JP2010188198A - 膨張可能なガイドシースおよび当該シースを使用する装置および方法 - Google Patents
膨張可能なガイドシースおよび当該シースを使用する装置および方法 Download PDFInfo
- Publication number
- JP2010188198A JP2010188198A JP2010132478A JP2010132478A JP2010188198A JP 2010188198 A JP2010188198 A JP 2010188198A JP 2010132478 A JP2010132478 A JP 2010132478A JP 2010132478 A JP2010132478 A JP 2010132478A JP 2010188198 A JP2010188198 A JP 2010188198A
- Authority
- JP
- Japan
- Prior art keywords
- sheath
- lumen
- reinforcing member
- distal end
- proximal
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 238000000034 method Methods 0.000 title abstract description 47
- 230000003014 reinforcing effect Effects 0.000 claims description 143
- 230000002787 reinforcement Effects 0.000 claims description 27
- 238000003780 insertion Methods 0.000 claims description 16
- 230000037431 insertion Effects 0.000 claims description 16
- 239000011248 coating agent Substances 0.000 claims description 4
- 238000000576 coating method Methods 0.000 claims description 4
- 239000012530 fluid Substances 0.000 abstract description 24
- 210000005166 vasculature Anatomy 0.000 abstract description 24
- 210000003484 anatomy Anatomy 0.000 abstract description 7
- 239000000463 material Substances 0.000 description 37
- 210000004204 blood vessel Anatomy 0.000 description 32
- 210000003462 vein Anatomy 0.000 description 30
- 208000031481 Pathologic Constriction Diseases 0.000 description 16
- 238000003384 imaging method Methods 0.000 description 16
- 230000036262 stenosis Effects 0.000 description 16
- 208000037804 stenosis Diseases 0.000 description 16
- 239000000853 adhesive Substances 0.000 description 8
- 230000001070 adhesive effect Effects 0.000 description 8
- 210000001072 colon Anatomy 0.000 description 8
- 238000004891 communication Methods 0.000 description 8
- 229940079593 drug Drugs 0.000 description 7
- 239000003814 drug Substances 0.000 description 7
- 238000000926 separation method Methods 0.000 description 7
- 239000002872 contrast media Substances 0.000 description 6
- 239000013013 elastic material Substances 0.000 description 6
- 238000002405 diagnostic procedure Methods 0.000 description 5
- 239000012528 membrane Substances 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 238000002560 therapeutic procedure Methods 0.000 description 5
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 4
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 4
- 210000001035 gastrointestinal tract Anatomy 0.000 description 4
- 210000004324 lymphatic system Anatomy 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 230000002093 peripheral effect Effects 0.000 description 4
- 210000002345 respiratory system Anatomy 0.000 description 4
- 230000002792 vascular Effects 0.000 description 4
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 3
- 239000004812 Fluorinated ethylene propylene Substances 0.000 description 3
- 239000004698 Polyethylene Substances 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 238000013019 agitation Methods 0.000 description 3
- 239000003146 anticoagulant agent Substances 0.000 description 3
- 229960004676 antithrombotic agent Drugs 0.000 description 3
- 239000002131 composite material Substances 0.000 description 3
- 230000008602 contraction Effects 0.000 description 3
- 238000002224 dissection Methods 0.000 description 3
- 230000003073 embolic effect Effects 0.000 description 3
- 229920000295 expanded polytetrafluoroethylene Polymers 0.000 description 3
- 238000002594 fluoroscopy Methods 0.000 description 3
- 238000002608 intravascular ultrasound Methods 0.000 description 3
- 229920009441 perflouroethylene propylene Polymers 0.000 description 3
- 239000004033 plastic Substances 0.000 description 3
- 229920003023 plastic Polymers 0.000 description 3
- 229920000573 polyethylene Polymers 0.000 description 3
- 229920000139 polyethylene terephthalate Polymers 0.000 description 3
- 239000005020 polyethylene terephthalate Substances 0.000 description 3
- 229920001296 polysiloxane Polymers 0.000 description 3
- -1 polytetrafluoroethylene Polymers 0.000 description 3
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 3
- 239000004810 polytetrafluoroethylene Substances 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 238000013151 thrombectomy Methods 0.000 description 3
- 238000002604 ultrasonography Methods 0.000 description 3
- 238000002679 ablation Methods 0.000 description 2
- 150000001336 alkenes Chemical class 0.000 description 2
- 238000002399 angioplasty Methods 0.000 description 2
- 210000001367 artery Anatomy 0.000 description 2
- 238000005452 bending Methods 0.000 description 2
- 229910002092 carbon dioxide Inorganic materials 0.000 description 2
- 239000001569 carbon dioxide Substances 0.000 description 2
- 230000000747 cardiac effect Effects 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 238000002788 crimping Methods 0.000 description 2
- 238000005520 cutting process Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000001125 extrusion Methods 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 238000001802 infusion Methods 0.000 description 2
- 239000004816 latex Substances 0.000 description 2
- 229920000126 latex Polymers 0.000 description 2
- 210000005240 left ventricle Anatomy 0.000 description 2
- 238000002595 magnetic resonance imaging Methods 0.000 description 2
- 238000013507 mapping Methods 0.000 description 2
- 238000002483 medication Methods 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 2
- 230000010412 perfusion Effects 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 208000005189 Embolism Diseases 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 229940124599 anti-inflammatory drug Drugs 0.000 description 1
- 239000002473 artificial blood Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 238000001574 biopsy Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 210000004720 cerebrum Anatomy 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 210000003748 coronary sinus Anatomy 0.000 description 1
- 230000010102 embolization Effects 0.000 description 1
- 239000008393 encapsulating agent Substances 0.000 description 1
- HQQADJVZYDDRJT-UHFFFAOYSA-N ethene;prop-1-ene Chemical group C=C.CC=C HQQADJVZYDDRJT-UHFFFAOYSA-N 0.000 description 1
- 210000003191 femoral vein Anatomy 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 230000002439 hemostatic effect Effects 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000002324 minimally invasive surgery Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical class [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000002250 progressing effect Effects 0.000 description 1
- 230000000541 pulsatile effect Effects 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 210000005245 right atrium Anatomy 0.000 description 1
- 238000007790 scraping Methods 0.000 description 1
- 238000009958 sewing Methods 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 229910000679 solder Inorganic materials 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 210000001321 subclavian vein Anatomy 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
- 238000003466 welding Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/1027—Making of balloon catheters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/34—Trocars; Puncturing needles
- A61B17/3417—Details of tips or shafts, e.g. grooves, expandable, bendable; Multiple coaxial sliding cannulas, e.g. for dilating
- A61B17/3421—Cannulas
- A61B17/3431—Cannulas being collapsible, e.g. made of thin flexible material
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/34—Trocars; Puncturing needles
- A61B17/3417—Details of tips or shafts, e.g. grooves, expandable, bendable; Multiple coaxial sliding cannulas, e.g. for dilating
- A61B17/3421—Cannulas
- A61B17/3439—Cannulas with means for changing the inner diameter of the cannula, e.g. expandable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/06—Body-piercing guide needles or the like
- A61M25/0662—Guide tubes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/1002—Balloon catheters characterised by balloon shape
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/104—Balloon catheters used for angioplasty
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2/011—Instruments for their placement or removal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/95—Instruments specially adapted for placement or removal of stents or stent-grafts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/95—Instruments specially adapted for placement or removal of stents or stent-grafts
- A61F2002/9505—Instruments specially adapted for placement or removal of stents or stent-grafts having retaining means other than an outer sleeve, e.g. male-female connector between stent and instrument
- A61F2002/9511—Instruments specially adapted for placement or removal of stents or stent-grafts having retaining means other than an outer sleeve, e.g. male-female connector between stent and instrument the retaining means being filaments or wires
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M2025/0177—Introducing, guiding, advancing, emplacing or holding catheters having external means for receiving guide wires, wires or stiffening members, e.g. loops, clamps or lateral tubes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/06—Body-piercing guide needles or the like
- A61M25/0662—Guide tubes
- A61M2025/0681—Systems with catheter and outer tubing, e.g. sheath, sleeve or guide tube
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/1002—Balloon catheters characterised by balloon shape
- A61M2025/1004—Balloons with folds, e.g. folded or multifolded
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M2025/1043—Balloon catheters with special features or adapted for special applications
- A61M2025/1088—Balloon catheters with special features or adapted for special applications having special surface characteristics depending on material properties or added substances, e.g. for reducing friction
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M29/00—Dilators with or without means for introducing media, e.g. remedies
- A61M29/02—Dilators made of swellable material
- A61M2029/025—Dilators made of swellable material characterised by the guiding element
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/06—Body-piercing guide needles or the like
- A61M25/0662—Guide tubes
- A61M25/0668—Guide tubes splittable, tear apart
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Surgery (AREA)
- Biophysics (AREA)
- Pulmonology (AREA)
- Anesthesiology (AREA)
- Hematology (AREA)
- Child & Adolescent Psychology (AREA)
- Molecular Biology (AREA)
- Medical Informatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pathology (AREA)
- Vascular Medicine (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Cardiology (AREA)
- Transplantation (AREA)
- Media Introduction/Drainage Providing Device (AREA)
- Endoscopes (AREA)
- Materials For Medical Uses (AREA)
- Shaping By String And By Release Of Stress In Plastics And The Like (AREA)
- Catching Or Destruction (AREA)
- Prostheses (AREA)
Abstract
【解決手段】収縮されている状態から、少なくとも部分的に内部で内腔を規定する拡張した状態にまで膨張できる可撓性を有するシースが提供される。シースは滑らかで比較的薄い壁を有し、それにより、曲がりくねったアナトミーを経由して及び/または比較的狭い通路中に、流体及び/又は器具を運ぶための折り畳み可能/膨張可能であるガイドを提供する。シースは、収縮した状態で、入口サイトから体管腔内に進行させられる。シースが目的とする体管腔に達すると、シースは拡張した状態に膨み、それによりシース内に内腔を規定し、流体および/または器具がシースの内腔を経由して体内に導入される。処理が終了すると、シースは体管腔から除去される。
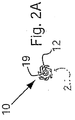
【選択図】図2A
Description
最小限の侵襲性の処置が、種々の医療の場において、例えば、脈官介入(例えば、脈管形成術、ステント植込み術、塞栓の予防、心臓電気刺激、心臓写像および明視化等)のために実施されてきた。これらの処置は、概して、器具を患者の脈管構造内に正確に誘導し、配置することに依存する。
そのような処置の間、ターゲット(または目的)とする管は、介在する脈管構造を経由して、ターゲットとする管に進行するガイドワイヤを用いてアクセスされ、それにより管に「線路」を与えることができる。1または複数の器具(例えば、カテーテル、シース等)がガイドワイヤまたは「レール」の上を進行して、管内に進行する。したがって、診断および/または治療処置は、これらの線路の上を1または複数の器具を進行させることにより実施され得る。
本発明は、医療処置の間、体管腔にアクセスするための、ならびに/または体管腔に器具および/もしくは薬剤を運ぶための装置および方法を概して対象としている。より特には、本発明は、ガイドシース、および当該ガイドシースを使用して患者の体管腔内(例えば、患者の冠状、神経および/または末梢の脈管構造内、患者の胃腸管、尿生殖管、呼吸器管、リンパ管系、および/または外科的に形成された流路内)に器具および/または薬剤を運ぶことを容易ならしめる方法を対象とする。
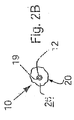
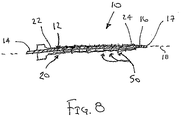
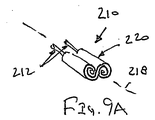
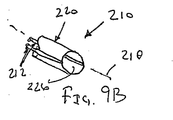
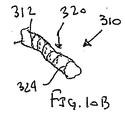
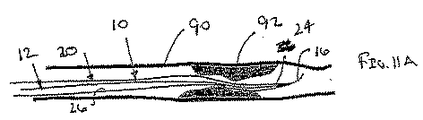
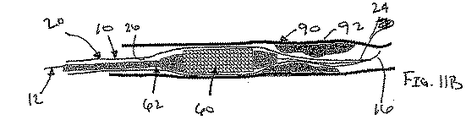
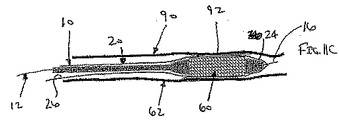
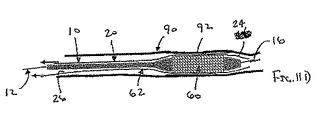
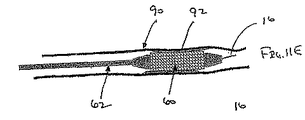
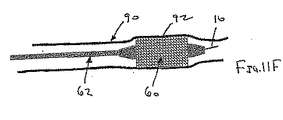
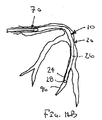
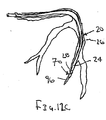
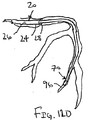
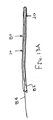
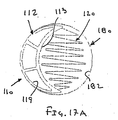
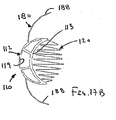
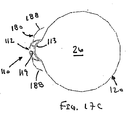
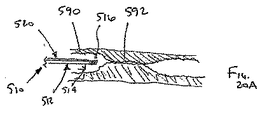
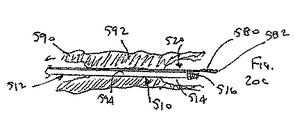
ここで図面を参照すると、図1A〜図2Bは、体管腔(図示せず)内へのアクセスを提供するための及び/または1または複数の器具(図示せず)を体管腔内へ送るための装置10の第1の好ましい形態を示す。体管腔は、例えば、患者の脈管構造内の管、尿生殖器管、呼吸器管、リンパ系、および患者の胃腸管内の通路等である。
(態様1)
患者の体管腔へアクセスする装置であって、
体管腔に挿入するための寸法および形状を有する遠位端部および近位端部を有する可撓性の補強部材;および
補強部材の近位端部と遠位端部との間で延在する膨張可能なシースであって、シースの輪郭を最小にして補強部材とともに管腔に挿入することを許容する収縮した状態から膨張することができ、補強部材の近位端部と遠位端部との間でシースが少なくとも部分的に内腔を規定する拡張した状態になる、シース
を含む装置。
(態様2)
シースがなめらかな材料を含む、態様1の装置。
(態様3)
シースがポリマーを含む態様1の装置。
(態様4)
ポリマーが、PTFE、EPTFE、FEP、PET、ウレタン、オレフィンおよびPEのうち少なくとも1つを含む、態様3の装置。
(態様5)
シースが弾性材料を含む、態様1の装置。
(態様6)
弾性材料が、シリコーン、ラテックス、イソプレンおよびクロノプレンのうち少なくとも1つを含む、態様5の装置。
(態様7)
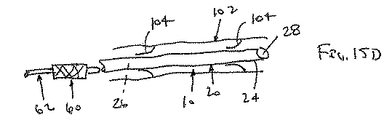
シースが、補強部材の近位端部と遠位端部との間の1または複数の箇所にて補強部材に取り付けられている管状部材を含む、態様1の装置。
(態様8)
補強部材が、シースに沿って延びる1または複数のストランドを含む、態様7の装置。
(態様9)
前記1または複数のストランドがシースに沿って、軸方向、らせん方向および円周方向のうち少なくとも1つの方向に延びる、態様8の装置。
(態様10)
シースが、シートおよび補強部材がともに内腔を規定するように、先端が補強部材に取り付けられているシート材料を含む、態様1の装置。
(態様11)
シースが、内腔と連絡している開口部を有する遠位端部を含み、内腔を通過して挿入される器具が開口部から体管腔に進行し得るようになっている、態様1の装置。
(態様12)
シースが実質的に閉じた遠位端部を含み、当該遠位端部が開口部を形成するための分離部分を含み、内腔を通過して挿入される器具が開口部から体管腔に進行し得るようになっている、態様1の装置。
(態様13)
分離部分が、シースを構成する材料が弱くされた領域された領域を含む、態様12の装置。
(態様14)
シースが補強部材の遠位端部に取り付けられた実質的に閉じた遠位端部を含み、シースの遠位端部が開口部を形成するように補強部材から分離可能であり、内腔を通過して挿入される器具が開口部から体管腔に進行し得るようになっている、態様1の装置。
(態様15)
シースが裂けるように構成されて、シースが内腔内に配置された器具の周囲から除去され得るようになっている、態様1の装置。
(態様16)
シースがシースの近位端部と遠位端部との間で延びる弱くされた領域を含む、態様15の装置。
(態様17)
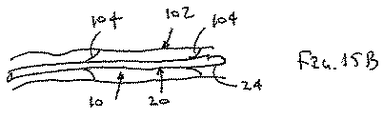
シースが、約0.01インチ(0.25mm)以下の壁厚さを有する、態様1の装置。
(態様18)
補強部材が、近位端部を押すことにより捩れる又は歪むおそれがなく遠位端部が体管腔を通過して進行し得るほど十分なコラム強さを有する、態様1の装置。
(態様19)
補強部材がガイドワイヤを含む、態様1の装置。
(態様20)
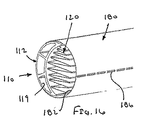
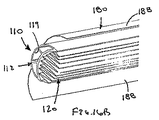
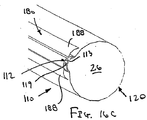
補強部材が弓形の断面を有し、それにより、近位端部と遠位端部との間で延びるグルーブであって、その内部にて、シースの少なくとも一部が、装置の断面輪郭を最小にする収縮した状態で配置されているグルーブを規定している、態様1の装置。
(態様21)
補強部材が、シースの長手方向の軸に実質的に平行に延びる長尺部材を含む、態様1の装置。
(態様22)
シースが、1または複数の箇所にて、補強部材に取り付けられている、態様1の装置。
(態様23)
シースが補強部材の近位端部と遠位端部に取り付けられている、態様21の装置。
(態様24)
シースが補強部材の遠位端部と近位端部との間で実質的に連続的に補強部材に取り付けられている、態様21の装置。
(態様25)
シースが取り外し可能なように補強部材に固定されている、態様21の装置。
(態様26)
補強部材が、シースの長手方向の軸に実質的に平行に延びる複数の長尺部材を含む、態様1の装置。
(態様27)
シースを収縮した状態に選択的に保持する拘束体をさらに含む、態様1の装置。
(態様28)
拘束体が、その内部に補強部材およびシースが配置され得る内腔を有する管状部材を含む、態様27の装置。
(態様29)
拘束体が、収縮した状態のシースを囲む外側シースを含み、当該外側シースが、シースが拡張した状態に膨張することを許容するように分離可能である1または複数の弱くされた領域を含む、態様27の装置。
(態様30)
外側シースが補強部材の近位端部と遠位端部との間で1または複数の箇所にて補強部材に取り付けられている、態様29の装置。
(態様31)
外側シースが、シースが収縮した状態にて配置され得る内腔を有し、当該内腔が流体をその内部に受け入れるように構成されていて、流体圧力が前記1または複数の弱くされた領域を分離させるようになっている、態様29の装置。
(態様32)
拘束体が膨張可能な部材の周囲に配置される1または複数のストランドを含む、態様27の装置。
(態様33)
拘束体が接着剤および生体吸収可能な封入材料のうち少なくとも1つを含む、態様27の装置。
(態様34)
シースが拡張した状態で近位端部と遠位端部との間で延びる複数の内腔を有する、態様1の装置。
(態様35)
前記複数の内腔の各々が、注入、吸引、潅流および攪拌のうち少なくとも1つのために構成されている、態様34の装置。
(態様36)
患者の体管腔へアクセスする装置であって、膨張可能な薄弱なシースであって、シースが患者の体の中への入口サイトとターゲットとする体管腔との間で延びるのに十分な長さ、近位端部、および遠位端部を含む、シースを含み、
シースが、シースの輪郭を最小にして体管腔への挿入を許容する収縮した状態から膨張可能であり、且つ近位端部と遠位端部との間で延びる内腔を少なくとも部分的に規定する拡張した状態になるものである、装置。
(態様37)
シースがなめらかな材料を含む、態様36の装置。
(態様38)
シースがポリマーを含む、態様36の装置。
(態様39)
シースが弾性材料を含む、態様36の装置。
(態様40)
シースが管状部材を含む、態様36の装置。
(態様41)
シースに沿って延びる1または複数の補強部材をさらに含む、態様36の装置。
(態様42)
1または複数の補強部材がシースに沿って、軸方向、らせん方向および周方向の少なくとも1つの方向に延びる、態様41の装置。
(態様43)
長尺の補強部材をさらに含み、シースが、先端が補強部材に取り付けられているシート材料を含み、シートおよび補強部材がともに内腔を規定するようになっている、態様36の装置。
(態様44)
シースが、内腔と連絡している開口部を有する遠位端部を含み、内腔を通過して挿入された器具が開口部から体管腔内に進行し得るようになっている、態様36の装置。
(態様45)
シースが実質的に閉じた遠位端部を含み、当該遠位端部が開口部を形成するための分離部分を含み、内腔を通過して挿入された器具が開口部から体管腔内に進行し得るようになっている、態様36の装置。
(態様46)
シースが裂けるように構成されていて、シースが内腔内に配置された器具の周囲から除去され得るようになっている、態様36の装置。
(態様47)
シースが約0.01インチ(0.25mm)以下の壁厚さを有する、態様36の装置。
(態様48)
シースを収縮した状態に選択的に維持する拘束体をさらに含む、態様36の装置。
(態様49)
シースの内腔を経由して挿入可能な1または複数の器具をさらに含む、態様36の装置。
(態様50)
患者の体管腔内への搬送装置であって、
近位端部および遠位端部を含む可撓性のカテーテル;
カテーテルの外側表面に取り付けられた膨張可能なシースであって、シースの近位端部と遠位端部との間で延びる内腔を含み、シースの輪郭を最小にする収縮した状態から膨張可能であって、且つシースの近位端部と遠位端部との間で延びる補助的な内腔をシースが少なくとも部分的に規定する拡張した状態になる、シースを含む、装置。
(態様51)
シースがなめらかな材料を含む、態様50の装置。
(態様52)
シースは薄弱な物質を含む、態様50の装置。
(態様53)
シースが実質的に閉じた遠位端部を含み、当該遠位端部が開口部を形成するための分離部分を含み、内腔を通過して挿入された器具が開口部から体管腔内に進行し得るようになっている、態様50の装置。
(態様54)
シースが約0.01インチ(0.25mm)以下の壁厚さを有する、態様50の装置。
(態様55)
シースを収縮した状態に選択的に維持する拘束体をさらに含む、態様50の装置。
Claims (12)
- 患者の体管腔へアクセスする装置であって、
体管腔に挿入するための寸法および形状を有する遠位端部および近位端部を有する可撓性の補強部材であって、遠位端部と近位端部との間で延在する1または複数の内腔を有する補強部材;および
補強部材の近位端部と遠位端部に取り付けられ、かつ近位端部と遠位端部との間で延在する膨張可能なシースであって、シースの輪郭を最小にして補強部材とともに体管腔に挿入することを許容する収縮した状態から膨張することができ、補強部材の近位端部と遠位端部との間でシースが少なくとも部分的に内腔を規定する拡張した状態になる、シース
を含み、
補強部材が、膨張可能なシースの遠位端部を越えて延びる実質的に非外傷性の遠位先端を有する、
装置。 - 膨張可能なシースが、補強部材の遠位先端に隣接して、テーパー形状となって終わっている、請求項1に記載の装置。
- テーパー形状が、丸くされた形状および斜めに切断された形状のうちの一方を含む、請求項2に記載の装置。
- 補強部材の遠位先端が、J字の先端および丸くされた先端のうちの一方を含む、請求項1〜3のいずれか1項に記載の装置。
- 補強部材が、シースの近位端部と遠位端部との間で延びるシースの独立したポケットまたは内腔に収容されている、請求項1〜4のいずれか1項に記載の装置。
- 患者の体管腔へアクセスする装置であって、
体管腔に挿入するための寸法および形状を有する遠位端部および近位端部を有する可撓性の補強部材であって、遠位端部と近位端部との間で延在する1または複数の内腔を有する補強部材;および
補強部材の近位端部と遠位端部に取り付けられ、かつ近位端部と遠位端部との間で延在する、薄弱かつ膨張可能なシースであって、シースの輪郭を最小にして補強部材とともに体管腔に挿入することを許容する収縮した状態から膨張することができ、補強部材の近位端部と遠位端部との間でシースが少なくとも部分的に内腔を規定する拡張した状態になる、シース
を含み、
補強部材が、シースの近位端部と遠位端部との間で延びるシースの独立したポケットまたは内腔に収容されている、
装置。 - 補強部材が、ポケットまたは内腔内で固定されていない、請求項5または6に記載の装置。
- シースが、1つ、2つまたはそれよりも多い部分に分割されて、シースの内腔を通って進行する電気リードから容易に除去され得る、請求項1〜7のいずれか1項に記載の装置。
- 電気リードをさらに含み、電気リードは、シースが補強部材に沿って進行させられてアクセスすべき部位に達した後に、シースの内腔を通って進行しうる、請求項1〜8のいずれか1項に記載の装置。
- 体管腔を経由して患者を処置するシステムであって、
体管腔に挿入するための寸法および形状を有する遠位端部および近位端部を有する可撓性の補強部材;
補強部材の近位端部と遠位端部との間で延在する膨張可能なシースであって、シースの輪郭を最小にして補強部材とともに体管腔に挿入することを許容する収縮した状態から膨張することができ、補強部材の近位端部と遠位端部との間でシースが少なくとも部分的に内腔を規定する拡張した状態になる、シース;および
シースが補強部材とともに進行させられて患者の所望の体管腔内に達した後に、シースの内腔を通って進行しうる、電気リード
を含む、システム。 - シースが、リードを内腔内に挿入することを容易にするために、滑らかなコーティングを含む、請求項10に記載のシステム。
- シースが、内腔に配置される器具の周囲から除去され得るように裂けるように構成されている、請求項10または11に記載のシステム。
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US37606502P | 2002-04-25 | 2002-04-25 | |
| US60/376,065 | 2002-04-25 | ||
| US10/423,321 US7762995B2 (en) | 2002-04-25 | 2003-04-24 | Expandable guide sheath and apparatus and methods using such sheaths |
| US10/423,321 | 2003-04-24 |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003587459A Division JP4583032B2 (ja) | 2002-04-25 | 2003-04-24 | 膨張可能なガイドシースおよび当該シースを使用する装置および方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010188198A true JP2010188198A (ja) | 2010-09-02 |
| JP5487015B2 JP5487015B2 (ja) | 2014-05-07 |
Family
ID=29273046
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003587459A Expired - Lifetime JP4583032B2 (ja) | 2002-04-25 | 2003-04-24 | 膨張可能なガイドシースおよび当該シースを使用する装置および方法 |
| JP2010132478A Expired - Lifetime JP5487015B2 (ja) | 2002-04-25 | 2010-06-09 | 膨張可能なガイドシースおよび当該シースを使用する装置および方法 |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003587459A Expired - Lifetime JP4583032B2 (ja) | 2002-04-25 | 2003-04-24 | 膨張可能なガイドシースおよび当該シースを使用する装置および方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (2) | US7762995B2 (ja) |
| EP (2) | EP2067499B1 (ja) |
| JP (2) | JP4583032B2 (ja) |
| AT (1) | ATE427768T1 (ja) |
| AU (1) | AU2003223749A1 (ja) |
| DE (1) | DE60327067D1 (ja) |
| WO (1) | WO2003090834A2 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016505309A (ja) * | 2012-12-18 | 2016-02-25 | コーニンクレッカ フィリップス エヌ ヴェKoninklijke Philips N.V. | 表面及び身体温度測定に対する再使用可能なmr安全温度プローブ |
Families Citing this family (261)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6306166B1 (en) * | 1997-08-13 | 2001-10-23 | Scimed Life Systems, Inc. | Loading and release of water-insoluble drugs |
| US20030088240A1 (en) * | 2001-11-02 | 2003-05-08 | Vahid Saadat | Methods and apparatus for cryo-therapy |
| AU2003223749A1 (en) | 2002-04-25 | 2003-11-10 | The Board Of Trustees Of The Leland Stanford Junior University | Expandable guide sheath and apparatus and methods using such sheaths |
| US8956280B2 (en) | 2002-05-30 | 2015-02-17 | Intuitive Surgical Operations, Inc. | Apparatus and methods for placing leads using direct visualization |
| US6793678B2 (en) | 2002-06-27 | 2004-09-21 | Depuy Acromed, Inc. | Prosthetic intervertebral motion disc having dampening |
| US7309334B2 (en) | 2002-07-23 | 2007-12-18 | Von Hoffmann Gerard | Intracranial aspiration catheter |
| US8425549B2 (en) | 2002-07-23 | 2013-04-23 | Reverse Medical Corporation | Systems and methods for removing obstructive matter from body lumens and treating vascular defects |
| US7591832B2 (en) * | 2003-04-24 | 2009-09-22 | Medtronic, Inc. | Expandable guide sheath and apparatus with distal protection and methods for use |
| US8388628B2 (en) * | 2003-04-24 | 2013-03-05 | Medtronic, Inc. | Expandable sheath for delivering instruments and agents into a body lumen and methods for use |
| US7909766B2 (en) * | 2003-05-21 | 2011-03-22 | Scimed Life Systems, Inc. | Systems and methods for improving the imaging resolution of an imaging transducer |
| WO2005032642A2 (en) * | 2003-10-03 | 2005-04-14 | Acumen Medical, Inc. | Expandable guide sheath and apparatus and methods for making them |
| US9241735B2 (en) * | 2003-12-05 | 2016-01-26 | Onset Medical Corporation | Expandable percutaneous sheath |
| US7780692B2 (en) | 2003-12-05 | 2010-08-24 | Onset Medical Corporation | Expandable percutaneous sheath |
| ATE468830T1 (de) * | 2004-03-31 | 2010-06-15 | Wilson Cook Medical Inc | Stenteinführsystem |
| US20050245984A1 (en) * | 2004-04-30 | 2005-11-03 | Medtronic, Inc. | Implantable medical device with lubricious material |
| US7422559B2 (en) | 2004-06-16 | 2008-09-09 | Ge Inspection Technologies, Lp | Borescope comprising fluid supply system |
| US9387313B2 (en) * | 2004-08-03 | 2016-07-12 | Interventional Spine, Inc. | Telescopic percutaneous tissue dilation systems and related methods |
| US20060030872A1 (en) * | 2004-08-03 | 2006-02-09 | Brad Culbert | Dilation introducer for orthopedic surgery |
| US7892203B2 (en) | 2004-09-09 | 2011-02-22 | Onset Medical Corporation | Expandable transluminal sheath |
| US20060135962A1 (en) | 2004-09-09 | 2006-06-22 | Kick George F | Expandable trans-septal sheath |
| US7993350B2 (en) * | 2004-10-04 | 2011-08-09 | Medtronic, Inc. | Shapeable or steerable guide sheaths and methods for making and using them |
| US7875049B2 (en) * | 2004-10-04 | 2011-01-25 | Medtronic, Inc. | Expandable guide sheath with steerable backbone and methods for making and using them |
| US8872906B2 (en) | 2005-01-05 | 2014-10-28 | Avantis Medical Systems, Inc. | Endoscope assembly with a polarizing filter |
| US8182422B2 (en) | 2005-12-13 | 2012-05-22 | Avantis Medical Systems, Inc. | Endoscope having detachable imaging device and method of using |
| US8289381B2 (en) | 2005-01-05 | 2012-10-16 | Avantis Medical Systems, Inc. | Endoscope with an imaging catheter assembly and method of configuring an endoscope |
| US8797392B2 (en) | 2005-01-05 | 2014-08-05 | Avantis Medical Sytems, Inc. | Endoscope assembly with a polarizing filter |
| US20060184105A1 (en) * | 2005-02-15 | 2006-08-17 | Townsend Gregory L | Thin wall catheter and method of placing same |
| WO2006122061A1 (en) * | 2005-05-06 | 2006-11-16 | Acumen Medical, Inc. | Complexly shaped steerable catheters and methods for making and using them |
| US8092481B2 (en) | 2005-06-03 | 2012-01-10 | Onset Medical Corporation | Expandable percutaneous sheath |
| US8801744B2 (en) * | 2006-06-28 | 2014-08-12 | Abbott Laboratories | Expandable introducer sheath to preserve guidewire access |
| US20080004571A1 (en) * | 2006-06-28 | 2008-01-03 | Abbott Laboratories | Expandable introducer sheath |
| US9168359B2 (en) | 2005-06-30 | 2015-10-27 | Abbott Laboratories | Modular introducer and exchange sheath |
| US8440122B2 (en) | 2005-06-30 | 2013-05-14 | Abbott Vascular Inc. | Introducer sheath and methods of making |
| US9352118B2 (en) * | 2005-06-30 | 2016-05-31 | Abbott Laboratories | Modular introducer and exchange sheath |
| US20100130937A1 (en) * | 2005-06-30 | 2010-05-27 | Abbott Vascular Inc. | Introducer sheath and methods of making |
| US8359723B2 (en) | 2005-06-30 | 2013-01-29 | Abbott Vascular Inc. | Introducer sheath and methods of making |
| US9597063B2 (en) * | 2006-06-28 | 2017-03-21 | Abbott Laboratories | Expandable introducer sheath to preserve guidewire access |
| US7575569B2 (en) * | 2005-08-16 | 2009-08-18 | Medtronic, Inc. | Apparatus and methods for delivering stem cells and other agents into cardiac tissue |
| US7765014B2 (en) | 2005-08-16 | 2010-07-27 | Medtronic, Inc. | Apparatus and methods for delivering transvenous leads |
| US7556710B2 (en) * | 2005-10-04 | 2009-07-07 | Ilh, Llc | Catheters with lubricious linings and methods for making and using them |
| WO2009114556A2 (en) * | 2005-10-04 | 2009-09-17 | Ilh, Llc | Catheters with lubricious linings and methods for making and using them |
| US7553387B2 (en) | 2005-10-04 | 2009-06-30 | Ilh, Llc | Catheters with lubricious linings and methods for making and using them |
| US9974887B2 (en) | 2005-10-04 | 2018-05-22 | Clph, Llc | Catheters with lubricious linings and methods for making and using them |
| US7550053B2 (en) * | 2006-01-26 | 2009-06-23 | Ilh, Llc | Catheters with lubricious linings and methods for making and using them |
| US20070088296A1 (en) * | 2005-10-04 | 2007-04-19 | Leeflang Stephen A | Catheters with lubricious linings and methods for making and using them |
| US7637902B2 (en) | 2005-11-23 | 2009-12-29 | Medtronic, Inc. | Slittable and peelable sheaths and methods for making and using them |
| WO2007087421A2 (en) | 2006-01-23 | 2007-08-02 | Avantis Medical Systems, Inc. | Endoscope |
| US20070185444A1 (en) * | 2006-02-03 | 2007-08-09 | Starfire Medical, Inc. | Vascular clip-on occlusion system |
| US20070185457A1 (en) * | 2006-02-03 | 2007-08-09 | Starfire Medical, Inc. | Vascular occlusion delivery |
| US20070185442A1 (en) * | 2006-02-03 | 2007-08-09 | Starfire Medical, Inc. | Rapid balloon coupling system |
| US20070185443A1 (en) * | 2006-02-03 | 2007-08-09 | Starfire Medical, Inc. | Vascular therapy delivery system |
| US8287446B2 (en) | 2006-04-18 | 2012-10-16 | Avantis Medical Systems, Inc. | Vibratory device, endoscope having such a device, method for configuring an endoscope, and method of reducing looping of an endoscope |
| US9084901B2 (en) | 2006-04-28 | 2015-07-21 | Medtronic, Inc. | Cranial implant |
| JP2009537283A (ja) | 2006-05-19 | 2009-10-29 | アヴァンティス メディカル システムズ インコーポレイテッド | ビデオアーチファクトの影響を低減するための装置および方法 |
| US9889275B2 (en) | 2006-06-28 | 2018-02-13 | Abbott Laboratories | Expandable introducer sheath to preserve guidewire access |
| US20100198160A1 (en) * | 2006-06-28 | 2010-08-05 | Abbott Vascular Inc. | Expandable Introducer Sheaths and Methods for Manufacture and Use |
| US20080033396A1 (en) * | 2006-08-01 | 2008-02-07 | Percutaneous Systems, Inc. | Vascular sheaths and methods for their deployment |
| US7927272B2 (en) | 2006-08-04 | 2011-04-19 | Avantis Medical Systems, Inc. | Surgical port with embedded imaging device |
| CA2976839C (en) * | 2006-09-08 | 2020-04-28 | Edwards Lifesciences Corporation | Integrated heart valve delivery system |
| US8105382B2 (en) | 2006-12-07 | 2012-01-31 | Interventional Spine, Inc. | Intervertebral implant |
| WO2008079828A2 (en) | 2006-12-20 | 2008-07-03 | Onset Medical Corporation | Expandable trans-septal sheath |
| CA2673991C (en) | 2007-01-21 | 2012-02-07 | Hemoteq Ag | Methods for coating catheter balloons with a defined quantity of active agent |
| US7722568B2 (en) | 2007-01-29 | 2010-05-25 | Onset Medical Corporation | Expandable intra-aortic balloon pump sheath |
| US7686783B2 (en) | 2007-03-30 | 2010-03-30 | Boston Scientific Scimed, Inc. | Perfusion and embolic protection |
| US7780630B2 (en) * | 2007-03-30 | 2010-08-24 | Boston Scientific Scimed, Inc. | Perfusion device |
| WO2008121710A1 (en) * | 2007-03-30 | 2008-10-09 | Boston Scientific Scimed, Inc. | Catheter for perfusion and embolic protection |
| WO2008121888A1 (en) | 2007-03-30 | 2008-10-09 | Onset Medical Corporation | Expandable trans-septal sheath |
| US8064666B2 (en) | 2007-04-10 | 2011-11-22 | Avantis Medical Systems, Inc. | Method and device for examining or imaging an interior surface of a cavity |
| US8900307B2 (en) | 2007-06-26 | 2014-12-02 | DePuy Synthes Products, LLC | Highly lordosed fusion cage |
| US9192697B2 (en) | 2007-07-03 | 2015-11-24 | Hemoteq Ag | Balloon catheter for treating stenosis of body passages and for preventing threatening restenosis |
| US8075575B2 (en) | 2007-08-14 | 2011-12-13 | Toby Orthopaedics, Llc | Device and method for assisting in flexor tendon repair and rehabilitation |
| WO2009046164A1 (en) | 2007-10-05 | 2009-04-09 | Tyco Healthcare Group Lp | Seal anchor for use in surgical procedures |
| US8673100B2 (en) * | 2007-10-19 | 2014-03-18 | Stephen A. Leeflang | Strip lined catheters and methods for constructing and processing strip lined catheters |
| EP2471493A1 (en) | 2008-01-17 | 2012-07-04 | Synthes GmbH | An expandable intervertebral implant and associated method of manufacturing the same |
| US8109908B1 (en) | 2008-01-22 | 2012-02-07 | Greatbatch Ltd. | Biodegradable shroud for a dilator/sheath assembly |
| CN102036623A (zh) | 2008-04-05 | 2011-04-27 | 斯恩蒂斯有限公司 | 可膨胀的椎间植入体 |
| US20090259283A1 (en) * | 2008-04-09 | 2009-10-15 | Brandt Michael S | Sheathed lead for pacing or defibrillation |
| US8668668B2 (en) | 2008-05-14 | 2014-03-11 | Onset Medical Corporation | Expandable iliac sheath and method of use |
| US8562559B2 (en) | 2008-05-14 | 2013-10-22 | Onset Medical Corporation | Expandable iliac sheath and method of use |
| FR2932979B1 (fr) * | 2008-06-25 | 2012-04-06 | Perouse Lab | Dispositif introducteur s'etendant entre un point proximal et un point distal et necessaire de traitement associe. |
| US8403889B2 (en) | 2008-06-25 | 2013-03-26 | Covidien Lp | Access assembly |
| KR20160049027A (ko) * | 2008-08-28 | 2016-05-04 | 마르코 안토니오 페나 듀크 | 인트라루미널 장치의 지향성 팽창 |
| US8403896B2 (en) * | 2008-08-29 | 2013-03-26 | AUST Development, LLC | Apparatus and methods for making coated liners and tubular devices including such liners |
| USD738500S1 (en) | 2008-10-02 | 2015-09-08 | Covidien Lp | Seal anchor for use in surgical procedures |
| US8790387B2 (en) | 2008-10-10 | 2014-07-29 | Edwards Lifesciences Corporation | Expandable sheath for introducing an endovascular delivery device into a body |
| US7951110B2 (en) | 2008-11-10 | 2011-05-31 | Onset Medical Corporation | Expandable spinal sheath and method of use |
| US9468364B2 (en) | 2008-11-14 | 2016-10-18 | Intuitive Surgical Operations, Inc. | Intravascular catheter with hood and image processing systems |
| US8992558B2 (en) | 2008-12-18 | 2015-03-31 | Osteomed, Llc | Lateral access system for the lumbar spine |
| US8758847B2 (en) * | 2009-02-18 | 2014-06-24 | AUST Development, LLC | Apparatus and methods for making coated liners and tubular devices including such liners |
| US8845582B2 (en) | 2009-03-09 | 2014-09-30 | Flip Technologies Limited | Device for protecting a catheter, and a method for locating a catheter in a remote location in a human or animal body |
| US9526620B2 (en) | 2009-03-30 | 2016-12-27 | DePuy Synthes Products, Inc. | Zero profile spinal fusion cage |
| ES2550634T3 (es) | 2009-07-10 | 2015-11-11 | Boston Scientific Scimed, Inc. | Uso de nanocristales para un balón de suministro de fármaco |
| EP2453938B1 (en) | 2009-07-17 | 2015-08-19 | Boston Scientific Scimed, Inc. | Nucleation of drug delivery balloons to provide improved crystal size and density |
| US20110082370A1 (en) * | 2009-10-02 | 2011-04-07 | Wilson-Cook Medical Inc. | Endoscopic fascia tunneling |
| US9539081B2 (en) | 2009-12-02 | 2017-01-10 | Surefire Medical, Inc. | Method of operating a microvalve protection device |
| US9393129B2 (en) | 2009-12-10 | 2016-07-19 | DePuy Synthes Products, Inc. | Bellows-like expandable interbody fusion cage |
| US20110152917A1 (en) * | 2009-12-22 | 2011-06-23 | Boston Scientific Scimed, Inc. | Intravascular filter restraining device |
| US20110160645A1 (en) * | 2009-12-31 | 2011-06-30 | Boston Scientific Scimed, Inc. | Cryo Activated Drug Delivery and Cutting Balloons |
| US8556896B2 (en) * | 2010-01-27 | 2013-10-15 | Zimmer, Inc. | Distal relief for a surgical device |
| US8728162B2 (en) | 2010-04-15 | 2014-05-20 | Osteomed, Llc | Direct lateral spine system instruments, implants and associated methods |
| US20110282353A1 (en) * | 2010-05-11 | 2011-11-17 | Mchugo Vincent | Biliary access sheath |
| US8979860B2 (en) | 2010-06-24 | 2015-03-17 | DePuy Synthes Products. LLC | Enhanced cage insertion device |
| US9282979B2 (en) | 2010-06-24 | 2016-03-15 | DePuy Synthes Products, Inc. | Instruments and methods for non-parallel disc space preparation |
| AU2011271465B2 (en) | 2010-06-29 | 2015-03-19 | Synthes Gmbh | Distractible intervertebral implant |
| AU2011272764B2 (en) * | 2010-06-30 | 2015-11-19 | Muffin Incorporated | Percutaneous, ultrasound-guided introduction of medical devices |
| US10390889B2 (en) * | 2010-07-26 | 2019-08-27 | St Jude Medical International Holding S.Á R.L. | Removable navigation system and method for a medical device |
| US8889211B2 (en) | 2010-09-02 | 2014-11-18 | Boston Scientific Scimed, Inc. | Coating process for drug delivery balloons using heat-induced rewrap memory |
| US20120071975A1 (en) * | 2010-09-10 | 2012-03-22 | Eduardo Gonzalez-Hernandez | Device and method for assisting in flexor tendon repair and rehabilitation |
| US9402732B2 (en) | 2010-10-11 | 2016-08-02 | DePuy Synthes Products, Inc. | Expandable interspinous process spacer implant |
| US9414944B2 (en) * | 2010-11-11 | 2016-08-16 | W. L. Gore & Associates, Inc. | Deployment sleeve shortening mechanism |
| US9675487B2 (en) * | 2010-11-17 | 2017-06-13 | Cook Medical Technologies Llc | Prosthesis deployment system for vascular repair |
| EP2455018B1 (de) * | 2010-11-22 | 2014-05-21 | Karl Storz GmbH & Co. KG | Trokarhülse zum Einsetzen in eine Hautinzision |
| US8948848B2 (en) | 2011-01-07 | 2015-02-03 | Innovative Cardiovascular Solutions, Llc | Angiography catheter |
| KR20140008446A (ko) * | 2011-05-06 | 2014-01-21 | 더블유.엘. 고어 앤드 어소시에이트스, 인코포레이티드 | 에코발생 슬리브 |
| DE102011050173A1 (de) * | 2011-05-06 | 2012-11-08 | Vilmos Nagy | Dehnbare Einführschleuse und Schleusensystem |
| WO2013022458A1 (en) | 2011-08-05 | 2013-02-14 | Boston Scientific Scimed, Inc. | Methods of converting amorphous drug substance into crystalline form |
| US10213329B2 (en) * | 2011-08-12 | 2019-02-26 | W. L. Gore & Associates, Inc. | Evertable sheath devices, systems, and methods |
| WO2013028208A1 (en) | 2011-08-25 | 2013-02-28 | Boston Scientific Scimed, Inc. | Medical device with crystalline drug coating |
| US10238837B2 (en) | 2011-10-14 | 2019-03-26 | Intuitive Surgical Operations, Inc. | Catheters with control modes for interchangeable probes |
| US9452276B2 (en) | 2011-10-14 | 2016-09-27 | Intuitive Surgical Operations, Inc. | Catheter with removable vision probe |
| WO2013060740A1 (en) * | 2011-10-28 | 2013-05-02 | Advant Medical Limited | Protection device for a prostheses and/or balloon catheter and method making and using the same |
| US11213318B2 (en) | 2011-11-10 | 2022-01-04 | Medtronic Vascular, Inc. | Expandable introducer sheath and method |
| CA3082091C (en) | 2011-11-10 | 2023-01-31 | Transaortic Medical, Inc. | System for deploying a device to a distal location across a diseased vessel |
| US20130338677A1 (en) * | 2012-06-18 | 2013-12-19 | Biotronik Ag | Insertion element and insertion device |
| US9283072B2 (en) | 2012-07-25 | 2016-03-15 | W. L. Gore & Associates, Inc. | Everting transcatheter valve and methods |
| WO2014018098A1 (en) | 2012-07-26 | 2014-01-30 | DePuy Synthes Products, LLC | Expandable implant |
| US10376360B2 (en) | 2012-07-27 | 2019-08-13 | W. L. Gore & Associates, Inc. | Multi-frame prosthetic valve apparatus and methods |
| US20140067069A1 (en) | 2012-08-30 | 2014-03-06 | Interventional Spine, Inc. | Artificial disc |
| US9192751B2 (en) | 2012-10-26 | 2015-11-24 | Medtronic, Inc. | Elastic introducer sheath |
| US9907931B2 (en) | 2012-10-26 | 2018-03-06 | Medtronic, Inc. | Elastic introducer sheath |
| AU2013337807B2 (en) | 2012-11-01 | 2016-12-08 | Muffin Incorporated | Implements for identifying sheath migration |
| US9414752B2 (en) | 2012-11-09 | 2016-08-16 | Elwha Llc | Embolism deflector |
| US9737398B2 (en) | 2012-12-19 | 2017-08-22 | W. L. Gore & Associates, Inc. | Prosthetic valves, frames and leaflets and methods thereof |
| US10039638B2 (en) | 2012-12-19 | 2018-08-07 | W. L. Gore & Associates, Inc. | Geometric prosthetic heart valves |
| US10966820B2 (en) | 2012-12-19 | 2021-04-06 | W. L. Gore & Associates, Inc. | Geometric control of bending character in prosthetic heart valve leaflets |
| US9144492B2 (en) | 2012-12-19 | 2015-09-29 | W. L. Gore & Associates, Inc. | Truncated leaflet for prosthetic heart valves, preformed valve |
| US9101469B2 (en) | 2012-12-19 | 2015-08-11 | W. L. Gore & Associates, Inc. | Prosthetic heart valve with leaflet shelving |
| US10321986B2 (en) | 2012-12-19 | 2019-06-18 | W. L. Gore & Associates, Inc. | Multi-frame prosthetic heart valve |
| US9968443B2 (en) | 2012-12-19 | 2018-05-15 | W. L. Gore & Associates, Inc. | Vertical coaptation zone in a planar portion of prosthetic heart valve leaflet |
| BR112015013364B1 (pt) * | 2012-12-31 | 2021-04-20 | Clearstream Technologies Limited | aparelho para tratar uma área de tratamento em uma vasculatura e método de montagem de um conjunto que inclui um cateter que inclui um balão |
| US20140228661A1 (en) * | 2013-02-08 | 2014-08-14 | Liviu Popa-Simil | Biopsy method and gun set devices |
| US20140228814A1 (en) * | 2013-02-09 | 2014-08-14 | Boston Scientific Scimed, Inc. | Expandable sheath |
| US9717601B2 (en) | 2013-02-28 | 2017-08-01 | DePuy Synthes Products, Inc. | Expandable intervertebral implant, system, kit and method |
| US9522070B2 (en) | 2013-03-07 | 2016-12-20 | Interventional Spine, Inc. | Intervertebral implant |
| US9539411B2 (en) * | 2013-03-13 | 2017-01-10 | W. L. Gore & Associates, Inc. | Deconstructable endoluminal devices and related systems and methods |
| US20140276905A1 (en) * | 2013-03-18 | 2014-09-18 | Biotronik Ag | Insertion Element and Insertion Device |
| AU2014262488B2 (en) * | 2013-05-10 | 2018-08-30 | Medtronic, Inc. | System for deploying a device to a distal location across a diseased vessel |
| CA2912204A1 (en) | 2013-05-17 | 2014-11-20 | Transaortic Medical, Inc. | Expandable introducer sheath |
| US9629978B2 (en) | 2013-05-20 | 2017-04-25 | Clph, Llc | Catheters with intermediate layers and methods for making them |
| US10434307B2 (en) | 2013-10-15 | 2019-10-08 | Medtronic, Inc. | Methods and devices for subcutaneous lead implantation |
| US9610436B2 (en) | 2013-11-12 | 2017-04-04 | Medtronic, Inc. | Implant tools with attachment feature and multi-positional sheath and implant techniques utilizing such tools |
| US10792490B2 (en) | 2013-11-12 | 2020-10-06 | Medtronic, Inc. | Open channel implant tools and implant techniques utilizing such tools |
| US10966850B2 (en) | 2014-03-06 | 2021-04-06 | W. L. Gore & Associates, Inc. | Implantable medical device constraint and deployment apparatus |
| US9968740B2 (en) | 2014-03-25 | 2018-05-15 | Surefire Medical, Inc. | Closed tip dynamic microvalve protection device |
| JP2016005547A (ja) * | 2014-05-27 | 2016-01-14 | 公立大学法人横浜市立大学 | リンパ管位置検出用ワイヤ |
| GB201411486D0 (en) * | 2014-06-27 | 2014-08-13 | Isis Innovation | Apparatus for providing and maintaining access to a surgical site |
| US10064649B2 (en) | 2014-07-07 | 2018-09-04 | Covidien Lp | Pleated seal for surgical hand or instrument access |
| CN106659567B (zh) | 2014-08-18 | 2019-06-14 | W.L.戈尔及同仁股份有限公司 | 带有用于假体瓣膜的一体缝合封套的框架 |
| US9827094B2 (en) | 2014-09-15 | 2017-11-28 | W. L. Gore & Associates, Inc. | Prosthetic heart valve with retention elements |
| US10569063B2 (en) | 2014-10-03 | 2020-02-25 | W. L. Gore & Associates, Inc. | Removable covers for drug eluting medical devices |
| WO2016064449A1 (en) | 2014-10-20 | 2016-04-28 | Research Development International Corporation | Steerable micro-endoscope |
| US9707011B2 (en) | 2014-11-12 | 2017-07-18 | Covidien Lp | Attachments for use with a surgical access device |
| US11083491B2 (en) | 2014-12-09 | 2021-08-10 | Medtronic, Inc. | Extravascular implant tools utilizing a bore-in mechanism and implant techniques using such tools |
| US10349978B2 (en) | 2014-12-18 | 2019-07-16 | Medtronic, Inc. | Open channel implant tool with additional lumen and implant techniques utilizing such tools |
| US11426290B2 (en) | 2015-03-06 | 2022-08-30 | DePuy Synthes Products, Inc. | Expandable intervertebral implant, system, kit and method |
| US20160287839A1 (en) | 2015-03-31 | 2016-10-06 | Surefire Medical, Inc. | Apparatus and Method for Infusing an Immunotherapy Agent to a Solid Tumor for Treatment |
| US10327896B2 (en) | 2015-04-10 | 2019-06-25 | Edwards Lifesciences Corporation | Expandable sheath with elastomeric cross sectional portions |
| US10792471B2 (en) | 2015-04-10 | 2020-10-06 | Edwards Lifesciences Corporation | Expandable sheath |
| US12194256B2 (en) | 2015-04-10 | 2025-01-14 | Edwards Lifesciences Corporation | Expandable sheath |
| US9913727B2 (en) | 2015-07-02 | 2018-03-13 | Medos International Sarl | Expandable implant |
| US20170189059A1 (en) * | 2016-01-06 | 2017-07-06 | Boston Scientific Scimed, Inc. | Percutaneous access device |
| US10548631B2 (en) | 2016-03-04 | 2020-02-04 | Boston Scientific Scimed Inc. | Introducer with expandable capabilities |
| US10940294B2 (en) * | 2016-04-25 | 2021-03-09 | Medtronic Vascular, Inc. | Balloon catheter including a drug delivery sheath |
| EP3474783B1 (en) | 2016-06-28 | 2023-05-03 | Eit Emerging Implant Technologies GmbH | Expandable, angularly adjustable intervertebral cages |
| WO2018002715A2 (en) | 2016-06-28 | 2018-01-04 | Eit Emerging Implant Technologies Gmbh | Expandable and angularly adjustable articulating intervertebral cages |
| WO2018039228A1 (en) | 2016-08-23 | 2018-03-01 | Stryker European Holdings I, Llc | Instrumentation for the implantation of spinal implants |
| WO2018039000A1 (en) * | 2016-08-24 | 2018-03-01 | W. L. Gore & Associates, Inc. | Sleeves for expandable medical devices |
| US11400263B1 (en) | 2016-09-19 | 2022-08-02 | Trisalus Life Sciences, Inc. | System and method for selective pressure-controlled therapeutic delivery |
| US10780250B1 (en) | 2016-09-19 | 2020-09-22 | Surefire Medical, Inc. | System and method for selective pressure-controlled therapeutic delivery |
| US10537436B2 (en) | 2016-11-01 | 2020-01-21 | DePuy Synthes Products, Inc. | Curved expandable cage |
| US10888433B2 (en) | 2016-12-14 | 2021-01-12 | DePuy Synthes Products, Inc. | Intervertebral implant inserter and related methods |
| US10806893B2 (en) | 2017-01-10 | 2020-10-20 | Surefire Medical, Inc. | Guiding catheter having shape-retentive distal end |
| US10912919B2 (en) | 2017-01-23 | 2021-02-09 | Edwards Lifesciences Corporation | Expandable sheath |
| EP4649901A3 (en) | 2017-02-23 | 2026-01-21 | Boston Scientific Scimed, Inc. | Medical drain device |
| US10799685B2 (en) | 2017-03-09 | 2020-10-13 | Edwards Lifesciences Corporation | Expandable sheath with longitudinally extending reinforcing members |
| US10588636B2 (en) | 2017-03-20 | 2020-03-17 | Surefire Medical, Inc. | Dynamic reconfigurable microvalve protection device |
| US10398563B2 (en) | 2017-05-08 | 2019-09-03 | Medos International Sarl | Expandable cage |
| US11026719B2 (en) | 2017-05-15 | 2021-06-08 | Boston Scientific Scimed, Inc. | Radially expandable introducer sheath |
| US11344424B2 (en) | 2017-06-14 | 2022-05-31 | Medos International Sarl | Expandable intervertebral implant and related methods |
| US11160682B2 (en) | 2017-06-19 | 2021-11-02 | Covidien Lp | Method and apparatus for accessing matter disposed within an internal body vessel |
| US10940016B2 (en) | 2017-07-05 | 2021-03-09 | Medos International Sarl | Expandable intervertebral fusion cage |
| US10828065B2 (en) | 2017-08-28 | 2020-11-10 | Covidien Lp | Surgical access system |
| US10675056B2 (en) | 2017-09-07 | 2020-06-09 | Covidien Lp | Access apparatus with integrated fluid connector and control valve |
| CN111182856B (zh) | 2017-09-12 | 2022-04-29 | W.L.戈尔及同仁股份有限公司 | 用于假体瓣膜的瓣叶框架附连件 |
| EP3618723B1 (en) | 2017-09-15 | 2024-07-24 | INFRAREDEX, Inc. | Imaging catheter |
| CN111163728B (zh) | 2017-09-27 | 2022-04-29 | W.L.戈尔及同仁股份有限公司 | 具有机械联接的瓣叶的假体瓣膜 |
| CN111132636B (zh) | 2017-09-27 | 2022-04-08 | W.L.戈尔及同仁股份有限公司 | 带有可扩张框架的假体瓣膜以及相关系统和方法 |
| US11090153B2 (en) | 2017-10-13 | 2021-08-17 | W. L. Gore & Associates, Inc. | Telescoping prosthetic valve and delivery system |
| US11154397B2 (en) | 2017-10-31 | 2021-10-26 | W. L. Gore & Associates, Inc. | Jacket for surgical heart valve |
| EP3703615B1 (en) | 2017-10-31 | 2024-05-15 | W. L. Gore & Associates, Inc. | Transcatheter deployment systems and associated methods |
| US11439502B2 (en) | 2017-10-31 | 2022-09-13 | W. L. Gore & Associates, Inc. | Medical valve and leaflet promoting tissue ingrowth |
| US11123183B2 (en) | 2017-10-31 | 2021-09-21 | W. L. Gore & Associates, Inc. | Prosthetic heart valve |
| US10945765B2 (en) | 2017-12-06 | 2021-03-16 | Austin Miller Trauma LLC | Fixation clamp with spacer |
| EP3752236B1 (en) | 2018-02-15 | 2023-12-27 | Boston Scientific Scimed, Inc. | Introducer with expandable capabilities |
| US20190307487A1 (en) * | 2018-02-15 | 2019-10-10 | Brown University | Peel-away electrode placement catheter |
| US11957899B2 (en) | 2018-02-15 | 2024-04-16 | Brown University | Coupled annulus and catheter system for placement of biocompatible brain electrodes and like devices |
| KR20250069966A (ko) | 2018-03-07 | 2025-05-20 | 이너베이티브 카디오배스큘러 솔류션스, 엘엘씨 | 색전 보호 디바이스 |
| US11191532B2 (en) | 2018-03-30 | 2021-12-07 | Stryker European Operations Holdings Llc | Lateral access retractor and core insertion |
| WO2019199692A1 (en) | 2018-04-09 | 2019-10-17 | Edwards Lifesciences Corporation | Expandable sheath |
| KR102947035B1 (ko) | 2018-04-12 | 2026-04-02 | 로켓 사이언스 헬스 코퍼레이션 | 비강 내 약물 전달 기구 |
| JP2019187506A (ja) * | 2018-04-18 | 2019-10-31 | 大塚テクノ株式会社 | バルブアセンブリおよび尿道カテーテル |
| US11786695B2 (en) | 2018-07-25 | 2023-10-17 | Edwards Lifesciences Corporation | Methods of making an expandable sheath |
| US11850398B2 (en) | 2018-08-01 | 2023-12-26 | Trisalus Life Sciences, Inc. | Systems and methods for pressure-facilitated therapeutic agent delivery |
| EP3849646B1 (en) | 2018-09-10 | 2022-10-26 | Boston Scientific Scimed, Inc. | Introducer with expandable capabilities |
| EP4275629A3 (en) | 2018-09-20 | 2023-12-27 | Three Peaks Medical Pty Ltd | Adjustable sheath device |
| US11389193B2 (en) | 2018-10-02 | 2022-07-19 | Covidien Lp | Surgical access device with fascial closure system |
| US11338117B2 (en) | 2018-10-08 | 2022-05-24 | Trisalus Life Sciences, Inc. | Implantable dual pathway therapeutic agent delivery port |
| US11612413B2 (en) * | 2018-10-10 | 2023-03-28 | Merit Medical Systems, Inc. | Telescoping atrial septum needle |
| US11457949B2 (en) | 2018-10-12 | 2022-10-04 | Covidien Lp | Surgical access device and seal guard for use therewith |
| US11446156B2 (en) | 2018-10-25 | 2022-09-20 | Medos International Sarl | Expandable intervertebral implant, inserter instrument, and related methods |
| USD926322S1 (en) | 2018-11-07 | 2021-07-27 | W. L. Gore & Associates, Inc. | Heart valve cover |
| CA3119460A1 (en) * | 2018-11-15 | 2020-05-22 | Baleen Medical Llc | Methods, systems, and devices for embolic protection |
| US11166748B2 (en) | 2019-02-11 | 2021-11-09 | Covidien Lp | Seal assemblies for surgical access assemblies |
| US10792071B2 (en) | 2019-02-11 | 2020-10-06 | Covidien Lp | Seals for surgical access assemblies |
| US11497601B2 (en) | 2019-03-01 | 2022-11-15 | W. L. Gore & Associates, Inc. | Telescoping prosthetic valve with retention element |
| JP7381601B2 (ja) | 2019-04-12 | 2023-11-15 | エドワーズ ライフサイエンシーズ コーポレイション | マルチパートフレーム及び関連するレジリエントブリッジ特徴部を備えた弁 |
| US20200338316A1 (en) * | 2019-04-24 | 2020-10-29 | DePuy Synthes Products, Inc. | Vascular Access System |
| US11000313B2 (en) | 2019-04-25 | 2021-05-11 | Covidien Lp | Seals for surgical access devices |
| US11413068B2 (en) | 2019-05-09 | 2022-08-16 | Covidien Lp | Seal assemblies for surgical access assemblies |
| US12433597B2 (en) | 2019-06-04 | 2025-10-07 | Trisalus Life Sciences, Inc. | Atraumatic occlusive system with compartment for measurement of vascular pressure change |
| US11259840B2 (en) | 2019-06-21 | 2022-03-01 | Covidien Lp | Valve assemblies for surgical access assemblies |
| US11259841B2 (en) | 2019-06-21 | 2022-03-01 | Covidien Lp | Seal assemblies for surgical access assemblies |
| US11357542B2 (en) | 2019-06-21 | 2022-06-14 | Covidien Lp | Valve assembly and retainer for surgical access assembly |
| US11413065B2 (en) | 2019-06-28 | 2022-08-16 | Covidien Lp | Seal assemblies for surgical access assemblies |
| US11399865B2 (en) | 2019-08-02 | 2022-08-02 | Covidien Lp | Seal assemblies for surgical access assemblies |
| CN110604873B (zh) * | 2019-09-06 | 2025-12-30 | 无锡市人民医院 | 用于心脏传导束起搏的磁导航引导可撕开鞘管 |
| US11432843B2 (en) | 2019-09-09 | 2022-09-06 | Covidien Lp | Centering mechanisms for a surgical access assembly |
| US11523842B2 (en) | 2019-09-09 | 2022-12-13 | Covidien Lp | Reusable surgical port with disposable seal assembly |
| US11812991B2 (en) | 2019-10-18 | 2023-11-14 | Covidien Lp | Seal assemblies for surgical access assemblies |
| US11564674B2 (en) | 2019-11-27 | 2023-01-31 | K2M, Inc. | Lateral access system and method of use |
| US11464540B2 (en) | 2020-01-17 | 2022-10-11 | Covidien Lp | Surgical access device with fixation mechanism |
| US12324606B2 (en) | 2020-01-28 | 2025-06-10 | Covidien Lp | Seal assemblies for surgical access assemblies |
| AU2021216372B2 (en) * | 2020-02-04 | 2023-11-02 | Boston Scientific Scimed, Inc. | Devices, systems, and methods for an instrument accessory |
| US11576701B2 (en) | 2020-03-05 | 2023-02-14 | Covidien Lp | Surgical access assembly having a pump |
| US11426286B2 (en) | 2020-03-06 | 2022-08-30 | Eit Emerging Implant Technologies Gmbh | Expandable intervertebral implant |
| US11642153B2 (en) | 2020-03-19 | 2023-05-09 | Covidien Lp | Instrument seal for surgical access assembly |
| US11541218B2 (en) | 2020-03-20 | 2023-01-03 | Covidien Lp | Seal assembly for a surgical access assembly and method of manufacturing the same |
| US11446058B2 (en) | 2020-03-27 | 2022-09-20 | Covidien Lp | Fixture device for folding a seal member |
| US11717321B2 (en) | 2020-04-24 | 2023-08-08 | Covidien Lp | Access assembly with retention mechanism |
| US12127939B2 (en) * | 2020-04-30 | 2024-10-29 | Cephea Valve Technologies, Inc. | Catheter lumen lubricant |
| US11622790B2 (en) | 2020-05-21 | 2023-04-11 | Covidien Lp | Obturators for surgical access assemblies and methods of assembly thereof |
| US11751908B2 (en) | 2020-06-19 | 2023-09-12 | Covidien Lp | Seal assembly for surgical access assemblies |
| US11850160B2 (en) | 2021-03-26 | 2023-12-26 | Medos International Sarl | Expandable lordotic intervertebral fusion cage |
| US11752009B2 (en) | 2021-04-06 | 2023-09-12 | Medos International Sarl | Expandable intervertebral fusion cage |
| WO2022221869A1 (en) * | 2021-04-15 | 2022-10-20 | Retriever Medical, Inc. | Catheters with expandable and collapsible lumens |
| US11679195B2 (en) | 2021-04-27 | 2023-06-20 | Contego Medical, Inc. | Thrombus aspiration system and methods for controlling blood loss |
| EP4468973A1 (en) | 2022-01-27 | 2024-12-04 | Contego Medical, Inc. | Thrombectomy and aspiration system and methods of use |
| US12090064B2 (en) | 2022-03-01 | 2024-09-17 | Medos International Sarl | Stabilization members for expandable intervertebral implants, and related systems and methods |
| US20230364393A1 (en) * | 2022-05-12 | 2023-11-16 | Abiomed, Inc. | Expandable sheath and liner therefor |
| US12502178B2 (en) | 2022-08-16 | 2025-12-23 | Boston Scientific Scimed, Inc. | Medical device for occluding a left atrial appendage |
| WO2025090651A1 (en) * | 2023-10-23 | 2025-05-01 | TriReme Medical, LLC | Balloon catheter |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06197972A (ja) * | 1992-03-30 | 1994-07-19 | Pameda Nv | 迅速交換カテーテルシステム |
| JPH0751381A (ja) * | 1993-06-30 | 1995-02-28 | Cook Inc | 導入シース組立体装置 |
Family Cites Families (58)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US900726A (en) * | 1907-10-15 | 1908-10-13 | John H Gwynne | Wire-fence stretcher. |
| US4401433A (en) | 1980-06-13 | 1983-08-30 | Luther Ronald B | Apparatus for advancing oversized catheter through cannula, and the like |
| US4392853A (en) | 1981-03-16 | 1983-07-12 | Rudolph Muto | Sterile assembly for protecting and fastening an indwelling device |
| DE3117802A1 (de) | 1981-05-06 | 1982-11-25 | Max Dr. 8520 Erlangen Hubmann | Katheterbesteck |
| US4406656A (en) | 1981-06-01 | 1983-09-27 | Brack Gillium Hattler | Venous catheter having collapsible multi-lumens |
| DK447982A (da) * | 1982-10-11 | 1984-04-12 | Richard Bent Nissen | Fremgangsmaade til indfoering af et kateter eller en slange i en blodaare og instrumenter til udoevelse af fremgangsmaaden |
| US4569347A (en) | 1984-05-30 | 1986-02-11 | Advanced Cardiovascular Systems, Inc. | Catheter introducing device, assembly and method |
| US4710181A (en) | 1985-06-11 | 1987-12-01 | Genus Catheter Technologies, Inc. | Variable diameter catheter |
| US4601713A (en) | 1985-06-11 | 1986-07-22 | Genus Catheter Technologies, Inc. | Variable diameter catheter |
| US4738666A (en) | 1985-06-11 | 1988-04-19 | Genus Catheter Technologies, Inc. | Variable diameter catheter |
| US4798193A (en) | 1987-05-18 | 1989-01-17 | Thomas J. Fogarty | Protective sheath instrument carrier |
| US4921479A (en) | 1987-10-02 | 1990-05-01 | Joseph Grayzel | Catheter sheath with longitudinal seam |
| US5234425A (en) | 1989-03-03 | 1993-08-10 | Thomas J. Fogarty | Variable diameter sheath method and apparatus for use in body passages |
| US5015239A (en) | 1989-03-22 | 1991-05-14 | Browne Kevin F | Sheath introducer apparatus |
| US5176660A (en) * | 1989-10-23 | 1993-01-05 | Cordis Corporation | Catheter having reinforcing strands |
| US5209728B1 (en) | 1989-11-02 | 1998-04-14 | Danforth Biomedical Inc | Low profile high performance interventional catheters |
| US5201756A (en) | 1990-06-20 | 1993-04-13 | Danforth Biomedical, Inc. | Radially-expandable tubular elements for use in the construction of medical devices |
| US5256150A (en) | 1991-12-13 | 1993-10-26 | Endovascular Technologies, Inc. | Large-diameter expandable sheath and method |
| US6652492B1 (en) | 1991-12-13 | 2003-11-25 | Endovascular Technologies, Inc. | Dual valve, flexible sheath and method |
| US5935122A (en) * | 1991-12-13 | 1999-08-10 | Endovascular Technologies, Inc. | Dual valve, flexible expandable sheath and method |
| US5395349A (en) | 1991-12-13 | 1995-03-07 | Endovascular Technologies, Inc. | Dual valve reinforced sheath and method |
| US5304134A (en) | 1992-01-17 | 1994-04-19 | Danforth Biomedical, Inc. | Lubricious yet bondable catheter channel sleeve for over-the-wire catheters |
| US6312442B1 (en) * | 1992-06-02 | 2001-11-06 | General Surgical Innovations, Inc. | Method for developing an anatomic space for laparoscopic hernia repair |
| US6090072A (en) * | 1992-10-15 | 2000-07-18 | Scimed Life Systems, Inc. | Expandable introducer sheath |
| US5320611A (en) | 1993-02-04 | 1994-06-14 | Peter M. Bonutti | Expandable cannula having longitudinal wire and method of use |
| US5674240A (en) | 1993-02-04 | 1997-10-07 | Peter M. Bonutti | Expandable cannula |
| US5961499A (en) | 1993-02-04 | 1999-10-05 | Peter M. Bonutti | Expandable cannula |
| US6338730B1 (en) | 1993-02-04 | 2002-01-15 | Peter M. Bonutti | Method of using expandable cannula |
| US5431676A (en) | 1993-03-05 | 1995-07-11 | Innerdyne Medical, Inc. | Trocar system having expandable port |
| US5254084A (en) | 1993-03-26 | 1993-10-19 | Geary Gregory L | Peritoneal catheter device for dialysis |
| US6277107B1 (en) * | 1993-08-13 | 2001-08-21 | Daig Corporation | Guiding introducer for introducing medical devices into the coronary sinus and process for using same |
| US6159565A (en) | 1993-08-18 | 2000-12-12 | W. L. Gore & Associates, Inc. | Thin-wall intraluminal graft |
| US5824041A (en) | 1994-06-08 | 1998-10-20 | Medtronic, Inc. | Apparatus and methods for placement and repositioning of intraluminal prostheses |
| US5472418A (en) | 1994-07-28 | 1995-12-05 | Palestrant; Aubrey M. | Flattened collapsible vascular catheter |
| US5647857A (en) * | 1995-03-16 | 1997-07-15 | Endotex Interventional Systems, Inc. | Protective intraluminal sheath |
| US5902311A (en) * | 1995-06-15 | 1999-05-11 | Perclose, Inc. | Low profile intraluminal suturing device and method |
| US5868704A (en) | 1995-09-18 | 1999-02-09 | W. L. Gore & Associates, Inc. | Balloon catheter device |
| JP2000507119A (ja) | 1996-02-13 | 2000-06-13 | イマジン メディカル インコーポレイティド | 外科アクセス装置及び外科アクセス装置を構成する方法 |
| US5749889A (en) * | 1996-02-13 | 1998-05-12 | Imagyn Medical, Inc. | Method and apparatus for performing biopsy |
| US5997508A (en) | 1996-03-28 | 1999-12-07 | Medtronic, Inc. | Expandable percutaneous introducer sheath |
| US5735831A (en) | 1996-07-10 | 1998-04-07 | Cordis Corporation | Expandable flowrate catheter assembly and method of making same |
| US5827227A (en) | 1996-07-17 | 1998-10-27 | Delago; Augustin J. | Catheter having a radially adjustable sheath |
| US6007517A (en) | 1996-08-19 | 1999-12-28 | Anderson; R. David | Rapid exchange/perfusion angioplasty catheter |
| US6007522A (en) | 1996-09-13 | 1999-12-28 | Boston Scientific Corporation | Single operator exchange biliary catheter |
| US5944691A (en) | 1996-11-04 | 1999-08-31 | Cordis Corporation | Catheter having an expandable shaft |
| AU5621198A (en) | 1996-12-30 | 1998-07-31 | Imagyn Medical Technologies, Inc. | Expandable access device and method of constructing and using same |
| US5911702A (en) | 1997-11-06 | 1999-06-15 | Heartport, Inc. | Methods and devices for cannulating a patient's blood vessel |
| US6564101B1 (en) * | 1998-02-02 | 2003-05-13 | The Trustees Of Columbia University In The City Of New York | Electrical system for weight loss and laparoscopic implanation thereof |
| US6179827B1 (en) * | 1998-03-16 | 2001-01-30 | Chase Medical | Catheter having integral expandable/collapsible lumen |
| WO2000032264A1 (en) * | 1998-12-01 | 2000-06-08 | Chase Medical Inc. | Catheter having integral expandable/collapsible lumen |
| US6361528B1 (en) | 1999-04-05 | 2002-03-26 | Acist Medical Systems, Inc. | Dynamically compliant catheter |
| US20030065353A1 (en) | 2000-11-22 | 2003-04-03 | Danforth Biomedical, Inc, A California Corporation | Radially-expandable tubular elements for use in the construction of medical devices |
| JP4111829B2 (ja) * | 2001-01-11 | 2008-07-02 | リタ メディカル システムズ インコーポレイテッド | 骨処置器具 |
| US6899727B2 (en) | 2001-01-22 | 2005-05-31 | Gore Enterprise Holdings, Inc. | Deployment system for intraluminal devices |
| US7422579B2 (en) | 2001-05-01 | 2008-09-09 | St. Jude Medical Cardiology Divison, Inc. | Emboli protection devices and related methods of use |
| AU2003223749A1 (en) | 2002-04-25 | 2003-11-10 | The Board Of Trustees Of The Leland Stanford Junior University | Expandable guide sheath and apparatus and methods using such sheaths |
| US7329268B2 (en) | 2002-07-02 | 2008-02-12 | Warsaw Orthopedic, Inc. | Expandable percutaneous sheath |
| AU2003284976A1 (en) | 2002-10-25 | 2004-05-13 | Nmt Medical, Inc. | Expandable sheath tubing |
-
2003
- 2003-04-24 AU AU2003223749A patent/AU2003223749A1/en not_active Abandoned
- 2003-04-24 EP EP09156041A patent/EP2067499B1/en not_active Expired - Lifetime
- 2003-04-24 AT AT03719952T patent/ATE427768T1/de not_active IP Right Cessation
- 2003-04-24 US US10/423,321 patent/US7762995B2/en active Active
- 2003-04-24 DE DE60327067T patent/DE60327067D1/de not_active Expired - Lifetime
- 2003-04-24 EP EP03719952A patent/EP1496975B1/en not_active Expired - Lifetime
- 2003-04-24 JP JP2003587459A patent/JP4583032B2/ja not_active Expired - Lifetime
- 2003-04-24 WO PCT/US2003/013024 patent/WO2003090834A2/en not_active Ceased
-
2009
- 2009-03-18 US US12/406,897 patent/US9839771B2/en active Active
-
2010
- 2010-06-09 JP JP2010132478A patent/JP5487015B2/ja not_active Expired - Lifetime
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06197972A (ja) * | 1992-03-30 | 1994-07-19 | Pameda Nv | 迅速交換カテーテルシステム |
| JPH0751381A (ja) * | 1993-06-30 | 1995-02-28 | Cook Inc | 導入シース組立体装置 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016505309A (ja) * | 2012-12-18 | 2016-02-25 | コーニンクレッカ フィリップス エヌ ヴェKoninklijke Philips N.V. | 表面及び身体温度測定に対する再使用可能なmr安全温度プローブ |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2003223749A1 (en) | 2003-11-10 |
| US20090182278A1 (en) | 2009-07-16 |
| US7762995B2 (en) | 2010-07-27 |
| WO2003090834A2 (en) | 2003-11-06 |
| US20030233115A1 (en) | 2003-12-18 |
| EP1496975A2 (en) | 2005-01-19 |
| JP4583032B2 (ja) | 2010-11-17 |
| ATE427768T1 (de) | 2009-04-15 |
| JP2006500970A (ja) | 2006-01-12 |
| EP1496975B1 (en) | 2009-04-08 |
| EP2067499A1 (en) | 2009-06-10 |
| JP5487015B2 (ja) | 2014-05-07 |
| DE60327067D1 (de) | 2009-05-20 |
| EP2067499B1 (en) | 2012-05-16 |
| WO2003090834A3 (en) | 2004-02-19 |
| US9839771B2 (en) | 2017-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5487015B2 (ja) | 膨張可能なガイドシースおよび当該シースを使用する装置および方法 | |
| US7591832B2 (en) | Expandable guide sheath and apparatus with distal protection and methods for use | |
| US8388628B2 (en) | Expandable sheath for delivering instruments and agents into a body lumen and methods for use | |
| JP4691640B2 (ja) | 膨張可能なシースおよび装置ならびにその製造方法 | |
| US20200368053A1 (en) | Varying diameter vascular implant and balloon | |
| US7875049B2 (en) | Expandable guide sheath with steerable backbone and methods for making and using them | |
| US7744564B2 (en) | Apparatus and methods for delivering stem cells and other agents into cardiac tissue | |
| US7993350B2 (en) | Shapeable or steerable guide sheaths and methods for making and using them | |
| US20040116897A1 (en) | Minimally invasive bypass system and related methods | |
| US20050131447A1 (en) | Emboli protection devices and related methods of use | |
| EP2306939B1 (en) | Device delivery catheter having a curved distal tip | |
| CN111407479B (zh) | 用于覆盖穿刺部位的可旋转支架递送设备 | |
| CN111407480B (zh) | 用于覆盖穿刺部位的可膨胀支架递送设备 | |
| CN111407478B (zh) | 用于支架递送和定位以覆盖穿刺部位的方法 | |
| HK1152224B (en) | Device delivery catheter having a curved distal tip |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20100609 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120306 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120531 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130108 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130405 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130410 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130507 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130510 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20130607 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20130612 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130708 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20140204 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20140224 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5487015 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| EXPY | Cancellation because of completion of term |