JP4932498B2 - 創傷用多層包帯 - Google Patents
創傷用多層包帯 Download PDFInfo
- Publication number
- JP4932498B2 JP4932498B2 JP2006552691A JP2006552691A JP4932498B2 JP 4932498 B2 JP4932498 B2 JP 4932498B2 JP 2006552691 A JP2006552691 A JP 2006552691A JP 2006552691 A JP2006552691 A JP 2006552691A JP 4932498 B2 JP4932498 B2 JP 4932498B2
- Authority
- JP
- Japan
- Prior art keywords
- wound
- layer
- dressing
- absorbent core
- fibers
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 206010052428 Wound Diseases 0.000 claims abstract description 182
- 208000027418 Wounds and injury Diseases 0.000 claims abstract description 182
- 230000002745 absorbent Effects 0.000 claims abstract description 91
- 239000002250 absorbent Substances 0.000 claims abstract description 91
- 210000000416 exudates and transudate Anatomy 0.000 claims abstract description 44
- 239000010410 layer Substances 0.000 claims description 180
- 239000011162 core material Substances 0.000 claims description 82
- 239000000835 fiber Substances 0.000 claims description 76
- 239000012530 fluid Substances 0.000 claims description 52
- 239000000853 adhesive Substances 0.000 claims description 44
- 230000001070 adhesive effect Effects 0.000 claims description 43
- 239000012790 adhesive layer Substances 0.000 claims description 41
- 239000000203 mixture Substances 0.000 claims description 28
- 230000007480 spreading Effects 0.000 claims description 28
- 238000003892 spreading Methods 0.000 claims description 28
- 238000004873 anchoring Methods 0.000 claims description 19
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 8
- 239000004952 Polyamide Substances 0.000 claims description 7
- 238000010521 absorption reaction Methods 0.000 claims description 7
- 229920002647 polyamide Polymers 0.000 claims description 7
- 229920003043 Cellulose fiber Polymers 0.000 claims description 6
- 229920005830 Polyurethane Foam Polymers 0.000 claims description 4
- 239000011496 polyurethane foam Substances 0.000 claims description 4
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 claims description 3
- 239000006260 foam Substances 0.000 claims description 3
- 229920003123 carboxymethyl cellulose sodium Polymers 0.000 claims description 2
- 229940063834 carboxymethylcellulose sodium Drugs 0.000 claims description 2
- 229940079593 drug Drugs 0.000 claims description 2
- 239000003814 drug Substances 0.000 claims description 2
- 229920006264 polyurethane film Polymers 0.000 claims description 2
- 230000035699 permeability Effects 0.000 claims 1
- 230000005540 biological transmission Effects 0.000 abstract description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 11
- 230000006835 compression Effects 0.000 description 11
- 238000007906 compression Methods 0.000 description 11
- 229920000297 Rayon Polymers 0.000 description 8
- 229920002367 Polyisobutene Polymers 0.000 description 6
- 239000004744 fabric Substances 0.000 description 6
- 239000011780 sodium chloride Substances 0.000 description 6
- 229920000433 Lyocell Polymers 0.000 description 5
- 230000009471 action Effects 0.000 description 5
- 239000000416 hydrocolloid Substances 0.000 description 5
- 239000004745 nonwoven fabric Substances 0.000 description 5
- 230000008901 benefit Effects 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 229920000728 polyester Polymers 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 239000004753 textile Substances 0.000 description 4
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 3
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 3
- 229940072056 alginate Drugs 0.000 description 3
- 235000010443 alginic acid Nutrition 0.000 description 3
- 229920000615 alginic acid Polymers 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 229920001661 Chitosan Polymers 0.000 description 2
- 208000001840 Dandruff Diseases 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000001110 calcium chloride Substances 0.000 description 2
- 229910001628 calcium chloride Inorganic materials 0.000 description 2
- 125000002057 carboxymethyl group Chemical class [H]OC(=O)C([H])([H])[*] 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 125000002791 glucosyl group Chemical group C1([C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 2
- 150000004676 glycans Chemical class 0.000 description 2
- 239000001814 pectin Substances 0.000 description 2
- 235000010987 pectin Nutrition 0.000 description 2
- 229920001277 pectin Polymers 0.000 description 2
- 229920001282 polysaccharide Polymers 0.000 description 2
- 239000005017 polysaccharide Substances 0.000 description 2
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 229920000569 Gum karaya Polymers 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- 244000046052 Phaseolus vulgaris Species 0.000 description 1
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 1
- 239000004820 Pressure-sensitive adhesive Substances 0.000 description 1
- 241000934878 Sterculia Species 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 210000003484 anatomy Anatomy 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 238000003287 bathing Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229920005549 butyl rubber Polymers 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 238000009960 carding Methods 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- FOCAUTSVDIKZOP-UHFFFAOYSA-N chloroacetic acid Chemical compound OC(=O)CCl FOCAUTSVDIKZOP-UHFFFAOYSA-N 0.000 description 1
- 229940106681 chloroacetic acid Drugs 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 238000002788 crimping Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 230000032798 delamination Effects 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 229940014259 gelatin Drugs 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 239000000231 karaya gum Substances 0.000 description 1
- 235000010494 karaya gum Nutrition 0.000 description 1
- 229940039371 karaya gum Drugs 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 229960000292 pectin Drugs 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 239000004627 regenerated cellulose Substances 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 238000009987 spinning Methods 0.000 description 1
- 229920006132 styrene block copolymer Polymers 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 231100000397 ulcer Toxicity 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
- 239000002759 woven fabric Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/02—Adhesive bandages or dressings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/02—Adhesive bandages or dressings
- A61F13/0203—Adhesive bandages or dressings with fluid retention members
- A61F13/022—Adhesive bandages or dressings with fluid retention members having more than one layer with different fluid retention characteristics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/02—Adhesive bandages or dressings
- A61F13/0203—Adhesive bandages or dressings with fluid retention members
- A61F13/0223—Adhesive bandages or dressings with fluid retention members characterized by parametric properties of the fluid retention layer, e.g. absorbency, wicking capacity, liquid distribution
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/02—Adhesive bandages or dressings
- A61F13/0246—Adhesive bandages or dressings characterised by the skin-adhering layer
- A61F13/0253—Adhesive bandages or dressings characterised by the skin-adhering layer characterized by the adhesive material
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F2013/00089—Wound bandages
- A61F2013/00246—Wound bandages in a special way pervious to air or vapours
- A61F2013/00263—Wound bandages in a special way pervious to air or vapours vapour permeability >500 g/m2/24h
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F2013/00361—Plasters
- A61F2013/00544—Plasters form or structure
- A61F2013/00604—Multilayer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F2013/00361—Plasters
- A61F2013/00727—Plasters means for wound humidity control
- A61F2013/00731—Plasters means for wound humidity control with absorbing pads
- A61F2013/00744—Plasters means for wound humidity control with absorbing pads containing non-woven
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F2013/00361—Plasters
- A61F2013/00855—Plasters pervious to air or vapours
- A61F2013/00876—Plasters pervious to air or vapours vapour permeability >500 g/mg/24h
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Dermatology (AREA)
- Materials For Medical Uses (AREA)
- Golf Clubs (AREA)
- Storage Of Web-Like Or Filamentary Materials (AREA)
Description
さらに公知の包帯、特にALLEVYN(登録商標)などのフォーム包帯の欠点は、たとえば圧縮バンデージシステム(compression bandage system)下で、使用中に包帯に圧力が加わると、包帯によって吸収される滲出液は包帯からしぼり出されることが少なくないことである。さらに、包帯の滲出液を吸収する能力は、いったん圧縮されると、減少する。従って、このような包帯は、圧縮を必要あるいは体験する場合の創傷への使用は適切ではない。
高MVTRを有する透過層、
滲出液を吸収し、保持しうる吸収心材、
滲出液を吸収心材に伝送する創傷接触層、および
包帯中の滲出液の創傷領域への横広がりを制限する吸収心材および創傷接触層
から成ることを特徴とする創傷用多層包帯が提供される。
(a)高MVTRを有する透過層;
(b)接着剤;
(c)高吸収性および低い横吸上作用を有する吸収心材;および
(d)創傷接触層
から成ることを特徴とする創傷用多層包帯が提供される。
透過層は接着剤の上に重なり、接着剤は吸収心材の上に重なり、次いで吸収心材は創傷接触層の上に重なることが好ましい。
創傷接触層は好ましくは、非粘着性で滲出液を吸収心材へ伝送するように形成されている。創傷接触層は好ましくは、創傷面に湿った環境を作り、該環境は創傷治癒を導き、かつ創傷乾燥の危険を縮小する。さらに、創傷接触層の吸収特性は、典型例としてバンデージもしくは等価の圧縮部材によって加えられる圧縮下でそれほど低下しないことが好ましい。バンデージは、約40mmHgの圧力を加えるように配置されてよい。
創傷接触層は好ましくは、包帯に構造保全性を付与し、かつ吸収心材を物理的に圧迫する。使用中、創傷接触層はゲル化吸収層を物理的に圧迫するのを助けることができ、さもなければ、包帯を離層し、スライドさせる傾向を有するかもしれない。
吸収心材の好ましい横吸上作用は低く、好ましくは20mm/分である。好ましくは1〜15mm/分、より好ましくは1〜10mm/分である。
本発明での使用に好適な繊維は、通常の繊維機械を用い、たとえば切断、カーディングおよびニードリング(needling)、および要すればクリンプ加工、延伸および紡績を含むステープルルートで加工することができる。
存在する場合の接着剤は、包帯の各層を共に保持するのに役立ち、かつ好ましい粘着性包帯の実施態様において、包帯を皮膚に密着させるのに使用されてよい。接着剤組成物は好ましくは、EP−B−92999に記載されているような、水溶性ハイドロコロイドの1種以上と低分子量ポリイソブチレンの1種以上の均質ブレンドからなる。
本発明の透過層は好ましくは、1993 BP Appendix XX J1 に記載の方法で測定した、少なくとも300gm2/24時間または100〜1000gm2/24時間範囲のMVTRを有する層である。透過層は、フィルム/フォーム積層体、たとえば発泡ポリウレタンフォームがポリウレタンフォームに積層した形状にあってよい。
好ましくは透過層は、患者が入浴またはシャワーを浴びている間、創傷を湿らすことなく包帯の着用を可能ならしめる。
透過層は好ましくは、バクテリヤ、ウィルスおよび外的汚染菌に対するバリヤーであって、これによって、創傷を感染から保護する。
また該包帯は、流体広げ層(spreading layer)を含有してもよい。流体広げ層の役割は、包帯によって吸収される流体を、高MVTR透過層を横切って横に広げることである。この層は、吸収心材の創傷に面しない側に設置されてよい。流体広げ層は、100%のビスコース、ポリオレフィン型繊維またはビスコース/ポリエステルブレンドで構成されてよい。より好ましい流体広げ層は、ビスコース/ポリエステルの水を巻き込む不織布層である。
包帯の総厚みは好ましくは2〜4mmで、より好ましくは2.2〜3.7mmである。これは、包帯の使用中の適合性や選択の自由性を高める。
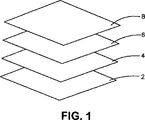
図1は、創傷用多層包帯の非粘着性の具体例の概略図である。
図2は、図1の包帯の概略横断面図である。
図3は、創傷用多層包帯の粘着性の具体例の概略図である。
図4は、図3の包帯の概略横断面図である。
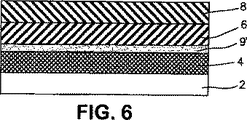
図6は、本発明に係る創傷用多層包帯であって、接着剤層と吸収心材の間に追加の定着層を含有する図2の包帯の概略横断面図である。
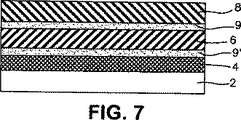
図7は、本発明に係る創傷用多層包帯であって、創傷接触層と吸収心材の間および吸収心材と接着剤層の間に追加の定着層を含有する図2の包帯の概略横断面図である。
図8は、追加の流体広げ層を含有する図5の包帯の概略横断面図である。
図10は、本発明に係る創傷用多層包帯であって、創傷接触層と吸収心材の間に追加の定着層を含有する図4の包帯の概略横断面図である。
図11は、本発明に係る創傷用多層包帯であって、吸収心材と接着剤層の間に追加の定着層を含有する図4の包帯の概略横断面図である。
図12は、本発明に係る創傷用多層包帯であって、創傷接触層と吸収心材の間および吸収心材と接着剤層の間に追加の定着層を含有する図4の包帯の概略横断面図である。
図14は、追加の流体広げ層を含有する図12の包帯の概略横断面図である。
図15は、追加の接着剤層を含有する図9の包帯の概略横断面図である。
図16は、追加の接着剤層を含有する図14の包帯の概略横断面図である。
創傷接触層は、ゲル形成繊維からなる、35gm2の水を巻き込む不織布から作られる。
透過層は、ポリウレタンフォーム/フィルム積層体である。
本発明に係る創傷用包帯は、公知の包帯に比し、圧縮下でも高い流体取扱能力を有する。典型的な圧縮は、約40mmHgで適用される。
かかる包帯で用いる材料や、該包帯の厚みは、他の公知包帯に比し、包帯の使用中の適合性や選択の自由性を高める。
比較実験から、本発明の粘着性および非粘着性バージョンは、重要な利点を有することが証明された。
2:透過層
3:中央領域
4:接着剤層
6:吸収心材
8:創傷接触層
9:定着層
9’:定着層
10:流体広げ層
14:接着剤層
15:周囲リム
16:吸収心材
18:創傷接触層
19:定着層
19’:定着層
20:流体広げ層
21:接着剤層
Claims (29)
- 多量の滲出液をもたらす創傷に用いる創傷用多層包帯であって、
少なくとも300gm2/24時間の透湿度(MVTR)を有する透過層、
滲出液を吸収し、保持しうる、ゲル形成繊維を備えている吸収心材、
滲出液を吸収心材に伝送するゲル形成繊維を備えている創傷接触層、および
吸収心材上に設置する定着層を備えており、
吸収心材および創傷接触層は、包帯中の滲出液の創傷領域への横広がりを制限する、ことを特徴とする創傷用多層包帯。 - 包帯10cm2当り少なくとも6gの流体を24時間以内で取扱うことができる請求項1に記載の創傷用多層包帯。
- 包帯10cm2当り8〜15gの流体を24時間以内で取扱うことができる請求項1又は2に記載の創傷用多層包帯。
- 包帯が粘着性である請求項1乃至3のいずれか1に記載の創傷用多層包帯。
- 包帯が非粘着性である請求項1乃至4のいずれか1に記載の創傷用多層包帯。
- ゲル形成繊維が、織物形状の化学変性セルロース系繊維である請求項1に記載の創傷用多層包帯。
- 繊維が、カルボキシメチル化セルロース繊維である請求項1に記載の創傷用多層包帯。
- 創傷接触層が、5〜40mm/分の低い横吸上速度を有する請求項1乃至7のいずれか1に記載の創傷用多層包帯。
- 創傷接触層が、25〜55gm2の繊維密度を有する請求項1乃至8のいずれか1に記載の創傷用多層包帯。
- 創傷接触層が、35gm2の繊維密度を有する請求項9に記載の創傷用多層包帯。
- 吸収心材が、少なくとも10g/gの高い滲出液吸収性を有する請求項1乃至10のいずれか1に記載の創傷用多層包帯。
- 吸収心材が、20mm/分以下の低い横吸上速度を有する請求項1乃至11のいずれか1に記載の創傷用多層包帯。
- ゲル形成繊維が好ましくは、カルボキシメチルセルロース・ナトリウム繊維である請求項1に記載の創傷用多層包帯。
- 吸収心材が、ゲル形成繊維とセルロース系繊維のブレンドである請求項1乃至13のいずれか1に記載の創傷用多層包帯。
- 吸収心材が、25重量%以下のセルロース系繊維と75〜100重量%のゲル形成繊維のブレンドである請求項14に記載の創傷用多層包帯。
- ブレンドが、50重量%以下のセルロース系繊維と50〜100重量%のゲル形成繊維である請求項14に記載の創傷用多層包帯。
- ブレンドが、約50重量%のセルロース系繊維と約50重量%のゲル形成繊維である請求項14に記載の創傷用多層包帯。
- 吸収心材の繊維密度が、150〜250gm2である請求項1乃至17のいずれか1に記載の創傷用多層包帯。
- 繊維密度が、200gm2である請求項18に記載の創傷用多層包帯。
- 包帯が湿ったときの収縮率が、25%以下である請求項1乃至19のいずれか1に記載の創傷用多層包帯。
- 収縮率が15%以下である請求項1乃至20のいずれか1に記載の創傷用多層包帯。
- 透過層がフォームである請求項1乃至21のいずれか1に記載の創傷用多層包帯。
- 透過層が、ポリウレタンフィルムに積層したポリウレタンフォームである請求項1乃至22のいずれか1に記載の創傷用多層包帯。
- 薬物を添加した可溶性フィルム層、臭気吸収層、流体広げ層、定着層および追加の接着剤層からなる群から選ばれる1つ以上の層を含有する請求項1乃至23のいずれか1に記載の創傷用多層包帯。
- 厚みが2〜4mmである請求項1乃至24のいずれか1に記載の創傷用多層包帯。
- 定着層が、吸収心材を隣接層に結合させる請求項1乃至25のいずれか1に記載の創傷用多層包帯。
- 定着層が、吸収心材の創傷に面する側または吸収心材の創傷に面しない側に設置されている請求項1乃至26のいずれか1に記載の創傷用多層包帯。
- 吸収心材と創傷接触層の間に定着層が設置されている請求項1乃至27のいずれか1に記載の創傷用多層包帯。
- 定着層がポリアミドウェブである請求項1乃至28のいずれか1に記載の創傷用多層包帯。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0403245.4 | 2004-02-13 | ||
| GBGB0403245.4A GB0403245D0 (en) | 2004-02-13 | 2004-02-13 | Multi layered wound dressing |
| GBGB0405769.1A GB0405769D0 (en) | 2004-03-15 | 2004-03-15 | Multi layered wound dressing |
| GB0405769.1 | 2004-03-15 | ||
| PCT/GB2005/000517 WO2005079718A1 (en) | 2004-02-13 | 2005-02-11 | Multi layered wound dressing |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2007521903A JP2007521903A (ja) | 2007-08-09 |
| JP4932498B2 true JP4932498B2 (ja) | 2012-05-16 |
Family
ID=34839916
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2006552691A Active JP4932498B2 (ja) | 2004-02-13 | 2005-02-11 | 創傷用多層包帯 |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US7759537B2 (ja) |
| EP (1) | EP1718257B1 (ja) |
| JP (1) | JP4932498B2 (ja) |
| AT (1) | ATE476947T1 (ja) |
| AU (1) | AU2005215212B2 (ja) |
| CA (1) | CA2556151C (ja) |
| DE (1) | DE602005022831D1 (ja) |
| DK (1) | DK1718257T3 (ja) |
| ES (1) | ES2348306T3 (ja) |
| GB (1) | GB2429161B (ja) |
| NZ (1) | NZ549226A (ja) |
| PL (1) | PL1718257T3 (ja) |
| PT (1) | PT1718257E (ja) |
| WO (1) | WO2005079718A1 (ja) |
Families Citing this family (161)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003094811A1 (en) * | 2002-05-07 | 2003-11-20 | Polyremedy Llc | Method for treating wound, dressing for use therewith and apparatus and system for fabricating dressing |
| US8407065B2 (en) * | 2002-05-07 | 2013-03-26 | Polyremedy, Inc. | Wound care treatment service using automatic wound dressing fabricator |
| GB0224986D0 (en) | 2002-10-28 | 2002-12-04 | Smith & Nephew | Apparatus |
| GB0325129D0 (en) | 2003-10-28 | 2003-12-03 | Smith & Nephew | Apparatus in situ |
| US8062272B2 (en) | 2004-05-21 | 2011-11-22 | Bluesky Medical Group Incorporated | Flexible reduced pressure treatment appliance |
| US10058642B2 (en) | 2004-04-05 | 2018-08-28 | Bluesky Medical Group Incorporated | Reduced pressure treatment system |
| US7708724B2 (en) * | 2004-04-05 | 2010-05-04 | Blue Sky Medical Group Incorporated | Reduced pressure wound cupping treatment system |
| US7776028B2 (en) | 2004-04-05 | 2010-08-17 | Bluesky Medical Group Incorporated | Adjustable overlay reduced pressure wound treatment system |
| US7909805B2 (en) | 2004-04-05 | 2011-03-22 | Bluesky Medical Group Incorporated | Flexible reduced pressure treatment appliance |
| GB0409446D0 (en) | 2004-04-28 | 2004-06-02 | Smith & Nephew | Apparatus |
| GB0409444D0 (en) * | 2004-04-28 | 2004-06-02 | Smith & Nephew | Apparatus |
| US8529548B2 (en) | 2004-04-27 | 2013-09-10 | Smith & Nephew Plc | Wound treatment apparatus and method |
| CN102715984B (zh) | 2005-09-06 | 2014-07-09 | 施乐辉股份有限公司 | 具有微型泵的独立伤口敷料 |
| AU2006287460A1 (en) | 2005-09-07 | 2007-03-15 | Tyco Healthcare Group Lp | Wound dressing with vacuum reservoir |
| WO2007089763A2 (en) | 2006-01-31 | 2007-08-09 | Tyco Healthcare Group Lp | Super soft foams |
| US7779625B2 (en) | 2006-05-11 | 2010-08-24 | Kalypto Medical, Inc. | Device and method for wound therapy |
| JP4874723B2 (ja) * | 2006-06-26 | 2012-02-15 | 国防部軍備局中山科学研究院 | 銀ナノ医療湿布材 |
| US9820888B2 (en) | 2006-09-26 | 2017-11-21 | Smith & Nephew, Inc. | Wound dressing |
| EP1905465B2 (en) | 2006-09-28 | 2013-11-27 | Smith & Nephew, Inc. | Portable wound therapy system |
| GB0710846D0 (en) * | 2007-06-06 | 2007-07-18 | Bristol Myers Squibb Co | A wound dressing |
| US8729121B2 (en) | 2007-06-25 | 2014-05-20 | Adhezion Biomedical, Llc | Curing accelerator and method of making |
| US7943811B2 (en) * | 2007-10-24 | 2011-05-17 | Johnson & Johnson Industrial Ltda. | Adhesive bandage and a process for manufacturing an adhesive bandage |
| EP2222261B1 (de) * | 2007-11-13 | 2018-05-09 | Medela Holding AG | Wunddrainageauflage |
| US8613952B2 (en) | 2007-11-14 | 2013-12-24 | Adhezion Biomedical, Llc | Cyanoacrylate tissue adhesives |
| GB0722820D0 (en) | 2007-11-21 | 2008-01-02 | Smith & Nephew | Vacuum assisted wound dressing |
| MX2010005553A (es) | 2007-11-21 | 2010-06-01 | Smith & Nephew | Aposito para heridas. |
| EP3360519B1 (en) | 2007-11-21 | 2020-11-18 | Smith & Nephew plc | Wound dressing |
| US8808259B2 (en) | 2007-11-21 | 2014-08-19 | T.J. Smith & Nephew Limited | Suction device and dressing |
| US11253399B2 (en) | 2007-12-06 | 2022-02-22 | Smith & Nephew Plc | Wound filling apparatuses and methods |
| US20130096518A1 (en) | 2007-12-06 | 2013-04-18 | Smith & Nephew Plc | Wound filling apparatuses and methods |
| GB0723855D0 (en) | 2007-12-06 | 2008-01-16 | Smith & Nephew | Apparatus and method for wound volume measurement |
| GB0723875D0 (en) | 2007-12-06 | 2008-01-16 | Smith & Nephew | Wound management |
| US8298200B2 (en) | 2009-06-01 | 2012-10-30 | Tyco Healthcare Group Lp | System for providing continual drainage in negative pressure wound therapy |
| US9033942B2 (en) | 2008-03-07 | 2015-05-19 | Smith & Nephew, Inc. | Wound dressing port and associated wound dressing |
| US8152785B2 (en) | 2008-03-13 | 2012-04-10 | Tyco Healthcare Group Lp | Vacuum port for vacuum wound therapy |
| US20090234306A1 (en) | 2008-03-13 | 2009-09-17 | Tyco Healthcare Group Lp | Vacuum wound therapy wound dressing with variable performance zones |
| SE533170C2 (sv) * | 2008-04-09 | 2010-07-13 | Moelnlycke Health Care Ab | Anordning för att behandla sår med undertryck |
| US20100241447A1 (en) * | 2008-04-25 | 2010-09-23 | Polyremedy, Inc. | Customization of wound dressing using rule-based algorithm |
| US10912869B2 (en) | 2008-05-21 | 2021-02-09 | Smith & Nephew, Inc. | Wound therapy system with related methods therefor |
| US8414519B2 (en) | 2008-05-21 | 2013-04-09 | Covidien Lp | Wound therapy system with portable container apparatus |
| US8293838B2 (en) | 2008-06-20 | 2012-10-23 | Adhezion Biomedical, Llc | Stable and sterile tissue adhesive composition with a controlled high viscosity |
| US8198344B2 (en) | 2008-06-20 | 2012-06-12 | Adhezion Biomedical, Llc | Method of preparing adhesive compositions for medical use: single additive as both the thickening agent and the accelerator |
| US20110117047A1 (en) | 2008-06-23 | 2011-05-19 | Adhezion Biomedical, Llc | Cyanoacrylate tissue adhesives with desirable permeability and tensile strength |
| US8237009B2 (en) * | 2008-06-30 | 2012-08-07 | Polyremedy, Inc. | Custom patterned wound dressings having patterned fluid flow barriers and methods of manufacturing and using same |
| US8454990B2 (en) * | 2008-08-01 | 2013-06-04 | Milliken & Company | Composite article suitable for use as a wound dressing |
| EP2309961B1 (en) | 2008-08-08 | 2017-12-06 | Smith & Nephew, Inc. | Wound dressing of continuous fibers |
| US9414968B2 (en) | 2008-09-05 | 2016-08-16 | Smith & Nephew, Inc. | Three-dimensional porous film contact layer with improved wound healing |
| US8609128B2 (en) | 2008-10-31 | 2013-12-17 | Adhezion Biomedical, Llc | Cyanoacrylate-based liquid microbial sealant drape |
| US9254133B2 (en) | 2008-10-31 | 2016-02-09 | Adhezion Biomedical, Llc | Sterilized liquid compositions of cyanoacrylate monomer mixtures |
| US8652510B2 (en) | 2008-10-31 | 2014-02-18 | Adhezion Biomedical, Llc | Sterilized liquid compositions of cyanoacrylate monomer mixtures |
| GB0902368D0 (en) | 2009-02-13 | 2009-04-01 | Smith & Nephew | Wound packing |
| US8663198B2 (en) | 2009-04-17 | 2014-03-04 | Kalypto Medical, Inc. | Negative pressure wound therapy device |
| US20100275942A1 (en) * | 2009-05-01 | 2010-11-04 | Barile Maria A | Cosmetic nail covering |
| SE533841C2 (sv) * | 2009-06-15 | 2011-02-01 | Moelnlycke Health Care Ab | Sårförband med hög vätskehanteringskapacitet |
| US20100324516A1 (en) | 2009-06-18 | 2010-12-23 | Tyco Healthcare Group Lp | Apparatus for Vacuum Bridging and/or Exudate Collection |
| US20110054429A1 (en) * | 2009-08-25 | 2011-03-03 | Sns Nano Fiber Technology, Llc | Textile Composite Material for Decontaminating the Skin |
| GB0919659D0 (en) * | 2009-11-10 | 2009-12-23 | Convatec Technologies Inc | A component for a wound dressing |
| CN102770165B (zh) | 2009-12-22 | 2016-07-06 | 史密夫和内修有限公司 | 用于负压伤口治疗的设备 |
| WO2011104388A1 (en) * | 2010-02-26 | 2011-09-01 | Vir I Kinda Ab | Wound dressing comprising a superabsorbent substance |
| US8791315B2 (en) | 2010-02-26 | 2014-07-29 | Smith & Nephew, Inc. | Systems and methods for using negative pressure wound therapy to manage open abdominal wounds |
| US9061095B2 (en) | 2010-04-27 | 2015-06-23 | Smith & Nephew Plc | Wound dressing and method of use |
| USRE48117E1 (en) | 2010-05-07 | 2020-07-28 | Smith & Nephew, Inc. | Apparatuses and methods for negative pressure wound therapy |
| US9309019B2 (en) | 2010-05-21 | 2016-04-12 | Adhezion Biomedical, Llc | Low dose gamma sterilization of liquid adhesives |
| GB201015656D0 (en) | 2010-09-20 | 2010-10-27 | Smith & Nephew | Pressure control apparatus |
| CA140189S (en) | 2010-10-15 | 2011-11-07 | Smith & Nephew | Medical dressing |
| CA140188S (en) | 2010-10-15 | 2011-11-07 | Smith & Nephew | Medical dressing |
| GB201020005D0 (en) | 2010-11-25 | 2011-01-12 | Smith & Nephew | Composition 1-1 |
| WO2012069794A1 (en) | 2010-11-25 | 2012-05-31 | Smith & Nephew Plc | Composition i-ii and products and uses thereof |
| USD714433S1 (en) | 2010-12-22 | 2014-09-30 | Smith & Nephew, Inc. | Suction adapter |
| RU2016111981A (ru) | 2010-12-22 | 2018-11-27 | Смит Энд Нефью, Инк. | Устройство и способ лечения ран отрицательным давлением |
| WO2012116099A1 (en) | 2011-02-22 | 2012-08-30 | Oleg Siniaguine | Adaptive and optionally also otherwise adaptable wound dressing |
| GB201108229D0 (en) | 2011-05-17 | 2011-06-29 | Smith & Nephew | Tissue healing |
| US9058634B2 (en) | 2011-05-24 | 2015-06-16 | Kalypto Medical, Inc. | Method for providing a negative pressure wound therapy pump device |
| BR112013030071A2 (pt) | 2011-05-24 | 2016-09-20 | Kalypto Medical Inc | dispositivo com módulos de controlador e bomba para prover uma pressão negativa para terapia de ferimento |
| US9067003B2 (en) | 2011-05-26 | 2015-06-30 | Kalypto Medical, Inc. | Method for providing negative pressure to a negative pressure wound therapy bandage |
| ITTO20110499A1 (it) | 2011-06-08 | 2012-12-09 | Emiliano Lepore | Medicazione compressiva e procedimento per la sua realizzazione |
| HUE045003T2 (hu) | 2011-07-14 | 2019-11-28 | Smith & Nephew | Sebkötözõ és kezelési eljárás |
| GB201113515D0 (en) * | 2011-08-04 | 2011-09-21 | Convatec Technologies Inc | A dressing |
| WO2013064831A1 (en) * | 2011-11-01 | 2013-05-10 | Brightwake Limited | Wound dressings, and yarn useful therein |
| US9084845B2 (en) | 2011-11-02 | 2015-07-21 | Smith & Nephew Plc | Reduced pressure therapy apparatuses and methods of using same |
| US20150159066A1 (en) | 2011-11-25 | 2015-06-11 | Smith & Nephew Plc | Composition, apparatus, kit and method and uses thereof |
| AU2013234034B2 (en) | 2012-03-12 | 2017-03-30 | Smith & Nephew Plc | Reduced pressure apparatus and methods |
| EP2827917B1 (en) | 2012-03-20 | 2019-10-16 | Smith & Nephew plc | Controlling operation of a reduced pressure therapy system based on dynamic duty cycle threshold determination |
| GB201207852D0 (en) * | 2012-05-04 | 2012-06-20 | Adv Med Solutions Ltd | Wound dressing |
| US9427505B2 (en) | 2012-05-15 | 2016-08-30 | Smith & Nephew Plc | Negative pressure wound therapy apparatus |
| AU346291S (en) | 2012-05-15 | 2013-01-09 | Smith & Nephew | Medical dressing |
| HUE047600T2 (hu) | 2012-05-23 | 2020-04-28 | Smith & Nephew | Berendezések negatív nyomású sebgyógyításhoz |
| WO2014020440A1 (en) | 2012-08-01 | 2014-02-06 | Smith & Nephew Plc | Wound dressing |
| CN104661626B (zh) | 2012-08-01 | 2018-04-06 | 史密夫及内修公开有限公司 | 伤口敷料 |
| GB201317746D0 (en) | 2013-10-08 | 2013-11-20 | Smith & Nephew | PH indicator |
| CA2902392C (en) | 2013-03-15 | 2023-08-01 | Smith & Nephew Plc | Wound dressing for negative pressure wound therapy |
| US20160120706A1 (en) | 2013-03-15 | 2016-05-05 | Smith & Nephew Plc | Wound dressing sealant and use thereof |
| US10695226B2 (en) | 2013-03-15 | 2020-06-30 | Smith & Nephew Plc | Wound dressing and method of treatment |
| EP3003236B1 (en) * | 2013-06-04 | 2019-05-15 | Gui Global Products, Ltd. | Prophylactic dressing and use of same in the prevention of infection |
| US10238547B2 (en) | 2013-06-04 | 2019-03-26 | Gui Global Products, Ltd. | Prophylactic dressing and use of same in the prevention of infection |
| EP2910224A1 (en) * | 2014-02-19 | 2015-08-26 | Swiss VX Venentherapie und Forschung GmbH | Adhesive film bandage for medical compression |
| US9421297B2 (en) | 2014-04-02 | 2016-08-23 | Adhezion Biomedical, Llc | Sterilized compositions of cyanoacrylate monomers and naphthoquinone 2,3-oxides |
| JP6586431B2 (ja) | 2014-06-18 | 2019-10-02 | スミス アンド ネフュー ピーエルシーSmith & Nephew Public Limited Company | 創傷包帯および治療方法 |
| GB201414147D0 (en) * | 2014-08-08 | 2014-09-24 | Medtrade Products Ltd | Wound dressing |
| GB201414146D0 (en) * | 2014-08-08 | 2014-09-24 | Medtrade Products Ltd | Wound dressing |
| EP3191041B1 (en) | 2014-09-10 | 2022-06-08 | C. R. Bard, Inc. | Protective dressing for skin-placed medical device |
| TW201622668A (zh) * | 2014-12-16 | 2016-07-01 | 準訊生醫股份有限公司 | 長效型貼片敷料結構 |
| AU2015370586B2 (en) | 2014-12-22 | 2020-07-16 | Smith & Nephew Plc | Negative pressure wound therapy apparatus and methods |
| US10076594B2 (en) | 2015-05-18 | 2018-09-18 | Smith & Nephew Plc | Fluidic connector for negative pressure wound therapy |
| EP3397219B1 (en) | 2015-12-30 | 2020-10-21 | Smith & Nephew plc | Absorbent negative pressure wound therapy dressing |
| CA3009878A1 (en) | 2015-12-30 | 2017-07-06 | Smith & Nephew Plc | Negative pressure wound therapy apparatus |
| JP1586115S (ja) | 2016-02-29 | 2017-09-19 | ||
| USD796735S1 (en) | 2016-02-29 | 2017-09-05 | Smith & Nephew Plc | Mount apparatus for portable negative pressure apparatus |
| CN109069708B (zh) | 2016-03-04 | 2022-04-12 | 史密夫及内修公开有限公司 | 用于乳房外科手术后的伤口的负压伤口治疗设备 |
| EP3228330A1 (en) | 2016-04-08 | 2017-10-11 | Mölnlycke Health Care AB | Composite materials in wound treatment |
| GB2555584B (en) | 2016-10-28 | 2020-05-27 | Smith & Nephew | Multi-layered wound dressing and method of manufacture |
| US11806217B2 (en) | 2016-12-12 | 2023-11-07 | Smith & Nephew Plc | Wound dressing |
| EP3384882A1 (en) | 2017-04-05 | 2018-10-10 | Mölnlycke Health Care AB | Composite material for fluid flow management |
| GB201800057D0 (en) | 2018-01-03 | 2018-02-14 | Smith & Nephew Inc | Component Positioning And stress Relief For Sensor Enabled Wound Dressings |
| JP7069219B2 (ja) | 2017-05-10 | 2022-05-17 | メンリッケ・ヘルス・ケア・アーベー | 創傷治療における複合フォーム |
| JP7272962B2 (ja) | 2017-05-15 | 2023-05-12 | スミス アンド ネフュー ピーエルシー | 創傷分析装置 |
| CN110870021A (zh) | 2017-05-15 | 2020-03-06 | 史密夫及内修公开有限公司 | 使用欧拉视频放大的负压伤口治疗系统 |
| EP3638167B1 (en) | 2017-06-14 | 2023-11-01 | T.J. Smith and Nephew, Limited | Negative pressure wound therapy apparatus |
| JP7189159B2 (ja) | 2017-06-23 | 2022-12-13 | スミス アンド ネフュー ピーエルシー | センサを有効化した創傷モニタリングまたは治療のためのセンサの配置 |
| US11554051B2 (en) | 2017-06-30 | 2023-01-17 | T.J. Smith And Nephew, Limited | Negative pressure wound therapy apparatus |
| GB201804502D0 (en) | 2018-03-21 | 2018-05-02 | Smith & Nephew | Biocompatible encapsulation and component stress relief for sensor enabled negative pressure wound therapy dressings |
| GB201803496D0 (en) | 2018-03-05 | 2018-04-18 | Smith & Nephew | Skewing pads for impedance measurement |
| GB201809007D0 (en) | 2018-06-01 | 2018-07-18 | Smith & Nephew | Restriction of sensor-monitored region for sensor-enabled wound dressings |
| WO2019030384A2 (en) | 2017-08-10 | 2019-02-14 | Smith & Nephew Plc | SENSOR POSITIONING FOR MONITORING OR TREATING SENSOR ACTIVATED WOUNDS |
| GB201713511D0 (en) | 2017-08-23 | 2017-10-04 | Scapa Uk Ltd | Wound dressing |
| CN111093477B (zh) | 2017-09-10 | 2023-09-12 | 史密夫及内修公开有限公司 | 用于检查装配有传感器的伤口敷料中的封装和部件的系统和方法 |
| GB201804971D0 (en) | 2018-03-28 | 2018-05-09 | Smith & Nephew | Electrostatic discharge protection for sensors in wound therapy |
| US11596553B2 (en) | 2017-09-27 | 2023-03-07 | Smith & Nephew Plc | Ph sensing for sensor enabled negative pressure wound monitoring and therapy apparatuses |
| WO2019072531A1 (en) | 2017-09-28 | 2019-04-18 | Smith & Nephew Plc | NEUROSTIMULATION AND MONITORING USING A SENSOR ACTIVATED WOUND SURVEILLANCE AND TREATMENT APPARATUS |
| GB201718851D0 (en) | 2017-11-15 | 2017-12-27 | Smith & Nephew | Flocked conformable circuit boards for sensor enabled wound therapy dressings and systems |
| WO2019089856A1 (en) * | 2017-10-31 | 2019-05-09 | InMEDBio, LLC | Absorbent, breathable and pathogen blocking/killing wound care dressing and fabrication thereof |
| GB201809914D0 (en) | 2018-06-18 | 2018-08-01 | Smith & Nephew | Foot Loading Monitoring Apparatus |
| US20200000640A1 (en) * | 2018-06-29 | 2020-01-02 | Milliken & Company | Multi-Layer Wound Care Device Having Absorption and Fluid Transfer Properties |
| GB201811449D0 (en) | 2018-07-12 | 2018-08-29 | Smith & Nephew | Apparatuses and methods for negative pressure wound therapy |
| GB201814011D0 (en) | 2018-08-29 | 2018-10-10 | Smith & Nephew | Componet positioning and encapsulation for sensor enabled wound dressings |
| JP2022500106A (ja) * | 2018-09-06 | 2022-01-04 | コンバテック・テクノロジーズ・インコーポレイテッドConvatec Technologies Inc | 適応可能接着性創傷被覆材 |
| US11944418B2 (en) | 2018-09-12 | 2024-04-02 | Smith & Nephew Plc | Device, apparatus and method of determining skin perfusion pressure |
| WO2020064937A1 (en) | 2018-09-28 | 2020-04-02 | T.J.Smith And Nephew,Limited | Optical fibers for optically sensing through wound dressings |
| GB201816838D0 (en) | 2018-10-16 | 2018-11-28 | Smith & Nephew | Systems and method for applying biocompatible encapsulation to sensor enabled wound monitoring and therapy dressings |
| GB2611267B (en) | 2018-10-18 | 2023-06-28 | Smith & Nephew | Tissue treatment device |
| GB201817052D0 (en) | 2018-10-19 | 2018-12-05 | Smith & Nephew | Tissue treatment device |
| GB201901242D0 (en) | 2019-01-30 | 2019-03-20 | Smith & Nephew | Optical sensing systems and methods for sensing enabled wound dressings and systems |
| GB2611914B (en) | 2019-01-30 | 2023-09-20 | Smith & Nephew | Sensor integrated dressings and systems |
| CN113365588B (zh) * | 2019-02-01 | 2022-08-30 | 凯希特许有限公司 | 部分透明的伤口敷料 |
| SG11202108551SA (en) | 2019-02-27 | 2021-09-29 | Lucas Hector Izard Smith | Breathable adhesive bandages |
| JP7529681B2 (ja) | 2019-03-18 | 2024-08-06 | スミス アンド ネフュー ピーエルシー | センサ統合基板用設計ルール |
| EP3941346A1 (en) | 2019-03-19 | 2022-01-26 | Smith & Nephew plc | Systems and methods for measuring tissue impedance |
| GB201909947D0 (en) | 2019-07-11 | 2019-08-28 | Smith & Nephew | Sensor sheet with digital distributed data acquisition for wound monitoring and treatment |
| EP4010037A4 (en) * | 2019-08-09 | 2023-09-06 | SWM Luxembourg Sarl | LOW-TRAUMA DRESSINGS AND ADHESIVES THEREFOR |
| GB201914443D0 (en) | 2019-10-07 | 2019-11-20 | Smith & Nephew | Sensor enabled negative pressure wound monitoring apparatus with different impedances inks |
| WO2021070048A1 (en) * | 2019-10-08 | 2021-04-15 | Kci Licensing, Inc. | Compression-resistant wound dressing |
| WO2021111370A1 (en) * | 2019-12-04 | 2021-06-10 | Kci Licensing, Inc. | Dressing with spacer fabric to promote medial tension |
| WO2021122701A1 (en) | 2019-12-16 | 2021-06-24 | T.J.Smith And Nephew,Limited | Monitoring and therapy devices and methods of using same |
| GB201918856D0 (en) | 2019-12-19 | 2020-02-05 | Smith & Nephew | Sensor integrated dressings and systems |
| GB202000574D0 (en) | 2020-01-15 | 2020-02-26 | Smith & Nephew | Fluidic connectors for negative pressure wound therapy |
| GB202001212D0 (en) | 2020-01-29 | 2020-03-11 | Smith & Nephew | Systems and methods for measuring and tracking wound volume |
| GB202003203D0 (en) | 2020-03-05 | 2020-04-22 | Smith & Nephew | Sensor integrated dressings and systems |
| GB202003586D0 (en) | 2020-03-12 | 2020-04-29 | Smith & Nephew | Device, apparatus and method of determining skin perfusion pressure |
| GB202005867D0 (en) | 2020-04-22 | 2020-06-03 | Smith & Nephew | Tissue treatment device |
| DK3915532T3 (da) * | 2020-05-25 | 2024-06-17 | Moelnlycke Health Care Ab | Forbinding til undertryksbhehandling af sår (npwt) |
| GB202100527D0 (en) | 2021-01-15 | 2021-03-03 | Smith & Nephew | Systems and methods for managing operation of wound dressings or wound treatment devices |
Family Cites Families (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3062209A (en) * | 1957-04-23 | 1962-11-06 | Lohmann Kg | Bandaging material for the treatment of wounds |
| US4196281A (en) * | 1976-10-20 | 1980-04-01 | Regents Of The University Of California | Psoralens |
| US4246221A (en) * | 1979-03-02 | 1981-01-20 | Akzona Incorporated | Process for shaped cellulose article prepared from a solution containing cellulose dissolved in a tertiary amine N-oxide solvent |
| US4649909A (en) * | 1981-12-11 | 1987-03-17 | Johnson & Johnson Products, Inc. | Composite surgical dressing |
| NZ203706A (en) | 1982-04-22 | 1986-03-14 | Squibb & Sons Inc | Occlusive multi-layer wound dressing and hydrocolloidal granules for wound treatment |
| US4551490A (en) | 1983-06-27 | 1985-11-05 | E. R. Squibb & Sons, Inc. | Adhesive composition resistant to biological fluids |
| US4875473A (en) * | 1986-04-03 | 1989-10-24 | Bioderm, Inc. | Multi-layer wound dressing having oxygen permeable and oxygen impermeable layers |
| US4909244B1 (en) * | 1986-11-26 | 1994-07-05 | Kendall & Co | Hydrogel wound dressing |
| GB8629566D0 (en) * | 1986-12-10 | 1987-09-09 | Lantor Uk Ltd | Composite material |
| GB2208277B (en) | 1987-07-30 | 1991-11-13 | Courtaulds Plc | Cellulosic fibre |
| EP0304536A3 (en) | 1987-08-25 | 1990-05-09 | The B.F. Goodrich Company | Polymeric wound dressings |
| GB8820564D0 (en) | 1988-08-31 | 1988-09-28 | Britcair Ltd | Wound dressing |
| US5056510A (en) | 1989-04-13 | 1991-10-15 | The Kendall Company | Vented wound dressing |
| IL97142A0 (en) * | 1990-03-02 | 1992-05-25 | Duphar Int Res | Wound dressing and its preparation |
| GB9020278D0 (en) | 1990-09-17 | 1990-10-31 | Britcair Ltd | Alginate fabric |
| DK122791D0 (da) * | 1991-06-24 | 1991-06-24 | Coloplast As | Saarbandage |
| AU2625192A (en) * | 1991-11-01 | 1993-05-06 | Ferris Mfg. Corp. | Window dressing |
| GB9126193D0 (en) | 1991-12-10 | 1992-02-12 | Courtaulds Plc | Cellulosic fibres |
| IL108373A (en) | 1993-01-20 | 1996-11-14 | C V Lab Ltd | Alginate fibers with a high absorption capacity and their preparation |
| GB9301258D0 (en) | 1993-01-22 | 1993-03-17 | Courtaulds Plc | Use of absorbent fibre |
| US5681579A (en) * | 1993-03-22 | 1997-10-28 | E.R. Squibb & Sons, Inc. | Polymeric support wound dressing |
| US5695777A (en) * | 1994-05-10 | 1997-12-09 | Medtronic, Inc. | Absorptive wound dressing for wound healing promotion |
| US5607388A (en) * | 1994-06-16 | 1997-03-04 | Hercules Incorporated | Multi-purpose wound dressing |
| US5653699A (en) * | 1994-09-13 | 1997-08-05 | Polymedica Industries, Inc. | Spyrosorbent wound dressings for exudate management |
| US5512041A (en) * | 1994-10-07 | 1996-04-30 | Scott Health Care | Wound dressing for promoting moist wound healing |
| US5571079A (en) * | 1994-12-30 | 1996-11-05 | Algos Pharmaceutical Corporation | Wound dressing |
| ES2157568T3 (es) | 1996-04-12 | 2001-08-16 | Bristol Myers Squibb Co | Fibras compuestas, apositos para heridas que incorporan dichas fibras y procedimiento de fabricacion de las mismas. |
| US5968001A (en) * | 1996-05-14 | 1999-10-19 | Bristol-Myers Squibb Company | Wound dressings with leak prevention seal |
| GB9701178D0 (en) * | 1997-01-21 | 1997-03-12 | Smith & Nephew | Dressings |
| JP2002519153A (ja) | 1998-07-01 | 2002-07-02 | アコーディス スペシャリティー ファイバーズ リミティド | 傷手当用品及びその製造並びにその使用に適切な材料の製造 |
| GB9900348D0 (en) * | 1999-01-09 | 1999-02-24 | Bristol Myers Squibb Co | Multi layered wound dressing |
| GB2380945B (en) | 1999-01-09 | 2003-06-04 | Bristol Myers Squibb Co | Multi layered wound dressing |
| GB2356145B (en) | 1999-11-10 | 2004-07-28 | Mas Mfg Ltd | Dressing |
| US6566575B1 (en) * | 2000-02-15 | 2003-05-20 | 3M Innovative Properties Company | Patterned absorbent article for wound dressing |
| GB0017080D0 (en) * | 2000-07-12 | 2000-08-30 | Bristol Myers Squibb Co | Multi layered wound dresssing |
| US6903243B1 (en) * | 2000-09-08 | 2005-06-07 | 3M Innovative Properties Company | Multi-layer absorbent wound dressing |
| GB2370226A (en) * | 2000-09-21 | 2002-06-26 | Acordis Speciality Fibres Ltd | Wound dressing |
| GB0124530D0 (en) * | 2001-10-12 | 2001-12-05 | Acordis Speciality Fibres Ltd | Improved fabric |
| GB2382305B (en) * | 2001-11-23 | 2004-12-15 | Johnson & Johnson Medical Ltd | Absorbent wound dressings containing a hydrogel layer |
| JP2006527007A (ja) * | 2003-06-10 | 2006-11-30 | ビーエスエヌ メディカル,インク. | 横糸挿入伸縮性接着包帯及びその製造方法 |
-
2005
- 2005-02-11 NZ NZ549226A patent/NZ549226A/en not_active IP Right Cessation
- 2005-02-11 JP JP2006552691A patent/JP4932498B2/ja active Active
- 2005-02-11 EP EP05708336A patent/EP1718257B1/en active Active
- 2005-02-11 GB GB0617356A patent/GB2429161B/en active Active
- 2005-02-11 PT PT05708336T patent/PT1718257E/pt unknown
- 2005-02-11 WO PCT/GB2005/000517 patent/WO2005079718A1/en active Application Filing
- 2005-02-11 DK DK05708336.2T patent/DK1718257T3/da active
- 2005-02-11 US US11/055,931 patent/US7759537B2/en active Active
- 2005-02-11 ES ES05708336T patent/ES2348306T3/es active Active
- 2005-02-11 DE DE602005022831T patent/DE602005022831D1/de active Active
- 2005-02-11 PL PL05708336T patent/PL1718257T3/pl unknown
- 2005-02-11 AT AT05708336T patent/ATE476947T1/de active
- 2005-02-11 CA CA2556151A patent/CA2556151C/en active Active
- 2005-02-11 AU AU2005215212A patent/AU2005215212B2/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| ATE476947T1 (de) | 2010-08-15 |
| ES2348306T3 (es) | 2010-12-02 |
| DE602005022831D1 (de) | 2010-09-23 |
| US20050182347A1 (en) | 2005-08-18 |
| PL1718257T3 (pl) | 2011-01-31 |
| NZ549226A (en) | 2010-06-25 |
| JP2007521903A (ja) | 2007-08-09 |
| EP1718257B1 (en) | 2010-08-11 |
| US7759537B2 (en) | 2010-07-20 |
| GB0617356D0 (en) | 2006-10-18 |
| GB2429161B (en) | 2008-12-24 |
| WO2005079718A1 (en) | 2005-09-01 |
| CA2556151A1 (en) | 2005-09-01 |
| AU2005215212A1 (en) | 2005-09-01 |
| GB2429161A (en) | 2007-02-21 |
| AU2005215212B2 (en) | 2010-11-25 |
| DK1718257T3 (da) | 2010-10-18 |
| PT1718257E (pt) | 2010-10-21 |
| EP1718257A1 (en) | 2006-11-08 |
| CA2556151C (en) | 2013-07-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4932498B2 (ja) | 創傷用多層包帯 | |
| US8404921B2 (en) | Wound dressing | |
| US6552244B1 (en) | Multi-layered wound dressing | |
| JP4783540B2 (ja) | 多層創傷包帯 | |
| US10543133B2 (en) | Component for a wound dressing | |
| JP4662488B2 (ja) | 創傷用包帯 | |
| JP2005102790A (ja) | 創傷被覆材 | |
| GB2380945A (en) | Multi-layered wound dressing |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20080208 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20110111 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20110408 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20110415 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20110511 |
|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A711 Effective date: 20110511 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20110608 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20110613 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20110620 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110711 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20120131 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20120215 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4932498 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20150224 Year of fee payment: 3 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |