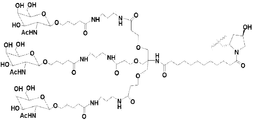
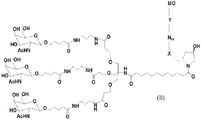
KR20170033439A - 대사 장애에 대한 마이크로rna의 표적화 - Google Patents
대사 장애에 대한 마이크로rna의 표적화 Download PDFInfo
- Publication number
- KR20170033439A KR20170033439A KR1020177005873A KR20177005873A KR20170033439A KR 20170033439 A KR20170033439 A KR 20170033439A KR 1020177005873 A KR1020177005873 A KR 1020177005873A KR 20177005873 A KR20177005873 A KR 20177005873A KR 20170033439 A KR20170033439 A KR 20170033439A
- Authority
- KR
- South Korea
- Prior art keywords
- certain embodiments
- compound
- nucleoside
- mir
- modified
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 0 CC*(C)NCCC1CC(CC*C(C)(C[C@](C*)C23CC2)[C@@](*)[C@]3*=*)C1 Chemical compound CC*(C)NCCC1CC(CC*C(C)(C[C@](C*)C23CC2)[C@@](*)[C@]3*=*)C1 0.000 description 3
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/549—Sugars, nucleosides, nucleotides or nucleic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
- C12N2310/113—Antisense targeting other non-coding nucleic acids, e.g. antagomirs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering nucleic acids [NA]
- C12N2310/141—MicroRNAs, miRNAs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/315—Phosphorothioates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/321—2'-O-R Modification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/322—2'-R Modification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/323—Chemical structure of the sugar modified ring structure
- C12N2310/3231—Chemical structure of the sugar modified ring structure having an additional ring, e.g. LNA, ENA
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/34—Spatial arrangement of the modifications
- C12N2310/345—Spatial arrangement of the modifications having at least two different backbone modifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/34—Spatial arrangement of the modifications
- C12N2310/346—Spatial arrangement of the modifications having a combination of backbone and sugar modifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/50—Physical structure
- C12N2310/51—Physical structure in polymeric form, e.g. multimers, concatemers
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/30—Special therapeutic applications
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Epidemiology (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- General Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Biochemistry (AREA)
- Diabetes (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Biophysics (AREA)
- Physics & Mathematics (AREA)
- Microbiology (AREA)
- Plant Pathology (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Child & Adolescent Psychology (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Gastroenterology & Hepatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
Applications Claiming Priority (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462034739P | 2014-08-07 | 2014-08-07 | |
| US62/034,739 | 2014-08-07 | ||
| US201462062749P | 2014-10-10 | 2014-10-10 | |
| US62/062,749 | 2014-10-10 | ||
| US201562143434P | 2015-04-06 | 2015-04-06 | |
| US62/143,434 | 2015-04-06 | ||
| PCT/US2015/043926 WO2016022753A1 (en) | 2014-08-07 | 2015-08-06 | Targeting micrornas for metabolic disorders |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20170033439A true KR20170033439A (ko) | 2017-03-24 |
Family
ID=53836884
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020177005873A Withdrawn KR20170033439A (ko) | 2014-08-07 | 2015-08-06 | 대사 장애에 대한 마이크로rna의 표적화 |
Country Status (17)
| Country | Link |
|---|---|
| US (4) | US9487783B2 (enExample) |
| EP (1) | EP3177721A1 (enExample) |
| JP (1) | JP2017523790A (enExample) |
| KR (1) | KR20170033439A (enExample) |
| CN (1) | CN106559995A (enExample) |
| AU (1) | AU2015301057A1 (enExample) |
| BR (1) | BR112017001931A2 (enExample) |
| CA (1) | CA2953883A1 (enExample) |
| CL (1) | CL2017000312A1 (enExample) |
| CO (1) | CO2017000950A2 (enExample) |
| IL (1) | IL249819A0 (enExample) |
| MA (1) | MA40463A (enExample) |
| MX (1) | MX2017001493A (enExample) |
| RU (1) | RU2017105342A (enExample) |
| SG (1) | SG11201610877PA (enExample) |
| TW (1) | TW201613949A (enExample) |
| WO (1) | WO2016022753A1 (enExample) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2017105342A (ru) | 2014-08-07 | 2018-09-13 | Регулус Терапьютикс Инк. | НАЦЕЛИВАНИЕ НА микроРНК ПРИ РАССТРОЙСТВАХ ОБМЕНА ВЕЩЕСТВ |
| KR102608220B1 (ko) * | 2016-05-06 | 2023-11-29 | 아이오니스 파마수티컬즈, 인코포레이티드 | Glp-1 수용체 리간드 모이어티 컨쥬게이트된 올리고뉴클레오티드 및 이의 용도 |
| AU2017368050A1 (en) | 2016-11-29 | 2019-06-20 | Puretech Lyt, Inc. | Exosomes for delivery of therapeutic agents |
| KR101869308B1 (ko) * | 2017-02-28 | 2018-06-20 | 대구가톨릭대학교산학협력단 | Srebp-1 디코이 올리고디옥시뉴클레오티드 및 이를 유효성분으로 함유하는 지방간 질환의 예방 또는 치료용 약학 조성물 |
| AU2019209491B2 (en) * | 2018-01-17 | 2022-05-19 | Joslin Diabetes Center, Inc. | Methods for treating diabetes, hepatitis, and/or inflammatory liver disease |
| WO2020010059A1 (en) * | 2018-07-02 | 2020-01-09 | Aptamir Therapeutics, Inc. | Targeted delivery of therapeutic agents to human adipocytes |
| EP3824087A1 (en) * | 2018-07-20 | 2021-05-26 | Regulus Therapeutics Inc. | Methods for oral delivery of oligonucleotides |
| US10844383B1 (en) | 2019-09-18 | 2020-11-24 | Dasman Diabetes Institute | Microrna dyslipidemia inhibitor |
Family Cites Families (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0862439A4 (en) | 1995-11-22 | 2001-01-10 | O Paul O P Ts | LIGANDS FOR INCREASING THE CELLULAR UPtake OF BIOMOLECULES |
| US7491805B2 (en) | 2001-05-18 | 2009-02-17 | Sirna Therapeutics, Inc. | Conjugates and compositions for cellular delivery |
| CA2504694C (en) | 2002-11-05 | 2013-10-01 | Isis Pharmaceuticals, Inc. | Polycyclic sugar surrogate-containing oligomeric compounds and compositions for use in gene modulation |
| EP1648914A4 (en) | 2003-07-31 | 2009-12-16 | Regulus Therapeutics Inc | OLIGOMERIC COMPOUNDS AND COMPOSITIONS USEFUL FOR MODULATING SMALL NON-CODING RNA |
| JP4731324B2 (ja) | 2003-08-28 | 2011-07-20 | 武 今西 | N−o結合性架橋構造型新規人工核酸 |
| WO2006078278A2 (en) | 2004-04-27 | 2006-07-27 | Alnylam Pharmaceuticals, Inc. | Single-stranded and double-stranded oligonucleotides comprising a 2-arylpropyl moiety |
| EP2290071B1 (en) | 2004-05-28 | 2014-12-31 | Asuragen, Inc. | Methods and compositions involving microRNA |
| EP2990410A1 (en) | 2004-08-10 | 2016-03-02 | Alnylam Pharmaceuticals Inc. | Chemically modified oligonucleotides |
| EP2314688B1 (en) | 2004-11-12 | 2014-07-16 | Asuragen, Inc. | Methods and compositions involving miRNA and miRNA inhibitor molecules |
| US20060185027A1 (en) | 2004-12-23 | 2006-08-17 | David Bartel | Systems and methods for identifying miRNA targets and for altering miRNA and target expression |
| AU2006279906B2 (en) | 2005-08-10 | 2012-05-10 | Alnylam Pharmaceuticals, Inc. | Chemically modified oligonucleotides for use in modulating micro RNA and uses thereof |
| WO2007027894A2 (en) | 2005-08-29 | 2007-03-08 | Isis Pharmaceuticals, Inc. | Antisense compounds having enhanced anti-microrna activity |
| EP2388327A1 (en) | 2006-01-27 | 2011-11-23 | Isis Pharmaceuticals, Inc. | Oligomeric compounds and compositions for the use in modulation of micrornas |
| ES2516815T3 (es) | 2006-01-27 | 2014-10-31 | Isis Pharmaceuticals, Inc. | Análogos de ácidos nucleicos bicíclicos modificados en la posición 6 |
| KR101407707B1 (ko) | 2006-04-03 | 2014-06-19 | 산타리스 팔마 에이/에스 | Anti-mirna 안티센스 올리고뉴클레오타이드를 함유하는 약학적 조성물 |
| CA2927045A1 (en) | 2006-10-03 | 2008-04-10 | Muthiah Manoharan | Lipid containing formulations |
| CN101121934B (zh) | 2007-04-11 | 2011-12-07 | 哈尔滨医科大学 | 多靶点miRNA反义核苷酸的制备方法 |
| WO2008150729A2 (en) | 2007-05-30 | 2008-12-11 | Isis Pharmaceuticals, Inc. | N-substituted-aminomethylene bridged bicyclic nucleic acid analogs |
| EP2173760B2 (en) | 2007-06-08 | 2015-11-04 | Isis Pharmaceuticals, Inc. | Carbocyclic bicyclic nucleic acid analogs |
| AU2008272918B2 (en) | 2007-07-05 | 2012-09-13 | Isis Pharmaceuticals, Inc. | 6-disubstituted bicyclic nucleic acid analogs |
| US8288356B2 (en) | 2007-10-04 | 2012-10-16 | Santaris Pharma A/S | MicroRNAs |
| EP3705125B1 (en) | 2007-12-04 | 2023-07-05 | Alnylam Pharmaceuticals, Inc. | Carbohydrate conjugates as delivery agents for oligonucleotides |
| EP2268811A1 (en) | 2008-03-07 | 2011-01-05 | Santaris Pharma A/S | Pharmaceutical compositions for treatment of microrna related diseases |
| AU2010250838B2 (en) * | 2009-05-20 | 2016-01-21 | Eth Zurich | Targeting microRNAs for metabolic disorders |
| SG176716A1 (en) | 2009-06-08 | 2012-01-30 | Miragen Therapeutics | CHEMICAL MODIFICATION MOTIFS FOR miRNA INHIBITORS AND MIMETICS |
| ES2562817T3 (es) | 2010-02-24 | 2016-03-08 | Arrowhead Research Corporation | Composiciones para el suministro dirigido de ARNip |
| WO2011117353A1 (en) * | 2010-03-24 | 2011-09-29 | Mirrx Therapeutics A/S | Bivalent antisense oligonucleotides |
| KR20130132475A (ko) | 2010-12-17 | 2013-12-04 | 애로우헤드 리서치 코오포레이션 | siRNA의 갈락토오스 클러스터-약동학적 조절제 표적 물질 |
| US8877722B2 (en) | 2011-03-25 | 2014-11-04 | Idera Pharmaceuticals, Inc. | Compositions for inhibiting gene expression and uses thereof |
| EP2751270B1 (en) | 2011-08-29 | 2018-08-22 | Ionis Pharmaceuticals, Inc. | Oligomer-conjugate complexes and their use |
| EP3533873A1 (en) * | 2011-09-14 | 2019-09-04 | Translate Bio MA, Inc. | Multimeric oligonucleotide compounds |
| JP5536963B2 (ja) | 2012-03-04 | 2014-07-02 | 株式会社ボナック | microRNA阻害剤 |
| JP6208228B2 (ja) | 2012-06-21 | 2017-10-04 | ミラゲン セラピューティクス, インコーポレイテッド | ロックド核酸モチーフを含むオリゴヌクレオチドベースの阻害剤 |
| US9163235B2 (en) | 2012-06-21 | 2015-10-20 | MiRagen Therapeutics, Inc. | Inhibitors of the miR-15 family of micro-RNAs |
| WO2014043544A1 (en) | 2012-09-14 | 2014-03-20 | Rana Therapeutics, Inc. | Multimeric oligonucleotide compounds |
| US9725721B2 (en) | 2012-09-26 | 2017-08-08 | Mirrx Therapeutics | Oligomers with improved off-target profile |
| SG11201503821YA (en) * | 2012-11-15 | 2015-06-29 | Roche Innovation Ct Copenhagen As | Oligonucleotide conjugates |
| RU2649367C2 (ru) | 2013-01-30 | 2018-04-02 | Ф. Хоффманн-Ля Рош Аг | Конъюгаты углевода и lna-олигонуклеотида |
| WO2014118272A1 (en) | 2013-01-30 | 2014-08-07 | Santaris Pharma A/S | Antimir-122 oligonucleotide carbohydrate conjugates |
| CA2909868C (en) | 2013-05-01 | 2021-10-19 | Regulus Therapeutics Inc. | Compounds and methods for enhanced cellular uptake |
| KR102482890B1 (ko) | 2013-05-01 | 2022-12-30 | 아이오니스 파마수티컬즈, 인코포레이티드 | 아포지질단백질 (a) 발현을 조절하는 조성물 및 방법 |
| CA2935426C (en) | 2014-01-30 | 2023-07-25 | F. Hoffmann-La Roche Ag | Polyoligomer compound with biocleavable conjugates for reducing or inhibiting expression of a nucleic acid target |
| RU2017105342A (ru) | 2014-08-07 | 2018-09-13 | Регулус Терапьютикс Инк. | НАЦЕЛИВАНИЕ НА микроРНК ПРИ РАССТРОЙСТВАХ ОБМЕНА ВЕЩЕСТВ |
-
2015
- 2015-08-06 RU RU2017105342A patent/RU2017105342A/ru not_active Application Discontinuation
- 2015-08-06 BR BR112017001931A patent/BR112017001931A2/pt not_active Application Discontinuation
- 2015-08-06 CN CN201580039229.9A patent/CN106559995A/zh active Pending
- 2015-08-06 WO PCT/US2015/043926 patent/WO2016022753A1/en not_active Ceased
- 2015-08-06 AU AU2015301057A patent/AU2015301057A1/en not_active Abandoned
- 2015-08-06 US US14/819,648 patent/US9487783B2/en not_active Expired - Fee Related
- 2015-08-06 MX MX2017001493A patent/MX2017001493A/es unknown
- 2015-08-06 JP JP2017506667A patent/JP2017523790A/ja not_active Ceased
- 2015-08-06 KR KR1020177005873A patent/KR20170033439A/ko not_active Withdrawn
- 2015-08-06 CA CA2953883A patent/CA2953883A1/en not_active Abandoned
- 2015-08-06 MA MA040463A patent/MA40463A/fr unknown
- 2015-08-06 SG SG11201610877PA patent/SG11201610877PA/en unknown
- 2015-08-06 TW TW104125684A patent/TW201613949A/zh unknown
- 2015-08-06 EP EP15750573.6A patent/EP3177721A1/en not_active Withdrawn
-
2016
- 2016-09-22 US US15/272,619 patent/US9862950B2/en not_active Expired - Fee Related
- 2016-12-28 IL IL249819A patent/IL249819A0/en unknown
-
2017
- 2017-02-01 CO CONC2017/0000950A patent/CO2017000950A2/es unknown
- 2017-02-07 CL CL2017000312A patent/CL2017000312A1/es unknown
- 2017-10-05 US US15/725,843 patent/US10138484B2/en not_active Expired - Fee Related
-
2018
- 2018-09-25 US US16/140,881 patent/US20190153444A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| EP3177721A1 (en) | 2017-06-14 |
| US20160046940A1 (en) | 2016-02-18 |
| US9862950B2 (en) | 2018-01-09 |
| AU2015301057A1 (en) | 2017-01-19 |
| RU2017105342A3 (enExample) | 2019-03-05 |
| MA40463A (fr) | 2017-06-14 |
| SG11201610877PA (en) | 2017-02-27 |
| TW201613949A (en) | 2016-04-16 |
| WO2016022753A1 (en) | 2016-02-11 |
| RU2017105342A (ru) | 2018-09-13 |
| MX2017001493A (es) | 2017-05-09 |
| CA2953883A1 (en) | 2016-02-11 |
| US9487783B2 (en) | 2016-11-08 |
| CL2017000312A1 (es) | 2017-10-06 |
| BR112017001931A2 (pt) | 2017-11-28 |
| IL249819A0 (en) | 2017-03-30 |
| US10138484B2 (en) | 2018-11-27 |
| US20170067051A1 (en) | 2017-03-09 |
| CN106559995A (zh) | 2017-04-05 |
| JP2017523790A (ja) | 2017-08-24 |
| US20180171334A1 (en) | 2018-06-21 |
| CO2017000950A2 (es) | 2017-06-30 |
| US20190153444A1 (en) | 2019-05-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10941400B2 (en) | Compounds and methods for enhanced cellular uptake | |
| JP6300775B2 (ja) | 代謝障害に関するターゲッティングマイクロrna | |
| US10138484B2 (en) | Targeting microRNAs for metabolic disorders | |
| JP6092226B2 (ja) | Gcgr発現のアンチセンス調整 | |
| JP2017521045A (ja) | アンジオポエチン様因子3発現を調節するための組成物及び方法 | |
| EA034731B1 (ru) | Соединения на основе микрорнк и способы модулирования активности mir-122 | |
| JP6043347B2 (ja) | 線維芽細胞増殖因子受容体4の発現のアンチセンス調節 | |
| JP2014512186A (ja) | Ptp1b発現のアンチセンス調節 | |
| WO2012145374A1 (en) | TARGETING miR-378 FAMILY MEMBERS FOR THE TREATMENT OF METABOLIC DISORDERS | |
| US20220098581A1 (en) | Methods for Oral Delivery of Oligonucleotides | |
| WO2020037797A1 (zh) | miR-221/222及其抑制剂用于制备调控肝脏脂肪沉积、肝纤维化和肝细胞肝癌的药物及检测靶点 | |
| HK1221257B (en) | Compounds and methods for enhanced cellular uptake |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20170302 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PC1203 | Withdrawal of no request for examination |