KR20180098301A - 아로마틱 콤플렉스에서의 톨루엔 메틸화 방법 및 장치 - Google Patents
아로마틱 콤플렉스에서의 톨루엔 메틸화 방법 및 장치 Download PDFInfo
- Publication number
- KR20180098301A KR20180098301A KR1020187020239A KR20187020239A KR20180098301A KR 20180098301 A KR20180098301 A KR 20180098301A KR 1020187020239 A KR1020187020239 A KR 1020187020239A KR 20187020239 A KR20187020239 A KR 20187020239A KR 20180098301 A KR20180098301 A KR 20180098301A
- Authority
- KR
- South Korea
- Prior art keywords
- xylene
- aromatic
- stream
- transalkylation
- benzene
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 title claims abstract description 213
- 238000000034 method Methods 0.000 title claims abstract description 64
- 125000003118 aryl group Chemical group 0.000 title claims abstract description 58
- 230000011987 methylation Effects 0.000 title claims abstract description 35
- 238000007069 methylation reaction Methods 0.000 title claims abstract description 35
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims abstract description 153
- URLKBWYHVLBVBO-UHFFFAOYSA-N Para-Xylene Chemical group CC1=CC=C(C)C=C1 URLKBWYHVLBVBO-UHFFFAOYSA-N 0.000 claims abstract description 96
- 238000010555 transalkylation reaction Methods 0.000 claims abstract description 57
- 238000004519 manufacturing process Methods 0.000 claims abstract description 28
- 239000006227 byproduct Substances 0.000 claims abstract description 15
- 238000004064 recycling Methods 0.000 claims abstract description 6
- 239000000047 product Substances 0.000 claims description 46
- 238000006317 isomerization reaction Methods 0.000 claims description 33
- 239000008096 xylene Substances 0.000 claims description 28
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 27
- 238000009835 boiling Methods 0.000 claims description 27
- 239000003054 catalyst Substances 0.000 claims description 26
- 239000000203 mixture Substances 0.000 claims description 21
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N Ethylbenzene Chemical compound CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 claims description 18
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 18
- 229910021536 Zeolite Inorganic materials 0.000 claims description 17
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 claims description 17
- 239000010457 zeolite Substances 0.000 claims description 17
- 229910052751 metal Inorganic materials 0.000 claims description 11
- 239000002184 metal Substances 0.000 claims description 11
- 238000005984 hydrogenation reaction Methods 0.000 claims description 10
- 229910052799 carbon Inorganic materials 0.000 claims description 7
- 238000000926 separation method Methods 0.000 claims description 7
- 150000001491 aromatic compounds Chemical class 0.000 claims description 6
- 230000020335 dealkylation Effects 0.000 claims description 6
- 238000006900 dealkylation reaction Methods 0.000 claims description 6
- 238000006243 chemical reaction Methods 0.000 claims description 5
- GNFTZDOKVXKIBK-UHFFFAOYSA-N 3-(2-methoxyethoxy)benzohydrazide Chemical compound COCCOC1=CC=CC(C(=O)NN)=C1 GNFTZDOKVXKIBK-UHFFFAOYSA-N 0.000 claims description 4
- 239000007791 liquid phase Substances 0.000 claims description 3
- 239000012808 vapor phase Substances 0.000 claims description 3
- 238000002360 preparation method Methods 0.000 claims description 2
- 238000002407 reforming Methods 0.000 description 28
- 229930195733 hydrocarbon Natural products 0.000 description 21
- 150000002430 hydrocarbons Chemical class 0.000 description 21
- 239000004215 Carbon black (E152) Substances 0.000 description 12
- 239000003921 oil Substances 0.000 description 12
- 150000003738 xylenes Chemical class 0.000 description 12
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 8
- 230000008901 benefit Effects 0.000 description 8
- 150000001336 alkenes Chemical class 0.000 description 6
- IVSZLXZYQVIEFR-UHFFFAOYSA-N m-xylene Chemical group CC1=CC=CC(C)=C1 IVSZLXZYQVIEFR-UHFFFAOYSA-N 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- OFBQJSOFQDEBGM-UHFFFAOYSA-N n-pentane Natural products CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 6
- 238000001179 sorption measurement Methods 0.000 description 6
- 238000005194 fractionation Methods 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- -1 For example Chemical class 0.000 description 4
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 4
- 238000001833 catalytic reforming Methods 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 238000004821 distillation Methods 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 4
- QMMFVYPAHWMCMS-UHFFFAOYSA-N Dimethyl sulfide Chemical compound CSC QMMFVYPAHWMCMS-UHFFFAOYSA-N 0.000 description 3
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 3
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 3
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 3
- 125000000217 alkyl group Chemical group 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 239000003208 petroleum Substances 0.000 description 3
- 239000012071 phase Substances 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 239000011593 sulfur Substances 0.000 description 3
- 150000003464 sulfur compounds Chemical class 0.000 description 3
- CXWXQJXEFPUFDZ-UHFFFAOYSA-N tetralin Chemical compound C1=CC=C2CCCCC2=C1 CXWXQJXEFPUFDZ-UHFFFAOYSA-N 0.000 description 3
- DSNHSQKRULAAEI-UHFFFAOYSA-N 1,4-Diethylbenzene Chemical compound CCC1=CC=C(CC)C=C1 DSNHSQKRULAAEI-UHFFFAOYSA-N 0.000 description 2
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical compound C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 2
- JRLPEMVDPFPYPJ-UHFFFAOYSA-N 1-ethyl-4-methylbenzene Chemical compound CCC1=CC=C(C)C=C1 JRLPEMVDPFPYPJ-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 2
- 239000001273 butane Substances 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 239000003317 industrial substance Substances 0.000 description 2
- 229910052809 inorganic oxide Inorganic materials 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 239000012188 paraffin wax Substances 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 238000000197 pyrolysis Methods 0.000 description 2
- SPPWGCYEYAMHDT-UHFFFAOYSA-N 1,4-di(propan-2-yl)benzene Chemical compound CC(C)C1=CC=C(C(C)C)C=C1 SPPWGCYEYAMHDT-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 1
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001345 alkine derivatives Chemical class 0.000 description 1
- 239000000987 azo dye Substances 0.000 description 1
- 150000001555 benzenes Chemical class 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000004523 catalytic cracking Methods 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 150000001924 cycloalkanes Chemical class 0.000 description 1
- 238000006356 dehydrogenation reaction Methods 0.000 description 1
- QDOXWKRWXJOMAK-UHFFFAOYSA-N dichromium trioxide Chemical compound O=[Cr]O[Cr]=O QDOXWKRWXJOMAK-UHFFFAOYSA-N 0.000 description 1
- 150000001993 dienes Chemical class 0.000 description 1
- 230000003467 diminishing effect Effects 0.000 description 1
- 238000007323 disproportionation reaction Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000000895 extractive distillation Methods 0.000 description 1
- 239000000295 fuel oil Substances 0.000 description 1
- 229910001385 heavy metal Inorganic materials 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 229910017464 nitrogen compound Inorganic materials 0.000 description 1
- 150000002830 nitrogen compounds Chemical class 0.000 description 1
- 229910000510 noble metal Inorganic materials 0.000 description 1
- 229910052762 osmium Inorganic materials 0.000 description 1
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 238000005504 petroleum refining Methods 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- LGRFSURHDFAFJT-UHFFFAOYSA-N phthalic anhydride Chemical class C1=CC=C2C(=O)OC(=O)C2=C1 LGRFSURHDFAFJT-UHFFFAOYSA-N 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 239000010948 rhodium Substances 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 238000004230 steam cracking Methods 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C6/00—Preparation of hydrocarbons from hydrocarbons containing a different number of carbon atoms by redistribution reactions
- C07C6/08—Preparation of hydrocarbons from hydrocarbons containing a different number of carbon atoms by redistribution reactions by conversion at a saturated carbon-to-carbon bond
- C07C6/12—Preparation of hydrocarbons from hydrocarbons containing a different number of carbon atoms by redistribution reactions by conversion at a saturated carbon-to-carbon bond of exclusively hydrocarbons containing a six-membered aromatic ring
- C07C6/126—Preparation of hydrocarbons from hydrocarbons containing a different number of carbon atoms by redistribution reactions by conversion at a saturated carbon-to-carbon bond of exclusively hydrocarbons containing a six-membered aromatic ring of more than one hydrocarbon
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/02—Monocyclic hydrocarbons
- C07C15/04—Benzene
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C15/00—Cyclic hydrocarbons containing only six-membered aromatic rings as cyclic parts
- C07C15/02—Monocyclic hydrocarbons
- C07C15/067—C8H10 hydrocarbons
- C07C15/08—Xylenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2/00—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms
- C07C2/86—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms by condensation between a hydrocarbon and a non-hydrocarbon
- C07C2/862—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms by condensation between a hydrocarbon and a non-hydrocarbon the non-hydrocarbon contains only oxygen as hetero-atoms
- C07C2/864—Preparation of hydrocarbons from hydrocarbons containing a smaller number of carbon atoms by condensation between a hydrocarbon and a non-hydrocarbon the non-hydrocarbon contains only oxygen as hetero-atoms the non-hydrocarbon is an alcohol
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C4/00—Preparation of hydrocarbons from hydrocarbons containing a larger number of carbon atoms
- C07C4/08—Preparation of hydrocarbons from hydrocarbons containing a larger number of carbon atoms by splitting-off an aliphatic or cycloaliphatic part from the molecule
- C07C4/12—Preparation of hydrocarbons from hydrocarbons containing a larger number of carbon atoms by splitting-off an aliphatic or cycloaliphatic part from the molecule from hydrocarbons containing a six-membered aromatic ring, e.g. propyltoluene to vinyltoluene
- C07C4/14—Preparation of hydrocarbons from hydrocarbons containing a larger number of carbon atoms by splitting-off an aliphatic or cycloaliphatic part from the molecule from hydrocarbons containing a six-membered aromatic ring, e.g. propyltoluene to vinyltoluene splitting taking place at an aromatic-aliphatic bond
- C07C4/18—Catalytic processes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C5/00—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms
- C07C5/22—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms by isomerisation
- C07C5/2206—Catalytic processes not covered by C07C5/23 - C07C5/31
- C07C5/222—Catalytic processes not covered by C07C5/23 - C07C5/31 with crystalline alumino-silicates, e.g. molecular sieves
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C5/00—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms
- C07C5/22—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms by isomerisation
- C07C5/27—Rearrangement of carbon atoms in the hydrocarbon skeleton
- C07C5/2729—Changing the branching point of an open chain or the point of substitution on a ring
- C07C5/2732—Catalytic processes
- C07C5/2737—Catalytic processes with crystalline alumino-silicates, e.g. molecular sieves
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C5/00—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms
- C07C5/22—Preparation of hydrocarbons from hydrocarbons containing the same number of carbon atoms by isomerisation
- C07C5/27—Rearrangement of carbon atoms in the hydrocarbon skeleton
- C07C5/2767—Changing the number of side-chains
- C07C5/277—Catalytic processes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C7/00—Purification; Separation; Use of additives
- C07C7/04—Purification; Separation; Use of additives by distillation
- C07C7/05—Purification; Separation; Use of additives by distillation with the aid of auxiliary compounds
- C07C7/08—Purification; Separation; Use of additives by distillation with the aid of auxiliary compounds by extractive distillation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C7/00—Purification; Separation; Use of additives
- C07C7/12—Purification; Separation; Use of additives by adsorption, i.e. purification or separation of hydrocarbons with the aid of solids, e.g. with ion-exchangers
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G29/00—Refining of hydrocarbon oils, in the absence of hydrogen, with other chemicals
- C10G29/20—Organic compounds not containing metal atoms
- C10G29/205—Organic compounds not containing metal atoms by reaction with hydrocarbons added to the hydrocarbon oil
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G69/00—Treatment of hydrocarbon oils by at least one hydrotreatment process and at least one other conversion process
- C10G69/02—Treatment of hydrocarbon oils by at least one hydrotreatment process and at least one other conversion process plural serial stages only
- C10G69/12—Treatment of hydrocarbon oils by at least one hydrotreatment process and at least one other conversion process plural serial stages only including at least one polymerisation or alkylation step
- C10G69/123—Treatment of hydrocarbon oils by at least one hydrotreatment process and at least one other conversion process plural serial stages only including at least one polymerisation or alkylation step alkylation
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G2400/00—Products obtained by processes covered by groups C10G9/00 - C10G69/14
- C10G2400/30—Aromatics
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Water Supply & Treatment (AREA)
- Crystallography & Structural Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
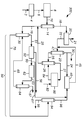
도 2는 톨루엔 메틸화 구역이 통합되어 있는 아로마틱 콤플렉스의 다른 실시양태를 도시한 것이다.
도 3은 톨루엔 메틸화 구역이 통합되어 있는 아로마틱 콤플렉스의 또 다른 실시양태를 도시한 것이다.
도 4는 톨루엔 메틸화 구역이 통합되어 있는 아로마틱 콤플렉스의 다른 실시양태를 도시한 것이다.
몇몇 도면을 통해 상응하는 도면 부호는 상응하는 부품을 지시한다. 당업자라면 도면의 요소들은 간단하고 간결하게 도시된 것이고 반드시 실제 척도로 도시된 것이 아님을 이해할 것이다. 예컨대, 본 개시내용의 여러 실시양태들의 이해 증진을 돕기 위하여 도면에서 일부 요소들의 치수는 다른 요소들에 비해 과장되어 있을 수 있다. 또한, 상업적으로 실행가능한 실시양태에 유용하거나 필요한 통상적이고 잘 알려진 요소들은 본 개시내용의 여러 실시양태들의 도시가 덜 방해되도록 흔히 도시되지 않는다.
Claims (10)
- 벤젠 부산물을 포함하지 않는 파라크실렌의 제조 방법으로서,
a) 벤젠을 함유하는 더 경질의 방향족 스트림 및 C9-C10 방향족 화합물을 함유하는 더 중질의 방향족 스트림을 트랜스알킬화 구역에 통과시키는 단계;
b) 상기 더 경질의 방향족 스트림 및 더 중질의 방향족 스트림을 트랜스알킬화 구역에서 제1 촉매의 존재를 포함하는 트랜스알킬화 조건에 두어, 더 높은 농도의 톨루엔 내지 C8 방향족을 갖는 트랜스알킬화 생성물 스트림을 제공하는 단계;
c) 상기 트랜스알킬화 생성물 스트림으로부터 벤젠을 포함하는 제1 비점 유분, 톨루엔을 포함하는 제2 비점 유분, C8 방향족을 포함하는 제3 비점 유분, 및 C9+ 방향족을 포함하는 제4 비점 유분을 분류에 의해 분리하는 단계;
d) 트랜스알킬화 생성물 스트림으로부터 벤젠의 적어도 일부를 트랜스알킬화 구역으로 다시 재순환시키는 단계;
e) 단계 c, g 및 i로부터의 제2 비점 유분의 적어도 일부 및 메탄올 스트림을 톨루엔 메틸화 조건 하에 운전되는 톨루엔 메틸화 구역에 통과시켜 톨루엔 메틸화 생성물 스트림을 생성하는 단계;
f) 톨루엔 메틸화 생성물 스트림으로부터 단계 c에 기재한 동일한 유분을 분류에 의해 분리하는 단계;
g) 단계 c, g 및 i의 C8 방향족을 포함하는 제3 비점 유분의 적어도 일부를 분리 구역에 두어, 파라크실렌 생성물을 선택적으로 제거하고 C8 방향족의 비평형 혼합물을 제공하는 단계;
h) C8 방향족의 비평형 혼합물의 일부를 제2 촉매의 존재를 포함하는 크실렌 이성화 조건에 두어 이성화 생성물을 제공하는 단계;
i) 단계 g로부터의 C8 방향족의 비평형 혼합물의 일부를 트랜스알킬화 구역에 통과시키는 단계; 및
j) 이성화 생성물 스트림으로부터 단계 c에 기재한 동일한 유분을 분류에 의해 분리하는 단계
를 포함하는, 벤젠 부산물을 포함하지 않는 파라크실렌의 제조 방법. - 제1항에 있어서, 트랜스알킬화 조건은 320℃ 내지 440℃의 온도를 포함하는 것인 제조 방법.
- 제1항에 있어서, 제1 촉매는 트랜스알킬화에 적합한 하나 이상의 제올라이트 성분, 탈알킬화에 적합한 하나 이상의 제올라이트 성분 및 수소첨가에 적합한 하나 이상의 금속 성분을 포함하는 것인 제조 방법.
- 제1항에 있어서, 톨루엔 메틸화 생성물 스트림은 전체 크실렌에 대한 파라크실렌의 비가 0.2 이상, 또는 바람직하게는 0.5 이상, 또는 더 바람직하게는 0.8∼0.95인 것인 제조 방법.
- 제1항에 있어서, 이성화 조건은 240℃ 내지 440℃의 온도를 포함하는 것인 방법.
- 제1항에 있어서, 제2 촉매는 크실렌 이성화에 적합한 하나 이상의 제올라이트 성분, 에틸벤젠 전환에 적합한 하나 이상의 제올라이트 성분, 및 수소첨가에 적합한 하나 이상의 금속 성분을 포함하는 것인 제조 방법.
- 제1항에 있어서, 이성화 공정은 증기상으로 실시되는 것인 제조 방법.
- 제1항에 있어서, 이성화 공정은 탈알킬화에 의해 에틸벤젠을 전환하여 벤젠을 생성하는 것인 제조 방법.
- 제1항에 있어서, 이성화 공정은 이성화에 의해 에틸벤젠을 전환하여 크실렌을 생성하는 것인 제조 방법.
- 제1항에 있어서, 이성화 공정은 액상으로 실시되는 것인 제조 방법.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201562267966P | 2015-12-16 | 2015-12-16 | |
| US62/267,966 | 2015-12-16 | ||
| PCT/US2016/064306 WO2017105848A1 (en) | 2015-12-16 | 2016-12-01 | Processes and apparatuses for toluene methylation in an aromatics complex |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20180098301A true KR20180098301A (ko) | 2018-09-03 |
| KR102159219B1 KR102159219B1 (ko) | 2020-09-23 |
Family
ID=59057401
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187020239A Active KR102159219B1 (ko) | 2015-12-16 | 2016-12-01 | 아로마틱 콤플렉스에서의 톨루엔 메틸화 방법 및 장치 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US10239802B2 (ko) |
| KR (1) | KR102159219B1 (ko) |
| CN (1) | CN108430958B (ko) |
| WO (1) | WO2017105848A1 (ko) |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11208365B2 (en) | 2016-12-20 | 2021-12-28 | Uop Llc | Processes and apparatuses for methylation of aromatics in an aromatics complex |
| CN107473918A (zh) * | 2017-07-14 | 2017-12-15 | 同济大学 | 煤基原料生产对二甲苯、邻二甲苯和偏三甲苯的方法 |
| US10308571B2 (en) | 2017-10-11 | 2019-06-04 | Uop Llc | Process for minimizing benzene, toluene, and a recycle loop in a zero benzene aromatics complex |
| US10392316B2 (en) * | 2017-10-11 | 2019-08-27 | Uop Llc | Process for minimizing benzene, toluene, and a recycle loop in a zero benzene aromatics complex |
| US11130719B2 (en) * | 2017-12-05 | 2021-09-28 | Uop Llc | Processes and apparatuses for methylation of aromatics in an aromatics complex |
| US10556847B2 (en) * | 2017-12-06 | 2020-02-11 | Uop Llc | Processes and apparatuses for methylation of aromatics in an aromatics complex |
| US11130720B2 (en) | 2018-03-23 | 2021-09-28 | Uop Llc | Processes for methylation of aromatics in an aromatics complex |
| US20200354293A1 (en) * | 2019-05-10 | 2020-11-12 | Uop Llc | Process and apparatus for dual feed para-xylene separation |
| US10626067B1 (en) | 2019-05-10 | 2020-04-21 | Uop Llc | Processes for separating para-xylene from toluene |
| US20210206704A1 (en) * | 2020-01-06 | 2021-07-08 | Uop Llc | Oxygenate removal for para-xylene purification via adsorption separation |
| US12065401B1 (en) | 2023-03-14 | 2024-08-20 | Saudi Arabian Oil Company | Integrated processes and systems for producing para-xylenes |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110178354A1 (en) * | 2010-01-19 | 2011-07-21 | Antoine Negiz | Aromatic aklylating agent and an aromatic production apparatus |
| US20120149958A1 (en) * | 2010-12-10 | 2012-06-14 | Ellrich Justin M | Method and Apparatus for Obtaining Aromatics from Diverse Feedstock |
| KR20180042856A (ko) * | 2015-09-10 | 2018-04-26 | 유오피 엘엘씨 | 아로마틱스 콤플렉스에서의 톨루엔 메틸화 방법 및 장치 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6740788B1 (en) * | 2002-12-19 | 2004-05-25 | Uop Llc | Integrated process for aromatics production |
| FR2883283B1 (fr) | 2005-03-16 | 2007-05-18 | Inst Francais Du Petrole | Procede de production combinee de paraxylene et de benzene de productivite amelioree |
| US7405335B1 (en) * | 2005-06-30 | 2008-07-29 | Uop Llc | Integrated process for producing xylenes and high purity benzene |
| KR100821428B1 (ko) | 2005-12-26 | 2008-04-11 | 삼성토탈 주식회사 | 탈알킬화 반응을 사용하여 파라-크실렌의 생산성이 향상된방향족 화합물 처리공정 |
| CN101993333B (zh) | 2009-08-31 | 2014-01-22 | 中国石油化工股份有限公司 | 芳烃生产中增产对二甲苯的组合方法 |
| CN102040461B (zh) | 2009-10-13 | 2014-07-02 | 中国石油化工股份有限公司 | 用于芳烃生产中增产对二甲苯的组合方法 |
| CN102190553B (zh) | 2010-03-03 | 2014-03-26 | 中国石油化工股份有限公司 | 生产苯和对二甲苯的芳烃烷基转移方法 |
| CN103596907B (zh) * | 2011-06-10 | 2016-04-27 | 埃克森美孚化学专利公司 | 对二甲苯的生产 |
| SG11201502727YA (en) * | 2012-10-10 | 2015-05-28 | Gtc Technology Us Llc | Processes and systems for obtaining aromatics from catalytic cracking hydrocarbons |
| WO2015142454A1 (en) | 2014-03-20 | 2015-09-24 | Exxonmobil Chemical Patents Inc. | Paraxylene separation process |
| US20150376087A1 (en) * | 2014-06-30 | 2015-12-31 | Exxonmobil Chemical Patents Inc. | Process for the Production of Xylenes |
-
2016
- 2016-12-01 WO PCT/US2016/064306 patent/WO2017105848A1/en not_active Ceased
- 2016-12-01 KR KR1020187020239A patent/KR102159219B1/ko active Active
- 2016-12-01 CN CN201680073295.2A patent/CN108430958B/zh active Active
-
2018
- 2018-05-24 US US15/989,084 patent/US10239802B2/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110178354A1 (en) * | 2010-01-19 | 2011-07-21 | Antoine Negiz | Aromatic aklylating agent and an aromatic production apparatus |
| US20120149958A1 (en) * | 2010-12-10 | 2012-06-14 | Ellrich Justin M | Method and Apparatus for Obtaining Aromatics from Diverse Feedstock |
| KR20130103766A (ko) * | 2010-12-10 | 2013-09-24 | 엑손모빌 케미칼 패턴츠 인코포레이티드 | 각종 공급원료로부터 방향족 화합물을 얻기 위한 방법 및 장치 |
| KR20180042856A (ko) * | 2015-09-10 | 2018-04-26 | 유오피 엘엘씨 | 아로마틱스 콤플렉스에서의 톨루엔 메틸화 방법 및 장치 |
Also Published As
| Publication number | Publication date |
|---|---|
| US10239802B2 (en) | 2019-03-26 |
| CN108430958A (zh) | 2018-08-21 |
| KR102159219B1 (ko) | 2020-09-23 |
| US20180265429A1 (en) | 2018-09-20 |
| WO2017105848A1 (en) | 2017-06-22 |
| CN108430958B (zh) | 2021-01-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10239802B2 (en) | Processes and apparatuses for toluene methylation in an aromatics complex | |
| TWI666194B (zh) | 用於芳香族複合設備中之芳香族甲基化之方法及裝置 | |
| US10508066B2 (en) | Methods and systems of upgrading heavy aromatics stream to petrochemical feedstock | |
| US10876054B2 (en) | Olefin and BTX production using aliphatic cracking reactor | |
| US9434894B2 (en) | Process for converting FCC naphtha into aromatics | |
| US20170073285A1 (en) | Processes and apparatuses for toluene methylation in an aromatics complex | |
| WO2008094255A1 (en) | Integrated apparatus for aromatics production | |
| CN108368003B (zh) | 在芳族化合物联合装置中用于烯烃饱和的方法和装置 | |
| JP7372346B2 (ja) | 二重供給パラキシレン分離のためのプロセス及び装置 | |
| US11066344B2 (en) | Methods and systems of upgrading heavy aromatics stream to petrochemical feedstock | |
| US20210206704A1 (en) | Oxygenate removal for para-xylene purification via adsorption separation | |
| WO2020214872A1 (en) | Methods and systems of upgrading heavy aromatics stream to petrochemical feedstock | |
| WO2021087163A1 (en) | Methods and systems of upgrading heavy aromatics stream to petrochemical feedstock |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0105 | International application |
Patent event date: 20180713 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PA0201 | Request for examination | ||
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20190823 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20200625 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20200917 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20200918 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration | ||
| PR1001 | Payment of annual fee |
Payment date: 20230906 Start annual number: 4 End annual number: 4 |
|
| PR1001 | Payment of annual fee |
Payment date: 20240909 Start annual number: 5 End annual number: 5 |