KR20160037201A - Bet-브로모도메인 억제제를 사용한 미만성 거대 b-세포 림프종 (dlbcl)의 치료 방법 - Google Patents
Bet-브로모도메인 억제제를 사용한 미만성 거대 b-세포 림프종 (dlbcl)의 치료 방법 Download PDFInfo
- Publication number
- KR20160037201A KR20160037201A KR1020167004844A KR20167004844A KR20160037201A KR 20160037201 A KR20160037201 A KR 20160037201A KR 1020167004844 A KR1020167004844 A KR 1020167004844A KR 20167004844 A KR20167004844 A KR 20167004844A KR 20160037201 A KR20160037201 A KR 20160037201A
- Authority
- KR
- South Korea
- Prior art keywords
- compound
- carbon atoms
- gene
- alkyl
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 238000000034 method Methods 0.000 title claims abstract description 50
- 206010012818 diffuse large B-cell lymphoma Diseases 0.000 title claims abstract description 36
- 208000031671 Large B-Cell Diffuse Lymphoma Diseases 0.000 title claims abstract description 35
- 229940125763 bromodomain inhibitor Drugs 0.000 title description 2
- 150000001875 compounds Chemical class 0.000 claims abstract description 291
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 94
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 73
- 125000005843 halogen group Chemical group 0.000 claims abstract description 60
- 239000000203 mixture Substances 0.000 claims abstract description 57
- 150000003839 salts Chemical class 0.000 claims abstract description 57
- 239000012453 solvate Substances 0.000 claims abstract description 30
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims abstract description 26
- 125000003545 alkoxy group Chemical group 0.000 claims abstract description 25
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 23
- 210000003719 b-lymphocyte Anatomy 0.000 claims abstract description 20
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims abstract description 16
- 125000004076 pyridyl group Chemical group 0.000 claims abstract description 16
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 11
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims abstract description 8
- 125000004093 cyano group Chemical group *C#N 0.000 claims abstract description 8
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims abstract description 8
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims abstract description 8
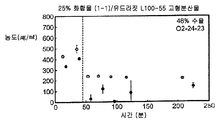
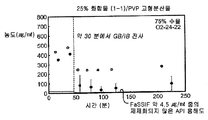
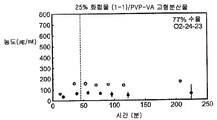
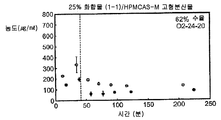
- 239000007962 solid dispersion Substances 0.000 claims description 184
- 229920000642 polymer Polymers 0.000 claims description 61
- -1 3'-cyanobiphenyl-4-yl Chemical group 0.000 claims description 46
- 229920000639 hydroxypropylmethylcellulose acetate succinate Polymers 0.000 claims description 44
- 108090000623 proteins and genes Proteins 0.000 claims description 41
- 239000008194 pharmaceutical composition Substances 0.000 claims description 39
- ZUAAPNNKRHMPKG-UHFFFAOYSA-N acetic acid;butanedioic acid;methanol;propane-1,2-diol Chemical compound OC.CC(O)=O.CC(O)CO.OC(=O)CCC(O)=O ZUAAPNNKRHMPKG-UHFFFAOYSA-N 0.000 claims description 32
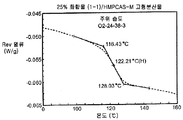
- 238000000634 powder X-ray diffraction Methods 0.000 claims description 29
- 230000014509 gene expression Effects 0.000 claims description 22
- 230000009477 glass transition Effects 0.000 claims description 22
- GNMUEVRJHCWKTO-FQEVSTJZSA-N 6h-thieno[3,2-f][1,2,4]triazolo[4,3-a][1,4]diazepine-6-acetamide, 4-(4-chlorophenyl)-n-(4-hydroxyphenyl)-2,3,9-trimethyl-, (6s)- Chemical compound C([C@@H]1N=C(C2=C(N3C(C)=NN=C31)SC(=C2C)C)C=1C=CC(Cl)=CC=1)C(=O)NC1=CC=C(O)C=C1 GNMUEVRJHCWKTO-FQEVSTJZSA-N 0.000 claims description 19
- 101150053046 MYD88 gene Proteins 0.000 claims description 19
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 18
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 15
- 230000037361 pathway Effects 0.000 claims description 10
- 108700003785 Baculoviral IAP Repeat-Containing 3 Proteins 0.000 claims description 7
- 102100021662 Baculoviral IAP repeat-containing protein 3 Human genes 0.000 claims description 7
- 101150104237 Birc3 gene Proteins 0.000 claims description 7
- 101100219559 Homo sapiens CARD11 gene Proteins 0.000 claims description 7
- 101001011441 Homo sapiens Interferon regulatory factor 4 Proteins 0.000 claims description 7
- 102100030126 Interferon regulatory factor 4 Human genes 0.000 claims description 7
- 101100379220 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) API2 gene Proteins 0.000 claims description 7
- 108010047933 Tumor Necrosis Factor alpha-Induced Protein 3 Proteins 0.000 claims description 7
- 102100024596 Tumor necrosis factor alpha-induced protein 3 Human genes 0.000 claims description 7
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 claims description 7
- 101150001392 CD79B gene Proteins 0.000 claims description 6
- 101150101999 IL6 gene Proteins 0.000 claims description 6
- 101150046106 IRAK1 gene Proteins 0.000 claims description 6
- 101150099493 STAT3 gene Proteins 0.000 claims description 6
- 101150095338 TLR6 gene Proteins 0.000 claims description 6
- 101150090104 TNFRSF17 gene Proteins 0.000 claims description 6
- 101150080074 TP53 gene Proteins 0.000 claims description 6
- 108700025694 p53 Genes Proteins 0.000 claims description 6
- 238000002560 therapeutic procedure Methods 0.000 claims description 6
- 230000008569 process Effects 0.000 claims description 4
- 206010069754 Acquired gene mutation Diseases 0.000 claims description 3
- 230000037439 somatic mutation Effects 0.000 claims description 3
- 229940049706 benzodiazepine Drugs 0.000 claims 2
- 230000002222 downregulating effect Effects 0.000 claims 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 44
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 44
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 43
- 239000006185 dispersion Substances 0.000 description 37
- 238000009472 formulation Methods 0.000 description 34
- 210000004027 cell Anatomy 0.000 description 17
- 239000002552 dosage form Substances 0.000 description 17
- 238000012360 testing method Methods 0.000 description 17
- 239000002775 capsule Substances 0.000 description 15
- 238000004090 dissolution Methods 0.000 description 14
- 229940079593 drug Drugs 0.000 description 14
- 239000003814 drug Substances 0.000 description 14
- 239000000243 solution Substances 0.000 description 14
- 239000007921 spray Substances 0.000 description 14
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 12
- 238000002360 preparation method Methods 0.000 description 12
- 241001465754 Metazoa Species 0.000 description 11
- 239000008187 granular material Substances 0.000 description 11
- 229950000080 birabresib Drugs 0.000 description 10
- 239000003112 inhibitor Substances 0.000 description 10
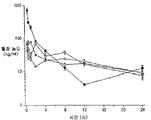
- 230000036470 plasma concentration Effects 0.000 description 10
- 239000002904 solvent Substances 0.000 description 9
- 206010025323 Lymphomas Diseases 0.000 description 8
- 239000002253 acid Substances 0.000 description 8
- 238000004458 analytical method Methods 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 8
- 239000000314 lubricant Substances 0.000 description 8
- 239000000546 pharmaceutical excipient Substances 0.000 description 8
- 102100024134 Myeloid differentiation primary response protein MyD88 Human genes 0.000 description 7
- 206010028980 Neoplasm Diseases 0.000 description 7
- 241000700159 Rattus Species 0.000 description 7
- IYKJEILNJZQJPU-UHFFFAOYSA-N acetic acid;butanedioic acid Chemical compound CC(O)=O.OC(=O)CCC(O)=O IYKJEILNJZQJPU-UHFFFAOYSA-N 0.000 description 7
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 7
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 7
- 238000000338 in vitro Methods 0.000 description 7
- 238000001990 intravenous administration Methods 0.000 description 7
- 239000002609 medium Substances 0.000 description 7
- 230000001105 regulatory effect Effects 0.000 description 7
- 238000001694 spray drying Methods 0.000 description 7
- 230000002195 synergetic effect Effects 0.000 description 7
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- 102100021663 Baculoviral IAP repeat-containing protein 5 Human genes 0.000 description 6
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 6
- 108010002687 Survivin Proteins 0.000 description 6
- 239000000945 filler Substances 0.000 description 6
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 6
- 238000001727 in vivo Methods 0.000 description 6
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 6
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 6
- 239000002245 particle Substances 0.000 description 6
- 229920000191 poly(N-vinyl pyrrolidone) Polymers 0.000 description 6
- 239000000843 powder Substances 0.000 description 6
- 102000001805 Bromodomains Human genes 0.000 description 5
- 108050009021 Bromodomains Proteins 0.000 description 5
- 241000282472 Canis lupus familiaris Species 0.000 description 5
- 241000124008 Mammalia Species 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- 201000011510 cancer Diseases 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- 229920001577 copolymer Polymers 0.000 description 5
- 238000000113 differential scanning calorimetry Methods 0.000 description 5
- 150000004683 dihydrates Chemical class 0.000 description 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 5
- 239000007884 disintegrant Substances 0.000 description 5
- 229940124302 mTOR inhibitor Drugs 0.000 description 5
- 239000003628 mammalian target of rapamycin inhibitor Substances 0.000 description 5
- 239000008108 microcrystalline cellulose Substances 0.000 description 5
- 229940016286 microcrystalline cellulose Drugs 0.000 description 5
- 230000035772 mutation Effects 0.000 description 5
- 230000004044 response Effects 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- 229940124291 BTK inhibitor Drugs 0.000 description 4
- 102100026596 Bcl-2-like protein 1 Human genes 0.000 description 4
- 101150008012 Bcl2l1 gene Proteins 0.000 description 4
- 229920003135 Eudragit® L 100-55 Polymers 0.000 description 4
- 229920003134 Eudragit® polymer Polymers 0.000 description 4
- 241000282412 Homo Species 0.000 description 4
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 4
- 239000002177 L01XE27 - Ibrutinib Substances 0.000 description 4
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 239000013543 active substance Substances 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 125000003342 alkenyl group Chemical group 0.000 description 4
- 230000006907 apoptotic process Effects 0.000 description 4
- 108700000711 bcl-X Proteins 0.000 description 4
- 239000011324 bead Substances 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 238000011161 development Methods 0.000 description 4
- 238000001035 drying Methods 0.000 description 4
- 230000008030 elimination Effects 0.000 description 4
- 238000003379 elimination reaction Methods 0.000 description 4
- GDCRSXZBSIRSFR-UHFFFAOYSA-N ethyl prop-2-enoate;2-methylprop-2-enoic acid Chemical compound CC(=C)C(O)=O.CCOC(=O)C=C GDCRSXZBSIRSFR-UHFFFAOYSA-N 0.000 description 4
- 229960001507 ibrutinib Drugs 0.000 description 4
- XYFPWWZEPKGCCK-GOSISDBHSA-N ibrutinib Chemical compound C1=2C(N)=NC=NC=2N([C@H]2CN(CCC2)C(=O)C=C)N=C1C(C=C1)=CC=C1OC1=CC=CC=C1 XYFPWWZEPKGCCK-GOSISDBHSA-N 0.000 description 4
- 238000012750 in vivo screening Methods 0.000 description 4
- 150000007522 mineralic acids Chemical class 0.000 description 4
- 150000007524 organic acids Chemical class 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 238000010922 spray-dried dispersion Methods 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- 102100029897 Bromodomain-containing protein 7 Human genes 0.000 description 3
- 102100035186 DNA excision repair protein ERCC-1 Human genes 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- HKVAMNSJSFKALM-GKUWKFKPSA-N Everolimus Chemical compound C1C[C@@H](OCCO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 HKVAMNSJSFKALM-GKUWKFKPSA-N 0.000 description 3
- 101000794019 Homo sapiens Bromodomain-containing protein 7 Proteins 0.000 description 3
- 101000876529 Homo sapiens DNA excision repair protein ERCC-1 Proteins 0.000 description 3
- 101000838035 Homo sapiens TATA box-binding protein-associated factor RNA polymerase I subunit A Proteins 0.000 description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 3
- 102000004889 Interleukin-6 Human genes 0.000 description 3
- 108090001005 Interleukin-6 Proteins 0.000 description 3
- 230000004163 JAK-STAT signaling pathway Effects 0.000 description 3
- 239000002202 Polyethylene glycol Substances 0.000 description 3
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 102100028553 TATA box-binding protein-associated factor RNA polymerase I subunit A Human genes 0.000 description 3
- 125000000218 acetic acid group Chemical group C(C)(=O)* 0.000 description 3
- 230000000996 additive effect Effects 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 150000001721 carbon Chemical group 0.000 description 3
- 239000001913 cellulose Substances 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 3
- 229960005167 everolimus Drugs 0.000 description 3
- 230000004153 glucose metabolism Effects 0.000 description 3
- 238000005469 granulation Methods 0.000 description 3
- 230000003179 granulation Effects 0.000 description 3
- 150000004677 hydrates Chemical class 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 235000019359 magnesium stearate Nutrition 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 239000008185 minitablet Substances 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- XNGIFLGASWRNHJ-UHFFFAOYSA-L phthalate(2-) Chemical compound [O-]C(=O)C1=CC=CC=C1C([O-])=O XNGIFLGASWRNHJ-UHFFFAOYSA-L 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 229960004641 rituximab Drugs 0.000 description 3
- 230000035945 sensitivity Effects 0.000 description 3
- 238000000935 solvent evaporation Methods 0.000 description 3
- 230000000707 stereoselective effect Effects 0.000 description 3
- 239000000454 talc Substances 0.000 description 3
- 229910052623 talc Inorganic materials 0.000 description 3
- 235000012222 talc Nutrition 0.000 description 3
- 238000013518 transcription Methods 0.000 description 3
- 230000035897 transcription Effects 0.000 description 3
- 239000000080 wetting agent Substances 0.000 description 3
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 2
- LRANPJDWHYRCER-UHFFFAOYSA-N 1,2-diazepine Chemical compound N1C=CC=CC=N1 LRANPJDWHYRCER-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- WXTMDXOMEHJXQO-UHFFFAOYSA-N 2,5-dihydroxybenzoic acid Chemical compound OC(=O)C1=CC(O)=CC=C1O WXTMDXOMEHJXQO-UHFFFAOYSA-N 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- XAUDJQYHKZQPEU-KVQBGUIXSA-N 5-aza-2'-deoxycytidine Chemical compound O=C1N=C(N)N=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 XAUDJQYHKZQPEU-KVQBGUIXSA-N 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- WSVLPVUVIUVCRA-KPKNDVKVSA-N Alpha-lactose monohydrate Chemical compound O.O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O WSVLPVUVIUVCRA-KPKNDVKVSA-N 0.000 description 2
- 241000272517 Anseriformes Species 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 102100027203 B-cell antigen receptor complex-associated protein beta chain Human genes 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 239000004322 Butylated hydroxytoluene Substances 0.000 description 2
- NLZUEZXRPGMBCV-UHFFFAOYSA-N Butylhydroxytoluene Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NLZUEZXRPGMBCV-UHFFFAOYSA-N 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 2
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 2
- 108010077544 Chromatin Proteins 0.000 description 2
- 229920002785 Croscarmellose sodium Polymers 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- 230000033616 DNA repair Effects 0.000 description 2
- 101100278318 Dictyostelium discoideum dohh-2 gene Proteins 0.000 description 2
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- 229920003152 Eudragit® RS polymer Polymers 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- IAJILQKETJEXLJ-UHFFFAOYSA-N Galacturonsaeure Natural products O=CC(O)C(O)C(O)C(O)C(O)=O IAJILQKETJEXLJ-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- 101000914491 Homo sapiens B-cell antigen receptor complex-associated protein beta chain Proteins 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical class Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 102000003814 Interleukin-10 Human genes 0.000 description 2
- 108090000174 Interleukin-10 Proteins 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 208000025205 Mantle-Cell Lymphoma Diseases 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- 102100023050 Nuclear factor NF-kappa-B p105 subunit Human genes 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical class OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 102000009097 Phosphorylases Human genes 0.000 description 2
- 108010073135 Phosphorylases Proteins 0.000 description 2
- ZTHYODDOHIVTJV-UHFFFAOYSA-N Propyl gallate Chemical compound CCCOC(=O)C1=CC(O)=C(O)C(O)=C1 ZTHYODDOHIVTJV-UHFFFAOYSA-N 0.000 description 2
- 108010017324 STAT3 Transcription Factor Proteins 0.000 description 2
- 102100024040 Signal transducer and activator of transcription 3 Human genes 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- 235000021355 Stearic acid Nutrition 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical class OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 102000002689 Toll-like receptor Human genes 0.000 description 2
- 108020000411 Toll-like receptor Proteins 0.000 description 2
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- RWZYAGGXGHYGMB-UHFFFAOYSA-N anthranilic acid Chemical compound NC1=CC=CC=C1C(O)=O RWZYAGGXGHYGMB-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 235000010354 butylated hydroxytoluene Nutrition 0.000 description 2
- 229940095259 butylated hydroxytoluene Drugs 0.000 description 2
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 2
- 210000003483 chromatin Anatomy 0.000 description 2
- 229940075614 colloidal silicon dioxide Drugs 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 238000012937 correction Methods 0.000 description 2
- 229960001681 croscarmellose sodium Drugs 0.000 description 2
- 235000010947 crosslinked sodium carboxy methyl cellulose Nutrition 0.000 description 2
- 238000007418 data mining Methods 0.000 description 2
- 229960003603 decitabine Drugs 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 239000008121 dextrose Substances 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- FSBVERYRVPGNGG-UHFFFAOYSA-N dimagnesium dioxido-bis[[oxido(oxo)silyl]oxy]silane hydrate Chemical compound O.[Mg+2].[Mg+2].[O-][Si](=O)O[Si]([O-])([O-])O[Si]([O-])=O FSBVERYRVPGNGG-UHFFFAOYSA-N 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 239000002270 dispersing agent Substances 0.000 description 2
- 238000007922 dissolution test Methods 0.000 description 2
- 239000002702 enteric coating Substances 0.000 description 2
- 238000009505 enteric coating Methods 0.000 description 2
- FSXVSUSRJXIJHB-UHFFFAOYSA-M ethyl prop-2-enoate;methyl 2-methylprop-2-enoate;trimethyl-[2-(2-methylprop-2-enoyloxy)ethyl]azanium;chloride Chemical compound [Cl-].CCOC(=O)C=C.COC(=O)C(C)=C.CC(=C)C(=O)OCC[N+](C)(C)C FSXVSUSRJXIJHB-UHFFFAOYSA-M 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- 238000013213 extrapolation Methods 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 235000013305 food Nutrition 0.000 description 2
- 230000002496 gastric effect Effects 0.000 description 2
- 210000001035 gastrointestinal tract Anatomy 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 239000007903 gelatin capsule Substances 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 230000002068 genetic effect Effects 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 239000003276 histone deacetylase inhibitor Substances 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 2
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 2
- 150000007529 inorganic bases Chemical class 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- TWBYWOBDOCUKOW-UHFFFAOYSA-N isonicotinic acid Chemical compound OC(=O)C1=CC=NC=C1 TWBYWOBDOCUKOW-UHFFFAOYSA-N 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 229960001375 lactose Drugs 0.000 description 2
- 229960001021 lactose monohydrate Drugs 0.000 description 2
- 229960004942 lenalidomide Drugs 0.000 description 2
- GOTYRUGSSMKFNF-UHFFFAOYSA-N lenalidomide Chemical compound C1C=2C(N)=CC=CC=2C(=O)N1C1CCC(=O)NC1=O GOTYRUGSSMKFNF-UHFFFAOYSA-N 0.000 description 2
- 230000003902 lesion Effects 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 239000000391 magnesium silicate Substances 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 229920000515 polycarbonate Polymers 0.000 description 2
- 229920003124 powdered cellulose Polymers 0.000 description 2
- 235000019814 powdered cellulose Nutrition 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 238000011002 quantification Methods 0.000 description 2
- ZAHRKKWIAAJSAO-UHFFFAOYSA-N rapamycin Natural products COCC(O)C(=C/C(C)C(=O)CC(OC(=O)C1CCCCN1C(=O)C(=O)C2(O)OC(CC(OC)C(=CC=CC=CC(C)CC(C)C(=O)C)C)CCC2C)C(C)CC3CCC(O)C(C3)OC)C ZAHRKKWIAAJSAO-UHFFFAOYSA-N 0.000 description 2
- 238000001953 recrystallisation Methods 0.000 description 2
- 238000000611 regression analysis Methods 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 229960002930 sirolimus Drugs 0.000 description 2
- QFJCIRLUMZQUOT-HPLJOQBZSA-N sirolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 QFJCIRLUMZQUOT-HPLJOQBZSA-N 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000007909 solid dosage form Substances 0.000 description 2
- 206010062113 splenic marginal zone lymphoma Diseases 0.000 description 2
- 238000013112 stability test Methods 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 239000008117 stearic acid Substances 0.000 description 2
- 239000008223 sterile water Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 2
- NAOLWIGVYRIGTP-UHFFFAOYSA-N 1,3,5-trihydroxyanthracene-9,10-dione Chemical compound C1=CC(O)=C2C(=O)C3=CC(O)=CC(O)=C3C(=O)C2=C1 NAOLWIGVYRIGTP-UHFFFAOYSA-N 0.000 description 1
- OKMWKBLSFKFYGZ-UHFFFAOYSA-N 1-behenoylglycerol Chemical compound CCCCCCCCCCCCCCCCCCCCCC(=O)OCC(O)CO OKMWKBLSFKFYGZ-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical class NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- 102000004121 Annexin A5 Human genes 0.000 description 1
- 108090000672 Annexin A5 Proteins 0.000 description 1
- 241000271566 Aves Species 0.000 description 1
- 208000010839 B-cell chronic lymphocytic leukemia Diseases 0.000 description 1
- LSPHULWDVZXLIL-UHFFFAOYSA-N Camphoric acid Natural products CC1(C)C(C(O)=O)CCC1(C)C(O)=O LSPHULWDVZXLIL-UHFFFAOYSA-N 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 102100025064 Cellular tumor antigen p53 Human genes 0.000 description 1
- GHOKWGTUZJEAQD-UHFFFAOYSA-N Chick antidermatitis factor Natural products OCC(C)(C)C(O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-UHFFFAOYSA-N 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 208000011231 Crohn disease Diseases 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 102000012078 E2F2 Transcription Factor Human genes 0.000 description 1
- 108010036466 E2F2 Transcription Factor Proteins 0.000 description 1
- 102100038616 E3 ubiquitin-protein ligase MARCHF1 Human genes 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 238000000729 Fisher's exact test Methods 0.000 description 1
- DSLZVSRJTYRBFB-UHFFFAOYSA-N Galactaric acid Natural products OC(=O)C(O)C(O)C(O)C(O)C(O)=O DSLZVSRJTYRBFB-UHFFFAOYSA-N 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 239000001828 Gelatine Substances 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 208000002250 Hematologic Neoplasms Diseases 0.000 description 1
- 108010033040 Histones Proteins 0.000 description 1
- 102000006947 Histones Human genes 0.000 description 1
- 101000957748 Homo sapiens E3 ubiquitin-protein ligase MARCHF1 Proteins 0.000 description 1
- 101000852483 Homo sapiens Interleukin-1 receptor-associated kinase 1 Proteins 0.000 description 1
- 101001090713 Homo sapiens L-lactate dehydrogenase A chain Proteins 0.000 description 1
- 101000979342 Homo sapiens Nuclear factor NF-kappa-B p105 subunit Proteins 0.000 description 1
- 101000864800 Homo sapiens Serine/threonine-protein kinase Sgk1 Proteins 0.000 description 1
- 101000617830 Homo sapiens Sterol O-acyltransferase 1 Proteins 0.000 description 1
- 101000634866 Homo sapiens TRAF-type zinc finger domain-containing protein 1 Proteins 0.000 description 1
- 101000669406 Homo sapiens Toll-like receptor 6 Proteins 0.000 description 1
- 101000801255 Homo sapiens Tumor necrosis factor receptor superfamily member 17 Proteins 0.000 description 1
- 101001009087 Homo sapiens Tyrosine-protein kinase HCK Proteins 0.000 description 1
- 101000808011 Homo sapiens Vascular endothelial growth factor A Proteins 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 206010021143 Hypoxia Diseases 0.000 description 1
- 241000370541 Idia Species 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 102100036342 Interleukin-1 receptor-associated kinase 1 Human genes 0.000 description 1
- 239000012565 Kollidon 17 Substances 0.000 description 1
- QAQJMLQRFWZOBN-LAUBAEHRSA-N L-ascorbyl-6-palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](O)[C@H]1OC(=O)C(O)=C1O QAQJMLQRFWZOBN-LAUBAEHRSA-N 0.000 description 1
- 239000011786 L-ascorbyl-6-palmitate Substances 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- 102100034671 L-lactate dehydrogenase A chain Human genes 0.000 description 1
- 208000031422 Lymphocytic Chronic B-Cell Leukemia Diseases 0.000 description 1
- 208000028018 Lymphocytic leukaemia Diseases 0.000 description 1
- 231100000002 MTT assay Toxicity 0.000 description 1
- 238000000134 MTT assay Methods 0.000 description 1
- 239000004425 Makrolon Substances 0.000 description 1
- UEZVMMHDMIWARA-UHFFFAOYSA-N Metaphosphoric acid Chemical class OP(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-N 0.000 description 1
- 229920003091 Methocel™ Polymers 0.000 description 1
- 208000034578 Multiple myelomas Diseases 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical class O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 241000286209 Phasianidae Species 0.000 description 1
- 241000233805 Phoenix Species 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 229920002565 Polyethylene Glycol 400 Polymers 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 1
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 102100030070 Serine/threonine-protein kinase Sgk1 Human genes 0.000 description 1
- 102100021993 Sterol O-acyltransferase 1 Human genes 0.000 description 1
- 101000697584 Streptomyces lavendulae Streptothricin acetyltransferase Proteins 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- 206010042971 T-cell lymphoma Diseases 0.000 description 1
- 208000027585 T-cell non-Hodgkin lymphoma Diseases 0.000 description 1
- 102100029451 TRAF-type zinc finger domain-containing protein 1 Human genes 0.000 description 1
- 102100039387 Toll-like receptor 6 Human genes 0.000 description 1
- 108700025716 Tumor Suppressor Genes Proteins 0.000 description 1
- 102000044209 Tumor Suppressor Genes Human genes 0.000 description 1
- 108010078814 Tumor Suppressor Protein p53 Proteins 0.000 description 1
- 102100033726 Tumor necrosis factor receptor superfamily member 17 Human genes 0.000 description 1
- 102100029823 Tyrosine-protein kinase BTK Human genes 0.000 description 1
- 102100027389 Tyrosine-protein kinase HCK Human genes 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 102100039037 Vascular endothelial growth factor A Human genes 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- ABRVLXLNVJHDRQ-UHFFFAOYSA-N [2-pyridin-3-yl-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound FC(C1=CC(=CC(=N1)C=1C=NC=CC=1)CN)(F)F ABRVLXLNVJHDRQ-UHFFFAOYSA-N 0.000 description 1
- IAJILQKETJEXLJ-RSJOWCBRSA-N aldehydo-D-galacturonic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-RSJOWCBRSA-N 0.000 description 1
- IAJILQKETJEXLJ-QTBDOELSSA-N aldehydo-D-glucuronic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-QTBDOELSSA-N 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- CEGOLXSVJUTHNZ-UHFFFAOYSA-K aluminium tristearate Chemical compound [Al+3].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CEGOLXSVJUTHNZ-UHFFFAOYSA-K 0.000 description 1
- 229940063655 aluminum stearate Drugs 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000002424 anti-apoptotic effect Effects 0.000 description 1
- 230000001028 anti-proliverative effect Effects 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000002216 antistatic agent Substances 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 235000010385 ascorbyl palmitate Nutrition 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 238000000889 atomisation Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid group Chemical group C(C1=CC=CC=C1)(=O)O WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- LSPHULWDVZXLIL-QUBYGPBYSA-N camphoric acid Chemical compound CC1(C)[C@H](C(O)=O)CC[C@]1(C)C(O)=O LSPHULWDVZXLIL-QUBYGPBYSA-N 0.000 description 1
- 239000007894 caplet Substances 0.000 description 1
- 239000007963 capsule composition Substances 0.000 description 1
- 235000011089 carbon dioxide Nutrition 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 229960001777 castor oil Drugs 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 208000032852 chronic lymphocytic leukemia Diseases 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 239000008119 colloidal silica Substances 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000000875 corresponding effect Effects 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 229940127089 cytotoxic agent Drugs 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 229940096516 dextrates Drugs 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 230000003828 downregulation Effects 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- RDYMFSUJUZBWLH-UHFFFAOYSA-N endosulfan Chemical compound C12COS(=O)OCC2C2(Cl)C(Cl)=C(Cl)C1(Cl)C2(Cl)Cl RDYMFSUJUZBWLH-UHFFFAOYSA-N 0.000 description 1
- 238000006911 enzymatic reaction Methods 0.000 description 1
- 239000002532 enzyme inhibitor Substances 0.000 description 1
- 238000011067 equilibration Methods 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- MVPICKVDHDWCJQ-UHFFFAOYSA-N ethyl 3-pyrrolidin-1-ylpropanoate Chemical compound CCOC(=O)CCN1CCCC1 MVPICKVDHDWCJQ-UHFFFAOYSA-N 0.000 description 1
- 239000000374 eutectic mixture Substances 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000010195 expression analysis Methods 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 229960002598 fumaric acid Drugs 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- DSLZVSRJTYRBFB-DUHBMQHGSA-N galactaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O DSLZVSRJTYRBFB-DUHBMQHGSA-N 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 238000011223 gene expression profiling Methods 0.000 description 1
- 229960005219 gentisic acid Drugs 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229960001031 glucose Drugs 0.000 description 1
- 229940097043 glucuronic acid Drugs 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 229940049654 glyceryl behenate Drugs 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- LHGVFZTZFXWLCP-UHFFFAOYSA-N guaiacol Chemical class COC1=CC=CC=C1O LHGVFZTZFXWLCP-UHFFFAOYSA-N 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 201000005787 hematologic cancer Diseases 0.000 description 1
- 230000002489 hematologic effect Effects 0.000 description 1
- 239000008241 heterogeneous mixture Substances 0.000 description 1
- 229920001903 high density polyethylene Polymers 0.000 description 1
- 239000004700 high-density polyethylene Substances 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 125000001183 hydrocarbyl group Chemical group 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 239000012052 hydrophilic carrier Substances 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 229940075628 hypomethylating agent Drugs 0.000 description 1
- 230000007954 hypoxia Effects 0.000 description 1
- 229960003943 hypromellose Drugs 0.000 description 1
- 238000005286 illumination Methods 0.000 description 1
- 238000003119 immunoblot Methods 0.000 description 1
- 238000003364 immunohistochemistry Methods 0.000 description 1
- 239000002955 immunomodulating agent Substances 0.000 description 1
- 229940121354 immunomodulator Drugs 0.000 description 1
- 230000002584 immunomodulator Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 230000031261 interleukin-10 production Effects 0.000 description 1
- 230000017307 interleukin-4 production Effects 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- 210000004731 jugular vein Anatomy 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229920001684 low density polyethylene Polymers 0.000 description 1
- 239000004702 low-density polyethylene Substances 0.000 description 1
- 208000003747 lymphoid leukemia Diseases 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- 235000019793 magnesium trisilicate Nutrition 0.000 description 1
- 229910000386 magnesium trisilicate Inorganic materials 0.000 description 1
- 229940099273 magnesium trisilicate Drugs 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 229960001855 mannitol Drugs 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 229940117841 methacrylic acid copolymer Drugs 0.000 description 1
- 229920003145 methacrylic acid copolymer Polymers 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 238000003801 milling Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000001565 modulated differential scanning calorimetry Methods 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- 210000004985 myeloid-derived suppressor cell Anatomy 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 230000030648 nucleus localization Effects 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 231100000822 oral exposure Toxicity 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003002 pH adjusting agent Substances 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 229940055726 pantothenic acid Drugs 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 238000011056 performance test Methods 0.000 description 1
- 238000005191 phase separation Methods 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 150000004804 polysaccharides Chemical class 0.000 description 1
- 229940069328 povidone Drugs 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 239000000473 propyl gallate Substances 0.000 description 1
- 235000010388 propyl gallate Nutrition 0.000 description 1
- 229940075579 propyl gallate Drugs 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000022983 regulation of cell cycle Effects 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 229940045902 sodium stearyl fumarate Drugs 0.000 description 1
- JAJWGJBVLPIOOH-IZYKLYLVSA-M sodium taurocholate Chemical compound [Na+].C([C@H]1C[C@H]2O)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(=O)NCCS([O-])(=O)=O)C)[C@@]2(C)[C@@H](O)C1 JAJWGJBVLPIOOH-IZYKLYLVSA-M 0.000 description 1
- 239000012265 solid product Substances 0.000 description 1
- 239000006104 solid solution Substances 0.000 description 1
- 230000007928 solubilization Effects 0.000 description 1
- 238000005063 solubilization Methods 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 125000000547 substituted alkyl group Chemical group 0.000 description 1
- BUUPQKDIAURBJP-UHFFFAOYSA-N sulfinic acid Chemical compound OS=O BUUPQKDIAURBJP-UHFFFAOYSA-N 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 238000004885 tandem mass spectrometry Methods 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical class CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 235000019731 tricalcium phosphate Nutrition 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1605—Excipients; Inactive ingredients
- A61K9/1629—Organic macromolecular compounds
- A61K9/1652—Polysaccharides, e.g. alginate, cellulose derivatives; Cyclodextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
- A61K31/5517—1,4-Benzodiazepines, e.g. diazepam or clozapine condensed with five-membered rings having nitrogen as a ring hetero atom, e.g. imidazobenzodiazepines, triazolam
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
- A61K47/38—Cellulose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/141—Intimate drug-carrier mixtures characterised by the carrier, e.g. ordered mixtures, adsorbates, solid solutions, eutectica, co-dried, co-solubilised, co-kneaded, co-milled, co-ground products, co-precipitates, co-evaporates, co-extrudates, co-melts; Drug nanoparticles with adsorbed surface modifiers
- A61K9/146—Intimate drug-carrier mixtures characterised by the carrier, e.g. ordered mixtures, adsorbates, solid solutions, eutectica, co-dried, co-solubilised, co-kneaded, co-milled, co-ground products, co-precipitates, co-evaporates, co-extrudates, co-melts; Drug nanoparticles with adsorbed surface modifiers with organic macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1605—Excipients; Inactive ingredients
- A61K9/1629—Organic macromolecular compounds
- A61K9/1635—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4808—Preparations in capsules, e.g. of gelatin, of chocolate characterised by the form of the capsule or the structure of the filling; Capsules containing small tablets; Capsules with outer layer for immediate drug release
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Inorganic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Applications Claiming Priority (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361862772P | 2013-08-06 | 2013-08-06 | |
| US201361862752P | 2013-08-06 | 2013-08-06 | |
| US61/862,772 | 2013-08-06 | ||
| US61/862,752 | 2013-08-06 | ||
| US201361909703P | 2013-11-27 | 2013-11-27 | |
| US61/909,703 | 2013-11-27 | ||
| PCT/EP2014/002164 WO2015018521A1 (en) | 2013-08-06 | 2014-08-06 | Method of treating diffuse large b-cell lymphoma (dlbcl) using a bet-bromodomain inhibitor |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20160037201A true KR20160037201A (ko) | 2016-04-05 |
Family
ID=51298706
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167004844A Withdrawn KR20160037201A (ko) | 2013-08-06 | 2014-08-06 | Bet-브로모도메인 억제제를 사용한 미만성 거대 b-세포 림프종 (dlbcl)의 치료 방법 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20160158246A1 (cg-RX-API-DMAC7.html) |
| EP (1) | EP3030242A1 (cg-RX-API-DMAC7.html) |
| JP (1) | JP2016529246A (cg-RX-API-DMAC7.html) |
| KR (1) | KR20160037201A (cg-RX-API-DMAC7.html) |
| CN (1) | CN105899212A (cg-RX-API-DMAC7.html) |
| WO (1) | WO2015018521A1 (cg-RX-API-DMAC7.html) |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MX384074B (es) | 2010-05-14 | 2025-03-12 | Dana Farber Cancer Inst Inc | Composiciones y métodos para tratar neoplasias, enfermedades inflamatorias y otros trastornos. |
| CN103154246B (zh) | 2010-05-14 | 2015-11-25 | 达那-法伯癌症研究所 | 用于治疗白血病的组合物和方法 |
| US11446309B2 (en) | 2013-11-08 | 2022-09-20 | Dana-Farber Cancer Institute, Inc. | Combination therapy for cancer using bromodomain and extra-terminal (BET) protein inhibitors |
| EP3110424A1 (en) | 2014-02-28 | 2017-01-04 | Tensha Therapeutics, Inc. | Treatment of conditions associated with hyperinsulinaemia |
| KR20170037670A (ko) * | 2014-08-19 | 2017-04-04 | 온코에틱스 게엠베하 | 티에노트리아졸로디아제핀 화합물을 사용한 림프종의 치료 방법 |
| MX376748B (es) | 2014-10-27 | 2025-03-07 | Tensha Therapeutics Inc | Inhibidores del bromodominio. |
| US10702517B2 (en) | 2015-04-22 | 2020-07-07 | Celgene Quanticel Research, Inc. | Bromodomain inhibitor |
| WO2016176335A1 (en) | 2015-04-27 | 2016-11-03 | Concert Pharmaceuticals, Inc. | Deuterated otx-015 |
| MX2018003824A (es) * | 2015-10-02 | 2019-04-01 | Dana Farber Cancer Inst Inc | Terapia de combinación de inhibidores de bromodominios y bloqueo de puntos de control. |
| CN114569606B (zh) | 2016-07-29 | 2024-04-19 | 英克特诺治疗公司 | 吲哚啉酮化合物的用途 |
| JP7129973B2 (ja) * | 2016-10-20 | 2022-09-02 | セルジーン クオンティセル リサーチ,インク. | ブロモドメイン阻害剤 |
| UA127413C2 (uk) * | 2017-07-04 | 2023-08-16 | Джянгсу Хенгруй Медісін Ко., Лтд. | Фармацевтична композиція та спосіб її отримання |
| CN107879989B (zh) * | 2017-11-29 | 2020-01-03 | 重庆市中药研究院 | 具有生物活性的3,4,5-取代苯并二氮卓2-酮类药物分子及其制备方法 |
| CN107759607B (zh) * | 2017-11-29 | 2019-08-23 | 上海万巷制药有限公司 | 具有抗肿瘤活性的三氮唑并二氮卓化合物及其制备方法 |
| JP2021517119A (ja) * | 2018-03-05 | 2021-07-15 | クリニクム レヒツ デア イザール デア テクニシェン ウニフェルジテート ミュンヘン | 腫瘍溶解性アデノウイルスとcdk4/6阻害剤との組み合わせによる腫瘍の処置 |
| EP4074842A4 (en) * | 2019-12-11 | 2023-09-06 | Tsinghua University | LONG NON-CODING RNA LETN AS A TUMOR MARKER AND THERAPEUTIC TARGET |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2412776C (en) * | 2000-06-16 | 2011-03-15 | Mitsubishi Pharma Corporation | Compositions controlling release ph range and/or speed |
| US8476260B2 (en) * | 2007-12-28 | 2013-07-02 | Mitsubishi Tanabe Pharma Corporation | Antitumor agent |
| JP5715241B2 (ja) * | 2010-05-14 | 2015-05-07 | ダナ−ファーバー キャンサー インスティテュート, インコーポレイテッド | 新生物、炎症性疾患、およびその他の障害を治療するための組成物および方法 |
| MX384074B (es) * | 2010-05-14 | 2025-03-12 | Dana Farber Cancer Inst Inc | Composiciones y métodos para tratar neoplasias, enfermedades inflamatorias y otros trastornos. |
| AR084070A1 (es) * | 2010-12-02 | 2013-04-17 | Constellation Pharmaceuticals Inc | Inhibidores del bromodominio y usos de los mismos |
| US9249161B2 (en) * | 2010-12-02 | 2016-02-02 | Constellation Pharmaceuticals, Inc. | Bromodomain inhibitors and uses thereof |
| JP6265979B2 (ja) * | 2012-06-25 | 2018-01-24 | オンコエシックス ゲーエムベーハー | チエノトリアゾロジアゼピン化合物を使用するリンパ腫の治療方法 |
| HK1211209A1 (en) * | 2012-09-28 | 2016-05-20 | Oncoethix Gmbh | Pharmaceutical formulation containing thienotriazolodiazepine compounds |
-
2014
- 2014-08-06 WO PCT/EP2014/002164 patent/WO2015018521A1/en not_active Ceased
- 2014-08-06 US US14/910,344 patent/US20160158246A1/en not_active Abandoned
- 2014-08-06 CN CN201480055171.2A patent/CN105899212A/zh active Pending
- 2014-08-06 EP EP14748126.1A patent/EP3030242A1/en not_active Withdrawn
- 2014-08-06 KR KR1020167004844A patent/KR20160037201A/ko not_active Withdrawn
- 2014-08-06 JP JP2016532263A patent/JP2016529246A/ja not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| US20160158246A1 (en) | 2016-06-09 |
| EP3030242A1 (en) | 2016-06-15 |
| JP2016529246A (ja) | 2016-09-23 |
| WO2015018521A1 (en) | 2015-02-12 |
| CN105899212A (zh) | 2016-08-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20160037201A (ko) | Bet-브로모도메인 억제제를 사용한 미만성 거대 b-세포 림프종 (dlbcl)의 치료 방법 | |
| WO2015018522A1 (en) | Bet-bromodomain inhibitor shows synergism with several anti-cancer agents in pre-clinical models of diffuse large b-cell lymphoma (dlbcl) | |
| KR20170044172A (ko) | 티에노트리아졸로디아제핀 화합물을 함유하는 약학적 조성물을 사용하여 급성 골수성 백혈병 또는 급성 림프구성 백혈병을 치료하는 방법 | |
| EP3137086A1 (en) | Method of treating resistant non-hodgkin lymphoma, medulloblastoma, and/or alk+non-small cell lung cancer using thienotriazolodiazepine compounds | |
| KR20160145833A (ko) | 티에노트리아졸로디아제핀 화합물을 사용한 급성 골수성 백혈병 및/또는 급성 림프아구성 백혈병의 치료 방법 | |
| KR20170002610A (ko) | 티에노트리아졸로디아제핀 화합물을 사용한 삼중 음성 유방암의 치료 방법 | |
| WO2015169953A1 (en) | Method of treating glioma using thienotriazolodiazepine compounds | |
| EP3074018A1 (en) | Method of treating leukemia using pharmaceutical formulation containing thienotriazolodiazepine compounds | |
| US9795612B2 (en) | Pharmaceutical formulation containing thienotriazolodiazepine compounds | |
| WO2015018520A1 (en) | A bet-brd inhibitor represents a novel agent for alk positive anaplastic large cell lymphoma | |
| KR20170016858A (ko) | 티에노트리아졸로디아제핀 화합물을 사용하는 비소세포성 폐암 및/또는 소세포성 폐암의 치료 방법 | |
| US9820992B2 (en) | Method of treating non-small-cell lung cancer using pharmaceutical formulation containing thienotriazolodiazepine compounds | |
| KR20170037670A (ko) | 티에노트리아졸로디아제핀 화합물을 사용한 림프종의 치료 방법 | |
| WO2015018523A1 (en) | A novel bet-brd inhibitor for treatment of solid tumors | |
| WO2015168587A1 (en) | Method of treating resistant multiple myeloma and mantle cell lymphoma using thienotriazolodiazepine compounds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20160224 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PC1203 | Withdrawal of no request for examination | ||
| WITN | Application deemed withdrawn, e.g. because no request for examination was filed or no examination fee was paid |