EP1077952B1 - Polymerisierbare hydrophobe monomere, die ultraviolettes licht absorbieren - Google Patents
Polymerisierbare hydrophobe monomere, die ultraviolettes licht absorbieren Download PDFInfo
- Publication number
- EP1077952B1 EP1077952B1 EP99922824A EP99922824A EP1077952B1 EP 1077952 B1 EP1077952 B1 EP 1077952B1 EP 99922824 A EP99922824 A EP 99922824A EP 99922824 A EP99922824 A EP 99922824A EP 1077952 B1 EP1077952 B1 EP 1077952B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- hydrogel
- methacrylate
- acrylate
- hydrogels
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 Cc(cc1)cc(*)c1O Chemical compound Cc(cc1)cc(*)c1O 0.000 description 2
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/16—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms condensed with carbocyclic rings or ring systems
- C07D249/18—Benzotriazoles
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F20/00—Homopolymers and copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride, ester, amide, imide or nitrile thereof
- C08F20/02—Monocarboxylic acids having less than ten carbon atoms, Derivatives thereof
- C08F20/10—Esters
- C08F20/34—Esters containing nitrogen, e.g. N,N-dimethylaminoethyl (meth)acrylate
- C08F20/36—Esters containing nitrogen, e.g. N,N-dimethylaminoethyl (meth)acrylate containing oxygen in addition to the carboxy oxygen, e.g. 2-N-morpholinoethyl (meth)acrylate or 2-isocyanatoethyl (meth)acrylate
-
- G—PHYSICS
- G02—OPTICS
- G02C—SPECTACLES; SUNGLASSES OR GOGGLES INSOFAR AS THEY HAVE THE SAME FEATURES AS SPECTACLES; CONTACT LENSES
- G02C7/00—Optical parts
- G02C7/02—Lenses; Lens systems ; Methods of designing lenses
- G02C7/04—Contact lenses for the eyes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S524/00—Synthetic resins or natural rubbers -- part of the class 520 series
- Y10S524/916—Hydrogel compositions
Definitions
- the present invention relates generally to hydrophilic ultraviolet light absorbing monomers. More particularly, this invention relates to ultraviolet light absorbing monomers polymerizable as co-monomers with other polymerizable monomers and co-polymers. In one of its more particular aspects, this invention relates to an ultraviolet light absorbing monomer polymerizable as a co-monomer with other suitable hydrophilic monomers into optically transparent, high refractive index hydrogels which are especially useful in the fabrication of intraocular lenses and contact lenses.
- Optical devices in the form of intraocular lenses and contact lenses have been commercially available for several decades.
- the primary indication for use has been as an adjunct for improving the wearer's visual acuity. This is accomplished by adding or subtracting small amounts of diopter power to the surface of the cornea.
- the contact lens also may have correction for astigmatism.
- a contact lens should be stable at temperatures at, or below, body temperature in an aqueous environment, non-toxic and not contain leachable compounds.
- intraocular lenses With intraocular lenses, the primary indication for use has been for the replacement of the natural crystalline lens of humans and other mammals that were lost to injury and/or cataract formation.
- the natural lens is generally a biconvex lens, from 6 to 13 mm in width, that has considerable optical power, nearly 20 diopters. Therefore, compared to a contact lens, replacing a damaged natural lens requires the use of a substantially larger, thicker, intraocular implant lens.
- an intraocular lens should be stable at body temperature in an aqueous environment, non-toxic and not contain leachable compounds.
- the materials of choice for forming intraocular and contact lenses were the acrylates and methacrylates, particularly polymethylmethacrylate. These materials form rigid, glass-like lenses that are easily shaped to the desired optical correction. These compounds are successful as contact lenses and are generally known as the "hard" contact lenses.
- various hydrogels and elastomeric silicones have been developed that are rollable, foldable or deformable, yet resilient.
- the lens When folded or deformed, the lens may be inserted into the eye through incisions as small as 2-3 mm.
- the resiliency of these materials provides for these lenses to re-assume their original biconvex optical shape after insertion.
- the materials used in these soft lenses have proven to provide optically clear lenses with sufficient indices of refraction, yet are strong or resilient enough to withstand the folding, deformation or rolling processes needed to achieve the smaller incision sizes.
- the folding, deforming or rolling capabilities of these substances, providing for smaller incisions, is a substantial improvement for the patient in terms of reduced trauma to the eye, improved post surgical healing and reduction in complications.
- UV light protection is not the only improvement that has been sought. Another improvement being sought is ultraviolet (UV) light protection.
- UV light absorption for contact and intraocular lenses is at least as important as UV absorption for skin found in sun screens. What amount of UV light protection a native lens provides is lost when it is removed, increasing the risk to the retina from deleterious exposure to UV light if that protection is not restored. UV light protection for the eye may be enhanced by providing UV absorbers in contact lenses or in intraocular lens implants.
- UV light absorbers in hydrogels for use in contact and intraocular lenses pose other problems, as well.
- Optical hydrogels suitable for use in contact or intraocular lenses need clarity, good optical power, stability and resilience. Because of the long term use of contact and intraocular lenses, especially for intraocular lenses, the UV light absorbing compound should stay put within the copolymer. If the UV light absorber leaches out, there is the risk to the surrounding tissue from the chemical exposure. There is also the increasing risk from UV exposure as the UV light absorption capability diminishes over time.
- UV light absorbers for use in hydrogels should be polymerizable as a comonomer in the hydrogel.
- Benzophenone based UV absorbers are polymerizable, but the resultant polymers may not be thermally stable, particularly when hydrated.
- the UV absorbing portion cleaves and leaches out of the polymer.
- UV absorbers are hydrophobic and not very soluble with hydrophilic hydrogel comonomers and copolymers. Even though these compounds are somewhat soluble with hydrophilic comonomers and copolymers, when hydrated within a hydrogel, they tend to coalesce from microphase separation. This coalescence clouds the material rendering it undesirable for use as a lens.
- UV light absorber is the class of phenylbenzotriazoles, such as 2-(2'-hydroxy-5'-methacryloxyethylphenyl)-2H-benzotriazole and its derivatives. These derivatives are polymerizable and stable against hydrolysis, and like the phenol based UV light absorbers, these compounds tend to be hydrophobic. The hydrophobic characteristic leads to microphase separation and clouding.
- the hydrophobicity of the UV light absorber also decreases the amount of water absorption into the hydrogel.
- the decreased water absorption creates a harder, less resilient hydrogel material.
- the amount of UV light absorber is kept to a minimum to make a resilient, optically clear hydrogel.
- current use of hydrophobic UV light absorbers in optically clear material do not produce hydrogels with substantial UV light absorbing characteristics.
- substantial UV light absorption is at least 90% absorption of light at or below 372 nm wavelength.
- US-A-4,526,311 (Beard at al) describes ultraviolet light absorbing polymer compositions comprising copolymers of 2-Hydroxy-5-acrylyloxyphenyl-2H-benzotriazoles with one or more other monomers copolymerizable therewith, particularly acrylic monomers are useful in the manufacture of occular devices, particularly intraoccular lenses and contact lenses.
- Polymers of the disclosed benzotriazoles may be added to other organic materials to impart ultraviolet absorbing properties thereo.
- US -A-4,611,061 (Beard et al)describes that the compounds 2'-hydroxy-5'-(hydroxyalkyl)phenyl-2H-benzotriazoles are useful as intermediate alcohols in the preparation of corresponding 2'-hydroxy-5'-acrylyloxyphenyl-2H-benzotriazole monomers, which in turn are copolymerizable with ethylenically unsaturated monomers, particularly acrylic monomers, to impart ultraviolet absorbing properties to the resulting copolymers.
- US-A-5,384,235 describes a photographic element with particular polymeric ultraviolet absorbers..
- Elements of the invention have good fresh D min and image dye stability, and the ultraviolet absorbers therein are highly light stable EP-A-0,274,844 (Iolab Corp.) describes ultraviolet light absorbing compositions comprising 2-(2'-hydroxy-5'-acryloyloxyalkoxyphenyl)-2H-benzotriazoles, and copolymers with one or more other monomers copolymerizeable therewith. They are useful in the manufacture of ocular devices, particularly intraocular lenses and contact lenses.
- Another object of the present invention is to provide stable increased hydrophilicity UV light absorbing hydrogels having the properties of optical transparency and resiliency while substantially absorbing UV light.
- the present invention provides a monomer as defined in Claim I of the accompanying claims.
- Copolymers formed from monomers of the present invention upon cross-linking and hydration, provide for hydrogels having high optical clarity, high water content, high index of refraction, are stable with good resiliency, and absorb at least 90% of the incident UV light at wavelengths at or below 400 nm.
- the invention thus further provides for homopolymers of monomers as defined in Claim 1 and hydrogels comprising cross-linked copolymers comprising one or a mixture of comonomers including as a comonomer the monomers as defined in Claim 1 and at least one polymerizable comonomer.
- optical clarity shall refer to above 90% light transmission for wavelengths in the visual spectrum.
- the hydrogels are non-toxic and suitable for implantation within living systems.
- these hydrogels are suitable for use as UV light absorbing contact and intraocular lenses, and these are also within the scope of the invention.
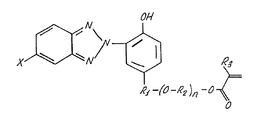
- novel hydrophilic UV light absorbing compositions comprise 2-(2'-hydroxy-5'-acryloxyalkoxyalkylphenyl)-2H-benzotriazoles. These novel hydrophilic UV light absorbing compositions are exemplified by 2-(2'-hydroxy-5'-methacryloxyethyoxymethylphenyl)-2H-benzotriazole.
- the benzotriazole based UV absorbers like other UV light absorbing compositions, are generally hydrophobic and are not soluble in water.
- the exemplary compound, 2-(2'-hydroxy-5'-methacryloxyalkoxyalkylphenyl)-2H-benzotriazole may be synthesized using the corresponding 2-(2'-hydroxy-5'-hydroxyalkoxyalkylphenyl)-2H-benzotriazole reacting with methacryloyl chloride in the presence of pyridine in co-solvents of ethyl ether and dichloromethane at 0-10° C.
- the choices for other comonomers for use in the hydrogels of the present invention may either be derivatives of acrylic acid, such as acrylates, methacrylates, acrylamides or methacrylamides; vinyl-substituted amides; or nitrogen-containing heterocyclic compounds which are substituted with unsaturated sidechains, such as vinyl or acryloyl sidechains.
- Hydrogel materials of the present invention include copolymers formed of at least one hydrophilic or water soluble monomer. Other, additional comonomers may be hydrophobic or hydrophilic. Particular examples are copolymers of various acrylate and acrylamide compounds such as 2-hydroxyethyl methacrylate, N,N-dimethylacrylamide, and N-benzyl-N-methylacrylamide, along with a cross-linking compound such as ethylene glycol dimethacrylate. These compounds are allowed to undergo sufficient cross-linking to hydrate to hydrated equilibrium water contents ranging from about 15% to about 65% and have refractive indices, n D 20 , ranging from 1.41 to 1.52, wet.
- n D 20 refractive indices
- the comonomers are polymerized with from about 1% to about 5% of the hydrophilic polymerizable UV light absorbing comonomers of the present invention, resulting in stable, non-toxic, hydrogels that also exhibit UV light absorption of at least 90% of light at or below 400 nm wavelength. These optically clear UV light absorbing hydrogels are useful in intraocular lenses, contact lenses and related applications.
- the present invention provides novel hydrophilic UV light absorbing polymerizable compositions which provide further for hydrophilic UV light absorbing polymeric materials which are suitable for forming hydrogels.
- the UV light absorbing compositions within the hydrogels are hydrolytically stable and non-leachable.
- Hydrogels of the present invention are optically clear UV light absorbing cross-linked polymers and copolymers. Hydrogels, generally, and processes for their formation are well documented in the literature.
- An exemplary class of UV light absorbing hydrogel-forming polymers includes cross-linked polymers and copolymers which hydrate to a relatively high hydrated equilibrium water content.
- high water content UV light absorbing hydrogels generally have difficulty with microphase separation of the hydrophobic UV light absorbers which dramatically interfere with the optical clarity of the hydrogel. Decreasing the content of hydrophobic UV light absorber may solve the microphase problem, only to substantially decrease the efficacy of the UV light absorption characteristic.
- the hydrogel polymers and copolymers of the present invention have water equilibrium contents of 15% or greater.
- the hydrogel polymers of the present invention have refractive indices of at least 1.41.
- the hydrogel polymers of the present invention have sufficient content of hydrophilic UV light absorbers to provide at least 90% absorption of incident UV light at or below 400 nm wavelength without appreciable loss of optical clarity.
- the present invention provides novel hydrophilic polymerizable UV light absorbing compositions having the general formula 2-(2'-hydroxy-5'-acryloxy(alkyloxy)alkylphenyl)-2H-benzotriazole.
- the alky portion of the compositions are methylene or repeating units of methylene and is hydrophobic.
- the alkyloxy portion of the compound is alkylene oxide, such as ethylene oxide and is hydrophilic.
- the coexistence of the hydrophobic methylene repeating units with the hydrophilic alkylene oxide unit is part of the unique character of this novel class of UV light absorbers.
- Exemplary compounds of the present invention include 2-(2'-hydroxy-5'-methacryloxyethoxymethylphenyl)-2H-benzotriazole and 2-(2'-hydroxy-5'-methacryloxyethoxyethylphenyl)-2H-benzotriazole.
- the exemplary 2-(2'-hydroxy-5'-acryloxy(alkoxy)alkylphenyl)-2H-benzotriazoles can be synthesized from 2-(2'-hydroxy-5'-hydroxy(alkoxy)alkylphenyl-2H-benzotriazole by reaction with methacryloyl chloride in the presence of pyridine in co-solvents of ethyl ether and dichloromethane at 0-10°C.
- 2-(2'-hydroxy-5'-acryloxy(alkoxy)alkylphenyl)-2H-benzotriazole is polymerizable as a comonomer with a wide variety of comonomers to form polymers and copolymers.
- the exemplary hydrophilic comonomers yield hydrogels having high water content, high refractive index n D 20 /n D 37 , and good strength, resiliency and stability.
- 2-(2'-hydroxy-5'-acryloxy(alkoxy)alkylphenyl-2H-benzotriazole provides the hydrogels with a UV light absorption of at least 90% absorption of light at or below 400 nm wavelengths.
- hydrogel forming polymers are cross-linked polymers of water soluble or hydrophilic monomers or copolymers of water soluble and water insoluble monomers.
- cross-linking agents which can be used to produce the hydrogels of the present invention include 1,3-propanediol diacrylate, 1,4-butanediol diacrylate, 1,6-hexamethylene diacrylate, 1,4-phenylene diacrylate, glycerol tris (acryloxypropyl), ether, ethylene glycol dimethacrylate, 1,3-propanediol dimethacrylate, 1,6-hexamethylene dimethacrylate, 1,10-decanediol dimethacrylate, 1,12-dodecanediol dimethacrylate, triethylene glycol dimethacrylate, glycerol trimethacrylate, N,N'-octamethylenebisacrylamide, N,N'-dodecanomethylenebisacrylamide, N,N'-(1,2-dihydroxyethylene)bisacrylamide, allyl methacrylamide, divinylpyridine, 4,6-divinylpyrimidine, 2,5
- Hydrogels prepared using the exemplary 2-(2'-hydroxy-5'-acryloxy(alkoxy)alkylphenyl-2H-benzotriazoles of the present invention have the properties desired for use in a wide variety of applications for UV absorption in hydrogels that require high UV absorption, strength, hydrophilicity and long term stability.
- an exemplary UV light absorbing hydrogel copolymer is 2-hydroxyethyl methacrylate polymerized with N,N-dimethylacrylamide and 2-(2'-hydroxy-5'-methacryloxyethoxymethylphenyl)-2H-benzotriazole cross-linked with ethylene glycol dimethacrylate.
- An additional exemplary hydrogel copolymer is 2-hydroxyethyl methacrylate polymerized with N,N-dimethylacrylamide and 2-(2'-hydroxy-5'-methacryloxyethoxyethylphenyl)-2H-benzotriazole cross-linked with ethylene glycol dimethacrylate.
- a quantity of 180 ml of methylene chloride was poured into a three necked flask containing 30 g of 2-(2'-hydroxy-5'-hydroxyethoxymethylphenyl)-2H-benzotriazole and stirred until starting chemicals were dissolved.
- To this reaction system 13.5 g of pyridine and 120 ml of anhydrous ethyl ether were sequentially added. 15.3 g of methacryloyl chloride in 60 ml of anhydrous ethyl ether were placed in an addition funnel.
- the reaction vessel was cooled over ice water to 0-10°.
- the solution from the addition funnel was dropped, with stirring, into the reaction mixture over 45 minutes. Stirring was continued at this temperature for 2-3 hours.
- the residue was dissolved into 150 ml of ethanol and then cooled in a freezer to -20 to - 45° C for 12 to 24 hours to form a precipitate.
- the white powdery precipitate was filtered quickly under low temperature and dried under vacuum at room temperature. Under ultrasound, the dried powder was dissolved into 150 ml of a mixture of ethanol and methanol (3:2) and filtered to remove remaining impurities yielding approximately 20 g of 2-(2'-hydroxy-5'-methacryloxyethoxymethylphenyl)-2H-benzotriazole.
- Example 1 The procedure of Example 1 was repeated using instead 2-(2'-hydroxy-5'-hydroxyethoxyethylphenyl)-2H-benzotriazole as the starting material as follows.
- a quantity of 180 ml of methylene chloride was poured into a three necked flask containing 30 g of 2-(2'-hydroxy-5'-hydroxyethoxyethylphenyl)-2H-benzotriazole and stirred until starting chemicals were dissolved.
- To this reaction system 13.5 g of pyridine and 120 ml of anhydrous ethyl ether were sequentially added. 15.3 g of methacryloyl chloride in 60 ml of anhydrous ethyl ether were placed in an addition funnel.
- the reaction vessel was cooled over ice water to 0-10°.
- the solution from the addition funnel was dropped, with stirring, into the reaction mixture over 45 minutes. Stirring was continued at this temperature for 2-3 hours.
- the residue was dissolved into 150 ml of ethanol and then cooled in a freezer to -20 to -45° C for 12 to 24 hours to form a precipitate.
- the white powdery precipitate was filtered quickly under low temperature and dried under vacuum at room temperature. Under ultrasound, the dried powder was dissolved into 150 ml of a mixture of ethanol and methanol (3:2) and filtered to remove remaining impurities yielding approximately 20 g of 2-(2'-hydroxy-5'-methacryloxyethoxyethylphenyl)-2H-benzotriazole.
- the following example illustrates the polymerization of 2-(2'-hydroxy-5'-acryloxy(alkoxy)alkylphenyl-2H-benzotriazoles and various other monomers.
- Each polymerization procedure was carried out by first mixing the appropriate amounts of the monomers and cross-linkers with 2,2'-azobisisobutyronitrile as a polymerization initiator. Then each mixture was transferred to an ampule which was pretreated with a trimethylchlorosilane mold releasing agent. Each ampule was then attached to a vacuum system and cooled with liquid nitrogen. After the mixture was frozen, the mixture was placed under vacuum. When a constant pressure was achieved, the vacuum was turned off and the mixture was allowed to thaw, assisted by warming in a water bath. This freeze-thaw cycle was repeated two to four times in order to provide sufficient degassing of the mixture. Finally, each mixture was sealed in the ampule under vacuum or an inert gas, such as nitrogen or argon, and polymerized at a temperature of 60°C for a period of 24 hours, then at 135°C for 10 hours.
- an inert gas such as nitrogen or argon
- each ampule was broken open and the resulting polymer rods were cut into blanks.
- Each blank was then machined to an intraocular lens in its dehydrated state.
- the machined dehydrated lenses had diameters ranging from approximately 6 to 13 mm and central lens thicknesses ranging from approximately 0.5 to 2.0 mm.
- the hydrophilic UV light absorbing monomers of the present invention provide for the creation of UV light absorbing polymers, copolymers and hydrogels. These products are useful in a large number applications under a number of different circumstances.
- the hydrophilic UV light absorbing monomers balance the hydrophobic moieties of the compositions with a hydrophilic moiety while providing for substantial UV light absorption, at least 90%, without loss of optical clarity.
- a higher number of alkylene oxide groups, with fewer methylene groups are used in the UV light absorbers of the present invention.
- the desired optical clarity was achieved through balancing the presence of the hydrophilic moiety necessary to keep the UV light absorber hydrated at higher concentrations of UV light absorber and/or higher water contents against the need to keep the UV light absorber from undergoing appreciable microphase separation.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Physics & Mathematics (AREA)
- Ophthalmology & Optometry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Materials For Medical Uses (AREA)
- Eyeglasses (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Prostheses (AREA)
Claims (15)
- Homopolymer umfassend ein polymerisiertes Monomer nach Anspruch 1.
- Hydrogel umfassend ein vernetztes Copolymer umfassend ein oder eine Mischung von Comonomeren, einschließlich des Monomers nach Anspruch 1 und mindestens eines polymerisierbaren Comonomers als Comonomer.
- Hydrogel nach Anspruch 3, wobei mindestens ein polymerisierbares Comonomer aus der Gruppe ausgewählt wird bestehend aus Acrylaten, Methacrylaten, Acrylamiden, Methacrylamiden, Vinyl-substitutierten Amiden, Vinyl-substituierten Stickstoff enthaltenden heterocyclischen Verbindungen und Acryloylsubstitutierten Stickstoff enthaltenden heterocyclischen Verbindungen.
- Hydrogel nach Anspruch 3, wobei mindestens ein polymerisierbares Comonomer aus der Gruppe ausgewählt ist bestehend aus Methylacrylat, Ethylacrylat, Propylacrylat, Butylacrylat, Amylacrylat, Hexylacrylat, Phenylacrylat, Hydroxyethylacrylat, Hydroxypropylacrylat, Hydroxybutylacrylat, Glycerinmonoacrylat, 2-Phenoxyethylacrylat, 2-N-Morpholinoethylacrylat, 2-(2-Ethoxyethoxy)ethylacrylat, 2-(N,N-Dimethylamino) ethylacrylat, 3- (N,N-Dimethylamino)propylacrylat, Methylmethacrylat, Ethylmethacrylat, Propylmethacrylat, Butylmethacrylat, Amylmethacrylat, Hexylmethacrylat, Furfurylmethacrylat, Hydroxyethylmethacrylat, Hydroxypropylmethacrylat, Hydroxybutylmethacrylat, Glycerinmonomethacrylat, 2-Phenoxyethylmethacrylat, 2-N-Morpholinoethylmethacrylat, 2-(N,N-Dimethylamino)ethylmethacrylat, 3-(N,N-Dimethylamino)propylmethacrylat, 2-Pyrrolidinonylethylmethacrylat, N-Methylacrylamid, N-Ethylacrylamid, N-Propylacrylamid, N-Butylacrylamid, N-Amylacrylamid, N-Hexylacrylamid, N-Heptylacrylamid, N-Octylacrylamid, N-(N-Octadecylacrylamid), 3-N,N-Dimethylamino)propylacrylamid, Allylacrylamid, Hydroxymethyldiacetonacrylamid, N,N-Dimethylacrylamid, N,N-Diethylacrylamid, N-Ethyl-N-methylacrylamid, N-Methylmethacrylamid, N-Methylolmethacrylamid, N-(2-Hydroxypropyl)methacrylamid, N-4-(Hydroxyphenyl)methacrylamid, N-(3-Picolyl)methacrylamid, 3-Vinylpyridin, 4-Vinylpyridin, N-Vinylpyrrolidinon, Vinylpyrazin, 2-Methyl-5-vinylpyrazin, 4-Vinylpyrimidin, Vinylpyridazin, N-Vinylimidazol, N-Vinylcarbazol, N-Vinylsuccinimid, 4-Methyl-5-vinylthiazol, N-Acryloylmorpholin und N-Methyl-N-vinylacetamid.
- Hydrogel nach einem der Ansprüche 3 bis 5, einschließlich eines Vernetzungsmittels ausgewählt aus der Gruppe bestehend 1,3-Propandioldiacrylat, 1,4-Butandioldiacrylat, 1, 6-Hexamethylendiacrylat, 1,4-Phenylendiacrylat, Glycerin-tris(acryloxypropyl) ether, Ethylenglykoldimethacrylat, 1, 3-Propandioldimethacrylat, 1,6-Hexamethylendimethacrylat, 1,10-Decandioldimethacrylat, 1,12-Dodecandioldimethacrylat, Triethylenglykoldimethacrylat, Glycerintrimethacrylat, N,N'-Octamethylenbisacrylamid, N,N'-Dodecanmethylenbisacrylamid, N,N'-(1,2-Dihydroxyethylen)bisacrylamid, Allylmethacrylamid, Divinylpyridin, 4,6-Divinylpyrimidin, 2,5-Divinylpyrazin, 1,4-Divinylimidazol, 1,5-Divinylimidazol und Divinylbenzol.
- Intraokulare Linse, hergestellt aus dem Homopolymer nach Anspruch 2.
- Intraokulare Linse, hergestellt aus dem Hydrogel nach einem der Ansprüche 3 bis 6.
- Hydrogel nach einem der Ansprüche 3 bis 6, das so formuliert ist, dass es im voll hydratisierten Zustand einen Brechungsindex nD37 von 1,41 bis 1,52 aufweist.
- Hydrogel nach einem der Ansprüche 3 bis 6, das einen Gleichgewichtswassergehalt von 15% bis 65% ausweist.
- Hydrogel nach einem der Ansprüche 3 bis 6, wobei das Hydrogel 1 Gew.-% bis 5 Gew.-% des Comonomers 2-(2'-Hydroxy-5'-methacryloxyalkoxyalkylphenyl)-2Hbenzotriazol enthält.
- Hydrogel nach einem der Ansprüche 3 bis 6, das 2-(2'-Hydroxy-5'-methacryloxyalkoxyalkylphenyl)-2Hbenzotriazol enthält und das eine UV-Lichtabsorption von mindestens 90% des in das Hydrogel einfallenden UV-Lichts aufweist.
- Optisch klares Hydrogel, umfassend ein Monomer nach Anspruch 1.
- Optisch klares Hydrogel nach Anspruch 13, das einen Gleichgewichtswassergehalt von mindestens 15 Gew.-% aufweist.
- Optisch klares Hydrogel nach Anspruch 1, das so formuliert ist, dass es einen Brechungsindex nD37 von mindestens 1,41 aufweist.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/075,753 US6036891A (en) | 1998-05-11 | 1998-05-11 | Polymerizable hydrophilic ultraviolet light absorbing monomers |
| US75753 | 1998-05-11 | ||
| PCT/US1999/009961 WO1999058507A1 (en) | 1998-05-11 | 1999-05-05 | Polymerizable hydrophilic ultraviolet light absorbing monomers |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1077952A1 EP1077952A1 (de) | 2001-02-28 |
| EP1077952A4 EP1077952A4 (de) | 2001-08-22 |
| EP1077952B1 true EP1077952B1 (de) | 2003-11-12 |
Family
ID=22127776
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP99922824A Expired - Lifetime EP1077952B1 (de) | 1998-05-11 | 1999-05-05 | Polymerisierbare hydrophobe monomere, die ultraviolettes licht absorbieren |
Country Status (17)
| Country | Link |
|---|---|
| US (1) | US6036891A (de) |
| EP (1) | EP1077952B1 (de) |
| JP (1) | JP4347523B2 (de) |
| KR (1) | KR20010071228A (de) |
| CN (1) | CN1186330C (de) |
| AT (1) | ATE254113T1 (de) |
| AU (1) | AU754155B2 (de) |
| BR (1) | BR9910358A (de) |
| CA (1) | CA2328912A1 (de) |
| DE (1) | DE69912766T2 (de) |
| DK (1) | DK1077952T3 (de) |
| ES (1) | ES2211085T3 (de) |
| NZ (1) | NZ507496A (de) |
| PT (1) | PT1077952E (de) |
| TR (1) | TR200003336T2 (de) |
| WO (1) | WO1999058507A1 (de) |
| ZA (1) | ZA200007285B (de) |
Families Citing this family (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6329485B1 (en) * | 1998-12-11 | 2001-12-11 | Bausch & Lomb Incorporated | High refractive index hydrogel compositions for ophthalmic implants |
| JP4149068B2 (ja) * | 1999-03-02 | 2008-09-10 | 株式会社メニコン | 眼用レンズ材料 |
| US20030223954A1 (en) * | 2002-05-31 | 2003-12-04 | Ruscio Dominic V. | Polymeric materials for use as photoablatable inlays |
| US6737448B2 (en) * | 2002-06-03 | 2004-05-18 | Staar Surgical Company | High refractive index, optically clear and soft hydrophobic acrylamide copolymers |
| US20040056371A1 (en) * | 2002-09-25 | 2004-03-25 | Medennium, Inc. | Method of manufacturing ophthalmic lenses made from hydrophobic acrylic polymers |
| DK1581272T3 (da) | 2003-01-09 | 2006-10-02 | Alcon Inc | UV-absorbere med dobbelt funktion til oftalmiske linsematerialer |
| CN101018513B (zh) * | 2004-08-13 | 2011-11-16 | 渥太华健康研究所 | 视力增强眼科器件及相关方法和组合物 |
| CN102323629B (zh) * | 2004-08-27 | 2015-08-19 | 库柏维景国际控股公司 | 硅水凝胶隐形眼镜 |
| US9322958B2 (en) | 2004-08-27 | 2016-04-26 | Coopervision International Holding Company, Lp | Silicone hydrogel contact lenses |
| JP5264177B2 (ja) * | 2004-11-22 | 2013-08-14 | アボット・メディカル・オプティクス・インコーポレイテッド | 共重合性メチンおよびアントラキノン化合物およびそれらを含有する物品 |
| US20060197067A1 (en) * | 2005-03-04 | 2006-09-07 | Erning Xia | Radiation-absorbing materials, ophthalmic compositions containing same, and method of treating ophthalmic devices |
| US20070092830A1 (en) * | 2005-10-24 | 2007-04-26 | Bausch & Lomb Incorporated | Polymeric radiation-absorbing materials and ophthalmic devices comprising same |
| DE502005011196D1 (de) * | 2005-12-01 | 2011-05-12 | Coronis Gmbh | Polymerzusammensetzung mit hohem Brechungsindex |
| TWI435915B (zh) * | 2007-08-09 | 2014-05-01 | Alcon Inc | 含有吸收紫外光(uv)及短波長可見光二者的發色團之眼科鏡體材料(一) |
| US7915322B2 (en) * | 2008-04-30 | 2011-03-29 | Everlight Usa, Inc. | Polymerizable water-soluble or alcohol-soluble ultraviolet absorber |
| NZ598405A (en) * | 2009-09-15 | 2013-05-31 | Novartis Ag | Prepolymers suitable for making ultra-violet absorbing contact lenses |
| EP2542183A4 (de) * | 2010-03-04 | 2014-04-30 | Perfect Ip Llc | System zur herstellung und veränderung von linsen und damit geformte linsen |
| CN102219877A (zh) * | 2010-04-16 | 2011-10-19 | 品青企业股份有限公司 | 具紫外线吸收官能基的高分子微粒及含该高分子微粒的化妆料 |
| KR101869746B1 (ko) * | 2010-04-29 | 2018-06-21 | 노파르티스 아게 | 자외선 흡수제들 및 청색광 발색단들의 조합을 이용한 인공수정체들 |
| WO2012016097A2 (en) | 2010-07-30 | 2012-02-02 | Novartis Ag | Amphiphilic polysiloxane prepolymers and uses thereof |
| CA2811013C (en) | 2010-10-06 | 2016-01-19 | Novartis Ag | Chain-extended polysiloxane crosslinkers with dangling hydrophilic polymer chains |
| WO2012047961A1 (en) | 2010-10-06 | 2012-04-12 | Novartis Ag | Polymerizable chain-extended polysiloxanes with pendant hydrophilic groups |
| US9187601B2 (en) | 2010-10-06 | 2015-11-17 | Novartis Ag | Water-processable silicone-containing prepolymers and uses thereof |
| CN102382237B (zh) * | 2011-08-12 | 2013-07-10 | 北京自然美光学有限公司 | 一种防辐射滤光隐形眼镜及其制造方法 |
| WO2013055746A1 (en) * | 2011-10-12 | 2013-04-18 | Novartis Ag | Method for making uv-absorbing ophthalmic lenses by coating |
| US20150210651A1 (en) | 2012-08-23 | 2015-07-30 | Bayer Materialscience Ag | Vapour deposition of organic uv absorbers onto plastic substrates |
| US9974646B2 (en) | 2012-09-05 | 2018-05-22 | University Of Miami | Keratoprosthesis, and system and method of corneal repair using same |
| EP2932314B1 (de) * | 2012-12-17 | 2017-02-01 | Novartis AG | Verfahren zur herstellung von uv-licht-absorbierenden brillengläsern |
| EP3689338A1 (de) * | 2014-02-25 | 2020-08-05 | Oculeve, Inc. | Polymerformulierungen zur nasolacrimalen stimulation |
| CA2956678C (en) * | 2014-07-30 | 2023-03-14 | Tiger Coatings Gmbh & Co. Kg | Uv-curable coating composition having improved resistance to sunlight |
| CN111265331B (zh) | 2014-09-09 | 2022-09-09 | 斯塔尔外科有限公司 | 具有扩展的景深和增强的远距视力的眼科植入物 |
| CA2965742C (en) * | 2014-12-16 | 2019-10-01 | Novartis Ag | Low-water content acrylate-acrylamide copolymers for ophthalmic devices |
| KR101704546B1 (ko) * | 2015-06-19 | 2017-02-08 | 연세대학교 산학협력단 | 양친성 고분자를 포함하는 다공성 필름 및 이의 제조방법 |
| WO2017124370A1 (en) * | 2016-01-21 | 2017-07-27 | Rohm And Haas Company | Polymer dispersion for durable coating, and the coating comprising the same |
| JP6953423B2 (ja) | 2016-03-09 | 2021-10-27 | スター サージカル カンパニー | 被写界深度延長及び遠見視力向上を伴う眼科インプラント |
| KR102560250B1 (ko) | 2018-08-17 | 2023-07-27 | 스타 서지컬 컴퍼니 | 나노 구배의 굴절률을 나타내는 중합체 조성물 |
| EP3894909B1 (de) * | 2018-12-10 | 2024-07-31 | Seed Co., Ltd. | Uv-absorbierende augenlinse |
| CN114058142B (zh) * | 2020-08-04 | 2023-05-12 | 浙江省化工研究院有限公司 | 一种透明含氟聚合物薄膜 |
| CN114775279B (zh) * | 2022-06-20 | 2022-09-09 | 河南源宏高分子新材料有限公司 | 一种抗静电阻燃的聚酯材料 |
| DE102022125341B3 (de) | 2022-09-30 | 2024-01-04 | Carl Zeiss Meditec Ag | Ophthalmologische Zusammensetzung mit mehreren Comonomergruppen und ophthalmologische Linse |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NZ208751A (en) * | 1983-07-11 | 1987-04-30 | Iolab Corp | 2-hydroxy-5-acrylyloxyalkylphenyl-2h-benzotriazole derivatives and polymers and copolymers thereof and use as uv absorbing additives in polymer compositions |
| US4528311A (en) * | 1983-07-11 | 1985-07-09 | Iolab Corporation | Ultraviolet absorbing polymers comprising 2-hydroxy-5-acrylyloxyphenyl-2H-benzotriazoles |
| US4611061A (en) * | 1984-03-26 | 1986-09-09 | Iolab Corporation | 2'-hydroxy-5'-(hydroxyalkyl)phenyl-2H-benzotriazoles |
| US5135965A (en) * | 1984-12-18 | 1992-08-04 | T. R. Developments, Ltd. | Hydrogel-forming polymers used in intraocular lenses |
| US4716234A (en) * | 1986-12-01 | 1987-12-29 | Iolab Corporation | Ultraviolet absorbing polymers comprising 2-(2'-hydroxy-5'-acryloyloxyalkoxyphenyl)-2H-benzotriazole |
| US5384235A (en) * | 1992-07-01 | 1995-01-24 | Eastman Kodak Company | Photographic elements incorporating polymeric ultraviolet absorbers |
| JPH08151415A (ja) * | 1994-09-29 | 1996-06-11 | Otsuka Chem Co Ltd | 耐候性組成物 |
| JP3672370B2 (ja) * | 1996-02-29 | 2005-07-20 | 株式会社メニコン | 重合性基を有しない紫外線吸収性ポリマーを含む眼用レンズ材料及びその製造方法 |
| JP3655061B2 (ja) * | 1997-07-31 | 2005-06-02 | 株式会社ニデック | 紫外線吸収性基材 |
| JPH11286569A (ja) * | 1998-04-01 | 1999-10-19 | Nippon Shokubai Co Ltd | 樹脂組成物 |
-
1998
- 1998-05-11 US US09/075,753 patent/US6036891A/en not_active Expired - Lifetime
-
1999
- 1999-05-05 JP JP2000548311A patent/JP4347523B2/ja not_active Expired - Lifetime
- 1999-05-05 AT AT99922824T patent/ATE254113T1/de not_active IP Right Cessation
- 1999-05-05 KR KR1020007012524A patent/KR20010071228A/ko not_active Application Discontinuation
- 1999-05-05 NZ NZ507496A patent/NZ507496A/xx unknown
- 1999-05-05 ES ES99922824T patent/ES2211085T3/es not_active Expired - Lifetime
- 1999-05-05 BR BR9910358-3A patent/BR9910358A/pt not_active Application Discontinuation
- 1999-05-05 PT PT99922824T patent/PT1077952E/pt unknown
- 1999-05-05 WO PCT/US1999/009961 patent/WO1999058507A1/en not_active Application Discontinuation
- 1999-05-05 CA CA002328912A patent/CA2328912A1/en not_active Abandoned
- 1999-05-05 DK DK99922824T patent/DK1077952T3/da active
- 1999-05-05 DE DE69912766T patent/DE69912766T2/de not_active Expired - Lifetime
- 1999-05-05 EP EP99922824A patent/EP1077952B1/de not_active Expired - Lifetime
- 1999-05-05 AU AU39731/99A patent/AU754155B2/en not_active Ceased
- 1999-05-05 CN CNB998060232A patent/CN1186330C/zh not_active Expired - Fee Related
- 1999-05-05 TR TR2000/03336T patent/TR200003336T2/xx unknown
-
2000
- 2000-12-08 ZA ZA200007285A patent/ZA200007285B/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| ES2211085T3 (es) | 2004-07-01 |
| JP4347523B2 (ja) | 2009-10-21 |
| AU3973199A (en) | 1999-11-29 |
| PT1077952E (pt) | 2004-03-31 |
| TR200003336T2 (tr) | 2001-02-21 |
| NZ507496A (en) | 2002-09-27 |
| BR9910358A (pt) | 2001-01-23 |
| ATE254113T1 (de) | 2003-11-15 |
| DE69912766T2 (de) | 2004-09-30 |
| CA2328912A1 (en) | 1999-11-18 |
| US6036891A (en) | 2000-03-14 |
| JP2002514662A (ja) | 2002-05-21 |
| DK1077952T3 (da) | 2004-03-29 |
| EP1077952A1 (de) | 2001-02-28 |
| CN1300283A (zh) | 2001-06-20 |
| CN1186330C (zh) | 2005-01-26 |
| EP1077952A4 (de) | 2001-08-22 |
| AU754155B2 (en) | 2002-11-07 |
| ZA200007285B (en) | 2002-10-08 |
| WO1999058507A1 (en) | 1999-11-18 |
| DE69912766D1 (de) | 2003-12-18 |
| KR20010071228A (ko) | 2001-07-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1077952B1 (de) | Polymerisierbare hydrophobe monomere, die ultraviolettes licht absorbieren | |
| US6673886B2 (en) | High refractive index hydrogel compositions for ophthalmic implants | |
| CA2945961C (en) | (meth)acrylamide polymers for contact lens and intraocular lens | |
| WO2007050394A2 (en) | Polymeric radiation-absorbing materials and ophthalmic devices comprising same | |
| US5135965A (en) | Hydrogel-forming polymers used in intraocular lenses | |
| US20040054026A1 (en) | Elastomeric, expandable hydrogel compositions | |
| US5717049A (en) | High refractive index hydrogels prepared from polymers and copolymers of N-benzyl-N-methylacrylamide | |
| CA1311095C (en) | Hydrogel-forming polymers | |
| JPH09235309A (ja) | 重合性基を有しない紫外線吸収性ポリマーを含む眼用レンズ材料及びその製造方法 | |
| MXPA00011063A (en) | Polymerizable hydrophilic ultraviolet light absorbing monomers | |
| KR100470247B1 (ko) | N-벤질-n-메틸아크릴아미드의중합체및공중합체로제조된고굴절율의하이드로겔 | |
| JPH0651101A (ja) | 光学レンズ用樹脂 | |
| KR20050023312A (ko) | 수분 함량이 낮고 굴절률이 높으며 유연한 중합체 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20001025 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20010710 |
|
| AK | Designated contracting states |
Kind code of ref document: A4 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| RIC1 | Information provided on ipc code assigned before grant |
Free format text: 7C 07D 249/20 A, 7C 08F 120/36 B |
|
| 17Q | First examination report despatched |
Effective date: 20020624 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REF | Corresponds to: |
Ref document number: 69912766 Country of ref document: DE Date of ref document: 20031218 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20040400559 Country of ref document: GR |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 20040130 Ref country code: CH Ref legal event code: NV Representative=s name: KIRKER & CIE SA |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20040617 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: MC Payment date: 20040621 Year of fee payment: 6 Ref country code: FI Payment date: 20040621 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PT Payment date: 20040624 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 20040625 Year of fee payment: 6 Ref country code: DK Payment date: 20040625 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20040630 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2211085 Country of ref document: ES Kind code of ref document: T3 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 20040721 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IE Payment date: 20040722 Year of fee payment: 6 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20040813 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050505 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050505 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050505 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CY Payment date: 20050505 Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050506 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050531 Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050531 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050531 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20050609 Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20051107 |
|
| BERE | Be: lapsed |
Owner name: *PHARMACIA & UPJOHN GRONINGEN B.V. Effective date: 20050531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20051205 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| NLT1 | Nl: modifications of names registered in virtue of documents presented to the patent office pursuant to art. 16 a, paragraph 1 |
Owner name: PHARMACIA GRONINGEN B.V. Owner name: AMO GRONINGEN B.V. |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060505 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060506 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: CD |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20060506 |
|
| BERE | Be: lapsed |
Owner name: *PHARMACIA & UPJOHN GRONINGEN B.V. Effective date: 20050531 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20130507 Year of fee payment: 15 Ref country code: CH Payment date: 20130426 Year of fee payment: 15 Ref country code: DE Payment date: 20130531 Year of fee payment: 15 Ref country code: GB Payment date: 20130425 Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20130531 Year of fee payment: 15 Ref country code: IT Payment date: 20130521 Year of fee payment: 15 Ref country code: NL Payment date: 20130514 Year of fee payment: 15 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 69912766 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20141201 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20140505 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140531 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140531 Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140506 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: EUG |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 69912766 Country of ref document: DE Effective date: 20141202 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20141201 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20150130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140505 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20141202 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140505 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140602 |