KR20180098651A - 발광 화합물 - Google Patents
발광 화합물 Download PDFInfo
- Publication number
- KR20180098651A KR20180098651A KR1020187021898A KR20187021898A KR20180098651A KR 20180098651 A KR20180098651 A KR 20180098651A KR 1020187021898 A KR1020187021898 A KR 1020187021898A KR 20187021898 A KR20187021898 A KR 20187021898A KR 20180098651 A KR20180098651 A KR 20180098651A
- Authority
- KR
- South Korea
- Prior art keywords
- ring
- substituted
- tadf
- species
- ring system
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent materials, e.g. electroluminescent or chemiluminescent
- C09K11/06—Luminescent materials, e.g. electroluminescent or chemiluminescent containing organic luminescent materials
-
- H01L51/50—
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
- H10K50/135—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers comprising mobile ions
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1033—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1037—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with sulfur
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/10—Triplet emission
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/20—Delayed fluorescence emission
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Organic Chemistry (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Indole Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Nitrogen- Or Sulfur-Containing Heterocyclic Ring Compounds With Rings Of Six Or More Members (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
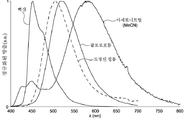
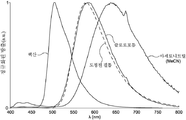
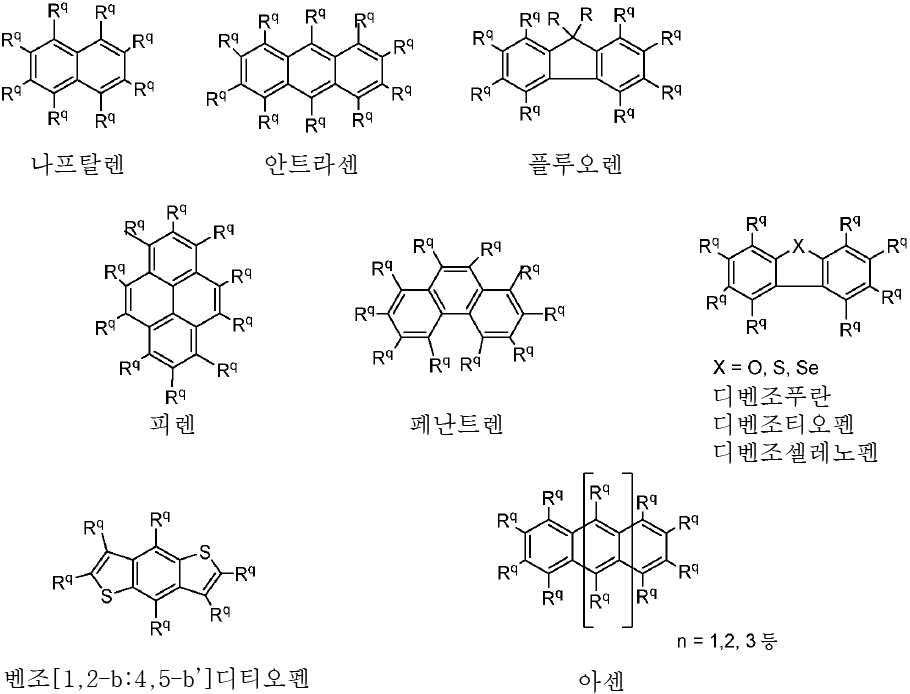
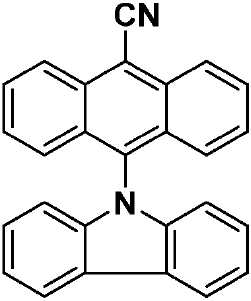
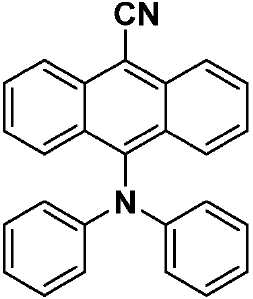
도 2는 TADF 종의 순환 전압곡선(cyclic voltammogram)을 나타내고;
도 3a는 TADF 종의 방출 스펙트럼을 나타내고;
도 3b는 TADF 종의 방출 스펙트럼을 나타낸다.
| 화학식 VII, 화학식 VIII 및 화학식 IX의 흡수 및 전기 화학적 데이터 | ||
| 분자 | λabs a (㎚)[ε(x104M-1㎝-1)] | 전기 화학론 b (eV) |
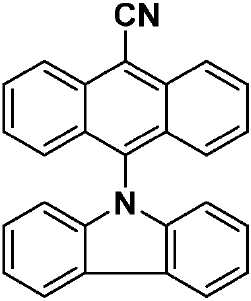
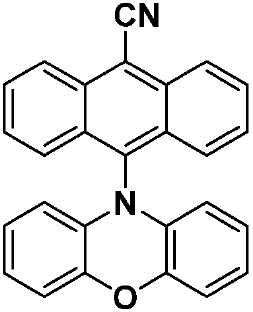
| CzAnCN(화학식 VII) | 310 [0.29], 322 [0.45], 334 [0.63], 352 [0.46], 370 [0.87], 390 [0.91], 408 [0.84], 435(sh) [0.31] | HOMO: -5.99 LUMO: -3.15 △E: 2.84 |
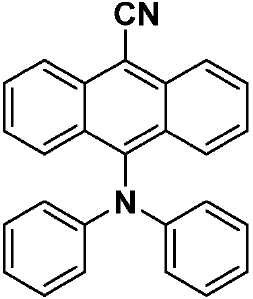
| TPAAnCN(화학식 VIII) | 343(sh) [0.23], 361(sh) [0.47], 373 [0.62], 381 [0.64], 402 [0.58], 472 [0.64] | HOMO: -5.68 LUMO: -3.20 △E: 2.84 |
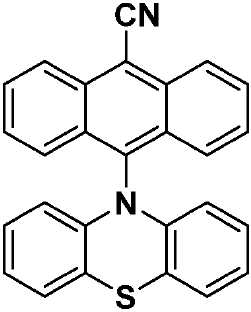
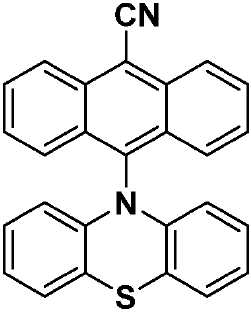
| PTZAnCN(화학식 IX) | 328(br) [0.58], 353 [0.69], 370 [1.24], 388 [1.53], 411 [1.46], 480 [0.04] | HOMO: -5.19 LUMO: -3.15 △E: 2.04 |
| a . 298K에서 DCM 중에서 측정. b . 지지 전해질로서 0.1M [nBu4N]PF6을, 그리고 내부 기준물질(SCE 대비 0.38V)로서 Fc/Fc+를 사용하여 MeCN 중에서 측정. HOMO 및 LUMO 에너지는 하기 식을 이용하여 계산되었다: EHOMO/LUMO = -(Eox pa,1/ERED PC,1 + 4.8)eV17(여기서, Eox pa 및 ERED PC는 각각 양극 및 음극 피크 전위임). ΔΕ = -(EHOMO-ELUMO). | ||
| 화학식 VII, 화학식 VIII 및 화학식 IX의 광 물리학 데이터 | ||||
| CzAnCN(화학식 VII) | TPAAnCN(화학식 VIII) | PTZAnCN(화학식 IX) | ||
| 헥산 | λem a (㎚) | 452(49) | 506(55) | 641(105) |
| ΦPL b (%) | 63.0(27.8) | 69.8(47.2) | n.d. | |
| Te(ns) | 22.3, 760 | 26.3, 858 | 5.3, 1,320 | |
| CHCl3 | λem a (㎚) | 518(83) | 588(98) | N/A |
| ΦPL b (%) | 32.8(14.7) | 43.2(37.5) | N/A | |
| Te(ns) | 38.8, 708 | 30.1 | N/A | |
| MeCN | λem a (㎚) | 581(124) | 638(137) | N/A |
| ΦPL b (%) | 5.7(2.9) | 6.7(6.3) | N/A | |
| Te(ns) | 13.8, 1,050 | 8.3 | N/A | |
| 박막 c | λem a (㎚) | 504(78) | 580(92) | 674(212) |
| ΦPL d (%) | 49.2(47.0) | 63.4(62.6) | 1.4(1.0) | |
| Te(ns) | 21.6, 416 | 29.4, 644 | 8.6, 1,032 | |
| a . 방출 최대치 및 반치전폭(FWHM)은 탈기 용액에서 보고된다. b . H2SO4(수성) 중의 0.5M 황산퀴닌은 기준물질(ΦPL: 54.6%)로서 사용되었다. 인용된 값은 탈기 용액에 대한 것이다. 괄호 한의 값은 통기 용액에 대한 것이다. c . 박막은 PMMA에서 10중량%의 도핑된 샘플을 스핀-코팅함으로써 제조되었다. d. 값은 적분구를 이용하여 결정된다. 탈기는 N2 퍼지(N2 purge)에 의한 것이다. | ||||
Claims (18)
- 제 1 항에 있어서, 상기 고리 시스템(Q)은 적어도 하나의 고도 불포화 고리를 포함하는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 1 항에 있어서, 상기 고리 시스템(Q)은 적어도 하나의 다른 고리에 융합된 적어도 하나의 벤젠 고리를 포함하는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 1 항에 있어서, 상기 고리 시스템(Q)에서 함께 융합된 적어도 2개의 고리 둘 모두는 방향족 및/또는 헤테로 방향족 고리인 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 1 항에 있어서, 상기 고리 시스템(Q)은 어닐레이팅(annelating)된 벤젠 또는 어닐레이팅된 헤테로아렌 고리 시스템인 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 1 항 내지 제 5 항 중 어느 한 항에 있어서, 상기 공여체(D) 및 수용체(A) 모이어티는 상기 고리 시스템(Q)의 동일한 고리 상에 위치하는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 1 항 내지 제 6 항 중 어느 한 항에 있어서, 상기 공여체(D) 및 수용체(A) 모이어티는 벤젠 고리 상에서 서로에 대해 파라(para)-위치에 위치하는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
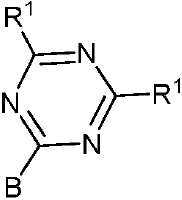
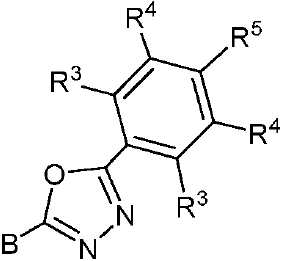
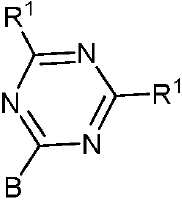
- 제 1 항 내지 제 7 항 중 어느 한 항에 있어서, 상기 수용체(A) 모이어티는 시아노(-CN), 케톤, 에스테르류, 아미드류, 알데히드류, 설폰류, 설폭시드류, 산화포스핀, 케톤류, 에스테르류, 아미드류, 알데히드류, 설폰류, 설폭시드류, 산화포스핀류, 및 치환 및 비치환된 피리미딘, 피라진, 1,2,4-트리아졸, 1,3,5-트리아진 및 1,3,4-옥사디아졸 모이어티로 이루어진 군으로부터 선택되는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 8 항에 있어서, 상기 적어도 하나의 수용체 모이어티는 화학식 II, 화학식 III 및 화학식 IIIa 중 하나에 따르는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종:
[화학식 II]
[화학식 III]
[화학식 IIIa]
상기 식에서, -B는 고리 시스템(Q)에 대한 결합 위치를 나타내고;
-R2는 환형일 수 있고 불포화될 수 있는 치환 또는 비치환된 1차, 2차 또는 3차 알킬(예를 들어, C1-C10 또는 심지어는 C1-C4)을 나타내고;
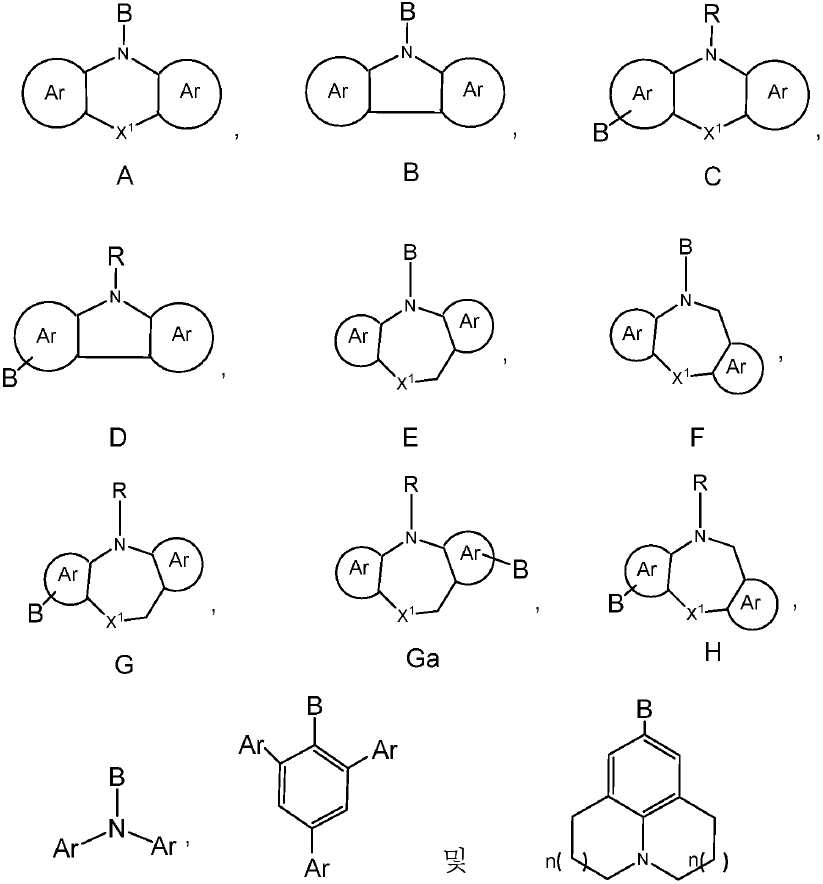
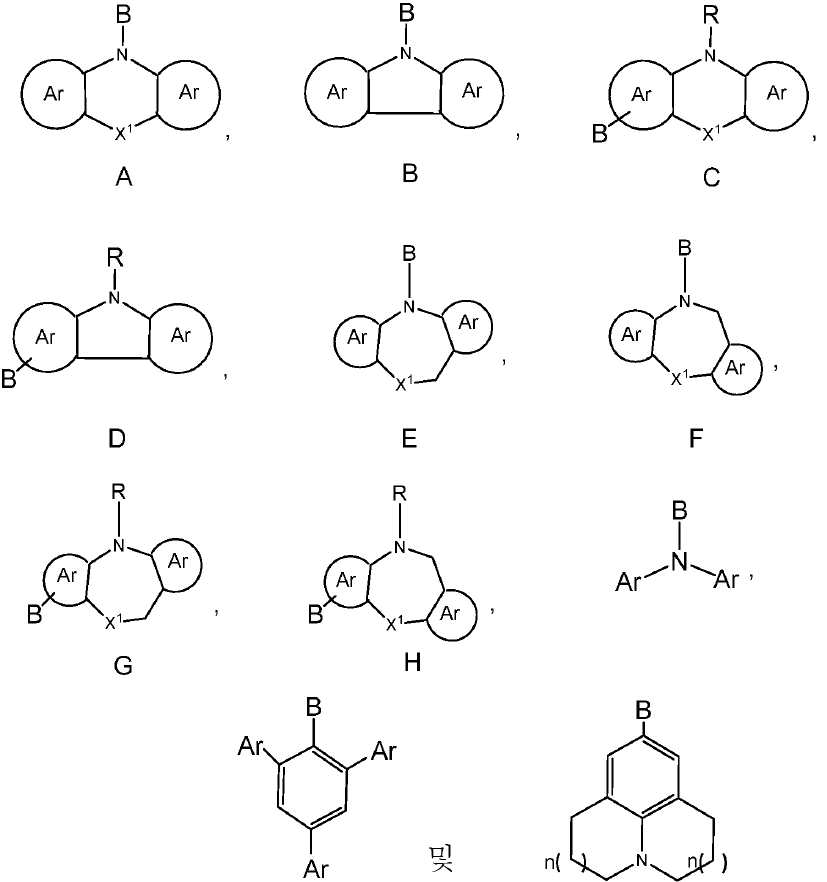
R1, R3, R4 및 R5 기는 각각의 경우에 대해 독립적으로는 -H, 환형일 수 있고 불포화될 수 있는 치환 또는 비치환된 1차, 2차 또는 3차 알킬(예를 들어, C1-C10 또는 심지어는 C1-C4); 치환 또는 비치환된 아릴 또는 헤테로아릴, -CF3, -OMe, -SF5, -NO2, 할로, 아릴 하이드록시, 아미노, 알콕시, 알킬티오, 카르복시, 시아노, 티오, 포르밀, 에스테르, 아실, 티오아실, 아미도, 설폰아미도, 카바메이트, 산화포스핀 및 황화포스핀으로 이루어진 군으로부터 선택된다. - 제 1 항 내지 제 9 항 중 어느 한 항에 있어서, 상기 공여체(D) 모이어티는 하기 화합물로 이루어진 군으로부터 선택되는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종:
상기 식에서, -B는 고리 시스템(Q)에 대한 결합 위치를 나타내되, 화학식 C, 화학식 D, 화학식 G 및 화학식 H의 구조 내에서는 질소에 대해 파라-위치에 있고;
X1은 O, S, NR, SiR2, PR 및 CR2로 이루어진 군으로부터 선택되고;
R 각각은 -H, 알킬, 아릴 또는 헤테로아릴(예를 들어, 치환 또는 비치환된 C1-C20 또는 심지어는 C1-C10 알킬)로 이루어진 군으로부터 독립적으로 선택되고;
Ar 각각은 각각의 경우에 대해 독립적으로는 치환 또는 비치환된 아릴 또는 헤테로아릴로 이루어진 군으로부터 선택되고;
은 각각의 경우에 대해 독립적으로는 화학식 A, 화학식 B, 화학식 C, 화학식 D, 화학식 E 또는 화학식 F의 구조의 중심 고리에 융합된 치환 또는 비치환된 아릴 또는 헤테로아릴 고리; 예를 들어, 5원 또는 6원의 치환 또는 비치환된 아릴 또는 헤테로아릴 고리를 나타내고;
n( )는 상기 벤젠 고리에 어닐레이팅된 고리 내의 포화된 -CH2- 기의 선택적 존재를 나타내고;
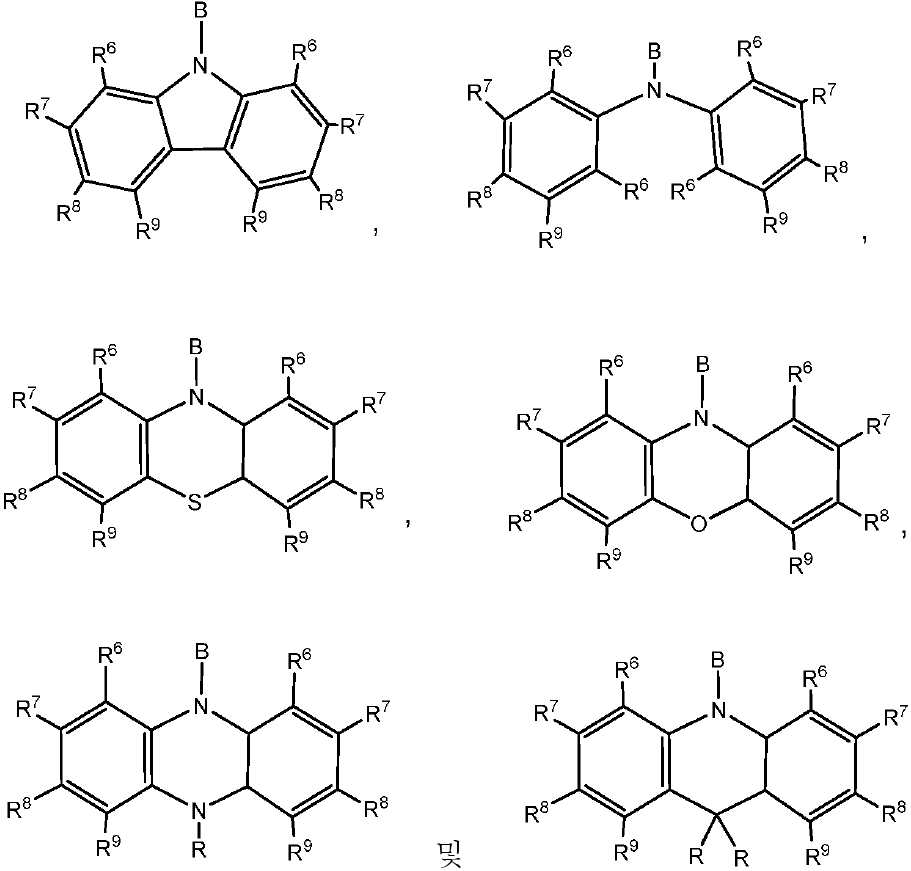
n은 각각의 경우에 대해 독립적으로는 0, 1 또는 2이다. - 제 1 항 내지 제 10 항 중 어느 한 항에 있어서, 상기 공여체(D) 모이어티는 하기 화합물로 이루어진 군으로부터 선택되는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종:
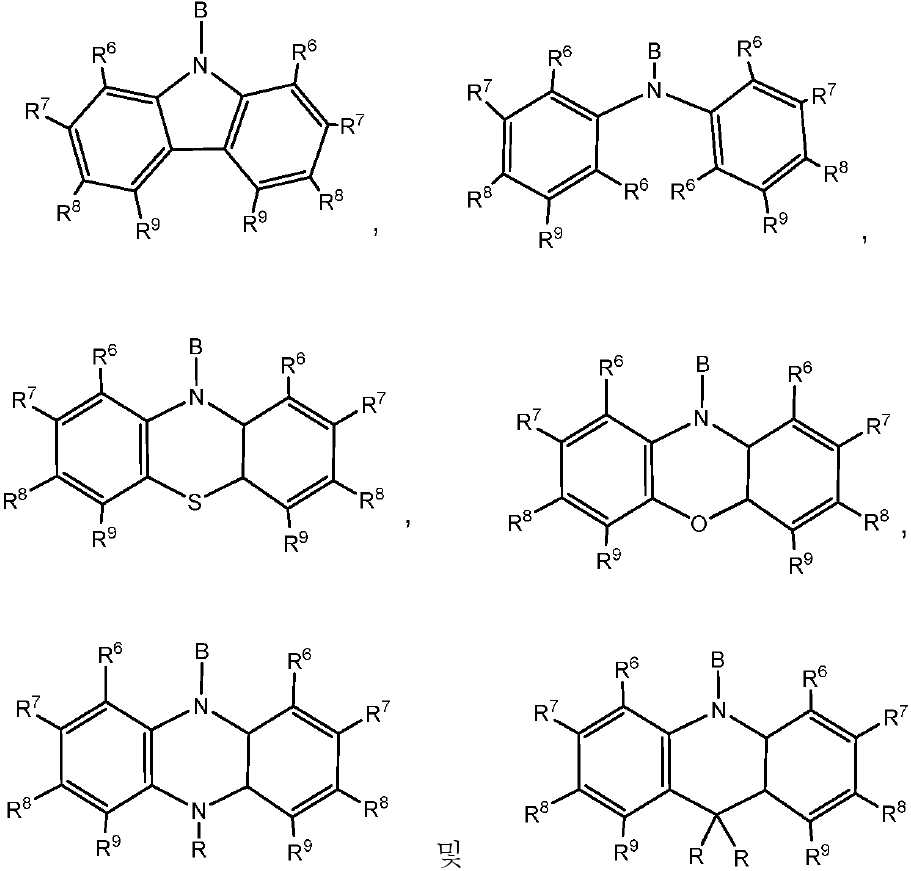
상기 식에서, -B는 고리 시스템(Q)에 대한 결합 위치를 나타내고;
R6, R7, R8 및 R9 기 각각은 각각의 경우에 대해 독립적으로는 -H, 환형일 수 있고 불포화될 수 있는 치환 또는 비치환된 1차, 2차 또는 3차 알킬(예를 들어, C1-C10 또는 심지어는 C1-C4); 치환 또는 비치환된 아릴 또는 헤테로아릴, -CF3, -OMe, -SF5, -NO2, 할로(예를 들어, 플루오로, 클로로, 브로모 및 요오도), 아릴, 아릴 하이드록시, 아미노, 알콕시, 알킬티오, 카르복시, 시아노, 티오, 포르밀, 에스테르, 아실, 티오아실, 아미도, 설폰아미도, 카바메이트, 산화포스핀 및 황화포스핀으로 이루어진 군으로부터 선택되고;
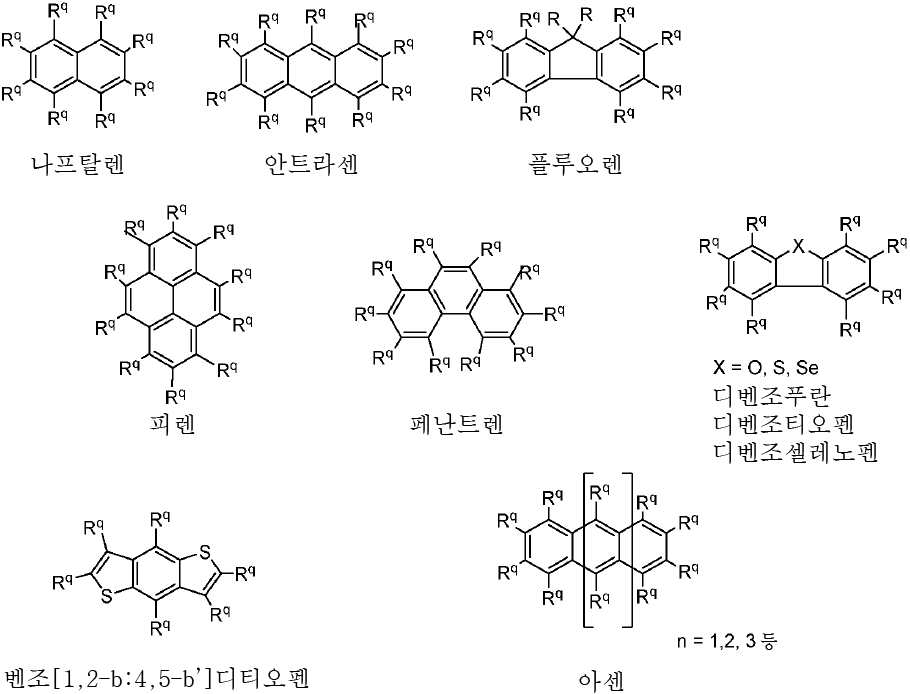
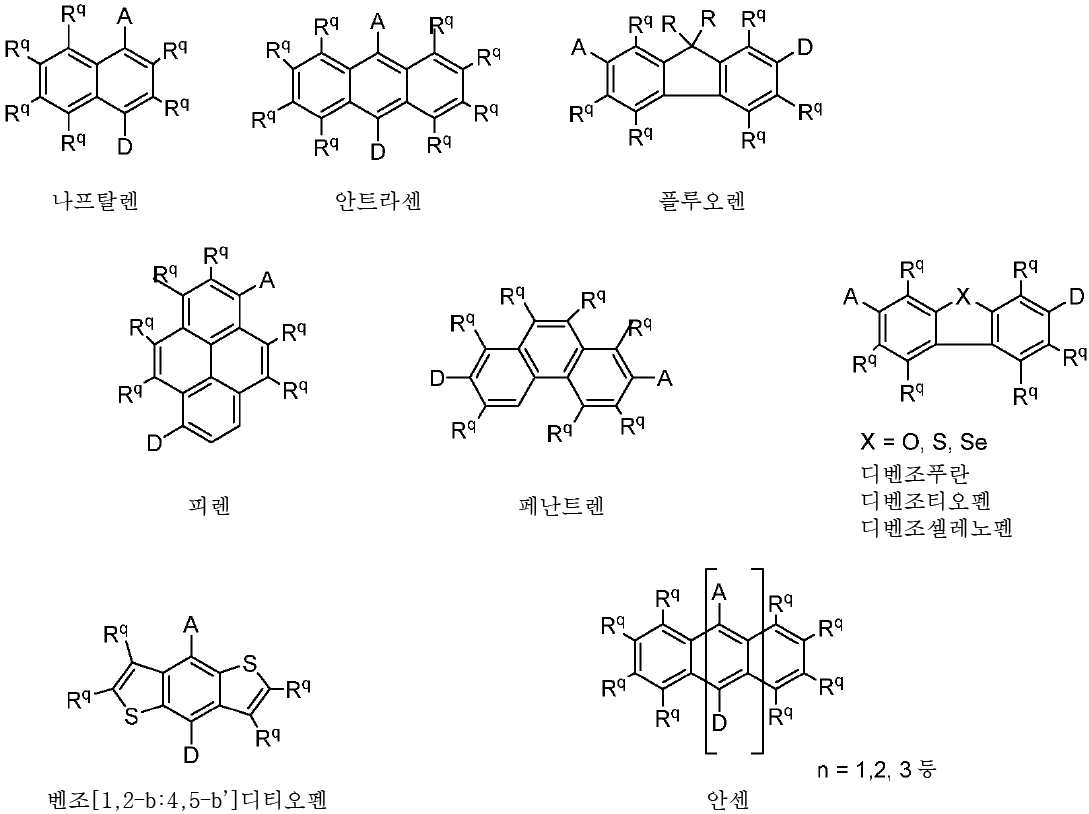
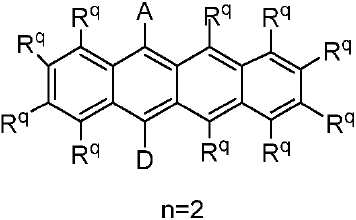
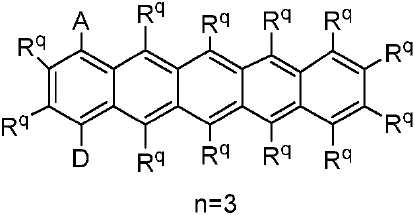
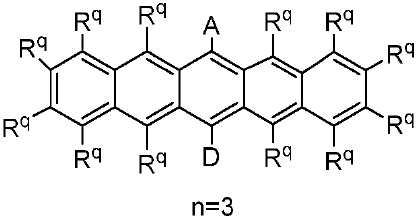
R 각각은 -H, 알킬, 아릴 또는 헤테로아릴(예를 들어, 치환 또는 비치환된 C1-C20 또는 심지어는 C1-C10 알킬)로 이루어진 군으로부터 독립적으로 선택된다. - 제 1 항에 있어서, 상기 고리 시스템(Q)은 하기 화합물로 이루어진 군으로부터 선택되는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종:
상기 식에서, Rq 치환기들 중 적어도 하나는 공여체(D) 모이어티이고, Rq 치환기들 중 적어도 하나는 수용체(A) 모이어티이고;
나머지 Rq 치환기는 각각의 경우에 대해 독립적으로는 -H, 환형일 수 있고 불포화될 수 있는 치환 또는 비치환된 1차, 2차 또는 3차 알킬(예를 들어, C1-C10 또는 심지어는 C1-C4); 치환 또는 비치환된 아릴 또는 헤테로아릴, -CF3, -OMe, -SF5, -NO2, 할로, 아릴, 아릴 하이드록시, 아미노, 알콕시, 알킬티오, 카르복시, 시아노, 티오, 포르밀, 에스테르, 아실, 티오아실, 아미도, 설폰아미도, 카바메이트, 산화포스핀 및 황화포스핀으로 이루어진 군으로부터 선택되고;
n은 1 이상이다. - 제 12 항에 있어서, 상기 n은 1 내지 10의 범위인 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 12 항에 있어서, 상기 플루오렌 상의 R 치환기는 -H, 알킬, 아릴 또는 헤테로아릴(예를 들어, 치환 또는 비치환된 C1-C20 또는 심지어는 C1-C10)로 이루어진 군으로부터 독립적으로 선택되는 것을 특징으로 하는 유기 열 활성화 지연 형광(TADF) 종.
- 제 1 항 내지 제 15 항 중 어느 한 항에 따른 유기 열 활성화 지연 형광(TADF) 종을 에미터 물질(emitter material)로서 포함하는 발광 장치.
- 제 16 항에 있어서, 상기 발광 장치는 유기 발광 다이오드(OLED)인 것을 특징으로 하는 발광 장치.
- 제 16 항에 있어서, 상기 발광 장치는 발광 전기화학 전지(LEEC)인 것을 특징으로 하는 발광 장치.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB1523037.8 | 2015-12-29 | ||
| GBGB1523037.8A GB201523037D0 (en) | 2015-12-29 | 2015-12-29 | Light emitting compounds |
| PCT/GB2016/053858 WO2017115068A1 (en) | 2015-12-29 | 2016-12-08 | Light emitting compounds |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20180098651A true KR20180098651A (ko) | 2018-09-04 |
Family
ID=55359160
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187021898A Ceased KR20180098651A (ko) | 2015-12-29 | 2016-12-08 | 발광 화합물 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20190027693A1 (ko) |
| EP (1) | EP3398217A1 (ko) |
| JP (1) | JP2019509977A (ko) |
| KR (1) | KR20180098651A (ko) |
| CN (1) | CN108780852B (ko) |
| GB (1) | GB201523037D0 (ko) |
| WO (1) | WO2017115068A1 (ko) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11844274B2 (en) | 2019-04-05 | 2023-12-12 | Samsung Display Co., Ltd. | Organic electroluminescence device and compound for organic electroluminescence device |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10680188B2 (en) | 2016-11-11 | 2020-06-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11910699B2 (en) * | 2017-08-10 | 2024-02-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| GB201717193D0 (en) * | 2017-10-19 | 2017-12-06 | Univ Durham | Thermally activated delayed fluorescence molecules, materials comprising said molecules, and devices comprising said materials |
| CN108864068B (zh) * | 2018-07-27 | 2021-12-28 | 武汉天马微电子有限公司 | 一种化合物以及有机发光显示装置 |
| CN109456276A (zh) | 2018-10-22 | 2019-03-12 | 武汉华星光电半导体显示技术有限公司 | 深红光热活化延迟荧光材料及其合成方法、电致发光器件 |
| CN109503508A (zh) * | 2018-11-15 | 2019-03-22 | 武汉华星光电半导体显示技术有限公司 | 绿光热活化延迟荧光材料及其合成方法、电致发光器件 |
| CN111303009B (zh) * | 2018-12-12 | 2022-02-15 | 华中科技大学 | 一种具有高效率、低滚降的蒽基深蓝光有机电致发光材料 |
| CN110054643B (zh) * | 2019-04-25 | 2021-10-15 | 上海天马有机发光显示技术有限公司 | 一种化合物、发光材料、有机发光显示面板及有机发光显示装置 |
| US20260036466A1 (en) * | 2022-07-26 | 2026-02-05 | Mississippi State University | Methods and systems for integrating-sphere-assisted resonance synchronous (isars) spectroscopy |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3008897B2 (ja) * | 1997-07-14 | 2000-02-14 | 日本電気株式会社 | 有機エレクトロルミネッセンス素子 |
| JPH11111458A (ja) * | 1997-09-29 | 1999-04-23 | Toyo Ink Mfg Co Ltd | 有機エレクトロルミネッセンス素子材料およびそれを使用した有機エレクトロルミネッセンス素子 |
| JP3633236B2 (ja) * | 1997-10-06 | 2005-03-30 | 東洋インキ製造株式会社 | 有機エレクトロルミネッセンス素子材料およびそれを使用した有機エレクトロルミネッセンス素子 |
| JP4404473B2 (ja) * | 2000-12-25 | 2010-01-27 | 富士フイルム株式会社 | 新規含窒素へテロ環化合物、発光素子材料およびそれらを使用した発光素子 |
| US20070141393A1 (en) * | 2005-12-21 | 2007-06-21 | Eastman Kodak Company | Amino anthracene compounds in OLED devices |
| KR100852328B1 (ko) * | 2006-03-15 | 2008-08-14 | 주식회사 엘지화학 | 신규한 안트라센 유도체, 이의 제조방법 및 이를 이용한유기 전기 발광 소자 |
| JP5624270B2 (ja) * | 2007-09-18 | 2014-11-12 | ユー・ディー・シー アイルランド リミテッド | 有機電界発光素子 |
| KR100989815B1 (ko) * | 2008-03-20 | 2010-10-29 | 다우어드밴스드디스플레이머티리얼 유한회사 | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 |
| KR20130020503A (ko) * | 2011-08-17 | 2013-02-27 | (주)씨에스엘쏠라 | 유기발광화합물 및 이를 이용한 유기 광소자 |
| JP2013179198A (ja) * | 2012-02-29 | 2013-09-09 | Toyo Ink Sc Holdings Co Ltd | 有機エレクトロルミネッセンス素子用材料およびその用途 |
| TWI638472B (zh) * | 2012-08-03 | 2018-10-11 | 日商半導體能源研究所股份有限公司 | 發光元件 |
| TWI661589B (zh) * | 2013-03-29 | 2019-06-01 | 日商九州有機光材股份有限公司 | 有機電致發光元件 |
| WO2014166585A1 (de) * | 2013-04-08 | 2014-10-16 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung |
| WO2015029964A1 (ja) * | 2013-08-30 | 2015-03-05 | コニカミノルタ株式会社 | 有機エレクトロルミネッセンス素子、発光材料、発光性薄膜、表示装置及び照明装置 |
| KR101502316B1 (ko) * | 2014-04-18 | 2015-03-13 | 롬엔드하스전자재료코리아유한회사 | 복수종의 호스트 재료 및 이를 포함하는 유기 전계 발광 소자 |
| KR20150126755A (ko) * | 2014-05-02 | 2015-11-13 | 삼성디스플레이 주식회사 | 유기 발광 소자 |
| WO2016017514A1 (ja) * | 2014-07-31 | 2016-02-04 | コニカミノルタ株式会社 | 有機エレクトロルミネッセンス素子、発光性薄膜、表示装置及び照明装置 |
| JP6674542B2 (ja) * | 2015-07-30 | 2020-04-01 | 四川知本快車創新科技研究院有限公司Sichuan Knowledge Express Institute For Innovative Technologies Co, Ltd. | 供与体と受容体との間に2つの非共役ブリッジを有する光電変換デバイス用の熱活性化遅延蛍光有機分子 |
-
2015
- 2015-12-29 GB GBGB1523037.8A patent/GB201523037D0/en not_active Ceased
-
2016
- 2016-12-08 JP JP2018534633A patent/JP2019509977A/ja active Pending
- 2016-12-08 CN CN201680077271.4A patent/CN108780852B/zh not_active Expired - Fee Related
- 2016-12-08 EP EP16812793.4A patent/EP3398217A1/en not_active Withdrawn
- 2016-12-08 WO PCT/GB2016/053858 patent/WO2017115068A1/en not_active Ceased
- 2016-12-08 US US16/067,390 patent/US20190027693A1/en not_active Abandoned
- 2016-12-08 KR KR1020187021898A patent/KR20180098651A/ko not_active Ceased
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11844274B2 (en) | 2019-04-05 | 2023-12-12 | Samsung Display Co., Ltd. | Organic electroluminescence device and compound for organic electroluminescence device |
Also Published As
| Publication number | Publication date |
|---|---|
| US20190027693A1 (en) | 2019-01-24 |
| WO2017115068A1 (en) | 2017-07-06 |
| CN108780852A (zh) | 2018-11-09 |
| JP2019509977A (ja) | 2019-04-11 |
| CN108780852B (zh) | 2020-12-15 |
| EP3398217A1 (en) | 2018-11-07 |
| GB201523037D0 (en) | 2016-02-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20180098651A (ko) | 발광 화합물 | |
| Cui et al. | Organoboron compounds with an 8-hydroxyquinolato chelate and its derivatives: substituent effects on structures and luminescence | |
| JP6861644B2 (ja) | 発光デバイス及び化合物 | |
| KR102082528B1 (ko) | 유기 발광 소자 그리고 그것에 사용하는 발광 재료 및 화합물 | |
| Zeng et al. | Cationic Bis‐cyclometallated Iridium (iii) Phenanthroline Complexes with Pendant Fluorenyl Substituents: Synthesis, Redox, Photophysical Properties and Light‐Emitting Cells | |
| Slodek et al. | Fluorene vs carbazole substituent at quinoline core toward organic electronics | |
| Zhang et al. | New platinum (II) one-armed Schiff base complexes for blue and orange PHOLEDs applications | |
| Slodek et al. | Does the length matter?-Synthesis, photophysical, and theoretical study of novel quinolines based on carbazoles with different length of alkyl chain | |
| Skardziute et al. | Synthesis and optical properties of the isomeric pyrimidine and carbazole derivatives: effects of polar substituents and linking topology | |
| Li et al. | Effects of electron affinity and steric hindrance of the trifluoromethyl group on the π‐bridge in designing blue thermally activated delayed fluorescence emitters | |
| Yang et al. | Charged dinuclear Cu (I) complexes for solution-processed single-emitter warm white organic light-emitting devices | |
| Sun et al. | Multi-substituted dibenzo [a, c] phenazine derivatives as solution-processable thermally activated delayed fluorescence materials for orange–red organic light-emitting diodes | |
| Karthik et al. | Synthesis, characterization and electroluminescence of carbazole-benzimidazole hybrids with thiophene/phenyl linker | |
| Patil et al. | Universal blue emitters for high efficiency thermally activated delayed fluorescence and fluorescent organic light-emitting diodes | |
| Arsenyan et al. | Dual versus normal TADF of pyridines ornamented with multiple donor moieties and their performance in OLEDs | |
| Shan et al. | Manipulating the AIE and low-temperature phosphorescence properties of o-carborane-imidazole derivatives via fine tuning their structural features | |
| Gupta et al. | Deep-red to NIR solution-processed OLEDs of donor-decorated quinoxaline-based TADF aggregates | |
| Zhang et al. | An efficient blue thermally activated delayed fluorescence material based on 4-fluorocyanobenzene derivative for organic light-emitting diodes | |
| Kang et al. | Effective exciplex host for solution-processed narrowband blue TADF-OLEDs using a 9-(dibenzo [b, d] thiophen-2-yl)-9H-carbazole analogue with an adamantane substituent | |
| Zhong et al. | Synthesis and luminescence properties of polymeric complexes of Cu (II), Zn (II) and Al (III) with functionalized polybenzimidazole containing 8-hydroxyquinoline side group | |
| Kumar et al. | Facile color tuning of thermally activated delayed fluorescence by substituted ortho-carbazole-appended triarylboron emitters | |
| Mubarok et al. | Boron‐and nitrogen‐embedded blue multi‐resonance emitters with low triplet energy | |
| Tang et al. | Achieving high efficiency at high luminance in fluorescent organic light‐emitting diodes through triplet–triplet fusion based on phenanthroimidazole‐benzothiadiazole derivatives | |
| Xiao et al. | Synthesis and optoelectronic properties of a series of novel spirobifluorene derivatives starting from the readily available reagent 4, 4′-bisalkylated biphenyl | |
| Tian et al. | Synthesis and thermal, electrochemical, photophysical properties of novel symmetric/asymmetric indolo [2, 3-a] carbazole derivatives bearing different aryl substituents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20180727 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20211116 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20240408 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20240613 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20240408 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |