JP7163733B2 - 液晶組成物及び液晶表示素子 - Google Patents
液晶組成物及び液晶表示素子 Download PDFInfo
- Publication number
- JP7163733B2 JP7163733B2 JP2018213765A JP2018213765A JP7163733B2 JP 7163733 B2 JP7163733 B2 JP 7163733B2 JP 2018213765 A JP2018213765 A JP 2018213765A JP 2018213765 A JP2018213765 A JP 2018213765A JP 7163733 B2 JP7163733 B2 JP 7163733B2
- Authority
- JP
- Japan
- Prior art keywords
- liquid crystal
- general formula
- group
- mass
- carbon atoms
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000004973 liquid crystal related substance Substances 0.000 title claims description 151
- 239000000203 mixture Substances 0.000 title claims description 120
- 150000001875 compounds Chemical class 0.000 claims description 206
- 125000004432 carbon atom Chemical group C* 0.000 claims description 63
- 239000000758 substrate Substances 0.000 claims description 32
- 125000000217 alkyl group Chemical group 0.000 claims description 24
- 125000003545 alkoxy group Chemical group 0.000 claims description 23
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 14
- 125000001153 fluoro group Chemical group F* 0.000 claims description 12
- 229910052731 fluorine Inorganic materials 0.000 claims description 11
- 125000003342 alkenyl group Chemical group 0.000 claims description 10
- 239000003963 antioxidant agent Substances 0.000 claims description 10
- RGOVYLWUIBMPGK-UHFFFAOYSA-N nonivamide Chemical compound CCCCCCCCC(=O)NCC1=CC=C(O)C(OC)=C1 RGOVYLWUIBMPGK-UHFFFAOYSA-N 0.000 claims description 10
- 125000002947 alkylene group Chemical group 0.000 claims description 8
- 230000003078 antioxidant effect Effects 0.000 claims description 8
- 125000003302 alkenyloxy group Chemical group 0.000 claims description 2
- 239000011159 matrix material Substances 0.000 claims description 2
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 claims 1
- 238000000034 method Methods 0.000 description 12
- 239000010408 film Substances 0.000 description 11
- 230000000052 comparative effect Effects 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 238000006116 polymerization reaction Methods 0.000 description 6
- 230000000379 polymerizing effect Effects 0.000 description 5
- 230000005684 electric field Effects 0.000 description 4
- 238000009281 ultraviolet germicidal irradiation Methods 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 210000004027 cell Anatomy 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 230000006866 deterioration Effects 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- 230000000704 physical effect Effects 0.000 description 3
- 125000001140 1,4-phenylene group Chemical group [H]C1=C([H])C([*:2])=C([H])C([H])=C1[*:1] 0.000 description 2
- -1 2-methylbenzene-1,4-diyl groups Chemical group 0.000 description 2
- 239000006096 absorbing agent Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 230000007547 defect Effects 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical group C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 239000007791 liquid phase Substances 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 2
- 229910052753 mercury Inorganic materials 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 238000007789 sealing Methods 0.000 description 2
- 125000006850 spacer group Chemical group 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 239000010409 thin film Substances 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 1
- 239000004986 Cholesteric liquid crystals (ChLC) Substances 0.000 description 1
- 239000004593 Epoxy Substances 0.000 description 1
- 239000004988 Nematic liquid crystal Substances 0.000 description 1
- 239000004990 Smectic liquid crystal Substances 0.000 description 1
- 206010047571 Visual impairment Diseases 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 210000002858 crystal cell Anatomy 0.000 description 1
- 125000005725 cyclohexenylene group Chemical group 0.000 description 1
- 238000004043 dyeing Methods 0.000 description 1
- 238000004070 electrodeposition Methods 0.000 description 1
- 238000010894 electron beam technology Methods 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 1
- 239000012212 insulator Substances 0.000 description 1
- 230000001678 irradiating effect Effects 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 150000004965 peroxy acids Chemical class 0.000 description 1
- 229920002120 photoresistant polymer Polymers 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 230000002269 spontaneous effect Effects 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 125000005407 trans-1,4-cyclohexylene group Chemical group [H]C1([H])C([H])([H])[C@]([H])([*:2])C([H])([H])C([H])([H])[C@@]1([H])[*:1] 0.000 description 1
- 239000012780 transparent material Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/38—Polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/42—Mixtures of liquid crystal compounds covered by two or more of the preceding groups C09K19/06 - C09K19/40
- C09K19/46—Mixtures of liquid crystal compounds covered by two or more of the preceding groups C09K19/06 - C09K19/40 containing esters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F20/00—Homopolymers and copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride, ester, amide, imide or nitrile thereof
- C08F20/02—Monocarboxylic acids having less than ten carbon atoms, Derivatives thereof
- C08F20/10—Esters
- C08F20/22—Esters containing halogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F20/00—Homopolymers and copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride, ester, amide, imide or nitrile thereof
- C08F20/02—Monocarboxylic acids having less than ten carbon atoms, Derivatives thereof
- C08F20/10—Esters
- C08F20/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F20/30—Esters containing oxygen in addition to the carboxy oxygen containing aromatic rings in the alcohol moiety
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/0008—Organic ingredients according to more than one of the "one dot" groups of C08K5/01 - C08K5/59
- C08K5/005—Stabilisers against oxidation, heat, light, ozone
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/13—Phenols; Phenolates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/15—Heterocyclic compounds having oxygen in the ring
- C08K5/156—Heterocyclic compounds having oxygen in the ring having two oxygen atoms in the ring
- C08K5/1575—Six-membered rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/54—Additives having no specific mesophase characterised by their chemical composition
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/137—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells characterised by the electro-optical or magneto-optical effect, e.g. field-induced phase transition, orientation effect, guest-host interaction or dynamic scattering
Description
第二成分として、一般式(RM-22)
更に詳述すると、P3及びP4は、それぞれ独立して式(PG-1)又は式(PG-2)であることが好ましく、式(PG-1)であることが特に好ましい。
R22は、炭素原子数1~8のアルキル基又は炭素原子数1から8のアルコキシ基であることが好ましく、炭素原子数1~5のアルキル基又は炭素原子数1から4のアルコキシ基がより好ましく、炭素原子数1~4のアルコキシ基が更に好ましい。
R21及びR22がアルケニル基である場合は、式(R1)から式(R5)
なお、本願において実質的に含有しないとは、意図せずに含有する物を除いて含有しないという意味である。
本発明の液晶組成物は、第一成分の重合性化合物及び第二成分の重合性化合物を含有し、更に一般式(N-01)、一般式(N-02)、一般式(N-03)、一般式(N-04)及び一般式(N-05)で表される化合物群から選ばれる化合物を1種類又は2種類以上含有し、更に一般式(NU-01)から(NU-08)で表される化合物群から選ばれる化合物を1種又は2種以上含有することが好ましく、これらの含有量の合計の上限値は、100質量%、99質量%、98質量%、97質量%、96質量%、95質量%、94質量%、93質量%、92質量%、91質量%、90質量%、89質量%、88質量%、87質量%、86質量%、85質量%、84質量%であることが好ましく、これらの含有量の合計の下限値が、78質量%、80質量%、81質量%、83質量%、85質量%、86質量%、87質量%、88質量%、89質量%、90質量%、91質量%、92質量%、93質量%、94質量%、95質量%、96質量%、97質量%、98質量%、99質量%、100質量%であることが好ましい。
-n -CnH2n+1 炭素数nの直鎖状のアルキル基
n- CnH2n+1- 炭素数nの直鎖状のアルキル基
-On -OCnH2n+1 炭素数nの直鎖状のアルコキシ基
nO- CnH2n+1O- 炭素数nの直鎖状のアルコキシ基
-V -CH=CH2
V- CH2=CH-
-V1 -CH=CH-CH3
1V- CH3-CH=CH-
-F -F
-OCF3 -OCF3
(連結基)
-1O- -CH2-O-
-O1- -O-CH2-
-2- -CH2-CH2-
-COO- -COO-
-OCO- -OCO-
- 単結合
(環構造)
Δn :20℃における屈折率異方性
Δε :20℃における誘電率異方性
γ1 :20℃における回転粘性(mPa・s)
RM :313nmの照度3mW/cm2のUV光を20分照射した後の液晶表示素子の中の残存モノマー量(ppm)
Tilt :313nmの照度3mW/cm2のUV光を2分照射した後の液晶表示素子のプレチルト角(°)であり、90°からの変化量を表す。
VHR :313nmの照度3mW/cm2のUV光を60分照射した後の液晶表示素子を用意し、1V、60Hz、60℃で測定したときの電圧保持率(%)
(液晶組成物の調製と評価結果)
液晶組成物(LC-1)から(LC-4)を調製し、その物性値を測定した。これらの液晶組成物の成分比とその物性値は表1のとおりであった。
Claims (11)
- 第一成分として、一般式(RM-13)
P3及びP4は、それぞれ独立して、式(PG-1)から式(PG-5)
S3は、単結合又は炭素原子数1~5のアルキレン基を表し、
Y1からY12はそれぞれ独立してフッ素原子又は水素原子を表すが、少なくとも一つはフッ素原子を表す。)
で表される重合性化合物を含有し、
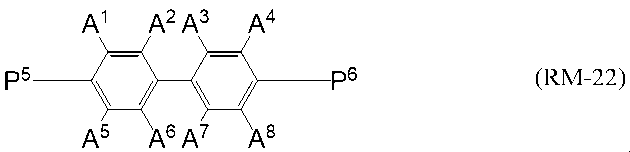
第二成分として、一般式(RM-22)
P5及びP6は、それぞれ独立して、式(PG-1)から式(PG-5)
A1からA8は、それぞれ独立して水素原子又は炭素原子数1~5のアルコキシ基を表すが、
A 1 からA 8 のうち1つ又は2つが炭素原子数1~3のアルコキシ基を表し、
残りが全て水素原子を表す。)
で表される重合性化合物を含有する液晶組成物。 - 一般式(N-01)、一般式(N-02)、一般式(N-03)、一般式(N-04)及び一般式(N-05)
R21及びR22は、それぞれ独立して、炭素原子数1~8のアルキル基、炭素原子数1~8のアルコキシ基、炭素原子数2~8のアルケニル基又は炭素原子数2~8のアルケニルオキシ基を表し、
該基中の1個又は非隣接の2個以上の-CH2-はそれぞれ独立して-CH=CH-、-C≡C-、-O-、-CO-、-COO-又は-OCO-によって置換されていても良く、
Z1は、それぞれ独立して、単結合、-CH2CH2-、-OCH2-、-CH2O-、-COO-、-OCO-、-OCF2-、-CF2O-、-CH=CH-、-CF=CF-又は-C≡C-を表し、
mは、それぞれ独立して、1又は2を表す。)
で表される化合物群から選ばれる化合物を1種又は2種以上含有する請求項1に記載の液晶組成物。 - 一般式(NU-01)から一般式(NU-08)
RNU11、RNU12、RNU21、RNU22、RNU31、RNU32、RNU41、RNU42、RNU51、RNU52、RNU61、RNU62、RNU71、RNU72、RNU81及びRNU82は、それぞれ独立して、炭素原子数1~8のアルキル基、炭素原子数1~8のアルコキシ基、炭素原子数2~8のアルケニル基又は炭素原子数2~8のアルケニルオキシ基を表し、
該基中の1個又は非隣接の2個以上の-CH2-はそれぞれ独立して-CH=CH-、-C≡C-、-O-、-CO-、-COO-又は-OCO-によって置換されていても良い。)
で表される化合物群から選ばれる化合物を1種又は2種以上含有する請求項1又は2に記載の液晶組成物。 - 前記第一成分、前記第二成分、前記一般式(N-01)~前記一般式(N-04)で表される化合物群から選ばれる化合物、及び前記一般式(NU-01)~前記一般式(NU-08)で表される化合物群から選ばれる化合物の含有量の合計が、組成物の総量に対して80質量%から100質量%である請求項1~5のいずれか一項に記載の液晶組成物。
- 二つの基板と、該二つの基板の間に設けられた請求項1~7のいずれか一項に記載の液晶組成物を含む液晶相と、を備える液晶表示素子。
- アクティブマトリックス駆動用である請求項8に記載の液晶表示素子。
- PSA型又はPSVA型である請求項8又は9に記載の液晶表示素子。
- 前記二つの基板のうち少なくとも一方の基板が配向膜を有さない請求項8~10のいずれか一項に記載の液晶表示素子。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018213765A JP7163733B2 (ja) | 2018-11-14 | 2018-11-14 | 液晶組成物及び液晶表示素子 |
| CN201910911103.6A CN111187629A (zh) | 2018-11-14 | 2019-09-25 | 液晶组合物和液晶显示元件 |
| KR1020190137313A KR20200056299A (ko) | 2018-11-14 | 2019-10-31 | 액정 조성물 및 액정 표시 소자 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018213765A JP7163733B2 (ja) | 2018-11-14 | 2018-11-14 | 液晶組成物及び液晶表示素子 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2020079371A JP2020079371A (ja) | 2020-05-28 |
| JP7163733B2 true JP7163733B2 (ja) | 2022-11-01 |
Family
ID=70704584
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018213765A Active JP7163733B2 (ja) | 2018-11-14 | 2018-11-14 | 液晶組成物及び液晶表示素子 |
Country Status (3)
| Country | Link |
|---|---|
| JP (1) | JP7163733B2 (ja) |
| KR (1) | KR20200056299A (ja) |
| CN (1) | CN111187629A (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115651669A (zh) * | 2022-11-14 | 2023-01-31 | 北京八亿时空液晶科技股份有限公司 | 一种负介电各向异性的液晶组合物及其应用 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013538250A (ja) | 2010-07-21 | 2013-10-10 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | 重合性混合物および液晶ディスプレイにおけるそれらの使用 |
| JP2018509507A (ja) | 2015-03-13 | 2018-04-05 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | 液晶媒体 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5566756A (en) | 1978-11-14 | 1980-05-20 | Toyo Soda Mfg Co Ltd | Carrier for ion exchange liquid chromatography and its manufacturing method |
| JPS608065B2 (ja) | 1979-04-28 | 1985-02-28 | シャープ株式会社 | ホツトメルト系有機−無機混合接着剤 |
| JP5593890B2 (ja) * | 2010-07-06 | 2014-09-24 | Dic株式会社 | 液晶組成物および液晶デバイス |
| KR102088166B1 (ko) | 2011-03-29 | 2020-03-12 | 메르크 파텐트 게엠베하 | 액정 매질 |
| CN105733607B (zh) * | 2013-03-26 | 2018-10-19 | Dic株式会社 | 液晶组合物以及使用其的液晶显示元件和液晶显示器 |
| CN104342167B (zh) | 2013-08-02 | 2016-12-28 | 江苏和成显示科技股份有限公司 | 聚合性液晶组合物及其显示器件 |
| CN104342170B (zh) | 2013-08-02 | 2016-12-28 | 江苏和成显示科技股份有限公司 | 聚合性液晶组合物及其显示器件 |
| DE112014006109B4 (de) * | 2014-01-06 | 2018-09-20 | Dic Corporation | Nematische Flüssigkristallzusammensetzung und ihre Verwendung zur Herstellung eines Flüssigkristallanzeigeelements |
| CN107075375B (zh) * | 2014-12-25 | 2020-11-13 | Dic株式会社 | 向列液晶组合物及使用其的液晶显示元件 |
| EP3121247B1 (en) | 2015-06-09 | 2019-10-02 | Merck Patent GmbH | Polymerisable compounds and the use thereof in liquid-crystal displays |
| EP3450523B1 (en) * | 2016-04-27 | 2021-01-06 | DIC Corporation | Liquid crystal composition and liquid crystal display device |
| KR20180134851A (ko) * | 2016-04-27 | 2018-12-19 | 디아이씨 가부시끼가이샤 | 네마틱 액정 조성물 및 이를 사용한 액정 표시 소자 |
| KR101988816B1 (ko) * | 2016-11-22 | 2019-06-12 | 디아이씨 가부시끼가이샤 | 액정 조성물 및 액정 표시 소자 |
| JP6380825B1 (ja) | 2016-12-26 | 2018-08-29 | Dic株式会社 | 液晶組成物及び液晶表示素子 |
-
2018
- 2018-11-14 JP JP2018213765A patent/JP7163733B2/ja active Active
-
2019
- 2019-09-25 CN CN201910911103.6A patent/CN111187629A/zh active Pending
- 2019-10-31 KR KR1020190137313A patent/KR20200056299A/ko unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013538250A (ja) | 2010-07-21 | 2013-10-10 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | 重合性混合物および液晶ディスプレイにおけるそれらの使用 |
| JP2018509507A (ja) | 2015-03-13 | 2018-04-05 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | 液晶媒体 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20200056299A (ko) | 2020-05-22 |
| CN111187629A (zh) | 2020-05-22 |
| JP2020079371A (ja) | 2020-05-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8906472B2 (en) | Nematic liquid crystal composition and liquid crystal display element using the same | |
| TWI567178B (zh) | A nematic liquid crystal composition, and a liquid crystal display device using the same | |
| JP7139797B2 (ja) | 液晶組成物及び液晶表示素子 | |
| CN111234844B (zh) | 液晶组合物和液晶显示元件 | |
| CN106062134B (zh) | 向列液晶组合物及使用其的液晶显示元件 | |
| JP7024254B2 (ja) | 液晶組成物及び液晶表示素子 | |
| TWI773789B (zh) | 液晶組成物及液晶顯示元件 | |
| TW201623585A (zh) | 向列型液晶組成物及使用其之液晶顯示元件 | |
| CN111373017B (zh) | 液晶组合物和液晶显示元件 | |
| JPWO2019021838A1 (ja) | 液晶組成物及び液晶表示素子 | |
| JP7163733B2 (ja) | 液晶組成物及び液晶表示素子 | |
| TWI786218B (zh) | 液晶組成物及液晶顯示元件 | |
| US9411207B2 (en) | Nematic liquid crystal composition and liquid crystal display element using the same | |
| JP7290061B2 (ja) | 液晶組成物及び液晶表示素子 | |
| CN112080290A (zh) | 含聚合性化合物的液晶组合物和液晶显示元件 | |
| JP7392434B2 (ja) | 液晶組成物及び液晶表示素子 | |
| JP7318204B2 (ja) | 液晶組成物及び液晶表示素子 | |
| JP2020097677A (ja) | 液晶組成物及び液晶表示素子 | |
| JP7395940B2 (ja) | 液晶組成物及び液晶表示素子 | |
| CN113493690A (zh) | 化合物、液晶组合物和液晶显示元件 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20190624 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20210916 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220830 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220906 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20220920 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20221003 |
|
| R151 | Written notification of patent or utility model registration |
Ref document number: 7163733 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R151 |
|
| R154 | Certificate of patent or utility model (reissue) |
Free format text: JAPANESE INTERMEDIATE CODE: R154 |