JP4723584B2 - 生物学的、生物分解性若しくは合成重合体又は繊維が巻き付けられた体腔を閉塞するための金属製コイル - Google Patents
生物学的、生物分解性若しくは合成重合体又は繊維が巻き付けられた体腔を閉塞するための金属製コイル Download PDFInfo
- Publication number
- JP4723584B2 JP4723584B2 JP2007527550A JP2007527550A JP4723584B2 JP 4723584 B2 JP4723584 B2 JP 4723584B2 JP 2007527550 A JP2007527550 A JP 2007527550A JP 2007527550 A JP2007527550 A JP 2007527550A JP 4723584 B2 JP4723584 B2 JP 4723584B2
- Authority
- JP
- Japan
- Prior art keywords
- coil
- fiber
- fibers
- stretchable member
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000000835 fiber Substances 0.000 title claims abstract description 118
- 229910052751 metal Inorganic materials 0.000 title claims description 32
- 239000002184 metal Substances 0.000 title claims description 32
- 229920002988 biodegradable polymer Polymers 0.000 title claims description 11
- 229920001059 synthetic polymer Polymers 0.000 title description 2
- 239000000463 material Substances 0.000 claims description 61
- 230000000975 bioactive effect Effects 0.000 claims description 44
- 238000000576 coating method Methods 0.000 claims description 36
- 239000011248 coating agent Substances 0.000 claims description 34
- -1 di-saccharides Polymers 0.000 claims description 18
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 17
- 238000004804 winding Methods 0.000 claims description 17
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 claims description 15
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 claims description 14
- 238000000034 method Methods 0.000 claims description 13
- 229920001577 copolymer Polymers 0.000 claims description 10
- 229920000642 polymer Polymers 0.000 claims description 10
- 108010035532 Collagen Proteins 0.000 claims description 9
- 102000008186 Collagen Human genes 0.000 claims description 9
- 239000004621 biodegradable polymer Substances 0.000 claims description 9
- 229920001436 collagen Polymers 0.000 claims description 9
- 150000002739 metals Chemical class 0.000 claims description 8
- 235000014655 lactic acid Nutrition 0.000 claims description 7
- 239000004310 lactic acid Substances 0.000 claims description 7
- 229910052697 platinum Inorganic materials 0.000 claims description 7
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 7
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims description 6
- 229920000954 Polyglycolide Polymers 0.000 claims description 6
- 208000007536 Thrombosis Diseases 0.000 claims description 6
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 6
- 239000004677 Nylon Substances 0.000 claims description 5
- 230000017531 blood circulation Effects 0.000 claims description 5
- 239000003102 growth factor Substances 0.000 claims description 5
- 229920001778 nylon Polymers 0.000 claims description 5
- 239000004633 polyglycolic acid Substances 0.000 claims description 5
- 108091034117 Oligonucleotide Proteins 0.000 claims description 4
- 239000004372 Polyvinyl alcohol Substances 0.000 claims description 4
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 claims description 4
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 claims description 4
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 claims description 4
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 claims description 4
- 230000015572 biosynthetic process Effects 0.000 claims description 4
- 230000021164 cell adhesion Effects 0.000 claims description 4
- 239000005020 polyethylene terephthalate Substances 0.000 claims description 4
- 229920002451 polyvinyl alcohol Polymers 0.000 claims description 4
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims description 4
- 239000001267 polyvinylpyrrolidone Substances 0.000 claims description 4
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims description 4
- 108090000623 proteins and genes Proteins 0.000 claims description 4
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 claims description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 3
- 239000004952 Polyamide Substances 0.000 claims description 3
- 229920001710 Polyorthoester Polymers 0.000 claims description 3
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 claims description 3
- 239000002253 acid Substances 0.000 claims description 3
- 229910045601 alloy Inorganic materials 0.000 claims description 3
- 239000000956 alloy Substances 0.000 claims description 3
- 150000004676 glycans Polymers 0.000 claims description 3
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 claims description 3
- 229910052737 gold Inorganic materials 0.000 claims description 3
- 239000010931 gold Substances 0.000 claims description 3
- 230000002209 hydrophobic effect Effects 0.000 claims description 3
- 229910052741 iridium Inorganic materials 0.000 claims description 3
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 claims description 3
- 229910052763 palladium Inorganic materials 0.000 claims description 3
- 239000002745 poly(ortho ester) Substances 0.000 claims description 3
- 229920002647 polyamide Polymers 0.000 claims description 3
- 229920001610 polycaprolactone Polymers 0.000 claims description 3
- 239000004632 polycaprolactone Substances 0.000 claims description 3
- 229920000728 polyester Polymers 0.000 claims description 3
- 239000004626 polylactic acid Substances 0.000 claims description 3
- 239000005017 polysaccharide Substances 0.000 claims description 3
- 229910052702 rhenium Inorganic materials 0.000 claims description 3
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 claims description 3
- 229910052703 rhodium Inorganic materials 0.000 claims description 3
- 239000010948 rhodium Substances 0.000 claims description 3
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 claims description 3
- 229910052709 silver Inorganic materials 0.000 claims description 3
- 239000004332 silver Substances 0.000 claims description 3
- 229910052715 tantalum Inorganic materials 0.000 claims description 3
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 claims description 3
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims description 3
- 229910052721 tungsten Inorganic materials 0.000 claims description 3
- 239000010937 tungsten Substances 0.000 claims description 3
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 claims description 3
- 229920000742 Cotton Polymers 0.000 claims description 2
- 102000008946 Fibrinogen Human genes 0.000 claims description 2
- 108010049003 Fibrinogen Proteins 0.000 claims description 2
- 239000004698 Polyethylene Substances 0.000 claims description 2
- 229920000331 Polyhydroxybutyrate Polymers 0.000 claims description 2
- 239000004743 Polypropylene Substances 0.000 claims description 2
- 239000004793 Polystyrene Substances 0.000 claims description 2
- 108010031318 Vitronectin Proteins 0.000 claims description 2
- 102100035140 Vitronectin Human genes 0.000 claims description 2
- 229920002301 cellulose acetate Polymers 0.000 claims description 2
- 229940012952 fibrinogen Drugs 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 239000005015 poly(hydroxybutyrate) Substances 0.000 claims description 2
- 229920000747 poly(lactic acid) Polymers 0.000 claims description 2
- 229920001707 polybutylene terephthalate Polymers 0.000 claims description 2
- 239000004417 polycarbonate Substances 0.000 claims description 2
- 229920000515 polycarbonate Polymers 0.000 claims description 2
- 229920000573 polyethylene Polymers 0.000 claims description 2
- 229920000139 polyethylene terephthalate Polymers 0.000 claims description 2
- 229920001155 polypropylene Polymers 0.000 claims description 2
- 229920001299 polypropylene fumarate Polymers 0.000 claims description 2
- 229920002223 polystyrene Polymers 0.000 claims description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 claims description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 claims description 2
- 229920002635 polyurethane Polymers 0.000 claims description 2
- 239000004814 polyurethane Substances 0.000 claims description 2
- 229920002689 polyvinyl acetate Polymers 0.000 claims description 2
- 239000011118 polyvinyl acetate Substances 0.000 claims description 2
- 239000004800 polyvinyl chloride Substances 0.000 claims description 2
- 229920000915 polyvinyl chloride Polymers 0.000 claims description 2
- 229940070710 valerate Drugs 0.000 claims description 2
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 claims description 2
- IYMAXBFPHPZYIK-BQBZGAKWSA-N Arg-Gly-Asp Chemical compound NC(N)=NCCC[C@H](N)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(O)=O IYMAXBFPHPZYIK-BQBZGAKWSA-N 0.000 claims 2
- 229920001222 biopolymer Polymers 0.000 claims 2
- 150000002772 monosaccharides Chemical class 0.000 claims 2
- 150000004804 polysaccharides Polymers 0.000 claims 2
- 239000002356 single layer Substances 0.000 claims 2
- 229920002125 Sokalan® Polymers 0.000 claims 1
- 206010053648 Vascular occlusion Diseases 0.000 claims 1
- 229920000117 poly(dioxanone) Polymers 0.000 claims 1
- 239000004584 polyacrylic acid Substances 0.000 claims 1
- 230000002040 relaxant effect Effects 0.000 claims 1
- 230000002792 vascular Effects 0.000 claims 1
- 208000021331 vascular occlusion disease Diseases 0.000 claims 1
- 206010052428 Wound Diseases 0.000 description 23
- 208000027418 Wounds and injury Diseases 0.000 description 23
- 206010002329 Aneurysm Diseases 0.000 description 18
- 210000004204 blood vessel Anatomy 0.000 description 5
- 230000010102 embolization Effects 0.000 description 5
- 239000002657 fibrous material Substances 0.000 description 5
- 239000000243 solution Substances 0.000 description 5
- 239000008280 blood Substances 0.000 description 4
- 210000004369 blood Anatomy 0.000 description 4
- 230000035876 healing Effects 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- 206010061218 Inflammation Diseases 0.000 description 3
- 239000012867 bioactive agent Substances 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 239000000017 hydrogel Substances 0.000 description 3
- 230000004054 inflammatory process Effects 0.000 description 3
- 229920002994 synthetic fiber Polymers 0.000 description 3
- 229940124597 therapeutic agent Drugs 0.000 description 3
- RKDVKSZUMVYZHH-UHFFFAOYSA-N 1,4-dioxane-2,5-dione Chemical compound O=C1COC(=O)CO1 RKDVKSZUMVYZHH-UHFFFAOYSA-N 0.000 description 2
- 229920004934 Dacron® Polymers 0.000 description 2
- 208000005189 Embolism Diseases 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 201000008450 Intracranial aneurysm Diseases 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 108090000373 Tissue Plasminogen Activator Proteins 0.000 description 2
- 102000003978 Tissue Plasminogen Activator Human genes 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 210000001105 femoral artery Anatomy 0.000 description 2
- JJTUDXZGHPGLLC-UHFFFAOYSA-N lactide Chemical compound CC1OC(=O)C(C)OC1=O JJTUDXZGHPGLLC-UHFFFAOYSA-N 0.000 description 2
- 229910001000 nickel titanium Inorganic materials 0.000 description 2
- 229910000510 noble metal Inorganic materials 0.000 description 2
- 229920002401 polyacrylamide Polymers 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 229960000187 tissue plasminogen activator Drugs 0.000 description 2
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- JJTUDXZGHPGLLC-IMJSIDKUSA-N 4511-42-6 Chemical compound C[C@@H]1OC(=O)[C@H](C)OC1=O JJTUDXZGHPGLLC-IMJSIDKUSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 208000031104 Arterial Occlusive disease Diseases 0.000 description 1
- 108010017384 Blood Proteins Proteins 0.000 description 1
- 102000004506 Blood Proteins Human genes 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- 206010053567 Coagulopathies Diseases 0.000 description 1
- 102000012422 Collagen Type I Human genes 0.000 description 1
- 108010022452 Collagen Type I Proteins 0.000 description 1
- 102000004266 Collagen Type IV Human genes 0.000 description 1
- 108010042086 Collagen Type IV Proteins 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 102000007625 Hirudins Human genes 0.000 description 1
- 108010007267 Hirudins Proteins 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- 229920001410 Microfiber Polymers 0.000 description 1
- 239000004721 Polyphenylene oxide Substances 0.000 description 1
- NPXOKRUENSOPAO-UHFFFAOYSA-N Raney nickel Chemical class [Al].[Ni] NPXOKRUENSOPAO-UHFFFAOYSA-N 0.000 description 1
- 108010023197 Streptokinase Proteins 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 108090000435 Urokinase-type plasminogen activator Proteins 0.000 description 1
- 102000003990 Urokinase-type plasminogen activator Human genes 0.000 description 1
- 229910001080 W alloy Inorganic materials 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 150000008064 anhydrides Chemical class 0.000 description 1
- 210000000709 aorta Anatomy 0.000 description 1
- 210000002376 aorta thoracic Anatomy 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 230000008321 arterial blood flow Effects 0.000 description 1
- 208000021328 arterial occlusion Diseases 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 230000004323 axial length Effects 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 239000003181 biological factor Substances 0.000 description 1
- 239000012620 biological material Substances 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 210000004899 c-terminal region Anatomy 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 230000035602 clotting Effects 0.000 description 1
- 229940096422 collagen type i Drugs 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 239000013013 elastic material Substances 0.000 description 1
- 230000003073 embolic effect Effects 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 239000003527 fibrinolytic agent Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000004811 fluoropolymer Substances 0.000 description 1
- 229920002313 fluoropolymer Polymers 0.000 description 1
- 238000002594 fluoroscopy Methods 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 210000004013 groin Anatomy 0.000 description 1
- 230000023597 hemostasis Effects 0.000 description 1
- 229940006607 hirudin Drugs 0.000 description 1
- WQPDUTSPKFMPDP-OUMQNGNKSA-N hirudin Chemical compound C([C@@H](C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C=CC(OS(O)(=O)=O)=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCCCN)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H]1NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@@H]2CSSC[C@@H](C(=O)N[C@@H](CCC(O)=O)C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](C(=O)N[C@H](C(NCC(=O)N[C@@H](CCC(N)=O)C(=O)NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N2)=O)CSSC1)C(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=2C=CC(O)=CC=2)NC(=O)[C@@H](NC(=O)[C@@H](N)C(C)C)C(C)C)[C@@H](C)O)CSSC1)C(C)C)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 WQPDUTSPKFMPDP-OUMQNGNKSA-N 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- 150000002433 hydrophilic molecules Chemical class 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 239000003658 microfiber Substances 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 239000005445 natural material Substances 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- YHHSONZFOIEMCP-UHFFFAOYSA-O phosphocholine Chemical group C[N+](C)(C)CCOP(O)(O)=O YHHSONZFOIEMCP-UHFFFAOYSA-O 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- ZONODCCBXBRQEZ-UHFFFAOYSA-N platinum tungsten Chemical compound [W].[Pt] ZONODCCBXBRQEZ-UHFFFAOYSA-N 0.000 description 1
- HWLDNSXPUQTBOD-UHFFFAOYSA-N platinum-iridium alloy Chemical compound [Ir].[Pt] HWLDNSXPUQTBOD-UHFFFAOYSA-N 0.000 description 1
- 229920001432 poly(L-lactide) Polymers 0.000 description 1
- 229920002463 poly(p-dioxanone) polymer Polymers 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920005594 polymer fiber Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 235000018102 proteins Nutrition 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000007634 remodeling Methods 0.000 description 1
- 229960005202 streptokinase Drugs 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 229960000103 thrombolytic agent Drugs 0.000 description 1
- 230000009772 tissue formation Effects 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 229960005356 urokinase Drugs 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/18—Materials at least partially X-ray or laser opaque
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
- A61B17/12113—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel within an aneurysm
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
- A61B17/12145—Coils or wires having a pre-set deployed three-dimensional shape
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
- A61B17/1215—Coils or wires comprising additional materials, e.g. thrombogenic, having filaments, having fibers, being coated
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B17/12—Surgical instruments, devices or methods, e.g. tourniquets for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels, umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/1214—Coils or wires
- A61B17/12154—Coils or wires having stretch limiting means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/02—Inorganic materials
- A61L31/022—Metals or alloys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/08—Materials for coatings
- A61L31/10—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/12—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material
- A61L31/125—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material having a macromolecular matrix
- A61L31/128—Composite materials, i.e. containing one material dispersed in a matrix of the same or different material having a macromolecular matrix containing other specific inorganic fillers not covered by A61L31/126 or A61L31/127
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/16—Biologically active materials, e.g. therapeutic substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B2017/00004—(bio)absorbable, (bio)resorbable, resorptive
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods, e.g. tourniquets
- A61B2017/00526—Methods of manufacturing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/60—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a special physical form
- A61L2300/606—Coatings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/36—Materials or treatment for tissue regeneration for embolization or occlusion, e.g. vaso-occlusive compositions or devices
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Surgery (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Medical Informatics (AREA)
- Reproductive Health (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Epidemiology (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Composite Materials (AREA)
- Neurosurgery (AREA)
- Medicinal Chemistry (AREA)
- Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Materials For Medical Uses (AREA)
- Surgical Instruments (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
Description
金属コイルと装置構成、非伸縮性部材、繊維材料、生物活性材料、適用
[金属コイルと装置構成]
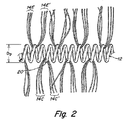
本発明の閉栓装置は、望ましくは繊維が巻き付けられている金属コイル等のコイルで作られている。ある形態では、装置は螺旋の形をし、単純に一つの螺旋で形成されている。しかし他の形態では、図1に示されるように、コイルは、2次螺旋、又は、繊維が一次コイルを通して巻き付けられた他の2次形態に巻き付けられた螺旋に付着している1次螺旋コイルを有するコイルオンコイルである。図1で、装置10は直径Dsの大きい又は二次の螺旋と、直径Dpの小さい又は一次の螺旋とを持つ金属コイル12を示している。複数の繊維14、16、18は2-2’、3-3’、4-4’に示された構成で一次螺旋のコイルを通して巻きつけられており、図2、3、3A、4にそれぞれ拡大して示されている。これら4つの巻き付け構成は代表的な例に過ぎず、繊維とコイルとが適切に結合された均等な構成もまた本発明の範囲に含まれる。
本発明の装置は非伸縮性部材を含み得る。非伸縮性部材は装置の除去、再設置等の過程での移動、変形を防ぎ得る。非伸縮性部材は少なくとも装置の一端に固定的に付着され得る。ある形態では、非伸縮性部材は装置の両端に固定的に付着され、コイルの内部を通り得る。
ある形態では、繊維は重合体材料で構成し得る。重合体材料は体内移植に許可された、あるいは許可を取得し得る材料を含み得る。これらの素材はポリエチレン、ポリアクリル酸(polyacrylics)、ポリプロピレン、ポリ塩化ビニル、ナイロン6.6等のナイロンのようなポリアミド、ポリウレタン、ポリビニルピロリドン、ポリビニルアルコール、ポリ酢酸ビニル、酢酸セルロース、ポリスチレン、ポリテトラフルオロエチレン、ポリエチレンテレフタレート(Dacron)等のポリエステル、絹、綿、などの非生物的分解性材料を含み得る。ある特定の形態では重合体要素用の非分解性材料としてポリエステル、ポリエ−テル、ポリアミド、又はポリフルオロカーボンで構成し得る。
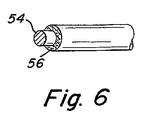
ある形態では、繊維はその中に生物活性材料を含み得る。別の形態では、生物活性材料はコイル、非伸縮性部材、繊維、又はこれらのさまざまな組み合わせを包むコーティングの中に含まれ得る。本発明のある形態では、生物活性材料のコーティングはコイルに一つの層として付けられ得て、生物活性材料を伴う又は伴わない繊維は除外することもできる。しかし、他の形態では生物活性材料は繊維の中に分散されているか、繊維の表面にコーティングされている。図6に示されるように、この構成は金属コイル又は繊維の芯30と生物活性材料からなる一層の外側コーティング32を含んでいる。
本発明の装置は特定の場所に設置し得る。したがって、本発明の装置は様々な疾患に適応し得る。例えば、動脈瘤の治療には、複数の装置で動脈瘤を埋め尽くし得る。もしくは、ただ1個の装置も投入し得る。動脈瘤に装置が設置されてからまもなく、生物活性材料の影響で塞栓が任意的に形成し得る。
AT−10のプラチナコイルが得られ、その端で平坦表面に固定された。コイルは直径0.028mmの一次螺旋を有する。直径0.099〜0.029mmの複数のVicryl90/100PGLAの縫合(suture)が得られた。これらの縫合は直径約12μmの小さなマイクロ繊維6−7本の束から構成されている。同様のサイズの単一フィラメントも使用し得る。繊維は2cm長さに切断され、図2〜5に示される構造のT-10コイルの巻きの間で物理的に巻き付けられる。この巻き付けは、1cm当り全体で約10〜20本の繊維がコイルから外方に延びるまでなされる。繊維はコイルから延びる長さが2〜4cmの長さになるよう刈り込まれる。
1.氷コンテナに冷水を満たし、温度が4〜6℃に到達するのに必要なだけ氷を追加する。
2.コンテナ/バケットに15mlの遠心管ラックを浸漬する。
3.(pH7.4±2のコラーゲン溶液で満たされた)15mlの遠心管を遠心ラックに配置する。
4.プラチナコイルを孔の開いた遠心カップを通して溶液中に挿入する。
5.コイルを溶液中に20分間放置する。
6.コイル及び(コイルをコラーゲン溶液中に確保する)管をコンテナ/バケットから除去する。
7.コイルを37℃で4時間置く。
8.コイルをコラーゲン溶液から取り出し、コイルをPBS中で3回、蒸留水中で3回リンスする。
9.コートされたコイルを1晩中乾燥させる。
Claims (36)
- a)腔(lumen)並びに第1及び第2の端部を備え、患者の血管ネットワーク中の使用部位に挿入される大きさのコイルと、
b)前記コイルの前記第1の端部に固定して取り付けられる第1の端部と前記コイルの前記第2の端部に固定して取り付けられる第2の端部とを有する非伸縮性部材と、
c)その各々が前記非伸縮性部材の回りに少なくとも2回巻かれた(wrapped)複数の繊維と、
を備える、患者の血流を閉塞するための血管閉塞装置。 - 前記非伸縮性部材は、前記コイルの前記腔(lumen)を軸方向に横切ることを特徴とする請求項1記載の装置。
- 前記非伸縮性部材は、金属ワイヤで構成されることを特徴とする請求項1記載の装置。
- 前記金属ワイヤは、白金、パラジウム、ロジウム、レニウム、イリジウム、金、銀、タングステン、タンタル、又は、これら金属のうちの2つ以上の合金、又は、超弾性金属から選択される金属から構成されることを特徴とする請求項3記載の装置。
- 前記非伸縮性部材は重合体から構成されることを特徴とする請求項1記載の装置。
- 前記重合体は、非生物分解性重合体及び生物分解性重合体のうちの一つであることを特徴とする請求項5記載の装置。
- 前記非伸縮性部材は、生物活性コーティングをさらに備えることを特徴とする請求項1記載の装置。
- 前記生物活性コーティングは、成長因子、遺伝子、オリゴヌクレオチド、ペプチド、海洋生物由来高分子(marine biopolymer)、モノ−サッカライド、ジ−サッカライド、ポリ−サッカライド、コラーゲン及びこれらの組み合わせから選択されることを特徴とする請求項7記載の装置。
- 前記生物活性コーティングは重合体を備えることを特徴とする請求項7記載の装置。
- 前記コイルは金属ワイヤで構成されることを特徴とする請求項1記載の装置。
- 前記金属ワイヤは、白金、パラジウム、ロジウム、レニウム、イリジウム、金、銀、タングステン、タンタル、又は、これら金属のうちの2つ以上の合金、又は、超弾性金属から選択される金属から構成されることを特徴とする請求項10記載の装置。
- 前記コイルは、それ自体巻かれて(wound)二次的な形状となっている一次螺旋から構成されることを特徴とする請求項1記載の装置。
- 前記二次的な形状は螺旋であることを特徴とする請求項12記載の装置。
- 前記繊維は、生物活性材料及び生物不活性材料のうちの一つであることを特徴とする請求項1記載の装置。
- 前記繊維は、グリコール酸と乳酸との共重合体から構成されることを特徴とする請求項1記載の装置。
- 前記繊維は、生物活性コーティングをさらに備えることを特徴とする請求項1記載の装置。
- 前記生物活性コーティングは、潤滑性の親水性コーティング及び疎水性コーティングのうちの一つであることを特徴とする請求項16記載の装置。
- 前記生物活性コーティングは共重合体を備えることを特徴とする請求項16記載の装置。
- 前記共重合体は乳酸とグリコール酸とから構成されることを特徴とする請求項18記載の装置。
- 前記繊維は、非生物分解性重合体から構成されることを特徴とする請求項1記載の装置。
- 前記非生物分解性重合体は、ポリエチレン、ポリアクリル酸(polyacrylic)、ポリプロピレン、ポリ塩化ビニル、ポリアミド、ポリウレタン、ポリビニルピロリドン、ポリビニルアルコール、ポリ酢酸ビニル、酢酸セルロース、ポリスチレン、ポリテトラフルオロエチレン、ポリエステル、絹、ナイロン、ポリエチレンテレフタレート又は綿から選択されることを特徴とする請求項20記載の装置。
- 前記繊維は、生物分解性重合体から構成されることを特徴とする請求項1記載の装置。
- 前記生物分解性重合体は、ポリグリコール酸、ポリカプロラクトン、ポリヒドロキシブチレートバレレート(Polyhydroxybutyrate valerate)、ポリオルトエステル(Polyorthoester)、ポリ乳酸、ポリ−p−ジオキサノン(poly(p-dioxanone))、ポリバレトラクトン(Poly(valetolactone))、ポリタルトロン酸、ポリ−β−マロン酸、ポリプロピレンフマレート、チロシンを基にしたポリカーボネート(tyrosine based polycarbonate)、及び、ポリエチレンオキサイドとポリブチレンテレフタレートとの共重合体から選択されることを特徴とする請求項22記載の装置。
- グリコール酸の乳酸に対する比は、99:1、1:99、95:5、5:95、50:50、90:10及び10:90のうちの一つに選択されることを特徴とする請求項19記載の装置。
- 前記コーティング材料は、成長因子、遺伝子、オリゴヌクレオチド、ペプチド、海洋生物由来高分子(marine biopolymer)、モノ−サッカライド、ジ−サッカライド、ポリ−サッカライド、コラーゲン及びこれらの組み合わせから選択されることを特徴とする請求項16記載の装置。
- 前記生物活性コーティングは、前記コイル及び前記非伸縮性部材に適用されることを特徴とする請求項16記載の装置。
- 前記生物活性コーティングは前記繊維に単一層として適用されることを特徴とする請求項16記載の装置。
- 前記生物活性コーティングは、前記コイル及び前記非伸縮性部材に単一層として適用されることを特徴とする請求項26記載の装置。
- 前記生物活性コーティングは、前記繊維全体にわたって分散されていることを特徴とする請求項16記載の装置。
- 前記生物活性コーティングは、細胞接着を促進し、血栓形成を促進するものであることを特徴とする請求項16記載の装置。
- 前記生物活性コーティング材料は、コラーゲン、フィブリノーゲン、ビトロネクチン、血管内皮増殖因子、及び、アルギニン−グリシン−アスパラギン酸(RGD)配列を備えるペプチドから選択されることを特徴とする請求項16記載の装置。
- 前記繊維のそれぞれは第1及び第2の端部を備え、前記第1及び第2の繊維端部のそれぞれは前記コイルから自由に延びていることを特徴とする請求項1記載の装置。
- a)腔(lumen)、第1及び第2の端部、並びに、張力がかかっていないもとで隣接するワインディング(winding)間で繊維を掴む間隔を備える螺旋状に巻かれたワイヤ芯を得る工程と、
b)前記ワイヤ芯を延伸された状態まで延伸する工程と、
c)前記螺旋状に巻かれたワイヤの前記腔(lumen)の中へ非伸縮性部材を挿入し、前記非伸縮性部材の第1の端部を前記螺旋状に巻かれたワイヤの少なくとも一つの端部に固定して取り付ける工程と、
d)前記非伸縮性部材の回りに複数の繊維を少なくとも2回巻く(wrap)工程と、
e)前記コイルを緩和し、それにより前記コイルの前記隣接するワインディング(winding)に前記繊維を掴まえさせる工程と、
を備える、血管閉塞装置を製造する方法。 - 前記非伸縮性部材の第2の端部を前記螺旋状に巻かれたワイヤの第2の端部に固定して取り付ける工程をさらに備えることを特徴とする請求項33記載の方法。
- 前記巻く工程は前記コイルの複数のワインディング(winding)を通して行われることを特徴とする請求項33記載の方法。
- 前記繊維のそれぞれは第1及び第2の端部を備え、
前記方法は、前記繊維のそれぞれの前記第1及び第2の端部を前記コイルから自由に延ばすよう構成する工程をさらに備えることを特徴とする請求項33記載の方法。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US57371004P | 2004-05-21 | 2004-05-21 | |
| US60/573,710 | 2004-05-21 | ||
| PCT/US2005/018087 WO2005113035A2 (en) | 2004-05-21 | 2005-05-23 | Metallic coils enlaced with biological or biodegradable or synthetic polymers or fibers for embolization of a body cavity |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011037486A Division JP5052682B2 (ja) | 2004-05-21 | 2011-02-23 | 生物学的、生物分解性若しくは合成重合体又は繊維が巻き付けられた体腔を閉塞するための金属製コイル |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2008500148A JP2008500148A (ja) | 2008-01-10 |
| JP2008500148A5 JP2008500148A5 (ja) | 2008-07-03 |
| JP4723584B2 true JP4723584B2 (ja) | 2011-07-13 |
Family
ID=34978919
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2007527550A Expired - Fee Related JP4723584B2 (ja) | 2004-05-21 | 2005-05-23 | 生物学的、生物分解性若しくは合成重合体又は繊維が巻き付けられた体腔を閉塞するための金属製コイル |
| JP2011037486A Active JP5052682B2 (ja) | 2004-05-21 | 2011-02-23 | 生物学的、生物分解性若しくは合成重合体又は繊維が巻き付けられた体腔を閉塞するための金属製コイル |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011037486A Active JP5052682B2 (ja) | 2004-05-21 | 2011-02-23 | 生物学的、生物分解性若しくは合成重合体又は繊維が巻き付けられた体腔を閉塞するための金属製コイル |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US7896899B2 (ja) |
| EP (2) | EP2316355B1 (ja) |
| JP (2) | JP4723584B2 (ja) |
| AT (1) | ATE490736T1 (ja) |
| CA (1) | CA2567331C (ja) |
| DE (1) | DE602005025215D1 (ja) |
| ES (2) | ES2357243T3 (ja) |
| WO (1) | WO2005113035A2 (ja) |
Families Citing this family (119)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070093889A1 (en) * | 1999-01-27 | 2007-04-26 | Wu Benjamin M | Non-Fragmenting Low Friction Bioactive Absorbable Coils for Brain Aneurysm Therapy |
| US6280457B1 (en) * | 1999-06-04 | 2001-08-28 | Scimed Life Systems, Inc. | Polymer covered vaso-occlusive devices and methods of producing such devices |
| US8048104B2 (en) * | 2000-10-30 | 2011-11-01 | Dendron Gmbh | Device for the implantation of occlusion spirals |
| AU2002345328A1 (en) | 2001-06-27 | 2003-03-03 | Remon Medical Technologies Ltd. | Method and device for electrochemical formation of therapeutic species in vivo |
| JP4351405B2 (ja) * | 2001-08-29 | 2009-10-28 | インターナショナル・ビジネス・マシーンズ・コーポレーション | トランスコーディングシステム及びアノテーション管理装置 |
| US8425549B2 (en) | 2002-07-23 | 2013-04-23 | Reverse Medical Corporation | Systems and methods for removing obstructive matter from body lumens and treating vascular defects |
| JP2004261234A (ja) * | 2003-02-20 | 2004-09-24 | Kaneka Medix Corp | 塞栓形成用体内留置具 |
| DE102004003265A1 (de) | 2004-01-21 | 2005-08-11 | Dendron Gmbh | Vorrichtung zur Implantation von elektrisch isolierten Okklusionswendeln |
| ES2357243T3 (es) | 2004-05-21 | 2011-04-20 | Micro Therapeutics, Inc. | Espirales metálicas entrelazadas con polímeros o fibras biológicos o biodegradables o sintéticos para la embolización de una cavidad corporal. |
| EP1793744B1 (de) * | 2004-09-22 | 2008-12-17 | Dendron GmbH | Medizinisches implantat |
| DE502004010411D1 (de) * | 2004-09-22 | 2009-12-31 | Dendron Gmbh | Vorrichtung zur implantation von mikrowendeln |
| US8535345B2 (en) * | 2004-10-07 | 2013-09-17 | DePuy Synthes Products, LLC | Vasoocclusive coil with biplex windings to improve mechanical properties |
| DE102005019782A1 (de) * | 2005-04-28 | 2006-11-09 | Dendron Gmbh | Vorrichtung zur Implantation von Okklusionswendeln mit innenliegendem Sicherungsmittel |
| US20070142859A1 (en) * | 2005-12-19 | 2007-06-21 | Boston Scientific Scimed, Inc. | Embolic coils |
| US8152839B2 (en) * | 2005-12-19 | 2012-04-10 | Boston Scientific Scimed, Inc. | Embolic coils |
| US8840660B2 (en) | 2006-01-05 | 2014-09-23 | Boston Scientific Scimed, Inc. | Bioerodible endoprostheses and methods of making the same |
| US8089029B2 (en) | 2006-02-01 | 2012-01-03 | Boston Scientific Scimed, Inc. | Bioabsorbable metal medical device and method of manufacture |
| US20070239194A1 (en) * | 2006-04-05 | 2007-10-11 | Boston Scientific Scimed, Inc. | Vaso-occlusive devices having expandable fibers |
| US8048150B2 (en) | 2006-04-12 | 2011-11-01 | Boston Scientific Scimed, Inc. | Endoprosthesis having a fiber meshwork disposed thereon |
| EP2015683B1 (en) * | 2006-04-17 | 2015-12-09 | Covidien LP | System for mechanically positioning intravascular implants |
| US8777979B2 (en) | 2006-04-17 | 2014-07-15 | Covidien Lp | System and method for mechanically positioning intravascular implants |
| JP2009545407A (ja) | 2006-08-02 | 2009-12-24 | ボストン サイエンティフィック サイムド,インコーポレイテッド | 三次元分解制御を備えたエンドプロテーゼ |
| ATE517590T1 (de) | 2006-09-15 | 2011-08-15 | Boston Scient Ltd | Biologisch erodierbare endoprothesen |
| JP2010503485A (ja) | 2006-09-15 | 2010-02-04 | ボストン サイエンティフィック リミテッド | 医療用デバイスおよび同デバイスの製造方法 |
| ES2368125T3 (es) | 2006-09-15 | 2011-11-14 | Boston Scientific Scimed, Inc. | Endoprótesis bioerosionable con capas inorgánicas bioestables. |
| US8808726B2 (en) | 2006-09-15 | 2014-08-19 | Boston Scientific Scimed. Inc. | Bioerodible endoprostheses and methods of making the same |
| US8002821B2 (en) | 2006-09-18 | 2011-08-23 | Boston Scientific Scimed, Inc. | Bioerodible metallic ENDOPROSTHESES |
| ATE488259T1 (de) | 2006-12-28 | 2010-12-15 | Boston Scient Ltd | Bioerodierbare endoprothesen und herstellungsverfahren dafür |
| CA2679112C (en) * | 2007-03-05 | 2015-08-11 | Boston Scientific Limited | Deploying embolic coils |
| JP5249249B2 (ja) * | 2007-03-13 | 2013-07-31 | コヴィディエン リミテッド パートナーシップ | コイルと耐伸張性部材とが含まれているインプラント |
| JP5227344B2 (ja) * | 2007-03-13 | 2013-07-03 | タイコ ヘルスケア グループ リミテッド パートナーシップ | インプラント、マンドレル、およびインプラント形成方法 |
| AU2008359873B8 (en) | 2007-07-27 | 2015-09-10 | Microvention, Inc. | Detachable coil incorporating stretch resistance |
| DE102007038446A1 (de) * | 2007-08-14 | 2009-02-19 | pfm Produkte für die Medizin AG | Embolisiereinrichtung |
| US8052745B2 (en) * | 2007-09-13 | 2011-11-08 | Boston Scientific Scimed, Inc. | Endoprosthesis |
| US20100174309A1 (en) * | 2008-05-19 | 2010-07-08 | Mindframe, Inc. | Recanalization/revascularization and embolus addressing systems including expandable tip neuro-microcatheter |
| US9220522B2 (en) * | 2007-10-17 | 2015-12-29 | Covidien Lp | Embolus removal systems with baskets |
| US8585713B2 (en) | 2007-10-17 | 2013-11-19 | Covidien Lp | Expandable tip assembly for thrombus management |
| US8088140B2 (en) | 2008-05-19 | 2012-01-03 | Mindframe, Inc. | Blood flow restorative and embolus removal methods |
| US8066757B2 (en) * | 2007-10-17 | 2011-11-29 | Mindframe, Inc. | Blood flow restoration and thrombus management methods |
| US8926680B2 (en) * | 2007-11-12 | 2015-01-06 | Covidien Lp | Aneurysm neck bridging processes with revascularization systems methods and products thereby |
| US20100256600A1 (en) * | 2009-04-04 | 2010-10-07 | Ferrera David A | Neurovascular otw pta balloon catheter and delivery system |
| US20100022951A1 (en) * | 2008-05-19 | 2010-01-28 | Luce, Forward, Hamilton 7 Scripps, Llp | Detachable hub/luer device and processes |
| US9198687B2 (en) * | 2007-10-17 | 2015-12-01 | Covidien Lp | Acute stroke revascularization/recanalization systems processes and products thereby |
| US10123803B2 (en) | 2007-10-17 | 2018-11-13 | Covidien Lp | Methods of managing neurovascular obstructions |
| US11337714B2 (en) | 2007-10-17 | 2022-05-24 | Covidien Lp | Restoring blood flow and clot removal during acute ischemic stroke |
| US8034075B2 (en) | 2007-11-09 | 2011-10-11 | Micrus Endovascular Corporation | Tethered coil for treatment of body lumens |
| CN102065779B (zh) | 2007-12-21 | 2014-02-12 | 微排放器公司 | 用于定位可脱卸植入物的脱卸区域的系统和方法 |
| WO2009089297A2 (en) * | 2008-01-07 | 2009-07-16 | Intersect Partners, Llc | Novel enhanced ptna rapid exchange type of catheter system |
| US8940003B2 (en) | 2008-02-22 | 2015-01-27 | Covidien Lp | Methods and apparatus for flow restoration |
| EP2271390A4 (en) * | 2008-04-11 | 2016-07-20 | Covidien Lp | MONORAIL NEURO MICRO CATHETER FOR DISTRIBUTING MEDICAL DEVICES FOR THE TREATMENT OF STROKE, PROCESSES AND PRODUCTS |
| AU2009239424B9 (en) * | 2008-04-21 | 2014-10-09 | Covidien Lp | Braid-ball embolic devices and delivery systems |
| US10716573B2 (en) | 2008-05-01 | 2020-07-21 | Aneuclose | Janjua aneurysm net with a resilient neck-bridging portion for occluding a cerebral aneurysm |
| US10028747B2 (en) | 2008-05-01 | 2018-07-24 | Aneuclose Llc | Coils with a series of proximally-and-distally-connected loops for occluding a cerebral aneurysm |
| WO2009137058A1 (en) | 2008-05-06 | 2009-11-12 | Metabolix, Inc. | Biodegradable polyester blends |
| US7998192B2 (en) | 2008-05-09 | 2011-08-16 | Boston Scientific Scimed, Inc. | Endoprostheses |
| US8236046B2 (en) | 2008-06-10 | 2012-08-07 | Boston Scientific Scimed, Inc. | Bioerodible endoprosthesis |
| JP5553750B2 (ja) * | 2008-06-27 | 2014-07-16 | 株式会社 京都医療設計 | 脈管用ステント |
| US7985252B2 (en) | 2008-07-30 | 2011-07-26 | Boston Scientific Scimed, Inc. | Bioerodible endoprosthesis |
| US20100030319A1 (en) * | 2008-07-31 | 2010-02-04 | Boston Scientific Scimed, Inc. | Coils for vascular implants or other uses |
| US8382824B2 (en) | 2008-10-03 | 2013-02-26 | Boston Scientific Scimed, Inc. | Medical implant having NANO-crystal grains with barrier layers of metal nitrides or fluorides |
| EP2403546A2 (en) | 2009-03-02 | 2012-01-11 | Boston Scientific Scimed, Inc. | Self-buffering medical implants |
| KR20110043799A (ko) * | 2009-10-16 | 2011-04-28 | 강호창 | 마이크로코일 어셈블리 |
| US9814562B2 (en) | 2009-11-09 | 2017-11-14 | Covidien Lp | Interference-relief type delivery detachment systems |
| US9358140B1 (en) | 2009-11-18 | 2016-06-07 | Aneuclose Llc | Stent with outer member to embolize an aneurysm |
| US8668732B2 (en) | 2010-03-23 | 2014-03-11 | Boston Scientific Scimed, Inc. | Surface treated bioerodible metal endoprostheses |
| US8353952B2 (en) * | 2010-04-07 | 2013-01-15 | Medtronic Vascular, Inc. | Stent with therapeutic substance |
| US8945171B2 (en) | 2011-09-29 | 2015-02-03 | Covidien Lp | Delivery system for implantable devices |
| US8795313B2 (en) | 2011-09-29 | 2014-08-05 | Covidien Lp | Device detachment systems with indicators |
| US9579104B2 (en) | 2011-11-30 | 2017-02-28 | Covidien Lp | Positioning and detaching implants |
| KR101315443B1 (ko) * | 2011-12-02 | 2013-10-07 | 강호창 | 마이크로코일 어셈블리 |
| US10342548B2 (en) | 2012-01-13 | 2019-07-09 | W. L. Gore & Associates, Inc. | Occlusion devices and methods of their manufacture and use |
| US9011480B2 (en) | 2012-01-20 | 2015-04-21 | Covidien Lp | Aneurysm treatment coils |
| US9687245B2 (en) | 2012-03-23 | 2017-06-27 | Covidien Lp | Occlusive devices and methods of use |
| US9326774B2 (en) | 2012-08-03 | 2016-05-03 | Covidien Lp | Device for implantation of medical devices |
| US9475930B2 (en) | 2012-08-17 | 2016-10-25 | Metabolix, Inc. | Biobased rubber modifiers for polymer blends |
| US9119948B2 (en) | 2013-02-20 | 2015-09-01 | Covidien Lp | Occlusive implants for hollow anatomical structures, delivery systems, and related methods |
| WO2014165023A1 (en) * | 2013-03-12 | 2014-10-09 | Carnegie Mellon University | Coated vaso-occclusive device for treatment of aneurysms |
| US9763667B2 (en) * | 2013-03-14 | 2017-09-19 | Cook Medical Technologies Llc | Embolization coil with barbed fiber |
| CN112716556A (zh) | 2013-03-15 | 2021-04-30 | 恩波医疗有限公司 | 栓塞系统 |
| US10660645B2 (en) | 2013-03-15 | 2020-05-26 | Embo Medical Limited | Embolization systems |
| US10076336B2 (en) | 2013-03-15 | 2018-09-18 | Covidien Lp | Delivery and detachment mechanisms for vascular implants |
| US10675039B2 (en) | 2013-03-15 | 2020-06-09 | Embo Medical Limited | Embolisation systems |
| US10669417B2 (en) | 2013-05-30 | 2020-06-02 | Cj Cheiljedang Corporation | Recyclate blends |
| FR3008304B1 (fr) * | 2013-07-15 | 2016-04-01 | Balt Extrusion | Endoprothese pour le traitement notamment d'un anevrisme |
| US10010328B2 (en) | 2013-07-31 | 2018-07-03 | NeuVT Limited | Endovascular occlusion device with hemodynamically enhanced sealing and anchoring |
| US9681876B2 (en) | 2013-07-31 | 2017-06-20 | EMBA Medical Limited | Methods and devices for endovascular embolization |
| US9877729B2 (en) | 2013-12-20 | 2018-01-30 | Microvention, Inc. | Catheter system |
| US10611903B2 (en) | 2014-03-27 | 2020-04-07 | Cj Cheiljedang Corporation | Highly filled polymer systems |
| US9713475B2 (en) | 2014-04-18 | 2017-07-25 | Covidien Lp | Embolic medical devices |
| US9808256B2 (en) | 2014-08-08 | 2017-11-07 | Covidien Lp | Electrolytic detachment elements for implant delivery systems |
| US9814466B2 (en) | 2014-08-08 | 2017-11-14 | Covidien Lp | Electrolytic and mechanical detachment for implant delivery systems |
| WO2016044188A1 (en) * | 2014-09-15 | 2016-03-24 | Jones Donald K | Intralumenal occlusion devices having improved properties |
| US10736730B2 (en) | 2015-01-20 | 2020-08-11 | Neurogami Medical, Inc. | Vascular implant |
| US11484319B2 (en) | 2015-01-20 | 2022-11-01 | Neurogami Medical, Inc. | Delivery system for micrograft for treating intracranial aneurysms |
| CA2972620C (en) | 2015-01-20 | 2023-08-01 | Neurogami Medical, Inc. | Micrograft for the treatment of intracranial aneurysms and method for use |
| US10925611B2 (en) | 2015-01-20 | 2021-02-23 | Neurogami Medical, Inc. | Packaging for surgical implant |
| US10857012B2 (en) | 2015-01-20 | 2020-12-08 | Neurogami Medical, Inc. | Vascular implant |
| JP6708200B2 (ja) * | 2015-03-03 | 2020-06-10 | 株式会社カネカメディックス | 血管塞栓用具、およびその製造方法 |
| DE202015102060U1 (de) * | 2015-04-24 | 2016-07-27 | Pfm Medical Ag | Medizinisches Implantat zum Verschluss einer Defektöffnung, eines Gefäßes, eines Organwegs oder einer anderen Öffnung in einem menschlichen oder tierischen Körper |
| US9717503B2 (en) | 2015-05-11 | 2017-08-01 | Covidien Lp | Electrolytic detachment for implant delivery systems |
| CA3014316A1 (en) | 2016-02-10 | 2017-08-17 | Microvention, Inc. | Devices for vascular occlusion |
| US20190076231A1 (en) | 2016-03-10 | 2019-03-14 | Keystone Heart Ltd. | Intra-Aortic Device |
| US10828039B2 (en) | 2016-06-27 | 2020-11-10 | Covidien Lp | Electrolytic detachment for implantable devices |
| US10828037B2 (en) | 2016-06-27 | 2020-11-10 | Covidien Lp | Electrolytic detachment with fluid electrical connection |
| US11051822B2 (en) | 2016-06-28 | 2021-07-06 | Covidien Lp | Implant detachment with thermal activation |
| WO2019027966A1 (en) | 2017-07-31 | 2019-02-07 | Boston Scientific Scimed, Inc. | DILATOR WITH REGION OF PRACTICE |
| JP7114691B2 (ja) | 2017-08-15 | 2022-08-08 | ボストン サイエンティフィック サイムド,インコーポレイテッド | 閉塞性医療装置システム |
| US10874402B2 (en) | 2017-10-10 | 2020-12-29 | Boston Scientific Scimed, Inc. | Detachable RF energized occlusive device |
| US20220104839A1 (en) | 2017-10-16 | 2022-04-07 | Retriever Medical, Inc. | Clot Removal Methods and Devices with Multiple Independently Controllable Elements |
| US20190110804A1 (en) | 2017-10-16 | 2019-04-18 | Michael Bruce Horowitz | Catheter based retrieval device with proximal body having axial freedom of movement |
| AU2021362245A1 (en) | 2017-10-16 | 2023-05-25 | Retriever Medical, Inc. | Clot removal methods and devices with multiple independently controllable elements |
| US11284902B2 (en) | 2018-02-01 | 2022-03-29 | Boston Scientific Scimed, Inc. | Method of making a vascular occlusion device |
| JP7034307B2 (ja) | 2018-02-01 | 2022-03-11 | ボストン サイエンティフィック サイムド,インコーポレイテッド | 医療デバイス解放システム |
| CA3107152A1 (en) * | 2019-04-18 | 2020-10-22 | Clearstream Technologies Limited | Embolization devices and methods of manufacturing the same |
| US11672946B2 (en) | 2019-09-24 | 2023-06-13 | Boston Scientific Scimed, Inc. | Protection and actuation mechanism for controlled release of implantable embolic devices |
| CN113017745A (zh) * | 2019-12-24 | 2021-06-25 | 上海微创心脉医疗科技(集团)股份有限公司 | 一种封堵器及系统 |
| AU2020448478B2 (en) | 2020-05-22 | 2023-02-16 | Clearstream Technologies Limited | Embolisation devices and methods of manufacturing the same |
| US20220313267A1 (en) * | 2021-03-31 | 2022-10-06 | DePuy Synthes Products, Inc. | Bio-absorbable coiled fiber |
| CN114081570B (zh) * | 2021-12-08 | 2024-03-08 | 杭州拓脉医疗科技有限公司 | 栓塞动脉瘤、血管的弹簧圈 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08336596A (ja) * | 1995-04-28 | 1996-12-24 | Target Therapeutics Inc | 熱固定されたポリマー繊維を有する血管閉塞デバイス |
| US5658308A (en) * | 1995-12-04 | 1997-08-19 | Target Therapeutics, Inc. | Bioactive occlusion coil |
| JP2000271131A (ja) * | 1996-04-10 | 2000-10-03 | Target Therapeutics Inc | 柔軟な末端の繊維を備えた微小脈管閉塞デバイス |
| US6193728B1 (en) * | 1995-06-30 | 2001-02-27 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils (II) |
Family Cites Families (74)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3174851A (en) | 1961-12-01 | 1965-03-23 | William J Buehler | Nickel-base alloys |
| US4238194A (en) * | 1966-10-04 | 1980-12-09 | General Electric Company | Nuclei analysis of solid substances |
| US3834394A (en) * | 1969-11-21 | 1974-09-10 | R Sessions | Occlusion device and method and apparatus for inserting the same |
| US3753700A (en) | 1970-07-02 | 1973-08-21 | Raychem Corp | Heat recoverable alloy |
| US4346712A (en) * | 1979-04-06 | 1982-08-31 | Kuraray Company, Ltd. | Releasable balloon catheter |
| US4545367A (en) * | 1982-07-16 | 1985-10-08 | Cordis Corporation | Detachable balloon catheter and method of use |
| CA1236641A (en) * | 1984-07-06 | 1988-05-10 | Motoaki Tanaka | Copolymer of lactic acid and glycolic acid and method for producing same |
| US4994069A (en) * | 1988-11-02 | 1991-02-19 | Target Therapeutics | Vaso-occlusion coil and method |
| JP2714454B2 (ja) * | 1988-11-07 | 1998-02-16 | 三井東圧化学株式会社 | 生体吸収性ポリエステルの製造方法 |
| US5354295A (en) * | 1990-03-13 | 1994-10-11 | Target Therapeutics, Inc. | In an endovascular electrolytically detachable wire and tip for the formation of thrombus in arteries, veins, aneurysms, vascular malformations and arteriovenous fistulas |
| US6083220A (en) * | 1990-03-13 | 2000-07-04 | The Regents Of The University Of California | Endovascular electrolytically detachable wire and tip for the formation of thrombus in arteries, veins, aneurysms, vascular malformations and arteriovenous fistulas |
| US5122136A (en) * | 1990-03-13 | 1992-06-16 | The Regents Of The University Of California | Endovascular electrolytically detachable guidewire tip for the electroformation of thrombus in arteries, veins, aneurysms, vascular malformations and arteriovenous fistulas |
| USRE41029E1 (en) * | 1990-03-13 | 2009-12-01 | The Regents Of The University Of California | Endovascular electrolytically detachable wire and tip for the formation of thrombus in arteries, veins, aneurysms, vascular malformations and arteriovenous fistulas |
| US5304194A (en) * | 1991-10-02 | 1994-04-19 | Target Therapeutics | Vasoocclusion coil with attached fibrous element(s) |
| US5226911A (en) | 1991-10-02 | 1993-07-13 | Target Therapeutics | Vasoocclusion coil with attached fibrous element(s) |
| US5256146A (en) * | 1991-10-11 | 1993-10-26 | W. D. Ensminger | Vascular catheterization system with catheter anchoring feature |
| US5443478A (en) * | 1992-09-02 | 1995-08-22 | Board Of Regents, The University Of Texas System | Multi-element intravascular occlusion device |
| US5382259A (en) * | 1992-10-26 | 1995-01-17 | Target Therapeutics, Inc. | Vasoocclusion coil with attached tubular woven or braided fibrous covering |
| US5382260A (en) * | 1992-10-30 | 1995-01-17 | Interventional Therapeutics Corp. | Embolization device and apparatus including an introducer cartridge and method for delivering the same |
| US5423849A (en) * | 1993-01-15 | 1995-06-13 | Target Therapeutics, Inc. | Vasoocclusion device containing radiopaque fibers |
| US6123007A (en) * | 1993-05-19 | 2000-09-26 | Metal Storm Limited | Barrel assembly |
| US5624449A (en) * | 1993-11-03 | 1997-04-29 | Target Therapeutics | Electrolytically severable joint for endovascular embolic devices |
| US5423829A (en) * | 1993-11-03 | 1995-06-13 | Target Therapeutics, Inc. | Electrolytically severable joint for endovascular embolic devices |
| US5549624A (en) * | 1994-06-24 | 1996-08-27 | Target Therapeutics, Inc. | Fibered vasooclusion coils |
| US6001092A (en) * | 1994-06-24 | 1999-12-14 | Target Therapeutics, Inc. | Complex coils having fibered centers |
| US5485496A (en) * | 1994-09-22 | 1996-01-16 | Cornell Research Foundation, Inc. | Gamma irradiation sterilizing of biomaterial medical devices or products, with improved degradation and mechanical properties |
| IL116561A0 (en) * | 1994-12-30 | 1996-03-31 | Target Therapeutics Inc | Severable joint for detachable devices placed within the body |
| US5911731A (en) * | 1995-04-20 | 1999-06-15 | Target Therapeutics, Inc. | Anatomically shaped vasoocclusive devices |
| US5645558A (en) * | 1995-04-20 | 1997-07-08 | Medical University Of South Carolina | Anatomically shaped vasoocclusive device and method of making the same |
| US5624461A (en) * | 1995-06-06 | 1997-04-29 | Target Therapeutics, Inc. | Three dimensional in-filling vaso-occlusive coils |
| US5853418A (en) * | 1995-06-30 | 1998-12-29 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils (II) |
| NO962761L (no) * | 1995-06-30 | 1997-01-02 | Target Therapeutics Inc | Strekkmotstandige vasookklusive spiraler |
| US5582619A (en) * | 1995-06-30 | 1996-12-10 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils |
| US5847963A (en) * | 1995-10-19 | 1998-12-08 | Gaiski; Stephen N. | Method for monitoring the effect of adjustments of paint application equipment |
| AU690862B2 (en) * | 1995-12-04 | 1998-04-30 | Target Therapeutics, Inc. | Fibered micro vaso-occlusive devices |
| US5797126A (en) * | 1996-02-16 | 1998-08-18 | Helbling; Edward | Automatic theater ticket concierge |
| DE19703482A1 (de) * | 1997-01-31 | 1998-08-06 | Ernst Peter Prof Dr M Strecker | Stent |
| US5891192A (en) * | 1997-05-22 | 1999-04-06 | The Regents Of The University Of California | Ion-implanted protein-coated intralumenal implants |
| US6766219B1 (en) * | 1997-05-29 | 2004-07-20 | Gary Marcel Hasey | Apparatus for controlling and monitoring of dispensing of oral medications |
| US5984929A (en) * | 1997-08-29 | 1999-11-16 | Target Therapeutics, Inc. | Fast detaching electronically isolated implant |
| US6146373A (en) * | 1997-10-17 | 2000-11-14 | Micro Therapeutics, Inc. | Catheter system and method for injection of a liquid embolic composition and a solidification agent |
| US6511468B1 (en) * | 1997-10-17 | 2003-01-28 | Micro Therapeutics, Inc. | Device and method for controlling injection of liquid embolic composition |
| AU2565099A (en) * | 1998-01-27 | 1999-09-20 | Regents Of The University Of California, The | Biodegradable polymer/protein based coils for intralumenal implants |
| US7070607B2 (en) * | 1998-01-27 | 2006-07-04 | The Regents Of The University Of California | Bioabsorbable polymeric implants and a method of using the same to create occlusions |
| US5935145A (en) * | 1998-02-13 | 1999-08-10 | Target Therapeutics, Inc. | Vaso-occlusive device with attached polymeric materials |
| US5941888A (en) * | 1998-02-18 | 1999-08-24 | Target Therapeutics, Inc. | Vaso-occlusive member assembly with multiple detaching points |
| US6013089A (en) * | 1998-04-17 | 2000-01-11 | Goldberg; Barry A. | Tongue cleaner |
| US20020087184A1 (en) * | 1998-06-18 | 2002-07-04 | Eder Joseph C. | Water-soluble coating for bioactive devices |
| US5980550A (en) * | 1998-06-18 | 1999-11-09 | Target Therapeutics, Inc. | Water-soluble coating for bioactive vasoocclusive devices |
| EP1148895B1 (en) * | 1999-02-05 | 2004-11-17 | The Regents of the University of California | Thermo-reversible polymer for intralumenal implant |
| US6221066B1 (en) * | 1999-03-09 | 2001-04-24 | Micrus Corporation | Shape memory segmented detachable coil |
| US6280457B1 (en) * | 1999-06-04 | 2001-08-28 | Scimed Life Systems, Inc. | Polymer covered vaso-occlusive devices and methods of producing such devices |
| DE10010840A1 (de) * | 1999-10-30 | 2001-09-20 | Dendron Gmbh | Vorrichtung zur Implantation von Occlusionswendeln |
| US8048104B2 (en) * | 2000-10-30 | 2011-11-01 | Dendron Gmbh | Device for the implantation of occlusion spirals |
| US6635069B1 (en) * | 2000-10-18 | 2003-10-21 | Scimed Life Systems, Inc. | Non-overlapping spherical three-dimensional coil |
| DE10118017B4 (de) * | 2001-04-10 | 2017-04-13 | Dendron Gmbh | Occlusionswendel und Vorrichtung zur Implantation von Occlusionswendeln |
| CA2459403A1 (en) * | 2001-08-27 | 2003-03-06 | Dendron Gmbh | Device for the implantation of occlusion means |
| EP1296467B1 (en) * | 2001-09-24 | 2004-02-25 | Evolium S.A.S. | Method for synchronizing terrestrial nodes equipped with GNSS receivers and belonging to a terrestrial network |
| DE10155191A1 (de) * | 2001-11-12 | 2003-05-22 | Dendron Gmbh | Medizinisches Implantat |
| US20040002732A1 (en) | 2002-06-27 | 2004-01-01 | Clifford Teoh | Stretch-resistant vaso-occlusive assembly with multiple detaching points |
| DE10233085B4 (de) * | 2002-07-19 | 2014-02-20 | Dendron Gmbh | Stent mit Führungsdraht |
| CA2513443A1 (en) * | 2003-02-26 | 2004-09-10 | Medivas, Llc | Bioactive stents and methods for use thereof |
| DE102004003265A1 (de) * | 2004-01-21 | 2005-08-11 | Dendron Gmbh | Vorrichtung zur Implantation von elektrisch isolierten Okklusionswendeln |
| US7323006B2 (en) * | 2004-03-30 | 2008-01-29 | Xtent, Inc. | Rapid exchange interventional devices and methods |
| ES2357243T3 (es) | 2004-05-21 | 2011-04-20 | Micro Therapeutics, Inc. | Espirales metálicas entrelazadas con polímeros o fibras biológicos o biodegradables o sintéticos para la embolización de una cavidad corporal. |
| EP1793744B1 (de) * | 2004-09-22 | 2008-12-17 | Dendron GmbH | Medizinisches implantat |
| DE502004010411D1 (de) * | 2004-09-22 | 2009-12-31 | Dendron Gmbh | Vorrichtung zur implantation von mikrowendeln |
| DE102005019782A1 (de) * | 2005-04-28 | 2006-11-09 | Dendron Gmbh | Vorrichtung zur Implantation von Okklusionswendeln mit innenliegendem Sicherungsmittel |
| EP2015683B1 (en) * | 2006-04-17 | 2015-12-09 | Covidien LP | System for mechanically positioning intravascular implants |
| US7689889B2 (en) * | 2006-08-24 | 2010-03-30 | Cisco Technology, Inc. | Content addressable memory entry coding for error detection and correction |
| JP5227344B2 (ja) * | 2007-03-13 | 2013-07-03 | タイコ ヘルスケア グループ リミテッド パートナーシップ | インプラント、マンドレル、およびインプラント形成方法 |
| JP5249249B2 (ja) * | 2007-03-13 | 2013-07-31 | コヴィディエン リミテッド パートナーシップ | コイルと耐伸張性部材とが含まれているインプラント |
| RU2011102994A (ru) * | 2008-07-22 | 2012-08-27 | Микро Терапьютикс, Инк. (Us) | Устройство для реконструкции сосудов |
| EP2444116B1 (en) * | 2008-08-19 | 2016-01-06 | Covidien LP | Detachable tip microcatheter |
-
2005
- 2005-05-23 ES ES05756033T patent/ES2357243T3/es active Active
- 2005-05-23 EP EP10194009.6A patent/EP2316355B1/en active Active
- 2005-05-23 DE DE602005025215T patent/DE602005025215D1/de active Active
- 2005-05-23 JP JP2007527550A patent/JP4723584B2/ja not_active Expired - Fee Related
- 2005-05-23 EP EP05756033A patent/EP1761178B1/en active Active
- 2005-05-23 US US11/134,998 patent/US7896899B2/en active Active
- 2005-05-23 AT AT05756033T patent/ATE490736T1/de not_active IP Right Cessation
- 2005-05-23 ES ES10194009.6T patent/ES2440650T3/es active Active
- 2005-05-23 WO PCT/US2005/018087 patent/WO2005113035A2/en active Application Filing
- 2005-05-23 CA CA2567331A patent/CA2567331C/en not_active Expired - Fee Related
-
2011
- 2011-01-20 US US13/010,671 patent/US8267955B2/en active Active
- 2011-02-23 JP JP2011037486A patent/JP5052682B2/ja active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08336596A (ja) * | 1995-04-28 | 1996-12-24 | Target Therapeutics Inc | 熱固定されたポリマー繊維を有する血管閉塞デバイス |
| US6193728B1 (en) * | 1995-06-30 | 2001-02-27 | Target Therapeutics, Inc. | Stretch resistant vaso-occlusive coils (II) |
| US5658308A (en) * | 1995-12-04 | 1997-08-19 | Target Therapeutics, Inc. | Bioactive occlusion coil |
| JP2000271131A (ja) * | 1996-04-10 | 2000-10-03 | Target Therapeutics Inc | 柔軟な末端の繊維を備えた微小脈管閉塞デバイス |
Also Published As
| Publication number | Publication date |
|---|---|
| ATE490736T1 (de) | 2010-12-15 |
| JP2008500148A (ja) | 2008-01-10 |
| EP1761178A2 (en) | 2007-03-14 |
| CA2567331A1 (en) | 2005-12-01 |
| JP2011136189A (ja) | 2011-07-14 |
| US8267955B2 (en) | 2012-09-18 |
| EP2316355A1 (en) | 2011-05-04 |
| WO2005113035A3 (en) | 2006-02-23 |
| DE602005025215D1 (de) | 2011-01-20 |
| JP5052682B2 (ja) | 2012-10-17 |
| US20060036281A1 (en) | 2006-02-16 |
| EP1761178B1 (en) | 2010-12-08 |
| WO2005113035A2 (en) | 2005-12-01 |
| US7896899B2 (en) | 2011-03-01 |
| ES2357243T3 (es) | 2011-04-20 |
| CA2567331C (en) | 2012-08-14 |
| EP2316355B1 (en) | 2013-09-18 |
| ES2440650T3 (es) | 2014-01-29 |
| US20110118777A1 (en) | 2011-05-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4723584B2 (ja) | 生物学的、生物分解性若しくは合成重合体又は繊維が巻き付けられた体腔を閉塞するための金属製コイル | |
| US11723667B2 (en) | Filamentary devices for treatment of vascular defects | |
| US20230270441A1 (en) | Devices for therapeutic vascular procedures | |
| US20230338035A1 (en) | Filamentary devices for treatment of vascular defects | |
| US10238393B2 (en) | Multiple layer filamentary devices for treatment of vascular defects | |
| US20120283768A1 (en) | Method and apparatus for the treatment of large and giant vascular defects | |
| US20050021072A1 (en) | Method and system for delivering an implant utilizing a lumen reducing member | |
| WO2002089865A2 (en) | Coated combination vaso-occlusive device | |
| EP2001370A1 (en) | Stretch-resistant vaso-occlusive devices with distal anchor link |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20080514 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20080514 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20101206 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20110223 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20110318 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20110407 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20140415 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4723584 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20140415 Year of fee payment: 3 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313113 Free format text: JAPANESE INTERMEDIATE CODE: R313111 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20140415 Year of fee payment: 3 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |