JP7508317B2 - 塞栓コイル近位接続要素及び耐延伸性繊維 - Google Patents
塞栓コイル近位接続要素及び耐延伸性繊維 Download PDFInfo
- Publication number
- JP7508317B2 JP7508317B2 JP2020155306A JP2020155306A JP7508317B2 JP 7508317 B2 JP7508317 B2 JP 7508317B2 JP 2020155306 A JP2020155306 A JP 2020155306A JP 2020155306 A JP2020155306 A JP 2020155306A JP 7508317 B2 JP7508317 B2 JP 7508317B2
- Authority
- JP
- Japan
- Prior art keywords
- embolic coil
- opening
- embolic
- lumen
- stretch
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 230000003073 embolic effect Effects 0.000 title claims description 205
- 239000000835 fiber Substances 0.000 title claims description 104
- 239000007943 implant Substances 0.000 claims description 119
- 238000000034 method Methods 0.000 claims description 30
- 238000000926 separation method Methods 0.000 claims description 15
- 238000004804 winding Methods 0.000 claims description 15
- 239000000463 material Substances 0.000 claims description 14
- 238000005520 cutting process Methods 0.000 claims description 12
- 238000003466 welding Methods 0.000 claims description 2
- 206010002329 Aneurysm Diseases 0.000 description 36
- 238000011282 treatment Methods 0.000 description 15
- 238000005452 bending Methods 0.000 description 6
- 230000002028 premature Effects 0.000 description 5
- 230000017531 blood circulation Effects 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 238000002513 implantation Methods 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 230000009977 dual effect Effects 0.000 description 3
- 230000001732 thrombotic effect Effects 0.000 description 3
- 238000004873 anchoring Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 206010020565 Hyperaemia Diseases 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000023555 blood coagulation Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 230000035602 clotting Effects 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 210000005166 vasculature Anatomy 0.000 description 1
- 230000009724 venous congestion Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
- A61B17/12113—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
- A61B17/1215—Coils or wires comprising additional materials, e.g. thrombogenic, having filaments, having fibers, being coated
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
- A61B17/12154—Coils or wires having stretch limiting means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B2017/00477—Coupling
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B2017/00526—Methods of manufacturing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B2017/00831—Material properties
- A61B2017/00902—Material properties transparent or translucent
- A61B2017/00915—Material properties transparent or translucent for radioactive radiation
- A61B2017/0092—Material properties transparent or translucent for radioactive radiation for X-rays
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B2017/1205—Introduction devices
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B2017/1205—Introduction devices
- A61B2017/12054—Details concerning the detachment of the occluding device from the introduction device
Landscapes
- Health & Medical Sciences (AREA)
- Surgery (AREA)
- Life Sciences & Earth Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Molecular Biology (AREA)
- Vascular Medicine (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Reproductive Health (AREA)
- Medical Informatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Neurosurgery (AREA)
- Surgical Instruments (AREA)
Description
添付の図面と併せて以下の説明を参照して更に考察され、様々な図面において、同様の数字は、同様の構造要素及び特徴を示す。図面は、必ずしも縮尺どおりではなく、代わりに、本発明の原理を例示することが重視されている。図は、限定としてではなく単なる例示として、本発明の装置の1つ又は2つ以上の実装形態を描写している。
(1) 方法であって、
塞栓コイル及び耐延伸性繊維を含むインプラントの一部又は全てを動脈瘤内に位置決めすることと、
前記塞栓コイルの第1の部分を前記動脈瘤から後退させることと、
前記第1の部分を前記動脈瘤から後退させる際に、前記耐延伸性繊維で前記塞栓コイルの前記第1の部分の延長を制限することと、を含む、方法。
(2) 前記塞栓コイルの管腔内に延在させるように前記耐延伸性繊維を位置決めすることを更に含む、実施態様1に記載の方法。
(3) 前記塞栓コイルの第2の部分を屈曲させることと、
前記塞栓コイルの前記第2の部分を屈曲させながら、前記耐延伸性繊維を用いて、前記塞栓コイルの前記第2の部分内の巻線の分離を制限することと、を更に含む、実施態様1に記載の方法。
(4) 前記耐延伸性繊維に係合した鍵によって前記インプラントを送達システムに固設することと、
前記送達システムから前記鍵を解放し、それによって、前記インプラントを前記送達システムから解放することと、更に含む、実施態様1に記載の方法。
(5) 前記インプラントを前記送達システムに固設する前記工程が、
前記鍵を通して前記送達システムのループワイヤを位置決めすることと、
前記ループワイヤの開口部を通して前記送達システムのプルワイヤを位置決めすることと、を更に含み、
前記方法が、
前記鍵によって、前記ループ開口部に対して遠位方向に位置決めされた前記プルワイヤの第1の部分を支持することと、
前記鍵によって、前記ループ開口部に対して近位方向に位置決めされた前記プルワイヤの第2の部分を支持することと、を更に含む、実施態様4に記載の方法。
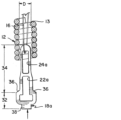
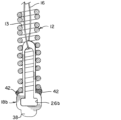
内部を通る管腔、近位端、及び遠位端を含む、塞栓コイルと、
前記塞栓コイルの前記近位端近くで前記塞栓コイルに貼着された脱離特徴部と、
前記脱離特徴部に係合し、前記塞栓コイルの前記管腔を通って延在し、前記塞栓コイルの前記遠位端近くで前記塞栓コイルに貼着された耐延伸性繊維と、を備え、
前記耐延伸性繊維は、前記塞栓コイルが再成形される際に、前記塞栓コイルの巻線の分離を制限するのに有効である、塞栓インプラント。
(7) 前記耐延伸性繊維が、縫合糸である、実施態様6に記載の塞栓インプラント。
(8) 前記耐延伸性繊維が、非弾性である、実施態様6に記載の塞栓インプラント。
(9) 前記脱離特徴部が、放射線不透過性である、実施態様6に記載の塞栓インプラント。
(10) 前記脱離特徴部が、内部を通る開口部を備え、
前記耐延伸性繊維が、前記脱離特徴部の前記開口部を通過し、
前記開口部が、前記塞栓コイルの前記近位端から近位に延在し、
前記開口部が、機械的送達システムのループワイヤを受容するようにサイズ決め及び位置決めされている、実施態様6に記載の塞栓インプラント。
前記耐延伸性繊維が、前記第1の開口部を通過し、
前記第1の開口部の少なくとも一部分が、前記塞栓コイルの前記管腔内に位置決めされており、
前記第2の開口部の少なくとも一部分が、前記塞栓コイルの前記近位端から近位に位置決めされている、実施態様6に記載の塞栓インプラント。
(12) システムであって、
実施態様11に記載の塞栓インプラントと、
前記第2の開口部を通して位置決めされたループワイヤ、及び前記ループワイヤの開口部を通して位置決めされたプルワイヤ、を含む、機械的送達システムと、を備え、
前記脱離特徴部が、前記第1の開口部と前記第2の開口部とを分離するブリッジを更に備え、
前記ブリッジが、前記ループワイヤの前記開口部から遠位方向に前記プルワイヤの一部分を支持する、システム。
(13) 前記塞栓コイルの前記管腔が、内径を含み、
前記脱離特徴部が、前記管腔から近位に配設された近位部分と、前記管腔内に配設された遠位部分と、を備え、
前記近位部分が、前記管腔の前記内径よりも大きい寸法の第1の幅を含み、
前記遠位部分が、前記管腔の前記内径とほぼ等しい寸法の第2の幅を含む、実施態様6に記載の塞栓インプラント。
(14) 方法であって、
脱離特徴部の第1の開口部に耐延伸性繊維を通すことと、
塞栓コイルの管腔を通して前記耐延伸性繊維を延在させることと、
前記塞栓コイルの第1の端部に前記脱離特徴部を貼着することと、
前記第1の端部とは反対側の前記塞栓コイルの前記第2の端部に前記耐延伸性繊維を貼着することと、
前記脱離特徴部と前記塞栓コイルの前記第2の端部との間に、前記耐延伸性繊維に沿って張力を提供することと、を含む、方法。
(15) 平坦なシート材料から前記脱離特徴部を切断することと、
前記脱離特徴部内の前記第1の開口部を切断することと、
前記第1の開口部(26b)を通して機械的展開システムの一部分を延在させて、前記脱離特徴部を送達チューブに係合させることと、を更に含む、実施態様14に記載の方法。
前記脱離特徴部内の前記第1の開口部を切断することと、
前記第1の開口部とは別個の、前記脱離特徴部内の第2の開口部を切断することと、
前記第2の開口部を通して機械的展開システムの一部分を延在させて、前記脱離特徴部を送達チューブに係合させることと、を更に含む、実施態様14に記載の方法。
(17) 放射線不透過性の平坦なシート材料から前記脱離特徴部を切断することを更に含む、実施態様14に記載の方法。
(18) 前記塞栓コイルの前記管腔内に前記脱離特徴部の遠位部分を挿入することと、
前記塞栓コイルの前記近位端から近位に前記脱離特徴部の近位部分を延在させることと、を更に含む、実施態様14に記載の方法。
(19) 前記管腔が内径を含むように前記塞栓コイルを選択することと、
前記脱離特徴部の前記近位部分が、前記管腔の前記内径よりも大きい寸法の第1の幅を含むように、かつ前記脱離特徴部の前記遠位部分が、前記管腔の前記内径とほぼ等しい寸法の第2の幅を含むように、前記脱離特徴部を選択することと、を更に含む、実施態様18に記載の方法。
(20) 前記塞栓コイルの第1の端部に前記脱離特徴部を貼着することが、前記塞栓コイルの前記第1の端部に前記脱離特徴部を溶接することを含む、実施態様14に記載の方法。
Claims (13)
- 塞栓インプラントであって、
内部を通る管腔、近位端、及び遠位端を含む、塞栓コイルと、
前記塞栓コイルの前記近位端近くで前記塞栓コイルに溶接された脱離特徴部と、
前記脱離特徴部に係合し、前記塞栓コイルの前記管腔を通って延在し、前記塞栓コイルの前記遠位端近くで前記塞栓コイルに貼着された耐延伸性繊維と、を備え、
前記塞栓コイルの前記管腔が、内径を含み、
前記脱離特徴部が、前記管腔から近位に配設された近位部分と、前記管腔内に配設された遠位部分と、を備え、
前記近位部分が、前記管腔の前記内径よりも大きい第1の幅を含み、
前記遠位部分が、前記管腔の前記内径とほぼ等しい第2の幅を有する幅広セクションと、前記管腔の前記内径よりも小さい第3の幅を有する幅狭セクションと、前記幅広セクションと前記幅狭セクションとの間のテーパ状セクションと、を含み、
前記耐延伸性繊維は、前記塞栓コイルが再成形される際に、前記塞栓コイルの巻線の分離を制限するのに有効である、塞栓インプラント。 - 前記耐延伸性繊維が、縫合糸である、請求項1に記載の塞栓インプラント。
- 前記耐延伸性繊維が、非弾性である、請求項1に記載の塞栓インプラント。
- 前記脱離特徴部が、放射線不透過性である、請求項1に記載の塞栓インプラント。
- 前記脱離特徴部が、内部を通る開口部を備え、
前記耐延伸性繊維が、前記脱離特徴部の前記開口部を通過し、
前記開口部が、前記塞栓コイルの前記近位端から近位に延在し、
前記開口部が、機械的送達システムのループワイヤを受容するようにサイズ決め及び位置決めされている、請求項1に記載の塞栓インプラント。 - 前記脱離特徴部が、内部を通る第1の開口部と、前記第1の開口部とは別個の、内部を通る第2の開口部と、を備え、
前記耐延伸性繊維が、前記第1の開口部を通過し、
前記第1の開口部の少なくとも一部分が、前記塞栓コイルの前記管腔内に位置決めされており、
前記第2の開口部の少なくとも一部分が、前記塞栓コイルの前記近位端から近位に位置決めされている、請求項1に記載の塞栓インプラント。 - システムであって、
請求項6に記載の塞栓インプラントと、
前記第2の開口部を通して位置決めされたループワイヤ、及び前記ループワイヤの開口部を通して位置決めされたプルワイヤ、を含む、機械的送達システムと、を備え、
前記脱離特徴部が、前記第1の開口部と前記第2の開口部とを分離するブリッジを更に備え、
前記ブリッジが、前記ループワイヤの前記開口部から遠位方向に前記プルワイヤの一部分を支持する、システム。 - 前記内径が第1の内径であり、
前記塞栓コイルの前記管腔が、非拡張部分と拡張部分とを含み、
前記拡張部分が前記第1の内径を有し、前記非拡張部分が前記第1の内径よりも小さい第2の内径を有する、請求項1に記載の塞栓インプラント。 - 方法であって、
脱離特徴部の第1の開口部に耐延伸性繊維を通すことと、
塞栓コイルの管腔を通して前記耐延伸性繊維を延在させることと、
前記塞栓コイルの近位端から近位に前記脱離特徴部の近位部分が延在するように、前記塞栓コイルの前記管腔内に前記脱離特徴部の遠位部分を挿入することと、
前記塞栓コイルの前記近位端に前記脱離特徴部を溶接することと、
前記塞栓コイルの遠位端に前記耐延伸性繊維を貼着することと、
前記脱離特徴部と前記塞栓コイルの前記遠位端との間に、前記耐延伸性繊維に沿って張力を提供することと、を含み、
前記塞栓コイルの前記管腔が、内径を含み、
前記近位部分が、前記管腔の前記内径よりも大きい第1の幅を含み、
前記遠位部分が、前記管腔の前記内径とほぼ等しい第2の幅を有する幅広セクションと、前記管腔の前記内径よりも小さい第3の幅を有する幅狭セクションと、前記幅広セクションと前記幅狭セクションとの間のテーパ状セクションと、を含む、方法。 - 平坦なシート材料から前記脱離特徴部を切断することと、
前記脱離特徴部内の前記第1の開口部を切断することと、
前記第1の開口部を通して機械的送達システムの一部分を延在させて、前記脱離特徴部を送達チューブに係合させることと、を更に含む、請求項9に記載の方法。 - 平坦なシート材料から前記脱離特徴部を切断することと、
前記脱離特徴部内の前記第1の開口部を切断することと、
前記第1の開口部とは別個の、前記脱離特徴部内の第2の開口部を切断することと、
前記第2の開口部を通して機械的送達システムの一部分を延在させて、前記脱離特徴部を送達チューブに係合させることと、を更に含む、請求項9に記載の方法。 - 放射線不透過性の平坦なシート材料から前記脱離特徴部を切断することを更に含む、請求項9に記載の方法。
- 前記内径が第1の内径であり、
前記塞栓コイルの前記管腔が、非拡張部分と拡張部分とを含み、
前記拡張部分が前記第1の内径を有し、前記非拡張部分が前記第1の内径よりも小さい第2の内径を有する、請求項9に記載の方法。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/573,469 | 2019-09-17 | ||
| US16/573,469 US11439403B2 (en) | 2019-09-17 | 2019-09-17 | Embolic coil proximal connecting element and stretch resistant fiber |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2021045547A JP2021045547A (ja) | 2021-03-25 |
| JP7508317B2 true JP7508317B2 (ja) | 2024-07-01 |
Family
ID=72560334
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020155306A Active JP7508317B2 (ja) | 2019-09-17 | 2020-09-16 | 塞栓コイル近位接続要素及び耐延伸性繊維 |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US11439403B2 (ja) |
| EP (1) | EP3795097B1 (ja) |
| JP (1) | JP7508317B2 (ja) |
| KR (1) | KR20210032908A (ja) |
| CN (1) | CN112515726B (ja) |
| ES (1) | ES2999631T3 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2021058586A (ja) * | 2019-10-03 | 2021-04-15 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 可撓性の耐伸張性機械的解放を有する医療装置送達部材 |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10806462B2 (en) | 2017-12-21 | 2020-10-20 | DePuy Synthes Products, Inc. | Implantable medical device detachment system with split tube and cylindrical coupling |
| US11253265B2 (en) | 2019-06-18 | 2022-02-22 | DePuy Synthes Products, Inc. | Pull wire detachment for intravascular devices |
| US11207494B2 (en) | 2019-07-03 | 2021-12-28 | DePuy Synthes Products, Inc. | Medical device delivery member with flexible stretch resistant distal portion |
| JPWO2023048142A1 (ja) * | 2021-09-24 | 2023-03-30 | ||
| US12508032B2 (en) | 2021-12-31 | 2025-12-30 | DePuy Synthes Products, Inc. | Medical device delivery systems with twisting loop wires |
| US12011171B2 (en) | 2022-01-06 | 2024-06-18 | DePuy Synthes Products, Inc. | Systems and methods for inhibiting premature embolic implant deployment |
| US11937825B2 (en) | 2022-03-02 | 2024-03-26 | DePuy Synthes Products, Inc. | Hook wire for preventing premature embolic implant detachment |
| US12471924B2 (en) | 2022-03-02 | 2025-11-18 | DePuy Synthes Products, Inc. | Flexible feature for embolic implant deployment |
| US11937826B2 (en) | 2022-03-14 | 2024-03-26 | DePuy Synthes Products, Inc. | Proximal link wire for preventing premature implant detachment |
| CN119789821A (zh) * | 2022-06-21 | 2025-04-08 | 库克医疗科技有限责任公司 | 栓塞线圈输送系统和部件 |
| US12402886B2 (en) | 2022-06-23 | 2025-09-02 | DePuy Synthes Products, Inc. | Detachment indicator for implant deployment |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012000464A (ja) | 2010-06-16 | 2012-01-05 | Codman & Shurtleff Inc | 伸長抵抗性部材及びアンカーフィラメントを備えた閉塞デバイス |
| JP2013212372A (ja) | 2012-03-30 | 2013-10-17 | Depuy Synthes Products Llc | 可撓性の遠位部材と結合継手とを有する塞栓用コイル取り外し機構 |
| JP2019111339A (ja) | 2017-12-21 | 2019-07-11 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 分割チューブと筒状継手とを備えた移植可能医療装置取り外しシステム |
Family Cites Families (220)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3429408A (en) | 1967-04-25 | 1969-02-25 | Associated Spring Corp | Actuator sleeves for spring clutch |
| US5484409A (en) | 1989-08-25 | 1996-01-16 | Scimed Life Systems, Inc. | Intravascular catheter and method for use thereof |
| US5122136A (en) | 1990-03-13 | 1992-06-16 | The Regents Of The University Of California | Endovascular electrolytically detachable guidewire tip for the electroformation of thrombus in arteries, veins, aneurysms, vascular malformations and arteriovenous fistulas |
| US5108407A (en) | 1990-06-08 | 1992-04-28 | Rush-Presbyterian St. Luke's Medical Center | Method and apparatus for placement of an embolic coil |
| US5636639A (en) | 1992-02-18 | 1997-06-10 | Symbiosis Corporation | Endoscopic multiple sample bioptome with enhanced biting action |
| US5263964A (en) | 1992-05-06 | 1993-11-23 | Coil Partners Ltd. | Coaxial traction detachment apparatus and method |
| US5350397A (en) | 1992-11-13 | 1994-09-27 | Target Therapeutics, Inc. | Axially detachable embolic coil assembly |
| US5250071A (en) | 1992-09-22 | 1993-10-05 | Target Therapeutics, Inc. | Detachable embolic coil assembly using interlocking clasps and method of use |
| US5382259A (en) | 1992-10-26 | 1995-01-17 | Target Therapeutics, Inc. | Vasoocclusion coil with attached tubular woven or braided fibrous covering |
| US5334210A (en) | 1993-04-09 | 1994-08-02 | Cook Incorporated | Vascular occlusion assembly |
| US5925059A (en) | 1993-04-19 | 1999-07-20 | Target Therapeutics, Inc. | Detachable embolic coil assembly |
| US5569221A (en) | 1994-07-07 | 1996-10-29 | Ep Technologies, Inc. | Catheter component bond and method |
| US6273404B1 (en) | 1995-06-05 | 2001-08-14 | Scimed Life Systems, Inc. | Method of making monolithic hub and strain relief |
| US5582619A (en) * | 1995-06-30 | 1996-12-10 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils |
| US5853418A (en) | 1995-06-30 | 1998-12-29 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils (II) |
| US6168622B1 (en) | 1996-01-24 | 2001-01-02 | Microvena Corporation | Method and apparatus for occluding aneurysms |
| US5899935A (en) | 1997-08-04 | 1999-05-04 | Schneider (Usa) Inc. | Balloon expandable braided stent with restraint |
| US6203547B1 (en) | 1997-12-19 | 2001-03-20 | Target Therapeutics, Inc. | Vaso-occlusion apparatus having a manipulable mechanical detachment joint and a method for using the apparatus |
| US6379374B1 (en) * | 1998-10-22 | 2002-04-30 | Cordis Neurovascular, Inc. | Small diameter embolic coil hydraulic deployment system |
| US6113622A (en) | 1998-03-10 | 2000-09-05 | Cordis Corporation | Embolic coil hydraulic deployment system |
| US6835185B2 (en) | 1998-12-21 | 2004-12-28 | Micrus Corporation | Intravascular device deployment mechanism incorporating mechanical detachment |
| US6391037B1 (en) | 2000-03-02 | 2002-05-21 | Prodesco, Inc. | Bag for use in the intravascular treatment of saccular aneurysms |
| US6689141B2 (en) * | 2000-10-18 | 2004-02-10 | Microvention, Inc. | Mechanism for the deployment of endovascular implants |
| US6623504B2 (en) | 2000-12-08 | 2003-09-23 | Scimed Life Systems, Inc. | Balloon catheter with radiopaque distal tip |
| DE60224502T2 (de) | 2001-01-10 | 2008-12-24 | Cordis Neurovascular, Inc., Miami Lakes | System zum Einführen einer Emboliespirale |
| US6454780B1 (en) | 2001-06-21 | 2002-09-24 | Scimed Life Systems, Inc. | Aneurysm neck obstruction device |
| US8252040B2 (en) | 2001-07-20 | 2012-08-28 | Microvention, Inc. | Aneurysm treatment device and method of use |
| US8715312B2 (en) | 2001-07-20 | 2014-05-06 | Microvention, Inc. | Aneurysm treatment device and method of use |
| US20030195553A1 (en) | 2002-04-12 | 2003-10-16 | Scimed Life Systems, Inc. | System and method for retaining vaso-occlusive devices within an aneurysm |
| US20040002732A1 (en) * | 2002-06-27 | 2004-01-01 | Clifford Teoh | Stretch-resistant vaso-occlusive assembly with multiple detaching points |
| US7608058B2 (en) | 2002-07-23 | 2009-10-27 | Micrus Corporation | Stretch resistant therapeutic device |
| US8425549B2 (en) | 2002-07-23 | 2013-04-23 | Reverse Medical Corporation | Systems and methods for removing obstructive matter from body lumens and treating vascular defects |
| US7208003B2 (en) | 2002-09-20 | 2007-04-24 | Cordis Neurovascular, Inc. | Reattachable introducer for a medical device deployment system |
| FR2853521B1 (fr) | 2003-04-10 | 2005-12-02 | Claude Mialhe | Dispositif de dilatation d'un vaisseau et introducteur d'implant vasculaire |
| US7371228B2 (en) | 2003-09-19 | 2008-05-13 | Medtronic Vascular, Inc. | Delivery of therapeutics to treat aneurysms |
| US20050149108A1 (en) | 2003-12-17 | 2005-07-07 | Microvention, Inc. | Implant delivery and detachment system and method |
| US9308382B2 (en) | 2004-06-10 | 2016-04-12 | Medtronic Urinary Solutions, Inc. | Implantable pulse generator systems and methods for providing functional and/or therapeutic stimulation of muscles and/or nerves and/or central nervous system tissue |
| US20060025802A1 (en) | 2004-07-30 | 2006-02-02 | Sowers William W | Embolic coil delivery system with U-shaped fiber release mechanism |
| US9655633B2 (en) | 2004-09-10 | 2017-05-23 | Penumbra, Inc. | System and method for treating ischemic stroke |
| WO2006052322A2 (en) | 2004-09-22 | 2006-05-18 | Guterman Lee R | Cranial aneurysm treatment arrangement |
| US20060089637A1 (en) | 2004-10-14 | 2006-04-27 | Werneth Randell L | Ablation catheter |
| US8562672B2 (en) | 2004-11-19 | 2013-10-22 | Medtronic, Inc. | Apparatus for treatment of cardiac valves and method of its manufacture |
| US8425550B2 (en) | 2004-12-01 | 2013-04-23 | Boston Scientific Scimed, Inc. | Embolic coils |
| US7238423B2 (en) * | 2004-12-20 | 2007-07-03 | Kimberly-Clark Worldwide, Inc. | Multicomponent fiber including elastic elements |
| US7819892B2 (en) | 2005-06-02 | 2010-10-26 | Codman & Shurtleff, Inc. | Embolic coil delivery system with spring wire release mechanism |
| US7367987B2 (en) | 2005-06-02 | 2008-05-06 | Cordis Neurovascular, Inc. | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US7799052B2 (en) | 2005-06-02 | 2010-09-21 | Codman & Shurtleff, Inc. | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US7985238B2 (en) | 2005-06-02 | 2011-07-26 | Codman & Shurtleff, Inc. | Embolic coil delivery system with spring wire release mechanism |
| US7811305B2 (en) | 2005-06-02 | 2010-10-12 | Codman & Shurtleff, Inc. | Stretch resistant embolic coil delivery system with spring release mechanism |
| US7371251B2 (en) | 2005-06-02 | 2008-05-13 | Cordis Neurovascular, Inc. | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US20060276833A1 (en) | 2005-06-02 | 2006-12-07 | Keith Balgobin | Stretch resistant embolic coil delivery system with spring assisted release mechanism |
| US7819891B2 (en) | 2005-06-02 | 2010-10-26 | Codman & Shurtleff, Inc. | Stretch resistant embolic coil delivery system with spring release mechanism |
| US20060276825A1 (en) | 2005-06-02 | 2006-12-07 | Vladimir Mitelberg | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US7377932B2 (en) | 2005-06-02 | 2008-05-27 | Cordis Neurovascular, Inc. | Embolic coil delivery system with mechanical release mechanism |
| US20060276826A1 (en) | 2005-06-02 | 2006-12-07 | Vladimir Mitelberg | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US7708754B2 (en) | 2005-06-02 | 2010-05-04 | Codman & Shurtleff, Pc | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US20060276830A1 (en) | 2005-06-02 | 2006-12-07 | Keith Balgobin | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US7708755B2 (en) | 2005-06-02 | 2010-05-04 | Codman & Shurtleff Inc. | Stretch resistant embolic coil delivery system with combined mechanical and pressure release mechanism |
| US7371252B2 (en) | 2005-06-02 | 2008-05-13 | Cordis Neurovascular, Inc. | Stretch resistant embolic coil delivery system with mechanical release mechanism |
| US9636115B2 (en) | 2005-06-14 | 2017-05-02 | Stryker Corporation | Vaso-occlusive delivery device with kink resistant, flexible distal end |
| AU2006262447A1 (en) | 2005-06-20 | 2007-01-04 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US20070083132A1 (en) | 2005-10-11 | 2007-04-12 | Sharrow James S | Medical device coil |
| EP1973680B1 (en) | 2005-11-17 | 2018-01-10 | Microvention, Inc. | Three-dimensional complex coil |
| US7942894B2 (en) | 2006-01-31 | 2011-05-17 | Codman & Shurtleff, Inc. | Embolic device delivery system |
| US9757260B2 (en) | 2006-03-30 | 2017-09-12 | Medtronic Vascular, Inc. | Prosthesis with guide lumen |
| US7766933B2 (en) | 2006-03-31 | 2010-08-03 | Codman & Shurtleff, Inc. | Stretch resistant design for embolic coils with stabilization bead |
| US7553321B2 (en) | 2006-03-31 | 2009-06-30 | Cordis Development Corporation | Chemically based vascular occlusion device deployment |
| US9615832B2 (en) | 2006-04-07 | 2017-04-11 | Penumbra, Inc. | Aneurysm occlusion system and method |
| US8777979B2 (en) | 2006-04-17 | 2014-07-15 | Covidien Lp | System and method for mechanically positioning intravascular implants |
| EP2027729A2 (en) | 2006-06-15 | 2009-02-25 | MicroVention, Inc. | Embolization device constructed from expansible polymer |
| US7934506B2 (en) | 2006-06-21 | 2011-05-03 | Koninklijke Philips Electronics N.V. | System and method for temporary tongue suspension |
| US8062325B2 (en) | 2006-07-31 | 2011-11-22 | Codman & Shurtleff, Inc. | Implantable medical device detachment system and methods of using the same |
| US8366720B2 (en) | 2006-07-31 | 2013-02-05 | Codman & Shurtleff, Inc. | Interventional medical device system having an elongation retarding portion and method of using the same |
| US7901444B2 (en) | 2006-09-29 | 2011-03-08 | Codman & Shurtleff, Inc. | Embolic coil delivery system with mechanical release mechanism |
| EP2076214A2 (en) * | 2006-10-03 | 2009-07-08 | Alure Medical, Inc. | Minimally invasive tissue support |
| US8926650B2 (en) | 2006-11-20 | 2015-01-06 | Boston Scientific Scimed, Inc. | Mechanically detachable vaso-occlusive device |
| WO2008064205A2 (en) | 2006-11-20 | 2008-05-29 | Boston Scientific Limited | Mechanically detachable vaso-occlusive device |
| WO2008064209A1 (en) | 2006-11-20 | 2008-05-29 | Boston Scientific Scimed, Inc. | Mechanically detachable vaso-occlusive device |
| US20080281350A1 (en) | 2006-12-13 | 2008-11-13 | Biomerix Corporation | Aneurysm Occlusion Devices |
| US8197442B2 (en) | 2007-04-27 | 2012-06-12 | Codman & Shurtleff, Inc. | Interventional medical device system having a slotted section and radiopaque marker and method of making the same |
| EP2444010B1 (en) | 2007-05-18 | 2017-03-01 | Stryker European Holdings I, LLC | Medical implant detachment systems |
| DE102007038446A1 (de) | 2007-08-14 | 2009-02-19 | pfm Produkte für die Medizin AG | Embolisiereinrichtung |
| US20090099592A1 (en) | 2007-10-15 | 2009-04-16 | Boston Scientific Scimed, Inc. | Detachable Interlock Systems and Methods of Use |
| WO2009086208A2 (en) | 2007-12-21 | 2009-07-09 | Microvention, Inc. | Hydrogel filaments for biomedical uses |
| US8974518B2 (en) | 2008-03-25 | 2015-03-10 | Medtronic Vascular, Inc. | Eversible branch stent-graft and deployment method |
| US20090276022A1 (en) | 2008-04-30 | 2009-11-05 | Medtronic , Inc. | Techniques for placing medical leads for electrical stimulation of nerve tissue |
| US20090312748A1 (en) | 2008-06-11 | 2009-12-17 | Johnson Kirk L | Rotational detachment mechanism |
| US8070694B2 (en) | 2008-07-14 | 2011-12-06 | Medtronic Vascular, Inc. | Fiber based medical devices and aspiration catheters |
| US8333796B2 (en) | 2008-07-15 | 2012-12-18 | Penumbra, Inc. | Embolic coil implant system and implantation method |
| US9232992B2 (en) | 2008-07-24 | 2016-01-12 | Aga Medical Corporation | Multi-layered medical device for treating a target site and associated method |
| US8721714B2 (en) | 2008-09-17 | 2014-05-13 | Medtronic Corevalve Llc | Delivery system for deployment of medical devices |
| CN102186426B (zh) * | 2008-10-13 | 2013-05-15 | 斯瑞克公司 | 血管闭塞线圈输送系统 |
| US20100160944A1 (en) * | 2008-12-24 | 2010-06-24 | Boston Scientific Scimed, Inc. | Thermally detachable embolic assemblies |
| AU2010236337B2 (en) | 2009-04-15 | 2015-01-29 | Microvention, Inc. | Implant delivery system |
| EP2421482B1 (en) | 2009-04-20 | 2019-11-13 | Achieva Medical Limited | Delivery assembly for occlusion device using mechanical interlocking coupling mechanism |
| US8758423B2 (en) | 2009-06-18 | 2014-06-24 | Graftcraft I Goteborg Ab | Device and method for treating ruptured aneurysms |
| US8911487B2 (en) | 2009-09-22 | 2014-12-16 | Penumbra, Inc. | Manual actuation system for deployment of implant |
| KR20110043799A (ko) | 2009-10-16 | 2011-04-28 | 강호창 | 마이크로코일 어셈블리 |
| CN102188300B (zh) | 2010-03-02 | 2014-05-28 | 上海微创医疗器械(集团)有限公司 | 一种动脉瘤手术装置 |
| CN103037776B (zh) | 2010-04-14 | 2017-07-04 | 微排放器公司 | 植入物输送装置 |
| US8764811B2 (en) | 2010-04-20 | 2014-07-01 | Medtronic Vascular, Inc. | Controlled tip release stent graft delivery system and method |
| US8876878B2 (en) | 2010-07-23 | 2014-11-04 | Medtronic, Inc. | Attachment mechanism for stent release |
| US8616040B2 (en) | 2010-09-17 | 2013-12-31 | Medtronic Vascular, Inc. | Method of forming a drug-eluting medical device |
| EP2654820A1 (en) | 2010-12-20 | 2013-10-30 | Microvention, Inc. | Polymer stents and methods of manufacture |
| US20120283768A1 (en) | 2011-05-05 | 2012-11-08 | Sequent Medical Inc. | Method and apparatus for the treatment of large and giant vascular defects |
| US9486604B2 (en) | 2011-05-12 | 2016-11-08 | Medtronic, Inc. | Packaging and preparation tray for a delivery system |
| CN106333749B (zh) | 2011-05-13 | 2020-01-03 | 斯波瑞申有限公司 | 展开导管 |
| WO2012158668A1 (en) | 2011-05-17 | 2012-11-22 | Stryker Corporation | Method of fabricating an implantable medical device that includes one or more thin film polymer support layers |
| WO2012166467A1 (en) | 2011-05-27 | 2012-12-06 | Stryker Corporation | Assembly for percutaneously inserting an implantable medical device, steering the device to a target location and deploying the device |
| US9750565B2 (en) | 2011-09-30 | 2017-09-05 | Medtronic Advanced Energy Llc | Electrosurgical balloons |
| CN104487024B (zh) | 2012-03-16 | 2017-08-29 | 微仙美国有限公司 | 支架和支架送递装置 |
| US9717421B2 (en) | 2012-03-26 | 2017-08-01 | Medtronic, Inc. | Implantable medical device delivery catheter with tether |
| US9833625B2 (en) | 2012-03-26 | 2017-12-05 | Medtronic, Inc. | Implantable medical device delivery with inner and outer sheaths |
| US9155540B2 (en) | 2012-03-30 | 2015-10-13 | DePuy Synthes Products, Inc. | Embolic coil detachment mechanism with heating element and kicker |
| US8920459B2 (en) * | 2012-03-30 | 2014-12-30 | DePuy Synthes Products, LLC | Embolic coil detachment mechanism with flexible distal member and resistive electrical heating element |
| US9242290B2 (en) | 2012-04-03 | 2016-01-26 | Medtronic Vascular, Inc. | Method and apparatus for creating formed elements used to make wound stents |
| US9549832B2 (en) | 2012-04-26 | 2017-01-24 | Medtronic Vascular, Inc. | Apparatus and methods for filling a drug eluting medical device via capillary action |
| US9700399B2 (en) | 2012-04-26 | 2017-07-11 | Medtronic Vascular, Inc. | Stopper to prevent graft material slippage in a closed web stent-graft |
| US9149190B2 (en) | 2012-07-17 | 2015-10-06 | Stryker Corporation | Notification system of deviation from predefined conditions |
| WO2014028528A1 (en) | 2012-08-13 | 2014-02-20 | Microvention, Inc. | Shaped removal device |
| US20140058435A1 (en) | 2012-08-21 | 2014-02-27 | Donald K. Jones | Implant delivery and release system |
| US9504476B2 (en) | 2012-10-01 | 2016-11-29 | Microvention, Inc. | Catheter markers |
| AU2013331439B2 (en) | 2012-10-15 | 2016-05-12 | Microvention, Inc. | Polymeric treatment compositions |
| CN108354645B (zh) | 2012-11-13 | 2026-03-13 | 柯惠有限合伙公司 | 封堵装置 |
| US9539022B2 (en) | 2012-11-28 | 2017-01-10 | Microvention, Inc. | Matter conveyance system |
| EP2928550B1 (en) | 2012-12-07 | 2023-06-07 | Medtronic, Inc. | Minimally invasive implantable neurostimulation system |
| US9943313B2 (en) | 2013-01-03 | 2018-04-17 | Empirilon Technology Llc | Detachable coil release system and handle assembly |
| US10342546B2 (en) | 2013-01-14 | 2019-07-09 | Microvention, Inc. | Occlusive device |
| GB2509952B (en) | 2013-01-18 | 2015-01-28 | Cook Medical Technologies Llc | Medical device loading and carrier tool |
| US9539382B2 (en) | 2013-03-12 | 2017-01-10 | Medtronic, Inc. | Stepped catheters with flow restrictors and infusion systems using the same |
| CN107468298A (zh) | 2013-03-14 | 2017-12-15 | 因库麦迪斯有限公司 | 可植入组件及制造可植入组件的方法 |
| WO2014150824A1 (en) | 2013-03-14 | 2014-09-25 | Stryker Corporation | Vaso-occlusive device delivery system |
| EP2967573B1 (en) | 2013-03-14 | 2021-04-21 | Stryker Corporation | Vaso-occlusive device delivery system |
| EP3009084B1 (en) | 2013-03-14 | 2017-09-13 | Stryker Corporation | Vaso-occlusive device delivery system |
| KR102366362B1 (ko) | 2013-03-15 | 2022-02-23 | 테루모 코퍼레이션 | 색전 방지 장치 |
| US9833252B2 (en) | 2013-03-15 | 2017-12-05 | Microvention, Inc. | Multi-component obstruction removal system and method |
| US9398966B2 (en) | 2013-03-15 | 2016-07-26 | Medtronic Vascular, Inc. | Welded stent and stent delivery system |
| ES2717678T3 (es) | 2013-04-22 | 2019-06-24 | Stryker European Holdings I Llc | Procedimiento para la carga de fármacos sobre superficies de implantes recubiertos de hidroxiapatita |
| US9445928B2 (en) | 2013-05-30 | 2016-09-20 | Medtronic Vascular, Inc. | Delivery system having a single handed deployment handle for a retractable outer sheath |
| US9662120B2 (en) | 2013-08-23 | 2017-05-30 | Cook Medical Technologies Llc | Detachable treatment device delivery system utilizing compression at attachment zone |
| US9675782B2 (en) | 2013-10-10 | 2017-06-13 | Medtronic Vascular, Inc. | Catheter pull wire actuation mechanism |
| US9439827B2 (en) | 2013-10-25 | 2016-09-13 | Medtronic Vascular, Inc. | Tissue compression device with pressure indicator |
| US9833604B2 (en) | 2013-12-20 | 2017-12-05 | Microvention, Inc. | Delivery adapter |
| WO2015095806A2 (en) | 2013-12-20 | 2015-06-25 | Microvention, Inc. | Device delivery system |
| US9566072B2 (en) | 2013-12-27 | 2017-02-14 | Blockade Medical, LLC | Coil system |
| US9788839B2 (en) | 2014-02-14 | 2017-10-17 | Cook Medical Technologies Llc | Stable screw-type detachment mechanism |
| US9775732B2 (en) | 2014-04-08 | 2017-10-03 | Stryker Corporation | Implant delivery system and method of use |
| WO2015167997A1 (en) | 2014-04-30 | 2015-11-05 | Stryker Corporation | Implant delivery system and method of use |
| US9060777B1 (en) | 2014-05-28 | 2015-06-23 | Tw Medical Technologies, Llc | Vaso-occlusive devices and methods of use |
| US9668898B2 (en) | 2014-07-24 | 2017-06-06 | Medtronic Vascular, Inc. | Stent delivery system having dynamic deployment and methods of manufacturing same |
| CA2955953A1 (en) | 2014-07-25 | 2016-01-28 | Incumedx, Inc. | Covered embolic coils |
| US9918718B2 (en) | 2014-08-08 | 2018-03-20 | DePuy Synthes Products, Inc. | Embolic coil delivery system with retractable mechanical release mechanism |
| US9770577B2 (en) | 2014-09-15 | 2017-09-26 | Medtronic Xomed, Inc. | Pressure relief for a catheter balloon device |
| RU2721288C2 (ru) | 2014-09-17 | 2020-05-18 | Метэктив Медикал, Инк. | Медицинское устройство для лечения мешотчатых аневризм |
| US9579484B2 (en) | 2014-09-19 | 2017-02-28 | Medtronic Vascular, Inc. | Sterile molded dispenser |
| GB2533087B (en) | 2014-12-08 | 2018-08-08 | Cook Medical Technologies Llc | Medical implant detachment mechanism and introducer assembly |
| US10925611B2 (en) | 2015-01-20 | 2021-02-23 | Neurogami Medical, Inc. | Packaging for surgical implant |
| US10857012B2 (en) | 2015-01-20 | 2020-12-08 | Neurogami Medical, Inc. | Vascular implant |
| US9692557B2 (en) | 2015-02-04 | 2017-06-27 | Stryker European Holdings I, Llc | Apparatus and methods for administering treatment within a bodily duct of a patient |
| EP3256058A1 (en) | 2015-02-10 | 2017-12-20 | Boston Scientific Scimed, Inc. | Active release of embolic coils |
| EP3291773B1 (en) | 2015-05-07 | 2026-01-07 | The Medical Research, Infrastructure, And Health Services Fund Of The Tel Aviv Medical Center | Temporary interatrial shunts |
| US9717503B2 (en) | 2015-05-11 | 2017-08-01 | Covidien Lp | Electrolytic detachment for implant delivery systems |
| US10398874B2 (en) | 2015-05-29 | 2019-09-03 | Covidien Lp | Catheter distal tip configuration |
| US10154905B2 (en) | 2015-08-07 | 2018-12-18 | Medtronic Vascular, Inc. | System and method for deflecting a delivery catheter |
| US10307168B2 (en) | 2015-08-07 | 2019-06-04 | Terumo Corporation | Complex coil and manufacturing techniques |
| JP6735814B2 (ja) | 2015-08-11 | 2020-08-05 | テルモ株式会社 | インプラント配送のためのシステムおよび方法 |
| JP6816126B2 (ja) | 2015-09-18 | 2021-01-20 | マイクロベンション インコーポレイテッドMicrovention, Inc. | 解放可能な送達システム |
| EP3349689B1 (en) | 2015-09-18 | 2023-12-27 | Microvention, Inc. | Implant retention, detachment, and delivery system |
| JP6816127B2 (ja) | 2015-09-18 | 2021-01-20 | テルモ株式会社 | 血管プロテーゼ |
| EP4327786A3 (en) | 2015-09-18 | 2024-05-01 | Terumo Corporation | Pushable implant delivery system |
| ES2788738T3 (es) | 2015-09-21 | 2020-10-22 | Stryker Corp | Dispositivos de embolectomía |
| ES2716926T3 (es) | 2015-09-21 | 2019-06-18 | Stryker Corp | Dispositivos de embolectomía |
| US10172632B2 (en) | 2015-09-22 | 2019-01-08 | Medtronic Vascular, Inc. | Occlusion bypassing apparatus with a re-entry needle and a stabilization tube |
| US20170100143A1 (en) | 2015-10-07 | 2017-04-13 | Stryker Corporation | Multiple barrel clot removal devices |
| US10327791B2 (en) | 2015-10-07 | 2019-06-25 | Medtronic Vascular, Inc. | Occlusion bypassing apparatus with a re-entry needle and a distal stabilization balloon |
| US10786302B2 (en) | 2015-10-09 | 2020-09-29 | Medtronic, Inc. | Method for closure and ablation of atrial appendage |
| WO2017066386A1 (en) | 2015-10-14 | 2017-04-20 | Three Rivers Medical Inc. | Mechanical embolization delivery apparatus and methods |
| US10271873B2 (en) | 2015-10-26 | 2019-04-30 | Medtronic Vascular, Inc. | Sheathless guide catheter assembly |
| WO2017087816A1 (en) | 2015-11-19 | 2017-05-26 | Penumbra, Inc. | Systems and methods for treatment of stroke |
| US10631946B2 (en) | 2015-11-30 | 2020-04-28 | Penumbra, Inc. | System for endoscopic intracranial procedures |
| WO2017100472A1 (en) | 2015-12-09 | 2017-06-15 | Medtronic Vascular Inc. | Catheter with a lumen shaped as an identification symbol |
| US10159568B2 (en) | 2015-12-14 | 2018-12-25 | Medtronic, Inc. | Delivery system having retractable wires as a coupling mechanism and a deployment mechanism for a self-expanding prosthesis |
| US10500046B2 (en) | 2015-12-14 | 2019-12-10 | Medtronic, Inc. | Delivery system having retractable wires as a coupling mechanism and a deployment mechanism for a self-expanding prosthesis |
| US10743883B2 (en) | 2015-12-30 | 2020-08-18 | Stryker European Holdings I, Llc | Embolic devices and methods of manufacturing same |
| US20170189033A1 (en) | 2016-01-06 | 2017-07-06 | Microvention, Inc. | Occlusive Embolic Coil |
| US10070950B2 (en) | 2016-02-09 | 2018-09-11 | Medtronic Vascular, Inc. | Endoluminal prosthetic assemblies, and associated systems and methods for percutaneous repair of a vascular tissue defect |
| EP3413808B1 (en) | 2016-02-10 | 2023-07-26 | Microvention, Inc. | Devices for vascular occlusion |
| EP3413963A4 (en) | 2016-02-10 | 2019-09-18 | Microvention, Inc. | ACCESS TO INTRAVASCULAR TREATMENT CENTER |
| US10188500B2 (en) | 2016-02-12 | 2019-01-29 | Medtronic Vascular, Inc. | Stent graft with external scaffolding and method |
| US10485579B2 (en) | 2016-02-25 | 2019-11-26 | Indian Wells Medical, Inc. | Steerable endoluminal punch |
| US20170258476A1 (en) | 2016-03-08 | 2017-09-14 | Terumo Kabushiki Kaisha | Blood vessel treatment method |
| CN108601647B (zh) | 2016-03-31 | 2020-10-02 | 美敦力瓦斯科尔勒公司 | 具有流体吸收性成分的修复脉管组织缺损用腔内假体装置 |
| CN108883254B (zh) | 2016-03-31 | 2022-03-29 | 美敦力瓦斯科尔勒公司 | 具有转向机构的可扩展导引器护套 |
| US10695542B2 (en) | 2016-04-04 | 2020-06-30 | Medtronic Vascular, Inc. | Drug coated balloon |
| US10252024B2 (en) | 2016-04-05 | 2019-04-09 | Stryker Corporation | Medical devices and methods of manufacturing same |
| US10441407B2 (en) | 2016-04-12 | 2019-10-15 | Medtronic Vascular, Inc. | Gutter filling stent-graft and method |
| US9987122B2 (en) | 2016-04-13 | 2018-06-05 | Medtronic Vascular, Inc. | Iliac branch device and method |
| US10010403B2 (en) | 2016-04-18 | 2018-07-03 | Medtronic Vascular, Inc. | Stent-graft prosthesis and method of manufacture |
| US20170304097A1 (en) | 2016-04-21 | 2017-10-26 | Medtronic Vascular, Inc. | Stent-graft delivery system having an inner shaft component with a loading pad or covering on a distal segment thereof for stent retention |
| US10517711B2 (en) | 2016-04-25 | 2019-12-31 | Medtronic Vascular, Inc. | Dissection prosthesis system and method |
| US10888342B2 (en) | 2016-04-25 | 2021-01-12 | Stryker Corporation | Anti-jamming and macerating thrombectomy apparatuses and methods |
| JP6924256B2 (ja) | 2016-04-25 | 2021-08-25 | ストライカー コーポレイションStryker Corporation | 予め組み込まれた反転トラクタの血栓除去装置および方法 |
| CN109414272B (zh) | 2016-04-25 | 2021-09-28 | 斯瑞克公司 | 翻转机械血栓切除装置及在血管系统中使用的方法 |
| US10940294B2 (en) | 2016-04-25 | 2021-03-09 | Medtronic Vascular, Inc. | Balloon catheter including a drug delivery sheath |
| US11147952B2 (en) | 2016-04-28 | 2021-10-19 | Medtronic Vascular, Inc. | Drug coated inflatable balloon having a thermal dependent release layer |
| US10191615B2 (en) | 2016-04-28 | 2019-01-29 | Medtronic Navigation, Inc. | Method and apparatus for image-based navigation |
| US10406011B2 (en) | 2016-04-28 | 2019-09-10 | Medtronic Vascular, Inc. | Implantable medical device delivery system |
| US10292844B2 (en) | 2016-05-17 | 2019-05-21 | Medtronic Vascular, Inc. | Method for compressing a stented prosthesis |
| WO2017210092A1 (en) | 2016-06-01 | 2017-12-07 | Microvention, Inc. | Improved reinforced balloon catheter |
| US10285710B2 (en) | 2016-06-01 | 2019-05-14 | DePuy Synthes Products, Inc. | Endovascular detachment system with flexible distal end and heater activated detachment |
| US10517624B2 (en) | 2016-06-03 | 2019-12-31 | Stryker Corporation | Inverting thrombectomy apparatuses and methods |
| US10646689B2 (en) | 2016-07-29 | 2020-05-12 | Cephea Valve Technologies, Inc. | Mechanical interlock for catheters |
| KR20180047232A (ko) | 2016-10-31 | 2018-05-10 | 삼성에스디에스 주식회사 | 실시간 영상 스트리밍의 네트워크 패킷의 패턴 분석 기반 영상 분석 방법 및 그 장치 |
| US10258492B2 (en) | 2017-03-03 | 2019-04-16 | Cook Medical Technologies Llc | Prosthesis delivery system with axially collapsible sheath |
| US11305095B2 (en) | 2018-02-22 | 2022-04-19 | Scientia Vascular, Llc | Microfabricated catheter having an intermediate preferred bending section |
| US10806461B2 (en) | 2018-04-27 | 2020-10-20 | DePuy Synthes Products, Inc. | Implantable medical device detachment system with split tube |
| US10631791B2 (en) | 2018-06-25 | 2020-04-28 | Caption Health, Inc. | Video clip selector for medical imaging and diagnosis |
| US11051928B2 (en) | 2019-04-11 | 2021-07-06 | Neuravi Limited | Floating carotid filter |
| US10653425B1 (en) | 2019-05-21 | 2020-05-19 | DePuy Synthes Products, Inc. | Layered braided aneurysm treatment device |
| US11207494B2 (en) | 2019-07-03 | 2021-12-28 | DePuy Synthes Products, Inc. | Medical device delivery member with flexible stretch resistant distal portion |
-
2019
- 2019-09-17 US US16/573,469 patent/US11439403B2/en active Active
-
2020
- 2020-09-11 KR KR1020200116735A patent/KR20210032908A/ko not_active Ceased
- 2020-09-16 JP JP2020155306A patent/JP7508317B2/ja active Active
- 2020-09-16 ES ES20196478T patent/ES2999631T3/es active Active
- 2020-09-16 EP EP20196478.0A patent/EP3795097B1/en active Active
- 2020-09-17 CN CN202010979459.6A patent/CN112515726B/zh active Active
-
2022
- 2022-08-16 US US17/888,677 patent/US12127744B2/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012000464A (ja) | 2010-06-16 | 2012-01-05 | Codman & Shurtleff Inc | 伸長抵抗性部材及びアンカーフィラメントを備えた閉塞デバイス |
| JP2013212372A (ja) | 2012-03-30 | 2013-10-17 | Depuy Synthes Products Llc | 可撓性の遠位部材と結合継手とを有する塞栓用コイル取り外し機構 |
| JP2019111339A (ja) | 2017-12-21 | 2019-07-11 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 分割チューブと筒状継手とを備えた移植可能医療装置取り外しシステム |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2021058586A (ja) * | 2019-10-03 | 2021-04-15 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 可撓性の耐伸張性機械的解放を有する医療装置送達部材 |
| JP7593549B2 (ja) | 2019-10-03 | 2024-12-03 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 可撓性の耐伸張性機械的解放を有する医療装置送達部材 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3795097B1 (en) | 2024-10-30 |
| JP2021045547A (ja) | 2021-03-25 |
| ES2999631T3 (en) | 2025-02-26 |
| US12127744B2 (en) | 2024-10-29 |
| US20220387045A1 (en) | 2022-12-08 |
| CN112515726A (zh) | 2021-03-19 |
| KR20210032908A (ko) | 2021-03-25 |
| EP3795097A1 (en) | 2021-03-24 |
| CN112515726B (zh) | 2025-09-05 |
| US20210077118A1 (en) | 2021-03-18 |
| EP3795097C0 (en) | 2024-10-30 |
| US11439403B2 (en) | 2022-09-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7508317B2 (ja) | 塞栓コイル近位接続要素及び耐延伸性繊維 | |
| US12376859B2 (en) | Embolic coil proximal connecting element and stretch resistant fiber | |
| JP7680112B2 (ja) | 可撓性の耐伸張性遠位部分を有する医療装置送達部材 | |
| EP4119065B1 (en) | Implant with modified detachment feature and pull wire engagement | |
| US9034026B2 (en) | Implant for influencing blood flow | |
| EP3181064B1 (en) | Occlusive device delivery system with mechanical detachment | |
| EP2854704B1 (en) | Aneurysm occlusion system | |
| US20170172581A1 (en) | Aneursym occlusion system and method | |
| EP2397081B1 (en) | Occlusive device with stretch resistant member and anchor filament | |
| KR20210103957A (ko) | 혈관내 임플란트 전개 시스템 | |
| US20240138843A1 (en) | Implant detachment systems with a modified pull wire | |
| EP4119064B1 (en) | Embolic coil proximal connecting element and stretch resistant fiber | |
| JP7852194B2 (ja) | 塞栓コイル近位接続要素及び耐延伸性繊維 | |
| WO2024125147A1 (zh) | 血管内介入治疗装置 | |
| JP2025507638A (ja) | 神経血管用フローダイバータおよび送達システム | |
| EP4205671B1 (en) | Medical device delivery systems with twisting loop wires | |
| EP4398814B1 (en) | Vaso-occlusive device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20230726 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20240220 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20240222 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20240517 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20240604 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20240619 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7508317 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |