JP2021522071A - 半透過性装置 - Google Patents
半透過性装置 Download PDFInfo
- Publication number
- JP2021522071A JP2021522071A JP2021510546A JP2021510546A JP2021522071A JP 2021522071 A JP2021522071 A JP 2021522071A JP 2021510546 A JP2021510546 A JP 2021510546A JP 2021510546 A JP2021510546 A JP 2021510546A JP 2021522071 A JP2021522071 A JP 2021522071A
- Authority
- JP
- Japan
- Prior art keywords
- semi
- structural
- self
- ideally
- porous member
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000004140 cleaning Methods 0.000 claims abstract description 215
- 239000012530 fluid Substances 0.000 claims abstract description 86
- 239000000126 substance Substances 0.000 claims abstract description 71
- 230000001050 lubricating effect Effects 0.000 claims abstract description 69
- 239000000463 material Substances 0.000 claims abstract description 55
- TXEYQDLBPFQVAA-UHFFFAOYSA-N tetrafluoromethane Chemical compound FC(F)(F)F TXEYQDLBPFQVAA-UHFFFAOYSA-N 0.000 claims abstract description 50
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 34
- 229920000295 expanded polytetrafluoroethylene Polymers 0.000 claims abstract description 26
- 230000004888 barrier function Effects 0.000 claims abstract description 20
- 230000007613 environmental effect Effects 0.000 claims abstract description 15
- 230000002934 lysing effect Effects 0.000 claims abstract description 8
- 238000001914 filtration Methods 0.000 claims description 153
- 239000007788 liquid Substances 0.000 claims description 50
- 239000002245 particle Substances 0.000 claims description 41
- 229920001343 polytetrafluoroethylene Polymers 0.000 claims description 38
- 239000004810 polytetrafluoroethylene Substances 0.000 claims description 38
- 239000011148 porous material Substances 0.000 claims description 30
- 229920000642 polymer Polymers 0.000 claims description 24
- 239000004812 Fluorinated ethylene propylene Substances 0.000 claims description 22
- 229920009441 perflouroethylene propylene Polymers 0.000 claims description 22
- 230000000844 anti-bacterial effect Effects 0.000 claims description 17
- 239000004814 polyurethane Substances 0.000 claims description 17
- 229920002635 polyurethane Polymers 0.000 claims description 14
- 239000000654 additive Substances 0.000 claims description 12
- 238000002844 melting Methods 0.000 claims description 12
- 230000008018 melting Effects 0.000 claims description 11
- 230000000996 additive effect Effects 0.000 claims description 8
- 238000003825 pressing Methods 0.000 claims description 8
- 230000000472 traumatic effect Effects 0.000 claims description 5
- 238000002513 implantation Methods 0.000 claims description 4
- 239000003795 chemical substances by application Substances 0.000 claims 1
- 238000010586 diagram Methods 0.000 abstract 1
- 238000000034 method Methods 0.000 description 168
- 239000010410 layer Substances 0.000 description 83
- 239000000835 fiber Substances 0.000 description 60
- 238000007789 sealing Methods 0.000 description 50
- 238000004519 manufacturing process Methods 0.000 description 49
- AGOYDEPGAOXOCK-KCBOHYOISA-N clarithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@](C)([C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)OC)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 AGOYDEPGAOXOCK-KCBOHYOISA-N 0.000 description 27
- 238000001523 electrospinning Methods 0.000 description 23
- 239000012528 membrane Substances 0.000 description 23
- 239000000314 lubricant Substances 0.000 description 19
- 239000003570 air Substances 0.000 description 15
- 230000008859 change Effects 0.000 description 12
- 239000003814 drug Substances 0.000 description 12
- 229940079593 drug Drugs 0.000 description 12
- 244000052769 pathogen Species 0.000 description 12
- 210000001519 tissue Anatomy 0.000 description 10
- 230000009471 action Effects 0.000 description 9
- 230000008569 process Effects 0.000 description 7
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 6
- 238000000576 coating method Methods 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 244000005700 microbiome Species 0.000 description 6
- 238000005245 sintering Methods 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000002209 hydrophobic effect Effects 0.000 description 5
- 238000005304 joining Methods 0.000 description 5
- -1 polytetrafluoroethylene Polymers 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 230000003014 reinforcing effect Effects 0.000 description 5
- 241000894006 Bacteria Species 0.000 description 4
- 230000012010 growth Effects 0.000 description 4
- 208000015181 infectious disease Diseases 0.000 description 4
- 239000002356 single layer Substances 0.000 description 4
- 229920001169 thermoplastic Polymers 0.000 description 4
- 239000004743 Polypropylene Substances 0.000 description 3
- 241000191967 Staphylococcus aureus Species 0.000 description 3
- 208000027418 Wounds and injury Diseases 0.000 description 3
- 239000003242 anti bacterial agent Substances 0.000 description 3
- 239000000560 biocompatible material Substances 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 230000002349 favourable effect Effects 0.000 description 3
- 229920002313 fluoropolymer Polymers 0.000 description 3
- 239000004811 fluoropolymer Substances 0.000 description 3
- 230000000813 microbial effect Effects 0.000 description 3
- 229950011087 perflunafene Drugs 0.000 description 3
- UWEYRJFJVCLAGH-IJWZVTFUSA-N perfluorodecalin Chemical compound FC1(F)C(F)(F)C(F)(F)C(F)(F)[C@@]2(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)[C@@]21F UWEYRJFJVCLAGH-IJWZVTFUSA-N 0.000 description 3
- QKENRHXGDUPTEM-UHFFFAOYSA-N perfluorophenanthrene Chemical compound FC1(F)C(F)(F)C(F)(F)C(F)(F)C2(F)C3(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C3(F)C(F)(F)C(F)(F)C21F QKENRHXGDUPTEM-UHFFFAOYSA-N 0.000 description 3
- 230000035699 permeability Effects 0.000 description 3
- 229920001155 polypropylene Polymers 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 238000005452 bending Methods 0.000 description 2
- 230000032770 biofilm formation Effects 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 238000012937 correction Methods 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 238000005538 encapsulation Methods 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 230000001976 improved effect Effects 0.000 description 2
- 230000001788 irregular Effects 0.000 description 2
- 238000002955 isolation Methods 0.000 description 2
- 239000010702 perfluoropolyether Substances 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 229920005594 polymer fiber Polymers 0.000 description 2
- 238000004080 punching Methods 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 238000009738 saturating Methods 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 238000009941 weaving Methods 0.000 description 2
- 208000037193 Device lead damage Diseases 0.000 description 1
- 229920001410 Microfiber Polymers 0.000 description 1
- 206010038848 Retinal detachment Diseases 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000003633 blood substitute Substances 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 239000011538 cleaning material Substances 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 230000001066 destructive effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000005553 drilling Methods 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- HQQADJVZYDDRJT-UHFFFAOYSA-N ethene;prop-1-ene Chemical group C=C.CC=C HQQADJVZYDDRJT-UHFFFAOYSA-N 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 230000001815 facial effect Effects 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 239000003658 microfiber Substances 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 206010033675 panniculitis Diseases 0.000 description 1
- 239000002831 pharmacologic agent Substances 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 230000001846 repelling effect Effects 0.000 description 1
- 230000029058 respiratory gaseous exchange Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000004264 retinal detachment Effects 0.000 description 1
- 239000002195 soluble material Substances 0.000 description 1
- 210000000278 spinal cord Anatomy 0.000 description 1
- 239000003351 stiffener Substances 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 210000004304 subcutaneous tissue Anatomy 0.000 description 1
- 230000008093 supporting effect Effects 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 230000008467 tissue growth Effects 0.000 description 1
- 238000002834 transmittance Methods 0.000 description 1
- 230000001515 vagal effect Effects 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D65/00—Accessories or auxiliary operations, in general, for separation processes or apparatus using semi-permeable membranes
- B01D65/08—Prevention of membrane fouling or of concentration polarisation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/02—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor characterised by their properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/10—Supported membranes; Membrane supports
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/10—Supported membranes; Membrane supports
- B01D69/106—Membranes in the pores of a support, e.g. polymerized in the pores or voids
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/10—Supported membranes; Membrane supports
- B01D69/107—Organic support material
- B01D69/1071—Woven, non-woven or net mesh
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/12—Composite membranes; Ultra-thin membranes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/12—Composite membranes; Ultra-thin membranes
- B01D69/1214—Chemically bonded layers, e.g. cross-linking
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/14—Dynamic membranes
- B01D69/141—Heterogeneous membranes, e.g. containing dispersed material; Mixed matrix membranes
- B01D69/1411—Heterogeneous membranes, e.g. containing dispersed material; Mixed matrix membranes containing dispersed material in a continuous matrix
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/30—Polyalkenyl halides
- B01D71/32—Polyalkenyl halides containing fluorine atoms
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/30—Polyalkenyl halides
- B01D71/32—Polyalkenyl halides containing fluorine atoms
- B01D71/36—Polytetrafluoroethene
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/54—Polyureas; Polyurethanes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2002/018—Filters implantable into blood vessels made from tubes or sheets of material, e.g. by etching or laser-cutting
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2321/00—Details relating to membrane cleaning, regeneration, sterilization or to the prevention of fouling
- B01D2321/281—Details relating to membrane cleaning, regeneration, sterilization or to the prevention of fouling by applying a special coating to the membrane or to any module element
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2325/00—Details relating to properties of membranes
- B01D2325/08—Patterned membranes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2325/00—Details relating to properties of membranes
- B01D2325/28—Degradation or stability over time
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2325/00—Details relating to properties of membranes
- B01D2325/48—Antimicrobial properties
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dispersion Chemistry (AREA)
- Filtering Materials (AREA)
- Separation Using Semi-Permeable Membranes (AREA)
- Materials For Medical Uses (AREA)
- External Artificial Organs (AREA)
Abstract
【選択図】図32
Description
Claims (39)
- 臨床、農業、工業及び/又は環境の現場において使用される、構造手段を備える半透過性装置であって、
前記構造手段は、潤滑流体に親和性を有する材料から少なくとも部分的に形成され、潤滑流体を注入できるように適合され、
前記半透過性装置は、前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する手段と共に構成され、
前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段は、前記構造手段を通じた空気、水及び溶解物質等の流体の移動を可能にする1つ又は複数の通路を有する状態で構成される、
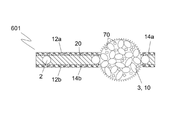
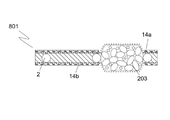
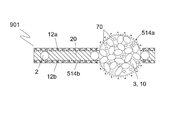
半透過性装置。 - 前記構造手段は、多孔性であることを特徴とする、請求項1に記載の半透過性装置。
- 前記構造手段は、複数の小繊維を備え、前記小繊維間の間隔は、1.0μm以下であることを特徴とする、請求項2に記載の半透過性装置。
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段は、前記構造手段の細孔内に位置する障壁を含むことを特徴とする、請求項2又は3に記載の半透過性装置。
- 前記障壁は、PU又はFEP等のポリマーを前記構造手段の細孔内に押圧及び/又は熱溶融することによって設けられることを特徴とする、請求項4に記載の半透過性装置。
- 前記構造手段は、1つ又は複数の層から形成されることを特徴とする、請求項1から5のいずれか一項に記載の半透過性装置。
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段は、前記構造手段内に保持されることを特徴とする、請求項1から6のいずれか一項に記載の半透過性装置。
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段は、前記構造手段の主平面から少なくとも1つの方向で延出することを特徴とする、請求項7に記載の半透過性装置。
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段は、前記構造手段の第1の層と第2の層との間に配設されることを特徴とする、請求項6に従属する請求項7又は8に記載の半透過性装置。
- 前記1つ又は複数の層は、ePTFEから形成されることを特徴とする、請求項6に記載の半透過性装置。
- 前記構造手段は、ePTFEの第1の層と第2の層とを備えることを特徴とする、請求項10に記載の半透過性装置。
- 前記第1の層は、前記第1の層内の小繊維の長尺方向が前記第2の層内の小繊維の長尺方向と位置がずれるように、前記第2の層に対して配置されることを特徴とする、請求項11に記載の半透過性装置。
- 前記第1の層は、前記第1の層内の小繊維の長尺方向が前記第2の層内の小繊維の長尺方向に直交して延在するように、前記第2の層に対して配置されることを特徴とする、請求項12に記載の半透過性装置。
- 前記ePTFE層は、一緒に結合されることを特徴とする、請求項10から12のいずれか一項に記載の半透過性装置。
- 前記ePTFE層は、前記層の間に延在するFEP等のポリマーを結合することによって一緒に結合されることを特徴とする、請求項14に記載の半透過性装置。
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段は、少なくとも1つの多孔性部材を備えることを特徴とする、請求項1から15のいずれか一項に記載の半透過性装置。
- 前記多孔性部材は、一緒に接着及び/又は焼結した複数の粒子を含むことを特徴とする、請求項16に記載の半透過性装置。
- 前記多孔性部材は、形状が球形、ほぼ球形若しくは偏平の球体、球形キャップ、半球体、卵形体、立方体又は直方体であることを特徴とする、請求項16又は17に記載の半透過性装置。
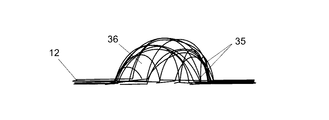
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段は、少なくとも1つの実質的に中空の部材を備え、前記実質的に中空の部材は、前記構造手段の一方の側に配置可能な第1の開口と、前記構造手段のもう一方の側に配置可能な第2の開口とを有することを特徴とする、請求項1から18のいずれか一項に記載の半透過性装置。
- 前記実質的に中空の部材は、管であることを特徴とする、請求項19に記載の半透過性装置。
- 前記実質的に中空の部材は、1つ又は複数のポリマー物質、例えば、ポリウレタン(PU)、フッ素化エチレンプロピレン(FEP)及び/又はPTFEから少なくとも部分的に形成されることを特徴とする、請求項19又は20に記載の半透過性装置。
- 前記実質的に中空の部材は、硬い最内部分とあまり硬くない最外部分とを有する状態で形成されることを特徴とする、請求項21に記載の半透過性装置。
- 前記実質的に中空の部材は、比較的硬いポリマー物質から少なくとも部分的に形成され、前記実質的に中空の部材は、FEP若しくはPU等の可撓性物質又は比較的低い溶融点を有する物質から形成される最外部分を備えることを特徴とする、請求項22に記載の半透過性装置。
- 前記実質的に中空の部材の前記最外部分は、前記構造手段内に延在するように配置され、これにより、前記実質的に中空の部材を前記構造手段内に保持し、前記構造手段を通じた前記潤滑流体の移動に対する障壁をもたらすことを特徴とする、請求項23に記載の半透過性装置。
- 前記実質的に中空の部材は、前記構造手段の前記第1の層と前記第2の層との間に配設されるように適合した突縁を有することを特徴とする、請求項6に従属する請求項19から24のいずれか一項に記載の半透過性装置。
- 前記構造手段は、前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段の通路の開口にわたり延在することを特徴とする、請求項1から25のいずれか一項に記載の半透過性装置。
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段の部分は、少なくとも部分的に親水性であることを特徴とする、請求項26に記載の半透過性装置。
- 潤滑流体を含む、請求項1から27のいずれか一項に記載の半透過性装置。
- パーフルオロカーボン液体を含む、請求項28に記載の半透過性装置。
- 前記構造手段の少なくとも一部を通じた前記潤滑流体の移動を防止又は制限する前記手段の前記通路は、添加剤を含むことを特徴とする、請求項1から29のいずれか一項に記載の半透過性装置。
- 前記添加剤は、可溶性抗菌性物質又は薬剤を含むことを特徴とする、請求項30に記載の半透過性装置。
- 前記添加剤は、水及び/又は空気浄化物質を含むことを特徴とする、請求項30に記載の半透過性装置。
- 前記構造手段は、構造枠を備えることを特徴とする、請求項1から32のいずれか一項に記載の半透過性装置。
- ろ過手段を備える、請求項1から33のいずれか一項に記載の半透過性装置。
- 前記ろ過手段は、ろ過ウェブを備えることを特徴とする、請求項34に記載の半透過性装置。
- 前記ろ過ウェブは、1.0μm以下の間隔を有することを特徴とする、請求項35に記載の半透過性装置。
- 自己洗浄手段を備える、請求項1から36のいずれか一項に記載の半透過性装置。
- 自己洗浄ウェブを備える、請求項37に記載の半透過性装置。
- レシピエントへの植え込み、MAVIEDでの使用、外傷用包帯としての使用、顔マスクとしての使用、水ろ過器としての使用、若しくは空気ろ過器としての使用に適応可能であるか又は適合される、請求項1から38のいずれか一項に記載の半透過性装置。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB1807322.1 | 2018-05-03 | ||
| GBGB1807322.1A GB201807322D0 (en) | 2018-05-03 | 2018-05-03 | A semipermeable arrangement and method of manufacture |
| GB1812846.2 | 2018-08-07 | ||
| GBGB1812846.2A GB201812846D0 (en) | 2018-05-03 | 2018-08-07 | A semipermeable arrangement and method of manufacture |
| PCT/EP2019/061465 WO2019211479A2 (en) | 2018-05-03 | 2019-05-03 | A semipermeable arrangement |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2021522071A true JP2021522071A (ja) | 2021-08-30 |
| JPWO2019211479A5 JPWO2019211479A5 (ja) | 2022-05-09 |
Family
ID=62598141
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2021510546A Pending JP2021522071A (ja) | 2018-05-03 | 2019-05-03 | 半透過性装置 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20210236995A1 (ja) |
| EP (1) | EP3796992A2 (ja) |
| JP (1) | JP2021522071A (ja) |
| CN (1) | CN112384293A (ja) |
| CA (1) | CA3120219A1 (ja) |
| GB (2) | GB201807322D0 (ja) |
| WO (1) | WO2019211479A2 (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB202106255D0 (en) * | 2020-04-30 | 2021-06-16 | Lau Ernest Wai Yin | An article of personal protective equipment and a pump |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04122412A (ja) * | 1990-09-12 | 1992-04-22 | Hitachi Ltd | 酸素の溶解方法及び酸素溶解装置 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS641216U (ja) * | 1987-06-16 | 1989-01-06 | ||
| JPH0433859Y2 (ja) * | 1987-06-16 | 1992-08-13 | ||
| DE19544912A1 (de) * | 1995-12-01 | 1997-06-05 | Gore W L & Ass Gmbh | PTFE-Körper aus mikroporösem Polytetrafluorethylen mit Füllstoff und Verfahren zu dessen Herstellung |
| MXPA02006641A (es) * | 2000-01-03 | 2004-09-10 | Biomed Sciences Inc | Aposito novedoso para heridas, proceso de fabricacion y articulos utiles del mismo. |
| JP3851864B2 (ja) * | 2002-10-23 | 2006-11-29 | 住友電工ファインポリマー株式会社 | 多孔質複層中空糸および該多孔質複層中空糸を備えた濾過モジュール |
| EP2155370A1 (en) * | 2007-04-23 | 2010-02-24 | W.L. GORE & ASSOCIATES GmbH | Composite material |
| US8193406B2 (en) * | 2007-05-17 | 2012-06-05 | Ut-Battelle, Llc | Super-hydrophobic bandages and method of making the same |
| US20110052900A1 (en) * | 2009-02-16 | 2011-03-03 | Sumitomo Electric Fine Polymer, Inc. | Porous multilayer filter and method for producing same |
| JP6388541B2 (ja) * | 2012-01-10 | 2018-09-12 | プレジデント アンド フェローズ オブ ハーバード カレッジ | 流体および固体反発性のための表面の改質 |
| US9522072B2 (en) * | 2013-03-15 | 2016-12-20 | W. L. Gore & Associates, Inc. | Porous materials having a fibrillar microstructure and a fracturable coating |
| JP6862642B2 (ja) * | 2015-10-15 | 2021-04-21 | 住友電工ファインポリマー株式会社 | 半透膜及び半透膜の製造方法 |
-
2018
- 2018-05-03 GB GBGB1807322.1A patent/GB201807322D0/en not_active Ceased
- 2018-08-07 GB GBGB1812846.2A patent/GB201812846D0/en not_active Ceased
-
2019
- 2019-05-03 EP EP19734690.1A patent/EP3796992A2/en not_active Withdrawn
- 2019-05-03 WO PCT/EP2019/061465 patent/WO2019211479A2/en unknown
- 2019-05-03 US US17/052,629 patent/US20210236995A1/en not_active Abandoned
- 2019-05-03 JP JP2021510546A patent/JP2021522071A/ja active Pending
- 2019-05-03 CN CN201980045157.7A patent/CN112384293A/zh active Pending
- 2019-05-03 CA CA3120219A patent/CA3120219A1/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04122412A (ja) * | 1990-09-12 | 1992-04-22 | Hitachi Ltd | 酸素の溶解方法及び酸素溶解装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20210236995A1 (en) | 2021-08-05 |
| CN112384293A (zh) | 2021-02-19 |
| GB201807322D0 (en) | 2018-06-20 |
| GB201812846D0 (en) | 2018-09-19 |
| WO2019211479A2 (en) | 2019-11-07 |
| WO2019211479A3 (en) | 2020-01-09 |
| CA3120219A1 (en) | 2019-11-07 |
| EP3796992A2 (en) | 2021-03-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5566911B2 (ja) | 創傷治療用の繊維微小球の生体吸収性複合体足場 | |
| EP2897661B1 (en) | Drug-eluting rotational spun coatings and methods of use | |
| KR102178233B1 (ko) | 공간적으로 배열된 섬유를 갖는 생의학 패치 | |
| CN103442741B (zh) | 具有快速治疗剂可吸收的组织修复装置 | |
| KR101813300B1 (ko) | 미세다공성 표면 층을 가지는 이식형 의료 장치 및 이에 대한 이물 반응을 감소시키는 방법 | |
| EP1570806B1 (de) | Flächiges Implantat, Verfahren zu seiner Herstellung und Verwendung in der Chirurgie | |
| EP2642960B1 (de) | Wundauflage für die unterdrucktherapie | |
| US5686090A (en) | Multi-layered implant | |
| JP2019048104A (ja) | 皮膚移植片採取用の吸収性基材 | |
| CN103228229B (zh) | 具有支持和径向扩散支架的可植入细胞装置 | |
| EP2002853A2 (en) | Biocompatible implant system and method | |
| KR101150964B1 (ko) | 항균 드레싱 적층체 및 그의 제조방법 | |
| EP3206725B1 (en) | "device for wound dressing" | |
| WO2006032497A1 (de) | Antimikrobielles implantat mit einer flexiblen porösen struktur | |
| JP2009542353A (ja) | 改良された接触表面を有する成長刺激創傷用ドレッシング | |
| DE102008037888A1 (de) | Wundpflegeartikel, aufweisend Textilbänder mit Fasern mit gelbildenden Eigenschaften sowie Fasern mit nicht gelbildenden Eigenschaften | |
| JP2023174811A (ja) | 薬物療法デリバリーシステム及び方法 | |
| RU2578458C2 (ru) | Медицинская многослойная повязка с многофункциональными наномембранами и изделия на ее основе | |
| DE102012025125A1 (de) | Saugkörper zur therapeutischen Behandlung einer Wunde mittels Unterdruck | |
| JP2021522071A (ja) | 半透過性装置 | |
| DE102012025124A1 (de) | Saugkörper zur therapeutischen Behandlung einer Wunde mittels Unterdruck | |
| DE102004062798A1 (de) | Flächiges Implantat und Verfahren zu seiner Herstellung | |
| WO2020176923A1 (en) | An attachment means for attaching a medical device to tissue, a system for attaching a medical device to tissue, a medical device having an attachment means, a method of attaching a medical device to tissue, and a method of manufacturing an attachment means. | |
| KR101168121B1 (ko) | 압출, 전기방사 및 슬러리 공정을 이용한 인공골의 제조방법 | |
| JP2019076783A (ja) | 空間的に配置された繊維を有する医用パッチ |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220425 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20220426 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220427 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20230130 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20230131 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20230419 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20231003 |