KR20200043555A - Supplement food composition for diet - Google Patents
Supplement food composition for diet Download PDFInfo
- Publication number
- KR20200043555A KR20200043555A KR1020180123383A KR20180123383A KR20200043555A KR 20200043555 A KR20200043555 A KR 20200043555A KR 1020180123383 A KR1020180123383 A KR 1020180123383A KR 20180123383 A KR20180123383 A KR 20180123383A KR 20200043555 A KR20200043555 A KR 20200043555A
- Authority
- KR
- South Korea
- Prior art keywords
- weight
- extract
- chitosan
- carnitine
- parts
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 53
- 235000005911 diet Nutrition 0.000 title claims abstract description 19
- 230000037213 diet Effects 0.000 title claims abstract description 19
- 235000013305 food Nutrition 0.000 title abstract description 30
- 239000013589 supplement Substances 0.000 title abstract description 6
- 239000000284 extract Substances 0.000 claims abstract description 29
- 229920001661 Chitosan Polymers 0.000 claims abstract description 27
- PHIQHXFUZVPYII-ZCFIWIBFSA-N (R)-carnitine Chemical compound C[N+](C)(C)C[C@H](O)CC([O-])=O PHIQHXFUZVPYII-ZCFIWIBFSA-N 0.000 claims abstract description 24
- 102000004169 proteins and genes Human genes 0.000 claims abstract description 23
- 108090000623 proteins and genes Proteins 0.000 claims abstract description 23
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims abstract description 21
- 239000011777 magnesium Substances 0.000 claims abstract description 20
- 229910052749 magnesium Inorganic materials 0.000 claims abstract description 20
- 244000134552 Plantago ovata Species 0.000 claims abstract description 8
- 235000003421 Plantago ovata Nutrition 0.000 claims abstract description 8
- 235000015872 dietary supplement Nutrition 0.000 claims description 28
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 claims description 28
- 244000046052 Phaseolus vulgaris Species 0.000 claims description 24
- ZMJBYMUCKBYSCP-UHFFFAOYSA-N Hydroxycitric acid Chemical compound OC(=O)C(O)C(O)(C(O)=O)CC(O)=O ZMJBYMUCKBYSCP-UHFFFAOYSA-N 0.000 claims description 22
- 235000018102 proteins Nutrition 0.000 claims description 22
- 239000004310 lactic acid Substances 0.000 claims description 14
- 235000014655 lactic acid Nutrition 0.000 claims description 14
- 241000894006 Bacteria Species 0.000 claims description 13
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 claims description 12
- 229920002774 Maltodextrin Polymers 0.000 claims description 11
- 239000005913 Maltodextrin Substances 0.000 claims description 10
- 229940035034 maltodextrin Drugs 0.000 claims description 10
- 244000269722 Thea sinensis Species 0.000 claims description 8
- 108010024636 Glutathione Proteins 0.000 claims description 6
- ODKSFYDXXFIFQN-BYPYZUCNSA-N L-arginine Chemical compound OC(=O)[C@@H](N)CCCN=C(N)N ODKSFYDXXFIFQN-BYPYZUCNSA-N 0.000 claims description 6
- 229930064664 L-arginine Natural products 0.000 claims description 6
- 235000014852 L-arginine Nutrition 0.000 claims description 6
- 239000009223 Psyllium Substances 0.000 claims description 6
- 229960003180 glutathione Drugs 0.000 claims description 6
- 229940070687 psyllium Drugs 0.000 claims description 6
- 239000010903 husk Substances 0.000 claims description 4
- 241000593508 Garcinia Species 0.000 claims 1
- 235000000885 Garcinia xanthochymus Nutrition 0.000 claims 1
- 210000003734 kidney Anatomy 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 24
- 235000012054 meals Nutrition 0.000 abstract description 9
- 235000015097 nutrients Nutrition 0.000 abstract description 8
- 210000000577 adipose tissue Anatomy 0.000 abstract description 6
- 210000001789 adipocyte Anatomy 0.000 abstract description 5
- 230000004069 differentiation Effects 0.000 abstract description 5
- 230000035755 proliferation Effects 0.000 abstract description 5
- 241001627231 Garcinia gummi-gutta Species 0.000 abstract 1
- 244000045930 Phaseolus coccineus Species 0.000 abstract 1
- 230000011759 adipose tissue development Effects 0.000 abstract 1
- 235000008590 ayocote Nutrition 0.000 abstract 1
- 239000000843 powder Substances 0.000 description 31
- 208000008589 Obesity Diseases 0.000 description 11
- 235000020824 obesity Nutrition 0.000 description 11
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 10
- 239000003925 fat Substances 0.000 description 10
- 239000000796 flavoring agent Substances 0.000 description 10
- 235000000346 sugar Nutrition 0.000 description 10
- 235000013355 food flavoring agent Nutrition 0.000 description 9
- 235000002789 Panax ginseng Nutrition 0.000 description 8
- 210000004369 blood Anatomy 0.000 description 8
- 239000008280 blood Substances 0.000 description 8
- 238000010521 absorption reaction Methods 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- 230000000996 additive effect Effects 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 230000001965 increasing effect Effects 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 206010012735 Diarrhoea Diseases 0.000 description 5
- 150000001720 carbohydrates Chemical class 0.000 description 5
- 235000014633 carbohydrates Nutrition 0.000 description 5
- -1 etc.) Substances 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- 235000013616 tea Nutrition 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 208000016261 weight loss Diseases 0.000 description 5
- 230000004580 weight loss Effects 0.000 description 5
- 206010010774 Constipation Diseases 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- 240000004371 Panax ginseng Species 0.000 description 4
- 235000005035 Panax pseudoginseng ssp. pseudoginseng Nutrition 0.000 description 4
- 235000003140 Panax quinquefolius Nutrition 0.000 description 4
- 235000001014 amino acid Nutrition 0.000 description 4
- 150000001413 amino acids Chemical class 0.000 description 4
- 230000036528 appetite Effects 0.000 description 4
- 235000019789 appetite Nutrition 0.000 description 4
- 235000012000 cholesterol Nutrition 0.000 description 4
- 206010061428 decreased appetite Diseases 0.000 description 4
- 235000013325 dietary fiber Nutrition 0.000 description 4
- 230000037149 energy metabolism Effects 0.000 description 4
- 235000003599 food sweetener Nutrition 0.000 description 4
- 235000008434 ginseng Nutrition 0.000 description 4
- 150000004668 long chain fatty acids Chemical class 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 210000003470 mitochondria Anatomy 0.000 description 4
- 230000003880 negative regulation of appetite Effects 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- 239000003765 sweetening agent Substances 0.000 description 4
- 229940088594 vitamin Drugs 0.000 description 4
- 229930003231 vitamin Natural products 0.000 description 4
- 235000013343 vitamin Nutrition 0.000 description 4
- 239000011782 vitamin Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 108090000790 Enzymes Proteins 0.000 description 3
- 102000004190 Enzymes Human genes 0.000 description 3
- 206010020772 Hypertension Diseases 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 235000014113 dietary fatty acids Nutrition 0.000 description 3
- 229940088598 enzyme Drugs 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 229930195729 fatty acid Natural products 0.000 description 3
- 239000000194 fatty acid Substances 0.000 description 3
- 150000004665 fatty acids Chemical class 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 210000000936 intestine Anatomy 0.000 description 3
- 230000004060 metabolic process Effects 0.000 description 3
- 230000002438 mitochondrial effect Effects 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- 102000013142 Amylases Human genes 0.000 description 2
- 108010065511 Amylases Proteins 0.000 description 2
- 206010003210 Arteriosclerosis Diseases 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- 208000010201 Exanthema Diseases 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 244000068988 Glycine max Species 0.000 description 2
- 235000010469 Glycine max Nutrition 0.000 description 2
- 229920002527 Glycogen Polymers 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 206010033307 Overweight Diseases 0.000 description 2
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 2
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 2
- 206010047700 Vomiting Diseases 0.000 description 2
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 2
- 102000004139 alpha-Amylases Human genes 0.000 description 2
- 108090000637 alpha-Amylases Proteins 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 208000011775 arteriosclerosis disease Diseases 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 230000000975 bioactive effect Effects 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 235000021152 breakfast Nutrition 0.000 description 2
- ADRVNXBAWSRFAJ-UHFFFAOYSA-N catechin Natural products OC1Cc2cc(O)cc(O)c2OC1c3ccc(O)c(O)c3 ADRVNXBAWSRFAJ-UHFFFAOYSA-N 0.000 description 2
- 235000005487 catechin Nutrition 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 229960001231 choline Drugs 0.000 description 2
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 239000010941 cobalt Substances 0.000 description 2
- 229910017052 cobalt Inorganic materials 0.000 description 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 230000001276 controlling effect Effects 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 238000000502 dialysis Methods 0.000 description 2
- 230000029087 digestion Effects 0.000 description 2
- 230000001882 diuretic effect Effects 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- XMOCLSLCDHWDHP-IUODEOHRSA-N epi-Gallocatechin Chemical compound C1([C@H]2OC3=CC(O)=CC(O)=C3C[C@H]2O)=CC(O)=C(O)C(O)=C1 XMOCLSLCDHWDHP-IUODEOHRSA-N 0.000 description 2
- 201000005884 exanthem Diseases 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- OVBPIULPVIDEAO-LBPRGKRZSA-N folic acid Chemical compound C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-LBPRGKRZSA-N 0.000 description 2
- 235000021588 free fatty acids Nutrition 0.000 description 2
- 238000004108 freeze drying Methods 0.000 description 2
- 229910052732 germanium Inorganic materials 0.000 description 2
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 229940096919 glycogen Drugs 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 230000003871 intestinal function Effects 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 235000021332 kidney beans Nutrition 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 210000003205 muscle Anatomy 0.000 description 2
- 235000021096 natural sweeteners Nutrition 0.000 description 2
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 2
- 235000019629 palatability Nutrition 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 239000011574 phosphorus Substances 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 230000002335 preservative effect Effects 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 238000000751 protein extraction Methods 0.000 description 2
- 206010037844 rash Diseases 0.000 description 2
- 210000000813 small intestine Anatomy 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 230000035882 stress Effects 0.000 description 2
- XOAAWQZATWQOTB-UHFFFAOYSA-N taurine Chemical compound NCCS(O)(=O)=O XOAAWQZATWQOTB-UHFFFAOYSA-N 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 230000032258 transport Effects 0.000 description 2
- 230000007723 transport mechanism Effects 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 2
- 230000008673 vomiting Effects 0.000 description 2
- PFTAWBLQPZVEMU-DZGCQCFKSA-N (+)-catechin Chemical compound C1([C@H]2OC3=CC(O)=CC(O)=C3C[C@@H]2O)=CC=C(O)C(O)=C1 PFTAWBLQPZVEMU-DZGCQCFKSA-N 0.000 description 1
- PFTAWBLQPZVEMU-ZFWWWQNUSA-N (+)-epicatechin Natural products C1([C@@H]2OC3=CC(O)=CC(O)=C3C[C@@H]2O)=CC=C(O)C(O)=C1 PFTAWBLQPZVEMU-ZFWWWQNUSA-N 0.000 description 1
- PFTAWBLQPZVEMU-UKRRQHHQSA-N (-)-epicatechin Chemical compound C1([C@H]2OC3=CC(O)=CC(O)=C3C[C@H]2O)=CC=C(O)C(O)=C1 PFTAWBLQPZVEMU-UKRRQHHQSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 description 1
- CHHHXKFHOYLYRE-UHFFFAOYSA-M 2,4-Hexadienoic acid, potassium salt (1:1), (2E,4E)- Chemical compound [K+].CC=CC=CC([O-])=O CHHHXKFHOYLYRE-UHFFFAOYSA-M 0.000 description 1
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 1
- 208000004998 Abdominal Pain Diseases 0.000 description 1
- 206010000087 Abdominal pain upper Diseases 0.000 description 1
- UCTWMZQNUQWSLP-UHFFFAOYSA-N Adrenaline Natural products CNCC(O)C1=CC=C(O)C(O)=C1 UCTWMZQNUQWSLP-UHFFFAOYSA-N 0.000 description 1
- 244000144927 Aloe barbadensis Species 0.000 description 1
- 235000002961 Aloe barbadensis Nutrition 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 244000144730 Amygdalus persica Species 0.000 description 1
- 239000004382 Amylase Substances 0.000 description 1
- 208000019901 Anxiety disease Diseases 0.000 description 1
- 241000512259 Ascophyllum nodosum Species 0.000 description 1
- 244000063299 Bacillus subtilis Species 0.000 description 1
- 108700029181 Bacteria lipase activator Proteins 0.000 description 1
- 235000017166 Bambusa arundinacea Nutrition 0.000 description 1
- 235000017491 Bambusa tulda Nutrition 0.000 description 1
- 208000035985 Body Odor Diseases 0.000 description 1
- 208000031648 Body Weight Changes Diseases 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 206010006326 Breath odour Diseases 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000238366 Cephalopoda Species 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- GHOKWGTUZJEAQD-UHFFFAOYSA-N Chick antidermatitis factor Natural products OCC(C)(C)C(O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 235000007516 Chrysanthemum Nutrition 0.000 description 1
- 244000189548 Chrysanthemum x morifolium Species 0.000 description 1
- 244000223760 Cinnamomum zeylanicum Species 0.000 description 1
- 235000005979 Citrus limon Nutrition 0.000 description 1
- 244000131522 Citrus pyriformis Species 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 241000238424 Crustacea Species 0.000 description 1
- AUNGANRZJHBGPY-UHFFFAOYSA-N D-Lyxoflavin Natural products OCC(O)C(O)C(O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-UHFFFAOYSA-N 0.000 description 1
- 241000238557 Decapoda Species 0.000 description 1
- 206010014733 Endometrial cancer Diseases 0.000 description 1
- 206010014759 Endometrial neoplasm Diseases 0.000 description 1
- 208000004930 Fatty Liver Diseases 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- 240000009088 Fragaria x ananassa Species 0.000 description 1
- 235000011201 Ginkgo Nutrition 0.000 description 1
- 244000194101 Ginkgo biloba Species 0.000 description 1
- 235000008100 Ginkgo biloba Nutrition 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 102000006587 Glutathione peroxidase Human genes 0.000 description 1
- 108700016172 Glutathione peroxidases Proteins 0.000 description 1
- 102000005720 Glutathione transferase Human genes 0.000 description 1
- 108010070675 Glutathione transferase Proteins 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 201000005569 Gout Diseases 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 206010019708 Hepatic steatosis Diseases 0.000 description 1
- SQUHHTBVTRBESD-UHFFFAOYSA-N Hexa-Ac-myo-Inositol Natural products CC(=O)OC1C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C1OC(C)=O SQUHHTBVTRBESD-UHFFFAOYSA-N 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 235000010254 Jasminum officinale Nutrition 0.000 description 1
- 240000005385 Jasminum sambac Species 0.000 description 1
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 1
- XMOCLSLCDHWDHP-UHFFFAOYSA-N L-Epigallocatechin Natural products OC1CC2=C(O)C=C(O)C=C2OC1C1=CC(O)=C(O)C(O)=C1 XMOCLSLCDHWDHP-UHFFFAOYSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- 108010028554 LDL Cholesterol Proteins 0.000 description 1
- 238000008214 LDL Cholesterol Methods 0.000 description 1
- 244000199866 Lactobacillus casei Species 0.000 description 1
- 235000013958 Lactobacillus casei Nutrition 0.000 description 1
- 240000006024 Lactobacillus plantarum Species 0.000 description 1
- 235000013965 Lactobacillus plantarum Nutrition 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 244000070406 Malus silvestris Species 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- 229920000881 Modified starch Polymers 0.000 description 1
- 239000004368 Modified starch Substances 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- OVBPIULPVIDEAO-UHFFFAOYSA-N N-Pteroyl-L-glutaminsaeure Natural products C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-UHFFFAOYSA-N 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- SKPPEIDJGJGRGK-UHFFFAOYSA-N Panacen Natural products CCC1=CC=CC2=C1C1OC(C=C=CBr)CC1O2 SKPPEIDJGJGRGK-UHFFFAOYSA-N 0.000 description 1
- 206010033664 Panic attack Diseases 0.000 description 1
- 235000004347 Perilla Nutrition 0.000 description 1
- 244000124853 Perilla frutescens Species 0.000 description 1
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 1
- 244000082204 Phyllostachys viridis Species 0.000 description 1
- 235000015334 Phyllostachys viridis Nutrition 0.000 description 1
- 241001127637 Plantago Species 0.000 description 1
- 208000004880 Polyuria Diseases 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 235000006040 Prunus persica var persica Nutrition 0.000 description 1
- MEFKEPWMEQBLKI-AIRLBKTGSA-O S-adenosyl-L-methionine Chemical compound O[C@@H]1[C@H](O)[C@@H](C[S+](CC[C@H]([NH3+])C([O-])=O)C)O[C@H]1N1C2=NC=NC(N)=C2N=C1 MEFKEPWMEQBLKI-AIRLBKTGSA-O 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 206010040904 Skin odour abnormal Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 239000004283 Sodium sorbate Substances 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- JZRWCGZRTZMZEH-UHFFFAOYSA-N Thiamine Natural products CC1=C(CCO)SC=[N+]1CC1=CN=C(C)N=C1N JZRWCGZRTZMZEH-UHFFFAOYSA-N 0.000 description 1
- 208000037063 Thinness Diseases 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 241000219094 Vitaceae Species 0.000 description 1
- MECHNRXZTMCUDQ-UHFFFAOYSA-N Vitamin D2 Natural products C1CCC2(C)C(C(C)C=CC(C)C(C)C)CCC2C1=CC=C1CC(O)CCC1=C MECHNRXZTMCUDQ-UHFFFAOYSA-N 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- PSUBPFRWJHQJIA-UHFFFAOYSA-K [Ca+2].C(C=CC=CC)(=O)[O-].[Na+].C(C=CC=CC)(=O)[O-].C(C=CC=CC)(=O)[O-] Chemical compound [Ca+2].C(C=CC=CC)(=O)[O-].[Na+].C(C=CC=CC)(=O)[O-].C(C=CC=CC)(=O)[O-] PSUBPFRWJHQJIA-UHFFFAOYSA-K 0.000 description 1
- 208000019790 abdominal distention Diseases 0.000 description 1
- 230000035508 accumulation Effects 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 235000011399 aloe vera Nutrition 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- PECIYKGSSMCNHN-UHFFFAOYSA-N aminophylline Chemical compound NCCN.O=C1N(C)C(=O)N(C)C2=NC=N[C]21.O=C1N(C)C(=O)N(C)C2=NC=N[C]21 PECIYKGSSMCNHN-UHFFFAOYSA-N 0.000 description 1
- 229960003556 aminophylline Drugs 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 235000019418 amylase Nutrition 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 230000000259 anti-tumor effect Effects 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 230000036506 anxiety Effects 0.000 description 1
- 239000002830 appetite depressant Substances 0.000 description 1
- 235000021016 apples Nutrition 0.000 description 1
- 229910052785 arsenic Inorganic materials 0.000 description 1
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 239000011425 bamboo Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 239000003613 bile acid Substances 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- 230000004579 body weight change Effects 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 230000003925 brain function Effects 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 239000004301 calcium benzoate Substances 0.000 description 1
- 235000010237 calcium benzoate Nutrition 0.000 description 1
- HZQXCUSDXIKLGS-UHFFFAOYSA-L calcium;dibenzoate;trihydrate Chemical compound O.O.O.[Ca+2].[O-]C(=O)C1=CC=CC=C1.[O-]C(=O)C1=CC=CC=C1 HZQXCUSDXIKLGS-UHFFFAOYSA-L 0.000 description 1
- 235000019577 caloric intake Nutrition 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 150000001765 catechin Chemical class 0.000 description 1
- 230000032677 cell aging Effects 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 201000001883 cholelithiasis Diseases 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 229950001002 cianidanol Drugs 0.000 description 1
- 235000017803 cinnamon Nutrition 0.000 description 1
- 235000020971 citrus fruits Nutrition 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- GVJHHUAWPYXKBD-UHFFFAOYSA-N d-alpha-tocopherol Natural products OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 230000001079 digestive effect Effects 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 230000002500 effect on skin Effects 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- LPTRNLNOHUVQMS-UHFFFAOYSA-N epicatechin Natural products Cc1cc(O)cc2OC(C(O)Cc12)c1ccc(O)c(O)c1 LPTRNLNOHUVQMS-UHFFFAOYSA-N 0.000 description 1
- 235000012734 epicatechin Nutrition 0.000 description 1
- DZYNKLUGCOSVKS-UHFFFAOYSA-N epigallocatechin Natural products OC1Cc2cc(O)cc(O)c2OC1c3cc(O)c(O)c(O)c3 DZYNKLUGCOSVKS-UHFFFAOYSA-N 0.000 description 1
- 229960002061 ergocalciferol Drugs 0.000 description 1
- 239000003797 essential amino acid Substances 0.000 description 1
- 235000020776 essential amino acid Nutrition 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 235000005523 excessive nutrition Nutrition 0.000 description 1
- 230000002964 excitative effect Effects 0.000 description 1
- 206010016256 fatigue Diseases 0.000 description 1
- 230000004129 fatty acid metabolism Effects 0.000 description 1
- 208000010706 fatty liver disease Diseases 0.000 description 1
- 229930003935 flavonoid Natural products 0.000 description 1
- 235000017173 flavonoids Nutrition 0.000 description 1
- 150000002215 flavonoids Chemical class 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 235000019152 folic acid Nutrition 0.000 description 1
- 229960000304 folic acid Drugs 0.000 description 1
- 239000011724 folic acid Substances 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 229960002442 glucosamine Drugs 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 229930182470 glycoside Natural products 0.000 description 1
- 150000002338 glycosides Chemical class 0.000 description 1
- 235000021021 grapes Nutrition 0.000 description 1
- 235000009569 green tea Nutrition 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 230000004217 heart function Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 241000411851 herbal medicine Species 0.000 description 1
- 235000008216 herbs Nutrition 0.000 description 1
- 238000000265 homogenisation Methods 0.000 description 1
- 235000012907 honey Nutrition 0.000 description 1
- 230000000887 hydrating effect Effects 0.000 description 1
- 150000002432 hydroperoxides Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 210000003016 hypothalamus Anatomy 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 229960000367 inositol Drugs 0.000 description 1
- CDAISMWEOUEBRE-GPIVLXJGSA-N inositol Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@@H]1O CDAISMWEOUEBRE-GPIVLXJGSA-N 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 229920000831 ionic polymer Polymers 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 229940017800 lactobacillus casei Drugs 0.000 description 1
- 229940072205 lactobacillus plantarum Drugs 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 230000006372 lipid accumulation Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 235000014666 liquid concentrate Nutrition 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 238000012792 lyophilization process Methods 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- 239000011859 microparticle Substances 0.000 description 1
- 235000019426 modified starch Nutrition 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 235000001968 nicotinic acid Nutrition 0.000 description 1
- 229960003512 nicotinic acid Drugs 0.000 description 1
- 239000011664 nicotinic acid Substances 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 208000019906 panic disease Diseases 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 229940055726 pantothenic acid Drugs 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 229920001197 polyacetylene Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 239000004300 potassium benzoate Substances 0.000 description 1
- 235000010235 potassium benzoate Nutrition 0.000 description 1
- 229940103091 potassium benzoate Drugs 0.000 description 1
- 235000010241 potassium sorbate Nutrition 0.000 description 1
- 239000004302 potassium sorbate Substances 0.000 description 1
- 229940069338 potassium sorbate Drugs 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 235000020944 retinol Nutrition 0.000 description 1
- 229960003471 retinol Drugs 0.000 description 1
- 239000011607 retinol Substances 0.000 description 1
- 235000019192 riboflavin Nutrition 0.000 description 1
- 229960002477 riboflavin Drugs 0.000 description 1
- 239000002151 riboflavin Substances 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 235000017709 saponins Nutrition 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- CDAISMWEOUEBRE-UHFFFAOYSA-N scyllo-inosotol Natural products OC1C(O)C(O)C(O)C(O)C1O CDAISMWEOUEBRE-UHFFFAOYSA-N 0.000 description 1
- 239000000932 sedative agent Substances 0.000 description 1
- 230000001624 sedative effect Effects 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 231100000046 skin rash Toxicity 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- 229960003885 sodium benzoate Drugs 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- LROWVYNUWKVTCU-STWYSWDKSA-M sodium sorbate Chemical compound [Na+].C\C=C\C=C\C([O-])=O LROWVYNUWKVTCU-STWYSWDKSA-M 0.000 description 1
- 235000019250 sodium sorbate Nutrition 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 230000007103 stamina Effects 0.000 description 1
- 231100000240 steatosis hepatitis Toxicity 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 235000021012 strawberries Nutrition 0.000 description 1
- 238000005728 strengthening Methods 0.000 description 1
- 239000002438 stress hormone Substances 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 230000000576 supplementary effect Effects 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 235000018553 tannin Nutrition 0.000 description 1
- 229920001864 tannin Polymers 0.000 description 1
- 239000001648 tannin Substances 0.000 description 1
- 229960003080 taurine Drugs 0.000 description 1
- 150000003505 terpenes Chemical class 0.000 description 1
- 235000007586 terpenes Nutrition 0.000 description 1
- 235000019157 thiamine Nutrition 0.000 description 1
- 229960003495 thiamine Drugs 0.000 description 1
- KYMBYSLLVAOCFI-UHFFFAOYSA-N thiamine Chemical compound CC1=C(CCO)SCN1CC1=CN=C(C)N=C1N KYMBYSLLVAOCFI-UHFFFAOYSA-N 0.000 description 1
- 239000011721 thiamine Substances 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 229960001295 tocopherol Drugs 0.000 description 1
- 235000010384 tocopherol Nutrition 0.000 description 1
- 229930003799 tocopherol Natural products 0.000 description 1
- 239000011732 tocopherol Substances 0.000 description 1
- 239000011573 trace mineral Substances 0.000 description 1
- 235000013619 trace mineral Nutrition 0.000 description 1
- 206010048828 underweight Diseases 0.000 description 1
- 235000001892 vitamin D2 Nutrition 0.000 description 1
- MECHNRXZTMCUDQ-RKHKHRCZSA-N vitamin D2 Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)/C=C/[C@H](C)C(C)C)=C\C=C1\C[C@@H](O)CCC1=C MECHNRXZTMCUDQ-RKHKHRCZSA-N 0.000 description 1
- 239000011653 vitamin D2 Substances 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 238000004260 weight control Methods 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/16—Inorganic salts, minerals or trace elements
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/17—Amino acids, peptides or proteins
- A23L33/185—Vegetable proteins
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/20—Reducing nutritive value; Dietetic products with reduced nutritive value
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2200/00—Function of food ingredients
- A23V2200/30—Foods, ingredients or supplements having a functional effect on health
- A23V2200/332—Promoters of weight control and weight loss
Abstract
Description
본 발명은 다이어트용 보조식품 조성물에 관한 것으로, 보다 상세하게는 기존의 다이어트 식품들이 갖는 여러 부작용을 억지하고, 지방세포의 증식과 분화를 억제하며, 식사량 부족으로 인한 부족 영양소를 보충할 수 있는 다이어트용 보조식품 조성물에 관한 것이다.The present invention relates to a dietary supplement composition, more specifically, to suppress various side effects of the existing diet foods, suppress the proliferation and differentiation of fat cells, and supplement the nutrients due to lack of food. It relates to an auxiliary food composition.
경제 발전에 따른 생활수준의 향상과, 바쁜 생활환경에 따른 운동 부족, 및 영양의 과잉 섭취 등으로 비만 인구가 급속히 늘고 있다. 우리나라의 비만 인구 비율은 성인 여성의 경우 1995년 11.7%이던 것이 2001년 29.4%, 성인 남성의 경우 1995년 18.0%이던 것이 2001년 32.6%로 빠른 증가세를 보이고 있다.The obesity population is rapidly increasing due to the improvement of living standards due to economic development, lack of exercise due to the busy living environment, and excessive nutrition. The proportion of the obese population in Korea was 11.7% in 1995 for adults, 29.4% in 2001, and 18.0% in 1995 for adults, with a rapid increase of 32.6% in 2001.
비만은 열량의 섭취와 소비의 불균형으로 지방 조직이 비정상적으로 증가된 상태를 말한다. 임상적으로는 BMI(Body Mass Index: 체질량 지수)가 25.0 내지 30.0을 과체중으로 정의하고 30.0 이상인 경우를 비만으로 정의한다.Obesity is a condition in which fat tissue is abnormally increased due to an imbalance in consumption and consumption of calories. Clinically, a body mass index (BMI) of 25.0 to 30.0 is defined as overweight, and a case of 30.0 or more is defined as obesity.
비만은 지속되면 고혈압, 동맥경화, 당뇨, 지방간, 담석증, 통풍 등의 질환의 원인으로 작용하며, 특히 비만 여성에게서는 자궁내막암, 자궁경부암, 난소암, 유방암 등이 더 흔히 발생한다고 보고가 있으며, 또한 사망률에 있어서도 비만자는 정상 체중자들 보다 1.3배 이상 더 높은 것으로 보고되어 있다.If obesity persists, hypertension, arteriosclerosis, diabetes, fatty liver, cholelithiasis, gout act as a cause of diseases, especially in obese women, endometrial cancer, cervical cancer, ovarian cancer, breast cancer, etc. are reported to occur more frequently, In addition, obesity is also reported to be 1.3 times higher than that of normal weight.
비만 치료제는 일반적으로 3가지의 범주 즉 식욕 억제제, 체내 에너지 대사 촉진제 및 소화 흡수 억제제로 구분된다.Treatment for obesity is generally divided into three categories: appetite suppressants, energy metabolism accelerators in the body, and digestive absorption inhibitors.
이중 식욕을 억제하는 식사대용 식품조성물로는 특허 제10-0439094호 "복령을 이용한 다이어트 식품"이 공지된 바가 있다. 이러한 다이어트 식품은 탄수화물을 중심으로 하는 주식품군과 식욕억제기능이 있는 제2구성분으로 구성된 것이다. 그러나, 이러한 다이어트 식품은 식사대용으로 섭취하는 식품이기 때문에 정상적인 식사를 금하게 하여, 식사 모임에의 참석 등이 불가능하므로 정상적인 사회활동을 방해할 수 있다는 문제점이 있었다. Among them, as a food composition for appetite suppressing appetite, Patent No. 10-0439094, "Diet Food Using Pueryeong," has been known. These diet foods consist of a group of stocks, mainly carbohydrates, and a second component with appetite suppression. However, since these diet foods are foods that are consumed as meal substitutes, they are prohibited from eating normally, and thus, it is impossible to attend a meal meeting, and thus there is a problem that they can interfere with normal social activities.
또한, 특허 제10-0602281호 "인삼가공편이 포함된 선식의 조성물 및 이를 이용한 다이어트 선식 조성물"은 인삼가공편을 주원료로 하고 진피분말 등 비만억제 및 치료 작용을 하는 소재를 첨가한 것으로, 이 또한 식사대용품으로 사용할 경우 전술한 문제점을 같은 문제점을 가지게 된다. 그리고 이를 식사량 조절을 위한 보조식품으로 사용하는 경우에는 필요한 영양소의 보충 작용이 충분치 못하다는 문제점도 있다. In addition, Patent No. 10-0602281, "a composition of a wire containing a ginseng processed piece and a diet wire composition using the same" is a ginseng processed piece as a main raw material and is added with a material that suppresses and treats obesity such as dermal powder. When used as a meal replacement, the above-described problems have the same problems. In addition, when it is used as an auxiliary food for controlling the amount of food, there is a problem that the supplementary action of necessary nutrients is insufficient.
한편, 비만을 예방하거나 치료하는 약리 작용을 가지는 식품을 첨가하여서 되는 다이어트 식품으로는 특허 제10-0448260호 "다시마 및 한방 생약재를 이용한 체중 조절용 영양식품 및 제조방법"이 공지된 바 있다. 이는 다시마 및 한방 생약재를 일부는 분말 상태로, 일부는 추출액으로 하여 혼합된 식품이나, 그 제조 방법이 복잡하다는 문제점이 있다.On the other hand, as a diet food by adding a food that has a pharmacological action to prevent or treat obesity, Patent No. 10-0448260, "Tashima and Herbal Herbs for Nutritional Food and Method for Weight Control" has been known. This is a part of the kelp and herbal herbal medicines in powder form, and some of them are mixed foods, but there is a problem in that the manufacturing method is complicated.
또한, 특허 제10-0853940호 "다시마, 함초, 톳, 콩의 조성물에 바실러스 서브틸리스균을 접종하여 발효시킨 다이어트 조성물"이 공지된 바가 있으나, 이는 식사량 부족에서 오는 부족 영양소의 보충하기 어렵다는 문제점이 있다. In addition, there is a known patent No. 10-0853940, "a diet composition fermented by inoculating Bacillus subtilis bacteria in a composition of Dashima, Hamcho, Soybean, and Soybean," but this is a problem that it is difficult to supplement insufficient nutrients coming from a lack of food. have.
따라서, 본 발명의 목적은 영향학적으로 균형된 식사 생활을 영위할 수 있도록 도와주고, 이뇨작용에 따른 수분감소로 인한 급격한 체중감소, 식욕억제, 근력감소, 요요현상의 발생을 억제하며, 지방세포의 증식과 분화를 억제하여 체지방을 감소시킬 수 있는 다이어트용 보조식품 조성물을 제공하는데 있다. Accordingly, the object of the present invention is to help lead an impactfully balanced eating life, and to suppress the occurrence of rapid weight loss, appetite suppression, muscle strength reduction, yo-yo phenomenon due to water loss due to diuretic action, fat cells It is to provide a dietary supplement composition that can reduce the body fat by inhibiting the proliferation and differentiation of.
상술한 본 발명의 목적을 달성하기 위하여, 본 발명의 일 실시예에서는 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘을 포함하는 다이어트용 보조식품 조성물을 제공한다.In order to achieve the object of the present invention described above, in one embodiment of the present invention provides a dietary supplement composition comprising a tea bean, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium.
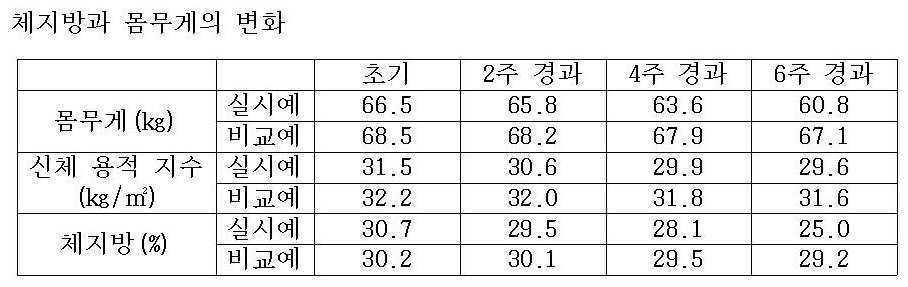
본 발명에 의한 다이어트용 보조식품 조성물은 식사 전후로 1포씩 복용함으로서, 식사를 통해 영양분을 섭취하더라도 지방생성 뿐만 아니라 지방세포의 증식과 분화를 억제함으로써 체지방을 감소시킬 수 있다.The dietary supplement composition for diet according to the present invention can reduce body fat by suppressing proliferation and differentiation of fat cells as well as fat production, even by taking one packet before and after a meal.
또한, 본 발명은 이뇨작용이나 식욕억제 효능이 있는 소재를 이용하지 않고 천연재료 유래 성분을 사용함으로서 기존의 다이어트 식품들이 갖는 여러 부작용의 발생을 방지하며, 일상의 식생활을 즐길 수 있도록 하면서도 몸무게를 감소시킬 수 있다. In addition, the present invention prevents the occurrence of various side effects of the existing diet foods by using ingredients derived from natural ingredients without using a material having a diuretic effect or an appetite suppression effect, and reduces the weight while allowing you to enjoy a daily diet. I can do it.
아울러, 본 발명은 섭취자에게 풍부한 영양소를 공급하여 식사량 부족으로 인한 영양소 부족 염려를 해소시킬 수 있다.In addition, the present invention can solve the concern of insufficient nutrients due to insufficient food supply by supplying rich nutrients to the intake.
이하, 본 발명의 바람직한 실시예들에 의한 다이어트용 보조식품 조성물을 상세하게 설명한다. Hereinafter, a dietary supplement composition according to preferred embodiments of the present invention will be described in detail.
본 발명에 따른 다이어트용 보조식품 조성물은 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아(Garcinia Cambogia) 추출물, 키토산, L-카르니틴, 및 마그네슘을 포함한다.Dietary supplement composition for diet according to the present invention includes chajeonpi, white kidney bean protein extract, Garcinia Cambogia extract, chitosan, L-carnitine, and magnesium.
이러한 다이어트용 보조식품 조성물은 소지가 편하도록 4g 내지 5g씩 소포장하는 것이 바람직하며, 아침과 저녁 식전에 1포로 1일 2회 충분한 물과 함께 섭취한다. These dietary supplement compositions are preferably packaged in small portions of 4g to 5g for ease of possession, and consumed with sufficient water twice a day in one packet before breakfast and dinner.
이하, 각 구성요소별로 보다 구체적으로 설명한다. Hereinafter, each component will be described in more detail.
본 발명에 따른 다이어트용 보조식품 조성물은 차전자피를 포함한다.Dietary supplement composition for diet according to the present invention comprises a tea puff.
상기 차전자피는 식이섬유로, 체중감량 중임에도 불구하고 변비발생을 억제하는 것은 물론 적극적으로 배변활동을 강화하는 역할을 위해 첨가된다. The chamokpi is a dietary fiber, and is added for the role of actively suppressing constipation and strengthening bowel movements despite losing weight.
상기 차전자피는 차전자(Plantago ovata 또는 Plantago spp.)의 껍질을 분쇄하여 식용에 적합하도록 만든 것으로, 차전자피 분말, 바람직하게는 식이섬유를 79% 이상 함유한 차전자피 분말을 사용하는 것이 좋다. The psyllium husk is made to be suitable for edible food by crushing the shell of a psia (Plantago ovata or Plantago spp.), And it is preferable to use a psyllium stalk powder, preferably a psyllium hull powder containing at least 79% dietary fiber.
일반적으로 차전자피는 총 콜레스테롤과 LDL-콜레스테롤을 감소시키는 기능을 제공하나, 본 발명에서는 원활한 배변활동이 일어나도록 유도하는 기능을 제공하기 위해 첨가된다. 구체적으로, 차전자피는 수분을 흡수력이 뛰어나고 이때 놀라울 정도로 부피가 증가하여 장에서 배변활동을 촉진하게 된다. In general, psyllium husk provides a function of reducing total cholesterol and LDL-cholesterol, but is added to provide a function of inducing smooth bowel activity in the present invention. Specifically, the chaerion blood is excellent in absorbing moisture, and at this time, the volume is surprisingly increased to promote bowel movement in the intestine.
이러한 차전자피는 전체 100 중량%를 기준으로 40 내지 50 중량%가 포함된다. 이때, 차전자피는 전체 100 중량%를 기준으로 함량이 40 중량% 미만으로 첨가되는 경우 다이어트로 인해 유발되는 변비의 개선효과가 미미하고, 50 중량%를 초과하면 체질에 따라 설사가 유발되는 문제가 발생될 수 있다. The psyllium husk contains 40 to 50% by weight based on 100% by weight. At this time, when the content is added to less than 40% by weight based on 100% by weight, Chaejeonpi has a slight improvement effect of constipation caused by diet, and when it exceeds 50% by weight, diarrhea may occur depending on the constitution. Can be.
필요에 따라, 상기 차전자피로는 장용성 고분자에 의해 코팅된 차전자피 분말을 사용할 수 있다. 이는, 배변활동 강화 효과를 높이기 위해 차전자피 분말의 흡수가 위가 아닌 장에서 일어나도록 하기 위함이다. If necessary, the electron barrier powder coated with the enteric polymer may be used as the electron barrier blood. This is to ensure that the absorption of the tea powder is caused in the intestine, not the stomach, in order to enhance the effect of enhancing bowel movement.
본 발명에 따른 다이어트용 보조식품 조성물은 흰강낭콩 단백질 추출물을 포함한다.Dietary supplement composition according to the present invention includes a white kidney bean protein extract.
상기 흰강낭콩 단백질 추출물은 소화 효소인 알파아밀라아(α-amylases)의 활성을 저해하여 탄수화물의 흡수를 억제하는 기능을 제공하도록 첨가된 것으로, 전체 100 중량%를 기준으로 15 내지 25 중량%가 포함된다. 이러한 흰강낭콩 단백질 추출물로는 흰강남콩에서 자연친화적으로 추출한 추출물 분말을 사용한다. The white kidney bean protein extract is added to provide the function of inhibiting the absorption of carbohydrates by inhibiting the activity of the digestive enzyme alpha-amylase (α-amylases), 15 to 25% by weight based on 100% by weight Is included. As the white kidney bean protein extract, an extract powder extracted naturally from white kidney beans is used.
또한, 흰강낭콩 단백질 추출물은 전체 100 중량%를 기준으로 함량이 15 중량% 미만으로 첨가되는 경우 첨가효과가 없고, 25 중량%를 초과하면 기호성이 떨어지는 문제가 발생된다.In addition, the white kidney bean protein extract has no additive effect when the content is added to less than 15% by weight based on 100% by weight, and the problem of poor palatability occurs when it exceeds 25% by weight.
한편, 흰강낭콩 단백질 추출물은 액상추출 과정과, 단백질 추출과정과, 원심분리 과정, 및 동결건조 과정을 통해 제조할 수 있다. Meanwhile, the white kidney bean protein extract may be prepared through a liquid extraction process, a protein extraction process, a centrifugation process, and a lyophilization process.
구체적으로, 흰강낭콩 단백질 추출물의 제조는 1% 식염수를 강낭콩 분말 1㎏당 1.5ℓ를 가하여 실온에서 2시간 동안 추출물을 추출하는 액상추출 과정과, 상기 추출물을 원심분리 하여 상등액을 수득하고 상기 상등액에 탄닌 추출물 또는 에탄올을 가하여 침전물을 생성하는 단백질 추출과정과, 상기 침전물을 원심분리 하여 상등액을 농축하는 원심분리 과정, 및 상기 상등액을 투석막으로 투석한 후 동결건조 하는 동결건조 과정으로 이루어진다. Specifically, the production of the white kidney bean protein extract is a liquid extraction process of extracting the extract for 2 hours at room temperature by adding 1.5 l of 1% saline to 1 kg of kidney bean powder, and centrifuging the extract to obtain a supernatant and the supernatant. It consists of a protein extraction process to produce a precipitate by adding tannin extract or ethanol, a centrifugation process to concentrate the supernatant by centrifuging the precipitate, and a freeze-drying process to lyophilize the supernatant after dialysis with a dialysis membrane.
본 발명에 따른 다이어트용 보조식품 조성물은 가르시니아 캄보지아 추출물을 포함한다.Dietary supplement composition according to the present invention includes a garcinia cambogia extract.
상기 가르시니아 캄보지아 추출물은 지방산과 콜레스테롤 합성에 관여하는 효소의 저해제인 하이드록실 시트르산(Hydroxycitric Acid : HCA)을 함유하여 지방 합성을 저해, 식욕 억제 효과와 지방의 분해 촉진을 유도하는 기능을 제공하는 것으로, 전체 100 중량%를 기준으로 20 내지 30 중량%가 포함된다. 이러한 가르시니아 캄보지아 추출물로는 흰강남콩에서 자연친화적으로 추출한 추출물 분말을 사용한다. The garcinia cambogia extract contains hydroxyl citric acid (HCA), an inhibitor of enzymes involved in the synthesis of fatty acids and cholesterol, to inhibit fat synthesis, to suppress appetite, and to provide a function to induce fat decomposition. 20 to 30% by weight based on the total 100% by weight is included. As the Garcinia cambogia extract, an extract powder extracted naturally from white kidney beans is used.
또한, 가르시니아 캄보지아 추출물 분말은 곡류 분말 100 중량%를 기준으로 함량이 20 중량% 미만으로 첨가되는 경우 가르시니아 캄보지아 추출물 분말에 의한 효능과 효과가 미약하고, 30 중량%를 초과하면 피로감, 피부 발진, 두통, 구취, 소화 장애 등의 문제가 발생될 수 있다.In addition, when the content of the garcinia cambogia extract powder is less than 20% by weight based on 100% by weight of the grain powder, the efficacy and effect by the garcinia cambogia extract powder is weak, and when it exceeds 30% by weight, fatigue, skin rash, headache , Bad breath, problems with digestion, etc. may occur.
이러한 가르시니아 캄보지아 추출물 분말은 과잉 당질이 비장으로 합성되는 경로를 차단, 지방 합성을 방해하는 요인으로 작용하여 신체 내 에너지 생산이 길어져 총에너지 생산량을 증대시킨다. 특히 가르시니아 캄보지아 추출물 분말은 인체 내에서 아세틸조효소에이(acetyl-coenzymeA)를 감소시키므로, 결과적으로 간장과 글리코겐 합성 속도를 증가시킨다. The garcinia cambogia extract powder blocks the pathway of excess sugar synthesis into the spleen and acts as a factor that interferes with fat synthesis, thus increasing the energy production in the body and increasing the total energy production. In particular, Garcinia cambogia extract powder reduces acetyl-coenzymeA in the human body, resulting in increased hepatic and glycogen synthesis rate.
이에 의해, 과량의 글리코겐이 축적되면 뇌의 시상하부에 신체가 필요로 하는 에너지가 충분히 축적되어 있음을 인지하므로, 섭취자는 더 이상의 에너지 섭취를 줄이게 된다. 즉, 본 발명에 따른 다이어트용 보조식품 조성물의 섭취자는 자연히 식욕이 억제되어 체중 감소가 발생된다.As a result, when the excess amount of glycogen is accumulated, it is recognized that the energy required by the body is sufficiently accumulated in the hypothalamus of the brain, thereby reducing the intake of energy. That is, the intake of the dietary supplement composition according to the present invention naturally suppresses appetite, resulting in weight loss.
본 발명에 따른 다이어트용 보조식품 조성물은 키토산을 포함한다.Dietary supplement composition according to the present invention comprises chitosan.
상기 키토산은 새우, 게, 곤충의 껍질, 오징어 뼈 등 갑각류에 많이 함유되어 있는 성분을 추출, 정제한 것으로 화학 구조는 천연물 중에서 유일한 플러스 이온을 갖는 글루코사민이 수천개의 고리로 연결되어 있는 거대한 고분자 물질로 구성되어 있다.The chitosan is a large polymer material in which glucosamine, which is the only positive ion among natural products, is connected by thousands of rings, by extracting and purifying components contained in crustaceans such as shrimp, crab, insect shells, and squid bones. Consists of.
그리고 플러스(+) 이온 상태인 고분자 키토산은 혈압 상승 원인의 하나인 염소나 콜레스테롤, 담즙산 등의 마이너스(-) 이온과 중금속 등을 흡착하여 체외로 배출하고, 인지질과 결합하여 지방의 흡수를 억제하는 기능을 갖고 있으며, 소화 촉진과 알콜 대사 촉진 및 암세포 억제 등 생체 조절 기능도 제공한다. In addition, positive (+) ionic polymer chitosan adsorbs negative (-) ions such as chlorine, cholesterol, and bile acids, which are one of the causes of increased blood pressure, and discharges them to the body, and combines them with phospholipids to suppress fat absorption. It has a function and provides bio-regulation functions such as promoting digestion, promoting alcohol metabolism, and suppressing cancer cells.
특히, 비만과 관계되는 효능으로는 고분자 상태의 키토산 분자가 과잉의 지방과 콜레스테롤 등을 흡착 배설시키므로 칼로리 섭취를 줄여 주고, 탄수화물도 흡착하여 서서히 방출함으로써 세포의 완전 연소를 유도하여 비만 해소 및 혈당 상승을 억제하는 효과를 제공한다. Particularly, as an effect related to obesity, high molecular weight chitosan molecules adsorb and excrete excess fat and cholesterol, thereby reducing calorie intake and absorbing carbohydrates, gradually releasing them to induce complete combustion of cells to relieve obesity and increase blood sugar. It provides the effect of suppressing.
이러한 키토산은 전체 100 중량%를 기준으로 3 내지 10 중량%가 포함된다. 이때, 키토산은 전체 100 중량%를 기준으로 함량이 3 중량% 미만으로 첨가되는 경우 첨가효과가 없고, 10 중량%를 초과하면 구토나, 설사, 복부 팽창 등 위장 관련 문제가 발생될 수 있다. The chitosan is 3 to 10% by weight based on 100% by weight. At this time, chitosan has no additive effect when the content is added in an amount of less than 3% by weight based on 100% by weight, and when it exceeds 10% by weight, gastrointestinal problems such as vomiting, diarrhea, and abdominal distention may occur.
상기 키토산으로는 키토산 분말을 사용할 수 있으며, 선택적으로는 키토산 미립자 분말을 사용할 수 있다.As the chitosan, chitosan powder may be used, and optionally, chitosan particulate powder may be used.
구체적으로, 2 내지 50㎛ 키토산 미립자 분말의 제조는 키토산을 1 내지 5중량%의 초산 또는 염산용액에 키토산을 0.5 내지 10중량%로 용해시키고 1000rpm 내지 1500rpm으로 교반하면서, 여기에 초산 또는 염산용액의 당량비의 수산화나트륨, 수산화칼륨, 암모니아수 등의 용액으로 pH 6.8 내지 7.5로 중화시켜 겔화 시키며, 이 겔 용액을 충분히 세척하여 염을 제거한 다음 동결 건조 또는 스프레이 건조하는 과정으로 이루어진다. Specifically, in the preparation of 2 to 50 μm chitosan particulate powder, chitosan is dissolved in 1 to 5% by weight of acetic acid or hydrochloric acid solution at 0.5 to 10% by weight of chitosan, and stirred at 1000 rpm to 1500 rpm. Equivalent ratio of sodium hydroxide, potassium hydroxide, ammonia water and the like is neutralized to pH 6.8 to 7.5 to gel, and the gel solution is washed sufficiently to remove salt, followed by freeze drying or spray drying.
본 발명에 따른 다이어트용 보조식품 조성물은 L-카르니틴을 포함한다.Dietary supplement composition according to the invention comprises L-carnitine.
상기 L-카르니틴은 β-hydroxy-γ-N-trimethylaminobutyrate의 화학명을 가지며, 구조적으로 콜린(choline)과 비슷한 아미노산 유사 물질이다. 그러나 엄밀하게 말하면 L-카르니틴은 신체 필요량의 일부를 생합성에 의하여 충족할 수 있기 때문에 실질적인 비타민은 아니며, 콜린, 타우린(taurine), 이노시톨(inocitol) 등과 같은 비타민 형태의 영양소라고 말할 수 있다. The L-carnitine has a chemical name of β-hydroxy-γ-N-trimethylaminobutyrate, and is an amino acid-like substance structurally similar to choline. However, strictly speaking, L-carnitine is not a practical vitamin because it can satisfy a part of body needs by biosynthesis, and it can be said that it is a nutrient in the form of vitamins such as choline, taurine, and inositol.
이러한 L-카르니틴의 가장 중요한 생체 기작은 지방산 대사의 필수 보조인자로서 체내에서 지방산을 미토콘드리아 내로 원활히 수송하여 지방을 세포의 에너지로 전환시키는 운반분자의 역할이다. 이러한 역할은 세포 내 지방산 및 당 대사를 개선하게 된다. The most important biomechanical mechanism of L-carnitine is an essential cofactor of fatty acid metabolism, and is a transport molecule that smoothly transports fatty acids into the mitochondria in the body to convert fat into energy of cells. This role improves fatty acid and sugar metabolism in the cell.
특히, 긴 사슬 지방산은 미토콘드리아의 외막에서 활성화되어 미토콘드리아의 기질에서 산화된다. 상기 긴 사슬 지방산은 특별한 운반기구 없이는 미토콘드리아의 내막을 통과할 수 없는데, 이때 운반기구로 작용하는 것이 L-카르니틴이다. 이와 같이, 미토콘드리아 외막에서 활성화된 긴 사슬 지방산은 L-카르니틴의 하이드록시기에 일시적으로 결합하여 긴 사슬 지방산 아실카르니틴(acyl-carnitine)을 형성하며, 미토콘드리아 내로 수송되어 대사된다. In particular, long chain fatty acids are activated in the mitochondrial outer membrane and oxidized in the mitochondrial substrate. The long chain fatty acids cannot pass through the inner membrane of the mitochondria without a special transport mechanism, where L-carnitine acts as a transport mechanism. As such, the long chain fatty acids activated in the mitochondrial outer membrane temporarily bind to the hydroxy group of L-carnitine to form the long chain fatty acid acyl-carnitine, which is transported into the mitochondria and metabolized.
또한, L-카르니틴은 체중감량에 있어 생체의 여러 가지 상태에서 혈중 및 조직 속의 지질 축척을 감소시키는 것이 증명되었으며, 근육이 일을 하는데 있어서 필수적으로 사용되는 에너지를 공급하므로 심 기능의 향상, 산소 흡입량의 증가, 체력유지, 지방 감소 등의 효과를 제공한다.In addition, L-carnitine has been proven to reduce lipid accumulation in the blood and tissues in various states of the body in terms of weight loss, and improves heart function and oxygen intake by supplying energy that is essential for muscle work. Provides effects such as increase, stamina, and fat reduction.
이러한 L-카르니틴은 전체 100 중량%를 기준으로 5 내지 15 중량%가 포함된다. 이때, L-카르니틴은 전체 100 중량%를 기준으로 함량이 5 중량% 미만으로 첨가되는 경우 첨가효과가 없고, 15 중량%를 초과하면 맛과 냄새로 인하여 기호성과 제품성이 저하되는 문제가 발생될 수 있다. The L-carnitine is included in an amount of 5 to 15% by weight based on 100% by weight. In this case, L-carnitine has no additive effect when the content is added to less than 5% by weight based on 100% by weight, and if it exceeds 15% by weight, problems such as palatability and productability may be deteriorated due to taste and odor. You can.
본 발명에 따른 다이어트용 보조식품 조성물은 마그네슘을 포함한다.Dietary supplement composition according to the present invention includes magnesium.
상기 마그네슘은 에너지대사 활성효과로 지방의 미토콘드리아 내의 에너지 대사를 촉진시킨다. 이는 아드레날린 수용체의 활성화로 에너지대사를 활성화시키는 아미노필린의 지방대사 기전과는 다른 것이므로 내성이 생기지 않으며, 사용을 중단하고 난 후에 체중이 반등하는 효과도 적다.The magnesium promotes energy metabolism in the mitochondria of fats with an energy metabolism activity effect. This is different from the fatty metabolism mechanism of aminophylline, which activates energy metabolism by activating the adrenaline receptor, so it does not develop resistance and has little effect in regaining weight after stopping use.
또한, 마그네슘은 스트레스 호르몬과 뇌 기능을 조절해주어 다이어트로의 스트레스로 인한 우울증, 불안, 공황 발작 등의 발생을 완화시켜 준다. In addition, magnesium regulates stress hormones and brain function, thereby reducing the occurrence of depression, anxiety, and panic attacks caused by stress in the diet.
이러한 마그네슘은 전체 100 중량%를 기준으로 5 내지 10 중량%가 포함된다. 이때, 차전자피는 전체 100 중량%를 기준으로 함량이 5 중량% 미만으로 첨가되는 경우 첨가효과가 없고, 10 중량%를 초과하면 체질에 따라 설사가 유발되는 문제가 발생될 수 있다. The magnesium contains 5 to 10% by weight based on 100% by weight. At this time, when the content is added to less than 5% by weight based on 100% by weight of the whole chajeonpi, there is no additive effect, and if it exceeds 10% by weight, diarrhea may occur depending on the constitution.
본 발명에 따른 다이어트용 보조식품 조성물은 홍삼 분말을 더 포함할 수 있다. Dietary supplement composition according to the present invention may further include a red ginseng powder.
상기 홍삼 분말은 사포닌, 유리지방산, 함질소 성분 등을 통해 비만치료 및 예방, 식욕억제 효과를 제공하는 구성이다.The red ginseng powder is a component that provides obesity treatment and prevention, and appetite suppression effects through saponins, free fatty acids, nitrogen-containing components, and the like.
이때, 홍삼 분말은 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 0.5 내지 5 중량부가 포함된다.At this time, the red ginseng powder contains 0.5 to 5 parts by weight based on 100 parts by weight of a mixture of Chaeongpi, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium.
여기서, 홍삼 분말이 0.5 중량부 미만으로 사용되면 홍삼 분말에 의한 효능과 효과를 발현하기 어려우며, 5 중량부를 초과하여 사용되면 홍삼의 맛이 강하게 느껴지게 되어 기호성이 떨어지는 문제가 발생된다.Here, when the red ginseng powder is used in less than 0.5 parts by weight, it is difficult to express the efficacy and effect by the red ginseng powder, and when it is used in excess of 5 parts by weight, the taste of red ginseng is strongly felt, resulting in a problem of poor tasteability.
또한, 홍삼 분말은 배당체(glycosides), 인삼향성분(panacen), 폴리아세틸렌계 화합물, 함질소 성분, 플라보노이드(flavonoid), 비타민(B복합체, 니코틴산, 판토텐산, 폴산, 비오틴 등), 미량원소(망간, 구리, 바나듐, 코발트, 비소, 게르마늄, 인, 알루미늄, 니켈 등), 효소(아밀라아제, 페놀라아제), 항산화물질, 아미노산(21종), 유리지방산(24종) 등을 주요 성분으로 포함한다. In addition, red ginseng powder includes glycosides, ginseng flavor components (panacen), polyacetylene compounds, nitrogen-containing components, flavonoids, vitamins (B complex, nicotinic acid, pantothenic acid, folic acid, biotin, etc.), trace elements (manganese) , Copper, vanadium, cobalt, arsenic, germanium, phosphorus, aluminum, nickel, etc.), enzymes (amylase, phenolase), antioxidants, amino acids (21 species), free fatty acids (24 species), etc. .
이 성분들은 중추신경에 대해서 진정작용과 흥분작용이 있으며, 순환계에 작용하여 고혈압이나 동맥경화의 예방효과가 있고, 조혈작용을 하면서 혈당치를 저하시켜 주며, 간을 보호하며, 항염 및 항종양작용을 하고, 스트레스 해소에도 효과가 있음이 입증되었다.These components have sedative and excitatory effects on the central nervous system, act on the circulatory system to prevent hypertension or arteriosclerosis, reduce blood sugar levels while hematopoietic, protect the liver, and have anti-inflammatory and anti-tumor effects. And it was proved to be effective in relieving stress.
본 발명에 따른 다이어트용 보조식품 조성물은 L-아르기닌을 더 포함할 수 있다. Dietary supplement composition according to the present invention may further include L-arginine.
상기 L-아르기닌은 필수 아미노산계열이긴 하지만 체내에서 합성되지 않거나 합성되더라도 그 양이 매우 적어 외부로부터 공급받아야 하는 아미노산으로서, 체지방을 감소시키는 기능이 있다.Although the L-arginine is an essential amino acid family, it is an amino acid that must be supplied from the outside due to its very small amount even though it is not synthesized or synthesized in the body, and has a function of reducing body fat.
이러한 L-아르기닌은 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 1 내지 5 중량부가 포함된다.The L-arginine contains 1 to 5 parts by weight based on 100 parts by weight of a mixture of chamopi, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium.
이때, L-아르기닌은 1 중량부 미만으로 첨가되는 경우 첨가효과가 없고, 5 중량부를 초과하면 체질에 따라 체내 암모니아 축적으로 인해 체취가 발생될 수 있다. At this time, when L-arginine is added in an amount of less than 1 part by weight, there is no addition effect, and if it exceeds 5 parts by weight, body odor may be generated due to accumulation of ammonia in the body depending on the constitution.
본 발명에 따른 다이어트용 보조식품 조성물은 글루타티온을 더 포함할 수 있다. Dietary supplement composition according to the present invention may further include glutathione.
상기 글루타티온(glutathione)은 아미노산이 배열된 화합물의 일종로, 글루타민산과 시스테인 및 글리신이라는 3개의 아미노산이 펩티드 결합된 트리 펩타이드 구조를 갖는다. 이는 세포내에서 효소 글루타티온-전이효소 및 글루타티온-과산화효소(이는 치명적 분자, 예컨대 과산화수소를 파괴하기 위해 세포에 의해 사용됨)의 보조 인자로 작용한다. 설프하이드릴기의 존재에 기인하여, 글루타티온은 환원된 형태로부터 산화된 형태로 바뀌어 산화방지제로도 작용한다. The glutathione is a kind of a compound in which amino acids are arranged, and has a tripeptide structure in which three amino acids, glutamic acid and cysteine and glycine, are peptide-bonded. It acts as a cofactor of the enzymes glutathione-transferase and glutathione-peroxidase in the cell (which is used by cells to destroy lethal molecules, such as hydrogen peroxide). Due to the presence of sulfhydryl groups, glutathione changes from reduced form to oxidized form, which also acts as an antioxidant.
또한, 글루타티온은 산화 물질, 예컨대 유리 라디칼, 하이드로과산화물 및 지질과산화물과 반응하는 그의 능력에 기인하여 세포 노화를 방지하는 주요 효소로 작용한다. In addition, glutathione acts as a major enzyme that prevents cell aging due to its ability to react with oxidizing substances such as free radicals, hydroperoxides and lipid peroxides.
이러한 글루타티온은 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 0.5 내지 3 중량부가 포함된다. 이때, L-아르기닌은 0.5 중량부 미만으로 첨가되는 경우 첨가효과가 없고, 3 중량부를 초과하면 체질에 따라 발진, 위통, 오심, 구토가 발생될 수 있다. The glutathione contains 0.5 to 3 parts by weight based on 100 parts by weight of a mixture of tea stalk, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium. At this time, L-arginine has no added effect when added in less than 0.5 parts by weight, and if it exceeds 3 parts by weight, rash, stomach pain, nausea, and vomiting may occur depending on the constitution.
본 발명에 따른 다이어트용 보조식품 조성물은 유산균을 더 포함할 수 있다. Dietary supplement composition according to the present invention may further include lactic acid bacteria.
상기 유산균은 포도당 또는 탄수화물을 분해 이용하여 유산을 많이 생성하는 박테리아로서 단백질을 분해하지만 부패시키는 기능은 없으며, 인체에 유익한 작용을 한다.The lactic acid bacteria are bacteria that produce a lot of lactic acid by decomposing glucose or carbohydrates, but they decompose protein, but do not decompose, and have a beneficial effect on the human body.
구체적으로, 유산균은 섭취자의 장내에서 작용하여 변비 및 설사 개선, 장 기능 증진 등의 효과를 제공한다.Specifically, lactic acid bacteria act in the intestine of the ingestor to provide effects such as improving constipation and diarrhea, and improving bowel function.
이러한 유산균으로는 락토바실러스 카제이(Lactobacillus casei), 락토바실러스 플란타룸(Lactobacillus plantarum) 등을 사용할 수 있다.As such lactic acid bacteria, Lactobacillus casei, Lactobacillus plantarum, or the like can be used.
필요에 따라, 상기 유산균으로는 캡슐화 된 유산균을 사용하는 것이 바람직하다. 이는, 유산균의 흡수율과 안정성을 높이기 위함이다. If necessary, it is preferable to use encapsulated lactic acid bacteria as the lactic acid bacteria. This is to increase the absorption rate and stability of lactic acid bacteria.
예를 들면, 캡슐화 된 유산균의 제조는 변성전분과 말토덱스트린(DE16)을 증류수에 수화하여 수조에서 75℃ 까지 가온한 다음 균질기 압력 700psi에서 균질화하는 균질화 과정과, 탑스프레이가 장착된 유동층과립건조기에 유산균을 넣고 혼합한 후 피복물질, sodium alginate, cirtric acid를 H2O2에 넣어 제조하여 유산균 원료에 순간 분사하여 피복시키면서 건조하는 피복과정, 및 건조된 마이크로 입자를 메쉬망을 통해 50㎛ 이하의 사이즈를 갖는 미세캡슐만을 선별하는 선별과정으로 이루어진다. For example, the production of encapsulated lactic acid bacteria is a homogenization process of hydrating modified starch and maltodextrin (DE16) in distilled water and heating it to 75 ° C in a water bath, then homogenizing it at a pressure of 700 psi of a homogenizer, and a fluidized bed granulator equipped with a top spray After adding lactic acid bacteria to the mixture, the coating material, sodium alginate, and cirtric acid are prepared in H 2 O 2 and sprayed on the raw material of lactic acid bacteria to coat and dry while coating, and the dried microparticles are 50 μm or less through the mesh network. It consists of a screening process to select only the fine capsules having a size of.
이러한 유산균은 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 1 내지 3 중량부가 포함된다. 이때, 유산균은 1 중량부 미만으로 첨가되는 경우 첨가효과가 없고, 3 중량부를 초과하면 체질에 따라 변비, 복통, 설사가 발생될 수 있다. These lactic acid bacteria include 1 to 3 parts by weight based on 100 parts by weight of a mixture of chamokpi, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium. At this time, when the lactic acid bacteria are added less than 1 part by weight, there is no additive effect, and when it exceeds 3 parts by weight, constipation, abdominal pain, and diarrhea may occur depending on the constitution.
본 발명에 따른 다이어트용 보조식품 조성물은 난소화성말토덱스트린을 더 포함할 수 있다. Dietary supplement composition according to the present invention may further include an indigestible maltodextrin.
상기 난소화성말토덱스트린(indigestible maltodextrin)은 인간이 소화하기 어려운 말토덱스트린으로 설탕의 15% 정도의 단맛을 가지고 있으며, 보통 식이섬유의 원료로 사용된다.The indigestible maltodextrin is a maltodextrin that is difficult for humans to digest and has a sweetness of about 15% of sugar, and is usually used as a raw material for dietary fiber.
그리고 난소화성말토덱스트린은 장기능 개선, 식후 혈당상승조절, 혈중콜레스테롤조절, 중성지질조절을 돕는 효과를 제공한다.In addition, the indigestible maltodextrin provides an effect of improving intestinal function, controlling blood sugar elevation after eating, regulating blood cholesterol, and neutral lipid control.
상기 난소화성말토덱스트린은 옥수수 전분에서 얻어지며 소화되기 어려운 구조로 되어 있어 소화효소인 아밀라제에 의해 분해되기 어렵다. 이와 같은 난소화성말토덱스트린은 식사와 함께 섭취하면 당의 흡수를 억제시켜 식후 혈당 조절에 도움을 줄 수 있다. 음식으로 섭취한 당질은 단당류(포도당, 과당, 갈락토즈)로 분해되고 소장에서 흡수되어 간으로 가게 되는데, 이때 난소화성말토덱스트린은 소화되기가 어려워, 소장에서 통과시간을 지연시키거나 식사로 섭취한 당의 흡수를 방해하게 된다.The indigestible maltodextrin is obtained from corn starch and has a structure that is difficult to digest, making it difficult to decompose by the digestive enzyme amylase. These indigestible maltodextrins, when taken with meals, can inhibit sugar absorption and help control blood sugar after meals. Carbohydrates ingested as food are broken down into monosaccharides (glucose, fructose, galactose), absorbed from the small intestine, and passed to the liver. At this time, indigestible maltodextrin is difficult to digest, delaying the passage time in the small intestine, or consumed as a meal. It interferes with the absorption of sugar.
이러한 난소화성말토덱스트린은 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 1 내지 7 중량부가 포함된다. 이때, 난소화성말토덱스트린은 1 중량부 미만으로 첨가되는 경우 첨가효과가 없고, 7 중량부를 초과하면 과량의 식이섬유로 인한 장 트러블이 발생될 수 있다. The indigestible maltodextrin contains 1 to 7 parts by weight based on 100 parts by weight of a mixture of chamopi, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium. At this time, when the indigestible maltodextrin is added in an amount of less than 1 part by weight, there is no additive effect, and when it exceeds 7 parts by weight, intestinal trouble due to excess dietary fiber may occur.
필요에 따라, 본 발명에 따른 다이어트용 보조식품 조성물에는 그 유효성분 이외에 감미제, 풍미제, 생리활성 성분, 미네랄 등이 포함될 수 있다.If necessary, the dietary supplement composition according to the present invention may include sweeteners, flavoring agents, bioactive ingredients, minerals, and the like, in addition to the active ingredients.
상기 감미제는 식품이 적당한 단맛을 나게 사용되는 것으로, 천연 감미제나 합성 감지제를 사용할 수 있으며, 천연 감지제를 사용하는 것이 바람직하다. 이러한 천연 감미제로는 옥수수 시럽 고형물, 꿀, 수크로오스, 프룩토오스, 락토오스, 말토오스 등의 당 감미제를 사용할 수 있다.The sweetener is that the food is used to have a moderate sweetness, and a natural sweetener or a synthetic sensing agent may be used, and it is preferable to use a natural sensing agent. As the natural sweetener, sugar sweeteners such as corn syrup solids, honey, sucrose, fructose, lactose, and maltose can be used.
상기 풍미제는 맛이나 향을 좋게 하기 위하여 사용되는 것으로, 천연 풍미제나 합성 풍미제를 사용할 수 있으며, 천연 풍미제를 사용하는 것이 바람직하다. 이러한 천연 풍미제로는 사과, 레몬, 감귤, 포도, 딸기, 복숭아 등에서 얻어진 것이거나 녹차잎, 둥굴레, 대잎, 계피, 국화 잎, 자스민 등에서 얻어진 것을 사용할 수 있다. 또한, 인삼(홍삼), 죽순, 알로에 베라, 은행 등에서 얻어진 것을 사용할 수도 있다. The flavoring agent is used to improve the taste or aroma, natural flavoring agents or synthetic flavoring agents may be used, and it is preferable to use natural flavoring agents. Such natural flavoring agents may be obtained from apples, lemons, citrus fruits, grapes, strawberries, peaches, etc., or those obtained from green tea leaves, perilla, large leaves, cinnamon, chrysanthemum leaves, jasmine, and the like. In addition, those obtained from ginseng (red ginseng), bamboo shoots, aloe vera, and ginkgo can also be used.
아울러, 천연 풍미제는 액상의 농축액이나 고형상의 추출물을 사용할 수 있다. 경우에 따라서 합성 풍미제가 사용될 수 있는데, 합성 풍미제로는 에스테르, 알콜, 알데하이드, 테르펜 등이 이용될 수 있다.In addition, the natural flavoring agent may be a liquid concentrate or a solid extract. In some cases, synthetic flavoring agents may be used. As the synthetic flavoring agent, esters, alcohols, aldehydes, terpenes, and the like may be used.
상기 생리 활성 물질로는 카테킨, 에피카테킨, 갈로가테킨, 에피갈로카테킨 등의 카테킨류나, 레티놀, 아스코르브산, 토코페롤, 칼시페롤, 티아민, 리보플라빈 등의 비타민류 등이 사용될 수 있다.As the bioactive material, catechins such as catechin, epicatechin, galogatechin, and epigallocatechin, vitamins such as retinol, ascorbic acid, tocopherol, calciferol, thiamine, riboflavin, and the like can be used.
상기 미네랄로서는 칼슘, 마그네슘, 크롬, 코발트, 구리, 불소화물, 게르마늄, 요오드, 철, 리튬, 마그네슘, 망간, 몰리브덴, 인, 칼륨, 셀레늄, 규소, 나트륨, 황, 바나듐, 아연 등이 사용될 수 있다.As the mineral, calcium, magnesium, chromium, cobalt, copper, fluoride, germanium, iodine, iron, lithium, magnesium, manganese, molybdenum, phosphorus, potassium, selenium, silicon, sodium, sulfur, vanadium, zinc, etc. may be used. .
필요에 따라, 본 발명의 식품 조성물은 상기 감미제 등 이외에도 보존제, 유화제, 산미료, 점증제 등을 더 포함할 수 있다. If necessary, the food composition of the present invention may further include a preservative, emulsifier, acidulant, thickener, and the like in addition to the sweetener.
이러한 보존제, 유화제 등은 그것이 첨가되는 용도를 달성할 수 있는 한 극미량으로 첨가되어 사용되는 것이 바람직하다. 여기서, 극미량이란 수치적으로 표현할 때 식품 조성물 전체 중량을 기준으로 할 때 0.0005 중량% 내지 약 0.5중량% 범위를 의미한다.It is preferable that such preservatives, emulsifiers, etc. are added and used in a very small amount as long as it can achieve the use to which they are added. Here, the trace amount when expressed numerically refers to a range of 0.0005% by weight to about 0.5% by weight based on the total weight of the food composition.
상기 보존제로는 식품첨가용 글리세린, 소듐 소르브산칼슘, 소르브산나트륨, 소르브산칼륨, 벤조산칼슘, 벤조산나트륨, 벤조산칼륨, EDTA(에틸렌디아민테트라아세트산) 등을 사용할 수 있다.As the preservative, food additives such as glycerin, sodium calcium sorbate, sodium sorbate, potassium sorbate, calcium benzoate, sodium benzoate, potassium benzoate, and EDTA (ethylenediaminetetraacetic acid) can be used.
이하, 본 발명의 구체적인 실시예 및 실험예를 통하여 보다 구체적으로 기술한다. 다만 본 실시예 및 실험예는 상술한 발명의 특정예의 이해를 돕기 위한 것으로 이에 의하여 권리범위 등이 제한적으로 해석되어서는 아니된다.Hereinafter, it will be described in more detail through specific examples and experimental examples of the present invention. However, the present examples and experimental examples are intended to help understand the specific examples of the above-described invention, and the scope of rights and the like should not be interpreted thereby.
[실시예 1][Example 1]
차전자피 분말 4.5㎏, 흰강낭콩 단백질 추출물 분말 1.5㎏, 가르시니아 캄보지아 추출물 분말 2㎏, 키토산 분말 0.5㎏, L-카르니틴 분말 1㎏, 마그네슘 분말 0.5㎏을 혼합하여 식품 조성물을 제조하였다. A food composition was prepared by mixing 4.5 kg of tea powder, 1.5 kg of white kidney bean protein extract powder, 2 kg of garcinia cambogia extract powder, 0.5 kg of chitosan powder, 1 kg of L-carnitine powder, and 0.5 kg of magnesium powder.
[비교예][Comparative example]
흰강낭콩 단백질 추출물 분말 1.5㎏과 가르시니아 캄보지아 추출물 분말 2㎏을 혼합하여 식품 조성물을 제조하였다.Food composition was prepared by mixing 1.5 kg of white kidney bean protein extract powder and 2 kg of garcinia cambogia extract powder.
[실험예][Experimental Example]
직업에 상관없이 30~40대 성인 여성 40명을 나이에 관계없이 무작위 추출방법으로 선택한 뒤 다이어트용 보조식품 조성물을 섭취할 시험군과 곡류 분말만을 섭취할 대조군으로 나누어 6주간 임상실험을 하였다. 구체적으로, 다이어트용 보조식품 조성물은 아침과 저녁 식전 30분 이내에 1포(4.5g)씩 섭취하도록 하였으며, 복용 시에는 물을 1컵 이상 충분히 섭취하도록 하였다.Forty weeks, 40 female adults in their 30s and 40s were selected as random sampling methods regardless of age, and divided into a test group to consume dietary supplement composition and a control group to consume only grain powder for 6 weeks. Specifically, the dietary supplement composition was intended to be consumed 1 packet (4.5g) within 30 minutes before breakfast and dinner, and sufficient water was consumed at least 1 cup when taken.
실험에 참여한 여성들은 모두 과거에 특이한 신체질병이나 결함이 없었으며, 실험 기간 동안 연구결과에 영향을 미칠만한 다른 약제의 섭취는 금하도록 하였다. 과다체중이나 저체중의 여성들은 체중의 변화가 다르게 나올 것으로 예상되어 여성들의 체중은 55㎏에서 75㎏까지로 제한하였다.All women participating in the experiment had no specific physical illness or defects in the past, and they were prohibited from taking other medications that could affect the study results during the experiment. Women who were overweight or underweight were expected to have different body weight changes, limiting women's weight from 55 to 75 kg.
실험 참가자의 신체정보는 [표 1]과 같이 관찰되었고, 실험 결과는 [표 2]로 나타내었다.The physical information of the experiment participants was observed as shown in [Table 1], and the experimental results are shown in [Table 2].
[표 2]를 참조하면, 시험군은 4주 후부터 체중감소 효과가 나타났고, 각 기간에서 집단 간 차이가 확인되었다. 체중의 유의한 감소에 따라 BMI에서도 대조군에 비해 유의한 감소와 집단 간 차이가 확인되었다. 대조군에서도 체중과 체지방에서 감소가 나타났으나 유의한 변화는 나타나지 않았다. 최종 6주 후에는 대조군과 비교 시 체중과 체지방 모두에서 유의한 감소효과를 나타내었다.Referring to [Table 2], the test group showed a weight loss effect after 4 weeks, and differences between groups were confirmed in each period. According to the significant weight loss, the BMI also showed a significant decrease and differences between groups compared to the control group. The control group also showed a decrease in body weight and body fat, but no significant change. After the final 6 weeks, there was a significant reduction effect in both body weight and body fat compared to the control group.
이상에서 본 발명의 바람직한 실시예를 참조하여 설명하였지만, 해당 기술분야의 숙련된 당업자는 하기의 특허청구범위에 기재된 본 발명의 사상 및 영역으로부터 벗어나지 않는 범위 내에서 본 발명을 다양하게 수정 및 변경시킬 수 있음을 이해할 수 있을 것이다.Although described above with reference to preferred embodiments of the present invention, those skilled in the art can variously modify and change the present invention without departing from the spirit and scope of the present invention as set forth in the claims below. You will understand that you can.
Claims (6)
차전자피 40 내지 50 중량%, 흰강낭콩 단백질 추출물 15 내지 25 중량%, 가르시니아 캄보지아 추출물 20 내지 30 중량%, 키토산 3 내지 10 중량%, L-카르니틴 5 내지 15 중량%, 및 마그네슘 5 내지 10 중량%로 구성된 것을 특징으로 하는 다이어트용 보조식품 조성물.According to claim 1,
40 to 50% by weight of psyllium husk, 15 to 25% by weight of white kidney protein extract, 20 to 30% by weight of garcinia cambogia, 3 to 10% by weight of chitosan, 5 to 15% by weight of L-carnitine, and 5 to 10% by weight of magnesium Dietary supplement composition characterized in that the composition.
상기 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 L-아르기닌 1 내지 5 중량부를 더 포함하는 것을 특징으로 하는 다이어트용 보조식품 조성물.According to claim 2,
Dietary supplement composition for diet, characterized in that it further comprises 1 to 5 parts by weight of L-arginine based on 100 parts by weight of the mixture of the Chaeongpi, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium.
상기 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 글루타티온 0.5 내지 3 중량부를 더 포함하는 것을 특징으로 하는 다이어트용 보조식품 조성물.According to claim 2,
Dietary supplement composition characterized in that it further comprises 0.5 to 3 parts by weight of glutathione, based on 100 parts by weight of the mixture of the tea seed skin, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium.
상기 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 유산균 1 내지 3 중량부를 더 포함하는 것을 특징으로 하는 다이어트용 보조식품 조성물.According to claim 2,
Dietary supplement composition, characterized in that it further comprises 1 to 3 parts by weight of lactic acid bacteria based on 100 parts by weight of the mixture of the Chaeongpi, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium.
상기 차전자피, 흰강낭콩 단백질 추출물, 가르시니아 캄보지아 추출물, 키토산, L-카르니틴, 및 마그네슘의 혼합물 100 중량부를 기준으로 난소화성말토덱스트린 1 내지 7 중량부를 더 포함하는 것을 특징으로 하는 다이어트용 보조식품 조성물. According to claim 2,
Dietary supplement composition, characterized in that it further comprises 1 to 7 parts by weight of the indigestible maltodextrin based on 100 parts by weight of the mixture of the Chaeongpi, white kidney bean protein extract, garcinia cambogia extract, chitosan, L-carnitine, and magnesium.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180123383A KR20200043555A (en) | 2018-10-16 | 2018-10-16 | Supplement food composition for diet |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180123383A KR20200043555A (en) | 2018-10-16 | 2018-10-16 | Supplement food composition for diet |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20200043555A true KR20200043555A (en) | 2020-04-28 |
Family
ID=70456007
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020180123383A KR20200043555A (en) | 2018-10-16 | 2018-10-16 | Supplement food composition for diet |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR20200043555A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210158585A (en) | 2020-06-24 | 2021-12-31 | 홍연희 | Supplement food composition for diet |
| KR102461437B1 (en) * | 2022-05-02 | 2022-11-02 | 최진원 | Pharmaceutical composition for preventing or treating obesity having garcinia cambogia extract and health functional food having the same |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100439094B1 (en) | 2002-07-09 | 2004-07-05 | 이재문 | Dietary food using by Hoelen |
| KR100448260B1 (en) | 2001-04-02 | 2004-09-10 | (주)명세당 | Processing Methods of Health Food for the Control of Body Weight |
| KR100602281B1 (en) | 2004-06-10 | 2006-07-19 | 풍기특산물영농조합법인 | The diet-meal power composite which is contained slices of process-genseng |
| KR100853940B1 (en) | 2007-02-20 | 2008-08-25 | 유상훈 | Anti-obesity constituents made by fermented tangle, salty pasture, laver, soybean |
-
2018
- 2018-10-16 KR KR1020180123383A patent/KR20200043555A/en not_active Application Discontinuation
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100448260B1 (en) | 2001-04-02 | 2004-09-10 | (주)명세당 | Processing Methods of Health Food for the Control of Body Weight |
| KR100439094B1 (en) | 2002-07-09 | 2004-07-05 | 이재문 | Dietary food using by Hoelen |
| KR100602281B1 (en) | 2004-06-10 | 2006-07-19 | 풍기특산물영농조합법인 | The diet-meal power composite which is contained slices of process-genseng |
| KR100853940B1 (en) | 2007-02-20 | 2008-08-25 | 유상훈 | Anti-obesity constituents made by fermented tangle, salty pasture, laver, soybean |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210158585A (en) | 2020-06-24 | 2021-12-31 | 홍연희 | Supplement food composition for diet |
| KR102461437B1 (en) * | 2022-05-02 | 2022-11-02 | 최진원 | Pharmaceutical composition for preventing or treating obesity having garcinia cambogia extract and health functional food having the same |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6900173B2 (en) | Perioperative multivitamin protein bar for use in preparing an individual for fast surgical recovery | |
| US20040253227A1 (en) | Perioperative multivitamin protein beverage and additive for use in preparing an individual for fast surgical recovery | |
| CN111528483A (en) | Functional meal replacement powder with balanced nutrition and preparation method thereof | |
| EP1427297B1 (en) | Composition for reducing appetite in mammals comprising procyanidin | |
| CN108719984B (en) | Meal replacement nutrition bar for gout population and preparation method thereof | |
| CN108685100B (en) | Formula and preparation method of composite short peptide powder | |
| KR101539382B1 (en) | Food composition for improvement of bowel function and relief of constipation | |
| KR102020586B1 (en) | Healthy foods for elderly comprising animal and plant extracts and process for preparation thereof | |
| CN112042862A (en) | Weight-losing meal-replacing food and application | |
| CN108618126A (en) | A kind of Chinese medicine compound prescription weight-reducing special diet food and preparation method thereof | |
| CN108887564A (en) | A kind of selenoprotein coarse cereal powder formula and preparation method thereof | |
| CN107373672A (en) | A kind of irregular menstruation women tailored version clinical nutrition formula and preparation method thereof | |
| KR101896024B1 (en) | Functional food compositions comprising extracts of Garcinia Cambogia as active ingredients and method for preparing the same | |
| CN106858215A (en) | A kind of passion fruit composite beverage | |
| KR101722481B1 (en) | functional cereal containing sweet potato chip, broccoli granule and dietary fiber for constipation improving and manufacturing method thereof | |
| KR102422527B1 (en) | Supplement food composition for diet | |
| KR101456415B1 (en) | The method of manufacturing bar-shaped functional food for diet | |
| CN101606701B (en) | Natto pollen preparation | |
| CN114208989A (en) | Ketogenic meal replacement nutrition powder solid beverage and preparation method thereof | |
| KR20200043555A (en) | Supplement food composition for diet | |
| KR20040095949A (en) | Composition comprising herb with usage to prevent obeseness | |
| CN108541912A (en) | A kind of anti-oxidant blood-fat reducing composition and its preparation method and application | |
| CN108308621A (en) | A kind of weight losing meal-replacing powder rich in rice oligopeptide | |
| CN112189839A (en) | Composition for assisting in reducing blood fat and application thereof | |
| KR20210035592A (en) | galeusinialeul iyonghan biman haeso gineung-ui saengsigtaib cha joseongmul |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AMND | Amendment | ||
| E601 | Decision to refuse application | ||
| X091 | Application refused [patent] | ||
| E601 | Decision to refuse application | ||
| E801 | Decision on dismissal of amendment |