KR100939627B1 - 신규한 화합물 및 이를 이용한 유기 발광 소자 - Google Patents
신규한 화합물 및 이를 이용한 유기 발광 소자 Download PDFInfo
- Publication number
- KR100939627B1 KR100939627B1 KR1020060120560A KR20060120560A KR100939627B1 KR 100939627 B1 KR100939627 B1 KR 100939627B1 KR 1020060120560 A KR1020060120560 A KR 1020060120560A KR 20060120560 A KR20060120560 A KR 20060120560A KR 100939627 B1 KR100939627 B1 KR 100939627B1
- Authority
- KR
- South Korea
- Prior art keywords
- group
- formula
- compound
- groups
- light emitting
- Prior art date
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 131
- 230000005525 hole transport Effects 0.000 claims abstract description 38
- 239000011368 organic material Substances 0.000 claims abstract description 33
- 238000002347 injection Methods 0.000 claims abstract description 25
- 239000007924 injection Substances 0.000 claims abstract description 25
- 239000010410 layer Substances 0.000 claims description 100
- 239000000126 substance Substances 0.000 claims description 51
- 125000000623 heterocyclic group Chemical group 0.000 claims description 33
- 125000001424 substituent group Chemical group 0.000 claims description 31
- -1 furylene Chemical group 0.000 claims description 29
- 239000012044 organic layer Substances 0.000 claims description 28
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical compound C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 27
- 125000003118 aryl group Chemical group 0.000 claims description 25
- 150000004982 aromatic amines Chemical class 0.000 claims description 24
- 150000004945 aromatic hydrocarbons Chemical class 0.000 claims description 24
- 125000003545 alkoxy group Chemical group 0.000 claims description 23
- 125000000217 alkyl group Chemical group 0.000 claims description 22
- 125000003277 amino group Chemical group 0.000 claims description 20
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 16
- 150000002825 nitriles Chemical group 0.000 claims description 14
- 125000005264 aryl amine group Chemical group 0.000 claims description 13
- 229910052736 halogen Inorganic materials 0.000 claims description 13
- 150000002367 halogens Chemical group 0.000 claims description 13
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 claims description 11
- 125000003342 alkenyl group Chemical group 0.000 claims description 11
- 125000004432 carbon atom Chemical group C* 0.000 claims description 11
- 229910052739 hydrogen Inorganic materials 0.000 claims description 10
- 239000001257 hydrogen Substances 0.000 claims description 10
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 claims description 8
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 8
- 125000002560 nitrile group Chemical group 0.000 claims description 7
- 229910052799 carbon Inorganic materials 0.000 claims description 6
- 125000004185 ester group Chemical group 0.000 claims description 6
- 125000005842 heteroatom Chemical group 0.000 claims description 6
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 claims description 6
- 125000001931 aliphatic group Chemical group 0.000 claims description 5
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 claims description 5
- 229910052760 oxygen Inorganic materials 0.000 claims description 5
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 5
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 claims description 4
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical group C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 claims description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical group [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 4
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 claims description 4
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 claims description 4
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 claims description 4
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 claims description 4
- 125000003368 amide group Chemical group 0.000 claims description 4
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 claims description 4
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 claims description 4
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 claims description 4
- 125000001624 naphthyl group Chemical group 0.000 claims description 4
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical compound C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 claims description 4
- 125000001725 pyrenyl group Chemical group 0.000 claims description 4
- 125000004076 pyridyl group Chemical group 0.000 claims description 4
- 229910052717 sulfur Inorganic materials 0.000 claims description 4
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 claims description 3
- 125000005843 halogen group Chemical group 0.000 claims description 3
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical group [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims description 2
- XQVWYOYUZDUNRW-UHFFFAOYSA-N N-Phenyl-1-naphthylamine Chemical group C=1C=CC2=CC=CC=C2C=1NC1=CC=CC=C1 XQVWYOYUZDUNRW-UHFFFAOYSA-N 0.000 claims description 2
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 claims description 2
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 claims description 2
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 2
- PJANXHGTPQOBST-VAWYXSNFSA-N Stilbene Natural products C=1C=CC=CC=1/C=C/C1=CC=CC=C1 PJANXHGTPQOBST-VAWYXSNFSA-N 0.000 claims description 2
- 125000004653 anthracenylene group Chemical group 0.000 claims description 2
- 239000004305 biphenyl Substances 0.000 claims description 2
- 235000010290 biphenyl Nutrition 0.000 claims description 2
- 125000006267 biphenyl group Chemical group 0.000 claims description 2
- 125000002529 biphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C12)* 0.000 claims description 2
- DMBHHRLKUKUOEG-UHFFFAOYSA-N diphenylamine Chemical group C=1C=CC=CC=1NC1=CC=CC=C1 DMBHHRLKUKUOEG-UHFFFAOYSA-N 0.000 claims description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 2
- 150000008282 halocarbons Chemical group 0.000 claims description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 2
- 125000002883 imidazolyl group Chemical group 0.000 claims description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 claims description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 2
- 125000004957 naphthylene group Chemical group 0.000 claims description 2
- 125000005565 oxadiazolylene group Chemical group 0.000 claims description 2
- 125000002971 oxazolyl group Chemical group 0.000 claims description 2
- 125000005564 oxazolylene group Chemical group 0.000 claims description 2
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 claims description 2
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 claims description 2
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 claims description 2
- 125000003373 pyrazinyl group Chemical group 0.000 claims description 2
- 125000005550 pyrazinylene group Chemical group 0.000 claims description 2
- 125000005548 pyrenylene group Chemical group 0.000 claims description 2
- 125000005551 pyridylene group Chemical group 0.000 claims description 2
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 2
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 claims description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 2
- 229910052710 silicon Inorganic materials 0.000 claims description 2
- 150000003413 spiro compounds Chemical class 0.000 claims description 2
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical compound C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 claims description 2
- 235000021286 stilbenes Nutrition 0.000 claims description 2
- 125000005504 styryl group Chemical group 0.000 claims description 2
- 125000006836 terphenylene group Chemical group 0.000 claims description 2
- VLLMWSRANPNYQX-UHFFFAOYSA-N thiadiazole Chemical compound C1=CSN=N1.C1=CSN=N1 VLLMWSRANPNYQX-UHFFFAOYSA-N 0.000 claims description 2
- 125000005557 thiazolylene group Chemical group 0.000 claims description 2
- 229930192474 thiophene Natural products 0.000 claims description 2
- 125000005730 thiophenylene group Chemical group 0.000 claims description 2
- 125000003944 tolyl group Chemical group 0.000 claims description 2
- 150000003852 triazoles Chemical class 0.000 claims description 2
- 125000001425 triazolyl group Chemical group 0.000 claims description 2
- 125000005559 triazolylene group Chemical group 0.000 claims description 2
- 125000006617 triphenylamine group Chemical group 0.000 claims description 2
- 150000001408 amides Chemical group 0.000 claims 2
- 150000002431 hydrogen Chemical class 0.000 claims 2
- 125000005561 phenanthryl group Chemical group 0.000 claims 2
- WONYVCKUEUULQN-UHFFFAOYSA-N 2-methyl-n-(2-methylphenyl)aniline Chemical group CC1=CC=CC=C1NC1=CC=CC=C1C WONYVCKUEUULQN-UHFFFAOYSA-N 0.000 claims 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 claims 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims 1
- 239000000463 material Substances 0.000 abstract description 43
- DZBUGLKDJFMEHC-UHFFFAOYSA-N acridine Chemical compound C1=CC=CC2=CC3=CC=CC=C3N=C21 DZBUGLKDJFMEHC-UHFFFAOYSA-N 0.000 abstract 2
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 63
- 239000000243 solution Substances 0.000 description 47
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 46
- 238000006243 chemical reaction Methods 0.000 description 45
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 44
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 42
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 27
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 25
- 229910052757 nitrogen Inorganic materials 0.000 description 24
- 239000012153 distilled water Substances 0.000 description 21
- 239000003208 petroleum Substances 0.000 description 21
- 238000004519 manufacturing process Methods 0.000 description 19
- 238000002360 preparation method Methods 0.000 description 19
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 18
- BZHGXQBPJUHVFW-UHFFFAOYSA-N toluene;tritert-butylphosphane Chemical compound CC1=CC=CC=C1.CC(C)(C)P(C(C)(C)C)C(C)(C)C BZHGXQBPJUHVFW-UHFFFAOYSA-N 0.000 description 17
- 230000015572 biosynthetic process Effects 0.000 description 14
- 230000005684 electric field Effects 0.000 description 12
- 238000001228 spectrum Methods 0.000 description 12
- 238000003786 synthesis reaction Methods 0.000 description 12
- 230000003287 optical effect Effects 0.000 description 11
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- 238000002474 experimental method Methods 0.000 description 10
- 238000000034 method Methods 0.000 description 9
- 239000000203 mixture Substances 0.000 description 9
- 229960005235 piperonyl butoxide Drugs 0.000 description 9
- RUFPHBVGCFYCNW-UHFFFAOYSA-N 1-naphthylamine Chemical compound C1=CC=C2C(N)=CC=CC2=C1 RUFPHBVGCFYCNW-UHFFFAOYSA-N 0.000 description 8
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 8
- 238000010992 reflux Methods 0.000 description 7
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 6
- DMVOXQPQNTYEKQ-UHFFFAOYSA-N biphenyl-4-amine Chemical group C1=CC(N)=CC=C1C1=CC=CC=C1 DMVOXQPQNTYEKQ-UHFFFAOYSA-N 0.000 description 6
- 230000021615 conjugation Effects 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- 238000000576 coating method Methods 0.000 description 5
- 238000000151 deposition Methods 0.000 description 5
- 230000003647 oxidation Effects 0.000 description 5
- 238000007254 oxidation reaction Methods 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- FYKJPLLUCCMVTQ-UHFFFAOYSA-N 1-n-naphthalen-1-yl-4-n,4-n-diphenylbenzene-1,4-diamine Chemical compound C=1C=CC2=CC=CC=C2C=1NC(C=C1)=CC=C1N(C=1C=CC=CC=1)C1=CC=CC=C1 FYKJPLLUCCMVTQ-UHFFFAOYSA-N 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- DWUOVFDSVGXTDO-UHFFFAOYSA-N CC1=CC=CC=C1.C(CCC)C(CCCP)(CCCC)CCCC Chemical compound CC1=CC=CC=C1.C(CCC)C(CCCP)(CCCC)CCCC DWUOVFDSVGXTDO-UHFFFAOYSA-N 0.000 description 4
- 230000008021 deposition Effects 0.000 description 4
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 4
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 4
- 229920001467 poly(styrenesulfonates) Polymers 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 239000007858 starting material Substances 0.000 description 4
- 239000010409 thin film Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- IUXIPAVGALBLKZ-UHFFFAOYSA-N C1=CC(Cl)=CC=C1C1=CC=CC=C1.C=1C=CC2=CC=CC=C2C=1NC1=CC=CC=C1 Chemical compound C1=CC(Cl)=CC=C1C1=CC=CC=C1.C=1C=CC2=CC=CC=C2C=1NC1=CC=CC=C1 IUXIPAVGALBLKZ-UHFFFAOYSA-N 0.000 description 3
- RBPQKCJTCHSTME-UHFFFAOYSA-N C=1C=CC=CC=1NC1=CC=CC=C1.C1=CC(Cl)=CC=C1C1=CC=CC=C1 Chemical compound C=1C=CC=CC=1NC1=CC=CC=C1.C1=CC(Cl)=CC=C1C1=CC=CC=C1 RBPQKCJTCHSTME-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 230000009477 glass transition Effects 0.000 description 3
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- 150000002894 organic compounds Chemical class 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- UJOBWOGCFQCDNV-UHFFFAOYSA-N 9H-carbazole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=C1 UJOBWOGCFQCDNV-UHFFFAOYSA-N 0.000 description 2
- 229920001609 Poly(3,4-ethylenedioxythiophene) Polymers 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 238000004776 molecular orbital Methods 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- 125000003367 polycyclic group Chemical group 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 125000003003 spiro group Chemical group 0.000 description 2
- 238000001771 vacuum deposition Methods 0.000 description 2
- KEOLYBMGRQYQTN-UHFFFAOYSA-N (4-bromophenyl)-phenylmethanone Chemical compound C1=CC(Br)=CC=C1C(=O)C1=CC=CC=C1 KEOLYBMGRQYQTN-UHFFFAOYSA-N 0.000 description 1
- CAYQIZIAYYNFCS-UHFFFAOYSA-N (4-chlorophenyl)boronic acid Chemical compound OB(O)C1=CC=C(Cl)C=C1 CAYQIZIAYYNFCS-UHFFFAOYSA-N 0.000 description 1
- 239000001211 (E)-4-phenylbut-3-en-2-one Substances 0.000 description 1
- OIRHKGBNGGSCGS-UHFFFAOYSA-N 1-bromo-2-iodobenzene Chemical compound BrC1=CC=CC=C1I OIRHKGBNGGSCGS-UHFFFAOYSA-N 0.000 description 1
- DMEVMYSQZPJFOK-UHFFFAOYSA-N 3,4,5,6,9,10-hexazatetracyclo[12.4.0.02,7.08,13]octadeca-1(18),2(7),3,5,8(13),9,11,14,16-nonaene Chemical group N1=NN=C2C3=CC=CC=C3C3=CC=NN=C3C2=N1 DMEVMYSQZPJFOK-UHFFFAOYSA-N 0.000 description 1
- SQTLUXJWUCHKMT-UHFFFAOYSA-N 4-bromo-n,n-diphenylaniline Chemical compound C1=CC(Br)=CC=C1N(C=1C=CC=CC=1)C1=CC=CC=C1 SQTLUXJWUCHKMT-UHFFFAOYSA-N 0.000 description 1
- FIPWRIJSWJWJAI-UHFFFAOYSA-N Butyl carbitol 6-propylpiperonyl ether Chemical compound C1=C(CCC)C(COCCOCCOCCCC)=CC2=C1OCO2 FIPWRIJSWJWJAI-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 239000003377 acid catalyst Substances 0.000 description 1
- 125000000641 acridinyl group Chemical class C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical group C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 229930008407 benzylideneacetone Natural products 0.000 description 1
- GBRBMTNGQBKBQE-UHFFFAOYSA-L copper;diiodide Chemical compound I[Cu]I GBRBMTNGQBKBQE-UHFFFAOYSA-L 0.000 description 1
- 238000002484 cyclic voltammetry Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 238000007641 inkjet printing Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 125000000468 ketone group Chemical group 0.000 description 1
- DLEDOFVPSDKWEF-UHFFFAOYSA-N lithium butane Chemical compound [Li+].CCC[CH2-] DLEDOFVPSDKWEF-UHFFFAOYSA-N 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- MZRVEZGGRBJDDB-UHFFFAOYSA-N n-Butyllithium Substances [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Chemical group C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 150000003509 tertiary alcohols Chemical class 0.000 description 1
- BWHOZHOGCMHOBV-BQYQJAHWSA-N trans-benzylideneacetone Chemical compound CC(=O)\C=C\C1=CC=CC=C1 BWHOZHOGCMHOBV-BQYQJAHWSA-N 0.000 description 1
- 238000002371 ultraviolet--visible spectrum Methods 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D219/00—Heterocyclic compounds containing acridine or hydrogenated acridine ring systems
- C07D219/02—Heterocyclic compounds containing acridine or hydrogenated acridine ring systems with only hydrogen, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/06—Peri-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
- C07F7/0812—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring
- C07F7/0816—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring said ring comprising Si as a ring atom
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/636—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising heteroaromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/17—Carrier injection layers
Abstract
Description
Claims (16)
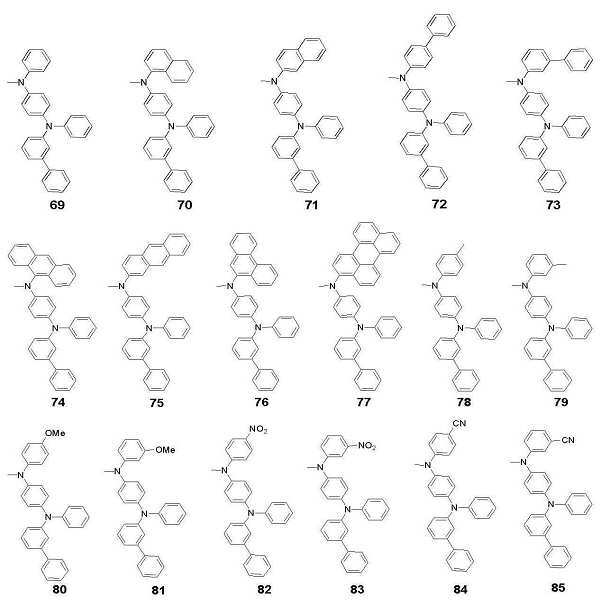
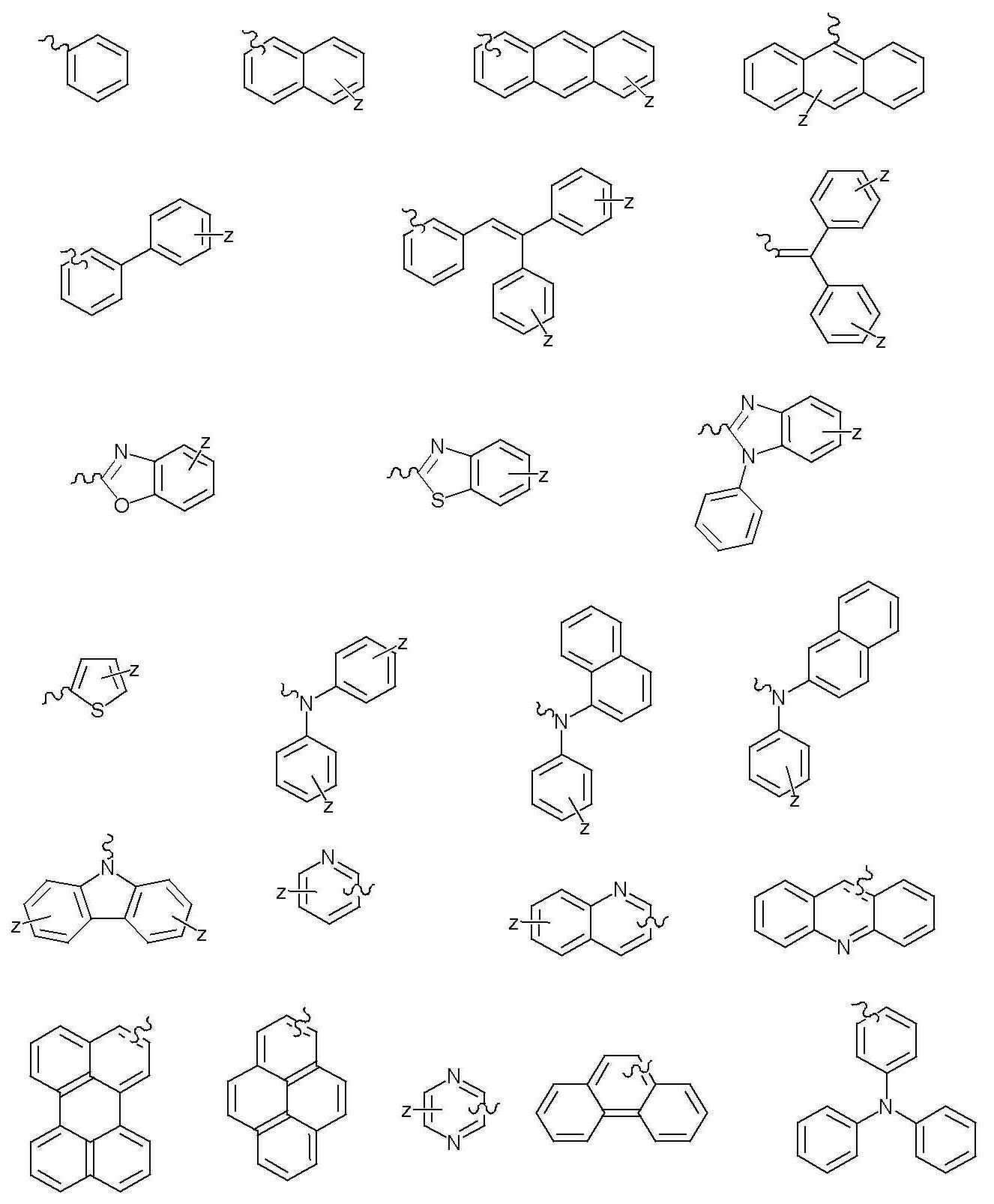
- 하기 화학식 1로 표시되는 화합물:[화학식 1]상기 화학식 1에 있어서,X는 C 또는 Si이고,Y는 직접 결합; 2가 방향족 탄화수소; 니트로, 니트릴, 할로겐, 알킬기, 알콕시기 및 아미노기로 이루어진 군으로부터 선택되는 1개 이상의 치환기로 치환된 2가 방향족 탄화수소; 2가 헤테로 고리기; 또는 니트로, 니트릴, 할로겐, 알킬기, 알콕시기 및 아미노기로 이루어진 군으로부터 선택되는 1개 이상의 치환기로 치환된 2가 헤테로 고리기이고,R1 내지 R11은 서로 동일하거나 상이하고, 각각 독립적으로 수소, 알킬기, 알콕시기, 알케닐기, 아릴기, 아릴아민기, 헤테로 고리기, 아미노기, 니트릴기, 니트로기, 할로겐기, 아미드기 또는 에스테르기이며, 여기서 이들은 서로 인접하는 기와 지방족, 방향족 또는 헤테로의 축합 고리를 형성할 수 있고,R12 내지 R16은 서로 동일하거나 상이하고, 각각 독립적으로 수소, 알킬기, 알콕시기, 알케닐기, 아릴기, 헤테로 고리기, 아미노기, 니트릴기, 니트로기, 할로겐기, 아미드기 또는 에스테르기이며, 여기서 이들은 서로 인접하는 기와 지방족, 방향족 또는 헤테로의 축합 고리를 형성할 수 있고,R7 및 R8는 직접 연결되거나, O, S, NR, PR, C=O, CRR' 및 SiRR'으로 이루어진 군에서 선택되는 기와 함께 축합고리를 형성할 수 있으며, 여기서 R 및 R'은 서로 동일하거나 상이하고, 각각 독립적으로 수소, 알킬기, 알콕시기, 알케닐기, 아릴기, 아릴 아민기, 헤테로 고리기, 니트릴기, 아미드기 또는 에스테르기이며, 여기서 R과 R'은 축합고리를 형성하여 스피로 화합물을 형성할 수 있고,p는 1 내지 5의 정수이고,a는 0 내지 10의 정수이고,Y1 및 Y2는 서로 동일하거나 상이하고, 각각 독립적으로 2가 방향족 탄화수소; 니트로, 니트릴, 할로겐, 알킬기, 알콕시기 및 아미노기로 이루어진 군으로부터 선택되는 1개 이상의 치환기로 치환된 2가 방향족 탄화수소; 2가 헤테로 고리기; 또는 니트로, 니트릴, 할로겐, 알킬기, 알콕시기 및 아미노기로 이루어진 군으로부터 선택되는 1개 이상의 치환기로 치환된 2가 헤테로 고리기이고,Z1 내지 Z4는 서로 동일하거나 상이하고, 각각 독립적으로 수소; 탄소수 1-20의 지방족 탄화수소; 방향족 탄화수소; 니트로, 니트릴, 할로겐, 알킬기, 알콕시기, 아미노기, 방향족 탄화수소 및 헤테로 고리기로 이루어진 군으로부터 선택되는 1개 이상의 치환기로 치환된 방항족 탄화수소; 방향족 탄화수소로 치환된 실리콘기; 헤테로고리기; 니트로, 니트릴, 할로겐, 알킬기, 알콕시기, 아미노기, 방향족 탄화수소 및 헤테로 고리기로 이루어진 군으로부터 선택되는 1개 이상의 치환기로 치환된 헤테로고리기; 탄소수 1 내지 20의 지방족 탄화수소 또는 탄소수 6 내지 20의 방향족 탄화수소로 치환된 티오펜기; 또는 방향족 탄화수소로 치환된 붕소기이다.
- 청구항 1에 있어서, 상기 화학식 1의 Y, Y1 및 Y2의 2가 방향족 탄화수소는 페닐렌, 비페닐렌, 터페닐렌, 나프틸렌, 안트라세닐렌, 파이레닐렌, 페릴레닐렌 및 페난트릴렌 중에서 선택되는 것을 특징으로 하는 화합물.
- 청구항 1에 있어서, 상기 화학식 1의 Y, Y1 및 Y2의 2가 헤테로고리기는 티오페닐렌, 퓨릴렌, 피롤릴렌, 이미다졸릴렌, 티아졸릴렌, 옥사졸릴렌, 옥사디아졸릴렌, 티아디아졸릴렌, 트리아졸릴렌, 피리딜렌, 피리다질렌, 피라지닐렌, 퀴놀릴렌 및 이소퀴놀릴렌 중에서 선택되는 것을 특징으로 하는 화합물.
- 청구항 1에 있어서, 상기 화학식 1의 Z1 내지 Z4의 방향족 탄화수소는 페닐, 비페닐, 터페닐, 나프틸, 안트라세닐, 파이레닐, 페릴레닐 및 페난트릴 중에서 선택되는 것을 특징으로 하는 화합물.
- 청구항 1에 있어서, 상기 화학식 1의 Z1 내지 Z4의 헤테로고리기는 티오펜, 퓨란, 피롤, 이미다졸, 티아졸, 옥사졸, 옥사디아졸, 티아디아졸, 트리아졸, 피리딜, 피리다질, 피라진, 퀴놀린 및 이소퀴놀린 중에서 선택되는 것을 특징으로 하는 화합물.
- 청구항 1에 있어서, 상기 화학식 1의 Z1 내지 Z4의 탄소수 1 내지 20의 지방족 탄화수소는 메틸기, 에틸기, 노말-프로필기, 이소-프로필기, 노말-부틸기, sec-부틸기, 이소-부틸기, 터-부틸기, 펜틸기, 헥실기, 스티릴 및 아세틸렌기 중에서 선택되는 것을 특징으로 하는 화합물.
- 청구항 1에 있어서, 상기 화학식 1의 R1 내지 R16의 아릴기는 페닐기, 비페닐기, 터페닐기, 스틸벤, 나프틸기, 안트라세닐기, 페나트렌기, 파이레닐기, 페릴레닐기 및 페난트릴기 중에서 선택되는 것을 특징으로 하는 화합물.
- 청구항 1에 있어서, 상기 화학식 1의 R1 내지 R11의 아릴아민기는 디페닐 아민기, 디나프틸 아민기, 디비페닐 아민기, 페닐 나프틸 아민기, 페닐 디페틸 아민기, 디톨릴 아민기, 페닐 톨릴 아민기, 카바졸기 및 트리페닐 아민기 중에서 선택되는 것을 특징으로 하는 화합물.
- 청구항 1에 있어서, 상기 화학식 1의 R1 내지 R16의 헤테로고리기는 티오펜기, 퓨란기, 피롤기, 이미다졸기, 티아졸기, 옥사졸기, 옥사디아졸기, 트리아졸기, 피리딜기, 피라다진기, 퀴놀리닐기, 이소퀴톨린기 및 아크리딜기 중에서 선택되는 것을 특징으로 하는 화합물.
- 제1 전극, 1층 이상의 유기물층 및 제2 전극을 적층된 형태로 포함하는 유기 발광 소자에 있어서, 상기 유기물층 중 1층 이상이 청구항 1 내지 청구항 12 중 어느 한 항의 화합물을 포함하는 것을 특징으로 하는 유기 발광 소자.
- 청구항 13에 있어서, 상기 유기물층은 정공 수송층을 포함하고, 이 정공 수송층이 화학식 1로 표시되는 화합물을 포함하는 것을 특징으로 하는 유기 발광 소자.
- 청구항 13에 있어서, 상기 유기물층은 정공 주입층을 포함하고, 이 정공 주입층이 화학식 1로 표시되는 화합물을 포함하는 것을 특징으로 하는 유기 발광 소자.
- 청구항 13에 있어서, 상기 유기물층은 정공 주입과 정공 수송을 동시에 하는 층을 포함하고, 이 층이 화학식 1로 표시되는 화합물을 포함하는 것을 특징으로 하는 유기 발광 소자.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020060120560A KR100939627B1 (ko) | 2006-12-01 | 2006-12-01 | 신규한 화합물 및 이를 이용한 유기 발광 소자 |
| US12/312,847 US8410475B2 (en) | 2006-12-01 | 2007-12-03 | Compounds and organic light emitting diode using the same |
| JP2009539190A JP5128610B2 (ja) | 2006-12-01 | 2007-12-03 | 新規な化合物およびこれを用いた有機発光素子 |
| PCT/KR2007/006176 WO2008066357A1 (en) | 2006-12-01 | 2007-12-03 | New compounds and organic light emitting diode using the same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020060120560A KR100939627B1 (ko) | 2006-12-01 | 2006-12-01 | 신규한 화합물 및 이를 이용한 유기 발광 소자 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20080049944A KR20080049944A (ko) | 2008-06-05 |
| KR100939627B1 true KR100939627B1 (ko) | 2010-01-29 |
Family
ID=39468092
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020060120560A KR100939627B1 (ko) | 2006-12-01 | 2006-12-01 | 신규한 화합물 및 이를 이용한 유기 발광 소자 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US8410475B2 (ko) |
| JP (1) | JP5128610B2 (ko) |
| KR (1) | KR100939627B1 (ko) |
| WO (1) | WO2008066357A1 (ko) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5615261B2 (ja) * | 2009-03-11 | 2014-10-29 | 学校法人関西学院 | 多環芳香族化合物 |
| DE102010005697A1 (de) * | 2010-01-25 | 2011-07-28 | Merck Patent GmbH, 64293 | Verbindungen für elektronische Vorrichtungen |
| JP5724588B2 (ja) * | 2011-04-28 | 2015-05-27 | コニカミノルタ株式会社 | 有機エレクトロルミネッセンス素子材料、有機エレクトロルミネッセンス素子、表示装置および照明装置 |
| KR102567881B1 (ko) * | 2017-11-15 | 2023-08-18 | 삼성디스플레이 주식회사 | 함질소 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR102254306B1 (ko) * | 2018-01-18 | 2021-05-20 | 주식회사 엘지화학 | 아크리딘 유도체 및 이를 포함하는 유기 발광 소자 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002069161A (ja) | 2000-08-29 | 2002-03-08 | National Institute Of Advanced Industrial & Technology | ケイ素で縮環されたtpdポリマー |
| JP2004250536A (ja) | 2003-02-19 | 2004-09-09 | Nagoya City | 蛍光顔料及びそれを含有する蛍光性樹脂 |
| US20040219386A1 (en) | 2003-04-29 | 2004-11-04 | Canon Kabushiki Kaisha | Heterogeneous spiro compounds in organic light emitting device elements |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05107784A (ja) * | 1991-04-29 | 1993-04-30 | Kao Corp | 電子写真感光体 |
| JP3079909B2 (ja) | 1993-10-01 | 2000-08-21 | 東洋インキ製造株式会社 | 正孔輸送材料およびその用途 |
| JP3984488B2 (ja) * | 2001-03-27 | 2007-10-03 | 日本ペイント株式会社 | 硬化性塗料組成物および塗膜形成方法 |
| JP4581355B2 (ja) | 2003-09-09 | 2010-11-17 | 東洋インキ製造株式会社 | 有機エレクトロルミネッセンス素子 |
| US7842404B2 (en) | 2004-09-24 | 2010-11-30 | Lg Chem, Ltd. | Compound and organic light emitting device using the same |
| WO2006080645A1 (en) * | 2004-09-24 | 2006-08-03 | Lg Chem. Ltd. | New compound and organic light emitting device using the same(9) |
| WO2006033563A1 (en) * | 2004-09-24 | 2006-03-30 | Lg Chem. Ltd. | Organic light emitting device |
-
2006
- 2006-12-01 KR KR1020060120560A patent/KR100939627B1/ko active IP Right Grant
-
2007
- 2007-12-03 JP JP2009539190A patent/JP5128610B2/ja active Active
- 2007-12-03 US US12/312,847 patent/US8410475B2/en active Active
- 2007-12-03 WO PCT/KR2007/006176 patent/WO2008066357A1/en active Application Filing
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002069161A (ja) | 2000-08-29 | 2002-03-08 | National Institute Of Advanced Industrial & Technology | ケイ素で縮環されたtpdポリマー |
| JP2004250536A (ja) | 2003-02-19 | 2004-09-09 | Nagoya City | 蛍光顔料及びそれを含有する蛍光性樹脂 |
| US20040219386A1 (en) | 2003-04-29 | 2004-11-04 | Canon Kabushiki Kaisha | Heterogeneous spiro compounds in organic light emitting device elements |
Also Published As
| Publication number | Publication date |
|---|---|
| US20100065827A1 (en) | 2010-03-18 |
| JP5128610B2 (ja) | 2013-01-23 |
| WO2008066357A1 (en) | 2008-06-05 |
| US8410475B2 (en) | 2013-04-02 |
| JP2010511605A (ja) | 2010-04-15 |
| KR20080049944A (ko) | 2008-06-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR100645030B1 (ko) | 새로운 유기 발광 소자 재료 및 이를 이용한 유기 발광소자(5) | |
| KR100645026B1 (ko) | 새로운 유기 발광 소자 재료 및 이를 이용한 유기 발광소자(6) | |
| KR100925323B1 (ko) | 신규한 화합물 및 이를 이용한 유기 발광 소자 | |
| KR20090051140A (ko) | 새로운 유기 발광 소자 재료 및 이를 이용한 유기 발광 소자 | |
| KR100939627B1 (ko) | 신규한 화합물 및 이를 이용한 유기 발광 소자 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| E902 | Notification of reason for refusal | ||
| E701 | Decision to grant or registration of patent right | ||
| GRNT | Written decision to grant | ||
| FPAY | Annual fee payment |
Payment date: 20130111 Year of fee payment: 4 |
|
| FPAY | Annual fee payment |
Payment date: 20140103 Year of fee payment: 5 |
|
| FPAY | Annual fee payment |
Payment date: 20150119 Year of fee payment: 6 |
|
| FPAY | Annual fee payment |
Payment date: 20160118 Year of fee payment: 7 |
|
| FPAY | Annual fee payment |
Payment date: 20161227 Year of fee payment: 8 |
|
| FPAY | Annual fee payment |
Payment date: 20180116 Year of fee payment: 9 |
|
| FPAY | Annual fee payment |
Payment date: 20190116 Year of fee payment: 10 |