JP2020518404A - 補強されたカテーテルを用いた血液ポンプ - Google Patents
補強されたカテーテルを用いた血液ポンプ Download PDFInfo
- Publication number
- JP2020518404A JP2020518404A JP2019560767A JP2019560767A JP2020518404A JP 2020518404 A JP2020518404 A JP 2020518404A JP 2019560767 A JP2019560767 A JP 2019560767A JP 2019560767 A JP2019560767 A JP 2019560767A JP 2020518404 A JP2020518404 A JP 2020518404A
- Authority
- JP
- Japan
- Prior art keywords
- catheter
- blood pump
- stiffening structure
- lumen
- proximal end
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000008280 blood Substances 0.000 title claims abstract description 59
- 210000004369 blood Anatomy 0.000 title claims abstract description 59
- 239000012530 fluid Substances 0.000 claims abstract description 32
- 238000005086 pumping Methods 0.000 claims abstract description 18
- 238000003780 insertion Methods 0.000 claims description 17
- 230000037431 insertion Effects 0.000 claims description 17
- 210000004204 blood vessel Anatomy 0.000 claims description 15
- 239000000463 material Substances 0.000 claims description 9
- 239000012781 shape memory material Substances 0.000 claims description 8
- 238000005452 bending Methods 0.000 claims description 6
- 229910001000 nickel titanium Inorganic materials 0.000 claims description 4
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 claims description 4
- 229910001285 shape-memory alloy Inorganic materials 0.000 claims description 3
- 238000010926 purge Methods 0.000 description 12
- 210000000709 aorta Anatomy 0.000 description 10
- 230000017531 blood circulation Effects 0.000 description 6
- 210000005240 left ventricle Anatomy 0.000 description 6
- 210000002376 aorta thoracic Anatomy 0.000 description 5
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 230000005489 elastic deformation Effects 0.000 description 2
- 210000001105 femoral artery Anatomy 0.000 description 2
- 238000007667 floating Methods 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 229910000734 martensite Inorganic materials 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 229920002545 silicone oil Polymers 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 210000004191 axillary artery Anatomy 0.000 description 1
- 230000006399 behavior Effects 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000000560 biocompatible material Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000004891 communication Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- 210000004731 jugular vein Anatomy 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 210000005241 right ventricle Anatomy 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 210000004872 soft tissue Anatomy 0.000 description 1
- 210000003270 subclavian artery Anatomy 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/0102—Insertion or introduction using an inner stiffening member, e.g. stylet or push-rod
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/80—Constructional details other than related to driving
- A61M60/855—Constructional details other than related to driving of implantable pumps or pumping devices
- A61M60/857—Implantable blood tubes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L29/00—Materials for catheters, medical tubing, cannulae, or endoscopes or for coating catheters
- A61L29/02—Inorganic materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L29/00—Materials for catheters, medical tubing, cannulae, or endoscopes or for coating catheters
- A61L29/04—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/10—Location thereof with respect to the patient's body
- A61M60/122—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body
- A61M60/126—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable via, into, inside, in line, branching on, or around a blood vessel
- A61M60/13—Implantable pumps or pumping devices, i.e. the blood being pumped inside the patient's body implantable via, into, inside, in line, branching on, or around a blood vessel by means of a catheter allowing explantation, e.g. catheter pumps temporarily introduced via the vascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/20—Type thereof
- A61M60/205—Non-positive displacement blood pumps
- A61M60/216—Non-positive displacement blood pumps including a rotating member acting on the blood, e.g. impeller
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M60/00—Blood pumps; Devices for mechanical circulatory actuation; Balloon pumps for circulatory assistance
- A61M60/80—Constructional details other than related to driving
- A61M60/802—Constructional details other than related to driving of non-positive displacement blood pumps
- A61M60/827—Sealings between moving parts
- A61M60/829—Sealings between moving parts having a purge fluid supply
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/02—General characteristics of the apparatus characterised by a particular materials
- A61M2205/0266—Shape memory materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2210/00—Anatomical parts of the body
- A61M2210/12—Blood circulatory system
- A61M2210/125—Heart
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0043—Catheters; Hollow probes characterised by structural features
- A61M25/005—Catheters; Hollow probes characterised by structural features with embedded materials for reinforcement, e.g. wires, coils, braids
Landscapes
- Health & Medical Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Anesthesiology (AREA)
- Hematology (AREA)
- Biomedical Technology (AREA)
- Cardiology (AREA)
- Mechanical Engineering (AREA)
- Vascular Medicine (AREA)
- Epidemiology (AREA)
- Pulmonology (AREA)
- Biophysics (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- External Artificial Organs (AREA)
- Transplantation (AREA)
- Prostheses (AREA)
Abstract
Description
Claims (13)
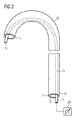
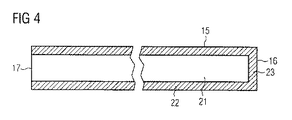
- 患者の血管内に経皮挿入するための血管内血液ポンプであって、カテーテル(10)と、前記カテーテル(10)に取り付けられたポンピングデバイス(1)とを備え、前記カテーテル(10)は、長手方向軸に沿って延び、前記長手方向軸に沿って、遠位端(11)と、前記遠位端(11)の反対側の近位端(12)とを有し、前記カテーテル(10)は、或る長さを有し、前記カテーテルの前記近位端(12)および前記遠位端(11)間に長手方向に延びる、管状補剛構造(15)を含み、前記管状補剛構造(15)は、管腔(21)と、近位端(17)と、前記近位端(17)の反対側の閉じた遠位端(16)とを有し、前記補剛構造(15)の前記管腔(21)は、少なくとも5バールの過剰圧力を有する加圧流体を受けるように構成されることを特徴とする血管内血液ポンプ。
- 請求項1に記載の血液ポンプであって、前記カテーテル(10)は、前記近位端(12)から前記遠位端(11)まで前記カテーテル(10)を通って延びる管腔(13)を有し、前記補剛構造(15)は前記カテーテル(10)の前記管腔(13)内に配設されることを特徴とする血液ポンプ。
- 請求項2に記載の血液ポンプであって、前記補剛構造は、前記カテーテル(10)の前記管腔(13)内部で緩んでいることを特徴とする血液ポンプ。
- 請求項1〜3のいずれか1項に記載の血液ポンプであって、前記補剛構造(15)の前記管腔(21)は、5バール〜150バールの過剰圧力を有する加圧流体を受けるように構成されることを特徴とする血液ポンプ。
- 請求項1〜4のいずれか1項に記載の血液ポンプであって、前記補剛構造(15)は、形状記憶材料、好ましくは形状記憶合金、より好ましくはニチノールを含むことを特徴とする血液ポンプ。
- 請求項1〜5のいずれか1項に記載の血液ポンプであって、前記補剛構造(15)はポリマー材料を含むことを特徴とする血液ポンプ。
- 請求項1〜6のいずれか1項に記載の血液ポンプであって、前記管状補剛構造(15)のうちの少なくとも2つを含み、前記少なくとも2つの管状補剛構造(15)は撚り合わされていることを特徴とする血液ポンプ。
- 請求項1〜6のいずれか1項に記載の血液ポンプであって、前記管状補剛構造(15)のうちの少なくとも3つを含み、前記少なくとも3つの管状補剛構造(15)は、中実の編み紐または編組された中空の管状部材を形成するように編組されていることを特徴とする血液ポンプ。
- 請求項1〜8のいずれか1項に記載の血液ポンプであって、前記補剛構造(15)は、前記血液ポンプの動作中、前記カテーテル(10)内に留まるように構成されることを特徴とする血液ポンプ。
- 請求項1〜8のいずれか1項に記載の血液ポンプであって、前記補剛構造(15)は、前記血液ポンプを患者の身体内に配置した後、前記カテーテル(10)から除去されるように構成されることを特徴とする血液ポンプ。
- 請求項1〜10のいずれか1項に記載の血液ポンプであって、前記補剛構造(15)は、前記カテーテル(10)が、塑性変形を生じることなく、10mm以下の曲げで弾性変形されることを可能にすることを特徴とする血液ポンプ。
- 請求項1〜11のいずれか1項に記載の血液ポンプと、前記補剛構造(15)の前記近位端(17)に接続された圧力源(20)とを備え、加圧流体を前記補剛構造(15)の前記管腔(21)に供給するように構成されることを特徴とするシステム。
- 請求項12に記載のシステムであって、前記加圧流体の圧力を調整するように構成されたコントローラを更に備えることを特徴とするシステム。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023049130A JP2023068164A (ja) | 2017-05-04 | 2023-03-27 | 補強されたカテーテルを用いた血液ポンプ |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP17169486.2 | 2017-05-04 | ||
| EP17169486.2A EP3398624A1 (en) | 2017-05-04 | 2017-05-04 | Blood pump with reinforced catheter |
| PCT/EP2018/061349 WO2018202775A1 (en) | 2017-05-04 | 2018-05-03 | Blood pump with reinforced catheter |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2023049130A Division JP2023068164A (ja) | 2017-05-04 | 2023-03-27 | 補強されたカテーテルを用いた血液ポンプ |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2020518404A true JP2020518404A (ja) | 2020-06-25 |
| JP2020518404A5 JP2020518404A5 (ja) | 2021-06-17 |
Family
ID=58669727
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2019560767A Pending JP2020518404A (ja) | 2017-05-04 | 2018-05-03 | 補強されたカテーテルを用いた血液ポンプ |
| JP2023049130A Pending JP2023068164A (ja) | 2017-05-04 | 2023-03-27 | 補強されたカテーテルを用いた血液ポンプ |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2023049130A Pending JP2023068164A (ja) | 2017-05-04 | 2023-03-27 | 補強されたカテーテルを用いた血液ポンプ |
Country Status (10)
| Country | Link |
|---|---|
| US (2) | US11744987B2 (ja) |
| EP (3) | EP3398624A1 (ja) |
| JP (2) | JP2020518404A (ja) |
| KR (2) | KR102605513B1 (ja) |
| CN (1) | CN110603064A (ja) |
| AU (2) | AU2018262629B2 (ja) |
| DK (1) | DK3618884T3 (ja) |
| ES (1) | ES2902688T3 (ja) |
| SG (1) | SG11201909507RA (ja) |
| WO (1) | WO2018202775A1 (ja) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108742951B (zh) | 2012-06-06 | 2021-05-25 | 洋红医疗有限公司 | 人工肾脏瓣膜 |
| US10583231B2 (en) | 2013-03-13 | 2020-03-10 | Magenta Medical Ltd. | Blood pump |
| CN105473063B (zh) | 2013-03-13 | 2019-03-08 | 马真塔医药有限公司 | 血液泵浦及其制造方法 |
| US11291824B2 (en) | 2015-05-18 | 2022-04-05 | Magenta Medical Ltd. | Blood pump |
| EP3532120B1 (en) | 2016-10-25 | 2024-05-01 | Magenta Medical Ltd. | Ventricular assist device |
| JP7094279B2 (ja) | 2016-11-23 | 2022-07-01 | マジェンタ・メディカル・リミテッド | 血液ポンプ |
| EP3693038B1 (en) | 2018-01-10 | 2024-06-05 | Magenta Medical Ltd. | Ventricular assist device |
| US10905808B2 (en) | 2018-01-10 | 2021-02-02 | Magenta Medical Ltd. | Drive cable for use with a blood pump |
| EP3782665B1 (en) | 2019-01-24 | 2021-08-25 | Magenta Medical Ltd. | Ventricular assist device |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009506839A (ja) * | 2005-09-02 | 2009-02-19 | ボストン サイエンティフィック リミテッド | 調整可能な剛性を有するカテーテル |
| JP2010536522A (ja) * | 2007-08-29 | 2010-12-02 | アクロシュターク コーポレイション | 被験者に小線源療法を施す(delivery)方法及びキット |
| US20130116701A1 (en) * | 2011-11-09 | 2013-05-09 | Boston Scientific Scimed, Inc. | Guide extension catheter |
| JP2015515348A (ja) * | 2012-04-27 | 2015-05-28 | アビオメド オイローパ ゲーエムベーハー | カテーテルシステムおよびカテーテルシステムを備える血管内血液ポンプ |
| US20160271363A1 (en) * | 2013-03-14 | 2016-09-22 | Abbott Cardiovascular Systems Inc. | Stiffness adjustable catheter |
Family Cites Families (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4909787A (en) * | 1986-08-14 | 1990-03-20 | Danforth John W | Controllable flexibility catheter with eccentric stiffener |
| JPH01158973A (ja) * | 1987-08-31 | 1989-06-22 | John W Danforth | カテーテル及びその使用方法 |
| WO1994003229A1 (en) * | 1992-07-31 | 1994-02-17 | Scimed Life Systems, Inc. | Catheter with distal tip guide wire lumen |
| US5336178A (en) * | 1992-11-02 | 1994-08-09 | Localmed, Inc. | Intravascular catheter with infusion array |
| US6508777B1 (en) * | 1998-05-08 | 2003-01-21 | Cardeon Corporation | Circulatory support system and method of use for isolated segmental perfusion |
| DE19821307C1 (de) * | 1998-05-13 | 1999-10-21 | Impella Cardiotech Gmbh | Intrakardiale Blutpumpe |
| US7780628B1 (en) * | 1999-01-11 | 2010-08-24 | Angiodynamics, Inc. | Apparatus and methods for treating congestive heart disease |
| EP1034808A1 (en) * | 1999-03-09 | 2000-09-13 | Paul Frederik Gründeman | A device for transventricular mechanical circulatory support |
| CH696424A5 (de) * | 2002-10-15 | 2007-06-15 | Alexander Von Weymarn Schaerli | Führungseinrichtung insbesondere zum Positionieren von Kathetern in einem Körpergang. |
| DE10336902C5 (de) * | 2003-08-08 | 2019-04-25 | Abiomed Europe Gmbh | Intrakardiale Pumpvorrichtung |
| WO2005051224A2 (en) * | 2003-11-25 | 2005-06-09 | F.D. Cardio Ltd. | Stent positioning using inflation tube |
| DE102004017834B4 (de) * | 2004-04-13 | 2011-01-27 | Siemens Ag | Kathetereinrichtung |
| US7841976B2 (en) * | 2006-03-23 | 2010-11-30 | Thoratec Corporation | Heart assist device with expandable impeller pump |
| US20060264907A1 (en) * | 2005-05-02 | 2006-11-23 | Pulsar Vascular, Inc. | Catheters having stiffening mechanisms |
| US7544201B2 (en) * | 2005-07-05 | 2009-06-09 | Futurematrix Interventional, Inc. | Rapid exchange balloon dilation catheter having reinforced multi-lumen distal portion |
| US7608056B2 (en) * | 2005-10-31 | 2009-10-27 | Wilson-Cook Medical Inc. | Steerable catheter devices and methods of articulating catheter devices |
| DE102006018489A1 (de) * | 2006-02-15 | 2007-10-25 | Epflex Feinwerktechnik Gmbh | Gesteuert versteifbarer Schlauch |
| US10123683B2 (en) * | 2006-03-02 | 2018-11-13 | Syntheon, Llc | Variably flexible insertion device and method for variably flexing an insertion device |
| DE102007014224A1 (de) * | 2007-03-24 | 2008-09-25 | Abiomed Europe Gmbh | Blutpumpe mit Mikromotor |
| FR2931680B1 (fr) * | 2008-05-27 | 2010-07-30 | Sayed Nour | Appareil permettant d'appliquer une pression pulsatile determinee sur un dispositif medical. |
| US9439721B2 (en) * | 2009-08-25 | 2016-09-13 | Medtronic Ablation Frontiers Llc | Bi-modal linear and loop ablation catheter, and method |
| EP2298371A1 (de) * | 2009-09-22 | 2011-03-23 | ECP Entwicklungsgesellschaft mbH | Funktionselement, insbesondere Fluidpumpe, mit einem Gehäuse und einem Förderelement |
| DE102009047845A1 (de) * | 2009-09-30 | 2011-03-31 | Abiomed Europe Gmbh | Herzunterstützungssystem |
| US20120179097A1 (en) * | 2011-01-06 | 2012-07-12 | Cully Edward H | Methods and apparatus for an adjustable stiffness catheter |
| US10058235B2 (en) * | 2011-03-01 | 2018-08-28 | Sanovas Intellectual Property, Llc | Steerable catheter |
| WO2012176189A1 (en) * | 2011-06-21 | 2012-12-27 | Dr. Eyal Bressler Ltd. | A catheter with an enhanced pushability |
| EP2802373B1 (en) * | 2012-01-11 | 2020-11-04 | UC-Care Ltd. | Catheterization apparatus |
| DE102012207053A1 (de) * | 2012-04-27 | 2013-10-31 | Abiomed Europe Gmbh | Intravasale rotationsblutpumpe |
| DE102012207049A1 (de) * | 2012-04-27 | 2015-08-13 | Abiomed Europe Gmbh | Intravasale rotationsblutpumpe |
| US10071222B2 (en) * | 2013-03-16 | 2018-09-11 | Clph, Llc | Electrode catheters and methods for making them |
| CN103536384A (zh) | 2013-10-21 | 2014-01-29 | 张�荣 | 阴茎多功能套 |
| DE102013021998A1 (de) * | 2013-12-30 | 2015-07-02 | Alexander Rübben | Ballonkatheter |
| WO2015175718A1 (en) * | 2014-05-13 | 2015-11-19 | Abiomed, Inc. | Cannula assembly |
| EP3042684A1 (en) * | 2015-01-07 | 2016-07-13 | Abiomed Europe GmbH | Introducer set |
| JP7044552B2 (ja) * | 2015-01-20 | 2022-03-30 | キューアペル メディカル, エルエルシー | 可変支持を有する管状構造 |
| EP3598986B1 (en) * | 2015-01-22 | 2021-02-17 | Tc1 Llc | Motor assembly with heat exchanger for catheter pump |
| CN108601925B (zh) * | 2015-12-04 | 2021-06-29 | 项目莫里股份有限公司 | 用于导管和其他用途的输入和铰接系统 |
| WO2018057650A1 (en) * | 2016-09-20 | 2018-03-29 | The Regents Of The University Of California | Hydraulically driven surgical apparatus |
| EP3398625A1 (en) * | 2017-05-04 | 2018-11-07 | Abiomed Europe GmbH | Blood pump with reinforced catheter |
-
2017
- 2017-05-04 EP EP17169486.2A patent/EP3398624A1/en not_active Withdrawn
-
2018
- 2018-05-03 ES ES18720632T patent/ES2902688T3/es active Active
- 2018-05-03 US US16/610,743 patent/US11744987B2/en active Active
- 2018-05-03 CN CN201880029851.5A patent/CN110603064A/zh active Pending
- 2018-05-03 WO PCT/EP2018/061349 patent/WO2018202775A1/en unknown
- 2018-05-03 KR KR1020197035501A patent/KR102605513B1/ko active IP Right Grant
- 2018-05-03 EP EP18720632.1A patent/EP3618884B1/en active Active
- 2018-05-03 SG SG11201909507R patent/SG11201909507RA/en unknown
- 2018-05-03 JP JP2019560767A patent/JP2020518404A/ja active Pending
- 2018-05-03 AU AU2018262629A patent/AU2018262629B2/en active Active
- 2018-05-03 DK DK18720632.1T patent/DK3618884T3/da active
- 2018-05-03 EP EP21209718.2A patent/EP3998103A1/en active Pending
- 2018-05-03 KR KR1020237039855A patent/KR20230164210A/ko not_active Application Discontinuation
-
2023
- 2023-03-27 JP JP2023049130A patent/JP2023068164A/ja active Pending
- 2023-07-18 US US18/223,109 patent/US20240050698A1/en active Pending
- 2023-09-15 AU AU2023229616A patent/AU2023229616A1/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009506839A (ja) * | 2005-09-02 | 2009-02-19 | ボストン サイエンティフィック リミテッド | 調整可能な剛性を有するカテーテル |
| JP2010536522A (ja) * | 2007-08-29 | 2010-12-02 | アクロシュターク コーポレイション | 被験者に小線源療法を施す(delivery)方法及びキット |
| US20130116701A1 (en) * | 2011-11-09 | 2013-05-09 | Boston Scientific Scimed, Inc. | Guide extension catheter |
| JP2015515348A (ja) * | 2012-04-27 | 2015-05-28 | アビオメド オイローパ ゲーエムベーハー | カテーテルシステムおよびカテーテルシステムを備える血管内血液ポンプ |
| US20160271363A1 (en) * | 2013-03-14 | 2016-09-22 | Abbott Cardiovascular Systems Inc. | Stiffness adjustable catheter |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2023229616A1 (en) | 2023-10-05 |
| DK3618884T3 (da) | 2022-01-10 |
| US11744987B2 (en) | 2023-09-05 |
| KR102605513B1 (ko) | 2023-11-23 |
| AU2018262629B2 (en) | 2023-06-22 |
| EP3998103A1 (en) | 2022-05-18 |
| WO2018202775A1 (en) | 2018-11-08 |
| EP3618884B1 (en) | 2021-11-24 |
| US20200094019A1 (en) | 2020-03-26 |
| JP2023068164A (ja) | 2023-05-16 |
| KR20230164210A (ko) | 2023-12-01 |
| CN110603064A (zh) | 2019-12-20 |
| EP3618884A1 (en) | 2020-03-11 |
| EP3398624A1 (en) | 2018-11-07 |
| KR20200003862A (ko) | 2020-01-10 |
| SG11201909507RA (en) | 2019-11-28 |
| ES2902688T3 (es) | 2022-03-29 |
| US20240050698A1 (en) | 2024-02-15 |
| AU2018262629A1 (en) | 2019-10-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240050698A1 (en) | Blood pump with reinforced catheter | |
| US11986604B2 (en) | Blood pump with reinforced catheter | |
| JP5899280B2 (ja) | 拡張可能な腸骨シースおよび使用方法 | |
| US8591538B2 (en) | Self-expanding cannula and a method for applying and positioning a self-expanding cannula | |
| JP5499375B2 (ja) | 腔内配送装置 | |
| KR20200038312A (ko) | 능동 도입기 외장 시스템 | |
| US8591539B2 (en) | Expandable conduit-guide and a method for applying and positioning an expandable conduit-guide | |
| JP3820270B2 (ja) | 大動脈内バルーンカテーテル | |
| KR20180075493A (ko) | 캐뉼러 배치를 위한 가이드 와이어 | |
| JP2020518404A5 (ja) | ||
| JP2022529938A (ja) | 血液ポンプシステムのための可変剛性遠位延長部 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210430 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20210430 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20220510 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220808 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20221129 |