JP5198577B2 - 配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその形成方法 - Google Patents
配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその形成方法 Download PDFInfo
- Publication number
- JP5198577B2 JP5198577B2 JP2010534653A JP2010534653A JP5198577B2 JP 5198577 B2 JP5198577 B2 JP 5198577B2 JP 2010534653 A JP2010534653 A JP 2010534653A JP 2010534653 A JP2010534653 A JP 2010534653A JP 5198577 B2 JP5198577 B2 JP 5198577B2
- Authority
- JP
- Japan
- Prior art keywords
- alignment film
- alignment
- polyimide
- liquid crystal
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000004973 liquid crystal related substance Substances 0.000 title claims description 271
- 239000000463 material Substances 0.000 title claims description 166
- 238000000034 method Methods 0.000 title claims description 103
- 239000004642 Polyimide Substances 0.000 claims description 254
- 229920001721 polyimide Polymers 0.000 claims description 254
- 229920002554 vinyl polymer Polymers 0.000 claims description 174
- 239000000178 monomer Substances 0.000 claims description 166
- 239000000758 substrate Substances 0.000 claims description 148
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 97
- 150000001875 compounds Chemical class 0.000 claims description 83
- 239000002243 precursor Substances 0.000 claims description 82
- 125000001153 fluoro group Chemical group F* 0.000 claims description 75
- 238000010438 heat treatment Methods 0.000 claims description 65
- 239000011159 matrix material Substances 0.000 claims description 58
- -1 naphthalene-2,6-diyl Chemical group 0.000 claims description 27
- 125000000217 alkyl group Chemical group 0.000 claims description 21
- 229910052731 fluorine Inorganic materials 0.000 claims description 19
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 18
- 239000011737 fluorine Substances 0.000 claims description 15
- 125000000524 functional group Chemical group 0.000 claims description 14
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 claims description 13
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 claims description 13
- 238000007639 printing Methods 0.000 claims description 13
- 238000004528 spin coating Methods 0.000 claims description 13
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims description 12
- 229910052736 halogen Inorganic materials 0.000 claims description 12
- 150000002367 halogens Chemical class 0.000 claims description 12
- FQPSGWSUVKBHSU-UHFFFAOYSA-N methacrylamide Chemical compound CC(=C)C(N)=O FQPSGWSUVKBHSU-UHFFFAOYSA-N 0.000 claims description 12
- 125000001140 1,4-phenylene group Chemical group [H]C1=C([H])C([*:2])=C([H])C([H])=C1[*:1] 0.000 claims description 11
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 11
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 11
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 9
- 125000004432 carbon atom Chemical group C* 0.000 claims description 9
- 229910052801 chlorine Inorganic materials 0.000 claims description 9
- 239000000460 chlorine Substances 0.000 claims description 9
- 125000004122 cyclic group Chemical group 0.000 claims description 9
- WBYWAXJHAXSJNI-VOTSOKGWSA-M trans-cinnamate Chemical group [O-]C(=O)\C=C\C1=CC=CC=C1 WBYWAXJHAXSJNI-VOTSOKGWSA-M 0.000 claims description 9
- 125000006850 spacer group Chemical group 0.000 claims description 8
- 125000003118 aryl group Chemical group 0.000 claims description 4
- 125000005731 2,5-thiophenylene group Chemical group [H]C1=C([*:1])SC([*:2])=C1[H] 0.000 claims description 3
- 125000004959 2,6-naphthylene group Chemical group [H]C1=C([H])C2=C([H])C([*:1])=C([H])C([H])=C2C([H])=C1[*:2] 0.000 claims description 3
- 229910003849 O-Si Inorganic materials 0.000 claims description 3
- 229910003872 O—Si Inorganic materials 0.000 claims description 3
- 125000002723 alicyclic group Chemical group 0.000 claims description 3
- DMLAVOWQYNRWNQ-UHFFFAOYSA-N azobenzene Chemical group C1=CC=CC=C1N=NC1=CC=CC=C1 DMLAVOWQYNRWNQ-UHFFFAOYSA-N 0.000 claims description 3
- 230000001588 bifunctional effect Effects 0.000 claims description 3
- JRXXLCKWQFKACW-UHFFFAOYSA-N biphenylacetylene Chemical group C1=CC=CC=C1C#CC1=CC=CC=C1 JRXXLCKWQFKACW-UHFFFAOYSA-N 0.000 claims description 3
- 125000000332 coumarinyl group Chemical group O1C(=O)C(=CC2=CC=CC=C12)* 0.000 claims description 3
- 125000005843 halogen group Chemical group 0.000 claims description 3
- 229910052739 hydrogen Inorganic materials 0.000 claims description 3
- 239000001257 hydrogen Substances 0.000 claims description 3
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 3
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 claims description 3
- 230000000379 polymerizing effect Effects 0.000 claims description 3
- DQFBYFPFKXHELB-VAWYXSNFSA-N trans-chalcone Chemical group C=1C=CC=CC=1C(=O)\C=C\C1=CC=CC=C1 DQFBYFPFKXHELB-VAWYXSNFSA-N 0.000 claims description 3
- 150000001252 acrylic acid derivatives Chemical class 0.000 claims 1
- 239000010408 film Substances 0.000 description 418
- 239000010410 layer Substances 0.000 description 146
- 230000008569 process Effects 0.000 description 49
- 238000011282 treatment Methods 0.000 description 36
- 239000002904 solvent Substances 0.000 description 35
- 230000008859 change Effects 0.000 description 32
- 229920005575 poly(amic acid) Polymers 0.000 description 24
- 229920000642 polymer Polymers 0.000 description 19
- 230000005611 electricity Effects 0.000 description 17
- 238000006116 polymerization reaction Methods 0.000 description 16
- 238000012360 testing method Methods 0.000 description 16
- 230000001678 irradiating effect Effects 0.000 description 15
- 238000012545 processing Methods 0.000 description 13
- 230000007423 decrease Effects 0.000 description 12
- 238000010586 diagram Methods 0.000 description 12
- 239000004988 Nematic liquid crystal Substances 0.000 description 10
- 238000006471 dimerization reaction Methods 0.000 description 10
- 230000001105 regulatory effect Effects 0.000 description 10
- VZSRBBMJRBPUNF-UHFFFAOYSA-N 2-(2,3-dihydro-1H-inden-2-ylamino)-N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]pyrimidine-5-carboxamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C(=O)NCCC(N1CC2=C(CC1)NN=N2)=O VZSRBBMJRBPUNF-UHFFFAOYSA-N 0.000 description 9
- 238000006358 imidation reaction Methods 0.000 description 9
- 238000010304 firing Methods 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- 230000003287 optical effect Effects 0.000 description 7
- 238000004833 X-ray photoelectron spectroscopy Methods 0.000 description 6
- 238000010884 ion-beam technique Methods 0.000 description 6
- 239000012535 impurity Substances 0.000 description 5
- 150000002500 ions Chemical class 0.000 description 5
- 125000004429 atom Chemical group 0.000 description 4
- 125000004386 diacrylate group Chemical group 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 239000011521 glass Substances 0.000 description 4
- 230000004044 response Effects 0.000 description 4
- MKYBYDHXWVHEJW-UHFFFAOYSA-N N-[1-oxo-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propan-2-yl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(C(C)NC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 MKYBYDHXWVHEJW-UHFFFAOYSA-N 0.000 description 3
- NIPNSKYNPDTRPC-UHFFFAOYSA-N N-[2-oxo-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 NIPNSKYNPDTRPC-UHFFFAOYSA-N 0.000 description 3
- AFCARXCZXQIEQB-UHFFFAOYSA-N N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CCNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 AFCARXCZXQIEQB-UHFFFAOYSA-N 0.000 description 3
- 238000009825 accumulation Methods 0.000 description 3
- 230000005684 electric field Effects 0.000 description 3
- 230000010287 polarization Effects 0.000 description 3
- 238000005011 time of flight secondary ion mass spectroscopy Methods 0.000 description 3
- 238000002042 time-of-flight secondary ion mass spectrometry Methods 0.000 description 3
- VCNIZVZNSLZWPZ-UHFFFAOYSA-N 1,1'-biphenyl;2-methylprop-2-enoic acid Chemical compound CC(=C)C(O)=O.CC(=C)C(O)=O.C1=CC=CC=C1C1=CC=CC=C1 VCNIZVZNSLZWPZ-UHFFFAOYSA-N 0.000 description 2
- UWCWUCKPEYNDNV-LBPRGKRZSA-N 2,6-dimethyl-n-[[(2s)-pyrrolidin-2-yl]methyl]aniline Chemical compound CC1=CC=CC(C)=C1NC[C@H]1NCCC1 UWCWUCKPEYNDNV-LBPRGKRZSA-N 0.000 description 2
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- 150000001491 aromatic compounds Chemical class 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 230000001629 suppression Effects 0.000 description 2
- 238000002834 transmittance Methods 0.000 description 2
- 0 CC(*)(*)C(CC1)(CC2)*C12N Chemical compound CC(*)(*)C(CC1)(CC2)*C12N 0.000 description 1
- XIAHSJTUQAWUAW-UHFFFAOYSA-N CC(C)(c1ccc(C)cc1)c1ccc(C)cc1 Chemical compound CC(C)(c1ccc(C)cc1)c1ccc(C)cc1 XIAHSJTUQAWUAW-UHFFFAOYSA-N 0.000 description 1
- PMPVIKIVABFJJI-UHFFFAOYSA-N Cyclobutane Chemical compound C1CCC1 PMPVIKIVABFJJI-UHFFFAOYSA-N 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000001941 electron spectroscopy Methods 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- 150000002221 fluorine Chemical group 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 125000005641 methacryl group Chemical group 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- CHDKQNHKDMEASZ-UHFFFAOYSA-N n-prop-2-enoylprop-2-enamide Chemical compound C=CC(=O)NC(=O)C=C CHDKQNHKDMEASZ-UHFFFAOYSA-N 0.000 description 1
- 239000005416 organic matter Substances 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08L79/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1039—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors comprising halogen-containing substituents
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1046—Polyimides containing oxygen in the form of ether bonds in the main chain
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1046—Polyimides containing oxygen in the form of ether bonds in the main chain
- C08G73/105—Polyimides containing oxygen in the form of ether bonds in the main chain with oxygen only in the diamino moiety
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1067—Wholly aromatic polyimides, i.e. having both tetracarboxylic and diamino moieties aromatically bound
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1075—Partially aromatic polyimides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1075—Partially aromatic polyimides
- C08G73/1078—Partially aromatic polyimides wholly aromatic in the diamino moiety
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/133—Constructional arrangements; Operation of liquid crystal cells; Circuit arrangements
- G02F1/1333—Constructional arrangements; Manufacturing methods
- G02F1/1335—Structural association of cells with optical devices, e.g. polarisers or reflectors
- G02F1/13363—Birefringent elements, e.g. for optical compensation
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/133—Constructional arrangements; Operation of liquid crystal cells; Circuit arrangements
- G02F1/1333—Constructional arrangements; Manufacturing methods
- G02F1/1337—Surface-induced orientation of the liquid crystal molecules, e.g. by alignment layers
- G02F1/133711—Surface-induced orientation of the liquid crystal molecules, e.g. by alignment layers by organic films, e.g. polymeric films
- G02F1/133723—Polyimide, polyamide-imide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F222/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a carboxyl radical and containing at least one other carboxyl radical in the molecule; Salts, anhydrides, esters, amides, imides, or nitriles thereof
- C08F222/10—Esters
- C08F222/1006—Esters of polyhydric alcohols or polyhydric phenols
- C08F222/102—Esters of polyhydric alcohols or polyhydric phenols of dialcohols, e.g. ethylene glycol di(meth)acrylate or 1,4-butanediol dimethacrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/133—Constructional arrangements; Operation of liquid crystal cells; Circuit arrangements
- G02F1/1333—Constructional arrangements; Manufacturing methods
- G02F1/1337—Surface-induced orientation of the liquid crystal molecules, e.g. by alignment layers
- G02F1/133742—Surface-induced orientation of the liquid crystal molecules, e.g. by alignment layers for homeotropic alignment
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/137—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells characterised by the electro-optical or magneto-optical effect, e.g. field-induced phase transition, orientation effect, guest-host interaction or dynamic scattering
- G02F1/139—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells characterised by the electro-optical or magneto-optical effect, e.g. field-induced phase transition, orientation effect, guest-host interaction or dynamic scattering based on orientation effects in which the liquid crystal remains transparent
- G02F1/1393—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells characterised by the electro-optical or magneto-optical effect, e.g. field-induced phase transition, orientation effect, guest-host interaction or dynamic scattering based on orientation effects in which the liquid crystal remains transparent the birefringence of the liquid crystal being electrically controlled, e.g. ECB-, DAP-, HAN-, PI-LC cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31721—Of polyimide
Description
P1−A1−(Z1−A2)n−P2 (1)
P1−A1−(Z1−A2)n−P2 (1)
(実施例1−1)
以下、図2および図6を参照して、実施例1−1の配向膜および液晶表示装置を説明する。実施例1−1の液晶表示装置もRTNモードで動作する。
以下、図2および図6を参照して、実施例1−2の配向膜および液晶表示装置を説明する。実施例1−2の液晶表示装置もRTNモードで動作する。
以下、図2および図6を参照して、実施例1−3の配向膜および液晶表示装置を説明する。実施例1−3の液晶表示装置もRTNモードで動作する。
以下、図2および図6を参照して、実施例1−4の配向膜および液晶表示装置を説明する。実施例1−4の液晶表示装置もRTNモードで動作する。
以下、参考例の配向膜および液晶表示装置を説明する。参考例の液晶表示装置もRTNモードで動作する。
以下、図2および図6を参照して、実施例2の配向膜および液晶表示装置を説明する。実施例2の液晶表示装置もRTNモードで動作する。
以下、図2および図6を参照して、実施例3の配向膜および液晶表示装置を説明する。実施例3の液晶表示装置もRTNモードで動作する。
以下、図2および図7を参照して、実施例4の配向膜および液晶表示装置を説明する。実施例4の液晶表示装置もRTNモードで動作する。
以下、図2および図8を参照して、実施例5の配向膜および液晶表示装置を説明する。実施例5の液晶表示装置もRTNモードで動作する。
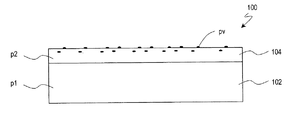
p1 第1ポリイミド
p2 第2ポリイミド
pv ポリビニル化合物
102 第1配向層
104 第2配向層
110 第1配向膜
112 第1配向層
114 第2配向層
120 第2配向膜
122 第1配向層
124 第2配向層
200 液晶表示装置
220 アクティブマトリクス基板
222 第1絶縁基板
224 画素電極
240 対向基板
242 第2絶縁基板
244 対向電極
260 液晶層
262 液晶分子
300 液晶パネル
Claims (25)
- 第1ポリイミドの前駆体と、
前記第1ポリイミドとは異なる第2ポリイミドおよびその前駆体の少なくとも一方と、
ビニル基を有するビニル系モノマーと
を含有する、配向膜材料であって、
前記ビニル系モノマーは、一般式(1)P1−A1−(Z1−A2)n−P2(一般式(1)において、P1およびP2は、独立に、アクリレート、メタクリレート、アクリルアミドまたはメタクリルアミドであり、A1およびA2は、独立に、1,4−フェニレン、1,4−シクロヘキサンまたは2,5−チオフェン、もしくは、ナフタレン−2,6−ジイルまたはアントラセン−2,7−ジイルを表し、A1およびA2の少なくとも一方は少なくとも1個のフッ素基で置換されており、Z1は−COO−、−OCO−、−O−、−CONH−基または単結合であり、nは0または1である)で表される、配向膜材料。 - 前記ビニル系モノマーは対称な構造を有する2官能モノマーである、請求項1に記載の配向膜材料。
- 前記ビニル系モノマーはジメタクリレートモノマーである、請求項1または2に記載の配向膜材料。
- 前記第1ポリイミドの前駆体の側鎖は垂直配向性基を有しない、請求項1から5のいずれかに記載の配向膜材料。
- 前記第2ポリイミドはフッ素基を含む側鎖を有する、請求項1から7のいずれかに記載の配向膜材料。
- 前記第2ポリイミドは光反応性官能基を有する、請求項1から8のいずれかに記載の配向膜材料。
- 前記光反応性官能基は、シンナメート基、カルコン基、トラン基、クマリン基およびアゾベンゼン基からなる群から選択された少なくとも一つである、請求項9に記載の配向膜材料。
- 前記第2ポリイミドおよびその前駆体の少なくとも一方は一般式(4)
Aは、場合によりフッ素、塩素、シアノから選択される基によるか、またはC 1-18 環式、直鎖状若しくは分岐鎖状のアルキル残基(これは、場合により1個のシアノ基または1個以上のハロゲン原子で置換されており、そして、場合により、アルキルの隣接しない1個以上の−CH 2 −基は、基Qで置き換えられている)で置換されている、ピリミジン−2,5−ジイル、ピリジン−2,5−ジイル、2,5−チオフェニレン、2,5−フラニレン、1,4−若しくは2,6−ナフチレンまたはフェニレンを表し、
Bは、非置換か、シアノ若しくはハロゲンで単置換されているか、またはハロゲンで多置換されている、炭素原子3〜18個を有する直鎖状または分岐鎖状のアルキル残基(ここで、隣接しない1個以上のCH 2 基は、独立して基Qで置き換えられていてもよい)であり、
C 1 およびC 2 は、互いに独立して、芳香族または脂環式基(これは、非置換か、あるいはフッ素、塩素、シアノまたは環式、直鎖状若しくは分岐鎖状のアルキル残基(これは、非置換か、シアノ若しくはハロゲンで単置換されているか、またはハロゲンで多置換されており、炭素原子1〜18個を有し、隣接しない1個以上のCH 2 基は、独立して基Qで置き換えられていてもよい)で置換されている)を表し、
Dは、酸素原子または−NR 1 −(ここで、R 1 は、水素原子または低級アルキルを表す)を表し、
S 1 およびS 2 は、互いに独立して、共有単結合またはスペーサ単位を表し、
S 3 は、スペーサ単位を表し、
Qは、−O−、−CO−、−CO−O−、−O−CO−、−Si(CH 3 ) 2 −O−Si(CH 3 ) 2 −、−NR 1 −、−NR 1 −CO−、−CO−NR 1 −、−NR 1 −CO−O−、−O−CO−NR 1 −、−NR 1 −CO−NR 1 −、−CH=CH−、−C≡C−および−O−CO−O−(ここで、R 1 は、水素原子または低級アルキルを表す)から選択される基を表し、
E、Fは、互いに独立して、水素、フッ素、塩素、シアノ、場合によりフッ素で置換され、炭素原子1〜12個を有するアルキル(ここで、場合により隣接しない1個以上のCH 2 基は、−O−、−CO−O−、−O−CO−および/または−CH=CH−で置き換えられている)を表し、
n 1 、n 2 は、正の整数である、請求項9または10に記載の配向膜材料。 - 前記第2ポリイミドおよびその前駆体の少なくとも一方は垂直配向性基を含む側鎖を有する、請求項1から8のいずれかに記載の配向膜材料。
- 前記配向膜材料に対する前記ビニル系モノマーの濃度は2wt%以上20wt%以下である、請求項1から12のいずれかに記載の配向膜材料。
- 第1ポリイミドと、
前記第1ポリイミドとは異なる第2ポリイミドと、
ビニル系モノマーの重合したポリビニル化合物と
を備える、配向膜であって、
前記ビニル系モノマーは、一般式(1)P1−A1−(Z1−A2)n−P2(一般式(1)において、P1およびP2は、独立に、アクリレート、メタクリレート、アクリルアミドまたはメタクリルアミドであり、A1およびA2は、独立に、1,4−フェニレン、1,4−シクロヘキサンまたは2,5−チオフェン、もしくは、ナフタレン−2,6−ジイルまたはアントラセン−2,7−ジイルを表し、A1およびA2の少なくとも一方は少なくとも1個のフッ素基で置換されており、Z1は−COO−、−OCO−、−O−、−CONH−基または単結合であり、nは0または1である)で表される、配向膜。 - 前記配向膜は、前記第1ポリイミドを含む第1配向層と、前記第2ポリイミドを含む第2配向層とを有している、請求項14に記載の配向膜。
- 前記ポリビニル化合物は、前記第2配向層内および前記第2配向層の表面に存在する、請求項15に記載の配向膜。
- 画素電極を有するアクティブマトリクス基板と、
対向電極を有する対向基板と、
前記アクティブマトリクス基板と前記対向基板との間に設けられた垂直配向型の液晶層と
を備える、液晶表示装置であって、
前記アクティブマトリクス基板および前記対向基板の少なくとも一方は前記液晶層側に設けられた配向膜をさらに有しており、
前記配向膜は、
第1ポリイミドと、
前記第1ポリイミドとは異なる第2ポリイミドと、
ビニル系モノマーの重合したポリビニル化合物と
を有しており、
前記ビニル系モノマーは、一般式(1)P1−A1−(Z1−A2)n−P2(一般式(1)において、P1およびP2は、独立に、アクリレート、メタクリレート、アクリルアミドまたはメタクリルアミドであり、A1およびA2は、独立に、1,4−フェニレン、1,4−シクロヘキサンまたは2,5−チオフェン、もしくは、ナフタレン−2,6−ジイルまたはアントラセン−2,7−ジイルを表し、A1およびA2の少なくとも一方は少なくとも1個のフッ素基で置換されており、Z1は−COO−、−OCO−、−O−、−CONH−基または単結合であり、nは0または1である)で表される、液晶表示装置。 - 前記配向膜は、前記第1ポリイミドを含む第1配向層と、前記第2ポリイミドを含む第2配向層とを有している、請求項17に記載の液晶表示装置。
- 前記第2配向層は前記第1配向層よりも前記液晶層側に設けられており、
前記ポリビニル化合物は前記第2配向層内および前記第2配向層の表面に存在する、請求項18に記載の液晶表示装置。 - 前記配向膜は、電圧無印加時に前記液晶層の液晶分子が前記配向膜の主面の法線方向から傾くように前記液晶分子を規定する、請求項17から19のいずれかに記載の液晶表示装置。
- 前記液晶表示装置は複数の画素を有しており、
前記液晶層は、前記複数の画素のそれぞれに対して、基準配向方位の互いに異なる複数の液晶ドメインを有している、請求項17から20のいずれかに記載の液晶表示装置。 - 前記複数の液晶ドメインは4つの液晶ドメインである、請求項21に記載の液晶表示装置。
- 第1ポリイミドの前駆体と、前記第1ポリイミドとは異なる第2ポリイミドおよびその前駆体の少なくとも一方と、ビニル系モノマーとを含有する配向膜材料を用意する工程と、
前記配向膜材料を塗布する工程と、
前記配向膜材料を加熱する工程であって、前記第1ポリイミドの前駆体の少なくとも一部を前記第1ポリイミドにイミド化し、前記ビニル系モノマーを重合してポリビニル化合物を形成する工程と
を包含し、
前記配向膜材料を用意する工程において、前記ビニル系モノマーは、一般式(1)P1−A1−(Z1−A2)n−P2(一般式(1)において、P1およびP2は、独立に、アクリレート、メタクリレート、アクリルアミドまたはメタクリルアミドであり、A1およびA2は、独立に、1,4−フェニレン、1,4−シクロヘキサンまたは2,5−チオフェン、もしくは、ナフタレン−2,6−ジイルまたはアントラセン−2,7−ジイルを表し、A1およびA2の少なくとも一方は少なくとも1個のフッ素基で置換されており、Z1は−COO−、−OCO−、−O−、−CONH−基または単結合であり、nは0または1である)で表される、配向膜の形成方法。 - 前記加熱する工程の後、前記第2ポリイミドのイミド化率は前記第1ポリイミドのイミド化率よりも大きい、請求項23に記載の配向膜の形成方法。
- 前記配向膜材料を塗布する工程は、前記配向膜材料の塗布を印刷法、インクジェット法またはスピンコート法で行う工程を含む、請求項23または24に記載の配向膜の形成方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010534653A JP5198577B2 (ja) | 2008-10-21 | 2009-06-23 | 配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその形成方法 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008271376 | 2008-10-21 | ||
| JP2008271376 | 2008-10-21 | ||
| JP2010534653A JP5198577B2 (ja) | 2008-10-21 | 2009-06-23 | 配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその形成方法 |
| PCT/JP2009/002862 WO2010047011A1 (ja) | 2008-10-21 | 2009-06-23 | 配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその形成方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2010047011A1 JPWO2010047011A1 (ja) | 2012-03-15 |
| JP5198577B2 true JP5198577B2 (ja) | 2013-05-15 |
Family
ID=42119069
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2010534653A Active JP5198577B2 (ja) | 2008-10-21 | 2009-06-23 | 配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその形成方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8345201B2 (ja) |
| EP (1) | EP2352060B1 (ja) |
| JP (1) | JP5198577B2 (ja) |
| CN (1) | CN102197333B (ja) |
| BR (1) | BRPI0919916A2 (ja) |
| WO (1) | WO2010047011A1 (ja) |
Families Citing this family (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8358391B2 (en) * | 2008-05-06 | 2013-01-22 | The Hong Kong University Of Science And Technology | Method to obtain a controlled pretilt and azimuthal angles in a liquid crystal cell |
| US20110164213A1 (en) * | 2008-09-03 | 2011-07-07 | Yohei Nakanishi | Alignment film, alignment film material, liquid crystal display device comprising alignment film, and method for manufacturing same |
| CN102216839B (zh) | 2008-09-17 | 2014-09-17 | 三星显示有限公司 | 定向材料、定向层、液晶显示装置及其制造方法 |
| US8514357B2 (en) * | 2008-09-17 | 2013-08-20 | Samsung Display Co., Ltd. | Alignment material, alignment layer, liquid crystal display device and manufacturing method thereof |
| RU2011126148A (ru) * | 2008-11-27 | 2013-01-10 | Шарп Кабусики Кайся | Жидкокристаллическое устройство отображения и способ его изготовления |
| US8597739B2 (en) | 2008-11-27 | 2013-12-03 | Sharp Kabushiki Kaisha | Orientation film, liquid crystal display having orientation film, and method for forming orientation film |
| WO2010131392A1 (ja) * | 2009-05-15 | 2010-11-18 | シャープ株式会社 | 液晶表示装置 |
| JP5884258B2 (ja) * | 2009-09-18 | 2016-03-15 | Jnc株式会社 | 液晶配向剤、液晶配向膜、液晶配向膜の製造方法および液晶表示素子 |
| WO2012017883A1 (ja) * | 2010-08-03 | 2012-02-09 | シャープ株式会社 | 液晶表示装置及び液晶表示装置の製造方法 |
| US9164325B2 (en) | 2010-08-03 | 2015-10-20 | Sharp Kabushiki Kaisha | Liquid crystal display device and process for producing liquid crystal display device |
| US9195097B2 (en) | 2010-08-03 | 2015-11-24 | Sharp Kabushiki Kaisha | Liquid crystal display device and process for producing liquid-crystal display device |
| CN103080824B (zh) | 2010-09-07 | 2016-03-09 | 夏普株式会社 | 液晶层形成用组合物、液晶显示装置和液晶显示装置的制造方法 |
| CN103154809B (zh) * | 2010-10-14 | 2016-06-29 | 夏普株式会社 | 液晶显示装置和液晶显示装置的制造方法 |
| TWI545372B (zh) | 2010-10-14 | 2016-08-11 | Merck Patent Gmbh | Liquid crystal display device |
| US9798179B2 (en) | 2010-10-14 | 2017-10-24 | Merck Patent Gmbh | Liquid crystal display device |
| KR101779510B1 (ko) * | 2010-11-11 | 2017-09-19 | 삼성디스플레이 주식회사 | 액정 표시 장치 및 그 제조 방법 |
| US9182632B2 (en) | 2010-12-06 | 2015-11-10 | Sharp Kabushiki Kaisha | Liquid crystal display device and method for manufacturing liquid crystal display device |
| US9239493B2 (en) | 2010-12-22 | 2016-01-19 | Sharp Kabushiki Kaisha | Liquid crystal alignment agent, liquid crystal display, and method for manufacturing liquid crystal display |
| KR101806351B1 (ko) * | 2011-02-01 | 2018-01-11 | 삼성디스플레이 주식회사 | 액정 표시 장치 |
| KR101894547B1 (ko) * | 2011-09-09 | 2018-09-04 | 삼성디스플레이 주식회사 | 액정 표시 장치 |
| CN104126148B (zh) * | 2011-12-21 | 2017-05-31 | 日产化学工业株式会社 | 液晶取向处理剂、液晶取向膜和使用该液晶取向膜的液晶显示元件以及化合物 |
| KR101986398B1 (ko) * | 2011-12-28 | 2019-06-05 | 닛산 가가쿠 가부시키가이샤 | 액정 배향제, 액정 배향막, 액정 표시 소자 및 액정 표시 소자의 제조 방법 |
| KR101998907B1 (ko) * | 2011-12-28 | 2019-07-10 | 닛산 가가쿠 가부시키가이샤 | 액정 배향제, 액정 배향막, 액정 표시 소자 및 디아민 화합물 |
| US9733524B2 (en) | 2012-01-30 | 2017-08-15 | Sharp Kabushiki Kaisha | Liquid crystal display device and manufacturing method therefor |
| CN102732264B (zh) * | 2012-06-04 | 2013-12-25 | 深圳市华星光电技术有限公司 | 一种液晶活性单体及液晶面板 |
| CN102732265B (zh) * | 2012-06-15 | 2014-04-09 | 深圳市华星光电技术有限公司 | 用于液晶显示器的液晶介质组合物 |
| CN103626660A (zh) * | 2012-08-23 | 2014-03-12 | 奇美电子股份有限公司 | 用于液晶层或配向层的感旋光性单体、使用其的液晶显示面板及其制作方法 |
| US9228130B2 (en) * | 2013-06-25 | 2016-01-05 | Shenzhen China Star Optoelectronics Technology Co., Ltd | Alignment film, a method of fabricating the same, and a liquid crystal display using the same |
| US9274378B2 (en) * | 2013-06-25 | 2016-03-01 | Shenzhen China Star Optoelectronics Technology Co., Ltd. | Alignment film, a method of fabricating the same, and a liquid crystal display using the same |
| US20160076798A1 (en) * | 2014-09-15 | 2016-03-17 | Nascent Devices Llc | Methods to enhance the performance of electrocaloric dielectric polymer |
| KR20160056197A (ko) * | 2014-11-11 | 2016-05-19 | 삼성전자주식회사 | 디스플레이 제어 방법 및 그 전자 장치 |
| CN107003573A (zh) * | 2014-11-28 | 2017-08-01 | 夏普株式会社 | 液晶显示装置的制造方法 |
| KR20170037763A (ko) * | 2015-09-25 | 2017-04-05 | 삼성디스플레이 주식회사 | 액정표시장치 및 이의 제조방법 |
| CN105487301B (zh) * | 2016-02-15 | 2018-11-23 | 深圳市华星光电技术有限公司 | 垂直光配向方法及液晶显示面板的制作方法 |
| CN106085464B (zh) * | 2016-06-02 | 2019-01-15 | 深圳市华星光电技术有限公司 | 配向膜材料、液晶显示面板的制作方法及液晶显示面板 |
| CN106188540B (zh) * | 2016-07-12 | 2019-01-22 | 深圳市华星光电技术有限公司 | 配向膜材料及配向膜的制作方法、液晶显示面板及其制作方法 |
| CN106009015B (zh) * | 2016-07-15 | 2019-04-02 | 深圳市华星光电技术有限公司 | 导电聚合物薄膜及其制作方法与液晶显示面板 |
| WO2018021093A1 (ja) | 2016-07-26 | 2018-02-01 | シャープ株式会社 | 走査アンテナおよび走査アンテナの製造方法 |
| US11067860B2 (en) * | 2016-11-18 | 2021-07-20 | Magic Leap, Inc. | Liquid crystal diffractive devices with nano-scale pattern and methods of manufacturing the same |
| CN107247369B (zh) * | 2017-07-20 | 2019-12-31 | 深圳市华星光电半导体显示技术有限公司 | 一种液晶滴下方法 |
| JP2019128411A (ja) * | 2018-01-23 | 2019-08-01 | シャープ株式会社 | 配向膜付き基板、及び、液晶表示装置 |
| JP2019152835A (ja) * | 2018-03-06 | 2019-09-12 | シャープ株式会社 | 配向膜付き基板の製造方法 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06202118A (ja) * | 1992-12-28 | 1994-07-22 | Chisso Corp | 液晶素子 |
| JPH07101904A (ja) * | 1993-10-01 | 1995-04-18 | Seiko Epson Corp | ビフェニルメタクリレート誘導体及びそれを用いた高分子分散型液晶表示素子 |
| JPH09185066A (ja) * | 1995-12-28 | 1997-07-15 | Japan Synthetic Rubber Co Ltd | 液晶配向剤 |
| JP2003261511A (ja) * | 2001-12-27 | 2003-09-19 | Merck Patent Gmbh | 重合可能な単環式化合物 |
| JP2007304509A (ja) * | 2006-05-15 | 2007-11-22 | Jsr Corp | 液晶配向剤および液晶表示素子 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5786041A (en) | 1995-06-07 | 1998-07-28 | International Business Machines Corporation | Alignment film, a method for producing the alignment film and a liquid crystal display device using the alignment film |
| JP3850002B2 (ja) | 1998-06-08 | 2006-11-29 | シャープ株式会社 | 液晶電気光学装置 |
| CN100373237C (zh) * | 2002-05-23 | 2008-03-05 | 日产化学工业株式会社 | 液晶取向处理剂、液晶取向膜和液晶显示元件 |
| AU2003218476A1 (en) * | 2002-05-31 | 2003-12-19 | Elsicon, Inc. | Hybrid polymer materials for liquid crystal alignment layers |
| TWI337679B (en) * | 2003-02-04 | 2011-02-21 | Sipix Imaging Inc | Novel compositions and assembly process for liquid crystal display |
| JP4666398B2 (ja) | 2005-05-13 | 2011-04-06 | シャープ株式会社 | 液晶表示装置 |
| JP5183165B2 (ja) * | 2006-11-21 | 2013-04-17 | 富士フイルム株式会社 | 複屈折パターンを有する物品の製造方法 |
| JP5007145B2 (ja) | 2007-04-24 | 2012-08-22 | パナソニック株式会社 | 無線認証システムおよびそのセンサ |
| DE102008056221A1 (de) | 2007-11-30 | 2009-06-04 | Merck Patent Gmbh | Polymerisierbare Verbindungen |
-
2009
- 2009-06-23 CN CN200980142070.8A patent/CN102197333B/zh active Active
- 2009-06-23 US US13/125,404 patent/US8345201B2/en active Active
- 2009-06-23 JP JP2010534653A patent/JP5198577B2/ja active Active
- 2009-06-23 WO PCT/JP2009/002862 patent/WO2010047011A1/ja active Application Filing
- 2009-06-23 BR BRPI0919916A patent/BRPI0919916A2/pt not_active IP Right Cessation
- 2009-06-23 EP EP20090821712 patent/EP2352060B1/en not_active Not-in-force
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06202118A (ja) * | 1992-12-28 | 1994-07-22 | Chisso Corp | 液晶素子 |
| JPH07101904A (ja) * | 1993-10-01 | 1995-04-18 | Seiko Epson Corp | ビフェニルメタクリレート誘導体及びそれを用いた高分子分散型液晶表示素子 |
| JPH09185066A (ja) * | 1995-12-28 | 1997-07-15 | Japan Synthetic Rubber Co Ltd | 液晶配向剤 |
| JP2003261511A (ja) * | 2001-12-27 | 2003-09-19 | Merck Patent Gmbh | 重合可能な単環式化合物 |
| JP2007304509A (ja) * | 2006-05-15 | 2007-11-22 | Jsr Corp | 液晶配向剤および液晶表示素子 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102197333B (zh) | 2014-04-16 |
| EP2352060A4 (en) | 2012-03-21 |
| US8345201B2 (en) | 2013-01-01 |
| CN102197333A (zh) | 2011-09-21 |
| US20110199566A1 (en) | 2011-08-18 |
| JPWO2010047011A1 (ja) | 2012-03-15 |
| EP2352060A1 (en) | 2011-08-03 |
| BRPI0919916A2 (pt) | 2016-03-29 |
| EP2352060B1 (en) | 2013-05-15 |
| WO2010047011A1 (ja) | 2010-04-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5198577B2 (ja) | 配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその形成方法 | |
| JP5198580B2 (ja) | 配向膜および配向膜を有する液晶表示装置ならびに配向膜の形成方法 | |
| JP5357163B2 (ja) | 配向膜、配向膜材料および配向膜を有する液晶表示装置ならびにその製造方法 | |
| JP5296096B2 (ja) | 液晶表示装置及びその製造方法 | |
| JP5357153B2 (ja) | 液晶表示装置およびその製造方法 | |
| JP5237439B2 (ja) | 液晶表示装置、液晶表示装置の製造方法、psa層形成用組成物、及び、psa層形成用液晶組成物 | |
| US20110267574A1 (en) | Liquid crystal display device and composition for forming liquid crystal layer | |
| WO2010106915A1 (ja) | 液晶表示装置及びその製造方法 | |
| WO2013080777A1 (ja) | 液晶表示装置 | |
| WO2012014803A1 (ja) | 液晶表示装置およびその製造方法 | |
| WO2012063936A1 (ja) | 液晶表示装置、及び、液晶表示装置の製造方法 | |
| WO2012063938A1 (ja) | 液晶表示装置、及び、液晶表示装置の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20121113 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20121213 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20130115 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20130206 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20160215 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 Ref document number: 5198577 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313113 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |