RU2720509C2 - Комбинированная терапия pac-1 - Google Patents
Комбинированная терапия pac-1 Download PDFInfo
- Publication number
- RU2720509C2 RU2720509C2 RU2017146346A RU2017146346A RU2720509C2 RU 2720509 C2 RU2720509 C2 RU 2720509C2 RU 2017146346 A RU2017146346 A RU 2017146346A RU 2017146346 A RU2017146346 A RU 2017146346A RU 2720509 C2 RU2720509 C2 RU 2720509C2
- Authority
- RU
- Russia
- Prior art keywords
- pac
- cancer
- vemurafenib
- cells
- active substance
- Prior art date
Links
- YQNRVGJCPCNMKT-LFVJCYFKSA-N 2-[(e)-[[2-(4-benzylpiperazin-1-ium-1-yl)acetyl]hydrazinylidene]methyl]-6-prop-2-enylphenolate Chemical compound [O-]C1=C(CC=C)C=CC=C1\C=N\NC(=O)C[NH+]1CCN(CC=2C=CC=CC=2)CC1 YQNRVGJCPCNMKT-LFVJCYFKSA-N 0.000 title claims abstract description 260
- 238000002648 combination therapy Methods 0.000 title description 12
- 101100462537 Caenorhabditis elegans pac-1 gene Proteins 0.000 title description 3
- 101100117764 Mus musculus Dusp2 gene Proteins 0.000 title description 3
- GPXBXXGIAQBQNI-UHFFFAOYSA-N vemurafenib Chemical compound CCCS(=O)(=O)NC1=CC=C(F)C(C(=O)C=2C3=CC(=CN=C3NC=2)C=2C=CC(Cl)=CC=2)=C1F GPXBXXGIAQBQNI-UHFFFAOYSA-N 0.000 claims abstract description 224
- 229960003862 vemurafenib Drugs 0.000 claims abstract description 223
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 200
- 201000011510 cancer Diseases 0.000 claims abstract description 129
- 150000001875 compounds Chemical class 0.000 claims abstract description 75
- 101000984753 Homo sapiens Serine/threonine-protein kinase B-raf Proteins 0.000 claims abstract description 74
- 102100027103 Serine/threonine-protein kinase B-raf Human genes 0.000 claims abstract description 74
- 238000000034 method Methods 0.000 claims abstract description 64
- 230000035772 mutation Effects 0.000 claims abstract description 46
- 230000001939 inductive effect Effects 0.000 claims abstract description 11
- 239000002532 enzyme inhibitor Substances 0.000 claims abstract description 8
- 239000003085 diluting agent Substances 0.000 claims abstract description 6
- 229940125532 enzyme inhibitor Drugs 0.000 claims abstract description 6
- 101000838335 Homo sapiens Dual specificity protein phosphatase 2 Proteins 0.000 claims abstract 14
- 101001080401 Homo sapiens Proteasome assembly chaperone 1 Proteins 0.000 claims abstract 14
- 229960005552 PAC-1 Drugs 0.000 claims abstract 14
- 102100027583 Proteasome assembly chaperone 1 Human genes 0.000 claims abstract 14
- 210000004027 cell Anatomy 0.000 claims description 252
- 239000013543 active substance Substances 0.000 claims description 94
- 238000011282 treatment Methods 0.000 claims description 75
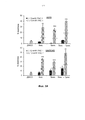
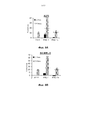
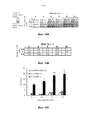
- 230000006907 apoptotic process Effects 0.000 claims description 74
- 201000001441 melanoma Diseases 0.000 claims description 54
- 239000003112 inhibitor Substances 0.000 claims description 33
- 229940124647 MEK inhibitor Drugs 0.000 claims description 28
- 230000012010 growth Effects 0.000 claims description 28
- 229960004066 trametinib Drugs 0.000 claims description 26
- LIRYPHYGHXZJBZ-UHFFFAOYSA-N trametinib Chemical group CC(=O)NC1=CC=CC(N2C(N(C3CC3)C(=O)C3=C(NC=4C(=CC(I)=CC=4)F)N(C)C(=O)C(C)=C32)=O)=C1 LIRYPHYGHXZJBZ-UHFFFAOYSA-N 0.000 claims description 26
- 239000002829 mitogen activated protein kinase inhibitor Substances 0.000 claims description 25
- 238000001727 in vivo Methods 0.000 claims description 17
- -1 PAC-1 compound Chemical class 0.000 claims description 11
- 206010009944 Colon cancer Diseases 0.000 claims description 10
- 208000032839 leukemia Diseases 0.000 claims description 10
- 230000035755 proliferation Effects 0.000 claims description 10
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 9
- 230000002401 inhibitory effect Effects 0.000 claims description 9
- 201000005202 lung cancer Diseases 0.000 claims description 9
- 208000020816 lung neoplasm Diseases 0.000 claims description 9
- 208000026310 Breast neoplasm Diseases 0.000 claims description 8
- 206010025323 Lymphomas Diseases 0.000 claims description 8
- 206010033128 Ovarian cancer Diseases 0.000 claims description 8
- 206010006187 Breast cancer Diseases 0.000 claims description 7
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 7
- 208000001333 Colorectal Neoplasms Diseases 0.000 claims description 6
- 208000024770 Thyroid neoplasm Diseases 0.000 claims description 6
- 201000002510 thyroid cancer Diseases 0.000 claims description 6
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 5
- 208000025127 Erdheim-Chester disease Diseases 0.000 claims description 4
- 201000005099 Langerhans cell histiocytosis Diseases 0.000 claims description 4
- 201000008968 osteosarcoma Diseases 0.000 claims description 4
- 102200055464 rs113488022 Human genes 0.000 claims description 4
- 235000000346 sugar Nutrition 0.000 claims description 4
- 229920000858 Cyclodextrin Polymers 0.000 claims description 3
- 229920002678 cellulose Polymers 0.000 claims description 3
- 239000001913 cellulose Substances 0.000 claims description 2
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 claims description 2
- 239000012928 buffer substance Substances 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 103
- 239000003814 drug Substances 0.000 abstract description 38
- 239000004480 active ingredient Substances 0.000 abstract description 28
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 19
- 238000002560 therapeutic procedure Methods 0.000 abstract description 13
- 230000006882 induction of apoptosis Effects 0.000 abstract description 11
- 239000000126 substance Substances 0.000 abstract description 5
- 239000000945 filler Substances 0.000 abstract description 2
- 230000002887 neurotoxic effect Effects 0.000 abstract description 2
- 230000007761 synergistic anti-cancer Effects 0.000 abstract description 2
- 230000004611 cancer cell death Effects 0.000 abstract 1
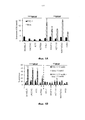
- 108090000397 Caspase 3 Proteins 0.000 description 76
- 102000003952 Caspase 3 Human genes 0.000 description 57
- 239000000203 mixture Substances 0.000 description 37
- 241000699670 Mus sp. Species 0.000 description 31
- 229940079593 drug Drugs 0.000 description 31
- 102100023712 Poly [ADP-ribose] polymerase 1 Human genes 0.000 description 30
- 108010064218 Poly (ADP-Ribose) Polymerase-1 Proteins 0.000 description 29
- 238000003776 cleavage reaction Methods 0.000 description 25
- 230000007017 scission Effects 0.000 description 25
- 230000001640 apoptogenic effect Effects 0.000 description 24
- 230000004913 activation Effects 0.000 description 22
- 239000002552 dosage form Substances 0.000 description 22
- 230000000996 additive effect Effects 0.000 description 21
- 230000001965 increasing effect Effects 0.000 description 21
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 20
- 230000001575 pathological effect Effects 0.000 description 20
- 102100029855 Caspase-3 Human genes 0.000 description 19
- 102000019149 MAP kinase activity proteins Human genes 0.000 description 18
- 108040008097 MAP kinase activity proteins Proteins 0.000 description 18
- 102000047934 Caspase-3/7 Human genes 0.000 description 16
- 108700037887 Caspase-3/7 Proteins 0.000 description 16
- 238000002474 experimental method Methods 0.000 description 16
- 230000026731 phosphorylation Effects 0.000 description 16
- 238000006366 phosphorylation reaction Methods 0.000 description 16
- 230000005764 inhibitory process Effects 0.000 description 15
- 150000003839 salts Chemical class 0.000 description 15
- 230000002195 synergetic effect Effects 0.000 description 15
- 230000007246 mechanism Effects 0.000 description 14
- 201000010099 disease Diseases 0.000 description 13
- 230000001225 therapeutic effect Effects 0.000 description 13
- 102000011727 Caspases Human genes 0.000 description 12
- 108010076667 Caspases Proteins 0.000 description 12
- 230000030833 cell death Effects 0.000 description 12
- 230000000259 anti-tumor effect Effects 0.000 description 11
- 230000003211 malignant effect Effects 0.000 description 11
- 108090000623 proteins and genes Proteins 0.000 description 11
- 210000001519 tissue Anatomy 0.000 description 11
- 108091054455 MAP kinase family Proteins 0.000 description 10
- 102000043136 MAP kinase family Human genes 0.000 description 10
- 238000004458 analytical method Methods 0.000 description 10
- 230000010261 cell growth Effects 0.000 description 10
- 239000007788 liquid Substances 0.000 description 10
- 102000004169 proteins and genes Human genes 0.000 description 10
- 238000001262 western blot Methods 0.000 description 10
- 201000009030 Carcinoma Diseases 0.000 description 9
- 239000003795 chemical substances by application Substances 0.000 description 9
- 230000007423 decrease Effects 0.000 description 9
- 238000011161 development Methods 0.000 description 9
- 238000002360 preparation method Methods 0.000 description 9
- 210000004881 tumor cell Anatomy 0.000 description 9
- 208000003174 Brain Neoplasms Diseases 0.000 description 8
- BFSMGDJOXZAERB-UHFFFAOYSA-N dabrafenib Chemical compound S1C(C(C)(C)C)=NC(C=2C(=C(NS(=O)(=O)C=3C(=CC=CC=3F)F)C=CC=2)F)=C1C1=CC=NC(N)=N1 BFSMGDJOXZAERB-UHFFFAOYSA-N 0.000 description 8
- 230000006698 induction Effects 0.000 description 8
- 239000004615 ingredient Substances 0.000 description 8
- 230000009826 neoplastic cell growth Effects 0.000 description 8
- 231100000419 toxicity Toxicity 0.000 description 8
- 230000001988 toxicity Effects 0.000 description 8
- 102100031480 Dual specificity mitogen-activated protein kinase kinase 1 Human genes 0.000 description 7
- 101710146526 Dual specificity mitogen-activated protein kinase kinase 1 Proteins 0.000 description 7
- 238000002835 absorbance Methods 0.000 description 7
- 238000011319 anticancer therapy Methods 0.000 description 7
- 229960002465 dabrafenib Drugs 0.000 description 7
- HKSZLNNOFSGOKW-UHFFFAOYSA-N ent-staurosporine Natural products C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1C1CC(NC)C(OC)C4(C)O1 HKSZLNNOFSGOKW-UHFFFAOYSA-N 0.000 description 7
- 230000003993 interaction Effects 0.000 description 7
- 239000000463 material Substances 0.000 description 7
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 7
- 239000000651 prodrug Substances 0.000 description 7
- 229940002612 prodrug Drugs 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 238000010186 staining Methods 0.000 description 7
- HKSZLNNOFSGOKW-FYTWVXJKSA-N staurosporine Chemical compound C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1[C@H]1C[C@@H](NC)[C@@H](OC)[C@]4(C)O1 HKSZLNNOFSGOKW-FYTWVXJKSA-N 0.000 description 7
- 239000003826 tablet Substances 0.000 description 7
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 7
- 229940125431 BRAF inhibitor Drugs 0.000 description 6
- 208000018084 Bone neoplasm Diseases 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- 206010029260 Neuroblastoma Diseases 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 230000003213 activating effect Effects 0.000 description 6
- 238000003556 assay Methods 0.000 description 6
- 230000008901 benefit Effects 0.000 description 6
- 238000004113 cell culture Methods 0.000 description 6
- 238000006243 chemical reaction Methods 0.000 description 6
- 230000002255 enzymatic effect Effects 0.000 description 6
- 238000002347 injection Methods 0.000 description 6
- 239000007924 injection Substances 0.000 description 6
- 239000013641 positive control Substances 0.000 description 6
- 230000000861 pro-apoptotic effect Effects 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- 230000004083 survival effect Effects 0.000 description 6
- GZDRODOYEFEHGG-NUDCOPPTSA-N (4s)-4-[[(2s)-2-acetamido-3-carboxypropanoyl]amino]-5-[[(2s)-1-[[(2s)-3-carboxy-1-oxo-1-[[2-oxo-4-(trifluoromethyl)chromen-7-yl]amino]propan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]amino]-5-oxopentanoic acid Chemical compound FC(F)(F)C1=CC(=O)OC2=CC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(O)=O)NC(C)=O)C(C)C)=CC=C21 GZDRODOYEFEHGG-NUDCOPPTSA-N 0.000 description 5
- 108090000672 Annexin A5 Proteins 0.000 description 5
- 102000004121 Annexin A5 Human genes 0.000 description 5
- 101100327819 Caenorhabditis elegans chl-1 gene Proteins 0.000 description 5
- 101100247317 Physarum polycephalum RAS1 gene Proteins 0.000 description 5
- PTFCDOFLOPIGGS-UHFFFAOYSA-N Zinc dication Chemical compound [Zn+2] PTFCDOFLOPIGGS-UHFFFAOYSA-N 0.000 description 5
- 230000001093 anti-cancer Effects 0.000 description 5
- 239000002246 antineoplastic agent Substances 0.000 description 5
- 230000009925 apoptotic mechanism Effects 0.000 description 5
- 239000002775 capsule Substances 0.000 description 5
- 239000013592 cell lysate Substances 0.000 description 5
- 230000000973 chemotherapeutic effect Effects 0.000 description 5
- 229940127089 cytotoxic agent Drugs 0.000 description 5
- 230000034994 death Effects 0.000 description 5
- 238000000684 flow cytometry Methods 0.000 description 5
- 206010020718 hyperplasia Diseases 0.000 description 5
- 230000003463 hyperproliferative effect Effects 0.000 description 5
- 230000002055 immunohistochemical effect Effects 0.000 description 5
- 238000000338 in vitro Methods 0.000 description 5
- 231100000252 nontoxic Toxicity 0.000 description 5
- 230000003000 nontoxic effect Effects 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 230000002829 reductive effect Effects 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 206010005949 Bone cancer Diseases 0.000 description 4
- 241000282414 Homo sapiens Species 0.000 description 4
- 241000124008 Mammalia Species 0.000 description 4
- 206010039491 Sarcoma Diseases 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 238000003782 apoptosis assay Methods 0.000 description 4
- 230000033228 biological regulation Effects 0.000 description 4
- 210000004556 brain Anatomy 0.000 description 4
- 230000005907 cancer growth Effects 0.000 description 4
- 230000004663 cell proliferation Effects 0.000 description 4
- 208000029742 colonic neoplasm Diseases 0.000 description 4
- 208000035475 disorder Diseases 0.000 description 4
- 239000006185 dispersion Substances 0.000 description 4
- 239000000975 dye Substances 0.000 description 4
- 239000012634 fragment Substances 0.000 description 4
- 230000014509 gene expression Effects 0.000 description 4
- 230000001404 mediated effect Effects 0.000 description 4
- 108020004999 messenger RNA Proteins 0.000 description 4
- 206010061289 metastatic neoplasm Diseases 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 210000005170 neoplastic cell Anatomy 0.000 description 4
- 210000000056 organ Anatomy 0.000 description 4
- 239000008194 pharmaceutical composition Substances 0.000 description 4
- 230000005522 programmed cell death Effects 0.000 description 4
- 230000008054 signal transmission Effects 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 229940124597 therapeutic agent Drugs 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 208000003200 Adenoma Diseases 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 108010010803 Gelatin Proteins 0.000 description 3
- 208000008839 Kidney Neoplasms Diseases 0.000 description 3
- 108010047230 Member 1 Subfamily B ATP Binding Cassette Transporter Proteins 0.000 description 3
- 206010027476 Metastases Diseases 0.000 description 3
- 206010027480 Metastatic malignant melanoma Diseases 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 206010038389 Renal cancer Diseases 0.000 description 3
- 208000000453 Skin Neoplasms Diseases 0.000 description 3
- 102100030306 TBC1 domain family member 9 Human genes 0.000 description 3
- 239000007983 Tris buffer Substances 0.000 description 3
- 230000002159 abnormal effect Effects 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 208000004064 acoustic neuroma Diseases 0.000 description 3
- 239000012190 activator Substances 0.000 description 3
- 230000005756 apoptotic signaling Effects 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 238000011278 co-treatment Methods 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 239000008273 gelatin Substances 0.000 description 3
- 229920000159 gelatin Polymers 0.000 description 3
- 235000019322 gelatine Nutrition 0.000 description 3
- 235000011852 gelatine desserts Nutrition 0.000 description 3
- 230000002068 genetic effect Effects 0.000 description 3
- 208000005017 glioblastoma Diseases 0.000 description 3
- 210000003734 kidney Anatomy 0.000 description 3
- 201000010982 kidney cancer Diseases 0.000 description 3
- 231100000518 lethal Toxicity 0.000 description 3
- 230000001665 lethal effect Effects 0.000 description 3
- 208000014018 liver neoplasm Diseases 0.000 description 3
- 238000011866 long-term treatment Methods 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 230000001394 metastastic effect Effects 0.000 description 3
- 208000021039 metastatic melanoma Diseases 0.000 description 3
- MMNNTJYFHUDSKL-UHFFFAOYSA-N methyl n-[6-[2-(5-chloro-2-methylphenyl)-1-hydroxy-3-oxoisoindol-1-yl]-1h-benzimidazol-2-yl]carbamate Chemical compound C=1C=C2NC(NC(=O)OC)=NC2=CC=1C(C1=CC=CC=C1C1=O)(O)N1C1=CC(Cl)=CC=C1C MMNNTJYFHUDSKL-UHFFFAOYSA-N 0.000 description 3
- 239000013642 negative control Substances 0.000 description 3
- 239000006187 pill Substances 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 230000011664 signaling Effects 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 239000012453 solvate Chemical class 0.000 description 3
- 206010041823 squamous cell carcinoma Diseases 0.000 description 3
- 125000001424 substituent group Chemical group 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical compound OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 3
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 3
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 2
- YABJJWZLRMPFSI-UHFFFAOYSA-N 1-methyl-5-[[2-[5-(trifluoromethyl)-1H-imidazol-2-yl]-4-pyridinyl]oxy]-N-[4-(trifluoromethyl)phenyl]-2-benzimidazolamine Chemical compound N=1C2=CC(OC=3C=C(N=CC=3)C=3NC(=CN=3)C(F)(F)F)=CC=C2N(C)C=1NC1=CC=C(C(F)(F)F)C=C1 YABJJWZLRMPFSI-UHFFFAOYSA-N 0.000 description 2
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 2
- 102000007469 Actins Human genes 0.000 description 2
- 108010085238 Actins Proteins 0.000 description 2
- 208000031261 Acute myeloid leukaemia Diseases 0.000 description 2
- 206010067484 Adverse reaction Diseases 0.000 description 2
- 102000010565 Apoptosis Regulatory Proteins Human genes 0.000 description 2
- 108010063104 Apoptosis Regulatory Proteins Proteins 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 206010003571 Astrocytoma Diseases 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 241000167854 Bourreria succulenta Species 0.000 description 2
- 201000005262 Chondroma Diseases 0.000 description 2
- 208000005243 Chondrosarcoma Diseases 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 2
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- 102100030708 GTPase KRas Human genes 0.000 description 2
- 206010018338 Glioma Diseases 0.000 description 2
- DHCLVCXQIBBOPH-UHFFFAOYSA-N Glycerol 2-phosphate Chemical compound OCC(CO)OP(O)(O)=O DHCLVCXQIBBOPH-UHFFFAOYSA-N 0.000 description 2
- 239000007995 HEPES buffer Substances 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 101000584612 Homo sapiens GTPase KRas Proteins 0.000 description 2
- 208000000172 Medulloblastoma Diseases 0.000 description 2
- 241001529936 Murinae Species 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- 208000033776 Myeloid Acute Leukemia Diseases 0.000 description 2
- 206010029350 Neurotoxicity Diseases 0.000 description 2
- 201000010133 Oligodendroglioma Diseases 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- 208000012868 Overgrowth Diseases 0.000 description 2
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- 241000288906 Primates Species 0.000 description 2
- 206010060862 Prostate cancer Diseases 0.000 description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 2
- 201000000582 Retinoblastoma Diseases 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- 206010044221 Toxic encephalopathy Diseases 0.000 description 2
- 208000008383 Wilms tumor Diseases 0.000 description 2
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 208000009956 adenocarcinoma Diseases 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 208000024447 adrenal gland neoplasm Diseases 0.000 description 2
- 230000006838 adverse reaction Effects 0.000 description 2
- 239000004479 aerosol dispenser Substances 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 210000000988 bone and bone Anatomy 0.000 description 2
- 101150048834 braF gene Proteins 0.000 description 2
- 208000003362 bronchogenic carcinoma Diseases 0.000 description 2
- 239000006172 buffering agent Substances 0.000 description 2
- 230000001413 cellular effect Effects 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 210000003169 central nervous system Anatomy 0.000 description 2
- 235000019693 cherries Nutrition 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- 238000011284 combination treatment Methods 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 208000035250 cutaneous malignant susceptibility to 1 melanoma Diseases 0.000 description 2
- 239000002254 cytotoxic agent Substances 0.000 description 2
- 231100000599 cytotoxic agent Toxicity 0.000 description 2
- 238000007405 data analysis Methods 0.000 description 2
- 230000007547 defect Effects 0.000 description 2
- 230000002950 deficient Effects 0.000 description 2
- 230000001934 delay Effects 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000000890 drug combination Substances 0.000 description 2
- 238000002651 drug therapy Methods 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 235000003599 food sweetener Nutrition 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 201000009277 hairy cell leukemia Diseases 0.000 description 2
- 230000002489 hematologic effect Effects 0.000 description 2
- 230000002390 hyperplastic effect Effects 0.000 description 2
- 238000011532 immunohistochemical staining Methods 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000001802 infusion Methods 0.000 description 2
- 230000002147 killing effect Effects 0.000 description 2
- CMJCXYNUCSMDBY-ZDUSSCGKSA-N lgx818 Chemical compound COC(=O)N[C@@H](C)CNC1=NC=CC(C=2C(=NN(C=2)C(C)C)C=2C(=C(NS(C)(=O)=O)C=C(Cl)C=2)F)=N1 CMJCXYNUCSMDBY-ZDUSSCGKSA-N 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 201000007270 liver cancer Diseases 0.000 description 2
- 210000004072 lung Anatomy 0.000 description 2
- 210000003563 lymphoid tissue Anatomy 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 206010027191 meningioma Diseases 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 238000010172 mouse model Methods 0.000 description 2
- 230000001613 neoplastic effect Effects 0.000 description 2
- 201000008026 nephroblastoma Diseases 0.000 description 2
- 231100000228 neurotoxicity Toxicity 0.000 description 2
- 230000007135 neurotoxicity Effects 0.000 description 2
- 230000002188 osteogenic effect Effects 0.000 description 2
- 230000002018 overexpression Effects 0.000 description 2
- 201000002528 pancreatic cancer Diseases 0.000 description 2
- 208000008443 pancreatic carcinoma Diseases 0.000 description 2
- 238000009520 phase I clinical trial Methods 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 208000029340 primitive neuroectodermal tumor Diseases 0.000 description 2
- 230000000069 prophylactic effect Effects 0.000 description 2
- 238000011002 quantification Methods 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 230000007420 reactivation Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 238000012216 screening Methods 0.000 description 2
- 201000000849 skin cancer Diseases 0.000 description 2
- 210000003625 skull Anatomy 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 239000003765 sweetening agent Substances 0.000 description 2
- 239000011885 synergistic combination Substances 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 238000002626 targeted therapy Methods 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- URAYPUMNDPQOKB-UHFFFAOYSA-N triacetin Chemical compound CC(=O)OCC(OC(C)=O)COC(C)=O URAYPUMNDPQOKB-UHFFFAOYSA-N 0.000 description 2
- 230000004614 tumor growth Effects 0.000 description 2
- 230000004222 uncontrolled growth Effects 0.000 description 2
- 235000015112 vegetable and seed oil Nutrition 0.000 description 2
- 239000008158 vegetable oil Substances 0.000 description 2
- GFMMXOIFOQCCGU-UHFFFAOYSA-N 2-(2-chloro-4-iodoanilino)-N-(cyclopropylmethoxy)-3,4-difluorobenzamide Chemical compound C=1C=C(I)C=C(Cl)C=1NC1=C(F)C(F)=CC=C1C(=O)NOCC1CC1 GFMMXOIFOQCCGU-UHFFFAOYSA-N 0.000 description 1
- KPGXRSRHYNQIFN-UHFFFAOYSA-N 2-oxoglutaric acid Chemical compound OC(=O)CCC(=O)C(O)=O KPGXRSRHYNQIFN-UHFFFAOYSA-N 0.000 description 1
- RCLQNICOARASSR-SECBINFHSA-N 3-[(2r)-2,3-dihydroxypropyl]-6-fluoro-5-(2-fluoro-4-iodoanilino)-8-methylpyrido[2,3-d]pyrimidine-4,7-dione Chemical compound FC=1C(=O)N(C)C=2N=CN(C[C@@H](O)CO)C(=O)C=2C=1NC1=CC=C(I)C=C1F RCLQNICOARASSR-SECBINFHSA-N 0.000 description 1
- HFDKKNHCYWNNNQ-YOGANYHLSA-N 75976-10-2 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@@H](NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](C)N)C(C)C)[C@@H](C)O)C1=CC=C(O)C=C1 HFDKKNHCYWNNNQ-YOGANYHLSA-N 0.000 description 1
- GLANOOJJBKXTMI-UHFFFAOYSA-N 9-(9-formylfluoren-9-yl)fluorene-9-carbaldehyde Chemical compound C12=CC=CC=C2C2=CC=CC=C2C1(C=O)C1(C=O)C2=CC=CC=C2C2=CC=CC=C21 GLANOOJJBKXTMI-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 208000024893 Acute lymphoblastic leukemia Diseases 0.000 description 1
- 208000014697 Acute lymphocytic leukaemia Diseases 0.000 description 1
- 206010001233 Adenoma benign Diseases 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 108091093088 Amplicon Proteins 0.000 description 1
- 206010061424 Anal cancer Diseases 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- 208000032791 BCR-ABL1 positive chronic myelogenous leukemia Diseases 0.000 description 1
- 206010004146 Basal cell carcinoma Diseases 0.000 description 1
- KHBQMWCZKVMBLN-UHFFFAOYSA-N Benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1 KHBQMWCZKVMBLN-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical class OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 206010005003 Bladder cancer Diseases 0.000 description 1
- 208000008720 Bone Marrow Neoplasms Diseases 0.000 description 1
- 206010065553 Bone marrow failure Diseases 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000282465 Canis Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 208000005623 Carcinogenesis Diseases 0.000 description 1
- 201000000274 Carcinosarcoma Diseases 0.000 description 1
- 102000004041 Caspase 7 Human genes 0.000 description 1
- 108090000567 Caspase 7 Proteins 0.000 description 1
- 108090000426 Caspase-1 Proteins 0.000 description 1
- 102000004091 Caspase-8 Human genes 0.000 description 1
- 108090000538 Caspase-8 Proteins 0.000 description 1
- 102100026550 Caspase-9 Human genes 0.000 description 1
- 108090000566 Caspase-9 Proteins 0.000 description 1
- 206010007953 Central nervous system lymphoma Diseases 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- 201000009047 Chordoma Diseases 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 208000009798 Craniopharyngioma Diseases 0.000 description 1
- 201000005171 Cystadenoma Diseases 0.000 description 1
- 102000009058 Death Domain Receptors Human genes 0.000 description 1
- 108010049207 Death Domain Receptors Proteins 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 235000019739 Dicalciumphosphate Nutrition 0.000 description 1
- 102100023266 Dual specificity mitogen-activated protein kinase kinase 2 Human genes 0.000 description 1
- 101710146529 Dual specificity mitogen-activated protein kinase kinase 2 Proteins 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 206010057649 Endometrial sarcoma Diseases 0.000 description 1
- 208000005431 Endometrioid Carcinoma Diseases 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 206010014967 Ependymoma Diseases 0.000 description 1
- 201000005231 Epithelioid sarcoma Diseases 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- 208000000461 Esophageal Neoplasms Diseases 0.000 description 1
- 208000006168 Ewing Sarcoma Diseases 0.000 description 1
- 108010040476 FITC-annexin A5 Proteins 0.000 description 1
- 241000282324 Felis Species 0.000 description 1
- 201000008808 Fibrosarcoma Diseases 0.000 description 1
- 102100039788 GTPase NRas Human genes 0.000 description 1
- 208000022072 Gallbladder Neoplasms Diseases 0.000 description 1
- 201000004066 Ganglioglioma Diseases 0.000 description 1
- 208000032612 Glial tumor Diseases 0.000 description 1
- 201000010915 Glioblastoma multiforme Diseases 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 208000002125 Hemangioendothelioma Diseases 0.000 description 1
- 208000017604 Hodgkin disease Diseases 0.000 description 1
- 208000021519 Hodgkin lymphoma Diseases 0.000 description 1
- 208000010747 Hodgkins lymphoma Diseases 0.000 description 1
- 101000744505 Homo sapiens GTPase NRas Proteins 0.000 description 1
- 208000029966 Hutchinson Melanotic Freckle Diseases 0.000 description 1
- 206010062767 Hypophysitis Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 208000018142 Leiomyosarcoma Diseases 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 208000036241 Liver adenomatosis Diseases 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-L Malonate Chemical compound [O-]C(=O)CC([O-])=O OFOBLEOULBTSOW-UHFFFAOYSA-L 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 108090000744 Mitogen-Activated Protein Kinase Kinases Proteins 0.000 description 1
- 102000004232 Mitogen-Activated Protein Kinase Kinases Human genes 0.000 description 1
- 241001430197 Mollicutes Species 0.000 description 1
- 241000204031 Mycoplasma Species 0.000 description 1
- 206010029488 Nodular melanoma Diseases 0.000 description 1
- 208000015914 Non-Hodgkin lymphomas Diseases 0.000 description 1
- 206010030155 Oesophageal carcinoma Diseases 0.000 description 1
- 238000009004 PCR Kit Methods 0.000 description 1
- SUDAHWBOROXANE-SECBINFHSA-N PD 0325901 Chemical compound OC[C@@H](O)CONC(=O)C1=CC=C(F)C(F)=C1NC1=CC=C(I)C=C1F SUDAHWBOROXANE-SECBINFHSA-N 0.000 description 1
- 102000018886 Pancreatic Polypeptide Human genes 0.000 description 1
- 206010033701 Papillary thyroid cancer Diseases 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 102000004160 Phosphoric Monoester Hydrolases Human genes 0.000 description 1
- 108090000608 Phosphoric Monoester Hydrolases Proteins 0.000 description 1
- 108091000080 Phosphotransferase Proteins 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 208000007452 Plasmacytoma Diseases 0.000 description 1
- 229920000776 Poly(Adenosine diphosphate-ribose) polymerase Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 208000006994 Precancerous Conditions Diseases 0.000 description 1
- 208000006664 Precursor Cell Lymphoblastic Leukemia-Lymphoma Diseases 0.000 description 1
- 102000001253 Protein Kinase Human genes 0.000 description 1
- 206010051807 Pseudosarcoma Diseases 0.000 description 1
- 201000008183 Pulmonary blastoma Diseases 0.000 description 1
- 239000012083 RIPA buffer Substances 0.000 description 1
- 239000012979 RPMI medium Substances 0.000 description 1
- 238000011529 RT qPCR Methods 0.000 description 1
- 208000006265 Renal cell carcinoma Diseases 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 229920001800 Shellac Polymers 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 206010041067 Small cell lung cancer Diseases 0.000 description 1
- 208000021712 Soft tissue sarcoma Diseases 0.000 description 1
- 102000005157 Somatostatin Human genes 0.000 description 1
- 108010056088 Somatostatin Proteins 0.000 description 1
- 208000005718 Stomach Neoplasms Diseases 0.000 description 1
- 238000000692 Student's t-test Methods 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 101000983124 Sus scrofa Pancreatic prohormone precursor Proteins 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 1
- 208000014070 Vestibular schwannoma Diseases 0.000 description 1
- 208000012018 Yolk sac tumor Diseases 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 239000002250 absorbent Substances 0.000 description 1
- 230000002745 absorbent Effects 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 206010000583 acral lentiginous melanoma Diseases 0.000 description 1
- 208000009621 actinic keratosis Diseases 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 208000002517 adenoid cystic carcinoma Diseases 0.000 description 1
- 201000001256 adenosarcoma Diseases 0.000 description 1
- 201000008395 adenosquamous carcinoma Diseases 0.000 description 1
- 201000005188 adrenal gland cancer Diseases 0.000 description 1
- 238000012382 advanced drug delivery Methods 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 239000011543 agarose gel Substances 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 210000004727 amygdala Anatomy 0.000 description 1
- 201000007696 anal canal cancer Diseases 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 229940124650 anti-cancer therapies Drugs 0.000 description 1
- 230000003466 anti-cipated effect Effects 0.000 description 1
- 230000003127 anti-melanomic effect Effects 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 229940041181 antineoplastic drug Drugs 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 229940072107 ascorbate Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- 239000002774 b raf kinase inhibitor Substances 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 150000007514 bases Chemical class 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 229940050390 benzoate Drugs 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 230000001588 bifunctional effect Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- ACWZRVQXLIRSDF-UHFFFAOYSA-N binimetinib Chemical compound OCCONC(=O)C=1C=C2N(C)C=NC2=C(F)C=1NC1=CC=C(Br)C=C1F ACWZRVQXLIRSDF-UHFFFAOYSA-N 0.000 description 1
- 238000002306 biochemical method Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 230000008499 blood brain barrier function Effects 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 210000001218 blood-brain barrier Anatomy 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 201000006491 bone marrow cancer Diseases 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- 239000006189 buccal tablet Substances 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 238000010805 cDNA synthesis kit Methods 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 230000036952 cancer formation Effects 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 231100000504 carcinogenesis Toxicity 0.000 description 1
- 208000002458 carcinoid tumor Diseases 0.000 description 1
- 238000001516 cell proliferation assay Methods 0.000 description 1
- 230000005754 cellular signaling Effects 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- 208000006990 cholangiocarcinoma Diseases 0.000 description 1
- 208000006571 choroid plexus carcinoma Diseases 0.000 description 1
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 1
- 229960004316 cisplatin Drugs 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 208000009060 clear cell adenocarcinoma Diseases 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000002301 combined effect Effects 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 210000003792 cranial nerve Anatomy 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- NEFBYIFKOOEVPA-UHFFFAOYSA-K dicalcium phosphate Chemical compound [Ca+2].[Ca+2].[O-]P([O-])([O-])=O NEFBYIFKOOEVPA-UHFFFAOYSA-K 0.000 description 1
- 229940038472 dicalcium phosphate Drugs 0.000 description 1
- 229910000390 dicalcium phosphate Inorganic materials 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- UGMCXQCYOVCMTB-UHFFFAOYSA-K dihydroxy(stearato)aluminium Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[Al](O)O UGMCXQCYOVCMTB-UHFFFAOYSA-K 0.000 description 1
- 229940042399 direct acting antivirals protease inhibitors Drugs 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002612 dispersion medium Substances 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- 238000009510 drug design Methods 0.000 description 1
- 238000009509 drug development Methods 0.000 description 1
- 230000002900 effect on cell Effects 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 208000001991 endodermal sinus tumor Diseases 0.000 description 1
- 201000003908 endometrial adenocarcinoma Diseases 0.000 description 1
- 201000006828 endometrial hyperplasia Diseases 0.000 description 1
- 208000028730 endometrioid adenocarcinoma Diseases 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 201000004101 esophageal cancer Diseases 0.000 description 1
- 210000003238 esophagus Anatomy 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 208000030533 eye disease Diseases 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 150000002191 fatty alcohols Chemical class 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 239000012737 fresh medium Substances 0.000 description 1
- 210000000232 gallbladder Anatomy 0.000 description 1
- 201000010175 gallbladder cancer Diseases 0.000 description 1
- 201000008361 ganglioneuroma Diseases 0.000 description 1
- 206010017758 gastric cancer Diseases 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 125000005908 glyceryl ester group Chemical group 0.000 description 1
- 239000001087 glyceryl triacetate Substances 0.000 description 1
- 235000013773 glyceryl triacetate Nutrition 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 208000037824 growth disorder Diseases 0.000 description 1
- 230000009036 growth inhibition Effects 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 201000010536 head and neck cancer Diseases 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 201000011066 hemangioma Diseases 0.000 description 1
- 210000000777 hematopoietic system Anatomy 0.000 description 1
- 206010073071 hepatocellular carcinoma Diseases 0.000 description 1
- 231100000844 hepatocellular carcinoma Toxicity 0.000 description 1
- 230000013632 homeostatic process Effects 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 125000000717 hydrazino group Chemical group [H]N([*])N([H])[H] 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 238000003119 immunoblot Methods 0.000 description 1
- 238000009169 immunotherapy Methods 0.000 description 1
- 201000004933 in situ carcinoma Diseases 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 239000003978 infusion fluid Substances 0.000 description 1
- 239000007972 injectable composition Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 206010022498 insulinoma Diseases 0.000 description 1
- 238000007917 intracranial administration Methods 0.000 description 1
- 208000020082 intraepithelial neoplasia Diseases 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 150000008040 ionic compounds Chemical class 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- 229940043355 kinase inhibitor Drugs 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 208000003849 large cell carcinoma Diseases 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 231100000845 liver adenoma Toxicity 0.000 description 1
- 208000019423 liver disease Diseases 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 210000004324 lymphatic system Anatomy 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 201000010893 malignant breast melanoma Diseases 0.000 description 1
- 208000006178 malignant mesothelioma Diseases 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 108010082117 matrigel Proteins 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- 229940126601 medicinal product Drugs 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 239000001525 mentha piperita l. herb oil Substances 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- OSWPMRLSEDHDFF-UHFFFAOYSA-N methyl salicylate Chemical compound COC(=O)C1=CC=CC=C1O OSWPMRLSEDHDFF-UHFFFAOYSA-N 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 210000003470 mitochondria Anatomy 0.000 description 1
- 230000000869 mutational effect Effects 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- UPSFMJHZUCSEHU-JYGUBCOQSA-N n-[(2s,3r,4r,5s,6r)-2-[(2r,3s,4r,5r,6s)-5-acetamido-4-hydroxy-2-(hydroxymethyl)-6-(4-methyl-2-oxochromen-7-yl)oxyoxan-3-yl]oxy-4,5-dihydroxy-6-(hydroxymethyl)oxan-3-yl]acetamide Chemical compound CC(=O)N[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@H]1[C@H](O)[C@@H](NC(C)=O)[C@H](OC=2C=C3OC(=O)C=C(C)C3=CC=2)O[C@@H]1CO UPSFMJHZUCSEHU-JYGUBCOQSA-N 0.000 description 1
- 239000006199 nebulizer Substances 0.000 description 1
- 230000008479 neoplastic tissue growth Effects 0.000 description 1
- 208000007538 neurilemmoma Diseases 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 201000000032 nodular malignant melanoma Diseases 0.000 description 1
- 229920002113 octoxynol Polymers 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 231100000590 oncogenic Toxicity 0.000 description 1
- 230000002246 oncogenic effect Effects 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 208000008798 osteoma Diseases 0.000 description 1
- 210000001672 ovary Anatomy 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 208000021255 pancreatic insulinoma Diseases 0.000 description 1
- 201000005163 papillary serous adenocarcinoma Diseases 0.000 description 1
- 208000024641 papillary serous cystadenocarcinoma Diseases 0.000 description 1
- 208000003154 papilloma Diseases 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 235000019477 peppermint oil Nutrition 0.000 description 1
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 1
- 229960003742 phenol Drugs 0.000 description 1
- 102000020233 phosphotransferase Human genes 0.000 description 1
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 1
- 210000004560 pineal gland Anatomy 0.000 description 1
- 125000004194 piperazin-1-yl group Chemical group [H]N1C([H])([H])C([H])([H])N(*)C([H])([H])C1([H])[H] 0.000 description 1
- 230000001817 pituitary effect Effects 0.000 description 1
- 210000003635 pituitary gland Anatomy 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000037452 priming Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 210000002307 prostate Anatomy 0.000 description 1
- 108060006633 protein kinase Proteins 0.000 description 1
- 230000017854 proteolysis Effects 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 238000003753 real-time PCR Methods 0.000 description 1
- 230000011514 reflex Effects 0.000 description 1
- 201000000443 refractory hairy cell leukemia Diseases 0.000 description 1
- 208000016691 refractory malignant neoplasm Diseases 0.000 description 1
- 238000010839 reverse transcription Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 201000009410 rhabdomyosarcoma Diseases 0.000 description 1
- 201000007416 salivary gland adenoid cystic carcinoma Diseases 0.000 description 1
- 206010039667 schwannoma Diseases 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- CYOHGALHFOKKQC-UHFFFAOYSA-N selumetinib Chemical compound OCCONC(=O)C=1C=C2N(C)C=NC2=C(F)C=1NC1=CC=C(Br)C=C1Cl CYOHGALHFOKKQC-UHFFFAOYSA-N 0.000 description 1
- 229950010746 selumetinib Drugs 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000011452 sequencing regimen Methods 0.000 description 1
- 238000012163 sequencing technique Methods 0.000 description 1
- 208000004548 serous cystadenocarcinoma Diseases 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- ZLGIYFNHBLSMPS-ATJNOEHPSA-N shellac Chemical compound OCCCCCC(O)C(O)CCCCCCCC(O)=O.C1C23[C@H](C(O)=O)CCC2[C@](C)(CO)[C@@H]1C(C(O)=O)=C[C@@H]3O ZLGIYFNHBLSMPS-ATJNOEHPSA-N 0.000 description 1
- 239000004208 shellac Substances 0.000 description 1
- 229940113147 shellac Drugs 0.000 description 1
- 235000013874 shellac Nutrition 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 231100000161 signs of toxicity Toxicity 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 208000000649 small cell carcinoma Diseases 0.000 description 1
- 208000000587 small cell lung carcinoma Diseases 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 210000004872 soft tissue Anatomy 0.000 description 1
- 239000007909 solid dosage form Substances 0.000 description 1
- 239000007892 solid unit dosage form Substances 0.000 description 1
- NHXLMOGPVYXJNR-ATOGVRKGSA-N somatostatin Chemical compound C([C@H]1C(=O)N[C@H](C(N[C@@H](CO)C(=O)N[C@@H](CSSC[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(=O)N1)[C@@H](C)O)NC(=O)CNC(=O)[C@H](C)N)C(O)=O)=O)[C@H](O)C)C1=CC=CC=C1 NHXLMOGPVYXJNR-ATOGVRKGSA-N 0.000 description 1
- 229960000553 somatostatin Drugs 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 238000011255 standard chemotherapy Methods 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 238000011301 standard therapy Methods 0.000 description 1
- CGPUWJWCVCFERF-UHFFFAOYSA-N staurosporine Natural products C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1C1CC(NC)C(OC)C4(OC)O1 CGPUWJWCVCFERF-UHFFFAOYSA-N 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 201000011549 stomach cancer Diseases 0.000 description 1
- 229940086735 succinate Drugs 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 238000011477 surgical intervention Methods 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 238000012353 t test Methods 0.000 description 1
- 229940081616 tafinlar Drugs 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- RTKIYNMVFMVABJ-UHFFFAOYSA-L thimerosal Chemical compound [Na+].CC[Hg]SC1=CC=CC=C1C([O-])=O RTKIYNMVFMVABJ-UHFFFAOYSA-L 0.000 description 1
- 229940033663 thimerosal Drugs 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 208000030045 thyroid gland papillary carcinoma Diseases 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 206010044412 transitional cell carcinoma Diseases 0.000 description 1
- ODLHGICHYURWBS-LKONHMLTSA-N trappsol cyclo Chemical compound CC(O)COC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)COCC(O)C)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1COCC(C)O ODLHGICHYURWBS-LKONHMLTSA-N 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 229960002622 triacetin Drugs 0.000 description 1
- 230000003827 upregulation Effects 0.000 description 1
- 201000005112 urinary bladder cancer Diseases 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 201000011531 vascular cancer Diseases 0.000 description 1
- 206010055031 vascular neoplasm Diseases 0.000 description 1
- 238000012795 verification Methods 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 239000009637 wintergreen oil Substances 0.000 description 1
- 229940034727 zelboraf Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/4184—1,3-Diazoles condensed with carbocyclic rings, e.g. benzimidazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/427—Thiazoles not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/437—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a five-membered ring having nitrogen as a ring hetero atom, e.g. indolizine, beta-carboline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/12—Aerosols; Foams
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2054—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2059—Starch, including chemically or physically modified derivatives; Amylose; Amylopectin; Dextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4841—Filling excipients; Inactive ingredients
- A61K9/4866—Organic macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Dispersion Chemistry (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Plant Substances (AREA)
- Medicinal Preparation (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201562171882P | 2015-06-05 | 2015-06-05 | |
| US62/171,882 | 2015-06-05 | ||
| US201662345629P | 2016-06-03 | 2016-06-03 | |
| US62/345,629 | 2016-06-03 | ||
| PCT/US2016/036063 WO2016197129A1 (en) | 2015-06-05 | 2016-06-06 | Pac-1 combination therapy |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| RU2017146346A RU2017146346A (ru) | 2019-07-09 |
| RU2017146346A3 RU2017146346A3 (enExample) | 2019-11-11 |
| RU2720509C2 true RU2720509C2 (ru) | 2020-04-30 |
Family
ID=57442182
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2017146346A RU2720509C2 (ru) | 2015-06-05 | 2016-06-06 | Комбинированная терапия pac-1 |
Country Status (14)
| Country | Link |
|---|---|
| US (3) | US10350207B2 (enExample) |
| EP (1) | EP3302478B1 (enExample) |
| JP (1) | JP7033451B2 (enExample) |
| KR (1) | KR102656027B1 (enExample) |
| CN (2) | CN108135896A (enExample) |
| AU (1) | AU2016271527B2 (enExample) |
| BR (1) | BR112017026140A2 (enExample) |
| CA (1) | CA2987340C (enExample) |
| DK (1) | DK3302478T3 (enExample) |
| ES (1) | ES2906763T3 (enExample) |
| IL (1) | IL256111B2 (enExample) |
| MX (1) | MX380835B (enExample) |
| RU (1) | RU2720509C2 (enExample) |
| WO (1) | WO2016197129A1 (enExample) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2866021C (en) * | 2012-03-06 | 2020-09-22 | The Board Of Trustees Of The University Of Illinois | Procaspace 3 activation by pac-1 combination therapy |
| IL256111B2 (en) * | 2015-06-05 | 2023-09-01 | Univ Illinois | Treatment of pac combinations |
| ES2928773T3 (es) | 2017-01-17 | 2022-11-22 | Heparegenix Gmbh | Inhibidores de proteína cinasas para fomentar la regeneración hepática o reducir o prevenir la muerte de hepatocitos |
| AU2018368453B2 (en) * | 2017-11-17 | 2024-05-30 | The Board Of Trustees Of The University Of Illinois | Cancer therapy by degrading dual MEK signaling |
| CA3114385A1 (en) * | 2018-10-05 | 2020-04-09 | The Board Of Trustees Of The University Of Illinois | Combination therapy for the treatment of uveal melanoma |
| EP3976062A4 (en) * | 2019-05-30 | 2023-06-14 | The Board Of Trustees Of The University Of Illinois | PROCASPASE-3 ACTIVATION AND IMMUNOTHERAPY FOR THE TREATMENT OF CANCER |
| US20230355581A1 (en) * | 2020-07-02 | 2023-11-09 | The Board of Regents of the Unversity of Texas System | Methods of treatment for melanoma |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010091383A2 (en) * | 2009-02-09 | 2010-08-12 | St. Jude Medical | Inflatable minimally invasive system for delivering and securing an annular implant |
| WO2010091382A1 (en) * | 2009-02-09 | 2010-08-12 | The Board Of Trustees Of The University Of Illinois | Design, synthesis and evaluation of procaspase activating compounds as personalized anti-cancer drugs |
| WO2013134307A1 (en) * | 2012-03-05 | 2013-09-12 | Ronald Mark Buck | Top mounting bottle container |
| WO2013134398A1 (en) * | 2012-03-06 | 2013-09-12 | The Board Of Trustees Of The University Of Illinois | Procaspase combination therapy for glioblastoma |
| WO2014072357A1 (en) * | 2012-11-06 | 2014-05-15 | Interna Technologies B.V. | Combination for use in treating diseases or conditions associated with melanoma, or treating diseases or conditions associated with activated b-raf pathway |
| WO2014138279A1 (en) * | 2013-03-05 | 2014-09-12 | University Of Tennessee Research Foundation | Compounds for treatment of cancer |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4938949A (en) | 1988-09-12 | 1990-07-03 | University Of New York | Treatment of damaged bone marrow and dosage units therefor |
| FR2757866B1 (fr) | 1996-12-30 | 2004-12-17 | Catalyse | Polymeres comportant des groupes ammoniums quaternaires, leur utilisation pour la fabrication d'un materiau a propretes antibacteriennes et leurs procedes de preparation |
| US6833373B1 (en) | 1998-12-23 | 2004-12-21 | G.D. Searle & Co. | Method of using an integrin antagonist and one or more antineoplastic agents as a combination therapy in the treatment of neoplasia |
| WO2008134474A2 (en) | 2007-04-27 | 2008-11-06 | The Board Of Trustees Of The University Of Illinois | Compositions and methods including cell death inducers and procaspase activation |
| US20070049602A1 (en) | 2005-05-26 | 2007-03-01 | The Board Of Trustees Of The University Of Illinois | Selective Apoptotic Induction in Cancer Cells Including Activation of Procaspase-3 |
| WO2012178038A1 (en) * | 2011-06-24 | 2012-12-27 | The Broad Institute, Inc. | Methods of treating cancer |
| US8916705B2 (en) | 2011-10-14 | 2014-12-23 | The Board of Trustees of The University of Illilnois | Procaspase-activating compounds and compositions |
| AU2013225682B2 (en) | 2012-03-02 | 2016-04-14 | The Board Of Trustees Of The University Of Illinois | Potent anticancer activity via dual compound activation |
| CA2866021C (en) * | 2012-03-06 | 2020-09-22 | The Board Of Trustees Of The University Of Illinois | Procaspace 3 activation by pac-1 combination therapy |
| US20150141470A1 (en) * | 2012-05-08 | 2015-05-21 | The Broad Institute, Inc. | Diagnostic and treatment methods in patients having or at risk of developing resistance to cancer therapy |
| US9249116B2 (en) | 2012-08-03 | 2016-02-02 | The Board Of Trustees Of The University Of Illinois | Enzyme-activating compounds and compositions |
| ES2822665T3 (es) * | 2013-05-31 | 2021-05-04 | Merck Sharp & Dohme | Terapias de combinación para el cáncer |
| CN111053768B (zh) * | 2013-07-12 | 2023-12-12 | 皮拉马尔企业有限公司 | 用于治疗黑素瘤的药物组合 |
| IL256111B2 (en) * | 2015-06-05 | 2023-09-01 | Univ Illinois | Treatment of pac combinations |
-
2016
- 2016-06-06 IL IL256111A patent/IL256111B2/en unknown
- 2016-06-06 BR BR112017026140-5A patent/BR112017026140A2/pt not_active Application Discontinuation
- 2016-06-06 AU AU2016271527A patent/AU2016271527B2/en active Active
- 2016-06-06 CN CN201680045123.4A patent/CN108135896A/zh active Pending
- 2016-06-06 EP EP16804655.5A patent/EP3302478B1/en active Active
- 2016-06-06 RU RU2017146346A patent/RU2720509C2/ru active
- 2016-06-06 CN CN202311252422.3A patent/CN117257802A/zh active Pending
- 2016-06-06 CA CA2987340A patent/CA2987340C/en active Active
- 2016-06-06 ES ES16804655T patent/ES2906763T3/es active Active
- 2016-06-06 WO PCT/US2016/036063 patent/WO2016197129A1/en not_active Ceased
- 2016-06-06 JP JP2017562987A patent/JP7033451B2/ja active Active
- 2016-06-06 US US15/579,689 patent/US10350207B2/en active Active
- 2016-06-06 DK DK16804655.5T patent/DK3302478T3/da active
- 2016-06-06 KR KR1020187000157A patent/KR102656027B1/ko active Active
- 2016-06-06 MX MX2017015532A patent/MX380835B/es unknown
-
2019
- 2019-06-10 US US16/436,542 patent/US11129830B2/en active Active
-
2021
- 2021-09-28 US US17/487,473 patent/US12336992B2/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010091383A2 (en) * | 2009-02-09 | 2010-08-12 | St. Jude Medical | Inflatable minimally invasive system for delivering and securing an annular implant |
| WO2010091382A1 (en) * | 2009-02-09 | 2010-08-12 | The Board Of Trustees Of The University Of Illinois | Design, synthesis and evaluation of procaspase activating compounds as personalized anti-cancer drugs |
| WO2013134307A1 (en) * | 2012-03-05 | 2013-09-12 | Ronald Mark Buck | Top mounting bottle container |
| WO2013134398A1 (en) * | 2012-03-06 | 2013-09-12 | The Board Of Trustees Of The University Of Illinois | Procaspase combination therapy for glioblastoma |
| WO2014072357A1 (en) * | 2012-11-06 | 2014-05-15 | Interna Technologies B.V. | Combination for use in treating diseases or conditions associated with melanoma, or treating diseases or conditions associated with activated b-raf pathway |
| WO2014138279A1 (en) * | 2013-03-05 | 2014-09-12 | University Of Tennessee Research Foundation | Compounds for treatment of cancer |
Also Published As
| Publication number | Publication date |
|---|---|
| RU2017146346A (ru) | 2019-07-09 |
| WO2016197129A1 (en) | 2016-12-08 |
| US10350207B2 (en) | 2019-07-16 |
| KR20180083842A (ko) | 2018-07-23 |
| EP3302478A4 (en) | 2019-02-27 |
| DK3302478T3 (da) | 2022-02-14 |
| IL256111A (en) | 2018-02-28 |
| US20180161326A1 (en) | 2018-06-14 |
| EP3302478B1 (en) | 2021-11-17 |
| CA2987340C (en) | 2024-04-02 |
| AU2016271527B2 (en) | 2021-09-02 |
| US11129830B2 (en) | 2021-09-28 |
| CA2987340A1 (en) | 2016-12-08 |
| MX2017015532A (es) | 2018-04-30 |
| ES2906763T3 (es) | 2022-04-20 |
| CN108135896A (zh) | 2018-06-08 |
| MX380835B (es) | 2025-03-12 |
| JP2018516936A (ja) | 2018-06-28 |
| US20190358229A1 (en) | 2019-11-28 |
| US20220088019A1 (en) | 2022-03-24 |
| JP7033451B2 (ja) | 2022-03-10 |
| US12336992B2 (en) | 2025-06-24 |
| IL256111B2 (en) | 2023-09-01 |
| IL256111B1 (en) | 2023-05-01 |
| KR102656027B1 (ko) | 2024-04-08 |
| BR112017026140A2 (pt) | 2018-08-14 |
| RU2017146346A3 (enExample) | 2019-11-11 |
| EP3302478A1 (en) | 2018-04-11 |
| AU2016271527A1 (en) | 2017-12-14 |
| CN117257802A (zh) | 2023-12-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2720509C2 (ru) | Комбинированная терапия pac-1 | |
| AU2013203637B2 (en) | Combination therapy for proliferative disorders | |
| JP2023145467A (ja) | 組み合わせ療法 | |
| RU2695228C2 (ru) | Прерывистое введение ингибитора mdm2 | |
| JP4130179B2 (ja) | 骨髄腫を処置するためのc−kit阻害剤の使用 | |
| RU2607596C2 (ru) | Комбинированная противораковая терапия | |
| CA2992221A1 (en) | Mdm2 inhibitors and combinations thereof | |
| US20210290616A1 (en) | Procaspase combination therapy for glioblastoma | |
| US20140348819A1 (en) | Methods of Treating Cancer | |
| CN107427522A (zh) | 用于治疗黑素瘤的阿吡莫德 | |
| CN119486713A (zh) | 尼罗加司他治疗 | |
| JP7696299B2 (ja) | Gapdhを阻害する方法及び組成物 | |
| WO2024220427A1 (en) | Treatment of cancer with a mek kinase inhibitor | |
| RU2724341C1 (ru) | Комбинированная композиция для предупреждения или лечения рака, содержащая производные бензофенонтиазола в качестве vda и ингибитор топоизомеразы | |
| HK1253828B (en) | Pac-1 combination therapy | |
| Peh | Procaspase-3 activation as a strategy to overcome resistance to targeted anticancer therapies |