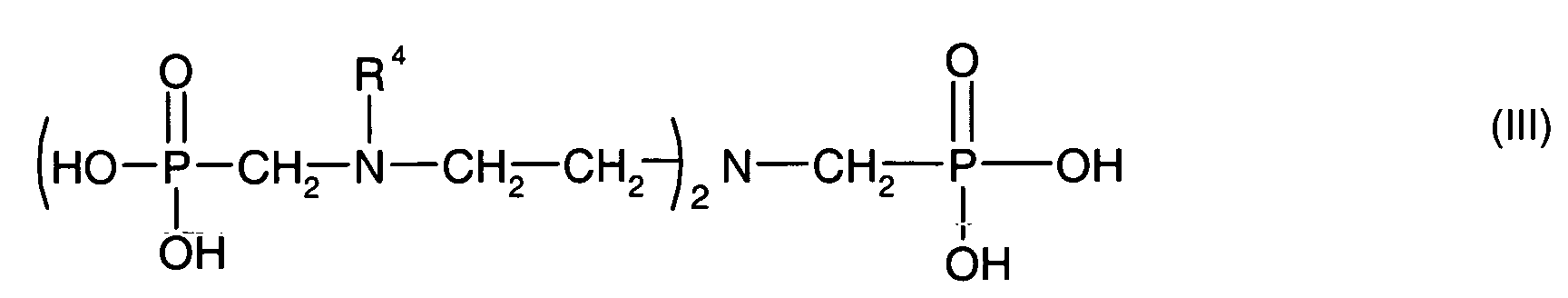EP1538261A1 - Process for flame-proofing of fibrous products - Google Patents
Process for flame-proofing of fibrous products Download PDFInfo
- Publication number
- EP1538261A1 EP1538261A1 EP03027959A EP03027959A EP1538261A1 EP 1538261 A1 EP1538261 A1 EP 1538261A1 EP 03027959 A EP03027959 A EP 03027959A EP 03027959 A EP03027959 A EP 03027959A EP 1538261 A1 EP1538261 A1 EP 1538261A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- component
- formula
- amino groups
- mixture
- weight
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/61—Polyamines polyimines
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/244—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus
- D06M13/282—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus with compounds containing phosphorus
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/244—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus
- D06M13/282—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus with compounds containing phosphorus
- D06M13/288—Phosphonic or phosphonous acids or derivatives thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/244—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus
- D06M13/282—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus with compounds containing phosphorus
- D06M13/292—Mono-, di- or triesters of phosphoric or phosphorous acids; Salts thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/322—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing nitrogen
- D06M13/325—Amines
- D06M13/332—Di- or polyamines
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H21/00—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties
- D21H21/14—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties characterised by function or properties in or on the paper
- D21H21/34—Ignifugeants
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/30—Flame or heat resistance, fire retardancy properties
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/03—Non-macromolecular organic compounds
- D21H17/05—Non-macromolecular organic compounds containing elements other than carbon and hydrogen only
- D21H17/10—Phosphorus-containing compounds
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/20—Macromolecular organic compounds
- D21H17/33—Synthetic macromolecular compounds
- D21H17/46—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D21H17/54—Synthetic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen
- D21H17/56—Polyamines; Polyimines; Polyester-imides
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H17/00—Non-fibrous material added to the pulp, characterised by its constitution; Paper-impregnating material characterised by its constitution
- D21H17/71—Mixtures of material ; Pulp or paper comprising several different materials not incorporated by special processes
- D21H17/74—Mixtures of material ; Pulp or paper comprising several different materials not incorporated by special processes of organic and inorganic material
Definitions
- the invention relates to a process for the flame-retardant finishing of fiber products.
- RM Rowell describes the chemical modification of lignocelluloses in "Proceedings, International Workshop on Frontiers of Surface Modification and Characterization of Lignocellulosic Fibers” (Sweden, May 30-31, 1996) (ISBN 91-7197-593-4).
- DE-A 30 03 648 and DE-A 42 44 194 describe the use of nitrogen-containing condensation products in papermaking.
- EP-A 542 071 describes wood preservatives which contain copper salts and which may additionally contain polyethyleneimine and / or phosphonic acid.
- the object of the present invention was to develop an improved method, to provide fiber products flame-retardant, in particular also fiber products with a high proportion of cellulose fibers produced by a wet process, Good flame retardant effects can be imparted.
- wet processes are namely the problems often greater than in dry processes, so that in known wet processes the danger is that the flame retardant component in the course of the preparation of the Fiber product is washed out. In this case, deterioration usually occurs the flame-retardant properties of the final article.
- fiber products are products which contain 20 to 100% by weight of cellulose fibers. This range of cellulose fiber content refers to the anhydrous fiber product.
- the fiber products may be finished products, such as paper, press plates (eg medium-density fibreboards or high-density fibreboards), which can be used in the furniture, packaging, construction and automotive industries.
- Such fiber or press plates often contain in addition to fibers still a fixing binder, wherein the fibers are the component which determines the strength of the press plates.
- the inventive method presented here is preferably used to achieve the flame-retardant properties of such fiberboard or press plates. For many uses of such press plates flame retardant properties are required.
- Cellulosic fibers may be blended with additives such as binder resins or hydrophobizing agents to make such fiberboards, optionally adding water to obtain an aqueous fiber suspension.
- the resulting mixture is shaped, dried and pressed into plates under the action of heat and pressure.
- a preferred embodiment is to carry it out during the manufacturing process of the finished fiber products, ie at a precursor of the finished fiber product.
- This precursor is preferably an aqueous suspension which contains cellulose fibers and optionally further additives, for example those of the type mentioned above.
- such fiber suspensions may be, for example, precursors in papermaking.
- aqueous fiber suspensions of cellulose fibers to fiberboard such as press plates or insulation boards
- the process of the invention is advantageously applicable, wherein the aqueous fiber suspension, for example a pulp (English Pulp) is treated with the components A and B.
- the fiber suspension is poured onto a filter screen, wherein a thin layer is formed, starting from which the finished fiber product is prepared by drying and pressing under heat and pressure.
- the said fiber suspension which contains cellulose fibers, water and optionally the abovementioned further components, is normally 0.3 to 15% by weight of cellulose fibers, preferably 0.5 to 1.5% by weight.
- This proportion of cellulose fibers must be such that the finished fiber product after removal of the water contains 20 to 100% by weight of cellulose fibers, based on the fiber product without water and without the components A and B. It is advantageous not to carry out the process according to the invention on the finished fiber product (fibreboard, paper) but on a precursor thereof.
- This precursor is a cellulosic fiber-containing product which is formed during the manufacture of the finished fiber product and processed into the finished fiber product.
- aqueous cellulose fiber suspensions are well suited as precursors for carrying out the process according to the invention.
- the inventive method not on the finished fiber product perform (although this is also possible in certain cases), but on a precursor of the type mentioned, is therefore advantageous because this normally a more effective flame retardancy is achieved. It is believed that this is because in this case better bonding of component A and / or B to the cellulosic fibers is achieved, favored by the subsequent action of heat and pressure.
- An advantageous embodiment of the method according to the invention is characterized in that the cellulose fibers of the fiber product are partly or completely present in the form of lignocellulose-containing fibers.
- Lignocellulose is a vegetable composite of cellulose, polyoses and lignin.
- a fiber product or, preferably, a precursor thereof is treated successively or simultaneously with a component A and a component B.
- a and B can be applied simultaneously, for example in the form of a mixture containing components A and B.
- this method is less preferred and in many cases not suitable. Rather, it is more advantageous to apply the components A and B successively, wherein it is further preferred to apply the component A (polyethyleneimine) earlier on the fiber product than component B (phosphonic acid). It has been found that in many cases with this approach, a more effective flame retardant effect can be achieved than with the other mentioned process variants. It has already been mentioned that it is preferable not to apply components A and B to the finished fiber product but to a precursor thereof.
- This precursor is preferably an aqueous suspension containing the cellulose fibers.
- component A and / or the component B are not applied in a pure form to the fiber product or its precursor, but in the form of a mixture. It is particularly advantageous if both component A and component B are applied in each case in the form of a mixture which contains component A or component B and additionally water.
- component A can be used in the form of a mixture containing 50 to 500 parts by weight of water per 100 parts by weight of component A, and component B in the form of a mixture containing 20 to 300 parts by weight of water per 100 parts by weight Component B contains.
- One or both of these mixtures may contain other components, for example polymaleic acid or partially hydrolyzed polymaleic anhydride.
- the addition of partially or fully hydrolyzed polymaleic anhydride is, if such an additive is used, preferably in the range of 1 to 5% by weight, based on total mixture containing component A or component B and water.
- polymaleic acid or partially hydrolyzed polymaleic anhydride is used, it is preferably added to a mixture containing component A and water. This addition causes, in a number of cases, an increase in the permanence of the flame retardant effect. This could be because the additional use of partially or completely hydrolyzed polymaleic anhydride leads to a better fixation of component A and / or component B on the fiber product.
- the fiber product or its precursor Contains 10 to 25% lignin, in addition to a partial ester of orthophosphoric acid on the Apply fiber product or its precursor.
- the application of this partial ester can simultaneously with the application of component A or component B or, which is preferred is to be done separately in a separate operation.
- the amount of orthophosphoric acid partial ester, which is applied is preferably in the range from 2 to 10% by weight, based on anhydrous fiber product or anhydrous precursor.
- a phosphoric acid partial ester include mono- or diesters of orthophosphoric acid having 6 to 12 carbon atoms suitable in the alcohol component of the ester, or mixtures of such mono- and diesters.
- An example of this is diisooctyl phosphate or diphenyl phosphate or bis (t-butyl-phenyl) phosphate.
- the addition of such esters is often the flame retardant effect increase.
- This is an advantage for cost and environmental reasons and also prevents the finished fiber products from being colored by metal ions are.
- component B in up to 50% of the bound to phosphorus Hydroxy groups are the hydrogen atoms by alkali metal or ammonium ions replaced, but this is not preferred.
- component A, component B or of a mixture which contains water other than component A or component B, to the fiber product or its precursor can be carried out by any desired methods. It is best to use an aqueous suspension containing cellulose fibers as a precursor and to apply to this precursor a mixture containing water and component A and then a mixture containing water and component B. Regardless of whether the components A and B are each applied as a mixture with water or in a pure form on the fiber product or the precursor, a preferred embodiment of the method according to the invention is that the weight ratio of the applied to the fiber product or its precursor amount Component A to the amount of applied component B is in the range of 1: 1.3 to 1: 4.0.
- the amount of component A and component B, which on the fiber product or its Pre-applied, is preferably such that on the finished fiber product 3 to 10% by weight of component A and 7 to 20% by weight of component B are present on anhydrous fiber product.
- Component A is a polyethyleneimine. As is customary with polymers, this is normally not a product which consists of all the same molecules, but is a mixture of products of different chain lengths.
- polyethyleneimines the fact known from the literature is added that under normal conditions there is a mixture of branched polymers whose individual molecules also differ in the number of branching units. This is expressed by the ratio of the number of secondary to primary amino groups as detailed below and to tertiary amino groups.
- Polyethyleneimines are products known from the literature. They can be prepared inter alia by reacting 1,2-ethylenediamine with 1,2-dichloroethane.
- polyethyleneimines which can be prepared by polymerization of unsubstituted aziridine (ethyleneimine). This polymerization can be carried out by known methods, if appropriate with addition of acidic catalysts, for example hydrochloric acid, and optionally in the presence of water.
- acidic catalysts for example hydrochloric acid
- Polyethyleneimines suitable for the process according to the invention are available on the market, for example from BASF, Germany (LUPASOL® grades and POLYMIN® grades) or from Nippon Shokubai Co. Ltd., Japan. US Pat. No. 6,451,961 B2 and US Pat. No. 5,977,293 describe polyethyleneimines and processes for their preparation.
- polyethyleneimines described there can be used for carrying out the process according to the invention, provided they meet the conditions mentioned above and in claim 1. Further, DA Tomalia et al in "Encyclopedia of Polymer Science and Engineering, Vol. 1, Wiley NY 1985, pages 680-739, describes suitable polyethyleneimines and methods for their preparation. Polyethyleneimines, their preparation and properties are also described in D. Horn, “Polyethyleneimine Physicochemical Properties and Applications”, “Polymeric Amines and Ammonium Salts", Goethals EJ, Pergamon Press: Oxford, New York 1980, pages 333-355 .
- the polyethyleneimines suitable as component A for the process according to the invention are branched. That is, the polymer having end groups of formula H 2 N-CH 2 -CH 2 - and within the polymer chain, units of the formula -CH 2 -CH 2 -NH-CH 2 -CH 2 -NH- additionally has units of the formula within the chain contains.
- the polymer thus contains primary, secondary and tertiary amino groups.
- the numerical ratios between the individual amino groups must assume values in a certain range.
- the ratio of the number of secondary amino groups to the number of primary amino groups must be in the range of 1.00: 1 to 2.50: 1, and the ratio of the number of secondary amino groups to the number of tertiary amino groups in the range of 1.20: 1 to 2.00: 1.
- the values for the numerical ratios of the various amino groups present in a particular polyethyleneimine or mixture of polyethylenimines can be determined by 13 C-NMR spectroscopy. This is discussed in T. St. Pierre and M. Geckle, 13 C-NMR Analysis of Branched Polyethyleneimines, J. Macromol SCI.-CHEM., Vol. A 22 (5-7), pages 877-887 ( 1985) ".
- Component A which, as is usual in polymers, is normally a mixture of polymers and consists of polyethyleneimine molecules of different molecular weight and different degrees of branching, has a weight average molecular weight in the range of 5,000 to 1,500,000, preferably in the range of 10,000 to 1,000,000.
- the individual value for this average molecular weight can be determined by methods known from the polymer literature, for example by gel permeation chromatography and detection by means of light scattering.
- the column used is one or more "PSS Suprema” types (available from “Polymer Standards Service GmbH”, Mainz, DE), which are adjusted to the intended molecular weight range; Eluant 1.5% formic acid in water; Multi-angle light scattering detector MALLS (also available from, among others, "Polymer Standards Service”); an internal standard may be used in addition.
- PSS Suprema available from “Polymer Standards Service GmbH”, Mainz, DE
- MALLS Multi-angle light scattering detector
- an internal standard may be used in addition.
- the values for the weight-average molecular weight referred to above and in claim 1 relate to this determination method.
- the average molecular weight of polyethyleneimines can be controlled by varying the parameters in their preparation.
- a preferred embodiment of the method according to the invention is that component A a Polyethyleneimine formed by polymerization of ethyleneimine and having the following structure (formula (V)) wherein the polymerization is optionally catalysed acid, wherein the individual units which contain tertiary amino groups and the individual units which contain secondary amino groups can be distributed as desired over the polymer chain, in which b is greater than a and wherein a and b have such values that the conditions given in claim 1 for the molecular weight and for the numerical ratios of Amino groups are satisfied with each other or wherein component A is a mixture of such polyethyleneimines.
- component A is normally a mixture of polyethylenimines.
- component A being a mixture of compounds of the formula (V).
- the values of a and b in the compounds of the formula (V) must, of course, be chosen so that the values for the ratios of the individual amino groups to one another and for the average molecular weight determined in the mixture are in the ranges stated above and in claim 1 , The control of these values can, as mentioned, be done via the parameters in the production of the polyethyleneimines.
- Component B is a phosphonic acid of the formula (I), of the formula (II) or of the formula (III)
- Component B may also be a mixture of compounds selected from compounds of formula (I), formula (II) and formula (III).
- R represents a linear or branched alkyl radical. This alkyl group contains 1 to 7 carbon atoms in the case that the below-mentioned R 1 is a hydroxy group. If R 1 is hydrogen, the radical R contains 3 to 7 carbon atoms.
- the radical R 1 in formula (I) is H or OH.
- the radical R 2 is the radical
- y can take the values 0, 1 or 2. Preferably, y has the value 0, which analogously to the case described above results in an increase in the phosphorus content on the fiber product.
- All radicals R 4 present in compounds of the formula (III) independently of one another represent hydrogen or or for a radical of the formula (IV)
- t is 0 or a number from 1 to 10.
- 50 to 100% of all radicals R 4 present are
- Phosphonic acids of formulas (I), (II) and (III) are commercially available products, e.g. Masquol P 210-1 Protex-Extrosa or Briquest 301-50 A from Rhodia or the Cublen products D50 (Zschimmer & Schwarz, DE), or Diquest 2060 S (Solutia, Belgium). Phosphonic acids of the formulas (I), (II) and (III) can be generally known from the literature Establish methods.
- a particularly advantageous embodiment of the process according to the invention is characterized in that component B is a mixture of phosphonic acids of the formula (I) and of the formula (II), both of which are present in completely unneutralized form.
- the mixing ratio of phosphonic acid of the formula (I) and phosphonic acid of the formula (II) may take any values.
- the weight ratio of the two types of phosphonic acid can assume values of 0: 100 to 100: 0.
- suspension 1 10 g of fiber raw material were slurried in 300 g of water at room temperature with stirring.
- the fiber raw material consisted of about 90% by weight of cellulose fibers and 10% by weight of lignin). Subsequently, this slurry was diluted with water with stirring to a total weight of 1050 g.
- suspension 2 10 g of a fiber raw material were slurried with 600 g of water with stirring. (This fiber raw material consisted of 70-75% by weight of cellulose fibers and 25-30% by weight of lignin).
- the products obtained from suspensions 1 and 2 were further treated as follows: First, the products were sucked through a suction filter and pressed while already a significant portion of the water was removed. Subsequently, some of the samples were pressed at room temperature and a pressure of 35 kp / cm 2 for 3 minutes and then dried at 120 ° C for 20 minutes and then conditioned at room temperature for 10 minutes. Some other samples were not pressed at room temperature but at elevated temperature. These samples were then no longer dried. The weight was then determined on all the samples thus obtained.
- DIOP di-iso-octyl phosphate
- the following table gives the quantities of suspension 1 or suspension 2 used, the amounts of components A and B used and optionally DIOP used, as well as the conditions of the pressing and drying process and the weight of the finished fibreboard.
- the firing time referred to in the right-hand column of the table as "BZ” represents a measure of the flame-retarding effect of the combination of component A and component B used in the process according to the invention.
- the “BZ” indicates the time in seconds during which the pattern still burns after being exposed to a flame for 15 seconds and then that flame is removed. A higher value for "BZ” thus means worse flame retardant properties of the sample.
- Patterns 3 to 7 show significantly better flame-retardant properties than Patterns 1 and 2 (non-inventive comparative experiments).
- a comparison of the patterns 6 shows and 7 that in the case of higher lignin contents (suspension 2) in the fiber suspension an additive from DIOP can bring a further improvement.
Abstract
Description
Die Erfindung betrifft ein Verfahren zur flammhemmenden Ausrüstung von Faserprodukten.The invention relates to a process for the flame-retardant finishing of fiber products.
Es ist bekannt, Faserprodukte wie z.B. Faserplatten oder -matten oder Vorstufen von Faserprodukten
mit bestimmten Produkten zu behandeln, um ihnen erwünschte Eigenschaften zu
verleihen. So beschreibt beispielsweise R.M. Rowell in "Proceedings, International Workshop
on Frontiers of Surface Modification and Characterization of Lignocellulosic Fibers" (Schweden
30. - 31. Mai 1996) (ISBN 91-7197-593-4) die chemische Modifizierung von Lignocellulosen.
Die DE-A 30 03 648 und DE-A 42 44 194 beschreiben die Verwendung stickstoffhaltiger Kondensationsprodukte
bei der Papierherstellung.
Die EP-A 542 071 beschreibt Holzschutzmittel, welche Kupfersalze enthalten und welche zusätzlich
Polyethylenimin und/oder Phosphonsäure enthalten können.
Aus S.C. Juneja, "Stable and Leach-Resistant Fire Retardants for Wood" in "Forest Products
Journal", Vol 22, Nr. 6 (1972) S. 17 - 23, geht die flammhemmende Behandlung von Holzmaterialien
hervor. S. Ishihara und T. Maku berichten in "Wood Research", No. 72 (1972) S. 72 bis
89 über die flammhemmende Behandlung von Holz und Filterpapier mit kationischen Produkten.It is known to treat fiber products such as fiberboard or mats or precursors of fiber products with certain products to give them desirable properties. For example, RM Rowell describes the chemical modification of lignocelluloses in "Proceedings, International Workshop on Frontiers of Surface Modification and Characterization of Lignocellulosic Fibers" (Sweden, May 30-31, 1996) (ISBN 91-7197-593-4).
DE-A 30 03 648 and DE-A 42 44 194 describe the use of nitrogen-containing condensation products in papermaking.
EP-A 542 071 describes wood preservatives which contain copper salts and which may additionally contain polyethyleneimine and / or phosphonic acid.
SC Juneja, "Stable and Leach-Resistant Fire Retardants for Wood" in "Forest Products Journal ", Vol. 22, No. 6 (1972) pp. 17-23, discloses the flame-retardant treatment of wood materials. S. Ishihara and T. Maku report in "Wood Research , No. 72 (1972) pp 72 to 89 on the flame retardant treatment of wood and filter paper with cationic products.
Die aus dem Stand der Technik bekannten Verfahren zur Behandlung von Faserprodukten sind nicht optimal, wenn es darum geht, Cellulosefasern enthaltende Produkte flammhemmend auszurüsten. Dies gilt insbesondere dann, wenn die Cellulose enthaltenden Faserprodukte nach einem Nassverfahren zu Faserplatten oder Fasermatten weiterverarbeitet werden. Vielfach lässt sich hierbei mit bekannten Verfahren eine genügende flammhemmende Ausrüstung nicht erzielen. Dies kann unter anderem daran liegen, dass es nicht gelingt, eine genügende Menge des flammhemmend wirksamen Produkts an das Fasermaterial zu binden. The methods known from the prior art for the treatment of fiber products are not optimal when it comes to equip flame retardant products containing cellulose fibers. This is especially true when the cellulose-containing fiber products after a wet process to fiberboard or fiber mats are further processed. Many times this can not be achieved with known methods sufficient flame retardant equipment. This may be due, among other things, to the failure to produce a sufficient amount of flame-retardant product to bind to the fiber material.
Der vorliegenden Erfindung lag die Aufgabe zugrunde, ein verbessertes Verfahren zu entwickeln, um Faserprodukte flammhemmend auszurüsten, wobei insbesondere auch Faserprodukten mit hohem Anteil an Cellulosefasern, die nach einem Nassverfahren hergestellt wurden, gute flammhemmende Wirkungen verliehen werden können. Bei Nassverfahren sind nämlich die Probleme oft größer als bei Trockenverfahren, sodaß bei bekannten Naßverfahren die Gefahr besteht, dass die flammhemmend wirksame Komponente im Verlauf der Herstellung des Faserprodukts ausgewaschen wird. In diesem Fall tritt normalerweise eine Verschlechterung der flammhemmenden Eigenschaften des Endartikels ein.The object of the present invention was to develop an improved method, to provide fiber products flame-retardant, in particular also fiber products with a high proportion of cellulose fibers produced by a wet process, Good flame retardant effects can be imparted. For wet processes are namely the problems often greater than in dry processes, so that in known wet processes the danger is that the flame retardant component in the course of the preparation of the Fiber product is washed out. In this case, deterioration usually occurs the flame-retardant properties of the final article.
Die Aufgabe wurde gelöst durch ein Verfahren zur flammhemmenden Ausrüstung eines Faserprodukts,
welches 20 bis 100 Gew% Cellulosefasern enthält, bezogen auf das Gewicht des
wasserfreien Faserprodukts, wobei man das Faserprodukt oder vorzugsweise eine Vorstufe
davon nacheinander oder gleichzeitig mit einer Komponente A und einer Komponente B behandelt,
wobei Komponente A ein verzweigtes Polyethylenimin ist, das primäre, sekundäre und
tertiäre Aminogruppen enthält und das ein Gewichtsmittel des Molgewichts im Bereich von
5.000 bis 1.500.000, vorzugsweise 10.000 bis 1.000.000, besitzt und bei dem das zahlenmäßige
Verhältnis von sekundären Aminogruppen zu primären Aminogruppen im Bereich von
1,00 : 1 bis 2,50 : 1 und das zahlenmäßige Verhältnis von sekundären Aminogruppen zu tertiären
Aminogruppen im Bereich von 1,20 : 1 bis 2,00 : 1 liegt,
oder wobei Komponente A ein Gemisch solcher Polyethyleninime ist,
wobei Komponente B eine Phosphonsäure der Formel (I), (II) oder der Formel (III) ist
wobei in den Formeln (I), (II) oder (III) in bis zu 50 % der an Phosphor gebundenen OH-Gruppen
das Wasserstoffatom durch ein Alkalimetall oder eine Ammoniumgruppe substituiert
sein kann, wobei jedoch vorzugsweise 100 % dieser OH-Gruppen in nicht-neutralisierter Form
vorliegen,
oder wobei Komponente B ein Gemisch von Verbindungen ist, die aus Verbindungen der Formel
(I), (II) oder (III) ausgewählt sind,
wobei
wobei R3 für H oder R2, vorzugsweise für R2 steht und wobei
alle Reste R4 unabhängig voneinander für H oder für oder für einen Rest der Formel (IV) stehen wobei es bevorzugt ist, wenn 50 bis 100 % aller anwesenden Reste R4 für stehen,
wobei t 0 oder eine Zahl von 1 bis 10 ist.The object has been achieved by a process for the flame-retardant finishing of a fiber product containing from 20 to 100% by weight of cellulose fibers based on the weight of the anhydrous fiber product, wherein the fiber product or preferably a precursor thereof is successively or simultaneously with a component A and a component B wherein component A is a branched polyethylenimine containing primary, secondary and tertiary amino groups and having a weight average molecular weight in the range of 5,000 to 1,500,000, preferably 10,000 to 1,000,000, and in which the numerical ratio of secondary amino groups to primary amino groups in the range of 1.00: 1 to 2.50: 1 and the ratio of secondary amino groups to tertiary amino groups ranges from 1.20: 1 to 2.00: 1, or wherein component A is a mixture of such Polyethylene inime is,
wherein component B is a phosphonic acid of formula (I), (II) or formula (III) wherein in the formulas (I), (II) or (III) in up to 50% of the phosphorus bound OH groups, the hydrogen atom may be substituted by an alkali metal or an ammonium group, but preferably 100% of these OH groups in non neutralized form,
or wherein component B is a mixture of compounds selected from compounds of formula (I), (II) or (III),
in which
wherein R 3 is H or R 2 , preferably R 2 and wherein
all radicals R 4 independently of one another for H or for or represents a radical of the formula (IV) wherein it is preferred that 50 to 100% of all radicals R 4 present for stand,
where t is 0 or a number from 1 to 10.
Im Rahmen der hier beschriebenen Erfindung werden unter Faserprodukten Produkte verstanden,
welche 20 bis 100 Gewichts-% Cellulosefasern enthalten. Dieser Bereich für den Gehalt
an Cellulosefasern bezieht sich auf das wasserfreie Faserprodukt. Die Faserprodukte können
Fertigprodukte sein, wie z.B. Papier, Pressplatten (z.B. Medium-Density-Fibreboards oder High-Density-Fibreboards),
welche in der Möbel-, Verpackungsindustrie, im Baugewerbe und im Automobilbau
eingesetzt werden können. Solche Faser- bzw. Pressplatten enthalten häufig neben
Fasern noch ein fixierendes Bindemittel, wobei die Fasern die Komponente sind, welche die
Festigkeit der Pressplatten bestimmt. Das hier vorgestellte erfindungsgemäße Verfahren wird
vorzugsweise zur Erzielung flammhemmender Eigenschaften solcher Faserplatten bzw. Pressplatten
verwendet. Für viele Verwendungszwecke solcher Pressplatten werden flammhemmende
Eigenschaften gefordert. Zur Herstellung solcher Faserplatten können Cellulosefasern mit
Zusatzmitteln wie Bindeharzen oder Hydrophobiermitteln vermischt werden, gegebenenfalls
wird Wasser hinzugefügt, um eine wässrige Fasersuspension zu erhalten. Das so erhaltene
Gemisch wird geformt, getrocknet und unter Einwirkung von Hitze und Druck zu Platten verpresst.
Obwohl prinzipiell das erfindungsgemäße Verfahren auch am fertigen Endprodukt durchgeführt
werden kann, besteht eine bevorzugte Ausführungsform darin, es während des Herstellungsprozesses
der fertigen Faserprodukte durchzuführen, also an einer Vorstufe des fertigen Faserprodukts.
Diese Vorstufe ist vorzugsweise eine wässrige Suspension, welche Cellulosefasern
und gegebenenfalls weitere Zusätze z.B. solche der oben erwähnten Art enthält. Solche Fasersuspensionen
können im Zusammenhang mit dem erfindungsgemäßen Verfahren beispielsweise
Vorstufen bei der Papierherstellung sein. Vorzugsweise sind sie aber Vorstufen bei der Herstellung
von Faserplatten oder Fasermatten.
Die Verarbeitung solcher wässrigen Suspensionen von Cellulosefasern zu Faserplatten, z.B.
Pressplatten oder Dämmplatten erfolgt nach dem sogenannten Nassverfahren. Insbesondere
bei Nassverfahren dieser Art ist das erfindungsgemäße Verfahren vorteilhaft anwendbar, wobei
die wässrige Fasersuspension, z.B. eine Pulpe (engl. pulp) mit den Komponenten A und B behandelt
wird. Hierbei wird beispielsweise die Fasersuspension auf ein Filtersieb gegossen, wobei
eine dünne Schicht gebildet wird, ausgehend von der das fertige Faserprodukt durch Trocknen
und Pressen unter Hitze- und Druckeinwirkung hergestellt wird.
Die genannte Fasersuspension (Vorstufe), die Cellulosefasern, Wasser und gegebenenfalls die
oben genannten weiteren Komponenten enthält, besteht im Normalfall zu 0,3 bis 15 Gew% aus
Cellulosefasern, vorzugsweise zu 0,5 bis 1,5 Gew%. Dieser Anteil an Cellulosefasern muß so
bemessen sein, dass das fertige Faserprodukt nach Entfernung des Wassers 20 bis 100 Gew%
Cellulosefasern enthält, bezogen auf das Faserprodukt ohne Wasser und ohne die Komponenten
A und B.
Es ist vorteilhaft, nicht an dem fertigen Faserprodukt (Faserplatte, Papier) das erfindungsgemäße
Verfahren durchzuführen, sondern an einer Vorstufe davon. Diese Vorstufe ist ein Cellulosefasern-enthaltendes
Produkt, das während der Herstellung des fertigen Faserprodukts gebildet
und zu dem fertigen Faserprodukt weiterverarbeitet wird. Insbesondere sind wässrige Cellulosefasersuspensionen
als Vorprodukte für die Durchführung des erfindungsgemäßen Verfahren gut
geeignet.
Das erfindungsgemäße Verfahren nicht am fertigen Faserprodukt durchzuführen (obwohl dies
auch in gewissen Fällen möglich ist), sondern an einem Vorprodukt der genannten Art, ist deshalb
von Vorteil, weil hierdurch im Normalfall ein effektiverer Flammschutz erreicht wird. Man
nimmt an, dass dies daran liegt, dass in diesem Fall eine bessere Bindung der Komponente A
und/oder B an die Cellulosefasern erreicht wird, begünstigt durch die nachfolgende Einwirkung
von Hitze und Druck.
Eine vorteilhafte Ausführungsform des erfindungsgemäßen Verfahrens ist dadurch gekennzeichnet,
dass die Cellulosefasern des Faserprodukts zum Teil oder vollständig in Form von
Lignocellulose-enthaltenden Fasern vorliegen. Lignocellulose ist ein pflanzlicher Materialverbund
aus Cellulose, Polyosen und Lignin.
Die chemische Zusammensetzung von Lignocellulose-enthaltenden Fasern ist beschrieben in
der eingangs genannten Literaturstelle (R.M. Rowell, ISBN 91-7197-593-4), 2. Seite, "Features
of Lignocellulosics", ferner in der EP-A 406 783.In the context of the invention described here, fiber products are products which contain 20 to 100% by weight of cellulose fibers. This range of cellulose fiber content refers to the anhydrous fiber product. The fiber products may be finished products, such as paper, press plates (eg medium-density fibreboards or high-density fibreboards), which can be used in the furniture, packaging, construction and automotive industries. Such fiber or press plates often contain in addition to fibers still a fixing binder, wherein the fibers are the component which determines the strength of the press plates. The inventive method presented here is preferably used to achieve the flame-retardant properties of such fiberboard or press plates. For many uses of such press plates flame retardant properties are required. Cellulosic fibers may be blended with additives such as binder resins or hydrophobizing agents to make such fiberboards, optionally adding water to obtain an aqueous fiber suspension. The resulting mixture is shaped, dried and pressed into plates under the action of heat and pressure.
Although in principle the method according to the invention can also be carried out on the finished end product, a preferred embodiment is to carry it out during the manufacturing process of the finished fiber products, ie at a precursor of the finished fiber product. This precursor is preferably an aqueous suspension which contains cellulose fibers and optionally further additives, for example those of the type mentioned above. In the context of the process according to the invention, such fiber suspensions may be, for example, precursors in papermaking. Preferably, however, they are precursors in the production of fiberboard or fiber mats.
The processing of such aqueous suspensions of cellulose fibers to fiberboard, such as press plates or insulation boards is carried out by the so-called wet process. In particular, in wet processes of this type, the process of the invention is advantageously applicable, wherein the aqueous fiber suspension, for example a pulp (English Pulp) is treated with the components A and B. Here, for example, the fiber suspension is poured onto a filter screen, wherein a thin layer is formed, starting from which the finished fiber product is prepared by drying and pressing under heat and pressure.
The said fiber suspension (precursor), which contains cellulose fibers, water and optionally the abovementioned further components, is normally 0.3 to 15% by weight of cellulose fibers, preferably 0.5 to 1.5% by weight. This proportion of cellulose fibers must be such that the finished fiber product after removal of the water contains 20 to 100% by weight of cellulose fibers, based on the fiber product without water and without the components A and B.
It is advantageous not to carry out the process according to the invention on the finished fiber product (fibreboard, paper) but on a precursor thereof. This precursor is a cellulosic fiber-containing product which is formed during the manufacture of the finished fiber product and processed into the finished fiber product. In particular, aqueous cellulose fiber suspensions are well suited as precursors for carrying out the process according to the invention.
The inventive method not on the finished fiber product perform (although this is also possible in certain cases), but on a precursor of the type mentioned, is therefore advantageous because this normally a more effective flame retardancy is achieved. It is believed that this is because in this case better bonding of component A and / or B to the cellulosic fibers is achieved, favored by the subsequent action of heat and pressure.
An advantageous embodiment of the method according to the invention is characterized in that the cellulose fibers of the fiber product are partly or completely present in the form of lignocellulose-containing fibers. Lignocellulose is a vegetable composite of cellulose, polyoses and lignin.
The chemical composition of lignocellulose-containing fibers is described in the cited reference (RM Rowell, ISBN 91-7197-593-4), 2nd page, "Features of Lignocellulosics" , further in EP-A 406 783 .
Bei dem erfindungsgemäßen Verfahren wird ein Faserprodukt oder, was bevorzugt ist, eine
Vorstufe davon, nacheinander oder gleichzeitig mit einer Komponente A und einer Komponente
B behandelt. Es können also A und B gleichzeitig aufgebracht werden, z.B. in Form eines Gemischs,
das die Komponenten A und B enthält. Diese Methode ist jedoch weniger bevorzugt
und sogar in vielen Fällen nicht geeignet. Vorteilhafter ist es vielmehr, die Komponenten A und
B nacheinander aufzubringen, wobei es ferner bevorzugt ist, die Komponente A (Polyethylenimin)
früher auf das Faserprodukt aufzubringen als Komponente B (Phosphonsäure). Es hat sich
nämlich herausgestellt, dass in vielen Fällen mit dieser Vorgehensweise eine effektivere flammhemmende
Wirkung erzielt werden kann als mit den anderen genannten Verfahrensvarianten.
Es wurde bereits erwähnt, dass es bevorzugt ist, die Komponenten A und B nicht auf das fertige
Faserprodukt aufzubringen, sondern auf eine Vorstufe davon. Diese Vorstufe ist vorzugsweise
eine wässrige Suspension, welche die Cellulosefasern enthält.
In analoger Weise ist es häufig von Vorteil, wenn man die Komponente A und/oder die Komponente
B nicht in reiner Form auf das Faserprodukt oder dessen Vorstufe aufbringt, sondern in
Form eines Gemischs. Besonders günstig ist es, wenn man sowohl Komponente A als auch
Komponente B jeweils in Form eines Gemischs aufbringt, das Komponente A bzw. Komponente
B und zusätzlich Wasser enthält. So kann man Komponente A beispielsweise in Form eines
Gemischs einsetzen, das 50 bis 500 Gew.teile Wasser pro 100 Gew.teile Komponente A enthält,
und Komponente B in Form eines Gemischs, das 20 bis 300 Gew.teile Wasser pro 100
Gew.teile Komponente B enthält. Eines oder beide dieser Gemische können weitere Komponenten
enthalten, z.B. Polymaleinsäure oder teilhydrolysiertes Polymaleinsäureanhydrid. Der
Zusatz von teil- oder vollhydrolysiertem Polymaleinsäureanhydrid liegt, wenn ein solcher Zusatz
verwendet wird, bevorzugt im Bereich von 1 bis 5 Gew%, bezogen auf Gesamtgemisch, das
Komponente A bzw. Komponente B und Wasser enthält.
Wenn Polymaleinsäure oder teilhydrolysiertes Polymaleinsäureanhydrid verwendet werden,
setzt man dieses vorzugsweise einem Gemisch zu, das Komponente A und Wasser enthält.
Dieser Zusatz bewirkt in einer Reihe von Fällen eine Erhöhung der Permanenz des flammhemmenden
Effekts. Dies könnte daran liegen, dass die zusätzliche Verwendung von teilweise oder
vollständig hydrolysiertem Polymaleinsäureanhydrid zu einer besseren Fixierung der Komponente
A und/oder Komponente B auf dem Faserprodukt führt.In the process according to the invention, a fiber product or, preferably, a precursor thereof, is treated successively or simultaneously with a component A and a component B. Thus, A and B can be applied simultaneously, for example in the form of a mixture containing components A and B. However, this method is less preferred and in many cases not suitable. Rather, it is more advantageous to apply the components A and B successively, wherein it is further preferred to apply the component A (polyethyleneimine) earlier on the fiber product than component B (phosphonic acid). It has been found that in many cases with this approach, a more effective flame retardant effect can be achieved than with the other mentioned process variants. It has already been mentioned that it is preferable not to apply components A and B to the finished fiber product but to a precursor thereof. This precursor is preferably an aqueous suspension containing the cellulose fibers.
In an analogous manner, it is often advantageous if the component A and / or the component B are not applied in a pure form to the fiber product or its precursor, but in the form of a mixture. It is particularly advantageous if both component A and component B are applied in each case in the form of a mixture which contains component A or component B and additionally water. For example, component A can be used in the form of a mixture containing 50 to 500 parts by weight of water per 100 parts by weight of component A, and component B in the form of a mixture containing 20 to 300 parts by weight of water per 100 parts by weight Component B contains. One or both of these mixtures may contain other components, for example polymaleic acid or partially hydrolyzed polymaleic anhydride. The addition of partially or fully hydrolyzed polymaleic anhydride is, if such an additive is used, preferably in the range of 1 to 5% by weight, based on total mixture containing component A or component B and water.
When polymaleic acid or partially hydrolyzed polymaleic anhydride is used, it is preferably added to a mixture containing component A and water.
This addition causes, in a number of cases, an increase in the permanence of the flame retardant effect. This could be because the additional use of partially or completely hydrolyzed polymaleic anhydride leads to a better fixation of component A and / or component B on the fiber product.
Ferner kann es von Vorteil sein, insbesondere dann, wenn das Faserprodukt oder dessen Vorstufe 10 bis 25 % Lignin enthält, zusätzlich einen Teilester der Orthophosphorsäure auf das Faserprodukt oder dessen Vorstufe aufzubringen. Das Aufbringen dieses Teilesters kann gleichzeitig mit dem Aufbringen der Komponente A oder der Komponente B oder, was bevorzugt ist, getrennt davon in einem separaten Arbeitsgang erfolgen. Die Menge an Orthophosphorsäureteilester, die aufgebracht wird, liegt bevorzugt im Bereich von 2 bis 10 Gew%, bezogen auf wasserfreies Faserprodukt bzw. wasserfreie Vorstufe. Als Phosphorsäureteilester sind unter anderem Mono- oder Diester der Orthophosphorsäure mit 6 bis 12 Kohlenstoffatomen in der Alkoholkomponente des Esters geeignet, oder Gemische solcher Mono- und Diester. Ein Beispiel hierfür ist Di-isooctyl-phosphat oder Diphenylphosphat oder Bis-(t-butyl-phenyl-) phosphat. Durch den Zusatz solcher Ester lässt sich vielfach die flammhemmende Wirkung steigern.Furthermore, it may be advantageous, especially if the fiber product or its precursor Contains 10 to 25% lignin, in addition to a partial ester of orthophosphoric acid on the Apply fiber product or its precursor. The application of this partial ester can simultaneously with the application of component A or component B or, which is preferred is to be done separately in a separate operation. The amount of orthophosphoric acid partial ester, which is applied is preferably in the range from 2 to 10% by weight, based on anhydrous fiber product or anhydrous precursor. As a phosphoric acid partial ester include mono- or diesters of orthophosphoric acid having 6 to 12 carbon atoms suitable in the alcohol component of the ester, or mixtures of such mono- and diesters. An example of this is diisooctyl phosphate or diphenyl phosphate or bis (t-butyl-phenyl) phosphate. The addition of such esters is often the flame retardant effect increase.
Vorzugsweise enthalten weder Komponente A noch Komponente B noch die Gemische aus Komponente A oder Komponente B und Wasser Metalle oder Metallverbindungen, abgesehen von unwesentlichen Verunreinigungen. Dies ist ein Vorteil aus Kosten- und aus Umweltgründen und außerdem wird dadurch vermieden, dass die fertigen Faserprodukte durch Metallionen gefärbt sind. Gegebenenfalls können zwar in Komponente B in bis zu 50 % der an Phosphor gebundenen Hydroxygruppen die Wasserstoffatome durch Alkalimetall- oder Ammoniumionen ersetzt sein, jedoch ist dies nicht bevorzugt. Preferably neither component A nor component B nor contain the mixtures Component A or Component B and water metals or metal compounds, except of negligible impurities. This is an advantage for cost and environmental reasons and also prevents the finished fiber products from being colored by metal ions are. Optionally, although in component B in up to 50% of the bound to phosphorus Hydroxy groups are the hydrogen atoms by alkali metal or ammonium ions replaced, but this is not preferred.
Das Aufbringen von Komponente A, Komponente B oder von einem Gemisch, das außer Komponente
A oder Komponente B noch Wasser enthält, auf das Faserprodukt oder dessen Vorstufe
kann nach beliebigen Methoden erfolgen. Am günstigsten ist es, eine wässrige Suspension,
die Cellulosefasern enthält, als Vorstufe zu verwenden und auf diese Vorstufe ein Gemisch zu
applizieren, das Wasser und Komponente A enthält und anschließend ein Gemisch, das Wasser
und Komponente B enthält.
Unabhängig davon, ob die Komponenten A und B jeweils als Gemisch mit Wasser oder in reiner
Form auf das Faserprodukt oder die Vorstufe aufgebracht werden, besteht eine bevorzugte
Ausführungsform des erfindungsgemäßen Verfahrens darin, dass das Gewichtsverhältnis der
auf das Faserprodukt oder auf dessen Vorstufe aufgebrachten Menge an Komponente A zur
Menge an aufgebrachter Komponente B im Bereich von 1 : 1,3 bis 1 : 4,0 liegt.The application of component A, component B or of a mixture which contains water other than component A or component B, to the fiber product or its precursor can be carried out by any desired methods. It is best to use an aqueous suspension containing cellulose fibers as a precursor and to apply to this precursor a mixture containing water and component A and then a mixture containing water and component B.
Regardless of whether the components A and B are each applied as a mixture with water or in a pure form on the fiber product or the precursor, a preferred embodiment of the method according to the invention is that the weight ratio of the applied to the fiber product or its precursor amount Component A to the amount of applied component B is in the range of 1: 1.3 to 1: 4.0.
Die Menge an Komponente A und an Komponente B, die auf das Faserprodukt oder dessen Vorstufe aufgebracht werden, wird vorzugsweise so bemessen, dass auf dem fertigen Faserprodukt 3 bis 10 Gew% Komponente A und 7 bis 20 Gew% Komponente B vorliegen, bezogen auf wasserfreies Faserprodukt.The amount of component A and component B, which on the fiber product or its Pre-applied, is preferably such that on the finished fiber product 3 to 10% by weight of component A and 7 to 20% by weight of component B are present on anhydrous fiber product.
Die Komponente A ist ein Polyethylenimin. Wie bei Polymeren üblich, handelt es sich hierbei im
Normalfall nicht um ein Produkt, das aus lauter gleichen Molekülen besteht, sondern das ein
Gemisch aus Produkten unterschiedlicher Kettenlänge ist. Bei Polyethyleniminen kommt noch
die aus der Literatur bekannte Tatsache hinzu, dass im Normalfall ein Gemisch verzweigter
Polymerer vorliegt, dessen einzelne Moleküle sich auch noch in der Anzahl der Verzweigungseinheiten
unterscheiden. Dies wird durch das unten näher erläuterte Verhältnis der Anzahl von
sekundären zu primären Aminogruppen und zu tertiären Aminogruppen ausgedrückt.
Polyethylenimine sind aus der Literatur bekannte Produkte. Sie können unter anderem durch
Umsetzung von 1.2-Ethylendiamin mit 1.2-Dichlorethan hergestellt werden. Zur Durchführung
des erfindungsgemäßen Verfahrens verwendet man bevorzugt Polyethylenimine, die durch Polymerisation
von unsubstituierten Aziridin (Ethylenimin) hergestellt werden können. Diese Polymerisation
kann nach bekannten Methoden, gegebenenfalls unter Zusatz saurer Katalysatoren,
z.B. Salzsäure, und gegebenenfalls in Gegenwart von Wasser durchgeführt werden.
Für das erfindungsgemäße Verfahren geeignete Polyethylenimine sind auf dem Markt erhältlich,
z.B. von der Firma BASF, Deutschland (LUPASOL®-Typen und POLYMIN®-Typen) oder
von der Firma Nippon Shokubai Co. Ltd., Japan.
Die US 6 451 961 B2, und die US 5 977 293 beschreiben Polyethylenimine und Verfahren zu
deren Herstellung. Die dort beschriebenen Polyethylenimine können zur Durchführung des erfindungsgemäßen
Verfahrens verwendet werden, sofern sie die oben und in Anspruch 1 genannten
Bedingungen erfüllen. Ferner beschreiben D.A. Tomalia et al in "Encyclopedia of Polymer
Science and Engineering, Vol. 1, Wiley N.Y. 1985, Seiten 680 - 739, geeignete Polyethylenimine
und Verfahren zu ihrer Herstellung.
Polyethylenimine, ihre Herstellung und Eigenschaften werden auch in D. Horn, "Polyethylenimine-Physicochemical
Properties and Applications, in "Polymeric Amines and Ammonium
Salts", Goethals E.J., Pergamon Press: Oxford, New York 1980, Seiten 333 - 355, beschrieben. Component A is a polyethyleneimine. As is customary with polymers, this is normally not a product which consists of all the same molecules, but is a mixture of products of different chain lengths. In the case of polyethyleneimines, the fact known from the literature is added that under normal conditions there is a mixture of branched polymers whose individual molecules also differ in the number of branching units. This is expressed by the ratio of the number of secondary to primary amino groups as detailed below and to tertiary amino groups. Polyethyleneimines are products known from the literature. They can be prepared inter alia by reacting 1,2-ethylenediamine with 1,2-dichloroethane. For carrying out the process according to the invention it is preferred to use polyethyleneimines which can be prepared by polymerization of unsubstituted aziridine (ethyleneimine). This polymerization can be carried out by known methods, if appropriate with addition of acidic catalysts, for example hydrochloric acid, and optionally in the presence of water.
Polyethyleneimines suitable for the process according to the invention are available on the market, for example from BASF, Germany (LUPASOL® grades and POLYMIN® grades) or from Nippon Shokubai Co. Ltd., Japan.
US Pat. No. 6,451,961 B2 and US Pat. No. 5,977,293 describe polyethyleneimines and processes for their preparation. The polyethyleneimines described there can be used for carrying out the process according to the invention, provided they meet the conditions mentioned above and in claim 1. Further, DA Tomalia et al in "Encyclopedia of Polymer Science and Engineering, Vol. 1, Wiley NY 1985, pages 680-739, describes suitable polyethyleneimines and methods for their preparation.
Polyethyleneimines, their preparation and properties are also described in D. Horn, "Polyethyleneimine Physicochemical Properties and Applications", "Polymeric Amines and Ammonium Salts", Goethals EJ, Pergamon Press: Oxford, New York 1980, pages 333-355 .
Die für das erfindungsgemäße Verfahren als Komponente A geeigneten Polyethylenimine sind
verzweigt. Das heißt, dass das Polymer, welches Endgruppen der Formel
Das Polymer enthält also primäre, sekundäre und tertiäre Aminogruppen.
Damit die Durchführung des erfindungsgemäßen Verfahrens gute Effekte bezüglich flammhemmender
Eigenschaften der Faserprodukte liefert, müssen die zahlenmäßigen Verhältnisse
zwischen den einzelnen Aminogruppen Werte in einem bestimmten Bereich annehmen. So
muß in Komponente A das Verhältnis der Anzahl sekundärer Aminogruppen zur Anzahl primärer
Aminogruppen im Bereich von 1,00 : 1 bis 2,50 : 1 liegen, und das Verhältnis der Anzahl
sekundärer Aminogruppen zur Anzahl tertiärer Aminogruppen im Bereich von 1,20 : 1 bis
2,00 : 1. Diese zahlenmäßigen Werte lassen sich steuern über die Parameter bei der Herstellung
der Polyethylenimine.
Die in einem bestimmten Polyethylenimin oder Gemisch von Polyethyleniminen vorliegenden
Werte für die genannten zahlenmäßigen Verhältnisse der verschiedenen Aminogruppen lassen
sich über 13C-NMR-Spektroskopie bestimmen. Dies wird erläutert in "T. St. Pierre and
M.Geckle, 13C-NMR-Analysis of Branched Polyethyleneimine, J. Macromol. SCI.-CHEM., Vol. A
22 (5 - 7), Seiten 877 - 887 (1985)". The polyethyleneimines suitable as component A for the process according to the invention are branched. That is, the polymer having end groups of formula
The polymer thus contains primary, secondary and tertiary amino groups.
In order for the implementation of the method according to the invention to provide good effects with respect to flame retardant properties of the fiber products, the numerical ratios between the individual amino groups must assume values in a certain range. Thus, in component A, the ratio of the number of secondary amino groups to the number of primary amino groups must be in the range of 1.00: 1 to 2.50: 1, and the ratio of the number of secondary amino groups to the number of tertiary amino groups in the range of 1.20: 1 to 2.00: 1. These numerical values can be controlled via the parameters in the production of polyethyleneimines.
The values for the numerical ratios of the various amino groups present in a particular polyethyleneimine or mixture of polyethylenimines can be determined by 13 C-NMR spectroscopy. This is discussed in T. St. Pierre and M. Geckle, 13 C-NMR Analysis of Branched Polyethyleneimines, J. Macromol SCI.-CHEM., Vol. A 22 (5-7), pages 877-887 ( 1985) ".
Komponente A, die, wie bei Polymeren üblich, normalerweise ein Gemisch von Polymeren ist
und aus Polyethyleniminmolekülen unterschiedlichen Molgewichts und unterschiedlichen Verzweigungsgrades
besteht, besitzt ein Gewichtsmittel des Molgewichts im Bereich von 5.000 bis
1.500.000, vorzugsweise im Bereich von 10.000 bis 1.000.000. Der im Einzelfall vorliegende
Wert für dieses mittlere Molgewicht lässt sich nach Verfahren bestimmen, wie sie aus der Polymerliteratur
bekannt sind, z.B. mittels Gelpermeationschromatographie und Detektion mittels
Lichtstreuung. Hierzu kann man folgendermaßen vorgehen:
Man verwendet als Säule eine oder mehrere "PSS-Suprema"-Typen (beziehbar von "Polymer
Standards Service GmbH", Mainz, DE), die auf den vorgesehenen Molgewichtsbereich eingestellt
sind; Elutionsmittel 1,5%ige Ameisensäure in Wasser; Mehrwinkel-Lichtstreudetektor
MALLS (ebenfalls beziehbar u.a. von "Polymer Standards Service"); ein interner Standard kann
gegebenenfalls zusätzlich verwendet werden.
Die oben und in Anspruch 1 genannten Werte für das Gewichtsmittel des Molgewichts beziehen
sind auf diese Bestimmungsmethode.
Das mittlere Molgewicht von Polyethyleniminen lässt sich steuern durch Variation der Parameter
bei ihrer Herstellung.Component A, which, as is usual in polymers, is normally a mixture of polymers and consists of polyethyleneimine molecules of different molecular weight and different degrees of branching, has a weight average molecular weight in the range of 5,000 to 1,500,000, preferably in the range of 10,000 to 1,000,000. The individual value for this average molecular weight can be determined by methods known from the polymer literature, for example by gel permeation chromatography and detection by means of light scattering. This can be done as follows:
The column used is one or more "PSS Suprema" types (available from "Polymer Standards Service GmbH", Mainz, DE), which are adjusted to the intended molecular weight range; Eluant 1.5% formic acid in water; Multi-angle light scattering detector MALLS (also available from, among others, "Polymer Standards Service"); an internal standard may be used in addition.
The values for the weight-average molecular weight referred to above and in claim 1 relate to this determination method.
The average molecular weight of polyethyleneimines can be controlled by varying the parameters in their preparation.
Eine bevorzugte Ausführungsform des erfindungsgemäßen Verfahrens besteht darin, dass
Komponente A ein
Polyethylenimin ist, das durch Polymerisation von Ethylenimin gebildet wird und das folgende
Struktur aufweist (Formel (V))
wobei die Polymerisation gegebenenfalls sauer katalysiert wird,
wobei die einzelnen Einheiten, welche tertiäre Aminogruppen enthalten und die einzelnen Einheiten,
welche sekundäre Aminogruppen enthalten, beliebig über die Polymerkette verteilt sein
können,
wobei
b größer als a ist und wobei a und b solche Werte besitzen, dass die in Anspruch 1 genannten
Bedingungen für das Molgewicht und für die zahlenmäßigen Verhältnisse der
Aminogruppen untereinander erfüllt sind
oder wobei Komponente A ein Gemisch solcher Polyethylenimine ist.
Wie erwähnt, ist Komponente A normalerweise ein Gemisch von Polyethyleninimen. Die oben
genannte bevorzugte Ausführungsform besteht also im Normalfall darin, dass Komponente A
ein Gemisch von Verbindungen der Formel (V) ist. Die Werte von a und b in den Verbindungen
der Formel (V) müssen natürlich so gewählt werden, dass die an dem Gemisch ermittelten Werte
für die zahlenmäßigen Verhältnisse der einzelnen Aminogruppen zueinander und für das
mittlere Molgewicht in den oben und in Anspruch 1 genannten Bereichen liegen. Die Steuerung
dieser Werte kann, wie erwähnt, über die Parameter bei der Herstellung der Polyethylenimine
erfolgen.
Komponente B ist eine Phosphonsäure der Formel (I), der Formel (II) oder der Formel (III)
A preferred embodiment of the method according to the invention is that component A a
Polyethyleneimine formed by polymerization of ethyleneimine and having the following structure (formula (V)) wherein the polymerization is optionally catalysed acid,
wherein the individual units which contain tertiary amino groups and the individual units which contain secondary amino groups can be distributed as desired over the polymer chain,
in which
b is greater than a and wherein a and b have such values that the conditions given in claim 1 for the molecular weight and for the numerical ratios of
Amino groups are satisfied with each other
or wherein component A is a mixture of such polyethyleneimines.
As mentioned, component A is normally a mixture of polyethylenimines. The abovementioned preferred embodiment thus normally consists in component A being a mixture of compounds of the formula (V). The values of a and b in the compounds of the formula (V) must, of course, be chosen so that the values for the ratios of the individual amino groups to one another and for the average molecular weight determined in the mixture are in the ranges stated above and in claim 1 , The control of these values can, as mentioned, be done via the parameters in the production of the polyethyleneimines.
Component B is a phosphonic acid of the formula (I), of the formula (II) or of the formula (III)
Komponente B kann auch ein Gemisch von Verbindungen sein, die aus Verbindungen der Formel
(I), der Formel (II) und der Formel (III) ausgewählt sind.
In Formel (I) bedeutet R einen linearen oder verzweigten Alkylrest. Dieser Alkylrest enthält für
den Fall, dass der unten genannte Rest R1 eine Hydroxygruppe ist, 1 bis 7 Kohlenstoffatome.
Falls R1 Wasserstoff ist, enthält der Rest R 3 bis 7 Kohlenstoffatome.
Der Rest R1 in Formel (I) steht für H oder OH.
In Formel (I) steht der Rest R2 für den Rest
Component B may also be a mixture of compounds selected from compounds of formula (I), formula (II) and formula (III).
In formula (I), R represents a linear or branched alkyl radical. This alkyl group contains 1 to 7 carbon atoms in the case that the below-mentioned R 1 is a hydroxy group. If R 1 is hydrogen, the radical R contains 3 to 7 carbon atoms.
The radical R 1 in formula (I) is H or OH.
In formula (I), the radical R 2 is the radical
Der Rest R3 in Formel (I) kann für Wasserstoff stehen. Bevorzugt jedoch steht er für einen Rest
R2. Hierdurch lässt es sich erreichen, dass der Gehalt an Phosphor auf dem fertigen Faserprodukt
höher ist als für den Fall R3=H, wodurch im Normalfall ein verbesserter Flammschutz resultiert.
In Formel (II) kann y die Werte 0, 1 oder 2 annehmen. Vorzugsweise besitzt y den Wert 0, was
analog zu dem oben geschilderten Fall in einer Erhöhung des Phosphorgehalts auf dem Faserprodukt
resultiert.
Alle in Verbindungen der Formel (III) anwesenden Reste R4 stehen unabhängig voneinander für
Wasserstoff oder für
oder für einen Rest der Formel (IV)
The radical R 3 in formula (I) may be hydrogen. However, it preferably stands for a radical R 2 . As a result, the content of phosphorus on the finished fiber product is higher than in the case of R 3 = H, which normally results in improved flame retardancy.
In formula (II) y can take the values 0, 1 or 2. Preferably, y has the value 0, which analogously to the case described above results in an increase in the phosphorus content on the fiber product.
All radicals R 4 present in compounds of the formula (III) independently of one another represent hydrogen or or for a radical of the formula (IV)
In dieser Formel (IV) steht t für 0 oder für eine Zahl von 1 bis 10. Vorzugsweise stehen 50 bis 100 % aller anwesenden Reste R4 für In this formula (IV), t is 0 or a number from 1 to 10. Preferably, 50 to 100% of all radicals R 4 present are
Nicht alle in Komponente B vorliegenden Phosphonsäuren müssen in vollständig nicht-neutralisierter Form vorliegen. Vielmehr können in bis zu 50 % der vorliegenden, an Phosphor gebundenen, OH-Gruppen die sauren Wasserstoffatome durch Alkalimetall oder Ammoniumionen ersetzt sein. Vorzugsweise jedoch liegen alle Phosphonsäuren von Komponente B in vollständig nicht-neutralisierter Form vor, sodaß also alle OH-Gruppen in saurer Form vorliegen. Phosphonsäuren der Formeln (I), (II) und (III) sind marktübliche Produkte, z.B. Masquol P 210-1 der Firma Protex-Extrosa oder Briquest 301-50 A der Firma Rhodia oder die Produkte Cublen D50 (Fa. Zschimmer & Schwarz, DE), oder Diquest 2060 S (Fa. Solutia, Belgien). Phosphonsäuren der Formeln (I), (II) und (III) lassen sich nach aus der Literatur allgemein bekannten Methoden herstellen.Not all phosphonic acids present in component B must be completely unneutralized Form present. Rather, in up to 50% of the present, to phosphorus bonded, OH groups, the acidic hydrogen atoms by alkali metal or ammonium ions be replaced. Preferably, however, all the phosphonic acids of component B are complete non-neutralized form, so that all OH groups are present in acidic form. Phosphonic acids of formulas (I), (II) and (III) are commercially available products, e.g. Masquol P 210-1 Protex-Extrosa or Briquest 301-50 A from Rhodia or the Cublen products D50 (Zschimmer & Schwarz, DE), or Diquest 2060 S (Solutia, Belgium). Phosphonic acids of the formulas (I), (II) and (III) can be generally known from the literature Establish methods.
Eine besonders vorteilhafte Ausführungsform des erfindungsgemäßen Verfahrens ist dadurch
gekennzeichnet, dass Komponente B ein Gemisch aus Phosphonsäuren der Formel (I) und der
Formel (II) ist, die beide in vollständig nicht-neutralisierter Form vorliegen.
In einem solchen Gemisch kann das Mischungsverhältnis von Phosphonsäure der Formel (I)
und Phosphonsäure der Formel (II) beliebige Werte annehmen. So kann das Gewichtsverhältnis
der beiden Typen von Phosphonsäure Werte von 0 : 100 bis 100 : 0 annehmen. Gute Ergebnisse
werden beispielsweise erzielt, wenn man als Komponente B ein Gemisch verwendet,
das 70 bis 95 Gew% an einer Verbindung oder einem Gemisch von Verbindungen der Formel
(I) und 5 bis 30 Gew% an einer Verbindung oder einem Gemisch von Verbindungen der Formel
(II) enthält. Besonders günstig ist es, hierbei eine Verbindung der Formel (I) zu verwenden, bei
der
In such a mixture, the mixing ratio of phosphonic acid of the formula (I) and phosphonic acid of the formula (II) may take any values. Thus, the weight ratio of the two types of phosphonic acid can assume values of 0: 100 to 100: 0. Good results are achieved, for example, when using as component B a mixture containing 70 to 95% by weight of a compound or a mixture of compounds of formula (I) and 5 to 30% by weight of a compound or a mixture of compounds of the formula (II). It is particularly advantageous to use a compound of the formula (I) in which
Die Erfindung wird nunmehr durch Ausführungsbeispiele näher veranschaulicht.The invention will now be illustrated by exemplary embodiments.
4,8 kg einer kommerziell erhältlichen wässrigen Lösung (LUPASOL® P, BASF, DE), die 50 Gew% Wasser und 50 Gew% Polyethylenimin enthielt, wurden mit 4,8 kg Wasser und 0,35 kg einer 50 %igen wässrigen Lösung eines hydrolysierten Polymaleinsäureanhydrids gemischt. Das fertige Gemisch enthielt also etwa 24 Gew% Komponente A.4.8 kg of a commercially available aqueous solution (LUPASOL® P, BASF, DE), the 50th % By weight of water and 50% by weight of polyethylenimine were mixed with 4.8 kg of water and 0.35 kg a 50% aqueous solution of a hydrolyzed polymaleic anhydride. The finished mixture thus contained about 24% by weight of component A.
9,2 kg einer wässrigen Lösung, die 40 Gew% Wasser und 60 Gew% einer Phosphonsäure der oben angegebenen Formel (I) (mit enthielt, wurden mit 0,8 kg einer wässrigen Lösung vereinigt, die 50 Gew% Wasser und 50 Gew% einer Phosphonsäure der Formel (II) (mit y = 0) enthielt. Das fertige Gemisch enthielt also etwa 59 Gew% Komponente B.9.2 kg of an aqueous solution containing 40% by weight of water and 60% by weight of a phosphonic acid of the above formula (I) (with were combined with 0.8 kg of an aqueous solution containing 50% by weight of water and 50% by weight of a phosphonic acid of formula (II) (where y = 0). The finished mixture thus contained about 59% by weight of component B.
Es wurden zwei unterschiedliche wässrige Suspensionen hergestellt, welche Cellulosefasern
enthielten (= Suspensionen 1 und 2).
Zur Herstellung von Suspension 1 wurden 10 g Faserrohmasse in 300 g Wasser bei Raumtemperatur
unter Rühren aufgeschlämmt. (Die Faserrohmasse bestand aus etwa 90 Gew% Cellulosefasern
und 10 Gew% Lignin). Anschließend wurde diese Aufschlämmung mit Wasser unter
Rühren auf ein Gesamtgewicht von 1050 g verdünnt.
Zur Herstellung der Suspension 2 wurden 10 g einer Faserrohmasse mit 600 g Wasser unter
Rühren aufgeschlämmt. (Diese Faserrohmasse bestand aus 70 - 75 Gew% Cellulosefasern
und 25 - 30 Gew% Lignin).
Nach der Behandlung mit den Komponenten A und B, die unten beschrieben wird, wurden die
aus den Suspensionen 1 und 2 erhaltenen Produkte folgendermaßen weiterbehandelt:
Zuerst wurden die Produkte über eine Nutsche abgesaugt und dabei abgepresst, wobei bereits
ein erheblicher Teil des Wassers entfernt wurde. Anschließend wurden einige der Proben bei
Raumtemperatur und einem Druck von 35 kp/cm2 3 Minuten lang gepresst und dann 20 Minuten
bei 120°C getrocknet und dann 10 Minuten bei Raumtemperatur konditioniert. Einige andere
Proben wurden nicht bei Raumtemperatur gepresst, sondern bei erhöhter Temperatur. Diese
Proben wurden anschließend nicht mehr getrocknet. An allen so erhaltenen Proben wurde anschließend
das Gewicht bestimmt.
Jeweils mehrere Proben der Suspension 1 und der Suspension 2 wurden vor dem Pressen mit
Komponenten A und B behandelt, wobei die Komponente A in allen Fällen in Form des gemäß
Beispiel 1 a) erhaltenen Gemischs und Komponente B in Form des gemäß Beispiel 1 b) erhaltenen
Gemischs aufgebracht wurde. In allen Fällen wurde Komponente A früher als Komponente
B aufgebracht. Bei manchen Proben wurde zusätzlich noch Di-isooctylphosphat (DIOP) aufgebracht
und zwar zeitlich vor dem Aufbringen von Komponente B.
Außerdem wurde in 2 Fällen (= "Muster 1" und "Muster 2") nur entweder Komponente A oder
Komponente B aufgebracht, die andere der beiden Komponenten wurde nicht verwendet. Die
Muster 1 und 2 sind demnach nicht-erfindungsgemäße Vergleichsproben.
Die nachfolgende Tabelle gibt die verwendeten Mengen an Suspension 1 bzw. Suspension 2,
die verwendeten Mengen an Komponenten A und B und ggf. DIOP wieder, sowie die Bedingungen
des Preß- und Trockenvorgangs und das Gewicht der fertigen Faserplatten. Die in der
rechten Spalte der Tabelle als "BZ" bezeichnete Brennzeit stellt ein Maß für die flammhemmende
Wirkung der beim erfindungsgemäßen Verfahren verwendeten Kombination von Komponente
A und Komponente B dar.
Die "BZ" bezeichnet die Zeit in Sekunden, während der das betreffende Muster noch nachbrennt,
nachdem es 15 Sekunden einer Flamme ausgesetzt war und diese Flamme dann entfernt
wurde.
Ein höherer Wert für "BZ" bedeutet also schlechtere flammhemmende Eigenschaften des Musters.
Two different aqueous suspensions were prepared containing cellulose fibers (= suspensions 1 and 2).
For the preparation of suspension 1, 10 g of fiber raw material were slurried in 300 g of water at room temperature with stirring. (The fiber raw material consisted of about 90% by weight of cellulose fibers and 10% by weight of lignin). Subsequently, this slurry was diluted with water with stirring to a total weight of 1050 g.
To prepare the suspension 2, 10 g of a fiber raw material were slurried with 600 g of water with stirring. (This fiber raw material consisted of 70-75% by weight of cellulose fibers and 25-30% by weight of lignin).
After treatment with components A and B described below, the products obtained from suspensions 1 and 2 were further treated as follows:
First, the products were sucked through a suction filter and pressed while already a significant portion of the water was removed. Subsequently, some of the samples were pressed at room temperature and a pressure of 35 kp / cm 2 for 3 minutes and then dried at 120 ° C for 20 minutes and then conditioned at room temperature for 10 minutes. Some other samples were not pressed at room temperature but at elevated temperature. These samples were then no longer dried. The weight was then determined on all the samples thus obtained.
In each case several samples of suspension 1 and suspension 2 were treated prior to pressing with components A and B, the component A in all cases in the form of the mixture obtained in Example 1 a) and component B in the form of according to Example 1 b) Mixture was applied. In all cases component A was applied earlier as component B. For some samples, di-iso-octyl phosphate (DIOP) was additionally applied, before the application of component B.
In addition, in 2 cases (= "Pattern 1" and "Pattern 2") only either Component A or Component B was applied, the other of the two components was not used. The samples 1 and 2 are therefore noninventive comparative samples.
The following table gives the quantities of suspension 1 or suspension 2 used, the amounts of components A and B used and optionally DIOP used, as well as the conditions of the pressing and drying process and the weight of the finished fibreboard. The firing time referred to in the right-hand column of the table as "BZ" represents a measure of the flame-retarding effect of the combination of component A and component B used in the process according to the invention.
The "BZ" indicates the time in seconds during which the pattern still burns after being exposed to a flame for 15 seconds and then that flame is removed.
A higher value for "BZ" thus means worse flame retardant properties of the sample.
Es ist deutlich zu erkennen, dass die nach dem erfindungsgemäßen Verfahren behandelten Muster 3 bis 7 wesentlich bessere flammhemmende Eigenschaften zeigen als die Muster 1 und 2 (nicht-erfindungsgemäße Vergleichsversuche). Außerdem zeigt ein Vergleich der Muster 6 und 7, dass im Fall höherer Ligningehalte (Suspension 2) in der Fasersuspension ein Zusatz von DIOP eine weitere Verbesserung bringen kann.It can be clearly seen that the treated by the method according to the invention Patterns 3 to 7 show significantly better flame-retardant properties than Patterns 1 and 2 (non-inventive comparative experiments). In addition, a comparison of the patterns 6 shows and 7 that in the case of higher lignin contents (suspension 2) in the fiber suspension an additive from DIOP can bring a further improvement.
Claims (8)
wobei man das Faserprodukt oder vorzugsweise eine Vorstufe davon nacheinander oder gleichzeitig mit einer Komponente A und einer Komponente B behandelt, wobei Komponente A ein verzweigtes Polyethylenimin ist, das primäre, sekundäre und tertiäre Aminogruppen enthält und das ein Gewichtsmittel des Molgewichts im Bereich von 5.000 bis 1.500.000, vorzugsweise 10.000 bis 1.000.000, besitzt und bei dem das zahlenmäßige Verhältnis von sekundären Aminogruppen zu primären Aminogruppen im Bereich von 1,00 : 1 bis 2,50 : 1 und das zahlenmäßige Verhältnis von sekundären Aminogruppen zu tertiären Aminogruppen im Bereich von 1,20 : 1 bis 2,00 : 1 liegt,
oder wobei Komponente A ein Gemisch solcher Polyethyleninime ist,
wobei Komponente B eine Phosphonsäure der Formel (I), (II) oder der Formel (III) ist wobei in den Formeln (I), (II) oder (III) in bis zu 50 % der an Phosphor gebundenen OH-Gruppen das Wasserstoffatom durch ein Alkalimetall oder eine Ammoniumgruppe substituiert sein kann, wobei jedoch vorzugsweise 100 % dieser OH-Gruppen in nicht-neutralisierter Form vorliegen,
oder wobei Komponente B ein Gemisch von Verbindungen ist, die aus Verbindungen der Formel (I), (II) oder (III) ausgewählt sind,
wobei
wobei R3 für H oder R2, vorzugsweise für R2 steht und wobei
alle Reste R4 unabhängig voneinander für H oder für oder für einen Rest der Formel (IV) stehen wobei es bevorzugt ist, wenn 50 bis 100 % aller anwesenden Reste R4 für stehen,
wobei t 0 oder eine Zahl von 1 bis 10 ist.A method of flame-retarding a fibrous product containing from 20 to 100% by weight of cellulose fibers by weight of the anhydrous fibrous product,
wherein the fibrous product, or preferably a precursor thereof, is treated sequentially or simultaneously with a component A and a component B, wherein component A is a branched polyethylenimine containing primary, secondary and tertiary amino groups and having a weight average molecular weight in the range of 5,000 to 1,500 .000, preferably 10,000 to 1,000,000, and in which the number ratio of secondary amino groups to primary amino groups in the range of 1.00: 1 to 2.50: 1 and the numerical ratio of secondary amino groups to tertiary amino groups in the range of 1.20: 1 to 2.00: 1,
or wherein component A is a mixture of such polyethyleninimes,
wherein component B is a phosphonic acid of formula (I), (II) or formula (III) wherein in the formulas (I), (II) or (III) in up to 50% of the phosphorus bound OH groups, the hydrogen atom may be substituted by an alkali metal or an ammonium group, but preferably 100% of these OH groups in non neutralized form,
or wherein component B is a mixture of compounds selected from compounds of formula (I), (II) or (III),
in which
wherein R 3 is H or R 2 , preferably R 2 and wherein
all radicals R 4 independently of one another for H or for or represents a radical of the formula (IV) wherein it is preferred that 50 to 100% of all radicals R 4 present for stand,
where t is 0 or a number from 1 to 10.
wobei die einzelnen Einheiten, welche tertiäre Aminogruppen enthalten und die einzelnen Einheiten, welche sekundäre Aminogruppen enthalten, beliebig über die Polymerkette verteilt sein können,
wobei
b größer als a ist und wobei a und b solche Werte besitzen, dass die in Anspruch 1 genannten Bedingungen für das Molgewicht und für die zahlenmäßigen Verhältnisse der Aminogruppen untereinander erfüllt sind
oder wobei Komponente A ein Gemisch solcher Polyethylenimine ist.A process according to claim 1 or 2, characterized in that component A is a polyethyleneimine formed by polymerization of ethyleneimine and having the following structure (formula (V)) wherein the polymerization is optionally catalysed acid,
wherein the individual units which contain tertiary amino groups and the individual units which contain secondary amino groups can be distributed as desired over the polymer chain,
in which
b is greater than a and wherein a and b have such values that the conditions mentioned in claim 1 for the molecular weight and for the numerical ratios of the amino groups with one another are met
or wherein component A is a mixture of such polyethyleneimines.
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP03027959A EP1538261A1 (en) | 2003-12-05 | 2003-12-05 | Process for flame-proofing of fibrous products |
| CA002541504A CA2541504A1 (en) | 2003-12-05 | 2004-11-25 | Process for the flame-retardant of fiber products |
| EP04819618A EP1697589A1 (en) | 2003-12-05 | 2004-11-25 | Process for the flame-retardant of fiber products |
| AU2004295417A AU2004295417A1 (en) | 2003-12-05 | 2004-11-25 | Process for the flame-retardant treatment of fiber products |
| US10/580,739 US20070082139A1 (en) | 2003-12-05 | 2004-11-25 | Process for the flame-retardant treatment of fiber products |
| PCT/EP2004/013357 WO2005054576A1 (en) | 2003-12-05 | 2004-11-25 | Process for the flame-retardant of fiber products |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP03027959A EP1538261A1 (en) | 2003-12-05 | 2003-12-05 | Process for flame-proofing of fibrous products |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP1538261A1 true EP1538261A1 (en) | 2005-06-08 |
Family
ID=34442966
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP03027959A Withdrawn EP1538261A1 (en) | 2003-12-05 | 2003-12-05 | Process for flame-proofing of fibrous products |
| EP04819618A Withdrawn EP1697589A1 (en) | 2003-12-05 | 2004-11-25 | Process for the flame-retardant of fiber products |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP04819618A Withdrawn EP1697589A1 (en) | 2003-12-05 | 2004-11-25 | Process for the flame-retardant of fiber products |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20070082139A1 (en) |
| EP (2) | EP1538261A1 (en) |
| AU (1) | AU2004295417A1 (en) |
| CA (1) | CA2541504A1 (en) |
| WO (1) | WO2005054576A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006105833A1 (en) * | 2005-04-02 | 2006-10-12 | Huntsman Textile Effects (Germany) Gmbh | Process for the flame-retardant treatment of fiber products |
| CN108642962A (en) * | 2018-05-21 | 2018-10-12 | 浙江鸿浩科技有限公司 | A kind of high temperature resistant paper halogen-free flame retardants |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2680371A1 (en) | 2007-03-15 | 2008-09-18 | Basf Se | Heat-sensitive coating compositions based on resorcinyl triazine derivatives |
| EP2181163B1 (en) | 2007-08-22 | 2011-12-21 | DataLase Ltd | Laser-sensitive coating composition |
| KR20100074334A (en) * | 2007-11-07 | 2010-07-01 | 바스프 에스이 | New fiber products |
| JP5645832B2 (en) | 2008-10-27 | 2014-12-24 | データレース リミテッドDatalase Ltd. | Laser sensitive aqueous composition for marking substrates |
| ITTO20130670A1 (en) * | 2013-08-05 | 2015-02-06 | Torcitura Padana S P A | CELLULOSIC SUBSTRATE WITH ANTI-FLAME PROPERTIES AND RELATED PRODUCTION PROCEDURE |
| CN108586815A (en) * | 2018-05-21 | 2018-09-28 | 浙江鸿浩科技有限公司 | One kind is for polyester carpet phosphorus flame retardant and preparation method thereof |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2632743A (en) * | 1952-01-19 | 1953-03-24 | Armstrong Cork Co | Fire-resistant coating composition for fiberboard and the like |
| GB1450446A (en) * | 1972-12-13 | 1976-09-22 | United Merchants & Mfg | Fire retardant fabrics and method for preparation thereof |
| EP0401538A2 (en) * | 1989-06-07 | 1990-12-12 | Chisso Corporation | Cellulose-based, inflammable, bulky processed sheets and method for making such sheets |
| EP0451665A1 (en) * | 1990-04-12 | 1991-10-16 | Albright & Wilson Limited | Treatment of fabrics |
| EP0542071A1 (en) * | 1991-11-15 | 1993-05-19 | Dr. Wolman GmbH | Wood preservative |
| WO2000029490A2 (en) * | 1998-11-14 | 2000-05-25 | Colloid Surface Technologies Gmbh | Ski coating, compound for producing said coating and method for producing the compound |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2286726A (en) * | 1939-01-06 | 1942-06-16 | Du Pont | Process of flameproofing cellulosic material and products thereof |
| US3398019A (en) * | 1963-02-21 | 1968-08-20 | Monsanto Co | Method for fireproofing cellulosic material |
| US3367828A (en) * | 1964-08-26 | 1968-02-06 | Johns Manville | Hot, wet pressing technique of forming fiberboard |
| US3801333A (en) * | 1968-05-03 | 1974-04-02 | Atlas Chem Ind | Flame retardant and intumescent compositions |
| US3864135A (en) * | 1972-10-13 | 1975-02-04 | Atlas Chem Ind | Flame retardant and intumescent compositions |
| US3963437A (en) * | 1974-04-15 | 1976-06-15 | Cotton Incorporated | Flame retardant process for cellulosic material including cyanamide, phosphonic acid, antimony oxide and polymeric halogen-containing material |
| US4013813A (en) * | 1975-02-27 | 1977-03-22 | Leblanc Research Corporation | Aminoalkylphosphonic acid ester-based textile fire retardants |
| DE3003648A1 (en) * | 1980-02-01 | 1981-08-06 | Basf Ag, 6700 Ludwigshafen | METHOD FOR THE PREPARATION OF WATER-SOLUBLE, NITROGEN-CONDENSING PRODUCTS AND THE USE THEREOF IN PAPER PRODUCTION |
| US4479912A (en) * | 1981-10-20 | 1984-10-30 | Bernadine A. Eggan | Fiber board composition |
| DE4133753C2 (en) * | 1991-10-11 | 1996-03-28 | Budenheim Rud A Oetker Chemie | Process for flame retarding phenol-formaldehyde resin, the resins obtainable thereby and their use |
| DE4244194A1 (en) * | 1992-12-24 | 1994-06-30 | Basf Ag | Water-soluble condensation products from compounds containing amino groups and crosslinking agents, processes for their preparation and their use |
| DE19545874A1 (en) * | 1995-12-08 | 1997-06-12 | Basf Ag | Process for the continuous production of homopolymers of ethyleneimine |
| US6451961B2 (en) * | 2000-02-03 | 2002-09-17 | Nippon Shokubai Co Ltd | Ethylenimine polymer, aqueous solution of ethylenimine polymer, production process for the same and purifying process therefor |
-
2003
- 2003-12-05 EP EP03027959A patent/EP1538261A1/en not_active Withdrawn
-
2004
- 2004-11-25 WO PCT/EP2004/013357 patent/WO2005054576A1/en active Application Filing
- 2004-11-25 AU AU2004295417A patent/AU2004295417A1/en not_active Abandoned
- 2004-11-25 EP EP04819618A patent/EP1697589A1/en not_active Withdrawn
- 2004-11-25 CA CA002541504A patent/CA2541504A1/en not_active Abandoned
- 2004-11-25 US US10/580,739 patent/US20070082139A1/en not_active Abandoned
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2632743A (en) * | 1952-01-19 | 1953-03-24 | Armstrong Cork Co | Fire-resistant coating composition for fiberboard and the like |
| GB1450446A (en) * | 1972-12-13 | 1976-09-22 | United Merchants & Mfg | Fire retardant fabrics and method for preparation thereof |
| EP0401538A2 (en) * | 1989-06-07 | 1990-12-12 | Chisso Corporation | Cellulose-based, inflammable, bulky processed sheets and method for making such sheets |
| EP0599812A2 (en) * | 1989-06-07 | 1994-06-01 | Chisso Corporation | Cellulose-based, inflammable, bulky processed sheets and method for making such sheets |
| EP0451665A1 (en) * | 1990-04-12 | 1991-10-16 | Albright & Wilson Limited | Treatment of fabrics |
| EP0542071A1 (en) * | 1991-11-15 | 1993-05-19 | Dr. Wolman GmbH | Wood preservative |
| WO2000029490A2 (en) * | 1998-11-14 | 2000-05-25 | Colloid Surface Technologies Gmbh | Ski coating, compound for producing said coating and method for producing the compound |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006105833A1 (en) * | 2005-04-02 | 2006-10-12 | Huntsman Textile Effects (Germany) Gmbh | Process for the flame-retardant treatment of fiber products |
| CN108642962A (en) * | 2018-05-21 | 2018-10-12 | 浙江鸿浩科技有限公司 | A kind of high temperature resistant paper halogen-free flame retardants |
| CN108642962B (en) * | 2018-05-21 | 2020-11-24 | 浙江鸿浩科技有限公司 | High-temperature-resistant halogen-free paper flame retardant |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2005054576A1 (en) | 2005-06-16 |
| AU2004295417A1 (en) | 2005-06-16 |
| CA2541504A1 (en) | 2005-06-16 |
| US20070082139A1 (en) | 2007-04-12 |
| EP1697589A1 (en) | 2006-09-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE102005015196A1 (en) | Flameproof finishing of fibrous products, useful particularly for materials of high wool or cellulose content, by treatment with a branched polyethyleneimine and a phosphonic acid | |
| DE3630139C2 (en) | ||
| EP1707665A1 (en) | Process for flame-proofing of fibrous materials | |
| DE2934007A1 (en) | PAPER OR PAPER PRODUCT WITH A HIGH FILLER CONTENT | |
| EP0061073A1 (en) | Fibrous material | |
| DE1546445B1 (en) | Method of making layered synthetic resin laminates | |
| EP1982809B1 (en) | Flame resistant wooden materials | |
| EP1538261A1 (en) | Process for flame-proofing of fibrous products | |
| DE2443074B2 (en) | Flame-retardant thermosetting resin composition and method for its preparation | |
| EP3117032B1 (en) | Flame retardant shaped objects made from cellulose produced by a direct solution process | |
| DE60308537T2 (en) | FLAME PROTECTION TREATMENT SOLUTION AND FLAME-RESISTANT MATERIAL THEREOF | |
| EP2425945A2 (en) | Flame resistant wooden materials | |
| EP0656438A1 (en) | Composition for fire-proofing of fibrous materials | |
| DE1771693B2 (en) | Flameproof paper and a method for its manufacture | |
| DE19709405B4 (en) | Heat insulating material and method for its production | |
| EP0121162B1 (en) | Flame-resistant paper and a process for its manufacture | |
| DE3537241A1 (en) | Flame-inhibiting and formaldehyde elimination-reducing material which can be added to chipboard | |
| DE3245997T1 (en) | MIXTURE OF CHEMICAL COMPOUNDS FOR THE TREATMENT OF CELLULOSE FIBERS CONTAINING OR PRODUCTS COMPOSING THEREOF TO IMPROVE THEIR FIRE RESISTANCE AND METHOD FOR CARRYING OUT THE TREATMENT | |
| EP1160305B1 (en) | Process for the production of a fireproofing agent for combustible materials | |
| EP0025480B1 (en) | Flameproofing agent for paper and carboard, and its use | |
| EP0570435B1 (en) | Process for the production of wood-fibre boards | |
| DE2005589A1 (en) | Process for the production of flame-retardant textiles | |
| EP1229165B1 (en) | Flameproof fleece containing regenerated cellulose fibres | |
| DE2547700C2 (en) | Starch derivative of a starch base and its use | |
| DD234252A5 (en) | EASILY FLAMMABLE STRIPPING PRESSURE BOXES, IN PARTICULAR STRAND PRESSING PLATES, AS WELL AS FLAMMABLE VENEERED OR COATED STRIP PRESSING PLATES, A METHOD OF MANUFACTURING THEM AND THEIR USE |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK |
|
| AKX | Designation fees paid | ||
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20051209 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: 8566 |