (en) * | 2019-06-05 | 2021-01-14 | Anaptysbio, Inc. | Pd-1 agonist and method of using same |
| WO2021009365A1 (en) | 2019-07-18 | 2021-01-21 | Ctxt Pty Limited | Benzothiophene, thienopyridine and thienopyrimidine derivatives for the modulation of sting |
| WO2021009362A1 (en) | 2019-07-18 | 2021-01-21 | Ctxt Pty Limited | Benzothiophene, thienopyridine and thienopyrimidine derivatives for the modulation of sting |
| US10899840B2 (en) | 2014-02-04 | 2021-01-26 | Pfizer Inc. | Combination of a PD-1 antagonist and a 4-1BB agonist for treating cancer |
| WO2021020336A1 (en) | 2019-07-26 | 2021-02-04 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Pharmaceutical composition for treating tumor |
| WO2021025140A1 (en) | 2019-08-08 | 2021-02-11 | 小野薬品工業株式会社 | Dual-specific protein |
| AU2015276978B2 (en) * | 2014-06-19 | 2021-02-25 | Regeneron Pharmaceuticals, Inc. | Non-human animals having a humanized programmed cell death 1 gene |
| US10934356B2 (en) | 2014-11-13 | 2021-03-02 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| WO2021048292A1 (en) | 2019-09-11 | 2021-03-18 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| US10961310B2 (en) | 2017-03-15 | 2021-03-30 | Pandion Operations, Inc. | Targeted immunotolerance |
| WO2021058711A2 (en) | 2019-09-27 | 2021-04-01 | Glaxosmithkline Intellectual Property Development Limited | Antigen binding proteins |
| WO2021064180A1 (en) | 2019-10-03 | 2021-04-08 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for modulating macrophages polarization |
| WO2021074391A1 (en) | 2019-10-17 | 2021-04-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for diagnosing nasal intestinal type adenocarcinomas |
| WO2021078910A1 (en) | 2019-10-22 | 2021-04-29 | Institut Curie | Immunotherapy targeting tumor neoantigenic peptides |
| WO2021083959A1 (en) | 2019-10-29 | 2021-05-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating uveal melanoma |
| WO2021083060A1 (en) | 2019-10-28 | 2021-05-06 | 中国科学院上海药物研究所 | Five-membered heterocyclic oxocarboxylic acid compound and medical use thereof |
| WO2021086909A1 (en) | 2019-10-29 | 2021-05-06 | Eisai R&D Managment Co., Ltd. | Combination of a pd-1 antagonist, a vegfr/fgfr/ret tyrosine kinase inhibitor and a cbp/beta-catenin inhibitor for treating cancer |
| US11014983B2 (en) | 2015-08-20 | 2021-05-25 | Sutro Biopharma, Inc. | Anti-Tim-3 antibodies, compositions comprising anti-Tim-3 antibodies and methods of making and using anti-Tim-3 antibodies |
| WO2021099511A1 (en) | 2019-11-22 | 2021-05-27 | Institut Curie | Device, apparatus and method for minibeam radiation therapy |
| WO2021102468A1 (en) | 2019-11-22 | 2021-05-27 | Theravance Biopharma R&D Ip, Llc | Substituted 1,5-naphthyridines or quinolines as alk5 inhibitors |
| WO2021108025A1 (en) | 2019-11-26 | 2021-06-03 | Massachusetts Institute Of Technology | Cell-based cancer vaccines and cancer therapies |
| WO2021113679A1 (en) | 2019-12-06 | 2021-06-10 | Mersana Therapeutics, Inc. | Dimeric compounds as sting agonists |
| US11040948B2 (en) | 2017-09-29 | 2021-06-22 | Curis, Inc. | Crystal forms of immunomodulators |
| WO2021123243A1 (en) | 2019-12-19 | 2021-06-24 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and vaccine compositions to treat cancers |
| WO2021127217A1 (en) | 2019-12-17 | 2021-06-24 | Flagship Pioneering Innovations V, Inc. | Combination anti-cancer therapies with inducers of iron-dependent cellular disassembly |
| US11046781B2 (en) | 2016-01-07 | 2021-06-29 | Birdie Biopharmaceuticals, Inc. | Anti-HER2 combinations for treating tumors |
| US11053240B2 (en) | 2017-04-27 | 2021-07-06 | Birdie Biopharmaceuticals, Inc. | 2-amino-quinoline derivatives |
| WO2021144426A1 (en) | 2020-01-17 | 2021-07-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| US11072653B2 (en) | 2015-06-08 | 2021-07-27 | Macrogenics, Inc. | LAG-3-binding molecules and methods of use thereof |
| US11078279B2 (en) | 2015-06-12 | 2021-08-03 | Macrogenics, Inc. | Combination therapy for the treatment of cancer |
| WO2021156360A1 (en) | 2020-02-05 | 2021-08-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for discontinuing a treatment with a tyrosine kinase inhibitor (tki) |
| US11091526B2 (en) | 2017-12-06 | 2021-08-17 | Pandion Operations, Inc. | IL-2 muteins and uses thereof |
| WO2021170777A1 (en) | 2020-02-28 | 2021-09-02 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for diagnosing, prognosing and managing treatment of breast cancer |
| US11136397B2 (en) | 2016-01-07 | 2021-10-05 | Birdie Pharmaceuticals, Inc. | Anti-EGFR combinations for treating tumors |
| US11136300B2 (en) | 2017-10-11 | 2021-10-05 | Aurigene Discovery Technologies Limited | Crystalline forms of 3-substituted 1,2,4-oxadiazole |
| WO2021203131A1 (en) | 2020-03-31 | 2021-10-07 | Theravance Biopharma R&D Ip, Llc | Substituted pyrimidines and methods of use |
| WO2021209357A1 (en) | 2020-04-14 | 2021-10-21 | Glaxosmithkline Intellectual Property Development Limited | Combination treatment for cancer involving anti-icos and anti-pd1 antibodies, optionally further involving anti-tim3 antibodies |
| WO2021209356A1 (en) | 2020-04-14 | 2021-10-21 | Glaxosmithkline Intellectual Property Development Limited | Combination treatment for cancer |
| US11155624B2 (en) | 2016-11-01 | 2021-10-26 | Anaptysbio, Inc. | Antibodies directed against programmed death-1 (PD-1) |
| WO2021222188A1 (en) | 2020-04-27 | 2021-11-04 | Seagen Inc. | Anti-cd40 antibody combination treatment for cancer |
| WO2021226003A1 (en) | 2020-05-06 | 2021-11-11 | Merck Sharp & Dohme Corp. | Il4i1 inhibitors and methods of use |
| WO2021224186A1 (en) | 2020-05-04 | 2021-11-11 | Institut Curie | New pyridine derivatives as radiosensitizers |
| WO2021231350A1 (en) | 2020-05-13 | 2021-11-18 | Massachusetts Institute Of Technology | Compositions of polymeric microdevices and their use in cancer immunotherapy |
| WO2021234110A1 (en) | 2020-05-20 | 2021-11-25 | Institut Curie | Single domain antibodies and their use in cancer therapies |
| WO2021248065A1 (en) | 2020-06-05 | 2021-12-09 | Theraly Fibrosis, Inc. | Trail compositions with reduced immunogenicity |
| WO2021253041A1 (en) | 2020-06-10 | 2021-12-16 | Theravance Biopharma R&D Ip, Llc | Naphthyridine derivatives useful as alk5 inhibitors |
| WO2021260675A1 (en) | 2020-06-24 | 2021-12-30 | Yeda Research And Development Co. Ltd. | Agents for sensitizing solid tumors to treatment |
| WO2022006179A1 (en) | 2020-06-29 | 2022-01-06 | Flagship Pioneering Innovations V, Inc. | Viruses engineered to promote thanotransmission and their use in treating cancer |
| US11220552B2 (en) | 2016-01-07 | 2022-01-11 | Birdie Biopharmaceuticals, Inc. | Anti-CD20 combinations for treating tumors |
| WO2022023379A1 (en) | 2020-07-28 | 2022-02-03 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for preventing and treating a cancer |
| CN114133448A (en) * | 2015-06-23 | 2022-03-04 | 纪念斯隆-凯特琳癌症中心 | Novel PD-1 immunomodulators |
| WO2022049526A1 (en) | 2020-09-02 | 2022-03-10 | Pharmabcine Inc. | Combination therapy of a pd-1 antagonist and an antagonist for vegfr-2 for treating patients with cancer |
| US11274154B2 (en) | 2016-10-06 | 2022-03-15 | Pfizer Inc. | Dosing regimen of avelumab for the treatment of cancer |
| US11285131B2 (en) | 2017-08-04 | 2022-03-29 | Merck Sharp & Dohme Corp. | Benzo[b]thiophene STING agonists for cancer treatment |
| US11292842B2 (en) | 2017-02-21 | 2022-04-05 | Regeneron Pharmaceuticals, Inc. | Anti-PD-1 antibodies for treatment of lung cancer |
| WO2022079270A1 (en) | 2020-10-16 | 2022-04-21 | Université D'aix-Marseille | Anti-gpc4 single domain antibodies |
| US11312772B2 (en) | 2017-08-04 | 2022-04-26 | Merck Sharp & Dohme Corp. | Combinations of PD-1 antagonists and benzo [b] thiophene STING agonists for cancer treatment |
| WO2022084325A1 (en) | 2020-10-20 | 2022-04-28 | Institut Curie | Metallic trans-(n-heterocyclic carbene)-amine-platinum complexes and uses thereof for treating cancer |
| WO2022084531A1 (en) | 2020-10-23 | 2022-04-28 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating glioma |
| WO2022092085A1 (en) | 2020-10-28 | 2022-05-05 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Pharmaceutical composition for treating tumors |
| US11326170B2 (en) | 2017-04-18 | 2022-05-10 | Changchun Huapu Biotechnology Co. Ltd. | Immunomodulatory polynucleotides and uses thereof |
| WO2022101481A1 (en) | 2020-11-16 | 2022-05-19 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for predicting and treating uveal melanoma |
| WO2022101484A1 (en) | 2020-11-16 | 2022-05-19 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for predicting and treating uveal melanoma |
| WO2022106505A1 (en) | 2020-11-18 | 2022-05-27 | Institut Curie | Dimers of biguanidines and their therapeutic uses |
| US11344620B2 (en) | 2014-09-13 | 2022-05-31 | Novartis Ag | Combination therapies |
| WO2022130206A1 (en) | 2020-12-16 | 2022-06-23 | Pfizer Inc. | TGFβr1 INHIBITOR COMBINATION THERAPIES |
| WO2022152862A1 (en) | 2021-01-14 | 2022-07-21 | Institut Curie | Her2 single domain antibodies variants and cars thereof |
| WO2022165403A1 (en) | 2021-02-01 | 2022-08-04 | Yale University | Chemotherapeutic bioadhesive particles with immunostimulatory molecules for cancer treatment |
| US11407830B2 (en) | 2017-01-09 | 2022-08-09 | Tesaro, Inc. | Methods of treating cancer with anti-PD-1 antibodies |
| US11413331B2 (en) | 2017-04-03 | 2022-08-16 | Hoffmann-La Roche Inc. | Immunoconjugates |
| WO2022185160A1 (en) | 2021-03-02 | 2022-09-09 | Glaxosmithkline Intellectual Property Development Limited | Substituted pyridines as dnmt1 inhibitors |
| WO2022189618A1 (en) | 2021-03-12 | 2022-09-15 | Institut Curie | Nitrogen-containing heterocycles as radiosensitizers |
| WO2022194908A1 (en) | 2021-03-17 | 2022-09-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| US11453697B1 (en) | 2015-08-13 | 2022-09-27 | Merck Sharp & Dohme Llc | Cyclic di-nucleotide compounds as sting agonists |
| US11459394B2 (en) | 2017-02-24 | 2022-10-04 | Macrogenics, Inc. | Bispecific binding molecules that are capable of binding CD137 and tumor antigens, and uses thereof |
| WO2022212784A1 (en) | 2021-03-31 | 2022-10-06 | Flagship Pioneering Innovations V, Inc. | Thanotransmission polypeptides and their use in treating cancer |
| WO2022208353A1 (en) | 2021-03-31 | 2022-10-06 | Glaxosmithkline Intellectual Property Development Limited | Antigen binding proteins and combinations thereof |
| US11466068B2 (en) | 2017-05-24 | 2022-10-11 | Pandion Operations, Inc. | Targeted immunotolerance |
| US11466047B2 (en) | 2017-05-12 | 2022-10-11 | Merck Sharp & Dohme Llc | Cyclic di-nucleotide compounds as sting agonists |
| WO2022217123A2 (en) | 2021-04-08 | 2022-10-13 | Nurix Therapeutics, Inc. | Combination therapies with cbl-b inhibitor compounds |
| EP3818146A4 (en) * | 2018-07-04 | 2022-10-19 | Cytoimmune Therapeutics, Inc. | Compositions and methods for immunotherapy targeting flt3, pd-1, and/or pd-l1 |
| WO2022223791A1 (en) | 2021-04-23 | 2022-10-27 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating cell senescence accumulation related disease |
| WO2022232333A1 (en) | 2021-04-30 | 2022-11-03 | Merck Sharp & Dohme Llc | Il4i1 inhibitors and methods of use |
| US11497734B2 (en) | 2017-11-03 | 2022-11-15 | Aurigene Discovery Technologies Limited | Dual inhibitors of TIM-3 and PD-1 pathways |
| US11497735B2 (en) | 2017-11-06 | 2022-11-15 | Aurigene Discovery Technologies Limited | Conjoint therapies for immunomodulation |
| WO2022239820A1 (en) | 2021-05-13 | 2022-11-17 | 公益財団法人神戸医療産業都市推進機構 | Anti-human pd-1 agonist antibody for treating or preventing inflammatory diseases, and pharmaceutical composition containing same |
| WO2022238386A1 (en) | 2021-05-10 | 2022-11-17 | Institut Curie | Methods for the treatment of cancer, inflammatory diseases and autoimmune diseases |
| WO2022251359A1 (en) | 2021-05-26 | 2022-12-01 | Theravance Biopharma R&D Ip, Llc | Bicyclic inhibitors of alk5 and methods of use |
| US11517567B2 (en) | 2017-06-23 | 2022-12-06 | Birdie Biopharmaceuticals, Inc. | Pharmaceutical compositions |
| WO2022256534A1 (en) | 2021-06-03 | 2022-12-08 | Synthorx, Inc. | Head and neck cancer combination therapy comprising an il-2 conjugate and pembrolizumab |
| WO2022254227A1 (en) | 2021-06-04 | 2022-12-08 | Kymab Limited | Treatment of pd-l1 negative or low expressing cancer with anti-icos antibodies |
| WO2022261018A1 (en) | 2021-06-07 | 2022-12-15 | Providence Health & Services - Oregon | Cxcr5, pd-1, and icos expressing tumor reactive cd4 t cells and their use |
| US11542505B1 (en) | 2018-04-20 | 2023-01-03 | Merck Sharp & Dohme Llc | Substituted RIG-I agonists: compositions and methods thereof |
| US11542328B2 (en) | 2008-11-14 | 2023-01-03 | The Brigham And Women's Hospital, Inc. | Therapeutic and diagnostic methods relating to cancer stem cells |
| WO2023278641A1 (en) | 2021-06-29 | 2023-01-05 | Flagship Pioneering Innovations V, Inc. | Immune cells engineered to promote thanotransmission and uses thereof |
| US11559504B2 (en) | 2017-06-02 | 2023-01-24 | The Penn State Research Foundation | Ceramide nanoliposomes, compositions and methods of using for immunotherapy |
| WO2023010080A1 (en) | 2021-07-30 | 2023-02-02 | Seagen Inc. | Treatment for cancer |
| WO2023011879A1 (en) | 2021-08-05 | 2023-02-09 | Institut Curie | Scanning dynamic device for minibeams production |
| US11603407B2 (en) | 2017-04-06 | 2023-03-14 | Regeneron Pharmaceuticals, Inc. | Stable antibody formulation |
| WO2023041744A1 (en) | 2021-09-17 | 2023-03-23 | Institut Curie | Bet inhibitors for treating pab1 deficient cancer |
| WO2023057882A1 (en) | 2021-10-05 | 2023-04-13 | Pfizer Inc. | Combinations of azalactam compounds with a pd-1 axis binding antagonist for the treatment of cancer |
| US11633476B2 (en) | 2017-05-02 | 2023-04-25 | Merck Sharp & Dohme Llc | Stable formulations of programmed death receptor 1 (PD-1) antibodies and methods of use thereof |
| WO2023078900A1 (en) | 2021-11-03 | 2023-05-11 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating triple negative breast cancer (tnbc) |
| WO2023079428A1 (en) | 2021-11-03 | 2023-05-11 | Pfizer Inc. | Combination therapies using tlr7/8 agonist |
| WO2023089032A1 (en) | 2021-11-19 | 2023-05-25 | Institut Curie | Methods for the treatment of hrd cancer and brca-associated cancer |
| WO2023099763A1 (en) | 2021-12-03 | 2023-06-08 | Institut Curie | Sirt6 inhibitors for use in treating resistant hrd cancer |
| US11685761B2 (en) | 2017-12-20 | 2023-06-27 | Merck Sharp & Dohme Llc | Cyclic di-nucleotide compounds as sting agonists |
| WO2023118165A1 (en) | 2021-12-21 | 2023-06-29 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2023122573A1 (en) | 2021-12-20 | 2023-06-29 | Synthorx, Inc. | Head and neck cancer combination therapy comprising an il-2 conjugate and pembrolizumab |
| US11702430B2 (en) | 2018-04-03 | 2023-07-18 | Merck Sharp & Dohme Llc | Aza-benzothiophene compounds as STING agonists |
| US11739146B2 (en) | 2019-05-20 | 2023-08-29 | Pandion Operations, Inc. | MAdCAM targeted immunotolerance |
| WO2023180552A1 (en) | 2022-03-24 | 2023-09-28 | Institut Curie | Immunotherapy targeting tumor transposable element derived neoantigenic peptides in glioblastoma |
| WO2023039243A3 (en) * | 2021-09-13 | 2023-09-28 | Achelois Biopharma, Inc. | Hepatitis b virus antivirus (hbv-antivirus) compositions and methods of use |
| WO2023187024A1 (en) | 2022-03-31 | 2023-10-05 | Institut Curie | Modified rela protein for inducing interferon expression and engineered immune cells with improved interferon expression |
| US11779632B2 (en) | 2017-12-06 | 2023-10-10 | Pandion Operation, Inc. | IL-2 muteins and uses thereof |
| WO2023194607A1 (en) | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor with cd40 and uses thereof for anti-cancer therapy |
| WO2023194608A1 (en) | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor and uses thereof for anti-cancer therapy |
| US11795219B2 (en) | 2016-05-18 | 2023-10-24 | Boehringer Ingelheim International Gmbh | Antibody molecules for cancer treatment |
| WO2023213764A1 (en) | 2022-05-02 | 2023-11-09 | Transgene | Fusion polypeptide comprising an anti-pd-l1 sdab and a member of the tnfsf |
| WO2023213763A1 (en) | 2022-05-02 | 2023-11-09 | Transgene | Poxvirus encoding a binding agent comprising an anti- pd-l1 sdab |
| WO2023230541A1 (en) | 2022-05-27 | 2023-11-30 | Viiv Healthcare Company | Piperazine derivatives useful in hiv therapy |
| WO2023230554A1 (en) | 2022-05-25 | 2023-11-30 | Pfizer Inc. | Combination of a braf inhibitor, an egfr inhibitor, and a pd-1 antagonist for the treatment of braf v600e-mutant, msi-h/dmmr colorectal cancer |
| US11845798B2 (en) | 2017-05-02 | 2023-12-19 | Merck Sharp & Dohme Llc | Formulations of anti-LAG3 antibodies and co-formulations of anti-LAG3 antibodies and anti-PD-1 antibodies |
| WO2024003353A1 (en) | 2022-07-01 | 2024-01-04 | Transgene | Fusion protein comprising a surfactant-protein-d and a member of the tnfsf |
| WO2024033399A1 (en) | 2022-08-10 | 2024-02-15 | Institut National de la Santé et de la Recherche Médicale | Sigmar1 ligand for the treatment of pancreatic cancer |
| WO2024033400A1 (en) | 2022-08-10 | 2024-02-15 | Institut National de la Santé et de la Recherche Médicale | Sk2 inhibitor for the treatment of pancreatic cancer |
| WO2024040264A1 (en) | 2022-08-19 | 2024-02-22 | Massachusetts Institute Of Technology | Compositions and methods for targeting dendritic cell lectins |
| US11932692B2 (en) | 2020-09-03 | 2024-03-19 | Regeneron Pharmaceuticals, Inc. | Methods of treating cancer pain by administering a PD-1 inhibitor |
| WO2024056716A1 (en) | 2022-09-14 | 2024-03-21 | Institut National de la Santé et de la Recherche Médicale | Methods and pharmaceutical compositions for the treatment of dilated cardiomyopathy |
| US11939343B2 (en) | 2019-08-02 | 2024-03-26 | Mersana Therapeutics, Inc. | Sting agonist compounds and methods of use |
| US11946094B2 (en) | 2017-12-10 | 2024-04-02 | Augusta University Research Institute, Inc. | Combination therapies and methods of use thereof |
| WO2024068617A1 (en) | 2022-09-26 | 2024-04-04 | Institut Curie | Myeloid cells expressing il-2 and uses thereof for quick anticancer therapy |
| WO2024077191A1 (en) | 2022-10-05 | 2024-04-11 | Flagship Pioneering Innovations V, Inc. | Nucleic acid molecules encoding trif and additionalpolypeptides and their use in treating cancer |
| WO2024081736A2 (en) | 2022-10-11 | 2024-04-18 | Yale University | Compositions and methods of using cell-penetrating antibodies |
| WO2024084034A1 (en) | 2022-10-21 | 2024-04-25 | Institut National de la Santé et de la Recherche Médicale | Methods and pharmaceutical compositions for the treatment of osteoarthritis |
| US11981715B2 (en) | 2020-02-21 | 2024-05-14 | Pandion Operations, Inc. | Tissue targeted immunotolerance with a CD39 effector |
| WO2024112867A1 (en) | 2022-11-23 | 2024-05-30 | University Of Georgia Research Foundation, Inc. | Compositions and methods of use thereof for increasing immune responses |
| US12029724B2 (en) | 2016-04-28 | 2024-07-09 | Eisai R&D Management Co., Ltd. | Method for inhibiting tumor growth |
| US12036204B2 (en) | 2019-07-26 | 2024-07-16 | Eisai R&D Management Co., Ltd. | Pharmaceutical composition for treating tumor |
| WO2024151687A1 (en) | 2023-01-09 | 2024-07-18 | Flagship Pioneering Innovations V, Inc. | Genetic switches and their use in treating cancer |
| US12042560B2 (en) | 2009-03-30 | 2024-07-23 | Eisai R&D Management Co., Ltd. | Liposome composition |
| US12054557B2 (en) | 2015-12-22 | 2024-08-06 | Regeneron Pharmaceuticals, Inc. | Combination of anti-PD-1 antibodies and bispecific anti-CD20/anti-CD3 antibodies to treat cancer |
| WO2024160721A1 (en) | 2023-01-30 | 2024-08-08 | Kymab Limited | Antibodies |
| WO2024163477A1 (en) | 2023-01-31 | 2024-08-08 | University Of Rochester | Immune checkpoint blockade therapy for treating staphylococcus aureus infections |
| WO2024194692A1 (en) | 2023-03-21 | 2024-09-26 | Institut Curie | Tgf-beta inhibitor for use in the treatment of dedifferentiated liposarcoma |
| WO2024194402A1 (en) | 2023-03-21 | 2024-09-26 | Institut Curie | Farnesyl transferase inhibitor for use in methods for the treatment of hrd cancer |
| WO2024194401A1 (en) | 2023-03-21 | 2024-09-26 | Institut Curie | Vps4b inhibitor for use in methods for the treatment of hrd cancer |
| WO2024200571A1 (en) | 2023-03-28 | 2024-10-03 | Institut National de la Santé et de la Recherche Médicale | Method for discriminating mono-immunotherapy from combined immunotherapy in cancers |
| WO2024206357A1 (en) | 2023-03-29 | 2024-10-03 | Merck Sharp & Dohme Llc | Il4i1 inhibitors and methods of use |
Families Citing this family (826)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7030219B2 (en) | 2000-04-28 | 2006-04-18 | Johns Hopkins University | B7-DC, Dendritic cell co-stimulatory molecules |
| JP2007530029A (en) * | 2004-03-26 | 2007-11-01 | クアーク・ファーマスーティカルス、インコーポレイテッド | Annexin II and uses thereof |
| PT2347775T (en) | 2005-12-13 | 2020-07-14 | The President And Fellows Of Harvard College | Scaffolds for cell transplantation |
| CN101663323A (en) | 2006-12-27 | 2010-03-03 | 埃默里大学 | The composition and the method that are used for the treatment of transmissible disease and tumour |
| AU2014201367B2 (en) * | 2007-06-18 | 2016-01-28 | Merck Sharp & Dohme B.V. | Antibodies to human programmed death receptor pd-1 |
| US8263078B2 (en) | 2007-09-05 | 2012-09-11 | Inotek Pharmaceuticals Corporation | Antibodies against flagellin and uses thereof |
| CA2715460C (en) | 2008-02-13 | 2020-02-18 | President And Fellows Of Harvard College | Continuous cell programming devices |
| CN102203125A (en) * | 2008-08-25 | 2011-09-28 | 安普利穆尼股份有限公司 | Pd-1 antagonists and methods of use thereof |
| EP2344540B1 (en) | 2008-10-02 | 2017-11-29 | Aptevo Research and Development LLC | Cd86 antagonist multi-target binding proteins |
| US20100239583A1 (en) * | 2009-03-04 | 2010-09-23 | Inotek Pharmaceuticals Corporation | Antibodies against flagellin and uses thereof |
| US8709417B2 (en) | 2009-09-30 | 2014-04-29 | Memorial Sloan-Kettering Cancer Center | Combination immunotherapy for the treatment of cancer |
| CA2790134A1 (en) * | 2010-02-16 | 2011-08-25 | Valorisation-Recherche, Limited Partnership | Pd-1 modulation and uses thereof for modulating hiv replication |
| US20150231215A1 (en) | 2012-06-22 | 2015-08-20 | Randolph J. Noelle | VISTA Antagonist and Methods of Use |
| US10745467B2 (en) | 2010-03-26 | 2020-08-18 | The Trustees Of Dartmouth College | VISTA-Ig for treatment of autoimmune, allergic and inflammatory disorders |
| EP2552947A4 (en) | 2010-03-26 | 2013-11-13 | Dartmouth College | Vista regulatory t cell mediator protein, vista binding agents and use thereof |
| CA2802344C (en) * | 2010-06-18 | 2023-06-13 | The Brigham And Women's Hospital, Inc. | Bi-specific antibodies against tim-3 and pd-1 for immunotherapy in chronic immune conditions |
| JP6126991B2 (en) * | 2010-09-27 | 2017-05-10 | ヤンセン バイオテツク,インコーポレーテツド | Antibody binding to human type II collagen |
| US8906649B2 (en) | 2010-09-27 | 2014-12-09 | Janssen Biotech, Inc. | Antibodies binding human collagen II |
| US11202759B2 (en) | 2010-10-06 | 2021-12-21 | President And Fellows Of Harvard College | Injectable, pore-forming hydrogels for materials-based cell therapies |
| CN103596985B (en) | 2011-04-19 | 2016-06-08 | 美国政府(由卫生和人类服务部的部长所代表) | The human monoclonal antibodies special to glypican-3 and purposes thereof |
| JP2014525904A (en) | 2011-06-28 | 2014-10-02 | ホワイトヘッド・インスティテュート・フォー・バイオメディカル・リサーチ | Using sortase to install click chemistry handles for protein ligation |
| WO2013013188A1 (en) | 2011-07-21 | 2013-01-24 | Tolero Pharmaceuticals, Inc. | Heterocyclic protein kinase inhibitors |
| EP4079319A1 (en) * | 2011-10-17 | 2022-10-26 | IO Biotech ApS | Pd-l1 based immunotherapy |
| SI2838515T1 (en) | 2012-04-16 | 2020-07-31 | President And Fellows Of Harvard College | Mesoporous silica compositions for modulating immune responses |
| CN115093480A (en) * | 2012-05-31 | 2022-09-23 | 索伦托药业有限公司 | Antigen binding proteins that bind to PD-L1 |
| CN107652289B (en) | 2012-06-13 | 2020-07-21 | 因塞特控股公司 | Substituted tricyclic compounds as FGFR inhibitors |
| US9890215B2 (en) | 2012-06-22 | 2018-02-13 | King's College London | Vista modulators for diagnosis and treatment of cancer |
| JP6457940B2 (en) | 2012-08-30 | 2019-01-23 | アムジエン・インコーポレーテツド | Method for treating melanoma using herpes simplex virus and immune checkpoint inhibitors |
| CA2884704C (en) | 2012-09-07 | 2023-04-04 | Randolph J. Noelle | Vista modulators for diagnosis and treatment of cancer |
| RU2708032C2 (en) | 2013-02-20 | 2019-12-03 | Новартис Аг | CANCER TREATMENT USING CHIMERIC ANTIGEN-SPECIFIC RECEPTOR BASED ON HUMANISED ANTI-EGFRvIII ANTIBODY |
| EP2958942B1 (en) | 2013-02-20 | 2020-06-03 | Novartis AG | Effective targeting of primary human leukemia using anti-cd123 chimeric antigen receptor engineered t cells |
| MX362247B (en) | 2013-03-06 | 2019-01-09 | Astrazeneca Ab | Quinazoline inhibitors of activating mutant forms of epidermal growth factor receptor. |
| WO2014165082A2 (en) * | 2013-03-13 | 2014-10-09 | Medimmune, Llc | Antibodies and methods of detection |
| US9308236B2 (en) | 2013-03-15 | 2016-04-12 | Bristol-Myers Squibb Company | Macrocyclic inhibitors of the PD-1/PD-L1 and CD80(B7-1)/PD-L1 protein/protein interactions |
| TWI654206B (en) | 2013-03-16 | 2019-03-21 | 諾華公司 | Treatment of cancer with a humanized anti-CD19 chimeric antigen receptor |
| US20160084839A1 (en) | 2013-04-02 | 2016-03-24 | Marisa Dolled-Filhart | Immunohistochemical assay for detecting expression of programmed death ligand 1 (pd-l1) in tumor tissue |
| EP2983661B1 (en) | 2013-04-09 | 2024-05-29 | Lixte Biotechnology, Inc. | Formulations of oxabicycloheptanes and oxabicycloheptenes |
| WO2014172392A1 (en) | 2013-04-18 | 2014-10-23 | Armo Biosciences, Inc. | Methods of using interleukin-10 for treating diseases and disorders |
| JP6449244B2 (en) | 2013-04-19 | 2019-01-09 | インサイト・ホールディングス・コーポレイションIncyte Holdings Corporation | Bicyclic heterocycles as FGFR inhibitors |
| EP2994491A4 (en) | 2013-05-10 | 2016-12-07 | Whitehead Inst Biomedical Res | In vitro production of red blood cells with sortaggable proteins |
| JP6603209B2 (en) | 2013-05-10 | 2019-11-06 | ホワイトヘッド・インスティテュート・フォー・バイオメディカル・リサーチ | Protein modification of living cells using sortase |
| ES2688206T3 (en) | 2013-06-17 | 2018-10-31 | Armo Biosciences, Inc. | Procedure for assessing protein identity and stability |
| KR102160389B1 (en) | 2013-08-05 | 2020-09-28 | 트위스트 바이오사이언스 코포레이션 | De novo synthesized gene libraries |
| AR097306A1 (en) | 2013-08-20 | 2016-03-02 | Merck Sharp & Dohme | MODULATION OF TUMOR IMMUNITY |
| MX2016002273A (en) | 2013-08-20 | 2016-05-31 | Merck Sharp & Dohme | Treating cancer with a combination of a pd-1 antagonist and dinaciclib. |
| US10010588B2 (en) | 2013-08-30 | 2018-07-03 | Armo Biosciences, Inc. | Methods of using pegylated interleukin-10 for treating hyperlipidemia |
| CN112457403B (en) | 2013-09-13 | 2022-11-29 | 广州百济神州生物制药有限公司 | anti-PD 1 antibodies and their use as therapeutic and diagnostic agents |
| CN104558177B (en) * | 2013-10-25 | 2020-02-18 | 苏州思坦维生物技术股份有限公司 | Monoclonal antibody for antagonizing and inhibiting programmed death receptor PD-1and ligand combination thereof, and coding sequence and application thereof |
| WO2015066413A1 (en) | 2013-11-01 | 2015-05-07 | Novartis Ag | Oxazolidinone hydroxamic acid compounds for the treatment of bacterial infections |
| RU2016122957A (en) | 2013-11-11 | 2017-12-19 | Армо Байосайенсиз, Инк. | Methods of using interleukin-10 for the treatment of diseases and disorders |
| US10556024B2 (en) | 2013-11-13 | 2020-02-11 | Whitehead Institute For Biomedical Research | 18F labeling of proteins using sortases |
| US20150140036A1 (en) | 2013-11-13 | 2015-05-21 | Novartis Institutes For Biomedical Research, Inc. | Low, immune enhancing, dose mtor inhibitors and uses thereof |
| EP3079772B1 (en) | 2013-12-10 | 2020-02-05 | Merck Sharp & Dohme Corp. | Immunohistochemical proximity assay for pd-1 positive cells and pd-ligand positive cells in tumor tissue |
| HUE046249T2 (en) | 2013-12-12 | 2020-02-28 | Shanghai hengrui pharmaceutical co ltd | Pd-1 antibody, antigen-binding fragment thereof, and medical application thereof |
| US20160304969A1 (en) | 2013-12-17 | 2016-10-20 | Merck Sharp & Dohme Corp. | Ifn-gamma gene signature biomarkers of tumor response to pd-1 antagonists |
| CN116478927A (en) | 2013-12-19 | 2023-07-25 | 诺华股份有限公司 | Human mesothelin chimeric antigen receptor and application thereof |
| US11014987B2 (en) | 2013-12-24 | 2021-05-25 | Janssen Pharmaceutics Nv | Anti-vista antibodies and fragments, uses thereof, and methods of identifying same |
| CN106661107B (en) | 2013-12-24 | 2021-12-24 | 杨森制药公司 | anti-VISTA antibodies and fragments |
| JO3517B1 (en) | 2014-01-17 | 2020-07-05 | Novartis Ag | N-azaspirocycloalkane substituted n-heteroaryl compounds and compositions for inhibiting the activity of shp2 |
| US20170088626A1 (en) | 2014-03-05 | 2017-03-30 | Bristol-Myers Squibb Company | Treatment of renal cancer using a combination of an anti-pd-1 antibody and another anti-cancer agent |
| CU24481B1 (en) | 2014-03-14 | 2020-03-04 | Immutep Sas | ANTIBODY MOLECULES THAT JOIN LAG-3 |
| EP3593812A3 (en) | 2014-03-15 | 2020-05-27 | Novartis AG | Treatment of cancer using chimeric antigen receptor |
| PL3122745T3 (en) | 2014-03-24 | 2019-08-30 | Novartis Ag | Monobactam organic compounds for the treatment of bacterial infections |
| SG10202109752XA (en) | 2014-04-07 | 2021-10-28 | Novartis Ag | Treatment of cancer using anti-cd19 chimeric antigen receptor |
| US10682400B2 (en) * | 2014-04-30 | 2020-06-16 | President And Fellows Of Harvard College | Combination vaccine devices and methods of killing cancer cells |
| KR20230144099A (en) | 2014-05-15 | 2023-10-13 | 브리스톨-마이어스 스큅 컴퍼니 | Treatment of lung cancer using a combination of an anti-pd-1 antibody and another anti-cancer agent |
| CN106572993B (en) | 2014-05-23 | 2019-07-16 | 卫材R&D管理有限公司 | Application of the EP4 antagonist in the drug of preparation treating cancer |
| SG11201609721WA (en) | 2014-05-28 | 2016-12-29 | Agenus Inc | Anti-gitr antibodies and methods of use thereof |
| US10293043B2 (en) | 2014-06-02 | 2019-05-21 | Armo Biosciences, Inc. | Methods of lowering serum cholesterol |
| EA037006B1 (en) | 2014-06-06 | 2021-01-26 | Бристол-Майерс Сквибб Компани | Antibodies against glucocorticoid-induced tumor necrosis factor receptor (gitr) and uses thereof |
| WO2015191881A2 (en) | 2014-06-11 | 2015-12-17 | Green Kathy A | Use of vista agonists and antagonists to suppress or enhance humoral immunity |
| US10092645B2 (en) | 2014-06-17 | 2018-10-09 | Medimmune Limited | Methods of treatment with antagonists against PD-1 and PD-L1 in combination with radiation therapy |
| KR102130600B1 (en) | 2014-07-03 | 2020-07-08 | 베이진 엘티디 | Anti-PD-L1 Antibodies and Their Use as Therapeutics and Diagnostics |
| WO2016014576A1 (en) | 2014-07-21 | 2016-01-28 | Novartis Ag | Treatment of cancer using a cd33 chimeric antigen receptor |
| EP3193915A1 (en) | 2014-07-21 | 2017-07-26 | Novartis AG | Combinations of low, immune enhancing. doses of mtor inhibitors and cars |
| US11542488B2 (en) | 2014-07-21 | 2023-01-03 | Novartis Ag | Sortase synthesized chimeric antigen receptors |
| RU2711141C2 (en) | 2014-07-22 | 2020-01-15 | СиБи ТЕРЕПЬЮТИКС, ИНК. | Anti-pd-1 antibodies |
| CN105330740B (en) * | 2014-07-30 | 2018-08-17 | 珠海市丽珠单抗生物技术有限公司 | Anti- PD-1 antibody and its application |
| EP4205749A1 (en) | 2014-07-31 | 2023-07-05 | Novartis AG | Subset-optimized chimeric antigen receptor-containing cells |
| US9982052B2 (en) | 2014-08-05 | 2018-05-29 | MabQuest, SA | Immunological reagents |
| SG10201901057UA (en) | 2014-08-05 | 2019-03-28 | Cb Therapeutics Inc | Anti-pd-l1 antibodies |
| WO2016020836A1 (en) | 2014-08-06 | 2016-02-11 | Novartis Ag | Quinolone derivatives as antibacterials |
| JP6919118B2 (en) | 2014-08-14 | 2021-08-18 | ノバルティス アーゲー | Treatment of cancer with GFRα-4 chimeric antigen receptor |
| RU2724999C2 (en) | 2014-08-19 | 2020-06-29 | Новартис Аг | Chimeric antigenic (car) receptor against cd123 for use in treating malignant tumors |
| US9535074B2 (en) | 2014-09-08 | 2017-01-03 | Merck Sharp & Dohme Corp. | Immunoassay for soluble PD-L1 |
| CN113929782B (en) * | 2014-09-16 | 2024-06-21 | 依奈特制药公司 | Neutralization of inhibitory pathways in lymphocytes |
| WO2016044605A1 (en) | 2014-09-17 | 2016-03-24 | Beatty, Gregory | Targeting cytotoxic cells with chimeric receptors for adoptive immunotherapy |
| US10053683B2 (en) | 2014-10-03 | 2018-08-21 | Whitehead Institute For Biomedical Research | Intercellular labeling of ligand-receptor interactions |
| AU2015327868A1 (en) | 2014-10-03 | 2017-04-20 | Novartis Ag | Combination therapies |
| WO2016057705A1 (en) | 2014-10-08 | 2016-04-14 | Novartis Ag | Biomarkers predictive of therapeutic responsiveness to chimeric antigen receptor therapy and uses thereof |
| MA41044A (en) | 2014-10-08 | 2017-08-15 | Novartis Ag | COMPOSITIONS AND METHODS OF USE FOR INCREASED IMMUNE RESPONSE AND CANCER TREATMENT |
| US9732119B2 (en) | 2014-10-10 | 2017-08-15 | Bristol-Myers Squibb Company | Immunomodulators |
| US10350270B2 (en) | 2014-10-14 | 2019-07-16 | Armo Biosciences, Inc. | Interleukin-15 compositions and uses thereof |
| CA2963995A1 (en) | 2014-10-22 | 2016-04-28 | Armo Biosciences, Inc. | Methods of using interleukin-10 for treating diseases and disorders |
| WO2016069727A1 (en) | 2014-10-29 | 2016-05-06 | Five Prime Therapeutics, Inc. | Combination therapy for cancer |
| EP3215182B1 (en) | 2014-11-05 | 2023-01-04 | The Regents of The University of California | Combination immunotherapy |
| US9856292B2 (en) | 2014-11-14 | 2018-01-02 | Bristol-Myers Squibb Company | Immunomodulators |
| KR20170084202A (en) | 2014-11-14 | 2017-07-19 | 노파르티스 아게 | Antibody drug conjugates |
| ES2905615T3 (en) | 2014-11-20 | 2022-04-11 | Promega Corp | Systems and Methods for Assessing Immune Checkpoint Modulators |
| US20180334490A1 (en) | 2014-12-03 | 2018-11-22 | Qilong H. Wu | Methods for b cell preconditioning in car therapy |
| JP2017537927A (en) | 2014-12-04 | 2017-12-21 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Combination of anti-CS1 and anti-PD1 antibodies to treat cancer (myeloma) |
| CN107405398A (en) | 2014-12-05 | 2017-11-28 | 伊穆奈克斯特股份有限公司 | VSIG8 is identified as presumption VISTA acceptors and its to produce the purposes of VISTA/VSIG8 activators and antagonist |
| TWI595006B (en) | 2014-12-09 | 2017-08-11 | 禮納特神經系統科學公司 | Anti-pd-1 antibodies and methods of use thereof |
| BR112017012222A2 (en) | 2014-12-09 | 2018-01-30 | Merck Sharp & Dohme | methods for deriving a gene signature biomarker and for treating a patient having a tumor, method and system for testing a tumor sample removed from a patient, and kit. |
| RU2711729C2 (en) | 2014-12-09 | 2020-01-21 | Регенерон Фармасьютикалз, Инк. | Non-human animals having humanized gene of differentiation cluster 274 |
| PL3233843T3 (en) | 2014-12-16 | 2020-04-30 | Novartis Ag | Isoxazole hydroxamic acid compounds as lpxc inhibitors |
| US9861680B2 (en) | 2014-12-18 | 2018-01-09 | Bristol-Myers Squibb Company | Immunomodulators |
| KR102549746B1 (en) | 2014-12-18 | 2023-07-03 | 암젠 인크 | Stable frozen herpes simplex virus formulation |
| WO2016100882A1 (en) | 2014-12-19 | 2016-06-23 | Novartis Ag | Combination therapies |
| WO2016100975A1 (en) | 2014-12-19 | 2016-06-23 | Massachsetts Institute Ot Technology | Molecular biomarkers for cancer immunotherapy |
| US9944678B2 (en) | 2014-12-19 | 2018-04-17 | Bristol-Myers Squibb Company | Immunomodulators |
| WO2016109310A1 (en) | 2014-12-31 | 2016-07-07 | Checkmate Pharmaceuticals, Llc | Combination tumor immunotherapy |
| CN107428818A (en) | 2015-01-29 | 2017-12-01 | 密西根州立大学校董会 | Hide polypeptide and application thereof |
| US11786457B2 (en) | 2015-01-30 | 2023-10-17 | President And Fellows Of Harvard College | Peritumoral and intratumoral materials for cancer therapy |
| US11161907B2 (en) | 2015-02-02 | 2021-11-02 | Novartis Ag | Car-expressing cells against multiple tumor antigens and uses thereof |
| WO2016126615A1 (en) | 2015-02-03 | 2016-08-11 | Armo Biosciences, Inc. | Methods of using interleukin-10 for treating diseases and disorders |
| US20160222060A1 (en) | 2015-02-04 | 2016-08-04 | Bristol-Myers Squibb Company | Immunomodulators |
| MA41551A (en) | 2015-02-20 | 2017-12-26 | Incyte Corp | BICYCLIC HETEROCYCLES USED AS FGFR4 INHIBITORS |
| WO2016134320A1 (en) | 2015-02-20 | 2016-08-25 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| US10238650B2 (en) | 2015-03-06 | 2019-03-26 | Beyondspring Pharmaceuticals, Inc. | Method of treating cancer associated with a RAS mutation |
| US10076518B2 (en) | 2015-03-06 | 2018-09-18 | Beyondspring Pharmaceuticals, Inc. | Method of treating a brain tumor |
| TN2017000375A1 (en) | 2015-03-10 | 2019-01-16 | Aduro Biotech Inc | Compositions and methods for activating "stimulator of interferon gene" -dependent signalling |
| US9809625B2 (en) | 2015-03-18 | 2017-11-07 | Bristol-Myers Squibb Company | Immunomodulators |
| CN107530428B (en) | 2015-03-23 | 2022-05-13 | 震动疗法股份有限公司 | Antibodies to ICOS |
| US10836827B2 (en) * | 2015-03-30 | 2020-11-17 | Stcube, Inc. | Antibodies specific to glycosylated PD-L1 and methods of use thereof |
| WO2016161410A2 (en) | 2015-04-03 | 2016-10-06 | Xoma Technology Ltd. | Treatment of cancer using inhibitors of tgf-beta and pd-1 |
| US20180140602A1 (en) | 2015-04-07 | 2018-05-24 | Novartis Ag | Combination of chimeric antigen receptor therapy and amino pyrimidine derivatives |
| WO2016164705A1 (en) | 2015-04-10 | 2016-10-13 | Omar Abdel-Rahman Ali | Immune cell trapping devices and methods for making and using the same |
| AU2016249005B2 (en) | 2015-04-17 | 2022-06-16 | Novartis Ag | Methods for improving the efficacy and expansion of chimeric antigen receptor-expressing cells |
| AU2016249395B2 (en) | 2015-04-17 | 2022-04-07 | Bristol-Myers Squibb Company | Compositions comprising a combination of an anti-PD-1 antibody and another antibody |
| EP3839510A3 (en) | 2015-04-17 | 2021-08-25 | Merck Sharp & Dohme Corp. | Blood-based biomarkers of tumor sensitivity to pd-1 antagonists |
| US9981239B2 (en) | 2015-04-21 | 2018-05-29 | Twist Bioscience Corporation | Devices and methods for oligonucleic acid library synthesis |
| US20180298068A1 (en) | 2015-04-23 | 2018-10-18 | Novartis Ag | Treatment of cancer using chimeric antigen receptor and protein kinase a blocker |
| US20160362489A1 (en) | 2015-04-28 | 2016-12-15 | Bristol-Myers Squibb Company | Treatment of PD-L1-Positive Melanoma Using an Anti-PD-1 Antibody |
| JP2018514550A (en) | 2015-04-28 | 2018-06-07 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Treatment of PD-L1 negative melanoma using anti-PD-1 antibody and anti-CTLA-4 antibody |
| US20180104331A1 (en) | 2015-05-11 | 2018-04-19 | The Johns Hopkins University | Autoimmune antibodies for use in inhibiting cancer cell growth |
| EP3297672B1 (en) | 2015-05-21 | 2021-09-01 | Harpoon Therapeutics, Inc. | Trispecific binding proteins and methods of use |
| CN107847583A (en) | 2015-05-28 | 2018-03-27 | 阿尔莫生物科技股份有限公司 | PEGylated Interleukin 10 for treating cancer |
| US20180155429A1 (en) * | 2015-05-28 | 2018-06-07 | Bristol-Myers Squibb Company | Treatment of pd-l1 positive lung cancer using an anti-pd-1 antibody |
| US11078278B2 (en) | 2015-05-29 | 2021-08-03 | Bristol-Myers Squibb Company | Treatment of renal cell carcinoma |
| MA53355A (en) | 2015-05-29 | 2022-03-16 | Agenus Inc | ANTI-CTLA-4 ANTIBODIES AND METHODS OF USE THEREOF |
| MX2017015811A (en) | 2015-06-12 | 2018-04-10 | Squibb Bristol Myers Co | Treatment of cancer by combined blockade of the pd-1 and cxcr4 signaling pathways. |
| EP3916018A1 (en) * | 2015-06-16 | 2021-12-01 | Genentech, Inc. | Anti-cd3 antibodies and methods of use |
| EP3310813A1 (en) | 2015-06-17 | 2018-04-25 | Novartis AG | Antibody drug conjugates |
| DK3313882T3 (en) | 2015-06-24 | 2020-05-11 | Janssen Pharmaceutica Nv | Anti-VISTA antibodies and fragments |
| EA201890199A1 (en) | 2015-07-02 | 2018-06-29 | Селджин Корпорейшн | COMBINED THERAPY FOR THE TREATMENT OF HEMOBLASTOSIS AND SOLID TUMORS |
| AU2016291708B2 (en) | 2015-07-13 | 2020-12-24 | Beyondspring Pharmaceuticals, Inc | Plinabulin compositions |
| SG10201913303XA (en) | 2015-07-13 | 2020-03-30 | Cytomx Therapeutics Inc | Anti-pd-1 antibodies, activatable anti-pd-1 antibodies, and methods of use thereof |
| EP3858859A1 (en) | 2015-07-14 | 2021-08-04 | Bristol-Myers Squibb Company | Method of treating cancer using immune checkpoint inhibitor; antibody that binds to programmed death-1 receptor (pd-1) or programmed death ligand 1 (pd-l1) |
| CN108136025B (en) | 2015-07-16 | 2022-09-06 | 比奥克斯塞尔医疗股份有限公司 | A novel method of treating cancer using immunomodulation |
| CN109476722A (en) | 2015-07-21 | 2019-03-15 | 诺华股份有限公司 | The method of the effect of for improving immunocyte and expansion |
| PT3317301T (en) | 2015-07-29 | 2021-07-09 | Novartis Ag | Combination therapies comprising antibody molecules to lag-3 |
| CN108136003A (en) | 2015-07-29 | 2018-06-08 | 诺华股份有限公司 | The use in conjunction of anti-PD-1 and anti-M-CSF antibody in treatment of cancer |
| BR112018001640A2 (en) | 2015-07-29 | 2018-09-18 | Novartis Ag | combination of pd-1 antagonist with an egfr inhibitor |
| WO2017019896A1 (en) | 2015-07-29 | 2017-02-02 | Novartis Ag | Combination therapies comprising antibody molecules to pd-1 |
| US20180207273A1 (en) | 2015-07-29 | 2018-07-26 | Novartis Ag | Combination therapies comprising antibody molecules to tim-3 |
| US20180222989A1 (en) | 2015-08-04 | 2018-08-09 | Glaxosmithkline Intellectual Property Development Limited | Combination treatments and uses and methods thereof |
| KR20180036996A (en) | 2015-08-04 | 2018-04-10 | 글락소스미스클라인 인털렉츄얼 프로퍼티 디벨로프먼트 리미티드 | Combination therapy and its uses and methods |
| EP3331901A1 (en) | 2015-08-07 | 2018-06-13 | Pieris Pharmaceuticals GmbH | Novel fusion polypeptide specific for lag-3 and pd-1 |
| US20180230431A1 (en) | 2015-08-07 | 2018-08-16 | Glaxosmithkline Intellectual Property Development Limited | Combination Therapy |
| CN108025040A (en) | 2015-08-25 | 2018-05-11 | 阿尔莫生物科技股份有限公司 | The method that disease and illness are treated using interleukin-10 |
| EP3341732B1 (en) | 2015-08-27 | 2023-07-12 | INSERM - Institut National de la Santé et de la Recherche Médicale | Methods for predicting the survival time of patients suffering from a lung cancer |
| CN114605548A (en) | 2015-09-01 | 2022-06-10 | 艾吉纳斯公司 | anti-PD-1 antibodies and methods of use thereof |
| ES2842306T3 (en) | 2015-09-02 | 2021-07-13 | Immutep Sas | Anti LAG-3 antibodies |
| US11747346B2 (en) | 2015-09-03 | 2023-09-05 | Novartis Ag | Biomarkers predictive of cytokine release syndrome |
| CN108348571B (en) | 2015-09-03 | 2022-03-22 | 艾瑞朗医疗公司 | Peptidomimetic macrocycles and uses thereof |
| EP3350314A4 (en) | 2015-09-18 | 2019-02-06 | Twist Bioscience Corporation | Oligonucleic acid variant libraries and synthesis thereof |
| KR20180058772A (en) | 2015-09-22 | 2018-06-01 | 트위스트 바이오사이언스 코포레이션 | Flexible substrate for nucleic acid synthesis |
| WO2017055321A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of fibroblasts in a tissue sample |
| WO2017055322A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of neutrophils in a tissue sample |
| WO2017055326A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of myeloid dendritic cells in a tissue sample |
| WO2017055325A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of nk cells in a tissue sample |
| WO2017055320A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of cytotoxic lymphocytes in a tissue sample |
| WO2017055319A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of b cells in a tissue sample |
| KR20180054824A (en) | 2015-09-29 | 2018-05-24 | 셀진 코포레이션 | PD-1 binding protein and method of use thereof |
| WO2017055324A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of cells of monocytic origin in a tissue sample |
| WO2017055327A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of endothelial cells in a tissue sample |
| ES2941968T3 (en) | 2015-10-01 | 2023-05-29 | The Whitehead Institute For Biomedical Res | Antibody labeling |
| WO2017059224A2 (en) | 2015-10-01 | 2017-04-06 | Gilead Sciences, Inc. | Combination of a btk inhibitor and a checkpoint inhibitor for treating cancers |
| SI3356404T1 (en) | 2015-10-02 | 2021-11-30 | F. Hoffmann-La Roche Ag | Anti-pd1 antibodies and methods of use |
| WO2017060397A1 (en) | 2015-10-09 | 2017-04-13 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for predicting the survival time of subjects suffering from melanoma metastases |
| EP3362074B1 (en) | 2015-10-16 | 2023-08-09 | President and Fellows of Harvard College | Regulatory t cell pd-1 modulation for regulating t cell effector immune responses |
| CN108135934B (en) | 2015-10-19 | 2024-09-10 | 永恒生物科技股份有限公司 | Methods of treating solid or lymphoid tumors by combination therapy |
| AU2016342269A1 (en) | 2015-10-22 | 2018-03-29 | Jounce Therapeutics, Inc. | Gene signatures for determining icos expression |
| CN105238762A (en) * | 2015-10-26 | 2016-01-13 | 无锡傲锐东源生物科技有限公司 | Anti-PD-1 protein monoclonal antibody hybridomas cell, anti- PD-1 monoclonal antibody generated by same and application |
| MA44334A (en) | 2015-10-29 | 2018-09-05 | Novartis Ag | ANTIBODY CONJUGATES INCLUDING A TOLL-TYPE RECEPTOR AGONIST |
| EA201891106A1 (en) | 2015-11-02 | 2018-12-28 | Файв Прайм Терапьютикс, Инк. | POLYPEPTIDES OF CD80 OUT-CELL DOMAIN AND THEIR APPLICATION IN CANCER TREATMENT |
| WO2017077382A1 (en) | 2015-11-06 | 2017-05-11 | Orionis Biosciences Nv | Bi-functional chimeric proteins and uses thereof |
| TWI795347B (en) | 2015-11-18 | 2023-03-11 | 美商必治妥施貴寶公司 | Treatment of lung cancer using a combination of an anti-pd-1 antibody and an anti-ctla-4 antibody |
| EP3380523A1 (en) | 2015-11-23 | 2018-10-03 | Five Prime Therapeutics, Inc. | Fgfr2 inhibitors alone or in combination with immune stimulating agents in cancer treatment |
| IL299072A (en) | 2015-12-02 | 2023-02-01 | Memorial Sloan Kettering Cancer Center | Antibodies and methods of use thereof |
| JP7003036B2 (en) * | 2015-12-02 | 2022-02-04 | エスティーキューブ,インコーポレイテッド | Antibodies specific for glycosylated PD-1 and how to use them |
| CN105837692A (en) * | 2015-12-10 | 2016-08-10 | 苏州佰通生物科技有限公司 | Chimeric antigen receptor for blocking immunodetection point and use thereof |
| EP4374926A3 (en) | 2015-12-15 | 2024-08-07 | OncoC4, Inc. | Chimeric and humanized anti-human ctla4 monoclonal antibodies and uses thereof |
| AU2016369537B2 (en) | 2015-12-17 | 2024-03-14 | Novartis Ag | Antibody molecules to PD-1 and uses thereof |
| US10392442B2 (en) | 2015-12-17 | 2019-08-27 | Bristol-Myers Squibb Company | Use of anti-PD-1 antibody in combination with anti-CD27 antibody in cancer treatment |
| KR20180089510A (en) | 2015-12-18 | 2018-08-08 | 노파르티스 아게 | Antibodies targeting CD32b and methods of using the same |
| MA44140A (en) | 2015-12-22 | 2021-05-19 | Dana Farber Cancer Inst Inc | CHEMERICAL ANTIGEN RECEPTOR (CAR) AGAINST MESOTHELIN AND ANTIBODY AGAINST PD-L1 INHIBITOR FOR COMBINED USE IN ANTI-CANCER THERAPY |
| PE20230731A1 (en) | 2015-12-22 | 2023-05-03 | Incyte Corp | HETEROCYCLIC COMPOUNDS AS IMMUNOMODULATORS |
| EA037508B1 (en) | 2016-01-08 | 2021-04-06 | Селджин Корпорейшн | Formulations of 2-(4-chlorophenyl)-n-((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide |
| CN108712904B (en) | 2016-01-08 | 2022-08-02 | 细胞基因公司 | Solid forms of 2- (4-chlorophenyl) -N- ((2- (2, 6-dioxopiperidin-3-yl) -1-oxoisoindolin-5-yl) methyl) -2, 2-difluoroacetamide and pharmaceutical compositions and uses thereof |
| ES2832475T3 (en) | 2016-01-08 | 2021-06-10 | Celgene Corp | Antiproliferative Compounds and Their Pharmaceutical Compositions and Uses |
| BR112018014150A2 (en) | 2016-01-11 | 2018-12-11 | Novartis Ag | immunostimulating humanized monoclonal antibodies to human interleukin-2, and fusion proteins thereof |
| EP3964529A1 (en) | 2016-01-22 | 2022-03-09 | Mabquest SA | Non-blocking pd1 specific antibodies |
| US11214617B2 (en) | 2016-01-22 | 2022-01-04 | MabQuest SA | Immunological reagents |
| JP6970099B2 (en) | 2016-01-28 | 2021-11-24 | アンスティチュ ナショナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシュ メディカル | Methods and pharmaceutical compositions for the treatment of cancer |
| WO2017129763A1 (en) | 2016-01-28 | 2017-08-03 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for the treatment of signet ring cell gastric cancer |
| EP3407912B1 (en) | 2016-01-28 | 2022-05-18 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for enhancing the potency of the immune checkpoint inhibitors |
| WO2017133175A1 (en) * | 2016-02-04 | 2017-08-10 | Nanjing Legend Biotech Co., Ltd. | Engineered mammalian cells for cancer therapy |
| DK3411398T3 (en) | 2016-02-05 | 2024-06-24 | Orionis Biosciences BV | TARGETED THERAPEUTICS AND THEIR USE |
| CN109072197A (en) | 2016-02-06 | 2018-12-21 | 哈佛学院校长同事会 | It is immune to rebuild to remold hematopoiesis nest |
| SG11201806583XA (en) | 2016-02-08 | 2018-09-27 | Beyondspring Pharmaceuticals Inc | Compositions containing tucaresol or its analogs |
| CA3014013A1 (en) | 2016-02-12 | 2017-08-17 | Janssen Pharmaceutica Nv | Anti-vista (b7h5) antibodies |
| RU2018131123A (en) | 2016-02-17 | 2020-03-17 | Новартис Аг | ANTIBODIES TO TGF-BETA2 |
| WO2017140821A1 (en) | 2016-02-19 | 2017-08-24 | Novartis Ag | Tetracyclic pyridone compounds as antivirals |
| BR112018067525A2 (en) | 2016-02-26 | 2019-02-05 | Centre Nat Rech Scient | antibodies having specificity for btla and their uses |
| US11725247B2 (en) | 2016-02-29 | 2023-08-15 | Foundation Medicine, Inc. | Methods of treating cancer |
| KR20180118175A (en) | 2016-03-04 | 2018-10-30 | 노파르티스 아게 | Cells expressing multiple chimeric antigen receptor (CAR) molecules and their uses |
| US10143746B2 (en) | 2016-03-04 | 2018-12-04 | Bristol-Myers Squibb Company | Immunomodulators |
| WO2017156349A1 (en) | 2016-03-10 | 2017-09-14 | Cold Genesys, Inc. | Methods of treating solid or lymphatic tumors by combination therapy |
| US20210309965A1 (en) | 2016-03-21 | 2021-10-07 | Dana-Farber Cancer Institute, Inc. | T-cell exhaustion state-specific gene expression regulators and uses thereof |
| EP3432924A1 (en) | 2016-03-23 | 2019-01-30 | Novartis AG | Cell secreted minibodies and uses thereof |
| US11760803B2 (en) | 2016-03-24 | 2023-09-19 | Takeda Pharmaceutical Company Limited | Methods of treating gastrointestinal immune-related adverse events in immune oncology treatments |
| WO2017165742A1 (en) | 2016-03-24 | 2017-09-28 | Millennium Pharmaceuticals, Inc. | Methods of treating gastrointestinal immune-related adverse events in anti-ctla4 anti-pd-1 combination treatments |
| EP3433257B1 (en) | 2016-03-24 | 2023-10-11 | Novartis AG | Alkynyl nucleoside analogs as inhibitors of human rhinovirus |
| EP3225253A1 (en) | 2016-04-01 | 2017-10-04 | Deutsches Krebsforschungszentrum Stiftung des Öffentlichen Rechts | Cancer therapy with an oncolytic virus combined with a checkpoint inhibitor |
| EP3436066A1 (en) | 2016-04-01 | 2019-02-06 | Checkmate Pharmaceuticals, Inc. | Fc receptor-mediated drug delivery |
| WO2017176925A1 (en) | 2016-04-05 | 2017-10-12 | Bristol-Myers Squibb Company | Cytokine profiling analysis for predicting prognosis of a patient in need of an anti-cancer treatment |
| US10358463B2 (en) | 2016-04-05 | 2019-07-23 | Bristol-Myers Squibb Company | Immunomodulators |
| IL295538B2 (en) | 2016-04-13 | 2024-02-01 | Vivia Biotech Sl | Ex vivo bite-activated t cells |
| CR20180537A (en) | 2016-04-15 | 2019-03-04 | Immunext Inc | ANTIHUMAN VIEW ANTIBODIES AND THEIR USE |
| US20190298824A1 (en) | 2016-05-04 | 2019-10-03 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Serv | Albumin-binding immunomodulatory compositions and methods of use thereof |
| CN109563141A (en) | 2016-05-13 | 2019-04-02 | 奥里尼斯生物科学公司 | To the therapeutic targeting of cellular structures |
| CA3023883A1 (en) | 2016-05-13 | 2017-11-16 | Orionis Biosciences Nv | Targeted mutant interferon-beta and uses thereof |
| SG11201810268YA (en) | 2016-05-20 | 2018-12-28 | Lilly Co Eli | Combination therapy with notch and pd-1 or pd-l1 inhibitors |
| US11623958B2 (en) | 2016-05-20 | 2023-04-11 | Harpoon Therapeutics, Inc. | Single chain variable fragment CD3 binding proteins |
| CN105968200B (en) | 2016-05-20 | 2019-03-15 | 瑞阳(苏州)生物科技有限公司 | Anti human PD-L 1 Humanized monoclonal antibodies and its application |
| US20190292259A1 (en) | 2016-05-24 | 2019-09-26 | Inserm (Institut National De La Sante Et De La Recherche Medicale) | Methods and pharmaceutical compositions for the treatment of non small cell lung cancer (nsclc) that coexists with chronic obstructive pulmonary disease (copd) |
| CN106008714B (en) * | 2016-05-24 | 2019-03-15 | 瑞阳(苏州)生物科技有限公司 | Anti-human PD-1 Humanized monoclonal antibodies and its application |
| BR112018074192A8 (en) | 2016-05-25 | 2022-11-08 | Inst Nat Sante Rech Med | METHOD FOR TREATMENT OF A SUBJECT, METHOD FOR PRODUCING A CELL COMPOSITION, CELL COMPOSITION, VACCINE COMPOSITION AND KIT |
| CN109476751B (en) | 2016-05-27 | 2024-04-19 | 艾吉纳斯公司 | Anti-TIM-3 antibodies and methods of use thereof |
| WO2017210453A1 (en) | 2016-06-02 | 2017-12-07 | Bristol-Myers Squibb Company | Pd-1 blockade with nivolumab in refractory hodgkin's lymphoma |
| WO2017207814A1 (en) | 2016-06-02 | 2017-12-07 | Ultimovacs As | A vaccine in combination with an immune checkpoint inhibitor for use in treating cancer |
| HUE063911T2 (en) * | 2016-06-02 | 2024-02-28 | Bristol Myers Squibb Co | Use of an anti-pd-1 antibody in combination with an anti-cd30 antibody in lymphoma treatment |
| KR102515509B1 (en) | 2016-06-03 | 2023-03-28 | 브리스톨-마이어스 스큅 컴퍼니 | Use of Anti-PD-1 Antibodies in the Treatment of Patients with Colorectal Cancer |
| CN109476753A (en) | 2016-06-03 | 2019-03-15 | 百时美施贵宝公司 | For treating the anti-PD-1 antibody of the method for tumour |
| WO2017210631A1 (en) | 2016-06-03 | 2017-12-07 | Bristol-Myers Squibb Company | Anti-pd-1 antibody for use in a method of treatment of recurrent small cell lung cancer |
| CN109475524A (en) | 2016-06-06 | 2019-03-15 | 万春药业公司 | For reducing the composition and method of neutrophilic granulocytopenia |
| JP7185530B2 (en) | 2016-06-13 | 2022-12-07 | トルク セラピューティクス, インコーポレイテッド | Methods and compositions for promoting immune cell function |
| CA3026151A1 (en) * | 2016-06-14 | 2017-12-21 | Xencor, Inc. | Bispecific checkpoint inhibitor antibodies |
| US10071973B2 (en) | 2016-06-14 | 2018-09-11 | Novartis Ag | Crystalline isoxazole hydroxamic acid compounds |
| WO2017216686A1 (en) | 2016-06-16 | 2017-12-21 | Novartis Ag | 8,9-fused 2-oxo-6,7-dihydropyrido-isoquinoline compounds as antivirals |
| WO2017216685A1 (en) | 2016-06-16 | 2017-12-21 | Novartis Ag | Pentacyclic pyridone compounds as antivirals |
| TWI771305B (en) | 2016-06-20 | 2022-07-21 | 美商英塞特公司 | Heterocyclic compounds as immunomodulators |
| SG10201912456RA (en) | 2016-06-24 | 2020-02-27 | Infinity Pharmaceuticals Inc | Combination therapies |
| WO2018009466A1 (en) | 2016-07-05 | 2018-01-11 | Aduro Biotech, Inc. | Locked nucleic acid cyclic dinucleotide compounds and uses thereof |
| CN109475536B (en) | 2016-07-05 | 2022-05-27 | 百济神州有限公司 | Combination of a PD-l antagonist and a RAF inhibitor for the treatment of cancer |
| WO2018011166A2 (en) | 2016-07-12 | 2018-01-18 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for quantifying the population of myeloid dendritic cells in a tissue sample |
| SG11201900157RA (en) | 2016-07-12 | 2019-02-27 | Revolution Medicines Inc | 2,5-disubstituted 3-methyl pyrazines and 2,5,6-trisubstituted 3-methyl pyrazines as allosteric shp2 inhibitors |
| US11555177B2 (en) | 2016-07-13 | 2023-01-17 | President And Fellows Of Harvard College | Antigen-presenting cell-mimetic scaffolds and methods for making and using the same |
| CA3031542A1 (en) | 2016-07-20 | 2018-01-25 | University Of Utah Research Foundation | Cd229 car t cells and methods of use thereof |
| CN118021943A (en) | 2016-07-28 | 2024-05-14 | 诺华股份有限公司 | Combination therapy of chimeric antigen receptor and PD-1 inhibitor |
| WO2018022438A1 (en) | 2016-07-29 | 2018-02-01 | Eli Lilly And Company | Combination therapy with merestinib and anti-pd-l1 or anti-pd-1 inhibitors for use in the treatment of cancer |
| EP3490548A4 (en) | 2016-08-01 | 2020-04-15 | Molecular Templates, Inc. | Administration of hypoxia activated prodrugs in combination with immune modulatory agents for treating cancer |
| US11649289B2 (en) | 2016-08-04 | 2023-05-16 | Glaxosmithkline Intellectual Property Development Limited | Anti-ICOS and anti-PD-1 antibody combination therapy |
| WO2018029336A1 (en) | 2016-08-12 | 2018-02-15 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for determining whether a subject was administered with an activator of the ppar beta/delta pathway. |
| CN118252927A (en) | 2016-08-19 | 2024-06-28 | 百济神州有限公司 | Treatment of cancer using combination products comprising BTK inhibitors |
| CA3034769A1 (en) | 2016-08-22 | 2018-03-01 | Twist Bioscience Corporation | De novo synthesized nucleic acid libraries |
| WO2018035710A1 (en) | 2016-08-23 | 2018-03-01 | Akeso Biopharma, Inc. | Anti-ctla4 antibodies |
| CN106967172B (en) | 2016-08-23 | 2019-01-08 | 康方药业有限公司 | The anti-PD-1 bifunctional antibody of anti-CTLA 4-, its medical composition and its use |
| CN106977602B (en) | 2016-08-23 | 2018-09-25 | 中山康方生物医药有限公司 | A kind of anti-PD1 monoclonal antibodies, its medical composition and its use |
| CN110121352B (en) | 2016-09-01 | 2020-12-11 | 嵌合体生物工程公司 | GOLD-optimized CAR T-cells |
| AU2017322501A1 (en) | 2016-09-09 | 2019-03-28 | Laboratoire Francais Du Fractionnement Et Des Biotechnologies | Combination of an anti-CD20 antibody, PI3 kinase-delta inhibitor, and anti-PD-1 or anti-PD-L1 antibody for treating hematological cancers |
| WO2018048975A1 (en) | 2016-09-09 | 2018-03-15 | Bristol-Myers Squibb Company | Use of an anti-pd-1 antibody in combination with an anti-mesothelin antibody in cancer treatment |
| TW201811788A (en) | 2016-09-09 | 2018-04-01 | 瑞士商諾華公司 | Polycyclic pyridone compounds as antivirals |
| WO2018046738A1 (en) | 2016-09-12 | 2018-03-15 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for predicting the survival time of patients suffering from cancer |
| WO2018046736A1 (en) | 2016-09-12 | 2018-03-15 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for predicting the survival time of patients suffering from cancer |
| US10766958B2 (en) | 2016-09-19 | 2020-09-08 | Celgene Corporation | Methods of treating vitiligo using PD-1 binding antibodies |
| BR112019004733A2 (en) | 2016-09-19 | 2019-05-28 | Celgene Corp | Methods of treating immune disorders using pd-1 binding proteins |
| EP4360714A3 (en) | 2016-09-21 | 2024-07-24 | Nextcure, Inc. | Antibodies for siglec-15 and methods of use thereof |
| KR102644544B1 (en) | 2016-09-21 | 2024-03-11 | 넥스트큐어 인코포레이티드 | Antibodies for SIGLEC-15 and methods of using the same |
| US10417457B2 (en) | 2016-09-21 | 2019-09-17 | Twist Bioscience Corporation | Nucleic acid based data storage |
| EP3515475B1 (en) | 2016-09-21 | 2024-05-01 | The U.S.A. as represented by the Secretary, Department of Health and Human Services | Chimeric antigen receptor (car) that targets chemokine receptor ccr4 and its use |
| WO2018055080A1 (en) | 2016-09-22 | 2018-03-29 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for reprograming immune environment in a subject in need thereof |
| WO2018057955A1 (en) | 2016-09-23 | 2018-03-29 | Elstar Therapeutics, Inc. | Multispecific antibody molecules comprising lambda and kappa light chains |
| JOP20190061A1 (en) | 2016-09-28 | 2019-03-26 | Novartis Ag | Beta-lactamase inhibitors |
| TW202340473A (en) | 2016-10-07 | 2023-10-16 | 瑞士商諾華公司 | Treatment of cancer using chimeric antigen receptors |
| WO2018069927A1 (en) | 2016-10-10 | 2018-04-19 | The National Institute for Biotechnology in the Negev Ltd. | Non-cytotoxic modified cells and use thereof |
| AU2017343621B2 (en) | 2016-10-11 | 2021-12-02 | Agenus Inc. | Anti-LAG-3 antibodies and methods of use thereof |
| WO2018071576A1 (en) | 2016-10-14 | 2018-04-19 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Treatment of tumors by inhibition of cd300f |
| WO2018073753A1 (en) | 2016-10-18 | 2018-04-26 | Novartis Ag | Fused tetracyclic pyridone compounds as antivirals |
| WO2018075447A1 (en) | 2016-10-19 | 2018-04-26 | The Trustees Of Columbia University In The City Of New York | Combination of braf inhibitor, talimogene laherparepvec, and immune checkpoint inhibitor for use in the treatment cancer (melanoma) |
| US11084859B2 (en) | 2016-10-24 | 2021-08-10 | Orionis Biosciences BV | Targeted mutant interferon-gamma and uses thereof |
| EP3532504A1 (en) | 2016-10-28 | 2019-09-04 | Bristol-Myers Squibb Company | Methods of treating urothelial carcinoma using an anti-pd-1 antibody |
| KR102687833B1 (en) | 2016-11-02 | 2024-07-24 | 브리스톨-마이어스 스큅 컴퍼니 | Combination use of dual specific antibodies and immunological drugs against BCMA and CD3 for the treatment of multiple myeloma |
| WO2018083087A2 (en) | 2016-11-02 | 2018-05-11 | Glaxosmithkline Intellectual Property (No.2) Limited | Binding proteins |
| WO2018085555A1 (en) | 2016-11-03 | 2018-05-11 | Bristol-Myers Squibb Company | Activatable anti-ctla-4 antibodies and uses thereof |
| US10342785B2 (en) | 2016-11-04 | 2019-07-09 | Askat Inc. | Use of EP4 receptor antagonists for the treatment of NASH-associated liver cancer |
| WO2018085750A2 (en) | 2016-11-07 | 2018-05-11 | Bristol-Myers Squibb Company | Immunomodulators |
| IT201600111877A1 (en) * | 2016-11-07 | 2018-05-07 | Biouniversa Srl | Anti-BAG3 antibodies in combination with inhibitors of immune check-point for therapeutic use |
| WO2018087391A1 (en) | 2016-11-14 | 2018-05-17 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for modulating stem cells proliferation or differentiation |
| WO2018094275A1 (en) | 2016-11-18 | 2018-05-24 | Tolero Pharmaceuticals, Inc. | Alvocidib prodrugs and their use as protein kinase inhibitors |
| WO2018101448A1 (en) | 2016-11-30 | 2018-06-07 | Kyowa Hakko Kirin Co., Ltd. | Method of treating cancer using anti-ccr4 antibody and anti-pd-1 antibody |
| CA3045508A1 (en) | 2016-12-03 | 2018-06-07 | Juno Therapeutics, Inc. | Methods for modulation of car-t cells |
| PT3551660T (en) | 2016-12-07 | 2023-11-30 | Ludwig Inst For Cancer Res Ltd | Anti-ctla-4 antibodies and methods of use thereof |
| MA50948A (en) | 2016-12-07 | 2020-10-14 | Agenus Inc | ANTIBODIES AND METHODS OF USING THE SAME |
| WO2018119266A1 (en) | 2016-12-22 | 2018-06-28 | Incyte Corporation | Benzooxazole derivatives as immunomodulators |
| CA3047508A1 (en) | 2016-12-23 | 2018-06-28 | Virttu Biologics Limited | Treatment of cancer |
| WO2018119474A2 (en) * | 2016-12-23 | 2018-06-28 | Remd Biotherapeutics, Inc. | Immunotherapy using antibodies that bind programmed death 1 (pd-1) |
| WO2018122245A1 (en) | 2016-12-28 | 2018-07-05 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods of predicting the survival time of patients suffering from cms3 colorectal cancer |
| WO2018122249A1 (en) | 2016-12-28 | 2018-07-05 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for predicting the survival time of patients suffering from a microsatellite stable colorectal cancer |
| CN110431135A (en) | 2017-01-06 | 2019-11-08 | 大连万春布林医药有限公司 | Tubulin binding compound and its therapeutical uses |
| EP3565560B1 (en) | 2017-01-09 | 2024-05-29 | OnkosXcel Therapeutics, LLC | Predictive and diagnostic methods for prostate cancer |
| MX2019008346A (en) | 2017-01-13 | 2019-09-09 | Agenus Inc | T cell receptors that bind to ny-eso-1 and methods of use thereof. |
| WO2018134279A1 (en) | 2017-01-18 | 2018-07-26 | Pieris Pharmaceuticals Gmbh | Novel fusion polypeptides specific for lag-3 and pd-1 |
| WO2018133837A1 (en) * | 2017-01-20 | 2018-07-26 | Tayu Huaxia Biotech Medical Group Co., Ltd. | Anti-pd-1 antibodies and uses thereof |
| WO2018134784A1 (en) | 2017-01-20 | 2018-07-26 | Novartis Ag | Combination therapy for the treatment of cancer |
| CN117327075A (en) | 2017-01-23 | 2024-01-02 | 锐新医药公司 | Bicyclic compounds as allosteric SHP2 inhibitors |
| MX2019008696A (en) | 2017-01-23 | 2019-09-13 | Revolution Medicines Inc | Pyridine compounds as allosteric shp2 inhibitors. |
| CN110461847B (en) | 2017-01-25 | 2022-06-07 | 百济神州有限公司 | Crystalline forms of (S) -7- (1- (but-2-alkynoyl) piperidin-4-yl) -2- (4-phenoxyphenyl) -4,5,6, 7-tetrahydropyrazolo [1,5-a ] pyrimidine-3-carboxamide, preparation and use thereof |
| JP7062010B2 (en) | 2017-01-27 | 2022-05-02 | セルジーン コーポレイション | 3- (1-oxo-4-((4-((3-oxomorpholino) methyl) benzyl) oxy) isoindoline-2-yl) piperidine-2,6-dione and its isotopolog |
| KR20190109479A (en) | 2017-02-01 | 2019-09-25 | 비욘드스프링 파마수티컬스, 인코포레이티드. | How to reduce neutropenia |
| JOP20190187A1 (en) | 2017-02-03 | 2019-08-01 | Novartis Ag | Anti-ccr7 antibody drug conjugates |
| CN110573172A (en) | 2017-02-06 | 2019-12-13 | 奥里尼斯生物科学有限公司 | Targeted engineered interferons and uses thereof |
| KR102642385B1 (en) | 2017-02-06 | 2024-03-04 | 오리오니스 바이오사이언시스 엔브이 | Targeted chimeric proteins and uses thereof |
| WO2018146148A1 (en) | 2017-02-07 | 2018-08-16 | INSERM (Institut National de la Santé et de la Recherche Médicale) | A method for predicting the response to checkpoint blockade cancer immunotherapy |
| WO2018146128A1 (en) | 2017-02-07 | 2018-08-16 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Detection of kit polymorphism for predicting the response to checkpoint blockade cancer immunotherapy |
| PL3579874T3 (en) | 2017-02-10 | 2022-02-28 | Novartis Ag | 1-(4-amino-5-bromo-6-(1 h-pyrazol-1-yl)pyrimidin-2-yl)-1 h-pyrazol-4-ol and use thereof in the treatment of cancer |
| US20190375847A1 (en) | 2017-02-15 | 2019-12-12 | Glaxosmithkline Intellectual Property Development Limited | Combination treatment for cancer |
| WO2018151820A1 (en) | 2017-02-16 | 2018-08-23 | Elstar Therapeutics, Inc. | Multifunctional molecules comprising a trimeric ligand and uses thereof |
| CA3054303A1 (en) | 2017-02-22 | 2018-08-30 | Twist Bioscience Corporation | Nucleic acid based data storage |
| WO2018163051A1 (en) | 2017-03-06 | 2018-09-13 | Novartis Ag | Methods of treatment of cancer with reduced ubb expression |
| JP2020510050A (en) | 2017-03-15 | 2020-04-02 | アムジエン・インコーポレーテツド | Use of an oncolytic virus alone or in combination with a checkpoint inhibitor to treat cancer |
| US10894959B2 (en) | 2017-03-15 | 2021-01-19 | Twist Bioscience Corporation | Variant libraries of the immunological synapse and synthesis thereof |
| EP3601353A1 (en) | 2017-03-31 | 2020-02-05 | Five Prime Therapeutics, Inc. | Combination therapy for cancer using anti-gitr antibodies |
| IL269026B1 (en) | 2017-03-31 | 2024-08-01 | Bristol Myers Squibb Co | Anti-pd-1 antibodies for treating tumors in high tumor mutational burden (tmb) patients |
| CN106987631A (en) * | 2017-04-01 | 2017-07-28 | 武汉赛云博生物科技有限公司 | A kind of immune group sequencing technologies for the adjoint diagnosis of PD 1/PD L1 blocking treatments |
| WO2018185618A1 (en) | 2017-04-03 | 2018-10-11 | Novartis Ag | Anti-cdh6 antibody drug conjugates and anti-gitr antibody combinations and methods of treatment |
| TWI788340B (en) | 2017-04-07 | 2023-01-01 | 美商必治妥美雅史谷比公司 | Anti-icos agonist antibodies and uses thereof |
| MX2019012223A (en) | 2017-04-13 | 2019-12-09 | Agenus Inc | Anti-cd137 antibodies and methods of use thereof. |
| KR20190134786A (en) | 2017-04-14 | 2019-12-04 | 콜드 제네시스, 인크. | How to treat bladder cancer |
| BR112019021790A2 (en) | 2017-04-18 | 2020-05-05 | Tempest Therapeutics Inc | bicyclic compounds and methods of use |
| JP2020517256A (en) | 2017-04-19 | 2020-06-18 | エルスター セラピューティクス, インコーポレイテッド | Multispecific molecules and uses thereof |
| AR111419A1 (en) | 2017-04-27 | 2019-07-10 | Novartis Ag | INDAZOL PIRIDONA FUSIONED COMPOUNDS AS ANTIVIRALS |
| UY37695A (en) | 2017-04-28 | 2018-11-30 | Novartis Ag | BIS 2’-5’-RR- (3’F-A) (3’F-A) CYCLE DINUCLEOTIDE COMPOUND AND USES OF THE SAME |
| EP3615068A1 (en) | 2017-04-28 | 2020-03-04 | Novartis AG | Bcma-targeting agent, and combination therapy with a gamma secretase inhibitor |
| EP3615055A1 (en) | 2017-04-28 | 2020-03-04 | Novartis AG | Cells expressing a bcma-targeting chimeric antigen receptor, and combination therapy with a gamma secretase inhibitor |
| EP3615566B1 (en) | 2017-04-28 | 2023-12-20 | Marengo Therapeutics, Inc. | Multispecific molecules comprising a non-immunoglobulin heterodimerization domain and uses thereof |
| AR111651A1 (en) | 2017-04-28 | 2019-08-07 | Novartis Ag | CONJUGATES OF ANTIBODIES THAT INCLUDE TOLL TYPE RECEIVER AGONISTS AND COMBINATION THERAPIES |
| TWI805582B (en) | 2017-05-01 | 2023-06-21 | 美商艾吉納斯公司 | Anti-tigit antibodies and methods of use thereof |
| AR111658A1 (en) | 2017-05-05 | 2019-08-07 | Novartis Ag | 2-TRICYCLINAL CHINOLINONES AS ANTIBACTERIAL AGENTS |
| JOP20190256A1 (en) | 2017-05-12 | 2019-10-28 | Icahn School Med Mount Sinai | Newcastle disease viruses and uses thereof |
| CA3063359A1 (en) | 2017-05-12 | 2018-11-15 | Harpoon Therapeutics, Inc. | Mesothelin binding proteins |
| JP7274426B2 (en) | 2017-05-16 | 2023-05-16 | ブリストル-マイヤーズ スクイブ カンパニー | Treatment of cancer with anti-GITR agonist antibodies |
| AR111760A1 (en) | 2017-05-19 | 2019-08-14 | Novartis Ag | COMPOUNDS AND COMPOSITIONS FOR THE TREATMENT OF SOLID TUMORS THROUGH INTRATUMORAL ADMINISTRATION |
| EP3630162A1 (en) | 2017-05-24 | 2020-04-08 | Novartis AG | Antibody-cytokine engrafted proteins and methods of use |
| WO2018215937A1 (en) | 2017-05-24 | 2018-11-29 | Novartis Ag | Interleukin-7 antibody cytokine engrafted proteins and methods of use in the treatment of cancer |
| AU2018274216A1 (en) | 2017-05-24 | 2019-12-12 | Novartis Ag | Antibody-cytokine engrafted proteins and methods of use in the treatment of cancer |
| AR111960A1 (en) | 2017-05-26 | 2019-09-04 | Incyte Corp | CRYSTALLINE FORMS OF A FGFR INHIBITOR AND PROCESSES FOR ITS PREPARATION |
| MX2019012076A (en) | 2017-05-30 | 2019-12-09 | Bristol Myers Squibb Co | Compositions comprising an anti-lag-3 antibody or an anti-lag-3 antibody and an anti-pd-1 or anti-pd-l1 antibody. |
| CA3060989A1 (en) | 2017-05-30 | 2018-12-06 | Bristol-Myers Squibb Company | Compositions comprising a combination of an anti-lag-3 antibody, a pd-1 pathway inhibitor, and an immunotherapeutic agent |
| MX2019012032A (en) | 2017-05-30 | 2019-10-30 | Bristol Myers Squibb Co | Treatment of lag-3 positive tumors. |
| JP2020522254A (en) | 2017-05-31 | 2020-07-30 | エルスター セラピューティクス, インコーポレイテッド | Multispecific molecules that bind myeloproliferative leukemia (MPL) proteins and uses thereof |
| JOP20190279A1 (en) | 2017-05-31 | 2019-11-28 | Novartis Ag | Crystalline forms of 5-bromo-2,6-di(1 h-pyrazol-1-yl)pyrimidin-4-amine and new salts |
| EP3630840A1 (en) | 2017-06-01 | 2020-04-08 | Bristol-Myers Squibb Company | Methods of treating a tumor using an anti-pd-1 antibody |
| WO2018223004A1 (en) | 2017-06-01 | 2018-12-06 | Xencor, Inc. | Bispecific antibodies that bind cd20 and cd3 |
| CA3065929A1 (en) | 2017-06-01 | 2018-12-06 | Michael Wayne SAVILLE | Bispecific antibodies that bind cd123 and cd3 |
| BR112019025403A2 (en) | 2017-06-02 | 2020-08-18 | Juno Therapeutics Inc | manufacturing articles and methods for treatment using adoptive cell therapy |
| JP2020522562A (en) | 2017-06-06 | 2020-07-30 | ストキューブ アンド シーオー., インコーポレイテッド | Methods of treating cancer with antibodies and molecules that bind to BTN1A1 or BTN1A1 ligand |
| AU2018284227B2 (en) | 2017-06-12 | 2024-05-02 | Twist Bioscience Corporation | Methods for seamless nucleic acid assembly |
| WO2018231864A1 (en) | 2017-06-12 | 2018-12-20 | Twist Bioscience Corporation | Methods for seamless nucleic acid assembly |
| WO2018229715A1 (en) | 2017-06-16 | 2018-12-20 | Novartis Ag | Compositions comprising anti-cd32b antibodies and methods of use thereof |
| EP3641739A1 (en) | 2017-06-20 | 2020-04-29 | Institut Curie | Inhibitor of suv39h1 histone methyltransferase for use in cancer combination therapy |
| WO2018235056A1 (en) | 2017-06-22 | 2018-12-27 | Novartis Ag | Il-1beta binding antibodies for use in treating cancer |
| KR20200019229A (en) | 2017-06-22 | 2020-02-21 | 셀진 코포레이션 | Treatment of Hepatocellular Carcinoma Characterized by Hepatitis B Virus Infection |
| WO2018237173A1 (en) | 2017-06-22 | 2018-12-27 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| US20190048072A1 (en) | 2017-06-22 | 2019-02-14 | Novartis Ag | USE OF IL-1beta BINDING ANTIBODIES |
| SG11201912473PA (en) | 2017-06-22 | 2020-01-30 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| US11066445B2 (en) | 2017-06-23 | 2021-07-20 | Bristol-Myers Squibb Company | Immunomodulators acting as antagonists of PD-1 |
| CN110799537B (en) * | 2017-06-25 | 2023-07-28 | 西雅图免疫公司 | anti-PD-1 antibodies and methods of making and using the same |
| AU2018290532A1 (en) | 2017-06-26 | 2019-11-21 | Beigene, Ltd. | Immunotherapy for hepatocellular carcinoma |
| WO2019006007A1 (en) | 2017-06-27 | 2019-01-03 | Novartis Ag | Dosage regimens for anti-tim-3 antibodies and uses thereof |
| JP2020526194A (en) | 2017-06-29 | 2020-08-31 | ジュノー セラピューティクス インコーポレイテッド | Mouse model for assessing toxicity associated with immunotherapeutic agents |
| RS64029B1 (en) | 2017-06-30 | 2023-03-31 | Celgene Corp | Compositions and methods of use of 2-(4-chlorophenyl)-n-((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-5-yl) methyl) -2,2-difluoroacetamide |
| JP7258009B2 (en) | 2017-07-10 | 2023-04-14 | セルジーン コーポレイション | Antiproliferative compounds and methods of use thereof |
| WO2019016174A1 (en) | 2017-07-18 | 2019-01-24 | Institut Gustave Roussy | Method for assessing the response to pd-1/pdl-1 targeting drugs |
| US20200172617A1 (en) | 2017-07-20 | 2020-06-04 | Novartis Ag | Dosage regimens of anti-lag-3 antibodies and uses thereof |
| US11926664B2 (en) | 2017-07-25 | 2024-03-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for modulating monocytopoiesis |
| US11899017B2 (en) | 2017-07-28 | 2024-02-13 | Bristol-Myers Squibb Company | Predictive peripheral blood biomarker for checkpoint inhibitors |
| MX2020001980A (en) | 2017-08-28 | 2020-03-24 | Bristol Myers Squibb Co | Tim-3 antagonists for the treatment and diagnosis of cancers. |
| CA3073055A1 (en) | 2017-09-04 | 2019-03-07 | Agenus Inc. | T cell receptors that bind to mixed lineage leukemia (mll)-specific phosphopeptides and methods of use thereof |
| WO2019051164A1 (en) * | 2017-09-07 | 2019-03-14 | Augusta University Research Institute, Inc. | Antibodies to programmed cell death protein 1 |
| BR112020004246A2 (en) | 2017-09-07 | 2020-09-01 | Revolution Medicines, Inc. | shp2 inhibitory compositions and methods for the treatment of cancer |
| CN111566125A (en) | 2017-09-11 | 2020-08-21 | 特韦斯特生物科学公司 | GPCR binding proteins and synthesis thereof |
| US11497756B2 (en) | 2017-09-12 | 2022-11-15 | Sumitomo Pharma Oncology, Inc. | Treatment regimen for cancers that are insensitive to BCL-2 inhibitors using the MCL-1 inhibitor alvocidib |
| EP3684410A1 (en) | 2017-09-19 | 2020-07-29 | Institut Curie | Agonist of aryl hydrocarbon receptor for use in cancer combination therapy |
| CN109554349B (en) * | 2017-09-27 | 2022-06-24 | 亘喜生物科技(上海)有限公司 | Engineered immune cells with silenced PD-1 gene expression |
| US11492375B2 (en) | 2017-10-03 | 2022-11-08 | Bristol-Myers Squibb Company | Cyclic peptide immunomodulators |
| KR20200063147A (en) * | 2017-10-10 | 2020-06-04 | 누맙 세러퓨틱스 아게 | PDL1 targeting antibodies and methods of use thereof |
| WO2019075265A1 (en) | 2017-10-12 | 2019-04-18 | Revolution Medicines, Inc. | Pyridine, pyrazine, and triazine compounds as allosteric shp2 inhibitors |
| JP7066837B2 (en) | 2017-10-13 | 2022-05-13 | ハープーン セラピューティクス,インク. | B cell maturation antigen binding protein |
| WO2019075468A1 (en) | 2017-10-15 | 2019-04-18 | Bristol-Myers Squibb Company | Methods of treating tumor |
| US20210189336A1 (en) | 2017-10-18 | 2021-06-24 | Vivia Biotech, S.L. | Bite-activated car-t cells |
| GB2583590A (en) | 2017-10-20 | 2020-11-04 | Twist Bioscience Corp | Heated nanowells for polynucleotide synthesis |
| US20210040205A1 (en) | 2017-10-25 | 2021-02-11 | Novartis Ag | Antibodies targeting cd32b and methods of use thereof |
| US12031975B2 (en) | 2017-11-01 | 2024-07-09 | Juno Therapeutics, Inc. | Methods of assessing or monitoring a response to a cell therapy |
| TW201932482A (en) | 2017-11-01 | 2019-08-16 | 美商奇諾治療有限公司 | Chimeric antigen receptors specific for B-cell maturation antigen and encoding polynucleotides |
| WO2019089969A2 (en) | 2017-11-01 | 2019-05-09 | Juno Therapeutics, Inc. | Antibodies and chimeric antigen receptors specific for b-cell maturation antigen |
| EP3706778A1 (en) | 2017-11-06 | 2020-09-16 | Bristol-Myers Squibb Company | Methods of treating a tumor |
| US20200353050A1 (en) | 2017-11-10 | 2020-11-12 | Armo Biosciences, Inc. | Compositions and methods of use of interleukin-10 in combination with immune check-point pathway inhibitors |
| CN111655288A (en) | 2017-11-16 | 2020-09-11 | 诺华股份有限公司 | Combination therapy |
| CN111315749A (en) | 2017-11-17 | 2020-06-19 | 诺华股份有限公司 | Novel dihydroisoxazole compounds and their use in the treatment of hepatitis b |
| CR20200204A (en) | 2017-11-17 | 2020-06-18 | Merck Sharp & Dohme | Antibodies specific for immunoglobulin-like transcript 3 (ilt3) and uses thereof |
| KR20200111168A (en) | 2017-11-24 | 2020-09-28 | 인쎄름 (엥스띠뛰 나씨오날 드 라 쌍떼 에 드 라 흐쉐르슈 메디깔) | Methods and compositions for cancer treatment |
| WO2019108795A1 (en) | 2017-11-29 | 2019-06-06 | Beigene Switzerland Gmbh | Treatment of indolent or aggressive b-cell lymphomas using a combination comprising btk inhibitors |
| WO2019108900A1 (en) | 2017-11-30 | 2019-06-06 | Novartis Ag | Bcma-targeting chimeric antigen receptor, and uses thereof |
| EP3720881A1 (en) | 2017-12-08 | 2020-10-14 | Elstar Therapeutics, Inc. | Multispecific molecules and uses thereof |
| MA51184A (en) | 2017-12-15 | 2020-10-21 | Juno Therapeutics Inc | ANTI-CCT5 BINDING MOLECULES AND RELATED METHODS OF USE |
| BR112020009757A2 (en) | 2017-12-15 | 2020-11-03 | Revolution Medicines, Inc. | polycyclic compounds as allosteric inhibitors of shp2 |
| JP2021507906A (en) | 2017-12-20 | 2021-02-25 | ノバルティス アーゲー | Fusion tricyclic pyrazolo-dihydropyrazinyl-pyridone compound as an antiviral agent |
| CN109966487B (en) * | 2017-12-28 | 2023-08-25 | 上海复宏汉霖生物制药有限公司 | Pharmaceutical formulation comprising anti-PD-L1 monoclonal antibody |
| AU2019205269A1 (en) | 2018-01-04 | 2020-07-30 | Twist Bioscience Corporation | DNA-based digital information storage |
| US11324774B2 (en) | 2018-01-05 | 2022-05-10 | Augusta University Research Institute, Inc. | Compositions of oral alkaline salts and metabolic acid inducers and uses thereof |
| US20200368268A1 (en) | 2018-01-08 | 2020-11-26 | Novartis Ag | Immune-enhancing rnas for combination with chimeric antigen receptor therapy |
| SG11202005323SA (en) | 2018-01-12 | 2020-07-29 | Bristol Myers Squibb Co | Combination therapy with anti-il-8 antibodies and anti-pd-1 antibodies for treating cancer |
| KR20200109339A (en) | 2018-01-16 | 2020-09-22 | 브리스톨-마이어스 스큅 컴퍼니 | How to treat cancer using antibodies to TIM3 |
| CA3096287A1 (en) | 2018-01-22 | 2019-07-25 | Pascal Biosciences Inc. | Cannabinoids and derivatives for promoting immunogenicity of tumor and infected cells |
| CA3088542A1 (en) | 2018-01-22 | 2019-07-25 | Bristol-Myers Squibb Company | Compositions and methods of treating cancer |
| US11786523B2 (en) | 2018-01-24 | 2023-10-17 | Beyondspring Pharmaceuticals, Inc. | Composition and method for reducing thrombocytopenia |
| WO2019152743A1 (en) | 2018-01-31 | 2019-08-08 | Celgene Corporation | Combination therapy using adoptive cell therapy and checkpoint inhibitor |
| AU2019215031A1 (en) | 2018-01-31 | 2020-08-20 | Novartis Ag | Combination therapy using a chimeric antigen receptor |
| WO2019157124A1 (en) | 2018-02-08 | 2019-08-15 | Bristol-Myers Squibb Company | Combination of a tetanus toxoid, anti-ox40 antibody and/or anti-pd-1 antibody to treat tumors |
| TWI849895B (en) | 2018-02-09 | 2024-07-21 | 日商小野藥品工業股份有限公司 | Bispecific antibody |
| NL2020422B1 (en) | 2018-02-12 | 2019-08-19 | Stichting Het Nederlands Kanker Inst Antoni Van Leeuwenhoek Ziekenhuis | Methods for Predicting Treatment Outcome and/or for Selecting a Subject Suitable for Immune Checkpoint Therapy. |
| US20200399383A1 (en) | 2018-02-13 | 2020-12-24 | Novartis Ag | Chimeric antigen receptor therapy in combination with il-15r and il15 |
| IL303087B1 (en) | 2018-02-27 | 2024-08-01 | Incyte Corp | Imidazopyrimidines and triazolopyrimidines as a2a / a2b inhibitors |
| JP2021514982A (en) | 2018-02-28 | 2021-06-17 | ノバルティス アーゲー | Indole-2-carbonyl compounds and their use for the treatment of hepatitis B |
| US20210002373A1 (en) | 2018-03-01 | 2021-01-07 | Nextcure, Inc. | KLRG1 Binding Compositions and Methods of Use Thereof |
| EP3762105A1 (en) | 2018-03-06 | 2021-01-13 | Institut Curie | Inhibitor of setdb1 histone methyltransferase for use in cancer combination therapy |
| CA3093742A1 (en) | 2018-03-12 | 2019-09-19 | Inserm (Institut National De La Sante Et De La Recherche Medicale) | Use of caloric restriction mimetics for potentiating chemo-immunotherapy for the treatment of cancers |
| WO2019178364A2 (en) | 2018-03-14 | 2019-09-19 | Elstar Therapeutics, Inc. | Multifunctional molecules and uses thereof |
| WO2019178362A1 (en) | 2018-03-14 | 2019-09-19 | Elstar Therapeutics, Inc. | Multifunctional molecules that bind to calreticulin and uses thereof |
| TW202003565A (en) | 2018-03-23 | 2020-01-16 | 美商必治妥美雅史谷比公司 | Antibodies against MICA and/or MICB and uses thereof |
| CN108530537B (en) * | 2018-03-29 | 2019-07-02 | 中国人民解放军军事科学院军事医学研究院 | PD-1/PD-L1 signal pathway inhibitor |
| WO2019185792A1 (en) | 2018-03-29 | 2019-10-03 | Philogen S.P.A | Cancer treatment using immunoconjugates and immune check-point inhibitors |
| KR20200139724A (en) | 2018-03-30 | 2020-12-14 | 브리스톨-마이어스 스큅 컴퍼니 | How to treat a tumor |
| US12110337B2 (en) | 2018-04-04 | 2024-10-08 | Bristol-Myers Squibb Company | Anti-CD27 antibodies and uses thereof |
| JP7511478B2 (en) | 2018-04-09 | 2024-07-05 | チェックメイト ファーマシューティカルズ | Packaging of oligonucleotides into virus-like particles |
| AU2019252795A1 (en) | 2018-04-12 | 2020-10-29 | Bristol-Myers Squibb Company | Anticancer combination therapy with cd73 antagonist antibody and pd-1/pd-l1 axis antagonist antibody |
| US20210147547A1 (en) | 2018-04-13 | 2021-05-20 | Novartis Ag | Dosage Regimens For Anti-Pd-L1 Antibodies And Uses Thereof |
| BR112020021042A2 (en) | 2018-04-17 | 2021-01-19 | Tempest Therapeutics, Inc. | BICYCLIC CARBOXAMIDES AND METHODS OF USE OF THE SAME |
| US11524991B2 (en) | 2018-04-18 | 2022-12-13 | Xencor, Inc. | PD-1 targeted heterodimeric fusion proteins containing IL-15/IL-15Ra Fc-fusion proteins and PD-1 antigen binding domains and uses thereof |
| JP7516254B2 (en) | 2018-04-18 | 2024-07-16 | ゼンコア インコーポレイテッド | IL-15/IL-15RA HETERODIMERIC FC FUSION PROTEINS AND USES THEREOF |
| JP2021522239A (en) | 2018-04-26 | 2021-08-30 | アジェナス インコーポレイテッド | Heat shock protein-binding peptide composition and how to use it |
| WO2019207030A1 (en) | 2018-04-26 | 2019-10-31 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for predicting a response with an immune checkpoint inhibitor in a patient suffering from a lung cancer |
| WO2019210153A1 (en) | 2018-04-27 | 2019-10-31 | Novartis Ag | Car t cell therapies with enhanced efficacy |
| US12048745B2 (en) | 2018-05-01 | 2024-07-30 | Augusta University Research Institute, Inc. | Methods for detecting and reversing immune therapy resistance |
| EP3788369A1 (en) | 2018-05-01 | 2021-03-10 | Novartis Ag | Biomarkers for evaluating car-t cells to predict clinical outcome |
| SG11202010882XA (en) | 2018-05-04 | 2020-11-27 | Incyte Corp | Salts of an fgfr inhibitor |
| DK3788047T3 (en) | 2018-05-04 | 2024-09-16 | Incyte Corp | Solid forms of an FGFR inhibitor and methods of making the same |
| CN112639130B (en) | 2018-05-18 | 2024-08-09 | 特韦斯特生物科学公司 | Polynucleotides, reagents and methods for nucleic acid hybridization |
| US11168089B2 (en) | 2018-05-18 | 2021-11-09 | Incyte Corporation | Fused pyrimidine derivatives as A2A / A2B inhibitors |
| FI3796912T3 (en) | 2018-05-23 | 2023-05-11 | Celgene Corp | Antiproliferative compounds and bispecific antibody against bcma and cd3 for combined use |
| BR112020023756A2 (en) | 2018-05-23 | 2021-02-09 | Celgene Corporation | treatment of multiple myeloma and use of biomarkers for 4- (4- (4 - (((2- (2,6-dioxopiperidin-3-yl) -1-oxoisoindolin-4-yl) oxy) methyl) benzyl) piperazin- 1-yl) -3-fluorobenzonitrile |
| AR126019A1 (en) | 2018-05-30 | 2023-09-06 | Novartis Ag | ANTIBODIES AGAINST ENTPD2, COMBINATION THERAPIES AND METHODS OF USE OF ANTIBODIES AND COMBINATION THERAPIES |
| WO2019229699A1 (en) | 2018-05-31 | 2019-12-05 | Novartis Ag | Hepatitis b antibodies |
| US20210214459A1 (en) | 2018-05-31 | 2021-07-15 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| US20210205449A1 (en) | 2018-06-01 | 2021-07-08 | Novartis Ag | Dosing of a bispecific antibody that bind cd123 and cd3 |
| US11492409B2 (en) | 2018-06-01 | 2022-11-08 | Novartis Ag | Binding molecules against BCMA and uses thereof |
| KR20210029158A (en) | 2018-06-03 | 2021-03-15 | 람카프 바이오 베타 엘티디. | Bispecific antibodies to CEACAM5 and CD47 |
| TW202016139A (en) | 2018-06-13 | 2020-05-01 | 瑞士商諾華公司 | Bcma chimeric antigen receptors and uses thereof |
| US20210347842A1 (en) | 2018-06-19 | 2021-11-11 | Eli Lilly And Company | Compositions and methods of use of il-10 agents in conjunction with chimeric antigen receptor cell therapy |
| AU2019288276A1 (en) | 2018-06-20 | 2021-01-14 | Incyte Corporation | Anti-PD-1 antibodies and uses thereof |
| TW202005985A (en) | 2018-06-21 | 2020-02-01 | 美商再生元醫藥公司 | Methods for treating cancer with bispecific anti-CD3xMUC16 antibodies and anti-PD-1 antibodies |
| WO2020005068A2 (en) | 2018-06-29 | 2020-01-02 | Stichting Het Nederlands Kanker Instituut-Antoni van Leeuwenhoek Ziekenhuis | Gene signatures and method for predicting response to pd-1 antagonists and ctla-4 antagonists, and combination thereof |
| AU2019297451A1 (en) | 2018-07-03 | 2021-01-28 | Marengo Therapeutics, Inc. | Anti-TCR antibody molecules and uses thereof |
| CN113166153A (en) | 2018-07-05 | 2021-07-23 | 因赛特公司 | Fused pyrazine derivatives as A2A/A2B inhibitors |
| AR116109A1 (en) | 2018-07-10 | 2021-03-31 | Novartis Ag | DERIVATIVES OF 3- (5-AMINO-1-OXOISOINDOLIN-2-IL) PIPERIDINE-2,6-DIONA AND USES OF THE SAME |
| JOP20210001A1 (en) | 2018-07-10 | 2021-01-05 | Novartis Ag | 3-(5-hydroxy-1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives and their use in the treatment of ikaros family zinc finger 2 (ikzf2)-dependent diseases |
| WO2020014583A1 (en) | 2018-07-13 | 2020-01-16 | Bristol-Myers Squibb Company | Ox-40 agonist, pd-1 pathway inhibitor and ctla-4 inhibitor combination for use in a mehtod of treating a cancer or a solid tumor |
| WO2020021465A1 (en) | 2018-07-25 | 2020-01-30 | Advanced Accelerator Applications (Italy) S.R.L. | Method of treatment of neuroendocrine tumors |
| EP3826660A1 (en) | 2018-07-26 | 2021-06-02 | Bristol-Myers Squibb Company | Lag-3 combination therapy for the treatment of cancer |
| WO2020021061A1 (en) | 2018-07-26 | 2020-01-30 | Pieris Pharmaceuticals Gmbh | Humanized anti-pd-1 antibodies and uses thereof |
| EP3833383A1 (en) | 2018-08-06 | 2021-06-16 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating cancers |
| WO2020030571A1 (en) | 2018-08-06 | 2020-02-13 | Glaxosmithkline Intellectual Property Development Limited | Combinations of a pd-1 antibody and a tlr4 modulator and uses thereof |
| EP3833762A4 (en) | 2018-08-09 | 2022-09-28 | Verseau Therapeutics, Inc. | Oligonucleotide compositions for targeting ccr2 and csf1r and uses thereof |
| JP7459058B2 (en) | 2018-08-27 | 2024-04-01 | ピエリス ファーマシューティカルズ ゲーエムベーハー | Combination therapy comprising a CD137/HER2 bispecific substance and a PD-1 system inhibitor and its use |
| AU2019328313A1 (en) | 2018-08-30 | 2021-02-25 | Immunitybio, Inc. | Single-chain chimeric polypeptides and uses thereof |
| US11730762B2 (en) | 2018-08-30 | 2023-08-22 | HCW Biologics, Inc. | Methods for stimulating proliferation or differentiation of an immune cell with a multi-chain chimeric polypeptide |
| AU2019328290B2 (en) | 2018-08-30 | 2024-10-10 | Immunitybio, Inc. | Multi-chain chimeric polypeptides and uses thereof |
| WO2020044252A1 (en) | 2018-08-31 | 2020-03-05 | Novartis Ag | Dosage regimes for anti-m-csf antibodies and uses thereof |
| WO2020048942A1 (en) | 2018-09-04 | 2020-03-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for enhancing cytotoxic t lymphocyte-dependent immune responses |
| EP3847194A1 (en) * | 2018-09-07 | 2021-07-14 | Pfizer Inc. | Anti-avb8 antibodies and compositions and uses thereof |
| WO2020049534A1 (en) | 2018-09-07 | 2020-03-12 | Novartis Ag | Sting agonist and combination therapy thereof for the treatment of cancer |
| WO2020053742A2 (en) | 2018-09-10 | 2020-03-19 | Novartis Ag | Anti-hla-hbv peptide antibodies |
| EP3849554A4 (en) | 2018-09-11 | 2022-06-01 | Curis, Inc. | Combination therapy with a phosphoinositide 3-kinase inhibitor with a zinc binding moiety |
| CN112996789A (en) | 2018-09-12 | 2021-06-18 | 诺华股份有限公司 | Antiviral pyridopyrazinedione compounds |
| WO2020058372A1 (en) | 2018-09-19 | 2020-03-26 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical composition for the treatment of cancers resistant to immune checkpoint therapy |
| AU2019345151A1 (en) | 2018-09-19 | 2021-04-29 | Alpine Immune Sciences, Inc. | Methods and uses of variant CD80 fusion proteins and related constructs |
| WO2020069028A1 (en) | 2018-09-25 | 2020-04-02 | Harpoon Therapeutics, Inc. | Dll3 binding proteins and methods of use |
| US20220242957A1 (en) | 2018-09-27 | 2022-08-04 | Marengo Therapeutics, Inc. | Csf1r/ccr2 multispecific antibodies |
| EP3856782A1 (en) | 2018-09-28 | 2021-08-04 | Novartis AG | Cd19 chimeric antigen receptor (car) and cd22 car combination therapies |
| EP3856779A1 (en) | 2018-09-28 | 2021-08-04 | Novartis AG | Cd22 chimeric antigen receptor (car) therapies |
| KR20210068473A (en) | 2018-09-29 | 2021-06-09 | 노파르티스 아게 | Method for preparing a compound for inhibiting SHP2 activity |
| US20220040183A1 (en) | 2018-10-01 | 2022-02-10 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of inhibitors of stress granule formation for targeting the regulation of immune responses |
| SG11202103192RA (en) | 2018-10-03 | 2021-04-29 | Xencor Inc | Il-12 heterodimeric fc-fusion proteins |
| JP2022512642A (en) | 2018-10-09 | 2022-02-07 | ブリストル-マイヤーズ スクイブ カンパニー | Anti-MerTK antibody to treat cancer |
| US11066404B2 (en) | 2018-10-11 | 2021-07-20 | Incyte Corporation | Dihydropyrido[2,3-d]pyrimidinone compounds as CDK2 inhibitors |
| MA53862A (en) | 2018-10-12 | 2022-01-19 | Xencor Inc | FC FUSION PROTEINS OF IL-15/IL-15RALPHA TARGETTING PD-1 AND USES IN COMBINATION THERAPIES INVOLVING THE SAME |
| JP2022504905A (en) | 2018-10-16 | 2022-01-13 | ノバルティス アーゲー | Tumor mutation loading alone or in combination with immune markers as a biomarker to predict response to targeted therapy |
| CN112955462B (en) | 2018-10-18 | 2024-05-07 | 国家医疗保健研究所 | Combination of a beta IG-H3 antagonist and an immune checkpoint inhibitor for the treatment of solid tumors |
| US20210338813A1 (en) | 2018-10-19 | 2021-11-04 | Bristol-Myers Squibb Company | Combination Therapy for Melanoma |
| WO2020086724A1 (en) | 2018-10-23 | 2020-04-30 | Bristol-Myers Squibb Company | Methods of treating tumor |
| EP3873532A1 (en) | 2018-10-31 | 2021-09-08 | Novartis AG | Dc-sign antibody drug conjugates |
| AU2019372331A1 (en) | 2018-11-01 | 2021-05-27 | Juno Therapeutics, Inc. | Methods for treatment using chimeric antigen receptors specific for B-cell maturation antigen |
| SG11202103873VA (en) | 2018-11-01 | 2021-05-28 | Juno Therapeutics Inc | Chimeric antigen receptors specific for g protein-coupled receptor class c group 5 member d (gprc5d) |
| JP7513604B2 (en) | 2018-11-14 | 2024-07-09 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | Intralesional administration of PD-1 inhibitors to treat skin cancer |
| TW202028222A (en) | 2018-11-14 | 2020-08-01 | 美商Ionis製藥公司 | Modulators of foxp3 expression |
| US20220008515A1 (en) | 2018-11-16 | 2022-01-13 | Neoimmunetech, Inc. | Method of treating a tumor with a combination of il-7 protein and an immune checkpoint inhibitor |
| US20220008465A1 (en) | 2018-11-16 | 2022-01-13 | Juno Therapeutics, Inc. | Methods of dosing engineered t cells for the treatment of b cell malignancies |
| WO2020102501A1 (en) | 2018-11-16 | 2020-05-22 | Bristol-Myers Squibb Company | Anti-nkg2a antibodies and uses thereof |
| CN113453678A (en) | 2018-11-26 | 2021-09-28 | 德彪药业国际股份公司 | Combination therapy for HIV infection |
| ES2971964T3 (en) | 2018-11-28 | 2024-06-10 | Inst Nat Sante Rech Med | Methods and kit for testing the lytic potential of immune effector cells |
| SG11202105502RA (en) | 2018-11-30 | 2021-06-29 | Juno Therapeutics Inc | Methods for treatment using adoptive cell therapy |
| US11034710B2 (en) | 2018-12-04 | 2021-06-15 | Sumitomo Dainippon Pharma Oncology, Inc. | CDK9 inhibitors and polymorphs thereof for use as agents for treatment of cancer |
| WO2020115262A1 (en) | 2018-12-07 | 2020-06-11 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of cd26 and cd39 as new phenotypic markers for assessing maturation of foxp3+ t cells and uses thereof for diagnostic purposes |
| WO2020127059A1 (en) | 2018-12-17 | 2020-06-25 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of sulconazole as a furin inhibitor |
| CN113438961A (en) | 2018-12-20 | 2021-09-24 | Xencor股份有限公司 | Targeting heterodimeric Fc fusion proteins containing IL-15/IL-15R α and NKG2D antigen binding domains |
| KR20210106437A (en) | 2018-12-20 | 2021-08-30 | 노파르티스 아게 | Dosage regimens and pharmaceutical combinations comprising 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives |
| WO2020128613A1 (en) | 2018-12-21 | 2020-06-25 | Novartis Ag | Use of il-1beta binding antibodies |
| US20220056123A1 (en) | 2018-12-21 | 2022-02-24 | Novartis Ag | Use of il-1beta binding antibodies |
| CN113195541B (en) | 2018-12-21 | 2024-08-30 | 诺华股份有限公司 | Antibodies to PMEL17 and conjugates thereof |
| WO2020128637A1 (en) | 2018-12-21 | 2020-06-25 | Novartis Ag | Use of il-1 binding antibodies in the treatment of a msi-h cancer |
| AU2019408408A1 (en) | 2018-12-21 | 2021-06-03 | Valerio Therapeutics | New conjugated nucleic acid molecules and their uses |
| KR20210107730A (en) | 2018-12-21 | 2021-09-01 | 노파르티스 아게 | Use of IL-1 beta antibodies in the treatment or prevention of myelodysplastic syndrome |
| TW202039835A (en) | 2018-12-27 | 2020-11-01 | 美商安進公司 | Lyophilized virus formulations |
| US20220073626A1 (en) | 2019-01-03 | 2022-03-10 | Institut National De La Santé Et De La Recheche Médicale (Inserm) | Methods and pharmaceutical compositions for enhancing cd8+ t cell-dependent immune responses in subjects suffering from cancer |
| CN118178417A (en) | 2019-01-09 | 2024-06-14 | 细胞基因公司 | Antiproliferative compounds and second active agents for the treatment of multiple myeloma |
| JP7489989B2 (en) | 2019-01-09 | 2024-05-24 | セルジーン コーポレイション | Pharmaceutical compositions comprising (S)-4-(4-(4-(((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-4-yl)oxy)methyl)benzyl)piperazin-1-yl)-3-fluorobenzonitrile and methods of use thereof |
| US11370777B2 (en) | 2019-01-09 | 2022-06-28 | Celgene Corporation | Solid forms comprising (s)-4-(4-(4-(((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-4-yl)oxy)methyl)benzyl)piperazin-1-yl)-3-fluorobenzonitrile and salts thereof, and compositions comprising and methods of using the same |
| AU2020207408A1 (en) * | 2019-01-11 | 2021-07-29 | The Wistar Institute Of Anatomy And Biology | DNA monoclonal antibodies targeting PD-1 for the treatment and prevention of cancer |
| SG11202107606VA (en) | 2019-01-15 | 2021-08-30 | Inst Nat Sante Rech Med | Mutated interleukin-34 (il-34) polypeptides and uses thereof in therapy |
| JP2022518207A (en) | 2019-01-17 | 2022-03-14 | ジョージア テック リサーチ コーポレイション | Drug delivery system containing oxidized cholesterol |
| PE20212198A1 (en) | 2019-01-29 | 2021-11-16 | Juno Therapeutics Inc | ANTIBODIES AND CHIMERIC RECEPTORS OF SPECIFIC ANTIGENS TO ORPHAN RECEPTOR 1, RECEPTOR TYROSINE KINASE TYPE (ROR1) |
| TWI829857B (en) | 2019-01-29 | 2024-01-21 | 美商英塞特公司 | Pyrazolopyridines and triazolopyridines as a2a / a2b inhibitors |
| CA3129031A1 (en) | 2019-02-12 | 2020-08-20 | Novartis Ag | Pharmaceutical combination comprising tno155 and a pd-1 inhibitor |
| MX2021009371A (en) | 2019-02-12 | 2021-09-10 | Sumitomo Pharma Oncology Inc | Formulations comprising heterocyclic protein kinase inhibitors. |
| EP3924055B1 (en) | 2019-02-15 | 2024-04-03 | Novartis AG | Substituted 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives and uses thereof |
| EP3923949A1 (en) | 2019-02-15 | 2021-12-22 | Incyte Corporation | Cyclin-dependent kinase 2 biomarkers and uses thereof |
| CN113490528A (en) | 2019-02-15 | 2021-10-08 | 诺华股份有限公司 | 3- (1-oxo-5- (piperidine-4-yl) isoindoline-2-yl) piperidine-2, 6-dione derivatives and uses thereof |
| WO2020168197A1 (en) | 2019-02-15 | 2020-08-20 | Incyte Corporation | Pyrrolo[2,3-d]pyrimidinone compounds as cdk2 inhibitors |
| EP3924521A4 (en) | 2019-02-15 | 2023-03-29 | IncellDx, Inc. | Assaying bladder-associated samples, identifying and treating bladder-associated neoplasia, and kits for use therein |
| WO2020169472A2 (en) | 2019-02-18 | 2020-08-27 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods of inducing phenotypic changes in macrophages |
| US20220088075A1 (en) | 2019-02-22 | 2022-03-24 | The Trustees Of The University Of Pennsylvania | Combination therapies of egfrviii chimeric antigen receptors and pd-1 inhibitors |
| JP2022522668A (en) * | 2019-02-26 | 2022-04-20 | ツイスト バイオサイエンス コーポレーション | Mutant nucleic acid library for antibody optimization |
| CN113490529A (en) | 2019-02-28 | 2021-10-08 | 瑞泽恩制药公司 | Administration of PD-1 inhibitors for the treatment of skin cancer |
| US11472791B2 (en) | 2019-03-05 | 2022-10-18 | Incyte Corporation | Pyrazolyl pyrimidinylamine compounds as CDK2 inhibitors |
| CA3131529A1 (en) | 2019-03-05 | 2020-09-10 | Amgen Inc. | Use of oncolytic viruses for the treatment of cancer |
| KR20210136071A (en) | 2019-03-06 | 2021-11-16 | 리제너론 파아마슈티컬스, 인크. | IL-4/IL-13 Pathway Inhibitors for Enhanced Efficacy in Treating Cancer |
| US11628162B2 (en) | 2019-03-08 | 2023-04-18 | Incyte Corporation | Methods of treating cancer with an FGFR inhibitor |
| MX2021011320A (en) | 2019-03-19 | 2021-12-10 | Fundacio Privada Inst Dinvestigacio Oncològica De Vall Hebron | Combination therapy with omomyc and an antibody binding pd-1 or ctla-4 for the treatment of cancer. |
| US11793802B2 (en) | 2019-03-20 | 2023-10-24 | Sumitomo Pharma Oncology, Inc. | Treatment of acute myeloid leukemia (AML) with venetoclax failure |
| MX2021011289A (en) | 2019-03-22 | 2021-11-03 | Sumitomo Pharma Oncology Inc | Compositions comprising pkm2 modulators and methods of treatment using the same. |
| EP3946625A1 (en) | 2019-03-28 | 2022-02-09 | Bristol-Myers Squibb Company | Methods of treating tumor |
| KR20210146349A (en) | 2019-03-28 | 2021-12-03 | 브리스톨-마이어스 스큅 컴퍼니 | how to treat a tumor |
| TW202102543A (en) | 2019-03-29 | 2021-01-16 | 美商安進公司 | Use of oncolytic viruses in the neoadjuvant therapy of cancer |
| WO2020205560A1 (en) | 2019-03-29 | 2020-10-08 | Incyte Corporation | Sulfonylamide compounds as cdk2 inhibitors |
| JP2022527972A (en) | 2019-04-02 | 2022-06-07 | アンスティチュ ナショナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシュ メディカル | How to predict and prevent cancer in patients with premalignant lesions |
| WO2020208060A1 (en) | 2019-04-09 | 2020-10-15 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of sk2 inhibitors in combination with immune checkpoint blockade therapy for the treatment of cancer |
| US20220220480A1 (en) | 2019-04-17 | 2022-07-14 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treatment of nlrp3 inflammasome mediated il-1beta dependent disorders |
| WO2020223469A1 (en) | 2019-05-01 | 2020-11-05 | Incyte Corporation | N-(1-(methylsulfonyl)piperidin-4-yl)-4,5-di hydro-1h-imidazo[4,5-h]quinazolin-8-amine derivatives and related compounds as cyclin-dependent kinase 2 (cdk2) inhibitors for treating cancer |
| US11447494B2 (en) | 2019-05-01 | 2022-09-20 | Incyte Corporation | Tricyclic amine compounds as CDK2 inhibitors |
| CA3139410A1 (en) | 2019-05-07 | 2020-11-12 | Immunicom, Inc. | Increasing responses to checkpoint inhibitors by extracorporeal apheresis |
| JP2022532490A (en) | 2019-05-13 | 2022-07-15 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | Combination of PD-1 and LAG-3 inhibitors to enhance efficacy in the treatment of cancer |
| WO2020243563A1 (en) | 2019-05-30 | 2020-12-03 | Bristol-Myers Squibb Company | Multi-tumor gene signatures for suitability to immuno-oncology therapy |
| EP3976832A1 (en) | 2019-05-30 | 2022-04-06 | Bristol-Myers Squibb Company | Methods of identifying a subject suitable for an immuno-oncology (i-o) therapy |
| WO2020243570A1 (en) | 2019-05-30 | 2020-12-03 | Bristol-Myers Squibb Company | Cell localization signature and combination therapy |
| US20210038684A1 (en) | 2019-06-11 | 2021-02-11 | Alkermes Pharma Ireland Limited | Compositions and Methods for Cancer Immunotherapy |
| US20220251604A1 (en) | 2019-06-14 | 2022-08-11 | Tilt Biotherapeutics Oy | Oncolytic adenovirus and checkpoint inhibitor combination therapy |
| CA3143634A1 (en) | 2019-06-18 | 2020-12-24 | Janssen Sciences Ireland Unlimited Company | Combination of hepatitis b virus (hbv) vaccines and anti-pd-1 antibody |
| MA56523A (en) | 2019-06-18 | 2022-04-27 | Janssen Sciences Ireland Unlimited Co | COMBINATION OF HEPATITIS B VIRUS (HBV) VACCINES AND ANTI-PD-1 OR ANTI-PD-L1 ANTIBODIES |
| EP3987010A1 (en) | 2019-06-21 | 2022-04-27 | HCW Biologics, Inc. | Multi-chain chimeric polypeptides and uses thereof |
| CA3144644A1 (en) | 2019-06-21 | 2020-12-24 | Twist Bioscience Corporation | Barcode-based nucleic acid sequence assembly |
| MX2021015433A (en) | 2019-07-02 | 2022-06-08 | Hutchinson Fred Cancer Res | Recombinant ad35 vectors and related gene therapy improvements. |
| CA3145864A1 (en) | 2019-07-03 | 2021-01-07 | Sumitomo Dainippon Pharma Oncology, Inc. | Tyrosine kinase non-receptor 1 (tnk1) inhibitors and uses thereof |
| WO2021007269A1 (en) | 2019-07-09 | 2021-01-14 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| CN110384657A (en) * | 2019-07-15 | 2019-10-29 | 三峡大学 | Target the preparation method and the application on the drug that preparation inhibits cervical carcinoma that PD-L1 carries miR-34a microvesicle |
| CN114514032A (en) | 2019-08-02 | 2022-05-17 | 兰提欧派普有限公司 | Angiotensin type 2 (AT2) receptor agonists for the treatment of cancer |
| WO2021024020A1 (en) | 2019-08-06 | 2021-02-11 | Astellas Pharma Inc. | Combination therapy involving antibodies against claudin 18.2 and immune checkpoint inhibitors for treatment of cancer |
| CA3149494A1 (en) | 2019-08-12 | 2021-02-18 | Purinomia Biotech, Inc. | Methods and compositions for promoting and potentiating t-cell mediated immune responses through adcc targeting of cd39 expressing cells |
| WO2021030537A1 (en) | 2019-08-14 | 2021-02-18 | Incyte Corporation | Imidazolyl pyrimidinylamine compounds as cdk2 inhibitors |
| GB201912107D0 (en) | 2019-08-22 | 2019-10-09 | Amazentis Sa | Combination |
| CA3152027A1 (en) | 2019-08-30 | 2021-03-04 | Agenus Inc. | Anti-cd96 antibodies and methods of use thereof |
| BR112022004791A2 (en) | 2019-09-17 | 2022-06-21 | Bial R&D Invest S A | Substituted imidazole carboxamides and their use in the treatment of medical disorders |
| KR20220100859A (en) | 2019-09-17 | 2022-07-18 | 비알 - 알&디 인베스트먼츠, 에스.에이. | Substituted N-heterocyclic carboxamides as acid ceramidase inhibitors, and their use as medicaments |
| US20220380314A1 (en) | 2019-09-17 | 2022-12-01 | Bial - R&D Investments, S.A. | Substituted, saturated and unsaturated n-heterocyclic carboxamides and related compounds for their use in the treatment of medical disorders |
| WO2021053587A1 (en) | 2019-09-18 | 2021-03-25 | Klaus Strein | Bispecific antibodies against ceacam5 and cd3 |
| WO2021053556A1 (en) | 2019-09-18 | 2021-03-25 | Novartis Ag | Nkg2d fusion proteins and uses thereof |
| EP4031578A1 (en) | 2019-09-18 | 2022-07-27 | Novartis AG | Entpd2 antibodies, combination therapies, and methods of using the antibodies and combination therapies |
| TW202124446A (en) | 2019-09-18 | 2021-07-01 | 瑞士商諾華公司 | Combination therapies with entpd2 antibodies |
| WO2021055994A1 (en) | 2019-09-22 | 2021-03-25 | Bristol-Myers Squibb Company | Quantitative spatial profiling for lag-3 antagonist therapy |
| AU2020356471A1 (en) | 2019-09-23 | 2022-04-21 | Twist Bioscience Corporation | Variant nucleic acid libraries for CRTH2 |
| KR20220069964A (en) | 2019-09-25 | 2022-05-27 | 씨젠 인크. | Anti-CD30 ADC, Anti-PD-1 and Chemotherapeutic Agent Combination for Treatment of Hematopoietic Cancer |
| CA3152263A1 (en) | 2019-09-25 | 2021-04-01 | Julia SANTUCCI PEREIRA DEL BUONO | Composite biomarker for cancer therapy |
| KR20220070005A (en) | 2019-09-26 | 2022-05-27 | 노파르티스 아게 | Antiviral pyrazolopyridinone compounds |
| EP3800201A1 (en) | 2019-10-01 | 2021-04-07 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Cd28h stimulation enhances nk cell killing activities |
| CA3151322A1 (en) | 2019-10-01 | 2021-04-08 | Silverback Therapeutics, Inc. | Combination therapy with immune stimulatory conjugates |
| CA3157024A1 (en) | 2019-10-03 | 2021-04-08 | Xencor, Inc. | Targeted il-12 heterodimeric fc-fusion proteins |
| EP4037710A1 (en) | 2019-10-04 | 2022-08-10 | Institut National de la Santé et de la Recherche Médicale (INSERM) | Methods and pharmaceutical composition for the treatment of ovarian cancer, breast cancer or pancreatic cancer |
| TW202128757A (en) | 2019-10-11 | 2021-08-01 | 美商建南德克公司 | Pd-1 targeted il-15/il-15ralpha fc fusion proteins with improved properties |
| PE20221905A1 (en) | 2019-10-11 | 2022-12-23 | Incyte Corp | BICYCLIC AMINES AS INHIBITORS OF CDK2 |
| MX2022004513A (en) | 2019-10-14 | 2022-07-19 | Incyte Corp | Bicyclic heterocycles as fgfr inhibitors. |
| WO2021076728A1 (en) | 2019-10-16 | 2021-04-22 | Incyte Corporation | Bicyclic heterocycles as fgfr inhibitors |
| KR20220103947A (en) | 2019-10-21 | 2022-07-25 | 노파르티스 아게 | Combination Therapy with Venetoclax and TIM-3 Inhibitors |
| AU2020370832A1 (en) | 2019-10-21 | 2022-05-19 | Novartis Ag | TIM-3 inhibitors and uses thereof |
| NL2024108B1 (en) | 2019-10-26 | 2021-07-19 | Vitroscan B V | Methods and apparatus for measuring immune-cell mediated anti-tumoroid responses |
| MX2022005371A (en) | 2019-11-05 | 2022-08-04 | Celgene Corp | Combination therapy with 2-(4-chlorophenyl)-n-((2-(2,6-dioxopiper idin-3-yl)-1-oxoisoindolin-5-yl) methyl)-2,2-difluoroacetamide. |
| EP4055392A1 (en) | 2019-11-05 | 2022-09-14 | Bristol-Myers Squibb Company | M-protein assays and uses thereof |
| WO2021092221A1 (en) | 2019-11-06 | 2021-05-14 | Bristol-Myers Squibb Company | Methods of identifying a subject with a tumor suitable for a checkpoint inhibitor therapy |
| WO2021092220A1 (en) | 2019-11-06 | 2021-05-14 | Bristol-Myers Squibb Company | Methods of identifying a subject with a tumor suitable for a checkpoint inhibitor therapy |
| AU2020380384A1 (en) | 2019-11-08 | 2022-05-26 | Bristol-Myers Squibb Company | LAG-3 antagonist therapy for melanoma |
| MX2022005651A (en) | 2019-11-11 | 2022-07-27 | Incyte Corp | Salts and crystalline forms of a pd-1/pd-l1 inhibitor. |
| WO2021097256A1 (en) | 2019-11-14 | 2021-05-20 | Cohbar, Inc. | Cxcr4 antagonist peptides |
| US20230000864A1 (en) | 2019-11-22 | 2023-01-05 | Sumitomo Pharma Oncology, Inc. | Solid dose pharmaceutical composition |
| BR112022009679A2 (en) | 2019-11-26 | 2022-08-09 | Novartis Ag | CD19 AND CD22 CHIMERIC ANTIGEN RECEPTORS AND USES THEREOF |
| EP3831849A1 (en) | 2019-12-02 | 2021-06-09 | LamKap Bio beta AG | Bispecific antibodies against ceacam5 and cd47 |
| BR112022010664A2 (en) | 2019-12-04 | 2022-08-16 | Incyte Corp | DERIVATIVES OF A FGFR INHIBITOR |
| JP2023505258A (en) | 2019-12-04 | 2023-02-08 | インサイト・コーポレイション | Tricyclic heterocycles as FGFR inhibitors |
| JP2023504568A (en) | 2019-12-04 | 2023-02-03 | オルナ セラピューティクス インコーポレイテッド | Circular RNA compositions and methods |
| US11897950B2 (en) | 2019-12-06 | 2024-02-13 | Augusta University Research Institute, Inc. | Osteopontin monoclonal antibodies |
| WO2021119105A1 (en) | 2019-12-09 | 2021-06-17 | Seagen Inc. | Combination therapy with liv1-adc and pd-1 antagonist |
| WO2021127554A1 (en) | 2019-12-19 | 2021-06-24 | Bristol-Myers Squibb Company | Combinations of dgk inhibitors and checkpoint antagonists |
| MX2022007759A (en) | 2019-12-20 | 2022-07-19 | Novartis Ag | Combination of anti tim-3 antibody mbg453 and anti tgf-beta antibody nis793, with or without decitabine or the anti pd-1 antibody spartalizumab, for treating myelofibrosis and myelodysplastic syndrome. |
| WO2021138407A2 (en) | 2020-01-03 | 2021-07-08 | Marengo Therapeutics, Inc. | Multifunctional molecules that bind to cd33 and uses thereof |
| CN115279766A (en) | 2020-01-03 | 2022-11-01 | 因赛特公司 | Combination therapy comprising A2A/A2B and PD-1/PD-L1 inhibitors |
| US12012409B2 (en) | 2020-01-15 | 2024-06-18 | Incyte Corporation | Bicyclic heterocycles as FGFR inhibitors |
| KR20220128389A (en) | 2020-01-17 | 2022-09-20 | 노파르티스 아게 | A combination comprising a TIM-3 inhibitor and a hypomethylating agent for use in treating myelodysplastic syndrome or chronic myelomonocytic leukemia |
| TW202136318A (en) | 2020-01-28 | 2021-10-01 | 美商建南德克公司 | Il15/il15r alpha heterodimeric fc-fusion proteins for the treatment of cancer |
| US20230086099A1 (en) | 2020-01-30 | 2023-03-23 | Ona Therapeutics, S.L. | Combination therapy for treatment of cancer and cancer metastasis |
| JP2023514152A (en) | 2020-02-06 | 2023-04-05 | ブリストル-マイヤーズ スクイブ カンパニー | IL-10 and its uses |
| IL295083A (en) | 2020-02-11 | 2022-09-01 | Hcw Biologics Inc | Methods of activating regulatory t cells |
| AU2021219720A1 (en) | 2020-02-11 | 2022-08-04 | Immunitybio, Inc. | Chromatography resin and uses thereof |
| CA3169231A1 (en) | 2020-02-11 | 2021-08-19 | HCW Biologics, Inc. | Methods of treating age-related and inflammatory diseases |
| TW202146452A (en) | 2020-02-28 | 2021-12-16 | 瑞士商諾華公司 | Dosing of a bispecific antibody that binds cd123 and cd3 |
| AU2021225491A1 (en) | 2020-02-28 | 2022-10-20 | Novartis Ag | A triple pharmaceutical combination comprising dabrafenib, an Erk inhibitor and a RAF inhibitor |
| CA3170369A1 (en) | 2020-03-05 | 2022-04-14 | Michal Shahar | Methods and compositions for treating cancer with immune cells |
| CN115768465A (en) | 2020-03-06 | 2023-03-07 | Ona疗法有限公司 | anti-CD 36 antibodies and their use to treat cancer |
| EP4114401A1 (en) | 2020-03-06 | 2023-01-11 | Incyte Corporation | Combination therapy comprising axl/mer and pd-1/pd-l1 inhibitors |
| WO2021177822A1 (en) | 2020-03-06 | 2021-09-10 | Stichting Het Nederlands Kanker Instituut-Antoni van Leeuwenhoek Ziekenhuis | Modulating anti-tumor immunity |
| US20230093147A1 (en) | 2020-03-09 | 2023-03-23 | President And Fellows Of Harvard College | Methods and compositions relating to improved combination therapies |
| WO2021189059A2 (en) | 2020-03-20 | 2021-09-23 | Orna Therapeutics, Inc. | Circular rna compositions and methods |
| WO2021194942A1 (en) | 2020-03-23 | 2021-09-30 | Bristol-Myers Squibb Company | Anti-ccr8 antibodies for treating cancer |
| WO2021207689A2 (en) | 2020-04-10 | 2021-10-14 | Juno Therapeutics, Inc. | Methods and uses related to cell therapy engineered with a chimeric antigen receptor targeting b-cell maturation antigen |
| EP4135844A1 (en) | 2020-04-16 | 2023-02-22 | Incyte Corporation | Fused tricyclic kras inhibitors |
| KR20230004635A (en) | 2020-04-21 | 2023-01-06 | 노파르티스 아게 | Dosage regimen for treating diseases modulated by CSF-1R |
| AU2021262794A1 (en) | 2020-04-29 | 2022-11-24 | Immunitybio, Inc. | Anti-CD26 proteins and uses thereof |
| US20230181756A1 (en) | 2020-04-30 | 2023-06-15 | Novartis Ag | Ccr7 antibody drug conjugates for treating cancer |
| MX2022013864A (en) | 2020-05-05 | 2023-03-09 | Teon Therapeutics Inc | Cannabinoid receptor type 2 (cb2) modulators and uses thereof. |
| WO2021231526A1 (en) | 2020-05-13 | 2021-11-18 | Incyte Corporation | Fused pyrimidine compounds as kras inhibitors |
| JP2023528017A (en) | 2020-05-26 | 2023-07-03 | アンセルム(アンスティチュート・ナシオナル・ドゥ・ラ・サンテ・エ・ドゥ・ラ・ルシェルシュ・メディカル) | Severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) polypeptide and its use for vaccine purposes |
| MX2022014734A (en) | 2020-05-26 | 2023-03-15 | Regeneron Pharma | Methods of treating cervical cancer by administering the pd-1 inhibitor antibody cemiplimab. |
| CA3180060A1 (en) | 2020-05-29 | 2021-12-02 | Zongmin ZHAO | Living cells engineered with polyphenol-functionalized biologically active nanocomplexes |
| US12024545B2 (en) | 2020-06-01 | 2024-07-02 | HCW Biologics, Inc. | Methods of treating aging-related disorders |
| WO2021247003A1 (en) | 2020-06-01 | 2021-12-09 | HCW Biologics, Inc. | Methods of treating aging-related disorders |
| WO2021247604A1 (en) | 2020-06-01 | 2021-12-09 | HCW Biologics, Inc. | Methods of treating aging-related disorders |
| AU2021288224A1 (en) | 2020-06-11 | 2023-01-05 | Novartis Ag | ZBTB32 inhibitors and uses thereof |
| TW202214857A (en) | 2020-06-19 | 2022-04-16 | 法商昂席歐公司 | New conjugated nucleic acid molecules and their uses |
| CN115916199A (en) | 2020-06-23 | 2023-04-04 | 诺华股份有限公司 | Dosing regimens comprising 3- (1-oxoisoindolin-2-yl) piperidine-2, 6-dione derivatives |
| US20230235077A1 (en) | 2020-06-24 | 2023-07-27 | The General Hospital Corporation | Materials and methods of treating cancer |
| KR20230027082A (en) | 2020-06-25 | 2023-02-27 | 셀진 코포레이션 | Methods of treating cancer using combination therapy |
| UY39298A (en) | 2020-06-26 | 2022-01-31 | Amgen Inc | IL-10 MUTEINS AND FUSION PROTEINS THEREOF |
| JP2023531305A (en) | 2020-06-30 | 2023-07-21 | アンスティチュ ナショナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシュ メディカル | A method for predicting the risk of recurrence and/or death in patients with solid tumors after neoadjuvant therapy. |
| JP2023531290A (en) | 2020-06-30 | 2023-07-21 | アンスティチュ ナショナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシュ メディカル | A method for predicting the risk of recurrence and/or death in patients with solid tumors after neoadjuvant therapy and definitive surgery |
| AU2021305052A1 (en) | 2020-07-07 | 2023-02-09 | Celgene Corporation | Pharmaceutical compositions comprising (S)-4-(4-(4-(((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-4-yl)oxy)methyl) benzyl)piperazin-1-yl)-3-fluorobenzonitrile and methods of using the same |
| MX2023000197A (en) | 2020-07-07 | 2023-02-22 | BioNTech SE | Therapeutic rna for hpv-positive cancer. |
| WO2022009157A1 (en) | 2020-07-10 | 2022-01-13 | Novartis Ag | Lhc165 and spartalizumab combinations for treating solid tumors |
| EP4188549A1 (en) | 2020-08-03 | 2023-06-07 | Novartis AG | Heteroaryl substituted 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives and uses thereof |
| TW202227490A (en) * | 2020-08-19 | 2022-07-16 | 美商潘迪恩營運公司 | Multi-paratopic anti-pd-1 antibodies and uses thereof |
| CN116761818A (en) | 2020-08-26 | 2023-09-15 | 马伦戈治疗公司 | Method for detecting TRBC1 or TRBC2 |
| CA3168743A1 (en) | 2020-08-26 | 2022-03-03 | Matthew G. Fury | Methods of treating cancer by administering a pd-1 inhibitor |
| AU2021331476A1 (en) | 2020-08-28 | 2023-05-04 | Bristol-Myers Squibb Company | Lag-3 antagonist therapy for hepatocellular carcinoma |
| US11999752B2 (en) | 2020-08-28 | 2024-06-04 | Incyte Corporation | Vinyl imidazole compounds as inhibitors of KRAS |
| WO2022043557A1 (en) | 2020-08-31 | 2022-03-03 | Advanced Accelerator Applications International Sa | Method of treating psma-expressing cancers |
| WO2022043558A1 (en) | 2020-08-31 | 2022-03-03 | Advanced Accelerator Applications International Sa | Method of treating psma-expressing cancers |
| JP2023538955A (en) | 2020-08-31 | 2023-09-12 | ブリストル-マイヤーズ スクイブ カンパニー | Cellular localization signatures and immunotherapy |
| WO2022072783A1 (en) | 2020-10-02 | 2022-04-07 | Incyte Corporation | Bicyclic dione compounds as inhibitors of kras |
| WO2022072762A1 (en) | 2020-10-02 | 2022-04-07 | Regeneron Pharmaceuticals, Inc. | Combination of antibodies for treating cancer with reduced cytokine release syndrome |
| KR20230080460A (en) | 2020-10-05 | 2023-06-07 | 브리스톨-마이어스 스큅 컴퍼니 | How to Concentrate Protein |
| MX2023004493A (en) | 2020-10-23 | 2023-05-10 | Bristol Myers Squibb Co | Lag-3 antagonist therapy for lung cancer. |
| EP4236960A1 (en) | 2020-10-28 | 2023-09-06 | Ikena Oncology, Inc. | Combination of an ahr inhibitor with a pdx inhibitor or doxorubicine |
| WO2022097060A1 (en) | 2020-11-06 | 2022-05-12 | Novartis Ag | Cd19 binding molecules and uses thereof |
| US11780836B2 (en) | 2020-11-06 | 2023-10-10 | Incyte Corporation | Process of preparing a PD-1/PD-L1 inhibitor |
| TW202233615A (en) | 2020-11-06 | 2022-09-01 | 美商英塞特公司 | Crystalline form of a pd-1/pd-l1 inhibitor |
| KR20230117573A (en) | 2020-11-06 | 2023-08-08 | 인사이트 코포레이션 | Methods for preparing PD-1 and PD-L1 inhibitors and salts and crystalline forms thereof |
| KR20230104659A (en) | 2020-11-08 | 2023-07-10 | 씨젠 인크. | Combination-Therapy Antibody Drug Conjugates Using Immune Cell Inhibitors |
| JP2023554587A (en) | 2020-11-12 | 2023-12-28 | アンセルム(アンスティチュート・ナシオナル・ドゥ・ラ・サンテ・エ・ドゥ・ラ・ルシェルシュ・メディカル) | Antibodies conjugated or fused to the receptor binding domain of the SARS-COV-2 spike protein and their use for vaccine purposes |
| WO2022101463A1 (en) | 2020-11-16 | 2022-05-19 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of the last c-terminal residues m31/41 of zikv m ectodomain for triggering apoptotic cell death |
| TW202227089A (en) | 2020-11-30 | 2022-07-16 | 大陸商杭州阿諾生物醫藥科技有限公司 | Combination therapy for the treatment of pik3ca mutant cancer |
| WO2022120179A1 (en) | 2020-12-03 | 2022-06-09 | Bristol-Myers Squibb Company | Multi-tumor gene signatures and uses thereof |
| MX2023006604A (en) | 2020-12-04 | 2023-06-19 | Tidal Therapeutics Inc | Ionizable cationic lipids and lipid nanoparticles, and methods of synthesis and use thereof. |
| EP4259149A1 (en) | 2020-12-08 | 2023-10-18 | Infinity Pharmaceuticals, Inc. | Eganelisib for use in the treatment of pd-l1 negative cancer |
| TW202237119A (en) | 2020-12-10 | 2022-10-01 | 美商住友製藥腫瘤公司 | Alk-5 inhibitors and uses thereof |
| HUE065728T2 (en) | 2020-12-18 | 2024-06-28 | Lamkap Bio Beta Ag | Bispecific antibodies against ceacam5 and cd47 |
| WO2022135667A1 (en) | 2020-12-21 | 2022-06-30 | BioNTech SE | Therapeutic rna for treating cancer |
| WO2022135666A1 (en) | 2020-12-21 | 2022-06-30 | BioNTech SE | Treatment schedule for cytokine proteins |
| TW202245808A (en) | 2020-12-21 | 2022-12-01 | 德商拜恩迪克公司 | Therapeutic rna for treating cancer |
| WO2022146948A1 (en) | 2020-12-28 | 2022-07-07 | Bristol-Myers Squibb Company | Subcutaneous administration of pd1/pd-l1 antibodies |
| WO2022146947A1 (en) | 2020-12-28 | 2022-07-07 | Bristol-Myers Squibb Company | Antibody compositions and methods of use thereof |
| JP2024502005A (en) | 2020-12-29 | 2024-01-17 | インサイト・コーポレイション | Combination therapy including A2A/A2B inhibitors, PD-1/PD-L1 inhibitors, and anti-CD73 antibodies |
| JP2024504923A (en) | 2021-01-11 | 2024-02-02 | シンセカイン インコーポレイテッド | Compositions and methods for receptor pairing |
| EP4284510A1 (en) | 2021-01-29 | 2023-12-06 | Novartis AG | Dosage regimes for anti-cd73 and anti-entpd2 antibodies and uses thereof |
| TW202241508A (en) | 2021-01-29 | 2022-11-01 | 美商艾歐凡斯生物治療公司 | Cytokine associated tumor infiltrating lymphocytes compositions and methods |
| US20240139198A1 (en) | 2021-02-10 | 2024-05-02 | Curon Biopharmaceutical (Shanghai) Co., Limited | Method and combination for treating tumors |
| TW202302148A (en) | 2021-02-12 | 2023-01-16 | 美商欣爍克斯公司 | Lung cancer combination therapy with il-2 conjugates and an anti-pd-1 antibody or antigen-binding fragment thereof |
| WO2022184937A1 (en) | 2021-03-05 | 2022-09-09 | Leadartis, S.L. | Trimeric polypeptides and uses thereof in the treatment of cancer |
| WO2022195551A1 (en) | 2021-03-18 | 2022-09-22 | Novartis Ag | Biomarkers for cancer and methods of use thereof |
| BR112023018950A2 (en) | 2021-03-19 | 2024-02-27 | Icahn School Med Mount Sinai | COMPOUND, NANOBIOLOGICAL AND PHARMACEUTICAL COMPOSITIONS, METHODS FOR TREATING A CELL PROLIFERATION DISORDER, FOR TREATING SEPSIS AND FOR ACTIVATING A NOD2 RECEPTOR, PROCESS FOR MANUFACTURING A NANOBIOLOGICAL COMPOSITION AND KIT |
| EP4313123A1 (en) | 2021-03-23 | 2024-02-07 | Regeneron Pharmaceuticals, Inc. | Methods of treating cancer in immunosuppressed or immunocompromised patients by administering a pd-1 inhibitor |
| TW202304506A (en) | 2021-03-25 | 2023-02-01 | 日商安斯泰來製藥公司 | Combination therapy involving antibodies against claudin 18.2 for treatment of cancer |
| US20240181052A1 (en) | 2021-03-29 | 2024-06-06 | Juno Therapeutics, Inc. | Methods for dosing and treatment with a combination of a checkpoint inhibitor therapy and a car t cell therapy |
| WO2022212876A1 (en) | 2021-04-02 | 2022-10-06 | The Regents Of The University Of California | Antibodies against cleaved cdcp1 and uses thereof |
| TW202304979A (en) | 2021-04-07 | 2023-02-01 | 瑞士商諾華公司 | USES OF ANTI-TGFβ ANTIBODIES AND OTHER THERAPEUTIC AGENTS FOR THE TREATMENT OF PROLIFERATIVE DISEASES |
| CA3214757A1 (en) | 2021-04-08 | 2022-10-13 | Andreas Loew | Multifuntional molecules binding to tcr and uses thereof |
| AU2022254727A1 (en) | 2021-04-09 | 2023-10-12 | Seagen Inc. | Methods of treating cancer with anti-tigit antibodies |
| EP4323405A1 (en) | 2021-04-12 | 2024-02-21 | Incyte Corporation | Combination therapy comprising an fgfr inhibitor and a nectin-4 targeting agent |
| PE20240327A1 (en) | 2021-04-13 | 2024-02-22 | Nuvalent Inc | HETEROCYCLES WITH AMINO SUBSTITUTION TO TREAT CANCERS WITH EGFR MUTATIONS |
| WO2022219080A1 (en) | 2021-04-14 | 2022-10-20 | INSERM (Institut National de la Santé et de la Recherche Médicale) | New method to improve nk cells cytotoxicity |
| WO2022221720A1 (en) | 2021-04-16 | 2022-10-20 | Novartis Ag | Antibody drug conjugates and methods for making thereof |
| MX2023012364A (en) | 2021-04-20 | 2023-11-01 | Seagen Inc | Modulation of antibody-dependent cellular cytotoxicity. |
| AR125874A1 (en) | 2021-05-18 | 2023-08-23 | Novartis Ag | COMBINATION THERAPIES |
| CN117396222A (en) | 2021-05-21 | 2024-01-12 | 天津立博美华基因科技有限责任公司 | Pharmaceutical combination and use thereof |
| WO2022254337A1 (en) | 2021-06-01 | 2022-12-08 | Novartis Ag | Cd19 and cd22 chimeric antigen receptors and uses thereof |
| WO2022261159A1 (en) | 2021-06-09 | 2022-12-15 | Incyte Corporation | Tricyclic heterocycles as fgfr inhibitors |
| CA3220274A1 (en) | 2021-06-09 | 2022-12-15 | Incyte Corporation | Tricyclic heterocycles as fgfr inhibitors |
| CN115466329A (en) * | 2021-06-11 | 2022-12-13 | 广东菲鹏制药股份有限公司 | anti-PD-1 humanized antibody and application thereof |
| US11981671B2 (en) | 2021-06-21 | 2024-05-14 | Incyte Corporation | Bicyclic pyrazolyl amines as CDK2 inhibitors |
| WO2023280790A1 (en) | 2021-07-05 | 2023-01-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Gene signatures for predicting survival time in patients suffering from renal cell carcinoma |
| TW202317565A (en) | 2021-07-07 | 2023-05-01 | 美商英塞特公司 | Tricyclic compounds as inhibitors of kras |
| KR20240046323A (en) | 2021-07-13 | 2024-04-08 | 비온테크 에스이 | Multispecific binding agent for CD40 and CD137 in combination therapy for cancer |
| JP2024529347A (en) | 2021-07-14 | 2024-08-06 | インサイト・コーポレイション | Tricyclic Compounds as Inhibitors of KRAS |
| CA3226163A1 (en) | 2021-07-14 | 2023-01-19 | Synthekine, Inc. | Methods and compositions for use in cell therapy of neoplastic disease |
| US20230139492A1 (en) | 2021-07-19 | 2023-05-04 | Regeneron Pharmaceuticals, Inc. | Combination of checkpoint inhibitors and an oncolytic virus for treating cancer |
| WO2023007472A1 (en) | 2021-07-30 | 2023-02-02 | ONA Therapeutics S.L. | Anti-cd36 antibodies and their use to treat cancer |
| EP4380980A1 (en) | 2021-08-03 | 2024-06-12 | F. Hoffmann-La Roche AG | Bispecific antibodies and methods of use |
| EP4380596A1 (en) | 2021-08-04 | 2024-06-12 | Genentech, Inc. | Il15/il15r alpha heterodimeric fc-fusion proteins for the expansion of nk cells in the treatment of solid tumours |
| MX2024002281A (en) | 2021-08-23 | 2024-05-20 | Immunitas Therapeutics Inc | Anti-cd161 antibodies and uses thereof. |
| EP4396187A1 (en) | 2021-08-31 | 2024-07-10 | Incyte Corporation | Naphthyridine compounds as inhibitors of kras |
| WO2023034530A1 (en) | 2021-09-02 | 2023-03-09 | Teon Therapeutics, Inc. | Methods of improving growth and function of immune cells |
| CA3231180A1 (en) | 2021-09-08 | 2023-03-16 | Redona Therapeutics, Inc. | Papd5 and/or papd7 inhibiting 4-oxo-1,4-dihydroquinoline-3-carboxylic acid derivatives |
| US12030883B2 (en) | 2021-09-21 | 2024-07-09 | Incyte Corporation | Hetero-tricyclic compounds as inhibitors of KRAS |
| WO2023051926A1 (en) | 2021-09-30 | 2023-04-06 | BioNTech SE | Treatment involving non-immunogenic rna for antigen vaccination and pd-1 axis binding antagonists |
| JP2024537824A (en) | 2021-10-01 | 2024-10-16 | インサイト・コーポレイション | Pyrazoloquinoline KRAS inhibitors |
| IL311771A (en) | 2021-10-06 | 2024-05-01 | BioNTech SE | Multispecific binding agents against pd-l1 and cd137 in combination |
| TW202333802A (en) | 2021-10-11 | 2023-09-01 | 德商拜恩迪克公司 | Therapeutic rna for lung cancer |
| IL312114A (en) | 2021-10-14 | 2024-06-01 | Incyte Corp | Quinoline compounds as inhibitors of kras |
| CN118159568A (en) | 2021-10-21 | 2024-06-07 | 杭州阿诺生物医药科技有限公司 | Fusion polypeptide and application thereof |
| CA3234821A1 (en) | 2021-10-28 | 2023-05-04 | Suman Kumar VODNALA | Methods for culturing immune cells |
| MX2024005053A (en) | 2021-10-29 | 2024-05-10 | Bristol Myers Squibb Co | Lag-3 antagonist therapy for hematological cancer. |
| WO2023081730A1 (en) | 2021-11-03 | 2023-05-11 | Teon Therapeutics, Inc. | 4-hydroxy-2-oxo-1,2-dihydro-1,8-naphthyridine-3-carboxamide derivatives as cannabinoid cb2 receptor modulators for the treatment of cancer |
| WO2023083439A1 (en) | 2021-11-09 | 2023-05-19 | BioNTech SE | Tlr7 agonist and combinations for cancer treatment |
| CA3238029A1 (en) * | 2021-11-11 | 2023-05-19 | Regeneron Pharmaceuticals, Inc. | Cd20-pd1 binding molecules and methods of use thereof |
| KR20240103007A (en) | 2021-11-12 | 2024-07-03 | 노파르티스 아게 | Combination therapy for lung cancer treatment |
| CA3238283A1 (en) | 2021-11-15 | 2023-05-19 | Yangxin Fu | Fusion protein construct taking interleukin 15 as active ingredient and use thereof |
| CN118765283A (en) | 2021-11-17 | 2024-10-11 | 国家健康科学研究所 | Universal sand Bei Bingdu vaccine |
| TW202320792A (en) | 2021-11-22 | 2023-06-01 | 美商英塞特公司 | Combination therapy comprising an fgfr inhibitor and a kras inhibitor |
| WO2023097211A1 (en) | 2021-11-24 | 2023-06-01 | The University Of Southern California | Methods for enhancing immune checkpoint inhibitor therapy |
| WO2023102184A1 (en) | 2021-12-03 | 2023-06-08 | Incyte Corporation | Bicyclic amine compounds as cdk12 inhibitors |
| US12084453B2 (en) | 2021-12-10 | 2024-09-10 | Incyte Corporation | Bicyclic amines as CDK12 inhibitors |
| US11976073B2 (en) | 2021-12-10 | 2024-05-07 | Incyte Corporation | Bicyclic amines as CDK2 inhibitors |
| IL313439A (en) | 2021-12-16 | 2024-08-01 | Valerio Therapeutics | New conjugated nucleic acid molecules and their uses |
| AR128043A1 (en) | 2021-12-22 | 2024-03-20 | Incyte Corp | SALTS AND SOLID FORMS OF AN FGFR INHIBITOR AND PROCESSES FOR THEIR PREPARATION |
| WO2023130081A1 (en) | 2021-12-30 | 2023-07-06 | Neoimmunetech, Inc. | Method of treating a tumor with a combination of il-7 protein and vegf antagonist |
| WO2023133424A2 (en) * | 2022-01-05 | 2023-07-13 | TCR2 Therapeutics Inc. | Compositions and methods for tcr reprogramming using fusion proteins and anti-pd-1 fusion peptides |
| WO2023133280A1 (en) | 2022-01-07 | 2023-07-13 | Regeneron Pharmaceuticals, Inc. | Methods of treating recurrent ovarian cancer with bispecific anti-muc16 x anti-cd3 antibodies alone or in combination with anti-pd-1 antibodies |
| MX2024008831A (en) | 2022-01-26 | 2024-07-25 | Bristol Myers Squibb Company | Combination therapy for hepatocellular carcinoma. |
| CN118742325A (en) | 2022-01-28 | 2024-10-01 | 上海岸阔医药科技有限公司 | Methods for preventing or treating diseases or conditions associated with antineoplastic agents |
| WO2023147488A1 (en) | 2022-01-28 | 2023-08-03 | Iovance Biotherapeutics, Inc. | Cytokine associated tumor infiltrating lymphocytes compositions and methods |
| TW202342474A (en) | 2022-02-14 | 2023-11-01 | 美商基利科學股份有限公司 | Antiviral pyrazolopyridinone compounds |
| WO2023154799A1 (en) | 2022-02-14 | 2023-08-17 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Combination immunotherapy for treating cancer |
| WO2023159102A1 (en) | 2022-02-17 | 2023-08-24 | Regeneron Pharmaceuticals, Inc. | Combinations of checkpoint inhibitors and oncolytic virus for treating cancer |
| WO2023155905A1 (en) | 2022-02-21 | 2023-08-24 | 上海岸阔医药科技有限公司 | Compound and use thereof |
| WO2023161453A1 (en) | 2022-02-24 | 2023-08-31 | Amazentis Sa | Uses of urolithins |
| AU2023226078A1 (en) | 2022-02-25 | 2024-08-22 | Bristol-Myers Squibb Company | Combination therapy for colorectal carcinoma. |
| WO2023168363A1 (en) | 2022-03-02 | 2023-09-07 | HCW Biologics, Inc. | Method of treating pancreatic cancer |
| WO2023168404A1 (en) | 2022-03-04 | 2023-09-07 | Bristol-Myers Squibb Company | Methods of treating a tumor |
| AR128717A1 (en) | 2022-03-07 | 2024-06-05 | Incyte Corp | SOLID FORMS, SALTS AND PREPARATION PROCESSES OF A CDK2 INHIBITOR |
| WO2023170606A1 (en) | 2022-03-08 | 2023-09-14 | Alentis Therapeutics Ag | Use of anti-claudin-1 antibodies to increase t cell availability |
| WO2023177772A1 (en) | 2022-03-17 | 2023-09-21 | Regeneron Pharmaceuticals, Inc. | Methods of treating recurrent epithelioid sarcoma with bispecific anti-muc16 x anti-cd3 antibodies alone or in combination with anti-pd-1 antibodies |
| WO2023178329A1 (en) | 2022-03-18 | 2023-09-21 | Bristol-Myers Squibb Company | Methods of isolating polypeptides |
| WO2023192478A1 (en) | 2022-04-01 | 2023-10-05 | Bristol-Myers Squibb Company | Combination therapy with anti-il-8 antibodies and anti-pd-1 antibodies for treating cancer |
| WO2023196987A1 (en) | 2022-04-07 | 2023-10-12 | Bristol-Myers Squibb Company | Methods of treating tumor |
| US20230326022A1 (en) | 2022-04-08 | 2023-10-12 | Bristol-Myers Squibb Company | Machine Learning Identification, Classification, and Quantification of Tertiary Lymphoid Structures |
| WO2023214325A1 (en) | 2022-05-05 | 2023-11-09 | Novartis Ag | Pyrazolopyrimidine derivatives and uses thereof as tet2 inhibitors |
| WO2023218046A1 (en) | 2022-05-12 | 2023-11-16 | Genmab A/S | Binding agents capable of binding to cd27 in combination therapy |
| WO2023224912A1 (en) | 2022-05-16 | 2023-11-23 | Regeneron Pharmaceuticals, Inc. | Methods of treating metastatic castration-resistant prostate cancer with bispecific anti-psma x anti-cd3 antibodies alone or in combination with anti-pd-1 antibodies |
| TW202410916A (en) | 2022-05-27 | 2024-03-16 | 日商武田藥品工業股份有限公司 | Dosing of cd38-binding fusion protein |
| WO2023235847A1 (en) | 2022-06-02 | 2023-12-07 | Bristol-Myers Squibb Company | Antibody compositions and methods of use thereof |
| US20240002331A1 (en) | 2022-06-08 | 2024-01-04 | Tidal Therapeutics, Inc. | Ionizable cationic lipids and lipid nanoparticles, and methods of synthesis and use thereof |
| US20230399342A1 (en) | 2022-06-08 | 2023-12-14 | Incyte Corporation | Tricyclic triazolo compounds as dgk inhibitors |
| AU2023291779A1 (en) | 2022-06-16 | 2024-10-17 | Lamkap Bio Beta Ltd | Combination therapy of bispecific antibodies against ceacam5 and cd47 and bispecific antibodies against ceacam5 and cd3 |
| WO2023250400A1 (en) | 2022-06-22 | 2023-12-28 | Juno Therapeutics, Inc. | Treatment methods for second line therapy of cd19-targeted car t cells |
| WO2023250430A1 (en) | 2022-06-22 | 2023-12-28 | Incyte Corporation | Bicyclic amine cdk12 inhibitors |
| US20240101557A1 (en) | 2022-07-11 | 2024-03-28 | Incyte Corporation | Fused tricyclic compounds as inhibitors of kras g12v mutants |
| WO2024015372A1 (en) | 2022-07-14 | 2024-01-18 | Teon Therapeutics, Inc. | Adenosine receptor antagonists and uses thereof |
| EP4310197A1 (en) | 2022-07-21 | 2024-01-24 | Fundación para la Investigación Biomédica del Hospital Universitario Puerta de Hierro Majadahonda | Method for identifying lung cancer patients for a combination treatment of immuno- and chemotherapy |
| WO2024030453A1 (en) | 2022-08-02 | 2024-02-08 | Regeneron Pharmaceuticals, Inc. | Methods of treating metastatic castration-resistant prostate cancer with bispecific anti-psma x anti-cd28 antibodies in combination with anti-pd-1 antibodies |
| US20240041929A1 (en) | 2022-08-05 | 2024-02-08 | Juno Therapeutics, Inc. | Chimeric antigen receptors specific for gprc5d and bcma |
| WO2024040175A1 (en) | 2022-08-18 | 2024-02-22 | Pulmatrix Operating Company, Inc. | Methods for treating cancer using inhaled angiogenesis inhibitor |
| WO2024052356A1 (en) | 2022-09-06 | 2024-03-14 | Institut National de la Santé et de la Recherche Médicale | Inhibitors of the ceramide metabolic pathway for overcoming immunotherapy resistance in cancer |
| WO2024069009A1 (en) | 2022-09-30 | 2024-04-04 | Alentis Therapeutics Ag | Treatment of drug-resistant hepatocellular carcinoma |
| US20240270849A1 (en) | 2022-10-03 | 2024-08-15 | Regeneron Pharmaceuticals, Inc. | Methods of treating cancer with bispecific egfr xcd28 antibodies alone or in combination with anti-pd-1 antibodies |
| WO2024084013A1 (en) | 2022-10-20 | 2024-04-25 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Combination therapy for the treatment of cancer |
| WO2024086827A2 (en) | 2022-10-20 | 2024-04-25 | Repertoire Immune Medicines, Inc. | Cd8 t cell targeted il2 |
| WO2024083988A1 (en) | 2022-10-20 | 2024-04-25 | Fundación Para La Investigación Médica Aplicada | Nanobodies for cancer therapy |
| WO2024102722A1 (en) | 2022-11-07 | 2024-05-16 | Neoimmunetech, Inc. | Methods of treating a tumor with an unmethylated mgmt promoter |
| TW202428272A (en) | 2022-11-18 | 2024-07-16 | 美商英塞特公司 | Heteroaryl fluoroalkenes as dgk inhibitors |
| WO2024115725A1 (en) | 2022-12-01 | 2024-06-06 | BioNTech SE | Multispecific antibody against cd40 and cd137 in combination therapy with anti-pd1 ab and chemotherapy |
| WO2024116140A1 (en) | 2022-12-01 | 2024-06-06 | Medimmune Limited | Combination therapy for treatment of cancer comprising anti-pd-l1 and anti-cd73 antibodies |
| US20240226298A1 (en) | 2022-12-13 | 2024-07-11 | Juno Therapeutics, Inc. | Chimeric antigen receptors specific for baff-r and cd19 and methods and uses thereof |
| WO2024126457A1 (en) | 2022-12-14 | 2024-06-20 | Astellas Pharma Europe Bv | Combination therapy involving bispecific binding agents binding to cldn18.2 and cd3 and immune checkpoint inhibitors |
| WO2024137776A1 (en) | 2022-12-21 | 2024-06-27 | Bristol-Myers Squibb Company | Combination therapy for lung cancer |
| WO2024150177A1 (en) | 2023-01-11 | 2024-07-18 | Advesya | Treatment methods for solid tumors |
| TW202428575A (en) | 2023-01-12 | 2024-07-16 | 美商英塞特公司 | Heteroaryl fluoroalkenes as dgk inhibitors |
| WO2024192033A1 (en) | 2023-03-13 | 2024-09-19 | Regeneron Pharmaceuticals, Inc. | Combination of pd-1 inhibitors and lag-3 inhibitors for enhanced efficacy in treating melanoma |
| WO2024196952A1 (en) | 2023-03-20 | 2024-09-26 | Bristol-Myers Squibb Company | Tumor subtype assessment for cancer therapy |
| WO2024209072A1 (en) | 2023-04-06 | 2024-10-10 | Genmab A/S | Multispecific binding agents against pd-l1 and cd137 for treating cancer |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5837845A (en) * | 1991-06-28 | 1998-11-17 | Mitsubishi Chemical Corporation | Human monoclonal antibody specifically binding to surface antigen of cancer cell membrane |
| EP0945464A1 (en) * | 1995-10-06 | 1999-09-29 | Cambridge Antibody Technology Limited | Specific binding members for human transforming growth factor beta; materials and methods |
| WO2001027272A1 (en) * | 1999-10-08 | 2001-04-19 | Hamilton Brook Smith & Reynold | Oculomedin and glaucoma |
| WO2001027279A1 (en) * | 1999-10-12 | 2001-04-19 | Cambridge Antibody Technology | Human anti-adipocyte monoclonal antibodies and their use |
| WO2002078731A1 (en) * | 2001-04-02 | 2002-10-10 | Wyeth | Module of pd-1 interactions with its ligands |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5859205A (en) * | 1989-12-21 | 1999-01-12 | Celltech Limited | Humanised antibodies |
| US5580756A (en) * | 1990-03-26 | 1996-12-03 | Bristol-Myers Squibb Co. | B7Ig fusion protein |
| JPH09500788A (en) | 1993-07-26 | 1997-01-28 | ダナ・ファーバー・キャンサー・インスティテュート・インコーポレイテッド | B7-2: CTLA4 / CD28 counter receptor |
| CA2143491C (en) * | 1994-03-01 | 2011-02-22 | Yasumasa Ishida | A novel peptide related to human programmed cell death and dna encoding it |
| IL147972A0 (en) * | 1999-08-23 | 2002-09-12 | Dana Farber Cancer Inst Inc Ge | Pd-1, a receptor for b7-4 and uses therefor |
| HUP0202882A2 (en) | 1999-08-23 | 2002-12-28 | Dana Farber Cancer Inst Inc | Novel b7-4 molecules and uses therefor |
| WO2003011911A1 (en) * | 2001-07-31 | 2003-02-13 | Ono Pharmaceutical Co., Ltd. | Substance specific to pd-1 |
| IL149820A0 (en) * | 2002-05-23 | 2002-11-10 | Curetech Ltd | Humanized immunomodulatory monoclonal antibodies for the treatment of neoplastic disease or immunodeficiency |
| ATE514713T1 (en) * | 2002-12-23 | 2011-07-15 | Wyeth Llc | ANTIBODIES TO PD-1 AND THEIR USE |
-
2003
- 2003-12-22 AT AT03780521T patent/ATE514713T1/en not_active IP Right Cessation
- 2003-12-22 MX MXPA05006828A patent/MXPA05006828A/en active IP Right Grant
- 2003-12-22 US US10/741,481 patent/US7488802B2/en not_active Expired - Fee Related
- 2003-12-22 ES ES03780521T patent/ES2367430T3/en not_active Expired - Lifetime
- 2003-12-22 BR BR0316880-8A patent/BR0316880A/en not_active IP Right Cessation
- 2003-12-22 JP JP2004561922A patent/JP4511943B2/en not_active Expired - Fee Related
- 2003-12-22 US US10/540,084 patent/US7521051B2/en not_active Expired - Fee Related
- 2003-12-22 AU AU2003288675A patent/AU2003288675B2/en not_active Ceased
- 2003-12-22 CA CA2508660A patent/CA2508660C/en not_active Expired - Fee Related
- 2003-12-22 CN CN2003801099298A patent/CN1753912B/en not_active Expired - Fee Related
- 2003-12-22 WO PCT/IB2003/006304 patent/WO2004056875A1/en active Application Filing
- 2003-12-22 EP EP03780521A patent/EP1576014B1/en not_active Expired - Lifetime
- 2003-12-22 CN CN2010101700224A patent/CN101899114A/en active Pending
-
2005
- 2005-06-14 IL IL169152A patent/IL169152A/en not_active IP Right Cessation
- 2005-07-12 NO NO20053389A patent/NO336442B1/en not_active IP Right Cessation
-
2006
- 2006-03-17 HK HK06103433.3A patent/HK1083510A1/en not_active IP Right Cessation
-
2007
- 2007-08-17 US US11/893,989 patent/US20080311117A1/en not_active Abandoned
-
2009
- 2009-03-16 US US12/405,058 patent/US8088905B2/en not_active Expired - Fee Related
-
2010
- 2010-03-16 JP JP2010059987A patent/JP2010189395A/en active Pending
- 2010-10-22 AU AU2010235966A patent/AU2010235966A1/en not_active Abandoned
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5837845A (en) * | 1991-06-28 | 1998-11-17 | Mitsubishi Chemical Corporation | Human monoclonal antibody specifically binding to surface antigen of cancer cell membrane |
| EP0945464A1 (en) * | 1995-10-06 | 1999-09-29 | Cambridge Antibody Technology Limited | Specific binding members for human transforming growth factor beta; materials and methods |
| WO2001027272A1 (en) * | 1999-10-08 | 2001-04-19 | Hamilton Brook Smith & Reynold | Oculomedin and glaucoma |
| WO2001027279A1 (en) * | 1999-10-12 | 2001-04-19 | Cambridge Antibody Technology | Human anti-adipocyte monoclonal antibodies and their use |
| WO2002078731A1 (en) * | 2001-04-02 | 2002-10-10 | Wyeth | Module of pd-1 interactions with its ligands |
Non-Patent Citations (3)
| Title |
|---|
| AGATA Y ET AL: "Expression of the PD-1 antigen on the surface of stimulated mouse T and B lymphocytes.", INTERNATIONAL IMMUNOLOGY. ENGLAND MAY 1996, vol. 8, no. 5, May 1996 (1996-05-01), pages 765 - 772, XP000971773, ISSN: 0953-8178 * |
| DAVIES J ET AL: "Affinity improvement of single antibody VH domains: residues in all three hypervariable regions affect antigen binding", IMMUNOTECHNOLOGY, ELSEVIER SCIENCE PUBLISHERS BV, NL, vol. 2, no. 3, 1 September 1996 (1996-09-01), pages 169 - 179, XP004070292, ISSN: 1380-2933 * |
| VAUGHAN T J ET AL: "HUMAN ANTIBODIES WITH SUB-NANOMOLAR AFFINITIES ISOLATED FROM A LARGE NON-IMMUNIZED PHAGE DISPLAY LIBRARY", NATURE BIOTECHNOLOGY, NATURE PUBLISHING, US, vol. 14, 1 March 1996 (1996-03-01), pages 309 - 314, XP000196144, ISSN: 1087-0156 * |
Cited By (638)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2243493A1 (en) * | 2002-07-03 | 2010-10-27 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| US8728474B2 (en) | 2002-07-03 | 2014-05-20 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| US9067999B1 (en) | 2002-07-03 | 2015-06-30 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| US9073994B2 (en) | 2002-07-03 | 2015-07-07 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| US9439962B2 (en) | 2002-07-03 | 2016-09-13 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| US8168179B2 (en) | 2002-07-03 | 2012-05-01 | Ono Pharmaceutical Co., Ltd. | Treatment method using anti-PD-L1 antibody |
| US9402899B2 (en) | 2002-07-03 | 2016-08-02 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| EP3287144A1 (en) * | 2002-07-03 | 2018-02-28 | ONO Pharmaceutical Co., Ltd. | Immunopotentiating compositions |
| US9393301B2 (en) | 2002-07-03 | 2016-07-19 | Ono Pharmaceutical Co., Ltd. | Immunopotentiative composition |
| US8088905B2 (en) | 2002-12-23 | 2012-01-03 | Wyeth | Nucleic acids encoding antibodies against PD-1 |
| US7488802B2 (en) | 2002-12-23 | 2009-02-10 | Wyeth | Antibodies against PD-1 |
| US7521051B2 (en) | 2002-12-23 | 2009-04-21 | Medimmune Limited | Methods of upmodulating adaptive immune response using anti-PD-1 antibodies |
| US9803015B2 (en) | 2004-10-06 | 2017-10-31 | Mayo Foundation For Medical Education And Research | Costimulatory B7-H1 in renal cell carcinoma patients: indicator of tumor aggressiveness and potential therapeutic target |
| US8747833B2 (en) | 2004-10-06 | 2014-06-10 | Mayo Foundation For Medical Education And Research | B7-H1 and methods of diagnosis, prognosis, and treatment of cancer |
| US11939378B2 (en) | 2004-10-06 | 2024-03-26 | Mayo Foundation For Medical Education And Research | Costimulatory B7-H1 in renal cell carcinoma patients: indicator of tumor aggressiveness and potential therapeutic target |
| EP1810026B1 (en) | 2004-10-06 | 2018-04-18 | Mayo Foundation For Medical Education And Research | B7-h1 and pd-1 in treatment of renal cell carcinoma |
| US11242387B2 (en) | 2004-10-06 | 2022-02-08 | Mayo Foundation For Medical Education And Research | Costimulatory B7-H1 in renal cell carcinoma patients: indicator of tumor aggressiveness and potential therapeutic target |
| US9492539B2 (en) | 2005-05-09 | 2016-11-15 | Ono Pharmaceutical Co., Ltd. | Monoclonal antibodies to Programmed Death 1 (PD-1) |
| EP2439273A3 (en) * | 2005-05-09 | 2012-10-03 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| US8008449B2 (en) | 2005-05-09 | 2011-08-30 | Medarex, Inc. | Human monoclonal antibodies to programmed death 1 (PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| JP2009155338A (en) * | 2005-05-09 | 2009-07-16 | Ono Pharmaceut Co Ltd | Cancer therapeutic method singly using human monoclonal antibody to programmed death 1 (pd-1) and anti-pd-1 antibody or concurrently using with other immunotherapy |
| NO341219B1 (en) * | 2005-05-09 | 2017-09-18 | Ono Pharmaceutical Co | Isolated human monoclonal antibody, immunoconjugate, composition and bispecific molecule |
| KR101339628B1 (en) * | 2005-05-09 | 2013-12-09 | 메다렉스, 인코포레이티드 | Human monoclonal antibodies to programmed death 1 (pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| US9084776B2 (en) | 2005-05-09 | 2015-07-21 | E.R. Squibb & Sons, L.L.C. | Methods for treating cancer using anti-PD-1 antibodies |
| CN105315373A (en) * | 2005-05-09 | 2016-02-10 | 小野药品工业株式会社 | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies |
| JP2021191793A (en) * | 2005-05-09 | 2021-12-16 | 小野薬品工業株式会社 | Human monoclonal antibody against programmed death 1 (pd-1) and method for treating cancer using anti-pd-1 antibody alone or in combination with other immunotherapy |
| EP2161336B2 (en) † | 2005-05-09 | 2017-03-29 | ONO Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| EP2418278A3 (en) * | 2005-05-09 | 2012-07-04 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| EP3530736A3 (en) * | 2005-05-09 | 2019-11-06 | ONO Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1 (pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| NO344818B1 (en) * | 2005-05-09 | 2020-05-04 | Ono Pharmaceutical Co | Monoclonal antibody or an antigen-binding portion thereof |
| US10441655B2 (en) | 2005-05-09 | 2019-10-15 | Ono Pharmaceutical Co., Ltd. | Monoclonal antibodies to programmed death 1 (PD-1) |
| US9358289B2 (en) | 2005-05-09 | 2016-06-07 | Ono Pharmaceutical Co., Ltd. | Methods for treating cancer using anti-PD-1 antibodies in combination with anti-CTLA-4 antibodies |
| JP7443302B2 (en) | 2005-05-09 | 2024-03-05 | 小野薬品工業株式会社 | Human monoclonal antibodies against Programmed Death 1 (PD-1) and anti-PD-1 antibodies alone or in combination with other immunotherapies to treat cancer |
| CN105315373B (en) * | 2005-05-09 | 2018-11-09 | 小野药品工业株式会社 | The human monoclonal antibodies of programmed death-1 (PD-1) and the method for carrying out treating cancer using anti-PD-1 antibody |
| US8779105B2 (en) | 2005-05-09 | 2014-07-15 | Medarex, L.L.C. | Monoclonal antibodies to programmed death 1 (PD-1) |
| JP2019103504A (en) * | 2005-05-09 | 2019-06-27 | 小野薬品工業株式会社 | Human monoclonal antibody against programmed death 1 (pd-1) and method for treating cancer using anti-pd-1 antibody alone or in combination with other immunotherapy |
| US9387247B2 (en) | 2005-05-09 | 2016-07-12 | Ono Pharmaceutical Co., Ltd. | Monoclonal antibodies to programmed death 1 (PD-1) |
| CN103059138A (en) * | 2005-05-09 | 2013-04-24 | 小野药品工业株式会社 | Human monoclonal antibodies to programmed death 1 (pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| US9492540B2 (en) | 2005-05-09 | 2016-11-15 | Ono Pharmaceutical Co., Ltd. | Methods for treating cancer using anti-PD-1 antibodies |
| EP2161336B1 (en) † | 2005-05-09 | 2013-07-31 | ONO Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| EP2439272A3 (en) * | 2005-05-09 | 2013-07-31 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| JP2014077015A (en) * | 2005-05-09 | 2014-05-01 | Ono Pharmaceut Co Ltd | Human monoclonal antibody to programmed death 1 (pd-1), and cancer therapeutic method singly using anti-pd-1 antibody or concurrently using with other immunotherapy |
| CN109485727A (en) * | 2005-05-09 | 2019-03-19 | 小野药品工业株式会社 | The human monoclonal antibodies of programmed death-1 (PD-1) and the method for carrying out treating cancer using anti-PD-1 antibody |
| KR101318469B1 (en) * | 2005-05-09 | 2013-10-23 | 메다렉스, 인코포레이티드 | Human monoclonal antibodies to programmed death 1 (pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| EP2439273B1 (en) | 2005-05-09 | 2019-02-27 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| EP4242655A3 (en) * | 2005-06-08 | 2024-02-21 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (pd-1)pathway |
| EP2397156B1 (en) | 2005-06-08 | 2016-11-02 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1)pathway |
| AU2012204032B2 (en) * | 2005-06-08 | 2014-01-16 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| US8652465B2 (en) | 2005-06-08 | 2014-02-18 | Emory University | Methods and compositions for the treatment of persistent infections |
| US11359013B2 (en) | 2005-06-08 | 2022-06-14 | Emory University | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| WO2006133396A3 (en) * | 2005-06-08 | 2007-08-09 | Dana Farber Cancer Inst Inc | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (pd-1) pathway |
| EP2397155A1 (en) * | 2005-06-08 | 2011-12-21 | Dana Farber Cancer Institute | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (pd-1)pathway |
| AU2006254902B8 (en) * | 2005-06-08 | 2012-08-16 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| US9457080B2 (en) | 2005-06-08 | 2016-10-04 | Emory University | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| RU2596491C2 (en) * | 2005-06-08 | 2016-09-10 | Дана-Фарбер Кэнсер Инститьют | Methods and compositions for treating persistent infections |
| AU2006254902A8 (en) * | 2005-06-08 | 2012-08-16 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| EP2397156A1 (en) * | 2005-06-08 | 2011-12-21 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1)pathway |
| WO2006133396A2 (en) * | 2005-06-08 | 2006-12-14 | Dana-Farber Cancer Institute | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (pd-1) pathway |
| RU2746383C2 (en) * | 2005-06-08 | 2021-04-12 | Дана-Фарбер Кэнсер Инститьют | Methods and compositions for treatment of persistent infections |
| AU2006254902B2 (en) * | 2005-06-08 | 2012-04-12 | Dana-Farber Cancer Institute, Inc. | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| US10370446B2 (en) | 2005-06-08 | 2019-08-06 | Emory University | Methods and compositions for the treatment of persistent infections and cancer by inhibiting the programmed cell death 1 (PD-1) pathway |
| US9580507B2 (en) | 2005-07-01 | 2017-02-28 | E.R. Squibb & Sons, L. L. C. | Human monoclonal antibodies to programmed death ligand 1 (PD-L1) |
| US9580505B2 (en) | 2005-07-01 | 2017-02-28 | E.R. Squibb & Sons, L. L. C. | Human monoclonal antibodies to programmed death ligand 1 (PD-L1) |
| US20160075782A1 (en) | 2005-07-01 | 2016-03-17 | E.R. Squibb & Sons, L. L. C. | Human monoclonal antibodies to programmed death ligand 1 (pd-l1) |
| WO2007100098A1 (en) | 2006-03-03 | 2007-09-07 | Kyoto University | Multimer of extracellular domain of cell surface functional molecule |
| EP2468765A1 (en) | 2006-03-03 | 2012-06-27 | ONO Pharmaceutical Co., Ltd. | Multimer of extracellular domain of cell surface functional molecule |
| EP2889309A1 (en) | 2006-03-03 | 2015-07-01 | Ono Pharmaceutical Co., Ltd. | Tetramer of extracellular domain of PD-L1 |
| US11624054B2 (en) | 2006-05-31 | 2023-04-11 | The Brigham And Women's Hospital, Inc. | ABCB5 positive mesenchymal stem cells as immunomodulators |
| US10017738B2 (en) | 2006-05-31 | 2018-07-10 | Children's Medical Center Corporation | ABCB5 positive mesenchymal stem cells as immunomodulators |
| US9855342B2 (en) | 2007-04-12 | 2018-01-02 | The Brigham And Women's Hospital, Inc. | Targeting ABCB5 for cancer therapy |
| US9533042B2 (en) | 2007-06-04 | 2017-01-03 | Genentech, Inc. | Anti-notch NRR antibodies and methods using same |
| US10005844B2 (en) | 2007-06-04 | 2018-06-26 | Genentech, Inc. | Polynucleotides encoding anti-Notch1 NRR antibody polypeptides |
| US8952136B2 (en) | 2007-06-18 | 2015-02-10 | Merck Sharp & Dohme B.V. | Antibodies to human programmed death receptor PD-1 |
| US9834605B2 (en) | 2007-06-18 | 2017-12-05 | Merck Sharpe & Dohme B.V. | Antibodies to human programmed death receptor PD-1 |
| US11117961B2 (en) | 2007-06-18 | 2021-09-14 | Merck Sharp & Dohme B.V. | Antibodies to human programmed death receptor PD-1 |
| US8354509B2 (en) | 2007-06-18 | 2013-01-15 | Msd Oss B.V. | Antibodies to human programmed death receptor PD-1 |
| US8900587B2 (en) | 2007-06-18 | 2014-12-02 | Merck Sharp & Dohme Corp. | Antibodies to human programmed death receptor PD-1 |
| EP2581441A1 (en) * | 2007-08-09 | 2013-04-17 | Genzyme Corporation | Method of treating autoimmune disease with mesenchymal stem cells |
| US8481022B2 (en) | 2007-08-09 | 2013-07-09 | Genzyme Corporation | Method of treating autoimmune disease with mesenchymal stem cells |
| WO2009023566A2 (en) * | 2007-08-09 | 2009-02-19 | Genzyme Corporation | Method of treating autoimmune disease with mesenchymal stem cells |
| WO2009023566A3 (en) * | 2007-08-09 | 2009-05-07 | Genzyme Corp | Method of treating autoimmune disease with mesenchymal stem cells |
| US8168757B2 (en) | 2008-03-12 | 2012-05-01 | Merck Sharp & Dohme Corp. | PD-1 binding proteins |
| EP2262837A2 (en) * | 2008-03-12 | 2010-12-22 | Merck Sharp & Dohme Corp. | Pd-1 binding proteins |
| EP2262837A4 (en) * | 2008-03-12 | 2011-04-06 | Merck Sharp & Dohme | Pd-1 binding proteins |
| US9650639B2 (en) | 2008-05-19 | 2017-05-16 | Advaxis, Inc. | Dual delivery system for heterologous antigens |
| US9644212B2 (en) | 2008-05-19 | 2017-05-09 | Advaxis, Inc. | Dual delivery system for heterologous antigens |
| US8609089B2 (en) | 2008-08-25 | 2013-12-17 | Amplimmune, Inc. | Compositions of PD-1 antagonists and methods of use |
| WO2010027423A2 (en) | 2008-08-25 | 2010-03-11 | Amplimmune, Inc. | Compositions of pd-1 antagonists and methods of use |
| US8709416B2 (en) | 2008-08-25 | 2014-04-29 | Amplimmune, Inc. | Compositions of PD-1 antagonists and methods of use |
| EP2927240A1 (en) | 2008-08-25 | 2015-10-07 | Amplimmune, Inc. | Compositions of pd-1 antagonists and methods of use |
| AU2009290543B2 (en) * | 2008-09-12 | 2015-09-03 | Oxford University Innovation Limited | PD-1 specific antibodies and uses thereof |
| US9181342B2 (en) | 2008-09-12 | 2015-11-10 | Isis Innovation Limited | PD-1 specific antibodies and uses thereof |
| US20110171215A1 (en) * | 2008-09-12 | 2011-07-14 | Isis Innovation Limited | Pd-1 specific antibodies and uses thereof |
| US8927697B2 (en) | 2008-09-12 | 2015-01-06 | Isis Innovation Limited | PD-1 specific antibodies and uses thereof |
| WO2010029434A1 (en) * | 2008-09-12 | 2010-03-18 | Isis Innovation Limited | Pd-1 specific antibodies and uses thereof |
| AU2009290544B2 (en) * | 2008-09-12 | 2015-07-16 | Oxford University Innovation Limited | PD-1 specific antibodies and uses thereof |
| WO2010029435A1 (en) * | 2008-09-12 | 2010-03-18 | Isis Innovation Limited | Pd-1 specific antibodies and uses thereof |
| US9683043B2 (en) | 2008-09-12 | 2017-06-20 | Oxford University Innovation Limited | PD-1 specific antibodies and uses thereof |
| US20110171220A1 (en) * | 2008-09-12 | 2011-07-14 | Isis Innovation Limited | Pd-1 specific antibodies and uses thereof |
| KR20190107756A (en) * | 2008-09-26 | 2019-09-20 | 다나-파버 캔서 인스티튜트 인크. | Human anti-pd-1, pd-l1, and pd-l2 antibodies and uses therefor |
| US11261251B2 (en) | 2008-09-26 | 2022-03-01 | Dana-Farber Cancer Institute, Inc. | Human anti-PD-1, PD-L1, and PD-L2 antibodies and uses therefor |
| KR102197527B1 (en) | 2008-09-26 | 2020-12-31 | 다나-파버 캔서 인스티튜트 인크. | Human anti-pd-1, pd-l1, and pd-l2 antibodies and uses therefor |
| US10370448B2 (en) | 2008-09-26 | 2019-08-06 | Emory University | Human anti-PD-1, PD-L1, and PD-L2 antibodies and uses therefor |
| EP3130923A1 (en) * | 2008-11-14 | 2017-02-15 | The Brigham and Women's Hospital, Inc. | Therapeutic and diagnostic methods relating to cancer stem cells |
| EP3978928A1 (en) * | 2008-11-14 | 2022-04-06 | The Brigham and Women's Hospital, Inc. | Therapeutic and diagnostic methods relating to cancer stem cells |
| US11542328B2 (en) | 2008-11-14 | 2023-01-03 | The Brigham And Women's Hospital, Inc. | Therapeutic and diagnostic methods relating to cancer stem cells |
| US10316085B2 (en) | 2008-11-14 | 2019-06-11 | Children's Medical Center Corporation | Therapeutic and diagnostic methods relating to cancer stem cells |
| US9598491B2 (en) | 2008-11-28 | 2017-03-21 | Emory University | Methods for the treatment of infections and tumors |
| US9920123B2 (en) | 2008-12-09 | 2018-03-20 | Genentech, Inc. | Anti-PD-L1 antibodies, compositions and articles of manufacture |
| EP3192811A1 (en) * | 2009-02-09 | 2017-07-19 | Université d'Aix-Marseille | Pd-1 antibodies and pd-l1 antibodies and uses thereof |
| US12042560B2 (en) | 2009-03-30 | 2024-07-23 | Eisai R&D Management Co., Ltd. | Liposome composition |
| US10016617B2 (en) | 2009-11-11 | 2018-07-10 | The Trustees Of The University Of Pennsylvania | Combination immuno therapy and radiotherapy for the treatment of Her-2-positive cancers |
| CN102892786A (en) * | 2010-03-11 | 2013-01-23 | Ucb医药有限公司 | Pd-1 antibody |
| WO2011110621A1 (en) | 2010-03-11 | 2011-09-15 | Ucb Pharma, S.A. | Biological products: humanised agonistic anti-pd-1 antibodies |
| US9102728B2 (en) | 2010-03-11 | 2015-08-11 | Ucb Biopharma Sprl | PD-1 antibodies |
| CN102892786B (en) * | 2010-03-11 | 2016-03-16 | Ucb医药有限公司 | Pd-1 antibody |
| WO2011110604A1 (en) | 2010-03-11 | 2011-09-15 | Ucb Pharma, S.A. | Pd-1 antibody |
| US8993731B2 (en) | 2010-03-11 | 2015-03-31 | Ucb Biopharma Sprl | PD-1 antibody |
| US8907053B2 (en) | 2010-06-25 | 2014-12-09 | Aurigene Discovery Technologies Limited | Immunosuppression modulating compounds |
| WO2011161699A2 (en) | 2010-06-25 | 2011-12-29 | Aurigene Discovery Technologies Limited | Immunosuppression modulating compounds |
| US9783578B2 (en) | 2010-06-25 | 2017-10-10 | Aurigene Discovery Technologies Limited | Immunosuppression modulating compounds |
| US9226958B2 (en) | 2010-10-01 | 2016-01-05 | University Of Georgia Research Foundation, Inc. | Use of Listeria vaccine vectors to reverse vaccine unresponsiveness in parasitically infected individuals |
| US9943590B2 (en) | 2010-10-01 | 2018-04-17 | The Trustees Of The University Of Pennsylvania | Use of Listeria vaccine vectors to reverse vaccine unresponsiveness in parasitically infected individuals |
| US9463227B2 (en) | 2011-03-11 | 2016-10-11 | Advaxis, Inc. | Listeria-based adjuvants |
| US10064898B2 (en) | 2011-03-11 | 2018-09-04 | Advaxis, Inc. | Listeria-based adjuvants |
| US9220776B2 (en) | 2011-03-31 | 2015-12-29 | Merck Sharp & Dohme Corp. | Stable formulations of antibodies to human programmed death receptor PD-1 and related treatments |
| WO2012145493A1 (en) | 2011-04-20 | 2012-10-26 | Amplimmune, Inc. | Antibodies and other molecules that bind b7-h1 and pd-1 |
| US9205148B2 (en) | 2011-04-20 | 2015-12-08 | Medimmune, Llc | Antibodies and other molecules that bind B7-H1 and PD-1 |
| EP2699264B1 (en) * | 2011-04-20 | 2018-03-14 | Medlmmune, LLC | Antibodies and other molecules that bind b7-h1 and pd-1 |
| RU2625034C2 (en) * | 2011-04-20 | 2017-07-11 | МЕДИММЬЮН, ЭлЭлСи | Antibodies and other molecules binding b7-h1 and pd-1 |
| EP3403672A1 (en) | 2011-04-20 | 2018-11-21 | Medlmmune, LLC | Antibodies and other molecules that bind b7-h1 and pd-1 |
| US9096642B2 (en) | 2011-06-08 | 2015-08-04 | Aurigene Discovery Technologies Limited | Therapeutic compounds for immunomodulation |
| WO2012168944A1 (en) | 2011-06-08 | 2012-12-13 | Aurigene Discovery Technologies Limited | Therapeutic compounds for immunomodulation |
| EP3939613A1 (en) | 2011-08-11 | 2022-01-19 | ONO Pharmaceutical Co., Ltd. | Therapeutic agent for autoimmune diseases comprising pd-1 agonist |
| WO2013022091A1 (en) | 2011-08-11 | 2013-02-14 | 小野薬品工業株式会社 | Therapeutic agent for autoimmune diseases comprising pd-1 agonist |
| WO2013132317A1 (en) | 2012-03-07 | 2013-09-12 | Aurigene Discovery Technologies Limited | Peptidomimetic compounds as immunomodulators |
| US10058599B2 (en) | 2012-03-12 | 2018-08-28 | Advaxis, Inc. | Suppressor cell function inhibition following Listeria vaccine treatment |
| US9422339B2 (en) | 2012-03-29 | 2016-08-23 | Aurigene Discovery Technologies Limited | Immunomodulating cyclic compounds |
| WO2013144704A1 (en) | 2012-03-29 | 2013-10-03 | Aurigene Discovery Technologies Limited | Immunomodulating cyclic compounds from the bc loop of human pd1 |
| US10709664B2 (en) | 2012-04-12 | 2020-07-14 | Yale University | Nanolipogel comprising a polymeric matrix and a lipid shell |
| US10603276B2 (en) | 2012-04-12 | 2020-03-31 | Yale University | Nanolipogel vehicles for controlled delivery of different pharmaceutical agents |
| US9610250B2 (en) | 2012-04-12 | 2017-04-04 | Yale University | Nanolipogel vehicles for controlled delivery of different pharmaceutical agents |
| US11173119B2 (en) | 2012-04-12 | 2021-11-16 | Yale University | Nanolipogel vehicles for controlled delivery of different pharmaceutical agents |
| US9603800B2 (en) | 2012-04-12 | 2017-03-28 | Yale University | Methods of treating inflammatory and autoimmune diseases and disorders using nanolipogels |
| US10500157B2 (en) | 2012-04-12 | 2019-12-10 | Yale University | Nanoparticle-mediated delivery of cytokines for maintenance of the regulatory T cell phenotype |
| US9856320B2 (en) | 2012-05-15 | 2018-01-02 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10316090B2 (en) | 2012-05-15 | 2019-06-11 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| AU2019202416B2 (en) * | 2012-05-15 | 2020-10-01 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting pd-1/pd-l1 signaling |
| US10072082B2 (en) | 2012-05-15 | 2018-09-11 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10316091B2 (en) | 2012-05-15 | 2019-06-11 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10308714B2 (en) | 2012-05-15 | 2019-06-04 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| WO2013173223A1 (en) * | 2012-05-15 | 2013-11-21 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting pd-1/pd-l1 signaling |
| EA037351B1 (en) * | 2012-05-15 | 2021-03-16 | Бристол-Майерс Сквибб Компани | Method of treating cancer using an anti-pd-1 and anti-ctl-4 antibody combination |
| US10266595B2 (en) | 2012-05-15 | 2019-04-23 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10323092B2 (en) | 2012-05-15 | 2019-06-18 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10577423B2 (en) | 2012-05-15 | 2020-03-03 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| CN113967253A (en) * | 2012-05-15 | 2022-01-25 | 百时美施贵宝公司 | Immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10604575B2 (en) | 2012-05-15 | 2020-03-31 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10584170B2 (en) | 2012-05-15 | 2020-03-10 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10323093B2 (en) | 2012-05-15 | 2019-06-18 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US9212224B2 (en) | 2012-05-15 | 2015-12-15 | Bristol-Myers Squibb Company | Antibodies that bind PD-L1 and uses thereof |
| US10266596B1 (en) | 2012-05-15 | 2019-04-23 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10138299B2 (en) | 2012-05-15 | 2018-11-27 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10266594B1 (en) | 2012-05-15 | 2019-04-23 | Bristol-Myers Squibb Company | Cancer immunotherapy by disrupting PD-1/PD-L1 signaling |
| US10660971B2 (en) | 2012-07-18 | 2020-05-26 | Birdie Biopharmaceuticals, Inc. | Compounds for targeted immunotherapy |
| US10548988B2 (en) | 2012-07-18 | 2020-02-04 | Birdie Biopharmaceuticals, Inc. | Compounds for targeted immunotherapy |
| WO2014122271A1 (en) | 2013-02-07 | 2014-08-14 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for predicting the survival time of patients suffering from diffuse large b-cell lymphomas |
| WO2014179664A3 (en) * | 2013-05-02 | 2015-02-19 | Anaptysbio, Inc. | Antibodies directed against programmed death-1 (pd-1) |
| US9815897B2 (en) | 2013-05-02 | 2017-11-14 | Anaptysbio, Inc. | Antibodies directed against programmed death-1 (PD-1) |
| US10738117B2 (en) | 2013-05-02 | 2020-08-11 | Anaptysbio, Inc. | Antibodies directed against programmed death-1 (PD-1) |
| WO2014194293A1 (en) | 2013-05-30 | 2014-12-04 | Amplimmune, Inc. | Improved methods for the selection of patients for pd-1 or b7-h4 targeted therapies, and combination therapies thereof |
| US9676853B2 (en) | 2013-05-31 | 2017-06-13 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind PD-1 |
| US11242391B2 (en) | 2013-05-31 | 2022-02-08 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind PD-1 |
| WO2014194302A3 (en) * | 2013-05-31 | 2015-02-26 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind pd-1 |
| EP4269441A2 (en) | 2013-08-08 | 2023-11-01 | Cytune Pharma | Il-15 and il-15ralpha sushi domain based on modulokines |
| EP3444271A1 (en) | 2013-08-08 | 2019-02-20 | Cytune Pharma | Il-15 and il-15raplha sushi domain based modulokines |
| EP3659622A1 (en) | 2013-08-08 | 2020-06-03 | Cytune Pharma | Combined pharmaceutical composition |
| EP3995507A1 (en) | 2013-08-08 | 2022-05-11 | Cytune Pharma | Il-15 and il-15ralpha sushi domain based on modulokines |
| US9771338B2 (en) | 2013-09-06 | 2017-09-26 | Auirgene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| EP3366289A1 (en) | 2013-09-06 | 2018-08-29 | Aurigene Discovery Technologies Limited | Cyclic peptidomimetic compounds as immunomodulators |
| US12037321B2 (en) | 2013-09-06 | 2024-07-16 | Aurigene Oncology Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| WO2015033303A1 (en) | 2013-09-06 | 2015-03-12 | Aurigene Discovery Technologies Limited | Cyclic peptidomimetic compounds as immunomodulators |
| EP3385257A1 (en) | 2013-09-06 | 2018-10-10 | Aurigene Discovery Technologies Limited | 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives as immunomodulators |
| US10106581B2 (en) | 2013-09-06 | 2018-10-23 | Aurigene Discovery Technologies Limited | Cyclic peptidomimetic compounds as immunomodulators |
| WO2015033299A1 (en) | 2013-09-06 | 2015-03-12 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| WO2015033301A1 (en) | 2013-09-06 | 2015-03-12 | Aurigene Discovery Technologies Limited | 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives as immunomodulators |
| US11512060B2 (en) | 2013-09-06 | 2022-11-29 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| US10961205B2 (en) | 2013-09-06 | 2021-03-30 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| EP3363790A1 (en) | 2013-09-06 | 2018-08-22 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| US9776978B2 (en) | 2013-09-06 | 2017-10-03 | Aurigene Discovery Technologies Limited | 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives as immunomodulators |
| US10160736B2 (en) | 2013-09-06 | 2018-12-25 | Aurigene Discovery Technologies Limited | 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives as immunomodulators |
| US10173989B2 (en) | 2013-09-06 | 2019-01-08 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| US10590093B2 (en) | 2013-09-06 | 2020-03-17 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole derivatives as immunomodulators |
| US10077305B2 (en) | 2013-09-10 | 2018-09-18 | Medimmune Limited | Antibodies against PD-1 and uses thereof |
| WO2015036394A1 (en) * | 2013-09-10 | 2015-03-19 | Medimmune Limited | Antibodies against pd-1 and uses thereof |
| US10570204B2 (en) | 2013-09-26 | 2020-02-25 | The Medical College Of Wisconsin, Inc. | Methods for treating hematologic cancers |
| US11708412B2 (en) | 2013-09-26 | 2023-07-25 | Novartis Ag | Methods for treating hematologic cancers |
| US10751291B2 (en) | 2013-11-01 | 2020-08-25 | Yale University | Nanoparticulate compositions comprising interferon gamma and losartan for immunotherapy |
| US9884026B2 (en) | 2013-11-01 | 2018-02-06 | Yale University | Modular particles for immunotherapy |
| US10780180B2 (en) | 2014-01-10 | 2020-09-22 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for immunotherapy |
| US10744206B2 (en) | 2014-01-10 | 2020-08-18 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for immunotherapy |
| US10548985B2 (en) | 2014-01-10 | 2020-02-04 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for treating EGFR expressing tumors |
| US11633494B2 (en) | 2014-01-10 | 2023-04-25 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for immunotherapy |
| US11633495B2 (en) | 2014-01-10 | 2023-04-25 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for immunotherapy |
| US10328158B2 (en) | 2014-01-10 | 2019-06-25 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for immunotherapy |
| US11786604B2 (en) | 2014-01-10 | 2023-10-17 | Birdie Biopharmaceuticals, Inc. | Compounds and compositions for treating HER2 positive tumors |
| CN106068275B (en) * | 2014-01-23 | 2021-06-01 | 瑞泽恩制药公司 | Human antibodies against PD-1 |
| CN106068275A (en) * | 2014-01-23 | 2016-11-02 | 瑞泽恩制药公司 | People's antibody of anti-PD 1 |
| US9938345B2 (en) | 2014-01-23 | 2018-04-10 | Regeneron Pharmaceuticals, Inc. | Human antibodies to PD-L1 |
| EA034770B1 (en) * | 2014-01-23 | 2020-03-18 | Ридженерон Фармасьютикалз, Инк. | Human antibodies to pd-1 |
| EP3967710A1 (en) * | 2014-01-23 | 2022-03-16 | Regeneron Pharmaceuticals, Inc. | Human antibodies to pd-1 |
| WO2015112800A1 (en) * | 2014-01-23 | 2015-07-30 | Regeneron Pharmaceuticals, Inc. | Human antibodies to pd-1 |
| US9987500B2 (en) | 2014-01-23 | 2018-06-05 | Regeneron Pharmaceuticals, Inc. | Human antibodies to PD-1 |
| US10737113B2 (en) | 2014-01-23 | 2020-08-11 | Regeneron Pharmaceuticals, Inc. | Human antibodies to PD-1 |
| US11117970B2 (en) | 2014-01-23 | 2021-09-14 | Regeneron Pharmaceuticals, Inc. | Human antibodies to PD-L1 |
| EA034770B8 (en) * | 2014-01-23 | 2020-07-10 | Ридженерон Фармасьютикалз, Инк. | Human antibodies to pd-1 |
| US9683048B2 (en) | 2014-01-24 | 2017-06-20 | Novartis Ag | Antibody molecules to PD-1 and uses thereof |
| US11827704B2 (en) | 2014-01-24 | 2023-11-28 | Novartis Ag | Antibody molecules to PD-1 and uses thereof |
| CN106103485A (en) * | 2014-01-24 | 2016-11-09 | 达纳-法伯癌症研究公司 | Antibody molecule of PD 1 and application thereof |
| CN106103485B (en) * | 2014-01-24 | 2020-06-30 | 达纳-法伯癌症研究公司 | Antibody molecules to PD-1 and uses thereof |
| WO2015112900A1 (en) * | 2014-01-24 | 2015-07-30 | Dana-Farber Cancer Institue, Inc. | Antibody molecules to pd-1 and uses thereof |
| KR102357621B1 (en) | 2014-01-24 | 2022-02-04 | 다나-파버 캔서 인스티튜트 인크. | Antibody molecules to pd-1 and uses thereof |
| US9815898B2 (en) | 2014-01-24 | 2017-11-14 | Novartis Ag | Antibody molecules to PD-1 and uses thereof |
| TWI692484B (en) * | 2014-01-24 | 2020-05-01 | 美商達納 法伯癌症協會 | Antibody molecules to pd-1 and uses thereof |
| KR20160110995A (en) * | 2014-01-24 | 2016-09-23 | 다나-파버 캔서 인스티튜트 인크. | Antibody molecules to pd-1 and uses thereof |
| US10752687B2 (en) | 2014-01-24 | 2020-08-25 | Novartis Ag | Antibody molecules to PD-1 and uses thereof |
| US10472419B2 (en) | 2014-01-31 | 2019-11-12 | Novartis Ag | Antibody molecules to TIM-3 and uses thereof |
| US11155620B2 (en) | 2014-01-31 | 2021-10-26 | Novartis Ag | Method of detecting TIM-3 using antibody molecules to TIM-3 |
| US10981990B2 (en) | 2014-01-31 | 2021-04-20 | Novartis Ag | Antibody molecules to TIM-3 and uses thereof |
| EP3971209A1 (en) | 2014-02-04 | 2022-03-23 | Pfizer Inc. | Combination of a pd-1 antagonist and a vegfr inhibitor for treating cancer |
| WO2015119944A1 (en) | 2014-02-04 | 2015-08-13 | Incyte Corporation | Combination of a pd-1 antagonist and an ido1 inhibitor for treating cancer |
| EP3498734A1 (en) | 2014-02-04 | 2019-06-19 | Pfizer Inc | Combination of a pd-1 antagonist and a vegfr inhibitor for treating cancer |
| US10899840B2 (en) | 2014-02-04 | 2021-01-26 | Pfizer Inc. | Combination of a PD-1 antagonist and a 4-1BB agonist for treating cancer |
| US10570202B2 (en) | 2014-02-04 | 2020-02-25 | Pfizer Inc. | Combination of a PD-1 antagonist and a VEGFR inhibitor for treating cancer |
| WO2015181624A2 (en) | 2014-05-28 | 2015-12-03 | Idenix Pharmaceuticals, Inc | Nucleoside derivatives for the treatment of cancer |
| AU2015276978B2 (en) * | 2014-06-19 | 2021-02-25 | Regeneron Pharmaceuticals, Inc. | Non-human animals having a humanized programmed cell death 1 gene |
| US11684050B2 (en) | 2014-06-19 | 2023-06-27 | Regeneran Pharmaceuticals, Inc. | Non-human animals having a humanized programmed cell death 1 gene |
| US10160806B2 (en) | 2014-06-26 | 2018-12-25 | Macrogenics, Inc. | Covalently bonded diabodies having immunoreactivity with PD-1 and LAG-3, and methods of use thereof |
| US11098119B2 (en) | 2014-06-26 | 2021-08-24 | Macrogenics, Inc. | Covalently bonded diabodies having immunoreactivity with PD-1 and LAG-3, and methods of use thereof |
| EP4001311A1 (en) | 2014-07-09 | 2022-05-25 | Birdie Biopharmaceuticals Inc. | Anti-pd-l1 combinations for treating tumors |
| WO2016004876A1 (en) | 2014-07-09 | 2016-01-14 | Shanghai Birdie Biotech, Inc. | Anti-pd-l1 combinations for treating tumors |
| US11279761B2 (en) | 2014-07-09 | 2022-03-22 | Birdie Biopharmaceuticals, Inc. | Anti-PD-L1 combinations for treating tumors |
| US11779619B2 (en) | 2014-07-16 | 2023-10-10 | Transgene Sa | Oncolytic virus for expression of immune checkpoint modulators |
| US10765710B2 (en) | 2014-07-16 | 2020-09-08 | Institut Gustave-Roussy | Combination of oncolytic virus with immune checkpoint modulators |
| US10555981B2 (en) | 2014-07-16 | 2020-02-11 | Transgene S.A. | Oncolytic virus for expression of immune checkpoint modulators |
| EP3552615A1 (en) | 2014-07-16 | 2019-10-16 | Transgene SA | Oncolytic virus for expression of immune checkpoint modulators |
| US9907849B2 (en) | 2014-07-18 | 2018-03-06 | Advaxis, Inc. | Combination of a PD-1 antagonist and a listeria-based vaccine for treating prostate cancer |
| WO2016020856A3 (en) * | 2014-08-05 | 2016-03-31 | MabQuest SA | Immunological reagents binding to pd-1 |
| KR20170069996A (en) * | 2014-08-05 | 2017-06-21 | 맵퀘스트 에스아 | Immunological reagents binding to pd-1 |
| AU2015298356B2 (en) * | 2014-08-05 | 2020-11-19 | MabQuest SA | Immunological reagents binding to pd-1 |
| KR102357893B1 (en) * | 2014-08-05 | 2022-02-04 | 맵퀘스트 에스아 | Immunological reagents binding to pd-1 |
| US10695426B2 (en) | 2014-08-25 | 2020-06-30 | Pfizer Inc. | Combination of a PD-1 antagonist and an ALK inhibitor for treating cancer |
| US11130812B2 (en) | 2014-09-01 | 2021-09-28 | Birdie Biopharmaceuticals, Inc. | Anti PD-L1 conjugates for treating tumors |
| EP4148069A1 (en) | 2014-09-01 | 2023-03-15 | Birdie Biopharmaceuticals Inc. | Anti-pd-l1 conjugates for treating tumors |
| EP3763742A1 (en) | 2014-09-01 | 2021-01-13 | Birdie Biopharmaceuticals Inc. | Anti-pd-l1 conjugates for treating tumors |
| US11344620B2 (en) | 2014-09-13 | 2022-05-31 | Novartis Ag | Combination therapies |
| EP4029508A1 (en) | 2014-10-10 | 2022-07-20 | Idera Pharmaceuticals, Inc. | Treatment of cancer using tlr9 agonists and checkpoint inhibitors |
| WO2016057898A1 (en) | 2014-10-10 | 2016-04-14 | Idera Pharmaceuticals, Inc. | Treatment of cancer using tlr9 agonist with checkpoint inhibitors |
| US10851165B2 (en) | 2014-10-14 | 2020-12-01 | Novartis Ag | Antibody molecules to PD-L1 and methods of treating cancer |
| US9988452B2 (en) | 2014-10-14 | 2018-06-05 | Novartis Ag | Antibody molecules to PD-L1 and uses thereof |
| US10280224B2 (en) | 2014-10-27 | 2019-05-07 | Agency For Science, Technology And Research | Anti-PD-1 antibodies |
| WO2016068801A1 (en) * | 2014-10-27 | 2016-05-06 | Agency For Science, Technology And Research | Anti-pd-1 antibodies |
| US9771425B2 (en) | 2014-10-27 | 2017-09-26 | Agency For Science, Technology And Research | Anti-PD-1 antibodies |
| US11072659B2 (en) | 2014-10-27 | 2021-07-27 | Agency For Science, Technology And Research | Anti-PD-1 antibodies |
| WO2016077397A3 (en) * | 2014-11-11 | 2016-09-01 | Sutro Biopharma, Inc. | Anti-pd-1 antibodies, compositions comprising anti-pd-1 antibodies and methods of using anti-pd-1 antibodies |
| US10822414B2 (en) | 2014-11-11 | 2020-11-03 | Sutro Biopharma, Inc. | Anti-PD-1 antibodies, compositions comprising anti-PD-1 antibodies and methods of using anti-PD-1 antibodies |
| US11718668B2 (en) | 2014-11-13 | 2023-08-08 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11325974B2 (en) | 2014-11-13 | 2022-05-10 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11591393B2 (en) | 2014-11-13 | 2023-02-28 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11325975B2 (en) | 2014-11-13 | 2022-05-10 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11753468B2 (en) | 2014-11-13 | 2023-09-12 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11649287B2 (en) | 2014-11-13 | 2023-05-16 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11339219B2 (en) | 2014-11-13 | 2022-05-24 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11643462B2 (en) | 2014-11-13 | 2023-05-09 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US10934356B2 (en) | 2014-11-13 | 2021-03-02 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11634491B2 (en) | 2014-11-13 | 2023-04-25 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| US11629187B2 (en) | 2014-11-13 | 2023-04-18 | The Johns Hopkins University | Checkpoint blockade and microsatellite instability |
| WO2016089797A1 (en) | 2014-12-05 | 2016-06-09 | Merck Sharp & Dohme Corp. | Novel tricyclic compounds as inhibitors of mutant idh enzymes |
| WO2016089830A1 (en) | 2014-12-05 | 2016-06-09 | Merck Sharp & Dohme Corp. | Novel tricyclic compounds as inhibitors of mutant idh enzymes |
| WO2016089833A1 (en) | 2014-12-05 | 2016-06-09 | Merck Sharp & Dohme Corp. | Novel tricyclic compounds as inhibitors of mutant idh enzymes |
| US10800846B2 (en) | 2015-02-26 | 2020-10-13 | Merck Patent Gmbh | PD-1/PD-L1 inhibitors for the treatment of cancer |
| WO2016140717A1 (en) | 2015-03-04 | 2016-09-09 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and a vegfr/fgfr/ret tyrosine kinase inhibitor for treating cancer |
| US12083112B2 (en) | 2015-03-04 | 2024-09-10 | Eisai R&D Management Co., Ltd. | Combination of a PD-1 antagonist and a VEGFR/FGFR/RET tyrosine kinase inhibitor for treating cancer |
| WO2016141209A1 (en) | 2015-03-04 | 2016-09-09 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and eribulin for treating cancer |
| US10945990B2 (en) | 2015-03-04 | 2021-03-16 | Eisai R&D Management Co., Ltd. | Combination of a PD-1 antagonist and eribulin for treating cancer |
| WO2016141218A1 (en) | 2015-03-04 | 2016-09-09 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and a vegfr/fgfr/ret tyrosine kinase inhibitor for treating cancer |
| US11547705B2 (en) | 2015-03-04 | 2023-01-10 | Merck Sharp & Dohme Llc | Combination of a PD-1 antagonist and a VEGF-R/FGFR/RET tyrosine kinase inhibitor for treating cancer |
| EP4023645A1 (en) | 2015-03-10 | 2022-07-06 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole and thiadiazole compounds as immunomodulators |
| US11465976B2 (en) | 2015-03-10 | 2022-10-11 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole and thiadiazole compounds as immunomodulators |
| US10781189B2 (en) | 2015-03-10 | 2020-09-22 | Aurigene Discovery Technologies Limited | 1,2,4-Oxadiazole and thiadiazole compounds as immunomodulators |
| WO2016142833A1 (en) | 2015-03-10 | 2016-09-15 | Aurigene Discovery Technologies Limited | 1,2,4-oxadiazole and thiadiazole compounds as immunomodulators |
| US10815264B2 (en) | 2015-05-27 | 2020-10-27 | Southern Research Institute | Nucleotides for the treatment of cancer |
| WO2016189055A1 (en) | 2015-05-27 | 2016-12-01 | Idenix Pharmaceuticals Llc | Nucleotides for the treatment of cancer |
| US10751412B2 (en) | 2015-05-29 | 2020-08-25 | Merck Sharp & Dohme Corp. | Combination of a PD-1 antagonist and CPG-C type oligonucleotide for treating cancer |
| US11918648B2 (en) | 2015-05-29 | 2024-03-05 | Merck Sharp & Dohme Llc | Combination of a PD-1 antagonist and CpG-C type oligonucleotide for treating cancer |
| WO2016196173A1 (en) | 2015-05-29 | 2016-12-08 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and cpg-c type oligonucleotide for treating cancer |
| EP3892284A1 (en) | 2015-05-29 | 2021-10-13 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and cpg-c type oligonucleotide for treating cancer |
| WO2016196218A1 (en) | 2015-05-31 | 2016-12-08 | Curegenix Corporation | Combination compositions for immunotherapy |
| US11072653B2 (en) | 2015-06-08 | 2021-07-27 | Macrogenics, Inc. | LAG-3-binding molecules and methods of use thereof |
| US11858991B2 (en) | 2015-06-08 | 2024-01-02 | Macrogenics, Inc. | LAG-3-binding molecules and methods of use thereof |
| US11078279B2 (en) | 2015-06-12 | 2021-08-03 | Macrogenics, Inc. | Combination therapy for the treatment of cancer |
| US10869924B2 (en) | 2015-06-16 | 2020-12-22 | Merck Patent Gmbh | PD-L1 antagonist combination treatments |
| CN114133448A (en) * | 2015-06-23 | 2022-03-04 | 纪念斯隆-凯特琳癌症中心 | Novel PD-1 immunomodulators |
| WO2017007700A1 (en) | 2015-07-06 | 2017-01-12 | Iomet Pharma Ltd. | Pharmaceutical compound |
| EP3744340A2 (en) | 2015-07-16 | 2020-12-02 | Biokine Therapeutics Ltd. | Compositions and methods for treating cancer |
| WO2017009842A2 (en) | 2015-07-16 | 2017-01-19 | Biokine Therapeutics Ltd. | Compositions and methods for treating cancer |
| EP3943098A2 (en) | 2015-07-16 | 2022-01-26 | Biokine Therapeutics Ltd. | Compositions and methods for treating cancer |
| US10973915B2 (en) | 2015-07-28 | 2021-04-13 | Shanghai Yunyi Healthcare And Technology Co., Ltd. | Anti-PD-1 antibodies and uses thereof |
| CN107922494A (en) * | 2015-07-28 | 2018-04-17 | 钜川生物医药 | Anti- 1 antibody of PD and its application |
| WO2017016497A1 (en) * | 2015-07-28 | 2017-02-02 | Harbour Biomed Limited | Anti-pd-1 antibodies and uses thereof |
| CN107922494B (en) * | 2015-07-28 | 2021-05-07 | 上海昀怡健康科技发展有限公司 | anti-PD-1 antibodies and uses thereof |
| US10577422B2 (en) | 2015-07-30 | 2020-03-03 | Macrogenics, Inc. | PD-1-binding molecules and methods of use thereof |
| EP3456346A1 (en) | 2015-07-30 | 2019-03-20 | MacroGenics, Inc. | Pd-1 and lag-3 binding molecules and methods of use thereof |
| EP3981792A1 (en) | 2015-07-30 | 2022-04-13 | MacroGenics, Inc. | Pd-1-binding molecules and methods of use thereof |
| US11623959B2 (en) | 2015-07-30 | 2023-04-11 | Macrogenics, Inc. | PD-1-binding molecules and methods of use thereof |
| US10316089B2 (en) | 2015-08-10 | 2019-06-11 | Innovent Biologics (Suzhou) Co. Ltd. | PD-1 antibodies |
| AU2016307002B2 (en) * | 2015-08-10 | 2019-07-11 | Innovent Biologics (Suzhou) Co., Ltd. | PD-1 antibodies |
| RU2686612C1 (en) * | 2015-08-10 | 2019-04-29 | Инновент Биолоджикс (Сучжоу) Ко., Лтд. | Pd-1 antibodies |
| WO2017025016A1 (en) * | 2015-08-10 | 2017-02-16 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibodies |
| TWI674275B (en) * | 2015-08-10 | 2019-10-11 | 中國大陸商信達生物製藥(蘇州)有限公司 | Pd-1 antibodies |
| IL257062B (en) * | 2015-08-11 | 2022-07-01 | Wuxi Biologics Cayman Inc | Novel anti-pd-1 antibodies |
| EP3334763A4 (en) * | 2015-08-11 | 2019-04-03 | Wuxi Biologics (Cayman) Inc. | Novel anti-pd-1 antibodies |
| US11008391B2 (en) | 2015-08-11 | 2021-05-18 | WuXi Biologics Ireland Limited | Anti-PD-1 antibodies |
| RU2729830C2 (en) * | 2015-08-11 | 2020-08-12 | Уси Байолоджикс (Кайман) Инк. | Novel anti-pd-1 antibodies |
| US11643465B2 (en) | 2015-08-11 | 2023-05-09 | WuXi Biologics Ireland Limited | Anti-PD-1 antibodies |
| AU2016305697B2 (en) * | 2015-08-11 | 2023-02-02 | WuXi Biologics Ireland Limited | Novel anti-PD-1 antibodies |
| WO2017027646A1 (en) | 2015-08-13 | 2017-02-16 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| US10759825B2 (en) | 2015-08-13 | 2020-09-01 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as STING agonists |
| US11453697B1 (en) | 2015-08-13 | 2022-09-27 | Merck Sharp & Dohme Llc | Cyclic di-nucleotide compounds as sting agonists |
| US10738074B2 (en) | 2015-08-13 | 2020-08-11 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as STING agonists |
| US10766919B2 (en) | 2015-08-13 | 2020-09-08 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| WO2017027645A1 (en) | 2015-08-13 | 2017-02-16 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| US10106574B2 (en) | 2015-08-13 | 2018-10-23 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| US11014983B2 (en) | 2015-08-20 | 2021-05-25 | Sutro Biopharma, Inc. | Anti-Tim-3 antibodies, compositions comprising anti-Tim-3 antibodies and methods of making and using anti-Tim-3 antibodies |
| WO2017055484A1 (en) | 2015-09-29 | 2017-04-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for determining the metabolic status of lymphomas |
| US11130810B2 (en) | 2015-10-02 | 2021-09-28 | Hoffmann-La Roche Inc. | Bispecific antibodies specific for PD1 and TIM3 |
| WO2017055404A1 (en) | 2015-10-02 | 2017-04-06 | F. Hoffmann-La Roche Ag | Bispecific antibodies specific for pd1 and tim3 |
| US10287352B2 (en) | 2015-10-02 | 2019-05-14 | Hoffman-La Roche Inc. | Bispecific antibodies specific for PD1 and TIM3 |
| US11174315B2 (en) | 2015-10-08 | 2021-11-16 | Macrogenics, Inc. | Combination therapy for the treatment of cancer |
| JP2018536632A (en) * | 2015-10-08 | 2018-12-13 | マクロジェニクス,インコーポレーテッド | Combination therapy for the treatment of cancer |
| WO2017071625A1 (en) * | 2015-10-30 | 2017-05-04 | 中山康方生物医药有限公司 | Anti-pd-1 monoclonal antibody, and pharmaceutical composition and use thereof |
| US10894830B2 (en) | 2015-11-03 | 2021-01-19 | Janssen Biotech, Inc. | Antibodies specifically binding PD-1, TIM-3 or PD-1 and TIM-3 and their uses |
| WO2017079115A1 (en) | 2015-11-03 | 2017-05-11 | Janssen Biotech, Inc. | Antibodies specifically binding tim-3 and their uses |
| WO2017079112A1 (en) | 2015-11-03 | 2017-05-11 | Janssen Biotech, Inc. | Antibodies specifically binding pd-1 and their uses |
| WO2017079116A2 (en) | 2015-11-03 | 2017-05-11 | Janssen Biotech, Inc. | Antibodies specifically binding pd-1 and tim-3 and their uses |
| EP4046655A1 (en) | 2015-11-03 | 2022-08-24 | Janssen Biotech, Inc. | Antibodies specifically binding pd-1 and their uses |
| WO2017093933A1 (en) | 2015-12-03 | 2017-06-08 | Glaxosmithkline Intellectual Property Development Limited | Cyclic purine dinucleotides as modulators of sting |
| EP3366691A1 (en) | 2015-12-03 | 2018-08-29 | GlaxoSmithKline Intellectual Property Development Limited | Cyclic purine dinucleotides as modulators of sting |
| WO2017098421A1 (en) | 2015-12-08 | 2017-06-15 | Glaxosmithkline Intellectual Property Development Limited | Benzothiadiazine compounds |
| WO2017106061A1 (en) | 2015-12-14 | 2017-06-22 | Macrogenics, Inc. | Bispecific molecules having immunoreactivity with pd-1 and ctla-4, and methods of use thereof |
| US10954301B2 (en) | 2015-12-14 | 2021-03-23 | Macrogenics, Inc. | Bispecific molecules having immunoreactivity with PD-1 and CTLA-4, and methods of use thereof |
| US11840571B2 (en) | 2015-12-14 | 2023-12-12 | Macrogenics, Inc. | Methods of using bispecific molecules having immunoreactivity with PD-1 and CTLA-4 |
| WO2017106062A1 (en) | 2015-12-15 | 2017-06-22 | Merck Sharp & Dohme Corp. | Novel compounds as indoleamine 2,3-dioxygenase inhibitors |
| US12054557B2 (en) | 2015-12-22 | 2024-08-06 | Regeneron Pharmaceuticals, Inc. | Combination of anti-PD-1 antibodies and bispecific anti-CD20/anti-CD3 antibodies to treat cancer |
| CN108473580A (en) * | 2015-12-23 | 2018-08-31 | 杭州尚健生物技术有限公司 | Anti-human 1 antibody of programmed death receptor and its preparation method and application |
| US10774144B2 (en) | 2015-12-23 | 2020-09-15 | Hangzhou Sumgen Biotechnology Co., Ltd | Human programmed cell death 1 receptor antibody, method of preparing same, and use thereof |
| WO2017118634A1 (en) | 2016-01-04 | 2017-07-13 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of pd-1 and tim-3 as a measure for cd8+ cells in predicting and treating renal cell carcinoma |
| US11136397B2 (en) | 2016-01-07 | 2021-10-05 | Birdie Pharmaceuticals, Inc. | Anti-EGFR combinations for treating tumors |
| US11702476B2 (en) | 2016-01-07 | 2023-07-18 | Birdie Biopharmaceuticals, Inc. | Anti-EGFR combinations for treating tumors |
| US11046781B2 (en) | 2016-01-07 | 2021-06-29 | Birdie Biopharmaceuticals, Inc. | Anti-HER2 combinations for treating tumors |
| US11220552B2 (en) | 2016-01-07 | 2022-01-11 | Birdie Biopharmaceuticals, Inc. | Anti-CD20 combinations for treating tumors |
| WO2017155981A1 (en) | 2016-03-07 | 2017-09-14 | Massachusetts Institute Of Technology | Protein-chaperoned t-cell vaccines |
| WO2017153952A1 (en) | 2016-03-10 | 2017-09-14 | Glaxosmithkline Intellectual Property Development Limited | 5-sulfamoyl-2-hydroxybenzamide derivatives |
| WO2017160599A1 (en) | 2016-03-14 | 2017-09-21 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Use of cd300b antagonists to treat sepsis and septic shock |
| WO2017160754A1 (en) | 2016-03-15 | 2017-09-21 | Mersana Therapeutics,Inc. | Napi2b-targeted antibody-drug conjugates and methods of use thereof |
| EP4302782A2 (en) | 2016-03-15 | 2024-01-10 | Mersana Therapeutics, Inc. | Napi2b-targeted antibody-drug conjugates and methods of use thereof |
| WO2017160975A1 (en) * | 2016-03-16 | 2017-09-21 | Bristol-Myers Squibb Company | Methods of diagnosing and treating lupus |
| US11104734B2 (en) | 2016-04-01 | 2021-08-31 | Akeso Biopharma, Inc. | Anti-PD-1 monoclonal antibody |
| EP3441086A4 (en) * | 2016-04-01 | 2019-11-13 | Akeso Biopharma, Inc. | Anti-pd-1 monoclonal antibody |
| JP2019519247A (en) * | 2016-04-01 | 2019-07-11 | アケソ・バイオファーマ・インコーポレイテッド | Anti-PD-1 monoclonal antibody |
| JP2021058204A (en) * | 2016-04-01 | 2021-04-15 | アケソ・バイオファーマ・インコーポレイテッド | Anti-PD-1 monoclonal antibody |
| JP7121792B2 (en) | 2016-04-01 | 2022-08-18 | アケソ・バイオファーマ・インコーポレイテッド | Anti-PD-1 monoclonal antibody |
| EP4032885A1 (en) | 2016-04-07 | 2022-07-27 | GlaxoSmithKline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| WO2017175156A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| WO2017175147A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| US12029724B2 (en) | 2016-04-28 | 2024-07-09 | Eisai R&D Management Co., Ltd. | Method for inhibiting tumor growth |
| WO2017191545A1 (en) | 2016-05-05 | 2017-11-09 | Glaxosmithkline Intellectual Property (No.2) Limited | Enhancer of zeste homolog 2 inhibitors |
| EP3243832A1 (en) | 2016-05-13 | 2017-11-15 | F. Hoffmann-La Roche AG | Antigen binding molecules comprising a tnf family ligand trimer and pd1 binding moiety |
| US11505600B2 (en) | 2016-05-13 | 2022-11-22 | Regeneron Pharmaceuticals, Inc. | Methods of treating skin cancer by administering a PD-1 inhibitor |
| US10457725B2 (en) | 2016-05-13 | 2019-10-29 | Regeneron Pharmaceuticals, Inc. | Methods of treating skin cancer by administering a PD-1 inhibitor |
| US11795219B2 (en) | 2016-05-18 | 2023-10-24 | Boehringer Ingelheim International Gmbh | Antibody molecules for cancer treatment |
| WO2017212423A1 (en) | 2016-06-08 | 2017-12-14 | Glaxosmithkline Intellectual Property Development Limited | Chemcical compounds |
| WO2017212425A1 (en) | 2016-06-08 | 2017-12-14 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds as atf4 pathway inhibitors |
| WO2017220990A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Anti-pd-l1 antibodies |
| WO2017220988A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Multispecific antibodies for immuno-oncology |
| WO2017220989A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Anti-pd-l1 and il-2 cytokines |
| WO2018015879A1 (en) | 2016-07-20 | 2018-01-25 | Glaxosmithkline Intellectual Property Development Limited | Isoquinoline derivatives as perk inhibitors |
| WO2018027039A1 (en) | 2016-08-03 | 2018-02-08 | Nextcure, Inc. | Compositions and methods for modulating lair signal transduction |
| US12084495B2 (en) | 2016-08-03 | 2024-09-10 | Nextcure, Inc. | Compositions and methods for modulating LAIR signal transduction |
| US11130809B2 (en) | 2016-08-09 | 2021-09-28 | Innovent Biologics (Suzhou) Co., Ltd. | PD-1 antibody formulation |
| WO2018028383A1 (en) | 2016-08-09 | 2018-02-15 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibody formulation |
| US9914783B1 (en) | 2016-09-14 | 2018-03-13 | Abbvie Biotherapeutics Inc. | Anti-PD-1 antibodies and their uses |
| US10730953B2 (en) | 2016-09-14 | 2020-08-04 | Abbvie Biotherapeutics Inc. | Anti-PD-1 antibodies and their uses |
| US11090391B2 (en) | 2016-09-16 | 2021-08-17 | The Johns Hopkins University | Protein nanocages with enhanced mucus penetration for targeted tissue and intracellular delivery |
| WO2018053434A1 (en) | 2016-09-16 | 2018-03-22 | The Johns Hopkins University | Protein nanocages with enhanced mucus penetration for targeted tissue and intracellular delivery |
| US10703738B2 (en) | 2016-10-04 | 2020-07-07 | Merck Sharp & Dohme Corp. | Benzo[b]thiophene compounds as STING agonists |
| WO2018067423A1 (en) | 2016-10-04 | 2018-04-12 | Merck Sharp & Dohme Corp. | BENZO[b]THIOPHENE COMPOUNDS AS STING AGONISTS |
| US10730849B2 (en) | 2016-10-04 | 2020-08-04 | Merck Sharp & Dohme Corp. | Benzo[b]thiophene compounds as STING agonists |
| US10414747B2 (en) | 2016-10-04 | 2019-09-17 | Merck Sharp & Dohme Corp. | Benzo[b]thiophene compounds as sting agonists |
| US11274154B2 (en) | 2016-10-06 | 2022-03-15 | Pfizer Inc. | Dosing regimen of avelumab for the treatment of cancer |
| WO2018071792A1 (en) | 2016-10-14 | 2018-04-19 | Merck Sharp & Dohme Corp. | Combination of a pd-1 antagonist and eribulin for treating urothelial cancer |
| US11155624B2 (en) | 2016-11-01 | 2021-10-26 | Anaptysbio, Inc. | Antibodies directed against programmed death-1 (PD-1) |
| US11730748B2 (en) | 2016-11-21 | 2023-08-22 | Msd International Gmbh | Cyclic phosphate substituted nucleoside derivatives for the treatment of liver diseases |
| WO2018091542A1 (en) | 2016-11-21 | 2018-05-24 | Idenix Pharmaceuticals Llc | Cyclic phosphate substituted nucleoside derivatives for the treatment of liver diseases |
| WO2018098269A2 (en) | 2016-11-23 | 2018-05-31 | Mersana Therapeutics, Inc. | Peptide-containing linkers for antibody-drug conjugates |
| WO2018100534A1 (en) | 2016-12-01 | 2018-06-07 | Glaxosmithkline Intellectual Property Development Limited | Combination therapy |
| WO2018100535A1 (en) | 2016-12-01 | 2018-06-07 | Glaxosmithkline Intellectual Property Development Limited | Combination therapy |
| US11407830B2 (en) | 2017-01-09 | 2022-08-09 | Tesaro, Inc. | Methods of treating cancer with anti-PD-1 antibodies |
| US11292842B2 (en) | 2017-02-21 | 2022-04-05 | Regeneron Pharmaceuticals, Inc. | Anti-PD-1 antibodies for treatment of lung cancer |
| US11926668B2 (en) | 2017-02-21 | 2024-03-12 | Regeneron Pharmaceuticals Inc. | Anti-PD-1 antibodies for treatment of lung cancer |
| WO2018156785A1 (en) * | 2017-02-22 | 2018-08-30 | Sutro Biopharma, Inc. | Pd-1/tim-3 bi-specific antibodies, compositions thereof, and methods of making and using the same |
| US11942149B2 (en) | 2017-02-24 | 2024-03-26 | Macrogenics, Inc. | Bispecific binding molecules that are capable of binding CD137 and tumor antigens, and uses thereof |
| EP4389226A2 (en) | 2017-02-24 | 2024-06-26 | MacroGenics, Inc. | Bispecific binding molecules that are capable of binding cd137 and tumor antigens, and uses thereof |
| US11459394B2 (en) | 2017-02-24 | 2022-10-04 | Macrogenics, Inc. | Bispecific binding molecules that are capable of binding CD137 and tumor antigens, and uses thereof |
| WO2018154520A1 (en) | 2017-02-27 | 2018-08-30 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides as kinase inhibitors |
| WO2018160538A1 (en) | 2017-02-28 | 2018-09-07 | Mersana Therapeutics, Inc. | Combination therapies of her2-targeted antibody-drug conjugates |
| US11161905B2 (en) | 2017-03-04 | 2021-11-02 | Xiangtan Tenghua Bioscience | Recombinant antibodies to programmed death 1 (PD-1) and uses thereof |
| WO2018162944A1 (en) * | 2017-03-04 | 2018-09-13 | Shenzhen Runshin Bioscience | Recombinant antibodies to programmed death 1 (pd-1) and uses therefor |
| WO2018167778A1 (en) | 2017-03-12 | 2018-09-20 | Yeda Research And Development Co. Ltd. | Methods of diagnosing and prognosing cancer |
| WO2018167780A1 (en) | 2017-03-12 | 2018-09-20 | Yeda Research And Development Co. Ltd. | Methods of prognosing and treating cancer |
| US10961310B2 (en) | 2017-03-15 | 2021-03-30 | Pandion Operations, Inc. | Targeted immunotolerance |
| WO2018172508A1 (en) | 2017-03-24 | 2018-09-27 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| US11413331B2 (en) | 2017-04-03 | 2022-08-16 | Hoffmann-La Roche Inc. | Immunoconjugates |
| US12023368B2 (en) | 2017-04-03 | 2024-07-02 | Hoffmann-La Roche Inc. | Immunoconjugates |
| WO2018185043A1 (en) | 2017-04-05 | 2018-10-11 | F. Hoffmann-La Roche Ag | Bispecific antibodies specifically binding to pd1 and lag3 |
| US11285207B2 (en) | 2017-04-05 | 2022-03-29 | Hoffmann-La Roche Inc. | Bispecific antibodies specifically binding to PD1 and LAG3 |
| US11603407B2 (en) | 2017-04-06 | 2023-03-14 | Regeneron Pharmaceuticals, Inc. | Stable antibody formulation |
| US11326170B2 (en) | 2017-04-18 | 2022-05-10 | Changchun Huapu Biotechnology Co. Ltd. | Immunomodulatory polynucleotides and uses thereof |
| EP3613768A4 (en) * | 2017-04-20 | 2020-06-17 | Suzhou Stainwei Biotech Inc. | Monoclonal antibody antagonizing and inhibiting binding between human pd-1 antigen and ligand thereof, preparation method therefor and application thereof |
| EP4286009A2 (en) | 2017-04-21 | 2023-12-06 | Sillajen, Inc. | Oncolytic vaccinia virus and checkpoint inhibitor combination therapy |
| WO2018195552A1 (en) | 2017-04-21 | 2018-10-25 | Sillajen, Inc. | Oncolytic vaccinia virus and checkpoint inhibitor combination therapy |
| US11053240B2 (en) | 2017-04-27 | 2021-07-06 | Birdie Biopharmaceuticals, Inc. | 2-amino-quinoline derivatives |
| US11834448B2 (en) | 2017-04-27 | 2023-12-05 | Birdie Biopharmaceuticals, Inc. | 2-amino-quinoline derivatives |
| US11633476B2 (en) | 2017-05-02 | 2023-04-25 | Merck Sharp & Dohme Llc | Stable formulations of programmed death receptor 1 (PD-1) antibodies and methods of use thereof |
| US11845798B2 (en) | 2017-05-02 | 2023-12-19 | Merck Sharp & Dohme Llc | Formulations of anti-LAG3 antibodies and co-formulations of anti-LAG3 antibodies and anti-PD-1 antibodies |
| US11466047B2 (en) | 2017-05-12 | 2022-10-11 | Merck Sharp & Dohme Llc | Cyclic di-nucleotide compounds as sting agonists |
| US11466068B2 (en) | 2017-05-24 | 2022-10-11 | Pandion Operations, Inc. | Targeted immunotolerance |
| US20200131266A1 (en) * | 2017-05-31 | 2020-04-30 | Stcube & Co., Inc. | Methods of treating cancer using antibodies and molecules that immunospecifically bind to btn1a1 |
| US11559504B2 (en) | 2017-06-02 | 2023-01-24 | The Penn State Research Foundation | Ceramide nanoliposomes, compositions and methods of using for immunotherapy |
| WO2018225093A1 (en) | 2017-06-07 | 2018-12-13 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds as atf4 pathway inhibitors |
| WO2018226336A1 (en) | 2017-06-09 | 2018-12-13 | Providence Health & Services - Oregon | Utilization of cd39 and cd103 for identification of human tumor reactive cells for treatment of cancer |
| WO2018225033A1 (en) | 2017-06-09 | 2018-12-13 | Glaxosmithkline Intellectual Property Development Limited | Combination therapy |
| US11517567B2 (en) | 2017-06-23 | 2022-12-06 | Birdie Biopharmaceuticals, Inc. | Pharmaceutical compositions |
| WO2019008506A1 (en) | 2017-07-03 | 2019-01-10 | Glaxosmithkline Intellectual Property Development Limited | N-(3-(2-(4-chlorophenoxy)acetamido)bicyclo[1.1.1]pentan-1-yl)-2-cyclobutane-1-carboxamide derivatives and related compounds as atf4 inhibitors for treating cancer and other diseases |
| WO2019008507A1 (en) | 2017-07-03 | 2019-01-10 | Glaxosmithkline Intellectual Property Development Limited | 2-(4-chlorophenoxy)-n-((1 -(2-(4-chlorophenoxy)ethynazetidin-3-yl)methyl)acetamide derivatives and related compounds as atf4 inhibitors for treating cancer and other diseases |
| WO2019021208A1 (en) | 2017-07-27 | 2019-01-31 | Glaxosmithkline Intellectual Property Development Limited | Indazole derivatives useful as perk inhibitors |
| US11312772B2 (en) | 2017-08-04 | 2022-04-26 | Merck Sharp & Dohme Corp. | Combinations of PD-1 antagonists and benzo [b] thiophene STING agonists for cancer treatment |
| US11285131B2 (en) | 2017-08-04 | 2022-03-29 | Merck Sharp & Dohme Corp. | Benzo[b]thiophene STING agonists for cancer treatment |
| WO2019049061A1 (en) | 2017-09-07 | 2019-03-14 | Glaxosmithkline Intellectual Property Development Limited | 5-(1 h-benzo[d]imidazo-2-yl)-pyridin-2-amine and 5-(3h-imidazo[4,5-b]pyridin-6-yl)-pyridin-2-amine derivatives as c-myc and p300/cbp histone acetyltransferase inhibitors for treating cancer |
| WO2019053617A1 (en) | 2017-09-12 | 2019-03-21 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| US11643401B2 (en) | 2017-09-29 | 2023-05-09 | Curis, Inc. | Crystal forms of immunomodulators |
| US11040948B2 (en) | 2017-09-29 | 2021-06-22 | Curis, Inc. | Crystal forms of immunomodulators |
| US11939306B2 (en) | 2017-09-29 | 2024-03-26 | Curis, Inc. | Crystal forms of immunomodulators |
| WO2019069270A1 (en) | 2017-10-05 | 2019-04-11 | Glaxosmithkline Intellectual Property Development Limited | Modulators of stimulator of interferon genes (sting) |
| WO2019069269A1 (en) | 2017-10-05 | 2019-04-11 | Glaxosmithkline Intellectual Property Development Limited | Modulators of stimulator of interferon genes (sting) useful in treating hiv |
| US11680051B2 (en) | 2017-10-11 | 2023-06-20 | Aurigene Discovery Technologies Limited | Crystalline forms of 3-substituted 1,2,4-oxadiazole |
| US11136300B2 (en) | 2017-10-11 | 2021-10-05 | Aurigene Discovery Technologies Limited | Crystalline forms of 3-substituted 1,2,4-oxadiazole |
| WO2019089412A1 (en) | 2017-11-01 | 2019-05-09 | Merck Sharp & Dohme Corp. | Novel substituted tetrahydroquinolin compounds as indoleamine 2,3-dioxygenase (ido) inhibitors |
| US11497734B2 (en) | 2017-11-03 | 2022-11-15 | Aurigene Discovery Technologies Limited | Dual inhibitors of TIM-3 and PD-1 pathways |
| US11497735B2 (en) | 2017-11-06 | 2022-11-15 | Aurigene Discovery Technologies Limited | Conjoint therapies for immunomodulation |
| US12064418B2 (en) | 2017-11-06 | 2024-08-20 | Curis, Inc. | Conjoint therapies for immunomodulation |
| WO2019099294A1 (en) | 2017-11-14 | 2019-05-23 | Merck Sharp & Dohme Corp. | Novel substituted biaryl compounds as indoleamine 2,3-dioxygenase (ido) inhibitors |
| WO2019099314A1 (en) | 2017-11-14 | 2019-05-23 | Merck Sharp & Dohme Corp. | Novel substituted biaryl compounds as indoleamine 2,3-dioxygenase (ido) inhibitors |
| WO2019104289A1 (en) | 2017-11-27 | 2019-05-31 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepine antibody conjugates |
| US11638760B2 (en) | 2017-11-27 | 2023-05-02 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepine antibody conjugates |
| US11779632B2 (en) | 2017-12-06 | 2023-10-10 | Pandion Operation, Inc. | IL-2 muteins and uses thereof |
| US11091526B2 (en) | 2017-12-06 | 2021-08-17 | Pandion Operations, Inc. | IL-2 muteins and uses thereof |
| US11945852B2 (en) | 2017-12-06 | 2024-04-02 | Pandion Operations, Inc. | IL-2 muteins and uses thereof |
| US11965008B2 (en) | 2017-12-06 | 2024-04-23 | Pandion Operations, Inc. | IL-2 muteins and uses thereof |
| US11946094B2 (en) | 2017-12-10 | 2024-04-02 | Augusta University Research Institute, Inc. | Combination therapies and methods of use thereof |
| US11685761B2 (en) | 2017-12-20 | 2023-06-27 | Merck Sharp & Dohme Llc | Cyclic di-nucleotide compounds as sting agonists |
| WO2019126691A1 (en) | 2017-12-21 | 2019-06-27 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepine antibody conjugates |
| WO2019134946A1 (en) | 2018-01-04 | 2019-07-11 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma resistant |
| WO2019147670A1 (en) | 2018-01-23 | 2019-08-01 | Nextcure, Inc. | B7-h4 antibodies and methods of use thereof |
| WO2019149716A1 (en) | 2018-01-31 | 2019-08-08 | F. Hoffmann-La Roche Ag | Bispecific antibodies comprising an antigen-binding site binding to lag3 |
| WO2019162325A1 (en) | 2018-02-21 | 2019-08-29 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of sk1 as biomarker for predicting response to immunecheckpoint inhibitors |
| US10493148B2 (en) | 2018-03-02 | 2019-12-03 | Eli Lilly And Company | PD-1 agonist antibodies and uses thereof |
| US10793557B2 (en) | 2018-04-03 | 2020-10-06 | Merck Sharp & Dohme Corp. | Sting agonist compounds |
| US11702430B2 (en) | 2018-04-03 | 2023-07-18 | Merck Sharp & Dohme Llc | Aza-benzothiophene compounds as STING agonists |
| WO2019195124A1 (en) | 2018-04-03 | 2019-10-10 | Merck Sharp & Dohme Corp. | Benzothiophenes and related compounds as sting agonists |
| WO2019193540A1 (en) | 2018-04-06 | 2019-10-10 | Glaxosmithkline Intellectual Property Development Limited | Heteroaryl derivatives of formula (i) as atf4 inhibitors |
| WO2019193541A1 (en) | 2018-04-06 | 2019-10-10 | Glaxosmithkline Intellectual Property Development Limited | Bicyclic aromatic ring derivatives of formula (i) as atf4 inhibitors |
| US11542505B1 (en) | 2018-04-20 | 2023-01-03 | Merck Sharp & Dohme Llc | Substituted RIG-I agonists: compositions and methods thereof |
| WO2019219709A1 (en) | 2018-05-14 | 2019-11-21 | Immunocore Limited | Bifunctional binding polypeptides |
| WO2019219820A1 (en) | 2018-05-16 | 2019-11-21 | Ctxt Pty Limited | Substituted condensed thiophenes as modulators of sting |
| US11613525B2 (en) | 2018-05-16 | 2023-03-28 | Ctxt Pty Limited | Substituted condensed thiophenes as modulators of sting |
| WO2019232319A1 (en) | 2018-05-31 | 2019-12-05 | Peloton Therapeutics, Inc. | Compositions and methods for inhibiting cd73 |
| WO2019231870A1 (en) | 2018-05-31 | 2019-12-05 | Merck Sharp & Dohme Corp. | Novel substituted [1.1.1] bicyclo compounds as indoleamine 2,3-dioxygenase inhibitors |
| WO2019241730A2 (en) | 2018-06-15 | 2019-12-19 | Flagship Pioneering Innovations V, Inc. | Increasing immune activity through modulation of postcellular signaling factors |
| EP3818146A4 (en) * | 2018-07-04 | 2022-10-19 | Cytoimmune Therapeutics, Inc. | Compositions and methods for immunotherapy targeting flt3, pd-1, and/or pd-l1 |
| WO2020012339A1 (en) | 2018-07-09 | 2020-01-16 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| WO2020011972A1 (en) | 2018-07-12 | 2020-01-16 | F-Star Beta Limited | Fc binding fragments comprising cd137 antigne binding side |
| WO2020023268A1 (en) | 2018-07-24 | 2020-01-30 | Amgen Inc. | Combination of lilrb1/2 pathway inhibitors and pd-1 pathway inhibitors |
| WO2020031107A1 (en) | 2018-08-08 | 2020-02-13 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| WO2020044206A1 (en) | 2018-08-29 | 2020-03-05 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides as kinase inhibitors for use in the treatment cancer |
| WO2020047345A1 (en) | 2018-08-31 | 2020-03-05 | Yale University | Compositions and methods of using cell-penetrating antibodies in combination with immune checkpoint modulators |
| WO2020055702A1 (en) | 2018-09-13 | 2020-03-19 | Merck Sharp & Dohme Corp. | Combination of pd-1 antagonist and lag3 antagonist for treating non-microsatellite instablity-high/proficient mismatch repair colorectal cancer |
| WO2020079692A1 (en) | 2018-10-17 | 2020-04-23 | Biolinerx Ltd. | Treatment of metastatic pancreatic adenocarcinoma |
| WO2020086476A1 (en) | 2018-10-22 | 2020-04-30 | Glaxosmithkline Intellectual Property Development Limited | Dosing |
| WO2020086479A1 (en) | 2018-10-22 | 2020-04-30 | Glaxosmithkline Intellectual Property Development Limited | Dosing |
| WO2020092385A1 (en) | 2018-10-29 | 2020-05-07 | Mersana Therapeutics, Inc. | Cysteine engineered antibody-drug conjugates with peptide-containing linkers |
| WO2020092183A1 (en) | 2018-11-01 | 2020-05-07 | Merck Sharp & Dohme Corp. | Novel substituted pyrazole compounds as indoleamine 2,3-dioxygenase inhibitors |
| WO2020096871A1 (en) | 2018-11-06 | 2020-05-14 | Merck Sharp & Dohme Corp. | Novel substituted tricyclic compounds as indoleamine 2,3-dioxygenase inhibitors |
| WO2020102804A2 (en) | 2018-11-16 | 2020-05-22 | Arqule, Inc. | Pharmaceutical combination for treatment of cancer |
| WO2020104479A1 (en) | 2018-11-20 | 2020-05-28 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating cancers and resistant cancers with anti transferrin receptor 1 antibodies |
| WO2020104496A1 (en) | 2018-11-20 | 2020-05-28 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Bispecific antibody targeting transferrin receptor 1 and soluble antigen |
| WO2020106560A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020106558A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020112581A1 (en) | 2018-11-28 | 2020-06-04 | Merck Sharp & Dohme Corp. | Novel substituted piperazine amide compounds as indoleamine 2, 3-dioxygenase (ido) inhibitors |
| EP4342473A2 (en) | 2018-11-30 | 2024-03-27 | GlaxoSmithKline Intellectual Property Development Limited | Compounds useful in hiv therapy |
| WO2020112700A1 (en) | 2018-11-30 | 2020-06-04 | Merck Sharp & Dohme Corp. | 9-substituted amino triazolo quinazoline derivatives as adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020110056A1 (en) | 2018-11-30 | 2020-06-04 | Glaxosmithkline Intellectual Property Development Limited | Compounds useful in hiv therapy |
| WO2020115261A1 (en) | 2018-12-07 | 2020-06-11 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2020123453A2 (en) | 2018-12-11 | 2020-06-18 | Theravance Biopharma R&D Ip, Llc | Alk5 inhibitors |
| WO2020120592A1 (en) | 2018-12-12 | 2020-06-18 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for predicting and treating melanoma |
| WO2020128447A1 (en) | 2018-12-17 | 2020-06-25 | Oxford University Innovation Limited | Modified antibodies |
| GB201820547D0 (en) | 2018-12-17 | 2019-01-30 | Oxford Univ Innovation | Modified antibodies |
| WO2020127411A1 (en) | 2018-12-19 | 2020-06-25 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating cancers by immuno-modulation using antibodies against cathespin-d |
| WO2020132560A2 (en) | 2018-12-21 | 2020-06-25 | Aim Immunotech Inc. | Compositions and methods for cancer therapy |
| WO2020127885A1 (en) | 2018-12-21 | 2020-06-25 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Compositions for treating cancers and resistant cancers |
| WO2020157131A1 (en) | 2019-01-30 | 2020-08-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for identifying whether a subject suffering from a cancer will achieve a response with an immune-checkpoint inhibitor |
| WO2020161083A1 (en) | 2019-02-04 | 2020-08-13 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for modulating blood-brain barrier |
| WO2020165370A1 (en) | 2019-02-13 | 2020-08-20 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for selecting a cancer treatment in a subject suffering from cancer |
| WO2020183011A1 (en) | 2019-03-14 | 2020-09-17 | Institut Curie | Htr1d inhibitors and uses thereof in the treatment of cancer |
| WO2020201095A1 (en) | 2019-03-29 | 2020-10-08 | Institut Curie | Interleukin-2 variants with modified biological activity |
| WO2020205688A1 (en) | 2019-04-04 | 2020-10-08 | Merck Sharp & Dohme Corp. | Inhibitors of histone deacetylase-3 useful for the treatment of cancer, inflammation, neurodegeneration diseases and diabetes |
| WO2020221796A1 (en) | 2019-04-30 | 2020-11-05 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2020227159A2 (en) | 2019-05-03 | 2020-11-12 | Flagship Pioneering Innovations V, Inc. | Methods of modulating immune activity |
| WO2020232375A1 (en) | 2019-05-16 | 2020-11-19 | Silicon Swat, Inc. | Oxoacridinyl acetic acid derivatives and methods of use |
| WO2020232378A1 (en) | 2019-05-16 | 2020-11-19 | Silicon Swat, Inc. | Benzo[b][1,8]naphthyridine acetic acid derivatives and methods of use |
| US11739146B2 (en) | 2019-05-20 | 2023-08-29 | Pandion Operations, Inc. | MAdCAM targeted immunotolerance |
| WO2020239558A1 (en) | 2019-05-24 | 2020-12-03 | Pfizer Inc. | Combination therapies using cdk inhibitors |
| WO2020247648A3 (en) * | 2019-06-05 | 2021-01-14 | Anaptysbio, Inc. | Pd-1 agonist and method of using same |
| WO2020260547A1 (en) | 2019-06-27 | 2020-12-30 | Rigontec Gmbh | Design method for optimized rig-i ligands |
| WO2021006199A1 (en) | 2019-07-05 | 2021-01-14 | 小野薬品工業株式会社 | Treatment of hematologic cancer with pd-1/cd3 dual specificity protein |
| WO2021009365A1 (en) | 2019-07-18 | 2021-01-21 | Ctxt Pty Limited | Benzothiophene, thienopyridine and thienopyrimidine derivatives for the modulation of sting |
| WO2021009362A1 (en) | 2019-07-18 | 2021-01-21 | Ctxt Pty Limited | Benzothiophene, thienopyridine and thienopyrimidine derivatives for the modulation of sting |
| US11083705B2 (en) | 2019-07-26 | 2021-08-10 | Eisai R&D Management Co., Ltd. | Pharmaceutical composition for treating tumor |
| US12036204B2 (en) | 2019-07-26 | 2024-07-16 | Eisai R&D Management Co., Ltd. | Pharmaceutical composition for treating tumor |
| WO2021020336A1 (en) | 2019-07-26 | 2021-02-04 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Pharmaceutical composition for treating tumor |
| US11939343B2 (en) | 2019-08-02 | 2024-03-26 | Mersana Therapeutics, Inc. | Sting agonist compounds and methods of use |
| WO2021025140A1 (en) | 2019-08-08 | 2021-02-11 | 小野薬品工業株式会社 | Dual-specific protein |
| WO2021048292A1 (en) | 2019-09-11 | 2021-03-18 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2021058711A2 (en) | 2019-09-27 | 2021-04-01 | Glaxosmithkline Intellectual Property Development Limited | Antigen binding proteins |
| WO2021064180A1 (en) | 2019-10-03 | 2021-04-08 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for modulating macrophages polarization |
| WO2021074391A1 (en) | 2019-10-17 | 2021-04-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for diagnosing nasal intestinal type adenocarcinomas |
| WO2021078910A1 (en) | 2019-10-22 | 2021-04-29 | Institut Curie | Immunotherapy targeting tumor neoantigenic peptides |
| WO2021083060A1 (en) | 2019-10-28 | 2021-05-06 | 中国科学院上海药物研究所 | Five-membered heterocyclic oxocarboxylic acid compound and medical use thereof |
| WO2021083959A1 (en) | 2019-10-29 | 2021-05-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating uveal melanoma |
| WO2021086909A1 (en) | 2019-10-29 | 2021-05-06 | Eisai R&D Managment Co., Ltd. | Combination of a pd-1 antagonist, a vegfr/fgfr/ret tyrosine kinase inhibitor and a cbp/beta-catenin inhibitor for treating cancer |
| WO2021099511A1 (en) | 2019-11-22 | 2021-05-27 | Institut Curie | Device, apparatus and method for minibeam radiation therapy |
| WO2021102468A1 (en) | 2019-11-22 | 2021-05-27 | Theravance Biopharma R&D Ip, Llc | Substituted 1,5-naphthyridines or quinolines as alk5 inhibitors |
| WO2021108025A1 (en) | 2019-11-26 | 2021-06-03 | Massachusetts Institute Of Technology | Cell-based cancer vaccines and cancer therapies |
| WO2021113679A1 (en) | 2019-12-06 | 2021-06-10 | Mersana Therapeutics, Inc. | Dimeric compounds as sting agonists |
| WO2021127217A1 (en) | 2019-12-17 | 2021-06-24 | Flagship Pioneering Innovations V, Inc. | Combination anti-cancer therapies with inducers of iron-dependent cellular disassembly |
| WO2021123243A1 (en) | 2019-12-19 | 2021-06-24 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and vaccine compositions to treat cancers |
| WO2021144426A1 (en) | 2020-01-17 | 2021-07-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2021156360A1 (en) | 2020-02-05 | 2021-08-12 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for discontinuing a treatment with a tyrosine kinase inhibitor (tki) |
| US11981715B2 (en) | 2020-02-21 | 2024-05-14 | Pandion Operations, Inc. | Tissue targeted immunotolerance with a CD39 effector |
| WO2021170777A1 (en) | 2020-02-28 | 2021-09-02 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for diagnosing, prognosing and managing treatment of breast cancer |
| WO2021203131A1 (en) | 2020-03-31 | 2021-10-07 | Theravance Biopharma R&D Ip, Llc | Substituted pyrimidines and methods of use |
| WO2021209356A1 (en) | 2020-04-14 | 2021-10-21 | Glaxosmithkline Intellectual Property Development Limited | Combination treatment for cancer |
| WO2021209357A1 (en) | 2020-04-14 | 2021-10-21 | Glaxosmithkline Intellectual Property Development Limited | Combination treatment for cancer involving anti-icos and anti-pd1 antibodies, optionally further involving anti-tim3 antibodies |
| WO2021222188A1 (en) | 2020-04-27 | 2021-11-04 | Seagen Inc. | Anti-cd40 antibody combination treatment for cancer |
| WO2021224186A1 (en) | 2020-05-04 | 2021-11-11 | Institut Curie | New pyridine derivatives as radiosensitizers |
| WO2021226003A1 (en) | 2020-05-06 | 2021-11-11 | Merck Sharp & Dohme Corp. | Il4i1 inhibitors and methods of use |
| WO2021231350A1 (en) | 2020-05-13 | 2021-11-18 | Massachusetts Institute Of Technology | Compositions of polymeric microdevices and their use in cancer immunotherapy |
| WO2021234110A1 (en) | 2020-05-20 | 2021-11-25 | Institut Curie | Single domain antibodies and their use in cancer therapies |
| WO2021248065A1 (en) | 2020-06-05 | 2021-12-09 | Theraly Fibrosis, Inc. | Trail compositions with reduced immunogenicity |
| WO2021253041A1 (en) | 2020-06-10 | 2021-12-16 | Theravance Biopharma R&D Ip, Llc | Naphthyridine derivatives useful as alk5 inhibitors |
| WO2021260675A1 (en) | 2020-06-24 | 2021-12-30 | Yeda Research And Development Co. Ltd. | Agents for sensitizing solid tumors to treatment |
| WO2022006179A1 (en) | 2020-06-29 | 2022-01-06 | Flagship Pioneering Innovations V, Inc. | Viruses engineered to promote thanotransmission and their use in treating cancer |
| WO2022023379A1 (en) | 2020-07-28 | 2022-02-03 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for preventing and treating a cancer |
| WO2022049526A1 (en) | 2020-09-02 | 2022-03-10 | Pharmabcine Inc. | Combination therapy of a pd-1 antagonist and an antagonist for vegfr-2 for treating patients with cancer |
| US11932692B2 (en) | 2020-09-03 | 2024-03-19 | Regeneron Pharmaceuticals, Inc. | Methods of treating cancer pain by administering a PD-1 inhibitor |
| WO2022079270A1 (en) | 2020-10-16 | 2022-04-21 | Université D'aix-Marseille | Anti-gpc4 single domain antibodies |
| WO2022084325A1 (en) | 2020-10-20 | 2022-04-28 | Institut Curie | Metallic trans-(n-heterocyclic carbene)-amine-platinum complexes and uses thereof for treating cancer |
| WO2022084531A1 (en) | 2020-10-23 | 2022-04-28 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating glioma |
| WO2022092085A1 (en) | 2020-10-28 | 2022-05-05 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Pharmaceutical composition for treating tumors |
| WO2022101484A1 (en) | 2020-11-16 | 2022-05-19 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for predicting and treating uveal melanoma |
| WO2022101481A1 (en) | 2020-11-16 | 2022-05-19 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for predicting and treating uveal melanoma |
| WO2022106505A1 (en) | 2020-11-18 | 2022-05-27 | Institut Curie | Dimers of biguanidines and their therapeutic uses |
| WO2022130206A1 (en) | 2020-12-16 | 2022-06-23 | Pfizer Inc. | TGFβr1 INHIBITOR COMBINATION THERAPIES |
| WO2022152862A1 (en) | 2021-01-14 | 2022-07-21 | Institut Curie | Her2 single domain antibodies variants and cars thereof |
| WO2022165403A1 (en) | 2021-02-01 | 2022-08-04 | Yale University | Chemotherapeutic bioadhesive particles with immunostimulatory molecules for cancer treatment |
| WO2022185160A1 (en) | 2021-03-02 | 2022-09-09 | Glaxosmithkline Intellectual Property Development Limited | Substituted pyridines as dnmt1 inhibitors |
| WO2022189618A1 (en) | 2021-03-12 | 2022-09-15 | Institut Curie | Nitrogen-containing heterocycles as radiosensitizers |
| WO2022194908A1 (en) | 2021-03-17 | 2022-09-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2022212784A1 (en) | 2021-03-31 | 2022-10-06 | Flagship Pioneering Innovations V, Inc. | Thanotransmission polypeptides and their use in treating cancer |
| WO2022208353A1 (en) | 2021-03-31 | 2022-10-06 | Glaxosmithkline Intellectual Property Development Limited | Antigen binding proteins and combinations thereof |
| WO2022217123A2 (en) | 2021-04-08 | 2022-10-13 | Nurix Therapeutics, Inc. | Combination therapies with cbl-b inhibitor compounds |
| WO2022223791A1 (en) | 2021-04-23 | 2022-10-27 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating cell senescence accumulation related disease |
| WO2022232333A1 (en) | 2021-04-30 | 2022-11-03 | Merck Sharp & Dohme Llc | Il4i1 inhibitors and methods of use |
| WO2022238386A1 (en) | 2021-05-10 | 2022-11-17 | Institut Curie | Methods for the treatment of cancer, inflammatory diseases and autoimmune diseases |
| WO2022239820A1 (en) | 2021-05-13 | 2022-11-17 | 公益財団法人神戸医療産業都市推進機構 | Anti-human pd-1 agonist antibody for treating or preventing inflammatory diseases, and pharmaceutical composition containing same |
| WO2022251359A1 (en) | 2021-05-26 | 2022-12-01 | Theravance Biopharma R&D Ip, Llc | Bicyclic inhibitors of alk5 and methods of use |
| WO2022256534A1 (en) | 2021-06-03 | 2022-12-08 | Synthorx, Inc. | Head and neck cancer combination therapy comprising an il-2 conjugate and pembrolizumab |
| WO2022254227A1 (en) | 2021-06-04 | 2022-12-08 | Kymab Limited | Treatment of pd-l1 negative or low expressing cancer with anti-icos antibodies |
| WO2022261018A1 (en) | 2021-06-07 | 2022-12-15 | Providence Health & Services - Oregon | Cxcr5, pd-1, and icos expressing tumor reactive cd4 t cells and their use |
| WO2023278641A1 (en) | 2021-06-29 | 2023-01-05 | Flagship Pioneering Innovations V, Inc. | Immune cells engineered to promote thanotransmission and uses thereof |
| WO2023010080A1 (en) | 2021-07-30 | 2023-02-02 | Seagen Inc. | Treatment for cancer |
| WO2023011879A1 (en) | 2021-08-05 | 2023-02-09 | Institut Curie | Scanning dynamic device for minibeams production |
| WO2023039243A3 (en) * | 2021-09-13 | 2023-09-28 | Achelois Biopharma, Inc. | Hepatitis b virus antivirus (hbv-antivirus) compositions and methods of use |
| WO2023041744A1 (en) | 2021-09-17 | 2023-03-23 | Institut Curie | Bet inhibitors for treating pab1 deficient cancer |
| WO2023057882A1 (en) | 2021-10-05 | 2023-04-13 | Pfizer Inc. | Combinations of azalactam compounds with a pd-1 axis binding antagonist for the treatment of cancer |
| WO2023078900A1 (en) | 2021-11-03 | 2023-05-11 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating triple negative breast cancer (tnbc) |
| WO2023079428A1 (en) | 2021-11-03 | 2023-05-11 | Pfizer Inc. | Combination therapies using tlr7/8 agonist |
| WO2023089032A1 (en) | 2021-11-19 | 2023-05-25 | Institut Curie | Methods for the treatment of hrd cancer and brca-associated cancer |
| WO2023099763A1 (en) | 2021-12-03 | 2023-06-08 | Institut Curie | Sirt6 inhibitors for use in treating resistant hrd cancer |
| WO2023122573A1 (en) | 2021-12-20 | 2023-06-29 | Synthorx, Inc. | Head and neck cancer combination therapy comprising an il-2 conjugate and pembrolizumab |
| WO2023118165A1 (en) | 2021-12-21 | 2023-06-29 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and compositions for treating melanoma |
| WO2023180552A1 (en) | 2022-03-24 | 2023-09-28 | Institut Curie | Immunotherapy targeting tumor transposable element derived neoantigenic peptides in glioblastoma |
| WO2023187024A1 (en) | 2022-03-31 | 2023-10-05 | Institut Curie | Modified rela protein for inducing interferon expression and engineered immune cells with improved interferon expression |
| WO2023194607A1 (en) | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor with cd40 and uses thereof for anti-cancer therapy |
| WO2023194608A1 (en) | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor and uses thereof for anti-cancer therapy |
| WO2023213764A1 (en) | 2022-05-02 | 2023-11-09 | Transgene | Fusion polypeptide comprising an anti-pd-l1 sdab and a member of the tnfsf |
| WO2023213763A1 (en) | 2022-05-02 | 2023-11-09 | Transgene | Poxvirus encoding a binding agent comprising an anti- pd-l1 sdab |
| WO2023230554A1 (en) | 2022-05-25 | 2023-11-30 | Pfizer Inc. | Combination of a braf inhibitor, an egfr inhibitor, and a pd-1 antagonist for the treatment of braf v600e-mutant, msi-h/dmmr colorectal cancer |
| WO2023230541A1 (en) | 2022-05-27 | 2023-11-30 | Viiv Healthcare Company | Piperazine derivatives useful in hiv therapy |
| WO2024003353A1 (en) | 2022-07-01 | 2024-01-04 | Transgene | Fusion protein comprising a surfactant-protein-d and a member of the tnfsf |
| WO2024033400A1 (en) | 2022-08-10 | 2024-02-15 | Institut National de la Santé et de la Recherche Médicale | Sk2 inhibitor for the treatment of pancreatic cancer |
| WO2024033399A1 (en) | 2022-08-10 | 2024-02-15 | Institut National de la Santé et de la Recherche Médicale | Sigmar1 ligand for the treatment of pancreatic cancer |
| WO2024040264A1 (en) | 2022-08-19 | 2024-02-22 | Massachusetts Institute Of Technology | Compositions and methods for targeting dendritic cell lectins |
| WO2024056716A1 (en) | 2022-09-14 | 2024-03-21 | Institut National de la Santé et de la Recherche Médicale | Methods and pharmaceutical compositions for the treatment of dilated cardiomyopathy |
| WO2024068617A1 (en) | 2022-09-26 | 2024-04-04 | Institut Curie | Myeloid cells expressing il-2 and uses thereof for quick anticancer therapy |
| WO2024077191A1 (en) | 2022-10-05 | 2024-04-11 | Flagship Pioneering Innovations V, Inc. | Nucleic acid molecules encoding trif and additionalpolypeptides and their use in treating cancer |
| WO2024081736A2 (en) | 2022-10-11 | 2024-04-18 | Yale University | Compositions and methods of using cell-penetrating antibodies |
| WO2024084034A1 (en) | 2022-10-21 | 2024-04-25 | Institut National de la Santé et de la Recherche Médicale | Methods and pharmaceutical compositions for the treatment of osteoarthritis |
| WO2024112867A1 (en) | 2022-11-23 | 2024-05-30 | University Of Georgia Research Foundation, Inc. | Compositions and methods of use thereof for increasing immune responses |
| WO2024151687A1 (en) | 2023-01-09 | 2024-07-18 | Flagship Pioneering Innovations V, Inc. | Genetic switches and their use in treating cancer |
| WO2024160721A1 (en) | 2023-01-30 | 2024-08-08 | Kymab Limited | Antibodies |
| WO2024163477A1 (en) | 2023-01-31 | 2024-08-08 | University Of Rochester | Immune checkpoint blockade therapy for treating staphylococcus aureus infections |
| WO2024194692A1 (en) | 2023-03-21 | 2024-09-26 | Institut Curie | Tgf-beta inhibitor for use in the treatment of dedifferentiated liposarcoma |
| WO2024194402A1 (en) | 2023-03-21 | 2024-09-26 | Institut Curie | Farnesyl transferase inhibitor for use in methods for the treatment of hrd cancer |
| WO2024194401A1 (en) | 2023-03-21 | 2024-09-26 | Institut Curie | Vps4b inhibitor for use in methods for the treatment of hrd cancer |
| WO2024194673A1 (en) | 2023-03-21 | 2024-09-26 | Institut Curie | Methods for the treatment of dedifferentiated liposarcoma |
| WO2024200571A1 (en) | 2023-03-28 | 2024-10-03 | Institut National de la Santé et de la Recherche Médicale | Method for discriminating mono-immunotherapy from combined immunotherapy in cancers |
| WO2024206357A1 (en) | 2023-03-29 | 2024-10-03 | Merck Sharp & Dohme Llc | Il4i1 inhibitors and methods of use |
Also Published As
| Publication number | Publication date |
|---|---|
| HK1083510A1 (en) | 2006-07-07 |
| ATE514713T1 (en) | 2011-07-15 |
| IL169152A0 (en) | 2007-07-04 |
| MXPA05006828A (en) | 2005-09-08 |
| US20060210567A1 (en) | 2006-09-21 |
| ES2367430T3 (en) | 2011-11-03 |
| US20040213795A1 (en) | 2004-10-28 |
| EP1576014B1 (en) | 2011-06-29 |
| US20080311117A1 (en) | 2008-12-18 |
| AU2010235966A1 (en) | 2010-11-11 |
| NO20053389D0 (en) | 2005-07-12 |
| JP4511943B2 (en) | 2010-07-28 |
| CN1753912B (en) | 2011-11-02 |
| CA2508660C (en) | 2013-08-20 |
| AU2003288675A1 (en) | 2004-07-14 |
| IL169152A (en) | 2010-11-30 |
| US20100028330A1 (en) | 2010-02-04 |
| BR0316880A (en) | 2005-10-25 |
| US7521051B2 (en) | 2009-04-21 |
| JP2010189395A (en) | 2010-09-02 |
| CA2508660A1 (en) | 2004-07-08 |
| CN101899114A (en) | 2010-12-01 |
| US8088905B2 (en) | 2012-01-03 |
| NO336442B1 (en) | 2015-08-17 |
| JP2006521783A (en) | 2006-09-28 |
| NO20053389L (en) | 2005-07-12 |
| US7488802B2 (en) | 2009-02-10 |
| CN1753912A (en) | 2006-03-29 |
| EP1576014A1 (en) | 2005-09-21 |
| AU2003288675A8 (en) | 2004-07-14 |
| AU2003288675B2 (en) | 2010-07-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2003288675B2 (en) | Antibodies against PD-1 and uses therefor | |
| US10077305B2 (en) | Antibodies against PD-1 and uses thereof | |
| JP3653093B2 (en) | Humanized antibody | |
| US9273136B2 (en) | Fully human anti-human NKG2D monoclonal antibodies | |
| JP4320381B2 (en) | Cells expressing anti-CD80 antibody | |
| US11739146B2 (en) | MAdCAM targeted immunotolerance | |
| CA2518371A1 (en) | Antibodies against human il-21 receptor and uses therefor | |
| JP2021508449A (en) | Specific PD-L1 binding sequence inserted into the CH3 domain | |
| US20210347899A1 (en) | Anti-klrg1 antibodies | |
| CA3229113A1 (en) | Anti-b7-h4/anti-4-1bb bispecific antibodies and use thereof | |
| CN112771075A (en) | Prolonged administration of bispecific antibody constructs that bind to CD33 and CD3 | |
| EP1708961B1 (en) | Anti-ip-10 antibodies | |
| KR20120092767A (en) | A human anti-cd23 fab antibody and a pharmaceutical composition for treating tumor comprising the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2508660 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 169152 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004561922 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2005/006828 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003780521 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1334/KOLNP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003288675 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20038A99298 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003780521 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: PI0316880 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10540084 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 10540084 Country of ref document: US |